

Bestiarium of
the future - Birds |
 |
| In this section descriptions of various animals and plants, which could live on the Earth in Neocene epoch. The section will be supplemented as new ideas about possible ways of evolution of life will appear. If readers will not find here any species placed here earlier, it means that it is a reason to search for a new chapter in English version of "The Neocene Project". |  |
| Birds |
Birds
|
Lilliputian
ostrich (Nanostruthella lilliputiana)
Order: Ostriches (Struthioniformes)
Family: Ostriches (Struthionidae)
Habitat: tropical woods of Zinj Land.

Picture by Pavel Volkov
Initial picture by Alexey Tatarinov |
During a history of order known to people ostriches never flied.
Already at fossil ostriches characteristic adaptation to running, disappearance
of external toes, had been visible. Prehistoric ostriches were rather uniform
group of birds, but they had successfully entered plain ecosystems of any geological
epoch of Cenozoic era. In Pleistocene ostriches had not best times: at first
their variability of species had reduced, and only one species had survived
till historical epoch. And later, in historical epoch, its area and number also
had reduced because of hunting of these birds by people. But ostriches had revealed
the considerable adaptive potential when as the fates decree they appeared free
from the main enemy, the human, and sets of competitors at change of geological
epoch. And in Neocene one African species of ostriches became the highest animal
in the world. But, as if a caprice of destiny, at the Eastern African subcontinent
(or Zinj Land) which had broken away from Africa, its distant relative evolved
in opposite way.
In soils of tropical wood there is lack of mineral substances: they are soaked
up immediately by roots of trees. Therefore it is no wonder, that at apparent
riches of a forest life in underbrush tiny species live, despite of the fact
that their savanna relatives are rather large animals. So there dwarf wood antelopes,
elephants, and even people (pygmies) had appeared. And the tiny representative
of ostriches – the Lilliputian ostrich – lives in tropical forest of Zinj Land
in Neocene epoch. This bird as if sneers at the fact that its ancestor, the
African ostrich, was one of the largest birds of Earth in historical epoch.
Growth of the adult Lilliputian ostrich up to top does not exceed one meter
at weight up to 25 kilograms. This bird has kept typical features of its ancestor
- long naked legs and neck, two toes, underdeveloped wings with long feathers
and a homogeneous feathering on all body.
Living conditions in tropical forest strongly differ from savanna, and the Lilliputian
ostrich has developed some adaptations to such conditions. For successful recognizing
of congeners in twilight of tropical wood at the bird bright spots on the body
had appeared: the head and neck at birds of both genders have caesious color,
and on throat there is a rounded site of red skin – at males it is larger, than
at females. Legs at this ostrich are black, and the feathering at birds of both
genders has identical cryptic colouring: each feather is grey with longitudinal
black strip. When the bird escapes from enemies, it can simply lie on the ground
and keep fixity, becoming imperceptible among shadows and roots. But usually
Lilliputian ostriches run away from the enemy: they accelerate the high speed
and can jump highly, jumping over tabular roots of trees. At their prosecution
along the cross-country terrain the predator lags behind from the first meters
when it could not attack birds suddenly.
The Lilliputian ostrich eats various vegetative foods which can be found in
wood litter: fruits dropping from trees, firm seeds, sprouts of trees. To fray
seeds, the bird searches on riverbanks and swallows small pebbles. Also this
ostrich eats any small animals which may be caught – insects, frogs, tiny mammal.
These birds often leave forest to feed on sandy banks of rivers where they dig
out turtle nests. Sometimes
this ostrich even eats carrion. Due to absence of narrow food specialization
these birds are widely settled in forests of Zinj Land.
At males on wings long feathers grow, but they are not curly as at this bird’s
far ancestor, and have no characteristic white colouring. During courtship demonstrations
the male involves females, opening wings like fans, "waltzing" and
stamping legs by the ground. Thus it loudly cries, and its voice is similar
to goose cackle. During the “serenade” displaying male puffs throat, and it
serves as the resonator. The site of red skin on throat during courtship demonstrations
is increased: it is an attribute of the male maturity. At young birds the red
spot on throat is not present, and they have no success during courtship games
though they start to perform elements of courtship dance, yet not having reached
maturity.
Similarly to all flightless birds this species nests on the ground. In the past
when people had intensively exterminated males of the African ostrich, it had
passed from monogamy to polygamy. The Lilliputian ostrich, of course, had not
undergone to such prosecution, therefore family relations at it represent not
strict monogamy. Usually the male forms family with one female, but sometimes
it allows other female to lay eggs in the nest, and birds look at the common
hatch three together. Outside of nesting season hatches and parents unite to
herds numbering 40 – 50 adult birds. One female lays up to ten rounded eggs
like goose one by size, covered with grey shell with dark points and spots.
The incubating lasts about 40 days Chicks hatch with opened eyes and well developed,
covered with rich down, and abandon nest, having dried under parents. They are
colored yellowish-grey with large longitudinal brown dabs and spots. Approximately
since the second week of life at them the juvenile feathering of grey color
without strips on feathers starts to grow. Head and neck of young birds are
black. By the end of the first year of life young ostriches grow up to 75 –
80 cm, the juvenile feathering is replaced by the adult one, and head and neck
brighten. At the second year of life sites of red skin on the throat appear,
and young birds participate in nesting.
Pyrenean ostrich (Brachystruthio pyrenaicus)
Order: Ostriches (Struthioniformes)
Family: Ostriches (Struthionidae)
Habitat: Western Europe, plains, maquis and woodlands.

Picture by Alexander Smyslov
Due to fast breeding rate ostriches (Struthio camelus) were
among few large inhabitants of African savannas survived in human epoch. The
ability of ostriches
to live in conditions of lack of water has played a considerable role in
their success. Populations of ostriches large enough for a survival of
species were
kept in lonely and uninhabited areas at edges of Sahara, and therefrom new
victorious procession of these birds in Old World after human disappearance.
In savannas of Northern Africa giraffe
ostrich (Deinostruthio altissimus) lives – it is the highest species of ever existing birds. And dwarf
representative of these birds inhabits tropical forests of Zinj Land.
One more descendant of African ostrich, Pyrenean ostrich, was settled to
the north, to Western
Europe. It is adapted to existence in conditions of seasonal climate and
became
one of characteristic species of woodland and plain fauna.
Pyrenean ostrich differs from the ancestor in more thickset constitution
– it is an adaptation to reduction of heat emitting in winter. Growth of
an adult
bird is up to 1.5 m; weight is about 110 kg. This ostrich has heavier constitution
comparatively to ancestor: short thick neck and thick legs. Head of this
species also seems larger and more massive. The lateral toe on legs is reduced
in great
degree, almost useless and only slightly touches the ground. Despite of this
feature, characteristic for quickly running birds, Pyrenean ostrich can accelerate
momentum only about 50 kms per hour. Possible, it descends from quickly running
ancestors, but has lost a part of speed due to development of northern habitats
of colder climate.
The plumage of Pyrenean ostrich is rich and resembles wool more. In feather
colouring the sexual dimorphism is expressed: male is dark grey, but its
neck keeps reddish shade; female is colored lighter – it is brownish with
grey spots
on plumage. Male lacks white feathers in wings characteristic for the African
species. The neck of these birds in bottom part is covered with large feathers
like on body – approximately the half of its general length from shoulders.
The top part of neck and head are covered with thin hairlike feathers.
Beak of this bird is thick and deep. Pyrenean ostrich eats fruits, roots
and grass. With the help of strong hoof-like claws it is capable to dig bulbs
out
of the ground. Occasionally this bird tramples and pecks lizards and small
snakes, and also may eat any rodent.
Similarly to African ostriches prior to the beginning of their extermination,
these birds are monogamous and live in permanently existing pairs or in groups
of three birds; in the south of area male may form family with two females
at once, and they hatch eggs alternately and look after chicks together.
If the herd is larger, it means two or three parents lead the grown up chicks.
The breeding season at this species begins in early spring. The female lays
up to eight eggs, and all members of family incubate the clutch alternately:
male at night, and female in day time. In hot weather female only protects
clutch from the sun. The hatching proceeds till 45 days. Breeding rate at
these birds is rather slow: chicks follow parents more than year and for
this time
they reach approximately 70% of weight of adult individual. Nesting and rearing
of posterity takes away many efforts from adult birds; therefore nesting
at these ostriches repeats once in two years. Young birds become sexually
mature
at the third year of life. Life expectancy reaches 25 – 28 years.
The idea about existence of this species of birds was proposed by Nick, the forum member.
One-toed ostrich (Unistruthio velox)
Order: Ostriches (Struthioniformes)
Family: Ostriches (Struthionidae)
Habitat: Arabia, Asian Near East (Levant), southern border of Mediterranean
hollow.

Picture by Alexey Tatarinov
Ostrich order in human epoch demonstrated expressed enough uniformity of
its representatives. Despite of a wide area, ostriches of Holocene epoch
were representatives
of one genus and several close enough species. Hunting activity of people
and secular changes of climate in Pleistocene had caused great reduction
of number
and area of ostriches. In historical time these birds had completely died
out in Asia, and their number in Africa was reduced many times over. But
ostriches
had rather successfully gone through human epoch – their populations had
been kept in areas where human economic activities had been very complicated.
Their
maintenance in semi-domestic condition had played an important role in preservation
of ostriches.
In early Neocene ostriches appeared among few groups of large vertebrates
in impoverished ecosystems remained after epoch of human domination. It became
a stimulus for their rough evolution, and the various species of ostriches
differing in ecology and way of life had appeared right in first millions
years
after ice age at the boundary of Holocene and Neocene. In Neocene the divergency
inside this group has proceeded, and some genera of ostriches, including giraffe
ostrich (Deinostruthio altissimus), the largest birds species of Neocene,
had appeared.
One genus of ostriches had adapted to fast running and had occupied dry plains
and deserts around of the Mediterranean hollow. This genus includes an only
species – one-toed ostrich.
The anatomy of this species of birds is unique, because on its legs only
one toe, the middle one is well advanced and completely functional. The lateral
toe has not reduced completely, but it is very short, almost completely hidden
under skin. From outside only its claw is seen. The claw of middle toe takes
up weight of body of the bird during walking and run. It is wide and similar
to horse hoof; traces of this species of ostriches have very characteristic
outlines. Growth of adult bird of this species exceeds two meters, and weight
reaches up to 80 kg – a constitution of this ostrich is more gracile, rather
than at its ancestor of Holocene epoch.
One-toed ostrich runs quickly – at the short distance it accelerates momentum
up to 80 kms per hour and can run at speed of about 60 kms per hour till
one hour. Evolution of this species was directed to increase of endurance.
It had
allowed to one-toed ostrich to develop a habitat of very severe conditions
– the Mediterranean hollow.
In due time ancient people compared ostrich to camel because of similarity
of their legs, and this similarity is reflected even in the scientific name
of the ostrich lived on the Earth in human epoch. One-toed ostrich with its
legs resembles the horse more, but it quite can be compared to a camel in
endurance. It can live without water for very long time, receiving metabolic
water from
eaten plants. This ostrich also easily endures very hot weather – its plumage
is rather thin, and head and neck are covered with absolutely naked skin.
On the throat of this bird there is a longitudinal skin plica penetrated
plentifully
with blood vessels and having bright red color. Through it ostrich radiates
the superfluous heat. During the rest this ostrich faces with other problem
– it should lie down on the heated ground or simply on salt crust. But the
bird has the adaptation allowing having a rest this way without harm. On
bird’s tarso-metatarsi there are advanced thick corneous callouses on which
the bird
laying on stomach leans. Moreover, fat layer gathers on stomach of bird,
and from outside stomach is covered with thick cornificated featherless skin.
Color
of plumage also protects this bird from overheat – plumage of birds of both
genders is colored very light, bluish-grey at males and yellowish at females.
Chicks are covered with pale-yellow down with an indistinct longitudinal
strip of grey color on back. At males demonstration feathers on wings are
advanced,
but they are much shorter, than at ostriches of human epoch.
This bird can come far into salty desert of the Mediterranean hollow where
large predators appear only casually. Herds of one-toed ostriches numbering
up to two tens of birds, wander on edges of Mediterranean hollow and in stony
deserts of Near East. This species nests in one of the safest places of this
territory – in island “oases” of the Mediterranean. Mountains serve as places
of condensation of moisture in salt desert, and ostriches find there a favorable
place for nesting. In the beginning of breeding season these birds gather
in herds numbering up to hundred of individuals. They make migrations across
salt
desert to “oases”, moving on firm ground and avoiding salt swamps where the
lost birds perish sometimes.
One-toed ostriches nest on the ground, forming the congestions numbering
some tens of nests. Pairs are formed only to one nesting season. The female
lays
up to ten eggs in primitive nest representing a hole in the ground. Both
birds from pair alternately incubate the clutch irrespectively of time. Chicks
hatch
after 40 days of incubation. They find enough food in bushes and under trees.
They spend the first weeks of life in “oases” but after that the most important
test in life is necessary for them: the grown up young birds make tiresome
migration across salt desert together with their parents. Many chicks lag
behind and are lost in desert where perish soon from heat and lack of food.
Some of
them stick in salty bogs. Strangely enough, but chicks lost in “oases” have
chance to live about the next year and to make the passage across desert
together with adult birds.
Life expectancy of one-toed ostrich makes 20 – 25 years.
In Pleistocene at the islands of Mediterranean Sea there was an original
faunistic complex of dwarf species of elephants, rhinoceroses, hippopotamuses,
deer and
other animals appeared in conditions of isolation when the sea level had risen.
In Neocene the situation repeated in some degree except that the sea has completely
dried up and the extensive spaces of dry saline desert became the main obstacle
for animal settling. At the former islands of the Mediterranean close species
of one-toed ostriches differing in dwarf size and settled way of life live.
Maltese one-toed ostrich (Unistruthio humilis) inhabits
Malta mountain area in the Mediterranean hollow. Growth of this dwarf ostrich
does not exceed 1 meter.
It has a gracile constitution, and its colouring of plumage id darker, rather
than at its large relative: background colouring is pale-yellow, back is dark
grey, and faltering longitudinal strips atretch on each side. Actually, this
species has kept juvenile colouring of its ancestor.
Cretan one-toed ostrich (Unistruthio cretensis) inhabits
mountains being once Crete island. It differs in light grey, almost white plumage
at birds of both
genders, and more robust constitution. Growth of these ostriches is about one
and a half meters, but their legs are rather shorter, than at close species.
This species is original version of “wild goat” among the ostriches, adapted
for movement on hillsides. These ostriches are able to make jumps up to five
meters long and to move on narrow stone eaves.
The complex of indistinctly differed from each other local forms united to
modular
species, dwarf
one-toed ostrich (Unistruthio intermedius), lives
in “oases” of northern part of the Mediterranean hollow. This species is remarkable
in very
wide variability of appearance. Growth at different populations varies from
90 up to 150 cm and is in straight dependence on the size of “oasis” where
they
live. Their constitution, as a rule, is gracile, but the forms close to Cretan
one-toed ostrich have heavier constitution and are less inclined to migrations.
Shade of plumage varies from white and straw-coloured up to beige and light
brown with several longitudinal strips. Between various local forms there are
transitive
variants freely crossed with each other and supporting the certain unity of
this species.
Pampas
giant tinamou (Supercrypturus nandoides)
Order: Tinamous (Tinamiformes)
Family: Tinamous (Tinamidae)
Habitat: pampas of South America, including Patagonia, Chaco; Andean foothills.
Tinamous, or crypturids, have rather successfully gone through human epoch.
Some species and subspecies of these birds had died out in epoch of human domination,
but the most usual species of this group were widely distributed in epoch of
disappearance of forests in South America. Disappearance of rheas, South American
ratites, in human epoch also has urged the evolution of these birds. Clearing
of ecological niche of large ground birds has allowed some descendants tinamou
to increase considerably in size. Reluctantly flying in human epoch, tinamous
had easily left flight ability in order to receive all advantages of ground
way of life.
The most usual representative of these birds is pampas giant tinamou, the inhabitant
of plains overgrown with grass in warm-temperate and subtropical zones of South
America. It is an original analogue of bustards (Otis) of Old World, flightless
bird weighting about 20 kg. In appearance this tinamou resembles a little any
giant chick of emu: it has the remarkable longitudinal-striped colouring, allowing
masking in grass. The basic background of colouring is straw-yellow with individual
variations from yellowish-white up to ochre-red. On back along every feather
shaft the longitudinal dark strip stretches. They form in common the striped
pattern, allowing masking in grass. On wings primarily feathers, on the contrary,
are dark with longitudinal yellow strip in the middle. There are only thin longitudinal
strokes on feathers of neck, breast and stomach.
Head of pampas giant tinamou sharply differs in colouring from other parts of
body. It is black with a cop of several long silky feathers of white color.
Eyes are surrounded with rings of featherless skin of pink color. At displaying
male blood flows to skin in these places, and this skin inflates strongly, turning
red and warty.
Pampas giant tinamou is similar to small ostrich in the constitution. It has
long and strong legs with short toes and thick blunt claws. Growth of this bird
is about 1 meter. Similarly to ostriches of Old World and South American rheas,
this species has kept rather well advanced wings. They are not used for flight:
keel bone at giant tinamous is reduced. But during the run birds use them to
change direction of movement. Chicks of this species, however, still can take
wing and fly short distance; adult birds lose such ability. The tail at this
species is reduced.
This species lives among undersized vegetation: in pampas and bush. This bird
avoids places overgrown with forest. Pampas giant tinamou runs quickly: even
chicks are remarkable in endurance and can deceive the opponent. Using the favourite
tactics of protection of this species, birds sharply change direction of run
and alternate high speed jerks with hiding in grass. Suddenly having hidden
in grass and having freezed for some seconds, bird suddenly jumps and runs,
having changed direction of movement. Birds escape from the enemy this way at
any age. The main enemy of these birds is flightless hunting
heron; obviously, protection tactics of these birds was generated for escaping
from these birds, capable to run for long distances, but lack of maneuverability.
Beyond breeding season pampas giant tinamou lives in herds numbering up to hundred
birds, without the expressed hierarchy. This bird is omnivorous and willingly
eats both greens and seeds, and food of animal origin – lizards, small mammals
and nestlings of ground birds. In rare cases birds peck carrion – it happens
more often on southern border of their area, after severe winters.
Like all flightless birds, pampas giant tinamou nests in pairs. Male displays
itself, extending its body vertically. Calling females, he utters loud buzzing
call, directing the straightened neck forward. When female comes nearer to him,
he begins courtship ritual: opens wings, displaying himself, and pursues in
such pose the female he liked. At this moment rings of skin around of eyes are
poured by blood and swell, giving to him the grotesque shape, and cop on head
rises up. When the female stops as a sign of submission, male folds wings and
cautiously touches her feathers on head by beak. At this time the skin around
of his eyes is still swollen, and cop trembles.
Nest of this species represents simple hole in the ground without any litter.
In clutch of these birds it is about ten eggs in green shell weighting up to
400 grammes. Female lays one egg each day and starts hatching after laying of
the last egg. The hatching lasts till 50 days; usually the female incubates
eggs, but at night male replaces her. Chicks come into light well advanced,
with opened eyes and covered with down. They are capable to walk in two hours
after hatching, and at the first week of life primarily feathers grow at them,
and chicks get ability to fly for a short time. At the age of three years young
bird becomes capable to breeding.
Great
inamu (Noctotinamus solitarius)
Order: Tinamous (Tinamiformes)
Family: Tinamous (Tinamidae)
Habitat: forests of south-eastern part of South America.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. The
genus Noctotinamus, the nocturnal tinamous, is formed by descendants of tinamids
that survived the anthropogenic onslaught. All the representatives of this genus
developed nocturnal habits. The type species of the genus is the great inamu,
an inhabitant of the Atlantic forests of Southeastern South America. Its name
comes from the name used by indigenous people of the Brazilian state of Mato
Grosso to the tinamids.
The great inamu have 53 cm of length and a wingspan of 71 cm. It has a dark
olive-green plumage with a whitish throat and belly. The flanks are barred black,
and the undertail is cinnamon. Crown and neck are rufous with a black supercilium.
The legs are blue-grey in color and the beak is dark grey. They have large eyes
covered with tapetum lucidum that allow them to have a good sight during the
night. While they are capable of flying, these birds rarely do it. They prefer
to run away from the predators or to use their cryptic plumage as camouflage
to hide, with the flight being a last resort.
These birds eats fruit off the ground or low-lying bushes, as well as invertebrates,
flower buds, tender leaves, seeds, and roots.
The great inamu is a shy and solitary tinamou. They are nocturnal, spending
the day perched in trees. Its high pitched vocalization is common to be heard
in the forest at night, but the birds themselves are difficult to be seen. Their
vocalization becomes more frequent during the nesting season, which starts during
the end of the winter and ends in the end of summer. The nests are usually rudimentary
scrapings in the ground, hidden in dense bushes or among buttress roots of trees.
This species is polygynandrous, featuring exclusive male parental care. A female
will mate with a male and lay an average of four eggs which he then incubates
until hatching after 21 days. The eggs are large, shiny, and bright violet in
color. He cares for the chicks for approximately 3 weeks before moving on to
find another female. Meanwhile, the female has left clutches of eggs with other
males. She may start nests with five or six males during each breeding season,
leaving all parental care to the males. Their reach sexual maturity with 13
months and have a lifespan of 17 years.
Another representative of this genus is the
Amazon inamu (Noctotinamus amazonicus). This species inhabits
the "terra firme" forests of the Amazon. This species has 49 cm of
body length and a wingspan of 63 cm. It has a yellowish lower mandible, dark
gray upper mandible, bluish-gray legs, a dark gray crown and neck, with a whitish
throat. The upperparts are slate gray, with black bars. The underparts are lighter
brownish-gray. This species has a longer tail compared with the great inamu,
giving this species more capacity of flight. Their eggs have a bright greenish-blue
shell.
This bird species was discovered by João Vitor Coutinho, Brazil.
False-nandu
(Paranandu pumilus)
Order: Tinamous (Tinamiformes)
Family: Tinamous (Tinamidae)
Habitat: Easter Island.
During the Holocene, the man introduced many species to other continents. Those
that were able to survive left descendants in the Neocene. The false-nandu is
one of them. This species is a large and flightless descendant of the Chilean
tinamou (Nothoprocta perdicaria) which were introduced by the humans in Easter
Island. This species inhabits all areas of the Island, being the largest herbivorous
animal living on it.
As its name says, the false-nandu is similar to the extinct South American rhea.
This species stands at 85 to 90 cm and has a length of 100 to 120 cm. The false-nandu
is sexually dysmorphic. The males are brown above, barred with blackish colouration,
and white below, with a long grey neck and head. Their breast and lower neck
sides are chestnut and there is a pale brown wash to the back. They also have
a black crest that is long, thin, and straight. The breast and neck of the female
are buff, with brown and pale colouration over the rest of the plumage, also
lacking the crests. Immature birds resemble females.
Differently from many flightless birds, the false-nandu maintained its wings.
However, they are small and can't be used to fly. Their wing feathers were also
modified, with the males having fluffy pure white primaries and secondaries,
while in the females they are pale grey. Such fluffy feathers aren’t useful
to fly, being used by the males for courtship.
The tail is very short. They have a gray and curved bill. The legs are pinkish
red and strong, allowing them to run. The feet have long claws, which are used
both to excavate the ground and to defend against a possible predator, or to
be used in territorial fights.
The false-nandu eats seeds, berries, leaves, fruits and small animals. Mostly
of the elements in their diet are collected in low branches or in the ground.
The fruits and seeds are usually those that fell from the trees.
These birds are solitary during most of the year, but in the summer, when the
mating season starts, they form pairs. The male will construct a rough nest
in a semi-sheltered hollow on the ground in the center of his territory, using
bark, grass, sticks and leaves to line it. Then, he will start to vocalize,
as an invitation for the females. They will enter the male’s territory and follow
his calls until reaching the place of the nest. Once he sees her, the male will
flap his wings and sit on the nest with wings open, allowing the female to see
his white feathers here. She will proceed to circle him and the nest, pulling
her neck back while puffing out the feathers and flapping her wings. If she
approves the male, the female will emit a low call to invite him to join her.
Then, he will move closer, stretching his neck and erecting his feathers and
will start to circle the nest with her. If another female appears, the incumbent
female will try to repel them, usually by chasing and kicking.
The pair mate every day or two, and every second or third day the female lays
one of a clutch of 6 to 15 very large, thick-shelled, green eggs. The eggs are
incubated only by the male. During this period he does not eat, drink, or defecate,
and stands only to turn the eggs, which he does about ten times a day. The incubation
period lasts 50 days, during this period the female stays and defends the nest.
When the chicks start hatching, she returns to her territory. Newly hatched
chicks are active and can leave the nest within a few days of hatching; they
will stay with the father until 6 months, when they will disperse to find their
own territory. The lifespan of the false-nandu is 28 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Yagil
(Carnocasuarius yagil)
Order: Cassowaries (Casuariformes)
Family: Cassowaries (Casuaridae)
Habitat: tropical woods of the north of Meganesia.

Picture by Timothy Donald Morris, colorization by Alexander Smyslov
Initial image by Timothy Morris |
North of Australia, New Guinea and islands of Indonesia were
few places at Earth where sites of the rainforest were kept during the ice age.
Some of rainforest inhabitants could go through epoch of climatic changes and
had occupied rainforests widely spreaded in tropical areas after the ice age.
Among them there were not only small creatures like warblers and insects, but
also rather large vertebrates. One of the largest inhabitants of Meganesian
rainforests in early Neocene was the dwarf cassowary (Casuarius benetti). This
bird had survived in epoch of people chasing due to secrecy of habit of life.
But in human era the area of this species had been fragmented, and different
populations had developed different features of behavior. After human extinction
birds had settled again within the borders of former area, but distinctions
in behavior reliably isolated different populations even in common habitats,
and it had become an incitement to speciation.
One of descendants of the cassowary is yagil, rather large bird, up to 170 cm
height, weighting up to 80 kg (female is smaller than male). Yagil had kept
a characteristic for cassowaries constitution: moderately long neck and legs.
But it became less bright, than its ancestor was: the feathering similar to
wool is colored olive tone, on shoulders a little bit darkest, than on sides
and back. In coloring of the bird there are no “excesses”: “lobes” and outgrowths,
characteristic for cassowaries, at yagil are not present, and naked skin on
head and neck is ash-grey. Only under beak of males there is brightly-yellow
spot. Corneous “helmet” on head is small – it looks as small outgrowth sticking
up back. Beak of bird is high, slightly bent down on the tip.
Soft coloring may be clarified simply enough: this bird is a predator, the active
hunter killing various large ground animals. The bird chases prey, imperceptibly
creeping to it hiding in thickets. And as soon as the distance between predator
and prey will be enough shorten, the yagil sharply rushes from shelter on catch.
Its killing instrument is long, powerful and sharp claw on internal toe. The
bird rips up belly or side of prey by it, and then jumps aside, dumping preyed
animal on side by strong push of the body. This bird is named after the hero
of one myth of New Guinea used cassowary leg instead of the weapon. If the victim
had survived after such furious attack, the predatory cassowary will chase it
up to exhaustion. But the bird will not spend superfluous efforts, and it simply
will walk follow the prey, waiting while that will expire with blood.
When the animal is killed, the yagil cleans and tears it apart to pieces by
strong beak. The bird usually eats only soft tissues of large catch, and on
the rests of carcass there is some meat for various scavengers. Except for large
animals the yagil eats small creatures: lizards, rodents and insects.
This predator had kept many features of behavior of cassowary: yagil is the
solitary predator zealously protecting extensive fodder territory. In case of
excitation the yagil involves head in shoulders, fluffs feathers on back and
straightens the rests of wings - the needle-shaped feathers which have turned
into horn spikes even at its ancestor. This bird utters voice seldom, only sometimes
it hisses during conflicts with congeners.
The courtship season at yagils lasts the year round. The female ready to pairing
comes itself into the territory of the male. In the beginning she is cautious,
and keeps at some distance from the male. Then, when he gets used to her presence,
the female starts “close acquaintance”: she leaves to the male right after how
he preys a large animal and is saturated so, that looses interest to catch.
At this time he is less aggressive, and the female can involve him any more
as probable meal: occasionally yagil males attack careless females. When contact
is established, pair in common chooses nesting place, and arranges the nest:
birds simply dig small hole without any litter. In clutch it is up to 10 eggs
having bluish-green shell, they are hatched by the male during 45 days. For
this time he strongly grows thin, as is compelled to eat casual catch, down
to small invertebrates. Constant pair at yagils is not formed, and right after
egg laying the female is expelled from territory of the male.
Young yagils differ in colouring from adults: these birds are spotty, black
with white spots merging to longitudinal lines. Right after hatching they have
“fighting” claw, and they try to attack insects and small vertebrates. Gradually
birds shed feathers, and their colouring becomes one-color, as at adults. Sexual
maturity at yagil comes at the age of about 3 years, at females earlier, than
at males.
This species of birds had been discovered by Simon, the forum member.
Savanna
false moa (Pseudodinornis savannum)
Order: Cassowaries (Casuariformes)
Family: False moas (Pseudodinornitidae)
Habitat: savannas of central and southern parts of Meganesia (Australian-New
Guinean continent).
Neocene epoch differs from droughty and cool Holocene in significant biological
efficiency. The areas of savannas and deserts were reduced and wood and bush
areas had extended. In territory of Australia new habitats able to support numerous
populations of large animals – thickets of eucalyptuses and perennial graminoids
– had appeared. Accordingly, in fauna of this continent appreciable changes
had cased: many large animals had evolved. A part of them was descendants of
introduced species, and the other part had evolved from native species of continent.
Among birds the group of false moas – large flightless descendants of emu –
had appeared. They had occupied all Australian-New Guinean continent, and had
formed some species adapted to different conditions of inhabiting.
In plain districts overgrown with grass, the savanna false moa, the largest
species of this family, lives. Growth of adult bird may reach 350 cm at weight
up to 350 kg. This bird differs in rather massive constitution and thick legs.
Therefore is not able to run quickly. But due to large growth this bird beforehand
notices danger, and life in herd gives advantage of collective defense.
The feathering of savanna false moa is colored sand tone. Feathers are narrow
and more similar to wool. They cover all body in regular intervals, but sides
of head, throat and forward part of neck of this bird are covered with featherless
skin of violet-red color. White coloring of feathers on nape sharply contrasts
with it. These feathers are long and also form thin crest which is used for
submission of signals and recognition of congeners. At males it is larger, than
at females.
At savanna false moa the beak is not specialized to any certain kind of food.
It is natural feature: the bird eats practically everything that is possible
to be found in savanna. The most part of diet of these species includes elevated
parts of grassy plants, but this bird frequently digs out tubers and bulbs by
sharp claws. Also savanna false moa willingly pecks insects and small vertebrates,
down to young rabbits (this bird can swallow animals of such size entirely).
To digest food better, savanna false moas swallow chicken egg-sized stones.
Usually herds of these birds numbering up to 30 - 40 birds wander on savannas,
not adhering to the certain territory. As the need for water at birds is less
than at mammals, they appear on watering places approximately once in two -
three days, using the moisture contained in eaten plants in other time. Due
to this feature birds can feed in districts unsuitable for life of local herbivorous
animals like camelopes. There is no strict hierarchy in herd of birds, and the
competition between males begins mainly during courtship season – shortly before
rain season. During courtship season males start to show aggression to each
other. They start to drive away each other from females, uttering loud blares
and strongly puffing throat.
Savanna false moas are not strict polydins: in various cases one male can form
pair family, or nesting group with several females – it depends on number of
females in herd. Accordingly, the size of clutch of this species varies from
ten up to twenty eggs and more in one nest. The clutch is hatched by both parents
(or male and all females of breeding group) alternately. Chicks hatch after
45 days; they are well advanced, covered with down of yellowish color with narrow
dark longitudinal strips. They can feed independently from the first day of
life. Chicks keep mainly with female, and male protects territory where the
hatch is feeding.
At the age of ten days at chicks feathers start to grow. Young birds till the
first year of life reach about half of weight of adult bird. At three-year age
they start to take part in courtship displays.
False moas have achieved the big success in struggle for existence, that is
expressed in variety and prevalence of these birds in Meganesia. Close species
of this genus inhabit other biotopes:

Picture by Eugeny Hontor
Desert
false moa (Pseudodinornis desertophilum) lives in droughty areas of
south-west of continent. It differs in graceful constitution and ability to
live in hot dry climate.
Mountain
false moa (Pseudodinornis orophilum) lives in completely other conditions,
than the previous species: in mountain woods and bush thickets of Great Dividing
Ridge. It is the smallest species in family: adult individuals grow up to 150
cm in height, and weigh about 100 kg. Birds of this species differ in stumpy
constitution: neck and legs at them are rather short. Mountain false moas do
not differ in run abilities, but they can skip on stones and easily move on
abrupt slopes.
Featherless sites of skin on throat are not present, but at males fluffy “beard”,
which it shows during courtship displays, develops. Feathering of this bird
is rather homogeneous; feathers are narrow, a little similar to wool. To winter
feathers become wider, improving thermal protection of birds. Coloring of body
is soft, brownish-olive tones; head is lighter than body. Legs are feathered
up to middle of hypotarsus, are covered with dark feathers. To winter feathers
on legs extend, forming warm “trousers”.
These birds are steadier against a cold, than local mammal, mountain
camelopes. They live at the greater height, than these animals, and avoid
competition to them due to it. However, to winter mountain false moas of this
species go down in valleys. They eat grass and branches of evergreen bushes.
Mountain false moas are monodins; they nest in rich bush, hiding the nest location.
In their clutch it is to 5 - 8 large eggs.
Wood
false moa (Pseudodinornis sylvaticus) lives in rich tropical woods
at the north of Meganesia. This species is medium-sized representative of the
genus: it reaches only 2 meters in height, and weighs 150 – 200 kg. Its feathering
is colored greyish-green tone with white spots on trunk, imitating light spots
on wood litter. Due to such colouring the bird can remain imperceptible, freezing
in bushes or near the fallen tree. Besides this bird is able to run quickly,
because it is combined rather gracefully, resembling an ostrich by proportions.
For recognition of congeners on throat of this bird there is a bright label
- the blue area of naked skin bordered by silver-gray feathers. Except for this
label, bird is differing in “eyebrows” of lengthened black feathers well distinguished
on grey feathering of head.
Wood false moa is monodin, and pairs at these species are kept till all life.
Male and female build in common nest somewhere in shelter, usually in bush in
the fallen site of wood. The nest represents a small hole in the ground without
litter. In clutch there are about green-shelled 10 eggs. The incubating lasts
about 35 days. Chicks have striped colouring: along back and on sides wide longitudinal
brown stripes stretch on yellowish background.
Striped,
or jarra false moa (Pseudodinornis jarrae) lives in
rarefied eucalyptine woods (this vegetative community is named “jarra”) at coast
of gulf of Eyre and at southern coast of continent. As against other species,
at jarra false moa it is striped colouring of body of alternating brown and
yellow strips, representing a juvenile attribute. Head of bird is black; neck
is white with pale longitudinal strips which become brighter on body.
It is one of large species of genus: the adult male reaches 2,5 - 2,8 m in height
and weighs about 300 kg. Jarra false moa is rather sluggish bird which keeps
in small groups – about 10 – 15 adult birds. It lives in districts overgrown
with bush and undersized wood species of eucalyptus (the characteristic Australian
vegetative community named “jarra”).
Jarra false moa eats eucalyptus leaves – this kind of forage is not accessible
to many herbivores. Leaves of eucalyptus are poisonous enough, and contain a
plenty of oils, therefore only the most specialized herbivores can eat them.
Due to special enzymes this bird neutralizes and decomposes poisonous substances,
and can browse even young sprouts of eucalyptuses. In districts where such birds
are fed, eucalyptuses grow as low trees with “pruned” crone.
Jarra false moas are inclined to social life – birds nest together, and nests
in colony are placed at the distance of several meters from each other. In clutch
there are no more than eight large eggs with shell of brown color. Both parents
hatch it, but the posterity is looked after only by the female. Usually chicks
keep near mother only the first week of life, and later then unite to “kindergarten”,
and females in common look at them. Young birds eat ground vegetation, and start
to use for food eucalyptus leaves only from bi-monthly age.
These species of birds had been discovered by Simon, the forum member.
White
Rumped Swan (Cygnus albicauda)
Order: Anserine birds (Anseriformes)
Family: Ducks and Geese (Anatidae)
Habitat: Waterways and freshwater bodies of Southeastern Meganesia, Murray River
system, mouth of the Murray River, coastal areas of Eyre Gulf.

Picture by Pavel Volkov
During the age of man, certain animals survived by being unpretentious
and even endearing themselves to humans. In Holocene Australia, waterfowl were
a common fixture of public watercourses, and were generally encouraged to settle
there.
And so we see in Neocene Meganesia with the White Rumped Swan, its ancestor,
the Black Swan was a common and favoured animal in any parks which contained
watercourses, and they had mostly escaped persecution.
This bird usually measures about 120 centimetres long and weighs up to 5 kilograms,
wingspan reaches as much as 190 centimetres, they generally resemble their ancestor,
except in colour. The head and neck are a dark black color, graduating into
ashy grey on the shoulders and wings, which also have white patches on the primaries,
the underside, rump and tail are dirty white. The bill is similar to its ancestor
but is an orange-red colour instead of pinkish red, with a white tip.
As with all swans, these birds feed on submerged vegetation, which they reach
on the riverbed by plunging their heads underwater, they can sometimes be found
in coastal shallows where they will feed on seaweed. They may travel in groups
of 6 or 7, and produce a trumpeting call that ends on a higher note. Flies exceedingly
well and can travel long distances between watercourses. This bird is mostly
diurnal but will socialize more in the morning.
As with all swans, they are monogamous, mating for life. Nesting occurs from
March to September, and the pair will build a platform nest standing about 1
meter above the water, this nest can be reused every year. Up to 8 eggs are
laid which are whitish green and these take up to 40 days to incubate. Both
parents take turns to sit on the eggs, and after hatching, the cygnets stay
with their parents for 9 months, at which stage they are fully fledged. The
parents will vigorously defend their young.
Predators include the Bunyip,
as well as large marsupial carnivores. Lifespan is up to 12 years.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Chatham
barocygnus (Barocygnus chathamensis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Chatham islands (to the east from New Zealand), bush thickets.

Picture by Amplion
Before the beginning of human settling the majority of ocean
islands belonged to birds. These creatures, able to overcome sea open spaces
independently, were among the first immigrants at islands and occupied ecological
niches of large vertebrates. At various islands flightless feathered giants
– moas, elephant birds and dodoes – appeared so. Practically at all islands
of tropical and subtropical zone of Earth endemic species of rails and other
flightless birds lived. But in human epoch the situation had sharply changed:
all large islands appeared occupied with descendants of mammals introduced by
people. Flightless birds were exterminated either people, or introduced mammals.
After the disappearance of people species, introduced by them, had continued
the existence and evolution. Thus they had excluded the new occurrence of feathered
giants in their former habitats. But nevertheless in some places flightless
birds appeared even in Neocene. They managed to occupy a leading position in
ecosystems of some islands. For example, at Hawaiian Islands very large geese
have appeared. And in other hemisphere their success had been repeated by the
descendant of related birds – swans.
Chatham Islands are in Pacific Ocean, in rather high latitudes. In Holocene
at them cool and damp climate dominated. In Neocene due to global warming, character
of vegetation of islands had changed. At these islands trees and bushes have
appeared, being mainly of New Zealandian origin. In Holocene due to human mammals
(rodents) had got to islands. They damaged to the local bird's population, but
in Neocene the fauna of islands had replenished with large bird successfully
resisting to them.
In XX century people had introduced the Australian black swan (Cygnus atratus)
to New Zealand. The bird had received “training” in struggle for existence at
the continent successfully superseded local species of ducks and occupied new
habitats. Black swan had gone through ecological crisis, and even began to settle
over New Zealand and nearest islands independently. After human disappearance
black swan had appeared at Chatham archipelago (to the east of New Zealand)
where it became the largest local inhabitant. Due to aggression it successfully
defended nests from rats and cats dwelt at archipelago, and had formed a stable
population. Though there were only few freshwater reservoirs at islands, descendants
of black swan managed to survive. They passed to terrestrial habit of life and
had changed a ration: they began to eat grass and leaves of local bushes. Gradually
the descendant of black swan had turned to dominant species of herbivores of
archipelago.
The former water bird had strongly changed to Neocene. Distinctions between
an ancestor and the descendant appeared significant enough to find the Chatham
species of birds as the representative of separate genus Barocygnus (it literally
means: “heavy swan”). It is very large bird: the height of barocygnus makes
about one and half meters at weight up to 40 kg. It is a sluggish bird unable
neither to run nor to fly. Legs of barocygnus are moderately long, strong, and
with thick toes; the palama between toes is reduced (like at Hawaiian nene).
By constitution barocygnus is a little similar to ostrich: it has long neck
and rather small head. The beak of bird is short and thick; it had lost filtering
device on edges characteristic for ducks. On jaws of bird small corneous denticles
have remained, assisting to browse rigid ground vegetation.
Barocygnus is too heavy for flight. Because at islands there are no large and
fast predators from which it is possible to escape only in flight, it had completely
lost this ability. Wings of barocygnus are strongly reduced, though they have
remained appreciable outside. They are used only for submission of signals to
congeners. The tail is also reduced and presented only by several very short
feathers, almost imperceptible at first sight.
Feathering of barocygnus is black like at ancestor, but wings are completely
white and well appreciable. They are narrow, but primary feathers are strongly
lengthened and very appreciable. Opening wings and flapping by them, barocygnuses
exchange signals with congeners. The beak of barocygnus is brightly marked on
background of rather strict colouring of feathering. It is shining and coral-red.
Legs of bird are covered with black skin.
Barocygnus eats leaves of bushes and seeds of graminoids. Due to rather tall
growth it can eat branches of low trees. Barocygnuses living on ocean coast
can go to coast and to feed with seaweed cast ashore after storm. Also barocygnuses
can eat small rat-sized animals. The large size gives advantage to birds: in
long intestines of bird swallowed plants are digested maximum fully. But to
eat plenty, the bird is compelled to feed almost all day.
Similarly to ancestors, swans, barocygnus lives in pairs which form to all life.
Each pair keeps very amicably and preserves nesting territory against contenders
in common. Protecting the territory, barocygnuses hiss, open wings and stamp
legs, calling synchronously. The voice of barocygnus is similar to loud and
lingering goose cackle. Out of nesting time birds form small herds of adult
birds and their posterity.
Barocygnus nests on the ground, choosing place among bushes well protected from
bad weather. In clutch of this bird it may be only 2 – 3 large eggs with bluish-gray
shell. Both partners hatch clutch and protect it by turns. Birds hatching posterity
are very aggressive to congeners and drive them off by impacts of body and legs.
Birds beat small animals (for example, rodents which can steal their eggs) by
legs.
The incubating lasts approximately 40 days. Nestlings appear quite advanced.
They have opened eyes and also are covered with grey down with black spots.
Parents at once withdraw them from nest and do not come back to it any more.
Young barocygnus becomes fully fledged at the age of about two months, and stays
with parents up to four-monthly age. It differs from parents in colouring: feathering
of young bird has an appreciable brownish shade, and beak is black. Adult birds
are less aggressive in relation to young growth. But when young barocygnus casts
feathers and gets adult colouring, and its beak becomes bright red, it should
test force of impacts of beaks and legs of adult birds on itself.
Barocygnus becomes able to nesting at the age of three years. Life expectancy
of bird makes approximately 40 years.
The idea about the existence of this species was proposed by Simon, the forum member.
Kelp
sea swan (Nereolor algophagus)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Atlantic coast of South America, marshlands and sea coasts in zone
of temperate climate.
For seas of temperate latitudes thickets of brown algae, or kelp, are very characteristic.
They form a special productive zone of shoaliness which can be compared to deciduous
or coniferous forest of temperate latitudes on land. The main feature of algae
is very fast growth. Therefore in coastal waters of South America at the kelp
even huge sea mammals algocetuses
feed. In parallel to them some representatives of anserine birds had adapted
to feeding on algae. Near to family groups of algocetuses, in coastal zone where
these animals swim seldom, sea swans, large herbivorous waterfowl, feed. Despite
of the name, these birds are the special genus of large ducks which are similar
to swans only remotely. Sea swans belong to number of large birds – weight an
adult bird may reach 15 kgs. Long flexible neck pemits such bird to get seaweed
from depth of about half meter, almost not diving. Sea swans of South America
are ecological analogues of flightless algal goose from
northern part of Pacific Ocean. However, as against to it, sea swans have kept
ability to flight. Wingspan of adult bird makes about 3 meters. For rise the
massive bird runs up on surface of water against wind, clapping by wings. Despite
of weight, these birds are good flyers, and are capable to extended migrations.
All sea swans live in areas where seasonal changes of climate are expressed,
and are birds of passage.
Kelp sea swan nests on the ground in places inaccessible to ground predators.
Usually its nesting areas are located on small islets separated from mainland
by strip of fenny sea bogs. At the lack of suitable places for nesting bird
can arrange nest on rocky islets. On small islands where large predators are
absent, kelp sea swans nest in bush, and even simply in forest. This bird is
a social species and forms large colonies in which it may be up to two hundred
of adult birds, and also their posterity of several previous seasons. In case
of occurrence of predator the colony shows to it concurrent resistance.
The voice of kelp sea swan is similar to duck quacking, but is more sonorous.
The annoyed bird hisses.
The inhabiting in sea water had caused changes in physiology of bird. For secretion
of surplus of salt at all species of sea swans very big salt gland above the
beak is developed. It is divided to two symmetric halves which are covered with
corneous cover stretching from beak back. Salt glands look like two smooth corneous
crests sticking out of feathering of head and form similarity of eyebrowes.
These glands open in cavity of nostrils, and secret the surplus of salt as dense
mucous brine when the bird immerses head in water.
Kelp sea swan has monotonously grey colouring of body with blue shade and black
secondary feathers. Black areas of feathering become appreciable when bird stretches
wings or flies. They serve for recognition of congeners, and are used till courtship
games. Bright spots in colouring of bird are blue paws and light yellow horn
cover on beak and salt glands.
Inside colonies birds keep in nesting pairs of approximately identical rank.
The pair forms for some seasons; separate pairs can be kept till tens years,
and each year successfully hatch chicks. Even if the pair is kept till many
years, in courtship season male all the same courts after the female. Courtship
ritual takes place in sea, often far from the coast. During the courtship male
swims around the female, touching to her plumage by beak. He also raises and
slightly opens wings, showing black feathers. The older and stronger male, there
is more black feathers in its wings. Young females not always permit the courting
male to touch their plumage, and the male during the courtship ritual only touches
by beak a surface of water at some distance from the female.
These birds nest on the ground. The nest represents a heap of dry plants which
is raken by the female from above, when she forms the pit of nest. She covers
the bottom of nest with its own down, pulling it out from breast and stomach.
In clutch of kelp sea swan there are only 2 – 3 large eggs (weighting up to
500 grams) with greenish brown shell. Both birds alternately hatch them within
approximately 50 days. Chicks hatch well advanced. They are covered with black
down, and beak at them is white. At chicks the salt gland is already advanced,
and they are able to feed independently. Chicks are exposed to great danger
at feeding in the sea – they are attacked often by fishes seizing them from
below. Therefore chicks, having heard an alarm signal, prefer to get on back
of parents. If danger has overtaken them far from parents, chicks have one more
way of protection against underwater predators. They simply make the same, as
adult birds going to fly up. The escaping chick runs very quickly on sea surface,
splashing on water by webby paws. It can run so up to 9 – 10 meters, and has
an opportunity to reach a back of the parent. But only the smallest chicks can
use such protective reception.
Young birds become able to breeding at the age of three years. The maximal life
expectancy of sea swans makes over 50 years, but the significant part of birds
lives no more than 30 years.
The temperate climatic zone of Southern hemisphere is inhabited by some species
of sea swans. The species closely related to kelp sea swan inhabits coast of
Chile. It is Pacific
sea swan (Nereolor pacificus) which prefers the areas near the current
flowing from polar latitudes of Southern hemisphere (in human epoch it was Humboldt
current). Pacific sea swan has darker colouring, than its Atlantic colleague
– it is dark grey, and covert feathers of wing at this one are almost black.
Legs at these birds are colored grey, and beak is snow-white, clearly visible
on background of plumage. At Tierra del Fuego this species forms mixed colonies
with kelp sea swan, but birds of different species never form a pair, because
they precisely distinguish relatives by voice and plumage. At Pacific sea swan
there is sonorous loud voice, and the congestion of birds feeding in sea is
easily distinguishing even in rich fog. These birds migrate along Pacific coast
of South America, and reach even Galapagos Islands in winter. In diet of Pacific
sea swan there is a significant part the food of animal origin – various invertebrates
with soft covers, and even nonpoisonous jellyfishes.
Antarctic
sea swan (Nereolor frigophilus) lives at coasts of Antarctica, and
flies out for wintering to Patagonia and Tierra del Fuego, replacing there migrated
to the north Pacific and kelp sea swans. The special settled population of this
species lives at Falkland Islands where this bird shares habitats with Falkland
paralgocetus – large herbivorous sea mammal. Earlier the number of Antarctic
sea swan at the coast to Antarctic Region had reached several hundreds thousand
individuals, and they migrated to more northern latitudes of the whole Southern
hemisphere, reaching up to the south of Africa, subantarctic volcanic islands
and the coast of New Zealand. During several thousand years number of this species
had considerably decreased due to the settling of Falkland paralgocetus at the
Antarctic coast. Between these species there is a certain competition for food
resources, but both species coexist rather successfully. The Antarctic sea swan
lives at coasts and eats algae mainly at shoalinesses where large algocetids
can not get. This species nests at sea coast in thickets of high grass.
Antarctic sea swan has plumage of light grey, almost white color. Legs and beak
of bird have yellow color. At males of this species plumage around of eyes forms
black “glasses”.
One species of sea swans had changed the way of life, and had developed productive,
but hard for life inhabitancy – graminoid and sedge meadows of Antarctica. Antarctic
meadow swan (Nereolor pratensis) actually replaces large herbivorous
mammals lack at this continent. Antarctic meadow swan leads terrestrial habit
of life, and is not able to swim at all. It nests in meadows and in bushes of
Antarctic in colonies numbering several tens of birds. Chicks of this species
at once after hatching leave nest and uneven-age herds of birds wander on meadows
of Antarctica, eating leaves of graminoids and sedges, and also sprouts of local
bushes. At this bird palamas are reduced, toes are strong and short, adapted
to walking, and legs are lengthened. In connection with inhabiting on land salt
gland at this bird is only slightly advanced and represents small knob at the
basis of beak of the bird.
Plumage of Antarctic meadow swan is grey with cross strips on feathers. Because
of it its colouring resembles colouring of some breeds of domestic chicken of
human epoch. Head at this bird white, and beak and legs have reddish-orange
color.
The seasonal climate of Antarctica represents the main difficulty for life of
terrestrial inhabitants of this continent. Some of them had adapted to outlive
winters and hide under snow, and others prefer to spend winter time in other
places. Antarctic meadow swan had not lost ability to flight, and easily migrates,
spending winter at the territory of South America.
Antarctic meadow swan nests on the ground. The pair of birds digs not deep pit
in soft ground, which bottom is covered with dry last year's grass. In clutch
of these birds there are 4 – 5 eggs. Chicks of Antarctic meadow swan have striped
colouring – they are grey with several longitudinal black strips on back and
black head. They eat not only grass, but also small invertebrates. Consuming
food rich in protein they grow quickly and to an autumn have time to study to
fly to avoid severe Antarctic winter. Antarctic meadow swans remain in Antarctica
longer, than other birds, and fly out to South America only when the continuous
snow cover is formed on meadows of Antarctica.
Carunculated
swan (Neocygnus carunculatus)
Order: Anserine birds (Anseriformes)
Family: Ducks and geese (Anatidae)
Habitat: North America, freshwater bodies in the area of temperate climate.
The anthropogenic interference in the nature during the Holocene caused the
extinction of several species. These that survived were able to continue their
evolution and their descendants appeared in the Neocene. The Carunculated swan
is the descendant of one of these survivors. This species lives at rivers, lakes,
estuaries and costal lagoons in the temperate regions of North America. The
largest population of this bird is located at the Mishe-Nama Lake, which is
also the Northernmost point of the distribution of this species. When the winter
arrives at the Northern Hemisphere, the carunculated swan migrates to Southeastern
and Southern South America, where it stays in freshwater and brackish habitats.
The great distance covered by it during the migration is a result of the Ice
Age that happened during the end of the Holocene, seeking to flee from the cold,
the ancestor of these birds were forced to migrate to low latitudes, reaching
South America. This migratory habit was passed to its descendants.
As their name says, the main characteristic of the carunculated swan is the
pronounced reddish-pink caruncles at the base of the bill present in the males,
the females lack this. Both sexes have nude reddish-pink faces, which together
with the caruncle become bright red in the males during the mating season. This
species have 130–165 cm in length and 185-250 cm in wingspan. The males are
larger than the females. Carunculated swans are heavy birds, with weight ranging
from 12 to 18 kg. This swan beak is red and its large webbed feet are dark grey,
the same color of the legs. Like many species of swan of the Holocene, the carunculated
swan is totally covered in pure white feathers. The cygnets are grey, and their
bill is dull greyish-black, not red, for the first year.
These birds feed on various plant matter, small aquatic insects, small fish,
fish eggs and small crustaceans. They will eat both the leaves and stems of
submerged and emergent vegetation. To reach the submerged vegetation these birds
use their long necks. They will also dig into muddy substrate underwater to
extract roots and tubers. Carunculated swans use their beak to turn the mud
in search of crustaceans. Grazing in land is more common while in their wintering
areas. They will often feed at night as well as by day. Feeding activity and
the birds' weights increase in the spring as they prepare for the breeding season.
Carunculated swans nest on large mounds that they build with waterside vegetation
in shallow water on islands in the middle or at the very edge of a lake. They
are monogamous and often reuse the same nest each year, restoring or rebuilding
it as needed. The female lays 3–12 eggs and both she and the male share the
care of the nest, and once the cygnets are fledged it is not uncommon to see
whole families looking for food. The incubation period is of 35 days, and the
young are able to swim within two days and usually are capable of feeding themselves
after, at most, two weeks. The fledging stage is reached at roughly 3 to 4 months.
While nesting, these birds are strongly territorial with just a single pair
on smaller habitats, though in a few locations where a large area of suitable
feeding habitat is found they can be colonial. Adults go through a summer moult
when they temporarily lose their flight feathers. The females become flightless
shortly after the young hatch; the males go through this process about a month
later when the females have completed their moult. The youngs will remain with
their parents during the migration and in the wintering grounds, only leaving
them after having returned to the nesting areas.
The carunculated swan is a relatively vocal bird, having high-pitched honking
calls. These calls are more common to be heard while these birds are flying
in their migration, this way the communication between the birds in maintained.
To communicate with the cygnets, the parents make a variety of grunting, hoarse
whistling, and snorting noises. A loud hiss is used to intimidate predators
and conspecifics, who enter the nesting area. If this is not sufficient to drive
off the predator, the swans attack by smashing at their enemy with bony spurs
in the wings, accompanied by biting with their large bill, while smaller waterbirds
are normally grabbed with the swan's bill and dragged or thrown clear of the
swan and its offspring.
The carunculated swan has the ability to grieve for a lost or dead mate or cygnet.
Swans will go through a mourning process, and in the case of the loss of their
mate, may either stay where its counterpart lived, or fly off to join a flock.
Should one of the pair die while there are cygnets present, the remaining parent
will take up their partner's duties in raising the clutch. The carunculated
swan starts to form pairs with 7 years and have a lifespan of 24 years.
The
Florida swan (Neocygnus floridanus) is a close relative of the carunculated
swan. This bird lives in the freshwater reservoirs of the Florida Peninsula,
Cuba and Great Antigua. In comparison to its Northern relative, the Florida
swan is smaller, with 87-115 cm of length and a wingspan of 155-160 cm. The
male is larger than the female, but morphologically they are similar. In both
sexes the beak is totally black. A dark red knob can be seen at the bill base,
which is similar in colour to the bare skin of the face. Although not migratory,
individuals of this species can be seen as vagrants in freshwater habitats in
Mexico.
This bird species was discovered by João Vitor Coutinho, Brazil.
Greater
paradise duck (Paradisanas magnifica)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Far East – Amur basin, Northern China, south of Big Kurils and the
north of Japan Islands; freshwater reservoirs.

Picture by Alexander Smyslov
Damp winds from Pacific Ocean carry huge amount of water to
the coast of Eurasia. They penetrate rather far deep into the continent, and
water which they carry spills as rains even in Mongolia represented the dry
desert in human era. Flatland areas of Far East in Neocene represent a fine
habitat for near-water animals – rains constantly fill rivers and lakes. Coasts
of reservoirs are overgrown with high marsh plants in which the set of species
of birds and other small animals lives.
Among water birds of Neocene epoch the important place is occupied by anserine
birds. Some species of these birds had adapted even to overland
inhabiting and had grew up to huge
size at ocean islands, but the majority of
representatives of this group remained faithful to traditional habit of life,
and settles near water. Some species of anserine birds had suffered in human
epoch from hunting and destruction of habitats. But after extinction of mankind
number of these birds had been restored, and till some millions years new species
had replaced irrevocably dyed out ones.
On the background of other anserine birds the representatives of separate genus
of paradise ducks look like birds of paradise. These birds differing in size
from teal-sized up to small goose-sized ones are endemics of temperate and subtropic
areas of Far East. They differ in beautiful plumage in which bright colors and
metal shine are combined. Various species differ from each other in color of
plumage and presence of “ornaments” of overgrown feathers. But such attributes
are characteristic mainly for males. Females of the majority of paradise ducks
have dimly colored plumage with few elements of “ornaments”, only remotely similar
to magnificent plumage of drakes.
Greater paradise duck, the largest representative of genus (equal to small goose
by size), simultaneously is the most northern species of these birds. In the
early spring it flies from wintering and up to middle of an autumn is the true
gem of reservoirs in its habitats. In colouring of drake of this species white
color prevails – the body of bird and the most part of wings is colored so.
On covert feathers of wings there are black tips forming a pattern of faltering
black cross strips. Primal feathers have crimson pink color, and are clearly
marked out at flying bird. Some middle pairs of quill feathers are very much
lengthened, and because of it the tail of bird resembles pheasant’s tail a little.
These feathers have white color with black cross strokes. Paradise ducks have
one more special “ornament” in plumage. On shoulders of these birds the long
narrow feathers forming some kind of “shoulder-straps” grow. Vanes of these
feathers are friable, barbs are not linked, and shafts are thin and flexible.
During the courtship displays drake lifts them vertically, and they flutter
from wind. At the female “shoulder-straps” are shorter, than at male.
The head of drake is colored bright – it is orange with silky shine, and from
forehead to nape the strip of shining cyan feathers stretches. In courtship
season male displays to females the fluffed cop sparkling with blue sparks.
At the background of bright head of male black beak with white tip is contrastly
marked.
Colouring of the female is considerably duller. The female of greater paradise
duck is grey with dark striped pattern on wings. On breast and neck on tips
of feathers there are dark spots, therefore the colouring of these parts of
body is speckled. On nape and back of the female feathers are bordered by black
strip, and colouring of the top part of body of bird looks “scaly”. Due to not
striking coloring the female of greater paradise duck can easily hide from predators
in time of clutch hatching. It would be difficult to recognize her as a partner
of magnificent male with sparkling head, but the body shape indicates it. On
head of the female of greater paradise duck there is mobile cop, and feathers
of tail are as lengthened and peaked, as at male, and have the same striped
pattern, but on grey background.
This bird eats plants, and also small aquatic invertebrates – snails and water
insects. Due to long neck birds of this species can get food from greater depth,
rather than other ducks, and thus they avoid a competition to them.
Beautiful males of paradise ducks get in claws and teeth to predators more often,
than females. Therefore at paradise ducks in population females prevail, and
males are inclined to polygamy - usually at the territory of strong male two
females nest.
Greater paradise duck nests in shelters. More often it arranges nest in reeds,
but sometimes it can nest at some distance from water, under roots of trees,
in trunks of trees rotten from within, or in holes of any large mammals which
do not represent danger to clutch. The female builds nest of thin rods and grasses,
and covers the bottom by its own down and thin leaves of grass. In clutch of
greater paradise duck there is about 12 small eggs with grey green shell. At
these birds exclusively the female incubates eggs till 32 – 33 days. Ducklings
hatch active and mobile, covered by grey down with longitudinal black strips
on back and head. Having dried under the female, they at once leave nest. Male
because of bright plumage is too visible for predators, and first some days
of life ducklings almost do not see him. At this time male keeps at the edge
of territory of nesting pair, protecting borders from neighbours and withdrawing
predators from hatch. Only when ducklings become strong enough and hardy to
escape from danger, he joins a family.
The first feathering of young birds is similar to plumage of the female. Young
birds have such plumage within the first year of life. Only after the first
mew males get characteristic bright colouring.
At the Far East other species of paradise ducks live:

Picture by Alexander Smyslov
Butterfly-faced paradise duck (Paradisanas papiliofrons)
is a small bird similar by size to domestic duck. It has received the name because
of features of appearance and ritual of display. Drake is bluish-grey with bright
green “mirrors” on wings; duck has brown plumage with small specks on covert
feathers. In tail of birds of both genders only two middle feathers are lengthened.
On head at birds of both genders the double cop of white color with black edges
of feathers grows. At drake on each side of beak there are large feather “whiskers”
of the same color, as a cop. But on these feathers it may be more of black color,
and at separate males “whiskers” are completely black or have only few white
spots in the basis. The displaying drake stretches feather “ornaments” in sides,
and feathers form the figure similar to stretched wings of butterfly.
This species lives at Big Kurils, Japan Islands and Pacific coast of Asia in
area of subtropical climate.

Picture by Alexander Smyslov
Necktied paradise duck (Paradisanas focalefera)
is similar in colouring to the previous species, but lives at the south – at
Korean Peninsula and in adjoining areas of China. It has another type of feather
ornaments: from the bottom jaw down to breast of drake the strip of feathers
of black color with green metal shine lasts. Besides on neck of drake there
are some freely hanging down lengthened feathers of the same color. The displaying
bird bends neck back, puffs throat, and feathers of “tie” stick upwards. When
drake utters an advertisement call, these feathers tremble and shine in sunlight.
Tail at birds of both genders is fanlike; at the male tail feathers are white
with brown tips. Displaying male lifts and unwraps tail like a fan. Feathers
on waist of male have blue color with metal shine. Till the courtship displays
they fluff and brightly shine, shading strict colouring of tail. “Shoulder-straps”
have white colouring; at the display male stretches them in sides, and they
shudder at each movement of bird.
 |
 |
Picture by Alexander Smyslov
Cuckoo paradise duck (Paradisanas parasitica)
differs from other species of these birds in some features of appearance and
way of life. At these birds male and female are colored equally brightly – they
are rusty with brownish wings, white fluffy “shoulder-straps” and dark blue
head with greenish metal shine which sharply contrasts with the general colouring
of bird. The female differs from the male only by darker wings and completely
black beak (at drakes beak has bone color with black tip). This feature is closely
connected to nesting behaviour of bird: cuckoo paradise duck does not build
its own nests, and only lays eggs in nests of other ducks – as of the same genus,
as of less related ones. Such feature of behaviour was more or less characteristic
for many ducks of Holocene epoch, and has reached the top at South American
duck Heteronetta which has completely passed to nest parasitism. In Neocene
the cuckoo paradise duck had independently repeated the same step of evolution
and consequently does not require masking colouring.
Before the sexual maturity young birds of this species have the modest order
– brown with black tips of feathers on wings and black primal feathers. Extremely
seldom sexually mature females of cuckoo paradise duck keep such plumage till
all further life. Presumably, before the transition to nest parasitism females
of the ancestral species had been colored similarly, and then as a result of
mutation at them the colouring incorporated in male genes, which the female
should transfer to posterity, simply began to express. Because of bright colouring
the female is compelled to be very cautious, laying eggs to other ducks. Usually
it tracks down for a long time ducks which may become foster mothers for its
posterity, and remembers the location of their nests in coastal thickets. The
bird chooses the best hidden nests for egg laying, and bright colouring does
not cause any obstacle for egg laying to another’s nests. Besides at the moment
of laying of eggs the female of cuckoo paradise duck is very cautious.
It is a small species of ducks, and this circumstance permits the cuckoo paradise
duck to be the nest parasite at the much greater number of species of ducks
living in the neighbourhood. The cuckoo paradise duckinhabits Japan Islands
and subtropical areas of China, and also the south of Korean Peninsula.
Dwarf
paradise duck (Microparadisanas lemnivora)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: the south of Japan Islands, forest marshes and rich thickets of swamp
plants.

Picture by Alexander Smyslov
As a rule, development of fantastical “decorations” of feathers,
skin or hair at various species of animals is a result of sexual selection.
At ducks sexual selection was expressed, first of all, in apparent sexual dimorphism:
drakes of the majority of duck species appreciably differ from females. At Japan
Islands one of the most colorful ducks of Neocene epoch – paradise
ducks distinguished by especially bright colouring – live. The majority
of paradise ducks has rather large size, but among them there is one tiny species
– dwarf paradise duck, the representative of the separate genus of these birds,
related to typical paradise ducks. This species is very tiny bird: the body
length of adult duck does not exceed 15 cm. Because of small weight it dives
not well, but flies perfectly. Flight of dwarf paradise duck is maneuverable
and fast; escaping from feathered predator, the bird sharply changes direction
of flight, therefore it is very difficult for preying, even if it is well appreciable.
Flying up above reeds, dwarf paradise duck sparkles in sunlight by bright feathering
of head.
Characteristic feature of this bird is the presence of strip of bright crimson
feathers with red metal shine on head. These feathers form the mobile crest
similar to crest of cockatoo parrot. At drake head is much brighter, rather
than at female: on each side of the head at it feathers with blue metal shine
grow. At females head is brown from sides. In courtship display time drake fluffs
feathers on head and involves female with their shine in sunlight, trembling
up head. The head of displaying drake at this moment turns to sparkling fluffy
sphere of feathers.
In colouring of the body of male and female there is an expressed sexual dimorphism.
The body of male is colored grey with silvery metal shine on wings. Edges of
covert feathers of wings have black border with dark blue metal shine, and primary
feathers are coal-black. The female has black feathering on the top side of
a body; sides and stomach are colored white, and on back under wings there is
an area of cream colored feathering. In tail of male middle pair of feathers
is lengthened; in courtship display time drake raises tail vertically, showing
these feathers. Paws at birds of both sexes are bright yellow.
Despite of bright colouring it is the extremely difficult to notice these birds
in their habitat. They live in marshland in small lakes surrounded with a zone
of bogs and almost impassable thickets of reed and other coastal plants. Dwarf
paradise ducks are very reserved and at occurrence of predators hide among water
plants. Due to small size they are able to hide among reeds dexterously. On
their wings there is special corneous spur, with which help ducks can cling
to stalks of reed by wings. Main enemies of dwarf paradise ducks are water snakes
easily chasing these ducks among reed thickets. Also these tiny birds may be
attacked by fishes, and ducklings are eaten by large frogs.
Dwarf paradise duck eats mainly the duckweed. This food source is widely distributed
in nature and differs in high rate of renewal; therefore dwarf paradise ducks
concern to congeners rather tolerantly. Out of nesting season they gather in
reservoirs to small groups and feed in common. Also this bird willingly eats
forage of animal origin: tiny snails, worms, leeches. Because of small size
dwarf paradise duck eats rather large quantity of forage – for day it eats the
amount of forage approximately equal to half of its body weight. The forage
of animal origin makes in its diet the bigger share, than at other species of
ducks: it is connected to fast growth and high rate of metabolism at small bird.
Dwarf paradise duck does not dive: feathering serves as a good float to it.
Therefore it gathers food only from surface of water. Birds are able to catch
larvae of mosquitoes dexterously, and hunt for them even among thickets of floating
water plants. These tiny ducks are one of main fighters of mosquitoes in swamps
of Japan Islands.
This bird arranges nest in reed thickets, choosing sites of swamps inaccessible
to ground predators. Frequently it occupies old nests of warblers, having repaired
them a little. Dwarf paradise duck breeds very quickly: the incubating lasts
about 2 weeks, and for one nesting season female has time to make two hatches.
Daily ducklings of this species are tiny creatures equal to large bumblebee
in size, covered with yellow down, with black longitudinal strip on back and
black head. They at once leave nest, and do not come back to it any more. Ducklings
bravely jump in water out from nest, even if they were hatched in old nest of
any warblers, made high above water. At week age they already completely change
down for juvenile feathering.
Young birds do not have metal shine on feathers of head, and male looks similar
to female. But after the mew young drake gets bright order characteristic for
males of this species. Nestlings of first hatch grow very quickly, and the next
year they nest normally. In favorable years separate individuals have time to
hatch chicks already till the first year of life. Life expectancy of dwarf paradise
duck is rather short – no more than 5 – 6 years.
Common
forest duck (Sylvanas sudamericana)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: lakes, rivers and marshes of forests and woodlands of tropical South
America.
The Ice Age that occurred during the transition of Holocene to Neocene forced
some species to migrate to southern regions. Certain species, even with the
end of the ice age, remained in their new localities giving rise to new animals.
The genus Sylvanas, the forest ducks is one of these cases. This genus originated
from mallard (Anas platyrhynchos) x northern pintail (Anas acuta) hybrids which
migrated to southern regions to avoid the Ice Age. When these ducks reached
these new areas, they interacted with local birds of the same Genus. This resulted
in new hybridization with these species, resulting in the genus Sylvanas having
genes from them. These animals have genes of the domestic duck (Anas platyrhynchos
domesticus) that went feral, the Mexican duck (Anas diazi) and the white-cheeked
teal (Anas bahamensis). The type species of this new genus is the common forest
duck.
The common forest duck has 60 of length and a wingspan of 93 cm. All members
of the genus Sylvanas are sexually dysmorphic. The males of common forest ducks
have a glossy bottle-green head and a cinnamon breast, throat and cheeks. The
males also have a long tail, a characteristic inherited from the pintails. This
characteristic is present in all members of the Genus. Their wings are greyish
brown, and a white belly. The females are mottled, with each individual feather
showing sharp contrast from buff to very dark brown. They also have white feathers
on their cheeks and throat. Both sexes have iridescent violet feathers in the
speculum and white feathers in the tail. The beak is reddish orange with a grey
base, the colors are more bright in the males. The legs and webbed feet are
orange. Young males are similar to the females, except for a pale brown breast,
throat and cheeks. They acquire adult plumage with 3 years, with this age they
reach sexual maturity.
These birds are omnivorous, eating from plant material to invertebrates, such
as molluscs, insects and crustaceans.
Common forest ducks usually form pairs until the female lays eggs at the start
of the nesting season. At this time she is left by the male who joins up with
other males to wait for the molting period, which begins in the rainy season.
During the brief time before this, however, the males are still sexually potent
and some of them either remain on standby to sire replacement clutches (for
female ducks that have lost or abandoned their previous clutch) or forcibly
mate with females that appear to be isolated or unattached. A characteristic
that differs this Genus of their ancestors is that they build floating nests
hidden in the aquatic vegetation of lakes, calm rivers and other freshwater
reservoirs. Egg clutches number 8 to 14 creamy white eggs. The eggs are laid
on alternate days, and incubation begins when the clutch is almost complete.
Incubation takes 28 days and fledging takes 50 days. The ducklings are precocial
and fully capable of swimming as soon as they hatch. The lifespan of these birds
is of 16 years.
Others species of the genus Sylvanas:
Masked forest
duck (Sylvanas andina)
Living in freshwater reservoirs of forests in the foothills of the Andes, this
bird has 56 cm of length and a wingspan of 87 cm. The male of this species has
grey flanks and back, a white underbelly and a green head. The head is glossy
bottle-green with a black strip around the eyes. The female has plumage similar
to that of the common forest duck, with the exception of a pale grey stripe
around the eyes. Both sexes have iridescent blue feathers in the speculum and
a white tail.
Crowned
forest duck (Sylvanas coronata)
Living in freshwater reservoirs of the island of Great Antigua, these birds
have 35 cm of length and a wingspan of 47 cm. They are poor flyers, but compensate
by flapping their wings into the water as well as using their feet to swim fast,
in a similar way to the modern steamer ducks. The male of this species have
a glossy bottle-green crown and nape. They also have grey flanks and backs.
The female is similar to the female of the common forest duck. Both sexes have
iridescent blueish green feathers in the speculum and a brown tail.
Northern
forest duck (Sylvanas septentrionalis)
Living in the freshwater reservoirs of tropical North America, the birds have
58 cm of length and a wingspan of 89 cm. The male of this species has a glossy
bottle-green head and a dark grey collar that demarcates the head from the cinnamon
breast, grey wings, and a white belly. The female is similar to that of the
common forest duck. Both sexes have green feathers in the speculum and a brown
tail, which is longer in the males.
This bird species was discovered by João Vitor Coutinho, Brazil.
Graceful
maned duck (Caballoanas gracilis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: mountain areas of China, lakes and rivers in mountain valleys.
Sexual selection leads to the formation of bizarre structures in living organisms.
Bright colors, long feathers, various combs and wattles are elements of the
appearance of birds that are the result of sexual selection. In the Neocene
epoch, a distinct group of bird species – maned ducks – is characteristic of
the mountain regions of East Asia. These are duck species with a characteristic
decoration on the back of the neck – here a strip of long narrow feathers along
the backbone forms something like a mane. These feathers are movable, and the
bird can raise and lower them at will. The courting male opens them and swims
around the female, displaying this decoration to her. Hence the genus name meaning
“horse duck”.
The size of maned ducks, as a rule, is small: the weight of an adult is no more
than 700-800 grams. Their typical representative is a graceful maned duck. It
is a non-migrating bird, occasionally making small migrations within its range.
As a rule, these birds spend the summer on high-altitude lakes, and in winter
migrate to warmer mountain valleys.
Sexual dimorphism is clearly expressed in these birds. The male is slightly
larger than the female. The head is dark green with white cheeks and throat,
the body and wings are brown. The secondary feathers have bright blue “speculums”
with a white border. The mane feathers on its neck are white with red bases,
with an expressed metallic sheen. The female has a camouflage color of the plumage
– it is brown with a greenish tinge on the neck and shoulders; there is a dark
scaly pattern on the wings. The beak of males and females is black with a white
tip, the legs are gray. Other species are also characterized by sharp sexual
dimorphism.
The head is wedge-shaped with a relatively long beak. This species feeds on
soft aquatic plants, dipping its head into the water in the process of feeding.
In case of danger, birds can dive to a depth of up to 2 meters.
The mating season begins in spring, after returning to the mountain lakes. Males
divide the coastal zone of the lake into individual territories, where there
are necessarily thickets of coastal marsh plants – a place for the nest. Part
of the mating ritual is dipping the head with the mane into the water and a
small tremor of the neck, which is accompanied by splashing water. The male
also swims around the chosen female with his mane spread out, vocalizing loudly.
The nest is arranged in thickets of marsh plants. Clutch numbers up to 10 eggs
with a dark green spotted shell. Incubation lasts 19 days; the chicks leave
the nest the day after hatching. Young birds fully fledge and acquire the ability
to fly at the age of 38-40 days. Sexual maturity comes at the age of 10-11 months,
life expectancy is up to 15 years.
Mountain lakes of East Asia are inhabited by similar species:
Cobaltic
maned duck (Caballoanas cyanea) is a small species weighing up to 600
grams. The head, neck and back of the male are dark blue with a metallic sheen;
the “mane” is white. There is a V-shaped white spot on the chest, the body is
brown. In the female, the blue color is replaced by dark green with a faint
metallic luster. This species lives in the Yunnan Mountains. There are 5-6 eggs
in a clutch.
Modest
maned duck (Caballoanas modesta) belongs to medium-sized species: the
weight of the male is up to 1.2 kg, the female is about 1 kg. It inhabits the
Sichuan Mountains and spends the winter in warm valleys. This species is distinguished
by a faint plumage color: the male and female are dark brown, differ only in
the presence of a longitudinal white stripe on the male’s throat and upper neck.
The male’s “mane” is also white. Birds of both sexes have dark blue speculums
on their wings, visible only in flight. These birds are cautious, prefer to
stay close to reeds, and are able to dive to a depth of 3 meters, escaping from
predators. There are up to 10 eggs in a clutch.
Isolated from these species, Japan
maned duck (Caballoanas nipponensis) lives on the Japan Islands. This
species inhabits the mountain reservoirs of the Japan Archipelago. This is a
bird weighing 700-900 grams, with clearly expressed sexual dimorphism. Male
is black with a green metallic sheen on the wings, white neck and a “tie” on
the chest. The head is dark blue with a metallic sheen; the beak is yellow with
a black tip. The “mane” is orange-red with black tips of feathers. The female
has brownish-gray plumage with small mottling on the head, neck and chest, and
with a dark scaly pattern on the wings. Birds feed on underwater vegetation
and ground grasses with thin leaves. There are up to 8 eggs in a clutch. Escaping
from the enemy, these ducks can dive to a depth of 4-5 meters and stay under
water for up to 2 minutes.
Golden-maned
duck (Caballoanas juba-aurata)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: rivers of the plain area of China in the zone of tropical and subtropical
climate.
A group of species of maned ducks is very characteristic for freshwater reservoirs
of East Asia, where they are found from mountainous areas to plain rivers. The
greatest diversity of these birds is observed in mountainous areas, where many
local species of these birds have formed
due to isolation. Lowland species are less diverse and have a wide range, differing
in their diet and behavior. The largest species in the group of maned ducks
is golden-maned duck: the weight of an adult is up to 2 kilograms.
The plumage color of the male is reddish-brown with light gray wings; the head
is chocolate-brown. The elongated feathers on the back of the male’s neck are
bright yellow. Sometimes birds of a dark color morph appear – the background
color of males is dark brown, the wings are dark gray, and the “mane” is orange.
Females are grey with a dark scaly pattern on the wings and longitudinal brown
strokes on the feathers of the head and neck.
The mating season begins in spring. Males occupy individual areas along the
riverbanks and arrange courtship displays, swimming parallel to each other with
their manes open. On the borders of individual territories there are duels between
males during which birds call loudly, “nodding” their heads and showing rampant
“manes” of feathers. Sometimes displays turn into fights, during which birds
can tear each other’s feathers out of the “mane”.
Courting the female, the male swims around her, bending his head and trembling
his “mane”.
This duck species feeds on swamp and terrestrial vegetation, eating a significant
number of invertebrates together with plants. Especially often this duck settles
on islets in the river channel inaccessible to terrestrial predators. In such
places, birds arrange nests on the ground among tall swamp vegetation. Clutch
numbers up to 8 eggs with a greenish-blue shell lacking spots. Chicks are led
by both parents, the male mainly protects the brood from predators, and the
female warms the chicks. Young birds have modest plumage and are similar in
color to the female; males have only slightly lighter wings. In juvenile plumage,
the “mane” on the neck is not developed – it appears only with the onset of
sexual maturity. Young birds become capable of reproduction the next year. Life
expectancy is up to 20 years.
In the warm-temperate regions of East Asia, a similar species is found – Transbaikalian
maned duck (Caballoanas transbaikalica). This is the northernmost species
of the genus, living east and south of Lake Baikal, as well as in the Amur basin.
This species has a memorable coloring: the “mane” of males is red with white
tips of feathers. The plumage of the drake is gray with a darker back and a
metallic sheen on the neck, the head is almost black with a snow-white beak.
The wings have dark blue speculums with a white edge. The female is brown with
blue speculums on the wings; there are numerous mottled spots on the neck and
chest. The female’s “mane” is shortened. The weight of the male is about 1500
g, the female – up to 1300 g. These birds feed on soft aquatic vegetation. There
are up to 6-7 eggs in a clutch. This species is migratory, winters on the plain
rivers of China, occasionally forms hybrids with the golden-maned duck.
Bunbulama
(Barobiziura bunbulama)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: South and southeast of Meganesia, Tasmania Island.
Neocene ducks are as widespread in the world as they were in the human era.
Many of them are descendants of mallards, including descendants of birds domesticated
by people. However, the descendants of other species of ducks are quite numerous,
and among them there are very peculiar forms. The lifestyle of most of them
has changed little – ducks occupy a convenient ecological niche where they have
practically no competitors. The large bunbulama duck inhabiting Meganesia looks
rather strange and unusual against their background. This species is a descendant
of the musk duck (Biziura lobata) of the Holocene epoch. The name is given after
the rain spirit of the Australian Aborigines.
This bird species is a very large representative of the family. The body length
of the male is 110 cm, the female is 85 cm, and the wingspan is up to 170 cm.
The weight of an adult is 6-8 kg. At the male bunbulama the throat “blade” of
the ancestor turned into a resonator that amplifies the bird’s voice. Birds
of both sexes have gray plumage with mottling on the wings, back and tail, the
head and upper half of the neck of the male are red-brown. Bunbulamas feed mainly
on food of animal origin – crustaceans, fish and amphibians; they can eat small
reptiles or rodents. Birds often peck snails, insects, and occasionally spiders.
Very rarely, bunbulams eat ripe berries, using them as a source of vitamins
and some minerals. Outside of the breeding season, these birds are relatively
tolerant of their relatives, however, not letting them get too close. This allows
the birds not to deplete their food resources. Outside of the breeding season,
a significant part of the population leaves the rivers and goes to feed in the
Eyre Gulf and in the shallow waters of the southern coast of Meganesia.
The mating season begins in spring, in September-October, and often coincides
with the onset of rains. At this time, the birds return to the rivers and lakes,
where they begin to occupy convenient territories, preparing for nesting. Males
at this time vocalize loudly, inflating their resonators. They try to win a
harem of 7-8 females, and weaker males have to live in monogamy or remain single
until the next breeding season. At this time, all convenient places on swampy
and simply damp areas of the coasts become a battlefield – males are aggressive
towards their relatives. During the fight, they strike the opponents with their
wings and pinch with their beaks, tearing out patches of feathers from each
other. Weaker males often go for a trick: they hide in the thickets on the edge
of the nesting territory of the harem owner, and try to mate with a female who
is out of sight of the male. Some females meet such courtship aggressively,
but in other females 1-2 eggs in a clutch are fertilized by such a third-party
male. The owner of the harem regularly patrols the borders of the territory
and checks possible shelters, but still he has to raise a certain number of
other male’s chicks.
A few days after mating, 5-6 eggs with a dark green shell with small brown spots
appear in a cup-shaped nest made of grass. Bunbulamas nest in groups, so single
females are stressed, and the number of eggs in their nest is 1-2 less. Eggs
are incubated mainly by females, but males with a small harem can replace them
on the nest, so fewer eggs die in such birds, and less number of eggs is compensated
by the better survival of chicks. Both parents take care of the chicks. Females
of the same harem, as a rule, do not care about other female’s chicks. When
young birds are in danger, the male tries to attract the predator’s attention
by honking loudly and simulating injury, while the female takes the chicks to
the shelter. The male attacks small predators and strikes them with his beak
and wings. Chicks become independent in the period from January to February.
At this time they are fully fledged, only in males the head has a gray color.
At the age of 2, they are already sexually mature. Life expectancy is up to
20 years.
This bird species was discovered by Nick, the forum member.
Bustardgoose
(Terrestranser otiformis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Middle and Central Asia – Southern Siberia, Mongolia, the north of
China; bushes and grasslands.
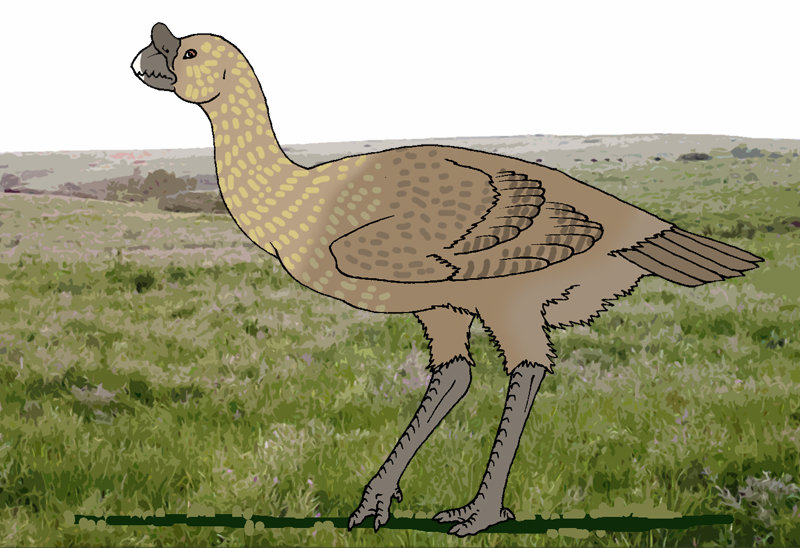
Picture by Tim Morris
The brief period of decrease of number and specific variety
of herbivorous mammals at the end of Holocene had caused by influence from the
side of people. Only few species of wild ungulates could survive – they became
the ancestors of hoofed mammals of Neocene. Simultaneously birds had taken advantage
of this circumstance; in early Neocene they gave a number of specialized herbivorous
of forms partly replacing mammals. Later the majority of them had become extinct,
but some species had continued the existence in Neocene.
Deserts of Central Asia in more humid climate of Neocene are overgrown with
various bushes created a special mosaic landscape, alternating with sites of
grass vegetation. Among bushes, under legs of large herbivorous saigochenias,
large birds bustardgeese feed. They are terrestrial not swimming geese without
palamas on legs, adapted to life in dry area. They had partly repeated the evolutional
experiment of extinct in Neocene Hawaiian goose, or nene (Branta sandvicensis)
which also had lost palamas, having adapted to terrestrial life. Bustardgoose
is very large bird more than meter in height and weight of about 15 kgs, having
strong constitution.
The feathering of this bird combines in colouring yellowish and brown tones.
On head and neck plumage is brown with yellow longitudinal strips along every
feather. Because of it the head of bird looks lighter, than the body. On wings
feathers have darker edge; when wings of bird are fold up, edges of feathers
form dark cross-striped pattern.
Legs of bustardgoose are rather long, with thick short toes. The palama between
them is reduced. The bird is rather heavy to fly long, therefore bustardgeese
spend the most part of life on the ground, searching for food. Nevertheless,
the bustardgoose has kept ability to fly. These birds are able to run very quickly,
accelerating momentum up to 50 kms per hour at the short distance. The running
bustardgoose extends head forward, and becomes less appreciable among high grass.
If there is no other way to escape from predator, bustardgoose flies up, loudly
flapping wings. Skill to fly is vital for bustardgoose for other reason: it
is the bird of passage wintering in Southern Asia – at the south of Hindustan
and in mountain areas of Jakarta Coast. Wingspan of bustardgoose reaches 3 meters.
Neck of bird has moderate length and is mobile. Beak is rather short and deep,
slightly similar to beak of turtle, dark grey with white tip. Above the beak
of sexually mature male big corneous knob grows. Edges of beak are jagged –
it is an adaptation to tear off grass and leaves of bushes. However, bustardgoose
can put strong bites to the opposer in case of need.
By their nature bustardgeese are peaceful herbivorous birds. They eat mainly
the variety of grasses and leaves of bushes, avoiding continuous thickets of
graminoids with rigid stalks and leaves. These birds willingly eat sappy stalks
of non-graminoid grasses – members of carrot family, mints and other species.
Because of specific character of feed bustardgeese live mainly at the edge of
bush thickets where non-graminoid grasses dominate and where herbivorous mammals
from plains do not come.
These birds are strict monodins nesting on the ground. The pair at this species
is forms to many seasons, and sometimes to all further life. Nesting cares of
bustardgoose begin right after birds come back from wintering – pairs of birds
hasten to occupy best protected places for nesting in the center of colony.
Males till the courtship season fiercely combat with each other, winning the
right to the nesting site. During the fight they strike each other strong impacts
by wings and paws. Females, supporting males, can combat with each other, not
entering a duel of males.
The colony of bustardgeese arranges nesting place at the plain area - so it
is easier to birds to find out the predator in time. The main protection of
bustardgeese is harmonious active defense. Operating together, they easily can
drive away from a nest the majority of species of four-footed predators, living
in the neighbourhood.
The clutch of bustardgoose totals about 5 large eggs with spotty brown shell.
The clutch is hatched mainly by female, and male occasionally replaces her on
the nest. The hatching lasts 44 – 45 days. Chicks of bustardgoose come into
the world covered with down and well advanced; they at once leave nest. The
down of young birds is light brown with longitudinal black strips – it provides
fine masking to them. Young birds eat not only vegetative food, but also small
animals; therefore they grow rather quickly, growing up to an autumn up to the
weight of about 7 kg. In the beginning of an autumn young birds try to fly,
and shortly before approach of winter fly out to wintering places in common
with parents.
The young growth becomes able to nesting at the age of about 3 years. Life expectancy
of this species reaches 40 years.
Sargasso
eider (Eusomateria algophila)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: algal fields near New Azora island.
Ice age at the boundary of Holocene and Neocene rendered significant influence
to distribution of some groups of typical polar animals. In ice age many inhabitants
of polar latitudes had moved to low latitudes. At the receding of glacier some
immigrants had died out, others had receded after glacier, and the third ones
had adapted to changing conditions of life, having remained at the former area.
The Sargasso eider, the representative of ducks living in algal fields near
New Azora island, belongs to the number of such immigrants. During the ice age
its ancestor, one species of eiders (Somateria spp.) had moved to the south.
Expansion of Atlantic Ocean and occurrence of cold Antigulf Stream current promoted
the naturalization of immigrants in new habitats.
Sargasso eider leads very specific way of life – this duck spends almost whole
life, except for the nesting periods, among algal fields of New Azora. It has
a rest on floating trunks of trees or on carpets of tangled sea macroalgae,
and eats sea animals living in algae thickets. It is a medium-sized duck – weight
of adult bird is about 500 grammes.
The remarkable feature in appearance of this duck is the original comb with
straight top edge – an outgrowth of corneous cover of beak. Inside it there
is a pair of large salt-secreting glands – it is the necessary adaptation for
permanent inhabiting in sea water. Cornificated cover of beak and comb is colored
very brightly and is appreciable from apart. Females have beak of black color
and bright pink comb with white edges. At male beak is cherry red, and comb
is orange-red with wide yellow edge.
The sexual dimorphism in colouring of plumage at this species is expressed in
high degree. Male has black-and-white plumage characteristic for eiders: it
has white wings with black secondary feathers, black “cap” on head including
the area of around eyes, black back part of neck and horseshoe-like black spot
on breast. Other parts of its plumage are snow-white. The skin of legs is black
– such color is less attractive for underwater predators. Female has modest
brown colouring with black dapples on back and wings. Only around of eyes it
has thin rings of white feathers. Its legs are also black.
At algal fields of New Azora Sargasso eider eats small gastropods and bivalves
settling in algae. It occasionally eats small crabs. Bird dives for them, pressing
wings to body and paddling by paws alternately. This bird can hold the breath
for two minutes, but dives not too deep – not deeper than to five meters. The
haw is transparent – under water it serves as glasses, allowing searching for
food with the help of sight.
Abundance of forage permits sargasso eiders to be tolerant to the presence of
congeners. Birds frequently rest in common, gathering in numerous groups on
floating tree trunks. They are sociable, and in their sound “dictionary” there
are some signals used in various situations. The loud voice helps these birds
to communicate at great distance and to reproduce alarm signals. The main hunter
of these ducks is the sea harrier, the
bird of prey living on algal fields. Escaping from it, sargasso eiders dive
and hide in algae thickets, having jutted out at the surface only a beak under
covering of seaweed thalli floating on water. Under water predatory fishes and
cephalopods threaten to these ducks, because sometimes they attack diving birds.
Seasonal prevalence in breeding at these birds is not expressed, and in algae
fields birds and their posterity may be observed at any time. This species of
birds is strictly monogamous; the pair is formed for one nesting cycle only.
Displaying male shows itself to the female, inflating throat and stirring up
a head.
Sargasso eider hatches posterity at New Azora island. Birds nest on rocky islets
near coasts where there are no ground predators, and often side by side with
plesioloons. Female
litters nest with its own down and hatches eggs alone. Nevertheless, male constantly
keeps near the nest, protecting it from encroachments of other sea birds. The
incubation lasts till 35 days. Ducklings, having dried under the female, at
once make long sea transition, following parents from island coast up to algae
fields, and further permanently live in algal fields. They live with parents
within first two months of life, and then become completely independent. Young
males before sexual maturity have the plumage similar to plumage of female,
with only wider rings of white feathers around of eyes. At the age of two years
their plumage is replaced by one characteristic for adult individuals. Within
one year birds succeed to make two hatches.
American
non-goose (Anseroanas chenus)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae), subfamily: True ducks (Anatinae)
Habitat: temperate and subarctic regions of North America, freshwater reservoirs.

Male

Female
Pictures by Pavel Volkov
Anserine birds represent one of the oldest orders of birds:
their first representatives appeared in the Mesozoic, during the time of the
dinosaurs. Over the next tens of millions of years, they successfully survived
many changes in the biosphere: the mass extinction that destroyed the dinosaurs,
other episodes of extinction, glaciations, and even the epoch of anthropogenic
pressure. In the Neocene, new families and species appeared among these birds,
sometimes of a rather unusual shape, but more often of a fairly recognizable
appearance. One of these “background” common species is the American non-goose.
This is a fairly large bird, 45-60 cm in length. The body of this bird is similar
in proportions to a duck, and on land this bird also waddles, but with a long
neck and head it looks more like true geese. This bird feeds on plants, both
terrestrial and growing in shallow water – American non-goose in unable to dive,
but, like its ancestor, the American wigeon (Anas americana) of the Holocene
epoch, the bird can plunge its head into the water quite deeply, standing in
the water with its tail up. These birds almost do not eat food of animal origin:
only those small animals that accidentally are swollen along with plant food.
This bird often feeds on land, especially in autumn, when berries and similar
food ripen.
Age variability and sexual dimorphism are expressed in the plumage of American
non-goose. Females and juveniles have ripply brown-red plumage, darker on the
neck. In adult females, the skin around the eyes is white, but these areas of
skin at rest and outside the courtship season are covered with plumage. During
the courtship season, the female spreads her feathers around her eyes, and the
blood vessels under the areas of white skin fill with blood. Then the white
markings on the head become more noticeable. Mature males are brighter colored
than females: they have black-and-blue wings and a green “mask” on the head.
The chicks are covered with yellowish down with longitudinal stripes of gray
color. The legs of young birds are black, coral pink in adult males, pinkish-white
in females.
The American non-goose breeds on the shores of small reservoirs, often among
swamps and in other places inaccessible to terrestrial predators. This is a
fairly aggressive bird that lives in separate families and forms large congestions
only closer to the time of migration. Breeding couples often last a lifetime.
The main enemies of this species are birds of prey and carnivores, predatory
fish occasionally attack chicks; epizootics also occur, caused by mass infection
with internal parasites.
The American non-goose is a migratory species, and in the northern part of its
range in the second half of autumn this bird flies to the south of the North
American continent for the wintering. Birds living in the southern part of the
range make relatively small migrations, and individuals of northern populations
fly much farther south. In the spring, birds usually return to their old nests,
but young individuals first of all have to win a place for nesting, often fighting
fiercely for it. Also, young birds that have not yet nested are easier to change
their place of residence, flying from wintering to new places for themselves;
males do this especially often. Fighting for territory, non-geese beat each
other with their wings and try to pinch the opponent’s neck. The male can drag
the defeated opponent by grabbing him by the plumage on the neck or on the nape.
Females have a stronger attachment to their native places, and they prefer to
nest in their native pond or near their native places. Pairs of this species
are formed in the south, during wintering.
The males of the American non-goose court females very gently, swimming around,
quacking and “hugging” with their wings from the side. If the female accepts
courtship, the ritual delivery of food – parts of plants – begins, as well as
the rubbing of mating partners against each other with necks and heads. The
nest is land-based, arranged in a shelter, in places inaccessible to terrestrial
predators. Egg laying occurs in the second half of spring, with an average of
about 6-7 eggs in the clutch. Only the female incubates, the male does not replace
her, but guards the borders of the territory and drives away enemies, aggressively
attacking them and striking with wings and beak. Sometimes widowed females stay
on the edge of the territory of breeding couples and mate with a male from a
pair, but lay eggs and raise offspring alone. Such females lay less number of
eggs and sometimes lay their egg in the nest of another female. Chicks are fed
and protected by both parents, but parental bonds break up during wintering,
when young birds choose their mating partners.
The usual life expectancy of the American non-goose is 13 years, but some individuals
live up to 20 years.
This bird species was discovered by Bhut, the forum member.
California
brant (Branta insularis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: California Island.

Picture by Pavel Volkov
The ice Age that separated the Holocene from the Neocene forced
many polar species to retreat to lower latitudes, towards the tropics. The black
geese inhabiting the Holarctic zoogeographic realm were no exception. The species
diversity of these tundra birds declined shortly after the extinction of man,
but during the ice age they restored their numbers. And after the retreat of
the glacier, some of the newly appeared species remained in more southern areas,
having adapting to climate change. The southernmost species of Neocene-era black
geese is the California brant from the California island, a descendant of the
Canadian goose (Branta canadensis).
This is a relatively small representative of the family: the body length is
61-67 cm, the wingspan is 121-145 cm, and the body weight of an adult is 1-1.6
kg. In its proportions, the bird resembles a goose, but has elongated legs.
The palamas are partially reduced: they reach only the middle of the length
of the toes. The color of the plumage is inconspicuous – brownish-gray with
white transverse mottling, wider on the belly, which makes it seem lighter.
Of all the Neocene black geese, this species is the most undemanding to the
presence of reservoirs. The native island of this species is quite dry, and
most of the fresh water here are temporary ponds and short rapid rivers. Birds
of this species prefer to stay in the forest, among rocks and in thickets of
cacti, but often gather on the seashore, especially when freshwater bodies dry
up. This bird moves steadily on the ground, runs fast and often escapes from
danger in this way during molting, when it completely loses the ability to fly.
The California brant is a good swimmer, but a poor flier – the bird is able
to fly several hundred meters only. Sometimes birds of this species fly into
the sea and feed on brown algae on coastal shoals, but they do not fly away
from the island.
This species has switched exclusively to plant food: leaves, shoots of herbaceous
plants, berries (including cactus fruits) and algae.
The California brant is a monogamous species, pairs persist until the death
of one of the partners. The breeding season falls on October-November, when
winter rains begin and ground grassy vegetation develops rapidly. This species
nests in pairs or scattered colonies numbering 4-5 pairs, often on a cliff and
under the protection of some bird of prey that has settled in the neighborhood.
Courtship displays include ritual plunge of the beak in water or simply touching
the ground with the beak if there is no water nearby (this element is performed
by both birds), and the vertical pose of the male, in which he presses the female
to the ground. After mating, the brants fully spread their wings, fluff their
tails and stretch their necks with a call. The female lays 5-7 eggs in a hole
lined with grass and down. Incubation lasts 29 days, only the female hatches.
The hatched chicks are covered with brown down. At the age of 8-9 weeks, they
fledge, and become independent at 7 months, although parents can tolerate their
presence until the beginning of the next breeding season. A pair of birds, often
with the last brood, keeps and feeds together. Before the onset of sexual maturity,
at the age of 2, birds of the same brood keep together; later, flocks break
up and young birds look for a mate. Life expectancy reaches 20 years.
This bird species was discovered by Nick, the forum member.
Kuihi-nui
(Notobranta vocifer)
Order: Anserine birds (Anseriformes)
Family: Ducks and Geese (Anatidae)
Habitat: North and South Islands of New Zealand, lowland vegetated habitats
adjacent to fresh water sources.

Picture by Pavel Volkov
Game birds were introduced into New Zealand by man during
the Holocene, along with large game animals for shooting. One such bird was
the Canada Goose (Branta canadensis), that also served as an ornamental waterfowl.
The Kuihi-nui is a descendant of this usually migratory bird, which was introduced
to New Zealand by man during the Holocene. This goose is somewhat heavier and
longer-legged than its ancestor, up to 130 centimeters long with a 2.2 meter
wingspan and weighing up to 4.5 kilograms. Unlike its ancestor it does not migrate,
and spends more time on land, mainly grazing, wings are somewhat shorter and
rounder in proportion. Head a neck is colored dark blackish brown, without the
white “chinstrap”; bill is shorter and more robust bearing coarse serrations.
Its body is an all over brownish grey, with darker brown on the primary wing
feathers, and a whitish rump, legs and bill are yellowish.
Diet consists of mainly grass, herbs, grains, the leaves of low bushes, and
various water plants, this bird spends less time feeding in the water than its
ancestor. Its call is rather typical for a goose but louder, an abrupt honk,
name of this bird translates to “loud goose” in native Maori language. Males
are antagonistic toward each other both in and out of the breeding season, both
sexes are territorial and audacious, chasing away smaller animals they consider
a threat. They generally congregate in large flocks in preferable feeding grounds,
and though not migratory, will fly in groups from place to place.
Kuihi-nui seek out a mate during their second year, forming pairs that remain
together their whole lives. In early spring, up to 9 eggs are laid on a nest
which is usually located on the ground in a secluded, well-vegetated spot, both
male and female help incubate the eggs. The nest is a shallow bowl-depression
lined thickly with leaves and down, incubation lasts up to 33 days. Goslings
are precocial, immediately able to follow their parents, swim and feed themselves;
parents will lead them in a line from place to place. Multiple groups of parents
may form crèches of young. Goslings start to fledge from about 7 to 9 weeks
old; by 3 months old they have reached adult size; sexual maturity is reached
at 2 years old. Life expectancy is up to 25 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Great
gerda (Gravianas gerd)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Europe to the north of the Alps and the Fourseas, rivers, Wenedian
Lake.

Male

Female
Picture by Pavel Volkov
Hunting and habitat destruction have undermined the numbers
of shorebirds and waterfowl. Some of them became extinct, and others survived
only due to hybridization with related species. Some species turned out to be
numerous enough to survive the anthropogenic crisis and the ice age. One of
these birds was tufted pochard (Aythya fuligula). Driven south by the glacier,
it succeeded to restore the population and become the ancestor of new species.
The largest of them is the great gerda (the species is named after the Scandinavian
giantess – the mistress of rivers and lakes).
This species is one of the largest waterfowl and flying birds in the world.
The body length of an adult bird is up to 150 cm, the wingspan is about 3 m,
the weight is up to 15 kg (the male is larger than the female). Sexual dimorphism
is expressed in the appearance of the bird. The color of the males is black
with a green metallic sheen on the neck, shoulders and back; on the head there
is a large crest of elongated rusty-red and white feathers. The female is grey
with a black head and neck, with a small tuft of black feathers on her head.
The body is somewhat heavy, the neck is short. The bird’s head is relatively
massive; the beak is large, equipped with a significant number of corneous serrations
that allow picking plants. Gerda looks heavier than Holocene swans, but leggier.
The palamas between the toes are normally developed, although the bird spends
a significant part of the time on the shore. The voice of these birds is a rasping
quacking and cawing, the alarm signal is a loud trumpet sound.
This bird species lives on the shores of large lakes and rivers, less often
near ponds and in river estuaries, and sometimes is seen on the seashore. A
prerequisite for bird life is the development of coastal grassy vegetation.
The great gerda feeds on plant food – stems and leaves of cane, reeds, underwater
vegetation. A strong beak helps the bird to tear off even the tough foliage
of sedges and cereals. Sometimes gerda dives for food to a depth of 5 m and
digs up the rhizomes of water lilies under water; the birds eat the surfaced
rhizomes on the surface of the water. This species is a kind of ecological analogue
of ruminant mammals in swamps. On land, the bird moves easily and freely, and
can even run, but it is almost unable to take off, so it prefers to graze in
swampy meadows, inaccessible to terrestrial predators. From the water, the bird
flies up heavily, noisily, from a running start, but the flight is fast and
easy.
The great gerda breeds in colonies of 10-15 pairs of birds, and in wintering
areas it gathers in flocks of hundreds of individuals. This species of birds
flies off for the winter one of the last – already when ice begins to appear
on the surface of reservoirs. Until a snow cover has formed on land, birds eat
withered autumn vegetation and shoots of evergreen wetland shrubs. Gerda winters
in North Africa (the basin of the Saharan Nile), at Zinj Land, New Azora and
the Canarian islands. Some flocks reach Madagascar.
The breeding season begins in early spring, in April-May, depending on the location
of nesting place. At this time birds squabble over the places of nesting and
young or single males also over females. At this time, males arrange courtship
dances: they open their crests and stamp their feet on the ground, and young
birds also scream, flapping their wings. The nest is a bowl of last year’s vegetation
gathered by the female. The inner diameter of the nest is about 140-150 cm.
The building of the nest begins immediately after the spring flood. The nest
is arranged on islands among marshes, in thickets of cane, reeds or cattails,
less often in shallow water, where the nest rests on the bottom of the reservoir.
The bottom of the nest is lined with grass and moss, as well as the female’s
own down. The clutch contains 5-7 large eggs with a greenish shell without spots,
which the female incubates for 30-32 days. Males guard the colony at this time
and behave aggressively towards small and medium-sized terrestrial predators.
In the case of the destruction of the clutch, the birds do not repeat it – this
is due to the long period of development of the young. Ducklings hatch well
developed, follow the female 20 minutes after hatching. They feed actively and
grow quickly, at the age of 4 months they are completely independent and acquire
a juvenile feather dress. By the time of migration, young birds weigh up to
8 kilograms. During the wintering, they separate from their parents and begin
an independent life. The growth of young birds continues during the wintering,
birds returning in the spring weigh up to 11 kg. Sexual maturity occurs at the
age of 1 year, but some birds participate in reproduction only from the age
of 2. Life expectancy is up to 40 years.
In the swamps of Siberia and the Far East, a close relative lives – grey
gerda (Gravianas cinerea), a duck 110 cm long, with gray plumage. Females
of this species do not have a crest. These birds winter in Southeast Asia, India,
Indonesia and Meganesia. Their migration way goes around the Himalayan mountain
range – they do not fly high. This species does not overlap with its European
relative; the barrier between their ranges is the Urals and the steppes of Central
Asia. On the shores of the Fourseas, the sea
or lesser gerda (Gravianas minor) lives – the smallest species
of the genus, about 95 cm long and weighing up to 6 kg. This is a bird with
brown plumage, the male has a black head with a black crest; the females have
no crest, or only some elongated feathers are present. It feeds both in rivers
and in the sea, plucking thickets of pondweed and other hydrophytes. Nesting
takes place in March, colonies are located on islands in river deltas. This
bird species does not undertake long migrations, often wintering simply on the
southern shore of the Fourseas.
This bird species was discovered by Nick, the forum member.
Shellfish-eating
duck (Pelagotachyeres ostracophagus)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Antarctica and nearby islands.

Picture by Sauron
During the Holocene, human interference in nature caused the
extinction of many species. Those that were lucky enough to survive, left behind
descendants in the Neocene. One of these survivors was the flying steamer duck
(Tachyeres patachonicus), which gave rise to the Genus Pelagotachyeres, with
the shellfish-eating duck being its type species. This species nests in Antarctica
and nearby islands, migrating during the winter to the sea near the coast of
Southern South America.
The shellfish-eating duck is a larger species of duck, with 68 cm of length
and 97 cm of wingspan. This is a sexual dismorphic species, the male is larger
than the female. They have white heads with a bright orange bill. Their webs
and feet are orange. The females have brown heads and necks and their bills
and feet have a pale yellow color. The rest of the plumage of both sexes is
gray on the bodies and white in the bellies.
This species feeds on mollusks, crustaceans like crabs, and on sea urchins,
starfish and sea anemones. They dive using their large feet for propulsion,
then they capture and crush the prey by using their beaks, which are strong
enough to break the exoskeleton of crustaceans and shells of molluscs. Although
they can upend when feeding in shallower waters, they mainly dive to find their
prey on the seafloor.
This species is monogamous, spending most of its time in small family groups
composed of the female, the male and the chicks. However, during the winter,
these birds gather in larger groups of up to 300 individuals. During the breeding
season, the males become aggressive with others of the same species, attacking
them violently to the point of causing severe injuries. Sometimes the female
will participate in these attacks. This is the reason why they only stay in
pairs during this period.
Their mating happens at the sea and is highly ritualized. The male and female
begin by dipping their bills into the water at increasingly fast paces before
the female submerges almost completely into the water and is mounted by the
male, after which the female and male raise their bills high into the air in
a hostile fashion and grunt audibly.
Once mating has occurred, females lay between 6 and 14 eggs in down-lined nests
and incubate them for about 34 days. The males stay with the female to defend
the nest and participate in the incubation process only when the female briefly
leaves the nest to bathe and preen. The nest is usually located in cliffs near
the sea, but can also be built in plain areas near it. After 23 days of incubation,
the ducklings will hatch. As soon as it happens, the parents will move with
them to the sea. For those that were born in cliffs, the young will need to
jump. Their brown and white duvet, small size and very light weight helps to
protect them from serious injury when they hit the rocks below, but many die
from the impact. After 12 week, the ducklings get their first plumage and sexual
maturity is reached after 24 months. Adults can live for up to 20 years old.
Another representative of the Genus Pelagotachyeres is the Masked
shellfish-eating duck (Pelagotachyeres personatus) this species inhabits
the temperate and tropical coast of Southern and Southeastern South America.
This species has 64 cm of length and 89 cm of wingspan, with the male being
slightly larger than the female. The male and female have white heads and necks.
The males have a black patch around their eyes, which is brown on the females.
The rest of their plumage is gray, with a chestnut-brown chest.
This bird species was discovered by João Vitor Coutinho, Brazil.
White-breasted
goose (Gainsboro albicollum)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: tundra of Northern Atlantic coast of North America, Greenland.

Picture by Pavel Volkov
In human epoch the number of waterfowl decreased rapidly due
to hunting and the destroying of habitats. Because of it in rests of former
habitats mixed populations of some species of geese appeared. Before the human-driven
destroying of natural habitats their ancestors lived separatedly and the interspecific
contacts had been minimal. But in epoch of global biological crisis the hybridization
appeared the way of survival of the residual populations of these species. As
a result the Genus Gainsboro, the pale grey geese, appeared.
This genus has a polyphyletic origin: they descends from the white-fronted goose
(Anser albifrons), however there is the presence of genes of feral domestic
geese (Anser anser domesticus + Anser cygnoides domesticus) and the snow goose
(Anser (Chen) caerulescens). These genes express themselves in phenotypes similar
to those of their ancestors.
Their feathers have white and pale gray shades; however the predominance of
these two colors varies with the species. They have orange feet and legs. Their
beaks are similar in size to that of the snow goose, but with the beak being
dark grey and with a knob, like that of the African goose, the domestic breed
of swan goose (Anser cygnoides domesticus), and one of the ancestors of the
feral population of domestic goose that give rise to this genus. The size of
their knob varies according to species. Originating from North America, Gainsboro
geese spread to Eurasia and Meganesia and formed some distinct species there.
The typical representative of this genus is white-breasted goose from North
America. It has a distinctive coloration of feathers: white feathers in the
breast, neck and head, in the other parts of its body there is the presence
of pale grey feathers. This bird is about 93 cm long and has a wingspan about
150 cm.
Geese of this genus normally pair for life, so courtship only occurs at the
time of first maturity. Females are strongly philopatric, meaning they will
return to the place they hatched to breed. The female chooses a nest site and
builds the nest at the area of high ground to avoid the casual flooding. The
nest is a shallow depression lined with plant material and may be reused from
year to year. It is built from pieces of reed, sprigs of heather, grasses and
moss, mixed with small feathers and down. A typical clutch is 6 to 8 eggs, but
greater or lesser number of eggs is not unusual. Eggs are creamy-white at first
but soon become stained. They are mostly laid on successive days and incubation
starts after the last one is laid. The female does the incubation, which lasts
about 29 days, while the male remains on guard somewhere near. Goslings are
precocial and able to leave the nest soon after hatching. Both parents are involved
in their care, and they learn soon to feed independently and become fully-fledged
at 8 or 9 weeks about the same time as their parents regain their ability to
fly after moulting their main wing and tail feathers a month earlier. Immature
birds undergo a similar moult, and move to traditional, safe locations before
doing so because of their vulnerability while being flightless.
These birds are herbivorous, eating grasses and aquatic plants. Avoiding the
competition to another species, traveler goose (Chen migratorius),
this species searches for food mainly in water and in coastal zone.
They are gregarious, forming large flocks and breeding colonies. It has an advantage
for the birds, because the vigilance of some individuals in the group allows
the rest ones to feed without having to constantly be alert to the approach
of predators. After the eggs hatch, some grouping of families takes place, enabling
the geese to defend their young by their joint actions, such as mobbing or attacking
predators. After driving off a predator, a goose returns to its mate and gives
a “triumphal call”, a resonant honk followed by a low-pitched cackle, uttered
with neck extended forward parallel to the ground. The mate and even unfledged
young reciprocate in kind.
Young geese stay with their parents as a family group; in the migratory species
they stay united in a larger flock, and only dispersing when the adults drive
them away from their newly established breeding territory the next year. These
birds migrate to marshes of southern part of North America for wintering.
This genus has five more species widespread in the world:

Picture by Pavel Volkov
White-headed goose (Gainsboro leucocephalus) nests in tundra of Arctic Ocean coast of North America and Beringia. Their size is about 88 cm, and only their head is white. It winters at the Pacific coast of North America.

Picture by Pavel Volkov
Boreal goose (Gainsboro nivalis) nests in tundra regions of Eurasia, migrating to the southern coast of Fourseas in winter. Adults of this species are 86 cm long and are almost completely white, with only their primary feathers pale grey. Sometimes it forms mixed pairs with the previous species, and hybrid birds may be seen in their flocks.

Picture by Pavel Volkov
Grey-winged goose (Gainsboro griseopterus)
inhabits lakes and rivers of Europe, migrating to the southern coast of the
Fourseas in the winter. They grow to 80 cm and have their wing feathers completely
pale grey and the rest of the body is white.
Great
knobbed goose (Gainsboro magnodus) prefers warmer climate and nests
in marshes of Far Eastern Asia in regions of temperate climate, migrating to
swamps of Southeast Asia in winter. They have the biggest knob of the genus.
Its length is 79 cm and the feathers of its underbelly are white, with other
feathers pale gray.
Lesser
knobbed goose (Gainsboro micronodus) is the southernmost species living
exclusively in tropical areas in rivers and lakes of the north of Meganesia
and Jacarta Coast. This is a smallest species of the genus compared to its northern
relatives (only 70 cm long) and has the same color as a previous species. The
difference between the two species is their size and the size of their knobs,
the knob of the lesser knobbed goose is medium sized, smaller than that of the
great knobbed goose, but bigger if compared with the others species. It is a
non-migratory bird species also.
This bird was discovered by João Vitor Coutinho, Brazil.
Ciconianser
(Ciconianser gracilis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: North America, Mishe-Nama Lake and surroundings.

Picture by Pavel Volkov
In the Neocene, anserine birds are a diverse group, including
species of both familiar appearance and
atypical, including very
large ones. One of the largest representatives of the order is a ciconianser
inhabiting the shores of Mishe-Nama Lake. It is a descendant of the white-fronted
goose (Anser albifrons).
Ciconianser is a large bird of atypical proportions for geese, with long legs
and a neck (hence the name, literally meaning “stork goose”): its height reaches
150-160 cm, the body length is about 1 meter. The wingspan reaches 170 cm: the
bird can fly, but does it reluctantly, preferring to walk on the ground. Ciconianser
also has a long movable neck with a small head. The legs are long, with a reduced
swimming membrane – it forms only scalloped outgrowths on the toes. The beak
is up to 17-18 cm long, with denticles characteristic of geese on the corneous
cover; the tip of the beak is expanded. The back, upper neck and wings are light
brown with fine black ripples; the shoulders, chest and belly are white, the
tail is glaucous. A bright crimson beak stands out against the background of
the plumage; the legs are orange. The eyes of mature individuals are light green,
sometimes yellowish; the chicks have black eyes. Sexual dimorphism is not pronounced.
These birds communicate with the help of a ringing cackle and usually behave
very noisily. They have well-developed eyesight and hearing; during feeding,
birds behave cautiously and raise the alarm when a predator approaches.
Ciconiansers inhabit the shallow waters of Mishe-Nama Lake and are found on
the swampy shores of rivers flowing into it. These birds swim poorly, so they
are found only in coastal shallow waters, at a depth not exceeding the length
of the bird’s legs. They are social, usually live in groups of up to 20 individuals.
With the exception of the nesting period, groups of ciconiansers are not tied
to a specific territory and wander through the shallow waters of the lake. They
live in places where there are many filamentous algae and long-stemmed aquatic
plants with soft foliage, which they feed on: large birds lower their heads
to the water and cut and browse plants with their beaks. Together with them,
the bird catches and swallows small animals. Also, the diet of the ciconianser
includes leaves of terrestrial plants, grass and berries. In the water, this
anserine bird also eats worms and other invertebrates, small fish and amphibians,
their eggs. Other anserine birds, as well as rails and grebes, often feed together
with groups of ciconiansers, since some species that are dangerous to smaller
birds do not attack ciconiansers. The enemies of this species are large predatory
mammals and birds, from which long-legged geese try to run or fly away. Chicks
have more enemies: these are birds of prey and small predatory mammals, as well
as various predatory fish.
The ciconianser is monogamous, pairs are kept for life. In early spring, the
female lays up to 6 eggs. These birds do not build nests and simply lay eggs
in a hole in the ground, lining it with their own down. At this time, the male
keeps not far from the nest and drives away small predators and birds, ravaging
the clutches, from the incubating female. For safety, ciconiansers nest in colonies
on islets – among swamps or located at some distance from the shores of the
lake. After the hatching of offspring, all birds leave the nesting grounds and
continue to roam along the shores of the lake. Interestingly, the chicks do
not have such long legs as adults, and they swim on the water like ordinary
geese. Adult birds at this time surround the offspring to keep predators away
from it. Often several families unite and jointly guard their broods.
By autumn, young birds reach about 75% of the weight of an adult, their plumage
is fully formed, and they are preparing for migration. Ciconiansers winter on
the Gulf of Mexico coast, at Cuba and Great Antigua. A significant part of the
population winters in Central America. For wintering, birds prefer grassy swamps
and lakes.
Sexual maturity occurs at the age of 2 years, life expectancy is up to 36 years.
This bird species was discovered by Mamont, the forum member.
Hemiswan
(Xenolor borealis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: tundra and taiga of Eurasia, rivers and lakes.

Picture by Pavel Volkov
Anserine birds, despite hunting, habitat pollution and climate
change, survived the human era relatively successfully, although with noticeable
losses, especially among narrow-range and small-numbered species. After the
disappearance of humankind, they quickly restored the range and species diversity
of the order. Most of the anserines are birds of a
familiar appearance; the exception is specialized island
forms. In Eurasia, a large representative of the duck family is a hemiswan,
a descendant of the taiga bean goose (Anser fabalis), which survived in small
numbers in the Urals and the Far East.
The body length of this bird is about 1.1 m, wingspan up to 2 m, weight about
5.5 kg. The coloration is not bright, which is typical for geese; two color
morphs are known – gray with a silvery tint and bluish wings, back and back
of the neck, and white with a mesh pattern of darker edges of feathers, while
the neck seems darker due to a thicker scaly pattern. Outwardly, the hemiswan
resembles a cross between geese and swans: a goose’s head on a long flexible
neck and a body relatively longer than at geese. Hemiswans feed on plants –
both terrestrial grasses and soft aquatic vegetation.
Usually birds settle, nest and feed in colonies, behave noisily and actively
communicate with cackle; aggression is expressed by hissing with an arched neck.
Birds migrate for the wintering in mid-October, spending winter mainly on the
coast of Fourseas and on the ocean coast of Southeast Asia. The Scandinavian
population migrates to the Atlantic coasts of Western Europe, Gibraltar Isthmus
and north-west Africa.
The nesting season begins in April, soon after the return to the nesting sites.
Sometimes hemiswans nest near the eyries of large birds of prey – they do not
hunt near the house, but perceive attempts of other animals to attack geese
as an encroachment on their own eyrie. Away from colonies, birds defend themselves
from small-sized predators by beating their wings. Pairs are formed during the
winter; partners are strongly attached to each other. Usually, the male returns
from the wintering grounds to the native land of the female.
The nest is simple, ground-based – a shallow hole lined with the down of the
hemiswan itself, mainly of the female. In a clutch there are 5-6 eggs with a
creamy shell. After 28 days, the chicks hatch. After drying out under the wings
of the female, they leave the nest, and in the future they are led and protected
by both parents. After 2 days, the adult geese lead the chicks to the water,
where they learn to search for food. At 50 days, young birds take to the wing,
but they become completely independent after the first autumn migration – the
bond between young and adult birds is broken already in the wintering areas,
sometimes only shortly before the spring return to the nesting sites. Sexual
maturity occurs at 3 years of age. Life expectancy is up to 25 years or more.
This bird species was discovered by Nick, the forum member.
Antarctic
ruddy-headed goose (Chloephaga pratensis)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: nesting in grass-sedge meadows of Antarctica, wintering in South America.
In the Neocene, due to the general warming of the Earth’s climate, the surface
of Antarctica was mostly freed from ice. In open areas, a soil layer appeared
and productive ecosystems were formed: thickets of low-growing evergreen shrubs
and grass-sedge meadows. Numerous herds of herbivorous birds graze on these
meadows. The smallest herbivorous birds are herbivorous Antarctic
chicken seedsnipes, descendants of South American seedsnipes, the largest
are Antarctic meadow swans. But meadow
swans are not the only anserine birds that functionally replace herbivorous
mammals in the meadows of Antarctica.
A characteristic species of Antarctic herbivorous anserine birds is the Antarctic
ruddy-headed goose, a large but flying goose of the genus Chloephaga, a descendant
of the South American ruddy-headed goose (Chloephaga rubidiceps). Huge flocks
of these geese, migrating for the summer from South America, graze on the grass-sedge
meadows of Antarctica, eating not only grass, but also numerous small invertebrates:
Antarctic springtails, tardigrades,
nematodes, larvae of crane
flies, as well as small mole woodlice.
The body length of the Antarctic ruddy-headed goose is 75-100 cm. The constitution
of birds is robust; they have a relatively short neck and a large head with
a wide beak. The plumage color of the male and female is similar, but males
are usually slightly larger than females. The body weight is about 3.5-6.8 kg.
There is a distinct difference in voices between birds of different sexes. Females
honk in a low voice, while males have a higher one.
The color of the head is variable - from ochre-yellow to brownish-red, the wide
ring around the eye is white. The belly is brown with a thin ripple, the chest
has an intense red-brown color, and the back and tail are brown. On the wings
there are large coverts of shining green color – during threatening displays,
birds open their wings, and these colored spots become clearly visible. The
color of the primary flight feathers is beige with black transverse strokes.
The blows of strong wings are one of the main means of protection of these geese,
and the aggressor remembers such a noticeable color. The legs are thick, yellow-orange,
the palama is reduced a little bit – birds spend a significant part of their
time on the ground. Young birds look similar to adults, but their juvenile plumage
is slightly duller than adult feathering and lacks metallic shine. The belly
of young birds is yellow-brown, the large coverts are matte and dark gray. The
coloration of the legs is also slightly paler than that of mature birds.
The nesting areas are located on the grass-sedge meadows of Antarctica. This
is a monogamous bird species; pairs are formed for life and remain faithful
to each other. During the breeding season, males become very aggressive, vigorously
defending their large nesting area. The nest is built in October on dry ground
in deepenings dug under bushes, in tall grass or just in an open area. The lining
for the nest is plant material and down of the incubating female. In a clutch
of 4 to 11 eggs, incubation lasts from 30 to 32 days; occasionally birds lay
eggs in the nests of relatives, and on average 1-2 eggs out of 10 are laid by
neighbors. Only the female incubates, but both adult birds take care of the
chicks. At this time, the birds are molting and they are unable to fly, but
they show increased aggressiveness to possible enemies. Chicks become independent
in the period from January to February; at this time they actively feed and
fledge. The chicks are hunted by local birds of prey, skuas and gulls. Adult
birds actively protect their offspring by striking predators with their wings.
Their primary feathers are very strong, and birds that have changed their feather
coat use them as beaters. Closer to the time of migration, the birds feed in
large common herds, sometimes plucking grass by the root. Herds of this species
can feed on sea coasts, eating seaweed and marine invertebrates, especially
shortly before migration for the winter.
Antarctic ruddy-headed geese winter in Tierra del Fuego, but a significant part
of the population flies further north, bypassing the Andes, and winters in Chile
and Argentina. Some flocks fly to the Falkland Islands.
Birds of this species develop quite slowly. Sexual maturity occurs at the age
of 2 years, life expectancy reaches 40-45 years.
This bird species was discovered by Wovoka, the forum member.
Yallal,
or Common sea merganser (Thalassomergus poseidonii)
Order: Anserine birds (Anseriformes)
Family: Ducks (Anatidae)
Habitat: Pacific coast of North America and Asia, temperate area.
After the ending of human epoch the long period of recovery of ecosystems had
taken place. During this period the descendants of survived species developed
new habitats, and new species evolved due to the ecological differences expressed
increasingly better with every new generation. Among anserine birds the new
group evolved rapidly to occupy marine habitats – sea-dwelling descendants of
mergansers (or sawbills), freshwater fish-eating ducks of human epoch. Red-breasted
merganser (Mergus serrator) – rather common and widespread species wintered
at the sea coasts of both Atlantic and Pacific oceans – evolved to specialized
marine fish-eating waterfowl of separate genus Thalassomergus adapted to life
in water of oceanic salinity. Birds of this genus live in waters of Northern
hemisphere from cold to tropical waters. The most common species of these birds
is yallal, or common sea merganser (“yallal” is the name of sawbill in Koryak
language). This species of merganser nests in the costal forests of Beringia
and spends winter in the costal waters of Japan.
It is a rather large aquatic bird – the total body length is about 72-78 cm
at weight of about 3-4 kg. Body of this bird is of streamlined shape, with rather
short, but mobile neck. Head is rather large, with elongated beak prominently
serrated along its edges. Wings are strong; these birds make long annual migrations
for wintering to warmer Asiatic waters of Pacific Ocean. Their feet are strong
and highly adapted for swimming; body is elongated, and legs are shifted far
back. So these birds have difficulty walking on land, keeping the body in semi-erect
position. In water these birds are highly mobile and can dive deeply. They use
only legs and wide webbed feet to swim, in a similar way to loons and grebes
ща human epoch. Making fast turns under water, they use wings to steer, but
not for “underwater flight” like penguins and auks. Wings of these birds are
pointed, and tail is short and rounded.
One more adaptation to marine life is visible in appearance of these birds:
both males and females have large outgrowths in the base of upper jaw. These
structures contain salt-removing glands inside and allow these birds drinking
salt water without any harm for their health.
Like other marine birds, they have a “standard” coloration of body – darker
upper side and wings, and light breast and belly, like auks and penguins. But
they inherited bright colors of heads from their freshwater ancestors. Yallal
has black back and wings with blue or greenish metallic shine, and white underside
of body. Male’s head is of dark blue color with prominent green shine. Females
have black upper side of body with brownish shade and dark brown head without
shine. The bright spot in its coloration is the color of the beak including
salt-removing gland. At females beak is orange with yellowish outgrowth containing
salt gland. At males both beak and outgrowth are of bright red color. Birds
of both sexes have some elongated feathers on the crown.
Yallal is a strictly zoophagous bird adapted to underwater hunting. Its beak
is serrated and thus allows keeping slimy prey like fish and small squids. Feeding
birds gather in flocks and arrange coordinated cooperative hunt in sea. These
birds can dive to 30-40 meters and stay in water for 10 minutes, but at fast
swimming they inhale fresh air every 2-3 minutes.
Breeding takes place in spring, after returning from wintering places. Breeding
pairs at these birds form only to one breeding season. Displaying males utter
loud hoarse calls and swim in water, following the female ready to breeding.
Sometimes the combats take place between competing males, but females are choosy
and may form a pair with males they like, despite of their success in combats.
These birds breed in islands of the Bering Sea, preferring nesting in places
inaccessible for land-dwelling predators. They construct their nests in rock
cliffs and are able to climb on rocks skillfully. Nest is made of dry seaweed
and grass, and clutch numbers up to 6 eggs of greenish color with brown speckles.
Incubation lasts for about 35 days. The youngs hatch well-developed with opened
eyes. Being about two hours old, they jump off the cliff and fall; their small
size, feathery down, and very light weight helps to protect them from serious
injury when they hit the rocks below, but some of them die. After jumping they
walk to the sea with their parents. Juvenile feathering is of dark grey color
with darker head and white belly. Young birds become mature at the age of two
years. Life expectancy is about 20 years.
This genus of anserine birds is relatively common in the northern part of Pacific.
There are some species of these birds at the Pacific coast of Old and New World.
Asian coast is populated by Asiatic
sea merganser (Thalassomergus asiaticus) – non-migrating species very
common in Sea of Okhotsk, Big Kurils, Sakhalin and Japan Archipelago coasts.
It has a physical appearance more similar to yallal, being 72-76 cm long, but
heavier compared to it. It differs from yallal in robust constitution, lesser
ability to fly and prominent green shine of feathering at males. Females are
very similar to ones of yallal species, and sometimes in northern part of the
range mixed pairs form. This bird prefers benthic prey and often eats crabs,
shrimps, gobies and small flatfishes.
Warmer waters of Pacific are inhabited by three island species having no seasonal
rhythm in nesting:
Dwarf
sea merganser (Thalassomergus nanus) is geographically isolated form
inhabiting Hawaii. It is a smallest of the genus, with 40-45 cm of length. It
walks well and prefers to nest in bushes of small rocky islands not populated
by rodents and land-dwelling predators. It has bright red bill and paws at both
sexes. Primary feathers of wings have white tips, and it also has white spots
on shoulders. These birds are good flyers and are able to swim fast, jumping
from under water like penguins.
Californian
sea merganser (Thalassomergus californicus) is also geographically
isolated from forms of northern part of Pacific Ocean. This species from California
Island is remarkable in its large size (80-100 cm long) and strong bill. Its
color is less contrast compared to yallal – light grey belly and dark grey back.
Males have chocolate-brown heads and thick orange beaks. It is a poorly flying
species able to keep in air no more than 10-15 minutes. It feeds on large fish
and squids.
Atarapan
flightless sea merganser (Thalassomergus atarapaensis) is the biggest
representative of the genus, being 120-150 cm long. Life in isolation made it
flightless; its wings are short and robust, used for underwater “flight” like
at auks. Its beak of pink color is long and has rather large “teeth” near to
tip. Body coloration is less contrast – entirely grey with dark speckles on
upper part of the body. Heads are black at both sexes, sexual dimorphism is
poorly expressed – males are larger and have loud vocalization. This one is
very territorial bird; each breeding couple protects its feeding area from neighbours.
These birds are adapted to fishing in coastal reeves, pulling fishes and invertebrates
from crevices and cracks of the reef.
This bird was discovered by João Vitor Coutinho, Brazil.
Crowned
screamer (Chauna coronata)
Order: Anserine birds (Anseriformes)
Family: Screamers (Anhimidae)
Habitat: savannas of the central part of South America.

Picture by Pavel Volkov
Many biomes changed in the Neocene. In South America, the
great wetlands of the Pantanal disappeared, being replaced by savannas. The
species that lived here were forced to adapt or would become extinct. The Southern
screamer (Chauna torquata) became adapted to these changes. Its descendant,
the crowned screamer, is now capable of surviving in the savannas of Central
South America, while occasionally visiting shallow lakes and rivers.
The crowned screamer is one of the largest flying birds of southern South America,
measuring 95 cm in length. Their flat wing measures 60 cm and the tail is 26
cm. They are stout bodied with a disproportionately small head and a “chicken-like”
grayish brown bill. The sexes have the same plumage. Their head and upperparts
are black with a velvety white “collar” around the base of the neck. Their wings
and tail are dark brown and the wing has two sharp spurs at its manus, which
are longer than that of their ancestors. The front of their neck, their breast,
and their sides are pale gray mottled and streaked with black. Their belly is
unmarked white. Their legs and feet are red. Their main characteristic is a
crown made up of long white feathers with black tips arising from the nape.
This species feeds on leaves, stems and seeds that it collects in the grasslands
of the savannas where it lives. Occasionally, they will also visit shallow lakes
and rivers to eat aquatic plants. They usually graze like geese, but can also
dig for food. Flocks of up to about 100 forage together in the non-breeding
season.
The crowned screamer forms long-term pair bonds that in some cases last for
life. Males and females court with mutual preening and duet calling. They build
a large nest of sticks and reeds on trees and often nest in about the same location
for several years. The typical clutch size is three to five eggs but can be
up to seven. Both parents incubate the eggs and care for the young. The incubation
period is 43 days, with the chicks leaving the nest immediately after hatching,
jumping from the trees. The fledging occurs eight to ten weeks after hatch and
the young are independent after about 12 weeks. This species reach sexual maturity
with one year and have a lifespan of 13 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Genefal
(Acrogallus otidocephalus)
Order: Gallinaceous birds (Galliformes)
Family: Guinea fowls (Numididae)
Habitat: Karoo and Cape Province, arid areas.
Due to domestication and partly to a wide range in the Holocene, guinea fowls
managed to survive the anthropogenic crisis with relatively small losses. Of
course, the forest species of the family have suffered greatly from the economic
activity of people. But the helmeted guineafowl (Numida meleagris) bred by humans
easily went wild and even spread more widely than in the wild before domestication.
Among its descendants there are also large species, for example the Madagascar
numidornis. In the south and southwest of Africa, there is a related
species – genefal (the name in Afrikaans means “guinea fowl”).
Genefal occupies an ecological niche of large terrestrial birds like bustards.
The body length of an adult is 95 cm, the weight is up to 16 kg, and the wingspan
is about 2 m. The bird spends almost all the time on the ground and runs fast,
escaping from enemies, but takes off only if absolutely necessary and does not
fly for long. The plumage color is grayish-red with small white speckles on
the wings, back and short tail. Naked head is bluish in color; the shape of
the head of the genefal resembles a bustard’s one – it is elongated and flattened.
Only pink skin wattles in the corners of the beak and a flat helmet-shaped crest
on the back of the head make the origin of the bird clearer.
Genefal is omnivorous: the bird eats seeds, stems and roots and bulbs dug out
with its paws, as well as arthropods, small reptiles that it tramples, and even
fresh carrion. Genefal is able not to drink for a long time due to the production
of metabolic water, but when the opportunity arises, it drinks willingly and
to the full. These birds live in flocks of 20 individuals, where there are 2-3
males (one of which is dominant), females and young. A fairly rigid hierarchy
is maintained in the flock; the dominant male or the main female seriously shakes
the violators with its beak, pinching the featherless skin on neck, and kicks
them. Sometimes serious fights break out for rank in the flock when one bird
tries to displace the other one.
Courtship season begins at the end of the rain season. At this time, the wattles
of males swell and become large and red, and a collar of long white feathers
grows on the neck. At this time, males become especially aggressive, spending
a lot of energy protecting the harem from young and cheeky rivals. Subdominants
usually manage to mate with some females while the leader drives away young
birds or is engaged in mating.
These birds nest on the ground, among the bushes. The nest is an ordinary hole
in the ground with a litter of grass. The male arranges a “dust bath” for the
females near the nesting sites, carefully tearing out the greensward on a small
plot of land. There are 3-4 large eggs with a greenish shell in the clutch.
Incubation lasts 35 days. Females hatch, and males from the flock are certainly
nearby and patrol the boundaries of the nesting site. Only one of them can go
further for food, but then returns quickly. The chicks are well developed and
follow the mother as soon as they dry off. At 1.5 months they are covered with
juvenile plumage, by 3 months they are already flying well. By the age of two
years, young genefals are expelled from the parent flock and stay together for
a while. They join other groups of relatives, or conquer their own females from
a ready-made harem. Sexual maturity occurs at the age of 2 years, although males
can start breeding a year later. Life expectancy is up to 30 years. The natural
enemies of this species are large predatory mammals and birds, as well as monitor
lizards.
This bird species was discovered by Nick, the forum member.
Fan-tailed
Quail-Grouse (Notogalloides specula)
Order: Gallinaceous birds (Galliformes)
Family: New World Quails (Odontophoridae)
Habitat: open country such as meadows, grassland, scrub, bush and sparse forest,
North and South Island, New Zealand.

Picture by Pavel Volkov
New Zealand was styled as a “hunters paradise” during human
occupation in the Holocene, here, many forms of game were introduced to satisfy
a very popular hunting sport. These included deer, feral pig, pheasant and quail.
It is the California Quail (Callipela californica) that descended into the most
common genus of game birds in Neocene New Zealand, the Quail-Grouse.
Fan-tailed Quail-Grouse appears in many ways a typical game bird, but is very
large when you consider that it is a quail, it reaches 55 centimeters long from
head to tail, and weighs up to 760 grams. In shape and size, it more resembles
a grouse or small pheasant. Males differ in overall colour, bright reddish brown
with a bluish breast; feathers along its sides are each striped with black and
white, with a black erectile crest on its head, unlike the overhanging crest
of its ancestor, the California Quail. Tail feathers are enlarged in the male
to form a short, broad fan that is used in mating displays, marked with contrasting
thick bars of reflective bright orange. Females lack the ornamental tail fan,
and have a body colour resembling a paler, duller version of that of the male.
As with all game birds, legs are strong and well developed, and the bird usually
only takes flight when alarmed.
These birds occur in mid-sized flocks and spend almost all of their time foraging
on the ground. They are omnivorous, feeding on a large range of foods including
insects, small vertebrates, berries, buds, leaves, stems and seeds. As with
all game birds they are diurnal, and shelter in trees or under vegetative cover
at night. Call consists of a piping clucking noise, and an alarm call which
is a loud piping chirp.
Courtship and nesting occur in the spring, from September to October, males
are a lekking species and try to form large harems of females to mate with.
Males perform an elaborate display in which they fan out their ornamental tails,
and spread their wings, puffing up their breast and producing a chuckling, piping
sound as they circle the female. Males may compete over females and the best
territory to display in, and fights consist of leaping and kicking. After mating,
the female makes a scrape in the ground, lined with leaves and twigs, in lee
of some piece of vegetative cover, the clutch consist of as many as 11 eggs,
laid over a 2 week period, incubation lasts up to 26 days. The chicks follow
the mother and grow quickly, being able to fly after about 17 days, and fledging
fully at 20 weeks old. Lifespan usually reaches up to 10 years.
Related species is the Ocellated
Quail-Grouse (Notogalloides ocellaris), which differs by darker colour.
With male coloration, body is chocolate brown chequered with grey and having
a dark grey chest, and the fan-tail is longer and marked with a reflective blue-green
“eyes” pattern. This species is found mostly in the North Island and the northern
half of the South Island, overlapping in range with the Fan-tailed Quail Grouse.
Smaller, more ornate form is the Ornamental
Quail Grouse (Notogalloides nanus), which is more secretive in habits,
and is found only in the sub-tropical forested areas of the northern part of
North Island. It is about half the size of the other species, males possess
a powder blue breast and blood red body, chequered along the sides in yellow-white.
The male’s tail fan is long, and is banded in alternating bars of reflective
red and blue, separated by strips of black. This form prefers densely-vegetated
areas and is very shy.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Common
austral grouse (Tetraoinus australis)
Order: Gallinaceous birds (Galliformes)
Family: New World quails (Odontophoridae)
Habitat: the forests of southern part of South America.
During the Holocene, the man introduced many exotic species to different habitats;
the ancestor of the austral grouse was one of these species. Their ancestor
was not a true grouse, but the Californian quail, due to evolutionary convergence
they acquired characteristics similar to the true grouses of North America.
Living in, the male austral grouse are 55 cm long and the female have 44 cm
of length. The male have a dark grey body plumage, black-and-white bridled head
pattern, black back and a greyish-blue belly. They have a curving crest or plume,
made of six feathers that droops forward (long and black in males, short and
brown in females) and long forked blackish tail with white undertail coverts.
Females and immature birds are mainly greyish-brown with a light-brown belly.
Both sexes have a black bill and relatively long grey legs.
Their diet consists mainly of seeds and leaves, but they also eat some berries
and insects. In some regions their main source of food are the seeds of trees
of the Genus Araucaria. These birds are not elegant fliers, however they sleep
perched in branches. Given a choice, they will normally escape on foot.
During the courtship, the male austral grouse do displays on a lek, each male
have a “personal space”. The males strut around their chose space, doing a display.
The display consists of the male posturing himself with the head near the ground.
Then, they start to move it from right to left showing their crests, simultaneously
they raising the tail feathers, showing their withe undertail coverts. After
that, they will raise their heads abruptly and whilst make a highly distinctive
mating call. When another male invade the personal space of other, a fight happens,
in this case the male will try to take of the crest of the rival, this way they
will not be capable to display for the females.
The female usually lays approximately 12 spotted eggs. The nest is a shallow
scrape lined with vegetation on the ground. Incubation lasts from 21–25 days,
usually performed by the female and rarely by the male. The chicks are precocial,
leaving the nest with their parents within hours of hatching. The male stay
with the female until the young leave, with 3 months. The young reach sexual
maturity with 1 year, with this age the young males acquire the coloration of
an adult male. Their lifespan is of 12 years.
In the forests of Tierra del Fuego another species lives, the Black
austral grouse (Tetraoinus nigrus). Their main difference to the other
species is that the males have an all-black plumage, with only a faint shade
of blue in the chest and brown undertail coverts. The females are similar to
that of the common austral grouse. The males have 52 cm and the females 41 cm.
This bird species was discovered by João Vitor Coutinho, Brazil.
Andean
mountain-cock (Montanogallus andinus)
Order: Gallinaceous birds (Galliformes)
Family: New World quails (Odontophoridae)
Habitat: Central Andes, mountain slopes.
During the Holocene the man introduced many species. In South America the Californian
quail was one of these species. In the Neocene these birds gave rise to two
new Genus: Tetraoinus, the austral grouse,
and the Montanogallus, the mountain-cock. The Andean mountain-cock is one of
the representatives of this Genus.
Living in the slopes of Central Andes, the Andean mountain-cock is a snowcock-like
bird, both sexes have 63 cm of length and a light brown tail. The male has a
dark brown plumage, a white chest, a golden scaled belly and a black head. They
also have a short curving crest, made of six feathers; the crest is nonexistent
in females. Females and immature birds are mainly brown with a grey belly and
white chest. Legs and beaks are grey in both sexes.
This species lives in higher altitudes. During winter, they descend to lower
altitudes and move around in convoy. When approached from below on a hill slope,
they move up, stopping every now and then to look at the intruder, but when
alarmed they fly away. The flight is swift and will often make a whistling call
in flight.
They feed on seeds, bulbs, grass shoots and invertebrates. During the feeding,
keep sentries and while resting in the middle of the day, one or more of adult
birds stay in high boulders and keep a watch, warning the flocks on the approach
of danger with loud prolonged whistles.
Their nest is a scrape, sparsely lined and sheltered under a stone or bush usually
on the leeward side of a bare hill, and avoiding ground with vegetation. About
6 to 10 spotted eggs are laid. The male stands sentinel while the female incubate
them for 27-28 days. Both parent birds accompany the brood and adults perform
distraction displays when the young are threatened, while the chicks crouch
or hide between stones. Often, families group together, into multifamily "communal
broods" which include at least two females, multiple males and many offspring.
Males associated with families are not always the genetic fathers. The younger
reach sexual maturity with 2 years. Their lifespan is of 27 years.
In the Andean Altiplano lives the White-headed
mountain-cock (Montanogallus leucocephalus). Their biggest difference
when compared to the Andean mountain-cock is the white head of the males. They
have 58 cm of length.
Another species, the Blue-chested
mountain-cock (Montanogallus cyanopectus), lives in the slopes of the
Andes in the extreme south of South America. The main characteristic of this
species is the grey-blue plumage in the chest of males. They have 60 cm of length.
The females of all species are similar, except that the female of blue-chested
mountain-cock has bluish-grey chest.
This bird species was discovered by João Vitor Coutinho, Brazil.
Heather
Azorean partridge (Azoturnix ericae)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: New Azora, mountain woods.

Picture by Tony Johnes
In human epoch Azores were a place of wintering of numerous
species of European birds. Therefore in epoch of congelation they became original
“refuge” for the rests of European ornithofauna. During the ice age Europe had
represented extremely inhospitable place. On the north it was held down by ice,
and in the south significant territories had turned to salt desert when Mediterranean
Sea had dried up. Rather acceptable to life conditions were kept on narrow strip
between glaciers and desert. Because of it some of migrating birds had simply
ceased to come back to Europe to summer, and had formed settled forms on Azores.
Among them there were quails (Coturnix) - unique true birds of passage among
gallinaceous birds. They have started to master new to them habitats, and in
Neocene epoch on the island New Azora some species of ground birds - their lineal
descendants –had already appeared. They differ from the ancestors enough to
be classified as the separate genus of Azorean partridges. All of them are united
with the common features of appearance: dense rounded trunk, moderately long
and strong beak, rather short wings and very short tail. Legs at all birds of
this genus are strong, with well advanced toes. Azorean partridges spend a lot
of time on the ground, and prefer to run away from enemies. They fly up only
in case of emergency.
The most typical and usual species of this genus is heather Azorean partridge
inhabiting thickets of heather and other bushes, covering slopes of mountains
of New Azora. It is a bird approximately of size of small domestic hen: its
growth is about 20 cm and weight is about 400 grams. Feathering of this bird
is cross-striped, yellowish brown with black strips. On wings strips are denser
and wider, that’s why wings seem darker. Legs of this bird are colored bright
orange. At males in courtship season legs get an appreciable reddish shade.
All species of Azorean partridges have distinctive features of feathering -
bright and contrast spot on throat and head. Heather Azorean partridge has white
throat bordered by black feathers. This ornament is used for courtship display
occurring in spring.
Heather Azorean partridge is omnivorous: it equally willingly pecks seeds of
grasses, soft leaves and insects. Similarly to many gallinaceous birds, heather
Azorean partridges willingly rummage in ground, supplementing their diet with
insects and roots of grasses. Birds of this species outside of nesting season
keep in small flocks, constantly supporting contact to neighbours with the help
of sound signals. Voice of this bird is long, rolling, but monotonous warble.
More often heather Azorean partridges “sing” during the dawn, and the voice
of flight of these birds can be heard almost at half-kilometer distance.
In courtship season heather Azorean partridges gather on lekking ground – places
on edges of bush thickets. The courtship display begins early in the morning
when there is a rich fog above bush thickets, and the sun had risen only a little.
Males gather on lekking ground the first. They keep at some distance from each
other, from time to time rising on legs almost vertically. At this moment they
puff up throats, and black feathers bordering white spot, are spread wide in
sides. If it becomes too much males on lekking ground, strongest males drive
off competitors and continue demonstrations. Gradually their movements synchronize,
and heads of males start to be shown from a grass almost simultaneously. Females
observe of courtship displays of males from bushes, and appear on lekking ground
when courtship displays reach apogee. Males start to chase females coming nearer
to them, continuing demonstrations in the moving. After several minutes of chase
the female strikes an attitude of submission, short pairing takes place, and
the male loses interest to it, continuing demonstrations.
The nest of these birds is arranged on the ground. In clutch there is up to
ten eggs with brown spotty shell. Only the female hatches them within approximately
18 days. Chickens hatch with opened eyes, covered with rich striped down. For
the second day of life feathers on wings start growing at them, and the ten-day
chicken is covered with monochromic juvenile feathering. The next year young
birds are able to take part in courtship displays and to give posterity.
On New Azora other species of Azorean partridges live also. These birds occupy
different habitats, and between them there is no competition.

Picture by Lambert
Regal
Azorean partridge (Azoturnix regius) inhabits warm lowland woods of
New Azora. It is the largest species of genus - the adult bird weighs up to
2 kg (the male up to 2,5 kg). The basic food of these birds includes fruits
of trees falling on the ground. Also regal Azorean partridges eat seeds and
grassy plants. They willingly feed with rhizomes of ferns, digging them out
by strong paws.
It is the most brightly colored species of genus. At adult birds feathering
on head and the top part of body is dark red. At edges of feathers there is
the black border giving to colouring of bird “scaly” pattern. The bottom part
of body at females is brown with short light strokes (along the middle part
of ever feather the yellowish strip passes); at males a stomach is black. Also
on throat of the male black feathers with strong green metal shine grow. Primary
feathers of males have white tips. This bird is monodin; pair rears posterity
in common. The male mainly drives off contenders and preserves hatch against
small ground predators, and female is occupied with incubating of eggs and care
of chickens.
In the morning birds of this species begin loud “muster”. The voice of this
bird reminds eagle squawk repeating by series of 4 - 5 “syllables”.
Rock
Azorean partridge (Azoturnix rupestris) is the smallest species of
this genus. By the size this bird is like a grey partridge (Perdix perdix).
Similarly to regal Azorean partridge it also has “scaly” pattern on body, but
this bird is colored much more dimly: back is grey with narrow black borders
on feathers, stomach and area under tail are white. The throat is silvery-white,
bordered by strip of black feathers. It is high-mountainous species does not
forming big congestions. Usually birds keep one by one, and in courtship period
only occasionally it is possible to see more than ten birds simultaneously.
Rock Azorean partridges eat mainly seeds of grasses, juicy bases of leaves and
roots.
Moss
Azorean partridge (Azoturnix bryophilus) lives in moss bogs and on
border of bush thickets in mountains, but along bogs in flood-lands of rivers
this species settles down to mountain valleys. This is medium-sized bird: it
weighs about 1 kg. Colouring of this bird is typically cryptic: back is brown
with light yellow longitudinal strips; sides are lighter. On throat there is
an area of red feathers with strong metal shine. Voice of this bird is terse
lingering cry. This species keeps in flocks numbering 5 - 6 birds.
Moss Azorean partridge is very cautious bird, and in case of necessity it is
able to mask skillfully in grass and bush, nestling against the ground. As against
other species of genus, it prefers to eat insects and other invertebrates, and
digs them out from the ground not by paws, but by strong sharp beak.
Basan
(Gymnocoturnix basan)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: mountain forests of the Japan Islands, undergrowth.
Among gallinaceous birds, not all species managed to survive the mass extinction
of the end of Holocene – species from tropical forests and human-developed territories
suffered the most. Among them there was the Japanese quail (Coturnix japonica),
from which a large forest species – basan (a giant bird breathing cold fire
and similar to a rooster, from Japanese mythology) – originated in Neocene.
This inhabitant of mountain forests is a large bird of Japan Archipelago. The
body length is up to 80 centimeters. The plumage of the body and wings is ochre-brown
with black spots on the wing coverts. The tail is short, visible only a little
from under the folded wings; in the female it is of the same color as the trunk,
in the male it is black, and it also has a white undertail. The legs are featherless,
red, long and strong; males have spurs. Head and neck are bald, the males have
leathery red “eyebrows” and a throat fold of skin – a “beard”. Beak is thick
ans deep, with a slightly curved tip, of coal-black color. Basan flies reluctantly
and only when necessary, but runs very well. During the take-off, bird flaps
its wings very loudly.
Basan feeds on various plants, buds, leaves, herbs, berries, fruits, seeds,
taking part in the distribution of seeds of some species of trees and bushes.
In addition, the bird eats small vertebrates and various invertebrates.
The bird escapes from large predators in the undergrowth, often running through
dense bushes and abruptly changing the direction of run. Birds with a brood
can protect their offspring by attacking the enemy and striking with spurs and
claws.
Courtship displays at basan begin in March, when the snow melts in the mountains.
During the display, males gather in groups, call “vake-vake” loudly and flap
their wings, attracting females. Displaying male opens its tail and bends it
on the back, showing a fluffed, well-visible undertail. At this time, its “eyebrows”
become very bloodshot, becoming like horns, and the throat fold is greatly lengthened.
In the displaying pose, the male utters continuous buzzing sounds. Fights often
occur between displaying birds; at this time, males peck and beat each other
with spurs, but fights usually do not lead to severe damage to rivals. These
birds are polygamous; there can be up to 3-4 females in a male’s harem.
The female makes a nest on the ground among bushes, lining it with forest debris
and grass. It lays 13-15 light yellow-brown eggs with black spots and an individually
variable pattern. The female incubates them for 26 days. Like its ancestor,
basan is a precocial bird. Chicks are light brown with a longitudinal stripe
on the back and head, with small light and dark spots. In the process of plumage
growth, juvenile fluff sheds, and the head and neck become bare, but have a
gray color. The female takes care of the chicks for about 2 months. By August,
young basans begin to lead an independent life. By the spring of the next year,
they become sexually mature and get adult plumage color and skin outgrowths
on the head.
Basan males live up to 12 years, and females about 14 years.
This bird species was discovered by Feldwebel, the forum member.
Meganesian
Jungle Fowl (Gallus australis)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Rainforests and forest margins in the north of Meganesia.
Domesticated food animals frequently became feral in the wake of man, island
continents such as Australia were badly affected by this. Relatively few kinds
of introduced bird left descendants in Neocene Meganesia, with some exceptions.
Meganesian Jungle Fowl is a descendant of run wild domestic chickens initially
used by man for meat and eggs. This species greatly resembles the modern Red
Jungle Fowl more so than a domestic chicken, sexually dimorphic, males being
ornate, and females being a speckled brown. Male of this species differs from
the ancestral species in that its tail plumes are bluish iridescent green, the
wattle and comb are purplish.
Habits are very similar to the current jungle fowl; these birds only take flight
to escape predators or to roost in trees at night. Individuals frequently take
dust baths for personal hygiene. Diet includes mainly fallen fruit and grain,
as well as insects, spiders, lizards and small rodents, ungulate droppings are
sometimes eaten. Dominant males keep small territories which they defend vigorously;
method of fighting is with leaping and fierce kicking, as with its ancestor.
There is a typical pecking order within a flock, with the dominant male servicing
the females fighting off other males. Males crow in the early morning hours
to proclaim territory.
Breeding occurs during the dry season mainly, and the male begins courting a
female by picking up pieces of food for her and clucking, female accepting the
food allows the male to mount. After the laying period begins, the female lays
one egg per day, and the eggs incubate within 20 days. Chicks are precocious
and follow their mother; they fledge by 5 weeks and reach sexual maturity at
5 months. Lifespan is up to 10 years and predators include carnivorous marsupials,
snakes and raptorial birds.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Flag-tailed
grass pheasanet (Herbogallus vexillifer)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: savannas of Northern Africa, Gibraltar isthmus, Western Europe.
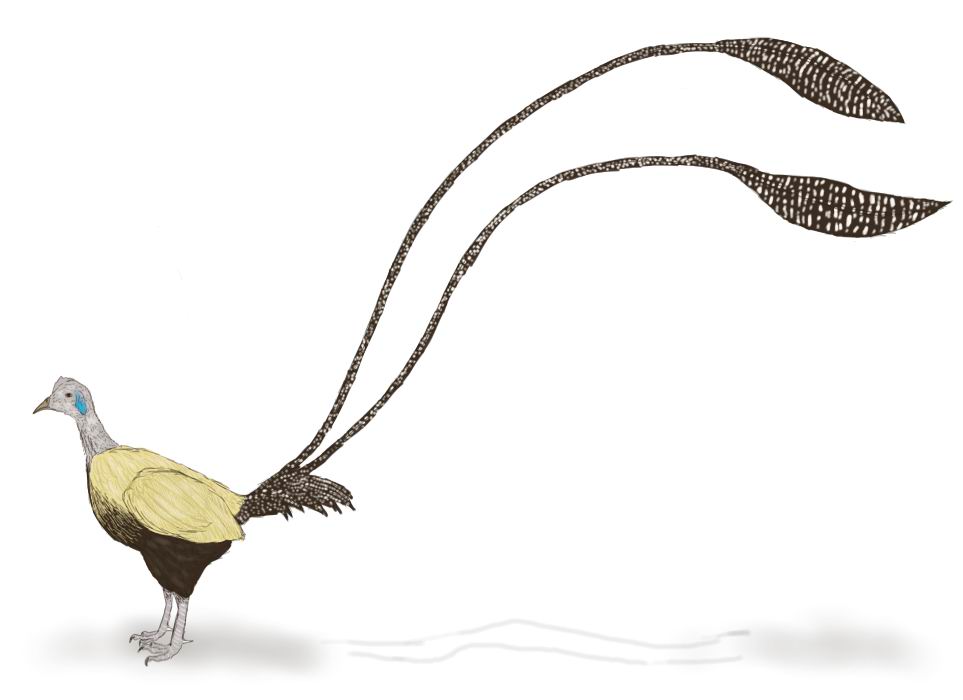
Picture by FanboyPhilosopher
In various places of Neocenic Earth there are species of animals
descending from feral domestic species. Usually they are mammal though occasionally
there are descendants of domestic birds among them. The reason of unsuccess
of domestic birds in struggle for existence is that the most part of domestic
breeds of birds had too strongly changed in comparison with wild ancestor. Their
dependence in relation to human had increased: many breeds of hens bred industrially,
had lost ability to hatch eggs. Also some other domestic birds had lost the
parental instinct. But in areas where intensive poultry farming had not developed,
there had been populations of domestic birds able to independent existence and
reproduction. In human epoch populations of wild species of gallinaceous birds
had strongly reduced and even had disappeared as a result of hunting and destruction
of habitats. But after human disappearance his former pets which could adapt
to wild life, began to take a place in formed ecosystems of Neocene. In tropical
areas there were domestic hens among them.
Living in savannas of Africa grass pheasanets are descendants of feral domestic
hens (Gallus domestica). They form the special genus of gallinaceous birds which
had adapted for life in plain district overgrown with grasses. Their appearance
is rather remarkable: the constitution of these birds is graceful; at them there
are long legs and neck. Growth of birds of this genus varies from 30 cm up to
half meter. Adult birds weigh from 0.6 up to 2 kg, males are larger than females.
Head of grass pheasanets is naked and at large species neck is also naked. This
is the effective adaptation for emission of excessive heat: in heat weather
blood vessels dilate, and surplus of heat is emitting from the organism.
Colouring of feathering of males at all species is bright and multi-coloured,
with metal shine; females are colored more similar to each other, to not striking
brownish shades with more – less expressed cross strips. Such colouring permits
birds to mask in grass during the posterity hatching. Bright colouring raises
probability of death of male in teeth or claws of predators, but it is not resulted
in reproduction of species: all grass pheasanets are polydins similarly to their
ancestors, domestic chicken.
Wings of grass pheasanets are short and rounded: birds are able to fly, but
do it reluctantly. The frightened away bird prefers to run away, dexterously
moving among grass and sharply changing direction of run. In case of necessety
grass pheasanet can fly up, loudly clapping by wings. Such sound is an alarm
signal for majority of herbivorous inhabitants of savanna.
Males of grass pheasanets are as pugnacious, as domestic cocks. At them on legs
sharp straight corneous heels develop, with which birds fight in courtship season
and strikes smaller predators like snakes or lizards. Females are more tolerant
to each other, but also may arrange noisy fights. But, despite of aggression,
grass pheasanets support contact with each other. Loud voice serves to them
for this purpose. Males of these birds from time to time utter the cry similar
to resonant call heard from apart. And for maintenance of visual contact the
magnificent tail, or, exactly, feathers of uropygium, serves to them. The tail
of grass pheasanets is insignificant – no longer than half of length of body.
But the central pair of uropygium feathers at these birds exceeds four times
in length growth of adult bird. Shafts of these feathers are strong, and the
basis of tail is very movable. Due to it the bird can lift feathers of uropygium
vertically.
The vane of long uropygium feathers is expanded at the tip to original “flags”
(approximately for the one fifth of length of the feather) and brightly painted.
Colouring is supplemented with metal shine. At various species colouring of
“flags” is different due to what males of different species do not clash in
vain. Showing aggression to the congener, the bird trembles by tail because
of what “flags” shake, and solar beams sparkle on them, making their owner more
appreciable to the opponent. At females “flags” are less expressed, and colouring
of tips of feathers is not such bright. Till the season of egg hatching females
cast these feathers at all.
Flag-tailed grass pheasanet is the typical representative of this genus. It
reaches height about 40 cm at weight of the male about 1.25 kg. Females of this
species are smaller than males. Head and neck at this species are naked; on
the crown there is a small crest-like outgrowth. Ear lobes are large and colored
bluish. In courtship period at males they get bright blue color.
The male is colored brightly: it has black stomach, straw-coloured feathering
on body and wings. Tail of the male is black with small white speckles. “Flags”
of covert feathers of tail reach the length of one and half meters. Vanes of
“flags” are colored black with large white spots merging to rough cross strips.
Displaying male gathers a harem of approximately ten females. It looks after
them, drives off contenders and from time to time arranges “show” for them.
In displaying time he lifts the tail up highly, having bent it forward, and
lifts opened wings, showing white sides. “Flags” are shaken before a head of
displaying male. He trembles by all body, and then “dances” around of the chosen
female, “drumming” fast by legs on the ground.
After courtship games the female retires and rears posterity independently.
For nest arrangement the female chooses dense bush or old burrow under tree
roots. In clutch of this species there are about ten small eggs with brown shell.
The incubating lasts about 20 days. Chicks hatch well advanced and independent.
They have longitudinal-striped colouring, and can hide from predators in grass.
They become sexual mature at the age of 10 months.
Other species of grass pheasanets live in Africa and Southwest Europe:
Dwarf,
or flame-tailed grass pheasanet (Herbogallus flammeocaudis)
is the smallest species of the family. Its growth is about 30 cm, and weight
of the male is only 600 grams. This bird inhabits grassy plains of Northern
Africa and keeps in places where grass and bushes are thined out by large herbivores.
Male and female of this species are colored similarly in general: they are brown
with cross black strips. But at the male wings are straw-coloured with shining
feathers on bends. At males the skin on head is naked, colored bright pink;
at females head is feathered. In its tail there are two long feathers forming
“flags”. Shafts of these feathers are covered with thin long barbs as at the
peacock, but only on one side. “Flags” are colored bright orange with strong
metal shine, shaded from below by black color.
Fan-tailed
grass pheasanet (Herbogallus flabellicaudis) is the largest species
of the genus: growth of adult male reaches half meter, and the length of its
tail “flags” exceeds one and half meter. This bird lives in high grass and among
bushes in Southwest Europe and Northern Africa. At birds of both genders head
and neck are naked; male differs from female in the large white ear lobes hanging
down like ear-rings. It also has light yellow feathering on back and wings,
and rusty red stomach. “Flags” on tail of the male are black with white tips
- such colouring is better visible on the background of drying up grass. Female
of this species is also a little lighter, than at close species.
This species of grass pheasanets differs from other ones by structure of tail:
except for two “flags” at it there are some more pairs of elongated feathers,
also having small expansion of vane on tips. During courtship games the male
spreads wings and inclines body forward. Also he opens tail like a fan, and
spoiled long feathers quiver at each his movement. Displaying male turns around
on the spot, involving the female, “cooing” for a long time and nodding head.
Ear lobes of the male are poured by blood, strongly increasing in sizes, and
touch the ground at his bows.
Bornean
deinogallus (Deinogallus borneoensis)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Kalimantan Island, tropical forests.
Man has domesticated a number of animals, and in process of selection has bred
a number of breeds that are not adapted to life in nature. Among such animals,
most breeds of domestic chickens turned out to be – breeds turned into “egg
production machines” with a lost hatching instinct or rapidly gaining weight
have become the most widespread in agriculture. After the human disappearance,
such breeds were doomed to extinction. But in addition to these breeds, in areas
of extensive agriculture, there were various unproductive populations of domestic
chickens close to the wild ancestor. After human disappearance, they adapted
rather easily to life in the wild. Perhaps in Southeast Asia and Indonesia they
mixed with the rests of the wild population of the ancestral species – red junglefowl
(Gallus gallus). The genetically heterogeneous population turned out to be quite
viable, and in Neocene in Southeast Asia and on the islands of Indonesia there
are many species of pheasant-like birds descended from domestic chickens. One
of the peculiar species of these birds is deinogallus, a peculiar cassowary
analogue.
The body length of this bird is up to 150 cm, the weight is about 30-55 kg (males
are larger than females). At this weight, these birds cannot fly, but their
wings are not reduced and retain their normal structure. Despite its large size,
deinogallus is a bird of light build, slender, agile and strong. In its constitution,
it somewhat resembles an ostrich. Sexual dimorphism is pronounced in these birds.
The background color of the female’s plumage is brownish, tips of feathers on
the neck and back are black, wing coverts are ochre-red. Female’s tail is long
and narrow, a bit like a shortened pheasant’s tail. Male has well-developed
narrow reddish hackle feathers, the background color of the body is reddish-brown;
tail consists of long elastic black feathers with a blue metallic shimmer and
white tips. Uppertail coverts are very thin and silky, black, constantly hanging
down. Male has a broad fleshy outgrowth on its head, similar to a helmet with
slightly raised edges; in females, this outgrowth is weakly expressed – most
often it is an outgrowth, similar to a walnut in shape. Large spurs grow on
the legs of birds, especially well developed in males. The male’s voice is a
long-drawn-out sound, similar to a howl or a buzz.
These birds are omnivorous and indiscriminate in food; they can peck any object
that seems edible for them: fruits, mushrooms, many flowers and leaves, large
invertebrates and small vertebrates, carrion (clearly preferring fresh one),
bones and pieces of skin remaining from the prey of large predators. These birds
regularly swallow the stones needed to grind food. Deinogallus lives in groups
of 6-8 adult birds: a male and his harem, as well as chicks of the current year.
Chicks of the first months of life are able to fly and are able to take off
for the night on tree branches 2-3 meters above the ground. Young birds that
have not reached maturity gather in herds of up to 20-30 individuals, where
the number of males and females is approximately equal. They are very careful:
deinogallus is an aggressive bird; the male regularly shows his power to females
and juveniles. Sometimes he even attacks local herbivorous animals and drives
them away from his harem. The enemies of adult birds are large cats and monitor
lizards, the chicks are stolen by snakes and small predators. These birds flee
from large enemies: they run at speeds up to 50 km/h, often making sharp turns
and changing the direction of run.
The breeding time of deinogallus falls on July-August. At the male by this time
feathers on the neck and tail develop in full degree. Displaying male crouches,
spreading his wings, and makes grumbling sounds. Usually, females remain faithful
to their male, and the male is not required to do more to express his feelings.
But this kind of relationship is built exclusively on force demonstration. Young
and widowed males display more intensively: from time to time they stretch their
bodies almost vertically, open their wings and begin to flap them forcefully
for several seconds. The tips of wings clap loudly against each other behind
the back and in front of the belly of the bird, and this sound is audible at
the great distance in the forest. Such leks of young birds may be recognized
from a distance of one kilometer. Sometimes a young and ambitious male tries
to recapture a harem from an adult one. This often results in a fight, but sometimes
he manages to win one or two females from the harem. More cautious males approach
the harem of a mature male without challenge, and they manage to mate quickly
with one of his females.
These birds nest on the ground, arranging several nests nearby to each other.
This tactic helps to avoid the ravaging of nests by monkeys and small predators.
Two or three females lay eggs in each nest; each bird lays up to 7-8 eggs with
a dark green speckled shell. They alternately incubate the clutch and jointly
protect the nest from enemies. At this time, the male guards the boundaries
of the nesting area and attacks trespassers. Incubation lasts 40 days; chicks
hatch well developed; they are covered with gray down with black longitudinal
stripes. All females of the harem take care of the brood, making no distinction
between their own and other’s chicks. At the age of one year, young birds leave
the flock, because adults begin to show aggression towards them. Sexual maturity
occurs at the age of 3 years, upon reaching the average weight of an adult bird.
Life expectancy is no more than 20 years.
This bird species was discovered by Nick, the forum member.
Emperor
junglefowl (Gigantogallus imperator)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Nicobar Island.
During the Holocene, the man introduced many species to other continents. Some
only because of their ornamental value, while others were introduced because
they were useful for agriculture. Among the birds, the domestic chicken (Gallus
domesticus) was the species that reached more continents. With the disappearance
of humanity, they were left behind to fend for themselves. Some were successful
in surviving and continued to evolve. In the Neocene, the chickens that were
left in Nicobar Island had evolved into a large flightless species, the Emperor
junglefowl. Such characteristic was only possible due to the disappearance of
terrestrial predators in the island.
The emperor junglefowl is the largest animal of Nicobar Island. A completely
adult male can reach 175 cm of height, surpassing their relative of the Genus
Aepygallus from Mauritius. The female has a more modest size, 147 cm.
These birds are also heavy, with the male having 32 kg while the female has
23 kg. To sustain such weight, their bones had evolved to be denser, like that
of an ostrich. They have a large skull with a deep and laterally compressed
orange beak. Its yellow colored legs are rather short, but have strong toes
with long claws. Most of their height comes from their long necks. The wings
are reduced, making it impossible for them to fly.
Beyond the size, the only other difference between the sexes is that the males
have a bare face with red skin that forms a comb and wattles. These bare parts
become bright during the nesting season. The comb is large, reaching 20 cm in
older males. The wattles are long and come down from both sides of their head,
below the auricular hole. This gives the bird the appearance of having earlobes.
The female lacks such structures and the skin on their face is pale pink. Both
sexes have the same dark brown plumage, with yellowish-brown underparts. The
tails are short, being metallic green in the males and brown in the females.
The emperor junglefowl is omnivorous. It'll feed on arthropods, terrestrial
gastropods, seeds, fruits and leaves. They use their long necks to reach fruits
and leaves in tree branches. These birds will also visit beaches in search of
algae and shellfish, which is excavated out of the sand. Their shell is crushed
by the birds' strong beak.
This species lives in harems formed by a dominant male and two to four females.
Their nesting season starts during the end of the dry season. The females will
make nests in the territory of the group, usually hidden among bushes and below
trees, near their roots. Usually three to six reddish-brown eggs are laid, which
are incubated solely by the female for 30 days. The male will stay patrolling
his territory in search of possible rivals and invasor, which will be expelled
with kicks of their strong legs. After hatching, the chicks are already capable
of leaving the nest. They will follow the mother back to the group and if other
females already have chicks, a crèche is formed by them. The young reach full
adult size in about six months. At this point, the male chicks are expelled
by their father. The females can remain indefinitely, but they leave as soon
as sexual maturity is reached with two years of age. At this point, they will
leave, with some even traveling in small female-only groups in search of a male
to form their own harem. The lifespan of this species is 15 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Terpsychornis,
“dancing chicken” (Terpsychornis ludio)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: steppe and semi-desert areas of Europe south of the Alps, northern
and eastern edges of Mediterranean lowland.
In the human era, domestic chickens were among the most numerous birds on the
Earth. However, many of the breeds of these birds depended too much on humans
and lost the ability to reproduce independently and defend themselves from enemies.
Therefore, the human disappearance of man was accompanied by the mass disappearance
of domestic chickens (however, the same fate befell many other domestic animals).
Primitive populations of chickens from the Middle East, retained the ability
to reproduce independently and search for food, managed to survive and develop
new habitats. In arid areas in southern Europe, where forests are found only
in the valleys of rivers flowing down from the Alps, one of the descendants
of domestic chickens – terpsychornis, a peculiar “dancing chicken” – lives.
This species is a bird of light and slender build: the height of an adult bird
is about 70 cm, the wingspan is up to 1.5 m, and the weight is about 10 kg.
The legs of terpsychornis are quite long, armed with spurs, which present in
both males and females. Unlike some other descendants of chickens, terpsychornis
has retained the ability to fly – it escapes from dangerous predators by taking
off into the air. But this is rather an extreme, since these birds are able
to run fast, and if necessary, they can deftly hide in the grass.
The main color of the plumage is gray-yellow; the male almost does not differ
from the female in the color of the plumage. Male is larger than female; wide
skin wattles grow on the sides of his beak, but the crest on his head is rather
poorly developed. In females, the “wattles” are much smaller. The tail is narrow,
straight and elongated. This species lives in small groups: male, 2-3 females
and the recent brood of chicks. Terpsychornis feeds mainly on vegetative food,
although it does not disdain insects. If it founds other food of animal origin,
it feeds on it: bird can peck at the remains of predator’s prey or stillborn
cubs of herbivorous animals. For successful digestion, terpsychornis swallows
pebbles. It easily beats dead a snake, lizard or rodent with its feet, and also
digs out roots with its beak.
Terpsichornis’ natural enemies are cats and large viverrids, foxes and some
owls catching adult birds. Young birds have much more enemies: even snakes and
large lizards eat them. But parents take care of their offspring selflessly,
striking the enemy with spurs.
Mating season takes place in April-May, immediately after the end of spring
molting. At this time, in open places far from possible shelters for predators,
leks are formed, where dozens of young and single males gather. After sunrise,
females gather on the outskirts of the lekking grounds and courtship displays
begin: males stamp their feet, jump up, run in zigzags and in a circle around
the females, “grumbling” from time to time and greatly inflating their throats.
After performing such dance figures, they squat and flap their wings – one by
one, then synchronously. Then the flapping of wings can be heard many hundreds
of meters away from the lekking ground. These mating dances are especially picturesque
and prolonged in young birds that do not have a harem – when they attract females
or try to recapture them from an adult male. The “family” male is usually limited
to a few “pas”, confirming his primacy. As a rule, females prefer such males,
but they can join his younger rival if his dance turns out to be longer and
more intricate. A “dance” performed entirely is a tedious competition with a
young rival, sometimes ending in a fight.
In the clutch, each female usually has 2-3 eggs laid at intervals of one day.
Incubation lasts up to 40 days. The nests of females from the same harem are
located nearby and the male additionally digs a “dust bath” for the females,
as well as patrols the territory and distracts predators. The chicks hatch out
quite developed and do not lag behind their mother within an hour after hatching.
Their down color is sandy yellow with several black longitudinal stripes on
the back. At first they feed on invertebrates, but in 1.5-2 months they completely
pass to an adult diet. Already at the age of one week, they can take off and
fly several meters. Young terpsychornises stay with their parents until spring,
after which they form groups of individuals of the same age. Being 3-4 years
old, birds begin to take part in reproduction. Life expectancy is about 40 years.
This bird species was discovered by Nick, the forum member.
Perfect
ayamornis (Ayamornis magnificens)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Southeast Asia, Jakarta Coast; tropical forests.
The most widespread of the domesticated bird species was red junglefowl (Gallus
gallus), eggs and meat of domesticated forms of which people ate. But, as a
result of artificial selection, a significant part of the chicken breeds lost
the ability to defend themselves from enemies and hatch eggs after the extinction
of humans was doomed to death. But primitive breeds from the tropical regions
of the Earth, less dependent on man, managed to survive and leave descendants
in the Neocene. They achieved the greatest diversity in Southeast Asia, where
these birds were domesticated, and where the greatest species diversity of pheasants
was observed in the human era. If in Borneo the descendants of feral chickens
turned into deinogallus, a peculiar
analogue of cassowaries, then on the mainland there lives an equally amazing
species – perfect ayamornis (“ayam” in Malay means “chicken”).
For this bird, as well as for arguses or peacocks of the human era, a strongly
pronounced sexual dimorphism is characteristic: males of this species, convergently
similar to some Japanese breeds of chickens bred by man, can reach up to two
and a half meters in length. At the same time, most of the length of the bird
falls on the tail; the mass of the male is about four kilograms, which is twice
the mass of the female. In addition to larger size, male is distinguished by
blue-black plumage on the back, sides, chest and tail, white primary feathers,
tail and hackle feathers on the neck, forming a kind of “mane”, as in chickens
of the human era. Bright red leather wattles and a comb grow on the head; head
skin is featherless and has dark gray coloration. Females look much more modest
– their tails do not reach such an impressive size, and the bird itself has
dusky brown plumage, which is due to the fact that the female hatching the clutch
should not attract the attention of predators. Wings are normally developed,
but ayamornis, like most other gallinaceous birds, flies poorly and reluctantly,
preferring a terrestrial lifestyle. In case of danger, the bird quickly takes
off and flies several tens of meters, but then lands and runs away into the
undergrowth. There are spurs on the legs, developed, however, not as well as
in the ancestor – sexual selection has led to the fact that the roosters do
not fight with each other, limiting themselves only to the displaying of their
tails. Only occasionally do they resort to power struggle, but they are limited
only to chest blows into the opponent’s body and pecks.
Ayamornis feeds mainly on herbs, seeds, leaves and fruits, less often on food
of animal origin: small vertebrates, insects and snails. Birds also swallow
the pebbles needed to grind food. These birds live in groups including a male,
two or three females and their recent brood, which has not reached maturity.
The breeding season of these birds falls on June and July. Males display, stretching
their wings and vocalizing loudly. The voice of ayamornis resembles the first
part of a cockcrow, turning into a ringing “stroke”. Usually, displaying males
with harems do not approach each other, and only try to protect the harem from
young and single males.
After mating, the female lays 8-10 eggs in a nest located on the ground. Each
female builds a nest and incubates her own clutch on her own. At this time,
the male patrols the territory, and in case of danger leads the predator away
from the nests, behaving boldly and not hiding. Incubation lasts about a month,
after which fully formed and down-covered chicks hatch from the eggs; they leave
the nest after a few hours and follow their parents. The downy dress of chicks
is gray with longitudinal black stripes on the back. The juvenile plumage of
males and females is the same, brownish in color with black speckles, only the
feathers on the wings of males are slightly lighter. During the first year of
life, males are similar to females. Ayamornises reach sexual maturity at the
age of 2 years; life expectancy reaches about 15 years.
This bird species was discovered by Anonim, the forum member.
Copper
jacumitan (Jacumita cuprina)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: savannas and forests of central part of South America.
During the Holocene, the man introduced many species in other continents. The
jacumitan is a descendant of domestic chicken (Gallus gallus domesticus) that
are introduced in South America. It replaced the birds in the family Cracidae,
which are extinct in the Neocene, in some areas. In a matter of fact, the name
jacumitan is the junction of the words jacu, the popular name in Brazil for
guans of the Genus Penelope, and the tupi word mitã (mitan) that means new,
this way the name means “new guan”. The copper jacumitan is the type species
of this genus and lives in the savannas and woodlands of Central South America.
The copper jacumitan is sexually dysmorphic. The male measure 190 cm, due to
the long tail, it has a bright coppery brown upperbody plumage and reddish-brown
feathers below. They also have metallic dark green feathers on the tail and
wings. Only males have a bright red naked skin on the face, with two wattles
that conceal the sides of its head. This characteristic is shared by all males
of the genus Jacumita. The males of the genus also have crests formed by white
feathers with black tips. During courtship and panic moments, they rise the
crests. The females have 75 cm of length and are cryptic brown and adapted to
camouflage, its naked skin on the face is pale pink and don’t have flesh wattles.
The young males are similar to the females until they reach 9 weeks, at this
time the wattle begin to develop. Both sexes have long orange colored legs and
grey beak. The males have spurs that they use in fights for dominance. The neck
is long and slim. Copper jacumitans can fly, but prefer to run from the predators
flying only as last resort.
These birds are omnivorous and feed on insects, seeds, and fruits. Its forage
for food by scratching the ground, usually near herbivores this way there is
other animals observing for the presence of predators. Often the male sits on
a high perch, to serve as a lookout for his group. He sounds an alarm call if
predators are nearby. At night it will sleep perched on branches.
Copper jacumitans are polygamous; the male will have a harem with 5 to 12 females
and will guards the area where his females are nesting, and attack other males
that enter his territory. To initiate courting, the males will dance in a circle
around or near a female, raising their crests and lowering the wing which is
closest to the hen. Then, he will vocalize and when she responds to his call,
the male will mount her and proceed with the mating. The females make their
nest in the ground, laying 6 to 14 red eggs which are incubated for 22 days.
Chicks are precocious, leaving the nest shortly they are born. They fledge in
about 4 to 5 weeks, and at 13 weeks old are chased out of the group by their
mother, at which point the young males start to form a harem and the females
join an existing one. Sexual maturity is reached at 6 months and the lifespan
of this species is of 13 years, however is common for males to die early due
to predation.
Other species in the genus Jacumita are:
Golden jacumitan
(Jacumita aurea)
Living in the forests of the Atlantic coast of South America, the males of this
species has 200 cm of length, from beak to tail, while the females have 83 cm.
Males have a bright golden-yellow plumage in the upperparts, being scarlet in
the chest and belly. They have dark metallic green wing feathers and tail while
the female is cryptic brown. This species prefers feeding on hard seeds and
fruits.
Red jacumitan
(Jacumita amazonica)
Living in the "terra firme" forests of the Amazon , the males of this
species has 185 cm of length, from beak to tail, while females have 70 cm. The
plumage of male is bright orange-red in the upperparts, while their chest and
belly is yellow. The males also have dark metallic blue wing feathers and tail
while the female is cryptic brown. The considerable part of its diet is made
of tubers and roots of herbaceous plants.
Andean jacumitan
(Jacumita andina)
Living in the highland forests in the slopes of the Andes, the males of this
species has 170 cm of length, from beak to tail, while females have 68 cm. The
plumage of the males is white in the upperparts and yellowish-red in the belly
and chest. Like in previous species, males also have dark metallic blue wing
feathers and tail while the female is cryptic brown. This species is omnivorous;
the food of animal origin, including small rodents, makes up to a half of its
diet.
This bird species was discovered by João Vitor Coutinho, Brazil.
Transalpine
(Southern European) pheasant (Euphasianus transalpinus)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: southern Europe – Balkans, Apennines and Pyrenees, southern slopes
of the Alps; forests, sometimes meadows.
In historical time, the common pheasant (Phasianus colchicus) was introduced
to Europe, where it got acclimatized and spread widely, including in human-modified
habitats. It survived in the epoch of climate change at the boundary of Holocene
and Neocene, and later evolved into new species. Transalpine or Southern European
pheasant is one of its descendants.
Body length of this bird without a tail is 40 cm; the tail feathers are long.
The main plumage is brown, darker on the back and upper sides. There are darker
thin stripes on the feathers, forming a cross-striped pattern that well masks
a bird in the forest. On the belly, the plumage has a yellowish tint. Bright
color spots in the male’s coloration are a green neck with a metallic sheen
and a red head with a double green tuft in the form of horns. The secondary
feathers are dark blue in color with an intense blue metallic shimmer. The male
taking off flaps its wings loudly and the reflections of its plumage attract
the attention of the inhabitants of the forest, becoming a universal alarm signal.
The plumage of the female is much duller – it is brown with transverse ripples.
The length of the tail feathers is up to 40-50 cm, they are ribbon-shaped with
a blunted tip, brown or reddish in color, and the background color becomes darker
towards the tip of the feather. There is a cross-striped pattern on the surface
of the vane. Legs of both males and females are pink in color; beak is thick
and strong, usually bluish in color; individuals with a yellow or greenish beak
are very rare. Males of this species have one of the brightest colors among
European birds.
This bird is almost always found in forests and keeps in dense undergrowth,
among tall bushes and ferns. Less often, the species occurs in sparse woodlands
with areas of high grass. Outside the nesting season, these birds live in flocks
of up to 20 individuals, and always keep a close distance from each other –
it is easier for them to notice the danger in time together. At night, the birds
fly up into the dense crowns of trees – usually conifers – perch in rows on
the branches, cling to each other and fall asleep.
The diet of Transalpine pheasant is diverse – leaves and grass, seeds, berries
and acorns, mushrooms, invertebrates and small vertebrates. Pheasants often
run under the feet of large herbivores, pecking insects they scare away. At
such moments, they are relatively safe from small predators. Their enemies are
snakes, birds of prey and small predatory mammals. Transalpine pheasants usually
run away from enemies, hiding in dense vegetation; in case of extreme danger,
they fly away, signaling danger with flapping of their wings and vocalization.
At the beginning of spring, these pheasants arrange bizarre courtship displays:
males go out into a well-lit clearing, where they walk on outstretched legs,
holding body erect, display bright plumage on their wings, flap them and coo,
inflating the goiter, while the females choose their males. These birds are
polygamous; male virtually does not take part in the care of offspring. Females
build nests on the ground among bushes, lay eggs, which are incubated for 3
weeks. There are up to 10-12 eggs in a clutch. The chicks are well developed,
at the age of one week they already are able to fly and fly up to the lower
branches of trees for the night. Females of this species often take care to
several broods at once together, and the deceased female can be replaced by
a foster mother, ensuring high survival rate of the offspring.
Sexual mturity occurs at the age of 2 years, life expectancy is up to 15 years.
This bird species was discovered by Mamont, the forum member.
Aotearoan
Green Pheasant (Notophasianus viridis)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: New Zealand, North and South Islands, found in woodland, open country,
scrubland, bush and wetlands.

Picture by Wovoka
The Common Pheasant (Phasianus colchicus) was introduced to
New Zealand, as with so many others, to facilitate game shooting, but it left
successful descendants there in the Neocene. There are a few rather typical
pheasant species in Neocene New Zealand, but the largest and most ornate is
the Aotearoan Green Pheasant.
This pheasant is quite large for a game bird, reaching a similar size to a female
turkey, but with a much longer tail, males may weigh up to 6.5 kilograms and
measure 90 centimetres long without the tail feathers. Females are an all-over
chequered tawny brown, and have relatively much shorter tails. Males have a
dark red wattle that forms a mask around the face, and the head, neck and front
of the chest are a dark iridescent bottle green, dark color continues onto the
back where the sheen is a pure blue. Sides are a reflective dark red with each
primary feather being striped with yellow so that it shows when wings are folded,
and the rump is bright orange. The tail feathers are longer than its ancestor,
more than a meter long, and barred with dark, reflective blue and bright orange.
As with all game birds, legs are long and strong, and the male’s ankles bear
a large spur.
These birds are gregarious and form loose flocks which range over the ground,
foraging by day, and sheltering by night. Diet consists of grain, stems, buds,
berries, insects, and occasionally small vertebrates. When alarmed, they prefer
to run on foot, but when pressed will fly upward suddenly and cruise off in
flight to a safe distance. Calls are generally similar to its ancestor, a chuckling
recurring cluck when alarmed.
These birds are lek-breeders, males compete viciously to form and defend harems
of females, and breeding season runs from September to November. Males will
compete by leaping up at each other and kicking with their spurs. In courting
the female, the male will make overtures by circling her and fanning his tail
in her direction, spreading his wings. The female will make a nest that is a
shallow scrape in the ground, lined with leaves, in lee of a bush or small tree.
Ten to 12 eggs are laid over a 2 and a half week period, incubation is up to
29 days. Chicks are well developed and able to follow their mother, they start
to fly after 20 days, and resemble their parents after a month. Lifespan may
be up to 12 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Golden
Fenghuang (Fenghuang aurata)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Eastern Asia, forests.
In the Holocene, some pheasant’s species are among the most beautiful birds,
with males having vivid colors. In the Neocene the pheasants continued with
their beautiful colors, an example is the golden fenghuang (fenghuang in mythology
is the name of the Chinese phoenix), a descendant of the golden pheasant.
Living in the forests of Far East, the male golden fenghuang have a length of
195 cm from the beak to their long tail and the female have 87 cm. The males
have a golden erectile crest, which is usually left flat, giving a hammerhead
shape to head. During the courtship, the male open their crests like a fan and
start to display to the female, standing in front of them with their crest raised
and wings outstretched, vocalizing. When raised, the crest displays a remarkable
combination of scarlet and yellow. They form leks of close related males. At
a lek, the males maintain small territories next to each other.
Besides the crest, the males have more colorful plumage in their bodies. Their
face, throat, chin, and the sides of neck are orange. The wattles and orbital
skin are both yellow in color. The upper back is green and the rest of the back
and rump is golden-yellow. The tertiaries are metallic blue whereas the scapulars
are metallic red. Their long central tail feathers are reddish golden with black
spots. The upper tail coverts are the same color as the central tail feathers.
The male also has a scarlet breast, and scarlet and light yellow flanks and
underparts. The females have a duller mottled brown plumage all over and their
tail is short (50 cm) if compared to the males (100 cm). In both sexes the beaks,
legs and feet are a dull yellow. They eat berries, grubs, seeds and invertebrates.
The golden fenghuang is polygamous, the male will mate with several females.
The nest is a shallow scrape in the ground lined with leaves, sticks and other
debris. The female lay 7 to 12 eggs and are incubated only by her for 24-28
days. The chicks are nidifugous and follow the mother around after hatching,
staying with the female for two months. The young males are similar to the female
until they reach their sexual maturity with six months, at this time they acquire
the plumage of the male. Their lifespan is of 7 years.
Another species, the Silver
fenghuang (Fenghuang argentata) lives in the montane forests near the
Himalayas. Their main features of the males are the metallic white crest and
white central tail feathers with black spots. The females are similar to the
previous species. The males have 120 cm and the females 79 cm.
This bird species was discovered by João Vitor Coutinho, Brazil.
Malac
tavus, peacock partridge (Pavotoris malac-tavus)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: The Balkans, the eastern and northeastern edge of the Mediterranean
lowland, Asia Minor and the southern coast of Fourseas.
Rock partridges, while remaining common birds for their region, suffered minimal
damage in the human era. Their survival was greatly facilitated by life in mountainous
areas, which remained a kind of “refuge” for representatives of fauna due to
the difficulties of economic activity. Then, climate changes in the Neocene
allowed some of their descendants to acquire an appearance significantly different
from the usual one for their ancestors. Malac tavus is one of these “deviant”
species, a descendant of the chukar partridge (Alectoris chukar).
This species has become much larger than its ancestor: the average body length
is about 50 centimeters, the wingspan is up to 1 meter. The appearance of the
bird somewhat resembles the short-tailed pheasants of the human era. Body is
muscular, rounded, with a wide chest: pectoral muscles are well developed and
allow the bird to take off quickly. Beak and legs are dark brown. Plumage of
birds is multicolored, but the color is generally unshowy. The upper part of
the body is rippled – brown with thin cross stripes on the feathers; sides and
tail are ochre-red. There are dark vertical stripes on the sides, characteristic
for the ancestral species. There are no spurs on the legs.
A crest grows on the male’s head, representing a bunch of erect black feathers;
it also has naked bright red eyelids, which swell strongly during the courtship
display. Both male and female have white throat, bordered by a narrow black
stripe. Tail feathers of the representatives of this species are greatly elongated:
25 cm long in males and 13 cm in females. Males have bright oval spots on the
tips of tail feathers, like peacocks, but much more modest in color: a black
spot with a bluish-green metallic luster and a white spot in the middle. In
females, dark brown spots without gloss sometimes appear on the uppertail coverts.
Also, the female lacks crest – there is only a black spot on the crown – and
the ochre-red color in the plumage is replaced by grayish-brown.
Peacock partridge feeds mainly on vegetative food – herbs, berries and leaves
– but does not disdain insects; it is especially typical for chicks during the
period of active growth. The bird’s favorite food is acorns of low-growing
oak species growing on the outskirts of Mediterranean lowland. During seasonal
droughts, malac tavus birds live in groups of 15-20 one, and in autumn they
split into pairs or threes. Malac tavus flies rarely and reluctantly, preferring
to cling to the ground and freeze when a predator appears, or to escape and
hide in the bushes. Taking off, the bird loudly flaps its wings; it behaves
this way only if the predator is getting close. Such an unexpected take-off
often saves the bird’s life, distracting the predator for a few seconds.
The mating season begins in February, along with the winter rains. Males begin
attracting females by actively displaying. They gather in open areas in groups
of 10-20 birds, and begin courtship displays. Birds utter throaty bubbling sounds,
strongly inflating the neck and ruffling the plumage on it, and spread the tail
coverts, leaning forward strongly. Demonstrating himself to the female, male
turns his head to her and begins to shake his whole body; at the same time,
the ocular spots on the tail coverts shine and shimmer in the sun rays. Females
walk among males, choosing a suitable mate, and actively compete with each other,
driving rivals away from the male they like. A successful male has 5-7 females
in the “harem”.
Males do not take part in the rearing of offspring. There are five to eight
eggs in the clutch, which the female lays in a shallow hole on the ground, usually
among bushes or under the cover of a large stone. The eggs are pale yellow with
brown speckles. The incubation period lasts about 25 days. The chicks hatch
with eyes opened and covered with down; they leave the nest forever 2 hours
after hatching. At the age of 3 months, they can digest the tough grasses and
seeds that make up the diet of adult birds. In autumn, young birds are already
completely independent, reaching sexual maturity at the age of 18 months. Life
expectancy is about 10 years.
This bird species was discovered by Nick, the forum member.
Motley
aguna (Aguna colorata)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: northern Europe from Scandinavia to the Urals, mountain forests, taiga.
During the glaciation period at the boundary of Holocene and Neocene, many species
of temperate latitudes faced a choice: move to the south, become extinct or
adapt to new conditions. In such conditions, one of the most common species
of Eurasian galliform birds – grey partridge (Perdix perdix) – appeared. In
due course of evolution, this species gave rise to a separate genus of birds
that spread widely across Eurasia in temperate climates and in the highlands.
One of the most common species of this genus is motley aguna (aguna is the name
for the partridge among the Uighurs). These birds are much larger than their
ancestor, having the size of a large rooster and weighing up to 4-5 kg. This
makes it easier for them to tolerate cold, and in a humid climate, precipitation
will increase, and birds will be able to find a thick enough layer of snow for
most of the winter.
Motley aguna looks like a large partridge: it has a compact body of dense build,
short tail and wide rounded wings. Head is relatively small, on a movable neck.
Beak is of moderate length, thick, adapted for digging up frozen ground and
biting the buds of shrubs.
All species of this genus are distinguished by their specialization to cold
climates. They have strong legs with a feather border on their toes, due to
which these birds will be able to walk on the snow without falling through,
and rake it more successfully – a parallel evolution with grouse birds is seen
here. Aguna has no spurs.
In addition, these birds are characterized by dense winter plumage and the absence
of bare skin areas on the head and chest, which are often found in tropical
pheasant birds – this is an adaptation to life in a cold climate. During the
courtship season and when defending the territory, aguna male utters loud cries
with the help of a special resonator, which is formed by the easily stretchable
anterior wall of the trachea. During the courtship display, a bubble the size
of an orange swells under the male’s head.
The color of the plumage of the motley aguna combines sharply delimited spots
of different colors. The bird’s head is ochre-red with black “eyebrows” and
a spot under the beak. Back, tail and wing coverts are ripply, gray-brown with
a thin white pattern. Primary and secondary feathers are fawn with black speckles
near the base. Belly and tail are white. In winter, the color of the plumage
does not change.
Motley aguna lives in flocks of one male and 6-8 females, which make up its
harem and nest on its territory not far from each other. The existence in a
harem helps the agunas survive harsh winters – a group of birds simply gathers
in a hole dug in the snow and the birds snuggle together, keeping warm. This
behavior increases the likelihood of survival in a cold winter. The consequence
of this type of family arrangement is expressed sexual dimorphism: the male
is larger and weighs about 1 kg more than the female. He drives away strangers
from the territory of the flock, and during the courtship season he tries to
recapture from the enemy a part of his harem. In spring, the male grows two
bands of elongated white feathers on the sides of the neck and chest. This is
a feature for courtship display in conditions of a short breeding season. The
displaying male inflates the resonator bubble and utters a loud “grunt”, fluffing
the feathers on the sides of the chest and neck into a kind of shield. At this
moment, it seems much larger, which allows it to establish a dominance relationship
without a fight. If aguna males arrange a duel, the fight can be very persistent;
opponents seize each other’s plumage with their beaks and strike with their
legs. It happens that fighting birds roll on the ground, almost not paying attention
to the approach of a predator.
The diet of aguna includes mainly vegetative food – seeds, berries, young shoots
of plants. Birds also eat insects and worms, peck small frogs and chicks of
birds nesting on the ground.
Nesting begins in spring, when leaves grow on the trees. Females of the same
harem arrange nests at a distance of several tens of meters from each other.
Near the nest, they behave very cautiously and quietly. The male, on the contrary,
behaves deliberately noticeably and, if necessary, distracts predators from
the nest. Clutch numbers up to 9 eggs; incubation lasts 25 days. Females from
the same harem jointly raise offspring. Young birds become capable of breeding
the next year.
Motley aguna and related species in the north of the genus range compete with
a larger species of galllinaceous birds, aganak
(Gravilagopus aganak). Unlike aganak, agunas prefer more wooded terrain
and mountainous areas, so competition between them is minimal.
On the territory of Eurasia and North America, various aguna species successively
replace each other. Beyond the Urals, motley aguna is replaced by a close species
– barbed aguna
(Aguna barbata). It differs from the European species in the features
of the courtship dress: the male grows long gray feathers, similar to a beard,
on the front of the neck and upper chest. The voice of the displaying male is
a rolling monotonous trill. The plumage of this species has a camouflage grayish-brown
color with a thin cross-striped pattern on the back, chest and wings. There
is a black “mask” on the head of this bird, and the sides under the wings and
the underside of the wings are white with separate black feathers. When the
bird takes off, these color spots become clearly visible and serve as an alarm
signal.
The territory of Kamchatka, Beringia and Alaska is inhabited by Beringian
aguna (Aguna beringica). In this species, “beard” is very short and
occurs in both males and females. Male in courtship dress grows elongated “epaulettes”
of thin fluffy feathers on the shoulders. The main color of birds of this species
is dark brown with a white “scaly” pattern on the underside of the body. “Beard”
is also brown. Voice of this species represents separate sonorous calls and
series of monotonous clicks. At the border of the ranges of barbed and Beringian
agunas, hybrid and intermediate populations are found, differing in intermediate
shades of body coloration and a gradually decreasing size of the “beard”. “Epaulettes”represent
a recessive feature and are characteristic only for pure-blooded Beringian aguna.
For the northern borders of the forest zone of North America Sioux
aguna (Aguna sioux) is characteristic – a large species (the weight
of an adult bird is up to 5.5 kg), remarkable by a very dark color. The upper
part of the body of this bird is dark brown, almost black; the underside of
the body is ochre-red. The lower part of head, throat and chest are black; white
spots on feather tips form a pattern on the chest. Sometimes a light color morph
appears, in which black color is replaced by smoky gray, and red by fawn; in
eastern populations it occurs more often – up to 10% of the total number of
population. In the male of this species, during the mating season, very developed
“epaulettes” of long white fluffy feathers grow on the shoulders. During the
courtship display, they open on the sides of the bird, forming two fans. The
male shakes them during the dance and utters loud whistling sounds.
Greenland is home to polar
aguna (Aguna borealis) – the northernmost species of the genus. In
color, it is similar to the light color morph of Sioux aguna: back is bluish-gray,
black is only at the tips of the secondary feathers. The belly of this bird
is grayish-white. Unlike other species, it lacks the feathery “ornaments” characteristic
for males’ courtship dress; courtship displays include a dance with wings opened,
accompanied by ringing calls. This bird lives in a harsh polar climate, so it
makes annual migrations along the coast of Greenland: in summer it is found
at the northernmost edge of the island, and by winter it migrates south to the
seashore. Also, this species is omnivorous and can even feed on seaweed and
dead fish washed ashore.
Several species of smaller mountain agunas inhabit the mountain ranges of Asia.
Singing aguna (Aguna
cantans) inhabits the Altai mountain meadows and differs from the northern
species in smaller size: adult bird weighs about 3 kg. Plumage of this species
is brightly colored: throat and belly are straw-yellow, undertail is white,
and tail feathers are white with red tips. Back and wing coverts of this bird
are gray with fine brown ripples, and the primary and secondary feathers are
black with a bright purple and green metallic luster. During the courtship season,
male of singing aguna grows “sideburns” of short black feathers. He “dances”
in front of the female, keeping his body vertically and stretching his wings
and tail. At the same time, the male inflates the throat resonator and utters
a bubbling melodic whistle.
On the high-altitude plateaus and in the valleys of the vast Himalayan range,
there are several close species of aguna with multicolored coloration and dark-colored
wings, vaguely similar in color to singing aguna. On the southern slopes of
the Himalayas there lives a small pheasant-like
aguna (Aguna phasianoides) characterized by a greenish-yellow color
with black ripples, turning to “scaly” pattern on the wings. Primary feathers
of this species are black with a noticeable blue sheen in males. In females,
black color is replaced with dark brown and lacks metallic luster. Male’s throat
at this species is black, and in courtship season, thin white feathers grow
on the sides of the black spot, fluttering in the wind during the flight of
the bird.
The highlands of the Himalayas are inhabited by silverish
aguna (Aguna argentea), in which almost all the plumage, except the
wings, has a gray color with a distinct metallic luster. At both males and females,
elongated plumage on the head forms “eyebrows” for protection from the bright
sun. Due to the limited food resources in the mountains, this species often
keeps in pairs or families of two females and one male.
On the northern slopes of the Himalayas, black
aguna (Aguna tenebrosa) lives, whose plumage is almost entirely black,
except for the feathers on the lower back and tail, which are snow-white in
color and serve as a signal for relatives. They are clearly visible only during
the bird’s take-off. By the courtship season, male of this species grows a “mane”
of thin white feathers with a fluffed vane and a flexible soft rachis. During
courtship, male fluffs these feathers and shakes them, trembling his whole body
and “beating the drum” with his feet.
Limited hybridization takes place between Himalayan aguna species; birds with
transitional color and plumage pattern, forming stable populations, are often
found in mountain valleys.
Aganak
(Gravilagopus aganak)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Eastern Siberia, Putorana plateau, tundra steppe in the Far North.
Grouse and allies appeared on the Earth during the Pleistocene glaciation. In
the Holocene, some species and subspecies of these birds became extinct or became
very rare due to human persecution. In the epoch of the biological crisis at
the boundary of Holocene and Neocene, the central regions of Eurasia with a
harsh climate, less susceptible to human activity, became a real “reserve” for
these birds. And in early Neocene, many representatives of gallinaceous birds
adapted to rather harsh living conditions appeared on the territory of temperate
and cold climate regions. The descendants of the willow ptarmigan (Lagopus lagopus)
have achieved great success in the struggle for existence. In the harsh conditions
of the continental climate on the territory of Eurasia, one of these birds lives
– aganak (Tuvan name of the willow ptarmigan). This species inhabits open areas
where low-growing trees and shrubs do not form a continuous cover. Aganaks willingly
feed on the “obda roads”, where
grasses and low shrubs grow.
Aganak is a fairly large bird compared to its ancestor: an adult individual
weighs up to 6-7 kg, and up to 8 kg in large males. At such a size, the bird
has not lost the ability to fly, although it does it with difficulty and only
in case of extreme danger. The bird’s wings are short and wide, with rounded
tips. The flight of the aganak is fast and straightforward, accompanied by a
loud wing flapping, which serves as an alarm signal for relatives. Aganak is
able to fly about 200-300 meters, after which it lands on the ground. If the
predator has not stopped the pursuit, the aganaks escape by run. On toes of
these birds “brushes” of feathers grow, bordering the toes. In summer, they
almost completely disappear, but in winter they expand greatly. Due to this
feature, aganak does not fall into the snow and can dig it out more efficiently
in search of food. Aganak has longer and stronger legs than its ancestor – it
allows the bird to run quickly, fleeing from an enemy, and walk long distances
in search of food. Legs are feathered up to the toes.
The body of the aganak has rounded outlines and is covered with dense plumage.
In winter plumage of the bird, the down part of the feathers is thicker and
longer than in the summer one. Bird’s tail is short and fan-shaped. Male has
a longer tail than female and uses it during courtship displays. Aganak retained
the ability of its ancestor, the willow ptarmigan, to change the color of its
plumage depending on the season. The winter plumage of the aganak is snow-white,
thick and very warm. Summer plumage is similar to the plumage of many grouse
birds: the main background is brown; there are large black ripples on the wings,
a black border on the tail, and chest is rusty-red with thin black ripples.
Rings of red feathers around the eyes of males are bright spots on this dress.
Females have only narrow red “eyebrows” above their eyes.
Aganak is omnivorous, although in winter it is forced to completely switch to
vegetative food. The beak of this bird is thick, conical, slightly curved and
compressed from the sides. With the help of such a beak, aganak easily bites
off the leaves and branches of shrubs and dwarf trees. Small pebbles (gastroliths)
are constantly present in the muscular stomach of this bird. In order to replenish
their stock in winter, aganaks migrate to places where there are wind-blown
rocks and where they can replenish the stock of gastroliths. If possible, aganak
catches small rodents and pecks eggs and chicks of small birds nesting on the
ground.
In the spring, when the snow finally melts, aganaks begin courtship displays.
At this time, the male displays himself to the females: he walks in front of
them, turning sideways and opening his wings to the ground. During the courtship
displays, he inflates his goiter and grumbles hoarsely. Eyebrows and areas of
skin around the eyes are filled with blood, which makes them seem larger. Fights
often occur between aganak males on the lekking ground, during which the birds
beat each other with their legs, jump on the opponent and try to peck him or
grab the feathers on his head.
It is a monogamous bird. The pair is preserved for the entire breeding season
and raises offspring together. At this time, the male is very aggressive and
often attacks small predators trying to kill his chicks. In the brood, aganak
has up to 4-6 chicks, which grow quickly and gain about 5 kg by autumn. Usually
about a third of the brood survives until winter.
The life expectancy of aganak is about 15 years.
Eastern
steppe hazel-hen (Pavonasia asiatica)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: Far East, forest-steppe.
The end of Holocene and the beginning of Neocene were marked by another ice
age. The giant glacier wall pushed the northern species to the south, making
the north of the Earth a very uncomfortable place for life. After the retreat
and melting of the glacier, the descendants of the migrated species returned
to the north, but some descendants of the northerners continued to live further
south, producing new species that adapted to changing conditions. In the swampy
forests of Siberia and the Far East, the fantail
hazel grouse (Pavonasia paludiphila) lives – a rather large bird that looks
like a capercaillie, and a little to the south, where the taiga is replaced
by dry woodlands with separate areas of the steppe, a related species lives
– eastern steppe hazel-hen.
This bird has typical features of a resident of the steppes, and resembles outwardly
not so much its forest relatives as bustards of Holocene epoch. It has a relatively
light build, weighs about 1.5-1.8 kg, and has longer legs and neck. Male has
a feather tuft growing on the head, which he spreads in front of the females
during the courtship display. Sexual dimorphism is clearly expressed. The plumage
of the male is darker than that of the ancestor, with a less pronounced pattern:
the striped plumage is present, but poorly expressed on black-brown feathers.
Red feathers, forming “eyebrows” over the eyes, are characteristic. In a displaying
male, feathers of the “eyebrows” fluff, and the skin under them is filled with
blood, increasing their visible size. Tail feathers have white tips. The color
of the female’s plumage is rippled, yellowish-gray with numerous cross stripes,
masking the bird on the nest.
When the eastern steppe hazel-hen is resting, it usually hides its head and
neck “under the wing”, bends its legs and resembles a boulder stone among steppe
grasses. If disturbed, it takes off with a loud wings flapping, scaring away
and stunning the enemy with a lighter, almost white plumage of the inner side
of the wings and belly. After flying several tens of meters, bird lands on the
ground and runs away. Chicks have a camouflage color with dark stripes and ripples
on the light down. Usually, eastern steppe hazel-hen does not like to fly, spending
almost all the time on the ground. Only in the forest-steppe and sparsely wooded
areas, bird can fly up trees, but usually it runs away from the enemy quite
quickly and hides in the bushes or among the grass.
The repertoire of sound signals of these birds is quite diverse – loud prolonged
alarm signals, grumbling and squeaking for communication of adult birds with
chicks, whistling for signals of adult individuals to each other.
Eastern steppe hazel-hen is a relatively omnivorous bird – it eats both vegetative
food, especially seeds of various steppe grasses and cereals (more often in
autumn and winter), and animals – mainly various invertebrates, occasionally
small vertebrates. The bird falls prey of various predators – predatory birds
and mammals catch adult birds, and large reptiles can ravage the clutch or attack
the chicks. Younger and older birds sometimes die during winter starvation.
The courtship season begins early, in the first half-mid-spring. Males find
a suitable place for lekking ground – usually it is an elevation like a large
stone, a hill or a low tree – and begis a courtship display, resounding the
surroundings with a loud piercing whistle. Eastern steppe hazel-hen is mainly
a solitary bird, it does not form large lekking grounds; usually 3-5 males gather
at the lek, and each of them displays independently. Males of this species are
monogamous, do not take part in incubation of the clutch, but take care of the
chicks and protect the brood. Male can drive away even herbivorous beasts from
the nest.
There are 8-10 eggs in a clutch; the female incubates them for 18 days. The
nest itself looks rather unremarkable – it is a simple hole in the ground in
a secluded place (usually under a bush or among tall grass), lined with grass
and feathers of the bird itself. The parents immediately take the chicks out
of the nest, and do not return to it anymore. Chicks are able to run already
at the age of one day, and after a week they can already fly short distances.
They can fly in full degree at about a month of age. By autumn, they have already
left their parents, but usually keep in a flock in the first winter before the
coming of sexual maturity – it happens already the next year.
The maximum lifespan of eastern steppe hazel-hen is up to 16 years, but most
of these birds die much earlier.
This bird species was discovered by Bhut, the forum member.
Migratory
grouse (Neobonasa migratoria)
Order: Gallinaceous birds (Galliformes)
Family: Pheasants (Phasianidae)
Habitat: forests of temperate zone in North America.
The Ice Age during the end of the Holocene led many species to adapt to survive.
Some developed migratory habits, moving to the South so they could escape the
cold weather. The ruffed grouse (Bonasa umbellus) was one of these species,
with its descendant in the Neocene, the migratory grouse, becoming a fully migratory
bird. During the summer, this bird lives in forests from the Appalachian Mountains
across Canada to Beringia. During the winter, these birds migrate to the Mexican
Plateau.
The migratory grouse are relatively slender, medium-sized birds that measure
from 40 to 50 cm in length and span 80–100 cm across their strong wings. The
head, neck, and back are grey-brown; the breast is light with barring. There
is much white on the underside and flanks. Overall, the birds have a variegated
appearance; the throat is often distinctly lighter. The tail is essentially
the same brownish grey, with regular barring and a broad black band near the
end. The ruffs are on the sides of the neck in both sexes. They also have a
crest on top of their head, which sometimes lies flat. Both sexes are similarly
marked and sized, making them difficult to tell apart, even in hand. The female
often has a broken subterminal tail band. At the same time, males tend to have
unbroken tail bands, though the opposite of either can occur. Females may also
do a display similar to the male. Another fairly accurate sign is that during
the displays, the males will inflate their throat, revealing a bright red patch
of skin.
Like most grouse, they spend most of their time on the ground; mixed woodland
rich in aspen seems to be particularly well-liked. These birds forage on the
ground or in trees. They are omnivores, eating buds, leaves, berries, seeds,
and insects.
These birds are polygynous, and males may mate with several females during the
breeding season. The migratory grouse relies mostly entirely on a non-vocal
acoustic display, known as drumming, unlike other grouse species. The drumming
itself is a rapid, wing-beating display that creates a low-frequency sound,
starting slow and speeding up. The male will also inflate their throats during
the ritual. Hens build nests and lay and incubate their eggs under the cover
of bushes. The hen uses grass and forbs between patches of bushes for additional
cover. During incubation, female migratory grouse undertake recesses, where
they leave the nest to undertake self-maintenance activities, thought these
recess activities are typically within 250 m of the nest. They lay four to six
eggs that are incubated for 21 days. Chicks can walk as soon as they are hatched
and are able to fly short distances within two weeks. Within five weeks they
are able to fly longer distances.
They spend most of their time quietly on the ground, and when surprised, may
explode into flight, beating its wings very loudly. During the autumn, the migratory
grouse will fly to the South. Their migration will happen both by flight and
walking. These birds use long patches of grass in the prairies regions that
they cross to hide their terrestrial movements from predators. The average lifespan
of a migratory grouse is seven years, although some birds can live for eleven
years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Volcanic
lake grebe (Nanopodiceps volcanophila)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: Japan, high-mountainous volcanic lake at Hokkaido Island.
Picture by Fanboyphilosopher
In human epoch birds of grebes order were widely settled on
Earth and lived at all continents, except for Antarctica. These birds live in
fresh-water reservoirs, perfectly swim and dive, and hunt small fish. In human
epoch among these birds there was a plenty of local and endemic species, and
some of them could inhabit only one small lake. Such species appeared the most
vulnerable ones at destruction of the natural environment as a result of economic
activity of people, and in human epoch some similar species of grebes had irrevocably
died out. But most common and widespread species had escaped, and after human
disappearance they continued evolution.
Japan Islands became a favorable place for evolution of this group of birds.
In Neocene at the archipelago some kinds of local flightless grebes had evolved
at once. The landscape of islands quite favours to this event: it is heterogenous
enough, differing in abundance of isolated lakes, and in conditions of warm
climate of Neocene and volcanic heating of some lakes their biological efficiency
is high enough to support a settled population of gluttonous and fastidious
in food birds.
All Japanese lake grebes belong to one genus Nanopodiceps, and are descendants
of one species widely settled on islands. The ancestral species is able to fly,
but its local descendants had completely left skills of flight. They are birds
with narrow head, long beak, wide paws and almost completely absent wings. At
all lake grebes of Japan Islands the body is streamlined, and tail is almost
absent. Swimming under water, these birds extend head forward, and paddle by
paws, working by them alternately, or making simultaneous pushes. Wings of flightless
species are advanced in various degrees – from small, but appreciable up to
completely reduced ones. The reduction of wings at these birds occured independently
from each other.
Japanese lake grebes have bright colored “collars” similar to plumage of great
crested grebe’s head, and some species have “horns” of lengthened feathers.
It is connected to necessity of identification of relatives – the ancestral
species frequently appears on lakes populated with its descendants, and strongly
expressed distinctive attributes help to exclude formation of hybrids.
Food of lake grebes includes small fishes, crabs and shrimps. Birds are able
to dive and easily chase prey under water. They eat exclusively live prey, swallowing
it entirely.
All species nest among coastal vegetation, arranging primitive flat nest. Frequently
nest happens half submerged in water, but even in this case the incubation of
eggs passes successfully. Usually in clutch may be 3 – 4 eggs – at small species
clutch is lesser, at large it is more (up to 6 eggs).
Volcanic lake grebe lives at the north of area of this genus. Its habitat is
unusual: it inhabits an only mountain valley, in which the lake about ten square
kilometers by the area is stretched. At the bottom of this lake volcanic thermal
springs spurt, and temperature of water even in winter is not less +20°С. Volcanic
lake grebes nest in thickets of reed bordering this lake. Due to original microclimate
water and marsh plants do not stop to grow even in winter, providing birds with
shelter, and their prey with food.
Volcanic lake grebe is tiny bird; its weight does not exceed 200 grammes. Plumage
on the body of this bird is monotonously grey, darker on neck. Colouring of
head of volcanic lake grebe is very bright and appreciable from apart. Feathers
on nape and back part of male’s neck form bright “flag” – they are strongly
extended and directed back. Females lack of such “flag”, but colouring of plumage
of the appropriate parts of their heads is the same, as at males. Forward part
of head and top of head at birds of both genders is black and lateral parts
of head and “flag” are bright orange with reddish shade. At some males tips
of feathers of “flag” are white. The iris of eyes at male has impressive white
color; at female it is brown.
Wings at this bird are very small – their length does not exceed 3 – 4 cm.
At Japan Islands other species of lake grebes live:
Common
lake grebe (Nanopodiceps volans) is the only species of the genus had
not lost ability to fly. Obviously, this species or its direct ancestor became
the ancestor of all flightless endemic grebes of Japan Islands. This bird has
bright blue paws (it is the unique colouring which is not meeting at other species
of genus), brown plumage on the body and white “saddle” – site of plumage on
back and under wings, well appreciable during flight of bird. From a nape on
back side of neck small “crest” of white feathers with black tips grows. In
courtship season males of this species show it to females, trembling by head
and neck.
Common lake grebe is settled at all Japan Islands and lives mainly on mountain
rivers, avoiding a competition to settled flightless lake species. Flightless
species of lake grebes have similar biology and differ from each other in colouring
and features of courtship ritual.
Southern
lake grebe (Nanopodiceps australis) inhabits the south of Hokkaido
Island. It has light colouring – pale grey plumage with brownish shade on back
and snow-white stomach. The cop at male is black with dark blue metal shine,
and around of eyes there is a ring of black feathers. The female of this species
has only black spot on nape, and it shows this mark to the male as a symbol
of submission during the courtship ritual. This bird weighs no more than 300
grammes. Wings at this bird are very small; only few reduced primarily feathers
are appreciably from outside.
Semi-striped
lake grebe (Nanopodiceps striatocollis) inhabits lake at the south
of Honshu Island. This species has darkest colouring of plumage – black with
separate white sites. From nape along back part of neck up to shoulders on black
background narrow longitudinal strips of white color stretch. During courtship
display male fluffs feathers on nape and neck, and strips become wider and more
appreciable. He displays to the female plumage on neck, swimming around her
and having turned a head by nape to her. Wings at this species are reduced stronger,
than at other species of genus: they are completely invisible from outside,
and only on skeleton the rudiment of humeral bone is visible.
Yellow-headed
lake grebe (Nanopodiceps flavicephalus) lives in several mountain lakes
and rivers of Shikoku Island (Shikoku-Sanchi mountains). It has grey color of
body plumage, and head and top part of male’s neck are bright yellow. At the
female yellow color is replaced by yellowish-brown one. On nape and back part
of neck of this bird the strip of black feathers passing to black plumage of
back stretches. It is the largest species of genus – weight of adult bird reaches
700 – 800 grammes.
This bird nests in reeds, preferring the thickets growing at the distance from
coast, when it is impossible to reach to it from the coast. Frequently these
birds arrange nests on islets of floating vegetation, and nesting pair specially
rakes large heap of plants, pulling them from ground, and strengthens the construction
with rods.
The idea about existence of this group of bird species was proposed by Simon, the forum member.
Chatham
sea grebe (Parapodiceps chathamensis)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: Chatham islands.

Picture by Alexander Smyslov
Due to ability to flight birds had occupied almost all ocean
islands, from cold Arctic regions up to equally cold Antarctic Region. During
the ice age Chatham Archipelago in Pacific Ocean represented the ecosystem very
poor in species variety and economically using poor resources. But in Neocene
the situation at the islands has changed: numerous birds and insects had appeared,
descendants of rats and mice introduced by people also evolved, and islands
have become overgrown with bushes and trees. The largest terrestrial species
of local fauna is Chatham barocygnus
– flightless descendant of black swan.
At the islands numerous freshwater reservoirs appeared – small lakes and bogs.
In them mosquitoes, dragonflies and other insects breed plentifully. Larvae
of insects represent one of main food sources for other inhabitant of islands
– for Chatham sea grebe. This bird, the descendant of great crested grebe (Podiceps
cristatus), is original analogue of loons (order Gaviiformes) of Northern hemisphere
in southern temperate latitudes.
Chatham sea grebe is rather large representative of the order, well flying bird
weighting about 400 grammes. It has lengthened body of streamline outlines;
legs are shifted to back part of body, therefore bird is not able to walk on
land, but only creeps on stomach, pushing by legs. Flexible mobile neck and
pointed awl-like beak give out the skilful hunter for water animals in this
bird. Chatham sea grebe can dive for a long time and swims under water up to
50 meters in searches of prey.
This species of birds has very characteristic colouring: back, wings, top part
of neck and head are grayish-blue; stomach and the bottom part of neck are white.
On cheeks of bird there are large white spots. Eyes are yellow and ringed with
a thin strip of black feathers. Feather “collar” characteristic for its ancestor
has almost completely disappeared: only “whiskers” of grayish feathers, more
advanced at male, remained from it. Feather “horns” also have decreased in size
and became very short. In courtship season displaying male raises them upwards,
swimming around the female. The beak of Chatham sea grebe is colored bright
red – birds display it to the partner during the courtship.
This species of birds has found original enough decision of a problem of limitation
of resources in freshwater reservoirs of small islands of archipelago. Freshwater
reservoirs are used by these birds only for nesting and rearing of posterity,
and adult birds search for food in sea. Chatham sea grebe eats various invertebrates
and fish fry. In fresh waters of Chatham archipelago there are no fishes, except
for the separate species coming from the sea, therefore the young growth of
this species finds plentiful food like mosquitos and other insect larvae. Adult
birds, replacing each other on the nest, fly to the sea for hunting. In coastal
zone, among thickets of brown macroalgae, adult birds hunt shrimps, small crabs
and octopuses. Besides high sea waves do not enable birds to build floating
nests. The seasonal climate of islands allows birds to nest only once per one
year.
The main problem at life in the sea is the necessity of removing of superfluous
amount of salt from an organism. At Chatham sea grebethe function of salt removing
the modified part of lachrymal gland has undertaken, separated from actually
lachrymal gland and opening as a separate channel in eye-socket. From time to
time the bird “cries”, splashing brine out as small jets, and at once washes
out eyes, dipping head in water. These birds hide from storm among thalli of
brown algae which soften impacts of waves in great degree, or in the coastal
caves protected from surf by stones.
Chatham sea grebe nests in freshwater lakes and bogs in numerous congestions.
As adult birds have almost unlimited source of food, this species forms significant
congestions – up to several tens couples of birds in the same small lake. At
this species the nest is typical for grebes – it is a floating heap of plants.
Sometimes some nests hook together, and birds continue eggs hatching, not paying
attention to new neighbours.
In clutch of this species there are up to three eggs with motley shell. Chicks
are well advanced and independent. Having dried under mother, they at once start
to search for food independently, diving to the depth up to 50 – 60 cm. Chicks
have brown down with narrow longitudinal straw-coloured strips and rather short
beak. Salt-emitting gland at them is yet underdeveloped, and therefore they
are not able to live in sea water. Parents warm and preserve posterity within
the first month of life. Only at the age of three weeks at young birds salt-removing
gland starts to develop. At this time they undertake very dangerous travel overland
to the sea coast. Chatham sea grebes especially willingly nest in lakes from
which the small river flows to the sea even if it forms a falls. These birds
almost do not settle in lakes far from the sea, or chicks should overcome some
hundreds meters of land in total, passing from lake to lake before they will
succeed to reach the sea coast. Chicks can move on land, keeping body in almost
vertical position. They hardly keep balance, helping to themselves with wings.
During this transition parents fly from lake to lake, calling up chicks with
loud cries. Not seeing parents, chicks choose a correct direction of movement,
being guided on their voices.
Having reached up to the sea, young birds study larger prey hunting, and parents
complete their rearing within one month. Young birds begin independent life,
not being able to fly at all. They start nesting at one-year-old age.
Antarctic
grebe (Cryopodiceps australissimus)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: freshwater reservoirs of Antarctica.

Picture by Simon
In Neocene Antarctica pushed aside by South America, had moved
aside Australia. Shift was not far, but it has permitted to weaken considerably
the cold Antarctic Circumpolar current, which did not let warm waters from equator
to the continent. Climate in Antarctica of Neocene epoch is still seasonal with
severe winter, but some birds had adapted
to all-the-year-round life in this severe place. Antarctic summer in Neocene
is favorable enough for life of birds and almost right after snow thawing bird
flocks from South America, Australia and New Zealand direct to this continent.
Among summer visitors of Antarctica there is one species of grebes, which is
the descendant of Tasmanian population of great crested grebe (Podiceps cristata),
which has rather easily gone through human epoch due to vast area, having kept
in “islets” of the earlier solid area. Possible, casual moving of these birds
from Tasmania through subantarctic islands has permitted
them to reach Antarctica and to join other summer visitors of this continent.
Antarctic grebe lives in rivers and lakes of Antarctica. The size of this bird
is medium for grebes: it weighs about 200 grammes. Larger birds hardly could
maintain successfully rather slowly renewing food resources of freshwater reservoirs
of cold Antarctica. For Antarctic grebe the streamline body shape is characteristic;
legs are shifted far back, and this bird is not able to walk on land. The body
feathering is colored black. It does not prevent this bird to hunt, because
its basic prey is made of larvae of insects and crustaceans. But in conditions
of cool Antarctic summer such colouring helps to be warmed quickly during the
rest. Favourite resting places of Antarctic grebes are flat stones, which one
edge is inclined to water. The bird creeps on such stone, pushing by legs, and
has a rest on it for a long time, having fluffed up feathers and basked in sun.
Bright “collar” of cross-striped black-and-white feathers with expressed metal
shine contrasts with dim color of plumage. In courtship season this “collar”
is stretched as a semicircle – feathers on nape rise upwards, and “moustaches”
are turned downwards. Male involves the female, shivering these feathers which
brightly shine in sunlight. If the female is ready to accept his court, she
arranges some kind of game: swimming out from male, she calls attractively,
and male follows her, stirring up feathers of “collar”. If male succeeds to
bar the way to the female, it means her consent to nesting.
This bird belongs to flying species and spends in Antarctica only summer when
the rivers and lakes are free from ice. These birds fly to Antarctica in second
half of spring when the ice on rivers of continent already starts to broken
up. The first days after migration to the continent birds have a rest, but,
in process of ice thawing, they start courtship games.
Antarctic grebe is strictly zoophagous species of birds. It eats mainly invertebrates,
catches shrimps and larvae of dragonflies. Occasionally it can seize a fish,
but, as a rule, it prefers to hunt in extensive shallow lakes, stiring ground
by beak and frightening invertebrates. This grebe dives to 1 – 2 minutes, but
moves under water very quickly and cucceeds to seize several larvae of fish
dragonflies – characteristic freshwater insects of Antarctica. Bird almost
does not put efforts to emerge: its density is lighter than water, and it is
enough for it to stop paddling by paws, and water pushes it out to the surface.
The Antarctic grebe nests on rafts of sedge and other plants which freely float
on water surface. It builds rather big rafts with high heap of grass in middle.
This is an adaptation for life in cold lakes: even in middle of summer water
in Antarctic lakes all the same is cold, and in high nest at the hatching cold
water does not fill in clutch. In clutch of this species there are only 2 –
3 large eggs. Chicks hatch after 18 – 19 days of incubation. They at once leave
nest and swim with parents. During the brood rearing birds keep in shallow parts
of water bodies – due to it chicks have an opportunity to feed on larvae of
small insects, midges and mosquitoes. Chicks grow intensively, and within one
summer have time to reach the size of adult individuals and to learn flight
skills. They should be ready to go to a long journey for wintering when first
frosts will come.
For wintering Antarctic grebe migrates to New Zealand, stopping in its way only
at subantarctic volcanic islets. At these islets it has a rest and refreshes
forces with the invertebrates caught in local freshwater reservoirs. This bird
is not adapted to life in sea, because it can not excrete the salts recieved
to its organism with sea water. Only birds tired during their flight alight
on sea surface, but they do it only for rest. They are afraid to dive during
the rest, and try to fly up and to continue their way as soon as possible. In
New Zealand these birds prefer to settle in mountain lakes and in upper courses
of rivers where climate is more habitual for them.
The idea about existence of present species was proposed by Simon, the forum member.
Dusky
grebe (Podilymbus columbianus)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: North America, Miche-Nama Lake, freshwater reservoirs.

Picture by Tony Johnes
Among the overall diversity of birds, grebes are a relatively
modestly represented group – they are not numerous and often small in size.
Like loons, they are almost unable to walk on land and live almost exclusively
in water – some of them even nest on floating islets of vegetation. Despite
the relatively beautiful plumage, people almost never hunted these birds, and
their specialization in the aquatic lifestyle helped them survive natural disasters
at the turn of the Holocene and Neocene. Many local species of these birds became
extinct in the human era, but in the Neocene, the active speciation of the surviving
species compensated for this damage.
In Neocene, grebes live all over the world; there are flightless species among
them, but this is an exception to the rule – most of them are relatively good
flyers, although they feel more confident on the water and under water. Dusky
grebe, a descendant of the pied-billed grebe (Podilymbus podiceps) is one such
bird.
Compared to its ancestor, it is really large bird, reaching up to 1.3 m in length,
mainly due to its elongated neck and long beak. It has retained the proportions
of a diving bird and is covered with water-repellent plumage of a soft pale
brown color. Sometimes, especially in northern populations, there are also lighter
“roan” birds. The beak, like that of the ancestral species, is brighter than
the plumage – yellow with a black vertical stripe stretching along the upper
and lower mandibles.
Dusky grebe is an exclusively zoophagous bird feeding on fish, amphibians, less
often crayfish and other large aquatic invertebrates. Its enemies are predatory
fish, less often birds and water snakes; on Mishe-Nama Lake, it sometimes comes
into conflict with the dwarf gannetwhale,
but it rarely happens. Dusky grebe is not a territorial bird and prefers to
retreat in such conflicts, losing also to the dwarf gannetwhale in weight; in
addition, it prefers to live alone in the coastal area of the lake, and therefore
is not a competitor to the flocking gannetwhale living in the deep middle part
of the lake. The attacked bird, however, is quite a serious opponent and strikes
the enemy hard with its wings and beak. Therefore, predators prefer to attack
young birds, grabbing them from under the water or attacking from the air. Adult
grebes are attacked by feathered predators during migrations.
Like related species, dusky grebe makes a floating nest by throwing a large
amount of twigs and grass into the water. During the nesting season, it usually
avoids appearing on Mishe-Nama Lake, and nests in shallower and overgrown reservoirs,
where it is easier for it to find both food and nest material. This bird also
avoids swamps, preferring to settle on large lakes and rivers with a large expanse
of open water. These birds usually avoid each other’s company and live quietly
– only in spring, during the courtship season, they resound North American reservoirs
with a loud high-pitched whistle: these are the males calling the females to
them. The females came at their call join the males and the birds begin to whistle
in a duet. But by the time the nest is built, the birds stop vocalizing.
Dusky grebe forms families for only one season; both birds build a nest and
hatch eggs, numbering from three to eight. The chicks hatch in a month, and
at the age of one and a half weeks they are already able to dive as well as
their parents, and by the beginning of autumn they take wing and migrate.
Dusky grebe is a migratory bird, and flies to the south of the continent for
the winter; families fly together at this time, but they usually break up at
the wintering site, and in the spring young birds already form new pairs. The
average life expectancy of this bird is up to 15 years, but many birds die in
the first years of their life.
This bird species was discovered by Bhut, the forum member.
Volcanic
lake grebe (Nanopodiceps volcanophila)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: Japan, high-mountainous volcanic lake at Hokkaido Island.

Picture by Fanboyphilosopher
In human epoch birds of grebes order were widely settled on
Earth and lived at all continents, except for Antarctica. These birds live in
fresh-water reservoirs, perfectly swim and dive, and hunt small fish. In human
epoch among these birds there was a plenty of local and endemic species, and
some of them could inhabit only one small lake. Such species appeared the most
vulnerable ones at destruction of the natural environment as a result of economic
activity of people, and in human epoch some similar species of grebes had irrevocably
died out. But most common and widespread species had escaped, and after human
disappearance they continued evolution.
Japan Islands became a favorable place for evolution of this group of birds.
In Neocene at the archipelago some kinds of local flightless grebes had evolved
at once. The landscape of islands quite favours to this event: it is heterogenous
enough, differing in abundance of isolated lakes, and in conditions of warm
climate of Neocene and volcanic heating of some lakes their biological efficiency
is high enough to support a settled population of gluttonous and fastidious
in food birds.
All Japanese lake grebes belong to one genus Nanopodiceps, and are descendants
of one species widely settled on islands. The ancestral species is able to fly,
but its local descendants had completely left skills of flight. They are birds
with narrow head, long beak, wide paws and almost completely absent wings. At
all lake grebes of Japan Islands the body is streamlined, and tail is almost
absent. Swimming under water, these birds extend head forward, and paddle by
paws, working by them alternately, or making simultaneous pushes. Wings of flightless
species are advanced in various degrees – from small, but appreciable up to
completely reduced ones. The reduction of wings at these birds occured independently
from each other.
Japanese lake grebes have bright colored “collars” similar to plumage of great
crested grebe’s head, and some species have “horns” of lengthened feathers.
It is connected to necessity of identification of relatives – the ancestral
species frequently appears on lakes populated with its descendants, and strongly
expressed distinctive attributes help to exclude formation of hybrids.
Food of lake grebes includes small fishes, crabs and shrimps. Birds are able
to dive and easily chase prey under water. They eat exclusively live prey, swallowing
it entirely.
All species nest among coastal vegetation, arranging primitive flat nest. Frequently
nest happens half submerged in water, but even in this case the incubation of
eggs passes successfully. Usually in clutch may be 3 – 4 eggs – at small species
clutch is lesser, at large it is more (up to 6 eggs).
Volcanic lake grebe lives at the north of area of this genus. Its habitat is
unusual: it inhabits an only mountain valley, in which the lake about ten square
kilometers by the area is stretched. At the bottom of this lake volcanic thermal
springs spurt, and temperature of water even in winter is not less +20°С. Volcanic
lake grebes nest in thickets of reed bordering this lake. Due to original microclimate
water and marsh plants do not stop to grow even in winter, providing birds with
shelter, and their prey with food.
Volcanic lake grebe is tiny bird; its weight does not exceed 200 grammes. Plumage
on the body of this bird is monotonously grey, darker on neck. Colouring of
head of volcanic lake grebe is very bright and appreciable from apart. Feathers
on nape and back part of male’s neck form bright “flag” – they are strongly
extended and directed back. Females lack of such “flag”, but colouring of plumage
of the appropriate parts of their heads is the same, as at males. Forward part
of head and top of head at birds of both genders is black and lateral parts
of head and “flag” are bright orange with reddish shade. At some males tips
of feathers of “flag” are white. The iris of eyes at male has impressive white
color; at female it is brown.
Wings at this bird are very small – their length does not exceed 3 – 4 cm.
At Japan Islands other species of lake grebes live:
Common
lake grebe (Nanopodiceps volans) is the only species of the genus had
not lost ability to fly. Obviously, this species or its direct ancestor became
the ancestor of all flightless endemic grebes of Japan Islands. This bird has
bright blue paws (it is the unique colouring which is not meeting at other species
of genus), brown plumage on the body and white “saddle” – site of plumage on
back and under wings, well appreciable during flight of bird. From a nape on
back side of neck small “crest” of white feathers with black tips grows. In
courtship season males of this species show it to females, trembling by head
and neck.
Common lake grebe is settled at all Japan Islands and lives mainly on mountain
rivers, avoiding a competition to settled flightless lake species. Flightless
species of lake grebes have similar biology and differ from each other in colouring
and features of courtship ritual.
Southern
lake grebe (Nanopodiceps australis) inhabits the south of Hokkaido
Island. It has light colouring – pale grey plumage with brownish shade on back
and snow-white stomach. The cop at male is black with dark blue metal shine,
and around of eyes there is a ring of black feathers. The female of this species
has only black spot on nape, and it shows this mark to the male as a symbol
of submission during the courtship ritual. This bird weighs no more than 300
grammes. Wings at this bird are very small; only few reduced primarily feathers
are appreciably from outside.
Semi-striped
lake grebe (Nanopodiceps striatocollis) inhabits lake at the south
of Honshu Island. This species has darkest colouring of plumage – black with
separate white sites. From nape along back part of neck up to shoulders on black
background narrow longitudinal strips of white color stretch. During courtship
display male fluffs feathers on nape and neck, and strips become wider and more
appreciable. He displays to the female plumage on neck, swimming around her
and having turned a head by nape to her. Wings at this species are reduced stronger,
than at other species of genus: they are completely invisible from outside,
and only on skeleton the rudiment of humeral bone is visible.
Yellow-headed
lake grebe (Nanopodiceps flavicephalus) lives in several mountain lakes
and rivers of Shikoku Island (Shikoku-Sanchi mountains). It has grey color of
body plumage, and head and top part of male’s neck are bright yellow. At the
female yellow color is replaced by yellowish-brown one. On nape and back part
of neck of this bird the strip of black feathers passing to black plumage of
back stretches. It is the largest species of genus – weight of adult bird reaches
700 – 800 grammes.
This bird nests in reeds, preferring the thickets growing at the distance from
coast, when it is impossible to reach to it from the coast. Frequently these
birds arrange nests on islets of floating vegetation, and nesting pair specially
rakes large heap of plants, pulling them from ground, and strengthens the construction
with rods.
The idea about existence of this group of bird species was proposed by Simon, the forum member.
Chatham
sea grebe (Parapodiceps chathamensis)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: Chatham islands.

Picture by Alexander Smyslov
Due to ability to flight birds had occupied almost all ocean
islands, from cold Arctic regions up to equally cold Antarctic Region. During
the ice age Chatham Archipelago in Pacific Ocean represented the ecosystem very
poor in species variety and economically using poor resources. But in Neocene
the situation at the islands has changed: numerous birds and insects had appeared,
descendants of rats and mice introduced by people also evolved, and islands
have become overgrown with bushes and trees. The largest terrestrial species
of local fauna is Chatham barocygnus
– flightless descendant of black swan.
At the islands numerous freshwater reservoirs appeared – small lakes and bogs.
In them mosquitoes, dragonflies and other insects breed plentifully. Larvae
of insects represent one of main food sources for other inhabitant of islands
– for Chatham sea grebe. This bird, the descendant of great crested grebe (Podiceps
cristatus), is original analogue of loons (order Gaviiformes) of Northern hemisphere
in southern temperate latitudes.
Chatham sea grebe is rather large representative of the order, well flying bird
weighting about 400 grammes. It has lengthened body of streamline outlines;
legs are shifted to back part of body, therefore bird is not able to walk on
land, but only creeps on stomach, pushing by legs. Flexible mobile neck and
pointed awl-like beak give out the skilful hunter for water animals in this
bird. Chatham sea grebe can dive for a long time and swims under water up to
50 meters in searches of prey.
This species of birds has very characteristic colouring: back, wings, top part
of neck and head are grayish-blue; stomach and the bottom part of neck are white.
On cheeks of bird there are large white spots. Eyes are yellow and ringed with
a thin strip of black feathers. Feather “collar” characteristic for its ancestor
has almost completely disappeared: only “whiskers” of grayish feathers, more
advanced at male, remained from it. Feather “horns” also have decreased in size
and became very short. In courtship season displaying male raises them upwards,
swimming around the female. The beak of Chatham sea grebe is colored bright
red – birds display it to the partner during the courtship.
This species of birds has found original enough decision of a problem of limitation
of resources in freshwater reservoirs of small islands of archipelago. Freshwater
reservoirs are used by these birds only for nesting and rearing of posterity,
and adult birds search for food in sea. Chatham sea grebe eats various invertebrates
and fish fry. In fresh waters of Chatham archipelago there are no fishes, except
for the separate species coming from the sea, therefore the young growth of
this species finds plentiful food like mosquitos and other insect larvae. Adult
birds, replacing each other on the nest, fly to the sea for hunting. In coastal
zone, among thickets of brown macroalgae, adult birds hunt shrimps, small crabs
and octopuses. Besides high sea waves do not enable birds to build floating
nests. The seasonal climate of islands allows birds to nest only once per one
year.
The main problem at life in the sea is the necessity of removing of superfluous
amount of salt from an organism. At Chatham sea grebethe function of salt removing
the modified part of lachrymal gland has undertaken, separated from actually
lachrymal gland and opening as a separate channel in eye-socket. From time to
time the bird “cries”, splashing brine out as small jets, and at once washes
out eyes, dipping head in water. These birds hide from storm among thalli of
brown algae which soften impacts of waves in great degree, or in the coastal
caves protected from surf by stones.
Chatham sea grebe nests in freshwater lakes and bogs in numerous congestions.
As adult birds have almost unlimited source of food, this species forms significant
congestions – up to several tens couples of birds in the same small lake. At
this species the nest is typical for grebes – it is a floating heap of plants.
Sometimes some nests hook together, and birds continue eggs hatching, not paying
attention to new neighbours.
In clutch of this species there are up to three eggs with motley shell. Chicks
are well advanced and independent. Having dried under mother, they at once start
to search for food independently, diving to the depth up to 50 – 60 cm. Chicks
have brown down with narrow longitudinal straw-coloured strips and rather short
beak. Salt-emitting gland at them is yet underdeveloped, and therefore they
are not able to live in sea water. Parents warm and preserve posterity within
the first month of life. Only at the age of three weeks at young birds salt-removing
gland starts to develop. At this time they undertake very dangerous travel overland
to the sea coast. Chatham sea grebes especially willingly nest in lakes from
which the small river flows to the sea even if it forms a falls. These birds
almost do not settle in lakes far from the sea, or chicks should overcome some
hundreds meters of land in total, passing from lake to lake before they will
succeed to reach the sea coast. Chicks can move on land, keeping body in almost
vertical position. They hardly keep balance, helping to themselves with wings.
During this transition parents fly from lake to lake, calling up chicks with
loud cries. Not seeing parents, chicks choose a correct direction of movement,
being guided on their voices.
Having reached up to the sea, young birds study larger prey hunting, and parents
complete their rearing within one month. Young birds begin independent life,
not being able to fly at all. They start nesting at one-year-old age.
Antarctic
grebe (Cryopodiceps australissimus)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: freshwater reservoirs of Antarctica.

Picture by Simon
In Neocene Antarctica pushed aside by South America, had moved
aside Australia. Shift was not far, but it has permitted to weaken considerably
the cold Antarctic Circumpolar current, which did not let warm waters from equator
to the continent. Climate in Antarctica of Neocene epoch is still seasonal with
severe winter, but some birds had adapted
to all-the-year-round life in this severe place. Antarctic summer in Neocene
is favorable enough for life of birds and almost right after snow thawing bird
flocks from South America, Australia and New Zealand direct to this continent.
Among summer visitors of Antarctica there is one species of grebes, which is
the descendant of Tasmanian population of great crested grebe (Podiceps cristata),
which has rather easily gone through human epoch due to vast area, having kept
in “islets” of the earlier solid area. Possible, casual moving of these birds
from Tasmania through subantarctic islands has permitted
them to reach Antarctica and to join other summer visitors of this continent.
Antarctic grebe lives in rivers and lakes of Antarctica. The size of this bird
is medium for grebes: it weighs about 200 grammes. Larger birds hardly could
maintain successfully rather slowly renewing food resources of freshwater reservoirs
of cold Antarctica. For Antarctic grebe the streamline body shape is characteristic;
legs are shifted far back, and this bird is not able to walk on land. The body
feathering is colored black. It does not prevent this bird to hunt, because
its basic prey is made of larvae of insects and crustaceans. But in conditions
of cool Antarctic summer such colouring helps to be warmed quickly during the
rest. Favourite resting places of Antarctic grebes are flat stones, which one
edge is inclined to water. The bird creeps on such stone, pushing by legs, and
has a rest on it for a long time, having fluffed up feathers and basked in sun.
Bright “collar” of cross-striped black-and-white feathers with expressed metal
shine contrasts with dim color of plumage. In courtship season this “collar”
is stretched as a semicircle – feathers on nape rise upwards, and “moustaches”
are turned downwards. Male involves the female, shivering these feathers which
brightly shine in sunlight. If the female is ready to accept his court, she
arranges some kind of game: swimming out from male, she calls attractively,
and male follows her, stirring up feathers of “collar”. If male succeeds to
bar the way to the female, it means her consent to nesting.
This bird belongs to flying species and spends in Antarctica only summer when
the rivers and lakes are free from ice. These birds fly to Antarctica in second
half of spring when the ice on rivers of continent already starts to broken
up. The first days after migration to the continent birds have a rest, but,
in process of ice thawing, they start courtship games.
Antarctic grebe is strictly zoophagous species of birds. It eats mainly invertebrates,
catches shrimps and larvae of dragonflies. Occasionally it can seize a fish,
but, as a rule, it prefers to hunt in extensive shallow lakes, stiring ground
by beak and frightening invertebrates. This grebe dives to 1 – 2 minutes, but
moves under water very quickly and cucceeds to seize several larvae of fish
dragonflies – characteristic freshwater insects of Antarctica. Bird almost
does not put efforts to emerge: its density is lighter than water, and it is
enough for it to stop paddling by paws, and water pushes it out to the surface.
The Antarctic grebe nests on rafts of sedge and other plants which freely float
on water surface. It builds rather big rafts with high heap of grass in middle.
This is an adaptation for life in cold lakes: even in middle of summer water
in Antarctic lakes all the same is cold, and in high nest at the hatching cold
water does not fill in clutch. In clutch of this species there are only 2 –
3 large eggs. Chicks hatch after 18 – 19 days of incubation. They at once leave
nest and swim with parents. During the brood rearing birds keep in shallow parts
of water bodies – due to it chicks have an opportunity to feed on larvae of
small insects, midges and mosquitoes. Chicks grow intensively, and within one
summer have time to reach the size of adult individuals and to learn flight
skills. They should be ready to go to a long journey for wintering when first
frosts will come.
For wintering Antarctic grebe migrates to New Zealand, stopping in its way only
at subantarctic volcanic islets. At these islets it has a rest and refreshes
forces with the invertebrates caught in local freshwater reservoirs. This bird
is not adapted to life in sea, because it can not excrete the salts recieved
to its organism with sea water. Only birds tired during their flight alight
on sea surface, but they do it only for rest. They are afraid to dive during
the rest, and try to fly up and to continue their way as soon as possible. In
New Zealand these birds prefer to settle in mountain lakes and in upper courses
of rivers where climate is more habitual for them.
The idea about existence of present species was proposed by Simon, the forum member.
Dusky
grebe (Podilymbus columbianus)
Order: Grebes (Podicipediformes)
Family: Grebes (Podicipedidae)
Habitat: North America, Miche-Nama Lake, freshwater reservoirs.

Picture by Tony Johnes
Among the overall diversity of birds, grebes are a relatively
modestly represented group – they are not numerous and often small in size.
Like loons, they are almost unable to walk on land and live almost exclusively
in water – some of them even nest on floating islets of vegetation. Despite
the relatively beautiful plumage, people almost never hunted these birds, and
their specialization in the aquatic lifestyle helped them survive natural disasters
at the turn of the Holocene and Neocene. Many local species of these birds became
extinct in the human era, but in the Neocene, the active speciation of the surviving
species compensated for this damage.
In Neocene, grebes live all over the world; there are flightless species among
them, but this is an exception to the rule – most of them are relatively good
flyers, although they feel more confident on the water and under water. Dusky
grebe, a descendant of the pied-billed grebe (Podilymbus podiceps) is one such
bird.
Compared to its ancestor, it is really large bird, reaching up to 1.3 m in length,
mainly due to its elongated neck and long beak. It has retained the proportions
of a diving bird and is covered with water-repellent plumage of a soft pale
brown color. Sometimes, especially in northern populations, there are also lighter
“roan” birds. The beak, like that of the ancestral species, is brighter than
the plumage – yellow with a black vertical stripe stretching along the upper
and lower mandibles.
Dusky grebe is an exclusively zoophagous bird feeding on fish, amphibians, less
often crayfish and other large aquatic invertebrates. Its enemies are predatory
fish, less often birds and water snakes; on Mishe-Nama Lake, it sometimes comes
into conflict with the dwarf gannetwhale,
but it rarely happens. Dusky grebe is not a territorial bird and prefers to
retreat in such conflicts, losing also to the dwarf gannetwhale in weight; in
addition, it prefers to live alone in the coastal area of the lake, and therefore
is not a competitor to the flocking gannetwhale living in the deep middle part
of the lake. The attacked bird, however, is quite a serious opponent and strikes
the enemy hard with its wings and beak. Therefore, predators prefer to attack
young birds, grabbing them from under the water or attacking from the air. Adult
grebes are attacked by feathered predators during migrations.
Like related species, dusky grebe makes a floating nest by throwing a large
amount of twigs and grass into the water. During the nesting season, it usually
avoids appearing on Mishe-Nama Lake, and nests in shallower and overgrown reservoirs,
where it is easier for it to find both food and nest material. This bird also
avoids swamps, preferring to settle on large lakes and rivers with a large expanse
of open water. These birds usually avoid each other’s company and live quietly
– only in spring, during the courtship season, they resound North American reservoirs
with a loud high-pitched whistle: these are the males calling the females to
them. The females came at their call join the males and the birds begin to whistle
in a duet. But by the time the nest is built, the birds stop vocalizing.
Dusky grebe forms families for only one season; both birds build a nest and
hatch eggs, numbering from three to eight. The chicks hatch in a month, and
at the age of one and a half weeks they are already able to dive as well as
their parents, and by the beginning of autumn they take wing and migrate.
Dusky grebe is a migratory bird, and flies to the south of the continent for
the winter; families fly together at this time, but they usually break up at
the wintering site, and in the spring young birds already form new pairs. The
average life expectancy of this bird is up to 15 years, but many birds die in
the first years of their life.
This bird species was discovered by Bhut, the forum member.
Golden-crawed
collar pigeon (Collaricolumba chrysocervix)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: Japan Islands, woods in foothills.
Japan Islands are surprising from the point of view of fauna and flora – here
there is one of points of contact of nature of cold north and hot south. Northern
species move to tropics along mountain ridges. And animals and plants of tropical
origin settle to the north due to warm ocean currents warming islands in winter.
Japan Islands represent the extended mountain circuit rising from oceanic bottom.
They serve as though as a barrier for a way of damp winds from Pacific Ocean,
therefore life conditions at the islands are very favorable for forest growth.
Especially dense woods grow on east slopes of mountains turned to ocean. Wood
habitats always were very favorable for various birds.
Pigeons belong to the number of characteristic wood birds of Old World. A lot
of species of pigeons had suffered from human activity and became a rarity (and
some ones had vanished completely), but some pigeons appeared one of the most
successful birds of human epoch due to ability to coexist people. Some species
of these birds settled in anthropogenous landscape and widely settled at the
Earth. After human extinction the part of populations of these species had become
extinct, but some ones had successfully adapted to independent existence. Till
the process of evolution among descendants of former people satellites the original
species evolved, distinguished by interesting features of anatomy. Usually various
kinds of “ornament” are the result of the action of sexual selection and have
high specific specificity, strongly differing even at related species.
One of results of sexual selection is shown by collar pigeons – forest birds
of medium and large size living at Japan Islands. At first sight birds of various
species of this genus are very similar to each other – their back and wings
are colored light brown and have slight cross ripple; and stomach is dark. At
all collar pigeons there are rounded wings and rather long tail (approximately
half of general length of a bird): it indicates that they are adapted to maneuverable
flight among branches.
These birds have very prominent feature of the feathering which has determined
their name. Collar pigeons have the lengthened feathers like a mane on the top
part of neck. On sides and front part of neck feathers are small. But they have
strong metal shine, which color differs at various species. When the bird is
quiet, the lengthened covering feathers hide shining feathers of neck, as if
the cloak. But in courtship display time collar pigeons are difficult for not
noticing.
Courtship display at all species of collar pigeons represents a spectacular
show. Displaying bird perches on branch vertically, turns feathers of mane in
sides, puffs craw and coos loudly. Feathers of mane stick up in sides like fans,
creating an impression of magnificent collar of medieval Spanish fashion. At
displaying bird feathering of craw reflecting sunlight becomes well appreciable.
To present itself to females to the best advantage, males gather at well lighted
tops of trees, and display the feathering to females flying above them. For
courtship display males gather to flocks of some tens birds. When they start
display ritual simultaneously, their voices are similar to roar of drums or
mutter of crowd.
The female chooses the nesting partner to itself for some years, and sometimes
for whole further life. Generated pairs take part in joint display, but females
do not leave displaying males, and keep near to them. Collective display, strangely
enough, strengthens family ties. After it partners much more willingly start
nesting – obviously, at this time at them the contents of hormones in blood
raises.
As well as all species of pigeons, collar pigeons do not differ in nest-building
talents. Their nest represents the friable heap of twigs heaped up in forked
branches. The inner part of nest is covered with locks of moss and epiphytic
plants. On poor litter the female lays two eggs, and both partners alternately
hatch them till 20 – 21 days. Nestlings come into the world covered with thin
down, helpless and blind. Parents feed them with “bird milk” – curdled secretions
of craw walls. Nestlings grow quickly, and already at the age of 30 days leave
nest. Young birds differ from adults in shorter mane and dim feathers on craw.
They get colouring of adult birds approximately at five-monthly age when they
cast feathers first time. At the age of approximately 11 months young birds
take part in courtship display and can nest first time. At large species terms
of nestlings development are approximately two weeks longer, than at smaller
ones.
Golden-crawed collar pigeon is colored rather impressively: feathers on its
neck have golden yellow shine, and this feature determined its specific name.
Other feathering is colored very modestly: head is black with white “eyebrows”;
top part of body is brown with dark scaly pattern. Tips of long tail feathers
are colored white. Feathers under the tail of the bird are also white.
At golder-crawed collar pigeon there is rather strict feeding specialization,
which allows it to avoid the competition to closely related species. This bird
eats fruits of plants of ginseng family (Araliaceae), including ones inedible
for other birds and poisonous for mammals.
At Japan Islands other species of collar pigeons differing in food predilections
live:
Black
collar pigeon (Collaricolumba nigra) differs from other pigeons of
this genus by very dark colouring – body of this bird is black with hardly appreciable
brownish shade on wings. On this background snow-white “glasses” and feathers
of breast – white with bluish metal shine – are brightly marked out. This bird
avoids a competition to other species of collar pigeons, because it is specialized
to feeding on fruits of trees of Sapindaceae family. Such fruits frequently
are poisonous for forest mammals and birds, but these pigeons eat them even
unripe or beginning to rot, when they are especially poisonous. At this time
even meat of pigeons had a meal such fruits, can become inedible. This bird
lives in small flocks in forests at the south of Japan Islands where the typical
rainforest grows.
Tiny
collar pigeon (Collaricolumba pumilis) is the smallest species of genus:
by size it is equal to smallest turtledoves of Holocene epoch. At these birds
feathers on breast are red with metal shine. It eats seeds of various plants,
preferring seeds of plants of carrots family, which often are poisonous for
other birds and mammals, and lives in flocks of some tens birds. These fine
pigeons can be met in thickets of huge carrot grass, the fennel tree. It was
settled far to the north in places of fennel tree growth, and nests in coastal
woods of Japan Islands and Big Kurils. In summer separate flocks of these pigeons
reach the south of Kamchatka.
Macaw-like
collar pigeon (Collaricolumba araroides) is the largest species of
genus: up to one meter long including long tail. It lives in western half of
Japan Islands, and even reaches Asian coast, where it had formed settled population.
Its colouring is brighter, rather than at other collar pigeons: bluish-grey
top of body with very thin cross ripple and yellow stomach (by size and colouring
this pigeon is a little similar to one species of macaw parrots from South America).
Feathers forming collar on neck of bird have the expressed metal shine, and
the forward part of neck and top of breast of bird are covered with black feathers
with strong green metal shine. This species eats seeds and small fruits, swallowing
them entirely. In connection with such food beak of bird is rather long and
thin. Bird does not crush seeds like parrots, but at first ferments them in
craw (secretions of special glands in walls of craw promote this process), and
then frays in muscular stomach with the help of pebbles. Sometimes pigeon searches
and swallows small beetles, having firm armour, for same purpose.
Hatoyoku
(Barotreron phyllophagus)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: Japan Islands, lowland evergreen forests in the southern part of the
archipelago.

Picture by Pavel Volkov
The evolution of living organisms on isolated islands can lead
to the appearing of living creatures of unusual appearance, which may occupy
unusual ecological niches. In the evergreen forests in the south of Japan Islands,
one of these peculiar species lives – large hatoyoku pigeon, descended from
one species of green pigeons (Treron) of the human era. An adult bird of this
species weighs about 2 kg – about the size of a chicken, hence the name of the
bird, meaning “rooster pigeon” in Japanese; the wingspan is up to 120 cm. Hatoyoku
retains the characteristic appearance of pigeons, adjusted only for the large
size of the bird. In this species, the background color of the plumage is grassy-green,
with a faint bronze sheen on the neck in males; the secondary feathers are black
with white tips, clearly visible during flight. Legs are bare, of meaty red
color. Beak is deep and wide, black with a white cere; mouth is wide. Birds
of both sexes have light yellow eyes with a round pupil and bright pink eyelids.
This species inhabits exclusively evergreen forests in the southern part of
the archipelago, because it has a special food specialization – it feeds mainly
on young leaves of trees. Such food specialization has led to changes in the
anatomy of the internal organs of hatoyoku. The crop of this bird is very voluminous;
it allows this bird to process a large volume of greenery at the same time with
the help of numerous symbiotic bacteria. A partial fermentation of fiber occurs
in the crop, which continues in the long intestine. Due to the large body weight,
hatoyoku flies reluctantly and for a short time, mainly flying from one tree
to another and occasionally flying over narrow rivers. An additional factor
that reduces the bird’s ability to fly is a highly developed crop, due to which
the size of the sternum and the flying muscles attached to it has decreased.
This species feeds on the leaves of various trees, mainly eating soft young
leaves at the tips of branches. Hatoyoku is able to climb trees deftly and cling
to branches with the help of rough corneous pads on the underside of its toes.
When digesting food, hatoyoku alternates periods of active feeding with periods
of inactivity, when food is processed in the crop. At this time, bird prefers
to hide in the foliage, next to the trunk of a tree. Defending itself from the
enemy, hatoyoku spits out a stream of semidigested feed from the crop into it.
If the enemy gets too close, hatoyoku turns back to it and shoots stinky sticky
droppings. The bird flies away from the enemy only in case of extreme danger:
hatoyoku is able to fly no more than 200-300 meters at a time, after which it
must rest. This bird flies very poorly against the wind and prefers to wait
out storms on a tree or among rocks, firmly clutching the support with its paws.
At this species, a pair is formed for life. During the courtship display, the
male coos in front of the female, greatly inflating the crop, and the skin on
his eyelids is filled with blood, which makes the eyelids become clearly visible;
warty growths appear on them. The displaying male spreads his tail like a fan
and lowers his wings, making visible the bright color of the secondary feathers.
Hatoyoku arranges a large nest in the upper part of the tree crown. Birds build
it from broken branches and twigs heaped into the fork of the branches, renewing
it every year if the nest has not been occupied by birds of other species. There
are 2 eggs in the clutch, only the female incubates. The male feeds her with
a leaf pulp partially processed in the crop. Incubation lasts about 20 days.
Nestlings are naked and blind, at the age of one week they grow a coverof thin
down and fully develop at the age of 40-45 days. They are fed with crop milk,
later they switch to eating fermented leaves. Birds nest twice a year. Sexual
maturity occurs at the age of 1 year, life expectancy is about 15 years.
The anonymus from Dobrochan helped to clarify the name of the bird.
Owl-pigeon
(Tytomimus tytomimus)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: tropical forests of Jakarta Coast and Southeast Asia.
In the nature of Neocene epoch, many examples of cryptic coloration and mimicry
can be found, which are demonstrated by representatives of various groups of
living organisms. This survival tactic was widely known on the Earth in the
human epoch, and became equally widespread in the subsequent epoch.
One of the species demonstrating this kind of adaptation is large pigeon species
that lives in the tropical forest canopy of Southeast Asia and nearby islands.
It is a bird weighing about 1 kg, with a wingspan of up to 1 meter. The appearance
of this bird is quite typical for pigeons; it has a small head and a thin beak
that can open wide. Wings of this bird are of rounded outlines: owl-pigeon lives
in the dense forest canopy and its flight is quite slow. Tail of this bird is
wide and fan-shaped. A feature that distinguishes this species from most pigeons
is relatively long legs. Owl-pigeon often walks along the branches, only fluttering
from one branch to another if necessary.
The plumage on the neck and on the top of the head of this species is dense,
and the feathers have greatly expanded vanes. Because of this, the bird’s neck
seems thicker than in most other pigeons.
The coloring of this pigeon is very peculiar, but its true meaning is clear
when the bird is defending itself from the enemy. The body of this pigeon has
a reddish-beige color with numerous individual variations from grayish and pinkish
to ochre-red. In different populations of owl-pigeons, a different background
shade prevails. Numerous spots of dark brown color are scattered over this background;
they are most common on the primary and tail feathers, as well as on the crown.
The lower part of the head, cheeks, front part of the neck and the upper half
of the chest of representatives of all populations are of snow-white color;
sometimes a dark border stretches along the lower edge of the white area. In
the middle of this area there is a large black spot stretched horizontally.
Sometimes there are some white feathers on black background.
Disturbed by a predator, owl-pigeon takes a protective pose. To do it, the bird
stretches the body vertically, strongly fluffs the plumage on the neck and head
and bends the head to the chest, laying it across the black spot. In this position,
the bird imitates a barn owl very accurately. The white plumage on the chest
mimics the owl’s face disk, and the lateral edges of the black spot become similar
to its eyes. The illusion is complemented by the movements of the bird (the
pigeon slightly opens its wings and stamps on its feet) and the sounds it utters.
Defending itself, the bird makes humming sounds, vaguely similar to an owl’s
hoot. Usually such an imitation is enough to scare monkeys or small tree-climbing
predators. If the deception is revealed, the pigeon flies away, flapping its
wings very loudly – this is an alarm signal.
The imitation of an owl is purely external: in behavior, this pigeon differs
little from most of its relatives. It feeds on the tree fruits, swallowing them
whole. This bird often pecks insects, getting the necessary protein feed.
Owl-pigeon is a solitary bird. Pairs of this species are formed for several
seasons, but outside of the nesting time, the birds stay alone and rarely meet
each other.
The courtship at these birds is accompanied by spectacular displays of the male.
Perching on the branch, he inflates his crop, throws his head back and fluffs
his feathers, visually enlarging the area of white plumage and the black spot
on it, which at this moment is facing upwards. The displaying male utters prolonged
humming sounds, while shaking his whole body.
The nest of twigs is arranged in the fork of the branches of a tall tree and
reaches about one meter in diameter. There are only two eggs in the clutch of
this species, and parents, as a rule, successfully feed both juveniles. Incubation
lasts 16 days, nestlings hatch covered with sparse down. They stay in the nest
for about 2 months, after which they leave it forever and their parents feed
them for about another week. The young bird lacks white area on the chest and
neck, and instead of a black spot, it has a dark brown one. After the first
molt, the bird has a plumage color characteristic of this species.
Several closely related species live on the islands of Southeast Asia:
Minor owl-pigeon
(Tytomimus minor) inhabits the forests of Sunda Land. It is a jackdaw-sized
bird with similar coloring features, but with darker plumage on the body.
Spectacled
owl-pigeon (Tytomimus strigops) lives on Philippine Islands. This crow-sized
species is distinguished by the presence of two eye-like spots on the chest,
black with a yellow border. The rest of the plumage is mottled, gray-brown with
spots, imitating the owl plumage.
Horned
owl-pigeon (Columbubo cornutus) also lives on Philippine Islands. This
is a large species (the weight of an adult is about 1500 g, the wingspan is
up to 120 cm) with a “mane” of elongated feathers on the neck and two large
ocular spots on the chest. Two tufts of elongated feathers directed backwards
and to the sides grow from the bird’s lower jaw. When a bird takes a protective
pose, it fluffs its plumage, exaggerating its own size. With the head bent to
the chest, the tufts of feathers look like the “horns” of an eagle-owl.
Mangrove
dove (Columba mangle)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: the coastal areas of South America in the tropical climatic zone.

Picture by Pavel Volkov
During the Holocene, the man introduced species in other continents.
Some of these introduced species were capable to survive in their new habitats
and give rise to descendants. The mangrove dove is one of these cases, a descendant
of the rock dove (Columba livia).
The mangrove dove has 30 cm of length and a wingspan of 63 cm. Its plumage is
mainly light brown in color, becoming darker on the back. The tail and primary
flight feathers are dark grey, the bill is red with a conspicuous white cere,
and the legs are purple-red. It has a grennish-purple iridescence along its
neck and breast feathers. The adult female is almost identical to the male,
but the iridescence on her neck is less intense and more restricted to the rear
and sides, while that on the breast is often very obscure.
This bird eats seeds, berries and invertebrates. The invertebrates are captured
when they forage in the mud of mangroves during the low tide, thus the name
mangrove dove. They also are seen in areas with sand near the sea searching
for small crustaceans, these doves will also eat beached algae and the invertebrates
hidden in it.
They feed in flocks, this way they use their numbers to avoid predation. They
also roost together at mangrove trees during the night. These doves are often
found in pairs during the breeding season, but usually these animals are gregarious,
living in flocks of 40 birds.
The mangrove dove breeds at any time of the year, but peak times are spring
and summer. The male on the ground puffs up the feathers on his neck to appear
larger. He approaches the female at a rapid walking pace while emitting repetitive
quiet notes, often bowing and turning as he comes closer. At first, the female
invariably walks or flies a short distance away and the male follows her until
she stops. At this point, he continues the bowing motion and very often makes
full- or half-pirouettes in front of the female.
Nesting sites are in coastal mangrove forests. The nest is a flimsy platform
of straw and sticks. Two white eggs are laid; incubation, shared by both parents,
lasts 17 to 19 days. The newly hatched nestling has pale yellow down and a flesh-coloured
bill with a dark band. For the first few days, the chicks are tended and fed
exclusively on “crop milk”. The fledging period is about 32 days. The young
reach sexual maturity with 8 months. The lifespan of this species is of 15 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Hawaiian
mountain dove (Paracolumba montana)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: mountains of Hawaiian islands.

Picture by Pavel Volkov
During the human epoch and also soon after its ending Hawaiian
islands had undergone the great number of changes, too strong for vunerable
island ecosystem. Anthropogenic pressure and climatic changes had left the traces
in local ecosystem, having almost completely destroyed native fauna and flora.
Representatives of new families and species replaced the extinct kinds. In many
cases birds were new migrants: usually they arrived to archipelago or storms
brought them from Polynesia, North America or Southeast Asia. Hawaiian mountain
dove is one of new birds of Hawaiian fauna, but it is the descendant of rock
pigeon (Columba livia) introduced to these islands in human epoch.
It is rather small bird: 20-25 cm long, wingspan about 40 cm and weighting about
200-250 grams. It nests high enough on slopes of Hawaiian mountains, in zone
of Alpine meadows, on the ground between stones. Alpine zones of Hawaii are
rather poor in vegetation, especially in places of recent volcanic eruptions,
and the predators dangerous to these doves are almost absent here. More often
this species nests in niches and between stones on abrupt mountain slopes, where
four-legged predators can not make the way.
Appearance of Hawaiian mountain dove has kept all recognizeable features of
pigeons. This bird has greenish-grey plumage, and at males well-visible black-and-white
mask is formed on face, indicating its maturity. Beak of these birds is deep
and has wide mouth cut, allowing swallowing large fruits entirely. The voice
resembles usual pigeon cooing, but sounds louder and less melodiously, rather
than at the ancestor of this species.
As Hawaiian islands are located in tropics, this pigeon has no expressed nesting
season and can nest all year round. This species is monogamous, and couple nests
during some years in succession. Courtship ritual represents “dance” of the
male with swollen craw around the female. At this time male opens tail like
fan and opens wings. The culmination of dance is the trembling of the whole
body of the male standing on extended legs.
The nest is primitive and represents heap of grass thrown on the bottom of shelter
and trampled down. Usually the couple of birds nests in the same place every
year, each time simply throwing a new layer atop of old grass. Both parents
hatch eggs in turns. In clutch there are 4-6 eggs with brown shell, ornamented
with pale specks for camouflage. Incubation lasts 16 days; nestlings hatch naked
and blind, eyes open at 5-th day of life, and they fledge completely and leave
nest at 35-37-th day of life. Within approximately one week adult birds finish
feeding of young ones. Birds can nest twice a year.
The food includes seeds and fruits found in forests growing down the mountain
slopes. At the time of bringing up of posterity the diet includes insects also.
Doves fly to feeding places in flocks numbering up to 200-300 birds. This dove
falls prey to various birds of prey and large bats, especially young ones. At
the nestlings staying in nest death rate is lesser because of nesting places
remote for the majority of predators, but they are attacked by some bats.
Life expectancy is about 7-10 years.
This species of birds is discovered by Bhut, the forum member.
White
crested pigeon (Lophocolumba elegans)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: Easter Island.

Picture by Pavel Volkov
During the Holocene, the man introduced many species to other
continents. Those that were able to survive left descendants in the Neocene.
The white crested pigeon is one of them. This species is a descendant of the
rock pigeon (Columba livia) which was introduced by the humans in Easter Island.
This species now inhabits all areas of the Island, being one of the members
of its endemic fauna.
The white crested pigeon has 40 cm of length and 72 cm of wingspan. The head,
back and rump are slate-gray. The tail is dark gray with white tips. Their main
characteristic is the pure white crest coming from the cere to the forehead.
Their neck, chest and belly are also white, while the wings are iridescent green.
The beak is red, while the legs and feet are pale pink. The male has a bright
blue cere, while it's pale in the females and subadults.
These birds are omnivorous, but prefer plant matter: chiefly fruits and grains.
They forage both in open areas and in trees. Larger flocks of white crested
pigeons can be seen feeding on fruits at trees.
The white crested pigeon is gregarious, forming flocks of 20 to 50 individuals.
Such groups can become larger when there is too much food. They are socially
monogamous, but extra-pair matings do occur, often initiated by males. This
bird breeds at any time of the year, but peak times are spring and summer. Nesting
sites are along coastal cliff faces, sometimes close to the colonies of seabirds.
Due to their ability to produce crop milk, pigeons can breed at any time of
year.
During courtship, the male will stay on the top of a tree cooing loudly. If
a female is interested, she will land in a branch near to him. Then, he will
start to puff up the feathers on his neck to appear larger, while flapping his
iridescent wings. At first, the female invariably flies a short distance away
and the male follows her until she stops. At this point, he repeats the same
ritual in front of her. The male then proceeds to feed the female by regurgitating
food, as they do when feeding the young. The male then mounts the female. The
mating is very brief, with the male flapping his wings to maintain balance on
top of the female.
The nest is a flimsy platform of straw and sticks, laid on the ledge of rocks,
under cover, often on cliffs. Two white eggs are laid; incubation, shared by
both parents, lasts 19 days. The newly hatched nestling has pale yellow down
and a flesh-coloured bill with a dark band. For the first few days, the chicks
are tended and fed exclusively on “crop milk”. The pigeon milk is produced in
the crops of both parents. The fledging period is about 30 days. They reach
sexual maturity in 6 month age and have a lifespan of 7-9 years, but many are
killed early by predators.
This bird species was discovered by João Vitor Coutinho, Brazil.
Hopi
(Zenaida hopi)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: deciduous forests of North America.
Picture by Sergey Ivanov
Despite of mass extinction at the boundary of Holocene and
Neocene which reasons were at first human influence, and then climatic changes,
various pigeons and doves had quite successfully overpassed this boundary and
had given rise to various, sometimes rather exotic new species. The majority
of “atypical” pigeons and allies lives in tropics, and in areas of more severe
climate species of quite recognizeable appearance live. One of these species
is hopi, pigeon from North America.
Hopi is a descendant of small mourning dove (Zenaida macroura) of Holocene epoch,
but it reaches 50 cm in length and seems even larger bird. Actually the significant
part of length falls on its motley tail which frequently distracts attention
of predator from the bird and gives hopi a chance to escape at an attack, having
lost only few feathers. Feathering colouring on body is soft, monotonous pearly-grey
and a little lighter on belly. But tail at this species is long, peaked and
resembling pheasant’s one a little bit. Tail feathers are colored with alternating
cross strips of light red and dark grey, almost black color.
Similarly to the ancestor, hopi eats various kinds of food as of animal, and
of vegetable origin. At the end of summer and in autumn these pigeons willingly
peck berries and even fly down on the ground to be fed. And in spring they eat
mostly insects as preparation for courtship season demands more energy, and
the female also must lay normal eggs. As a rule, these are flocking birds forming
close flocks of 3 – 4 breeding couples. Thus it is easier to them to escape
from predatory birds, mammals and snakes which are the main enemies of these
birds. Flocks of birds break up only for the nesting period.
Habitat of hopi is a zone of deciduous forests at the west of North America,
in Rocky Mountains foothills. These birds do not bear too bad cold and fierce
heat and because of it prefer to live in a zone of temperate climate. In winter
these birds migrate to the south along the ridges of Rocky Mountains. Thus they
rise in mountain forests, avoiding the heat weather in foothills.
The displaying period at hopi takes place at the end of winter or in the beginning
of spring as soon as birds return from wintering. For display males of this
species gather on branches of trees where leaves are not completely developed
yet, or on dry dead trees, and display themselves in front of females. Displaying
males lingeringly coo and show the tails, lifting them upwards, and lowering
heads down. At this time the craw is strongly inflated, and cooing becomes especially
loud. Young birds or the birds that have lost a parthner gather at the display
trees. The settled pairs are kept during several years, and in spring male displays
elements of courtship behaviour only to its female.
To the beginning of summer females already finish hatching of clutch numbering
2 – 3 eggs, nestlings have time to grow the down order, and from it the first
feathers start to grow. As well as other pigeon birds, hopi pigeons are bad
nest builders and, as a rule, they prefer to nest in abandoned nests of other
birds, rather than to build their own one. The couple of adult birds has time
to bring up two hatches for a season. Young growth of last hatch keeps with
their parents up to the wintering migration. The next year young birds become
capable to nesting. Life expectancy of hopi is up to 8 years.
This species of birds was discovered by Bhut, the forum member.
Wandering
dove (Zenaida erranta)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: South America, forests. Migrating species.

Picture by Pavel Volkov
A descendant of the eared dove (Zenaida auriculata), the wandering
dove is a nomadic and gregarious species, constantly migrating in search of
food, shelter and nesting grounds. They form enormous flocks with millions of
individuals, in constant migration from the forests of Tierra del Fuego to the
Southern Amazonian rainforest.
The wandering dove is 20 cm long with a long wedge-shaped tail. Adult males
have mainly olive-brown upperpart plumage, with black spots on the wings. The
head has a reddish-brown crown and on the sides of the neck and the upper mantle
they have iridescent display feathers of the colors violet or golden-green,
depending on the angle of the light. The underparts are vinous, and the tail
is tipped with cinnamon. The female is duller than the male, lacking the iridescent
feathers. The immature birds are greyish-brown, very dull, with pale barring.
In both sexes the bill is black and the legs dark red. Their diet consists of
seeds, fruits and shoots.
Their nomadic journey occurs as follows: Taking as a starting point the nesting
areas in the forests of Southern South America, they will fly to north after
the young are fledged. When the wandering doves reach the plains of Central
South America, the enormous flock separates into small groups that will stay
for a time in the plains. In the lasts days of autumn they will continue to
migrate north. These small groups will congregate again when they reach their
Amazonian nesting sites. After nesting here, they will fly back to the south,
staying for a time in the plains and going to the Southern nesting sites when
the conditions are favorable.
They form immense breeding colonies and realize cooperative breeding, with the
young receiving care not only from their parents, but also of others group members.
Courtship took place at the nesting colony, on a branch or perch. The male,
with a flourish of the wings, vocalize while near a female. The male then grip
tightly to the branch and vigorously flap his wings up and down. When the male
is close to the female, he will press against her on the perch with his head
held high and pointing at her. If receptive, the female press back against the
male. When ready to mate, the pair preens each other. As both sexes took care
of the nest, the pairs were monogamous for the duration of the nesting.
The wandering dove has two different nesting areas, one in the forests of Southern
South America and another in the Southern region of the Amazonian rainforest.
It builds a small stick nest several meters up in a tree and lays two white
eggs. These hatch in 13–15 days with another 9 days to fledging. This species
programs its reproduction to coincide with the fruiting of trees, in Southern
South America they arrive in their nesting grounds when the trees of the Family
Nothofagaceae have fruits, while in their Amazonian nesting areas they arrive
when the first fruits starts to appear. If the fruits take time to appear there
will be a high mortality of young. The young reach sexual maturity with 10 months.
Their lifespan is 10 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Pin-tailed
dove (Zenaida apicauda)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: Archipelago of Fernando de Noronha, forests.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. One
of these new species is the pin-tailed dove, a descendant of the eared dove
(Zenaida auriculata). This species inhabits the forests of the Archipelago of
Fernando de Noronha, but small populations inhabits the coastal areas of the
continent that are close to the Archipelago.
The males of pin-tailed doves are larger than the females. They have 28 cm of
length, while the females have 24 cm. Their size increases with the main characteristic
of the species, a 15 cm elongated central feather on their wedge-shaped tail.
They have a mainly chestnut upperpart plumage, with black spots on the wings.
The head has a grey crown, black line behind the eye, and an iridescent path
is found on the side of the neck. The underparts are dark brown, and the tail
is tipped with cinnamon. Female and juvenile coloration is slightly duller,
lacking the iridescence in their neck. The bill is black and the legs dark red.
These birds feed mainly on seeds, but they can also eat insects and small fruits.
They forage in the ground, forming small flocks of three to six individuals.
The pin-tailed dove is monogamous, with the male and female forming pairs that
last until one of them dies. When it happens, the surviving individual will
seek a new partner. It builds a solid but sparsely lined cup-shaped stick nest
in a tree and lays two white eggs. Incubation is 13 days with another 14 days
to fledging. There is no fixed breeding and provided with plentiful food, birds
breed almost continuously. Chick mortality through predation and falls from
the nest is high. They reach sexual maturity with one year and have a lifespan
of 13 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Blue-tailed
peafowl pigeon (Pavoenas cyanocauda)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: rainforests of Amazon basin.
Pigeons represent very common and widespread group of birds. In human epoch
some species of them got a great advantage from human activity, having widely
settled in human-changed habitats. In human epoch numerous large, endemic and
local forms of these birds had become extinct due to overhunting and destruction
of habitats, but some adaptable species became common faunal representatives
of urban and agricultural landscapes. In South America some pigeons of genus
Patagioenas appeared among synanthropes and widely settled in modified landscapes
throughout the continent. In post human era descendants of these birds appeared
among the common inhabitants of tropical forests restored in humid areas of
the continent.
In rainforests of South America new genus of pigeons evolved – large birds adapted
to feeding on leaves, buds and flowers of tropical trees. They usually walk
on large tree branches and lianas on strong legs. These birds are able to fly,
but do it reluctantly due to large size and partly reduced keel bone. These
features appeared due to the anatomy of digestive path of these birds – they
have large crop of two chambers and long intestines. It is a result of adaptation
to feeding on plant matter rich in cellulose. Since they have a tail capable
of being opened like those of peacocks of human era, their genus is named Pavoenas,
the peafowl pigeons. Their typical representative is blue-tailed peafowl pigeon,
the true gem of rainforest canopy.
Birds of this genus have clearly expressed sexual dimorphism. The females of
this species have short tail and are brownish green with blurred cross-striped
pattern on neck and back. Males are bright green with pink metallic shine on
neck, yellow feathers in their wings, and iridescent blue feathers in tail.
Males are about 84 cm long, including tail, and weight about 3 kg. Females are
smaller, about 70 cm long and weighting about 2.7 kg. It is the largest species
in its genus and inhabits the Amazon basin.
They are arboreal, but often search for fallen fruits, insects and small pebbles
on the ground. These birds can get water from bromeliads and find everything
they need in tree crowns. All peafowl pigeons are solitary birds, and only breeding
couples keep together. Male attract females with courtship dance with tail opened.
It chooses sunny places for the dance, where its tail sparkles and is visible
from far away. Dancing male inflates its crop and fluffs neck feathers. With
its wings opened it trembles with its whole body and is cooing loudly. Each
performance lasts for some minutes, and is longer when female appears nearby.
Attracting the female, male “bows”, raises widely opened tail and trembles in
sunlight.
After the mating female makes nest by itself in tree branches. Nest is simple
and is made of twigs and dry branches with litter of dry foliage and stalks
of grassy plants. Female lays only one or two eggs with opaque green shell and
hatches them for 32-33 days. Female makes three or four clutches in two years.
Male takes care to the incubating female, feeding her with regurgitated semi-digested
leaf pulp. Each male takes care to two or three females living on its territory.
When nestlings hatch, both parents feed them with semi-digested food and add
to it the “bird milk” characteristic to pigeons and rich in proteins. Nestlings
grow quickly, reach the size of adult bird and fully fledge in two months. They
leave nest and wander in forest canopy with female for two weeks, then pass
to independent life.
Young birds become sexually mature at the age of 1 year. Lifespan is about 10
years, but males usually fall prey of various predators at younger age.
Orange-tailed
peafowl pigeon (Pavoenas cirrhocauda), similar to the blue-tailed species,
lives in the coastal forests of southern part of South America. Male is bright
green with orange feathers in its tail, females are brownish green with cross-striped
feathers in wings. Tips of tail feathers at males have fringe of thin and elongated
feather barbs. It is slightly smaller: males are about 74 cm long and weight
only 2.5 kg. Females are 69 cm long and weight 1.9-2 kg. These birds eat lots
of tropical fruits, preferring figs and fruits of other cauliflorous trees.
The other species is the yellow-tailed
peafowl pigeon (Pavoenas flavicauda) – its tail feathers are yellow.
It lives in the forests of the northeast coast of South America. This species
differs from other peafowl pigeons in having a little mobile crest at males.
During the courtship male raises crest and cooes loudly to attract females.
At these moments its crop is inflated. Females are brownish green and have an
expressed cross-striped pattern on neck, body and wings, while the males have
the feathers of their back and wings bright green and those of their chest and
head yellow. Males are 78 cm long, but have longer tail and lighter weight compared
to other species. Females are 72 cm long. This species often flies down to the
ground and feeds on mushrooms and fallen fruits.
This bird was discovered by João Vitor Coutinho, Brazil.
Red-knobbed
pigeon (Nodoenas rubrus)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: tropical forests at the foothills of the Andes.
In South America the genus Patagioenas survived the environmental changes caused
by human activites and gave rise to two new genera: the genus Pavoenas, the
colorful peafowl pigeons, and the genus Nodoenas, with its only representative
being the red-knobbed pigeon.
Red-knobbed pigeon has physical characteristics very similar to those of its
ancestors; the only difference is a prominent and bright red knob above the
beak, present only in the males. This knob is responsible for naming the genus.
The overall appearance of these birds is typical for the family, with strong
legs allowing perching and even hanging head downwards on thin twigs in searches
of fruits. Beak is rather short, but the mouth cut is large and mouth may be
opened wide, allowing these birds swallowing of rather large fruits entirely.
Both the male and female are reddish brown, with the wings being dark gray.
They are small birds with only 35 cm both sexes. Wings have rounded shape fitting
to fast flight in forest canopy, and tail is fan-shaped. The sexual dimorphism
at these birds is expressed in presence at males of soft tissue knob made of
erectile tissues and able to fill with blood during the courtship games. Knob
is covered with featherless skin of red color. Females have reduced knobs on
their beaks.
Frugivores, they fly long distances in flocks of 10 to 16 individuals in search
of food. In times of great abundance of fruits, they form flocks of hundreds.
When the number of fruits decreases, the large flocks disperse.
The Red-knobbed pigeon program their breeding periods according to the availability
of food, in good years they can reproduce four times.
At the peak of the breeding season the male flies over the forest and is cooing,
then perches on tree branch and begins to display its knob while continues to
coo. The female chooses the males with the more colorful knob and the highest
coo. They are monogamous, living together for life.
The couple nests in territory marked by the male in high flights with a special
type of coo. They build the nests in trees using twigs from dry branches. The
nest is flat with loosely interlocking twigs. Female lays two white eggs, which
are incubated for 17-18 days. The couple takes care of nestlings, feeding them
with the “bird milk” characteristic to pigeons and rich in protein, which is
produced in crops in both sexes during that period. As the nestlings grow, small
fruits are added to their diet, semidigested first, and then fresh. They fledge
quickly, leaving the nest after one month and staying with the parents until
the two months age. Young birds reach sexual maturity in 1 year, when the males
show an increase in the size of their knobs that begin to acquire the characteristic
red color.
Their lifespan is of 9 years, with some young males being preyed during their
first mating season. Because of such kind of sexual disbalance some females
pair with “married” males and raise one nestling alone.
This bird was discovered by João Vitor Coutinho, Brazil.
Balkan
forest pigeon (Hesperocolumba balkanensis)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: subtropical forests of the Balkans.

Picture by Pavel Volkov
In Neocene, the Balkans have become one of the warmest and
most livable places in Europe. Peculiar natural conditions have been formed
here, favorable for the growth of evergreen drought-resistant forests characterized
by a peculiar flora and fauna. Balkan
cavedoves (Cavernicolumba strigops) nest in local caves – birds rather non-standard
in behavior with modest plumage color and a kind of face disk, which makes them
look like owls. However, the cavedove feeds on the fruits of plants, like its
closest relative, the Balkan forest pigeon.
In its general appearance, Balkan forest pigeon resembles ancestral pigeons,
differing only in a longer and thinner beak. It is a fairly small bird – about
20-22 cm long, with a wingspan of 30-35 cm, weighing no more than 200-250 grams.
The background color of the plumage is greenish with a brown spot on the shoulders
and back; only on the head sides there are feathers with a yellow tinge, but
around the eyes and up to the beak there are “glasses” of dark green feathers,
which gives the Balkan forest pigeon a distant resemblance to its cave-dwelling
relative. Males and females of this species have an identical coloration, but
the female’s plumage is duller than that of male.
Unlike the cavedove, this bird nests not in caves, but in the forest, in the
tree crowns. During the courtship display, males inflate the crop and utter
loud buzzing calls. A breeding pair is formed for several breeding seasons,
and outside of the breeding season, birds prefer to keep together.
The nest is a simple platform of twigs and branches, where the female lays 3-4
grayish or white eggs with dark specks. Both parents take turns hatching the
clutch, but the male often does it at night. Nestlings hatch on about the 20th
day, and spend another month in the nest. Then, for about another month, the
young birds are fed by their parents, and then they leave the young forever.
Nesting is repeated twice a season, young birds become capable of breeding the
next year. Balkan forest pigeon is an unsociable bird; these pigeons live in
forests in breeding pairs and do not form large flocks, as some other pigeon
birds do.
Like the cavedove, Balkan forest pigeon is a frugivorous bird, but it also eats
insects and other invertebrates, and feeds on seeds, buds and young leaves.
In summer, it keeps mainly in the crowns of trees, rarely descending to the
ground. In autumn and winter, birds often look for food on the ground, but they
are very cautious and take off at the slightest danger, loudly flapping their
wings. Their behavior is not particularly complex, and this pigeon is hunted
by local birds of prey, as well as tree snakes and predatory mammals (mainly
mustelids). Balkan forest pigeon lives no more than 10 years.
This bird species was discovered by Bhut, the forum member.
Booming
Fowl-Dove (Gouraphaps tardus)
Order: Doves (Columbiformes)
Family: Pigeons (Columbidae)
Habitat: Tropical rainforests of northern Meganesia.
Endemic species of Australian bird have generally prospered in the Neocene of
Meganesia, and the impact of introduced species is less than elsewhere.
The fowl-sized Papuan ground pigeons such as the genus Goura, became extinct
in the late Holocene because of man’s pressure from hunting and habitat loss.
A new kind has come to partly replace them, descended from the Wonga Pigeon
of Eastern Australia (Leucosarcia), which has spread much further north.
The Booming Fowl-Dove is a fairly large ground bird, reaching up to 75 centimetres
long and 3 kilograms in weight. The colour is a slaty blue, becoming more intense
powder blue on the chest and wings, with paired white bands extending across
the chest, and a whitish grey underside flecked with blue. The feet and flesh
around the eye are pinkish red, and the front of the face is whitish feathered.
The beak is a little longer than its ancestor, more similar to that of the genus
Goura.
These birds are gregarious and mostly diurnal, travelling in pairs or small
groups, they travel over the forest floor in search of food, which consists
of fallen fruit and seeds, occasionally insects. When alarmed, they will fly
and roost high in the branches of the trees until danger has passed. Males frequently
display to each other and the females in order to maintain dominance and make
overtures, this consists of puffing up the chest and making short charges and
beating of wings.
Breeding occurs late in the dry season, when calling and displaying to the female,
the male makes a loud humming boom sound and puffs up its chest. The female
will make a well built stick nest in an overhanging tree, laying a single large
white egg, which she broods exclusively without help from the male. Incubation
period is 30 days and the squabs leave the nest when they are still smaller
than their parents, but are attended to by the female for up to 11 weeks. Lifespan
is up to 25 years and predators include many large flesh-eating marsupials and
predatory birds, monitor lizards will eagerly eat their eggs.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Fruit
coorrow (Carpocolumba curro)
Order: Doves (Columbiformes)
Family: Fruit doves (Carpocolumbidae)
Habitat: tropical woods of Africa, Southern Asia, Indonesia and islands of Indian
Ocean.

Picture by Amplion
In Neocene borders of natural zones of Earth have started
to shift to poles: warming and humidifying of climate has resulted it. Tropical
rainforests which had practically missed during the ice age, have occupied extensive
territories again. But at the ice age the huge amount of species inhabited such
places, has died out, and Neocenic rainforests were occupied practically from
zero. A variety of some typically wood birds like parrots has appreciably decreased,
that has permitted to other groups of birds to master new habitats. Among settlers
of rainforests there were pigeons. Due to high adaptive ability some species
of these birds have survived and have replaced forest species of birds had dyed
out at the end of Holocene, including representatives of other groups.
Descendants of pigeons are large herbivorous birds coorrow (Carpocolumba). They
eat basically soft fruits and tree leaflets. These birds are rather large, and
are remarkable among the bird population with rather bright coloring.
Fruit coorrow is rather large bird (up to meter long including tail; its weight
is about 4 kg). It is the inhabitant of forest canopy, and appearance of this
bird indicates to its adaptation to this habitat. Wings are rather short and
rounded on ends, but tail is cuneate and long - such proportions provide maneuverable
flight among branches. Legs of bird are short, but tenacious, with bent claws.
Feathering is bright and contrast colored: wings and tail are green, stomach
is canary-yellow with black “tie” on chest, and head is rusty-red.
Beak is short, thick and grey-colored. At the basis of beak there is cere (site
of naked skin surrounding nostrils) characteristic for pigeons. The mouth cut
is wide, reaching vertical of back edge of an eye: it is the adaptation facilitating
feeding.
Fruit coorrow eats fruits of various tropical trees. Bird pecks large fruits
and swallows small ones entirely. In craw swallowed fruits are softened, and
then get to stomach. The stomach of fruit coorrow has some departments in which
symbiotic protozoans settle. Due to them speed of digestion at these birds appreciably
increases. Seeds of trees, covered with firm coat, are not digested in a stomach.
At some species of trees passage through intestines of frugivorous birds like
coorrow is necessary condition for germination: under action of gastric juice
of bird the seed coat becomes thinner and also more permeable. Thus, coorrow
takes the important part in seeds carrying and renewing of woods of tropical
zone.
In forest canopy it is often possible to hear a voice of these birds: characteristic
rolling coo (the name of this bird is onomatopoeic).
Fruit coorrows migrate in wood by small flocks of 6 - 10 birds. They practically
do not fly down to the ground finding all necessary for themselves at tops of
trees. Here it is possible to find even enough drinking water: in forest each
day there are rains, and water is accumulating in leaves of some trees. Birds
constantly move in wood, finding trees with ripe fruits. They are compelled
to do it, because fruits of different trees ripen in different time, and some
trees fructify irregularly. Inside each flight there are breeding pairs of birds
and single young growth.
Pairs at fruit coorrows form to all life. Male and female choose each other
by personal sympathy in flight of neighbours. Male cares for the female, cooing
and puffing craw. If the female shows reciprocity, male starts to feed her up
with semi-digested fruits. Thus the female as a sign of submission slightly
opens wings, representing nestling eliciting forage. Birds of one pair keep
together, clean each other’s feathers on head and neck. After pairing pair starts
to build nest. Coorrows nest in groups, at distance of several tens meters from
each other.
Art of nest building directly specifies relationship of these birds with pigeons.
Nest of coorrow simply represents big heap of dry branches thrown in forked
crown of large tree. In clutch there is only one large egg with greenish-white
shell. Male and female within 25 days hatch it alternately. During the year
pair of birds makes two clutches.
Nestling hatches blind, covered by thin down on head and back. At the second
day at him juvenile down starts to grow, and at week age it opens eyes. Similarly
to other pigeons parents feed up posterity by the “bird milk” – a product of
craw walls secretion.
Nestling grows in nest within 2 months, increasing for this time up to weight
about 3 kg. At young bird feathering is not as bright, as at adults: head is
dark-brown, and “tie” on stomach is not present. After nesting parents and brought
up young one join other pairs, forming flights, and migrate in forest.
Japanese
coorrow (Nipponocurro melanoleucus)
Order: Pigeons (Columbiformes)
Family: Fruit doves (Carpocolumbidae)
Habitat: Japan Islands; northern populations fly to Big Kurils, but migrate
to the south for wintering.
At the boundary of Holocene and Neocene tropical forests represented only small
islets in valleys of rivers within the areas of the former area of their growth.
It was a consequence of irrational economic activities of people, and also of
climatic changes. In such conditions the most part of typical forest groups
of animals fall into decline, and some of their groups died out almost completely.
At the boundary of epochs large species of parrots had become extinct because
of lack of the habitats, capable to support viable populations of such species.
In early Neocene, when the area of tropical forests began to increase, parrots
got competitors – forest species of pigeons. In tropics of Old World among pigeons
the separate family of fruit doves evolved – large birds resembling macaw parakeets
of Holocene epoch. These fruit-eating birds successfully compete to parrots
and consequently in Old World there are no large parrots and parakeets eating
soft vegetative food.
Typical representatives of fruit doves are coorrows, long-tailed birds of a
forest canopy remarkable in diverse colouring. They live mainly in tropics,
but one species also lives in warm-temperate zone of Northern hemisphere. It
is Japanese coorrow - a large bird having wingspan more than one meter and weighing
up to 3 kg. Tropical species of coorrows may be brightly colored, but this species
differs in contrast black-and-white colouring. Breast, back and tail of these
birds are black; on breast there is an expressed blue metallic shine. On wings
primarily and secondary feathers are white (sometimes with black spots in the
bottom part), and covert feathers of wings are black. White feathers also grow
on the bottom side of wings and on sides. The head of Japanese coorrow is white,
on it the red rings of featherless skin around of eyes are emphasized; it is
expressed at representatives of both genders. During a display ritual this skin
swells with blood and becomes clearly visible from apart. Male of this species
differs from the female in greater development of warts on skin around of eyes.
At some males the area of naked skin is stretched even to forehead, and during
the display their head gets absolutely grotesque shape – the warts swollen of
blood become very large. On bird’s forehead (at males above the area of skin
outgrowths) there is a thin mobile cop of several peaked white feathers.
Coorrows are forest birds very seldom flying down on the ground. Their body
shape is perfectly adapted to movement in forest canopy. Wings of Japanese coorrow
are rounded, and tail is very long, wedge-shaped and similar to tail of pheasant.
Due to such shape flight of coorrow is very maneuverable. In summer northern
populations of Japanese coorrow cross sea passages and reach up to Big Kurils,
where they nest.
Japanese coorrow eats soft tree fruits, occasionally adding to it young leaves
and food of animal origin – cockroaches, caterpillars and slugs. These birds
have short thick beak, a little bit similar to beak of finches. Jaws of bird
can be opened widely, and Japanese coorrow easily swallows even large fruits
(plum-sized ones) entirely. Coorrow eats fruits of treelike plants and lianas
of ginseng family, which are inedible for forest mammals. Seeds of these plants
do not lose viability, passing through intestines of bird.
Japanese coorrow lives in flocks numbering up to 50 adult birds. This species
of birds rather peacefully behaves relatively to the neighbours, but displays
aggression in relation to large species of collar
pigeons which are its food competitors. Coorrows attack on collar pigeons
and strike them impacts by wings and beak. At an opportunity Japanese coorrow
ravages nests of pigeons, pecking their eggs. But small collar pigeons can freely
nest even near to Japanese coorrow.
At Japanese coorrow breeding couple is formed for the whole following life.
Partners choose each other in flock of congeners and permanently keep together.
In courtship season male regularly renders signs of attention to the female.
It loudly coos, strongly inflating craw and demonstrating metallic shine of
feathers on it, carries twig or plant leaf in beak, walks round the female,
raising tail vertically and stirring by semi-opened wings. At the moment of
the greatest excitation at bird sites of naked skin around of eyes swell very
much.
Birds of this species nest in small colonies numbering up to ten nests. In clutch
there are 2 – 3 large eggs alternately hatching by both birds within 20 days.
Nesting happens once a year, and only in the south of an area birds have time
to rear two hatches within one year. Nestlings eat pulp of semidigested fruits
mixed with secretions of craw walls (it’s a characteristic feature of pigeons).
Young birds have monotonous grey plumage which is replaced with characteristic
black-and-white colouring only to six-monthly age.
Mergen
(Ungulavis trachypes)
Order: Sandgrouse (Pteroclidiformes)
Family: Sandgrouse (Pteroclididae)
Habitat: steppes and semi-deserts of Central Asia to the east of Fourseas.
In conditions the global ecological crisis sandgrouse suffered less than many
other bird species – these birds were adapted to extreme living conditions in
a desert area, so they were not harmed by the expansion of desert areas in the
human era. In addition, the ability to increase the number rapidly and to make
long-distance migrations and quickly change their place of residence when living
conditions deteriorate made it possible to preserve a sufficient number of viable
populations. Some of the local species became extinct and the number of other
species decreased, but they survived. A number of species already in the human
epoch were characterized by significant fluctuations in numbers, so after the
end of the human epoch, the surviving species simply realized their potential.
Their descendants evolved into new species, and one of them is megren, a descendant
of Pallas’s sandgrouse (Syrrhaptes paradoxus).
Compared with the ancestral species, mergen is a rather large bird: the body
length of an adult is 65-75 cm; weight is 3.5-4 kg. Male is larger than female,
but does not differ from it in coloration. The background color of the plumage
is gray with brown mottling, reddish-yellow on the belly. Wings are strong and
pointed; the flight is straight and fast. Birds are capable of making long flights
in search of water and food. The outer and middle toes of the bird, fused already
in the ancestor, formed a kind of hoof covered with a common corneous cover
– hence the name meaning “hoofed bird”. Such an adaptation allows it to walk
on hard and stony soil, as well as on stones that are strongly heated by the
sun. The inner toe is reduced.
Megren is unpretentious in nutrition: this bird eats herbaceous plants, including
dried ones, as well as leaves and berries. The digestive system of this species
does not digest hard seeds, and the bird carries them over long distances. From
spring to autumn, these birds keep alone (except for the time of rearing chicks),
in winter they form flocks of 10-15 birds of both sexes. At this time, mergen
follows the herds of harelopes and steppe obda,
feeding on grass excavated from under the snow.
The courtship season in this species lasts from the end of March to April. At
this time, the peculiar courtship games take place: males gather in certain
areas of the steppe with hard clay soil without vegetation and begin courtship
dances – they jump up, flapping their wings, and quickly stamp their feet. The
noise they make resembles dull thunderclaps, for which the bird was named after
the Mongolian god of thunderstorms. The feet “drumming” is very synchronous
and attracts other relatives very much. Each bird drums with its feet for about
ten seconds, making up to 3-5 beats with each foot per second. Each lekking
ground has its own “drumming” rhythm, which is set by the dominant males and
supported by the rest of the birds.
The female lays from 3 to 5 eggs in a hole lined with grass and incubates them
for 40 days under the protection of a male replacing her at night and morning.
Immediately after the hatch of offspring, the couple breaks up. The chicks hatch
quite developed and are immediately able to follow the mother and feed, pecking
the same plants as her. The mother only gives them a drink from the beak for
the first 20 days, until they learn to fly, and warms them at night. Later,
young birds stay with their mother for some time. At the age of 3 months, young
birds become completely independent, and by the next spring they reach sexual
maturity. Life expectancy reaches 10-12 years.
This bird species was discovered by Nick, the forum member.
Stripe-billed
tucanops (Tucanops dimorphus)
Order: Cuckoos (Cuculiformes)
Family: Cuckoos (Cuculidae)
Habitat: tropical forests of Northern Meganesia.

Picture by Pavel Volkov
In most cases, human activity led to a worsening of the situations
of species of wildlife. But some species have learned from this many benefits,
among them the large Channel-billed cuckoo (Scythrops novaehollandiae), adapted
to life in modified human habitats. After the disappearance of people its descendant
became an unusual bird, similar to a toucan: the stripe-billed tucanops that
inhabits tropical forests of northern Meganesia and around Arafura Lake and
Lake Carpentaria.
This species is one of the largest representatives of cuckoos: its wingspan
is up to 80 cm, and its weight is about 1 kg. The bird has elongated pointed
wings and a fan-shaped tail.
In the color and size of this bird species sexual dimorphism is clearly pronounced.
The male of this species is larger, bright and contrasting colored. The female
is smaller (adult weight of about 800 g) and much more modestly colored. The
plumage of the male is mostly black, the tips of primarily feathers are white,
the back is rusty-red. The area around the eyes of the male is covered with
white feathers. The plumage of the female is brownish with black ripples. A
characteristic feature of the appearance of these birds is a very large, deep,
laterally compressed beak filled with light spongy bone tissue. On the male’s
beak there is a horny ridge along the top side of the upper mandible. The coloring
of the beak of the male is lemon-yellow with a red base, the female’s is a dull
yellow. On the sides of the beak at the male and the female several slanted
strokes of black color stretch, contrasting with the background color of the
beak.
The stripe-billed tucanops is an exclusively carnivorous bird. This species
feeds on small mammals, birds, reptiles and large insects.
Like its ancestor, this species is a brood parasite. A pair forms for several
breeding seasons and controls a territory of 4 – 5 square kilometers at which
the nests of species feed the juveniles of this cuckoo are located. Stripe-billed
tucanops keep in the forest canopy, without going down to the ground. A population
of this species lives in mangrove forests separating the Arafura Lake from the
ocean. A pair of birds keeps contact with each other, calling to each other
in loud shrill voices.
For breeding this species uses the services of various species of birds of medium
and large size, including corvids. A pair of these birds lays in their nests
up to 20 eggs per season, laying one egg a day. Parental care does not end at
this: a pair of these birds controls the territory where the eggs are laid,
and expels from the territory of large birds of prey, which may threaten the
foster parents of the chicks of this species. Also, these birds can attack wood
mammals like cuscuses and rats, which would ravage nests. At the appearance
of these large predators the birds raise a collective alarm and harass them,
leading the bird flock combined of different species.
The nestlings of stripe-billed tucanopses develop very fast for such a large
bird and hatch on the 12th day of incubation. They immediately throw out of
the nest other eggs and chicks, remaining the sole object of parental care.
After 3 months, they are fully developed and fledged. Juvenile plumage of this
species is gray with black primarily feathers, identical for birds of both sexes.
The beak has a brown color, black smears on it are not yet visible. After 2
months of independent living the young bird sheds and acquires plumage characteristic
for adult birds. Sexual maturity comes at the age of 6 months. By this time
a young male grows a horny crest on the beak.
The idea about the existence of this species was proposed
by Tim Morris, Adelaide, Australia.
Translated by FanboyPhilosopher
Secretive
snake-catcher (Cryptophoenix ophiophagus)
Order: Cuckoos (Cuculiformes)
Family: Cuckoos (Cuculidae)
Habitat: tropical forests of Southeast Asia.
At the end of Holocene and the beginning of Neocene, birds of prey experienced
a significant crisis. Large species, whose numbers were undermined by man, gradually
became extinct. In some places of the Earth, they were replaced by descendants
of smaller species. But in Eurasia, the place of feathered predators was occupied
mainly by carnocorvids and
some shrikes. In the
tropics, the diversity of carnivorous birds is much higher, and among them there
are descendants of malkoha cuckoos (Phaenicophaeus). Having lost some species
during the destruction of tropical forests in the human epoch, the few surviving
species have evolved; one of these birds occupied a niche of a specialized herpetophage
in the forest canopy.
Secretive snake-catcher is a relatively large arboreal bird, resembling large
nightjars outwardly. The body length is about 70 cm, of which about half is
a narrow tail; male is slightly larger than female. The wingspan is 100 cm.
Wings are wide, with blunted tips, providing maneuverable flight among the trees.
The color of the plumage is greenish-gray; there are black and green strokes
on the wings. The bird has large yellow eyes surrounded by rings of gray featherless
skin, and a hooked beak with a wide mouth cut.
Secretive snake-catcher feeds mainly on lizards and snakes, regularly catches
amphibians, and occasionally eats large arthropods and rarely – rodents. Bird
lives in family couples, but both birds hunt alone. The border of the territory
of a family couple is not clearly determined, but competitors are not allowed
to the nest. This species flies to hunt later than others, and stops earlier
when the activity of its prey begins to decline. Secretive snake-catcher uses
an ambush method of hunting: the bird hides for a long time in the foliage or
clings to the tree bark or the bend of a liane, and the color of the plumage
hides it from both predators and prey. It’s not easy to spot a bird in an ambush.
When it sees the prey, it attacks, having previously let it get closer, or getting
closer on branches and a tree. The bird crushes the snake’s head with a bite,
and pecks other prey, simply killing it with an exact peck.
The courtship season begins at the end of the rainseason. At this time, single
males call females with monotonous hollow whistling calls. Family pairs vocalize
a duet. From time to time, the male brings the female his prey – usually a snake
or a large lizard. In single birds, it is an obligatory element of courtship.
The nest is small and loose, the head and tail of the hatching bird are often
visible from the outside. Birds often occupy other bird’s nests, sometimes expelling
the nest owners and ruining their clutch. There are 2-3 eggs in the clutch.
Sometimes a bird that is late with nesting manages to lay its eggs to neighbors
who usually accept another bird’s egg. After incubation lasting 16-17 days,
naked and blind nestlings hatch from the eggs. A month later, the young birds
fully fledge and try to fly, but they remain near the parent nest for some time.
They are duller than their parents, and spend a lot of time learning to hunt,
but for about another month their parents feed them. At the age of six to seven
months, they get adult plumage, at 2 years of age they reach sexual maturity.
Life expectancy is up to 20 years.
This bird speies was discovered by Nick, the forum member.
Saline
land nightjar (Halocaprimulgus halophilus)
Order: Nightjars (Caprimulgiformes)
Family: True nightjars (Caprimulgidae)
Habitat: “oases” in salt swamps of Mediterranean.

Picture by Alexander Smyslov
In Neocene Africa had continued movement in the direction
of Europe, and because of it strait of Gibraltar had closed for ever. The ice
age which had coincided in time with this event, differed in dryness of climate
- the huge quantity of water appeared frozen in glacial caps. By virtue of these
circumstances Mediterranean Sea began to dry up catastrophically quickly, and
soon it had completely disappeared, having left after itself an extensive fruitless
hollow where the ground was covered with layer of salt. In Neocene the climate
became much more favorable, and these changes had touched even salt desert of
Mediterranean. At the place of former desert the circuit of lakes and swamps
with water of very high salinity many times over exceeding oceanic one had appeared.
Heat and absence of potable water are circumstances because of which only few
animals can survive here. Salt lakes block access to “oases” – heights among
desert where in ground there is no salt, and plants can grow there. In such
places only those ones, which are able to overcome salt desert, can live. And
one of such species is the saline land nightjar.
This pigeon-sized bird is similar by constitution to the ordinary nightjar.
The feathering of saline land nightjar is color white with slight greenish shade.
Such colouring helps bird to mask among salt plain – bird simply nestles up
to the ground, and becomes imperceptible on the background of saline soil and
dried films of microscopic algae. On feathers tiny brown speckles are scattered.
Eyes of bird are black and less by size than at common nightjar – this species
hunts in the afternoon when above salt swamps flies,
which larvae live in salt water, hover. Paws of this nightjar are small, with
“brushes” of rigid feathers which raise a bird above sharp crystals of salt
and preserve legs of bird against cuts. At this nightjar there are long narrow
wings similar to wings of swift. The male differs from the female by two “pendants”
– the lengthened middle feathers in tail.
Nostrils of bird are expanded and extended in tubules, assisting to remove from
the organism surplus of salt (as at birds of order Procellariiformes). Various
birds differ in various strategies in the decision of one problem – disposals
of salt surplus being received with food. At saline land nightjar such large
salt gland, as at its neighbour, the crested
flamingo, is not present. The saline land nightjar drinks seldom, receiving
a necessary moisture from the had eaten insects. Besides this bird presumes
to drink from fresh springs in mountains, to which it quite often flies to slake.
Near streams the bird behaves cautiously: it is too noticeable because of white
feathering on the background of dark ground and vegetation of “oasis”.
This bird eats blood-sucking flies hovering about the flamingo, robberflies,
and also winged males of water
turtlebeetles. For this reason the nightjar accompanies with flights of
wingless flamingoes, or single Antheos
tortoises near which the “laid table” – set of large two-winged flies –
is ready for it. Thus, the saline land nightjar replaces in “oases” many species
of birds: fly-catchers, swifts and swallows which could not appear in swamps
of Mediterranean because of various reasons.
Saline land nightjar nests on the ground, in thickets of berry
salttrees and other bushes, at any time of year. Only in winter time when
from the north cool winds blow, and in short season of rains the bird does not
nest. Male involves female to the place of nesting, uttering long chirring warble.
It displays mainly in the morning and in the evening when it is enough light,
but it is cool. The laying is hatched by the male and the female serially, till
approximately 20 days. Nestling hatches covered with rich down of white color:
it is necessary for protection from overheat. It stays in nest about six weeks.
The bird sitting in nest closes nestlings by wings from scorching sunlight.
Parents bring up posterity with insects, and even bring to nestlings fresh water
in stomach.
One of the biggest dangers to inhabitants of Mediterranean “oases” is summer
storm. At this time wind lifts salty dust in air, and carries it, as if a blizzard.
During storms these birds hide in bushes, but hatching birds do not abandon
nests, and sometimes can even perish, filled up with salt dust. At saline soils
sometimes it is possible to find mummies of such birds stifled in salt dust.
This species of birds was discovered by Simon, the forum member.
Flagwing
(Vexillipteryx cantabrarius)
Order: Nightjars (Caprimulgiformes)
Family: True nightjars (Caprimulgidae)
Habitat: savannas and semi-deserts of South Africa.
Nightjars, although they lost part of the species diversity in human era, relatively
safely survived to Neocene. Tropical forest species suffered the most from human
activity, and species from savannas, steppes and deserts survived relatively
successfully. Nightjars had no serious competitors in their ecological niche,
so they have changed little by Neocene. Although specialized forms quite unusual
for this group, such as merlette (Merlette aviphaga)
and saline land nightjar (Halocaprimulgus
halophilus), appeared among them, most nightjars remained nocturnal insect
hunters, like most species known in human era. Among them, flagwing stands out
for its unusual appearance – it is a strange descendant of the pennant-winged
nightjar (Macrodipteryx vexillarius).
It is a relatively large bird compared to most nightjars: body length 32-34
cm, male body weight 303-365 g, female body weight 286-290 g, wingspan 63-70
cm. The background body color is grayish-brown, reddish on the back. Wings have
a pattern of small brown spots on a gray background. This bird species differs
from the rest of the nightjars by some features of the structure of the plumage,
which give it a very recognizable appearance. The secondary feathers of males
outside the breeding season are somewhat elongated, which adds a specific easily
recognizable shape the silhouette of a flying bird: wings seem angular, expanded
at the ends. In females, it is expressed to a lesser extent.
Birds of this species have large eyes, short beak, but a wide mouth, typical
for nightjars. Like other nightjars, flagwing is a nocturnal bird and sleeps
during the day – most often in bushes or on tree branches, lying along a branch.
At night it flies to hunt for grasshoppers, horseflies, gadflies, beetles and
moths. These birds are most often found near the herds of large herbivorous
mammals. The voice of this species is a long, rattling cry.
The courtship season at flagwings is in spring, in September-October. At this
time, among the secondary feathers of males, one pair turns into large ribbon-shaped
rusty-red feathers resembling flags. At these feathers, the shaft is soft and
flexible, and at the tips these feathers expand into rounded lobes. The color
of these lobes contrasts with the background coloring of the bird’s body: they
are covered with a cross-striped black-and-white pattern and end with a white
“eye” spot on the black background. At this time, they greatly hinder the movement
of the bird: they prevent its walking and prevent it from taking off normally.
Because of this, a fairly large number of males die when attacked by predators
– this bird species has appeared in the “trap” of sexual selection. This species
is polygamous, no permanent pairs are formed. During the breeding season, the
male mates with a large number of females, so the death of males is compensated
by a large number of offspring from the survivors. After intensive courtship
display for 1-2 weeks, the feathers of the courtship dress shed and the surviving
males continue to lead a habitual lifestyle. Courtship display in this species
includes a fluttering flight of the male low above the ground, accompanied by
a prolonged chirping trill. Females stay on the ground, from time to time flying
from one place to another, and indicate their presence by calls. During the
courtship flight of the male, they accompany him on the ground, flitting from
one place to another. Mating takes place on the ground, at this time the male
manages to mate with 3-4 females, after which he continues his flight. By the
end of the mating season, the male loses a lot of weight and weakens.
The nest is arranged on the ground and represents a hole in the grass or in
a bush. The clutch usually contains 2 eggs of an elongated ellipsoid shape,
measuring 27-37x20-25 mm. Incubation lasts about 17-18 days, exclusively the
female is incubating. 25 days after hatching, the chicks are ready to leave
the nest and live independently. At the age of 1 year, they are already sexually
mature. Life expectancy reaches 14 years, but males usually live much less because
of their vulnerability during the mating season.
This bird species was discovered by Nick, the forum member.
Hawk
potoo (Teratopotoo venator)
Order: Nightjars (Caprimulgiformes)
Family: Potoos (Nyctibiidae)
Habitat: South America, rainforests.

Picture by Amplion
In Neocene some nightjars passed to predatory habit of life
and feeding on vertebrates. Huge nightjar merlette (Merlette aviphaga) is found
in steppes of Europe – it is the largest (eagle-sized) species of the order
for all time of its existence. The second-largest species of nightjars lives
in rainforests of South America – it is hawk potoo, huge predatory nightjar.
Wingspan of this bird is about two meters, and weight is up to three kgs.
This bird leads passive way of life – it does not chase prey, but attacks it
by sharp throw when it will move closer to predator. In rest this bird skilfully
masks, perching on bark vertically. At hawk potoo there are short trunk and
rather large flattened head. The beak of this bird is short and hook-like, and
mouth cut is very wide – it stretches far for the level of back edge of eye.
The bird has partly binocular sight – its eyes are shifted forward, and their
fields of vision substantially overlap each other. The iris of eye is colored
camouflage pattern – it is brown with black nerves. Therefore bird can observe
of environmental conditions, not giving out itself.
Plumage of hawk potoo skilfully imitates coloring of bark – it has greyish-brown
color with black longitudinal strokes and randomly scattered black spots. Feathers
of bird are soft; therefore flight of this predator is silent like at owl. Wide
wings have rounded tips. Bird is not able to fly long, but is able to make fast
and unexpected throws and to change direction of flight. Tail of hawk potoo
is wide and fanlike; with its help bird makes sharp turns in air.
All nightjars have short paws and badly move on flat ground. Hawk potoo is not
exception here. It does not seize prey by paws as birds of prey of Holocene,
but catches it by widely open mouth. Its tenacious paws serve basically to perch
on bark in ambush, or to creep clumsily on tree trunk. This bird is not able
to move downwards the tree trunk, and can climb only upwards or sideways. Usually
hawk potoo chooses for an ambush the place near thickets of epiphytic plants
where it is less appreciable. The hidden bird densely nestles against tree bark
and slightly opens wings to not give out its location by shadow. As against
the majority of nightjars, hawk potoo is diurnal bird.
Hawk potoo almost does not catch insects – it attacks only largest beetles and
moths. The basis of diet of this bird is made of vertebrates. When any careless
bird or bat flies too close, hawk potoo pushes from trunk, silently takes wing,
and seizes it right in air. It kills prey by pointed tip of beak and comes back
to ambush. Here bird swallows prey entirely. Also hawk potoo seizes from tree
trunk various climbing reptiles and frogs, flying right above them. This bird
is able to swallow entirely pigeon-sized prey.
This bird is solitary species. Nesting at hawk potoo proceeds once a year, and
only at this time birds meet in pairs. The pair is formed at the territory of
the female, and female dominates in family. It is larger and more aggressive,
rather than male, therefore in courtship season the male should be very cautious.
If he will hasten with court, female can attack him and put to him serious wounds.
Male shows to the female intention to create a pair, uttering continuous bitter
trill. In the beginning he cautiously comes nearer to the female and “sings”,
in order that female has got accustomed to his presence. If the female answers
him by short trills, male moves nearer to her behind and sideways, keeping as
it is possible further from her beak. At this time female keeps eye on male,
ready to fly up and to attack him from air in any second. If the female is ready
to pairing, she admits male to herself and allows him to clean her plumage.
One more element in court of these birds is feeding of female on fresh prey.
Male holds prey in beak, and the female eats it, tearing pieces off from it.
The nest of these birds is arranged in small pit on thick inclined branch. The
only egg with spotty grey shell is hatched alternately by both parents, cautiously
replacing each other on nest. The hatching lasts 45 days. The nestling is covered
with rich down of grey color. It seizes bark by well advanced claws, and spends
the most part of time in immovability, trying to not draw attention of predators.
Parents feed it on small birds and frogs, and it swallows food entirely. Development
of nestling lasts till about three months.
Great
false owl (Pseudostrix grandis)
Order: Nightjars (Caprimulgiformes)
Family: Owlet-nightjars (Aegothelidae)
Habitat: Borneo island, tropical forests.
The human actions during the Holocene caused the extinction of many creatures.
However some were capable of surviving and in the Neocene they gave rise to
new species. It was the case of the genus Pseudostrix, the false owls. These
birds evolved from the owlet-nightjar (family Aegothelidae). The type species
of this Genus is the Great false owl, which lives in the forests of Borneo.
The great false owl is the largest member of its genus, with 49 cm of length
and a wingspan of 54 cm. It’s morphologically similar to a long tailed owl,
with feathers forming a facial disk and large eyes. The facial disk collects
sound waves and directs those waves towards the false owl's ears. The feathers
in the eyebrows form tuffs in the end, in a similar way to the ear tufts of
some owls. The back of these birds is dark grey with buff vermiculations and
the underside is pale with dark barring. The face is greyish brown with some
dark feathers forming the facial disk. The dark grey beak is curved and the
mouth is wide. The legs are pale grey and the feet have long claws.
These nocturnal birds feed on large insects, birds, mammals and reptiles. They
will stay on a branch of a tree waiting for the prey, when the prey approaches
they swoop down and swallow the prey whole. The false owls are great predators
of bats, actively pursuing these mammals. Once the prey is captured, they use
their strong beaks to crush it and then proceed to swallow it whole. Sometimes
it happens while they are flying, but they will wait in the entrance of caves
to surprise the bats. Small passerines are captured while they rest and small
nightjars are also eaten.
Due to their nocturnal habits, these birds rest during the day in cavities or
next to tree trunks. Great false owls live solitary or in pairs. They breed
year round. The nest is made in holes in trees where the female lays 3 to 4
white eggs which are incubated solely by her for 33 days. During this time,
the male will provide food to the female. The young fledge with 31 days after
hatching, but stay close to the parents for more than 4 months. The Meganesian
false owl reaches sexual maturity with 3 years and has a lifespan of 17 years.
Other species in the genus Pseudostrix:
Sunda
false-owl (Pseudostrix sundaicus)
Living in the forests of Sunda Land, this species have 43 cm of length and a
wingspan of 50 cm. This bird has greyish-brown upperparts with dark streaking
and the underparts are greyish-brown, with brown barring and buff spots. The
face is brown with light brown feathers forming the facial disk.
Black
false owl (Pseudostrix nigris)
Living in the forests and woods of Meganesia, this species has 44 cm of length
and a wingspan of 51 cm. This bird is blackish all over and densely striated
with horizontal, wavy, white bars. A black face mask encircles its eyes and
the facial disk is grey.
Lesser
false owl (Pseudostrix minor)
Living in the montane woods of Tasmania, this species has 37 cm of length and
a wingspan of 48 cm. Being the smallest member of it genus, the lesser false
owl eats mostly insects and small reptiles. Its plumage is greyish-brown with
a dark brown face. The facial disk is formed by grey feathers.
This bird species was discovered by João Vitor Coutinho, Brazil.
Spitting
swift (Viscapus viscosus)
Order: Apodiformes (Apodiformes)
Family: Swifts (Apodidae)
Habitat: tropical forests of Southern and South-Eastern Asia.

Picture by Lambert
Swifts are remarkable among birds in considerable development
of salivary glands. Their secretion are used by these birds for various purposes
– some birds “pack” caught insects into saliva, forming a capsule convenient
for feeding of nestling, and others in addition use saliva as glue, building
a nest. The swift Collocalia salangana in human epoch built nests exclusively
of saliva.
Swifts are specialized to life in flight, and can even sleep on wing. The source
of their food – small flying insects – during the whole human epoch differed
in stability, therefore even in epoch of global ecological crisis the significant
amount of species of swifts had survived, and in Neocene these birds still furrow
the sky in large flocks, living from temperate latitudes of both hemispheres
up to equator. During the evolution at them original vital strategy had developed.
Being dexterous flyers, swifts are completely helpless on the ground, and even
are not able to fly up independently from plain surface. Their nestlings also
are very vulnerable. But the species of swifts which has developed the original
adaptation for protection – spitting swift – has settled in tropics of Asia.
This bird uses special sticky and quickly congealing saliva for self-defense.
Adults spit predator with it, flying above it, and nestlings spit to nest ravager
similarly to nestlings of fulmar. Defending from the enemy, nestlings aim to
its eyes, and adult birds splash to climbing predator’s muzzle, and to wings
of birds of prey. Saliva rich in proteins secreting in one pair of hypertrophied
salivary glands, stiffens till few seconds as viscous gum-like strings on air.
It guaranteedly stops a predator.
Appearance of spitting swift is quite standard for these birds – it has small
size (body length is about 10 cm at wingspan up to 25 cm), short tenacious paws
and large head with short beak and wide mouth. Two hypertrophied salivary glands
stretch under skin of this bird up to a breast, and opens in mouth as two short
tubes under tongue. Glands have expansion as a sac, in which liquid contents
accumulate. Without contact to air it represents transparent liquid, a little
more viscous, rather than water. At oxidation its transformation to gum-like
mass proceeds. By contraction of ring muscles surrounding these sacs, bird spits
liquid as two jets to the distance of up to one meter.
These birds nest on trees, preferring the extensive hollows rotten out to several
meters down. In such hollows birds nest in colonies numbering up to two hundreds
of birds. Sometimes suitable hollow appears occupied – more often by bats. But
these swifts, when they prevail in number, can banish bats to other place, splashing
them with saliva.
The nest of spitting swift represents small “pocket” stuck of feathers and fluffy
seeds with the help of “fight” saliva. In clutch there is only one egg, but
during one year birds can repeat nesting up to four times. The main enemies
of nestlings of these birds are snakes and bats, but due to collective defense
birds in many cases succeed to repel an attack.
Young birds start nesting at the age of 10 months.
This species of birds was discovered by Simon, the forum member.
Tube-making
swift (Glutinornis tubicolus)
Order: Apodiformes (Apodiformes)
Family: Swifts (Apodidae)
Habitat: tropical forests of Africa, areas of young forest.
The human era has significantly influenced the diversity of living organisms.
The destruction of tropical forests led to the mass extinction of diverse forest
flora and fauna, and after the human disappearance, forests began to recover
from very few surviving fragments. The plants and animals inhabiting them began
to evolve rapidly, occupying vacant ecological niches and redistributing species
diversity between old and new inhabitants of the recovering tropical forests
of the early Neocene.
Swifts fly over the canopy of the forest – they have changed little in appearance
since the human era. In Asia, unusual birds appeared among the swifts – basket-mouths,
distinguished by a particular hunting device. African swifts are more conservative
in terms of morphology, but some of their species have interesting behavioral
features. Among them, sticky swifts have interesting nesting features – these
are birds with developed salivary glands that secrete a large amount of viscous
and rapidly solidifying saliva.
A characteristic representative of sticky swifts is a tube-making swift, a small,
modestly colored bird. The span of narrow long wings of an adult does not exceed
40-45 cm; the length of the body and tail is no more than 15 cm. The plumage
of this bird is colored gray with a dark V-shaped stripe on the upper part of
the body, stretching from the shoulders to the middle of the back. The underside
of the body is only slightly lighter than the top one; tips of wings and tips
of the forked tail are dark gray.
This bird has a short beak and a wide slit of the mouth that stretches beyond
the vertical of the posterior edge of an eye. Eyes are large and almost black.
These birds are characterized by highly developed salivary glands. Saliva allows
the bird to “pack” captured insects for feeding offspring, as well as build
nests.
Tube-making swifts nest inside large tree leaves, twisting them into tubes,
inside which birds glue a small nest and hatch offspring from an egg also glued
to this nest. Due to this peculiarity of choosing a place for the nest, tube-making
swifts prefer nesting in damaged forest areas, usually along the beds of large
rivers. They choose fast-growing trees with wide non-dissected simple leaves
and occupy branches in the upper part of the crown, under which there is enough
free space.
The breeding season of this species is not pronounced. Courtship flights continue
all year round and are accompanied by loud screeching calls. The choice of a
partner is accompanied with a joint synchronous flight, mating also takes place
in the air.
Male and female build a nest together. Choosing a leaf of a tree, one of the
birds twists it and holds it with its claws, while the second one at this time
smears with sticky saliva and glues its edges. The tube from the leaf is twisted
tightly enough – there is only room inside it for a nest and a pair of birds.
Birds cannot turn around inside the nest, so they leave it by simply unclenching
their claws – they fall down from inside the nest through the entrance and open
their wings already in the air. A small nest of down and flluffy seeds of trees
is arranged inside the leaf. There is only one egg with a white shell in the
clutch, which these birds glue to the bottom of the nest and incubate in an
upright position. Incubation lasts 12-13 days; the chick grows and develops
for about 20 more days. Sexual maturity of a young bird occurs at the age of
4 months, life expectancy does not exceed 5 years.
A leaf shelter has a rather long life: it is used and repaired by several pairs
of birds one after another until the leaf from which the shelter is twisted
dies off. Sometimes two pairs of birds use the same shelter, breeding their
offspring alternately. In this case, they jointly protect their shelter from
the claims of other relatives, although they tolerate each other. Nesting of
one pair can repeat up to 3-4 times a year.
A close species, lesser
sticky swift (Glutinornis hemisocialis) lives along the banks of narrow
forest rivers and prefers to catch insects among forest crowns. This is a miniature
species with a wingspan of no more than 25 cm. The plumage is light gray with
a bluish tinge; the tips of the edge tail feathers are white. Unlike the previous
species, it nests collectively. Two or three pairs together twist a large leaf
of the plant and arrange nests inside the tube on its opposite sides.
Pastry
swift (Glutinornis pistor), by its behavior, represents a more primitive
stage of development of this method of nest construction. The nesting pair folds
the leaf in half at the base and glues its edges to half, which makes the leaf
look a little like a pie. Birds strengthen the glue join from the inside with
an additional layer of saliva with down and feathers, and the place of gluing
becomes the foundation for the pocket-shaped nest. The entrance to the nest
is the open lower part of the glued leaf. The wingspan of this species is up
to 35 cm. Its plumage is dark gray with white “eyebrows” above the eyes. This
species prefers to settle in thickets of large-leaved grasses in the early stages
of restoration of the rainforest stand; it lives in Central Africa.
Chinese
swamp basket-mouth (Corbistoma paludiphila)
Order: Apodiformes (Apodiformes)
Family: Basket-mouths (Calathostomornitidae)
Habitat: China, swamp and lake areas.
In Neocene productive ecosystems giving the wide spectrum of forages to species
inhabiting them had received the wide distribution. It had caused occurrence
in Neocene of various groups of animals strictly specialized relatively to food.
Among birds the separate family of swifts strongly improved the way of prey
catching evolved. They differ in interesting shape of feathers surrounding mouth.
Elastic feathers with reduced vanes and small number of barbs expand trapping
surface, forming similarity of net for tiny flying insects. Trap feathers are
mobile: while the bird does not eat, they lie down on head and throat of bird,
not preventing flight. But during hunting they are widely opened, literally
filtering tiny flying insects from air. These birds are named basket-mouths,
and their scientific name means “bird with basket-like mouth”. Obviously, they
had evolved in tropical areas of Eastern Asia as they have kept attachment to
them.
Swamps of China are hundreds square kilometers of inaccessible terrain, the
circuit of shallow reservoirs, rivers and small sites of forests. The abundance
of moisture and set of shelters promotes prosperity of small two-winged insects
– midges and mosquitoes. Their larvae find food and shelter in water vegetation,
and adult individuals furiously attack mammals and birds inhabiting these areas.
But clouds of midges and mosquitoes involve birds specialized in feeding on
them. Through flights of insects small black birds sweep, and in sun beams ruby-red
spots on throats of some of them sparkle. These are tireless fighters of blood-sucking
insects, Chinese swamp basket-mouths. These fine long-winged birds form large
flocks, and nest in colonies. They are close relatives of Himalayan wiskered
lustrer, but only a little bit more smaller: their length of body is about 8
cm. Chinese swamp basket-mouth is similar to it externally, but in its anatomy
there is no specialization to extreme mountain conditions. Its feathering does
not reflect ultra-violet light, and haw does not protect eye from it. Colouring
of Chinese swamp basket-mouth is coal-black. At males throat is covered with
shining ruby-red feathers sparkling in sunlight, at females throat is lack of
metal shine. This color spot is a distinctive attribute by which birds distinguish
congeners from close species.
Beak of Chinese swamp basket-mouth is very short, but mouth cut is big. Jaws
of this bird can be opened at the large corner. This bird is specialized to
catching of small insects, due to what it avoids competition to larger birds
catching larger flying insects. Basket-mouths, as against Himalayan wiskered
lustrer, have one primitive feature – top feathers surrounding the mouth have
small parts of normal vane on tips.
Basket-mouths eat insects – forage which availability varies due to seasons.
Therefore they are birds of passage, even living in subtropical latitudes. Chinese
swamp basket-mouth migrates only a little – its flights make only several hundreds
kilometers. Close species living to the north from it, make considerably longer
flights.
Approximately from middle of spring, when mosquitoes start to swarm above swamps
of China, courtship rituals of Chinese swamp basket-mouths begin. These birds
as if compete in skill of flight, promptly flying by whole flocks and synchronously
turning in air. Flocks of basket-mouths first gather to dense sphere, then stretch
to long wide veil, or scatter to separate birds, and then gather again, as if
the invisible conductor operates them from the side. Courtship flight takes
place in the morning and in the evening before the sunset. At this time males
show the special impressively looking form of courtship flight: bird in prompt
flight is turned around of longitudinal axis, and its throat “flashes”, sparkling
in sun beams.
A significant part of adult life of birds passes in flight – they eat, drink,
pair, and even can sleep in flight. During the sleep at bird one cerebral hemisphere
falls asleep for some seconds. Then it wakes up, and bird is awake for some
time; and then the second hemisphere falls asleep. But sleep in flight is most
probably compelled circumstance: it is expressed during the migration. Out of
migration basket-mouths gather to spending the night in shelters inaccessible
to ground predators – more often in rotted through tree trunks. There birds
cling by claws to wall and sleep similarly to other birds. During the dream
the body temperature of these tiny birds lowers considerably – this feature
allows to save the energy.
Nesting of Chinese swamp basket-mouth occurs in colonies. For this purpose birds
choose trunks of high trees. The nest of this bird represents small cup of down
and flying seeds of plants, pasted to bark of tree by viscous saliva of birds.
The female pastes sole egg to internal part of nest and hatches it, having seized
by short paws to lateral parts of nest. Development at these birds occurs very
quickly. Nestling hatches at 12-th day of incubating; it is naked and blind,
but as soon as it leaves egg shell, it seizes nest by claws at once. It is fed
by both parents. Approximately from the third – fourth day of life nestling
starts to grow roughly. From now it eats per day approximately as much food,
as much it weighs itself. At the same time it starts to fledge. Trapping feathers
start to grow the first. They appear on edges of mouth at still naked nestling,
and stay in curtailed condition for a long time. Approximately to the end of
second week of life the nestling becomes almost completely fledged, and only
at this time its trapping feathers are unwrapped. At the third week of life
nestling is completely advanced. Approximately at monthly age it leaves nest,
and begins independent life. The young bird differs from parents in absence
of red spot on breast, which appears only at the approaching of sexual maturity.
Life expectancy of Chinese swamp basket-mouths is short – no more than three
years. But for one nesting season birds have time to hatch one by one four nestlings,
and one more generation appears in wintering places. Young birds also develop
quickly: they reach maturity at three-monthly age, and the first generation
of young growth has time to make an own hatch before flying away to wintering
places.
Chinese swamp basket-mouths are the smallest birds in family. Except for them,
in Asia there are some more species of basket-mouths:
Siberian
basket-mouth (Corbistoma migratoria) is larger bird (length of body
is up to 15 cm). Its feathering is colored not as contrastly, as at Chinese
swamp basket-mouth. This bird has almost uniform grey colouring, and throat
at males is red without metal shine. Throat of female is white. This species
is the bird of passage. It spends summer in Siberia, where in warm season masses
of two-winged flies appear, providing plentiful forage to these birds. In summer
it is possible to meet Siberian basket-mouths in huge territory – from east
spurs of Ural up to Kamchatka. During the migration Siberian basket-mouth overcomes
mountain ridges of Central Asia. In summer it is possible to see it often above
herds of large mammals of Siberia – obda
and shurga. In late summer,
shortly before first light frosts, the amount of flying insects is reduced,
and it serves as a signal for the beginning of winter migration. Literally for
one – two days all birds of this species gather in flights, and fly out to the
south, to South-Eastern Asia. In tropics Siberian basket-mouths keep in mountain
areas overgrown with forest. This bird nests on rocks overgrown with epiphytic
plants. In wintering places Siberian basket-mouth hatches one generation of
nestlings, and at the beginning of summer in Siberia it migrates to the north
again. At this species in clutch there is up to three eggs; development of nestlings
takes about two months.
Far
East (Japanese) basket-mouth (Corbistoma nipponica) differs from congeners
in brighter colouring. Feathering of this bird is black with dark blue metal
shine from above; stomach, sides and the bottom part of wings are white. At
males of this species on breast there is bright red spot. During courtship flight
the flock of males synchronously hangs in air till some seconds highly above
forest canopy. Males show to females feathering on breast, and then simultaneously
rush down and make an arch right above tree crones.
This species of basket-mouths spends warm season in woods of Far East, Japan
and Big Kurils. In hottest years single individuals of Far East basket-mouth
meet even at Kamchatka. Usually birds have time to hatch two generations of
nestlings for one year - one hatch in summer at the north, and another one in
winter at the south in wintering places. In its clutch there are two eggs; in
very favorable and rich in food years it may be three ones. This bird hibernates
in South-Eastern Asia – at Sunda Island and Jakarta Coast peninsula.
Nocturnal
basket-mouth (Macrocorbistoma nocturna) lives in Indochina and at the
Jakarta Coast. It differs in secretive habit of life and large size – the length
of this bird reaches 30 cm at wingspan from above 60 cm. Feathering of this
bird is light grey with cross strokes and dabs. This species is active in twilight
and at night, and has keen night sight. Eyes of this bird “shine” in darkness
like eyes of predators. Nocturnal basket-mouth lives in tropical forests and
eats large insects – moths, beetles and winged individuals of ants and termites.
For catching of such prey trapping feathers had changed – they became elastic
and similar to trap feathers of Himalayan wiskered lustrer. Only on tips of
two top feathers small parts of vane is kept. Barbs of these feathers are thin,
but very strong – even the beetle clashed in bird, will be knocked by them and
will get in mouth of winged hunter.
Nocturnal basket-mouths form very small congestions – no more than two tens
birds. They spend day in tree-trunk hollows, perching vertically and having
caught by short paws for wall of hollow. Seasonal prevalence in nesting is not
expressed at them. The same nest may be renewed and used serially by different
breeding pairs of the same colony.
Motley
basket-mouth (Xiao asiaticus)
Order: Apodiformes (Apodiformes)
Family: Basket-mouths (Calathostomornitidae)
Habitat: Far East, subtropical and temperate deciduous forests.
In Neocene, the species diversity of insect-eating birds increased, which is
associated with the restoration of forests after the human era. The heavens
of the Earth were inhabited by new families of birds, among which basket-mouths
– one of the most unusual descendants of the swifts of the human era – are remarkable
in their characteristic appearance. Rings of mobile elastic feathers with sparse
barbs grow around their beaks, which help the basketmouths deflect insects on
the fly and direct prey right into their mouths.
Most basket-mouths are small birds, and motley basket-mouth is one of the large
species of the family. It reaches 20 cm in length, has a wingspan of up to 55
cm, and weighs up to 50 grams. Compared to other members of its family, it is
distinguished by a more massive constitution with proportionally longer wings.
Its plumage is brown from above and gray or brownish from below, covered with
white spots and strokes forming a camouflage pattern imitating tree bark. There
are also white rings around the eyes. The beak is light colored, gray or pinkish,
with a very wide mouth slit.
Motley basket-mouth hunts, like its related species, catching insects on the
fly with an open beak and deflecting them into the mouth with hunting feathers.
But it is a larger bird that eats larger insects, and therefore sometimes it
attacks from ambush: it perches on the tree trunk or on the underside of a branch
and waits until a large insect flies past it. Attacking its prey, the basket-mouth
quickly flies off its branch, catches and swallows its prey.
This lifestyle has led to the appearing of some other differences in motley
basket-mouth from related species – it is not such a hardy flyer and prefers
to sleep perching on a tree, although it can also sleep on the fly and does
so during migrations. Like other swift birds and allies, it is unable to move
on the ground, but freely climbs branches and tree trunks – the bird only needs
space to open its wings in the fall. Its legs are short but tenacious, and its
claws are short, hooked and pointed. All four toes are pointed forward; their
underside is covered with bristles that improve seizing the branch.
Motley basket-mouth does not make nests, but simply glues its eggs with saliva
to a tree branch, less often to the wall of an empty tree-trunk hollow. There
are 1-2 eggs in the clutch, they are white or pinkish and very noticeable, so
parents always hatch them and feed in turn. The chicks hatch blind, but covered
with cryptically colored down and have tenacious paws that allow them to cling
tightly to the substrate. They require little heating and can stay alone for
quite a long time, since both parents get food for them, heating them only at
night. Chicks grow quickly, and by the end of summer they are already flying
as well as their parents. When the chick leaves the nest, the family breaks
up; this species is monogamous and does not form permanent pairs.
Motley basket-mouths fly south quite early – already at the end of summer, with
the first night cold spells. The wintering grounds of motley basket-mouth are
Southeast Asia and the Philippines. During the wintering, the birds manage to
breed offspring, and in the spring the young bird migrates to the north, where
it can nest in the first year of life. The lifespan of motley basket-mouth does
not exceed 7-8 years.
This bird species was discovered by Bhut, the forum member.
Bacororo,
left-sided crookbill hummingbird (Asymmetrochilus bakororo)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: humid tropical forests of Amazonia.
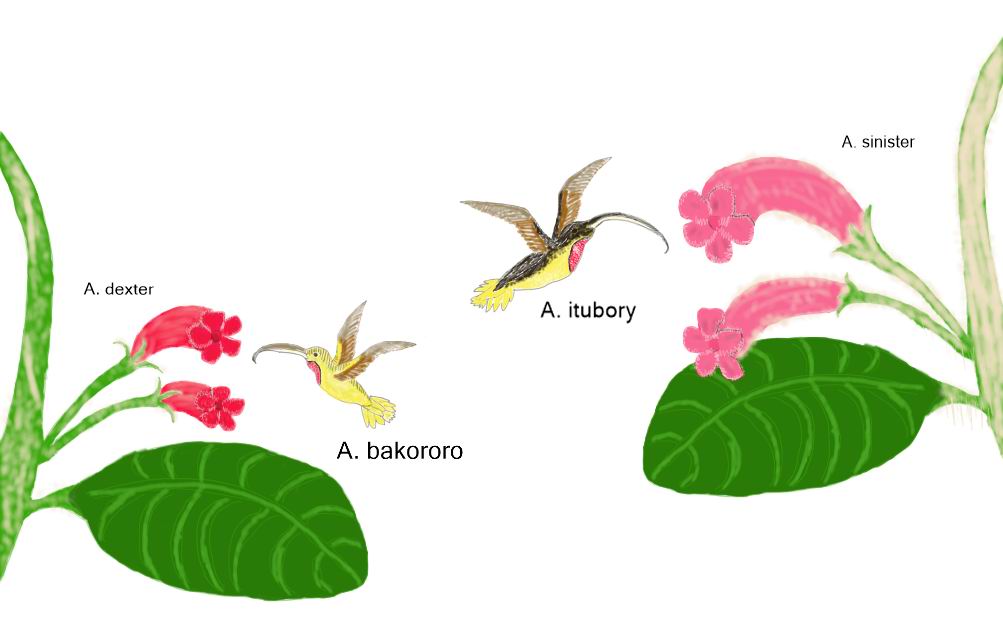
Picture by Fanboyphilosopher
Specialization during the evolution has two sides. On the
one hand, it helps to closely related species to avoid a competition. But, on
the other hand, the specialized species may die out much more probable at the
changes of inhabitancy. Deep specialization limits adaptive abilities of species
and consequently very much specialized species meet in rather stable conditions.
In tropical areas of South America during the evolution two “twin” species of
hummingbirds had evolved; they are adapted to feeding on nectar of asymmetrocalyx
– plant of Gesneriaceae family having asymmetrical flowers. Hummingbirds of
genus Asymmetrochilus and plants of genus Asymmetrocalyx represent a surprising
example of coevolution, resulted in formation of strictest interdependence of
plant and bird. Species of hummingbirds are named after twin brothers from fairy
tales of Indians of bororo tribe.
Bacororo is the typical representative of hummingbirds family. It is tiny bird
(its length does not exceed 6 centimeters, not including beak) having intensive
metabolism; at night bacororo runs into the catalepsy accompanying with body
temperature decreasing. Bacororo is exclusively nectarivorous species of birds,
and because of features of beak structure it is simply not capable to eat any
other food. Its beak is rather long – at adult bird its length is comparable
to body length (not including tail). The beak shape at bacororo is very original:
it is strongly bent to the left. The bird feeds on flowers of right-sided
asymmetrocalyx (Asymmetrocalyx dexter), having a bend to the right, and
the beak of bird precisely conforms to flower by form. Besides this this species
of hummingbird takes from flowers of asymmetrocalyx small insects and spiders
making the important source of proteins especially necessary for nestlings.
Colouring of bacororo is very bright – the top part of body of bird and its
tail are golden with greenish shade and expressed metal shine. On head, shoulders
and the top part of back feathers are patterned with thin ripples. The throat
of bacororo has red color and is bordered by thin black strip. Secondary feathers
are brown, and primaries have grey color with expressed metal shine. When the
bird gets in sunlight beam, shining wing tips outline a trajectory of their
movement on each side of bird’s body. Tail of bacororo is short and fanlike.
Bird does not have cops and other ornaments of feathers.
Strict food specialization of bacororo means, that food resources are the factor
limiting settling of this species of hummingbirds. Limitation of food resources
during the evolution has resulted in occurrence of aggressive territorial behaviour.
Bacororo does not suffer presence of congeners in the territory, and makes exception
only for females during the posterity rearing. This bird aggressively expels
relatives from the territory. Being near to fodder plants, bacororo aggressively
attacks even butterflies and large bees, accepting them for competitors. Each
bird preciously remembers places of growth of fodder plant and every day visits
them by fixed route. Bacororo is very good flyer, able to manoeuvre in thicket
of lianas and branches, to hang on the spot and even to fly a tail forward.
The nesting pair at bacororo forms only for the nesting period. The nesting
cycle in tropical climate repeats up to three times a year. Male involves the
female to its territory, making courtship flight around the large and well blossoming
fodder plant. Thus it hangs in air above a plant for a long time, especially
in sunlight beams. Courtship ritual of these birds is very simple: male only
allows the female to feed on plant, and does not drive her away. The pair of
birds builds nest of spider web and thin plant fibers, skilfully weaving it
under leaf of fan palms. Thus leaf segments bound by fibers are used as a basis
on which the conic nest is suspended.
In clutch there is up to five eggs, but usually two or three nestlings survive
to the flight from the nest. At young birds colouring of body is lack of metal
shine and beak is straight. Due to such beak young birds can feed on some species
of plants and survive successfully in critical period of search of their own
territory. Shortly before the coming of sexual maturity, at the age of four
months, the beak gets the shape characteristic for representatives of the present
species. Life expectancy of bacororo seldom exceeds three years.
In cooler foothills and on heights, in area of cloud forests, the close species
lives - itubory,
or right-sided crookbill hummingbird (Asymmetrochilus itubory).
It differs from bacororo by beak bent to the right. Also itubory is итубори
is larger a little (its length is up to 8 cm); it is connected with cooler mountain
climate, in which this bird lives. Being the close relative of the previous
species, itubory differs from it by features of colouring. In back and head
colouring of this bird there is more black color, and metal shine on the top
part of trunk is lack – these features are connected to necessity to be warmed
up faster after night cold. The throat of itubory has very intense metal shine,
due to which bird can warn relatives of its presence from apart.
This species nests only twice per year, and in clutch there are no more than
three eggs. A fodder plant at itubory is left-sided
asymmetrocalyx (Asymmetrocalyx sinister), the epiphyte from cloud forests
of South America.
The idea about existence of these species of birds was proposed by Simon, the forum member.
Wasp-mimic
hummingbird (Vespimimornis flammeocarbunculoides)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: tropical forests of Amazonia.


Pictures by Amplion
The mimicry is one of the most effective ways of protection
against predators. In any case of this phenomenon around well protected model
species the complex of species imitating it is forming. They may be equally
well protected, or similarity to the protected species is their only protection.
In South American selva there is the species of insects inspiring fear to the
majority of smaller forest inhabitants – corsair
hornet (Toxivespula flammeocarbunculus), the large species of social wasps
armed with strong poison. Colouring of these insects, black with red spots on
thorax and tip of abdomen, is very well familiar to all animals living in the
neighbourhood. These wasps are imitated successfully by black
waspmoth (Hymenursula vespiptera) from bears family (Arctiidae). Alongside
with it the external similarity to these wasps protects another inhabitant of
Amazonian forests – one of small species of hummingbirds.
Wasp-mimic humming-bird is a tiny bird, whose length does not exceed 6 centimeters.
This bird resembles wasp in colouring, and also in body shape. The plumage of
this hummingbird is black, but on its back there is reddish-orange spot. Short
rounded tail also has orange colouring, imitating a coloring of tip of abdomen
of that wasp. The first primarily feathers have grayish color with silvery shine.
When wasp-mimic hummingbird flies, buzzing by wings, it creates complete illusion
of flying wasp. This similarity saves this bird from enemies – large birds,
snakes and small predatory mammals. Nevertheless, corsair hornets correctly
define a deceit, and frequently attack wasp-mimic hummingbirds, perceiving them
as a kind of prey.
Similarity to wasp is additionally amplified by two special modified feathers,
growing above eyes. They have thick shafts, and vane is advanced only along
the terminal one third part of feather; these feathers imitate short antennae
of insect. In their structure some sexual dimorphismis expressed – male has
a little bit longer “antennae”.
Beak of wasp-mimic hummingbird is long and thin, little bit bent downwards.
Nectar makes up to 70 % of its diet; other part of diet is made of tiny insects.
This bird is not specialized to feeding on the certain species of plants, as
some species
living in the same place. Therefore wasp-mimic hummingbird is widely distributed
in tropical areas of South America.
This species of birds lives in forest canopy and feeds in flowers of epiphytic
plants and trees for which the formation of flowers on trunk (cauliflory) is
typical. Wasp-mimic hummingbird does not avoid a neighbourhood of congeners,
and frequently up to ten birds may feed simultaneously in the same small site
of tree crone.
This bird nests in couples keeping during one nesting season. The couple of
birds in common builds small and deep cup-like nest of spiderweb, plant fibres
and lichens. In clutch there is up to five eggs with white shell, which are
incubated mainly by the female. Within one year wasp-mimic hummingbirds can
rear up to three hatches.
The idea about existence of this species was proposed by Simon, the forum member.
Tayin
(Tayin migratorius)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: the south of Chilean region and Patagonia, Tierra del Fuego; wood areas.
Wintering in Andes foothills.

Picture by FanboyPhilosopher

Picture by Pavel Volkov
Hummingbirds are typical representatives of New World ornithofauna.
In human epoch they were settled from Alaska up to Tierra del Fuego. In moderate
and cold areas these birds were migrating ones, but in areas of tropical climate
they lived constantly and expressed the greatest specific variety and specialization.
A certain number of species of these birds had died out because of habitat destruction,
but some of less specialized species, mostly from the number of ones not connected
to forest habitats, had successfully gone through epoch of human domination
and climatic changes of the boundary of Holocene and Neocene. In early Neocene
inside this group of birds active speciation began, and new species of hummingbird
began restoring of their lost positions. One species of hummingbirds, tayin,
is the most southern species of these birds – it lives in the southernmost part
of South America. The name of bird descends from the local name of one species
of hummingbird lived in Tierra del Fuego in human epoch and became a hero of
folk tales of natives.
Hummingbirds are creatures, for which specialization to feeding on nectar is
characteristic. And in areas of seasonal climate such food source is accessible
only during the short part of year. Therefore tayin, living in such conditions,
is a migrating species. It appears in nesting places among the last of migrants,
and flies out among the first ones. It is bird up to 10 sm in length (not including
beak).
This species of birds feeds in flowers Dendrofuchsia,
or fuchsia tree. Some species of
hummingbirds of Neocene epoch demonstrate an extreme degree of specialization
to feeding on the certain species of plants. As against them, tayin has very
low specialization to any certain kind of food. It allows it to exist in conditions
of seasonal climate and to eat various kinds of food during the seasonal migrations.
Tayin has long straight beak which length makes approximately one third of the
general length of body (including tail). With such type of beak it can feed
equally successfully in opened flowers of grassy plants and in tubular flowers
with straight corolla which fuchsia tree produces. At the end of summer, when
flowering of plants finishes gradually, tayin passes to feeding on insects,
seizing them by beak, as if by tweezers. Tayin prefers to eat tiny and soft-bodied
insects – plant lice and larvae of butterflies. At the end of summer when berries
of fuchsia tree ripen, this bird willingly pecks overripe fruits.
At night, especially in spring and in the beginning of an autumn, tayin runs
into condition of deep catalepsy – body temperature decreases and physiological
processes are slowed down in great degree. It permits this bird to save energy.
In the summer, in conditions of long day, catalepsy is superficial, and the
bird quickly returns to normal life. In order to be warmed faster after long
night, tayin has velvet black colouring. In the morning bird fluffs feathers
and quickly gets warm in beams of rising sun. On feathers of head feathers have
purple metal shine, and on wings shine is greenish. In middle part of bird’s
tail two lengthened feathers colored white grow. Wings of tayin are peaked;
flapping them, bird utters silent hum.
Tayin is a solitary species of birds. Only for nesting period couples are formed,
but they break up right after nestlings leave nest. Courtship displays begin
right after the spring migration of birds. Males occupy territories on which
dendrofuchsia grows, and protect them from contenders. Courting the female,
male makes figured flight around of her, inviting on the territory. The nest
is plait of plant fibres and spider web. Birds arrange it somewhere in remote
place – usually on very thin branch where the predator can not get. In clutch
there are two or three eggs hatched only by the female. Nestlings are brought
up by both parents. Within one year tayin has time to rear two hatches in the
north of an area, and only one in the south. Young birds have time to leave
nest before the dendrofuchsia deflowering and feed on it for any time.
Tayin flies away for wintering shortly before the first frosts. This species
makes distant flights along the belt of mountain forests in Andes, keeping in
areas of moderately warm climate, and does not fly high in mountains where the
wood vegetation vanishes.
Giant
archilochus (Archilochus gigas)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: southeastern North America, northeastern Central America, islands of
Caribbean Sea.
Can a hummingbird become a giant? Yes, but only out of necessity – usually these
birds belong to the smallest birds. Back in Holocene, a finch-sized giant hummingbird
(Patagona gigas) lived and flourished in South America, but it was rather an
extreme, since most hummingbird species were small and tiny birds. A distant
relative of this species, giant archilochus, a descendant of ruby-throated hummingbird
(Archilochus colubris) of Holocene epoch, also reached similar sizes. The reason
of it was a special lifestyle, different from that of most hummingbirds, especially
from the tropics of Central and South America. It is a migratory species that
performs long-distance migrations.
Its ancestor also migrated from North America to Central America and back, while
remaining the typical size for a hummingbird. This helped it survive at the
beginning of Neocene, when the climate changed dramatically and the boundaries
of natural zones shifted. Part of the population of the ancestral species became
extinct; the other part began to migrate over Caribbean Sea and the Gulf of
Mexico between North and Central America. Various islands of Caribbean Sea served
as resting places for the ancestral species.
Then the ice age ended and the world changed again, but Archilochus continued
to migrate over the sea along the already established routes, bypassing the
mountains and deserts of Mexico. These birds gradually formed a new species
– giant archilochus.
It is a very large hummingbird by the standards of the family: equal in size
to a swallow of the human era and resembling it in proportions. Unlike other
hummingbirds and swifts, giant archilochus can take off from the ground, making
a high jump, but still prefers to live in trees and spends a lot of time in
the air. These birds have a solid green plumage with a metallic sheen, especially
on the belly, which makes them less noticeable against the sky when viewed from
below and among the foliage when viewed from above. Like at the ancestor, males
of this species have a ruby-red spot with a metallic sheen on the throat. Compared
to its ancestor, it is a larger, stronger and hardier bird species, capable
of doing long distances in a day. Its diet has also changed – it has relatively
less amount of pollen and nectar and more invertebrates (even large ones), and
also fruits and berries with soft flesh have appeared, which giant archilochus
pecks without problems: by the standards of hummingbirds, its beak is wider,
shorter and stronger. The main source of nectar for this species is cactus and
agave flowers.
Giant archilochus nests on the territory of North America when it returns there
in the spring, less often on islands of Caribbean Sea, if it has no opportunity
to return to North America in proper time. This species does not form permanent
pairs, but looks for a new nesting partner every year. In a cup-shaped or conical
nest made of plant material on a thin tree branch, there are 2 white eggs. The
female incubates them for about 2 weeks, and the male feeds her and drives away
enemies with stabs of his pointed beak. 3 weeks later, the fledglings of this
species take wing and by autumn they are ready to migrate south across the sea.
Some of them do not fly to Central America, but remain on the islands, maintaining
and vivifying populations of this species there. Young birds can already nest
on wintering grounds, and more than half of adult birds capable of reproduction
also manage to make a second brood there, and these young birds migrate to North
America in spring.
The maximum lifespan of giant archilochus is 4 years.
This bird species was discovered by Bhut, the forum member.
Cactus
glimmer hummingbird (Micrarchilochus cactobius)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: deserts and semi-deserts of Mexico, thickets of “old virgo” cactus.
In Neocene, hummingbirds from the Americas continue to compete for the brightest
plumage and unusual appearance. Some species have mastered new habitats, and
one of them is cactus glimmer hummingbird, a miniature inhabitant of deserts
and semi-deserts of Mexico.
Body length of this bird is up to 7 cm. Another 2.5-3 cm falls on straight thin
beak. Tail is short and fan-shaped. Wingspan is up to 13 cm. Legs are short,
adapted only for perching on branches.
Like many other members of the family, cactus glimmer hummingbird has bright
plumage: almost the entire body is pink with a metallic sheen, wings are blue,
tail is light gray with black spots along the edge; on the head there is a shiny
bright green “cap”. By the nesting season, females shed and get a dull color
– grayish-green with dark green stripes on the head and back and numerous dark
longitudinal strokes on head and throat feathers.
Cactus glimmer hummingbird lives in Mexican deserts and semi-deserts. During
the day, it can fly very long distances in search of flowering desert plants.
This species is not specialized for feeding on a certain kind of plants, and
feeds on various species of desert plants as they bloom – more often on cacti,
agaves and herbs of lily family. In winter, birds migrate to Central America
and feed on flowers of mountain cacti. Cactus glimmer hummingbirds use water
very efficiently, and therefore they can live in desert. At night, birds unite
in flocks and sleep in various shelters, mainly among cacti. The enemies of
this species are birds of prey, small mammals, snakes and lizards.
Breeding season of cactus glimmer hummingbird comes in spring, during the mass
flowering of cacti. Males display on top of a cactus, usually an “old
virgo” one, threatening each other with squeaking and trying to drive
them off the cactus. When a pair is formed, male and female begin to build a
nest together. To do it, both birds choose a bunch of cactus “hairs”, twist
its tip so that it looks like a cup, and then glue this cup to the surface of
the cactus with saliva and continue to weave it with neighboring hair strands,
adding cobwebs and plant fibers. In this case, “hairs” growing above the nest
cover the nest from the sun and mask it from predators. One cactus can carry
up to 15 nests disguised in this way.
This species is monogamous. Female lays 2-3 eggs in the nest. At this time male
flies to cactus flowers and sucks nectar, feeding a part of it to the female
and pollinating the cactus in addition. After 2 weeks, nestlings hatch from
the eggs. At first, the female protects nestlings from the scorching sun during
the day and from the cold at night, while the male brings nectar for the whole
family. As the juveniles grow, the female begins to leave the nest and feed
them. After fledging, young birds leave the nest at the age of about 20 days,
and the pair breaks up.
Life expectancy is up to 5 years, usually less.
This bird species was discovered by Mamont, the forum member.
Yaremusi
(Trochiliviator yaremusi)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: forests of Venezuela, the Antilles, Mississippi walley.
In Neocene, the geography of the Central American region has undergone significant
changes. The Panama Strait appeared, and the Antilles formed an intermittent
land connection between the continents. In Neocene at the shores of Caribbean
Sea, there is a small genus of wandering hummingbirds that make long migrations,
including across the sea straits. They all descend from the species Colibri
cyanotus, and one species of the genus is yaremusi.
This species is a small bird: the body length without a beak is 7-8 cm; the
long beak adds 6-7 cm more. In the body shape this bird resembles an ancestral
species – body of light build, not very long wings, pointed at the tips, small
wide tail and small head. Legs are short, bird can perch on branches, but walks
with difficulty. The wingspan is 13-14 cm. The basic body color is turquoise,
belly is light yellow, primary and secondary feathers are pink. On the back
edge of the tail there is a red stripe shaded with blue. There are three black
cross smears on the chest. On head there is a blue “cap”, and throat is light
green. Legs and beak are gray-blue.
Yaremusi is a migratory species. Every year they migrate along a single route
in thousands of flocks. Yaremusi spends the winter in Venezuela, where these
birds live in groups of 20-30 individuals at the same area of the forest. But
at the beginning of the migration, yaremusi flocks unite into huge flocks that
move from their homes and fly north along the chain of the Antilles. During
flights across the sea, hummingbirds fly in fairly dense flocks, demonstrating
the phenomenon of murmuration, so from the distance their flock looks like one
living organism and takes bizarre outlines during the movement. Sometimes, during
flights, birds do not find any food sources at all, so before the flight and
at all stops these hummingbirds feed actively, accumulating large fat stocks.
Flocks of birds break up over land, and birds migrate in small groups through
the forests, uniting only before forcing water barriers.
After 2-3 weeks, yaremusi hummingbirds fly to North America, where they live
in the same way as in South America. In the warm season, birds migrate further
north and feed on the nectar of large herbaceous plants. In autumn, birds migrate
back to South America.
Throughout the migration route, yaremusi hummingbirds feed on the nectar of
various plant species. They play an important role in pollination of a wide
variety of species, like other hummingbirds. Birds get protein by consuming
small insects together with nectar. The enemies of these birds are tree snakes
and lizards, and spiders and parasitic insects often attack their nestlings.
Yaremusi make two clutches per year – in South and North America. Male and female
build a nest together of twigs, blades of grass, cobwebs and down. Both parents
alternately incubate the clutch for 11-12 days. Nestlings are fed by both parents.
After 2-3 weeks, young birds leave the nest. Life expectancy is up to 10 years.
Also in the north of South America a similar hummingbird species yaremuna
(Trochiliviator yaremuna) lives. It is smaller (up to 5-6 cm long),
and has red longitudinal stripes on the primary feathers; throat is light red.
This species turns west during the flight along Greater Antigua and flies to
Yucatan.
The names of these species mean “hummingbird” in Caribbean languages.
This bird species was discovered by Mamont, the forum member.
Violet
guarasyaba (Guarasyaba violaceus)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: Atlantic forests of South America.
Even with the impacts caused by human activity during the Holocene, some species
persisted. One of these species was the swallow-tailed hummingbird (Eupetomena
macroura), which in the Neocene give rise to the genus Guarasyaba (“hair of
Guaraci” in Tupi language; Guaraci is the personification of the sun). The type
species of this genus is the Violet guarasyaba, which lives in the forests of
the Atlantic coast of South America.
All members of the genus Guarasyaba are sexually dismorphic. The males are large
with 22 cm, they have a crest and a gorget with iridescent violet feathers.
These feathers are also present in the belly, the rest of their body is bright
green. Males also have a short forked tail with two long tail coverts, characteristic
shared by all males of this genus. All females in the genus are similar; they
are smaller than the males, with 14 cm and lacking the crest, gorget and iridescent
feathers of the males and with a white underbelly and throat. The rest of their
feathers are dull green. The females have a short forked tail, but lack the
long tail coverts. Both sexes have medium sized straight grey beaks.
These hummingbirds mainly feed on nectar of flowering trees in the canopy of
the forest. They also eat tiny insects.
Violet guarasyabas are aggressive, attacking birds twice their size. The breeding
season occurs during all the year. In courtship, the male hovers in front of
the sitting female and chases her through the air, and the two may perform a
'zig-zag flight' together. The nest is a cup-shaped structure lined with soft
plant fibres and clad on the outside with lichen and mosses, held together with
spider webs. It is attached to a horizontal twig in smallish trees. The female
lays two eggs and only her is take care of them. The chicks hatch after 15 days;
they are initially naked, save for some grey down on the back, and have dark
skin. They start to grow feathers 5 days after hatching, starting with the remiges.
The young are fed 1-2 times per hour, and the female spends about half of the
day brooding and feeding her offspring, and the other half flying around and
feeding. The young fledge after 24 days but still return to the nest to sleep
and be brooded for some more days; they are independent some 3 weeks after fledging.
Two broods may be raised subsequently, sometimes reusing the nest; due to the
prolonged breeding season. This species reach sexual maturity with 2 years and
its lifespan is of 9 years.
Other species in the genus Guarasyaba:
Blue
guarasyaba (Guarasyaba caeruleus)
Living in the montane forests of Southeastern South America, males of this species
are 21 cm long with iridescent blue feathers in the crest, gorget and belly.
Golden
guarasyaba (Guarasyaba auratus)
Living in the savannas and woodlands of Central South America, males of this
species are 23 cm long with iridescent yellow feathers in the crest, gorget
and belly. This species is a common polinator of the violet
trumpet tree (Tabebuia violacea).
Ruby
guarasyaba (Guarasyaba puniceus)
Living in the Amazon forest, male of this species are 24 cm long with iridescent
red feathers in the crest, gorget and belly.
This bird species was discovered by João Vitor Coutinho,
Brazil
Mountain
redgem (Rubetrochilis andinus)
Order: Apodiformes (Apodiformes)
Family: Hummingbirds (Trochilidae)
Habitat: South America, Andean Plateau.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The hummingbirds were one of these survivors and now
their descendants are the proof of their survival. In the Andean Plateau, a
new species appeared. It's the mountain redgem, a bird that is adapted to survive
in this region. During the cold nights, these birds will enter in torpor, congregating
in small flocks inside rock crevices or in protected branches. By staying together,
they are capable of staying warm. During the winter, these birds will migrate
to the lowlands close to the Andes.
The mountain redgem is a small bird. They have 11 cm of length, with males having
a 10 cm long tail. Both sexes have scaly brown upperparts, but only the males
have red iridescent feathers in the throat and chest. In females, this part
of the body is reddish ocre. Both male and female have a dark striped belly
and a dark patch through the eye, with a white supercilium. The males have dark
purple uppertail coverts with light ochre bands and central tail feathers are
long with white tips. The tail of the females are short.
Like all other species of hummingbird, the mountain redgem feeds on nectar.
However, they will eat a variety of insects, including mosquitoes, fruit flies,
gnats in flight, or aphids on leaves and spiders in their webs.
When foraging, the mountain redgem is a solitary bird, with males even being
aggressive with each other when searching for food. However, when they find
a place to spend the night, these hummingbirds will congregate in flocks to
stay warm during the torpor. The nesting season starts immediately after they
return to the Andean Plateau during spring. The male sings during courtship.
It's thin and squeaky, interspersed with buzzes and chirps, and is drawn to
over 10 seconds in duration. During the breeding season, males can be observed
performing an aerial display dive over their territories. When a female flies
onto a male's territory, the male rises up about 30 m before diving over the
female. As the male approaches the bottom of the dive, it reaches an average
speed of 27 m/s. At the bottom of the dive, the male travels 23 m/s, and produces
an audible sound by the tail feathers. During this period, the males will attack
each other aggressively.
The female raises the young without the assistance of the male; there are three
broods per season. The female bird builds a nest in a shrub or tree, or in vines.
It’s constructed of plant fibers, downy feathers and animal hair; the exterior
is camouflaged with chips of lichen and plant debris. The nest materials are
bound together with spider silk. The female lays two to three white eggs that
are incubated for 13 days. The young fledge for 3 weeks. They can already breed
in the next spring. This species has a lifespan of 5 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Motley
fossorrallus, digging rail (Fossorrallus guttatus)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Antarctica; tundra, swamps, meadows, crooked forests, coastal thickets.
In the Holocene, Antarctica virtually lacked any terrestrial life. In the Neocene,
the climate has warmed, and the biodiversity of the polar continent has increased,
but it is still a harsh land where it is extremely difficult to survive. Terrestrial
vertebrates are extremely few there and limited to birds; many of them are migratory
and winter in South America, Africa, Meganesia or New Zealand. The few flightless
species are mostly small. Most often in various habitats of the continent there
are mousebirds, small burrowing
passerines. Apart from them, few birds remain to winter on the continent. One
of them is the spotted fossorrallus – a burrowing rail, a descendant of one
of the Gallirallus species (most likely the buff-banded rail (Gallirallus philippensis)).
Fossorrallus is a stocky, heavy-built bird with a muscular body, large head,
rather massive long beak, as well as wide, powerful legs resembling legs of
gallinaceous birds. The wings are greatly reduced and hidden in the plumage,
and the feathers are soft (much like the feathers of paleognaths) and at the
same time thick. The eyes are relatively small; eyesight is poor by the standards
of birds. The nostrils are shifted closer to the tip of the beak; the sense
of smell is very good, which compensates for the vision deficiency. There are
also long feathers around the beak, functioning like the vibrissae, like in
kiwi (Apteryx). The down is very dense and provides protection from the cold.
Feathers cover the bird’s legs to toes. The underside of the toes is covered
with thick keratinized skin. The feathers are light brown with small specks.
Males have 2 black stripes on each side of the head, stretching from the beak
to the throat. The beak and toes are reddish-brown. There is no tail made of
feathers – the tail feathers resemble contour feathers in texture. The size
of the bird is quite large by Antarctic standards – the body length is about
37 cm, the weight is up to 550-800 g. Females and males are about the same size.
Spotted fossorralluses are omnivorous birds: they eat rhizomes, berries, shoots
and leaves of plants, lichens, invertebrates, small birds and carrion. In summer,
these birds usually feed in dense thickets of plants, where they are difficult
to spot by flying predators. Fossorralluses
run well, although while running they waddle from side to side and shake the
vegetation – a massive physique and adaptation to life in burrows has an effect.
During the polar day, birds are active around the clock with sleep breaks. During
the change of day and night (autumn and spring), fossorralluses prefer to feed
at night.
Birds spend a significant part of their time in burrows, which represent complex
systems of passages and chambers, vaguely similar to badger’s hole.
In late summer and autumn, fossorralluses feed especially actively in order
to store enough fat for the winter, when it is more difficult to get food. At
the same time, they store food in burrows, bringing there the rhizomes and tubers
of plants, as well as killed small animals (more often mousebirds). In winter,
these birds almost do not leave underground and undersnow galleries, feeding
on food stocks, as well as lichens, plant rhizomes, underground invertebrates
and sometimes live mousebirds. Around the settlements of these birds, whole
systems of tunnels appear under the snow on the surface of the ground. In especially
severe cold, fossorralluses fall into a winter sleep, and at this time even
their body temperature decreases by several degrees.
With the arrival of spring, the breeding season begins – at this time snow cover
often is still on the ground. Fossorrallus males display, inflating their chests,
raising underdeveloped wings and uttering throaty, muffled calls “uuuummmmmmm-khhhh”.
Such calls are audible from afar. After mating, the pair usually moves into
the female’s burrow, where she lays light pink speckled eggs in a nest of plant
debris, quite large in size, in the amount of 3-5 ones. During the season, the
birds make only one clutch, both parents incubate alternately. Chicks covered
with black down hatch in a month. They are semi-precocial, which is typical
for rails – during the first 3 months they are fed by their parents, although
young birds begin to look for food themselves very early. Young birds spend
the whole winter in the parent burrow, and by spring they leave it. The parent
pair does not break up after that – the fossorralluses are monogamous and dig
and renovate the burrow together. They often use the tunnels of neighboring
pairs, uniting them into systems stretching for hundreds of meters.
The natural enemies of fossorralluses are primarily flying birds of prey, from
which these rails hide in dense vegetation and in their burrows. Young birds
often die in winter from starvation.
Young birds become sexually mature at 2 years old, and fossorralluses live up
to 20 years.
This bird species was discovered by Dohlozavr, the forum member.
Kiwi
tinihanga (Vermirallus sylvanus)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Heavily forested areas and woodland, both mountainous and lowland,
North and South Island of New Zealand.
The iconic Kiwi (Apteryx), was a pugnacious bird, but one of the first to succumb
to the depredations of the introduced predators and their descendants, in late
Holocene New Zealand. However, the niche of an adaptable, pugnacious worm-eating
bird was not one lacking viability, and it was soon filled again.
Descended from the Buff Banded Rail (Hypotaenidia philippensis), the New Zealand
False Kiwi is a fair sized bird, similar in size to a standard rooster, with
a long, slightly curved bill greatly resembling that of the true kiwi (hence
the name: “Fraud Kiwi” in Maori). Similarly, this bird finds its food by probing
the forest floor in search of earthworms, though it will also opportunistically
eat grubs and insects. The bill is covered with lines of mechanoreceptors inside
and out, especially at the tip, and it finds its prey mainly by touch, unlike
the kiwi which also used its sense of smell.
Though this bird can fly well in short bursts to escape predators, it vaguely
resembles the Weka (Gallirallus australis) in shape and coloration, with overall
dark brown feathers that contain some lighter and darker longitudinal streaks.
Feet are large and bear big claws, sometimes when foraging it will turn over
the ground as a chicken does with its feet. This bird is generally solitary,
and they may antagonize their own kind outside of the breeding season. Like
the true kiwi, it is nocturnal, but also similarly, its behaviour is flexible
and it may be seen by day or at dusk.
Breeding occurs from August to October, and females may produce more than one
clutch, to separate males in one year. Courtship is brief and consists of the
male attracting females with a loud piping call from far away, females will
judge males based on the strength of their call. Up to 4 eggs are laid, chicks
are mature by six months, the nest is a deep scrape made in lee of a large tree
or even sometimes dug some way into the ground. Non-mating call is a series
of chirping whistles or a rasping chirr when annoyed. Able to run quickly in
order to escape mammalian predators, it will defend itself by pecking and kicking
if cornered. Lifespan is up to 15 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Black-chested
rail (Pseudogallirallus nigrothorax)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Antarctica, coastal areas.
Rails are birds that paradoxically combine limited flight qualities and the
ability to colonize the most remote oceanic islands. They were among the first
permanent inhabitants of Antarctica, and among them several species capable
of living in harsh Antarctic conditions all year round evolved. The black-chested
rail is one of the largest Antarctic rail species. It reaches a length of 70
cm with a weight of about 2.8-3 kg. In the constitution this species resembles
gallinaceous birds: it has strong moderately long legs with thick toes and powerful
claws. The beak is moderately long, with a hooked tip.
Black-chested rails are not capable of flight, and even their wings have turned
into useless rudiments, almost invisible from the outside; they can run at speeds
up to 35-40 km/h. The wool-like feathers are long (up to 8 cm), especially on
the neck and back, grey-colored with blurred cross striation; there is a black
spot on the chest, three longitudinal black stripes stretch on the back, and
there is a black “cap” on the head. Beak and legs are grayish-yellow. Eyes are
relatively large, equipped with bristle-like “eyelash” feathers.
Black-chested rails are carnivorous birds, feed on insects, nymphs of insectfishes,
false jabbies, ravage
the nests of seabirds, peck carrion, often ambush mousebirds
at burrows and kill with a well-aimed peck in the neck. Quite often they attack
the nests of sea swans and steal eggs,
often even kill and eat the chicks. Less often, they feed on seeds and tubers
of plants.
By winter, birds grow very fat and shed; winter plumage is lighter and thicker
than summer one.
The nesting behavior of birds is very interesting. The male, using his beak
and paws, digs a burrow up to 2 m deep and up to 90 cm wide. He lines the bottom
of the nest chamber with dry plant stems, fern fronds, feathers and moss. The
male’s display is accompanied by dull cracking sounds and a demonstration of
a ready burrow. To strengthen the relationship, birds eat prey together. The
female lays 3-4 large brownish-green eggs in a burrow and incubates them for
about 26 days. The chicks are black and fluffy, like at all rails. Young birds
spend the first winter together with their parents, learning to search for food.
This species is rarely found far from the shores of the continent, because in
winter these birds look for food mainly by the sea. In the case of blizzards
or strong winds, they hide in their own burrow, or use other natural shelters
in which they huddle together and heat each other. In winter, they feed mainly
on the corpses of marine animals washed ashore, littoral mollusks and crustaceans,
and can also attack seabirds. In winter, kleptoparasitism is also characteristic
for them – they often terrorize penguigulls
and kaveskars coming out of the sea, beating
them and forcing them to regurgitate part of the catch.
This bird species was discovered by Corvus, the forum member.
Rantakana,
Finnish diving rail (Natanorallus rantakana)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Europe, wetlands, lakes, slow-flowing rivers and other calm bodies
of water. Wintering along the shores of Fourseas, populations in places with
a mild climate are sedentary.
In the Holocene, rails were common on all continents (except Antarctica) and
on many islands. These poorly flying (and in many cases completely flightless)
birds suffered greatly from humans, but quite numerous species managed to survive
and in the Neocene they gave a new radiation of species, including large crane-like,
Phorusracus-like and ostrich-like
forms. At the same time, many Neocene rails are small, often even tiny birds.
One such example is the rantakana, or Finnish diving rail (“rantakana” – rail
in Finnish), a descendant of one of the species of Porzana genus.
This bird is widespread in Europe, from the taiga in the north to the subtropics
in the south. Nevertheless, in all places of its range, rantakana keeps to the
water – after all, this bird is even more aquatic than its ancestor. Outwardly,
rantakana resembles a spotted crake, but has visible differences – for example,
the body is more elongated and massive, the toes are equipped with leathery
fringes, like at coots, beak is thicker and more powerful. The coloration of
rantakana is typical for secretive near-water birds – brown with darker small
spots on the back. In both sexes, the front part of the head, chest and belly
have a dull blue-lilac color, in males somewhat brighter than in females. Beak
and legs are icterinous; eyes are red. Rantakana is somewhat larger in size
than spotted crake – about 29-35 cm long, weighing 180-300 g. Several similar
species live in other regions of Eurasia.
Rantakana swims longer and more actively than spotted crake, and spends more
time in the water. The bird is omnivorous, but more carnivorous than most other
small rails, and due to its stronger beak is able to attack and kill fairly
large prey, such as small birds and mammals (for example, various shrews). Rantakana
also feeds on invertebrates, small fish, amphibian larvae and plant food. This
bird feeds like spotted crake, but swims more actively and walks less along
the shore. Activity is mainly nocturnal, during the day the bird keeps hidden.
The mating period of the rantakana is from early April to mid-May in different
parts of the range. The mating call of the male is the call “u-ki, u-ki, u-kiii”,
clearly distinguishable from afar (the rest of the time, the rantakans usually
utter a low “gurgling” or “buzzing”). It is a monogamous species; birds make
nests in thickets of coastal plants, from dry stems and leaves. The clutch consists
of 6-10 marsh-colored eggs with speckles; both parents incubate. The chicks
are covered with black down (which is typical for rails), are of semi-precocial,
after hatching they can walk and follow their parents, but they still cannot
regulate their body temperature themselves, and in the first few days they can’t
search for food. The chicks begin to feed themselves after 16-22 days; they
take wing at the age of about 50 days.
Migration for wintering occurs from the end of August to the first half of October,
depending on the climatic zone; some populations do not migrate. Rantakana winters
in the subtropics of Fourseas, usually on the southern shore of the reservoir.
The return to the nesting sites occurs in late spring.
The life expectancy of the Finnish swimming rail is up to 8 years.
This bird species was discovered by a Dohlozavr, the forum member.
Striped
False Woodhen (Ralligallina paludiphila)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Swamps, swamp-forest and mangroves surrounding Arafura and Carpentary
Lakes.
Rails are a very successful widespread group of birds, able to colonize islands
with ease, and having an almost global distribution. The genus Hypotaenidia
was notable in giving rise to many flightless and some oversized forms.
In northern Meganesia, in the swamps and mangroves surrounding Arafura and Carpentary
sea lakes, lives the Striped False Woodhen, a descendant of the Banded Rail
(genus Hypotaenidia). This is a large rail similar in size to a chicken, with
long legs and toes, and an elongated, conical beak. Bird slightly resembles
the New Zealand Weka (Gallirallus) but is fully able to fly, being mainly diurnal.
The bird is dark brown above, with the wing feathers scalloped with black, underside
is barred in contrasting black and white, face is pale and there is a dark stripe
through the eye. Bill and feet are bright red. Generally reluctant to fly, it
only does so when pursued by a predator. Spending most of its time on the ground,
it stalks and wades about on marshy ground and mudflats, in search of prey,
which consists of insects, small fish, crabs, worms, lizards and occasionally
small snails. The beak is relatively long for a rail and serves to aid it in
snatching or selecting prey, though it can be used to kill larger prey with
a sharp peck.
Breeding occurs all year round, and females may produce up to 10 clutches per
year. Courting consists of overtures made to the female including gifts of food,
male displays by bowing head and fanning out the tail and wing tips. The nest
is a shallow cup of woven leaves and reeds usually made in the crook of a low
tree. Up to 4 eggs are laid, chicks are mature by six months, and young are
expelled from the pair’s territory once fully grown. Birds pair for life and
forage by ranging a small distance from one another, pairs have distinct territories,
and fights will break out over territory, which involve pecking, leaping and
flapping. The call is a loud series of whistles and is made to maintain contact
within the group, individuals invariably respond to calls in case of territorial
interlopers. They are generally shy and secretive, fleeing by running for cover
at the slightest sign of danger. These birds are frequently preyed upon by crocodiles,
large monitors, pythons and other reptiles, raptorial birds and carnivorous
marsupials, rats and other birds may consume their eggs. Lifespan is up to 15
years.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Takahe
ki te mere (Deinonotornis socialis)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Upland and mountain slopes where tussock grass and other grasses grow,
South Island, New Zealand.
The effects upon native New Zealand birds by Holocene invasive predators is
best exemplified by the plight of Takahe (Porphyrio hochstetteri). This flightless
rail was a slow-moving specialist, eating the stems of grasses, and had no defences
against introduced mustelids. However, its ancestor and near neighbour, the
Pukeko (Porphyrio melanotus) was extremely common and prosperous all over New
Zealand and Australia.
Subsequently, the Pukeko served to evolve a replacement for the Takahe in the
Neocene. Indeed, it had already given rise to both the North Island and South
Island Takahes on separate occasions during the early Holocene.
The False Takahe, however, differs from its Holocene counterpart in being more
formidable, in the face of both marsupial and placental predators, and it some
ways resembles Australian Native Hens (Tribonyx) habitually. This bird is about
the size of a large domestic hen, coloration is indeed similar to both of its
relatives, with a bright green back, and a head and chest which is dark purplish
blue, turning to navy on the face, bill and feet are bright candy red. Its bill
is deep and triangular in profile, suited to shear off the bases of mountain
grasses, which makes up the majority of its diet, but also includes insects
and sedges. Feet are large and prehensile, it will often hold food using its
feet. Unlike the true Takahe, it is flighted, able to retreat in short bursts
of flight much like a grouse. Exceedingly aggressive towards predators, in a
similar way to a hen or rooster, it lives in loose flocks of up to 10, which
actively patrol their territory. When confronted with a predator, it brings
to bear its sizeable bill, and also large, blunt spur-like growths on the ends
of the wings (hence the name, meaning “takahe with war club”, in Maori, referring
to the wings), opponents are beaten over the head with wings and pecked. False
Takahe is diurnal, but also most active in the morning and afternoon. These
birds are generally silent, relying on visual signals within the flock, though
they will make a puffing hiss when angry.
Breeding season runs from July to September, depending on the lushness and availability
of grasses on which it feeds and amongst which it nests. Social groups are led
by an alpha pair, with the female also mating with other males in the group.
The nest is a shallow, round grassy mattress in which is laid up to 8 eggs.
Young are attended by the female, but are able to forage and follow their mother
on their own a few days after hatching, initially feeding on insects but switching
to grasses after they reach half adult size. Lifespan is up to 10 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Timid
rail (Pardirallus timidus)
Order: Gruiform birds (Gruiformes)
Family: Rails (Rallidae)
Habitat: Easter Island.
The human actions during the Holocene caused the collapse of many ecosystems.
Easter Island was one of these cases. This place was completely deforested and
many of its endemic species perished. However, with the arrival of the Neocene
it was able to recover, developing a new endemic fauna. One of the species living
here is the timid rail, a descendant of the plumbeous rail (Pardirallus sanguinolentus)
that arrived in the island coming from South America and evolved here to this
new species. This species lives close to freshwater reservoirs, hidden among
the reeds.
The timid rail has 27 cm of length with very reduced wings, making it impossible
for them to fly. It has a long, downward curving pale gray bill and is grayish
brown with a pale chestnut breast and conspicuous whitish rump patch. This plumage
allows these birds to camouflage in the reeds. The legs are dark gray.
These birds are omnivorous, feeding on invertebrates and plant material. When
foraging, they rarely leave the cover of reeds and bushes, hoping to avoid the
attack of flying predators.
The timid rail is a mostly solitary bird, with only pairs living together. Due
to staying hidden in the reeds and bushes during most of the time, it’s most
common to hear their loud callings. The pair builds the nest together and shares
the incubation of the eggs. The structure is made of branches and pieces of
grass, constructed among the vegetation. The female lays a clutch of 7 white
eggs with brown spots. The eggs are incubated for 14 days. The brown, downy
precocial chicks soon leave the nest but are fed and protected by the parents.
Fledging occurs after four to five weeks. They reach sexual maturity at the
age of 18 months and have a lifespan of 15 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Ostrich
rail (Otiocrex struthiopus)
Order: Gruiform birds (Gruiformes)
Family Running rallids (Dromorallidae)
Habitat: steppes of Central Asia.

Picture by Amplion
In Holocene epoch in steppes and savannas of Old World and
Australia different species of bustards, large birds of Craniformes order were
widely settled. But human activity like destruction of habitats and unlimited
hunting these birds have come to naught survival chances of bustards during
geological cataclysms of the end of Holocene.
In Neocene when in the Central Asia extensive steppes have appeared from the
east of Fourseas, they were occupied by large running birds similar to ostriches.
Probably, if man has not interfered with destiny of bustards in Holocene, it
most likely would be descendants of any bustard species. But in connection with
bustard extinction their ecological niche was occupied by descendants of other
bird: smaller neighbour and distant relative of bustard, the crake (Crex crex).
These birds, large flightless ostrich rails migrate by small flocks in steppes
of Central Asia from coast of Fourseas up to bush thickets of Mongolia.
Ostrich rail is the average-sized bird: its growth is about one and half meters,
and weight is about 30 kgs. Appearance of this bird is typical for steppe flightless
birds: at it there are long legs permitting to run quickly, and long neck strongly
increasing the field of vision. In connection with specialization to running
at this bird on legs only two toes remained: middle and external ones (as at
ostriches). It reduces friction against the ground and permits to develop high
speed – bird can run within approximately hour having speed up to 40 kms per
hour.
Ostrich rail is not able to fly at all though it had kept wings. They are wide
and short, and are using for maneuvering during run, and males also use them
for courtship demonstrations. On wings of males there are long soft primary
feathers shown during courtship rituals. Tail of this bird is very short: it
is not seen from under wings.
Colouring of ostrich rail feathering is characteristic for inhabitants of steppes
and savannas: yellowish-brown background is banded by thin cross strips; neck
is covered with short light yellow feathers. Legs of bird are featherless and
light brown.
Ostrich rail has strong wide beak: bird eats all kinds of forage that are possible
to be found in steppe – small vertebrates and large insects, and also firm seeds
of graminoids.
Usually these birds wander in steppe in small flocks – about 10 – 20 birds at
all. In flock of ostrich rails there are both males and females. They do not
differ almost from each other in colouring, only males have more bright color
of feathering, and females seem “faded” near them. These birds are very cautious:
while some birds search for livelihood, others look in sides searching for possible
predators. Frightened away birds
run the same way as the crake, their far ancestor: having extended head forward
and shaking it at each step. Not reducing speed of run they can make very sharp
turns opening thus wings.
New habit of life has made males of this species more tolerant to each other
outside of nesting season, but during nesting steppe appears shared between
them to nesting sites. Each male signs the right to its possession of the certain
territory, loudly shouting. The voice of ostrich rail is more similar to long
rolling chirring. During shout male extends neck upwards and blows throat which
acts as resonator. Borders of sites during nesting are determined by any remarkable
reference points: high bushes, separate trees or termitariums. Each male supervises
territory approximately three hundred meters in diameter.
This bird is monodin; pair is formed only to one nesting season. The male has
occupied territory, actively shows itself, making courtship dance: it opens
wings, spins in its place, stamps legs, “bows” and loudly shouts. At this time
its voice is most similar to hoarse croak. If on its territory other male gets,
skirmish with the most unpredictable consequences is possible: the lawful owner
of territory will snatch at the newcomer, striking it impacts by legs and beak.
Usually conflict does not reach it and stranger bird escapes. If, however, newcomer
is audacious and is not going to surrender, it is impossible to escape fight.
Being in heat, males drag each other by feathers, tearing them out, kick and
try to tumble down the contender on the ground. When fight comes to an end by
flight of one contender, winner some time chases it loudly shouting.
Ostrich cranes arrange nest on the ground. Pair of birds rears posterity in
common: male protects borders of territory for a short time replacing hatching
female at the nest. At this time parents movable by instinct are able to drive
off from nest practically any predator meeting in steppe. Protecting nest birds
blow throat, stand on end feathers on neck and loudly hiss. If the enemy approaches
too close, impacts by legs and beak are put to use. Due to such protection the
survival rate of ostrich rail chicks is rather high.
In clutch there are 4 – 6 large eggs. Shell of eggs is motley: on yellowish
background there are randomly scattered brown and black stains. The incubating
lasts about four weeks. Chicks are covered with black down; along their backs
longitudinal yellow strip passes. Having dried they at once abandon nest and
do not come back any more to it. Parents in the beginning feed nestlings from
beak, keeping food in mouth and enabling nestlings to peck it. Nestlings quickly
study to eat independently and parents provide them more protection than food.
To the end of second week of life at young birds feathers start to appear, and
they change black chick down for juvenile yellowish-brown feathering. To this
time territorial claims of adult birds gradually disappear and families with
posterity unite to rather numerous flocks. At one-year-old age young birds get
colouring of adults, and being one year old it can take part in nesting.
Life expectancy of ostrich rail does not exceed 15 years.
Lesser
crail (Rallogeranus minor)
Order: Gruiform birds (Gruiformes)
Family: Crails (Neogruidae)
Habitat: North America – woodlands, deciduous forests, prairie areas in the
east of the continent.
The Neocene epoch is characterized by changes in the structure of the fauna
– large and vulnerable representatives of various groups of animals died out
as a result of human activity, leaving no descendants. One of the extinct groups
were the true cranes (Gruidae). In the Neocene, their ecological niches were
occupied by other birds – usually by unrelated stork birds (storks, herons,
ibises, etc.), as well as representatives of a new branch of large gruiform
birds – representatives of the crail family, descendants of small rails (Rallidae).
Crails are common in Eurasia and North America, and one of their species is
the lesser crail.
Lesser crail is a relatively large bird: its height is about 90 cm; wingspan
is no more than 140 cm. It is a bird of a more robust constitution than other
species of the genus, with shorter and thicker legs, neck and beak. It is a
bird with relatively bright plumage of green and rust-brown color with a light
beak. The color of the wings and body is brown with thin cross stripes. Head,
neck and chest are dark green with a metallic sheen. The legs are dark gray.
This species, unlike other, larger crail species, does not have a throat sac.
Instead, this species has a mobile fiery-red crest, which birds can raise and
lower, giving each other alarms, warnings and threats, as well as using it during
courtship.
The coloration and small size of the lesser crail are associated with a lifestyle
atypical for these birds: it avoids wetland areas and prefers dry places – undergrowth
of deciduous forests, woodlands, prairies, rarely forests at the foothills,
especially in the Appalachian region. Like other crails, lesser crail feeds
on animal food – usually insects and other invertebrates, less often small vertebrates.
This bird does not eat carrion.
Lesser crail usually nests in a secluded place on the ground. In the forest
zone, birds can occupy empty nests of large birds nesting in trees. Near the
nest, the birds are very quiet and secretive. There are 2-3 speckled eggs in
the clutch; both parents incubate them alternately for 5 weeks. The chicks hatch
covered with down and having eyes opened, and after 2-3 days they leave their
nest together with their parents forever. To do it, the chicks fearlessly jump
off the tree and land safely in the bushes due to their low weight. Their color
is brown with a black longitudinal stripe from the crown to the tip of the tail
and black spots on the sides of the head, which allows them to disguise themselves
in the undergrowth when enemies appear.
Lesser crail forms permanent pairs, often remaining until the death of one of
the mates, but the couple does not have a permanent nest, so every year bird
couples either build new nests, or find and renovate abandoned ones – of relatives
or some other birds. By winter, the chicks of lesser crail become independent.
In the south of the range, birds lead a sedentary lifestyle; in the north they
are migratory. Northern populations winter in the south, in the Mississippi
Delta, on the Gulf of Mexico coast and in Florida. Separate birds fly to Cuba.
Sexual maturity in this species occurs only at the age of 2-3 years. Lesser
crail can live for several decades.
This bird species was discovered by Bhut, the forum member.
Steppe
crail (Rallogeranus aridophilus)
Order: Gruiform birds (Gruiformes)
Family: Crails (Neogruidae)
Habitat: Central Asia, steppes and shrub thickets.
Among the groups of terrestrial animals that became extinct at the boundary
of Holocene and Neocene, there were true cranes (family Gruidae). The main reason
for their disappearance was the decline in numbers associated with the disappearance
of habitual places of living and the increased level of disturbance due to human
activity. The number of these birds has greatly decreased, the range has been
fragmented, and separate populations have disappeared one by one. Their ecological
niches were occupied mainly by stork birds,
as well as by crails – large descendants of rails (Rallidae), a characteristic
group of birds of the Holarctic. Most of the species of these birds live in
wetlands, but there are species that prefer
forests and dry open areas. Steppe crail is one of the steppe species of
this genus.
Steppe crail is not the largest representative of the genus: about a meter long,
it weighs about 3.5 kg and has a relatively large wingspan – about 1.8 m. The
basic color of the plumage is gray with large cross ripples formed of the cross-striped
coloring of the feathers. On the head there is a movable crest of darker, coal-black
feathers with a greenish and blue metallic shine. On the cheeks of the bird
there are large red-and-white spots – areas of red feathers with white tips
forming a spotted pattern. At the base of the neck there is an outgrowth of
the trachea – an elastic resonator sac covered with gray skin; using it, steppe
crails can communicate to each other at a distance of several kilometers. During
the call, this outgrowth is greatly inflated. The voice of this species is a
high-pitched acute whistle.
Beak at this species is proportionally shorter than that of its semi-aquatic
relatives, since this bird species forages mainly on land, eating small vertebrates,
insects, grass tubers. This bird is especially fond of cereals; in autumn, when
the young birds have already left the nest, the families of these birds roam
the steppes and peck the ears of cereals.
Despite living in steppe areas, steppe crail prefers to nest not on the ground,
but on steppe trees or among tall shrub thickets in order to be safer from predators.
In addition, this bird is more territorial and aggressive than other species
of the genus, and drives away from the vicinity of its nest not only relatives,
but also other large birds, and even mammals, including medium-sized predators.
It happens that a pair of nesting birds even jumps on large saigochenias,
and tries to peck them in the head, driving them away from the nest.
The mating season of the steppe crail comes in the spring, after the return
from wintering grounds: adult males renew or rebuild nests and call females
to them, and younger bachelors try to drive them away from the territory or
take over the nest. At such moments, serious fights arise between the males
of the steppe crail: the birds fight, kicking and striking each other with their
wings. When a female appears, the male invites her to the nest, inflating the
resonator and uttering sounds similar to cooing.
The nest is a large platform on a tree or in the crown of a tall shrub. Birds
throw a large pile of branches into the crown, on top of which there is a flat
nest of branches and grass laid with a thick layer. Usually there are 2 eggs
in a clutch, less often 1 or 3. Both parents incubate for 5 weeks; at the daily
age, the chick leaves the nest and wanders the steppe with its parents. Large
reptiles can eat a chick; adult birds are attacked by eagleravens and other
predators. At about 10 weeks old, steppe crail chicks are fully fledged and
can already fly, and at 18 weeks they become independent and are expelled from
the parent territory. Birds fly for the winter to the high-altitude plateaus
of the Himalayas, where they winter in a cold climate in snowless valleys.
Sexual maturity occurs in 2-3 years, in females earlier than in males. Steppe
crail can live for several decades.
This bird species was discovered by Bhut, the forum member.
Central
European crail, willia (Rallogeranus willia)
Order: Gruiform birds (Gruiformes)
Family: Crails (Neogruidae)
Habitat: Central and Eastern Europe, riverbanks, lake coasts.
The true cranes died out shortly after the end of the human era. Their evolutionary
decline was superimposed by habitat destruction, which fragmented and reduced
the ranges of these birds, which accelerated their extinction. Their place in
the Neocene was taken by related birds, rails. Their descendants, cranils, are
convergently similar to cranes and are their ecological counterparts in the
Holarctic.
Willia is a large crail inhabiting central and eastern Europe. The stature of
an adult bird is about 140 cm, the wingspan is 2.25 m, the weight is up to 6
kg; the male is slightly larger than the female. The plumage of most of the
body in adult birds is dark gray or brown with mottling on the back and wings
– there are two color morphs, the frequency of which varies from one population
to another. The underside of the wings is always white, which, along with the
large size, allows recognizing these birds in flight. Unlike other crails, willia
prefers to live near rivers and flowing lakes, or other bodies of water with
clean water, although it regularly hunts in wetlands.
The main food of birds of this species is small aquatic and terrestrial animals:
from small fish and large invertebrates to rodents, reptiles and amphibians.
Occasionally a bird eats berries, usually in autumn. This is a migratory species:
willia winters in the savannas of North Africa and in the valley of Saharan
Nile, where young river vipers (Potamobitis planiceps)
become part of its prey. Outside of the nesting season, birds keep in
pairs, sometimes in harems of three birds: a male can mate with two females.
The breeding season is at the end of April – May, when the willias return from
wintering. At this time, males grow thin and long white feathers on the wings
(wing coverts) and on the breast; females have a tuft of feathers. In this way
they resemble their mythological prototypes – rusalkas (vilas, willias, from
Russian mythology). In the established family, birds claim the territory with
a series of prolonged calls and beak clicks – a signal that the nesting place
is occupied. In addition, the action is accompanied by characteristic male and
female dances, which include bouncing, flapping wings, tossing twigs and bowing.
In single or widowed birds, such “dances” are much longer and more intense,
and are usually not accompanied by vocalization.
The nest is arranged on a hill in a swampy area, or on an island in the middle
of a reservoir, but more often in bushes on the shore. There are two or three
eggs in the clutch of these birds, which are incubated only by the female. The
male guards the borders of the territory of the pair and drives away small predators.
Incubation lasts 30 days. Chicks appear well developed, sighted and covered
with down. After drying out, they quickly learn to walk and leave the nest.
Young birds fully fledge after about 70-75 days. By this time they are already
beginning to learn to fly. The bond between parents and offspring persists for
a long time: young birds fly to winter with their parents, becoming completely
independent already in Africa, from where they return to their homeland on their
own. Sexual maturity occurs at the age of 3 years, life expectancy is up to
25-30 years.
This bird species was discovered by Nick, the forum member.
Perkele,
horned crail (Rallogeranus perkele)
Order: Gruiform birds (Gruiformes)
Family: Crails (Neogruidae)
Habitat: tundra and forests of Northern Europe from Scandinavia to the Urals.
Crails represent a young group of birds that evolved after humans. They are
descended from small rails, but are ecological analogues of cranes that died
out at the end of the human epoch. These birds are a very characteristic element
of the Holarctic fauna and are widespread across the Northern Hemisphere – from
Greenland to Siberia, and from the northern tundra to the south of the taiga
zone. The north of Europe is inhabited by perkele, a relative of the northern
crail (Rallogeranus borealis).
The height of this bird is about 1.25 m, the wingspan is up to 1.95 m, and the
weight is about 5.5 kg. The appearance is very characteristic: two narrow tufts
of elongated feathers grow on the head behind the eyes, resembling horns (hence
the name meaning “evil spirit” in Finnish). The color of the back and wings
is gray with red and brown mottling, like fire under ashes. The belly is white.
Sexual dimorphism in coloration is not pronounced. Perkeles live in small groups
– male, female and juveniles of the last year. Birds strictly guard the territory
from their relatives, driving them away with wing beats and chest pushes. The
bird defends itself from predators with beak blows, inflicting deep wounds,
and often tries to peck the enemy in the eyes or nose. Birds prefer to live
in impassable areas of swampy forest surrounded by deep marshy areas. Perkeles
feed mainly on small vertebrates – fish, amphibians, chicks of wading birds
– and crustaceans, less often snails. At the end of summer, a significant part
of the birds’ diet consists of seeds and berries of marsh plants. Outside of
the breeding season, birds are usually silent, but chicks are called by rumbling.
Birds return from wintering grounds by the time the snow melts on reservoirs,
and in the first days after the flight they eat off. The mating season begins
at the end of April, when relatively stable warm weather is established. Pairs
are formed only for one breeding season. Males call females with screeches resembling
woman screaming, uttering them many times in a row during the morning hours.
Each of the males has a different timbre. When a pair of perkele has formed,
the birds already vocalize together, claiming the territory; a couple can even
slightly expand the boundaries of their own possessions, especially at the expense
of solitary birds. These birds nest on islands in the swamp and in thickets
of reeds; the nest is a pile of grass based on broken reed stalks. There are
3-4 eggs in the clutch, which are incubated by both parents; the incubation
period is 25 days. The chicks hatch sighted and covered with down, the next
day they leave the nest and follow their parents. At first, the parents feed
them, then from about the 2nd week of life, the chicks get food themselves,
and the parents only protect them and take them to places rich in food. Young
birds treat each other relatively peacefully, showing aggression only when there
is a lack of food. From about 52 days of age, young birds are already able to
fly, but they are completely independent only from the autumn migration. Horned
crails winter on the shores of Fourseas and on the Iberian Peninsula. Females
take part in reproduction at the age of 2 years, males at 3 years; life expectancy
is up to 26-30 years.
This bird species was discovered by Nick, the forum member.
Polar
oyster-cracker (Molliaphagus arcticus)
Order: Charadriiformes (Charadriiformes), suborder Charadrii (Charadrii)
Family: Oystercatchers (Haematopodidae)
Habitat: coast of Arctic Ocean, northern Atlantic.
Decrease of variety and efficiency of ecosystems in human epoch, human economic
activity and also global ecological crisis at the boundary of Holocene and Neocene
rendered significant influence to the fauna of sea coasts. The part of species
of sea birds had died out, and the survived species had been compelled to adapt
to new conditions. Among survived birds there were oystercatchers (Haematopus)
– usual birds of sea coasts in Holocene epoch, widely settled at the Earth.
In ice age some populations of these birds had receded from coast of northern
seas to the south and had survived, passed to feeding on river invertebrates.
In the beginning of Neocene warming river descendants of oystercatchers settled
back to habitats of their ancestors. One sea species had widely settled at coast
of Arctic Ocean and became the main consumer of bivalve molluscs. According
to its diet the whole genus of birds has received the name “oyster-crackers”
though some species of these birds live in places, where oysters do not meet
at all.
Polar oyster-cracker is rather large representative of plovers: by size it is
equal to mallard and weighs up to 700 grammes. Contrast black-and-white colouring
of ancestral species has strongly changed. Top of head, back part of neck, wings,
back and waist of polar oyster-cracker are speckled with numerous brown, black,
white and ochre speckles on yellowish background, forming cross lines on every
feather. The bottom part of body, throat and bottom part of head are white.
Beak and paws of polar oyster-cracker are bright yellow; at male their color
is more sated. Color of beak and legs at each species of oyster-crackers is
special – it helps them to distinguish individuals of the same species. Paws
at all species are rather long and lack of back toe; other toes are edged by
wide leathery “scallops”, as at grebes.
Beak of oyster-crackers is long and strong. Polar oyster-cracker eats mainly
bivalve molluscs, searching them in littoral zone. Having found a shell at the
coast, it wedges beak in crack between shell parts, and, using it as a can opener,
opens shell, cutting adductor muscle, and then quietly eats mollusc. If there
are no shells at the coast, oyster-cracker dives for them under water – it is
able to swim and to dive as well, as duck. Rowing by paws and having pressed
wings to a body, it tries to find shells under water. Having found a mollusc,
it catches it by beak or by several strong impacts tears off shell from substratum,
and carries to the shore, where opens and eats. Quite often oyster-cracker diversifies
its diet with invertebrates gathered at the sea coast – it digs out from sand
shrimps, scuds and also worms. The competitor of this bird in polar areas is
“sea woodpecker” (Picicorvus
albiventris), the bird of corvid family, also able to dive. Sometimes “sea
woodpeckers” dive near to polar oyster-cracker, and snatch out from under its
beak molluscs torn off by it from substratum. On land these birds also enter
conflicts – usually “sea woodpeckers” ravage nests of oyster-crackers and steal
forage from their nestlings which behave rather passively during feeding by
nature.
Pairs at oyster-crackers form to one nesting season, and birds in common bring
up nestlings during the long time. The nest is located on rocks, among pebble
or in sand. Actually, the nest is not present as such – birds dig a small pit
in sand, where eggs are laid. In clutch there are 2 – 3 eggs.
Polar oyster-crackers fly perfectly, and for winter they migrate in warmer areas
– eastern populations winter at Fourseas, and western ones migrate to the Atlantic
coast of Europe.
At the territory of Eurasia some close species of this genus live:
River
oyster-cracker (Molliaphagus potamophilus) is a pigeon-sized bird that
lives at the rivers of Eurasia from the Western Europe up to swamps of Western
Siberia. It is colored in olive tone, masking the female hatching eggs in grass.
The bottom part of body is colored lighter, rather than top. The top part of
body has dark speckled pattern. Beak and legs are orange-yellow.
Pacific
oyster-cracker (Molliaphagus larvatus) by size is equal to duck. It
lives at Asian coast of Pacific Ocean, at Big Kurils, and separate individuals
fly to the coast of Beringia in summer. It has grayish color with specks; across
eyes “mask” – wide black horizontal strip – is stretched. Legs and beak are
red.
Fourseas
oyster-cracker (Molliaphagus cyanipes) is a small species, equal to
pigeon in size. It lives at the coast of Fourseas, and by colouring is similar
to polar oyster-cracker, but white color in plumage is replaced to yellowish.
Obviously, this species is the descendant of polar oyster-cracker wintering
at Fourseas. But two these species very precisely differ from each other: at
Fourseas oyster-cracker legs and beak have strange blue color.
These species of birds were discovered by Simon, the forum member.
Shore
awlbill (Gravicharadrius pseudoalatus)
Order: Charadriiformes (Charadriiformes)
Family: Plovers (Charadriidae)
Habitat: North Atlantic, Loki Island.
The islands represent a real laboratory of evolution: here the processes of
speciation occur very quickly. The few species that managed to settle these
isolated habitats quickly produce descendants differing in ecology, which soon
occupy all possible ecological niches and form a stable ecosystem. The degree
of endemism among island species is very high, especially if these species have
limited ability to settle.
Loki Island appeared in the temperate latitudes of the North Atlantic after
the human disappearance, and its fauna and flora are truly endemic, whereas
the islands that existed in the human era are inhabited by descendants of human-introduced
species that could not have appeared on the islands in natural way: rabbits,
weasels, hedgehogs, rats and mice. The presence of such species greatly influenced
the further historical development of fauna and flora.
One of the typical birds of Loki Island is shore awlbill. This is a large flightless
sandpiper species, a descendant of the Kentish plover (Charadrius alexandrinus)
widespread on Earth in the human era. There are evidences of its origin in its
appearance, but this bird has some unique features. This is a chicken-sized
bird weighing about 2 kilograms. Shore awlbill has an elongated body with a
long forked tail of black color. The coloration of the bird retains typical
features of plovers: the upper part of the body is colored brown; the belly,
chest and throat are white, and there is a bright red cap on the head, turning
into a thin pointed tuft. A black “mask” covers the forehead and sides of the
head, and a thin ring of white skin emphasizes the large eyes of the bird.
Shore awlbill has a long straight pointed beak of black color. This bird feeds
on a variety of terrestrial invertebrates – insect larvae, beetles and spiders.
These birds often go to the seashore and catch marine invertebrates in the littoral
zone and in the sand on the shallows.
The bird’s legs are bright red, strong and three-toed. Shore awlbill can run
fast, often changing the direction of running, especially in case of danger.
In open places, the bird keeps very carefully, preferring to hide in the thickets
in case of danger. In the bushes and in the forest, the bird runs, stretching
its head and neck forward.
In due course of evolution, shore awlbill not only became a large bird; it also
completely lost the ability to fly. It has very short underdeveloped wings.
They are very small, barely more than 4-5 centimeters long, and covered with
short feathers. A notable feature of this species is the development of three
long primary feathers at the tip of each wing. They are so long that their tips
lie over the end of the tail when the wings are folded. At the same time, it
seems that the wings of the bird are not reduced – hence the name. These feathers
with wide vanes serve only for courtship display; they are white with black
tips, longer in the male than in the female.
During the courtship ritual, the male and female run next to each other, opening
and turning up their underdeveloped wings; at this moment, the catchy color
of the feathers is clearly visible to the chosen breeding partner. These birds
gather on a sandy beach with short and sparse vegetation for courtship displays.
Displaying males open and turn up their wings, trying to look larger, and stamp
their feet on the sand. If the female pays attention to the male, he rushes
after her, screaming and raising slightly his chest and head. While running,
the female opens her wings and turns up her feathers, and the male does the
same. When the male catches up with the female, he grabs her by the wing with
his beak and stops running. This is a sign of the forming of a new pair.
Shore awlbill nests on the ground, arranging a simple nest in a hole. The nest
is often hidden among plants or under the roots of a tree. There are 3 to 4
eggs in the clutch, which are incubated exclusively by the female. After the
completion of egg laying, the male leaves her, and the female takes care of
all the offspring. She is able to distract enemies from the nest by pretending
to be wounded, vocalizing loudly and showing brightly colored “mating” feathers
on her wings. The chicks of this species are spotted, with large spots on a
light background of down. They hatch mobile and active, can feed themselves
and escape from enemies. In case of danger, they crouch to the ground and remain
motionless, but if they are found, they run away from the enemy. When fleeing,
the chicks can abruptly change direction of their run. The female leads a brood
up to two months of age; there are two broods per year.
Chicks fledge at the age of 1 week. The juvenile plumage is very different from
the adult – it is gray with black marks, larger and more numerous on the back.
The legs of a young bird are brown. At the age of six months, the plumage is
replaced by a two-tone one characteristic for adult birds. But the “mating”
feathers grow later – only by the age of one year, when the birds reach sexual
maturity. Life expectancy is no more than 5-6 years.
Arafura
Turtle-Cleaner (Lacustrocharadrius cheloniphilus)
Order: Shorebirds (Charadriiformes)
Family: Plovers (Charadriidae)
Habitat: Arafura Lake, lake margins and shallows, symbiote of Brontochelys.
Shorebirds had varied experiences during the age of man, some became extinct
due to loss of habitat, but the persistence of suitable waterside habitat over
a wider area assured their survival into the Neocene in some way, with even
some specialized forms arising.
Arafura Turtle Cleaner is a small plover descended from the genus Charadrius.
It has mostly maintained a similar size and shape to its ancestor, but differs
in fully webbed feet, connected to its ability to occasionally dive underwater.
Individuals are bright white below, and contrasting black above, with a double
chevron of white over the nape; bill and feet are bright pink. Colour of this
animal relates to its niche and the need to advertise itself to its host. It
lives by congregating around the giant Arafura Brontochelys,
feeding off of parasitic invertebrates and epifauna that accumulate on these
turtles, as well as other small prey stirred up as the animals move. This bird
is diurnal, but synchronizes its activity with the movements of its host.
An individual bird will constantly seek out a turtle or more than one, if it
is without a host, it will advertise its availability by making aerial displays
and producing a high piping call. Individuals may stay with a single turtle
for up to a week before moving on to another, and they generally stay close
to them as much as they are able to. Individuals may squabble over feeding rights
and space, pecking and making short swoops. Diet consists of various kinds of
parasitic and epifaunal invertebrate that accumulate on the turtle’s shells
and skin, including barnacles, small bivalves, snails, sea lice, young crabs,
other crustaceans, and leeches, as well as commonly feeding on shrimp and worms
stirred up in the water as the turtles move. The birds will walk over the exposed
backs and heads of the turtles, or flit about from one to the other as they
submerge, this bird can also dive to feed on submerged parts of the turtles,
or to chase prey that is stirred up.
During the dry season from August to October, males will start displaying and
calling in order to attract females, flicking their wings and making small flights.
When a female selects a suitable male, the male will begin to court by making
offerings of food, after several mounts they will begin to seek out an area
of dry ground on the lake margins in which they can make a nest. The female
makes a shallow depression hidden amongst vegetation some distance from the
waterline, in which she will lay up to six eggs at intervals of 48 hours. During
nesting the female devotes herself to caring for the nest, and the male devotes
himself to defending the nesting ground and gathering food. The eggs incubate
within 28 days, and the young are precocial, parents lead them to a vegetated
area to hide from predators, during this time the pair will alternate attending,
feeding and defending the chicks. As soon as the chicks fledge at about 30 days
they are brought by the pair to start to feed around the turtles again. Pairs
usually disband after the young leave, but may mate together again on subsequent
years. Lifespan reaches about 10 years, and these birds are commonly preyed
upon by snakes, large fish, and occasionally other birds.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Eurasian
false bustard (Charadrotis eurasiaticus)
Order: Shorebirds (Charadriiformes)
Family: False cranes (Pseudogruidae)
Habitat: steppes of Eurasia.
The human actions in the Holocene caused many impacts in the ecosystems. Because
of these actions, many species went extinct in the Neocene. However some species
survived and give raise to descendants, one of these descendants is the Eurasian
false bustard. As its name already says, these birds are not bustards, in fact
these birds descended from the plovers (Charadrius), but through evolutionary
convergence acquired characteristics similar to that of birds of the Family
Otididae, this new species is very similar to a bustard of human era. They are
part of the Family Pseudogruidae, being closely related to the large-billed
crabstork. This bird lives in the steppes and prairies of Europe and Central
Asia. With the exception of one species, all false bustards are migratory, being
able to fly long distances. The Eurasian false bustard flies to savannas of
Africa and Zinj Land during the winter.
These birds are larger than their relatives of the Genus Pseudogrus. They have
97 cm of height, with a length of 110 cm and a wingspan of 200 cm. They have
a small head set on a long neck, and a bulky body with large legs. Their plumage
is brown above and white below, with black stripes down the sides of their long
neck. In flight, the long wings show large areas of black and brown on the flight
feathers and a white patch on the darker primaries. The long legs are yellowish
in color and the bill is yellow with a black tip.
They eat insects and other small invertebrates, small reptiles, frogs and rodents.
Occasionally the Eurasian false bustard will also eat shoots and seeds. To kill
the vertebrates, they stomp on them until they are rendered stunned or unconscious
enough to swallow.
During the spring and summer, these birds are solitary, with only mated pairs
being seen together. During migration, they form small familiar flocks formed
by the parents and their chicks. The breeding season occurs in the spring. The
normal clutch is 2 to 3 stone colored eggs laid inside a scrape on bare ground.
The eggs are incubated by both adults for 25 to 27 days. One of the parents
will stand guard nearby while the other incubates the eggs. The nidifugous chicks
are cryptically colored and follow the parents soon after hatching. The young
chicks freeze and crouch when alarmed and the cryptic plumage makes them hard
to detect. The parents will aggressively attack any predator that approaches
the nest. Juveniles are independent by their first winter, but normally stay
with their parents until the next breeding season. The lifespan of this species
is 24 years.
Other species in the Genus Charadrotis:
Indian
false bustard (Charadrotis nigrocephalus)
Living in the plains of the Middle East and Indian Subcontinent, this species
has 72 cm of height, with a length of 83 cm and a wingspan of 165 cm. Their
plumage is similar with the Eurasian species, with the exception of their head
that is black. It’s a non-migratory species.
Siberian
false bustard (Charadrotis sibiricus)
Living in the regions of tundra in Siberia, this species has 65 cm of height,
with a length of 78 cm and a wingspan of 157 cm. They have brown upperparts
with dark grey marks on a tawny buff background; the underparts are black. The
head is boldly patterned with black, white and buff. The neck, long and thin,
is buffy brown with a thin black line down the front that joins the black breast.
The tail is dark brown. During the winter, this species migrates to the grasslands
of Meganesia.
Tundra
false bustard (Charadrotis leucoventris)
Living in the regions of tundra in North America, this is the only member of
the genus Charadrotis that reached the Americas. Their closest relative is the
Siberian false bustard. This species has a height of 69 cm, with a length of
80 cm and a wingspan of 163 cm. Their plumage is similar to that of the Siberian
species, with the exception that their underparts are white rather than black.
During winter, this species migrates to the savannas and grasslands of South
America. Before reaching here, they make brief stops at the Great Plains.
This bird species was discovered by João Vitor Coutinho, Brazil.
Antarctic
chicken seedsnipe (Antarcthinocorus migrans)
Order: Charadriiformes (Charadriiformes)
Family: Seedsnipes (Thinocoridae)
Habitat: Antarctica, meadows and marshlands; wintering in South America
Neocene Antarctica favors the existence of various species of waders. Some of
their species find food on the coast, and others – in the interior of the continent,
some are omnivores, and others are highly specialized. One of the most unusual
species of waders is the Antarctic chicken seedsnipe, which has become an ecological
analogue of gallinaceous birds and ground pigeons that do not live in Antarctica.
Chicken seedsnipes are medium-sized waders, their weight reaches 800 grams.
In their constitution, these birds are similar to willow ptarmigan: they have
a robust body, short strong legs and a small head with a short beak. However,
unlike their “doppelgangers” of galliform birds, seedsnipes have long pointed
wings and fly well. The tail of these birds is quite short, of rounded shape.
The coloration of chicken seedsnipes is not bright. The upper body is colored
light brown with a black-and-white scaly pattern formed due to the fact that
each feather is edged with thin white and black stripes along the edge. This
coloring provides the seedsnipes with an excellent disguise: a bird hiding (for
example, hatching eggs) blends into the background of vegetation and becomes
inconspicuous. The belly and lower part of the breast of chicken seedsnipes
are white. The sides of the head and throat of the female are ash-gray, and
at the male they are red-brown with black mottling. Also, the male has a small
oval-shaped black spot on both sides of the head at the base of the beak. The
legs of these birds are light yellow; the beak is light brown with a dark tip.
The vocalization of the seedsnipes is a loud and repeated many times call “kli-kli-kli”.
They usually utter it when alarmed.
Antarctic chicken seedsnipes are strict vegetarians, which is unusual for the
Charadriiformes order, but typical for the seedsnipes family. Their food includes
the vegetative parts of plants – stems, leaves and buds, as well as flowers,
seeds and fruits.
In Antarctica, the only enemies of chicken seedsnipes are birds of prey – local
species of buzzard and caracara.
Chicks are much more vulnerable: they can fall victim to large grackles,
skuas, seagulls and even predatory
plants.
Chicken seedsnipes are monogamous: pairs are formed at wintering sites and exist
for one or more seasons. These birds begin nesting shortly after the returning
from wintering place, in the middle of the Antarctic spring – late September
and early October. They prefer to nest on dry hills, in separate pairs. The
nest is a simple hole in the ground, sometimes with poor plant lining. There
are 2-3 eggs of spotted coloring in the clutch. The male and female incubate
them for about a month. Chicks are precocial; soon after hatching they are able
to walk and feed themselves. In the first days of their life, insects play an
important role in their diet; only after a few weeks, the chicks switch to a
vegetarian diet of adult birds. About a month after hatching, they begin to
fly.
Soon after the juveniles take wing, the time of migrations begins for chicken
seedsnipes. Family groups wander the Antarctic, gradually uniting into flocks
and moving to the west of the continent. By the end of the Antarctic autumn,
flocks of chicken seedsnipes gather on the Antarctic Peninsula, where they fatten
up before flying across the Drake Strait. Having overcome this water barrier,
the birds fly to the plains of northern Patagonia, where they spend the winter.
They have a lot more enemies here than in Antarctica: these are numerous predatory
mammals and birds. To protect against them, chicken seedsnipes keep in large
flocks throughout the wintering period.
The lifespan of Antarctic chicken seedsnipes is up to 20 years.
This bird species was discovered by Simon, the forum member.
Plain
snipe or Nocturnal snipe (Nyctogallinago praticola)
Order: Charadriiforms (Charadriiformes)
Family: Sandpipers (Scolopacidae)
Habitat: plains of North America, migrating to South American pampas.
In human epoch numerous species of shorebirds had suffered due to draining of
wetlands and overhunting. Many species had become extinct, local forms in particular.
Other species survived due to their life in places non favorable for human activity
because of climatic conditions and features of the landscape – mainly in wetlands
and polar and subpolar regions. After the disappearance of humankind descendants
of survived species began active speciation and developed some new habitats.
One of these new species is plain snipe, the completely terrestrial species
of snipe from North America.
Living in the plains and grasslands of North America, plain snipe is a descendant
of the Wilson’s snipe (Gallinago delicata), but unlike their ancestors these
birds are completely terrestrial and nocturnal. Adults are 26-30 cm in length
with a wingspan of 45-50 cm. They have long legs to run through the vegetation
and a long beak to catch their main prey, insects and spiders. Also these birds
eat snails and earthworms.
Feathering coloration at this bird is cryptic: background color is straw-yellow
with thin brown cross strips on each feather. Tail and wing feathers have wider
cross strips with larger spaces between them. There is an individual variability
in coloration from lighter to darker shade of colors. The chest and sides vary
from yellowish white to rich tans. The nape of the head is black, with three
or four crossbars of deep buff or rufous color.
Eyes of this bird are adapted to the nocturnal life, being bigger and having
a more developed tapetum lucidum – that’s why its eyes shine in darkness like
at all nocturnal animals. During the daytime, these birds hide in tall grass
and bushes. Their eyes give them almost complete circular field of view. The
hearing of the plain snipe is good, allowing it to hear movements of insects
in the grass.
They comunicate with each other using loud calls. If a predator approaches,
they fly emitting loud calls to alert the others of the species.
Their mating season starts in the spring. During the night male tries to attract
females doing a serie of calls in the ground, after it he takes off and flies
from 50 to 100 yards into the air. He descends, zigzagging and banking while
singing a liquid, chirping song. This high spiralling flight produces a melodious
twittering sound as air rushes through the male’s outer primary wing feathers.
Females are attracted by the male display. After the mating, the female will
be the only responsible for making the nest – a shallow and rudimentary one
right on the ground with only some dead leaves and twigs as a litter. She lays
four eggs and the incubation lasts 20-25 days.
The down-covered young are precocial and leave the nest within a few hours of
hatching. The female broods her young and feeds them. When threatened, the fledglings
usually take cover and remain motionless, attempting to escape detection by
relying on their cryptic coloration. Plain snipe fledglings begin hunting for
insects when they have a few days after hatching. They develop quickly and can
make short flights after two weeks, can fly fairly well at three weeks, and
are independent after about five weeks. Plain snipe eats mostly insects, but
can eat some plant materials (small fruits and seeds). In the winter they migrate
to the plains of South America, flying at night. They avoid crossing Panama
Passage and fly to the east along the coast of Gulf of Mexico. During their
migration, they stop at the island of Great Antigua to have a rest, and then
fly along the coast of South America, bypassing tropical forests of northern
part of the continent.
Sexual maturity comes at the age of 10-11 month; life expectancy does not exceed
8 years.
This bird was discovered by João Vitor Coutinho, Brazil.
Forest
numenapteryx (Numenapteryx sylvaticus)
Order: Charadriiformes (Charadriiformes)
Family: Sandpipers (Scolopacidae)
Habitat: Eonesia, Burotu Islands, tropical forests.
Burotu Islands appeared on the map of the Earth after the human epoch, so the
fauna of this archipelago of Eonesia is typically insular, without descendants
of human-introduced species. It was formed from species that settled on the
archipelago naturally – birds, reptiles and invertebrates. The absence of predators
on the islands allowed the appearance of flightless bird species. Flight is
an ability that imposes too much stress and restrictions on physiology, so birds
refuse this privilege as soon as such an opportunity appears. The isolated remote
islands of Burotu are one of those places.
Several species of flightless birds inhabit the forests of Burotu, occupying
different ecological niches. One of them is a large forest numenapteryx, a flightless
sandpiper. This bird is a descendant of one of the widespread species of curlews
(genus Numenius) from the Northern Hemisphere. The most likely way for these
species to appear in the fauna of remote Pacific islands was the delay of birds
at wintering sites during migration. Gradually, a separate settled bird population
was formed from the ancestral species, and their descendants moved into the
forests and quickly lost the ability to fly.
In its appearance, numenapteryx resembles the New Zealand kiwis (Apteryx) of
the human era, but has a slimmer constitution. The height of an adult bird of
this species is about 40-45 cm, weight up to 2 kilograms (the female is larger
than the male). A very characteristic feature of the appearance of this bird
is a long beak bent downwards. The tip of the beak is equipped with a significant
number of receptors and allows it to search for prey in soft soil. The bird
feeds mainly on young land crabs, insects and soft tree fruits fallen to the
ground. Sticking its beak into the soft forest soil, the bird searches for invertebrate
burrows and digs them out with its paws, like a chicken. Numenapteryx takes
small prey with its beak directly from the ground.
The plumage of the bird bears characteristic features of flightless birds. It
is very soft; the feathers in the wings and tail are shortened. The background
color is reddish; the feathers are covered with thin dark cross ripples. There
are feathers with black tips on the crown, nape, back of the neck and back.
The wings are very poorly developed; the primary feathers are no more than 5
centimeters long. The elements of the wings retain mobility: the wings are used
for courtship display.
This bird species has long well-developed legs – numenapteryx is able to run
fast, often wanders through shallow sea waters in search of prey and can dig
up the ground. The color of the skin of the legs is grayish-yellow; the legs
of a female ready for breeding have a bright yellow color.
Due to living in a tropical climate, the seasonality of nesting in this species
is not expressed. The marital relationship in numenapteryx is clear polyandry.
During the year, the female meets with different males and makes up to 3 clutches.
The female also performs a courtship display. Calling the male, she emits long
“bubbling” whistles. The displaying female keeps in places where sunlight breaks
through the crowns of trees. Usually, several male applicants gather for the
female’s voice and arrange bloodless duels among themselves – one male drives
all the others away from the female. At this time, the female is displaying,
flapping rudimentary wings intensively and raising her beak up. After the formation
of a breeding pair, the female and male jointly make a nest – a simple hole
in the ground, in bushes or among large herbaceous plants.
The rate of reproduction of this bird is slow: there are only one or two eggs
in the clutch. The female does not take care of the offspring: the male exclusively
incubates and takes care to the chicks. The chicks have a longitudinally striped
coloration and black legs. The juvenile color of the plumage is also longitudinally
striped, but only three stripes remain – along the spine and on the sides of
the head and neck. With age, the color of the legs lightens.
Young birds grow slowly, reaching sexual maturity only in the third year of
life. Life expectancy reaches 20 years.
Stork-godwit
(Ardelimosa palustris)
Order: Charadriiformes (Charadriiformes)
Family: Sandpipers (Scolopacidae)
Habitat: marshy areas of Northern and Central Europe.
In Holocene, shorebirds were seriously affected by the drainage of swamps and
overhunting. Among such birds there were also godwits – several of their species
became extremely rare and became extinct in the human era. After the human disappearance,
their populations began to recover, and at this time the second blow to them
was inflicted by the ice age, which significantly reduced and fragmented the
ranges of these birds. However, after the retreat of the glacier, the godwits
regained their former species diversity. Their descendants became longer-legged,
somewhat resembling the early stages of the evolution of crails.
The central representative of this group in Neocene is the stork-godwit, a descendant
of the black-tailed godwit (Limosa limosa).
The body length of this species is 55 cm, wingspan 85 cm, weight up to 550 g.
The beak is long and straight, dark gray in color. The background color of the
plumage in different populations and color morphs varies from beige-brown to
gray-brown, always with a pattern of darker spots. The sides of the head lack
any pattern; a strip of narrow longitudinal specks (a dark stroke along the
rachis of each feather) stretches from the forehead to the back. Separate feathers
with a thin longitudinal stroke in the middle are scattered on the chest and
belly. The back and wing coverts are dark with a light border forming a scaly
pattern. The primary feathers are background colored with a dark area at the
base of the vane. The undertail is bright red. Sexual dimorphism is expressed
in the plumage color: females are colored duller. The legs are pink-red.
Birds can form clusters, especially in forage-rich areas of swamps, flood plains,
lake shores and banks of slowly flowing rivers. Outside the breeding season,
birds are tolerant of each other and form flocks of 10-20 ones. The basis of
the diet consists of small aquatic animals – small fish, tadpoles, frogs and
invertebrates. Stork-godwit is a migratory bird; it winters in southern Europe
along the banks of rivers, part of the population flies to Africa, to the basin
of Saharan Nile. A small number of birds overwinter on the southern coast of
Fourseas.
The breeding season lasts from April to mid-May. Adult birds form pairs for
life. During the mating season, they perform a courtship ritual: they vocalize,
stamp their feet together and chase each other. Young and widowed birds carry
sparrings for a female, accompanied by dancing and chasing each other. The nest
is a shallow hole in a flat, dry place; the nest is lined with grass. There
are 4 eggs with a mottled shell of camouflage color in the clutch, which both
parents incubate for 30 days – the female usually incubates during the day.
When predators appear, the male or both birds run in front of them, simulating
the wound and trying to lead them away from the nest. Chicks hatch well developed
and can follow their parents as early as 40 minutes after hatching. Young birds
take wing at the age of 35 days and begin to train hard, preparing for the migration.
The female flies away with the young first, the males are delayed with migration
for several days. The young of the stork-godwit becomes independent already
in the winter. Sexual maturity occurs at the age of 2 years, life expectancy
is up to 25 years.
This bird species was discovered by Nick, the forum member.
Yezherinis
(Tringa yezherinis)
Order: Charadriiformes (Charadriiformes)
Family: Sandpipers (Scolopacidae)
Habitat: Northern and Central Europe, flood plains and shores of freshwater
reservoirs.
Populations of sandpipers, as well as other shorebirds, suffered significant
damage in the human era. Swamps were drained for economic needs, reservoirs
were polluted, and a number of species were objects of hunting. But some small
species, having suffered some damage, managed to survive the anthropogenic crisis
and the ice age. One of such lucky species was common redshank (Tringa totanus),
a small European species of shanks. After the age of man and the ice age, it
literally went through a “bottleneck”, when the surviving populations were barely
enough for the species to avoid degeneration. Later, it restored the number
and gave rise to several small species, one of which is yezherinis (the name
means the Lithuanian lake spirit).
The body length of an adult is up to 40 cm, the wingspan is 65 cm, and the weight
is about 260 g. It is a long-legged bird with a short straight beak. The upper
side of the body is brown with black and gray mottling, and the belly is white.
The primary feathers are white, clearly visible in flight. The beak is black,
it is visually continued by a black stripe stretching across the eye and ending
behind the ear opening. It is shaded from below by a narrow white stripe. The
color of the legs is very bright and characteristic, crimson-pink. Yezherinis
feeds on flood plains and shallow waters of lakes and slow-flowing rivers, it
gathers in groups of up to 25 individuals for feeding. The bird is zoophagous
and feeds on insects, mollusks, worms and crustaceans, sometimes pecks the eggs
of fish and amphibians, as well as tadpoles. During feeding, the bird is silent;
when worried, it utters a loud call “tiu-tiu-tiu”, repeated many times. This
species is migratory; yezherinis winters in North Africa (the basin of Saharan
Nile) and on Zinj Land, where it leads the same lifestyle.
The mating season is quite long – from the end of April to the beginning of
June. Pairs are formed for one season. The beginning of breeding in spring is
preceded by courtship flights, during which the male emits a very melodious
whistle. The nest is simple – it’s just a hole in the sand, lightly lined with
grass. There are usually 4 pointed eggs in the clutch, which both parents incubate
for 30 days. In case of the destruction of the clutch, the couple quickly makes
the next one and manages to grow a new brood before autumn. The chicks hatch
quite developed and immediately begin to follow their parents and feed by themselves.
At 26 days, young birds take wing, and by autumn they are completely independent.
Sexual maturity comes the next year; life expectancy is up to 15 years. Hwever,
few individuals live up to this age, more often birds older than 10 years are
eaten by predators.
This bird speies was discovered by Nick, the forum member.
Running
sandpiper (Calidris dimorphus)
Order: Charadriiformes (Charadriiformes)
Family: Sandpipers (Scolopacidae)
Habitat: North American tundra, a separate population on Mishe-Nama Lake; wintering
in southern North America.
In Neocene, the geography of North America partially changed: the Beringian
Isthmus connected it with Asia, turning the Arctic Ocean into a “refrigerator”
of the Northern Hemisphere, and Hudson Bay closed and turned into a brackish
Mishe-Nama Lake. It is populated very densely, by former marine inhabitants
among others, as well as by those who moved here directly from fresh waters.
Running sandpiper, one of the numerous species of waders that the fauna of North
America is rich in, belongs to the natives of fresh waters. The ancestor of
this species is most likely the rock sandpiper (Calidris ptilocnemis), which
survived in the tundra zone during the ice age.
Running sandpiper is a peculiar bird species, which representatives clearly
fall into two dimensional categories. In length, this bird reaches from 27 to
40 cm, not counting the beak; the length of the beak is slightly less than the
length of the head, it is straight and pointed; the wingspan is from 40 to 70
cm. Such a large variation in size is due to the fact that this species has
a very wide range, and individuals from different populations have different
sizes. The population from Mishe-Nama Lake is remarkable in the smallest size
and is relatively isolated from others, and birds from the north are larger
and form the unite large population, little variable in size. In all populations,
the female is larger than the male.
This bird has long legs with small webbing and claws, and a rather short tail.
The head is relatively large in relation to the body, like in other waders.
The beak is thin, straight and pointed. The background color is reddish-brown,
and black, white and yellow mottlings are scattered on the upper side of the
body; the lower part of the body is lighter and lacks mottling. The eyes are
black, the tail and loin are also black, the legs and beak are dark gray.
Extensive sandy beaches are suitable for running sandpiper, and this species
does not occur on stone or pebbled areas. Usually these birds live alone or
in small groups, and during feeding they run along the sand, mincing their feet
quickly. The bird’s food consists of worms, mollusks and crustaceans, which
running sandpiper digs out of the sand with its beak. Less often, running sandpiper
eats fish eggs and fry. On Mish-Nama Lake, sandpipers from the local dwarf population
can land on dwarf gannetwhales
and clean their plumage from parasites. The danger to running sandpipers themselves
is represented by large fish, snakes, birds of prey and predatory mammals. The
bird quickly runs away from them, taking off only at the moment of extreme danger.
In the spring, courtship displays begin. Males gather on sandy beaches and display
themselves, bouncing and fluttering their wings in flight, accompanied by a
series of short whistles. The female chooses a male during the display, and
the pair leaves the lek. The pair is formed for one nesting season. The female
makes a nest in the sand, lays 4 eggs and incubates them and the male protects
the territory and distracts predators while the female hides. By the end of
incubation, the male increasingly replaces the female on the nest, and eventually
completes incubation himself. The eggshells are beige with numerous randomly
scattered brown specks, which mask them from the eyes of predators. After three
weeks, the chicks hatch. They dry out quickly and immediately leave the nest,
following the male. The female does not participate in the rearing of chicks.
Young birds feed on small invertebrates. After a month, the chicks are already
fully fledged, and at the age of 5 weeks they already are able to fly. By the
end of summer, families break up and the birds are fattened before the migration.
Running sandpipers of northern populations gather in huge flocks for migrations
and fly south across the continent along the beds of large rivers. Their wintering
grounds are the Mississippi Delta, Florida and the Caribbean Islands. The population
from Mishe-Nama Lake flies south along the Atlantic coast and winters in the
swamps of Florida.
Sexual maturity comes the next year; life expectancy is up to 15 years.
This bird species was discovered by Mamont, the forum member.
Masked
sitte (Sittus larvatus)
Order: Charadriiformes (Charadriiformes)
Family: Sandpipers (Scolopacidae)
Habitat: northern Eurasia from Ireland to Beringia and from tundra to Ladoga
in Europe and to the borders of the taiga and Siberian wetlands in Asia.
Birds of wetlands and coasts of reservoirs have suffered significantly due to
human fault – mainly due to hunting and habitat destruction. The northern snipe
species had a particularly hard time. Some of the species that survived the
ice age had to retreat to the south or become extinct during the Holocene-Neocene
glaciation. But those species that survived the ecological crisis and the ice
age have become more diverse. One of the surviving species was the Eurasian
whimbrel (Numenius phaeopus) widespread in the human epoch, which became the
ancestor of a number of bird species. One of its descendants is masked sitte
(the name means a ghost at the Sami).
This species is a fairly large sandpiper: body length 55 cm, wingspan up to
1 m, weight about 500 g. The plumage color is pale brown with numerous dark
longitudinal strokes, more dense on the back. On the wing coverts, the strokes
turn into broad spots, which make the wings look darker. On the front of the
head there is a characteristic black “mask”; the beak is long, thin and curved
downwards. In flight, white secondary feathers are visible. The color of the
legs is yellow. The call sounds like a long whistle.
The bird lives and feeds in swamps, flood plains, lake shores and riverbanks.
Masked sitte feeds on insects, their larvae, worms, mollusks and small crustaceans.
During feeding, the birds stay alone or in small groups. It is a migratory species,
wintering on the Atlantic coast of Europe, in North Africa, Zinj Land, Hindustan
and Southeast Asia. During the flights, birds try to circumvent high mountain
ranges.
The mating season begins in late spring, at the end of April – May. Attracting
females, males vocalize and flap their wings. When the rivalry becomes especially
active, the birds fly up and kick each other in flight. Pairs are formed for
one season. The nest is built in a flat depression on the ground, without litter.
In the clutch of masked sitte there are four eggs with a gray shell, speckled
with dark spots of various sizes. The male and female incubate, replacing each
other, for a total of 25 days. The chicks are well developed and, having dried
out, follow the mother. At the age of one month, they become independent. Sexual
maturity occurs at the age of one year, life expectancy is up to 19 years.
This bird speies was discovered by Nick, the forum member.
“Sea
vulture”, griffon skua (Thalassogryps nudiceps)
Order: Charadriiformes (Charadriiformes), suborder Gulls (Lari)
Family: Skuas (Stercorariidae)
Habitat: North Atlantic, from Iceland up to New Azora, coasts of Old and New
World in temperate and subtropical areas.
In human epoch gulls were one of prospering groups of aquatic birds. The pledge
of their success in human epoch was rather fast development (as opposed to them
the nestling of albatross, for example, developed, staying in nest till about
9 months), and adaptable behaviour, permitting to adapt to new conditions of
existence. In human epoch some species of gulls became usual inhabitants of
cities and dumps, and also of ports. Ease of adaptation and wide spreading had
helped these birds to survive in human epoch having suffered the minimal damage,
and in Neocene they have successfully continued the existence.
In Neocene one group of gulls, skuas, had reached significant evolutionary success.
Among them the species replaced procelariiform birds, which number had decreased
at the boundary of Holocene and Neocene, had appeared. And one species had turned
to true predator successfully competing to sea
eagleraven at the coast of northern seas.
In North Atlantic the usual inhabitant of sea coasts is the griffon skua also
named “sea vulture”. It is rather large carnivorous bird: it weighs up to 3
kg at wingspan of about 2 meters. Feathering of this bird is dark brown, and
only the tips of wings are white with several black primary feathers. Such contrast
marks are used for the bloodless solving of intraspecific conflicts: conflicting
birds simply stretch wings and display themselves to the contender. This way
they estimate the visual size and force of each other, not entering the combat.
At this time both birds utter their unpleasant shrill call.
The name of bird emphasizes both its appearance, and lifestyle. At griffon skua
the forward part of head and neck is covered with naked grafite-grey skin without
feathers. But feathers on top and sides of head are lengthened, forming the
kind of horizontally stretched crest. Raising it, the bird expresses its own
emotional condition and displays signals to relatives – it is very important
to avoid intraspecific conflicts which frequently happen at these birds.
Griffon skua eats carrion of various kinds. Usually it flies above the sea,
similarly to albatross, and picks up by beak dead or weakened fish and herring
squids from surface of water. Besides griffon skuas eat dead giant
sea birds
at coasts, and attack their nestlings in colonies. Groups of birds of this species
patrol coast in searches of carrion cast ashore. After storm big flights of
griffon skuas gather on carcasses of cachalot sharks
and other large sea creatures. Sharp beak hook-like bent tip helps bird to tear
even thick skin of shark.
Becoming a seashore predator, griffon skua has lost some skills which its ancestors
had. Palamas on paws of this bird are reduced, and griffon skua never gets on
water. In case of necessity (for example to pick up prey) it can only “run”
some distance on water against wind, fast splashing on water surface and having
stretched wings to create carrying power. Griffon skua is not able to dive at
all, therefore small seashore birds easily escape from this predator, hiding
under water.
This bird nests in pairs, not forming colonies, and flocks gather only on large
prey. Flocks of these birds are temporary, their structure is casual and in
them well defined hierarchy is not present. While it is a lot of food, birds
do not pay attention to relatives and tolerantly behave to their presence. If
prey is small, between feeding birds conflicts including display of force flash.
Griffon skua forms pairs only for one nesting season. It nests on the ground,
choosing for breeding small islands where there are no terrestrial predators.
In clutch of this species there is one egg, and only in favorable years it may
be two ones. Nestlings hatch in down, and at them there is already naked forward
part of head. First days they are inactive and sit in nest, reacting only on
approach of parents. From five-day age they start to walk and actively to elicit
food at parents, competing with each other. At lack of food stronger nestling
may kill weaker one and eat him.
At monthly age at the nestling growth of feathers on wings and in tail begins,
and three-monthly young bird is already fully fledged. Juvenile colouring at
griffon skua differs from adult one – at young birds there is white plumage
on head, and naked skin is black. At the age of five months young bird becomes
completely independent. At one-year-old age the plumage changes to characteristic
for adult birds one. At the age of one year young birds become able to nesting.
Griffon skua makes seasonal migrations along ocean coast. In spring this bird
flies far to the north and nests. In autumn adult birds and young growth migrate
to the south.
The idea about existence of this species was proposed by Simon, the forum member.
Narrow-beaked
warrior of Boreus (Boreofregata acutirostra)
Order: Charadriiformes (Charadriiformes), suborder Gulls (Lari)
Family: Skuas (Stercorariidae)
Habitat: coast of Arctic Ocean.

Picture by Alexander Smyslov
Summer in northern polar latitudes is a continuous polar day.
Sun shines all day and night, and it promotes development of plenty of plankton
in top layer of oceanic water. Abundance of plankton involves unnumerable schools
of fishes and cephalopods, which, in turn, are a food for various carnivores.
During the polar summer above ocean large long-winged birds appear. They promptly
sweep above a surface of water, and by dexterous movement snatch fishes and
squids from water. These birds are very similar to man-o-war birds (Fregata)
of Holocene epoch, but such similarity is only external. These birds are descendants
of skuas (Stercorarius), gulls known for predating bents. They make a separate
genus of birds – warriors of Boreus (Boreus in ancient Greek mythology is a
god of northern wind; this name emphasizes features of area of these birds in
comparison with true man-o-war birds, which had dyed out at the boundary of
Holocene and Neocene).
The most widespread species of these birds is narrow-beaked warrior of Boreus,
the long-winged predatory bird living at the coast of Arctic Ocean in flights
numbering up to one hundred of individuals. Feature of this species is long
beak compressed from sides and having hooked tip. By such beak bird dexterously
seizes prey from surface of water.
All warriors of Boreus are birds having magnificent flight abilities. Wings
of narrow-beaked warrior of Boreus are very long; their wingspan makes about
200 cm. The tail of bird is V-like doubled and is a little similar to swallow’s
tail. Such tail shape provides an excellent maneuverability to this bird. Despite
of large wingspan, narrow-beaked warrior of Boreus weighs not so much: no more
than one and a half kilograms.
The plumage of this bird has dark brown colouring, and only under tail and in
the bottom part of stomach it has ochre-red color. Primarily feathers are black
with metal shine. Colouring of beak contrasts with background of dark plumage
as a bright spot – at sexually mature birds it is bright yellow with black spot
on the tip. At not sexually mature birds beak has light brown color.
Narrow-beaked warrior of Boreus eats sea animals – fishes, squids and large
swimming crustaceans. It never alights on water, because has no swimming membranes
and is not able to swim. As against true man-o-war birds, at warriors of Boreus
there is an advanced coccygeal gland, due to which the plumage of bird has water-repellent
properties. Besides narrow-beaked warrior of Boreus is the masterly air hunter:
it also eats small sea and ground birds. Having gathered full speed, this bird
hurriedly forces down small birds by impact of beak, at once picks up them right
in air, and swallows entirely. Narrow-beaked warrior of Boreus hunts not only
in coastal zone of seas, but also above tundra and bushes. However, this bird
does not fly in forest and does not fly low above bushes, being afraid to injure
wings. It partly rescues small birds from this predator: they wait occurrence
of such air hunter, having hidden in bushes. Large species of warriors of Boreus
frequently get food using cleptoparasitism – they attack sea birds of other
species had caught prey: gannetwhales
and eagleravens. Striking
to them impacts by beak and wings, warriors of Boreus compel victim to let out
prey, and pick it on the spot.
As against true man-o-war birds, warriors of Boreus can walk on the ground,
and even fly up from flat surface against a wind; but they never perch on trees.
Warriors of Boreus nest in pairs at some distance from each other. Only right
after migration or before back migration they gather in big flocks. During the
nesting period birds occupy the certain territories of about one hundred square
meters, which borders are strictly protected. The alien bird has been late too
long at the territory of pair risks to cause aggression of owners of this territory.
In the centre of nested territory the nest is located; usually birds arrange
it in shelter – among stones or under bushes. The reason of it is the vunerability
of warriors of Boreus to frequent attacks of large predatory birds – sea eagleravens.
Approaching of this predator causes instant common alarm, and warriors of Boreus
surround predator by large flocks. They attack eagleraven, striking to it impacts
by beak and wings, and compel it to leave vicinities of nesting area.
In clutch of these birds it may be two large eggs with spotty brown shell. They
are hatched mainly by female, and male feeds her. The hatching lasts till about
43 days. Nestlings of warrior of Boreus hatch covered with down and having opened
eyes. They have rather long wings, and sometimes nestlings support on them at
walking. They eat fish and other sea animals, and later, from bi-monthly age,
start to eat entirely small birds brought by parents. To the autumn young birds
get juvenile plumage, and at the second year of life their colouring changes
to adult one.
Warriors of Boreus belong to number of migrating birds. In summer narrow-beaked
warrior of Boreus is fattened in rich by forage polar waters; for winter it
departs to the south. Western populations migrate along the Atlantic coast of
Europe, reaching up to Canary Islands; some individuals reach coast of New Azora.
Populations from Northern Asia migrate along more difficult route: they fly
up to Fourseas, and then make non-stop flight up to coast of Arabia and even
up to the north of Zinj Land. East Asian populations migrate via Beringia to
the north of Pacific Ocean and fly to the south up to Japan Islands.

Picture by Alexander Smyslov
At coasts of Pacific and Arctic oceans in North America the close species lives – pale-faced warrior of Boreus (Boreofregata pallidifrons). It differs from the Euroasian species in almost completely black plumage. Only the forward part of its head has white colouring, and beak is grey with black tip. By habit of life pale-faced warrior of Boreus is similar to narrow-beaked one, and at the boundary of areas of these species between birds of both species relations of antagonism are sharply expressed – birds of both species ravage nests of neighbours, kill their nestlings and attack single adult birds of other species.

Picture by Alexander Smyslov
At the Atlantic coast of North America one more species of this genus lives – smaller warrior of Boreus (Boreofregata parva). It is small species – its wingspan does not exceed 120 cm. Also for it grey plumage with brownish shade on covert feathers of wing and black primarily feathers are characteristic. This bird seldom attacks other birds, and prefers to hunt fish and squids far from coast. This species is widely settled in New World: it lives from the south of Greenland (where it is migrating species) up to Florida (where there are settled populations of this species). Some individuals reach Great Antigua.
Hakoakoa
(Sarcoscopus hakoakoa)
Order: Charadriiformes (Charadriiformes)
Family: Skuas (Stercorariidae)
Habitat: New Zealand, mountains and woodlands.
In the New Zealand of the human era, there were no specialized scavengers from
among vertebrates. But in the Neocene, when the archipelago acquired a megafauna
originating from introduced marsupials and ungulates, the situation changed.
Various New Zealand birds, such as corvids
and parrots, began to feed
sometimes on the corpses of large herbivores. But the most specialized type
of scavengers Ao-Tea-Roa does not belong to them, but comes from the skua famiy.
It is called hakoakoa – after the Maori name of the brown skua (Stercorarius
antarcticus), one of the possible ancestors of this bird.
Hakoakoa is a relatively large bird, comparable in size to an ordinary vulture:
the wingspan reaches 180 cm, and the weight is 2-2.5 kg. The limited resources
of the islands on which it lives do not allow this species to become larger.
The entire appearance of hakoakoa shows its deep specialization in feeding on
carrion: so, wide wings and a fan-shaped tail allow it to hover in the sky for
a long time, looking for food. The head and elongated neck are covered with
short gray down, and around the eyes and beak the skin is bare, colored light
gray – it is an adaptation that prevents the plumage from getting dirty when
the bird is feeding. The beak, colored black, is disproportionately large and
thick, with a sharp hook at the tip – due to it hakoakoa is able to cut open
the corpses of animals even with a thick skin. The plumage is colored brown,
with a scaly pattern on the back and wing coverts and a lighter area on the
belly; the iris of the eye is black. The legs are dark gray in color – they
lack palamas, because the bird (unlike another descendant of the skua, the griffon
skua from the Northern Hemisphere) has completely
interrupted its connection with water. The coloration of young individuals is
distinguished by light mottling all over the body.
Hakoakoas inhabit various landscapes of New Zealand, avoiding only dense forests.
They are most numerous in woodlands and in the mountains. These birds settle
where there are sufficiently large populations of herbivores that can provide
them with food. These birds search for food by soaring in the sky and carefully
studying the landscape. In part, the sense of smell helps in the search for
carrion – in hakoakoa it is somewhat better developed than in birds in general.
At the carcass of the dead animal, these birds gather in large groups, whose
members periodically arrange fights for a place in the hierarchy and the right
to feed first. In addition to their relatives, hakoakoa compete for food with
false ravens and eagle keas, as well as with mammals – but thanks to their quarrelsome
nature and courage, they are able to defend the carrion found and even drive
away competitors. The gastric juice of hakoakoa is very corrosive, which allows
it to digest carrion without harm to health. After feeding, these birds sit
in the sun rays for a long time, spreading their wings – ultraviolet radiation
kills pathogens. Mountain populations have invented several additional ways
of obtaining food – they can drop young mountain
herbivores from rocks and throw bones from a great height, then peck out
the marrow from them (like Holocene lammergeiers).
The transition to land life has led to changes in nesting behavior – due to
their size and the presence of predatory mammals
in the fauna of Ao-Tea-Roa, hakoakoas cannot afford to nest on flat ground.
Populations from mountainous areas build lax nests of dry grass and down on
ledges of rocks. Birds from the plains are forced to nest in trees – they either
occupy old nests of large birds such as false ravens and herons, or build lax
structures of branches on tree tops. The nesting season of hakoakoa is timed
to the spring of the Southern Hemisphere – the time when ungulates give birth
to cubs. In the nest of these birds there is one olive egg (sometimes two),
which is incubated by both parents. The male and female also feed the chick
together. Hakoakoas jealously guard their nests from predators and ravagers,
as well as from peaceful herbivores that happen to be nearby. After the juvenile
takes wing (after about 2-3 months), the couple breaks up – these birds become
attached to each other and to the nesting site for only one season, although
it is highly likely that the next year the same birds can form breeding pairs.
Young birds become ready to breed at the age of two years, and the total life
expectancy can be up to 35 years.
This bird species was discovered by Simon, the forum member.
Pilot
bird (Postcepphus navigator)
Order: Charadriiformes (Charadriiformes), suborder Auks (Alcae)
Family: Auks (Alcidae)
Habitat: Northern Atlantic.
At the end of human epoch sea ecosystems had suffered the big loss from human
economic activity. Overfishing for maintenance with feed of huge human population,
hunting, and also pollution and destruction of habitats had resulted in reduction
of number of many sea animals. Large vertebrates – cetaceans and pinnipeds had
suffered from it first of all. Their populations appeared too small and genetically
impoverished in order to be restored after disappearance of mankind. Therefore
in Neocene seas of northern and temperate latitudes are ruled by giant flightless
birds – gannetwhales and giant
loons of several species.
But side by side with them birds of other species, smaller and more numerous,
live.
When huge gannetwhale dives, at once some small birds with black plumage and
white wings direct in depth following it. They keep near to the feathered giant,
and move under water, rowing by paws and almost pressing to the body of gannetwhale.
For this feature of behaviour they received the name “pilot birds” by analogy
to pilot fishes (Naucrates ductor) which accompanied in the same way with sharks
in Holocene epoch.
Pilot bird is a descendant of short-billed guillemot (Cepphus grylle), the bird
of Atlantic, very characteristic and numerous in the past epoch. It has kept
ability to fly, and nests on coastal rocks in numerous noisy colonies. Its close
relative in Pacific Ocean is very large and completely flightless bird – ocean
hatchetbill (Megacepphus involans), the descendant of Pacific species
Cepphus crabro.
Pilot bird has kept similarity to ancestral species, but differs in larger size
– it weighs up to 700 grammes. This species has straight strong beak of black
color and wide red webby paws, with which help bird quickly swims under water.
This bird is too small to dive deep, but it successfully hunts, using the neighbourhood
of large sea birds. When gannetwhale dives, the pilot bird keeps in layer of
water, directly adjoining to the body of the giant and by that saves more, than
half of efforts necessary for swimming. It rows by paws only a little in order
not to lag behind large bird. It accompanies with huge sea birds during their
hunting to catch small fishes and squids frighten by them – when gannetwhale
searches for prey in thickets of brown algae, pilot birds literally hover near
it, picking up fishes to which the large bird does not pay attention. Pilot
bird also eats the rests of prey of giant, and sometimes steals a part of prey
literally “from under the nose” of gannetwhale. But it should be cautious: sometimes
gannetwhales seize these birds instead of fishes.
Pilot birds lead social way of life. They live and hunt fish by big flocks,
moving for fishing together with gannetwhales in the morning. Colonies of these
birds settle down at coast near to congestions of gannetwhales and plesioloons.
Pilot birds nest under stones, digging by beaks and paws holes up to two meters
deep. The nest is covered with small amount of dry grass and seaweed gathered
at the sea coast. The entrance to the hole usually is very narrow, and also
is made closely to big stone – this way it is more difficult to predators to
reach nesting birds. On islands remoted from continent coast, lack of ground
predators, holes of birds are not so deep. The colony totals over hundred of
birds, and at the continent pilot birds gather to many thousands colonies.
Nesting pair at these birds is kept during several seasons of nesting in succession.
They start nesting early in spring, still before the first gannetwhales will
appear at coast. Before the arrival of these birds pilot birds can feed independently,
but in this case search of forage takes more forces, and they can not dive as
deep, as at joint hunting with large birds.
Before egg laying even partners from already existing pair display a similarity
of courtship ritual: male chases female on the ground, accompanying the run
for her with loud cries. After that feeding the female on belched fish follows,
and next is transfer from beak to beak of symbolical “gift” – piece of seaweed
or feather. In clutch it usually may be two eggs hatching alternately by both
birds of pair. The incubation of eggs comes to an end, when gannetwhales already
appear at coast, and it is much easier to parent birds to catch food for nestlings,
accompanying with these huge birds. Nestlings are covered with rich down. They
remain in hole within 3 months, and strongly get fat, getting plentiful food
brought by parents: leaving a nest, young birds hardly squeeze through an entrance
of hole. Parents finish feeding of young birds within one week after their leaving
of nest.
Young birds in juvenile plumage have black wings. Only at the second year of
life young birds become able to nesting. Then, after mew, they take a colouring
characteristic for adult birds.
This species of birds was discovered by Simon, the forum member.
Baltic
auk (Eualle baltica)
Order: Charadriiformes (Charadriiformes), suborder Auks (Alcae)
Family: Auks (Alcidae)
Habitat: Wenedian Lake and water bodies located around of it.

Picture by Alexander Smyslov
In Holocene epoch little auk (Alle alle), numerous and medium-sized
sea bird, had very wide area in Northern hemisphere. For this species incidental
migrations far to the south, out of boundaries of its usual area had been characteristic.
Due to the small size this species could go through crisis at the boundary of
Holocene and Neocene, having kept enough number of viable populations within
the borders of former area. In early Neocene descendants of little auk had restored
number and area characteristic for their ancestor. Due to dissociation populations
of little auk descendants differed from each other, and they could occupy different
ecological niches in northern seas. Ability to resettlement has permitted to
descendants of little auk to expand an area and to form viable populations in
new habitats, beyond Arctic and Subarctic regions. One population settled at
the coast of Wenedian Lake – large water body of the glacial origin formed at
the place of Baltic Sea. During the evolution it had turned to separate species
– Baltic auk.
Similarly to its ancestor, it is a small bird up to 150 grammes in weight, well
flying and capable to dive. In colouring Baltic auk sharply enough differs from
the ancestor which had black-and-white colouring rather usual for auks. The
feathering of Baltic auk is colored sandy and straw tone; belly is white-grey,
beak and paws are dark brown. In courtship dress the breast at birds of both
genders turns snow-white, but right after hatching start white feathers drop
out.
Baltic auk has small beak and wide cut of mouth. This bird eats various small
animals: lake plankton, larvae of insects and fish fry. This species has inherited
from the ancestor such feature of behaviour, as night feeding. Birds catch prey,
diving for it in water.
The main nesting places of Baltic auk are located in northern part of Wenedian
Lake where there is a plenty of rocks and the islets formed by glaciers. In
southern part of lake small colonies of these birds live on the islets far from
the coast and overgrown with cane. Birds nest in natural shelters between stones,
and in canes at absence of them. As against the ancestor formed the large colonies
totaling thousands of birds, Baltic auk nests in small colonies – up to 40 couples
of adult birds. Colonies of these birds are not similar to rookeries at the
coasts of Arctic region and Atlantic – Baltic auks try to behave silently and
secretively, and their colonies are difficult for finding in canes. Adult birds
of this species are settled, and only at freezing of lake more then usually
happens they make short migrations to edge of ice cover, permanently returning
to nesting places in spring.
The nest arranging at Baltic auk is limited only to the choice of ready natural
shelter. In clutch of this species there is only one egg; nesting begins in
early spring. The nestling hatches well advanced. It is covered with light brown
down with longitudinal black strips; at danger it hides among plants and freezes
extended vertically, as if a bittern. Parents bring it up while it will fledge
completely. Young birds differ from adults in monotonous coloring of feathering.
They become adult at the second year of life.
Young birds spend the first winter in the central part of lake, at the edge
of ice cover. Here they are well protected from predators and find a lot of
food like planktonic crustaceans. The main enemy of these birds is lake
monster perch (Gorgoperca territaris), fish reaching three-meter length.
Large adult fishes of this species can eat Baltic auk only casually, but younger
fishes are more mobile, and purposefully hunt these birds at shoaliness. Also
other inhabitants of Wenedian Lake – huge lake crackens
(Crackenastacus cataphractus) attack Baltic auks.
This species of birds was discovered by Simon, the forum member.
Moustached
auk (Postalle glaucus)
Order: Charadriiformes (Charadriiformes), suborder Auks (Alcae)
Family: Auks (Alcidae)
Habitat: Mishe-Nama Lake, the central part of lake.
Little auk (Alle alle) had been one of numerous species of sea birds of Holocene
epoch. In epoch of global “planktonic accident” the majority of species of sea
birds, especially large ones, had died out, or their number and area had been
reduced. Some species came in decline and were gradually superseded by the neighbours,
and others could restore number quickly and expand an area. Little auk belonged
to the number of such species. Its descendants were settled in the seas of northern
hemisphere, and had appeared even in Wenedian Lake.
One more species being the descendant of little auk had settled in North America,
in Mishe-Nama Lake.
The North American species, moustached auk, belongs to separate genus. It is
a larger species, rather than its ancestor, a pigeon-sized bird. It has kept
bicoloured feathering, characteristic for its ancestor, but black color was
gradually clarified: the top part of body of moustached auk is grayish-blue,
stomach and throat are white. Moustached auk has dense constitution, large head,
short neck and thickset trunk. This species is very good flyer; bird has short
peaked wings and short fanlike tail. Flight of moustached auk is linear and
very fast. Bird swims under water, using a mode of “underwater flight” with
the help of wings, and easily passes from one environment to another: having
gathered speed in air, birds fold wings and enter into water almost without
splashes, and from under water, having gathered speed, jump out in air, and
continue flight just as usual birds. Birds walk on the ground, having extended
body vertically and basing not only on toes, but also on tarsometatarsus. Legs
are webby, and birds are able to swim on water very good.
Beak of moustached auk is blunt and short, coloured black. However, it has remarkably
wide mouth cut – the corner of mouth is located behind back edge of an eye.
From upper and lower jaws, near edges of corneous cover of beak, thin elastic
bristles stick out. They represent the modified feathers lack of vanes, with
thin flexible shaft. This adaptation helps birds to get food. Moustached auk
feeds on planktonic crustaceans and other organisms living in top layer of water.
Diving under water, bird filters water and keeps prey in mouth with the help
of bristles. While bird does not feel presence of prey, its mouth is closed.
But, having felt the impact against bristles, bird slightly opens mouth and
catches prey. It is interesting, that these birds also eat tiny flying insects
– mosquitoes, caddis flies and May flies. They gather them in air the same way.
This species of birds avoids coastal zone, and beyond of nesting time almost
constantly keeps above water. Flocks of moustached auks keep above congestions
of plankton crustaceans and fish fry, have a rest on subjects floating in water,
and on floating ice floes in winter. This species appears at the coast of lake
very seldom – usually birds lose road to a fog, but right after it dissipates,
they leave coast. For winter northern populations migrate to nonfreezing southern
part of lake.
Moustached auk is monogamous. Birds of this species nest at the islands in central
part of lake, in colonies numbering up to one thousand of individuals. The nest
is arranged in hollows and cracks between stones. Birds simply drag therea litter
of dry grass and feathers. Female lays two eggs, and parents more often succeed
to bring up both nestlings. Hatching lasts till 22 days; both birds take part
in it. Nestling stays in nest till about 40 days, being strongly eaten off.
Leaving the nest, it weighs approximately 20 % more than any of its parents.
The first days the young bird swims on water surface and trains in flight and
food getting. Colouring of young bird differs from adult one only in dimmer
color. At the age of two years moustached auks become able to breed.
Imperial,
or wingless murre (Ampluria breviptera)
Order: Charadriiformes (Charadriiformes)
Family: Auks (Alcidae)
Habitat: North Pacific Ocean, islands off the coast of Beringia to the north
of the Big Kurils.

Picture by Lambert
Murres, which easily survived the human epoch, suffered seriously
at the boundary of the Holocene and Neocene, before the beginning of the ice
Age. Their food supply was undermined by the “planktonic catastrophe”, and only
few populations of both murre species, known in the human epoch, survived in
the estuaries of rivers south of the edge of the glacier. Over time, murres
restored their number again and managed to regain the lost territories. In due
course of evolution, a whole bunch of diverse species, inhabiting various territories
of the Pacific and Arctic Oceans, have emerged among them. The largest of them
is imperial murre, a descendant of the thick-billed murre (Uria lomvia).
This species is convergently similar to penguins and great auks of the human
era. The body length of an adult is 75-85 cm, and weight is up to 8 kg. Because
of such a large size, birds of this species have lost the ability to fly; their
wings have turned into flippers used for swimming. Legs are strong: birds easily
walk a distance of several tens of meters from the nest to the sea and climb
steep banks, keeping the body in an upright position. The color of the plumage
has retained the combination of colors characteristic for auks: the plumage
on the head, back and sides is black, and the chest is white, which makes the
bird less noticeable to enemies in the water. The skin on the legs is dark gray,
almost black; the palamas are well developed.
The beak of this species is massive, pointed and elongated; the edges of the
beak are finely serrated. The corneous cover of the beak is black with a large
white spot at the tip and a yellowish vertical stripe at the base. The upper
mandible is slightly curved downwards.
Imperial murre feeds on small fish and crustaceans, preferring to look for prey
in the water at the bottom, usually at a depth of up to 10 meters, although
it is capable of diving 20 meters or more. The manner of movement of these birds
under water is “flight”, characteristic of penguins of the human epoch.
This species, like its ancestor, lives in colonies, only migrating to the south
by winter: Asian populations – on the coast from the southern islands of Japan
to Taiwan, and American ones – on the coast of the continent to the latitude
of California Island, as well as on this island itself. Outside of the nesting
season, birds are found on the coast of the continent. They are quite aggressive
towards small predators: when they approach, birds start screeching loudly,
and if the predator does not retreat, they counterattack it and bite hard. Birds
can protect themselves from large predators by purposefully regurgitating half-digested
food into them, although they prefer to hide in the water.
Nesting of these birds takes place on rocky islands where there are no terrestrial
predators. The mating season begins in early May. Established couples that persist
for life jointly arrange a nest and give each other signs of attention, preening
the feathers with their beaks. Young birds vocalize synchronously, chase each
other in the water, and then the male presents the female with a gift – one
or more fish, passing them and simultaneously trying to mate while swimming
on the surface of the water. For nesting, birds prefer the company of relatives,
and the signal for laying eggs is a certain density of pairs on the nest - on
average, one pair per one and a half square meters of the nest.
The female lays, as a rule, one large oblong egg, colored gray with dark spots.
If the egg died at the beginning of incubation, the bird is able to lay another
one and bring offspring in this nesting season. Partners incubate the egg, replacing
each other and alternately feeding in the sea. Incubation lasts up to 45 days.
The chick is covered with gray down. It is fed by both parents, and it grows
very quickly: at the age of 5 weeks, the young bird fully fledges, and two weeks
later begins to fish, swimming with its parents. During the winter, the young
bird is already completely independent. Young birds differ from adults only
in the color of the beak: it is dull gray without a white tip. Sexual maturity
occurs in the third year of life, life expectancy is up to 30 years.
This bird species was discovered by Nick, the forum member.
Nocturnal
seagull (Nycticreagrus hirundo-nocturna)
Order: Charadriiformes (Charadriiformes)
Family: Gulls (Laridae)
Habitat: Galapagos Islands, coastal rocky areas.

Picture by Lambert
In Neocene Galapagos islands had remained as same, as they
were in human epoch: they are the volcanic archipelago completely isolated from
continent. People had introduced to these islands representatives of continental
fauna which had stand a number of local species on the edge of extinction. But
rigorous conditions of islands – absence of constant fresh water sources, hot
climate and the intensive volcanic activity – had resulted to extinction of
numerous intruders of human epoch. The stayed species had continued to evolve,
and among some really surprising creatures had appeared.
When on islands night falls, day time inhabitants hide in shelters and fall
asleep. But the movement above sea coast does not stop. At night on the ocean
surface near the coast huge luminous spots appear: shoals of luminous fishes
and squids numbering millions of individs emerge from depths for feeding. And
in flickering light of shoals of these creatures the bird's shadows sweep over
above waves. Bursts are heard from time to time, and one light is separated
from luminous shoal, shooting upwards to become dim for ever. The feast of these
birds is accompanied by quiet bitter cries. It proceeds till morning, but with
the first sun beams birds disappear and as if are dissolved, similarly to night
darkness.
The mysterious night hunter is a nocturnal seagull, the sea bird endemic for
Galapagos Islands. It is the descendant of one local seagull (Creagrus furcatus),
nested at Galapagos isles in human epoch. Its ancestor had also led a nocturnal
habit of life, but at the nocturnal seagull the specialization in this sense
is deeper. It is river seagull-sized bird distinguished by long narrow wings.
Top of body of this bird is grey with small black specks and irregular stripes.
This colouring helps this bird to hide: the nocturnal seagull is shy and completely
defenceless creature, whose strategy of survival is to be as imperceptable as
possible for probable predators. Overtaken unawares, this seagull hides into
a deepening on surface of coastal rock, blinks and stretches wings at its surfaces
that nothing gave out its presence. Bottom of body of this species is ash-grey.
The beak contrasts with the general colouring of feathering: it is black with
large white spot near the tip.
Among feelings at this bird sight is especially well advanced. The nocturnal
seagull is distinguished with huge eyes making in weight over half of gross
weight of head of this bird. The retina is covered by a layer of cells in which
guanine is deposed, therefore eyes of this seagull “shine” in darkness, as eyes
of cats. Eyes differ in great light sensitivity, therefore in the afternoon
the disturbed bird covers them with third eyelid serving as “solar glasses”.
This bird is active exclusively at night, and sleeps in day time. It hunts at
night luminous fishes and squids swimming out from depths, and catches them
by beak from water surface.
Galapagos nocturnal seagulls lodge in colonies on sites of coast where there
are caves or heaps of stones: they nest in shelters protected from sunlight.
They are rather numerous, but disappear in dark places at dawn, and their number
is difficult for counting. Nests of these birds are built of stones, or seagulls
choose convenient deepening on ledges of cave walls. Very seldom separate birds
arrange nests in forest, in hollows of local trees. In clutch there is only
one egg, both parents at night alternately (and in day time till the rest even
simultaneously) hatch it. Laying repeats about three times per one year, but
the most part of nestlings do not live up to adult condition: about the half
of posterity in colony perishes from various reasons including predators. Nocturnal
seagulls nest in close groups because it is a little places convenient for their
nesting. Nestlings are fed by both parents. Adult birds find out the posterity
by the voice among tens of another's nestlings. The white spot on the tip of
beak of nocturnal seagull during the nestling feeding has the same function,
as similar to it by arrangement red spot at the herring gull: the nestling,
pecking in it, gets food. White color is better swept up, than red, in twilight;
therefore the spot on the beak of seagull is white. Besides nocturnal seagull
can see colors in ultra-violet range so the white spot in addition seems luminous
to nestling: it reflects ultra-violet light.
Nocturnal seagulls are not migrating birds, and also do not fly from the coast
more, than few kilometers. Such isolation had permitted to evolve to this remarkable
species of gulls at Galapagos Islands.
This species had been discovered by Simon, the participant of forum.
Miru
(Pezolarus miru)
Order: Charadriiformes (Charadriiformes)
Family: Gulls (Laridae)
Habitat: Eonesia, Burotu Archipelago.

Initial picture by Alexey Tatarinov |
Burotu Archipelago is located in the southern part of the chain
of Eonesia islands. Formed after the human disappearance, this archipelago was
settled exclusively in a natural way, so there are no representatives of many
terrestrial groups of animals on the Burotu Islands that do not have the ability
to fly, with the exception of lizards. Due to the
humid climate, the Burotu Islands are a favorable place for forest growth. The
productivity of the ecosystems that have developed on the islands is sufficient
to maintain the existence of a population of a peculiar predator – a very large
representative of seagulls, which is called miru (the name of Polynesian god
of the dead).
Since the time of settlement on these islands, the ancestors of this species
gradually switched to hunting for land prey, since they had no competitors on
the islands. This bird species is much larger than other seagulls – the height
of an adult bird reaches 70 cm, of which half is the height of the legs. The
weight of an adult bird reaches 5 kg; the male is smaller than the female. The
webbing between the toes is reduced to a barely noticeable skin fold. Despite
its large size, miru retains the ability to fly. This bird takes to the air
rarely and reluctantly, so its wings are relatively shorter than those of smaller
gulls. But from time to time individuals of this species move within the archipelago
from one island to another: this feature makes it possible to preserve the genetic
unity of the population and to avoid degeneration, and at the same time makes
it possible for small island subpopulations to exist.
The plumage of this seagull is gray with mottled spots on its back, a little
like the juvenile plumage of ordinary seagulls. The beak of an adult bird is
yellow; the female has a pink spot on the tip of the beak, turning red by the
time the chicks are hatched. The wing tips and tail are black. Miru usually
hunts alone or in pairs. These are monogamous birds that form a pair for life.
Each pair has its own territory, which both birds protect from competitors.
Usually the territories of forest birds are larger, and their borders are not
strictly observed. Couples living on the shores gather animals cast ashore and
ravage colonies of seabirds; their territory borders are strictly guarded, although
the territory itself is smaller in area.
Miru often attacks forest birds, slaughtering even larger individuals. Migratory
birds from South America wintering on the islands often become prey of miru.
Miru also willingly eats terrestrial crustaceans and is able to dig their burrows
with its beak. When hunting alone, the bird sneaks up to the prey, and then
attacks, inflicting dangerous wounds with its beak. When hunting in pairs (outside
the breeding season), one of the birds can drive the prey to the other one.
Miru nests in burrows, choosing the most elevated areas of the territory, which
are not reached by the highest tides. The mating season begins in October. At
this time, the pair renovates the nest hole or digs a new one. The male takes
care of the female, preening the feathers on her head. Sometimes birds start
“nodding” to each other and flapping their wings together. During the mating
season, fights often occur with young males trying to challenge the right to
a female and a burrow from an older male. Young males present the female with
an edible gift – a dead fish, a piece of meat, a lizard or a crab. If the female
has accepted the offering, the male opens his wings and begins to stamp and
nod, standing near the female. If the female accepts it, she repeats this “dance”.
Some of its elements are kept in the existing pair, but the male can limit himself
to a symbolic “gift” in the form of a tree leaf or twig. The female lays 2-3
eggs, measuring (65-81) × (41-54) mm, with a white or grayish shell right on
the ground. Incubation lasts 35 days. Usually parents manage to feed successfully
only one chick, less often two. The chicks are colored darker than their parents:
the downy dress is gray with spots and a white “mask”, the juvenile plumage
is dark gray, the beak is brownish yellow. The development of young birds is
slow. In the first 2 to 2.5 months of life, crabs are dangerous for chicks,
and even ants are dangerous before the age of one month. Young birds reach sexual
maturity and acquire adult coloration in the third year of life. Life expectancy
is 40-45 years.
This bird species was discovered by Nick, the forum member.
Satrap,
giant seagull (Larus satrapes)
Order: Charadriiformes (Charadriiformes)
Family: Gulls (Laridae)
Habitat: the Arctic coast of Eurasia and North America, as well as islands.
Despite the serious competition with the auks and the “plankton catastrophe”,
the seagulls survived not only the era of human domination, but also the ice
age. Some species survived due to their ability to coexist with humans, and
some due to the relatively small impact that human activity had on the coastal
polar waters in comparison with the seas of lower latitudes. In the Neocene
in the Northern Hemisphere, these omnivorous birds hold an honorable second
place among feathered hunters for ocean fish. The largest of the seagulls of
the northern seas is satrap, or giant seagull. This species is a descendant
of the glaucous gull, or burgomaster (Larus hyperboreus). The range of the species
covers the Arctic Ocean coast as a ring, and birds from the populations of Wrangel
Island and Beringia do not interbreed with each other – when representatives
of this species inhabited these areas from different directions, they accumulated
differences at the species level, although there are no such sharp morphological
differences between neighboring populations throughout the rest of the range.
Satrap is a large species of seagulls: body length up to 70 cm, wingspan 1.6
m. The color is characteristic for the “herring gulls” group of species – gray
back and wings, white primary and secondary feathers, head and belly; beak and
legs are yellow. The female has a vertical red stripe on her beak. This species
differs from its ancestors by a deeper and laterally compressed beak, the tip
of which is hooked, suitable for tearing meat and holding slippery prey. Populations
from Eurasia are distinguished by a lighter leg color and a short series of
calls, including no more than four “syllables”. Populations from Iceland, North
America and Beringia have darker legs (orange in color), an orange iris and
a “polysyllabic” call – up to six “syllables”. They also have gray secondary
feathers.
This seagull is exclusively zoophagous, feeding on fish, squid and small auks.
It often pecks carrion and after a storm wanders along the coast, looking for
dead marine animals. Satrap nests near colonies of seabirds, regularly stealing
their eggs and chicks, and sometimes forcing to regurgitate the caught fish.
Satrap is wary only of gannetwhales,
which can easily kill and peck an unwary seagull, but satraps regularly feed
on the corpses of these birds. A competitor of this species is the sea
eagleraven (Aquillorax maritimus), which also feeds on carrion and marine
animals. Usually these two species coexist in a kind of “armed neutrality”,
not attacking each other, but avoiding the neighborhood. Quarrels with eagleravens
arise only because of the place for the nest, or when both birds try to steal
eggs from the same nest. Because of the fish, both species do not quarrel, because
the prey of satrap swims in deep water, where the eagleraven flies quite rarely.
However, birds of both species try to rob each other after the luck in fishing.
The nesting period of satrap begins in April; pairs are formed for one nesting
season. Before the start of nesting, males and females perform complex and long
mating dances, during which they bow and rub their beaks against each other.
A special figure of the dance is walking with wings stretched wide towards each
other. The culmination of the mating ritual is beaks raised to the sky and a
loud synchronous call. Nests are most often arranged on rocks near cliffs. Birds
pay little attention to the nest arrangement – this is an ordinary deepening
in a stone or soil, lined with down and dry algae. Birds nest in separate pairs
or small colonies, numbering up to 3-4 breeding pairs. In a clutch there are
usually 2-3 eggs with a yellowish spotted shell. The clutch is incubated by
both parents for 27-28 days. Chicks are hatched covered with brown down with
black spots on the back. Young birds become independent at the age of 45-50
days. Juvenile plumage is white with a brownish tinge, darker on the wings and
back. The bird reaches sexual maturity at the age of 2 years; life expectancy
is up to 27 years.
This bird species was discovered by Nick, the forum member.
Tuyango
(Larovenator terribilis)
Order: Charadriiformes (Charadriiformes)
Family: Gulls (Laridae)
Habitat: coasts of South America.
Some species proved to be capable of surviving human interference. The kelp
gull (Larus dominicanus) was one of them. In the Neocene this species give rise
to a new one, tuyango (named after mythical predatory bird from Argentina),
or the raptorial gull. Living in the Pacific and Atlantic coastal areas of South
America, this species is an aggressive predator and scavenger. This species
is a vagrant in Africa, North America, Eonesia and Europe.
Tuyango has 82 cm of length and 179 cm of wingspan. The adult bird has black
upperparts and wings. The head, underparts, tail, and the wing tips are white.
Their thick and powerful bill is yellow with a red tip. Their legs and webbed
feet are bright orange. Juveniles have dull legs, a black bill, a dark band
in the tail, and an overall grey-brown plumage densely edged whitish. The young
stay with this plumage until they reach maturity within five years.
The diet of this species is composed of fish, mammals and other birds. Being
a successful predator, they are capable of taking down large prey while acting
in pairs. Birds that feed on beaches are frequent prey for them. Tuyango attacks
the flocks while they feed, killing those who are left behind during the panic.
This gull can also eat carrion, being capable of fighting with vultures and
other birds of prey for a carcass. They usually frequent colonies of seabirds
where they catch eggs and chicks. When attacking other flying birds, this species
often pursue them on the wing and attack them by jabbing with their bill, hoping
to bring down the other bird either by creating an open wound or simply via
exhaustion; this strategy is used to subdue young boobies and albatrosses when
they start to leave their nests. Some individuals learned that, while waiting
during the dusk, they may be able to surprise and kill a vesper bird returning
to the nest.
This species breeds singly, usually in areas close to colonies of other seabirds,
thus ensuring food supply to the offspring. Being monogamous, the pair will
hunt together, this way being capable of killing large prey. The nest is a shallow
depression on the ground lined with vegetation and feathers. The female usually
lays 2 or 3 greenish-brown, dark speckled eggs. Both parents participate in
the incubation stage, which lasts for approximately 29 days. The breeding pair
are devoted parents who both take shifts brooding the young, defending the nest
and gathering food. They will attack any intruder approaching its nest. Young
tuyangos leave the nest area at 52 days of age and remain with their parents
for an overall period of around six months. Their lifespan is 32 years.
Other species of the Genus Larovenator is Antarctic
tuyango (Larovenator griseus). Living in the Antarctic Ocean, this
species has 80 cm of length and 178 cm of wingspan. It has a white head, neck,
breast, and belly, a white tail, and pale gray wings and back. The ends of its
wings are white-tipped. Their beak is thick, powerful and yellow with a red
subterminal spot. During the winter, this species migrates to Meganesia and
New Zealand.
This bird species was discovered by João Vitor Coutinho, Brazil.
Antarctic
kaveskar (Kaveskar australis)
Order: Charadriiformes (Charadriiformes)
Family: Penguigulls (Sphenicilariidae)
Habitat: Antarctic Peninsula and nearby islands.

Picture by Pavel Volkov
In the Neocene, in the cold waters of the Southern Hemisphere
of the Earth, fish and invertebrates are caught by penguigulls,
representatives of a family convergently similar to puffins and penguins. True
penguins in the Neocene are relics inhabiting cold
freshwater reservoirs. The family of penguigulls includes several genera
that differ in behavior and details of anatomy. Its most spectacular representatives
belong to the kaveskar genus (Kaveskar), inhabiting the south of South America,
West Antarctica and nearby islands. This genus of birds is named after an Indian
tribe of hunters of marine animals and fishermen from the shores of the Strait
of Magellan.
The Antarctic kaveskar is the largest species of the genus; the adult bird reaches
80 cm in height. Its appearance shows a deep adaptability to underwater fishing:
the wings have turned into flippers, the toes are connected by palamas. The
relatively long and thick beak is used for catching prey. These birds feed on
fish, crustaceans and squid, which are caught underwater. When moving underwater,
they flap their wings, as when flying – in this way they are convergently similar
to penguins that lived in these places in the human era. Kaveskars prefer to
escape from predators on land: there are no land animals in Antarctica that
can be dangerous to them, but large predatory fish threaten them underwater.
The plumage of kaveskar is dense and at first glance resembles scales. Its coloration
is typical for aquatic animals: the back, head and wings are black, the neck
and belly are white. In mating plumage, both males and females are very beautiful:
a bunch of curved white feathers grows over the beak; the cheeks are also colored
white. During the mating season, the beak of kaveskars acquires a very bright
color: red cross stripes appear on a yellow background. At the tip of the mandible
there is a bright red spot, which serves as a signal for the beginning of feeding
for the chicks. In the non-breeding season, after molting, the bright feathers
above the beak shed, the cheeks turn black, and the beak is uniformly yellow.
Antarctic kaveskars live all year round on the coast of Antarctica and in Subantarctic
area. Their mating season begins in October. Birds form pairs, arranging spectacular
displays accompanied by loud trumpet calls. Kaveskars are monogamous, but the
“bonds of marriage” connect a male and a female for only one season. Kaveskars
nest in colonies numbering from several dozen to several hundred individuals.
These birds nest in burrows up to 2 m deep, with a nest chamber without litter
at the end. The newly formed pair either digs a new hole, or occupies one of
the old ones. Burrows often cause serious conflicts between family couples.
In the nest chamber, the female lays 2-3 eggs, which are incubated alternately
by both parents. After the chicks hatch, the male and female alternately go
to the sea for food for them. The remaining parent protects the nest from predators.
Although kaveskar chicks are of precocial type, they become independent only
by the end of the Antarctic summer. At this time, they leave their parents.
The young bird looks like an adult in non-nest plumage.
In addition to the Antarctic species, the genus also includes several species
living further north:
Patagonian
kaveskar (Kaveskar patagonensis) inhabits the eastern coast of South
America, from Tierra del Fuego north to La Plata. It is slightly smaller than
its Antarctic relative and reaches a height of 70 cm. The hair-like feathers
above the beak are yellow in color.
Chilean
kaveskar (Kaveskar pacificus) lives on the Pacific coast of South America,
north to Peru of human epoch and reaches the same size as the previous species.
The feathers above the beak of the Chilean kaveskar are orange, and the beak
itself is red with black stripes.
Falkland kaveskar
(Kaveskar nanus) is endemic to the Falkland Islands and the smallest
species of the genus, up to 60 cm tall. Dark tones dominate in coloration of
its plumage: the feathers above the beak are black with a violet metallic sheen,
and a black cross stripe stretches across the throat. The cheeks are not colored
white, but there is a thin ring of small white feathers around the eyes.
The ranges of the four species of the genus sometimes overlap, but due to strong
differences in mating coloration, cases of interspecific hybridization are very
rare.
These bird species were discovered by Simon, the forum member.
Common
vesper bird (Hesperides aurora)
Order: Charadriiformes (Charadriiformes)
Family: Vesper birds (Hesperidae)
Habitat: islands of Southern Atlantic.
During the Holocene, human interference caused the extinction of many species.
After the extinction of the man, the surviving species continued their evolution
giving rise to new ones. The common vesper bird is one of them, a member of
the Hesperidae Family that was part of the Charadriiformes. Their closest relatives
are the gulls and terns. This species lives in the tropical and temperate areas
of the Southern Atlantic Ocean, nesting in islands.
Its common name means “bird of the afternoon” and their binomial name is formed
by the words Hesperides, the Greek nymphs of the afternoon and Aurora, the Roman
goddess of dawn. These names are related to an adaptation presented by adults
during the nesting season. To feed their young, one of the parents will leave
the nest at the first hours of the dawn and will return at the last hours of
dusk, then the other parent will go search for food, returning in the dawn.
This way they avoid predators that can find the nest and kleptoparasitism by
other birds.
Morphologically, the common vesper bird is similar to the storm petrels (Genera
Hydrobatidae and Oceanitidae) of human era; they have a larger head, thicker
neck and strong long legs. Their wings are long, slender and pointed, this way
they soar over the sea. This is a case of convergent evolution. Their bills
are small, but strong. Their slightly curved tip helps in the capture of prey.
This species has 21 cm of body length and a wingspan of 48 cm. Most of their
plumage is dark grey, with the exception of the wings that are white and a crescent
shaped white mark that extends from one side of their head to the other. Their
tail is white and the males develop an elongated streamer. The bill is red and
the feet are orange. The precocial chick is yellowish with brown markings and
the bill is pale yellow. The eyes are large, allowing them to hunt during the
night.
Both males and females have large outgrowths in the base of the upper jaw. These
structures contain salt-removing glands inside and allow these birds drinking
salt water without any harm to their health. This characteristic allows these
birds to spend most of their lives in the ocean, only returning during the nesting
season.
This species had evolved to partially fill the niche of the storm petrels. They
feed on plankton, fishes, crustaceans and molluscs. Usually, these birds will
catch their prey directly from the surface by hovering above it and then taking
small morsels. However, they can seldom swim over it and more rarely dive, submerging
for some seconds, but to no more than 50 cm below the surface. It will also
track groups of predatory fish and other birds, waiting for their prey to be
driven to the sea's surface. As the chicks grow, they are fed increasingly larger
quantities of fish and crustaceans by their parents, generally partly digested
and regurgitated.
The common vesper bird forms pairs when they are at sea. The males initiate
an aerial courtship display of flying in large circles, alternating between
gliding, short periods of rapid wing-beating, and low flight within a few meters
of the water, while making sharp cackling calls. Initially flying in small groups,
birds then pair off to repeat the display in pairs before bonding. After bonding,
the pair will arrive on the island where they nest and the male will offer fish
to the female.
Nesting takes place in loose colonies on oceanic islands. A monogamous and philopatric
species, the pair will return to the same nesting place. The common vesper bird
makes their nests in crevices between or under rocks, or burrows in the soil.
When they make their own burrow, they loosen the earth with their bills and
kick out the debris with their feet. The construction of the nest occurs at
night. It usually lays a clutch of one pale purple egg with reddish-brown spots.
It is incubated by both sexes for 43 to 47 days. The chicks eventually fledge
in about 10 to 15 weeks after hatching and leave the nest at night with this
age. The juvenile reach sexual maturity with 4 years. Their lifespan is 23 years.
The Family Hesperidae is divided into two Genus: Hesperides and Erytheia.
The species that form the Genus Hesperides are:
Antarctic
vesper bird (Hesperides australis)
Living in the Antarctic Ocean, this species has 18 cm of length and a wingspan
of 43 cm. The Antarctic vesper bird is white, with pale grey plumage in their
wings and a black patch around and behind their eyes. The central feather of
the breeding males is very short compared to the other species of the Genus.
This species built their nests away from the ocean, being one of the few seabirds
of Antarctic that nests away from the sea. This is an attempt to avoid most
of the predators that stay close to the large nesting colonies of the other
seabirds. However, this characteristic forces the parents to make long travels
to reach the sea to find food for the chick. In the winter, this species migrates
to more warm areas in the Indian ocean.
New
Azora vesper bird (Hesperides atlantica)
Being endemic to New Azora island, this is the smallest representative of the
Genus, with only 14 cm in length and a wingspan of 32 cm. The plumage of this
species is mostly dark brown, except for the white rump and head.
Northern
vesper bird (Hesperides borealis)
Living in the North Atlantic Ocean, this species has 19 length and 41 cm of
wingspan. It has a pale brown to grey back, rump and wings with black flight
feathers. It is white below and has a white face with a black eye mask.
The species that form the Genus Erytheia are:
Pacific
vesper bird (Erytheia pacifica)
Living in the tropical and temperate areas of the Northern Pacific Ocean, this
species has 21 cm of length and 43 cm of wingspan. Most of their plumage is
pale grey which is contrasting with darker grey on the upper wings and the rump.
They also gave a distinct white head, and prominent dark eye patch.
Californian
vesper bird (Erytheia californiana)
This species is endemic to California island. The smallest of the genus, this
bird has 14 cm of length and 38 cm of wingspan. Its plumage is dark grey and
white, and it has a dark grey head with prominent white feathers on the cheeks
and throat. There is a white speckling on forehead along with a prominent dark
eye patch. Its underside is mostly white, which turned dark grey at underwings.
Solitary
vesper bird (Erytheia solitaria)
Living in the South Pacific Ocean, this species has 20 cm of length and 42 cm
of wingspan. Their plumage is similar to the Pacific vesper bird, except for
the absence of dark grey in the rump that is replaced by white.
This bird species was discovered by João Vitor Coutinho, Brazil.
Grumbling
loon (Gavia murmurara)
Order: Loons (Gaviformes)
Family: Loons (Gaviidae)
Habitat: Eurasia from Crimea and Ustyurt islands in the south to the Arctic
islands in the north and from the English Channel to Beringia, North America
to Greenland.

Picture by Cossus
In the human era, loons were a small group of birds widespread
mainly in the cold latitudes of the northern hemisphere. During the ice age,
loons were pushed south by the advancing ice along with other northern animals.
Later, when the glacier retreated, some populations remained in the south and
evolved into new forms, others
gradually migrated back to the north and settled in familiar conditions, and
others simply adapted to the changed habitat conditions. The grumbling loon,
a descendant of the black–throated loon (Gavia arctica), belongs to the latter.
This species is a relatively large aquatic bird: adult weight 4.0-6.4 kg, body
length 75-100 cm, wingspan 135-155 cm. The color of the plumage of the upper
side of the body is grayish-brown, a ring of small feathers with a metallic
green shine appears around the eyes in some populations. The belly is white
in representatives of all populations. A characteristic feature in the appearance
of birds is a bright pink beak. Geographical variability is clearly expressed
in this species: the closer to the south, the darker the color of the bird,
when moving to the east, the “ring” around the bird’s eyes becomes more expressed.
Birds prefer to choose partners similar to members of their population, so even
in wintering areas, different populations do not interbreed and avoid each other.
In addition, with the accidental hybridization of birds from remote populations,
the offspring turns out to be infertile – so strong are the intraspecific differences
at the edges of the range. The voice of this species is very characteristic:
it’s a “uorr-uorr-uorr-kerr” grunt. The alarm call resembles a horse neigh.
Grumbling loon feeds on any medium-sized pelagic and bottom-dwelling fish, in
the Fourseas the basis of the diet consists of gobies and fry of large fish.
Also, birds often eat arthropods – crustaceans and aquatic insects; frogs are
rarely hunted. The hunting territory of a pair of birds has an area of up to
3-5 hectares; birds regularly declare their rights to it by vocalization. The
northern populations of grumbling loon are migratory, the southern ones are
sedentary. Birds from Eastern Siberia migrate to Baikal and further to the wetlands
of China, ones from the Arctic Ocean cross the Beringian Isthmus and fly along
the Pacific coast, part of the population remains on the Sea of Okhotsk. Birds
from the Atlantic migrate south along the coasts of Eurasia and North America,
and part of the population winters on the Fourseas.
The mating season in the south begins in April, in the north – in May. Grumbling
loon nests in the same place from year to year, often using last year’s nest.
Moreover, on the pond inhabited by these birds, as a rule, only the same pair
of birds settles for several years in a row. The nest is located in lowland,
often at the water’s edge. Birds at the nest behave secretively; hunting sites
are often located several kilometers away from it. During courtship, males and
females in the established pair preen each other’s feathers and present a symbolic
gift – a bunch of water plants. Young birds arrange more rampant games, accompanied
by courtship flight: they synchronously run up on the water and take off, and
also synchronously land on the water, swim and dive. In this case the “gift”
is more often real – a small fish or crayfish. There are two eggs in the clutch,
sometimes one, 9 cm long with a greenish spotted eggshell. Both partners hatch
the eggs and feed the chicks; incubation lasts about 25 days. The chicks are
covered with down and have opened eyes. They immediately leave the nest and
learn to catch small animals on their own. Parents feed them for the first three
weeks of life, and then their care islimited mainly to protection of the chicks.
The young bird fledges at the age of about two weeks. At 5 weeks, the juveniles
are already flying, but they become independent only in winter. Birds from the
northern part of the range go to winter with their parents, but return to the
nesting sites on their own. Grumbling loons become sexually mature at 3 years
old. Life expectancy is up to 20 years.
This bird species was discovered by Nick, the forum member.
Azorean pseudododo (Terricolofulma pseudoraphoides)
Order: Petrels (Procellariiformes)
Family: Ground stormy petrels (Xenoprocellariidae)
Habitat: New Azora island, woods and bush thickets.
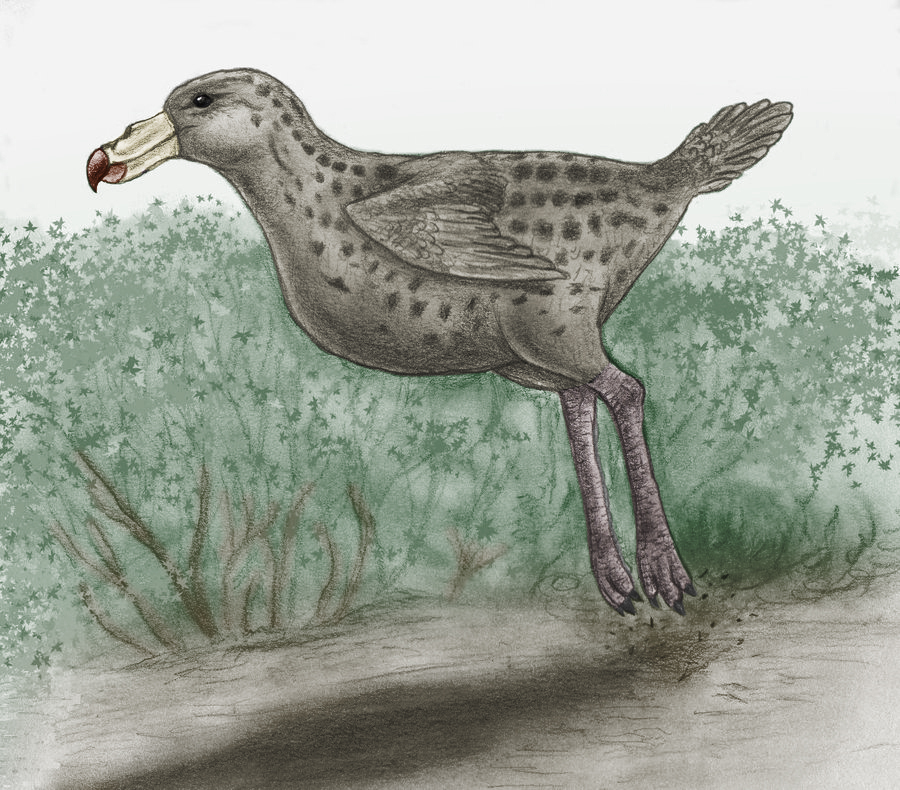
Picture by Carlos Pizcueta, colorization by Biolog
Initial image by Carlos Pizcueta |
Ecological crisis at the boundary of Holocene and Neocene had
caused changes in efficiency of ocean - the most important and simultaneously
the most sensitive to changes in biosphere source of food for set of species
of animals. The global cooling and change of direction of currents had caused
mass extinction of plankton and pelagic animals. As the result many species
of large-sized sea animal appeared doomed to starvation. Ecological crisis had
completely destroyed populations of sea cetaceans and other animals, undermined
in Holocene by human activity. Sea birds appeared harder to these changes, and
their species could evolve to new ones. And some of their descendants in Neocene
lead absolutely new habit of life.
Island habitats appear rather favorable for evolution of birds, especially if
on islands mammals are absent. In Holocene epoch people had introduced to various
islands animals which could not appear there in the natural way, therefore only
few islands have no mammal fauna. It is, for example, Iceland on which all ground
fauna had been destroyed by glaciers. And on New Azora placed at the south ground
animals had kept and evolved. But one of birds successfully competes to them,
taking a high place in food chain. This bird is the Azorean pseudododo.
This bird differs by many features from rather stupid and harmless Mauritian
dodo quickly exterminated by people and mammals introduced to its native island.
It is flightless carnivorous bird, the descendant of sea bird mallimauk (Fulmarus
glacialis) belonging to Procellariiformes order. The “true” dodo was herbivorous
bird, but this bird has kept and has strengthened abilities of carnivorous creature:
it eats small animals - murine rodents and small predators, nestlings of other
birds, and also carrion. Obviously, the adaptation of ancestors of this species
to feeding by ground animals was consequence of sharp falling of biological
efficiency of ocean in early Neocene (the ancestral species ate plankton, but
already in human epoch it had began to pass to feeding by waste products of
sea crafts). This species differs from dodo also by aggression that helps the
pseudododo to survive and succeed in the world of New-Azorean mammals. Besides
it differs in large size (the adult bird weighs about 6 - 7 kg), and the majority
of small inhabitants of New Azora represent delicious catch instead of enemies
for it.
Despite of so obvious distinctions, one feature strongly makes this bird similar
to dodo. Pseudododo, similarly to dodo, has kept many juvenile features of flying
ancestors, becoming, actually, an adult and sexual mature nestling. Body and
head of pseudododo are covered with soft down, feathers are only in tail and
wings. The body of bird is colored grey with large spots merging to cross strips:
such colouring makes bird undistinguished in underbrush of New-Azorean woods.
The beak of bird is well adapted to predating: it is big and high, with sharp
edges and hook on the tip. The characteristic feature of order, tubular nostrils,
was kept at the Azorean pseudododo. The beak of pseudododo has rather bright
colouring: it is ivory-colored with red - brown tip. At males the spot on tip
of beak is larger, than at females.
Wings of this bird as if had stopped growth in juvenile condition: they are
strongly reduced, narrow and short. Wings serve this bird for maintenance of
balance at fast movement. Tail is short and fanlike.
Legs of pseudododo are strong and rather long. The manner of movement overland
of this bird is original: these birds jump, similarly to kangaroo, pushing from
the ground by all hypotarsus (such way of movement on the ground was characteristic
for ancestor of this species). In case of necessity bird can run, chasing catch.
The palama is reduced, toes are rather short, have grown together to one third
of general length. On them thick strong claws are advanced, providing good coupling
with the ground at movement. On back (actually bottom) side of hypotarsus and
toes there is corneous calloused thickening.
Azorean pseudododoes develop rather slowly: the nestling develops in nest about
eight months, and the young bird becomes sexual matured only at the sixth year
of life. Pair at these birds is formed to all next life. Courtship ritual is
accompanied by complex caring, and during all life birds express attachment
to breeding partner. The bird chooses new breeding partner only at death of
previous one. This species always nests on the ground, frequently in holes dug
out by other animals. In clutch there is always only one egg. But nesting occurs
only alternate years, and theoretically breeding pair of birds can bring up
only 12 - 15 nestlings for all life. The incubating lasts 40 days; both parents
hatch and look after nestling, and thereof survival rate of posterity at pseudododoes
is very high. Parents feed posterity by small animals. While the nestling is
small, one of parents constantly stays in nest or nearby from it. Later, when
the nestling will grow up and will start to become fully fledged (approximately
at the third month of development in nest), both parents start to hunt, leaving
it alone. In case of necessity nestling can protect itself from enemies, spitting
out in them repulsively smelling oily liquid, and biting. Having abandoned the
nest at the age of seven and half or eight months, the nestling studies to search
for food independently: parents do not care any more of it. It is the critical
moment in life of pseudododoes, and the significant part of young growth perishes
in this period. Survived birds can expect for long life - up to 50 years and
more.
Great
diving albatross (Diomergulus magnus)
Order: Petrels (Procellariiformes)
Family: Diving albatrosses (Diomergulidae)
Habitat: temperate and tropical latitudes of Atlantic Ocean, islands.
During the Holocene, the human interference caused the extinction of many species.
After the extinction of the man, the surviving species continued their evolution
giving rise to new ones. In the Neocene a new Family of Procellariiformes appeared,
Diomergulidae, the diving albatrosses. These birds aren’t descendants of the
albatrosses of human era, but of shearwaters of the Genus Ardenna, which through
evolutionary convergence acquired physical characteristics similar to that of
the birds in the Genus Diomedea, thus the name of the family Diomergulidae,
Dio (from Diomedea and Diomedes) + Mergulus (from diving). The type species
of this group is the great diving albatross, a bird that lives in temperate
and tropical areas of the Atlantic Ocean, nesting in islands.
The great diving albatross is a large bird, with 88 cm of length and a wingspan
of 207 cm. This species is almost all white with exception of the wings, which
are dark brown. Their pale grey bill is large, strong, and sharp-edged, with
the upper mandible terminating in a large hook. Running from the top to the
sides of the bill there is two “tubes”, long nostrils that are characteristic
to the Procellariiformes. Their completely webbed legs are pink, in the same
way that their feet. This bird is not good walking on the land; because of this
they build their nests in areas near cliffs on islands, this way the birds can
more easily launch in the air. The flight is powerful and direct, with their
long wings held stiff and straight.
The great diving albatross eats fishes, cephalopods and crustaceans. This species
prefers to forage in the open sea. They catch their prey by plunge-diving, being
capable to reach 65 m in a dive. They use their wings to help swimming underwater
in a way similar to a flight; feet are also used to help in their swim. A gregarious
species during the nesting season, this bird can be seen in large numbers when
feeding. They will also track groups of predatory fish, waiting for their prey
to be driven to the sea's surface. They produce a stomach oil that is used both
against predators and as an energy rich food source for chicks and for the adults
during their flights.
Nesting takes place in large colonies in islands. Great diving albatrosses are
monogamous and highly philopatric, meaning they usually return to their natal
colony to breed. The males are the firsts to arrive at the colonies. They will
choose a place for the nest, those who choose the best places will be most likely
to attract a female. Fights between the males for a place are common, with them
hitting each other with their beaks and wings. After finding a good place for
the nest, the male will start to vocalize to attract the attention of a soaring
female. If the female accept the male, she will land next to him and they will
vocalize together, marking the place that they will construct their nest, after
that the pair will preen each other. The nest is a simple depression excavated
by the parents and lined with plant material. The female laid only one white
egg, which is incubated by both parents for 64 days. Once the chick hatches,
the parents raise their chick for 125 days. At first it is fed by its parents
every two days, with the feeding frequency reduced as it approaches fledging.
Having fledged, the young birds stays at sea until they reach sexual maturity
with five years, but only begins to breed with ten years. The great diving albatross
lifespan is of 58 years.
The family Diomergulidae has two genera:
The Genus Diomergulus, the Atlantic diving albatrosses. Besides the great diving
albatross, other species in this Genus are:
Southern
diving albatross (Diomergulus australis)
Living in the Antarctic Ocean and nesting in islands there, this bird has 90
cm of length and a wingspan of 210 cm. They have white plumage in the neck,
face, throat, lower breast, and belly; the other parts are dark brown.
Northern
diving albatross (Diomergulus borealis)
Living in the Arctic Ocean and nesting in islands there, this bird has 84 cm
of length and 205 cm of wingspan. They are nearly completely white, with only
their primaries and secondaries feathers being dark brown.
Brown diving
albatross (Diomergulus rufus)
The only species living in the Indian Ocean, this species is the smallest of
this Genus, with 78 cm of length and a wingspan of 195 cm. They nest in islands
near Jacarta Coast and Meganesia. This bird is completely brown, with the exception
of the head and throat, which are white.
The Genus Solitarimergulus, the Pacific diving albatrosses. Differently of the
birds in the Genus Diomergulus, which form large breeding colonies, the Pacific
species are more solitary, forming loose groups with widely spaced nests. Other
differences are that their dives are less deep, reaching 45 m, and they are
smaller than the Atlantic species. The species in this Genus are:
White-fronted
diving albatross (Solitarimergulus albifrons)
Living in North Pacific, this species has 70 cm of length and 187 cm of wingspan.
They are almost completely dark brown, with the exception of their characteristic
white forehead. They also have white tail feathers. White-fronted diving albatrosses
nests in islands near Japan and Bering Sea, forming small groups.
Solitary
diving albatross (Solitarimergulus eonesianus)
Living in tropical latitudes of the Pacific Ocean, this species has 69 cm of
length and 183 cm of wingspan. Their plumage is completely brown, with only
the tail feathers being white. The solitary diving albatross only nests in the
islands of Eonesia, with the pairs making their nest very far from each other,
because of this they are named “solitary”.
Sooty
diving albatross (Solitarimergulus fuscus)
Living in South Pacific, this species has 73 cm of length and a wingspan of
191 cm. They are sooty-brown with dark brown plumage in their wings and white
feathers in their tail. The sooty diving albatross nests in islands near New
Zealand and the Pacific Coast of Meganesia, forming small groups.
This bird species was discovered by João Vitor Coutinho, Brazil.
Lesser
nomad albatross (Thalassocrator minor)
Order: Petrels (Procellariiformes)
Family: False albatrosses (Neodiomedeidae)
Habitat: Northern and Central Atlantic, islands.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. One
of these new species is the lesser nomad albatross, a close relative of the
nomad albatross. This
species wanders the skies over the North and Central Atlantic, returning to
land only to nest.
The lesser nomad albatross is a large seabird, having 150 to 180 cm of length
and a wingspan up to 400 cm. It makes it one the largest flying birds of the
Neocene, being only behind the nomad albatross and the acatu.
Their plumage is similar to that of their large cousin. Beyond the size, the
main differences between the two species are the bright yellow beak with a dark
gray tip and a yellow-stained nape and crown that are present in the lesser
nomad albatross. Their feet and legs are red, with a partial web between the
fingers. Like their relatives, they also have a long beak, with the bottom jaw
having a "comb" which juts out downwards and the tip of the top jaw
is hooked.
This species travels vast distances and tends to feed further out in the open
sea than other members of the Genus Thalassocrator. They will also forage in
tropical areas, on rare occasions approaching the coast of South America and
Africa. They feed on cephalopods, small fish, and crustaceans and on animal
refuse that floats on the sea. They aren’t capable of diving, needing to catch
food that is located on the surface.
The lesser nomad albatross is a monogamous bird, living together for the rest
of their lives. The pairs are formed at the sea, with an aerial courtship and
vocalization of both the male and the female. The male will start to fly in
circles, while vocalising to the female. If she is receptive, she will mimic
him. After the pair is established, they will forage together. At breeding time
they occupy loose colonies on isolated island groups. The nests are a large
bowl built of grassy vegetation and soil peat. The female lays one large and
white egg, which is incubated by both parents for 37 weeks. During the early
stages of the chick's development, the parents take turns sitting on the nest
while the other searches for food. During this period they can be very aggressive
to any animal that approaches the nest. Later, both adults search for food and
visit the chick at irregular intervals. The chicks fledge after 310 days. The
adolescents return to the colony within six years; however they will not start
breeding until 11 to 15 years. They have a lifespan of 76 years, but a good
number of chicks will not survive until adulthood.
This bird species was discovered by João Vitor Coutinho, Brazil.
Giant
African stork (Megaciconia africana)
Order: Stork birds (Ciconiiformes)
Family: Storks (Ciconiidae)
Habitat: Western and Central Africa, Zinj Land, the north of Madagascar; river
valleys.
![]()
Picture by Amplion
At the boundary of Holocene and Neocene, during global ecological
and climatic catastrophe, the global fauna began to change radically – borders
of climatic zones changed, new groups of plants and animals appeared and old
ones disappeared. During such changes at the boundary of epoch cranes, being
among largest flying birds of Holocene, had become extinct. Typically their
ecological niche got to descendants of their far relatives – rails, buttonquails
and other gruiform birds. But it had taken place not everywhere. In Africa (and
also at the south of Asia) the ecological niche of cranes was occupied by giant
storks, representatives of new genus of stork birds.
At first sight giant African stork looks very big bird. It reaches 2 meters
in height, and its wingspan is about three meters and even more. But legs make
almost half of growth of this feathery giant, and this large bird weighs only
11 – 13 kgs. It is able to fly, but flies very reluctantly, preferring to hide
in thickets of marsh plants along the banks of Sahara Nile and other rivers.
Giant storks are direct descendants of Holocene storks, namely European white
stork Ciconia ciconia. Though primary, in Holocene, this bird lived mainly in
Europe, anthropogenous pressure and changed climate had forced it to move to
Africa and India, having left the European bogs to other birds. Nevertheless,
many features of their descendant’s appearance remained former. In colouring
of these storks sexual dimorphism is expressed. Male of giant African stork
is white bird with bright red beak, black legs and wings. Females are much dimmer;
they are monotonously grey. Nestlings and immature birds have camouflage brownish
colouring with longitudinal strokes making them undistinguished in nests and
among thickets. In case of danger nestlings and young birds freeze, relying
on masking.
Food of giant African stork includes various aquatic invertebrates, fishes and
crayfishes. Occasionally birds peck out seeds of wild graminoids at various
degrees of ripening. Sometimes giant African storks ravage clutches of African
crocoturtle.
Because these birds became rather large and heavy, they had to alter to behaviour
of ancestors and to begin nest building on the ground, like various flamingos
of Holocene and Neocene. Giant African stork builds its mound-like nests of
river clay, stalks of papyrus and other river plants, and also of large branches
of various trees growing near rivers. Mainly males are engaged in search of
building material for nest, and delivery of building material for nest became
even a part of courtship ritual. Nests are constructing in places difficultly
accessible to ground animals. But occasionally their nests appear plundered
by omnivorous boaropotamus.
Sometimes egg-eating snakes ravage nests of these storks: egg
drillsnake sucks out eggs of these birds entirely.
In full clutch of this species there are 2 – 3 eggs; female incubates them mainly.
She protects posterity aggressively, and at this time turns to dangerous adversary.
The beak of giant stork corresponds to its size; it resembles a spear by shape,
and force in neck of giant storks is great enough in order to wound hard, or
to kill with one beak blow even such large predators as deadlynetta
and young crocoturtles, which may try to diversify the menu with young stork
nestling. Only mighty boaropotamuses, lords of rivers and swamps of Central
Africa, can force parents to abandon their posterity. But it happens seldom;
much larger number of storks perishes from predatory animals and turtles, when
they leave nests and start to study to survive independently.
Nests of giant storks left after hatching of posterity are frequently used again
by other birds, including giant storks of other breeding pairs. If the nest
is high enough, small birds stick their nests to lateral faces of stork nests,
or make their own nest at the top of the stork’s one after when storks have
finished nesting. While birds stay at the nest, their tiny neighbours do not
venture to settle beside: young storks and nestlings would like to practise
hunting for tiny birdies.
In Asia, at Hindustan Peninsula the close relative of giant African stork lives:
giant
Indian stork (Megaciconia bharatus). It is smaller bird – its growth
is about 150 sm, and wingspan reaches 2.5 meters at weight of only 5 – 6 kgs.
Males of this species, as against the African relative, have not black, but
red legs. Besides on its head around of beak the original mask of black feathers
is advanced. By the biology it differs from the African relative not so strong;
the only amendment is the size of bird. It flies more actively, and frequently
arranges nests on trees at riverbanks. Occasionally these storks can migrate
and nest on islands of Indian Ocean, reaching even up to Mauritius.
These species of birds were discovered by Bhut, the forum member.
Anhanveve,
or diabolic stork (Deinociconia gigantea)
Order: Stork Birds (Ciconiiformes)
Family: Storks (Ciconiidae)
Habitat: tropical and subtropical areas of South America.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species and genera.
The anhanveve is one of these new species, a descendant of the jabiru stork
(Jabiru mycteria). The name anhanveve is formed by the combination of two words
of Guarani origin: aña that means devil and veve
that was used for the ability of flight. The anhanveve inhabits all the tropical
and subtropical biomes of South America, being capable of living both close
and away from water.
The anhanveve is a large stork, with a maximum height of 180 cm and a wingspan
of 230 cm. The males are 25% larger than the females. They are also heavier
than them. The plumage is mostly white, with the exception of the primaries
and secondary feathers that are black. The beak, which measures 35 cm, is black
and broad, slightly upturned, ending in a sharp point. The males have a yellow
casque on top of the beak that becomes bright during the nesting season. Their
long legs are black. The head and upper neck are featherless and black, with
a featherless stretchable pouch at the base. The skin of the throat and pouch
is bright red.
This bird eats eats fishes, molluscs, insects, other invertebrates and amphibians.
However, differently from their ancestors they prefer to search for prey in
dry areas. The anhanveve can be seen patrolling savannas in search of food.
They are also very predatory, hunting mammals and smaller birds, even visiting
colonies of seabirds and aquatic birds to pick eggs and nestlings. They will
frequently scavenger, using their size to intimidade and displace smaller scavengers.
The anhanveve can be seen in pairs or small flocks of five to ten individuals.
They are monogamous, but the pair stays separated outside of the nesting season,
which happens during the dry period. Their courtship consists of several bill-rattling
displays and vocalization with their pouch. They nest in large colonies, usually
close to that of herons and other aquatic birds. The populations of coastal
areas will nest close to the nesting areas of seabirds. The nest of sticks is
built on tall trees, and enlarged at each succeeding season growing to several
meters in diameter. Half a dozen nests may be built in close proximity, sometimes
among nests of herons and other birds. The parents take turns incubating the
clutch of two to five white eggs and are known to more territorial than usual
against other anhanveve during the brooding period. The eggs are incubated for
27 days and fledge within 110 days. They stay with their parents until reaching
3 months. The sexual maturity of the species in reached with 4 years and they
have a lifespan of 25 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Wattled
stork (Neociconia carunculata)
Order: Stork Birds (Ciconiiformes)
Family: Storks (Ciconiidae)
Habitat: Eurasia, freshwater reservoirs.
The anthropogenic interference in the nature during the Holocene caused the
extinction of several species. Those that survived were able to continue their
evolution and their descendants appeared in the Neocene. Some species of stork
survived the anthropogenic onslaught and gave rise to the Genus Neociconia.
The wattled stork, the type species of this Genus, inhabits ponds, rivers, edges
of lakes, estuaries and other freshwater wetlands. Their range is from Eastern
Asia to Central Europe. The Three-Rivers Land, Siberia and Fourseas are examples
of places where these storks live and nest. A migratory species, the wattled
stork winters in warm regions of the South. Birds from Central Europe will spend
the winter in the savannas and wetlands of Eastern Africa and Zinj Land whilst
the birds from Asia and Siberia will spend the winter in the Indian subcontinent.
The wattled stork is a large bird, measuring 110 cm in height with a 198 cm
wingspan. It has long red legs, a long neck and a long, straight, pointed red
beak. The main characteristic of this species are their two wattles that hang
down from under the upper throat, one in each side. Its plumage is glistening
black overall with a downy white breast and neck, while the crown is dark brown.
The breast feathers are long and shaggy, forming a ruff which is used in courtship
displays. The face is nude, with red skin in front of the eye extending to the
base of the beak and in the wattles, being covered by small round wart-like
bumps. These bare parts become bright during the nesting season. The sexes are
identical in appearance, except that males are larger than females on average.
The juvenile resembles the adult in plumage, but the areas corresponding to
the adult black feathers are browner and less glossy and they lack the bare
skin on the face, and have less prominent wattles.
These birds feed of fishes, amphibians, reptiles, mollusks, crustaceans, insects,
small mammals and birds. Foraging for food is more common to happen in fresh
water, but they will also do it in dry land. When foraging at water, this bird
wades patiently and slowly in shallow water in a small group and often foraging
in mixed species flocks. It uses the wings to shade the water while hunting.
The wattled stork also follows large mammals, seeking to eat the invertebrates
and other small animals flushed by their presence.
Wattled storks have to take one of the toughest migrations. In late summer and
middle autumn, they gather in flocks of up to 300 individuals and prepare for
their flight to their winter range. During their migratory flight south, these
birds reach altitudes of 4,900–7,900 metres. Along their arduous journey, the
Asiatic population has to cross the Himalayan Mountains to get to their over-wintering
grounds in India. Many die from fatigue, hunger and predation. The European
population has a less arduous migration, flying down to their wintering areas
in the savannas of mainland Eastern Africa and Zinj land. Large concentrations
of these birds are found in the Saharan Nile.
This species is monogamous. The courtship will start in the ground, usually
in areas of foraging. The male will spread their wings and call a female that
if interested, will approach. Then he will start to click his beak while displaying
his breast feathers and with wings spread. If the female accepts the male, she
will mimic him. The pairs in courtship also have aerial displays. The pair will
soar in parallel, usually over the nest territory early in the mornings or late
afternoons. The nest is large, constructed from sticks and twigs, and sometimes
also large branches, at an elevation of 4–25 m. The wattled stork prefers to
construct its nest in forest trees with large canopies where the nest can be
built far from the main trunk. The female lays two to five large oval grey-white
eggs, which become soiled during incubation. The eggs are laid with an interval
of two days. Incubation takes 38 days, with both sexes sharing duties, which
commence after the first or second egg is laid. Fledging takes 60 to 71 days,
after which the young joins the adults at their feeding grounds. However, for
another two weeks, the young continue to return to the nest, to be fed and to
roost at night. These birds reach sexual maturity at the age of 4 years and
their lifespan is of 39 years.
Other species in the Genus Neociconia are:
Black-headed
stork (Neociconia europea)
Living in wetlands of Western and Northern Europe, a small population can also
be found in the western shores of the Fourseas. These birds have 97 cm of height
and a wingspan of 180 cm. The plumage of these birds is similar to that of the
wattled stork, but their head is black and they lack the wattles. Their face
is bare, with the skin being red, which becomes bright during the nesting season.
During migration, these birds will fly in large flocks through the Atlantic
coast of Africa, reaching the western savannas of the continent.
Malagasy
stork (Neociconia calva)
This species is endemic to the wetlands of Madagascar. They have 98 cm of height
and a wingspan of 135 cm. The head of the adult is totally bare and orange in
color, which becomes bright during the nesting season. Their plumage is white,
with black flight feathers and a forked blacked tail. They aren’t migratory.
Solitary
stork (Neociconia solitaria)
The ancestors of this species crossed the Beringia and reached the taiga forests
of Northern North America, usually those near the tundra. They forage in drier
areas than their relatives of Eurasia, usually searching for food in the tundra.
They rarely form large flocks, being seen solitary or in pairs. They have 105
cm of height and a wingspan of 180 cm. Most of their plumage is black. The undertail
coverts and the back of neck are white with a black cap. Their face is bare,
with red skin that becomes bright during the nesting season. Migratory, these
birds will fly to the Mexican plateau and the marshes of Florida Peninsula during
winter. During migration, these birds form familiar flocks, with the parents
and their young.
This bird species was discovered by João Vitor Coutinho, Brazil.
Great
magnificent booby (Austrosula magnificens)
Order: Suliforms (Suliformes)
Family: Boobies (Sulidae)
Habitat: southern Atlantic, coasts of Africa and South America.
During the Holocene, human interference in nature caused the extinction of many
species. Those that were lucky enough to survive, left behind descendants in
the Neocene. The family Sulidae was one of these survivors, leaving descendants
that filled several different niches. While some like the gannetwhales
became very different from their ancestors, some remained with a familiar appearance.
The Genus Austrosula maintained a morphology and lifestyle similar to that of
their booby ancestors. The type species of this genus is the great magnificent
booby, a bird that inhabits the tropical and temperate areas of the Southern
Atlantic, nesting in islands and coastal areas of South America and Africa.
The great magnificent booby is sexually dimorphic, with the female reaching
87 cm of length and wingspan of 157 cm, while the male reaches 77 cm of length
with a wingspan of 149 cm. Outside the breeding season, both have similar plumage,
their upper body and neck is black, while the head is white. The feathers of
the belly, chest and wings are white. Their tail feathers are also white and
have black tips. The bare skin of their faces is pale red, their legs and feet
are this same color. Their beaks are yellow. During the mating season, the bare
skin of the males becomes bright red and they develop white tufts on their heads,
coming from over their eyes, like eyebrows. Their return to the non-breeding
appearance during late spring, when the time to form pairs ended.
Like their ancestors, the great magnificent booby feeds on small fish and squid
that they catch by plunging into the ocean at high speed. This species have
preference for foraging close to the coast. They will also occasionally catch
leaping fish while skimming the surface.
The pairs of magnificent boobies remain together for several seasons. During
the mating season, they perform elaborate rituals. The males are always the
first to arrive in the breeding grounds. They select a suitable place for the
nests to be built, sometimes even the one that they used in the last season.
Once it was done and they successfully defended it from rivals, the females
will start to arrive.
The courtship of this species consists of the male flaunting his red feet and
dancing to impress the female. The male begins by showing his feet, strutting
in front of the female. Then, he bows to the female, showing off his head and
the tufts. The dance continues with the “sky-pointing”, which involves the male
pointing his head and bill up to the sky while keeping the wings and tail raised.
It ends with the male presenting nest materials and with a final display of
his feet.
This bird nests in large colonies. The female lays two or three chalky blue
eggs, about four to five days apart. Both male and female take turns incubating
the eggs, while the nonsitting bird keeps watch. Their nests are made of vegetation.
They do not have brood patches, but their feet is very vascularized and hot,
and the birds place the eggs under the webs. Eggs lost during the first half
of incubation are replaced. The magnificent booby usually raises just one chick,
the second one to hatch being unable to compete for food with its older sibling,
or even ejected from the nest by it. The incubation period is 44 days. The male
and female share parental responsibilities. The male provides food for the young
in the first part of their lives because of his specialized diving. The female
takes over when the demand is higher. Chicks feed off the regurgitated fish
in the adult's mouth. The adults feed their offspring for around 13 weeks, right
up until the time they leave. The young birds fledge with 87 days old. Sexual
maturity is reached with 2 years and their lifespan is of 27 years.
Other representatives of the Genus Austrosula are:
Antartic
magnificent booby (Austrosula australis)
Nesting in islands and coastal areas of Antarctica, this bird spends the winter
at the sea. The females are larger than the males, having 82 cm of length and
154 cm of wingspan. The males have 71 cm of length and 143 cm of wingspan. Their
plumage is similar to that of their Tropical relatives, with the exception of
the neck and head that are white. The males also develop tufts over their eyes,
but they are longer than that of the Greater magnificent booby. The bare skin
in their face, legs and feet is pale orange outside of the nesting season, becoming
bright on the males during it.
Lesser magnificent
booby (Austrosula minor)
Nesting in islands and coastal areas of the Indian Ocean, this species is the
smallest of the Genus. The females are larger than the males, having 73 cm of
length and 137 cm of wingspan. The males have 67 cm of length and 132 cm of
wingspan. They are totally white, with the exception of the neck and wings.
The males also develop tufts over their eyes, but they are shorter than that
of any other representative of the genus, giving the impression that they really
have eyebrows. The bare skin in their face, legs and feet is pale yellow outside
of the nesting season, becoming bright on the males during it.
Moustached
magnificent booby (Austrosula pacifica)
This species nests in islands and coastal areas of the Southern Pacific Ocean.
The females are larger than the males, having 83 cm of length and 155 cm of
wingspan. The males have 77 cm of length and 144 cm of wingspan. They are mostly
slate-gray, with the exception of the head and belly that are white. The males
develop tufts of feathers over their beaks, giving the impression that they
have moustache. The bare skin in their face, legs and feet is pale greenish-yellow
outside of the nesting season, becoming bright on the males during it.
This bird species was discovered by João Vitor Coutinho, Brazil.
Caribbean
manchot (Manchotus caribaeus)
Order: Suliforms (Suliformes)
Family: Boobies (Sulidae)
Habitat: Caribbean islands, sea coasts.
The “plankton catastrophe” that occurred at the end of the Holocene caused a
mass extinction of marine pelagic animals, and cetaceans suffered the most from
it. In the human era, the number of many their species was undermined: they
were on the edge of extinction as a result of excessive whaling. In due course
of restoring of marine ecosystems, their place was taken by cephalopods,
fish and very peculiar
birds – gannetwhales. These
birds are actually “hyper-penguins”, and are well adapted to life in the cold
waters of the Atlantic and Arctic Ocean. But in the Neocene, the species from
which these huge birds originate have also retained: in the warm seas of the
New World, penguin boobies, or manchots, representatives of the group that became
the ancestors of gannetwhales, live. These are relics of the Ice Age that have
lost the ability to fly and resemble the penguins in their body shape.
Caribbean manchot is a large bird 75-85 cm long, weighing about 5 kg. Its physique
is more reminiscent of archaic Paleocene penguins: it has a relatively graceful
constitution and a more elongated neck and head. The wings are well developed,
flipper-like, with short stiff feathers. A large horny claw grows at the tip
of the hand. The tail is fan-shaped. The color of the plumage is characteristic
for pelagic animals: the back and head are gray, the belly is white, the beak
is yellow. Females have a red vertical stripe on it, which becomes bright during
the breeding season. This species feeds on various kinds of fish, squid and
crustaceans, chasing prey in the open sea. These birds can fish in groups, driving
fish into a shoal near the surface and snatching them. Their prey is smaller
than that of tropical gannetwhales.
Caribbean manchot is flightless, but walks well on land, keeping an upright
position when walking. It lives in colonies on rocky islands, where sometimes
it has to climb, jumping and climbing on ledges of rocks. The claw on the wing
helps birds cling to rocks and defend themselves when attacked by a predator.
The mating season of these birds begins in early-mid-January. Pairs of manchots
are formed for one season. During the courtship ritual, males call females with
loud buzzing cries. The nesting area turns out to be divided into many individual
nesting sites. During the struggle for a place for a nest and a female, males
quarrel loudly and jostle, striking each other with their wings. The nest of
this species is a simple bowl of sand and stones, and the males remember their
nests and occupy their former nesting sites from year to year, especially if
these are good places won from relatives. During the annual renovation of the
nests, quarrels occur over the stones for the nest, which the birds steal from
each other. There are usually two eggs in the clutch, which are laid with a
break of 1-2 days. The clutch is incubated by both parents for about 40 days.
During incubation, the blood vessels on the palamas dilate greatly, due to which
the eggs are heated.
The chicks are covered with sparse gray down and remain in the nest for some
time. They are fed primarily by females holding prey at the red stripe on the
beak, from where the chick takes it. At the age of one month, the chicks already
leave the nest and begin to walk around the nesting area, where they gather
in “kindergartens” under the supervision of young birds who are not engaged
in feeding offspring. At this time, they are fed by both parents, who recognize
their chick by voice. At the age of 3 months, the chick is fully fledged; it
leaves the nest and descends to the sea for the first time. Here the young bird
learns to swim and fish, although the parents continue to feed the young bird.
At the age of 5 months, young birds fish in the sea on their own, and at 8 some
birds leave the colony. Sexual maturity occurs at the age of 4 years, life expectancy
is up to 20-25 years.
A closely related species, Florida
manchot (Manchotus floridianus), lives on the coast of North America.
It goes further north than all other species, often adjacent to gannetwhales
of southern populations. It forms several colonies on the Gulf of Mexico coast
and in the Mississippi Delta. This species is larger (up to a meter long), with
a darker color of the upper part of the body – dark gray with a silvery sheen.
The beak is yellowish-brown, on the lower jaw at females there is a large cherry-red
spot. Birds prey on fish and cephalopods at depths of up to 50 meters. Pairs
in this species are formed for life.
Motley
manchot (Manchotus galapagoensis) inhabits the Galapagos. Body length
is 85-90 cm. The color is piebald: the head, wings and upper back are gray,
and other parts of the body are white. The beak is yellow with a red tip in
male and female. This species differs from the Atlantic species in nesting features:
it nests in burrows that are arranged at a distance of several tens of meters
from the shore. The colony consists of several dozen pairs that dig holes in
the neighborhood, at a distance of several meters between them – often among
shrubs, where the soil is held by their roots. Adult birds jointly protect nesting
sites. “Kindergartens” of the growing chicks gather on the coast 3 weeks after
hatching.
Crested
manchot (Manchotus cristatus) is an inhabitant of Burotu archipelago,
related to the Galapagos species. It is the smallest species in size: 60-65
cm long. The back is bluish-gray, the belly is white. Unlike other species,
a tuft of elongated filamentous yellow feathers grows on the head. Individual
forms from different islands vary in color (from yellow to orange) and size
(from large to several feathers above the eyes) of this crest. The species also
nests in burrows that are located 1-1.5 m far from each other; there are up
to 20-30 breeding pairs in the colony. The chicks gather in “kindergarten” at
the age of two weeks.
On the coast of Central America and the Panama Strait a large Panama
manchot (Manchotus colombiensis) lives, reaching 90 cm in length. It
resembles a Florida species, only the color is darker – the back is black and
there is a black transverse sling on the throat. The wings are black with a
white tip and back edge. Biology is similar to Florida manchot, but a significant
part of the diet is made up of crustaceans and cephalopods, as well as small
holothurias.
This bird species was discovered by Nick, the forum member.
Dwarf
gannetwhale (Nanodelphisula odontophora)
Order: Suliforms (Suliformes)
Family: Gannetwhales (Cetornitidae)
Habitat: North America, Mishe-Nama lake.

Picture by Tim Morris
Destruction of habitats, pollution of ocean and noncontrollable
hunting had caused extinction of sea mammals at the end of human epoch. Small
populations
of these animals continued the existing after the end of human epoch, but
they had died out soon as a result of gene pool poverty and degeneration. In
ice
age at the boundary of Holocene and Neocene in northern hemisphere they had
been replaced by gannetwhales, large descendants of sea birds. Because of
the restrictions connected to oviparity they never reached the size of the
majority
of cetaceans and the largest pinnipeds. These large flightless birds in Neocene
prosper in Arctic Ocean and reach a great number even in Neocene epoch, after
the general warming of planet climate.
One species of this original group of birds lives in Mishe-Nama lake – it is
dwarf gannetwhale. It is a typical relict of glacial epoch, the distant relative
of very large polar birds, evolved
independently of them, adapting to life in reservoir with the limited food resources.
Total length of dwarf gannetwhale does not exceed one and a half meters,
not including beak; weight of adult individual is about 60 kg. This bird
keeps
seal-like shape, characteristic for the present group of birds – powerful
flipper-like wings, horizontal position of body and legs shifted far back,
which help this
bird to push only on firm surface. This bird swims under water with the help
of wing movements, as penguins of human epoch did, and rudders by legs. Tail
of this bird is wide and flat, similar to beaver tail, but almost motionless.
The main task of tail is the creating of favorable conditions for egg incubation.
Tails of male and female do not differ in shape, because both birds equally
participate in egg incubation. On wings of a bird there is a pair of thick
corneous spurs, helping to clamber on rocks.
Plumage of dwarf gannetwhale is short and rigid. It had completely lost insulation
function, which was undertaken by hypodermic fat layer. The body of bird
is colored gray-blue; throat and stomach are white, tips of wings and tail
are
black. On head there is a black “bridle” – the thin ring of black feathers
surrounds the basis of beak and lasts up to eyes. Paws have grey color.
Beak of dwarf gannetwhale is shorter, rather than at sea forms, but is deep
and powerful, conic-shaped, of yellowish-brown color. On edges of beak teeth-like
outgrowths are advanced, allowing keeping the caught prey. This species eats
aquatic animals – fish and crayfishes.
Dwarf gannetwhale lives in numerous colonies numbering up to 200 individuals.
The whole population of this species is presented approximately by two tens
big colonies and set of small groups numbering no more than ten individuals.
This bird, clumsy on land, is aggressive enough and successfully protects
itself from land predators, biting them by beak. Frightening enemies off,
bird throws
out into them a jet of semidigested content of stomach. In winter birds keep
at edge of ice cover of lake. If winter is cold, birds keep ice-holes, breaking
away ice formed in them by beak.
Dwarf gannetwhales raise posterity at stony islands in the central part of
Mishe-Nama lake where almost whole population of this species gathers to
the end of winter. These islands are located just at the edge of the former
glacier
ard formed as a result of congelation at the boundary of Holocene and Neocene.
Here, among poor vegetation, in early spring the female lays an only egg
weighting about 3 kg. She hatches it, holding in paws kept like a dipper
and having covered
it by tail. Male replaces female, cautiously rolling egg in its own paws.
If the egg was casually broke during the incubation or had been infertile,
birds
do not repeat egg laying because the nestling develops for a long time and
birds will not have time to rear it. The incubation lasts till 48 days. The
nestling hatches naked and blind, but already at the first week of life it
becomes overgrown with rich black down. While the nestling is too small,
male incurs a duty on getting food for female and nestling. At the second
week of
its life parents stop holding the nestling on paws and leave it on the ground.
From this time the nestling receives food from both parents and starts to
grow much faster. By the end of the second month of life it(he) already completely
becomes covered by feathers, but continues to receive a forage from parents.
By the end of the third month of life the young bird has weight of about
50
kg. Parents gradually stop feeding of young bird, forcing it thus to creep
to water and to study food getting independently. Young dwarf gannetwhale
gains the weight typical for adult birds to an autumn. It becomes sexually
mature
only at the sixth year of life. Life expectancy of this species is about
60 years.
The idea of existence of the present group of birds was proposed by Dougal Dixon.
Hummock
gannetwhale (Delphisula glaciophila)
Order: Suliforms (Suliformes)
Family: Gannetwhales (Cetornitidae)
Habitat: polar seas, ice sheet of North Pole.
Picture by Tim Morris
Gannetwhales, large seal-like birds lost ability to flight
and walking on the ground, were a characteristic group of sea vertebrates of
Arctic Ocean in epoch
of Holocene – Neocene congelation. These birds, being, as a matter of fact,
“hyper-penguins” of Northern hemisphere, had evolved in Northern Atlantic,
but at the glacier receding they had been distributed farther to the north,
having formed some species in polar seas.
One of the largest species of this kind is hummock gannetwhale – the northernmost
species of genus, almost never appearing on firm ground. It is a huge flightless
bird weighting up to 500 kg at body length of about 3 meters. Prominent features
of appearance of this bird are long flipper-like wings, long beak with jagged
edges and large sensitive eyes, helping to hunt in sea during the polar night.
Eyes are surrounded with a ring of gristle plates protecting them from damages
during the diving. Plumage of this species is snow-white with black irregular-shaped
spots on breast and front side of neck. Feathering is considerably thicker,
rather than at other representatives of genus. Feather barbs do not form vane,
feather shaft is very short. As a result of such changes the plumage of this
bird is very similar to rich wool and also represents the additional protection
against cold along with a layer of hypodermic fat.
Hummock gannetwhale lives on floating ice sheet of Arctic ocean. It may appear
on firm ground only during summer migrations. This species is well adapted
to existence on ice field. The female lays egg in the beginning of polar night
and incubates it on paws, having covered it with wide tail. It lives mainly
due to stocks of fat, and presumes to itself to eat only some snow to satisfy
thirst. At this time male is actively fed, filling a full stomach of fish and
invertebrates. It may leave to the distance of about 100 kilometers from the
female for feeding, but it necessarily comes back to her approximately two
times per week. Having got out on ice sheet, males exactly find the females
hidden among snowdrifts. Having found out the female by its voice, male feeds
her, belching the swallowed food. For once he can bring up to 40 kg of food
in stomach. At the middle of winter at the female nestling is hatched. As against
to other species of these birds, the nestling is covered with rich down right
at hatching. At this time the female grows thin strongly – her organism starts
to produce a liquid rich in proteins and similar to “bird milk” of pigeons.
It is the first food of nestling, and female feeds it with these secretions
waiting for male with a stock of food. Till two first weeks the food is homogeneous
and resembles fat cream, but further the female starts “mixing” it with the
semidigested rests of food brought by male. When the nestling reaches the weight
of about 10 kg (at the age of two months, approximately in first half of March),
female leaves it in shelter representing a snow burrow. Parents go to the sea
for fishing and come back by turns to feed it. On breast and stomach of nestling
skin is thicker, and fat deposit provides good insulation. At this time uninterrupted
feeding is important for nestling – it should grow with the greatest possible
rate. Before summer will come and ice will melt, nestlings of hummock gannetwhale
reach weight of 200 kg and fledge. At this time they start swimming for the
first time, but yet are not able to get food independently, and parents feed
them up till about one month.
Hummock gannetwhales eat fish and cephalopods, and also crustaceans and echinoderms.
Birds of this species can dive to the depth of 150 – 200 meters and to hold
the breath for half an hour. They search for food with the help of sight sense.
Hummock gannetwhales migrate in Arctic Ocean with currents, stopping for a
short while at the northern coast of Greenland and at polar islands near coast
of Eurasia. At this time adult birds are fattened intensively in order to save
up stocks of fat to new breeding season. Sexually immature birds of various
ages wander near edges of the ice shield of Arctic Ocean and regularly winter
at the south, at the islands or at the coast of the continent. They become
capable to breeding at the age of eight years. Adult birds do not come far
to the south, and keep in area of drift ice. If ice-holes freeze, the female
can move closer to edge of ice, holding egg or nestling on paws. Life expectancy
of birds of this species may reach 60 years.
The idea of existence of the present group of birds was proposed by Dougal Dixon.
Wabun
(Crocorax wabun)
Order: Suliforms (Suliformes)
Family: Cormorants (Phalacrocoracidae)
Habitat: freshwater reservoirs of North America in the subtropical climate.
The boundary of two epochs, the Holocene and the Neocene, was marked by mass
extinctions associated with very drastic climate changes. Extensive glaciation
in the Northern Hemisphere forced the inhabitants of temperate latitudes to
move further south, following the changes in the boundaries of climatic zones.
Later, when the glaciers retreated again, many species returned to the lands
of their ancestors, but some of them simply adapted to changing conditions in
new habitats. Thus, representatives of groups of animals and plants more characteristic
of the polar regions appeared in low latitudes. Wabun is one of these species.
Wabun is one of the descendants of the double-crested cormorant (Phalacrocorax
auritus) of the Holocene epoch. Even in the human era, this bird had significant
adaptability and was represented by various subspecies; in the Neocene, several
new species, differing in ecology, evolved from this species. One of the species
is wabun, which inhabits rivers and lakes in North America.
The wabun is an aquatic bird about half a meter long. The coloration, like that
of the ancestral species, is dark (males are darker than females), but not monotonous,
but with a pronounced pattern of longitudinal stripes on the neck and chest.
This feature helps the bird to hide in the coastal thickets. The characteristic
feather crests of the ancestral species have turned into two narrow feather
“tufts”; the skin on the face is bare, of meaty-red color. The wings are small,
but the bird retains the ability to fly; the wabun cannot fly for a long time,
but at short distances it flies fast enough to escape from an underwater or
ground predator. If necessary, the bird jumps out of the water into the air
and immediately takes off, despite the wet plumage. Wabun escapes from aerial
enemies underwater: it is as good a diver as his ancestor. On the ground, this
bird is very careful: because of the long webbed toes, wabun walks very clumsily,
although it is able to take off from the ground.
The enemies of the wabun are not numerous – mainly predatory fish and snakes;
the meat of this bird has quite unpleasant taste, so many birds of prey and
carnivores do not hunt it, but ravage its nests in search of eggs and chicks.
Wabun itself hunts small pelagic fish, insects and other imvertebrates, as well
as amphibians. The bird will attack bottom-dwelling fish and crayfish less often,
preferring to hunt in open water.
Wabuns are sociable birds forming not numerous loose colonies. Each pair arranges
a nest individually, but relatively close to each other, and does not show aggression
to neighbors during hunting. In addition, they jointly defend themselves from
enemies, raising the general alarm and supporting each other with loud calls.
This is a non-migrating bird species that lives where large bodies of water
(especially rivers) usually do not freeze for the winter, or do not freeze completely
and for a short time. In winter, these birds search for wintering pits of fish,
diving to a depth of 20 meters.
Nesting begins in spring. Even at the end of winter, males of this species begin
to court of females, hunting together and feeding their mates with fish. The
courting male performs somersaults under water and often jumps into the air
vertically, and then falls into the water. During such courtship games, the
females assess the physical condition of the males before reciprocating. This
species is monogamous, and partners often stay together for life.
Wabun nests are a bit similar in structure to the great crested grebe’s nest
of human era. A pair of birds throws a bunch of twigs into the thickets of water
plants until a floating island is formed on which a nest is arranged. There
are no more than 6 eggs in the clutch; they are incubated by both parents in
turn. The chicks hatch by the end of spring, and by the end of summer they are
already able to swim and dive along with their parents. Usually young birds
overwinter with their parents, but in the spring the families break up, and
breeding pairs renovate the nests and make a new clutch. Sexual maturity occurs
at the 2nd year of life.
The average life expectancy of wabun is 10-15 years.
This bird species was discovered by Bhut, the forum member.
Ta'aroa
(Taaroa involans)
Order: Suliforms (Suliformes)
Family: Cormorants (Plalacrocoracidae)
Habitat: Eonesia, Fisaga Islands.

Picture by Alexey Tatarinov
Eonesia islands originated in the Pacific Ocean after the
human era, so they were settled in natural way by default. In the Neocene, their
nature is radically different from the nature of the islands that existed in
the human era, which are inhabited mainly by descendants of species introduced
by people. Seabirds were among the first settlers on the new islands. In the
new habitats, peculiar ecosystems were formed, in which the descendants of the
settlers evolved much more freely than on the continents, in conditions of intensive
competition and the presence of enemies. Therefore, island species turned out
to be peculiar in comparison with their mainland relatives. So, miru
(Pezolarus miru), a poorly flying gull, appeared on the Burotu Islands.
A large descendant of cormorants – ta’aroa (the name of this species comes from
the name of the Polynesian god of the sky) appeared on the young volcanically
active Fisaga islands. This species is a large flightless cormorant, forming
large colonies on the shores of the islands, numbering up to 300 adult birds
and more.
The loss of the ability to fly in island habitats is relatively easy: in the
human era, there were island-dwelling flightless cormorant species, which, however,
became extinct in the human era or immediately after it. Ta’aroa's body length
is up to 85-95 cm, weight about 4.5 kg. While walking on land, ta’aroa keeps
the body in almost erect posture. The wings of this species are shorter than
those of other cormorants that have retained the ability to fly. The primary
feathers are very strong and shortened, so the wings are a bit like the flippers
of penguins. On the bird’s head there is a short serrated corneous crest, similar
to a crown, under the beak on the throat pouch a red skin “wattle”, pierced
with blood vessels, grows – it is an adaptation for cooling when the bird is
on the shore. The area around the eyes and from the eyes to the beak is covered
with featherless light gray skin. The plumage color is mainly dull – the background
color is brown, the wings are black, the belly is gray. The skin on the legs
is black.
Taaroa is a monogamous species; pairs are formed for life and birds form a new
pair only after the death of a partner. This species feeds mainly on fish, crustaceans
and mollusks, does not disdain carrion and bird eggs. Sometimes taaroa attacks
small vertebrates caught on the coast – lizards, large insects and land crabs.
The bird swims well, flaps its wings under water, and paddles with webbed feet
on the surface. Some individuals swim across the straits between the islands
of the archipelago, so ta’aroa colonies exist on all the islands of the Fisaga
archipelago.
This bird species lives and nests exclusively in colonies, solitary birds experience
severe stress, when lack the society of relatives. Colonies are usually located
in the forest at a distance of several tens of meters from the beach. From the
colony to the sea well-trodden paths devoid of vegetation stretch. Each pair
has its own small nesting area. The mating season is very long; nesting birds
are present in the colony at any time of the year. Adult birds, preparing for
nesting, perform a mating ritual: they open their wings in front of each other
and call, raising their heads to the sky. Young birds also stomp on the ground,
lifting their legs high. The nest is very simple: it is just a hole in the forest
floor or in the sand. There are 4-5 eggs in the clutch, laid at intervals of
1-2 days. Incubation begins from the first egg, the chicks hatch after 30 days.
Development is relatively slow. For the first month and a half of their life,
they stay in the nest and are fed by their parents. At this time, quarrels often
break out in colonies: either one of the adult birds got too close to someone
else’s nest, or a chick penetrated to the neighbour’s brood, trying to get an
additional portion of food. Later, the young birds leave the nest and accompany
their parents to the sea, waiting for their return, then begin to swim with
their parents to the sea for fishing. At this time, they often become prey of
sharks. Ta’aroas become independent at the age of about 8 months and reach sexual
maturity at 4-6 years (females do it earlier). Life expectancy is very long
and can reach 50 years or more.
This bird species was discovered by Nick, the forum member.
Social
hammerkop (Scops socialis)
Order: Pelecaniforms (Pelecaniformes)
Family: Hammerkops (Scopidae)
Habitat: Africa, Madagascar, swamps and river banks.
The majority of stork birds had gone through the mass extinction had taken place
at the boundary of Holocene and Neocene. Exemption of ecological niches because
of extinction of various carnivores, and also because of extermination of some
species by people had permitted to representatives of order to evolve to new
species. But the part of stork birds had remained to live in the same place,
as earlier – near to water, in marshes and bush. Nevertheless, they also had
continued to evolve.
One of usual marsh birds in tropics of Old World in Neocene epoch is the social
hammerkop, the descendant of ordinary hammerkop (Scops umbretta). This bird
not so strongly differs from an ancestor in size and anatomy, but the habit
of life of this species is principally other.
The colonial hammerkop is rather small bird: it is like small hen in size, but
seems larger because of long legs and large head. The feathering of this bird
is colored imperceptible brown color, on back and stomach it is a little bit
lighter. Legs are naked, yellow, with tenacious toes and sharp claws; wings
are short and rounded. This bird reluctantly flies, but quickly runs on the
ground and is able to climb on trees dexterously, not using wings.
On the head of bird there is a characteristic crest, visually “equilibrating”
sharp chisel-looking beak. The colonial hammerkop is carnivorous, similarly
to the ancestor: it eats fish, frogs and shrimps, trapping them from tree roots,
or standing in water.
Main difference of this bird from the ancestor is the way of nesting. Colonial
hammerkops place their nests looking like huge spheres of branches and twigs,
on undersized branchy trees or bushes; the entrance is directed upward. In places,
where rivers floods happen oftenly, birds arrange nests only on trees, at height
about 3 – 5 meters above the ground. Inside the common nest there are some nesting
chambers in which some birds live at once: pair of adult birds and their posterity
of the current year. When the parental pair starts to nest, young birds leave
their dwelling, and start to build their own shelters. Sometimes they even attach
their nest to parental one. Thus birds form true colonies which inhabitants
are connected by relationship. In colony it can be totaled up to 50 and more
nests, located near to each other in rather small territory – about 1000 square
meters. The old colony of these birds forms practically integral construction.
The colony of these hammerkops may be found in rich thickets by strong noise.
Voice of these birds is loud, hoarse and unpleasant, similar to crow’s croak.
Usually birds do not worry that anybody will notice them: they choose a place
for nesting in remote places protected by bogs or river channels. But if birds
will notice a predator, they frighten it off by all colony, loudly crying and
striking to it impacts by beaks. In the afternoon when the most part of birds
departs to feeding, some “sentinel” birds stay in colony.
In nest of the social hammerkop it may be only two – three large (goose egg-sized)
white-shelled eggs. Nestlings hatch covered with down, but blind. At the age
of four days at them eyes open and they start to move in nest chambers. At fortnight
age when at nestlings feathers on wings start to grow, they move out from nest,
and wait for parents on its top.
The social hammerkop is widely spreaded in Africa and Madagascar, forming some
subspecies, distinguished from each other by size and colouring. In Madagascar
and Zinj Land eastern or Madagascar subspecies S. s.
madagascarensis lives. It is the smallest subspecies: adult birds are
only large pigeon-sized. It differs in lighter colouring of stomach; its beak
and legs are light yellow. Its nests are rather small – about one meter in diameter,
but colonies of this subspecies are more numerous, and total up to 80 – 100
nests. Continental African subspecies S. s. socialis meets
along Sahara Nile and other rivers; it is larger (the description mentioned
above concerns to it). These both subspecies live near freshwater reservoirs,
coming only in freshened enough sites of mangrove thickets.
The separate sea species, the Arabian
social hammerkop (S. arabicus), differs in sand color feathering. By
size it does not surpass the African continental subspecies of social hammer
cop. This bird lives in mangrove thickets along the coast of western part of
Indian ocean. The area of this species includes the Arabian coast and the north
of Zinj Land. At the coast of northern part of Tanganyica passage the area of
Arabian social hammerkop borders on areas of both subspecies of the African
species, but the competition between them never takes place, because this species
lodges at external edge of mangrove thickets, and searches for food (fish, shrimps,
molluscs) at the sea coast. Nests of this species are constructed stronger:
they are twisted in branches of mangrove trees, and can maintain a gale.
This species of birds had been discovered by Simon, the participant of forum.
Wide-billed
takatra (Takatra latirostra)
Order: Pelecaniforms (Pelecaniformes)
Family: Hammerkops (Scopidae)
Habitat: Madagascar, seasonal and permanent reservoirs, coastal forests.
After the human disappearance, the relative isolation of the various islands
was restored again, but with a new set of inhabitants, appeared there due to
human activity. Therefore, the evolution of life on the islands in the Neocene
is often the evolution of alien species that displace the endemic flora and
fauna of the islands, which in the human era could exist rather successfully
due to nature conservation activities. Only a few island species were able to
benefit from the changes in nature initiated by the introduction of alien representatives
of flora and fauna. In Madagascar, the invasion of exotic fish species has almost
completely destroyed the local fauna, and Neocene fish species are descendants
of introduced ancestors. And the evolution of fish contributed to the evolution
of new species of fish-eating birds. One of these species is wide-billed takatra,
a descendant of the local population of the hamerkop (Scopus umbretta). “Takatra”
is one of the local names for the hamerkop.
From a relatively small ancestor, a large shorebird with a powerful beak evolved.
The weight of an adult one is up to 3 kilograms, the wingspan is about 2 meters.
Unlike many species of near-water birds, takatra has a robust constitution,
relatively short legs and neck, and a large head with a heavy strong beak. This
bird prefers to walk and run on the ground and is distinguished by strong legs
with firm thick claws, similar to chicken ones. Nevertheless, takatra has not
lost the ability to fly: it takes off hard, but it flies well, and sometimes
it can even soar, especially in the dry season, when powerful updrafts rise
from the ground. The plumage of an adult bird is gray-blue: each feather is
covered with very thin cross ripples of gray color, which gives the impression
of a general bluish shade of plumage when viewed from afar. The head and shoulders,
as well as the wing coverts, are darker due to denser ripples. The tail feathers
are short, with wide tips of gray color. The wings are broad; primary feathers
are white with a dark base. Bird’s legs are covered with bluish skin. The male
is slightly larger than the female.
In its appearance, takatra has preserved some of the ancestor’s features: a
large erectile crest of thread-like feathers grows on the bird’s head. By rising
and folding it, the birds display their emotional condition. The takatra’s beak
is very remarkable – it is long and wide, like that of the shoebill (Balaeniceps
rex) of the human era. The color of the corneous cover of the beak is grayish-yellow.
The tip of the beak is bent to a powerful black hook. Numerous “teeth” grow
in the bird’s upper jaw: these are strong outgrowths of the cornea of the upper
mandible, allowing it to hold prey. In the lower jaw, they are only in the back
of the mouth.
This bird species feeds mainly on fish, less often eating frogs and large invertebrates
– insects and crustaceans. The fish species introduced by man rapidly evolved
in the conditions of island isolation, and the diverse fish population of Neocene
reservoirs of the island provided an excellent food base for water birds and
takatra in particular. This bird species has adapted to eat large fish, so takatra
is found mainly where there are large permanent or seasonal reservoirs. Snakeheads
and other fish capable of burrowing into the ground are particularly desirable
prey of takatra. The bird searches for them in dried-up reservoirs and digs
them out with its beak, shoveling dry mud with its feet. Usually takatras are
solitary hunters, but in the wet season, with an abundance of prey, they are
tolerant of each other. The relationship between them is greatly complicated
in the dry season, when competition for food increases. At this time, males
aggressively expel other birds from their territories.
Takatra’s voice resembles a squeaky laugh. Courting the female, the male stretches
his neck forward, raises the crest and makes ringing rattling sounds. He “sings”,
pacing back and forth in front of the female, interspersing calls with flapping
wings. The female, ready for nesting, quietly “responds” to the male.
Takatra is monogamous, but a pair is formed for one nesting cycle only. This
species has retained the building skills of its ancestor: the nest of takatra
is very large and covered, woven from branches. It consists of two chambers,
one of which is a nesting chamber – different females choose different chambers.
The nest is located on a tree at a height of up to ten meters, and it is owned
by a male who changes females with each new season.
Nesting begins shortly before the rainy season. Males call for females and choose
a mate for one breeding cycle. Before nesting, the couple renovates the nest,
repairs its walls and drapes it with vines of creeping plants. Epiphytic ferns,
specially brought by birds from the forest, often grow on the roof of the nest.
They take root well and grow on the roof and walls of the nest, masking it from
the air from eyes of birds of prey.
There are 2-3 eggs with a white shell in the clutch. The female incubates them
almost exclusively for 35-36 days. The chicks are covered with down and have
eyes opened, but remain in the nest for up to 2 months. Parents feed them on
small fish and frogs. Young birds fully fledge and leave the nest by the age
of 4 months. As a rule, parents manage to raise all the chicks that are not
aggressive towards each other. Juvenile plumage is darker than adult one – the
feathers of young birds are monotonous gray. At the age of 3 years, birds become
sexually mature. Life expectancy can reach 30-35 years.
Hanging
heron (Breviardea pendula)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: Equatorial Africa, gallery forests in Congo basin.

Picture by Alexander Smyslov
Congo basin is the African analogue of Amazonia in variety
of lifeforms. On marshy ground forests grow, which are flooded till some weeks
a year. The large efficiency of biotops permits to exist to specialized species
of animals which had developed original life strategies as a result of evolution.
On branches of trees hanging down above water, the small bird perches. It has
long beak and strong mobile neck. It is a special species of herons living only
in flooded forests of Equatorial Africa. This bird traps prey, having seized
in branches touching water surface. When prey is shown in sight of this heron,
the bird seizes it, not letting out a branch from toes. Having bent legs in
joints, bird pulls out prey from water. Such feature of behaviour has determined
the name of bird – hanging heron.
It is rather small species of herons – it is pigeon-sized
one. Proportions of body at hanging heron are not characteristic for shape of
heron – at this bird legs and neck are shorter, rather than at other herons.
Toes at this bird seem disproportionally long. They are very tenacious: due
to them hanging heron is able to swarm up branches of trees dexterously. Beak
at this bird is long and straight.
The feathering of hanging heron has camouflage colouring: it has soft brown
color with black cross strips. Stomach has rusty color, area under tail is white.
Head of hanging heron is dark brown; around of eyes there are rings of naked
bluish skin. On top of the head of bird the cop of several long narrow feathers
of white color grows. Beak and legs at this bird have yellowish color.
Male and female at hanging heron do not differ in color of plumage, but female
weighs approximately 20% heavier, rather than male. Pairs at these birds formed
to one nesting season, and break up, as young birds become independent. In courtship
season at the male similarity of egrets – long feathers with fluffy barbs –
grow on back. Male displays these feathers to the female, “bowing” and stretching
these feathers like a fan.
Hanging heron does not form numerous colonies that is typical for other species
of herons. The single nest of these birds is rather large – its diameter reaches
two meters. Birds can re-build for themselves the abandoned nest of storks or
birds of prey, or construct an own nest. In the basis of nest long firm branches
lay, on which adult birds put a heap of rods. The bottom of nest is covered
with grass and dry water plants.
In clutch of hanging heron there are up to three eggs. Nestlings hatch blind,
but covered with rich grey down. In some days they start to see and become more
active. At week age nestlings of hanging heron already well swarm up branches,
and wait for parents bringing food out of nest. They completely fledge at monthly
age, and at the age of six weeks try to hunt independently. The three-monthly
young bird already tries to fly. Young birds nest at two-year-old age.
Hanging heron prefers to nest on the same tree, from which branches it hunts
fish. Besides of fish it willingly eats snails and freshwater crabs. Having
seized prey, it swarms to nest on branches though if necessary it is able to
fly well. Dust and dung thrown out from nest involves fishes under nest of hanging
heron, and this circumstance helps bird to hunt.
Laka,
or Hawaiian dancing heron (Ardea ballerina)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: Hawaii; nesting in coastal forests, feeding along the entire coast
and throughout the islands, where there is an open area.
Laka, or Hawaiian dancing heron, is a descendant of grey heron (Ardea cinerea)
of Hawaiian population. In the process of changing the ecosystem of the Hawaiian
Islands under the influence of species introduced in the human era, herons survived
relatively successfully, and in due course of evolution in isolation, they developed
features of appearance and behavior that are not found in mainland forms. Bizarre
sexual selection turned the descendant of the herons of the human epoch into
a creature with bizarre behavior and remarkable appearance, Hawaiian dancing
heron.
This heron species is easily recognized among other birds by its colorful plumage.
The upper part of the bird’s body has bluish-gray color of various shades, the
underside – chest, belly and tail – is white. The head is white, but bright
red stripes stretch from the beak across the eyes towards the nape, which merge
there and turn into a red crest of several elongated feathers. The neck is light
grey with blue spots forming 2-3 longitudinal stripes.
The primary feathers of the wings are dark blue, but a little faded due to the
powder covering them, with which the bird rubs the feathers. The secondary feathers,
like the back, are bluish-grey. The mantle feathers are narrow and elongated,
hanging like a fringe on the wings and back. The color of these feathers is
light blue. The feathers on the throat and chest are elongated and become even
longer during the courtship season, forming a “wattle”, which is especially
noticeable in a perched bird.
Wide dark blue stripes stretch along the sides of the trunk. When the wings
of a heron are folded, the feathers form a prominent blue-white spot in the
area of the carpal fold. The tail is dark blue. Fragile powder down grows on
the chest, belly and groin. Its powder is applied by the heron with the help
of an elongated and serrated claw of the middle toe. During the courtship season,
the dresses of birds become especially bright, both in males and females: bright
green feathers appear on the back, and bright red on the chest. These feathers
shimmer in the sun as if bright emeralds and rubies are encrusted in the bird’s
plumage. At the end of the breeding season, the birds begin to molt, and the
plumage becomes duller.
The appearance of the dancing heron is very characteristic. Laka has a very
long neck, which is often folded in a perched bird, and long and thin legs.
This is a relatively large bird: the average weight is about one and a half
kilograms, while individuals can reach two kilograms; large males can weigh
2.5 kg. Body length up to 100 cm (including head and neck). The wingspan varies
from 1.5 to 1.75 m, in some specimens up to 2 m. Sexual dimorphism in this species
is weakly expressed – the male is only slightly larger than the female.
The beak is pointed and quite long – 10-15 cm. It has a slightly pronounced
conical shape and is laterally compressed. The color of the beak is dark blue,
while the upper mandible is usually darker than the lower mandible; the patch
of skin on the lore (the area between the eye and the beak) is reddish. The
legs are white, with bare skin.
Flying laka folds the neck in the form of a Latin letter “S”, so that the neck
loop lies on the back, and stretches the legs far back. When taking off, the
heron, especially startled, makes quick flaps of its wings, gaining altitude.
Having flown high enough, the bird switches to a slow and smooth flight, with
regular wing flaps. Occasionally it hovers, especially over the fields of hardened
lava, which are well warmed up and form ascending streams of air. During flight,
groups of herons often form a skein or line. Due to its ability to fly, this
species is found on all the islands of the Hawaiian Archipelago.
Dancing heron is an exclusively zoophagous bird. It is an active and very voracious
predator, eating almost all animals that it is able to cope with. Birds of this
species feed on the coast of the ocean and freshwater reservoirs or on land
– in an open area.
During low tide on the ocean coast, in mangroves and in the recesses of coastal
reefs, lakes of water remain, into which small fish and small crustaceans (crabs,
shrimps), as well as small octopuses and other mollusks are trapped. These lakes
are the main dining room of laka. Small marine animals, which have nowhere to
escape, turns out to be easily accessible prey of the heron, and even attempts
of shrimps and crabs to burrow into the sand do not save them from the sharp
gaze of the heron. As a rule, this heron smashes crabs and clam shells, and
also kills large and resisting fish by hitting against the nearest rock or reef
ledge. Often these birds arrange feasts, eating seal
mudskippers, especially young individuals, on the shore. It can also diversify
its diet sometimes by eating eggs or chicks, grabbing birds nesting on the shore.
Also, dancing heron can be seen on the coast of freshwater reservoirs, in the
swamps of Hawaiian Islands, where the bird also eats shellfish, arthropods,
fish (including eggs and fry), amphibians (including their eggs and tadpoles),
reptiles, small birds (as well as their eggs and chicks) and small near-aquatic
mammals.
Sick and parasite-infested fish swimming near the surface often fall prey of
these birds. Thus, the bird becomes an intermediate host of various parasites
and pathogens and plays a role in the spread of helminths of fish (flukes, nematodes
and tapeworms). But in the birds themselves, the mortality rate from helminths
is not very high.
It can also hunt on the edges of the forest and in the meadows of the Hawaiian
Islands, eating everything that moves and what it can kill – from small insects
to rodents (kapo). This heron
can often steal part of the prey from other predatory and fish-eating birds,
as well as from scavengers. The heron also does not disdain any carrion – it
knows how to tear apart a large carcass, tearing off pieces of meat with jerks
of the head. It also feeds on land amphibians and reptiles. Undigested remains
of eaten animals (chitinous integument, shells, eggshells, bones, feathers,
wool, etc.) are regurgitated in the form of compressed pellets.
The most frequently heard voice signal of a heron is a rough rasping call, similar
to a low short cawk. It is usually uttered by a heron on the fly. It is very
loud and can be heard at a great distance. There are a number of other voice
signals at the heron, but they can be heard almost exclusively at the nest,
in colonies (outside the colony they are usually silent). The alarm signal is
a guttural cackle. A prolonged throaty vibrating cry serves as an expression
of threat and a sign of aggressive intentions. The male, indicating his presence,
utters a short hollow cawing. Gathering in nesting colonies in large groups,
herons constantly utter cawk and croaking sounds. The landing heron utters a
sharp rasping cry, usually repeated.
The dancing heron also has a developed system of visual signals. With the help
of its long neck, the bird can express various emotions. The threat pose is
characteristic, in which the heron bends its neck, as if preparing for a rush,
and raises the crest on its head. This pose is usually accompanied by a threat
sound. Birds can greet each other by clicking their beaks quickly. It is also
used during the mating ritual. In general, the mating ritual of birds is a rather
complex and very beautiful dance. For this reason, the bird is called the dancing
heron, or laka, after the Hawaiian goddess Laka – the goddess of dance, love
and the guardian of the forest. During the courtship season, the coast is filled
with many cawing birds; several males dance around each female, opening their
wings wide and showing a wide chest, lifting their legs, as well as making sharp
head movements and ritual pecking of sand – in this way they display the health
and speed of movements of a successful hunter. Having chosen a male, the female
performs a return dance around her chosen one. However, it is only a preliminary
choice, which is supported by ritual feeding of the female by prey – fish, crab
or octopus. At this time, other males also rush to her with their prey: each
of them still has a chance to defeat their main rival by bringing more attractive
prey. At the same time, there is a fierce brawl between males for any large
prey. After the males return to their chosen one again, the female makes the
final choice among all the suitors around her. And in this case the choice not
always falls on the first chosen one. Such a complex two-stage process of choosing
a mating partner is explained by the fact that the female chooses a male for
the rest of her life and therefore it should be the most deserving one. After
that, the couple moves to the forest (either mangrove or swampy jungle) closest
to the coast and finds a suitable tree to build a nest. Birds nest in a tall
tree, often in the forest canopy. The male is usually engaged in choosing a
place and expelling possible competitors, but he had to change several places
for the nest before the female makes the final choice. When both partners like
the place for the nest, the male brings several branches to lay the basis of
the future nest. Having put them in place, the birds mate and then jointly build
a huge nest of large thin dry branches. The bottom of the nest is lined with
reed stems. The height of the nest is on average 50-60 cm, the diameter is up
to 2 meters. Females take part in nesting from the age of one year, and males
have a chance to form a pair only at the age of two or even three years.
Hawaiian dancing herons lead a sedentary lifestyle without leaving their traditional
habitats. The attachment of herons to their nesting place is very strong. Often
the nests are used for several years and are repaired every next year. Such
buildings are placed as if on a high basis and reach more than two meters across.
In some places, heron colonies can persist even for several centuries. These
herons are often kept in groups that can reach a significant number. Heron colonies
can be very numerous – from hundreds of individuals or more; birds of other
species can join them. The largest and oldest such bird colony is located on
the Big Island. This colony is more than a thousand years old and it numbers
up to two thousand birds.
Males of the dancing heron have a high level of aggressiveness; they are extremely
zealous in protecting their nests from other males. At the same time, the colony
is characterized by collective protection of nests and offspring from birds
of prey and tree-climbing predatory mammals. Although often eagleravens can
settle near a colony of dancing herons. Interestingly, in this case, the predator
perceives the colony as “its own” and does not hunt the heron chicks of “its
colony”, but can easily raid neighboring colonies. Also, local owls can settle
in the colony, preferring places below the herons’ nests.
It also happens that a young couple creates a new nest isolated from other herons,
and the nest of such a pair, as a rule, is built better than the lax nests of
colonial birds. But over time, a small colony of children, grandchildren and
great-grandchildren of the progenitor couple forms around this nest. Not every
such small colony is destined to grow to a megacolony: as a rule, the more the
colony grows, the more often there may be outbreaks of various diseases. There
are cases when epizootics mowed down up to 2/3 of the colony’s individuals in
especially large colonies.
The number of eggs in laka’s clutch is from 3 to 9, most often 5 or 6. The size
of eggs is on average 60 × 45 mm, the largest ones are 60.5 × 47.5 mm. The color
of the eggs is greenish-blue, often with white lime smears. Interestingly, heron
eggs do not always have the usual “egg-like” shape: they are often equally pointed
at both ends. The shell of freshly laid eggs has no luster, but if the eggs
are already slightly incubated, the shell acquires a slight luster. Egg laying
occurs with an interval of two days, and the female starts incubating immediately
after laying the first egg. Therefore, the chick hatched from the first egg
is always much larger than the subsequent ones, and even more so the last one.
Incubation lasts up to 21 days. Both parents incubate, but the female spends
more time in the nest than the male. The offspring of laka is of altricial type.
Chicks are sighted, covered with down, bluish above, whitish below the body,
their throat and belly are bare; feathers begin to grow on the 7-9 th day. The
newly hatched chick weighs 40-45 g. Dancing herons feed the chicks with food
regurgitated from the stomach. Food, before getting to the chicks, must necessarily
be already semi-digested.
At the daily age, chicks eat an average of 15.8 g of food per day, at the three
day age – 29.2 g. For the first five days, chicks only rest and almost do not
move in the intervals between feedings. At the age of one week, the chicks have
their own thermoregulation. Laka chicks are getting more active every day. In
the first week they only move in the nest on bent legs, but at the age of two
weeks they can stand on their feet and flap their wings. They activate the daytimevocalization;
the sound signals become diverse. In the feeding behavior of chicks, elements
of the hunting behavior of herons become clearly noticeable – aiming and rapid
rushes to capture food.
The main growth of the chicks’ body weight occurs before they take wing. They
grow most intensively at the age of 7-20 days. The average weight of a monthly
chick is 1 kg; a two-month-old one weighs 1.5 kg. Up to 20 days of age, the
development of the forelimbs and hind limbs is in the same rate, but then the
development of the wings begins to outpace the growth of the legs and other
parts of the body. This is due to the fact that in the wild herons move mainly
through the air, but their legs carry much less loads, therefore by the end
of the breeding period, the formation of wings is completed first of all. After
the leaving nests, the growth of other parts of the juvenile’s body continues
for some time. Young birds that have left the nest outwardly look almost like
adults. The final pass of young herons to independent life occurs at the age
of 3 months.
Each of the parents brings food to the chicks three times a day. However, not
all chicks receive food for one feeding. With a lack of food, older chicks can
kill and eat younger ones, which are inferior to them in size and strength.
“Freeloading” is also common, when older chicks take food from younger ones,
which leads to exhaustion and death of the latter. Younger chicks often die
from beatings by their elder siblings. Competition for food is expressed in
chicks early and is noted up to 35-36 days of age. Not only the older chicks
of the same brood can take food from the chicks, but also young herons from
neighboring nests. In general, the mortality rate of heron chicks is great.
The death of chicks often also occurs because adults often leave the nest when
alarmed.
The average life expectancy of a dancing heron is 5-8 years. Mortality of young
herons for various reasons is extremely high and especially significant in the
first year of life – up to 67%. From the second year of life, mortality decreases,
and in adult birds it is insignificant.
Due to the fact that the adult laka is a large and strong bird with a pointed
beak, it is able to defend itself from many enemies. However, even adult herons
often fall prey to predators.
The chicks are attacked by the erinia bat, neopueo,
Hawaiian bird-eating snail
or burning snail. Hawaiian
eagleraven can kill chicks and even a grown up young bird. An adult bird
feeding on the ground can be caught off guard by kuahana,
and a young bird – by a relatively large mongoose.
Colonies of herons attract a lot of ground-dwelling predators that pick up under
the trees chicks that have fallen out of the nest or catch young ones that are
still poorly flying, but are already trying to move inside the colony. Predators
are attracted to the colonies by the remnants of the brought food, which the
young herons did not have time to eat, falling abundantly from the nests. These
are mainly kuahanas and Hawaiian mongoose
species. Burning snails, monstrous
ants and other detritophages are also attracted by the herons’ regurgitated
pellets lying on the ground in great numbers, on which undigested pieces of
food remain. And snails and ants feeding under herons’ nests on the ground are
eaten by Hawaiian hookbills and
ant starlings, which
in turn are hunted by the neopueo and erinia bat.
This bird species was discovered by Wovoka, the forum member.
Papa-ratos
(Ardeola rattophaga)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: the coastal areas of Northeastern and Northern South America, Archipelago
of Fernando de Noronha.

Picture by Pavel Volkov
The birds were always one of the most mobile animals. With
their ability to fly, they were capable of going beyond their normal territory,
this way colonizing new areas. During the Neocene, the squacco heron (Ardeola
ralloides) had reached the Archipelago of Fernando de Noronha and the coast
of Northeastern Brazil coming from Africa and Europe. In the Neocene, a population
of this species remained here and gave origin to the papa-ratos. This name was
used for their ancestor in Portugal and means rat eater. The papa-ratos inhabit
the Archipelago of Fernando de Noronha and the coastal areas of Northeastern
and Northern South America.
The papa-ratos have 47 cm of length with a wingspan of 92 cm. When adult, its
coloration is a mixture of dark brown and orange on the back and flanks, abdomen,
lower back, wings and white tail, which becomes surprisingly apparent during
flight. It has a nuchal crest with elongated reddish-brown feathers and black
streaks that hang down the back of the neck. In the breeding season, the plumage
is more vivid, with an orange-colored crown, golden feathers on the lower part
of the neck and back, reddish-golden wing and chest-coverts, reddish-brown back,
with long golden plumes, and the rest being white plumage. The bare parts become
bright during courtship, the legs turn reddish with soon fading to pink and
back to dull yellowish-green, bright blue bill with dark tip that is pale greenish-yellow
with black tip and jaw in non-breeding season, blue face and yellow eyes. Juveniles
are less colorful, having bars on the head and neck and darker brownish back
than adults, no crest or back feathers, dark-tipped bill, gray underparts instead
of white and brown flight feathers.
It feeds on almost anything that moves, such as fish, frogs and their tadpoles,
snakes and small reptiles, small mammals and birds, molluscs, insects and their
larvae. However, they developed a preference for rodents, being capable to actively
chase these animals through the ground. Their ability in hunting rodents was
what gave them the name papa-ratos. The population of Fernando de Noronha learned
that if they stay near the colonies of sea birds, they will be able to catch
Noronha rats that came here to feed in the eggs and unattended chicks. Of course,
they will also eat chicks if the opportunity arises, but the sea birds mostly
tolerate their presence because of them hunting the nuisance that are the rats.
The papa-ratos are active during most hours of the day, but those of Fernando
de Noronha are more nocturnal due to this being the time that their main prey,
the Noronha rat, is mostly
active. It nests in large colonies, sometimes interspecific. The nest is a platform
of branches or reeds, well constructed, made in trees, near or on the water.
The nest is built by both sexes, and the material is collected by the male.
They usually lay 3 to 4 blue-colored eggs, with an incubation period of 24 days,
with the incubation being made mainly by the female. The chicks are fledged
after 35 days. Sexual maturity occurs around 2 years of age and they have a
lifespan of 12 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Doctor
heron (Neobubulcus medicinalis)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: various biotopes of Africa, Zinj Land and Madagascar.
In the Holocene, the western cattle egret (Bubulcus ibis) often accompanied
large mammals, eating parasites in their wool. Due to the high number and huge
range, as well as the ability to live in cultural landscapes, this bird has
persisted to Neocene. One of its descendants specialized in eating parasites
of large animals, expanded the range and broke with the aquatic habitat, and
so a species of doctor heron evolved – a common companion of many large animals
in the tropics of the Old World.
Doctor heron is a very small heron: the length of its body is 12 cm, the wingspan
is about 25 cm. However, this heron has long legs with thin toes and tenacious
claws, and a long thin beak and neck, which makes it seem larger. Head of this
bird is small, tail is short; face is not feathered. Doctor heron has a camouflage
plumage color – a yellow-brown background with longitudinal brown strokes, with
which the large red crest on the head contrasts vividly. The legs and beak are
dark gray; the skin on the face is yellow-orange. This bird has very well developed
eyesight and keen hearing.
Doctor heron inhabits various biotopes of Africa, Zinj Land and Madagascar –
from semi-deserts to tropical rainforests. It feeds on invertebrates that parasitize
mammals, birds and reptiles longer than a meter. It is actively looking for
a potential “client”, keeping near the noticeable features of the terrain –
single trees, flowering shrubs, or standalone stones and termite mounds. Attracting
“clients”, the bird perches in a prominent place, tramples with half-open wings
and raises its crest. At this time, doctor heron feeds on both herbivorous animals
and predators. Usually it walks slowly on the client's body and looks for various
parasites in its skin coverings. The heron’s diet includes skin parasites –
ticks, fleas, lice, subcutaneous botfly larvae, as well as flying dipterans
– flies, mosquitoes, horseflies, adult gadflies, etc. On the aquatic reptiles
that have crawled ashore, doctor heron also pecks off the blood-sucking crustaceans.
But it has not lost the ability to search for food in the environment without
the help of a large animal.
Most likely, this species originated in Africa and then settled to Zinj Land
and Madagascar. In North Africa, doctor heron usually feeds on large birds and
reptiles to avoid competition with weaverbirds.
The breeding season is not expressed, pairs form only for one season. Both birds
build a nest on a tree from grass stalks, twigs and their own feathers, although
they often occupy the nest of passerine birds or pigeons, destroying sometimes
their clutch and expelling the nest owners. Both parents incubate alternately;
there are 3 eggs in a clutch. Chicks hatch in two weeks. Parents feed them with
large insects (often not only parasitic), and chicks grow quickly,getting abundant
feeding. A month later, the juveniles are already fully fledged and learn to
fly. Soon after, they fly away and the parent couple breaks up. During the year,
there are most often two nesting cycles.
Sexual maturity occurs at the age of 7-8 months; life expectancy reaches 15
years.
This bird species was discovered by Mamont, the forum member.
Rail-like
heron (Paraardea ralloides)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: Galapagos Islands, mangrove and cactus forests.

Picture by Pavel Volkov
Rail-like heron is the most unusual representative of the
genus of Galapagos herons – birds that appeared at the archipelago after the
human disappearance. Due to the small number of terrestrial mammals on the islands,
this bird leads mainly a ground lifestyle, has poor flight abilities, but can
run quickly and climb trees. In length, the bird reaches 60 cm, while it has
very robust constitution and weighs about 1.5-2 kg. Its neck and legs are short
by the standards of herons, and its beak is of medium length, rather thick and
pointed. The beak is equipped with serrations on the edges of the jaws.
The color of the plumage is dusky, brownish-gray with yellow mottling on the
head and chest; the bill and legs are yellowish-brown. Bird is similar externally
to the Holocene night heron. Male has a black “cap” on the head with several
long plumes on the nape, like most herons.
The bird is exclusively zoophagous, feeds on invertebrates (crabs, insects,
mollusks), fish, small rodents and eggs of other birds. However, its main food
specialization is crabs and mollusks with hard shells. To break their shells,
the bird uses its powerful beak. Usually the bird presses its prey to the ground
and kills it with several pecks, but it can also knock it against a stone or
a hard tree trunk until its shell cracks. Birds have favorite places for prey
processing – “anvils”, around which fragments of shells of prey accumulate.
Rail-like heron nests in June-July on trees; the nest is made of branches and
leaves and represents a massive, loose build, constantly renewed by adult birds.
These herons are monogamous; pairs are formed for the rearing of offspring and
break up after the end of nesting cycle. Herons lay 3-5 eggs, incubate for about
20 days. Chicks grow quickly and leave the nest at the age of 2 months. Sexual
maturity occurs at 1.5 years; life expectancy is up to 15 years.
This bird species was discovered by Corvus, the forum member.
Black-headed
angler heron (Lophioardeola melanocephala)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: Meganesia, the south of Indonesia.

Picture by Alexander Smyslov
In human epoch a lot of species of animals had adapted to
inhabiting near to people. In tropical areas there are herons among synanthropic
species; they feed at fields and pastures, near cattle herds. After human disappearance
herons managed to keep sufficient variety, and in Neocene they keep strongly
their position among feathery inhabitants of freshwater reservoirs. In South
America one species of herons
turned to running predator, but it looks an exception in this group of birds.
Only in temperate and subtropical zone of Northern hemisphere herons are partly
displaced by crails (Rallogeranus
spp.) – crane-like descendants of small rails. But in forest reservoirs,
and even among mangrove thickets Neocene herons prosper and evolve.
In Meganesia there are two reservoirs, which had been a part of sea once: more
ancient by origin and almost fresh-water Carpentaria Lake and younger brackish
Arafura Lake. In extended zone of mangrove forests separating Arafura Lake from
ocean, the special variety of herons lives – it is small (equal to pigeon in
size) black-headed angler heron. It is the most widespread representative of
the genus of herons, developed during the evolution the interesting adaptation
for fishing.
In human epoch some herons fished, throwing in water any small objects – branches
and leaves – which involved fishes. Black-headed angler heron hunts in similar
mode, but it does not have necessity to search for suitable bait – this bird
always has it. One feather in cop of ancestor of this bird had almost completely
lost vane and had turned to fine “fishing tackle”. It became very long (its
length is about 25 cm), and the part of vane has remained only on its tip as
small black-and-white “tag”. The basis of this feather differs in mobility –
under skin some muscular fibres similar to mimic muscles of primates last to
it – they are attached by other end to skull of bird. Shaft of this feather
is flexible, thin and elastic. Heron is able to move it with the help of muscles,
and bird uses this skill during hunting. “Fishing tackle” to the moment of mew
is strongly tattered because of the often use – sometimes fishes manage to seize
it. During the mew bird does not suffer from famine – it catches prey in mode
usual for herons.
The bird lowers the tip of “fishing tackle” with bait in water, and starts to
move it, imitating movement of small fishes or shrimps near to surface of water.
Thus bird perches on roots or on tree trunk, having bent to water. From time
to time angler heron catches by beak fishes involved with its bait.
Angler heron keeps characteristic features of anatomy of this group of birds.
It has long legs with lengthened tenacious toes, mobile long neck and pointed
beak with tiny denticles on edges, helping to keep slippery prey. All angler
herons fly well and frequently make flights between islands of Indonesia. Wings
of these birds are rounded and wide. Colouring of this bird is remarkable: the
feathering on various areas of body has completely various colors, and this
heron seems tailored of two halves of different birds. Head and neck of black-headed
angler heron are completely black (hence its name) with bluish metal shine;
even corneous cover of beak is black. On this background eyes with shining yellow
iris are brightly emphasized. The body of bird is straw-coloured with black
longitudinal strokes on feathers. Primarily feathers have wide black cross strips.
Angler herons do not form separate colonies and prefer to nest in colonies of
other species. At small islands of Indonesia and in mangrove thickets of Arafura
Lake these birds nest in pairs in places protected from ground predators. Angler
herons eat small fishes, crustaceans and water beetles.
These birds are monodins. The pair forms for one breeding season, but till this
time birds have time to feed up in succession two hatches of 2 – 3 nestlings.
For courtship display at the male the courtship dress develops – long straw-coloured
egrets on waist. Legs at black-headed angler heron out of courtship season are
orange, but at displaying male they turn bright red. These birds prefer to occupy
and to repair old nests of other birds, rather than to build their own nest.
Involving the female, male searches for such nest, and starts the display. It
calls the female with cries similar to crow croak. When the female perches on
its nest or near it, male starts to display courtship dance. It widely opens
wings, showing striped colouring of primarily feathers, and fluffs egrets. In
such pose male bows forward and wings appear stretched in vertical plane. Male
continues courtship dance, holding in beak any twig and bowing to the female.
If the female accepts his court, she takes from him this twig and attaches it
to the nest.
Angler herons live in Meganesia, Indonesia and Southeast Asia. This genus includes
some species differing from each other in size and colouring.
Giant
angler heron (Lophioardeola gigantea) lives in Indonesia – at Jakarta
Coast and near islands. This bird avoids settling in mangrove thickets, because
it hunts only in fresh water. It lives at riverbanks and lake coasts and eats
fish and amphibians. It is the largest species of the genus – this bird weighs
about one kilogram, and its wingspan is 120 cm. Its plumage is greyish-white,
and head is covered with thin cross ripples and looks darker, than other plumage.
“Bait” on head of this bird is bicoloured – white with black tip.
Green
angler heron (Lophioardeola chloropluma) is common at islands of Indonesia
and lives mainly in mangrove thickets. It is small bird weighing no more than
200 grams at growth of about 30 cm. Its plumage has dark green color with brownish
shade on shoulders and back; head and neck are grey with longitudinal green
strips. Legs of this heron are rather short and tenacious, therefore it easily
swarms up roots and branches of mangrove trees and makes the way even through
the richest thickets. Green angler heron differs in boldness and curiosity while
it stays among mangrove thickets, but it is very timid, when it is compelled
to cross open area. Nevertheless, this species of herons frequently makes flights
between islands and consequently does not form any local forms along the whole
area.
Brush
angler heron (Lophioardeola penicillifera) has “bait” as loose vane
with long barbs forming brush of black color. It lives separately from an area
of other representatives of genus – at the south of Hindustan and Sri Lanka.
This bird has dim plumage – it is monotonous gray-blue with darker primarily
feathers. It is larger a little, than pigeon, and weighs about 400 grams. Brush
angler heron lives only near freshwater reservoirs – lakes and swamps.
Copper-winged
angler heron (Lophioardeola cuprea) lives in Southeast Asia and at
Jakarta Coast. It is small species: height of adult bird is about 30 cm. This
heron has reddish-brown plumage, and covert feathers of wings have faint metal
shine. “Bait” feather is colored white. Around of beak and eyes of this bird
there is featherless grey skin. Legs at male and female differ by color – at
female they are pale yellow, and orange at male. The present species of birds
lives at swamps and eats frogs, small fish and crabs. Copper-winged angler heron
does not avoid sea water and frequently settles in mangrove thickets.
The idea about existence of these species of birds was proposed by Simon, the forum member.
Bennu
(Nycticorax bennu)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: the basin of Saharan Nile, lakes and ponds of North Africa.
In the age of man, many species of stork birds suffered. Many large storks and
herons, as well as island local species have disappeared due to habitat destruction
as a result of human activity. But smaller species, such as the black-crowned
night heron (Nycticorax nycticorax), were able to survive due to their wide
range and ability to restore the population quickly. Having survived the ice
age at the boundary of Holocene and Neocene, these birds became the ancestors
of new species, among which there is bennu – one of the descendants of the black-crowned
night heron.
Externally, bennu is markedly different from the ancestral species; in proportions
it is a typical Holocene heron, larger in size than the ancestor. Its coloration
combines various shades of gray: on a light gray background there are darker
longitudinal stripes on the head and neck, and one stripe stretches from the
crown to the base of the tail. The cheeks and the inner side of the wings are
colored white. The legs, on the contrary, are very dark, almost black. On the
head, above the eyes, plumes grow, consisting of three or four narrow elongated
black feathers, which bennu can raise and lower: thus, birds of this species
court each other during the mating season and give signals to their relatives.
The beak is light yellow; its base is bordered by a ring of black feathers,
merging with the “cap” on the crown.
Bennu is a bird of rather large size: about 1 m high, with a wingspan of 2 m,
and weighing up to 3.5 kg. But compared to such neighbors as crocoturtle,
boaropotamus and giant
African stork, bennu looks a small and fragile creature; this bird leads
a rather secretive lifestyle. Bennu is mostly active in the morning and evening,
at dusk, and sometimes in moonlit nights. During the day, this heron usually
sleeps in thickets of reeds and papyrus, or on a tree growing near the water.
The bennu nest is usually located on a tree – it is a loose pile of branches
among the large branches of a tree, which can hardly withstand strong gusts
of wind. There are 2-4 eggs in the clutch; the shell is white with brown specks.
The clutch is incubated by both parents in turn: these birds are monogamous,
and although pairs break up after the breeding season, former spouses usually
stay close to each other, and last year’s pairs often reunite in the new nesting
season.
The breeding season for bennu begins in the second half of the dry season, shortly
before the first rains. At this time, the birds remain secretive, but their
loud two-syllable call – “ben-nuuu” (hence the name) – is heared quite far in
the vicinity of the nest. When a female comes to the displaying male, both birds
begin to dance around each other, jumping and showing the partner white feathers
on the inside of the wings, as well as displaying their “pigtails” by lifting
them and shaking them. At this time, fights between males of this species occur
more often – for females and for territory. Males beat each other with their
beaks, sometimes quite hard, but without fatal outcome.
Chicks are hatched at the beginning of the winter rainy season. They are covered
with white down, and in the first days of life their eyes are closed. At this
time, parents regularly replace each other at the nest – while one bird protects
the chicks, the second one forages for various fish, crayfish, other invertebrates,
frogs and lizards. At the age of about 2 months, young birds begin to fledge,
and at 4 months they already pass to independent life. By this time, the parents
manage to fatten them so much that the young bird weighs more than an adult
one.
The enemies of bennu are various birds of prey that hunt them in the air, and
crocoturtles that lie in wait for them in shallow water. Occasionally, monkeys
can ravage an unattended nest.
The average life expectancy of a bennu is 20 years.
This bird species was discovered by Bhut, the forum member.
Coal
black night heron (Nycticorax niger)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: freshwater reservoirs of North America in the areas of temperate and
subtropical climate.

Picture by Pavel Volkov
Although the boundary of Holocene and Neocene was a time of
mass extinction and climatic and ecological changes, many orders of living organisms
of the human epoch survived, although their species diversity has changed significantly.
Some of them have turned into small relict groups; others have increased species
diversity and occupied ecological niches of extinct animals.
Sometimes, in due course of evolution, new species changed their way of life
compared to their ancestors. Among the birds of freshwater reservoirs of North
America in Neocene, there are night herons, birds of a twilight lifestyle. Due
to their wide range, they were able to keep viable populations, and after the
mass extinction of some larger stork-like birds, as well as cranes, partially
occupied their niche of near-water birds. One of the widespread species is coal
black night heron, inhabiting reservoirs in the deciduous and mixed forests
zone of North America.
Coal black night heron is a fairly large bird: 75 cm in length, weighing about
2.5 kg, with a wingspan of about one and a half meters. Unlike most other stork-like
birds, it is covered with monotonous black plumage, which makes it look like
a huge crow and noticeable during the day. Therefore, during the day it hides
in secluded places – in tree hollows, thickets of reeds, and even in its nests:
this heron nests high in trees. As with many related species, the nest of coal
black night heron represents a huge “platform” made of several thick branches
in the forked crone, on which smaller branches are piled and rammed.
At night, coal black night heron is much more active than its ancestor: it wanders
through shallow waters and near water in search of its prey – mainly fish and
frogs, less often other small animals. In addition, it does not disdain carrion
– its beak is equipped with corneous “teeth” that can hold slippery prey and
inflict strong bites even to a large predator. The beak and legs of this bird
are black, only the claws are grayish. On the back of the bird’s head during
the mating season, several very thin feathers grow, forming a crest.
Coal black night herons are quite social birds, living both alone, in pairs
and in congestions numbering several adult breeding pairs and their fully fledged
offspring. Although conflicts may arise between separate birds over food, they
fight together with predators and competitors, striking them with powerful beaks
and screaming shrilly. The main enemies of coal black night heron are local
species of mustelids and owls.
The social way of life has another side: while some birds are eating, others
are watching the surroundings and may notice the appearance of enemies in time.
In the dark, this species of herons sees as good as owls and other nocturnal
birds. Eyes of coal black night heron are large, looking like owl’s eyes a little
bit. They have a red iris, and they glow orange in the dark.
Coal black night herons are monogamous and form breeding couples for many years;
in the south of their range, their mating period falls at the end of winter,
in the northern regions they return to nesting grounds early, as soon as the
ice disappears on the rivers. The females call the males with signals similar
to the crow’s cawing. Displaying males give females a ritual treat from their
beaks – fish or even a bunch of algae. At the same time, the male dances a kind
of “pas”: it jumps up and steps from one foot to the other. If the female likes
the male, she accepts his offering and the pair begins to build a nest together.
After about 2 weeks, the female lays eggs – they are pure white; there are usually
2-3 eggs in the clutch. Incubation begins with the first egg, so the chicks
in the brood differ in physical development. Sometimes the older chicks kill
the younger ones, but often all the chicks survive until they leave the nest.
In the south of the range, birds manage to make two clutches. Young birds of
this species grow fast, and by autumn they are ready to migrate to the south,
to wintering places. At this time, many coal black night herons die – this bird
is not a very fast flier, and during annual migrations it often becomes a prey
of birds of prey. The flight takes place mainly at night; in the morning the
birds gather in groups in natural shelters – on the branches of tall trees,
in rock niches. Wintering of these birds takes place at the islands of the Caribbean
Sea and at the coast of Central America. Some birds are found on the coast of
South America, where they get after crossing the Panama Strait, or via Great
Antigua.
Coal black night heron can live up to 17 years or more, but most of these birds
die much earlier.
This bird species was discovered by Bhut, the forum member.
Sea
bittern (Ixobrychus tetramarina)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: the coast of Fourseas, Crimea and Ustyurt islands, as well as large
lakes of Central and Middle Asia.
Holocene bitterns survived the human epoch relatively successfully. Their ranges
have significantly decreased due to habitat destruction, and a number of local
species have become extinct. During the ice age, due to changes in the boundaries
of natural zones, some more species became extinct, but with the retreat of
glaciers, the few surviving species began to populate the vacated habitats and
evolved actively. But in general, this group of birds has changed little in
the Neocene – despite the appearing of new species, the appearance of these
birds has remained quite recognizable, and the lifestyle is the same as that
of ancestral forms. However, even among these birds, specialized forms have
appeared, one of which is sea bittern.
This bird species is a descendant of a common little bittern (Ixobrychus minutus),
which has become a relatively large bird: the body length with the head is about
55 cm, the wing length is 30 cm, the weight is up to 500 g. Sea bittern inherited
from its ancestor a pronounced sexual dimorphism in the color of the plumage.
In the male, the plumage color on the upper part of the body is gray, each feather
is covered with thin cross ripples; on the head is a dark gray cap. The lower
part of the body is glaucous with longitudinal stripes of pale gray color. The
beak and legs are brown, and there is a narrow ring of darker plumage around
the eyes. The female has a camouflage brown plumage, each feather also with
transverse ripples. It has a darker “cap” on the head, turning into a stripe
stretching along the upper part of the neck to the shoulders. The plumage of
young birds is similar to the plumage of a female.
Sea bittern feeds mainly on fish, occasionally catching aquatic reptiles and
crustaceans, as well as small mammals appeared in the water. Usually this bird
hunts by standing in the thickets of water plants near the shore and remaining
motionless for a long time. After a storm, these birds often pick up stranded
aquatic animals. Outside of hunting, sea bittern hides in thickets of bushes,
or in the floodplains at the mouths of rivers. Sea bittern is very cautious
and interrupts fishing at the slightest alarm. A pair of adult birds can fish
together, but strangers are immediately expelled. The voice of this species
outside the breeding season is a characteristic croaking call: an abrupt single
cry serves as an alarm signal; a long series of cries serves to define rights
to the territory. During a direct conflict with relatives, the bird makes a
loud crack with its beak.
It is a strictly monogamous species. The breeding season is from March to May.
The clutch consists of 3-5 eggs, both parents take part in incubation and feeding
of chicks. This bird builds nests in swamps and reeds, usually in places inaccessible
to terrestrial vertebrates. Less often, the nest is arranged on trees near reservoirs
– on branches hanging directly over the water. The nest is a rather loose flat
platform with a depression in the middle, the base of which is composed, as
a rule, of rather thick sticks and branches, on top of which thinner branches
and stems of coastal plants are laid. Incubation lasts from 16 to 18 days. After
15 days, the offspring fully fledge and leave the nest. For some time, young
birds still return to the nest, where their parents feed them, but gradually
move on to independent life and learn to fly independently. A month later, the
parental instinct fades in adult birds; at this time, young birds are expelled
from the territory of the adult pair and begin to search for their own territory.
The next year, the sea bittern reaches sexual maturity. Life expectancy is up
to 13-15 years.
This bird species was discovered by Nick, the forum member.
Pygmy
nocturnal heron (Noctiardea minima)
Order: Pelecaniforms (Pelecaniformes)
Family: Herons (Ardeidae)
Habitat: wetland forests of Amazonia.
The human actions during the Neocene caused the extinction of many species.
However some survived and in the Neocene they give rise to new species. One
case is the pygmy nocturnal heron, a descendant of the zigzag heron (Zebrilus
undulatus). This bird lives in the Amazon forest. These animals are nocturnal,
resting during the day hidden between branches of trees near water.
Pygmy nocturnal herons are small birds, with 29 cm of height and a wingspan
of 46 cm. Adult plumage is dark grey with a pale underbelly. Juveniles have
a dark back with a pale brown head and underside. The legs are yellow and short
for a heron. The beak is bluish-grey. Their eyes are large and have a more developed
tapetum lucidum.
These birds feed on insects, such as grasshoppers and water bugs, spiders, small
fish, crabs, snails and frogs. Its hunts at night, feeding on moist soil, turning
the cover of leaves from the forest floor or mud around shallow pools in marshy
areas in search of preys, doing it silently and unobtrusively by standing and
by walking slowly.
Pygmy nocturnal herons lives solitary or in pairs. It makes its nest in trees
at the water's edge at low altitude from the ground or water. The nest is a
shallow platform of twigs, where the female lays 2 pure white eggs. The eggs
are incubated by both parents for 25 days. The young fledge with 28 days. These
birds reach sexual maturity with 4 years and their lifespan is of 19 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Haruspex
(Haruspex sarcophilus)
Order: Pelecaniforms (Pelecaniformes)
Family: Ibises (Threskiornithidae)
Habitat: savannas and woodlands of Meganesia.

Picture by Amplion, colorization by Biolog
Initial image by Amplion |
Neocene ibises cannot be called a very successful group. These
birds suffered losses during the era of human domination, as they suffered greatly
from habitat destruction (in particular, due to land reclamation), hunting and
the use of pesticides. The number of many species has decreased catastrophically
– in some cases, populations have been reduced to hundreds or even several dozen
individuals for several centuries. However, some ibises managed to survive due
to the fact that they either lived in places undeveloped by man, or began to
adapt to his neighborhood. In Neocene, they continued to develop, although in
some areas the niche of ibises was occupied by other birds (for example, by
sandpipers).
Haruspex, an inhabitant of the savannahs and woodlands of Meganesia – is one
of the most unusual ibises. Unlike related species, it broke the connection
with water and switched to a diet atypical for these birds – this species feeds
mainly on carrion. The peculiarities of nutrition are reflected in the name
of the bird: in ancient Rome, a haruspex was a fortune teller who predicted
the future from the entrails of sacrificial animals. In addition to the corpses
of dead animals, these birds often feed on live prey: they gather small vertebrates
and large insects in the grass, ravage the nests of local social
weaverbirds and catch fish in drying reservoirs.
Haruspex is a fairly large bird: its body length reaches a meter, and its weight
is up to 4 kg. The constitution of this bird is typical for ibises: it has long
legs and a neck. Wide wings allow it to soar in the sky for a long time, looking
for carrion. The plumage of the haruspex is black with a metallic blue shimmer.
After feeding, the birds disinfect it for a long time, opening their wings in
sunlight, getting rid of bacteria with the help of ultraviolet radiation (vultures
did the same in the human era). The head and neck of this bird are covered with
bare red skin – this is also an adaptation to carrion feeding. Sexual dimorphism
is not pronounced in haruspices.
The main tool for obtaining food from this bird is its beak, colored black.
Like at most ibises, it is long and curved, but, unlike them, it is much more
powerful and resembles rather the beak of hornbill of Bucorvus genus (ground
hornbills). Such a beak allows not only to butcher the corpses of animals, but
also to protect the bird's share of food from other scavengers
when birds of various species gather near the corpse.
The bird’s voice is a very loud hoarse “croak”.
Haruspices nest in colonies, making nests from branches in the crowns of trees.
Colonies usually include 10 to 20 breeding pairs of birds; there are also some
adult solitary birds in the colony that have lost a breeding partner or have
not yet formed a pair. These ibises are monogamous, and pairs sometimes persist
for many years and use the same nests; in this case, the nests are repaired
and renewed annually. The haruspex nest is cup-shaped, with a diameter of about
two meters. In the “foundation” of the nest there are thick branches, on top
of which the birds lay a thick layer of twigs; the nest is shallow. There are
2-3 eggs in the clutch at these birds. Both male and female incubate eggs and
feed the chicks. The chicks develop quite quickly and fly out of the nest for
the first time at the age of two months. Haruspices very actively repel the
attacks of predators on the nest: they beat the attacker with their wings and
peck it. The defense is accompanied by loud cries that attract birds that have
temporarily left the colony. To flying predators, these birds give a “dogfight”,
striking with their beaks and feet on the back and wings and grabbing feathers
with their beaks. The “collective defense” of several dozen birds is so effective
that there are few ones willing to attack the colony of these ibises, and they
successfully raise about 70% of the chicks to independence.
During non-breeding time, haruspices do not seek to maintain contacts with each
other – colonies break up, and nesting sites become empty. At this time, their
nests are used by other birds. Outside of the nesting season, each pair keeps
apart from the neighbors and the partners themselves are limited to formal signs
of attention to each other. However, haruspices often gather in flocks at the
source of food; at this time, conflicts are not uncommon, accompanied by displays
of the beak and stretched wings.
These birds have few enemies, so many of them can live to a significant age
of 35-40 years.
This bird species was discovered by Simon, the forum member.
Crowned
ibis (Magniplegadis coronatus)
Order: Pelecaniforms (Pelecaniformes)
Family: Ibises (Threskiornithidae)
Habitat: South-Eastern Europe, Fourseas; coastal reeds.
In human epoch glossy ibis (Plegadis falcinellus) had been one of the most widespread
ibises in the world, living both in Old and New World. This species of birds
had suffered in epoch of biological crisis – its range had been broken to some
separate areas. But some populations of glossy ibis kept the sufficient number
to survive in degraded habitats, and after the extinction of humankind they
formed new separate species.
One descendant of the glossy ibis, the crowned ibis, is a large bird 1.7 m long
with an upright standing height of 1.2 m and a wingspan of 2.3 m. This bird
has brownish curved bill, brown body, grayish green wings, dark facial skin
bordered from above and below with blue-gray, and reddish-brown legs. During
the breeding season the adults acquire reddish brown body feathers, their wings
became shiny bottle-green and their facial skin turns cobalt blue. Their main
characteristic is their crown of stiff brownish-red feathers that became vivid
red during the mating season.
These birds eat fishes, insects, amphibians, small birds, mammals and reptiles.
Sometimes they beat the prey against rock or ground to kill it.
It is a monogamous species. Crowned ibis males choose a site to perform a display.
They begin vocalization (these birds are silent during the most of the year)
until a female arrive to its chosen site. With the female coming closer male
begins head-shaking, exposing its “crown” and stick-shaking (grasping any twig
and shaking it briefly). If the female choses the male, she follows its movements,
copying them. After that, they continue with allopreening. During the next breeding
season the pair reinforces their bond by performing the same displays together.
They arrange their nests at the tops of trees near water, in mixed colonies
of aquatic birds. Female lays 3 eggs, which are incubated by both male and female
for between 31 and 33 days. Juveniles fledge in about 40 days, but keep dull
grey color until they reach sexual maturity, being two years old. They stay
with their parents during the migration, passing to independent life when they
return to their summer locations. Their lifespan is of 35 years.
Living in marshes of the northern part of Fourseas, Crimea Island and Three-Rivers-Land,
they migrate to Hindustan in autumn. During their migration, crowned ibises
form familiar flocks of the adult pair and their young. The flock flies in characteristic
V-like formation.
This bird was discovered by João Vitor Coutinho, Brazil.
Red-necked
ibis (Neoplegadis rubrocollis)
Order: Pelecaniforms (Pelecaniformes)
Family: Ibises (Threskiornithidae)
Habitat: wetlands of Southern and South-Eastern Asia, Far East.

Picture by Pavel Volkov
In human epoch glossy ibis (Plegadis falcinellus) was among
the most widespread birds of the world. In historical time this bird crossed
Atlantic ocean and colonized New World, having increased its natural range.
Due to the great range glossy ibis survived in human epoch, but its range had
been fragmented, and it favored to further evolution of new forms from the common
ancestor. A descendant of the glossy ibis, the red-necked ibis, represents the
genus Neoplegadis (“New sickle”). This bird is a mid-sized ibis, with 55 cm
of length and 85 cm of wingspan. It has brownish bill curved down, red legs,
grey body and greyish green wings (becoming shiny bottle-green when nesting).
Its main characteristic is brownish-red neck turning shiny red during the breeding
season.
These birds prey on insects such as aquatic beetles, dragonflies, damselflies,
grasshoppers, crickets, flies and caddisflies. Leeches, molluscs, crustaceans
and small vertebrates are also part of their diet.
Red-necked ibis is a monogamous species; male chooses a site and begins uttering
loud calls to attract the female. When the female had chosen the male, the pair
shows it by performing allopreening. In the next breeding season the pair will
do an allopreening and vocalize together to reinforce their bond.
Their nest is usually a platform of twigs and vegetation in large trees. Red-necked
ibis usually makes its nests in mixed colonies. The female lays 3-4 eggs, which
are incubated by both male and female for between 20 and 22 days. The young
fledge being about 25 days old, but stay with the parents for three months.
Juveniles reach sexual maturity at the age of one year; at this age their necks
acquire the brownish red color. Their lifespan is about 18 years.
These birds live in marshes at the margins of lakes and rivers, but can also
be found at lagoons, flood-plains, wet meadows and swamps from Hindustan to
the Far East. They do short migrations during the dry season.
Another member of the genus Neoplegadis is red-headed
ibis (Neoplegadis rubrocephalus). This species lives in marshes from
the Jakarta Coast to northern Meganesia. Its main characteristic is brownish
red head that turns shiny red during the breeding season. This species has 52
cm of length and wingspan of 79 cm. These birds are non-migrating.
This bird was discovered by João Vitor Coutinho, Brazil.
Grey
ibis (Griseoplegadis americanus)
Order: Pelecaniforms (Pelecaniformes)
Family: Ibises (Threskiornithidae)
Habitat: marshes, lakes and rivers of the northern part of South America, Great
Antigua and southern North America.

Picture by Pavel Volkov
In human era the glossy ibis was the most widespread ibis
species, with individuals on almost all continents except Antarctica. The individuals
in Asia gave origin to two new genera: the Neoplegadis
and the Magniplegadis. In the Americas
the genus Griseoplegadis evolved, with the grey ibis being this only representative.
The grey ibis has 49 cm of length and 75 cm of wingspan. It has brownish bill
curved down, red legs and shades of green in the wings (become shiny bottle-green
when nesting). Its main characteristic is the grey plumage, which covers the
whole body.
These birds prey on insects, leeches, snails, earthworms and small vertebrates
are also part of the diet. This species lives mainly in marshes at the margins
of lakes and rivers, tending to avoid coastal areas.
The grey ibis is a monogamous species; the male chooses a site and starts to
make a loud call to attract the female. When the female chose the male, the
pair will start to do an allopreening. The pair will never distance themselves,
they will search for food together and during flights they will stay together,
even in the great flocks that this species form.
Their nest is usually a platform of twigs and vegetation in large trees near
water. The grey ibis usually make their nests in mixed colonies. The female
lay 3-5 eggs, which are incubated by both male and female birds for between
17 and 22 days. The young fledge in about 24 days, staying with the parents
for 2 month, the young reach sexual maturity with 10 months. Their lifespan
is of 17 years.
This bird species was discovered by João Vitor Coutinho, Brazil
Pale
ibis (Paraplegadis pallidus)
Order: Pelecaniforms (Pelecaniformes)
Family: Ibises (Threskiornithidae)
Habitat: islands of Melanesia from the Solomon Islands to New Caledonia, separate
rookeries in the northeast of Meganesia.

Picture by Pavel Volkov
Ibises suffered quite seriously in the human epoch. Hunting,
destruction and pollution of habitats have undermined their numbers and caused
the extinction of a number of species. After the stabilization of natural conditions
in the early Neocene, the surviving species continued to evolve, forming new
species. One of the Neocene representatives of the family is pale ibis, a descendant
of the glossy ibis (Plegadis falcinellus).
Body length is 60-70 cm, wingspan is up to 1 m. The bird looks large because
of its long legs, neck and beak, although it is only slightly larger than a
crow in body size. The species has a wide range; throughout the range the color
of the plumage varies: it can be pale brown or greenish-gray in different populations.
Pale ibis feeds on any small animals – from aquatic invertebrates to small birds
and reptiles. In Meganesia, these birds catch frogs and small mammals. Not as
agile a hunter as the heron, pale ibis survives due to less specialization.
Groups of these birds, numbering 15-20 individuals, move through shallow waters,
lake shores, banks of small rivers, and also check the glades in the jungle.
In such groups, birds have no hierarchy and any of them can take prey from a
neighbor. However, acting together, they easily drive away herons and detect
predators. Their enemies are birds of prey and reptiles that attack individual
birds, but a group of pale ibises can repel small predators.
The mating season lasts from January to March. Pale ibises nest in colonies,
arranging rookeries on tree branches near the water. In colonies, birds quarrel
loudly, sharing convenient nesting sites and competing for females. The nest
is a loose construction of twigs on a foundation of large branches; males often
steal building material from each other, and sometimes they can simply “take
apart” the nest of an unlucky competitor to the foundation. Pairs in this species
are formed for one season, with females choosing a male and a nest. The clutch,
which is incubated by both parents, consists of 3-4 eggs. Incubation lasts 28
days. 40 days after hatching, young birds take wing. At 8 months, young birds
become completely independent and form their own groups. Sexual maturity occurs
at 2 years; life expectancy is up to 20 years.
This bird species was discovered by Nick, the forum member.
Pink
ibis (Eudocimus roseus)
Order: Pelecaniforms (Pelecaniformes)
Family: Ibises (Threskiornithidae)
Habitat: tropical areas of North America, islands of Central America.
The Ice Age, which occurred in the beginning of Neocene, forced some species
to migrate to warmer locations. One of these species was the American white
ibis (Eudocimus albus). This ibis expanded its area to more southern locations,
and so its new territory overlapped completely the territory of the scarlet
ibis (Eudocimus ruber). It resulted in hybridization, and after several generations
a new species appeared – pink ibis.
Pink ibis lives in mangroves and other coastal regions from Southern North America
to Great Antigua and the tropical coast of South America. Pink ibis have 50
cm in length and 87 cm of wingspan; bird looks larger because of long neck and
legs. It has pale pink plumage and bright orange bill and legs. Its facial skin
is pink and turns vivid red during the breeding season. Adults have dark pink
wingtips that are usually only visible in flight.
Pink ibis prefers to feed on crayfish and other crustaceans, using its thin
bill to probe for food in soft mud or under plants. The pink color is related
to its diet; this species performs the metabolism and selective incorporation
of carotenoids that is accumulated in its feathers – ability inherited from
its scarlet ibis ancestor.
Pink ibis is monogamous and breeds in numerous colonies, usually side by side
with other waterbird species. To attract a female, the male performs a variety
of mating rituals such as preening, shaking, bill popping, head rubbing, and
high flights, after united the pair is often seen doing an allopreening. The
female chooses the site in the branches of mangrove tree well above the water
and builds the nest, and male assists by bringing nest material; the nest is
a loose platform of sticks. If they can, they nest on islands far from land-dwelling
predators.
The female lays 5 eggs, which are incubated by both male and female birds for
between 20 and 23 days. The young fledge in about 26 days, staying with the
parents for 4 month. Juvenile birds have grey plumage turning pink when they
reach sexual maturity at the age of 2 years. Their lifespan is about 22 years.
This bird species was discovered by João Vitor Coutinho, Brazil
Marabou
vulture (Dolichocathartes velodromus)
Order: Cathartiforms (Cathartiformes)
Family: Cathartids (Cathartidae)
Habitat: south-west of Northern America, semi-deserts and plains.

Picture by Tim Morris
Many species of carnivorous birds, especially large species
of orders Falconiformes (daily birds of prey) and Strigiformes (owls) transferred
changes of inhabitancy and the anxiety, connected with human activity badly
enough, and their number in Holocene had been sharply reduced. But some “not
true” predatory birds at this time, on the contrary, had got significant advantage
from the neighborhood of people. One of such species, the turkey vulture (Cathartes
aura) concerned to typically synanthropic species. In Holocene this species
was settled enough and numerous, that its populations could survive in conditions
of anthropogenous pressure and at the subsequent change of inhabitancy in ice
age.
The descendant of the turkey vulture widely distributed in Northern America,
became a creature of rather frightening appearance. This Neocenic representative
of cathartids is flightless, but instead of wings it has got strong legs, becoming
tireless walker and sprinter. The marabou vulture by constitution is similar
to herons: at bird there are long legs and neck, and the general growth reaches
170 cm. This species is even more similar to “typical” stork birds by long skull
on which tip corneous cover of beak forms similarity of hook. Wings of marabou
vulture are still rather advanced, but the bird cannot fly any more. However
wings are not so useless to this bird: they serve mainly for courtship displays
(inherited from the turkey vulture), and with their help the bird can make sharp
turns at fast run.
The marabou vulture similarly to its Holocene “doubles” from Old World, eats
carrion. Outside of nesting season this bird frequently follows herds of herbivores
mammals - donkeyhorses,
desert
deermaras, or peccasons,
hoping to prey the weakened animal, or catching small creatures crushed by these
herbivores. Desert birds can follow hatches of huge ostrich
turkey, or “attach” to huge bear
porcupines, eating animals casually found out by it. At absence of
large catch marabou vultures catch small animal (rodents, reptiles, insects),
and even eat fruits from low trees. During mass fruit ripening this bird becomes
practically the vegetarian. Thanking to “firm” stomach marabou vulture can eat
even prickly fruits of prickly pear cactus. This bird finds food with the help
of sense of smell and sight. Sense of smell at marabou vulture is keen: nostrils
are through and very wide. Above nostrils the high corneous crest similar to
crest of Oviraptor dinosaur is formed. This formation serves for cooling: under
corneous cover a circuit of blood vessels passes, radiating surplus of heat.
At the same time they strongly advance olfactory epithelium of bird.
On head of males and females there is naked skin colored differently (such feature
is exception among cathartids). At females skin on head and neck is meat – red.
Male is smaller, than female, and its head is dimmer – it is brown. The skin
becomes especially bright in courtship season: at males it becomes reddish-violet,
at females head is scarlet with orange shade and yellow longitudinal strip from
level of eyes up to horn cover of beak. The feathering of birds of both sexes
is black, only on wings black color is replaced by dark grey.
This species lives (and follows herds of herbivores) in small groups. In courtship
season (at the end of drought season) small groups of birds gather to congestions
numbering up to fifty birds, and begin noisy courtship displays. At this time
females show bright skin of head and neck, loudly crying, and males perform
near them the original dances including wing flapping, jumps, “waltz” near the
chosen female and bows of partners to each other. Breeding couples at this species
are formed only to one nesting season.
For the period of nesting group breaks up, and each pair of birds brings up
posterity independently. The nest of marabou vulture is a simple small hole
in the ground, without any litter. In clutch there are 2 eggs hatched mainly
by the male. Nestlings hatch covered with down and with opened eyes, but remain
in nest for a long time. Approximately at four-monthly age young birds study
to run, and leave nest for ever. At this time they strongly differ externally
from adults: at them there are light grey feathering and black skin on head.
Within winter young birds keep with parents, training to search for food. In
spring when preparations for courtship displays begin, the not sexual matured
young growth forms one-age congestions – “gangs”: it is easier to birds to live
this way. Approximately at the fourth year of life young birds start to take
part in courtships.
Life expectancy of marabou vulture reaches 50 years.
Twilight
vulture (Cathartops crepuscularis)
Order: Cathartiforms (Cathartiformes)
Family: Cathartids (Cathartidae)
Habitat: South America, pampas and woodlands in the north and in the center
of the continent.
The New World, North and South America, represents a parallel world for the
world of Eurasia and Africa in terms of fauna and flora. There are various animals
and birds that are similar to their overseas counterparts, but often are not
their relatives. Among them there are cathartids, scavenging birds of New World,
which are close relatives of stork birds: storks and ibises. Being scavengers
and non-specialized predators capable of flying considerable distances, and
being unassuming to their environment, these birds were able to survive both
the neighborhood of people, and a new ice age, and even a new split of New World
into two separate continents. The latter even benefited the cathartids, as many
of their ancestral species evolved to new species on both continents.
One of these species was the turkey vulture. In North America, its descendant
is the marabou vulture (Dolichocathartes
velodromus), and in South America, where a much larger and more formidable
scavenger, the great acatu (Sciopterornis
acatou), rules in the skies, a different species has appeared – a twilight
vulture. Unlike its North American relative, it is a well-flying bird, reaching
about 70 cm in length. Plumage of this species is coal-black. Head remained
featherless, like at the ancestor, but the shape of this head has changed somewhat:
compared to the ancestral species, it is shorter and wider. These changes occurred
because the twilight vulture’s sense of smell is even more developed than that
of its ancestor, and the nostrils have become even larger relative to the overall
size of the head; those brain lobes that are responsible for the sense of smell
are also very large, which is actually rare for birds. The hearing of this bird
is also well developed, and the eyes are proportionally larger than at the ancestor;
twilight vulture has excellent vision.
However, the vision of this species has significant features: unlike many birds,
there are quite a few cone cells in the eyes of the twilight vulture, but there
are a lot of rod cells, which is why this bird weakly distinguishes many colors,
but it sees very well at dusk, and even in almost complete darkness, in the
light of stars. This helps a lot when twilight vulture flies out in search of
its prey – carrion and small animals, as its ancestor did; this species is looking
for food not during the day, when akatu rules in the sky, but in the morning
or evening twilight, or even at night, avoiding thus competitors in the form
of most other scavengers. Owls are not competitors of this species: carrion
is out of their interests.
Such a radical change in lifestyle has changed not only the head and brain of
twilight vulture, but also the rest of the body. While the ancestral species
preferred to hover passively enough in the sky, its descendant had to switch
to a more active flight; twilight vulture has longer and narrower wings, resembling
more the wings of a seagull than birds of prey, and in general this bird flies
more actively and orients itself in the air, catching odors.
Despite the changes in anatomy, the behavior of the twilight vulture has changed
less: it is still essentially a scavenger that feeds on already dead animals.
Of course, some of its prey dies during the day, during daylight hours, and
most of it goes to other species. But twilight vulture has a fairly short and
powerful beak, and a muscular head and neck: this bird may well scrape or tear
off even a small piece of meat from the bones, as well as crack small bones
with blows against stones and extract bone marrow from them – this bird also
has a long tongue that allows it to do it.
Such adaptations give the twilight vulture the opportunity to deal with live
prey, but since its paws are not adapted to grabbing, and the flight, unlike
the owl’s one, is not silent, most of the living creatures that fall prey to
the twilight vulture usually already die, or are old, sick, wounded, as well
as young or inexperienced. Occasionally, this bird attacks sleeping animals,
trying to surprise and kill a completely healthy animal. These birds prefer
to hunt in open spaces.
Twilight vulture lives in the north and center of the South American continent,
where the climate is quite warm or even tropical, so it does not have a pronounced
mating season, and at any time of the year males of this species are ready to
court females. Displaying himself to the female, the male spreads his wings
and clumsily dances in front of her, lifting his legs high. Usually nesting
occurs once every 18-20 months. The absence of seasonality in reproduction somewhat
compensates for the slow rate of reproduction of this species. There is no sexual
dimorphism in twilight vulture; both sexes are relatively equal in strength
and height, therefore, manifestations of aggression during communication between
the sexes are minimized.
Like most species of birds of prey, twilight vulture almost does not build a
nest: it occupies someone else’s one, or lays eggs (usually 1-2, less often
3) on a cliff, or directly on the ground, but in a place inaccessible to terrestrial
predators. The eggs are light brown in color; they are incubated by both parents
in turn, for about six weeks. The chicks are covered with light down; they grow
slowly, and live with their parents for about a year before they will be independent.
If the twilight vulture manages to survive the first two or three years of life,
then at the age of about 4 years the bird reaches sexual maturity and in the
future it will be able to live up to 50-60 years.
This bird species was discovered by Bhut, the forum member.
Long-necked
apitan, or long-necked vulture (Apitan longicollus)
Order: Cathartid birds (Cathartiformes)
Family: Cathartids (Cathartidae)
Habitat: tropical areas of South America – forests and grasslands.
During the Holocene, human interference in nature caused the extinction of many
species. Only those that were resilient enough were able to survive and left
descendants. One of these survivors was the black vulture (Coragyps atratus).
This species was widespread and was capable of adapting to anthropogenic interference,
by feeding on landfills and taking opportunity in the areas affected by man.
In the Neocene they give rise to the genus Apitan. This word is a regional name
for the black vulture in Brazil. The type species of the genus is the long-necked
apitan, which inhabits the grasslands and forests of tropical South America.
The long-necked apitan has 63 cm of length and 157 cm of wingspan. The main
characteristic of this species is their long bare neck, which gives them the
appearance of a stork while flying. Its plumage is mainly glossy black, with
only the wings being white. The skin of their head and neck is dark grey and
wrinkled, while both the beak, legs and feet are pale grey.
This bird eats mainly carrion, but also takes eggs, fruit (both ripe and rotting),
fish and can kill or injure newborn or incapacitated mammals. Its long bare
neck allows it to pick food inside deep crevices in the carcasses. Like other
vultures, it plays an important role in the ecosystem by disposing of carrion
which would otherwise be a breeding ground for disease. Like their ancestors,
the long-necked apitan locates food by sight. This species is aggressive when
feeding, and may chase the smaller scavengers from carcasses, but will flee
with the arrival of a viceroy vulture and of an Acatou. A characteristic of
this Genus is that they will remove and eat ticks from resting large mammals.
The apitans will also visit nesting colonies of herons and seabirds to kill
young. Young turtles leaving their nests will also be eaten.
The long-necked apitan is monogamous, with the pairs staying together for the
rest of their lives. Pairs are formed following a courtship ritual which is
performed on the ground: several males circle a female with their wings partially
open as they strut and bob their heads. Once the pair is formed, they will perform
courtship flights, diving or chasing each other over their chosen nest site.
The female lays one to three eggs on the ground in a wooded area, a hollow log,
or some other cavity. Both parents incubate the eggs, which hatch after 28 to
41 days. Upon hatching, the young are covered with a buffy down. Both parents
feed the nestlings, regurgitating food at the nest site. The young remain in
the nest for two months, and after 75 days they are able to fly skillfully.
They reach sexual maturity with 3 years and have lifespan of 26 years.
Another species of the Genus Apitan is the Northern
Apitan (Apitan mexicanus), which lives from the Mexican Plateau to
the forests of the Panama Peninsula, with some also being found in Great Antigua
and Florida. While the plumage of this bird is similar to that of the long-necked
apitan, they have a shorter neck. They are also smaller, with 54 cm of length
and 142 cm of wingspan.
This bird species was discovered by João Vitor Coutinho, Brazil.
Great
viceroy vulture (Pseudosarcoramphus vicerex)
Order: Cathartid birds (Cathartiformes)
Family: Cathartids (Cathartidae)
Habitat: tropical and temperate latitudes of South America.
During the Holocene, human interference in nature caused the extinction of many
species. Only those that were resilient enough were able to survive and left
descendants. One of these survivors was the lesser-yellow headed vulture (Cathartes
burrovianus). This species was widespread and was capable of adapting to anthropogenic
interference, by feeding on landfills. In the Neocene they give rise to the
genus Pseudosarcoramphus, the viceroy vultures. The type species of this genus
is the great viceroy vulture, a large scavenger that inhabits all the areas
of tropical and temperate South America, from the Amazon rainforest to Patagonia.
The viceroy vulture has 77 cm of length and 197 cm of wingspan. Their plumage
is predominantly black, which contrasts with their wing coverts, flight feathers
and tail that are white. Their neck is featherless, with the exposed skin being
yellow. These birds also have a grayish-blue comb. Their bill has a hooked tip
and a sharp cutting edge. It’s dark gray, like their legs.
The viceroy vulture eats anything from large mammal carcasses to beached fish
and dead lizards. While principally a carrion eater, they will occasionally
kill and eat injured animals, newborn animals and small lizards. This bird inherited
their ancestors’ good olfactory sense, being capable of finding a carcass several
kilometers of distance. The viceroy vulture primarily eats carrion found in
the forest, though it is known to venture onto nearby savannas in search of
food. This is an adaptation to avoid competition with the acatu.
Once it has found a carcass, this bird displaces the other vultures because
of its large size and strong bill. However, when it is at the same kill as the
larger acatu, the viceroy vulture always defers to it. Using its bill to tear,
it makes the initial cut in a fresh carcass. This allows the smaller, weaker-beaked
vultures, which can not open the hide of a carcass, access to the carcass after
the viceroy vulture has fed.
Viceroy vultures are monogamous, with the courtship consisting of several elaborate
air maneuvers. This species do not build nests, but rather lay eggs on the ground,
cliff ledges, the floors of caves, or in the hollow of a tree. The female lays
two eggs, both parents incubating it for 53 days before it hatches. The chicks
are altricial – they are blind, naked and relatively immobile upon hatching.
The parents feed their young by regurgitating pre-digested food into their beak,
where the chicks then drink it. The young fledge after two to three months.
Sexual maturity is reached with four years old and they have a lifespan of 38
years.
The
Lesser viceroy vulture (Pseudosarcoramphus minor) is another
representative of the Genus Pseudosarcoramphus. This bird lives from the grasslands
and woods of the Mexican Plateau to the tropical forests near the coast of the
Panama Peninsula. Their plumage is similar to that of their Southern relative,
but they are smaller. They have 63 cm of length and 168 cm of wingspan. This
species also lacks the comb present in the Great viceroy vulture.
This bird species was discovered by João Vitor Coutinho, Brazil.
Crested
wasp burglar (Pappopernis fur-vespinus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Europe from the Alpes up to Ural: hibernates in savannas of Northern
Africa to the south of Mediterranean swamps and in Arabia, occasionally reaches
northern coast of Zinj Land.

Picture by Sergey Ivanov
In Holocene the majority of birds of prey became extremely
rare as a result of pursuit from the side of people and destruction of native
habitats. Because of ecological crisis mainly large species – eagles and vultures
– had suffered. Populations of “noble” large falcons appeared undermined by
human activity, and these birds also had disappeared. But the few of birds of
prey nevertheless managed to survive in ecological crisis – they were mainly
low-specialized species eating rodents or insects. During ecological crisis,
right after human extinction, in Earth biosphere the ecosystems formed by smaller
species of animals dominated short time. This period had served as though as
the “filter” which had kept only predators eating small catch. But separate
specialized species of order also managed to survive because of specialization.
One of species of feathered predators, honey buzzard, or pern (Pernis apivorus)
ate insects – mainly social hymenopters (wasps and bees). Such numerous and
stable food source had permitted this bird to survive and evolve further to
the way of deeper specialization.
The descendant of pern, crested wasp burglar, lives in Europe. This bird inhabits
woods of temperate and subtropical zones, and spends winter in African savanna.
Because of specific diet crested wasp burglar does not compete to wood insectivorous
birds.
It is rather small bird: by size it is a little bit larger than pigeon. At crested
wasp burglar there are long tail and rounded wide wings. This bird does not
hunt for vertebrates, therefore the special maneuverability in flight is simply
not necessary for it.
The food of crested wasp burglar is made of social insects, mainly hymenopters.
This species is less specialized than waxeater
and beekeeper parrot: crested
wasp burglar eats not only bees, but also much more numerous wasps.
The main hunting weapons of predatory birds are hooked beak and sharp claws.
The beak of wasp burglar had kept the shape typical for these feathered predators,
but became more lengthened, with small hook at the tip – it is more convenient
so to pull out larvae of insects from nests. Claws of bird had a little changed:
they became blunt, thicker and shorter. Crested wasp burglar does not catch
prey by paws, but spends a lot of time on the ground. It can walk and run fast,
and is able even to dig out underground nests of hymenopters, working by legs
like gallinaceous birds. On sandy steeps of riverbanks crested wasp burglar
digs out nests of digger-wasps and eats their larvae both with stock of insects.
This bird has kept soft colouring similar to colouring of ancestor. Back, top
part of head, wings and tail of crested wasp burglar are colored brown without
spots. Bottom part of head of bird is white; at the neck on tips of some feathers
dark spots appear. On stomach some feathers have black fringing on edges, and
because of it on feathering of bird the faltering scale-looking pattern appears.
Crested wasp burglar is able to ravage nests of bees and wasps, not being afraid
of their stings. The presence of the special protective adaptation, very important
at attacking of nests of stinging insects is the main difference of this bird
from an ancestor. Above the beak of bird the crest of rigid feathers is advanced;
it is opened and protects eyes when bird ravages nest of wasps or bees. The
similar adaptation is present at the beekeeper parrot, and at the waxeater there
are “eyebrows” of rigid feathers protecting eyes. Thus, wasp burglar represents
a nice example of convergence connected with feeding at the same food source.
In connection with feeding by specific kind of food crested wasp burglar is
a territorial bird – pair of birds supervises territory about 9 square kilometers
in size. Birds declare rights to territory by loud repeating calls. Pair keeps
very amicably and in common expels competitors from their posession.
Because this species lives in areas of seasonal climate, it is the bird of passage.
At wintering places crested wasp burglar frequently accompanies insectivorous
animals and feeds on termitaries ravaged by them. Sometimes crested wasp burglar
obtains food in common with other bird eating hymenopters, the savanna
waxeater belonging to honeyguides family. These birds compete with each
other, and sometimes fight near the ravaging nest. Usually well armed crested
wasp burglar succeeds to win savanna waxeater, and common feeding actually turns
to robbery.
This bird nests two times per one year – in Neocene summer (understanding as
this period warm season suitable for nesting) became actually longer, and insects
meet longer in enough amount. The ancestor of this bird in Holocene had time
to make only one hatch for a season.
In clutch of crested wasp burglar there are about 3 eggs, they are hatched only
by the female within 15 days. The male feeds the female on nest and takes part
in feeding of posterity. The posterity develops in nest within 5 weeks. The
first feathering of young birds is dimmer, than at adults – the bottom part
of body is grey, and head is entirely brown.

Picture by Alexander Smyslov
The related species, Far East wasp burglar (Pappopernis orientalis), lives in woods of Far East, Japan and Indonesia. It is settled bird; only birds from northern part of area migrate to the south. It differs from crested wasp burglar in larger sizes (it is crow-sized bird), and brighter colouring – primary feathers are white with cross black strips, protective feathers of crest are also white. Individuals from southern populations also have white cross strips on feathers of tail (they are separated to subspecies Pappopernis orientalis callocaudis) and differ in smaller size.
This species of birds is discovered by Simon, the forum member.
Sea harrier (Algocircus longipes)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: algae fields near New Azora.
Volcanic activity in area of Mid-Atlantic Ridge had caused significant expansion
of Atlantic Ocean in Neocene epoch. Besides tops of this ridge had grown to
the surface of water, and the ridge was designated at the surface of water
as a circuit of islands and New Azora island is one of them. Currents form
near the island slow, but steady whirlpool, in which floating brown algae plentifully
expand. Algae form on water surface a kind of dense carpet, on which small
tetrapods can move. Some birds nesting at New Azora are able to walk on these
algae, searching for food. Among them large ghost wanderers, the birds similar
to ibises with long toes, are characteristic. Besides them, on algal fields
ducks of some varities, and also fog
swifts live. Fog swift does not perch
on algae: in this case it simply can not fly up from surface of this sea swamp.
It is involved with insects feeding on tissues of algae.
Such abundance of the live beings living at algal fields has caused occurrence
of the hunter preying these birds. An only species of birds of prey, which
could adapt to inhabiting in so specific conditions, was marsh harrier (Circus
aeruginosus). This migrant of the European origin had settled at New Azora
during an ice age – it migrated to this island from the Western Europe via
Canary Islands. The small population of this species lived at New Azora, followed
its prey, various birds, moving to algal fields. Changes in habit of life had
caused respective changings in its shape and had resulted in evolving of new
species – sea harrier.
By its size sea harrier does not differ from the relatives lived in Holocene
epoch. But it seems larger because of long legs and peaked wings. The tail
of this bird is also lengthened, and is similar to tail of swallow. Sea harrier
is fine flyer.
This bird is well adapted to life at algal fields. To move easier on algae
carpet, sea harrier developed long toes with straight claws; tarsometatarsus
of this bird is also lengthened, and the plumage almost does not touch water.
But for additional protection of feathers from wetting at this bird the coccygeal
gland is strongly advanced. Sea harrier spends a lot of time, clearing plumage
and greasing it with oily water-repellent secretions.
Plumage of sea harrier is bicoloured. Bird has gray-blue bottom of body – throat,
breast, stomach, bottom part of wings and primarily feathers. Therefore during
the flight this bird is poorly appreciable from below. The top part of head
and neck, back, covert feathers of wings and quill feathers of tail, on the
contrary, are brownish-green. Sea harrier occasionally hunts flying birds,
hiding on surface of carpet of floating algae. At this time colouring makes
this predator imperceptible at the sight from above. When the flying bird appears
beside, sea harrier promptly flies up and strikes a blow to it by strong claws
growing on back toes. Sea harrier frequently practises air hunting for fog
swifts above forest. In dawn time flocks of these birds fly to algal fields,
and at sunset come back to nesting place. Sea harrier hides in trees crones
near to favourite routes of daily flights of fog swifts. Hunting these fast
birds, it flies at full speed into their flock, having put forward claws, and
succeeds to kill one or two birds during the attack. Also this bird occasionally
can feed on carrion. At New Azora these birds frequently fly to beaches where
large sea birds, plesioloons and gannetwhales have a rest and raise posterity.
Sea harriers eat dead birds and nestlings there, not paying attention to healthy
individuals many times surpassing them in weight and size.
Sea harriers nest at New Azora. Nesting pair at this species is formed to one
nesting season. In lowland marshy areas of island these birds arrange nest
in reeds, but the part of population chooses for nesting thickets of high grass
at the mountain meadows. Sea harriers nest on the ground; the nest represents
a wide platform of grass and branches. Female is larger, rather than male,
and dominates in family relations. She hatches a clutch of 3 – 4 eggs with
dark brown spotty shell. Nestlings of sea harrier hatch with open eyes and
covered with rich grey down. Usually they rather peacefully behave relatively
to each other, and only at lack of food stronger nestlings kill weaker ones.
Parents feed nestlings on small birds, and during the raising of nestlings
prefer to hunt small prey at sea coast or at the edge of forests. They actively
protect posterity from ground animals. During an attack on enemy birds try
to strike in its eyes or nose. Development of nestlings lasts till about three
months, and usually 2 – 3 nestlings survive up to leaving of nest. They differ
from adult birds in dark grey colouring with lighter stomach. After the second
mew young birds get colouring characteristic for adult birds.
The idea about existence of present species of birds was proposed by Simon, the forum member.
Neopueo
(Strigocircus vespertinus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Hawaii, woods.

Picture by Alexander Smyslov
In Hawaiian “pueo” means “owl”. At Hawaiian archipelago inn
human era short-eared owl, obviously, bird of North American origin, lived.
Its descendant in Neocene epoch also inhabits these islands, but the new Hawaiian
predator partly competes with it – the descendant of harrier migrated to Hawaii,
one of recent settlers of these islands. This feathery predator is neopueo,
the nocturnal predatory bird which is very similar to owls. Probably, otherwise
owls simply wouldn't allow the appearing of such competitor at the islands,
but neopueo and owls are clearly differing in their activity time and therefore
compete for prey not so rigidly. Unlike owls, activity peak of neopueo takes
place twice per day: in late evening and early in the morning, in twilight.
It is so because this bird hunts mainly with the help of sight, relying on hearing
to the lesser extent. Therefore it spends the darkest night hours in relative
rest, and owls hunt right at this time.
Neopueo is rather small bird of the pigeon size. It hunts small vertebrates
and large insects. The nocturnal habit of life made a bird similar in shape
to an owl, of neopueo. The head of neopueo is roundish, and the feathers growing
around eyes form a characteristic face disk. The head of bird has dark brown
color, and the face disk is white, looking like a mask.
Long tail and rather short and wide wings allow the maneuvering among branches:
this predator lives only in forests. Primal feathers of neopueo are similar
to owl’s ones: on them there is a set of bristles along the vane edge. They
extinguish air turbulences at flaps of wings, and because of it flight of this
bird becomes almost silent as at true owls.
The body top of neopueo is colored brown with dark cross stripes; stomach is
grey. On the head stripes are more dense and narrow; therefore head seems darker
than body is. Primal feathers are light brown, and long tail feathers have black
tips.
One more “heritage” of harrier in shape of neopueo is the length of its legs.
Because of it the bird seems slightly larger, than it is actually. Legs of neopueo
are covered with smooth small scales of yellowish color, toes are armed with
hooked claws. Neopueo has an interesting feature which is characteristic for
some African birds of prey of Holocene: it has an exclusively mobile joint between
tarso-metatarsus and shin; it can bend not only forward, but also sideways,
and even back a little. Due to such adaptation neopueo can hunt small animals
hiding in hollows, just pushing paw in tree trunk hollow and seizing the animal
hidden there.
Neopueo is a solitary one, and only during the mating season a couple of birds
builds nest on high trees and grows up posterity in common. Nestbuilding skill
of neopueo is developed better, than at other birds of prey. Birds of this species
build in branches a big nest of brushwood looking like deep bowl though more
often they occupy and re-build the ready nest of other birds. The most remarkable
feature of the nest of neopueo is that birds build a friable roof of brushwood
over the nest, and because of it nest of this bird is more similar to thу nest
of magpie. In completely finished nest of neopueo there is a through pass, and
bird having got into the nest from one side, leaves it from another. And the
long tail of hatching bird sometimes even juts out from the pass. There are
some advantages in presence of the roof over the nest: it perfectly protects
from hot sun, and in the midday heat both birds hide in the nest. Neopueo has
very sensitive eyes, and daylight irritates them.
The neopueo female is larger than male, and she “inherits” the nest: it is located
on her territory, and males ready to make a couple migrate from the next sites
of forest. In clutch there are 2 - 3 eggs, and both birds of couple hatch them
in common. But despite of such care, only 1 or 2 young birds survive to the
age of the moving out from the nest: at these birds the cannibalism is usual,
and the weakest chick is sometimes eaten by stronger brothers and sisters. Sometimes
already grown up young bird throws out another one from the nest, or pecks it
to death, especially if the food is not enough (for example, when one bird of
the couple is perished, and the remained one can’t supply both chicks with food).
Translated by Alexander Smyslov
Australian
false hawk (Neoclanga accipiteroides)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Meganesia, areas of savanna with thickets of trees or rocks.
For birds of prey, the age of man proved to be a serious test of survival. The
destruction and pollution of the habitat, the human persecution and the undermining
of the food supply have destroyed many large species. Outside Eurasia, the role
of the largest feathered predators could
be occupied by the descendants of small birds of prey, which were relatively
diverse even in the epoch of ecological crisis. The exception was Meganesia,
where the niche of the top predators is still occupied by eagles.
But, as in the Holocene, next to large predators there are smaller ones that
occupy their own ecological niche. One of these species is the Australian false
hawk, a descendant of the swamp harrier (Circus approximans).
It is a relatively large bird species: body length is up to 66 cm, weight is
about 2.5 kg, wingspan is up to 1.8 m. The coloration is very variable and varies
from bluish-gray to black, with a lighter underside of the body. The legs are
always dark, the beak and claws are yellow. The false hawk feeds on mammals
ranging in size from a mouse to a rabbit and ground birds. Occasionally, this
predator attacks even well-protected marsupial
spiketails, although it happens mainly in case of hunger. From time to time,
it watches large animals, following their herds and waiting for them to scare
off its prey while moving. The false hawk leads a sedentary lifestyle and, as
a rule, adheres to the same territory throughout its life. Birds jealously guard
the territory from their relatives, arranging a real aerial battle with them,
while making false attacks, scaring each other.
Outside of nesting time, birds stay alone, although a pair of this species is
formed for life – the territories of mates are adjacent, and birds usually fly
into each other’s territory. As a sign of trust in the partner, both birds touch
each other’s feathers and can rest together on a tree, although the male usually
behaves more cautiously – the female is slightly larger than him. Nesting time
comes at the end of the dry season. Lonely birds actively call each other, uttering
a series of monotonous calls, and when forming a pair, males bring a gift –
prey – to the females. The formed pair repairs the old nest of another bird,
or builds a new one – a cup of twigs and green shoots about a meter in diameter.
There are 2-3 eggs in the clutch, which are laid not at the same time. The clutch
is incubated from the first egg for 40 days. The female hatches mainly, but
the male replaces her during hunting. After the hatch of offspring, the female
continues to stay in the nest for another 25 days, during which time all the
worries about prey for the female and offspring fall on the male. The youngest
chick, hatched from an egg laid by the second, is subjected to aggression from
the older one and often dies in the first two weeks of life. At the age of 8
weeks, the young bird is fully feathered and take wing, and at 4 months it is
completely independent. At the same time, parents expel offspring from their
territory. Sexual maturity occurs at the age of 1 year, and life expectancy
is up to 35 years.
A related species, marine
false hawk (Neoclanga maritimus) with a length of 60 cm, lives on the
islands of Melanesia. This bird with lighter (gray) plumage lives in coastal
forests, feeds on fish, and can even catch cephalopods in the tidal strip. In
New Zealand, a lesser
false hawk (Neoclanga minor) with a length of 50 cm lives. This bird
inhabits the lowland forests of the archipelago and feeds on small terrestrial
vertebrates.
This bird species was discovered by Nick, the forum member.
Salt
hawk (Haloaccipiter nudifrons)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Mediterranean lowland, swamps, desert and “oases”.

Picture by Alexander Smyslov
When Africa had collided with Europe and strait of Gibraltar
was closed, Mediterranean sea had gradually dried up in several thousand years.
It had turned to some
practically lifeless (bacteria were kept only) deep lakes with extremely
salt water. These lakes were surrounded with dry and hot desert in which ground
is impregnated with salt. Here it is difficult to survive to mammals exacting
to presence of water and not enduring too hot climate. But reptiles and birds
prosper in “oases” – former islands of Mediterranean Sea which represent
mountains
among lifeless desert. Here it is possible even to find water: at night stones
cool down, and on them the moisture from air is condensed. In some places
of mountain “oases” small streams flow, and there are more-less constant ponds
with fresh water. To get here, mammal should cross many kilometers of district
completely hostile to them.
In Neocene climate began more damp, and the continuous salt desert had receded:
in Mediterranean hollow the extensive salt swamps giving life to the whole
world of small live creatures were formed. Water in these swamps is unsuitable
for drink, and the deadly quagmire preserves “oases” of Mediterranean against
ground animals even more reliably. Only some species of reptiles managed
to colonize these islands among swamps and deserts.
But where ground animal can not pass, there are no barriers to flying creatures.
Therefore birds form the most part of the population of “oases”. Here it
is possible to meet various passerine birds, nightjars and
strange flightless
flamingoes. Here there are no ground predatory animals, but feathered
predators were kept. One of usual species of predatory birds of the Mediterranean
lowland
belongs to hawks. Hawks are among few predatory birds of Holocene managed
to survive in mass extinction which had finished human epoch. Due to ecological
plasticity and ability to coexist humans rather successfully these birds
could
be kept in amount, sufficient for survival.
One of hawks had survived from human epoch is sparrow hawk (Accipiter nisus).
In human epoch this small feathered predator was widely settled even in cities
where it hunted rodents and small birds. In ecological crisis epoch large
prey had disappeared, and because of it the majority of feathered predators
had
died out. Besides habitats of large predatory birds in Holocene were reduced
because of the anxiety factor from the side of people. In such conditions
this bird had received advantages in survival and was widely settled in Europe.
In Neocene descendants of this species meet in various districts. One of
original
descendants of sparrow hawk lives in swamps and desert of Mediterranean lowland.
It is a daw-sized bird; colouring of its feathering is white with black speckles.
On back there are few spots, and on stomach they are much numerous. When
the bird stands on the ground covered with crust of came out salt, it is
very difficultly
to see it from air. This feathered predator frequently keeps on saline soils,
for what it has received the name “salt hawk”. In salt deserts of Mediterranean
lowland it is not enough catch, and the amount of shelters is even lesser.
Therefore salt hawk had changed hunting tactics. This bird hides, dropping
to the ground, and catches flying birds, sharply flying up and dropping them
in air.
Orange beak and paws look bright spots on feathering of bird. “Face” of bird
is naked (it improves heat emission in hot climate) and yellowish. Paws of
salt hawk are lengthened, and the bird seems larger than it really is.
Salt hawk is not specialized to certain kind of catch: it is the universal
predator of salt plains. It catches not only flying birds and insects, but
also eats other kinds of prey. Using long paws, salt hawk snatches out from
water swimming lizards skinkfishes and water
turtlebeetles. Long legs help this bird
to chase on ground small lizards and beetles. Having an opportunity, this
bird willingly eats carrion and catches small inhabitants of Mediterranean
“oases”.
Despite of inclination to life in hot climate, in especially hot time salt
hawk migrates to the north and in mountain areas, to the border of maquis.
This species of birds nests in bushes, preferring richest and shady thickets.
Large ground predators are not found in its habitats, therefore this bird
does not try to make nest at high tree. At lack of bushes salt hawks can
arrange
nest among stones. Birds prefer for nesting shady places in which the weak
wind constantly blows: it protects clutch and nestlings agains overheat.
Nest of salt hawks is simple-looking and cup-shaped; among bushes it is located
at small height from the ground, and among stones birds arrange it so, that
in midday it remains in shadow.
Female and male of salt hawk do not differ in color, but female is approximately
20 % heavier than male. Pair at these birds is formed to all life. Male constantly
renders to female signs of attention, feeding her up with belched pieces
of meat from beak. Otherwise female can start to behave aggressively relatively
to it, and it may cost of life to the male.
In clutch of these birds there are only 2 eggs which female lays with an
interval of 2 – 3 days. Because female begins hatching from the first egg,
nestlings
hatch not simultaneously. They are covered with white down and have opened
eyes; they stay in nest about two months. Parents feed them mainly with birds.
Salt hawks nest two times per one year - in spring and in autumn, dating
the nesting to time of bird migration between Africa and Europe. In favorable
years
they cucceed to bring up both nestlings, but at lack of food stronger of
nestlings can kill and eat another one. Young bird becomes sexual matured
at the second
year of life.
This species of birds is discovered by Simon, the forum member
Owl-harrier
(Strigocircus solitarius)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Easter Island, forest.
The human actions during the Holocene caused the collapse of many ecosystems.
Easter Island was one of these cases. This place was completely deforested and
many of its endemic species perished. However, with the arrival of the Neocene
it was able to recover, developing a new endemic fauna. One of the species that
is now living here is the owl-harrier, a descendant of the cinereous harrier
(Circus cinereus) that reached the island already after the disappearance of
humanity.
When its ancestor reached the island, it found that no species of owl lived
here. This way the owl-harrier evolved to fill the niche of these birds. This
bird is crepuscular, being more active during the dusk and early hours before
dawn.
The female of the owl-harrier is larger than the male with an average size of
46 cm compared to the male’s 40 cm. The wingspan is 90 to 115 cm. Both sexes
have a 40 cm long tail. Morphologically, these birds are similar to an owl.
They have large eyes that allow them to see during the night and a good hearing.
Their plumage is dark grey above with black wingtips and a dark brown rump.
The underparts are pale grey, with a rufous streaked belly. The face is chestnut
brown with a black border. The beak and legs are dark grey.
This species eats mainly rodents and bats, but also birds that are sleeping
in branches during the night. Insects are also eaten, but more occasionally.
While being monogamous and sharing a territory, the pair stays separated during
most of the year, with only some occasional meetings happening. They come together
durig their breeding season, which starts during the summer. During this period,
males and females reforce their bond by engaging in nocturnal aerial courtship
displays and chatter very loudly. This is the only period that they vocalize
more frequently, being silently birds most of the time. The nest is built hidden
in rock crevices. Nests are made of sticks and are lined inside with grass and
leaves. Four to eight whitish eggs are laid by the female, who incubates them
for 32 days. When incubating eggs, the female sits on the nest while the male
hunts and brings food to her and then to its chicks. The male will help feed
chicks after they hatch, but does not usually watch them for a greater period
of time. The male usually passes off food to the female, which she then feeds
to the young, although later the female will capture food and simply drop into
the nest for her nestlings to eat. The chicks fledge at around 36 days old,
though breeding maturity is not reached until 2 years in females and 3 years
in males. Their lifespan is of 22 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Faitiri
(Ornitovenator vitiensis)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Fiji, forests and thickets of shrubs.

Picture by Pavel Volkov
Birds of prey in the human era have lost a significant part
of large species, as well as vulnerable species sensitive to the anxiety factor.
By the Neocene in Eurasia, they were replaced by predatory
corvids and some shrikes,
and in North Africa, woodpeckers
and parrots also appeared
among carnivorous birds. In other places, small birds of prey have survived
and managed to occupy the vacant niches of their large relatives. One such bird
is the faitiri, a descendant of the Fiji goshawk (Accipiter rufitorques), named
after the Maori god of lightning.
Faitiri belongs to the number of large species of feathered predators – the
body length is 80-100 cm, the wingspan is over 1.5 m, the weight is up to 3
kg; the male is smaller than the female. The constitution is gracile, the main
color of the plumage is gray with a darker loin; the top of the head and the
back of the neck are brown, the mature male has a tuft of rusty-red feathers
on his head. The underside of the body is colored lighter than the top side.
The eyes are large, with a yellow iris. This long-legged bird feeds on reptiles
and rodents, trampling them to death. Despite the normally developed wings,
sternum and musculature, faitiri flies rarely and reluctantly. Most often, young
birds do this, settling between the islands. Due to the peculiarities of the
way of life, the claws of this bird are shortened – it is more convenient to
move on the ground this way. Prey is killed with the help of a beak, on which
there is a small jag near the tip, convergently similar to the “tooth” of noble
falcons of the human epoch. Adult birds hunt alone, startling prey by stomping
and flapping their wings. However, breeding partners rarely move out of earshot
of each other, from time to time calling to each other with voices similar to
cawing. After catching up with the prey, the bird tries to press it to the ground
with its foot and bite into the skull or spine.
The mating season is not timed to a specific time, and displaying birds are
found at any time of the year. The leading role in courtship belongs to the
male. Displaying itself to the female, he stomps, nods and raises his tuft in
a mating dance. The most spectacular dance is performed by young and widowed
birds seeking to win the favor of the female. The dancing male holds his body
almost vertically, intensively tapping his feet on the ground or a tree trunk
lying on the ground. At the same time, he fluffs the feathers on the sides of
his head, acquiring a bizarre appearance. Interrupting the dance, the male flaps
his wings and walks in front of the female for a while, watching her reaction.
The nest is built on top of a bush, at its base a large pile of branches and
twigs lies. The female lays from one to three bluish-white eggs, which she incubates
for 45 days. At this time, the male supplies her with food and guards the nest.
Hatched chicks are covered with gray down with white spots. At the sight of
their parents, they vociferate loudly, and the brought prey usually goes to
the most vociferous one among them. If there is a shortage of food, the weakest
of the chicks in the brood is simply eaten by a sibling. At 2 months, young
birds fully fledge and take wing, and at one year of age they already become
sexually mature. Life expectancy is over 25 years.
This bird species was discovered by Nick, the forum member.
Yellow-crested
hawk (Fandraokibo dexter)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: forests of Eastern Madagascar.

Picture by Pavel Volkov
The human epoch is marked by the extinction of a significant
number of species of large birds of prey due to the destruction of habitats
and the impoverishment of the food supply. At the same time, many small hawkbirds
kept a fairly high number and retained viable populations by the Neocene. In
the Holarctic, they did not manage to become large – this niche was occupied
faster by corvid birds. But
in other regions, especially in tropical areas, they thrive, and many of their
representatives have reached large sizes.
One of these species is the yellow-crested hawk, a resident of the Madagascar
jungle and a descendant of the France’s sparrowhawk (Accipiter francesiae).
It is a large forest-dwelling bird of prey, a hunter from the upper levels of
the forest.
The body length of the yellow-crested hawk is up to 1 meter, the wingspan is
about 2 meters. It is a bird with a long tail, shortened legs and large claws,
a small head and a rather large beak. The wings are wide, with rounded ends,
suitable for fast and maneuverable flight among branches and clever aerial tricks.
The color of the upper side of the body and wings is light brown, the lower
part of the body is white; there are dark brown cross stripes on the primary
feathers and tail, and longitudinal short gray strokes on the belly – some feathers
have a dark middle. Legs are yellow, the claws are black. Beak is dark gray
with a yellow cere, the eyes are red. A mobile golden crest grows on the nape,
more developed in males, which is why the birds partly resemble huge parrots.
The female is larger than the male and weighs about 20% more.
Yellow-crested hawks nest on a huge tree, on the uppermost branches, preferring
trees with a broken top, branched in the upper part of the crown. The feathered
predator lives in pairs that last for life. A pair of birds controls an area
of about 50 square kilometers. The activity is daytime, at night the birds sleep
in the nest, huddled together. The pair is remarkable by the attachment of partners
to each other: birds periodically reinforce the relationship by mutual preening,
especially near the eyes.
The yellow-crested hawk’s diet includes mainly large arboreal vertebrates: reptiles,
birds and mammals. It is noteworthy that very large animals are also present
in the diet, including terrestrial ones: forest numidornis,
large species of lemurs and tenreks. When attacking large animals, the bird
tries to pierce vital organs with its claws and inflicts deep wounds with its
beak. Often the bird pursues wounded prey. The yellow-crested hawk can also
hunt from ambush, hiding in a tree among epiphytes and attacking with a short,
quick rush. A bird can easily snatch a hanging
lemur from a branch, turning upside down with its paws in flight. Having
caught numidornis, the hawk eats the prey on the spot, gorging itself to satiety,
and leaves a significant part of the carcass to local predators and scavengers.
Only felimalagasia
is dangerous for the adult yellow-crested hawk.
The breeding season is not pronounced, nesting can occur at any time of the
year. The female lays 2-3 eggs in the nest and begins to incubate them, while
the male at this time supplies her with prey, and most often with a small one.
The chicks hatch in a month. At first, the whole family is fed exclusively by
the male, and the female takes care of the chicks. But after two weeks, the
female also joins him, since young birds become very voracious by this time.
Usually they coexist relatively peacefully, but sometimes a stronger chick eats
a weaker one. In the fourth month of life, their plumage fully develops, although
the crest at this time is represented only by slightly elongated feathers of
background color. Young birds leave the nest and live on the territory of their
parents for a while, but then begin a completely independent life.
Sexual maturity in young birds occurs in the third year of life, but they manage
to occupy a suitable territory, create a pair and get offspring only around
the fifth year of life. Life expectancy is up to 60-70 years.
This bird species was discovered by Mamont, the forum member.
Humminghawk
(Nanastur pugnax)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: South-Eastern Asia – Sunda Land, Jacarta Coast.

Picture by Alexander Smyslov
In Neocene epoch diurnal predatory birds are submitted in
ornithofauna by small amount of species. Extinction of significant amount of
feathered predators had been caused by changes in biosphere at the boundary
of Holocene and Neocene, and also direct prosecution from the part of people
and destruction of habitats. Because of it many groups of predatory birds had
not gone through biosphere crisis. Only most plastic and low-specialized forms
had survived and evolved in Neocene to the set of new species. Mainly, various
hawks were among them. These birds had survived due to absence of strict specialization,
evolved in Neocene to various highly specialized descendants occupying ecological
niches being empty earlier. One of them lives in South-Eastern Asia and in nearby
islands.
When this bird hunts, it can be confused with large species of hummingbirds
dwelt in New World. The small size of body and quickly flapping wings strengthen
this similarity, but instead of thin and long beak of the nectar getting expert
the bird is armed with sharp hooked beak. This combination of features has determined
the name of bird – it is the humminghawk.
Despite of small body size this bird is still a predator – it is the tiniest
species of the order, smaller than birds of prey known to people. By size the
humminghawk is smaller, rather than sparrow, but small growth obviously does
not correspond to its gluttony and hunting courage. As the humminghawk is insignificant,
but belongs to endothermic animals, it has big losses of heat, and because of
it the high requirement for food was developed at this bird: for day it eats
approximately twice more than weighs. Humminghawk eats any catch which it can
overcome: it hunts tiny birds, lizards and amphibians, and also large insects.
Flight of humminghawk is very fast and maneuverable: it can make fast turns
in air and fly even among branches of forest canopy, chasing birds. Till the
flight bird emits muffled rustle by wings (making about 15 – 20 flaps per one
second). Attacking large catch, humminghawk tries to strike by beak in head
or to pierce by claws spinal cord of catch. The bird dexterously seizes fine
catch like insects from branches and eats it right in flight. It happens that
humminghawk attacks even lizards or snakes, whose weight exceeds its own twice
or even three times.
Outlines of body indicate that humminghawk is the skilful flyer: its wings are
narrow and peaked, and tail is wide and fan-shaped. In flight bird can make
sharp turns and somersaluts in air.
Hummingbird of Holocene epoch differed in bright colouring, but their Neocenic
“doubler” has much more dim colouring. The top of body of humminghawk has cryptic
colouring (like the color of bark) – it is brown with black longitudinal strokes
on each feather. Stomach of this bird is grayish-white, on the neck there is
wide black cross stripe with dark blue metal shine. The area around of eyes
of humminghawk featherless and naked skin is colored white. The bared skin serves
for emission of superfluous heat – metabolic processes at tiny bird are very
intensive, and the power consumption is great. One more important purpose of
this attribute in behaviour of birds is being a signal of pacification for aggressive
birds: turn of head removes beak aside and opens weak spot – the eye surrounded
with naked skin. At males “mask” of naked skin is larger, than at females, and
cross strip on neck is wide and black.
Life cycle of humminghawk is very fast – this bird lives not longer than three
years. Pairs at this species of birds are formed only to one nesting season.
The female of humminghawk is larger than the male, in nesting season it is engaged
in care of posterity, and the male continuously supplies hatch with food.
It may be 3 – 4 nestlings in hatch of this bird, but, as a rule, only two young
birds survive up to nest leaving. At lack of forage the weak nestling may fall
a victim to stronger brothers. The female actively protects posterity, attacking
any predators despite of their size. Usual tactics of the bird protecting posterity
is the attacking of eyes and nose of predator.
Till one year this species has about three clutches, and seasonal prevalence
in nesting is not expressed – in tropical climate with feebly marked seasonal
prevalence readiness for nesting depends on physical condition of bird. Feeding
the posterity adult birds grow thin strongly, therefore when the posterity abandons
the nest, adult birds feed intensively. Young birds become able to nesting at
the age of about half-year.
Pernilongo
hawk (Neogeranospiza dolichoscelis)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: South America, open areas, Archipelago of Fernando de Noronha.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. One
of these new species is the pernilongo hawk, a descendant of the crane hawk
(Geranospiza caerulescens). The word pernilongo means long-legged
in Portuguese. This species inhabits the open areas of South America, from the
savannas to the рестинги in the eastern coast of the continent. A population
had reached the Archipelago of Fernando de Noronha, where it became the apex
predator of the islands.
The pernilongo hawk has 55 cm of length and 100 cm of height, with 120 cm of
wingspan. The males are smaller than the females. They have grey upperparts
with a white rump. The central tail feathers are black tipped with white and
the outer tail feathers are white. The head and upper breast are dark grey while
the rest of the underparts are finely barred in dark grey and white. The bill
is bright red at the base and dark grey at the tip. The facial skin and the
characteristic long and strong legs are also bright red. This species developed
a curved, sickle-like claw in each foot in a similar fashion to the extinct
dromaeosaurid dinosaurs.
This species' main prey are small vertebrates, especially rodents, bats, lizards,
snakes, and small birds, but they have also been known to eat larger insects
and other arthropods and snails. These hawks often forage by scanning from a
perch or on the wing and swooping down to grab prey, killing it with stomps
and using their sickle-like claws. However, they are capable of running after
prey. Due to their long legs, they are fast on the ground. While doing it, they
held off their hunting claws from the ground.
The population that inhabits Fernando de Noronha hunts mostly chasing after
the koriko pexerumen, their main
prey on the Archipelago. They will try to subdue the rodent with hits of their
claws and by breaking their bones with stomps.
The pernilongo hawk is a monogamous bird, with the pairs living together even
outside the breeding season. They will also hunt large prey together in the
ground, with the male guiding it toward the hidden female, who will make the
kill.
They nest in small trees, with nests made of sticks, plant roots, and stems,
which are often lined with leaves, moss, bark and plant roots. They are built
mainly by females. She lays two to four bluish white eggs with brown speckling.
The female does most of the incubation. The eggs hatch in 31 to 36 days. The
young begin to explore outside the nest at 38 days, and fledge, 50 days. In
a year, the female can nest two times. Young birds stay with their parents for
7 months. During this period, large prey becomes a common source of food. They
reach sexual maturity with three years and have a lifespan of 20 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Fish
kite (Dolichomilvus piscivorus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: freshwater reservoirs of Europe north of the Alps.
Many birds of prey of the Holocene epoch became extinct as a result of the destruction
of their habitat by people, giving way to corvids
and other birds that showed a predatory tendency. Populations of many species
of small birds of prey turned out to be too sparse for them to recover, and
gradually faded away, unable to withstand competition with representatives of
competing groups of birds. A serious blow to their populations was caused by
the ice age: in particular, the number of osprey (Pandion haliaetus) has greatly
decreased, and this species has completely disappeared in the temperate zone
of the northern hemisphere. Its ecological niche was immediately occupied by
other animals. In tropical Asia, a fishing
warbler appeared, and in Europe this place was occupied by a descendant
of the black kite (Milvus migrans). Seprate populations of this species were
able to survive the ice age in the subtropics, and its descendants returned
to the forests of the temperate zone, where competition with large carnivorous
corvids forced them to choose a way of deeper specialization. In the Neocene,
one of the descendants of this species is the fish kite.
This species is a large, well-flying bird, partly an ecological analogue of
sea eagles (Haliaeetus). The body length is from 63 to 67 cm, the wingspan is
from 175 to 210 cm; females are slightly larger than males. Wings have wide
rounded tips, and tail is forked. The male and female do not differ from each
other in color: the back is brown, the wings have a pronounced reddish tinge,
the belly is gray with mottling. Cheeks have a pronounced ochre tinge, and the
beak is dark gray. These birds have unusual legs – relatively long, helping
to snatch fish out of the water. Like at the osprey, the underside of the toes
is covered with numerous corneous spikes that help to hold smooth slippery prey.
The claws are hooked, the claw on the hind toe is especially large.
Fish kite feeds mainly on fish, rarely eats frogs or snakes, sometimes feeds
on carrion. In the temperate zone, these birds migrate to the south with the
beginning of winter, in the southern areas of the range, where the water does
not freeze, they lead a sedentary life. Birds from the northern and eastern
regions of Europe migrate to the shores of the Fourseas and its islands shortly
before the beginning of autumn frosts. A small population keeps on the shores
of Wenedian Lake. Fish kites look out for prey, hovering in the sky, and grab
animals that are close to the surface of the water. During the breeding season,
they prefer to hunt larger fish species. In spring, birds often switch almost
completely to catching frogs.
It is a monogamous species, the pair persists until the death of one of the
partners. A pair of fish kites owns a territory covering a section of a riverbed
or the shore of some large body of water. There is competition between the couples,
but it is usually limited to display in the air. The beginning of the breeding
season varies somewhat depending on latitude, but in general nesting occurs
once a year, in mid-January – March. Sedentary populations nest early, migrating
ones do it almost immediately after returning to their territory. The male brings
fish to the female during courtship; often both birds preen each other's feathers.
The male regularly has to drive away young competitors from his female. Serious
battles for a female occur only between bachelors or widowed males, but in most
cases they are reduced to a force display, and a fight occurs only in the case
of fierce competition.
The nest is located on a tall tree, less often in bushes (this happens on islands
where terrestrial predators cannot reach), but always near water. This is a
large building up to 80 cm in diameter, consisting of branches and stems of
reeds and cattails. There are 1-3 (usually 2) whitish eggs in the clutch. Chicks
show aggression towards each other with age, often quarrel over food, and usually
only one of them survives before leaving the nest. They are covered with dark
down, the first feathers appear at the age of 10 days, and they fully fledge
in 8-9 weeks. At the same time, they leave the nest and learn to fly, but are
still under the care of their parents. After another 3-4 weeks, the young birds
are completely independent, and at the same time their parents expel them from
their territory. Sexual maturity occurs at the age of 1 year, life expectancy
reaches 27-30 years.
This bird species was discovered by Nick, the forum member.
Riverbank
kite (Pezomilvus unipes)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: freshwater reservoirs of southern Europe and North Africa – the basin
of the Saharan Nile.

Picture by Pavel Volkov
In ecosystems of the Neocene, birds of prey most often occupy
a subordinate position: in the temperate zone of the northern hemisphere, they
are forced to compete with carnivorous corvids and survive due to specialization.
One of the specialized species of feathered predators is the fish
kite (Dolichomilvus piscivorus) from Europe. South of the Alps mountain
range, along the outskirts of the Mediterranean hollow and in North Africa,
its closest relative, a representative of the genus of pedestrian kites (Pezomilvus),
lives – the riverbank kite, also a large bird species that feeds on fish. It
is similar in outline to its relative, but differs in a more graceful build,
which is associated with life in a hotter area. It is a long-legged bird of
prey with wide wings (wingspan up to 170 cm), flying well. The color of the
plumage is dominated by light tones: the plumage on the body is light gray,
the wings are bluish on top, and the belly is white with separate mottling on
the throat. The light color prevents overheating; for the same purpose, a thick
downy tuft develops on the head and back of the neck. The male is slightly smaller
than the female, but does not differ from her in color.
This species has a different tactic of foraging compared to its European relative:
the riverbank kite hunts in the coastal zone of rivers and lakes. This bird
can stand patiently in shallow water for a long time on one leg, and grabs the
approaching fish with the toes of another leg, which was raised while waiting.
Hence the specific name of the bird, meaning “one-legged one”. Sometimes, to
lure fish with a shadow, this kite opens its wings like a black heron. In addition
to fish, this species readily eats crabs, as well as frogs and baby water turtles.
The competitors of these birds are herons, which the riverbank kite tries to
expel from its territory. The claws of this species are very large and hooked;
rows of sharp corneous spikes develop on the tips of the toes from the underside,
helping to hold prey. When walking, special tendons on the upper side of the
toes lift the claws, and their tips do not touch the ground.
Living in a fairly stable climate, when reservoirs are free of ice all year
round, this species is sedentary. Riverbank kites nest in pairs, which can persist
for quite a long time: if nesting is successful from year to year, the pair
remains faithful to each other throughout their lives. Each couple owns a territory
that includes about a kilometer of coastline. This species nests in trees, often
remaking a heron nest for itself, including expelling its rightful owners. Riverbank
kites do not form colonies and treat their relatives rather intolerantly, attacking
them within the boundaries of their own territory. Nevertheless, there are peculiar
neutral territories in the population of coastal kites that are not occupied
by breeding pairs. Being in such places, adult birds show tolerance towards
their relatives, rest together, look for a mate and even hunt next to each other.
In the clutch of this species there are usually 2 eggs with a gray shell. The
chicks are covered with white down; they fledge at the age of about 2 months.
Juvenile plumage is darker than adult one: the wings are dark gray. Young birds
leave the nest and start hunting with their parents. Even in the nest, they
arrange peculiar games, “looking out” for pieces of branches for a long time,
after which they “catch” them. They also actively hunt insects flying by. At
the age of 6 months, their plumage changes to adult one, and they become sexually
mature. Life expectancy reaches 20 years.
Locust-eater
(Pezomilvus locustivorus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: savannas of North Africa, the Middle East, Zinj Land.
The genus of pedestrian kites (Pezomilvus) includes several species of birds
of prey that have adapted to forage on the ground. They have not lost the ability
to fly, but due to their long legs they can run fast. A fish-eating riverbank
kite (Pezomilvus unipes) lives on the coasts of reservoirs in northern Africa
and in Europe south of the Alps. More arid areas of Africa, the Middle East
and Zinj Land were mastered by another species of this genus, leading a completely
different way of life – a locust-eater adapted to feeding on large insects.
Compared to the riverbank kite, it is a bird of lighter build, with a small
head, elongated neck and long thin legs. The wingspan is about 1 meter, the
height of the bird is up to 80 cm, mainly due to long legs. The claws are pointed,
and beards grow on the sides of the toes, pointing to the sides and helping
to knock down and hold prey. The coloration of the locust-eater’s plumage is
camouflage: the background color is grayish-yellow, with thin cross stripes
on the back, neck and wings. The belly is white, the tips of the tail feathers
are black with a white border – when attacking this bird, the predator focuses
its attention mainly on them, which saves the bird from death at the cost of
several plucked feathers. The eyes are masked by a longitudinal smear of black
on each side of the head.
This bird species hunts on the ground. Its main prey is insects, which the locust-eater
grabs with its paws and brings to its beak. This bird is especially active in
hunting locusts and other orthopterans. When hunting them, the locust-eater
is able to make high jumps into the air and picks up insects right during their
jumps. In addition, the locust-eater can catch the orthopterans by ear, determining
the location of the insect by its chirping. Outside of the breeding season,
these birds migrate in small groups behind swarms of locusts, providing themselves
with an uninterrupted and abundant food source. In addition to insects, locust-eaters
eat reptiles, rodents and chicks of ground-nesting birds.
This species migrates widely within its range, occurring in foci, but abundantly.
Locust-eater’s nesting sites are associated with the main locust breeding sites,
and birds feed on these insects while they are at the nymphal stage and cannot
fly. There are up to 10 nests in the colony; birds treat each other quite peacefully
and even jointly defend the nesting site from predators. Nests are arranged
on the tops of trees.
There are up to 3 eggs in a white shell with a few brown specks in the clutch,
but only 2 chicks usually survive until leaving the nest. Juvenile down is light
gray; the plumage of young birds differs from the adult one in a slightly more
saturated color. Young birds leave the nest at the age of about 40 days and
stay together with their parents for up to 15 days. Sexual maturity occurs at
the age of 7 months; life expectancy is up to 15 years.
Vulture
kite (Paragyps afroaustralis)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: South Africa, deserts and semi-deserts.
The mass extinction of the end of the Holocene destroyed most of the large animals,
including predators. Vultures were among the disappeared creatures. They perished
because of initially low numbers and a sharp reduction in the food supply –
a decrease in the number of carcasses of large animals. In Neocene, during the
restoration of ecosystem productivity, the niches of these birds were occupied
by representatives of other bird groups. In North Africa, parrots
and even woodpeckers
did it, and in South Africa these are descendants of the black kite (Milvus
migrans).
The South African vulture kite is a very large bird: body length is 150 cm,
wingspan is up to 310 cm, and weight is about 13 kg. The plumage is brown and
creamy; adult birds are lighter than young ones. There are no feathers on the
head and neck. The skin on the front and upper part of the head is light orange;
a skin fold like a wattle hangs on the neck. The skin of this fold is penetrated
with blood vessels and in the heat it noticeably lengthens and turns red from
the rushing of blood. The beak is long and quite powerful.
For soaring, a heavy bird uses streams of heated air rising from the bare ground,
so the vulture kite lives in open areas, most often with rock outcrops, and
avoids forest areas. This species is most often found in the Karoo and Kalahari,
although it also breeds in eastern South Africa. The vulture kite feeds on carrion
– mainly on carcasses of large animals. But, unlike its Holocene analogues,
the vulture kite is still able to catch small animals. Up to 20 birds can gather
on the carcass of a large animal. With its strong beak, the vulture kite can
tear the skin of an antelope and even a moosehorn,
although it more often prefers to pick up the remains of prey from predators
like marafils. Each couple
controls a territory of several tens of square kilometers.
The breeding season lasts throughout the year, but most often the offspring
can be found in the nests from April to December. Pairs are formed for life,
and birds are very careful about choosing a mate. A great role in this is played
by personal sympathy – often almost formed couples break up without starting
nesting, and are looking for new partners. Courtship is a chase in the air and
joint flights and feeding. Formed pairs hover together for a long time; birds
in forming pairs often chase each other.
The nest with a diameter of 2 meters is located on a rock or a high single tree
and is used for several years in a row. Birds decorate their nests with pieces
of skins or long dry leaves, including palm leaves. Sometimes in the “collections”
there are horns or skulls, and even branches of aloe with leaves, rooting then
in the cracks of rocks and blooming. The stronger the bird, the more decorations;
the weakest birds have a simple nest. Often birds come to another bird’s territory
precisely for the sake of stealing unusual objects from neighbors’ nests.
The rate of reproduction is slow. There are 1-2 eggs in the clutch, which the
female incubates for 55 days. The chick hatches with opened eyes and covered
with down; it grows for a long time and takes wing at the age of 126 days. Sexual
maturity occurs at the age of 2 years, but in fact young birds take part in
reproduction at the age of 3-4 years; life expectancy is up to 75 years. Chicks
can be stolen by agogwes and lezas,
but adult birds have no enemies. Occasionally, birds become victims of predators,
if they are caught off guard on the ground, having eaten too much carrion –
they hardly take off, and prefer perch on a tree for a long time while the food
is digested.
This bird species was discovered by Nick, the forum member.
Fork-tailed
helang (Helang borneensis)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: tropical areas of South-Eastern Asia, north of Meganesia.

Picture by Pavel Volkov
The human actions in the Holocene caused many impacts in the ecosystems. Because
of these actions, many species went extinct in the Neocene. However some species
survived and gave rise to descendants. One of these descendants is the genus
Helang, the fork-tailed eagles. Rather than being a true eagle, these birds
evolved from the kites (subfamily Elaninae). The type species of this group
is the Fork-tailed helang. Helang is a word in Malay that means eagle.
This species lives on the tropical regions of Southeastern Asia and nearby islands,
such as Borneo, Sulawesi and the Philippines. A small population also occurs
in Northern and Central Meganesia.
These birds have a body length of 58 cm and a wingspan of 180 cm. The males
of this genus appear to be larger than the females due to their long forked
tail, which can reach 60 cm. The tail on the females is half the size of the
males, but they are slightly larger than the males in body length. The plumage
of both sexes is similar, they are predominantly white. The nape, rump and wings
are black. The tail is dark grey. The beak is black with a yellow base and the
legs are also yellow.
These birds feed on birds, mammals and reptiles. When hunting in open areas,
they will hover over lowland scrubs and grassland in search of their prey. When
the prey is found, the bird swoops down and takes it. Bats are also eaten, the
bird will wait near the entrance of caves to attack these mammals when they
leave during the sunset.
Fork-tailed helangs are monogamous, mating for life. The couple will defend
their territory from other members of the species fiercely, engaging in aerial
combats. The nesting season occurs at the end of the dry season. The female
lays 2 to 3 eggs on a platform nest high in the tree branches. The female spends
more effort in the construction of the nest than the male. The eggs are creamy
with brown spots. Both parents incubate but when the chicks hatch, the male
spends more time foraging for food. The incubation lasts for 32 days. The young
will fledge 37 days after hatching, but they will be dependent on food brought
by the parents until they reach 80 days of age. This species reaches sexual
maturity with 4 years and have a lifespan of 22 years.
Other species in the genus Helang:
Lesser fork-tailed
helang (Helang minor)
Living in the tropical areas of the Hindustan, this species have 55 cm of length
and a wingspan of 176 cm. The males have a tail that can reach 57 cm, while
that of the females have half of this size. The females are also slightly larger
than the males in body length. Its plumage is mainly white, with pale grey nape,
rump, back and tail. The wings are grey and the shoulders black.
This bird species was discovered by João Vitor Coutinho, Brazil.
Subahu
(Paraharpia ferox)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: endemic to Hindustan, forests of various types.

Picture by Pavel Volkov
Almost all large birds of prey of the Holocene became extinct
in the human epoch, unable to withstand persecution and destruction of habitats.
The remaining populations of the few surviving species gradually degraded and
disappeared shortly after the end of the human era. The fate of small species
of hawk-like birds has developed in different ways. In both
Americas and partly in Africa, they managed to regain their dominant position,
and in Eurasia, with rare exceptions, they remained
on the sidelines, giving place of dominant feathered predators to predatory
corvids. The kites, who were originally “scavengers”, were barely able to
break out into other ecological niches. The black-winged kite (Elanus caeruleus)
turned out to be one of the few Eurasian birds of prey that successfully tried
to break out into a large-sized class. Its descendant was subahu (it is the
name of one of the rakshasas) from the forests of India.
Subahu is a large feathered predator: body length is up to 1 m, wingspan about
2.2 m, and weight up to 8 kg. It is a forest-dwelling species; relatively short
wings with rounded tips and a long tail allow the subahu to maneuver in the
forest. A remarkable feature of the bird’s appearance is the beak – very large
and tall, ivory-colored. The color of the plumage is not bright, gray or light
brown; the male and female do not differ in color. The eyes are red.
Subahu feeds mainly on arboreal animals – small monkeys,
large birds and snakes. This bird also attacks predators hunting in the crowns,
including other birds of prey, and sometimes attacks large prey like the calves
of forest trumpeters.
The mating season is not pronounced, reproduction occurs at any time of the
year. A pair of birds is formed for life and builds a nest at a height of at
least 45 meters above the ground. The nest is renewed throughout the whole life
of the birds together and is used for many years in succession. The male is
ready to fight for the female; the rivals arrange an air fight, performing aerobatics
and arrange false attacks in the air. Usually the established pair easily drives
rivals away.
The rate of reproduction of the species is very slow. There is usually one white
egg in the clutch, less often 2, but almost always one of two chicks kills the
second one. Both parents incubate the eggs for about 40 days. The chick gets
on the wing on the 100th day of life, for another 4 months it learns to hunt
with its parents. Sexual maturity occurs at the age of 3 years, life expectancy
is significant, sometimes reaching 75 years.
This bird species was discovered by Nick, the forum member.
Antarctic
buzzard (Notobuteo ornithivorus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Antarctica and Subantarctic Islands, wintering in South America.

Picture by Pavel Volkov
Initial picture by Tony Johnes |
Birds of prey of Antarctica are not numerous, which is due
to the relative poverty of the continent’s ecosystems. The niche of small predators
here is occupied by peculiar representatives of the grackles,
and the “profession” of scavengers and gatherers was mastered by the caracaras.
The top predator of the ecosystem is an endemic species of buzzard, a descendant
of one of the South American species of these birds of prey.
Antarctic buzzard is a large bird of stocky build, whose weight can reach 2.5
kg, and the wingspan is 170 cm. Females are noticeably larger than males. The
wings are wide; the tail is quite short and slightly rounded. The tarsometatarsus
is feathered.
The main color of the Antarctic buzzard is light gray, almost white, with a
dark gray wavy pattern on the chest and belly. The wings are dark gray, the
primary feathers are black. The back and tail are dark brown, with a wide black
stripe at the edge of the tail. In the population, individuals with a coloration
different from the typical one are quite common: in such birds, the entire body
is colored either light gray or brown. The legs of Antarctic buzzards are yellowish,
and the beak is light gray, with a black tip. The eyes are light brown. Males
and females do not differ in color, and young birds are distinguished by numerous
speckles in plumage and a generally lighter color tone.
The voice of Antarctic buzzards is a loud nasal call.
Unlike other buzzards, which are often adapted to feeding on mouse-like rodents,
the Antarctic species is a specialized ornithophagus. It usually hunts medium-sized
birds: ducks, plovers and large passerines. Small passerines and insects are
hunted only by young birds during training in hunting skills. Antarctic buzzards
look out for prey by hovering above the ground for a long time or perching on
rocks. In the wind, these birds are able to hover in the air for a while, almost
without moving their wings.
A favorite nesting place for Antarctic buzzards is rocks, but where there are
no rocks, they nest among bushes or just on the ground on dry hills. The nest
is a building of shrub branches and (on the coast) dry algae, with a lining
of moss and dry grass. The size and design of the nest directly depend on the
availability of building material and vary from huge structures with a diameter
of more than a meter, existing for decades, to grass-lined holes in the ground
surrounded by several twigs. Adult birds often bring herbaceous plants and green
twigs of shrubs to the nest, which serve for disinfection. Antarctic buzzards
are monogamous and form pairs for several seasons, and sometimes for life, which
is more related to the attachment of birds to the nesting site than to each
other.
Buzzards return from wintering soon after fresh water bodies are released from
the ice and the bulk of Antarctic birds – their prey – flies to the continent.
Buzzard males, who have returned to the nesting sites, perform courtship flights
with calls. After the formation of pairs (or their reunion), the birds begin
to build and repair nests.
Antarctic buzzard females usually lay from two to seven eggs, and they begin
to incubate from the first laid egg. Incubation of each egg lasts about 35 days.
The male brings food to the female, and participates in incubation only occasionally.
After the chicks appear, the female also begins to hunt. Both parents fiercely
protect their offspring from nest destroyers, such as gulls, skuas and caracaras.
This is used by some small passerines that arrange their nests in the immediate
vicinity of the buzzards’ nest.
Chicks leave the nest at the age of about 7 weeks. Not all chicks live up to
this point: the youngest ones usually do not have enough food and they die,
or are eaten by older siblings. In general, the breeding success of Antarctic
buzzards is closely related to weather conditions that directly affect the amount
of food – in cold years, all offspring may die. Sometimes cold springs force
buzzards to completely abandon nesting.
The migration of Antarctic buzzards begins in early autumn, when they move to
the coast of the mainland, where they spend several weeks. Then the birds go
on to South America. Antarctic buzzards spend the winter on the plains of Patagonia.
However, populations on some Subantarctic islands lead a sedentary lifestyle,
hunting seabirds in winter.
This bird species was discovered by Simon, the forum member.
American
fishing eagle-hawk (Aetopiscator americanus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: North and South America, tropical to temperate areas, wetlands, rivers
and lakes.
During the end of the Holocene many members of the Accipitriform order had disappeared.
However, the few survivors continued their evolution. Roadside hawk (Rupornis
magnirostris) was one of these survivors; this bird was able to survive even
in cities during the human epoch. The disappearance of large eagles and forming
of new habitats gave roadside hawk an opportunity to evolve into the genus Aetopiscator,
fishing eagle-hawks. This new genus spread to both continents of New World,
benefiting from the extinction of ospreys and sea eagles. The type species of
this genus is American fishing eagle-hawk, which lives near lakes, rivers and
coastal areas from Southern South America to Mishe-Nama Lake in North America.
Populations living in North America migrate to South America during the winter.
Individuals from the populations living in South America can settle to North
America together with the northern populations and fishing eagle-hawks living
in North America can stay permanently in the wintering areas together with individuals
from southern populations.
American fishing eagle-hawks are large birds of prey, with female being large
than male. She has 83 cm of length, while he has 76 cm. The wingspan is of 178
cm for females, and 170 cm for males. Plumage of both sexes is greyish-brown
with white head with dark mask across eyes reaching to the sides of neck. Belly
and tail are also white. Breast and neck are brown with the wings dark brown
with blacker primaries on top and brown lower side. Hooked beak is dark grey
and feet are pale yellow. Birds have closable nostrils to keep out water during
dives and dense plumage which is oily and prevents its feathers from getting
waterlogged.
All birds of the genus Aetopiscator feed mostly on fish; however, they can occasionally
eat rodents, small birds, amphibians and lizards. Fisher eagle-hawks are capable
to detect prey under water. When the prey is detected, the bird hovers momentarily
then plunges feet first into the water. These birds are capable to take fish
in deeper water, using their strong wings to propel them back to the surface.
American fishing eagle-hawks are sexually mature at the age of 5 years. When
they are old enough to breed, they often return to the area where they were
born. These birds mate for life. However, if one member of a pair dies or disappears,
lone bird will choose a new mate. A pair which has repeatedly failed in breeding
attempts may split and look for new mates. The courtship involves elaborate
calls and flight displays. The flight includes swoops, chases, and cartwheels,
in which they fly high, lock talons, and free-fall, separating just before hitting
the ground.
American fishing eagle-hawk nests near freshwater lakes and rivers, and sometimes
is coastal areas of brackish waters. Nest represents a large heap of sticks,
driftwood, turf or seaweed built in forks of trees, on rocky outcrops or offshore
islets. Female lays two to four whitish with bold splotches of reddish-brown
eggs. Eggs are incubated for about 37 days to hatching and the chicks fledge
in 10 weeks. Its lifespan is about 30 years.
Another species in the genus Aetopiscator is austral
fishing eagle-hawk (Aetopiscator australissimus). Living in Antarctica,
the females of this species are large than the males, with 78 cm of length and
a wingspan of 169 cm, while males have 74 cm and a wingspan of 165 cm. The plumage
of both sexes is white with a dark grey face and wings. Differently of the previous
species, austral fishing eagle-hawk hunts more other birds than fish. This species
migrates in the winter to Tierra del Fuego and southern part of South America
(to the north up to Falkland Islands).
This bird species was discovered by João Vitor Coutinho, Brazil.
Gladiator
eagle (Gladiatornis maximus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Meganesia – plains, woodlands.
In human epoch many predatory birds of the order Falconiformes appeared at the
edge of extinction. It was the consequence of destruction of natural habitats,
chasing of these birds, the increased factor of anxiety and the result of use
of pesticides in agriculture. As a result species of feathery predators which
could not adapt to life near to people had died out. But human activity promoted
the prosperity of separate species of these birds. In Australia actively bred
rabbits turned to main food source of local wedge-tailed eagle (Aquila audax).
Due to presence of extensive territories not populated with people this species
managed to survive in human epoch, and had given rise to the several Neocene
species which had occupied various natural habitats of Meganesia, the continent
included Australia and New Guinea. In savannas of Meganesia the descendant of
this eagle is the monstrous gladiator eagle, a huge predatory bird: wingspan
of this species is over 4 meters, and weight reaches 14 kg. It is one of the
largest flying birds of the Neocene world. Only nomade
albatross (Thalassocrator magnificus), a sea bird of the Southern hemisphere,
valkyrie owl (Valkiriostrix robusta)
from temperate areas of Eurasia and North America, and huge bird acatu
(Sciopterornis acatou) from South America surpass this one.
Meganesian gladiator eagle is the analogue of dyed out in early historical time
New Zealand Haast’s eagle (Harpagornis moorei). This species is one of top predators
of plains of Meganesia: it eats large representatives of megafauna - large flightless
birds false mihirungs and false
moas, and also descendants of camels camelopes
and young caballocameluses.
It imposes essential restrictions to settling of gladiator eagle: it lives only
in places plentifully inhabited by representatives of megafauna. But out of
nesting period gladiator eagle moves far from usual habitats – to deserts of
southwest of Meganesia, to coast of Eyre Gulf, and even to foothills of Great
Dividing ridge.
The gladiator eagle is a large bird. Partly the impression from its size is
amplified by very big legs with long hypotarsuses of this bird. Growth of gladiator
eagle standing on the ground, reaches 80 cm. Feathering of this bird is yellowish-brown;
primal feathers are white. On head, shoulders and the top part of back feathers
are dark brown. The area of head around of eyes and beak is lack of feathers
and covered with naked grey skin – this adaptation helps to keep cleanliness
of plumage during the feeding.
Gladiator eagle looks for prey, soaring highly. This bird is lack of maneuverability
and speed of falcons of Holocene epoch – its prey is made of rather sluggish
creatures. But the weight of prey many times over surpasses the weight of feathery
hunter that creates the big danger to attacking bird. Attacking on prey, gladiator
eagle tries to kill it as soon as possible. It seizes by claws neck and back
of prey, and puts a strong bite by beak in basis of skull. The beak of gladiator
eagle is very strong, also it is similar a little to beak of parrot – the bird
easily crushes bones of small animals by it. In connection with habit of killing
of prey the skull of this eagle has strongly changed: bird has powerful maxillary
muscles, and for their attachment the skull of bird has extended. The head of
gladiator eagle seems flat and wide in comparison with heads of eagles of Holocene
epoch. Tip of beak of this eagle is strong and pointed. During the fatal bite
it destroys myelencephalon of prey where the vital nerve ganglions are concentrated,
and after that even the largest prey instantly perishes. Usually these eagles
attack young animals, but small camelopes become their prey at any age. Gladiator
eagles are able to break open a skull of prey, and often eat brain of killed
animals. Also these birds search for the rests of prey of other predators (including
congeners), and at lack of large prey catch small animals. If these eagles are
not occupied with nesting, they spend the day near the prey and feed on carcass
some times in succession; at this time they drive scavengers away from the prey.
The nesting pair does not come back to the prey the second time.
Gladiator eagle nests on high trees. The pair at this species forms to all further
life. Partners express attachment to each other by poses and movements. Male
is smaller, than female, and behaves very circumspectly in relation to her.
Shortly before nesting male starts to feed up the female, bringing for her pieces
of meat and cautiously delivering it from beak to beak. If birds hunt in common,
male of gladiator eagle gives up the place near prey to the female and from
time to time feeds up her.
Breeding rate at these birds is very low: nesting occurs once in two years,
and only one nestling lives up to flight from nest. Nesting of these birds has
no strict attachment to seasons, and in droughty years birds can not nest at
all till some years in succession. The pair of gladiator eagles builds eyrie
at top of tree – eucalyptus or large acacia. This eyrie is annually renewing
and is used by birds till many years. In clutch there are two eggs hatched only
by the female. Male feeds her in nest, and the female leaves clutch only for
short while to take dust-bath and to stretch wings. The hatching lasts till
about 40 days, and the nestling leaves nest at the age of 4 months. If two nestlings
hatch, weaker of them, as a rule, perishes from lack of food or aggression from
the side of stronger nestling. Connection of parents and posterity lasts for
very long time and is too strong. The young bird during 4 – 5 months after leaving
from nest feeds in common with parental pair, training simultaneously in receptions
of hunting. Young bird frequently plays, “attacking” prey killed by parents,
or frightening by false attacks herds of herbivores. The young growth nests
at the fifth year of life; life expectancy may reach 40 years.
This species of birds is discovered by Simon, the forum member
Aella
(Aella atropos)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: rainforests and mountain forests at the north of Meganesia.

Picture by Pavel Volkov
Rainforests formed at the north of Meganesia are very productive
ecosystems. They give a wide choice of refuges and kinds of food for various
live beings: invertebrates, reptiles, amphibians, birds and mammals. The majority
of inhabitants of forest canopy belongs to the number of vegetarians, omnivores
and insectivorous animals. Abundance of prey involves to forest canopy a number
of predators: snakes, carnivorous marsupial mammals and birds. The supreme feathered
predator of rainforest canopy of Meganesia by right is aella – forest species
of eagles, the descendant of Australian wedge-tailed eagle (Aquilla audax).
Other descendants of wedge-tailed eagle live in various biotops. Largest of
them is gladiator eagle (Gladiatornis maximus),
the inhabitant of savannas eating representatives of megafauna.
Aella differs from gladiator eagle in much smaller size: wingspan of this bird
seldom exceeds 250 cm. At this bird species males are smaller, than females.
Aella may be easily recognized among inhabitants of Meganesian forests because
of white plumage with black marks. Separate feathers on breast and stomach of
bird have black spots on tips, forming irregular pattern. In some cases the
plumage on stomach may be snow-white. On nape and back part of neck of this
eagle feathers are black more, than on half, and form a similarity of cop. Covert
feathers of wing are white with wide black border. On wings of perching bird
the characteristic scaly pattern is formed. Feathers on back and waist are almost
completely black.
Wings of aella are rather short and wide, and have rounded tips. Primarily feathers
are white with black tips. Long tail helps bird to make sharp turns in air,
when aella chases prey. Aella is a master of air acrobatics. Despite of large
size, this bird dexterously flies in forest canopy, and even can seize in flight
animals hidden from it on the bottom side of branches, having turned upside
down to one instant.
In the name of this bird two names of characters of antique mythology are joined:
one of harpies, violent spirits of storm, and moyra breaking off a string of
life. This ominous name is quite justified by predating bents of aella. The
present eagle is analogue of South American harpy eagle (Harpia harpyia) and
other forest eagles. Aella eats various birds, possums and reptiles. Skilfully
manoeuvring among branches, this eagle can easily seize the prey having hidden
on trunk or on branch. The bird can break from tree by one jerk and easily carry
away prey weighing up to 3 kgs. The animal caught by aella perishes instantly:
claws about 7 centimeters long are stuck into its body. Aella has deep cutting
beak with long bent tip, which easily tears skin of possum or dense scales of
reptiles. The cornea of beak is colored cream shade.
By its habits aella is furious and fearless predator. Birds protecting nest,
can attack without any doubt even marsupial
biruang many times surpassing them in size. Claws and beaks of birds protecting
the nest are dangerous weapons.
The nest of aella is arranged at the top of tree broken by wind, or among large
branches. If the forest grows on hillsides, aellas nest in rocky niche. The
nest of these birds exists till many years. Birds regularly renew it, spreading
a new layer of branches and leaves atop of last year’s one. In the bottom part
of nest of aella epiphytic plants, masking nest at sight from below, frequently
expand. The pair of birds occupies extensive territory – of about 30 square
kilometers.
Aella’s life is very long: average life expectancy of these birds exceeds fifty
years. The constant pair forms, when partners reach complete physical maturity
– at the eighth year of life. Up to this age birds can form temporary pairs,
which can break up easily though birds have time to bring up one or two hatches.
In clutch of aella there are only two eggs laid by female with an interval of
3 days; the hatching begins from the first egg and proceeds till about 50 days.
The nestling of aella sharply differs from adult birds by color: its down is
brown or almost black. Juvenile colouring suppresses aggression of adult birds
and stimulates their parental behaviour. Nestlings hatch not simultaneously,
and the difference in age renders decisive influence to the success of their
survival. Usually elder nestling since the first days of life starts to attack
younger one, and at lack of food simply eats him. If the forage is abundant,
the further development of nestlings smooths age distinction, and they start
to behave more tolerant to each other. But at food shortage aggression between
them may amplify at any age, and usually ends by pushing out of weaker one from
the nest by stronger nestling.
Development of nestling of aella lasts till about four months. The young bird
has dark juvenile plumage – it is grey with black marks. Parents finish feeding
of young bird about one month after it leaves nest, and later start to show
aggression to it, compelling young bird to leave their territory and to search
for new place for life.
Naked faced eagle (Griffonaquila nudifrons)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Meganesia, plains and woodlands.
In human epoch many species of birds of prey had hard times. Their number had
been decreased because of exterminating, destruction of habitats, anxiety and
pesticide using. When the human epoch was finished, the majority of species
of feathery predators had become extinct or was presented by residual populations,
too rarefied and genetically uniform for the further restoration of the species.
In Eurasia large predatory birds – eagles and see eagles – had been replaced
by eagleravens (Aquillorax spp.), which occupy various ecological niches and
live from steppes up to the coast of Arctic Ocean. But in other continents
representatives of true birds of prey, including descendants of Holocene eagles,
were kept. Some species of descendants of wedge-tailed eagle (Aquila audax)
inhabit Meganesia, living in various natural zones. In tropical forests at
the north of Meganesia large eagle aella (Aella
atropos) lives, and plains
of Meganesia are under the rule of huge gladiator
eagle (Gladiatornis maximus),
which prey includes representatives of local megafauna.
When gladiator eagle is lucky in hunting, around of it various meat eaters
gather very quickly. Usually eagle eats rather small part of carcass, and the
rest is a share of scavengers. Among the animals waiting for the share near
prey of the gladiator eagle, the large flesh-eating birds behaving safely and
independently are remarkable. They are perfectly adapted to feeding on corpses
of large animals – the forward part of their heads is lack of feathers and
covered with dark red skin. They are not vultures, but the close relatives
of gladiator eagle, descending from the common ancestor – naked faced eagles.
Naked faced eagle is rather sizable bird. The height of the bird standing on
the ground reaches 70 cm, and its wingspan is about 3 meters. In proportions
of these birds long legs and neck pay attention to themselves. These features
indicate the ability of bird to walk good on the ground and to feel free among
high grass of plains. Claws of naked faced eagle are rather short, thick and
blunt: this bird does not carry prey in claws, and brings food to nestlings
in stomach or in the beak. But such claws do not prevent walking and running
on the ground.
Wings of naked faced eagle are wide; they have long primarily feathers and
rounded tips. Using ascending streams of air, naked faced eagle can soar till
the hours above plains, looking out for dead animals or hunting predators.
These birds watch closely one after another and after hunting gladiator eagles.
If the gladiator eagle attacked prey successfully, naked faced eagles searching
for food within a radius of five kilometers from the place of its hunting quickly
gather to it. Tail of this species is short, and has the lengthened feathers
in middle part.
The feathering of naked faced eagle has soft colouring: brown with white primarily
feathers; back and neck are darker, than trunk and wings. Stomach and vent
of birds have sandy-yellow color. The forward part of head – forehead, beak,
area around of eyes, cheeks and throat – lacks of eathers and is covered with
skin of meaty red color. Large yellow eyes are surrounded from above with small
skin combs, which surface is covered with small warts. The beak of bird is
very long and resembles a little the beak of vulture of human epoch. These
birds are not able to tear thick skin on corpses of large tetrapods, and do
not start meal at once, having found out a corpse. Usually they wait for appearing
of birds with powerful beaks or the predatory mammals, capable to tear skin.
The long beak permits to penetrate deep inside the corpse and to eat away interiors
of prey. Naked faced eagles can hunt independently medium-sized prey – kangaroos,
cubs of local camels and chicks of large
flightless birds. But more often they
prefer to search for the rests of prey of large predators, even at early stages
of decomposition. Extremly acidic digestive juice interferes with infection
of these birds with bacteria.
Naked faced eagles inhabit plains of Meganesia in places where there are rocks
or large single trees on which they arrange nests. Pairs of these birds are
kept for many years, and each year they nest in the same place, renewing and
repairing old nest. In clutch at this species there are two eggs, but only
one nestling grows up to adult condition. The second nestling, if it hatches,
perishes during two first months of life from aggression of stronger congener.
The nestling develops for very long time: it stays in the nest till about half-year,
and keeps near parents till approximately the same time, training in hunt and
in search of carrion. The young bird reaches the sexual maturity at the fifth
year of life, and life expectancy of naked faced eagle reaches fifty years
and more.
Seashore
griffon eagle (Griffonaquila littoralis)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Accipitridae)
Habitat: Meganesia, the south of Indonesia, coasts of large reservoirs, seashores.
Descendants of Australian wedge-tailed eagle (Aquila audax) inhabit various
areas of Meganesia – from savannas up to tropical forests. But one species of
these birds had developed the completely particular habitats – coastal zones.
After storm sea coast represents a plentiful table for scavengers. In Northern
Atlantic sea coasts are patrolled by “sea
vultures”, very large birds of skua family. And at the opposite side of
the Earth, at the coast of Meganesia, the birds very similar to them and leading
the similar way of life wander – these ones are seashore griffon eagles. They
are descendants of wedge-tailed eagle and close relatives of naked
faced eagle from savannas of Meganesia. But these birds lead completely
different way of life. Couples of seashore griffon eagles patrol coast in searches
of the animals cast ashore by storm. Their prey includes large fish and various
sea animals.
Seashore griffon eagle, like its relative from savannas, is rather large bird:
its growth reaches 1 meter. But the constitution of this bird is very gracile
– this bird resembles secretary birds and herons. Long legs with sharp claws
make about a half of bird growth – this bird is able to keep small prey in paw
and to bring it to the mouth, as if a parrot. Neck of seashore griffon eagle
is also long enough, flexible and mobile. Head is large and has lengthened beak
which tip is hooked. Head and top part of neck of this bird lack of feathers,
and nostrils are supplied with leathery covers and can close densely. It is
connected to features of food behaviour of bird – seashore griffon eagle gathers
prey in water and frequently immerses head in water completely, gathering small
invertebrates.
This bird is good flyer. As against the savanna relative, seashore griffon eagle
has rather narrow wings and flies not due to ascending streams, and using permanent
light wind. Wingspan of this species is about two meters; the female is little
bit larger than male. The feathering of seashore griffon eagle has very light
colouring in comparison with savanna species – back and wings are almost white
with slight beige shade, and neck and breast are straw-coloured. Primarily feathers
have black tips, feathers of tail are almost entirely black, and only at the
basis they are light yellow. Head and featherless part of neck are covered with
smooth pinkish skin; above eyes there are small longitudinal combs.
Seashore griffon eagles live in couples and have extensive territory. Usually
the couple controls a narrow strip of coast about 20 kilometers long, but in
mangrove thickets and among islands territories may be shorter, but much wider.
These birds willingly settle near freshwater reservoirs and are usual representatives
of fauna in vicinities of Arafura and Carpentaria lakes.
These birds relate to congeners with the moderate degree of aggression. Near
large prey some couples of birds may gather, but if food is not enough, owners
of territory may banish newcomers, attacking them in air or pushing aside from
prey on the ground.
The pair at seashore griffon eagles is kept till the whole life. Partners concern
to each other very gently, constantly displaying signs of attention to each
other. Forms of expression of mutual attachment include feeding the partner
and cleaning of naked skin on its head, especially near to eyes. In courtship
season male displays to the female the clumsy dance including jumps with opened
wings. Right before nesting male and female exchange with “gifts”: they entrust
each other twigs, pieces of stalks of plants and other material, suitable for
nest repairing. The huge nest (up to three meters) is building and renewing
till the whole life of the couple. Sometimes the tree on which it is arranged,
does not maintain its weight and falls. On rocks nests are more durable – sometimes
they are used by some generations of birds in succession.
In clutch of this species there are two eggs. Female and male incubate them
alternately during two months. Nestlings relate to each other rather peacefully,
and approximately in two cases of three parents succeed to bring up both young
birds. Young birds leave nest at the age of half-year. For some time they live
in parent territory and fed in common with parents, but after 1 – 2 months parents
banish them.
Young birds become capable to nesting at the age of 4 years. Life expectancy
reaches 45 – 50 years.
Giant
crested Eagle (Megaetus cristatus)
Order: Hawk birds (Accipitriformes)
Family: Hawks (Acciptridae)
Habitat: Malayan peninsula, islands of Indonesia.
Usually mammals are the main terrestrial predators in Cenozoic. In early Cenozoic
birds had made an effort to become the dominant predators, but only in conditions
of isolation in South America this attempt had been successful. In other cases
birds lost the position of terrestrial apex predator to mammals.
The Neocene birds have evolved into large predators: in New Zealand there is
the ruakapangi, at the Hawaiian islands
kuahana lives, and in deserts of North America the praire
groundowl is found. These species can easily compete to mammals, living
near them. In South-Eastern Asia there is also the terrestrial bird of prey
of its own kind – one of the few species of accipitriform birds survived to
Neocene.
Giant crested eagle, reaching the body length of one meter, is one of the largest
birds of prey in South-Eastern Asia. A tireless hunter for smaller creatures,
this feathered giant is one of the few accipitriform birds almost completely
lost the ability to flight and passed to terrestrial habit of life.
Giant crested eagle looks externally like a typical eagle, but it has traits
of adaptation to new way of life. It has large hooked beak to kill its prey,
but the talons on its feet are less hooked to allow walking on the ground. This
eagle looks much larger than its relatives from Holocene era because of the
changes of body proportions. It has long and strong legs, so the height of this
eagle at the shoulder is over one meter. It has powerful pectoral muscles, and
its wingspan reaches three and a half meters. This eagle retains the ability
to fly, but it takes to the wing only in extremely rare cases. This bird’s wings
are broad and rounded at the tips, and tail is fan-shaped with smooth sides.
There is a small crest of white fluffy feathers on the head of this bird.
The plumage of those birds has dull black and white coloration and cross-striped
pattern. These stripes are thinner on the chest and head, and on the wings and
tail they are broader and pattern is clearly visible from the distance. This
pattern perfectly merges with the chiaroscuro world of the underbrush, where
the eagle hides in ambush. This powerful but not very agile bird prefers to
attack from ambush and kills its prey with powerful beak impacts; also it eats
rests of prey of more agile carnivores or searches for carrion. Adult eagles
are strong enough and well armed to fear predators, but various snakes and carnivorous
mammals sometimes eat eggs or chicks of these birds.
Giant crested eagles nest on the ground, gathering various fallen vegetation
and leaves to a pile and laying 1—2 eggs on top of it. Unlike their smaller
relatives only the female hatches eggs and a little bit smaller male brings
food. When the nestlings hatch, the parents switch roles: male remains near
the nest and takes care to chicks and female hunts and drives away the intruders.
Hatchlings grow quickly and soon they leave the nest and begin waiting nearby
for the parents bringing food to them. They resemble in constitution the gallinaceous
birds rather than smaller hawks or falcons. Young eaglets grow quickly and at
the age of one year they are completely independent. The juvenile coloration
of these birds is also striped, but there is rusty reddish color instead of
white. Three years old birds acquire the adult coloration and become sexually
mature. The life span of giant crested eagle is about 50 years.
Giant crested eagle competes successfully to carnivorous mammals because the
early stages of the evolution of this species took place in island conditions.
These habitats could not supply large carnivorous mammals with food, and large
population of eagles could exist in such conditions due to regular migration
of birds between the islands. The return of giant crested eagle to mainland
Asia seems a secondary event that took place after the forming of this species
of bird at the archipelago of Indonesia.
This species of birds is discovered by Bhut, the forum member.
Translated by Bhut.
Ethereal kite (Seraphimornis gracilissimus)
Order: Hawk birds (Accipitriformes)
Family: Ethereal kites (Seraphimornitidae)
Habitat: tropical areas of Pacific Ocean, seashores and large rivers near forests
tracts.

Picture by Amplion
Colorization by
Carlos Pizcueta
In Holocene flesh-eating falcon birds had great damage from human activity.
The majority of species of these predators became rarity – especially large
“noble” falcons and the most part of eagles. Ecological crisis at the boundary
of Holocene and Neocene had resulted in mass extinction of animals that became
the reason of disappearance of large feathered predators. Because of it up
to Neocene this group was present by not numerous low-specialized forms –
mainly by hawks. In Neocene corvid birds had substituted large feathered
predators
of Old World, and descendants of butcherbirds had replaced falcons. In such
conditions falcon birds evolved to very specialized forms.
Above river fauces, in mangrove thickets and above the coastal sea waters one
representative of Neocene accipitriforms hunts. It is a medium-sized bird with
long legs and peaked wings. Its wingspan makes about 2 meters, but long primarily
feathers make a significant part of this size. These birds were widely settled
in tropics of Indian Ocean and have reached even the far islands of Pacific
Ocean. This bird is named seraph kite or ethereal kite, and is one of the best
flyers on the Earth.
Ethereal kites are capable to stay in sea far from the coast till up to three
or four days. Due to long wings bird uses air streams with the same dexterity
as the albatross, and almost does not make efforts to flight. Forked tail
with firm edge feathers provides the bird good maneuverability in air.
Ethereal kite has long and rather weak legs. It spends a little time on the
ground – mainly during egg hatching and posterity care periods. This kite
seldom walks on the ground, and prefers to have a rest, perching on large
branches
high above the ground. Ethereal kites gather for rest on trees with the broken
top and thick branches especially frequently.
Weak legs do not permit this bird to hunt terrestrial animals, but ethereal
kite eats absolutely another prey. Its food includes fish and aquatic invertebrates;
bird snatches them out from water and brings to mouth, not interrupting its
flight. During their sea flights ethereal kites receive water strictly from
the eaten prey, but in estuaries, where the top layer of water is fresh,
they can drink it like swifts, flying right above water surface.
The sexual dimorphism at these birds is expressed precisely enough: the female
is approximately a quarter heavier than male is. Colouring at birds of both
genders is identical: light grey with thin cross ripples on breast and white
stomach. Edges of wings and tips of tail feathers are black. At males legs
are colored light orange, at females they are yellow. In courtship period
legs of male become bright orange, and at separate individuals they turn
almost
red.
These birds nest the year round – in tropics the regularity of nesting is
not expressed. Pairs at ethereal kite form only for one nesting season and
break
up when young birds become independent. Male starts the display. It chooses
a tree with flat top or forked branch to which it is possible to fly up easily.
It drops into branches some large rods making a basis of nest, and begins
courtship flight above the chosen place. The bird flies upwards almost vertically,
and
begins slow spiral descent, using ascending streams of air. Such flights
may proceed for some hours in succession. If the female is interested in
male,
he shows a place for nest to her, and birds build it up in common. Birds
make friable nest, throwing in suitable fork of branches rods and branches
picked
up on water surface. From time to time they fall to the nest and trample
it down, giving the necessary form to the basis of nest.
In clutch of these birds it may be one or two rather large eggs (compared
to large hen eggs in size). Egg shell is colored yellow with brown irregular-shaped
sopts. Nestlings hatch after 18 days of incubation and develop rather fast.
The bi-monthly bird already has completely advanced feathering. It leaves
the
nest and gradually learns flшпре skills. The young growth spends about one
month with parents, and then leads independent life. In first two years of
life plumage of bird is grey with bluish shade and without black marks. Life
expectancy of these birds makes about 30 years.
The idea about existence of this species of birds was proposed by Bhut, the forum member.
Safat
(Seraphimornis safatus)
Order: Hawk birds (Accipitriformes)
Family: Ethereal kites or Seraphims (Seraphimornithidae)
Habitat: mountains in north-west and north Africa, south and south-west Europe.
In the Holocene, many species of hawk birds falled victims of mass extinction
and disappeared, yielding their ecological niches to other predators – American
vultures, corvids, shrikes, etc. The descendants of the few surviving species
often acquired new skills and specializations; ethereal kites or seraphims are
one of such specialized forms of hawk birds of the Neocene. They have adapted
to a continuous flight and are quite capable of even crossing the oceans, traveling
between islands.
Safat is one of such beautiful flyers. Named after a mythical bird that has
never landed, this ethereal kite is, of course, connected with the ground, and
even more than its related species – it perches for the rest on tree branches
or on mountain cliffs. It has well-developed and strong claws and legs, but
safat prefers to grab and even eat its main prey – various insects, small birds
and other small animals – on the fly.
Safat is a medium-sized bird, 50-70 cm in length, including a long forked tail.
It is covered with light gray plumage, only the tail is darker. The color of
the beak is also dark; it is relatively short, but wide: safat swallows smaller
prey, such as flying locusts, grasshoppers or dragonflies in flight. Safat knocks
down larger prey, usually birds, with beak impacts due to a mobile and strong
neck. It also grabs prey with its feet and eats it, bringing it to the beak
and tearing pieces from it.
Safat hunts with the help of vision, and its nictitating membrane has evolved
into special glasses that allow it not to go blind in bright sunlight, including
one reflected from the salt desert of the former Mediterranean Sea – like other
seraphim kites, safat can not drink for several days and freely flies into these
inhospitable lands in search of prey – small birds or flock of orthopterans
migrating between Africa and Southern Europe. Sometimes it falls prey of local
hawks, but it happens more often with young and inexperienced, or with too
old birds. A healthy individual of this species can fly away from almost any
other bird, as well as escape from a salt storm, or fly over it, and wait until
it ends. Similarly, safat avoids dust storms in Africa and Europe, and then
returns to these places in search of animals killed by the disaster.
This ethereal kite nests in the mountains – on cliffs or in trees, in places
inaccessible to terrestrial mammals. The mating period in safat is not pronounced.
The nest arrangement is typical for birds of prey – it is a massive structure
of piled up thin branches topped with a loose nesting platform. In a clutch
there are 2-3, less often 4 eggs with a pale brown shell. Both parents hatch
– sexual dimorphism is absent in this species, males and females look the same
and have the same size.
The chicks hatch in a month. Covered with down, they are initially completely
helpless and depend on their parents. At this time, they are especially vulnerable,
and they are saved from predators only by the inaccessibility of the nest. They
grow fast enough, and at one and a half to two months old they are already fully
fledged, and at the age of three months they are able to fly. For the first
time after leaving the nest, they stay on the parental territory, but after
six months they leave their parents forever.
This bird does not show much intelligence or complexity of behavior; however,
it has a very good memory, which allows safat not to get lost even among the
monotonous landscape of the salt desert. Also, like swifts, safat can sleep
in the air for a while, which he does quite often when it is not busy in taking
care of chicks. Safat flies especially spectacularly when the male of this species
courts the female: he flies around her in circles, and then displays more complex
aerobatics, showing his force and endurance; if the female likes him, then she
joins him. It happens that birds catch small prey or just grab a piece of a
branch or a dry leaf, and start playing, throwing it to each other in flight.
These birds are strict monogamous, and couples often stay with each other for
life.
The average life expectancy of this species is 15-20 years, rarely more. Sexual
maturity in young birds occurs at the age of 2 years.
This bird species was discovered by Bhut, the forum member.
Ghost
Barn Owl (Tyto spectra)
Order: Owls (Strigiformes)
Family: Barn owls (Tytonidae)
Habitat: European maquis.
Neocene was a period when many large diurnal birds of prey became extinct, and
later their niche was occupied by birds of other orders and families – corvids,
shrikes, etc. Nocturnal predators – true owls and barn owls – were more fortunate:
the nocturnal lifestyle helped the species that survived during the Holocene-Neocene
ecological crisis to survive the initial rivalry with new birds of prey and
eventually even master a partially diurnal lifestyle, forming new species during
this process. One of these species is ghost barn owl.
This bird is one of the descendants of the Western barn owl (Tyto alba) of the
Holocene epoch. It is about the same size as its ancestor – on average about
50 cm long, but with stronger and longer legs and wings; unlike most other barn
owls, ghost barn owl feels free both in trees and on the ground. Compared to
its ancestor, it is colored quite variegated – on the brown background of back
and wings, lighter, even whitish spots and stripes are scattered, which makes
this bird less visible during the day among the light and shadows on tree branches
or in bushes, where it hides from other birds at this time of day. The head
is rounded; face disc is well developed, but less noticeable, since the feathers
on it are of the same color as the rest of the bird’s body. This species also
does not have feather “horns”, like other barn owls. The inner side of the wings
is white; the disturbed bird takes off, stunning a potential enemy with a bright
flash of white feathers and a loud unpleasant screech – a penetrative squeal
(hence the epithet “ghost”). An undisturbed bird prefers to run on the ground
rather than fly. It can exist relatively successfully even in a treeless area
on the outskirts of Mediterranean lowland.
Like all other owls, ghost barn owl is a bird of prey. Its main prey is small
rodents and insects, less often small birds. Ghost barn owl gets most of its
prey on the ground. In search of prey, it relies not only on sight, but also
on hearing. Having heard its prey, ghost barn owl usually sneaks up to it on
the ground, sometimes very slowly, and ometimes running fast. Only the last
few meters it usually takes off into the air and rushes at its prey from above.
Its legs somewhat resemble the legs of Cretaceous raptor dinosaurs, but without
specialized claws; ghost barn owl's claws are blunted due to a more terrestrial
lifestyle, and it kills its prey with a “bite” of its powerful beak. But toes
of ghost barn owl are tenacious and strong, this bird can press the caught animal
to the ground with them and perch on a branch, although it feels more comfortable
on the ground.
Courtship season at this barn owl takes place in the second half of the dry
season; this bird is monogamous, and pairs are often formed for life. Males
and females court each other, performing peculiar dances – they fly up alternately
or simultaneously, stamp their feet ceremoniously or run after each other. A
few days after that, the female equips the nest, lays 1-4 white eggs and hatches
them for about 5 weeks. Ghost barn owl does not build complex nests: it can
use the nests of other birds, tree hollows, caves and burrows, but it actually
does not make nests itself, adding the litter only. Male does not hatch the
clutch, but feeds his female. When chicks hatch from the eggs, both parents
feed them for about 5 weeks before juveniles take wing, but not all young birds
survive to independence – various predatory animals and snakes ravage nests
of these barn owls. These birds also often have cases of cainism, especially
when there is a shortage of prey.
The life expectancy of ghost barn owl is up to 30 years, but most of these birds
die much earlier.
This bird species was discovered by Bhut, the forum member.
Suindaraçu,
or Great Suindara (Tuidara magna)
Order: Owls (Strigiformes)
Family: Barn owls (Tytonidae)
Habitat: tropical forests of South America, old forest areas.

Picture by Alexey Tatarinov, final refinement by Pavel Volkov
During the Holocene, human interference in nature caused the
extinction of many species. Only those that were resilient enough were able
to survive and left descendants. One of these survivors was the American barn
owl (Tyto furcata). Being common and capable of surviving to the anthropogenic
impacts, this species continued their evolution and gave origin to a new genus
in the Neocene, the Tuidara (a local name used for the barn owl in Brazil).
The type species of this genus is the great Suindara. This species is endemic
to South America and inhabits tropical rainforests, being found mostly in areas
where dense, old-growth forest is profuse. However, it may enter secondary habitats,
such as forest edges, especially while hunting. Occasionally, they can also
be found in dry forests, wooded savanna plains and semi-open areas with trees.
The great suindara is a large bird, with 60 cm of length and a wingspan of 137
cm. The females are larger and heavier than the males. The large dark eyes are
set in a round large facial disk, which is dark gray-silver and has a heavy
black edge. The upperparts are dark greyish-brown, with white part of spots
prominent. The underparts are whitish to light buff with little speckling.
This species is a powerful species of owl, able to attack and kill large prey
far beyond the capacities of its ancestors. It preys principally on a wide array
of mammals, but it'll also eat invertebrates and birds. Being nocturnal, these
animals hunt during the night. They eat smaller owls, caprimulgids, bats, cubs
of small deermaras and artiodactyls. Hunting mainly consists of the owl watching
from a perch for prey activity and then swooping down swiftly once prey is spotted.
The prey is often killed quickly by the Great Suindara's powerful grip and talons
though is sometimes bitten on the head to be killed as well. Then the prey item
is swallowed whole or torn into pieces with the bill. Exceptionally large prey
is consumed on the ground where it is killed, possibly being torn apart into
manageable pieces.
The Great Suindara are usually monogamous, sticking to one partner for life
unless one of the pair dies. During the non-breeding season they may roost separately,
but as the breeding season approaches they return to their established nesting
site, showing considerable site fidelity. The nesting season happens during
the dry period. Once a pair-bond has been formed, the male will make short flights
at dusk around the nesting and roosting sites and then longer circuits to establish
a home range. When he is later joined by the female, there is much chasing,
turning and twisting in flight, and frequent screeches, the male's being high-pitched
and tremulous and the female's lower and harsher. At later stages of courtship,
the male emerges at dusk, climbs high into the sky and then swoops back to the
vicinity of the female at speed. He then sets off to forage. The female meanwhile
sits in an eminent position and preens, returning to the nest after some minutes
before the male arrives with food for her. Such feeding behaviour of the female
by the male is common, helps build the pair-bond and increases the female's
fitness before egg-laying commences.
These birds are cavity nesters. They choose holes in trees and fissures in cliff
faces. No nesting material is used as such but, as the female sits incubating
the eggs, she draws in the dry furry material of which her regurgitated pellets
are composed, so that by the time the chicks are hatched, they are surrounded
by a carpet of shredded pellets. Before commencing laying, the female spends
most of the time near the nest and all her food is entirely provisioned by the
male. When the female has reached peak weight, the male provides a ritual presentation
of food and copulation occurs at the nest. The female lays eggs on alternate
days and the clutch size averages about five eggs, but can reach nine. The eggs
are chalky white and incubation begins as soon as the first egg is laid. The
incubation period is about 30 days, hatching takes place over a prolonged period
and the youngest chick may be several weeks younger than its oldest sibling.
Not all chicks survive, with the weakest being devoured by their siblings. They
fully fledge with ten weeks, but remain dependent of the parents until they
have 15 weeks. These birds reach sexual maturity with 2 years and their lifespan
is of 27 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Utburd,
fan owl (Gravistrix islandicus)
Order: Owls (Strigiformes)
Family: Typical owls (Strigidae)
Habitat: Iceland, various landscapes: mountain woods, bushes, grasslands.
Till the ice age Iceland appeared completely buried under glacial cover. As
the result, all terrestrial flora and fauna were destroyed, except for tiny
“oases” near muzzles of active volcanoes. During the thawing of glacier whole
soil layer was washed off to the ocean, and in early Neocene the vegetative
cover of island was presented by poor grassy plants, mosses and lichens. The
ground fauna of islands was also destroyed, except for some invertebrate species.
Iceland never had the connection to continents, therefore in Neocene fauna of
this island is presented only by descendants of flying animals: insects, birds
and bats. Due to it Iceland can be named as “Atlantic New Zealand”. Birds became
main herbivores and predators of this island.
Main predator of the island is large running species of owls – fan owl, or utburd,
the descendant of one small European species of owls. Life in isolation and
out of competition had transformed it to amazing and frightening bird similar
to flightless owls of Mexican plateau.
Utburd weighs about 50 kg, and is not able to fly at such significant weight.
Wings at bird are advanced rather well, but the bird uses them for other purposes.
Wings help bird to support balance during the run, and at males serve for courtship
display. The feathering of fan owl changes colouring depending on season: among
birds such feature was characteristic earlier only for white partridge. Since
spring up to late autumn the feathering of utburd has color standard for owls:
grey top of body with longitudinal strokes on feathers, and brown bottom with
white spots on tips of all feathers. Secondary feathers at males of utburd are
colored bright – they are red-brown with black tips bordered by white strip.
In courtship season male opens wings like fans (hence the name) and shows them
to female, having bent head down to the ground.
Winter feathering of utburd is almost completely white; only separate feathers
on stomach are black with white tips. Due to such colouring bird has an opportunity
to hunt on snow successfully.
Face disk of bird is wide; feathers on forehead are extended and bent back;
they are especially advanced at females. Feathers on face disk of bird are white
with black border, especially wide at the female. The crest is used by female
during the courtship: raising crest, owl can “increase” the visual size of head.
Fan owl is able to run quickly. Legs at bird are strong and high, with rather
short toes. In winter on legs “stockings” of feathers develop, assisting to
not fail in snow. Chasing catch, this bird easily accelerates momentum up to
50 kms per hour. Common flying owls attack prey by claws, but because of specialization
to running fan owl has changed the hunting habits. This bird attacks prey by
beak, which is rather large at this bird.
Utburd is ecological analogue of wolf in ecosystems of Iceland. It hunts various
ground birds flying to the island in summer (hence the name: in Scandinavian
mythology utburd is murderer phantom looking like huge owl). In winter this
bird hunts even tiny catch and often migrates to ocean coast and gathers carrion.
At coast of Iceland various sea birds – ducks and auks – constantly live. Also
at the coast of Iceland feathered giants, gannetwhales
have a rest and nest. Utburd occasionally attacks these birds, choosing weak
and ill individuals which keep at the edge of colony. In summer this predator
searches in colonies of gannetwhales for dead nestlings, preferring midnight
hours when the sun stands low above horizon. When there some of these birds
gather, they become more courageous, and even attack young and old gannetwhales
having a rest at the coast. In winter among these birds cases of cannibalism
are typical.
In summer fan owl usually hunts from ambush, and does not chase prey for a long
time. But in winter fan owls search for food by various ways, including digging
out snow in searches of spending the night birds. In hunting the owl uses keen
hearing which allows it to hear even breath of bird hidden in snow. But sight
of these birds is also very good.
Fan owls are territorial birds forming constant families. Pair is kept till
all life; birds hunt and rear posterity in common. At this species the sexual
dimorphism is well expressed: male is almost twice lighter in weight than female.
In connection with this feature it became cautious. In behaviour of the male
there is a set of poses showing peace intentions and extinguishing aggression
of the female. Courtship dance of the male is complex and fantastical, similar
to waltz; it includes various movements with wings opened like fans. Male turns
round before the female, frequently “bowing” and stirring wings. At this time
the female shows the superiority, extending up and opening crest on head like
a fan. It constantly turns to male sideways or back, forcing it to interrupt
demonstration and to run in front of her. At the culmination moment the female
joins “bows” of the male, lies down on the ground and presses crest to head.
At this moment the pairing takes place, and after it courtship ritual is finished
this day. Demonstrations repeat till some days in succession, and are finished,
when the female ceases to show interest to courtship. The courtship season begins
right after snow thawing when owls finally change winter feathering to summer
one.
Fan owl nests on the ground in shelters. The nest represents small hole in the
ground among bushes. In mountain areas these birds nest in rocky niches, if
there is an opportunity to climb in them from the ground. The litter in nest
is very poor, and sometimes is simply absent. In clutch there are only two eggs
which are laid by the female with an interval of 2 – 3 days. It begins hatching
from the first eggs (it is the common feature of owls), and nestlings hatch
not simultaneously. Nestlings of utburd are blind and helpless, covered with
rich down. They develop slowly enough: eyes open at the second week of life,
and feathers start to grow at monthly age. In same time young birds start to
rise on legs and try to walk. Bi-monthly nestlings leave nest and hide in forest,
and parents feed them with food brought in craw. The young growth lives in common
with adult birds within the first winter, but to the beginning of new courtship
season adult birds banish young ones from the territory. The most part of young
birds perishes in first months of independent life, more often in skirmishes
with adult individuals.
Sexual maturity comes at the age of 3 years, and life expectancy may exceed
40 years.
The name to this species of birds was given by Simon, the participant of forum.
Forest scops owl (Neoscops sylvestris)
Order: Owls (Strigiformes)
Family: Typical owls (Strigidae)
Habitat: North America, forests of temperate climatic zone.
Being a separate group of birds, owls existed on the Earth long before occurrence
of human ancestors. They had successfully gone through anthropogenous pressure,
and also through the subsequent climatic changes. Only local endemic and the
largest species of these birds had disappeared. At the territory of North America
the ecological niche of eagle owl was occupied by even larger valkyrie
owl,
and in northern areas of continent the place of polar owl its direct descendants
had partly inherited, sharing it with descendants of marsh or short-eared owl
(Asio flammeus). To the south, in the area of temperate climate with the expressed
seasonal prevalence, descendants of Western screech owl (Otus (Megascops) kennicottii)
live. One of such descendants, forest scops owl (Neoscops silvestris) is one
of the most widespread owls in North America of Neocene epoch.
In comparison with its ancestor forest scops owl seems the true giant – its
growth is up to 50 centimeters at wingspan of up to one and a half meters.
When this owl opens the wings and fluffs feathers in threatening pose, accompanying
its display with shrill call, few predators will risk attacking an adult owl
of this species. Nevertheless, the size is mostly illusory also impression
of large bird is made due to friable and loosely feathering. As well as its
close relatives, forest scops owl has rather long legs and neck. Beak is also
lengthened enough in comparison with other bones of skull, and in its shape
it resembles the isosceles triangle inverted by sharp top downwards. Cut of
mouth is very wide; such jaw structure helps forest scops owl to swallow entirely
its basic food – small and medium-sized ground rodents and other animals; sometimes
it eats nestlings of other ground birds or attacks large animals like young
deer. As well as at other owls, at forest scops owl cases of cannibalism among
nestlings, and occasionally among adult individuals are often.
Forest scops owl leads mainly ground habit of life, and even nests on the ground
– in holes, in wood hollows and ravines, in bush thickets. Birds having no
their own nests, and also males (only females hatch eggs) usually spend the
day on the old stubs, tumbled down trees, or on thick branches near to the
ground as these owls fly rather badly and reluctantly. At this time they usually
close eyes, extend body vertically, lift the feather horns upwards, and in
such position resemble any continuation of trunk or tree branch due to dark
grey-brown plumage with indistinct marble pattern imitating a structure of
tree bark. Many small forest songbirds, certainly, can distinguish a hidden
predator in this simple camouflage, but as forest scops owl hunts them less
often, than other owls do, they usually ignore it, especially if the owl really
sleeps, but not watches for prey.
But at night forest scops owl actively searches for prey, running in forest
on strong legs, and exchange shrill calls with its breeding partner. Almost
exclusively nocturnal habit of life helps this owl to avoid meeting with large
ground predators like berl or coyote descendants preferring to hunt in day
time, and thus weakens a competition between them. This species has very good
night sight and large bright yellowish-green eyes similar to cat eyes.
Despite of large wings, forest scops owl flies reluctantly. It is a settled
species; it does not migrate to the south when there comes winter, and continues
hunting also in snowy season. At this time on its paws “snowshoes” of toe bordering
feathers grow, which permit forest scops owl to walk, almost not failing through
snow. In winter forest scops owl changes a diet and becomes not so fastidious
in food, as in summer. Hunting on snow, it is capable to attack even gallinaceous
birds frequently spending night on the ground in snowdrifts.
The courtship season of forest scops owl begins in February – March. At this
time males start to display, uttering shrill calls from sunset up to sunrise.
At this time they are malnourished and grow thin. It happens, if snowy weather
returns, the part of especially thin males loses an opportunity to hunt and
perishes. By the end of April when snow starts rough thawing, forest scops
owls arrange nests and lay 2 – 4 eggs. To the beginning of summer owlets hatch.
The down dress of young birds is brown with irregular marble pattern. Owlets
have the ability similar to catalepsy and “virtual death” of other animals.
If the potential enemy appears near, they freeze on the spot, trying to escape
with the help of masking. If they are found out, nestlings fall on the ground,
make some convulsive movements and freeze, simulating death. At this time their
bodies turn soft and weakened, and eyes are only slightly opened. “Dying” nestlings
of forest scops owl let out a portion of unpleasantly smelling dung. When danger
passed, they perch again to the former place, waiting for parents with food.
Parents feed them till two months. For this time young birds gain weight and
develop, becoming similar to their parents. When young birds leave nest, the
family breaks up and adult birds also leave each other.
One-year-old birds are already capable to breeding. Life expectancies of forest
scops owl is 12 – 15 years.
Besides the forest scops owl, in North America related species of owls live:
Marsh scops owl (Neoscops palustris) – the smallest species of this genus,
no larger 35 – 40 centimeters in height. It is also the most brightly colored
owl of genus: it has bright rusty-brown background colouring with black strips
that helps it to hide in canes and other marsh vegetation. Despite of life
in damp habitats, this owl does not like to come into water and prefers to
hunt at the coast, eating large reptiles and amphibians, and also invertebrates.
Sometimes it ravages clutches of other marsh birds. It has stronger wings,
and for winter this owl migrates to the south along river valleys to sea coast
where spends cold season.
Water scops owl (Neoscops aquatica) resembles marsh scops owl in size, but
has monotonous reddish-brown plumage without marks. As against the relatives,
this owl nests on trees and prefers to look out for prey from height. In this
habit it resembles some predatory fishing birds lived in human epoch. Besides
it hunts in day time, instead of at night as forest scops owl behaves, and
uses sight for search of food actively. The basic food of this owl includes
river fishes, crayfishes and amphibians. In order to catch such specific prey
this owl has hooked claws, and on the bottom side of toes the set of corneous
acute spikes develops – it is the typical feature of fish-eating birds. Legs
of this owl are rather longer, than at related species. This owl inhabits swampy
forests of temperate climatic zone at the east of the continent. For winter
when the rivers freeze, it migrates for wintering to the sea coast and in lower
reaches of rivers similarly to marsh scops owl. These owls frequently wander
at the coast of ocean, eating fish and invertebrates cast ashore by storm.
These species of birds were discovered by Bhut, the forum member.
Swift-winged
owl (Otus apodiptera)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: Caucasian Peninsula, forests on mountain slopes and in valleys.
The boundary between Holocene and Neocene was marked by climatic changes and
the mass extinction of many animal species, especially of larger ones. Smaller
species mostly survived and evolved into new species, developing new ecological
niches, sometimes quite unusual ones. One example of this kind is swift-winged
owl, a descendant of the Eurasian scops owl (Otus scops) of Holocene epoch.
Despite its name, swift-winged owl is a fairly large, crow-sized owl. Its appearance
is recognizable and characteristic of owls, including the face disk, as well
as feather horns, like other owls of Otus genus. The coloration is also characteristic
for owls – soft, gray or ochre-red (various color morphs are present) with a
variegated marble pattern imitating tree bark. However, in its proportions this
bird resembles swifts or swallows rather than other owls – bird has long narrow
wings, which make this owl a very effective flier. The bird’s legs are long
and agile, with tenacious toes and sharp claws.
Swift-winged owl is a migratory species: it is so due to the specifics of the
diet of this species. As soon as autumn comes in the Caucasus Mountains, these
owls leave their habitats and fly south to North Africa and the mountain valleys
of Persian Ridge, where they winter. Owls usually migrate at night to avoid
encounters with other birds that may attack the owl or give away its location
to predators with loud cries.
This owl is specialized for hunting chiropterans, and its fast flight allows
it to hunt even the fastest bats. Soft plumage suppresses the echolocation signals
of the animals, and they do not notice the approach of the owl until the last
moment. During the night, the bird eats up to five small bats or 2-3 large ones.
It swallows small prey whole, at large prey it pre-tears off and throws out
wings with long fragile bones. Less often, swift-winged owl catches large insects,
small birds and other arboreal vertebrates. It usually hunts at dusk, in the
morning and in the evening, resting in the middle of the night, but on clear
and moonlit nights it hunts without interruption. During the day, the bird hides
in the crowns of trees and sleeps.
When spring comes, swift-winged owls return to Caucasus, where new pairs form
in the mountain forests. At the same time, bats wake up, and birds begin to
hunt and eat off actively after migration. Males ready for nesting find suitable
hollows or abandoned nests of other birds, and call females with shrill cries.
If the female likes the male and the nest, it stays and they raise offspring
together.
Swift-winged owls protect the territory around the nest from relatives, and
declare their rights to the territory with rather loud screeches at dusk and
at night. There are usually 4-5 white eggs in the clutch; they are incubated
by both parents in turn. When the chicks hatch, the parents quickly begin to
leave them alone, actively supplying the brood with food. After two weeks, the
juvenile down is replaced by feathers, by one and a half months old they can
fly and learn to hunt, and in the fall they fly south along with their parents.
At the same time, adult couples break up.
The life span of swift-winged owl is no more than 10 years, but usually these
birds die much earlier, in wintering areas or during migrations.
This bird species was discovered by Bhut, the forum member.
Grey-faced
pygmy owl (Glaucidium griseorostrus)
Order: Owls (Strigiformes)
Family: Typical owls (Strigidae)
Habitat: savannas, plains and semi-open areas of South America.

Picture by Pavel Volkov
Even with the impacts caused by human activity during the
Holocene, some species persisted. Owls of the Genus Glaucidium survived and
in the Neocene a new species appeared, the grey-faced pygmy owl.
The grey-faced pygmy owl is small, with the females being slightly larger than
the males, with 17 cm against their 15 cm of length. The head is rounded, with
no ear tufts. As their name says, their facial disk is dark grey. The upper
part of the body is relatively dark auburn with small white spots. Wings are
light reddish-brown and relatively short. The tail is reddish-brown with dark
bars. The lower part of the body is white, with dark brown stripes. The throat
shows a rounded whitish area. The legs are feathered, with bristled and yellowish
toes with dark claws. The beak is yellow.
This species is crepuscular, but often hunts by day. It hunts a variety of birds,
lizards, mammals, and insects. They are able to catch birds in flight. The flight
is low to the ground and rapid with long swoops. Small birds will mob at it
while it is perched in a tree. Grey-faced pygmy owls can kill preys larger than
them.
The grey-faced pygmy owl nests in tree holes and hollowed-out termite nests,
however the individuals that live in the areas of occurrence of azure
parakeets (Sociopsitta cyanea) will make their nests almost exclusively
in the nest of these parakeets. Adults defend the nesting site with great aggression.
The birds nesting together with the azure parakeets will defend not only their
nests but he entire colony of parakeets. The pair will attack birds of prey,
other owls, snakes and mammals. The owls will also attack and consume striped
anguja (Anguja striatus), a species of predatory rodent that usually feeds
on eggs and chicks.This way both birds are benefited, the owls with a nest and
the parakeets with protection of their eggs and young. This rodent is bigger
than the owls, to kill it the birds attack constantly the rodent, forcing it
to fall from a branch or to blood until die.
This species is serially monogamous, forming bonded pairs for one or more breeding
seasons. The male is territorial and may use the same nesting territory for
up to seven years. The female lays about four to seven eggs in late winter to
early spring. They are incubated for four weeks, starting when the third egg
is laid. They hatch nearly simultaneously and the female remains with them for
nine to ten days, being fed by the male. After three weeks the young are active
and the female returns to the nest only to feed them and clean out waste. Fledging
occurs at 30 to 34 days. The chicks remain close to the nest for a few days
before departing. The lifespan of the grey-faced pygmy owl is of 16 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Rust-feathered
owl (Neoglaucus ferrugineus)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: southwest of North America, north of Central America.
The transition between Holocene and Neocene was marked by significant changes
in the climate and geography of the Earth. South America separated from the
North America again, and the latter connected with Eurasia via Beringia, as
a result of which vast areas of high young mountains were formed in the west
of the North American continent. The peaks of Rocky Mountains in Neocene epoch
are covered with glaciers even in summer, and even in the southern part of this
mountain range.
The new habitats were populated by migrants of different origins. Most often,
these are descendants of species from the north retreated to the south because
of the glacier, or from the west – descendants of Asian species who managed
to cross mountainous areas and get to a new continent. But the ancestors of
some species came to this corner of the planet from the south: one of them was
the ancestor of rust-feathered owl, which descends from one of the species of
Central American pygmy owls of the widespread genus Glaucidium. This bird got
its name for the characteristic rusty-brown background color of its plumage;
in the coloration of this species, there are also brighter white spots and stripes,
which give rust-feathered owl an appearance more characteristic for tropical
species. These light markings are especially noticeable when the bird is in
flight. There are no feather horns on the head. A characteristic feature in
the coloring of the bird is also the wide black “eyebrows” – bands of black
feathers over the eyes, converging over the beak. The facial disc is weakly
pronounced. Unlike the ancestors, this is a fairly large species - up to 50-60
cm long with a wingspan of about one and a half meters.
The voice of this species is a long, shrill whistle, different from the voices
of other local owl species. Rust-feathered owl is aggressive towards other species
of owls, especially large ones, and destroys their clutches and kills chicks.
Due to its aggressive disposition, this owl successfully survives when living
next to predatory mammals. The adult rust-feathered owl has virtually no enemies,
but this species often suffers from parasites got through the prey. This species
hunts various mammals at night, sometimes attacking sleeping birds. This species
has a characteristic behavioral feature: rust-feathered owl bites off the prey’s
head and swallows the body without it. Therefore, in the favorite places of
stay of this bird on the ground under the roost, piles of skulls and heads of
prey at various stages of decomposition gradually accumulate.
Despite the fact that this owl is a fairly large bird, it has a light build,
and its wings and tail are adapted for fast, maneuverable flight between trees
in pursuit of animals. It catches prey on the ground, but it often grabs birds
and bats with one foot in the air or from branches. During the day, rust-feathered
owl sleeps, usually hiding somewhere in a secluded place – among the tree foliage,
less often in a mountain cave. At this time, the coloring of the bird provides
it with an excellent disguise.
Rust-feathered owl is a non-migrating species, but the connection with the territory
is rather weak: if necessary, the birds easily travel long distances, moving
to places rich in food.
This species is monogamous. The courtship season begins in early spring, at
the end of the dry or beginning of the rain season. Males begin courtship games:
the displaying male actively calls the female, flying next to the tree in which
the chosen hollow is located, and screams and whistles shrilly. The female is
guided mainly by the quality of the hollow chosen by the male. This bird does
not form permanent pairs; after the end of nesting, the pairs break up. Sometimes
rusty-feathered owls nest not in hollows, but in mountain caves and crevices
between large boulders.
In the clutch of this species there are 1-4 white eggs. Rust-feathered owl,
like other owls, does not build its own nests, laying eggs directly in the natural
litter. Hatching begins with the first egg, so the older owlets turn out to
be much more developed and larger than the younger ones, and they may well eat
them if the year is poor. Due to cannibalism, usually only 1, rarely 2, offspring
survive to sexual maturity. The plumage of young birds is duller than that of
adults and lacks white markings. Sexual maturity begins at the age of 2 years.
The average life expectancy of rust-feathered owl is 8-12 years.
This bird species was discovered by Bhut, the forum member.
Flute
owl (Philomelostrix xenowekau)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: New Zealand, forests of various types.
The largest owl of New Zealand of the human era was the laughing owl, or whekau
(Sceloglaux albifacies). But by the beginning of the XX century, this species
became extinct, unable to withstand the pressure of civilization and introduced
species. In Neocene, a new large owl species appeared in the ecosystems of the
archipelago, descending from the human-introduced little owl (Athene noctua).
This bird is called flute owl. The body length of the flute owl reaches 70 cm,
and weight is up to 1800 g. The wingspan of this bird can reach 150 cm. Males
are slightly smaller and slimmer than females. Flute owl is distinguished from
most owls by its long and strong legs, feathered to the toes.
The coloration of these birds is rather inconspicuous. The underside of the
body is light gray with dark brown mottling. Back, wings and back of the head
are chestnut in color with a white scaly pattern. Tail is striped, of chestnut
color with white cross-stripes. Face is gray, bounded at the edges by thin black
stripe. Eyes are bright yellow; there are no “horns”. Beak is pale yellow, legs
are gray.
Flute owls got their name for the mating song of a male, which sounds as a simple
musical phrase repeated many times and vaguely resembling the singing of a Eurasian
golden oriole. Females often sing with males antiphonally. In general, these
owls are very “talkative” and make a variety of sounds that serve as alarm signals,
as well as for communication during the nesting period.
These birds, like most owls, are nocturnal, with the greatest activity occurring
in the twilight hours after sunset and before dawn. During the day, these owls
sleep or perch, hiding among the branches.
The food of flute owls includes various small and medium-sized vertebrates,
primarily rabbits, rodents,
ground birds, lizards and amphibians, as well as large insects and snails. Usually
these predators ambush prey, perching on a roost site, and attack it from above.
Also, flute owls often chase prey on the ground, developing a fairly high speed
and deftly overcoming obstacles. Sometimes male and female hunt in pair and
catch this way a larger prey than singly. When hunting, these owls rely equally
on sight and hearing. Silent flight is also a great help.
Flute owls are monogamous birds: a pair occupies the same hunting territory
all year round. However, outside of the breeding season, the male and female
stay in different parts of it. The reunion of the mates takes place at the end
of winter (in the Southern hemisphere it is August). At the same time, lonely
birds search for mates for themselves.
Flute owls nest in hollows located low above the ground, or on the ground: under
the roots of trees, in abandoned burrows of mammals, etc. Stems of herbaceous
plants are used as a litter. There are usually three to five eggs in the clutch,
which are laid one after the other, and the female begins to incubate only from
the moment the last egg is laid. The male feeds and protects her. After 30 days,
helpless down-covered chicks hatch. Both parents are engaged in feeding them.
Flute owls guard the nest very jealously; they bravely attack any animals that
happen to be near the chicks. However, if the parents are not around, the chicks
can fend for themselves: in case of danger, they make loud buzzing sounds resembling
the buzzing of local wasps (similarly, the chicks of Holocene burrowing owls
imitated the rattle of rattlesnakes). The survival rate of chicks depends on
the number of prey animals and the hunting skills of the parents. If there is
not enough food, the stronger chicks kill and eat the underdeveloped ones.
As young birds grow up, they gradually leave the nest, and begin to fly when
they reach the age of one and a half months. In the fall, they leave their parents’
territory. Already in the first year of life, young birds begin to reproduce,
and the life span of flute owls can reach more than 15 years.
Due to their size and strength, adult flute owls have almost no enemies – only
occasionally they can be attacked by large predatory mammals
and eagle keas acting as a group. However,
in the daytime, these birds, like all owls, may be mobbed by small birds.
This bird species was discovered by Simon, the forum member.
New
Zealand Ground Owl (Antipathene cursor)
Order: Owls (Strigiformes)
Family: True Owls (Strigidae)
Habitat: North and South Islands of New Zealand, open country, grassland, meadows,
scrubland, bush, open forest.
During man’s occupation of New Zealand during the Holocene, the Little Owl (Athene
noctua) was introduced to the archipelago in order to control plagues of sparrows
and other crop-eating birds. Since then it became very successful, leaving descendants
in the Neocene.
This mid-sized owl resembles its ancestor somewhat, but even more its relative
the Burrowing Owl (Athene cunicularia), with the exception of not settling in
the burrows of other animals. Reaching a length of 35 centimeters and a wingspan
of up to 70 centimeters, it has a compact body, with a small facial disk; coloration
is dark greyish brown, spotted all over with yellowish white, also having white
eyebrows and “bib” above the chest that is bordered with darker brown. Living
in open country, they are regularly found on the ground, having long slender
legs ending in compact feet with large claws, stance is upright and alert. There
is little or no sexual dimorphism.
These birds are often active during the day, but do most of their hunting between
dusk and dawn; they will stalk over the ground, seeking out higher spots from
which to spot prey. Diet includes small vertebrates such as frogs, rodents and
lizards, as well as large insects, slugs and earthworms. Usually they swoop
down on prey from their perch, though they may occasionally chase prey on foot.
They are territorial, and a male maintains a territory the boundaries of which
may change during its lifetime. Call is a high-pitched, repetitive hooting,
or a throaty hiss when alarmed.
Breeding occurs in spring, from August to November, birds are generally monogamous,
but may sometimes have a different mate from year to year. These birds seek
out a large patch of vegetative cover, tree stump or rocky outcrop, near which
to make their nest. Nests consist of a shallow scrape in the ground, in lee
of a piece of cover, and obscured by a cairn of gathered tufts of grass and
other vegetation, materials for the nest include grass and shed feathers. The
female will make a clutch of up to 6 eggs, laying one egg every few days over
a protracted period of time, she then broods the eggs for up to a month, with
the male bringing food for her to eat. Both parents feed the young, chicks become
active and able to make short flights after about 4 weeks, but may still be
fed by their parents during this time. Lifespan is up to 10 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
“Dead
head” owl (Athenops caput-mortuum)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: mixed and broadleaf forests of Europe - from the forests of the Atlantic
coast to the east to the Ural Mountains.
In the human epoch, the advantage in the struggle for existence was gained by
small synanthropic species, which, thanks to people, managed to maintain a high
number and a fairly wide range. One of these species was the little owl (Athene
noctua), which gave a variety of descendants in Neocene, including in
new habitats, where it was introduced by people. A descendant of the little
owl in the forests of Europe is “dead head” owl, a widespread owl species.
The appearance of “dead head” owl is very characteristic and easily recognizable:
the front part of the head around the eyes and beak is covered with white feathers,
which makes the white area slightly resemble the stylized outlines of the human
skull – hence the name of the species. The crown and the back of the bird’s
head are black; these areas of plumage shade the “mask”, making it more contrasting.
The background color of the plumage is dark brown, primary feathers are reddish.
Sexual dimorphism is weakly expressed, but the female is about 20% heavier than
the male and is also distinguished by a dark cross stripe on the wings, which
appears with the onset of maturity. The adult male reaches 25-30 cm in length
and 55-60 cm in wingspan, the female is proportionally larger.
This owl species preys on rodents, bats, small birds; sometimes in summer these
owls eat insects. In the northern part of the range, with the onset of winter,
this bird hides part of its prey in hollows – this stock helps out in case of
unsuccessful hunting. In hunting, these birds rely on very keen hearing and
almost silent flight. The hunting agility of this owl allows it to grab a bat
in flight or from the ceiling of the cave, quickly turning over in flight with
its belly up.
“Dead head” owl lives in hollows; it can occupy an abandoned squirrel nest,
if it is not badly destroyed. In mountainous areas, birds arrange nests in crevices
between rocks, but only as a last resort if they do not find a suitable place
for a nest. When arranging the nest, the birds confine themselves to laying
fresh litter only, and sometimes they simply rake the wood dust into a pile
on which they lay eggs. These owls are strictly nocturnal, fly out from the
shelter for feeding at late dusk, when many daytime birds living in the same
territory are already settling down for the night. Before the flight, these
owls spend a few minutes near the hollow, preen feathers with a beak and spread
their wings, flex their muscles with frequent flaps of their wings. Going for
hunting, these owls utter a characteristic screech before takeoff, notifying
neighbors that this territory is occupied. The usual vocalization of “dead head”
owl is a ringing hoot, uttering in a series of 3-4 cries with a long break between
a series of signals.
Nesting usually takes place in March-April. During the breeding season, birds
are very active, behave excitedly, often fly even during the day and scream
for a long time. Courtship ritual includes mutual feeding, preening and pinching
with the beak. There are 3-6 eggs in the clutch, the incubation period is 28-33
days, and the feeding of the offspring continues for about 3 more weeks. Young
owls reach sexual maturity at 2 years old and live 15-17 years.
This bird species was discovered by Nick, the forum member.
Fishing
owlet (Postathene longipes)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: reed thickets along the shores of Fourseas.

Picture by Pavel Volkov
In Neocene, in epoch of the restoration of ecosystems, some
land birds passed to a semi-aquatic lifestyle – for example, fishing
parrots of Mauritius island and ethereal
kites of the tropical seas of the Earth. Another bird from this category
is fishing owlet, a small fish-eating inhabitant Fourseas, a descendant of the
little owl (Athene noctua).
This species is larger than its ancestor, but is still not among the largest
owl species: about 40 cm tall, with wingspan of up to 70 cm. This bird species
has a very recognizable appearance: a rounded barrel-shaped body with a facial
disk, like in other owl species (due to the contour feathers), and very long
legs. The bird spends a lot of time on the ground and walks well on marshy soil:
there are skin membranes between the toes, reaching to the middle of toes. The
length of the claws reaches 6 cm; the tail is 13 cm long. In the color of plumage,
this owl is more like a bittern – the background color of the back and sides
includes many longitudinal yellow and brown stripes, masking the bird among
the vegetation. Face disc is beige. Legs are orange, beak and eyes are yellow.
Fishing owlets inhabit reed thickets along the shores of Fourseas. Unlike most
other owls, this species is active during the day and at dusk, and often lives
in colonies of larger waterfowl. During the day, fishing owlet forages for food.
It can walk along the banks of the channels or directly along the bottom of
shallow reservoirs, as well as fly over the channels and look for prey. The
diet consists of aquatic invertebrates, small fish, frogs and newts. It prefers
to snatch these creatures out of water with its claws and brings by paw to its
beak, tearing them to pieces. In case of hunger, fishing owlet can kill animals
the size of itself – for example, medium-sized fish. In summer, these owls can
find a drying duct and literally empty it, not leaving this place until they
eat all the animals got into this trap. Wandering in colonies of semiaquatic
birds (thalassocoraxes, herons), this owl looks for prey accidentally dropped
by birds and eats it. The enemies of this owl are terns and large predatory
fishes that can drown this bird.
Fishing owl is a migratory bird; for wintering it flies to the shores of Indian
Ocean, including to the north of Zinj Land. During the migrations, these birds
stick to the riverbeds and try not to fly high into the mountains.
In the spring, the owls returned from wintering form breeding pairs. They choose
colonies of shorebirds for nesting and occupy old bird nests or build their
own ones. The nest of this species is a loose building half a meter in diameter.
The female lays up to 3 eggs and hatches them for 5 weeks while the male brings
food to her. Chicks are covered with thick brown down with irregular white spots.
While the parents are away, the chicks huddle together and close their eyes,
not giving away their presence. During the feeding of chicks, adult birds may
not even fly far from the nest – they gather fish dropped by thalassocorax chicks.
Acute hearing helps the bird to determine where the fish has fallen. At the
age of 6 weeks, the chicks are already fully fledged and begin to learn to fly.
Sexual maturity in young birds occurs at the age of 2 years, life expectancy
is up to 40 years.
This bird species was discovered by Mamont, the forum member.
Ruru
e Loka (Notoninox agilis)
Order: Owls (Strigiformes)
Family: True Owls (Strigidae)
Habitat: Forested areas across the North and South Island of New Zealand, avoids
overly cold mountainous regions, and forests of stunted growth.
The hawk-owls known as Mopoke, Boobook or Morepork, (genus Ninox), were common
across Australia and New Zealand in the age of man. They were known to live
peacefully alongside mankind, roosting in parks and gardens and commonly hunting
rodent vermin by night. During the start of the Neocene, these birds successfully
spectated into new forms including very large ones.
In the forests of New Zealand, descendants of the Ruru or Morepork (Ninox novaeseelandiae),
whose ancestors traditionally fed on lizards and large insects, now actively
predate on the many kinds of introduced small mammal, as well as native kinds
of bat originating in Australia.
This bird is somewhat larger and more powerful than its ancestor, reaching up
to 38 centimetres long, and having a wingspan of 70 centimetres, weighing as
much as 550 grams, females are a little larger than males. It appears slightly
more like a typical owl than its ancestors the hawk-owls, the face has a distinct
“mask” of feathers that accompany finely adapted directional hearing, in order
to track the movement of rodents. Coloration is a middle tone of reddish brown,
checked with long streaks of darker and lighter brown, face and underparts are
greyish white. Primaries are cross-banded pale brown and dark brown. Wings are
somewhat short and broad, rounded in shape, adapted to fly quickly and manoeuvre
between trees in order to seize prey, it is even able to catch bats in flight.
Feet are feathered, and a little larger than in its ancestor, grip is very powerful,
talons are large and curved. Eyes are a bright yellow, and the beak is pale
grey. Call is a little more melodious than its ancestor, but is still a two-note
“Rhuu-rhuu”. The agility of this bird in flight has given it the Maori name
meaning “acrobat”.
They may live singly or in pairs, sometimes pairs may be seen with their adult
young. These owls are nocturnal, and roost upon secluded perches or in tree
holes by day. They are most active hunting in early evening and early morning,
continuing sporadically through the night, if the weather is bad or overcast,
they may hunt by day. Their diet mostly consists of small mammals such as bats,
possums and rodents, small birds and lizards are occasionally taken. Prey may
be caught on the wing after a short pursuit, this is especially how it catches
bats, though it will sometimes pounce from a high perch to seize prey on the
ground. Individuals are territorial, and males will call back and forth in a
“duel” by night to proclaim territory.
Breeding occurs between September and November, the female will lay up to three
eggs in a tree-hole, sheltered ledge or hollow log. Eggs are incubated alone
by the mother for up to 30 days, during which she is fed by the male. The owl
chicks fledge after about 7 weeks, being fed by both mother and father. Lifespan
is up to 30 years, but usually somewhat shorter, both adults and young may be
preyed upon by carnivorous marsupials
or cats, large rats may ruin their nests, adults in flight by day may be preyed
upon by large hawks.
This bird species was discovered by Timothy Morris, Adelaide, Australia.
Andean
lesser groundowl (Parvodeinostrix andina)
Order: Owls (Strigiformes)
Family: True owls (Strigidae)
Habitat: South America, Andean Plateau.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The genus Parvodeinostrix, the lesser groundowls, is
formed by descendants of the burrowing owl (Athene cunicularia). It's the sister
genus of the Deinostrix, large
owls that inhabit North America. Like their Northern relatives, these birds
have a long knife-like claw on their internal forward toe. One of its representatives
is the Andean lesser groundowl, a species that inhabits the Andean Plateau.
The Andean groundowl have 32 cm of length and 75 cm of wingspan. Adults have
chestnut brown heads and wings with white spotting. Their chests and abdomens
are cream white with variable black spotting or barring, also depending on the
subspecies. Juvenile owls are similar in appearance, but they lack most of the
white spotting above and brown barring below. The juveniles have a buff bar
across their upper wings and their breasts may be buff-colored rather than white.
Andean groundowls of all ages have long grayish legs. Their plumage is dense,
what helps them to stay warm during the winter of the Plateau; they are non-migrating
birds.
When hunting, they wait on a perch until they spot prey. The perch usually is
a rock, cactus or bush. Once they spot it, they swoop down on prey or fly up
to catch insects in flight. Sometimes, they chase prey on foot across the ground.
In these cases, they will kill the prey with a hit of their long claw. The highly
variable diet includes invertebrates and small vertebrates. These owls mainly
eat large insects and small rodents. Although they often live close to social
guinea pigs, they rarely prey upon them.
The nesting season happens during the spring. The lesser groundowls usually
only have one mate but occasionally a male will have two mates. The owls nest
in a burrow, usually these left empty by social guinea pigs. If burrows are
unavailable and the soil is not hard or rocky, the owls may excavate their own.
During the nesting season, they will collect a wide variety of materials to
line their nest, some of which are left around the entrance to the burrow. The
most common material is mammal dung, which helps to control the microclimate
inside the burrow and to attract insects, which the owls may eat. The female
lays an egg every one or two days until she has completed a clutch, which can
consist of four to 12 eggs. She then incubates the eggs for 4 weeks while the
male brings her food. After the eggs hatch, both parents feed the chicks. Four
weeks after hatching, the chicks can make short flights and begin leaving the
nest burrow. The parents still help feed the chicks for 3 months. They reach
sexual maturity with 7 months and have a lifespan of 9 years.
Another representative of the Genus is the Savanna
lesser groundowl (Parvodeinostrix savannae). This species inhabits
several biomes of South America, from the savannas of Central South America
to the plains of Patagonia. They have 28 cm of length and 70 cm of wingspan.
The upperparts are grey-brown, heavily spotted with white. The underparts are
white, streaked with brown. The facial disc is pale and the iris is yellow.
There is a white neckband and supercilium. The Patagonian population is migratory,
moving north during the winter.
This bird species was discovered by João Vitor Coutinho, Brazil.
Karnabo
(Asio karnabo)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: southern Europe – between the Alps and the Mediterranean lowland.

Picture by Pavel Volkov
The transitional epoch between Holocene and Neocene was marked
by climatic changes and the mass extinction of many animal species, especially
of large and vulnerable species. Smaller species mostly survived, evolved and
became the progenitors of a number of new species, including those having a
lifestyle atypical for an ancestor. Karnabo, a descendant of Holocene short-eared
owl (Asio flammeus), is one such species.
Karnabo is a rather small owl about 30 cm long with a wingspan of about 60 cm
and a weight of 150-250 grams (the female is larger). Plumage is colored inconspicuously,
but contrastingly: head and wings are mainly gray with thin black cross ripples.
Its chest and abdomen are white with gray ripples; the round face disk is outlined
by a narrow black stripe along the edge. There are no feather “horns”. Karnabo
lives along the entire southern slope of the mountain ranges of Europe to the
north of the Mediterranean lowland; the range is strongly elongated from west
to east, so local variations in the color of the plumage are very common – gray
color can be replaced by beige or ochre-red. Legs are long and strong – karnabo
spends more time on the ground than in trees, and runs well. The bird can fly
well, but prefers to stay on the ground in thickets of bushes, less often on
trees or in the burrows of small animals. The bird often chases wounded prey
by running.
Karnabo has two peaks of daily activity – day and night ones; in the morning
and evening, this bird prefers to sleep in a shelter. It hunts mainly small
vertebrates, less often daily active insects. In spring and autumn, during the
migration of birds, a significant part of the diet of this species consists
of small birds. Cannibalism is characteristic for this species, especially among
chicks. This owl does not like cold, and it is not found in Central, and even
more so in Northern Europe.
The breeding season of karnabo begins in the first half of spring, when rains
begin and seasonal vegetation develops abundantly. Pairs are formed for one
season. Males call females with shrill cries, but they introduce themselves
very carefully, because the female is larger and often behaves aggressively.
Within a few days, the birds get used to each other’s presence, after which
mating takes place. Karnabo has from 4 to 7 white eggs in the clutch. Karnabo
does not build its own nest, and usually occupies someone else’s one, or lives
in a hollow, or even in the burrow of some small mammal. Only the female incubates
the clutch for 17 days. The male does not take part in incubation, but brings
her food. Later, when the chicks hatch, they are fed by both parents. Owlets
grow quickly: in a month they fledge, at the age of 6 weeks they can fly, in
two months they are already independent, and the next year they can take part
in reproduction.
Karnabos usually don’t live more than 10 years.
This bird species was discovered by Bhut, the forum member.
Hawaiian
red owl (Pueotus rufus)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: forests of the Hawaiian Islands.

Picture by Pavel Volkov
At the beginning of Neocene, the avian colonization of Hawaiian
Islands continued. After the end of the human epoch, owls – descendants of the
long-eared owls (Asio otus) of American origin – arrived to the islands. In
competition with the descendants of human-introduced species, they gave rise
to several small forms, one of which is Hawaiian red owl.
It is a small bird: the total length of the bird is 20 centimeters with a wingspan
of about 30 cm; female is slightly larger than male, like in most birds of prey.
This is a bird of an elegant build: it has an elongated fan-shaped tail, as
well as rather long legs. The body is elongated; head is not very large, with
small tufts of feathers above the eyes. The background color of the plumage
is reddish-brown with a darker marble pattern on the wings, tail, back and nape.
On the chest and belly, the background color is lighter, tips of feathers are
white, and there are also light areas under the wings. Face disc is also light;
beak is grey and the iris is yellow. Tips of tail and wings are dark. Legs are
feathered almost to the toes; skin on toes is dark pink with gray claws. There
are numerous corneous bristles on the underside of toes.
This small predator lives in the lowland rainforests of Hawaiian Islands, but
a related form inhabits the mountain silver forests.
This bird is active at night and at dusk, often flies in the forest canopy and
at high altitude. This owl usually sleeps among foliage or in thickets of epiphytes
against the background of bark. Like all owls, this species searches for prey
mainly by ear and grabs it with its claws. The diet of Hawaiian red owl is diverse
– usually it includes large invertebrates (snails, crabs and insects), sometimes
tailless amphibians and lizards, often a variety of small birds and tree rats,
which the bird attacks, despite the fact that they are almost equal to the predator
in size. The owl itself can become a prey of larger birds of prey (like neopueo).
Hawaiian red owl is able to defend itself from the predatory erinia
bat by cutting its flying membrane with its claws.
Hawaiian red owl does not have a specific breeding season – in a tropical climate,
birds ready to breeding are found at any time of the year. A couple makes a
nest in the forest canopy, preferring an abandoned large nest of another bird
or a hollow rotten out in place of a broken tree branch. The same place can
be used for nesting by several pairs of these birds in succession. If suitable
nesting sites are present, the egg laying may repeat twice a year. There are
one or two blue eggs in the clutch; the clutch is incubating for up to 17 days.
Only the female hatches it, the male gets food for her. After the chick appears,
both parents can fly in search of food, especially when the juvenile grows up.
Chick feeding lasts up to 35 days. The young bird leaves the nest, and the parents
feed the fledgling for about one more week. Sexual maturity is at the age of
5 months, life expectancy is about 6-7 years.
This bird species was discovered by Mamont, the forum member.
Hawaiian
horned owl (Pueotus cornutus)
Order: Owls (Strigiformes)
Family: Typical owls (Strigdae)
Habitat: the canopy of the Hawaiian rainforest.
Among the endemics of Hawaii of Neocene epoch there are small owls, usually
insectivorous and having bright coloration. They descended from the descendants
of long-eared owl (Asio otus) settled at the islands from North America after
the end of the human era. They form a genus Pueotus (“pueo” is the owl name
in Hawaiian), uniting small species living in the tropical forest canopy. One
of their species is Hawaiian horned owl, a relative of Hawaiian red owl.
This species is a small bird the size of a starling, but with a slightly larger
head and beak, and with long legs. Because of this, as well as because of the
fluffy plumage, the bird seems larger than it actually is. It has not very long,
but wide wings that allow it to maneuver among the branches of the tropical
forest. The upper side of the body is red-brown with a pattern resembling tree
bark, lower side is beige or sandy yellow, face is white with a thin black cobweb
pattern, beak is gray, legs are feathered to the toes, yellow or occasionally
pinkish, equipped with large claws. On the upper side of the body there are
many fine spots on the plumage; on the white plumage of the face disk, a black
pattern is formed of thin black edges of the facial feathers. Above the eyes
there are two long pointed tufts of ochre-red feathers with white tips, for
which the bird got its name. Females are slightly larger and duller colored
than males.
The habitat of this small bird is the canopy of tropical rainforests. On its
broad wings, it flies among the trees at dusk and at night, looking for prey
and listening to the sounds of the forest. During the day, these owls gather
in groups and sleep in shelters on especially tall trees. They often hide among
epiphytic plants. Horned owls usually feed on insects, including large ones
– beetles, butterflies and orthopterans.
To do it, the bird uses a large beak and a strong bite, as well as powerful
claws that easily pierce insect shells. Hawaiian horned owl kills beetles by
sticking a claw into the articulation of the head with the chest. Part of the
diet consists of snails, including large ones, which shell the bird breaks easily.
Less often, this owl feeds on small birds, like honeybirds
– such prey sometimes appears equal in size to the predator itself. For Hawaiian
horned owl, erinia bats and larger
birds of prey are dangerous. When attacked by these predators, owl maneuvers
among the branches under the forest canopy, relying on its speed and small size.
There is no pronounced breeding season in this species. Like its ancestor, this
owl occupies the nests of various birds, without even making attempts to complete
or repair them. Sometimes birds lay eggs simply among epiphytic plants, and
even in the center of the rosette of an epiphytic fern. In the clutch, there
are 2-3 blue eggs without spots; during the year, the egg laying is repeated
2 times. Only the female incubates, and the male feeds her and sleeps near the
nest during the day. After 15 days, the chicks hatch. They are usually fed not
on insects, but on small birds and lizards, which their parents previously tear
into pieces and give their chicks with skin and bones. A little later, the chicks
switch to a diet of insects, after 1.5 months they begin to fly and soon leave
their parents and move on to independent life. Sexual maturity occurs at the
age of 5 months, life expectancy is up to 6 years.
This bird species was discovered by Mamont, the forum member.
Amazon
nhakurutukutu (Piscatostrix amazonica)
Order: Owls (Strigiformes)
Family: True owls (Strigidae)
Habitat: South America, tropical forests.
The human actions in the Holocene caused many impacts in the ecosystems. Because
of these actions, many species went extinct in the Neocene. However some species
survived and gave rise to descendants, one of these descendants is the amazon
nhakurutukutu, a type of species of the genus Piscatostrix. This animal is a
descendant of the owls in the family Strigidae, the true owls. The name nhakurutukutu
is the combination of two Tupi words, nhakurutu (which means owl) and kutu (which
means fishing). This reflects the habit of this animal to catch fish. It lives
in the Amazon rainforest.
These birds have 46 cm of length and a wingspan of 83 cm. The females are slightly
larger than the males. Their plumage is rufous brown and heavily streaked with
dark brown. The underparts are grey, with finer brown barring. The facial disk
is dark brown and their ear tuffs are white and long. The beak and legs are
dark grey.
Members of the genus Piscatostrix eats mostly fish, however they will also take
other aquatic animals such as frogs, freshwater crustaceans and large insects.
Small mammals and reptiles are also eaten, but more rarely.
Amazon nhakurutukutu nest during the dry season, which has the benefit of lower,
clearer water and thus more easily detectable fish. They are monogamous and
territorial, claiming a stretch of river or lakeshore for themselves. Territories
are claimed by hooting at the start of the breeding season. The nest is a natural
hollow or cavity in an old shady tree close to the water, quite often around
where thick branches emerge from the trunk. The clutch size is typically one
or two white eggs. The female lays the eggs when water's at its peak or starting
to fall, so that brood feeding coincides with low water and concentrated prey.
The female does all incubation for around 32 days, while the male feeds her.
If there is more than one egg, hatching occurs at 5 day intervals. The second
chick often disappears, probably due to starvation and fratricide. The young
fledge with 53 days and remain in their parent's territory for 6 to 9 months
after fledging and resemble the adult by around 10 months of age. At the first
sight of danger to their offspring, both parents may engage in distraction displays.
In these displays, the parents become slim and erect their ear tufts high, the
wings are stretched and they produce a sound similar to a scream. This species
reaches sexual maturity with 4 years and their lifespan is of 28 years.
Other species in the genus Piscatostrix:
Lesser
nhakurutukutu (Piscatostrix minor)
Living in the forests and woodlands at the foothill of the Andes, these birds
have 37 cm of length and a wingspan of 72 cm. Females are slightly larger than
the males. Their plumage is chocolate-brown with the facial disk being dark
brown. Their ear tuffs are white and shorter than that of the other species.
Coastal
nhakurutukutu (Piscatostrix atlantica)
Living in the forests near the Atlantic coast of South America, these birds
have 45 cm of length and a wingspan of 87 cm. Females are slightly larger than
the males. Their plumage is rufous-brown with the facial disk being light brown.
Their long ear tufts are white.
This bird species was discovered by João Vitor Coutinho, Brazil.
Valkyrie
owl (Valkiriostrix robusta)
Order: Owls (Strigiformes)
Family: Griffon owls (Griffonostrigidae)
Habitat: Eastern Asia, Beringia, west of North America.
Mass extinction of the end of Holocene has sharply negatively had an effect
at number of large species of predatory birds: their basic catch either had
died out, or its area was reduced. As a result large predatory birds - eagles,
condors and vultures - have simply died out: their rarefied populations were
so reduced, that the further reproduction of these species appeared impossible.
But after stabilization of conditions in Neocene many other species of predatory
birds have occupied become empty ecological niches had appeared. Species earlier
eating small mammals have especially succeeded: when the set of ecological niches
of large species has become empty, upsurge of number of small mammals began.
And when separate descendants of small animals became large, some of their natural
predators have turned to giants too. Owls had been especially lucky: these birds
in some areas of Earth had turned to rather frightening predators. One of such
predators inhabits woods and mountains of temperate zone of northern hemisphere
– it is large valkyrie owl which yields to South-American condor in size respect.
The valkyrie owl is the real giant among representatives of the group. Though
larger praire groundowl (Deinostrix
sphinga) lives in deserts of south of Northern America, the valkyrie owl
differs from it by its ability to fly. At weight up to 16 kg it has wingspan
over 4 meters. It considerably improves its hunting opportunities in comparison
with running relative from desert though limits weight of bird and as consequence
size of possible catch. But in hunting the main component of success is not
as many size of predator, as its hunting tactics. The valkyrie owl successfully
hunts large ground mammals – harelopes
and even obda cubs. Birds living
near of sea coasts frequently attack colonies of sea birds and eat bodies of
sea animals cast ashore after storm.
Valkyrie owl keeps features of characteristic “owl” appearance: on its head
there is an obverse disk formed by feathers though because of large beak its
head is similar a little to heads of predatory birds. Skull of this owl is rather
wide: binocular vision important for hunting is kept. Eyes at the valkyrie owl
are very large, this bird has excellent color vision: it can distinguish mouse
among grass at the distance over 100 meters. Under feathers big chink-shaped
ear apertures are latent: at the valkyrie owl there is keen hearing. Near ear
apertures characteristic for many owls feather “ears” have turned to two “tresses”,
sticking up back and to sides. At the male these “tresses” are longer, than
at the female, and at feathers forming them there are white tips: it is a unique
distinction in colouring between male and female.
Legs of the valkyrie owl are rather longer, rather than at other owls: it is
connected with its habit of life: birds of this species spend a lot of time
on the ground – prey of birds is frequently so large that the opportunity to
take it away to the tree (as other owls usually do) is excluded. On toes of
this bird claws of really monstrous size – up to 15 cm long – grow. This if
main weapon of valkyrie owls: attacking owl kills chased prey by them. With
the help of claws bird also divides catch: tears off big pieces of meat from
carcass and brings them to mouth. It swallows meat with skin and wool belching
them later as castings – it is a characteristic feature of owls. This bird can
swallow entirely relatively “small” catch weighting up to 2 kgs: mouth of this
owl is very wide (though it is almost imperceptible outside), jaw bones are
rather flexible and bottom jaw has elastic ligament.
Colouring of these birds is soft: dark-brown top of body with black spots and
cross strips on wings, and grey bottom with black tips of feathers on stomach.
The obverse disk of these owls is formed by yellowish-brown feathers and bordered
by black ones, therefore “face” of valkyrie owl is reminiscent somewhat of mask.
The habit of life of valkyrie owls considerably raises their chances of successful
hunting: these birds live in close breeding pairs keeping to all life. Pair
of birds supervises territory about 100 square kilometers not supposing occurrence
of competitors on it. In family relations at this species strict matriarchy
reigns: male weighs a little bit less than female and incurs significant part
of “female” family duties. It hatches eggs more than half of all term of incubating
and feeds nestlings up to their nest lefting with catch brought by female.
Pair of birds nests in the beginning of spring in rocky niches and other places
inaccessible to ground predators, each year in the same nidus which is strictly
protected and constantly renewed. In clutch there are only 2 large white eggs,
but usually only one young bird survives up to nest lefting. The incubating
of eggs lasts about 40 days. Nestlings hatch from eggs blind, but covered by
white dense down. Between nestlings since the first days of life the rigid competition
for forage takes place, and weaker one has chance to survive only at abundance
of forage. Usually parents feed only nestling shouting more loudly, therefore
nestling has weakened for famine, most likely, will not be fed: parents submitting
to parental instinct will give all prey to loudly wailing offspring. Having
received long-awaited piece of meat, nestling calms down to any time. If at
this time someone of parents has brought food it can get to the second nestling.
But such cases are the big rarity. More often it happens so that at lack of
forage stronger nestling eats weaker one.
Approximately at bi-monthly age survived nestling becomes completely fledged,
but about one month it spends in nest: primary feathers grow late. Nestling
actively trains muscles and studies to eat independently, tearing apart catch
brought by parents. The three-monthly nestling abandons nest and about one month
keeps near to parents. At this time lessons most important for it pass: it masters
receptions of large prey hunting. Young bird must study very quickly: that day
will come soon, when during prey sharing instead of share of catch it will take
some sound impacts by beak. It also will be a signal, that the childhood is
over and adult life began in which there is a place to famine and severe struggle
for a survival, to failures from hunting and to illnesses. But, if the young
bird will manage to survive and occupy good territory, at the age of four years
it can create own family and hatch posterity. Life expectancy of birds of this
species can be over 50 years.
Outs of nesting season valkyrie owls keep together and hunt in pairs. Except
for rather small catch (large rodents and cubs of animals) they can kill even
large animals. Hunting for large animals (for example, harelope)
these owls use the special tactics demanding coordination and mutual understanding.
Having tracked down herd of herbivores, pair owls divides. Female flies far
too the forest and hides, and male starts to frighten and chase animals. Usually
among animals there are such ones starting up panic flight, and all attention
of chasing bird is focused at one of them. Male drives prey in direction of
expecting female, which strikes crushing blow to preying animal sideways. Killing
prey, owl by one paw seizes shoulder of prey, and by another one top part of
its neck. Long claws of bird put deep wounds, and frequently simply pierce spinal
cord of prey, at once killing it. If the first impact appeared unsuccessful,
and the victim shows resistance, strong beak of owl is using.
Catch is eaten simultaneously by both birds from pair. Male cares to the female
signs of attention, tearing off from slices of meat carcass and handing them
to it from beak into beak. Thus he extinguishes aggression of the female. Males
behaving too “independently” sometimes exist, but in such pairs frequently there
are conflicts, which easily can finish by death of one bird (usually male as
weaker one).
Amicable pair of birds spends together almost all time. In nesting season these
birds, usually silent, arrange “duet” singing: male begins to “sing”, female
continues, and male finishes shout. Voice of this owl is bass lingering hoot.
Healer
trogon (Brevitrogon pediculivorus)
Order: Trogons (Trogoniformes)
Family: Trogons (Trogonidae)
Habitat: tropical forests of South America, preferring drier areas.
Trogons represent a group of birds of ancient origin, which turned out to be
very vulnerable to anthropogenic impact. Deforestation of tropical areras and
mass capture of these birds for zoos in Holocene, and then further reduction
of the area of tropical forests during the ice age greatly reduced the species
diversity of the order. As a result, only a few relict species were preserved
in the Neocene in dry and secondary forests. Despite further adaptive radiation,
trogons in Neocene represent a decaying group of birds. Healer trogon is one
of the unusual descendants of relic trogons of the ice age, a descendant of
the blue-crowned trogon (Trogon curucui). It avoids competition with passerine
birds due to its unusual lifestyle.
It is a small bird species: the body length of an adult is 15-18 cm along with
the tail. The coloration is quite striking – the male has red spots on black
wings, a greenish back and a blue head with a metallic sheen. Unlike the ancestral
species, the belly has a soft color – it is pinkish-gray. Female has brown body
and wings; head and shoulders are rusty-red. Birds of both sexes have a distinctive
feature: a cross stripe on the chest, slightly expanding in the middle. The
tail color of birds of both sexes is the same and catchy – the feathers are
covered with black and white cross-stripes. Paws are short and tenacious. Beak
is conical in shape, with a pointed tip.
Usually, outside of nesting time, this species keeps in small groups of 5-7
birds of both sexes on tree trunks and branches, as well as on lianas. Seeing
a relatively large mammal – notopongo, sidespiny
porcupine or other arboreal beast, healer trogons utter a characteristic
calling trills – a series of chirps and whistles, and also flutter along the
trunk. In addition, birds give visual signals to the beast by sharply stretching
and folding tails. Noticing the signals, the animal stops, allowing the birds
to land on the wool and search for parasites in it. Healer trogons have the
ability to extract larvae of parasitic dipterans from under the skin of animals.
Small queues sometimes gather at the “medical center” of the birds – up to 6-7
animals of various species waiting to be cleaned of parasites. Occasionally,
even predatory mammals use the services of these birds. The specialization of
this species to the wool cleaning of other animals is not deep. In the absence
of “clients”, the healer trogon is able to feed itself and feeds on caterpillars,
mollusks and slow-moving beetles. The main enemies of this species are birds
of prey and snakes.
The courtship season is not expressed. At any time of the year, both parents
with chicks, and ready-to-mate or incubating birds may be met. Male calls female
ready for mating with loud cries to the chosen place for the nest – usually
to a hollow or a cavity hollowed out in a termite mound or an abandoned wasp
nest. Eggs are laid directly on the bottom of the hollow, without any litter.
There are 2-4 rounded eggs of a greenish-rusty color in the clutch. The clutch
is hatched by both parents for about 2 weeks. The nestlings hatch naked and
blind, at 10 days they are covered with feathers and have eyes opened, at 3
weeks they already leave the nest and try to fly. The parents feed the juveniles
for some more time, and at the age of 6 weeks they are completely independent.
Young healer trogons form their own groups and clean arboreal mammals. Sexual
maturity occurs at the age of 7 months, life expectancy is up to 15 years.
This bird species was discovered by Nick, the forum member.
Appalachian
quetzal (Americotrogon appalachiensis)
Order: Trogons (Trogoniformes)
Family: Trogons (Trogonidae)
Habitat: the east of North America, the Appalachian mountains.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The eared quetzal (Euptilotis neoxenus), had not only
survived the anthropogenic onslaught, but also expanded their distribution to
the North. There this species gave rise to a new Genus, the Americotrogon. The
Appalachian quetzal is its type species. An inhabitant of the forest and woodlands
of the Appalachian mountains, this bird migrates to Great Antigua and other
Caribbean Islands during the winter.
The Appalachian quetzal has 36 cm of length, with a wingspan of 64 cm. Both
sexes have dark green backs and reddish brown heads, neck and chest. These birds
have iridescent dark blue central tail feathers, and outer tail feathers that
are predominantly white. The males have iridescent green feathers in their wing
coverts, while the females lack such feathers. Adult males have pale red lower
bellies, the females only have some patches of this color in this part of their
body. The bill is dull gray with a slightly darker band at the tip, the legs
are pale gray.
They feed on insects, small vertebrates, and fruit. Caterpillars, moths, katydids,
cicadas, small lizards, and other prey are fed to the young. Like other trogons,
they often pluck prey and fruit while hovering. Vertebrates and invertebrates
are more hunted when they need to feed their chicks.
Appalachian quetzals are territorial and monogamous. Males will repel other
members of the same species and even other hole-nesting species from around
their nesting sites. These birds nest in the beginning of the spring, as soon
as they return from migration. Males attract females by singing. Like other
trogons, they are cavity nesters. Nests are dug with their beaks, into rotting
wood or in the soft ground of cliffs. Nest digging is undertaken by the male
alone. In the case of nests dug into tree trunks, the wood must be strong enough
not to collapse but soft enough to dig out. When seeking a place to make the
nest, the male will land on dead tree trunks and slap the wood with its tail,
test the firmness.
The nests are usually unlined. Between two and four eggs are laid by the female.
These are round and lightly green. Both parents incubate the eggs, with the
male taking one long incubation stint a day and the female incubating the rest
of the time. The incubation begins after the last egg is laid. The incubation
usually lasts between 16–19 days. On hatching the chicks are altricial, blind
and naked. The chicks acquire feathers rapidly, usually in a week. They take
25 days to fledge. The young will follow their parents during the migration,
but will return alone. This species don’t form flocks during the migration,
with the only groups being that formed by pairs and their chicks. They reach
sexual maturity with 2 years and their lifespam is of 21 years.
Another representative of the Genus Americotrogon is the Florida
quetzal (Americotrogon flaviventer), an inhabitant of the forests and
wooded swamps of the Florida Peninsula. They have 33 cm of length and 58 cm
of wingspan. Both sexes have dark green backs and iridescent dark blue tail
feathers. The males have bright yellow heads, chests and bellies. In the females
these parts of their body are pale yellow. This species is non-migratory.
This bird species was discovered by João Vitor Coutinho, Brazil.
Snake-eating
hoopoe, jasper hoopoe (Euupupa serpentophaga)
Order: Hoopoes (Upupiformes)
Family: Hoopoes (Upupidae)
Habitat: south and middle zone of Europe (from Spain up to Ural, to the south
up to Fourseas), Central Asia, North Africa, Near East.
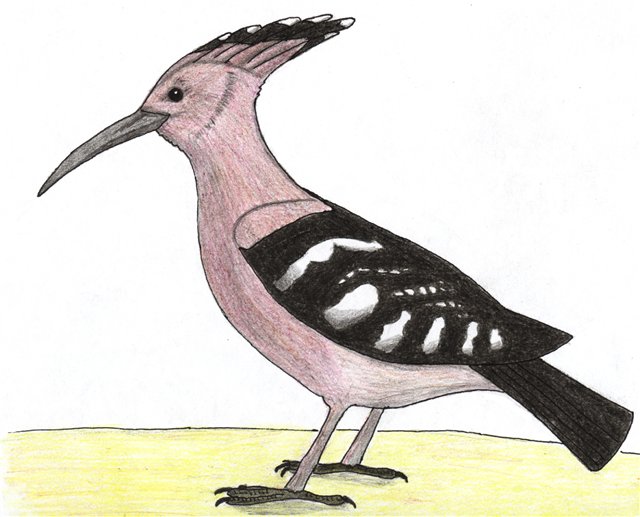
Picture by Simon
The greatest advantage in cases of mass extinctions is received
by unspecialized species of animals of smaller size. In this case it is easier
to them to survive, using various sources of food and habitats. And small size
helps to keep high number of population even in district rather poor in forage.
For this reason the majority of species of birds could go through mass extinction
at the boundary of Holocene and Neocene. Only highly specialized species (and
the majority of birds of prey among them), initially rare species, and also
inhabitants of tropical rainforests, which had missed during the ice age, had
died out. But inhabitants of dry open districts and light forests were widely
settled at Earth. Among them there were Euroasian species of hoopoes (Upupa).
In Neocene among descendants of hoopoes the new species had appeared, becoming
in some features the analogue of large ground hornbills and maned
crow belonging to corvine birds’ family.
The descendant of common hoopoe (Upupa epops), jasper hoopoe, is larger bird
than its ancestor. It reaches the size of kite. Feathering of bird is colored
greyish-pink with brownish shade (color of jasper mineral, one name is because
of it). On wings of jasper hoopoe the pattern inherited from the ancestor, black
and white cross strips stretched across the most part of wing, is kept. This
colouring is well appreciable to ground predators, even to hving no color sight
ones. Presence of such feature indicates that bird has effective protective
adaptation. Tail of bird is black.
On head of jasper hoopoe big mobile fan-like crest grows; it can fold and open.
The bottom part of crest feathers is colored dirty pink; top is black, and on
tips of feathers there are white specks. When the crest of bird is opened, the
black-and-white arch formed by the ends of feathers is appreciable from apart.
Legs of this hoopoe are strong, short, with sharp claws. They are equally well
adapted to walk on the ground and to clamber on branches. The hypotarsus is
feathered down to toes – this feature is connected to diet of bird. Beak is
long and sharp, slightly bent downwards: it is thicker, than at common hoopoe.
Jasper hoopoe eats amphibians and reptiles, but its favourite dish is snakes
(from here there is another name of bird), less often lizards. Near reservoirs
bird catches frogs. The way of food getting is hunting from the perch. Usually
jasper hoopoe arranges the ambush on alone tree or on high bush. Bird simply
catches frogs and lizards by beak, throws up and swallows. Snake is more dangerous
catch, therefore hoopoe kills it, beating by legs or piercing its head by sharp
long beak. Sometimes hoopoe can kill the snake, having seized it in beak to
the tail and striking by head against the ground. Bird swallows entirely even
rather large snake.
Jasper hoopoe arranges nest in tree-trunk hollows and natural crevices of rocks.
In clutch it may be up to 3 – 4 eggs with white shell; they are hatched mainly
by the female. Nestlings hatch in 30 days. They are naked and blind, and fledge
rather slowly: young birds leave nest approximately to 40-th day of life. They
are extremely inaccurate, defecate right in nest and leave in nest litter belched
parts of food. Because of it the nest of bird has a characteristic unpleasant
smell which is felt from apart. But birds are not afraid of predator attack
– they are well enough armed to resist to it. Cleanliness in nest is supported
by beetles and their larvae which at night eat the most part of the dross left
by nestlings. At the south of area birds make two clutches per one year, and
at the north only one clutch. Northern birds migrate to winter to the south,
overcoming the Alpes. Birds from Three-Rivers-Land steppes overcome Fourseas
through Crimea, Caucasus and Ustyurt Island.
Snake-eating hoopoe has the fine weapon which is effectively frightening off
the most part of enemies. The bird is able to shoot by dung to the enemy. Nestlings,
defending from a predator, sprinkle dung directly from the nest, having turned
back to the enemy. They attack any creature which has not warned about the approach
by the special sound, therefore adult birds, perching on edge of nest, utter
short signal to not be “shot” by own posterity. Adult birds apply “chemical
weapon” by much more effective and memorizing way. At first bird warns predator
of intention to defend, rising on legs and having stretched magnificent crest.
In such position the hoopoe rocks, turning head sideways to predator that the
crest was better swept up. If it has not conceived action, the hoopoe promptly
flies up from the ground vertically, loudly clapping by black-and-white wings.
At this moment its flight is similar to rise of gallinaceous birds. The hoopoe
hangs in air for one second at height about two meters. This way it shows warning
contrast colouring of wings, and their loud flapping is audible for tens meters.
If the enemy had not left intentions to attack, the bird turns tail downwards
and sprinkles to the enemy a jet of sticky and stinky dung. In cloaca of bird
the special gland opens, emitting a portion of protective liquid having unpleasant
smell. It strengthens efficiency of protection of bird even more.
This species of birds is discovered by Simon, the forum member.
Pink
lenaka (Lenaka callorostra)
Order: Hornbills (Bucerotiformes)
Family: Hornbills (Bucerotidae)
Habitat: deserts of South Africa.

Picture by Pavel Volkov
One of the characteristic groups of birds in Africa is hornbills.
Many forest species of this group became extinct in the human era due to the
destruction of tropical forests, and only some small savanna forms managed to
survive by Neocene. In Neocene, a number of their descendants live in Africa.
The common bird species include pink lenaka – an inhabitant of the deserts of
South Africa, a descendant of the species Tockus leucomelas. The species name
means “hornbill” in Sesoto language.
Pink lenaka is mainly a ground-dwelling bird. The height of a bird of this species
standing on the ground is up to 35 cm. In its proportions, lenaka is similar
to other birds leading a similar way of life: it has a medium-sized trunk on
long bare legs, wide rounded wings, a long narrow tail, an elongated movable
neck and a small head with a very large beak. The outgrowth on the beak represents
just a low crest on the upper side of the upper beak. The background color of
the plumage of lenaka is white with a grayish or beige tint, the legs are dark
gray; there is bare meat-red skin around the eyes and on the front of the head.
The back is light pink, the pink edges of the feathers on the wings form a scaly
pattern; tail and primary feathers are gray or beige. A remarkable feature of
the bird’s appearance is the bright beak: crimson with vertical yellow strokes
in males, light pink with pale spots in females. There are no other sexual differences
between male and female.
Pink lenaka is a fairly numerous species of birds; they can often be found in
the deserts of South Africa. Pink lenakas live in groups of up to 15 birds –
they are very sociable and love the company of relatives; alone, the bird experiences
obvious stress. The group does not have a specific place of residence and roams
a vast territory during the day, and at night the birds sleep in any random
shelter – more often on a tall aloe tree with prickly leaves. Usually flocks
of these birds constantly communicate noisily, so their approach can be noticed
from afar. These birds are generally quite intelligent, not inferior in intelligence
to corvids and parrots, and can use natural objects as tools.
The diet of this species is diverse: leaves, fruits, seeds, invertebrates and
small vertebrates, as well as carrion. Pink lenakas often follow large animals
– both herbivores and predators. For herds of herbivores, they serve as a kind
of sentinels, warning them of danger. Accompanying predators, birds behave quietly
and cautiously, because they eat the remains of their prey and are often the
first to be near the left remains of prey and have time to eat a lot before
the appearance of stronger scavengers. The enemies of this species, as a rule,
are large birds of prey, less often snakes and small predatory mammals. Pink
lenaka is characterized by group protection: noticing a predator, birds mob
it, surrounding it with their whole flock.
The breeding season is at the end of the drought. Males arrange courtship displays,
and females choose the one with the brightest beak. Birds build nests in rocks
from grass, twigs and feathers. Both parents incubate the clutch (up to 3 eggs
in it). After 2 weeks, the nestlings hatch, and both parents take care of them
– while one heats and protects them, the other carries food. By this time, the
wet season comes, and it is easier to feed the juveniles.
Life expectancy is up to 20 years.
This bird species was discovered by Mamont, the forum member.
Great
crested hornbill (Neobuceros magnus)
Order: Hornbills (Bucerotiformes)
Family: Hornbills (Bucerotidae)
Habitat: tropical forests of Borneo Island.
During the Holocene, human interference in nature caused the extinction of many
species. Only those that were resilient enough were able to survive and left
descendants. Among these was the oriental pied hornbill (Anthracoceros albirostris).
Differently from the other hornbills, this species was capable of surviving
in degraded habitats and continued to evolve. In the Neocene, it gave rise to
the Genus Neobuceros. The type of species of this genus is the Great crested
hornbill, a larger inhabitant of the forests of Borneo.
The great crested hornbill is a large bird, with 128 cm of length and a wingspan
of 145 cm. This bird have sexual dismorphism, the male have a larger red casque
and beak that can reach 50 cm. The females that lack such structure and are
smaller than the males. The male casques are laterally flattened, which may
form a bright red protruding crest. The plumage of both sexes are similar, with
the head, neck, back, wings and upper breast being black. The tail is white
with black tips. The plumage of their lower breast, lower abdomen, thighs, under-wing
and all the tips of the wings are white.
The great crested hornbill's diet consists mainly of fruit. Figs are particularly
important as a food source. They obtain water entirely from their diet of fruits.
They are important dispersers of many forest tree species. They will also eat
small mammals, birds, small reptiles and insects. Sometimes primates can be
seen foraging alongside these birds. They forage along branches, moving along
by hopping, looking for insects, nestling birds and small lizards, tearing up
bark and examining them. Prey are caught, tossed in the air and swallowed.
These birds are monogamous, their nesting season coincides with the onset of
rain depending on geographic location, and peak abundance of fruit. The great
crested hornbills are secondary cavity nesters, meaning that they typically
do not excavate their own nesting sites but use those created by other birds
or by branches breaking off. When females have selected and entered their nest,
they seal the cavity with a mixture of saliva, mud, fruit, droppings and tree
bark, leaving only a small opening through which food may be passed in. The
male forages for the female and chicks, and the female feeds the nestlings.
The clutch consists of one or two eggs, which she incubates for 39 days. After
20 days since they hatched, the female will leave the nest and the chicks seal
it again. They remain inside the nest for 3 months until they are ready to fledge.
They reach sexual maturity with 5 years and they have a lifespan of 32 years.
The lesser
crested hornbill (Neobuceros minor) is another representative of this
Genus. This species is more widespread than the Greater crested hornbill, living
in Borneo, Philippines, Celebes and Sunda lands, where it produces numerous
local forms. This species have 60 cm and a wingspan of 78 cm. The females are
smaller than the male and lack the casque, which in the males is bright yellow
and has 37 cm of length. Both male and females have bright yellow beaks. The
plumage of the neck, back, wings and upper breast is black, while the rest is
white.
This bird species was discovered by João Vitor Coutinho, Brazil.
Meganesian
hornbill (Nanobuceros meganesianus)
Order: Hornbills (Bucerotiformes)
Family: Hornbills (Bucerotidae)
Habitat: Northern Meganesia, tropical forests.
During the Holocene, human interference in nature caused the extinction of many
species. Only those that were resilient enough were able to survive and left
descendants. Among these was the oriental pied hornbill (Anthracoceros albirostris).
Differently from the other hornbills, this species was capable of surviving
in degraded habitats and continued to evolve. Through the Sunda Land, some of
its descendants were able to reach the tropical forests of Northern Meganesia
were they became the ancestors of the Meganesian hornbill.
This species has 32 cm of length and a wingspan of 47 cm. The upper parts of
their bodies are black, while the flight feathers of the wing are white. The
tail is also white with a black tip. The casque is only present on the males.
It is bright yellowish-orange, short and pointed. The female has pale yellow
beaks, while in the males it's bright yellow.
They are omnivorous, taking and eating insects, fruit, seeds and even small
rodents. They feed only on the forest canopy and will form flocks outside the
breeding season.
The Meganesian hornbill is monogamous, with pairs staying together for life.
The nesting season starts during the monsoon period and the clutch varies from
one to five very symmetrical white eggs. These birds usually nest in tree hollows
on tall trees. An existing hollow may be excavated further to suit. The female
enters the nest hollow and seals the nest hole, leaving only a small vertical
slit through which the male feeds her. The nest entrance is sealed by the female
using its excreta and mud-pellets supplied by the male. While inside the nest,
the female moults her flight feathers and incubates the eggs. The male provides
the nest inmates with a steady supply of bark pieces to ensure that the excreta
is removed from the nest by absorption and adsorption and to help maintain the
micro-climate within the next cavity.
The female incubates the eggs for 28 days. After 19 days, the chicks and the
female are too big for the nest. Then, the mother breaks out and rebuilds the
wall, leaving the chicks inside for their continued protection from predators.
Then both parents continue to feed the chicks. They remain inside the nest for
38 days until they are ready to fledge. They reach sexual maturity with 3 years
and they have a lifespan of 27 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Tiny
straw kingfisher (Micralcyone scirpophyla)
Order: Coraciiformes (Coraciiformes)
Family: River kingfishes (Alcedinidae)
Habitat: lower reaches of Three-Rivers-Land rivers, Fourseas reed beds, the
Kara Kum beach and Ust’-yurt island.

Picture by Alexander Smyslov
Reed thickets represent the fine place for reproduction of
various fishes and other water inhabitants. Here they find food and set of shelters.
And also large predators are absent here. But all the same they can't feel in
safety – one of the smallest birds of reed beds, tiny straw kingfisher hunts
them.
This is a tiny bird, the descendant of common kingfisher (Alcedo atthis), is
smaller in size rather than sparrow. It has short wings and tail, but large
head and pointed beak. Tiny straw kingfisher is perfectly adapted for life in
reeds. Its main color is light yellow, and wings, tail and top of head which
sparkled an azure at its ancestor, have got brownish color with gray longitudinal
strips. Having such coloring this bird is invisible to predators occasionally
visiting these places. Beak is black, but paws and eyelids of males are bright
red (at females they have dim orange color).
In behavior pattern this species is very similar to the ancestor, but it nests
not in holes dug out in steep river bank, but in cavities of trunks of papyrian
reed mace. Because this kingfisher is not able to peck a hollow in strong trunks
of reed mace, it depends very much on activity of another bird, tiny cane woodpecker.
It happens that a couple of kingfishers ready to nesting expels small woodpeckers
from their hollow and destroys their clutch, or waits while birds will peck
a hollow to occupy it immediately. If the number of woodpeckers is enough, kingfishers
peacefully adjoin to them, occupying their nests made the last year. Kingfishers
often arrange nest in the broken trunks of huge reed mace. In the absence of
a suitable hollow of a bird can dig out a hole in sandy steep (the birds from
Ust'-yurt island do it mainly).
Tiny straw kingfisher eats small fishes and invertebrates, tracking down them
from a reed trunk, or hanging in air, as if the hummingbird. During this moment
its wings make up to 20 waves in a second, but the bird is tired quickly and
can’t track down fish for a long time, flying in such way. But «hummingbird
flight» is an important point of courtship ritual of these birds.
The pair is formed for one nesting season. In clutch there are 4 – 5 eggs, the
laying repeats twice within summer and if the first clutch was lost at an early
stage of incubation, birds have time to make the third clutch and to grow up
nestlings. The rearing up of nestlings takes about 5 weeks, and till the last
week of this period parents finish feeding of the youngs which have already
left a nest.
This species of birds was discovered by Simon, the forum
member.
Translated by Alexander Smyslov.
Finnish
or diving kingfisher (Boreodacelo speciosus)
Order: Coraciiformes (Coraciiformes)
Family: Kingfishers (Alcedinidae)
Habitat: freshwater reservoirs of Northern Europe, wintering at the Fourseas
and to the south of it.

Picture by Alexander Smyslov
The common kingfisher (Alcedo atthis) was one of the bird
species that relatively successfully survived in the human era. Its tropical
relatives were significantly affected by human activity, but this species was
quite widespread and retained a high number and a wide range. In Neocene, due
to the warming of the climate, it settled further north, and the northernmost
of its descendants lives in Scandinavia and on the Kola Peninsula. This is Finnish,
or diving kingfisher.
Diving kingfisher is much larger than its ancestor, which is due to its inhabitation
in a colder climate: the body length of Finnish kingfisher is about 40 cm, the
wingspan is up to 70 cm. Finnish kingfisher has a very long pointed beak (about
17 cm) with a small hook on the upper jaw tip. Tail is quite short. Diving kingfisher
also has elongated legs with large hooked claws. Wings are wide, but pointed
at the tips. In its body shape the bird resembles its ancestor of the human
era. The color of the plumage is very bright: against the background of other
birds of Fennoscandia, Finnish kingfisher looks like a tropical bird. Back and
upper part of wings are of various shades of blue with an expressed metallic
luster. There are red cross stripes on primary feathers. Tail is turquoise,
undertail is yellow. Belly and chest are reddish-orange with purple tips of
feathers on the throat. On the crown and back of the head there is a dark green
stripe stretching to the shoulders; sides of the head are red. Beak is dark
gray, legs are pink. Sexual dimorphism in coloration is not pronounced.
Finnish kingfisher spends all the warm season on the Kola Peninsula and in Scandinavia.
For housing, the bird uses either a hole dug independently in a steep riverbank,
or a tree-trunk hollow. Diving kingfisher is active during the day, sleeping
at night in its shelter. The bird is very territorial; it drives relatives from
the territory, aggressively treats gulls and waders looking for food nearby.
This bird species is exclusively zoophagous. Its prey is very diverse: invertebrates
(worms, mollusks, crustaceans, aquatic insects), small fish and amphibians.
The bird hunts in several ways. Usually the Finnish kingfisher perches on the
branch of a tree growing near the water and looks out for prey. When the bird
sees it, it dives into the water, catches it with its beak and kills it by beating
against the roost, or pecks it, pressing it to the roost with its foot and then
eats it. Also, diving kingfisher can run along the river bottom against the
current, like a dipper, peck small prey and swallow it immediately. For the
kingfisher itself, some birds of prey are dangerous, and large fish and amphibians
are in the water.
Finnish kingfisher is a migratory species; it winters on the shores of Fourseas
and further south, and at the end of March it returns to the nest, unmistakably
finding its own territory. The male digs out a new nest and displays himself
in flight or by perching next to the nest. When the female makes her choice,
the birds mate, and then the female lays 2-3 eggs in the nest without litter.
Exclusively the female incubates them for a month while the male feeds her.
When the nestlings hatch, the female warms them during the first days of life,
and then leaves the nest and also begins to look for food for them. The nestlings
grow quickly and leave the nest in the middle of summer. They differ from adults
in their dull juvenile plumage, and their belly and chest are colored brown.
Young birds feed on the parent territory, learn to catch prey, and at this time
they are fed by their parents. The birds migrate independently, one by one;
young birds sometimes migrate for the winter in groups, but return to the north
alone.
Young birds become sexually mature by the following spring, although in the
first years of nesting they rarely manage to occupy a good feeding area and
feed the brood. Life expectancy is about 30 years.
This bird species was discovered by Mamont, the forum member.
Dire
Kookaburra (Archodacelon rapax)
Order: Coraciiformes (Coraciiformes)
Family: Kingfishers (Alcedinidae)
Habitat: Woodlands, forest, open forest, scrubland, tropical rainforest, across
most of eastern, northern and central Meganesia, absent from deserts.

Picture by Pavel Volkov
The kookaburra (genus Dacelo), was a successful bird in the
Holocene, able to live alongside people, well liked for its habit of eating
snakes and singing in a jolly way. During the Neocene, the numbers of raptorial
bird predators was greatly reduced, though some diversity remained in Meganesia,
other forms such as Ibis, Kookaburras,
day-flying owls and large bats,
competed with them.
Numerous descendants exist in the Neocene, of the kookaburra, including forms
that feed near water, and also one terrestrial form, a fair sized predator that
competes actively with other kinds of predatory bird. This bird, the Dire Kookaburra,
is about as large as a raven, with a wingspan of up to 90 centimetres, thus
larger than its ancestor. Size is connected to aggressive, predatory habits,
similarly to its ancestor it feeds mostly on small vertebrates and large arthropods,
including snakes, lizards, frogs, rodents, locusts, centipedes, beetles, as
well as certain small marsupials and their young. This bird is even known to
harass mid-sized marsupials such as phalangers and marsupial lemurs, in order
to prey upon ejected pouch young, nestlings and eggs of birds are also eaten.
Bodyshape and wings generally resemble ancestor, apart from somewhat longer
wings and stronger legs, as well as particulars of the beak. Feathers are a
mid-shade of brown, striped along each feather with light grey, in a “scalloped”
pattern. Body underside and lower part of face is yellowish white, face has
a dark brown “mask” of feathers, and an erectile crest of feathers sits atop
the head, a little more prominent than that of its ancestor. It mainly differs
from ancestor by its beak, which is deeper, a little shorter, and ends in a
large hooked notch, similar to the tomial tooth of a Falcon. Lower jaw is lined
with smaller keratinous “teeth”, colour of beak is a dark orange, with the tip
and upper edge being black. Males possess a greenish-blue patch of feathers
on the mid-wing, which is only seen when in flight. Call is similar to that
of its ancestor, but is a deeper, more menacing cackling sound as opposed to
jolly chuckle.
This bird hunts by perching on a low branch or tall rock, seeking about for
small animals to catch, when one is sighted, it swoops down and seizes it with
beak , landing and then dashing the prey head-first against the ground to kill
it. Unlike its ancestor, it is able to tear larger prey into small pieces with
its beak; in this way it is able to eat somewhat bigger snakes, lizards, and
even rats. This bird is completely diurnal, but during twilight may attack fruit-bat
colonies in order to consume young bats.
Dire Kookaburra usually live in mated pairs which defend a large territory,
calling loudly to warn off intruders, any interlopers are chased, swooped and
may be pecked heavily. Unlike relatives, pairs form for life and they call and
hunt cooperatively. Pair nests in the large hollows of trees during the wetter
months from June to September, 2 large eggs are laid and incubated alternately
by both parents. The young are fed by both parents, though the larger chick
invariably dominates the other, getting all the food and injuring the runt on
purpose. Young birds fledge within 2 months, leave the nest and will remain
with parents for another month learning to hunt, sexual maturity is reached
at 10 to 11 months. Lifespan may be over 20 years and as many as 30, though
usually shorter.
This bird species was discovered by Timothy Morris, Adelaide, Australia.
Waxeater
(Ceraphaga sylvatica)
Order: Woodpeckers (Piciformes)
Family: Honeyguides (Indicatoridae)
Habitat: tropical woods of Central Africa and Zinj Land.
In struggle for existence one of the most widespread ways of survival is narrow
specialization. Thus the species becoming the specialist simply avoids a competition
to relative ones because its vital interests are crossed with those at neighbours
in minimal degree. But such way of existence has the extremely unpleasant return
side: at change of conditions of environment highly specialized species die
out at first. But sometimes there are happy exceptions when at changes of inhabitancy
limiting vital resource is kept in rather constant amount. Then specialized
species have a chance of survival.
One of such “lucky beggars” lives in tropical woods of the Central Africa. It
is large crow-sized bird of chartreuse color with orange “epaulettes” on wings.
The bird is the descendant of honeyguide (Indicator), bird specialized for feeding
by wax and contents of bee combs. The beak of bird is black, massive, hooked,
resembling beaks of parrots, but more lengthened and not such hooked as at them.
This bird eats mainly… wax, as well as its ancestors therefore it has received
the name “waxeater”. Symbiotic protozoans which decompose wax live in stomach
of waxeater and synthesize vitamins necessary for bird.
Waxeater is more specialized to getting wax in comparison with honeyguides of
Holocene epoch. When to Holocene honeyguide the help of honey badger is required
for catch of food, waxeater manages this problem itself. Having found bee colony,
waxeater does not call anybody to the aid: with the help of beak the bird simply
breaks open an entrance in tree-trunk hollow where the bee nest is placed. Of
course, bees try to expel the uninvited visitor from the possession. But it
is not so simple to make it – feathering of waxeater is very dense, and stings
of insects simply do not reach skin. Only one weak spot remains – eyes of bird.
But here the waxeater also has additional protection: above eyes big mobile
“eyebrowes” of rigid bristle-like feathers of yellow color grow. When bird ravages
bee nest, eyes are closed by these “eyebrowes” which fall downwards during attack,
“curtaining off” eyes.
Ravaging colony of bees, waxeater eats only wax, almost not paying attention
to honey and bee larvae. Having had eaten the share of combs, bird simply departs.
I t is completely unprofitable to waxeater to ravage nest completely, therefore
for day the bird attacks two – three bee nests, feeding at every one only a
short time. This bird holds in memory the location of all bee nests at the territory,
and regularly visits them. Because food resources are limited, waxeater is very
much territorial and is not declined to communicating with congeners at all.
Bird notifies competitors that the territory is occupied by loud calls. Cry
of this bird resembles a hoarse croak. It may be heard at the distance of several
kilometers.
Waxeater is not unique bird feeding at bee nests. The nest ravaged by bird is
immediately attacked by various “easy money” lovers which, being unable to ravage
bee colony, watch for waxeater. When this bird finishes meal, they eat rests
not touched by this bird – some honey and larvae. Though it is uneasy to make
– bees are annoyed and ready to attack any live creature which has casually
appeared nearby.
Waxeater frequently applies other way of food getting. It watches the beekeeper
parrot living in the neighbourhood: it is also specialized to feeding in
bee nests. But parrot eats honey and bee larvae, leaving after the meal empty
combs. Therefore waxeater frequently simply expects aside while beekeeper parrot
licks honey from cells of comb. And the empty combs are the food of waxeater.
Sometimes waxeater simply settles near to the nest of parrots, and constantly
accompanies with these birds in search of food.
If it is not enough bee nests, waxeater is content with “substitute” – bird
scrapes out vegetative wax from leaves of some palms and young sprouts of tropical
trees. Features of digestive system of waxeater permit it to feed even with
such food which is not eaten by other animals, except for insects: bird pecks
drops of stiffening gum emitting on trunk of trees belonging to family Euphorbiaceae.
This substance is poisonous for other animals.
Eating wax and others hardly digesting substances, waxeater can not bring up
nestlings with the same food – other kinds of food rich in proteins are necessary
for nestlings’ development. Therefore waxeaters lay eggs in nests of other feathered
neighbours. While there is no hatching bird at the nest, waxeater lays in its
nest one egg, sometimes throwing out one of eggs of the host bird. Eggs of waxeaters
are very small in comparison with the size of body, and more similar to eggs
of smaller warblers. Nestling hatches naked and blind. It kills nestlings of
host bird, and receives all food alone. In another's nests nestlings of waxeater
are brung up with insects. Gradually nestling outgrows adoptive parents, and
even squeezes nest. Then it simply changes to branch, and adoptive parents feed
it, perching to its back. Nestling of waxeater becomes fully fledged rather
quickly: already at week age. It quickly grows, and gradually it becomes more
difficult to adoptive parents to warm the переростка stepchild.
Young waxeaters sharply differ in color of feathering from adult birds. Feathering
of young bird is brown with separate light feathers; wings are spotty, similar
to lichens by colouring. Young waxeaters are silent and cautious. After the
mew waxeater gets characteristic bright colouring of adult bird.
The close species living in savannas of Africa is savanna
waxeater (Ceraphaga savannophila). It differs in sand color of feathering,
grey wings, and larger sizes (it is raven-sized one). This bird spends a lot
of time on the ground, digs nests of ground bees and bees settling in trunk
hollows of baobabs. Savanna waxeater is not too specialized in diet: bird willingly
eats larvae of bees.
This species of birds is discovered by Simon, the forum member.
Cadaverornis,
corpse bird (Cadaverornis indicans)
Order: Woodpeckers (Piciformes)
Family: Honeyguides (Indicatoridae)
Habitat: tropical forests and woodlands of the Central Africa, Zinj Land.
In Neocene woodpeckers order has reached the great evolutionary successes. Among
woodpeckers, the central group of order, the species occupying various
(and sometimes unexpected)
ecological niches had evolved. But also “younger brothers” of woodpeckers, representatives
of other families of order, have continued the evolutional development. For
example, representatives of honeyguides family (Indicatoridae), known in human
epoch because of their unusual diet and way of food getting, had continued the
evolution in different ways. One of them, waxeater,
has deepened food specialization of its ancestors. Other species, named cadaverornis,
had cardinally changed the diet.
This bird eats mainly carrion. But the small size and weak beak do not permit
it to open bodies of dead animals independently. Therefore, having found a corpse
of large animal, it tries to draw attention of any omnivorous animal to it,
acting the same way, as Holocene honeyguide found a bee nest. When the “partner”
of cadaverornis tears carrion, bird receives a part of food. When the sated
animal leaves carcass, cadaverornis is fed still on carrion for any time, eating
not only meat, but also pecking larvae of insects. In addition to carrion this
bird searches for hollows with eggs and nestlings, holes with mammalian cubs
and clutches of reptiles in ground. It also draws attention to them of the animals,
capable to reach this source of food, having broken a hollow or having dug the
ground. In such cases cadaverornis usually also receives a share of prey. But
this bird can also get food independently: it quite often catches and eats large
beetles, small lizards and frogs.
Not only observation and the advanced intelligence permit cadaverornis to find
successfully carrion and other easy for getting sources of food in rich forests
and woodlands, but also other feature, much rarer for birds. The matter is that
at this bird sense of smell is well advanced. This feature, peculiar to honeyguides,
had been developed even deeper at their descendant. However cadaverornis has
lost some features characteristic for homeyguides. For example, thick skin protecting
from bee stings and symbiotic microflora of the stomach, assisting to digest
wax, are not necessary for bird never ravaging bee nests.
Cadaverornis has not striking greyish-green coloring. Only primarily and tail
feathers have black color with green shine, and underside of wings appreciable
in flight is white. Contrast colouring of these sites of plumage helps this
bird to draw attention of “partner” in search of food. Beak, resembling crow’s
one a little, is black, and legs are grey. “Face” of bird lacks feathering –
it is an adaptation to feeding on carrion which has independently appeared at
unrelated species of birds eating carrion; parts of featherless skin on head
have light grey color. In size and body shape cadaverornis resembles song thrush:
the total body length of this bird reaches 25 cm at wingspan of up to 35 cm.
The voice of cadaverornis sounds like loud dry crash or murmur.
Cadaverornis, as well as its ancestors, is brood parasite. But the unusual way
of life has left an effect on its breeding features: in order to avoid casual
cannibalism, cadaverornis never lays eggs in nests of birds nesting in hollows.
Usually nestlings of cadaverornis are brought up by the birds arranging open
nests. More often the foundling kills host nestlings.
This species of birds was discovered by Simon, the forum member.
Rainbow
bald-toucan (Capitocalvus versicolor)
Order: Woodpeckers (Piciformes)
Family: Barbets (Capitonidae)
Habitat: South America, tropical and subtropical forests.

Picture by Pavel Volkov
The human actions during the Holocene caused the decline of
many species. However the species that were resilient enough survived, with
their descendants now living in the Neocene. Some species of barbets (Capitonidae)
were capable of surviving. Due to great changes in their environment, these
birds ended up needing to adapt and even change much of their morphology. In
the Neocene, they give rise to the genus Capitocalvus, the bald-toucans. While
they aren’t true toucans, their appearance and lifestyle is similar to that
of true Ramphastidae. The type species of this genus is the rainbow bald-toucan,
an inhabitant of the tropical and subtropical forested areas of South America,
from Amazon to the Atlantic rainforest.
The rainbow bald-toucan is the largest representative of its genus. This species
has 54 cm of length and a wingspan of 97 cm. Their strong and thick beak is
14 cm long. It has a white color with a black tip. Such pale and simple coloration
is a stark difference from the true toucans and their colorful beaks. All males
of this genus have a flat projection on the top of their beaks, similar to the
casque of the hornbill. It reaches part of their skull, giving a bald appearance.
Both males and females have a colorful plumage. The head is bright red, while
the underparts are yellow. The back, tail and wing coverts are green, while
the primaries and secondary feathers are blue. Their legs are black and strong.
These birds are opportunistic omnivores. While fruits are a dominant part of
their diet, they will also target other animals as prey. Invertebrates such
as insects, scorpions, spiders and centipedes are common prey. Among the vertebrates,
the rainbow bald-toucan is capable of killing small mammals, small birds, frogs
and lizards.
The rainbow bald-toucan is usually found in pairs or small familiar groups.
These groups show a marked territorial behavior towards other groups or species,
which is usually made clear by the loud duets of breeding pairs, and it is actively
protected by the breeding pair by chasing off intruders. The family group's
helpers assist in this, especially near the nest. In the absence of interference,
these groups can occupy a particular territory for a year or longer. The rainbow
bald-toucan breeds cooperatively, with several helpers aiding the dominant breeding
pair with incubation and raising the young. Groups are larger outside the reproductive
season but generally shrink to three individuals in season, usually composed
of previous immature offspring that stay with their parents and help with the
new hatchlings. After the breeding season, the group increases due to greater
acceptance of non-family members. These helpers significantly increase the reproductive
success of the breeding pair. The breeding season of this species starts in
the middle of spring and lasts until the end of autumn. Pairs may have two or
even three broods per year.
They build their nests inside holes carved in cliffs. The construction of such
holes is made solely by the male, who uses their strong beak to remove the soil.
They also use the flat part on their beak and head to leave the soil inside
it compacted. The female will fiscalize the construction and when it's deep
enough, she will fill it with leaves and lianes. The female usually lays two
to four white eggs a few days after mating. The eggs are incubated by both sexes
and hatch after 18 days. The young will be attended in the nest by their parents
and siblings, until they fledge with 20 days. Sexual maturity is reached with
one year and they have a lifespan of 14 years.
Another species of the genus is the blue-throated
bald-toucan (Capitocalvus cyanogularis), an inhabitant of the cloud
forests at the slopes of the Andes. This species is 32 cm long and has a wingspan
of 68 cm. Their plumage is mostly green, with the exception of their throat
and chest that is blue, and head that is red.
This bird species was discovered by João Vitor Coutinho, Brazil.
Green
wryneck (Jynx viridis)
Order: Woodpeckers (Piciformes)
Family: True woodpeckers (Picidae)
Habitat: thickets of baobab opuntia in North Africa.
Around large baobab opuntia
cacti from North Africa a whole complex of species tied to this plant develops.
Birds are quite numerous among these species; one of them is green wryneck,
which is a descendant of the red-throated wryneck (Jynx ruficollis) settled
north of the rainforest region when the Sahara became savanna from the desert.
This species is a relatively small bird: the body length of green wryneck is
12-13 cm, the wingspan is up to 25 cm. In its proportions, green wryneck is
very similar to its ancestors: it has an elongated body, a straight-cut tail
of medium length, not very long legs with large claws and a small head on a
movable neck. Beak is longer and stronger than that of other wrynecks. Eyes
are large; green wryneck has a very good sight. The coloring is suitable for
masking on the shoots of baobab opuntia: the plumage of the bird has a light
green color and numerous spots, smears and strokes of light yellow color. Throat
is red. Legs and beak are grey, but the lower jaw is light blue.
Green wrynecks live in the sparse forests of baobab opuntia in North Africa.
Males and females lead different lifestyles: females are not territorial and
fly from one cactus to another; each male controls one tree and builds a dwelling.
This species is a cavity nester, uses either an abandoned hollow of any other
bird speies, or a hole in wood of another origin. There, the green wryneck male
arranges a dwelling: with his beak, he chops off pieces of soft cactus wood
and eventually makes a hollow of suitable size for himself, and then every morning
claims his rights to this nest by vocalization. Green wryneck is active during
the day, looking for food on a tree and on the ground near it. During the search
for food, green wryneck moves by jumps or slowly “crawls” along the trunk of
the cactus. The food of this bird includes small insects and arachnids, small
snakes and lizards, sometimes rodents or bird eggs and chicks. There are also
seeds and berries in the diet of green wryneck; the bird pecks the fruits of
the baobab opuntia. The enemies are large birds of prey, less often viverrids,
cats, snakes and large lizards. When they attack, the bird either tries to fly
away, or moves its neck in a characteristic way and hisses, mimicking a snake.
Nesting takes place during the wet season. The male “dances” near his nest and
calls loudly, attracting females. When the female flies and she likes the nest,
a pair is formed. The formed pair of wrynecks makes a new litter in the nest,
and then the female lays up to 10 light blue eggs in the nest. The clutch is
incubated alternately by both parents; the nestlings hatch after 2 weeks. They
are fed by both parents; young birds become independent at the age of 25 days.
Sexual maturity occurs at the age of 8-9 months, life expectancy is 13-15 years.
This bird species was discovered by Mamont, the forum member.
Bamboo
woodpecker (Dendrocopus bambusophilus)
Order: Woodpeckers (Piciformes)
Family: True woodpeckers (Picidae).
Habitat: bamboo woods of China, South-Eastern Asia and the Coast Jakarta.
Alongside with “atypical” woodpeckers of Africa, Southern Asia and Caribbean
islands in Neocene there is a plenty of “typical” woodpeckers of quite recognizable
appearance belonging to family of true woodpeckers existed in human epoch, and
even to genera, known to people. It indicates that woodpeckers have occupied
a convenient ecological niche from which nobody could press them essentially
till past millions years. Evolution of “true” woodpeckers proceeded in Neocene,
and some species of these birds have developed interesting adaptations to their
inhabitancy.
For mountain areas of East and South-Eastern Asia bamboo woods widespread in
hillsides to tens kilometers are characteristic. Here there is a little of large
animals, but various small creatures prosper. And among them there are woodpeckers
of particular species too.
The bamboo woodpecker is a medium-sized bird (starling-sized or a little bit
larger). Its addition is typical for woodpeckers: at it there are strong chisel-like
beak, pointed tail of rigid feathers, and tenacious four-toed legs. This species
has cryptic colouring: the general background is grayish-green, and on back
and wings there are brown longitudinal hyphens and strokes. On the head of this
woodpecker there is a “cap” of brick-red color characteristic for representatives
of family; at the male it is larger, than at the female. At the male there is
also bright red feathers under tail using for demonstration in courtship games.
Due to colouring the woodpecker masks well from predators of various kinds,
and in wood it is possible to hear its voice - loud nasal “kee-kee-kee!” more
often, rather than to notice a bird.
By features of behavior the bamboo woodpecker is similar to other representatives
of genus Dendrocopus. The bird eats insects, extracting them, pecking stalks
of bamboo. As against to other woodpeckers which can hollow firm wood, this
species searches for insects mainly in poorly lignified growing trunks of bamboo
where they live more often. Also the bamboo woodpecker searches for food on
the ground.
“Housing problem” is not critical for this species of birds: in bamboo wood
practically any lignified trunk represents a convenient place for life. The
bird only makes the hollow at the necessary height, and settles in cored internode.
Usually the male is engaged in construction of hollow. It possesses a leading
role in courtship games. The male ready to nesting, having arranged a bamboo-trunk
hollow, starts to invite females for nesting, “drumming” on well resounding
trunk of bamboo. When the female ready to nesting, flies to the site of the
male, he arranges “vertical races”: male tries to sit on trunk of bamboo above
the female, and shows to her red feathers under tail.
Pair is formed to one season of nesting, and breaks up after nestlings become
independent. In clutch of these woodpeckers there are 2 - 3 eggs, nestlings
are fed by both parents.
These birds are settled, and seldom leave rendered habitable territory. Unique
large “trouble” for bamboo woodpeckers is the mass flowering of bamboo after
which this graminoid dies off. In this case number of species is reduced tens
times, and usually birds lived in territory where some species of bamboo grow
survive: they have an opportunity to move to other species of bamboo.
To the north, on Japan Islands, Sakhalin and Big Kurils where bamboo is presented
by small species, the close in behavior species live: fennel
tree, or homeless woodpecker (Dendrocopus anethophyllumobius). It also
is approximately a starling-sized bird colored greenish. By features of colouring
this species is similar to the large spotted woodpecker (Dendrocopus major),
known in historical epoch. But where at the large spotted woodpecker black color
was in feathering, at fennel tree woodpecker these areas are dark green, and
white color is replaced by light green. Brightly colored feathers under tail
and “cap” at fennel tree woodpecker are kept, but have got more light orange
color. Similarly to the majority of woodpecker species, the male has larger
“cap”, than the female.
This bird inhabits thickets of fennel tree (Anethophyllum bambusoides), widespread
on Pacific coast of the Far East. Fennel tree woodpecker makes hollows in stalks
of this huge grass and rears posterity here. Each woodpecker preserves the territory
against contenders. Frequently the bird flies up on the highest stalk of plant,
and notifies neighbours on the claims on territory by loud creaking cries. This
interesting bird eats insects pecking them out from stalks of fennel tree just
like other woodpeckers take insects from under tree bark. The most desired catch
for this woodpecker is a nest of fennel tree leaf beetle (Anethophyla nanopicus).
This beetle arranges in hollow internodes of fennel tree “kindergartens”, and
the woodpecker destroys at once whole hatch of this beetle. Quite often fennel
tree woodpecker catches insects on the ground, and sometimes hunts tiny clambering
frogs living in leaf axils of fennel tree.
The nesting period at these birds begins at the end of April - the beginning
of May. At this time the male chooses dry last year's stalks of fennel tree,
and “drums” on them, involving the female. In clutch there are up to five round
eggs with white shell. Nestlings are feeding by both parents. Development of
posterity at this woodpecker occurs quickly, and for one year birds have time
to bring up two hatches.
When annual stalks of fennel tree die off (autumn - winter), the woodpecker
becomes “homeless”, justifying its second name. Then woodpeckers leave familiar
spots and migrate to woody areas of islands. They spend end of autumn, winter
and the beginning of spring in deciduous and coniferous woods living similarly
to “normal” woodpeckers. But at the end of spring when fennel tree grows anew
and stalks get stronger, homeless woodpeckers come back to native places, anew
hollowing out hollows in stalks of fennel tree.
This species of birds is discovered by Simon, the participant of forum.
Coconut
woodpecker (Cocopicus marinus)
Order: Woodpeckers (Piciformes)
Family: True woodpeckers (Picidae).
Habitat: coast of Indian ocean, from Southern Africa up to Jakarta Coast, through
Zinj Land, Arabia, India, Indochina; groves of coconut palms. This species does
not meet on Madagascar, in Australia and at the islands of Indian Ocean.

Picture by Alexander Smyslov
It is rather large representative of woodpeckers, the daw-sized
bird. The present species is the descendant of one of the African woodpecker
species. The bird has a constitution typical for woodpeckers. The basic colouring
is grayish-sandy with set of specks and strokes (tips of feathers on chest and
belly are dark). Wings of this bird are monotonous ash-grey. Similarly to many
other woodpeckers, this bird has red head contrasting with the general soft
colouring of feathering.
Voice of bird is sharp nasal cry. Males also “drum” on trunks of trees, involving
the female in courtship season. Coconut woodpeckers are settled birds, and also
do not make distant migrations. Besides these birds do not depart far to the
sea therefore they do not inhabit islands far from coast.
This woodpecker lives in groves of coconut palms at coast of Indian Ocean. It
eats insects pulling them out from under bark of coconut palms, sometimes gathers
any edible trifle at coast after storm, or hollows trunks of trees cast ashore,
searching for crustaceans and molluscs settling in them. Main and the most interesting
feature of this bird is the way of its nesting. The matter is that it rears
nestlings in … coconuts. Preparation for nesting begins from the flight of male
and the female to palm groves. They find the nut most attractive in their opinion.
They choose have not ripened yet and have not become covered by fibers coconuts.
Male and female hollow out an aperture in shell of nut, drink “coco milk”, and
scrape and throw out thin layer of pulp of the coco, yet not harden and looking
at this moment like jelly-looking substance. Sometimes they act even easier:
simply keep near to the chosen tree, protecting it. At this time in pulp of
coco larvae of insects settle and completely eat the rests of pulp from within
a nut. Woodpeckers occasionally check this coco, at the same time eating a part
of insects. When it becomes dry in coco hanging at this time on tree, birds
start laying and incubating of eggs. Eggs are hatched by the female, and the
male protects nest territory and brings food in nest. Almost all pairs have
time to hatch nestlings and to leave such “starling house” before it will fall
from a palm. But at some “losers” the nest falls from a top of palm earlier,
than the hatch leaves it. If the palm is not so high, and nut falls in soft
sand, nestlings can survive. In that case woodpeckers will continue to feed
them up, but already on the ground. In this case, to tell the truth, a significant
amount of young birds perishes, becoming catch of snakes or lizards.
This species of birds is discovered by Simon, the participant of forum.
Pine
pygmy woodpecker (Pinipicus ruber)
Order: Woodpeckers (Piciformes)
Family: True woodpeckers (Picidae)
Habitat: coniferous woods of the north and temperate zone of Eurasia, from Eastern
Siberia up to Scotland.

Picture by Alexander Smyslov
During an ice age the glacier some kilometers thick, as if
a plough, has ploughed the ground of Northern hemisphere, and is literally wiped
off coniferous woods of Northern Eurasia on which life of numerous species of
animals directly depend. Because of it Holocene crossbills (Loxia), known to
humans, were compelled to leave the most part of former area. At this time the
area of their inhabiting appeared broken off, both their number and variety
were reduced. Nevertheless crossbills have survived up to Neocene, but now they
live in other places, rather than 25 million years before in historical epoch.
In Northern Eurasia missed crossbills had been replaced by pine pygmy woodpeckers
– small birds a little bit larger than the sparrow. By appearance they are more
similar to any tropical exotic species, rather than to birds from cold areas
of Eurasia. These birds almost are entirely colored bright red color which has
appeared in their feathering because of carotenoids, containing in seeds of
coniferous trees which they eat. It is interesting, that in precisely same way
red colouring has appeared at completely other birds – the flamingoes; but they
receive carotene from crustaceans. The female of pine pygmy woodpecker is colored
duller than the male – it has orange color of feathering, only on stomach passing
to reddish-brown. Wings and tail are black at birds of both sexes. Also around
of eyes at males “glasses” of black feathers develop. On head of birds of both
sexes there is a mobile crest, by color the same, as colouring of feathering
on body of bird. Beak is rather long, but weak: pygmy woodpeckers can not hollow
wood.
Bright colouring of pine pygmy woodpeckers is warning one: the bird, certainly,
is not poisonous, but its meat has unpleasant bitter taste and resinous smell,
especially in winter. It is directly connected to diet of pine pygmy woodpecker:
it eats seeds of coniferous trees. Pygmy woodpeckers take seeds from under scales,
showing elements of acrobatic: the bird perches on pine cone from below, and
pushes beak under scales. In forage getting it is helped by exclusive flexibility
of neck. Less often pygmy woodpecker simply pecks out seeds from pine cones
similarly to other woodpeckers. At lack of forage pygmy woodpecker becomes less
legible in meal: it starts to eat berries and seeds of deciduous trees, and
at an opportunity pecks carcasses of frozen animals. Especially frequently pine
pygmy woodpeckers do it in spring when stocks of habitual forage are exhausted.
In summer when the staying seeds of coniferous trees ripen and are carried by
wind, pygmy woodpeckers eat mainly insects.
Outside of nesting season these birds live in flocks numbering up to 200-300
birds in every one. At this time pine pygmy woodpeckers are the nomades late
in places where it is a lot of cones. They rear nestlings in season when there
are more seeds of coniferous trees – in winter, similarly to crossbills. To
the moment of nesting flock splits up to separate pairs. Pine pygmy woodpeckers
do not hollow tree-trunk hollows – for this purpose at them beak is too weak.
These birds prefer to settle in natural cracks in trees, or in free tree-trunk
hollows which in the winter anyway stay empty.
In connection with nesting in the winter nestlings of pine pygmy woodpecker
already at the moment of hatching from eggs have rich grey down. Having dried
under the female, nestlings can not be afraid of cold, therefore pygmy woodpeckers
can settle in areas with sharply continental climate. In hatch usually there
are 3 - 5 nestlings. Parents rear them by seeds of coniferous trees, and add
insects to their diet. These birds frequently dig out ant hills by beak, gathering
slept ants, and also sound by beak cracks in tree bark, gathering insects hibernating
there.
Approximately in two months after hatching, in second half of winter, young
birds leave nest, and separate bird families gather to big flocks.
This species of birds is discovered by Simon, the participant of forum.
Gospodar
woodpecker (Regiopicus gospodar)
Order: Woodpeckers (Piciformes)
Family: Woodpeckers (Picidae)
Habitat: humid forests of southeast coast of Fourseas and Balkan.

Picture by Amplion
Warming of climate in Neocene had caused the formation of
productive ecosystems in places populated earlier with people and strongly changed
by their economical activity. It had turned so at the territory of Southeast
Europe, on coast of Fourseas – large continental reservoir with brackish water.
At Balkan and along southern and western coasts of this reservoir extensive
humid subtropical forests had grown up. Stability and efficiency of ecosystems
promoted occurrence of large species of animals.
In warm and humid forests of Balkan and Carpathian Mountains very large woodpecker
lives, being the original analogue of extinct in historical time large ivory-billed
woodpecker (Campephilus principalis) from forests of North America. This bird,
named gospodar woodpecker, is the descendant of one of widely widespread woodpeckers
of Eurasia, greater-spotted woodpecker (Dendrocopos major). The length of adult
bird including tail makes about 70 cm (hence its name – “gospodar”, the supreme
governor at some folks lived in historical time in Balkan region and near Black
Sea).
This bird is not too multicolor, but is colored contrastly. Sides of head at
this woodpecker are white; beak, forehead, top of head and nape are black, back
and back part of neck are also black. Covert feathers are black with white tips
which form striped pattern on folded up wings. Tail is formed of lengthened
rigid black feathers. As opposed to them forward part of neck and breast are
white. Red stomach at birds of both genders is a bright spot in strict black-and-white
colouring of bird. On head of male there is bright red cop of feathers; at the
female cop is brown-red.
Smaller species of woodpeckers are mainly insectivorous birds, but gospodar
woodpecker shows appreciable predatory bents. This bird eats not only large
wood-boring insects (grubs of capricorn beetles), but also ravages nests of
small birds and eats small mammals settling in tree-trunk hollows. Frequently
bats become its prey. This bird swallows small animals entirely and kills larger
ones by impact of beak and pecks, having hooked in fork of branches. In winter
this bird also eats seeds of various trees – chestnuts, oaks and horse chestnut.
Gospodar woodpecker almost never goes down on the ground, and spends entire
life in forest canopy and on trunks of trees.
Gospodar woodpecker is exacting enough relatively to habitats – this bird settles
only in old wood where there are high trees, suitable hollows for hollow arranging,
and old trees bored by insects.
This species of birds settles in pairs keeping to all further life. The pair
of birds makes for nesting a deep hollow – its depth is up to one and a half
meters, with width of entrance about 20 cm. At the bottom the hollow extends
to rounded nest chamber about 40 – 50 cm width. Gospodar woodpecker is strictly
territorial one – each pair of birds in common protects the extensive territory
(more than 10 square kilometers) from relatives. Birds notify congeners about
the rights to territory by loud sharp cry, especially in the morning. Both birds
of pair cry in common and continuously during 15 – 20 minutes.
The pair of birds starts to peck hollow for nesting at the end of winter. In
same time birds begin to cry long and loudly in the morning, warning congeners
that the territory is occupied. Construction of nest takes about two weeks in
total. Nesting begins in early spring, right after hollow making. During courtship
games birds chase each other, cry loudly and display bright cops to each other.
Nesting at gospodar woodpecker takes place only once a year, and in years poor
in food birds can not nest at all. In clutch at this species it may be 2 – 3
eggs; they are hatched only by the female. Male feeds her and protects territory
alone. The female leaves the nest only for a short while. The hatching lasts
about 25 days. Nestlings come into the world naked and blind. They are brought
up by both parents during two months. For this time young birds fledge and leave
a hollow. Later parents feed them till about two weeks, and then start to show
aggression to their own posterity. Young birds settle at edges of territory
of adult birds and in young forests. They start breeding at the age of three
years. Life expectancy of this woodpecker reaches 30 years.
The pair of these woodpeckers seldom settles in the same hollow longer than
2 years. On a twist of fate after woodpeckers their hollow is more often occupied
by bats which frequently are prey of these birds. Occasionally woodpeckers visit
old hollows at their territory, attack bats settling in them and eat their young
growth which can not escape from them.
Termite
woodpecker (Neogeocolaptes termitophilus)
Order: Woodpeckers (Piciformes)
Family: True woodpeckers (Picidae)
Habitat: the south of Africa, Cape province.

Picture by Alexey Tatarinov
In Neocene woodpeckers had developed ecological niches new
to this group of birds. Among them predatory
birds eating small animals had evolved. Largest woodpeckers of Neocene are
scavengers in savannas of Africa. At Great
Antigua island flightless woodpecker species lives,
and some more kinds live in mangrove thickets and
feed in reef zone near to the coast. However, such
changes in habit of life represent a rarity among woodpeckers. The majority
of species living at the continents have kept adherence to former way of life
and diet consisting of insects.
One more species of woodpeckers had partly broken with habit of life typical
for these birds lives in savannas of South Africa – it is termite woodpecker.
It is the descendant of ground woodpecker (Geocolaptes olivaceus) lived on the
Earth in human epoch. It had moved from mountain areas, where its ancestor lived,
to plains, and due to expansion of semideserts and savannas territory in ice
age had it easily survived in biological crisis epoch.
The descendant of ground woodpecker has kept many features of the ancestor,
but this bird hollows not trees, but termitaries with the help of strong beak.
Termite woodpecker easily breaks firm walls of termitary, and licks off insects
by long tongue. Termites are very common in savanna, and termite woodpeckers
are usual birds of plain areas. These birds are very cautious, and their loud
cry is heared far in savanna. Therefore termite woodpeckers render very important
service to herbivores, warning them about approaching of predators. Besides
the pair of birds of this species cries in the morning for a long time, designating
claims to the territory.
Termite woodpecker has very imperceptible brownish-grey colouring. On wings
and back there are numerous dark spots, and on waist feathers having metal shine
and bright red coloring grow. When this bird flies up, these feathers are well
visible, and serve as an alarm signal for many animals. Male and female have
some distinctions in colouring - at male’s head from ear up to ear across the
nape a cross strip of white color stretches. The plumage of termite woodpecker
is rigid, and it protects the bird from stings of insects.
This bird species nests in original way – it hollows out nesting chambers right
in termitary. During the hollow construction birds eat, seizing termites creeping
out for protection of the colony. Gradually they expand hollow and form the
nesting chamber in it. Damages, caused by woodpeckers, are so great, that termites
simply close up entrances in destroyed galleries, and woodpeckers need only
to level a surface of nest walls. The tunnel leading to nest chamber is abruptly
bent at the surface and is directed downwards to one meter deep. Such nest appears
strong and unapproachable, and any predator trying to destroy it will be attacked
by hundreds of termite soldiers. It is interesting, that the pair of birds does
not feed in termitary where their nest is arranged, and actively protects it
from congeners.
For one year, during the rain season, these birds may rear up two hatches. Female
lays up to three eggs and hatches them within 18 days. Fledglings leave nest
at the age of 35 days, and parents feed them within several days after it.
Wasp-eating
woodpecker (Vespicus auratus)
Order: Woodpeckers (Piciformes)
Family: Woodpeckers (Picidae)
Habitat: South America, rainforests.

Picture by Pavel Volkov
Rainforests of South America represent the habitat of numerous
species of insects – from herbivorous up to predatory ones. Representatives
of hymenopters order, solitary and social species of wasps, are very diverse
in canopy of South American forests. Some species of these insects became very
dangerous inhabitants of forest – for example, such one is predatory corsair
hornet (Toxivespula flammeocarbunculus) which is capable to attack any animal
living in the neighbourhood. However in nature there are various species of
birds, adapted to eat stinging insects and to ravage their colonies. Such birds
live in Africa – they are beekeeper parrot
(Melipsitta melivora) and waxeater (Cerophaga
spp.). In Europe crested wasp burglar
(Pappopernis fur-vespinus) is their ecological analogue. In South America
stinging insects become prey of a special kind of woodpeckers – wasp-eating
woodpecker.
This species of birds is adapted to feeding on insects well protected from enemies
– ants and wasps. Due to the adaptation to such diet it almost lacks competitors
in tropical forest.
The shape of wasp-eating woodpecker is typical for representatives of its family.
It is a small bird – its length does not exceed 20 cm including tail. This bird
does not have cop, only feathers on the top of male’s head are lengthened a
little. Wasp-eating woodpecker is very beautiful – it has straw-coloured body
feathering, white “face” with black specks at the basis of beak, and bright
red “cap”. For protection against insects feathering of this woodpecker is very
dense, and tips of feathers around of eyes are bristly.
The couple of birds occupies the vast site of forest where there is enough of
colonies of social insects on which wasp-eating woodpeckers are feeding. Birds
designate rights to the territory with the help of loud calls well heard from
apart.
Attacking on insects, this woodpecker uses an element of surprise. Bird feeding
lasts not for long, but at this time the bird works very quickly. For some seconds
woodpecker punches an aperture in cover of wasp nest by strong impacts of beak
and pulls out their larvae from honeycomb cells by long tongue. If bees or wasps
construct open honeycombs, bird hangs on them with the help of tenacious paws
and quickly pulls out the largest larvae. By tip of tongue woodpecker defines
larvae by their movements and consequently pupae of insects lying in closed
cells remain without its attention. Ant colonies in forest canopy, as a rule,
exist inside hollow stalks or petioles of myrmecophilous plants forming the
chambers right for this purpose. Also ants settle in wood, gnawing burrows through
it, or using already existing ones. Woodpecker gets ants the same way, as any
other insects, obviously preferring their larvae and pupae. Wasp-eating woodpecker
has very proof immunity to poisons of insects – bird can easily endure a doze
of poison enough to kill a mammal weighting up to 50 kg.
The couple of birds nests in a hollow which they make in tree trunk. In clutch
there are up to 4 eggs hatched mainly by the female. During one year birds have
time to make two clutches. Young birds are similar to adults, but “cap” on their
head is not red, but black. Young birds start to nest at the age of about 8-9
months.
Cereus
woodpecker (Leuconotopicus mexicanus)
Order: Woodpeckers (Piciformes)
Family: True woodpeckers (Picidae)
Habitat: cactus “forests” in the south of North and North Central America.
Woodpeckers are one of the most specialized and recognizable orders of birds.
Their way of life and behavior differ from other small forest birds strongly
enough that woodpeckers could live almost all over the world without being particularly
different from each other externally. But in Neocene epoch, species with very
specialized behavior appeared among them.
Most Neocene woodpeckers have retained ancestral skills, although a number of
species, especially in Africa and Asia, have become predators
or scavengers (they
are classified as a separate family). But most woodpeckers remained faithful
to their habitual way of life, changing only certain features. Cereus woodpecker
is one of these species. It is an ecological “heir” of Gila woodpecker (Melanerpes
uropygialis) of Holocene epoch, although it is a representative of another woodpecker
genus.
By the standards of woodpeckers, cereus woodpecker is a typical representative
of this group of birds, about 20 cm long. Its back and wings are colored with
brown and white cross-stripes; male has a red-brown cap on its head, belly is
pure white. The appearance of this species is typical for woodpeckers, but the
paws on the underside are covered with pads of keratinized skin; on the belly
under the feathers there is also an area of keratinized skin that protects against
pricking of cactus spikes.
Beak of cereus woodpecker is long and pointed, but the skeleton and musculature
are proportionally less powerful than those of other woodpeckers – this bird
makes its hollows not in trees with hard wood, but in large tree-like Cereus
cacti with a sappy soft core. Birds spend much less effort on the arrangement
of the hollow, and after them, other birds – owls, wrens and small parrots –
use their abandoned hollows.
The courtship season of this species falls at the end of the dry season – males
warn about their rights to the territory and call for females by drumming. For
this purpose, male uses a tree with hard wood, drumming with its beak on resonating
branch. When the pair has formed, both birds make a new (or update the old)
hollow in a large and tall cactus. There the female lays 3-4 white eggs and
both parents incubate them in turn for about 2 weeks. After another 3 weeks,
the juveniles are on the wing. Most often they stay with their parents until
the beginning of new rains and look for food in the same group, leaving their
parents before the new nesting season. This woodpecker is monogamous and often
forms pairs for life.
Cereus woodpecker has few enemies: these are mainly local birds of prey. It
is mainly nestlings that die from snakes, and nesting on cacti protected by
spikes saves these birds from predatory mammals. More often these woodpeckers
die from lack of food or from diseases in especially dry years.
The lifespan of this woodpecker is up to 20 years.
This bird species was discovered by Bhut, the forum member.
Tortoise
woodpecker (Testudopicus rubrocephalus)
Order: Woodpeckers (Piciformes)
Family: Predatory woodpeckers (Carnopicidae), a subfamily of true predatory
woodpeckers (Loricoflangerinae), “armor crushers”.
Habitat: coast of former Mediterranean sea – Asia Minor, Balkan, Appenines,
Pyrenees, Atlas mountains.

Picture by Amplion
One more species of the carnivorous woodpeckers had appeared
on Earth in Neocene. It is one of large species of woodpeckers: the bird is
crow-sized. It has rather short rounded tail and strong four-toed legs. The
bird is not able to walk, similarly to all woodpeckers, and moves on the ground
by jumps. The tortoise woodpecker has spotty colouring: on grey background there
are chestnut spots, head is entirely ruby-red (at females it is dimmer, than
at males). The beak at birds of both sexes has almost white color.
But not so much colouring, as food predilections of this species had determined
the name of bird.
Usually tortoise woodpecker eats various insects. But it does not hollow trees
in searches of forage, and simply pecks from the ground beetles, large grasshoppers
and other big insects. But meeting the tortoise or large snail bird applies
completely another way of food getting. It presses snails by paw against the
ground and, having broken their shells by one or two exact beak impacts, swallows
meat, having shaken from it splinters of shell. Attacking tortoises, it perches
on their carapace and orderly pecks it. The usual tactics of tortoises against
such attack (to hide in armor and to wait) does not rescue them. Even tortoises
belonging to species closing head and paws by parts of carapace, cannot feel
like protected from this bird. Eventually, the woodpecker punches carapace,
kills the reptile, and regales itself with its meat. The attacked turtle has
three ways of rescue: to creep near to animals which may frighten off a woodpecker
from its back, to creep in dense prickly bush, or to dive into water. Reservoirs
in savanna meet not so frequently, but it is the best way to protect from this
bird. Usually, if the tortoise creeps through bushes, the woodpecker watches
it, and attacks in a right moment. Some water turtles having long neck, can
bite a woodpecker, having contrived – but such way of protection is effective
only against young and inexperienced birds.
Tortoise woodpeckers are monodins, but pairs at them are formed only for the
nesting period. These birds nest in tree-trunk hollows hollowing them out, or
in cracks of rocks (in hilly terrain or in desert). The tortoise woodpecker
is relatively strong, and can break skull of sheep-sized animal; therefore predatory
animals avoid attacking this bird. If the predator tries to ravage nest, woodpeckers
at first warns it by cries (the voice of this species sounds as lingering “dry”
creaking warble), and then attacks it, striking impacts in its head and nose.
Woodpeckers sometimes can be predators, attacking rodents and small birds. They
stun and kill catch by strong impacts in head by beak. However, more often these
woodpeckers do not attack mammal and birds, preferring to them sluggish tortoises
and snails which cannot protect themselves actively. Usually these birds attack
small animals during nesting and at shortage of the basic forage.
In clutch of tortoise woodpeckers there is up to four white eggs. The posterity
hatches after three weeks of incubating.
There is close species of woodpeckers: crab
woodpecker (Testudopicus cancriphagus). Its ecology appreciably differs
from those at tortoise woodpecker. This species lives in mangrove thickets.
Its area is in separation from an area of tortoise woodpecker: it includes southern
and southeast coast of Eurasia – the Jakarta Coast and coast of Bay of Bengal.
This bird is similar to tortoise woodpecker in size, but strongly differs from
it in colouring. The crab woodpecker has monotonous ash-blue color of stomach
and neck, and its wings of sand color. Head at this species is also red, but
beaks at the male and females differ in coloring: at males beak is white, and
at females black. This species eat beetles and snails numerous in mangrove woods
therefore this bird it is not so aggressive and territorial like its desert
congener. This species is not interested in tortoises at all: in mangrove thickets
turtles live in water, and because of it are absolutely inaccessible to this
woodpecker. But the bird actively hunts crabs – from small, coin-sized ones,
up to huge mangrove robbers. By strong beak the crab woodpecker punches their
armors and pecks meat. But only largest and skilled birds hunt huge crabs because,
as against phlegmatic turtles being a food to tortoise woodpeckers, the crab
can catch and eat the inexperienced hunter itself.
Crab woodpeckers are monodins, forming pairs to all life, but reunite only during
nesting. Outside of nesting season birds feed at different edges of the common
territory, occasionally exchanging by cries.
Nesting of crab woodpecker occurs twice per one year. Nests are located in tree-trunk
hollows; birds hollow them out themselves, or expand available ones. By nesting
features this bird resembles tortoise woodpecker.
This species of birds is discovered by Simon, the participant of forum.
Egg
woodpecker (Ovipicus oviraptor)
Order: Woodpeckers (Piciformes)
Family: Predatory woodpeckers (Carnopicidae), subfamily of egg woodpeckers (Ovipicinae)
Habitat: Northern Africa, the Central Asia, not numerous populations exist in
Arabia. Everywhere this bird inhabits open landscapes with small sites of wood.
Picture by Alexander Smyslov
It is one of smallest species of family - a sparrow-sized
bird. The egg woodpecker is colored yellowish-white colors with dense brown
cross ripple. On wings there are dark edges of feathers forming scaly pattern.
On head of this bird the mobile pointed red crest grows.
The food behavior of egg woodpecker combines primitive features characteristic
for usual woodpeckers with features of extreme specialization to other, than
at other species of family, diet. Usually this bird eats various insects, getting
them from under bark as any other woodpecker. But it feeds so only half-year.
Other half-year, in nesting season of other birds, it eats their eggs.
It simply steals eggs of smaller birds from nests, breaking them by beak and
drinking out the contents. But it also can ravage even clutches of large birds,
up to huge giraffe ostrich, the largest feathered creature of ever living on
Earth. In this case it acts other way: while nest owners have left, the bird
perches on thick-walled egg, and, having caught by claws and supported by tail,
starts to hollow it by beak, as if it is the wood bark. Some times not one,
but some birds can peck a large egg together. When the egg-shell is broken open,
birds start to drink its contents, using long tongues. If in egg the embryo
is developed, and the egg-shell is broken, these small woodpeckers can peck
the embryo.
In Africa egg woodpeckers peck thus eggs of gallinaceous birds and giraffe ostriches,
in Arabia – garudas (local bird descended from gallinule), in Asia – ostrich
rails. In Africa the egg woodpecker has competitor – egg-eating drillsnake,
the specialized representative of reptiles. But their food predilections are
crossed in minimal degree: the woodpecker can eat eggs of wood birds, but the
eggs of turtles buried in sand are inaccessible to it. Therefore special rigid
competition between bird and reptile is absent. The special loss to slowly breeding
large birds from these woodpeckers is not present - in each clutch only 1 -
2 eggs are eaten, and sometimes feathered robbers pass nest of these birds by
if the family is amicable and can provide constant protection of nest.
The egg woodpecker nests in tree-trunk hollows, not changing “traditions” of
ancestors. These birds are monodins, forming pairs till one nesting season.
The female hatches out eggs and nestlings, and the male searches for food at
this time. Life cycle of these birds is interesting: eggs and nestlings appear
when at small passerines in eggs embryos start to develop. It is connected by
that it is difficultly to carry to the nest liquid contents of eggs for feeding
of nestlings. Therefore the female and nestlings of this woodpecker feed with
meat of embryos of birds: male pecks eggs of other birds, orderly ravaging near
nests one by one. This woodpecker has excellent memory, and it can remember
an arrangement up to three tens nests of various birds placed in its territory.
This species of birds is discovered by Simon, the participant of forum.
Laughing
seriema (Barocariama patagonica)
Order: Cariamiformes (Cariamiformes)
Family: Cariamidae (Cariamidae)
Habitat: Southern Patagonia, pampas.
South American seriemas were able to survive the epoch of human domination,
losing only part of their range, but retaining a sufficiently high number necessary
for survival. It was much more difficult for them during the ice age, when cold
and glaciation caused the displacement of vegetation zones, and pushed them
to lower latitudes. But part of the seriemas managed to adapt to the more severe
conditions of the ice age; after the retreat of the glaciers, these birds migrated
south to the conditions of the habitual climate. This is how the laughing seriema
appeared, a migratory species of the south of Patagonia, a descendant of the
red-legged seriema (Cariama cristata). The spread of this species to the north
is hindered by the presence of large flightless hunting
herons in the pampas, which are partly food competitors of this species.
In the appearance, laughing seriema resembles a bustard. The bird reaches 120
cm in length (including tail) with a weight of about 13-15 kg. But at the same
time, laughing seriema looks quite fleshy and stocky compared to the ancestors
of the Holocene epoch. The plumage color is brown with mottled spots, more numerous
on the back and wings. A tuft of narrow cream feathers grows on the head, in
males it is larger – it is the only feature of sexual dimorphism. The head is
elongated; the beak is more powerful than that of the ancestral species, reddish-brown
in color.
This bird species is omnivorous with a predatory bias. The bird eats seeds and
tubers of herbaceous plants, which make up about a quarter of the diet by weight.
All other food is of animal origin: it prefers rodents, reptiles, amphibians
and insects. Occasionally, the laughing seriema preys on young ungulates and
cursorial rodents, striking them with its feet and beak. At this time, the bird
is careful and attacks only when the adult animal has gone too far, and the
attack is relatively safe. It is wary of kicks from adult animals that can damage
its bones. Laughing seriema is able to fly, but does it poorly and reluctantly.
It flies up on a separate tree or termite mound, only if the predator persistently
pursues it; usually it moves on the ground. In mid-autumn, laughing seriema
flies north to Grand Chaco.
The breeding season begins shortly after the completion of the spring migration
of these birds – in October-early November. At this time, the males’ call is
audible through the pampas – a loud frequent call similar to laughter. Males
first choose several places in the bush, where they build the bases of the nests.
Females, in their turn, very carefully inspect the place and the nest base.
At this time, the male walks next to the female, stamps his feet and raises
his crest, displaying his physical condition. If two males claim the same female,
they can engage in a duel, during which they vocalize, stamp their feet and
jump on each other, pushing with their breasts. The female usually goes to the
winner, although she can leave his territory if she does not like the place
for the nest. The male can also continue his display, having arranged a new
base for the nest.
There are usually 2 elongated eggs in the clutch, covered with chocolate spots
and reddish-brown markings. The female incubates; the male guards the nest and
the territory around. After 25-26 days, the chicks hatch. In case of danger,
the female covers the nest until the last, then suddenly jumps out to meet the
enemy, loudly flapping her wings. Sometimes she attacks the enemy by kicking
and clinging to him with her beak. If she leads the chicks, then in case of
danger they hide, huddling to the ground. Sometimes the female simulates the
wound, pretending to have a sore leg, and leads the enemy away from the nest.
At the same time, the bird constantly changes the direction of movement, takes
off and flies low above the ground. After 2 weeks, the chicks leave the nest
and roam with their parents. In case of danger, the parents take the predator
away or attack him, and the chick at this time sits, pressed to the ground,
and does not move until the mother calls it. The feeding of chicks by adults
continues for about a month, and then the young bird just stays close to its
parents, searching for food independently. Young birds get adult plumage at
the age of 4 months, after another month they are completely independent. By
this time, a pair of adult birds breaks. At the age of 2, the laughing seriema
reaches sexual maturity. Life expectancy is about 40 years.
This bird species was discovered by Nick, the forum member.
Utiaritimirim,
or dwarf kestrel (Falco nanus)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: plains of South America.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The American kestrel (Falco sparverius) had survived
in South America, where it gave rise to the utiaritimirim, the dwarf kestrel.
His name is formed by two words: utiari, a name given by the Pareci people of
Rondônia to the bird’s ancestor, and by mirim, which means small in Guarani.
The utiaritimirim is a relatively common bird of prey, inhabiting the open areas
of South America. Birds that live in Southern areas migrate to the savannas
in the center of the continent during the winter.
The utiaritimirim, having between 14 to 18 cm of length and a wingspan of 40
cm, can be considered one of the smallest species of falcon in the Neocene.
Like other birds of prey, the female is larger than the male. Beyond the difference
of size, their plumage also show sexual dysmorphism. The male is reddish-brown
in the crown and back, with a dark brown band between it and the white rump.
The wing coverts and tail are also dark brown. The females have dark brown crowns,
back, tail and wing coverts. Both sexes have white faces, but the male has a
black malar strip that is absent in the female. The wing feathers are black,
while the chest is cream with dark marks. The tail has white bands. Both the
beak and legs are yellow.
The species feeds mainly on insects and smaller birds. The proportions of insects
and birds in the diet may vary according to availability. They are great hunters
of dragonflies, attacking the insects with great agility, but it’ll take other
insects such as bees, butterflies, grasshoppers, and beetles. The utiaritimirim's
primary mode of hunting is by perching and waiting for prey to come near. It
also hunts by kiting, hovering in the air with rapid wing beats and scanning
the ground for prey. Other hunting techniques include low flight over fields,
or chasing insects and birds in the air. Before striking, the kestrel characteristically
bobs its head and tail, then makes a direct flight toward the prey to grab it
in its talons.
Utiaritimirins are sexually mature by their first spring. In migratory populations,
the males arrive at the breeding ground before females, then the female selects
a mate. Pair bonds are strong, often permanent. Pairs usually use previous nesting
sites in consecutive years. This gives birds an advantage over younger or invading
individuals, as they would already be familiar with the hunting grounds, neighbors,
predators, and other features of the site. Males perform elaborate dive displays
to advertise their territory and attract a mate. These displays consist of several
climbs and dives, with three or four "kirií" calls at their peaks.
Females are promiscuous for about one to two weeks after their arrival at the
nesting site. Food transfers from the male to the female occur from about four
to five weeks prior to egg laying to one to two weeks after.
These birds are cavity nesters, but they are able to adapt to a wide variety
of nesting situations. They generally prefer natural cavities with closed tops
and tight-fitting entrances that provide for maximum protection of the eggs
and young. However, they will nest in holes created by large woodpeckers, or
use the abandoned nests of other birds, such as those made by the white-throathed
obrero or other ovenbirds. The obrero tolerates the presence of these birds
of prey in their colony because utiaritimirins will repel any predator that
tries to approach their territory, this way protecting the other birds that
are nesting near it. Even if they are small, these kestrels are aggressive and
can cause wounds with their sharp talons.
Three to seven eggs are laid approximately 24–72 hours apart. The eggs are white
to cream in color with brown or grey splotching. Incubation usually lasts 30
days and is mainly the responsibility of the female, although the male incubates
for some brief periods of time. Eggs that are lost are typically replaced in
11 days. Hatching takes place over three to four days. Hatchlings are altricial,
and are only able to sit up after five days. They grow rapidly, reaching an
adult weight after 14 days. After 28 days, their wings have developed and they
are able to leave the nest. The young adult utiaritimirim may breed from a year
old, and the species has approximately a lifespan of six years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Crested
singing falcon (Canorofalco cristatus)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: Atlantic forests of South America.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The laughing falcon (Herpetotheres cachinnans) was a
widespread species that lived in mostly of tropical South America. It had survived
the degradation of the environment by humans and gave rise to the genus Canorofalco,
the singing falcons. Its members are restricted to humid tropical and subtropical
forests, with the type species, the crested singing falcon, inhabiting the Atlantic
forests of Northeastern and Southeastern South America.
The crested singing falcon has 53 cm of length and a wingspan of 84 cm. Like
most birds of prey, the female is heavier than the male. The adults have a gray
back and wings. The breast and belly are white. The head has a broad black face
mask that stretches across the neck as a narrow collar, bordered with white.
They have a cinnamon crest that can be erected when they are excited or trying
to intimidate an adversary. The tail is black with a white tip and a white band
in the middle. In flight, they have a shape closer to a hawk than most of its
falcon relatives, with short, rounded wings and a long tail. The legs and beak
are yellow.
This species feeds primarily upon small birds, mammals (mainly rodents and marsupials)
and squamates. They often hunt prey by sitting quietly on tree branches and
waiting for their victims to appear. When the prey arrives, the crested singing
falcons quickly ambush them, attempting to catch them with a brief, flying pursuit.
As their name says, the crested singing falcon is a very vocal bird. Their vocalization
can be heard during the entire day, with pairs doing duets. Nesting territories
are occupied year after year. They do not build a nest, but lay their two or
three white eggs in cavities in trees. Laying occurs mainly late in the dry
season, with hatching taking place at the onset of the rainy season, a time
of increasing prey abundance. Eggs hatch 34 days after being laid, and nestlings
fledge 42 days after hatching. Fledglings disperse from their parents' territories
within four to seven weeks after fledging, achieving independence at that time.
They reach sexual maturity with three years and have a lifespan of 18 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Cuauhtli
(Caracara cuauhtli)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: Central America, Cuba, Greater Antigua.
Birds of prey of the order Falconiformes, like specialized carnivorous mammals,
were experiencing a serious crisis by the beginning of the Neocene: large species
disappeared due to habitat destruction, disturbing and direct persecution by
humans. However, a number of smaller and less specialized species survived,
and their descendants managed to restore the number and keep the niche of feathered
predators for themselves. The crested caracara (Caracara plancus) turned out
to be one of the largest successful feathered predators. Widespread and feeding
on a variety of prey, it not only survived the anthropogenic crisis and the
beginning of the ice Age: its descendants gave a range of new species, even
conquering Antarctica. In Central America,
having crossed the Panama Strait, cuauhtli – a large descendant of this species
– settled.
The body length of an adult is 71-96 centimeters (the female is larger than
the male; the species name means “eagle” in Nahuatl language of Aztecs), the
wingspan is about 168-224 centimeters, the weight is up to 4.5-5.5 kg. The bird
has a recognizable appearance, preserving the features of the ancestor’s anatomy.
A very characteristic feature of the species is a crest of elongated feathers
growing on its head, which the bird raises when displaying aggression. The background
color of the plumage is brown with faint ripples on the wings; the feathers
on the head are almost black with a greenish tint. There is no plumage on the
face and around the eyes; the naked skin is dark red. The legs are elongated,
with bare tarsometatarsus and toes, covered with bright yellow skin.
Cuauhtli is a quite widespread species – from Florida in the north to the Panama
Strait in the south, as well as on the large islands of the Caribbean. This
species inhabits a variety of landscapes – from open spaces to mountains and
tropical forests. Perhaps only in the Mexican Plateau these birds appear only
occasionally – it is too arid climate for them there and they “bypass” it along
the mountain ridges. It feeds on large birds and rodents; sometimes it can kill
a sick or old deermara or steal its cub. Birds fly well, but they can chase
small prey on the ground.
This is a territorial species that controls a feeding area of considerable size:
up to 100 square kilometers per pair. Detected rivals are immediately expelled;
an exception is made only for the breeding partner – the pair owns a more extensive
territory than a single bird. If one of the individuals in a pair dies and the
second bird does not find a mate, neighboring pairs of birds can expand their
possessions at the expense of a widowed bird.
The mating season begins early – in January-February. During courtship, birds
perform aerobatics in the air; the most characteristic among them is a “somersault”
with geared claws – during a somersault in the air, one bird holds another.
For an emerging pair, this is a test of the partner’s physical condition, for
an established pair, it is a strengthening of the bonds between the birds in
the pair.
A cup-shaped nest is built of large branches and twigs on top of a tall living
tree or on a rock, usually with a good view of the surroundings. One pair can
keep several nests on its territory, usually 3-4, nesting in them alternately.
In this way, birds get rid of the excess of parasites that settle in the nest.
There are 1-3 (most often 2) eggs in the clutch, which the female lays at intervals
of 2-3 days. In years rich in food, the female feeds all the chicks, although
more often one of them kills the others. Aggression is provoked by hunger and
the absence of adults in the nest for too long. Incubation lasts 40-45 days;
this is mainly done by the female, although the male can occasionally replace
her on the nest. The growth period is about 60 days. In May, young birds are
already flying, foraging with their parents. In October-November, they are already
completely independent and their parents expel them from the territory. By the
age of 2, the young cuauhtli already owns its territory, but many young birds
do not survive the first months of independent life – they become victims of
predators and aggression of adult relatives defending their feeding territories.
A significant part of young birds die of starvation. Birds begin breeding from
the age of four or five years; life expectancy is 35-40 years.
This bird species was discovered by Nick, the forum member.
Thick-billed
caracara (Crassirhynchus apiphagus)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: tropical forests of South America.
During the Holocene the humans caused great alterations in the ecosystems of
Earth, from the destruction of habitats to the introduction of exotic species.
Africanized bees were introduced in South America to produce honey; however
these insects escaped and spread across the continent. It prospered and in Neocene
its descendants evolved. At
the same time, animals evolved to prey on it. The thick-billed caracara is one
of these animals, a descendant of red-throated caracara (Ibycter americanus).
This bird lives in the tropical forests of South America.
The female of this species is larger than the male, she is 56 cm long with a
wingspan of 78 cm while males grow to 50 cm length and have a wingspan of 71
cm. Both sexes have black plumage with white undertail feathers. Their throat
is not bare like that of their ancestors, but the region around the eyes is
exposed, revealing their red skin. Their feet and legs are strong and yellow.
The grey-colored beak is thick and strong.
The diet of this bird consists mostly of bees, wasps and their larvae. Ants,
termites, other insects and fruits are also eaten. When hunting wasps, thick-billed
caracara makes an aerial-diving attack to knock nests down onto the forest floor,
while skillfully evading most wasp stings. The birds use air squadron precision,
repeatedly diving then scooping upward, to drive off or confuse angry defender
swarms around the hive. While doing it, it can catch adults and crush them with
the beak. Its strategy to hunt bees is different: it excavates the hive using
its thick bill and strong legs. When the defender bees start to attack, thick-billed
caracara makes fast aerial maneuvers to confuse and catch the bees. These birds
are also resistant to the bees poison, being capable to be stung many times
without suffering damage. Sometimes bee-eater
tanagers follow thick-billed caracara to attack and eat the adult insects
that escape. Thick-billed caracara also follows jaguapes
to search for bee hives; this way the birds can have an easy way to catch the
insects.
Thick-billed caracara is monogamous bird forming pairs for life. Its lives in
family groups, with the parents and their young, which help to take care of
the next generation of chicks. The nest is made of twigs and branches high in
the trees. The female lays 3 to 5 white eggs, which are incubated by both parents
and older siblings for 30 days. The chicks fledge at the age of 33 days. These
birds reach sexual maturity in 4 years, but can stay with the family group until
their sixth year. The lifespan of thick-billed caracara is about 28 years.
This bird species was discovered by João Vitor Coutinho, Brazil
Antarctic
carancho (Carancho antarctophron)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: Antarctica, wintering in South America.

Picture by Alexander Smyslov
At the end of human era the falcon family sustained heavy
losses: large species of true falcons disappeared because of destruction of
their habitats, disturbance and direct hunt from the man’s side. But in New
World other representatives of family – caracaras – survived and even reached
certain prosperity. After human disappearance they expanded an area and became
among the main feathered predators in new ecosystems. In South America caracaras
live at the South of the continent, in the field of temperate climate. And it
was the important circumstance which further had allowed these birds to occupy
the new continent, the Antarctica.
Meadows and bushes of Antarctica are occupied by the separate species of caracaras
– by antarctic carancho. This bird is similar to South American caracaras from
which it descended, but differs in number of unique features.
Wingspan of antarctic carancho reaches one and a half meters, and weight of
adult bird is about three kilograms. In cold climate these birds have denser
and thicker plumage in comparison with ancestors. Antarctic carancho has ash-gray
color with white primarily feathers and black “hat” on the head. Tips of tail
feathers are also black. The winter plumage is much denser than summer one.
The skin area near eyes and in the base of beak is covered with small feathers.
Some extended feathers on nape form a kind of crest. Their fans are strong untwisted,
and cores of these feathers are very elastic.
Legs of these birds are long, with rather short toes. The distinctive feature
of antarctic carancho is that feet of this bird are feathered almost to toes
(but toes are naked). Toes are rather short, and claws are blunt, therefore
bird can't tear prey using them. Antarctic carancho walks on the ground good
and for a long time and often looks on the ground for prey – small animals and
carrion. In spring after rivers overflow antarctic carancho explores the pools
remained in flood plains of the rivers, and catches the small fish had got to
these traps. Also these birds settle near colonies of sea birds and gather dead
chicks or the prey dropped or belched by adult birds.
The head of this bird is more extended in comparison with ancestors. Beak is
short, of black color, very sharp, with cutting edges and the hooked tip, adapted
for tearing of skin of large animals. Antarctic carancho is the main scavenger
of Antarctica and often wanders on an ocean coast, searching and eating large
fishes and other sea animals that had been cast ashore by storm.
The development of sense organs in this species is peculiar: as at New World
vultures, at antarctic carancho sense of smell is well developed. It is connected
with need of search of food: in the conditions of Antarctica ascending streams
of the air, allowing soaring at great height, aren’t formed. Therefore birds
are compelled to use other ways of search of food, and the main thing among
them is the recognition of smells. Due to the secondary development of olfactory
parts the head of bird has more extended shape, rather than at related species
from South America.
Pairs at antarctic carancho are formed for one season of nesting. Courting begins
in the spring when snow thaws and the grass begins the growth. The male chooses
a place for nesting – on the ground among bushes. It scatters the ground away
by feet, forming the nest basis, and pulls down in a being formed pole a last
year’s grass. During the courtship display male loudly claps by wings and makes
some circles over the nesting territory, uttering calls similar to crash. When
the female flies to examine the territory of the male, he displays himself to
her, walking up and down round the female with the semi-opened wings and the
raised crest. From time to time the courting male gives a blade of grass to
the female, and then puts it back to the nest. If the pair was formed, partners
in common complete the nest. In clutch at this species there are two eggs. Parents
bring up posterity in common, but more often they manage to grow up to leaving
the nest only one chick during the season. If the clutch or chicks are lost
for any reasons, birds don’t nest in the current year, and the pair breaks up.
Young antarctic caranchos differ from adult birds by one-colour gray plumage
of more dark shade, than at adult birds, and by the lack of black marks on plumage.
Only the corneous cover of beak is distinguished against their plumage background
color. The next year young birds gain the color of plumage typical for this
species. The sexual maturity at these birds comes at one-year-old age though
birds older than two years nest more successfully. Life expectancy of this species
makes till thirty years.
Antarctic carancho is a migrating bird. However, it flies to wintering places
late enough, with the first snow and even at the appearance of steady snow cover.
These birds cross Drake Strait and migrate to the north along the Pacific and
Atlantic coasts of South America. Many birds stay to winter on Tierra del Fuego,
and separate individuals move to Falkland Islands.
The idea about existence of this species was proposed by
Simon, the forum member.
Translated by Alexander Smyslov.
Caracaraptor
(Caracaraptor inexpectatus)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: California Island.

Picture by Pavel Volkov
The evolution of certain groups of living organisms on islands
often makes unexpected moves and may go in a different direction than on the
continents. In the Neocene, the evolution of island animals demonstrates the
same patterns as in the era before human colonization. A characteristic feature
of the fauna of the islands is a presence of flightless birds, and even birds
of prey, represented on the continents by well-flying forms, became flightless
there. In the Pleistocene, a large Jamaican caracara (Caracara tellustris),
which was larger than other species of these birds and had small wings, lived
in Jamaica. Perhaps it has completely lost the ability to fly. On the California
island, evolution repeated this experiment, and its result was a species of
terrestrial birds of prey resembling a small dinosaur.
In the arid landscapes of California island, favorable conditions are developing
for the dominance of birds that are less demanding of the availability of water
compared to mammals. That is why mammals are represented on this island by relatively
small herbivores, and the role of the top predator of the local ecosystem is
occupied by a large flightless carnivorous bird, caracaraptor.
Compared to the flightless predatory birds of the Cenozoic, this species is
a dwarf: the weight of an adult is about 5 kg; the female is larger than the
male. A notable feature of caracaraptor is its long legs with sharp claws, which
make the bird look bigger than it really is. It also has a relatively large
head compared to related flying species, and the wings have not completely disappeared,
but have become small and can no longer lift the bird into the air. Caracaraptor
is able to run fast, catching up with prey. The tail helps to maneuver during
the chase – it is long, and with sharp turns it opens like a fan, helping to
keep the balance.
The head of this bird is elongated, with a powerful hooked beak of gray color.
The tip of the beak is stretched down and slightly bent – it is an adaptation
for catching small mobile prey. The throat and sides of the head are featherless
and covered with smooth pinkish skin that helps radiate an excess of body heat.
The feathers on the crown are black, slightly elongated and resemble the crest
of an ancestral species. The plumage on the body is of light shades: yellowish-gray
in color, on the back and wings the feathers are covered with thin dark gray
ripples. This coloring helps to avoid overheating during daytime hunting. Occasionally
individuals of a light-colored morph with a grayish-white background color may
be seen. Chicks of both color morphs can be found quite often in the brood.
The main food of this bird species includes terrestrial mammals, reptiles and
birds. Caracaraptor often ravages nests of seabirds. These birds live in pairs
and singly, each individual occupies a certain hunting territory, which it protects
from the encroachments of relatives. After a storm, birds of this species can
often be seen on the shores of the island: they dig in the algae cast ashore
and eat the corpses of marine animals. At such moments, birds pay little attention
to their relatives and can feed together quite peacefully. When there is a lack
of food, caracaraptors treat each other aggressively and can even attack each
other, expelling strangers from their territories. At this time, the caracaraptors
utter loud screeches and snap their beaks. During an attack on a relative, birds
fight with their feet without using their beaks.
The pair of this species persists for several seasons in a row. The mating ritual
includes bouncing and flapping of wings. Also, the male walks around the selected
female, opening his tail like a fan and raising his legs ceremoniously with
each step. When a couple has formed, partners often hunt and eat prey together.
These birds nest on the ground, most often in the shade of cactus plants or
among bushes. The base of the nest is a depression dug in the ground and lined
with grass and thin twigs. In a clutch there are 2-3 eggs equal in the size
to goose egg, with a mottled shell: brown specks on a yellowish-white background.
Incubation is mainly done by the female, and the male feeds her on the nest.
Incubation lasts 44-45 days, the chicks are covered with down and have eyes
opened, but they stay in the nest for a long time. The parents feed the offspring
together for 4 months, and for about six months the offspring learn to hunt
on the parental territory. Usually a pair of birds manages to raise one chick
per season. The young bird is expelled from the territory of the couple shortly
before the mating season at the parents. Sexual maturity occurs at the age of
3 years; life expectancy is up to 40 years.
Genupillan
(Barocaracara aquiliforma)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: pampas of South America.
Changes in nature at the end of the human epoch had a negative impact on the
number and species diversity of large birds of prey. Large species of birds
of prey disappeared in many places due to habitat destruction and the disappearance
of prey, but some smaller species survived and gave rise to new species of the
Neocene epoch. In South America, there is a genupillan – a large bird of prey
species (named after the Araucanian sky god), which is a descendant of the crested
caracara (Caracara plancus), one of the species that successfully survived the
human era. Caracaras, very common birds, survived the ice age and evolved to
several species of birds of prey in the mountains and grasslands of both continents
of the New World.
Genupillan is an ecological analogue of eagles and hunts medium-sized mammals,
especially rodents, and ground birds. If there is an opportunity, this bird
can steal a cub of some ground predator. The physique of this species is similar
to eagles – it has wide wings and long legs with strong toes and pointed claws.
The color of the bird is not bright: the wing coverts are brown-black, the wings
are brown. The feathers on the chest and the front of the neck are straw yellow.
The head is light brown, with a large black beak and bright pink bare skin at
its base. In adult birds, white spots – “epaulettes” – can often appear on the
shoulders. The female is larger than the male; the birds form a close-knit monogamous
family and raise offspring together.
Adult body length is 72-84 cm, wingspan 180-215 cm, weight 2.4-4.5 kg. Due to
the wide wings rounded at the tips, these birds hover for a long time in ascending
air currents, looking for prey on the ground. At the same time, the voice of
this bird is heard – a ringing meowing cry. In case of alarm, birds utter a
sharp croaking sound. A formed pair of birds often hunts together: birds watch
each other and eat prey together. Genupillan rarely quarrels with relatives
outside the breeding season, if they do not approach the central part of the
territory of the pair and keep a distance of at least half a kilometer from
hunting birds. The only exception is the appearance of a lone akatu
(Sciopterornis acatou) – genupillans cruelly mob it, gathering in a flock
and forgetting at this time even about the boundaries of their own territories.
This is especially violent in the vicinity of the nest, if the birds already
have a clutch or chicks. After the attack, the birds quickly leave neighbour’s
territory.
The mating season begins in late spring (in the southern hemisphere – in November).
The male and female fly together for a long time, performing tricks in the air
– somersaults and dives. Sometimes they even fly, clutching claws. At this time,
males expel rivals of their own sex, and females – of their own. The nest is
arranged on rocks in a niche or on the top of a tree with a wide and flat top.
Their clutch numbers 1-3, more often 2 white eggs. Incubation lasts 39-45 days;
the birds take turns on the nest. The chicks are covered with gray down; their
feeding lasts about 75 days. In hungry years, a stronger chick can eat a weaker
one, but this does not happen so often. By the time of leaving the nest, the
young bird is covered with light brown feathers; the chest and throat are reddish.
The young bird stays with its parents almost until the next mating season, learning
hunting techniques, but then it is expelled from the territory. At the age of
4-5 years, a young bird acquires the plumage color characteristic for adults
and begins breeding. Life expectancy is 30-32 years.
This bird species was discovered by Nick, the forum member.
Kakazi,
the false vulture (Kakazi paravultur)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: South America, forests and light forests in areas of tropical and subtropical
climate.

Picture by Pavel Volkov
During the Holocene, human interference in nature caused the
extinction of many species. Only those that were resilient enough were able
to survive and left descendants. One of these survivors was the black caracara
(Daptrius ater). In the Neocene, this species gave rise to the Kakazi (this
word is a modification of the name ka-ka-zi, used by native people of Mato Grosso,
Brazil, to name its ancestor). This medium-sized bird of prey is endemic to
South America, inhabiting its subtropical or tropical forest and heavily degraded
former forest. It also occurs in gallery forests and wooded savannas.
The kakazi have 47 cm of length and 103 cm of wingspan. The females are slightly
larger than the males. Their plumage is glossy black except for the distinctive
white tail. They have bare bright orange heads, legs and feet. The beak is black.
Juveniles can be identified by their dull black plumage, pale yellow face.
These birds have evolved to feed mostly on carrion. Their bare head allows them
to not get their plumage dirty while feeding and their sense of smell and good
vision allows them to find a carcass. In dense forests, these animals will use
their olfactory perception to locate food, using their agility and maneuverability
to pass through the trees and vegetation. In the hierarchy of feeding of a carcass,
the kakazi is above the guyrauna, but below the
true vultures. His beak is strong enough to cut the meat open, with larger scavengers
such as the long-necked apitan allowing
them to do it before they scare the small bird to take the carcass for themselves.
While these birds are developing a scavenging lifestyle, the kakazi can still
eat fruits and actively hunt small prey such as other birds, lizards, amphibs
and small mammals. This bird will also attack nests.
The kakazi are monogamous birds living in small familiar flocks, formed by the
breeding pair and their young of the last nesting seasons. The nest is made
of twigs and built on the top of a high tree. The female lays one to four eggs,
which are incubated solely by her for 32 days. The male stays near guarding
the nest, while the juveniles will bring food for their mother and father. The
young fledge at 67 days old, and will remain with their familiar group until
reaching sexual maturity with 2 years. At this point they leave to stablish
their own family. The lifespan of this species is of 25 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Rapa
Nui caracara (Sociofalco insularis)
Order: Falcon birds (Falconiformes)
Family: Falcons (Falconidae)
Habitat: Easter Island.
The human actions during the Holocene caused the collapse of many ecosystems.
Easter Island was one of these cases. This place was completely deforested and
many of its endemic species perished. However, with the arrival of the Neocene
it was able to recover, developing a new endemic fauna. One of the species living
here is the Rapa Nui caracara, a descendant of the chimango caracara (Milvago
chimango) that arrived in the island coming from South America and evolved here
to this new species.
The Rapa Nui caracara is larger than its ancestor, having a total length of
65 cm and a wingspan of 132 cm. The female is larger than the male. This species
has a mantle and back with cinnamon brown feathers. Neck, chest, abdomen and
belly are dark brown, while the head is pale. The tail is light brown with a
white terminal band. The legs are orange in the male and light yellow in the
female and hatchlings. Their claws are long and sharp, being capable of inflicting
deep wounds. The wings are quite broad, an adaptation that increases maneuverability
in forested areas of their habitat. Their shoulders are chestnut and dark grey
primaries and secondaries, with white tips.
This species is the largest predator of Easter Island, hunting even the
false-nandu. While most raptors are solitary, only coming together for breeding
and migration, the Rapa Nui caracara will hunt in cooperative groups of two
to six. When hunting, all the caracaras spread around the prey and one bird
starts chasing it out. To bring the false-nandu down, these birds will take
turns to attack the prey, causing several wounds until the false-nandu bleeds
to death. These birds will often chase prey on foot, and are quite fast on the
ground and their long legs are adapted for this.
These birds are monogamous, forming pairs that remain together for the rest
of their lives. They nest in the top of trees. It’s made of sticks, plant roots,
and stems, and are often lined with leaves, moss, bark and plant roots. They
are built mainly by the female. There are usually two white to blueish white
eggs sometimes with a speckling of pale brown or gray. The nestlings start out
light buff, but in five to six days turn a rich brown. The incubation lasts
for 36 days. The young begin to explore outside the nest at 38 days, and fledge
at 45 to 50 days. The female sometimes breeds two or three times in a year.
Young stay with their parents for up to three years, helping to raise later
broods. They reach sexual maturity with 5 years and have a lifespan of 27 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Point-crested
dwarf cockatoo (Spinocacatua mimicus)
Order: Parrots (Psittaciformes)
Family: Cockatoos (Cacatoidae)
Habitat: South-Eastern Asia; Jakarta Coast and nearby islands.
In a nature two variants of a mimicry are probable: in one case the unprotected
species imitates well protected one (Bates’s mimicry), and in other case well
protected from enemies species develop common features of appearance (Mueller’s
mimicry); in this case the predator, learned to avoid representatives of one
species, does not attack on similarly colored and protected representatives
of other species that brings mutual benefit to these protected species. Such
phenomenon was known at insects of Holocene epoch, and in Neocene even rather
unrelated species of birds, developing the similar adaptation, had appeared.
Well protected species of Asian spike-headed starlings, the
porcupine bird from South-Eastern Asia, drives enemies off by stings of
long needles growing on head (these are strongly changed feathers). At such
“talent” at once two “admirers” had appeared. One of them is completely defenceless
false spike-headed bird from
flycatcher family, protected due to significant similarity to the porcupine
bird. But other imitator, living near the prickly feathered creature, is quite
able to protect itself independently. It is small parrot, which imitates the
porcupine bird and lives approximately in the same places, as prickly bird.
The mobile crest of pointed cross-striped feathers shows relations of this bird:
it is the descendant of one cockatoo species widely settled in Australia and
South-Eastern Asia in the past. It is named point-crested dwarf cockatoo.
Feathers making crest of this parrot, imitate needles of the porcupine bird,
though, certainly, they can not put any wounds to the predator. They are colored
light brown with cross black strips. Bases of feathers of crest are bright red;
this feature becomes especially appreciable when the bird raises crest up. Crown
and nape of this parrot are also colored red, and sides of its head are white.
By the colouring of body the point-crested dwarf cockatoo in general imitates
the porcupine bird, differing in some features. On neck of bird there is the
black “collar” forming on chest wedge-shaped “tie”. At males this “tie” is larger,
than at females. Back of bird is light brown with cross ripple; tail is black
with white tips of feathers, stomach is grayish white. Except for the size of
“tie”, males do not differ from females in colouring.
Certainly, similarity of this parrot with long-needled spike-headed starling
is not completed: it is spoilt a little by thick hooked beak of black color
characteristic for parrots and short paws on which two toes are directed forward
and two toes – back.
It is not enough only the colouring for successful protection and the bird skilfully
imitates behaviour of long-needled spike-headed starling. Ability to imitation
is characteristic for parrots, and the point-crested dwarf cockatoo uses it
for defense against a predator: it imitates movements of the porcupine bird,
shaking head in sides like it. This behaviour is accompanied by imitation of
aggressive cry of long-needled spike-headed starling that strengthens external
similarity of parrot to this bird. As against completely defenceless false spike-headed
bird, this parrot is not so harmless, and in case of necessary it can bite an
aggressor strongly.
Spike-headed starlings do not form strong families, but this parrot shows an
opposite example: it keeps in amicable pairs formed to all life. Partners feed
and have a rest together, frequently feeding up each other by belched food from
beak to beak. These birds do not form flights, but support contact to neighbour
pairs, in the evening and in dawn exchanging with them by loud shouts.
The center of territory at these birds is the rendered habitable tree-trunk
hollow in which nesting pair hatches nestlings from year to year. Till one year
pair of point-crested dwarfish cockatoos rears two hatches of 2 – 3 nestlings.
The posterity develops slowly: only at bi-monthly age nestlings leave nest.
The first feathering of young birds resembles adult feathering, but head at
them is not such bright as at adults – red color is replaced by brown one. The
first year of life at young birds red sites in feathering are not present, and
they behave very cautiously, preferring to disappear from predators. They study
to simulate aggression of the porcupine bird, observing for these birds or for
adult parrots of the species.
First time young birds nest approximately at three-year age. Life expectancy
at this species reaches 20-25 years.
Drummer
cockatoo, red-headed parrot (Paracacatua erythrocephala)
Order: Parrots (Psittaciformes)
Family: Cockatoos (Cacatuidae)
Habitat: tropical forests of Meganesia (north and east of the continent).
Most large parrots became extinct along with other large tropical birds during
the anthropogenic crisis as a result of human persecution, habitat destruction
and climate change during the Holocene-Neocene glaciation. Among the few large
parrots that survived the end of the glaciation there were some cockatoos –
characteristic birds of Australian zoogeographic realm. Several specialized
species have appeared among the Neocene cockatoos, but there are species of
a more typical appearance. One of these parrots is a drummer cockatoo, a descendant
of the palm cockatoo (Probosciger aterrimus).
This species belongs to the number of large parrots: the body length is about
1 m, the tail is 32 cm. The wingspan reaches 160 cm. The plumage color is black
with a greenish tint, especially intense on the wings and tail, head is red
with a black nape and a movable feather crest, which has an intense green tint.
Drummer cockatoo is a sociable bird that lives and feeds in pairs that last
for life. A characteristic feature of the bird’s appearance is a strong disproportionately
large beak, which allows it to feed on hard seeds of palm trees and nuts, as
well as dry seeds of acacias and other plants. Beetles and snails are an addition
to the diet. When foraging, the bird deftly crunches the shells of nuts and
peels the bark from the branches. If necessary, the bird is able to break wood
with its beak. Birds communicate with each other with murmuring sounds and separate
calls; the alarm signal is creaky and sharp. The enemies of this bird are mainly
feathered predators; drummer cockatoo protects
itself from snakes with bites. The bite of a bird’s beak is capable of crushing
the limb bones of a predatory mammal.
The mating season has no pronounced seasonality. Birds nest in old hollows,
expanding them according to their own needs. At the same time, courtship resembles
that of an ancestor – the male bites off a tree branch, claws into the bark
of a tree or perches on a branch near a hollow and knocks this branch against
the trunk at the hollow in a certain rhythm. Each male has his own rhythm of
beats, and the competitions of young males for a female resemble the roll of
drums in the forest. When a female appears, the intensity of the drumming increases.
Preening the mate’s plumage is a sign of sympathy and trust between nesting
partners.
This bird species reproduces quite slowly. Drummer cockatoo’s clutch usually
has one egg with a white or slightly bluish shell. In case of destruction of
the clutch, the female lays a new egg after 2-3 weeks. Incubation lasts about
30 days, and at the age of 10-12 weeks, the juvenile flies out of the nest and
stays under the care of parents for up to 4-5 months. Sexual maturity occurs
at the age of 6 years. The slow rate of reproduction is compensated by the significant
longevity of the bird – the life expectancy can reach 90-100 years.
This bird species was discovered by Nick, the forum member.
Cut-throat
Digging Cockatoo (Geocacatua cepaphaga)
Order: Parrots (Psittaciformes)
Family: Cockatoos (Cacatuidae)
Habitat: Savannahs and bushy plains of central and southern Meganesia.
Parrots were one of the great success stories of Australia during the Holocene,
these included lories, parakeets and cockatoos, and mankind became fond of them
too, keeping them as pets. Cockatoos were particularly diverse in Australia,
and remain so in the Neocene, despite some previous conflicts with mankind over
crops.
The Cut-throat Digging Cockatoo is a descendant of the Long Billed Corella (Cacatua
(Licmetis) tenuirostris), and is broadly similar in habits and size. They reach
38 centimetres in length, with a wingspan of up to 80 centimetres and a weight
of 550 grams. Outwardly they partly resemble both Galah and Corella, being yellowish
white with a bright pink head, which bears a red stripe across its throat (giving
it its name), undersides of wings are yellow and bases of chest and belly feathers
are pink. Beak is pale and the skin around the eye is puffy and bright blue.
The beak greatly resembles that of its ancestor, but slightly wider, with a
weak hook shape and the upper jaw overhanging the lower, forming a digging implement.
These birds feed mainly by digging in the ground with their beak, in search
of roots, seeds and bulbs of various grasses and weeds. They travel in flock
of up to 30, which may merge with neighbouring flocks to form large congregations,
which settle on open areas of grassland and sod. These birds are very destructive
not only in feeding but habitually to wear their bill down, and may tear at
dead or live wood to occupy themselves. Its call is a wavering screech when
communicating, or otherwise a harsh screech. They are diurnal, and socialize
a good deal in the early evening and late afternoon.
Breeding takes place from August to November, male and female pair for life
and share the nest making and incubating duties. The nest is generally made
in the hollow of a living or dead tree, and lined with plant debris and decayed
wood, these birds may extend these cavities by chewing and scraping them with
their beaks. 2 to 3 eggs are laid and incubation takes up to 23 days, chicks
spend about 52 days in the nest. Lifespan is considerate and can range into
several decades, as long as 70 or 80 years in some cases but usually shorter,
predators include large raptorial birds as well as some kinds of predatory marsupial.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Aotearoan
Crested Cockatoo (Notocacatua cristata)
Order: Parrots (Psittaciformes)
Family: Cockatoos (Cacatuidae)
Habitat: North and South Islands of New Zealand, vegetated habitats that have
trees, including various forests.

Picture by Wovoka, final refinement by Pavel Volkov
During the Holocene, the introduction of foreign species by
man into New Zealand included some Australian species, among them was the Sulphur-crested
Cockatoo (Cacatua galerita). This form was able to proliferate there, and gave
rise to an endemic form, Aotearoan Crested Cockatoo.
In general shape and particulars of anatomy, it greatly resembles its ancestor,
but is a bit smaller. Adult body length is up to 40 centimetres with a wingspan
of up to 85 centimetres, weights does not surpass 800 grams. Feathering differs
from its ancestor in that the chest, back of head and shoulders are smoky grey,
hindquarters and underparts are pale white with bases of feathers yellowish.
Facial feathers are a bright yellow, large pointed crest of feathers is more
intense orange-yellow, underside of tail and inside of wings is very pale yellow.
Feet are dark grey, skin around the eye and nose is pale blue and wrinkled,
beak is bluish grey. This bird has a fairly large powerful bill, diet consists
of large seeds and nuts from both flowering trees and conifers, pine nuts are
commonly eaten. Diet also includes soft leaves, flowers, fruit and insects.
This cockatoo is strictly diurnal, feeding in groups, one individual will usually
be on guard for predators at any one time. Calls consists loud screeching and
squawking calls, which are used to communicate within the flock, especially
in forests where they may lose sight of each other. It is incredibly intelligent
for a bird, as are most kinds of cockatoo, it will sometimes use problem-solving
skills to obtain food.
Breeding occurs between August and January, the parrots are aggressive and will
compete with other birds for nesting sites. Nest consists of a bed of chewed
wood chips in a large tree-hole, two or three large eggs are laid, incubation
lasts up to 27 days. Both parents participate in maintaining the nest and feeding
the nestlings, nesting period lasts no longer than 12 weeks. Fledged juveniles
stay with their parents for a number of months before going out on their own.
Lifespan may be extremely long, up to 70 years, but usually much shorter, predators
include meat-eating marsupials
and hawks, on the ground canids
and large mustelids may attack
it, eggs are vulnerable to large lizards, possums
and mustelids.
This bird species was discovered by Timothy Morris, Adelaide, Australia.
Roseate
parakeet (Europsittacula rosea)
Order: Parrots (Psittaciformes)
Family: Psittaculids (Psittaculidae)
Habitat: Central and Southern Europe, forests.

Picture by Pavel Volkov
During the Holocene, people introduced many species to other
continents. The members of the genus Europsittacula, which type species is roseate
parakeet, are descendants of feral parakeets of genus Psitacula introduced to
Europe. In fact, this genus is a result of the inter-specific hybridization
between rose-ringed parakeet (Psittacula krameri) and alexandrine parakeet (Psittacula
eupatria). Roseate parakeet lives in temperate forests of Europe, migrating
to southern latitudes during the winter. This species is noisy with different
calls for various situations.
The roseate parakeet is sexually dimorphic. The males show bright pink feathers
in their head and chest, while the female have pale pink feathers with shades
of green in these regions. Young parakeets are similar to the female, but the
tail is short. In both sexes the feathers of the back and wings are green, with
a red patch in the shoulders. The upper side of the tail passes from green at
the top to blue further down, and is yellow at the tip. Roseate parakeet measures
on average 45 cm in length, including the tail feathers, a large portion of
its total length. Its wingspan is of 50 cm. Bird has a powerful red beak, which
is used to open nuts and cones.
These birds eat buds, fruits, green parts of plants, nuts, berries, nectar,
flowers and seeds. Roseate parakeet is capable to open cones using its bill
to extract the pine seeds from inside and is capable to break nuts easily.
During the autumn these birds prepare to migration from Europe to tropical forests
and savannas of Africa. During this time, they form large flocks numbering hundreds
of individuals and these groups fly over the Gibraltar Isthmus to reach tropical
Africa.
Roseate parakeet forms small flocks when outside of migration. During the nesting
season they form monogamous pairs. The courtship includes bill rubbing and mutual
feeding. They usually nest in ready tree hollows, but sometimes use tree holes
excavated by themselves. The pair defends their hole fiercely and, if they found
a hole occupied by bat, rodent or any other bird, parakeets expel them violently
from the place. In these holes, the female lays 6 white eggs that are incubated
solely by her; the male is responsible to bring food for her and will protect
aggressively the nest from potential predators. The incubation lasts for 26
days; chicks hatch blind, naked and completely helpless. Only after 10 days,
they begin to open their eyes and their feather quills break through. Both parents
participate in feeding the brood. Juveniles depend on their parents for 8 to
11 weeks after hatching, and only become independent after 12 to 14 weeks. They
are sexually mature in the age of around 2 years and have a lifespan ranging
from 25 to 30 years.
Another species in the genus Europsittacula is red-sided
parakeet (Europsittacula rubroptera). Living in the forests of south-eastern
Europe, this species is larger than the previous one, reaching the length of
55 cm and wingspan of 55-60 cm. Red-winged parakeet is sexually dimorphic: males
show bright violet feathers in their head and chest, while females have pale
violet feathers with shades of green in this area. In both sexes feathers of
the back and tail are green, and the shoulder feathers, primaries and secondaries
are bright red. This species is a migratory bird wintering in forests of Persian
Ridge, and in the northern part of Zinj Land – it must cross the seaway between
this microcontinent and Asian mainland. In late spring birds return to their
nesting area. Some flocks of this species even cross Fourseas and nest in forests
of Three Rivers Land, but their nesting there is irregular.
This bird species was discovered by João Vitor Coutinho, Brazil.
New
Azora parakeet (Azoropsitta macaronesiensis)
Order: Parrots (Psittaciformes)
Family: Psittaculids (Psittaculidae)
Habitat: New Azora, forests of various types.
One of the hallmarks of human epoch was the mass appearance of various biological
species in places where they could not settle naturally for various reasons.
In Neocene, the descendants of many of these species became part of the newly
formed ecosystems. One of the species that in Holocene appeared in a place where
it had not been before is the rose-ringed parakeet (Psittacula krameri) introduced
from India to Europe. Moreover, its number was very high, and in Neocene in
Europe its descendants exist. In early
Neocene, the descendants of the rose-ringed parakeet managed to colonize New
Azora island, where they turned into a separate species – New Azora parakeet.
On the island, the parakeet has undergone some changes: it has become a larger,
crow-sized bird. Its legs have become much longer, and the tail is shorter than
that of the ancestor. The bird spends quite a lot of time on the ground in search
of food and is a good walker. Its claws are blunted compared to arboreal parakeets.
Plumage is green with a lighter belly. The color of the tail is red in males,
brownish in females. Beak is brightly colored: the upper mandible is gray, the
lower mandible is yellow. Due to such a bright color of the beak, birds notice
each other at a great distance in the semi-darkness of the undergrowth. Around
the eyes there is a narrow area of bare red skin.
These parakeets live in flocks of 15-25 birds in the forests of the island,
preferring the growing places of trees with edible fruits (mainly of the heather
family), and rarely leave the inhabited territories. Birds feed in the crowns
of trees, or walk on the ground and gather fallen fruits, clearly preferring
to eat large fruits with a soft peel. Large piles of droppings accumulate under
the trees where the New Azora parakeets live. After passing through the intestines
of the bird, the seeds germinate, and in such places the entire undergrowth
is occupied by young trees. Small birds make nests in these dense thickets,
and it is also a good place for insects to live. Parakeets are tolerant of small
birds and do not conflict with them. Their enemies are local mammals – mustelids
and hedgehogs, and birds
– large barn owls. To protect against them, parakeets gather in a large flock
and vocalize loudly, simultaneously attacking the enemy and mobbing it for a
long time.
Parakeets of this species nest in tree hollows, or dig holes in steep sea shores,
where the soil is strengthened with tree roots. The female lays up to 5 pinkish
eggs and incubates them for about 3 weeks. Birds feed their offspring with a
semi-digested pulp of fruits and seeds. At the age of 4 months, young birds
fly out of the nest; they reach sexual maturity at the age of 4 years. Life
expectancy is up to 50 years, but many birds die earlier.
This bird species was discovered by Mamont, the forum member.
Blue-throated
parrot (Paramazona cyanoguttur)
Order: Parrots (Psittaciformes)
Family: Psittaculids (Psittaculidae)
Habitat: tropical forests of Hindustan.
Through the history of evolution, many species developed analogous structures
that have similar form or function but were not present in their last common
ancestor; this occurrence is named convergent evolution. In the Neocene, the
blue-throated parrot is a case this. They are birds of the Genus Paramazona,
descendants of Loriculus parrots, which through convergent evolution acquired
physical characteristics similar to that of the Genus Amazona of human era.
This new species lives in the tropical forests of Indian subcontinent.
The blue-throated parrot has 35 cm of length and 84 cm of wingspan. They have
large heads, broad and rounded wings. The tail is short, rounded or square.
The beak is robust and the area around the eye is devoid of feathers. Most of
the body is covered with green plumage, however the male have vivid blue feathers
in the throat and red in the mantle and rump, while the females have faint shades
of blue in their throat and lacks the red rump. Immature birds have a duller
rump and lack the throat patch. Like their Loriculus ancestors, they are capable
to sleep upside down.
It is a gregarious species, continuously chattering with a range of whistling
and squawking calls. They eat fruits, seeds, flowers, nuts and leaves. It roosts
communally in trees, and large numbers can be seen at the roost sites at dawn
and dusk.
Blue-throated parrots nest in holes in trees, usually laying three to four white
eggs. The eggs are incubated by the female, starting after the second egg has
been laid, for about 27 to 28 days. Newly hatched chicks have a sparse white
down and they do not open their eyes until about two to three weeks after hatching.
They are dependent on the female for food and warmth who remains in the nest
most of the time until about four weeks after hatching when the chicks have
enough feathers for heat insulation. During this time the male brings food for
the female and chicks, and guards the nest site. From about two to four weeks
after hatching the female also begins to collect food for the chicks. The chicks
fly out of the nest at about 9 weeks and they become independent from their
parents at about 12 weeks after hatching They reaches sexual maturity with 5
years, when the young male acquire adult colors. A monogamous species, the pair
will mate for life. The lifespan of this species is of 35 years.
Other members of the Genus Paramazona are:
Himalayan
parrot (Paramazona himalayana)
The smallest member of the Genus, with only 28 cm of length. The male Himalayan
parrot has bright blue feathers in their cheeks and a bright red plumage in
their rump and mantle. The female lacks the red plumage and their face is faint
blue. They live in the forests at the foothills of the Himalaya and descend
to the valleys in winter.
Blue-chested
parrot (Paramazona cyanopecta)
With 38 cm, this species is the largest of the Genus. The male has a characteristic
bright blue plumage in the chest, which is absent in females. Both sexes have
the predominance of green feathers, there is also the presence of red rumps,
but in the female the color is duller. The blue-chested parrot lives in the
forests of South-Eastern Asia.
Blue-diademed
parrot (Paramazona diademata)
This species is endemic to the tropical forests of Borneo. With 30 cm of length,
the male has a distinctive blue circle in his head, which is absent in the female.
Both sexes are mostly green with an orange colored rump, which is duller in
the female.
Red-winged
parrot (Paramazona sundaica)
Living in the Jakarta Coast, this bird has 36 cm of length. They have a bright
red plumage in their rump, mantle and wings. In the female the red plumage is
dull. The rest of their bodies is green, but the male has blue plumage in their
forehead.
Green-bellied
parrot (Paramazona meganesiana)
Living in the tropical forests of Northern Meganesia, this species is very similar
in size (36 cm) and color to the red-winged parrot, the difference is that the
males of the green-bellied parrot don’t have the blue plumage in their forehead.
This bird species was discovered by João Vitor Coutinho, Brazil.
Long-billed
Lorikeet (Neotrichoglossus vocifer)
Order: Parrots (Psittaciformes)
Family: Parakeets and Lories (Psittaculidae)
Habitat: Different forested areas throughout eastern Meganesia, extending from
temperate forests of Great Dividing Range, through to tropical rainforests of
the north of the continent.
Numerous kinds of parrots saw their extinction during the late Holocene, due
to pressure from the pet trade and habitat loss, but many less pretentious kinds
prospered alongside man, and so leave descendants in the Neocene. Lorikeets
(Trichoglossus) proved their viability by easily living off ornamental trees
commonly used by man.
The Long-billed Lorikeet is descended from this genus, and has further adapted
to feeding on the nectar and pollen of flowers, such as wattles, acacia, bottlebrush
and eucalypt, in the north of their range they feed from various unspecialized
rainforest flowers, they will also eat the pulp of fruit. Their bill is less
parrot-shaped than its ancestor, being conical for reaching into flowers, the
lower jaw is deep and wedge shaped in order to house a large, brush-like tongue
which is even more extensive than its ancestor and can be extended further.
It is a medium sized parrot, reaching up to 30 centimetres long. The head is
a bright purplish blue, turning to bright red on the shoulders and chest, which
is orange tinged further towards the underside, the belly is dark blue. Wings
and back are colored bright orange red with the primaries being a dark green,
scalloped with brownish orange. The bill is yellow-orange and the iris of the
eye is red. Males and females are not dimorphic, and are identical in size and
colour.
These birds are very social, forming large loose flocks which travel from tree
to tree feeding on flowers; they are exclusively diurnal and make a good deal
of noise, mostly high pitched squealing and squawking. Breeding occurs from
August to January, the nest is made usually in tree hollows or ledges, 2 to
3 eggs are laid which incubate for up to 25 days, the female exclusively sits
on the eggs. A single tree may contain more than one breeding pair. Individuals
usually pair for a long time, usually for life.
A closely related species is the Rainforest
Lorikeet (Skoupaglossa splendida), found in the rainforests of Northern
Meganesia. Smaller than its relative, it measures only 20 centimeters long.
It differs by a longer more curved bill which contains a much longer, broom
shaped tongue, this species is intermediate in form between Long Billed Lorikeet,
and south-east Asian species of the genus Florilorius (Loreetos).
The head and shoulders are bright purple with a green back and tail; these feathers
are scalloped with yellow. Wings are bluish green scalloped with olive green.
This species feeds specifically on various large specialized rainforest flowers
that the Long-Billed Lorikeet is unable to feed on, competition to feed on these
flowers is usually high and thus this species is not as social as its relative.
Breeding occurs in the drier months and is generally similar to its relative.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Carunculated
Parrot (Prognathorhamphus nudifrons)
Order: Parrots (Psittaciformes)
Family: Parakeets and Lovebirds (Psittaculidae)
Habitat: Forested areas of North and South Islands of New Zealand, avoids overly
cold upland areas.
One of the few kinds of New Zealand bird to survive the age of man were the
Green Parakeets or Kakariki (genus Cyanorhamphus), their intelligence and willingness
to breed, as well as conservation efforts, assured their success.
During the Neocene an adaptive radiation occurred where these rather generalised,
unpretentious birds speciated into numerous new forms, including those that
take the role of woodpeckers, and
some species that are active only at
night.
Another new form is the Carunculated Parrot, an intelligent species that is
somewhat larger and more pugnacious than its ancestors. Reaches a body length
of about 55 centimetres (including the tail), and weighing up to 200 grams,
with a wingspan of about 40 centimetres. Its feathering is generally green,
the feathers around he head and chest are a dark olive green, scalloped with
brown, hindquarters and tail are a brighter leaf-green, pale yellowish on the
underparts. Wings are a deep bright green, primaries are cross-striped with
brownish olive, tips of wings are black. Head and face are distinctive, the
beak is much larger than its ancestor, strongly hooked upper beak is quite long,
bill is coloured bluish grey with a black tip. Facial area around the eyes,
behind the beak, and fore-part of head are bald, covered in wrinkled, thick
greyish skin, from this it gets its name “carunculated”. Feathers covering the
head and neck are coarse, dense and firm to the touch.
Its unusual face and beak are explained by peculiarities of its diet and behaviour,
it feeds regularly on various insects and their larvae, and will commonly break
open the nests of wasps, bumblebees and honey-bees, to feed on larvae and even
honey. Features of the face accommodate this, the nude facial skin is thick
and wrinkled, partly deprived of nerves, it can sustain stings without harm.
Coarse, dense feathering of the head and neck also prevent stinging insects
from penetrating the skin. This anatomy also serves it well when it preys on
larger more pugnacious insects, like Sawleg Grasshoppers or large mantis. Apart
from this behaviour, this bird will commonly feed on other kinds of insect (beetles,
crickets, grubs etc), as well as snails, lizards, small rodents, and the nests
of smaller birds, fruit and seeds are also eaten.
Calls of this bird include loud, high shrieks, and high-pitched trilling, repetitive
trilling calls are made when flying. It is usually diurnal, but most active
in the morning and afternoon. These birds do not generally flock, and tend to
form pair bonds which remain outside the breeding season, sometimes a pair may
be seen with grown offspring. The pair will become closer and more sociable
with each-other a few weeks before breeding commences. Nesting sites are large
tree-holes, which are dug out and extended by chewing with the beak and throwing
wood-chips to the ground. Nest-building is done by the female industriously,
and the male will attend to her and feed her during this time. Egg-laying commences
between late October and mid-November, up to 8 eggs are laid. Incubation lasts
up to 25 days and is done exclusively by the female, while she is fed by her
mate. Eggs do not hatch all at once, some may hatch a few days after the first,
young are altricial and pink when first hatched. For the first couple of days,
the mother regurgitates liquid food for them, and after this they will start
to consume solid particles of regurgitated food. Chicks open their eyes after
7 days, and after this both male and female will care for the young. After about
a month they are mostly feathered, but remain near the nest, 10 days after fledging
they are able to feed themselves, and they are fully independent about 3 weeks
after leaving the nest. Grown chicks may remain with their parents in a small
group for a year or two.
Predators of this bird include meat-eating
marsupials, cats and hawks, on the ground they may fall prey to mustelids,
canids and Ruacapangi
birds. Lifespan is short for a parrot, usually no longer than 35 years.
This bird species was discovered by Timothy Morris, Adelaide, Australia.
Night-robber
Parrot (Nyctipsittacula rex)
Order: Parrots (Psittaciformes)
Family: Parakeets and Lovebirds (Psittaculidae)
Habitat: Various forested areas of North and South Islands of New Zealand, avoids
overly cold upland areas. In particular commonly visits caves in order to roost
and nest.
The green parakeets of New Zealand (Cyanorhamphus genus), were able to survive
alongside introduced birds at the start of the Neocene, partly because of intelligence,
and changes to behaviour. Some species adapted to different food, or different
habits, some kinds became nocturnal, one of these is the Night-Robber Parrot.
This parrot is a little larger than its ancestor, body length including tail
maybe be up to 40 cm and wingspan is usually up to 30 cm, usually between 100
and 150 grams in weight. Coloring is a dull bluish grey, the head and foreparts
of the body are covered in a blackish-grey mottling and cross-strips, primaries
are cross-striped with dark blackish grey, face is black with a ring of pale
skin around the eye. Beak is larger than its ancestor, lower beak is deep and
upper is long and curved, colour of beak is a dark grey, darker at the tip of
the upper jaw. Legs are a coarse dark grey, and the underparts are feathered
smoky grey. Legs are a bit longer and more agile than those of other parakeets,
feet are strong and grasping, because of this it can climb very well among branches
or rock ledges, and even run quickly and surefooted on the ground.
The habits of this bird compared to its ancestor are unusual, but less so compared
to other birds of the island, it is nocturnal, and has a strong preference for
animal prey. Birds roost in small flocks of up to twenty, and are unusual in
sheltering by day in the ledges of caves, not too far from the entrance. After
dusk, they become active, and go in search of food, which is most often insects,
lizards, small rodents and their young, bird nestlings, foraging within caves
they commonly feed on baby bats and injured adult bats. About 70% of its diet
is composed of animal matter, the rest is usually seeds, fruit and some nectar.
Hunting and foraging is generally done alone, and while they often search the
forest they will eagerly hunt and forage some way into caves that have colonies
of bats.
Though shy and usually silent, during the day as they roost, they commonly socialise
and preen each other, making chittering noises and soft-crooning. These birds
pair for life, and remain with each other each year, young unattached males
will usually leave their home roost in search of a mate. Nest-building begins
in October, when each pair will gather mud and clay, and build a structure in
a secluded corner or on a ledge within the cave. When finished, it resembles
a small igloo, and can only admit one parent at a time, though both parents
are involved in building it. By late October, up to 6 eggs are laid inside the
back end of the structure, away from the entrance. After this, the female will
incubate them for about 20 days, being attended to and fed by the male. The
young are born pink and blind, altricial, and both parents take turn watching
over them and bringing back food. Unlike related forms, the chicks can eat solid
food almost right away, which usually consists of soft meat fed to them. Eyes
are open after 4 days, fledging occurs after 3 weeks, and the young are ready
to leave the nest about two weeks after fledging. Adolescents may roost alongside
their parents for a few years after, and it is usually adult male young that
find new flocks in order to seek a mate. Predators include predatory
marsupials, cats, hawks, owls, mustelids
and canids, large lizards
may eat very young chicks or eggs. Lifespan may be as long as 60 years.
This bird species was discovered by Timothy Morris, Adelaide, Australia.
Beekeeper
parrot (Melipsitta melivora)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: tropical woods of the Central Africa.
This rather large (daw-sized) parrot with short tail is the descendant of small
parrots – African love-birds (Agapornis). This bird has bright colouring: at
it there are green body, tail and wings, coffee-brown head and bright red “bridle”
– thin strip bordering beak and directed from corner of beak up to an eye. Beak,
as at the majority of Neocaenic parrots, is powerful and crushing; at the male
it is black, at the female brown. Above the bird’s beak crest of friable feathers
with rigid core and thin barbs, not forming dense vane as on other feathers,
sticks up. This crest opens not upwards, as, for example, at cockatoo, but to
sides across the head. Feathers on body are very dense and rather rigid. It
is connected to features of feeding of parrots of this species: contents of
bee nests, honey and larvae, makes up to half of diet at these birds. Ravaging
bee nest birds open crest and press it to eyes, protecting them from bee stings.
Parrots of this species also eat nectar of large tropical fabaceous trees (family
Fabaceae), occasionally being their pollinators. For feeding by liquid food
the long tongue covered with fibers of epithelium serves to them - with its
help it is possible to get nectar from flowers and to lick honey in cells of
combs. As against Holocene birds Indicatoridae, beekeeper parrots can not digest
wax. The diet is supplemented with larvae of bees and various other insects.
In connection with such specific diet parrots of this species meet in small
groups: only up to 4 - 6 birds, and sometimes at pairs at all. Birds communicate
among themselves, shouting to one another by hoarse voices.
Parrots nest 2 times per year, dating nesting to the flowering time of fodder
trees. Pair of birds is formed not at once: the first years nesting “alliance”
can break up after nesting, but pairs of adult birds usually keep fidelity to
each other. Such birds form new pair only after death of partner. This species
makes nests in tree-trunk hollows; in clutch there are 3 - 4 white eggs. Both
birds from pair hatch, feeding up the partner sitting on eggs. Nestlings hatch
after 16 days; they are naked and blind, as well as at all parrots. Development
of nestlings proceeds about 3 months, parents feed them with insects and nectar.
Becoming independent nestlings climb out of hollow and about one month keep
together with parents, training to get food.
Beekeeper parrots perfectly know territory on which they live, and remember
location of all bee nests existing there. They fly their possession, taking
away the share of honey in each nest, but not ravaging insect colony completely.
Having found bee colony somewhere in wide tree-trunk hollow or under branch,
birds cautiously approach to it, and then simultaneously attack, trying to break
out by beak pieces of combs with honey and larvae. It occurs very quickly –
all attack lasts about half-minute. After the attack parrots carry off the share
of catch in beak and eat it somewhere on branch of tree. Having pressed piece
of combs to branch by leg, parrot carefully licks honey, then takes and eats
larvae of bees. After its meal on branch only empty combs stay. Usually parrots
eat preyed honey at the same branch as usually, and in due course the true “warehouse”
of empty combs is formed there involving various moths whose larvae eat wax.
If it is not enough bee nests, parrots search for nectar in large flowers of
tropical trees, preferring superficial flowers of red and orange color. Some
trees of tropical forest are adapted to pollination almost exclusively by beekeeper
parrots.
Stiff-tailed,
or wood-pecking parrot (Boreagapornis piciformis)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: North Africa, savannas, thickets of baobab opuntia.
In the human era, the scale of the introduction of alien species exceeded the
natural rate of species exchange between continents by many orders of magnitude.
Invasive species often caused damage to the natural flora and fauna of new habitats,
and after the human disappearance continued to exist in new habitats. Their
descendants became natural components of new Neocene ecosystems, and even began
to play a leading role in communities. This is what happened to prickly pear
cacti in the Old World. In North Africa of Neocene epoch, their descendant,
baobab opuntia (Adansopuntia obesa),
grows – it is a giant tree-like cactus, around which a specific community of
living organisms has developed.
One of the bird species closely associated to baobab opuntia is stiff-tailed,
or wood-pecking parrot, one of the descendants of the yellow-collared lovebird
(Agapornis personatus) of Holocene epoch. It resembles outwardly the related
parrot species: relatively large head, hooked beak and fan-shaped tail, but
its habits are more like a woodpecker: it has tenacious paws, with which this
bird can climb vertically along the trunk, and short but wide wings that allow
maneuvering among the thorny tree branches. The bird’s tail seems ordinary at
first glance, but the shafts of the tail feathers are very strong and thick,
so it serves as a support for the bird when the parrot needs it. The bird’s
beak is lacquer-red, rather massive, slightly jagged at the edges – denser and
tougher areas stretch in strips in the corneous cover of the beak, which play
the role of “teeth” when wearing out the corneous material of the beak.
This bird has a discreet body color – brown head, back and wings, green chest,
belly and tail. Stiff-tailed parrot has a ring of yellow feathers on its neck
– it is a remnant of the yellow plumage areas of the ancestral species. Body
length of this bird reaches 30 cm, the wingspan is up to 50 cm.
The specific appearance indicates that the parrot is a “homebird” and does not
like to fly far away from fodder trees. This bird spends almost entire its life
among tree-like baobab opuntias, less often among other tree species of African
savannas and semi-deserts, and does not fly far from them, avoiding the dangers
of the savanna. Stiff-tailed parrot feeds on various insects that settle under
the bark and in the wood of baobab opuntia and other trees, including large
grubs. A specific kind of food is large tree-dwelling zulal
beetles (Moneilema zulal) living in the wood of baobab opuntia and their
larvae. This is a rather specific food, because these insects are poisonous,
and birds have adapted to neutralize their poison. In addition, some of the
metabolites accumulate in the tissues of the bird, making its meat inedible.
Such a diet has a side effect: the life expectancy of these parrots is less
than that of related species of similar size, but sexual maturity occurs already
at the age of 6-8 months. A significant part of the bird’s diet consists of
cactus fruits, which the bird easily bites. With a lack of food of animal origin,
the bird feeds on fresh cactus shoots, eating the pulp out of them.
The mating season is not pronounced at this parrot: its main food is available
all year round. During the year, the breeding pair nests up to 2 times. These
parrots nest in tree hollows – with their powerful beaks they easily gnaw out
hollows and even tunnels in the thickness of the soft wood of the treelike cactus.
The nest is quite primitive – it is a pile of twigs and grass stems, which the
birds drag into the hollow and spread in a layer along the walls. This species
is monogamous, pairs are formed for life. Colonies of several dozen breeding
pairs are formed in trees convenient for nesting.
At the bottom of the nest, the female lays two or three whitish-pink eggs, and
both parents hatch them in turn for 31 days. Then blind and naked nestlings
hatch from the eggs, but after a couple of weeks they open their eyes and grow
white down, and after another 6 weeks they leave the nest and are able to fly.
They stay with their parents for another 2-3 weeks, and adult birds finish feeding
them. Infant mortality level in this bird is also quite high, and at least half
of the nestlings and fledg;ings of stiff-tailed parrot die before reaching adulthood.
The maximum lifespan of stiff-tailed parrot is up to 8 years.
This bird species was discovered by Bhut, the forum member.
Arafura
mangrove parakeet (Mangropsitta lacustris)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Arafura Lake, mangrove forests.
In the age of man, Australia was one of the centers of species diversity of
parrots. At the end of the human epoch, the diversity of parrots decreased due
to the destruction of tropical forests and climate change, but in the early
Neocene it recovered approximately to the level of the pre-human epoch. Neocene
parrots often occupy new ecological niches not previously occupied by these
birds. Some species of parrots live in a vast zone of mangrove forests separating
Arafura Lake in northern Meganesia from the ocean. One of these species, Arafura
mangrove parakeet, is found exclusively in this habitat. It comes from one of
the species of rosella (Platycercus) – a common genus of parakeets of Australia
in the human era.
Arafura mangrove parakeet is a medium-sized bird: its body length including
its tail is about 50 cm. The tail is about half the total length of the bird.
The coloration of this parrot is quite noticeable, although it lacks bright
spots. Bird has a golden-green back, chest, neck and back of the head. The wing
coverts have a black center; they form a characteristic “scaly” pattern on the
folded wings of the bird. Primary feathers are black with a narrow green border
along the front edge and with green tips. The tail consists of narrow long feathers;
the middle four feathers are of the same length and blue in color, other tail
feathers are bluish-green. The undertail contrasts in color with the tail feathers
– it is rusty-red in color.
Against the background of the head plumage, a white beak outlooks, shaded by
a black “mask” on the front of the head. This species does not have a crest.
Beak is relatively large compared to ancestral forms, of crushing type.
This bird feeds mainly on seedlings and leaves of mangrove trees, as well as
on seeds of palm trees and pandanus (screw palms). Arafura mangrove parrot receives
a significant part of the necessary fresh water from vegetative food. The addition
to the diet are small invertebrates that live in mangrove forests – mollusks
and small crabs, which birds gather at low tide on the roots of mangrove trees.
Outside of nesting, this bird migrates in the mangrove forests in flocks of
20-30 birds. At high tide, this parrot feeds in the crowns of trees, at low
tide it descends to the roots and gathers invertebrates. After the rain, birds
willingly drink fresh rainwater accumulated on the surface of the channels.
These parrots are monogamous; the pair persists for several nesting cycles,
and often for life. During the nesting period, flocks break up or ready-to-nest
pairs separate from them, occupying their own territory. Like most parrots,
this species nests in tree-trunk hollows. If the found hollow is small, the
pair expands it with their beaks, digging up the wood and throwing chips out.
There are 2 clutches of 4-6 eggs per year. The parents feed the offspring for
three months, after which they nest again two months later. There are usually
fewer eggs in the second clutch than in the first one.
Sexual maturity occurs at the age of 14-16 months, life expectancy is usually
no more than 5-6 years.
The idea of the existence of this bird species was proposed by Timothy Donald Morris, Adelaide, Australia.
Phantom
parakeet (Nocturnopsitta phantomica)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: tropical woods of New Zealand.

Picture by Pavel Volkov
During evolution, mastering new habitats or avoiding a competition
to other species, live creatures can change radically former habit of life.
It had taken place so in forests of New Zealand where the descendant of one
local species of parakeets, avoiding the competition to introduced species,
had turned to nocturnal creature. Dim feathering and propensity to life in darkness
create the sensation of that it is the native of the beyond. Therefore this
parakeet has the name “phantom parakeet”. The basic opportunity of occurrence
of parakeet having nocturnal habit of life had been shown by dwelt in New Zealand
kakapo, or nocturnal parrot (Strigops). But it had disappeared in human epoch,
and the nocturnal habit of life at phantom parakeet was developed independently
of this bird.
At the sunset, when day time inhabitants of New Zealand fall asleep, this parakeet
leaves to feeding. Small flocks of phantom parrots fly in wood. They search
fruits of various trees, peck small animals, crush bark of trees in searches
of larvae of insects.
Phantom parakeet concerns to number of medium-sized birds: body length is about
20 cm, tail is about 30 cm long, and wingspan is up to 40 cm. It has characteristic
for all parrots tenacious paws and strong hooked beak. Because birds have twilight
and nocturnal habit of life, their feathering has dim colouring: it is grey
with glaucescent shade. Only on tips of wings primary feathers are colored black.
Paws and beak of bird are colored black too.
Phantom parakeet has large sensitive eyes. It almost does not distinguish color,
but visual acuity of this bird is amazing: phantom parakeet is able to distinguish
in starlight the ant creeping on bark. In eye retina of phantom parakeet there
is small number of cones, but the plenty of densely located rods provides to
bird fine night sight. This parakeet freely flies in wood even at night – it
sees well all obstacles arising at its way. The pupil of eye is round; it can
react to change of light exposure quickly. For protection of eyes against bright
daylight bird uses blinking membrane, therefore phantom parakeets disturbed
in day time are not so helpless at bright sunlight.
For feeding in night wood bird has the special adaptation: phantom parakeet
can distinguish ultra-violet beams. This parakeet finds fruits of plants by
wax coating reflecting ultra-violet beams. Therefore at night phantom parakeet
finds fruits as successfully as other birds do it in day time. But phantom parakeet
eats not only fruits. It is omnivorous, and willingly eats snails, insects and
small vertebrates. For their catching phantom parakeet has one more adaptation
– it has very good hearing which is not worse than owl’s one. Aural apertures
of this parrot are chink-like and rather big. They are covered with lengthened
feathers from above. In full darkness phantom parakeet can catch nocturnal animals
(invertebrates, amphibians, small mammals), listening to sounds uttering by
them.
Despite of well advanced hearing, phantom parakeet prefers to search for food
with the help of sight. Therefore it is most active in twilight and in dawn
when it is enough light. But in moonlight nights this bird can feed all night
without break.
Phantom parakeet is secretive forest bird. Usually these birds lodge in pairs,
and seldom form big flocks. But even if these parrots gather in flock, inside
it they keep in pairs, separately from other birds. Pair at this parakee is
formed to all life. Birds of one pair spend a lot of time together, showing
signs of attention to each other: they feed up each other with belched food,
clear feathering and call together for a long time at the sunset, communicating
with neighbours. Voice of these birds is loud and unpleasant as at the majority
of parrots, but in daily (to say more exact – “nightly”) life they communicate
with the help of silent whistles and clicks.
Birds of this species spend day in tree-trunk hollows and others shlters, flying
off to feed at night. Sometimes these parakeets keep in common with bats in
cavities of trunks of old trees. Spending day time, parakeets sleep, having
seized by claws in vertical walls of tree-trunk hollow away from entrance. To
keep easier on a wall of tree-trunk hollow, shafts of tail feathers form spikes
on tips, and some barbs are rigid and bristle-like. Setting the tail against
wall of tree-trunk hollow, parakeet may be sure that it will not fall down sleeping.
Sometimes in one old hollow tree some pairs of adult birds may spend day time.
In districts where there are caves, this parakeet lodges in them, forming constant
congestions of some tens of birds. Phantom parakeets get on with other animals
not so well – they catch gekkoes and small bats.
In nesting behavior phantom parakeet diligently continues traditions of representatives
of the order – it arranges nest in tree-trunk hollow. In caves this bird nests
in crack between stones, or even in deepening at stone eaves. Nest of this parakeet
actually represents natural shelter without any litter. Female lays 2-3 eggs,
and both birds hatch the clutch. In woody districts of New Zealand this parakeet
has time to make up to 3 clutches per one year, and in cooler mountain areas
only 2 layings. Nestlings hatch at 25-th day of incubating. They are naked and
blind; parents bring them up during 6-7 weeks. Approximately at the second week
of life nestlings start to fledge, and at 6-week age they already try to fly
and train wings. Young birds leave nest and do not come back to it any more.
Parents feed them about one week, and then young phantom parakeets start to
search for food independently. Life expectancy at this species reaches 15-18
years.
Wood-breaking
parakeet (Psittopicus trachyrhamphus)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: New Zealand, forests of various types.
The fauna of New Zealand has suffered significantly due to the introducing of
various mammals and birds in the human era. The Neocene fauna of the islands
is dominated by descendants of introduced species, which turned out to be much
more competitive in relation to local species. Only a few New Zealand animals
(especially among vertebrates) are descendants of native species.
The fauna of New Zealand initially lacked woodpeckers, widespread across the
rest of the globe (except for Australia (Meganesia) and Antarctica). People
did not bring introduce these birds to New Zealand and their ecological niche
was empty for a long time. But in due course of evolution, one of the local
parakeet species got a good chance of survival by mastering the nutrition of
tree-boring insects. However, unlike woodpeckers, this parakeet does not peck
wood, but gnaws it with a very hard and wide beak. For this feature, the bird
was named wood-breaking parakeet.
It is a small bird species: body length of an adult is 25-30 cm, the weight
is about 85 grams. The parakeet seems larger because of its long tail.
Wood-breaking parakeet inhabits forests in which there are large numbers of
old trees affected by woodworm insects. It is a very cautious bird, skillfully
hiding its presence. It is very difficult to notice a wood-breaking parakeet
perching on a tree: its plumage is colored to merge with the color of tree bark
– it is brown with a pattern of dark longitudinal strokes. The secondary feathers
are gray with black cross stripes; they are visible only during flight and serve
as an identification signal for relatives. This kind of bird prefers to perch
not on branches, but on the trunk of a tree, and climbs the branches, holding
its body not across, but along the branch. In this way it resembles large tropical
nightjars. The bird has very tenacious paws, covered on the underside with lateral
corneous combs, allowing it to hold on to the bark without much effort. Tail
feathers also have adaptations for climbing trees – the shafts of the tail feathers
are rigid, and their tips jut out of the vane and serve for support.
Wood-breaking parakeets are strict monogams. During the nesting period, each
pair occupies a large area of forest (up to three square kilometers) and protects
it from the invasion of relatives. To signal them, they use a bright spot –
a white crest of slightly elongated feathers. At rest, however, it is hidden
by a “cap” of elongated feathers of camouflage color. This adaptation allows
it to give signals to relatives: by raising the crest, the parakeet makes white
feathers visible from afar. Outside of the nesting season, birds gather in flocks
of up to a dozen birds and wander through vast forests, never staying anywhere
for a long time. Sexual dimorphism is not pronounced in this species.
Wood-breaking parakeet usually feeds on trees with dried branches or on dying
trees already damaged by insects. This species, unlike most parrots, is predominantly
insectivorous; it eats beetles and large grubs. It extracts them from the thickness
of the wood with a long tongue equipped with bristles, vaguely similar to the
woodpecker’s tongue. The tongue of woo-breaking parakeet can stick out of its
mouth at a distance almost twice the length of the bird’s head. Due to the flexibility
of the tongue, the parakeet can examine the winding passages of woodworm insects.
These birds become sedentary and stick to a certain territory only during the
nesting season, which takes place in September-December (spring and early summer
in the Southern Hemisphere). Like most parrots, this species nests in tree-trunk
hollows. The female lays 4-6 eggs directly on the bottom of the hollow without
litter and incubates them for 18-20 days. The male feeds the female at this
time and protects the territory from relatives. Nestlings hatch featherless
and blind; being about ten days old, they open eyes and at the age of one month
they fledge. They develop rapidly, and become independent at the age of 2 months.
The life span of this species is about 12 years.
The idea of the existence of this bird species was expressed by Nick, the forum member.
Chatham
digging parakeet (Speopsitta moriori)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Chatham Islands.

Picture by Alexey Tatarinov
To the southeast of the main islands of New Zealand, there
is a small Chatham archipelago (the locals – the Moriori people – called it
Rekohu, which meant “misty skies” in their language). By the beginning of Neocene,
the fauna of these islands was severely depleted by human activity and the ice
age that came at the end of Holocene. However, in Neocene, when the climate
became more favorable, the fauna of Chatham restored the diversity of species.
One of the unusual inhabitants of the archipelago is Chatham digging parakeet.
Birds of this species are remarkable in their strong constitution and large
size (body length is 50 cm, weight is up to 2 kg), which makes them look a little
like Holocene parrots kakapo (Strigops). However, in this case, the external
similarity is deceptive: by their origin, Chatham digging parakeets are not
related to kakapo in any way; they descended from the eastern rosella (Platycercus
eximius), an Australian parrot introduced to New Zealand by people. After the
human extinction and the end of the ice age, the descendants of this bird, developed
various habitats of the main islands of the archipelago, settled to more southern
islands.
Chatham parakeets lead mainly a terrestrial lifestyle, which has left its mark
on the appearance of this bird species. They are distinguished from most parrots
by well-developed strong legs with rather long toes, which not only allow them
to run fast, but also serve to dig up the ground and forest litter in search
of food. Other characteristic features of the digging parakeet’s appearance
include a powerful beak and a relatively short vedge-shaped tail. Wings of Chatham
parakeets are not very large and have a rounded shape. The Chatham Islands are
home to animals that are dangerous to these birds, so they have not lost the
ability to fly, although they usually walk on the ground. In case of danger,
they try to hide among plants or run away, but in extreme cases they fly vertically
upwards, like gallinaceous birds.
In its coloration, Chatham parakeet is very different from the rosella, its
brightly colored ancestor. The main color of its plumage is dark yellowish-green.
Throat and cheeks are white; a wide bright red stripe runs over the beak. There
are numerous black speckles on the back; the uppertail coverts are light brown.
The eyes of Chatham parakeets are black, beak is light gray and paws are dark
gray. Male differs from female in slightly brighter plumage.
Chatham digging parakeets have a rather pleasant voice, and they are very “talkative”:
birds often communicate with each other, uttering soft whistling and creaking
sounds. Louder whistles are used in courtship ceremonies and to alert relatives
of danger.
The food of these birds is mainly vegetative: seeds and vegetative parts of
grasses and shrubs, as well as flowers, berries, fruits and mushrooms. They
mostly gather food on the ground, but, using their beak and paws, they can climb
low into trees for seeds and fruits. Terrestrial mollusks and soft insects,
brought by parents, also play an important role in nestling feeding.
Outside of the breeding season, digging parakeets lead a collective lifestyle,
gathering in wandering groups of 15-20 birds – it increases the safety and efficiency
of food search. These birds are characterized by daytime activity, and they
spend the night low in trees or (during the breeding season) in burrows. Sometimes
flocks of Chatham parakeets follow large local birds – barocygnuses.
The Chatham Islands are located in high latitudes, so the birds inhabiting them
have a pronounced seasonality of reproduction. The courtship season of Chatham
parakeets begins in December, which corresponds to the beginning of summer in
the Southern hemisphere. Groups of birds break up into pairs – many of which,
thanks to the emotional attachment of partners, persist for decades – and move
to nesting sites. The formation of a pair is preceded by a courtship ceremony:
the male walks around the female in circles, opening his tail and lowering his
wings and she begs for food from him. This “spectacle” is performed both by
couples forming for the first time, and by birds who have been living together
for a long time.
The nest is arranged in a hole, which depth sometimes reaches 1.5 meters – birds
can dig it out on their own, using their paws and beaks, or use abandoned burrows
of rodents and seabirds. In rare cases, Chatham parakeets nest in low-lying
tree hollows, and inexperienced couples may try to lay eggs simply in a small
depression on the ground (in the latter case, the clutch is almost always doomed
to damage). A good burrow often causes quarrels between pairs of birds: the
owners of such a dwelling have to defend their right to it from the encroachments
of other family couples. In fights between relatives, beaks, blows with wings
and paws and “chicken style” throwing ground are used. Much more fiercely, although
using the same techniques, Chatham parakeets protect their offspring from predators
and ravagers.
There are up to three white eggs in the clutch of these birds, their incubation
lasts up to 28 days. The female incubates mainly, and the male is responsible
for supplying her with food. After hatching the nestlings, they are fed by both
parents. At the age of 45 days, young birds can already fly and feed themselves.
After the end of the breeding season, adult birds and their offspring unite
in small groups again. Thus, young individuals usually do not leave their parents
until the next breeding season. They will be able to reproduce only after reaching
the age of three years.
The only inhabitant of Chatham – a large predatory rat – represents a danger
to adult birds, although eggs and nestlings can also be attacked by smaller
rodents and omnivorous birds, such as gulls or false ravens.
Chatham digging parakeets are long-lived birds. The usual life expectancy is
40 years, and in record cases it reaches 55 years.
This bird species was discovered by Simon, the forum member.
Madagascar
giant false macaw (Pseudarara titan)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Madagascar, eastern and northern parts of island covered with rainforest.

Picture by Sauron from FurNation
Transition from Holocene to Neocene was marked by ice age,
and, as the consequence, the reduction of number and species variability of
tropical forest inhabitants.
Among “victims” of this phenomenon there were many species of typically forest
birds, including parrots. Practically all large parrots had died out to the
end of Holocene. Their destiny was predetermined by people to considerable
degree: people caught these birds for the keeping in captivity and destroyed
their habitats. As the result in early Neocene tropical woods restored after
mankind were occupied by few small species of parrots escaped after climatic
changes. Among these birds descendants of one species of lovebirds (Agapornis)
had reached the certain success, evolved to magnificent birds, the true gem
of tropical forest. These birds inhabit eastern part of Madagascar Island
overgrown with tropical rainforest. By bright and gaudy colouring, and also
by social
way of life these birds resemble Amazon macaw parrots (birds of genera Ara,
Anodorhynchus) which had not lived up to Neocene, having fallen victims of
chasing from the part of people and the reduction of rainforests area in
glacial epoch. For some similarity to American macaw parrots the genus of these
birds
is named false macaws.
False macaws live mainly in tropical woods at the east of Madagascar, and
some ones inhabit also mountain woods. Appearance of these birds has kept
the basic
features of parrots: at them there are powerful hooked beak, large head,
wide wings of rounded outlines and tenacious paws typical for parrots by
structure.
Tail of false macaws is straight and rather short, as against the long pointed
tail of their Amazon prototype. Sizes of birds of false macaw genus vary
strongly – from medium-sized birds about 40 cm long up to the giant 100 cm
long at wingspan
more, than one and half meters. These birds eat mainly dry fruits (nuts,
seeds), and from time to time eat leaves and unripe juicy fruits. Due to
specialization
to feeding by firm nuts these birds avoid competition to lemurs and other
inhabitants of Madagascar woods.
Giant false macaw belongs to largest parrots of the Neocenic world, being
also the largest species of the genus. This bird is colored very brightly:
large
beak is bright red, sharply visible on the background of white feathering
of head. Around of eyes there are rings of naked blue skin; at the male they
are
a little bit wider, than at the female. The body of bird is color green.
Wings are dark green; on back there is white spot, appreciable during the
bird’s
flight.
Madagascar giant false macaw is flocking social species of birds. Out of
nesting season flocks of several tens birds of this species constantly migrate
in forest.
This species belongs to strict monodins: pair of birds is formed to all life,
and breaks up only at the death of one of partners. Sometimes adult birds
lost the ability to breed form pairs only for communication to each other.
In flight
pairs of birds keep together, constantly supporting contact with the help
of sounds and movements. Mutual clearing of feathering and feeding each other
strengthen connections between nesting partners.
In nesting season flock breaks up to separate pairs nesting at significant
distance from each other. Due to good memory pairs of birds find out each
other after nesting, and structure of flight stays approximately identical
each year.
Nesting of these large birds occurs in tree-trunk hollows which are converted
by the special way. From their ancestors, lovebirds, this bird has inherited
building abilities which were transformed at the process of evolution. In
large tree-trunk hollow pair of birds constructs spherical or bow-like nest,
throwing
and plaiting leaves and twigs. On elastic litter female lays up to three
white shelled eggs. During the hatching and feeding of nestlings the dung
of birds
fails between twigs to the bottom part of nest, where it is eaten by various
symbiotic insects settling in nests of birds – beetles and cockroaches. For
nesting the pair returns each year to favourite tree-trunk hollow, and carefully
clears it of the rests of last year's nest.
Naked and blind nestlings hatch after 4 weeks of incubating and develop rather
slowly. Both birds of pair care of them. In first weeks of life of nestlings
the male does not feed them directly: it transfers food to the female which
feeds nestlings. Approximately at bi-monthly age young birds of this species
fledge completely. The juvenile feathering differs from the adult one – head
of young bird is green, and eyelids are light. Also at young birds there
is brown beak. Young growth leaves nest approximately at three-monthly age.
Parents
and nestlings had left nests gather to flocks where young birds study to
adjust social connections and prepare for independent life, adopting experience
of
adults. Before approaching of maturity flocks of young birds do not break
up.
Life expectancy of giant false macaw is very great: over 60 years.
At Madagascar other species of false macaw genus live:
Two-colored false macaw (Pseudarara bicolor) is
much smaller, rather than the previous species. It is flocking crow-sized
bird; however it seems larger because
of long tail making about half of length of bird. The colouring of this bird
which have determined the name, has strongly pronounced dismembering type
– in it two colors are combined, sharply bordered from each other. The top
part
of body – sides, wings and tail – has green feathering with blue metal shine.
Head, breast and the top part of back are colored milky-white with bluish
shade. The beak of this parrot is colored grey, and featherless skin around
of eyes
and in the basis of beak has yellow color. This parrot nests in groups, forming
colonies on large trees. It plaits large nests of twigs and long leaves of
tropical plants. Each year nests are renewed and completed, gradually forming
unified construction. For construction nests birds dexterously tear to strips
rigid palm leaves. The collective nest of two-color false macaws occupies
some tens cubic metres in tree crone, and weighs over one ton. In such constructions
various species of birds, small snakes and mammals frequently settle.
Celestial false macaw (Pseudarara caelestis) has
received the name because of unusual colouring – this bird is almost completely
colored blue color with
metal shine on wings. Only few parts of body of bird have other color: “cap”
on head of bird is colored dark grey, legs are black, and beak and skin around
of eyes have “ivory” yellowish-white color. Celestial false macaw is rather
small species of the genus – its length is about 40 cm. At this bird there
are short wide tail and rather massive constitution. Celestial false macaw
eats nuts and other firm fruits, and migrates in forest in searches of food
by small flocks which number no more of ten of birds (actually flock includes
two or three breeding pairs and their posterity). Flocks at this species
are kept for the nesting period – birds nest nearby from each other, and
in common
protect nest of any of pairs. They arrange nests in hollows of old trees,
and if necessary can make hollow in soft wood, using firm beak. Usually these
parrots
nest in sites of broken forest stand, or near the rivers where trees with
such wood grow.
Dawn false macaw (Pseudarara eos) has
rare enough pink colouring of the most part of body. Only primary feathers
and part of back of this bird have white
colouring, and the bottom part of stomach is ochre-red. This parrot up to
half meter long with short rounded tail gathers to flocks numbering of some
tens
of birds. In rich in food sites of forest these birds form flocks of several
hundreds birds, representing magnificent show in flight. This parrot has
rather weak beak, and eats soft food – fruits and flowers. It also willingly
eats
invertebrates – insects and snails. Dawn false macaws nest in big flocks
and build of twigs and stalks of grassy plants collective nests around of
the top
part of tree trunks.
The idea about the opportunity of existence of these birds was proposed by Simon, the forum member.
Basket-making
parrot (Megagapornis architector)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Madagascar, dry forests in the western part of the island, in river
valleys.
In the human epoch, among the most successful inhabitants of Madagascar there
were lovebirds (Agapornis). These small birds were able to live in a man-altered
landscape and did not depend on the presence of old trees with hollows for reproduction,
because they were able to make nests themselves. Their small size allowed them
to preserve quite numerous populations in conditions of strong fragmentation
of habitable landscapes. A significant part of the Madagascar parrots of Neocene
epoch descends from lovebirds. The most impressive among them are large false
macaws (Pseudarara), but these birds are not the only descendants of lovebirds
– in the forests and savannas of the island various parrots live, ranging in
size from small to quite large ones.
The dry forests to the west of the island’s mountain ranges are inhabited by
one of these species – a large basket-making parrot. It is crow-sized bird with
a short tail, wide wings and a massive beak. The background color of the plumage
of this species is emerald green, only on the rump there is a patch of white
feathers. The bird’s head stands out vividly against this background: it is
red and white, with a red “mask” and a white crown and nape. The bird’s beak
is light gray. Male and female almost do not differ in color: the female has
only a little more white color in the coloration of the head, and less red one.
Also, the female has a larger white spot on the rump: its display serves as
a signal for mating. Legs are featherless, dark gray in color.
Palm seeds and hard-shelled nuts compose a food for these birds, but up to a
third of their diet consists of soft fruits and the tops of tree shoots. Sometimes
these parrots eat food of animal origin – insects and snails. And due to their
powerful beaks, they are able to split the bones of large vertebrates in search
of bone marrow, so they can often be seen waiting in line around feasting predators.
They need food of animal origin mainly during the feeding of offspring.
These parrots live in stable pairs. For nesting, this species builds a large
twiggen nest, often intertwining branches of living trees. Birds interweave
them with twigs and fibers cut from palm trunks, arranging a nest up to one
meter in diameter among the branches. Such a nest turns out to be very durable,
and with regular repairs it serves for several years in a row. After parrots,
owls, small herons and bats often settle in such nests. The nest chamber is
lined with dry grass and palm fiber. Basket-making parrot nests in colonies
of several dozen pairs, occupying several trees growing next to each other.
In a clutch, there are 4-5 eggs with a white shell; the female incubates mainly.
Incubation lasts 20 days, young birds spend 2 months in the nest, fledging and
eating off. Young birds, leaving the nest, weigh up to 1500 g, exceeding the
weight of their parents. They differ from adult birds in plumage color: there
are no white areas in the plumage, and the red color is replaced with reddish-brown.
Beaks of young birds are dark gray. They learn to fly over the next few days
and begin to accompany their parents in search of food. At the age of one year,
young birds get adult plumage color, and two-year-old birds form breeding pairs
and nest. The life expectancy of this species reaches 50 years.
The mating ritual includes mutual feeding and preening. A peculiar feature of
the mating ritual is the male sticking grass into his plumage of the back and
displaying it to the female with outstretched wings and tail upraised. This
is an echo of the method of carrying of building material to the nest, characteristic
of small lovebirds. The formed pair of birds jointly builds a nest, or repairs
an old one. Usually, if the old nest is occupied by a heron or owl, parrots
prefer to build a new nest. Nevertheless, parrots do not try to expel the owl
from the colony, since its presence scares away terrestrial and arboreal predators.
Black
and white lovebird (Agapornis chiaroscuro)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: savannas of southwestern Africa.
After the mass extinction at the edge of Holocene and Neocene, when many species
of large mammals, reptiles and birds died out, the empty niches appeared occupied
by the descendants of their smaller neighbours. It happened so on Madagascar,
where the genus of parrots called lovebirds gave rise to false macaws belonging
to the number of largest parrots of Neocene. But it did not happen so everywhere:
in mainland Africa, where the enough number of large species of birds survived,
lovebirds remained rather small birds, and black and white lovebirds are among
them.
Black and white lovebirds are medium-sized birds: about 30 cm long, of which
6 cm wide and blunted tail takes. Actually, this is the very large representative
of its genus had already existed in Holocene epoch.
The females of this species are somewhat larger than males are, and it is the
only feature of sexual dimorphism.
The basic coloration of the body is speckled, of greenish-gray color. Due to
this color bird can hide from its’ possible predators. But head is colored much
more noticeably: the forehead is black, and the cheeks and the throat are white
(hence the name of the bird). Also, the tail of this species is covered with
cross stripes of black and white. The beak of the bird is also colored black,
and its eyelids are white.
These birds inhabit the savannas with parts of forest and scrub vegetation in
the southwest Africa, sometimes even not far from the seacoast. The nesting
period begins at the end of dry season and at the beginning of rain season,
when males and females start to call each other by the loud grumbling cries.
Breeding pair keeps together for many years, but during the nesting period birds
behave as if the pair is forming for the first time. After several weeks in
rough nest made of branches and bast fiber females lay from 2 to 8 eggs and
incubate them for about three weeks.
The nestlings covered with down of greenish color hatch from eggs. 3 months
after hatching the chicks are covered with feathers, and by 5 months of age
they leave their nest, but they remain with their parents prior to the beginning
of the next year. Out of breeding season the family groups gather in flocks
and migrate between islets of forest vegetation in savanna. At the next year
young birds take part in nesting for the first time.
Black and white lovebirds feed on berries, seeds and graminoids. It is not interested
in food of animal origin and becomes itself prey for various predatory birds
and snakes.
Its maximum lifespan is 9 or 10 years.
This species of birds is discovered by Bhut, the forum member.
Translated by Bhut.
Boreal
parrot (Neoaratinga flammea)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: temperate latitudes of North America, deciduous forests.
Aratinga erythrogenys, the original species, is a 30 cm. long macaw relative,
native both for the Tumbes region of Peru and Southern Ecuador, is a lean-looking
long-tailed bird, with plumage green-yellowish and splotches of crimson red
on face and front of the wings, plus a circle of white naked skin around the
eyes, and black beak. It lived in flocks from 10 – 1000 individuals, traveling
between foodsources (usually leguminous trees and fruiting trees) when the fruits
or seeds are mature, and also rarely eating insects such katydids, on a very
ample territory. They are very noisy, and the chicks are reared on tree hollows,
preferently on palms, the 2 – 4 young are all-green plumaged and leaves the
nest on 50 days; is a very adaptable species, living on dryforests, rainforest
edges, andean forests, farmlands and even urban landscapes; was also traded
as a pet, forming urban colonies on San Francisco and areas on Florida.
Once humans are extinct, the colonizing birds are forced to adapt to fully wild
landscapes, and diverged on many species.
The first is the boreal parrot (Neoaratinga flammea), 70 cm long, with thick
plumage dark green, all underbody red-orange, without eye rounding patch, and
a compact, squarish grey beak and chunky bodyshape. This species prefer to live
on pairs or family groups, on the southern conifer belt and chestnut-oak forest,
and eats mainly pinecones and other conifers seeds, and also chestnuts and hazels,
and is unusual among North American parrots from this latitudes in being sedentary,
enduring the winter instead to migrating. Unlike the rest of Aratinga-descendents,
this species only have one chick per clutch born on spring and leaving the nest
on 4 months.
It uses its thick beak to enlarge existen treeholes on parroty fashion, and
line up the interior with its feathers and large mammals hair.
The group of the southern residents:
Redwing
parakeet (Aratinga populator) is the least derived of all, even clasiffied
on the same genus of their ancestor, is smaller than it, on the 22 cm threshold,
having a yellowish-green plumage, with the primaries scarlet colored and a large
white patch around eyes, forms huge flocks of many thousands, on an area ranging
from Florida to the Mississipi delta zone, has abandoned the tree-nesting habit
and digs nests on soft substrate cliffs and riversides, cribbing the chosen
site with the thousands of individual nests, eats a lot of tree and shrub seeds,
but also eats leaves and sprouts, showing a folivorous trend, and frequently
defoliating entire trees. Usually their flocks are mixed with pigeons and other
parrots, seeking protection from predators like the parrot-specialist wolf-falcon
(Cynofalco gregarius) which follows the immense flocks.
Blue wedgetail
parrot (Deltura cyanaea) has a shortened tail forming an almost perfect
equilaterous triangle, and a plumage cobalt blue on males, with greenish-blue
females, the white ocular patch is a narrow horizontal streak. This bird 26
cm long having long legs giving a stilted appearance, lives on the southern
part of North America from Florida, overlapping with the anterior species, to
the Sonora-Texas desert band. This species prefers mixed habitat, with trees
to nest and open terrain, lives in groups of 20 – 60 birds, walking on ground
on search of seeds, insects and small vertebrates. It is a very quiet species,
except when a danger alerts the group, the alarm call is a loud screeching sound,
and the group flies to the cover of trees.
Whiteheaded
parrot (Deltura leucocephala) is a pale olive green 15 cm long bird,
with the short, triangular tail and stilt legs of the genus, and a greyish-white
head, with the oval eye patch. It lives on dry and desertic landscapes, with
a range from Sonora-Utah-California to the north of Mexico, having at least
two geographic races. This species is rather solitary, living on pairs or on
small temporary flocks, is mainly a nectarivore and frugivore on cacti, and
also stalks lizards and beetles from the highю It uses the strong beak to dig
nests on cacti.
Bluetail
(Lacrimopsitta longicauda) is the more common and widespread species,
with 40 cm of long, lives on variated forest landscapes, on small groups of
40 or less. It is dark olive green, with many orange-yellow horizontal bars
on the breast, teardrop-shaped white eyepatch, with the narrow edge leaning
to the ear, and sky-blue tail-feathers. It lives on many of the central North
America, and occasionally sighted on the boreal parrot territory. It has a mixed
diet, with diverse fruits and seeds, including also a lot of insects and deadwood
grubs.
The migratory species:

Picture by Carlos Pizcueta (Electreel)
Beach screecher (Lacrimopsitta vocifer),
also called “long-tailed kea”, although isn’t specially close to this NZ parrot,
is a 38 cm long bird,
has
a recurved scythe beak, robust but not specially long legs. Its plumage
is green-brownish, with orange throat and teardrop-shaped eyepatch. It has
unusually
long and pointed wings. , and is a strong flyer, they lives on groups of
20-25, and have a intense leaning for carnivory, they are known for attack
many waterbird
colonies for the eggs and chicks, and also can mob and kill, then eat isolated
gulls, ducks and other parrots. Also they eat carrion and sea invertebrates.
The distinct subpopulations live on a huge territory, with northern grounds
in area of coniferous forests, usually open lands near lakes or the sea,
and wintering on the coastal Gulf of Mexico and California. They are distinguished
from one another mainly on territory and diet, varying from molluscivores
seldomly
stealing seabird eggs to exclusive waterfowl predators. The breeding season
is on the northern territories, they are the only species of the group
to nest on ground burrows, usually on islets or other zones protected.
Fighting parakeet (Alloaratinga pugnax) is
an open land and riverside forest species, of 18 cm of long. Body is shiny
green, breast tan-colored
with pink
head and square-shaped white eyepatch. It eats variated shrub and herb
seeds, migrating between the Rockies latitude to the breeding grounds on
the gulf
of Mexico. When sedentary it lives in groups of more or less 40, but on
migratory season forms multitudinary flocks which can darken the sky. The
name is because
the species usually brawls and fights with other similarly-sized parrots
on roost sites or when coming to drink.
All species have plain greenish-colored young, adquiring the specific color
markings when achieve sexual maturity. Usually they live on flocks and
break on pairings on breeding season. The original species is extinct on
North
America on the Neocene.
These species of birds were discovered by Edgar Segovia, the Philosophica-Dixonia group member.
Yucatan
conure (Pyrrhura mexicana)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: tropical forests of the Yucatan Peninsula.
During the Holocene, human interference in nature caused the extinction of several
species. However, some species were able to survive and its descendants continued
their evolution in the Neocene, with the Genus Pyrrhura being one of them. This
genus was once widespread, reaching from subtropical South America and southern
Central America. These birds suffered with the human impacts over the ecosystems,
but after humanity became extinct, the survivors of this genus recovered and
spread, even reaching lands beyond their original range. During this time, the
Genus Pyrrhura gave rise to some new genera, among its descendants is the Genus
Araguari, large macaw-like psittacids. Now many of the Pyrrhura species
disappeared and the Genus became a relic taxon, with only two species still
living. One of these species is the Yucatan conure.
Yucatan conures have 22 cm of length and 47 cm of wingspan. Adult and juvenile
parakeets look similar in plumage. They have dark maroon faces and ear coverts,
with a blue crown. They have white eye rings surrounding their eyes above their
dark gray beaks. Their necks are covered in green feathers. The breasts are
colored blue with hints of white and brown, while their abdomen is red. The
wings have a blend of red, green and blue. Their tails are maroon with a green
base.
The diet of the Yucatan conure is mostly made up of fruits, berries, and seeds.
However, they will also eat certain flowers, insects, and their larvae.
This bird is social, being typically seen in pairs or flocks with 40 individuals.
These flocks are typically noisy when in flight but will be quiet when resting
or feeding in the canopy. Monogamous, after the pairs are formed, they will
stay together for life. A mated pair can be identified in a flock because the
male and the female will fly very close to each other. The nest is placed in
a tree cavity. The female lays between 4 and 6 white eggs and the incubation
lasts for 25 days. Only the female incubate the eggs, with the male being responsible
to protect the nest and bring food to her. The young will fledge with 24 days
and they reach sexual maturity with 2 years. The lifespan of the Yucatan conure
is of 27 years.
The other representative of the Genus Pyrrhura is the Amazon
conure (Pyrrhura amazonica). This species is restricted to the Northern
parts of the Amazon rainforest in South America. This bird have 23 cm of length
and a wingspan of 49 cm. The plumage is mostly green. The chest has dark grey
and whitish scaling on a green background, and the rump and belly are dark maroon.
The face and throat are dark red, the forehead is blue and there is a reddish
ear-patch. The long, graduated tail has a red underside.
This bird species was discovered by João Vitor Coutinho, Brazil.
Blue-fronted
macaw (Araguari cyanofrons)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: the forests of various kinds and forested savannas of the central part
of South America.
During the Holocene, human activities caused great damage to the Psittaciformes.
Many species became extinct, while others became critically endangered. However,
the psittacids that were able to survive the anthropogenic onslaught over natural
resources during the end of the Holocene, continued their evolution and their
descendants ended filling the niches left by the psittacids that weren’t able
to survive. One of these survivors were birds of the Genus Phyrrhura, which
were versatile enough to survive in great urban centers and in the Neocene they
gave rise to the Genus Araguari. Araguari is a word of the Guarani language
which was used as a name for some species of psittacids. These birds are morphologically
similar to the Ara macaws, with long tails and long narrow wings. The difference
between the members of these two genus is that the Araguari macaws lack the
bare face patch around the eyes that was characteristic to the Ara macaws. This
gives these birds the appearance of giant parakeets. The birds of this genus
filled the niches that were once occupied by the true macaws (Genus Ara) and
by large parrots. A successful group, the Araguari macaws originated first in
South America and spread to North America and Caribe before the disappearance
of the Isthmus of Panama. The type species of this genus is the blue-fronted
macaw. This bird lives from the savannas of Central South America to the forests
of the Atlantic coast of this continent, also reaching the Araucaria forests
in the South.
Blue-fronted macaws are large birds, with 80 cm from beak to tail and a wingspan
of 87 cm. Its plumage is mostly green, with exception of the primaries and secondaries
feathers which are red. It bears a vivid blue plumage in its forehead, crown
and nape. The tail is long and has feathers that are bright green above and
red below. The beak is strong and dark grey, the feet are of this same color.
Seeds and fruit are the major part of the diet of these birds. Their beaks are
well adapted for eating various seeds and nuts as they have the ability to break
open the hardest of shells with relative ease. Because of this, they are seed
predators not seed dispersers. All Araguari species consume clay to absorb toxic
compounds produced by some poison an to supplement the diet with sodium. Blue-fronted
macaws will leave their roosts in flocks around dawn and head to their feeding
areas.
Blue-fronted macaws are monogamous and remain with their mates for life. As
they fly in large flocks the mates fly together. They will also be found flying
in pairs in their feeding, roosting and nesting areas. The breeding season occurs
during the spring. The nest is made in cavities on trees, either live or dead.
Fissures on cliffs are also used. These birds will aggressively dislodge digger
parrots (genus Ampliforpus) from their nests in cliffs, which are then enlarged
by the macaws. Females will lay one to two eggs which only she will incubate
for a period of approximately 26 days and the chicks fledge from the nest about
90 days after hatching. Blue-fronted macaws reach sexual maturity in two to
four years. Their lifespan is of 50 years.
Other species in the Genus Araguari:
Blue-headed
macaw (Araguari cyanocephala)
Living in the savannas of Central South America and in the Gran Chaco, this
bird has 76 cm of length and a wingspan of 83 cm. They are mostly green with
the exception of the head that is blue and the primaries and secondaries feathers
that are red.
Green-chested
macaw (Araguari amazonica)
Living in the Amazon rainforest, this bird has 85 cm of length and a wingspan
of 92 cm. They have green plumage in the throat, underbelly and tail. The head,
nape, wings and back are blue. The primaries and secondaries feathers are red.
Red-headed
macaw (Araguari rubrocephala)
Living from the tropical forests of the Panama Peninsula to the tropical forests
of Mexico, this bird has 79 cm of length and a wingspan of 85 cm. It has a red
head, bright green body, blue wings and green greater coverts. The tail of this
bird is scarlet with blue on top.
Red-bellied
macaw (Araguari erythroventer)
Living in the subtropical forests of North America, from Texas to the Florida
Peninsula, this bird has 77 cm of length and a wingspan of 83 cm. Its plumage
is mostly green, with exception of the belly, underbelly and chest that are
red. This species also has a red nape and crown. The primaries and secundaries
feathers of this species are blue.
Antiguan macaw
(Araguari minor)
Living in the tropical forests of Great Antigua, this species is the smallest
member of the Genus Araguari, with 67 cm of length and a wingspan of 78 cm.
The plumage of this species is mostly green, with a red forehead, a red patch
over the ears and bright blue wing feather.
This bird species was discovered by João Vitor Coutinho, Brazil.
Blue-headed
digger parrot (Ampliforpus azureoceps)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: South America, Amazon basin, riverbanks in tropical rainforests.
In human epoch inhabitants of tropical rainforests had suffered seriously of
deforestation and climatic changes. Some of them had become extinct due to loss
of habitats, and others had to compete to species of other habitats, suffering
also of epizooties. From among parrots inhabited South America in human epoch,
genus Forpus (parrotlets) successfully survived in epoch of antropogenous crisis
due to smaller size, adaptability and developing of antropogenous landscapes.
Descendants of these birds take a prominent place in tropical ornithofauna of
South America in Neocene, forming a new genus Ampliforpus. Representatives of
this genus differ from their ancestors in larger size – hence the name: “amplis”
in Latin means “large”. Because of their nesting habits these birds are named
as “digging parrots”.
The typical representative of this genus is blue-headed digging parrot, large
bird (24 cm long) living in forests of Amazon basin and making nests in steep
slopes of riverbanks. It has a typical appearance inherited from its ancestor:
short fan-like tail, wide rounded wings, robust short-legged body and rather
large head. Its beak is wide and robust, adapted to cracking hard seeds and
nuts of tropical trees, and also for digging.
Birds of this genus have expressed sexual dimorphism in coloration. Background
color of plumage is green (like at its ancestors) with prominent yellow mark
on back at males. Male also has sky-blue coloration of head feathers gradually
changing to background green color on breast and hindneck. Female has green
color of feathering, and only its head has a lighter shade. Juvenile males do
not differ from female in coloration, and only after the first molt get a typical
male coloration. Beak and paws have dark grey color; in some local populations
beak is almost black.
Human activity in far past had influenced to the directions of evolution of
birds nesting in tree-trunk hollows. Due to competition with the Africanized
bees for holes in the trees, birds had to find new ways of building their nests.
These parrots adapted to build their nests in burrows dug by them, with their
legs and powerful beaks, in steep slopes of riverbanks. Blue-headed digging
parrot is a monogamus bird; the couple finds a suitable place to dig their nest
and takes turns in digging. When it is finished, the female covers the bottom
of the nest with leaves and lays four eggs of white color. Their incubation
lasts for 18 days. The incubation of eggs is performed by females, while the
male is responsible for guarding the nest and feeding the female and nestlings
once they are hatched. Juveniles fledge being about five weeks old and siblings
keep in close contact with one another for a few weeks post fledging. The nest
will continue to be used by the nesting couple for the rest of their lives.
If the nest is destroyed by natural reasons, birds easily make new hole for
nesting. Out of nesting time birds keep in flocks numbering up to 20-30 adult
birds and sexually immature subadults. Birds communicate with each other using
loud hoarse calls. Their calls represent a characteristic alarm sound when they
find owl, hawk or any land-dwelling predator.
These parrots eat seeds, nuts, leaves, berries and cactus stalks. Due the ingestion
of clay these parrots can eat a greater variety of seeds and leaves, which would
be toxic to other birds and the construction of nests in places with clay abundant
facilitates the obtaining of this substance and allows the hatchlings consuming
it when they are very young. Juveniles become sexually mature at the age of
2 years. Lifespan is about 35-40 years.
In tropical forests at the foot of Andes another species lives – yellow-headed
digger parrot (Ampliforpus xanthocephalus). It is smaller a little
– adult bird is 20 cm long. Typical feature of this species is yellow color
of male head; female has only yellow forehead and lores. Background coloration
of feathering is also green; beak is black. Males use the contrast of beack
and head feathering coloration for attracting females and warning stranger males
about their claims on nesting territory. It makes nests in steep slopes made
of clay. This species nests in small colonies of 3-4 nesting couples only, and
these birds protect their nesting area cooperatively.
Plain areas of South America are occupied by blue
digger parrot (Ampliforpus caesius). These large (23 cm long) birds
have the singular characteristic of building their nests in hollowed-out termite
mounds in pampas. This species feeds on seeds and friuts, and also adds a considerable
amount of grass seeds to its diet. It has pointed wings and flies fast, migrating
in pampas out of breeding season. The background coloration is light-blue with
distinct greenish shade on back at females. Beaks and legs are grey. These birds
are least social in genus – only two or three breeding couples nest together
and form flocks.
The fourth species in genus – green-headed
digger parrot (Ampliforpus robustus) – is a parrot having very distinct
color differing from other species of genus – it has yellow body (greenish shade
at females) and green head with almost white beak. It is a southernmost and
largest species in genus – 26 cm long. These birds live in southeastern South
America and make nests in steep slopes made of clay. They search for food in
newly formed Atlantic Forest and in Araucaria moist forests. Flocks of this
species number up to 20-30 breeding pairs.
This bird was discovered by João Vitor Coutinho, Brazil.
Lilac-faced
parrot (Amazona migratoria)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: California Island, Pacific coast of North America.
During the Holocene, the man introduced many species to other continents. Some
only because of their ornamental value, while others were introduced because
they were useful for agriculture. However, most introductions were those of
animals used as pets. The psicittacids were among those that were most introduced,
being spread across several continents. The red-crowned parrot (Amazona viridigenalis)
was a species that lived in northeastern Mexico and southern Texas. In their
natural range, the species was suffering with human pressure and their population
decreased. However, some of these birds escaped from their owners and became
feral in California. This population grew and thrived, leaving descendants in
the Neocene.
The lilac-faced parrot is their descendant, one of the last few representatives
of the genus Amazona in the Neocene. These birds are endemic to the forests
and woodlands of the region that once was the American state of California.
During the winter, these birds migrate to the forests of California Island and
of the Pacific slopes of Mexico.
They have 43 cm of length and a wingspan of 86 cm. These birds also have rounded
tails and wings. Their plumage is mostly green with the most notable features
being a bright red forehead and crown, with blue-lilac faces. Their beak and
legs are dark gray.
Their diet consists of seeds, nuts, fruits, flowers and nectar. The lilac-faced
parrot has a great preference for pines and the fruits of the oaks. They hold
the food with their legs and use their strong beak to break the hard shell.
The lilac-faced parrots are gregarious and monogamous. The pairs will be seen
flying close to each other in the flocks that can have 20 to 40 individuals.
During their migration, such flocks can have hundreds of birds. When flying
to their wintering grounds, the male and female will constantly vocalize to
each other. They start to breed in the middle of spring. The female lays 3 to
5 white eggs that can be laid with an interval of three to four days. Females
are responsible for the entire incubation period of 28 days, and only leave
the nest for short feeding periods. Fledging begins at nine weeks old. Their
nests are made inside tree cavities near other members of their flock, creating
a breeding colony. They will return to the same cavity every year to raise chicks
unless that cavity was previously attacked or they did not have reproductive
success there for a different reason. While rare, intraspecific fights can happen
if they find the hole already occupied by other parrots. The chicks leave the
nest after 60 days, but remain close to their parents during the first migration.
Sexual maturity is reached with 5 years and they have a lifespan of 34 years.
This bird species was discovered by João Vitor Coutinho,
Brazil
Greater
monkey parrot (Tardipsittacus pithecinus)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Eonesia, Burotu Islands.
Picture by Alexey Tatarinov
In Neocene, Eonesia islands represent one of the few places
on Earth whose fauna was formed in natural way. They were formed after the human
disappearance, and their settling occurred solely due to the own capabilities
of living organisms to settle and colonize new habitats. Therefore, the fauna
of Eonesia islands bears all those features of originality that were lost as
a result of human introducing of alien species to the islands in the historical
era. Flightless birds, sometimes very peculiar, are often found on these islands.
In the fauna of the Burotu Islands there are very peculiar birds – parrots that
have lost the ability to fly. In the dense forests of the islands, they occupy
an ecological niche roughly corresponding to primates. Their representative
is a greater monkey parrot, a large bird leading an arboreal lifestyle. The
weight of an adult is up to 4 kg: a bird is the size of a large rooster, only
with a proportionally larger head and a massive beak. Wings of this species
are developed, but have greatly decreased in size. Due to the large stomach,
the size of the sternum has greatly decreased, and the flight muscles are weak.
Because of it, the ability to fly in this species is limited to gliding from
one tree to another, without flapping wings. In its way of life, the bird resembles
hoatzin (Opisthocomus) of the human epoch. Tail is wedge-shaped, with very dense
and stiff feathers, slightly resembling the woodpecker’s tail. While climbing
trees, the tail is used as an additional support.
The main color of the plumage is dark green, orange on the back and at the base
of the tail; there is a white spot in the lower part of the head and throat.
In contrast to the wings, the legs are very well developed and proportionally
larger than those of parrots of ordinary appearance. The tarsometatarsus and
toes are covered with bare gray skin. Claws are pointed and hooked, with their
help the bird climbs trees and grabs food. The lower surface of the toes is
covered with numerous corneous spikes that improve grip with the support. Greater
monkey parrot is able to climb trees, and with the help of strong legs it jumps
from one branch to another without the help of wings. Sometimes, when a bird
jumps unsuccessfully, it grabs a branch with its beak and only then pulls up
and clings with its paws. Sometimes this parrot can hang from the support and
get food with its beak or another paw in a hanging position.
This species has a very strong crushing beak of gray color, darker at the base.
Greater monkey parrot feeds mainly on hard seeds and fruits with large kernels.
It supplements the diet with young foliage, which it “chews” before swallowing.
Marks of its feeding are very characteristic – these are branches, the ends
of which seem to have been cut off with a sharp knife.
Greater monkey parrots are strict monogamous; the pair persists for life. The
mating ritual is very simple: the male pursues the female he likes in the tree
crowns, displaying his dexterity to her at the same time. An important role
in the formation of a pair is played by the personal sympathies of birds – the
female treats aggressively the male she does not like. An established pair of
birds spends a lot of time preening and feeding together. Birds mark claims
to the feeding area with loud calls.
The nesting of this species occurs exclusively in the hollows of old trees,
so the birds avoid areas of young forest. If necessary, the pair gnaws out a
hollow of the proper size in rotting wood, eaten away by insects. Reproduction
rate is very slow. There are two eggs in the clutch, but the pair usually manages
to rear only one chick. Incubation lasts 23-24 days; nestlings hatch naked and
blind. The young bird fledges completely at the age of 80-82 days, leaves the
nest at the 120th day of life and migrates through the forest with its parents
for about 2 months. Sexual maturity comes at the age of 5 years. Life expectancy
is up to 60-70 years.
In the forests of Burotu there is a more common lesser
monkey parrot (Tardipsittacus versicolor). Obviously, it lost the ability
to fly on each of the islands independently, after settling across the archipelago.
The weight of this bird reaches 1 kilogram, in proportions it resembles its
larger relative. The ability to fly is limited to gliding between trees. The
plumage is light green, wings and tail are dark brown, and back is reddish-orange.
There is no white spot on the throat. On different islands there are different
color morphs, differing in the presence and shades of color marks in the background
color of the plumage. This species has greater fertility and a faster rate of
reproduction – a couple raises two chicks in one nesting cycle, sexual maturity
occurs at the age of 2 years. This species feeds exclusively on fruits and large
seeds.
Heavy-billed
parrot (Lophopsitta gravirhynchos)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Easter Island.
The human actions during the Holocene caused the collapse of many ecosystems.
Easter Island was one of these cases. This place was completely deforested and
many of its endemic species perished. However, with the arrival of the Neocene
it was able to recover, developing a new endemic fauna. One of the species living
here is the heavy-billed parrot. Its closest relatives are the parrots
that live in Eonesia. This bird is found widespread throughout the island.
The heavy-billed parrot has 28 cm of length and 57 cm of wingspan. The plumage
is mainly green, except for the blue crown and reddish-orange in the face, neck
and upper chest. The primaries and secondary feathers are violet. The tail is
rounded with yellow tips. These birds have a large, heavy and strong beak. The
beak is black, while the legs are pale gray.
This species feeds on a number of fruits and seeds. They use their strong beak
to break the hard shell of the Rapa
Nui coconut, a descendant of the coconut that was introduced by the humans
in the island.
The heavy-billed parrot is a gregarious bird, forming flocks of 15 to 40 individuals.
These birds are monogamous breeders who nest in tree cavities. Each mated pair
of parrots needs their own tree for their nest. The female lays two to four
eggs, which she incubates for 30 days while being fed by her mate. The adults
defend their nesting sites. The chicks require feeding and care from their parents
in the nest. The parents take care of them until 5 weeks after they are fledged.
Young leave the nest at the age of 12 weeks and reach sexual maturity with 2
years. They have a lifespan of 32 years, but many are killed by the Rapa
Nui caracaras before reaching this age.
This bird species was discovered by João Vitor Coutinho, Brazil.
Large-billed
infantopsitta (Infantopsitta raphomima)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: Eonesia, Burotu Islands.

Picture by Alexey Tatarinov, colorization by Biolog.
In Neocene, the consequences of human intervention in the
natural balance still remain in the fauna of the Earth – it is a significant
number of species that are descendants of alien invaders, purposefully or accidentally
introduced by people to new habitats. The amount of such species is especially
high among the representatives of the island fauna. “Uncontaminated’ island
fauna is found only on those islands that appeared after the human disappearance.
Such places include the large Eonesia archipelago in the Pacific Ocean. Here,
in due course of evolution, true island species arose, resembling the typical
fauna of the islands of the Earth before human colonization.
A peculiar bird species lives in the forests of the central island of Burotu
archipelago – large-billed infantopsitta, a ground-dwelling parrot. It is a
bird of a heavy constitution, completely lost the ability to fly and resembles
a dodo (Raphus), exterminated in human era. The process of transition to terrestrial
life in this species has gone even further than in kakapo (Strigops) and Mauritian
broad-billed parrot (Lophopsittacus) of human era. The evolution of infantopsitta
followed the path of neoteny and the preservation of infantile features – in
this respect this species resembles dodo of the human era: adult birds retain
the features of the juvenile in appearance. In this species, wings are greatly
reduced, although they still retain all the wing bones typical of flying birds.
The plumage has changed, having lost the features characteristic for flying
birds: it has become soft; the feathers are loose, more like wool. There are
several shortened primary feathers in the wings; tail is wedge-shaped, represented
by several elongated pointed feathers.
Adult infantopsitta weighs up to 8 kg. This species has strong legs with massive
bones, adapted for walking. Bird can walk for a long time and, if necessary,
run quickly for short distances, but it brings food to its beak with difficulty,
without showing the dexterity of its ancestors. Toes are covered with calluses
from below and have thick blunted claws that allow digging holes.
A characteristic feature of the appearance of this species is a massive black
beak. It allows the bird to crush the fruit kernels. Also, this parrot digs
edible plant roots with its beak and legs. A significant part of the diet of
infantopsitta consists of plant fruits and succulent leaves of low-growing plants.
Small seeds are not digested by the bird and fall with its droppings into the
ground, where they germinate.
Male and female do not differ from each other in coloration; male is slightly
smaller, with a larger beak. The background color of the plumage is green with
a white "mask" around the beak and eyes. Back has a brownish hue,
there is a dark brown half-ring on the neck, interrupted in front.
Infantopsitta is a strict monogamist; couples are formed for life. Male and
female constantly stay together, communicating with sound signals. The voice
of this parrot resembles a throat grunt; in case of irritation or fright, the
bird utters a loud unpleasant cry.
This species nests in wide burrows, which it digs under the roots of plants.
Preparing for nesting, the male chooses a secluded place in the forest and begins
to dig a hole, loosening the ground with his beak and raking it away with legs.
He invites the female to join the nest arrangement with nodding head movements
and “cooing”. The rate of reproduction in the infantopsitta is very slow. Nesting
takes place once a year; there is only one egg in the clutch, which is alternately
incubated by both parents. There is no nest litter in the burrow. Incubation
lasts about 30 days. If the egg turns out to be unfertilized or lost during
incubation, the birds undertake a new clutch only after 2-3 months. The nestling
hatches featherless and blind, stays in the nest for up to 3 months. In the
first weeks of its life, the female stays in the nest for a long time and takes
care of it. A young bird spends the first 3-4 months of life with its parents,
then begins to lead an independent life and unites in bachelor groups with other
relatives. New pairs are formed within such groups.
Sexual maturity in this species occurs at the 5th year of life, life expectancy
is up to 60 years.
Azure
parakeet (Sociopsitta cyanea)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: the gallery forests of the savannas of Northeastern and Central South
America.
During the Holocene, human activities have caused the extinction of many species
and the increase in the range of others, though the introduction of species
in others places. The monk parakeet (Myiopsitta monachus) was a common pet,
being transported to many other regions of South America and of the world. Some
individuals turned feral and in the Neocene they give rise to the Genus Sociopsitta,
with the azure parakeet being the type species. The name of the Genus refers
to their highly social habits, these birds live in large flocks and make large
communal nests.
The azure parakeet has 42 cm of length (including the long tail) and a wingspan
of 90 cm. The male is slightly larger than the female. The plumage of both sexes
is vivid blue, with the exception of their greyish blue breast. They have a
large and powerful grey beak, which is used to break nuts and seeds. The legs
and feet are brownish-black. They are noisy birds, vocalizing a lot while flying
in their large. Their flocks can have from 50 to 100 individuals.
This bird eats seeds, fruits, flower buds and nuts. They use their powerful
beak to break the hard shell of nuts and capsule fruits. The fruit most consumed
by them is that generated by the riverine
almond (Neoterminalia riparia), a descendant of the Terminalia catappa (a
species introduced by the man in the Holocene), they will also consume the seeds
of the violet trumpet tree (Tabebuia
violacea). Both these two species are the main trees that form the gallery
forests where the azure parakeet lives.
The azure parakeet mates for life. The nest is large structure made of sticks
with separate entrances for each pair. The nests can cover completely the branches
of trees. Other animals use these “apartments”, such as the grey-faced
pygmy owl (Glaucidium griseorostrus), a species of owl that generally uses
the nest of azure parakeet for reproduction. In these nests the female lays
8 white eggs, which are incubated only by her for 25 days. The chicks fledge
in 68 days and are independent in 100 days. The lifespan of the azure parakeet
is of 36 years.
Other species in the Genus Sociopsitta is the Emerald
parakeet (Sociopsitta viridis). This species lives in the areas of
the Gran Chaco. With 38 cm of length and a wingspan of 85 cm, this bird forms
flocks with less individuals than the azure parakeet, with 40 to 60 birds. Their
main characteristic is the bright green feathers in the back, nape and forehead.
The other parts of their body are vivid blue, with the exception of their greyish
blue breast.
This bird species was discovered by João Vitor Coutinho, Brazil.
Rapacious
parrot (Pseudonestor vorax)
Order: Parrots (Psittaciformes)
Family: True parrots (Psittacidae)
Habitat: tropical zone of South America, from the Amazon rainforest to the savannas
of Central South America.

Picture by Wovoka
During the Holocene, human interference in nature caused the
extinction of several species. In the Neocene, the niches left behind by the
species that went extinct were filled by new creatures. One of these is the
Rapacious parrot, a species of omnivorous psittacid with predatory tendencies.
The Rapacious parrot has 46 cm of length and 72 cm of wingspan. Its plumage
is green, with a reddish yellow belly and uppertail coverts. It has red feathers
on the undersides of its wing. It has a bare black facial skin and a relatively
long, strong, hooked dark grey bill, giving to the bird a vulture-like profile.
The bare part of the head is an adaptation to avoid the feathers of being dirty
from the blood of the carcasses that it eats.
An omnivore, the Rapacious parrot feeds on more than plants, insects, other
birds, mammals, and carrion. Its diet will change according to the disponibility
of food. When there is an abundance of fruits, these birds become nearly complete
herbivores. However, when these birds find carrion, even when there is an abundance
of fruits, they will eat it. Is common to see Rapacious parrots among birds
of prey and cathartids feeding on carcasses. When the number of fruits decreases,
these parrots become more carnivorous, actively hunting small mammals and birds.
These birds will also attack nests of birds to eat their eggs and chicks.
Rapacious parrots are monogamous breeders who nest in tree cavities. Each mated
pair of parrots needs their own tree for their nest. Couples will fight each
other for the nests. If they find a nest occupied by a smaller species of bird,
the couple will aggressively expel it, and if the size allows, the parrots can
kill and eat the other birds. The female lays three to five eggs, which she
incubates for 30 days while being fed by her mate. The adults aggressively defend
their nesting sites. The parents take care of them until 5 weeks after they
are fledged. The young leave the nest at the age of 12 weeks. The lifespan of
the Rapacious parrot is 45 years.

Picture by Wovoka
Another species in the Genus Pseudonestor is the Andean rapacious parrot (Pseudonestor andinus). This species inhabits the Andean Plateau and is more carnivorous than its relative, hunting efficiently other animals that live in the plateau. More robust that the Rapacious parrot, the Andean species have 52 cm of length and a wingspan of 92 cm. Its plumage is moss green, with a yellow belly and uppertail coverts. It has pale red feathers on the undersides of its wing. Like its relative, the Andean rapacious parrot have a bare black facial skin and a relatively long, strong, hooked dark grey bill. This species build its nest in rock crevices.
This bird species was discovered by João Vitor Coutinho, Brazil.
Chahuiztli
bird (Neodysithamnus pochtecophaga)
Order: Passerine birds (Passeriformes)
Family: Antbirds (Thamnophilidae)
Habitat: seasonally humid tropical forests and rainforests of Central America.

Male

Female
Pictures by Pavel Volkov
A descendant of the subspecies of plain antvireo Dysithamnus
mentalis septentrionalis, which lived in Holocene in Central America: from southern
Mexico to Panama. Birds of this species live largely exclusively on account
of the megacolony of pochteca ants and feed on
insects that are found in its vicinity. Per day, one bird eats up to one thousand
ants, thousands of mealybugs and up to a dozen
of caterpillars (it is inconvenient for birds to
catch them, since the caterpillars feed mainly on the underside of the calathea
leaf). Hence the name of this species: chahuiztli – “pest” in Nahuatl (Aztec),
for the damage caused by the bird to pochteca ants and to a lesser extent to
purepecha ants.
Up to a hundred chahuiztli birds “graze” over each megacolony every day, eating
up to 100,000 ants per day. But even such loss in the ant family is not too
much damage to pochteca ants, since the fertility of pochteca females in megacolonies
is very high, as is the fertility of their “livestock” – coccids. Moreover,
the birds themselves turn out to be useful for ants, since they give large quantities
of guano so necessary for calatheas fertilizing.
Chahuiztli is a small bird species: the largest male is no more than 10 cm long.
Chahuiztli has a powerful (for such a small bird) beak, which is curved at the
tip.
In the color of the plumage chahuiztli males slightly differ from the ancestor:
back and wings are dark green from above; throat, abdomen and wings are acid
green from below. This separation of coloration continues up the head and neck
all the way to the beak, and only the sides of the head are black. The plumage
of the females is entirely brown-green, darker on the back and olive only on
the undertail, with a few white spots on the wings. Birds lack any adaptations
that physically or chemically protect them from being bitten by ants. Chahuiztli
bird successfully hunts these dangerous insects due to the speed of movement:
it lands and pecks two or three ants or coccids on a path or bush, and immediately
flies to a safe place on the branches of shrubs or trees, from where it looks
for a new easy prey. The bird has very keen eyesight and hearing.
Males, ready to reproduce, sing for a long time, uttering a long trill of short
loud whistles, accelerating towards the end. Chahuiztli birds are monogamous,
create pairs for life and choose their own territory for life, which they actively
protect from relatives and food competitors. They usually lay two eggs in a
nest woven of fibrous stems and plant down, suspended from thin branches. Both
parents equally share responsibilities during incubation and feeding of nestlings.
Incubation lasts 16 days; the development of juveniles lasts up to 3 weeks.
When the juveniles fledge and leave the nest, each parent takes care of only
one of them. The nesting cycle repeats up to three times a year with short breaks.
Sexual maturity occurs at the age of two years, life expectancy is up to 10
years.
This bird species was discovered by Wovoka, the forum member.
Tototl,
marauder-antbird (Gymnopithys latronem)
Order: Passerine birds (Passeriformes)
Family: Antbirds (Thamnophilidae)
Habitat: seasonally humid tropical forests and rainforests of Central America.

Picture by Pavel Volkov
A descendant of the bicolored antbird of the subspecies Gymnopithys
bicolor olivascens, which lived in Central America from Honduras to Panama during
the human epoch. This bird species is a specialized insectivore, whose entire
life depends on conquistador ants.
It follows the wandering armies of ants, eating the insects they startled, like
a marauder following an army and plundering on the battlefields. Hence the second
name of the species. “Tototl” means “bird” in Nahuatl (Aztec). Per day, this
bird eats more than a thousand various arthropods that tried to escape from
the mandibles of conquistador ants. Every multimillion army of ants is followed
daily by more than a hundred of such birds, and by their voices the large inhabitants
of the forest learn about the approach of columns of insects.
Tototl is a small bird, no more than 10 cm long, with short wide wings and long
tenacious legs. The powerful beak, curved at the tip, allows this bird to open
the hard chitinous shells of beetles. The birds themselves are very nimble –
they are able to snatch prey literally from the mandibles of ants, thus eating
also the hunters clung to their prey.
A special feast for these birds comes after the grandiose battle of conquistador
ants and pochteca ants, after which hundreds of
thousands of dead insects remain on the battlefield. Birds feed in such areas
of the forest in groups of dozens of individuals, peacefully treating each other.
The color of the birds is slightly different from their ancestors, bicolored
antbirds: the back and wings are brown-green on top, and the throat, belly and
wings are white from below with a slight blueness. There is a blue spot of feathers
around the eye. Tototl has very keen eyesight and hearing, it is able to catch
prey on the fly.
Otherwise, the birds are very similar to chahuiztli, their distant relatives,
differing only in a more diverse diet. With the beginning of the rainseason,
the mating season begins at them. Single males fight for the attention of females,
arranging duels on the branches of shrubs and low trees. They chase each other,
catching at the tail and wing feathers with their beaks, and push them off branches
convenient for singing. The winner male attracts the female with a monotonous
accelerating melodic trill, uttering it repeatedly for several minutes, after
which he rests for a short time, and begins to sing again.
Like chahuiztli, tototls are monogamous and create couples for life. They usually
lay two eggs in a deep sac-shaped nest suspended on the tree – the incubating
bird is almost completely hidden from predators. Both parents take turns while
incubating eggs for 2 weeks, and then jointly take part in feeding the nestlings
– 2 weeks in the nest and about a week after the juveniles take wing. Each parent
raises one chick, and after a while the pair repeats nesting – in total, the
birds grow up to 2-3 broods per year, depending on the abundance of feed.
Sexual maturity occurs in two years, life expectancy is up to 10 years.
This bird species was discovered by Wovoka, the forum member.
Rufous-bellied
choca, or rufous-bellied antshrike (Neothamnophilus rufus)
Order: Passerine birds (Passeriformes)
Family: Antbirds (Thamnophilidae)
Habitat: lowland tropical Atlantic forests in South America.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. One
of these new species is the rufous-bellied choca, the type species of the Genus
Neothamnophilus. The direct ancestors of these birds were Thamnophilus antshrikes
that survived the anthropogenic onslaught during the Holocene. The word choca
was the popular name of several representatives of their ancestral genus. The
rufous-bellied choca lives in the lowland Atlantic forests of South America.
The rufous-bellied choca have 15 cm of length and a wingspan of 32 cm. This
species, like all the members of this genus, is sexually dismorphic. The male
is dark gray with a black crown and a rufous belly. They also have black wings,
with white spots in the coverts. The female is completely rufous, with the exception
of the crown and wings that are dark brown and lack spots in the coverts.
This species is insectivorous, feeding on insects and other arthropods that
are captured in low vegetation and on the ground.
The rufous-bellied choca lives singly or in pairs, often with dense undergrowth.
Breeding is seasonal, starting during the spring and continues through the summer.
The nest is a woven cup, generally placed rather low. Both sexes incubate 2
to 3 eggs. The incubation lasts for 10 days, taking 12 days for the chicks to
fledge. They remain with their parents for three more weeks. Sexual maturity
is reached with 6 months and they have a lifespan of 7 years.
The genus Neothamnophilus has other representatives. One of them is the
striped
choca (Neothamnophilus striatus), an inhabitant of the woodlands and
gallery forests of Central South America. This species has 13 cm of length and
a wingspan of 32 cm. The male is black with several white stripes on the belly.
They also have white spots in the coverts. The female is pale gray, with the
exception of the crown and wings that are black. The Amazon
choca (Neothamnophilus amazonicus) is another representative of this
genus. Living in the understory parts of "terra firme" forests in
the Amazon, this species have 14 cm of length and a wingspan of 31 cm. The male
is black with a white belly. They also have brown wings, with white spots in
the coverts. The female is completely gray, with the exception of the wings
that are dark brown. The Agreste
choca (Neothamnophilus minor) is endemic to the transition areas between
arid savanna and Atlantic forest located in Northeastern South America. The
word agreste was used in Brazil to name this region. These birds have
13 cm of length and a wingspan of 30 cm. The male is brown with a black crown
and a black belly. They also have black wings, with white spots in the coverts.
The female is completely light brown, with the exception of the crown and wings
that are gray.
This bird species was discovered by João Vitor Coutinho, Brazil.
Horned
gnateater (Conopophaga nigricornis)
Order: Passerine birds (Passeriformes)
Family: Gnateaters (Conopophagidae)
Habitat: chaparral and forest areas of Grand Chaco.
A group of birds characteristic of South America is suboscine passerines, a
suborder Tyranni that unites several bird families. In due course of evolution,
they turned into ecological analogues of Old World songbirds (suborder Oscines).
Gnateaters, widespread in the South American jungle, suffered from the deforestation
of tropical regions in the human era, and the ice age, which caused an even
greater reduction in their habitat, made a second blow to the diversity of the
family. The relict populations preserved by that time adapted to the new conditions,
but more often simply died out. Few of them survived to Neocene and gave new
species in the recovering tropical rainforests. One of the descendants of the
rufous gnateater (Conopophaga lineata), horned gnateater, adapted to life in
more arid conditions.
Compared to other species of the family, horned gnateater is a relatively large
species: the body length of an adult bird is 21-25 cm, wingspan is 34-39 cm,
weight is 55-100 g. It is a bird of dense build with large head, short tail
and long legs. Sexual dimorphism in coloration is clearly expressed in this
species. Male has a brown head and back, and a reddish belly. Primary feathers
are black; there is a large white spot on the back edge of the vane of each
feather. On the head, a horizontal black stripe stretches from the base of the
beak across the eye, passing on the nape into two narrow bundles of elongated
black feathers similar to horns. In the female, primary feathers are brown without
spots, and the black color is also replaced by brown on the head; a weakly pronounced
cross-striped pattern is noticeable on the back.
This bird species keeps mainly on the ground and in the bushes, where it gathers
food – insects and slugs. The horned gnateater’s voice is a series of monotonous
whistles and squeaks. At dawn and dusk, the bird flies over its territory of
about 4.2 hectares; at the same time, the wings produce a characteristic sound
like “fr-r-r”. When the bird spots an insect, it makes a short rush down to
the ground or to the upper leaves of plants to catch it.
Horned gnateater is monogamous, a pair is formed for one mating season. Courtship
among birds begins shortly before the end of the dry season; the juveiles leave
the nest shortly after the rains begin. Sometimes birds manage to feed a second
brood during the rainy season. The nest of this species is built in the thickets
of branches and looks like a deep bowl of twigs and leaves, lined with moss.
The female lays three dark green oval-shaped eggs with a slightly pointed tip.
After 17 days, blind and naked nestlings hatch, which begin to grow juvenile
fluff from the second day of life. At the age of 10 days, their eyes open and
feathers begin to sprout, and at 17 days they fully fledge and at the age of
3 weeks they try to fly and leave the nest, keeping on the branches of trees
or shrubs. Young birds have dull plumage, and lack characteristic feather horns.
They reach sexual maturity in the first year of life; life expectancy is up
to 7 years.
This bird species was discovered by Nick, the forum member.
Southern
night tapaculo (Nyctoscytalopus soricoides)
Order: Passerine birds (Passeriformes)
Family: Tapacolos (Rhinocryptidae)
Habitat: forests of the southeast of South America.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. Among the survivors were tapaculos (Rhinocryptidae).
Representatives of this family survived in the Atlantic forests of South America
and continued to evolve. In the Neocene, they gave rise to the Genus Nyctoscytalopus,
the night tapaculos. The type species of this genus is the Southern night tapaculo,
an inhabitant of the Atlantic lowland forests of Southeastern South America.
The Southern night tapaculo is a tiny bird, having 11 cm of length and 23 cm
of wingspan. Their plumage in the upperparts is dark gray, while in the lower
parts it's dark brown. They have large eyes with well developed tapetum lucidum
that are suitable for their nocturnal lifestyle. Their beak and legs are dark
gray.
This species forages during the night on the ground for insects, spiders, and
centipedes. All representatives of this genus are birds of the understory of
the forest, rarely leaving it. They are fast, moving swiftly in the ground like
a shrew. During the day, these birds rest in small cavities.
The Southern night tapaculo is a monogamous species, with the male and female
forming pairs for life. It nests in natural cavities in the ground or on fallen
logs. It makes its nest in the form of a sphere made of material found in the
vicinity of the couple's territory, such as roots, small twigs, lichens and
others. The female lays 2 white eggs that incubated solely by her for 17 days.
The chicks are feed by both parents. They fledge with 23 days, but remain with
the parents for more 4 weeks. They reach sexual maturity with 3 months and have
a lifespan of 5 years.
Another representative of the Genus Nyctoscytalopus is the northern
night tapaculo (Nyctoscytalopus minor), an inhabitant of the Atlantic
forests in Northeastern South America. They have a similar plumage to that of
their Southern relative, but are smaller, having 10 cm of length and 20 cm of
wingspan. The highland Atlantic forests in the Southeast have an endemic species,
the Serra
night tapaculo (Nyctoscytalopus montanus). Differently of their lowland
relatives, this species have light brown underparts. They have 12 cm of length
and 24 cm of wingspan.
This bird species was discovered by João Vitor Coutinho, Brazil.
White-throated
obrero (Sociofurnarius albogularis)
Order: Passerine birds (Passeriformes)
Family: Ovenbirds (Furnariidae)
Habitat: savannas, gallery forests of central part of South America.
In the Neocene, several new species appeared. While some of them were morphologically
similar to their ancestors of the Holocene, they developed habits that were
far different from them. An example is the genus Sociofurnarius, the obrero
birds. The name obrero means worker in Spanish. These birds are direct descendants
of the horneros and like their ancestors, they build their nests as structures
made of mud and other materials. However, different from them, these birds are
very gregarious, forming flocks of 20 or more individuals. Such lifestyle reflects
also in the construction of their nests, with these individuals working together
to make large structures that resemble a building with apartments. The type
species of the genus is the white-throated obrero, an inhabitant of the savannas
and gallery forests of Central South America.
The white-throated obrero has 15 cm of length and 37 cm of wingspan. Like all
the furnariids, their plumage has different shades of brown. Their upperparts
and wings are reddish-brown, while the underparts and tail are light brown.
They have black stripes around their eyes and a white throat. The legs and beak
are pale gray.
These birds feed on insects and other arthropods obtained by foraging on the
ground while walking. They forage in flocks, with birds of other species following
them. Songs in the white-throated obrero are sexually distinct. The rapid trill
that is usually heard as part of the duet is faster in the male, slower in the
female, and both beat their wings at their sides while singing and the wings
beat at the same rate as their trill.
The white-throated obrero breeds in the austral summer. The species is monogamous
and the pair bond is long term, sometimes for life. The nest of the species
is typical for the genus, the breeding pairs of the flock make their nests one
over the other, giving it the appearance of a building with each nest entrance
being a floor. Such structures can have at least five “floors”, but other “buildings”
are made close to each other, which make the entire “building” look like a wall
with several holes. These nests are built in tree branches or over large rocks.
Pairs remain together throughout the year and will work on the nest during that
time. The first nests to be built will be those in the base of the “building”.
Those are usually made by the oldest birds in the flock and its construction
influences the others to start their own. A clutch generally contains two to
four eggs. The eggs are laid every second day and incubated for 14–18 days.
Chicks are fed for 23–26 days before fledging; young birds remain in the parental
flock, but can disperse to others around 6 months after fledging. Both parents
incubate eggs and feed the young. Obreros may or may not reuse nests, therefore
it is quite common to see several abandoned “buildings”. However, a formerly
unused nest may be repaired for a new breeding season. They reach sexual maturity
with 9 months and have a lifespan of 12 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Great
guyrapyhare (Pseudopicus magnus)
Order: Passeriformes (Passeriformes)
Family: False woodpeckers (Pseudopicidae)
Habitat: Atlantic coast of South America, forests.
During the Holocene, the human interference caused the extinction of many creatures.
However some were capable to survive. The woodcreeper (Dendrocolaptidae) survived
and in the Neocene a new family appeared from they, the family Pseudopicidae,
the false woodpeckers, this name is due the morphological similarities with
the woodpeckers. This family is formed by three genus: Pseudopicus (Great guyrapyhare),
Lophornithes (Crested guyrapyhare) and Pictopicus (Painted guyrapyhare). This
name descends from two Tupi words, guyra (bird) and pyhare (night). All the
members of this family are mostly active during the crepuscular hours, both
dawn and dusk. The great guyrapyhare is the largest member of this family and
lives in the forests of the Atlantic coast of South America.
The great guyrapyhare has 56 cm of length and a wingspan of 76 cm. The plumage
of this bird is mostly reddish brown with cream underbelly and throat. The feathers
of the tail are rigid and are used for supporting the body when climbing tree
trunks. The tail can support most of the body weight and birds that lose their
tail find climbing difficult. The guyrapyhares climb by flexing their legs and
hopping up the trunk. The feet of these birds are also modified for climbing,
guyrapyhares have developed zygodactyly feet. Its beak is grey, long, thick
and strong. It’s used like a chisel, breaking the bark on trees in search of
food. These birds also have relatively long tongues, but not as long as that
of a true woodpecker. Their saliva is very sticky, which helps them catch insects.
It feeds on the larvae and adults of certain poisonous wood-boring beetles,
ants and termites, but also eats other insects seeds, amphibians, lizards, and
fruits. Unlike the true woodpeckers, they aren’t capable of pecking holes in
the trees. The bird uses its large and strong bill to peel the bark off dead
trees or of nests to find the insects. Then the long tongue and sticky is used
to catch the prey. They will also use their beak to destroy bromeliads and lichens
in search of insects and amphibians hiding here.
These birds are monogamous, forming pairs for life. The breeding season occurs
during all the year when there is abundant food. The nest is made in a dead
or partially dead tree. The female lays 2 to 4 eggs. Parents incubate the eggs
cooperatively for 24 days. The young fled with 28 days. This species reach sexual
maturity with 2 years. The lifespan of the great false woodpecker is 18 years.
Other species in the genus Pseudopicus:
White-bellied
guyrapyhare (Pseudopicus leucogaster)
Living in the savannas of Central South America, this species have 52 cm of
length and a wingspan of 68 cm. Its plumage is similar to that of the great
guyrapyhare, with the exception of the belly which is white.
Amazonian
guyrapyhare (Pseudopicus amazonicus)
Living in the rainforests of the Amazon Basin, this bird has 54 cm of length
and a wingspan of 70 cm. It has an auburn plumage, with the exception of the
throat and underbelly which are pale grey.
Southern
guyrapyhare (Pseudopicus australis)
Living in the forests of Southern South America, this bird has 49 cm of length
and a wingspan of 59 cm. It has pale brown plumage with a dark grey underbelly.
These birds migrate to northern areas during the winter.
The genus Lophornites is formed by the crested guyrapyhares. These birds are
smaller than that of the genus Pseudopicus and are more terrestrial, feeding
on the ground. They include in their diet various toxic insects, such as beetles,
having developed immunity to them.
Andean
crested guyrapyhare (Lophornites andinus)
The type species of this genus, the Andean crested guyrapyhare lives in the
woodlands at the slopes of the Andes. These birds have 32 cm of length and a
wingspan of 48 cm. Its plumage is yellowish-brown with grey throat and chest.
Auburn
crested guyrapyhare (Lophornites rufus)
Living at the forests near the foothills of the Andes, these birds have 35 cm
length and a wingspan of 50 cm. Its plumage is auburn with dark brown underbelly.
Genus Pictopicus:
This genus includes painted guyrapyhares. These birds share a similar size to
the Genus Pseudopicus. It is formed by only one species.
Painted
guyrapyhare (Pictopicus leucopteron)
The only member of this genus, the painted guyrapyhare is a large bird, with
48 cm of length and a wingspan of 54 cm. This bird lives in the tropical forests
of the Panama Peninsula. It is mostly reddish-brown with white primaries and
secundaries feathers. It also has a yellowish brown underbelly.
This bird species was discovered by João Vitor Coutinho, Brazil.
Sapphire-crowned
saltarin (Atacaran splendidus)
Order: Passerine birds (Passeriformes)
Family: Manakins (Pipridae)
Habitat: “terra firma” forests of Amazon region.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The golden-crowned manakin (Lepidothrix vilasboasi),
a species with a hybrid origin between the opal-crowned manakin (Lepidothrix
iris) and snow-capped manakin (Lepidothrix nattereri), survived the anthropogenic
onslaught by finding refuge in inaccessible areas of the Amazon rainforest.
There, this species continued to evolve, giving rise to the Genus Atacaran,
from the Tupi tata: walk and arã: around. It’s a reminder
of the courtship display made by the males, which is several jumps. It also
has genes of other Lepidothrix birds that ended up stranded in these forests
and hybridized to a lesser extent with the golden-crowned manakin.
When the forest started to recover, this genus spread to other areas of the
rainforest. The type species is the sapphire-crowned saltarin, an inhabitant
of the “terra firme” rainforests understory of the Amazon Basin. The word saltarin
is from Castellan language and refers to the male’s jumps during courtship.
The sapphire-crowned saltarin is a small bird, they have 9 cm of length. Like
all the manakins, all the representatives of the genus are sexually dismorphic.
The male have bright green feathers in their upperparts, while the underparts
are bright orange. They have a blue crown, which is the result of structural
colors produced by different nanostructural organizations of the feather barb
keratin matrix. Their secondary feathers are bright yellow. The female is mostly
dull green, with a pale yellow in the belly. They lack the blue crown.
Blue-crowned saltarins are frugivores. Their diet includes fruit from Melastomataceae,
Moraceae, Bromeliaceae, and Araceae. They can participate in mixed flocks, with
their preferred food being arthropods, including ants, flies, and other small
insects. While catching arthropods, their hunting technique was often a sally-strike
or sally-glean off of live foliage.
Breeding season coincides with the dry season. Males form leks of up to seven
males. Adult and juvenile males form territories though juveniles are unable
to maintain their own territory. Their territories range from 209 to 5050 m²
in size. They defend their territory and attract mates through song. They perform
their songs on song perches that are horizontal or slightly angled twigs. Their
display courts are 3–5 m in diameter and are located close to the ground in
the open understory. They use up to two courts at a time, but courts can change
location annually. Their courtship consists of the males constantly jumping
around the female. They will bow to her, showing their blue crowns and opening
their wings to reveal their yellow secondaries. Female’s home ranges overlap
with one lek on average.
Sapphire-crowned saltarin make simple, open-cup nests using dry palm leaves
and bark externally. The internal lining of their nest is pale brown, but can
also be whitish or yellow. They use spider webs to secure their nests to the
tree. They chose small shrubs and treelets that are less than 1 meter tall and
place their nests on horizontal forks. Females are the sole nest builders and
take care of their offspring alone. They lay two eggs per clutch. She incubates
them for 19 days. The chicks fledge within 21 days, but remain being fed by
the mother for more than three weeks. Sexual maturity is reached with 7 months
and they have a lifespan of 5 years, but males are killed by predators before
that.
The Sapphire-rumped
saltarin (Atacaran serranus) is another representative of this genus.
This species inhabits the forests in mountainous areas of the Amazon, replacing
the sapphire-crowned saltarin in these areas. They have 8,5 cm of length. The
plumage of the males is similar to that of their relatives, with the exception
of their bright blue rump and white primaries and secondaries.
This bird species was discovered by João Vitor Coutinho, Brazil.
Turquoise
panapana (Panapana azurea)
Order: Passerine birds (Passeriformes)
Family: Cotingas (Cotingidae)
Habitat: South America, evergreen Atlantic forests.
The extinction of the humans had been followed by restoring of habitats destroyed
in human epoch. When natural conditions stabilized, new species of plants and
animals emerged. One of these species was the Turquoise panapana, a descendant
of one representative of the genus Cotinga, the common bird genus of South American
forests in human epoch. Some cotingas had been able to live in heavily destroyed
forests, and their descendants are rather common in Neocene.
Living in the forests of the Atlantic coast of South America, the turquoise
panapana is a small bird 15 cm long, and it is a type representative of its
genus. Even being a small bird, the bright colors of the males make them easily
visible. They eat small insects and fruits, being commonly seen regurgitating
seeds. So they are among the important distributors of small seeds of some tropical
trees. They rarely descend to the ground, preferring to search for food in the
forest canopy.
Males have black feathers in the rump, and their breasts are dark blue. But
what makes them flashier is the bright turquoise color present in their tail,
head and in the primaries and secondaries. The females are greenish-gray: this
color allows them to camouflage while incubating the eggs. Both sexes have black
short beaks and legs.
During the breeding season, the males form leks of 10-12 individuals to perform
the courtship. The courtship starts with the male vocalizing to attract females
to them. When the needed number of females gathers, they begin to make short
flapping flights with wings and tails widely opened. Making series of short
and fast wing flaps, they hover in air like butterflies. These movements also
cause the wind to pass through the modified feathers present on its tail and
wings, producing sounds similar to a “whistling”. The male that performs the
“butterfly flight” for longer time is the winner.
Their characteristics of forming leks and “butterfly flight” gave them the name
“panapana”, which in the Tupi language means “cloud of butterflies” or “swarm
of butterflies”. During the courtship flight males are vunerable to attacks
of predatory birds and bats, so they listen for alarm calls of females and interrupt
their flight immediately. Each male pairs with some females during the courtship
performance.
After the courtship the female makes a small shallow cup-like nest made of twigs
in tree fork and lays two white eggs. The incubation lasts for 18 days and the
care for the young is undertaken by the female alone. Hatchlings are fed by
the female with semi-digested fruit pulp and insects until they have 3 weeks.
After that time they are fed with solid food and are able to swallow entire
berries and small fruits like adult birds eat.
The youngs fledge being 4 weeks old and keep with the female for 2 weeks. At
this age young males and females look similar to each other; the only difference
is that the males have shades of blue in their breasts. They reach the sexual
maturity at the age of 8 months – at this age young males acquire the coloration
of adult birds.
Their lifespan is about 7 years.
Genus Panapana includes some more species differing from each other mainly in
colors of male feathering and features of bird voices. Females of different
species look very similar to each other.
Violet
panapana (Panapana violacea) is closely related to previous species
and shares a common ancestry. This species is 14 cm long and males have bright
violet feathers in tail and head, the same color is present in their primaries
and secondaries. Other parts of their bodies are of dark gray color. They live
in the coastal rainforests of northern part of South America, from the Atlantic
coast to the Caribbean Sea coast. Part of their range along the Atlantic coast
overlaps part of the range of turquoise panapana; in this area hybridization
may occur and birds of intermediate color appear sometimes. Violet panapanas
form leks of 7-9 individuals.
Golden panapana
(Panapana aurea) has the most contrast coloration compared to related
species: male of this species has bright yellow feathers in tail and head, the
same color is present in their primaries and secondaries, and also in covert
feathers. Body feathers and legs are coal-black. Usual length is about 11 cm.
These birds live in the northern part of Amazon basin and prefer wetland forests.
They form leks of 12-15 individuals.
Scarlet panapana
(Panapana punicea) differs in almost strict frugivory, its beak is
short and deep, and mouth cut is larger than at related forms. Adult male of
this species is 11 cm long and has bright red feathers in the tail and head.
Tips of its primaries and secondaries are also of the same color, and lower
parts of these feathers are black, like the color of the body. Some coverts
of male’s wings are bright red on black background. They inhabit the southern
part of Amazon basin. Males form leks of 10-12 individuals.
Emerald panapana
(Panapana viridis) has a range isolated from other species of the genus:
it lives in the montane woods near the Andes. Being 10 cm long, male of this
species have bright green feathers in the tail and head, and also in wings.
Body color is chocolate-brown, and iris of the eye is white. They have less
dense populations compared to lowland species and form leks only of 4-7 individuals.
This species is also more insectivorous compared to lowland forms, and has longer
and thinner beak.
This bird was discovered by João Vitor Coutinho, Brazil.
Magenta
screaming anambe (Vociferornis roseoventer)
Order: Passerine birds (Passeriformes)
Family: Cotingas (Cotingidae)
Habitat: tropical forests of northern and northeastern parts of South America.
The human actions in the Holocene caused many impacts in the ecosystems. Because
of these actions, many species went extinct in the Neocene. However some species
survived and give raise to descendants, one of these descendants is the rose
screaming anambe (in Brazil, the word anambe was a common name used to birds
of the Family Cotingidae). A descendant of the Genus Lipaugus, the magenta screaming
anambe lives in the tropical forests.
These birds are sexually dysmorphic, with males being large and more colorful
than the females. They have 68 cm of length and a wingspan of 100 cm, while
females have 62 cm of length and a wingspan of 96 cm. The males have throat,
underparts and wings magenta-colored. The head and back are dark grey, while
the tail is yellow. The females are mostly dull grey, with rufous in the throat
and underparts. The young males are similar to the females until reaching sexual
maturity with 3 years. The beak and legs are dark grey.
Magenta screaming anambes feed mostly on fruits and berries, however flower,
insects, spiders and small amphibians are eaten opportunistically. This bird
never leaves the canopy, with all food being obtained there.
These birds are solitary, forming pairs for a brief period after mating. The
nesting season occurs during the end of the dry season. Males gather in large
leks and vocalize to attract the females. Their song is extraordinarily loud,
reaching even greater distances than that of the fiery
fruitcrow. Nests are built in trees or treeferns, high above the ground.
The female lay two white eggs and is the solely responsible for incubate them,
the male will leave her after the eggs are laid. The incubation lasts for 27
days. The chicks fledge with 30 days. The lifespan of this species is of 20
years.
Other species in the genus Vociferornis:
Bare-throated
screaming anambe (Vociferornis nudicollis)
Living in the forests at the foothills of the Andes, the males of this species
have 57 cm of length and a wingspan of 95 cm, while females have 50 cm and a
wingspan of 87 cm. Males of this species have a characteristic bare red patch
of skin around its eyes and beak and on its throat. Their plumage is similar
to that of the rose screaming anambe. Females of bare-throated screaming anambe
have plumage similar to that of the other species.
This bird species was discovered by João Vitor Coutinho, Brazil.
Purple
magnificent bellbird (Magnoprocnias purpuraceus)
Order: Passerine birds (Passeriformes)
Family: Cotingas (Cotingidae)
Habitat: South America, rainforest canopy in Amazonia.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. The
Genus Magnoprocnias, the magnificent bellbirds, is formed by descendants of
New World bellbirds that survived the anthropogenic onslaught during the Holocene.
The type species of this genus is the purple magnificent bellbird. This bird
is found in the Amazon Rainforest, only being seen in the canopy.
The purple magnificent bellbird is a sexually dismorphic bird, with males having
characteristic large purple wattles in the face and throat. The female lacks
such structures. The males have 42 cm of length and a wingspan of 63 cm, while
the females have 37 cm of length. Both sexes have broad dark gray hooked-tipped
bills, rounded wings and strong gray legs. The plumage of the male is mostly
white, with a brown back and rump. Their wings are green. The females are duller
in color with a black crown, olive-brown upper parts and yellowish underparts
streaked with olive green.
The species is completely frugivorous, swallowing the fruits from trees. The
magnificent bellbirds perform an important role in seed dispersal. It regurgitates
the seeds it cannot digest and deposits them in gap areas beneath song perches,
which nearly doubles seedling survival rate.
The purple magnificent bellbird is mostly solitary, living spread across the
canopy and only forming small flocks when there is an abundance of fruits. Their
breeding season starts during the beginning of the rainy season. The males will
fly up to the emergent layer of the forest and will vocalize loudly with a characteristic
sharp sound like that of a hammer striking an anvil. The sound is so loud that
it can be heard up to a mile away, being louder than a thunder. Once a female
approaches him, the male starts a presentation to the female. He will present
to her his purple wattle, the female will choose the most colorful males. The
same tree branch is used by the male for his presentation during all his life
and until the end of the breeding season, they will have attracted several females.
The construction of the nests and parental care is made only by the female.
She builds her nest between 6 and 10 meters from the ground in the form of a
small cup and decorates it externally with lichens and fungi, which works as
a camouflage. The female lays 2 eggs that are incubated by her for 23 days.
They fledge after 19 days and remain with their mother for two more weeks. Sexual
maturity is reached with 2 years, the time that the males develop the adult
plumage and wattles. The lifespan of this species is 18 years.
The Andean
magnificent bellbird (Magnoprocnias andinus) is another member of the
Genus Magnoprocnias. This bird inhabits the cloud forests in the slopes of the
Andes, descending to lowland forests during the winter. Like all species of
this genus, they are also sexually dismorphic. The male is mostly white, with
brown wings. They have 37 cm of length and a wingspan of 52 cm. Their characteristic
wattles are reddish-orange. The female has a plumage similar to that of the
purple magnificent bellbird, but is smaller, with 31 cm of length. Another representative
of this genus is the Red
magnificent bellbird (Magnoprocnias puniceus). This species inhabits
the Atlantic forests of Southeastern South America, reaching areas 760 m high.
The males have red wattles in the face and neck. The plumage is white in the
back and rump, while the chest and belly are brown. The wings are green. The
females are similar to that of the other two species of the genus. The males
are also larger than the females, with 40 cm of length and a wingspan of 58
cm. The females are 37 cm in length.
This bird species was discovered by João Vitor Coutinho, Brazil.
Fiery
fruitcrow (Calidornithes vociferans)
Order: Passerine birds (Passeriformes)
Family: Cotingas (Cotingidae)
Habitat: humid subtropical and tropical forests of South America.
The human actions in the Holocene caused many impacts in the ecosystems. Because
of these actions, many species went extinct in the Neocene. However some species
survived and gave rise to descendants, one of these descendants is the fiery
fruitcrow. This animal is a descendant of the birds in the Genus Cotinga.
Fiery fruitcrows are large passerines; the males can reach 40 cm o length with
wingspan of 65 cm, while females have 34 cm with wingspan of 60 cm. Males are
more colorful than the females, they have bright reddish orange feathers covering
their body with exception of the belly, tail and primaries feathers that are
black, they also have a bright yellow beak. The females have olive plumage with
yellowish green underparts and their beak is grey. Both sexes have grey legs
and feet. The young males are similar to the females until reaching sexual maturity,
which occurs with 2 years.
The diet of these birds is largely composed of fruits, the fruits of large seeds
are swallowed whole and the seed are regurgitated while the small are expelled
in the feces. Fiery fruitcrows will occasionally eat insects.
This species lives mostly on the canopy of the forests, rarely coming to low
levels. They are polygamous, during breeding season the males form loose leks
and start vocalizing to attract the females, the sound produced is loud and
can be hear for kilometers. Nests are built high in trees, far above the ground.
The female lays two creamy colored eggs, which are incubated only by her for
27 days. Only the female cares for the chicks, feeding them with invertebrates
and regurgitated fruits. The young fledge usually 23 days after hatching. The
lifespan of this species is of 14 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Plant
louse shrewbird (Aphidiornis aphidiphagus)
Order: Passerine birds (Passeriformes)
Family Shrewbirds (Aphidiornitidae)
Habitat: tropical woods of South America, forest canopy at height about 30 meters
above the ground level and higher.

Picture by Pavel Volkov
|
Initial image by |
Picture by Arseny Zolotnikov, |
The tropical wood occupied by set of species of live creatures,
represents the favorable environment for evolution. Here, in conditions of rigid
competition and variety of ecological niches the occurrence of highly specialized
species is more probable.
In South-American selva many specialized varieties of passerine birds had appeared;
mainly they are the representatives of so-called calling passerines (not concerning
to warblers). And the tiniest representative of this group, plant louse shrewbird,
by the size makes a competition to smallest species of hummingbirds. But, as
against the hummingbirds eating mainly nectar, this bird eats exclusively small
invertebrates, representing the kind of “flying shrew” in crones of tropical
wood. This bird never flies down to the ground, keeping in crones of trees.
The body of plant louse shrewbird is covered with rust-colored feathers; wings
are motley black with brilliant blue tips of feathers forming mesh pattern.
Wings are narrow and pointed. Flight of shrewbird is similar to flight of insects:
the bird makes up to 30 waves by wings per one second, uttering a silent buzzing
in flight. On the back between wings there is an area of bright orange feathers,
needing for recognition of neighbours.
The head seems large in relation to a body. At females it is of the same colors
as the body, at the male there is small “cap” of brilliant grayish feathers.
The beak is straight and long (being twice longer than other head, it is almost
equal to the body length); it serves as “tweezers” for gathering tiniest insects
and spiders - the food of this bird.
It is very territorial bird protecting fodder sites from neighbours. Food of
plant louse shrewbirds includes tiny soft-bodied insects, mainly plant louses
and termites. Each bird occupies territory on which there are some branches
with colonies of plant louses, and each day visits them by turns, feeding on
each colony. Sometimes this bird follows large insectivorous animals, and feeds
in insect colonies ravaged by them. Then near one animal some shrewbirds may
gather, but they prefer to keep distance to each other, and observe strict sequence
at feeding.
Metabolism of this bird is very intensive: for day the bird eats three times
more various small animals, than it weighs itself. Some other attributes of
bird are connected to features of physiology. So, the shrewbird falls into catalepsy
for night. At this time the bird perches to thinnest branches that it could
not be got to clambering on predators, seizes branch by claws, and simply hangs
upside down till morning. Breath and palpitation of sleeping bird are strongly
slowed down, and at this time the body of bird is cooled up to an ambient temperature.
The basic enemies of this species are small snakes, large climbing amphibians
and small mammals. Protecting itself from them, plant louse shrewbird utters
the sound of very high frequency passing to ultra sounds. It strongly irritates
sensitive hearing of predatory animals. If it does not frighten off predators,
the bird protects, striking impacts by beak in eyes and nostrils of predator.
Plant louse shrewbird lives its life some times faster, than large birds live.
It can nest up to five times per year. Nest of this species of birds represents
an accurate basket on branches which both parents plait of vegetative fibers
and spider webs. In clutch there are 4-5 eggs, but up to nest leaving only 2-3
fledglings survive. Development is very fast: the incubating lasts about one
week, nestlings develop in nest two weeks, and the young bird becomes sexual
matured at the age of 3 months, and general life expectancy is only about 2
years.
This species is discovered by Arseny Zolotnikov.
Butcher-kiskadee
(Confectorarius venator)
Order: Passerine birds (Passeriformes)
Family: Tyranns (Tyrannidae)
Habitat: South America, forests and savannas.

Picture by Pavel Volkov
Human interference during the Holocene caused the extinction
of many species. However some creatures were capable of surviving, one case
was the great kiskadee (Pitangus sulphuratus). These birds survive and in the
Neocene they give rise to the Genus Confectorarius (Confectorarius means slaughter;
butcher in Latin). The type species of this genus is the butcher-kiskadee, which
lives in tropical areas of South America.
These birds have 34 cm of length and a wingspan of 50 cm. The head is black
with a strong white supercilium and a concealed bright red crown stripe, which
is less bright in the females. The birds can make their crown stripe visible
during certain ocasions. The upperparts are greenish brown; the throat and breast
are rufus. The belly is yellow. The black beak is thick, long and has a sharp
hooked tip, a characteristic common to all species of the Genus. Their feet
are pale grey.
These birds feed on insects, arachnids, frogs, lizards, rodents and even other
birds. Butcher-kiskadees will also attack nests of other birds to eat their
chicks. The prey is caught in the middle of the vegetation. To kill the prey,
they use the strong beak to knock them against branches or rock. Small prey
is eaten whole, while large prey is eaten in small pieces which the bird take
from it by using their sharp beak while holding it with its legs. These birds
will store food by dismembering the prey in pieces and hidding it in tree holes
and rock crevices, sometimes distant from each other, to be eaten another time,
similar to what the shrikes (Laniidae) do with its prey. Is rare, but sometimes
the butcher-kiskadee can feed on fruits.
It lives in couples, during the breeding season the males sing intensely to
attract the female. If she becomes interested, she will fly to where he is singing.
Then, the male will vocalize while opening his wings and bowing his head to
the female, showing his red crown to her. If she accepts him, the female will
imitate the male. After that the birds will fly together while they vocalize.
These birds are aggressive, attacking other butcher-kiskadees in its territory.
Most of the construction of the nest is work of the female, which uses plant
fibers in the making of an open and deep bowl, coated with mosses. The nest
is well secured in a fork of trees with the aid of spider webs. She lays white-red
eggs with purple speckles. The male and female take turns incubating for about
19 days and feed the chicks. The young fledge with 21 days and reach sexual
maturity with 2 years. The lifespan of this animal is 13 years.
Other species in the Genus Confectorarius:
Streaked
butcher-kiskadee (Confectorarius cirratus)
Living in the tropical and subtropical forests from the Panama Strait to the
Florida Peninsula, this bird has 30 cm of length and a wingspan of 46 cm. The
head is dark brown with a concealed bright yellow crown patch, white supercilium
and dusky eye mask. The crown in the females is less bright. The upperparts
are greenish-brown with darker brown streaks on the back and rufous edges on
the wings, and wide white edges on the rump and tail. The underparts are yellow
streaked with brown.
Migratory
butcher-kiskadee (Confectorarius migratorius)
This species lives in the temperate areas of North America, including the Great
Plains. The Northernmost point of its distribution are the forests near the
Mishe-Nama lake. The migratory butcher-kiskadee has 32 cm of length and a wingspan
of 48 cm. The head is grey with a concealed bright reddish yellow crown patch,
white supercilium and dark brown eye mask. The crown in the females is less
bright. The upperparts are greenish-brown with dark grey streaks on the back
and rufous edges on the wings, and chestnut edges on the rump and tail. The
underparts are pale yellow streaked with rusty brown. During the winter, this
species migrates to Cuba, Great Antigua, Panama Peninsula and the tropical forests
of Northern South America.
This bird species was discovered by João Vitor Coutinho, Brazil.
Insular
rocket-tailed tyrant (Tyrannoelegans insularis)
Order: Passerine birds (Passeriformes)
Family: Tyranns (Tyrannidae)
Habitat: Archipelago of Fernando de Noronha, forests.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The genus Tyrannoelegans is part of the Tyrannidae Family.
These birds originated in the savannas of South America, but some species have
adapted to different biomes. One of the members of this genus is the insular
rocket-tailed tyrant, an endemic species of the Archipelago of Fernando de Noronha.
The insular rocket-tailed tyrant has 18 cm of length and a wingspan of 38 cm.
This species is sexually dimorphic, with the males having bright reddish-orange
underparts, while the rest of the plumage is dark brown. Their head is black
with a strong white supercilium and a concealed yellow crown stripe. The females
are drab and have a peach-colored belly with a dark gray upperside. Their head
is pale brown. The main characteristic of the genus are the conspicuous elongated
outer feathers on its tail, which have long streamers ending in paddle-shaped
tips. Both sexes have this characteristic but the length is different. In the
males it is 10 cm, while in females the length is 8 cm.
These birds are mostly insectivorous, but will occasionally eat small berries.
When hunting insects, they perch openly in trees, several meters above ground.
From such perches, they will sally out for considerable distances to catch insects
in flight, to which purpose they utilize a range of aerobatic maneuvers. They
also regularly hover and glean for prey. Such predatory behavior is shared with
other members of the genus.
The insular rocket-tailed tyrant is a monogamous bird, with the pairs defending
a territory against members of the same species. These birds are aggressive
around predators, mobbing them and alerting nearby prey with their aggressiveness.
This species breed during the spring and summer. Both sexes build a large domed
nest that has a side entrance. It's chiefly composed of grasses and small twigs.
The nest is placed in a wide range of sites, often in an exposed position high
up in a tree, but occasionally the nest is placed in a cavity. The clutch is
3 to 4 eggs. These are moderately glossy, light yellowish cream in colour with
purplish black and purplish brown spots. Only the female incubates the eggs;
the male guards the nest while she leaves the nest to feed. The eggs hatch after
16–17 days. The chicks are fed by both parents and fledge after 17-18 days.
They reach sexual maturity with 11 months and have a lifespan of 9 years.
The insular rocket-tailed tyrant closest relative is the scarlet
rocket-tailed tyrant (Tyrannoelegans rubroventer), a inhabitant of
the Atlantic forests of Southeastern and Northeastern South America. They have
22 cm of length and 40 cm of wingspan. The males are similar to that of the
insular species, with the exception of the vivid and bright red underparts that
are present in the continental species. The plumage of the female of this species
is also similar to that of the other. Another species of the genus is the Amazon
rocket-tailed tyrant (Tyrannoelegans amazonicus). This species inhabits
the rainforests of the Amazon basin. They have 20 cm of length and 38 cm of
wingspan. The males of this species have bright orange underparts.
This bird species was discovered by João Vitor Coutinho, Brazil.
Lamplighter
bird (Lanternariornis nocturnus)
Order: Passerine birds (Passeriformes)
Family: Bowerbirds (Ptilonorhynchidae)
Habitat: Meganesia, tropical rainforests.

Picture by Sauron from FurNation
In Neocene Australia and New Guinea had merged to unite continent
Meganesia. Under action of internal forces of Earth crust since human epoch
this continent had moved to the north, to the area of equatorial climate. It
had caused the occurrence of extensive and almost impassable rainforests at
the north of the continent. These forests are inhabited by set of odd-looking
and original species of animals and plants. One of the most surprising inhabitants
of these forests hides from sunlight in day time, and leaves the refuge only
at night. Its life passes in pale and greenish light of some species of luminescent
mushrooms growing on fallen and rotten trunks of trees. This creature is a special
kind of bowerbirds named lamplighter bird.
Usually males of bowerbirds build for courtship rituals original buildings,
“bowers”. In human epoch these constructions varied from the primitive round
platform cleared away from leaves on which fresh leaves are scattered up to
complex and very large in comparison with the bird constructions decorated with
various bright objects. Actually, these constructions represent an element of
the courtship dress of the male, transferred from the bird to objects of world
around.
In Neocene one species of these birds had replaced brightness of colors and
berries shining in sunlight to pale light of some species of luminescent
mushrooms settling on rotten wood. This bird began in literal sense to cultivate
luminescent mushrooms which grow plentifully and shine on building of this species.
For this feature the nocturnal species of bowerbirds is named lamplighter bird.
The lamplighter bird is small – it is not larger than any medium-sized finch.
It has thickset body on long legs, tail of moderate length and straight strong
beak. At males there is a small cop on head. Remarkable feature of birds of
this species is large eyes shining in darkness like at some nocturnal mammals.
In retina of lamplighter bird there is a special layer of cells containing guanine
which returns to photosensitive elements of retina light penetrating through
them.
The feathering of these birds has soft colouring; sexual dimorphism is clearly
expressed. Male has pale green color similar by shade to the light emitting
by mushrooms. The female which raises nestlings alone, has camouflage colouring
– brown with longitudinal black strips on feathers of wings and longitudinal
white strokes on feathers of head and chest.
Females and males differ in way of life. The female not busy in care of posterity,
is similar by habit of life to the majority of nocturnal insectivorous animals.
It hunts insects and other invertebrates living in wood litter and rotten wood.
Male till the most part of adult life is occupied with unique major work – it
builds a construction which at this species replaces magnificently decorated
complex bowers characteristic for other species of bowerbirds. Construction
of lamplighter bird represents a heap of pieces of rotten wood, heaped up between
plank-buttress roots of any tree. But it is only a basis for the main ornament
of construction because of which the lamplighter bird is named. When the heap
reaches the enough size, bird drags on pieces of rotten wood some luminescent
mushrooms found in forest. First time male is compelled to change frequently
mushrooms, because they, quite naturally, do not take roots and decay quickly.
But in some weeks the situation chamges. From the decayed fruit bodies of mushrooms
spores spilled out, which have sprouted and have formed a viable mycelium. And
diligence of lamplighter bird male get married success – the mycelium forms
fruit bodies which start to shine at night by strange ghosty chartreuse light.
The bird constantly looks after the construction – it regularly brings new pieces
of rotten wood, and accurately pecks out from mushrooms various insects flied
to feed on mushrooms. These insects make a significant part of ration of the
male of lamplighter bird.
With the first beams of sun when day time animals wake up, lamplighter birds
hide in shelters. The female usually spends day time time in hollows of trees
or under bark peeled off. Male chooses as shelter burrows and holes under roots,
usually near its construction. Frequently it gets into the tree trunk rotten
from within.
At night courtship displays of lamplighter birds begin. Male calls the female
by melodious single cries, audible far in night forest. When near male’s “mushroom
garden” the female appears, male begins courtship dance. It keeps between female
and construction, and light of mushrooms precisely depicts his silhouette. Самец
“bows” to the female, slightly spread wings and having stretched vertically
rised tail like a fan. If the female accepts his court, fast pairing follows,
and the male loses interest to this female.
The female broods clutch of 3 – 5 eggs, and raises hatch alone. She builds a
nest in tree hollow. Bringing up posterity, the female leaves a nest before
the sunset, and last feeding of nestlings takes place one – two hours after
sunrise. The female looks after posterity till nine – ten weeks while young
birds stay in the nest, and finishes the feeding of young birds till about one
week after they leave nest.
Young birds become sexually mature at the age of ten months.
The idea about existence of this species is proposed by Simon, the forum member.
Gardener
bowerbird (Hortulornis plantarum-cultor)
Order: Passerine birds (Passeriformes)
Family: Bowerbirds (Ptilonorhynchidae)
Habitat: rainforests at the north of Meganesia, areas of interrupted forest,
riverbanks.
The courtship behaviour of birds may turn odd forms, and according to them the
shape of the bird changes. At males of various species of birds freakish bright
feather “ornaments” grow, or colouring becomes bright and appreciable from apart.
But at representatives of bowerbirds family evolution has resulted in “replacing”
of features of courtship behaviour to extraneous object – artificial “construction”
which has actually replacing by itself a courtship dress of male. In Neocene
descendants of any species of bowerbirds with primitive building behaviour had
replaced artificial constructions to successfully cultivated living organisms
– usually in this way plants are used. They had actually transferred functions
of secondary sexual attribute from lifeless objects (various brightly colored
objects) to live plant. The result of such adaptations is simply surprising.
One of species of family, lamplighter
bird, began to lead nocturnal habit of life, and involves the female, cultivating
luminescent mushrooms on simple heap of rotten pieces of wood. The birds of
close species leading a traditional diurnal habit of life, use for similar courtship
displays of plants with bright leaves.
One of species having such features of behaviour is the gardener bowerbird,
ground bird smaller than sparrow in size. Male and female of this species distinguish
in sharply expressed sexual dimorphism. Male has black-and-white colouring -
white head with black nape and “cap”, white breast and stomach, black covert
feathers and tail, and black-and-white cross-striped primarily feathers. The
female is brown with black specks on wings, feathers of nape and back. At birds
of both genders there is thick beak of grey color. Iris of eyes is bright red
at male and yellow at female.
Gardener bowerbird developed close symbiotic relations with one species of balsam
- crimson balsam (Impatiens cinnabarifolia), grassy
plant of underbrush. This species of plants grows better at enough of light
– in sites of damaged forest, and also at riverbanks. Therefore males of gardener
bowerbird meet in these places more often.
Male of the bowerbird lives near the plant and carefully looks after it: bird
pecks out pest insects, throws out dry leaves falling on plant from trees, breaks
off dry and ill leaves, and also regularly rakes under stem friable fallen leaves.
It stimulates formation of adventitious roots at the plant, and it grows better.
The bird’s dung serves as additional fertilizer for the plant. The most surprising
feature of behaviour of gardener bowerbird is the skill of plant forming. Bird
picks by beak a top of main stalk, and thus forces it to branch. Leaves of crimson
balsam have bright anthocyan colouring – they are green with numerous red spots,
and the youngest leaflets are completely ruby-red. Male of gardener bowerbird
controls the growth of patronizing bush, from time to time pinching out tops
of too strongly grew sprouts and biting off part of stalk by strong beak. Such
operation results in development of the plant as a compact bush. Well formed
bush of this species has a plenty of tops and looks more impressive. On its
background the bowerbird displays black-and-white plumage. At the strong adult
male some plants form semicircular “stage”.
The female of gardener bowerbird visits “flower bed” of the male only for pairing.
She leads a secretive way of life, and prefers to hide in hollow or among branches
in day time. She is most active from dawn till midday, and also in the evening.
When the female comes nearer to male’s “flower bed”, she utters silent calling
whistle. Having heard it, male as if changes itself. He starts to fly excitedly
from branch to branch near “flower bed”, searching for the female. If at his
territory another male, or a bird even remotely similar to him by colouring
will appear at this time, male will attack them and banish. Meeting the female,
the male perch on plant grown up up by him, shakes stalks, and utters chirring
advertisement call alternating with loud resonant calls. Pairing takes place
on the ground near plants.
Male does not take part in care of posterity, but protects the site from other
males. As a rule, at the territory of one male one or two females may nest.
Nest of these birds is simple cup-like one; it is made of thin twigs and sturdy
fibrous leaves of giant grasses. The female chooses rich bushes for nesting
and builds a nest itself. In clutch of this species of birds there is up to
six rounded eggs with dark green smooth shell. Nesting repeats two times per
year.
The idea about existence of this species of birds is proposed by Simon, the forum member.
Mangrove
bowerbird (Onychoptilonorhynchus caementator)
Order: Passerine birds (Passeriformes)
Family: Bowerbirds (Ptilonorhynchidae)
Habitat: Meganesia, mangroves of Lake Arafura.
In the human era, the bowerbirds were famous for their art of erecting unusual
buildings, serving as a kind of “courtship dress” transferred from a live bird
to objects of the surrounding nature. The caprice of sexual selection led to
the improvement of the building skills of these birds, and in the process of
evolution, each bird species acquired its own type of buildings, differing from
those of other species to the same extent as anatomical and physiological characteristics.
In the human era, some of the bowerbird species became extinct due to the destruction
of their habitats. A few species survived that were able to coexist with humans
relatively successfully, and in Neocene they became the ancestors of various
species that developed equally unusual construction skills as human contemporaries.
Avoiding competition, one bowerbird species settled in the vast mangrove forests
separating Arafura Lake from the ocean. This species is a mangrove bowerbird
– a bird of inconspicuous appearance, but with a very peculiar construction
behavior.
This bowerbird species is a relatively large, jackdaw-sized bird. Wings and
tail are quite short, and the legs are relatively large, with tenacious toes
and large claws: mangrove bowerbird flies a little, but spends a lot of time
among the roots and trunks of mangrove trees, climbing on them. The startled
bird takes off and can fly several tens of meters, but prefers to hide among
the vegetation. Sexual dimorphism is very clearly expressed in the color of
the plumage. In this species, the male has emerald-green plumage; he has a black
back and a bright red loin. The iris of the male’s eyes is white. The female
has brown plumage with fine black ripples, and black eyes. Birds of both sexes
have grey legs. A peculiar feature of this species is the beak – deep, strongly
compressed from the sides, glossy black, especially large in the male.
The main difficulty of living in mangrove forests for terrestrial animals is
the lack of solid ground under their feet. The soil of the mangrove forest is
viscous silt that hides under water twice a day. Therefore, if the bird itself
easily escapes from the advancing water on the tree, then any ground buildings
are completely destroyed by tidal waves. Mangrove bowerbird got out of the predicament
by starting to erect its buildings on trees.
The basement of the building consists of branches thrown over the roots of mangrove
trees above the tide level. A layer of thin rods forming the base of the platform
is laid on top of them. The next stage is the construction of a platform for
courtship dances. The bird levels it by laying a layer of “cement” from a mixture
of silt, clay and the droppings of the bird itself on top of it. At this stage
of the work, the deep massive beak serves as a tool for the male: the bird scoops
up silt with it and smears it on the rods, mixing it with the bird’s own droppings
and clay. The area of this structure can be impressive: up to one square meter.
When the dance floor acquires the desired smoothness, the mangrove bowerbord
begins to finish: it pulls the elytra and even whole specimens of bright-colored
beetles onto the platform and tramples them into fresh “cement”, having previously
torn off and eaten their abdomens. Then the bird begins to gather a collection
of shells, searching the branches of trees and the tidal zone. The bird eats
live snails, carefully picking them out of the shells. Picking up a collection
of shells, the male prefers whole specimens with light or variegated coloring,
ignoring dark and broken shells. The shells are stacked in heaps of color, separate
beautiful specimens lie singly.
Having composed a collection, the male attracts females with loud calls, perching
on the branches of mangrove trees above the dance floor. Displaying in front
of the female, the male lowers the front part of the body, opens the tail like
a fan and fluffs the red feathers on the lower back, which contrast favorably
with the black color of the back and the white iris of the eyes. In this position,
the bird flutters its wings finely, uttering a rhythmic ticking sound. Suddenly
interrupting the courtship display, the male grabs the shells and shows them
to the female.
After mating, the female builds a simple cup-shaped nest among the branches,
into which she lays up to 3 eggs with a spotted shell, and incubates the offspring
alone. During the laying process, she returns to the male 1-2 more times and
mates with him, but can also mate with other males. Incubation lasts 20 days,
the nestlings hatch naked and blind. They fully fledge at the age of 40 days
and leave the nest. Seasonality in reproduction is not expressed; the female
feeds 2 broods per year. Sexual maturity occurs at the age of 10 months, life
expectancy is up to 8 years.
In the marshy areas in the east of Arafura Lake, a close species lives, the
eastern swampland bowerbird (Onychoptilonorhynchus plumophilus).
It differs from the mangrove bowerbird in larger sizes: the body length is about
40 cm. The plumage of the male is grassy-green, the back is red. The female
is similar in plumage color to the mangrove bowerbird. A noticeable feature
of the difference in the appearance of these species is the color of the legs:
in the eastern swampland bowerbird they are pale yellow in birds of both sexes.
The beak of this bird is also deep and compressed from the sides: the male makes
similar buildings from a mixture of clay, dung and silt on a foundation of twigs.
But the decoration of these buildings is made of feathers of various birds,
which the male sticks into the wet material of the building in two large groups,
and dances between them.
The idea of the existence of this bird species was proposed by Timothy Donald Morris, Adelaide, Australia.
Big-legged
chicken wren (Dromomalurus megapodus)
Order: Passerine birds (Passeriformes)
Family: Running malurs (Dromomaluridae)
Habitat: savannas and semi-deserts of Meganesia
In the human era, Australian zoogeographic realm was characterized by birds
of Maluridae family, which some ornithologists regard as relatives of the sylviid
babblers of Eurasia. Small and unpretentious, these songbirds survived the time
of human domination without significant losses and continued their evolution
in a new epoch. Neocene Meganesia is home to both typical Australasian wrens
– small insectivorous birds – and the endemic family of running malurs.
Running malurs descend from one of Australasian wren species, which has completely
passed to a ground lifestyle. This turn of evolution is natural: already in
Holocene, most of the Australasian wrens flew rather reluctantly, preferring
to run. The running malurs, represented in Meganesian fauna by several species,
are much larger than their ancestors; they have long strong legs and relatively
short wings. However, none of the species of running malur has lost the ability
to fly. The tail at all species of these birds is very long; they are characterized
by the habit of lifting it vertically and shaking it.
Big-legged chicken wren is a typical representative of the family. This species
has a wide range: savannas and semi-deserts of Meganesia. This bird externally
resembles American roadrunners (Geococcyx), but is somewhat smaller than they
are. Body length reaches 50 cm, with half falling on the tail. Big-legged chicken
wrens are characterized by pronounced sexual dimorphism. The female is rather
modestly colored: the underside of the body is straw yellow; back, wings, tail
and head are light brown with numerous dark mottling. The male is remarkable
in bright coloring of the head and tail: the tail is dark blue with a metallic
sheen; the cheeks and the top of the head have a shining bright blue color.
Throat is white, nape is black and a black stripe runs across the eye. Beak
and legs are black in the male and yellow in the female. Male and female do
not differ in size.
These birds are quite silent; their voice resembles a quiet squeak. The mating
song of the male is a soft trill.
Big-legged chicken wrens feed on large invertebrates and small vertebrates –
lizards, small snakes, rodents. Only sometimes they eat berries. These birds
are able to chase prey, running for a long time and reaching speeds of up to
25 km/h. They usually kill prey with their feet.
Big-legged chicken wrens are territorial birds. A family couple, preserved for
life, occupies a territory of 40 to 70 hectares and protects it from competitors.
In addition to them, their juveniles from previous broods live on the territory
of the parents, helping in the rearing of offspring. After the death of one
or both parents, their place is taken by helpers with the highest social status.
As a result of the practice of inheritance of nesting territories, inbreeding
is not uncommon among these birds. The settling of young birds to other territories
takes place only to nearby territories.
Reproduction of big-legged chicken wrens occurs after the rainy season. Parents
build a nest of twigs and grass and place it in a bush. There are from three
to four eggs in the clutch, the female incubates the clutch for 20-22 days.
After the nestlings hatch, both parents and helpers are engaged in feeding them.
After a month, they become completely independent. A small number of juveniles
leaves the territory of the parents.
The enemies of big-legged chicken wrens are birds of prey, predatory marsupials,
as well as monitor lizards and snakes. In case of danger, these birds try to
escape or hide in the tall grass. They take off only in case of extreme danger.
The life span of big-legged chicken wrens reaches 18-22 years. Such a long life
expectancy is explained by the social structure: adult birds, due to their helpers,
spend less effort for feeding offspring.
In jarra – eucalyptus woodland in the south of Meganesia – a close species lives
– running
magpie wren (Dromomalurus cyanoleucus). Birds of this species are slightly
smaller than big-legged chicken wrens and differ greatly in color. Females have
a dark gray top and light bottom. Males have a black head, dark blue wings,
back and tail. The underside of the body is white, the throat is also colored
white, a black belt runs across the chest. The social behavior of running magpie
wrens is similar to that of the previous species, but the size of the territories
is much smaller: no more than 20 hectares.
This bird species was discovered by Simon, the forum member.
Caped
Magpie (Neogymnorhina atrox)
Order: Passerine Birds (Passeriformes)
Family: Butcherbirds (Artamidae)
Habitat: Widespread in well vegetated areas throughout Meganesia.
One iconic bird of the Australian suburbs was the Australian Magpie (Gymnorhina),
this species remained successful in the face of human habitation, and often
became a favoured resident of parks and gardens. It has accordingly left a descendant
in the Neocene, Caped Magpie.
This bird is slightly larger than its ancestor, but resembles it in shape, up
to 47 centimetres long and resembling a crow or raven in general, wingspan is
up to 1.2 meters, and weight is up to 460 grams. Coloring is mostly glossy black,
with a large white area from the nape down the back and onto the front of the
wings, face is contrasted in pale grey, wing feathers are black with white tips,
underside is grey. The bill is conical and wedge shaped, and steel grey in colour,
iris of the eye is yellow, feet are dark grey. Juveniles are lighter and generally
greyish in colour. This bird is an accomplished singer and produces a variety
of warbling and melodious gurgling calls, as well as keening caws, juveniles
make a keening begging call.
This bird is mostly diurnal, and calls a lot in the early morning. Diet consists
mostly of various invertebrates found on the ground like worms, grubs and insects,
and it will dig with its bill in order to find them, small vertebrates, carrion,
roots and nuts are also eaten. Caped Magpie is a very intelligent bird, even
smarter than its ancestor, similar in cognitive abilities to a raven or crow.
Birds are territorial and may maintain a single territory for many years; each
territory is occupied by a single flock that may contain as many as 20 birds,
which range over a large area. These birds actively repel intruders upon their
territory, such as predatory birds, snakes and marsupials, they will attack
by swooping and striking blows with their sharp beaks, as well as producing
loud calls and generally antagonizing the attacker. Birds call as a group to
warn off other groups of the same species, and confrontations between groups
involve loud calling, posturing and aerial displays.
Breeding season varies depending on location, northern populations breed from
June to September, but in the south-east occur from August to September. Two
to five light green mid-sized eggs are laid in a large cup shaped nest woven
of twigs, reeds and vines. The incubation lasts 22 days and all chicks hatch
at once, chicks are altricial, pink and defenceless, being fed and attended
by both parents in turn, eyes open after 10 days and feathers begin to grow
within the first week. Juveniles take 4 weeks to fledge and start foraging away
from the nest after 3 weeks, though they will frequently beg their parents for
food, they forage on their own more after 6 months. Young reach adult size within
a year and are ejected from the group. Lifespan is up to 25 years, and predators
include predatory marsupials, snakes and raptorial birds, chicks may be killed
by other carnivorous birds.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
River
frogbutcher (Dolicholanius batrachovorus)
Order: Passerine birds (Passeriformes)
Family: Shrikes (Laniidae)
Habitat: Far East, tropics and subtropics, swamps, lakes and river basins.
In the human epoch, the number and diversity of birds of prey of the Falconiformes
order decreased significantly, and after the ice age at the boundary of Holocene
and Neocene, other species of carnivorous birds gradually began to occupy the
ecological niches that previously belonged to them. Among the small species
of feathered predators, a special place belongs to the representatives of shrike
family. Among these species, relatively large species have evolved, specializing
in various kinds of small prey.
In the wetland areas of China, in river basins and along the shores of lakes,
one of the largest species of shrikes, the river frogbutcher, lives. This species
is a relative of the fishing
warbler (Dolicholanius piscatorius), which lives in the same area, but differs
markedly from it in its preferred food. It is also smaller than its fish-eating
congener: the body length is about 40 cm; the wingspan is about 65-70 cm. The
upper side of the body is brown, turning into ochre-red on the head. The skin
area from the base of the beak to the eye is featherless, covered with yellow
skin. The forehead is black, a narrow belt of black feathers stretches from
the forehead behind the eyes to the throat. The beak is also black. The underside
of the body is light: throat and upper chest are yellowish, belly is white.
The female has a yellowish color replaced by white; she is more massive than
the male. The primary feathers are black, the secondary ones are black with
white tips; tail feathers have the same color. Tail is long and fan-shaped;
the wings are wide and rounded. The bird has a fast maneuverable flight, is
able to turn sharply in the air and even turn over backwards, grabbing its prey.
Like the fishing warbler, this species has long legs, hooked claws and numerous
corneous bristles on the underside of the toetips.
The bird has very large ear slits and acute hearing. The ear slits are covered
with elongated feathers forming a pair of short “horns” on the head. This adaptation
is associated with the peculiarities of the bird’s diet: the main part of the
prey consists of amphibians – toads and frogs, including poisonous species that
other animals do not feed on. For butchering the caught prey, the bird uses
a long laterally compressed beak with a hooked tip. There is also a serrature
at the tip of the mandible.
This species lives mainly in moist evergreen forests in river valleys and along
the shores of reservoirs overgrown with large-leaved marsh grasses. In addition
to amphibians, this species also eats small fish, less often young turtles.
The bird finds fish and turtles with the help of sight, and hunts amphibians
also with the help of hearing. The bird attacks the prey from an ambush, quickly
flying over its shelter and grabbing it with its paw. Claws and corneous spikes
help to hold slippery prey. This bird eats the caught animals on a branch with
thorns or a broken sharp twig, on which the prey is impaled. When eating poisonous
amphibians, this bird is careful: it pecks out the insides and muscles through
the belly, leaving intact the skin on the back, where the most poisonous glands
are.
River frogbutchers live in pairs on a vast territory, claiming rights to it
with characteristic loud trills. Male and female can hunt in different places
of the territory, but they spend the night together and breed 2-3 times a year.
The nest has a shape of a basket of grass and is woven among the branches, at
a height of 3-4 meters above the water, in places inaccessible to terrestrial
mammals. During the year, birds nest in different places of the territory. Courtship
games are accompanied by high-speed flights, during which birds tear off leaves
on the fly and grab them in the fall. The male finds a suitable place for the
nest and sings intensely, making sonorous trills. He begins the building of
the nest, but does not finish it, and protects the territory from relatives
and competitors. These birds are aggressive towards kingfishers, attack and
can kill them.
In the clutch there are 2-4 eggs, white with some small brown specks on the
shell. The female usually incubates them. Incubation lasts 20 days, offspring
is feeding up to 45 days. Juvenile plumage is distinguished by the white color
of the throat and chest, as in the female, and by the absence of a black stripe
bordering the face. Sexual maturity occurs at the age of 13-14 months, life
expectancy is up to 15 years.
Skew-billed
shrike (Dromolanius loxiirostris)
Order: Passerine birds (Passeriformes)
Family: Shrikes (Laniidae)
Habitat: Far East, tropics and subtropics, wetlands, lakes and river basins.
The tropics of East and Southeast Asia in Neocene became a place of significant
adaptive radiation of songbirds of the shrike family – a significant number
of large species of this group occupying ecological niches of birds of prey
are found here. Swampy forests in the tropics and subtropics of the Far East
are home to one of the specialized species of this genus – a skew-billed shrike.
It settles near reservoirs and in swampy forests, and occupies a specific ecological
niche of the consumer of gastropods.
Skew-billed shrike is a bird about 35-40 cm long (the female is larger); about
half of the body length is a fan-shaped tail. The bird has a large head; the
skin on the throat and on the face from the beak to the eyes lacks plumage and
has a grayish color. The plumage on the crown, nape and shoulders is gray; back,
wings and tail are colored ochre-brown. There are white spots on the tips of
the tail feathers that distract the predator’s attention when attacking this
bird. A characteristic feature of the behavior of the skew-billed shrike is
frequent shaking of the tail. The underside of the body is white in males, beige
in females.
Legs are relatively long and have no corneous bristles on the underside of the
toes. The claws are hooked, of moderate length, allow the bird to climb the
branches and stems of reeds. The bird flies well, but spends a lot of time on
the ground, moving with quick jumps in search of land snails.
The beak of this species is black and has a special shape, having determined
the name of the bird: the tip of the upper beak is slightly slanted to the left,
which allows the bird to extract the snail’s body from the right-hand shell.
In the shape of its beak, this bird is a bit like a crossbill (Loxia). The lower
jaw is shortened and massive, allowing holding the shell firmly. At the tip
of the movable tongue, several corneous bristles develop, helping the bird to
hook the slippery body of the mollusk.
Skew-billed shrike feeds on land and freshwater snails. The bird gathers them
on the ground and in shallow pools, as well as on the surface of the water in
thickets of marsh plants. Birds of this species often perch on the leaves of
large water lilies and examine the underside of the leaves in search of snails.
After getting a snail, the bird takes it to a tree branch, where it eats the
prey. Unlike species of shrikes that feed on vertebrates and insects, the skew-billed
shrike does not impale snails caught on thorns, but squeezes them into a narrow
fork of branches and extracts meat from the shell with its beak and tongue,
holding the shell with its paw. If necessary, the bird cracks the last turn
of the shell with the help of the lower jaw. Empty snail shells accumulate under
the feeding sites of the skew-billed shrike.
Due to the specifics of the diet, this bird species is very territorial and
aggressive towards its relatives outside the breeding season. The female and
male do not form permanent pairs and stay in their own territories outside of
nesting time, protecting their borders even from each other. Birds constantly
claim the rights to the territory with loud trills that end with a long whistle.
If an opponent appears, the owner of the territory pursues it, attacking from
above and clinging to its plumage with claws.
Birds nest 2 times a year. At this time, the male lingers for a long time on
the borders of the territories of the females and sings: at this time, his song
lacks the final whistle element, characteristic for the aggression signal. Usually
in the first days the female drives him away, but gradually the birds begin
to treat each other more loyally, and the pair begins to build a nest together.
It is located in dense thickets of swamp plants or high on branches and has
a shape of a bowl tightly woven of grass. In the clutch there are 3-5 eggs with
a greenish shell; the female incubates for 16 days. The nestlings are fed on
snails for 4 weeks, after which they leave the nest and the brood is divided:
part of the offspring is fed by the female; the other part is fed by the male.
The juvenile plumage differs from the adult one in the brown color of the feathers
on the head and shoulders. Sexual maturity occurs at the age of 9 months, life
expectancy is no more than 6 years. Due to its specific diet, this bird species
is highly infected with parasitic worms and represents an important intermediate
and final host in the life cycles of parasites.
Crayfish-eating
shrike (Dromolanius cancriphagus)
Order: Passerine birds (Passeriformes)
Family: Shrikes (Laniidae)
Habitat: the tropics of Southeast Asia, Jakarta Coast, riverbanks and lake shores.

Picture by Pavel Volkov
Shrikes represent one of the characteristic groups of small
carnivorous birds of the tropics. In the age of man, their diversity was limited
due to the widespread distribution of falcon birds and allies occupying ecological
niches of larger feathered predators. In the human era, the number of typical
birds of prey declined sharply, and many of their species became extinct at
the end of Holocene or in the epoch of the global ecological crisis at the boundary
of Holocene and Neocene. This circumstance allowed the shrikes to get rid of
competition. In early Neocene, the family experienced a period of active adaptive
radiation, mastering various new types of feed and passing into a large-sized
class. Among the shrikes of Neocene epoch there are several large species, as
well as a number of very specialized forms that consume a narrow range of kinds
of food. An example of a species of such kind is the crayfish-eating shrike
that inhabits tropical forests in swampy areas of Southeast Asia.
This bird species has a bright color: the male has a scarlet face, throat and
upper chest, a wide white cross stripe on the belly, and a rusty-red undertail.
The beak is black; a black stripe stretches from the beak across the eye, narrowing
behind the ear. Against the background of the black stripe, the greenish-white
iris of the eye stands in stark contrast. Crown, nape and shoulders of the bird
are green with brown longitudinal strokes on the feathers. The wings, back and
tail are brown; the tips of the tail feathers are white. The female differs
from the male in a duller color: the scarlet color is replaced by an ochre-red.
These birds have short rounded wings and a long narrow tail. The flight of the
crayfish-eating shrike is fast and straightforward.
This bird spends a lot of time on land, moving by leaps in the undergrowth and
among swamp vegetation, is often found along the shores of shallow oxbow lakes
and ponds. On the ground, the movements of the crayfish-eating shrike are fast
and “nervous”: the bird constantly shakes its tail and utters a short double
whistle – a call note for the mate. The long tail serves as a balancer and helps
during the turns. When attacked by a predator, the bird takes off with a loud
alarm cry.
The crayfish-eating shrike feeds mainly on crustaceans – crayfish, shrimp and
crabs. If necessary, the bird enters shallow water and plunges its head into
the water to catch its prey. The bird has a very thick and strong beak with
a small curved tip and two short serrations on the mandible, which helps to
crack the shells of crabs – the bird tries to kill a crayfish or crab as soon
as the bird grabs it to avoid pinching their claws. The thin tip of the beak
allows the bird to scrape meat from the shells. Like its snail-eating relative,
the crayfish-eating shrike does not pin prey on a thorn, but simply clamps in
the fork of the branches and pecks.
This species is a strict monogamist; pairs are formed for several nesting cycles
in a row. Birds make 2-3 clutches during the year. The nest shaped as a deep
bowl is made of grass and fibers, and is woven among the thin branches of a
tree, usually above water. In a clutch there are up to 4 eggs with a greenish-blue
shell. Juvenile coloration is similar to that of the female, but young birds
lack a black stripe stretching across the eye. Sexual maturity occurs at the
age of one year, life expectancy reaches 6-7 years.
A close species, mangrove
shrike (Dromolanius vorax), lives in the mangroves of East and Southeast
Asia, widespread on the coasts of many islands of Indonesia. It is distinguished
by wide black markings on the cheeks and around the eyes – in fact, the head
is black from the sides. The plumage from the forehead to the back of the head
and on the back of the neck has a bright green color without a brown pattern,
and the area of bright plumage covers only the throat and upper chest. On varioous
islands there are different color morphs of this species – with scarlet, orange
or fawn feathers on the throat and chest, and a white band on the belly may
have different widths and in some cases is missing. This species lives in mangrove
forests near the channels and feeds on crabs. In search of prey, the bird usually
jumps along the roots of trees, going down to the water, less often tracks crabs
from the lower branches. This bird species actively claims the rights to the
territory, uttering long trills consisting of two-syllable signals. When meeting
with a trespasser of the territory, the birds fluff their feathers on the throat
and chest, arranging a kind of ritual duels, accompanied by loud crackling calls.
These bird species are the final hosts of a number of parasite species transmitted
through crab meat.
Nocturnal
white-eared shrike (Noctliocton vespertiliovorum)
Order: Passerine birds (Passeriformes)
Family: Shrikes (Laniidae)
Habitat: Zinj Land, forests and mountainous areas.

Picture by Pavel Volkov
In Neocene, the tropical regions of the Earth became the place
of appearance of a new group of feathered predators. As a result of the reduction
in the number and species diversity of falconid and accipitrid birds, representatives
of the shrike family got a chance to occupy their ecological niche and move
into their size class. In the tropical regions of the Old World, a significant
number of species of large shrikes hunting various prey appeared. In the course
of evolution, various specialized forms appeared among these birds, avoiding
competition due to adaptation to feeding on special types of prey. One of these
shrike species lives in the mountainous areas of Zinj Land.
Nocturnal white-eared shrike is a specialized aerial predator that preys on
bats. Unlike contrast-colored relatives that hunt birds, rodents or insects,
this shrike species has a very dull color. The plumage on the chest and bely
has a dark gray color; on the upper part of the body and wings it is black with
a faint greenish reflection, noticeable in daylight. To recognize relatives,
behind the bird’s eyes there are two rounded spots of pure white color. In these
places, the bird’s plumage reflects ultraviolet rays, so these spots are clearly
visible to its relatives, but invisible to bats, its main prey. Birds of both
sexes have a pair of large white spots on their sides. When a bird is perching
on a branch, they are hidden under the wings, but in flight they are clearly
visible. Another feature associated with the specialization in hunting bats
is a very soft and fluffy plumage that suppresses their echolocation signals.
This bird species has a fast maneuverable flight. It has wide wings with rounded
tips and a fan-shaped tail, thanks to which the bird can make sharp turns in
the air, chasing its prey. The paws are short, with strong toes and curved claws.
The edges of the primary feathers are equipped with barbs like those of owls
– they muffle the sound and make the flight of the bird completely silent. The
body length of this bird is about 30 cm; the wingspan is up to 40 cm.
Large eyes reflecting light and burning bright red in the dark are a very characteristic
feature of the appearance of this bird. The beak is black, compressed laterally,
with a curved cutting tip. The nocturnal white-eared shrike hunts mainly bats,
less often birds and large moths. There are two peaks of activity in the behavior
of this bird – morning and evening ones. At this time, the birds gather near
the places where bats spend the night and watch for their flocks at the entrances
to bat shelters. Usually, the nocturnal white-eared shrike attacks prey in flight,
trying to grab the wing with its claws of one paw and inflict deep wounds on
the prey with its beak and other paw. Less often, a bird flies into a cave and
grabs bats hanging from its vaults. Also, the bird can examine tree-trunk hollows
in search of prey. At night, these shrikes attack sleeping birds ranging in
size from sparrow to pigeon. This bird grabs large moths and beetles with its
paw or beak in flight. The prey is impaled on thorny branches, and the bird
eats it, as ordinary shrikes do.
This species lives exclusively in forests, does not descend to the ground and
spends its entire life on rocky cliffs or in the tree crowns.
Seasonality in nesting is not pronounced; there are usually 2 clutches during
the year. A pair is formed for one breeding cycle. The mating song of the male
sounds like monotonous melodious trills that are usually heard in the dead of
night. This species nests in dense tree crowns, at a height of more than 10
meters above the ground. The cup-shaped nest is woven of plant fibers and leaves
of epiphytic ferns and is tightly intertwined with branches or lianas. A loose
woven canopy made of plant fibers is built over the nest of this bird, protecting
it from the bright sun. In the clutch, there are 2-4 eggs with a gray shell
covered with small black smears. Incubation lasts up to 15 days, the young are
fed for 5 weeks in the nest, and up to 2 weeks after leaving the nest. In young
birds, the base of the beak is surrounded by a narrow ring of white feathers
– this is an stimulus for parents to feed the juveniles. When young birds leave
the nest, these feathers shed very soon, and parents begin to show aggression
towards young birds instead of feeding. Sexual maturity occurs at the age of
9-10 months, life expectancy is no more than 6 years.
Polar
shrike (Boreolanius versatilis)
Order: Passerine birds (Passeriformes)
Family: Shrikes (Laniidae)
Habitat: northern regions of Eurasia and North America, Greenland.

Picture by Pavel Volkov
During ecological crises, carnivorous species standing at
the top of the food pyramid suffer the most damage – their well-being depends
on many factors, and their number is initially lower than that of herbivores.
Therefore, carnivorous species are characterized by a high rate of both speciation
and extinction. The decline in the number of birds of prey in the human era
allowed carnivorous bird species from other groups to partially occupy their
ecological niche. In Neocene, one group of shrikes in the tropical regions of
the Old World turned into peculiar analogues of falcons
and ospreys. But along
with such specialized species, typical shrikes – predators of small sizes –
remained in Neocene fauna. One of these species inhabits the subarctic regions
of the Northern Hemisphere, occurring in the tundra and krummholz zones. It
is a polar shrike – a specialized hunter of small vertebrates.
This species of songbirds is equal to a large finch in size – the body length
is up to 15 cm, including a wide fan-shaped tail. A characteristic feature of
this bird is the ability to change the color of its plumage for the winter,
which makes its hunting more successful. The summer plumage combines the ochre-red
coloration of the back, wings and tail, sandy-yellow belly, black top and white
sides of the head. The secondary feathers are black, but their coloration is
visible only in flight. The winter plumage is snow-white and dense; it allows
the bird surviving easily in winter frosts. The legs are feathered to the toes
– it is a characteristic feature of the genus. In summer, the feathers on the
legs are short, in winter they form long “pants”. The bird has long curved claws.
Beak is thick at the base, with a thin hooked tip.
Polar shrike keeps near thickets of bushes. Here it builds a nest and impales
prey on broken branches. The bird usually keeps on top of the bush and twitches
its tail up and down – this is a behavioral feature common to shrikes.
This species is capable of hunting rodents and small birds. From spring to autumn,
birds and large insects form the basis of the species’ nutrition, in winter
it almost completely switches to feeding on murine rodents. Polar shrike is
capable of killing prey twice its weight. In winter, the bird often tracks down
rodents in the snow.
Birds start egg laying in the middle of spring, after the snow melting and the
spring molt. Pairs are formed for one breeding season. The courtship ritual
involves aggressive pursuit of the female by the male and keeping her in the
nesting territory. After ritualized feeding, the pair usually stays together
and the birds feed each other on pieces of their prey. The bowl-shaped nest
is woven in the depths of a shrub; there are 2-3 eggs in a clutch. The female
incubates the clutch, but the male keeps on the territory at this time. A pair
of birds aggressively protects the nest and is able to attack even fox- or owl-sized
predators.
Young birds leave the nest by the end of summer and lead an independent life
in autumn. Sexual maturity occurs at the end of winter, and young birds are
able to give offspring next spring. Life expectancy in nature does not exceed
5 years.
Singing
Hookbill (Psittacolanius cohleaphagus)
Order: Passerine Birds (Passeriformes)
Family: Shrikes (Laniidae)
Habitat: Forested swamps and mangrove forest adjacent to Arafura and Carpentary
lakes in Meganesia.
Australia is notable for its richness in endemic bird families, but during the
Neocene and even before, new kinds have arrived via southeast Asia and the Indonesian
Archipelago. Shrikes (Laniidae) are among them now, not to be confused with
the native birds that are called the similar name of “butcherbird” (Cracticus).
Various species in the Laniidae exist in Northern Meganesia, and some have become
specialized, such as the Singing Hookbill. This bird is about the size of a
small crow, reaching 250 grams, and is notable for its deep, parrot-like bill,
of which the upper portion has a long slender hook. This bill betrays the diet
of the bird, it feeds almost entirely on various swamp and mangrove-dwelling
snails, and will also eat rainforest snails. Its technique varies by species
and size, some kinds are thin-shelled enough to simply be broken open with its
powerful bill, but various saltwater sorts have thicker shells and even opercula,
for these, the bird inserts the hook of its bill and dexterously severs the
muscle which retains the shellfish inside its shell. This bird will also occasionally
feed on land crabs or young crayfish, being able to break them open.
Coloration is buff brown on the upper side with a white belly, black wings and
a long black and white tail array. Wings are broader than some of the more active
types of shrike, allowing it to manoeuvre in the undergrowth and tangle of mangrove
roots. Bill is dark black and a single dark stripe goes through each eye. The
female is a duller brown with creamy colored underside. This bird is mostly
diurnal and structures its life around low tide, when various snails are exposed
on low vegetation and mud flats.
As indicated by its name, its calls are various melodious whistles and warbles,
as well as a more typical “shrike” noise when irritated. These birds are monogamous,
and will often pair for life, breeding occurs in the dry season from April to
September, when tides are less aggressive. The male will make overtures to the
female by singing, and will also prepare gifts of freshly caught snails which
he usually places in the crook of a tree. The pair will usually maintain a territory
together that they defend vigorously. Their nest is made up of a cup woven from
thin twigs and cemented with mud. Nestlings leave the nest at 3 months and are
independent by 5 months. Lifespan is up to 12 years.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Guyrauna
(Corvus notus)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: tropical and temperate areas of South America, forests of various kinds.

Picture by Pavel Volkov
The end of the Holocene was marked by several climatic changes,
one of them was an Ice Age. During this period, some species moved to regions
with a warm climate, among them was the American crow (Corvus brachyrhynchos).
Due to the cooler temperatures of North America, individuals of this species
moved down, crossed the Panama Isthmus before it was broken and reached South
America. There, they continued to evolve and in the Neocene they gave rise to
a new species, the guyrauna. Its common name is formed by two Tupi-guarani words:
Guyra (bird) and una (black). This species is widespread across tropical and
temperate South America, living from the Amazon rainforest to the Araucaria
forests.
Guyraunas are large birds, with 53 cm of length and a wingspan of 102 cm. Like
their ancestors, these birds have their bodies covered by black iridescent feathers.
However, they developed white patches in their wings. Both their beak and feet
is black.
These birds are omnivorous, with their diet changing according to the region
where they live. They will feed on invertebrates of all types, fruits, carrion,
Araucaria pine nuts, seeds, eggs, nestlings and stranded fish on the shore.
Guyraunas are active hunters and will prey on rodents, lizards, frogs, and other
small animals. They are among the first birds to arrive in a carcass, feeding
fast to avoid larger scavengers, such as vultures and birds of prey. Being clever
birds like their ancestors, these animals are capable of modifying and using
tools to obtain food.
Guyraunas are socially monogamous birds, breeding in a cooperative way. The
mated pairs form large families of up to 15 individuals from several breeding
seasons. The offspring from a previous nesting season will usually remain with
the family to assist in rearing new nestlings. These birds do not reach breeding
age for at least three years, which is the time when they leave the family.
They build bulky stick nests, nearly always in trees but sometimes also in large
bushes and, very rarely, on the ground. Three to six eggs are laid and incubated
for 18 days. The young are usually fledged by about 36 days after hatching.
Their lifespan is 23 years, but many of them are killed by predators before
reaching this age.
Their closest relative is the Mexican
crow (Corvus mexicanus), another descendant of the American crow. Living
from the Mexican Plateau to the tropical forests near the Panama Strait, this
species has the same plumage of their relatives in South America, but is smaller
with 47 cm of length and a wingspan of 95 cm.
This bird species was discovered by João Vitor Coutinho, Brazil.
Stripe-headed
daw (Callomonedula fasciatocephala)
Order: Passerine birds (Passeriformes)
Family: Corvine bird (Corvidae)
Habitat: Northern Africa, savannas and light forests.

Picture by Lambert
In Holocene the fauna of Earth had suffered from human activity.
It had especially touched species not suffering anxieties from the part of people.
But in same time synanthropic species could expand an area and occupy new habitats.
Among them some representatives of corvine bird family had especially succeeded.
These birds due to intelligence and abilities to get on with people had received
excellent chance of survival in Neocene. Having kept high number, representatives
of corvine birds had advantage after human extinction before other species,
and could occupy various ecological niches. Among them large predators
and scavengers had appeared,
and some species of this family had became analogues of other medium-sized warblers,
representatives of other families of the order.
In savannas of Northern Africa speckle-headed daws are – rather small-sized
birds making the separate genus – are characteristic representatives of corvine
birds. They are descendants of common daw (Corvus monedula), one of usual synanthropic
species in Holocene. The majority of species of these birds is starling-sized,
or a little bit larger. The characteristic feature speckle-headed daws – the
presence of well-distinguished and appreciable from afar pattern on back part
of head. It serves for demonstration at conflicts between birds, and also for
recognition of congeners. Wishing to show this pattern, bird rears feathers
on head, slightly having bent it forward. At different species of speckle-headed
daws pattern differs, that precisely permits them to find out congeners, not
reacting to other species. Crown, forehead and throat at birds of all species
of this genus are dark, and feathering on body has more varying colouring, than
at ancestor, due to metal shine. At all species characteristic feature of daws
is kept: light grey iris of eyes distinguishing them from other corvine birds.
Being in the majority small birds, speckle-headed daws eat insects. They keep
on backs of large herbivores of savanna – flathorns,
harelopes, giraffe
ostriches. Birds gather parasitic and blood-sucking insects from them therefore
both large herbivore and bird receive mutual benefit. Other service, which they
render to large animal, is the work of “sentinels”: due to keen sight birds
notice predators beforehand, and warn herd by loud cries.
Stripe-headed daw is the typical representative of genus. This bird is colored
lighter, than its far ancestor known to people. Feathers on its nape are hatched
in narrow cross strips, forming regular black-and-white pattern. These feathers
are lengthened and look like a wig. Beak is light grey. Feathers on stomach
and back of bird are also light grey, almost white. Wings and tail are black
with bright blue metal shine. Due to the neighbourhood with large animals stripe-headed
daw feels like protected from an attack of various feathered predators even
having such bright colouring.
Outside of nesting season stripe-headed daws migrate in savanna with herds of
herbivores, perching for a night on trees or bushes near to herd. Animals like
flathorn represent convenient “dining room” for them: on each animal some birds
constantly are “on duty”, gathering from the animal ticks and horseflies. These
birds keep on back, shoulders and groats of animals, gathering from them mainly
large flies. Sometimes they fly out from animal, and quickly catch insect in
flight.
Once a year, in the beginning of rain season, stripe-headed daws partly lose
interest to herds: the time of nesting comes. At this time they gather to large
colonies on big hollow trees, and arrange noisy courtship demonstrations. Males
start displaying: they hang on branches headfirst near the chosen tree-trunk
hollow, and start to shake, loudly crying. When any female shows interest to
nest and its owner, male arranges noisy race, pursuing her in air. So birds
estimate forces of each other. If birds have had a good first impression about
each other, they start to get acquainted closer: cautiously touch each other
by beaks and start to clean feathers to each other - in the beginning on wings
and back, and then when the trust will be established – near eyes of the partner.
At last the female shows the even greater trust to the male, allowing him to
feed her. For this purpose she squats and stretches wings, imitating a nestling,
and male feeds her from beak with insects. The generated pair keeps very amicably,
in common protecting nest from encroachments of competitors.
When eggs (4 – 5 white eggs) appear in nests, parents hatch them alternately.
Free from a hatch bird feeds and gathers food for the partner.
Nesting in colony has one essential advantage: safety of clutches and hatches
of stripe-headed daws is very high. Potentially dangerous enemy will be driven
off at once with the whole colony: birds snatch on aggressor, cry, beat him
by wings and, peck and scratch by claws. Force of joint attack of birds happens
so great, that even the deadlynetta or giraffe ostrich recedes from flock of
these birds.
Nestlings hatch after 17 – 18 days of hatching. They are naked and blind, but
develop quickly – at the second week of life they fledge, and to the end of
third week of life leave nest and perch on branches of tree. About two weeks
parents feed them, and then young birds start to feed independently near to
adults. The young growth becomes able to nesting from the eighth – the ninth
month of life, but usually young birds nests at one-year-old age, when new rain
season begins.
Close species of speckle-headed daws live in savannas of Northern Africa:
Silver-headed
daw (Callomonedula argenteocephala) is one of small species of family.
It is remarkable in color of feathers on nape: they are silvery colored with
strong metal shine. Showing them, bird trembles the head, and sun light sparkles
on feathers. That’s why the bird looks very effectively, especially in courtship
season. In other features the biology of this species is typical for genus.
Silver-headed daw feeds on rather small herbivores: harelopes and kangoohoppers.
In nesting biology of this species there are essential differences from congeners:
this bird does not form large colonies, nests in dense prickly bush and builds
dense spherical nests of branches.
Snow-headed
daw (Callomonedula grandis) is the largest species of genus: it is
little bit larger than common daw by size. Nape, sides of head and cheeks at
this bird are snow-white, beak is ivory-colored; on the head there is small
black “cap”, and there is dark site on a throat like narrow “tie”. During the
courtship demonstration bird fluffs feathers on head such way that black sites
are brightly visible on white background. The feathering on body sharply contrasts
with head in colouring: whole body of bird is coal-black, but on wings and tail
there is strong bluish-green metal shine. Paws of snow-headed daw have very
remarkable color: they are coral-red at birds of both genders.
This bird does not render services of the cleaner to large animals, but constantly
keeps near to them. The bird benefits by presence of large animals: moving,
they scare away small animals for which this bird hunts – rodents, lizards and
large insects. Usually such bird perches on bushes and trees near to grazing
herbivores, and catches frightened away animals by fast throws. And when it
joins the herd of flathorns, this bird at all perches on their wide corneous
head incrustations, and attacks catch therefrom. Also these birds may attack
miteeaters and bloodbirds
– small warblers feeding on large animals.
Silver-headed daw nests in small colonies numbering up to 3 – 5 pairs of adult
birds. It is much more aggressive than other representatives of genus, and attacks
even other species of speckle-headed daws, driving them away from nests.
Nice-headed
daw (Callomonedula callocephala) is starling-sized bird. It differs
in brightest feathering among congeners. All body of this bird is grayish-white,
tail is black with greenish metal shine. Occasionally birds of this species
have black primary feathers in wings - at various birds in varying amount, sometimes
wings of any bird are colored differently. Head of this species is very bright:
black “cap” is bordered behind and from sides by blue feathers with strong metal
shine. Blue “mane” gradually passes to glaucescent colouring of shoulders with
crimson-pink shine. Beak of this bird is snow-white.
Nice-headed daws keep in big flocks of rather constant structure, numbering
up to 50 birds. Out of nesting season they “serve” herds of various hoofed mammals,
clearing them from parasites. More often they keep on sides and stomach of animals,
gathering from them ticks and small horseflies. Thus, they do not compete to
close species of genus.
Maned
crow (Leontocorvula leonina)
Order: Passerine birds (Passeriformes)
Family: Corvine birds (Corvidae)
Habitat: Northern Africa, Southwest Europe, Apennines, “oases” of Mediterranean
swamps; savannas and light forests.
In Neocene corvine birds realized the advantages received from inhabiting near
to people. Due to private, but very favourable union with people they had an
opportunity to settle widely and to keep high number whereas other animals disappeared
in the world changed by people.
In Neocene the specific structure of many ecosystems had considerably changed:
the part of large species, characteristic for Holocene, had died out, and they
had been replaced by completely other animals. Extinction had affected the majority
of orders and families of world fauna, which representatives differed in large
size. Therefore among new species of herbivores in African savanna instead of
representatives of hornbills other birds, maned crows, representatives of completely
other family and order, wander.
Maned crow is rather large bird: it is up to 50 cm in height; its wingspan is
about one and half meters. By proportions it is a little similar to any hornbill
- the similar habit of life has an effect. At it there are long legs, short
body and large head with big beak. Tail of bird is straight and fan-shaped;
wings are wide and have rounded ends. Maned crow flies well and even can hover,
though it prefers to spend more time on the ground.
Colouring of the most part of body of this bird is rather dim: wings are black
with bluish metal shine; body is covered with brown feathering. On head and
neck long narrow feathers similar to domestic cock’s ones grow. They form some
kind of “mane”, having determined thus the name of bird. “Mane” is colored brightly:
feathers making it are yellowish, and along every feather black strip stretches.
Showing itself to the opponent during intraspecific conflicts, bird fluffs these
feathers, visually exaggerating its own size. At this moment it becomes a little
similar to the cock prepared to fight Beak is black and shining; at adult birds
its tip is white. Around of eyes there is an area of naked wrinkled grayish
skin covered only with separate bristle-looking feathers. At adult birds this
skin is lighter, than at young growth which had recently left nest.
Maned crow had became the original ecological analogue of ground hornbills (genus
Bucorvus) – African representatives of hornbill family. It wanders by the ground
and searches for small vertebrates and various insects. Occasionally this bird
eats carrion: maned crow observes of behavior of predators
and scavenging birds, and
due to powerful beak easily receives the share of meal at their feast. In Mediterranean
“oases” where the most part of year it is difficult to find potable water, this
bird willingly eats juicy fruits of local
plants.
Maned crow lives solitarily or in pairs. The amicable pair, which had successfully
brought up posterity, can be kept till several nesting seasons. This species
nests in big tree-trunk hollows, high from the ground. In “oases” of Mediterranean
swamps these birds can not find trees suitable to nesting, because not every
species of plants can sustain extreme conditions of salt swamps. Therefore maned
crows nest in rock cracks in former Mediterranean. Here nobody threatens them,
because ground predators can not reach “oases”: mammals prefer to not go deep
into disastrous district where there is no potable water. For nesting in such
places maned crows choose clefts where hot midday sun beams will not reach.
In clutch there is up to three large eggs (like big chicken egg by size) with
white shell. They are hatched mainly by female, and male feeds her and protects
nest. The posterity – blind naked nestlings – hatches after fifteen days of
incubating. The young growth develops rather slowly: nestlings completely fledge
only at bi-monthly age, and leave nest at the age of three months.
Maned crows living in Africa and Europe have the normal sizes typical for species.
The settled small form (L. l. minima) lives in “oases” of Mediterranean,
never leaving this place. It is widespread at Apennines, forming in Northern
Africa the transitive hybrid form with the nominative subspecies.
Mangrove
crow (Subcorvus arafurensis)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: Meganesia, Arafura Lake and surrounding mangrove woods.

Picture by Alexander Smyslov
Corvids are considered as one of tops of avian evolution. Possessing
good intelligence and flexible behavior, they easily adapt to changing conditions
of life. During the human epoch some representatives of corvids reached the
great successes in struggle for existence. The destruction of natural communities
in human era resulted in considerable decrease of specific variety of birds.
The appearance of new ecological niches in early Neocene allowed corvids to
expand considerably an area and among them the species adapted for life in new
conditions had evolved. Mangrove crow, the typical representative of family,
the inhabitant of mangrove woods in northern part of Meganesia, became one of
such types.
This bird species differs not so much in body shape from typical crows of human
epoch. Mangrove crow is a bird having wingspan up to 70 cm and weighting up
to 1 kg. The appearance of bird has some traits of adaptation to life in humid
habitats. Cheeks, throat and forward part of neck of this bird lack feathers
and are covered with light gray skin. Beak of mangrove crow is deeper and more
compressed from sides, rather than at the similar species living in the woods.
Such beak is well suitable for opening the shells of bivalves; this bird eats
them especially willingly.
The plumage of mangrove crow is lead-colored which passes into bluish on back
and the top of the neck. “Hat” of almost white color on crown and nape contrasts
with gray plumage on neck. Beak and feet of bird are black.
Mangrove crow is a sociable bird; out of the nesting period these birds gather
in small flocks and in common migrate in mangrove thickets in search of food.
Thus they constantly keep the contact with congeners with the help of abrupt
croak. In the “language” of these birds there are up to 20 various sound signals.
Inside the flock single birds find a partner to themselves, and adult birds
keep in common and do not leave each other. The nesting season in tropics is
not expressed, and pair ready to nesting simply leaves the flock and settles
at the certain territory in forest. This species nests in pairs which are kept
till the whole life. The nest of mangrove crow is opened from top, made of thin
twigs and arranged in places visible worse from air. In clutch there are up
to 4 eggs with spotty grey shell. Mainly the female hatches them (within 22
days), but nestlings are fed up by both parents. Young birds leave a nest at
the age of 2 months and become mature at the age of 1 year.
This bird species is omnivorous, but prefers food of animal origin. Mangrove
crow pecks up the snails, openly settling bivalve mollusks and wood-drilling
«shipworms» in roots of mangrove trees. Also this bird hunts crabs and catches
fishes in littoral pools. Mangrove crow is often feeding together with larger
animals; when Brontochelis turtles living in Arafura Lake uproot mangrove trees,
these birds catch the invertebrates seeking safety in flight or ones attached
to roots. Also mangrove crows often attack other animals fishing or hunting
small animals, and compel them to leave prey by their attacks.
Translated by Alexander Smyslov.
Chatham
false raven (Notocorax chathamensis)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: Chatham Islands, open habitats, shrub thickets, sea coast.

Picture by Alexander Smyslov
The rook (Corvus frugilegus), introduced by people to New
Zealand, acclimatized on the islands successfully and after the disappearance
of mankind in the process of evolution became the ancestor of a new genus of
corvid birds, southern ravens (Notocorax). One
species of this genus abundantly inhabits both large islands of New Zealand
and many islets off their coast. In addition, on the Chatham archipelago there
is another, close species – a Chatham false raven. In the historical era, the
island was inhabited by an endemic large raven species Corvus moriorum, which
became extinct in the early stages of human colonization of the islands.
This bird species differs somewhat in ecology from its New Zealand relative:
it avoids dense forests and is found mainly in coastal areas of islands, in
areas with sparse shrub vegetation or even among grasses. This species is slightly
larger than its New Zealand relative: the weight of an adult bird is up to 3
kg, the wingspan is about 2 meters. The appearance of this bird is similar to
its distant ancestor: it has wings with wide rounded tips and a fan-shaped tail.
The Chatham false raven has elongated legs and beak compared to its New Zealand
relative, and the strip of bare skin around the beak is much larger – it reaches
the posterior edge of the eyes and crown. The beak itself is much thicker and
is well suited for splitting solid food objects – crustaceans and small mollusks,
as well as echinoderms. The plumage of adult birds is black with a noticeable
bluish metallic shine on the wings and the back of the neck; in young individuals
the plumage lacks shine. Short corneous brushes grow on the toes, forming a
kind of fringe that helps to walk on viscous wet sand.
The diet of this bird species is dominated by seafood, although the bird does
not disdain to eat carrion. Usually these birds roam along the sea coast in
small groups, including several breeding pairs, or consisting of immature young
of approximately the same age. At low tide, birds wander around the littoral,
examining puddles in search of small marine animals, or dig out crustaceans
and mollusks from the sand. Also, these birds peck at the corpses of large fish
and seabirds that died during the storm, and ravage the nests of seabirds, eating
part of their eggs and chicks. Kleptoparasitism is also characteristic of this
species: using their force, birds attack penguigulls returning from the sea
with prey, and force them to regurgitate part of the catch. Chatham false raven
is not afraid to fly far into the sea in search of food, and some groups of
birds are found on islands far from the Chatham archipelago. Some individuals
of this species fly far to the south, to the subantarctic islands. They can
nest there, but do not form a permanent stable population. Most often, these
birds, together with the young, fly back to Chatham by winter.
This bird species prefers to nest among rocks, choosing niches more or less
protected from sea winds. Birds build a nest of grass and twigs, heaping them
in a big pile and in the upper part of it make the depression lined with grass
and feathers. In the clutch, there are usually two eggs with a gray shell covered
with numerous brown specks; both parents incubate for 20 days. The nestlings
hatch blind, covered with sparse gray down. They quickly become overgrown with
juvenile down of dark gray color, saving from hypothermia, and parents can leave
them for a long time, looking for food. The juveniles grow rapidly, fledging
at the age of 2 weeks, and leave the nest at the age of 50 days. Chatham false
ravens are social birds that nest close to each other in groups of 5-6 nests.
They notify neighbors of the appearance of enemies and jointly drive them away.
Due to this behavior, low fertility is compensated by the high survival rate
of young – on average, about 60% of hatched nestlings survive until leaving
the nest. Sexual maturity occurs at the age of 3 years, and life expectancy
is up to 50 years.
This bird species was discovered by Simon, the forum member.
New
Zealand False Magpie (Picorvus australis)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: New Zealand, most vegetated habitats.
Introduced birds which acclimatized in New Zealand sometimes came to converge
upon forms found elsewhere, and so we see with the descendants of the European
Rook (Corvus frugilegus). One form converges on familiar European Magpie (Pica
pica) in shape and habits.
New Zealand False Magpie is a mid-sized bird, reaching 50 centimeters long,
more than half of which is its long, upright tail, wingspan is up to 60 centimeters.
It is a rather typical crow-bird in shape, greatly recalling a Holocene Magpie
in form. Its coloration is bluish black on the upper wings, back and tail, primaries
are white edged with black. There is a bright white “vest” over the chest and
shoulders, the head and chest is blue-black becoming an intense dark blue on
the head, bill is mid length and blackish, as are its feet. Birds weigh about
275 grams, with males being larger than females.
These birds are diurnal, becoming first active in early morning. Its call is
a chuckling, repetitive cawing noise that increases in speed. It is an omnivore,
eating invertebrates, small mammals and reptiles, eggs and nestlings of other
birds, as well as seeds, nuts, stems and berries. As with many Corvids, this
bird is extremely intelligent and curious, it will often intently follow other
animals as they forage and feed, interested in potential prey that they disturb.
These birds may hone small twigs or long thorns to prize insect larvae out of
crevices in trees. Individuals are somewhat social and may form loose flocks
close to the breeding season.
Breeding does not always occur in the first year of life, but usually in the
second. They form monogamous pairs that may remain together all year long. Males
will display to their partner by raising and lowering the crest on their head,
and fanning out their tail and wings, while making a crooning noise, this is
followed by boisterous chases and eventually copulation. Nests are made generally
in very tall trees; and consists of a cup of twigs and sticks cemented together
with mud, lined with leaves and feathers. Clutches are laid in August or September,
and number up to 6 eggs, which are pale blue. Eggs are incubated for 20 days
by the female, who is kept fed while brooding by the male. Nestlings open their
eyes a week after hatching, and fledge after a month. Sexual maturity is reached
at one year, and life expectancy may be as much as 20 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Pied
False Jay (Cittacorax novazealandiae)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: New Zealand, most vegetated habitats.
The acclimatization of the European Rook (Corvus frugilegus) in New Zealand
created opportunities for this animal to fill niches left not only by the extinction
of native Corvids during the age of man, but also to converge upon other Corvid
groups found in the New and Old World.
One such creature is the New Zealand Pied False Jay, an analog of the jays of
the Old and New worlds (genera such as Cyanocitta, Garrulus). Resembling a true
jay in outward shape, it is a mid-sized crow-like bird with a long, upright
tail and a short erectile crest of feathers on its head. This bird weighs up
to 100 grams, reaches a wingspan of up to 40 centimeters and a total length
of about 33 centimeters. Coloration is mostly black and white, underbelly and
chest is white, back and neck are metallic black, tail and wing feathers are
chequered with black and white along their length. Its face is round and white,
with a black stripe through the eye, crest is black with prominent blue sheen.
Bill is mid-length and conical, being dark grey in colour, feet are grey.
As with true jays, they are omnivores with a strong preference for grain and
seeds, including the nuts of broadleaf and coniferous trees. The bird may hold
the nut under its foot as it proceeds to crack it open. Other foods include
insects and various small invertebrates. It is diurnal and generally noisy and
aggressive; they will chase and harass larger birds. Call is a high-pitched,
short, repetitive caw; it may also mimic the calls of other birds.
Breeding occurs at the end of winter (August to September), a nest is built
at a fair height in a suitable tree or large bush, consists of a large cup made
from mud, twigs, bark and other plant materials. These birds form monogamous
pair bonds for life, both sexes build the nest and rear the young, but only
the mother broods the eggs, which number 4 to 6 and incubate over about 18 days.
The young fledge by about 20 days after hatching; sexual maturity is reached
at one year, lifespan is up to 24 years.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Flaming
magpie (Pica flammea)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: Far East, tropical forests.
Magpies in Eurasia proved to be a successful group of birds back in the human
epoch. They are less social than some of their relatives like jackdaws and rooks,
ravens and crows, but they also proved to be able to act together and solve
complex problems, having shown their evolutionary success. They managed to survive
both the anthropogenic pressure of Holocene and the climatic changes of early
Neocene, and after the formation of the Beringian land bridge in the North Pacific
Ocean, they managed to expand their range by penetrating into North America.
But all the same, these are birds, more characteristic of the Old, not the New
World.
Flaming magpie is one of the unusual corvid birds. Its size is quite large:
the male reaches an average length of 70 cm with a weight of about 600 grams,
and the female is about 50 cm, weighing up to 500 grams. It resembles other
magpies in proportions, but its legs are longer and stronger – it spends a lot
of time on the ground. The plumage color is black and white, like other magpies,
but with a brighter, fiery orange metallic luster. In males, this shine is more
pronounced than in females.
The flaming magpie is an omnivorous bird. It eats both vegetative food and animal
food, but prefers vegetative food more: fruits and seeds of plants. It flies
well, but it has relatively short wings, so it gets tired pretty quickly, and
prefers to hide from enemies in the thickets on the ground or in the tree crowns.
Unlike other corvids, this is a tropical species inhabiting forests and not
found north of the latitude of Baikal Lake – in summer some birds fly north,
but in autumn they migrate back.
This species is polygamous: one male usually fertilizes 2-4 females that live
on his territory, protects their nests, but does not care about the offspring.
Females hatch and feed the brood on their own; there are usually 4-6 eggs in
a clutch, sometimes less. The nestlings hatch in 3 weeks, grow and develop quickly,
and after 6 weeks they are already independent. During the year, the female
grows up to 2 broods. The male usually does not show aggression to the offspring,
but the females quickly finish feeding the young and expel them from their territory.
In young birds, the white color in the plumage is replaced with light brown,
and the metallic sheen is almost absent. If a young bird does not die in the
first 2-3 years of life, then it has an opportunity to live for more than 20
years.
This bird species was discovered by Bhut, the forum member.
Forest
Azorean magpie (Azopica collector)
Order: Passerine birds (Passeriformes)
Family: Corvine bird (Corvidae)
Habitat: New Azora, tropical forest.

Picture by Simon
During the ice age separate populations of common magpies
(Pica pica), European corvine birds, turned to birds of passage, receding from
the edge of the approaching glacier. They began to migrate over drying up Mediterranean
Sea to Atlas Mountains. Other route of migrant magpies ran to the west – up
to Canary Islands. And separate individuals migrated even farther – to Azores;
there they had formed the settled population. In Neocene the archipelago had
turned to unite big island New Azora.
In human epoch magpies differed in propensity to larceny of small shining objects.
This feature had received continuation in Neocene, turning to the feature of
courtship behaviour of new species – forest Azorean magpie. Males of these birds
are known for the propensity to collect various bright objects for female attraction.
Azorean magpie is a large, approximately crow-sized bird. Females are much smaller,
rather than males – they are equal to Holocene magpie in size. The constitution
of this bird is robust and more resembles crow’s one, rather than magpie’s one.
Its tail has the size usual for corvine birds – it is shorter, than half of
body length; middle feathers are a little bit longer, than edge ones. The sexual
dimorphism of this species is expressed not only in size, but also in colouring.
Males are colored contrast black-and-white tones: tail, head and wings are black,
and sides, belly and primary feathers are white. Beak and paws at males are
bright yellow. In corners of the beak males have original “wattles” – skin outgrowths
of bluish color, at excitation reddening and increasing, swelling of blood.
The similar attribute was known at New Zealand birds of family Callaeidae (New
Zealand wattlebirds). Females are colored more modestly – they are grey with
black and white spots.
This bird lives on the ground – at the island there is less number of predators,
rather than at the continent. It has not lost ability to fly, but flies reluctantly
and hardly: wings of this bird are rather short. Azorean wood magpies are omnivorous:
they eat seeds, fruits fallen from trees, and various small live beings such
as snails, insects and lizards. These birds avoid a society of congeners – each
bird solitarily wanders in forest in searches of food. Azorean forest magpies
are clever and cautious birds: they beforehand try to escape from predators,
but if necessary, can hide in foliage. If it does not help, the bird actively
defends from the enemy by beak or hardly and noisily flies up, warning thus
other forest inhabitants about danger. Voice of this species of birds is drumming
chatter, similar to the voice of common magpie.
Males of forest Azorean magpie are strictly territorial: each of them has the
site in forest, which is strictly protected. Quite often at borders there are
skirmishes between males from the next sites. Thus they cry, fluff feathers
and “fence” by beaks, striking lateral impacts by beak to beak of the opponent.
Females observe borders of sites not so strictly: they freely wander in forest,
coming to the territory of one or another male.
This bird differs in especial courtship display – males of this species differ
in propensity to gathering various objects which they show to females, trying
to draw their attention. The male, like the bowerbird male, searches for various
bright objects: fallen fruits, flowers, egg shells, feathers etc. Bright elytrums
of beetles are especially appreciated to males; because of them birds can break
border of nother's site or arrange fight. The male spreads collected objects
on the platform trodden beforehand in bush in front of the “bower”, resembling
usual magpie nest (covered building with friable roof). The “bower” building
is strongly modified element of building behaviour of the male.
Besides the platform, male also decorates walls of “bower”. The most part of
time in courtship season male is busy with gathering and sorting of “collection”.
Also he furiously protects collection from other males and other small animals
of New Azora involved with shine or color of collected objects. When the female
comes to the territory of the male, he begins courtship displays. Male loudly
cries, fluffs feathering, straightens white primary feathers “dancing” near
the female, and, certainly, invites her in “bower”. Male shows gathered “collection”
to female, catching and dropping various objects. Sometimes he jumps on the
spot, keeping brightest objects of “collection” in beak. If it does not impress
the female, she simply goes out, but if she accepts courtship, the male pairs
with her … and his family duties come to end after it. At this species, as against
to common magpies, male does not take participation in posterity rearing, because
gathering of collection occupies too long time.
After pairing female builds in bushes cup-like nest with friable “roof” anywhere
in territory of the male, and lays in it 5 – 7 eggs, hatching and protecting
them from predators. She feeds hatched nestlings for a long time, and when they
become fledged and leave the nest, leads them in forest and trains to search
for food. Some weeks later the hatch breaks up.
At coasts of New Azora the close species lives: coastal
Azorean magpie (Azopica littoralica). It differs from the wood neighbour
in smaller size (it is daw-sized bird) and colouring. Males are colored greenish
with black beak and paws, and blue primary feathers; “wattles” are white. Females
of this species are grey with darker head. This bird lives in forests near the
coast of island. At sea coast males gather stones, the fish scale, feathers
of sea birds and shells for collections. Females search for food in coastal
woods and bushes. Other features of behaviour of this bird are similar to behaviour
of forest Azorean magpie. Fertility of this bird is less - 2 – 4 eggs. But the
egg laying can repeat twice per year.
These species of birds were discovered by Simon, the forum member.
Iberian
nocturnal magpie (Neopica afroeuropaea)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: South-western Europe, Atlas Mountains.
Like crows, jackdaws and rooks, magpies were among the most adaptable birds
of Holocene epoch; they were able to live with and without humans. It helped
them survive the era of biological crisis during the transition from Holocene
to Neocene, which was characterized by significant climatic changes. Magpies
have survived and gave rise to new species that often resemble their ancestors
in appearance. Significant changes most often occurred at the behavioral level.
One of these Neocene species is the Iberian nocturnal magpie, a descendant of
the common magpie (Pica pica) of Holocene epoch. Outwardly and in size, it resembles
its ancestor, only its color is darker – not contrasting black and white, but
gray-black with ripples on the plumage; the tail is long. A characteristic feature
of the appearance are long legs: this bird prefers to live in sparse woodlands,
rather than in dense forests, which are relatively rare in southwestern Europe;
the Iberian magpie spends no less time on the ground or among bushes than on
trees.
On the ground, it moves with big jumps, flies well, but not for long – usually
only to the nearest tree or thicket of bushes. The voice is a recognizable magpie
chatter. The bird is active mainly at dusk, in the morning and evening, less
often at night (especially during the full moon). During the day, this bird
prefers to hide in thickets, among rocks or on a tree.
Of the senses, this species has the best developed vision and hearing. With
the help of hearing, the bird is able to distinguish insects crawling in the
grass. Vision allows it distinguishing small prey among the grass at dusk.
The food of this species is mainly insects and other invertebrates, less often
small vertebrates, seeds and fruits of trees and bushes. The bird itself turns
prey of various predatory birds and mammals; large snakes can ravage its nests.
The nest of the Iberian nocturnal magpie is a spherical structure of branches
with a hole on one side and with a nest cavity inside. To build a nest, birds
choose branches of thorny shrubs with large thorns. Eggs are usually light,
with darker specks; incubation lasts 18-19 days. There are usually 4 to 6 nestlings
in the brood, which become independent after 3 weeks.
The breeding season begins at the end of winter. At this time, male builds a
new nest or repairs an old one, and calls for females with a chatter. The female
chooses the male on the basis of personal sympathy and the ability to choose
a nesting site and build a nest. The Iberian nocturnal magpie is a monogamous
species, but pairs are formed only for one nesting season. The Iberian magpie
lives 7-10 years in average.
This bird species was discovered by Bhut, the forum member.
Kermez
(Podoces kermez)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: steppe regions of Central Asia with sparse vegetation.

Picture by Alexander Smyslov
Many corvids of Holocene epoch were easily adaptable birds
and they perfectly tolerated the human neighborhood. The ice age destroyed only
tropical local species, and a number of island species of these birds disappeared
due to human fault. The corvids of temperate zone that lived on the continents
survived the ice age, and some of them gave rise to a whole range of new forms
that differ in diverse behavior and ecology. Other corvids, on the contrary,
remained relatively inconspicuous inhabitants of their biotopes, slightly differing
from their ancestors from the human epoch. One of these species is the Central
Asian kermez, a descendant of the Turkestan ground jay (Podoces panderi).
It is a relatively large bird: body length 60-65 cm, wingspan up to 90 cm, weight
about 1.2-1.5 kg. The color is mottled; it combines rusty-red, white, gray and
black colors. Sexual dimorphism is not developed, but representatives of various
populations may differ markedly in color. The background color of the body is
usually red with gray nape, back and wings. There is an indistinct cross ripple
on the wings. The tail is black with a bluish metallic sheen, and the primary
feathers are white with black tips. In some local populations, the red color
may be replaced by ochre-red or fawn, and the gray color may cover the entire
head of the bird. The legs are always black, with strong straight claws. The
beak is straight, pointed, colored from gray to black. Eyes have a blue iris
and a round pupil.
Kermez is mainly a zoophagous bird, a skilled hunter of small animals: reptiles,
murine rodents, large invertebrates (hence the name of the species: “kermez”
– an evil spirit in Kazakh mythology). It stealthily sneaks up to the prey and
kills it with blows of its beak and legs. Like the ancestor, kermez flies very
reluctantly, only in case of extreme danger, but runs great, escaping from terrestrial
predators. It is also able to hide skillfully in the grass. This species is
territorial: outside of the breeding season, no bird tolerates rivals on its
territory, expelling them ruthlessly. Claiming the rights to the territory,
the kermez utters a sharp chirping trill; at this moment, the bird usually jumps
on a low bush or other elevation. There are 2-3 burrows at the territory of
the bird, from which the bird has expelled the rodent hosts; sometimes they
occupy abandoned burrows. The burrow serves as a shelter from the rain and a
place for nesting. For this purpose, it is renewed and deepened. Kermez can
defend itself from some small predators by rushing at them and inflicting sensitive
blows with its wings and beak.
The breeding season begins in March-April. Males at this time vocalize loudly,
calling females, and actively patrol their areas, indicating their presence
by vocalization. Fights break out between rival males at this time, during which
birds kick and beat each other with their wings. A nest of twigs is arranged
in a dense thorny shrub or on a low tree. The clutch usually contains 5-7 eggs,
which are incubated by both parents alternately for 17-18 days, starting with
the fourth egg. They also feed the nestlins alternately; one of the parents
stays in the nest and warms the offspring for the first week when the partner
is away. Young birds leave the nest after 15-16 days and roam with their parents
for the first month, learning to hunt and escape from enemies. Later, broods
and pairs break up. Sexual maturity occurs at the age of 9-10 months, and life
expectancy is about 15 years.
This bird species was discovered by Nick, the forum member.
Southern
jay (Cyanocitta mexicana)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: open spaces of North and Central America.

Picture by Alexander Smyslov
In the human era, the genus of American blue jays included
only two species, reliably isolated from each other geographically. In Neocene,
these successful bird species of the human epoch gave rise to numerous descendants
occupying various ecological niches and spreading noticeably wider than the
ancestral species. These are mostly forest birds, but one of the descendants
of Steller’s jay has mastered life in the open: the southern jay inhabits the
prairies and semi-deserts of North and Central America.
It is a smaller bird than the ancestral species: 25-30 cm in length along with
the tail. The blue color of the plumage of the ancestral species is kept only
on the back and on the belly; head, wings, neck and tail are black with a pearly
luster (in males) or grayish (in females). But in a flying bird, secondary feathers
become noticeable – blue with black cross stripes and a cyan metallic luster.
When such a bird takes off with a loud alarm call, it attracts the attention
of animals that are nearby, and they begin to look for signs of danger.
The crest of the ancestral species is almost completely absent in southern jay
– the feathers on the crown are only slightly elongated. In a state of excitement,
the bird raises these feathers and its head, when viewed from the side, begins
to seem disproportionately large. Beak is thin and pointed, black in color.
Tail and legs of this bird are elongated compared to the ancestral species,
so the southern jay looks like a long-legged magpie. It spends a lot of time
on the ground and runs fast, catching its prey or hiding from enemies. In case
of extreme danger, the bird takes off quickly. If there are cactus thickets
nearby, it prefers to hide among them.
The behavior of the southern jay is typical for corvid birds. It is an omnivorous
bird, but its main food is invertebrates, rarely berries. The bird likes to
peck the juicy cactus fruits, and when there is a lack of water, it eats their
flesh. Foraging for food, the bird shows considerable intelligence – if necessary,
it picks up the stems of cacti with a twig in search of insects. The main enemies
of this species are various birds of prey, snakes and small mammals, from which
it escapes both in flight and running.
This species is a sociable bird that gathers outside the breeding season in
groups of up to 10 individuals. During feeding, the birds behave noisily, constantly
communicating with sharp crackling calls. They spend the night in tall trees,
also in groups. Several flocks of these birds can occur on one tree, and often
in the morning individual birds can move to a neighboring flock.
On the northern edge of the range, nesting begins in spring. In the south of
the range, this bird does not have a pronounced breeding season – at any time
a series of monotonous melodic cries of males of this species are heared – they
invite females to their nests. The nest is arranged somewhere in bushes or in
thickets near a pond – it is cup-shaped, with a loose roof of twigs. The clutch
numbers from 3 to 6 eggs of bluish or bright blue color. Females incubate the
clutch for about 2 weeks, after which both parents feed the nestlings for another
2 weeks. The chicks take wing, but live with their parents for about a month
before they pass to an independent life and the families break up. These birds
are monogamous, but do not form permanent pairs and do not show strong attachment
to a breeding partner outside of the breeding season.
Young birds begin to breed from the next year of life. The average life expectancy
of a southern jay can be up to 15 years.
This bird species was discovered by Bhut, the forum member.
Parrot
jay (Cyanocitta pecularia)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: broadleaf forests in eastern North America.

Picture by Pavel Volkov
The genus of American blue jays is not numerous – it included
the blue jay itself, which lived in Holocene in the eastern half of North America,
and the Steller’s (or long-crested) jay, which lived in the west of the continent,
so these related species have never had interspecific hybrids.
In Neocene, this situation has persisted. In the center of North American continent
there is now a long, almost continuous strip of open spaces – prairies, where
blue jays, originally forest birds, feel uncomfortable, and therefore the evolution
of the descendants of both species of blue jays continued in different ways;
the descendants of the first species are exclusively North American birds, but
among the descendants of the Steller’s jay there is a snow
jay, a resident of the snowy forests of Siberia, changing its plumage
for winter. There are no such descendants of the blue jay, but among its descendants
there are unusual species too.
One of the species of this group is the parrot jay. Despite its name, it does
not live in the tropics, but in the broadleaf and mixed forests of North America,
in a temperate climate with mild, snow-free winters. Outwardly, it resembles
an ancestral species, but differs in larger size – about the size of a common
raven of Holocene epoch, with a proportionally more powerful beak – straight
and only slightly curved downwards.
The plumage of this species bears features inherited from the ancestral species.
Primary feathers are blue with narrow black cross stripes; wings are rounded,
with blunted tips. The back is cobalt blue, with a greenish metallic sheen on
the neck. The upper part of the head is black with narrow white “eyebrows”;
cheeks and throat are white. Chest and belly are pale yellow, and the tail is
white. Tail feathers are long, the tail resembles a magpie’s one; because of
it, the bird seems much larger than a raven. The middle four feathers have a
bright blue color with black cross stripes, along the edges of the tail the
feathers are grayish-blue without stripes. On the head of birds of both sexes
there are large crests of elongated blue feathers with black tips, which can
rise and fall, expressing the emotional state of the bird. There is no sexual
dimorphism in this species.
This bird received the specific epithet for its lifestyle – it is a noisy and
sociable bird, like a parrot. Many corvids are sociable birds, but blue jays
of Holocene epoch preferred to live in pairs or families, rather than in large
flocks, as crows or jackdaws did. And the descendant of these birds, the parrot
jay, prefers to live in flocks that consist of two or three families: this bird
is quite tolerant of relatives, especially when there is a lot of food in the
habitat.
The diet of this species has also changed: the parrot jay is mainly a herbivorous
bird. Its main food includes nuts, acorns, seeds of other trees, sometimes juicy
fruits of trees and shrubs. Sometimes it catches insects, spiders, and other
invertebrates, but it does it mainly to feed nestlins; adult birds almost do
not eat animal food, and a significant part of animal protein is obtained from
insects swallowed with food.
The mating season of the parrot jay occurs in spring and summer, especially
in the south of its range, where the winter is mild and snowless. This species
is monogamous, and pairs are often formed for life. Single males call females
with a special signal, and they evaluate the volume of the voice and the force
of the male. The mating call of this species is a loud dry trill, sometimes
performed for up to thirty seconds in a row. Calling the female, the male perches
on the tree top so that he can be seen. Outside of the breeding season, pairs
and family groups of these birds are kept in flocks of up to 20 birds. Due to
it, they are able to notice the appearance of dangerous animals, and collective
protection contributes to the survival of these birds. The alarm cry of these
birds is a loud rattling cawing. When a flock of birds reveals an enemy, they
make a deafening noise. For the night, the birds perch on the branches of a
tree in a continuous row, warming each other.
These birds do not build nests on their own, but they take away the ready one
from another bird or look for last year’s one that requires a little repair.
There are 3-4 eggs in the clutch of this species; their shell is pale green,
with black specks. The nestlins hatch after a few weeks; their parents feed
them first on insects, and then gradually switch them to vegetative food. In
the second half of summer, the chicks take wing and migrate with their parents,
to the north, when nuts, acorns and similar fruits begin to ripen there. There
the parrot jays feed, despite the arrival of winter. In Neocene, the climate
in high latitudes is milder than in Holocene, winters are warmer and less snowy,
and parrot jays stay there for a long time, flying away by the middle of winter,
when autumn abundance is exhausted. They fall prey of various predators – tree
snakes and mustelids ravage their nests, and various birds of prey catch these
birds during migrations. The most dangerous enemies of this species are owls,
catching them at night in places of overnight stays.
The parrot jay can live 16 years or more, but many of the birds die much earlier.
This bird species was discovered by Bhut, the forum member.
Poisonous
jay (Geocitta unedibilis)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: Mexican deserts and semi-deserts.
Due to their ecological plasticity and the ability to adapt to various natural
conditions, corvids are represented by a number of unique and specialized forms
in Neocene. One of these species lives in the deserts of Mexico – a poisonous
jay, a descendant of the blue jay (Cyanocitta cristata).
The body length of poisonous jay is 50 cm; the wingspan is up to 60 cm. The
beak is long and strong, slightly curved at the tip. The bird’s build is light,
it has a long tail and legs. A small feather tuft grows on its head. The coloration
is very bright, clearly visible from afar: the tail is striated, black and blue,
the rump is bright red, noticeable in flight and with the wings open; crown,
nape and upper part of the neck are dark blue, the crest is red with a blue
tip. The cheeks, lower part of the head, front of the neck, chest and belly
are white. Wings are black with longitudinal red stripes on the primary feathers.
The legs and beak are bright red. The eyes are large, orange, and the vision
is sharp.
Poisonous jays inhabit vast deserts and semi-deserts in North America. According
to their behavior, they are solitary, but the poisonous jays do not have strictly
protected individual territories, and sometimes several birds feeding together
can be seen at once. During the day, poisonous jays fly in the desert in search
of food, and at night they sleep in natural shelters. The diet is very extensive:
plants (fruits, seeds, herbs and roots), invertebrates, reptiles, small birds
and their eggs, small animals, carrion. There are quite strange objects in the
diet of the poisonous jay: poisonous plants and insects – bees, beetles (blister
beetles, ladybugs, some leaf beetles, etc.) and others. Their poisons are not
harmful to the jay: it does not get rid of them, but accumulates in muscles
and fat, becoming poisonous to predators. Therefore, the bright color of the
plumage is essentially a warning. Thanks to the rigid plumage on the head, the
poisonous jay can ravae the bee nests: the bird breaks the wall of the nest,
pecks the escaping bees and eats them whole, supplementing its diet with honey.
Due to the accumulated poison, the poisonous jay has no enemies – only the nestlings
that have not accumulated yet enough poison in the tissues may be attacked by
parasitic dipterans. However, the price of such protection is the short life
of adult birds: the resistance of the bird’s organism to the action of poison
steals a lot of resources from the organism.
In spring, birds form pairs. They dig their own burrow or occupy an empty one,
and lay up to 3-4 eggs at the bottom of the nest chamber. Then the female remains
to incubate them, and the male feeds her all this time. After 2 weeks, the nestlings
hatch, and the male has to feed them, and he specifically adds a certain amount
of poisonous insects to the juveniles’ feed. After a month, the juveniles fledge
and leave the nest.
Sexual maturity occurs at the age of 8-9 months; life expectancy is about 10
years.
This bird species was discovered by Mamont, the forum member.
Kukat
(Parapica kukat)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: Eurasia, Siberian taiga.
Despite some losses in species diversity in the human era, corvids turned out
to be one of the most successful groups of birds. A significant number of species
of these birds managed to survive successfully the human era, having kept a
high number and a wide range. Being iIntelligent and evolutionarily flexible,
they not only regained their diversity, but managed to partially replace birds
of prey after the Holocene-Neocene crisis. Among this family, birds of Perisoreus
genus, remaining in the shadow of their larger relatives, turned out to be a
more conservative group of birds. However, a remarkable species also appeared
among them – the kukat, a descendant of the Siberian jay (Perisoreus infaustus).
The name of the bird is the name of the witch in the Khakass myths.
The body length of kukat is up to 50 cm; the wingspan is 75 cm. The plumage
color is brown; the male has a little crest of black feathers. This bird resembles
a more stocky and short-tailed magpie in appearance. Kukat is an omnivorous
bird with a predatory bias. It easily pecks nuts and berries, but much more
often attacks small animals, rodents, and squirrels among them, bats, birds
(including pigeons) and reptiles. Kukats are sociable birds that keep in family
groups: parents with juveniles of the current year, siblings from the same brood,
in winter also several pairs of adult birds from the neighbouring territories.
Living in a seasonal climate has left an imprint on the behavior of the bird.
For the winter, the kukat gathers food stocks: it hides seeds and carcasses
of small animals in secluded places, usually in hollows or rotted through tree
trunks. Such caches are ravaged from time to time by rodents and ground-dwelling
carnivores.
The breeding season begins early, in late March – early April. At this time,
the male behaves very aggressively towards other males, actively expelling them
from his site. Usually it is limited to sharp squeaky cries, although sometimes
males peck and beat each other with their wings. Young males, who have just
occupied the breeding site, try to keep the females on their territories, displaying
themselves by jumping and making crackling trills, and “married” males preen
the females’ plumage and jointly repair their nests after winter. There are
5-6 eggs in a clutch with a gray shell covered with small brown spots. Incubation
lasts 3 weeks; the parents replace each other on the nest and do not leave the
clutch. The nestlings hatch covered with thick juvenile down of grayish-white
color and are blind, but they open their eyes already on the fourth or fifth
day of life. The parents feed them for about six weeks, after which the young
birds leave the nest and accompany the adults until the beginning of March.
Sometimes the young birds leave their parents and roam in a flock along the
borders of the territories of adult birds. In juvenile plumage, the male lacks
a crest, but there is a black “cap” instead of it. In young birds, the eyes
have a gray iris, which is a signal that reduces the aggression of adult birds.
Sexual maturity occurs at the age of 2 years; life expectancy reaches 15 years.
This bird species was discovered by Nick, the forum member.
White-throated
resonatornis (Resonatornis albopectoralis)
Order: Passerine birds (Passeriformes)
Family: Corvids (Corvidae)
Habitat: Equatorial Africa, Congo.

Picture by fanboyphilosopher
Congo basin in Neocene represents the extensive territory
covered by circuit of lakes and bogs among sites of impassable tropical forest.
Flooded forests of Congo are adverse for life of large tetrapods, but represent
a perfect habitat for climbing and flying creatures. The variety of birds in
Congo basin of Neocene epoch is comparable to those in territory of Amazonia
of human epoch. Among birds of Congo basin corvine birds take an appreciable
place. This family has considerably succeeded in Neocene. These birds had managed
to receive advantages in struggle for existence, living in cultural landscapes
of human epoch. Natural ingenuity and ability to adaptation had helped these
birds to occupy various ecological niches in ecosystems of Neocene. For Eurasia
and North America large birds
of prey, related to corvids and descending from representatives of this
family, are characteristic, and in forests of Zinj Land the separate family
of tropical birds, crestbills, also related to
corvids, had evolved. But, in addition to them, in Neocene also various representatives
of true corvids exist, which in due course of evolution had developed the various
adaptations, helping to survive in specific conditions, characteristic for their
habitats.
Tropical rainforests of Equatorial Africa, growing in Congo basin, represent
a habitat of one interesting representative of Neocene corvids. It is rather
easy to hear it among thickets of branches and lianas. The voice of this bird
is very loud and unpleasant, resembling bull bellow. It is one of characteristic
sounds of gallery forests crossed by rivers.
The bird uttering such cry appears unexpectedly small for such loud voice. It
may be hardly noticed among lianas and in tree crones. During the cry under
its skin on breast two big air bubbles of white color strengthening loudness
of its voice are inflated. This feature has determined the name of this species
– it is white-throated resonatornis.
This species is a raven-sized bird weighting about 1 kg. Colouring of resonatornis
is not striking – plumage of bird is almost entirely black with greenish metal
shine on wings. On breast of bird there is large white spot stretching upwards
up to the throat. Besides for it, other ornamental features at this species
are small fluffy feather cop above the beak and blue skin of eyelids. The main
feature of bird is visible when it begins calling. Before uttering the impressive
call, white-throated resonatornis makes some deep breaths, rising on paws. The
trachea of this bird near to entrance to lungs forms two outgrowths which are
located under skin. They have elastic walls, capable to stretch very strong.
During an exhalation bird “pumps up” these outgrowths with air, and they are
increased up to the size of orange. The skin above them forms the pair of folded
“pockets” of white color hidden under feathers. When resonators are inflated,
they appear from under feathers of bird. Such inflated resonators are well appreciable
to competitors and partners of opposite gender. With the help of calls and displaying
of inflated resonators this bird declares the rights on territory.
Resonatornises are sociable birds, constantly exchanging signals with each other.
The usual voice of these birds is similar to croak with various modulations;
in “dictionary” of resonatornis there is about ten signals used by birds in
different situations. The birds free of nesting gather in small flocks and migrate
in forest. Wide wings and long tail permit this bird to move easily in forest
canopy, changing direction of flight.
Resonatornis the powerful deep beak assisting to crush firm seeds and to tear
meat equally easily. These birds are omnivorous with inclination to zoophagy.
Having an opportunity, they catch smaller birds, reptiles or mammals, ravage
the bird’s nests and eat invertebrates, even beetles and snails covered with
firm shell. These birds live strictly in forest canopy and do not fly down to
the ground.
Resonatornises nest in pairs which are kept till their whole further life. The
nest is built by both partners. It represents a construction of thin branches
and other vegetative material. Frequently resonatornises occupy available nests
of other birds, sometimes even attacking on the lawful owners and killing them.
In clutch of resonatornises it may be up to five eggs which are hatched exclusively
by the female. Male protects territory and gathers food for the female and posterity.
When nestlings hatch, both parents feed them. At the age of six weeks young
birds leave nest. They differ from adults in color of spot on the breast – it
is not white, but dark grey. Within one year birds rear two hatches. Young birds
become capable to breeding at the age of two years. Life expectancy of this
species reaches 50 years and more.
Close species of resonatornises live in forests of Central Africa:
Ornamented
resonatornis (Resonatornis ornatus) is smaller (daw-sized) species.
Feathering of this species is black-and-white: primarily feathers are white
with black tips; tail is long and white. Lateral parts of head are also white.
Black color on wings has intensive green, and on breast – purple metal shine.
Resonator bubbles at this species are bright orange. Voice sounds like long
“metal” ringing whistle more often heard in midday hours. This species eats
mainly invertebrates and small vertebrates – frogs and nestlings of small birds.
White
resonatornis (Resonatornis albus) differs in colouring from other species:
its plumage is almost entirely white, except for secondary feathers and waist
having black color. It is a large bird: weight of adult individual is about
800 grammes. It has short fan-shaped tail and thick beak adapted to feeding
on small animals and firm seeds. This species eats reptiles, including snakes
up to one meter long, attacks birds and eats crabs and insects with firm shells.
Head of this species is decorated with a bunch of fluffy feathers growing on
forehead with soft shaft and down-like vane. Resonator bubbles of this species
are grey; the voice is similar to sound of working mechanical saw. The adult
individuals, having formed breeding couple, frequently cry a duet, continuing
the call of the partner. This species cries in the morning more actively.
Green
resonatornis (Resonatornis viridis) is the smallest species of the
genus (starling-sized one). It differs in dark green color of plumage with intensive
green metal shine on wings, back and nape. It has tenacious paws; this species
can easily climb up trunks and branches of trees. Also it has pointed awl-shaped
beak of white color adapted to getting of insects from cracks and other shelters.
Instead of soft cop this species has elastic bristle-like feathers on forehead
and about nostrils protecting feathering against pollution. Resonators are colored
white. Voice of this bird is shrill long-lasting whistle. This species is active
in twilight, in the morning and in the evening.
Wotan’s
raven (Eoaquilorax wotanii)
Order: Passerine birds (Passeriformes)
Family: Predatory corvids (Carnocorvidae)
Habitat: Loki Island, various types of habitats.
In Neocene, a new group appeared among the carnivorous birds of the Holarctic
– eagleravens, large carnivorous corvids that occupied the ecological niche
of eagles, but remarkable in complex behavior and intelligence characteristic
of their ancestors of Holocene epoch. Various species of eagleravens inhabit
the steppes, forests
and sea coasts of Eurasia
and North America. On Loki Island in Atlantic a primitive representative of
their family lives – a Wotan’s raven. It is a relic of the Early Neocene adaptive
radiation of predatory corvids, an endemic of Loki Island remained in isolation
and the top predator of this island. It is a close relative of the forest eagleraven
of Europe. Since it has no enemies on the island, it has lost the ability to
fly.
Wotan’s raven is a brown bird with black spots appearing at the age of 18 months,
by the onset of sexual maturity. This species lives and hunts in groups of 6-8
individuals. It inhabits mainly the forests of the island, but often appears
on the coast, where it takes away the carcasses of gannetwhales
from griffon skuas.
The stature of an adult bird is up to 70 cm, half the height of the bird is
made up of strong legs. The weight of the bird is up to 8 kg. The wings have
not disappeared, but are greatly reduced – they are only about 50 cm long, with
thin bones; the sternum is very small. The bird has a large, laterally compressed
beak with a pointed curved tip.
Wotan’s raven is a predator; it attacks forest and sea birds, often surveys
the sea coast in search of corpses cast ashore by the sea and can attack young
gannetwhales. It also hunts the large local lizard fafnirella
(Fafnirella nigra). Wotan’s ravens often ambush prey at places frequented
by it. When hunting gannetwhales, predators drive the intended prey away from
the group and inflict deep wounds with their beaks, as a result of which the
prey dies from blood loss. Sometimes birds attack gannetwhales returning from
the sea after feeding and beat them, forcing them to regurgitate part of their
prey.
Sexual dimorphism is not pronounced in this species. It is a monogamous bird;
breeding pairs preserve for life. The courtship season begins in the middle
of spring. Males actively help their mates repair last year’s nests, or build
new ones. When meeting an opponent, they cawk loudly and kick each other, without
using beaks in intraspecific conflicts. Young and solitary adult males, not
having a mate, gather in the open glades of the island and arrange a courtship
display, more like a conflict: they vociferate, rise as high as possible on
their toes and sometimes fight, kicking and pushing too active competitors.
Females choose the largest and strongest males.
The nest is built in the thick of bushes and represents a pile of branches with
a depression lined with soft grass. Fertility is low: once every 2-3 years,
the female lays only one egg and incubates it for a month. The nestling is initially
naked and blind, until midsummer it stays in the nest, fed alternately by both
parents. After leaving the nest, the young bird learns for a long time to hunt
and interact with parents and their neighbors. For a year and a half, the young
bird lives in the parent group, and shortly before the new clutch it is expelled
and begins an independent life. At the age of 3, sexual maturity comes; life
expectancy is up to 50 years.
This bird species was discovered by Nick, the forum member.
Yel
(Aquillorax yel)
Order: Passerine birds (Passeriformes)
Family: Predatory corvids (Carnocorvidae)
Habitat: Pacific coast of North America, cold and temperate latitudes.
Eagleravens and their relatives represent one of the successful experiments
of nature in Early Neocene. These birds quickly took the place of large feathered
predators in ecosystems recovering from damage caused by man and nature at the
boundary of Holocene and Neocene. In the Northern Hemisphere, these birds inhabit
various natural zones: forests, mountains and sea coasts. One of the largest
species of this group is the sea
eagleraven (Aquillorax maritimus) from Eurasia. At the Pacific coast of
North America, it has a close relative – yel (the name of the Great Raven among
the Tlingit).
This bird species is only slightly inferior in size to its relative from Eurasia:
the body length of an adult bird is up to 100 cm, the wingspan is up to 2 m,
the weight is about 7 kg. The color of the yel’s plumage is much darker: back
is dark brown, belly is gray, and the tail feathers have white tips. Legs are
featherless and gray in color. Beak is very light in color – white with a yellowish
tinge. At the base of the beak and on the lower jaw, there is a bare skin of
light gray color. Yel looks slightly larger than the sea eagleraven, but this
impression arises because of its physique: it has longer legs, neck and beak.
The bird’s beak is also compressed from the sides and has a strong hook-shaped
tip. The nostrils are capable of closing with the help of movable fleshy valves.
Yel eats seafood and prefers to search for food in the coastal zone. Usually
birds of this species ransack shallow waters and littoral during low tide, but
some individuals can forage in rivers and large lakes. The basis of the diet
consists of marine mollusks. The bird crushes small mollusks with its beak and
swallows, having previously cleaned the shell fragments with a movable tongue
with horn brushes at the edges. To bivalve mollusks, yel cuts an adductor muscle
with its beak, dissecting it with the beak tip. In relation to large mollusks,
the bird uses a different tactic: it takes them ashore and splits them with
a stone, which it throws on the shell from above. In the same way, yel extracts
mollusks and barnacles attached firmly to the stones. Also, yel does not disdain
worms, cephalopods and, less often, dead fish. The bird shows aggression towards
its relatives and rarely allows them to get closer than thirty meters. Even
birds from a breeding pair prefer to keep their distance from each other during
the search for food and meet only near the nest. In the northern part of the
range, the yel is a migratory species, flying away for the winter in mid-September-October,
returning at the end of March-April. Wintering takes place at the southern tip
of the continent, in the area of the Panama Strait. Some individuals cross the
strait and reach the northern coast of South America.
This species is monogamous. Pairs persist for a long time: for several nesting
seasons, sometimes until the end of life. Despite the size of the birds, nesting
takes place in trees and steep cliff slopes in mid-April-early May. Before the
beginning of nesting, males gather in groups somewhere on the border territories
and arrange courtship displays: they jump, open their wings and cry. Fights
occur only between young males; less often an adult widowed bird enters them.
Established pairs of birds also visit the lekking grounds, but do not take part
in courtship games so actively.
A pair of yels has its own protected nesting area; however, with a large population
density, nests can be located at a very close distance from each other. Birds
build a nest of twigs and branches cast ashore by the sea, lining it with grass
and dry algae. From time to time, mates replace each other on the nest, giving
the another bird an opportunity to rest and feed. There are usually 3-4 eggs
in the clutch, less often 2 or 5. The incubation period is 25-27 days. Adult
birds protect nests jealously, so predators rarely encroach on eggs and offspring.
The nestlings are blind and helpless, but covered with thick black down. They
open eyes at the age of a week and begin to beg for food actively on the 10th
day. Young birds fully fledge at the 2nd month of life, but spend one more month
in the nest after that, gradually learning to fly. Birds of the first year of
life differ from adults by a darker beak and a wide strip of skin without feathers
at its base. During their stay in the nest, some of the juveniles die of starvation
due to competition for food: usually the parents manage to feed two young birds.
After leaving the nest, young birds stay with their parents until mid-autumn
(in the north – until the autumn migration), learning to extract shellfish and
open their shells. These birds usually live 33-36 years.
A smaller subspecies, the California yel (Aquillorax yel insularis),
lives at the California Island. It is a non-migrating subspecies of small size
– the wingspan of an adult is about 160 cm, weight up to 4 kg. The color of
the plumage of these birds is much lighter compared to the mainland subspecies:
rusty-red back, white belly and very light-colored legs. These birds nest on
the ground among rocks and in other shelters, and feed on land snails and small
vertebrates in addition to marine animals. Island birds sometimes appear on
the Pacific coast of the continent and form pairs with the mainland form, giving
fertile offspring with no limits.
This bird species was discovered by Nick, the forum member.
Susanoo,
Japanese eagleraven (Aquillorax insularis)
Order: Passerine birds (Passeriformes)
Family: Predatory corvids (Carnocorvidae)
Habitat: mountain forests of Japan Islands.
In Neocene avifauna of Holarctic, the role of large feathered predators was
undertaken by the descendants of corvids – large birds of prey that belong to
a separate family of predatory corvids. Due to their mental abilities, these
birds managed to replace the accipitriform birds of prey, which were significantly
affected by human activity. The ecological niches of small feathered predators
are occupied by the descendants of shrikes,
and predatory corvids became the analogues of eagles in Holarctic. The greatest
diversity of these birds is observed in temperate and cold climatic zones. One
of the southernmost species of these birds is the susanoo, or Japanese eagleraven,
named after the Japanese god of storm and wind.
It is a relatively small species of eagleravens: the body length of an adult
is about 70 cm, the wingspan is up to 160 cm. It is a close relative of the
sea eagleraven (Aquillorax maritimus),
a migratory species that lives along the coast of Arctic Ocean. These species
had one common ancestor, whose sedentary population inhabited the islands. In
the appearance and behavior of susanoo, there are many features characteristic
for its ancestor.
It has more graceful build compared to the sea species. Elongated legs have
hooked claws. The wings are wide; the tail is elongated and fan-shaped. Despite
its size, the bird is able to fly skillfully in the forest or among the rocks,
and can snatch off the prey from a rocky ledge with its paw. Unlike the forest
species from Eurasia, the susanoo has retained the light-colored plumage color
characteristic of its ancestor from the ocean coasts. On the head of adult bird,
there is a black “cap” covering the forehead, crown and nape. The upper part
of the body is glaucous with a blurred scaly pattern formed of the dark edges
of the feathers. The tail feathers are white, and approximately from the middle
of the length to the tip they are colored black. Belly is light gray, and some
feathers have black tips. Legs are not feathered, black in color. Sexual dimorphism
in coloration is not pronounced, the female is only slightly more massive than
the male.
Live prey predominates in the diet of this species. The beak of the susanoo
is strongly curved at the tip, deep, black with a white tip. Around the eyes
and the base of the beak there is a ring of bare skin of meat-red color, protecting
the plumage from contamination.
The usual voice of this species is harsh, raspy calls, which sound like a coughing,
but the mating call sounds like loud trumpet sounds.
The susanoo feeds on terrestrial mammals; while doing it, the pair often acts
together: one bird drives prey for the other, hidden in ambush. When there is
a lack of food, especially in winter in the mountains, breeding pairs unite
and jointly hunt larger prey like young deer and dwarf harelopes. In the spring,
such a union breaks up, and each couple lives on its own territory. But next
winter, the same pairs of birds can unite again for hunting.
This species is a strict monogamist; a couple is formed for life. Personal affection
plays a ggreat role in the life of susanoo: usually a pair of birds that poorly
fit to each other in temperament quickly disintegrates, and a successfully formed
pair persists for life. Even a formed couple in the spring utters a mating call,
which denotes the rights of birds to the territory.
Susanoo builds a nest on rocks, choosing places inaccessible to terrestrial
predators – this is a legacy of the behavior of a marine ancestor that nested
on the ground, a kind of compromise between behavioral features and a new habitat.
The nest is renewed every year and used for many years in a row. Usually birds
search for a new place to nest if the old one is destroyed by natural disasters.
Nesting begins in late winter or early spring. There are 2-3 eggs with a light
gray shell in a clutch; incubation lasts about 40 days. The nestlings hatch
blind, but covered with thick black down. They begin to fledge at the age of
1 month, but leave the nest only at the age of 3 months, and stay with their
parents until autumn, learning to procure food.
Young birds that have formed a pair learn to hunt together for a long time.
This species has a developed system of signs and poses that serve for communication.
The main sign of trust is cleaning the naked skin around the mate’s eyes with
a beak. The lifespan of these birds can reach 60 years. Sometimes old birds
can “cede” their territory to one of their own juveniles of the last broods
and help care for their offspring, as well as hunt together and take over a
part of the territory from neighboring pairs.
Koraka
(Koraka tropicalis)
Order: Passerine birds (Passeriformes)
Family: Predatory corvids (Carnocorvidae)
Habitat: Hawaii, forests of various types.
In Neocene, the characteristic birds of Holarctic realm are predatory corvids
– carnivorous descendants of corvids, which have become ecological analogues
of eagles and vultures. One of the species of predatory corvids settled at the
Hawaiian Islands through Japan, giving rise to the endemic genus koraka (the
name means “raven” in Hawaiian). It is a close relative of the Japanese susanoo,
and also descends from the sea
eagleraven. This species is the result of a rare event of settling of the
continental species to remote Hawaiian Islands.
On the archipelago, koraka mastered forest habitats, avoiding competition with
ethereal kites and other seabirds.
Body length is about 70 cm, wingspan up to 130 cm; koraka is shorter-winged
than susanoo. The tail is relatively short and wide. The legs are of medium
length, with blunted claws – the bird often walks on the ground and does not
use them to grab prey. The head is large, with a powerful beak similar to that
of the forest eagleraven,
but equipped with a more prominent hook at the tip than in continental forms.
The plumage of the koraka is pale brown with numerous longitudinal strokes on
the back; the cheeks, throat and middle of the chest are black, the rest of
the head is gray. Beak and legs are grey. Young birds are completely dark gray.
Skin on the head around the base of the beak to the eyes is covered only with
bright red skin. Sexual dimorphism is not pronounced. The calls of this bird
are similar to the cawing of a hooded crow; other signals are less often used.
This bird inhabits Hawaiian rainforests in valleys, as well as coastal forests,
and lives in groups of up to 10 individuals. Outside of nesting time, birds
roam through the forest, staying in one place for several days. The food is
very diverse, including small birds and mammals, young ungulates and chicks
of giant geese, large reptiles and amphibians, invertebrates. Korakas can attack
heron colonies and eat eggs and chicks, as
well as take away prey from larger predators. Korakas living in coastal forests
fly along the coast after a storm, eating sea creatures washed ashore. The nestlings
can be attacked by birds of prey
and lizards.
There is no pronounced breeding season, but if one pair in a flock begins nesting,
this serves as an incentive for the rest ones. This species is monogamous, with
1-2 eggs in a clutch. When nesting, they build nests at a close distance, and
at least two birds always guard the colony, replacing each other. A month later,
the gray-down-covered and blind nestlings hatch, to which the parents bring
food to the nest. Young korakas can stay in the parent pack, but more often
they keep in a separate group and gradually stop following their parents. They
create their own flock and move out of the parental territory. Flocks of young
birds stay in neutral territories or in less convenient places for life, gradually
displacing other relatives from their territories. Young birds often leave flocks
of young, forming pairs with birds from established flocks.
Sexual maturity is at the 3rd year of life, life expectancy is up to 70 years.
This bird species was discovered by Mamont, the forum member.
Alpine
thanatos, camulos (Thanatopteros camulos)
Order: Passerine birds (Passeriformes)
Family: Predatory corvids (Carnocorvidae)
Habitat: Alps and Pyrenees, some individuals migrate to the Atlas Mountains
in winter.
Alpine thanatos, or camulos (named after the Gallic god of war), is the closest
relative of thanatos, living
in the Himalayas, and simurg, that inhabits western
Asia. These large carnivorous birds belong to predatory corvid family, which
originates from the corvids of human era and is widespread in the Holarctic
zoogeographic realm. Representatives of predatory corvids are ecological analogues
of large birds of prey of the human era – eagles and vultures.
Camulos is smaller than its eastern relative: its wingspan is up to 2 meters.
The plumage is black and silvery, with a pronounced metallic sheen on the back,
darker on the belly. The tail is short, but wide, fan-shaped. Beak is very long,
with a small hook at the tip. Corneous cover of the beak is brightly colored:
the base of the beak is red, the lower jaw is yellow closer to the middle and
up to the tip, and the curved tip is black. Bare skin around the beak and eyes
is also red – it is needed so as not to contaminate the plumage while eating
meat. The Alpine thanatos has rather long legs, as it often hunts on the ground.
They are featherless, their skin is colored brown, and the claws are of medium
length and slightly curved.
This predator mainly hovers high above the mountains in search of food. Vision
allows him to look at the earth from a distance of up to five kilometers. Periodically,
the bird lands on large rocks somewhere in the mountains, or on trees in valleys,
and surveys the surroundings from there. At night, camulos hides in shelters
among rocks or in the crowns of large trees in valleys, where it rests until
the morning.
Harelopes and porcippulas are the main prey of camulos. The bird can kill them
by striking them with its beak, or pushing them off rocks. In addition to large
animals, it can also hunt rodents, rabbit
burrowers, birds and reptiles. Occasionally,
the camulos catches fish and amphibians in mountain rivers and streams with
its long beak. This bird willingly eats carrion and is its main consumer in
the mountains. An adult bird has no enemies, and mammals (cats, mustelids, predatory
dormice), smaller birds of prey and snakes can be dangerous for nestlings.
Camulos is a strict monogamist: these birds keep family pairs for life. After
the formation of a pair, the male and female constantly keep and patrol the
mountains together, watching each other. These birds of prey nest on a rock
or a tall tree, throwing a bunch of branches in the chosen place and covering
the nesting platform with grass and feathers. At the end of March, the female
lays 1-2 eggs. She is periodically replaced on the nest by a male so that she
can eat. After 41-42 days, a naked and blind nestling hatches. Both parents
bring food to it, and at first one of the parents protects and warms it. After
the appearance of juvenile fluff in the nestling, the parents begin to leave
it alone in the nest for a while. After the young camulos learns to fly (at
the age of 4 months), it accompanies its parents for several months while searching
for food and learns hunting techniques. Closer to the next year’s spring, the
young camulos begins an independent life; sexual maturity begins at 4 years
old.
Life expectancy reaches 70 years.
This bird species was discovered by Mamont, the forum member.
Unicorn
bird (Monocerornis chrysocornis)
Order: Passerine birds (Passeriformes)
Family: Crestbills (Ctenorhamphidae)
Habitat: Zinj Land, savannas and woodlands.
Birds of the crestbill family are endemic to the East African microcontinent
(Zinj Land). They are widespread mainly in forests, where most of their species
are found. Some crestbills have mastered the islands off the coast of Zinj Land,
forming small endemic forms on them. But in any case, most of these birds tend
to forest habitats. However, there are species in the family that have migrated
from the forest to the open area – to savannas and woodlands in the north and
northwest of the subcontinent.
The representative of the savanna crestbills is a unicorn bird, not inferior
in size to the great false
calao, a very large forest-dwelling species. It even seems larger than the
false calao because of its long legs, which make it easy to walk on the grass.
The unicorn bird is similar in lifestyle to the maned
crow (Leontocorvula leonina), although this similarity is due only to a
similar lifestyle.
The unicorn bird is a large crestbill species having a wingspan of up to 120
cm. It has a somewhat elongated neck, wide wings with rounded tips and an elongated
fan-shaped tail. Since the unicorn bird lives in an open area and spends a lot
of time on the ground, its legs are very long – the height of an adult bird
reaches 70 cm. A characteristic feature of the bird’s appearance is the growth
on the beak – growing vertically, narrow and long (its height exceeds the length
of the bird’s head). The coloration of the unicorn bird does not differ in variegation:
the main background is black with a purple reflection on the wings. The undertail
is snow-white, and there is a patch of white plumage on the back, usually hidden
under the wings. The tail feathers at the tips are black-and-white with a cross-striped
pattern. The beak and outgrowth on it are colored white with a slight yellowish
tinge, and the tip of the “horn” is bright yellow – hence the species name meaning
“golden-horned”). The areas around the eyes and beak are featherless and covered
with wrinkled gray skin. The bare legs are bluish-gray. The female differs from
the male in a larger size.
The unicorn bird’s voice is very loud and sounds like a shrill whistle. During
the vocalization, the bird’s throat is inflated noticeably. Due to its keen
eyesight, the unicorn bird notices the approach of a predator in advance and
is a good “watchman” for the goatlopes
– large local herbivores.
This bird feeds mainly on large invertebrates and small vertebrates, but about
40% of its diet consists of seeds and fruits of various plants. The unicorn
bird often feeds near the herds of goatlopes, snatching from the grass small
animals that they have scared away. These birds prefer to escape from predators
by fleeing, abruptly changing the direction of run and hiding in the bushes.
To protect themselves from large predators, birds stay close to herds of herbivorous
mammals.
The unicorn bird is a monogamous species. A pair of these birds is formed for
several nesting seasons and outside of the breeding season, the birds keep together.
Courtship display includes dancing of the male near the female and displaying
of the color of the beak and horn against the background of the plumage. To
do it, the male squats, leans forward and spreats his tail and wings like fans.
Fluffing the feathers on his back, he turns his head to one side and stomps
in front of the female, making a hoarse “cooing”.
The nesting of the unicorn bird begins at the end of the dry season, and the
nestlings appear at the beginning of the rainseason. There are up to 4 eggs
in the clutch of this species. A large open nest is arranged on a tree, in the
fork of the branches. Usually birds occupy any other bird’s nest, only slightly
repairing it. If there are no suitable nests in the breeding territory, the
birds build the nest on their own. The foundation of the nest is made of large
branches, which the birds heap among the branches. The nest itself is woven
of flexible twigs and lined with grass. Only the female hatches, and the male
feeds and protects her. At this time, he behaves aggressively and can attack
small animals, crying loudly and flapping his wings.
The nestlings grow in the nest for about one and a half months. Having fledged
and learned to fly, they leave the nest and wander around the parent territory
for a while. The plumage of young birds lacks luster, the legs are grayish-black,
and the horn-shaped outgrowth on the beak is only slightly noticeable. In the
second year of life, they become sexually mature and get an adult plumage color.
The life expectancy of this species is up to 25 years.
Multicolored
tufted crestbill (Cristoctenops polychromus)
Order: Passerine birds (Passeriformes)
Family: Crestbills (Ctenorhampidae)
Habitat: Zinj Land, tropical forests.
The Neocaenic family of crestbills is endemic to Zinj Land and is represented
primarily by small and medium-sized forest birds. In this family several genera
represent the natural group of “crestbills of paradise”, among which various
species differ in development of bright colors and ornaments of feathers. Representatives
of tufted crestbills develop on the head an erect or movable crest, supplementing
the growth on the upper mandible typical of the family.
The multicolored tufted crestbill is one of the most colorful-looking members
of the group. It is a bird of the size of a small daw with plumage which combines
several bright colors. The head of the male of this bird is painted in a charcoal
black color with a greenish metallic sheen. Against this backdrop the iris of
the eye of white or bluish color stands out. The bird’s beak and rounded horny
outgrowth on the upper mandible are white. The outgrowth on the upper mandible
is complemented by a thick black tuft on the crown and nape, the edge of which
continues the line of the outgrowth edge. The back of the neck, back and wings
of the male are colored bright green with a spotty pattern, which is formed
by the brownish tips of feathers. The front part of the neck is lemon-yellow
with a white bottom edge. The belly and undertail of the bird are crimson-red;
the tail is white. The female of this species is colored much more modestly:
it is a dull green with a white throat and a black head. The beak and growth
on her upper mandible are dark gray.
This species feeds mainly on small fruits and seeds, but often pecks insects
crawling on plants.
A courting male utters a loud buzzing trill, sitting on a branch without leaves
and exposing itself. The female behaves secretively and her special call reports
to the male her presence. Hearing the call, the male begins courtship displaying,
dancing and bouncing on a branch with his wings spread. At that he shakes his
head and fluffs yellow feathers on his neck.
The male of this species is not involved in nest building. His role in the preparation
of nesting is reduced exclusively to fertilizing the female, but it is usually
paired with only one female. This is due to the fact that after the start of
the nesting the male takes a certain care of female. He gathers insects and
puts them in a special sublingual pouch that can be stretched. The male keeps
on the edge of the nesting territory of the female and from time to time beckons
her, uttering a particular sound. The female briefly flies off of the nest,
flies to the male and receives a portion of the gathered insects. She behaves
the same way while rearing offspring. Due to this feature a couple can bring
up a brood of 7-8 chicks: more than that of other small crestbills. The juvenile
plumage of young birds looks like the color of the female’s plumage, but males
have darker heads. As the crest of the male grows to sexual maturity, at the
same time his plumage acquires multiple colors.
Sexual maturity comes at the age of 11-13 months; a lifetime is approximately
8 years.
Translated by FanboyPhilosopher
Emerald
crestbill (Iridonephele smaragdinus)
Order: Passerine birds (Passeriformes)
Family: Crestbills (Ctenorhamphidae)
Habitat: Zinj Land, tropical forests.
In Neocene, a new family of passerine birds appeared at the territory of East
African microcontinent (Zinj Land) – crestbills, descendants of corvids. In
due course of evolution, species settling various habitats in the rainforest
appeared among them, and one of their
species became a true savanna inhabitant. There is a special group of “crestbills
of paradise” among the forest species of crestbills, which includes some closely
related genera of small birds with bright coloration and feather decorations
– crests, elongated tails and tufts of thin elongated feathers on the back and
neck. The genus of maned crestbills (Iridonephele), which includes several small
species notable for their bright coloration, belongs to this group. These birds
are characterized by a sharp sexual dimorphism: the female differs from the
male in camouflage coloration and behaves very secretively.
The emerald crestbill is a typical representative of maned crestbills. It is
a starling-sized bird with a short fan-shaped tail and wide wings with rounded
tips. The plumage of the male of this species is mainly green with a metallic
sheen on the wings and neck. The area around the eyes is colored bluish-green;
there is a red plumage area on the lower back under the wings, and the undertail
is brown. The outgrowth on the bird’s beak has a rounded shape; the beak itself
is shortened, deep and strong. The beak and the outgrowth on it have a white
color with a creamy tint.
Elongated green feathers grow on the back of the neck and shoulders of the male
(the feathers on the shoulders are much longer). The male shows them during
the courtship display, which takes place several times a year.
On the contrary, the female of the emerald crestbill is distinguished by a very
inconspicuous plumage. The main color is dark green without metallic luster;
the edges of the feathers on the neck, back and wings are brown, forming a characteristic
scaly pattern. The beak and the outgrowth on it are of gray color.
Pronounced sexual dimorphism is associated with the peculiarities of reproduction
of this species: the care of offspring lies entirely with the female. There
are no permanent pairs in this species; one female can mate with several males,
and one male with several females. Because of this, there may not be juveniles
in the brood that have a common father.
The diet of the emerald crestbill includes mainly insects, although up to 40%
of the food may be soft-fleshed fruits and young leaves.
During the courtship display, the male shows himself, having chosen a branch
illuminated by the sun for the display. To be better visible, he often bites
leaves from a branch at a distance of up to a meter in both directions from
the place where he is displaying. The courtship display is preceded by loud
calls consisting of a long series of double jerky “syllables”: “haha-haha-haha-haha!”
The “laughter” of the emerald crestbill is often heard in areas of the damaged
forest canopy, where several males gather for courtship display. When the female
approaches the lek, she utters a short signal indicating her presence, but hides
among the foliage, flying out to the male she likes only for mating. The displaying
male fluffs the plumage on his neck and shoulders, lowers his wings and bends
his head. He emits specific buzzing trills and shakes his whole body finely,
showing feather ornaments. Due to the trembling of the feathers, it seems that
he is surrounded with a colored cloud (hence the name meaning “rainbow cloud”).
The female leaves her hiding place for a few seconds – she flies out of the
foliage and perches next to the male. At this point, he interrupts the current
and quickly mates with her, after which the female flies away again.
Despite the brightness of the color and noisy behavior, the emerald crestbill
rarely becomes prey to predators. This bird has sharp eyesight and a good reaction,
so the emerald crestbill notices the danger in time and manages to escape. In
addition, emerald crestbill males have a habit of mobbing of feathered and four-legged
predators. Noticing a predator, males gather in a flock and begin to cry loudly
and fly over the predator’s head, accompanying its movement through the forest.
Even a ground predator roaming in the undergrowth causes a mobbing behavior
in them.
The female builds a nest of twigs and scraps of fibrous palm leaves among the
branches. There are only 3 to 4 eggs in the clutch of this species. Only the
female incubates and feeds the nestlings. The offspring leaves the nest at the
age of 5 weeks and after another week begins to lead a completely independent
life. The juvenile plumage of the emerald crestbill resembles the coloration
of the female, only the male’s beak gets a characteristic color by the time
of leaving the nest. The young bird becomes sexually mature at the age of about
14 months. At this time, males have a plumage characteristic for an adult.
In the forests of Zinj Land there are some more close species of maned crested
beaks:
Greyish-white
crestbill (Iridonephele canus) inhabits the tops of the trees of the
rainforest and almost never descends into the lower levels of the forest. This
is small jackdaw-sized bird. The male has ivory-colored plumage with a grayish
“mane” on the back of the head, the back of the neck and shoulders. The corneous
shell of its beak is snow-white, but there is a red-orange spot in the upper
part of the crest. The feathers on the wings of the male have a characteristic
silvery sheen. The female has a brownish-gray color with separate patches of
brown and dark gray spots on the shoulders and wings. On the main background
of the coloring, it has thin transverse strokes of a darker shade, forming cross
ripples on the plumage of the bird. The corneous covering on its beak is dark
gray. The grayish-white crestbill is one of the main “sentinels” of the forest
from feathered predators. Thanks to life on the tops of the forest, this bird
notices the approach of feathered predators from afar and notifies the forest
with a long series of staccato nasal sounds. This bird does not take part in
mobbing of predators, preferring to hide from them. This species feeds mainly
on insects.
Azure
crestbill (Iridonephele azureus) is named so due to the coloration
of the male, which has blue plumage with iridescent sheen on the wings. There
is a dark blue “mask” on the bird’s head, and the tail is colored ash-gray.
The corneous cover on the male’s beak is grayish-white. The female has dark
gray plumage with black spots on the wings and back; her beak is dirty yellow.
This species is a starling-sized bird. According to many features of behavior,
the azure crestbill is close to the emerald one; it differs in the diet that
includes more plant foods, as well as vocalization similar to coughing and magpie’s
chatter. This species often takespart in mobbing of predators, sometimes together
with the emerald crestbill.
Oculate
crestbill (Macropetalornis ocellifer)
Order: Passerine birds (Passeriformes)
Family: Crestbills (Ctenorhamphidae)
Habitat: Zinj Land, tropical forests.
The family of crestbills, having appeared at Zinj Land, demonstrates the phenomenon
of adaptive radiation, thus reminding the birds of paradise of New Guinea or
Hawaiian honeycreepers from the Hawaiian Islands. Representatives of this family
have developed a variety of life strategies to reduce competition with both
related species and representatives of other groups of birds.
Most crestbills are diurnal birds, which often behave boldly and aggressively.
But among them there is one species that is the complete opposite by behavior
to the small crestbills of paradise – the oculate crestbill. It is a medium-sized
bird, equal to a pigeon in size. One of the main distinguishing features of
this species is a very large, petal-shaped outgrowth on the upper mandible.
Its background color is yellow, and in the middle, a little closer to the front
edge, there is a very large ocular spot – black with a blue border, a brown
middle and a white smear imitating the reflection of light on the iris of the
eye. In general, the outgrowth on the head of the oculate crestbill gives the
impression of a large bird’s head. Such an outgrowth with an eye spot is present
in both the male and the female and is an example of a frightening coloration,
in some ways echoing the
local swallowtail butterfly, mimicking a head of a crestbill.
The color of the bird’s head and upper body is black; the eyes almost do not
stand out in color against the background of the plumage. The wings and tail
of the bird are white. The chest and belly are black, but there are two white
spots on the sides of the chest, visually increasing the size of the folded
wings.
The oculate crestbill feeds mainly on insects and spiders, but also readily
eats snails or nestlings of small birds, if it manages to find them. In its
habits, it is a quiet and harmless bird that spends the day keeping motionless
among the branches. Disturbed during the day, the oculate crestbill tries to
get away from danger and hide as quickly as possible. On its territory, the
bird knows all the shelters perfectly well and most often flies from one shelter
to another during the day. The flight is fast and maneuverable – despite the
contrasting and noticeable color, the bird quickly hides from pursuit among
plants. This species becomes active at dusk and begins to feed actively, finding
the insects due to its keen eyesight and good hearing. In the darkest hours
of the night, the bird falls asleep, but closer to dawn it becomes active again
and feeds until morning, hiding in a shelter only 2 to 3 hours after sunrise.
At night, the bird’s defensive tactics change. Instead of flying away and hiding,
the oculate crestbill fluffs its feathers, opens its tail and lowers its wings
to look larger. If the enemy does not retreat, the bird flaps its wings and
emits a loud hoarse call. Hearing it, other birds of this species repeat the
signal and this is enough to disorient the enemy.
Unlike the small crestbills of paradise, this species is monogamous; male and
female jointly raise offspring. The nest is usually located in a hollow; there
are up to 4 eggs in the clutch, which are incubated by the male and female alternately.
There are only 2 clutches during the year. Incubation lasts about 15 days, young
birds spend up to 2 months in the nest. Leaving the nest, young birds weigh
about a quarter more than their parents. They immediately begin to lead an independent
life and their parents do not feed them any more. In young birds, the plumage
has a camouflage color, but there are already ocular spots on the crest, characteristic
for this species. Sexual maturity occurs at the age of 8 months, and life expectancy
reaches 10 years.
Sawbeak
fisher (Serratorostrum piscatoius)
Order: Passerine birds (Passeriformes)
Family: Sawbeaks (Serratorostridae)
Habitat: Northern Asia, Beringia. Bird lives at riverbanks.
Mass extinction at the boundary of Holocene and Neocene, figuratively speaking,
“had spoilt game”, having changed parity of forces in biosphere. Species from
earlier prospering and various taxons appeared completely destroyed, but species
from groups insignificant earlier received unexpected chance of further development.
At times representatives of any group had opportunity to occupy the ecological
niche not peculiar to this group earlier. Warblers used this way the chance
of survival: some representatives of this suborder in large and variable order
of passerine birds have occupied the “business” completely unusual for these
birds in the past. Sawbeak fisher, eating fish, began one of such “deviated”
species.
Sawbeak fisher is rather large representative of warblers: it is duck-sized
bird. This species is the descendant of corvine birds (Corvidae), known by their
ecological plasticity and advanced intelligence. It is similar to the halcyon
of huge size: at this bird there are short body, large head, long sharp beak,
short wings and tail. At edges of the end of beak of bird jags are developed:
this is the adaptation for keeping of slippery catch – fishes. And for more
reliable capture of catch on the end of tongue the horn oblique back teeth also
are developed. Toes on legs of bird are edged by skinny fringe which can fold
at forward movement of leg and fall out when bird pushes by paw. Sawbeak fisher
can dive and chase catch under water, making in chase of it dizzy turns. Under
water body of bird looks silvery: At the sawbeak coccygeal gland, producing
plentiful fat greasing which has water-repellent properties, is advanced. This
bird spends a lot of time, cleaning feathering and greasing it with fat.
The body of sawbeak is colored rather brightly: back, nape and wings are grey
with shade; sides of head, throat, chest, stomach and tail are colored blue
with metal shine. Such colouring makes dived bird rather imperceptible at look
from above and from below. At each side of head from eyes two “tresses” of lengthened
feathers direct back: at males they are red, at females grey. Beak is colored
yellowish-grey, and legs are dark grey. Wings of bird are short but strong:
the sawbeak fisher is the bird of passage, each year making long migrations.
The sawbeak fisher catches catch from any support: to look for underwater inhabitants
it chooses branch of tree growing horizontally above water. This bird eats fish
and crayfishes, chasing them safely diving in water. During swimming bird extends
body almost in straight line, presses wings to body (using them for turns),
and swims rowing by paws. It kills prey by impact of beak.
Sawbeak fisher is monodin; pairs at these birds form to one season. Male occupies
site of the river with riverside thickets of trees, builds the basis for nest
and starts to involve females to the territory with the help of abrupt cries
similar to crow croak. Female chooses site, views it, and only after that starts
acquaintance to “groom”. Acquaintance is accompanied by bows, original “dance”
of the male in air above the female, and an exchange of “gifts” – pieces of
bark, twigs or feathers. If birds have liked each other, they complete nest
together. The generated pair hunts together; birds make to each other signs
of attention: clean feathers to each other, treat with small fish and cry a
duet, declaring about rights to territory.
This species builds bowl-like nest of thin rods on tree near to water. Trees
growing on small islands, where ground predators do not reach, are especially
preferable at these birds. In clutch there are two large eggs, female hatches
basically, male substitutes her to short time, but basically only feeds female.
The incubating lasts about 4 weeks. Nestlings hatch blind, but covered by down.
They begin to see at the age of 1 week, in same time they start to cover with
feathers. Both parents take part in feeding of posterity: nestlings are very
gluttonous. At six-week age they abandon nest and start studying to swim and
hunt fishes and frogs. At one-year-old age young birds become sexual matured.
Sawbeak fisher is the bird of passage. For winter it flies away to southern
rivers and Pacific coast to islands of Kuril - Kamchatka zone, catching there
fishes in coastal sea waters.
Forest
ronin (Roninornis mortifer)
Order: Passerine birds (Passeriformes)
Family: Tits and Chickadees (Paridae)
Habitat: subtropical forests of Japan and southern part of Big Kurils.
In Holocene some species of titmouses have got the advantage, becoming synanthropic
birds. Were widely settled, these birds began development of new food sources.
Extinction of many species of predatory birds had caused occurrence of carnivorous
species among various groups of passerine birds. Large carnivorous butcherbirds,
characteristic for tropical and subtropical areas of Old World, had appeared
so. Titmouses, despite of pretty appearance, also differed in predating bents
in human epoch (this feature of behaviour was especially brightly shown at the
keeping of these birds in captivity). In Neocene this tendency had proceeded
and had to found the embodiment.
One species of titmouses developed predating bents lives in forests of Japan
Islands. For aggressive character the bird has received the name “forest ronin”
(ronin is the masterless wandering Samurai). It is one of the largest species
in titmouses family: by size it is larger, rather than starling. Beak of forest
ronin is of “universal” type – it is straight with pointed tip. At the bird
predating bents are strongly expressed – it attacks various small vertebrate
animals – rodents, lizards and birds, and kills them by strong impact of beak
in head. Forest ronin also eats large insects and snails, and birds living near
the rivers, catch small crabs creeping on land. This bird pecks catch, keeping
it by paw. Forest ronin eats large catch on the ground, and carries away small
prey in beak to the tree, fixes it in forked branches, and then pecks.
Forest ronin prospers in forests of Japan, and successfully competes with small
corvine birds due to features of behaviour. It is clever, courageous and curious
bird, able to procure various kinds of food. Forest ronin notices, where other
birds may nest, and willingly ravages their nests.
Appearance of forest ronin shows that this bird is typical forest inhabitant.
At it there are short wings with rounded tips, wide long tail and prompt maneuverable
flight. Forest ronin is able to turn sharply in air and quickly flies even among
dense branches. In branches and on the ground this bird moves by jumps.
Colouring of body of this bird is cryptic: top of body is grey with brown spots.
Back of bird is white, and stomach is emerald-green. The head of forest ronin
sharply differs in color from the body: it is black, and on nape there is a
site of red color feathers. This feature of colouring has the important value
in life of bird: it is the pacifying signal, which is vital for coexistence
of aggressive birds. Demonstration of such spot is the sign of submission, and
it quickly suppresses aggressive behaviour of the congener. Bird submitting
to the winner rises on legs, inclines the body forward, lowers head and shows
this spot. Usually young birds are colored dimmer, rather than adults. But forest
ronin represents one of few exceptions. At young birds colouring is much brighter,
than at adults – the red spot is larger; it makes up to about third of all surface
of head.
As a rule, carnivores are territorial species, and forest ronin is not exception
here. The male of this species occupies rather large site of forest – two males
do not live closer, than for hundred meters from each other. In courtship season
male loudly and sonorously sings, notifying on claims for territory. Its song
sounds monotonously, and represents a series of whistles, ending by long rolling
warble.
The male maintains two females at the site and simultaneously looks after two
hatches, feeding up nestlings. In relation to other males it behaves very aggressively,
actively expelling them from the territory. Usually in the beginning of the
territorial conflict competitor males loudly sing in a pointed manner, perching
in field of view of each other. This way they estimate forces of each other,
observing for the opponent. Usually “duel of singers” is enough for the newcomer
male to leave from another's territory. If it does not happen, severe fight
follows. During the fight males seize each other’s feathers by beak and paws,
and also beat the contender by wings. If the duel is especially severe, males
may even fall from branch to the ground and continue fighting in grass. At this
moment they are so keen on fight, that permit to come to themselves very close.
In courtship season one of them or even both ones easily may fall prey to various
ground predators.
If the female flies to the site protecting by male, the male tries to stop her
at his territory. Courting for the female, male pursues her, flies forward,
bends head down, showing the absence of aggression. At this time red spot on
his nape is especially appreciable. If the female does not fly out, the male
continues its demonstrations. In courtship displaying the male perches on tree
in front of the female, inclines the body forward strongly, and demonstrates
itself to her. Thus he stretches wings and quivers by them. White feathering
on its back becomes well seen, and male fluffs it to exaggerate itself visually.
As the result there is spectacularly looking combination of spots of three colors
– white back, red spot on nape and black forward part of bird’s head.
The nest of forest ronin is arranged in tree-trunk hollow. The female lays 3
– 5 eggs on litter of wood dust. Usually it feeds nestlings by its own, but
male helps her, delivering to nestlings about third of forage eaten by them.
Nestlings leave nest at the age of six weeks. The female finishes feeding them
till about one week, and then young birds become independent. For one year forest
ronin can rear up to two hatches.
Young birds become able to breeding at the age of 8 – 9 months. Life expectancy
of this bird makes about 6 – 7 years. More often males live much less, than
females – high intraspecific aggression of this bird expresses so. The same
circumstance constrains the number of population, regulating the density of
settlement of these birds.
Kuday
(Neopanurus kudai)
Order: Passerine birds (Passeriformes)
Family: Bearded reedlings (Panuridae)
Habitat: Altai, mountain valleys, thickets of marshland vegetation along the
riverbanks and lake shores.
Mainly small species of living creatures with a short life cycle and a wide
range managed to survive the human era successfully. Their range could be reduced
or even fragmented, but the number of the species could recover quickly and
remained high enough for survival. The bearded reedling (Panurus biarmicus)
of Holocene epoch managed to survive anthropogenic pressure and the ice age
in the Asian part of the once vast area, and to give rise to several new species
after the human epoch. One of them is kuday (the name of the supreme god in
the mythology of the Altaians) widespread in Altai.
This bird species is larger than its ancestor, measuring 28-30 cm; the tail
is very long – up to 15 cm. The wingspan is about 40 cm. There is no difference
in size between the female and the male.
The color of plumage of the body is light, brownish-yellow. Legs are long, coal-black,
with thin tenacious claws. Sexual dimorphism is expressed in the coloration.
Male has a white head with a dark gray forehead. Long black “moustaches” of
feathers stretch from beige eyes and almost to the wings. Under the wings of
males there are large red spots, noticeable in flight; the tail is ochre-red.
Females are more modestly colored: the body is light ochre; the head is bluish
from below. Beak of birds of both sexes is short and yellow; a captured bird
inflicts severe beak bites on a predator. Kuday’s advertisement call consists
of a sharp “pfish”, a long, rolling “chigirr” and a quiet “bett”, repeated sequentially.
Kuday lives near reservoirs, especially preferring thickets of marshland vegetation
in floodplains of rivers in mountain valleys. In summer this bird species feeds
on insects, spiders, other invertebrates and small mammals. In winter, its diet
consists of plant seeds.
These birds are monogamous, they nest twice a year (in April and in early July).
During the courtship display, males try to show all the beauty of their plumage
by spreading their tails, raising their wings, rotating their heads and spreading
their feathery “whiskers”. The mating song of the species is a murmuring trill
“pshik-jiv-chgerrre”. When the breeding pair has formed, the female builds a
cup-shaped nest of grass, attaching it to a reed near the water, and lays 2-3
small eggs, white with small spots. Both partners replace each other on the
nest, incubation lasts about 2 weeks. The nestlings are covered with white down.
They grow in the nest for 5 weeks, and their parents feed them for about 1 more
week.
Sexual maturity comes after wintering, life expectancy is about 12 years.
This bird species was discovered by Feldwebel, the forum member.
Three-Rivers-Land
reed warbler (Acrocephalus tripotama)
Order: Passerine birds (Passeriformes)
Family: Reed warblers (Acrocephalidae)
Habitat: thickets of reeds and cane in Three-Rivers-Land.
After the end of Holocene anthropogenic crisis of the biosphere, many small
birds got a chance to spread and further evolve. Among others, reed warblers,
characteristic songbirds of the Old World, also took advantage of this chance.
Their numbers in Eurasia declined during the ice age, but after its end, reed
warblers became as diverse as in the previous era. The extinct local species
have been replaced by the descendants of the surviving species, and many of
these birds are common species in Eurasia. One of them is Three-Rivers-Land
reed warbler, which remained at the place of appearance of its species and changed
along with changes in its habitats.
This species is a descendant of the Eurasian reed warbler (Acrocephalus scirpaceus)
of Holocene epoch. The appearance of this bird is similar to the appearance
of the ancestor in many ways: the length of the body including the tail is 20-21
cm, the wingspan is 19-23 cm, and the weight is 35-40 g. Plumage is nondescript
brown with darker specks on the back and wings; the chest and abdomen are slightly
lighter, yellowish-gray, the primary feathers are darker. Beak is pointed, elongated
and yellow in color. Legs are elongated, with tenacious toes.
Three-Rivers-Land reed warbler feeds on insects, spiders and mollusks inhabiting
the reed beds, occasionally eats frogs and mice cubs, pecks bird eggs. The bird
is mobile, climbs very deftly on large stalks of reeds and papyrus
cattail (Typha tetramarina). It is a solitary bird, denoting its rights
to an area of thickets with sharp, squeaky trills. Sometimes it imitates the
voices of other birds, alternating them with its own creaking song fragments.
This species is migratory, spending the winter at the southern shores of Fourseas,
along the shores of the Balkans, and even in the “oases” of the Mediterranean
lowland. The return to the nesting sites occurs by the beginning of April, when
the marsh plants have already formed a significant above-water mass of greenery.
The nesting period lasts from the end of April to the first half of June. Males
build nests of grassy stems in particularly dense thickets. The nest is cup-shaped
or conical in shape and is fixed above the water between three or four reed
stalks or two or three cattail stalks. Often the nest is arranged in the leaf
axil of giant papyrus cattail – the leaf and stem are intertwined with each
other with grass fibers. Birds nest in places inaccessible to terrestrial predators
– in the thicket at a safe distance from the shore.
Males sing, climbing to the tops of the stems above the nest. Sometimes short-term
fights break out between them, but usually they arrange only singing “duels”,
keeping on their territories and performing virtuoso trills with the inclusion
of “outsong” fragments of songs of other birds. Often young birds “outsing”
imitations of songs performed by more experienced relatives. Females choose
singers not only for the quality of singing, but also for the location and quality
of the nest. There are usually 3 to 5 bluish-white eggs in the clutch, which
are incubated alternately by both partners for 12 days. The offspring develops
quickly, and at the age of 14-16 days, the juveniles fly out of the nest, but
they are fed by their parents for another week. The birds are ready to breed
already the next year. Life expectancy is up to 8 years.
This bird species was discovered by Nick, the forum member.
Red-winged
swallow (Petrochelidon pseudohirundo)
Order: Passerine birds (Passeriformes)
Family: Swallows (Hirundinidae)
Habitat: mountain forests of North America.

Picture by Pavel Volkov
The change of natural conditions during the transition from
Holocene to Neocene had a relatively small impact on the species diversity of
swallows. Like swifts, these birds have evolved towards specialization towards
long high-speed flight. They were able to fly long distances, which helped them
survive at the beginning of Neocene, when the boundaries of the natural zones
of the Earth changed significantly. Species with a wide range, capable of making
extended migrations, were able to survive.
A significant number of swallow species disappeared during the era of the biological
crisis – mainly local species from tropical forests. Other species migrated
from their former habitats to more favorable conditions: for example, in Neocene,
the descendants of the Holocene barn swallow are found mainly in tropical regions
– in Africa, in southern Asia, in Meganesia. On other continents there are descendants
of other species of swallows: for example, in North America there is a descendant
of the American cliff swallow (Petrochelidon pyrrhonota) – a red-winged swallow.
Red-winged swallow looks like a barn swallow that disappeared from North America,
but its tail is not forked, but fan-shaped with a straight rear edge. Its back
is black and blue, its chest and belly are white, there is a white mask on its
face, the underside of the wings is dark red, of the rust color (hence the name).
this species is a bird of miniature size – body length up to 110 mm, wingspan
- 240 mm.
This species is a flocking bird, nesting in numerous colonies. Many species
of swallows formed colonies as early as the Holocene, and in the Neocene this
trend continued, although not in all species. Red-winged swallow prefers to
nest in mountain forests throughout North America. In the center of this continent,
in the Neocene, an extended strip of prairies and steppes appeared from north
to south, which is an obstacle to the settlement of many species of birds, especially
of small forest ones, but various species of swallows fly freely over it, so
North American swallows have a wide range and there is no division of species
into subspecies on a geographical basis.
A mandatory requirement for the formation of colonies at this swallow species
is the presence of permanent reservoirs with running water and clay banks: they
make their nests out of clay, attach them to rocks or high on trees, and line
them with feathers and grass. The nest of these birds has preserved a shape
characteristic for the ancestral species: a hemispherical nesting chamber with
a long corridor to the entrance. But in this species, the entrance to the nest
is facing down, and the corridor is stretched along the outer wall of the nest
from the side, often attaching to the neighboring nest. The end of the entrance
hangs down like a tube. This swallow species very often builds a new nest every
year, leaving the old nests to other bird species, so swallows of other species,
swifts and small songbirds often nest in colonies of the red-winged swallow.
The mating season falls on the second half of spring: males display to females
a special courtship flight, and if the females like it, they join the males,
and for some time they perform flight figures synchronously. After the formation
of a breeding pair, intensive construction of a nest of clay with the addition
of animal manure and grass fibers begins. In the nest, a pair of birds makes
a clutch of 3 to 8 eggs (usually 5-6) of white or pinkish color with brown specks
on the shell. Both parents incubate them for about 2 weeks, in turn. This species
is monogamous, but pairs are formed only for one breeding season. In the northern
parts of the range, birds make one clutch per year, and two in the southern
part. Males of this species often mate with females from neighboring pairs,
and females sometimes lay their eggs in the nests of relatives, so 1-2 chicks
from the brood may be the offspring of another pair of birds or have a different
father. 3-4 weeks after hatching, the juveniles take wing and leave their nest
forever. The next year they become capable of reproduction.
Red-winged swallow feeds on insects, less often on spiders, which it catches
on the fly. Sometimes this species eats berries (especially in autumn, before
flying south). Its enemies are various birds of prey, especially ethereal kites,
grabbing them on the fly, and various arboreal mammals sometimes ravage her
nests, although usually these birds arrange nests in inaccessible places. These
birds often suffer from parasites: ticks and blood-sucking dipterans attacking
nestlings.
Red-winged swallow can live up to 10 years, but more often it dies much earlier
from predators, diseases and lack of food.
This bird species was discovered by Bhut, the forum member.
Peregrine
swallow (Areniriparia peregrina)
Order: Passerine birds (Passeriformes)
Family: Swallows (Hirundinidae)
Habitat: Northern and North-Eastern Europe, riverbanks.

Picture by Pavel Volkov
Some species of swallows were among the birds got a considerable
benefit due to human activity. They settled in human buildings and human-modified
landscapes. One of these species had been sand martin (Riparia riparia) settled
in riverine habitats. Having kept rather numerous population and wide range,
this species survived in human epoch and in early Neocene it became an ancestor
of some new genera and species of Northern hemisphere. One of them is peregrine
swallow endemic to the European riverbanks. Because of their long migration
to winter in the marshes of Southern Asia, they are named peregrine swallows.
These are numerous migrating birds, very common in river walleys. They are small,
with general body length of 13 cm, but their outer tail feathers 5 cm long make
them look bigger. Wings of these fast and dexterous flyers are narrow and elongated.
Overall appearance of peregrine swallows is typical for their family. The chest
of the peregrine swallow is white, other parts of the body are blue-glossed
black; bill is short, wide and black, and the legs are brown. Their legs are
short but strong enough to dig the sand where they make their nests.
These birds feed on small insects they catch in air. Also they eat young spiders
settling on spiderweb threads by air. Being a social species, they form flocks
that may number thousands of individuals.
Both sexes have forked tails with long outer tail feathers (similar to that
of the wire-tailed swallow (Hirundo smithii) of human era); in the males these
long feathers are racquet-shaped, having rounded lobes of feather vanes at tips.
Males use these feathers in the courtship flight: the movements they perform
during the flight cause wind to shake these feathers, and the female will fly
next to the male she chose. Their breeding season starts in the end of spring.
Nesting takes place in colonies which may number from a dozen to hundred pairs;
their nests are excavated by breeding couples of birds in steep sand banks near
rivers and lakes. Despite of tiny size, birds dig long nesting holes – sometimes
the length of tunnel is about 100 cm, but usually it is about 50-60 cm. The
real nest is a litter of straw and feathers in a chamber at the end of the burrow.
Female lays four or five eggs and incubates the clutch for 14 days.
The hatchlings are fed with semi-digested insects, regurgitated by both parents.
They fledge at the age of one month, having the same coloration, as their parents,
but lack long outer tail feathers. Juveniles leave the nest at this age, but
keep with the parents until the end of summer, when migration begins.
Sexual maturity is reached within 7 months, when they develop their characteristic
outer tail feathers. Lifespan is about 8 years.
This bird was discovered by João Vitor Coutinho, Brazil.
Red-headed
bank swallow (Nigroriparia erythrocephala)
Order: Passerine birds (Passeriformes)
Family: Swallows (Hirundinidae)
Habitat: riverbanks of northern part of North America, migrating in the winter
to South America.

Picture by João Vitor Coutinho
For first millions of years of post-human epoch the burst
of active speciation among survived species had been characteristic. Human epoch
was the time of drastic decrease of species diversity. And the stabilization
of natural conditions promoted the increase of biological diversity leading
to stability of ecosystems.
Some species of swallows survived in human epoch, and sand martin (Riparia riparia)
among them. From it some new genera and species descended in Neocene. One of
them is red-headed bank swallow from North America – numerous and common species
of migrating passerines of the continent.
This is a small bird; its total length is only 12 cm. Its plumage is blue-glossed
black, with the exception of the head, which is brownish-red outside the breeding
season and bright red during the nesting season. Its bill and legs are black;
this bird has forked tail characteristic for swallows. With their strong legs,
these birds are capable of digging their nesting burrows in sandy soil of steep
riverbanks.
Red-headed bank swallow is exclusively insectivorous bird, consuming mosquitoes,
midges and other insects living in riverine habitats. Sometimes these brave
birds attack even large horseflies, but try to tear off their unpalatable head
and thorax in flight, swallowing only soft abdomen. They also catch plenty of
casual prey when flying above forests during their migration.
Highly sociable, they migrate in groups of hundreds of individuals, sometimes
uniting with other fast-flying birds like other species of swallows and swifts.
This species spends winter in tropical areas of South America. During the migration
these birds move along the river walleys to the shore of Gulf of Mexico. Then
they fly along the coast of Central America and cross the Panama Strait. In
wintering places they keep near river channels. The returning to nesting areas
coincides with the metamorphosis of blood-sucking dipterans making the base
of their ration.
They are also social in their nesting habits, with many pairs breeding close
together, according to avaiable space. Sometimes it may be up to 20 burrows
of these birds to one square meter of steep riverbank slope. After the wintering
young birds return to parental colony, but can easily move to another colony
or establish new one, having found a suitable place.
During the breeding season starting in late spring, male vocalizes while flying
very high, and then descends rapidly in spirals. This behavior is repeated until
a female joins it in the flight. Then, the couple digs a tunnel about half meter
long in sand banks in rivers or lakes. The nest litter is made of straw and
feather in a chamber in the end of the tunnel. The female lays two to four white
eggs that are incubated by both parents for 14-15 days.
The hatchlings are fed with semi-digested insects, regurgitated by both parents.
They fledge at the age of four weeks and leave the nest at this age. In juvenile
feathering their body feathers are dark grey and their heads are brown. They
become completely independent one week later and keep in partental colony until
the migration to the south. Young birds returning from the wintering are sexually
mature and have the coloration of adult birds. Their lifespan is about 7 years.
This bird was discovered by João Vitor Coutinho, Brazil.
Cactus
tree swallow (Neohirundo africana)
Order: Passerine birds (Passeriformes)
Family: Swallows (Hirundinidae)
Habitat: groves of baobab opuntia and trees in North Africa.
The barn swallow (Hirundo rustica) of Holocene epoch was one of the common synanthropic
bird species. This species no longer exists in Neocene, but it managed to become
the ancestor of a number of descendant species. Some of them live in North America
– for example, the red-winged swallow,
and others inhabit Africa. One of these species is a cactus tree swallow, which
has chosen giant tree-like cacti, baobab
opuntias, for life.
This swallow species has retained many recognizable features of the ancestral
species – pointed wings, short beak and characteristic two-tone body coloration.
This bird has a dark blue back with a metallic sheen and straw or pale rusty
belly and throat. This bird is a good and maneuverable flier, and can outrun
almost any other bird or bat in the air.
Like other swallows, it feeds on insects – usually the bird eats small flying
insects, but, unlike other swallows, it is able to pick them up from the surface
of the trunks and branches of baobab opuntia. It has a shorter tail than the
ancestral species, and there is no “fork” formed of the elongated outermost
tail feathers. It allows the cactus tree swallow to move easily along the trunks
of tree-like cacti. There they also find a drink – birds can drink cactus sap
in holes made in the trunks of baobab opuntias with the beaks of stiff-tailed
parrots. The sap of these cacti does not completely replace water, but cactus
tree swallows may leave opuntia groves and fly to rivers, lakes and other reservoirs
to drink water. While flying to the watering hole, they are attacked by their
main enemies – local birds of prey; a healthy swallow of this species is usually
able to fly away from many of its enemies.
These swallows also make nests of coastal mud mixed with fluffy plant seeds,
attach them to the branches of baobab opuntia and line them with grass and their
own feathers. There are 4 to 8 eggs in a clutch, white or pinkish with brown
specks. Both birds of the pair alternately incubate them for 2-3 weeks, and
the juveniles leave the nest after about the same time, but continue to stay
close to their parents, who finish feeding them. After another two weeks, the
young birds leave their parents, and the families break up. These birds do not
have a pronounced mating period, but successful nesting partners usually remember
each other and the pair reunites for the next nesting cycle. During the year,
the couple is able to make 2 clutches.
Sexual maturity occurs at the age of six months, life expectancy is up to 8
years.
This bird species was discovered by Bhut, the forum member.
Morning
honeybird (Melisugornis aurorae)
Order: Passerine birds (Passeriformes)
Family: Hawaiian honeybirds (Melisugornitidae)
Habitat: Hawaii, rainforests.

Picture by Alexander Smyslov
Before the human colonization of Hawaiian Islands their fauna
was represented mainly by birds and insects. Among the Hawaiian birds there
was a separate family of Hawaiian honeycreepers (Drepanididae) which had a great
importance for flora of islands. Various species of these birds had been connected
by the symbiotic relations with plants of islands: many species of local trees
had been pollinated by these birds, and often exclusively by them. Therefore
extinction of Hawaiian honeycreepers led to the disappearance of a number of
species of plants.
Flora of Hawaiian Islands of Neocene is represented by descendants of species
introduced in the past, and isn’t successive in relation to primary flora existed
here before people. But separate tendentions in nature tend to repeat, and in
new Hawaiian flora also symbiotic relations of some species of plants with birds
appeared. The place of the Hawaiian honeycreepers that had completely disappeared
by Neocene, had been occupied by another family of birds descended from white-eyes
(Zosterops) introduced to Hawaii. They showed such inclinations in type of feeding
during human era, and in Neocene this feature developed to the condition of
deep specialization.
Hawaiian honeybirds represent the clearly formed family of nectar-eating birds.
They live in forests of various types – from mountain to lowland humid tropical
ones. The only limiting factor for them is the permanent existence of blossoming
plants.
For Hawaiian honeybirds rather small body size is characteristic: the largest
species is starling-sized, and others are even smaller. All species have rather
bright coloring including various shades of yellow and green colors, supplemented
with metal shine and separate bright spots. One more characteristic feature
of family is rings of naked white skin around eyes, the heritage from the ancestor.
Wings of all honeybirds are peaked, and tails are wide. At some species tail
feathers have the elongated locks of feathers. Flight of these birds is fast
and maneuverable. Honeybirds of larger species during the feeding perch on branches,
and small species simply hover around the flower like hummingbird. Usually each
species of honey birds is connected with a certain set of species of plants,
feeding on them and at the same time making pollination.
Voices of all species of honeybirds represent more or less long ringing trill.
Different species prefer to sing at different times of the day – it helps to
avoid mistakes at recognition of congeners.
At all species of honeybirds the nest represents a small “basket” skillfully
weaved of plant fibers and hanged on thinnest twigs. Such feature is connected
with the appearing in forests of Hawaiian Islands of Neocene epoch of numerous
mammals, descendants of the species introduced by people, which can ravage nests
of birds. In clutch there are usually 4-5 eggs (at large species 3-4 ones) with
dark egg shell. Incubation lasts about 10 days. Chicks develop quickly: in three
weeks they grow, fledge and leave the nest. In a year the egg layings may repeat
three times (at small species up to 4 times). Young birds are able to nest already
at six-month-old age.
Morning honeybird lives in rainforests of lowlands of islands. It is a medium-sized
species: it is sparrow-sized if not including a long beak. It is colored bright
yellow, only on a back there is a site of extended feathers of green color.
During courtship season males of this species display it to females, raising
back feathers vertically. In addition over areas of featherless skin around
eyes this species has “eyebrows” of black feathers, allowing recognizing the
congeners visually.
Beak of this bird has white color and is rather long: it exceeds the head length.
Birds feed on nectar of tubular flowers, including species of treelike datura.
Usually they perch on branch of fodder plant, but if necessary they are able
to suck nectar in flight. Also these birds eat small insects, pecking them from
branches or out of flowers.
This species is named so because it sings earlier, rather than all other species:
since the earliest morning to sunrise. Voice of these birds sounds like long
buzzing trill alternated with separate ringing calls.
Other species of honey birds live in forests of Hawaiian islands:
Mountain
honeybird (Melisugornis grevilleophila) is the largest, starling-sized
species of this genus. This kind is feeding mainly on Grevillea (“silky-oak”)
forming “silver forests” in mountains of islands. It is a solitary bird with
greyish-green feathering which is remarkable in expressed metal shine. On waist
of this species of birds there is bright orange-yellow spot imperceptible when
bird folds wings. Beak is long (exceeding head length), crescent, adapted to
feeding on plants. The present bird eats mainly nectar, occasionally seizing
small invertebrates. This bird sings from sunrise, but for rather short time:
in total for about two hours. Its voice is rattling calls.
In marshlands, in underbrush of rainforests butterfly
honeybird (Melisugornis papilio) lives. It is very tiny bird: its total
length is about 10 cm including beak. It is feeding on flowers of forest-dwelling
grassy plants. It can be observed especially frequently in thickets of fragile
balsam bush – grassy plant growing in shadow of large grasses and trees near
to rivers. This species has dark green top of body, black primarily feathers
and pale yellow belly. Voice of this bird sounds like shrill squeak.
Short-beaked,
or spectacled honeybird (Melisugornis brevirostris) lives in
rainforests. It differs from other species of this genus having short beak:
no longer than half of head length. It takes place due to features of its diet
– bird eats nectar of widely opened flowers: passionflowers and Asteraceae.
This bird is larger a little than sparrow. It has rather short wings, and it
prefers to move on fodder plants “on foot”, occasionally flying from one branch
to another. This bird has grassy-green back and tail, wings of the same color
with dark spots on bends, and primarily feathers with the expressed metal shine.
The bottom part of body, breast and throat are bright yellow. White sites of
skin around of its eyes are bordered by small black feathers, hence the second
name of bird. This species sings late: from dawn till midday; song represents
prolonged murmuring trill.
Translated by Alexander Smyslov.
Crab-cleaning
rattlebird (Carcinornis crepitaculum)
Order: Passerine birds (Passeriformes)
Family: Nuthatches (Sittidae)
Habitat: South-Eastern Asia, Jakarta Coast, Sunda Land and nearby islands.

Picture by Simon
Background by Alexander Smyslov
During the process of evolution representatives of group
of passerine birds had occupied set of ecological niches. Among them it is
possible to find omnivorous
and herbivorous birds, quick hunters for insects and after mass extinction
on the boundary of epochs even predators. Some of these birds have concluded
mutually advantageous “union” with plants, becoming their pollinators. And
in tropics of South-Eastern Asia one of birds became the constant satellite
of animals, clearing them from parasites. Its presence can be found out from
afar by characteristic voice – abrupt bitter cries. This small bird (length
of body is only 6 – 7 cm including long beak) assiduously gathers flies and
mites from local animal – turtles and large crabs
“mangrove robbers” (Archocancer mangrophilus). For these features the
bird is named crab-cleaning rattlebird.
At it there are short wide trunk, rounded wings, big head and sharp beak.
The tail of this bird is also short – it is hardly seen from under wings.
But paws
of crab-cleaning rattlebird are very tenacious with hooked claws.
The bird is colored from above light green; bottom of body is pale yellow.
Beak and legs are black, and from corner of beak through an eye the narrow
black strip passes, ending near the ear aperture. At males this strip is
wider than at females.
The crab-cleaning rattlebird differs by mobility and characteristic fussy
movements. Having perched on the body of crab or turtle, it does not stop
even for a second.
The bird moves on the body of “patient” by characteristic short jumps, examining
plicas of skin or joints of shell, and taking from them catch – parasitic
two-winged flies and their larvae. Sometimes the bird simply traps them
sitting on the body of “patient” and catches them by sharp throw. Sometimes,
when giant “mangrove
robber” crabs bear posterity, this bird can harm to them, pecking part of
young crabs.
Crab-cleaning rattlebirds nest in pairs in tree-trunk hollows. Sometimes
they close up edges of tree-trunk hollow with clay in characteristic for
nuthatches
manner, reserving only small entrance aperture. In clutch there are 5 – 6
eggs; both parents hatch it. The incubating lasts about 10 days. Nestlings
hatch
naked and blind, but develop quickly. At the age of one month they already
leave nest. For one year these birds can rear 3 – 4 hatches.
This species was discovered by Arseny Zolotnikov.
Stovemaker nuthatch (Sitta furnaria)
Order: Passerine birds (Passeriformes)
Family: Nuthatches (Sittidae)
Habitat: humid forests at the southeastern coast of Fourseas and in Balkan.
Picture by Simon
For European nuthatch (Sitta europaea) widespread in human
epoch in forests of Europe, the special form of nesting behaviour was characteristic.
These birds nested in hollows and covered their entrance apertures with dirt
and clay, leaving the small aperture permitting to owners of nest to penetrate
inside hardly. One of Neocene descendants of European nuthatcher has deepened
this interesting feature of ancestor. It is named stovemaker nuthatch.
The shape of this bird is typical for the majority of nuthatches. But nevertheless
stovemaker nuthatches surpass the ancestors in size in great degree and are
among the largest representatives of family in Neocene. Birds of this species
reach the size of great spotted woodpecker: the length of their body reaches
25 cm, and weight is about 100 grammes. Plumage of stovemaker nuthatches
is rather bright. The top of their body is colored dark grey (at separate
individuals
it is even black), belly and breast are ochre-red, and “face” is white. Beak
is bright red; paws are colored black. The sexual dimorphism in colouring
of these birds is almost not expressed.
Stovemaker nuthatches eat mainly insects and spiders, finding them on tree
trunks. The large size gives them an opportunity to eat also small vertebrates.
Vegetative food – mainly fruits, nuts and acorns – has also an essential
role in a diet of these birds. In searches of food stovemaker nuthatch dexterously
moves on branches and trunks of trees. In “acrobatic abilities” this species
does not concede to ancestor and is capable to creep on trunks turning head
downwards equally dexterously. These birds go down to the ground reluctantly,
but sometimes also search for food there.
Stovemaker nuthatches utter a plenty of sounds, basically whistles and short
snuffling calls. In the beginning of nesting season male utters especially
loud cries, calling the female.
The most interesting feature of this species is its nesting behaviour. At
nest building stovemaker nuthatches behave similarly to the majority of hornbills
(Bucerotidae) of human epochs: male immures the female in hollow for the
time
of egg incubation and nestlings feeding, leaving only tiny aperture. Through
it male feeds the female and nestlings, bringing them food in beak. Such
tactics provides protection against the majority of predators (except for
snakes and large local woodpeckers).
Though stovemaker nuthatches are capable to make a hollow of the necessary
size in soft or rotten wood independently, they use more often natural hollows
or ones made by woodpeckers, quite often expelling former owners therefrom.
The bottom of hollow is covered with a layer of litter of thin slices of
bark and dry leaves. Male immures the female, using dirt and clay for this
purpose.
The female is “convicted” within approximately 1.5 months – at this time
nestlings hatch from eggs, fledge and become independent. Usually it may
be up to 5 –
7 nestlings in hatch; their number is less, than at an ancestor of this species,
Eurasian nuthatch. It is connected to higher survival rate of posterity.
Besides male simply can not feed the greater number of nestlings. At the
bottom of
nest a plenty of larvae of various insects constantly lives; they eat waste
products of birds’ vital activity – dung, shed feathers and the rests of
food. Usually in nest of these nuthatches flies settle, but occasionally
beetles
live. Usually grubs pass metamorphosises already after the hatch leaves nest.
Stovemaker nuthatches are monogamous birds, and after nestlings rearing breeding
couples do not break up. Beyond the nesting period birds live at the site
of forest which is protected from the relatives breaking its borders. These
birds
live up to 15 years.
This species of birds was discovered by Simon, the forum member.
Common
melodious wren, or Common eastern uirapuru (Cambaxirra canora)
Order: Passerine birds (Passeriformes)
Family: Wrens (Troglodytidae)
Habitat: tropical areas of the eastern coast of South America.
The human actions during the Holocene caused the decline of many species. However
some species survived and in the Neocene they gave rise to new species. One
of these new species is the common melodious wren, a descendant of the birds
of the Genus Troglodytes. These birds are also the type species of the Genus
Cambaxirra, the Eastern uirapurus. While cambaxirra was a common brazilian
name for Troglodytes birds, the word uirapuru was a name used by the
Tupi-guarani people to name some species of passerines, more famous used to
name birds of the Genus Cyphorhinus, which descendants in the Neocene are endemic
to the Amazon rainforest as members of the genus Irapuru.
The common melodious wren is endemic to the Atlantic rainforest of Southeastern
and Northeastern South America.
The common melodious wren is a small bird, the adults are 13 to 15 cm long with
a wingspan of 17 cm. Their plumage is brown, being darker on the upperparts
and lighter on the throat and chest. They have blackish barring to the wings,
tail and to the flanks. The bill is thin with a blackish upper mandible, and
a black-tipped pale grey lower mandible. The legs are pinkish or grey. The short
tail has a white tip and is typically held cocked.
These birds forage actively in the lower areas of the forest vegetation, never
going to the canopy. They mainly eat insects such as butterfly larvae, also
spiders and snails. The common melodious wren rarely attends mixed-species feeding
flocks.
The common melodious wren doesn't form flocks, being solitary or living in pairs.
These birds are monogamous, with the bonded pair being aggressive to members
of their own species. Such aggressiveness increases during the nesting season.
Their courtship starts during rainy summer months, with the male singing an
elaborated and agradable song to attract the female. Usually, he does it in
a branch close to the ground. The female will land near him and, if she is pleased
by it, will sing with him. This melodious species has different types of songs
to use on several occasions. For example, those used to mark territory is different
from those used by mated pairs in their "duet" to strengthen their
bond.
They usually construct a large cup nest in various sorts of cavities, taking
about a week to build. The nest is made from small dry sticks and is usually
lined with a variety of different materials. These include: feather, hair, wool,
spider cocoons, strips of bark, rootlets and moss. The male wren finds dry sticks,
which he adds to the nest. Once he is done, the female inspects the nest; but
if she does not approve of the construction, she will throw any unwanted sticks
to the ground. After this process, the female lines the nest. Nest cavities
are usually a few meters above ground at most, but they can occasionally have
nests in high cliffs.
The common melodious wrens' clutch is usually between two and eight red-blotched
cream eggs. Only the female incubates these, for around 12–19 days, and she
will every now and then leave the nest for various reasons. While she is in
the nest, the male provides her with food. The young, which like all passerines
hatch almost naked and helpless, take another 15–19 days or so to fledge. They
are fed by both parents, and need plenty of food given their tiny size. As the
young near fledgling, the parents spend much of their time procuring food for
them. They reach sexual maturity with 6 months and have a lifespan of 7 years.
There are other species in the genus Cambaxirra, with each one of them having
different songs. The Noronha
melodious wren (Cambaxirra noronhensis) is endemic to the Archipelago
of Fernando de Noronha. They have 11 cm of length and a wingspan of 22 cm. Their
plumage is rufous brown above, pale gray below, barred with darker brown and
black, even on wings and tail. The bill is dark brown, the legs pale brown.
Young birds are less distinctly barred. The restinga
melodious wren, or restinga eastern uirapuru (Cambaxirra litoralis)
is endemic to the Atlantic coast restingas, an ecoregion closely associated
with the Atlantic forest. They have 13 cm of length and 24 cm of wingspan. They
have brown head and upperparts, barred with black on the wings and narrow tail.
It has a stronger buff eyebrow stripe and black eyestripe. Their underparts
are gray and white in the throat. The flanks and undertail coverts have dark
brown bars. Another species is the serra
melodious wren (Cambaxirra serrana), which is endemic to the mountainous
Atlantic forests of Southeastern South America. They have 12 cm of length and
23 cm of wingspan. They have brown upperparts with small black and white spots
and pale grey underparts with a light brown rump. Additional distinctive features
include a dark grey line over the eye. The last representative of this genus
is the southern
melodious wren (Cambaxirra australis), which inhabits the forests of
Southern South America. They have brown upperparts with a reddish-brown belly
and flanks and a white throat and breast. The back is black with white stripes.
They have a dark cap with a white line over the eyes. During winter, this species
migrates to the areas of Southern Amazon rainforest.
This bird species was discovered by João Vitor Coutinho,
Brazil.
Marvelous
musician wren, or marvelous true-uirapuru (Irapuru mirus)
Order: Passerine birds (Passeriformes)
Family: Wrens (Troglodytidae)
Habitat: tropical forests of Amazonia.
The human actions during the Holocene caused the decline of many species. The
Amazon rainforest suffered with deforestation and exploitation of its soil for
the extraction of gold and other valuable minerals. Many species were only able
to survive such an onslaught because they retreated to remote areas that were
difficult for the humans to reach. Living safely in these areas, they continued
to evolve. Now, their descendants in the Neocene are the signals of their success.
One of these animals is the marvelous musician wren, the type species of the
Genus Irapuru. This bird is a descendant of the Genus Cyphorhinus, called as
uirapurus by the Tupi people of Brazil.
The marvelous musician wren have 13 cm of length and 20 cm of wingspan. The
adult has a dark brown forehead and forecrown that becomes chestnut brown in
the back and rump. It has a narrow white supercilium, a chestnut brown stripe
behind the eye, and an orange-brown throat and chest. The tail is brown with
cream-white tips. Its flanks and lower belly are yellowish brown. The juvenile
is similar but sometimes has faint barring on its lower belly. The beak and
legs are pale grey. Their vocalization is a series of a gradable whistles, varying
greatly in pitch.
This bird forages primarily on and near the ground, searching debris and leaf
litter. Its diet is mostly invertebrates, including insects, spiders, and crustaceans,
but it also occasionally eats berries. It usually forages alone or in pairs.
Though it sometimes follows army ant swarms it does not usually join mixed-species
foraging flocks.
The marvelous musician wren is monogamous, with pairs staying together for the
rest of their lives. The breeding season starts during the beginning of the
dry season. Males that don’t have pairs will congregate in a clean part of the
forest ground. Here they will realize a specific vocalization to attract the
females. With one land near them, the males will sing a song used for courtship.
If she chose one of them, the female will approach him and start to make a "duet"
with the male. These presentations can have from two to seven males and the
order of the singing to the female is decided through intimidation and sometimes
by fights between the males. Mated pairs will sing in "duet" to strengthen
their bond.
The parents work together to build the nest, which is spherical with an entrance
through a funnel-shaped path and is constructed of pieces of leaves and coarse
grass. The female lays two white eggs. She is the only responsible for incubating
them, with the male bringing food for her and protecting the area of the nest
against members of their own species. The incubation lasts for 15 days. The
young hatch almost naked and helpless, taking another 16 days to fledge. They
are fed by both parents, and need plenty of food given their tiny size. As the
young near fledgling, the parents spend much of their time procuring food for
them. They reach sexual maturity with 7 months and have a lifespan of 8 years.
The Tepui musician
wren, or Tepui true-uirapuru (Irapuru canorus) is
another species of the genus Irapuru. This species inhabits the tepuis located
in the Northern areas of Amazon. They have 11 cm of length and 18 cm of wingspan.
They have a similar plumage to that of the Marvelous musician wren, with the
only exception being the reddish-brown feathers in their underparts. Their song
is also different from that of the lowland species. Another member of the genus
is the Andean
musician wren, or Andean true-uirapuru (Irapuru andinus).
This species inhabits the altitude forests in the slopes of the Andes. They
have 12 cm of length and 19 cm of wingspan. Their plumage is similar to that
of their congenere, except for the lack of white supercilium and the presence
of creamy-white feathers in the belly. Their song is also different and the
males do not congregate in groups to present for the female, rather being very
aggressive to each other during the breeding.
This bird species was discovered by João Vitor Coutinho, Brazil.
Marine
runner (Thalassocinclus arcticus)
Order: Passerine birds (Passeriformes)
Family: Water ouzels (Cinclidae)
Habitat: coast of Arctic Ocean and islands.

Picture by Alexander Smyslov
In Holocene coasts of seas were a habitat of various birds,
but among them there were no representatives of largest order of Passerine birds.
After mass extinction at the boundary of Holocene and Neocene the bird's population
of coasts had “thinned” a little: it is the result of “plankton accident” which
was the consequence of change of ocean currents as a result of extensive congelation.
When ice had receded, new ecosystems had started to form, and the chance to
master new inhabitancy had appeared at warblers. In various places of Earth
species of passerine birds adapted to inhabiting in sea evolved. In sea waters
of Subantarctic yellow-headed
divesparrow hunts for small fish and crustaceans, and at the other pole,
at coast of Arctic Ocean, sea shallow waters are mastered by its distant relative,
marine runner, also belonging to water ouzels family.
The marine runner is an thrush-sized bird, the descendant of water ouzels (Cinclus)
living at sea coasts. This bird does not involve the special attention to itself,
when it searches for food at sea coast. It is colored softly: the main color
is chestnut, on chest there is lighter spot; beak and legs are black. Marine
runner differs in short body, but it has long strong legs. Bird can run fast
on the ground. Wings and tail of marine runner are short; bird flies quickly,
but reluctantly. The disturbed bird of this species keeps tail up and even bent
to back. And in this case marine runner becomes more appreciable: quill feathers
on edges of tail at this bird are white. When disturbed bird has lifted tail,
they are well visible on background of other dark feathering. It serves as an
original alarm signal – birds observe of congeners and also start to behave
more cautiously.
Marine runner eats small sea creatures – invertebrates, small fishes, their
fry and larvae. At this bird there is short chisel-like beak, by which it can
peck out armours and shells of invertebrates. But in general this bird hunts
at all not like its Antarctic relative, yellow-headed divesparrow. Divesparrow
swims in thickness of water, flapping wings as flippers, and marine runner runs
at the bottom searching for food, clinging by claws for stones and shells of
molluscs. Its hunting passes as follows: bird dives under water and searches
for catch, running on bottom. Each such immersing lasts not longer than one
minute: air pushes out marine runner from water, as a fuse, and bird spends
some efforts to be kept at the bottom. Thus nostrils of marine runner are densely
closed by skinny operculums. Almost like this hunting at water ouzels proceeded,
but they lived in streams, and marine runner applies similar behavior in sea.
Current helped water ouzels to keep under water – birds held wings so that current
pressed them to bottom. Marine runner uses sea surf for same purpose: when wave
surges up a coast, bird safely dives and hunts. During inflow or outflow marine
runner stays under water even longer.
To remain lighter than water and keep feathering dry, this bird is compelled
to spend a lot of time for feathers’ cleaning. Usually after successful hunting
bird shakes itself for a long time and carefully greases feathers with secretions
of coccygeal gland. Coccygeal gland of this bird is very much advanced: it is
some times larger, than at ground birds of the same size. But even during the
strongest rain this bird remains dry – it only stirres feathers up, dumping
drops of water. Marine runner easily emerges (to bird it is enough to unclench
claws for this purpose), and at once can fly up after emersion right from the
surface of water.
Courtship displays of marine runners begin in spring, right after snow thawing.
Male finds place convenient for nesting and protected from predators (usually
among stones or in bush), and involves female with singing. Marine runners sing
simply – their song differs in monotony and abundance of bitter sounds. Pairs
form to one nesting season. If female liked place chosen by male for nesting,
she shows to the male signs of submission: it “bows” with fanlikely opened tail.
The sign of attachment at pair of birds is ritual of mutual clearing and greasing
of feathering.
Nest looking like open bowl is made by both birds of pair of poor Arctic vegetation
and rests of dried up seaweed. In clutch there are about five eggs with spotty
brown shell. Female hatches clutch mainly, and male feeds her. After nestlings
hatching female warms them while they will become covered with down. At this
time male almost all the day is occupied with searches of food and strongly
grows thin. When nestlings become covered by down and become steadier to cold,
female also starts to feed them. The posterity leaves nest approximately at
the fifth week of life. Young birds differ in brown one-colour feathering which
changes to coloration characteristic for adult bird to an autumn.
The ocean may feed marine runner also in winter, but these birds do not withstand
bad weather and storm, therefore for winter they migrate to the south. Basic
ways of migrations of marine runner pass lengthways channels of large rivers,
but birds from Scandinavia migrate along the Atlantic coast up to the Western
Europe.
This species of birds is discovered by Simon, the forum member.
Hawaiian
hookbill (Curvirostrornis heliciphagus)
Order: Passerine birds (Passeriformes)
Family: Starlings (Sturnidae)
Habitat: Hawaiian Islands – Big Island rainforests.

Picture by Alexander Smyslov
The fauna of Hawaiian Islands has substantially suffered because
of human influence. To Neocene any species of the birds existing there before
human colonization of islands, or their descendants had not remained in Hawaii.
Descendants of species introduced by people, or later settlers had appeared
at these islands after disappearance of people compose mainly new fauna of Hawaiian
Islands. Numerous descendants of birds of various families – finches, weaverbirds,
and other ones – had appeared so at islands. The part of new settlers had not
died out after disappearance of people, and their descendants evolved also have
occupied various ecological niches at Hawaiian islands.
In conditions of competition strict specialization appears the favorable strategy
of evolution. The descendant of myna (Acridotheres tristis) introduced to Hawaii,
the Hawaiian hookbill, is one of such “narrow specialists”. This large finch-sized
warbler with curve long beak and wide short tail eats extremely ground
snails plentifully inhabiting woods of islands. It lives mainly in
wet woods and marshlands where more snails live.
The feathering of this bird is colored rather brightly in comparison with dusky
feathering of its ancestor. At the Hawaiian hookbill there is dark brown back
with light border on feathers, forming pattern similar to wood bark one. Primary
feathers of wings and steering feathers of tail are black. But the bottom part
of body is colored brightly: chest is bright red, and stomach is yellow. At
males in feathering there is more red color, than at females. At birds of both
sexes the area around of eyes is featherless and covered with white skin. And
on corners of beak there are skin lobes of pinkish color. At displaying male
they become bright red and increased in size because of blood inflow.
The beak of Hawaiian hookbill is long and bent, maxilla is longer than mandible,
and its tip slightly deviates to the left side. It is just connected to food
specialization of this species: bird eats snails, pulling them out from shells.
This bird tears off small snails from leaves and drags them to favorite branch
serving as “butchery”. Here this bird presses caught snail by paw against bark,
and by dexterous movement of beak tears its body from shell. Under “butcheries”
of hookbills a plenty of shells of had eaten molluscs is gathering.
When the bird attacks large snail, the mollusc usually presses edges of shell
to substratum, and feels like in relative safety. But the hookbill by prick
of beak under edge of shell forces mollusc to come unstuck from substratum and
to involve to the shell even more deeply. After that bird simply overturns shell,
and right on the spot pecks out body of mollusc.
Hookbills are solitary and territorial birds protecting the nesting site against
neighbours. Only in nesting season they keep in pairs and in common bring up
nestlings. These birds nest two times per one year, arranging nests in tree-trunk
hollows. In courtship season the male occupies a tree-trunk hollow, and starts
to sing, involving the female. Hookbills differ in abilities of imitator and
improvisator: as a rule, the male imitates voices of birds living in the neighbourhood
for courtship songs, adding to them the characteristic “buzzing” note. Usual
voice of these birds is lingering buzzing warbles. In clutch there are 3 - 4
rather large eggs with white shell. The female hatches mainly, and the male
feeds her.
The incubating lasts about two weeks, and next month the young growth stays
in tree-trunk hollow. When feathers complete growth at young birds, they abandon
nest and about one month migrate with parents. Gradually male starts to show
aggression to them, and young growth and female leave together his territory.
Having had a rest and having fed up, male starts to prepare for new nesting
season.
Young birds in juvenile feathering do not have red color, and skin on naked
parts of head is grey instead of white. They become able to nest at the age
of 12-15 months.
Cape
starling (Sturnus capensis)
Order: Passerine birds (Passeriformes)
Family: Starlings (Sturnidae)
Habitat: savannas of South Africa.

Picture by Pavel Volkov
Widespread, adaptable and fed by humans, European starlings
(Sturnus vulgaris) survived easily the biosphere crisis of Holocene and Early
Neocene. Once they were settled by people in North America, South Africa and
Australia, where people later tried to exterminate them, but to no avail. After
the human disappearance, the birds fit into the newly forming ecosystems and
evolved in their new homeland.
Cape starling, which lives in South Africa, is a relatively large bird by the
standards of the family. Body length of an adult bird is 35 cm, wingspan 55
cm, weight up to 210 grams. Back and head are black with white mottling; chest
and abdomen are beige. Beak is more powerful than that of the ancestor and dark.
Cape starling looks more like a jay. Sexual dimorphism is not pronounced.
This species is a monogamous bird; pairs are formed up to the death of one of
the mates. These birds keep and even nest in small groups of 5-7 pairs. Cape
starling feeds mainly on insects, preferring beetles and grasshoppers, and on
hard seeds – a hard beak helps them cope with solid food. Bird can easily kill
and eat a small lizard, a murine rodent or a chick. Flocks of birds protect
their feeding territory (with an area of approximately 2.5 square kilometers)
from adult competitors, but in case of losses from predators, their groups can
adopt young birds that get a chance to make a pair.
The breeding season lasts from August to January and during this time Cape starlings
manage to grow 2, and in some years even 3 broods. Courtship begins with squeaky
trills and whistles, which include the sounds of other animals – starlings often
mimic songs of other birds and improvise based on them. In family couples, everything
is limited to singing. Unmarried or widowed males, while singing, perch above
others and flutter their wings while displaying themselves. The female, choosing
a mate, perches next to him, taking the pose of a nestling begging for food.
The nest represents a cup of grass in a hollow or in deep crevices between stones.
Colonies of several dozen pairs, nesting synchronously and jointly protecting
the nesting place, are often formed in the rocks. Sometimes these birds drive
termite woodpeckers out of their
hollow and occupy their shelter. Egg laying begins synchronously in all females
of the group. There are 5-7 eggs in the clutch, mostly the female incubates
them. The incubation period is 2 weeks. The juveniles stay in the nest for 19
days and are fed by both parents. Young birds at first just wander in flocks,
trying to find or occupy a territory for themselves. Most of them die at this
time from stress, diseases or predators. Sexual maturity occurs at 8 months;
life expectancy is up to 14 years.
This bird species was discovered by Nick, the forum member.
Snow
or white-chested starling (Sturnus nivicola)
Order: Passerine birds (Passeriformes)
Family: Starlings (Sturnidae)
Habitat: northern Europe from Scandinavia to the Northern Urals.

Picture by Pavel Volkov
Initial image by Shervin |
European starlings (Sturnus vulgaris) easily survived the human
era. Both the abundance and unpretentiousness of birds and the attitude of people
who fed them and provided them with artificial dwellings played a role in their
survival. After the human disappearance, their numbers decreased slightly, but
starlings remained common birds in Eurasia and at other continents where they
were introduced. In due course of evolution, they have given rise to wast range
of new species, differing in lifestyle features and details of anatomy. One
of them is snow starling, which lives in northern Europe.
Snow starling is a bird about the same size as its ancestor: body length up
to 24 cm, wingspan 55 cm. The coloration of the bird is contrasting, bicolored:
head, back, tail and wings are black, breast and belly are white. There are
no lighter markings on the dark plumage, but there is a strongly pronounced
metallic luster – copper and sky blue. Snow starlings live and nest in small
groups of 10-12 ones, feed together and guard nesting sites. In relation to
small songbirds, snow starlings are quite aggressive – they can attack adult
birds, and if possible peck their eggs.
Snow starling is an omnivorous bird: it feeds on insects and worms, as well
as on buds, berries and seeds. Due to this diet, the bird’s lifestyle has changed
compared to its ancestor: up to 7% of birds remain to winter in their homeland
(hence the species epithet), in warm winters the proportion of wintering birds
increases. The rest of the birds migrate to Caucasus, the Balkans and the Apennines
for the winter, to the mountain oases of Mediterranean lowland and to North
Africa.
The mating season begins a week after the birds return from wintering – in early
April. At this time the singing of snow starlings, which includes elements of
imitation of the voices of other birds in Europe and Africa, is often heared.
This species nests in shelters – usually in crevices between rocks in mountainous
areas. In the forest area, the density of the settlement of starlings is limited
by the presence of hollow trees suitable for nesting. The nest is built of thin
twigs and grass stalks. In a clutch, there are 5-7 eggs with white or light
olive shell, which are incubated by the female for about 2 weeks. Nestlings
hatch naked and helpless. Parents take care of them for 22-25 days, after which
the young starlings leave the nest. In the years of abundance of insects, snow
starlings make a second clutch in the summer. Sexual maturity of young birds
occurs the next year; life expectancy is up to 12 years.
This bird species was discovered by Nick, the forum member.
Scaled
starling (Sturnus squamosus)
Order: Passerine birds (Passeriformes)
Family: Starlings (Sturnidae)
Habitat: South America, from warm temperate to cold areas.

Picture by Pavel Volkov
During the Holocene, the man introduced many species to other
continents. One of these species was the common starling (Sturnus vulgaris),
introduced in Southern South America. This species was capable of surviving,
giving rise to the scaled starling in the Neocene. This gregarious bird inhabits
all the biomes from the temperate forests of Tierra del Fuego to the Atlantic
forests of Southeastern South America. The population that inhabits the temperate
areas migrate in large flocks of thousand individuals to the Amazon rainforest
during the austral winter. Upon reaching their wintering areas, the flocks disperse.
Scaled starlings have 18 to 22 cm of length and a wingspan of 30 to 43 cm. The
plumage is iridescent purple with dark edges on the feathers, giving the bird
a "scaly" appearance. During the winter, their plumage becomes more
dull and full of white spots in the belly and chest. Juveniles are grey-brown
and by their first winter resemble adults though often retaining some brown
juvenile feathering, especially on the head. Their sharp, blade-like bill is
bright yellow, while the legs and feet are bright orange.
The scaled starling is largely insectivorous and feeds on several types of insects.
Prey are consumed in both adult and larvae stages of development, and scaled
starlings will also feed on earthworms, snails, small amphibians and lizards.
While the consumption of invertebrates is necessary for successful breeding,
scaled starlings are omnivorous and can also eat grains, seeds, fruits and nectar
if the opportunity arises.
These birds are monogamous, but the males can occasionally breed with more than
one female. For the population living in the temperate areas of South America,
the breeding season happens during the winter, while those living in tropical
areas can breed during the entire year. Unpaired males find a suitable cavity
and begin to build nests in order to attract single females, often decorating
the nest with ornaments such as flowers and fresh green material, which the
female later disassembles upon accepting him as a mate. The amount of green
material is not important, as long as some is present, but the presence of herbs
in the decorative material is important in attracting a mate.
The males sing throughout much of the construction and even more so when a female
approaches his nest. Following copulation, the male and female continue to build
the nest. Nests may be in any type of hole, common locations include inside
hollowed trees and tree stumps. The female lays eggs on a daily basis over a
period of several days. If an egg is lost during this time, she will lay another
to replace it. There are normally four or five eggs that are ovoid in shape
and white. Incubation lasts thirteen days, although the last egg laid may take
24 hours longer than the first to hatch. Both parents share the responsibility
of brooding the eggs, but the female spends more time incubating them than does
the male, and is the only parent to do so at night when the male returns to
the communal roost. The young are born blind and naked. They develop light fluffy
down within seven days of hatching and can see within nine days. Nestlings remain
in the nest for three weeks, where they are fed continuously by both parents.
Fledglings continue to be fed for another one or two weeks after leaving the
nest. The lifespan of this species is 7-8 years, but many perish before even
reaching 5 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Australian
Ornate Starling (Australosturnus splendidus)
Order: Passerine Birds (Passeriformes)
Family: Starlings (Sturnidae)
Habitat: open country and forest throughout Meganesia, avoids arid areas.

Picture by Pavel Volkov
Australia during the age of man had suffered many introduced
pest species which subsequently proved to dominate and displace local kinds.
Many of these continued to prosper during the Neocene, with some notable exceptions.
Introduced birds were less successful here, with the exception of, the Common
Starling (Sturnus vulgaris), which came to reproduce in great numbers.
The descendant of this form is the Australian Ornate Starling, a diurnal form
of roughly similar size and shape to its ancestor, but which is considerably
more beautiful. The eyes, feet and beak are a striking bright red, the feathers
of the head are an iridescent purple, with this becoming emerald green iridescence
on the chest and upper part of the wing, the wing feathers are a bright golden
yellow scalloped with black, and the underparts of the bird are a solid bright
yellow, the tail an iridescent bluish green. The eponymous “stars” of its ancestor
only remain on the chest and upper wing, and these are spots of reflective yellow.
In many ways it has come to resemble the tropical starlings of Africa.
Apart from its colour, it greatly resembles its ancestor; birds feed on the
ground in large flocks, looking for seeds, berries and insects. As with its
ancestor it forms great large murmurations in flight. The birds call resembles
that of its ancestor but is slightly harsher and less prone to mimic other birds.
As with many small birds, predators include raptorial birds such as hawks, and
in this case marsupial predators like Dasyurids, lizards and snakes will eagerly
consume their eggs.
Breeding can occur anytime but most often during spring and summer. Females
make a cup nest of twigs and grasses in any hollow they can find, in trees or
rock outcrops, they will actively drive other birds out of potential nesting
sites. Incubation is about 13 days and consists of 5 to 7 eggs, which are a
pale blue dotted with black. The young fledge in about three weeks. Pairs usually
produce 2 broods in a season. Lifespan is up to 7 years.
This species of birds was discovered by Timothy Donald Morris, Adelaide, Australia.
Narrow-billed
Mynah (Novaegracula acutirostris)
Order: Passerine Birds (Passeriformes)
Family: Starlings (Sturnidae)
Habitat: Forests of different sorts, scrub-land, shrubby grassland, North Island
and the northern part of South Island, New Zealand.
The bird fauna of Neocene New Zealand was formed in three main ways, partly
by rare surviving endemic birds that endured the age of man, to a small degree
also of newcomers that dispersed there naturally from Australia, and most of
all by introduced species that had prospered because of man.
One of the most notorious invasive birds was the Indian Mynah (Acridotheres
tristis), a confident, social bird that often harassed other kinds of bird,
and competed for nesting sites. In the Neocene of New Zealand, it has left a
few successful species there. One of these is the Narrow-billed Mynah, a common
sight in the forests and scrub-land of the warmer parts of the islands.
In appearance, it is about the same size and stature as its ancestor, averaging
about 25 centimetres long, with a wingspan of up to 40 centimeters, weighing
up to 140 grams. Appearance has not changed a great deal, the head and shoulders
are black and slightly glossy, body and wings are middle shade of greyish brown,
lighter on underparts, tips of the primaries are black. The legs, beak and skin
around the face are a bold yellow orange, there is a distinct ring of skin around
the eye, the very end of the bill is black. Its body-shape and posture is a
little slimmer than its ancestor, but the most important difference is its beak,
which is longer, and noticeably more slender, hence this bird's name.
This myna's bill is connected to its diet, whereas its ancestor was an omnivore
with a preference for insects, small vertebrates and scavenging, this bird feeds
almost entirely on various kinds of insects, which it forages actively for in
trees and vegetation, and on the ground. Insects such as grasshoppers, catterpillars,
bees, crickets, roaches, beetles and wasps are commonly taken, when attacking
a bee or wasp it carefully kills it and removes the sting. Occasionally, soft
berries or nectar are also eaten.
These birds are very social, living in large flocks that may number between
100 to even 700, they disperse far and wide in smaller groups throughout the
day, and nest communally at night in the crowns of large trees. Social life
of this species involves mutual grooming and sharing information about food
sources, as well as social hierarchies. The mynah generally pair for life, and
will tend to pair up as mature adolescents within their own flock, though imbalanced
numbers of either sex can cause the flock to break up and seek mates elsewhere.
They are able to breed throughout the year, with the time varying depending
on climate and abundance of food. Nest is usually made in the tree-trunk hollow
or in a rock crevice, out of grass, fine twigs and thin roots, as well as spider
silk and decayed plant matter. The female will lay a clutch of 4-6 eggs, which
hatch within about 18 days, the chicks are cared for by both parents, and fledge
within 36 days. Common predators include cats, predatory marsupials and hawks,
lizards and large saw-leg grasshoppers may steal their eggs. Lifespan is generally
between 4 and 10 years.
This bird species was discovered by Timothy Morris, Adelaide, Australia.
Bristle-headed
starling (Setocephalornis aculeatus)
Order: Passerine birds (Passeriformes)
Family: Spike-headed starlings (Echinosturnidae)
Habitat: Southern Asia, from the north of Hindustan up to Indochina; woods and
light forests at plains and in foothills.
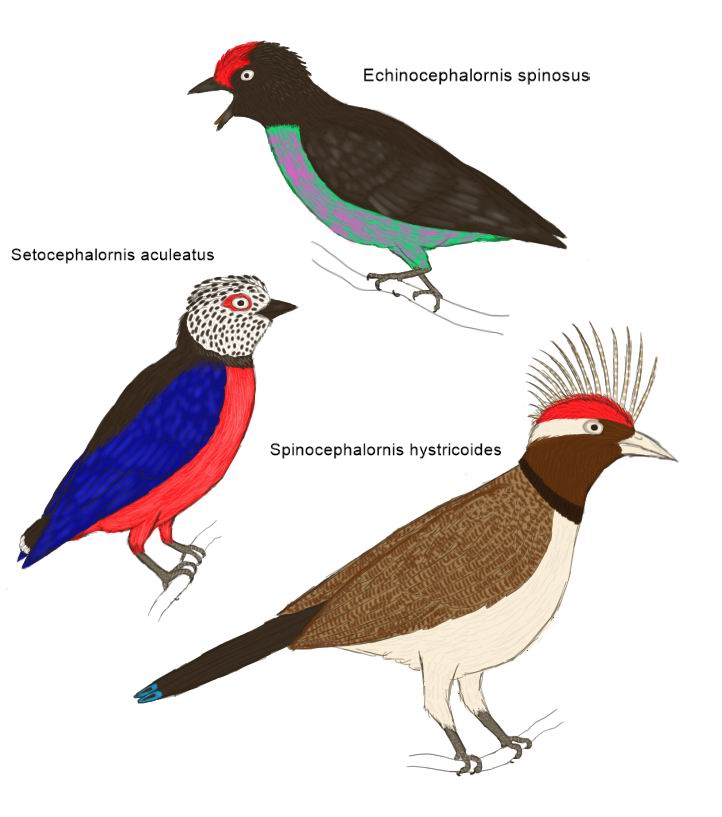
Picture by FanboyPhilosopher
Passerine birds order is the youngest and most actively evolving
group of birds. After mass extinction at the boundary of Holocene and Neocene
this group had kept the greater variety, rather than other groups of birds that
had permitted to various passerines to occupy new ecological niches, successfully
competing with relic predatory birds and even with small fine petrels. When
tropical woods were restored at the huge areas of continents and islands, it
had caused active speciation at small passerines. These birds, being earlier
rather similar in anatomic features, had formed some original groups remarkable
in special anatomy.
One family of small warblers descending from any tropical starlings had developed
the amazing protective adaptation protecting from small feathered and four-footed
predators. The feathering on head of these birds had changed, having turned
to the kind of weapon: shafts of feathers as if had “sprouted” through the vane,
forming sharp spikes on tips of feathers. Because of it the present group of
birds had been named “spike-headed starlings”. By the general shape they are
small passerine birds living in tropics and subtropics of Asia. One species
had crossed the passage between Asia and Zinj Land and had formed the particular
species of the separate genus at this subcontinent. But the majority of
species lives mainly in Asia.
Bristle-headed starling is one of primitive species of the family, settled very
widely. It is a bird about 20 cm long including short wide tail. Its feathers
on head still keep advanced vanes, and tips of feather shafts form only small
bristles serving mainly for displaying tournaments between males. Stings of
such bristles, possibly, may drive off from nest only predators having sensitive
soft skin (like wood amphibians or small mammals).
The head of bristle-headed starling has contrast dichromatic colouring: tips
of feathers are black and other vane is white. Such colouring may be discerned
well and may be remembered even by predators not having color sight. Chest of
bristle-headed starling is red with metal shine. Nape of bird, back part of
neck and back are black. On neck there is black “collar” – the strip of black
feathers having iridescent shine. The tail is also black, but on tips of feathers
there are white spots. Wings are dark blue with bluish metal shade, pointed
at the ends. Flight of this bird is fast, prompt and maneuverable. Bristle-headed
starling lives at tops of trees, very seldom flying down on the ground.
Beak of bristle-headed starling is straight, strong, thick and short (its length
is less than half of bird’s head), black. Eyes of bird are edged by site of
naked reddish skin – at males skin is brighter, than at females. An iris of
the eye is white.
Bristle-headed starlings eat fruits of various tree species, pecking through
the fruit skin. They swallow small berries entirely. The vegetative food is
supplemented with insects and small snails – this starling crushes them by beak
and eats entirely with shell splinters.
These birds nest up to three times per year in hollows of trees, occupying abandoned
nests of woodpeckers and parrots. Pair at this species is formed only for one
nesting season. Male finds suitable tree-trunk hollow and drives competitors
off from it, using strong beak as the weapon. For same purpose sharp prickly
feathers on head serve to it. When near the nest two competing males meet, they
arrange improvised duel: having fluffed feathers on head, they show to each
other contrastly colored feathers with pointed tips. Usually weaker male recedes
during the demonstration of head feathering. But when contenders are equal in
force and nobody wants to recede, the real struggle begins. Having bent head
forward, birds try to prick each other with tips of feathers in throat or chest.
Such battle receptions are safe for fighting birds: feathers bend easily, and
their tips are too weak to pierce skin of bird and to put any serious wound.
If competitors or predators are not present nearby, the male of bristle-headed
starling actively warbles, involving the female to the nest. As well as other
starlings, it imitates songs of other species of birds, adding to them some
specific warbles. When any female starts to show interest to male and its nest,
male shows to her prickly feathering of head, not interrupting warbling. At
these moments male, having fluffed feathers on head, alternates poses of domination
and humility. Showing the force, he extends on legs vertically and fluffs feathers
on head. Showing to the female readiness to accept her as an equal partner,
male squats, having pressed feathers to head and having bent forward.
In family of these birds the male dominates: he is larger than female and is
colored brighter then she is. Pair is formed only for one breeding season, and
breaks up, when young birds become independent. Birds do not equip the tree-trunk
hollow specially: they only rake up and throw out from it the dust. Female lays
4 – 5 eggs with white shell right on bottom of tree-trunk hollow in dust. Both
partners hatch eggs, but the male gives more time to protection of territory
and search of food for the female and hatch.
Nestlings hatch in 2 weeks. They are naked and blind, only on wings at them
germs of feathers appear. By the tenth day of life they become covered by down
and at them primary feathers of wings start to develop. Approximately at monthly
age young birds leave nest. They have dusky juvenile colouring – brown with
“marble” pattern similar to wood bark. Feathers on their heads still soft: they
will be replaced with adult bristle-like ones after the first mew. Then the
dusky juvenile feathering will give up the place to bright one characteristic
for adult birds. It will take place approximately at seven-mouth age – then
young birds become able to nesting.
More specialized species of spike-headed starlings live in woods of Southern
and South-Eastern Asia: their prickly feathers became more similar to true spikes;
they are strong and frequently may be gathered to mobile prickly crests. Birds
armed in similar way, do not use needles in fights with congeners, but safely
attack small predators, striking to them impacts by head.
Hedgehog
bird, or spiny spike-headed starling (Echinocephalornis spinosus)
is found in south of Hindustan, Sri Lanka. Sometimes it makes flights
between islands and is found even at nearest islands of Indian ocean.
On the head of this bird sharp needle-shaped feathers grow in regular intervals
among usual feathers. They are rather short and also are appreciable only when
bird lifts them specially. On the forehead of the hedgehog bird there is a site
of bright red feathers. When bird is angry, feathers on forehead rise, increasing
the visible area of spot. It is a mark of threat, which is showing by this species
to smaller predators. Threatening the enemy, bird starts to bounce head. If
this demonstration has not conceived action, bird attacks the aggressor, rushing
to it and striking it by head. The same receptions of intimidation are used
by males during courtship tournaments. But fights of males are strongly ritualistic
and also are more similar to demonstration: birds do not touch to each other
and show prickly feathering of head at a range. To courtship season forehead
feathers of males grow to high crest.
Feathering on chest of these birds is green with pinkish metal shine; wings
at males are black with steel shine, at females brown with white cross strips
on primary feathers.
Porcupine
bird, or long-needled spike-headed starling (Spinocephalornis
hystricoides) is larger than previous species: its size is about 30
cm including tail. This species lives in damp woods of South-Eastern Asia, at
the Jakarta Coast and nearby islands, frequently developing mangrove thickets.
Porcupine bird is armed better, than other congeners: along the middle part
of head the crest of long cross-striped brownish-white needle feathers grows.
The length of needles growing on the forehead exceeds length of head of this
bird. The crest of needles is very mobile - the bird any way can lift and lower
it depending on mood. Sides of head are colored brightly: along the basis of
crest of needles red strip passes, and from eye up to nape in parallel it there
is white one. The angry bird lifts crest and turns head from side to side, showing
it to aggressor and observing for its reaction. Feathers on each side of head
fluff up, increasing the area of brightly colored sites. Thus the porcupine
bird utters loud unpleasant cry.
Being well armed birds, porcupine birds behave very cautiously, sorting out
their relations, and their courtship duels are extremely ritualistic. Males,
sorting out their relations, limit only to demonstration of ornaments and bright
feathers on each side of head. Defensive tactic of long-needled spike-headed
starling is so successful, that it has two successful imitators: bird
of flycatchers family and parrot.
Shading bright strips along the crest, the bottom part of head of the porcupine
bird is dark brown. Beak at this species is white; an iris of the eye is white
too (because of it the eye is less visible on the background of head feathering).
On the top part of body of the porcupine bird there is brown “scaly” pattern
on lighter brown background; on neck there is dark cross strip and stomach is
white.
Tail of this bird is dark, long and narrow. On tips of several pairs of quill
feathers in middle of tail there are dark blue shining oculate spots. At males
such spots are present on all feathers, and at females they sometimes can be
absent. It is a unique attribute of sexual dimorphism at long-needled spike-headed
starling.
The biology of this species is similar to other hedgehog starlings, but long-needled
spike-headed starling nests less often – no more than twice a year. As against
“fighting call”, the warble of the porcupine bird male differs in complexity
and melody: it includes imitations of songs of other birds.
In hatch at this species it may be no more than four nestlings, but the survival
rate of them is very high. Needle-shaped feathers start to grow at them right
after the leaving of nest, and for some days they reach about half of length
of needles of adult bird. After the second mew the bird gets colouring, characteristic
for adult individuals, and its needles reach the normal size.
Double-crested spike-headed starling (Spinolophornis dicristatus)
Order: Passerine birds (Passeriformes)
Family: Spike-headed starlings (Echinosturnidae)
Habitat: Zinj Land, tropical woods at east coast and in central part of subcontinent.
This species of birds lives in separation from other species of family, characteristic
mainly for Asia. Its close relative is, most likely, long-needled spike-headed
starling, or the porcupine bird, able to make flights through sea passages
(most likely, at them there was common ancestor). But the biology of these
species differs considerably.
Double-crested spike-headed starling concerns to large species of family –
it is a small crow-sized bird. Similarly to all representatives of family,
this species has feathers modified to needles on head, but they grow very originally.
In middle part of head the bright red longitudinal strip of usual feathers
stretches, on each side of which strong and pointed needle-shaped feathers
form two parallel crests. At the angry bird crests rise upwards and in sides,
making red strip more appreciable. Warning an aggressor about the protecting,
double-crested spike-headed starling bends head forward, showing bright feathers.
This demonstration is accompanied by lingering unpleasant cries of bird similar
to gnashing.
Colouring of feathering of double-crested spike-headed starling is very motley
and bright: back is dark green with metal shine, chest is violet, and stomach
is creamy-white. Tail and wings are brown with dark tips of feathers. Sides
of head of this bird are black; on this background light blue iris of eyes
and white needle-shaped feathers with black tips are good visible. Beak is
white, thick and slightly deflexed.
This species of starlings is more carnivorous, rather than its Asian congeners:
it eats vegetative food extremely seldom, preferring insects and small vertebrates.
Occasionally it ravages nests of medium-sized birds, eating eggs and nestlings.
This bird kills small catch by sharp impacts against branch. Also the double-crested
spike-headed starling is aggressive to neighbours, and can repulse even to
large birds (for example, to crestbills – local birds closely related to corvine
birds). Protecting itself from predators, it strikes lateral impacts by head,
trying to strike the enemy by spikes.
Similarly to distant ancestors, starlings, this species nests in tree-trunk
hollows. But the good tree-trunk hollow in wood happens more often occupied.
However it is not problem for present species of birds: due to aggression the
double-crested spike-headed starling easily finds to itself suitable place
for nesting, expelling from tree-trunk hollow of ones lived in it earlier.
It easily attacks cat-sized animals, and prevails over them due to aggression
and sharp needles on head. Even parrots armed with powerful beak recede before
an impact of this bird. These birds feel like in wood so confidently, that
the male may begin courtship display even near to inhabited tree-trunk hollow
while its inhabitants are not expelled. When pair is formed, birds in common
attack owners of tree-trunk hollow and grasp to themselves a convenient residence.
Displaying male warbles for a long time near the chosen tree-trunk hollow,
occasionally driving away other birds which quite naturally try to attack it,
sometimes
even by whole flock.
In clutch of these birds there are no more than four eggs. They are hatched
only by the female, she also cares of nestlings the first time. Male during
the hatching protects territory and gathers forage for the female and nestlings.
At the age of six weeks nestlings leave nest and about one week live with parents.
Their juvenile feathering is dim: on green feathers of back there is no shine,
and head, chest and stomach are brown. Needle-shaped feathers on head at young
birds are short and black. Young birds are nonaggressive and very reserved;
they eat mainly insects. At the age of about nine months under influence of
hormones at maturing birds the juvenile feathering is replaced by adult one.
They become bright and cocky, start to hunt small vertebrates, to attack other
birds and to ravage their nests. And one-year-old birds start to nest.
Haltia
(Haltia neopilaris)
Order: Passerine birds (Passeriformes)
Family: Thrushes (Turdidae)
Habitat: krummholz of Northern Europe, wintering places in the Fourseas region.

Picture by Pavel Volkov
Passerine birds represent a relatively young group of birds that preserved
a significant part of their diversity at the level of families and genera
in the human era. Thrushes in Holarctic were characteristic synanthropic birds,
did not lose their diversity in Neocene and are still widespread in Eurasia.
One of the avifauna species of Northern Europe is haltia (the name means Finnish
helper spirit), a descendant of the fieldfare (Turdus pilaris).
Body length of this bird is up to 16 cm, wingspan is up to 35 cm. The appearance
remained characteristic for thrushes: long legs and tail, short wings, pointed
straight beak and a small head. Back and upper side of the wings are brown,
belly and underside of the wings are white. The lower part of the head (chin
and throat) is orange, the upper side of the tail and head are dark brown,
and there is an area of yellow feathers around the eyes. Legs and beak are
gray-blue.
Haltia live in flocks of up to 10-12 individuals, and during the breeding
season they build their nests at a close distance from each other, taking
advantage of collective protection from enemies. Outside of the nesting season,
they fly in a noisy flock through the krummholz, and at night they perch on
a branch or trunk close to each other, and sleep with their sides pressed
closely together to keep warm. In addition, with such grouping of birds, the
predator cannot distinguish separate individuals in the general outlines of
a group of birds, and so they disguise themselves from nocturnal predators
like owls. Haltia prefer to nest in close proximity to the nests of large
birds of prey – for example, the storyunkar hawk.
These creatures do not hunt near the nest, but they drive away predators that
can attack haltias.
This thrush feeds on berries, seeds and insects. The basis of diet is berries,
including of shrubs such as lingonberry, blueberry, cranberry – birds often
feed on the ground in the thickets of these shrubs. Sometimes they catch parasites
in the fur of large mammals. Birds catch insects mainly for feeding offspring.
Haltia itself can become a prey of small birds of prey, predatory mammals
and snakes.
The breeding season is in spring or in early summer in the northern parts
of the range. At this time, birds form breeding pairs and begin to build nests,
preferably near the nests of feathered predators. In such conditions, haltias
nest together with other bird species, safe due to the large number of neighbors.
The female lays 3-5 blue eggs in a cup-shaped nest made of blades of grass,
needles and twigs, and cemented with mud and clay. The litter is made of wool
and bird feathers. The female incubates the clutch, and the male searches
for food for her and occasionally replaces her on the nest. After 2 weeks,
the nestlings hatch. At first, the female warms the nestlings while the male
brings them food, but when the nestlings grow a juvenile downy dress, both
parents begin to participate in feeding the juveniles. At the age of 3 weeks,
young birds begin to fly. In the southern part of the range, birds manage
to rear two broods. Life expectancy is up to 10-12 years.
Haltia winters in the Fourseas region, usually on the southern shore of the
reservoir. Sometimes birds stay to winter in the oak forests of the southern
part of the forest zone of Eastern Europe. During the migration, birds form
numerous flocks, numbering several tens of thousands of individuals.
This bird species was discovered by Mamont, the forum member.
Mustarastas,
or Fennoscandian blackbird (Turdus mustarastas)
Order: Passerine birds (Passeriformes)
Family: Thrushes (Turdidae)
Habitat: coniferous and mixed forests and woodlands of Fennoscandia.

Picture by Pavel Volkov
Thrushes represent a characteristic group of birds of the
northern regions of Europe in Neocene epoch. They survived relatively successfully
in the human era, inhabiting agricultural and forest park landscapes, and retained
a relatively high number and species diversity, which allowed them to maintain
their place in the new ecosystems of Neocene. For the northern European forests,
a characteristic species is mustarastas, or Fennoscandian thrush – a large thrush,
reaching 25-30 centimeters in length and weighing 90-130 grams. Fennoscandian
thrush is one of the most numerous bird species in Fennoscandia. In summer,
it prefers to settle in coniferous and mixed forests and krummholz, especially
with thickets of shrubs with edible berries in the undergrowth or in wetlands
in the neighborhood.
Males of this species are colored entirely black, with a small blue-black crest,
with a yellow-orange beak and with a ring of yellow skin around the eyes. Hence
the name of the species: “mustarastas” – blackbird in Finnish; mustarastas is
a descendant of the blackbird (Turdus merula). Young birds and females are light
brown in color, with a dark blue tail, light throat and belly.
In cold winters, the bird migrates to southern Europe: the Balkans, the Apennines
and the Pyrenees, and some flocks even cross the Mediterranean lowland and even
reach Morocco. In warm winters, the bird may not migrate or migrate to the southern
border of coniferous forest zone.
The mating season begins in mid-spring, when males occupy territories in the
forest and sing actively. The song of the bird is sad flute whistles, and the
cry is a dry crackle and the chirping “tak-tak” and “hicks-hicks”. The male
sings especially actively early in the morning and in the evening before nightfall,
and at this time the songs of this bird species give the forest a special charm.
The nest of this species is cup-shaped, approximately 150-200 mm in diameter,
75-100 mm in height; the diameter of the inner part is 75-110 mm, the depth
is 45-70 mm. Nests can be located at a height of 8 m, on firs or pines, in mixed
forests also on birches and linden trees. The nest is woven of thin twigs and
grass, plaistered with clay from the inside. In spring, molting lagomoropuses
coming from the south rub against gnarly trees, leaving patches of falling out
winter wool on them, and this wool becomes a good material for nest bedding.
The female lays eggs in the second half of May, begins to incubate them at the
end of May. As a rule, in the clutch there are from 4 to 7 pale eggs of bluish-green
color with brownish-purple spots and smears thickening to its blunt end. The
eggs are 25-30 mm long and 20-23 mm wide. Nestlings hatch by mid-June, fledge
and leave the nest a month later. In case of the destruction of the clutch in
the early stages of incubation, the couple makes a second clutch. Juveniles
from the second brood usually leave the nest in July, sometimes in early August.
Sexual maturity in young birds occurs by the following spring.
Mustarastas is an omnivorous bird that feeds on various insects, earthworms,
slugs, seeds and berries. This bird often follows lagomoropuses, feeding on
skin parasites of these large animals, or preys on blood-sucking insects that
bother these beasts. If a bird is looking for food on the ground among the shady
undergrowth, it is almost invisible. On the ground, thrushes search for food,
moving in leaps and at the same time keeping their tails raised, and sometimes
stop to probe the soil with their beaks. They also dig up the ground under the
bushes.
Life expectancy does not exceed 7-10 years.
This bird species was discovered by Wovoka, the forum member.
Rapa
Nui thrush (Turdus leucourus)
Order: Passerine birds (Passeriformes)
Family: Thrushes (Turdidae)
Habitat: Easter Island, forest.

Picture by Pavel Volkov
The human actions during the Holocene caused the collapse
of many ecosystems. Easter Island was one of these cases. This place was completely
deforested and many of its endemic species perished. However, with the arrival
of the Neocene it was able to recover, developing a new endemic fauna. One of
the species living here is the Rapa Nui thrush, a descendant of birds that arrived
on the island from South America.
The Rapa Nui thrush has 23 cm of length and a wingspan of 42 cm. This species
has a brownish-black plumage in the upperparts and wing, while the chest and
belly are creamy. Their tails have white tips. The beak is yellow in the males
and light gray in the females. Both sexes have pale gray legs and feet.
This species is a generalist feeder, principally foraging for invertebrates
but also taking fruits. It tends to swallow fruits whole, but can peck those
that are too large. Activity peaks are at dawn and dusk.
The nesting season of the Rapa Nui thrush occurs during the spring. The male
will find a suitable territory to build a nest and will sing constantly to attract
the female. This bird is a polygamous species, with the male mating with two
females, usually one older than the other. Differently of other polygamous birds,
all of them raise the young. Together, the three birds build an open-cup nest,
usually 4 to 5 meters above ground. Inside it both females will lay two to three
creamy white eggs, with the older female being the first to lay them. During
13 days, the females will share the incubation while the male defends the territory.
The female that isn’t busy incubating, will help the male in his task. Together
they will vocalize in duet to show that the territory is theirs.
The nestlings are attended by the three birds. They fledge within 12 days, but
continue receiving food from the adults for more three weeks. After that they
are expelled from the territory. Once the young leave, the bond between the
adults is broken. However, they will usually come together once again during
the next nesting season. Sexual maturity is reached in 7 months and they have
a lifespan of 8 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
False
spike-headed bird (Pseudornitohystrix inaculeatus)
Order: Passerine birds (Passeriformes)
Family: Flycatchers (Muscicarpidae)
Habitat: south of Indochina, tropical woods.
Occurrence of species of live creatures, able to protect themselves from enemies
actively, results to the occurrence of some more or less successful imitators
at them. So butterflies imitate wasps, flies look like bees, and spiders imitate
ants. In Neocene such tactics of protection had received continuation: the bird
of flycatchers family found in woods of South-Eastern Asia, which dexterously
simulates the porcupine bird (Spinocephalornis
hystricoides), one of species of spike-headed
starlings family living in the same place.
False spike-headed bird is smaller than the porcupine bird: this creature is
about 25 cm long (porcupine bird is up to 30 cm long), but at a passing such
distinction is almost imperceptible. Being disturbed, this bird imitates movements
and aggressive demonstration of long-needled spike-headed starling, uttering
loud chirring cry and shaking head from side to side. But at closely examening
the basic difference becomes visible: instead of spikes on its head only thin
feathers forming crest stick up. Bravery of this bird is supported with nothing:
at the best it can peck the aggressor, but no more that. Such similarity of
the imitator and model represents an example of well-known in nature Bates’s
mimicry.
Except for absence of needles on head, false spike-headed bird differs from
the prototype in details of colouring. On its head there is wider red strip
getting on eyes (practically all top part of head, except for crest, is red),
and the white strip stretches from corners of mouth up to nape. As against the
porcupine bird at false spike-headed bird throat is black. Black colouring also
gets on throat and top part of chest; stomach is grayish-white. Back and wings
are covered with motley pattern, looking approximately like to scaly colouring
of the porcupine bird: on each feather there are some cross light stripes on
dark brown background.
Tail of false spike-headed bird is black, long and narrow. Two middle feathers
are little bit longer than other ones, and their tips are colored white. At
males of this bird tips of tail feathers are expanded – they are used in courtship
demonstrations.
Birds of this species have habit to sit on branches, having raised tail at an
angle to the back. And the disturbed bird starts to throb tail that is not characteristic
for the porcupine bird.
By the way of feeding false spike-headed bird strongly differs from spike-headed
starlings. Similarly to all flycatchers, it is insectivorous bird. Its beak
is pointed, rather thin and straight, and the mouth cut is very wide: it reaches
almost the level of back edge of eye. This bird eats insects and traps them,
motionlessly waiting on branch. The basic catch of false spike-headed bird is
made by large bugs, butterflies and flies which are caught in flight by short
exact throws. But frequently even well armed wasps and bees may occur on its
“dinner table”. False spike-headed bird is not afraid of their stings: it seizes
such insects across the abdomen, presses to branch by toes and pecks, throwing
out sting with poisonous glands.
As all its congeners, false spike-headed bird nests in tree-trunk hollows. Pairs
at this species form to one nesting season and break up, when nestlings mature
and become able to eat independently. Male finds suitable tree-trunk hollow
and sings near it, involving females. Attempts to form a pair at males have
chosen well disguised tree-trunk hollow are most successful.
In clutch there are from 4 up to 6 eggs; female hatches mainly, and male feeds
her, only occasionally replacing the female in nest. Till the posterity rearing
its duty is protection of nesting site from competitors, and also protection
of nest against predators. Till one year this species makes about 4 hatches.
Young birds had left nest approximately at monthly age, have monotonous dark
brown colouring. They will get bright protective colouring only up to the third
month of independent life, and before this event young false spike-headed birds
are very vulnerable for every possible predators.
Ukko,
sparkling waxwing (Bombycilla ignicapilla)
Order: Passerine birds (Passeriformes)
Family: Waxwings (Bombycillidae)
Habitat: forests of northern Eurasia, the Urals, the taiga of Siberia.
Waxwings (Bombycilla garrulus) easily survived the Holocene and human times.
Eating a variety of food and building nests on the edges of the forest, they
did not suffer much from human activity, and even found food in human settlements.
During the ice age, there were enough suitable habitats and food for them. The
common species of European birds is the most common descendant of the Bohemian
waxwing – ukko.
Ukko is similar in size and appearance to its ancestor: body length is 20 cm,
weight is about 62 g. The main color of the plumage is greenish-brown; wings
are darker, with a double white stripe along the edge of the wing coverts. A
large mobile bright yellow crest of feathers grows on the head, which is used
by birds in communication. The color of the crest is shaded by a black “mask”
on the crown and front of the head. When birds move among the trees crowns,
there is an impression of short flashes, which is referred in the name. Ukkos
are flocking birds, there are up to several dozen birds in each flock. From
time to time, the characteristic muffled call “ukko”, accompanied by the movement
of the crest is heared in the forest. Usually this signal is uttered by birds
heading to feed. They feed on berries, fruits and insects, and on the buds of
trees and shrubs in late winter and early spring. In winter, birds migrate from
nesting sites in search of berries. Populations living in the north of the range
– in Scandinavia, Khibiny, and along the tundra border – make real large scale
migrations. In lean years, separate ukko flocks appear even in the groves of
southern Europe and the steppes of Mongolia, competing for berries with local
animals.
Mating occurs in early spring. Birds build nests on the edges of the forest,
fire sites or near reservoirs. Tournaments for a female came down to the display
of the crest and the power of the voice. At this time, the male spreads its
crest like a fan in width and ruffles the head plumage so that it shades the
crest. To lay eggs, the female builds a small nest, which is more often attached
to the thin outer branches of tall trees. As a litter, last year’s grass and
wool are used, which ukkos gather from shedding mammals or from trees against
which they scratched themselves. There are 3-5 eggs in a clutch. The female
incubates the clutch for 13 to 14 days, while the male supplies her with food.
Near the nest, the birds are secretive and cautious. Juveniles leave the nest
at the age of 25 days. Sexual maturity comes at the age of 8 months, life expectancy
up to 14 years.
This bird species was discovered by Nick, the forum member.
Tropical
waxwing (Bombycilla tropicalis)
Order: Passerine birds (Passeriformes)
Family: Waxwings (Bombycillidae)
Habitat: tropical forests of Amazonia.
The Ice Age that occurred during the transition of Holocene to Neocene forced
some species to migrate to southern regions. Certain species, even with the
end of the ice age, remained in their new localities giving rise to new species.
One of these new species was the tropical waxwing, a descendant of the cedar
waxwing (Bombycilla cedrorum). It lives in the Amazon rainforest.
This bird has 17 cm of length and a wingspan of 30 cm. Both sexes are similar
with a brownish-grey plumage and a conspicuous crest on its head. It has a black
mask through the eyes. The lower belly is a rich chestnut color and there are
cinnamon-colored areas around the mask. The primaries and secondaries are red,
being brighter in the males. The tip of their rectrices is orange. The beak
and legs are dark grey.
Tropical waxwings feed mostly on fruits and berries. Insects become an important
part of the diet in the breeding season. Sometimes, these birds will eat fruit
that is overripe and has begun to ferment, intoxicating them.
These birds are sociable, staying in flocks year round. They are non-territorial
birds and will often groom each other. They move from place to place depending
on where they can find good sources of food. Nesting season for this bird begins
during the end of autumn. The male will do a "hopping dance" for the
female. If she is interested, she'll hop back. During courtship the male and
female will sit together and pass small objects back and forth, such as flower
petals or an insect. Mating pairs will sometimes rub their beaks together affectionately.
The nest is a loose open cup built with grass and twigs, lined with softer materials
and supported by a tree branch. Usually 5 or 6 eggs are laid and the female
incubates them for 12 days. The eggs are bluish grey with irregular, dark brown
spots. Both parents build the nest and feed the young. Young leave the nest
about 14 to 18 days after hatching. The tropical waxwing reach sexual maturity
with 1 year and its lifespan is of 12 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Amakuchi
(Florombycilla amakuchi)
Order: Passerine birds (Passeriformes)
Family: Waxwings (Bombycillidae)
Habitat: south of Japan Islands, forests.
In Holocene, representatives of several bird families were more or less adapted
to feeding on flower nectar – for example, hummingbirds, sunbirds and honeyeaters.
Their representatives continued their evolution in Neocene; in addition, new
groups with similar specialization appeared – flowerpeckers
and loreetos in Southeast
Asia and honeybirds in Hawaii. They descend
from finches, loriinae parrots and white-eyes, respectively. In the south of
the Japan Islands, far from the ranges of any nectarivorous birds, a descendant
of one species of Bombycilla genus, widespread in northern Eurasia, adapted
to feeding on nectar. This bird is called amakuchi, which means “sweet tooth”
in Japanese.
There is nothing surprising in the fact that such a species originated from
the waxwings: according to genetic studies, Hawaiian “honeyeaters” of Mohoidae
family, extinct by the end of the XX century, were close relatives of the waxwings.
In its size, amakuchi is slightly inferior to the ancestor, reaching the size
of a sparrow (length up to 16 cm). This bird has a rather short tail and pointed
wings. Paws are short, with tenacious toes; they allow amakuchi to climb deftly
the branches of trees (this bird never descends to the ground). A characteristic
feature of the appearance is a long and thin beak, slightly curved downwards.
The main plumage color of this bird is dark brown. The tail is colored pale
yellow, and there is a wide dark red stripe at the tip of the tail. Primary
feathers are black with a white border, at the tips of the secondary feathers
there are leathery plaques of white color – a kind of “family trait” of the
waxwings. The feathers on the bird’s head and neck have a reddish tint. On the
throat there is a wide “necktie” of black color with a white stripe along the
edge. A lush crest grows above the beak, which can fold and open. Eyes of this
bird are brown, beak and legs are dark gray. Male and female are outwardly indistinguishable.
Amakuchi’s voice vaguely resembles the voice of a waxwing, but the trills are
sharper and shorter.
The basis of amakuchi’s nutrition is flower nectar. They eat it while percking
on a branch next to the flower and dipping the beak into its corolla (they cannot
hover over the flower in flight, like hummingbirds and some sunbirds). An adaptation
for nectar feeding is a long tongue with a corneous brush at the tip. Amakuchis
are not specialized in feeding on certain plant species and visit a variety
of flower species. At the same time, they make a significant contribution to
their pollination.
In addition to nectar, these birds eat juicy fruits and berries: small ones
are swallowed whole and large ones are pecked and their juice is sucked out.
An indispensable element of nutrition (in particular, a source of protein) is
small insects, which amakuchis gather on flowers or catch from the perch, like
Old World flycatchers.
Outside of the breeding season, amakuchi lives in small flocks (up to 15 individuals),
roaming the forests within a limited area. In the middle of spring, the breeding
season begins for these birds and the flocks break up into separate pairs. The
male attracts the female by singing, as well as by a special courtsship dance
combining a fluttering flight and jumping on a branch. Mates build a large flat
nest in the crowns of tall trees using plant materials like twigs, grass and
lichen, with a small addition of down and wool. There are 3 to 6 blue eggs in
the clutch, which the female incubates (the male brings her food at this time).
After about 2 weeks, the nestlings hatch. The male and the female feed them
with a semidigested mixture of insects and nectar (the older the juveniles get,
the greater the proportion of nectar becomes). After 18-20 days, young birds
leave the nest. Soon after, the parent pair starts breeding for the second time.
After the second brood becomes independent, birds from different pairs unite
in groups and begin migrating. At the same time, birds from the north of the
range migrate to the south. Sometimes the wind brings amakuchi flocks far from
home – for example, to the Korean Peninsula.
The enemies of these birds are various birds of prey and carnivorous mammals,
as well as snakes. The maximum lifespan of amakuchi is no more than 12 years.
A similar species, lesser
amakuchi (Florobombycilla nanus), lives on the Kyushu Islands. It is
characterized by a small size (body length is up to 13 cm) and a darker color.
This bird species was discovered by Simon, the forum member.
Asian
tree-creeper (Nechlorus dendrobatoides)
Order: Passerine birds (Passeriformes)
Family: Fairy-bluebirds (Irenidae)
Habitat: southern China, Southeast Asia.

Picture by Alexander Smyslov
Neocene is the time of restoration of tropical forests, the
most productive natural communities of land. Degraded in great degree in human
epoch, forests have covered again the vast areas of land, and they are inhabited
by various creatures. The initial fauna had strongly suffered from destruction
of habitats, and tropical forests of early Neocene had been occupied by species
settled from other ecosystems. During the process of establishing of natural
balance evolution had produced the diverse forms of animals, birds and reptiles
which quite often may seem strange and unusual. The bird Asian tree-creeper
is one of them.
Tree-creeper is rather small bird: its length reaches 20 – 23 cm including tail.
This bird has monophonic, dim and dark plumage of blue-green color which makes
it imperceptible among various epiphytes, so plentiful in Asian jungle. The
most unusual feature of this bird is its body shape and a habit of life: tree-creeper
has almost completely lost the ability to fly, having kept rather small wings
and strongly reduced keel bone. It is rather unusual to forest bird, but in
rich forest canopy this species prefers to move a different way. During the
evolution at Asian tree-creeper very strong and tenacious legs had developed;
by means of them tree-creepers climb and jump from one branch to another and
even from tree to tree, especially if trees grow close enough. Besides rather
light weight and remained wings permit tree-creeper to glide from the top downward,
but the bird makes it reluctantly, preferring to use wings as signal flags at
courting for females or during tournaments against contenders. On inner side
of wings there are bright red spots, appreciable only when the bird opens wings.
As well as other tropical birds of small size, Asian tree-creepers eat small
invertebrates, and also soft fruits and sometimes nectar – everything that is
possible to find in tropical forest canopy in plenty. Therefore birds are rather
tolerant relatively to neighbours and in crones of large enough trees up to
6 breeding couples of these birds may live. Each couple, however, has strictly
protected territory which borders are observed. Displaying signals to neighbours,
bird stands on tiptoes and trembles wings, making visible at look from the side
their bright inner side. Though Asian tree-creepers can not fly, their constitution,
resembling an anatomy of nuthatches (Sitta), apermits them to move on trees
very quickly, hunting for prey or escaping from enemies – predatory mammals,
snakes and birds.
Asian tree-creeper starts to hatch eggs in second half of dry season, and to
the beginning of rainseason from eggs nestlings covered with rare yellowish-green
down hatch. The cup-like nest of these birds is constructed on thin branches
bound by rods and fibres. In clutch it may be up to 4 eggs with speckled brown
shell. Eggs are hatching only by the female, but nestlings are brought up by
both parents. They leave nestlings alone for a long time, and at this time nestlings
lay in nest, not moving – their camouflage is their only protection. At the
age of 2 months young birds leave nest. They differ from adult birds in colouring
– the top part of their body is coloured cryptic greyish-brown with dark speckles;
the bottom part of body is green. After the first shed they get colouring of
adult individuals and become capable to breeding at the age of 4 months.
In places with long-lasting rainseason Asian tree-creepers have time to bring
up two hatches within one year, and then have a rest during several months.
Life expectancy of Asian tree-creeper is about 7-8 years.
This species of birds was discovered by Bhut, the forum member.
Goldenback
crownhead bird (Umbellornis aurodorsalis)
Order: Passerine birds (Passeriformes)
Family: Umbellate birds (Umbellornitidae)
Habitat: Central Africa, flooded forests of Congo basin.

Picture by FanboyPhilosopher
At the ice age the area of tropical woods all over the world
had considerably reduced: the climate began drier because huge amounts of water
appeared accumulated at poles of planet as ice. At this time the variability
of species of wood birds had sharply decreased, but inhabitants of savannas
became more numerous and various. When the situation was stabilized, the climate
became more damp and warm, and the area of tropical woods began to increase
promptly, some inhabitants of savannas had adapted to life in forest. Among
immigrants there were birds, representatives of weaverbirds family (Ploceidae)
which became ancestors of new family of forest birds – crownhead birds, or umbellate
birds (Umbellornitidae). This family is presented by rather small birds – by
size from sparrow up to daw. Their characteristic attribute is hood on head
top looking like crown (it is especially advanced at males, at females can be
absent). The feathers making it have long core on which barbs grow only at top
(in any case make top two thirds of length of feather). These feathers stick
up to sides as an umbrella, but at excitation of bird they rise almost vertically,
as if a crown. The length of them at times exceeds length of head and neck of
bird. At various emotional conditions of bird such hood can open or close up.
Movements of hood are actively used by males during courtship games. At many
species males gather for courtship games at well appreciable branch or on trunk
of fallen tree by groups of 10 birds and more. Courtship demonstrations are
accompanied by loud melodious warbles and separate resonant cries. After pairing
pair of birds in common builds wattled hanging nest; at one species closely
related genus nests are large and collective, renewing and extending annually.
Representatives of family basically eat tree seeds, some eat insects with firm
covers.
Goldenback crownhead bird is rather large species in family: length of body
including tail is about 18 cm. Constitution of bird is rather compact; neck
and tail are short. Wings are moderately long, with pointed ends. Bird flies
well and is able to make sharp turns in air and to change direction of movement.
Goldenback crownhead bird eats seeds of trees, splitting them by strong short
beak. It feeds nestlings by insects and semi-digested seeds.
Colouring of goldenback crownhead bird combines cryptic colors with bright spots
used at courtship rituals. Body of bird is colored grayish-green with brown
primary feathers. Chest of bird is colored white, but tips of feathers are black;
that’s why common colouring looks as “speckled”. On head around of eyes there
are white “glasses” of naked skin. Feathers of hood are black-and-white: the
basic colouring of feathers is black, at top there is white wedge-shaped spot
(at females hoods are made by short brownish feathers). Main ornament of bird
is feathering of back, in rest closed by wings. At courtship ritual bird opens
and lowers wings, having fluffed up feathers on back. Then they become appreciable
from afar - feathers of back at males are painted golden-yellow color with appreciable
metal shine (at females they are simply brightly-yellow).
In nesting season males search for trees which stand at some distance from each
others, and where forest canopy between them is more friable. Each male occupies
branch on which sunlight falls, and starts to involve females, making courtship
dance. If the male has chosen an advantage-ground, the sunlight falls on his
back and makes this bird appreciable from afar. At one lek up to one and half
tens males gather. Courtship games in mass pass up to three times per year though
in intervals between “peaks” of nesting in forest it is possible to see separate
displaying males.
Females from afar notice bright “grooms”, and near lek at times twice more females,
than males gather. At this species polygamy exists “legally”, and sometimes
male simultaneously looks after three nests, replacing hatching females by turns.
Having coupled with one female, male begins nest building, from time to time
flying to lek to form new pair with other female, and sometimes with third one.
He forwards all females to the territory where birds together choose place for
the next nest. Flying “by circle” all females at the territory, male participates
in construction of their nests. Nest of this species represents wattled construction
with long hanging “corridor”, suspended on branch. As building material long
fibrous leaves of graminoids are using; birds gather them on bogs and river
banks. Birds tear apart leaves to lengthwise strips and drag them to the basis
of nest in beak. Some individuals do not squeamish “piracy”, taking away ready
building materials at neighbours, or ravaging and untwining nests of water birds.
After egg laying (up to 4 - 5 eggs in one nest) and the beginning of incubating
females serially are fed up and replaced at the nest by male. For the male it
very favorable time: his bright colouring is not visible, when he sits into
the nest. At this time female eats and cleans feathers. In time free from hatching
the male declares rights to territory: sitting on separately sticking up branch,
he songs loud warble similar to cricket chirr which comes to an end with melodious
“flourish”.
The incubating lasts 17 days, nestlings hatch blind, covered by down only on
back and head. At the age of one week they start to fledge, at 10-days age open
eyes, and at monthly age abandon nest. Parents finish feeding fledglings after
nest leaving two more weeks, and thus sometimes near hatches at the territory
of the same male can unite. Young birds by colouring are similar to female,
but feathers of hood at them are absent. Usually they migrate in wood, having
united in flocks. Puberty at males comes at one-year-old age, at females two
- three months earlier. Thus they leave “youthful” flocks earlier, forming pairs
with older mature males. Young males at approach of sexual maturity form own
leks at which bachelor flights break up. Life expectancy at this species is
about 5 - 6 years.
Close species of crownhead birds live in forests of Equatorial Africa:
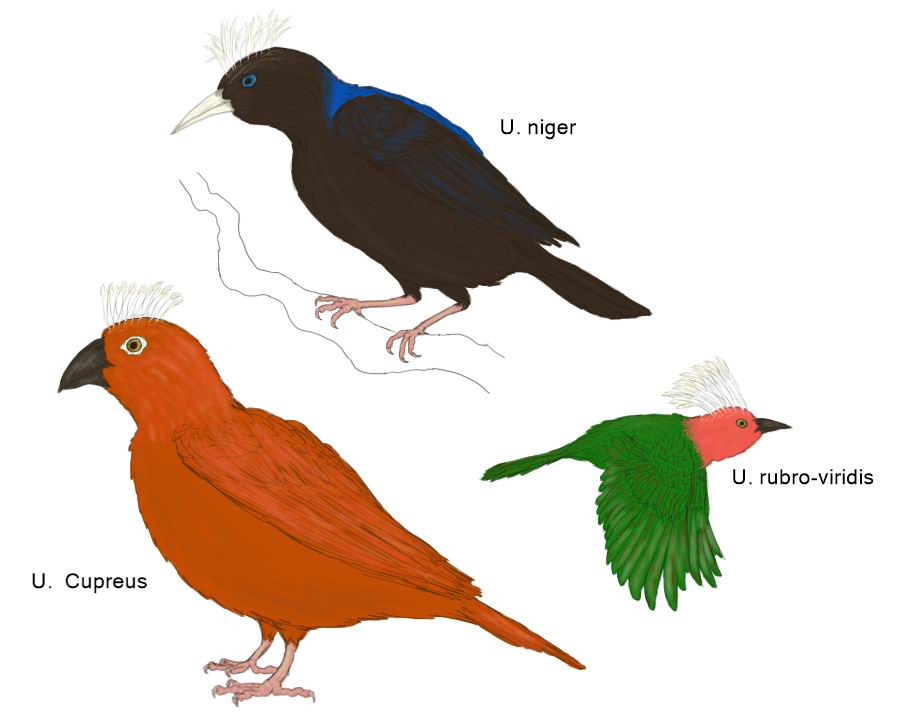
Picture by FanboyPhilosopher
Red-headed
crownhead bird (Umbellornis rubro-viridis) is rather small bird (like
finch by size). Males are colored brightly and contrast: head and neck are coral-red,
body, tail and wings are green with brown speckles. Females are entirely colored
green. Hood on head at the male is white; at the female it is green. Feathers
making it are very long; at them the vane amounts only about third of feather
length. Beak is moderately long; bird eats soft fruits with small seeds. It
is monodin; at the lek it gathers no more, than by ten birds. Nest is making
of long leaves of epiphytic plants and thin stalks of lianas; birds hang it
from below in forked crown of branches.
Copper
crownhead bird (Umbellornis cupreus) is medium-sized bird (pigeon-sized
one). The male is colored rather brightly: all his feathering is brown-red with
bright metal shine on head, wings and tail. Feathers of “crown” of the male
are white (vane makes all about 1/5 of feather length), around of eye there
is a ring of white naked skin – the male uses these contrast stains during courtship
demonstrations. Female is colored brown with dark spots and cross strips, without
metal shine. “Crown” of females is also brown; feathers making it are very short.
This species eats firm seeds of trees, therefore beak of copper crownhead birds
is very short and thick. Displaying takes place in branches of highest trees
of rainforest. In displaying time male involves females with shine of feathering,
and at closer “acquaintance” shows white “glasses” and “crown”. Usually it is
monodin, less often polydin: male participates in nesting no more, than with
two females, but approximately in half of cases (especially at lack of forage)
it forms pair only with one female. Nest is very original: it is the long “sleeve”
attached to branch from below and opening from the side of the end of branch.
Black crownhead
bird (Umbellornis niger) is bird the small pigeon-sized bird. Male
and female, as well as at the previous species, strongly differ in colouring.
Male is black with bluish metal shine on wings and bright blue feathers on back.
Irises at him are also blue, and beak is white. Feathers of “crown” at him are
very elastic and narrow, colored white. Female is colored brown with rust-colored
stomach; its iris of the eye is yellow; “crown” at her is not present. Beak
is direct and moderately long: this species eats soft fruits of palms, unripe
seeds of graminoids and insects. It lives at northern border of tropical forests
and meets even in woody parts of savanna. For nest building it splits to fibers
leaves of fan palms, and weaves these strips to spherical nest directly on petiole
of leaf, strengthening them with dry grass.
Umbellate
harlequin bird (Scurrulornis rubrocephalus)
Order: Passerine birds (Passeriformes)
Family: Umbellate birds (Umbellornitidae)
Habitat: Central Africa, flooding forests in Congo basin.

Picture by Tony Johnes
This species is the distant relative of goldenback
crownhead bird distinguished from it by features of anatomy and behaviour.
This bird is considerably smaller: it is sparrow-sized bird. The basic colouring
of body is black; on head of male there is mobile orange-red hood. Feathers
of hood have vane only at the top third of length. At females hood is shorter
and colored dark cherry. Around of eyes at birds of both sexes there are white
rings of naked skin. Under wings at males there are bright red stains. Colouring
of the adult male resembles motley scrappy suit of Harlequin – bird is named
because of it.
This bird eats mainly insects though it can eat small seeds of various trees.
In this connection beak of this species is thinner than at related species.
As against goldenback colleague, umbellate harlequin bird is only monodin. However
it does not avoid society of relatives though it is shown a little differently:
birds of this species build huge collective nest, occupying the chosen tree
for many years. The nest is building and renewing by members of colony practically
all year round. It is shared to set of “apartments” in which nesting pairs of
birds live. Breeding pairs at these birds formed to one nesting season, and
the nest is “inherited” by the male. It renews nest and performs near it ritual
of female attraction. For this purpose the male seizes by claws edge of nest
and hangs on it headfirst, having opened wings so, that red colouring of sides
becomes visible. Thus it chirrs attractively for females. If the female is ready
to form pair with him, she simply flies into the nest, and male gets after her.
In clutch of the harlequin bird there are 5 - 7 eggs, both birds hatch it alternately
within 14 days, and then feed nestlings together. At the age of 5 weeks young
birds leave nest then parents finish feeding them about one week. When fledglings
abandon parents, pair breaks, and the male has a rest and prepares nest for
new nesting cycle. In total during one year in one nest up to 4 hatches can
be made. The young bird becomes sexual matured at the age of 4 months.
Efficiency of breeding at birds of this species is increased due to features
of behavior: collective protection is applied to defense of nests at harlequin
birds. Having found the enemy – snake, large lizard or small predatory mammal,
birds swoop down on it by all flight, driving predator away from nest.
Despite of such features of behaviour allowing effective surviving, life expectancy
at this bird it is very short – no more than 3 years. It is common feature of
all animals having the small size of body.
Sea,
or green-faced weaverbird (Chlorochrysornis chlorops)
Order: Passerine birds (Passeriformes)
Family: Waxbills (Estrildidae)
Habitat: mangrove thickets at the coast of Eyre Gulf.

Picture by Pavel Volkov
In Neocene at the territory of Meganesia (the continent uniting
Australia and New Guinea) the group of passerine birds of waxbills family, Meganesian
weaverbirds, is widely settled. These birds are rather large in comparison with
their ancestors: the majority of species is larger than a sparrow, and some
surpass starling in size. They inhabit woodlands, mountain and rainforests of
continent. But among these birds there is one species strongly distinguished
by habit of life from other Meganesian weaverbirds. It lives in marshy and nearwater
biotops, and separate colonies meet even in thickets of Pandanus (screw palm)
on shoalinesses of Eyre Gulf having no any connection with coast. It is sea,
or green-faced weaverbird.
This species is rather large, starling-sized bird. Green-faced weaverbird differs
in very bright colouring – background color of bird feathering is dark yellow
with brownish shade (“honey”), and primary feathers have beige colouring. On
this background bright green with metal shine “mask” on head looks as a contrast
spot. At females it takes only forward part of head and partly stretches on
forehead, but at males the most part of head may have green color, and only
the top of head and nape keep background colouring.
At these birds there is straight and strong conic-shaped beak. Sea weaverbird
is unpretentious in meal and eats practically any kind of food. It pecks infructescences
of screw palms, cracks their seeds, and eats seeds of some other plants living
in mangrove thickets of Eyre Gulf. This bird willingly eats forages of animal
origin: sea weaverbirds frequently peck dead fish, molluscs and crustaceans.
There is one more interesting feature of behaviour of these birds - they had
learned to rob a web of mangrove
fishing spider living on the bottom part of mangrove trunks.
The nest of this bird differs in inaccessibility and is reliably protected from
predators. Sea weaverbird plaits nest in the bases of leaves of pandanus on
which edge long thorns grow. Bird braids the bases of several near growing leaves
with long plant fibres, and then plaits them together by shorter fibres. The
entrance to the nest represents the long “sleeve” hanging down. Thorns on leaves
of pandanus provide more durable attaching of nests to the plant, and make them
inaccessible to predators. These birds nest in colonies, and frequently one
trunk of pandanus forms the basis for the true “city” of sea weaverbirds numbering
some tens of nests.
Pairs at these birds form to one breeding cycle. Male involves the female, uttering
modulating advertisement call near the nest. When near the nest the female appears,
he begins courtship displays: male fluffs green feathers on head, displaying
their shine in sunlight. He hangs on leaves near the nest, trembling by wings
and having stretched tail. If the nest is acceptable in quality and protecting,
the female gets into it, and male follows her.
In clutch of this species there are about 6 eggs with greenish shell. The incubation
lasts till about 12 days, both birds hatch eggs alternately. Nestlings develop
quickly, and already at the age of 4 weeks leave the nest. Within one year these
birds may rear three hatches. The half year old bird already can nest. Life
expectancy of sea weaverbird does not exceed three years.
Sleeve
weaverbird (Manuleornis pandanophilus)
Order: Passerine birds (Passeriformes)
Family: Waxbills (Estrildidae)
Habitat: Meganesia, thickets of pandanus along the coast of reservoirs and at
the islands of Eyre Gulf.

Picture by Pavel Volkov
Eyre Gulf in southern part of Meganesia represents the extensive
shallow reservoir connected to the ocean and having changing level of water
salinity. It is shallow and is well warmed by sun. In some places above the
water surface the small islets rise; they are overgrown with pandanus and represent
the analogues of mangrove thickets at the coast of oceans. They do not contact
to land, and in them the community of insects, spiders,
fishes and birds is formed. One of the birds nesting here is the sleeve weaverbird.
This bird has received the strange name for very characteristic form of nest
– it is plaited of grass and fibres, long, resembling a sleeve, and attached
along the leaf of pandanus on its bottom side. The nest chamber is between leaves
of plant and is well protected from enemies like birds or snakes by the prickles
sticking out from edges of the nearest leaves. It is possible to penetrate into
the nest only through the narrow entrance, opened close to the tip of leaf.
As the leaf is spiny from sides and from below along the middle nerve, the entrance
also appears protected. Moreover, only the dexterous bird can get in it when
the leaf waves on wind.
Sleeve weaverbird is the representative of waxbills, group numerous in Meganesia;
it’s a bird of tiny size and bright colouring. Wingspan of this weaverbird does
not exceed 15 cm. They have rounded tips and permit this bird to maneuver dexterously
in thickets of pandanus forest. Thick conic beak helps to crush the core of
pandanus seeds and insects with strong covers. In outflow sleeve weaverbirds
move down to the bottom part of pandanus trunks and search for the snails stayed
after outflow, and for young crabs.
The plumage on back, head and wings of sleeve weaverbird has grass-green color.
Sides of head, throat, breast and stomach are straw-coloured. On back there
is site of silver-grey feathers. On background of plumage the beak of bright
red color is clearly visible. Near mouth corners there are small sites of the
featherless skin of white color; at male they are larger, rather than at female.
Paws are grey. The female differs from male in thinner beak and greyish shade
of plumage on stomach.
Birds of this species nest in small groups. At the same pandanus tree up to
ten nests may be arranged, poorly appreciable at sight from the side. Single
and young birds keep in flocks of some tens birds. They can make flights between
different islets of vegetation, easily overcoming distance of some kilometers
above the sea. The birds having a nest lead a settled way of life.
Nesting occurs irregularly, 2 – 3 times per one year. Male starts to prepare
for nesting, occupying a site in tree crone. On the chosen leaf of pandanus
bird starts to braid its bottom side with fibres, hooking them on prickles growing
along the edge. At the initial stage of building the nest represents deep cup
in the basis of leaf. Male gradually builds on an entrance, transforming it
to tube. It interrupts work for singing, uttering series of monotonous trills,
perching on leaf near the nest. If the female accepts its court and also likes
a place for nest, birds complete it in common. The female exclusively makes
a litter for the nest.
In clutch of these birds there are about five eggs; the female hatches only,
but male feeds her regularly. The incubation lasts till about 12 days. Male
and female in common feed nestlings during three weeks. Young birds keep together
for some time, searching for flock of congeners. After nestlings leave the nest,
the parental couple also breaks up and adult birds join one of flocks of congeners,
flying sometimes far from place of former nesting.
The young bird begins nesting at the age of half-year. Life expectancy of adult
birds does not exceed 4 years.
Red-faced
waxbill (Estrilda rubrorostris)
Order: Passerine birds (Passeriformes)
Family: Waxbills (Estrildidae)
Habitat: plains of central South America.

Picture by Pavel Volkov
During the Holocene, man introduced species in other continents.
Some of these introduced species were capable of surviving in their new habitats
and give rise to descendants. The red-faced waxbill is one of these cases, a
descendant of the common waxbill (Estrilda astrild). This bird lives in the
plains of Central South America.
This bird is small, with 12 cm of length. It has a slender body with short rounded
wings and a long graduated (darker on its tip) tail. The adults have a bright
red bill. Their plumage is mostly grey-brown, finely barred with dark brown.
In both sexes their head is red. They have a reddish stripe along the center
of the belly. Females are similar to the males but are paler with less red on
the belly and a dull red. Juveniles are duller with little or no red on the
belly and head, fainter dark barring and a black bill, until they reach sexual
maturity with 10 months. The red-faced waxbill has a variety of twittering and
buzzing calls and a distinctive high-pitched flight-call. The simple song is
harsh and nasal and descends on the last note.
Their diet consists mainly of grass seeds and insects are also eaten on occasions,
which are eaten in great quantities during the breeding season when more protein
is needed. The waxbills typically forage in flocks which may contain hundreds
or even thousands of birds. They usually feed by clinging to the stems with
their long, spindly claws and picking from the flower heads but they will also
search for fallen seeds on the ground. They need to drink regularly as the seeds
contain little water.
The nest is a large ball of grass stems with a long downward-pointing entrance
tube on one side. It is built in a tree and being a species that forms large
flocks, a tree can end up being filled with several nests. A rudimentary second
nest may be built on top where the male sleeps. Four to seven white eggs are
laid. They are incubated for 11 to 13 days and the young birds fledge 17 to
21 days after hatching. Both parents take part in incubating the eggs and feeding
the chicks. The juveniles are independent for 26 days. The lifespan of this
species is 4 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Snail-eating
or New Zealand accentor (Prunella helicivora)
Order: Passerine birds (Passeriformes)
Family: Accentors (Prunellidae)
Habitat: New Zealand, forests along riverbanks on plains and in mountains.

Picture by Pavel Volkov
Accentors are small songbirds, characteristic mainly of the
forest and mountainous regions of Eurasia. They were not greatly affected by
human activity in Holocene, as they often fed near human settlements and could
live in a landscape altered by humans. The ice age at the boundary of Holocene
and Neocene, which fragmented continuous woodlands into isolated areas, dealt
a much stronger blow to the accentors. Populations of some species have declined;
some of them have become extinct. In Neocene, accentors are still the common
species of the avifauna of the Eurasian forests and mountains. Only the fate
of the dunnock (Prunella modularis), introduced by people to New Zealand, has
developed in a special way. Here these birds survived after the end of the human
era, and the population of their descendants diverged into two species. One
of them is a snail-eating accentor, which lives in humid forests and along the
banks of rivers of the archipelago.
The body length is up to 18 cm along with the tail. Beak is massive, cone-shaped
and pale yellow in color, resembling the beaks of finches. The color of the
male’s plumage is brownish-black with a reddish tinge; the throat spot, primary
feathers and tail are black, on the wing coverts there are wide black edges
forming a scaly pattern. Females are brown with darker mottling. These birds
keep solitarily, except for periods of breeding and caring for nestlings. The
snail-eating accentor feeds mainly on snails and slugs, gathering them on trees
and in the forest floor and easily “cracking” their shells with a strong beak.
Occasionally it eats freshwater snails in shallow river waters and looks for
hard-shelled beetles in the forest litter. It is the only New Zealand bird capable
of eating a garlic
snail (Scorodonodora foetidissima) without harm to health.
The breeding season is long – from the end of spring and the whole summer. Snail-eating
accentor grows 2 broods per season. Males attract females by singing, which
sounds like a long, quiet, murmuring trill. Most often it turns out that 2 males
form a bond with 3 females and jointly guard the breeding territory. The nest
is arranged low above the ground, at a height of 0.5-3.5 m (usually 1-2 m) above
the ground, it is built only by the female. It is usually tightly woven of thin
twigs, roots and other thin and flexible building material. The clutch consists
of 4-5 eggs of a grayish-green color. The duration of incubation (mainly by
the female) is 14 days. The male hatches only when the female flies out for
feeding. While the female is hatching, the male feeds her and guards the nest.
Both parents take care of the brood. The juveniles stay in the nest for 11-14
days. Young birds of the first brood in the season stay on the parent territory
and help feed younger siblings from the second brood. At the age of 2 months,
snail-eating accentors are completely independent; they reach sexual maturity
at the age of one year. Life expectancy is no more than 10 years.
This bird species was discovered by Nick, the forum member.
Antipodean,
or bush accentor (Prunella antipodorum)
Order: Passerine birds (Passeriformes)
Family: Accentors (Prunellidae)
Habitat: New Zealand, forests on the plains.
Accentors, unlike many larger birds, survived the human era relatively easily.
Their unpretentiousness and ability to live next to people helped the most part
of their species to retain, and in Neocene they are one of the characteristic
components of the avifauna of Eurasia. The dunnock (Prunella modularis) was
introduced to New Zealand in the historical era. The ice age of the end of Holocene
proved to be a more serious test for these birds, as the area of forests decreased,
and bird ranges were fragmented into isolated areas. But the accentors survived
this glaciation as easily as the previous ones, and restored their diversity.
The New Zealand population diverged into two species of birds that easily get
along with each other and their competitors. One of them is antipodean, or bush
accentor.
It is smaller than its relative, the snail-eating accentor – the body length
is only 13 cm, and the weight is up to 19 g. It is a dim gray-brown bird with
a thin brown beak and a short tail. Sexual dimorphism, unlike a related species,
is not pronounced: head and body are colored gray with mottling on the forehead
and cheeks, wings have a brown scaly pattern; primary feathers, back and tail
are brown. Like its ancestor, it catches its prey mainly in bushes and undergrowth,
without flying into the tree crowns. The bird feeds on worms, spiders, insects
with not very hard cover – grasshoppers, ants, dipterans, butterflies, small
beetles, larvae and caterpillars. It lives in small colonies of 5-6 pairs, building
a common nest in the bush thickets.
These birds keep in pairs when searching for food, but the colony is protected
by all the individuals being there. The allert signal of birds sounds like separate
clicks “kluk... kluk...”, on approach to the nest, birds utter a series of three
clicks in a row. The alarm signal is a loud chatter, repeated many times in
short series. Bush accentors especially often mob with such signals birds of
prey, including ruacapangi and eagle
kea, and then other forest inhabitants join them in it.
The breeding season is long – from mid-October to January-February. In each
colony, the breeding cycles of its members are synchronized. As a rule, males
and females in colonies are connected by polygamous and polyandrous relationships.
Males present a symbolic gift to the female: a young or widowed bird brings
an edible prey, a member of the breeding pair carries a twig that is woven into
the common nest. During the season, 2 (on the Southern Island) or 3 (on the
Northern Island) clutches are made. Nest based on a common foundation of twigs
includes several sections separated by loose walls. Eggshells are light green;
there is up to 5 eggs in a clutch. The female incubates independently and almost
does not leave the clutch for 12-13 days. Nestlings are fed by both parents;
males often feed the rest of the incubating females. Already at the age of about
12 days, the juveniles begin to fly independently. At the age of one year, they
are sexually mature and the young birds of first broods have time to build a
nest and make one or two clutches. Life expectancy is up to 7 years.
This bird species was discovered by Nick, the forum member.
Architector
weaver (Heteropasser tectorius)
Order: Passerine birds (Passeriformes)
Family: Old World sparrows (Passeridae)
Habitat: savannas of Meganesia.
In human epoch the fauna of Australia, developed in isolation till millions
of years, has undergone the significant changes. People had introduced to this
continent the whole set of species of animals and plants from other continents,
and had broken the natural balance developed in the natural way. Due to nature
protection actions and flashes of infectious deseases number of placental predators
was so reduced, that their populations had died away. People had never managed
to cope with other species. House sparrows (Passer domesticus) belong to the
number of introduced species which have gone through human epoch. They were
successfully settled across the whole continent, and their descendants had taken
the place in ecosystems of Neocene epoch.
The southern part of Meganesia to the north and the west from Eyre Gulf represents
the savanna. In some places in extensive grassy plains high trees are scattered,
and on some of them the huge nests are arranged; these nests resemble nests
of sociable weavers (Philetairus socius) inhabited Africa in human epoch. They
are owned by descendants of house sparrow, the small birds settling in numerous
colonies. This species of birds is named architector weaver. Such building behaviour
and sociality are quite natural continuation of development of behavioural features
natural for ancestor of this species. But they had received very unusual continuation
at Neocene descendants of sparrows.
Architector weaver, sparrow-sized bird, has much in common with the ancestor
in shape. It has peaked wings of fast flyer and short tail – this bird has fast
and maneuverable flight; at an attack of flying predator it is capable to evase
from it, sharply changing direction of movement. Thick conic beak permits architector
weaver to use effectively the kind of food most widespread in savanna – seeds
of grasses of various stages of ripeness. Colouring of this species is soft,
but very recognizeable: brown back, white primarily feathers and tail, brownish-yellow
plumage on breast and stomach. Beak has bright red colouring at male and cherry
colouring at female. The forehead and feathers around of eyes have white colouring
which makes red colouring of beak better seen from apart. For this species of
birds it is vital to be appreciable for other birds with which it nests in common.
Architector weaver has a great skill in nest building. Its huge colonial nest
is made of grass, and all adult birds of the colony work simultaneously to construct
it. Construction of nest begins at once from the several sides, and the ready
construction represents the irregular-shaped structure firmly attached to large
branches in tree crone and separated to numerous nest chambers. In these chambers
various species of birds settle – in colony of architector weaver small parrots,
pigeons and weaverbirds of other species find a place for nesting. But in colony
of architector weaver two more closely related species of birds are always present,
and they descend from the same common ancestor, as architector weaver – from
house sparrow. Their mutual relation resembles more the relations of different
castes in ant colony.
Warrior
weaver (Heteropasser bellicosus) is larger bird with sharply expressed
sexual dimorphism: male is starling-sized bird, female is little bit larger
than the largest male of architector weaver. These birds are remarkable in their
powerful beak with several pairs of sharp triangular denses on lower jaw. Warrior
weavers have bright colouring: bright blue breast, black back, wings and nape,
white “face” and orange-red beak. This bird differs in aggressive behaviour
and is capable to attack the medium-sized mammals coming too close to nest in
which it lives. Attacking birds splash liquid dung into the enemy, beat it by
wings, tear out pieces of wool, and also put deep bleeding bites in head and
ears by beak. This species expresses flesh-eating inclination in diet and frequently
eats food rich in protein: large insects and seeds of plants of leguminous family.
Also at an opportunity this species willingly eats carrion, the rests of prey
of large local predators, and attacks small vertebrates – lizards and snakes.
Brooder
weaver (Heteropasser maya) is a species on which the survival of colony
depends completely. It is the modestly colored bird a little bit smaller than
sparrow. The basic colouring of plumage is brown with dark scaly pattern on
back. Tail feathers have white tips. Around of eyes and on forehead white feathers
form “mask” figure. Beak is rather thin and black in color. This species eats
mainly insects.
These three species of birds evolved together, adapting to each other, and have
shared with each other duties in maintenance of life in the colony. Architector
weaver builds nests and constantly repairs them, but it has completely lost
parental instinct. Also warrior weaver protecting a colony from predators also
has lost it. And brooder weaver raises not only its own posterity, but also
posterity of two other species which became its nest parasites. The parental
instinct at this species is so expressed, what even males take part actively
in incubation of eggs and rearing of posterity. Brooder weavers are monogamous
ones: their family is kept during the time of posterity rearing; at two other
species couples are formed only for the period of pairing and laying of eggs.
The ratio of birds in colony is approximately constant: 40 – 50% of brooder
weavers and 25 – 30% of each other species (warrior and architector weavers).
In areas of favorable climate the colony is used all-the-year-round, and in
it constantly there are eggs and nestlings of various ages. During the season
favorable for nesting brooder weavers have time to bring up two hatches of nestlings
of their own and another’s species. In places where all-the-year-round nesting
is possible, the number of hatches may reach four in one year. In areas of seasonal
drought the multispecific colony leaves nest and migrates in savanna; at this
time birds of all species keep together. Such migrating flock occupies an empty
collective nest to the beginning of nesting season, or quickly builds new one.
Usually the colonies from areas of seasonal climate are not as numerous, as
settled colony. New colonies are easily formed by division of old one: when
the colony reaches too large number, the part of it is separated and moves to
another tree. Less often the flock is formed by birds of one species – as a
rule, it is a result of disbalance of number of species, and the stress because
of aggravation of competition following it. Then the unispecific flock may join
already existing colonies, or gathers missing representatives of symbiotic species,
yet will reach a normal ratio of number of species.
These species of birds were discovered by Simon, the forum member.
Pueblo
sparrow, or building sparrow (Tectopasser aridophilus)
Order: Passerine birds (Passeriformes)
Family: Old World sparrows (Passeridae)
Habitat: cactus forests in North American deserts.
In Holocene, thanks to man, the house sparrow (Passer domesticus) settled almost
worldwide. In Neocene, numerous descendants of this species appeared in the
new homeland of their ancestor, including those distinguished by peculiar specializations.
In the deserts of North America in Neocene, building sparrow, or Pueblo sparrow
lives.
This species has become somewhat larger compared to its ancestor: the body length
is about 17-20 cm; the wingspan is up to 25-30 cm. The building sparrow has
rather long legs and tail, a strong build, large head and very strong thick
beak. Sexual dimorphism is expressed: the male is larger and colored differently
than the female. Back, wings, upper part of the tail and head are light brown;
the female has a lot of mottling, and the male has reddish-brown feathers on
the wing coverts, as well as a red nape. Legs are bright pink, and beak is dark
gray in both sexes.
Building sparrows prefer to live in dry and hot deserts, inhabiting cactus forests
formed of cacti taller than 5 meters. In such thickets there are always lots
of shelters and food is available. This feathered creature is very social and
lives in flocks of up to 30-40, or even 50 individuals. Building sparrows are
very sociable and constantly communicate with chirps. Flocks of young birds
of the same age, having simultaneously left their nests, often fly through the
desert.
The most interesting features of this species are its nest-building activity
and peculiar nests. Each nest represents a tube 50-60 cm long with a narrow
entrance and an extension at the very end. The tube is built of sand and clay
with the addition of plant parts – usually various parts of cacti, broken off
by the beak. The building sparrow shapes it with its beak, and then lubricates
it with saliva – the salivary glands are enlarged, especially during the building
of nests, and saliva is rich in glutinous protein, similar to that of swifts
and allies. Building sparrows build their nests on the ground, preferring inaccessible
ledges on cliffs. Nests are arranged close to each other, often in several floors;
birds often build their nests on the roofs of other bird’s nests. Such a settlement
of birds resembles the buildings of the settled Pueblo Indians. Cactus thorns
are often embedded in the outer layer of buildings.
Building sparrows search for food all over the cactus forest. The diet is very
diverse: any parts of cacti and other desert plants, insects, arachnids, small
lizards and snakes. A powerful beak allows the bird to crush such food. The
enemies of this species are mainly reptiles, birds of prey and mammals. Protecting
the nest from them, the Pueblo sparrows collectively attack them and cry loudly.
The only predator that can destroy their fortress is the gila-dragon.
The breeding season comes in spring, with the first rains. Shortly before it
begins, pairs are formed, and birds actively build a new nest, or repair an
old one. Due to well-coordinated work, they manage to finish the construction
in about a week. Old nests are also used, but they are often inhabited by parasites.
Mammalian wool and sometimes cactus hair is used as bedding. The female lays
up to 4 eggs and incubates them alone; the male feeds her. When the nestlings
hatch after 16-17 days, the female and male together carry food to them, because
at any time there are at least some birds near the nests that will make a noise
if they see an enemy. The juveniles leave the nest a month later.
Sexual maturity comes next year. Life expectancy is up to 12 years, but usually
it is less.
This bird species was discovered by Mamont, the forum member.
Jacana
wagtail (Dromotacilla nupharodroma)
Order: Passerine birds (Passeriformes)
Family: Wagtails (Motacillidae)
Habitat: freshwater reservoirs of Europe (east to the Three Rivers Land), wintering
in Africa in Saharan Nile basin.

Picture by Alexander Smyslov
The wagtail family got an opportunity to evolve actively when
vast treeless spaces overgrown with grasses appeared on the Earth. Most of the
species of these birds live in a relatively dry area, but some species have
mastered other habitats – the shores of freshwater reservoirs. Such species
existed already in the human epoch, but in Neocene the specialization of wagtails
to an aquatic lifestyle became even more pronounced. New Azora island is a home
to one of these species – the stream wagtail
(Motacinclus novazorae), capable of diving and running short distances underwater.
On the territory of Europe, there are other, less specialized nearwater species
– long-toed wagtails (Dromotacilla). These are small wagtails with thin long
beaks and very long toes, having the ability to walk on the floating leaves
of aquatic plants like water lilies and pondweeds, like jakanas. Wagtails of
the human era were sufficiently preadapted to such a life: a small weight and
a near-aquatic lifestyle allowed them to run their evolution in such a direction.
Long-toed wagtails are distinguished by very long toes: the length of each toe
exceeds the length of the tarsometatarsus. Toes are also equipped with long,
thin claws. Due to this device, these birds easily move on floating leaves.
In addition, long-toed wagtails are able to look for food by immersing their
heads in water, and if necessary and in case of danger, they dive. In general,
the appearance of these birds remains recognizable wagtail shape – large head,
long legs and long swaying tail. These birds retained the ability to fly, and
the primary feathers became dense and stiff, allowing them to paddle their wings
under water. Due to the content of melanin, which additionally strengthens the
feather, all species of long-toed wagtails have black or brown primary feathers.
Sexual dimorphism in coloration is not pronounced.
Jacana wagtail is a typical species of the genus that lives in river bays and
lakes with a slow current, heavily overgrown with aquatic plants. The body length
of an adult bird is about 10 cm, the tail is up to 8 cm. The head and back are
dark green; there are yellow “eyebrows” of thin feathers above the eyes. The
beak is almost as long as the head and slightly bent down. The nostrils are
able to close, which allows the bird to search for food underwater on the underside
of floating plant leaves. The primary feathers are black, the secondary ones
are bright yellow with black tips. Belly is grayish-white; there is a black
semicircle on the front of the neck. Tail is black. Skin on the legs is greenish-gray.
This bird species feeds on aquatic invertebrates: insects and their larvae,
small crustaceans, worms.
Unlike real jacanas, which have a precocial type of development of juveniles,
jacana wagtail belongs to the number of altricial birds. Therefore, it arranges
a strong reliable nest on land – usually under the roots of large trees, where
soil is washed out by water, or in low hollows of trees standing near the water,
as well as in the trunks of trees with soft wood that have rotted from the inside.
In a clutch there are up to 5 eggs, the female incubates exclusively, and male
feeds her. Both birds feed their nestlings together; during the summer, a pair
of birds makes two broods. This species is migratory and flies for the winter
to North Africa, to the Saharan Nile basin.

Picture by Alexander Smyslov
In the heavily overgrown impenetrable reservoirs of southeastern Europe, north of the mountain ranges of the Alps, in the Balkans and along the southern coast of the Fourseas, a close species lives – duckweed-running wagtail (Dromotacilla lemnodroma). This bird is very small in size – the length of an adult bird is about 6-7 cm, and the tail is of the same length. Due to its small size, it is able to walk on the thickets of duckweed and other small floating plants. The plumage color of these birds is bright green with black flight feathers and a weakly pronounced “scaly” pattern on the back and head – the contour plumage in these places has slightly darker feather edges. This species is more thermophilic and migrates to the Hindustan for the winter, flying to the wintering places mainly along the beds of large rivers.
The idea of the existence of these bird species was expressed by Simon, the forum member.
Wood
soilprober (Georimarornis motacilloides)
Order: Passerine birds (Passeriformes)
Family: Wagtails (Motacillidae)
Habitat: New Azora Island, tropical woods.

Picture by Alexander Smyslov
The animal population of New Azora volcanic island generated
from one of tops of Mid-Atlantic Ridge is presented mainly by European species
of birds. It is connected by that islands of Macaronesia (the area including
also Madiera and Canary Islands) still in historical epoch had been the wintering
place of plenty of European birds species. Till the ice age some birds had formed
in Macaronesia settled populations, and after formation of New Azora Island
they had successfully occupied it.
The nature of New Azora is very diverse: due to altitude zones various types
of vegetation are presented here: from tropical wood up to Alpine desert. The
fauna of island is presented by numerous species of birds and some species of
mammal – descendants of species introduced to islands by humans. Bird population
of island is especially rich.
In woods of New Azora the majority of birds live in tree crones. The gloomy
underbrush overgrown with ferns seems less suitable for their life, but it is
the deceptive impression. Many species of birds, varying in shape and feeding
habits, inhabit the underbrush of New Azora woods.
One of interesting New Azora birds is the wood soilprober. It had descended
from one of species of wagtails (Motacilla), spent winter at Azores in Holocene,
and formed the settled island form. Till millions years the descendant of small
wagtails had considerably changed: now it is rather large, starling-sized bird.
But the wood soilprober seems even larger because of long tail. It has kept
a characteristic for wagtail habit to shake tail up and down. Soilprober has
long and slightly bent beak: bird eats insects drawing them from wood litter.
In searches of forage this bird practically constantly stirs fallen leaves and
rakes ground. For such characteristic feature of behavior it has received the
name.
The body of this bird combines bright spots and cryptic colouring: at the wood
soilprober there are grey chest, brown wings with black spots and coal-black
tail. Tips of tail feathers have deliberately appreciable colouring: they are
snow-white with strong metal shine. “Mirrors” on the tail reflect the poor sunlight
getting in underbrush, and are used by birds of this species to submit signals
to congeners. Soilprober is the solitary bird strictly preserving the fodder
site from congeners. This bird moves in wood, running similarly to wagtails,
and shakes tail in same characteristic way. But, though it spends the most part
of time on the ground, this bird can fly well, and escapes from predators in
flight.
One more adaptation which permits to these birds to distinguish each other is
bright black-and-white head. Forehead, crown and nape at this bird are colored
black, from beak through the eye to a shoulder the white strip stretches, and
throat is black. At males white strip is appreciably wider, than at females.
During conflicts on border of territory birds rise opposite to each other, and
fluff feathering on head, making it similar to pompon. Thus they visually exaggerate
themselves, trying to impress the opponent.
Each individual of wood soilprober keeps in certain territory which borders
are well-known to it. Birds of this species eat small invertebrates, and frequently
feed near to other inhabitants of underbrush – Azorean partridges, local gallinaceous
birds. Due to quickness wood soilprober takes insects when partridges rake ground
by legs. Partridges try to drive off the importunate competitor, but all the
same the soilprober has time to eat more insects or seeds, than they get. But
nevertheless there is a benefit to birds from the presence of soilprober near
to them. It is very sensitive bird: at first attributes of danger it warns other
wood inhabitants by shrill resonant chirp.
Some times till one year soilprobers form pairs and rear posterity in common.
In courtship season the male executes original “dance”, perching on fallen branches
of trees or stubs sticking above leaves of ferns. It turns round, shakes tail,
stretches wings and utters modulating warble, involving females. Usually male
beforehand chooses the nesting place, and shows itself nearby to it. This bird
nests in hollow trunks of fallen trees, or in abandoned holes of various ground
animals. Similarly to all warblers, it is nidicolous species. In clutch there
are 3 – 5 eggs from which blind nestlings covered with thin grey down hatch.
They grow quickly, and at monthly age leave nest, keeping near to parents. Young
birds differ from adults in colouring: at them head is brown. It will get black-and-white
colouring to the first nesting season – at five-monthly age.

Picture by Alexander Smyslov
At coasts of island the close species lives: beach soilprober (Georimarornis psammophilus), which differs from wood congener in almost monochrome feathering of yellowish-grey color, masking bird on sand background. Also its beak is longer: it exceeds length of head. This bird feeds in ebb-tide strip, searching for small invertebrates burying in sand. Also beach soilprober clears large sea birds, living on beaches of New Azora, of parasites. At the same time it receives good protection against small feathered and four-footed predators of island. This species nests in cracks of rocks and among coastal vegetation, choosing places away from surf border.
Stream
wagtail (Motacinclus novazorae)
Order: Passerine birds (Passeriformes)
Family: Wagtails (Motacillidae)
Habitat: New Azora, mountain streams.

Picture by Alexander Smyslov
Expansion of Atlantic Ocean had resulted in occurrence of
faltering chain of islands in central part of this ocean. Already in human epoch
Iceland was large island, and Canary Islands and Azores represented archipelagoes.
In Neocene volcanic activity had resulted in formation of unite large New Azora
island instead of Azores.
The fauna of New Azora is formed as of descendants of species introduced by
people, as of species migrated in the natural way from Europe. They had occupied
all possible ecological niches at the island; tropical conditions and abundance
of food resources had allowed some of them to turn to highly specialized forms.
The small songbird able to dive and to run on the bottom dexterously, clinging
by paws for stones, lives in cool mountain streams of New Azora. It is similar
to ouzels (Cinclus), but it is not their descendant or the relative. This bird
is the stream wagtail, descended from blue-headed wagtail (Motacilla flava)
migrated to Azores in ice age at the boundary of Holocene and Neocene. This
bird initially lived near to water, and its descendants had developed this ability
even further.
Stream wagtail is approximately starling-sized bird. Its shape only by few features
specifies that the bird is adapted to life in water, but it is the full inhabitant
of fast mountain streams by the way of life. This bird has streamline body,
conic-shaped head, rather short and rounded wings, and the short fan-shaped
tail. This bird inherited the habit to shake by tail from wagtails, its ancestors.
This feature of behaviour expresses an emotional condition of bird and is used
in courtship displays. The plumage of stream wagtail is greased with plentiful
secretions of coccygeal gland, therefore, having jumped out from water, bird
remains absolutely dry. Besides it has kept one preadaptation existed at wagtails:
leathery covers at nostrils.
The basic plumage colouring of this bird is greenish. Covert feathers of wings
at this bird are dark with wide bright green border; therefore the body top
of the bird looks spotty. The bottom part of body sharply contrasts with top
– it is yellow; this feature is kept from an ancestor. From beak across eyes
on lateral faces of neck to wings black strips stretch, dividing colouring of
top and bottom parts of body. Short tail is black; on tips of feathers there
are white spots.
Legs of stream wagtail are well adapted to run on stones slippery of algae and
water. On the bottom surface of long toes the set of corneous bristles develops,
helping to cling to support. This bird runs, instead of jumps, as many songbirds
do. Stream wagtail flies rather seldom, and prefers to dive into the water for
the sake of food and protection against a predator. Sometimes the bird gathers
speed in air, folds wings and dives into water, as if the thrown stone. Under
water this bird due to special body shape does not spend efforts in order to
keep in depth – current presses it to the bottom. Food of stream wagtail includes
larvae of insects and young growth of small crabs. This bird also catches larger
crabs and pecks them on coastal stones. Sometimes these birds become prey of
fishes swimming to short rivers of New Azora from the sea, or large crabs, which
can ravage their nests and eat nestlings.
Stream wagtail inhabits coastal vegetation. Each individual declares rights
to the territory by series of loud dual calls. If the contender does not leave,
the owner of territory meets him, showing the colouring. It fluffs feathers
on stomach, extends body vertically and opens tail like a fan. After such demonstration
the owner of territory is ready to banish the newcomer using force.
Pair at these birds is formed to one season of nesting, and breaks up soon after
posterity rearing. Birds arrange nest in holes, cracks between stones and in
thers shelters, where the predator can not penetrate. In clutch of this species
there are about 4 eggs. Naked and blind nestlings are looked after by both parents.
Parents bring up them mainly with soft larvae of insects. Approximately after
43 – 45 days completely advanced fledglings do not require any more parental
care, and the pair of adult birds breaks up. Young birds nest for the first
time at the age of 7 – 8 months. Life expectancy of this bird does not exceed
4 years.
The idea about existence of this species of birds was proposed by Simon, the forum member.
Prairie
or blackish pipit (Anthus nigricans)
Order: Passerine birds (Passeriformes)
Family: Wagtails (Motacillidae)
Habitat: prairies of North America.
Prairies and other open spaces in Neocene North America are either snowy plains
in winter, or rather dense thickets (and sometimes even entire groves) in the
warm season, when herbaceous plants grow abundantly. With the disappearance
of people and civilization, nature has recovered, and open areas with moderate
moisture have again become the kingdom of herbs with a small admixture of shrubs.
There are very few trees in such places, shrubs are playing supporting roles,
and herbaceous plants, especially cereals, dominate. In addition to large herbivores,
prairies are inhabited by a large number of small animal species, and among
them is a songbird – a prairie pipit.
Outwardly, this distant relative of wagtails resembles a sparrow, but it is
of a slimmer build, with a longer and thinner beak. This bird eats mainly insects,
sometimes berries and small seeds. Prairie pipit has a pronounced sexual dimorphism:
the male is dark gray, almost black, only the tail feathers have white tips,
with which the bird signals to its relatives in flight. The female is paler,
gray-green in color with brownish wings and a dark stripe stretching along the
side of the head from the corner of the mouth across an eye to the ear opening.
A white “eyebrow” stretches over this dark stripe. There are characteristic
mottled spots on the female’s belly, and a light scaly pattern on the wing coverts.
The behavior of these birds is secretive: both females and males prefer to hide
in shrubby or grassy thickets. However, the song of these birds is loud, and
the prairie pipit is much easier to hear than to see.
In early autumn, prairie pipit migrates south to Central America, where it winters
in dry cactus-xerophytic forests at elevations. This species is monogamous,
but pairs keep only for the nesting period; after raising offspring, families
break up. In the spring, males returning to the prairies occupy individual territories
and begin to call females to create a family. This species is territorial, but
birds are relatively tolerant of relatives, and family couples nest in the same
bushes not very far from each other. In this case, their feeding grounds radiate
from common shrubs with nests, and different pairs do not compete with each
other.
The nest is made of grass, plant fibers and cobwebs above the ground – in bushes
or on a low tree. There are 3-4 eggs with a whitish shell covered with dark
gray spots in a clutch. The clutch is hatched by both parents for about 2 weeks,
with the male replacing the female in the morning and evening. Nestlings hatch
blind and naked, but they grow quickly, and at the age of 2 weeks they are already
able to leave the nest, and adult birds make a second clutch. By the end of
summer, young birds gain strength and are able already to migrate with their
parents. Before maturity, males and females do not differ in plumage color.
The main enemies of the prairie pipit are various birds of prey. Life expectancy
is no more than 8 years, but birds mostly die much earlier, especially during
migrations and wintering.
This bird species was discovered by Bhut, the forum member.
Elf
redpoll (Acanthis pygmaeus)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Loki Island.
Small, widespread and aggressive birds capable of finding food in human-altered
landscapes, redpolls (Acanthis flammea) survived the human era and the ice age
without losses, preserving their numbers and wide range. Moreover, a part of
the population of these birds maaged to adapt to life in the forests south of
the taiga zone after the retreat of the glacier, as well as to colonize Greenland
from America again. After the appearance of Loki Island, birds from the Greenland
population settled there – perhaps they got there with a storm, like many other
birds that inhabited this isolated world. On the island, these birds have become
smaller, and the color of their plumage is duller. Their descendant in Neocene
is elf redpoll.
In conditions of a relatively warm and mild climate, this bird species has become
very small – the body length is about 10 cm, the wing length is up to 6 cm.
The color of the plumage is variable – from a uniformly colored gray top and
lighter bottom to white with gray “ripples” on the wings, head and back. The
red markings on the plumage have almost completely disappeared, only occasionally
the breast in some males has a slight pinkish tinge. Elf redpolls feed on buds,
seeds, insects and arachnids. Due to their omnivorous nature, the birds managed
to populate the entire territory of the island – from the seashore to the craters
with hot springs. These birds live in flocks of 10-12 adults; they also nest
together and look for food both in tree crowns and on the ground. The calls
of birds in the flock sound a murmuring “che-che-che” with separate “chen” sounds;
the alarm signal is a rolling “al-al-frr”.
The mating season begins in March-April. At this time, males flutter in the
branches of shrubs, displaying themselves and uttering a buzzing trill. Nests
are built on deciduous trees, out of reach for terrestrial birds. These nests
often form a collective structure looking like a platform with a loose roof.
There are 5-7 eggs in the clutch, which are incubated by the female for 2 weeks.
Nestlings are fed initially on insects only, and then on mixed food, including
buds, flowers and seeds. After 17 days, the juveniles fly out of the nest. In
good years, elf redpolls manage to rear 2-3 broods.
These birds grow very fast and do not live long: sexual maturity at the age
of one year; life expectancy is up to 6 years, only few birds live up to 8 years
old. Many die in the first 3-4 years from predator attacks.
This bird species was discovered by Nick, the forum member.
Wood-crushing
rosefinch (Conorostra cactivora)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: large cacti in the deserts of North America.
The deserts of North America are home to a number of strange bird species –
for example, flightless turkey
ostriches, dwarf herons
and large ground-dwelling owls.
Another unusual bird from this area is a wood-crushing rosefinch, a descendant
of the house finch (Carpodacus mexicanus).
It is a small bird species: the body length of the wood-crushing rosefinch is
up to 15 cm. It has a solidly built body, a medium-length tail and paws, and
a disproportionately large, deep and strong beak; there is a small downward-curving
hook on the upper beak. The plumage is quite stiff, especially in the front
of the head. The feet have large claws. The outermost tail feathers have strong
shafts forming spikes protruding from the vane. The color of the plumage is
bright and light: the whole body is yellow of various shades depending on heredity
and population; on primary feathers, there are light brown elongated spots noticeable
in flight. There is a large orange spot on the marginal coverts. Throat is bright
red; tips of the tail feathers are cherry-red. Beak is dark grey.
Wood-crushing rosefinches inhabit deserts and cactus forests in southern North
America, in the deserts of the Mexican Plateau. Birds live in pairs, up to 3-4
pairs can nest on the same large cactus. Having found a suitable large cactus,
the birds begin to build their dwellings. They choose a small area on the surface
of the cactus, carefully tear out all the spikes and hairs from it, and then
begin to literally gnaw through a very long but narrow passage in the pulp of
the cactus trunk. The bird tears out the cactus tissue with its beak and either
eats it or throws it on the ground. Having built a tunnel of sufficient depth,
the wood-crushing rosefinch builds an expanded nesting chamber at its end. After
the finishing of nest building, the birds give the plant the opportunity to
dry the walls of the tunnel, after which they remain to live on the cactus.
Less often, wood-crushing rosefinches can settle in an abandoned bee nest or
in a woodpecker’s hollow. If several couples of them live on the same cactus,
they build tunnels so that their burrows in the pulp of the plant do not intersect.
Less often, birds gnaw out tunnels in the soft wood of other trees, including
rotten ones.
The diet of this species is very diverse: cactus pulp, flowers, fruits, parts
of other plants, insects and arachnids living on cacti, sometimes small reptiles.
The enemies of the wood-crushing rosefinch are birds of prey and reptiles. To
protect against them, these finches stay closer to the cacti and hide in their
own passages. Often, when a bird sees a predator, it cries loudly, warning the
other inhabitants of the cactus about the danger.
At the end of the dry season, the female lays 3-4 eggs and stays in nest to
incubate them, while the male feeds the female. By the time the nestlings are
hatched, the wet season comes, and there is more food for the offspring. The
juveniles grow and fledge quickly, and at the age of 17-18 days they get out
of the nest and fly away soon. The next year, the birds form pairs and look
for a cactus for housing.
Life expectancy is up to 9-11 years.
A variety of animals settle in the abandoned nests of the wood-crushing rosefinch:
insects, birds of similar sizes, as well as climbing reptiles.
This bird species was discovered by Mamont, the forum member.
Parrot
finch (Psittospiza pseudopsitta)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Galapagos Islands, thickets of treelike cactuses.

Picture by Pavel Volkov
Galapagos, or Darwin’s finches were birds on which example
the phenomenon of adaptive radiation – origin of set of related specialized
species from common unspecialized ancestor –had been considered for the first
time. In human epoch these birds had not from each other too strongly differed.
But gradually the world, in which Galapagos finches lived, began to change:
people had introduced new species of animals, and after the ending of historical
epoch new species of animals got to Galapagos Islands naturally. Gradually some
lines of Galapagos finches had died out, superseded by new migrants from continent,
and only few most specialized representatives had preserved in Neocene. One
of these species is the parrot finch a little similar to small parrot externally.
It is the single species of genus settled at all large islands of archipelago.
Parrot finch is a large-headed starling-sized bird, one of the largest Galapagos
finches. At it there are rounded wings and wide tail, permitting to maneuver
in rich thickets where it lives. Despite of much-promising name, parrot finch
is colored softly: at it there are brownish wings with yellowish stripes and
back, pale yellow stomach and white “cheeks”. On head of birds of both genders
there is a crest which may rise slightly when the bird is excited. The crest
is colored bright green, and bright spots of same color are under wings. They
are especially appreciable when the bird is excited: annoyed or busy in courtship
displays.
The beak of parrot finch strengthens its similarity to parrot: it is high and
strong, with the bent tip, black-colored. But, as against to beaks of parrots,
the beak of finch is compressed from sides on tip, forming cutting “edge”, and
the top jaw is motionless relatively to skull.
One more difference of parrot finch from parrot consists in nesting behavior.
When parrots nest mainly in tree-trunk hollows, parrot finch builds nest independently.
In the beginning of rain season the male starts to build some spherical nests
of twigs, and displays near to each one, gradually completing it. During courtship
demonstrations male opens and raises wings, freezing on unfinished nest with
twig in beak. Such demonstrations alternate with warbling similar to pipe sounds
and active construction of nest.
This bird is monodin. When the female is interested in male and its nest, birds
in common complete the nest which the female has liked, and other ones stay
unfinished.
Because parrots could not get to Galapagos Islands, this bird had occupied their
ecological niche: parrot finch eats firm seeds and fruits of various plants.
This species can even eat prickly fruits of treelike prickly pears – most typical
plants of islands. By sharp tip of beak finch cuts skin of fruit, and then accurately
scrapes out friable pulp with seeds by bottom jaw. Small seeds of treelike cactus
pass through its digestive path not digested, and are distributed with dung
of bird.
For one rain season parrot finches have time to make two clutches of 4 – 5 eggs,
and to bring up nestlings successfully. Young birds are colored softly: they
do not have green spots under wings, and yellow color is replaced by grey. They
get adult colouring only at the eighth month of life.
This species of birds had been discovered by Simon, the participant of forum.
Night
vampire (Dinogeospiza strix)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Galapagos Islands, woods.

Picture by Alexey Tatarinov

Picture by Sauron
In conditions of rigid competition for fodder resources the
most favourable strategy is narrow specialization and high fitness to the chosen
source of forage. In this case food predilections of adjoining species do not
coincide, and the competition between them is practically not present. It had
taken place so at Galapagos Islands where after end of human epoch many new
immigrant species had appeared. As the result some of ground finches, endemic
for these islands, had died out, and others had become narrowly specialized
species.
One of former ground finches had mastered very unusual food source, becoming
true parasite. This bird eats blood of various animals inhabiting islands, but
prefers to attack huge tortoises living in Neocene epoch at Galapagos Islands.
For its food predilections it is named “night vampire”. However, this bird also
eats parasitic insects and ticks living on tortoises. Probably, some millions
years ago ancestors of this bird were fed with parasitic arthropods, and only
later passed to feeding by blood of animals. It is interesting, that in Northern
Africa the same way of feeding was mastered by bird
of weaverbird family. That fact is even more remarkable, that one of subspecies
of ground finches already in human epoch had attacked gannets nested at Galapagos
Islands and had sucked their blood.
Appearance of this finch does not differ in any special frightening fig. Its
feathering is colored black with bluish lustre on neck which at male is expressed
stronger, than at female. Eyes of bird are rather large, dark, with transparent
haw. They are sensitive to light: the bird can find animals on which it is fed,
even at light of stars. And in the afternoon haw serves for protection of eyes
against bright light. Long sharp beak of night vampire is colored white, and
near its basis the skin is featherless – it permits to the bird to keep itself
clean.
Wings of night vampire are rounded and rather short. Edges of primary feathers
are covered with tiny barbs which guzzle noise of flaps and allow a bird to
fly silently like owls.
Night vampire is small species of finches: this bird is less than sparrow by
size. Probably, this circumstance rescues various inhabitants of islands from
it: for night each bird sucks at other animals only about 20 grams of blood.
This bird leads extremely nocturnal way of life and in day time hides in secluded
places – holes, tree-trunk hollows, cavities in trunks of fallen trees. At night
this finch flies out from shelter and searches for fodder animals, using sharp
sight and good hearing. Most frequently it attacks tortoises which are not so
active at night, as at the afternoon. Less often these birds feed in colonies
of sea birds spending the night at the coast. Going to feed, the bird flies
up to sleeping animal, accurately sits on it or beside, and pecks skin of victim.
Its saliva contains anticoagulant and proteins similar by action to snake poison
– they lightly paralyze nerves; because of it animal at which this bird feeds,
does not feel pain, and blood does not coagulates. All operation passes very
cautiously: the bird waits, while its saliva will work on victim, and only some
minutes later starts to peck wound and to suck blood.
In the afternoon night vampire sleeps in tree-trunk hollows and others shelters.
Usually birds of this species feed individually, but gather to day spending
in groups of some birds. Pair at this species is kept till all life: it helps
birds to survive. It happens, that one of partners could not be sated for night.
In this case other bird belches to it in beak a part of sucked blood. But such
mutual assistance exists not only within the limits of one pair: if it is necessary,
birds from different pairs feed each other. The bird eliciting forage, behaves
similarly to nestling: it squats before the supporter bird, and trembles wings,
having slightly opened mouth.
Male ready to nesting warbles in the morning and at sunset. Voice of this finch
sounds like melodious warbles, interrupting by separate cries. This species
nests in same shelters where it spends the day, but each pair builds separate
nest. Formation of pairs and nesting can pass at any time of year, and for one
year pair can rear up to three hatches. In clutch there are 2 – 3 eggs which
are hatched alternately by male and female within two weeks. Nestlings hatch
naked and blind; they start to fledge since the first week of life. At them
black feathering grows at once, but it has no metal shine characteristic for
adult birds. Female and male bring up nestlings at first with insects, and then
with blood within three weeks, and then accustom young birds to independent
feeding. Propensity to peck skin of animals at young birds is instinctive, but
skill to suck blood cautiously is developed at each bird individually.
This species of birds had been discovered by Simon, the participant of forum.
Cuckoo finch (Cuculispiza parasitica)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Galapagos islands, woods.

Picture by Alexander Smyslov
Supervision for Galapagos ground, or Darwin’s finches, realized
by people, had revealed one interesting feature of courtship behavior of these
birds:
too frequently males of finches had displayed near the unfinished nests of
congeners. Obviously, this feature of behavior had extended and also developed
further in Neocene epoch, because in Neocene fauna of Galapagos Islands one
species of finches becoming the nesting parasite of other smaller birds of
islands had appeared. This characteristic feature of its behaviour had determined
the name: cuckoo finch.
This sparrow-sized species differs in short wings and tenacious paws. Cuckoo
finches live in woods, searching for food both on the ground, and in tree
crones. They eat insects with firm covers, and also dry seeds of trees and
grasses.
In connection with such diet the beak of this species had kept characteristic
“finch” type: it is conic, thick at the basis.
The majority of ground finches differ in dim feathering, but the present
species is the exception among them. Besides at cuckoo finch the sharp sexual
dimorphism
is advanced. The male of this species is colored brightly and contrastly:
it is entirely white with black eyes and beak; its primary feathers are black
with white border, tail is black too. Female is brown with “agate” pattern
of yellowish-green shades, varying at various individuals. The difference
in
colour at these birds is connected to their role in breeding. This species
of finches is the polydin which is not forming constant families. The male
in courtship season involves as much females as possible, for what it keeps
openly and displays in the afternoon. The task of the female is right opposite:
to be imperceptible and to lay egg in another's nest so to not give out its
location to probable predators by its actions. Therefore females keep very
cautiously, and even cry only in extreme cases.
The male of cuckoo finch displays near nests of other birds, usually near
to unfinished ones, and even near to abandoned ones. Nest in this case is
powerful
stimulus of breeding behaviour. Sometimes the instinct has “malfunction”,
and male chooses for displaying plant sprouts struck with diseases, causing
ugly
growth of branches. Male does not make attempts to build nest, but elements
of building behaviour had remained at it: male “bows”, sitting near the nest
and holding in beak a blade of grass. Sometimes it throws grass and warbles,
uttering resonant warbles similar to reed-pipe sound. When the female flies
to him, male jumps around of her having semi-stretched wings, quickly couples
(continuing to hold a blade of grass in beak), and the female departs – she
does not interest the male any more at present. To lay an egg is her problem
in which male can not take part having so bright feathering.
As a rule, two males do not live beside, but one male is visited by some
females from next territories. One female can couple with several males living
in environs.
This bird lays eggs in nests of other small birds of islands. Female remembers
the location of several tens nests of other warblers (including finches to
which it lays eggs more often), and visits them in process of clutch formation.
To lay an egg, the female behaves very cautiously: she sneaks up to chosen
nest and hides nearby. Having seized the moment, she steals and eats fresh
egg of owner bird, then quickly lays her own egg and flies away. Till one
nesting season (about half-year in most damp months) the female lays in another's
nests
approximately 15 eggs – about three full clutches of finches of the comparable
size.
Nestlings of some nest parasites begin the life from the killing or throwing
out from nest of nestlings of the host bird. Nestlings of cuckoo finch, on
the contrary, peacefully get on in new family, and consume as much forage,
as much nestlings of host bird need. They grow approximately with equal grade,
as other small birds: become fully fledged approximately to the second week
of life, and leave nest a week later. The first feathering of the cuckoo
finch is modest and brownish. Males get contrast colouring at the age of
6 months
– when they become sexual matured.
Diverse-beaked finch (Heterospiza dimorphorostra)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Galapagos Islands.
Ground finches (Geospiza), lived at Galapagos Islands in human epoch, were
known as a vivid example of adaptive radiation – from the common ancestor the
set of descendants, differing by habitats, habit of life and shape, had evolved.
The part of species of these birds had not evolved any descendants and had
died out in epoch of global ecological crisis. Descendants of survived species
had occupied the exempted ecological niches and had given rise to new, sometimes
rather freakish species.
In forests of Galapagos Islands formed by cacti and treelike representatives
of Asteraceae, very original species of finches lives – diverse-beaked finch.
It is insectivorous bird living in crones of trees and seldom moving down to
the ground. Birds of this species are interesting in features of their beaks:
beaks of males and females strongly differ by shape. Beak of female of diverse-beaked
finch is long, thin and slightly deflexed, as at treecreeper (Certhia). It
helps the female to take insects and their larvae from under peeled bark, from
cracks in wood trunks and between stones. Beak of male is massive, pointed,
hooked on the tip like a beak of butcherbird. The reason of this sexual dimorphism
at diverse-beaked finches is rather original. The matter is that the male does
not extract food to itself independently – he is supplied with it by the female.
But the male, due to the beak, protects the female from predators and competitors,
constantly accompanying with her during searches of food. Also he helps the
female to take forage, crumbling wood and tearing off bark by beak. The similar
phenomenon of sexual dimorphism was typical for New Zealand wattlebird Heteralocha
acutirostris from Callaeidae family, which was exterminated by people approximately
at 1907.
Male of diverse-beaked finch is starling-sized bird; female is smaller a little.
Birds of both sexes are colored modestly: their plumage is monochromic, blackish-brown.
As opposed to plumage, the male’s beak is colored brightly: it is lacquered
pink with white tip, well appreciable from apart. It is a signal: such colouring
serves both for attraction of the female, and for the warning for predators,
that this bird can give solid repulse. Male of diverse-beaked finch is remarkable
by bellicose behaviour, and sometimes it rushes to the opponent considerably
surpassing him in size – he attacks, targeting by beak to eyes and naked areas
of skin.
These birds are monodins; pairs at this species form to all further life, and
partners almost never leave. If one bird of the pair dies, the second one urgently
looks for its replacement. The problem of partner search for widowed males,
which can not feed normally independently and survive only due to casual food,
is especially actual. Diverse-beaked finches build collective nests, where
at once some pairs lay eggs. It is connected to features of behaviour of these
birds, because at once both parents search for food. While some pairs search
for food, other ones stay in the nest, hatching eggs and protecting it. In
warm climate absence of both parents in the nest has not an effect for embryo
development in egg. Within one year these birds can nest up to three times.
In clutch of diverse-beaked finch there are 4 – 5 eggs incubating by male and
female alternately. Female, free from hatching, keeps in vicinities of nest,
expecting on protection from the part of congeners.
At just hatched nestlings beaks are typical for all finches – they are conic
and only slightly deflexed. They keep such shape until birds become adult.
The young birds had left nest successfully can feed alone. But at the coming
of sexual maturity (at the age of about 7 months) the situation starts to change.
At males and females beaks become different, and they are compelled to search
for the partner to themselves: male needs it for feeding, and female – for
protection against enemies.
This species of birds was discovered by Simon, the forum member.
Sumach finch (Unedulis rhucivorus)
Order: Passerines (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Japan, woods and bush thickets.

Picture by Simon
Some species of birds of family Colluricinclidae named pitohui
(Pitohui) lived in Holocene in New Guinea Island. These birds had a proof unpleasant
smell,
but their even more remarkable feature was the unique for birds attribute
– the poisoness. In skin and feathers of these small birds with a bright red-and-black
feathering strong poison accumulated. It protected birds both from ectoparasites
and from four-footed predators. For mankind the nature of pitohui poison
remained
a riddle for a long time. But it was also known that some other birds could
become inedible and even poisonous after ate poisonous plants. It was supposed,
that some species of fishes also became poisonous after the feeding on poisonous
algae. The poisoning with such fishes was known as “siguatera”. But the phenomenon
looking the exception for vertebrate animals appeared usual tactics for invertebrates.
In Holocene butterflies of milkweed butterflies family (Danaidae) used for
protection the poison of plants belong to devil’s milk family (Euphorbiaceae)
on which their caterpillars were fed. In Neocene such tactics of protection
has not lost the urgency: Asian menthol
leaf beetle and burning snail from
Hawaiian Islands protect from enemies the same way. Among vertebrates adherents
of such way of protection also had appeared: at the Sunda Land, the large
island in Indonesia, the beetlecracker
finch lives. It eats poisonous insects and
accumulates in body their poison for self-defense.
Beetlecracker has the relative at Japan Islands – the sumach finch. This
starling-sized bird is named so because it lives in thickets of poisonous
sumach bush (Rhus,
Toxicodendron) of various species, and even among branches of “death
tree” (Necrodendron omnimortalis) fatally poisonous for the majority
of animals. But it is not a simple whim in choice of an inhabitancy, but
vital necessity
for bird. Sumach finch eats exclusively seeds, flower buds and leaflets of
poisonous species of sumach. Such diet provides to the sumach finch the fine
protection: its meat also becomes poisonous; in it strong vegetative toxin
accumulates.
Sumach finch prefers to not depart far from thickets of plants in which it
lives. This bird is narrowly specialized to such unusual species of food
as seeds and greens of poisonous plants, therefore in the distribution it
is closely
connected to places of their growth. These finches often lodge in thickets
of “death tree” – very poisonous wood plant of Japan Islands. In connection
with relatively settled way of life wings of these birds are rounded, and
a tail is rather short and wide. Such constitution permits to move well among
rich branches of bushes. Legs of bird are long and have tenacious toes. Sumach
finches frequently swarm up trunks of “death tree”, searching for few insects
able to feed on this plant.
Colouring of feathering at sumach finch is very bright: the basic colouring
of bird is orange, on head there is reddish “cap”, and back is brightly yellow.
Primary feathers and tail are black with white tips, and around of eyes there
are “glasses” of black feathers. This colouring is typical warning – predator
risked to have eaten such bird may receive rather strong poisoning. Finches
keep deliberately safely: they do not hide at any feathered or four-footed
predator, and only meet its occurrence with disturbing calls. If the enemy
comes too near to bird, sumach finch starts to show the colouring: bird fluffs
feathering on head and breast, also opens wings and tail like fans, making
especially appreciable their black-and-white colouring. Usually the predator
had tried once meat of this bird, avoids the further attack.
Sumach finch eats rather firm food. In this connection at it the powerful
conic beak of black color with white strip in the basis was developed. At
males the
beak is thicker a little, rather than at females.
This bird is monodin; pairs at it are formed only to one nesting cycle. The
male ready to nesting begins courtship demonstrations: it chooses rich thickets
of young sumach plants and starts to sing for a long time, making a break
only in hottest afternoon time. The song of sumach finch includes series
of whistling
sounds. The disturbed bird utters lingering warble of “dry”, abrupt and sharp
sounds. It acts so, noticing any feathered or ground predator.
When the female has chosen a site suitable to nesting, she starts to build
nest of thin rods. The male at this time sings actively, protecting nesting
area, and occasionally visits the female, bringing to her some blades of
grass or sprigs for nest construction. The nest of sumach finch looks like
a deep
bowl in forked branches.
Usually this bird makes hatches per one year – in late spring and in the
middle of summer. But if the winter is warm enough, birds may nest earlier:
in this
case at the end of summer they have an opportunity to lay eggs third time.
The third hatch is usually small, but it survives not less successfully,
rather than posterity of spring and summer hatches. In clutch of sumach finch
it may
be up to 5 – 6 eggs. This bird has so well adapted to coexistence with this
plant, that even its eggs accumulate in yolk a small amount of alcaloids.
Therefore sumach finch is not afraid, that its clutch may be ravaged: it
nests at small
height from the ground, among sumach branches and among young plants of “death
tree”. Eggs of this bird have bright white shell with black speckles. Having
tried eggs of this bird for some times, nest ravager will not die, but will
remember well the relationship between these easily accessible eggs and the
subsequent sharp stomach-ache.
Eggs are hatched almost exclusively by the female, and the male catches insects
and gathers edible (only for these birds!) parts of sumach bushes. Also the
male protects the territory from contenders, though practically the rivalry
is not so sharp being out of competition with other bird species, and intraspecific
aggression at this species is weak.
First time parents feed nestlings on insects, then they add to food semidigested
seeds and buds of sumach and other plants. Nestlings accumulate poisonous
substances with such forage, and in their development at once get the bright
warning feathering
(as against to them, nestlings of beetlecracker have not striking cryptic
colouring up to the first mew).
Life expectancy of sumach finch does not exceed 4 years, and birds living
in thickets of “death tree” live up to the age of 3 years seldom. Obviously,
it
is the “payment” for inhabiting in so dangerous conditions – the organism
of bird should spend energy to neutralize the certain amount of poison. But
survival
rate of posterity at these birds is very high: they are practically not threatened
with predators, only casually appearing in thickets of plants where these
finches live.
Chaffrow
(Postfringilla novaezealandiae)
Order: Passerine Birds (Passeriformes)
Family: True Finches (Fringillidae)
Habitat: Open country and wooded plains, meadows North and South Islands of
New Zealand.
The Neocene bird fauna of New Zealand is typified by a mix of descendants of
both introduced and native species, and thus we see with descendants of numerous
kinds of English countryside finches released there during the age of man.
The Chaffrow is one such example, it has come to be the dominant kind of grain-eating
finch in New Zealand’s Neocene, descended from the Chaffinch (Fringilla coelebs).
A small bird, it almost identical in size to its ancestor, and is broadly similar
in coloration, but differs in having a bright red face and chest with pink underparts
in males, and a bright blue nape, females are a duller brown, moulting does
not appreciably change the colour.
This small bird is diurnal and feeds almost entirely on the seeds of grasses
and sedges, as well as insects. It forms large flocks outside of the breeding
season, which can be seen ranging over open ground, foraging for seeds. Chaffrow
fly exceptionally well, and flocks will take flight at the slightest provocation.
Breeding season occurs from September to November, with the flush of spring
growth, during this time their diet shifts partly to insects in order to feed
their young. Flocks break up into and seek wooded areas or trees in which to
court and nest. The male sings profusely and sweetly in order to attract a female,
with more than one kind of song alternating, otherwise these calls resemble
those of their ancestor, but dialects exist in different parts of their range.
Eggs are laid in the first year; the nest is built by the female, generally
in the fork of a tall bush or tree, out of reach of most predators. The nest
is a deep cup lined with roots and plucked feathers and covered on the outside
with lichen. A clutch of 4 or 5 eggs are laid and can be variable in colour.
Eggs are incubated for up to 15 days by the female, young are altricial and
helpless, attended and fed mainly by the female. Nestlings fledge after about
16 days but are still taught to feed by the parents. Second broods are usually
made except in bad years. Predators include raptorial birds and tree climbing
mustelids, lifespan is up to 6 years but usually as short as 3.
This species of birds was discovered by Timothy Morris, Adelaide, Australia.
Golden-fronted
erythrophonia (Erythrophonia flavifrons)
Order: Passerine birds (Passeriformes)
Family: Finches (Fringillidae)
Habitat: Atlantic forests of South America.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The Genus Erythrophonia originated from species of Euphonias
that survived the anthropogenic onslaught. The golden-fronted erythrophonia
is the type species of this genus. It inhabits the canopy of the Atlantic forest
of South America.
The golden-fronted erythrophonia have 12 cm of length and 21 of wingspan. The
male has entirely bright red underparts from the throat to vent save. The upperparts
are a dark blue-black except for the yellow crown patch which extends from the
bill over the head to just past the eye. The female, like most representatives
of this genus, is dull olive above and pale red below. In both sexes, the bill
and legs are gray.
It’s one of the best imitators among the South American birds. A single male
can manifest in a few minutes in the voice of 10 to 16 different bird species.
They are perfect imitations, but translated to their own reduced vocal strength.
The repertoire of the golden-fronted erythrophonia becomes a faithful copy of
the avifauna of the region in which it lives. Such characteristic is shared
by all representatives of the Genus.
They eat small fruit and berries, particularly mistletoe. Insects can be consumed
occasionally, being more frequent when they have chicks.
They are social birds, forming flocks of 7 to 20 individuals. Pairs are monogamous,
and stay together year round. Both parents build a globular nest of dried grass
and stems, lined with finer material and with a side entrance. The female lays
three to four cream-colored, brown-splotched eggs, which she alone incubates
for 15 days. They have 2 to 3 broods during the nesting season, which starts
during the end of winter. Both parents feed the hatched chicks. The chicks fledge
within 20 days. Sexual maturity is reached with 12 months and they have a lifespan
of 8 years.
The Genus Erythrophonia have other representatives. The black-throated
erythrophonia (Erythrophonia amazonica) replaces the golden-fronted
erythrophonia in the Amazon rainforest. This bird is more common in varzea forests
and areas that are flooded during the monsoon. They have 11 cm of length and
20 cm of wingspan. The male of this species have a red belly and chest, with
the rest of the plumage being dark blue-black, including the throat. They also
have a small yellow patch over their beak. The female is similar to the other
species. Another representative is the red-crowned
erythrophonia (Erythrophonia estemmenorubra), an inhabitant of the
woodlands and gallery forests of Central South America. This bird has 9 cm of
length and 19 cm of wingspan. The male is similar to that of the golden-fronted
erythrophonia, with the exception of having red plumage on the crown. The last
member of this genus is the Andean
erythrophonia (Erythrophonia andina), a bird endemic to the cloud forests
at the slopes of the Andes. They have 10 cm of length and 21 cm of wingspan.
The adult male has glossy olive upperparts, a yellow forehead, and a bright
red belly. The adult female has less glossy upperparts than the male, a yellow
crown and a small rufous belly patch.
This bird species was discovered by João Vitor Coutinho, Brazil.
Thick-billed
guiraguacu (Guiraguacu macrorhynchus)
Order: Passerine birds (Passeriformes)
Family: Troupials (Icteridae)
Habitat: rainforest of northern part of South America.
Oropendolas represent rather successful group of passerine birds of New World.
In South America some species of these birds inhabited Andean montane forests,
and in human epoch some of them survived in relic inaccessible forest patches
in places hard for human activity. During the period of restoration after global
ecological crisis descendants of these birds settled to rainforests of plain
areas of the continent, filling the empty ecological niches. In Neocene some
descendants of the russet-backed oropendola (Psarocolius angustifrons) are characteristic
birds of rainforests of northern part of South America.
These Neocene oropendolas form a separate genus Guyraguacu (açu and guaçu means
“big” in Tupi, and the whole name means “large winged animal”) and have a rather
large size and very thick bill compared to related forms. The most common species
of South American rainforests is thick-billed guiraguacu.
It is a largest representative of the genus: male is 53 cm long, female grows
to 44 cm. It inhabits the Amazon basin and lives in the rainforest canopy almost
never descending to the ground. Its striking feature is the thick and colorful
beak of the males; corneous cover of beak stretches to bird’s crown, making
birds looking as if “bald”. Females are smaller than the males and have beaks
less thick in size and pale in color. The color of male’s beak is black with
a bright red part at the tip; feathers of its body are green and tail feathers
are yellow. Red tip of its beak attracts the attention of females, being especially
noticeable on the background of male’s plumage. Female has black beak with yellowish-white
tip visible in the darkness of the nest.
Being solitary most of the time, they gather in harems in the summer, when the
mating season takes place. Males behave aggressively when protect their territory
and harem. Their harem numbers from 6 to 9 females building their individual
nests on tree tops close to each other.
These birds eat fruits, insects and eggs of other birds. But the individuals
of the Amazon basin began to have more predatory habits, resembling thus feeding
habits of toucans, being capable to eat eggs, small birds and reptiles. Larger
size facilitates them the capture and killing of prey animals.
The courtship consists in the male to vocalizing while showing their colorful
beaks. Their vocalizations can be of their own sounds or imitations of sounds
of other birds living nearby. The more colorful the beak and the more diverse
vocalizations are made, the greater will be the male’s harem. The females build
nests of plant fibres and vines. In a tree that is usually in the center of
the male’s territory, there are various long woven basket-like nests, each hanging
from the end of branch. The male protects the harem from predators and rival
males, being very aggressive in the mating season. Each female lays two blue-gray
eggs which hatch in 19 days, with the hatchlings fledging in 36 days. Life span
is about 10 years.
An allopatric speciation resulted in appearing of closely related species of
different ecology – lesser,
or mountain guiraguacu (Guiraguacu andinus). The populations
of this smaller (males 48 cm long, and females 39 cm long) species are numerous
in the tropical forests near the Andes. Males of this species have a harem of
5-7 females. This smaller species also differs from its larger cousin in appearance:
it has bright yellow beak, with the feathers of the body brown and the feathers
of the tail black. It is also more peaceful and feeds only on fruits and insects
in way similar to the oropendolas of human epoch.
This bird was discovered by João Vitor Coutinho, Brazil.
Sentinel
grackle (Leistes neglecta)
Order: Passerine birds (Passeriformes)
Family: Troupials (Icteridae)
Habitat: pampas of South America.
In Neocene epoch, South America returned to the state in which it was during
a significant part of the Cenozoic: this is an island continent again, lacking
a permanent land connection with all other continents. As a result of this new
isolation, its fauna began to accumulate differences from other continents again;
for example, large representatives
of caviomorph rodents live in the Pampas. Among the predators of the Pampas
birds dominate – hunting herons,
which hunt various animals, including large
cursorial rodents. Hunting herons themselves should beware of even larger
birds, like akatu, which
is one of the largest flying birds of Neocene world. But most South American
birds have a more familiar appearance, although they have acquired new behavioral
features. Sentinel grackle is one of these birds.
Outwardly, this feathered creature is not too different from other tropical
birds: it is about 20 cm long, thickly built, with an elongated pointed beak.
The background color of the plumage is black, but the plumage of the wings is
brightly colored: the secondary feathers are striated black and yellow. Males
are brighter than females – they additionally have chains of yellow spots along
the posterior edge of the primary feathers. Only the tail feathers of birds
of both sexes are white: these birds use them to signalize to their four-legged
neighbors.
Legs of the sentinel grackle are long, but wings are relatively short, with
blunted tips – this bird flies reluctantly and prefers to get its prey, various
invertebrates, on the ground rather than in the air. Therefore, outside of the
nesting time, the sentinel grackle follows the herds of various animals of the
South American Pampas and catches all the smaller animals that large animals
scare off in the grass when moving. Sometimes the bird even rides on their backs
to catch small animals from above, not from below. In return for this service,
the grackle notifies its partners about the approach of a predator from the
sky or from the grassy thickets, hence the name of this species.
The courtship season of this bird begins at the end of the dry season, when
males begin to notify females of its species with mating calls, signaling his
readiness for nesting. Females evaluate males by volume, duration and strength
of calls, and choose the best males with the best songs, although the singing
of these birds is rather monotonous and non-melodic. The nest of these birds
is woven from grass; it looks like a ball with a long “sleeve” of the entrance
and is attached to the branches of local trees, higher from the ground and away
from predators – small mammals and snakes that hunt these birds. In the nest,
there are usually 4 eggs with a plain pale blue shell. Only the female incubates
them for 2 weeks, and the male feeds her at this time. The nestlings stay in
the nest for about three weeks, after which they leave it forever and keep close
to their parents for the same amount of time. When they leave their parents
and begin to live an independent life, the pair breaks up – these birds form
pairs only for one nesting season, and every year the pair is formed anew.
In the juvenile plumage of birds, the yellow color on the wings is replaced
by brown, and only the tips of the tail feathers have a white color. Sexual
maturity comes the next year, at which time the birds get adult coloring. Sentinel
grackle can live for about 10-11 years, but, as a rule, it dies much earlier.
This bird species was discovered by Bhut, the forum member.
Godelo
(Molothrus mimus)
Order: Passerine birds (Passeriformes)
Family: Troupials (Icteridae)
Habitat: tropical South America, grasslands.
The human actions during the Holocene caused the decline of many species. However
the species that were resilient enough survived, with their descendants now
living in the Neocene. The shiny cowbird (Molothrus bonariensis) survived the
anthropogenic onslaught and in the Neocene gave rise to a new species, the godelo.
This word was used as the name of their ancestor in some regions of Brazil.
Like their ancestors, the godelo is an obligate brood parasite, laying its eggs
in the nests of many other bird species such as the white-throated
obrero. This species inhabits most of tropical South America, being more
common in open areas.
The godelo has 17 cm of length with a wingspan of 32 cm. This species is sexually
dismorphic; the male is black with purple-blue iridescent feathers. The females
have reddish-brown upperparts and pale brown underparts. Their appearance is
similar to that of an obrero. This characteristic, together with their ability
of mimicking the song of other birds, allow them to approach their colonies
to lay eggs without being attacked by these furnarids. This ability isn’t only
used with obreros, but also to scare other hosts from their nests. To do that
they will copy the vocalization of birds of prey. The beak and legs are dark
gray.
This species has an omnivorous diet, feeding mainly on insects and seeds, but
occasionally eats fruits and flowers. It can also feed on ticks on large
mammals.
The godelo do not form monogamous pairs. They have a promiscuous mating system
where individuals will copulate with many different mates. During courtship,
the male performs a song while circling a female, and when the song is finished
they bow to their prospective mate. This bow is a display used in both mating
rituals and as a show of aggression toward other males. It consists of the male
ruffling his feathers while arching his wings and lowering his tail. The display
is performed either on the ground, in a tree, or while flying. Following a successful
mating display, the pair will copulate once.
These birds are obligate brood parasites, with adult females laying their eggs
in the nests of other species and their offspring relying entirely on their
hosts for parental care. They are generalists, and have several different host
species, including the white-throated obrero. As a host generalist, their young
are non-mimetic, meaning they do not attempt to replicate the behaviours of
host chicks like a host specialist species might.
The eggs are similar to that of their hosts, but do vary in shape. Godelos can
lay 6 to 12 eggs, but usually one pair will lay only 2 eggs in the host nest.
The eggs have a short incubation period of about 12-13 days. Even if they don’t
destroy the remaining eggs of their foster parents, the chicks will vocalize
so much that all the food and attention will end up going to them and their
siblings will have little sustenance.
Female godelos do not build nests, as they rely on their hosts to care for their
offspring, but they will preferentially select hosts that build enclosed nests
such as nests built in cavities. They will look for host nests both actively,
and by silently watching for hosts. When a host nest is found, they will flush
the host away from the nest by noisily flying around the area. When dealing
with the obrero, the female will wait until the nest is left unattended to approach
it. Individual godelos may lay their eggs across many different nests. They
reach sexual maturity with 6 months and they have a lifespan of 9 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Paradise
false-cuckoo (Pseudococcyzus cirrhoventer)
Order: Passerine birds (Passeriformes)
Family: False cuckoos (Pseudococcyzidae)
Habitat: Atlantic forests of South America.
During the Holocene, humans caused many impacts on nature. However the survivors
give rise in the Neocene to new species. One case is the Genus Pseudococcyzus,
the false cuckoo. This genus is part of the new Family Pseudococcyzidae in the
order Passeriformes. The type species of this group is the paradise false cuckoo
that lives in the Atlantic forests of South America. These birds are brood parasites
like the cuckoos and descend from birds of Icteridae family having the same
feature of breeding behaviour. Other characteristic of these birds is their
ability to mimic the sound of other birds.
The paradise false-cuckoo is a sexually dismorphic bird. The male is large with
44 cm of length and 57 cm of wingspan. Half of their size is due to their long
wide forked tail. The plumage is iridescent blue as are most of the upperparts,
the wings are bronze-green with blue primaries, the throat and upper breast
are glossy purple, and the tail is glossy blue-green. The middle breast and
underparts are orange-buff. The female is smaller, with 37 cm of length and
a wingspan of 48 cm. All females of this genus have the same plumage. The head,
upperparts and tail of female birds are rusty brown, while their throat and
underparts are white. The tail of females is short and less wide than that of
the males. Young males are similar to females until reaching sexual maturity
with 2 years. Beaks and legs in both sexes are grey colored.
These birds are omnivorous, consuming a variety of insects, caterpillars, eggs
and small vertebrates. Adults feed mainly on fruit. They will sometimes defend
fruiting trees that they forage in and chase away other frugivores.
All species of false-cuckoos are brood parasites. They form pairs during the
breeding season which occurs during the end of the dry season. The male will
attract the female copying the vocalization of other birds and will proceed
to give her food. The pair will work together when the female needs to lay the
eggs. The male will stay near the area of the host nest and will start to mimic
sounds of predatory birds and attack the nesting bird. This spook the host,
making it flees of the nest, allowing the female to lay its eggs. The female
lay one egg per nest, she will consume one of the original eggs. The female
lays 4 eggs on different birds’ nests. Birds of the Families Cotingidae and
Thraupidae are chosen by the paradise false-cuckoo. The eggs are incubated by
the foster parents for 16 days. When it hatches, the young false-cuckoo will
not expel its foster siblings from the nest. The chick fledges with 28 days,
but stays being fed by the foster parents for more than 10 days. Paradise false-cuckoo
has a lifespan of 17 years.
Other species in the genus Pseudococcyzus:
Emerald
false-cuckoo (Pseudococcyzus amazonicus)
Living in the Amazon rainforest, males of this species have 46 cm and a wingspan
of 60 cm. Its plumage is mostly metallic blue, with exception of the belly that
is yellow. The female has 39 cm of length and a wingspan of 57 cm. The preferred
hosts for its eggs are birds of the families Icteridae and Cotingidae.
Andean
false-cuckoo (Pseudococcyzus andinus)
Living in the forests near the foothills of the Andes, males of this species
have 42 cm of length and a wingspan of 56 cm. Its plumage is mostly metallic
green, with the exception of the underbelly that is metallic purple. The females
have 34 cm of length and a wingspan of 48 cm. The preferred hosts for its eggs
are birds of the families Icteridae and Cotingidae.
This bird species was discovered by João Vitor Coutinho, Brazil.
Marsh
warbler (Geothlypis palustris)
Order: Passerine birds (Passeriformes)
Family: New World warblers (Parulidae)
Habitat: wetlands, swampy forests of North America.

Picture by Pavel Volkov
Neocene is the time of the emergence of new species and often
incredible steps of evolution. Freed forever from the pressure of human activity
and taking advantage of the warming after the Holocene-Neocene glaciation, animals
and plants often evolved in a variety of directions, but among them there were
always representatives of conservative groups, which survived, preserving their
universality and evolutionary flexibility. Such groups include New World warblers,
a family of songbirds endemic to the New World. Marsh warbler is one of the
widespread common bird species in the temperate zone of the continent.
Marsh warbler has a fairly standard appearance: it is a bird about 15-17 cm
long, with a thin pointed beak and relatively long legs. The back of adult bird
is greenish-gray, the lower part of the body is dark green; male has a wide
reddish mask on its face, shaded with white from below. Female almost lacks
mask: there are only narrow reddish “eyebrows” stretching from the base of the
beak to the ear above the eye. The female’s cheeks are greenish-white.
The basis of the diet of this species is insects; in late summer and autumn,
unripe plant seeds are added to them. Marsh warbler is a migratory bird that
flies to the south of North America for the winter. It returns to its nesting
sites in the second half of spring. Males return first and start looking for
suitable sites for nests – they prefer vast expanses of swamps or slow-flowing
rivers with swampy banks overgrown with reeds or other tall marsh grasses. Having
found a suitable place, male begins to build the basis for the nest and warns
rivals of his presence with songs. The females fly later, inspect the males
and their nests, choose suitable mates, then finish the nests and lay eggs there.
Marsh warblers nest in the reeds. Despite of its small size, this bird is quite
strong and can bend and weave together several reeds. Birds build a deep conical
nest where the female lays eggs, usually 3-4, with a pale green speckled shell.
The incubating bird sits on the eggs with its head and tail up. After two weeks,
the nestlings hatch. Parents feed them with insects and other invertebrates,
and in the first half of summer the juveniles already leave the nests. For several
days they are fed by their parents, who manage tp raise successfully two broods
per season in the southern part of the range. The sexual maturity of these birds
comes next spring, and the life expectancy is no more than 9 years, but only
a few live up to this age.
Marshwarbler has quite a lot of enemies: birds of prey hunt adult birds, especially
during autumn and spring migrations, and their clutches become the object of
nesting parasitism of cuckoos and other birds.
This bird species was discovered by Bhut, the forum member.
Black-headed
cardinal (Cardinalis nigrocephalus)
Order: Passerine birds (Passeriformes)
Family: Cardinal-grosbeaks (Cardinalidae)
Habitat: tropical forests of Northern and Northeastern South America.
The Ice Age that occurred during the transition of Holocene to Neocene forced
some species to migrate to southern regions. Certain species, even with the
end of the Ice Age, remained in their new localities giving rise to new species.
One of these new species was the black-headed cardinal, a descendant of the
northern cardinal (Cardinalis cardinalis).
The black-headed cardinal is a mid-sized songbird with a body length of 21-24
cm and a wingspan of 25-31 cm. The male is slightly larger than the female.
The adult male is a brilliant crimson red color with a black head and a reddish
orange crest. The color becomes duller and darker on his back and wings. The
female is brown, with mostly grayish-brown tones and a slight reddish tint on
the wings, the crest, and the tail feathers. The females head is dark grey.
Both sexes possess prominent raised crests and bright coral-colored beaks. The
beak is cone-shaped and strong. Young birds, of both sexes, show coloring similar
to the adult female until their first year, when they reach sexual maturity.
Their diet consists mainly of seeds and fruits. It will also consume insects,
including beetles, cicadas, grasshoppers, and snails; it feeds its young almost
exclusively on insect
The black-headed cardinal is a territorial song bird. The male sings in a loud,
clear whistle from the top of a tree or another high location to defend his
territory. He will chase off other males entering his territory. Pairs mate
for life and stay together year-round. Mated pairs sing together before nesting.
During courtship they participate in a bonding behavior where the male collects
food and brings it to the female, feeding her beak-to-beak. If the mating is
successful, this mate-feeding may continue throughout the period of incubation.
The male bring nest material to the female, who does most of the building. The
female builds a cup nest in a well-concealed spot in a tree. Three or four eggs
are laid and incubated by the female for 13 to 14 days. Young fledge 11 to 12
days after hatching. Two to three, and even four, broods are raised each year.
The male cares for and feeds each brood as the female incubates the next clutch
of eggs. Their lifespan is 12 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
White-chested
bunting (Passerina leucopecta)
Order: Passerine birds (Passeriformes)
Family: Cardinal-grosbeaks (Cardinalidae)
Habitat: forests of Southeastern South America.
The Ice Age that occurred during the transition of Holocene to Neocene forced
some species to migrate to southern regions. Certain species, even with the
end of the Ice Age, remained in their new localities giving rise to new species.
One of these new species was the white-chested bunting, a descendant of hybrids
between the indigo bunting (Passerina cyanea) and the lazuli bunting (Passerina
amoena). It lives in the forests of Southeastern South America.
The white-chested bunting is a small songbird. It measures 13 cm long, with
a wingspan of 21 cm. The adult males have a vibrant cerulean blue plumage in
their head and back. The wings and tail are black with cerulean blue edges.
Their chest and belly is white. The adult female is brown on the upperparts
and lighter brown on the underparts, with a grey belly. Both sexes have white
wingbars. The beak is short and conical. In the adult female, the beak is light
brown tinged with blue, and in the adult male the upper half is brownish-black
while the lower is light blue. The feet and legs are black or gray. The juveniles’
plumage is similar to the female until they reach sexual maturity at 2 months,
at this age the young males acquire the blue coloration of the adults.
These birds eat mostly seeds and insects. They may feed conspicuously on the
ground or in bushes, but singing males are often very elusive in treetops.
These birds are generally monogamous but not always faithful to their partner.
They make a loose cup nest of grasses and rootlets placed in a low tree. It
lays three or four pale blue eggs, which are incubated for 12 to 13 days. Chicks
fledge 10 to 12 days after hatching. Most pairs raise two broods per year, and
the male may feed newly fledged young while the females incubate the next clutch
of eggs. The lifespan of this species is 3-4 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Green
bee-eater tanager (Apiphagus viridis)
Order: Passerine birds (Passeriformes)
Family: Tanagers (Thraupidae)
Habitat: tropical woodlands and savannas of South America.

Picture by João Vitor Coutinho

Picture by Pavel Volkov
The introduction of species by the man caused many impacts
over the environment. In some cases these impacts proved to be not negative.
The genus Apiphagus, in the family Thraupidae evolved in Neocene. The type species
of this genus is green bee-eater tanager, a bird that lives in the savannas
and woodlands of Tropical South America.
The green bee-eater tanager has 23 cm of length and 25 cm of wingspan. Adult
males have bright green plumage with black wings and tail, while the females
are greyish-green with dark gray wings and tail. The bill of green bee-eater
tanager is curved, long and ending in a sharp point. Young birds of both sexes
look similar to the female until reaching sexual maturity in 7 months.
As their name says, these birds are adapted to eat bees. Green bee-eater tanagers
catch these insects, seizing their abdomens. To kill bee, bird strikes it against
branch or rock. After that, it removes bee’s sting using the beak, and finally
eats its prey. Not only green bee-eater tanager, but all species in the genus
Apiphagus have an ecological relationship with jaguape
(mammal of genus Manducomelifer). When these mammals approach a bee nest, flocks
of bee-eater tanagers fly closer to them, vocalizing to attract jaguape. When
bees begin attacking the invader, these birds rush and catch the insects. In
addition to bees, these passerines can eat wasps (with the same method used
to kill bees) and other insects, but they also feed on fruits and flowers.
Green bee-eater tanagers are monogamous; male vocalizes to attract the female
and once the pair is formed, they will stay together until death of one them.
The nesting season takes place all the year round, with the pair building a
nest made of small roots, mosses and leaf petioles. It is hidden in dense epiphytic
vegetation in forked tree trunk. Female lays 2 to 3 eggs of white color patterned
brown speckles and is responsible for the incubation that lasts of 14 days.
The couple feeds the chicks, which leave the nest at the age of 20 days. The
sexual maturity comes at the age of 7-8 month, and lifespan of these birds is
up to 11 years.
There are some more species in the genus Apiphagus:
Amazonian
bee-eater tanager (Apiphagus caeruleus) lives in Amazon rainforest.
This bird is 22 cm long and its wingspan is 24 cm. Males are bright blue with
black primaries, secondaries, tertials and tail. Female is grayish-blue with
dark grey wings and tail.
Turquoise
bee-eater tanager (Apiphagus atlanticus) inhabiting the forests in
the Atlantic coast of South America, is 23 cm long with a wingspan of 24 cm.
Males are bright turquoise with black feathers on wings and tail. Females are
grayish-green with dark grey wings and tail. The range of this species overlaps
that of the green bee-eater tanager in the regions where the Atlantic forests
transit to savannas. In these areas hybrids of intermediate coloration appear.
Andean
bee-eater tanager (Apiphagus andinus) lives in elevated areas of the
Andes. This bird is 21 cm long and has a wingspan of 23 cm. The males are yellow
with dark gray feathers on wings and tail. Female is brown with shades of yellow,
its wings and tail are dark brown. Its plumage is much denser than at flatland
forms. In winter it migrates to warmer lowlands.
Fiery
bee-eater tanager (Apiphagus australis) lives in forests of southern
part of South America. This bird grows to 22 cm with a wingspan of 24 cm. Males
have reddish-orange plumage with the exception of wings and tail which are black.
Females are pale grey with some shades of red. During the winter these birds
migrate to northern areas, reaching the savannas of central part of South America.
This bird species was discovered by João Vitor Coutinho, Brazil
Emerald
sibite (Carpophagus viridis)
Order: Passerine birds (Passeriformes)
Family: Tanagers (Thraupidae)
Habitat: South America, Atlantic forests.

Picture by Pavel Volkov
During the Holocene, humans created gardens in the cities,
which attracted many animals. One of these animals was the bananaquit (Coereba
flaveola). These birds were common across all of tropical and warm areas of
the American continent. Bannaquits prospered during the Holocene and survived
in to the Neocene, giving rise to the Genus Carpophagus, the sibites (this word
was one of its popular names on Brazil). The type species of the genus is the
emerald sibite, which lives in the Atlantic forests of South America.
These birds have 11 cm of length and a wingspan of 20 cm. All species of sibite
are sexually dysmorphic, males are bright colored. In the case of the emerald
sibite, males are bright green with black around the eyes, on the throat, back,
wings and tail. Females and juveniles are similar, being dull green with grey
on the throat, back, wings and tail. Both sexes have grey legs and feet. The
beak is grey and sharp.
The sibites eat mostly fruits and nectar, rarely feeding on small invertebrates.
They use their sharp beak to perforate the peel of large fruits and proceed
to consume the pulp. When eating nectar, these birds pierces the flowers from
the side, taking the nectar without pollinating the plant. Sometimes small insects
are consumed.
This species is monogamous, mating for life. It builds a spherical lined nest
with a side entrance hole, laying up to three spotted eggs, which are incubated
solely by the female for 20 days. The male stay away from the nest most of the
time (because of its colorful feathers) defending the territory and return to
bring food for the female. These birds breed all year regardless of season and
build new nests throughout the year. The young fledge usually with 18 days and
reach sexual maturity with 4 months. The lifespan of this species is of 8 years.
Other species in the genus Carpohagus:
Amazonian
sibite (Carpophagus amazonicus)
Living in the Amazon rainforest, this bird has a similar size to the emerald
sibite. The male of this species is bright yellow with black around the eyes,
on the throat, back, wings and tail. The females and juveniles have dull colors.
Dwarf sibite
(Carpophagus nanus)
This is the smallest species on the genus, with only 9 cm of length and 17 cm
of wingspan. This is an inhabitant of the forests and woodlands near the Andes.
The male is bright greenish yellow with black around the eyes, on the throat,
back, wings and tail. The females and young have dull colors.
The genus Carpophagus belong to the family Thraupidae and together with the
genera Sentrentriornithes (northern sibites) and Neocoereba (insular sibite)
they form the subfamily Carpophagiinae.
Species in the genus Setentriornithes:
Black-headed
northern sibite (Setentriornithes nigrocephalus)
Living in the tropical forests of North America, this bird has 13 cm of length
and a wingspan of 23 cm. The males have bright yellow feathers at the throat,
abdomen and belly. They also have dark plumage on the head, back, wings and
tail with the presence of white around the eyes. Females and young are pale
grey with shadows of yellow in the underparts.
Black
northern sibite (Setentriornithes nigrus)
Living in the subtropical forests of the Florida Peninsula, this bird has 14
cm of length and a wingspan of 25 cm. The males are completely black with white
around the eyes. They also have a bright yellow rump. The females and juveniles
are pale grey.
Species in the genus Neocoereba:
Insular
sibite (Neocoereba insularis)
The only member of it genus, the insular sibite lives in the forests of Great
Antigua. It has 10 cm of length and a wingspan of 18 cm. Differently of sibites
of other genera, the insular sibites don’t have sexual dimorphism, both the
male and female have whitish grey heads, back, wings and tail. The belly and
underpart of both sexes is yellow.
This bird species was discovered by João Vitor Coutinho, Brazil.
Atarapan
symphonic tanager (Paratanagra atarapaensis)
Order: Passerine birds (Passeriformes)
Family: Tanagers (Thraupidae)
Habitat: Atarapa islands, tropical forests.

With the passing of the Holocene to the Neocene, the movements
of tectonic plates caused the changes in the disposition of the continents and
the appearance of new islands. The islands of Eonesia are some of these that
appeared during this new geological period. These new habitats gave opportunity
for the animals that could reach them. There they found new resources and evolved
into new species. The symphonic tanager of the Genus Paratangara, part of the
Family Thraupidae, is endemic to the islands. Their ancestors reached Eonesia
coming from Central America. The type species of the genus is the Atarapan symphonic
tanager, an endemic species to the forests of the Atarapa islands.
The Atarapan symphonic tanagers have 15 cm of length and 27 cm of wingspan.
These birds have a bright red plumage on the head, neck, back and rump. The
chest and belly are bright green. The wings, ear-coverts and tail is black.
While there wasn’t a great sexual dismorphism, the females' plumage is a bit
duller. The beak and legs are dark gray.
These birds eat mainly fruits and small buds. The fruits are usually swallowed
whole, with the seeds being regurgitated. Insects are also taken, mainly from
the underside of branches.
The Atarapan symphonic tanager is usually found in pairs, but sometimes small
groups are formed. The males have elaborate songs that they use to attract the
females. This species is monogamous, with pairs staying together for the rest
of their lives. While they are mostly silent, the females will vocalize with
the males. They form a "duet" that is used to strengthen the pair's
bond. When they are in a flock, the birds will vocalize in such a way that reminds
of a natural symphony. The parents work together to build the nest, but only
the female incubates them. One to three, usually two, dark-marked whitish to
gray-green eggs are laid in a deep cup nest in a high tree fork. Incubation
lasts for 14 days with the chicks fledging after 17 days. They reach sexual
maturity with 11 months and have a lifespan of 12 years.
The Genus Paratangara have other two species that also inhabits Eonesia:
Burotu
symphonic tanager (Paratanagra burotuensis)
This species inhabits the Burotu islands. They have 13 cm of length and 25 cm
of wingspan. These birds have a bright orange plumage on the head, neck, back
and rump. The chest and belly are bright green. The wings, ear-coverts and tail
is black. Their song is also different of the Atarapan species.
Fisaga
symphonic tanager (Paratanagra fisagaensis)
This species inhabits the Fisaga islands. They have 16 cm of length and 31 cm
of wingspan. These birds have a bright yellow plumage on the head, neck, back
and rump. The chest and belly are bright bluish-green in the males, with the
females being a duller green. The wings, ear-coverts and tail is black. Their
song is also different from the other previous species.
This bird species was discovered by João Vitor Coutinho, Brazil.
Common
cabocolino, or white-winged seedeater (Neosporophila albopterus)
Order: Passerine birds (Passeriformes)
Family: Tanagers (Thraupidae)
Habitat: grasslands and wetlands in tropical areas of South America.

Picture by Pavel Volkov
The human actions during the Holocene caused the decline of
many species. However some species survived and in the Neocene they gave rise
to new species and genera. A new genus that appeared was the Neosporophila,
a direct descendant of birds of the Genus Sporophila that survived the anthropogenic
onslaught. The type species of this genus is the common cabocolino, an inhabitant
of open fields and marshy areas with tall grass in tropical South America, from
Amazon to the plains. The word cabocolino was the common name given by the people
from the Brazilian states of Pará and Ceará to some Sporophila.
The common cabocolino has 10 cm of length and a wingspan of 32 cm. The males
have a mostly cinnamon plumage with tail, rump and coverts black. The primaries
and secondaries feathers are white. The females are olive-brown in the upperparts
and yellowish-white in the lower parts, such plumage is shared by all females
of the genus. Their beak is black while the legs are light gray.
These birds feed mostly on seeds, and occasionally berries and insects. It often
forages on herbaceous plants, and less often on the ground. The common cabocolino
will sometimes participate in mixed flocks when foraging.
The common cabocolino is a gregarious bird, forming small flocks when outside
of the nesting season. They are monogamous, with the pair staying close to each
other even when they are part of flocks. The nesting season starts at the beginning
of spring and lasts until the middle of summer, with at least 2 to 3 clutches
during this period. Both parents work together to build a cup-shaped nest made
of grasses in a bush or a small tree. The female lays 2 to 3 eggs, which are
incubated solely by her for 13 days. The male will remain nearby protecting
the nest's area from intruders and bringing food for the female. The chicks
are fed by both parents. They fledge with 14 days and are still fed by their
parents for more two weeks. They reach sexual maturity with 10 months and have
a lifespan of 9 years.
Like their ancestors of the genus Sporophila, they have a great evolutive radiation
with several species inhabiting different ecosystems of the continent. One of
these species is the thick-billed
cabocolino, or thick-billed seedeater (Neosporophila crassirhynchus).
This species is endemic to the North areas of South America, from coastal areas
to the Northern side of the Amazon river. They have 13 cm of length and a wingspan
of 37 cm. The males have a mostly black plumage, with a chestnut-brown chest
and belly. The female has the typical olive-brown plumage of the genus. Their
beak is white and thick, allowing them to crush hard seeds that other seedeaters
can't. Another representative of the genus is the Southern
cabocolino, or Southern seedeater (Neosporophila rufoventer).
This species inhabits the plains and prairies of Southern South America, migrating
to the savannas in the center of the continent during the winter. They have
10 cm of length and a wingspan of 32 cm. The males have a cinnamon chest with
head, rump, wings and tails with blueish-gray. The cerrado
cabocolino, or savanna seedeater (Neosporophila savanna)
is a species endemic to the savannas of Central South America. They have 11
cm of length with 31 cm of wingspan. The males have chestnut-brown chest and
belly, while the rest of the body is black. The white-throated
cabocolino, or white-throated seedeater (Neosporophila albogularis)
is another species in the genus. They are endemic to the areas of transition
between the Savanna and Atlantic forests of Northeastern South America. They
have 10 cm of length with 30 cm of wingspan. The males have a cinnamon chest
and belly, while the rest of the body is black. They also have a distinctive
white throat. Another species of the genus is the collared
cabocolino, or collared seedeater (Neosporophila collaris).
This bird inhabits the savannas in Northeastern South America. They have 11
cm of length with a 32 cm wingspan. The male has a chestnut belly with the rest
of the plumage black and a characteristic white plumage around the neck, similar
to a necklace. The range of this bird overlaps with the white-throated cabocolino
and the cerrado cabocolino, resulting in hybridization.
This bird species was discovered by João Vitor Coutinho, Brazil.
Noronha
cardinal (Paroaria noronhensis)
Order: Passerine birds (Passeriformes)
Family: Tanagers (Thraupidae)
Habitat: forests of the archipelago of Fernando de Noronha.

Picture by Pavel Volkov
During the Holocene, the man introduced many species to other
continents. The Archipelago of Fernando de Noronha received many exotic species,
not only through the humans but also that arrived there by themselves. The red-cowled
cardinal (Paroaria dominicana) was introduced in the Archipelago where they
found good conditions to their survival. Due to that, they survived and gave
origin to the Noronha cardinal in the Neocene. This species is endemic to the
Archipelago, with most of the population being found in the largest island.
The Noronha cardinal has 14 cm of length and 31 cm of wingspan. Both sexes have
similar plumage, with a bright scarlet head and throat, with black ear-coverts
that give the appearance of masks. They have a short crest that is raised by
the birds when excited. Belly, breast and undertail are white, with a gray back,
wings, and tail. Wing coverts are gray, but the primaries, secondaries, and
rectrices show a darker gray. Juveniles are similar to the adults, but they
show a dull brownish orange head and bib. The beak and legs are pale gray.
This species feeds mainly on seeds, fruits, insects and other small arthropods.
They generally search for food in the ground, forming pairs or small groups.
The Noronha cardinal is a monogamous bird, with the pairs staying together while
they are in flock. During the breeding season they live strictly in pairs, being
extremely faithful to a territory, defended by the male. Each brood usually
has between 2 and 3 eggs, with 2 to 4 broods per season. The chicks are born
after 13 days and are fed by both parents. They fledge with 21 day and reaches
sexual maturity at 10 months. The lifespan of the Noronha cardinal is 4 years.
This bird species was discovered by João Vitor Coutinho, Brazil.
Red-billed
bullfinch (Parapyrrhula erythroramphus)
Order: Passerine birds (Passeriformes)
Family: Tanagers (Thraupidae)
Habitat: South America, Andean Plateau.

Picture by Pavel Volkov
The human actions during the Holocene caused the decline of
many species. However the species that were resilient enough survived, with
their descendants now living in the Neocene. Even during the Holocene, the family
Thraupidae always showed a great morphological diversity. Its members evolved
to fill the most different niches. From the tropical rainforests, to the Andes,
the members of this family could be seen in all of them. In the cold Andean
Plateau region, the red-billed bullfinch appeared. This species has a similar
lifestyle to the true bullfinches from Eurasia (Genus Pyrrhula).
The red-billed bullfinch is a bulky bull-headed bird. They have 13 cm of length
and a wingspan of 29 cm. The males have a slaty gray plumage with black wings,
back and head. Their short and thick bills are bright red. The females are pale
gray with patches of brown in the wings and head. Their bill is pale pink. Both
sexes have dark gray legs.
They feed mainly on seeds and buds, but can occasionally eat invertebrates.
When winters arrive in the Andean Plateau, these birds engages in altitudinal
movements by winter, traveling to as low as 1,800 m or even to 1,060 m.
The red-billed bullfinch lives in pairs or small flocks that usually perch near
the tops of bushes, but forage often in lower vegetation or even on the ground.
They fly in tight flocks in a fast, direct but sometimes undulating flying style.
Their nesting season starts early in the spring and until the end of summer,
the pairs can have 3 to 5 clutches. The nests are built in cavities, usually
rock crevices. The female lays 3 brown-speckled pale blue eggs, which are incubated
solely by her for 11 days. The chicks fledge after 4 weeks, but can remain with
parents until the migration. During this period, they help their parents take
care of the other clutches. This species reaches sexual maturity with 6 months,
but mist only start to nest in the next spring. They have a lifespan of 7 years.
This bird species was discovered by João Vitor Coutinho, Brazil.