

Bestiarium of
the future - Mammals
|
 |
| In this page descriptions of various animals and plants not included to basic chapters are gathered. The wanishing of the description from this part means that you just must search for new chapter in English version of "Tour to Neocene". |  |
|
Mammals |
Mammals
|
Wood
echidna (Dendroglossus scandens)
Order: Monotremes (Monotremata)
Family: Echidnas (Tachyglossidae)
Habitat: tropical rainforests of Meganesia.

Picture by Alexander Smyslov
Climatic change in Neocene has strongly affected upon life of various Earth
inhabitants. Some of them had to abandon their habitats, other oness had
died out, and third ones had received great opportunities for evolution
and had
turned to prospering species. Humidifying of climate has caused reduction
of desert areas and expansion of tropical rainforests zone. Some of inhabitants
of open spaces have to found convenient places for life in forests, and than
evolved to true wood inhabitants. In Australia which even after intervention
of humans has kept an originality of fauna and flora, amazing abilities to
adaptation were shown by echidna - the inhabitant of savannas and light forests.
It had turned to true wood inhabitant, and besides it had changed to the
excellent
tree-climber.
To see such animal on the tree is not so such unusual thing as it seems at
first sight. Among tree-climbers in different continents there were kangaroo,
porcupines and even ground tortoises, not to mention toads and frogs. Therefore
the wood echidna of Neocene epoch, sauntering along tree branches, does not
seem as something unusual on their background.
This is rather large animal: its body length is up to 70 cm, heads with long
proboscis is about 30 cm long. Slowly moving in tree crones wood echidna
outwardly resembles a hybrid of bear and anteater. The wood echidna had kept
characteristic
shape of these animals, but had got additional features of adaptation to
clambering habit of life. Its paws which are quickly digging off the firm
dry ground,
now equally easily break rotten wood or strong cover of termitary. Claws
becoming more bent cling to bark and branches, permitting the animal to make
walks at
30-meter height so easily, as if on firm ground. For more tenacious grip
thumbs on forepaws began to oppose partly to fingers. On hinder legs feet
are turned
back, but it at all does not prevent this animal to move. Even on the contrary
to it, using such feet it is easier to climb down headfirst along the tree
trunk. The little toe on hinder legs became a certain analogue of the thumb:
it can be opposed to other toes too and serves for branch gripping. This
animal is very strong: by paws the echidna breaks trunks of trees beginning
to rot
and turns out from trunk thick boughs, searching for ants and termites.
Body of the wood echidna is massive and heavy-build. The tail of animal is
very short; it serves practically only for fat accumulation.
The muzzle of this echidna is long, tiny mouth opens on its tip. Main food
of animal includes insects, usually ants and termites, and the main instrument
of their catching is the long tongue covered with sticky saliva. Except for
ants and termites, the echidna eats larvae of beetles searching them in pierced
trunks of dead trees, and also insects living among epiphytic plants. The
wood echidna is able even to plunder trap leaves of insectivorous plants,
catching
from them insects floating in digestive liquid secreted by leaves. Sometimes
it simply drinks this digestive juice with insects. One more dainty for the
echidna includes bird eggs. Having found a nest, the echidna drives hatching
bird out from it, accurately pierces an egg-shell by claw tip and licks by
tongue the egg completely. The animal exhausts all eggs in nest though sometimes
it is attacked not only by nest owners, but also by their numerous neighbours
have flied to help. But what can birds make with the prickly robber surpassing
them many times in weight?
Eyes of the wood echidna are very small, and auricles are not present at
all. This animal is short-sighted, and the prickly armor perfectly protects
it from
any predator of Australian and New Guinean woods, therefore the wood echidna
can not react (at least, externally) to loud alarm cries of wood inhabitants.
But sense of smell and touch at it are very sharp: the animal easily will
feel, as on branch any bug or spider creeps, not seeing it at all.
Colouring of animal began more contrast, than at its ancestor: needles are
ivory-colored, top of body is light, and stomach is dark, covered with short
thin fur. At the wood echidna there is very dense and thick skin, therefore
stings of ants and termites are not harmful to it. Even army ants killing
all alive on their way, do not harm this phlegmatic animal: unless they will
inflict
some stings to sensitive proboscis or between toes. The animal has natural
immunity to their poison, therefore the echidna does not avoid their attacks:
such attack does not harm to it, and in it there is even such benefit – ants
at the same time kill blood-sucking insects in wool of the huge echidna.
If insects (and also larger enemies) appear too importunate, the wood echidna
sprinkles out a jet of musk liquid from special glands. Its sickening smell
easily will drive off the most persevering predator.
The wood echidna is real wood tramp. It has no precisely bordered territory,
and does not protect residence from neighbours. Some animals can peacefully
food on the same tree, and even to ravage one termitary in common, not paying
attention against each other. During such meetings pairing happens more often.
Courtship ritual at the wood echidna is not present, and the breeding season
lasts all the year round.
Not changing traditions of ancestors, wood echidna lays eggs. In its clutch
there are 1 - 2 rather large eggs (with small chicken egg-sized ones), covered
with white skinny shell. During formation of eggs because of hormonal influence
the cross skin plica on the stomach of the female is increasing and turning
to brood pouch. The female lays eggs, lying on back so that the tail appeared
above a head. Then the egg rolls down to the pouch itself from oviduct. The
incubating lasts about three weeks. Cubs hatch underdeveloped, looking more
similar to embryons. They lick milk, squeezing from pouch walls. Usually
from two cubs only one cub survives. It grows quickly, and at the age of
two months
(when it leaves mother’s pouch), the cub weighs already about half-kilogrann.
It is covered with grey naked skin, but at it the short proboscis characteristic
for these animals is visible. Claws of it still soft and short but after
the cub has left the pouch, it literally starts to increase from different
directions:
practically simultaneously at it needles, wool and claws grow. While the
cub is still small, its unique protection is musk liquid with which it excretes
plentifully at a fright. The female regularly visits it (approximately twice
per day) and feeds with milk. For this purpose it lies down on back in forked
crown of branches, and the cub licks off the milk flowing down from pouch
to
stomach. But gradually the cub also starts to catch insects. At the age of
three months the cub already starts to wander in wood canopy with mother,
studiing to swarm up lianas and to search for insect nests. At the age of
eight months
it reaches length about 70 cm and already is completely independent. At the
age of 20 months the young female already can lay the first egg and this
surprising animal can live up to the age of 60 - 70 years.
Spurred
Echidna (Plectroechidna toxica)
Order: Monotremes (Monotremata)
Family: Echidnas (Tachyglossidae)
Habitat: Forests of northern Meganesia.
The Neocene is, in some ways, much like the Holocene, in other ways not. The
disappearance of humans is notable, but nature has carried on regardless. Some
animals present in the Holocene have diversified, yet stayed fairly similar
to their original ancestor. Some monotremes are a good example of this.
Echidnas of the Neocene are noteworthy for the presence of arboreal,
giant, and even small burrowing
forms. However, many have kept to the original mastery of their kind, the consumption
of social insects. The forests of northern Meganesia hide one such creature,
the Spurred Echidna. Outwardly it is very similar to its ancestor, with the
exception of one remarkable feature, venom spurs. Venom spurs were typical of
almost all primitive mammals, including monotremes, and simple atavisms were
all that was needed to reactivate the vestigial spurs of echidnas into working
ones, like the platypus.
This animal looks similar to the Holocene short-beaked echidna. It possesses
spines slightly shorter than its ancestor, they are a secondary defense, spurs
being a more effective deterrent. It's shape is typical, a squat body with sprawling
limbs, it is about 30 cm long and weighs generally around 2 – 4 kilograms. It
has a pointed rostrum, its fur is dark brown, and partly covers the spines.
Its diet is mostly social ants and termites, but sometimes other insects.
Why has it grown the extra defense of a spur? Possibly because of the presence
of rather large and fairly intelligent predators, that may be able to expose
the animal's underbelly, Marsupial Panther, and the flightless Yagil bird both
do this.
The spur is situated on each ankle, and is long and yellow. When approached
by a predator, Spurred Echidna first produces a loud hiss, almost like a snake
(echidnas of human epoch were only able to make a quiet hiss through the nose).
It can also attack from a distance by spraying venom with the spurs. If the
predator grabs the echidna, it will actively defend by stabbing with spurs.
The venomous sting of the spur is not only very painful, but causes fatigue
and sickness that can last some days.
Breeding takes place during the dry season, when floodwaters are gone, and suitable
dry bedding is available for nest making. Females and males secrete a musky
odor from the cloaca that allows them to find one another. Many males may pursue
a female at once, but she will deny unsuitable males by showing her spines.
As with the modern echidna, the penis is strangely shaped, similar to that of
reptiles. 2 or 3 eggs are layed into her pouch about a month after copulation.
Babies are small, and blind, and are suckled and raised in the nursery burrow.
The milk is rich, and the babies grow relatively quickly, leaving the burrow
when spines and spurs are fully developed.
This mammal is discovered by Timothy Donald Morris, Adelaide, Australia.
Crested
Echidna (Neoglossus spinosa)
Order: Monotremes (Monotremata)
Family: Echidnas (Tachyglossidae)
Habitat: savanna and open forest of central and southeastern Meganesia.

Pictures by Alexander Smyslov
 Monotremes
of the Neocene are considerably more varied than those of the Holocene. Both
platypi and echidna occupy diverse niches, mostly consuming small animals, insects
and forage.
Monotremes
of the Neocene are considerably more varied than those of the Holocene. Both
platypi and echidna occupy diverse niches, mostly consuming small animals, insects
and forage.
The Crested Echidna is a fairly large echidna, being similar in size and shape
to the long-beaked echidna of human times (Zaglossus). It is about 60-90 cm
long, and weighs up to 14 kilograms. It has relatively long, almost erect legs
for an echidna, suitable for walking long distances in search of its food, ants,
termites, and beetle grubs. It has a long snout, and large claws, which aid
it in digging open insect nests, or grub burrows. The most obvious feature of
this animal, however, are its spines. It's back is covered in a large crest
of very long spines, similar to those of a porcupine, such spines can be up
to 40 cm long. Shorter, stouter spines cover the rest of the body, and the tail
has a cluster of fairly long spines too. Its fur is a light, dusty brown, and
the long crest spines are banded in black and white, serving as a warning for
predators. Shorter spines are yellow. rostrum, face, and feet are covered in
black skin.
For this newer kind of echidna, spines have been taken from their initial purpose
of passive defense, to more active vigorous protection similar to some porcupines.
Firstly, at sighting a predator, echidna turns around, erects spines, and makes
abrupt whistling noises through its nose, this is a warning. Then it will confront
a predator, facing backward, with spines pointed at the hunter, and will make
violent thrusting motions and short, backward charges. If the predator gets
too close, these motions will intensify, and the predator may be badly stabbed
by such long spines. Unlike the porcupine, these spines are firmly rooted in
the echidna's thick hide, and do not come off when driven into a predator. In
fact, after stabbing a predator, the echidna will make efforts to extract itself
(spines are not barbed), and retreat.
During the wet season, when plant growth and abundance of insects is best, females
become receptive, and smell musky. Males will not stab each other, when competing
for mates, but posture and shake their spines vigorously. After copulation,
1 large egg is layed into the pouch. The pouch is forward facing, and well developed,
this species does not make a nursery burrow. The female carries the egg, and
then baby, until the young is mobile, and large enough to follow her, spines
grow soon after.
This mammal is discovered by Timothy Donald Morris, Adelaide, Australia.
Starbill
(Astrorhynchus condyluroides)
Order: Monotremes (Monotremata)
Family: Duck-bills (Ornithorhynchidae)
Habitat: lower reaches of rivers of southeast of Meganesia, Flinders Island
in Eyre Gulf.

Pictured by Carlos Pizcueta (Electreel)
colorization by Alexander Smyslov
Climatic changes in Neocene favour to evolution of thermophilic and moistureloving
species. The most part of territory of Australia in Holocene was covered
by droughty deserts. In Neocene this continent had moved to the north and
had
joined to New Guinea forming unite continent Meganesia. Rivers supplying
with water plains of the central part of Meganesia spring from the north
and east
of the continent. The Eastern Meganesia prospers due to large river system
occupying the territory of former Murray River. It runs into extensive shallow-water
Eyre Gulf filled with warmed up brackish water. Expansion of habitats stimulated
the evolution of water inhabitants.
The genealogical branch of monotremes declined till the Cenozoic Era had
received new opportunities in Neocene. The occurrence of productive ecosystems
has allowed
to evolve to the various specialized species of these primitive mammal. The
large piscivorous duck-bill mergotherium
(Mergotherium piscivorus) inhabits
rivers, and in mountain streams its tiny aggressive relative ancistrotherium
(Ancistrotherium radulus) lives. In lower reaches of rivers running into
Eyre Gulf, and also at small Flinders Island, one more descendant of duck-bill,
the starbill, lives.
This species of duck-bills resembles the American star-nosed mole in many
respects – it leads similar way of life, and even has few common features
in appearance.
This is small (rat-sized) animal with powerful forepaws using for digging.
Between toes membranes are advanced; the animal is nice swimmer. At this
animal the swimming membrane on forepaws is reduced, and they are more adapted
to
digging, than forepaws of duck-bill. During the swimming starbill presses
them to the body. Besides the starbill is able to climb on roots in mangrove
thickets,
and often catches ground insects and snails. The tail at starbill is weak
and short – this animal swims with the help of strong hind paws.
The most original part of appearance of starbill is its beak, the family
feature of duck-bills. Beak at this species is toothless, short and wide.
But on it
the set of sensitive leathery outgrowths grows; that’s why the beak of starbill
resembles a little the snout of star-nosed mole (hence the name: Condylura
– the American star-nosed mole).
This animal is protected from predators by poisonous spurs (it is one more
feature inherited from duck-bill) colored bright orange. At males spurs are
longer, than at females. Fur of animal is black or dark brown with contrast
white marks on head and shoulders. The predator attacking starbill will remember
its colouring, having received a painful prick of poisonous spurs.
Starbill lives in damp ground of riverbanks and in marshlands. By this feature
it also resembles its prototype from the insectivores order. It eats various
ground and water invertebrates – earthworms, leeches, snails and larvae of
insects. The animal searches for them with the help of sensitive outgrowths
on edges of beak. These outgrowths are rich in chemo- and electroreceptors,
and permits to define presence of live creatures even in darkness. Under
water the starbill closes eyes, and is guided exclusively with the help of
touch
sence and receptors of beak.
Starbill is a solitary animal. Each individual digs complex system of tunnels
in riverbank. The nest for posterity rearing represents the long hole terminating
in the nesting chamber. This chamber is located above maximum water level,
and has an emergency exit.
In lower reaches of rivers starbills of different genders lodge in different
places. Males prefer to live in territories completely filling with water
during the inflow. Here the amount of forage is more, and for spending the
night animal
often uses any casual shelter. Females prefer to settle closer to riverbanks
where their long-term holes will not be filled with inflow.
At Flinders Island the separate population of this species differing in smaller
sizes (animals weigh approximately 20% smaller, than their relatives from
the “motherland”) lives. Conditions of life at this island are more severe,
than
at the continent – permanent sources of fresh water are not present there.
Therefore starbills of Flinders Island receive necessary moisture exclusively
from food. Also at these animals there is stronger poison secreting in smaller
amount.
This animal was discovered by Simon, the forum member.
Fat-tailed
marsupial dormouse (Notoglis crassipyga)
Order: Paucituberculata (Paucituberculata)
Family: Caenolestids (Caenolestidae)
Habitat: far south of South America, Tierra del Fuego, Andes – areas of seasonal
climate.

Picture by Alexander Smyslov
Marsupials represent the characteristic group of mammals of Southern hemisphere.
They are diverse in Meganesia and in South America, but in Pleistocene opossums
had penetrated for the second time into North America where they lived up
to Miocene, and in Neocene they widespreaded up to Greenland, having evolved
a
separate hardy species. One more northern advanced post of marsupials is
at Hawaii where these animals had appeared due to human activity and could
go
through the epoch of people domination on the Earth. But all the same at
southern continents marsupials are more diverse, rather than free or involuntary
immigrants
to the north. They lead various habits of life and occupy diverse habitats:
from humid tropical forests up to snow-covered tops of Andes and cold deciduous
forests of Tierra del Fuego.
Representatives of caenolestid family are tiny insectivorous forms from South
America. One species of this group lives even in wool of huge groundsloth
rodents. One more unusual representative of this family lives in areas of
temperate
and cold climate of South America – it is fat-tailed marsupial dormouse.
It is very large representative of family: an adult individual is guinea
pig-sized
one, but it looks larger because of long fluffy tail which exceeds length
of other parts of body. Head of this mammal is large and bears small auricles
and large bulging eyes.
Fat-tailed marsupial dormouse leads tree-climbing habit of life and lives
in forested areas. Because of rather short paws it is almost unable to jump
in
branches and prefers to climb, clinging by hooked claws. The tail of this
marsupial is partly prehensile – its tip can twist around of branches, and
the animal
can hang, fixing only on tail. The fur of animal has beautiful ash-grey color
with white underfur. On head and shoulders fur is darker.
The mouth of this marsupial is full of pointed teeth: fat-tailed marsupial
dormouse is omnivore and has obvious tend to carnivory. Its food includes
small mammals and birds, and amphibians and lizards in summer season. Also
it willingly
eats the carrion, also even frozen one. Similarly to the majority of animals
of such kind, it is a solitary species of primitive behaviour, displaying
aggression to relatives almost the year round, except for time of hibernation
and courtship
season.
Fur of fat-tailed marsupial dormouse is dense, short and velvety in summer
and growing much longer to winter. This animal is strongly eaten off in autumn;
in tail and buttocks fat is laid (hence the specific name “crassipyga”, meaning
“fat back”). With approach of steady cold weather this marsupial falls into
the true hibernation. For this purpose fat-tailed marsupial dormouse makes
the shelter – it covers a hollow with dry leaves and constructs grass-made
fuse to stop up an entrance of the hollow.
During the hibernation the body temperature of animal falls up to a minimum;
breath and palpitation are slowed down in great degree. In such condition
animal spends approximately two thirds of winter. In second half of winter
at the
female cubs are born. Shortly before the birth moment physiological processes
of female become more active. She clears pouch and licks wool. Female gives
birth up to ten underdeveloped cubs independently reaching her pouch. This
species has only six nipples, therefore the part of cubs perishes within
one hour after birth. When all nipples are occupied by cubs, the female falls
asleep
again, but dream is less deep. She produces milk which is periodically sprinkled
in mouths of cubs. They gradually grow and develop, and the female wakes
up after winter dream having already well advanced cubs. Within the summer
cubs
grow and pass to adult diet. At the beginning of an autumn they become independent
and reach sexual maturity at the third year of life. Life expectancy of this
species is up to 30 years.
Marsupial
sharp-toother (Carnomacropus raptor)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Predatory marsupials (Dasyuridae)
Habitat: savannas and light forests of Meganesia.
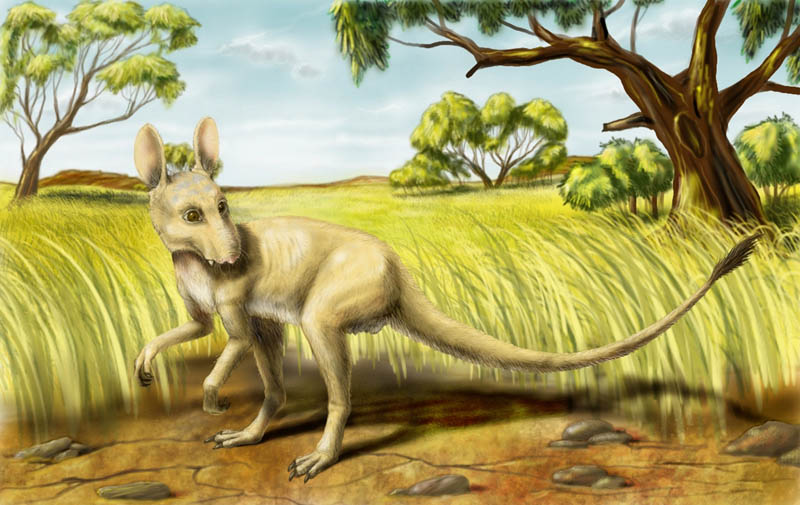
Picture by Alexander Smyslov
Looking at this carnivorous creature, especially during its
hunting, it is possible to think involuntarily, that dinosaurs have returned
to Earth. But actually it is not so, though this animal by habits and constitution
is very similar to reptiles missed for a long time from the face of Earth. The
marsupial sharp-toother is very original animal, and in fauna of Earth of human
epoch there were no analogues to it. It is animal about meter height at a shoulder
and weighting up to 50 kgs, the huge descendant of small marsupial jerboas (Antechinomys).
It has inherited and has developed characteristic carnivorous habits of ancestors,
and increasing the size this animal has had an opportunity to attack not only
small animals, but also to overpower even sheep- or antelope-sized animals.
Marsupial sharp-toothers became original "cheetahs" of the Meganesian
savanna attacking cursorial herbivores. They are solitaries but if they have
mastered concerted group hunting, it would not be more terrible animal in Meganesia
than this creature.
The constitution of the marsupial sharp-toother is adapted to fast chasing of
catch: back legs of animal are advanced as well as at kangaroo, and armed with
moderately long non-plungering claws (three claws on each foot). At short distances
the animal can accelerate momentum up to 80 kms per hour, and at long distance
it keeps speed about 40 kms per hour during tens minutes. Forepaws of animal
are rather big, there are advanced long fingers with the sharp claws on them,
permitting to grip and kill small catch. Tail of animal is long (the general
body length is of 2 meters, half of which tail amounts), well permits to turn
and to keep balance at sharp turns.
The head of the marsupial sharp-toother is armed with powerful jaws and well
advanced canines: tips of canines are visible from the closed mouth. Incisors
are very sharp, molars are powerful and wide: at lack of alive catch the animal
eats carrion, even dried up at the sun. Frequently marsupial sharp-toothers
eat up the rests of catch of other large predators. The chewing muscles are
well advanced: the animal kills large animals by bite in neck, or inflicts by
teeth extensive wounds to its prey and drives it up to exhaustion. Ears of animal
are wide, in them there is advansed the circuit of blood vessels: in heat weather
they serve for heat emission. Nasal cavity is high, nostrils form a kind of
small proboscis – it simultaneously increases olfactory surface and clears inhaled
air of dust: at internal edge of nostrils some numbers of the hair playing a
part of dust filter grow.
Eyes of animal are directed forward, sight is binocular.
Colouring of body of the marsupial sharp-toother is basically sandy-yellow
with faltering pale grey strips on shoulders and head, stomach is lighter,
on tail tip there is a hairy brush of long black hair.
Marsupial sharp-toothers are solitary animals meeting each other only for the
period of pairing. Also the grown up cubs some time keep near to mother, training
in hunting technique. Males at this species are larger and more aggressive than
females. Courtship relations of animals remind fight more: the male forces the
female to pairing by every possible way, bites its skin on back and forepaws.
At adult female’s ears are frequently torn up and bleed: these are traces of
similar “grooming” of males. Pairing repeats some times within two - three days,
then the male abandons the female and does not accept any participation in posterity
rearing. Pregnancy lasts not for long as at all marsupials: about one week;
newborn cubs are underdeveloped, more similar to embryons. Usually delivery
begins at night, when there is no danger of drying of the cub on the sun. The
female lies down in the dug out small hole so that the aperture of maternal
passages is little bit higher than an entrance to the marsupium (which opens
back), and gives rise up to 5 - 8 cubs. But because in pouch there are only
4 nipples, only those survive who reached to the pouch the first. The aperture
of pouch is surrounded with elastic ring muscle which pulls it during movement
of animal; therefore cubs do not drop out of pouch during sharp throws at catch
chasing. Cubs stay in pouch for about 5 months, thus from four only two, and
sometimes only one cum may survive. But its chances of the further survival
are rather great: the female furiously protects cubs, attacking even on the
largest herbivores if it will seem that cubs are threatened with danger. At
the age of six months the cub freely follows mother and can catch small vertebrates,
and one year old cub takes part in hunting on equal terms with mother. As a
rule, soon after this she can give birth to new litter again. Young animals
become sexual mature rather early: one-year-old females continue to grow, but
already can rear posterity. Males take part in breeding from the age of two
or three years.
Hunting small animals, for example, rabbits, the marsupial sharp-toother tries
to creep to them as closely as it is possible under covering of grass and bushes,
and then overtakes catch in short throw. Having overtaken with it, it inflicts
lateral impact by claws of forepaws, trying to knock down and to wound catch,
and then turns around and inflicts to chase a fatal bite. Hnting larger animals,
for example, camelopes (antelope-like camels), the marsupial sharp-toother
separates the planned chase from herd, and then drives it some hours along
the plain, not developing maximal speed. Usually during continuous chasing
the prey weakens so, that the predator succeeds to overtake it by one conclusive
throw and to seize its throat.
Usually marsupial sharp-toother is active since early morning till midday and
in the evening. The animal spends hot daytime in shadow of trees or in rich
bushes. If it is a lot of catch, the animal can any time stay in feeding area.
Then in bushes or river breakage it digs to itself wide hole where it has a
rest in the afternoon and sleeps at night. At lack of catch the animal becomes
the nomade and lives in temporary shelters.
Marsupial
crab-eater (Cancrivenator dexter)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Predatory marsupials (Dasyuridae)
Habitat: zone of mangrove forests separating Arafura Lake from ocean.
Mangrove forests at the northwest of Meganesia became the house for numerous
and various species of animals. There are many species of various mammals among
inhabitants of these forests. They can be vegetarians or predators, but one
feature unites all of them – they perfectly climb on trees. Some of them eat
only the food given by trees, and others feed in swamps, using trees only as
a place for life. The strange descendant of marsupial mice belongs to such species
– it is mobile and dexterous creature named marsupial crab-eater. The shape
the marsupial crab-eater resembles tarsiers (Tarsius) of Holocene epoch, but
it strongly differs from them by habit of life.
Marsupial crab-eater is medium-sized mammal. It weighs about 400 grams and it
seems rather large because of lengthened paws and long flexible tail. Rear legs
of this animal are much longer than front ones, and long tail serves as the
balance weight at jumps. Besides the bottom side of tail is naked, and tail
is also prehensile. The convergence of marsupial crab-eater and many climbing
mammals of New World also had such adaptations ise shown in it. Fingers and
toes of marsupial crab-eater are prehensile and mobile. The animal is able to
jump and to swarm up trees. Moving down from mangrove trees to the swamp, marsupial
crab-eater does not jump off on the ground – it may easily stick in silt and
die. It dexterously skips on roots of mangrove trees only on hind legs, and
climbs on branches in tree crone with the help of all four paws and tail. Marsupial
crab-eater can make long jumps from tree to tree, especially being pursued by
predators.
The wool of this animal has reddish-brown colouring with yellow spots forming
longitudinal lines on back. Throat, breast, stomach, forepaws and the bottom
part of hinder legs are white.
Marsupial crab-eater has large eyes directed forward and providing almost completely
binocular sight. Also it has wide rounded ears and long mobile proboscis-like
nose. Marsupial crab-eater is an exclusive predator with sharp incisors and
multituberculate molars. It eats small animals, searching them on roots of mangrove
trees during the outflow time. The basic prey of this animal includes crabs;
animal prefers to catch young crabs and crabs of small species. It catches crabs
by very original way: in order to seize the crab, the animal overhangs headfirst
and keeps by hinder legs and tail for roots of mangrove trees. By free forepaws
marsupial crab-eater seizes crab for the bases of claws, and thus does not give
it to protect itself, and then it puts to prey fast bite by powerful incisors.
This animal eats prey, sitting vertically on tree branch and holding it in forepaws.
Marsupial crab-eaters drink rain water, licking it from leaves, or cautiously
drinking up the top layer of fresh water from the surface of mangrove swamp
channels.
Marsupial crab-eater is a solitary animal meeting congeners only in courtship
season. These animals declare rights to territory, uttering loud shrill cries.
If two animals meet each other at border of territory, they display force to
each other, having seized branch by all paws and shaking it. After rain animals
mark borders of the territory with urine.
In the most remote place of the territory each animal builds on tree wum sphere-shaped
nest among live branches, bending and plaiting branches with fibres and strong
leaves of plants like pandanus (screw palm). Branches continue growing, and
the nest becomes strong and imperceptible. In this nest animal spends night,
and female leaves the grown up cubs.
The courtship season at marsupial crab-eater proceeds during all year round.
Males of this species search for females ready to pairing by smell. Courtship
at marsupial crab-eater is rough enough and hard: male interferes to the territory
of the female, pursues her and forces to stop for pairing, biting her tail and
hips. At this time he utters the special sounds similar to grumble.
After very short pregnancy at the female up to five underdeveloped cubs are
born. But in pouch opening forward like at kangaroo, there are only two nipples;
therefore only two cubs survive. They develop in pouch for a long time (till
about four months). The grown up cubs still some time sit on back of the female,
keeping for its wool and having clasped its tail. The female leaves grown up
and already heavy cubs in nest and comes back to them for feeding 4 – 5 times
a day. At this time cubs gradually start to study climbing and jumping on trees.
When they become independent enough and start to accompany with the female during
hunting, she has already time to couple and soon gives rise to new cubs.
Marsupial crab-eater becomes sexually mature at the age of one year. Life expectancy
of these animals makes 10 – 12 years.
Marsupial
potto (Neodasyrus auridae)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Dasyurids (Dasyuridae)
Habitat: mountain forests of Meganesia.

Picture by Alexey Tatarinov
Like other carnivorous marsupial, the quolls had experienced
hard times in Holocene epoch: their number decreased greatly due to anthropogenic
pressure and competition to introduced placental predators. However, unlike
their larger relatives, the thylacine and the Tasmanian Devil, quolls managed
to go through anthropogenic pressure, the co-existing with more evolutionally
advanced animals, climatic disasters and much more.
However, such “tests” have not gone unnoticed for the quolls: some of their
species became extinct, and the descendants of others sometimes began to develop
in quite different evolutional directions, acquiring completely different features.
Marsupial potto is one of such deviating species.
Like its placental analogue it is a small animal about 26-33 centimeters long,
having short tail. This tail is a legacy of the ancestral condition: marsupial
potto moves too slowly and does not leap, so it does not really need a tail.
The ancestors of marsupial potto had spotted coloration, but nocturnal lifestyle
made the fur of this animal monotonously dark brown with a golden tint. Also
it has shorter and broader snout, larger eyes and ears, and longer whiskers.
The teeth are small, but strong, able to bite through even a thick bone. Marsupial
potto is an active predator that feeds on insects and spiders, slugs and snails,
tree frogs and lizards, small mammals and birds – everything that it can catch
and kill with a strong bite. Dark fur of this animal makes it hard to see at
night when it sneaks up onto its prey – its short legs are poorly adapted for
longer leaps or a long chase, but it can sneak up to sleeping birds very cautiously.
Marsupial potto comes down to the ground very rarely – mainly to slake its thirst.
But more often this animal finds rosettes of epiphytic plants where water accumulates
and so it does not need to go down to the ground at all. Twice a year the females
begin to call out to males with barking sounds, indicating the beginning of
heat. If two or more males meet near the same female, there will be severe fighting,
and sometimes they bite each other. The winner mates with the female and leaves
it – the female of this species raises its litter alone. The pouch has only
two nipples, but the female gives birth to up to 5 offspring – but only those
ones survive, that reach the nipples first. At the age of three months the young
begin to leave the pouch and being 8 months old they are independent. By that
time the female is ready to breed again. If any female loses its cubs for some
reason, it comes into heat much faster.
The life expectancy of marsupial potto is 8-10 years; the sexual maturity comes
at two-and-a-half to three years.
This species of mammals is discovered by Bhut, the forum
member.
Translated by Bhut.
Filicide
embryotherium (Embryotherium infanticidum)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Predatory marsupials (Dasyuridae)
Habitat: Meganesia, woodlands and forests of various types.

Picture by Alexey Tatarinov
People-caused introducing of placental mammals to Australia
had strongly reflected in evolution of local marsupials. Many carnivorous species
of marsupials had been exterminated by people or had died out, when people had
exterminated large herbivorous marsupials. And small marsupials frequently did
not maintain a competition to placental predators. But one group of Australian
predatory marsupials had got great advantage from occurrence of placentals in
Australia – these ones were small predators, marsupial mice. When epizootias
had reduced the number of placental predators occupied Australia, marsupial
predators had not missed their chance and had returned in new quality – as clever,
furious and easily adapting species. Gradually they had taken a place in new
ecosystems of Australia and further in Meganesia.
The descendant of one Holocene species of marsupial mice, tiny (the total body
length is no more 10 cm, not including short tail) species of predatory marsupial
mammals, had developed the unique habit of life combining predating with original
kind of parasitism. It lives in places where many species of large marsupials
live, and spends a part of life, parasitizing in their brooding pouch. This
animal penetrates into pouches of large marsupial mammals and eats still tiny
newborn cub bearing by them. Then adult animal is attached instead of it to
the nipple and feeds on milk secreting by the female involuntarily becoming
the host of parasite. This animal is named filicide embryotherium.
At embryotherium many infantile features are kept in appearance. The skin of
this mammal is covered by thin soft wool and is completely naked in some places.
In appearance of animal the sharp sexual dimorphism is expressed. Wool of females
is thin and sparse; its color is yellowish-white. Males have richer greyish-yellow
wool with cross brownish strips.
Embryotherium, however, spends a significant part of life out of host animal
pouch. It is connected mainly with search of sexual partner, settling of young
animals and search of host animal by them. Embryotherium has well advanced sensitive
eyes and large ears – not living in host pouch animal leads nocturnal habit
of life and prefers to hide in various shelters in day time. Its sense of smell
is very keen – animal easily distinguishes by smell the female bearing the newborn
cub on the background of smells of other part of herd. Paws of embryotherium
are strong and supplied with well advanced claws. This animal is rather mobile:
embryotherium can swarm up trees, run and jump fast. Its mobility is connected
to necessity of search of host animal.
By the appearance embryotherium resembles a mouse. It has rather large head
with the short muzzle; muzzle, lateral parts of head and throat are almost hairless.
Teeth of animal are short and sharp; only incisors and canines develop. The
reason of it is a fact that till the significant part of life animal eats soft
food and has no necessity to chew it. Milk-teeth of embryotherium are advanced
better, than second ones – it is connected to necessity to lead an active habit
of life till the first weeks of independent life, up to the meeting of host
animal. Some more interesting adaptations to existence in pouch of host animal
are the features of excretory system. Embryotherium produces very dry dung and
concentrated urine. Leaving the nipple of host animal, embryotherium puts out
back from its pouch, sharply throws outside dung and urine, and then hides again.
Embryotherium is able to hunt and to eat small animals – worms, slugs and soft-bodied
insects like cockroaches and young crickets. Usually such diet is characteristic
for young individuals. Males spend in pouches of host animals rather small part
of life – they grow up, eating milk of the host animal, and at the approach
of sexual maturity leave the safe refuge. Adult males search for females by
smell and pair with them actively. Sperm keeps viability in sexual ducts of
the female till the long time, and it can bring up to three litters after one
pairing. It is an adaptation to secretive parasitic habit of life. After pairing
the female at which cubs develop, searches for the host animal. It settles in
its pouch, kills and eats its cub, sticks to nipple of the host animal, and
raises its own posterity in safety and food abundance. In litter of the embryotherium
it may be up to 6 – 7 cubs. The pouch of embryotherium looks like two thin and
elastic longitudinal plicas surrounding nipples from two sides. Cubs eat parent
milk for very long time – it is their only food till about 4 months, and during
the entire life they keep ability to digest milk proteins. Young embryotheriums
grown up sizable enough, leave host animal’s pouch and lead independent life
within several months. Females reach sexual maturity rather early – at the second
month of independent life. After pairing embryos in their bodies run to a diapause
and do not develop while the female will reach normal physical standard. Free-living
males seldom survive over two years, but separate individuals succeeded to find
the host animal after rough courtship season, quickly restore forces and can
live up to four-year-old age.
The idea about existence of this species was proposed by Simon and Mutant, forum members.
Marsupial
bloodsucker (Vampyrotherium nosferatu)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Dasyurids (Dasyuridae)
Habitat: Meganesia, forests, woodlands and bush.

Picture by Alexey Tatarinov
Marsupial mice appeared one of the most successful groups
of carnivorous marsupials of Neocene epoch. Large species of predatory marsupial
had not sustained a competition with placental carnivors introduced in human
epoch. But placentals could not displace marsupials in ecological niches of
small predators. The survival of small carnivorous marsupials became a basis
of their future success in Neocene. Among Neocene descendants of marsupial mice
the true giants, marsupial panthers,
had appeared. Other descendants of marsupial mice, embryotheres,
had got very strict feeding specialization and had developed the special form
of predatoriness which could appear only in the world where large marsupial
mammals live. And one more descendant of marsupial mice, marsupial bloodsucker,
represents the kind of haematophagous parasite – it is a very unusual phenomenon
among vertebrates.
Marsupial bloodsucker is tiny species of mammals: its length is no more than
5 cm (not including tail). Only marsupial
shrew – another dwarf species of marsupials – is smaller than this one.
In its constitution marsupial bloodsucker differs only a little from other species
of marsupial mice – it is mouse-looking mammal with short ears and thin naked
tail about 4 cm long. Wool colouring of this little mammal is ash-grey, but
head and shoulders are black, and a tip of muzzle is white. Such colouring is
disruptive, and it prevents a probable predator to distinguish this mammal in
conditions of bad lighting. In case of danger this little mammal hisses threateningly
and rushes on predator, putting to it deep wounds with teeth.
Marsupial bloodsucker lives in wood areas of Meganesia, avoiding of semideserts.
In savanna it keeps near to reservoirs where bushes and trees grow. In open
area this animal has two peaks of daily activity: in early morning and at the
beginning of night. It is too hot in savanna or in woodlands in the afternoon
and probable predators are more active. And at night, on the contrary, it is
also too cold. In tropical forest where day and night temperature difference
is less expressed, marsupial bloodsuckers are active in the afternoon. These
animals do not have determined territory and lead solitary way of life.
Marsupial bloodsucker eats almost exclusively blood of warm-blooded animals,
attacking them while they sleep. It finds potential prey by smell. This little
mammal cautiously climbs up in convenient place on their body, precisely determining
where the skin is thinner and blood vessels lay close to its surface. For this
purpose it is served with a site of naked skin on throat in which the set of
receptors is concentrated, and also with receptors on lips. Having found the
suitable place marsupial bloodsucker bites through skin of the donor animal
and licks off the streaming blood.
Teeth and jaws of marsupial bloodsucker are specialized to this type of feeding.
It has broadened and flattened (“bulldog-like”) muzzle with the mobile lips
permitting more effective blood sucking. Incisors in upper jaw of animal are
compressed from sides and work like blades – they make a vertical cut on skin
of prey. The saliva of animal contains anticoagulants (as at American bats Desmodus)
and the substances rendering anesthetizing effect.
Marsupials bloodsuckers find each other in forest and savanna with the help
of smell. At these animals there are specific glands from which the animal emits
a secret having strong characteristic smell as required. During the search of
donor animal gland ducts are closed by special sphincter muscles. Also before
search of donor animal marsupial bloodsucker carefully licks wool around of
gland ducts, hiding its own specific smell.
The courtship period at marsupial bloodsuckers lasts for all year. Male is tinier
than the female and approximately 20% lighter. Within one year the female can
bring up two litters of cubs. Female has 8 nipples surrounded by ring-like plica
of skin forming a brooding pouch, which entrance aperture it is shifted back
to the basis of tail. She gives rise simultaneously up to 20 underdeveloped
cubs; only ones managed to reach up to nipples can survive. It happens that
even four young animals from one litter rarely can survive up to independent
condition. They reach sexual maturity at the age of one year; life expectancy
of marsupial bloodsucker reaches six years.
These blood eating animals are carriers of filariae
– parasitic nematodes. It happens especially frequently in tropical forest.
Filariae pass one stage of development in mucous epithelium of mouth of animal
and infect donor animals, getting into the blood flowing from the wound made
by marsupial bloodsucker.
The idea of existence of this species was proposed by Simon, the forum member.
Marsupial
Suricate (Phascolomungo nanus)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Dasyurids (Dasyuridae)
Habitat: Tropical bushy savannahs of central Meganesia, arid deserts of Western
Meganesia, also around the semi-arid coasts of Eyre Gulf.
Dasyurid marsupials, predators of various kinds, are remarkable in their short
life cycle and usually anti-social behaviour. Individuals of some species may
not live longer than a year, but produce as many young as possible. One Neocene
species has taken this fecundity to its advantage and has become a social forager,
Marsupial Suricate.
This animal is small, no larger than a rat at about 20 centimeters body length,
head is wedge shaped with a pointed snout and sharp high-cusped teeth for consuming
invertebrates such as beetles and scorpions. Body is low to the ground with
slightly longer hind legs than forelegs, but forelegs bear large claws for digging.
Tail is short and bushy, with a black tip. This animal is colored a sandy tawny
brown, with brindling and striping of darker brown along the spine, paws are
white with dark soles, and face has distinctive black “glasses”. Unlike most
carnivorous marsupials, Marsupial Suricate is a social animal, living in large
groups of over 20 and as much as 35. Social hierarchy dominated by breeding
females, as males die soon after mating, breeding depends on constant fights
among males in order to court the females.
Individuals in the group forage together during the day, feeding on insects,
scorpions, centipedes, bird nestlings and small reptiles, even the young of
various venomous snakes. Individuals may work together to harass and kill more
formidable prey such as large scorpions or small snakes. As with its namesake,
individuals in the group remain alert for threats by predators, climbing bushes
or mounds in order to keep sight of potential danger. This is not an individual
role and any member of the group performs this action sporadically. Marsupial
Suricate is mainly diurnal but does most of its socializing in the late afternoon.
When faced with a predator, the group will mobilize together and seek refuge,
unless the combined might of the group may allow them to repel it, in the case
of smaller predators such as snakes or birds. The group shelters in a large
communal burrow that has several entrances and exits, individuals sleep in a
large huddle to maintain body heat, as metabolism is very high. Breeding is
constant throughout the year, and males are constantly fighting for the favour
of receptive females, unlike a true suricate there is no dominant pair. Several
females may bear young at the same time. Males die about a month after mating,
and are soon replaced by the new generation.
Females produce up to 15 joeys at a time, and have 13 teats with which to nurse
their young. Babies mature in about 5 weeks but do not breed until they are
a year old. Males generally do not live longer than a year, dying soon after
successfully mating, females may live as long as 5 years and produce many generations
of joeys. Vocalisation is a small chirring or a high pitched yarring when alarmed.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Budjagan
(Phascolofelis ferox)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Predatory Marsupials (Dasyuridae)
Habitat: Forests of various sorts, Eastern and Northeastern Meganesia.
A typical trait of evolution is convergence among unrelated forms, and one of
the classic examples of this is the Australian marsupials. Here we see analogues
of leopards, foxes, weasels, ungulates, moles, hedgehogs etc. During the Neocene
this has become even more pronounced in places, with Phalangers converging on
primates, and Dasyures converging on many kinds of placental carnivore. Phascogales
of the Holocene gave rise to some of the most cat-like of Neocene Dasyures,
including the Marsupial Panther,
but also the Budjagan, which takes its name from an Aboriginal Australian word
meaning feral cat.
Outwardly this animal does indeed resemble a domestic cat, being of a similar
size and weight to a typical housecat. Coloration is a greyish fawn color, with
dark bands on the tail and hips, turning into spots along the sides, face has
a contrasting “mask” of dark and light hairs, feet are dark furred with pink
soles, nose is small and pink. The feet are semi-digitigrade, and the limbs
are muscular and fairly long, both fore and hind feet have large claws for both
climbing trees and capturing prey. Tail is long and bushy, with a white tip.
The snout is a good deal shorter than that of a typical dasyure like a quoll,
with a broad face and large forward facing eyes, ears are large and pointed,
with a coat of fine hair. Canine teeth are long and slender for tearing the
throat out of small vertebrate prey, and molars have the carnassial shape for
cutting meat, with the rearmost molar being adapted to crush small bones such
as the skull of prey. Budjagan has a slightly longer midsection than a placental
cat, and in this way slightly resembles a large marten, apart from the short
blocky head. Vocalisations range from a hissing growl to a high-pitched yarring
squeal. Budjagan is most active at night and does most of its hunting then,
but it may also be active occasionally during the day.
This animal is found mainly in forested areas, and is a great climber, commonly
hunting birds and possums among the branches. Prey also includes small reptiles
and rodents, or even large insects such as moths. It is a solitary animal, only
meeting up in order to breed, during the dry season. Females produce a strong
musky scent that attracts males from a distance, and more than one male may
fight over the right to mate with a single female. Mating is swift and noisy
with growling and yarring, but the male does not die after mating, and may produce
more than one generation. The pouch is a simple fold on the stomach, and the
young will stick out of it when they get larger. After a gestation of 18 days,
a litter of up to 10 joeys are born, of which only six may survive the first
few weeks. They remain in the pouch for 8 weeks, and in the ninth week will
start to cling to their mother’s back to travel, as well as venturing short
distances. Joeys mature within a year, and both males and females have a lifespan
of up to 15 years. Predators include large birds of prey and any larger kind
of predatory reptile; they are usually pugnacious enough to repel other marsupial
predators.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Marsupial
digger (Pseudotalpa geobia)
Order: Dasyuromorphia (Dasyuromorphia)
Family: False marsupial moles (Pseudonotoryctidae)
Habitat: bush savannas of Meganesia.

Picture by Alexander Smyslov
Marsupials in Australia have shown to the full an opportunity
of evolution of separate limited group of animals. Among them in different time
ecological analogues of placentary mammals, frequently belonging to different
orders, had appeared. Even the marsupial mole lived in Holocene at the territory
of Australian deserts – the kind of blind digging marsupials.
In Neocene among marsupials the second time the species having underground habit
of life had appeared. It can seem strangely enough, it had evolved due to activity
of human for a long time disappeared on Earth. In due time people had introduced
rabbits to Australia, and they have successfully gone through cataclysms on
border of Holocene and Neocene due to the abilities to adaptation and high fertility.
In rabbit holes spreading under ground to many tens of meters and forming ramified
systems various species of animals settled. Among them any species of marsupial
mice (Antechinus), subsequently adapted to completely underground way of life
was. This small predator had to found in rabbit holes practically everything
necessary for its life: relative safety and food – numerous insects hiding in
holes from day time heat. In holes it was always possible to find larger catch
– dead rabbit cubs. Evolution had picked up and had improved this idea and as
a result savannas of Neocenic Meganesia were occupied by one more species of
digging marsupials – by marsupial digger.
This animal is medium-sized one: length of body is about 20 cm, and height
at a shoulder is only about 6 – 8 cm. Body of this small mammal is cylinder-shaped
and muscled; it is covered with short velvety brown-colored fur. Paws of marsupial
digger are short, on strong forepaws there are three long strong claws of yellowish
color (on II, III and IV fingers) with which help the animal carves its burrows.
Hinder legs are very short, with narrow feet and four fingers (there is no
thumb at the foot). Tail of this animal is short and very thick; it serves
for accumulation of fat stock. The layer of fat also is laid on waist of animal.
At the marsupial digger there are short wide head and very strong skull. Bones
of skull are thick and also have grown together with each other to solid vault.
Eyes and auricles are absolutely not present at it, and nose is covered with
corneous scute, as at marsupial mole (Notoryctes). Nasal cavity of animal is
strongly expanded, and its internal surface is covered with set of plicas –
animal has keen sense of smell. Looking at teeth of marsupial digger everybody
will understand that it is furious predator in its weight category: all teeth
in its mouth are peaked and cutting; and canines are rather long, even jutting
out from mouth.
The marsupial digger lives exclusively under ground and never leaves on surface.
If the marsupial mole of Holocene epoch preferred to live in drift sands, the
marsupial digger builds constant system of constant holes in dense ground,
in which it waits for catch – every possible small animals. These small mammals
are very strong, and are able to kill animal larger than itself. Metabolism
at marsupial digger is rather intensive: for day it eats quantity of food only
twice less, than it weighs itself. Having found a plenty of food, the animal
eats it all and quickly accumulates fat in back part of body and tail. The
full digger becomes inactive to any time. It creeps to the hole, closes an
entrance by cover of friable ground and vegetative rests, and sleeps. After
the food will be digested, it wakes up and begins to hunt again.
The hole of marsupial digger includes main chamber located at the depth about
2 meters under ground, additional chamber which incorporates with main one
by unique vertical corridor, and system of radial burrows spreading from the
additional chamber to sides at the depth about meter. On the ground surface
it is possible to heaps of ground pulled outside by this animal. The marsupial
digger throws out ground through an inclined course, instead of vertical shaft,
as moles do.

Burrow systems of different animals of this species never incorporate directly
with each other: marsupial diggers are soliyary animals. But it yet does
not mean, that this animal avoids other “colleagues” in underground life:
whenever
possible this animal builds joint systems of holes with rabbits. Some burrow
systems of marsupial diggers frequently adjoin from edges to one settlement
of rabbits. Obviously, rabbits are not in delight from such neighbourhood:
the marsupial digger makes it having especially gastronomic interests – it
can attack rabbit cubs, and at times attempts even healthy adult rabbit if
it will manage to cut off way to escape to prey. Besides having attached
the dwelling to hole of rabbits, the animal saves forces at burrowing of
own holes,
chasing every possible inhabitant of rabbit holes. More often rodents – mice
and rats appear on its “lunch table”. Sometimes even other small predators
- marsupial hermins (Mustelogale
dolichocranus) become its prey, getting for hunting in holes of rabbits.
Obviously, at an attack against such rather dangerous animals the horn scute
on muzzle
rescues the marsupial digger against wounds.
During breeding season males leave holes and wander in underground burrows
searching for females. They are extremely aggressive relatively to each other
and if two males will meet in hole, one of them can kill and to have eaten
another one.
At these animals constant pairs have not formed, and the destiny of posterity
is completely indifferent to the male. At any opportunity male even can eat
young animals if will to find a hole with cubs. The female gives rise only
to three – four underdeveloped cubs. They creep in pouch (opening back) and
grow in it about one month. After that the female leaves them in hole, and
regularly comes to feed. For one year the female has about three packs.
Three-month aged cubs already can freely move in underground holes. They
leave territory of the female and live independently. Within the first months
of
life repugnatorial glands at them are not advanced, therefore their presence
can remain unnoticed by adult animals of this species. But up to the period
of puberty (at the age of about seven months) at young animals the smell
appears, and aggression sharply grows. The marsupial digger lives seldomly
more than
four years.
Marsupial
spiketail (Spinurotherium leucospinus)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Spike-tailed marsupial rats (Spinurotheriidae)
Habitat: bushy savannas of Meganesia.

Picture by Alexander Smyslov
In Neocene climatic conditions in Australia/Meganesia became
more favorable for life. Water had come to central areas of this island continent,
having transformed deserts to savanna with circuit of shallow-water rivers and
lakes. In such favorable conditions many new species of animals have appeared
on continent.
The fauna of Meganesia of Neocene epoch represents fantastical combination of
descendants of native marsupial mammals and introduced by people placentary
ones. Epizooties of rabies and plague of carnivores have reduced chances of
survival of introduced placentary predators to zero, but the competition to
them had perfected features of adaptation at local marsupial Carnivores, and
had permited to few survived ones to take place at top of food pyramid. Among
large-sized herbivores it is possible to meet not only graceful and harmonous
descendants of camels found due to the people new native land in Meganesia,
but massive marsupial animals too. And among small and medium-sized animals
marsupials successfully compete to rabbits and rodents introduced to this continent.
Among dense grass of Meganesian savanna it is possible to see strange object:
something similar to white flower, sticking up on hairy stalk. But, when this
object starts to move among grass stalks, it becomes clearly seening, that it
is not a plant but any animal. From time to time the owner of this “flower”
rears on hinder legs, and it can be seen more detailly. It is a cat-sized animal
covered with short wool. At it there are long flexible nose surrounded with
thin wiskers, round eyes and ears similar to rabbit’s ones. Well advanced claws
are appreciable on strong paws of this animal. And the thing looking like certain
fantastic “flower” actually is very long tail. At the tip of tail hairs are
modified to white-colored needles with black bases forming hairy brush. For
such appearance of this structure the animal had received the name “spiketail”.
It has habit to lift from time to time tail vertically upwards: this way spiketail
communicates with neighbours at long distance. A certain species of Australian
marsupial mice had become an ancestor of the spiketail.
Colouring of animal is rather motley: the body is covered with brown wool with
dim black speckles, the mouth is led round by thin strip of white wool. On the
back of marsupial spiketail there are some alternating black and white cross
strips. All tail is black, and white needles are brightly appreciable on its
background. Being attacked by predator, spiketail warns it that it is armed,
curving back like a cat, lifting up tail, and shrilly squealing. If it does
not stop predator, the spiketail protects itself against it by impacts of tail.
In the basis of each spike there is a muscle attached by one end to backbone.
With the help of such muscles the spiketail can press spikes to each other,
forming “beater” or to spread them widely in sides, forming true weapon which
impact can be rather painful. Defending itself against predator, marsupial spiketail
turns around on hinder legs, keeping to aggressor by side and swinging by tail
extensively.
In mouth of spiketail it is possible to see both sharp incisors and canines,
and wide knobby molars. The marsupial spiketail has a little deviated from diet
of ancestors: it is omnivorous (marsupial mice are predators), and the most
part of its diet consists of plants. The animal is able to dig out roots from
ground, dexterously swarms up bushes and low trees in searches of fruits, chews
unripe seeds of grasses. But it also likes to diversify the diet with food of
animal origin. On trees and in bush marsupial spiketail searches for small reptiles,
large insects and bird’s nests. In the ground except for roots spiketail willingly
finds and swallows worm or larva of any insect. By smell it finds the rests
of predator meal, and with pleasure gnaws bones and cartilages.
These animals are solitaries, but they support visual contact to neighbours,
from time to time rising on hinder legs and looking around. They usually avoid
close meetings, preferring to leave an urine mark on objects allocated among
savanna – trunks of trees, termitaries and stones. Unique exception is a time
of breeding season. At this time marsupial spiketails gather to small groups
– approximately fifty-fifty females and males. Males compete with each other
for females, establishing hierarchy in short duels. During such tournaments
they beat each other by tail, but in this case needles are densely compressed
and do not cause wounds to the opponent. After pairing the male loses interest
to the female and starts to look after for next one. Thus it jumps after chosen
female on hinder legs, uttering keen chirp and snort. Each female couples with
several males, therefore paternity at these animals is difficult to establish.
Sometimes in one pack cubs at once from two males can appear.
After short pregnancy the female gives rise to about ten tiny underdeveloped
cubs. But the chance to survive is not at all of them: at the female there are
only six nipples. The pouch at this animal is presented by two longitudinal
plicas of skin, surrounding nipples from two sides. This skin is elastic, therefore
cubs are not threatened with danger to drop out of it. They develop in pouch
about three months, and to this time from six cubs only three or four ones survive.
Grown up cubs move on back of mother, and it drags them on herself one month
more. At this time on tails of young animals needles start to grow. Gradually
cubs pass from milk to adult diet, and start to feed near mother independently,
at the same time training at it to ways of food getting. Half-year old cub already
becomes completely independent. The young female can bear posterity first time
already at one-year-old age.
Stinktail
(Malodorocauda catta)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Spike-tailed marsupial rats (Spinurotheriidae)
Habitat: Bushy savannas and light forests of central Meganesia, forested areas
of south eastern Meganesia.
During the Holocene, the role of small and mid-sized omnivores in Australia
belonged to two kinds of marsupial, Bandicoots and Potoroos. This situation
has changed in the Neocene, previous predation pressures from feral carnivores
during the age of man eradicated the bandicoots, and potoroos diversified into
different forms in the early Neocene, proboscis kangaroos, and also various
small carnivores, though some forms remain close to the ancestral shape and
lifestyle. Additional to this came the event of some kinds of marsupial rat
becoming more equable to consuming plant matter, thus producing omnivores. One
example is the family of spike-tailed marsupial rats.
This family is exemplified by forms which bear a brush of sharp quills at the
end of the tail, but more primitive members of this family also exist, such
as the Stinktail. These forms have only bushy hairy tails, but still use them
for defence, chemical defense. The Stinktail is about the size of small housecat
but differs by large noticeable claws on the forepaws, and pointed snout, the
tail is long and bushy, colored with alternating dark and white bands. This
marsupial produces a powerful chemical from its anal glands, which smells very
noxious and strong; it habitually grooms and rubs this secretion into the fur
of its hindquarters and tail. When faced with a predator, the tail is brought
upward and forward, batted and fanned at the opponent, subjecting it to the
unbearable smell. If the predator is not repelled by this, the Stinktail will
squirt glandular secretions directly at it, much like a skunk. The body of the
animal is a mid-toned greyish brown, with darker brindle beginning on the hips
and becoming full dark bands on the tail. Paws are lighter, bearing white fur
and pink soles, and the face has a dark “mask”.
This creature, as with the Marsupial Spiketail, feeds on both plant and animal
matter, but Stinktail is predisposed to feed on softer food, and does not consume
seeds or stems. Vegetable food includes roots, fungus, fallen fruit, and buds
of new growth, various insects, larva and small vertebrates are eagerly eaten,
as well as the eggs of birds or lizards. Stinktail has large foreclaws, resembling
bandicoot a little, and will search for food by digging, as well as climbing
low trees or searching in the undergrowth. Vocalisations consist of a throaty
hiss when annoyed or a high pitched growl when establishing dominance. This
animal is crepuscular, being most active during dawn and early morning, and
again during late afternoon and dusk.
These animals live in loose family groups of up to 6, and will forage over a
large area during the day. The banded tail is usually carried aloft to maintain
contact in the group, and individuals may rear on hind legs to keep sight of
others. At the sign of a predator, more than one individual may undertake the
threatening display of chemically armed tails, driving it away. In the breeding
season, which falls to the dry season, family groups merge with each other in
some areas, and males will display and posture in an attempt to court females.
Males produce a special kind of sweeter scent during the mating season, and
compete by vigorously shaking their tails and stamping their feet. Females will
mate with numerous suitable males, and give birth to a litter of 12 joeys, only
6 of which usually survive, as the folded pouch only contains six teats. After
about 3 months, the young move from the pouch to cling to the back and tail
of the mother instead, by six months they are fully weaned and become independent.
Young females become fertile at one year. Lifespan is short and generally does
not surpass 5 years, and with steady breeding, numbers within the group stay
constant.
Relative in the rainforests of northern Meganesia is the Spicetail
(Malodorocauda capsaicis). This animal is slightly smaller, only as
heavy as a rabbit, and is a dark reddish color, tail is banded chocolate brown
and white. This animal differs in its chemical defence; its anal secretions
contain a poison that is extremely bad tasting to predators, overly spicy and
painful. The animal rubs this secretion all over itself including its tail,
and will lick up the secretion in order to coat its teeth. When faced by a predator,
it will fan and direct the smell with its tail, but if seized in the opponent’s
mouth it is quickly dropped because of the unbearable taste, Spicetail may also
administer bites which allow the poison to penetrate into the predator’s blood
and cause extreme pain. The diet of this marsupial is mostly similar to its
central relative, though its lifespan is slightly shorter. Also, Spicetail forms
smaller family groups that range widely apart from each other during the day.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Erucotherium,
caterpillar beast (Erucotherium quasiapodus)
Order: Dasyuromorphia (Dasyuromorphia)
Family: Erucotheres (Erucotheriidae)
Habitat: tropical forests of Meganesia, leaf litter.

Picture by Alexander Smyslov
In Neocene restoration of tropical forests stimulated intensive
evolution of forest inhabitants and occurrence among them of specialized forms
occupying
strict ecological niche. One species of mammals living in forests of Meganesia
reached an extreme degree of specialization to its way of life. This creature
is the distant relative of marsupial mice passed to burrowing habit of life.
Its specialization had gone very far; the result of it is a significant reduction
of some parts of body. The name of animal means “almost legless caterpillar
beast”. It lives in friable leaf litter of Meganesian forests, not being
dug deeply in firm ground. In the body shape it has convergent similarity to wedgehead
(Herpethotalpa caeca), digging lizard of skink family living in the same
places. The body of erucotherium is about 40 cm long at the maximal thickness
up to
7 cm. It is cylindrical, truncated on the back end and wedge-shaped from
head end. Wool is short, dense and velvety, of grey, brown or reddish color.
On
head there is a flat corneous plate of yellowish color is advanced; it serves
for protection of skin at digging.
Forepaws of erucotherium are strongly reduced. Bones are very short; ulna
is reduced and radius is thickened. Hand bones fuse and lose mobility. Only
humeral
and wrist joints keep partial mobility. From outside from forward limbs two
large claws (3-rd and 4-th fingers) located on small lumps protrude – it
is an only seen part of forepaws. Claws serve for fastening in the ground
and
for pushes. Hinder legs are less reduced in comparison to front ones and
keep the limited mobility. Two-toed foot with thin and mobile 2-nd and 3-rd
toes
and normally advanced ankle joint is visible outside. The role of these toes
in life of erucotherium is great: they serve for clearing of pouch at females
and for keeping of the female during the pairing at males. Strong flattened
tail with the wide basis helps to push forward in the ground. The tip of
tail is dulled and gristly – it serves as a support at burrowing in the ground.
Tail vertebrae have wide and powerful lateral outgrowths.
The chest of animal is motionless, ribs strongly fuse to backbone. The animal
breathes exclusively with the help of diaphragm movements. The lumbar part
of backbone is long and very flexible: animal can easily bite its own heel.
Despite of deep specialization to underground habit of life erucotherium
is not helpless if it should leave a usual inhabitancy. In case of need it
is
able to swim perfectly, waving the whole body as if a dolphin. With the help
of sensitive bristles on muzzle it perceives the waves reflected from subjects,
and can be guided in water and bypass large obstacles at least partly.
Eyes of erucotherium are strongly reduced and hidden under skin. The skull
of animal is strong and thicken; it serves for tunnel burrowing. It is flattened
and has advanced cross crests for an attachment of the muscles raising head
up. External ears are lack; the acoustic duct represents a small aperture.
This animal perceives good the sounds getting in ear from the ground through
bones of lower jaw. The similar mechanism of sound perception is characteristic
for cetaceans, but in this case it was developed independently. This animal
hears badly the sounds spreading in air. Erucotheres communicate to each
other with the help of the odorous labels left in thickness of leaf layer.
This mammal eats any animals living in leaf litter. In order to get food
erucotherium developed long tongue which can be extended forward for length
of head. Edges
of tongue are covered with corneous combs, and its basis had shifted far
back and is attached to sternum. Tongue serves for digging and capture of
worms,
insects and their larvae, and also helps to clean wool. Due to flexible backbone
animal can lick wool on all parts of body.
Erucotherium is reliably protected from enemies. In the basis of tail it
has a pair of glands secreting badly smelling oily liquid. Defending against
enemies,
this animal can bite strongly. It has pointed teeth adapted to feeding on
insects. Besides the saliva of erucotherium is toxic for small animals and
in large
predators it causes long weakness and pain if gets in open wounds.
Seasonal prevalence in breeding of this species is not present. Male finds
the female ready to breeding by smell, pairs with her keeping her by teeth
and by hinder legs, and leaves female right after pairings. Pregnancy is
short as at all marsupials – not longer than one week. The brooding pouch
having
two nipples is opened back and before birth of cubs the female clears it
by toes. The female raises only two cubs two times per one year. During the
cub
birth she turns on back and cubs have an opportunity to creep into the pouch.
Up to five cubs are born simultaneously, but only ones managed to reach nipples
can survive. They develop in pouch till two months and then the female cares
of them till about one month, feeding them up on caught insects. Young animals
reach sexual maturity at the age of 8 – 9 months. Life expectancy does not
exceed 8 years.
Marsupial
sloth (Bradyphalanger scandens)
Order: Diprotodontia (Diprotodontia)
Family: Phalangers, or Climbing marsupials (Phalangeridae)
Habitat: tropical woods of Northern Meganesia.

Picture by Alexander Smyslov
In Neocene Australia moving to the north has collided against New Guinea
Island. As a result in northern area of continent young mountains have
started to increase.
This area is in the zone of equatorial climate, and mountain covered by rainforest
giving shelters and food to various live creatures. As well as in other parts
of the world, life in Meganesian rainforests is concentrated in
forest canopy at height 20 - 30 meters above the ground. Many original live
creatures, descendants of Holocene inhabitants of this region, live here.
One of them is the original herbivorous representative of marsupials –
marsupial
sloth, the descendant of cuscuses (Phalanger).
Similarly to South-American “colleague” of Holocene epoch, marsupial sloth
is the inactive herbivorous animal slowly moving in tree crones, suspending
on them from below with the help of hooked claws. This animal weighs about
15 kgs; length of its body is about half meter. However, as against the “true”
sloths of South America, the Australian marsupial sloth has also long prehensile
tail, by the size exceeding body length. The bottom side of this tail is
covered with naked skin with set of cross combs (similar to papillar patterns
on hands
of primates) – it raises durability of capture of support. Working as the
“fifth paw”, tail helps this animal to keep on branches. But basic organs
of movement
are unusual paws of the marsupial sloth.
Forepaws of this animal are similar to monkey’s ones - the thumb opposes
to fingers. But on hinder legs from one side the thumb and joined together
second
and third toes, and from other side fourth and fifth toes are opposed against
each other. Back legs of the marsupial sloth are powerful “pincers” by which
it grips branches when it takes to itself food by forepaws. Backbone and
neck of the marsupial sloth are very flexible: hanging a back downwards,
the animal
easily can bend a half-revolution, touching by forepaws the bottom branch
of tree.
Head of the marsupial sloth is rather flat, short and wide: at it chewing
muscles are strongly advanced. Molars of animal are wide and knobby – leaves
of trees
and firm seeds which this mammal cracks with loud crunch make significant
part of its diet. The marsupial sloth willingly adds food of animal origin
to its
diet: eggs and nestlings of birds, and also large insects and snails.
The marsupial sloth is colored according to the habit of life: because more
than half of time it spends hanging under branches, its stomach is darker,
than back is. On the back of animal fur is gray-brown with numerous narrow
faltering cross strips – it is an excellent imitation of tree bark. On stomach
wool is monocoloured, dark-brown. Hands and feet of animal are naked, covered
with rosy-gray skin. Around of big orange-brown eyes there are circles of
yellowish wool, tip of muzzle is of the same color. Male and female do not
differ in
colouring; the male is simply a little larger.
At this animal color binocular sight is advanced, there is sharp sense of
smell, and the hearing is a little bit dulled. Nasal cavity of the marsupial
sloth
is short and wide, and ears are almost latent in fur. By the “intellectual
development” this animal continues “traditions” of marsupials: brain cavity
at it is small. But at its habit of life the special ingenuity is practically
not needed.
Marsupials sloths are sluggish solitary animals. They are practically omnivorous
and do not depend on any limited fodder resource, therefore they tolerantly
concern to presence of each other and have no protected territories. Only
the female bearing posterity in pouch becomes a little more aggressive to
neighbours.
Pairing and birth of cubs at marsupial sloths occurs practically in any season.
The female ready to pairing, during movement marks branches and leaves by
the smell: repugnatorial glands at it are in axillary area, and it simply
drags
a branch between paw and side, having griped it. The male finds the female
using these marks; then he couples to her without any rituals, and marks
wool of the female by his smell. Such feature of behaviour is characteristic
for
some Holocene species of cuscuses.
Pregnancy lasts not a long time, and after 8 - 9 days tiny underdeveloped
cubs are born. Usually up to 10 cubs are born, but at the female there are
only
six nipples, therefore some newborn marsupials sloths at once are doomed
for death. Cubs independently get into pouch opening back, and start to suck
milk
and to grow. Approximately at the age of two months they can leave mother’s
pouch and remain on its body, clinging for its paws. Usually up to this moment
only three or four cubs from all survive, and only two or three cubs from
litter can become independent completely. At the age of four months youngsters
already
depend on mother a little, and to the age of five months they become completely
independent. At this time the female already carries a new pack in pouch.
For one year it can give birth up to three packs of cubs.
Young marsupial sloths become sexual mature at one-year-old age though active
growth proceeds at them till two years. Life expectancy of these animals
is no more than 8 – 9 years.
Drummer cuscus (Onychowirlda tympanista)
Order: Diprotodontia (Diprotodontia)
Family: Phalangers, or Climbing marsupials (Phalangeridae)
Habitat: plain rainforests and mountain fog woods in Northern Meganesia.
Neocene is the epoch of the occurrence of extensive large forests in Northern
Meganesia, and the development of rich fauna of tree-climbing animals. In forest
canopy, at height of several tens meters above the ground, various mammals
live: rodents, chiropters and marsupials. For similar ecosystems the combination
of two factors is typical: on the one hand, the abundance of kinds of food,
but on the other hand, rather rigid competition. Therefore in such ecosystems
very much specialized species frequently evolve.
In the evening and at night in forest canopy frequently it is possible to hear
rolling knock on tree, similar a little bit to drumming which a beak of woodpecker
beats out. Woodpeckers do not live in Meganesia, therefore instead of them
absolutely other animals hunt wood-boring insects. One of these creatures is
rather large (cat-sized) species of cuscuses. This creature is vaguely similar
to lemur aye-aye (Daubentonia), dwelt on Madagascar in Holocene epoch. But
instead of thin sensitive middle fingers thumbs armed with thick massive claw
are strongly advanced on forepaws of this cuscus. On other four fingers there
are very sensitive subungiuses covered with thin skin. Searching for insects,
this cuscus “drums” on bark and wood by thumb claws and catches vibrations
of wood by subungiuses of other fingers. Because of this feature the animal
has received the name drummer cuscus. Most likely, it is the descendant of
fox brush-tailed cuscus (Trichosurus vulpecula), one of few cuscuses had received
advantage from the neighbourhood with people in Holocene epoch.
This marsupial is fine tree-climber, spending all life in crones of trees and
never going down to the ground. All paws of this animal are prehensile, and
the long fluffy tail serves for maintenance of balance. The short velvety fur
of the drummer cuscus is colored grayish-beige color with marble pattern of
cross strips. It provides fine masking for animal swarming up on bark. Drummer
cuscus is too massive to jump from branch to branch similarly to marsupial
lemurs, cats or primates. But its paws differ in great strength of seize –
this animal can hang freely on one paw, and during the combat with small predator
cuscus can simply strangle it. Even dead animals of this species seize branches
so strong, that they fall on the ground already semi-decomposed.
At this marsupial there is the head with short wide muzzle and large sensitive
ears. Eyes of drummer cuscus are large and yellow, with strong shine characteristic
for nocturnal animals.
Distinctive feature of this animal is strongly advanced incisors similar to
incisors of rodents, and very strong chewing muscles. This is the adaptation
for feeding on insects hidden in thickness of wood. “Percussing” wood by claws,
animal listens to the tonality of sound, and feels by fingers changes in character
of vibration of wood. By these attributes drummer cuscus defines presence and
direction of tonnels made by larvae of insects in wood. Having found out a
tunnel pierced by beetle larva or other insect, cuscus opens it by several
strong bites.
Other feature of this mammal is very long tongue which can extend from a mouth
to ten centimeters. The basis of tongue is strongly shifted back - almost to
breast bone. On the tip of tongue the cross comb of corneous hooks, which can
be shifted relatively to each other a little, grows. Having opened dwelling
of insect, drummer cuscus pushes tongue in hole, and hooks larva on by these
hooks.
This animal goes to feeding in the evening and at night, and in dawn time it
hides in deep tree-trunk hollows or covered bird nests. Drummer cuscus dozes
hottest time of day, having covered sensitive eyes by ears and paws. At this
time it is enough vulnerable. Day time sleep of animal is sensitive, and, being
disturbed, drummer cuscus makes sharp movements by claws of forepaws, trying
to strike the enemy by them.
Drummer cuscuses are strictly solitary animals; only the female and her posterity
can keep together till some time. At night each animal from time to time reminds
to neighbours of its existence, uttering shrill high-frequency squeak. Male
searches for female ready to pairing, using such sound and special odorous
labels.
The male of drummer cuscus is approximately a quarter lighter, than the female
is; besides at him there is darker pattern on skin. Seasonal prevalence in
breeding at this animal is not expressed, and at any time of year in wood there
are females having posterity at various stages of development. Per one year
the female of this species gives rise two times to up to ten cubs, which leave
the brooding pouch at the 3-rd month of life. To this moment from pack only
two or three cubs survives. Till about one month they move in wood on back
of mother, and then start to lead independent life. Young animals reach the
sexual maturity at the second year of life. General life expectancy of animals
of this species does not exceed 10 years.
The idea about existence of this animal was stated by Simon, the forum member.
Woolly
False Cuscus (Enanticuscus novaezealandiae)
Order: Two-front Teeth (Diprotodontia)
Family: Possums and Cuscus (Phalangeridae)
Habitat: Cool mountain forests and temperate lowland forests of the South Island
of New Zealand, forest canopy.
Though the introduction of the Brushtail Possum (Trichosurus vulpecula) into
New Zealand, served to catalyse an adaptive radiation of unusual forms, including
predators and giant
ones, some forms came to more closely resemble their Meganesian cousins.
The Woolly False Cuscus is a large arboreal possum found in the temperate woods
and cool mountain forests of the South Island. Body length not including tail
may be up to 60 centimetres, and the tail is the same length again, weight does
not exceed 7 kilograms. Build and particulars of the body resemble both its
ancestor, and the Cuscus of New Guineas Holocene, a bulky climber with powerful
limbs, gripping hands and feet, and a large rounded head with big circular eyes.
Its coat is dense and woolly, even more so than its ancestor, and very soft
to the pouch, tail is muscular and prehensile, and covered in a coat of mid-length
dense fur, tail underside is naked. Fur is coloured a pale grey, which darkens
to almost black on the rump, shoulders and back are covered in irregular black
and light-grey patches. The skin of the face is covered in a fine coat of dark
fur, appearing like a mask around the eyes and nose. Eyelids, nose, soles of
the feet and underside of tail are naked and dark pink.
Diet consists of soft leaves, fungus, fruit and seeds, large insects and caterpillars
are also eaten, this animal will eagerly consume bird eggs and nestlings, in
lowland woods the nuts of conifer cones are often eaten. This animal is an adept
climber, spending most of its time in the branches of trees, but it may also
occasionally seek food on the ground, such as fallen fruit or mushrooms. False
Cuscus is mostly nocturnal, foraging and feeding at night, by day it sleeps
I large tree holes or in the fork of a large tree limb. It is generally shy,
and will flee at the first sign of an intruder or predator, if cornered it can
bite or scratch fiercely. Generally solitary, males mark their territory with
musky secretions, and by chewing the ends of branches, confrontations usually
occur when males meet, and will involve grunting and snarling loudly.
Mating occurs year round, but is most frequent between October and February,
in the warmer months. Courting takes place on tree limbs, and the male will
consort with the female for up to two and a half weeks. Mating is promiscuous,
and may take place with multiple partners within the year. Gestation is up to
13 days; two small pink joeys crawl into the pouch and develop there for as
long as long as 7 months, growing and suckling. Lifespan is up to 12 years,
sexual maturity is reached at 1 year. Predators most often are the eagle
kea, on the ground it may fall prey to the Ruacapangi
bird or Marsupial Pardus, it is usually too large to be targeted by hawks.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Banjora,
False Koala (Pseudophascolarctos rotundus)
Order: Two Fore Teeth (Diprotodontia)
Family: Phalangers and cuscus (Phalangeridae), Subfamily Pseudophascolarctidae
(monotypic)
Habitat: Jarrah forests of the southeast of Meganesia, temperate forests of
eastern Meganesia, exclusively where eucalyptus trees grow.
Specialized kinds of animal often suffer the worst effects of mass extinction,
and thus the Koala suffered extinctions at the hands of man’s forest clearing
and disturbance in Australia during the Late Holocene. But the niche of a slow-clambering
exclusive leaf-eater is one that has often been repeated by evolution, and so
it happens again with the False Koala, or Banjora (coming from an Aboriginal
boy’s name meaning Koala). The ancestor of this species was the rather generalist
Brushtail Possum (Trichosurus vulpecula), as we often see, specialists commonly
evolve from more generalized ancestors.
Outwardly it is a stocky round animal with a large hindquarters and a stubby
tail, this tail serves some purpose of fat storage but is otherwise useless.
An adult male can weigh up to 9 kilograms, with a body length of 78 centimetres,
females are slightly shorter but weigh about the same. Its head is large and
supplied with heavy chewing muscles, the snout is short and arched. They feed
entirely on the leaves of various kinds of eucalyptus tree. The original Koala
was restricted to consuming only a few species, but the Banjora can gain sustenance
from most kinds of eucalypt. Its nose is dark in color but resembles that of
a possum more so than a true koala, eyes are small and brown, vision is poor
but hearing and smell are acute. Ears are slightly enlarged but are pointed
and do not resemble those of a koala, having a thin coat of white hairs. Colour
and softness of the fur is intermediate between that of a koala and that of
a Brushtail Possum, thick, fluffy and colored brownish grey, but the rear and
face are darker, almost black. Hands and feet are both built for gripping branches,
and bear very large curved claws, both hands and feet bear prehensile thumbs
but that of the foot does not bear a claw, syndactyly of the foot is not overly
pronounced.
Banjora are able to clamber about in the treetops and even make short leaps
between trees, but are more adept on the ground than a true koala, often crossing
larger distances between trees on foot. These marsupials are diurnal but are
most active in the afternoon. Predators include various carnivorous climbing
marsupials, some larger kinds of monitor lizard, and raptorial predatory birds
such as eagles, hawks and owls. Banjora may defend themselves by biting hard
or by scratching with their large fore claws. Vocalisations vary from a guttural
hiss or chatter when annoyed, to a loud scream uttered by the male during breeding
season.
Banjora are generally solitary, but breeding is sporadic all year long, peaking
in the spring, from September to November. Reproduction is slower than their
ancestor, and usually only one young is born. As with all marsupials this baby
is born poorly developed, and matures in a pouch, which faces outwards and slightly
towards the mother’s chest, two teats are present. At 6 or 7 months, the joey
has left the pouch, it is fully weaned and independent by 12 months. The mother’s
milk is poorer in nutrients than with other marsupials, and the joey is fed
milk for a longer time. Joeys will often consume its mothers faeces in order
to gain the correct gut flora of bacteria in order to digest eucalyptus leaves
in the future, but the mother does not produce special pap in the way of a true
Koala. Home ranges are generally small and overlap with others, but these animals
are not very social, and fights often break out. Several males may court a single
female when she is receptive, and some amount of fighting and resistance preludes
the mounting and insemination. Both males and females produce a strong musky
scent which is stronger when they are breeding. Lifespan is 12 to 15 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Marsupial grizzly (Gravipossum ursinus)
Order: Diprotodontia (Diprotodontia)
Family: Predatory possums (Carnopossumidae)
Habitat: forests and woodlands of Meganesia, plains of southern part of continent
(up to coast of Eyre Gulf) and Great Dividing Ridge.

Picture by Alexander Smyslov
In Holocene Australian marsupials had suffered significant
damage from the introduction placentary predators to Australia, and from destruction
of megafauna by first aborigenes. At the continental part of Australia marsupial
lions (Thylacoleo), marsupial wolves (Thylacinus) and marsupial devils (Sarcophilus)
had died out. In historical time the Tasmania Island remained last shelter of
marsupial wolf (smaller, rather than its continental species) and marsupial
devil (also smaller than species exterminated at the continent). The place of
marsupial predators on continent had been actually occupied by people and feral
dogs, dingoes. In Neocene when both people, and placentary predators had died
out of different reasons, marsupials had started to evolve actively, occupying
the exempted ecological niches. Among new marsupial predators of Australia descendants
of both “true” predatory marsupials (Dasyuridae), and other animals passed to
predating in parallel to them had appeared. In Neocene possums had made the
second attempt to become carnivorous creatures (marsupial lion was the first
and rather successful attempt). Being not able to compete with dasyurids, carnivorous
possums had turned to massive animals, similar to bears by constitution. The
size of marsupial grizzly, largest one of them, reaches 3 meters in length at
weight about 500 kg.
Marsupial grizzly is one of largest species of marsupial mammals of the world.
Longteeth wombats,
descendants of Holocene wombats, reach even bigger size, but among possums this
species is true giant. Even huge New
Zealandian species of possum is much smaller.
Marsupial grizzly is very similar to massive bear with rather short extremities
and back curved upwards. At it there are plantigrade extremities armed with
sharp claws up to 10 cm long. Being attacked by predator, the marsupial grizzly
defends, rearing on hinder legs and delivering crushing blows by front ones.
It had not lost the ability to climb on trees though adult animals do it hardly.
But young marsupial grizzlies spend a lot of time in crones of trees where they
eat foliage and fruits, and ravage bird's nests. Adult animals feed mainly on
the ground.
Possums known to people differed in long tails. At marsupial grizzly which spends
on trees only small part of life, the tail became short and thick. It has lost
past function of the balance weight and the “fifth paw”, and now in it the stock
of fat is accumulated, helping to go through fodder shortage.
Head at this marsupial is rather large and massive. Ears of animal are short
and round; nose is big and lips are mobile. Eyes of marsupial grizzly are small
– the animal is short-sighted, and in search of food bases at sharp sense of
smell. Marsupial grizzly has yellowish-grey fur with dark paws and nose bridge.
Stomach of animal is white, on back from waist up to the basis of tail narrow
black strip stretches. Feet and hands of animal are covered with thick cornificate
skin.
Jaws of marsupial grizzly are short and have slightly increased canines and
wide molars. Strong chewing muscles help this marsupial to chew fibrous parts
of plants and heads of bones of large animals. This species of animals partly
plays a role of scavenger in ecosystems of southern and eastern parts of Meganesia.
It finds rests of catch of large local predators, and eats them almost completely:
after the feast of marsupial grizzly only separate vertebrae and bones of legs
remain from carcass of animal. Also it can drive away smaller predators from
catch. The males of this species not burdened with cubs bearing can go fishing
in shallow rivers and lakes.
Marsupial grizzly is not completely carnivorous species of marsupials: it is
omnivore, and whenever possible willingly eats vegetative food. With the help
of long claws animal digs out roots of ferns and other eatable plants. It can
dig out holes of small animals just as it was done by bears in Holocene epoch.
Because this animal is omnivorous, at it the increased intraspecific aggression
characteristic for carnivorous species is not shown. Besides females at marsupial
grizzly are larger, rather than males, therefore cubs are not threatened with
danger of attack from their side while they stay with mother.
Brooding pouch at this species of mammals opens back, and there are four nipples
in it. Fertility of the female of marsupial grizzly reaches ten cubs, but only
four of them receive an opportunity to survive, and usually two cubs survive
up to independence. In the beginning the posterity of marsupial grizzly is “worn”
in mother’s brooding pouch over half-year. After that cubs change to back of
mother, and keep by paws for her wool. The posterity stays with mother up to
one-year-old age, and right when young animals abandon the female, it becomes
ready to pairing again.
Picture by Alexander Smyslov
At the eastern slope of Great Dividing Ridge, in humid mountain
woods the relative of this species, the marsupial panda (Gravipossum
paraailuropoda) lives. This species is much smaller, rather than marsupial
grizzly: the adult female weighs about 300 kg at length of body up to 2 meters,
and the male is 25 – 30 kg lighter. In food predilections marsupial panda differs
from marsupial grizzly by appreciable bias in vegetarianism. The food of animal
origin amounts a small part of diet – marsupial pandas eat eggs of ground-nesting
birds, and willingly consume carrion. Frequently animals search for forage on
banks of mountain rivers, feeding on water invertebrates and local migrating
fishes of Galaxiiformes order. But the basic food of this animal is made of
roots and leaves of grassy plants, and also sprouts of bushes. Living in cool
mountain climate, marsupial panda differs in rich wool. At this animal there
are black head, shoulders and back, rounded white spots above eyes, grey sides
and back part of body. The tail of marsupial panda has snow-white inner side
which is used by animals for submission of signals to congeners.
Fertility of marsupial panda is insignificant: the female gives rise to no more
than five cubs from which only one or two ones survive up to independence. Life
expectancy of this animal reaches 40 years.
Marsupial
biruang (Carnopossum heliarctoides)
Order: Diprotodontia (Diprotodontia)
Family: Predatory possums (Carnopossumidae)
Habitat: mountain and lowland rainforests of north and northeast of Meganesia.

Picture by Alexander Smyslov
In Neocene Australia and New Guinea had merged to unite land
massive named Meganesia. This continent continuously moves to the north, to
the equator, “crushing” edges
of lithosperic plates of Indonesia. Actually, this circumstance became the
reason of raising of northern part of Meganesian lithosperic plate.
Movement of continent has caused the considerable changes in climate of territories
included in continent. The northern Meganesia in Neocene is in the area of
equatorial climate that has positively had an effect on nature of continent.
The north is
covered with rich rainforests. And in forest swamps rivers originate and flow
to the south and carry water to areas represented rigorous waterless deserts
in Holocene.
Rainforests of the north of Meganesia are rich in life. Various birds, reptiles
and amphibians, and also unnumerable species of insects live here. Besides,
the forest canopy is inhabited by various small and medium-sized mammals, and
large
species of mammals are found in underbrush. Among them there are descendants
both of native Australian and New Guinean species, and descendants of species
introduced by people.
Rainforest is a habitat of rather large species of marsupial mammal which is
named marsupial biruang. It belongs to group of carnivorous possums classified
in family Carnopossumidae. Its close relatives are huge marsupial grizzly living
in the south of continent, and marsupial panda – smaller species from mountain
areas at the east of the continent. Marsupial biruang is the smallest representative
of carnopossumids: weight of adult animal is only about 60 kg. It is the ecological
analogue of Malayan bear, or biruang, known in human epoch – hence the name
of this species.
Marsupial biruang has robust constitution and rather short limbs. Similarly
to all carnopossums, it has very short tail which does not take part in tree-climbing.
On paws of animal long hooked claws grow; with their help marsupial biruang
easily
gets on high trees. Fingers of animal are mobile and permit it to gather very
small objects and to catch insects. The wool of this animal has black or dark
brown color, and only above eyes there are two light grey spots. At young animals
spots above eyes are absent. On hands and feet wool is thin, and through it
grey skin of animal is visible.
Eyes of animal are directed forward, and provide enough wide sector of three-dimensional
vision. Ears at marsupial biruang are short, rounded, slightly jutting out
from wool. At this animal there are rather short muzzle and increased forward
cutters.
Molars are obtused: their shape is characteristic for omnivores.
Marsupial biruang uses for life all levels of rainforest – from underbrush
up to forest canopy at height over 20 meters. This is sluggish single animal
marking
borders of territory with the help of musky liquid secreted by two glands at
the root of tail.
This animal is omnivorous and easily finds enough of food. It eats fruits of
various tropical trees, sappy leaves and stalks of epiphytic plants. Also this
animal catches large insects and digs out of ground worms and grubs. If the
opportunity is represented, marsupial biruang catches little mammals and ravages
nests of
birds. Any animals from insects and snails up to vertebrates weighting up to
3 – 4 kgs become its prey.
Having caught prey, marsupial biruang perches on wide strong branch of tree,
sits on hinder legs and eats food, holding it in forepaws. After meal the animal
licks wool by long tongue, and “washes” by forepaws like cat. Marsupial biruang
is active in day time, and only in hottest time of day sleeps till some hours,
having chosen a shady place.
At this species of marsupials the female is larger and heavier, than the male.
Animals do not form constant pairs, and meet only for the period of pairing.
The pouch at the female opens forward – when female swarms up a tree, such
position of pouch is safer for the large cub. The female bears only 2 – 3 rather
large
cubs (large to measures of marsupials – newborn marsupial biruang weighs about
10 grammes). Some months later only one cub from the litter usually survives.
The female looks after it for a long time: the cub stays in pouch till about
seven months. Having left the pouch, it follows the female and learns to search
for food till about three – four months. At this time the female can couple
with another male and bear new posterity. When the new cub is born and gets
into a
bag, the female starts to show aggression to the previous cub, and it passes
to independent life.
Sexual maturity at marsupial biruang comes at the age of four years, and life
expectancy reaches 40 years and more.
The idea about existence of this species of animals was proposed by Simon, the forum member.
Pouched
Un-margay (Thylofelis arboricola)
Order: Two-front Teeth (Diprotodontia)
Family: New Zealand Predatory Marsupials (Paradasyuridae)
Habitat: Forested areas of the North and South Island of New Zealand, including
mountain forest, conifer forest, tropical forest etc.
The introduction of different foreign mammal species to New Zealand by man,
had a devastating effect on its fauna in the Holocene, but presented new opportunities
for evolution in the Neocene.
The Brush-Tailed Possum (Trichosurus vulpecula) gave rise to several Neocene
forms here, including the predatory Paradasyuridae, which converged upon both
cats, and extinct marsupial lions (Thylacoleo). This group includes great apex
predators such as the Marsupial
Pardus, but also smaller cat-like arboreal predators such as the Pouched
Un-margay.
This marsupial resembles a partway between a small jungle cat, and its ancestral
form, the possum. Adults reach up to 50 centimetres in body-length, tail is
a little longer again than the body, reaching 60 centimetres, weight may be
up to 4.5 kilograms. Limbs are powerful and flexed, ending in gripping paws
that bear large curved claws, hands have a large semi-opposable thumb while
the hind foot has a large opposable toe to grip branches. This animal appears
more slender than its ancestor, spine is sinuous and flexible, animal is able
to leap between branches, or pounce upon prey with ease. Tail is long and bushy,
and ends in a large black tuft. Skull is large and rounded, eyes are large and
face forward, in this way it resembles a small cat. Coloration is yellowish
grey with a darker back, irregular black spots and cross-stripes towards the
rump, tail tip is black. Ears are large, pointed and movable, resembling its
ancestor, hearing is keen. Face is covered in a black “mask” around the eyes
and mouth, nose is naked and dark pink. Paws are covered in black hair on the
upper side, soles of feet are bald and pink. Its dentition recalls both Holocene
possums and extinct marsupial lions, incisors are large and pointed, prey is
killed with a bite to the throat. Premolars are large and blade-like, serrated,
molars bear short pointed cusps, rearmost molars have rounded cusps for crunching
small bones.
Pouched Un-margay is an adept tree-climber and spends most of its time in the
trees, it can quickly run along branches and up and down trunks headfirst. This
animal is a nocturnal predator of usually vertebrate prey, diet includes birds,
rodents, and other kinds of small marsupial such as possums, large insects and
lizards are sometimes taken, and they will eagerly consume nests of bird eggs
or hatchlings. Prey is stalked from cover, and leapt upon in a long pounce much
like a cat, they may drop down from branches above onto prey, or drive them
to the tips of branches. Most prey is killed with a powerful bite to the throat,
large incisors sever the windpipe and crush the neck. Calls of this animal include
a coarse hiss or guttural screech when alarmed or excited.
Adults are solitary outside mating and courting, mating may occur year round
but reaches its peak from September to November. Mating is promiscuous and opportunistic,
individual males may sire multiple young within a year. The male will consort
with the female for up to 2 weeks, mating several times, gestation is up to
16 days. Female gives birth to a single joey, small and pink, which migrates
to the pouch, development inside the pouch lasts up to 6 months. After this
time, the joey may be left in the mother's den or cling to her mothers back,
and remains with her for up to 7 months. Sexual maturity is reached at 1 year
old, and lifespan may be up to 16 years, predators include hawks, eagle
keas, and on the ground they are vulnerable to canids
and the Ruacapangi bird.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Night
marsupial lemur (Nyctiphalangeropsis strigops)
Order: Diprotodontia (Diprotodontia)
Family: Marsupial lemurs (Phalangeropsidae)
Habitat: rain tropical woods of Meganesia.

Picture by fanboyphilosopher
The canopy of tropical rainforests stretched at height 20
- 40 meters gives a plenty of opportunities for adaptation to various live creatures.
Among its inhabitants it is a lot of marsupial mammals. Those species have survived
at the border of Holocene and Neocene, have inherited from missed species many
free ecological niches, and have occupied them, having evolved to fantastical
lifeforms. The competition between inhabitants of rainforest canopy can be very
rigid, therefore during evolution live creatures developed to lower it as much
as possible. Certainly, it is possible to become the specialized “expert” in
consumption of the certain kindss of forage, and it is possible simply to live
and not to prevent to another ones. One of animals living in this wood had made
it.
When night falls on wood, the majority of wood inhabitants hides in tree-trunk
hollows and nests and falls asleep. But some animals just at this time wake
up and quicken. Among them there are various insects, frogs, lizards, some birds,
and also small mammals covered by black wool with white marks. Their movements
are very similar to movements of monkeys; their heads are big, have high cheek-bones
and large “owline” eyes. Animals keep in small group, supporting contact with
the help of quiet chirp. They are similar to small monkeys, but they are not
primates at all, but descendants of cuscuses – night marsupial lemurs.
The night marsupial lemur is insignificant: length of its body is about 20 cm,
tail length – about 30 cm. All paws of these small mammals are adapted to branch
gripping: on forepaw there is well advanced thumb, and toes on hinder legs are
opposed to each other by two groups: joined together I, II and III against joined
IV and V. Bent claws grow on ends of fingers and toes. The animal moves on trees
like squirrel: it runs on branches and swarms up trunks, clinging by claws against
bark. Due to tenacious paws night marsupial the lemur dexterously swarms up
the thinest branches. It is able to jump from tree to tree like monkey: having
shaken on thin elastic branch, the small mammal makes jump some meters long.
During flight in air the mammal stretches paws in sides to glide a little bit
and thus to increase range of jump. The prehensile tail increases opportunities
of animal: due to it night marsupial lemur can hang on branches.
The nocturnal habit of life is reflected at the appearance of this animal: eyes
of the night marsupial lemur are very large, and because of it the obverse part
of skull looks rather wide. On muzzle of mammal long whiskers grow with which
help the animal finds way in dark night wood. Colouring of wool is coal-black,
but for recognition of neighbours at night marsupial lemurs there are contrast
marks – white stains on chest and tail tip. Around of animal’s eyes there are
rings of white wool because of what this small mammal looks a little horribly:
rings create illusion of eyes belonging to larger animal. Eyes of this marsupial
lemur are yellowish-brown; pupils are vertical, as at cats. This animal distinguishes
colors badly, but it is capable to see world around even at weak light of stars
in moonless night.
At night marsupial lemurs there is good hearing, though auricles very short.
But the ear aperture at them looks like a crack, and on back edge it is edged
by fringe of rigid fur functionally replacing an auricle. Due to sharp hearing
the animal can search for insects in darkness, and frequently to it succeeds
even to hear flying owl: it is rather useful property for nocturnal animals
of small and medium size.
The night marsupial lemur is a sociable animal; it lives in groups of 3 – 5
individuals. All group consists of male and several females, and also of cubs
have not reached independence. These animals spend day in tree-trunk hollow
on their territory. They know an arrangement of all suitable shelters on their
area, and each morning they stay in new one. By this way animals prevent the
parasite breeding and hide their resting place from predators.
Night marsupial lemurs wake up, when day time heat falls down, and day time
predators search shelter to lodge for a night. Usually dominant male or the
main female shows itself first from tree-trunk hollow. The animal cautiously
smells air, convincing of safety, and then comes out on the branch. After it
other members of group appear. Animals clean wool, stretch, and start to inspect
territory. They move on branches in a line, keeping a distance. During the night
small mammals become especially cautious: at this time owls fly to hunt. To
protect itself against attacks of these birds, night marsupial lemurs choose
way in richest branches, and jump from tree to tree one by one, stopping for
a little time on trunk after landing. Other enemies of these animals are snakes.
The silent rustle similar to sound of the moving snake is the most terrible
sound for night marsupial lemurs.
The group marks borders of the territory with the help of secretions of repugnatorial
glands located in armpits. For this purpose they simply drag branch between
body and paw. All members of group mark territory in same place, but on different
branches, therefore smells of separate animals do not mix up also any newcomer
can estimate number of animals protecting this territory. However, dominating
male will do not be against at all if the newcomer will appear the female. When
it is a lot of forage, it can admit to clan even other male, but already beforehand
a role of satellite and “whipping boy” will be determined to it, and it will
druggle at the end of group. But the solitary animal in almost cases is restrained
with such role: it is easier to notice danger together. During the movement
group supports contact: animals utter chirp and abrupt sounds similar to bark.
Night marsupial lemurs are omnivorous: they will ravage the bird's nest or will
tear off ripe fruits from branch with the same pleasure. In searches of vegetative
forage they are guided basically by sense of smell, correctly distinguishing
ripest fruits by smell. One more dainty is thick juicy stalks of some orchids.
The best food of animal origin includes insects and spiders. If fruit appeared
worm-eaten, these mammals will to have eaten it entirely, right including caterpillar.
The special dainty is delicate soft termites. At night these blind moisture-loving
creatures leave their shelters, and night marsupial lemurs willingly lick them
off from bark.
Seasonal prevalence in breeding at night marsupial lemurs is not present: practically
at any time it is possible to find groups with cubs. Female twice per one year
gives rise to 7 - 8 cubs though it can feed only four ones. The pouch opens
back at this species. Cubs spend in it about two months, then they get out on
back of mother and travel on it, seizing its wool. This time is the most important
for their further life: cubs study to search for food and seize skills of tree-climber.
When they wean from dairy diet, their connection with mother weakens. Cubs can
get on backs to other members of group though those can be not so pleased with
it, and seldomly bear too long driving another's “youngs” on the back.
Sooner or later young animals abandon clan. They go to “neutral” (“drawn”) boundary
territories of different clans, and gradually teenage groups break up. Young
females more often succeed to join to other clan, and the destiny of males develops
more dramatically: at them probability to finish the barchelor life in stomach
of the snake or an owl is most of all. Occasionally the young male finds teenage
group and expels from it other males, forming a high-grade clan. Being lucky
it can “overthrow” the old male and take hold of available clan.
Life expectancy of the night marsupial lemur is short: it seldomly lasts more
than 10 years.
Hook-fingered kangaroo (Gravimacropus macrocheirus)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroos (Macropodidae)
Habitat: light forests and bush thickets of Southern, Central and Eastern
Australia, foothills of Great Dividing ridge.

Picture by Timothy Donald Morris
During the evolutionary history kangaroes belonged to among the most successful
marsupial animals of Australia. They evolved rather quickly, adapting to
changes of world around, and in historical epoch competed on equal terms
with sheeps
and camels introduced by people. In Neocene the significant part of domestic
animals introduced by people to Australia, had died out, and only camels
evolved to some species adapted to various habitats became real competitors
of kangaroes.
Kangaroes had also evolved, and among them various kinds had appeared:
some of them eat foliage, others graze, and some ones even eat insects
and carrion.
Climatic conditions of Neocenic Australia favour to development of large
life forms, and among kangaroes one such species had appeared.
The largest kangaroo of Australia is the hook-fingered kangaroo. It is
huge species up to 3 meters in height in biped pose. The hook-fingered
kangaroo
has shape characteristic for representatives of this family, but adjusted
for larger size and massive constitution. This animal weighs about 250
- 300 kg,
therefore it jumps slowly and hardly, more preferring to move by original
slowed down “gallop”, moving alternately pairs of front and hind legs.
Forepaws of
the hook-fingered kangaroo are adapted to such type of movement: the animal
leans on the external side of long hands covered with thick skin, similarly
to gorilla. On fingers of this animal thick hooked claws grow. At movement
the animal pushes from the ground by tail which is considerably short in
comparison with tails of usual species of kangaroes. On the bottom side
of tail the cornificated
covering, similar to callouses developing on legs of camels, is advanced.
But this “callous” does not serve for protection against burns as at camels,
but
provides the best cohesion with ground at the movement.
Hinder legs of the hook-fingered kangaroo are lengthened: it is a characteristic
attribute of all kangaroes. But they are not much longer than forepaws,
short of a foot. Foot at this species of kangaroo is long and elastic,
and claws
are not sharp, but to a greater extent hoof-like and blunt. The hook-fingered
kangaroo does not concern to list of fast runners: it prefers to not escape
from predators in flight similarly to the majority of its congeners, and
to protect itself in close combat. This animal defends against enemies
by impacts
of forepaws, gashing it by terrible lateral impact strengthened with a
turn of all body. This animal counter-attacks large enemies and tries to
tear
them by claws of hinder legs.
The wool of hook-fingered kangaroo is colored bright - red. Head and forepaws
at it are dark, almost black, and hinder legs and the bottom part of tail
are lighter, than the basic colouring of body is. On head there is the
extensive white “mask” covering muzzle, eyes and a part of bottom jaw.
This animal is original analogue nowadays extinct sharp-clawed perissodactyls
chalicotheres (Chalicotheriidae). The hook-fingered kangaroo eats leaves
of bushes and trees. Usually it is feeding, standing on hinder legs and
tightening tree branch to a mouth by long forepaws. Also it eats soft species
of grasses,
not competing with other species of kangaroes eating rigid siliceous graminoids.
Chewing muscles are advanced rather poorly at it: the animal chews a little,
and the food is processed mainly in the complex stomach consisting of three
chambers. Symbiotic protozoans improving quality of food digesting, live
in stomach of this species. The feature of physiology of the hook-fingered
kangaroo
is the ability to make some of metabolic water which is formed at fat oxidation.
This adaptation helps this animal to live long time without water: the
hook-fingered kangaroo comes to the watering place only once a week, and
even less often.
Jaws of hook-fingered kangaroo are short, and the obverse part of skull
is short and expanded. Eyes are shifted in sides and provide the good circular
field of view. Also at the animal there are wide mobile ears: hearing at
the hook-fingered kangaroo is keen. The olfactory cavity also is expanded:
therefore
the head of animal has no “deer” shape usually characteristic for kangaroes.
Wide ears and nasal cavities help large animal to cool itself in strong
heat.
This species of kangaroo is solitary animal though it does not avoid a
company of congeners. In places rich in forage this species meets by small
groups
of 4 - 5 adult animals. Usually animals keep together during pairing. The
courtship
season at this species is strongly dragged out, and females ready to pairing
meet the most part of year, except for the droughtiest months. The female
ready to get posterity, emits the special smell on which it is found by
males. Usually
one female is looked after with true “retinue” of males. But one of them
shows the greatest activity, driving other ones away. Fights between males
are limited
to struggle with the help of forepaws. Kicks in stomach of the contender,
characteristic for other kangaroes, at this species are excluded: animals
are too massive
for it.
After short pregnancy the female gives rise to one small cub, which should
get into parental pouch itself similarly to posterity of all marsupials.
The period of bearing of posterity in pouch at the hook-fingered kangaroo
is very
long: about half-year the cub constantly sits in pouch, sucking milk, and
still almost half-year it is hidden in it at the case of danger. But up
to age of
fifteen months the cub continues to suck milk of mother though to another
nipple its younger brother has already attached. Young animals differ from
adults
in monotonous colouring of body. It changes approximately to three-year
age, and four-year-old animals already can breed. Life expectancy at this
species
may be over forty years.
The idea of existence of this animal was proposed by Simon, the member of forum.
Hobyah-Wallaby
(Arborogale pithecoides)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroos (Macropodidae)
Habitat: Warm and temperate forests of South-Eastern Australia, cool forests
of Great Dividing Range.
During the Pleistocene and Holocene, kangaroos and wallabies experimented successfully
with tree-dwelling habits. Not only were there Tree-Kangaroos (Dendrolagus),
but a separate kind of Ice Age wallaby capable of climbing trees to feed (Congruus).
In the Neocene, there are no descendants of Dendrolagus due to endangered status
and habitat loss, but more unpretentious wallabies (Petrogale), have taken to
the trees and produced broadly similar forms. Not only this, but it also occurred
convergently in Hawaiian Islands, from invasive members of the same group, Monkey
Wallaby.
These Meganesian Tree-Wallabies are typified by the Hobyah-Wallaby, which is
found over a large range in south-eastern Meganesia. It is named after mischievous
imp of Australian folkloric fiction. Bodily it resembles a tree-kangaroo, having
a short body, flexible hunched back, shortened feet, and powerful arms as long
as the hind legs, with big hand claws. Hind feet are flexible and able to turn
soles inward, feet can move independently to “walk” along branches. The neck
is upright and flexible, and its head is somewhat more massive than a typical
wallaby. Snout is of fair length but deeper with an arched nose, lips are large
and flexible, not split, forming a small proboscis to aid in feeding. Eyes face
more forward, allowing animal to judge distance when climbing and jumping, it
is able to see in ultraviolet and can perceive the colours of fruits or flower
heads. Ears are large and mobile, but rounded at the outer edge, resembling
mouse’s ones. Tail is long and flexible with a “brush” at the end, in order
to balance the animal; it is able to make short leaps between trees and even
may descend to the ground by leaping as far as 6 meters. It moves about by clambering
between and along large branches, and it is able to ascend vertical trunks.
Hobyah-Wallaby reaches the size of a monkey or a child, up to 20 kilograms,
males are larger than females, but both are equally muscular to aid in climbing.
Pouch is large and faces forward and a little upward so that joey will not fall
out. Coloration is a dark brownish grey, with thick blackish-brown cross-bands
on the back and hindquarters, extremities of limbs, tail and face are all darker
blackish brown. Skin of ears, nose and soles of feet are black. Its diet consists
of the soft leaves of broadleaf trees, eucalyptus and other coarse leaves are
not taken, fruits and flowers are eagerly consumed, as are wood fungus.
As with Holocene Tree Kangaroos, it is generally solitary, individuals have
a distinct large territory, and only come together to mate. Males court females
in the cooler wetter part of the year, but female is receptive for much of the
year. Males seek females by smell, both sexes release a musky glandular odour
advertising receptiveness, and in the case of males, vigour. Opposing males
fight by biting and “boxing” with fore-paws, and attempt to drive the other
to climb down and retreat. This wallaby is usually only active during morning
and late afternoon, including dawn and dusk, individuals hide during the middle
of the day.
Females will give birth twice a year, and they are able to rear a new fetus
when the older joey is still suckling. Joey starts to leave pouch and climb
at 3 and a half months, and moves independently at 4 and a half months, weaning
2 months later. During following year’s courtship the young joey will leave
the female, becoming sexually mature at 2 years old. Lifespan is up to 30 years.
Predators include large raptorial birds, and climbing predatory marsupials,
stray joeys may be consumed by pythons or monitors.
Related species is the Imp-Wallaby
(Arborogale agilis) found in the tropical woods and rainforests of
north-eastern Meganesia. It differs by somewhat smaller size, weight usually
of only 15 kilograms. Colour is a yellowish brown, with dark-reddish cross-bands
along the back and hips, extremities are coloured chocolate brown, as is the
face. It also differs by more slightly shaped head, a little more similar to
a typical wallaby, lip forms a flexible point as with its relative. Fruit makes
up a greater portion of its diet than its relative; it will consume the flesh
of many non-poisonous tropical fruit. As against relative, breeding is preferentially
done in the drier months when there is less risk of storms and flooding.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Caprine
Wallaby (Capromys dexter)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroo (Macropodidae)
Habitat: Woods, scrub land and forests of Eastern Meganesia, from Great Dividing
Range into sub-tropical zone.
Smaller herbivorous marsupials such as wallabies, managed better against invasive
species, and habitat loss during the Holocene human occupation of Australia.
The forms left after the extinction of man, radiated into niches made absent
after the Holocene extinctions.
The genus Petrogale, rock-wallabies, survived and speciated in some numbers,
giving rise to not only larger species, but arboreal and even predatory forms.
Caprine Wallaby is a kind of remaining transitional form between rock-wallabies
and Neocene tree-dwelling species, a fair-sized wallaby that is able to feed
both on the ground, as well as by climbing rock faces or large trees and shrubs.
This species generally resembles a rock-wallaby, apart from somewhat shorter
and broader hind-feet, and longer more muscular arms, ending in powerful clawed
hands. Wallaby reaches a weight of about 22 kilograms and a length of up to
130 centimeters, and is a mixed feeder with a strong bias towards the leaves
and buds of trees and shrubs, as well as soft herbs. This marsupial spends a
great deal of its time climbing trees and shrubs in order to feed on leaves,
adults are able to scale the trunks of large and mid-sized trees that to dot
have a long shaft of bare trunk. Individuals mainly climb by clambering between
branches, and walking along boughs (foot bones are flexible enough to alternate
stride), branches and leaves are pulled towards the mouth using its hands. Face
is elegant and generally similar to a Holocene wallaby, but slightly deeper
in the jaw; dentition is adapted to chew a wide range of kinds of leaves, shoots
and buds. It also spends time feeding on the ground, on understory plants such
as shrubs and herbs, as well as sometimes climbing hilly outcrops and crags
in order to feed on inaccessible growth. In this physical adaptability of feeding,
it is so named Caprine Wallaby, similar to a feral goat which may climb trees
and rocks to consume plants.
Coloration is a fawn-grey colour, with a pale cream coloured belly, hands, feet
and tip of tail are all a dark chocolate brown, head is dark brown with a pale
“spectacle” marking around the eyes and mouth. Skin of nose, inside of ears
and soles of hands and feet are all brownish pink. Ears are long and slightly
pointed, recalling a rabbit or other wallaby, lip is split and rather long,
flexible in order to aid feeding. Eyes are large and dark, sensitive to movement
and shape especially in order to detect predators.
Caprine Wallaby is usually most active in the morning and afternoon, including
dawn and twilight, by day it will usually hide in lee of a crag or up a tree
in order to rest. This species lives usually in small groups consisting of a
breeding male, one or two females, and their mature young, male youth are usually
expelled from the group upon reaching sexual maturity. Mating occurs in the
cooler wetter part of the year, August until mid November, females are receptive
throughout this time but usually only bear one young each season, second developing
fetus may be delayed until the following season. Groups have fair-sized territories,
where female strangers are tolerated, and interloping males driven out. During
the breeding season, stray males may challenge family leaders for access to
females, fighting by “boxing” with arms and kicking, as well as vigorous displays
by thrashing and throwing branches and leaves with arms. Joey starts to venture
out of pouch after 10 weeks, and is independent of pouch and weaned by 6 months,
sexual maturity is reached at 2 and a half years usually. Lifespan is up to
25 years but usually shorter; predators include large carnivorous marsupials,
birds of prey, varanid lizards and sometimes large snakes.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Monkey
wallaby (Pithecomacropus agilis)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroos (Macropodidae)
Habitat: rainforest of Hawaiian islands (Oahu, Molokai, Lanai, Maui, Kahulawi),
forest canopy.

Picture by Tim Morris
Kangaroos never lived on Hawaiian Islands, similarly to representatives
of other groups of ground and flightless mammal. But in human epoch they have
appeared even in this isolated corner of Earth, being introduced there by people.
And it was species of mountain wallabies Petrogale penicillata (brush-tailed
wallaby) became extremely rare in true native land, in Australia, later. Pair
of these animals has run away from a menagerie in the beginning of XX century,
and was multiplied on Oahu Island. Decrease of ocean level till an ice age had
permitted to this animal to settled on the near islands: Molokai, Lanai, Maui
and Kahulawi. Obviously, this moving was unitary: Oahu Island is separated from
them by rather deep Kaiwi passage which had kept in ice age, and obviously,
it had been crossed casually by a little group of animals – founders of new
population. And the further moving of wallabies to the east on islands of the
Hawaiian ridge was blocked with Alenuihaha passage separating these islands
from Hawaii Island. Therefore moving of descendants of marsupial immigrants
on islands is limited to these islands. In the period when the volcanic island
Hekeua had been formed, the part of populations of this kangaroos has almost
disappeared, having kept in small amount only in rests of woods at coast. But
later, due to characteristic for marsupials ability to fast breeding, the population
of Hawaiian wallabies was restored. So these marsupials relieved by the nature
of competitors, others large mammals, have remained on Hawaii.
The monkey wallaby has considerably changed a habit of life characteristic for
ancestors. Now this is a forest animal, something similar to the “true” Holocene
tree kangaroos of New Guinea. It is the small species of kangaroos: it is cat-sized
animal, but seems larger because of long tail. This animal has found a fine
ecological niche in woods and became the largest species of herbivorous animals
in rich canopy of the Hawaiian rainforest.
Monkey wallaby dexterously swarms up trees. Joints of back extremities at it
are very flexible, and the foot is partly swivels sideways: the animal clasps
so a tree trunk more densely. On front and hinder legs hooked claws are advanced.
Grown together toes on hinder legs serve for the toilet purposes. This animal
clambers on trees “galloping”, working synchronously in pairs by forward and
back extremities. Perhaps, the monkey wallaby is unique species among kangaroos,
able to move back: this animal goes down from trees a tail first, serially grasping
a trunk by front and hind legs. Similarly to monkeys, this animal can make big
jumps from tree to tree, and is able to swarm up thin lianas dexterously. If
the branch or liana is especially thin, animal is able to move on it similarly
to sloth - a back downwards, clinging for a support by bent claws. A tail of
this species is thin and long: in length it exceeds body and head taken together.
Tail is not prehensile, but elastic: it serves as the balance weight during
jumps and movement among branches.
The skull of monkey wallaby is rather short and wide, as gives to this marsupial
the “monkey” shape. It is connected to several features of its habit of life.
First, the important advantage to this species is the binocular sight assisting
to estimate distance up to the next branch precisely. And second, the animal
eats rather soft food: fruits and soft leaflets, therefore its jaws became weaker
and have decreased in size.
Sight of the monkey wallaby has one feature: the animal can perceive ultra-violet
light. Therefore animals of these species easily find among foliage green fruits
covered with wax, reflecting this part of solar spectrum. Ears of the monkey
wallaby are wide and mobile; animal has very good hearing. Ears are hairless,
pink due to blood vessels appearing through skin: in hot weather vessels extend,
and surplus of heat is emitting through ears.
Coloring of body of monkey wallaby is disruptive: head is black with longitudinal
white strips stretching from nose to ear through eye, on forepaws and shoulders
there is light red “cloak”, tail is grey with black tip. Waist and hinder legs
are dark brown.
Being similar to monkeys externally, this animal is absolutely not similar to
them by habits: monkey wallaby is solitary animal, and two adult animals can
be together only in short courtship season. Monkey wallabies are very territorial
animals. Each individual occupies territory about a quarter of square kilometer,
and marks its borders by urine. This animal does not arrange constants shelters,
and spends night in casual refuges. And adult strong animals spend the night
even simply on branches.
Two times per one year the female brings posterity: one underdeveloped cub growing
in pouch similarly to other species of marsupials. The three-monthly cub starts
to get out of pouch, and at the age of four months it leaves mother’s pouch
finally. About one month the young animal studies at mother in searches of forage,
and then in courtship season she banishes the posterity. Young monkey wallabies
begin able to breed at two-year-old age.
Deer
wallaby (Neogale sylvanus)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroo (Macropodidae)
Habitat: forests in the north and east of Meganesia, undergrowth.

Picture by Alexey Tatarinov
Unlike large marsupials, which became extinct soon after people
arrival to Australia and New Guinea, the animals of small and medium sizes were
able to adapt successfully to neighbouring humans and introduced placental mammals
in Holocene. This trend expressed in many cases not only in Australia, but in
the fact that the majority of native mammals belonged to marsupials, that gave
a unique identity to the fauna made of descendants of survived species.
Among the marsupial survivors in the era of anthropogenic pressure there were
the relatives of larger kangaroos – brush-tailed rock-wallabies (Petrogale penicillata).
Their relatively flexible behavior, small size and wide range of habitats allowed
them to outlive not only humans, but also the climatic cataclysms that greatly
reduced the species diversity of the larger kangaroos. And the formation of
a single continent of Meganesia had opened new ecological niches for development.
Deer wallaby of Neocene is one of the descendants of rock-wallaby. This is quite
large animal, growing to sizes of red kangaroos of Holocene – 1-1.6 meters in
height (the females are smaller), but much more lightweight, not heavier than
40 kg. Furthermore, when compared with other bipedal marsupials, deer wallaby
has very gracile constitution; great part of its body seems to consist of highly
elongated hind legs and tail, due to it deer wallaby, it seems, walks “on stilts”.
Despite the long legs, this animal is not very fast and gets tired very quickly
during the chase – lung volume does not allow this animal to run for a long
time. Therefore, like placental deer and antelopes it has very large ears and
eyes, and is constantly on guard. At the slightest danger deer wallaby rushes
away and hides in forest thickets: it has spotted coloration like deer of Holocene
epoch – white spots on yellowish-brown background – and it is its main mean
of defence.
Deer wallabies feed mostly on foliage of trees and shrubs, as well as on berries,
flowers, low-growing epiphytes and other herbaceous plants. Their height and
the ability to jump permit them even to climb on low trees and to browse their
crowns, or to escape from predators. When climbing up a tree, deer wallaby makes
vertical jump along the trunk, and at the highest point of the jump it grabs
tree with its front paws. Then animal grabs tree with its hind legs and climbs
onwards, pushing the tail into the trunk.
Deer wallabies can mate every six months. Like with the other marsupials, the
pregnancy is very shortt and the youngs in the pouch also grow quickly. At one
year age they are already independent and do not depend on their mothers, and
may already have their own offspring. Young animals keep next to the older ones:
they live in small herds numbering of 6 to 10 individuals of both genders and
all ages. Males, however, usually leave their family upon reaching sexual maturity
and go look for a new life companion. These are phlegmatic animals and the fights
for females almost never happen. But while establishing relationships of domination,
the dominant male of herd may bite the subordinate males in the areas of the
scruff and shoulders. It also constantly displays its’ dominance in the group,
pushing the subordinate males away from females and food sources. Thus it forces
the competitors to leave the herd.
The life expectancy of deer wallaby is 15 – 20 years, but usually less than
that.
This species of mammals is discovered by Bhut, the forum
member.
Translated by Bhut.
Ostrich kangaroo (Theriostruthio unidactylus)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroos (Macropodidae)
Habitat: grasslands and semideserts of Meganesia.
Picture by Tim Morris
Right after disappearances of mankind Australia represented
the continent the most part of which had been occupied with deserts and poor
savanna. In Neocene
when Australia had merged with New Guinea to unite continent Meganesia, the
climate promoted the occurrence of productive savanna. This area is inhabited
by large flightless birds and
descendants of camels introduced to Australia in ancient historic time, similar
to giraffes and antelopes.
But representatives of indigenous Australian fauna, marsupial mammals, successfully
compete to
them. Among them the original species became ecological analogues of antelopes
and other quickly running herbivores had evolved.
The extreme degree of specialization to such habit of life is demonstrated
by huge descendants of medium-sized wallabies of Holocene epoch, representatives
of genus Theriostruthio (literally: “ostrich beast”) for which strongly expressed
gracile constitution and ability to high-speed moving on a long distance
are characteristic. These animals are a vivid example of convergence with
running
birds and fossil ostrich dinosaurs (Ornithomimidae).
Ostrich kangaroo is settled at the significant part of Meganesia where savannas
were stretched. This animal lives in big herds numbering up to 100 individuals.
These animals are constantly in movement, and continuously migrate, not staying
at one place for a long time. Only in rain season when the grass plentifully
grows, animals do not hurry up to leave rendered habitable places. It is
very large species of marsupials – growth of the adult individual standing
on rear
legs reaches 3 meters, and the length including the tail is up to 4,5 meters.
Ostrich kangaroos strongly differ from species known in human epoch. At them
there are very long and rather thin extremities. On hind legs of these animals
there is only one middle toe with wide blunt claw similar to horse hoof.
It is an attribute of quickly moving animal: usual speed of ostrich kangaroo
makes
45 – 50 kms per hour, and animals pursued by predator accelerate momentum
up to 80 - 90 kms per hour. These animals perfectly move on firm ground,
but are
compelled to avoid areas with soft and sandy ground.
Forepaws of this animal are approximately one third shorter, than back ones,
and on them only three fingers with thick dulled claws remained. They are
mobile enough to permit the animal to make some actions with their help.
With the
help of these fingers the female of ostrich kangaroo clears the pouch before
the cub birth. Also ostrich kangaroos are able to break off tops of grasses
by forepaws and to eat them, holding body in vertical position. Such pose
is very characteristic for these animals: from height of the growth ostrich
kangaroos
easily notice predators.
Neck of this animal is longer and more mobile, than at usual kangaroos. If
grass is short and thin, ostrich kangaroos can graze, moving on four limbs
by slow “gallop” characteristic for kangaroos (ostrich kangaroos are not
able to walk). Thus they graze grass by mouth.
The diet of ostrich kangaroos includes rigid and dry grasses, and also plants
which can be inedible for the camelopes. In connection with such food jaws
of ostrich kangaroos are short (their muzzle is similar to muzzle of large
fossil kangaroo Procoptodon), and teeth have folded enamel and are adapted
for masticating of highly abrasive food. Besides they accrue for rather long
time, before their roots will be formed finally. It helps to compensate deterioration
as a result of feeding on rigid vegetative food. Muzzle of ostrich kangaroos
is wide and short, with strong chewing muscles. Its outlines are especially
emphasized by large auricles. Such auricles are necessary for more effective
heat emitting – ostrich kangaroos inhabit hot and dry areas of Meganesia.
They can not drink for a long time, being content with metabolic water which
turns
out at oxidation of food and fat.
Wool of ostrich kangaroos is short and velvety, colored in light shades –
at different individuals it varies from yellow up to almost snow-white and
light
grey. Light color of wool rescues animals from overheating – in places of
their inhabiting it is almost impossible to find trees giving enough shadow.
Each year at the female of ostrich kangaroo only one cub is born. It comes
into the world small and underdeveloped, and independently creeps to the
pouch of mother. In pouch it attaches by mouth to the nipple and within several
months
almost continuously sucks milk. The eight-monthly cub ventures to leave the
pouch for the first time, but at first attribute of danger it hides in it
again though it is rather difficult for it to make it with its long legs.
Gradually
young animal starts to explore the world around, and passes to adult diet.
At the age of fourteen months the young animal finally abandons the parent
pouch and does not suck milk any more. The three-year animal already almost
grows till the size of adult individuals, and at the age of five years ostrich
kangaroos become able to breeding. Life expectancy of this animal reaches
40 years and more.
In territory of Meganesia more some species of ostrich kangaroos still:
Dwarf ostrich kangaroo (Theriostruthio nanus) lives
in the strip of dry savannas and semideserts to the west of Eyre Gulf. Its
length does not exceed 150 cm
including the tail, and growth of sitting animal is no higher than one meter.
On the body of this animal there is a pale pattern of dense vertical strips
which pass to more contrast cross-striped pattern on tail and rear legs.
Such colouring of wool appears fine masking at life in high grass. This species
differs in high speed – at the short distances dwarf ostrich kangaroo can
reach
the speed of more than 80 kms per hour. The female of this species can bring
posterity two times per one year – in spring and autumn. This animal grazes
on four legs only in thin and short grass. Usually dwarf ostrich kangaroos
eat, biting off tops of high grasses during the movement.
Striped ostrich kangaroo (Theriostruthio zebratus) lives
in rather damp district – in bushy savanna at the western coast of Eyre Gulf.
It differs from common
ostrich kangaroo in considerably smaller size (length of body is up to 3
meters) and darker colouring. On yellowish-brown skin of this animal brown
vertical
strips stretch. Due to such colouring grazed animals are poorly appreciable
among grass and in light forest. These animals have also special feature
of behaviour – during movement each animal from time to time makes almost
vertical
jumps of height up to 4 meters.
Forest ostrich kangaroo (Theriostruthio sylvestris) has
the same size, as the previous species. It lives in light forests growing
in river valleys at the
east of Meganesia. Jaws of this animal are weaker and lengthened more, than
at common ostrich kangaroo. It is connected to a diet of this animal – it
eats leaves of undersized trees and bushes, and also large grasses. The body
of
this kangaroo has spotty colouring – reddish-brown with vertical white strips
on back and shoulders. Also the throat and bottom part of head have white
colouring. Ears of this animal are rounded and have smaller size, rather
than at species
living in plain district.
Mirriuula (Diablowallabia camelophoneus)
Order: Diprotodontia (Diprotodontia)
Family: Kangaroos (Macropodidae)
Habitat: Meganesia, mountains in the east, to the south up to Flinders Range;
forests and bush.

Picture by Carlos Pizcueta (Electreel)
The majority of kangaroo species represents herbivores of plains, bush and
mountains. But it is not only ecological niche which these animals could occupy.
The species of carnivorous kangaroo rats Ekaltadeta ima weight about 15 kgs,
and also one more predatory kangaroo, Propleopus oscillans lived in the past
in territory of Australia. In human epoch some wallabies differed in propensity
to zoophagy. After extinction of placentary predators because of epizooties
various species of marsupials occupied an ecological niche of carnivores during
Neocene with different degrees of success. The small naked-breasted kangaroo,
scavenger kangaroo rat (Harpotorous
microraptor), lives in semideserts of Meganesia,
eating carrion and the rests of prey of large
predatory reptiles. The result
of other attempt of kangaroos to develop predatoriness is mirriuula, the descendant
of mountain wallaby (Petrogale) living in mountain areas of Meganesia.
Mirriuula is named after mythical demonic dog from folklore of Australian aborigenes,
and quite justifies this name. It is a furious kangaroo weighting about 100
kgs, with flexible torso and long fluffy tail serving for balance. It is a
predator eating birds, herbivorous kangaroos and mountain
camelopes.
Mirriuula is not too similar to usual kangaroos by its constitution. It has
rather short hind legs, and rather seldom rears on them. The mode of its movement
is galloping jumping and true gallop. Mirriuula moves only on hind legs very
seldom – usually it does it, attacking prey. At this time its forepaws are
free, and mirriuula seizes prey by them. Mirriuula can run quickly both on
short, and on long distances, but on open district or in foothills it frequently
conflicts with other marsupial predators. But in mountains this species is
out of competition. It gets big advantage in hilly terrain over marsupial jaguar
and marsupial panther, surpassing them in dexterity. Besides these predators
are more heat-loving and prefer not to come into mountains. And mirriuula is
able to chase prey on hillsides; it arranges ambushes on tracks and frequently
hunts in mountain forests and among thickets of undersized eucalypti. Colouring
of wool of the mirriuula helps it to remain unnoticed. The basic background
of colouring is brownish-grey, darker on head and paws. Tail is light grey,
and its tip is almost white. On back of animal there are some longitudinal
lines of dark spots.
Having long and strong limbs and hard bent claws, mirriuula can easily catch
up and kill the goat-sized prey. It snatches on prey and brings it down on
the ground with the help of strong forepaws, and then tears its throat by long
pointed incisors slightly directed forward and closed, as a nipper. With the
help of incisors this predator can easily bite through neck of prey. In strong
jaws cutting molars with jagged edge and peaked molars easily cracking bones
and tearing meat from them grow.
The muzzle of animal resembles cat's one a little – it is short and high, with
large mobile ears. In nasal cavity of animal the olfactory epithelium forms
many plicas; therefore sense of smell of this animal is very keen.
This is solitary animal strictly protecting its territory. At the territory
of mirriuula there are some shelters, one of which is used constantly, and
others are temporary; animal spends night in them, when eats large prey left
near them.
At the territory of Meganesia smaller relative of mirriuula lives – fox
wallaby (Diablowallabia omnivorodentus). This species inhabits plains overgrown with
high grass, where it builds shelter representing deep hole. It hunts or traps
in ambush small vertebrates, eats insects, digs out grubs, nutritious stalks
and tubers of plants. It can run very quickly – as quickly, as its large relative.
This animal has larger ears resembling ears of fennec fox – it is the adaptation
for heat emitting. The wool of fox wallaby is colored yellowish-grey with several
longitudinal lines of brown spots.
These species of animals were discovered by Tim Morris, Adelaide, Australia.
Bladdernose
Kangaroo (Rhinoroo desertus)
Order: Diprotodontia (Diprotodontia)
Family: Trunked Kangaroos (Nasalotheriidae)
Habitat: Desert of western Meganesia.

Picture by Alexander Smyslov
Australia is characterised in the age of man by arid deserts,
this was not always typical of Australia. In the Neocene, Meganesia has parts
that are similarly desrtified, and creatures living there have unique adaptations.
The Bladdernose Kangaroo is a very distinctive form, seemingly comical in appearance,
with swollen red nose.
This kangaroo is small at 20-25 Kilograms, and reaching a height of 70 centimeters
at the hips. It’s sleek fur is a dusty cream colour with white underparts, this
helps to keep the animal from overheating. Certain areas of high bloodlflow,
thighs, and chest are bald and pinkish for heat dissipation, which can be licked
to aid cooling. It has powerful forelimbs that are long, with large hands for
grasping food, and its hindlimbs are about half again as long, with long feet.
This kangaroo typically moves on all fours when foraging, but is able to hop
at high speeds to escape danger. It has a short-compact torso, and a short neck
ending in a large head, and a long tail.
The head is short and blocky, and the snout is a large inflatable sac that is
round and swollen (by this feature it resembles saiga or hooded seal). This
sac is infused with blood-vessels, and helps to cool the animal as it breathes,
it can be flushed with blood, as can other bald spots, to help the animal cool.
This sac is also a resonating chamber for producing loud calls, and can be inflated
during the mating display. Call is typically short and loud nasal bark, but
breeding calls are very loud prolonged roar.
Bladdernose Kangaroo feeds on desert herbs, grasses and flowers, as well as
digging up roots and tubers and eating small desert fruits, it is completely
herbivorous.
Breeding season is always during high productivity and rainfall, normally in
the winter or spring. Males produce loud roaring calls to attract females, males
generally have their own territory in which to attract a harem. Females congregate
with males producing loudest call. During mating ritual, male inflates nasal
sack considerably, resmbling baloon. During this display the sack is bright
pink from circulation.
Female produces one cub that becomes mobile during the flush of spring growth,
and suckles for 3 months. Lifespan is typically 20-30 years.
This species of mammals was discovered by Tim Morris, Adelaide, Australia.
Forest Probo (Probosciroo sylvanus)
Order: Diprotodontia (Diprotodontia)
Family: Trunked Kangaroos (Nasalotheriidae)
Habitat: Rainforests and Woods of northern Meganesia
During the age of man, kangaroos remained successful despite human influence.
With the extinction of mankind and the advent of the neocene, kangaroos competed
successfully with the surviving introduced domestic herbivores. Camels produced
numerous forms, and pigs became more specialised towards herbivory. Eventually,
kangaroos produced omnivorous and carnivorous forms, which competed sucessfully
with their placental counterparts.
The Forest Probo is descended from small omnivorous rat-kangaroos of the human
age, such creatures were successful foragers. This animal has become much larger
than it’s ancestor, becoming as heavy as a medium sized pig, at 40-60 kilograms.
It is 1.4 meters tall at the hips, and is usually bent over close to the ground
to forage. It has a thickset body and a long tail; colored blackish brown with
creamy underparts and a white dorsal stripe, fur is coarse generally. The forelimbs
are long and powerfully built with large claws for digging, hind legs are only
slightly longer with pointed claws, it moves mainly on all fours, only hopping
when it needs to move quickly.
Its head is long-snouted, with a large thick horny pad on the upper side for
digging with, its ears are long. The nose terminates in a moveable proboscis
20 centimetres long that has a flattened knob at its tip; this knob is very
sensitive and can be used to pass food to the mouth.
This creature constantly forages in the leaf litter, moving earth with its
snout similarly to a pig, but with the addition of a manipulative trunk.
When heavily pushing earth with it's nasal pad, it tucks it's trunk close to
its mouth, however, the trunk can also move litter and overturn small stones.
More extensive digging can be done with the claws.
The diet of this animal is as varied as that of a swine, fruit, roots, seeds,
buds and shoots are consumed, and the animal voraciously raids nests of ground
birds. Also small vertebrates, any insect or grub, and some carrion. It’s teeth
have large cusps for crushing, and the premolars have a large shearing edge
for cutting.
Animal vocalises with nasal squeals and coughs produced by trunk.
It is generally solitary, but mates all year long, to take advantage of any
fortuitous encounters with the opposite sex. The female has a distinct musky
aroma to alert potential suitors. The mating ritual consists of mock fighting
and kicking up wood litter by paws. Female produces one cub, which crawls from
the cloaca to the pouch. Babies leave the pouch by 3 months, but still use
it for safety, and weans by 8 months, during which a new cub will be developing.
Young are colored more pale than their parents, but the colors become dark
by one year of age. Lifespan is generally 25-30 years.
Similar relative, the Bush Probo (Probosciroo porcinus) is
found in the forest and bush of southeastern Meganesia, including the mountain
forests of the Great
Dividing Range and the forested Murray River valleys. Differs mainly by longer
legs, and lighter, dark grey coloration, the proboscis is shorter and possesses
a square tip. Diet is more tolerant to dry, tough forage such as twigs.
This species of mammals was discovered by Tim Morris, Adelaide, Australia.
Long Snouted Kangaroo (Dolichoops nasalis)
Order: Diprotodontia (Diprotodontia)
Family: Trunked Kangaroos (Nasalotheriidae)
Habitat: savannah and semidesert of central Meganesia
Occasionally during evolution in any animal group, new features distinguish
an animal group, becoming a new taxonomic grouping. The Trunked kangaroo group
has produced many different forms during the neocene. Evolution has been versatile,
with large omnivorous forms and also numerous herbivores. Example is the long-snouted
kangaroo.
This kangaroo is moderate in size, being 1 meter tall and weighing 30 kilograms.
This animal is sandy colored with soft, smooth fur, dusty white underparts
and a black dorsal stripe, the hands, feet and face are pale. Its forelimbs
are long and powerful, with bald patches along the inside which it licks to
keep cool; the paws are large and have long claws for digging. The tail is
long and its hindlimbs are of medium length, with long feet.
Ears are large, almost like a rabbit, and the head is long and low, recalling
a horse.
There is a well developed muscular upper lip and nose which forms a swelling,
this is very flexible and comes to a rounded tip, like a horse. The nasal cavity
is enlarged, and the nostrils can be closed.
This animal feeds on roots, tubers, some grasses, herbs, and soft foliage,
but will also eat insects. It forages bent over, close to the ground, moving
generally by a slow bounding motion on all fours. It stands up periodically
to sense any danger, but large ears and sensitive nose constantly provide early
warning system while foraging. When forced to flee, it can either bound like
a rabbit, or at higher speeds hop very quickly. These animals are generally
found in small groups of 2 or three, which grow in number when breeding.
Mating occures in spring and winter, during higher producticity and rainfall.
Males will fight over females using clawed hands and kicking feet. Females
produce one cub which comes about in the typical marsupial fashion. Able to
leave the pouch at 4 months, but will continue to use it as refuge, and to
suckle, until 1 year, when another cub will have started to grow and suckle
on the second teat.
Vocalisations are nasal cough or loud resonant sneeze. Lifespan is typically
30-35 years.
This species of mammals was discovered by Tim Morris, Adelaide, Australia.
Elephant-Kangaroo
(Marsupielephas proboscoides)
Order: Diprotodontia (Diprotodontia)
Family: Trunk Kangaroos (Nasalotheriidae)
Habitat: tropical rainforest and gallery forest of Northern Meganesia, occasionally
ranges into bushland or treed savannah.
Picture by Amplion
Evolution is characterized during stages of disaster by a
rapid increase in size in a recovering ecosystem. Primitive hoofed animals quickly
became as large as bears after the extinction of the dinosaurs, reptiles became
as large as crocodiles quickly after the Permian extinction, and modern animals
quickly became larger in the abscence of competitors, see the feral cats of
anthropocene Australia. And thus, the evolution of the trunk kangaroos in the
early Neocene is characterized by the Elephant Kangaroo. Most trunked kangaroos
are relatively small or medium-sized, but the bountiful jungle of northern Meganesia
provided an opportunity for a giant of the group.
Unlike its relatives, it is a leaf eater, reaching high into the trees to gather
food. The teeth are high crowned and ridged, like those of a tapir or the extinct
deinothere. 80 percent of its diet is leaves and bark, 10 percent is fruit,
and the other 10 percent is rare animal forage. It will still eat animal matter
on occasion like its ancestors. Still having a slicing premolar, it will rarely
consume the eggs of ground birds, eat large beetles or stick insects, and scavenge
flesh from the kills of marsupial panthers and the yagil. It has been known
to eat ejected pouch young from the ground.
This creature by far surpasses any present kangaroo in size and stature, weighing
up to 800 kilograms. It is no longer able to hop, it can only bound slowly and
clumsily, recalling dinosaur. Its head is of medium length, deep and massive,
with a relatively long snout, ending in a trunk that is 1.3 meters long. Its
pelage is made of short, woolly hair. Its color recalls the Malayan tapir, with
a large white saddle on a black background, it also a white facial “mask” and
a white dorsal stripe, characteristic of its group, with white “socks” on hands
and feet. Its arms are somewhat like that of a ground sloth, only less thick-boned,
the claws are very long for pulling down branches, the longest claw is 20cm
long. It has a fairly long, barrel chest and a short “humped” neck with thick
shoulders. With wide hips and massive legs it can carry large, well developed
joeys, its thigh is still relatively long, but its foot is short and flat, with
massive “hooved” toes. The tail is fairly long and thick, with a white tip.
By many features it is analogous the the long extinct Palorchestes, including
a long, elephant-like trunk. It stands 2 meters tall at rest, which is bent
forward. At a rearing height, it reaches 4 meters from the head to the ground,
but due to its proboscis it can reach even higher. The trunk ends in a “finger”,
not unlike those of the elephant of human era.
The mating season falls at the start of monsoon season, when they migrate to
upland areas or clearings to court. The males make characteristic braying and
trumpeting noises, accompanied by deep grunts and clicks, to attract a harem
of females. If a dispute breaks out between males, it is settled by displays
of physical strength, pulling down branches and biting them apart, throwing
them away. Males do not risk physical harm due to their long sensitive noses.
Babies are born at the end of the monsoon season, allowing enough time to develop
into large joeys the size of small children. They defend their young viciously,
lashing out with the forelimbs and biting, as they will also defend themselves
alone using this method. The legs are too degenerate for large kicks.
Lifespan can reach 20-40 years, large adults have no predators except in times
of drought when marsupial panther
can hunt cooperatively.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Nagual (Tatu nagual)
Order: Edentates (Edentata)
Family: Armadilloes (Dasypodidae)
Habitat: North America, the south of prairies and the north of Mexican Desert.

Picture by Simon, colorization by Biolog
Initial picture by Simon |
Nine-banded armadillo (Dasypus novemcinctus) alongside with
opossum (Didelphis) and American porcupines (Erethizon) was one of the most
successful descendants of South American fauna settled to the north in time
of “Great Interchange” in Plio-Pleistocene. In human epoch this species had
overcome Mexican plateau and was settled rather far to the north, having successfully
mastered the south of the United States of America. During the ice age at the
boundary of Holocene and Neocene armadilloes among other thermophilic animals
had receded to the south, but with the subsequent warming they have returned
to former positions again.
Nagual, the large species of armadilloes, lives at the southern border of prairies
where the grassy vegetation is gradually replaced by water-stocking succulents
and deciduous prickly bushes. It is large enough representative of family: its
length is about 130 cm, and weight reaches 100 kg and more. The big size of
animal is one of ways of survival in desert: the large animal has the big stocks
of water in organism, and the strong armour protects not only from enemies,
but also from the hot sun. At the north of the area nagual leads a day time
habit of life, but at the south, in deserts of Mexico, activity is displaced
to night time. It is also a way to avoid overheat and losses of water.
The armour of animal consists of two strong plates – forward and back ones –
and between them some belts of cornificated skin with elastic sites between
them are located. Under armour there is a layer of fat which is the heat insulator.
As against the ancestor, nagual is not able to turn off in ball. Being attacked
by predator, animal simply lays on the ground, having turned up under armour
head and thick short tail. Because of large weight it is difficult to turn it,
but if predator manages to do it, animal protects itself, sharply waving forepaws
armed with sharp claws. The bottom part of animal body is covered with rough
yellowish wool. Armour is colored bright yellow with grayish shade.
The head of animal is covered with strong corneous plate with skin ossifications
which jut out on plate surface like rounded outgrowths. Eyes are small – nagual
has bad sense of sight. Animal has mobile rounded ears which are penetrated
with blood vessels and partly help to radiate the surplus of heat. Muzzle of
nagual is short and triangular; teeth are numerous and have sharp cutting edge.
They are closed in such a manner that edges of upper and lower teeth are constantly
erased against each other. On teeth of upper jaw cutting edge is external, on
lower jaw internal. Despite of size and slowness nagual is zoophagous species.
It willingly eats carrion searching it by smell. Nagual is frequently fed with
the rests of prey of local
predators. Due to massive armour this animal does not pay attention to attempts
of predator to protect the prey. Furthermore nagual eats medium-sized vertebrates
and occasionally feeds on underground and sapful parts of plants, especially
during the drought season.
Forepaws of nagual are perfectly adapted to digging. The third and fourth fingers
are armed with huge claws – about 7 cm thick and up to 18 – 20 cm long. On scapulae
ledges and crests to which strong muscles attach are advanced. Nagual easily
moves the stones comparable in weight to the animal and can dig and throw out
on ground surface up to half of cubic metre of the ground in some minutes. Defending
itself against enemies, the animal drops to the ground and sticks claws into
it deeply. In that case it is very difficult to turn nagual upside down. But
if the predator had made it, the animal defends, making sharp attacks by claws.
If it succeeds to grasp a predator, this armadillo seizes it in death grip and
can easily strangle it or break its ribs. Therefore, when nagual appears near
prey, lucky hunters prefer not to pay attention to this one.
The sexual dimorphism at this species is not expressed. Pregnancy is long because
of latent stage: it proceeds till about 11 months, and cubs always are born
in favorable season. Up to 10 cubs may be born; they have the same gender because
develop from one initial ovocyte. Newborn armadilloes have opened eyes and are
covered with soft leathery armour. In some hours after birth they are already
capable to walk, and in same time the armour hardens quickly. Young animals
remain with mother within 8 months. They reach sexual maturity in 4 years.
The idea about existence of this species of mammals is proposed by Nick, the forum member.
Aquatic armadillo (Hydrotatu potamophilus)
Order: Edentates (Edentata)
Family: Armadilloes (Dasypodidae)
Habitat: southern coast of North America: Florida, deltas of rivers at the
coast of gulf of Mexico.

Picture by Alexander Smyslov
Armadilloes were one of groups of South American mammals managed to overcome
isthmus of Panama and colonized North America successfully. In human epoch
nine-banded armadillo (Dasypus novemcinctus) had moved far to the north,
having overcome Mexican plateau, and had settled in southern extremities
of North
American prairies. In ice age armadilloes had been thrusted back far to the
south, and along coast of gulf of Mexico only tiny relic populations of these
animals remained. In such populations speciation and accumulation of new
features passed roughly, and at the ending of ice age the southern part
of North America
had been inhabited with some species of armadilloes differing in shape and
in habit of life. Some of them had died out subsequently, and others managed
to fix in habitats for a long time, and their descendants make a part of
Neocene fauna of North America.
In tropical areas of North America where various freshwater reservoirs are
present, one species of armadillos lives, which had mastered a habit of life
unusual to these animals – the aquatic armadillo. This animal is about half
meter long including tail. By its shape it differs from other armadilloes
only a little – its body on back and sides is covered with armour divided
to thoracal
and lumbar shields. Between them some (usually 6 to 8) narrow belts lay,
permitting a body of animal to be bent. The armour of this animal, however,
is much thinner,
rather than at ground species. Tail of aquatic armadillo is of oval outlines,
short and wide. Head from above is covered with corneous plate. Bottom part
of body is covered with thin rough wool. This animal has dark colouring –
reddish-brown with lighter edges of thoracal and lumbar shields.
Paws of aquatic armadillo are perfectly adapted to movement on fenny ground
– between fingers and toes there are skin crosspieces increasing the support
area and interfering with immersing of animal in marsh; also it helps in
swimming. On forepaws strong claws are advanced. They are rather thinner,
than at ground
species which should dig firm deserted ground. With the help of claws aquatic
armadillo digs from silt rhizomes and tubers of water plants making a part
of its diet and also digs holes in riverbanks and among roots of trees.
Ears of this animal are short and mobile. In the basis of ear there is a
skin valve which closes ear duct when animal dives. Aquatic armadillo orientates
itself under water freely with the help of sight – it has rather large eyes.
But in muddy water it orientates itself and searches for a forage by means
of touch: it also has mobile lengthened snout richly supplied with receptors.
By means of snout it finds out food of animal origin – worms, crayfishes,
snails
and larvae of insects. Animal and vegetative food in its diet is present
approximately fifty-fifty.
Aquatic armadillo is active mainly in twilight, in the morning and in the
evening. In hottest time of day it falls asleep in the hole dug out in a
place where
danger to be flooded at rising of water lavel to be minimal. This is very
mobile animal able to run fast in underbrush or among thickets of reed in
case of
need. It is good swimmer due to the layer of hypodermic fat which serves
as a float, and dives without any efforts due to rather big density. This
armadillo
can stay under water till about five minutes.
Breeding of this mammal is not dated for the certain season – the posterity
of this species may be observed in nature the year round, and the female
can bring posterity up to two times for one year. After the pregnancy continuing
about three months, the female gives rise to from four up to six cubs – they
always are enzygotic twins. They are advanced well enough and at one week
age
they do not concede to adult individuals in swimming ability. Young animals
can only dive no longer than for one minute. They remain with mother till
about five weeks and then start to lead independent life. Because of too
short intestines
the young growth eats food of animal origin mainly. At the age of 16 months
young animals become sexually mature; life expectancy is about 10 – 12 years.
Digging armadillo (Postpichisiego trachycephalus)
Order: Edentates (Edentata)
Family: Digging armadilloes (Armotalpidae)
Habitat: South America, pampas
and light forests from warm temperate up to tropical zone.

Picture by Alexander Smyslov
In human epoch in South America mammals constantly leading
digging habit of life (the analogues of moles of Holarctic region) were absent.
At the same time in territory of this continent there were many kinds of burrowing
rodents and armadilloes. Fairy armadillo (Chlamydophorus truncatus), or pichisiego
died out in human epoch had led the way of life closest to moles.
In Neocene of South America armadilloes had taken a significant place in fauna.
They successfully compete to rodents, and one species became a
large scavenger. Among Neocene armadilloes one species which conducts
practically completely underground way of life had evolved. This small animal
is named digging armadillo.
Digging armadillo is deeply adapted to a underground habit of life. The body
of animal is about 25 cm long. It has almost cylindrical shape and is covered
with strong armour. In the middle of trunk the armour of animal forms five belts
connected by elastic skin. These belts allow digging armadillo to turn off in
ball and to turn round in its own tunnels. Tail of this animal is short and
wide in the basis and has triangular outlines. Edges of corneous belts of tail
form thick strong prickles. Resting them in floor and walls of hole, animal
provides a reliable support at digging to itself.
At this animal there are short limbs. Hinder legs of digging armadillo are rather
weak and three-toed. Forepaws are wide and muscled, armed with three huge claws
(the largest claw on middle finger reaches length of 4 cm, claws on third finger
and forefinger are shorter). During the digging animal supports on prickles
on edges of tail, and rakes ground out by claws of forepaws. Friable ground
is raked back by hind legs. When the rather large amount of it is gathered,
digging armadillo turns tail under stomach, and moves back, pushing ground to
the exit of hole. On the ground surface this animal is very cautious, and the
predator practically can not creep to it imperceptibly. Digging armadillo feels
keenly vibrations of ground, and feels approaching of large animals how they
will see it.
Digging armadillo is practically blind. Its eyes are shifted in the bottom part
of head and are very small. In them there is no crystalline lens, and the animal
can distinguish light from darkness only. The head of digging armadillo is covered
with thick corneous plate in which basis thick skull bones lay. Ears of animal
are very short, protected from ground by rigid bristles. The forepart of muzzle
is protected by “visor” of corneous plate, and nostrils are supplied with muscles
permitting them to close. At digging of friable and dry ground animal can hold
the breath till 1-2 minutes. Due to such features of anatomy the animal is able
not only to dig tunnels but also to swim. Digging armadillo can successfully
swim across small rivers, being guided on smell of air above water. During the
swimming it rows by forepaws, and turns rear legs in sides, and they serve for
balance.
Digging armadillo eats various ground invertebrates, but the vegetative food
– roots and tubers – makes about 20 % of its diet. This animal prefers to settle
in areas with small amount of wood vegetation. Digging armadillo often builds
the common systems of holes with some local rodents, but such neighbourhood
not always passes without conflicts – digging armadilloes willingly eat newborn
rodents.
In southern regions of area this animal runs into not deep winter dormation,
does not dig new tunnels and does not leave on ground surface.
In the beginning of spring at digging armadilloes the courtship season begins.
Male is larger, than female, and behaves more aggressively. It searches in tunnels
of females ready to breeding, pairs with them and does not accept participation
in care of posterity any more. In 7 weeks after pairing in deep hole the female
gives rise to posterity – 4 cubs which always are enzygotic twins. While they
are small, the female often comes back to feed them, but since the third week
of life cubs can follow freely the female in tunnels and gradually pass to diet
of adult animals. They become sexually mature at the age of half-year.
Aotearoan
Ridgeback Hedgehog (Notoechinus vorax)
Order: Insectivores (Insectivora)
Family: Hedgehogs (Erinaceidae)
Habitat: North Island of New Zealand, woods, scrub and sub-tropical forest.
Species introduced by Englishmen to acclimatize themselves to the country of
New Zealand were very varied, including many European species, among them, the
Common Hedgehog (Erinaceus europaeus). This species leaves various
insectivorous descendants in Neocene New Zealand, but one form differs in
being more generalized.
Being found in the forests of the North Island, this hedgehog is relatively
large, about 45 centimetres long, its ability to curl up has reduced, and instead
it has a distinct ridge of short, very sharp spines down its back and along
its flanks. Reason for loss of defensive posture is also evident by this animals
large head, relatively long with sharp front teeth. Front claws are relatively
long and straight, hind and fore legs are longer than its ancestor, giving it
an upright shape more resembling an oversized, tail-less rat. Coloration can
vary from a straw-brown colour to a reddish brown, but always with darker spots,
each spine is yellow with a black tip, nose and feet are black.
It is an omnivore with some preference for animal food, large insects, worms,
slugs and snails, as well as small rodents, frogs, lizards and young rabbits,
and the eggs of ground-nesting birds. Vegetable food includes fallen fruit,
large seeds and nuts, as well as softer root vegetables.
It is mostly solitary, being active during twilight and early evening. Sense
of smell and hearing is very keen, and it is by these that it usually finds
its food, eyesight is relatively poor. When attacked by a predator, it will
arch its back and produce a yarring chirr; if this fails it will bite fiercely,
or flee. This animal breeds in the summer, from November until mid January,
males will compete for the attention of females and follow them around ardently
until they submit to an overture of nuzzling, and males may mate with multiple
females. Mating is brief, but can occur multiple times. The female raises the
litter alone, which generally numbers 4 or 5; the young are born blind and only
grow their prickles later. Weaning occurs at about 6 weeks, sexual maturity
is reached at one year, and lifespan is up to 10 years but usually less.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Azorean
tree hedgehog (Dendrogalerix scandens)
Order: Hedgehogs (Erinaceomorpha)
Family: Hedgehogs (Erinaceidae)
Habitat: New Azora, lowland and mountain woods.

Picture by Alexander Smyslov
Usually island ecosystems differ in poverty in comparison with continental
ones. Few species can get on islands and survive in conditions of isolation.
Island ecosystems differ in vulnerability, and strongly suffer from introduction
of new species. Newly appeared species change a course of evolution of other
inhabitants of ecosystem because actually they are the new factor of environment
for native ones. But the introduced species change not less, than others,
having got in new conditions.
On New Azora Island the majority of mammalian species is descendants of species
introduced by people. The special species of insectivores, the Azorean tree
hedgehog – belongs to them. It is tree-climbing species of animals, the descendant
of European hedgehog (Erinaceus europaeus) introduced in historical epoch.
Earlier unite species had given two branches of descendants. The massive
line of ground animals is presented by large azogalerix,
and small descendants of European hedgehog turned to tree-climbing species.
Change of way of life had resulted in strongly expressed difference of new
species from ancestor in constitution and behaviour. Azorean tree hedgehog
is not able to turn in ball, and the hypodermic muscles characteristic for
ancestor became very weak at it. The body of this animal is still covered
with the pointed spikes serving for self-defense. In case of danger the animal
nestles
against bark of tree and exposes towards to the enemy spikes which can rise
and fall with the help of special muscles in the basis of each spike.
In connection with tree-climbing habit of life at this animal there are relatively
larger paws, than at ground hedgehog. Azorean tree hedgehog dexterously and
quickly swarms up trees, and confidently keeps even on vertical surface of
tree trunk. Fingers at Azorean tree hedgehog are more mobile; with their
help animal can cling even to thin twigs. Besides this mammal has one more
adaptation
permitting to keep on trees better. On the back side of paws at this hedgehog
especially strong and thick, slightly bent spikes grow in line. With their
help the hedgehog can cling to bark, swarming up tree trunk and branches.
Head at this animal is extended and flattened. The muzzle is extended in
mobile proboscis with which help animal searches for forage. Ears at Azorean
tree
hedgehog are wide and mobile – it is necessary not only for search of prey,
but also for more effective heat exchange in conditions of hot climate. At
animal there is keen hearing due to which this hedgehog can hear noise of
insect creeping on bark in night darkness. At this animal there is good night
sight,
but Azorean tree hedgehog distinguishes colors badly. The brain at this animal
is small, and behaviour differs in primitiveness.
Azorean tree hedgehog occupies an ecological niche of small prosimians in
ecosystem of this island. This is a solitary animal eating mainly small animals
– insects,
tree-climbing land crabs and nestlings of various birds. Due to ability to
climb on trees this animal can attack colonies of fog
swifts settling in cavities
of trunks of large trees. Also Azorean tree hedgehog eats mushrooms and juicy
soft fruits. Among mushrooms the animal chooses the most worm-eaten ones,
and eats them along with larvae of insects settling in them. The poison accumulating
in some mushrooms does not harm to it – at Azorean tree hedgehog there is
strong
immunity to poisons, inherited from ancestor.
Each individual occupies the certain territory which is marked by odorous
secretions. Out of breeding season there are no constant shelters in territory
of this
little mammal. These animals meet only in period of pairing, and in another
time protect the territory from relatives. The posterity at these hedgehogs
is born two – three times per one year. In litter it may be up to 5 – 6 small,
blind and helpless cubs. Before their birth the female searches for shelter
in which it will raise cubs. Usually for this purpose it occupies hollows
or old bird nests. If necessary the female can expel medium-sized birds from
liked
nests: it eats their laying or nestlings, and occupies the empty nest.
Young animals abandon nest at the age of about two months. At this time they
already see well and able to climb on branches. The litter keeps near to
the female till two next weeks, training in ways of getting of food. One-year-old
animals already can bring posterity. Life expectancy of this mammal does
not
exceed 6 years.
Tchagrin(Aepierinaceus tchagrin)
Order: Hedgehogs (Erinaceomorpha)
Family: Hedgehogs (Erinaceidae)
Habitat: Balkans, South-Eastern Europe, south coast of Fourseas, Caucasian
peninsula.

Picture by Alexander Smyslov
Early Neocene lacked various and diverse ecosystems; its’ ecosystems
lacked big herbivores and carnivores. So the situation was a bit like early
Paleocene
when the representatives of different animal groups were having their “tryouts”
of different environmental niches. That happened in Eurasia where nature was
once under the especially strong influence of mankind. The representative of
different species and orders were having their “tryouts’ for the ecological
niche of big predator. During a few millions of years true predators from the
Carnivora order had to co-exist with different predators representing insectivores
and primates. Finally they retook their rightful places in ecosystem which
were abandoned during the dominance of mankind, but some of the competitors
are still there. In the northern European forests spinywolf
(Erinalupus spinosus) lives, and the Mediterranean maquis is inhabited
by jackal hedgehog (Erinalupus mediterraneus). The subtropical belt of Europe-
Balcans, Asia Minor
and Caucasus-
are inhabited by another representative of the same family- huge hedgehog tchagrin.
That animal has more primitive features compared to its relatives. Tchagrin
is a stocky animal, reminding badger in proportions. Body length is around
140 cm. The visible tail is absent, the head is relatively big, with long movable
snout. The fur is grey with brown shades, lighter on the belly; around mouth
the fur is white. Tchagrin has strong jaws with sharp teeth of carnivorous
animal. But pursuing its prey isn’t one of strong sides of tchagrin: his limbs
are relatively short (longer than those of badger but shorter than those of
dog) and it’s plantigrade animal. So maximal speed of sprinting tchagrin is
around 30 km\h.
Tchagrin has poor eyesight- it’s myopic. But the animal has keen sences of
hearing and smell which help him to locate his prey and the sources of possible
danger.
From its ancestors the animal has inherited another specific trait- among its
fur on the back you can find 5 cm-long hard quills. Those cover upper part
of the head, neck, shoulders and the middle of the back along the spine. Tchagrin
lost the ability to roll itself up in the ball for protection cause he's too
large to do that. But the large size gives him the possibility of active defence.
While defending himself, tchagrin opens his mouth wide to demonstrate sharp
teeth, and actively tries to bite its opponent or attack him with claws. Rudimentary
muscles under skin allow the animal to lift the quills during the attack of
predators. That’s a solitary animal who is very aggressive towards its own
species outside of mating season. During the fight two tchagrins "box" with
their heads while quills are up. During the courting in the mating season male
pushes female to the bodyside with his head, quills are down.
The mating season of tchagrins starts early in the spring. The female, who
is ready for the coupling, has specific strong musk smell that attracts males.
One female can attract up to 5 males. They engage in strong rivalry, pushing
the rivals away from desired female with their heads. Female doesn't care about
her mate, so sometimes mating happens while the other males nearby are fighting.
The winner walks behind the female on stretched legs, sniffing loudly and pushing
the female with his head. Once the mating is over, the female sends males away
from her.
The pregnancy is about 5 weeks long. The female makes the nest for her cubs
from branches and ground. She makes a pile of the branches and digs something
akin to dugout under it. The dug earth is thrown over the branches to make
a cover from the rain and the wind. The brood (usually 3-5 cubs) is born naked
and blind. The eyes open at the age of a week, and at the age of 6 weeks they
leave the den together with their mother and start searching for the food with
her. The maturity is reached at the age of 3 years, the life span of the species
is 40-45 years.
Translated by Charles, the forum guest
Scaly hedgehog (Squamoechinus loricatus)
Order: Hedgehogs (Erinaceomorpha)
Family: Hedgehogs (Erinaceidae)
Habitat: Caucasian Peninsula, Asia Minor, Middle Asia to the east from Fourseas.

Picture by Alexander Smyslov
Small mammals had gone through epoch of anthropogenous pressure
much better, than large ones. In the majority they had kept biological variety
and had quickly
restored their number after human disappearance. In Neocene among them species
developed new features and strategies of survival had evolved.
One unusual species of insectivorous mammals lives in droughty areas in Western
Asia to the south of Fourseas – it is scaly hedgehog. Hedgehogs are rather
conservative group of mammals though occasionally among them freakish species
evolve like fossil Deinogalerix or Neocene spinywolf;
both species are large predators. But the majority of hedgehogs even in Neocene
are presented by small
animals eating insects and other invertebrates.
Scaly hedgehog is a small animal: the length of its body does not exceed
30 cm. Its constitution is typical for hedgehogs: massive trunk, short tail,
unspecialized
five-fingered and five-toed paws and large head on short neck. Living in
hot areas this hedgehog is covered with thin wool of bright yellow color.
Back,
sides and the top part of head of this animal are spiny – it has inherited
this feature from ancestors. But spikes have an unusual structure.
The part of spikes on sides and back is modified into wide scales which gradually
transform to the ordinarly looking spikes kept only in forward part of head.
Scales have light brown or straw colouring – it helps this animal to avoid
overheat. But tips of scales are colored black that unmasks this animal on
the background of landscape.
Scaly hedgehog is not afraid of predator attacks, but uses different protection
tactics depending on the kind of attacker. If the attacking predator belongs
to mammals, this hedgehog is curled off in ball. The scales skintight to
each other, forming strong enough armour, and their pointed tips do not allow
a
predator to unwrap scaly hedgehog by teeth or by paws. Being under attack
of reptiles swallowing prey entirely, scaly hedgehog, on the contrary, rises
on
paws and stretches scales in sides in order to seem larger. Thus black tips
of scales are well appreciable on background of light colouring of animal,
exaggerating its visual size. Thus hedgehog does not recede even if the reptile
touches it with tongue – having convinced that it cannot be bitten, reptile
does not continue the attack. Protecting itself against reptiles, this hedgehog
turns head to them and rotates on the spot if the predator tries to go and
to attack from side.
The diet of scaly hedgehog includes invertebrates (also poisonous ones),
small vertebrates and occasionally berries of plants from which it gets needed
moisture.
This animal is capable to live for a long time without water, getting moisture
from prey.
This species of animals is solitary one. Each hedgehog occupies the certain
territory from which it expels all strangers. Only a female raising cubs
has any constant shelter – usually it is the abandoned hole or crevice between
stones. At this species the posterity is born in spring. In a litter there
are 3 – 6 naked and blind cubs which grow spikes to the fifth day of life.
At the age of 4 weeks the part of needles on back and sides drops out, but
at their place at once scales start to grow. At six-week age young animals
leave mother and lead independent life. At the age of 1 year they reach maturity.
Life expectancy of scaly hedgehog does not exceed 10 years.
New Zealand hedgehog (Austroerinaceus myrmecophagus)
Order: Hedgehogs (Erinaceomorpha)
Family: Hedgehogs (Erinaceidae)
Habitat: New Zealand, forests of Northern Island in area of subtropical climate.

Picture by Alexander Smyslov
Before human arrival to New Zealand there were no mammals,
except for flying and marine ones. People had introduced to these islands a
plenty of mammals
of various groups which had started to evolve after human disappearance,
changing themselves and occupying new ecological niches. The descendant of
European
hedgehog introduced to New Zealand by people is New Zealand hedgehog, living
like ecological analogue of anteater. By its appearance this hedgehog as
if parodies the echidna of human epoch because of the muzzle extended to proboscis,
long tongue and the forepaws specialized for digging.
New Zealand hedgehog is rather large animal in standards of insectivores:
it grows up to 40 cm long and weighs about 800 grammes. All its back, back
part
of body, top half of sides and even forehead are covered with spikes, straighter
and shorter than at echidna. Fur of New Zealand hedgehog is short, but dense,
especially at individuals from southern parts of area. It is colored monotonous
gray-brown color, but sometimes there are also lighter colored individuals
and even partial albinos having white irregular-shaped spots scattered on
body.
Forepaws of New Zealand hedgehog are partly specialized for digging holes
of underground invertebrates and vertebrates, and resemble paws of American
ground
squirrels with long and powerful claws. Nevertheless, as against animals
like echidna and American anteaters, these claws are not bent and do not
prevent
this hedgehog to walk on the ground. Hinder legs of hedgehog are considerably
less specialized rather than forepaws, and are even a little bit longer.
It gives this hedgehog a little bit humpbacked appearance and prevents it
to curl
in ball similarly to its ancestors. But ability to curl in ball is not too
important any more for it as it can escape rather successfully from enemies,
having dug in ground like echidna. But longer rear legs and powerful back
permit New Zealand hedgehog to rise vertically, that help it to climb on
small tree
for ants or other invertebrates.
New Zealand hedgehogs live mainly on Northern Island of New Zealand plentifully
overgrown with forest. There these hedgehogs efficiently dig forest litter
in searches of ants, worms, bugs etc. pulling them out from underground holes
by long tongues covered with sticky saliva. Sometimes they creep in underground
tunnels of castle rabbits and can put some loss
to young rabbit cubs before leaving to the surface. New Zealand hedgehog
has still kept sharp enough and
strong molars suitable for eating of animal like newborn rabbit cub, but
adult rabbits can not be afraid of this hedgehog: they are too large and
strong for
this prickly animal. Nevertheless, they can make of nothing when hedgehog
got in their “fortress” because to answer their attacks hedgehog turns spikes
aside
the opponent and it cannot neither be pushed away nor bitten.
More mountainous Southern Island is less inhabited by these hedgehogs and
they live basically in lowlands, in bushy thickets along the rivers where
there
is lesser opportunity to meet ruacapangi –
large flightless bird of rallid group which can punch through this hedgehog
by long beak despite of its spikes,
break it off by claws and peck off all edible parts from prickly skin. Besides
there is larger amount of food for hedgehog in such places, rather than in
drier high-mountainous areas of island.
The peak of activity of New Zealand hedgehog falls to night time when it
walks across its territory and updates its borders with odorous marks. In
the afternoon
time hedgehogs sleep, having buried in the ground or in leaf layer except
the moments when they feel the rain coming. These animals do not like rain,
especially
individuals from Southern Island and if they feel approaching thunder-storm,
they immediately leave the shelters and search for places more protected
from a rain without dependence from time.
The courtship period at New Zealand hedgehogs takes place in drier season,
since May till July. At this time males actively court females, arranging
competitions during which they “box” against contenders by prickly forehead.
Approximately
5 weeks later females bring a litter of cubs, from 4 up to 6 ones in litter,
but at the north it may be up to 7 – 8 ones. Cubs are born almost helpless
and blind, but in one and a half month they turn quite independent, and at
the third month of life leave mother.
Usual life expectancy of New Zealand hedgehog is about 5 – 6 years.
This species of mammals was discovered by Bhut, the forum member.
Sabre-toothed
shrew (Smilosorex venator)
Order: Shrew-form (Soricomorpha)
Family: Shrews (Soricidae)
Habitat: forests of temperate and subtropical areas of Eastern Asia, Far East.

Picture by Alexander Smyslov
The mass extinction at the boundary of Holocene and Neocene freed multiple
ecological niches. In early Neocene a peculiar experiment began in nature: the
descendants of the surviving species started making attempts to develop new
life forms. In deciduous forests of East Asia one representative of the relicts
of early Neocenic insectivore radiation lives – the sabre-toothed shrew, a descendant
of the common shrew (Sorex araneus).
This species has become somewhat larger than most shrews, known in the human
epoch: the body length is 12-15 cm, the tail is 6.5-8 cm, and the skull size
is 30.5-34 mm. At this species sexual dimorphism is expressed: the female is
smaller than the male. Its ears are relatively small, the eyes are tiny. The
muzzle ends in a mobile proboscis, swollen at the base and enclosing large olfactory
chambers – the sense of smell at this species is very well developed and is
the main sense used when searching for prey. The build is solid, stocky; the
tail length is about half the length of the body. Hair on the tail is very sparse.
Like its ancestors, this is a carnivorous species. The specialization of its
teeth is noteworthy: the incisors and canines have diminished and grow much
closer to each other, and the first molars in the upper jaw have become larger
than the others and stick out of the mouth. They erupt much later than the other
teeth during the transition of the animal to adulthood. The base of the tooth
is reinforced by bone, while the skull is shortened compared to other shrews
and its front part is reduced. Molars, following the saber-like ones and growing
opposite of them in the lower jaw, are enlarged and have a cutting edge with
a thick layer of enamel. The jaws can open up very widely, and the bite force
is large enough to be able to bite through the cervical vertebrae of a rabbit-sized
animal.
Wool of animals of this species is brown, the belly and throat are colored lighter.
The sabre-toothed shrew is a solitary territorial species; when meeting outside
of the breeding season, these shrews growl, showing their teeth and trying to
exaggerate their size, pacing in front of an opponent on outstretched legs and
raising up hair on their backs.
This species feeds on relatively large prey: large invertebrates, mice, voles
and rat cubs. In the hunt for rodents, the animals are helped by their long
teeth; using them, the sabre-toothed shrew also can pierce the chitinous covers
of beetles. Perhaps the sabre tooth developed mainly as a weapon for hunting
for armored invertebrates. On occasion, this shrew attacks ground-dwelling birds,
far exceeding its size, and often emerges victorious. Killed bird may be enough
for it for feeding till some days, until the meat decays so much that it could
not eat it. The sabre-toothed shrew also does not disdain carrion, and sometimes
eats the remains of someone else’s prey. The relatively large size of this shrew
reduced its appetite, but because of its intense metabolism it eats per day
as much food as it weighs.
The breeding season lasts from May to September, the females manage to raise
2 litters of cubs. Males at this time are intolerant to each other and between
them combats take place, in which the teeth are never used – the opponents fight
and scratch each other with their paws. Pregnancy lasts for 3 weeks; the litter
size is up to 5 cubs. Cubs remain in a shelter for 15 – 20 days, and then they
keep near their mother for the same time. Brood follows the female in single
chain, holding on by their teeth to the base of the tail of that which is ahead,
the first one holding the tail of the mother. At the age of 50 days, females
are already able to reproduce. Life expectancy is about 2.5 years.
This species of mammals was discovered by Nick, the forum member
Translated by FanboyPhilosopher.
Wormtonguer
(Vermiglossorex formicivora)
Order: Shrew-form (Soricomorpha)
Family: Wormtonguers (Vermiglossotheriidae)
Habitat: Central and Southern Africa, savanna with small areas of wood
vegetation, wood in foothills.
Picture by Timothy Donald Morris
Colorization by Carlos Pizcueta
Initial image made by Pavel Volkov. |
When an environment had stabilized after Holocene accident , the climate of
Earth became considerably warmer and more humid. At animals the huge amount
of new fodder resources had appeared, number and species variability of some
animal groups had considerably increased. In warm Neocenic climate the area
of various social insects had considerably extended, and they became a fodder
resource for some species of animals representing new families, appeared
in Neocene.
The wormtonguer is African variant of anteater being the descendant of any
species of numerous African shrews (Soricidae). It is rather large representative
of order: cat-sized, but it seems even more largly because of features of
constitution. The head at animal is very long: it is the adaptation for feed
in nests of
social insects. The muzzle of shrews is usually extended to short flexible
proboscis. This feature has received the further development at their descendant:
the skull of animal is short and expanded in front, and increased nasal cartilages
serve as support for very long mobile proboscis (making two thirds of length
of head) with nostrils and mouth on the end. Nostrils have ring contracting
muscles – it is the adaptation for protection against insect stings. Being
feeding this mammal breathes by mouth in a step to tongue movements or holds
the breath at all. Tongue is very long - it may be extended forward for length
of proboscis of animal. The basis of tongue fastens to hypoglossal bone which
has moved far back, to middle of chest. In mouth salivary glands producing
sticky saliva thickening in air are well advanced. Teeth are original – incisors,
canines and premolars are not present in both jaws, only sharp-edged molars
have remained – two pairs in each jaw. With their help animal crushes caught
insects frayed then by corneous outgrowths of stomach walls. But at young
animals at which the proboscis is not advanced yet, among milk-teeth two
cutters in
each jaw develop nevertheless. Ears are short, they can fold across with
the help of special muscle. It protects them from stings of insects.
The animal spends practically all life on the ground, but if it is necessary
(for example, during flooding) it can dexterously climb on trees and even
swim. Usually the wormtonguer moves on the ground on four legs. Hinder legs
are plantigrade
with straight claws and non-joined toes. On forepaws there are sharp claws,
therefore animal at walking supports on lateral side of fingers (claws at
walking are turned by edges to each other). Tail is strong, with thick basis
where
the fat accumulates, assisting to go through fodder shortage. On waist of
animal there is special gland, secreting musk liquid. Colouring of wool of
animal
is not striking: head of males is dark-brown colored with white tip of proboscis,
back is grey with brown cross strips, and tail is black. At females head
is grey without white spots. Claws at animals of both sexes are sandy-yellow.
Wormtonguers are active in the afternoon: from dawn till midday and some
hours before sunset. At this time insects are not so active and not so strongly
bite,
or hasten to nest and are not so aggressive. Having found the nest of termites
or ants, animal rises on hinder legs, rests tail against the ground, and
digs out the house of insects by strong movements of forepaws. It licks off
running
outside insects by tongue. Besides the animal can dig out of the ground larvae
of beetles and gathers single insects from plants. Usually wormtonguers live
single life, zealously protecting fodder site from encroachments of neighbours.
Borders of this territory are diligently marked by sharply smelling musk
liquid. In territory at animal there are some constant shelters, in one of
which it
spends the night and will spend hottest time of day. Only in breeding season
borders of territories can be broken, and animals sometimes use this circumstance,
redistributing fodder areas. Usually one female and some males take part
in courtship ritual. Trying to impress the female, they walk on hinder legs,
show
claws and loudly snort. The female couples with strongest of applicant males
though it frequently prefers most strongly smelling of them.
Two - three times per one year the female gives rise to three – four cubs.
Newborn wormtonguers are naked, blind, with closed ears. At them there are
very short proboscises, therefore they can suck milk without difficulties.
The female hides them in specially dug hole and feeds with milk about three
weeks. At three-week age young animals leave(abandon) a hole and gradually
pass to an adult diet. About one week the female teaches them to find forage,
than one week the young growth keeps near to it for protection. Later young
animals pass to independent life, searching free territory for themselves.
Young animals are not so specialized, as adults: their diet includes much
more species of insects. When the proboscis starts to grow, the wormtonguer
passes
to eat extremely ants and termites. They become sexual mature at the second
year of life; life expectancy is about 10 - 12 years.
Enemies of wormtonguers are predatory birds and mammals, sometimes large
snakes and lizards attack them. The animal applies sharp claws to protect
against
enemies with which it inflicts to aggressor serious lacerations. Warning
about intention to defend, animal emits odorous liquid and rubs itself’s
back against
stones or tree trunks. Also, sharply exhaling air through mouth, it utters
loud sound – shrill whistle. Usually the predator has got from wormtonguer
deep wound, remembers its shout of threat and pungent smell for a long time.
Snailer
(Vermiglossorex malacovora)
Order: Shrew-form (Soricomorpha)
Family: Wormtonguers (Vermiglossotheriidae)
Habitat: Central and Southern Africa, flatland and mountain woods.

Picture by Alexander Smyslov
This mammal is similar to the previous species (and it is no wonder, they
are close relatives!), but it lives mainly in damp places – in tropical
woods and
thickets of bushes on river banks. The reason is, that the food of snailer
lives here - this animal eats basically snails of various species. For eating
of such food at snailer the special adaptation was developed: on the end
of its tongue horn thorns directed back have appeared. It permits to take
spineless
invertebrates from their refuges and shells. The muzzle of snailer in connection
with difference in diet became much shorter than at wormtonguer, and teeth
are advanced very poorly and submitted by one pair of molars (with very thin
layer of enamel) in each jaw.
As against to extremely ground-dwelling wormtonguer, the snailer uses various
biotopes for life. More often this animal meets among trees in damp tropical
rainforest. It perfectly swarms up trees: fingers of snailer are thin and
sensitive similarly to fingers of primates. But they are armed with sharp
claws, permitting
to cling, clamber and if necessary to break rotten wood. Tail of snailer
is rather flexible and partly prehensile. Colouring of this mammal is very
contrast,
cryptic: the forward part of body (head, shoulders and chest) is colored
black, back one is light grey, almost white. On forepaws of animal there
are white
“gloves” up to elbows and on waist and basis of tail – black round stains.
The tail of animal partly imitates colouring of some wood snakes that protects
animal from predators.
This species eats spineless invertebrates – larvae, worms and snails. Snailer
is able to break rotten tree and to take by tongue from burrows larvae of
capricorn beetles. Animal determines their presence, sniffing at apertures,
bored by
larvae in wood. The most usual catch of this animal includes ground snails
making over half of its diet. Snailer takes them from shell, having cut edge
of mollusc body by claw of the thumb near the edge. After that animal pins
snail body on tongue, having pushed it in shell deeply to all coils, and
by one jerk takes mollusc meat entirely, has left shell empty.
If it is a little number of ground snails in its habitat, animals from populations
living near to rivers can dive into water, catching water snails. Having
pulled out from water such catch, snailer carefully shakes off, and then
deals with
mollusc. Usually water snails, as against to ground ones, have protective
corneous operculum on their shells. Snailer cuts its contractor by claw and
then eats
mollusc.
Snailers are solitary territorial animals. But as against to wormtonguers
they eat more accessible and various forage therefore they do not have sharp
intraspecific
competition, and they are more tolerant concern to presence of neighbours.
As well as wormtonguer, snailer breeds some times per one year. Two times
per year female gives rise to 5 – 6 cubs. Male finds female in its territory
(usually
it is the male from the fodder site located near), and some time both animals
keep and fed together. Pairing repeats some times but when female will feel
approach of pregnancy, it banishes male.
Cubs are born naked and blind, but grow quickly. At monthly age they actively
study world around, observing for mother and founding food under her supervision.
And at the age of three months they become completely independent. At the
age of one and half years female is capable to bear posterity. Life expectancy
of snailer is usually rather short – no more than 8 years. Obviously, it
is
connected with fact that adult animals are practically without exception
infected with helminths, receiving them from forage – ground snails.
Ferocious condylutra (Condylutra ferox)
Order: Shrew-form (Soricomorpha)
Family: Moles (Talpidae)
Habitat: North America, temperate and subtropical latitudes, to the north up
to Mishe-Nama Lake (southern and western coast); freshwater reservoirs of various
kinds – from lakes up to swamps.

Picture by Alexander Smyslov
During the evolution process low-specialized forms receive
advantage in survival first of all. Due to absence of specialization they show
a wide spectrum of
variability and can occupy various ecological niches. Highly specialized
species, on the contrary, can develop only aside the amplification of specialization.
This principle is shown evidently by insectivorous mammals of mole family.
In Neocene their specialized representatives have kept characteristic shape
of digging animal, and evolution of unspecialized forms had resulted in completely
unexpected results.
In areas of temperate climate of North America one of unusual descendants
of moles of Holocene epoch lives. The structure of muzzle gives out its origin:
it is crowned up by long mobile proboscis which tip is surrounded with fleshy
outgrowths. This animal is very large descendant of star-nosed mole (Condylura
cristata) which leads semi-aquatic habit of life. The name of this animal,
“Condylutra”, combines two names: “Condylura” – star-nosed mole (an ancestor
of this species), and “Lutra” – otter (this animal partly replaces it in
ecosystems).
Condylutra represents the ecological analogue of platypus and mink – it is
aquatic carnivore eating large invertebrates and small vertebrates. This
is an animal of streamline shape up to 60 cm long not including tail; individuals
from southern populations are smaller.
Head of condylutra is flat and lengthened, with low brain cavity. Eyes of
this mammal are very small, and sight is bad. But this animal searches for
prey
under water not with the help of sight. Its main sensitive organ is long
snout extended to flexible mobile proboscis and covered with naked pink skin.
On
tip of muzzle of its ancestor mobile sensitive shoots grew, helping to search
for food. In evolution process condylutra had further improvement of this
sensitive organ. Overgrown shoots of condylutra’s snout have electrosensitivity.
With
the help of this feature animal can search in muddy water for small animals
by their electric field. External auricles at this animal are reduced, and
small ear apertures are closed by reduction of special ring muscle at diving.
Wool of condylutra is rich and velvety, colored grey. To give it the water-repellent
properties animal regularly greases hair with oily liquid secreting by two
large glands at the basis of tail. The animal finds a lot of time to look
after wool, combing it by claws of hind legs.
Condylutra leads aquatic way of life and is able to swim quickly. Diving
for prey, animal can stay under water till about 5 minutes. Short (about
10 cm
long) tail of condylutra is similar to beaver’s tail a little. It is rounded
from sides, and its back edge is dulled. Tail is covered with dense rigid
skin and thin hair; it helps to speed-up at swimming and is usually used,
if it
is necessary for animal to swim quickly, not turning off (for example, escaping
from predator or banishing the congener). In this case condylutra swims with
the help of wavy movements of backbone in vertical plane. For underwater
hunting animal uses other style of swimming. Forepaws of condylutra are transformed
to narrow paddles with well advanced claws; animal uses them when it is necessary
to swim slowly and maneuverably, surveying places in which prey may hide.
Diving
for food, this animal digs out bottom by claws of forepaws, overturns snags
and climbs among stalks of water plants. Also with the help of forepaws condylutra
digs at the riverbanks and lake coasts long holes with several exits. The
main exit opens right under water, and some emergency exits in case of flooding
of hole or occurrence of predator lead to shelters and to the ground surface.
Condylutra has kept predating habits of ancestor: it is exclusivaly carnivore.
Teeth of condylutra are numerous (44 ones) and pointed. This animal eats
any food of animal origin which may be found in water and near water: worms,
molluscs,
crayfishes and crabs, dead or weakened fish. Condylutra can ravage clutches
and even attacks chicks of waterfowl, snatching them from under water.
It is solitary species with primitive behaviour, not forming pairs even in
courtship season. Each individual marks the territory with musky secretions
and protects it from relatives. Pairing at this species takes place in early
spring, after clearing of reservoirs from ice. In southern part of area the
repeated courtship season occurs in second half of summer. Male searches
for the female ready to pairing guiding by smell, pairs with it quickly and
after
pairing the female banishes him (she is larger and stronger, rather than
male). Once a year (at the south of an area 2 times per year) the female
gives rise
to 4 – 5 cubs in deep hole. The nesting chamber is always above water level.
Newborn cubs are naked and blind. They develop within 2 months, leave the
female and lead independent life. Young animals become sexually mature at
the age
of 6 – 7 months: in southern parts of area young animals from the second
litter already participate in pairing and give rise to posterity the next
year.
Life expectancy of condylutra does not exceed 5 years.
Long-spiked
maurihystrix (Maurihystrix longispinosus)
Order: Afrosoricida (Afrosoricida)
Family: Tenrecs (Tenrecidae)
Habitat: Mauritius Island, highlands.

Picture by Alexander Smyslov
Representatives of ancient family of tenrecs in human epoch
lived mainly at Madagascar and Comoro Islands (from this family only African
water shrews lived in Africa). One of their representatives, rather large species,
tailless tenrec (Tenrec ecaudatus), had been acclimatized by people at Mauritius
Island. After extinction of mankind activity of introduced species had completely
changed the nature of island, and evolution at Mauritius Island has gone in
other direction.
The descendant of tailless tenrec is one of species of Neocene fauna of this
island. At tenrecs there is very small brain, and this animal does not differ
in complexity of behaviour. For this reason evolution of tenrec descendant has
gone to the way of improvement of passive protection that was promoted by fine
inclinations getting from the ancestor. The body of ancestral species was covered
with wool with bristle-like spikes. This feature had received powerful development
at the Neocene descendant, and this way the maurihystrix (literally means “Mauritian
porcupine”) – the large tenrec with long spikes, similar to a porcupine, had
evolved at this island.
The body length of maurihystrix reaches 60 cm at the weight of about 5 kgs.
This is plantigrade sluggish animal with short limbs and rather large head.
Paws of animal are tenacious, and on fingers strong claws grow, with which help
animal gets various food. Head of maurihystrix is lengthened, with peaked forward
teeth and nose turned to short mobile proboscis.
Wool of maurihystrix is short and bristly, grey-colored; on stomach it is lighter.
On top of head of animal wool forms long hair “cop”. The top part of body (back,
nape, top part of neck and shoulders) of this species is covered with spikes
up to 5 cm long. Along the middle of back the crest of lengthened (up to 15
cm) spikes grows; with its help animal puts to predators dangerous wounds. Spikes
of young animals are dark, at adults they gradually brighten.
This mammal lives in mountains and other dry places, thus avoiding a competition
to Mauritian dwarf pigs, which are more moisture-loving. Maurihystrix is able
to climb on trees. Adult individuals do it very clumsily, but young maurihystrixes
climb quickly and dexterously.
This is solitary animal. Maurihystrix does not avoid relatives, but concerns
to them indifferently, not establishing hierarchy. Only at appreciable lack
of forage animals show aggression to relatives. Sight at maurihystrix is weak,
and eyes are small. For it keen hearing and sense of smell are more important,
because the animal often searches for food in high grass where visibility is
very bad. This species is omnivorous. The significant part of its diet the vegetative
food– roots and fruits of plants – occupies. Also it willingly eats insects
and small reptiles, digs grubs and worms from ground. At an opportunity maurihystrix
eats carrion.
The disturbed animal hisses and rears spikes, having turned sideways aside the
enemy. The attacked animal actively shows an opportunity of self-defense, jumping
up on four paws and having curved back like a cat.
Seasonal prevalence in breeding of maurihystrixes is not expressed. Pairing
is not anticipated by the courtsip ritual, and the female raises posterity alone.
Twice per one year it gives rise to many cubs – up to 15 ones in one litter.
Newborn maurihystrixes are completely defenceless. They are blind, covered with
thin wool without spikes. The female arranges for them a den in rich bush, or
digs a hole under roots of tree. At week-aged cubs eyes are already opened,
and wool becomes thicker. At three-week age they start to move in den actively
and try to explore vicinities. Monthly cubs can already not lag behind the female.
They abandon the nest and follow mother in wood. At this time at them spikes
start to grow. The highest death rate at maurihystrixes takes place during the
pass to independent life. The sexual maturity at young animals comes at the
age of 18 months. Life expectancy of maurihystrixes reaches 10 years.
Flabellitherium,
fan beast (Flabellitherium semisolaris)
Order: Afrosoricida (Afrosoricida)
Family: Tenrecs (Tenrecidae)
Habitat: Madagascar, savannas of the western part of the island.
In human era tenrecs were among the typical representatives of mammalian fauna
of Madagascar. This group of mammals demonstrated a significant species diversity
and differences in lifestyle. Among tenrecs arboreal, digging, aquatic and terrestrial
forms existed. In human epoch there was a significant reduction in the number
of these animals, and a number of species became extinct during the Ice Age
at the boundary of Holocene and Neocene. But the survived species continued
evolving, and among them new and unusual forms appeared, sometimes
reaching large sizes. The species richness of small and medium-sized
tenrecs also increased, and among them forms having unusual anatomical and behavioral
characteristics appeared. One of these species is the whimsical flabellitherium,
the “fan beast”, living in savannas in the west of the island.
The flabellitherium is a descendant of the tailless tenrec (Tenrec ecaudatus),
a widespread and easily adapting species of this family. This species evolved
to strengthen its passive protection, and eventually on the plains of Madagascar
a tenrec similar to a porcupine appeared. The flabellitherium is a chunky and
slowly moving beast; the adult weighs about 8 kilograms and resembles a small
badger in constitution. If necessary, it can run fast for short distances, but
cannot run for a long time. In fact, it prefers not to run away from enemies,
but to use other methods of self-defense.
The back of flabellitherium is covered with durable movable spikes up to 20
cm long, which provide it the main protection from enemies. Spikes also grow
on the animal’s head and neck, and also there is a stripe-like zone of their
growth along the midline of the back up to the base of the very short tail.
At the base of the spikes sparse and rigid elongated wool grows, visually enhancing
the outline of the animal. Some spikes are thickened and hollow – they are used
as rattles, further warning predators of intent of the beast to protect itself.
The colouring of sides and belly of the flabellitherium is grayish-brown; on
the back colors are more contrasting: a white background, on which black spots
and strokes of various shapes are scattered. Spikes and wool between the spikes
are grayish-white. This is warning coloring, reinforcing its threatening demonstration.
When frightened the flabellitherium raises upright its spikes that stick out
in all directions and make the outline of the beast look like a fan. At the
same time the flabellitherium turns its side to the predator, so the black-and-white
color of his back is clearly visible to the aggressor. Until the predator keeps
its distance, the flabellitherium loudly hisses and squeals, occasionally slightly
trembling wit its body. At this point the spikes knock against each other, and
a loud cracking sound is heard. If the predator attacks, the beast turns to
it by spikes and tries to prick it. During the fight flabellitheriums bite strongly.
This animal has an elongated snout and a movable prominent proboscis-like nose.
Its weak long jaws are equipped with pointed teeth. Resembling a porcupine outwardly,
flabellitherium differs from it in diet: this is a zoophagous species, feeding
on insects, small vertebrates and carrion. On occasion it willingly ravages
bird nests.
Seasonality in reproduction at flabellitheriums is not expressed. This species
does not form permanent pairs and each animal usually occupies a certain territory,
banishing its relatives from it. Male finds a female ready to pair by smell
and encroaching on her territory. It displays itself to the female, pacing in
front of her on outstretched legs, slightly trembling and chattering its spikes.
At the same time it opens its spike fan and bars the way of the female. If she
tries to leave, the male rattles its spikes stronger and runs at her head, trying
to stop her. If the female does not try to move out, the male gradually calms
down and lowers its spikes, showing thus the absence of aggression. After mating
the pair separates. The female can mate, still taking care for cubs of its previous
litter.
Pregnancy lasts up to 60 days; during the year the female can make 2-3 litters
of cubs, and then rests for about six months. The female gives birth to 1-10
cubs in a hole that she digs herself or occupies someone else’s. At birth, the
cubs are hairless and blind, but within a few days after birth they are covered
with hair, and at the age of 2 weeks their eyes open and spikes begin to grow.
At the age of 6-7 weeks cubs can already leave the hole and walk through the
territory with their mother, learning to search of food. Three-month young animals
already leave their mother and live independently. At the age of 1 year, the
animals reach sexual maturity. Life expectancy is 10 years.
Translated by FanboyPhilosopher.
Shield-faced mole tenrec (Malagasytalpa scutifera)
Order: Afrosoricida (Afrosoricida)
Family: Tenrecs (Tenrecidae)
Habitat: Madagascar, savannas and woodlands.
The fauna of Madagascar got very serious damage because of human activity.
The large species lived at the island before human colonization had died out
within several centuries. The species living in forests also had suffered.
But even in human epoch island had been inhabited by groups of small endemic
animals. Among them the tenrec family expressed remarkable variety. It included
the species occupying various ecological niches and having shapes differing
in great degree. This family has kept a variety in human epoch, and in Neocene
it entered new heyday. Among Neocene tenrecs there are huge
species, but all
the same the majority of their species has the small size.
Insectivorous mammals of families typical for continental Africa do not live
on Madagascar in Neocene, and tenrecs have occupied their ecological niches.
In forests and bushes, in humus-rich soil many kinds of invertebrates live.
And the top predator of underground habitats is the separate species of tenrecs
looking like mole. This is a blind creature having powerful digging paws –
shield-faced mole tenrec, the descendant of rice tenrecs (Oryzorictes spp.)
inhabited this island in human epoch. It is specialized deeper to burrowing
habit of life, rather than its ancestors. This animal is up to 20 cm long with
long naked tail, short head and cylindrical body. The basic digging instruments
are large forepaws; 3-rd and 4-th fingers carry huge claws on wide bases. 2-nd
finger is short, and thumb can oppose against fingers. Nestling to digging
claws it helps in keeping food and to bringing it to the mouth. Hinder legs
have four toes, narrow foot and long claws. Tail is covered with corneous scales
of grey color and thin hairs.
At this animal eyes had completely disappeared, and even the optic nerve had
undergone a significant degeneration. Auricles visible outside are lack at
this animal, and the acoustic duct is protected by small skin valve. Animal
has massive bones; therefore it is not able to swim. But bones conduct the
sounds extending through the ground, and this feature helps an animal to orientate
in environmental conditions. Also whiskers growing on muzzle of animal serve
for orientation. The muzzle of animal takes part in underground tunnel burrowing,
and the top part of head of this tenrec is protected by strong hairless corneous
plate of brownish-yellow color comparable in hardness with claws of animal;
hence the name of animal and specific epithet “scutifera” meaning “shield-bearing”.
Short velvety fur of black color with brownish shade permits this animal to
move back and forth in tunnels equally easy.
Shield-faced mole tenrec is flesh-eater. It feeds on various invertebrates,
attacks frogs, reptiles and the small rodents get into its tunnels. This species
leads solitary way of life, and two individuals of this species behave aggressively
having met each other. Among shield-faced mole tenrecs cannibalism is usual,
and adult individuals frequently eat young growth, and even attack newborn
cubs. Pairing is possible at any time of year. The female ready to meeting
with male leaves odorous marks of pungent smell in tunnels. Pregnancy lasts
till only 10 – 12 days. The female brings up to 10 naked helpless cubs quickly
growing and becoming independent at the age of 2 months. In 5 months age young
animals become adults. Feeding up posterity the female grows thin considerably,
and after young animals left her, it restores the physical shape for a long
time. For one year it brings up to three litters. Life expectancy of shield-faced
mole tenrec does not exceed 4 years.
Stream
elephant shrew (Desmanella natans)
Order: Elephant shrews (Macroscelidea)
Family: Elephant shrews (Macroscelididae)
Habitat: mountain areas of Zinj Land.

Picture by fanboyphilosopher
Split off from African continent, Eastern Africa, or Zinj
Land, had carried away the highest mountain tops of the continent. In Neocene
mountain tops of Zinj Land are still the kind of water wapour condenser, and
the numerous streams merging in rough rivers full of rapids flowing down from
them. In cold water water mosses grow, and their sprouts wave in current. Stones
and plants are populated with numerous larvae of insects – May flies, caddis
flies, midges and mosquitoes. Some of them scrape bacterial layer from stones
and plants, others eat plants, and the third ones are predators and hunt not
only invertebrates, but also fish fry. And insects, in turn, represent the food
of original representative of elephant shrews adapted to aquatic lifestyle.
This species, stream elephant shrew, feels like very well in cold water and
easily swims against fast current of streams. It descends from one species of
elephant shrews of genus Elephantulus widely settled in Eastern Africa. Stream
elephant shrew is a small species: it is like young brown rat the size. Fur
of this animal is water-repellent, very dense and velvety. Colouring of fur
is black: it helps this animal to be warmed quickly in sunny place, and it is
very important at life in cold water of mountain streams. This mammal dived
under water seems silvery because of air layer kept in wool. In order to support
water-repellent properties of fur, stream elephant shrew takes a great care
to it, combs it by claws and greases with oily secretions of glands developed
in armpits. Forepaws of this animal are mobile and have sharp claws. Hind legs
are much longer than forepaws, and the animal moves overland by jumps like jerboa.
If necessary stream elephant shrew is capable to make jumps up to three meters
long, and easily crosses the river by several jumps on stones, escaping from
predators. This animal swims with the help of hind legs, having pressed forepaws
against chest. Edges of feet are covered with long rigid bristles unbending
during the swimming and increasing a rowing surface. Swimming animal makes pushes
by two hind legs simultaneously. The tail of animal is long and thick – a stock
of fat gathers in it being used in case of starvation.
Stream elephant shrew has very small auricles. The external ear of animal seems
hidden in skin, and the top edge of auricle blocks an acoustic duct under water
serving as a valve. The mobile proboscis helps this animal to search for food
in water. Due to large eyes stream elephant shrew has good eyesight both on
land and under water.
This exclusively species is an original kind of “desman” in mountain areas of
Zinj Land. His diet includes insects and their larvae, snails, crustaceans and
small fishes. Within the day one animal eats food in amount approximately equal
to two thirds of its body weight. Stream elephant shrew leads exclusively solitaru
habit of life, only the female and her posterity up to achievement of sexual
maturity live together. This animal is strictly territorial, and marks the territory
with the same secretions as it greases wool. For this purpose little mammal
simply clamps a stalk of a grass between a body and a forepaw, and drags it
through an armpit.
The courtship season at this species is not expressed clearly, and posterity
is born at any time of year. Male searches for the females ready to pairing,
guided by smell of their territorial marks. At this time it is extremely aggressive
to contenders, and frequently kills their by bite in head. Three times per one
year, after the pregnancy continuing about seven weeks, the female gives rise
to posterity – 2 – 3 blind and helpless cubs covered with rich wool. The female
hides a litter in shelter – usually in hole dug out under stones and covered
with dry leaves. Within one month cubs grow and study to extract food, then
the female stops to feed them with milk and only enables to spend the night
in her shelter. The bi-monthly cub already leads independent life, and the four-monthly
female is ready to pairing.
In especially cold time this species can run into short-term hibernation of
which delay of physiological processes is typical and body temperature drops
down to + 10 – 12°С. Due to this feature the organism of animal grows old
not as quickly as at constantly active animals having similar physiology. Stream
elephant shrew from high-mountainous areas are able to live 8 – 9 years, and
individuals from warmer areas live no more than 4 years.
Ant
elephant shrew (Myrmisengi saltator)
Order: Elephant shrews (Macroscelidea)
Family: Elephant shrews (Macroscelididae)
Habitat: Zinj Land, rainforests.

Picture by Alexander Smyslov
Elephant shrews represent a group of mammals very characteristic
for Africa and kept primitive body plan. Split of Africa and separating of East-African
subcontinent (Zinj Land) have resulted in isolation of the species survived
in human epoch. And restoration of tropical forests has allowed some species
to develop a new inhabitancy – forest underbrush.
The large species of elephant shrews lives in forests of Zinj Land – shaggy
elephantine sengi (Rhynchoporcus monstrosus). It is one of largest species
– its length is from above half meter. But in the neighbourhood of it the tiny
mammal moving by jumps on two hinder legs lives – ant elephant shrew. This tiny
mammal is about 10 cm long, and tail makes approximately the same length. It
has wide mobile ears, large eyes and mobile sensitive proboscis. Hind legs of
this little mammal are longer than front ones, on bottom side of toes corneous
callouses develop, improving traction with a ground. Forepaws are mobile and
have thin sensitive fingers.
The wool of this mammal is short and has reddish-brown color with black longitudinal
strip from nape up to the basis of tail. Tail, paws and ears are hairless, covered
with dark skin. Above eyes two bright yellow spots are located, helping to distinguish
relatives in twilight of forest floor.
This species of animals leads an unusual way of life and searches for meeting
of creatures carrying fear and panic to the majority of forest inhabitants.
Ant elephant shrew searches for the columns of army ants creeping on the ground
and keeps near them. The presence of ants frightens inhabitants of underbrush
and forces them to search for a new refuge. And ant elephant shrew easily notices
and catches them. One more source of forage is everything carried by columns
of ants accompanied with it: the caught insects and larvae, and also ants’ pupae.
Due to speed this little mammal can snatch out ants burdened with load from
a column, and furious soldier ants simply have no time to do anything against
it. Also this elephant shrew can hunt insects itself. It licks prey off by long
tongue covered with sticky saliva.
These animals lead a solitary way of life, but have no precisely determined
territory. If near to ant column two animal of this species meet, between them
the fight flashes, accompanying with shrill squeak and bites of the opponent
to shoulders and ears.
Pairing does not have the expressed seasonal character. Male finds the female
by smell, pairs with her and immediately leaves her. After the pregnancy lasting
about 6 weeks, well advanced cubs are born, capable to move independently. The
female brings three litters (1 – 2 cubs each time) during one year. Posterity
stays with her till first four weeks of life, and then starts hunting independently.
Ant elephant shrew grows very quickly: the three-monthly female is already able
to bring posterity. But life expectancy at this species does not exceed three
years.
This species of mammals was discovered by Simon, the forum member.
Tree
micropangola, “walking pinecone” (Micropangola scandens)
Order: Pangolins (Pholidota)
Family: Pangolins (Manidae)
Habitat: tropical rainforests of Hindustan, South East Asia, tree crones.

Picture by Alexander Smyslov
The order Pholidota was never especially numerous and diverse.
At the beginning of their evolution these animals had adapted to feed on insects,
having become ecological analogues of anteaters in Old World. This specialization
caused their conservatism and imposed certain restrictions to the body size
and way of life, having allowed animals to vary in quite strict limits. The
upper limit of the body size of pangolins is rather rigidly limited, but the
lower limit is limited by nothing, and one of Neocene species of pangolins –
tree micropangola – shows it.
This species is adapted to life in tropical rainforest where it inhabits forest
canopy – the place richest in life. To make it possible the animal had to offer
in the sizes and physical power. Tree micropangola differs from its relatives
in small size and truncated snout. The body length of this animal is about a
half a meter, and wide muscled tail takes a half of this length. The body of
micropangola is covered with corneous scales, as at its ancestor. But the scales
are rather thick and strong, and the scales along the spine have pointed tips.
On head and paws scales actually turn to corneous scutes located closely to
each other. Primary color of scales is reddish-brown; sides of head are coloured
lighter a little.
This animal has short snout, but the tongue is long and flexible. Its base is
shifted far back and is attached to chest bone. Due to elasticity of tissues
the tongue may extend from the mouth at the distance equal to double length
of the head. Ears and eyes of this animal are small, and sight sense is bad.
But it is compensated very much by keen senses of smell and touch which are
used in food search. The feeding animal constantly taps on tree with claws and
determines by the type of wood vibration the existence of cavities in wood mass
– the tunnels and the chambers gnawed by insects. Having felt their presence,
it breaks wood by claws and penetrates the tongue into the opened tunnels, licking
off the insects.
Paws of tree micropangola are adapted for grabbing of branches. On forepaws
strong and slightly bent claws nestle against the palm and form strong grap,
allowing the animal to cling even on thin branches. Feet of hinder legs are
covered with corneous combs allowing clinging to bark and branches much stronger.
The tip of tail of this animal is expanded, and scales bordering the body edges
are strong and peaked – it is an additional adaptation for fixation on trunk
and branches. A usual pose of feeding animal is the perching a tail downwards.
Thus scales at the back edge of tail stick to the bark and take up weight of
the animal.
The main protective adaptation at this species is the set of strong corneous
scales. They defend tree micropangola against birds and small arboreal predators.
Defending itself, micropangola sits across the branch, envelopes it with tail
like a ring, and covers the head with tail tip. Due to strong muscles the animal
can keep such pose till about one hour. If the predator doesn’t stop attempts
of attack or is too strong, tree micropangola can defend itself actively, imposing
on it the near battle which can finish with serious injuries for the attacker.
Thus, having grab the branch by hind legs and having clasped it by the tail,
the animal sharply throws up a body, strikes to the enemy some fast blows by
large claws of forepaws, and then clasps a branch again and hides the head under
tail tip.
The seasonality in reproduction of this speciees isn’t expressed, but in areas
of monsoonal climate the most number of youngs appears in rain season. The courtship
ritual of tree micropangola is primitive and is reduced to chase of female by
male. After pairing animals abandon each other. There is only one cub always;
it stays with mother up to three-monthly age and then starts to lead independent
life. Life expectancy doesn’t exceed 12 years.
The idea about existence of this species of mammals was suggested
by Simon, the forum member.
Translated by Alexander Smyslov.
Mole
pangolin (Talpangola laticauda)
Order: Pangolins (Pholidota)
Family: Digging pangolins (Talpangolidae)
Habitat: Southern and South-Eastern Asia, woods and light forests.
Order of pangolins is rather conservative group of mammals. From early Cenozoic,
according known fossils, they almost did not change structure and habit of life.
But in epoch of mass extinction few successful forms which have standed “durability
test” start to evolve actively, giving rise to new lifeforms.
Obviously on this wave one of pangolin species living in woods of South-Eastern
Asia, had mastered new to this group habit of life, becoming practically completely
underground animal.
Common length of the Asian mole pangolin is up to half meter including tail.
Head of animal is wedge-shaped, flattened, covered with corneous plates forming
continuous protective armour. With the help of such head animal can ram friable
wood ground, digging a hole to it. Eyes are very small, protected from above
by corneous “eyebrowes”. To protect eyes from ground at animal in addition there
are advanced dense brushy eyelashes. Auricles are not present; ear apertures
are covered by special corneous plates. Sight and hearing at mole pangolin are
bad, but sense of smell with which help the animal searches for the forage (ground
invertebrates, including social insects) is perfectly advanced. Nasal cavities
occupy significant part of skull.
Forepaws with powerful claws are the basic “digging instrument” of mole pangolin.
Claws on II and III fingers are especially advanced. Back legs are short, plantigrade,
with large feet. Almost all loading during digging and movement falls on them.
At all species of pangolins known in human epoch tails were very long. Turning
such tail around of body, pangolin could feel like in full safety from predators.
At the mole pangolin the protective role of tail is lost, as the animal spends
almost all life in holes, getting out from them only in case of emergency. Tail
of the mole pangolin is wide and short, during short travel on surface of the
ground it can protect an animal: the disturbed mole pangolin can turn having
covered belly and partly head with tail. If the enemy will not recede, the animal
makes sharp movements by forepaws, trying to wound the enemy by sharp claws.
Body is covered with scale armour, as at all representatives of order. Between
scales it is only few wool, it is very thin. Edges of scales are covered with
corneous hairs that strengthens coupling of scales and make their abutment more
dense. Colouring of scales is rosy-brown. Because of continual friction against
ground scales of animal are “polished” and gleam slightly. Horn hairs are formed
at rubbing of corneous layer on edge of scale, and represent ends of fibres
forming every separate scale.
During digging mole pangolin serially shovels ground by claws under belly and
throws it back by legs. The main reason of special form of tail of this animal
is its use during digging holes: from time to time the animal lowers shovel-like
tail and starts to move back, raking by it away shoveled up ground to hole entrance.
This mammal eats insects and other invertebrates licking them off by long tongue.
Basis of its diet are larvae of beetles rummaging in ground, earthworms and
snails dug in the ground. Frequently this pangolin digs burrows to termitaries,
visiting them by turns and breaking underground part of insect nests. When from
the damaged nest hundreds of “soldiers” and working individuals run out for
repair of it, animal simply licks them off by tongue, staying still.
Twice per year female gives rise to 1 – 2 cubs. As opposed to other pangolins,
cubs keep on stomach of mother - if they’ll sat on back as at ground and climbing
pangolins, female simply would dump them during movement. The female carrying
cubs, digs ground more cautiously, preferring to move in ready holes.
Cubs leave mother’s stomach at fortnight age, but about one month they are fed
with her milk. They become completely adult at the age of half-year. Life expectancy
of this animal reaches 10 – 12 years.
Lupardus (Lupardus tenuis)
Order: Carnivores (Carnivora)
Family: Canids (Canidae)
Habitat: Northern America, plains and semidesert.

Picture by Eugeny Hontor
During anthropogenous pressure on nature number and variety of most part
of large “noble” predators (felines, bears, canines etc.) had strongly
decreased,
and some species had completely disappeared. Populations of predators were
so strongly undermined by human activity, that the most part of carnivores
species had not gone through ecological crisis at the boundary of Holocene
and Neocene, and had simply died out during several hundreds of thousand
years when on Earth unproductive ecosystems had expressed. But completely
new
opportunities had been received by species which had successfully coexisted
humans. In Northern America such species was coyote (Canis latrans) which
had widely settled at the continent due to extermination of wolf and woods
slash.
This species had reached prosperity in Northern America, having evolved to
some new species of predators.
One of descendants of coyote inhabits plains overgrown with grasses in the
central part of the continent, stretched like wide strip along Rocky mountains
from the north to the south. This animal is externally similar not to coyote,
and not to the representative of canine family at all, and more to someone
of felines. This similarity is reflected in the name of an animal: derivative
of words Lupus (Latin “wolf”) and “-pardus” – the ending of names of some
felines.
This species is specialized in catching of swift-footed medium-sized catch
that had resulted in formation of running predator of the special appearance.
A constitution of lupardus is rather light: animal has muscled sinewy body,
thin long legs and flexible backbone. Claws at lupardus are short, but thick
and sharp: they improve coupling with ground at run. By appearance it resembles
other swift-footed predatory animals of Neocene: African pardinia and
Middle-Asian zibetonyx (both animals are
representatives of civet family). Losing to other predators in physical strength,
lupardus surpasses them in speed: it is the
fastest animal of the American continent, accelerating momentum up to 90
kms per hour at the short distance. During chasing of swift-footed catch
it is
practically out of competition.
At the lupardus there is small head with short muzzle. Jaws of the animal
are rather short, but strong, with moderately long canines. Eyes are directed
forward
and provide good three-dimensional sight. The nasal cavity is short, but
wide, and mucous membrane forms inside it numerous plicas. It considerably
aggravates
sense of smell of predator.
During intensive muscular work the animal is exposed to serious danger of
overheat. The lupardus partly escapes from it due to fragile constitution,
but it has
also extra for cooling – very wide ears similar to ears of some desert foxes.
When the lupardus chases catch, blood vessels penetrating ears, dilate, and
excessive heat dissipates. Big auricles provide to the lupardus good hearing,
that is especially important at hunting for small catch in high grass, where
the visibility is bad.
Skin of the lupardus is colored light rusty with thin close black cross strips.
This colouring resembles a little “tiger-like” colour of some breeds of domestic
dogs. Legs of lupardus are dark; strips are not expressed on them. Internal
side of wide ears is grey, but on external side of ear there is the big white
spot edged with black wool. When lupardus is turned by muzzle directly to
catch, its prey does not see the predator on the background of grass, but
congeners
standing behind perfectly notice white spots on its ears.
The basic catch of luparduses is deermara –
the running rodent of South-American origin similar to small antelope. Except
for them these predators hunt large
flightless ostrich turkeys.
Less often luparduses attack larger animals of American plains: donkeyhorses and
young peccasons. At lack
of large prey lupardus eats small animals: rodents, lizards and snakes. Usually
it avoids to hunt
among bushes or in high grass, preferring plains where grass is short because
numerous herbivores eat and tramble it. Tracking down its catch, lupardus
relies first of all on sharp sight; therefore for it the good field of view
is important.
Besides the high grass complicates run, and animal simply may fall, having
hooked for stalks and having injured its legs. Having caught up prey, luparduses
put to it strong bites in sides and hips, trying to pull out a piece of meat.
With each such wound animal weakens more and more, and soon it falls, becoming
catch of predators.
Swift-footed animals have one general feature: they get tired very quickly,
and can not accelerate the maximal speed for a long time. Lupardus has the
same feature, but it easily compensates such lack by other features of behaviour.
As against cats and pardinias, and similarly to zibetonyx, it is packing
hunter. Luparduses lead the chasing of catch by organized way, and in pack
always there
are animals, ready to join chasing. The pack of luparduses cuts the selected
catch from the common herd, and drives it up to exhaustion. Thus hunting
animals arrange ambushes, lining up. Herbivores cut from herd are “transferred”
by
hunters from one to another. In this case the prey is every time chased by
all new animals, and by the end of chasing it is literally ready to fall
down of tiredness.
Such hunting tactics can not be organized without close interaction between
members of clan. Outside of hunting lupardus is the sociable family animal
closely communicating with congeners. Similarly to other canids, lupardus
uses for communication a tail with a white tip. By position of tail and ears
animal
expresses emotion and shows the mood. The clan is ruled by main pair in which
the male is mainer than the female. Dominant male rules all males in clan
and all females submit to the main female. Youngest animals are outside of
the
common hierarchy: the relation to cubs at luparduses is the most gentle and
touching. Each female twice per one year brings posterity: up to three –
four clumsy long-legged puppies. They differ from adult animals in black
colouring,
and it forces even dominants to suffer all their children's pranks.
The grown up animals also outside of hierarchy, but for other reason: they
are driven off by adult animals, and young, but not developed to the full,
animals lead hard life, eating scraps. Gradually they study to hunt. It occurs
during the big hunting for large prey. In the beginning young animals are
on supporting parts, and then most capable of them quickly rise on a scale
of
ranks up to the high position. And once any young strong lupardus will overthrow
the leader, proving its own hunting abilities.
Nearctic
hyena-toother (Neohyaenognathus macrodens)
Order: Carnivores (Carnivora)
Family: Canids (Canidae)
Habitat: cold and temperate areas of North America, various landscapes – from
plains up to deciduous and coniferous woods.
Picture by Carlos Pizcueta (Electreel)

Picture by Eugeny Hontor
Canids are one of ancient groups of predators. They had made great progress
at flat landscapes being habitat of numerous herbivores. In Neocene their destiny
was changeable: having reached short-term success at plains of ice age, canids
had conceded their positions to other groups of predators better adapted for
woody districts and bushes. In such conditions canids had kept in very small
amount. Besides number of many species had been undermined by people, and they
had not gone through climatic cataclysms at the boundary of Holocene and Neocene.
But some species managed to survive.
In North America descendants of coyote (Canis latrans) had achieved the great
success in struggle for existence. Coyotes had got the big advantage from the
neighbourhood with people, having expanded the area and having got rid from
wolf, their main competitor. Possible, in first time after human disappearance
hybridization of coyote and descendants of domestic dogs took place; it had
resulted in enrichment of genetic information of new species becoming ancestors
Neocenic of North-American canids. Besides preservation of canids in Neocene
was promoted by geography of continent. Prairies formed in “wind shadow” of
Rocky Mountains, had continued the existence in Neocene. They became the house
for swift-footed herbivorous mammals and “range” for occurrence of new species
of canids. Among them two lines separated, leading radically differing habit
of life. One line of canids is represented by swift-footed packing predator
lupardus, and another one is occupied with massive animal specializing to feeding
by large catch – Nearctic hyena-toother.
Hyena-toother has occupied a convenient ecological niche of scavenger and large
prey hunter. Other representatives of American predators - large cat balam
and packing swift-footed canid lupardus catch small and swift-footed animals.
But huge peccasons, giant pigs of American prairies, are too large and strong
for them. But hyena-toothers are able to overcome such strong herbivore. This
predator lives in small packs (up to 10 – 12 individuals) consisting of dominant
pair and several subordinated pairs: they succeed to overcome large catch due
to harmonious actions. However, they easily snatch catch away at other predators
of prairies. This species effectively consumes large catch: after feast of
pack of hyena-toothers only tears of skin, jaws and leg bones remain from catch.
Other parts of prey are eaten completely.
Hyena-toother is a sizable predator (height at a shoulder is more than 1 meter)
of massive constitution: the adult animal weighs up to 300 kg. It is similar
to large and rather short-legged dog. Shoulders of animal are little bit higher
than waist, and back of animal is sloping. Colouring of animals strongly varies:
from light grey and even yellowish in southern and southwest areas up to grey
with black back and almost completely black at the north.
Paws of hyena-toother are rather short, animal can not run fast and for a long
time, accelerating maximal speed about 40 kms per hour. Toes of this animal
are thick and short, and hyena-toother looks almost plantigrade. Not able to
run fast, it swims well and easily walks on marshy ground due to wide feet.
Often hyena-toothers preys swift-footed herbivores, pushing them to lakes and
swamps and chasing them on fenny ground.
Jaws of hyena-toother are adapted to feeding in carrion and crushing bones
of large animals: they are short and high, with powerful molars. Canines are
big (especially at males: it is an attribute of a sexual dimorphism) and thick,
with powerful bases. Tips of canines jut out from closed mouth of predator.
But at some external similarity to sabertooth cats it is not their analogue
by the habit of prey killing. If sabertooth cats operated by canines as by
daggers, hyena-toother operates by them differently: it puts by canines shallow
lacerations after which catch bleeds profusely.
Eyes of animal are small: sight of hyena-toother is rather weak; it badly distinguishes
colors. But the hearing and sense of smell at it are excellently advanced.
Also at hyena-toother there is high and wide nasal cavity, and olfactory epithelium
forms longitudinal plicas. Ears of animal are big, rounded and mobile.
During search of catch hyena-toothers support contact with each other with
the help of movements of tail with white hairy brush on the tip. When animals
do not hunt, they actively use sounds for communication: hyena-toothers howl,
declaring rights of clan to the territory, and also growl and whimper, expressing
the mood. Puppies can bark, but adult animals lose this ability.
The social habit of life gives big advantages to the hyena-toother in hunting
for large herbivores. Animals of different clans develop special tactics of
hunting for large animals which transfers from generation to generation. Usually
hyena-toothers choose the most perspective catch in herd of herbivores– usually
it is young or rather old animal. They rush into herd, separate the planned
victim from the common herd and drive it up to exhaustion. Hyena-toothers are
not able to run quickly; therefore they drive prey, walking behind it. They
have special places for killing catch where animals prefer to make decisive
throw and to eat catch. Usually for these purposes places fare chosen, from
which it is more convenient to reach lair and cubs.
At the period of hunting the young growth is looked after by old animals of
clan. Usually young animals start to accompany with adults approximately from
four-monthly age. Young males usually remain in parental clan at subordinated
roles with strict hierarchy, and females either pass to other clans, or form
their own one, which is leading by the male come from other clan.
Distribution of hyena-toother in North America to the south is limited by
the border of temperate climatic zone. This animal is too massive; that’s why
it badly tolerates heat. Therefore at Mexican plateau it is replaced by other
American scavenger – marsupial
hyaena (Phascohyaena tigrina), the specialized
predatory descendant of the opossum.
Atshehaske,
Great saber-toothed coyote (Smilodonocyon regius)
Order: Carnivors (Carnivora)
Family: Canids (Canidae)
Habitat: North America – lake coasts and riverbanks, wet grassy sites of prairies
and meadows.
Picture by Pavel Volkov, colorization by Biolog

Picture by Cossus
Initial picture by Pavel Volkov |
Coyote (Canis latrans) was one of the most successful predators
of North America in late Holocene epoch. In Neocene coyote descendants had radiated
to numerous species of predators of New and Old World. Among them Nearctic
hyena-toother (Neohyaenognathus macrodens) is remarkable – it is huge canid
having the certain features of saber teeth. Hyena-toothers did not become true
saber-toothed predators, because during the evolution process they had adapted
to carrion eating. But one canid descended from the same ancestors as hyena-toother
turned to the real saber-toothed predator. It is atshehaske, huge saber-toothed
coyot. The name “atshehaske” descends from Great Coyote – the Supreme deity
of Havajo Indians. Like its “divine namesake” atshehaske differs in complex
behaviour – this predator has remarkable cunning, insidiousness and nasty customs.
The Latin name of species means “regal saber-toothed dog” and emphasizes appearance
of this animal as well as possible.
Atshehaske is extraordinary beautiful and majestic animal. It is built lighter,
rather than hyena-toother (the maximal weight of atshehaske does not exceed
150 kg.), and has more graceful proportions. The reddish skin of atshehaske
is patterned with beautiful spots and strips of yellowish-grey, brownish and
black colors. Males have beautiful yellowish-grey shabrack with reddish shade,
and females lack of it. On paws there are cross strips breaking to separate
spots on shoulders and hips. Tail is short enough, but fluffy, with black tip.
Like hyena-toother, atshehaske has sloping back, but on the whole its constitution
is much more graceful. Muzzle of atshehaske is rather short. The extended obverse
part of a muzzle characteristic for coyotes evolved to rather massive forward
part of the lower jaw. The upper jaw smoothly passes into powerful and strong
forehead. Prominent feature of the upper jaw is the presence of huge canines
– up to 25 cm long at males and up to 20 cm at females. Animal has large enough
peaked ears on which tips there are beautiful brushes of yellowish-grey fur.
In the whole, despite of low, flat and massive forehead, atshehaske is rather
majestic animal.
On a way of hunting atshehaske is the real analogue of extinct large saber-toothed
cats. It is an ambush hunter for large prey, unting in small groups. Attacking
its prey atshehaske knocks it down and puts a bite. The predator uses canines
as daggers – at the moment of bite in neck of prey canines of atshehaske simultaneously
injure the large blood vessels carrying blood to brain and block respiratory
ways. Prey dies from asphyxia and the termination of blood supply of brain.
One more way of prey killing is putting large deep wounds in prey's belly, that
causes fast death from bleeding. Atshehaske hunts most effectively in thickets
of grass at riverbanks and shores of lakes where large mammals come to watering
place.
Atshehaske is a social species, like the most part of canid species, and lives
in clans united by consanguinity (usually patrilineal). Each clan jealously
protects the hunting territory, marking its borders with urine. The courtship
season begins in winter. At this time males gather near the lair of female ready
to pairing, and arrange severe duels for the right of pairing. They do not use
canines against contenders, but tear skin of each other by incisors, pulling
out large pieces of wool and skin. But direct skirmish of competing males happens
rather seldom. Usually duels are limited to demonstration of canines and an
exchange of ritual poses. After pairing male stays with the female and in common
they build a lair – wide hole under roots of tree. Animals also frequently occupy
caves of natural origin. Male feeds up the female and stays with her until while
his puppies will become independent. In litter it may be up to 3 – 4 puppies
which will become independent hunters at the age of approximately one and half
years. After posterity rearing the female has the year long break in breeding,
and at this time male can couple to another female.
Life expectancy of atshehaske makes about 30 years.
This species of canids in Neocene is at the edge of extinction due to rather
recent occurrence in their area of saber-toothed cat missopeho,
the large predator. Populations of atshehaske are isolated also, and gene exchange
between them proceeds slowly. Therefore small populations quickly degenerate
because of inbreeding depressions. The most successful populations of this species
live in Rocky Mountains and in mixed forests of the north. The large population
lives in Appalachian Mountains, and along the northern border of prairies this
species is present as small isolated populations. In significant area of northern
forests this species was superseded by missopeho.
This species of mammals was discovered by Odin, the forum member.
Common xolot (Anconocyon pratensis)
Order: Carnivors (Carnivora)
Family: Canids (Canidae)
Habitat: North America, meadows and prairies.

Picture by Alexander Smyslov

Picture by Wovoka
Coyote (Canis latrans) was one of the most successful predators
of North America. In human epoch it had considerably expanded an area and had
partly replaced wolf strongly exterminated at that time. Due to smaller size
and variability of behaviour this species had freely adapted to life in anthropogenous
landscape. After human disappearance coyote had kept the positions occupied
in nature and had fixed its success, having penetrated along Beringian Isthmus
to Old World. In North America coyote descendants evolved to animals looking
unlike each other such as swift-footed gracile lupardus
(Lupardus tenuis) and the hyena-toother
(Neohyaenognathus macrodens), massive carrion devourer. But, undoubtedly,
the most exotic and deviated descendant of coyote is xolot – the unusual underground
coyote kind. The name “xolot” descends from the name of Xolotl – Aztec “coyote-headed”
god of underground world.
There are some species of this animal living in North America. Common xolot
is the most widespread among them. Common xolots inhabit meadows and prairies
far from large rivers where there is no danger of flooding. They lead substantially
underground habit of life and settle in small colonies in burrow systems dug
out in prairies by large rodents. Xolots are omnivores with significant inclination
to carnivory, and their food is diverse; they eat food of animal origin (ground
birds, lizards, and also rodents – “builders” of their burrows) and vegetative
food (roots and tubers of plants, young leaves and unripe seeds of graminoids).
Though they rather frequently eat rodents in which holes they settle, xolots
do not cause irreparable damage to colonies of rodents. Besides they rather
jealously relate to their own territory, and do not let other predators to their
home area. Therefore residing of xolots in holes of rodents is favourable for
rodents to some extent. Though xolots attack them, they also simultaneously
protect them from other predators. Damage caused by xolots to rodents is much
less than possible damage other predators can cause to these rodents. Thus,
between predatory xolots and their neighbours the certain biological balance
is observed.
Common xolot is a medium-sized animal: its length including tail reaches 60-70
cm. The fur of this animal is short, rich and velvety, that interferes with
its dirtying with ground and sand. Colouring of fur is light enough – usually
it has bright sandy color, but there are individual deviations from grey and
white up to red and yellow color. Paws of animal are strong, rather short and
slightly bent. Xolot runs in its burrows and if necessary can easily dig out
its own holes or expand already existing ones. The body of this animal is extended,
and ears are rather big and wide, that is obviously atypical for burrowing animal.
Anatomic feature of xolots as of separate genus is the presence in their ear
of additional skinny membrane which simultaneously improves hearing and protects
ear ducts from the ingress of sand and ground. Besides the auricle at underground
movement turns back so that dust did not get into acoustic duct. Sight at xolots
is rather weak, but the hearing and sense of smell are perfectly advanced. At
night xolots go to the ground surface and feed on small animals and berries.
They usually get roots and tubers during the tunnel digging.
Xolots live in small colonies numbering up to 10-15 individuals, consisting
of breeding couple and their posterity. As a rule, males remain in group, and
females leave it to settle in other place when reaching sexual maturity. They
prefer to move to other colonies, using holes of rodents. These animals are
afraid of bright sunlight, and if they appear on ground surface during the resettlement,
they try to find any casual shelter.
The female brings posterity once a year. To give birth it arranges shelter in
deep and well protected hole. Litter in shelter is made of rests of skins of
prey or flocks of furs; the female adds to it a little portion of grass. For
disinfection the female brings into the hole bunches of wormwood greenery and
other odorous grasses, in order to get rid of parasites. In litter it may be
up to 6-9 pups. They remain in shelter during first two months, gradually surveying
holes in vicinities of their home. Young animals become sexually mature at the
age of one and half years. Life expectancy is about 15-16 years.
In foothills of Rocky Mountains other two species of xolot live – nimble
xolot (Anconocyon alacer) and mantled
xolot (Anconocyon stragulumornatus), distinguished in smaller size
and darker colouring. Males of mantled xolot have clearly expressed shabrack
of reddish-grey color on back; females lack of it. As against to others xolots,
sight at mantled xolot is better advanced, rather than at other representatives
of genus.
At Pacific coast of North America false
xolot (Subterracyon occidentalis) lives. It is also the coyote descendant,
but it evolved independently of “true” xolots. False xolot is a bright example
of convergent evolution. As against “true” xolots, false xolot has rather small
ears, an additional membrane in ear is absent, and for protection against ingress
of ground the auricle can fold lengthways with the help of special muscle. This
species is a predator and builds holes independently, not using rodent colonies
for the purposes. This species has strong forepaws on which the second and third
fingers are armed with larger claws, than the others.
This species of mammals was discovered by Odin, the forum member.
Kurī
hou (Notokami pseudolupus)
Order: Carnivores (Carnivora)
Family: Dogs (Canidae)
Habitat: New Zealand, North and South Islands, most vegetated habitats, including
mountain slopes, grassland, forest and marshes.

Picture by Cossus
The domestic dog (Canis lupus domesticus) has been a constant
companion and loyal friend of man, used for companionship, food, war and hard
work. Ancient Maoris brought dingo-like domestic dogs with them to New Zealand,
the Kurī, which later became extinct in modern times. White man’s coming to
New Zealand brought farm dogs, pet dogs, and large dogs used for hunting pigs.
As the Holocene ended and man left these animals to run wild, they became a
more homogenous, dingo-like mongrel again, and eventually evolved smaller size
due to their island habitat.
The result is the Kurī hou (Maori for “new dog”), a creature partway resembling
a dingo, and also a small wolf. Height at the shoulder is only 60 centimetres,
its legs are shorter proportionately than a wolf or dingo, head is shorter and
its tail is bushy and curls upward a little, traits derived from its domestic
ancestry, weight is up to 31 kilograms. Colour is a general tawny brown, with
a dark brown back, the end of the tail and face are darker, belly and underside
are whitish. Populations found on the North Island are darker, and melanism
is relatively common there.
This dog lives in packs, similarly to a wolf or dingo, that consist of a mated
pair and their grown offspring, as many as 8 individuals, the older animals
(including parents) are usually the dominant. Lone individuals consist of grown
young which have been pushed out of the pack to start one of their own, but
offspring may stay with the family pack for as long as 3 years. As with wolves,
they establish a large hunting territory to maintain a good supply of food,
prey hunted as a pack may consist of large birds such as pheasant or geese,
rabbits, and various fair sized ungulates including certain kinds of deer and
sheep. Smaller prey such as rodents, lizards and insects are also taken, as
well as carrion and fallen fruit such as blackberries. These dogs proclaim their
territory by howling communally, but as with the dingo, they are unable to bark,
scent marking is also used. Other vocalizations include yips, yelps, whines
and growls.
Kurī hou are monogamous, and a single mated pair will remain together for life,
sexual maturity is reached at 2 years. Oestrus and rut begins in spring, such
as September or October, and lasts for 3 weeks. The female constructs a large
comfortable den, which is usually dug in lee of a tree root or large rock for
protection, and a convenient distance from water-sources. Gestation is up to
70 days, and litters consist of up to four young, which are nursed in the den,
leaving it first after 3 weeks and being very mobile at 6 weeks. Pups start
to eat solid food regurgitated by the mother after four weeks, and during initial
denning time, father returns to regurgitate food for the mother. Pups begin
being able to hunt with their parents after 3 months. Lifespan is up to 7 years
but usually shorter.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
New
Zealand False Fox (Vulpecanis aotearoae)
Order: Carnivores (Carnivora)
Family: Dogs (Canidae)
Habitat: North and South Islands of New Zealand, forest, woods, scrub, bushland,
foothills.

Picture by Cossus
Unlike in Australia, foxes were never established as an invasive
animal in New Zealand, but the descendants of the run-wild domestic dogs have
prevailed there in the Neocene, producing convergent fox-like forms.
The New Zealand False Fox is descended from the domestic dog of the Holocene
human occupation, but has adapted towards a more fox like shape and size. This
animal has an elongate body and relatively short limbs, like a fox, and a mid-length
bushy tail. Fur colour is a light reddish brown, with the back being slightly
darker, underbelly and cheeks are whitish, and extremities of limbs and tail
are dark brown. False Foxes reach 35 centimetres tall at the shoulder, with
a body length of up to 48 centimetres, and a 28 cm long tail, weight is up to
7 kilograms. Fur is denser, fluffier and darker in colour, in the southern part
of its range where climate is colder. The snout is somewhat more elongate proportionately
than its ancestor and the face is fairly broad, the molars are a little broader
to accommodate an omnivorous diet, ears are large and pointed.
Diet is that of an omnivore, including birds, fish, lizards and rodents, insects,
fallen fruit and nuts, roots, mushrooms and carrion.
Territories are shared by family groups, and males will mark their territorial
boundaries with urine. Within any one family there may be subordinate individuals
sharing the territory. Individuals dig dens to shelter in, or use the abandoned
holes of other animals, dens are also used for raising cubs. Calls vary from
a loud earnest barking call to communicate over long distances, to whining and
snickering noises when communicating among themselves at close range. Unlike
its ancestor, they are able to sometimes climb trees to escape predators.
Mating occurs in the spring, females have their oestrus in September or October.
Males will mate with the female several times during the 3 week receptive period,
mating is generally monogamous but litters with multiple sires do occur. Litters
consist of between 4 and 7 cubs, which are born with their eyes closed and are
an overall dark brown. Mothers stay in the den with the kits for up to 2 weeks
until they open their eyes and start to move about, nursing lasts up to 7 weeks.
Young resemble their parents after about 7 months, reaching sexual maturity
at about 10 months to a year old. Life expectancy is up to 5 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Tsitsu,
the wild hound (Neocanis tsitsu)
Order: Carnivors (Carnivora)
Family: Canids (Canidae)
Habitat: steppe areas of Europe.

Picture by Wovoka
Initial picture by Lambert |
Descendants of coyote (Canis latrans) have given rise to numerous
Neocene predators of both Americas. But right at the beginning of Neocene congelation
coyotes had penetrated into Eurasia through Beringia. At the end of Holocene
only Bering Strait prevented coyotes to begin a gain of Eurasia, and at early
stages of congelation when the ocean level had gone down, but Beringia had not
completely disappeared yet under glacier shield, coyotes realized the opportunity
opened to them and had intruded Old World from the east. As against their American
relatives, the destiny of Euroasian coyote descendants was changeable. Having
reached short-term success during Neocene congelation when they had widespreaded
practically across the whole Eurasia, after climate change they were in the
majority superseded by descendants of local predators and partly newcomers from
the south. But coyote descendants had confidently reserved for themselves one
climatic zone of Eurasia – steppes of Southern Europe and Three-Rivers-Land
where wild hound tsitsu lives, a swift-footed canid species of gracile constitution.
In steppes of Eurasia tsitsu occupies the ecological niche similar to occupied
by African hunting dogs (Lycaon pictus) in African savannas at the end of Holocene.
If to be exact, tsitsu is the coyote descendant not in full degree: they descend
not only from it. Hybridization as a way of speciation is rare phenomenon in
nature, but it also takes place. The majority of Holocene representatives of
Canis genus (except for black-backed and side-striped jackals) had identical
number of chromosomes and was able to interbreeding (interspecific hybridization).
Tsitsu has polyphyletic origin. The basis of tsitsu ancestral line is made of
Alaskan coyote (Canis latrans incolatus) descendants, but among tsitsu ancestors
there were also feral descendants of domestic dog (C. familiaris), and also
descendants of Asian jackal (C. aureus).
Tsitsu is highly social pack hunters. The average number of individuals in one
tsitsu pack changes from 70 up to 90 individuals (not including pups), the record
number of individuals in pack can make 146 adult individuals. Each pack controls
an area of approximately from one and half to two thousand square kilometers.
There are two subspecies of tsitsu, one (N. t. europaeus) of which lives in
Southern Europe, and another (N. t. rufus) inhabits Three-Rivers-Land. Subspecies
differ in colouration. N. t. rufus has “standard coyote” monophonic reddish
color while yellowish-red skin of N. t. europaeus is speckled with spots and
small strips of white and grey colors which help this animal to be imperceptible
in grass. In winter the wool turns denser and lighter a little. Tail is reddish
with black tip; its position is used by these canids for communication and expression
of emotional condition. Ears are big and pointed; paws are narrow. Canines are
rather long, much longer than at Holocene coyote.

Picture by Wovoka
Tsitsu is medium-sized animal. In dimensions it approximately
corresponds to polar subspecies of Holocene coyote. Weight of males makes about
20 kg, females are about 16 – 17 kg. Despite of the dimensions, packs of swift-footed
(developing speed up to 75 - 80 kms per hour in jerk and able to hold speed
of 60 kms per hour for a long time) tsitsu easily hunt animals like harelopes
and porcippulas. Also tsitsu actively compete to
other species of carnivores, for example, with zibetonyx.
Due to superiority in strength they frequently take off prey of other predators.
Even large predators finding themselves in position “two or three against hundred”
are compelled to concede the prey to aggressively attacking tsitsu pack.
Similarly to Holocene hunting dogs tsitsu nurse even old and ill individuals,
bringing food to them. The pack lives in the wast territory, but in the evening
after hunting adult individuals hasten to shelter – colony numbering up to several
tens of holes dug in soft ground. These dogs rear young growth in holes, and
individuals do not take part in hunting – old or injured animals – look after
posterity. For communication at the large distances tsitsu uses sounds. The
sound repertoir of tsitsu is diverse and includes howl and bark of varying tonality
and duration, and also yelping and growl. A characteristic sound of night steppe
is thin shrilly howling of the whole tsitsu pack.
Speed used in hunting has the return side – the organism of fast runner is used
up in greater degree. Average life expectancy of tsitsu is about 15 years, but
usually it is much less.
This species of mammals was discovered by Odin, the forum member.
Chootie (Chuti chuti)
Order: Carnivors (Carnivora)
Family: Canids (Canidae)
Habitat: salt swamps of Mediterranean lowlands.

Picture by Cossus
Initial image by Odin, the forum member |
For many mammals hot salt swamps have arisen in saline desert
replaced former Mediterranean Sea are the most improper place for life. Any
unadapted animal casually got in these places is doomed for death within several
days. In border areas of Mediterranean swamps frequently there are corpses animal,
lost from thirst and overheat. Salt can keep their bodies for long years, impregnating
them when wind wears out the dead animals with salty sand and dust. But some
mammals could adapt to these severe conditions, and there are even species prospering
in this inhospitable environment. Usually rodents make the majority among champions
on survival, but representatives of predatory mammals also live in severe desert
at the border between Europe and Africa. They are descendants of the fennec
fox (Vulpes zerda), the small big-eared fox. This tiny Holocene canid, at which
the significant part of diet was made with insects, was perfectly adapted to
survival in extreme conditions of critical shortage of water resources. Holocene
fennec foxes could live without water for some days even in conditions of heated
deserts of Holocene North Africa. Therefore right at the beginning of Neocene
descendants of fennec fox could colonize droughty open spaces of Mediterranean
lowland while the rests of sea still existed in hollow between continents. In
due course descendants of fennec fox turned even smaller and passed to completely
insectivorous diet, having formed a separate fennecine subfamily (Fennecinae)
within canid family. The fennecine subfamily includes one genus Chuti and two
species – chootie and adjule. Chootie is a typical species of genus. It is a
predator of very unusual shape: a tiny mammal which length does not exceed 12
cm including tail. Thus ears of this one have kept the same proportional size
relatively to body size, which was at fennec fox, its far ancestor. Running
chootie from apart looks like any rodent, and its belonging to predators can
be defined only at more attentive survey.
Chootie has rather short legs in comparison with its ancestor, and shorter tail.
Loss of water and overheating are the basic dangers at life in salt desert.
Chootie escapes from overheat with the help of wide auricles penetrated with
blood vessels and helping to radiate superfluous heat. Its wool is dense and
velvety despite of a hot climate in its habitat. Such wool provides protection
from overheat to skin of animal. Colouring of chootie is camouflaging – reddish
or yellowish. In the populations living in saline soils of Mediterranean hollow,
white and light grey color variations are most usual. Chootie avoids loss of
water due to habit of life and features of physiology. This animal is active
at twilight hours and in dawn time, spends its day in one of regularly visited
shelters at its territory, and sleeps within some hours at night in temporary
shelters. Kidneys of this animal work very effectively, and urine has very high
concentration of nitrogenous substances.
Chootie is extreme cautious mammal relying on sharp hearing and keen sense of
smell in detection of danger. Out of breeding period lonely chooties constantly
migrate from one small source of fresh water to another. Moreover chootie receive
a significant part of moisture from food. The basis of chootie diet is made
of insects and other small invertebrates. The liver of animal effectively destroys
some poisons of organic origin, therefore chootie can eat poisonous beetles,
millipeds and spiders entirely. Chooties become prey of reptiles and birds of
prey. Despite of adaptations to life in desert, chootie always faces real danger
to die from dehydration. In dust storm animals may simply choke with salty dust
if the shelter can not protect them.
The care of posterity at this species is original. At males long fur jabot grows
on breast soon after pairing. The wool in this jabot has one feature – at contact
to water it is capable to accumulate and keep moisture in significant amount.
While the female feeds cubs, male searches for water. At night stones in desert
quickly cool down, and to morning plentiful dew accumulates on them. Male literally
wallows on surface of stone and creeps on it on belly, carefully absorbing dew
by wool. When he comes back to the female and cubs, they lick off this moisture
from fur jabot, receiving thus the additional portion of water. Within one year
the female has time to draw up to three litters of 4 – 5 cubs in each. Like
all animals of small size, chootie grows quickly: the month-aged cub already
reaches the size of adult individual and becomes independent. And five month
old animals can raise their first litter. More successfully chootie breeds in
winter and in the spring when from the Alpes and Atlas Mountains streams flow
down to the Mediterranean hollow, and air is damper. In summer animals do not
breed, and if the female is pregnant, embryos are “conserved” at the early stage
of development, or simply resolve.
Life expectancy of chootie makes about 7-8 years.

Also in eastern part of Mediterranean swamps and at the territory of former coast of Asia Minor larger species lives – adjule (Chuti adjule). Its adult males reach the size up to 20 cm. It has similar biology, but can attack small reptiles occasionally. This species always keeps in pairs and lives near to constantly existing sources of water. Colouring of wool is grey with black strip along the back.
This species of mammals is discovered by Odin, the forum member.
Anteater Fox (Neootocyon insectophagus)
Order: Carnivores (Carnivora)
Family: Canids (Canidae)
Habitat: southern and eastern Africa, savannas and woodlands.

Picture by Alexander Smyslov
The border of Neocene and Holocene epochs was marked with
the mass extinction of various animal species and even familles. That included
the extinction of
Proteles cristatus, unusual predator from southern and north-eastern Africa,
who was almost completely insectivorous. The human intervetion and new rivalring
species caused the extinction of that insectivorous hyena. But that environmental
niche hasn't stayed vacant for too long, as there were several applicants to
fill it: in the North Africa it was grumbling
ant-mungo from the family closely related to civets, while in the south
it was the representative of another carnivorous family – the bigear fox (Otocyon
megalotis) descendant – anteater
fox.
It's a middle sized animal with thick fur (size varies from 80 cm to 1 m, weight
- 6 -10 kg) and relatively long legs which is typical for most open environment
animals. Thick fur saves the animal from the ants and termites' "soldiers" bites.
Thick and shaggy fur of that animal has relatively monotone brown colour, with
just the ears' tips being a bit darker. Nose, chest and neck have extra protection
from insects, spiders and scorpions bites- unusualy dense skin. The vulnerable
nose has extra protection- a small piece of cornual "armour".Unlike
another 'anteaters' (Holocene true anteaters from Edentates Order and pangolines,
Neocene echidnas, lizards and wormtonguers)
its teeth system isn't reduced- it has plenty of sharp small teeth, resembling
insectivors teeth a lot. Being
very careful, anteater fox hunts only in twilight and in the night. In the
day anteater foxes rest in selfmade holes or in the other animals dens. Very
often they share their dens with armor-headed
wart hogs for extra protection.
The sensitive ears of anteater fox are inclined a bit forward and down; keen
hearing helps it successfully find insects even in deep darkness. Its paws
aren't specialized simply for breaking termitarys, so the fox has to vary its
diet, adding another invertebtates (beetles, spiders or ants) into it. Also
its diet includes eggs and small vertebrates. One of the main alternate food
sourses are locusts, who have survived all the ecological disasters on the
border of Holocene and Noeocene. Its invasions are comparable to lemming invasions,
and as regular in Neocene as it was in Holocene. To the anteater fox, it's
just like lemming invasion to polar fox- it "forgets" about hunting
in twilight only and hunts for the locust even in the daylight.
Just like their ancestors, anteater fox are monogamous. That is explained by
the diet pecularities- they hunt for small preys so the nessesary population
density just can't be reached. The female gives birth to 3-7 cubs but not all
of them reach senior age, especially in dry lean years, when there's little
food and all savannah inhabitants, including large predators, are starving.
The maximal lifetime of anteater fox is 10-15 years.
In the northeastern Africa, where the insectivorous enviromental niche is occupied
by other animals, lives the anteater fox relative - coastal fox (Neootocyon
oraannona). Its diet includes various seafood- shellfishes, dead fish,
mollusks,which they gather during the falling tide. Unlike its southern relative,
coastal
fox body is more "foxy"- it has shorter legs and smaller ears. The
lower part of coastal fox' acoustic duct is protected by skinny valve to prevent
water from coming into ears. The fur is short and hydrofuge (especially on
lower body half). Despite living at the seashore, coastal fox is not a good
swimmer. It prefers to search for its food on the littoral fringe, but sometimes
comes to feed before the falling tide ends or stays on the littoral fringe
till the rising tide starts. The fur of coastal fox is more light-colored that
the one of anteatrer fox- it's yellow with grey shades. The fingers on the
paws are mobile and broad-placed, which helps the coastal fox to run on the
sand with ease as well as hold its preys. The coastal fox is very careful animal,
who is very tolerant to its relatives. Several couples of those anmals can
feed on the littoral fringe simultaneously without disturbing each other, and
fights rarely happen during the feeding.
The breeding and home life resembles those of an anteater fox. Coastal fox
makes its dens at coastal rocks or at the bushes (higher that the highest rising
tide mark). The maximal lifespan is around 12 years.
These species are discovered
by Bhut ,the forum member.
Translated by Charles.
Lynx
fox (Megacyon feliformis)
Order: Carnivors (Carnivora)
Family: Canids (Canidae)
Habitat: North America, forests of temperate zone.

Picture by Alexander Smyslov
As against larger wild dogs, such as wolves and African hunting
dogs, smaller canid representatives had quite safely gone through both anthropogenous
pressure, and climatic changes of the boundary of Holocene and Neocene. These
animals – jackals, coyotes, foxes and even small varieties of feral domestic
dogs – were successfully fixed in ecological niches of small and medium-sized
predators, having caused new species in due course. So, in Siberia waheela,
the descendant of polar fox, had evolved and in South America the feral representatives
of domestic dogs became wild pack predators again, despite of approximately
16 – 20 thousand years of life near people.
In North America lupardus is one of the largest
and widespread wild canids became ecological analogue of cheetahs, running felid
representatives. It lives in prairies, but in areas where open spaces are replaced
with forests and groves, other canid species live – the descendants of red and
grey foxes.
The grey fox (Urocyon cinereoargenteus) lived in North and Central America to
the south from Canada, and had, as well as other foxes, good amount of hardiness
and ecological flexibility. In Neocene, despite of slightly increased pressure
from the side of more progressive descendants of red foxes and coyotes, the
grey fox quite safely evolved into new canid species – the lynx fox. In forests
it occupies the become empty ecological niche of Canadian lynx which had become
extinct in Holocene.
Lynx fox is very large animal in canid standards: large самец can reach height
of 130 cm rearing on hind legs of about 40 cm long. Weight of animal quite corresponds
to growth – large foxes may reach weight of 60 kg: as much, as Euroasian waheela
weighs. But, as against waheela, lynx fox can not change wool color in winter
and in summer: the year round this animal has silver-gray fur which appreciablly
turns white on the tip of tail. At adults males “collar” of pale-yellow fur
grows on neck. On back of these animals there is an indistinct spotty pattern.
It is clearly expressed at young individuals, and it helps them to escape from
predators, hiding among grass and foliage. In course of growth animals lose
this pattern, but at the southern borders of the area where more evergreen plants
grow and winter is snowless, up to a fourth part of individuals keeps spotty
colouring in adult condition, and separate populations include almost exclusively
spotty individuals.
The tail of this fox is long and thin – it serves as the balance weight at the
movement on trees. Carpal and talocrural joints of animal are mobile and allow
to hand and foot to turn on small angle – it helps lynx fox to climb on trees
better.
Muzzle of this animal is short and has large canines. However, canines serve
mainly for demonstration – the angry animal starts “yawning”, turning to the
opponent and widely opening mouth. Auricles are rather large, mobile, peaked,
and have small hair brushes on tips.
Despite of the growth and impressive canines, lynx fox has rather peaceful behaviour
and is not squeamish in feeding: it is omnivorous and in autumn, in the time
of fruiting of wild berries and trees it almost completely passes to vegetative
food. It eats the greatest amount of food of animal origin at the end of winter
and in early spring when stocks of vegetative forage accessible to this animal
are exhausted. At this time lynx fox can even eat carrion. It also has kept
abilities of ancestor to tree climbing and is quite capable to find prey and
shelter for rest on trees. Forage of animal origin of this animal includes various
animals and birds, but especial frequently it hunts hares, as well as its ecological
prototype – Canadian lynx. The fox is afraid only of few animals: berls,
huge mustelid representatives, and also of missopeho,
Neocene American saber-toothed cat. But due to its secretive twilight habit
of life and ability to tree climbing lynx fox almost does not face with these
predators.
As against others large canids, lynx foxes do not form family groups. Females
raise cubs independently; usually there are from 1 to 3 cubs in litter. They
meet males only during a courtship season. It takes place at the end of winter;
that time males arrange long duels, rising on hind legs. Also the winner male
pursues the female for some time, estimating her force and the relation to him.
Usually the female shows resistance only right at the beginning of courtship,
if the male is pleasant to her. Avoiding male, the female, on the contrary,
shows long resistance and tries to come nearer to other males which perceive
her persecutor as the contender and attack him.
Voice of lynx fox is long lingering bark which may be heard at the great distance.
With the help of voice animal declares rights to territory and calls the courtship
partner. Cubs have thin squeaky voice and can utter sounds similar to twittering.
Usual life expectancy of lynx fox is about 10 – 12 years.
This species of mammals was discovered by Bhut, the forum member.
Aoskiar
(Megacyon aoskiar)
Order: Carnivores (Carnivora)
Family: Canids (Canidae)
Habitat: deciduous forests and woodlands of North America.

Picture by Cossus
By the beginning of the Neocene representatives of North American
canids were represented for the most part by descendants of the coyote (Canis
latrans) of the Holocene, by predators of medium and large sizes. The only exceptions
are waheela, a descendant of
the arctic fox, and various descendants of grey fox (Urocyon cineroargenteus),
one of the most unusual canids of the Holocene. One of its descendants, the
lynx fox (Megacyon feliformis), has changed
relatively little and in many respects is similar to its ancestor, only changed
by its larger size. But another descendant of the gray fox, the aoskiar, is
markedly different in its appearance, as well as behavior.
The aoskiar is quite a large beast: the height of an adult at the shoulder is
about 90 cm, a body length of about five feet, and a tail length of about half
a meter. In its proportions this animal resembles a beagle dog, and only the
head, and especially the whiskers and ears, retain the features of a “portrait
likeness” to the fox’s ones. Its eyes are greenish-yellow, its eyebrows are
bushy and light colored, at a sexually mature male on the neck a thick “beard”
grows. The hair color is usually gray, almost monochromatic, lighter on the
belly. Northern populations of aoskiars differ in darker hair compared to southern
ones; sometimes some of them are melanistic. In southern populations “blondes”
having straw-yellow hair are occasionally found.
The aoskiar is a terrestrial carnivorous mammal that eats very little amount
of plant food, unlike the related lynx fox. It has weak jaws, not adapted for
chewing, but armed with teeth with sharp cutting edges. It is an active predator
that can hunt for a variety of prey. Its diet consists of small and medium-sized
rodents, lagomorphs, juvenile ungulates, terrestrial birds and their chicks,
frogs, small non-venomous snakes and lizards, various insects and carrion. Due
to the light constitution and good running abilities this animal is capable
of preying on the swiftest animals, driving them to exhaustion. In this regard,
the competitor of the aoskiar is the lupardus,
but these two species rarely compete as the aoskiar is larger and preys on larger
animals. In addition, the aoskiar prefers a “mosaic” landscape with thickets
of shrubs and trees, while the lupardus inhabits plains, overgrown with grassy.
Aoskiars themselves are often victims of waheelas, berls and other predators
which ravage their dens.
Aoskiars live alone or in pairs or small packs (3-8 individuals) whose members
are usually connected by family relationships. They hunt large prey, combining
hunting from an ambush with a running hunt – one animal drives prey towards
others, hidden in an ambush. Family life of aoskiars takes place in a den (usually
a wide burrow or a hole under the roots of a tree), where adults raise pups
(there are typically 2-4 puppies in litter). Both parents take care to puppies,
and sometimes they are helped by “nurse”, the grown up cub of the previous litter
or a subordinate female (usually the sister of dominant one).
Aoskiars are usually monogamous, but sometimes one male has two or three females.
In this case, offspring appear only at one dominant female. The struggle for
the establishment of domination relations occurs only if the females are unrelated
to each other. Courtship at aoskiars occurs in late winter, and the posterity
is born a few months later at the beginning of summer.
Aoskiars are cautious animals, when they meet with enemies they prefer not to
fight, but to hide in the forest or scrub. They enter an open confrontation
only when there is a threat to their offspring, and inflict deep bites on the
aggressor. The adaptation to rapid running largely affects the physiology of
animals: due to frequent exposure to intense physical stress the life expectancy
of an aoskiar is no more than 8 – 10 years.
This species of mammals was discovered by Bhut, the forum member.
Translated by FanboyPhilosopher.
African
tufted fox (Dromovulpes africanus)
Order: Carnivors (Carnivora)
Family: Canids (Canidae)
Habitat: North Africa, savannas and woodlands.
During Holocene epoch the common red fox (Vulpes vulpes) lived in northern hemisphere
everywhere from Europe through Asia up to North America, being one of the most
widespread smaller canids. Its well-being was directly connected to anthropogenous
pressure which had destroyed or strongly reduced number of larger predators,
having given to foxes more freedom for existence and having given new habitats.
Red fox existed successfully in landscapes changed by people that had allowed
it to keep large number and wide area.
But the situation had changed in Neocene. People had disappeared, and red foxes
had lost the advantages connected to development of anthropogenous landscapes.
Some waves of ice ages had thrown heat-loving foxes farther to equator therefore
this species of animals had disappeared in New World absolutely, and in Old
World it had conceded northern part of the area to polar foxes and to waheelas,
their descendants. But in new habitat – in Africa and southern Asia – foxes
had quite accustomed, had evolved to new species and even had returned to Europe
as it was made by grass fox,
solitary running predator of savannas and woodlands. But nevertheless it is
a rare case – the majority of its relatives lives in warmer parts of the world.
African tufted fox is a species related to grass fox, but distinguished from
it with a number of anatomy and behaviour features. First, it hunts not only
alone, but also in pairs. In this case one animal exhausts prey, and the second
one jumps out from an ambush for it and puts the final impact. Sometimes the
grown up cubs help thus to parents and at the same time study hunting methods.
However, as against the grass fox, tufted fox usually accelerates speed no more
than 18 – 20 kms per hour, but at the same time it has the great endurance and
is able to pursue prey for a long time. Moreover, rear legs are longer and stronger
a little bit than front ones, and it allows the fox to jump on stones and low-growing
trees having curved trunk in order to leave from a pursuit or to catch small
prey.
Second, tufted fox is colored conservatively enough. It has monophonic sandy
color (there are also darker individuals) with lighter belly and tail tip, and
also with dark “stockings” on legs. Males additionally grow a crest or a forelock
– a bunch of longer and rigid hair on head between ears, which condition shows
age, health and force of its owner. At females wool on head is only slightly
lengthened, and only at the strongest and most aggressive individuals testosterone
surplus results in formation of small hair cop.
Tufted fox is an omnivore. It usually eats carrion, and also various rodents
– down to cubs of huge grasscutter
rats. Attacking less dangerous animals, like kangoohoppers,
this animal can chase also healthy adults. Steppe
harelopes especially suffer from it – tufted foxes eats more than 20 % of
their young growth, despite of all attempts of parents to protect the posterity.
Usually hunting foxes separate young animal from herd and drive it in savanna,
not allowing it to have a rest. After one hour of such pursuit one of animals
snatches on it and kills, biting through the neck. Also tufted fox ravages nests
of small birds and eats various reptiles, but at the same time willingly eats
various ripe fruit and graminoids at the stage of milky ripeness.
At the same time tufted foxes are objects of hunting of larger predators. Crocoturtle
sometimes catches them at the watering place whereas barbed
herzogcats sometimes purposefully hunt these animals, considering them as
food competitors and danger to their posterity. Tufted fox prefers to avoid
furiobaboons and other monkeys
living in groups though sometimes the strong individual can kill the young monkey.
But in this case foxes quickly leave a place of hunting as the flight of monkeys
represents force which is insuperable for foxes.
Tufted foxes breed once a year, from December to January. Female’s heat lasts
for some weeks. The animal notifies on readiness for pairing by the shrill squeal
which is a little bit distinct from usual bark of these animals. The voice of
the female involves several males. They actively compete for the right of pairing;
showing the crests to each other in the beginning, and then rising on hind legs
and “boxing” by front legs to each other’s muzzle. The strongest and hardiest
animal wins, as a rule. If male has found the female in loneliness, pairing
usually begins after long and persevering courtship when male cajoles the female
actively, and sings the “serenade” to her, uttering the sounds similar to grumble
and cooing.
In some weeks after pairing the female brings posterity – from 5 up to 10 cubs.
They are born small, almost blind and covered with white-yellow fur, but within
several weeks at them eyes open and teeth start to appear. At bi-monthly age
cubs leave shelter and start to learn the world around. At the age of half-year
they are already capable to follow parents for hunting, and by the end of the
first year of life they are already quite independent. At the second year of
life the female brings posterity the first time. General life expectancy of
tufted foxes is from 8 up to 13 years.
This species of mammals was discovered by Bhut, the forum member.
Giant tanooka (Tanuka gigantea)
Order: Carnivors (Carnivora)
Family: Bear-like dogs (Prolesnyctereuteidae)
Habitat: Siberia, Altai, Northern Europe.

Picture by Amplion
At the boundary of Holocene and Neocene the mass extinction
took place. It had influence at plenty of animals and was caused by human activity
and reduction of territories, suitable for existence of normal populations of
large animals. Bears also had died out. In Neocene animals of other families
started to occupy an ecological niche of bears. In Southern Asia their place
was occupied by viverrid descendants and
in Central and East Europe by bearaccoons,
descendants of raccoons. At the north of Holarctic niche of bears was occupied
by large mustelids, berls, and also by
animals of new Neocene family of carnivores – bear-like dogs (Prolesnyctereuteidae)
separated from true canids. An ancestor of this family was a raccoon dog (Nyctereutes
procyonoides), medium-sized unpretentious omnivorous canid. Eventually descendants
of raccoon dogs had grown larger. Similarly to their ancestors, raccoon dogs
(and also similarly to bears), the majority of bear-like dogs run into hibernation;
this feature has allowed them to settle at the north of Eurasia remarkable in
severe snowy winter. The family includes one genus (Tanuka) and two species.
The largest species is giant tanooka. The weight of females usually reaches
500 kg, males – up to 600 kg; some individuals in northern part of area can
grow to 800 kg. Thus, giant tanooka can compete to South African awfulest
marafil for the right to be named as “the largest representative of carnivores
order in Neocene”.
By appearance giant tanooka resembles a hybrid between bear and fossil predator
Amphicyon. Because of rather short fingers and toes animal looks almost plantigrade.
Fur of tanooka is brownish, spotted with numerous albesent and black spots on
sides, shoulders and hips. Animal has very big claws, but, similarly to the
ancestors, raccoon dogs, tanooka can not climb up trees. This animal runs slowly,
but can make far passages for food within the borders of its territory.
Similarly to Holocene bears, giant tanooka is an omnivore actively consuming
both vegetative and animal food. In spite of the fact that giant tanooka is
a predator nevertheless, it prefers not to hunt itself, but to play a role of
scavenger or to take off prey of other predators. Giant tanookas very often
track down Siberian sabertoothes,
but not as prey, but as original “suppliers” of food. After their successful
hunting tanooka eats up numerous uneaten rests of their prey. Frequently (especially
in autumn, right before hibernation) giant tanookas attack waheela
packs, taking off their prey. If the autumn was poor in food, tanooka trying
to take prey may risk and attack even the lonely berl. As against the berl,
giant tanooka is active only in rather warm season. In winter, similarly to
its ancestor, raccoon dog, giant tanooka runs into hibernation. In spite of
the fact that giant tanooka is heavier than berl, it is not a dominant predator
because of three factors: first, its population is extremely rarefied (the average
territory controlled by one individual is about 2 – 3 thousand square kilometers),
second, in winter it runs into hibernation, and, thirdly, tanooka is more omnivorous
scavenger, rather than active predator. Besides in summer and in autumn more
than 60 % of its diet the vegetable food makes. The main contender of giant
tanooka is berl. Tanooka does not bear the presence of berl in its territory
and relates to it agressively: it will try to banish berl, using the size and
force, and also to kill it if the opportunity will appear. In a case if tanooka
meets berl female with cubs, it will try to kill cubs by all means, despite
of all attempts of berl female to protect them. In such situation tanooka will
attack berl cubs and female protecting them, despite of repulse which large
predator can render it. In winter the situation frequently changes to opposite
one. From time to time berls attack tanookas in hibernation condition right
in their dens. To tell the truth, it finishes with berl victory not always.
Winter sleeping of tanooka is not too deep, more similar to bear ibernation.
If tanooka will have time to wake up during such attack, it will kill or will
seriously cripple the attacking berl most likely.
Because of great rarefaction of populations at giant tanooka the original courtship
behaviour had evolved. Closer to approach of courtship season tanooka males
actively mark borders of their territory with extremely odorous labels. To breeding
season urine of tanooka males gets very proof and pungent smell which is gradually
spreaded to many tens kilometers, and the marks left in winter continue intensive
smelling even at the beginning of summer. Females find males using smell due
to very keen sense of smell. Heat proceeds intensively at the female, and the
female stays ready to pairing within several days in succession. It is enough
to her to find a male ready to pairing. Heat at any individual tanooka female
happens alternate years, at the end of winter.
Pregnancy lasts within about three months. The female gives rise up to five
cubs, but usually it happens three or four of them. From the big litter only
two cubs may survive to approach of independence, and per lean years only one
may stay alive. To autumn young animals reach weight 90 – 100 kg. They will
spend the first winter of their life with mother, and young females sometimes
remain with her for the second winter and leave mother when new heat starts
at her.
Life expectancy of giant tanooka is about 45 – 50 years.

Рисунок Александра Смыслова
At the Far East and in mountains of Central Asia this species is replaced by mountain tanooka (Tanuka monticola). It differs in smaller size (weight of adult individual does not exceed 350 kg), richer winter fur, which is colored lighter, rather then summer one, and annual breeding. This species is more herbivorous (the vegetative food makes up to 70% of a diet), and prefers small vertebrates, insects and carrion from food of animal origin. Mountain tanooka is more mobile, easily swarms up abrupt slopes and walks on narrow mountain tracks.
This species of mammals was discovered by Odin, the forum member.
Flag-tailed
pampanasua (Pampanasua vexillicauda)
Order: Carnivors (Carnivora)
Family: Raccoons (Procyonidae)
Habitat: pampas of South America.

Picture by Timothy Donald Morris, colorization by Biolog
Initial picture by Timothy Donald Morris |
In Neocene Atlantic Ocean had continued to extend, and, as
if a wedge, had separated North and South Americas, having broken off isthmus
of Panama. South America has moved little bit closer to South Pole, and it has
resulted in expansion of temperate climate area and pampas – plain areas covered
with grassy vegetation. In pampas herds of large animals wander – huge tapirotheriums,
descendants of tapirs little bit similar to elephants, deermaras
and giant paca – cursorial
caviomorph rodents. They are pursued by hunting
heron, large flightless bird, and also by acatou
– the heaviest flying bird of the world. Among smaller animals inhabiting pampas,
rodents, armadilloes (including rather large species) and numerous birds are
characteristic. Among South American inhabitants of pampas monkeys are lack
– not so numerous ground New World monkeys prefer to stay in woody regions and
bushes. Original pampas analogue of baboons and other ground monkeys of Old
World is the representative of raccoon family adapted to life in grassy plains
– flag-tailed pampanasua. It is the descendant of one species of coatimundi
(Nasua), successfully adapted to life in forestless areas after disappearance
of significant part of South American forests as a result of human activity.
Compared to its ancestors, pampanasua has changed in significant degree. It
is long-legged animal of gracile constitution about 60 cm high at a shoulder;
body length of adult individual is up to 70 cm, not including tail. The body
of pampanasua is covered with rusty-red wool with small dark spots on back and
sides. Muzzle is long and has a mobile nose and well advanced olfactory chambers.
Due to keen sense of smell pampanasua easily finds insect larvae and edible
parts of plants under ground at the depth of about 20 cm.
Fingers of animal are short, but mobile, and claws are blunt: this animal swarms
up trees worse, rather than its ancestor, and spends almost whole life in open
areas where it moves on four paws more often. In order to look around pampanasua
can rear on hind legs and keep vertical position within several minutes. For
recognition of relatives this animal has contrast marks on muzzle: forward part
of muzzle is white with black “glasses” around of eyes. The size of these “glasses”
depends on testosterone content in blood, and at dominant males they are especially
large. The size of marks allows quickly establishing of hierarchical relations,
not resorting to combat. Males have long canines which tips stick out of the
closed mouth. Defending itself from the enemy, pampanasua puts very painful
bites.
Ears of pampanasua are long and have white hair brushes on tips. Auricles serve
for removal of superfluous heat from organism.
Pampanasua lives the groups numbering up to 20 members – males and females of
various ages. Animals communicate with each other with the help of various sounds
– hissing, snorting, cooing and grunting. For communication with congeners at
the long distance this species uses tail reaching 1 meter length. In the basis
of tail pale cross-sriped pattern is visible, and on the tip the one-sided brush
of lengthened hair of white color is developed; tail tip is black. In rest animal
holds tail vertically and its movements are smooth. Quivering tail is the feature
of alert animal. Usually animals signal this way to each other about snake found
out. Pampanasua submits alarm signal in the voice – it is a series of abrupt
shrill sounds similar to bark of small dog.
Usually pampanasua moves on the ground, but also can clamber on trees. The main
enemies of these animals are hunting herons. Pampanasua hides from them, dropping
to the ground and drawing tail in, or getting on trees. The group can frighten
off a medium-sized single predator with menacing demonstrations.
Pampanasua is omnivorous and equally willingly eats food of vegetative and animal
origin. Animals dig out tubers and bulbs, eat sapful bases of leaves of grasses,
young shoots of bushes. Among food of animal origin pampanasua prefers insects,
small reptiles and nestlings of ground birds. Frequently the group of pampanasua
keeps near to large rodents and tapirotheres, and catch near them frightened
insects and small reptiles. Due to keen sight and good hearing pampanasua right
in time distinguishes approaching of enemies and warns large animals of their
occurrence.
Twice per one year the female gives rise to one or two cubs. As this species
leads migrating habit of life and does not build long-term shelters, the female
must carry her posterity herself. The cub is born blind and covered with short
wool. Right after birth it is able to cling to wool of mother and to twist her
tail with its one. Due to it cub does not fall on the ground even if the female
darts off sharply. Cub’s eyes open at week age, and at the age of one month
young animals spend a significant part of time on the ground, getting on mother’s
body only at the alarm signal. At one-year-old age young animal leads independent
life. Males usually stay in parental clan, and females pass from one clan to
another.
Sexual maturity comes at the age of two years, and life expectancy reaches 25
– 30 years.
Ozomatli (Procyolemur ozomatli)
Order: Carnivores (Carnivora)
Family: Raccoon lemuroids (Procyolemuridae)
Habitat: North America, evergreen deciduous woods at the coast of Gulf of Mexico
and in Florida.
In Holocene representatives of primates order had gradually lost positions
occupied in prehistoric time. Only one primate – man – had achieved fantastic
power and prosperity at the expense of all ecosystem of planet. And only in
some places unique species of small monkeys could coexist with people. Other
primates became victims of hunting and human initiated change of natural environment.
And once this vicious regime had failed: the mankind had disappeared, having
fall a victim of natural laws which acted this species the same order as other
ones. Stress, illnesses and adverse heredity had undermined reproduction of
mankind as the species, and people had disappeared till some millenniums when
some waves of global epidemics had swept across the world. Taken together with
people, the part of primate species had died out: they are also subject to
human illnesses. In addition the ice age rendered large influence to the number
of primates, having reduced areas of suitable for them habitats. In Neocene
when the situation had stabilized, some ecological niches earlier belonging
to primates appeared occupied by absolutely other creatures.
One of such animals lives in woods along the coast of Gulf of Mexico. This
creature resembles a little the monkey by shape, for what it has received the
name “ozomatli” (in Aztec language it means “monkey”). Ozomatli dexterously
swarms up trees, using paws with well advanced fingers. When ozomatli yawns,
in its jaws six incisors instead of four ones characteristic for primates,
are clearly visible. It unequivocally indicates the family tree of the present
creature: ozomatli is predator very specialized to climbing habit of life,
the descendant of animal cacomistle (Bassaricus astutus), belonging to raccoons
family (Procyonidae).
Ozomatli is rather large creature: the adult male weights more than 20 kg,
the female – about 15 kg. By separate features of appearance ozomatli resembles
any monkey, but it has features, not characteristic for primates: on paws short,
but sharp claws grow instead of nails. By proportions ozomatli resembles long-legged
lemur: its head is rounded with large ears, body is lengthened, and mobile
tail approximately one and half time exceeds the body length. Paws have sharp
claws and well advanced fingers.
Rear legs are a little longer than front ones, due to what ozomatli can skip
from branch to branch. At this animal feature of structure of extremities,
more characteristic for primates, has developed: this animal does not cling
to branches by claws, and clasps them by fingers and toes. To strengthen durability
of clench, palms and feet of ozomatli are covered with wrinkled skin forming
a semblance of papillar combs characteristic for primates.
At ozomatli there is short and rather wide muzzle: animal eats mainly vegetative
food and small animals, and the part of molars had reduced. But force of bite
has appreciably increased, that permits ozomatli to chew firm seeds and to crack
nuts. Canines of this animal are also shorter in comparison with other predators.
Wool of ozomatli is short and rather thin: this creature prefers areas of warm
and damp climate, not living in woodless areas. This animal has light brown colouring
with thin cross black strips on back. Colouring of tail sharply contrasts to
the body: it is light-yellow with black-and-white hairy brush of long hair on
the tip. This hairy brush serves to the animal for communication with congeners:
ozomatlies, as a rule, are silent, and only in case of danger they utter shrilly
cry of alarm. Animals communicate with each other by means of tail movements.
Thus the hairy brush is appreciable from apart, and permits animals to not give
out themselves by cries.
Ears of ozomatli are rather wide, peaked and sticking in sides – they provide
not only excellent hearing, but also protection from overheat in warm climate.
Eyes of this animal are also big and shifted in forward part of scull. As the
muzzle of animal is short, such position of eyes provides to it three-dimensional
sight. Due to this feature ozomatli is able to jump in branches dexterously,
covering the distance up to 5 – 6 meters and more. This animal is active in day
time, as against to the nocturnal ancestor.
Ozomatli lives in deciduous woods at the southern coast of North America. It
does not avoid swamp forests of Florida and Mexican coast, but does not live
in woodless districts and light forests. This species is omnivorous and inclined
to vegetarianism. The basic food of ozomatli includes fruits, seeds and nuts
of various trees. Also this animal eats juicy stalks of orchids and other plants,
and tree leaflets. It supplements the diet with invertebrates – snails, spiders
and insects which this animal gathers in tree crones. Ozomatlies almost do not
go down on the ground, preferring to travel in crones. These creatures keep in
groups of several breeding pairs of approximately equal age, among which one
pair has leading position. Pairs at ozomatli form to all life. Partners constantly
care about each other, exchanging marks of attention: they clean each other,
constantly keep beside and exchange by silent cooing sounds with each other.
Because this animal lives in districts where change of seasons is almost not
expressed, animals having posterity may be met at any time of year. Two times
per year the female brings posterity: one large cub covered with wool. The cub
is born blind and with closed ears, and till first days of life it can only cling
to mother’s wool. It holds for wool of female, and any time she holds the cub
by one paw during jumps. At three-day age the cub starts to see and to hear,
and its grasp becomes stronger. A week later it “settles” on mother’s back and
starts to play with other cubs. Young ozomatlies keep near to parents almost
before the birth of next cub: shortly before its birth female banishes grown
up posterity, and young animals start to lead independent life. As a rule, they
leave parental clan, joining other, small group, or form groups of same aged
animals. Such groups differ in increased aggression, and can even supersede adult
animals from rendered habitable territories. Ozomatli become adults approximately
at the third year of life.
Life expectancy of this animal reaches 15 – 19 years.
Hopping carnocebus (Carnocebus saltator)
Order: Carnivores (Carnivora)
Family: South-American tail-grippers (Carnocebidae)
Habitat: tropical rainforests of South America, forest canopy.

Picture by Amplion, colorization by Lambert
Initial picture by Amplion |
In glacial epoch rainforests of South America had seriously
suffered from climatic changes. Because of dry and cooler climate of ice age
their area had been strongly reduced, and they had remained only along the Atlantic
coast of South America, and in lower reaches of rivers. Accordingly, the variety
of local fauna and flora also had reduced. Especially large damage was incurred
by groups of specialized wood animals among which only few species had remained.
It had taken place with earlier numerous monkeys of New World.
In Neocene the climate became favorable for growth of rainforests again, but
only few species of local monkeys had survived up to this time. In dense forest
canopy they are compelled to compete to numerous animals of other groups adapted
to tree-climbing and jumps.
One of such animals is very similar to Madagascar lemurs of Holocene epoch:
it goes to feed at night, and hides in tree-trunk hollow in day time, covering
by tail and paws large sensitive eyes. But it is not the primate, and a predator
– carnocebus, the descendant of specialized tree-climbing raccoon kinkajou (Potos
flavus). The name “carnocebus” emphasizes two features of this animal: the belonging
to Carnivores order (Carnivora) and external similarity to South-American cebid
monkeys (Cebidae).
Carnocebus inhabits tropical rainforests of the north of South America. This
mammal keeps in forest canopy, practically never going down on the ground. Carnocebus
is perfectly adapted to such way of life. It has fine inheritance from the ancestor
– long tenacious tail which exceeds a little common length of body and head.
The tip of tail is covered from below with hairless cornificate skin which makes
grasp of branch stronger. When carnocebus climbs on branches, its tail, it seems,
independently grasps the next branches, providing secure if the branch appears
too thin. Paws of carnocebus are rather long because of what the animal is similar
to monkey. Rear legs are a little bit longer than front legs because of what
the back of animal is inclined forward. Due to strong rear legs carnocebus can
make long jumps in forest canopy.
Fingers and toes of this animal are supplied with short, but strong and sharp
claws. Till the climbing carnocebus simply clasps branches by fingers, but not
clings to bark, as a cat. Thumbs on hand and foot can oppose to other fingers
or toes.
Wool of carnocebus is short and velvety. General colouring of wool is brown;
on back it is darker, than on paws and stomach. Eyes are bordered by thin ring
of white wool; muzzle and area around of mouth are also white. The tip of tail
is dark, almost black.
Carnocebus has inherited the habit of life of its ancestor: this is twilight
and nocturnal animal. At it there are good hearing and very sharp sight. Ears
of carnocebus are large, rounded, covered with wool on outer side, with fluffy
fringe of long hairs on edge. Eyes of animal are big, round, directed forward:
the muzzle of carnocebus is rather short, that strengthens its similarity to
monkeys even more. Eyes of animal “shine” at night, as at cats: the retina has
a layer of cells containing guanine which additionally reflects faint light
rays to photosensitive layer of retina.
Carnocebus is omnivorous, but it obviously prefers food of animal origin. It
eats insects and small vertebrates: mammals, frogs, lizards and sleeping birds.
Carnocebus is so agile, that it can catch by jump even flying by birds and bats.
The animal eats catch, holding it in forepaws, and sitting on branch vertically.
In such moments animal additionally securing itself by tail clinging to the
branch.
This is solitary animal spending day in shelter – usually in hollow of big tree.
Less often carnocebus uses abandoned bird nest as shelter. When it is placed
at top of tree, the animal arranges the original “canopy” of branches protecting
from solar beams.
Carnocebuses meet and keep for some time in pairs only in breeding season. At
this time the male some time cares at the female: cleans her fur and feeds her
from the mouth. When animals sit on branch, the male can twist by tail the tail
of the female. Animals spend day in common shelter, more often belonging to
the female. When the female feels the approach of pregnancy, she starts to behave
aggressively relatively to the male, and banishes him from the territory. Pregnancy
lasts about three months, and on the average for two years at the female it
happens three litters. It may be two cubs in litter. They are born rather advanced.
Right after birth they can cling by paws to mother’s wool, additionally twisting
her tail by their ones. They are colored darker than adult animals and have
no white marks on muzzle. When the female moves in forest canopy, cubs sit on
her hips and keep so strongly, that it can jump from tree to tree not being
afraid for their life.
Young animals keep near to the female about four months. Usually at the end
of this term the female again ready to pairing again, and cubs are banished
by the male starting to court after her. Young carnocebuses spend first months
of independent life together, and then each of them finds new territory to settle
in. To this time animals become sexual mature. Duration of their life makes
about 14 years.
Gliding weasel (Ventomustela zewanna)
Order: Carnivores (Carnivora)
Family: Mustelids (Mustelidae)
Habitat: Balkan, woods to the north of the Alps; forest canopy.

Picture by Alexander Smyslov
In Neocene subtropical woods had returned to Europe. But trees forming them
were not natives from the south. Mediterranean Sea, the shelter of thermophilic
flora, had turned to heated salt desert, and the rich southern flora was
lost, having replaced by drought-resistant plants of desert. Trees of European
forests
are descendants of species of flora of temperate latitudes: broad-leaved
oaks and maples, relic plane trees, nut trees, beeches and chestnut trees.
They
had managed to survive in mountain valleys during the ice age, and after
restoration of conditions favorable for life, had formed rich shady forests,
stretched
by strip from the Central Europe (to the north of the Alpes) through Tatra
Mountains and Carpathian Mountains up to coast of Fourseas.
On the ground under forest canopy a few kinds of animals live, and the majority
of inhabitants of European deciduous forests live in forest canopy. As against
tropical rainforests, in European subtropical forests there are only few
species of lianes, and crones of separate trees are not connected by them
together.
Therefore in canopy of European forests only ones live, able to fly well
or … to make long gliding jumps. Abundance of prey had involved to forest
canopy
the predator perfectly adapted to movement in such inhabitancy. It is the
representative of mustelid family, the gliding weasel.
The European gliding weasel has kept small size, characteristic for many
mustelid species. The length of body of this animal is about 30 cm, and about
20 cm
falls to fluffy tail. Between paws of gliding weasel skin membrane is advanced,
permitting this little mammal to make long gliding jumps from tree to tree.
Head of animal is small and rounded, with short ears. Eyes are shifted to
forward part of skull and provide good three-dimensional vision – it is vital
for animal
living in a forest canopy to estimate distance before a jump more precisely.
The short wool of gliding weasel is colored brown, and on tail there are
some black rings. The bottom part of body of gliding weasel is snow-white.
To winter
at individuals in northern part of area wool turns longer and a little lighter.
This animal is active and successful predator (it has received the specific
name in honour of goddess of hunting of western Slavs). At gliding weasel
there are large canines, and mouth can open very widely. Gliding weasel eats
various
forest rodents, bats and birds. This
predator kills prey by bite in head. Gliding weasel often ravages bird nests,
showing miracles of acrobatics and dexterously
landing even on very thin twigs where the nest is built. This animal can
climb in hollows to bee nests in searches of honey, and eats other insects
and large
spiders. Gliding weasel spends not whole life in tree crones. It often goes
down on the ground in searches of forage. Being a predator by its nature,
it does not disdain ripe berries, and in autumn cracks large oily seeds of
beeches
and chestnuts which grow in forests of Southern Europe much. On the ground
this animal eats worms and insects. This mammal is able to climb on trees
as dexterously, as squirrel, despite of presence of gliding membrane.
Gliding weasel is a solitary animal. Each individual has site in forest and
marks it with odorous musky secretions. The territory of animal includes
not only a part of crones of trees, but also trunks and the ground under
trees.
Gliding weasel pair two times per year. First time courtship games occur
at the end of winter when night frosts stop. Males start to come in territory
of females and to leave their own odorous marks near female’s ones. If the
female is ready to pairing, her musky secretions get a special smell well
distinguishing
by males. When the male feels it, he tries to reach the female as soon as
possible. Sometimes at once some males follow one female. They constantly
compete with
each other, and sometimes can bite each other, putting deep painful wounds.
Sometimes competing males seize each other’s gliding membrane, and one of
them can pay for failure in courtship season by loss of ability to make gliding
jumps. Eventually, near the female only one male stays, which pairs with
her.
After the pairing the female banishes the male. Pregnancy lasts till about
two months, and in second half of spring the female gives rise to 4 – 6 cubs.
They are blind and helpless, and female looks after them within 2 months
before they become independent. She hides cubs in deep hollow of tree at
the big height.
Young gliding weasels differ from adults in darker colouring of wool and
absence of strips on tail.
Second time for a season the female is ready to pairing approximately in
one month after cubs had ceased to suck milk. The first days of pregnancy
the female
concerns to grown up cubs tolerantly, but later starts to express aggression
to them more often, and compels them to live independently. Second time it
brings posterity at the end of summer. In the second litter it is less number
of cubs – only two or three ones. But they are larger, than ones in first
litter, and become quite independent predators to the beginning of winter.
The main enemy of these animals is large gospodar
woodpecker which pecks out
tree-trunk hollows and can eat newborn cubs of gliding weasel.
The idea about existence of this animal was proposed by Momus, the forum member.
Antipod’s unotter (Xenolutra antipodorum)
Order: Carnivores (Carnivora)
Family: Mustelids (Mustelidae)
Habitat: New Zealand, sea coast and lower stream of rivers.
Freshwater ecosystems of New Zealand differed in poverty and high degree of
endemism in comparison with continental ones. It is connected with very early
isolation of archipelago, which had taken place before occurrence of the most
typical families of freshwater fishes and other water vertebrates. The situation
had sharply changed in human epoch when various species of freshwater fishes
and predatory mammals had been introduced to islands. After disappearance of
mankind their descendants had changed freshwater ecosystems up to unrecognizability.
One of such species lives in lower reaches of rivers of New Zealand; it is
a predatory aquatic mammal, crowning with itself the food pyramid of ecosystem.
By shape this animal is very similar to otter, but is connected to it with
only remote relationship by origin: it is the descendant of the domesticated
American mink (Mustela vison) introduced to islands in human epoch. This animal
is named antipod’s unotter. It is much larger, than the ancestor: up to one
and half meters long including wide flattened tail.
At antipod’s unotter there are rather short paws with long fingers connected
by elastic membrane. The antipod’s unotter is very vulnerable on land, but
in water only few animals can make a competition to it. For slow and maneuverable
swimming animal uses paws, but at hunting in open water unotter presses paws
to body and swims with the help of vertical bends of backbone and movements
of tail. Tail of antipod’s unotter is wide and flat, similar to beaver’s one.
It is covered with dense skin and shorter fur, than the body.
Antipod’s unotter has flattened head with short strong jaws which easily crack
skull of fish up to 40 – 50 cm long, or the armour of large crab. Ears of animal
are short and rounded. On muzzle of unotter long rigid whiskers – vibrisses
– grow; with their help animal searches for prey under water.
The body of unotter is covered with dense rich fur of grey color with white
spot on throat which passes to longitudinal strip on stomach. Fur of antipod’s
unotter is rich and dense. It reliably keeps air, and consequently animal is
not cold even in cool water. Unotter carefully looks after fur, and combs it
with two wide claws of forepaws which bottom surface forms “combs”. The animal
also greases fur with secretions of former musky glands, located near the anal
aperture. In this connection they had strongly changed: having lost smell component
(the strong smell is characteristic for many mustelids), they became plentiful
and oily. Cubs at which glands poorly function yet, use for care of fur secretions
of the female.
Antipod’s unotter is active piscivorous predator. It usually hunts in coastal
zone and in thickets of brown algae. These animals especially willingly catch
pelagic fishes which shoals swim up close to the coast. The group of animals
attacks fishes, and drive their shoal into algae thickets where fishes lose
orientation: in such situation it is easier to catch them. Besides of fish,
antipod’s unotter eats crustaceans and small snails. After storm these animals
gather on coast dead sea animals cast ashore.
By the way of life the antipod’s unotter is social species. It settles at sea
coast or in mouth of the river in big groups numbering up to ten breeding pairs
of adult animals. Besides of them in groups there are some single adult individuals,
and also numerous youngsters – the posterity, yet not completely passed to
independent life. The colony lodges in places where there is an opportunity
to arrange shelter. Animals dig in the ground some common holes, or lodge in
cave well protected from storm waves.
The pair at antipod’s unotters is formed to all further life. Partners differ
in attachment to each other – they have a rest and hunt together. Male is larger,
than female and female regularly renders to him signs of attention and submission
– she clears and greases his fur and licks his muzzle. Once a year, in early
spring the female gives rise to 3 – 5 cubs. They are born blind and deaf, but
covered with wool. The female gives rise to posterity in shelter, on litter
of grass and seaweed. First some days she does not admit to posterity even
her male. Week-aged cubs start to hear, little bit later eyes open at them.
At the age of 17 days cubs start to creep out from den and to explore the world
around. The female feeds them with milk approximately up to bi-monthly age,
but already fortnight cubs start to try food of adult animals: male brings
in the den fish, and the female allows cubs to eat a little. At the age of
two months young animals already leave a den and start to study to swim and
to get food independently.
As a rule, males abandon the group at the age of half-year. They lead barchelor
life, and can found their own colony if they find a place suitable to life.
If the parental colony is overpopulated, adult individuals can show aggression
to young growth. Then even young females leave a colony; later they can join
a colony of bachelor males and form breeding pairs with them. At the age of
two years young animals become able to breeding.
Littoral mink (Neovison dicynodonta)
Order: Carnivors (Carnivora)
Family: Mustelids (Mustelidae)
Habitat: sea coasts of North America from polar to temperate waters.
Among mammals the mustelid family shows rather different shape of species included,
varying from tiny weasel conceding to some rats in length, up to glutton close
to bear cub in size. Mustelids live on trees (marten, American mink, etc.),
under ground (badger, black-footed ferret etc.), and even in water (otters
and minks). In Holocene separate representatives of this family even devepoled
oceanic habitat! Also it is necessary to note that seals descend from early
mustelids.
Short, but severe epoch of anthropogenous pressure and climatic changes took
place right after it had been a true test for durability for mustelids. The
area of some species had been reduced, and some of them had become extincy
completely.
In due course mustelids began to return former positions and to occupy again
ecological niches lost earlier. American mink (Neovison vison) had occupied
a niche of otters extinct on human fault, and in due course was settled up
to ocean. It had not become high-grade analogue of sea otter (Enhydra lutris)
dyed out at the boundary of Holocene and Neocene, but evolved to one of the
most unusual mustelid representatives of Neocene – littoral mink.
Littoral mink is an animal kept features of the ancestor in many respects:
lengthened body and tail, and short webby paws. But it is also adapted to new
inhabitancy and had developed special attributes, not characteristic for ancestor.
One of the main features of this species is the structure of teeth. Incisors
are still sharp, but strongly reduced. As opposed to them molars are low, but
wide and massive, with the powerful roots, resembling nutcrackers. Canines,
though they stick out of mouth, as if “sabers” of other predators, are absolutely
straight and smooth, resembling tusks of Holocene walrus. Littoral mink is
its ecological analogue, though in miniature.
This animal has rich fur of brown color. Littoral mink carefully looks after
it, cleaning the dirt with claws of forepaws and greasing it with fat musky
secretions of anal glands.
Bones of mink skeleton turned appreciablly heavier, and muscles have become
stronger: it is twice longer than its ancestor and almost ten times heavier.
It allows an animal to dive under water and to remain there till 15 – 20 minutes
without any serious problems. Certainly, it is not enough to catch quickly
swimming pelagic fishes and squids, but this mink does not hunt them: its prey
includes bentic fishes and crustaceans, bivalves and gastropods, less often
sea urchins and starfishes. It easily cracks shells and carapaces of animals
by molars, and tears off snails and bivalve shells from stones by canines.
As well as other sea mammals, mink swims, waving by the whole body in vertical
plane, and uses short and wide hind feet and extended tail as rudders. Forepaws
also have palamas between fingers, but do not take part in underwater movement:
sea minks use them for movement overland where they move in seal manners –
on all fours, pushing by hind legs.
The main enemies of littoral mink are predatory fishes and orcine
squids. On
land it is attacked by coastal predatory birds and carnivorous mammals searching
for food in coastal zone. But ground predators are usually dangerous only for
cubs: adult minks, having powerful jaw bones and muscles, can be formidable
opponents. Protecting themselves, they put deep bites. Pain sensitivity is
reduced at them, therefore littoral mink, protecting itself from large predator,
can hang on it in death grip and does not unclench jaws even if it will be
gnawed and biten.
Littoral minks live at both coasts of North America. The Pacific population
is isolated from Atlantic one by Beringean Isthmus at the north and by tropical
areas at the south. Probably, it is formed as a result of unitary settling
of the Atlantic population to the south during any short-term ice age. To winter
animals of northern populations swim to the south: despite of rich fur and
underskin fat, these animals bear cold Canadian winters not so successfully.
They migrate in large packs, pursued by sea predators hunting weakened and
left behind animals. In spring migration takes place in opposite direction.
These animals keep in family groups including 1 – 2 males and 4 – 6 females.
Females of littoral minks bring posterity in second half of spring, after the
arrival to summer residence. In litter it may be up to 7 – 10 cubs, but within
the first year of life less then half of them will survive – the others perish
from predators or during autumn migration.
The maximal life expectancy of littoral minks is 12 – 15 years.
This species of mammals was discovered by Bhut, the forum member.
Grokk
(Quasiursus grokk)
Order: Carnivores (Carnivora)
Family: Mustelids (Mustelidae)
Habitat: Western, Central and Southern Europe, Atlas Mountains, Asia Minor,
the Caucasus peninsula.
During the first millions of years of early Neocene world resembled a little
the early Palaeocene, as small animals quickly increased in size to occupy the
vacant niches in emerging ecosystems. The ecological niche of bears at the area
of Holarctic had been taken over by the large descendants of ferrets (Mustela
putoris) that form a single berl (Quasiursus)
genus. Only in the Far East they are replaced by two more predators: arctomeles
(Arctomeles tardus) and giant tanooka (Tanuka
gigantea), natives of Oriental zoogeographical realm. South of the Alps,
Western Europe and North Africa are inhabited by grokk, another representative
of berl genus. It evolved in relative isolation from another species and is
slightly different from them. This is a predator and a scavenger, an ecological
equivalent of the Holocene bear, but more carnivorous: plant food makes not
more than 10% of the diet.
The grokk is reminiscent in appearance of a cross between a ferret and a badger,
but is an animal of a large bear size; its shoulder height is about 1 meter.
It has an elongated body, pretty short strong paws, large head and short tail.
The fur of the grokk has sandy-yellow shade with darker legs and a “mask” on
the muzzle. Tail is also dark, but with a white tip. Grokk is a sluggish animal
with plantigrade feet, but over short distances, it can run at a very high speed,
catching up with its prey. This animal’s claws are long and non-retractable.
This mammal is a carnivore, it has strong jaws, and molars have a well-developed
cutting edge; the canines are elongated. Grokk’s sense of smell is very acute
– the nasal cavity is quite extensive, and due to it animal’s head is shaped
more like a cat’s than a bear’s. The eyesight is comparatively weak, the eyes
are small. Hearing is sharp – the external ears are moveable and are located
at the top of the head.
The grokk is a solitary predator, specializing in hunting for large herbivores
– tapiroids and embolohyraxes.
This species has tremendous strength, and the adult animal is capable of toppling
a young embolohyrax on the ground and breaking its neck. Also, this mammal willingly
eats carrion, finding it by smell. Using its superior strength, this species
easily seizes prey from other predators. In the Atlas Mountains, this mammal
attacks young giraffe ostriches.
Due to this feeding specialization the population of the predator is widely
dispersed. Each female occupies approximately 100 square kilometres of territory
and in fact spends its whole life there. On contrary, the males travel much
more: they roam vast territories, partly overlapping those of other individuals.
The territory of each male includes territories of several females. During the
mating season the male indulges in long wanderings, fertilizing females that
live within its posessions. Each individual marks its territory by using secretions
of musk glands, inherited from mustelid ancestors, and communication of different
individuals occurs mainly through scent marks that these animals leave on the
noticeable elements of the landscape – on large stones and single trees.
Grokk is a quiet animal. Using sounds in communication occurs mostly between
females and their offsprings, and when meeting each other, two adult grokks
roar loudly, trying to oust a rival from its territory. The voice of the grokk
in calm is a short snarl, hence the onomatopoeic name of the animal.
The mating season begins in the fall. When ready to mate, the female produces
a distinctive odor, which attracts males. After the end of estrus the female
chases away the male. 2-3 cubs are born in spring, within the first few weeks
of life intense competition arises between them, and fights take place often.
Only one cub of the litter usually reaches the independent condition. The sexual
maturity comes at the age of 3 years; the life expectancy is up to 50 years
Translated by Bhut.
Aotearoan
False Glutton (Pseudogulo australis)
Order: Carnivores (Carnivora)
Family: Weasels (Mustelidae)
Habitat: Well vegetated habitats across New Zealand, with a preference for those
that are not swampy, including mountain slopes, woodlands, meadows, savannah
and coasts.

Picture by Alexander Smyslov
The introduction of ermine and ferrets to New Zealand by mankind
during the Holocene was a catastrophe for many kinds of native animal there,
particularly ground-nesting birds. But these animals were provided later opportunities
to prosper in the Neocene, radiating into new niches, omnivores,
burrowers, and scavengers.
One notable scavenging form is the Aotearoan False Glutton, this is a large,
bulky Mustelid descended from the ferret, recalling a wolverine in shape. Body
length is up to a 85 centimeters, and weight in large individuals can be up
to 25 kilograms. Animal is built low to the ground with strong, powerfully built
limbs, tail is short and bushy. Head and jaws are massive, cutting teeth are
large and accompanied by heavy crested back molars for processing bone, canines
are long. Colour recalls partway between ferret and wolverine, with a dark back
and limbs, lighter sides and a dark “mask” around the eyes.
Diet consists partly of mid-sized and small animals caught for itself, rabbits,
birds, large rodents and snakes, but a great deal of its diet comes from scavenging
carcasses left behind by other predators, Kurī
hou, Ruacapangi bird
or Marsupial Pardus. In this
way it recalls a miniature hyena, as it is able to gnaw and crush bones as well
as consuming flesh and organs. Sometimes as many as four or five may congregate
around a carcass, and only the larger bones and ends of some bones usually remain,
some may be cached to eat later. This animal is active the most in the late
afternoon and early morning, including twilight. Calls vary from a chirring
growl to a yarring squeal. Males produce an extremely strong glandular odour,
and mark their territory with glandular secretions, but it does not use glands
in defence as a skunk would.
Though generally solitary and quarrelsome amongst themselves, dominant males
will allow multiple females within their home territory, mating with each of
them and preventing mating by smaller males. Breeding season is in winter from
June to September. Gestation is up to 35 days with litters of two or three young
born in late spring, weaning at 10 weeks, cubs reach adult size within a year
and are overall darker than their parents. Lifespan may be up to 14 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Black
jaguape (Manducomellifer niger)
Order: Carnivores (Carnivora)
Family: Mustelids (Mustelidae)
Habitat: South America, tropical forested areas.
The introduction of species to new areas by people during the Holocene epoch
caused many impacts over the indigenous species of these habitats. Some species
were capable to adapt to the presence of the invasive ones, and even to develop
ecological relations with them. The mammalian genus Manducomellifer appeared
in this context, descending from the tayra (Eira barbara). These mammals are
adapted to eat honey, larvae and pupae of bees, mainly of those belonging to
the genus Apis introduced to South America during the Holocene. The type species
of this genus is black jaguape (the Tupi word jaguape is used to refer to the
tayra). This animal lives in the tropical areas of South America.
Black jaguape is 65 cm long with 40 cm long tail; female is smaller than male.
These animals have short black fur on the body, limbs and tail, with two yellow
stripes coming from their snout, passing around the eyes, to the back. These
stripes represent a feature common to all species of Manducomellifer genus.
Their appearance resembles that of a slender badger; this mammal has slender
body with short ears, small eyes and short snout. This species has skin so thick,
that bee stings rarely penetrate it. They have long claws on the forepaws, which
they use to excavate hives in the ground or in hollowed trees. Black jaguapes
are good in tree climbing, being capable to leap from treetop to treetop when
pursued, using their tails for balance. Mostly diurnal, they sleep in hollow
trees or burrows at night.
They are opportunistic omnivores, hunting rodents and other small mammals, as
well as birds, lizards and invertebrates, and climbing trees to get fruits.
However they have a great appreciation for honey, larvae and pupae of bees –
these are the most common elements in their diet. When black jaguapes find an
underground bee colony, they dig it and not suffer too much from bee stings,
due to their thick skin. All jaguape species developed an ecological relationship
with bee-eater tanager, bird
of the subfamily Apiphagiinae in Thraupidae family. When jaguape appears near
them, these birds perch on tree near bee nest and start to vocalize. It attracts
the attention of the mammal to bee colony, and when the mustelid begins to ravage
the nest, bees try to defend it. As the jaguape begins to devour honey and larvae,
the birds begin attacking and hunting bees defending the colony.
Black jaguapes breed year-round, with the females entering estrus several times
per year for 15 days at a time. The territory of a successful male overlaps
that of two to five females. The female gives birth to one to three young and
takes care to them for alone. Newborn cubs are blind and have closed ears, but
are already covered in a full coat of black fur. Their eyes open at 46 days,
and they leave the den shortly thereafter. They begin to take solid food around
70 days of age, and are fully weaned by 105 days. Hunting behavior begins as
early as at three months, and the mother initially brings to her young any wounded
or slow prey to practice on as they improve their killing technique. The female
also teaches them to hear and to follow the vocalization of bee-eater tanagers
to find bees. The young animals are fully grown being around 6 months old, and
leave their mother to establish their own territory in 10 months. The lifespan
of this species is about 15 years.
There is another species in the genus Manducomellifer – Andean
jaguape (Manducomellifer andinus). Living in elevated areas of the
Andes, this species has 50 cm of length, with 29 cm long tail. Its fur is longer
and has light brown shade. These animals have the same yellow stripes as previous
species, but their coloration is duller. Andean jaguape is also less agile in
trees and climbs rarely. In southern part of South America it lives not only
in mountains, but also in southern temperate forests, forming a separate subspecies
M. a. australis that is 67 cm long with 33 cm long tail. Its
fur turns longer in winter and is dark brown, and stripes on head are yellowish-orange.
When bees gather in their nests to spend the winter, austral jaguape passes
to a more carnivorous diet.
This mammal species was discovered by João Vitor Coutinho, Brazil
Neosurilio
(Neosurilio zebropygus)
Order: Carnivores (Carnivora)
Family: Mustelids (Mustelidae)
Habitat: forests of the east of North America and along the coast of Gulf of
Mexico.
In epoch of biological crisis at the boundary of Holocene and Neocene the species
had not having strict specialization received advantage in survival. The part
of these species belonged to the group of synanthropic ones, and had adapted
to live near people, having received from it additional benefits – an abundance
of forage, refuge and rather quiet existence. But after human disappearance
all species had received equal opportunities for further evolution.
Among mustelid family among survived ones there were North American skunks –
the omnivores having an effective way of protection against enemies. One of
descendants of spotty skunk (Spilogale gracialis), neosurilio (“surilio” is
the name of one species of skunks in human epoch) lives in Neocene epoch in
forests of North America. It is the ecological analogue of badger, the sluggish
plantigrade animal having massive constitution. Length of body of this animal
is about 70 – 80 cm, and length of tail is about 40 cm.
Neosurilio is noticeable from apart due to contrast warning colouring. The animal
as if consists of two halves sharply differing by colouring. On back part of
body, hips and waist there are alternating black and white strips (hence the
scientific specific name meaning “having back of zebra”). Tip of tail is also
white. Forward part of body of neosurilio is brown with white throat and stomach.
Around of eyes there are “glasses” of white wool, and on top of head there is
black “cap”.
Neosurilio has kept way of protection characteristic for skunk: it defends against
enemies splashing smelly liquid from strongly advanced anal glands. In special
sacs the oily liquid containing ethylmercaptane accumulates in amount enough
to make some “shots” in succession to distance up to 3 meters. Before application
of the weapon animal shows to aggressor the black-and-white back, having turned
it to aggressor. At aggressively behaving neosurilio wool on back part of body
rises on end and fluffs, increasing a little the visual sizes of this part of
animal. If this warning had not worked, jet of smelly liquid flies to predator.
Neosurilio consumes various kinds of food, and due to the omnivorousity it does
not stay hungry in any district. On forepaws of animal thick strong claws grow,
with which help neosurilio easily digs out holes of small animals and gets roots
and tubers of plants. More often neosurilio eats small animal – invertebrates,
amphibians; also it ravages nests and eats eggs and nestlings of small birds.
Up to half of diet of this animal the vegetative food makes – tubers, roots
of plants and berries. Neosurilio eats food “aristocratically” – it sits on
hind legs and brings it by front paws to the mouth. At the end of summer and
in autumn neosurilio almost completely passes to vegetative food, and during
winter and spring food shortage does not squeamish about carrion. Using the
chemical weapon, neosurilio behaves very impudently, and can drive away from
prey even large predators.
Each individual builds in the territory some temporary shelters and spends the
night in them during wandering on the territory. Also neosurilio does not overlook
to mark border of territory, splashing some drops of combat liquid on roots
of large trees. The female ready to birth of posterity becomes homebody. Right
before birth of posterity she stays best arranged shelter, and starts to expand
it even more. She digs a hole up to 5 meters deep with several emergency exits.
Pregnancy at neosurilio lasts about three months, and proceeds from the end
of winter up to middle of spring. The female gives rise to 2 – 3 large cubs.
They are born blind and deaf, but already at birth are covered with wool. The
female cares of them for a long time, feeding them by milk up to 4-month's age.
But to this time cubs already develop enough to leave a hole and to follow the
female. At this time at them also odorous glands develop and they become able
to splash smelly liquid. Young animals spend the first winter with the female,
learning the search of food, and leave her shortly before courtship season when
she starts to behave aggressively to them.
Common
fruit civet (Paradoxuroides frutophagus)
Order: Carnivores (Carnivora)
Family: Palm civets (Nandiniidae)
Habitat: tropical forests of Central Africa, forest canopy.

Picture by JOrnitho
In Holocene Equatorial Africa was a sanctuary of African palm
civet (Nandinia binotata), an arboreal civet-like mammal. In fact, this animal
isn’t a true viverrid, but a member of it’s own family inside the feliforms.
African palm civet survived in Neocene and gave rise to new genus.
A habitant of the forest canopy, fruit civet is a very agile animal in the trees,
being capable to grasp branches equally well with its fore- and hind paws and
can hang from branches only on one foot and use its long tail for balance. This
way it takes its main food – fruits. This species rarely descends to the ground,
being capable to jump from a tree to another – a necessary ability, since its
habitat represents dense tropical forests of Congo Basin.
Fruit civet is similar in appearance to Holocene palm civets of the genus Paradoxurus,
true members of viverrid family. It has a slender, cat-like body, small head
and large eyes. Its body has 45 to 56 cm of length and tail measuring 34 to
54 cm; males usually are larger than females. Its legs are long, with cat-like
feet and semi-retractile claws. Its fur is dense and soft, and the coat is greyish
brown, with dark spots on the back. There is also a black stripe coming from
the forehead, along the spine and ending in the base of the tail – this feature
is present in all species of the genus. Dark patches of wool are present beneath
the eyes, which are offset against the reddish brown fur of the chin and throat.
Tail is cross-striped with thin black rings. This species is nocturnal and crepuscular,
with its highest levels of activity following sunset and just prior to sunrise.
During the day, these animals rest in hollow trees.
They communicate with each other using sounds, scents, and visual signals. Their
scents are sweet, similar to ripe fruit. Both males and females scent mark in
their home ranges. Females mark their territory using scent glands on their
flanks, hind legs and perineum. Males mark less frequently than females, often
spraying urine, rather than using their scent glands, and behave so primarily
during the breeding season. Scent marks of both sexes allow individuals identifying
the reproductive and social status of other civets. Fruit civets also defecate
at specific latrine sites, which are often located at the edges of their territories,
and perform a similar function to other scent marks.
Fruits, mainly figs, make up the most part of its diet, but, when necessary,
these animals can eat small vertebrates like birds, mammals, frogs and lizards.
Eggs are also consumed. They locate their prey primarily by scent, chasing them
agilely through the trees, and kill it with a bite to the neck like felids do.
To reach larger fruits they stand up, using their front paws to hold the fruit
and proceed to eat, while using their tail for balance.
This species is solitary, only meeting in the breeding period that starts in
the last days of the dry season. In rainforest areas breeding season takes place
the year round. Female leaves a trail of scent for the male to follow; when
he finds her they copulate. If another male finds them, they will fight in the
trees, chasing one another through the branches until the loser runs away. The
victorious male will stay in the female’s territory for two weeks, making sure
that no one another male appears there.
After a gestation period of 12 to 13 weeks, up to six cubs are born in a hollow
tree. They start eating solid food at around seven weeks of age, and are fully
weaned at four months of age. Being five months old, they are skilled in foraging
for food on their own. At the age of seven months they leave mother’s territory.
When 19 months old, they start marking their territories, and are sexually mature
at the age of two years. The lifespan of fruit civet is about 13 years.
Other species of this genus exist in Africa:
Ring-tailed
fruit civet (Paradoxuroides annulatus) lives in the forests of the
Atlantic coast of Africa. This species has body length of 41 to 52 cm and tail
34 to 50 cm long. Ring-tailed fruit civet is similar to the nominal species,
but the rings on its tail are wider and more accentuated, justifying thus its
name. Its fur is pale brown with small spots.
Masked
fruit civet (Paradoxuroides civetta) is less arboreal and frugivorous
species of this genus. It lives in woods and savannas of Central Africa. Its
lifestyle is similar to that of the Holocene African civet (Civettictis civetta).
It has body length from 42 to 54 cm and short tail 29 to 36 cm long. Its main
characteristic is a spot of black fur around eyes similar to a mask. The general
fur coloration is brown with dark spots and tail has thin dark rings.
Banded
fruit civet (Paradoxuroides fasciatus) inhabits coastal forests of
Eastern Africa (Tanganyika Passage). This species grows to 43 to 53 cm with
a tail measuring 35 to 52 cm. Its name comes from four dark bands on its back.
Background color of fur is reddish brown, it is spotless. Tail has thin dark
rings.
This mammal species was discovered by João Vitor Coutinho, Brazil
Ursine
civet (Ursivetta aromatica)
Order: Carnivores (Carnivora)
Family: Civets (Viverridae)
Habitat: mountain woods of Southern Asia.

Picture by Alexander Smyslov
Mass extinction of live creatures at the border of Holocene
and Neocene had given few chances of survival to animals weighting
more than 20 kgs. Predators
dependent on abundance of large prey – cats, dogs, hyaenas and bears have
especially suffered from it. But small predators like raccoons, weasels and
civets have
easily survived and have “privately divided” the planet: weasels and their
descendants became main predators of temperate and cold areas of Earth,
and civets were spreaded in tropical zone of Old World. Only in Africa and
partly in America relic representatives of cats, descendants of small cat
species compete with them. Holocene representatives of civet family by the
constitution
are analogues of primitive predators of early Cenozoic, therefore it is possible
to assume, that they have the big evolutionary future. Among them there are
ecological analogues of omnivorous bears and carnivorous cats, therefore
it is possible to assume, that in the future they will occupy place of these
animals.
Ursine civet is the huge representative of civet family. This animal is similar
to bear with long tail (prehistoric analogue of ursine civet is the huge
fossil marten Perunium dwelt in Eurasia). Constitution of this animal is,
however,
more graceful, than at bear: neck is longer and more mobile, and muzzle is
more extended. Adult animal weighs up to 250 - 300 kg (males are larger).
Legs are almost plantigrade, with long non-retracting claws. Fur of animal
is rich
and rather long (the animal frequently meets in mountains, sometimes in summer
rising up to zone of Alpine vegetation), chestnut-brown with mesh white pattern
on neck and back. On tail there are some cross rings of white wool, the tail
tip is black. With the help of tail position ursine civet expresses its mood
at the meeting with neighbours. In the basis of tail there are repugnatorial
glands which emit pleasant musk smell for territory marking.
This animal is solitary one living in territory numbering some square kilometers
in area and strictly protected from neighbours. Only female and cubs can
live any time in group.
The ursine civet is omnivorous species with deviation to predating. Teeth
at it are wide, knobby and dulled – vegetative food makes about 40 % of diet.
Among plants ursine civet prefers fruits of different palms rich in oil.
Also
it digs out by claws roots of huge grasses of family Apiaceae growing on
mountain slopes, and tubers of various wood grasses. On river banks ursine
civet eats
roots of reed mace and other marsh plants. The food of animal origin includes
larvae of various insects, frogs and lizards, eggs and nestlings of ground
birds. If there is an opportunity, ursine civet willingly eats carrion and
attacks cubs of ground animals. Due to claws this animal can climb trees,
ravaging bird nests.
Ursine civet is mainly day time animal. It spends night in shelter which
it is usually a little number in its territory. From time to time animal
moves
to other shelter, giving to ants and the other insects the right to eat staying
in lair litter larvae of fleas and ticks. For disposal of parasites animal
willingly bathes in river and wallows on odorous grasses, especially on greenery
of plants of Apiaceae family (having eaten its roots).
Borders of territories can be broken only in season of pairing. Usually on
border of possession of several animals there is special remarkable place
like alone growing tree or large stone. Animals make here original “message
board”,
marking it with odorous secretions. When the male notices on smell, that
the female is ready to pairing, he puts the mark atop of mark of this female,
and
waits nearby from “message board” driving away other males. Also he puts
marks in territory of chosen female: peels bark from tree and marks this
place with
odorous secretions, declaring about its presence. When the female comes to
“message board” and sniffs at it, she becomes acquainted “in absentia” with
the male, and if the male is pleasant to her, some time it allows him to
remain nearby for pairing.
When the female feels approach of pregnancy, it banishes out the male, sometimes
should win anew his own territory if on it in his absence the neighbour had
lodged. Pregnancy lasts about five months. Shortly before cub birth the female
builds lair – she digs out hole under tree roots or finds ready cave in mountains.
She drags moss and branches for litter, and carefully hides traces of her
presence near to chosen refuge.
In pack there are usually two cubs though sometimes there are triplets, and
young females bear only one cub. Young growth is born blind and with closed
ears, covered by short wool. Colouring of newborn cubs is spotty: chestnut
stains on red or straw-coloured background (characteristic colouring of Asian
civet (Viverricula), an ancestor of the ursine civet, was spotty), later
cubs darken. At ten-day age at them eyes and ears open, and month-aged cubs
already
can play near lair entrance. At bi-monthly age cubs leave shelter, and female
teaches them in ways of food getting. Young growth stay with mother up to
eight-monthly age then starts to live independently. Sexual maturity comes
at them at three-year
age, and life expectancy can reach up to 30 - 35 years.
Pardinia
(Pseudacinonyx gracilis)
Order: Carnivores (Carnivora)
Family: Civets (Viverridae)
Habitat: savannas and light forests of Southern Africa.

Picture by Viergacht
Mass extinction of large herbivore animals has caused disappearance
of large predators eating them. After stabilization of situation and restoration
of biological balance as the reply to evolution of herbivores large predators
have started to appear – they were descendants of rather small forms which have
survived during mass extinction.
Pardinia is one of such predatory animals, the descendant of genette (Genetta
genetta), widely spreaded in Holocene at the territory of Northern Africa and
Southern Europe. Its close relative is the huge deadlynetta
(Necrogenetta deima), living in North-African savannas. But if deadlynetta is
the specialized sabertooth hunter for large prey, pardinia is graceful analogue
of the cheetah chasing swift-footed herbivores. By constitution pardinia resembles
fleet dog with long tail and rather short muzzle. Height of animal at a shoulder
is about 80 cm, body length is up to 120 cm, tail lenght is about meter; back
legs are little bit longer than front ones. Body of pardinia is covered with
short wool, on edges of ears there is fringe of long soft hair. Wool is straw-coloured
with scattered brown stains on back merging to longitudinal strips. On throat
of males there is big white spot bordered by dark-brown strip. Head of pardinia
is similar to cat’s one, but is more extended. Canines are well advanced, their
ends are visible from closed mouth of animal; bases of canines are very wide.
Chewing muscles are well advanced: animal kills catch by bite in backbone or
in the basis of skull.
Similarly to cheetah pardinia chases catch by fast throw. But as against cheetahs
it also can chase lonely catch walking for a long time, yet will drive it up
to exhaustion. Chasing prey, pardinia tries to fall catch down on the ground
by impact of paws against side or shoulder, and then presses it by own weight
against ground and bites in backbone.
Pardinia is solitary predator. Each female has territory marking it with secret
of musk glands. Male, as a rule, has no constant territory: he wanders on territories
of several females, remaining for the pairing period at one of them. Pregnancy
lasts for 3 months. Shortly before cub birth female builds shelter in rich bushes
or among stones where it gives rise to posterity: 4-6 cubs. Cubs are born blind,
covered by dark one-colour wool. Later their colouring replaces to motley one,
characteristic for adults. Cubs develop rather slowly: they start to leave shelter
at monthly age, and study to hunt approximately from three-monthly age. About
half-year they hunt together with mother, leaving her shortly before occurrence
of new pack. Sexual maturity at young animals comes at the second year of life.
Pardinia hunts mainly quickly running herbivore animals - hoofed mammals, kept
in Southern Africa as relicts of Holocene fauna. Hunting usually occurs on dawn
though the female having cubs can hunt even in hottest time of day. Pardinia
creeps to herd hiding in bushes and grasses, stopping at slightest sign of alarm
at herbivores. Having chosen prey animal tries to catch it by one throw – pardinia
is not able to run fast at long distance. Usually only every 6-th or 7-th attack
ends successfully.
If number of large catch is not enough, pardinia willingly foods with small
vertebrates – frogs, lizards and also birds. Dexterity of the pardinia is great:
it can drop flying up birds in jump by long paws.
Organism of pardinia as against to organism of deadlynetta related to it works
literally “for deterioration” – during run plenty of forces is spent. Therefore
life expectancy of this animal is not so great: in total up to 12 – 14 years.
Gelluda
(Pardonetta gelloudes)
Order: Carnivores (Carnivora)
Family: Viverrides (Viverridae)
Habitat: forests of South-East of Europe, forested areas of Fourseas coast,
Caucasian Peninsula.
During Holocene, the human epoch, the native European fauna had reduced in number
and in diversity. Many groups of animals, especially the large-sized ones, did
not go through co-existence with humanity and died out without living descendants.
Among them there were carnivorans – wolves, brown bears, European lynxes, etc…
They had been replaced by the descendants of the predators of the “second wave”
– mustelids and viverrids, including the descendants of the common genet. The
panther genet inhabits the west and also in center and in the south of Europe,
and to the east it is replaced by gelluda, a closely related species.
Like its relatives, the panther genet and the deadlynetta,
gelluda is a very large animal, which reaches 160 centimeters in length (tail
length is about one meter) and weighs approximately 70 kilograms. Its fur has
grayish-white or pale yellow color, on which there are darker spots of irregular
shape that merge to vertical stripes on the rear half of body and into rings
on tail.
The appearance of gelluda has clear features of specialized predator: in has
a robust skull and sharp teeth, especially the incisors. Unlike the deadlynetta,
it does not have sabre-like canine teeth – on the contrary, its canines and
incisors are close in size and are equally sharp, capable of breaking through
almost any skin and of cutting meat. The molars, on the contrary, are blunt
slightly and act like forceps. They are capable of breaking through even bones
of medium thickness.
The muzzle of this animal is shortened and has strong jaw muscles – the role
of gelluda in ecosystem is like the role of hyena, because it is able to eat
its prey including the most part of bones.
When hunting, gelluda prefers to use its claws and weight, but not its teeth
on contrary to felines: after jumping out from an ambush, it smashes into the
side of herbivore, and begins to rake it with claws, trying to bring down its
prey. Pain, the loss of the blood and the weight of the predator force its prey
to fall and gelluda usually bites through cervical vertebrae of its prey to
kill it. Gelluda hunts stronger and larger herbivores more carefully, putting
smaller wounds to it and chasing it for a long time. Gelluda almost always hunts
on the ground, and almost never uses trees for this purpose; therefore its basic
prey includes medium-sized and large animals rather than the small ones, but
during the period of hunger (usually in winter) it is capable of eating any
animal, which it will be able to overpower.
In contrast to their relatives, gelludas are comparatively sociable animals,
and live usually in pairs (both parents rear their 2-4 youngs), and in winter
several (usually 2-3) pairs can act together, attacking even such animals, which
they would not risk attacking in spring or in summer. At this time the territories
of these animals are also used in common.Sometimes lonely animal joins the family
and forms a pair with one young animal.
The mating period begins at the end of winter. At that time gelluda females
begin to leave odorous marks on trunks of trees, stumps, large boulders and
so forth. Males find these markers by smell and find females ready to mating
following their marks. If several males gather near one female, they fight,
punching each other on the snout with their front paws. They sit on rear ones
when dueling. Head and shoulders of adult male may be covered with a pattern
of numerous scars, the traces of such duels. If the pair of adult animals is
stable, they do not take part in mating duels and behave aggressively to contenders.
In the second half of spring the females give birth to cubs covered with brighter
and monotonous fur (melanistic gelludas appear in population rarely); usually
it happens in secluded place, in natural shelter or in specially made lair.
The female rears them with milk for the first two or three months, but in the
middle of summer young animals leave their home forever, and at the autumn they
are ready to follow their parents and to accompany them on the hunting.
Life expectancy of this species is 20-25 years.
This species of mammals is discovered by Bhut, the forum
member.
Translated by Bhut.
Squirrel
mungo (Mungartes scandens)
Order: Carnivors (Carnivora)
Family: Viverrids (Viverridae)
Habitat: tropical rainforests of East Madagascar.

Picture by Timothy Donald Morris
During the climatic changes marked the change of geological
epochs, the area of tropical forests of Madagascar was reduced up to several
isolated sites in river valleys. Large forest species like Malagasy fossa had
died out, but some small omnivorous predators had successfully survived. Among
them there were rather small Malagasy mongooses (Galidictis), whose descendants
have added new fauna of Madagascar.
In forests of Madagascar of Neocene epoch small animals can not feel like in
full safety – at night one of local predators goes to hunt. Medium-sized mammal
looking like squirrel quickly skips from one branch to another. It is about
cat-sized, but looks larger because of long fluffy tail. With the help of such
tail animal can change direction jump and support balance on branches and in
jump. This creature is a squirrel mungo, one of Neocene Malagasy predators,
ruthless hunter of any animals weighting up to 3 – 5 kgs.
The skin of squirrel mungo is rusty-red, patterned with wide longitudinal strips
of black color, breaking on shoulders to separate strokes. Tail is red with
white tip, on eyes there is white “mask”; hands and feet are dark grey.
Paws of squirrel mungo are rather long – in proportions animal looks like huge
squirrel. Claws are rather short, but fingers and toes are long and tenacious
- animal is good tree-climber and spends the whole life in forest canopy.
Muzzle of animal is extended; ears are short and rounded. Canines are long,
slightly juiting out from mouth. Squirrel mungo takes prey by forepaws, presses
to branch and kills, putting deep bites to it. Squirrel mungo eats reptiles,
birds and small mammals, and can attack even lemur cubs. It also supplements
diet with bird eggs and insects. This predator eats small prey like squirrel,
holding it in forepaws.
Squirrel mungoes live and hunt in pairs keeping together till some years, and
sometimes till the whole life. Hollow of large tree, especially if the trunk
is rotten from within to needed depth, is best dwelling for these animals. In
afternoon time squirrel mungos sleep, and right before evening they begin the
restless life: stretch, cleaned wool for some time, and go to search for food,
renewing boundary marks in passing. Animals mark borders with musk secreting
by glands in the basis of tail. For this purpose this mammal nestles back against
tree trunk or branch, and rubs against this place for some time. Usually the
couple of animals marks territory in common, putting a personal mark near to
partner’s one.
The pair protects its fodder site in common. If the stranger interferes the
site occupied by couple of animals, lawful owners of territory meet it, loudly
squalling and keeping nervously shivering tails up. Usually single animal does
not get involved in fight with breeding couple and leaves another’s possession.
Two times per one year the female brings posterity; in litter there is one cub
differing from adults in monotonous reddish colouring without spots. It spends
first two weeks of life in shelter (usually in tree-trunk hollow or in abandoned
bird nest). When it gets stronger, parents start to train it in hunting receptions.
Young animal keeps with parents almost up to the birth of the next cub at them.
Shortly before delivery female starts to express aggression to its own cub.
She can even kill it, protecting the future posterity if the cub appears not
ready to begin independent life.
The young animal reaches about 2/3 of the weight of adult animal to the moment
of exile from family. At the age of half-year young animals occupy their own
territory and after two more months can bring posterity.
Sugar mungo (Floromungo flosculiphagus)
Order: Carnivors (Carnivora)
Family: Viverrids (Viverridae)

Picture by Alexander Smyslov
Habitat: tropical rainforests of Eastern Madagascar.
The tropical rainforest gives the huge variety of kinds of food to the inhabitants;
therefore it is possible to meet the set of highly specialized species of animals
here. Among them at times there are species depending on the most unexpected
food sources.
Predators turn to vegetarians rather seldom. Pandas, partly bears, and the
African palm-nut vulture among birds are the examples of this phenomenon known
in human epoch. But in Neocene in Madagascar forests the descendant of carnivores
evolved, which quite may add this list with itself – sugar mungo. This species
of former predators lives in canopy of Madagascar rainforests at the great
height. The small mammal with bright red wool and long tail quickly clambers
among lianas and branches. It is similar to any rodent or small lemur, but
at close examination first impression appears deceptive. It differs from primates
in claws and short fingers and toes bearing characteristic small pads. Also
it is lack of large incisors being a “card” of rodents.
This animal moves in a manner characteristic for predators: it quickly jumps
among branches, frequently sniffing. And it seems that sensitive long nose
prompts its owner where the food is. In several jumps small predator reaches
the purpose: branches of plentifully blossoming tree. Having found recently
opened labiate flowers emitting aroma with slight putrefactive shade, little
mammal thrusts head in flower. In the weight it lowers the bottom petal, densely
pressed against top one, and narrow muzzle allows it to penetrate deep into
flower fauces. Little mammal does not search for small animals it can easily
cope with – it feeds on nectar. When it pulls head out from flower, its nape
appears powdered with brownish pollen. Having cleaned and having sneezed squeaky,
sugar mungo gets into other flowers by turns, licking nectar from them and
making pollination in passing.
Sugar mungo, perhaps, is one of the smallest and most specialized species of
carnivores; it is the descendant of Malagasy mongoose (Galidictis). Having
adapted to life in forest canopy, it has turned to skilful acrobat, and small
size allows it to clamber easily on thinest branches. It is only about rat-sizeв
one, though it seems larger because of longer fluffy tail. Bright red wool
of sugar mungo is short and rich. Throat and breast are white, bordered with
thin black strip. On back there passes black longitudinal strip; sometimes
it is supplemented with pale faltering parallel strips on each side. Tenacious
paws of animal are supplied with sharp bent claws, allowing clambering on branches
and tree trunks. Tail serves as a counterbalance when this mammal moves on
lianas and thin branches.
Head is extended and is on rather long and flexible neck. Brown eyes are large
– sugar mungo has good color sight; it distinguishes the most part of colors
(except for dark blue) and especially likes to feed on red flowers. Muzzle
of animal is long and narrow, slightly lowered downwards; on each side of nose
long whiskers stick out. Teeth are small and short – sugar mungo eats firm
food seldom. But tongue of this animal is a principal organ of food getting.
It can be put far out from mouth, and on its tip there is brush of fibers,
assisting to gather nectar. In addition to nectar sugar mungo eats friable
pulp of ripe fruits. Animal licks it by tongue from cracks in peel, or tears
peel of fruit by claws for feeding. Occasionally sugar mungo catches tiny insects
and slugs, supplementing a diet with necessary animal protein. Pregnant and
nursing females, and also young animals especially frequently catch invertebrates.
Sugar mungo is a solitary animal and is active mainly in afternoon time. Females
of this species are very much adhered to the territory, and each individual
carefully marks borders of territory with musky smell. Males lack musky glands,
and they have no constant territory which may be marked at all. Every male
lives at the large site including some territories of females. Males are more
tolerant, rather than females, concerning to each other and some animals can
even spend the night in the same hollow.
This species brings posterity up to 3 – 4 times per year. In a litter there
is only one cub, twins are rarity. The young growth is born rather advanced,
covered with short wool. It is remarkable, that at this species habits like
at cuscuses and lemurs were developed: the grown up cub leaves nest very early
(at the age of one week, just having opened eyes), and gets on back of mother.
It travels so approximately two weeks more, and then starts to follow mother
independently. At this time it remembers species of plants having nectar-bearing
flowers. At the age of two months it becomes completely independent and leaves
mother which already bears the new cub at that time. Five-monthly sugar mungoes
are already adult and also are capable to breed. Life expectancy is short,
as well as at all small mammals: no more than 5 years.

Picture by Carlos Pizcueta (Electreel)
The related species also lives in rainforests of East Madagascar – it is bee
mungo (Floromungo apiphilus). This species differs from sugar mungo
in dim brown colouring of the top part of body with longitudinal black strips
on sides.
Spots on its throat are yellowish and rather small.
The diet of bee mungo includes mostly honey which it steals in bee nests,
and also larvae of these insects which represent the main source of protein
of
animal origin in its diet. It is dangerous to get such food: bees can kill
small animal, having stung it enough of times. But bee mungo uses the special
adaptation to ravage bee nests: it has strongly advanced musky glands, and
the smell of their secretion frightens insects off. Getting into bee nest,
animal lets out some drops of musky liquid. The extending smell forces bees
to leave nest and to get off in dense clusters on branch at some distance
while the animal quietly licks honey and eats larvae in their nest. In other
features
the biology of this species is similar to biology of sugar mungo.
Long-bodied pterogenettula (Pterogenettula prolonga)
Order: Carnivores (Carnivora)
Family: Civets (Viverridae)
Habitat: Equatorial Africa, the top level of rainforest canopy.

Picture by Alexander Smyslov
Development of various anatomical adaptations at live beings
depends on features of their environment. And the generality of inhabitancy
results in formation
of similar adaptations at unrelated species. It has taken place in woods
of Equatorial Africa where on top-sized trees many species of mammals able
to
make long jumps were developed. If the forest canopy is dense, in it tree-climbing
animals receive advantages. But in friable multileveled canopy experts in
long jumps dominate. And on top of food pyramide there is also living “glider
pilot”,
but it is a true predator.
Predator of the top level of African woods is the long-bodied pterogenettula,
small animal with extended body. It had descended from one of species of
wood civets managed to go through the time of active hunting in last equatorial
woods of Africa.
Body length of this predator is about 30 cm at all, and tail lenght is about
20 cm. Externally this animal is similar to any small weasel: at the pterogenettula
there are rather short legs and the extended head. The animal has “marble”
spotty colouring of large ring-formed spots on light background. It makes
this predator imperceptible on the background of bark spotted with shadows
of leaves.
On head there are dark strips between eyes and nose, needing for recognition
of neighbours.
Extremities are rather short, supplied with tenacious claws. But the most
remarkable feature of appearance of these small mammals is the presence of
small flying
membrane between front and hind legs. For improvement of its “flying” properties
on edge of membrane the fringe of black fluffy fur (as on sides at Holocene
monkey Colobus) is stretched. The long tail of animal serves as rudder. Skilfully
using force of wind, the pterogenettula can make jumps up to 20 meters long,
losing in height only 8 - 10 meters. Operating flight, the pterogenettula
can even catch small birds, preying them in sudden jump from an ambush. If
attack
was unsuccessful, the animal simply continues a jump, and sits on a tree.
Flying up to the final point of jump, the animal weakens flying membrane,
and seizes
by claws of all four paws branch or bark of tree. Also this predator is able
to swarm up trees dexterously, being not inferior to any squirrel in speed.
The pterogenettula frequently hunts also a way traditional for predators,
chasing catch among branches.
Favourite catch of the pterogenettula includes birds, but also it frequently
hunts lizards, squirrels
and squirrel
guenons - dwarf monkeys of forest canopy.
This animal is strict solitary predator. Each individual chooses to itself
the certain site in forest canopy, reaching not only in sides, but also upwards.
Borders are marked by odorous secretions of specific glands of anal area.
Seasonal prevalence in breeding of pterogenettulas is not present, and young
animals may be met at any time of year. Per one year the female brings 2
packs (4 - 5 cubs in each litter). Young animals differ in darker colouring
(light
intervals between dark spots so are small, that colouring seems mesh-looking).
Approximately at four-monthly age young animals start to study “flying” -
to this time the flying membrane at them develops to the full. Sexual maturity
comes at them at the age of 8 – 9 months, and life expectancy may be about
6 years.
Zibetonyx
(Zibetonyx velox)
Order: Carnivores (Carnivora)
Family: Civets (Viverridae)
Habitat: steppes of Central Asia (to the east from Fourseas).
Evolution proceeds by certain principles and laws which were just in the past,
and undoubtfully will be just in the future. Therefore it is quite logical
to expect of recurrence in the future of some phenomena taking place both in
the far past, and within people.
One of such phenomena, convergence with representatives of canine family (Canidae)
and Neocenic African predator pardinia, is demonstrating by zibetonyx, the
inhabitant of plains in Central and Middle Asia. This gracefully build predatory
animal is the descendant of mongooses (Herpestes) widely settled in Asia. Ecological
plasticity of these small predators has permitted them to occupy temperate
steppes of Asia successfully. And specific conditions of inhabiting - open
plains with high grass - had generated shape of new predator.
Being quickly running predator, zibetonyx has characteristic shape: at it there
are long legs and flexible backbone, small head on rather long and mobile neck.
Jaws of zibetonyx are long and rather weak, its head resembles canine one.
As against cats and pardinia very similar to it, the zibetonyx kills catch
not by one sting: the pack inflicts to the chosen prey numerous wounds and
exhausts it by long pursuit.
At zibetonyx there are sharp sense of smell and good hearing: animal frequently
should hunt in high grass where sight can be practically useless. Ears of zibetonyx
are rounded and wide, but they are located at head sides as at its ancestors.
On the top part of neck the mane especially big at the adult male grows.
Colouring of wool is yellowish with large brown spots merging on sides and
back to short longitudinal dabs. Up to winter (in habitats of zibetonyx snow
does not fall, but weather becomes appreciable more cold in winter) wool becomes
longer and then the mane of males is almost not appreciable.
Zibetonyx is ecological analogue of wolf, but a little bit reduced size. Animals
of this species hunt in packs up to 10 – 15 animals preying approximately goat-sized
herbivores.
The pack of zibetonyxes represents clan with expressed social connections and
hierarchy. Clan is formed of posterity of one parental pair: all females from
packs remain in clan, and maturing males leave it. Females in clan appear connected
by consanguinty and represent two – three generations of one family. Males
come to clan “from the side”, and between them and main pair (clan founders)
there are relations of submission. If friction between males appears strong,
clan can divide at all.
The clan is original “super-organism”: harmonious collective hunting is much
more effective, than single’s one. Zibetonyxes are specialized for hunting
quickly running herbivores: usually harelopes are their catch. However the
important place in menu of zibetonyx is occupied also with small catch: animal
willingly eats lizards, birds and large insects – beetles and grasshoppers.
Main way of hunting is prey chasing. For this purpose zibetonyxes from one
clan stay in line and hide in grass at some distance from each other. Some
individuals (as a rule males are they) drive harelope out from herd and chase
it to animals waiting in ambush. “Passing on” chased animal to each other,
predatorss absolutely not for long time take part in hunting but can “give
all the best” completely, similarly to cheetah. Because these animals combine
hunting tactic of wolf and speed of cheetah it has allowed them to become successful
and prospering plain predators in hunting.
Daily life of clan takes place near natural shelters – small groups of bushes
in which females arrange little lairs and practically in common rear young
growth. Here animals have a rest after hunting. Near shelters zibetonyxes keep
rather carefully and silently, and do not hunt here to not unmask place where
cubs are reared. But borders of clan are plentifully marked by urine and musk
secretions of repugnatorial glands. Usually adult males put marks, placing
them on big stones, bushes and trunks of lonely growing trees.
Cubs (2 - 3 in pack) are born blind and covered by short wool. Their colouring
is very dark: brown, paws and ears colored black. For the fourth day of life
at them eyes open and then they start to hear. And ten-day cubs already try
to walk. Only first two weeks of life young zibetonyx knows exclusively care
of native mother. Growing up, it becomes object of gentle care at once of several
females of clan, including old ones which can not bear posterity any more.
They play role of “baby-sitters” when clan goes hunting. Such feature raises
chances of survival even at deserted cubs. One female is capable to bear posterity
twice per year.
At the age of four months young animals already change youthful dark colouring
to spotty adult one. They study to hunt about two months, and then when females
of clan have new cubs, young males are driven away. They can live vagrant life
on border of territories of several clans, satisfying by remains of their catch.
Frequently young males form independent packs try to drive out adult males
from any not numerous clans, or to drive off young females from near clans.
Usually bachelors are much more aggressive than males have formed pair. They
easily get involved in fights though they can suffer serious loss from strong
clans: females protecting posterity can fight up to death. Usually many young
males perish in such skirmishes, and sex ratio in generated and breeding clan
is 1 male for 2 – 4 females.
Sexual maturity at females takes place at second year of life, at males – at
third year. Life expectancy is about 15 – 18 years.
Mauritian paralutra (Paralutra nana)
Order: Predatory (Carnivora)
Family: Civets (Viverridae)
Habitat: rivers of Mauritius Island.
The fauna of Mauritius Island had strongly suffered from people influence.
Many species of animals representing groups which could not appear on island
by natural way had been introduced to the island in different times. These
animals were monkeys, rodents, various predators and domestic hoofed mammals.
Such inflow of species new to the ecosystem of the island had taken place in
very short term according evolutionary measures – within several centuries.
As a result the native fauna and flora of island had degraded. People could
interfere with influence of introduced species to the endemic nature of island
with the help of nature protection actions till some time. But after the disappearance
of mankind the rests of island primordial nature were destroyed by introduced
species. In Neocene flora and fauna of Mauritius are presented mainly by descendants
of introduced species had formed the complete and balanced ecosystem. In most
cases inhabitants of islands differ in diminutiveness, and only few species
of reptiles and birds are the exception. Descendants of introduced species
had occupied all ecological niches were given to them by the nature.
Rivers of island are shallow and short. Descendants of the fishes introduced
by people – true gourami (Osphronemus goramy) and goldfish (Carassius auratus
auratus) live here. Occasionally sea fishes swim into the rivers of island,
and there are some species of river shrimps and crabs. These species make the
basis of food pyramid at which top there is one endemic species of predators
adapted to life in water – the Mauritian paralutra. This animal is the descendant
of mongooses (Herpestes) introduced to the island and perfectly swimming predator.
Paralutra eats any water animals which can be caught, and supplements a diet
with tubers and sappy leaf petioles of water plants.
Mauritian paralutra is a small animal. This is the natural phenomenon in the
world of small island, where resources and vital space are strictly limited.
Length of adult animal is about 60 cm including long tail. By appearance and
constitution paralutra resembles true otters (Lutra) of Holocene epoch: this
is a short-legged animal with flexible body and membranes between toes. Fingers
of forepaws of paralutra are not connected by membrane. They are sensitive
and mobile, and have small claws. With their help paralutra searches in thickets
of plants for shrimps and small fish. On the coast paralutra clears and combs
fur by fingers. The wool of animal has grey color, on throat there is white
spot, and cheeks are also white.
Head of paralutra is flattened, with short wide nose and long postorbital part
of skull. Eyes are shifted to the forward part of head and upwards, therefore
animal can breathe and look over vicinities, having put out from water only
a part of head. Above the nose of animal there is a site of hairless skin with
which help paralutra digs bottom in searches of crustaceans and roots of plants.
External auricles at paralutra are lack, and the ear aperture at diving animal
is closed by ring muscle. Tail of paralutra is long, flattened, covered with
naked skin. At movement under water animal presses forepaws to body, and swims,
wavy beding by all trunk. For slow and maneuverable swimming among underwater
thickets animal uses hinder legs.
Paralutra eats small fish, not attacking large fishes. Hunting animals try
to push aside school of fishes to the bank and attack them in common. Also
this animal eats shrimps and crabs, and ravages nests of coastal birds.
Musk glands located under tail produce plentiful oily secret with which help
paralutra makes its fur water-repellent. To not frighten off fish, this product
of secretion till the process of evolution had lost smell that is completely
atypical for civet. This mammal smears secretions of musk glands on forepaws,
and pounds secretions on fur, carefully rubring them in hairs and combing them
by claws. Ritual of mutual fur caring helps to support friendly relations between
the representatives of species living together.
This is sociable mobile animal. Paralutras settle on steep riverbanks by whole
families including some females connected by consanguinty, and males not related
to them. The family of these animals digs out some inhabited holes, one of
which is occupied with main pair. These animals breed the year round. The female
can bring posterity two times per one year, 3 – 4 cubs in one litter. Preparing
to birth of posterity, female deepens the hole and digs out wider inhabited
chamber, rather than usually does. Cubs are born blind and deaf, covered with
short dark wool. They start to move in hole at the age of about 12 days, and
monthly cubs already are able to swim as well, as adults. At the age of 3 months
young animal becomes completely independent; half-year old female already gives
rise to posterity.
Paralutras constantly support contact with each other with the help of voice.
They can utter various sounds – they chirr, peep and whistle. At danger animals
yelp like small dog. After feeding animals gather and clean wool to each other.
At the rest paralutras are cautious and prefer to gather at shallows and small
islands in the middle of the river where it is impossible to creep to them
imperceptibly. At suspicious sounds and sudden occurrence of large animals
at the riverbank they disappear under water. Main enemies of paralutras are
maurosmiluses – small saber-toothed ground predators of feline family; in water
some sea fishes swimming to rivers of island can attack these animals.
Grumbling
ant-mungo (Myrmungopsis myrmivorus)
Order: Carnivores (Carnivora)
Family: Ant-mungoes (Myrmivoridae)
Habitat: Atlas Mountains, Middle East, Southern Europe (coast line of Mediterranean
swamps).

Picture by Tim Morris
Some species of live creatures have success in struggle for
existence due to development of habitats and food resources inaccessible to
other species. This
mammal species belongs to such animals. It is the representative of new family
of Neocenic mammal, eating exclusively social insects. The ant-mungoes family
had descended from civet family: some small species of family had specialized
in feeding by insects, and further this specialization has resulted in occurrence
of new group of animals. Any species of small mungoes - animals proved their
success in struggle for existing - became direct ancestor of representatives
of present family.
The ant-mungo during evolution has got common for insectivorous animals appearance
features: at this animal there are wedge-shaped extended head, small mouth,
reduced lower jaw and teeth. The basic feeding organ is tongue, capable to
extend forward approximately for one and half lengths of head. Nostrils are
located on tip of muzzle, during the feeding process they close and animal
holds the breath. Claws on ant-mungo forepaws are long, during movement the
animal turns them in and supports on the back side of hand similarly to anteater
or gorilla (in these places “callouses” of dense thick skin develop). Claws
are used for digging insect nests and for protection against enemies. The
wool of ant-mungo is short, dense and rigid, bristle-like. It protects animal
from
stings of insects. Besides animal has significant immunity to insect poison.
Colouring of wool is dingy: monochromic gray-brown, stomach is light grey,
legs and tail are darker. Forepaws are shorter then rear legs; at movement
the back of animal is inclined forward. Digging out an ant hill or termitary
ant-mungo can stand on hinder legs for a long time.
The grumbling ant-mungo reaches length 40 cm (tail is up to 30 cm) and weighs
about 5 kgs.
Ant-mungo leads social habit of life differing
in it from other Neocenic “anteaters”. This species lives in families of
4 - 7 animals (breeding pair of adult animals and cubs of the current year).
Family occupies rather extensive territory with thickets of bushes and light
forest where many nests of termites and ants are located. Except for them
animals
eat small snails, swallowing them entirely with shell, and also larvae of
beetles and caterpillars of butterflies. Animals communicate among themselves
with
the help of sounds similar to rumbling and grunt.
This animal lives in hot, almost waterless areas, therefore it shows greatest
activity in twilight. The group of ant-mungoes spends day in temporary hole
dug on the eve on morning, and at the sunset leaves it for feeding. In the
evening animals eat mainly ants gathering in ant hill for the night, at night
– by termites coming on surface, and gather snails crept out on plants covered
with dew in the morning, or search for beetle larvae under stones and tree
bark.
Pairing of ant-mungoes occurs approximately for two months before rain season,
and cubs are born just in this season. It is justified biologically: milk
producing at the female takes plenty of water, and in rain season it is easier
to fill
up internal water stocks. Once a year, in the beginning of rain season female
gives rise to five - eight cubs. They are born blind and deaf, female hides
them in specially dug hole. She feeds posterity, coming back in hole some
times for night. At this time male shows original care of her: he gorges
on for future
use, and then in addition feeds up the female, belching food to her mouth.
At one month after birth the young growth already can follow adult animals,
and the female actively trains posterity to search for food. Usually pack
stays with mother up to the following season of pairing when the parental
pair banishes
posterity from the territory. The young growth reaches sexual maturity at
half-year age, and life expectancy is about 8 years. Main enemies of grumbling
ant-mungoes
are predatory mammals and snakes, but most of all these animals perishes
during long droughts.
Awfulest marafil (Marafil perhorridus)
Order: Carnivores (Carnivora)
Family: Hyenas (Hyaenidae)
Habitat: South Africa, mountains and plains.

Picture by Alexey Tatarinov
Improvement by Carlos Pizcueta (Electreel)

Picture by Eugeny Hontor
In Neocene South Africa appeared separated from Holarctic zoogeographic
area by the zone of impassable tropical woods. They had appeared when East Africa
(Zinj Land) had separated from the main part of the continent, and the area
of dry climate had considerably reduced because of evaporation of water from
Tanganyica Passage.
The majority of groups of herbivores appeared in Neocene had not got here; therefore
Southern Africa became one of places where many representatives of hoofed mammal
fauna characteristic for Ethiopic zoogeographic area had kept. In South Africa
in savanna descendants of water chevrotains
similar to deer wander, and tall descendants
of dwarf duiker antelopes similar to primitive giraffes browse foliage of
trees. Representatives of animal kingdom of new epoch – huge descendants
of pigs armed with massive bone outgrowths on head – also are found here.
By the efficiency ecosystem of Southern Africa does not concede to North African
savannas of early historical epoch.
The abundance of catch involves various predators, and relative stability of
this region had allowed to some representatives of Holocene fauna of predators
to be kept in South Africa. Among civets and cats of South Africa one local
predator looks separated – it is the last representative of hyenas’ family.
This family had been widely distributed in Eurasia in Holocene. Till the ice
age some of its representatives had wandered in snow-covered woods of Europe,
tolerating severe winters. But after mass extinction of hoofed mammals in the
areas mastered by people the area of family was sharply reduced. In historical
epoch the family was submitted only by five species, one of which was extremely
rare.
Awfulest marafil (“marafil” is the Arabian name of hyaena), the descendant of
spotted hyena (Crocuta crocuta) widely settled in past, lives in South Africa
This animal represents the deification of “predator” concept, doubtlessly occupying
the top of food pyramid in local ecosystems.
Awfulest marafil is the largest species of hyenas among ever dwelt on Earth,
and, obviously, it is the largest representative of the carnivore order in Neocene.
Growth of the adult female at a shoulder reaches two meters at weight about
700 kgs. Male is smaller, rather than female: it weighs about 500 kgs and differs
in lighter constitution. By the appearance marafil resembles fossil Arctodus
bear dwelt in Pleistocene at the territory of North America. The female of marafil
differs in sloping back (back legs are shorter a little, rather than front ones)
and wide chest. Forepaws at it are very muscular – with their help the animal
knocks prey off feet. Feet at the female of marafil are rather wide due to what
animal can chase prey on marshy ground. At the animal there is short tail with
hairy brush of lengthened hair on the tip.
At marafil there is short head with powerful high jaws. Canines are rather short;
molars and premolars of animal work as crushing tool – marafil female can shatter
the backbone of rhinoceros-sized prey by one bite.
The skin of marafil female is covered with rough short wool of brown color.
There are vertical strips of black color on shoulders and sides and some black
spots on hips (sometimes they may be absent). Wool on head of animal is black,
but around of mouth there is a strip of white wool – this attribute has the
important value at the communication of animals. Marafils, especially females
of this species, are rather aggressive animals, and in many cases it is vital
for them to be able to distinguish mood of the congener from afar to behave
in appropriate way.
Very sharp sexual dimorphism is characteristic for marafils. It is shown not
only in difference of body sizes, but even in colouring. Skin of marafil male
is grey with numerous black spots merging on shoulders to vertical strips. On
the neck of male there is a mane of long grey hair. Males look more harmonous
and long-legged, rather than females and can run much faster. Also at the male
there is longer and narrow muzzle with more advanced canines which partly jut
out from closed mouth.
Males live and hunt in packs of ten individuals, living mainly at the boundary
of territories of female clans. They do not make constant refuges, and spend
the night in the same place no more than two nights in succession. Females of
marafils, on the contrary, make constant lairs in bush and come back there every
day. They lodge in clans of 4 – 5 individuals connected by consanguinty (as
a rule, it is the female and her senior daughters). The number of marafil males
in population is smaller, than the number of females: it is connected with the
high mortality level of these animals at young age. When young marafils grow
up, young females dominate over males compelled to be content with the rests
of their catch. When the young marafil male reaches about one-year-old age,
it is expelled from clan, and some time it leads single life, eating the scraps
had remained after hunting of parental clan. Groups of marafil males can unite,
forming male packs.
Sexual dimorphism of marafils is shown also in hunting preferences: there are
different catch and different hunting tactics at males and females. Females
chase mainly sluggish catch – huge bullhogs. Males prefer to hunt swift-footed
hoofed mammals, badgering them by pack like wolves. Hunting habits at different
genders also differ – females hunt at night, attacking resting and feeding animals,
and use during hunting sharp sense of smell. Males hunt in day time with the
help of sight.
Marafil females being in group are very aggressive to neighbours. It happens
they can attack males of their own species, kill and eat them as if other prey.
Therefore pairing at marafils occurs outside of territory of female clan. For
pairing males mark with urine objects well appreciable from afar (for example,
lonely growing tree, large stone or termitary) at the edge of territory of females,
and wait for female near this place. Thus they drive other males off from the
marked sign. Also males leave some of labels at the territory where the female
clan lives. Usually they do at the afternoon before females will leave on hunting.
But at the same time males try to keep together and to be on alert. They succeed
to have a sleep only in second half of night and in the morning approximately
till midday when females come back home from hunting. The female ready to pairing
leaves clan, finds the site marked by males, and males pair with it by turns.
Before pairing the female shows absence of aggression to males: she lowers head
and presses ears. Males showing the superiority run near her by whole pack with
lifted heads and occasionally bite the female to shoulders and groats.
Similarly to all large animals, marafils breed seldom: each female gives rise
to posterity alternate years. Pregnancy lasts about half-year, and cubs are
born to the beginning of rain season. They suck milk till four – five months,
but start to try meat food at the third month of life. In litter at one marafil
female it happens three – four cubs. Usually only half of posterity survives
up to independence.
Young females become able to breeding at the age of 4 years, and males since
3 years. Life expectancy at females of this species reaches 50 years and more,
and males seldom live up to 40 years.
Awful daggerclawer (Pugionyx deimus)
Order: Carnivores (Carnivora)
Family: Cats (Felidae)
Habitat: Eastern Asia, Far East (area of moderate and subtropical climate),
flatland woods, foothills.

Picture by Sauron from FurNation
In Holocene the cats family has suffered the big damage from human activity.
Number of large cats was sharply reduced, and their areas have catastrophically
decreased. Small cats have suffered from hunting for the sake of valuable
fur. The unique representative of family has received the big advantages
in a survival
in human epoch is the domestic cat Felis catus. Due to people this predator
was settled practically in whole world. It became a stimul for evolution
of the present species. As against the majority of domestic animals dependent
on people, the domestic cat easily runs wild. Therefore after disappearance
of people this species have not died out in common with them, and have continued
to exist. The majority of Neocene felines descend from several species of
rather
small cats have survived hunting and ecological crisis.
Cats are successful predators; therefore descendants of domestic cats have
taken the high place appropriate to their abilities in food pyramids of new
biosphere. And in Neocene among them large animals from the number of the
most terrible land predators of Earth have appeared. One of them is the awful
daggerclawer, specialized large prey hunter.
It is very large cat: it is lion-sized, but it seems larger because of longer
legs. At daggerclawers characteristic features of appearance, common for
cats, were kept: rather short jaws, large canines, binocular sight. Daggerclawers
are solitary animals.
The body of daggerclawers (in the Neocene world there are few species of
these cats) is covered with fluffy wool. At the awful daggerclawer the colour
of
wool is similar to “tortoise-shell” color of some domestic cats - on grey
background there are big, almost symmetric dark spots. Tail of animal is
long, on it spots
merge to non-uniform cross strips. A tip of tail of animal is white. White
sites are on the bottom part of muzzle; throat and breast are also white.
On hinder legs of animals of this species occasionally there are white “stockings”.
Forepaws of daggerclawers are colored contrastly and brightly: from an elbow
up to fingers they are covered with alternating wide black and white strips.
This colouring is the important element in behaviour of animals as it is
used
in rituals of an establishment of domination. It is shown only at adult animal
of both sexes. Not sexual matured animals have dark forepaws.
At capture of catch cats actively use forepaws. And this feature of behaviour
has received development at the present species and has reached high specialization.
On paws of cats there are five fingers from which four ones base on the ground,
and the first, the shortest one, does not touch ground. At daggerclawers
on first fingers of forepaws crescent claws of huge size (its length is up
to
20 cm) were developed. In rest this claw is pressed to the paw and its end
is directed back, not preventing walking and running. On the internal sides
of wrists of the animal there are sites of very thick cornificate skin, and
this claw adjoins directly to this one. Also this claw is partly retracted
in skin cover. The “fighting” claw of the daggerclawer is partly similar
to claws of small predatory dinosaurs like Deinonychus and Velociraptor.
The daggerclawer eats large ground animals. Attacking prey, it at first creeps
to it to probably closer distance, and then rushes to the planned victim.
Due to long legs it can accelerate momentum up to 60 kms per hour on the
short
distance. At the moment of solving jump the animal shows its “fighting” claws,
and puts to the victim deep wounds by them. When impact has falled on the
neck, catch of the daggerclawer, even having escaped from its claws, quickly
bleeds
profusely and perishes. In case of successful attack the daggerclawer, except
for impact by claws, delivers to prey sting in neck or in the basis of skull:
this is the “inheritance” stayed from its ancestor, the domestic cat.
All daggerclawers are solitary territorial animals. Usually at them as at
tigers, the territory of the male includes territories of several females.
Animals
mark territory, fleecing bark from trees, and rubring the bared wood with
a secret of chin repugnatorial glands. Males also mark the territory by urine.
During an establishment of domination relations animals pay special attention
to colouring of forepaws of the contender: the more contrast coloring, the
animal having it is stronger. At young animal paws are dark; at old ones
they
are “gray-haired”: strips become not so contrast. Duels for territory and
place in hierarchy, as a rule, are limited by demonstration of “stretching”
of contenders:
thus animals by turns bend backs and extend forward forepaws, showing their
colouring. “Stretching”, each animal tears off by claws a part of grass,
breaking through deep furrow by “fighting” claw. Such ritual behaviour helps
to avoid
fight, and contenders can estimate force each other. It is accompanied by
loud mewing and squeal.
Pairing occurs in middle of winter, kittens (2 - 4) are born in first half
of spring. They are covered with dark wool, blind and deaf. The female feeds
with them with milk about four months, and then even more half-year young
animals keep with mother. They abandon her in winter, and more often they
are banished
by males during a rut.
Sexual maturity comes approximately at three-year age. At this time at males
strips on forepaws start to appear. But most successfully males are coupled
approximately since the fifth year of life: females give obvious preference
to males with striped forepaws.
Life expectancy of the awful daggerclawer reaches 30 years.
There are some other species of daggerclawers distinguished in smaller size
in East Asia in moderate and subtropical latitudes:
Fisher daggerclawer (Pugionyx piscivorus) is
a predator up to half meter at a shoulder. Length of its body is about one
meter; a tail is about 60 cm. The
head is dark with white area on the tip of muzzle and throat, above eyes
there are white spots; wool has striped colouring. Forepaws up to an elbow
are white,
occasionally on them there are one - two cross strips. The claw is rather
larger, than at the awful daggerclawer: its length is about 15 cm. In rest
it is strongly
unbend back, resembling cock heel.
The animal inhabits marshlands, riverbanks and lakes. The fisher daggerclawer
willingly swims and prefers to not leave water far. The animal eats fish
and aquatic animals: turtles, frogs, waterfowl and rodents.
Bird-catching daggerclawer (P. saltatus) is
the smallst species of genus (length of body not including tail is about
60 cm; tail is
about half meter
long). It is settled more widely than other species: this cat inhabits areas
from plains up to mountain woods. It also differs in especial addition: hinder
legs are strong, appreciably longer than forepaws (back is inclined forward).
Colouring of wool is spotty: on background from sand up to red there are
dark brown spots. Forepaws armed with sharp crescent claws are colored black
with
white spots. This animal eats mainly birds. It dexterously forces down flying
up birds, killing for one jump up to five ones from flight. It eats catch,
keeping it in forepaws and sitting on back legs (by same habits Holocene
caracal (Felis caracal) had differed).
The idea of the present animals is prompted by Simon, the participant of forum.
Menkw (Parafelis menkw)
Order: Carnivors (Carnivora)
Family: Felids (Felidae)
Habitat: mountain forests of Altai, the Far East.

Picture by Amplion, colorization by Alexander Smyslov
Исходное изображение. Рисунок Amplion |
Among pets only few species had gone through disappearance
of mankind. Selecting breeds of various domestic animals, people had changed
their physiology and behaviour so deep, that many breeds appeared as a result
of domestication were completely unable to exist independently. But nevertheless
there were the exceptions – primitive breeds of domestic animals ran wild easily.
Also the process of adaptation to wild life passed successfully in conditions
of isolation and absence of predators – strange
sheep descendants
in New Zealand have appeared this way. But domestic cat (Felis cattus) appeared
the champion in adaptation to life in a nature, and people did not manage to
subordinate it to their will completely. Descendants of domestic cats live on
various continents and islands.
At the territory of Central and Eastern Asia extensive mountain areas are inhabited
by one of such species – menkw*, the universal predator, the expert in survival
in mountain forests.
In its ecology menkw is the analogue of leopard, but in shape it resembles lion
a little. The basic colouring of this animal is brown with a pattern of spots
and cross strips on paws and tail. The wool on trunk is almost monophonic, pattern
of vertical strips is present only at kittens to the age of about one year,
and completely vanishes during an autumn fall-off. Male of this species is larger
than female and has short small mane covering nape, neck and forward part of
breast. In autumn animals change wool to winter fur, longer and lighter in color.
The length of menkw body from head up to a tail is about 140 cm; tail is 90
– 100 cm long. Weight of adult individual may reach 75 kg, mature males frequently
weigh over 80 kg. This predator hunts any animals it can catch, from a rat up
to boar in size. This animal usually hunts from an ambush, suiting it near tracks
of herbivores in bushes or behind the fallen tree. This predator frequently
jumps on prey from tree, choosing for ambush thick branch among foliage. This
predator kills prey by strong bite in throat, simultaneously throttling it and
biting through blood vessels of neck by canines.
This species is territorial; adult male occupies territory measuring over 60
square kilometers. In male territory up to 5 females – its harem – and posterity
up to one and half years old live. Each morning male declares its rights to
the territory with loud call. It has no constant shelter and within several
days walks riond its territory, renewing border marks. For this purpose it tears
bark on trees and also waters it with urine. Nevertheless, these animals are
rather tolerant to each other: the meeting of two males out of breeding season
is usually finished only in force demonstrations. Males rear wool and yell loudly,
trying to impress the opponent, but enter fight seldom.
Pairing at menkw takes place in winter or in early spring. At this time boundary
skirmishes between males become more often and the number of fights grows. They
frequently come to an end with ragged ears and scars on shoulders. In especially
severe duels one of males can even receive fatal traumas. Pregnancy of female
lasts till 90 – 100 days; usually 3 – 4 kittens are born. For delivery female
chooses reliable shelter, as a rule, a tree trunk rotten out from within or
a cave. She feeds cubs with milk within 2 months. Kittens are born blind and
open eyes at the age of 3 weeks. The young growth of this species differs from
adult individuals with visible vertical strips on sides which vanish with the
years. They are independent at the age of 5 months and reach sexual maturity
at two-year-old age. With approach of this age females join the next groups
of menkws, and males should win territory and females to themselves, or wander
on boundary territories of stronger males. Life expectancy is about 35 years.
* Menkw is a forest goblin in mythology of Siberian folks.
This species of mammals was discovered by Nick, the forum member.
Felimalagasia
(Felimalagasia gracilissima)
Order: Carnivores (Carnivora)
Family: Felids (Felidae)
Habitat: Madagascar, various types of forests.

Picture by Amplion
When humans appeared on the Madagascar, they heavily damaged
if not outright destroyed the unique ecosystem of the island. The large animals
became extinct due to the hunting and deforestation. Destroying of natural habitants
at the end of the mankind era led to the extinction of fossa (Cryptoprocta ferox),
the largest predator of the island. The smaller viverrids survived but they
have become mostly land-based predators or small climbing animals. The ecological
niche of large climbing predator was given to another animal descending from
domestic cat. It has developed a set of adaptation to hunt fast-climbing and
careful prey and has become a beast who looks much like a huge marten. It’s
felimalgasia, the domineering predator of forest canopy occupying the top of
food chain.
Felimalgasia spends almost all the time in the trees but sometimes gets down
to the earth during the hunt. The build of the animal is adopted for fast movement
in forest canopy. The legs of felimalgasia are relatively shorter than those
of cats, the body is longer- the height of animal is only 50 cm while the length
is up to 150 cm without the tail. The spine is incredibly flexible- the long
neck permits to turn the head around on 180°, and before the jump the animal
can fold itself in two. The long tail serves as balancing pole during the jumps,
the movements of tail provide necessary balance.
Small head with short stout is inherited from domestic cats. The ears are relatively
wide, with rounded tips: the hearing of felimalgasia is very acute. The eyes
in the front of head provide stereoscopic eyesight which is really important
for tree-climbers. The mouth opens wide. The animal uses its columnar teeth
to kill birds with relatively fragile bones, biting them through the spine or
the base of scull. Big animals such as lemurs are killed with bites in the tissue
of the neck while being held in place with paws. Felimalgasia has relatively
good sense of smell, well-developed whiskers help the animal to orient in forest
canopy even in the dark.
The colour of fur serves camouflaging purposes. The main colour is a shade of
yellow, it’s covered with multiple wide spots, forming long lengthwise stripes
on head and neck. The pattern of spots is individual. On the tail stripes form
rings and the main colour changes from yellow to white. With the stripy end
of the tail felimalgasia sends visual signals to the others of her species,
but that animal being solitary rarely has need for communication save for the
mating season.
The usual prey of felimalagasia is lemurs and birds; though sometimes it’s smaller
animals such as frogs, lizards and even small insects.
The animal tags its territory by tearing the rind of the trees with its claws.
The territory of each adult animal is about 30 square cm. Only during the heat
the female tags the territory with urine which has specific smell during that
period. Felimalgasia rarely climbs down from the trees so the animal doesn’t
dig its feces, but urinates and defecates from the trees and feces fall down
to the ground. Because of that the inhabitants of forest canopy can’t detect
its presence, and hunt in the underbush happens relatively rarely.
The litter of female happens once in a year and includes up to 5 kittens, but
only two or three of them reach maturity. The female makes its den in relatively
large hollow or usurps big bird nest (for example the one of false
macaws) which is adopted to suit its basic needs. The kittens are different
from adults in colour, the dark spots on their fur are relatively bigger. The
kittens stay with their mother till they’re 5 month old – usually by that time
female has already mated with another male and is pregnant again. The maturity
is reached at the age of 2 years; the life span of felimalgasia is up to 20
years.
Translated by Charles, the forum guest.
Ngeru
hou (Australofelis postdomesticus)
Order: Carnivores (Carnivora)
Family: Cats (Felidae)
Habitat: New Zealand, North and South Island, forests, woods, bushland, mountain
forests.
Initially, introduced domestic cats became a feral pest in New Zealand and caused
many problems for native wildlife, especially birds and reptiles, but this creature
became a lesser player in the long term. Equally pugnacious and more adaptable
feral Possum (Trichosurus vulpecula), increased in size and became the dominant
predator of Aotearoa, Marsupial Pardus.
Feral cats instead formed an analogy of their ancestor, Felis sylvestris, becoming
the Ngeru-hou (“new-cat” in Maori). General appearance recalls a Wildcat or
a large tabby. Body is up to 60 centimetres long, with a tail up to 20 centimetres
long, relative shortness of tail is retained from domestic ancestor. Legs are
a little longer than that of a domestic cat, and skull is a little longer and
more robust. Coloration is a spotted tabby pattern on a light brown background,
with a dark ringed tail. Coat is shorter and paler, and animal is overall thinner,
in the sub-tropical parts of its range.
As with most wildcats, it is solitary, though it will sometimes tolerate others
of its own kind, as with its domestic ancestor. Diet consists of prey caught
from ambush, small and mid-sized birds, rodents, rabbits and reptiles, as well
as large insects such as moths. This cat is most active by night, and retreats
to sleep in secluded places by day. Vocalisations generally recall that of its
ancestor, yowls, hisses, mewling and meowing, but is somewhat louder and more
eager in tone.
Sexual maturity is reached at 10 months, female has one oestrous in March or
April, and gestation is up to 65 days. Mating and contests for mating result
in loud yowls and screams. Litter sizes vary from 3 to 8 kittens, which are
suckled in a den, usually under a bush, rock outcrop or tree roots. Kittens
learn to hunt at 10 to 12 weeks and are fully weaned by 14 weeks. They leave
their mothers at about 6 or 7 months but may remain in her territory a little
longer. Lifespan is generally up to 15 years but can be longer.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Island
Tigrillo (Arbofelis pseudotigrillo)
Order: Carnivores (Carnivora)
Family: Felids (Felidae)
Habitat: Galapagos Islands, main large island, forest.
Among the animals introduced to the Galapagos Islands was the Domestic Cat (Felis
catus), which has left some successful descendants here in the Neocene.
Island Tigrillo gets its name for its small size and the resemblance of its
coat to a Holocene kind of cat, the Margay (Leopardus wiedii). However, build
of this animal is different, legs are shorter and more flexed at the knee and
body is longer, almost resembling a Marten. Animal weighs about 2.5 kilograms
and body may be up to 45 centimetres long, tail about 40 centimetres long. This
cat spends most of its life in the branches and tops of trees; prey includes
birds, lizards and tree-climbing rodents as well as large insects. The spine
is very flexible and legs are bent, allowing the animal to make prodigious leaps
from tree to tree. Head is longer and more muscled than its ancestor, canine
teeth are slightly longer proportionally. Ears are large and rounded, bearing
fur on the outside, facial whiskers are well developed. Coat is yellowish in
colour with a pattern of spots serving as camouflage, resembling a leopard or
its namesake the Margay, tail is banded in black and yellow.
This animal is mainly nocturnal but may hunt in late afternoon and early morning.
They are solitary and maintain distinct territories; individuals will yowl loudly
to proclaim their territory. Animal marks boundaries with urine and does not
leave the trees to defecate.
Breeding occurs once a year, litters consist of up to 6 kittens, which are reared
in a tree hole lined with leaves. Kittens initially have eyes closed and are
darker than their mother in colour, but still bear the beginnings of spots.
Kittens remain with the mother for 3 and a half months, sexual maturity is reached
at 5 months old, individuals may have a lifespan of up to 10 years but usually
much less.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Galapagos
Nano-Otorongo (Tigrefelis ferox)
Order: Carnivores (Carnivora)
Family: Felids (Felidae)
Habitat: Galapagos Islands main large island, open habitats, scrub, woods, ground
dwelling predator.
It has occurred many times in the Neocene that introduced invasive predators
have produced apex predators in their new domain, and so we see with Domestic
Cats (Felis catus) in the Galapagos.
Apex predator of the here is the Galapagos Nano-Otorongo, a formidably built
small cat which may reach up to 12 kilograms, name refers to Otorongo, Peruvian
word for Jaguar. Animal generally resembles a large, short-haired domestic cat;
body may be up to 60 centimetres long, with tail about 50 centimetres. Animal
differs in shape noticeably by more muscular build, larger head and prominent
canine teeth. Canines of this cat are long both on the top and bottom jaws,
resembling those of the Clouded Leopard of Holocene (Neofelis). Such weaponry
is connected to the diet of this animal; it hunts the larger animals inhabiting
the island, including pigs, iguanas and big rodents. Colouring of this animal
is a typical “tabby” pattern, but somewhat less distinct than in a domestic
cat, background of fur is light brown and spots are a darker brown.
Animal is diurnal but may hunt in twilight or at night occasionally, these cats
are solitary, forming distinct territories which are proclaimed with loud screeching
yowls, and marked with urine. Other calls include a deep mewling sound or a
hiss when angry. This cat mainly hunts from ambush, hiding amongst vegetation
in order to chase and pounce at relatively close range. Prey is killed with
a throttling bite to the throat, or puncturing of the jugular vein.
Breeding occurs once a year, during the wet season, oestrus of the female lasts
up to 8 days. The female makes a den that is a scrape hidden in a covered area
of undergrowth or bushes, and gives birth to up to 5 kittens. Kittens stay with
mother for about 4 months; sexual maturity is reached at about 6 months. Lifespan
may be up to 15 years, but usually much shorter.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Gatito
(Nanofelis musteloides)
Order: Carnivores (Carnivora)
Family: Felids (Felidae)
Habitat: main island of Fernando de Noronha Archipelago.

Picture by Wovoka
During the Holocene, humans had caused several impacts on
the environment. Among their actions was the introduction of exotic species
and the places that most suffered with it were the islands. With the disappearance
of the human species, these animals remained and some evolved in new species
during the Neocene. One of these cases was the gatito, a descendant of the domestic
cats that were left in Fernando de Noronha after the disappearance of the humans.
The word gatito means kitten in Spanish and can also be used informally in Portuguese
to name the same thing. This species is the only representative of the Genus
Nanofelis and is endemic to the largest island of the Archipelago.
The gatito is one of the smallest felines of the Neocene, with the males having
a body length of 30 cm, while females have 26 cm. The tail is 8 cm short in
males, while in females it's 6 cm. They have a slim and flexible body with short
limbs, which gives them the appearance of a weasel. Their fur is reddish-brown
and entirely covered with black spots, while their tail is black with faint
brown-colored rings.
This species eats insects, lizards and small birds. However, their main prey
is the Noronha rat. The gatito developed
such a slim body exactly to hunt this rodent. Using their flexibility, these
small felines are capable of invading the dens made by the rats, killing them
while they are resting. Such dens are occupied and enlarged by the felines,
which turn them on their home. While they can be active at any hour of the day,
gatitos prefer to hunt during the night. This occurs not only because the Noronha
rat is mostly a nocturnal animal, but also because they want to avoid predation
by the long-legged hawk
and raptorial gulls.
The gatito is solitary, with males and females meeting only when mating, which
can occur at any time of the year. The female have a 37 day gestation period.
During this period she prepares her den to give birth by feeling it with grass
and leaves. She raises its kittens without help from the male. The newborn are
born pink, naked, blind and deaf, but gain a brownish coat of downy fur at the
age of 4 days. At 10 days, the fur starts to become reddish-brown and the spots
start to appear. The milk teeth erupt at 2 to 3 weeks of age, at which point
the young start to eat solid food, though lactation can last 12 weeks. The eyes
and ears open at 3 to 4 weeks of age, and by 8 weeks, killing behaviour is developed.
The family breaks up after 9 to 12 weeks. They reach sexual maturity with 4
months and can live for 8 years.
This mammal species was discovered by João Vitor Coutinho, Brazil.
Tiger
raptor cat (Felinoraptor tigrinus)
Order: Carnivores (Carnivora)
Family: Felids (Felidae)
Habitat: South America, subtropical and temperate areas; light forests and woods
in foothills.

Picture by Amplion
At the boundary of Holocene and Neocene human activity had
caused the mass extinction of large herbivores. It had resulted in extinction
of set of predators, including large cats of genera Panthera (the majority of
large cats), Uncia (snow leopard), Acinonyx (cheetah). In same time many smaller
cats became the extremely rare as a result of hunting or destruction of natural
environment. However, the felid family had survived in Neocene. But in most
cases it is the consequence of worldwide settling of domestic cats which descendants
had run wild and had taken part in formation of new ecosystems in various parts
of the world.
In Neocene connection of South America with other continents had interrupted
again. Some time in early Neocene the Antilles land bridge existed, and using
it land animals could settle, but then it had disappeared. In conditions of
isolation descendants of puma (Felis concolor) had turned to very large predators
of frightening appearance. These cats of South America represent the unite group
which has the general distinctive attribute – the original adaptation for killing
of large prey. The inner toe on hind leg of these cats had strongly changed:
it became little bit shorter than the others, but on it the huge strong claw
is developed. In rest it is retracted into special sac formed by skin plica.
When these cats attack prey, they seize it by forepaws and tear prey’s flesh
between ribs or on the stomach by “battle” claw of rear legs. This adaptation
resembles very much the feature of hind legs anatomy of small theropod dinosaurs
of Cretaceous, and hence the name of such predators – raptor cats. It is the
original experiment of evolution: in South America the kind of contrast to Asian
daggerclawers, massive cats with similar “battle” claw developed on forepaw
had evolved.
The constitution of these predators is rather light and “universal”, permitting
them to swarm up trees and to move on rocks good. Due to this feature raptor
cats are widely settled in South America in places where enough plenty of large
herbivores lives. They are the specialized hunters on large sluggish extraction
- large rodents barocavia and giant paca. Occasionally they attack on young
tapirotheres and groundsloth rodents. These cats attack prey from an ambush,
strike it a blow by “battle” claws, and jump off aside. If the animal had not
died from the first attack, the cat pursues prey, not repeating attack while
the victim will die from loss of blood.
In South America three well differing species of these large cats live. Largest
of them is tiger raptor cat – the lion-sized predator. This species has dark-rusty
skin with numerous vertical strips of black color on body and paws. Ears and
top part of head of this cat are black, and cheeks and throat are white. Tiger
raptor cat inhabits flat districts and foothills, preferring the mosaic landscape
where areas of grasslands, woods and rivers are combined. It is a solitary predator;
each individual occupies extensive territory up to 30 square kilometers in area.
Despite of large size, tiger raptor cat is freely able to swarm up trees and
rocks and also to swim. It easily attacks prey in water, and its prey may often
become barocavia or giant paca – semi-aquatic rodents. Occasionally these cats
catch young thyranncharaxes - huge predatory characid fishes.
This species of cats marks borders of territory by urine, leaving it on bushes,
tree trunks and termitaries. The territory of the male substantially overlaps
territories of several females living in the neighbourhood. Usually females
concern tolerantly enough to the presence of male out of breeding season, though
the female with cubs or near the prey can be aggressive to him. Males concern
to each other much more aggressively, and at the meeting necessarily display
the force to each other. This demonstration of force at well armed predators
passes very originally, practically without the direct contact: they sort out
their relationships in original competition, by turns or nearly so simultaneously
jumping on tree trunk. Having seized claws in bark, males hang on a trunk till
some seconds, hissing against each other, and then jump off. Usually the winner
is the one jumping higher and keeping on tree for longer time. If males are
equal in force, they can combat. Also the adult male concerns aggressively to
male youngsters; the majority of them perish till first months of independent
life from paws of adult males. In rare cases the old male can become cannibal
and steal cubs at females living in the neighbourhood.
Courting the female, the male leaves marks in her territory to give the female
the opportunity to get accustom gradually to his presence. “Unready” female
can rush on the male if he will appear too suddenly – usually it happens so
at young males. The female is ready to pairing till several days, and all this
time the male preserves her against other males, and frequently pairs with her.
When the rut stops, female banishes the male, showing aggression to him.
After six-monthly pregnancy, in the beginning of rain season female gives rise
to the litter of from two to eight cubs. In big litter usually only three or
four cubs have an opportunity to grow up to independence. For rearing of posterity
the female arranges the den in bush or in wide hole dug out additionally to
the necessary size. Cubs are born blind and deaf. Their wool has the same strips
as at adult individuals, but strips are rather wide; therefore cubs seem darker,
than adult individuals. At three-monthly age they leave a den and start to study
to hunt actively. Young animals spend with mother about one and a half years,
therefore the female brings posterity only once in two years. Young animals
become sexually mature at the age of three years.
The close species lives in mountains of South America: the Andean
raptor cat (Felinoraptor andinus). It is little bit lower growth, than
tiger raptor cat, but is built much stronger and heavier than it. The “battle”
claw on hind legs at this species is advanced not so extremely – in mountains
there is less number of large prey, and Andean raptor cat eats mostly smaller
animals – birds and rodents. But it also hunts young mountain tapirotheres.

Picture by Alexander Smyslov
One more species, the Chilean, or western raptor cat (Felinoraptor occidentalis), lives in narrow land strip between Andes and Pacific coast of South America. It is settled from equatorial latitudes up to Tierra del Fuego. This species of cats differs in pale yellow skin with some dark spots. Northern populations living in warm latitudes have more gracile constitution and wider ears, and southern ones, living in areas of cool climate, are smaller and short-legged, with almost one-colour rich fur. In the extreme south of South America this species of cats is superseded by larger Andean raptor cat. Chilean raptor cat almost does not live in mountains, preferring woods in foothills and in plain areas. These predators search for food even at the coastal strip, and frequently eat bodies of dead algocetids cast ashore during the storm.
Noonda
(Deinothera nunda)
Order: Carnivors (Carnivora)
Family: Felids (Felidae)
Habitat: Zinj Land, tropical forests and woodlands.

Picture by Wovoka
In human epoch many large cats had practically died out because
of disappearance of animals being their food, and because of direct extermination
by people which saw the competitors and direct threat in them. Cats of genus
Panthera included the largest representatives of family had suffered the greatest
losses. Only leopard (Panthera pardus) could go through mass extinction at the
boundary of Holocene and Neocene because it could eat small prey and was ecologically
plastic species. This species also had suffered strongly because of hunting
and destruction of habitats, and by the end of Holocene there were only isolated
populations of these animals that was an incitement to speciation after human
disappearance. One of Neocene descendants of leopard is noonda, the largest
predator of Zinj Land, East African subcontinent. The name “noonday” descends
from Swahili fairy tales where the huge man-eating leopard was called so.
Noonda is very large, tiger-sized cat. It is a predator of heavy constitution
adapted to hunting for large prey. Forepaws are little bit longer and thicker
than rear legs are, because of what noonda’s back is sloping a little. This
species has bright red fur with large dark irregular-shaped spots. Ears of animal
are black with white tips; on cheeks characteristic “whiskers” of lengthened
hair grow, as if at Holocene tigers. Tail is very long; at males it is covered
with rich wool with alternating black and white strips; at females and sexually
immature animals wool on tail is less rich, colored monophonic black. Colouring
of tail is very important at establishment of domination relations; at old males
wool on tail is thicker.
Noonda prefers to hunt large prey – great goatlopes,
bikifaru, megaardvarks
and cataphractheriums. These cats
attack prey from an ambush and actively use forepaws killing it. If prey is
of rather gracile constitution, predator simply breaks its backbone by heavy
paws; for strengthening of impact pads of forepaws are covered with thick cornificated
skin. If prey has heavy constitution, noonda delivers to it blows by 10-centimeter
long claws on internal side of forepaws. If the animal had not died after the
first attack, noonda stops attack and simply follows prey while it will be lost
from loss of blood. Because of heavy constitution noonda can not swarm up trees
as good, as its ancestor did, and hides prey in bushes more often.
This cat is solitary territorial animal; each individual needs about 20 – 30
square kilometers of territory. Noonda males mark borders of the territory with
urine, leaving it on stubs, branches and trunks of trees; females also use secretion
of anal glands. The male’s territory includes partly or completely territories
of several females. Meeting each other, males behave aggressively, but more
often the meeting finishes without fight which can be resulted in drawing of
dangerous wounds. Establishing their relations, males swing tail in sides, scratch
ground by forepaws and growl loudly. Usually one having thicker wool on tail
wins – it is a sign of presence of plenty of testosterone hormone in blood.
Male learns about readiness of the female for pairing by smell of urine, with
which she marks her territory. The heat period at female lasts till about a
week. All this time male keeps near to the female and frequently copulated with
her, and leaves her at the end of heat period. Breeding rate at this species
is rather slow. After three-monthly pregnancy the female gives birth to one
up to four cubs in reliable refuge. Cubs are born blind and only 700 grammes
weigh; spots on their wool are located denser. Since three months age cubs already
start to show independence and even try to hunt small prey. After the cub has
reached age of two years, mother banishes it. At the age of four years noondas
reach sexual maturity.
Life expectancy of noonday is up to 30 years.
This species of mammals was discovered by Ayren, the forum member.
Ant
lemur (Myrmelemur pumilis)
Order: Primates (Primates)
Family: Dwarf lemurs (Cheirogalidae)
Habitat: Madagascar, light forests and shrubby savannas.

Pictures by Alexander Smyslov
Strangely enough, there is some advantage in the phenomenon
of mass extinction for certain representatives of fauna and flora. When the
great number of live beings dies out, the survived species have a possibility
to fill become empty ecological niches and to begin active speciation. After
the extinction which had taken place at the boundary of Holocene and Neocene,
among animals of Madagascar this process started actively, and the forests of
the island were again filled with life.
In twilight and at sunrise among bushes in Madagascar light forests it is possible
to see one of representatives of new fauna of this island: the small animal
climbs among branches, from time to time tearing off pieces of bark and raking
fallen leaves in search of insects. It has an especial interests for the nests
of social insects – termites or ants. Having found such nest, this small animal
stops fussy movements and starts to eat slowly: having risen on hind legs, it
digs out the dwelling of insects and quickly licks off its inhabitants.
This animal, the direct descendant of representatives of native fauna of Madagascar,
is named ant lemur. It is a small species of primates: length of the body is
up to 15 cm, the tail – up to 20 cm.
Ant lemur is adapted for feeding on insects. Its tongue is elongated a little
and is covered with sticky saliva which is exuded from large salivary glands.
On fore- and third fingers of this primate the main tool for destruction of
nests of insects grows: these are big claws. Fingers are joined by the common
cover of skin, and look like one thick finger with double claw. Bones of these
fingers are thick – due to such fingers ant lemur ruins even firm nests of termites.
If fingers can’t cope with a nest of insects, teeth are used: forward incisors
on upper and lower jaws are very large, similar a little to incisors of rodents.
Ant lemur leads mainly arboreal way of life though could be found on the ground
often. Because of very large claws ant lemur moves on the ground on two legs,
holding forepaws hanging. Forepaws of this lemur keep the characteristic for
primates ability to grasping. The thumb is opposed to fourth and little finger,
forming strong hold. Fore- and third fingers during the climbing rise, and don’t
prevent to clasp branches. Hands are covered with dark wool. Hind legs of this
animal are not so specialised, but the thumb is opposed to the rest fingers.
 The
head of ant lemur is roundish, with the flattened face and short muzzle. Canines
are very short, and premolars have sharp cutting edge. The face is covered with
dense short fur of black color. Eyes are large; iris of eyes is brown. Ears
are short.
The
head of ant lemur is roundish, with the flattened face and short muzzle. Canines
are very short, and premolars have sharp cutting edge. The face is covered with
dense short fur of black color. Eyes are large; iris of eyes is brown. Ears
are short.
Hair of male is bright red, straw-colored at female. Tail is long; it is used
as the balance weight during the climbing. On the tip of the tail bright black-and-white
tuft grows. Threat sign at ant lemurs is vibrating tail in the air.
Once a year the female gives birth to only one young. It is covered with dark
wool, and gets coloring of adult animals only at maturity. The first two months
of life it stays on mother’s back, gradually training to climb on trees and
to search for food. It seizes wool of the mother by large fingers. When claws
start to grow strongly and cause pain to its mother, she allows the cub to travel
astride her less often. Within about five months the young keeps near mother,
then leaves her and leads solitary life.
Social insects represent the specific food resource: their nests are located
rather seldom. Because of it ant lemurs live solitarily; only the female and
young may be met together. Ant lemur marks the territory with urine, regularly
renewing the marks.
This small animal carefully protects the territory from congeners. If two ant
lemurs meet each other on the border of the individual territories, they start
to display the rights to the territory: having risen up tails, they show black-and-white
tufts and stridently squeal and grin simultaneously. Only the female ready to
pairing allows a male to the territory, but right after pairing she banishes
him.
This lemur spends day and night in shelters – usually in hollows or hollow trunks
of trees. The most active time of feeding is at sunrise when nocturnal predators
already leave to sleep, and daytime ones didn’t wake up yet. In the evening
this lemur also leaves to feeding, but that time it is more careful.
As at all small animals, life expectancy of ant lemurs is not so great: no more
than 10 years. Their natural enemies are snakes and birds of prey. Young animals
occasionally perish having not distinguished dangerous species of ants: they
sting them to death.
Translated by Alexander Smyslov.
Red-masked
lemur (Eulemur rubrifrons)
Order: Primates (Primates)
Family: True Lemurs (Lemuridae)
Habitat: forests and wooded plains of Madagascar
In Neocene the geography of Madagascar island did not undergo any significant
changes – only after the split of African continent and separating of Zinj Land
this island was displaced eastwards in common with this subcontinent. Climate
of the Earth became more humid, and the regions of Madagascar covered with woodlands
had spreaded considerably. Also the human disappearance benefited to various
inhabitants of the island, including the lemurs – the distant relatives of people.
Red-masked lemur is one of the more widespread Malagasy species. This is a rather
large primate that reaches up to one-and-a-half meters in length, and one meter
goes to the tail, which is covered with especially bright red fur. The snout
of this mammal looks like dog’s one in shape, but is shorter and wider. Ears
are small and bordered with a strip of elongated brown fur. The eyes are large;
the color of the iris is sexually dimorphic: in males the iris is yellow, in
females brown or dark orange.
The color of tail noticeably contrasts with the color of fur on animal’s body
- this primate is covered in light grey fur with darker vertical strips. The
nape, the back of the head, the rear side of neck and arms of animals of both
genders are covered in dark-brown fur. A spot of ochre-red fur, which is located
on forehead, temples and nose bridge in the adult beasts (hence the name) sharply
contrasts with it. The snout, the cheeks and the lower jaw of these animals
are white. The coloration of red-masked lemurs has an expressed sexual dimorphism
and age-related changeability: females are colored lighter than males (the background
color of fur may be almost white or slightly beige), and the adolescents have
less developed stripes. These special features of fur coloration help the animals
to follow and to track each other: red-masked lemur has well-developed chromatic
vision, and animals exchange visual signals with each other with the aid of
their vividly colored tails. But this primate also frequently uses its sense
of smell in the search for food and to mark the boundaries of its territory
with the aid of odorous urine.
The basic food of red-masked lemur includes various fruits, which these animals
eat in a great quantity, up to 80% of the mass of their day ration. Nevertheless,
they also diversify their diet with various invertebrates (mostly insects),
and also with honey, nectar and some species of tree fungi, and also with fresh
leaves.
Red-masked lemurs are social mammals living in rather large groups, which number
from 15 to 25 animals. In each group there is the dominant male and its underlings,
but the majority of animals are females and juveniles.
Due to the features of their diet, red-masked lemurs are migrating animals.
Each group inhabits an extensive territory up to several dozens of square kilometers.
The boundaries of each territory are not too strictly observed, and the border
zones of adjacent group may take up to one third of each separate group territory.
The markers left by animals rather tell the news about each group instead of
claiming exceptional rights to the territory. The group of animals constantly
roams through its territory in search of fruit-bearing trees, staying in one
or another territory no more than till several days. Groups of lemurs are frequently
followed by predatory birds and mammals attacking the old, young or sick individuals
that fell behind.
Red-masked lemurs are equally confident both on the ground and in trees: their
limbs are long and strong enough even to run at a galloping speed for short
distances. And they often escape on the ground from predatory birds and snakes,
especially in the woodlands.
The mating period of red-masked lemur takes place in the second half of spring,
when mature males chase away the adolescents and fight with each other for the
leadership in the group. Hierarchy is established by crying contests – the rivals
shout to each other until one of them gives up and becomes silent. The alpha
male obtains harem of 3-4 females, which have high rank in the hierarchy of
the group, and the vanquished males can count only to mate with one female that
stands on one of the lower steps in the group hierarchy. The alpha male does
not prevent it, but it does not allow other males to approach to his own females,
while they are in estrus. After half a year after mating females give birth
to one or two rather large offspring.
The sexual maturity of young animals comes at the age of two years. The usual
life expectancy of red-masked lemur is 10-12 years.
This species of mammals is discovered by Bhut, the forum
member.
Translated by Bhut.
Hanging
lemur (Scyolemur quadrimanus)
Order: Primates (Primates)
Family: Hanging lemurs (Pendulemuridae)
Habitat: Madagascar, rainforests.

Picture by Alexander Smyslov
In early historical epoch on Madagascar representatives of
original family of lemurs still lived – so-called “sloth lemurs” (Paleopropithecidae).
But after human colonization of island their destiny appeared sad: “freshest”
of the found bones of these animals carry the traces testifying to hunting and
cutting of carcasses of these animals. Thus, it is obvious, that they became
one of many victims of human activity promoting impoverishment of island fauna.
After human disappearance and the ending of epoch of mass extinction the few
survived species of lemurs had actively started to evolve, developing new forms
and occupying accessible ecological niches. Species of lemur family (Lemuridae)
changed especially actively. One of their descendants had mastered an ecological
niche occupied earlier by sloths in America, and by lemurs of Paleopropithecidae
family at Madagascar. It differs from ancestors in so great degree that represents
the separate family Pendulemuridae. This species is hanging lemur.
Hanging lemur is medium-sized primate: it weighs about 10 kg at body length
up to half meter. This animal has long tail which usually hangs downwards. The
role of this tail in life of hanging lemur is great: it is used for recognition
of congeners. At the tip of tail the switch of long hair of black-and-white
coloring grows, carrying out alarm function. It is possible to tell, that it
is “the mirror reflection” of its distant ancestor, ring-tailed lemur (Lemur
catta), only turned upside down. Watching such switches, appreciable from afar,
animals distinguish presence of congeners on next trees. In case of danger tail
is drawn in, and animal nestles against the branch, densely clasping it by paws.
In such position it is more difficult to attack it for enemy (usually for bird
of prey). This lemur protects itself against predators from among mammals, biting
strongly.
The wool of animal has the colouring appropriate to its usual position on tree:
back is yellowish-brown, and stomach is dark, almost black. It is obviously
expressed countershadow colouring: when sun lights up hanging lemur, light transforms
it into flat silhouette difficultly distinct among branches.
This species of primates spends almost all life, hanging back downwards on tree
branches (hence the name “Scyolemur”, meaning “lemur in shadow”). All paws of
animal are prehensile and can work as hands, grasping branches. Because of it
Hip joint is very mobile: if necessary, lemur can freely tear food from branch
and bring it to mouth by hind leg. Fingers and toes are very long (the palm
length is approximately equal to forearm lenght, and fingers make 2/3 of its
length); thumb and big toe can oppose to other fingers/toes. The surface of
palms and soles is covered with rough skin with advanced papillary combs. If
necessary this lemur can swarm up tree trunk in the same manner as other lemurs,
back upwards, clasping branches by hands and feet. This animal moves on the
ground extremely seldom and very hardly, basing on external edge of feet and
palms.
Neck of hanging lemur is almost as flexible, as at owls: the head of animal
can turn to 270 degrees. Muzzle of animal is short and wide: the basis of diet
of this animal is made of rigid leaves, including the leaves of trees containing
rubber. Therefore hanging lemur has wide tuberculous molars and strong chewing
muscles giving the characteristic shape to its head. Canines are strongly shortened
in order to improve chewing. Stomach of this lemur is complex, five-chambered,
with volumed “fermentive” department. Intestines are long, that allows taking
as much as possible nutrients from such hardly digestive food. But due to food
specialization hanging lemur can feed, not having a competition from the part
of other species of animals. On head of animal there is white “mask” covering
forehead, area around of eyes and nose bridge – it is visible from apart, and
serves for recognition by animals of each other at the distance. Ears of hanging
lemur are short, at their edges fringe of rich white wool grows.
Hanging lemurs are solitary animals. Only young lemurs, passing to independent
life, spend any time at the same branch as their mothers and follow them. But
later they start to lead independent life, and leave parents. Hanging lemurs
use calls for communication. They are especially “vociferous” in the morning
and right before rain, when they utter lingering call similar to sheep bleating.
Families are not formed at these lemurs, and male right after pairing leaves
female. Two times per one year the female brings posterity. The cub is always
sole; it is born with open eyes and the first days of life keeps on stomach
of mother, clinging for her wool. Approximately at monthly age it studies swarming
up trees, coming back to the female for feeding. At four-monthly age young lemur
becomes completely independent. If the cub was born during spring rains, female
is already pregnant again to the moment of its complete independence. In rather
dry winter period female restores forces.
Young animals begin capable to give posterity at the age of about one year.
Life expectancy of representatives of this species reaches 14 – 17 years.
Banshi
saki (Neomyrikina banshi)
Order: Primates (Primates)
Family: Cebids (Cebidae)
Habitat: Amason region, rainforests and woods in foothills.
On boundary of Holocene and Neocene big tests for durability expected the primates
order. The majority of representatives of this order in the daily life is strictly
connected with woods. Here they find shelters and food. And when on boundary
of Holocene and Neocene the global congelation began, the majority of primates
has died out. In New World in Holocene this group was submitted by three families
which number and variety were fairly undermined by human activity. Therefore
chances of survival at most part of species were minimal. But nevertheless,
among these animals some ones were to found has gone through global ecological
crisis and has successfully occupied Neocenic woods.
When night goes down on rainforests of Amason region, it is possible to see
some representatives of Neocenic monkeys of New World. In moonlight in underbrush
small groups of night monkeys banshi saki wander. These animals are rather
large for monkeys of New World: length of body of adult individual is up to
50 cm not including long tail. They are colored rather dimly in comparison
with monkeys of Holocene epoch. At these primates head is covered with white
wool, on neck, cheeks and crown long white hair forming original “pallium”
grow. Very big brown eyes with round pupils indicate that it is nocturnal animal.
In addition eyes are “penciled” from above by black wool. Only on chin males
and females have red beard (at males it is much longer, than at females). Males
in addition are decorated with rich white moustaches which are hanging down
downwards from corners of mouth. Bodies of these monkeys are covered with dense
grey wool, on chest and stomach wool is darker. The tail tip is white, but
set off by black ring.
Jaws of these monkeys are short; at males large canines slightly jutting out
from closed mouth are advanced. Nostrils as at all wide-nosed monkeys are bordered
by wide nasal sept. The forward part of muzzle at the adult banshi saki is
covered with thin wool, and at old animals it may be hairless at all. Ears
are short, rounded; only their tips jut out from wool.
Hands and feet at banshi saki are hairless, covered with grayish-pink skin.
The thumb at these monkeys does not oppose to others (this feature distinguishes
monkeys of New World from other monkeys).
Banshi saki is one of the cleverest inhabitants of Amazon woods. At them there
are rather large brain and complex behaviour including set of acquired skills,
carefully kept in collective experience. Monkeys keep contact with each other
uttering various sounds: they howl, chirp, smack and squeal silently. In total
in their “dictionary” there are some tens of sounds and their combinations.
Besides they watch white tips of tails of the neighbours, flashing in darkness.
These monkeys distinguish colors rather badly, but their night sight is very
keen.
Groups of these original primates amount up to 20 individuals, and they are
headed with dominant pairs: all males submit to the male, the female bosses
over all females of group. If the dominant male chooses to itself the female
not belonging to number of animals of the supreme rank (at these monkeys pair
is forming according personal sympathies), the group status of “Cinderella”
automatically grows.
These monkeys wander in forest, finding drop fruits, insects and small vertebrates
on the ground. But it is rather dangerous to make it: at night numerous predators
wake up and go to hunt. Usually these monkeys are very cautious, and some individuals
of medium rank are sentinels of group. Such duty allows them to lift the social
status though prevents to search for food normally. Feeling approaching of
any predator sentinels notify group about it by sharp squeal. Usually monkeys
try to save from predators on trees. If they can not make it for any reasons
they play awful performance for which have received the name.
Frightening off the enemy, banshi saki spread manes asides, having turned by
faces to the enemy. Their heads turn to terrible masks: stains above their
eyes look on the background of white mane as huge eyes, and spreaded beard
seems an aperture of huge opened mouth. Moustaches of males besides create
impression of canines in “mouth”. This performance is accompanied by loud shrill
squeal (in the English folklore banshies are creatures remarkable in heart-breaking
voices). To seem larger monkeys rise on hinder legs. Then the white mane is
even more brightly appreciable on the background of black stomach. Usually
not any predator endures monkey’s “performance” for a long time, preferring
to run away. But even if its hunting appears successful and one of monkeys
becomes its prey, banshi sakis all the same cry at the predator by all group,
chasing it forthwith – it can secure them against the following attack.
Once a year the female gives rise to two cubs. They at once seize wool on her
back, and first two weeks of life they travel on mother. Gradually young monkeys
start to master the world, to try food of adult animals and to play with neighbours.
Half year old animals already move independently, and one-year-old ones live
independent life. At the age of two years young animals can have posterity.
Life expectancy of banshi saki reaches 15 years.
South-American barbudo (Barbudo papioformis)
Order: Primates (Primates)
Family: Cebids (Cebidae)
Habitat: north of South America, forests and bush in foothills.
Primates had appeared in South America in early Oligocene, and during their
natural history were represented by exclusively arboreal forms having greater
or lesser degree of specialization to tree-climbing. Some species of South
American monkeys had evolved tenacious tail allowing to cling to branches and
to hang on it. All these monkeys form a group of New World monkeys, which prominent
feature were very widely placed nostrils.
In Holocene epoch many kinds of New World monkeys were destroyed as a result
of hunting, catching for zoos and deforestation of Amazon basin. The most specialize
arboreal and local endemic species had especially strongly suffered. But some
species managed to survive, to go through climatic changes at the boundary
of Holocene and Neocene, and to inhabit again forests of South America, which
had gradually started to restore in early Neocene. Common squirrel monkey (Saimiri
sciureus) became the most successful species of South American monkeys. Its
success had been determined by a number of factors: they easily adapted to
life in broken forests and bush, frequently went down and could easily move
on the ground, were numerous and differed in small size. The majority of monkeys
of Neocene South America descends from Saimiri. One of these species is large
South American barbudo.
Barbudo are medium-sized monkeys (body length is about 40 cm) having rather
massive constitution. They live in big groups on the ground in forests of various
types in central and northern part of South America. These monkeys meet especially
frequently in foothills and among bushes, where they find a lot of food. They
are able to climb on trees, but do it reluctantly, because they are more adapted
to life on the ground. Young individuals climb on trees more often.
Barbudo has massive trunk with voluminous stomach. Front and hind legs are
of approximately equal length, therefore the back of animal is slightly inclined
back. Tail of barbudo is very short (about 10 centimeters long); with its help
monkey can only express the mood, and it does not take any part in movement
on trees.
Head of barbudo is rounded and has short strong jaws. Males have large canines;
they frighten contenders and defend against predators with their help. Females
have lesser canines, and during the repulse of predator attacks they keep near
cubs in rear of the group while all males of the group in common attack predator,
uttering loud cries.
The body of barbudo is covered with greyish-yellow wool; back and waist are
brown. Very long wool on head is very remarkable feature of appearance of barbudo.
On nape and neck the mop of rigid black hair grows as a mane, falling on shoulders
and reaching up to the middle of back and closing neck. At males the magnificent
beard covering throat and partly breast grows (hence the name, literally meaning
“bearded”). Hair of the top part of head are short and light, and consequently
monkey looks as though grown bald though actually only the small part of skin
on head is lack of wool. When monkeys move through grass and bush, long hair
of mane protect body against pricks and scratches. Mane and beard serve for
demonstration in male duels – despite of frightening appearance, males of barbudo
do not enter fight with each other, and confine themselves only to demonstration
of menace and loud cry. In heat of the conflict male can seize by paws a trunk
of small tree or bush and start shaking them, but never rushes to another male.
The face of barbudo is covered with short white wool. Both males and females
have rich eyebrowes – it is a protection against insects and dust, important
at life on the ground. On the face of barbudo there is rather small site of
hairless skin – it is a nose and the bottom part of nose bridge, area between
nose and mouth, and area around of mouth. This skin is brightly coloured, and
the shade of colouring varies depending on physical and emotional condition
of animal. At males the naked skin on muzzle is colored crimson-red color,
at females it is pink. At young animals skin on muzzle is bright pink. At males
there is also a site of naked skin of red color on breast. During menacing
demonstration males can rear on hind legs, showing brightly painted skin of
breast to each other. Thus they raise head, in order not to close breast by
beard, and it gives to their conflict comic similarity to dispute of old men.
In general, by behaviour barbudos resemble African baboons of Holocene epoch,
but differ from them in considerably weaker aggression.
Barbudo has large eyes and good full color stereoscopic sight got from ancestors.
With the help of keen sight animals notice berries and insects making a significant
part of their diet. On hands at these monkeys thick strong nails grow, due
to which animals can rummage in ground in searches of larvae and roots. But
at them the thumb can not oppose to fingers – it is a common feature of New
World monkeys.
Monkeys spend a lot of time in bush and underbrush, therefore sound communication
occupies the significant place in their life. The voice of these monkeys is
rich in various sounds; it includes grumble and grunt, cries of various tonalities
and melodious twittering. Very much advanced hearing helps to warn beforehand
about approach of predators. Having found out a predator, monkeys give the
alarm, squalling shrilly. Their voices serve as the warning for forest inhabitants,
like in Holocene voices of deer, langurs and peacocks notified jungle on presence
of tiger.
In group of barbudo there are some leading males, which copulate with all females
of the group by turns, therefore it is rather inconveniently to define, from
what male cubs are born. Young males had not got to the circle of group “elite”
have no opportunity to couple to females: females do not admit to themselves
males of low rank. It is possible to raise the rank in group by only way: to
protect herd against predators and strangers from other groups, and to concern
loyally to females, occasionally allowing them to steal the found food. Such
behaviour reduces the amount of disputed situations in group.
Once a year the female gives rise to one cub. It strongly differs in colouring
from adult individuals: it has golden wool, and nape and lateral parts of head
are grey. The cub is looked after by the female, but she allows dominant males
to clean wool of the cub and to treat him with found forage when it grows up.
At the age of one year the cub becomes independent, and the difficult period
of its life begins – it becomes “adolescent” to whom children’s pranks are
not allowed. “Adolescents” keep by unite group, and frequently unite with coevals
from other groups, or pass to the next groups one by one, especially if those
groups are small. Three-year old male reaches blossoming of physical force,
and young female at this age already gives rise to the first cub. Life expectancy
of barbudo reaches 15 – 17 years.
This species of mammals is discovered by Momus, the forum member.
Mapinguari (Mapinguari giganteus)
Order: Primates (Primates)
Family: Sakis (Pitheciidae)
Habitat: mountain forests of South America, bush thickets.
In human epoch South American monkeys represented completely or mainly arboreal
forms. Their sizes varied from very small up to medium. The species comparable
in size to African gorilla lack among primates of New World.
At the end of human epoch the situation had changed: forests were cut down
and only small islets remained of solid forested areas lasting continuously
tens and hundreds of kilometers. In ice age the rests of humid tropical forests
had receded to banks of large rivers. As a result of these changes the part
of forest inhabitants had died out (it had affected large species in especially
great degree), and another part had been compelled to adapt to other habit
of life – life in open spaces, bush thickets and mountain forests.
Bush thickets and forests with rich underbrush are inhabited by one descendant
of former forest inhabitants – huge species of ground New World monkeys, mapinguari.
It is a huge descendant of white-faced saki (Pithecia leucocephala) - one species
of South American monkeys of Holocene epoch. Its ancestors survived in ice
age in relic forest islets and this way got an opportunity for the further
evolution. In due course some descendants of this species had increased in
size and had settled in ground habitats along the northern border of South
American pampas and in Andes foothills. This primate inhabits tropical area
of South America.
This species of primates considerably exceeds in sizes gorilla of human epoch.
Length of this animal including tail is about 3 m, height at a shoulder of
adult individual is up to 155 cm, weight of adult male is 300 kg, female –
about 200 kg. As against an ancestor having dark colouring of wool, mapinguari
has shaggy bright red fur. On back of adult individual wool is darker, on buttocks
the naked sciatic callouses of blue color bordered by strip of black wool develop.
The face of animal is hairless, skin is pigmented in great degree – usually
it is brown though there are grey-faced individuals. Face is bordered from
below by “beard” of white wool.
Tail is short – it has completely lost balance function, because only young
mapinguaris can climb on trees. Adult individuals spend the whole life only
on the ground. The top side of tail is covered with dark wool; bottom side
is white. Raising tail, this monkey shows warning signals to congeners.
Fingers have the limited flexibility: thumb is not opposable, and the animal
only picks up and tightens branches, using hands as hooks. Toes are short and
joined together in common skin cover almost by halves. Usually mapinguari moves
on four limbs, but during the feeding it rises on legs. This species eats leaves
and young shoots of trees, and also nuts and other firm fruits. Teeth of this
species are large and wide, adapted to chewing. Canines also are short, and
due to this feature lower jaw can move in sides.
Mapinguaris live solitarily or in small groups. Single individuals usually
are old animal, doing not wish to leave from places, rich in food. Adult individuals
live in family groups including male and one or two females with cubs. Animals
from different groups are not aggressive relatively to each other and frequently
feed in common. Borders of territory of each family group are very indistinct.
Usually mapinguari is silent; animals communicate with each other with the
help of rumbling and hooting. Threatening the opponent, this monkey utters
loud throat growl, and call of alarm is loud long squeal. Using such cry mapinguaris
warn of their detection of large predators – raptor
cats or jagueira.
In breeding of this species the certain cyclicity is expressed. Courtship period
takes place in the beginning of dry season. At this time males arrange ritual
combats for females – they growl, push each other and strike impacts by edge
of palm on each other’s shoulders. Such duels do not result in fatal outcome,
but allow establishing of domination relations in struggle for the female.
If the structure of family group is rather constant, male shows to females
the favour, allowing them stealing his food, and shows force, snatching with
noise and cries to other animals. Thus he only tries to frighten them, but
does not attack really.
Pregnancy lasts till about 9 months, one cub is usually born. Twins represent
a rarity – one case for some tens of deliveries. Within five months cub eats
milk, but already at the second month of life it starts to try food of adult
individuals. Till four years young individuals live with mother, but then they
are expelled by her from group. Soon after that the female is ready to pairing
again. Sexual maturity comes at the age of 6 years. Life expectancy does not
exceed 50 years.
This species of mammals is discovered by Nick, the forum member.
Kong
(Titanosimia kong)
Order: Primates (Primates)
Family: Para-apes (Parapongidae)
Habitat: Jakarta Coast, South-East Asia, tropical forests.

Picture by Eugeny Hontor
In late Cenozoic apes were the largest primates. Growth of
largest of them, Pleistocene Gigantopithecus, was estimated at 3 meters. At
Madagascar, however, lemurs of genus Megaladapis approximately equal to gorilla
in size lived. In human epoch large species of primates had suffered serious
damage from economic activity of people and direct chasing. Giant lemurs, undoubtedly,
had died out in early historical epoch because of hunting for them. Gorillas
and other apes became rarity because of hunting and destruction of habitats.
Their populations kept to the end of human epoch in some reserves, appeared
too small in order to restore these species. No one species of apes had lived
up to Neocene. But in Southern Asia evolution prepared the replacement to extinct
large primates. The descendant of one macaque (Macaca) species had passed to
ground habit of life and began the settling in all suitable habitats in tropical
zone of Southern and South-Eastern Asia. In tropical forests of South-Eastern
Asia and at the Jacarta Coast peninsula kong, the largest of ever living primates,
had evolved. Weight of adult male of this species exceeds 700 kg, and his growth
reaches 4 meters. The female of this species is smaller – her weight is about
450 kg at growth up to 3 meters.
Kong is an ecological analogue of Pleistocene ground sloths of South America
and chalicotheres of Old World. It is too massive to climb on trees, and is
exclusively ground species. Only young individuals are able to climb on trees.
By constitution kong is similar to gorilla – this primate has very long arms,
and back is inclined. Tail is not present at this species.
Living in twilight of underbrush, these primates are compelled to use very appreciable
color spots to distinguish relatives. Sciatic callouses of kong are colored
bright red and bordered by contrast ring of white wool. Sexual dimorphism is
expressed not only in size, but also in colouring – male has black wool with
brownish shade, and female is reddish-brown. Additionally male has brightly
colored face – white with bluish spots on cheeks. Face skin of females is grey.
The male’s neck is decorated with magnificent white mane contrasting with the
general colouring of body. At females wool on neck is only slightly lengthened
and darker, than wool on the body.
Jaws of kong are short and wide. Canines at females are very short and consequently
don’t prevent to lateral movements of jaws at chewing. At males canines are
larger and more appreciable – they are used in courtship displays and during
an establishment of domination relations. The diet of kong is diverse and includes
mainly vegetable food. These primates easily pull out plants from the ground
and chew their tubers and rhizomes by strong molars. The significant part of
their diet is made of foliage of young trees. In order to reach it, these para-apes
rise on rear legs and bend trunk and branches of trees to themselves. Kong likes
to feed in shallow water of tropical rivers with soft aquatic vegetation.
The food of animal origin in diet of kong is presented mainly by carrion and
the rests of prey of large predators. Kong differs in massive constitution and
sluggishness, and therefore it is very bad hunter. Its prey includes very large
invertebrates and sluggish reptiles – tortoise or snake insufficiently quick
after nourishing dinner may easily get for dinner to this primate. This species
can easily drive away predators from their prey, using huge force and superiority
in strength.
Kong lives the small groups numbering no more than ten individuals. In group
there is a well defined leader – it is large adult male. Also there are subdominants
in group – one of senior sons of group leader, or male came from the side and
showing loyalty to the leader and supporting him. As a rule, dominant male descends
from other family, rather than females of group.
Kong breeds very slowly. Females turn able to give birth to cub at the age of
about five or six years. Pregnancy lasts till about one year, and till next
three years the cub depends on mother in many respects. The wool of newborn
kong is bright rusty and darkens in course of time. It develops slowly enough,
and can independently move on four paws only at the age of five months. The
cub is fed with milk till about one year, and only since the tenth month of
life starts to use vegetative forage for food. During the life the female of
kong can bring up no more than five or six cubs. At this species of primates
twins are frequently born – one case to approximately 10 – 12 births. Obviously,
this is an adaptation permitting to increase rate of reproduction at these animals.
These primates have well advanced parental instinct, and frequently the female,
too old to give birth to its own cubs, helps young females to look after cubs,
and even transports the cub to the back. Adult kong does not have enemies, and
this primate has an opportunity to live up to 50 – 60 years.
Tibetan
yeti (Yeti tibetianus)
Order: Primates (Primates)
Family: Para-apes (Parapongidae)
Habitat: Himalayas and Tibetan Plateau, mountain forests.

Picture by Alexey Tatarinov
The most progressive primates, monkeys, in Neocene express
the variety comparable to their variety in human epoch. Large apes had not survived
up to Neocene, but their place in this epoch is occupied by large species descending
from macaques (Macaca) of Holocene epoch, which represent the special parapongid
family. Area of this family is limited to mainly Southern and South-Eastern
Asia. Parapongids include kong (Titanosimia kong)
– the largest Neocene species of primates, the inhabitant of tropical forests.
Area of primates is limited mainly to areas with sufficient duration of day.
In human epoch a number of monkey species lived in mountains where these animals
endured normally temperatures below zero. In Neocene one species of primates,
Tibetan yeti, also settled in mountain areas of Central Asia. This monkey is
named so because of significant similarity to mysterious “yeti” in appearance
and behaviour.
Because it is cold enough in mountains of Central Asia, Tibetan yeti is large
creature: weight of adult male makes about 70 kg, female is smaller – up to
50 kg. Body of this primate is covered with rich wool due to what it is not
afraid of severe mountain winds. For additional insulation at Tibetan yeti all
projecting parts of body – ears, face and limbs – are rather short. In comparison
with its close relatives from tropical forests Tibetan yeti looks rather thickset
creature. Primates from warm habitats often have sites of hairless skin on face,
breast and in genital area. This skin is frequently bright colored and serves
for demonstration during the communication and establishment of domination relations.
In cold mountain climate at intensive solar radiation Tibetan yeti has a minimum
of naked skin on face and body – the whole muzzle of this monkey is covered
with short, but rich wool; wool grows even on ears from external side. The back
side of feet and hands, and also fingers and toes from above are also covered
with wool. Perhaps, only lips and eyelids are not protected by wool.
For protection against overcooling at this primate the extensive sciatic callouses
formed of thick layer of cornificated skin are advanced. Due to such adaptation
animal can sit on cold stones without harm for an organism. Soles are also covered
with cornificated skin.
In mountains at high altitudes trees which could sustain weight of this monkey
do not grow. Tibetan yeti has substantially lost skills of tree-climbing, and
the structure of its feet had undergone significant changes. On foot of this
monkey the big toe is short, and other toes are connected in the basis by soft
tissues and had considerably lost mobility. Actually, only distal halves of
toes, which are short in themselves, remain free. Tail of Tibetan yeti has not
disappeared completely, but is considerably reduced. It keeps mobility, but
is very short and covered with rich wool. Such tail can not be the balance weight
at movement any more, but is quite functional as means of expression of emotions.
Male and female at this primate appreciably differ in colouring. The male’s
wool has chestnut color with reddish shade, darker on back. At females wool
is almost black. Male also has wide white “eyebrows”, white moustaches and short
beard. Females have only small sites is white wool above eyes.
In mountains the nature does not give a rich choice of food, on contrary to
tropical forest. Tibetan yeti is practically omnivorous: in summer its diet
includes leaves and berries, and also insects and other invertebrates. In winter
the nature of mountains is scanter, and from vegetative food in yeti’s diet
buds and needles remain. But at any time of year small vertebrates and carrion
make the desired additive to diet. Tibetan yeti frequently keeps eyes on birds
– scavengers and predators like tanatos
– and eats the rests of their prey.
In order to find enough amount of food it is necessary to be able to clamber
on stones. In summer yeti moves on four limbs more often, like other monkeys.
Winter snow and cold force it to move on hind legs, preserving hands from cold.
During a bad weather yetis crowd together, closing cubs by bodies (such behaviour
resembles the formation of “turtle” at penguins in human epoch), but search
shelter in caves or among stones more often.
This primate survives in severe mountain conditions due to social way of life
in many respects. It lives in small groups (up to 10 – 15 individuals), in which
the strict patriarchy-based hierarchy reigns. In extreme conditions for preservation
of group the survival rate of young growth is important. In connection to it
at young animals well expressed juvenile colouring – reddish fur with dark face
– develops and is kept till first two years of life (even after they become
independent). Such colouring gives the cub the right of best piece of food and
superfluous care from the side of adults. As it grows, colouring of young individual
changes to adult one, and at other individuals in group other favourites appear.
Young “adolescents” keep in group separately, and sometimes leave parental group
together, keeping friendship for long years. Such individuals challenge authority
of male from other group more successfully or simply entice females from his
group to their own one.
Tibetan yetis grow mature for very long time – not less than seven years pass
before a sexual maturity. First one and a half years young animal stays with
mother, being trained at her to receptions of food search and skills of social
behaviour. Life expectancy at this monkey is not less than 40 years. For this
time the female has time to bring into the world and rear no more than ten cubs.
Almas (Yeti almas)
Order: Primates (Primates)
Family: Para-apes (Parapongidae)
Habitat: western Himalayas slopes, mountains of Central Asia, mountain forests
and bush thickets.
Almas is a close relative of Tibetan yeti, the monkey living in high-mountainous
areas of Tibet and Himalayas. It belongs to parapongid family including mainly
large Old World monkeys descending from macaques, but convergent similar to
pongids and hominids extinct to Neocene. Almas differs considerably in ecology
from yeti: it is more thermophilic and in mountains does not rise up to snow
border and lives not above zones of deciduous forests and bushes. Also it is
much smaller than yeti: adult almases weigh no more 50 kg (males) and 30 kg
(females). As well as yeti, almas is covered with rich wool, but it has rusty
brown color, lighter, than at its eastern relative; there are also grey color
forms (up to 40 % of a population). The sexual dimorphism in colouring is expressed
in presence of white spot on breast at males and darker general colouring of
wool. At females head is lighter, rather than at males, and newborn cubs are
covered with silver-gray wool. At grey color morph cubs are almost white. Juvenile
colouring helps to moderate aggression of adult individuals during their games
with cubs.
This monkey species moves mainly on four paws, but has ability to keep in biped
pose for a long time, getting food with the help of hands. Feet are covered
with thick corneous callouses from the bottom side due to which animal can
move on stones. Big toe has kept mobility and helps animal to grip stones and
tree branches. Other four toes have grown together in the basis and are less
mobile, than at forest monkeys from tropics. Tail is short, but mobile.
Almas lives in family groups numbering 8 – 12 individuals: dominant male, 1
– 2 subdominant ones, sexually mature females and their cubs. These primates
eat various kinds of food, but, as against yeti, almases differ in omnivorousity
and willingly eat food of animal origin. They are capable to kill and to eat
even rather large animal, do not disdain carrion, drive away predators from
prey, and occasionally there are cases of cannibalism. Almases have kept more
skills of tree-climbing, than yeti did, and can escape on tree in case of predator
attack. The young growth swarms up trees especially good. Almases protected
themselves against small enemies on manners of macaques and baboons: males
show aggression and intention to fight, opening mouth and showing large canines
while females and young growth escape far away.
The sexual maturity of almas comes at the age of 5 – 6 years. Once a year the
female gives birth to 1 – 2 cubs. Newborn ones are helpless and only can grip
for wool of mother. Therefore childless females of group frequently incur a
significant part of care of cubs of other females. Life expectancy of almas
is about 30 – 35 years.
This species of mammals is discovered by Bhut, the forum member.
Enkidou
(Enkidou sylvaticus)
Order: Primates (Primates)
Family: Guenons (Cercopithecidae)
Habitat: Southern Asia, plain and foothill woods.

Picture by Alexey Tatarinov
Disappearance or significant reduction of areas of tropical
rainforests during the ice age had caused mass extinction of species connected
to these biotopes. Among irrevocably missed animals there were large primates
– apes. Gibbons, chimpansees, gorilla and orangutan have died out, not having
left descendants. But small species of monkeys were in incomparably best position.
Bieng ecologically plastic they have developed life in light forests of glacial
epoch, and when woods again have covered tropical zone of Earth, wood inhabitants
have appeared among them. Among new wood monkeys the special family of “false
apes” including very large species descended from macacas and convergently similar
to gorillas of Holocene epoch is especially remarkable. In some areas other
large primates, descendants of langoors (Presbytis) widely settled in Asia,
compete with them.
The enkidou named after beast-like hero of Sumer epos is huge primate similar
to gorilla having short tail. It is the primate of massive constitution moving
by the ground mainly on four paws, but sometimes rising on legs. In such position
this monkey can make some steps, holding something in hands. Growth of the rising
monkey reaches 3 meters. The body of animal is covered with shaggy black wool;
face skin is naked, colored red. Brightly colored face is a mark of recognition
by representatives of this species of neighbours. However the area of buttocks
serves as not less important recognition symbol at these species, as well as
at many species of monkeys. On buttocks of enkidou there are large sciatic callouses
covered with naked lilac-blue skin at males and bluish-grey skin at females.
These formations have also alarm function, by their appearance it is possible
to determine approximately the status of individual in group, its physiological
condition and readiness for pairing at female. Thus, sciatic callouses of this
animal serve as its original “passport”.
Wool of cubs is colored differently, than at adults: they are light grey, almost
white, with pink face. Sciatic callouses at them are covered with wool. Such
distinctions in colouring have the important value: they serve as the deterrent
for aggressive adult individuals, showing the “special status” of cub in group.
For this reason all pranks and theft of meal at adult monkeys are forgiven to
cubs, and the adult member of clan may have strong punishment for it.
At males of enkidou in mouth long canines used for demonstration at an establishment
of hierarchy jut out. During fights between adults they are not applied, but
at defense against predator or at rescue of the cub got to its claws adult males
inflict terrible wounds by canines. At females canines are much shorter. Molars
of enkidou are wide and knobby – the significant part of diet of this species
include with coarse sappy vegetative food.
Because of massive constitution enkidous spend the most part of life on the
ground though they can swarm up trees. Especially frequently cubs do it, finding
in branches fruits inaccessible to massive adult individuals. This species of
monkeys is omnivorous, equally willingly eating both vegetative food and meat
(if it will be possible to find it). Enkidous willingly eat tubers and sweetish
roots of ferns, soft leaves of bushes, fruits of different trees. The food of
animal origin includes insects (especially they like juicy and fat larvae of
beetles), bird eggs and nestlings, frogs and small mammals. Using the might
and large number enkidous can take away catch of different predatory animals,
or pick up rests of meals after them. During the meat sharing the severe hierarchy
is observed: the dominant male with huge canines tries this dish first, choosing
to itself best pieces. He personally endows with meat of females with which
he is coupled, after them males with whom supports friendly relations more often,
and further all cubs irrespective of whose are they. After that he leaves a
carcass for individuals of the lowest rank and does not care of its sharing
any more. Frequently lowest individuals in hierarchy get only uneatable pieces
of skin and cartilages, and sometimes they get only smell of meat. If someone
from animals of the lowest rank tries to steal a piece of meat out of turn,
the true fight accompanied with loud roar and menacing grimaces can be fastened.
At this time any monkey does not try to steal meat from fear to get “in the
heat of the moment” to the prepotent male. It happens that in an impulse of
anger the male inflicts to members of clan traumas and mutilations by impacts
of powerful hands.
The sharing of vegetative food never happen such noisy and strict because usually
it is enough of it for all ones. However dominant males watch closely members
of clan being always ready to take away from them something tasty, that they
have to found and had not time to hide and to have eaten. At the same time the
leader strictly watches that anybody, especially youngsters, did not take away
meal from cubs. Caring about cubs, the dominant male strengthens his social
status in clan - a lot of females will want to be coupled to him and will support
him if there will be any threat of leadership change in clan.
But the dominant male should fulfil such privileges to the full: at an attack
of predators or at conflict with the next clan he acts in the forefront among
defenders, and at times outcome of the conflict depends on his behaviour.
Time of pairing at enkidou is stretched for half-year falling to rainseason.
At the female ready to pairing sciatic callouses increase and become bright
blue. Usually each male occupying high enough position in clan has one female
to which he couples regularly and supports friendly relations during all life.
The female from such steady pair even can abandon clan together with the male
when the readiness for pairing comes, spending some days at some distance from
the basic group of clan.
Pregnancy at these species of monkeys lasts about half-year. The newborn cub
is covered with wool, at it eyes are opened, and it practically at once can
move head and grip for wool of mother. First time mother constantly carries
the cub at breast. Later it gets over to her back. At bi-monthly age cub climbs
down from mother and starts to play with coevals, at three-monthly age it starts
to try food of adult animals. Half year old cub ceases to suck milk and starts
to spend more time on the ground, moving together with adults. At one-year-old
age the wool of the cub starts to darken: it starts to mature and turns to the
“teenager”. At this time it usually receives the first punishment from adult
animals that marks the ending of the carefree childhood. But connection between
young animal and its mother is kept for long years, even when the female has
other cub. At the age of three years the young female is ready to give rise
to first cub.
Life expectancy at enkidou is 40 - 50 years. For this time up to 6 - 7 cubs
can be born at the female.
Kappa (Potamopithecia kappa)
Order: Primates (Primates)
Family: Guenons (Cercopithecidae)
Habitat: rivers, lakes and swamps of Japan Islands, mainly in eastern part
of archipelago.
Primates almost never passed to completely aquatic way of life. Anyway, among
them there were no animals as closely connected to aquatic environment, as
otters and pinnipeds among carnivores or hippopotamuses and sirenians among
ungulates. Separate species of primates of Holocene epoch were connected to
aquatic environment only in part: these ones were Allen’s swamp monkeys (Allenopithecus)
from Africa and long-nosed monkeys (Nasalis) from Southeast Asia. Among monkeys
of New World there were at all no species adapted to existence in water or
near water. One more primate right in human epoch had shown obvious propensity
to life in water. This event took place in Japan Islands, where some populations
of Japanese macaques (Macaca fuscata) passed to life in water in part. Island
isolation promoted to evolution of their descendant into very special creature
– to the species of Neocene primates most adapted to life in water.
In Japanese legends the fantastic water creature, kappa, is mentioned. According
to legendary descriptions, kappa is similar partly to human being and frog
simultaneously, and differs in rather blood-thirsting inclination. In Neocene
in the native land of legendary kappa the monkey, became literally a material
embodiment of this creature, certainly, with the exception of some magic properties
and predatory bents, had evolved. Therefore aquatic primate from Japan Islands
is also named kappa.
Habitat of Japanese kappa includes banks of water bodies, rivers and lakes,
thickets of coastal vegetation. Kappa avoids life in rapid and cool mountain
rivers, preferring warm and rather slowly current water bodies, which banks
are overgrown with forest and bush. Especially numerous populations of kappa
inhabit eastern side of Japan Islands directed to ocean. Obviously, there it
was a centre of kappa speciation.
Appearance of kappa is very remarkable – animal combines attributes of tree-climbing
and aquatic animal. When kappa swims, rear limbs are principal organs of movement.
Feet of this primate are adapted to swimming: they are rather long and wide,
and between toes rudimentary palamas grow. Big toe is considerably reduced.
With the help of such feet kappa swims in water quickly, resembling a huge
frog at this moment, and also walks on fenny ground. Unique feature of this
monkey is ability to walk on rear legs for more or less long time. It amplifies
the similarity of Japanese kappa to its legendary prototype even more. Kappa
differs from human beings in hunched bearing – backbone of this primate is
slightly curved, and does not form S-shaped shock-absorbing bends. Moreover,
feet of kappa are not vaulted as human feet are, and do not spring at walking.
But they are adapted very much for walking on soft ground.
On temples, head sides and nape of kappa long dark hair grow. This is very
useful adaptation – the cub keeps for them, sitting on back of the female.
But on top of monkey’s head hair are short. On body wool is also short and
thin, yellowish-brown in color. Skin on kappa’s face is naked and rather brightly
colored: at males face is dark red, and pink at females. Male differs from
female also in wool colouring: at his breast dark wool grows, whereas at female
wool on breast is bright yellow. Moreover male has much longer and richier
“whiskers” growing down to the middle of breast. Male is 20 – 25 % heavier
than female and differs in larger jaws with long canines.
Kappa lacks of tail – it had disappeared at far ancestors of this species.
But at animals of both genders bright sciatic callouses are advanced. At males
they are light pink, not too visible on the background of wool. At females
sciatic callouses are bright red, shaded in addition with black wool on buttocks.
This deliberately bright colouring is connected to social behaviour of primates
in which the condition and demonstration of sciatic callouses and area surrounding
them has great importance. This is a powerful gesture of suppression of aggression,
which is important for this species – kappa males are rather aggressive, and
among them the strict hierarchy is supported.
Arms of kappa are about the same length, as legs are. Kappa’s hands have long
sensitive fingers – they are equally well adapted to grasping of tree branches
when animal climbs and to searching for small invertebrates in muddy water
– kappa frequently searches for food under water to the touch. When kappa swims,
it presses hands to breast.
Kappa has also other adaptations to aquatic habit of life: when animal dives,
its nostrils close. At diving kappa can hold the breath for one minute and
more. Under water kappa searches for food – roots and tubers of aquatic plants,
snails, crustaceans and frogs. Gathering trees fruits drop in water, kappa
keeps in water in upright pose, standing on rear legs. In this case wide feet
prevent sticking in oozy bottom. Diving skill belongs to the number of acquired
attributes; therefore kappa with very young cub appears very vulnerable. The
only way of rescue for such female is its skill of tree climbing – kappa had
not lost this ability though does it worse than other monkeys because of feet
specialization. But the female has got in a trouble, can expect to the aid
of relatives: having gathered in group, kappas can counterattack a predator.
Males may be especially dangerous to aggressor – attacking it, they put deep
wounds by canines.
Kappa female gives birth to one cub once a year; the expressed seasonal prevalence
in breeding of these monkeys is absent. Cub differs in colouring from adult
individuals: it has light grey wool and pink skin on face. As it grows, the
wool of young individual changes colouring: first it becomes plain yellowish,
and then starts turning darken.
Kappas spend a lot of time in games with each other and both cubs, and adult
individuals equally like to play. Animals conceive games not only on trees,
but also in water. Jumps in water represent the especially favourite game at
young kappas. Monkeys jump in water from height, having swinged on flexible
tree branches. Each group of kappas has favourite “game tree” on which animals
swing with pleasure when the group of animals comes back to this place.
Approximately at the age of eight months at young kappa long hair on head start
to grow. Individual development is rather long – the young animal keeps connection
with parents even after birth of the next cub. Young kappa becomes completely
independent approximately at two-year-old age. At this time young females study
to care of posterity, helping mother to look after their own younger brothers
and sisters. Females become capable to breeding at the age of three years,
males – for half-year later.
Like all primates, kappas are social animals. They live in groups including
several breeding couples and their posterity. As a rule, adult animals are
joined by consanguinty, but the significant part of group can be submitted
by individuals from other populations. Young kappas live for a long time in
parental group – females more often remain in it till whole their life, but
males in most cases pass to other groups. If male stays in parental group,
his social status will be low while his father is alive.
Kappa lives in groups, differs in ability to bipedal moving, and its hands
allow manipulating with small subjects. But it does not mean an opportunity
of intelligence development for them at all. These monkeys do not make tools,
and their brain is rather small. In addition to it this species is closely
connected to specific habitats – humid marshy forests. Populations of kappa
contact among themselves rather poorly, and in each population differences
in appearance of animals, and even in size, from other populations quickly
accumulate.
Kappas evolved at the islands, in area isolated from continent. Ways to distribution
for them are cut off by wide sea passages. Kappa is able to swim, but salt
sea water makes passages completely insuperable obstacle for this primate.
Therefore kappa appears only very specialized kind of monkeys; it does not
have prospects for development of intelligence, and has no one opportunity
to settle beyond Japan Islands.
Healer
monkey (Micropresbytis medicus)
Order: Primates (Primates)
Family: Guenons (Cercopithecidae)
Habitat: Southern Asia, tropical forests and bushes.

Picture by Lambert
In Neocene the primates order is presented by a plenty of
species of monkeys and prosimians. At the boundary of Holocene and Neocene hunting,
destruction of habitats and climate changes had resulted in reduction of number
and specific variety of primates. Warmer climate of Neocene has resulted in
expansion of territories suitable for life of primates, and their variety began
to grow again. Among them very original and specialized species had evolved.
Mountain ridges separated Eurasia into two sharply differing worlds: cold north
with obviously expressed change of seasons, and humid tropical south. To the
south of mountain zone, in tropics of India and Indochina, fauna is very rich.
Forests and bushes are populated with various large ungulated on which the set
of species of parasitic invertebrates lives. Usually the role of their cleaners
is carried out by birds, but in tropics of Asia small species of monkeys evolved,
which also specializes in gathering of parasites from wool of these animals.
Such behaviour is derivate of grooming, usual behavioural feature of monkeys.
Birds clear herbivores of parasites on open areas, and monkeys do the same in
forests. Due to such division they do not compete to each other.
Healer monkey living in tropical forests and woodlands is a small species of
primates. It is larger than squirrel
guenons living in Africa: weight of adult male is about 700 grammes, female
is up to 500 grammes. At this species of monkeys there is very bright and appreciable
colouring. The basic background is bright red, stomach and forepaws are white.
Skin on face, palms and feet is dark and hairless. In addition to size males
differ from females only in presence of small “beard” and longer canines. At
females canines are only a little longer than other teeth.
These primates live in small family groups numbering from one up to three males,
up to five females and their cubs. In group the strict hierarchy is supported.
Animals communicate with each other by means of various sounds; in “language”
of healer monkey there is about 20 various signals.
Usually monkeys keep on trees and among bushes, but spend a lot of time on the
ground. Usually they move in forest, keeping nearby from large herbivorous mammals.
It is connected to features of behaviour of this primate species. Healer monkey
is omnivorous and willingly eats fruits of various trees. When monkeys are fed,
they drop a part of fruits on the ground, and it additionally involves ground
animals. But the significant part of a diet of healer monkey is made with various
insects and other invertebrates gathering on bodies of herbivorous mammals.
Monkeys get on backs of animals and clear their wool of parasites, in exchange
receiving the additional protection against predators. Due to contrast colouring
they are easily found out by herbivorous mammals which behave quietly at the
presence of monkeys. Animals get one more advantage from the neighbourhood with
monkeys: these primates have good sight and beforehand warn animals cleared
by them of approach of predator by loud cries.
At the watering place these monkeys prefer to keep near large mammals which
presence frightens off snakes frequently attacking animals of this species.
Besides snakes, the main enemies of these primates are large birds of prey and
small mammalian predators.
These primates breed quickly enough - approximately in half of cases at the
female twins are born. Cubs are looked after by all females of group due to
what the survival rate of posterity is very high. The young monkey becomes capable
to breeding at the age of three years.
The idea about existence of this species of mammals was proposed by Momus, the forum member.
Jumping
squirrel guenon (Sciuropithecus semivolans)
Order: Primates (Primates)
Family: Guenons (Cercopithecidae)
Habitat: tropical woods of the Central Africa, highest trees.
At the end of Holocene tropical rainforests of equatorial zone of Earth
have had not best time: aridization and the cold of planet climate during the
ice age has sharply reduced their areas, having caused disintegration of earler
uniform zone of woods to the set of small islands. Representatives of fauna
of these natural communities have died out or their number was sharply reduced.
But some species used original strategy of survival which had helped to stay
number of species at former level: they have simply decreased the size and
at the reduced territory of woods the big population which survival is more
stable could live. Elephants and rhinoceroses from islands of Mediterranean
Sea, mammoths of Wrangel Island and Columbian elephants of islands near coast
North America had used this strategy in Pleistocene. And in forests of Africa
guenons (Cercopithecus) have acted so. One of species of these motley monkeys
had turned to real dwarf in its genus that has permitted to it to survive.
And this tactics had appeared so successful, that later this species had evolved
into some affiliated species occupying different ecological niches. So the
genus of squirrel guenons (Sciuropithecus) which representatives practically
do not exceed the common squirrel by size had appeared on Earth. After
increase of the forest area species of this genus were settled widely, but
the inhabiting at different “islands” of forest had resulted in formation of
different species distinguished from each other by behaviour and habit of life.
Squirrel guenons are very “large-headed” in comparison with other species of
monkeys – it is common feature of all small primates including South-American
marmosets (Callitrix) to which squirrel guenons are similar by ecology. Besides
these tiny primates differ from each other by numerous “ornaments” of hair,
color strips and spots serving for specific recognition.
Jumping squirrel guenon is the typical representative of genus. It lives on
highest trees of tropical wood and has the original adaptation for such life.
This monkey is the excellent hopper; at it has big hinder legs with tenacious
long toes. It easily surmounts distance of ten meters and more by jump. For
small creature it is a simple task: the monkey keeps in air due to long hair
growing on sides and shoulders forming original “parachute”. The magnificent
hairy brush on the tip of long tail helps to drive in flight. By it the jumping
squirrel guenon is convergently similar to monkeys of guereza genus (Colobus).
Pushing off from branch by legs, monkey stretches hands in sides and freely
soars in air, supported by ascending air streams. Having reached up to the
planned branch, it bends body, extends legs forward, deflects head back, and
seizes bark by toes. Due to abilities to “fly” it practically has no competitors
among neighbours, having mastered life on highest trees of tropical forest.
The jumping squirrel guenon is colored contrastly: almost all body is covered
with white wool, hairs of “parachute” are white too, and on back there is black
longitudinal strip expanded on shoulders. At males faces are covered with bluish
naked skin and have red “sidewhiskers” and beard, at females skin on the face
is pink and hair “ornaments” are not present. Besides at males of this species
long canines are advanced. Cubs sharply differ in colouring from adults: they
are covered with brown wool brightening later.
These monkeys keep in crones of the highest trees (at height over 30 meters)
in flights of 20 – 30 individuals. In group the main pair is precisely appreciable,
and other pairs implicitly submit to them. In rear guard of the group there
are young not sexual matured individuals and bachelors. They are afraid of
leader, and at the same time regularly challenge his right on leadership.
At these animals the “language” of sound signals is advanced including up to
30 various “words” like squeal, hoot, twitterling and cries.
Jumping squirrel guenons eat soft fruits, seeds with thin covers, both various
insects and spiders finding them in tree crones. They do not compete to wood
rodents specialized in feeding by firm forages. Besides squirrel guenons are
rather aggressive to them, and rodents, as a rule, avoid places where these
primates keep.
The female gives rise to one cub once of times 5 – 6 months. First time it
grips for wool on her breast, later gets over on mother’s back. While at the
cub “child's” colouring is kept, all individuals in flight irrespective of
rank willingly play with it. But later, approximately at four-monthly age,
colouring starts to brighten: the cub matures. It finally leaves mother at
the age of seven - eight months though from time to time it communicates with
her during all life. The grown up young growth keeps aloof of the leader seeing
threat to the leadership in bachelors. To prove the right of domination the
male resorts to menacing demonstrations: he opens a mouth wide, showing sharp
canines, and squeals shrilly. Thus he shows the force, jumping in branches
or shaking branch on which it is sitting. Usually such reminders on leadership
are bloodless and are limited only to demonstrations but if the opponent is
audacious and is not going to submit, demonstration of forces passes in fight,
being accompanied by bites and peces of pulled out wool. When the hierarchy
is indisputable, the group can food peacefully, except for moments when the
individual lowest by rank does not succeed to hide a lucky find from vigilant
look of the clan leader. Social connections in clan become stronger with the
help of grooming. Squirrel guenons as however all monkeys express this way
the loyalty or favour to the neighbour.
As well as all small animals squirrel guenons live not so long though due to
the brain size they can be considered as long-livers in comparison, for example,
with rodents: they live up to 10 – 13 years.
Representatives of closely related species living in forests of Equatorial
Africa are:
Marsh
squirrel guenon (Sciuropithecus paludiphylus). This primate belongs
to the same genus as previous one. It differs from jumping squirrel guenon
in
more
massive
constitution, shorter extremities, absence of hair fringe on sides and also
in other wool coloring. Colouring of this species is much darker: head of
black color, grey back and white stomach. Along sides longitudinal rusty-brown
strips spread, on hands there are “gloves” of red wool. Around of eyes this
monkey has rounded sites of naked white colored skin. It is the frightening
colouring which is making an impression of huge unblinking “eyes” of any
large animal. Cubs have no such false “eyes”, and they do not have sites
of red wool on the body. Tail at monkeys of this species is covered with
black wool with white hairy brush on the tip.
This species of monkeys is larger rather than jumping squirrel guenon: adult
monkey is young cat-sizeed and male is larger than female. Living in marshy
woods marsh squirrel guenon frequently searches for food near to water: it
is able to catch crabs and tadpoles in wood pools. Also these monkeys are willingly
fed with tubers and greenery of marsh plants. The significant part of diet
of this species includes tree fruits have fallen on the ground, therefore monkeys
of this species frequently watch groups of monkeys of other species, gathering
fruits thrown by them.
Decorated
squirrel guenon (Sciuropithecus ornatus). This representative of
the genus differs in very bright colouring: bright-red body, white crest
on head
at
animals
of both sexes, long tail at the male is white, at the female black. The back
side of palms and feet is colored coffee-brown. Face skin is naked and yellow.
Males have small black “barb”. Cubs are colored uniform brown color; only
face skin is yellow, as at adult monkeys.
Decorated squirrel guenon lives on the trees making forest canopy and practically
does not go down to ground. This species even searches for water in leaves
and crowns of epiphytic plants. Food specialization is available at these species:
fruits of trees of family Sapindaceae including inedible for other monkeys
contain more than half of diet of these monkeys. Fruits of some Sapindaceae
are poisonous being unripe or overripe, but features of digestion of monkeys
of this species permit to eat even them without harm for themselves. Due to
it decorated squirrel guenon avoids food competition to other species of monkeys
and also to rodents.
Blue-faced
squirrel guenon (Sciuropithecus cyanofrons). Distinctive features
of this species is naked bright-blue face skin at monkeys of both sexes and
of any
age, and
also cross-striped black-and-white tails (as at ring-tailed lemur or the
coati raccoon). The body is colored gray-brown color; wool on extremities
is lighter, at males on chin and throat there are long hair form “beard”,
and on head there is black “hat” contrasting with it. Cubs have uniform gray-brown
colouring, and they do not have strips on tails.
This species spends a lot of time on the ground, therefore at them the feature
similar with coati and ring-tailed lemur had developed: these monkeys keep
striped tails vertically, therefore any member of group can notice easily neighbours
among roots of trees. Life of these monkeys is closely connected to trees of
fig (Ficus) genus, which fruits frequently ripen on trunks practically at the
ground level. Other features of behaviour at these monkeys are common for representatives
of this genus.
Caribbean
crested guenon (Papiopithecus mitratus)
Order: Primates (Primates)
Family: Guenons (Cercopithecidae)
Habitat: Cuba, Great Antigua, sea coasts.
Picture by Alexey Tatarinov
Human activity rendered significant influence on fauna of
various areas of the Earth. One of its consequences was occurrence of representatives
of one or another group of animals and plants in places where they could not
reach in natural way. Before people Old World monkeys had been absent in New
World. Green monkeys (Chlorocebus sabaeus) had been introduced in human epoch
to islands of Caribbean Sea from Africa; and they had quite got accustomed in
new motherland and had successfully gone through miseries appeared because of
ice age. When islands of Caribbean Sea connected to each other, forming the
land bridge, descendants of these monkeys were settled at the largest islands,
having formed some species well distinguished from each other, but had not undertaken
attempts of colonization of continents.
One descendants of green monkeys lives in coastal forests of Cuba – it is Caribbean
crested guenon, rather large and almost terrestrial monkey. Body length of adult
individual of this species is up to 70 cm, weight is about 30 kg. In its shape
this monkey resembles baboon – it has large head, front and hind legs of approximately
equal length, rather small feet and hands, and the tail strongly shortened in
comparison to forest species. Wool of these monkeys has yellowish-brown color,
a little bit darker on back, but edged by strip of light, almost white wool
– such colouring helps monkeys to distinguish each other. Tail about 25 cm long
with white switch of long hair on the tip has completely lost functions of the
balance weight – Caribbean crested monkey spends on the ground almost all day
time, and only for night gets on trees for sleeping. Nevertheless, such tail
has the important alarm value – its position and movements of monkey express
its emotional condition. The important role in emotion expression is played
by demonstration of canines which are especially advanced at males.
Caribbean crested guenons have clearly expressed sexual dimorphism. Males are
larger, rather than females, have high dark cop looking like mitre on a top
of head. At females the top of head is covered with short wool, and only in
the middle of it the strip of dark wool stretches. Also at males hairless face
skin has grey-blue color, and at females it is dark grey. Cubs of these monkeys
have darker, almost black wool. Naked face skin which is colored light grey,
almost white, sharply contrasts with its color. Juvenile colouring is a guarantee
of the good reference to cubs: adults forgive monkey tricks and food theft to
them. But, as colouring changes to one characteristic for adult individuals,
the relation to cubs also changes and they are compelled to struggle for place
in group hierarchy.
These monkeys move in forest in groups: male, 3-4 females and their cubs of
several previous years. Male is the indisputable leader in group and it demonstrates
the superiority, taking off the found food at females and juveniles. Young individuals,
as a rule, leave parental group. The female almost always can join any other
group, but males keep in barchelor groups at the forest edge, and from there
they can encroach to harems of mature males in hope to take a place of the leader
there.
These monkeys inhabit seacoasts and coastal forests. In the afternoon they wander
on beaches, selecting the fishes thrown out by waves and crustaceans, and also
collect from stones seaweed. They supplement the diet with fruits of trees or
insects. At night these monkeys are arranged on branches of trees and sleep,
closely having nestled to each other.
The courtship season is not expressed, and at any time in herd there are young
individuals and females ready to breeding. Male confirms its rank with ritual
poses more often and drives away contenders, showing to them grinned canines.
Thus he rises on hinder legs and can make some steps while keeping such position
.
Pregnancy at the female lasts till about 150 days, and more often two cubs are
born. The lactation lasts up to about 8 months, but cubs start to try food of
adult animals right at the third week of life. Puberty comes in 3-5 years. Life
expectancy is till 25 years.
At Great Antigua Island subspecies of this monkey (P. m. antiguanus)
lives – it is larger one having bright red wool and grey back. Monkeys from
Great Antigua differ a little from inhabitants of Cuba in skill of gathering
food in freshwater reservoirs. They can dive for food (monkeys from Cuba do
not do it), but appear at sea coast less often.
The idea about existence of this species of mammals was proposed by Nick, the forum member.
Chemosit
(Gravipapio chemosit)
Order: Primates (Primates)
Family: Old World Monkeys (Cercopithecidae)
Habitat: South African savannas.

Picture by Alexey Tatarinov
Among the Old World monkeys, the baboon genus contained the
most heavy primates after great apes, including mandrills and drills, whose
number decreased greatly, however, in the era of Holocene human pressure. The
changes in nature at the conjunction of two eras had destroyed the chances of
survival for those species, as well as for great apes proper.
But besides mandrill and drill, among baboons there were many other relatively
large primates, more suited to survival in the face of changes in their habitats.
One of them was the Chacma baboon (Papio ursinus). An inhabitant of savanna,
not the jungle, omnivorous, inclined to carnivory, it succeeded greatly and
after the disappearance of people it evolved to chemosit, one of the largest
primates of Africa.
Chemosit is actually a baboon of very robust constitution. The length of chemosit
male may reach 3 meters, of female – 2-2.5 m; the short tail is 10-15 centimeters
long. Its body is covered with yellow-brown hair, except for muzzle and butt,
which are covered with black hairless skin; also mature males grow sideburns
and beard of brighter, whitish color.
Hands and feet of chemosit have kept the general similarity with those of other
primates, but its fingers are very rigid, almost non-bendable, with the exception
of thumbs, therefore their grabbing ability is very limited. Hind legs are somewhat
more powerful and longer than forelimbs, so chemosit can stand-up on them for
a while for a better view of its surroundings or to frighten its enemy or rival
congener. When moving, the rump of animal is slightly above the shoulders.
The main weapon of chemosit is its teeth. Both they and the jawbones are very
thick and powerful. Canines can cause deep lacerations and break a neck of even
a big animal, but chemosit hunts rarely. It prefers to eat carrion, or to steal
prey from other predators. It also eats vegetative food, like hard pods of acacias
and other plants. When searching for insects it breaks into anthills, termite
mounds, and wasp and bee nests; due to its thick skin this animal easily endures
their stings. The smaller females of this species especially love wild African
cereals and similar plants.
Among chemosit males loners are frequent; sometimes they live in groups of two
to three individuals. Successful males lead harems of 4-5 adult females, while
protecting their offspring. Each female typically gives birth to one cub once
in three years; twins are very rare.
The mating season of chemosit is not distinctive. Single males constantly impinge
on the harems of alpha males and roam on the borders of their territories, declaring
their intentions in loud roars. Periodically there are attempts of alpha males
to win over females from their neighbors; usually it is a displaying of force
and the length of canines, but on occasion real fights happen.
Young monkeys remain in their family’s clan until the age of three, and then
they are banished. Puberty in females occurs at the fifth year of life, in males
at the sixth or seventh year. The life expectancy of the chemosit can reach
35-40 years.
This species of mammals was discovered by Bhut, the forum
member.
Translated by Bhut.
Umingmak (Arctotitanolagus umingmak)
Order: Hoofed lagomorphs (Ungulagomorpha)
Family: Huge lagoids (Titanolagidae)
Habitat: Central and Northern Eurasia, Beringia and Alaska (it is the only
species of hoofed lagomorphs penetrated to New World).
Picture by Tim Morris
In Holocene fauna of Eurasia herbivorous mammal were presented
by hoofed mammals of various families – mainly by deer and bovines, and also
by less numerous pigs, elephants and perissodactyls (horses and rhinoceroses).
As a result of human activity, hunting and habitat destruction, the majority
of families of hoofed herbivores had disappeared. In Neocene bovines and deer
had kept mainly in southern part of Eurasia and Africa where after human disappearance
they had formed some separate families. To the north of ranges of Central Asia,
in Europe, Siberia and Far East, the fauna of herbivores is presented by various
descendants of pigs, and also by new group of animals – hoofed lagomorphs. This
order of mammals, descended from true lagomorphs, includes animals of two types
– gracefully built harelopes living in savannas,
woods, steppes
and mountains, and massive
herbivorous huge lagoids to which aurochid
from Northern Eurasia and huge obda
from Siberia belong. These animals inhabit woods of Eurasia, and mountains of
Eastern Siberia, Beringia, Alaska and plains of Far North are inhabited by one
more species of massive line of hoofed lagomorphs, the umingmak.
“Umingmak” is the Eskimo name of musk ox. It indicates a role of this animal
in ecosystems of northern latitudes: it is an ecological analogue of artiodactyls
of Holocene epoch. Umingmak is the herbivorous cow-sized mammal. It lives the
year round in conditions of seasonal sharply continental climate, not making
long migrations like obda. In winter in habitats of this animal there is the
thick snow layer and the temperature sinks up to -30°С, but umingmak is
adapted to survive and to exist normally even in such inclement conditions.
In severe conditions of the North it is better to struggle for survival in common.
Umingmak is gregarious animal keeping in herds of 30 – 40 ones and sometimes
even more. This animal prefers district where wood and plains are combined.
In winter umingmak avoids places where the thick layer of snow falls – in searches
of forage it should dig snow out; therefore these animals go to forests. Umingmak
frequently comes to frozen wood bogs where it is easier to find high last year's
grass.
The animal is well adapted to severe climate of Siberia. Foot of umingmak is
wide: it permits the animal to not fail in snow and to wander freely in snow-covered
woods. Umingmak runs badly – its maximal speed is about 45 kms per hour. But
the predator all the same can not catch it up on friable snow. Back legs of
the umingmak are little bit longer than front ones, and the back is inclined
forward – this animal meets in mountain areas of Central Eurasia and Beringia,
and grazes on slopes. In winter umingmaks often meet in mountains, especially
in areas where the wind blows snow off. They graze stalks of last year's grass
and young branches of bushes. On nose of this animal the cross horn comb is
advanced, assisting to dig snow. It gives to the animal some similarity to rhinoceros.
At the ground dug by umingmaks other herbivores, snow
harelopes, distant relatives of the umingmak, feed. Umingmaks frequently
search for forage in common with another large herbivore – Siberian
shurga, the huge boar descendant. But they keep aloof of this aggressive
animal, and feed with the rests of grass and bushes when shurga goes out.
Similarly to their far ancestors, hares, umingmak changes color of wool according
the season. Summer wool of this animal is short and brown, lighter on cheeks
and stomach. On throat and neck of males the “beard” of long hair develops –
it is the secondary sexual attribute characteristic for various species of huge
lagoids. Except for it, males of umingmaks differ in massiver head, rather than
females. In summer wool on back of animal dark “belt” stretches. Winter wool
of umingmak is long and white with grayish shade. At this time at the animal
rich thin underfur develops. In spring shedding umingmaks find coniferous trees
with dried up bottom branches, and scratch against them, leaving on branches
and trunk flocks of shedding winter wool. At this time small birds take wool
of umingmaks to build nests. And larger birds even sit on backs of grazed umingmaks
and pull wool out right at animals. It does not cause them any anxiety: wool
of this animal in general keeps in skin unsteadily, and easily drops out, if
it is pulled out strongly. This feature rescues young umingmaks from small predators.
Head of umingmak is massive, with thick frontal bones and short strong jaws.
The nasal cavity has grown up, nostrils are able to close – with their help
animal adjusts temperature of inhaled air. Ears of animal are short and rounded
– such feature had developed in cold climate where it is unprofitable to warm-blooded
animal to have strongly extending parts of a body increasing heat emission.
Molars of umingmak are plicated and constantly growing – it is connected to
diet of animal which includes rigid grass and branches of bushes. Forward pair
of top incisors is wide, and all incisors of animal grow constantly. The feeding
animal can bite across small branches of bushes. Umingmak is the exclusive vegetarian.
In summer it eats leaves and grass, occasionally browses branches of bushes.
In winter when it is very difficult to search for food, umingmak becomes less
legible in forage: by wide feet and muzzle it digs out snow in searches of last
year's grass and evergreen bushes. If the layer of snow is too thick, umingmak
can rear and eat branches of trees, basing forward legs on tree trunk. It can
eat even conifer needles.
The simple stomach of umingmak had actually turned to the fermenting chamber
in which symbiotic bacteria live. Umingmak does not chew cud like artiodactyls,
and digestion occurs, basically, in long bowels. For digestion of rough vegetative
forage at the umingmak the volumetric caecum also occupied by bacteria is developed.
Fermented food gets first to caecum, and then to bowels.
The rut of umingmaks takes place at the end of summer when animals are in the
best physical condition. The female ready to pairing emits the special smell
involving males. Around of such female some males gather and persistently follow
behind her. Males establish hierarchy among themselves: they fight, pushing
the contender away from the female by sides and shoulders. Occasionally, when
there are two males of about equal force, they can even bite each other. The
female emitting the specific smell can be attractive to males till some days,
but it is ready to fertilisation till some hours per year. At this time one
of strongest males from her “retinue” simply drives contenders off and protects
the female, frequently pairing with it.
Pregnancy at umingmaks lasts about 10 months, and in spring at the female one
cub (twins are extremely rare, and usually one of twins does not survive) is
born. It is well advanced, and right after birth rises on legs. Umingmaks walk
slowly, and run only in extreme cases, therefore the cub does not lag behind
herd already at the first day of life. It is fed with fat milk and quickly gathers
weight: at the autumn young umingmak weighs already about 300 kgs. The significant
part of cubs had not reached age of two years perishes of winter fodder shortage.
Having survived in first two years, young umingmak has an every prospect to
reach maturity. Males mature at the age of four years, and the female do it
at three years.
Black-headed
dwarf harelope (Lepolopella nigriceps)
Order: Hoofed lagomorphs (Ungulagomorpha)
Family: Harelopes (Lagolopidae)
Habitat: steppes of Central Asia (up to eastern coast of Fourseas) except for
bush thickets.

Picture by Cossus
Harelopes have replaced in Neocene numerous hoofed mammals
became mass species of herbivorous animals. Having developed plains, forests
and woodlands, they were evolved to set of species with various appearance distinguished
by different requirements in environment. In steppes adjoining to eastern coast
of Fourseas black-headed dwar harelope, one of tiniest representatives of this
group of animals, lives.
Growth of the dwarf harelope at a shoulder is about half meter. This is long-legged,
fragile, gracefully build animal. Legs of this harelope end with sharp hoof-like
claws providing excellent cohesion with ground at run (escaping from enemies
dwarf harelope can accelerate momentum up to 60 kms per hour at short distances
and also sharply change run direction).
Head of dwarf harelope is rather short and high, with strong chewing muscles:
rigid graminoids made the basis of food of this animal. Living in dry and hot
climate dwarf harelope has long ears penetrated with circuit of blood vessels
which promotes effective cooling in heat.
Short wool of dwarf harelope is colored yellowish-brown with narrow close cross
strips. Such colouring is consequence of special defensive strategy of dwarf
harelope: having run any distance animal makes a long jump sideways and hides
in grass having nestled to ground. Striped colouring reliably masks this harelope
from predators.
Remarkable feature of colouring of this species is head parren – black head
sides (areas around of eyes and cheeks). Nose bridge, nape, chin and throat
are colored white. Such colouring of head helps animals to identify each other
among grass and to support visual contact. Keen sight plays very big role in
life of dwarf harelope: with the help of head movements animals can transmit
signals to each other at big distances. If the grass is high, harelopes can
even rear to support visual contact to the neighbour. At cubs head is colored
the same color as body; only at approach of sexual maturity it gradually darkens.
Dwarf harelopes live in herds of 20 - 30 animals at wide grass plains with small
thickets of bushes. There is no well defined leader in herd: the hierarchy is
established by males only in short breeding season. Usually animals spend day
in shadow of bushes and graze since evening till morning with short breaks for
deep sleeping. And animals fall asleep by turns: a little from their number
stay on the alert all the same.
For winter harelops migrate to the east away from Fourseas coast: during winter
rains wool of animals can become wet that usually results in cold. And the ill
harelope is easy catch for predators.
In winter time rich underfur grows at them.
In the beginning of cool winter at harelopes the pairing season begins. At this
time males become pugnacious and intolerant to each other. Rising they beat
each other by forward legs, striking to opponent impacts by forward part of
wrist. Such impacts do not harm fighting animal but at protection of the cub
against small predator female can strike it direct impacts by hoof-like claws
which can wound the aggressor seriously. However protection against the enemy
with the help of hoof impacts is an extremely rare situation: usually dwarf
harelopes seek safety in flight.
During courtship season males carefully sniff at all females finding ones ready
to pairing. Such condition lasts at the female only some hours per year, and
here it is important to use it in time. Male protects such female within day,
repeating pairing some times. After the female will lose readiness for pairing
and will cease to accept his carings, he searches for new females.
Pregnancy lasts about three months, then in early spring female gives rise to
twin (sometimes even triplet) cubs. They are born very much advanced, with opened
eyes, and after some hours are able to follow herd. At two-day age cub is equal
in speed to adult animals.
Female feeds cubs with milk about 2 months, but already from week age the young
growth starts to try forage of adult animals and change of diet passes gradually.
At the age of eight months young animals get adult black-and-white colouring
of head. They form independent herds and migrate to the east. At this time females
become capable to pairing, and next spring they give rise to first litter. Males
take part in pairing only one year later.
Life expectancy of black-headed dwarf harelopes is rather short and seldomly
exceeds 10 years.
Australian
marsh rabbit (Aqualagus natans)
Order: Lagomorphs (Lagomorpha)
Family: Hares (Leporidae)

Picture by Carlos Pizcueta (Electreel)
Habitat: rivers and bogs of Eastern Australia.
During all Cenozoic in Australia marsupial mammals dominated. Due to absence
of competition they had occupied practically all ecological niches with one
exception. Because of features of cub bearing process marsupial mammals principally
can not develop water habitats. Only in South America the water opossum (Chironectes
minimus) had appeared but it had developed water habitat not in that degree
as, for example, otter or seal. But activity of people had resulted in occurrence
in Australia of big number of placentary mammal species. In Neocene among descendants
of these animals one species successfully occupied Australian waters had appeared.
The place of water predator in Australian ponds and rivers was shared among
different predatory reptiles. But the place of water herbivorous analogue of
beaver and musk rat was earlier vacant. It was occupied by Australian marsh
rabbit – descendant of rabbits introduced by humans. This small mammal had
kept rather recognizing appearance characteristic for rabbits though it has
got some adaptations to unusual habit of life.
This animal is able to swim perfectly and even to dive for 3 – 4 minutes. Strong
hinder legs are adapted to swimming: feet at them are wide. Usually this rabbit
swims having pressed forepaws to sides and making sharp simultaneous movements
by hinder legs. Ears of marsh rabbit are short and rounded, they can close
lengthways. Fur is short and rigid; it is white on stomach and cheeks and brown
on sides and head. Along back some faltering dark stripes pass. Tail is short,
white from wrong side.
Marsh rabbit eats leaves and tubers of water plants. Usually it foods in thickets
of marsh grasses but in case of necessary can dive to the bottom for tubers
of plants - arrowheads and aponogetons. Having dived marsh rabbit digs out
tubers by forepaws. Its ears thus are folded and pressed to shoulders.
Dwelling of this original rabbit is the hole in steep river bank. The entrance
to the hole is placed above maximal water level in the river and usually is
disguised. Holes of different individuals are united to one system and stretch
along river bank to tens meters. The settlement of marsh rabbits usually appears
in place where there are shallows with thickets of water plants and reeds.
In rabbit holes other animals, usually birds and small lizards, frequently
settle.
Settlements of Australian marsh rabbits include usually no more than one and
half tens of adult animals – food resources of river are more limited than
in places where rabbits lived earlier. Three - four times per year number of
colony considerably grows: at does posterity appears. Usually first time doe
of this species rises posterity being half year old. In litter usually there
are four - five cubs. It is less than at rabbits introduced to Australia by
people, but newborns of water rabbits are larger and also develop much faster.
At fortnight age young rabbit cubs start to investigate world around. They
leave a hole and gradually start to try plants eating by adult animals. Rabbit
cubs very cautiously investigate water and make first attempts to swim. It
is the most dangerous moment in their life – numerous predators – turtles,
snakes and fishes hunt for inexperienced young mammals not able to swim quickly.
And at any age predatory birds are dangerous to marsh rabbits and it is possible
to escape from them having dived or having hided in hole.
These animals feed in shallow waters having hided among leaves of marsh plants
rising above water. Because in this case the field of vision is limited at
marsh rabbits the system of sound dialogue is advanced. Resting rabbits communicate
with the help of short clicks. Having noticed the predatory bird flying by
above thickets rabbit utters shout of alarm – prolonged chirring. Having heard
this signal animals try to dive or hide under driftwood. To signal about the
underwater danger marsh rabbits have other signal – shrill whistles.
Despite of care and collective system of enemy watching hardly probable tenth
part from number of newborn rabbit cubs lives up to adult condition. And life
expectancy of adult rabbit seldomly exceeds three years.
Common
Australian rabbit (Australagus papiliotis)
Order: Lagomorphs (Lagomorpha)
Family: Hares (Leporidae)
Habitat: Western and central Meganesia (up to the eastern coast of Eyre gulf
and the zones bordering with the tropical rainforests at the north), various
types of biotopes, from dry savanna to temperate forest.
In late Eocene epoch, the southern remains of the large continent of Gondwana
broke in two great landmasses, Antarctica, and Australia, initiating the drift
of this enormous island to the north. As the result of many millions of years
of isolation from mainland, the Australian fauna became quite extraordinary
and unique; some of the most unusual creatures in the Cenozoic evolved here.
During late Holocene, the structure of this original fauna had considerably
changed, due to human activity: whereas many endemic species of this subcontinent
have disappeared, other ones, specially placental mammals were delivered from
other zones of the Earth. The introduction of foreign mammals in the historic
time, was in many cases more catastrophic than the direct action of human for
the native marsupials: then, kangaroos and other small herbivores had to compete
for food with enormous herds of sheeps, goats and rabbits, that eventually desertified
great areas of terrain. Foxes, dogs and cats, also competed with local predators,
and affected to the population of small marsupials and birds, by hunting them.
Though after the extinction of man, some of these ecological “aliens”, like
the placental carnivores, were eliminated by epizooties, others, for example,
dromedaries, rodents and rabbits, successfully adapted to the conditions, and
started to evolve, filling the left niches. Due to this event, Australian mammal
fauna during Neocene is a mix of three different stocks; most of the placentals
are constitued by herbivores; the descendants of dromedaries diversified into
various new species occupying many of the niches, nevertheless, there are also
several herbivorous marsupials, some of them reaching a great size. Species
of monotremes are not the most common creatures, though they’re more numerous
than during Holocene. Predators are basically marsupial; and the small and medium
sized ones usually prey on the ubiquitous and typical rodents and rabbits.
During the Holocene, the European rabbit (Oryctolagus cuninculus) was intentionally
introduced in Australia by the people. The propagation of these creatures occurred
incredibly fast and within some years they expanded almost to the whole subcontinent,
becoming a serious and uncontrollable plague affecting both humans and native
animals. Rabbit species successfully survived after the extinction of man and
endured the ecological accident that started to the Neocene epoch, becoming
one more important element in the local fauna... and the food chain.
During Neocene, Australian rabbits diversified into many species, occupying
some niches, some of them even leading a semiaquatic type of life. However those
species, still quite recognizable, they have kept the small size and the typical
shape of their ancestors. Because of the tendency of the ecosystems to rearrange
with the pass of the time, the population of rabbits is not so large as it was
during the historical time, it is controlled by the small predators that usually
prey on them.
Many of the species are contained in the new genus Australagus, that literally
represents the adaptation of rabbits to the different habitats in Australia.
The most abundant and less specialized species of the genus is the common Australian
rabbit. Body length in adult individuals is about 40-60 cm. Like all its relatives,
the Australian rabbit is quite similar to a normal rabbit, and differ in very
few aspects, nevertheless those species are quite remarkable due to a feature
that distinguish them from other lagomorphs. They possess typical rather long
and mobile ears, but when we look at their internal side, it can be appreciated
that an original pattern of speckles and oculate spots is scattered. This special
pattern, that vary in each species of the genus, quite remembers the colouring
pattern of butterfly wing. That determines the specific name of the common Australian
rabbit, A. papiliotis, which means “butterfly eared”. Ear colouring pattern
in these species is less advanced than others, but still it’s very captivating:
internal side of the ear is white, on it a great amber coloured oculate that
seems an eagle’s eye appears, there are also some dark speckles of different
shapes, including a stripe bordering the oculate spot by one side. This remarkable
colouring feature is a quite important element in the social behaviour of the
rabbits, used mainly in rituals and for recognition within neighbours, though
in some cases it can serve for frightening off predators. Ears can easily be
stretched widely, inclined to sides and folded back thanks to special nerves
on them and advanced muscles on their basis. Body colouration of the Australian
rabbit is yellowish brown with some dark spots at the sides, head, back and
paws are darker, stomach and tail are white. Around eyes there’s a white spot;
bordering it and along the nose bridge there is a black mask.
The common Australian rabbit has a wide diet range, it feeds on various types
of aliments, from grass, leaves, bush branches, roots, up to insects. As well
as its ancestors, Australian rabbit is a social animal living in colonies of
tens of individuals, in great, complex burrows that expand with the pass of
generations. The community is divided in social differentiated casts: there
are some dominant individuals, and various types of subordinates. While the
rank of the dominant females is immutable, in the case of males is constantly
changed. Cada certain period of time, in the colony, a ritual for the hierarchical
reestablishment is realised. In it, mature males flap their ears, and show the
intern colouration to their opponents. Those who have the most expressed pattern
become accepted by the dominant females.
6 times per year, females bring posterity to 8 blind, defenceless cubs. Dominant
females nest in the principal burrow, whereas the subordinated ones have to
do it in separated chambers.
When it’s reached a certain number of individuals, the overgrown colony has
to be divided into two groups, one stays in the burrow, and the other leaves,
searching for a new place of inhabiting.
Some more specialized relatives are adapted to different habitats:
Mountain
Australian rabbit (Australagus orophilus) inhabits the eastern mountains
up to the Great Dividing Ridge. It differs in greater size (adults can grow
up to 70 cm of length), strong constitution and quite small ears. These species
are adapted to life in the rigorous cold mountain habitats, they have broad
feet with slightly cornificated skin under them for jumping and skipping easily
on the rocks without receiving any harm. Fur is rich and fluffy, general body
coloration changes seasonally, during the winter is light grey, whereas in summer
it acquires a brownish shade. On the back some great longitudinal dark stripes
are stretched, stomach is white. Ears in these species are rather reduced and
rounded, an adaptation for living in cold conditions. Nevertheless the typical
ear pattern has not disappeared: A great yellow oculate with a black spot in
the centre almost occupies the whole ear, and some round dark stripes surround
it. It feeds on whatever edible that could find, including seeds, little bush
branches and moss.
Forest
Australian rabbit (Australagus sylvaticus) Similar to the common species,
though it is a bit smaller. Lives in woods and rainforests at northern and eastern
Meganesia. It possesses a contrasted coloration: top part of the body is rusty
brown with small light speckles, and stomach is white, both head and paws are
very dark brown. On cheeks and around eyes there’s a zone of white hair. In
these species, the colouration pattern of the ear characteristic of the genus
is taken to extreme level: a myriad of speckles and stripes and an oval oculate
of pink colour in the interior part of the ears, and a fringe of reddish long
hair on their top edges, form a spectacular, matchless ornament.
The forest Australian rabbit feeds on the various wood plants, bark and fruits
and nuts that fall from the trees.

Picture by Alexander Smyslov
Desert
Australian rabbit (Australagus lepoides) These species living in the
arid zones at the southwest part of the subcontinent resemble hares in many
respects, especially due to their slender constitution and long ears. This form
is adapted to the extreme conditions of the desert. It possesses rather long
feet for racing in the sand. Quite long ears (up to 20 cm), are penetrated by
a circuit of blood vessels; that serves for radiating the harmful heat. Fur
is short and velvety, coloured very light yellow. This colouration helps it
both to camouflage on the terrain and reflect the intense sunlight, avoiding
so the overheat. The sole dark zones are two speckles over the eyes and the
external part of ears. The ear pattern is rather degenerated, only some simple
faltering stripes and an elongated red coloured oculate appear. It feeds mainly
on grasses and roots, but during the severe drought season it can survive gnawing
little dry branches and even pecking on carrion.
Colonies of this species are quite small, each one is constituted by less than
ten individuals.
These species of mammals were discovered by Carlos Pizcueta.
Giant
lake rabbit (Barocuniculus capybaroides)
Order: Lagomorphs (Lagomorpha)
Family: Hares (Leporidae)
Habitat: marshlands around Carpentary Lake.
Evolution has given one serious limitation to marsupial mammals: due to their
mode of reproduction they are almost completely unable to develop an aquatic
lifestyle. Therefore, in the territory of Meganesia in the Neocene epoch aquatic
mammals belong to two other groups. These are various
platypi feeding on small aquatic animals and rabbits,
having become herbivorous animals.
Giant lake rabbit is one of aquatic species of rabbits inhabiting the coastal
zone of Carpentaria Lake, preferring wetlands with dry islands and extensive
thickets of marsh vegetation. This species of rabbit in the process of evolution
has acquired a significant convergent similarity with the capybara, South American
caviomorph rodent. This is a large herbivorous creature with broad paws and
sparse rough wool. It has almost lost the ability to jump: on land this animal
moves on outstretched legs, and only being in water it can push off with both
feet at once. Feet of the animal have elongated toes, which are connected by
a thick swimming membrane. The height of the adult at the shoulders is up to
40 cm, the weight is about 10 kg. In spite of the adaptation to an aquatic lifestyle,
this animal is able to run fast over short distances, but prefers to hide from
enemies in the water.
The coloring of its wool is gray with a white belly and a white spot on the
neck and shoulders, which prevents a predator to recognize the true contours
of the body. The area around the nose, the tip of the lower jaw and the cheeks
are white. The rear part of the body is darker than the front, and under the
tail there is a vast area of white wool. In case of alarm the animal lifts its
tail and shows this spot. Giant lake rabbit has large head with long whiskers
and powerful incisors. The ears are very short, with a valve inside that protects
the ear canal from water. The nostrils can close also. The eyes are large, shifted
to the top of the head.
This animal is a good swimmer; while underwater, it paddles all its legs alternately,
while swimming near the surface of the water. If it needs to dive, this animal
paddles its hind legs at the same time, holding its front legs on its stomach.
The basis of the diet is made of aquatic plants. Also this animal eats coastal
vegetation and tree seedlings in boggy areas of forests. With the claws of its
forepaws the rabbit digs up roots and tubers at the bottom of the lake and eats
them on the surface of the water. In case of danger the animal dives and can
stay underwater for up to 5 minutes at a depth of 5-6 meters.
It is a social species of animal, which settles in small groups of 2-5 breeding
pairs on dry islands in the marshes. Each pair of adult animals digs a burrow
under tree roots, a well-trodden path leads to the water among the vegetation.
The depth of the hole can reach 5 meters. The pair of adults bear cubs 2 to
5 times a year. The cubs stay with their parents up to 3 months, and then leave
the parent group. They lead an independent life in small groups which settle
on the banks of rivers and swamps. Typically, these groups give rise to new
settlements. Sexual maturity comes at the age of 4 months.
The life span of adult animals rarely exceeds 7 years. Enemies of this species
are large reptiles (for example, the giant snake eingana
(Eingana archonta)) and birds of prey.
The idea about the existence of this species of mammal was proposed by Tim Morris, Adelaide, Australia.
Translated by FanboyPhilosopher.
New
Azora tardolagus (Tardolagus novazorae)
Order: Lagomorphs (Lagomorpha)
Family: Hares (Leporidae)
Habitat: woods and bushy thickets of New Azora.

Picture by Amplion, colorization by Lambert
Initial image by Amplion |
In human epoch the fauna of islands had undergone changes:
various species of flora and fauna which could not appear at these islands by
natural way had been introduced to islands. Occurrence of these species has
broken natural balance of island nature, and ecosystem began to develop another
way. New species of animals, descendants of introduced species, had taken the
place in these ecosystems.
People had introduced rabbits to Azores (and to other numerous islands of the
Earth). These animals had survived in epoch of global ecological crisis and
evolved to species being a part of new ecosystems of Neocene. At the volcanic
island New Azora very large species of ground herbivorous animals had appeared,
the descendant of rabbit – New Azora tardolagus. It is the largest species of
terrestrial (not connected to sea) inhabitants of island. Tardolagus represents
herbivorous mammal weighting about 50 kg. By proportions of body this animal
resembles various herbivorous mammals evolved at the continents – fossil chalicotheres
(Chalicotherium), megatheres (Megatherium) and Neocene animals - ndipinotheres
(Ndipinotherium)and groundsloth
rodents (Tardimegamys). Tardolagus has long forepaws and inclined back.
Due to “semi-bipedal” constitution tardolagus can rise on strong hinder legs
with wide feet and gnaw branches of bushes and undersized trees. Standing on
hinder legs, this animal reaches growth of one and half meters. During the feeding
it tightens branches by forepaws on which large hooked claws grow, a little
bit similar to claws of ground sloths. At movement on four legs the animal bases
on external side of hands, turning claws by tips to each other. Because of massive
constitution tardolagus moves walking (its name literally means “the slow hare”),
and only in case of danger can run on small distances. Martillas,
large local predators, represent the main danger to this animal. Tail at tardolagus
is very short, long white colored hairs grow on it.
Wool on back, sides and hips is reddish-brown with dark spots forming faltering
longitudinal lines. Lateral parts of head are white, tip of muzzle is dark.
Forepaws from claws up to half of forearm are colored white.
Tardolagus eats rather soft forage – leaves, mushrooms and fruits of plants.
Its muzzle is extended, and forward incisors are rather weak. The animal willingly
eats dropped fruits of trees of laurel and heaths families, and with the help
of claws digs out roots of ferns and other plants.
This animal meets in places overgrown with bushes and undersized trees – at
riverbanks, in gorges and mountains of island. It avoids the areas overgrown
with woods of tropical type with poor underbrush, where there is small amount
of food. Tardolagus keeps in small groups of about ten adult individuals and
young growth. This animal had strongly changed habit of life in comparison with
ancestor: it had lost the ability to dig holes, and only makes beds in bush,
or uses natural shelters to spend the night.
At danger tardolagus prefers to protect itself actively – it rises on hind legs,
bites and beats the aggressor by long claws of forepaws. Thus it growls and
opens mouth, showing incisors.
Breeding rate at tardolagus is very low in comparison with ancestor. This animal
brings posterity once a year. At female one or two large cubs are born. They
are more advanced, than newborn rabbit cubs, but not so advanced, as posterity
of hares. For birth of posterity female leaves the group and searches for shelter
for itself and posterities. The newborn cub of tardolagus is covered with dark
wool, but blind and can not stand. Till first days the female hides posterity
in shelter – in cave or among bushes. The cub develops quickly: at the second
day it opens eyes and starts to explore world around. At week age the cub already
is able to walk. Grown up cubs leave shelter and follow mother to join the group
of congeners.
Young tardolagus begins able to bring posterity at the age of three years. Life
expectancy of this animal is till 20 years.
Falkland
xenolagus (Xenolagus subantarcticus)
Order: Lagomorphs (Lagomorpha)
Family: Hares (Leporidae)
Habitat: Falkland Islands, flatland and mountain areas overgrown with grassy
vegetation.

Picture by Alexander Smyslov
Studying of the biosphere of Earth of Neocene epoch indicates
that in various places of the planet there are animal species which couldn’t
appear in those places in natural way. Their existence is the result of human
activity in casual or purposeful introduction of species. Actually, human activity
broke the natural isolation of these or those districts and changed the course
of evolution of the species making ecosystems.
At Falkland Islands during the historical era there were no large herbivorous
mammals, and on islands the special type of tall grassy vegetation – tussock
– developed. In Neocene the vegetation of islands looks otherwise: the grassy
cover doesn’t reach great height and forms continuous extended turf. It is so
because the large descendant of introduced rabbit – Falkland xenolagus lives
on islands.
Xenolagus is very large species of lagomorphs: its weight reaches 30 kg and
more. Xenolagus has heavy constitution: it is four-legged animal with large
head and rather short plantigrade extremities. Unlike rabbit, xenolagus isn’t
able to jump, and in case of need it runs gallop, rising on finger-tips. The
body of animal is covered with dense fur which changes color depending on seasons.
In summer the body coloring of xenolagus is grayish-brown with dark chestnut
strip stretching on head, neck and along the back. Throat of animal is white;
white coloring sometimes stretches on breast and stomach. In winter fur hair
of animal are longer and colored lighter.
Life in cool Subantarctic climate led to reduction of the sizes of projecting
parts of animal body. Xenolagus has short rounded ears capable to turn in various
directions. The outer side of ears is black; on the base of ears there are white
strips. Ears risen up become appreciable – it is an alarm signal. Tail of this
animal is also short and covered with fluffy wool, white from the bottom side.
Xenolaguses are the sociable animals settling and feeding in common. Because
of large size these animals need the considerable territory for feeding; therefore
settlements of xenolaguses include no more than four or five pairs of adult
animals and their posterity. For housing xenolagus digs wide holes up to 5 meters
deep, ending with the living chamber. In holes of these animals flightless sea
birds cavescars often settle and dig for themselves lateral tunnels opening
in main tunnel. Holes of representatives of the same colony are located in the
neighbourhood, at distance of several meters between them. Animals from different
pairs know each other by smell and treat to “friends” rather tolerantly. Breeding
pair at this species is formed for one season of reproduction and breaks up
to the next courtship season; the hole is in possession of a female. Often happens
so that males of different pairs during new courtship season simply move to
another hole within the colony. The newcomer from outside should graze any time
in common with representatives of the colony that they got used to its smell
and presence. Usually young males move from one colony to another, whereas females
born in the colony dig their holes near parental one or occupy the free holes
in colony.
Due to size enlargement and increase of survival rate of posterity rate of reproduction
of xenolaguses became much slower, than at their ancestors. Once a year female
of this species gives rise to the litter of 4-6 cubs. The newborn xenolagus
weighs about 300 grams, but quickly gains weight and in two weeks weighs about
one kilogram. The newborn individual is covered with wool, but eyes and ears
open only at the end of the first week of life. The young animal starts to pass
to adult diet gradually at the age of 3-4 weeks, but stops the milk feeding
only at two month age.
The sexual maturity comes at the first year of life; in litter of young female
it may be only 1-2 cub. Life expectancy reaches 20 years.
Translated by Alexander Smyslov.
Alpine
burrower (Procaecolagus visibilis)
Order: Lagomorphs (Lagomorpha)
Family: Rabbit burrowers (Caecolagidae)
Habitats: Southern Europe, northern slopes of the Alps, mountain areas; the
area of forests and alpine meadows.

Picture by Alexey Tatarinov, colorization by Biolog
Initial image by Alexey Tatarinov |
Eurasia in real had never been an isolated continent. Only
in early Cenozoic the midland seas had separated it into some fragments for
a short time. But later water had receded, and the continent became the whole
again. Connecting in various geological epoch to Africa and North America, Eurasia
every time took part in exchange of flora and fauna representatives. Therefore
the evolution of new endemic family of animals in Eurasia is rather rare event.
One endemic family of Eurasian mammals is rabbit burrowers family (Caecolagidae),
including the descendants of rabbits specialized to burrowing habit of life.
In area of steppes and forests of Europe there are some species of rabbit
burrowers (Lepospalax), herbivorous mammals specialized to underground
life. The species of animals close to them, a relict of early radiation of family
is Alpine burrower living in the Alpes.
As against to species more evolutionally advanced, this animal has kept small
eyes, hardly distinct among wool. It has very bad sight sense: Alpine burrower
does not distinguish colors and is short-sighted. Lack of sight, however, is
quite compensated by keen sense of smell and hearing.
The high-mountainous climate is hard; therefore Alpine burrower is larger than
its plain and forest relatives and also has denser fur. It has muscled cylindric
body about 55 – 60 cm long. Wool of animal is one-colour, dark brown or black
at some individuals. Due to such colouring these animals are easily heated in
rays of cold mountain sun.
Skull of animal is very strong; frontal and jaw bones are very thick. The forward
pair of incisors is directed forward. These incisors are larger, than at plain-dwelling
species; enamel layer on them is folded, that additionally strengthens their
durability, and the teeth bases are stretched far back along the top part of
skull. Strengthening of incisors is connected to necessity to dig tunnels in
stony ground. Calvaria at Alpine burrower is wide. It carries out a role of
protective helmet and can sustain significant loading – with the help of head
the animal can raise and roll away stones weighting over 30 kg. Edges of calvaria
overhang above eyes, protecting them from ground ingress. Nostrils of animal
close at digging, and the mouth is protected from ingress of ground by lips
outgrowths. Auricles of animal are short, rounded and gristly. The special outgrowth
closes acoustic duct during the ground digging.
Paws of Alpine burrower are short and muscled. Forepaws are armed with strong
claws, and muscles are attached to wide shoulder-blades. Tail of animal is very
short.
Alpine burrower appears often enough on ground surface – at night animals eat
grass and browse leaves of bushes, and in day time occasionally come out to
get warm on sunny place. Similarly to their steppe relatives, Alpine burrowers
drag in holes bush branches and bunches of grass dug out at the surface, in
order to eat it in safety. It promotes enrichment of ground by nutrients. In
addition to it Alpine burrower eats sappy underground parts of plants. It has
immunity to plant poisons and can eat poisonous bulbs and tubers without any
harm for itself.
Alpine burrower digs the vast systems of tunnels. The significant part of its
holes is located at the depth of 20 – 30 cm, but tunnels for wintering stretch
to the depth of several meters. Its building activity promotes aeration and
hashing of ground, but this species can live only in places, where ground layer
is thick enough for digging of tunnels. This species lives in small colonies
numbering up to ten adult females and their posterity. Males have no constant
territories and move from one colony to another, searching by smell for females
ready to pairing.
Two times per one year female gives rise to cubs. In litter usually it is up
to 5 – 6 cubs though large female in good physical shape has an opportunity
to give birth to ten cubs at once and to bring them up successfully. They become
sexually mature at the age of 5 – 6 months.
Beringian hutmaker (Architectona beringica)
Order: Lagomorphs (Lagomorpha)
Family: Haymakers (Ochotonidae)
Habitat: Beringia, Northern Asia, Big Kuril Isles; in New World from Alaska
up to Greenland, to the south this species penetrates rather far along mountain
ridges.
During Holocene epoch in “shadow” of hares there were their less appreciable
relatives – haymakers (Ochotona). They are similar to rodents, and have no
features of anatomy characteristic for hares – long back legs, permitting to
develop high speed. But these lagomorphs have curious features of behavior
– they gather plants and make of them hayricks for winter (for what they are
named). In addition some species strengthen hayricks by stones and branches.
During the period of transition from Holocene to Neocene this course of evolution
had been picked up and successfully advanced. Evolution successfully realized
the task of survival of these small mammals in some steps. In glacial steppes
of Central Asia where it is a lot of grass, climate is typically continental
and winter is very cold, haymakers had became larger similarly to all polar
animal. Accordingly, they began to provide a plenty of hay, and to protect
it from wind at them feature of behavior was developed: animals began to throw
large branches atop of hay. Surviving in steppe in small colony is rather difficult,
and therefore, animals began to unite to the “towns” numbering tens of adult
individuals: it helps them to increase efficiency of hay reserve.
But to prepare dry grass for winter yet does not mean to spend winter successfully.
Large herbivores may easily find and to have eaten all hay provided by haymakers.
And in steppes of an ice age herbivores were numerous. It were representatives
of hoofed mammals, and also large rodents, new species have appeared in ice
age. To save forage gathered to the winter, separate animals began to protect
stocks, attacking on herbivores, and driving them away by cries, jumps and
even bites. And then some populations had made one more step: they had started
to use stocks of hay as shelters, gradually eating them up from edges and arranging
an inhabited hole in middle of haystack. Gradually among them animals never
settling in holes and other underground shelters had appeared. This step of
evolution had resulted to changes of “house-building” behavior: “having come
off” from dependence on available holes, rocks and crevices, animals have started
to improve “architectural” behavior. They have started to build inhabited tents
separately and did not mix them with stocks of forage. Inhabited tents of these
animals began to be improved in features of design: at their construction a
plenty of branches of trees and prickly bushes was used. For protection against
enemies animals began weaving of branches dense walls, and the internal part
of inhabited tent is covered with grass for warming. So the inhabited tent
is completely separated from stock of forage in behavior of animal, and the
building behavior evolved irrespective of food- providing one.
The Beringian hutmaker had kept features of haymakers in its appearance: it
is similar to short-legged tailless marmot. Auricles of animal are very small,
and eyes are large: it is an adaptation for life in conditions of polar night.
The body of Beringian hutmaker is covered with rich fur becoming longer to
the winter. The fur is colored red with longitudinal black strip along the
back. Wood populations of hutmakers also have dark speckles on back, and often
strip is faltering. The length of body of this animal is about 40 cm; female
is larger than male.
This species builds shelters as tents of branches, warmed from within with
grass and bird feathers. For construction of dwelling the Beringian hutmaker
chooses the prickliest branches, including coniferous trees ones. Building
their shelter, animals accurately cut them out by strong incisors from the
tree-trunk, and drag to building tent. To not give out their presence, animals
cut branches on bushes from the edge of feeding territory. For an internal
lining of house animals gather soft plants, thin stalks of grasses, wool of
sheded animals, and feathers.
In tent up to 2 meters in height there are some inhabited chambers at two or
three floors. The population of one tent makes approximately 4 – 5 pairs of
adult animals, and their posterity. Near to inhabited tent stocks of food for
winter are located: animals gather some stacks of grass, and surround an inhabited
tent by them – it complicates their search by predators. Beringian hutmakers
start to provide a forage approximately from second half of summer. To dry
up a grass and to not spoil it during a rain, some animals in clan constantly
watch weather: if there are signs of rain, they quickly carry off drying grass
under trees. In summer the majority of animals are occupied with preparation
of stocks of forage – they run to glades, cut the overgrown grass, and drag
bunches of picked plants to the common storehouse.
In winter to keep stocks of a forage is vital for clan, therefore in winter
in stacks of hutmakers adult animals and teenagers often “are on duty” – usually
they are males having subordinated position in clan or young animals not having
families.
The adult females already had giving rise to cubs, stand at top of hierarchy
in clan. Per one year at them it may be 2 litters. Cubs are born well advanced,
with opened eyes and covered with wool. They early become independent: already
at week age cubs actively eat forage of adult animals. The young growth, which
has been born at the end of summer, spends winter with parents, and the posterity
from first packs arranges new settlements independently. They quickly grow,
and can give posterity the next year. Life expectancy of Beringian hutmakers
can reach 10 years.
Young animals frequently “convert” for habitation old stocks of forage, strengthening
walls of stacks by branches, and arranging inside inhabited chambers. Doing
it, they as though repeat evolution of dwelling of the hutmaker - from stock
of forage to constant habitation. Usually young females from growing old clan
do it. Gradually they move to the true specially made tents, and their posterity
occupies their habitation.
Sun flying fox (Posteidolon solaris)
Order: Chiropters (Chiroptera)
Family: Megabats (Pteropodidae)
Habitat: Zinj Land, tropical woods.
When till the ecological crisis at the boundary of Holocene and Neocene some
birds had become extinct, including predators and the majority of tropical
groups like parrots, bats had an opportunity to occupy new habitats which were
formed in Neocene. Some species of these mammals began much more active in
day time, and in some cases compete to birds as equals. An example of such
species of chiropters is the sun flying fox, the descendant of palm flying
fox (Eidolon helvum), living at the Eastern-African subcontinent (Zinj Land).
Appearing in conditions of partial isolation, this species had appreciably
changed habit of life, had settled in rainforest, and had partly occupied an
ecological niche of animals of forest canopy.
This is rather large chiroptere: body length is about 30 cm, and wingspan is
almost 1 meter. The animal differs in rather massive and strong constitution,
and weighs about 2 kgs. Wings of sun flying fox are shorter and wider, than
at other representatives of order: it is directly connected with the habit
of life of this animal. Sunny flying fox does not like to fly long, limiting
to short flights from tree to tree. In rich tropical wood skill to fly quickly
is not always expedient, but well clambering animals get appreciable advantage
in survival. More often animals of this species prefer to climb on trees, clinging
by hind legs and hooked claws growing on free thumb of wing. Thumb at this
animal is advanced much better, than at other chiropters: it is thicker and
stronger, and claw on it permits this animal to hang under the branch.
Hanging to branches upside down, the sun flying fox dexterously moves on them
in searches of fruits. By manner of movement it resembles the sloth, but moves
much faster than it (this chiroptere accelerates speed up to 5 km per hour).
When it is necessary, animal can even make small jumps from branch to branch
like a monkey. This animal reluctantly flies, making it only in case of emergency
– flying to other tree in searches of food, or escaping from clambering predators.
In flight sun flying fox feels like not so confidently, as other species of
chiropters: it frequently flaps wings (its flight resembles flight of gallinaceous
birds), and sometimes at all prefers to glide from tree to tree similarly to
flying squirrel. The importance of flight in life of this species is so low,
than even the animal having injured flying membrane, can exist and take part
in breeding normally.
The color of wool at sun flying fox partly justifies its name: fur is darkly
yellow (“honey” shade) or sometimes light brown. On stomach wool is brighter,
than on back. Membranes of wings are covered with thin wool on the top side.
On neck of animal there is an ornament: collar of rusty-red fur, especially
advanced at males. Skin of wings is dark brown, contrasting with color of fur.
The head of the solar flying fox looks similar to head of dog with small ears
and large reddish eyes. It is 0rather wide, because animal eats various vegetative
foods and at it chewing muscles are advanced. On skull of this chiropters even
the low longitudinal crest serving for their attachment is developed. This
chiroptere usually eats fruits of any degree of ripening; from unripe up to
began to rot, but it also supplements a diet with leaves and young sprouts.
Sometimes sun flying fox includes food of animal origin to its diet: eggs and
nestlings of birds, frogs and insects. Animal eats fruits simply hanging on
branch near to them, and catches small animals by tenacious hinder legs. Their
flexibility permits to this chiroptere to bring the caught prey by hinder leg
right to the mouth.
As against flying foxes of other species, this species keeps solitarly. Sometimes
on one fructifying tree it is possible to meet several animals of this species,
but they keep separately, not coming nearer to each other. In morning time
animals of this species like to “sunbathe”: climbing up on light up branches,
they stretch wings, having put their inner side and stomach to beams of rising
sun.
The breeding season takes place till all year. Males ready to pairing, involve
females by special “warbles” sounding like long series of clicks. To meet the
female the male prepares special branch: on thick horizontal branch he makes
some odorous marks, using musk secretions of specific gland on chin. The same
secretions impregnate his fur collar, strengthening the smell of animal. Female
ready to pairing, finds these marks in forest canopy, and waits for the male
near of one of them. To draw his attention, the female utters short calls.
After pairing female leaves male’s territory and he continues marriage appeals.
Once per one year at the female one cub is born, occasionally there are twins.
The female holds posterity in the “cradle” formed by stomach and wings. First
some days of life the cub is so helpless, that the female, compelled to fly,
carries him in mouth. Later the cub becomes covered with wool and begins able
to keep by mother during her flight. Approximately at three-week age cub starts
to study to climb on branches. Posterity stays with mother for a long time:
even having studied to fly, and having reached 70 % of weight of adult animal,
the young animal does not leave mother. Because of these circumstances the
rate of reproduction at sun flying foxes is very slow: for one year the female
can give birth to posterity only once (or two times if the first birth happens
in the beginning of current year and the second one happens at the end of year).
But the posterity is teaching by mother in skills of food searching in tropical
wood, learns terms of fructification of various trees and ways of protection
against enemies. Besides the female actively protects posterity in first months
of life, and its survival rate is rather high.
The idea about existence of this species of animals was supposed by Bhut, the participant of forum.
Yellow
Browed Brush-tongued Bat (Florapteropus australis)
Order: Bats (Chiroptera)
Family: Megabats (Pteropodidae)
Habitat: The north of Meganesia, tropical rainforest.
Bats are among the most numerous of mammals in the age of man, one would expect
their success in later epochs, and so it goes in the Neocene. Bats reached Australia
early in the age of mammals, by being able to fly, and they still prosper there.
From the common Australian Flying Fox (genus Pteropus), descends a genus of
bats which feed entirely on the nectar of forest trees, avoiding competition
with birds by feeding at night. Previously during the Holocene, similar animals
had appeared, but were the victims of the anthropogenic extinctions caused by
forest clearing.
Nominal example is the Yellow Browed Brush-tongued Bat of the tropical north
of the continent, smaller than its large ancestors, with a wingspan of only
40cm and a weight of 80 grams. These bats typically rest by day in small groups
scattered throughout the forest, taking wing at night to forage in the treetops
upon various flowering trees, night blooming flowers and those that do not close
at night are preferred, though they will rob nectar from day flowers which have
closed.
The animal has a relatively narrow snout which is more acute than its ancestor,
but this is obscured by a brush of whiskers which is conducive to the spreading
of pollen from flower to flower, these bats importantly pollinate many kinds
of night blooming flower. The tongue of this bat is slender and covered in a
prominent brush of hair-like papillae which it uses to extract nectar from flowers.
Fur is colored fawn grey with a saddle of chocolate colour across the shoulders,
and the top of its head is a golden yellow. Wings are relatively broad but not
as big proportionately as those of typical fruit bats such as its ancestor.
The call of this bat is a high-pitched braying screech.
Breeding season occurs during the wet season when there is a flush of growth,
though in a good year there may be two mating seasons. It is at this time that
they accumulate in large colonies segregated by sex, bachelor groups of males
compete to woo females out of a large selection. Females will choose a male
to mate with based partly on his vigour and loudness, but also by a gift of
regurgitated food. A single pup is born but occasionally there may be twins,
gestation is about 155 days or slightly longer. Pups become independent from
their mothers after about 8 months. Lifespan is up to 14 years.
A close relative is the Ginger-Headed
Brush-tongued Bat (Florapteropus major), which is found in temperate
and subtropical woodlands in the southeast of the continent. This bat specializes
in feeding upon the nectar of proteas and eucalypts, which generally have flowers
which do not close at night, and which drip nectar profusely. This form is slightly
larger and more pugnacious than the nominal form, as well as having fur of a
darker grey; the color on top of the head is a ginger orange. Breeding season
varies in this form to reflect the seasonal climate, the main breeding season
is in early spring but a second season may occur in late summer.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Painted
Fruit Bat (Toxicopterus splendens)
Order: Bats (Chiroptera)
Family: Megabats (Pteropodidae)
Habitat: The north of Meganesia, tropical rainforest.
Having poisonous flesh in order to repel predators is usually the domain of
fish, amphibians and invertebrates, and also some rare birds, but in the Neocene,
some kinds of mammals evolved
the ability of accumulation of poisons from their food in their flesh.
Painted Fruit Bat is descended from smaller members of the genus Pteropus, and
like those, it feeds entirely on fruit, this particular bat only feeds from
a few distinct types of trees, which have exceedingly poisonous fruit. This
is related to its method of defence and survival, its flesh is extremely poisonous,
any animal consuming it will quickly get extremely ill and possibly even die,
so predators quickly learn to avoid it. Thanks to the nature of its defence
mechanism, this solitary fruit bat feeds by day.
Coloration is directly related to method of defence, fur is boldly striped in
black and white longitudinal stripes, almost recalling a skunk (but fur is shorter),
and the skin of wings is bright orange, blotched between the fingers with black
patches. Face has a “mask” of white fur on a black background, bare snout, feet
and hands are dark orange. Wingspan is a meter or less and weight is up to 600
grams. Apart from coloration, this bat greatly resembles its ancestor in shape
and appearance. Call is high pitched yapping or yarring, when confronted with
a predator it may spread its wings and produce a loud call of a high strident
pitch as a warning.
Breeding occurs during the wet season, and in a good year it may experience
more than one breeding event. This is when both males and females gather in
large groups in order to court. Males display their colourful wings and produce
loud calls in order to impress the females, and may perform acrobatic display
flights. Mates are chosen based on vigour and loudness, but also upon gifts
of regurgitated food. A single baby is born, but rarely twins, gestation is
up to 160 days, and the young become independent at 8 months. Initially, young
are an all over reddish brown with opaque wings, and it is only as they reach
adulthood, and feed profusely on poisonous fruit, that they gain their warning
colours. Lifespan may be up to twenty years, somewhat longer than its relatives.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Striped
singing bat (Musicilio striata)
Order: Chiropters (Chiroptera)
Family: Common bats (Vespertilionidae)
Habitat: subtropics of Europe, southwestern coast of Fourseas.
Chiropters had evolved at the Earth right at the beginning of Cenozoic, appreciably
later, rather than birds. Probably, the nocturnal habit of life had helped these
animals to avoid a competition to birds. Chiropters had achieved significant
success in evolution - in human epoch they made up about a quarter of variety
of mammalian species. Among them the species occupying various ecological niches
had evolved: herbivores, nectarivores, insectivores, predators and even blood-sucking
parasites. Chiropters of some species form colonies which number is measured
by millions of individuals.
Chiropters had rather safely gone through global ecological crisis. Among them
highly specialized tropical species, and some species of small total number
had become extinct. But right after stabilization of environment conditions
chiropters had entered new blossoming. Some Neocene chiropters during the process
of evolution had developed very original features of behaviour.
Some kinds of separate genus of chiropters – singing bats (Musicilio) live in
warm-temperate and subtropical areas of Europe. They belong to the number of
medium-sized representatives of group – wingspan at them does not exceed 30
cm, and more often it is even smaller.
These animals have not only echolocation, but also well advanced system of sound
communications. Voices of males of these mammal differ in originality. At different
species of these chiropters at similar sounding echolocating signals voices
in heard part of sound range strongly differ. These animals are able to utter
various sounds – from single clicks and simple monotonous trills up to rather
complex signals, including a sequence of sounds of varying frequency.
Striped singing bat lives in subtropical forests of Europe. It is one of typical
representatives of genus. At these animals wool is colored black, and on this
background from shoulders along the back two wide white strips stretch. Hairless
membrane of wings is also dark. Male and female at this species differ in colouring.
At males strips on back are wider, than at females, and on throat often there
is a white spot. The female of striped singing bat has two thin faltering strips
on back, and besides it is larger a little, rather than male. It is the most
impressively looking species of the genus, other species are colored less brigher.
Muzzle at all singing bats is short and wide. On the nose high leaf-shaped outgrowth
grows, on each side of which chink-like nostrils extended vertically open. Mobile
edges of nostrils are supplied with special muscles which change width of nostril
and density of its closing. “Playing” by edges of nostrils, singing bats “sing”
at the exhalation. They send echolocating signals by mouth, as usual bats.
At singing bats there are large oval ears with pointed tips and large hircuses.
At striped singing bat edges of ears are covered from external side with short
white fringe.
Wings of these chiropters are short and have rounded tips. Long tail is half
included in interfemoral membrane. Flight of these chiropters is slow and flitting.
Singing bats are active in twilight and at night. They are mainly omnivorous,
and also eat large flying insects (moths and beetles) and ripe fruits of plants.
Various species have preferences in diet, but the species eating more vegetative
food dominate at the south, and at the north migrating insectivorous species
prevail. But even northern species during the fruit ripening time pass to partly
vegetarian diet.
The courtship season at singing bats comes in summer, and one cub is born at
the female in spring of the next year. At each species of singing bats the specific
song differs from those at close species. Males of striped singing bat involve
females with the sounds similar to twittering. Every male displays itself, hanging
on the branch from below and arranging the “concert”. Sometimes on one tree
some males gather, and they organize the original musical competition. However,
at these animals males not always suffer the presence of competitors – it happens,
strong, but badly singing male simply disperses contenders to near trees. But
it has very insignificant chances of success – during fight it is compelled
to interrupt song, and females search for others males. Every male tries to
sing as long, as it is possible, not interrupting. Calling the female, male
utters short abrupt trills. When the female answers his appeal, male starts
to sing actually courtship song – series of whistling sounds. First “syllable”
of his songs is the longest, and each subsequent one is shorter, than previous,
and its tone is higher. During the song performance male stretches wings and
displays to the female its own size.
In the afternoon singing bats prefer to hide in shelters – usually in hollows
of trees or under bark. They do not form big colonies, and search for society
of congeners only during migrations or in courtship season. Striped singing
bat belongs to migrating species, and in autumn, with approach of time of cold
fogs going from Fourseas, it flies to the south – to mountain valleys of Asia
Minor. These chiropters make flights at night, and hide in various casual shelters,
gathering in groups of some tens individuals in day time. The disturbed animals
are very aggressive. They hiss to the enemy and put painful bites.
Some birds may be enemies of singing bats. During night hunting owls attack
them, and large gospodar woodpecker
pecks hollows where these animals hide, and eats them.
Along the coast of Fourseas, in subtropical areas of Europe with soft climate
other species of singing bats are found:
Common
singing bat (Musicilio cantans) is the most widespread species of genus.
It lives at the extensive territory: from woods in Central Europe at the west
up to forest areas in deltas of rivers flowing to Fourseas at the east. Populations
from edges of area make migrations, and inhabitants of southern areas – Caucasian
Peninsula and Balkan – are settled and run to short-term hibernation. This species
of chiropters has dim colouring: male is black with brownish shade and white
spot on throat; female has dark brown wool, and it does not have spot on throat.
The only ornament in colouring of this species is thin strip of white wool at
edges of ears. Ears are rounded; nasal outgrowth is wide and short. Wingspan
makes about 25 cm. The voice of male represents a simple trill of short abrupt
sounds.
Greater
singing bat (Musicilio grandis) is the largest species of the genus
– at some individuals wingspan exceeds 35 cm, though usually it is less. Colouring
of wool at this species is very light – white with yellowish shade; on head
there is darker “cap” (at the male it is bright red, at the female brown). This
species lives at the south – in mountain forests of Balkan and Asia Minor, near
borders of salt and hot Mediterranean Lowland. The basis of ration of this animal
is made of fruits of trees and large insects. Courtship call of the male sounds
like sharp abrupt serialized singing.
Caucasian
singing bat (Musicilio caucasica) inhabits mountain and coastal forests
of Caucasian Peninsula, and migrates for wintering to mountain valleys of Asia
Minor. This small species of bats (no more than 20 cm in wingspan) utters the
melodious modulating singing similar a few to the voice of small songbirds.
This species of chiropters is especially active in twilight and in dawn time.
The wool is colored grey with dark strip along the back; at males on throat
there is a yellowish spot. This species eats insects, and only in middle of
summer passes to feeding on soft and overripe fruits.
Aotearoan
Long-eared Bat (Novonyctophilus macrotis)
Order: Chiropters (Chiroptera)
Family: Common bats (Vespertilionidae)
Habitat: New Zealand, both forested areas and open country, sheltering in caves.
A natural vagrant migration of animals from both Australia and the South Pacific
to New Zealand has regularly occurred in the Holocene. Native bat species there
were badly endangered and subsequently became extinct, but were soon replaced
by Vesper Bats that had migrated from Australia. One such species is Aotearoan
Long-eared Bat, which has changed relatively little from its Australian ancestor.
This species is of mid size for a micro-bat, weighing as much as 12 grams with
a wingspan of up to 30 centimetres. It generally resembles its ancestor, body
fur is of dark colour and wings are pinkish grey and translucent, ears are long
and very prominent. Animal is an insectivore, feeding on flying insects of various
sorts but also plucking spiders from their webs, echolocation is high-pitch
and faint, but effective. This bat differs from its immediate ancestors in that
it roosts colonially, as opposed to singly in tree hollows and ledges, as its
ancestors did. This may be a strategy to adapt to the cooler conditions present
in some parts of New Zealand.
Breeding season runs from March to May, but most mating occurs around April.
Females retain sperm over the winter months and ovulating and conceiving in
August and September, young are born by November, when it is warm. Females give
birth to twins, which remain clinging to the mother, apart from when she departs
to feed at night, she finds them again by the sound of their call. Babies are
able to fly within 3 months and weaning occurs 2 months later. This species
forms large colonies in the mouths of glacially formed caves which are common
in New Zealand; a single colony may number up to a thousand or more individuals.
Large amounts of guano are produced which form the basis of a biota of scavengers
and predators. Commonly preyed upon by owls, as well as occasionally mammalian
predators such as wildcats or species of carnivorous bat, reptilian predators
may include large Mouse-eating
Gecko, which may venture into their caves. Lifespan may be up to 20 years
but usually shorter.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Cat-headed
pipistrelle (Hirtofalco felinocephalus)
Order: Chiropters (Chiroptera)
Family: Predatory bats (Carnonycteridae)
Habitat: tropical woods of Central Africa.

Picture by Lambert
After extinction of majority of predatory birds species representatives
of other groups of vertebrates began to develop their ecological niche. Usually
various birds became flying carnivorous creatures, but some species of mammals
from chiropters order successfully competed to them. Some bats passed from insects
to more sizeable forage: they began to hunt birds and small mammals.
Cat-headed pipistrelle is predatory day time species of chiropters, descendant
of one of African pipistrelle (Pipistrellus) species. Because of injurious habit
of life at this animal the structure of body has considerably changed. Cat-headed
pipistrelle had turned to day time predator, and now it hunts birds and small
animals in trees crones finding them with the help of sight. At this species
eyes are very large and directed forward: sight is binocular. Ears are long,
triangular and peaked. The skull of cat-headed pipistrelle is short, jaws are
rather wide: its head is similar to comical portrait of cat. The mouth of animal
can open widely – it is connected to mode of catch killing: this chiroptere
kills caught animals by sting in head. Its molars because of it have got the
special form: they are prolonged and work as edges of scissors crushing backbone
and skull of catch (the similar adaptation was at fossil “marsupial lion” Tylacoleo).
Outgrowths on nose characteristic for the majority of bats at this species are
small and echolocating abilities are reduced – catch can hear voice of bat and
hide. This animal weighs about 300 grammes having wingspan up to 70 cm. Colouring
of wool is reddish-brown, ears are black and above eyes there are black stains.
On chest of male there is yellowish spot.
This species of chiropters hunt in forest among branches, therefore its flight
differs in maneuverability and high speed. Wings are long but rather wide: bat
easily can change direction of movement and fly round branches. Back extremities
of animal are almost free: the flying membrane is attached only to external
edge of hips; short tail has only small skin edging on its basis. Hinder legs
at this bat are very tenacious, the toe and little toe can oppose to others,
why the grip of this animal becomes doubly stronger. On toes hooked claws grow.
Speed of reaction at cat-headed pipistrelle is so fast that this mammal can
easily grip birds by each paw promptly having flown by through their flight.
Mammal eats caught prey somewhere on branch. Pipistrelle plucks caught birds
by hinder leg holding in mouth (on the contrary it flays small mammals by teeth
holding in paw). The animal eats catch with bones easily chews them by powerful
jaws.
Each pipistrelle has hunting area in forest canopy vigilantly protecting from
neighbours. When two pipistrelles meet at the border of territories, they begin
menacing demonstrations which however do not pass to combat: animals are afraid
to injure flying membrane, therefore teeth and claws are out of use. Animals
open wings, flap by them to the contender’s side and loudly squeal at this moment,
widely opening mouth and showing canines. After this bloodless, but noisy duel
both animals mark borders of territory by urine and peacefully miss each other.
Twice per year in breeding season males “paste up” on borders of territory odorous
“announcements” for females with the help of secretions of musk glands on throat.
Female ready to pairing hangs to branch near to such mark and waits when male
begins to inspect borders of its possession. Sometimes it even rubs stomach
against male’s mark to interrupt its own smell and to not cause aggression in
it. If the male is to her liking female forms pair with him and moves on his
territory. Pairs at cat-headed pipistrelles are formed not only for breeding
season, but also for all time while female look after posterity – two and sometimes
even three cubs. At this time male is compelled to hunt almost constantly. Cubs
are born naked and blind, but quickly open eyes and become wooly. First time
female carries them on itself but later starts to leave them on branch. Approximately
since the second week of life cubs gradually pass from milk to meat food – in
the beginning they eat semidigested meat, then fresh one and at last at bi-monthly
age they study to catch small animals independently.
Sexual maturity comes at the age of half-year. Life expectancy is about 10 years.
Silent
flying wolf (Pterolupus silentiosus)
Order: Chiropters (Chiroptera)
Family: False vampires (Megadermatidae)
Habitat: light forests at the south of Meganesia.

Picture by Lambert
After mass extinction evolution can make unexpected moves
and then in ecosystems very original species of live creatures evolve, occupying
in ecosystems a role unusual from the point of view of the human. It has taken
place so in Neocene Meganesia (the continent uniting Australia and New Guinea).
Here predatory medium-sized birds were pressed by very unusual representative
of chiropters which has received the name the flying wolf.
In Holocene in tropics of Old World large chiropters, flying foxes, had widely
settled. They had received the name only for features of shape, because they
were vegetarians. The flying wolf from Meganesia bears the name more deservedly.
Its appearance does not resemble wolf’s one at all, but this chiropteran has
surpassed its ground wingless “prototype” in injurious bents and dexterity of
prey catching. Flying wolf belongs to suborder Microchiroptera and is the largest
species of bats of Neocene. Wingspan of this animal is about 150 cm, and weight
reaches 1 kg. Body of this animal is muscled, on chest the keel (atypical formation
for small chiropters, which is more typical for large flying foxes) is well
advanced. Wings of flying wolf are long and pointed, and flight is very fast
and maneuverable. The thumb on wing is well advanced and mobile; with its help
flying wolf holds catch when eats it, and also clings to bark of trees and rocks.
The wing membrane of this large species of chiropters in addition is strengthened
by collagen fibers which stretch from bones to edge of wing.
The tail of flying wolf is approximately equal by length to back extremities.
The interthign membrane completely includes it, but covers back legs approximately
up to knee joint. The membrane of wings also covers legs only up to knees. Shins
of this animal are lengthened and muscled, well adapted for catch seizing. Toes
are mobile and armed with hooked claws from which the small animal cannot escape.
On little toe the especially large claw, with which help the animal kills the
seized animals, is advanced.
The wool of animal is colored brown, on throat there is a spot of yellow wool.
Eyes are led round by rings of white wool. Membranes of wings are colored dark
grey.
Flying wolf hunts in day time and in twilight, and has good color sight. Echolocating
abilities at this chiropteran are substantially lost (this animal does not distinguish
small objects), and express only during the flight in twilight. During day time
hunting flying wolf does not use an echolocation at all (hence its specific
name). In connection with this feature the muzzle of flying wolf is partly lack
of skinny outgrowths characteristic for bats. On nose bridge of this animal
the short vertical outgrowth grows, and small skinny platen surrounds nostrils
from outside. Ears are rather short and triangular-shaped with pointed tips.
They are connected to each other in the top part of head, and their tips are
turned in sides.
This species of bats is an active predator, and eats various vertebrates of
small and medium sizes. More often it hunts birds and small ground or climbing
animals – mammals and reptiles. Large eyes of this predator are shifted forward
and provide good binocular sight.
During the hunting flying wolf grasps catch by hinder legs, and sticks claws
deeply into it. The sharp claw on little toe puts to catch deep wounds from
which it bleeds profusely and perishes. Flying wolf especially frequently attacks
various birds – pigeons, small parrots and large passerine birds. The predator
hunts them not hiding: it frightens away birds, and rushes through their flight,
trying to grasp catch by paws. Sometimes it succeeds to seize two birds at once.
This chiropteran attacks small animals from air, and tries to kill by claws
at once. Flying wolf does not like to hunt among rich trees where the probability
to injure the wing membrane is high. However it dexterously seizes various tree-climbing
animals from large branches and tree trunk.
The courtship season at flying wolf comes at the end of rain season. At these
aggressive bats each individual protects the territory from neighbours, and
because of it pairing can become complicated. The male is larger than the female,
but even in this case there are reasons to him to be afraid of her – protecting
the territory, female attacks wings of the male. Therefore, to constrain her
aggression, male presents the female with small gift – killed small animal.
If the female is ready to pairing, she accepts it, and till some days animals
keep together and sleep on one tree. For these days the male is coupled many
times to the female and drives off other males from her. Then the female banishes
him.
In the beginning of the next rain season female gives rise to one large, but
helpless cub. The first weeks of life she flies to hunting with it, but later
she starts to leave it in tree-trunk hollow. Development of the cub proceeds
about four months. At this time it passes from parent’s milk to meat food and
tries to catch small animals (insects and lizards) itself, not being able to
fly at all. At the age of about 15 weeks the cub starts to study to fly, and
in three weeks it leaves mother and leads independent life.
The young flying wolf reaches maturity at the second year of life. Life expectancy
of this chiropteran reaches 30 – 35 years.
Swift-winged whiskered bat (Setostomops hirundipterus)
Order: Chiropters (Chiroptera)
Family: Molossid bats (Molossidae)
Habitat: South America, edges of tropical forests, woodlands.
Among mammals chiropters concede in variety only to rodents. At the boundary
of Holocene and Neocene the part of species of this group had died out because
of destruction of habitats – mainly because of destruction of tropical forests.
But in any case some species were numerous enough, and in epoch of biological
crisis the order remained various all the same.
In Neocene in plains and light forests of South America herds of herbivores
wander – tapirotheres and huge coursing rodents – deermara and giant
paca.
Herds involve plenty of blood-sucking insects – flies, mosquitoes and midges.
And rather large bat hunts for insects – it is an animal having wingspan of
up to 50 cm and weighs of about 200 grammes. All bats are remarkable by more
or less advanced outgrowths on muzzle, and also by ears frequently of strange
shape – these are adaptations for echolocation. At this bat strange appearance
is added with a plenty of rigid long bristles surrounding its muzzle. For this
feature it has received the generic name “whiskered bat”, and the shape of
wings is a reason of specific epithet “swift-winged”.
Wings of this little mammal are pointed; flight is fast and prompt with sharp
turns. Tail of swift-winged whiskered bat is long; the interfemoral membrane
includes it completely, but hind legs from knee and below it are free of membrane.
This bat eats large insects which gather around of representatives of megafauna.
For prety catching animals of this species have got the special adaptation:
around of their mouth resilient hair grow, increasing trapping surface. The
similar adaptation is present at basket-mouths and lustrer birds of Apodiformes
order, living in Central and Eastern Asia, and it represents an example of
convergence – at representatives of different classes from germs different
by origin organs similar by function are formed.
Muzzle of swift-winged whiskered bat is very freakish. The nose of animal is
short, and narrow nostrils are extended vertically and almost parallel to each
other; the top of nose is slightly below forehead. Edges of nostrils are mobile;
nostrils can open or close as narrow slot. Small eyes are directed forward
and are located at height of half of length of nostrils. Upper lip is splitted
by vertical cut, and its halves can move independently from each other. Edges
of upper lip hang down and are covered on edge with eresilient trapping hairs.
The same hairs grow on the bottom jaw. When animal opens mouth during hunting,
halves of upper lip shift in sides, and the bottom jaw moves downwards. Bristles,
sticking in sides, form a funnel directing the met insects right to the mouth.
During the feeding this bat utters echolocating signals with the help of nostrils.
Ears of this species are small. They are pressed to the head and are opened
as two slits closed on top of the head – tips of ears are densely pressed to
each other, and the forward line of ear adheres to head with the help of skin
membrane.
Body and head of animal are covered with bright rusty wool. Male differs from
female in larger size.
Swift-winged whiskered bat is crepuscular and nocturnal animal not competing
to diurnal insectivorous birds. In day time this bat hides in hollows. It forms
small congestions numbering up to 20 – 30 individuals. These groups have rather
constant structure – no more than 20 % of individuals changes for one year.
Seasonal prevalence in breeding of whiskered bat is not present. One cub is
born by female in any season, but more often it happens in autumn months, in
rain season. Young individuals become capable to breeding at the second year
of life. Up to this time their wool differs by colouring from wool of adult
individuals – it is darker and dimmer.
Megaardvark (Megaardwark armatus)
Order: Aardvarks (Tubulidentata)
Family: Aardvarks (Orycteropodidae)
Habitat: Zinj Land, light forests and tropical woods.
Having African origin, the order of aardvarks never differed in variety, and
distribution of these animals was always limited to area of warm climate. These
animals are specialized in feeding on insects though by biochemical parameters
they are close to ungulates.
When the East-African subcontinent, or Zinj Land had splited off from Africa,
among its inhabitants there were aardvarks (Orycteropus afer). These animals
could go through the time of biological crisis, because they lived not in woods,
but in savanna, and social insects which these animals ate, were kept in enough.
Gradually the climate changed, and the significant part of the area of Zinj
Land was occupied by tropical woods. The descendant of aardvark – the megaardvark,
very large insectivorous animal, inhabits these areas.
Megaardvark is a plantigrade animal moving on hind legs and resembling old-fashioned
reconstruction of any dinosaur. The body length of megaardvark including tail
makes up about 3 meters, and height in a waist is up to one and half meters.
The long tail of animal similar to tail of kangaroo, but thicker, counterbalances
forward part of body. When the animal eats to satiety, in tail the stock of
fat is accumulated. Features of locomotion of megaardvark are reflected in
structure of its skeleton. Sacral bone of this animal is large; some last lumbar
vertebrae and first tail vertebrae are fused with it to unite rigid structure.
Hind legs of megaardvark are very strong and brawny. If necessary the animal
is able to run, but speed of its run is only about 20 kms per hour. The massive
animal can not run long, and runs keeping such speed only about from hundred
to two hundred meters.
If the megaardvark is attacked by predator, the animal prefers to not seek
safety in flight and to protect itself with the help of its main weapon. Forepaws
of this animal are armed with constantly growing thick claws. At this one 3-rd
and 4-th fingers on which claws reach the length of 20 cm are especially strongly
advanced. Usually megaardvark walks, having pressed forepaws to chest. Occasionally
animal bases by claws on the ground, especially during the feeding. But the
main function of claws of megaardvark is not defense, but destruction of nests
of social insects, ants and termites. Besides megaardvark breaks by claws rotten
trunks of trees in searches of grubs, and digs out wood litter. In addition
to insects it eats spiders and even small vertebrates – frogs, lizards and
nestlings of birds nesting on the ground. The megaardvark is convergently similar
to huge stegoechidna from tropical forests of Meganesia, but does not eat the
vegetative forage.
Megaardvark is specialized to eating of insects and other small animals. It
has weak bottom jaw and very small mouth on the tip of long narrow snout. Teeth
are strongly reduced and presented only by three or four pairs in each jaw.
They represent cylindrical formations with thin layer of dentine and easily
erased soft pulp. The animal frays caught animals by them. Tongue of megaardvark
has very remarkable structure. It is very long – the tongue is able to extend
from the mouth more, than to half-meter. The basis of tongue is shifted far
back, and attached to brest, as at anteaters of Holocene epoch. Tongue consists
of elastic muscles, and inside it the thin sinew retracting the tongue in mouth
is stretched. The surface of tongue is covered by large knobs and small corneous
thorns between which glands emitting sticky secret are located. Such tongue
allows not only to extract insects, but even to pull small lizards and frogs
out from holes.
Insects are not safe forage. Many kinds of social insects, which are searched
by megaardvark, are armed sharp mandibles, and ants have also poisonous glands.
Poison of some ants eaten by megaardvark, can easily kill cat-sized animal.
For protection against insects nostrils of megaardvark can close with the help
of special ring muscles. Eyes are another place of this animal vulnerable for
attacks of insects. Eyes of megaardvark are small; they are protected from
casual penetration of insects by dense rigid eyelashes. At megaardvark there
is bad sight, but it does not prevent it to live normally. Sharp sight is not
so necessary for life in rich and shady tropical forest. This animal searches
for forage with the help of very keen sense of smell, and the hearing provides
it the additional information about the world around.
The wool of megaardvark is thin and rough, similar to pig bristle. The whole
body of animal is covered with thick and strong wrinkled skin which can not
be penetrated through by mandibles of insects. Besides on back of this sluggish
animal some rows of dermal ossifications (as at fossil ground sloths like Megatherium)
are developed. The predator, risked to attack megaardvark, risks to break teeth
biting to its back. The adult megaardvark practically does not have enemies.
This animal usually eats termites and ants, destroying their nests. This is
one of few animals of Zinj Land which is not afraid of vagrant ants. Except
for them, megaardvark digs out digging insects from wood litter and searches
for larvae in rotten wood. Megaardvark often feeds on carrion and rests of
prey of local predators. It can not tear off and swallow pieces of meat, but
all the same it finds a lot of edible things on the rests of carcass – megaardvark
eats maggots and grubs developing on it. High acidity of gastric juice makes
such diet of animal safe, protecting it from bacterial infections.
The ancestor of megaardvark was well-known for skill of hole digger. Megaardvark
had practically left this skill. For spending the night the animal digs to
itself non-deep hole in wood litter, usually near the fallen tree. These animals
are solitary ones meeting only for pairing. Seasonal prevalence in breeding
of megaardvark is not expressed. Pregnancy lasts till about 17 months. Once
in two years the female gives rise to one large cub. It has soft skin, and
dermal ossifications are poorly advanced. Therefore the cub of megaardvark
is very much vulnerable not only for predators, but even for vagrant ants quietly
eaten by adult animal. However it is born well advanced, and in some hours
after birth is able to follow mother. The female looks at it within approximately
7 months (at this time she pairs once again). The young megaardvark becomes
able to breeding at the age of about five years. Life expectancy of this animal
is more than 60 years.
Forest horned cony (Ceratohyrax prolongocornis)
Order: Hyraxes (Hyracoidea)
Family: Hyraxes (Hyracidae)
Habitat: Zinj Land, various kinds of plain and mountain forests.

Picture by Alexander Smyslov
In early Neocene, after extinction of significant part of
species of ungulates, representatives of earlier not numerous and uniform group
of hyraxes had got an opportunity to show their evolutionary potential. Among
them representatives of separate phylogenetic line of running hyraxes had appeared
– subfamiliy Dromohyracinae in Hyracidae family. These animals gradually passed
from burrowing habit of life to existence in plain district. After great split
of Africa the part of species of this group had remained on continent and evolved
to giant species. And the species isolated at the East-African subcontinent,
had changed much less. At Zinj Land representatives of this basal group of hyraxes
had remained.
In forests growing on plains and in foothills one of such species of running
hyraxes lives. Its distinctive feature is a presence of pair of short horn-like
outgrowths on head; because of this feature the animal has received the name
forest horned cony. This is a pig-sized ground mammal: it weighs about 50 kg
at body length up to 70 cm. Legs of animal are longer, rather than at common
hyraxes of Holocene epoch, and forest horned cony is able to run quickly. Hand
and foot are digitigrade. Despite of massive constitution, animal dexterously
jumps over high plank-buttress roots of tropical trees, escaping from chasing
of predators, and is able to change direction of run sharply. By habit of life
it resembles caviomorph rodents of South America like agouti or paca very much.
This animal lacks tail appreciable from outside.
Bones of upper part of skull of forest horned cony are thickened and form a
kind of helmet. On nose bridge of animal two conic horns directed upwards and
forward grow; these structures are covered with dense cornificated hairless
skin. Both males and females have them, and their application is various: at
males horns serve for courtship tournaments and establishment of domination
out of breeding season, and representatives of both genders also use horns for
digging holes and protection against enemies. The attacked horned cony, not
having an opportunity to escape in flight, attacks on predator, and strikes
impacts by head, ramming the aggressor. Small eyes are slightly shifted downwards
and protected by edges of bone “helmet”.
Colouring of wool is dark brown with white spots on sides of head and on stomach.
Male and female do not differ in colouring. The cub does not have white spots.
Forest horned cony lives in family groups consisting of large male, several
females of various ages and their cubs. Shelter for family of animals is the
hole about half meter wide and over ten meters long, directing under ground
to almost three-meter depth. In hole there is well equipped inhabited chamber
covered by vegetative material, some temporary toilet holes where animals leave
dung and urine, masking their presence, and 2 – 3 carefully disguised emergency
exits.
These animals do not have seasonal prevalence in breeding, and in family group
always there are cubs of various ages. Pregnancy lasts till about three months.
The female gives rise to 3 – 4 well advanced cubs born with opened eyes. They
become sexually mature at the age of about two years.
At Zinj Land close species of horned conies live:

Picture by Alexander Smyslov
Mountain horned cony (Ceratohyrax crassipygus) lives in mountain areas of Zinj Land, preferring dry bushy districts and thickets of grasses. It is larger, rather than forest horned cony, because it lives in cooler climate with expressed daily and seasonal fluctuations of temperatures. At cold snap this species runs to not deep dormation. In warm season when food is plentiful, on buttocks of mountain horned cony thick layer of fat is accumulated; it is used in winter time. Colouring of wool at this animal is lighter, than at forest species – straw-coloured with grayish shade on back, hips and waist. Horns on skull are thicker and shorter, than at forest species. With their help animal can pick out from the ground stones during the hole digging. This species settles in pairs, but does not avoid presence of congeners.

Picture by Alexander Smyslov
Dwarf horned cony (Ceratohyrax minimus) inhabits high-mountainous meadows of Zinj Land. It is a marmot-sized species weighting no more than 6 kgs. It has short horn-looking outgrowths of skull, and rich wool has dark, almost black color without marks both at adult individuals, and at cubs. This animal is able to climb and jump on rocks, searching for grassy plants. In summer dwarf horned cony also accumulates fat which is depositing on buttocks and hips. In conditions of high mountains at downturn of temperature it runs into deep dormation which lasts not less than 3 months. At this time animal uses the saved up fat stocks.
Sluggish ventrohyrax, “Zinj sloth” (Ventrohyrax bradipus)
Order: Hyraxes (Hyracoidea)
Family: Ashkokos, or Climbing Damans (Ascendohyracidae)
Habitat: tropical woods of Zinj Land.

Picture by Timothy Donald Morris
In epochs of climatic changes some types of vegetative communities
may completely disappear, dooming to extinction the significant part of species
connected
to them. But at restoration of acceptable conditions of inhabiting settling
of again formed community can proceed by two ways. In one case it is occupied
with descendants of relic species had escaped in few “refuges” where the
former vegetation had kept. In other cases new habitats are accustomed by the
species
earlier not dwelt here. It had taken place, for example, at the East African
subcontinent (Zinj Land) where in tropical rainforests the original analogue
of sloth descending from one local group of animals had appeared.
For Zinj Land the special family of climbing damans, not meeting in continental
Africa, is typical. Its characteristic representative is ashkoko (Ashkoko
sylvaticus) - animal similar by constitution to loris - Asian prosimians
of Holocene epoch.
But this animal is not so specialized to tree-climbing habit of life, as
ventrohyrax – another animal living in the neighbourhood.
The name “ventrohyrax” means “a stomach of daman” and emphasizes characteristic
position of this animal – upwards a stomach. Ventrohyrax hangs under the
branch similarly to South-American sloths, for what it had received another
name –
“Zinj sloth”.
In connection to unusual way of life in anatomy of ventrohyrax there is a
number of interesting features. Paws of animal are adapted to clinging to
branches
– they are rather long, and the structure of hand and foot differs in prominent
features. Hand and foot of animal are lengthened (their length is about one
third of general length of the extremity), along their bottom side strong
sinews stretch, which are attached to strong muscles. For attaching of muscles
on
paws bones of ventrohyrax crests giving to bones fantastical appearance are
advanced. Fingers of ventrohyrax are very long and tenacious – animal clings
by them to branches, because claws became very small even at damans – far
ancestors of these animals. On forepaw two fingers are kept (only 2-nd and
3-rd, and
other ones, 1-st and 4-th fingers are reduced and do not touch to branch),
on hinder leg all three toes are kept. Fingers and toes are bent like hooks;
skin on palms and feet is naked, covered with cross plicas and callouses.
Due to this adaptation animal easily moves even on prickly branches. At grabbing
of branch muscles automatically contract, and very strong capture is formed:
even the died animal remains hanging on branch, yet will the decomposition
of corpse begins. Muscles of extremities of ventrohyrax have unusual dark
red
color: they contain many myoglobin – the substance accumulating oxygen, therefore
they can work any time in “automatic mode”.
Ventrohyrax spends all life among branches, and extremely seldom goes down
to the ground. On the ground this animal is very clumsy: ventrohyrax bases
on back surface of hooked fingers of forepaws and external lateral surface
of foot (ends of toes are turned under the body). The animal walks on the
ground slowly, and as soon as possible tries to find a tree suitable to life
to appear
again in world habitual for itself.
The neck of ventrohyrax is very mobile; due to it animal can eat leaves around
of itself, staying at one place. Muzzle of animal is short and wide; molars
are tuberculous with wide masticatory surface. Chewing muscles of animal
are well advanced: in case of necessity ventrohyrax can bite out small branches.
Ventrohyrax had kept features of dental system characteristic for the daman:
in each jaw it has two constantly growing self-sharpening incisors.
Ventrohyrax is covered with shaggy wool – though it lives in tropical climate,
it may easily catch a cold. In equatorial area each day rain may fall, and
only foliage of trees protects ventrohyrax from it. And in wood canopy strong
winds blow frequently. The wool of ventrohyrax is colored brown with darker
separate locks, precisely imitating the pattern of wood bark. The wool of
animal serves as fine masking. Only it and powerful impacts by claws are
its unique
protection against predators. Defending itself, animal seizes branch by hinder
legs and defends by both front paws.
Though ventrohyrax spends all life on branches in tropical forest canopy,
this animal does not make risky and fascinating jumps from tree to tree,
and in
general it differs in slowness. At such habit of life three-dimensional sight,
characteristic for damans, loses the value, but the wide field of view gets
much more value for life. Therefore eyes of ventrohyrax look more in sides,
than forward.
Ventrohyrax leads rather simple way of life and consequently does not differ
in high mental faculties. Its brain is too small, and brain department of
skull is lengthened and low.
Though the forest canopy is very productive part of tropical wood, here there
is very rigid competition for food resources. In forest canopy a lot of different
mammals, consuming various kinds of forage lives. The close relative of ventrohyrax,
ashkoko, is omnivorous animal, and other climbing animal, sun
flying fox of chiropters order, eats mainly fruits. Ventrohyrax does
not compete to them due to the diet: this animal eats exclusively leaves.
Being a sluggish animal,
not needing for long jumps and fast climbing, and having no big expenses
of energy, ventrohyrax eats poorly nutritious and hardly digestive leaves
of plants.
In such way it avoids a competition for food with other animals. The stomach
of this animal is complex and consists of several chambers. In the most voluminous
first chamber there is a food fermentation, in which symbiotic bacteria and
protozoans help ventrohyrax. Ventrohyrax easily digests even dense leaves
of figs, rich in rubber.
This species is social; it keeps in tree crones in groups of one male and
several adult females with cubs. The structure of these groups is changeable,
and animals
are not connected with strong ties of attachment (except for female and growing
up cubs). Frequently groups of animals, travelling in forest, unite or break
up. Also there are single males joining groups only for pairing.
Two times per one year the female of ventrohyrax gives rise to one cub. It
is well developed, covered with wool, with opened eyes. Color of wool at
the cub is darker than at female. Till first days of life the cub clings
to mother
and eats only her milk. Starting approximately from the week age cub of ventrohyrax
starts to try vegetative food. At first the female feeds it with slurry of
belched and semidigested plants. Young animal receives so necessary gastric
microflora. Gradually the cub passes to feed in vegetative forage.
Young ventrohyrax becomes sexual matured approximately at the fourth year
of life. Life expectancy of this animal reaches 30 – 35 years.
Embolohyrax (Embolohyrax brontotherioides)
Order: Hyraxes (Hyracoidea)
Family: Flathorns (Platyceratheriidae)
Habitat: the north of Africa, Atlas Mountains, Western Europe to the north
from the Alpes.
In human epoch large species of mammals got serious harm because
of human activity. Hunting and destruction of habitats had resulted in great
reduction of their number and variety. The part of large species had died out
even within people’s memory, and other species had become extinct soon after
people extinction – their small and dispersed at the former area populations
appeared invitable in conditions of the changing world meeting a new wave of
congelation. In new landscapes of early Neocene the place of missed giants was
occupied by new animals. Frequently they are descendants of ones grazed gingerly
in shadow of Holocene giants left to the past. Hyraxes were especially successful
from this point of view. Their descendants had quickly increased in size and
had turned to prevailing group of large herbivores in tropics of Old World.
The north of Africa, southwest of Europe, Near East and Hindustan became habitats
of large species of this group.
To the north of savannas of Sahara, at the Gibraltar Isthmus and in Southern
Europe at the edges of salt swamps and deserts of Mediterranean lowland one
species of these animals, huge embolohyrax, lives. It is the close relative
of flathorn (Platyceratherium
foetidus) from savannas of Africa. Embolohyrax looks like rhinoceros, but
has very large size: length of its body is over 4 meters, and growth at a shoulder
is up to 170 cm. Weight of such giant may reach 8 – 9 tons. Constitution of
this species is massive; limbs are digitigrade, with elastic pad of fatty tissue
under fingers and toes. Front and rear limbs are three-fingered (three-toed),
and on fingers/toes thick hooves grow. Embolohyrax lives in dry places, but
if necessary it easily moves in marshes and is able to swim. This species is
remarkable with its great ecological plasticity – embolohyraxes well feel like
as in savanna and maquis the Western Europe, as in deciduous forests of the
Central Europe and foothills of the Alpes. They equally well consume rigid bushes
forming maquis, sapful foliage of undersized trees in hillsides in the Alpes
and marsh grasses at water meadows of Central Europe.
This animal has bad sight, but very good hearing and keen sense of smell. Nostrils
of embolohyrax are wide and supplied with skin valves. When animal gets in sandstorm
or dives, valves close nostrils from sand or water. Wool of embolohyrax is thin
and short, and skin of grey color reaches thickness in 4 – 5 cm, especially
on shoulders and back of animal.
 Like
all flathorn relatives, embolohyrax has massive bone horn and very large head.
Horn represents the massive bone outgrowth covered with corneous layer, and
it stretches along the whole skull. In horn shape at this species the precise
sexual dimorphism is expressed. At females horn represents a low bone thickening,
the platen stretching along skull. Horn of female is covered with dense skin,
and in its forward part, above a nose, there is a small expansion in horizontal
plane. Male horn is much more massive and thicker. It is sharply expanded and
curved upwards in forward part, from the level of eye-sockets. The forward surface
of horn is covered with thick cornificated skin and represents the shock weapon
– hence the name meaning “ram hyrax”. The bone tissue of horn is well adapted
to endure significant loadings directed front to back; the basis of horn rests
against well advanced cross occipital crest. At the end of summer the courtship
season begins at embolohyraxes. At this time embolohyrax males establish hierarchical
relations, butting each other by horn – it does not cause traumas to them. If
struggle does not reveal the strongest one, or the contender behaves too arrogantly,
males pass to more aggressive way of relations establishment, and butt each
other’s sides by horn. As a result after courtship season males frequently get
broken ribs, and at “battle veterans” the most part of ribs has traces of repaired
traumas.
Like
all flathorn relatives, embolohyrax has massive bone horn and very large head.
Horn represents the massive bone outgrowth covered with corneous layer, and
it stretches along the whole skull. In horn shape at this species the precise
sexual dimorphism is expressed. At females horn represents a low bone thickening,
the platen stretching along skull. Horn of female is covered with dense skin,
and in its forward part, above a nose, there is a small expansion in horizontal
plane. Male horn is much more massive and thicker. It is sharply expanded and
curved upwards in forward part, from the level of eye-sockets. The forward surface
of horn is covered with thick cornificated skin and represents the shock weapon
– hence the name meaning “ram hyrax”. The bone tissue of horn is well adapted
to endure significant loadings directed front to back; the basis of horn rests
against well advanced cross occipital crest. At the end of summer the courtship
season begins at embolohyraxes. At this time embolohyrax males establish hierarchical
relations, butting each other by horn – it does not cause traumas to them. If
struggle does not reveal the strongest one, or the contender behaves too arrogantly,
males pass to more aggressive way of relations establishment, and butt each
other’s sides by horn. As a result after courtship season males frequently get
broken ribs, and at “battle veterans” the most part of ribs has traces of repaired
traumas.
On back of embolohyrax there is large musky gland closed by skin valve. In courtship
season it secrets oily liquid with sharp musky smell. Smells play the important
role in communication and behaviour of these animals. The smell of female secretions
raises male aggression, and male secretions serve for marking of territory and
females. Leaving odorous marks, male rubs back and waist against stones and
tree trunks. Usually mature males lead solitary life, and females gather in
herds numbering up to 20 animals. In areas rich in forage males join herds of
females and beyond courtship season are very tolerant to each other.
Pregnancy lasts till about 20 months. The cub is born in spring when female
has opportunity to feed on vitamin-rich plants. At birth cub weighs about 200
kgs and is covered with appreciablly richer wool, rather than adult animals.
It sucks mother’s milk up to two-year-old age, but right at the first autumn
of life it tries vegetative food. Sexual maturity at young animal comes at the
age of 4 years, and life expectancy reaches 60 years.
Xenondipina (Xenondipina agilis)
Order: Hyraxes (Hyracoidea)
Family: Ndipinotheriums (Ndipinotheriidae)
Habitat: marsh forests of Central Africa.

Picture by Tim Morris
In Neocene in territory of Africa to the south of Sahara
evolution of hyraxes proceeded actively. These animals have considerably increased
in size and have
got various specializations. Among descendants of hyraxes animals of ndipinotheres
family are remarkable – these are large herbivores, similar to gorilla in
constitution. Large ndipinotheriums inhabit mosaic landscape where sites of
forest alternate
with open area. And xenondipina, the relict of early stage of family evolution
lives in continuous tropical forests of Central Africa. As against to typical ndipinotheriums,
it is not so large animal capable even to swarm up trees. Body length of
xenondipina is about 2 meters. By its constitution it is similar
to gorilla – front limbs are elongated, back is appreciablly inclined back,
tail is lack absolutely. Hind legs of xenondipina are plantigrade and have
three large claws. Forepaws are digitigrade; animal supports on 3-rd and
4-th fingers having little peaked hooves. Mobile 2-nd and 5-th fingers help
animal
to keep food. This is sluggish, but mobile animal – adult xenondipina is
capable to swarm up trees and eats foliage, and if necessary it swims well
enough and
walks on fenny ground.
Head of this species is lengthened – xenondipina eats soft sapful vegetation
for which chewing significant efforts are not required. Animal has rounded
mobile ears, larger than ears of ndipinotheriums. Xenondipina has keen hearing
and sense of smell, but its sight is bad. In rich forest it is difficult
to be guided with the help of sight, and animal is helped more by other senses.
Body colouring of xenondipina is cross-striped: on brown background on shoulders,
back and crupper close and thin vertical strips of white color stretch, forming
slightly wrong striped pattern imitating spots of light and shadow on tree
trunk. On throat of animal there is a white spot which helps animals to distinguish
each other.
Xenondipina avoids competition to ground monkeys due to life in swampy habitats
with set of fenlands and rivers – this species is especially numerous in
Congo basin. Xenondipina eats foliage of trees, and frequently gets on trees
to the
height up to 10 meters. Also it supplements diet with marsh vegetation and
digs out tubers of perennial grasses by forepaws. Frequently xenondipina
goes for feeding to glades formed by fallen trees, and eats leaves of large
grassy
plants appearing at initial stage of forest restoration.
This species eats rough vegetative forage from which it is difficult to take
nutrients. Stomach of xenondipina is specialized and two-chambered. Its first
section is “fermentive chamber” where symbiotic protozoans and the bacteria,
providing initial stages of cellulose digesting, live. In second, ferruterous
section of stomach food is processed by digestive juice and the further digestion
proceeds in long intestines.
Xenondipina keeps in herds numbering up to 20 individuals – adults males,
females and their posterity about the second year of life. Between males
the certain
hierarchy is established, and dominant male has the right of priority in
pairing. But subordinate males also have an opportunity to leave posterity.
After 5-month
long pregnancy female gives rise to well advanced cub which in one hour after
birth is capable to walk, and at monthly age does not concede adult animal
in dexterity of tree-climbing. At the second year of life young animals leave
parental herd and join groups of animals of the same age, living at the edge
of territory of herds of adult individuals. At three-year age xenondipina
becomes sexually mature, and life expectancy of this animal makes till 30
years.
Sea
ipopo (Behemohyrax littoralis)
Order: Hyraxes (Hyracoidea)
Family: Ndipinotheriums (Ndipinotheriidae)
Habitat: Africa, Zinj Land (ocean coast), coast of Tanganyica passage.

Picture by Alexander Smyslov
In Holocene sea habitats were populated by mammals of three
orders: cetaceans, Carnivores (pinnipeds represent not uniform group, but descendants
of several branches of land predators) and sirens. First two groups were submitted
by carnivorous and planktophagous forms and sirens were sea herbivores. In Cenozoic
it also had exist the order of sea ungulates Desmostylia, but it had died out
long before the people occurrence. Hunting and reduction of places of inhabiting,
caused by human activity, and also climatic changes and “plankton accident”
at the boundary of Holocene and Neocene had resulted in complete disappearance
of the majority of representatives of these orders. Only separate relic species
of “old” orders had remained. But in Neocene mammals had made the second attempt
to develop sea habitats – various groups of mammals in different areas of Earth
passed to life in sea independently from each other. So, in Europe the flattooth,
the species of semi-aquatic boars had appeared. Sirens led the origin from African
mammals close to proboscids, and in Neocene the similar situation had repeated
again in Africa. Descendants of damans, also related to proboscids, are one
of succeeding Neocenic mammals. And among them animals leading semi-aquatic
habit of life had appeared.
At coast of Indian Ocean one of representatives of water herbivorous mammals
lives. This large animal is the descendant of damans by anatomy, but by ecology
it represents the analogue of fossil mammal Desmostylus. It browses in mangrove
thickets and at sea shallows, eating leaves of mangrove trees, seaweed and sea
grasses. Systematical position of this animal is between flathorns
and ndipinotheriums,
but by features of anatomy this mammal is closer to the last ones. By size this
four-footed animal is larger than rhinoceros, and is externally a little similar
to hippopotamus. Because of it there is its name – sea ipopo (“ipopo” is the
form of word “hippopotamus” distorted in African languages).
Sea ipopos meet in areas of warm climate, and their distribution in many respects
coincides with area of mangrove woods growth. The body of ipopo is covered with
short wool, and under skin the layer of fat improving buoyancy is advanced.
Tail is absolutely not present at this animal. The wool on body is light grey
– it makes the swimming ipopo less appreciable on the background of sea water.
On forehead and nose bridge the black wool grows. At swimming animal they are
above water, and do not give out this animal to underwater predators. The amount
of black wool depends on hormonal level of animal, and it is possible to determine
approximately the status of each individual in group by size of black spot -
at dominants black “cap” is largest.
Legs of ipopo are rather long (near to it the flathorn looks stumpy) but this
animal can not rise on hinder legs (like ndipinotheriums), and moves exclusively
on four extremities. Forward legs are little bit longer than back ones, and
back is sloping. Feet are wide, adapted to walk on fenny dirt and to row at
swimming. Hoofs on toes are flattened, similar to elephant’s ones; under foot
the fat pillow softening steps and increasing the area of support develops.
Due to such structure of legs ipopos easily browse in mangrove thickets where
any predator can not reach. Ipopo swims very well – it lives at all small islands
near coasts of Africa and Zinj Land, and separate animals even regularly reach
Madagascar though a stable population of ipopos had not appeared on this island.

Picture by Alexander Smyslov
Head of animal is large, rounded, with short jaws. Eyes are
located in forward part of skull. Neck at ipopo is mobile – animal easily feed
with branches of mangrove trees. Ears are rounded, mobile and short. The upper
lip of ipopo is adapted for browsing of parts of plants: it is flexible and
represents small proboscis, as at rhinoceros. The end of lip is slightly forked,
permitting this animal to break even thin stalks dexterously. Simultaneously
teeth of animal enable it to eat food of other kind – containing the big amount
of mineral substances. Incisors in bottom jaw of ipopo are wide, scraping by
type. They have deep roots and constantly accrue. Sea ipopo frequently leaves
mangrove woods and goes to shallows, where eats seaweed, sea grasses and sedentary
animals (in this habit it is the convergention with the European flattooth).
Due to universality these animals can live where mangrove trees do not grow
– at the rocky shallows overgrown with seaweed.
On back of ipopo there is the specific gland – it is the heritage remained from
damans. With its help animals submit each other chemical signals, and dominants
mark territory of herd. In mangrove woods they rub backs against low thick branches
of trees, and among rocks they prefer to mark stones forming the similarity
of eaves under which it is possible to stand.
Sea ipopos wander in herds on coasts, preferring the territories overgrown with
mangrove trees. On rocky sites they also live, but each group has extensive
territory – some times larger, than in mangrove wood. In herd of ipopos strict
matriarchy dominates. Males of this species live one by one or in small groups
in boundary territories of female herds. At bare coasts males live one by one
and follow herds of females.
Pairing at ipopos occurs at any time of year. Pregnancy lasts about 15 months.
In litter there is always one cub. But it is very well advanced, and rises on
legs in first some minutes after birth. It is born on the coast; during the
cub birth the female is surrounded by neighbours protecting her from possible
enemies – at this time they rush even to sea birds. When the cub rises on legs,
the female licks it, and some time does not let neighbours to it. After between
the female and cub connection was established, other neighbours sniff newborn
cub. They as though “accept” it so to the clan, and if necessary protect it
in common. Therefore the survival rate of posterity at ipopo is high. But the
cub depends on mother for a long time – it leaves her only at the third year
of life and then she begins able to bear posterity again. For all life at the
ipopo female it may be 4 – 5 cubs. The young animal becomes able to breed at
the age of 6 – 7 years.
This species has more primitive relative, reed
ipopo (Behemohyrax calamophilus) eating riverbank vegetation in rivers
of Africa. It lives in marshland at riverbanks. The basis of food of reed ipopo
is made with papyrus and other huge grasses. This is sluggish peaceful animal
little bit smaller by size then its sea relative. The skin of ipopo has striped
colouring – on light brown background there are vertical dark strips. Due to
such colouring animal easily masks in thickets. Tip of muzzle at animals of
both genders is white.

Picture by Alexander Smyslov
The role of reed ipopo in ecosystems of African rivers is
great – these giants, eating papyrus and other plants, preserve rivers from
overgrowing. On lighted
by them sites of thickets of papyrus soon larger sprouts of this plant appear.
Reed ipopo digs out plants from a bottom of river by strong forepaws for
what the claw on internal finger of his forepaw is especially strongly advanced.
By this claw reed ipopo “ploughs” river bottom, and then eats roots, tubers
and sappy stalks of water plants emerged on water surface. As well as the
sea
neighbour, this is the social animal. But this animal lives in families in
which there is a male. Male and main female mark territory by musk secretions
of back gland.
Possible, the ancestor of these species lived in rivers and lakes, and then
had settled down the current of rivers at first to deltas, and then to the
sea coast, having the respective alterations in habits and diet.
Fast hyracolopa (Hyracolopa velocipes)
Order: Hyraxes (Hyracoidea)
Family: Running damans (Dromohyracidae)
Habitat: savannas of Northern Africa.
Picture by Tim Morris
Unspecialized species of live creatures are one of main reserves
of evolution during mass extinctions. Absence of dependence on any limited
resources and
the ability to live in various conditions of environment are main “secret
weapons” of such species. And anatomical primitiveness, characteristic for
similar animals,
comprises a potential opportunity for realization of various specializations
in aspect of anatomy.
Among African animals damans (Hyracomorpha) were such group: they had represented
the order of rather ancient and rather primitive hoofed mammals. In Neocene,
after mass extinction, they had formed within the limits of order some groups,
differing by habit of life. One group is presented by massive ground tetrapods
– flathorns from Northern Africa. The second group specialized to tree-climbing
way of life and became analogues of lower primates like lories and lemurs.
It is ashkoko (Ashkoko sylvaticus) from Zinj Land, or the East-African subcontinent.
Except for them, in mountains of Central Africa, savannas of Northern Africa
and mountains of Southern Europe representatives of the third branch of damans
– running antelope-like forms, hyracolopas, live. They occupy an ecological
niche of small and medium-sized antelopes, partly competing with harelopes
of northern origin.
By constitution hyracolopas are similar to typical antelopes, but in closer
examination they have some obviously differences. Tail at hyracolopas is
not present: it was reduced at their ancestors, damans. Rear legs of these
animals
are little bit longer than front legs because of what the back of hyracolopas
is slightly inclined forward. This is the heritage of their past way of life:
obviously, hyracolopas evolved in mountains of Central or Eastern Africa,
and such structure of body is typical for animals grazing on hillsides. The
structure
of legs uindicates these animals are rock-climbers and runners. Extremities
of damans were plantigrade, and foot of rear leg had formed similarity of
sucker. At hyracolopas such adaptation had disappeared: on back side of rear
leg there
is only a callous, permitting to sit on stones. Claws had increased and became
similar to small hoofs. As against horses and antelopes, hyracolopa supports
not only on hoof-like claw, but also on the subungius covered with thick
cornificate skin. Lateral toes became small, and touch the ground at movement
only a little.
On forward leg only two fingers from four, characteristic for daman, remained:
second and larger third ones.
Head of hyracolopas is lengthened and low, eye-sockets are shifted in forward
part of skull. Brain cavity is low and long, that clearly indicates on –
alas! – low intellectual qualities of these animals. But at top of skull
small sagittal
crest stretches, to which strong chewing muscles attach. In dental system
of hyracolopas features, characteristic for ancestors are kept: gnawing type
of
incisors. The bottom jaw is short and a little bit “lengthened” by two large
incisors. With their help hyracolopas can dig out bunches of grass with roots.
If necessary incisors turn to fighting weapon: at the attack of small predators
hyracolopas can bite strongly.
At hyracolopas there are large eyes providing good sight. At mountain species
eyes look forward, rather than in sides: so the three-dimensional sight,
which helps to estimate distance before jump, is provided to an animal. Hearing
at
hyracolopas is good; ears are big, rounded and mobile. Sense of smell is
advanced well: nasal cavity is short, but high. Smells play the significant
role in
life of these animals: on back of hyracolopas there is the specific gland,
producing an odorous secret. With its help animals mark territory: for this
purpose the hyracolopa rears on back legs, nestles by back to the stone or
tree, and rubs against it.
Hyracolopas live in small herds numbering up to 15 - 30 animals. In herd
there is no strictly expressed hierarchy, and its structure may strongly
vary in
due course: the herd may break up, or two herds mix up at one pasture and
any time graze together. For night hyracolopas hide in bushes.
All species of this group of herbivores eat grassy plants, but can eat young
branches of trees and bushes. The stomach of hyracolopas is complex, three-chambered
and similar to stomach of ruminant animals. In forward chamber of stomach
the colony of symbiotic protozoans, similar to those at ruminant animals,
develops.
They help animal to digest rough cellulose. The blind gut is poorly advanced.
All hyracolopas have kept a habit, characteristic for damans: animals leave
dung always in the same place. Such behavior has the special role in their
life: by smell of dung animals learn about the congeners, not seeing them
at all. Some animals, living in one group, leave dung in same place.
Fast hyracolopa leads atypical for this genus way of life: it prefers plain
habitats, living in savannas of Northern Africa. This animal reaches height
about 60 cm at a shoulder. Wool of this animal is colored grayish-beige color
with narrow and dense cross strips on body. On all legs there are white “stockings”.
Head of fast hyracolopa is colored brighter, than body: through eyes and
forehead black “band” stretches, which ends wash out on neck; tip of muzzle
is white.
Such contrast colouring of head helps to warn congeners of danger: the disturbed
animal highly lifts head, and even rears, looking over district. And colouring
of its head becomes appreciable from afar, serving as an alarm signal for
neighbours.
Due to strong rear legs fast hyracolopa can make long jumps, escaping from
the enemy: up to six meters in length and two meters in height. Hoof-like
claws are wide; therefore animal can walk on fenny ground and swims rather
good.
Frequently hyracolopas of this species escape from predators in water or
in swamp.
The courtship season at all hyracolopas passes roughly: males gather small
harems of females, jealously preserving them from each other. Threatening
the opponent, they growl and show incisors, having opened mouth widely. If
contenders
do not concede each other, they can even fight, biting each other in back
and nape, and striking impacts by forward legs.
Once a year at the female well advanced independent twin cubs are born. They
are able to rise of legs at the first hour of life, and the daily cub does
not lag behind mother. The young growth is colored not so bright, as adults:
cubs don’t have “mask” and white sites on muzzle. They appear only at the
age of about one year. At one and half year age young females become able
to breeding.
Males grow longer, and mature only up to age of two years.
Other species of hyracolopas live in mountains, and their area is limited
to one or several ranges:
Rift hyracolopa (Hyracolopa riftica) lives
in mountain woods of Central Africa, where it is the analogue of small antelopes
like the duiker. Its growth at
a shoulder reaches approximately 40 cm. Wool of this hyracolopa is colored
dark brown, almost black color, and only tip of muzzle, throat and chest
are white. In mountains where it lives, the weather may be rather cool, and
ears
of rift hyracolopa are covered from external side with short wool. This animal
eats various kinds of vegetation: grass, ferns, tubers and bulbs of plants.
It meets in herds numbering up to fifty adult animals.
Wooly-eared hyracolopa (Hyracolopa trichotis) lives
even higher, than rift hyracolopa: on open hillsides with poor vegetation.
This animal differs in
dexterity and quickness: it can clamber on almost steep slopes. The wool
of this species is colored brown, and only on throat of the adult male white
wool
grows. On back in the area of specific gland long wool grows. For protection
against cold ears of this species are much shorter, than at relative species,
and are covered with long wool from external side. This animal gathers in
small groups and carefully marks borders of territory: fodder resources in
mountains
are poor, and the herd should have rather extensive territory.
Atlas hyracolopa (Hyracolopa atlassica) is
the species of savannas and foothills of Atlas Mountains, inhabiting also
rocky areas of south-west of Europe. It
partly competes with flatland harelopes, eating rigid plants. The body is
colored yellow, on back there is black longitudinal strip. On rear legs,
and at separate
individuals even on front ones, there are black cross strips on white background.
Cheeks and throat are white.
Persian hyracolopa (Hyracolopa persica) inhabits
mountains of Near East formed instead of Persian Gulf as a result of movement
of Arabian lithospheric plate.
The area of this species is partly crossed with area of rockloper (Lepotragus
nivalis) from the order of hoofed lagomorphs (Ungulagomorpha). But both species
do not compete with each other because of life in different habitats. Rockloper
prefers dry slopes of mountains, and Persian hyracolopa prefers damper valleys
overgrown with bushes. It frequently lives on hillsides overgrown by wood.
This species lives in herds numbering 10 – 15 individuals. Colouring of Persian
hyracolopa is spotty: general colouring is grey, on sides and groats small
black spots are scattered. Forehead and back part of neck are also black.
On internal side of forward and rear legs there are sites of white wool.
Alpine hyracolopa (Hyracolopa alpina) inhabits
mountains of Southern Europe from Pyrenees to Balkan Mountains along the
northern edge of Mediterranean
swamps. It is the largest species of genus, reaching the size of domestic
goat. It differs from other relatives in long wool with rich underfur that
permits
this animal to live in high mountains. General colouring of this animal is
grey with black longitudinal strip on back and black spots on groats; legs
are dark.
Tapiroid
(Tapirocephalus velox)
Order: Even-toed ungulates (Artiodactyla)
Family: Trunk boars (Proboscichoeridae)
Habitat: bush thrickets of Southern Europe.

Picture by Tim Morris
Picture by Pavel Volkov - the initial image |
During global changes in ecosystems unspecialized species able to live in various
conditions and to use for food plenty of forage kinds survive basically. During
change of Holocene by Neocene among few survived hoofed mammals pigs were such
universals and champions in survival. After ice age and extinction of big number
of European hoofed mammals pigs were among animals successfully existed in narrow
zone of vegetation clutched between glacier of kilometer thickness and heated
salty desert of Mediterranean. When glacier had receded pigs among the first
wave of migrants had started to develop free territories and later migrants
from Asia and Africa were compelled to compete with specialized descendants
of wild boar (Sus scrofa).
Boar family had becomed prospering group: among them special family of trunk
boars has appeared at which representatives muzzle had turned to short proboscis.
This adaptation has given appreciable advantages to its owners having expanded
opportunities of food getting. In family even such species started to develop
sea coasts had appeared – it is huge flattooth
(Belonoporcus dolichocephalus).
As against to massive flattooth tapiroid is terrestrial animal. Length of body
of adult animal is about 2 meters, height at a shoulder is 1.7 m. The constitution
indicates fast runner in it: at this animal there are muscled body and long
legs. It can accelerate momentum up to 40 kms per hour at the long distance
and at short time accelerate up to 60 kms per hour. On every leg of tapiroid
there are two large hoofs: characteristic for pigs hoofs on 2-nd and 5-th toes
are underdeveloped and sometimes are absent at all. In appearance of tapiroid
there is a feature making it similar to South-American tapir: massive head with
long proboscis. With the help of flexible proboscis tapiroid can browse leaves
and young branches of bushes containing basic part of its ration. Neck of tapiroid
is rather short but due to proboscis it does not have inconveniences in daily
life. Body of animal is covered with rigid thin wool and on back skin is very
thick – it is its protection against predators. On tips of long ears there are
hairy brushes raising sensory acuity. Eyes at tapiroid are small and sight is
rather weak. But sense of smell at it is very keen: due to presence of proboscis
the olfactory surface of nasal cavity is strongly increased.
Wool of tapiroid is colored brownish-yellow with dark longitudinal strips on
back (such colouring is characteristic for boar piglets). Due to inhabiting
in bush thickets this juvenile attribute became very useful to survival.
In mouth of tapiroid sharp tusks directed forward and in sides grow. At males
they reach significant size. Males of this species apply tusks to duels, establishing
hierarchy or combating for female. During fight opponents simply cling each
other by one tusk and try to tumble down the contender by movement of head and
body pressure. Dangerous wounds this way are completely excluded though at old
males tusks sometimes break. At the predator attack animal uses tusks as thrust
weapon causing to predator deep wounds similar to dabber ones.
Tapiroids live in family groups of 15 - 20 uneven-age animals (young growth
of the current year makes up about half of group) under the leading of large
male. Each group lives in the certain territory on which goes adhering about
constant routes. During movement herd treads and eats young growth of bushes,
therefore in habitat of these animals through continuous bush thickets wide
tracks pass. And the more herd living in certain territory, the wider tracks
left by it are. In tracks graminoids grow basically changing by othergrasses
at the edge of tracks. It gives food to many other bush inhabitants.
Tapiroids eat leaves and young sprouts of bushes, soft grasses and mushrooms.
Because of short neck they can not graze on grass and dig out roots and tubers
as their ancestors, but in bushes they find a lot of suitable forage.
At the watering place animals are cautious: they can reach water only having
bent forward legs, therefore any part of family group stands on the alert while
other animals drink water.
Cubs of tapiroids are born in spring when it is a lot of forage and water. Female
gives rise to two - three cubs covered with short wool. By colouring young growth
is similar to adult animals, colouring is only brighter appreciably and more
contrast. Cub rising occurs almost simultaneously at all females of family group.
Cubs spend first days in shelter among bushes, yet they will get stronger enough
to move after adult animals. On 3-rd - 4-th day they abandon shelter and herd
continues movement.
Females can already rise posterity at two-year-old age, and males though become
mature almost simultaneously with females, receive chance to pairing only at
the age of four years having developed to full might. Life expectancy at this
species is about 25 years.
Nozdrokh
(Probosciaper mammutoides)
Order: Even-toed ungulates (Artiodactyla)
Family: Trunk boars (Proboscichoeridae)
Habitat: woods of Central and Eastern Europe up to Ural.

Picture by Leonard Popov
Initial image - pictured by Pavel Volkov |
In Russian annals by the word “nozdrokh” any large animal,
most likely mammoth meant, whose frozen carcasses occasionally had been founded
by people, derivating every possible legends. Therefore the large animal of
Neocene epoch, rather reminiscent by the constitution proboscides missed a long
time ago, is named so.
The extinction of large animals caused by chasing from the side of people, destruction
of places of inhabiting and natural accidents, has released a way for evolution
of groups of animals, not numerous and not variable before it, having allowed
them to realize to the full the evolutionary potential. Among such “lucky beggars”
of Neocene epoch non-ruminant artiodactyl mammals appeared. In North America
dominant large animals peccasons
from peccary family (Tayassuidae) appeared. But in Old World pigs (Suidae) have
reached of the big success: among them ecological analogues of such various
hoofed mammals, as horses, bulls, hippopotamuses, tapirs, and even proboscides
have appeared.
Nozdrokh, the inhabitant of woods of Europe, is a huge descendant of the wild
boar, whose growth at a shoulder reaches 2 meters. It differs in heavy addition
and thick legs. Head of nozdrokh is rather big. Well-known tusks, the main weapon
of boar, at this species had started to increase, resembling tusks of mammoth:
they are bent upwards by half rings. It is convenient to rake by such tusks
a wood litter in searches of roots and acorns. And in winter with the help of
tusks nozdrokh easily scatters snow, reaching up to a last year's grass. Jaws
of animal are rather short and strong, in case of necessity it can feed even
with young branches of trees and bark which it peels out by tusks. As against
to proboscides, at nozdrokhs the “conveyor” of replacing teeth is not formed.
Snout characteristic for pigs at ancestors of nozdrokh started to grow in length,
and became short, but flexible high-grade trunk. Due to it the animal can lift
even a small acorn from the ground. Inside the trunk there are some pairs of
plicas of epithelium. They serve as original additional “vocal chords”, expanding
a spectrum of sounds uttering by the nozdrokh - from the trumpet roar similar
to elephant’s voice, up to silent chirp and squeak.
Ears of nozdrokh are short – it lives in rather cool climate, and problem of
overheat do not arise at it. The animal is covered with a shaggy rough wool
of brown color. To winter at it the rich underfur grows, permitting to endure
rigorous frosts.
In ecosystem of woods of Europe nozdrokh partly occupies an ecological niche
of large wood hoofed mammals – aurochs and urus. It mainly eats leaves of trees
and bushes, adding to them fern rhizomes, tubers and bulbs of various plants.
In summer nozdrokhs willingly feed near reservoirs, pulling out by tusks whole
bushes of reed. Near reservoirs nozdrokhs spend the hottest days of summer.
Due to wide feet they easily cross bogs, and frequently come to bogs to wallow
in dirt and to cool.
This animal keeps in small herds (up to 5 - 6 animals) under the leading of
adult male. It does not tolerate competitors, and expels young males from herd
when they become independent (at the age of approximately 2 years). Except for
him, in herd there are 2 - 3 sexual matured females and other individuals are
young growth. Young males expelled from family groups, form barchelor groups
of 2 - 4 animals. They more tolerantly concern to each other until will get
own harem. Animals in group constantly communicate with each other with the
help of various sound signals – this way of communication is especially good
in rich wood where animals frequently lose sight of each other.
Female every time gives rise to one cub once per 2 years. It feeds exclusively
with her milk, but in case of danger can expect for protection of any of females
of herd. The cub is born rather weak. It is covered with longitudinal strips
– this is the type of juvenile colouring, characteristic for pigs. First two
days of its life all herd stays at the place, preserving it and expecting while
it will start to walk and will get stronger enough. Gradually strips on its
back become dimmer and disappear to bi-monthly age. Sexual maturity comes at
the age of approximately 20 months, and life expectancy may account about 40
years.
Porceratella
(Porceratella bovina)
Order: Even-toed ungulates (Artiodactyla)
Family: Horned hogs (Porceratidae)
Habitat: tropical woods and marshlands of Zinj Land.
 |
 |
Initial image - pictured by Pavel
Volkov
|
Picture colorization made by Odin
|
Mass extinction of the majority of groups of hoofed mammals at the boundary
of Holocene and Neocene became mainly the result of human activity. Only most
unpretentious and ecologically plastic species have managed to survive, having
kept as small populations. After disappearance of mankind they have started
to fill in released ecological niches.
The geography of planet in Neocene epoch has changed, though it has remained
rather recognizeable. From Africa along the “seam” of Great Rift Valley the
large land site had “broken away” – Zinj Land. And here one of species of hoofed
mammals, river hog (or bush pig) (Potamochoerus porcus), had turned to the
interesting creature similar to bulls of Holocene epoch.
The river hog had small bone knobs before eyes. Evolution had picked up and
had developed this “idea”, and the descendant of river hog, porceratella, had
acquired a pair of sharp horns directed forward and a little in sides. It is
interesting, that at the continent descendants of bush pigs either have lost
rudiments of horns completely or have acquired one large unpaired horn similar
to horn of brontotheres. All these animals belong to the separate family of
horned hogs (Porceratidae).
Osseous horns of porceratella are precisely separated from each other, and
resemble horns of bovids. They are filled with friable bone tissue lowering
their weight, but their top layer is enough strong and they could be used as
the tournament weapon, and for protection against predators.
Externally porceratella resembles small cow with back curved upwards. Height
of animal at a shoulder is about one and half meters, weight of the male is
about 500 kg, female up to 400 kg. The wool is short, rather thin, brightly
colored: back is rusty-red, sides are white and the bottom part of body is
black. The area under tail is covered with white wool. Behind on hips between
sites of white and red color there passes a black strip. Wool on throat is
also black. Around of eyes there are small white “glasses” (sometimes they
may be absent), on tips of ears there are rich black hairy brushes. The tail
is covered with rich white wool, its tip is black.
The nose has turned to small mobile proboscis, shorter, than at saiga or tapir.
Characteristic for pigs ossifications in snout are reduced and the animal can
not dig out by it something from the ground, but can pick up edible parts of
plants. Due to proboscis the animal can utter various blares. Also the big
surface of nasal ducts promotes development of sharp sense of smell. Sight
plays smaller role in life of porceratella: the bases of horns form protective
“eyebrows” which prevent it to see object that is directly ahead: to make it,
the animal raises head a little.
On the top jaw there are sharp tusks directed forward and in sides. With their
help the animal digs ground in searches of food. In the case necessity porceratella
can put deep wounds by these tusks.
Hoofs can move apart, due to what porceratella is able to walk along fenny
marsh ground. Besides it does not avoid water: it willingly swims, feeds with
water plants, and searches in water for protection against ground predators.
Porceratella keeps in herds numbering 15 - 20 ones. Over the herd is predominated
by the female, and other members of herd are also females. Males keep in herd
only while depend on mother. The leader female expels become adult males. Males
form barchelor groups of 5 - 6 animals in which the not strict hierarchy is
observed. But in season of pairing (in the middle of rain season) they become
aggressive, and can even kill each other in tournament fights.
The posterity is born in the beginning of dry season, when there is no danger
of flooding of forests. In litter there are usually two cubs, at young females
only one cub is born. At the age of approximately three months at young animal
horns start to grow, and half year old males are already compelled to lead
independent life. They completely develop and become able to breeding at the
age of 2 – 2.5 years.
Bikifaru
(Dilophoporcula rhinocerotoides)
Order: Even-toed ungulates (Artiodactyla)
Family: Horned hogs (Porceratidae)
Habitat: Zinj Land, light forests and bush thickets in the north part of subcontinent.
Picture by Carlos Pizcueta (Electreel), colorization by Biolog
Picture by Carlos Pizcueta (Electreel) |
Artiodactyl mammals in Holocene turned to favourite objects
of people hunting. Human activity has resulted to strong reducing of the number
of various gregarious animals (mainly bovids), and the majority of species of
these animals had disappeared. Only few relic species kept in Asia and South
Africa. The place of small swift-footed antelopes in Neocene was occupied by
various animals of new groups: harelopes
of lagomorph group and hyracolopas, cursorial
descendants of hyraxes. And the place of larger herbivores – rhinoceroses and
buffaloes – representatives of other groups of mammals, various descendants
of damans, had shared. At the East-African subcontinent named Zinj Land, large
descendants of hyraxes are represented only by one
semi-aquatic species. At the subcontinent the large descendants of pigs
occupied ecological niches of ruminant animals had appeared. One of the most
widespread species is bikifaru, large species of horned descendants of pigs.
This animal is larger relative of porceratella,
which leads migrating way of life and differs in rather graceful constitution.
This is stout cow-sized animal, also similar to cows by proportions. At the
short distance bikifaru can accelerate momentum up to 50 kms per hour, but it
can not support such rate for a long time. Hoofs of this animal can move apart
- bikifaru frequently feed on juicy marsh vegetation at the fenny ground. Also
this animal is not afraid of water and is able to swim well.
Wool of animal, living in hot climate, is rough, short, sandy-yellow colored.
On back and hips there are some dim darker spots. The tail of animal is long
– with its help grazing bikifaru drives off blood-sucking insects. On tail there
is big black-and-white brush of long hair. Using it the animal submits signals
to congeners: the tail with hairy brush lifted upwards, well seen from apart,
is the alarm signal, clear not only for bikifarus, but also to their neighbours.
Neck is moderately long; head is rather small. But the structure of head of
animal has determined its name. “Kifaru” in suachili means “rhinoceros”, and
“bi …” means “double” in Greek. On head of this animal not horns, as at porceratella,
but two longitudinal bone crests grow. These outgrowths are too thin to be used
for combat, and serve more for demonstration. Also they help to move apart prickly
branches of bushes among which bikifaru lives and feeds, and protect eyes from
damages. Crests are semicircular; on nape they stick back like two horns. These
crests are covered with thick rough skin and short wool, and frequently grow
very large – at old males the size of crests can be almost equal to the seen
sideview of head. Crests of males have a border of black wool on edge, and due
to it are appreciable from apart. At females on crests there are only separate
black spots, and at young animals colouring of crests is the same, as at all
body. The width of black border depends on level of testosterone, and this feature
helps males to estimate force of each other, not entering combat. Females also
prefer to couple to males, at which there is more of black wool on crests. Also
at males at edges of crests knobby osseous outgrowths develop. At males the
small tusks, inflexed and turned in sides, also are advanced. This is the weapon
for demonstration, but the defending animal can strike a predator by lateral
movement of head. At females tusks are smaller and are visible only when the
animal opens mouth.
Lips of bikifaru are mobile, and the muzzle is extended to small proboscis.
Inflating proboscis and contracting nostrils, animal can utter various sounds.
In bushes where visibility can be very bad, this animal communicates with congeners
almost exclusively with the help of sounds.
Bikifaru is strict vegetarian; it eats leaves of bushes and branches of young
trees. Jaws of this animal are rather weak; therefore the animal does not eat
rigid grass. Due to the structure of hoofs bikifaru can graze in bogs, where
it eats leaves of huge grasses and gathers floating plants from surface of water.
This species lives the herds numbering up to 20 – 30 adult animals. The herd
is ruled by large male which constantly keeps up observance of hierarchy. Between
males there is rather rigid struggle for leadership: the dominant male can couple
to the majority of females of herd. Males standing a rank below the leader also
have opportunity to have posterity, especially in big herd. The old animals,
not able to support by force the place in hierarchy, quickly fall to the bottom
step of hierarchy of herd and only the young animals recently becoming independent
can be lower than they are. Old males usually keep in back part of herd, and
fall victims of predators the first.
Seasonal prevalence in breeding of bikifaru is not present. The female ready
to pairing involves at once many males which arrange combat for the right to
couple with her. Pregnancy lasts about eight months and is finished by birth
of one or two cubs (twins are born in third of cases). The cub is born advanced
and can walk at once. It differs from adults in absence of longitudinal crests
on muzzle. Its colouring is brighter, rather than at adults – on sides of young
animal there are black spots, merging to faltering longitudinal strips. At growing
animal they grow pale and vanish on the most part of body. At the animals living
in richer bushes and light forests in south of area, at the border of rainforests,
spots are kept to all life. These animals, larger than northern ones, are classified
as the subspecies Dilophoporcula rhinocerotoides maculata.
Brontothere
hornhog (Porceras brontotherioides)
Order: Even-toed ungulates (Artiodactyla)
Family: Horned hogs (Porceratidae)
Habitat: savannas and light forests of Southern Africa, dry sites, foothills.

Picture by Tim Morris
Picture by Pavel Volkov - the initial image |
Presence of small bone outgrowths before eyes at bush pigs
(Potamochoerus porcus) looks insignificant attribute at first sight. But evolution
had picked up this attribute, and it has received the development at descendants
of these animals: at them on head large bone outgrowths of various shapes have
appeared. So on life arena the family of non-ruminant artiodactyls, horned hogs
(Porceratidae), has appeared. At the representatives
of this family living at the Eastern African microcontinent, there is the
pair of horns a little bit similar to horns of bovids. And at inhabitants of
continental Africa on the head mainly unpaired outgrowths were developed, making
these animals little bit similar to rhinoceroses extinct at the end of Holocene
and brontotheres dyed out to end of Paleogene. In continental Africa these animals
are spreaded mainly to the south from zone of tropical woods, in damp savanna
and mountains of Southern Africa.
The brontothere hornhog is one of the most typical representatives of the family,
widely widespread in rather dry habitats. By constitution it is similar to bull
(growth at a shoulder is up to one and half meters), but its head strikingly
differs from bull’s one. It is larger, with rudimentary proboscis developed
from characteristic snout of pigs. The most remarkable feature is the presence
of huge osseous horn. It is formed by pulled and grown together pair bone knobs
characteristic for ancestor. The horn rises perpendicularly to forehead; it
is expanded in sides and partly doubled at top. It is rather demonstration,
than fighting weapon. But with the help of strong cervical muscles the brontothere
hornhog can hook by horn the attacking predator and reject it aside. During
intraspecific tournaments males of this species butt, having inclined a head
aside, and resting by bases of horns. But usually they avoid direct fight, and
only show horns to each other. At females horns are shorter, than at males.
Wool of this animal is very short. On chin hair are a little bit lengthened.
The horn is hairless, covered with cornificate skin. The basic colouring of
body is light grey, only on nape lengthened black hair form similarity of mane,
and on long tail there is black hairy brush.
In mouth there are two long incisors similar to teeth of fossil rhinoceros Chilotherium:
animal uses them to dig out roots and tubers. Also during courtship duels the
animal can put to the opponent stings. Jaws of brontothere hornhog are long
and rather weak. The basic food of animal includes young grass and leaves of
bushes.
The brontothere hornhog is able to run quickly and long – at it legs are rather
long. In this connection hoofs of this animal are small – this way friction
against the ground is less. But it imposes the certain difficulties: the animal
is compelled to avoid fenny areas, and comes into water only in case of emergency.
Animals avoid places where there are flooding. Differing in ability to predict
weather, they leave lowland areas to which flooding may threaten, and migrate
to foothills. At this time separate family groups may unite, and then between
males fights happen.
This species keep in small herds (up to ten adult animals), each of which represents
a harem of one male. Animals communicate with each other by means of various
sounds uttering with the help of short proboscis (at this moment it is slightly
puffed). The young growth keeps with parents before independence then males
are expelled by the leader, and females – by adult females. Seasonal prevalence
in breeding is expressed poorly, but the majority of cubs is born in the beginning
of rain season. In litter there is always one cub.
Bullhog
(Buchoerus crassoceras)
Order: Even-toed ungulates (Artiodactyla)
Family: Horned hogs (Porceratidae)
Habitat: savannas and light forests of Southern Africa, damp sites near
to rivers and bogs.

Picture by Tim Morris
Picture by Pavel Volkov - the initial image |
When during evolution process in nature a “successful” variation
of live beings appears, on its basis related forms adapted to different conditions
of inhabiting at once start to evolve. In Southern Africa at the basis of the
common ancestor different species belonging to new Neocenic family of horned
hogs were evolved. These animals have partly replaced large ruminant artiodactyl
animals, not sustained chasing from the side of people and destruction of habitats
during a catastrophic congelation. Hornhogs became analogues of buffaloes, large
antelopes, and partly even rhinoceroses. Bullhog is the largest representative
of family.
This species of mammals differs from brontothere
hornhog by more powerful addition: the animal is very similar to large buffalo.
The highest point of the body is at shoulders, back is a little bit sloping.
Wool is short, pale-yellow, and lighter on stomach. On cheekbones wool forms
black “whiskers” especially obviously expressed at males. Legs, partly shoulders
and groats of animal are covered with short horizontal strips of rusty wool.
Tail is long, with hairy brush on the tip.
The most characteristic attribute of animals of horned hog family is the horns
of various shapes developing in middle part of head. At bullhog, despite of
the name, horn it is completely not similar to bull horns: it is wide and flat,
reminiscenting somewhat the horn of living in North-African savannas
flathorn (Platyceratherium foetidus), huge animal from order of damans (Procaviiformes).
It is wide, covered with cornificate skin, and concave in middle part. The horn
of bullhog is mainly the tournament weapon, rather than fighting one: in courtship
season between males of this species there is a force struggle by necks (muscles
of neck are very strong). Horns serve as protective “helmet”: they are formed
by thick layer of bone tissue with a minimum quantity of cavities. But also
this horn may be used as the weapon: to attacking predator protecting bullhog
puts ramming impact by head. The horn protects eyes from casual wounds, therefore,
having “hooked” predator by flat horn, bullhog can lift it in air, and then
simply strike a predator against tree or throw off far.
As against the relative, the brontothere hornhog, bullhog prefers sites which
may be flooded. This animal willingly bathes and wallows in dirt, and also searches
near to reservoirs for significant part of food – soft vegetation. In this connection
at bullhog the wide hoofs able to move apart partly, when the animal grazes
at the bog, were developed. Also the animal is able to swim very well, and easily
crosses even wide rivers and lakes.
Bullhogs keep in small harems: one male and 3 - 4 females with cubs of the age
till one year. Such harem occupies the certain territory along the river bank,
or in bogs, and does not leave it even at flooding. Except for such families,
there are single males living at the narrow border lands between possessions
of family males. Usually they are either very old males expelled by young contenders
from own groups, or young males, yet not having own harem. In courtship season
young animals challenge family males, and between them there are skirmishes.
Once a year the female gives rise to 1 – 2 cubs. They differ from adults by
darker colouring in which there are more strips (they stretch even on back and
sides). The female carefully preserves them against predators, and easily may
attack any animal if it will seem that the invader represents threat for the
cub. The young growth is fed with milk till four months, and then completely
passes to vegetative forage. One-year-old youngsters abandon herd: shortly before
birth of the new cub the female starts to show aggression to them.
Sexual maturity comes at the age of about 3 years, life expectancy may account
till 40 years.
Hyena-like
deinaper (Deinaper crocutoides)
Order: Even-toed ungulates (Artiodactyla)
Family: Predatory boars (Carnoporcidae)
Habitat: grass plains and bush thickets of Central and East
Asia.


Pictured by Eugeny Hontor
Colorization by Alexander Smyslov
Early image - pictured by Pavel Volkov |
In Paleogene and
early Neogene among characteristic inhabitants of open spaces of Eurasia
and North
America there were entelodons (Entelodontidae) – family
of non-ruminant artiodactyls, related to pigs. By structure of teeth, they
were omnivores in which diet meat took an essential place. After epoch of
mass extinction when many ecological niches appeared free, pigs have taken
advantage
of a situation, and have taken a vacant place of scavengers. So at plains
and in bush of Asia the family of Predatory boars partly occupied a place
of
hyaenas and bears had appeared.
The typical representative of this family is deinaper (“deino-“ means “terrible”,
and “aper” – the boar), the largest species in family. An ancestor of this
animal was wild boar (Sus scrofa), the species widely widespread in Eurasia
and have successfully survived the period of anthropogenous pressure. The
important place in diet of boar was occupied with food of animal origin.
One of the evolutionary
lines, originated from this species, has gone to the way of adaptation to
searching and eating larger amount of food of animal origin. Gradually these
animals
have turned to scavengers, and even had become able to attack cubs of other
animals.
Deinaper is an animal of massive constitution, its growth at a shoulder is
about 0.8 m and weight is up to 140 kg. By constitution it is similar to
the spotted hyena: at it there is large head on mobile neck. Its body is
covered
with shaggy rough wool; to winter rich underfur grows. In connection with
unusual for hoofed mammals way of life at it spotty colouring developed –
strips characteristic
for boar juvenile colouring, break up to separate spots. It even more strengthens
similarity of deinaper with hyenas. Legs of animal are rather long, adapted
to fast running to the big distances. Because of long legs the body seems
a little bit short.
Jaw are short and high, able to crush bones. There are large sharp tusks
jutting out up and in sides. They are used as the weapon in tournament fights,
but
in case of need deinaper can use them against predators.
The appearance of muzzle of deinaper is characteristic for pigs. Sense of
smell is sharp - with its help animal searches for carrion and finds small
animals.
Deinaper is aggressive: it can drive away medium-sized predators from catch,
threatening them with tusks and making sharp attacks on aside a predator.
As this animal lives small groups (up to 4 – 6 adult animals), not any predator
can defend the right to the catch in the face of several strong and aggressively
adjusted animals.
The group of deinapers supervises extensive territory on which there are
some shelters. Borders of territory are marked by heaps of manure, and constantly
renewed. In shelters animals spend hottest time of day, becoming more active
in the morning and in the evening when the majority of predators hunts. If
won over catch at predators does not suffice, deinapers can hunt in the afternoon.
Then they attack small animals having a rest in a shadow.
Once per year the female brings posterity: up to 5 – 6 cubs. Feeding female
does not take part in hunting, protecting posterity. The only thing, that
it presumes to herself – to catch any tiny casual catch like reptiles and
rodents.
But males and females without cubs after successful hunting necessarily belch
for feeding female pieces of meat. Newborn cubs are weak and helpless: they
start to stand only at the age of one week, and pass to feeding by meat only
at bi-monthly age. Approximately at the age of three months young animals
leave lair, and lead nomadic life with parental group. Only at the second
year of
life the young female can give posterity the first time. And the young male
can really apply for domination in clan only since the fourth year of life.
Up to this time it leads life of the lonely hunter in boundary territories,
gaining strength and experience to challenge once in winter to any old leader
of clan and to take his place.
(This animal is discovered in common: by Tenek, the participant of forum (she has stated the assumption of existence of the present animal in Neocenic fauna), and by Arseny Zolotnikov (he created initial sketch of the present animal))
Varaha
(Barodeinaper waraha)
Order: Even-toed ungulates (Artiodactyla)
Family: Predatory boars (Carnoporcidae)
Habitat: Southern and South-East Asia, Hindustan; forests of various types and
bush.

Picture by Alexander Smyslov
Pig family is among champions on survival. These animals,
easily growing wild and easily adapting to various conditions, used in full
degree possibilities on development of new habitats opened after human disappearance.
In due course of evolution the North of Eurasia was occupied by trunk boars,
and in Africa and on Zinj Land horned forms of pigs evolved. For Asia the special
family of predatory boars is endemic – it is a peculiar attempt of pigs to become
carnivores as entelodons did in Tertiary. In temperate areas of Asia the hyena-like
deinaper (Deinaper crocutoides) is widespread – it is the carnivorous species
being ecological analog of hyenas. To the south of the Himalayas it is replaced
by varaha, the species of close genus.
Compared to deinaper varaha is larger species adapted for life in rainforest.
Its growth at a shoulder is about one and a half meters, body length to 2 meters,
weight is about 400 kg. Varaha has powerful jaws and extended wedge-shaped muzzle
which ended with the small snout. Skull bones are quite massive – in case of
need varaha strikes to competitors blows with a forehead. Tusks of animal are
well developed and inflexed, but are turned sideways only a little, therefore
they don’t hamper the movement of the animal through dense underbrush. The main
purpose of tusks is to be the duel weapon. During courtship season males hook
the contender by tusks and try to tumble down each other, using strong muscles
of neck and shoulders. Wide ears are used for heatreturn though in daytime heat
varaha more often simply searches for a reservoir where it has a rest during
the hottest time of day. On neck and shoulders of animal the standing bristly
mane is developed similar to mane of wild horses and creating the illusion of
larger size, than is in fact.
Skin on neck and shoulders of varaha is very thick and strong – even a large
predator hardly bites through it. For attaching of the muscles supporting the
head, at this animal spinous processes on cervical and chest vertebrae are well
developed. Because of it the back of an animal is sloping a little.
Legs of varaha are short, but strong, with two well developed hooves on 3rd
and 4th toes. The 2nd and 5th toes are underdeveloped, and their hooves are
appreciable as two small bumps on each side of fore- and hind legs of animal.
Unlike the spotty northern relative, varaha has longitudinally striped coloring.
The main background of coloring is reddish-brown, on the back narrow longitudinal
strips of black color stretch. Coloring of the top part of head of this animal
sharply contrasts with body color – it is black with two large white spots under
eyes. This coloring has obvious warning character – varaha has aggressive temper
and can easily rush on the competitor, driving it away from food. Lower part
of head is brown, and mane is black.
Varaha eats mainly food of animal origin. This animal often eats carrion and
searches for it, using keen sense of smell. Also varaha attacks small animals,
which are trampled or biten to death, and then eats them entirely. If necessary,
varaha can win away prey at predators – it attacks and tries to bite a predator
or to hook it by tusks. Due to skin “armour” and strong skull it easily endures
blows of predators and more often defeats them, forcing them out from prey or
to wait, while varaha will be sated. Bumps on teeth of animal have sharp tops
and cutting edges.
Varaha female gives birth once a year to three cubs. Piglets spend first two
weeks of life in den built in secluded place, and then leave it and lead nomadic
life, keeping near mother. At the age of about 5 months between them the competition
becomes aggravated and they start to lead independent life. The sexual maturity
at them comes at the age of 2 years, but males receive real possibility to take
part in reproduction not earlier than at the age of 4 years. Life expectancy
reaches 30 years.
Translated by Alexander Smyslov.
Armor-headed
wart hog (Geophacochoerus subterraneus)
Order: Even-toed ungulates (Artiodactyla)
Family: Pigs (Suidae)
Habitat: savannas and light forests of Southern Africa.

Picture by Amplion
Ancestor of present species is usual in Holocene African
wart hog (Phacochoerus). These hoofed mammals spend significant part of day
in holes dug independently.
And its descendant has finished this habit of life to extreme being shown
on ground surface only at night. This animal spends day in shelters – wide
holes
at times directed downwards to two meters depth and stretch under ground
to tens meters. In connection with such original habit of life the shape of
animal
had strongly changed.
The armor-headed wart hog reaches height only 40 cm at a shoulder. But its
body is very much extended – almost meter longwise not including tail. About
20 % of body length the head turned to tunneling mashine amounts. At this
animal there are large, slightly bent tusks sticking up forward and in sides
– this
animal digs and deepens holes, at feeding on surface overturns stones in
searches of insect larvae and digs out plant tubers and rhizomes by them.
At the top
part of head thick layer of cornificate skin protecting head from casual
fallings in holes is advanced. Tip of muzzle with “snout” characteristic
for pigs is
very mobile that partly compensates hindrances created by tusks at feeding.
Nostrils of animal are closing during digging. Nasal courses are rather short
but the olfactory surface forms numerous plicas: at armor-headed wart hog
it is very sharp sense of smell. Sight of animal is rather weak; eyes are
protected
from ground by dense eyelashes. Ears are small; at digging they fold lengthways
and nestle to sides of head. On cheekbones the large bone outgrowths especially
advanced at males stick up.
Body is long and legs are short. On forward legs in front, and on back ones
under hock there are calloused outgrowths serving as a support at ground
digging (animal digs a hole standing “on knees”). Hooves are wide; at digging
they
are used for shoveling up the dug ground. Hairs on body are submitted by
short thin bristle, “mane” on back characteristic for wart hog is not present.
Skin
is wrinkled and grayish-black. At the tip of long tail there is brush of
white hair: tail is used for communication and signal submission.
Armor-headed wart hogs live in groups: dominant male, female and cubs of
current litter (up to 5 cubs 2 times per one year are born). At new litter
birthing
at the female grown up young animals are driven out. The family of animals
lives in constant territory which is jealously preserved against neighbours.
At the territory usually there is a main hole and plenty of temporary refuges
well known to all family of hogs. In due course holes are extending, sometimes
systems of holes join to each other. At exhaustion of food sources around
of main hole the family moves (at night or dawn) to other hole, somewhere
to the
edge of territory. Gradually the centre of territory is actually displaced
here that inevitably results to revision of borders between neighbours.
In each hole there is a main chamber where the female with posterity lives,
and “throne hall” where the dominant male lives. They are located opposite
to each other and between them main burrow with two exits (one of which is
disguised outside by dust) passes through. On each side of this pass animals
dig out temporary toilet chambers leaving dung there. After filling the chamber
is filling up by ground and simultaneously the new chamber is making. This
way animals hide their presence from predators.
In holes of armor-headed wart hogs a plenty of “lodgers” settles: butterflies,
crickets, numerous fleas (they parasitize in thin wool on throat and stomach
of animal). Among vertebrates large tortoises and lizards (hogs simply can
to have eaten small reptiles) and also birds and small mammals settle in
holes. For protection against parasites wart hogs willingly take dust baths
sometimes
arranging place for bathing right in heap of ground thrown out from hole.
Family of armor-headed wart hogs goes for feeding at night. At first male
appears from hole; he looks, listens and smells around determining whether
there is
no danger. If all is quiet he walks out and after him female and cubs appear.
At night animals go to watering place (if the water source is far some burrows
to it are dug allowing to not show itself to superfluous danger at the watering
place) and then feed on meadow or under trees. The basic forage of armor-headed
wart hogs is grass. But in trees and bushes fructification season they willingly
eat drop fruits and berries. Animals willingly eat forage of animal origin:
worms and beetle larvae gathering them at hole digging, searching under stones
or in rotten wood. Differing in keen sense of smell such pig easily feels
presence of larva under thin layer of wood and if it is necessary splits
mouldering
tree by sharp tusk. If it is possible to find carrion or remains of predator’s
catch animals willingly feed with meat.
During feeding armor-headed wart hogs communicate wagging with white hairy
brush on the tail tip. Their voices at this time are similar to usual pork
grunt. Tail sharply lifted upwards is an alarm signal: having seen such mark
animals cease eating and start to smell around trying to find out predator.
In case of danger animals squealing shrilly rush to nearest hole trying to
keep near it at all. These pigs are fast and maneuverable runners though
they can not run long on short legs. At deviation male climbs to the hole
at last
having turned to predator by head. He loudly roars trying to frighten predator
and from time to time makes aggressive lunges trying to strike attacking
predator by tusk. Usually after that only a little number of wishing to continue
chasing
ones may be founded.
Maturity at females comes at two-year-old age, at males little bit later.
Young females at the second year of life pair the first time and approximately
after
three and half months give rise to posterity. The first litter is insignificant:
no more than three pigs and usually only two ones. But at the fourth year
of life in litter it may be up to five pigs.
Young growth abandons parents shortly before birth of next cubs or right
after it. Youngsters first time keep in parental territory using for life
old holes
- at them tusks are still insufficiently grown. But when the young growth
new litter grows up, the dominant male, their daddy, expels all of them from
territory.
At this time young animals perish in a plenty from predators. But ones managed
to find suitable place for life, have an every prospect to live up to old
age: the age limit of these animals reaches 15 years.
Cerdito-peluda
(Insulosus volcanica)
Order: Artiodactyls (Artiodactyla)
Family: Pigs (Suidae)
Habitat: Galapgos islands, open country, scrub and forested areas. Is distributed
on all large islands due to its ability to swim well.
Mankind managed to not wholly “deflower” the Galapagos Islands, early emphasis
placed on its uniqueness and conservation resulted in the eradication of some
invasive species, and the preservation of native ones. But introduced kinds
of animals still left their mark, and evolved new forms, among these founders
was the Domestic Pig (Sus domesticus). Cerdito-peluda is relatively small for
a pig, reaching about 60 centimetres long with a shoulder height of up to 40
centimetres. Coat is a pelage of soft bristles, denser than that of its ancestor
but not as thick as that of other wild pigs. From this it gains its common name,
Spanish for “hairy little pig”. Colour is a fawn brown, with darker stripes
running longitudinally along the body, this is a trait gained atavistically
from the Wild Boar (Sus scrofa), predecessor to the Domestic Pig. Males bear
an upright mane of stiff bristles of dark color, and have prominent tusks, especially
in older individuals. Head is fairly large proportionally, and bears a disc-shaped
“snout” by which it digs and forages in the soil, ears are large and hairy on
the outside. Exposed areas of skin are dark reddish-brown in color, the pink
of its ancestor being prone to sun-burn.
Social grouping consists of herds of up to 20 animals, a harem style arrangement
of a male with many females including their offspring. Young males live in bachelor
herds with weakly expressed hierarchy. Diet consist of fallen fruit and seeds,
soft vegetation, fungi, eggs of ground nesting birds, carrion, and occasionally
small vertebrates and insects.
Lead male within the herd maintains authority by antagonising subordinates,
but also helps to fiercely defends comrades from predators and territorial interlopers.
Females produce young 2 times a year, litters of up to 6 piglets, which are
nursed within a secluded “nest” made of dry grass, obscured and hidden by undergrowth
or bushes. Young mature quicker than at its domestic ancestor, individuals are
able to walk and run within a few days, and are sexually mature within 6 months.
Lifespan may be up to ten years but is usually as short as six years.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Crimean
pygmy boar (Neosus tauricus)
Order: Even-toed ungulates (Artiodactyla)
Family: Pigs (Suidae)
Habitat: Crimea Island in Fourseas, landscapes of various types.

Picture by Alexey Tatarinov
During late Cenozoic pigs had proved their evolutionary success,
having settled on to all Eurasia and Africa. In human epoch they have had the
minimal losses: areas of widespread species had been reduced, and some endemic
species and subspecies have died out. But after disappearance of mankind they
have quickly returned to initial positions. Large ruminants were strongly exterminated
by people, therefore pigs have taken advantage of the situation formed in biosphere,
and have evolved some new families becoming analogues of large ruminants in
certain degree. Representatives of the initial branch of pigs, true pigs, also
were kept in Neocene due to their universality and abilities to adaptation.
During the formation of Fourseas Crimea had turned to island. There the rests
of local fauna which evolved in isolation during several millions years were
kept. Prominent feature of local species is so called island dwarfism – very
small size in comparison to continental species. One characteristic local species
is Crimean pygmy boar, the descendant of Holocene wild boar (Sus scrofa), widely
settled and successful species of ungulates. Appearance of this animal is characteristic
and recognizeable. Crimean pygmy boar has shape characteristic for pigs, with
more flexible snout, rather than its ancestors, resembling early stages of evolution
of snout of trunk boars. Length of its body is about 60 cm at growth less than
half meter at a shoulder. At this species there are rather long legs permitting
to run quickly and to swarm up rocks. Colouring is skewbald – usually head and
legs are colored lighter than body, but extreme variants exist: almost entirely
black colouring with white legs and almost white one with dark longitudinal
strip on back. This species is adapted to life in rocky landscape: it swarms
up mountains excellently, and is strong and hardy. This pig can run on narrow
rocky eaves and clamber on slopes. By nature this wild boar is very cautious
and has good hearing and sight.
This is a gregarious animal; Crimean pygmy boar moves in small herds numbering
10 – 15 ones ruled by the most skilled and strong male. In places most frequently
visited by them tracks are well appreciable, and if the track stretches through
bushes, among thickets the tunnel on which animals move one by one is formed.
Having kept an omnivorousity characteristic for pigs, it eats animal food –
reptiles, rodents, insects, worms, bird eggs – much more often, than its ancestors.
At an opportunity this animal willingly eats carrion. But as it grows the diet
passes to herbivory, and the oldest individuals frequently leave in marshy sites
of island with plain relief and soft vegetation. Usually beyond the breeding
season males of Crimean pygmy boar are tolerant to each other.
Heat happens in December – January, but even that time male skirmishes seldom
are out of limits of simple display: males rise wool growing on back and nape,
scatter ground by hooves and stamp legs. Pregnancy lasts till about 90 days.
The female, as a rule, gives rise to two pigs of the same gender – it is connected
to limitation of habitats of this animal. Cubs are born in reliable lair: usually
in a cave in rocks or in specially dug holes if caves are not present. The same
holes and caves are used by years and pass from generation to generation. Pigs
are born striped, but as they grow their colouring changes: strips vanish, or,
on the contrary, extend. Depending on genetic features colouring of adult animals
varies in great degree. Young animals are fed with milk till 1 – 1.5 month;
at this time the female lives separately from herd and leads very secretive
life. After the young growth will get stronger enough, female bring cubs to
the common herd. Females become independent at the age of 8 – 8.5 months, males
– at 11 – 11.5 months. The sexual maturity comes in 2 years, and then young
males are frequently expelled from herd by the leader. Representatives of this
species live up to 37 years.
This species of mammals was discovered by Nick, the forum member.
Great
Porkoceros (Ceratodens babiroussoides)
Order: Artiodactyls (Artiodactyla)
Family: Pigs (Suidae)
Habitat: Lowland rainforest and swamp forest of northern Meganesia.

Picture by Alexey Tatarinov
During the Holocene, the continent of Australia had suffered
heavily from introduced animals of all kinds. Due to the harsh nature of the
climate, most hoofed animals have disappeared as the neocene progressed. Sheep
were too poorly adapted to the deserts, and too reliant on man, for example.
The camels, practically pre-disposed to an arid existence, proliferated and
came to dominate the dryer part of Meganesia. The other exception are the feral
pigs, versatile and adaptable, they evolved to Meganesian equivalent of rhinoceros,
the Great Porkoceros. This creature is a browser of low growth in the extensive
rainforest and swamp forest of northern Meganesia, much like the Holocene forest
rhinoceros.
This animal is built similarly to a rhinoceros, attaining 1500-2000 kilograms.
The animal has an extensive barrel-chest and a bulging stomach which possesses
an extensive gut. Unlike its predecessor, it subsists entirely on plant matter.
Its limbs are sturdy, but thinner compared to those of a rhinoceros. Broad hooves
spread the animal’s weight in soft or swampy ground. The shoulders come to a
distinct hump, which serves to support its thick neck and tremendous head.
Head of this animal is large and deep, with a broad snout compared to the body,
and has large flaring cheek bosses which serve as protection during combat.
The upper tusks of its ancestor have become the analogue of a rhinoceros horn.
The cylindrical upper tusks project through the top of the snout, like those
of a babiroussa, except that they project upward in the manner of 2 large horns,
the tusks of the female are smaller and more slender. The lower tusks are strongly
reduced. The animal is mostly hairless, possessing a thick, leathery hide of
chocolate colour. Hairs only persist on the tail tuft, ear tufts and along the
neck. The cartilaginous nasal cap is reduced and sits under the skin, while
the lip extends outward in a small proboscis like that of a rhinoceros.
The face and sides of this animal are covered in heavy, wart-covered calluses.
These are to protect the animal from serious injury during combat with one another.
As it browses, the Porkoceros clears distinct trails in the undergrowth; these
trails are also used by other forest dwelling animals, which browse on the regenerated
growth that emerges after the animal has passed. The Yagil
and the Marsuipial panther
are able to attack this beast, but they prefer to catch the more vulnerable
juveniles, which have less-developed tusks and dermal thickening. This animal
is predominantly solitary, only coming together to mate and to compete for mates.
Rutting and breeding season takes place at the height of the dry season, allowing
the females to raise the young in a safe, dry nest.
The female gives birth to 3 or 4 young at a time, and suckles them in a large
shelter made from branches and lined with soft bedding. The newborn piglets
possess lengthwise light stripes on a dark background. When the males compete
for females, they display their tusks as a stag would his antlers, rasing their
heads and showing them side-on. When in combat they make blows to the body and
face with their tusks, and push against each other with their shoulders, in
an attempt to exhaust one another. Great Porkoceros have a lifespan of up to
40 years; males that reach old age often have partly broken tusks and large
scars from fighting.
Smaller relative Lesser
Porkoceros (Ceratodens hirsutus), reaches only 150 kg, and is covered
in light brown hair, which forms a crest along its back, which is lighter. It
only possesses medium-sized protruding upper-tusks, which cross over each other;
lower tusks are present and quite large. It mainly inhabits the dryer central
scrub and grassland, where it feeds on herbage and shrubs, and some small amounts
of softer grass. It prefers to shelter in large areas of scrubland, where it
is able to hide more easily. The Marsuipial Panther frequently preys on this
animal. Lifespan of up to 35 years, but usually much shorter.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Poaka-ngaherehere
(Pseudochoerus sylvanus)
Order: Even Toed Ungulates (Artiodactyla)
Family: Pigs (Suidae)
Habitat: Lowland forests of New Zealand, North and South Islands, sub-tropical
woods of North Island.
Run wild domestic pigs have left numerous descendants in the Neocene of New
Zealand; Poaka-ngaherehere is one of them, name meaning “forest pig” in indigenous
Maori language.
Poaka-ngaherehere differs from their domestic ancestor in smaller size, being
only about 70 kilograms in weight and 60 centimetres tall at the shoulder. Also,
their face is distinctive by broad, warty outgrowths on the cheeks and snout,
supported by bony ridges underneath. Tusks are larger than the ancestral form,
even approaching those of a warthog (Phacochoerus) in size, coat is sparse,
short reddish bristles with pale stripes, retention of juvenile colour. Males
have a long erectile mane on the back which forms a crest, face has pale markings
around the eye and base of the snout, running in a stripe to the cheek, snout
and feet are dark pink. South Island populations form a separate subspecies,
having denser bristles and a slightly larger size.
This animal is most active at in the morning and afternoon, preferring twilight,
its diet is omnivorous, typical for a wild pig, roots, fungus, fallen fruit
and nuts, large invertebrates, carrion and eggs, rare live vertebrates are also
sometimes taken. Sounders are relatively large, dominated by a big female, and
groups follow trails worn down through the undergrowth, deviating from these
to forage. Males form small bachelor groups, but elder males are generally solitary.
Calls are reminiscent of those of a domestic pig, but higher in pitch.
Breeding season runs from July to October, when there are abundant stocks of
food. Males seek out sounders of sows by scent, and produce strong pheromone
secretions to communicate with rival males. Contests over males are less violent
and damaging than those of its ancestor, as tusks also form a display structure,
and warty growths on the face afford some protection. A single male will mate
with multiple sows, although the mating period does not last long. Gestation
is about 130 days; nest making precedes the birth of the litter by about a month.
Litters are large, consisting of up to twelve piglets. As with other pigs, young
can follow their mother on excursions at about two weeks old and are fully weaned
by 3 months, at which stage contrasting striped baby colouring gives way to
more subtle pattern of the adult. Lifespan is up to 8 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Poaka-maunga
(Notosus montanophilus)
Order: Even Toed Ungulates (Artiodactyla)
Family: Pigs (Suidae)
Habitat: Mountain forests and upland slopes of mountains, North and South Island
of New Zealand.
In the wild country of Neocene New Zealand, descendants of run-wild domestic
pig of Holocene occupation are notable. A generalized ancestor has radiated
into different habitats, producing new forms.
Poaka-maunga somewhat resembles Sus scrofa, the wild boar of the Holocene, but
it is somewhat smaller and more compact in shape, with proportionately longer
legs. Adults average about 80 kilograms in weight and shoulder height is 85
centimetres. There is a dense coat of bristles that gives it a shaggy appearance,
but this is thinner in the North Island subspecies. Coat colour varies from
light brown to dark brown or almost black, white markings stretch from the eye
to the cheek. Legs are relatively long and aid it in walking on sloped or rocky
ground; this animal is surefooted and can flee predators on slopes with ease.
Accordingly its name translates to “mountain pig” in native Maori language.
Diet is similar to ancestor, but comprises more vegetable matter, mainly grass
and roots, but also occasionally fungus, and fallen seeds from coniferous trees.
Animal forage is still taken, worms, birds eggs, carrion and insects, but it
takes up smaller portion of the diet in more open countryside. Poaka-maunga
is mostly diurnal, but will also forage at twilight. These pigs live in small
sounders compared to their ancestors, with males being solitary or in small
bachelor groups, seeking out females in the breeding season. Calls are almost
identical to their ancestor, grunts, squeaks and squeals.
Breeding occurs in late summer and early autumn, from February to April. Males
will seek out female-led sounders by scent, and will actively compete for females.
Males fight by biting and goring with their tusks, skin is thickened at this
time of year to resist severe injury. A single male will mate with multiple
sows, and after the mating period is over, he will leave. Gestation is up to
140 days, and nest-making starts in anticipation of this. The litter size varies
by how healthy and how old the sow is, but it can be up to 10 piglets. Newborns
weigh up to a kilogram, and compete for the best suckling. Piglets start following
their mother away from the nest at two weeks; lactation is up to 3 months, but
piglets star foraging at about 4 weeks old. Striped juvenile pattern fades to
adult coloration after about 3 months. Lifespan is 12-15 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Poaka-wai
(Australochoerus riparius)
Order: Even Toed Ungulates (Artiodactyla)
Family: Pigs (Suidae)
Habitat: Rivers, lake margins and marshes of North Island, New Zealand.
The domestic pig has followed Mankind to nearly every corner of the earth, in
the Holocene, so valued is it for its meat. Feral populations thrived, and gave
rise to many new forms in the Neocene, including in New Zealand.
One such form is the Poaka-wai, a large, semi-aquatic pig found in the rivers
and marshes of the North Island, taking its name from Maori words for “pig”
and “water”. In body shape it resembles a tapir, weighing about 250 kilograms
and having a large head with a long snout, disk-shaped rooting nose is absent,
and the animal has prominent lips with whiskers. In the shape of head and lack
of oversized tusks it tends to resemble Cenozoic Anthracotheres, ancestors of
the Hippopotamus. Tusks are mid-sized, pointed and do not wear against each
other, used in defence. The toes are long and enlarged, forming a spreading,
webbed foot for walking on marshy ground, hind legs are longer than forelegs
and back slopes forward. This pig has a coat of short, coarse hair, again like
a tapir, the animal is an all-over brownish grey, with the skin around the mouth
and anus being pinkish.
In habits, it is semi-aquatic, feeding on submerged vegetation and also plants
along the water margins, preferring water-weed, leaves and stems as opposed
to reeds and grasses. It is able to “walk” along the riverbed like a tapir or
hippo, but can also swim, as with all pigs. Though mostly solitary, females
may remain with their piglets until they are a third of adult size. They are
active mostly in the morning and evening, staying cool in the water in the high
heat of the afternoon. When surfacing they may produce a grunting “blow” of
spent air somewhat like a hippo does, and while feeding on land can make a soft
grunt, distressed animal makes a loud squeal.
Breeding occurs during summer rains, from November to mid January, females will
spend more time on land, and advertise receptiveness by producing a musky smell.
Males will compete actively over females, wrestling and shoving with their heads,
but not using their tusks. Males will also display by uprooting small trees
and shrubs and throwing them about, mating is a protracted, occurring multiple
times. The mother will find a suitably dry spot in which to make her nest, which
is a shallow scrape in the ground lined with dead leaves, gestation is about
140 days. Only 4 to 5 young are born, unlike its domestic ancestor, and they
are relatively better developed at a young age, having a faded striped colour.
Young are able to follow their mother in the water after a week, and are suckled
for about two months. Whelps start feeding on their own at about 5 weeks, and
sexual maturity is reached by the females after one year, and males after two
years. Life expectancy is up to 16 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Yesin
(Yesin bengalensis)
Order: Even-toed ungulates (Artiodactyla)
Family: Shevrotains (Tragulidae)
Habitat: coast of bay of Bengal and Jakarta Coast, mangrove forests.

Picture by Tim Morris
In human epoch people of Myanma (Burma) believed in
existence of tiny aquatic elephant. They named it as “yesin” and attributed
to it some unusual
properties: poisonous bite and mystical power above common elephants. In
opinion of some cryptozoologists, the real animal was behind a myth. But, unfortunately,
the mankind had died out, not having found out the truth.
Neocene yesin, the unusual representative of shevrotains, a family of primitive
artiodactyls, is named after this mythical character. As well as mythical
yesin, it resembles tiny elephant a little, leads semi-aquatic way of life
and lives
also in territory of former Myanma.
Appearance of yesin is rather original: in constitution this animal is vaguely
similar to tapir, but is much smaller than it is, and is built not so heavy.
Yesin reaches 130 cm in length; its height at a shoulder is about 60 cm and
weight is about 40 kg. The muzzle of yesin is extended as a proboscis used
at diving as a respiratory tube. Both at males and at females from the upper
jaw impressive canines grow: at males they reach the length of 15, at females
– of 10 cm. These canines not only allow to dig silt and sand in searches
of food, but also serve as a weapon. Yesin it is capable to wound seriously
any
animal attacked it, and males use canines for courtship tournaments. Ears
of yesin are of medium size, rounded and mobile. At diving they can fold
along
the head and nestle against it closely. As the adaptation to aquatic way
of life the special structure of hoofs serves: they are wide and have a membrane
between them, and allow the owner to swim and to walk on fenny ground perfectly.
The animal is covered with short wool of chocolate-brown color with light
yellow “apron” on throat and breast. The lower side of short tail is white
– it is
a mark which the female uses for communication with cubs. Cubs have masking
albesent strips and spots on body.
Yesin is secretive animal leading mainly twilight way of life. This animal
spends daytime in dream among rich roots of mangrove trees. It repulses possible
enemies with the help of advanced canines, but quite often seeks safety in
flight, diving and hiding under water for a long time.
Yesin is omnivorous, but prefers food of animal origin. The basis of its
diet is made of seafood – fish, crabs and molluscs which it catches in water
or
digs out of sand and silt during outflow. Also this animal digs clutches
of reptiles at sandy coast and ravages bird nests made on the ground. Also
it
does not disdain carrion. The small part of yesin food is made with vegetative
food: algae and fallen fruits.
Only for the pairing period these animals, usually not tolerating the presence
of relatives, meet each other: males arrange tournaments, fighting for females.
Usually these tournaments pass without blood and are limited to display actions.
Males inflate proboscis and puff air out through densely closed nostrils,
uttering high squeak. Thus they open mouth wide and show canines to the contender.
If
rivalry becomes especially sharp, contenders attack against each other and
strike each other by lateral impacts of head. Thus canines only slightly
touch skin of contender. Biting the contender, yesin male only bites its
skin by
forward teeth. But protecting itself from predator, yesin sticks points of
canines into skin of enemy.
The female gives rise to one, and in rare cases to two cubs depending on
mother until they will reach the age of 6 months. The female brings posterity
not
more than once per year. Young animals become sexually mature at the age
of two years; life expectancy in nature does not exceed 30 years.
This species of mammals was discovered by Simon, the forum member.
Deersimil
(Cervops platycornis)
Order: Even-toed ungulates (Artiodactyla)
Family: False deer (Neocervidae)
Habitat: savannas and light forests of Southern Africa.
After extinction of significant part of large hoofed mammals their place was
occupied with representatives of other groups of mammals: in Palearctic there
are large running descendants of lagomorphs, and in Nearctic and Neotropic
areas – descendants of South-American rodents. Only south of Africa appeared
rather isolated from the Sahara savanna by woods and mountains. Here again
species of herbivorus mammals have remained relicts of Holocene dominant: various
hoofed mammals, descendants of small antelopes and chevrotains.
Occupying ecological niches exempted after mass extinction they turned to species
similar to well-known in human epoch antelopes and deer. At some species even
big horns have appeared – it is a rarity among Neocenic herbivores.
The deersimil is not the most numerous species among South-African hoofed mammals.
It keeps in small herds sometimes together with other species of herbivores.
This animal is the descendant of small and short-legged African chevrotains
(Tragulus). Having remained out of competition from the side of bovines chevrotains
roughly have started to evolve having formed the separate family of false deer
– rather large swift-footed hoofed mammals.
In comparison with ancestors deersimil is a sizable animal: its growth at a
shoulder is up to 1.5 m, body length is up to 1.6 m. By constitution it resembles
small antelopes and gazelles. This is one of most swift-footed hoofed mammals
of Neocene: at short distances deersimil accelerates momentum up to 80 kms
per hour. Because of long legs usual pace of this animal is an amble but deersimil
develops maximal speed running gallop.
Colouring of deersimil helps it to hide among vegetation: body is covered with
chestnut-brown wool; on groats and hips there are thin vertical strips of yellow
color. Stomach, bottom part of neck and head sides are white. Cubs are colored
lighter: the basic colouring at them is straw-colour with longitudinal brown
strips on body (by colouring they are similar to boar piglets); on groats and
hips strips are bent downwards. In process of growth striped pattern on the
body vanishes and on hips becomes more contrast.
This animal has horns on head. But as against deer horns deersimil horns are
constantly growing (not dropping as at deer), shovel-like and with several
jags (“antlers”). These horns are a little similar to horns of African prehistoric
giraffes Sivatherium. Bases of horns are enough strong and extended along of
skull; horns grow on head of animal in parallel to each other. Horns of females
resemble more longitudinal crests with two jags at back edge. At males horns
are larger and it can be up to 5 - 7 jags on them; horn tops at males slightly
inclinate in sides. New jags appear on forward part of horn: at old males horns
are sometimes similar to rooster crest. Similarly to deer deersimil uses horns
as the tournament weapon - opponents approach to each other so that horn jags
get in dredgings on contender’s horns. At the duel basically a power struggle
takes place – contenders try to push aside and tumble down each other.
From senses at deersimil hearing and sense of smell are most sharply advanced.
Other herbivores use it watching for behaviour of these animals for detection
of probable danger. In case of alarm the deersimil utters special shout - sharp
whistle warning of neighbours and other animals grazed near about danger. Rescuing
from predator deersimil hopes for the speed in many respects - few
predators can catch up adult animal full of forces. Even two-day cubs are
already almost equal in speed to adults though, certainly, they get tired much
faster.
Deersimil eats mainly graminoids. Grazing animal bites off only their tops;
after feeding of this animal other species of herbivores can find enough food
at the same place to themselves. Especially deersimil likes cones of graminoids
in condition of wax ripeness: thus some graminoids differ in sweetish taste.
Usually deersimil feeds during dawn and sunset and spends hottest time of day
in tree shadow.
For protection from overheat deersimil uses the original adaptation – its own
horns. As against deer horns they are always combined by alive growing bone.
Outside of breeding season skin on horns is plentifully penetrated with blood
vessels serving as fine radiator: blood emits heat to air. But in breeding
season because of hormonal influence blood vessels in basis of horns are narrowed
and substantially block stream of blood to horns that protects males from blood
loss at casual wounds.
Breeding season at deersimils begins at the end of rain season. Between males
there are short skirmishes for “harem” of several females (depending on physical
condition male can declare rights to 4 – 10 females). Opponents usually avoid
to enter direct fight and all can be limited to demonstration of force: males
bellow bassy, dig ground by hoof, widely open mouthes and show teeth. Sometimes
they rear and showing to each other the might make some steps on rear legs.
Usually one of contenders leaves stadium without fight.
Time of pairing proceeds only about one week and then aggression of males is
reduced also they become tolerant concerning to each other again feeding sometimes
side by side.
After dry season female gives rise to one or two cubs (about half of females
fawns twins). As at all hoofed mammals cub is born advanced and in some hours
it is capable to follow mother. First two months it feeds by milk but from
fortnight age it already tries plants copying mother. To the end of rain season
it becomes independent, but stays in herd. Sexual maturity at females comes
at the age of one and half years, at males at two years.
Sunhorn deer (Heliocervus heliocornis)
Order: Artiodactyls (Artiodactyla)
Family: Deer (Cervidae)
Habitat: eastern part of North America, light forest and bushes in plain and
foothill areass of Appalachian Mountains.

Picture by Meh
Till whole human epoch, since prehistoric times, deer were
traditional object of hunting. Chasing of these animals had resulted in their
preservation only
in few places of their former area. In Northern hemisphere these places represented
remote northern grounds, Siberia in Eurasia and area of Rocky Mountains in
North America. After the ending of human epoch some small deer species preserved
in forests of Southeast Asia. These animals became ancestors of new species
of deer of Neocene epoch.
North American deer are descendants of white-tailed deer (Odocoileus virginianus)
– one of the most usual species of the family at this continent. One of descendants
of this species is magnificent sunhorn deer. It is medium-sized deer – adult
male weighs about 100 kg, female is much smaller. For this one gracile constitution
is typical – this deer is able to run quickly. Sunhorn deer lives in light
forest areas where bushes alternate with sites overgrown with grass. The
basis of ration of this animal is made of leaves of bushes and low trees
and also
of rather soft grasses not belonging to graminoids.
The body of sunhorn deer is covered with wool of bright yellow color; dark
longitudinal strip stretches from the nape up to the basis of tail. Occasionally
at separate individuals in adult condition attributes of juvenile colouring
keep – white spots on shoulders and hips. Stomach at females is white. At
males colouring of stomach is quite another – it is white with numerous black
spots.
This colouring is used by males for display in courtship season. The bottom
part of tail at individuals of both genders is white. Vertically rised tail
making seen a wrong side appreciable from apart is the traditional alarm
signal at deer.
Deer are an excellent example of action of sexual selection. Various species
of these animals during the history of this group developed horns of various
sizes and shapes. Extreme degrees of expression of consequences of action
of this kind of selection are fossil deer Megaloceras with huge horns and
Eucladoceras
on which fan-shaped horns more than ten antlers developed. Sunhorn deer had
actually repeated an evolutionary way of fossil deer Eucladoceras – its horns
have completely lost function of the tournament weapon, and have turned to
object of courtship display. The shape of horns of this deer has determined
its name. Horns of sunhorn deer have set of antlers which grow practically
in one plane. In the basis of such horn there is wide semicircular “shovel”
as at the elk, but turned vertically. On its edge numerous straight antlers
grow, because of what both horns of this deer create the impression of stylized
solar disk with beams. At adult males it may be up to 8 – 11 and even more
antlers on each horn. In courtship season males avoid combat, being limited
to displaying of horns. Females of this species are polled; it is typical
condition for all deer, except for odd-looking skewhorn from
Greenland.
Within summer season males of sunhorn deer concern to each other tolerantly
and even form barchelor herds numbering up to 20 individuals. The courtship
season begins in the middle of an autumn. At this time horns completely ossificate,
the rests of skin fall down from them, and deer males become aggressive.
Males of this species gather at “stadia”. They display themselves to contenders,
rearing. Keeping balance, male can make too many steps in vertical position.
Thus spotty colouring of its belly becomes especially good seen. Usually
males
compete; they try to keep longer a vertical position – this way the strongest
one is revealing among them. If rivalry is critical, males can pass to active
struggle against contenders: they push each other by chest, trying to tumble
the contender down. Occasionally they strike impacts to each other with the
help of forelegs.
The winner male gathers a harem of 5 – 8 females and within several days
repeatedly couples with them. He furiously rushes to every one, in which
it sees the contender
(even to animals of other species), and during the courtship season eats
almost of nothing. To the end of courtship season male is strongly exhausted,
and
some males even perish, including from a nervous exhaustion. In winter males
mew horns.
Pregnancy lasts about half-year, and cubs (usually twins) are born in spring.
Juvenile colouring of sunhorn deer is spotty: cub is darker, than adult individual,
and on brownish background of its wool white spots are scattered forming
faltering longitudinal strips on back and sides.
To an autumn young deer leaves mother, and at this time she is ready to new
pairing. At the second year of life young females take part in breeding season
the first time. Males can take part in courtship tournament only since the
fifth year of life. For the second year they grow primal small horns which
have few antlers and no “shovel”. Each new year of life horns become larger,
and at five years get the shape characteristic for horns of adult animal.
Maned
spirocervus (Spirocervus jubatus)
Order: Artiodactyls (Artiodactyla)
Family: Deer (Cervidae)
Habitat: Himalayas, mountain forests.
In human epoch the majority of large animals had been exterminated as a result
of hunting, or their areas and number had been strongly reduced because of destruction
of habitats. As a result after human disappearance the majority of large animals
had died out, not having left descendants. Their place was occupied by large
descendants of more successfully survived small species of animals.
Asia was the most densety populated continent of human epoch. This circumstance
became the reason of practically general degradation of natural ecosystems and
extinction of large animals. But in Neocene this continent appeared populated
by large animals again. One of such species is the descendant of small barking
deer (Muntiacus muntjak) which lives in Himalayas. This animal is equal to large
deer of human epoch in size (height at a shoulder up to 120 sm, weight over
150 kg) and is the approximate analogue of bezoar goat of human epoch. Similarity
is strengthened even more by the shape of horns of this deer – they have no
characteristic branchy shape and are mostly straight with spirally twirled edges.
This feature has determined the name of animal – spirocervus (literally “spiral
deer”).
Spirocervus has robust constitution; its rear legs are little bit longer than
front ones, and back is inclined forward. It is the prominent feature of herbivores
living on hillsides – it is easier so to graze. Also at spirocervus there are
strong hooves behind which the elastic small pad is located. Hooves can easily
move apart. Due to such features of structure of legs the animal is able to
move on abrupt stony slopes though prefers to live in mountain forests.
The high mountains differ in rather cold climate. The alternating of seasons
is clearly expressed here, and it is cold weather till the significant part
of year. Spirocervus is adapted to such features of mountain climate. The thickset
constitution reduces a relative surface of its body. Brown colored wool of this
animal is long and rich. In cold months it becomes thick, but does not change
color, as at snowlopper from
the number of hoofed lagomorphs, living higher. On breast and neck of spirocervus
males long mane grows, and tail is ended with magnificent switch of white color.
Head of spirocervus has odd appearance. On head of this animal two longitudinal
bone crests from nostrils up to the bases of horns grow – it is a characteristic
attribute of barking deer descendants, including massive bulldeer
of China swamplands. At the female bone crests are less expressed, and horns
are lack. And horns of the male have very unusual shape. They consist of two
antlers – the basic one growing upwards, and lateral one directed aside and
outside. The basic antler is straight; at the adult male it reaches the length
70 – 80 cm. It is three-edged in cross-section and is slightly twirled spirally.
Lateral antler is short, thick and hook-like. It serves for fixing horns at
courtship tournaments.
Bone crests pass to bases of horns. They are covered with long wool of yellowish-brown
color at males and grey at females. At males wool on bases of horns is so long,
that it seems the continuation of mane.
Spirocervus precisely differentiates habitats with snowlopper. It avoids places
where snow lays till the most part of year, and does not meet higher in mountains
where the snowlopper lives. Usually spirocervuses keep in herds of 10 – 15 animals
– females and young animals of both genders under leadership of the adult male.
Sometimes in large herd it may be two adult males. These deer live in mountain
forests and feed on leaves of bushes and low trees. In winter when leaves fall
from most part of plants and forage reserve of animals is reduced, spirocervuses
may eat leaves of evergreen rhododendrons which they usually do not touch in
summer.
In the beginning of an autumn the courtship season begins. At this time males
roar loudly, declaring rights to territory and females. Bachelor males (usually
these ones are young animals) challenge males, interfering to their territory
and roaring. Between competing males there are severe duels for harem. They
are reduced to force struggle of animals linked by horns. Lateral antlers on
horns serve as terminators, not allowing animals to injure each other; therefore
males usually go through this time without damage.
Pregnancy at the female lasts about half-year, and she gives rise only to one
well advanced cub. Such tactics is more favourable in conditions of limited
resources. Cubs of spirocervus differ from adults in dark, almost black colouring,
with several small white spots on throat. In the course of time they brighten,
and the wool changes shade to brown color. To winter the young animal becomes
almost independent. Females are ready to breeding at the third year of life.
At males at the second year of life simple forked horns with almost equal basic
and lateral antlers grow. Next years horns change the shape, in brief repeating
an evolutionary way of ancestors of this animal – vertical antler becomes longer,
and lateral one grows thick.
Life expectancy makes about 40 years.
Meganesian
False Moose, Samboose (Paludicervus altus)
Order: Even Toed Ungulates (Artiodactyla)
Family: Deer (Cervidae)
Habitat: Swamp-forest, marshes and vegetated lake margins of Carpentary and
Arafura lakes in northern Meganesia.
The urge to hunt trophies has caused man to propagate various large ungulates
outside of their natural range. Deer have been introduced into many places they
did not traditionally live, including New Zealand and Australia. In the north
of Meganesia, deer still thrive due to this, and some have become adapted to
the vegetated margins of the great sea lakes there.
The Meganesian False Moose is a very large deer, almost as tall as a true moose
and weighing up to 500 kilograms, with males being larger than females. They
have a massive body; humped shoulders, and long legs, feet are large with splayed
pointed hooves to aid it in walking on marshy ground. Colour is a general dull
brown, with darker parts along the spine and bases of the feet, a short mane
tops the shoulders. The snout is long and tapered, with a large nose, the lip
forms a short overhanging proboscis that aids it in gathering food, the antlers
can reach a span of 2 meters, and are palmate much like a moose, with as many
as 16 points.
This animal is an ecological analogue of a moose, but in a tropical setting.
It will consume low-growing vegetation as well as the young shoots of trees
and other waterside vegetation, twigs and bark, they frequently dredge the shallows
of the lake for water plants of various sorts, fallen fruit is also eaten. Food
is selected based on nutrient level as opposed to bulk, they are selective browsers.
Though found on the margins of both Carpentary and Arafura lakes, this animal
still needs to drink fresh water and will travel some distance to find it if
it needs to. Both males and females are aggressive if threatened by a predator
or rival, and will charge at threats, only the males have antlers, which are
regrown each season. These deer are mostly diurnal, and generally travel alone.
Rutting and mating occurs at the end of the dry season from September to November,
this coincides with the flush of new growth. At this time, rutting males become
sensitive to the smell of the does urine, males and females start calling to
each other, the rutting call of the male is a typical stag roar. Males will
congregate around females and compete to mate with them, females select mates
based on the size of antlers and males will display antlers vigorously in order
to compete for females. Stags will sometimes be driven to duel and lock antlers
in combat, with the weaker backing down. Gestation of the pregnant doe is 8
months, giving birth to a single large fawn. Fawns are a lighter colour than
their parents, and bear a weak spotted pattern, as with many deer, the newborn
will initially be hidden in a nest of thick vegetation until it is able to follow
its mother. The young will stay until the following year when the next fawn
is born. Lifespan is up to 20 years, and only the young are ever preyed upon
by marsupial panther and
yagil.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
White-headed
rapidocervus (Rapidocervus albocephalus)
Order: Even-toed ungulates (Artiodactyla)
Family: Deer (Cervidae)
Habitat: plains of North America – prairies and semideserts.
In human epoch in North America isolated populations of white-tailed deer (Odocoileus
virginianus) were kept. In epoch of Holocene – Neocene congelation they lived
in foothills of Rocky Mountains, in areas of humid enough climate, but far from
edge of glacier. These populations became ancestral stock for deer of Neocene
epoch, inhabiting North America.
Among American deer of Neocene epoch a special genus evolved, which representatives
live in open area and have ability to very fast running. These animals are rapidocervuses,
original analogues of pronghorns of Holocene epoch.
Rapidocervuses represent the small genus of deer including the forms living
in open areas – from grassy plains up to semideserts. These animals are remarkable
in their gracile constitution and long legs with strong hooves. Rapidocervuses
are high animals (over 1.5 m at a shoulder) and have long neck. Head at these
animals is short, and jaws are strong, adapted to feeding on rigid grassy vegetation.
Eye-sockets are shifted upwards and are placed at small bony eminences formed
also by antler pedicles.
Similarly to all deer species, rapidocervuses shed antlers every year. Antlers
of these deer are poorly branched, and because of it animals look like gazelles
or antelopes from apart. Usually antlers of adult male have no more than three
branches. The main branch of antler is directed back; antlers have lyre-like
shape with tips slightly bent in sides. The second large branch grows from the
basis of antler and is directed forward, and on the basic branch there is one
small branch directed upwards and inside. Females of rapidocervus species are
hornless.
Typical species of group is white-headed rapidocervus living in western part
of Great Plains, in dry area located in “rain shadow” of Rocky Mountains. It
is a large species of genus; growth of adult male at a shoulder is about 160
cm. Female is smaller compared to male – it typical for all species of rapidocervus.
At white-headed rapidocervus head is almost entirely white, except for top of
head and nape. White color also is stretched to sides and forward part of neck,
breast and stomach. Nape, back part of neck, shoulders, back, hips and crupper
of animal are yellowish-brown; tail from above is black, and white from underside.
White-headed rapidocervuses live in plains in herds numbering up to 50 ones.
They avoid thickets of bushes and trees, and any obstacles for run. Being frightened
by predators, white-headed rapidocervuses, and also other large species are
capable to accelerate momentum about 70 kms per hour, and small ones – up to
80 kms per hour on short distance. Rapidocervuses do not compete to deermaras,
cursorial rodents of South American origin, due to difference in habitats –
rapidocervuses are steadier against cold, live in northern areas and live in
deserted regions to the west from plains. Besides they eat more rigid grasses
uneatable for these rodents.
In rapidocervus herd there are approximately fifty-fifty males and females of
various ages. The hierarchy in herd is well expressed – large male is a leader
of herd. At meeting of two herds males try to establish leadership, rearing
and striking each other impacts by front legs. Heat at this species takes place
in the beginning of winter months, and the posterity is born in early spring.
At white-headed rapidocervus female gives rise to one fawn, but approximately
20 % of females give birth to twins. Calf has spotty colouring, as at deer in
human epoch. At change of colouring in places where there where white spots
at adult animal, spots of juvenile colouring start growing and merge with each
other. On one-colour sites of body they gradually decrease in size and vanish.
At not sexually mature individuals head is dark, and only throat and tip of
muzzle are white.
In territory of North America some close species of rapidocervuses live:
Grassland,
or striped rapidocervus (Rapidocervus zebratus) – medium-sized
species: growth of male at a shoulder is about 120 cm. At this species antlers
have only two branches. The basic branch of antler is directed back and upwards
a little, and on one third of its length, measuring from the antler basis, the
second stag branches at small corner to the main one and reaches about the same
lengths. Shoulders at this species are little bit higher then crupper, and neck
is rather short. Colouring of grassland rapidocervus is very rare for deer:
on yellowish-brown background dark narrow vertical strips stretch, being especially
dense on back part of the body and on neck. Lower side of tail is white.
It is the most “water-loving” species of this genus: it lives in well humidified
plains in eastern part of area of these deer, but keeps in the distance from
bushes and trees. For this deer run by jumps up to five meters long is characteristic.
Crowned,
or short horned rapidocervus (Rapidocervus coronatus) inhabits
dry areas of Mexican plateau. It is the large representative of genus: growth
at a shoulder of male reaches 140 – 150 cm. This species differs in colouring
of head appreciable from apart: from upper lip through an eye up to the basis
of ear wide black strip stretches; it is present at individuals of both genders.
Below this strip the narrow white strip lasts. The basic colouring of body is
monophonous sand-yellow with darker strip along the back; stomach is bright
yellow. Antlers of this deer are rather short and carry three branches directed
accordingly back, sideways and forward. All branches approximately one length
and their ends are slightly inflexed, because of what pair horns is remote is
similar to a crown. The bases of antlers are strongly expanded and covered with
large corneous lumps – “pearls”.
For improvement of heat emitting at crowned rapidocervus the plica of thin skin
on breast is developed, and ears of this species are rather larger, than at
related species. This species has interesting behavioural feature – it is able
to receive water from cactuses, breaking stalks of these plants by hoof.

Picture by Fanboyphilosopher
Badland,
or donkey-eared rapidocervus (Rapidocervus asellinus) is the
smallest species of genus: height of adult male at a shoulder is about 60 cm.
This species lives in droughty “badlands” kept along Rocky Mountains ridges.
The gracile constitution and plica of skin on breast help to radiate the superfluous
heat. But more effective bodies of heat emitting are auricles. They are long,
narrow and mobile as at hare, and their tissue is plentifully penetrated with
blood vessels.
Antlers at badland rapidocervus are advanced rather poorly. They have only two
branches – larger one is directed back, and shorter one, sometimes having doubled
top, is pointed forward. In constitution this species is very similar to gazelle.
At this species lips are covered with dense skin: it is an adaptation for feeding
on cactuses. Badland rapidocervus eats prickly pears covered with tiny hair-like
prickles and because of it inedible for other herbivores, except for insects.
Eating round stalks of cactuses, or simply looking around, this deer can stand
on rear legs for a long time, supporting against any firm subject, or without
a support at all. In diet of this species there are also dry grass and insects.
In spring these deer willingly eat even poisonous tubers and bulbs.
Colouring of wool of this deer is monotonous yellowish-brown. As an element
facilitating the recognizing of congeners at these deer there is only black
tail with fluffy white inside.
For badland rapidocervus the low requirement for water is characteristic. It
receives water from cactuses and excretes very concentrated urine containing
small amount of water.
Aotearoan
Wood-deer (Sylvadama novazealandiae)
Order: Even-toed Ungulates (Artiodactyla)
Family: Deer (Cervidae)
Habitat: Lowland forests, woods and bush, North and South Islands of New Zealand.

Picture by Alexander Smyslov
Deer had become a characteristic part New Zealand landscape
during the human occupation of the Holocene, being introduced for the purposes
of game hunting sport. Descendants of these animals remain in Neocene, and have
given rise to large and small forms. One kind, descendant of Fallow Deer (Dama
dama) became an exclusive inhabitant of woodlands and other well-vegetated areas.
Aotearoan Wood Deer is a fairly small kind of deer, converging somewhat upon
a Muntjac in body shape, but without tusks. Adults reach up to 65 centimetres
tall at the shoulder and 35 kilograms in weight, and males possess short, sharp
2 pronged antlers, ears are large and prominent. Body colouring is an all-over
reddish brown, retaining a juvenile pattern, rows of white spots. Face, nose
and feet are darker brown colour, tail and rump is white, used in signalling.
Coat is soft and dense, but thicker in the colder parts of its range, the South
Island. Facial glands are used to mark territory, and are larger in males.
A diet consists mostly of the leaves of trees and bushes, but also occasionally
bark, soft grasses and herbs, as well as fallen fruit. These animals are active
mainly during the twilight as well as late afternoon; they are generally shy
and will flee at the first sign of danger. Call is a whimpering bark when alarmed;
breeding males may make a high nasal trumpeting call to attract females. These
deer are mostly solitary, but several individuals may exist within one territorial
area, and they are generally tolerant of one-another.
Individuals reach sexual maturity at 2 years; females have more than one oestrous
cycle during the year, but prefer to breed when food is abundant. Gestation
lasts up to 7 months; fawns are sheltered in dense undergrowth for a week, and
grow quickly, leaving the mother after 6 months. Males are polygamous, and a
form small harem of females, fighting consists of displays where males thrash
and uproot plants with their antlers, males will lock antlers and attempt to
exhaust one another.

Picture by Alexander Smyslov
Related form, exclusive inhabitant of the dense sub-tropical woods of the northern part of North Island, is the Pygmy Wood Deer (Sylvadama nana). It is distinguished from its relative in much smaller size, 40 centimetres tall, and only 10 kilograms in weight, with underdeveloped antlers, single short, horn-like points. Coloration is darker, and spotted pattern is yellowish. This animal is a shy inhabitant of dense forest, and feeds selectively on low understory growth, leaves, fungus, stems, and fallen fruit, sometimes insects and small vertebrates are taken. Young is born larger and better developed than its relative.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Bald
rambull (Pachycephalovis grandis)
Order: Artiodactyls (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Himalayas, Plateau of Tibet, Altai; mountain woods and meadows at height
up to 4 – 5 thousand meters; in the north of area animal goes down, but does
not meet in plain regions.
At the boundary of Holocene and Neocene the variety of ungulates had sharply
reduced. The reason of it had been mainly human activity/ People had overpopulated
the planet and had destroyed habitats of many species. Agrarian landscapes changed
by people, where herds of domestic cattle were analogues of wild animals, became
the prevailing landscapes in many places of the Earth. After human extinction
the majority of domestic animals had also disappeared, but some species able
to adaptation, had survived and became ancestors of new forms of Neocene ungulates.
Among them there were primitive breeds of domestic sheeps which easily ran wild
and became a part of poor communities formed at the place of collapsing agrocenoses.
Many descendants of domestic sheeps live in Neocene New Zealand, but separate
species had evolved in other parts of the world.
In mountain areas of Asia large artiodactyl – bald rambull is found. Height
of this animal at a shoulders is up to 170 cm at body length of about 2,5 m.
Externally it is similar to golden goat antelope (Budorcas taxicolor), the original
representative of bovids, related to sheeps, lived at the territory of China.
The body of rambull is covered with rich shaggy wool of gray-blue color, on
back the longitudinal strip of darker wool stretches; the tail is covered with
long wool and is similar to horse’s one. Ears are moderately long and covered
from external side with wool.
As against New Zealand sheep
descendants, bald rambull has almost completely
lost horns. Instead of it the upper part of skull at this animal is strongly
thickened. The skull of male has the convex top representing a continuous bone
outgrowth about 15 cm thick. Males use skull for courtship tournaments during
which they put each other strong ramming impacts. Thus the massive cover of
skull protects brain of animal from concussion. On top of the head of animal
the skin is dense, thick, strongly cornificated, colored dark grey, completely
lack of hairs. Near ears of animal there are outgrowths hided in wool, also
covered with cornificated tissue – they are rudiments of horns. Females lack
of them more often. Also at females the top of skull is not as convex, as at
males, and they do not use it for fierce tournaments. Only occasionally females
strike each other easy impacts by head, reminding the subordinated individual
about their own position in herd hierarchy. Cervical vertebrae are adapted to
maintain the sizeable loadings.
The thick ossificated top occupies the most part of volume of skull. The brain
of bald rambull is very small, and the behaviour of animal differs in primitiveness.
Weakness of sight is compensated, however, by keen hearing and sense of smell,
and also by natural care. Animals move across their territory along strictly
determined routes, and only natural acts like avalanches or earthquakes force
them to search for new ways. This species of ungulates lives in small herds
including one dominant male, several females and their cubs of the age of up
to two – three years. In herd the not strict hierarchy is established.
Bald rambull eats leaves of undersized trees and bushes. At lack of habitual
forage it can eat even branches of coniferous trees. Also the animal is able
to dig snow in searches of grass and evergreen bushes. This species lives in
conditions of seasonal climate – winter in habitats of bald rambull is frosty
and snowy. Therefore right before colds it grows long rich wool of lighter color,
rather than summer one.
Pregnancy lasts 11 months; usually the female gives rise to one cub. In favorable
years twins are often born – one case to approximately 8 – 10 pregnant females.
Cubs are fed on very fat milk and quickly grow. At the age of about 8 months
the young animal completely passes to forage of adult animals. At young animals
approximately up to one-year-old age the head is covered with wool, as at other
representatives of family. At the age of four years animals completely develop
physically, and females already can bring posterity. Young male reaches the
blossoming of physical strengths to the seventh year of life, and supports this
physical condition at least till 15 years.
Sylvammon
(Sylvammon gracilis)
Order: Even-toed ungulates (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: evergreen woods of Northern island of New Zealand.
Forests of New Zealand had lost because of human destructive activity large
moa birds in Neocene had found new herbivorous inhabitants. Descendants of
sheeps, domestic animals introduced to New Zealand became them. In Neocene
when the human species had disappeared and set of new ecological niches had
appeared descendants of this domestic animal had started to evolve roughly
having evolved in conditions of island isolation to some species differing
by ecology.
The sylvammon is large species of artiodactyls living in woodlands and forests
of New Zealand. Withers height of adult male is up to 150 – 160 cm. By proportions
this species is a little similar to deer.
The long neck is covered with mane of lengthened hairs. At old males mane hangs
down almost up to knees of forward legs. Shoulders of sylvammon are higher
than crupper and back of animal is sloping. Due to such constitution animal
can browse leaves from trees at height up to 2 meters. Due to need animal can
even rear on back legs. Animal can run quickly and jump high.
Horns are similar to goat’s ones; at females and young males they are straight
and only at old males can bend semilunarly. Bases of horns are bulged and also
pulled together, and horn tips are directed back – in this case they do not
prevent to move in dense wood. Among females hornless animals appear frequently.
Horns at males are lighter than at females.
Wool of sylvammon is short and rather thin: animal lives in rather warm climate
not rising highly in mountains and not coming to southern areas of New Zealand.
Except for mane long hairs grow on tail of this animal more similar to horse’s
one. Colouring of body of sylvammon is dark-brown with white stomach and white
“glasses” around of eyes.
It lives in woods preferring to graze at sites where bushes and young trees
spread off: on marges and near river banks. Frequently it goes to plains overgrown
with bush. In wood animals have favourite tracks adhering them at movement.
Sylvammons have kept characteristic for sheeps gregarious habit of life. Herds
of these animals total up to 20 individuals and consist of several breeding
groups including male and three – four females. In forest herd moves having
stretched in line where ahead there are dominant individuals. The rank of females
in herd is determined by rank of the male which group they belong.
Due to change of habit of life proportions of animals have changed that was
resulted in their behaviour. More graceful constitution has made impossible
struggle by ram heads impacts characteristic for ancestors. Males sort out
their relations striking each other lateral impacts by head. Crushing blows
characteristic for rams were transformed at these animals to demonstration
of horns. The sylvammon male shows itself to the contender having bent head
down. Thus horns are lifted upwards that visually exaggerates his size. At
this time mane on neck of the male is fluffed. Males rear on back legs and
can make some steps in such position for strengthening of the impression created
at the opponent.
At any season in sylvammon herds it is possible to see cubs. Regularity in
breeding at this species is not present but most part of cubs is born in spring
(in October – November in Southern hemisphere). One cub usually is born though
about a quarter of all females eans twins. Cub is born well advanced as at
all hoofed mammals. At one hour after birth it already can walk and at the
second day of life does not lag behind herd.
Great goatlope (Gravicaper magnus)
Order: Artiodactyls (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Zinj Land, savannas and light forests at the west of subcontinent.

Picture by Alexander Smyslov
In human epoch ungulates of bovid family had suffered the
great damage. Many of their species had been objects of hunting, and practically
all wild species
had suffered from destruction of inhabitancy and the expansion of territories
occupied for agriculture. Subsequently the significant part of these grounds
had undergone to desertification, and began even less suitable for life of
ungulates. In Neocene new groups of running herbivorous mammals had appeared,
and the variety of ruminant artiodactyls had reduced. In human epoch herds
of various wild ruminants were replaced by monospecific herds of domestic
cattle which had become extinct after people in the majority of places. But
representatives
of primitive breeds of domestic ruminants became ancestors of some species
of Neocene ungulates. At the East-African subcontinent (Zinj Land) from feral
domestic goats many kinds of goatlopes had descended; these ones became dominant
herbivores of subcontinent alongside with horned suids of Porceratidae family.
In savannas at the west of Zinj Land small herds of very large ungulates
similar by a constitution to domestic humped ox or eland antelope of human
epoch graze.
It is the most massive representative of goatlopes, the great goatlope. Its
genus name “Gravicaper” means “heavy he-goat”, and emphasizes features of
its anatomy. Evolution of goatlopes directed to increasing of body size is
limited
by presence of large horned suids at Zinj Land. In Neocene among suids a
plenty of massive running forms occupying ecological niches of rhinoceroses,
tapirs
and bulls had appeared. Therefore great goatlope is an only exception among
swift-footed gracile relatives.
Great goatlope is an ecological analogue of elands of Holocene. Growth of
this animal at a shoulder is about 160 cm, and weight is up to 800 kgs. Legs
are
rather short, and hoofs are wide. Front and rear legs have approximately
equal length, therefore back of animal is horizontal. Great goatlope runs
rather
slowly and prefers to live on plains with firm ground.
The large animal in conditions of a hot climate faces with a problem of heat
emitting. At great goatlope on the neck “hanger” develops – wide plica of
skin stretched from chin up to back edge of thorax. This skin is penetrated
with
blood vessels, and in day heat they dilate, giving to air surplus of heat.
Skin of great goatlope is covered with very short light grey wool; from apart
its wool seems velvety. At young animals wool is rather long and darker,
rather than at adults. At adult animals long black hair growing on the bottom
edge
of “hanger” form a kind of “beard” from chin up to stomach. It is especially
well advanced at males and helps to estimate force and physical condition
of the male in courtship season. On long tail there is a switch of black
hair.
At great goatlope both females and males are horned. Horns of these animals
are short and have wide bases almost adjoining on forehead. Horns are lyre-shaped,
and the annual gain adds new relief ring in their basis. During courtship
duels males fall on knees of forward legs and combat, striking impacts by
head and
neck. Rut at great goatlope takes place at the beginning of dry season.
Great goatlope eats lower parts of grasses and undersized plants, therefore
it prefers to graze near to other species which eat top parts of plants.
This species does not compete to bikifaru, the representative of horned pigs,
because
it occupies various habitats, preferring dry ones: savannas overgrown with
grass and flat hillsides with small amount of bush.
Once a year, in the beginning of rainseason, the female gives rise to one
large, well advanced calf. One hour after birth it already can walk, and
at the second
day of life it runs on a level with adult animals. Sexual maturity at these
animals comes at the age of 3 years.
Donkey goatlope (Hippocapra asellina)
Order: Artiodactyls (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Zinj Land, savannas and woodlands at the west of subcontinent.
Goatlopes evolved at Zinj Land are descendants of the common ancestor, domestic
goat (Capra domestica). Till the evolution history, occupying various habitats,
these animals changed, adapting to new conditions. It allowed them to avoid
competition and to use resources of an environment more effectively.
The highest species of goatlopes is donkey goatlope living in dry regions
at the west of subcontinent. Its growth at a shoulder reaches 170 centimeters,
and animal can browse branches at height more than 2 meters. This is quickly
running animal of gracile constitution keeping in large herds.
Donkey goatlope is easily distinguishing due to the specific colouring similar
to colouring of Grevy’s zebra (Equus grevyi) lived in human era. But the
colouring of this species is less contrast: on body, shoulders, hips and
neck on light
greyish-beige background thin and dense vertical brown strips stretch. At
old animal strips on back part of body may turn pale. At young growth colouring
is more contrast, and calves of this species seem darker. The bottom part
of
legs at animals of any age is single-color greyish-white. For recognition
of congeners donkey goatlope has dark marks on head: wide black strip at
the edge
of ears and black top part of head.
At this species horns are present at animals of both genders. They are lengthened,
twisted as at koodoo antelope, and have smooth surface. At males horns are
larger, rather than at females.
Tail of donkey goatlope is similar to horse’s one – it is short and covered
with long white hair.
Distinctive feature of this species of goatlopes is long ears. This is an
adaptation helping to be cooled. At donkey goatlope the special form of social
behaviour
is developed: as a sign of attachment to each other animals lick each other’s
ears; the female also behaves so in relation to the calf. This ritual has
practical value: saliva evaporates, improving heat emitting. Besides on cheeks
of animal
wide lobe-like hangers. If the animal becomes hot, blood inflows to them,
and they increased in size, also emitting heat in air.
Due to high growth donkey goatlope does not compete to other species of ruminants
of Zinj Land. It eats top parts of graminoids and foliage of bushes, and
can browse bottom branches of low trees. Therefore more often it lives at
edges
of small forests and thickets of bushes. This species keeps in herds including
both males and females. Such herds may total some tens animals of various
ages. Out of courtship season males are tolerant relatively to each other.
In courtship
season, which begins at the end of rainseason, they start to behave aggressively
to each other, and the herd is separated into set of harems, each of which
is jealously protected by male. At this time males start to emit very strong
“goat” smell. Males battle against each other, having widely moved apart
front legs and having lowered heads. The winner male drives defeated male
away from
harem, uttering throat bleating, and brings its females in harem. At this
moment it starts to smell especially strongly. Male of donkey goatlope “marks”
females
by original mode – he urinates plentifully on the ground, rolls in dirt,
rubring urine in wool, and then rubs against females, leaving smell of urine
on them.
During courtship season male repeats this procedure each day.
At the beginning of the next rain season the female gives birth to one well
advanced calf. In first some minutes after birth it already rises on legs
and tries to walk. At the age of three months vegetative food contains more
than
half of its diet. The female, in addition to milk, feeds calf with belched
semidigested grass, thus supplying it with unicellular organisms necessary
for life, which live in stomach at all ruminants.
Young females become sexually mature at the second year of life, and males
at the third year. Life expectancy of these animals in nature does not exceed
20 years.
Swamp goatlope (Paludicapra pseudositatunga)
Order: Even-toed ungulates (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Zinj Land, marshlands in east and southeast of subcontinent.
People left after them the impoverished and broken ecosystems, and also a significant
amount of feral domestic species of animals. During the restoration of ecosystems,
in first millions years after disappearance of mankind, till the process of
adaptive radiation many new species of animals occupying various ecological
niches have appeared. It reduces the competition between them as such species
though related to each other, differ in requirements to inhabitancy and can
coexist in the same territory, using different food sources.
In East African subcontinent (Zinj Land) in Neocene the various bovids live;
they are goatlopes, descendants of feral domestic goats. Various species of
these animals differ in size, constitution and features of inhabitancy. Many
species of goatlopes inhabit open landscapes with firm ground, but one species,
swamp goatlope, has developed life in marshlands at the east of subcontinent.
Swamp goatlope is “antelope” of gracile constitution; growth of animal at a
shoulder is about one meter. The wool of this animal is short, colored reddish-brown
with thin vertical strips of white color – such pattern helps this animal to
mask in reeds. At male long beard grows, females have only bunch of hair on
chin. Both males and females have long skin “wattles” on lower jaw. At males
ducts of specific glands secreting characteristic goat smell open in them:
oily secretions are rich in caprylic acid and serve for marking of territory
and recognition of congeners. “Wattles” of males are covered with long wool
which absorbs secretion and keeps smell during long time. At meeting animals
sniff each other’s “wattles”. To make marks at the border of territory male
rubs “wattles” against tree trunks and reed stalks.
Swamp goatlope is able to swim and walks confidently in fenny places. On fenny
ground fingers and toes of animal move apart, increasing the area of support.
Hooves of marsh goatlope are long and wide, therefore it runs rather slowly
overland. Beyond the shelters swamp goatlope feels like uncertainly and prefers
to not appear in such places. But in reeds this animal lays a network of footpaths
on which it moves across its territory. The part of footpaths is directed to
shelters – to rich thickets and to swamps overgrown with floating plants where
it is difficult for predator to pursue this animal.
Horns of marsh goatlope are pointed and long. They are similar remotely to
horns of oryx antelope (Oryx), but are not acute and conic, and twirled as
a dense spiral along an axis. This animal lives in herds including several
tens of individuals of both genders. Beyond the breeding season male behave
peacefully to each other, but in heat time there is a strong competition between
them. Males of this species fight, hooking each other’s horns, and struggle,
having kneeled forward legs.
At female two calves are born almost always. Only young females give rise to
one cub, and in one case from ten even triplets may be born. But usually one
calf from triplets quickly dies, because it does not get enough milk. Calves
differ from adult animals in darker colouring – at them white strips are narrow,
and background color of wool is brown. Young animals pass to diet of adult
animals at the age of 4 – 5 months and remain in parental herd up to sexual
maturity (at the age of 22 – 25 months).
Longhorn goatlope (Laticornella frontalis)
Order: Even-toed ungulates (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Zinj Land, savannas in the north and west of subcontinent.
During the adaptive radiation in Zinj Land a plenty of goatlopes appeared –
these ones are descendants of feral goats which have occupied various ecological
niches in nature of subcontinent. Species of goatlopes adapted to various habitats
differ from each other in size and constitution. Adaptive radiation reduces
a competition between them and allows using of resources of inhabitancy more
effectively.
Herds of herbivores in dry plains of Zinj Land are formed by various goatlopes
and cursorial descendants of pigs - large bikifaru
(Dilophoporcula rhinocerotoides).
Among them representatives of original species of goatlopes, longhorn goatlopes,
are remarkable in their special appearance. Females of this species look rather
usual, but male horns differ in original shape having analogues only at some
prehistoric species of ungulates.
The height at a shoulder of adult male of longhorn goatlope reaches 150 cm;
females are smaller a little. This animal has gracile constitution, slightly
sloping back and rather long neck. This species of animals is adapted to fast
running: despite of great size, longhorn goatlope can accelerate momentum about
70 kms per hour at the short distance.
At this species sexual dimorphism is sharply expressed. The female of longhorn
goatlope is hornless; its wool has grey color with numerous cross strips on
legs and crupper. Stomach is white; between area of white wool on stomach and
background colouring wide black strip stretches. Eyes of longhorn goatlope
female are surrounded with area of white wool which at different individuals
varies from small crescent spot at back edge of an eye up to wide irregular-shaped
ring. Beard at female is small and grows only on lower jaw.
Male of this species has darker colouring – it has wide cross strips on legs
and crupper, and on body there are scattered numerous black spots on grey background.
Head of male has appreciable black-and-white colouring: back part of head,
area near ears, nose bridge and cheeks are black, and the tip of muzzle, lips,
beard, lower jaw and area around of eyes are white. Beard at males is more
advanced, rather than at females: it grows on lower jaw and strethes back up
to throat.
Males have long horns of original shape – hence the name of species. Span of
horns reaches almost 2 meters. They grow in sides and are curved upwards a
little. At old individuals horns are slightly bent back. Such horns can not
be used as the effective weapon against predators, therefore at predator attack
males of longhorn goatlope rely only upon their own speed. In courtship season
duels of males have exclusively ritual character – males fall on knees of front
legs and “measure” the length of horns, only slightly touching each other by
forehead and touching each other’s horns. In such duels animal with the longest
horns wins though occasionally male, at which one or both horns are broken,
may win due to larger size. At males and females of this species forehead is
wide, and frontal bones of skull are thickened a little.
Because of special shape of male’s horns longhorn goatlopes avoid woody areas
and the regions overgrown with high bushes, preferring grassy plains. They
keep near to herds of larger goatlope species, avoiding thus attacks of predators.
Due to high growth, keen sense of smell and good sight they can notice approaching
of predators in time, and notify animals surrounding them on danger with loud
bleating.
Usually female gives rise to one calf; twins are born approximately in quarter
of the general number of birth cases. Calf is well advanced, able to run in
one hour after birth. It has spotty colouring which helps to mask in grass.
As they grow, young females loose spots on body, and on legs color marks merge
to strips. At males juvenile colouring is partly kept – only the part of spots
vanishes. At the age of 2 years females already can take part in breeding.
Young males also reach sexual maturity, but usually they can not compete to
older congeners. At the age of 5 – 6 years male’s horns reach the size typical
for this species.
Crested
goatlope (Sylvicaprella sylvana)
Order: Even-toed ungulates (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Zinj Land, humid forests in east and southeast of subcontinent.

Picture by Alexey Tatarinov
After human disappearance some species of domestic animals
had an opportunity to evolve on a par with wild species. Primitive breeds of
domestic goats, widespread in tropical areas of Old World, not too dependent
on people care, had been among such animals. In Zinj Land descendants of these
animals had evolved to some species of goatlopes of various sizes, differing
from each other in shape and ecology. Large species of these ungulates are inhabitants
of plains, and small species, which growth at a shoulder does not exceed 30
cm, live in tropical forests at eastern coast of subcontinent.
One species of small goatlopes is crested goatlope widely settled in the east
of Zinj Land. It is an original ecological analogue of duikers, dwarf antelopes
known in human epoch. Growth of this animal at a shoulder is about 35 cm. In
its constitution this goatlope is similar to duikers or shevrotains – it has
rather short legs allowing run quickly in underbrush and sharp changes of run
direction, when escaping from the enemy. Neck of this animal is also short,
and head is prolonged and has wedge-shaped shape and mobile lips allowing leaves
browsing. The feeding animal frequently rears on two legs and can keep such
position of body at feeding for a long time.
Wool of crested goatlope has camouflage colouring – on head, neck and sides
it is grey with olive-green shade. From nape along the back black strip stretches,
faltering sometimes. It reaches a tip of tail. The bottom part of tail and back
part of buttocks are covered with white wool. In case of alarm fluffed up wool
of tail lifted upwards is an alarm signal. On head between the bases of horns
the long bunch of rigid white wool grows, well visible from apart. Using it,
crested goatlopes notice congeners in twilight of tropical forest underbrush.
Horns of this animal are straight, pointed and short; their tips are partly
directed in sides. Protecting itself against enemy, animal makes deep chipped
wounds by horns. Males of this species, establishing hierarchy relations, butt
each other. Crested goatlope lives in herds including 10 – 20 animals under
leadership of older male. They frequently form mixed herds with local flightless
birds – dwarf species of
ostriches. It allows representatives of both species to watch world around
– crested goatlope has sharp hearing and sense of smell better, and dwarf ostriches
have wider field of view because of the growth.
The courtship season of this species is very long, and during half-year there
are the females ready to pairing in herd. Males compete to each other for an
opportunity of pairing, and dominant male appears the father of calves born
in herd in most cases. Barchelor males live lonely or in small groups. Because
there are no females near them, they relate to each other much more peaceful,
rather than in common herd.
Usually female gives rise to one calf though about one third of females can
give rise to twins. Pregnancy lasts till about two months, and calves are born
two times per one year at the same female. Calf differs from adult individuals
in darker colouring with irregular-shaped cross strips. White marks appear at
the age of 16 – 18 months when young animal reaches full development and can
take part in breeding. Life expectancy of crested goatlope does not exceed 15
years.
In tropical forests of different areas of the Earth some animal kinds very similar
to crested goatlope have appeared. In forests of Mauritius dwarf dryocaprella
lives, descending from the same ancestor, but not related to this species, and
in Great Antigua Island cursorial rodent mazamara
lives, being only convergent similar to this species. Similar conditions had
independently resulted in formation of similar features of anatomy at these
species.
Antilopine
Forest Buffalo (Bubalotragus sylvanus)
Order: Even-toed ungulates (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: Tropical rainforests of northern Meganesia.
Among the mammals introduced to Australia by man, it is the ungulates that have
become the most successful during the Neocene. In the north of Meganesia, descendants
can be found of deer, pigs and water buffalo. Some descendants of the water
buffalo became analogues of hippopotamus,
but others stayed closer to the original bovid body type.
The Antilopine Forest Buffalo has lost the stocky body shape of its ancestor,
and looks almost like a forest antelope; it is a graceful browser which reaches
1.5 meters at the shoulder and a weight of about 300 kilograms. Legs are relatively
long and slender, and the neck is of medium length, it browses on low and mid
level vegetation, mostly the leaves of trees and bushes, as well as bark and
fallen fruit. Sometimes it will use trails cleared in the forest by other animals
in order to feed, and it will frequently venture into clearings and forest margins
for the same purpose. The profile of the head is more graceful and antelope-like
than its ancestor, with a tapering snout, horns are of medium length and spiral
weakly, very sharp, and shaped like a backswept lyre. Pelt is a coat of short
hair much like that of a horse, which is dark reddish brown, extremities of
the legs and the base of the snout are whitish, ears are large like a cow, with
blond “tassels” of hair, the tail is long and ends in a blond tuft.
Individuals lead mostly solitary life, males are completely solitary but females
and young may congregate occasionally in small groups. This bovid is mainly
crepuscular, being most active at dawn and early morning, and again at late
afternoon and dusk. They are generally shy and will mostly flee danger, but
can retaliate with their horns to fearsome effect if provoked. Males will spar
and display to each other with horns when they come across one another, and
they only seek out the females during the breeding season. Disagreements among
males over females are generally resolved with sparring and posturing, rarely
do fights take place. Mating is during the dry season and the female gestates
for 10 months, a single calf is born that is weaned at 6 months, young reach
sexual maturity at 2 years. Calves are kept in hiding in a special spot in the
undergrowth for up to two weeks after birth, until they are strong enough to
follow their mother. Though mostly silent, the call of this animal is partway
between a moo and a bleat, or a nasal snort. The young are preyed upon by the
marsupial panther and yagil,
but the adults are generally well defended and large enough to avoid predation.
Lifespan is up to 20 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Lembu
or Bornean false buffalo (Pseudotaurus lembu)
Order: Even-toed ungulates (Artiodactyla)
Family: Bovids (Bovidae)
Habitat: tropical forests of Indonesia (Borneo Island).
In Neocene feral descendants of primitive breeds of domestic goats (Capra domestica)
appeared the most successful survivors among bovids. They easily adapted to
new habitats and evolved in various directions to increase their biological
diversity. Lots of goat
descendants inhabit
Zinj Land – East African microcontinent splitted from African mainland. Island
part of South East Asia is another center of their biological diversity, while
the mainland is inhabited by large herbivores of other families – bulldeer
and thunderhorns.
Lembu is the largest mammal of Borneo Island. The name “lembu” means “bull”
in Malay language, and the appearance of this animal justifies this name.
Lembu has deep and narrow body, long legs and a humped back; hump is more expressed
in males. Horns of females are short and tightly curved, pointing inward at
the tips, while those of males arc upwards, growing 60 to 75 cm long. Spinous
processes of thoracic vertebrae represent a support for muscles that move male’s
large head. Male lembu has a head-and-body length of 240 to 310 cm, while the
females have 210 to 230 cm. Adult male is dark brown, approaching black in very
old individuals; the upper part of the head, from above the eyes to the nape
of the neck, is white; muzzle and lower parts of legs are also white. These
marks help animals to recognize each other in forest gloom. Females and juveniles
are chestnut with the same white parts as at male. Wool is short, but on lower
jaw a beard of elongated wool grows at males and older females. Tail is short,
covered with elongated hair forming a tuft.
Both males and females have paired leathery lobes on necks helping them to remove
an extra body heat. Flesh of these lobes is penetrated densely with blood vessels,
and when animal is overheated or excited, these lobes swell with blood and elongate.
Sometimes animals mutually lick these lobes to each other as a sign of good
relations between them.
These animals feed on grasses, young bamboo shoots, fruits, leaves and young
branches. Being active both at night and day, lembu pastures in the open areas
at night and under the forest cover during the day.
The herd is led by an old adult female, the matriarch. Herds number up to 20
individuals, generally females and calves, but males join them during the dry
season. Older males are solitary, but the young ones form bachelor herds.
After 278 days long gestation female gives birth to one calf. Calves are typically
weaned after seven to 12 months. Sexual maturity occurs in the second or third
year of life. Breeding takes place year-round. After reaching maturity, the
males are expelled from the herd. The usual lifespan reaches 30 years.
This species of mammals was discovered by João Vitor Coutinho, Brazil
Four
Horned Phobocorn (Quadrucornis armatus)
Order: Even-toed Ungulates (Artiodactyla)
Family: Bovids (Bovidae); subfamily: Phobocorns (Phobocorninae)
Habitat: Scrub and broad-leaf forest of eastern Madagascar.
During the beginning of the Neocene, some animals evolved into shapes and niches
seen previously in other animals. Less often, completely original forms appeared,
exhibiting features not seen in wild animals before. Some original groups became
entirely new kinds of animal, one such group is the representatives of Phobocorninae
subfamily.
Descended from run-wild domestic goats, these horned ruminants are in many ways
unremarkable. Their bodies and coloration resemble small and medium sized antelopes
of various kinds, such as impala or sitatunga. Their plain appearance stops,
however at the horns, a hallmark of their ancestors, greatly enhanced.
Phobocornines are defined by the presence of more than one pair of horns. Such
arrangements of horns had been rare in human era, present only in the Tetracerus
antelope, and some breeds of sheep. There are some genera, each having different
horns respectively, four or six, in various arrangements and sizes.
The four-horned Phobocorn, posses fairly impressive horns, 2 pairs, growing
from the brow, behind the eyes. One pair inclined upwards, the other down, in
this way, they resemble four-horned domestic sheep breeds of human occupation.
The horns have widely spaced ribs, and are slightly curved, and are only slightly
shorter than the length of the animal’s head. Unlike some other Phobocorns,
there are no horns on the base of the nose. Four horned Phobocorns are medium-sized
browsers of the forest thus having dark brown pelts with lighter vertical tripes,
similarly to some Holocene antelope, aiding in camouflage. In size, they are
equal to a medium sized antelope, like the sitatunga: the shoulder height is
about 130 cm at weight of about 140 kg.
Like all Phobocorns, breeding occurs in the wet season. At the middle of dry
season males display lekking behavior, with males competing physically by ramming
foreheads, and poking each other in sides with horns. Females give birth coinciding
with the flush of growth in the later part of the wet season.
Related species Lesser
Four-horn (Quadrucornis minor) inhabits sparse forest on the Western
side of Madagascar, and is also a browser. This one is mostly similar to its
larger relative, except for lighter coat color, body slightly smaller (shoulder
height is about 110 cm). Horns are not equal in size, outer pair are half the
length of the top pair, unlike eastern relative. It is relatively uncommon,
due to the presence of more successful Six Horned Phobocorn.
This species of mammals is discovered by Tim Morris, Adelaide, Australia.
Western
Dwarf Phobocorn (Neotetracerus dimorphus)
Order: Even-toed Ungulates (Artiodactyla)
Family: Bovids (Bovidae); subfamily: Phobocorns (Phobocorninae)
Habitat: Scrub and savanna of western Madagascar.
Dwarf Phobocorns are the smallest of the Phobocorns, and are mixed feeders on
both low browse, herbage, and soft grasses in scrub-land and grassland. They
posses smooth, short horns, one pair of typical brow horns, non spiraling, and
one smaller pair projecting from the base of the nose, in front of the eyes.
In this way they resemble the Indian Four Horned antelope of human era. Males
have both pairs of equal length, but females have the front pair larger, and
the posterior pair relatively smaller. This is connected to the fact that males
use their horns in territorial and lekking disputes with other males, but females
must use their horns to defend themselves and their young. These animals are
about the size of a miniature domestic goat, but more slender, possessing tan
pelts with a darker stripe down the back, the muzzle and eye area are darker.
Males joust and lock horns actively, competing for females. As with all phobocorns,
breeding occurs in the wet season. Young are relatively small, and will hide
in long grass similarly to a deer fawn.
Upland
Dwarf Phobocorn (Neotetracerus montanus) inhabits the plateu and mountain
slopes of central Madagascar, and is adapted to eat the tougher plants of upland
scrub and savanna. It is lightly colored, cream, with slightly darker patches
hardly discernible, dorsal stripe is paler than plain relative. Body and legs
are stouter, and the animal is quite surefooted.
This species of mammals is discovered by Tim Morris, Adelaide, Australia.
Pied-horn
Phobocorn (Cincatocornus ornatus)
Order: Even-toed Ungulates (Artiodactyla)
Family: Bovids (Bovidae); subfamily: Phobocorns (Phobocorninae)
Habitat: Grassland and sparse forest of western Madagascar.
The Pied-horn is a rather beautiful animal. It is pale greyish brown, with dark
"socks" on its lower legs, and light markings around the face. It
has 1 pair of long upward-backward pointing, spiral horns (like those of the
holocene Blackbuck antelope), in the typical place. There is also a smaller
pair of horns at the base of the nose, just before the forehead. The front pair
are ivory colored with black tips, whilst the larger pair are banded in black
and white, creating a striking appearance. Both males and females will use horns
for defense against predators, sometimes simply posturing with horns is enough
to deter attackers. This creature is about as large as a ram, with slightly
longer legs. It is a commonly seen grazer and only browses rarely, in times
of famine. Horns are arranged in such a way as to not impair vision when the
animal is grazing.
The Pied-horn is the most common and populous of the Phobocorns, forming large
herds. As might be expected, males do not joust or ram with such ornate horns.
Males simply posture and show horns, with elaborate head-sweeping motions, males
with most impressive horns generally win. Females give birth when the grass
is lush and green, at the end of the wet season, young are almost immediately
able to follow the herd.
This species of mammals is discovered by Tim Morris, Adelaide, Australia.
Six
Horned Phobocorn (Phobocornis apocalypticus)
Order: Even-toed Ungulates (Artiodactyla)
Family: Bovids (Bovidae); subfamily: Phobocorns (Phobocorninae)
Habitat: Open Forest, scrub, treed savanna, western Madagascar.
The most impressive phobocorn, truly a sight to behold. It is the size of a
pony, thus, the largest Phobocorn. So named for its uncanny resemblance to the
biblical multi-horned goat, harbinger of the Christian apocalypse.
This animal is colored ruddy brown, with faded greyish brown stripes, a light
underside, and white "socks". It possesses 2 pairs of large, ribbed
brow-horns, and a smaller pair of horns on the forehead. Paired horns are back-swept
so as to not interfere with feeding. Males and females have horns of similar
size, but males posses a "beard" of whitish hair on the neck and lower
shoulders.
This phobocorn is a generalist, mixed feeder, and has a large, chambered stomach.
Able to consume leaves of broadleaf trees or bushes, twigs and shoots, as well
as both soft and coarse grass. It derives some of its water consumption from
succulents, and will depend on them more heavily during the dry season, when
trees drop their leaves.
Breeding behavior is during the wet season, males use horns in competing for
females, but fighting is mostly ritualized. Horns lock, as with deer, and each
male tries to push the other's head to one side. Young are fairly large, and
move from suckling to regular browsing fairly quickly, but start with the more
tender parts of bushes and low trees.
This species of mammals is discovered by Tim Morris, Adelaide, Australia.
Duikapi
(Giraffotragus altus)
Order: Even-toed ungulates (Artiodactyla)
Family: Bonehorns (Osseocorni seu Parabovidae)
Habitat: savannas and light forests of Southern Africa.
After the ice age of boundary between Holocene and Neocene borders of some zoogeographic
areas have changed. So, after Mediterranean sea drying and Gibraltar isthmus
formation border of Holarctic zoogeographic area has moved to the south having
included the north zone of African savannas (the middle Sahara watercourse of
Niles (flowing now from the east to the west of Northern Africa and inflowing
to headstream of Niger river) serves as border between Holarctic and Ethiopian
zoogeographic areas). In Northern Africa there are descendants of Eurasian and
Near Eastern animals and small impurity of descendants of African forms. But
in Southern Africa separated from the north by continuous zone of mountains
and marshy woods stretched along the equator relicts of a Holocene have remained
and evolved - various species of even-toed hoofed mammals, descendants of different
African antelopes. They occupy different ecological niches and are not less
various than species of antelopes in early historical epoch.
Among herds of new South-African antelopes grazing in dense grass giants sedately
wandering by small groups and regaling themselves with soft leaflets of trees
inaccessible to anybody from other hoofed mammals are especially remarkable.
These giants of Africa are duikapi – descendants of one species of the African
wood duiker antelopes (Cephalophus). Though ancestors are too small, their descendant
is rather large herbivore mammal: its withers height is up to 170 cm at body
length up to 2 meters. Neck of duikapi is very long (the common growth of animal
from horns up to hoofs is almost 3 meters). Colouring of short animal’s wool
is sandy-yellow with brown cross strips on legs and neck; back is dark. Cubs
of duikapi seem darker than adults: on their body strips are narrower and denser.
These long-legged animals move with amble similarly to giraffes and camels.
In case of danger they can run gallop accelerating momentum up to 50 kilometers
per hour.
Skull of adult animals is strongly bent in middle part; large eyes and small
brain cavity tower above the lengthened obverse part. Due to high position of
eyes at these animals there is fine circular field of view and they notice predators
from afar. Similarly to giraffes and ostriches in Holocene epoch savannas duikapi
serve as though as “sentinels” for other herbivores. Large nasal cavities indicate
that at duikapi sense of smell is very keen.
On nape of duikapi the original cross wide “horn” is developed covered with
wool and doubled at top where reduced rests of true horns, two small horn knobs
are placed. Such bone ledge is used as the tournament weapon: contenders approach
to each other by chest, rest foreheads and “struggle” by heads trying to curve
neck of the opponent to force it to refuse struggle and to recede. Such fight
is the original compromise between natural habit to butt inherited from bovine
ancestors and rather fragile constitution of duikapi.
Duikapi keep in groups of 5 - 8 animals under the leading of elder female. In
such group there are some adult females and their under one year aged cubs.
Elder young males abandon mothers and young females can remain in parental group
or leave it and create own one. Males live solitarly or in groups of 2 - 3 animals.
Out of breeding season they tolerantly concern to each other but in breeding
season their aggression relatively to each other sharply increases.
Duikapi are out of competition among animals of South-African savannas and light
forests. These high animals browse branches of trees and bushes inaccessible
to their neighbours. If all accessible lower branches on tree are eaten duikapi
easily can rear on back legs resting hoofs against tree trunk and reaching up
to fresh branches.
Pregnancy at this species lasts about 6 months. At the female only one cub is
born at the end of dry season. Growth of newborn cub is about one and half meters.
First time it feeds exclusively by milk but from monthly age gradually passes
to vegetative food. It stops to depend on mother at the age of half-year, and
becomes completely adult at the third year of life.
Thick-horned
thunderhorn (Brontocornus magnus)
Order: Even-toed ungulates (Artiodactyla)
Family: Thunderhorns (Brontocornidae)
Habitat: Hindustan, South-Eastern Asia, woodlands, bushes, foothills.

In human epoch representatives hoofed mammals had a hard time.
They appeared among favourite game, number of many species had strongly reduced,
and some of them had disappeared at all as a result of hunting or change of
habitats. The ice age had finished this epoch appeared one more test for them.
Many species at this time had disappeared and variety of hoofed mammals had
sharply decreased. Some orders like proboscid and sirens had disappeared completely.
Therefore in Neocene hoofed mammals are presented by not numerous isolated families.
But their evolution was not finished - survived groups of animals evolved to
descendants perfectly adapted to changed inhabitancy.
In India separated from other Eurasia by Himalayas the whole new family of even-toed
mammal had appeared. Its representatives differ in various form of horns turned
to resonators for strengthening of voice. Because of this feature animals had
been named “thunderhorns”. They descend from bovines, obviously from any small
antelopes. Cavities of bovine horns had joined frontal and nasal cavities. At
thunderhorns, their descendants, these cavities had got more complex form and
horns had turned to resonators. At different species of these hoofed mammals
resonators have various shape and voices of these animals differ appreciably.
The voice serves for recognition of species representatives in bushes at a great
distance (all thunderhorns are inhabitants of forests and bushes). One more
role of loud voice is to inform the probable contender that any territory is
occupied and by that to avoid force methods of struggle. Resonator outgrowths
are rather fragile ones, also they are unsuitable for force struggle. In this
connection the horn cover on these outgrowths at different species is at different
stations of reduction up to its full disappearance. Females at all species of
thunderhorns have fewer horns than males.
Thick-horned thunderhorn, the central representative of family, has kept more
primitive ancestral features than other species. Its horns are similar to strongly
swollen and short ram horns having kept horn cover on the top side. The voice
of the present species resembles roar of any predator.
Thick-horned thunderhorn is small deer-sized animal differing in graceful constitution.
The basic colouring of wool is light brown. Neck, back and rear legs of animal
are covered with short black cross strips forming fine cryptic colouring. On
legs black wool forms “stockings”. Tail is white, on the bottom side of neck
also lengthened white wool grows. Tail put upright means signal of danger.
This animal keeps among bushes feeding by leaves and grass.
Thick-horned thunderhorn does not form herds and keeps in “harems”: one male
and 4 - 6 females. In breeding season between males duels take place accompanying
with loud frightening roar audible for kilometer and more. The rut falls to
the beginning of dry season. Pregnancy lasts about 5 months. As a rule, female
gives rise to one calf colored similarly to adults but having longer strips.
At the age of one year young animals leave parental herd and form their own
herds. Sexual maturity comes at three-year age (at females it happens some months
earlier, than at males). At this time herds of young animals separate to harems.
Life expectancy in nature reaches 12 - 14 years.
Forest
trumpeter (Buccinotherium magna-voce)
Order: Even-toed ungulates (Artiodactyla)
Family: Thunderhorns (Brontocornidae)
Habitat: Hindustan, foothills of Himalayas, light forests and thickets
of bushes.

This species is large hoofed mammals of heavy-build constitution,
one of the largest representatives of family: withers height of the adult male
reaches 2 meters, female is a little bit smaller. The skull of this species
is rather long; more than half of it the hollow resonating crest of the roundish
shape amounts. Horn covers have completely disappeared and remained bases of
horns are covered with velvety wool and rather brightly colored. The crest is
high, compressed from sides; its edge is bordered by wide strip of black wool,
and the middle part of crest is reddish-yellow. Body of this animal is colored
bluish-gray; stomach is white. On long tail rich brush of white wool grows.
Position and character of movements of tail at this animal expresses its condition
at present moment.
The voice of forest trumpeter has determined its name: it sounds like lingering
blares.
Jaws are long and rather weak: animal eats mainly soft food - leaves of trees
and bushes. To browse it from branches on top jaw nose and upper lip have formed
similarity of short proboscis, as if at tapir.
Wood trumpeters live in forests near large rivers where giant grasses and bushes
plentifully expand. Hoofs of this animal are well adapted to walking on fenny
ground and then forest trumpeter is able to swim well.
This mammal lives by couples kept for the whole year. The male incurs protection
of territory borders marking them by heaps of manure. Also he warns the probable
contender that the territory is occupied with the help of shouts. Wood trumpeters
are especially “vociferous” in the morning. They actively shout while above
river valleys there is dense fog: probability of predator attack is less so.
Once a year, mainly at the end of dry season female gives rise to one cub. By
colouring it is similar to adult animals but on the head instead of crest only
strip of black wool is. The young animal keeps with parents almost year, and
female banishes it shortly before birth of the next cub. The animal becomes
the adult at the age of four years.
Shadhavar
(Shadhavar fabulous)
Order: Even-toed ungulates (Artiodactyla)
Family: Thunderhorns (Brontocornidae)
Habitat: Hindustan, Southwest Asia up to the Persian ridge, plains and light
forests.

Picture by Leonard Popov
Initial image - picture by Pavel Volkov |
In Oriental (Persian) legends shadhavar is the fantastic creature : the antelope-like
one with unique hollow horn. According the legend when the wind blew, the horn
of this antelope sounded attractively to other animals. When curious animals
approached closer, shadhavar rushed on them and ate: the word of mouth attributed
to this fantastic animal furious behavior. And only the voice of the wild pigeon
could charm this animal so that hunters could catch it.
But legends have disappeared together with people, and in savannas and light
forests of Southern Asia the animal somewhat resembling the legendary creature
has appeared. Its behavior is not so furious, as at the fantastic prototype,
but some other properties rather approach this hoofed mammals from thunderhorn
family with fictious creature. The horn of this animal is especially remarkable:
bases of pair horns are pulled together and have grown together to the single
structure resembling the crest of dinosaur Parasaurolophus. Only this hollow
horn is straight, also continuing a line of animal head. The length of horn
exceeds length of other skull in one and half time. The corneous cover on this
structure has disappeared completely; "horn" is covered with short
thin wool. It is not more used for fight, its purpose is to be the resonator
and partly to carry out alarm function. The tip of horn is covered with black
wool: it is a mark for recognition of neighbours. During ritual combats males
bend head down so that the horn sticks upwards as it is possible. This way
they determine the strongest one without fight. The horn of the female is shorter,
and the black tip on it is not so big. Due to horn the male can utter lingering
trumpet shout of high tone. The voice of the female is more silent, than at
the male.
By other features shadhavar resembles other artiodactyls. It is herd animal
by proportions similar to fallow deer. It lives in bush thickets in foothills
and valleys, in summer rising highly in mountains. It keeps in herds by 10
- 20 ones. The basic background of body colouring is yellowish - grey, on wool
there are longitudinal black stripes, lasting along all body from shoulders
up to hips. These stripes help animals to disappear among bushes and do not
allow predators to distinguish separate animals in herd (colouring of zebras
works by similar mode). The animal can jump high.
It eats mainly leaves of bushes and soft grasses.
The coupling season falls to the end of summer. At this time males establish
hierarchy and gather harem from 5 - 6 females, protecting them from other males.
After coupling season the hierarchy is almost erased; and recent contenders
do not pay attention against each other. Once a year, in the beginning of spring
the female gives rise to two calves. They have no characteristic horn, and
their voices sounds like lamb bleating. Calves are covered with small spots
which merge in longitudinal strips later. Approximately to the age of one year
at them the horn starts to form. At the female it reaches full development
to the age of three years, at the male – up to the fifth year of life. Sexual
maturity comes in three years, but males really can participate in breeding
from five - six years.
Desert
whistlehorn (Stridocornis aridophilus)
Order: Artiodactyls (Artiodactyla)
Family: Thunderhorns (Brontocornidae)
Habitat: semi-deserts and deserts of Near East and Northern Africa.
In human epoch hoofed mammals have strongly suffered from people activity.
Destruction of their habitats and active hunting had resulted to that only
few of them have survived up to Neocene. Indirectly reduction of number of
hoofed mammals had resulted to that other groups of animals have started to
master their ecological niches. Nevertheless, evolution of “true” hoofed mammals
in Neocene has proceeded, and they even had formed new groups which had not
existed in human epoch. One of such groups is a family of thunderhorns. They
are mainly inhabitants of open spaces and light forests of Southern Asia and
Northern Africa. One of representatives of family, the desert whistlehorn,
has mastered life in rather difficult conditions: it lives in droughty district
bordering from south and east coasts of Mediterranean salt swamps.
This animal of graceful addition looks similar to gazelle. The desert whistlehorn
is medium-sized animal: it weighs only about 60 - 70 kg, its withers height
is about 1 meter. Its legs are long and thin, with rather small hoofs. The
desert whistlehorn can run very quickly, accelerating on a short distance to
speed over 80 kms per hour. Because of small hoofs it avoids viscous ground:
sandy sites of desert and boggy saline soils. In a dry season these animals
meet on clay sites of desert, but in short rain season they leave in territory
with dense and drier ground.
Body of the desert whistlehorn is one-color, sandy-yellow; wool on back is
lighter up to almost white. Tail is long, with hairy brush of light hair on
the tip. Characteristic feature of representatives of family is resonator chambers
of various shapes on the head. At the whistlehorn they look like the vertical
semicircular “crest” covered with black cross strips on light background. As
at all thunderhorns, this outgrowth is formed by cavities of former horns,
and serves for strengthening of sounds uttered by animals. However, the outgrowth
is not only the resonator: the bone forming it is very thin also is penetrated
with plenty of blood vessels. This adaptation also serves for cooling, effectively
radiating surplus of heat. Of course, animals with such structure on head are
not able to butt, therefore relations of hierarchy in herd are established
mainly with the help of loud voices, pushes by shoulders and impacts by hoofs.
But at whistlehorns the special form of expression of submission has appeared:
weaker animal licks crest to stronger one. The sense of this behavior is, that
evaporating saliva helps the licked animal to cool. This behavior became an
element of courtship ritual of animals: the male “licks” a crest of the female
like this, but only stronger ritualized, slightly touching it by tongue.
The desert whistlehorn lives in hot and droughty district, and eats various
plants, including bitter and even poisonous for other animals ones. It can
not drink for a long time, receiving a metabolic water from the eaten plants.
This animal keeps in big herds, in which hierarchical relations are established
only in pairing season. At this time males start to utter loud sounds: long
and also shrilly whistle through nostrils, blowing air through an outgrowth
on a head (of course, it is connected with nasal ducts). Showing the resonating
outgrowth to females, the male bends head back, showing it on the background
of light wool of back.
In epoch when the climate became drier, the part of population of whistlehorns
dwelt in Northern Africa has passed the Mediterranean bogs by way of isthmus
connecting territories of Tunis and Sicily. In territory of Southern Europe
they had turned to the special species – Etrurian whistlehorn (Stridocornis
etrurius). The European species differs from African one in larger sizes, dark
colouring (wool on sides and back is monotonous red), and propensity to life
in foothills. Etrurian whistlehorn runs not so quickly, but dexterously swarms
up rocks. It has stronger constitution, and its forward legs are appreciably
shorter then back ones: it is a feature of adaptation to inhabiting on mountain
slopes. Head of this animal is narrower, but higher, with short and high resonating
crest. Courtship cry of this animal also differs from voice of desert whistlehorn:
it sounds like abrupt loud sounds.
Nanditherium (Nanditherium altissimus)
Order: Even-toed ungulates (Artiodactyla)
Family: Thunderhorns (Bronthocornidae)
Habitat: Hindustan, forests of various types.

Picture by Lambert
Initial image by Pavel Volkov |
Restoration of tropical forests after disappearance of mankind
promoted expansion of areas of forest species of animals and occurrence of new
species adapted to life in such ecosystems. Occurrence of forests results in
impoverishment of ground vegetation and in moving of organic substances accessible
to consumption to the height inaccessible to many ground animals. At ground
herbivorous mammals the choice is very poor in this situation: resettlement
to any other habitats where habitual conditions of life are kept, or increase
of size, allowing reaching tree branches. In Africa the foliage from trees is
eaten by high giraffe ostriches to which ndipinotheriums similar to huge Pleistocene
sloths compete. In Southern Asia they have an ecological analogue belonging
to thunderhorns family – new group of artiodactyls, characteristic for tropics
and subtropics of Eurasia to the south from Himalayas.
Nanditherium, the original Indian analogue of giraffe lives in woodlands of
Hindustan. As against a giraffe, this animal has very massive constitution and
shorter legs. The name of this animal means “Nandi beast” after Nandi, saddle
buffalo of Shiva, the god of Hindu pantheon.
This is one of the largest mammals of Eurasia. Growth of adult nanditherium
male at a shoulder makes three meters, and skull is about one meter long. Animal
has thick two meter long neck mobile in the basis. Cervical vertebrae are light
and porous; it makes them much more lightweight at preservation of durability.
Bones of legs are very strong; under terminal phalanxes of fingers/toes shock-absorbers
of fatty tissue develop, increasing the area of support and reducing loading
on hooves. Skin on legs is dense (as at giraffe), that interferes with stagnation
of blood.
Nanditheriums browse leaves and young shoots of trees at height of about 4 –
5 meters, not having competitors among other ground mammals. Jaws of this species
are long and rather weak, and teeth are adapted to chewing of soft foliage.
Nanditherium’s tongue is long and mobile; it operates as a hand when animal
browses foliage – it twists and tightens to mouth of animal branches from which
this animal tears off leaves by mobile lips.
Colouring of nanditherium is very original and includes two types of the pattern
smoothly passing one to another. Head and top part of neck of animal are colored
contrastly and brightly – on white background there are large black irregular-shaped
spots. Shape and arrangement of these spots are individual, and animals find
out each other using characteristic pattern on head. In top part of neck white
color changes to brownish, and black passes to brown; spots turn to cross strips.
Neck and forward part of body are striped, strips stretch on shoulders downward
from the top. On back part of body strip pattern becomes poorly appreciable
and body has almost smooth yellowish-brown colouring. Bottom part of legs and
stomach of nanditherium are light colored, almost white. Young animal does not
have white sites on body, and strips are more contrast, rather than at adults.
Black-and-white colouring on head appears only after the beginning of independent
life – at males earlier, than at females.
Head resonator of nanditherium represents crest of rounded shape which lasts
along the whole head. Any rests of corneous cover are completely absent on it.
With the help of resonator animals of this species utter infrasounds for communication
at the large distance as elephants made it in Holocene epoch.
Nanditheriums live in small herds, including male, two or three females and
their posterity of several years. In forest groups of these animals tread out
ways which generations of animals use. Obda,
large herbivorous mammal of Siberia, changes landscape in similar way. In these
places forest canopy is broken and plentiful underbrush develops, giving food
to smaller herbivores. Along nanditherium tracks a steady complex of animal
and plant species develops.
Similarly to all large animals, nanditherium breeds very slowly. Female gives
rise to one calf once in three years: pregnancy lasts till about two years,
and in the rest of the time calf eats milk. After the female has stopped feeding
it, young nanditherium stays in parental group for about two years, and leaves
it at birth of the new calf at its mother. At this time the adult type of colouring
on head starts to appear at it, and adult animals start to behave more aggressively
to the grown up animal. Young animals gather in small groups and fed, moving
on tracks of adult animals. Due to large size death rate among young nanditheriums
is low. Sexual maturity at these animals comes at the age of seven years. Life
expectancy makes till 60 years and more.
Simon, the forum member, has helped to specify features of biology of present species.
Mountain
camelope (Leptocamelus montanophylus)
Order: Even-toed ungulates (Artiodactyla)
Family: Camels (Camelidae)
Habitat: mountain areas of Meganesia.
Picture
by Rafael Silva do Nascimento, Brazil
In Australia camels never were found in wild and they could not get to this
isolated continent naturally. Woods and sea passages reliably barred them the
way to the “lost world” of Australia. Only due to one species of primates,
Homo sapiens had become extinct at the end of Holocene they could get to this
continent as domestic animals. After human disappearance their ancestors, dromedaries
(Camelus dromedarius), have become wild and have given rise to several species
of herbivores competing with local marsupial animals. Among camels analogues
of flatland giraffes and antelopes, and also mountain goats have appeared.
The mountain camelope is strong build relative of swift-footed camelope Leptocamelus
gracilis, graceful runner of Australian plains. This species differ from plain
neighbour in shorter legs and neck, and also in larger narrow head. Besides
mountain camelope is covered with dense wool that helps to endure cold night
of Australian Alps. It is rather medium-sized species - its weathers height
is up to 1,2 m, body length is about one and half meters. Back of mountain
camelope is inclined forward because back legs of animal are appreciably longer
than forward ones – so it is more conveniently to graze on mountain slope.
Wool of animal is colored grey with brownish shade; on ridge there is longitudinal
black strip which at males is much wider and longer than at females. The hump
at these camels is expressed poorly - in middle of back small swelling is appreciable.
On chest and basis of neck at animals of both sexes mane of long hairs develops.
On head at males very long hairs grow a rich bun. To time of rut a liquid from
specific gland located on nape impregnates them – hair help to strengthen smell
and to keep it for a long time. At females such hairs are not present; head
is covered with short wool. Legs are ended as at all representatives of the
order by two hoof-like claws and special skin “pad”; the weight of body falls
to end toe phalanxes. For the best cohesion with stone surface leg soles are
covered with strongly cornificate skin.
Mountain camelopes live on western slopes of Australian Alps in conditions
of cool and rather dry climate. These animals prefer to live in forestless
districts not coming to thickets of trees occasionally growing in valleys.
They eat grass at times reaching up to plants on very abrupt slopes. Due to
features of legs these animals easily jump and swarm up hillsides surviving
in places where marsupials of size comparable to them can not live. Teeth of
mountain camelope have the special structure: incisors are strongly jutting
out forward as if a nipper, and molars grow all life: it is an adaptation to
grazing of food plentifully “flavoured” with sand and dust strongly erasing
teeth.
These animals live in herds of 10 – 15 ones under the rule of large male leader.
He constantly supports the authority in herd with the help of might demonstrations
and loud roar. In herd the significant part is presented by mature females
and their cubs. At mountain camelopes one cub alternate years is born as a
rule. It depends on mother for a long time studying at it to move in mountains
and to search for food. Usually it becomes completely independent at the third
year of life leaving mother at birth at her of the next cub. The leader of
herd does not support youngsters in herd as among them his probable future
contenders can grow. When young animal leaves from under parent trusteeship,
it has a lot from herd leader forcing young growth to submission. Because sexual
maturity at young females comes only at the fourth year they do not represent
interest for the main male and also are driven out. Young animals form small
herds either mixed or including only young males. At this time the part of
young growth perishes from various predators. Such herds of young growth live
in boundary territories between areas of breeding herds and occasionally between
old and young animals skirmishes happen. When young females become sexual matured,
between young males fights for “harem” possession begin. Contenders rear, collide
by chests, strike impacts by forward legs in sides and chest of contender and
also bite strongly. Thus fighting males loudly roar and sometimes such loud
demonstration of ardour serves bad service for them: having heard such roar
skilled adult males hasten to these sounds and disperse self-confident young
growth adding some young females to own herd. If at the herd “sharing” place
two adult males face, fight between them happens severe and frequently results
to mutilations or fatal outcome. Sometimes young male can live many years without
“harem”, from time to time attempting to “property” of any “married” neighbour.
Sometimes such bachelor literally chases herd of the contender trying to deliver
from it one or several females.
Mountain camelopes are threatened, as a rule, only with local marsupial predators.
If there is no opportunity to rescue in flight, herd can pass to active defense
trying to strike predator by forward legs or to bite.
Life expectancy of mountain camelopes can reach 40 years.
Caballocamelus, horse camel (Caballocamelus velox)
Order: Even-toed ungulates (Artiodactyla)
Family: Camels (Camelidae)
Habitat: savannas and semideserts of Meganesia.
Picture by Carlos Pizcueta (Electreel), colorization by Lambert
Initial image by Carlos Pizcueta |
Until human arrival it was no large running mammal in Australia.
Birds and kangaroo were their ecological analogues. The occurrence of camels
(Camelus dromedarius) introduced by people had changed the course of evolution
of continent inhabitants. Marsupials could not evolve to four-footed quickly
running animal, replacing hoofed mammals only from the point of view of ecology.
Despite of the competition to camels, they had survived and successfully evolved
in Neocenic Meganesia. They avoid a competition to descendants of camels due
to feeding by different kinds of forage, and also due to effective digestion
of accessible, but poorly nutritious food. As a result descendants of camels
had occupied only a part of ecological niches in plains of Australian part of
Meganesia. Among them there are camelopes
of gracile constitution, and high giraffamels
having no competitors in bottom and middle levels of tree tops. But these animals
live in districts where there is though any wood vegetation. In plains of Central
and Western Meganesia, to the south of the zone of forests and woodlands, another
descendant of camels, caballocamelus, the gregarious mammal similar to the horse,
lives.
Caballocamelus is a large animal of robust constitution: its withers height
is up to 160 cm, and the general length of body is about 3 m. At this animal
there is a large head on rather short and strong neck. Jaws are short and have
constantly growing teeth, adapted for grinding of rigid graminoids and bush
branches. The muzzle is extended to short almost immovable proboscis serving
for protection against dust and humidifying of inhaled air.
The hump is small; it is stretched along the back up to waist as the low wide
platen. The tail is turned to “fly-brush” having hairy brush on the tip.
The wool of caballocamelus is very short and velvety. On the bottom part of
neck at adult animals a lot of lengthened hair similar to beard grows. Colouring
of body is yellowish brown; on shoulders and hips there is a small amount of
faltering dark strips. On legs of animal there are white “stockings”. At adult
animals head is darker, rather than body; at male forehead and nose bridge have
coffee-brown color. At young growth the head has the same color as the body,
and on legs there is lack of “stockings”.
The foot of typical camel is adapted to rather slow movement on soft ground:
toes are widely separated, sheafs between them are lost, and the support falls
to some distal phalanxes. At caballocamelus the structure of foot has changed
according the adaptation to fast running. Toes of this animal are connected
by sinews to each other along almost all length. At young animal sinews are
elastic, but to the approach of maturity they ossify. Distal toe phalanxes are
thick and covered with “stockings” of thick skin. The characteristic for camels
subungius, at which the leg bases, is very small.
This animal lives in dry districts with firm ground, lack of wood vegetation.
Caballocamelus quickly runs, accelerating momentum about 50 kms per hour. Its
pace at short distances is gallop, at long one it moves by an amble (it is a
characteristic pace of typical camels). This species lives in herds numbering
20 – 40 animals.
Caballocamelus eats rigid grasses and can even gnaw prickly branches of bushes.
Its three-chambered stomach (inherited from the dromedary, its ancestor) helps
to digest such food. Lips covered with dense skin help to feed on prickly plants.
In rain season animals can not drink till many days, being content with moisture
received from plants. But in dry season these animals visit reservoirs approximately
once in three days. The leader of herd knows well all reservoirs in places of
inhabiting of herd, and each time animals visit new reservoir for watering.
The herd includes from 1 up to 3 harems leading by dominant males. Each male
owns 5 – 10 females; their posterity stays in herd up to the maturity (young
males are expelled from the herd a little bit earlier, rather than females).
Young females are more often quietly poured in other herds, and single males
form herds. Becoming stronger, they try to bereave any adult male of authority.
Males of this species are very aggressive in courtship season: they bite each
other and beat by forward legs. Skin of the male is thick, especially on shoulders
and in the basis of neck – it is the adaptation for intraspecific courtship
tournaments. Out of breeding season males treat to each other more tolerantly,
but at the watering place harems approach to water in sequence appropriate to
rank of the male in herd.
Pregnancy lasts about 10 months; one cub is born always. The female cares of
it for a long time, therefore the posterity at the female is born only once
in 2 years. The female does not admit neighbours to the newborn cub while it
will rise on legs, and some more hours after it. At this time the cub remembers
smell and voice of mother. It is very much adhered to mother, and does not depart
far from her for a long time. A parental instinct at this species is very strong:
in case of need all adult animals of a clan protect the cub; therefore the survival
rate of posterity at this species is rather high.
Siberian
digger (Paramarmota architector)
Order: Rodents (Rodentia)
Family: Squirrels (Sciuridae)
Habitat: Western Siberia, flat districts, river valleys.
Small animals had gone through mass extinction of the epoch at the boundary of
Holocene and Neocene much more successful, rather than large species. Their variety
at the level of families remained practically the same, as before the catastrophe;
separate genera had died out, but new ones appeared to replace them. Ecological
niches occupied by them had remained the same – these are small forest and grassland
animals, herbivores and omnivores. Among them also digging forms remain, for
example, suslic doggies from three-Rivers-Land steppes. In severe continental
climate of Siberia the separate species of digging animals settling in large
colonies, Siberian digger, also have appeared. The colonial habit of life provides
more successful survival of these animals in conditions of contrast continental
climate and severe long winter. This animal descends from one species of suslics
inhabited glacial steppes during the congelation at the boundary of Holocene
and Neocene.
From the point of view of anatomy Siberian digger represents very large suslic
– one average animal weighs up to 3 kg. Ecological analogue of Siberian digger
is the marmot – digging animal building extensive underground constructions.
The colony of Siberian diggers numbers up to 200 – 300 animals, and they construct
under the ground a huge settlement which part is placed at the depth up to 4
– 5 meters, inaccessible for predators.
Settlement of these rodents represents the circuit of underground tunnels supplied
with drainage “shafts” and drainage burrows which open in steep riverbank and
can stretch to tens meters from edge of colony. It is necessary for protection
of colony in spring from thawed snow. Besides exits to the surface are made as
high (up to two meters) earthen hills with an entrance at the top. At snow thawing
in them only a little of ice-cold water will get. From these hills the good view
opens, therefore in summer at tops of hills “sentries” – males from among defenders
of a colony –are constantly on duty.
Except for earthen hills – entrances to the colony, presence of colony is marked
by the bushes and young trees damaged by rodents. Siberian diggers influence
a landscape as essentially, as large herbivores like obda
or shurga: within the limits
of their colonies trees survive hardly (they are simply chewed at the sprout
stage). But above colonies of diggers graminoids and other plants, bearing drier
ground (presence of drainage system in colonies has an effect) and constant
“shear” of them by these rodents, prosper. Forager individuals from colony also
make sorties for the forage in thickets of bushes and forest.
Siberian diggers are animals having well expressed caste system and obvious intraspecific
polymorphism. Like Neocenic castle rabbits from New Zealand or naked mole rats
(Heterocephalus glaber) from Africa of Holocene epoch, these animals are divided
to castes. The caste system is not fixed genetically, and depends on age and
position of animal in hierarchy of colony. At these rodents there is large head
with very short ears, able to wrap when the animal moves in hole. The trunk of
Siberian digger is covered with rich velvety fur similar a little to mole fur:
hairs grow vertically, and do not prevent movement in hole forward or back. The
fur of animal is colored brown with small amount of white spots on back and shoulders.
On head of adult males there is an area of white fur on sides of head and around
of eyes. Depending on position of animal in hierarchy colouring of fur may vary
strongly.
Paws of Siberian digger are short, with wide feet, having edge of rigid hairs
–it is convenient to rake away the ground by such paws during the digging. Tail
is short, with white tip. Tail lifted upwards is an alarm signal, well appreciable
even from among grass.
The caste system of Siberian diggers includes some categories of animals:
1) “King” and “queen” – breeding pair of large fertile animals. But the amount
of these animals is not limited, as at insects. In large colony occurrence of
several “queens”, which are fertilized by one or two “kings”, is possible. If
two “kings” meet in tunnels of colony, between them severe combat is possible,
but usually “king” does not leave from the centre of colony. From these animals
all posterity of colony descends.
“Queen” represents the individual which almost continuously gives rise or brings
up young growth. It lives in special chamber; it and its posterity are looked
after by some females from the number of “nurses”. “Queen” is very large – it
is approximately twice larger than the adult forager female. At it milk glands
are strongly advanced, and it almost continuously produces milk (except for second
half of pregnancy). Because of it “queen” requires a plenty of juicy forages,
and “nurses” additionally give it to drink, dragging water from drainage “shafts”
or from the nearest reservoir in cheek pouches. Fertility of “queen” is about
15 naked, blind and helpless cubs in one litter. In some days after cub birth
it is ready to become pregnant again. For one year “queen” makes up to five litters.
“Queen” can not look after itself and posterity independently; it is only able
to creep hardly in the chamber. If in spring thawed snow fills in chambers where
“queens” live, they perish, being not able to escape.
“King” is the large fertile male with normal proportions of body, able to serve
itself independently. It freely moves in colony, independently eats in “pantries”
and searches for “queens” ready to fertilisation. At this male sexual attributes
in colouring of wool are obviously expressed: on head around of eyes and on cheeks
areas of white wool develop. Also “king” has repugnatorial glands: it has a specific
smell which influences other males and suppresses sexual behaviour at them.
2) “Soldiers” – aggressive adult males. They are in condition of constant stress
because of presence of “king” in colony. The stress causes in them formation
of plenty of adrenaline, and hormonal changes lead to irreversible changes in
their appearance. Incisors and jaws of “soldiers” become large; therefore the
head of such individuals looks disproportionately big relatively to the body.
Sexual function at them is suppressed from the childhood, especially if they
developed in conditions of density in the centre of colony. These animals can
hardly eat independently because of hypertrophied jaws; more often young individuals
from “nurses” caste feed and clean them. But at an attack on colony they leave
on surface the first, and furiously attack any animal had disturbed them, even
such large one, as obda or shurga. At the edges of colony where the stress is
less, features of “soldier” at males are less expressed, and among them fertile
individuals, from whom new “kings” may grow, frequently meet.
Colouring of “soldier” is actually warning: at them there is almost completely
white head with dark marks on forehead and nape, and white spots on shoulders
are very big. Such colouring is well remembered by animals which had undergone
to attack of “soldiers” of colony, and forces them to keep farther from these
animals.
3) “Foragers” and “nurses” – females of various ages. Adult and old females are
foragers. They constantly gather forages and drag them in holes serving as “pantries”.
These animals are able to feed and look after themselves independently. Young
females have function of “nurses” – they look after “queens” and “soldiers”,
clean and feed them. At them the parent instinct is early expressed, but it equally
quickly fades. If in the short period of maturing the female was not fertilized,
it becomes the forager. Young “nurses” have opportunity to become “queens”, if
they will be fertilized within first three months of life. At pregnancy there
is a hormonal reorganization of the organism, and young female develops to “queen”
occupied with reproduction.
If the colony becomes too large, in it “diarchy” can arise: at the edge of colony
the young male (from among the individuals less subject to stress) and the female
ready to breeding from the number of “nurses” can create new breeding family.
If founders of colony perish, their place is occupied quickly by individuals
from among young ones. Frequently young males from other colonies come in colony.
They lodge at edges, and also can become founders of new “dynasty”.
Siberian diggers eat exclusively vegetative food. Everything, that can be stored
within winter, is reserved in special chambers serving as “pantries”. These are
tubers and rhizomes of plants, and also seeds of graminoids. Daily food of animals
in summer includes young branches of bushes, greenery and roots; the autumn adds
to them fruits of various plants. Foragers of this species carry food in cheek
pouches. In them there are no salivary glands, therefore the transporting forage
stays dry and does not spoil in winter.
In winter activity of colony is reduced: animals spend the most part of day in
dream. When bad frosts come, all colony runs to true hibernation, at which the
body temperature of animals is strongly reduced, and vital processes are inhibited.
At this time even development of embryos at breeding females stops. Due to this
feature consumption of forages does not increase, and the colony successfully
exists up to the end of spring when there is an opportunity to pass to fresh
forage. When snow thaws, flooding of the part of colony is possible, but the
majority of animals usually survives. When all difficulties are left behind,
and in nature there is enough of fresh vitamin forage, in colony the burst of
birth rate takes place. The care of posterity raises survival rate of young growth,
and damage, caused to the colony by predators and weather, is restored.
Life expectancy of foragers and “soldiers” seldom exceeds three years, but “kings”
and “queens” can live till ten years and more.
Stripe-tailed squirrel (Callurosciurus fasciatus)
Order: Rodents (Rodentia)
Family: Squirrels (Sciuridae)
Habitat: Honshu Island (Japan Archipelago), tropical rainforest.

Picture by Alexander Smyslov
It is difficult to name squirrels as “living fossils”, because
this group of rodents is widely settled on the Earth and inhabits various biomes
– from deserts up to tropical forests. However, long before human appearing
squirrels were already quite recognizeable animals and their very successful
body plan was kept from Oligocene. The majority of squirrel species, except
for inhabitants of tropical forests and not numerous local species, has successfully
gone through human epoch, and in Neocene their species are as numerous and various,
as in pre-human epoch of Cenozoic.
In Japan Islands numerous species belonging to endemic for these islands genus
Callurosciurus live. Their typical representative is stripe-tailed squirrel,
the owner of spectacular tail. The area of this species includes tropical rainforests
at the east of Honshu Island. This species has all typical features of genus.
The body length of stripe-tailed squirrel is rather small – no more than 15
cm. But tail of this creature is very remarkable: its length is more than 20
cm; it is cross-striped, black-and-white, as at common lemur (Lemur catta) of
human epoch. Tail of stripe-tailed squirrel is rather thin, but covered with
rich wool and looks as cylindrical-shaped one. It is used as the balance weight
at climbing and jumps, but its basic function is a little bit other – with the
help of tail this squirrel declares to the congeners that the territory is occupied,
and also gives other signals. Similarly to all representatives of genus, stripe-tailed
squirrels are very territorial animals. Each individual chooses a site in forest
canopy, carefully marks its borders with nibbled areas of bark and odorous marks,
and jealously protects the occupied territory against intrusion of congeners.
Body of stripe-tailed squirrel is colored not as distinctly, as tail is. The
basic background of body colouring is grey with small brown specks. On head
there is a white “mask” covering forehead, top and lateral parts of head. Colouring
of ears sharply contrasts with this background: ears are black and edged with
red hair. Such colouring is well appreciable among foliage, and squirrel could
become easy prey for predators. However these animals have effective system
of the notification of congeners. In spite of the fact that stripe-tailed squirrel
does not endure the presence of congeners in the territory, it constantly exchanges
sound signals with them. Having noticed a predator, squirrel utters alarm signal
– shrill thin peeping. It is well known to all inhabitants of tree crones, and
stripe-tailed squirrels are among the main informants about presence of predators
in forests of Japan Islands. Having heard alarm signal of the congener, each
individual repeats it necessarily. Duration of repeated signal depends on loudness
of heard sound, therefore it is possible to define approximately where there
is a predator noticed by them, listening voices of squirrels.
Stripe-tailed squirrels arrange dreys on trees. They have completely passed
from life in hollows, and only build their own dreys. These rodents belong to
number of the most skilful builders among mammals. They plait spherical dreys,
remotely similar to nests of weaverbirds, on tips of long shaken branches. Building
its drey, squirrel bends branch in a ring and connects it with vegetative fibres,
or plaits to each other the lateral branches growing on it. This ring becomes
a basis of nest. New branches forming a skeleton of nest are added to it, and
space between them is braided with fibrous stalks of fern fronds or leaves of
epiphytic plants. From inside the drey is covered with leaves. The drey entrance
is located below, but the narrow corridor stretches along lateral wall and opens
in the top part of nest.
Stripe-tailed squirrels do not form constant breeding pairs. Pairing tales place
at any time of year, but most intensively two times per year: in spring and
in the middle of summer. Males leave their territories and search for females
ready to breeding. Females constantly mark borders of territories with urine,
and its smell helps the male to determine, whether it is possible to be coupled
to this or that female. Male courtship actions have formal character – it pursues
the female and tries to catch her tail. If to it succeeds to do it (accepting
male courtship, female allows it to make it), fast pairing follows, after which
female banishes male. Nevertheless, male keeps for some time on border of her
territory, driving away any contenders. When female loses interest to male courthship,
its urine gets other smell and male, feeling it, leaves borders of her territory.
Having built a drey, female uses it for some years, constantly renewing and
repairing, but then abandons it. In litter it may usually be up to 5 – 6 cubs.
They grow quickly, and at the age of 2 months already become completely independent.
As against adult individuals, at young animals the tail has no black-and-white
strips – it is almost completely grey with light tip. Also head has no white
“mask” which appears only closer to the time of sexual maturity (at the age
of 4 – 5 months).
Life expectancy of this species in nature can reach 4 – 5 years, but the majority
of animals does not live up to the third year of life.

Picture by Alexander Smyslov
Flame-tailed squirrel (Callurosciurus flammeocauda) is the most spectacular representative of genus. Its native land is the area of tropical forests of Shikoku Island. Body of this squirrel is black on back and sides; top of head, nose and sides of head are white, stomach is rusty-red. Tail looks very impressively: it is deep red; tip is yellow passing to white. Declaring rights to its territory, flame-tailed squirrel raises tail and trembles it. Thus it utters shrill high-frequency squeak; and some birds and predatory mammals can not endure it. This species of squirrels arranges drey, braiding two branches of near growing trees. In the beginning of drey construction squirrel should show miracles of acrobatic skills. Hanging on the branch, squirrel shakes it and tries to grasp a branch of near growing tree. When it succeeds to do it, squirrel gnaws out from one branch narrow strip of fibres and ties both branches together with it. After it I done, squirrel starts building of typical spherical drey between them.

Picture by Alexander Smyslov
Long-tailed squirrel (Callurosciurus longicauda) is one of large species of this genus, widespread in tropical and warm to temperate forests of eastern side of Honshu Island. The length of its body is about 20 cm, and very long striped tail reaches half-meter length. Body of this squirrel is grey and has glaucescent shade; along both sides wide black strips stretch. There is the same strip on each side of head – it stretches from tip of nose through eye up to the basis of ear. On neck and shoulders strip is fuzzy, turning to dark grey spot, and appears again on sides. Tail of this species is white with red tip.

Picture by Alexander Smyslov
Reed squirrel (Callurosciurus calamophila)
lives in swampy forests at the east of Honshu Island and prefers to keep in
thickets of reed instead of trees. It is a tiny species: body length of adult
individual does not exceed 10 cm; tail is of the same length. Body of this squirrel
has grey colouring with several longitudinal strips of rusty red color on nape,
neck and back. Ears and nose bridge, and also hands and feet are colored black.
Tail sharply contrasts with body in colouring: it is snow-white with bright
red tip. Declaring the rights to territory, this squirrel hangs upside down
on reed stalk, uttering thin squeak and having raised tail up. Drey of this
squirrel represents a kind of ball of reed plaited very densely and littered
with vegetative fibres.
In mountain forests of Honshu Island golden-tailed
squirrel (Callurosciurus chrysocaudata) lives; it is the largest and
hardy species. The length of body of this species reaches 40 cm; tail is about
50 cm long. The adult animal of this species weighs about 1,5 kg. This is an
animal of heavier constitution, than heat-loving species inhabiting tropical
forests: it has rather large head and shorter ears and paws. Golden-tailed squirrel
lives in coniferous and deciduous forests in mountains of island where snow
in winter lays, and eats seeds, buds and young shoots of wood plants. In summer
it willingly eats mushrooms growing on trunks of old trees. Wool on body of
this species is colored grey with glaucescent shade, darker on back. Tail is
straw-coloured with black tip. Also ears of animal and “eyebrows” above eyes
are colored black. Voice of this animal is similar to bark of small dog, and
warns other animals of presence of predatory birds and mammals.
Cutter squirrel (Gravisciurus forfex)
Order: Rodents (Rodentia)
Family: Squirrels (Sciuridae)
Habitat: mixed forests of Central and Southern Europe.
Genus of true squirrels (Sciurus) had proved the viability and evolutionary
success. Having evolved in Oligocene, these rodents have successfully gone
through epoch of anthropogenous pressure – only some local endemic species
and species sensitive to anthropogenous influence had died out. The majority
of squirrel species had kept sufficient number and after disappearance of mankind
in forests their various descendants had appeared.
The descendant of common squirrel (Sciurus vulgaris) widespread in whole Eurasia
in human epoch lives in European forests; it is very large cutter squirrel.
It has kept a significant amount of attributes of ancestral species, differing
greatly only in size and heavier constitution. Its color of wool is the same
as at ancestor, but on red background on hips darker spots are scattered, and
white tip of tail is shaded with a belt of brown color. Also this animal has
dark ears.
Paws of cutter squirrel are rather shorter and thicker, rather than at squirrels
of human epoch. But tail of this rodent is longer, than at an ancestor, and
is covered with long fluffy wool.
Cutter squirrel is one of the largest species of forest animals of non-tropical
part of Eurasia. The length of its body is about 50 cm, and weight reaches
3 kgs. The increase of body size is connected to features of its diet. This
species of squirrels eats vegetative food only: since spring up to late autumn
basically leaves and thin branches, and conifer needles in winter. After feeding
of these rodents the tree looks clipped awkwardly – if the family of cutter
squirrels stays on it for a long time, they eat a significant part of young
shoots which they can reach. The inhabited tree, however, is never eaten round,
and dung is dumped on the ground from near growing trees frequently growing
in tens meters from the location of drey. Bacteria helping rodents to acquire
nutrients from rough forage live in a stomach and intestines of these squirrels.
Due to large caecum these squirrels can digest conifer needles. Oils and resinous
substances are not acquired by them; therefore in winter dung of cutter squirrel
has characteristic resinous smell.
Cutter squirrel lives in small groups: male, female and 1 or 2 recent litters.
The group occupies territory of about 15 square kilometers. Here on strongest
trees some dreys changing by pair of parent animals before birth of the next
litter are located.
Drey of these squirrels represents massive construction of bound rods and split
branches. Incisors of these rodents are strong enough to bite off branches
up to one centimeter thick and to split them lengthways. The drey entrance
is placed near the trunk; therefore it is difficult to get there even for these
animals. But it gives additional protection against predators, because it is
difficult for them to make the way in drey of these rodents. Inside the drey
there are some inhabited chambers littered with soft foliage and grass.
Because of their large size cutter squirrels climb not as dexterously, as their
ancestors. In manner of movement they resemble more monkeys or lemurs: they
run on four paws along horizontal branches and make rare jumps. When animals
move, tails are raised vertically and contrastly painted tip is visible. During
the feeding these squirrels sit on hinder legs and tail hangs downwards.
The courtship season at this species is very long, but animals show the greatest
activity since January till May. Pregnancy lasts till about 35 days, in litter
there are 3 – 4 cubs. In northern parts of area usually there is only one litter
per one year, in southern parts there are two ones. Mother feeds the posterity
with milk till approximately one and half month, and till about one month young
animals keep with parents. They reach sexual maturity at one and a half years.
Life expectancy makes till 20 years.
This species of mammals was discovered by Nick, the forum member.
Beringean
mountain squirrel (Tamiasciurus beringea)
Order: Rodents (Rodentia)
Family: Squirrels (Scuiridae)
Habitat: mountain forests of Beringia, northwest of North America.

Picture by Alexander Smyslov
Similarly to many other rodents, various squirrels, including
North American red squirrel (Tamiasciurus hudsonicus) have rather safely gone
through the period of anthropogenous pressure at the end of Holocene. Climatic
changes which had shifted southern border of coniferous forests zone farther
to the south appeared very favourable for this species.
But the ice age is a temporary phenomenon, and after thawing of glacier various
deciduous trees had started to spread to the north, having pushed coniferous
forests of North America closer to polar areas, and also to the west, to Beringia.
The part of the population of American red squirrel had receded to these forests
where had evolved to new species – to Beringean mountain squirrel.
Beringean mountain squirrel is rather large rodent – males weigh 350 – 400 grams,
females up to 300 – 350 g. It grows up to 25 – 30 centimeters long with fluffy
tail of the same length. On body fur is also longer and fluffier, rather than
at ancestor, even in summer because of colder climate in their inhabitancy –
northern and mountain (even high-mountainous) coniferous forests.
As against the ancestor, Beringean mountain squirrel is not red, and most likely
bluish-gray, like a color of stones and dark needles of trees in its inhabitancy;
also it has kept lighter belly, characteristic for its ancestor, and white rings
around of eyes have increased to large light spots on cheeks. On ears of these
squirrels short signal brushes of darker color grow, and at sexually mature
males also there is a dark strip along back; occasionally there are melanistic
individuals.
Beringean mountain squirrel is easily adapting animal with diverse diet. It
eats seeds of many species of coniferous trees, also some berries, mushrooms,
various insects and spiders, and ravages nests of small birds. This species
equally freely feels like both on trees, and on the ground therefore it has
superseded from northwest of North America various chipmunks conceding to Beringean
mountain squirrel in force and quickness.
This squirrel lives on trees where it builds a typical squirrel drey of branches
of conifers, or occupies empty bird nest, reconstructing it as it needs. From
within this drey is warmed by female’s underfur, and in spring female gives
birth up to 5 – 6 blind and helpless cubs; she brings them up alone and looks
after them within 2 months. Elder cubs remain with mother, but study to search
for food and to escape from enemies. At the age of 5 months squirrel cubs become
independent and leave her, and to one-year-old age they become sexually mature.
While female looks after posterity, male stays in her territory, gathering food
and feeding up the female. When cubs leave drey, male joins family. When the
posterity leaves the female, male also goes away from female’s territory.
Beringean mountain squirrel is sociable enough animal. Breeding pairs living
in neighbourhood relate to each other loyally enough, but in courtship period
between solitary males rather severe fights break out, accompanied by very painful
bites and long pursuits in tree crones.
If Beringian mountain squirrel will be eaten by nobody (and it is a prey of
various small and medium-sized predatory mammals and birds of coniferous forests
in mountains of Beringia), it can live up to 7 – 8 years.
To the east from Beringia, in northern forests of North America the species
related to Beringean mountain squirrel lives – taiga
squirrel (Tamiasciurus borealis). It differs from the mountain relative
in more habitual red color of wool, but in the rest it is practically similar
to previous species. Only white marks on its head are larger, and wool on stomach
is snow-white.
This species of mammals was discovered by Bhut, the forum member.
Farmer
hamster (Agrocricetus agricola)
Order: Rodents (Rodentia)
Family: Hamsters (Cricetidae)
Habitat: steppes of Southern Europe (Three-Rivers-Land) to the east up to Southern
Ural and Central Asia.
Rodents are the most numerous global widespread order of mammals. Their species
increased variety till all Cenozoic era and had got advantage in Holocene.
Human economic activity in many cases promoted prosperity of rodents. People
superseded and destroyed wild hoofed mammals, ploughed steppes and cultivated
large plantations of graminoids. People had also exterminated many feathered
and four-footed predators, and it had favoured to breeding of separate species
of rodents. They grasped new habitats freely, getting of advantage from human
activity.
In Neocene extensive spaces in Southern Europe, Middle and Central Asia had
turned to grassy plains in which local herbivores graze. Their typical representatives
are steppe species of harelopes and massive porcippula similar
to heavy-build pony. They “cut” steppe grasses by teeth, but do it not everywhere.
Here and there in steppe sites are located, where the grass is much denser
and higher rather than usually. Graminoids in these places differ in rather
massive ears rocking in the wind – it is a separate species, the descendant
of feral wheat many MY ago cultivated by people. These thickets differ in strict
monotony - among them plants of other species are practically absent. Obviously,
these plants were specially selected and carefully protected - such sites are
precisely separated from other grasses. Local herbivores for any reasons avoid
eating this grass which looks much more appetizing, than everywhere.
The puzzle is disclosed very simply when among high grasses there are small
stumpy mammals with rich longitudinal – striped fur of gray-brown color. It
wanders in thickets of high grasses, from time to time stopping and breaking
off stems lasting among grasses. This is one decision of some puzzles at once:
the rodent really looks after this graminoids. Actually, these thickets represent
its extensive fodder site. And due to it the animal is named farmer hamster.
This animal is a little bit smaller, rather than common hamster (Cricetus cricetus),
the usual inhabitant of Eurasian steppes of Holocene epoch. Probably, it is
its descendant though it is possible, that its ancestor could be one of small
species of Central-Asian hamsters. Colouring of farmer hamster combines two
patterns. The top side of body of this animal is cryptically colored: on gray-brown
background narrow black strips stretch. Having dropped to the ground, this
animal becomes completely imperceptible from afar for feathered and four-footed
predators. Coloring of stomach strongly differs from coloring of the back:
on stomach and cheeks there are wide cross black strips on white background.
This coloring becomes appreciable from apart when this small mammal rears to
eat something or simply to look round. At this animal there is constitution
typical for hamsters: large head, short thick trunk and very short tail, hardly
appreciable among wool.
This mammal differs in unique strategy of survival. It not simply gathers seeds
of plants which serve as food to it, and specially creates conditions for their
growth. It carefully keeps up a site of steppe around of the hole and in summer
selectively eats plants which do not belong to graminoids. As the result at
the “allotment” occupied by this hamster amounting about one hundred square
meters graminoids grow well, providing the hamster by grain for winter. More
often at sites of farmer hamster the hamster
wheat grows – it is the special genus of graminoids again “cultivated”
by the hamster after people and actually entered symbiosis relations. In summer
seeds of graminoids have not ripened yet, and animal eats grass – weeds growing
at its site. Also it eats various insects, involuntarily relieving fodder graminoids
from wreckers. In the autumn when seeds of grasses ripen, farmer hamster gathers
“crop”: it chooses the ripest ears and carefully husks them, scattering dust
on site and carrying off in burrow well cleared grain. Gradually at the territory
“cultivated” for a long time graminoids start to grow especially well, and
obviously prevail of other plants.
Farmer hamster also fertilizes ground with dung – it does not have specially
removed “toilet”, and the part of mineral substances comes back in ground with
its dung. Only in winter it leaves dung in special chamber of hole, and throws
it out outside in common with used litter in spring - it additionally fertilizes
ground in the territory this way.
Well-groomed sites of these mammals are favourite object of attention from
the part of other grain and grass lovers. At sites of farmer hamster in autumn
birds feed, and large herbivores occasionally visit them. But the farmer hamster
can render their repulse: despite of small growth, it is very much martial
creature. It frightens off smaller pilferers by loud squeals and high jumps.
Thus small mammal strongly puffs cheek pouches to seem larger. Such reception
does not work against the large herbivorous animals: they are strong enough
simply to crush this animal by hoof. But the hamster uses one more effective
means of protection against them: it lies down on back, showing to the uninvited
visitor contrastly colored belly, and shrilly squeals. If threat has not worked,
it sprinkles to the stranger stinky substances from strongly advanced anal
glands similarly to skunk.
The specific habit of life had transformed this animal to even greater “homebody”,
rather than hamsters of Holocene epoch. But it does not avoid the society of
neighbours, and prefers to settle in rarefied colonies. In total “allotments”
of different individuals of this species gather to the colony rather closely
to each other - their borders are divided with some meters. In the center of
colony there are “allotments” of strongest individuals – they are richest and
better protected from herbivores. But in the centre of colony lack of place
is sharply felt, and for each superfluous plot of land a real battle takes
place. Individuals at edges of colonies feel first that they are not unique
local herbivores. But it is possible for them to expand their “allotment”,
having “cultivated” more parts of steppe. The certain balance and order in
a colony is reached so.
Outside of breeding season each individual diligently marks borders of the
territory, sprinkling on grass secretions of anal glands. Only being ready
for pairing the female can leave the site to meet the male – in any other time
it would meet her with bites of sharp incisors. In courtship season the fodder
site is an original secondary sexual attribute: the female chooses the male
for pairing looking the size and “cultivating” degree of its allotments. After
pairing female returns to the site and rears posterity itself. Within one summer
the female rears two – three hatches of 4 – 6 cubs. The most part of young
animals settled from colony can not find suitable site, and perishes in teeth
and claws of local predators. To the autumn survived young hamsters find site
for life, and gather in hole the first stock for winter – basically seeds of
wild-growing grasses. In autumn the farmer hamster fattens up intensively and
grows fat strongly: fat may make up to half of weight of animal.
In winter farmer hamster falls into deep hibernation: the temperature of its
body is reduced practically up to an ambient temperature, and pulse decreases
up to several beatings per minute. Some times for winter animal wakes up to
eat some of seeds from the stocks. It is awake till some hours, yet will empty
intestines completely, and then falls runs into hibernation again.
Though farmer hamsters are convinced and bilious egoists, all of them keep
the contact with each other: between their holes there are burrows connecting
them to whole system. To tell the truth, more often other steppe animals dig
these courses, but hamsters willingly use them. In such tunnels animals leave
from chasing of predators. Being on the ground surface, animals “communicate”
with each other with the help of whistling, as suslics. In such way they warn
each other of occurrence of predator – usually steppe
eagleraven or predatory mammal zibetonyx hunt
them.
The biggest complexity in life of farmer hamster is derivated by its habit
of life, to be more exact – by the way of “agriculture”. At their sites only
the unique species of graminoids frequently grow, therefore animals endure
heavy time when cultivated plants are attacked with illnesses and parasites.
On “hamster wheat” the ergot fungus grows often. But small ergot infection
of graminoids does not harm to animal – when this fungus appears, hamster specially
searches for such ears, chews fungus off from them eats. Ergot poison renders
intoxicating effect to these rodents – in the beginning animal becomes easily
excitable and aggressive, then gets into the hole and sleeps for a long time.
The activity of farmer hamster constrains the distribution of ergot, but at
mass disease of graminoids they can not cope with it, and the part of crop
perishes. After flare up of plant desease in colonies of farmer hamsters the
famine comes, and number of populations may decrease in tens times. And in
such situation the part of colony members simply leaves on new places where
new “plantations” appear in some years.
This species of animals is discovered by Momus, the forum member.
Whistling
hamster (Buccacricetus stridor)
Order: Rodents (Rodentia)
Family: Hamsters (Cricetidae)
Habitat: steppes of Tree-Rivers-Land, thickets of grasses.
Some species of rodents had received great advantages in survival in human
epoch. Expansion of agricultural areas had allowed them to settle widely; the
cultivating of cereals had provided constant and uninterrupted source of food,
and human presence and its activity had strongly reduced number of predators
in habitats of rodents. Disappearance of mankind has restored natural balance
of environment during several hundreds thousand years – the extensive areas
of monocultures had disappeared, predators had appeared, and gradual formation
of new ecosystems instead of ones destroyed by people began. Evolution had
gone in the natural way again, and at new species of animals new adaptations
for survival began to develop.
In extensive steppes of Tree-Rivers-Land reaching along northern and eastern
coasts of Fourseas, rodents are prevailing group of mammals. Quickly evolving,
they had formed many species, differing by requirements to inhabitancy. Representatives
of hamster family differ from other rodents by ability to gather the large
stock of forage for winter. One of their species, farmer
hamster, even is engaged in original “cultivating” of one
species of graminoids on its fodder area. But other hamsters lead more
traditional way of life.
Many predators live in steppes. One of widely widespread species is zibetonyx,
cursorial species of viverrids, the most northern species of this family. It
represents danger to many small rodents, but one of the large hamsters, living
in steppe, is not afraid of its attack: it is armed with sharp incisors and
is able to protect itself. This species is whistling hamster.
Whistling hamster is rather large rodent: the length of its body is about 40
centimeters, and weight reaches 2 kgs. By appearance it is similar to other
hamsters: it is short-legged and massive one with large head. Its wool on back,
nape and top part of neck has camouflage color – golden-yellow with darker
faltering longitudinal strips. The bottom part of body sharply differs by colouring:
stomach and forward part of head are white with sparse black spots.
The most remarkable feature of whistling hamster is its cheek pouches. Usually
they are used for dragging stocks of forage to the hole, but at the attack
they are used in completely unusual mode. The aggressive animal rears on hind
legs and starts to puff cheek pouches. Their skin is very extensable, and due
to it this rodent seems larger and more impressive. And then remembered colouring
of wool is expressed in full degree – on skin covering cheek pouches - cross-striped
pattern becomes visible. Skin under wool is also pigmented at the places where
black hairs grow; therefore this pattern stays appreciable, even when cheek
pouches are puffed completely. This is the first warning to predator. Usually
it is enough to the skilled predator to stop getting into touch with this rodent.
If attempts of attack proceed, hamster contracts ring muscles encircling cheek
pouches, and blows them off. Thus loud whistle is hearing. This is more powerful
prevention to predators. After that hamster rushes on the enemy and seizes
it by incisors. It happens, these animals attack even porcippulas –
large and harmless herbivores.
Whistling hamsters live solitarily in rather extensive territories – between
holes of separate individuals there is a distance not less than 200 meters.
Warning relatives that the territory is occupied, the owner of territory whistles
from time to time, puffing cheek pouches. Having heard its whistling, neighbours
usually also respond.
This hamster gathers seeds of graminoids in its extensive individual territory,
and to the end of an autumn it gathers in underground “pantry” up to 30 kgs
of grain. In addition to it, animal digs out and eats tubers and rhizomes of
various plants, a part from which it also drags to “pantry”. To winter this
hamster is strongly eaten off and runs into hibernation when autumn rains begin,
changed further by light frost and snow. For winter it wakes up many times
and eats a part of the stocks. In spring whistling hamster searches mushrooms
growing in steppe, and eats up the rests of winter stocks. Usually they suffice
up to growth of young grass.
In spring the courtship season begins at these rodents. Demonstration of puffed
cheek pouches and whistling are important elements of courtship ritual of whistling
hamster. Usually male comes itself to the territory of female ready to pairing
and starts to court after her, following her with blown up cheek pouches and
whistling. If near one female some males meet, they arrange a kind of “duel”,
trying to whistle louder and longer, than the competitor. If it does not help
to define superiority, combat begins.
Within one year the female gives rise to 2 litters of 3 – 4 cubs. Young animals
of the second litter winter together with the female and in common prepare
forage in autumn. The young growth of summer litter has time to find territory
suitable for life, to dig a hole and to prepare enough amount of food for successful
wintering. The next spring young animals from both litters can take part in
breeding.
The idea about existence of this species of animals was proposed by Simon, the forum member.
Ayapuh
(Pseudolutra ayapuh)
Order: Rodents (Rodentia)
Family: Neoichthyomyids (Neoichthyomyidae)
Habitat: the south of South America, Tierra del Fuego, Pacific coast.
In human epoch otters were the basic predatory mammals of fresh water reservoirs.
These animals were widely settled at all continents, except for Australia and
Antarctica. One species of otters had developed sea habitats in northern part
of Pacific Ocean. But in human epoch destruction of habitats and hunting for
otters for the sake of fur had reduced their number in great degree, and they
had not survived to Neocene. After human disappearance at various continents
the ecological niche of otters was occupied by animals from various systematic
groups. In South America otterodents, descendants of fish-eating hamsters of
genus Ichthyomys, became the most successful ones in development of aquatic
inhabitancy. Various species of these large carnivorous animals have mastered
reservoirs of various types, from tropics up to Tierra del Fuego.
Ayapuh is one of otterodent species, which, as against the majority of species
of this group, lives in brackish and sea water. The name of this animal is a
name of otter from fairy tales of Indians of Tierra del Fuego. Ayapuh does not
concede Amazonian otterodent
in length, but differs in more massive and thickset constitution. It has short
and wide tail of rounded outlines, large body, short neck and heavy head: it
is connected to necessity of heat preservation in conditions of colder climate.
Fur of ayapuh has rusty-brown color; at separate animals lower jaw and throat
are white, but more often between forepaws there is only small white spot. Fur
of ayapuh is very rich. The animal looks after it for a long time and greases
it plentifully with secretions of glands located in armpits. Due to care fur
gets additional water-repellent properties and keeps the air layer saving animal
from overcooling under water in winter time.
Hands, feet and tail of animal are covered with short thin hair, and through
naked skin animal could lose a lot of heat. But in paws and tail blood vessels
form a countercurrent system which helps to keep heat.
The dental system of ayapuh is typical for rodents – it includes two pairs of
sharp incisors of reddish-orange color representing its main instrument of getting
of food and protection against enemies. The length of incisors in lower jaw
of animal reaches 7 centimeters. For search of food ayapuh can dive to depth
up to 20 – 30 meters. Swimming under water, animal makes wavy movements by backbone
in vertical plane. Main movement bodies at this species are flattened tail and
webby hinder legs. Animal uses forepaws to feel sea bottom and to overturn stones
in searches of prey. Ayapuh frequently hunts near to large herbivorous mammals
algoceti when families of these animals
are fed in thickets of seaweed. At this time ayapuh catches fishes and crustaceans
frightened at movements of giants. Also strong teeth allow ayapuh eating echinoderms
– sea urchins and starfishes.
Under water animal searches for prey with the help of sight, but in muddy water
or in twilight it uses long sensitive whiskers.
Ayapuh is social species: this animal settles at sea coast in small groups including
up to five breeding pairs and several single individuals, young or old. Breeding
pair at ayapuh forms to some breeding seasons, and sometimes is kept till whole
life. Animals look after one after another, clearing fur of the partner, or
feeding it up with a part of prey.
In storm ayapuh hides between stones and in natural shelters like caves. Only
for posterity care female builds a hole, digging it with the help of forepaws.
It frequently simply digs holes built by sea birds, expands and converts them.
Once a year, in spring (in Southern Hemisphere in October – beginning of November),
female brings posterity – from two to four blind and deaf cubs covered with
short wool. They quickly acquire rich wool, but begin to hear only at the age
of 5 days, and eyes open at them at the age of 8 – 9 days. At monthly age cubs
start to leave their shelter; and bi-monthly ones study swimming. At the age
of two years young female is ready to breeding. Life expectancy reaches 17 –
20 years.
Plains
Hopping Mouse (Neonotomys agilis)
Order: Rodents (Rodentia)
Family: Mice (Muridae)
Habitat: Meganesia, tropical scrubland and savannah which extends in the centre
and west of the continent.
Success of rodents in the age of man, and subsequently in the Neocene, is undeniable.
It was only in the late Tertiary that rodents finally reached the bastion of
Australia from Asia, and much later further kinds were introduced by man in
the modern age. The original kinds of Australian rodent have proved viable even
against introduced kinds, in the Neocene, with specialized forms prevailing
in some places.
In the open country of the tropical savannah, lives the Plains Hopping Mouse,
a successful descendant of the current hopping mouse (Notomys). Specialized
towards an agile, hopping and jumping gait, this adaptation is even stronger
in this new form, than its ancestor. The hopping mouse is slightly larger than
its predecessor, being up to 5 inches long from head to rump, and weighing up
to 40 grams, the tail consists of an additional 5 to 6 inches. Its hind legs
and upper part of the foot are long, and the creature usually stands on the
tips of its toes, except when at rest. Forelimbs are short and dexterous to
aid in feeding; the tail is long and held against the ground, with a tuft of
fur at the end. Coat of fur is a pale fawn grey on top, and a whitish cream
underneath, the brush of the tail is darker. The face is rather similar to its
ancestor, but the ears have become larger in order to aid in dissipating heat,
hearing is very acute. This animal’s diet consists almost solely of the seeds
of grasses and herbs, but soft tips of new grass growth are also eaten. This
rodent is completely nocturnal, sleeping in a burrow throughout the heat of
the day.
These animals live generally in small family groups that share a burrow, which
is usually dug concealed under a tussock of grass or small hummock of soil.
Predators include birds like hawks and owls, as well as Dasyurid marsupials
like the marsupial hermin. Breeding takes place frequently and at any time of
year, but spring is preferred, pregnancy lasts about 40 days, litters consist
of up to 7 babies, which take up to 2.5 months to reach sexual maturity. Lifespan
is up to 3 and a half years.
A close relative is the Desert
Hopping Mouse (Neonotomys albipes), which inhabits the arid deserts
that exist in the far west of the continent of Meganesia. The animal is in general
much paler than its relative, being a sandy cream colour above, with white feet
and underparts and a ginger coloured tail tuft. It is also slightly smaller,
with proportionately larger ears, which are naked on the inner edge, to dissipate
heat. Habits and feeding are generally similar, consisting of the seeds of many
kinds of sparse desert grasses and sedges, but by necessity they eat the fleshy
parts of plants for water more regularly. Breeding is slightly different, being
regular but most often coinciding with wet periods and flushes of growth.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Murray
Shell Rat (Durohydromys aquatica)
Order: Rodents (Rodentia)
Family: Mice (Muridae)
Habitat: Found in the Murray River of southeastern Meganesia, depths of water,
water margins, swamps.
The rodents native to Australia stood fairly well against introduced ubiquitous
rats and mice brought by humans. Specialized forms existed, and continue to
exist in the Neocene.
Murray Shell Rat is a descendant of the Water Rat or Rakali (Hydromys). Animal
is similar in size to its ancestor, reaching 12 inches in length, and up to
a kilogram in weight, with females being smaller than males. Outwardly it looks
similar to its ancestor, original partway in appearance between otter and rat,
with long tail and broad webbed hind feet, fur is velvety and repellent to water.
Color is a general chocolate colour above, with a yellowish buff belly, the
tail is tipped with white. It differs from its ancestor in blockier robust head
bearing even bigger incisors, with a projecting face and profuse whiskers. This
is related to diet, which is predominately freshwater molluscs such as snails
and bivalves, as well as crayfish, shrimp and occasionally small fish. Hard
shelled prey are broken open by gnawing the outside until a large enough hole
exists to pry the shell further open. Individuals may habitually make middens
in the place where they prefer to consume their food, usually just outside their
nest.
This rodent is most active in early morning and late afternoon, individuals
are very territorial, fighting over space when they meet each other. Moulting
occurs in summer and autumn during the warmer weather. Breeding depends on seasonal
abundance of food, and the male will usually find a female with which he may
sire more than one litter if the young die, or if it is a good season. This
rodent constructs burrows in riverbanks, making a nest of reeds and grass in
which to raise the babies. Females are sexually mature at one year, mating occurs
from August to October, gestation is 30 days. Females have 4 nipples on the
belly and usually produce four young per litter. They are solitary outside the
breeding season, and are generally aggressive towards the opposite sex. Any
one area will contain multiple individuals that compete and antagonize each
other based on age and sex. Young reach full size within a year. Lifespan is
short, 3 to 4 years, and they are commonly preyed upon by raptorial birds and
occasionally large fish.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Noronha
rat (Rattus noronhensis)
Order: Rodents (Rodentia)
Family: Mice and rats (Muridae)
Habitat: Archipelago of Fernando de Noronha.
While humans purposefully introduced some species in new habitats, others were
inadvertently introduced. The black rat (Rattus rattus) followed the humans
to several places, benefiting from their presence. When they were extinct, some
of these rodents were able to survive and continue their evolution. In the Archipelago
of Fernando de Noronha, the populations of the black rat that survived evolved
into an endemic species, the Noronha rat.
The Noronha rat has 12 cm of body length, with a tail of 9 cm. They have strong
forelimbs that are used to excavate the ground to make tunnels to be used as
their dens. It has light brown fur with a white underside.
This species is an omnivore and eats a wide range of foods, including seeds,
fruit, stems, leaves, fungi, and a variety of invertebrates. Some individuals
will also occasionally eat eggs and newborn chicks of seabirds that are left
unprotected. However, they need to be careful when approaching the colonies.
The adults that nidify in the Archipelago had learned the danger of the rodents,
being aggressive with them. The large species of bird can even consider them
as a good snack.
Differently from their ancestor, the Noronha rat is monogamous and highly territorial.
The limited resources left in the Archipelago after the disappearance of the
humans forced these rodents to fight against each other for what they could
find. This way, only the mated pair forage together and they defend their dens
fiercely against intruders.
Their greatest predator is the gatito,
which is capable to invade their dens and to kill them while inside it. However,
these rodents don’t fall without a fight. Their fights against the feline could
be considered titanic for animals of such small proportion, but even then the
gatito ends winning most of the time.
The Noronha rat can breed throughout the year if conditions are suitable, with
a female producing up to five litters a year. The gestation period is only 21
days, and litters are formed by 7 to 14. The competition among the young is
fierce, with weak siblings of large litters perishing due to it. In these cases,
the parents will eat the dead. They reach sexual maturity in about five weeks,
which is when they are expelled from the den by their parents. When there is
enough food, the female becomes fertile again three weeks after giving birth.
The maximum life span is three years, although most barely manage to one. Unlike
their ancestors, they aren’t an invasive species in the Archipelago anymore.
The local fauna have evolved together with them and know that they can be a
good food source. Beyond the gatito, local
herons will also exploit them as such.
This mammal species was discovered by João Vitor Coutinho, Brazil.
Hawaiian
bamboo rat (Bambusiphagomys tector)
Order: Rodents (Rodentia)
Family: Murines (Muridae)

Picture by Alexander Smyslov
Habitat: bamboo woods of east slopes of mountains on Big Island (Hawaii).
In human epoch birds dwelt on Hawaiian Islands were a fine example of adaptive
radiation - the phenomena at which from one ancestor many descendants evolve.
They are connected by relationship, but adapted to different inhabitancies
and having various shape. But indigenous species of Hawaiian fauna have irrevocably
died out, not having sustained a competition with newcomer species introduced
by people. And newcomers, in turn, roughly began to evolve, showing on the
example of the same evolutionary phenomenon.
Three species of rats have got to Hawaii on human fault: black (Rattus rattus),
brown (R. norvegicus) and indigenous Pacific rat (R. exulans). The third species
was quickly superseded by descendants of larger neighbours, and had died out
till several subsequent thousand years. But two remained species had actively
evolved, having given many various descendants distinguished by appearance,
behavior and habit of life. Descendants of brown rats became mainly ground
animals, and black rat – wood inhabitants.
A few species of mammals live in bamboo woods. In this place it is difficult
to move quickly - trunks of bamboo form almost solid walls, and between them
the large animal simply cannot walk through. But smaller species of rodents,
adapted to existence in this original biotope, prosper. At first it is difficult
to find out about presence of these small mammals in dense bamboo wood, but
gradually these animals find themselves out. They run on wood litter, rearing
from time to time among trunks, and showing improbable, practically squirrel-like
speed, climb up on smooth and firm stalks of this graminoid where even for
cat it would be difficult to seize against something. It is the Hawaiian bamboo
rat, the descendant of black rat (Rattus rattus).
Hawaiian bamboo rats are small rodents, like only the young grey rat by size.
They have kept the appearance typical for representatives of family, but have
got some features, permitting to survive in specific biotope. Representatives
of this genus of rodents meet exclusively in bamboo thickets, and are not found
anywhere more. On each island of archipelago there is the species of these
rats distinguished from others in size and features of coloring.
The Hawaiian bamboo rat dexterously swarms up stalks of bamboo. It has tenacious
claws, but it is not enough to it to show such quickness like it swarms up
trunks of bamboo. On palms and feet of rodent the skin forms small holes acting
like suckers. With their help rat can quickly run on smooth trunks of bamboo.
To improve action of suckers, sudoriferous glands on their edges are hypertrophied,
and allocate stickier secret providing the best sticking to trunk.
The short wool of the rat is colored yellowish – brown. On back of rodent there
are dark longitudinal stripes – they mask this animal in the world of vertical
trunks of bamboo. The amount of strips and their pattern considerably vary
at different individuals.
Hawaiian bamboo rat completely depends mainly on large species of bamboo with
thick trunks. Such choice of habitat is connected with the fact that all species
of Hawaiian bamboo rats arrange shelters, simply gnawing out apertures in ripen
hollow trunks of bamboo. After rats birds use their tree-trunk hollow: on Hawaii
there are no woodpeckers, and, except for rodents, there is nobody able to
make tree-trunk hollows here. And the natural tree-trunk hollows formed in
trunks of some trees, does not suffice for all. Therefore some birds depend
on well-being of bamboo rats.
This rodent has one more important “duty”: because these rats eat young sprouts
of bamboo, they thin out thickets of this graminoid in such way. To gnaw hollows
and to chew firm stalks of bamboo, at this rodent large incisors and short
strong jaws were developed. Cheekbones of this rodent are wide, and jaw muscles
are very strong.
All bamboo rats are solitary animals preserving the territory from neighbours.
Only during breeding season males can come in territory of females for pairing.
Usually per one year at the female it happens three litters of 5 - 6 cubs.
They are born naked and blind, but develop rather quickly. Bi-monthly animals
leave mother, and search to themselves for suitable place for life. Usually
in territory of each adult animal it happens some shelters gnawed in trunks
of bamboo, and young animals frequently lodge in them, choosing ones in which
the lawful owner of territory was not for a long time.
Trunks of bamboo are very strong, and animals settling in them are perfectly
protected from many enemies. But occasionally the population of Hawaiian bamboo
rats has not best times. During flowering of bamboo (one - two times per one
century) number of Hawaiian bamboo rats is considerably reduced: the bamboo
dies off after flowering simultaneously in big territories, and rats sometimes
appear without food among a congestion of died out trunks of these graminoids.
The survived rats in small amount pass to other forage, eating young sprouts
of bamboo, slowing down naturally its distribution. Also at lack of the basic
forage they eat fleshy rhizomes and the bottom part of stalks of other grasses,
including other species of bamboo.
On other islands of the Hawaiian archipelago close species of bamboo rats live:

Picture by Alexander Smyslov
Black-backed bamboo rat (Bambusiphagomys melanodorsus) lives on Molokai Island. It is the small species representing by the size something mean between rat and mouse. This rodent lives on medium-sized species of bamboo and at tops of large species, choosing the internode of suitable size. The basic colouring of its wool is sandy – yellow, and on back from a nape up to the basis of tail one wide black strip stretches. Occasionally on sides there are pale longitudinal strokes.

Picture by Alexander Smyslov
Narrow-striped bamboo rat (B. angustizonus) is also small species meeting on Maui Island in thickets of small-leafed species of bamboo. It arranges shelter in the basis of trunks in the thickest internodes, but feeds mainly at tops of bamboo, eating insects and young leaves of this grass. On its back set of narrow longitudinal stripes of brown color stretches on grayish - white background; stomach is white.

Picture by Alexander Smyslov
Half-striped bamboo rat (B. hemifasciatus) lives on Lanai Island. This rat differs from close species in remarkable colouring: on forward part of body strips are absent; they begin only on middle of back, and cover waist and hips of animal. The basic colouring of this animal is light grey with black strips. On head and cheeks of this rat there are white spots using for recognition of congeners. Except for colouring, this species differs in more graceful addition: lengthened body, short forepaws, narrow muzzle and long tail. This rat reaches usual length of grey rat, but weighs approximately twice less than it.
Hawaiian gatherer rat (Pacifirattus collector)
Order: Rodents (Rodentia)
Family: Murines (Muridae)
Habitat: Hawaiian Islands, rain tropical woods.
Hawaiian islands differ from set of other islands in rather big area and huge
variety of possible habitats given to live creatures. The complex mountain
relief creates a plenty of rather isolated habitats and as the consequence
of it, set of various though also closely related species of small live creatures
lives on Hawaii. They differ from each other in features of appearance, way
of life and behavior. Such variety gets sometimes the extremely fantastical
forms.
Mammal of Hawaiian Islands evolved mainly from animals introduced by people.
And rodents reached the greatest variety among them. Having high speed of alternation
of generations and small size, they could occupy various ecological niches.
Island descendants of rats differ from each other in size, way of life and
features of behavior.
In human epoch in territory of Australia and New Guinea bower birds, and in
pampas of South America large rodents viscachas lived. These animals had differed
in one interesting feature of behavior: having predilection for various bright
objects, they collected them, arranging in special places original “exhibitions”.
The similar creature lives on Hawaii in Neocene epoch. It arranges round wicker
drays in branches of bushes, and places “collections” of shining exoskeletons
of beetles and snail shells around of them on broken off knots. Owners of building,
the breeding couple, from time to time flash in foliage, hastening to the dray
or back.
This four-pawed “collector” is Hawaiian gatherer rat, a creature like large
grey rat (Rattus norvegicus) by size, and by the way it is its strict descendant.
This rodent leads semi-climbing habit of life, swarms up bushes well, and arranges
drays at small height from the ground. For protection against enemies this
rat builds drays in prickly bushes.
Being a day time animal, Hawaiian gatherer rat has the bright colouring appreciably
distinguished from monotonous and dim colouring of ancestor. At this rat there
are red back with longitudinal lines of dark speckles, brown head and white
spots near black ears. Also there are white points above eyes. At this rat
there are large eyes and wide rounded ears through which there leaves surplus
of heat in hot day. Paws are armed with sharp tenacious claws; joints are very
mobile, hand and foot can turn aside to the significant corner.
This rat is omnivorous, but prefers food of animal origin. It eats various
snails including predatory ones, at an opportunity ravages nests of birds and
eats insects. Frequently this rat eats large beetles, and drags their shells
to the dray to supplement its “collection”. This rodent lives at edges and
damaged sites of wood where bushes plentifully grow. It is able to swarm up
branches perfectly, and here prehensile tail making more of half of the general
length of rodent helps the rodent. The tail is naked, covered with grey skin
with separate bristles. It is used as the balance weight more often. Jumping
from branch to another, this rat can drive by tail, or rotates it to turn over
in air and to land on paws like the cat. On the bottom side of tail the cross
horn combs permitting to catch for bark stronger when rat moves in bush, are
advanced.
Dray of this rodent is similar to squirrel’s one: it is spherical, wattled
of rods, arranged in densest part of bush thickets. Similar drays martenrats from Tonga islands plait independently of them, but at the Hawaiian rats, as
against to Tonga ones, the entrance to dray in turned not from below, but sideways.
This rat frequently alters for habitation bird's nests, preliminary having
attacked on owners and having ravaged their clutch. Drays made among prickly
bushes are especially appreciated. This species lodges in drays in pairs kept
for some seasons, and frequently till all life. For strengthening of relations
between partners rats use various symbolical “gifts”, and by their amount near
the dray it is possible to judge force of matrimonial connections: the strong
pair in the prime of life collects many various “gifts”, and successfully protects
them from encroachments on the part of congeners. Usually these rodents collect
bright snail shells under “butcheries” of local hookbill bird eating snails,
and decorate with them an entrance to the shelter. For the organization of
“exhibition” pair of rats gnaws off a part of branches near to bush in which
the dray is arranged, clears them of foliage and bark, and picks on them shells,
dead beetles of bright coloring and if their number is too little – live flowers.
The single male, having won itself a bush suitable for habitation, first of
all organizes “exhibition”, and arranges dray only later. The more bright and
large will be such collection of bright objects, the more obviously force and
dexterity of animal had collected and had kept it. Different pairs have different
predilections - near some drays there is more live flowers, near others shells
or elytrums of beetles are presented. Pairs at gatherer rats are to no small
degree formed on generality of “aesthetic views” of partners. The “exhibition”
is regularly updated: the withered flowers and dried berries are replaced by
fresh ones, and new “exhibits” found, or stolen at neighbours from time to
time are added there.
During courtship displays the male drags in teeth bright objects from “collection”,
showing them to female. It serves as original “substitute” of secondary sexual
attributes. Partners from amicable pair in common steal ornaments at neighbours
and protect “collections” from ravaging. If necessary these rodents can drive
off from bushes even local huge herbivore birds (giant forest geese), frightening
them by shrill squeal. If birds do not recede, rodents even attack them, biting
birds in unprotected paws. Some pairs of these rodents can operate in common
against the large enemy.
Seasonal prevalence in breeding of the Hawaiian gatherer rats is not present.
At any time year in drays of these rodents it is possible to find cubs of various
ages. In litter of this rodent three - four times per one year up to eight
- eleven naked blind cubs appear. The female cares after them, leaving posterity
only for feeding, and the male is completely occupied with protection of fodder
territory and “collection” from neighbours. At week age the young growth becomes
covered by wool, for the twelfth day starts to see and hear. Up to monthly
age rat cubs stay in dray, and later start to play already outside of home:
swarm up branches, drag small objects, trying to steal them at each other.
Bi-monthly cubs already receive punishment from parents, and one by one abandon
parental territory. In youth, not having an own home, they are very vulnerable,
and at this time perish from various predators. Up to half-year age, when the
young animal becomes strong enough to win to itself territory, only few individuals
survive. Life expectancy of these rodents is short – only few animals live
till three years, and the majority does not live even till two years.
Mangrove builder rat (Architectorattus piscatorius)
Order: Rodents (Rodentia)
Family: Murines (Muridae)
Habitat: Meganesia, mangrove forests of Arafura Lake.
Rodents were only terrestrial placental mammals in Australia before the human
arrival to this continent. Under human influence the fauna of Australia had
substantially lost the originality, and a plenty of non-native species introduced
from other continents had appeared in it. During the time pased from the moment
of extinction of mankind, some lines of placentals had declined, and others,
on the contrary, continued the prospering. Rodents are especially succeeding
group among them. The type of anatomy, characteristic for rats and mice, appeared
very successful, and numerous rats had settled in various biotops of Meganesia.
One of their species due to versatility of behaviour had developed mangrove
forests separating by thousand-kilometre wide barrier Arafura Lake from the
ocean. It is very specific environment where small islets of land alternate
with impassable marshes and rich mattings of roots and branches of mangrove
trees.
The large species of rats well adapted to life in such inhabitancy lives in
branches of mangrove trees. Its settlements represent “towns” of several tens
of nests looking like small huts arranged on branches. Because of this feature
the animal is named mangrove builder rat.
This mammal is medium-sized aquatic rodent: its body length is about 30 cm.
It has anatomy typical for rats, long flattened muzzle and short forepaws with
mobile fingers and sharp claws. Rear legs are big and strong, and have membranes
between toes. Auricles are small. When rat dives, acoustic duct is closed by
the special skin valve. Mangrove builder rat has long tail, which serves as
a counterbalance when it swarms up branches. Wool of animal is velvety and
rich, but short. This rat greases it with fat secretions of special glands,
and spends a lot of time cleaning it. Cleaning of wool at these animals is
a mark of mutual attachment, and the individuals standing below at the scale
of rank express submission to stronger individual this way. The top part of
body of mangrove biulder rat is colored beige; throat and breast are white.
This rat eats fish, crabs and insects, and also consumes fruits and young leaves
of mangrove trees. It is able to swim and dive well, and during the inflow
it hunts for fish and other water animals. Swimming under water this rodent
presses forepaws to breast and rows by hinder legs. Mangrove builder rat leads
social habit of life and settles among mangrove thickets in colonies numbering
some tens of individuals. The inhabiting in holes and hollows traditional for
rats does not fit to conditions of mangrove forest – ground is regularly flooded
here, and hollows in tree trunks are not so extensive to contain the numerous
colonies of animals. Therefore during the evolution at these rats the special
form of building behaviour had developed. Each breeding pair constructs for
itself the original dwelling – primitive conic-shaped “tent” which is located
above a level of the highest inflow. By this feature mangrove builder rat is
similar to martenrat from Tonga Islands – the rodent developed during the evolution
process the similar kind of shelters independently of it. In the basis of “tent”
there is a small wum platform made by animals of fibrous leaves of palm tree
and pandanus, or of bark of mangrove tree sprouts. Above it the dome of arched
bent rods is erected, on which the roof made of leaves is attached. The ready
construction has conic shape. Houses of mangrove builder rats form a kind of
“village” where up to ten pairs of adult animals and numerous young ones live.
At the exhaustion of fodder resources the colony abandons “village” and moves
to new places where new constructions are quickly made.
Within one year the pair of adult rats may bring into the world up to four
litters of 6 – 8 cubs. They develop quickly and already at the age of 6 – 7
weeks become independent and make their own constructions at the edge of “village”.
At the age of half-year they become sexually mature. Life expectancy at these
animals does not exceed three years.
Kapo (Kapo hawaiiensis)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: Hawaii Islands, Big Island.

Picture by Alexander Smyslov
Before the people arriving to Hawaii mammals of this archipelago
were presented only by bats and seals. In common with people new animals appeared
at the islands,
and among them there was a rat – one of the first ground mammals appeared
in Hawaii. Their occurrence has resulted in disappearance of significant number
of bird species existed at Hawaiian archipelago. Despite of human efforts
directed
to extermination of these animals, rats have not died out on Hawaii, and
managed go easily through the period of global ecological crisis. After stabilization
of climate various species of rats differing from each other in ecology and
way of life had evolved on Hawaii. The largest descendant of introduced rats
is kapo, animal similar to South American rodents agouti and paca, known
in
human epoch.
Kapo is very large species of rodents: the adult animal weighs about 5 kgs
at body length about 40 cm. In its constitution kapo resembles a pig – this
animal has short and muscled body, large head with narrow mobile muzzle and
long whiskers, rather long legs and short thin tail. Colouring of this animal
helps this rodent to hide in vegetation of underbrush: primary colour of
kapo wool is grey, along back some faltering black strips stretch, and the
top part
of head is black. This rodent has rather large eyes. Above kapo’s eyes there
are two big white spots helping this animal to distinguish congeners. Auricles
of kapo are large, rounded and rather wide – they serve for body cooling
in hot climate. From the outside edges of kapo’s auricles are covered with
white
wool that permits the animals to notice each other in shady underbrush. This
species of rodents is almost exclusively vegetarian though it does not miss
an opportunity to catch large insect, crab or snail. Its basic food includes
seeds, roots of grassy plants and green. Kapo moves on ground on straightened
legs. It can run very quickly, suddenly changing direction of run for protection
against enemies. This animal is heavy enough, and it is not able to climb
on trees. If the animal can not escape from the enemy, it protects itself,
putting
deep lacerations by incisors.
Kapo is especially active in twilight and in early morning, and prefers to
hide in various shelters in the afternoon. In middle of night kapo also has
a rest within several hours. These animals dig short holes with a characteristic
bend: the beginning of hole is almost horizontal, then it makes a sharp bend
downwards, and proceeds horizontally or slightly inclined upwards. It is
very difficult to dig out such hole, and it serves as good shelter for animal.
Kapos
make “stores” of seeds in forest, digging under roots of trees small holes
and filling them with gathered seeds. They especially frequently reserve
large seeds of palm trees and leguminous plants. The part of stocks will
be eaten,
but not completely – kapos frequently do not pull out all seeds from “store”,
or forget the stock at all if find enough food. And then through any time
in forest whole bunches of sprouts of various plants appear. Similarly to
South
American rodents, Hawaiian kapos carry seeds of palms and other trees with
large and heavy seeds. Hence there is their name: Kapo is a god of fertility
at ancient Hawaiians.
Kapos live in pairs or in small groups: pair of adult animals and their posterity.
Each pair has the certain territory where the shelter is arranged, but is
fed in the distance from it. Inhabited holes of kapo are made nearby one
from another,
and individuals of different pairs communicate with each other with the help
of sounds and movements of body. The pair is formed for some years, and brings
posterity two times per one year. In litter at kapo there are about five
naked blind cubs. Within two months of life they acquire wool, grow considerably
and become more mobile. At them eyes open, they start to get out of a hole
and to learn world around. In the beginning of the third month of life young
kapos start to lead the same way of life as adult individuals, and completely
pass to food of adult animals. At the age of 20 months young animals reach
the size of adults and can give posterity. Life expectancy of kapo rarely
exceeds
five years.
Poison-toothed
giant rat (Cobrodus toxicodus)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: Southeast Asia, tropical forests, the sites overgrown with grassy vegetation.

Picture by Alexey Tatarinov
In tropical forests of Neocene epoch rodents represent one
of the most typical groups of mammals. These animals have great range of habitats
– from underbrush up to forest canopy – and occupy various ecological niches.
Due to ecological plasticity rodents had successfully gone through the time
of anthropogenous pressure and had reached blossoming in Neocene epoch. Their
adaptations for survival are very diverse, and some Neocene species have developed
adaptations unknown in human epoch.
The massive sluggish rodents similar a little to badgers with long hairless
nonflexible tails live in forests of Southeast Asia. They are large descendants
of any from numerous species of rats (Rattus); the body length of adult individual
reaches 50 – 60 cm (and tail is about 40 cm long), and weight is up to 7 – 8
kg. They live in family couples and constantly search for food together. The
body of these rodents is covered with rigid wool of rusty-brown color, lighter
on stomach. Head is colored very brightly and sharply contrasts with body colouring:
on white background longitudinal black strips stretch – some narrow strips on
forehead and one wide stroke on each cheek. Such colouring is very well perceived
by predators lack of color vision, and is remembered by them for a long time
after acquaintance to protective adaptations of these rodents. The bite of these
rodents is poisonous.
Poisonous mammals are known in human epoch – they are monotremes and some insectivores.
They secreted their own poison from special glands: duck-bill from spurs on
legs, and shrews from modified salivary glands. Poisonous
shrew lives in Neocene in forests of Europe. Poison-toothed giant rat differs
from them in origin of poison: in modified salivary glands of this rodent the
poisons received from plants are accumulated. At an attack of predator poison-toothed
giant rat first of all displays to the enemy readiness to protect itself, widely
opening mouth and bristling wool on head. If the enemy continues attempts of
attack, the rodent counterattacks it and bites strongly. On top incisors of
poison-toothed giant rat from external edge there is a groove which gradually
goes deeper when animal is older and turns to closed channel. The duct of specialized
salivary gland opens in it. Obviously, at ancestors of these rats salivary glands
began initially to filter and to excrete a part of alcaloids received with food,
which has allowed rodents to eat poisonous plants, avoiding a competition to
other herbivores. Then additional function gradually turned to basic one, and
salivary glands had turned to stores of poison.
The diet of these rodents includes a significant part of poisonous plants of
various families, usually spurges and sumach. When poison-toothed giant rat
chews poisonous plants, poison from their sap is intensively absorbing by mucous
membranes of mouth and is immediately filtered in salivary glands. This species
has very expressed immunity to plant poisons, and also to poisons of snakes
and numerous invertebrates. Due to such diet poison-toothed giant rat avoids
food competition to other species of animals.
Voice of this rodent represents silent sounds similar to pig grunting and puffing.
Alarm call of poison-toothed giant rats is shrill whistle heard from very long
distance. Using it, these rodents warn each other of occurrence of predator.
Protecting itself from the enemy, poison-toothed giant rat displays to it contrast
colouring of head and utters loud squeal. If the predator does not stop an attack,
poison-toothed giant rat puts to it deep snaps and injects poison in wounds
at these moments.
These animals live in pairs in large holes arranged under roots of big trees.
The pair at poison-toothed giant rats is kept during very long time – partners
may raise posterity in common within several years in succession. Within one
year this rat gives birth to two litters numbering 2 – 4 cubs. They are born
blind, but covered with rare wool. Female looks after them within one month
while cubs will develop enough to search for food independently and to follow
parents.
The first months of life poison-toothed giant rat lacks characteristic poisonous
bite, because it has weak immunity to poisons, and eats nonpoisonous parts of
plants. At this time only protection of this animal is its own care. Young animal
gradually includes poisonous plants in diet, and later becomes capable to accumulate
poison in salivary glands.
North
Island Tree Rat (Neouromys carpophagus)
Order: Rodents (Rodentia)
Family: Mice and rats (Muridae)
Habitat: Sub-tropical woods and forests of the northern part of North Island,
New Zealand.
The brown rat (Rattus norvegicus) has been a constant commensal and pest of
mankind, and has spread wherever he went. Stowaway rats that came to New Zealand
soon became very successful, and have left descendants in the Neocene.
North Island Tree Rats are very large rats, living mainly in the branches of
forest trees, and feeding on fruit and seeds. Physical shape is generally more
elongate and less stout than its ancestor, though it is overall heavier. They
reach 35 centimetres long, not counting the tail, and can weigh up to 1 kilogram,
the tail is longer than the body, and used to balance itself when climbing.
Face is fairly long with a “roman nose” shape and large incisors, cheeks are
large and can be used to store food. Colour of fur is light greyish brown with
darker markings around the face, feet are pink, tail is naked and dark grey.
Diet consists of the fruit, nuts and large seeds of forest trees, individuals
make nests out of leaves in the branches where they store food they have gathered,
and also where females raise their young. This animal is nocturnal and rather
shy; it can flee through the branches into the treetops very quickly. Animals
communicate when they meet with various squeaks, some of which are ultrasonic,
individuals are mostly solitary but tolerant of other individuals, and any one
piece of territory may have up to 20 individuals ranging within it, of which
certain bigger individuals will be dominant.
Breeding occurs from September to December, sexual maturity is reached at 9
weeks. Males do not compete to breed, and any female may bear young sired by
a number of males, gestation is from 25 to 30 days, and litters consist of up
to six babies, which remain with their mother for up to three months. Females
groom their young to clean them and secure social bonds. Lifespan is up to 5
years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Aotearoan
Ratmarmot (Marmotarattus australis)
Order: Rodents (Rodentia)
Family: Mice and rats (Muridae)
Habitat: Open country, meadows, grassland, scrub, grassy mountain slopes, North
and South Islands of New Zealand.
The adaptive radiation of Rodents in New Zealand was seeded by stowaways on
ships, and has resulted in numerous novel and specialized forms, including those
converging upon kinds from other continents, such as marmots.
Ratmarmot is a common inhabitant of open country in New Zealand, pretty much
wherever there is grass, and ground which is not swampy. These rodents reach
about 30 centimeters in body length and about a kilogram in weight, constitution
is more muscular than its ancestor, with strong limbs bearing large digging
claws. The tail is relatively short, furred and thick, serving as a store of
fat. Fur colour is a overall dark brown, but northern populations are lighter
and more greyish, belly is dirty white, feet and nose are blackish.
Diet consists almost entirely of grass and herbage found in open country, as
well as seeds occasionally, individuals will graze by day and into twilight,
rushing into the safety of communal burrows at the slightest sign of danger.
This animal is highly social, building large underground warrens in which may
live many tens of individuals. Warrens consist of a network of tunnels; living
chambers, and nursing chambers, dung is thrown out certain exits that serve
as latrine piles. Warrens have many entries and exits, some hidden, and sentries
stand atop mound-like entrances, on their hind legs, calling with loud strident
chirps in case of danger.
In the group, dominant large females breed the most often, suppressing the hormones
of subordinate females by harassing them. When a group becomes too large, fighting
occurs and generally subordinate individuals are driven out to start new colonies.
Individuals groom and play fight to strengthen social bonds among comrades and
sexual partners, and females affectionately clean their young.
Breeding occurs from April to July, the male makes a recurring, chirping mating
call around an entrance to entice a female, showing his vigour by being exposed.
Mating occurs underground, fighting and grooming are an overture to mating.
Extra mating is prevented by a “copulatory plug” of semen. Gestation is up to
30 days, litters of babies consist of between 6 and 9 individuals. Babies are
nursed by the mother underground for six weeks, after which young start to venture
out, and soon begin adult feeding behaviour. Sexual maturity is reached at 12
weeks, and lifespan is up to 5 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
False-guatusa
(Dasyproctomys insularis)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: Galapagos islands, forests of the main large island.
Several species of mammal became invasive with the colonisation of the Galapagos
Islands, thankfully some like goats and dogs, were exterminated, but others,
like the Brown Rat (Rattus norvegicus) managed to avoid human interference and
prosper, leaving diverse descendants in the Neocene.
This large rodent is an ecological analog to the Holocene Agouti (Dasyprocta)
of South America, this is where it gains part of its name “guatusa” Ecuadorian
word for Agouti. They reach about 40 cm long and 4.7 kilograms on average, being
moderately large for a rodent. Coloration is a reddish brown, with numerous
pale spots, face and extremities bear darker fur than the rest of the body,
nose and soles of feet are bare and pink, ears are large and prominent, hairless.
It has a short, muscular body with a large rump; forelegs are shorter than hind-legs.
Limbs are rather long, and this animal is able to run fairly fast through the
undergrowth to escape predators. Head is blocky and muscular, with large orange
incisors, this is connected to its diet, hard seeds which have fallen to the
forest floor, other forage includes soft understory growth, fruit, occasionally
insects and eggs of nesting birds.
This animal is most active at twilight and early morning, but may also forage
by day. Nest of this animal consists of a large scrape in the ground, under
a bush or small tree, which is lined with dry leaves. Pseudo-guatusa will bury
caches of gathered seeds in certain spots within its territory, but it rarely
forgets where they are. Animal is solitary, but will tolerate others of its
kind, many individuals may share a common territory. Communications consists
of high chirping squeals and clicking of incisors, call when distressed or alarmed
is a high long squeal.
Breeding occurs year round, female may bear up to 3 litters a year. Males seek
out females by scent, and may confront each other in order to compete for mating
rights by posturing and attempting to bite one another. Litters consist of up
to 5 pups, which are born naked and blind but mature very quickly. After two
months young are fully furred and able to follow their parents and consume adult
food, sexual maturity is reached at one and a half years. Lifespan may be up
to 5 years but usually much shorter.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Common
anguja (Anguja robustus)
Order: Rodents (Rodentia)
Family: Mice and rats (Muridae)
Habitat: coastal forests of southeastern and northeastern South America.
Even with the impacts caused by human interference in the nature during the
Holocene, some species are capable to survive and continue with their evolution.
In the Neocene, a new genus of rodent appeared, the Genus Anguja, this name
means rat in the Guarani language. The members of this Genus are all predatory
rodents, acting like the carnivorous marsupial mice of Australia. The type species
of this Genus is the common anguja, a species that lives in the coastal forests
of southeastern and northeastern South America.
The male common anguja has 14 cm length, while the female has 12 cm. They are
greyish brown, with pale grey undersides and feet. The tail is long, bushy,
and flattened from base to tip. The ears are larger and rounded. Common anguja
are good climbers and have sharp claws. They use their long tails for balance
while climbing and jumping.
This rodent is a good hunter, specialized on hunting at night. Their diet consists
mostly of insects, centipedes, arachnids and other vertebrates. They will attack
nests of birds, eating eggs, chicks and sometimes the parents that are taking
care of the young. The common anguja is resistant to the poison of some species
of scorpions and spiders.
This rodent inhabits tunnels and pathways created by other rodents. Given their
good ability to climb in trees, they can also make dens inside hollowed trees,
preferably those that were once used by birds as nests, which eggs they could
have eaten.
Common anguja has about two or three litters a year consisting of two to seven
young. They have a gestation of 32–47 days, and the young are born in either
late fall or early winter in dens dug by the parents. Living in pairs, the male
gathers and provides food for the female while she is pregnant and taking care
of her young. The common anguja is an altricial species; young rodents are naked
with closed eyes when they are born, and weigh about three grams. Both sexes
reach their sexual maturity at three months. Their lifespan is of 6 years, but
many perish with few weeks and months.
Other members of the genus Anguja:
Striped anguja
(Anguja striatus)
This species inhabits the savannas of Northeastern and Central South America.
The male has 16 cm of length and the female 13 cm, thus being the largest member
of the genus. Their fur is yellowish brown with characteristic dark strips in
the back. They attack the communal nests of azure
parakeets, sometimes doing their dens there. The striped anguja is the favorite
prey of grey-faced pygmy owl
during nesting season.
Auburn anguja (Anguja
rufus)
This species inhabits the forests of Southern South America. The male has 13
of length and the female 11 cm, thus being the smallest member of the genus.
Their fur is reddish brown with pale grey underbelly. They prey mostly on invertebrates,
sometimes eating small frogs and lizards.
This mammal species was discovered by João Vitor Coutinho, Brazil.
Marinating
Coney Rat (Marinadomys herbosiros)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: North America, temperate zone, meadows, fields and grassland.
North America played host to its own endemic families of rodents during the
Holocene, which were later joined by introduced rats and mice of the family
Muridae, due to the actions of man. These animals have left descendants in the
Neocene.
Marinating Coney Rat is descended from the Brown Rat (Rattus norvegicus), it
looks outwardly a little similar to its ancestor, apart from lack of tail and
more rounded face, it reaches a length of 30 centimetres and a weight of 550
grams. Coloration is somewhat more distinct, with a white belly and dark fawn-colored
body, face is darker and extremities are naked and pink.
Its diet is more specialized than that of its ancestor, feeding mostly on herbs,
weeds, grass stems, the leaves of low shrubs, and grass seeds. It usually gathers
them in portions and stores them in its nest, which is a relatively deep hole,
usually made in lee of a rock or tree stump. Animal remains active through the
winter from making large stores of food. What it does with its stored food is
extraordinary, it will cache fibrous and tough grasses and herbs alongside large
amounts of weeds which have acidic, sour juices and sap. The stored food that
it does not immediately eat slowly starts to decompose, and the juices from
the weeds proceed to break down some of the structural chemicals within the
stored food. To protect it from the air oxygen, this rodent buries the stored
feed in the ground. It is from this habit that it gets the name “Marinating
Coney Rat”.
They are most active in early morning and late afternoon, individuals have distinct
territories, and will defend them against interlopers and competitors. Breeding
occurs in spring and early summer, from March to July. Females are sexually
mature at 3 months old, gestation is one month. Litter size is usually six babies,
which mature very quickly; young are reared in a burrow lined with plucked underfur
and dry grass. This rodent is generally solitary outside the breeding season,
competitors for food are driven out, animal is most active during early morning
and early evening. Lifespan is up to 5 years, they are commonly preyed upon
by raptorial birds, canids and felids.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Spiny
Jungle Rat (Echinorattus australis)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: mountainous rainforests of Northern Meganesia.
Introduced rats and mice that came to live in Australia managed to compete successfully
with native species, but also to find new niches as the Neocene progressed.
Tropical Rainforests in the north of Meganesia play host to a large range of
fruit and nut bearing trees, which are mainly fed upon by parrots and cockatoos,
but these birds only rarely descend to eat fallen fruits and seeds in such a
dense understory. This is the niche taken by the Spiny Jungle Rat, a creature
analogous to the agoutis of South American jungles. It feeds by gnawing open
the hard seeds of rainforest trees, as well as eating the flesh and seeds of
fallen fruit.
Animal reaches up to 45 centimetres in length, and has only a stubby, hairless
tail, weight is up to 4 kilograms. Outward appearance recalls both its ancestor,
the Brown Rat (Rattus norvegicus) and a South American Agouti, legs are fairly
long, hands are grasping, hind legs are longer than forelegs, and rump is large.
The head is blocky and profusely muscled, with large incisors of dark orange
colour. It is supplied with defence by the presence of sharp black spines, up
to 8 centimetres long, which are distributed amongst the fur, spines are denser
on the rump and shoulders. Coloration is chocolate brown on the upper parts,
with pale grey cross stripes, belly is yellowish white.
Spiny Jungle Rat is diurnal, solitary but very shy, and will flee when disturbed;
individuals tolerate each other outside the breeding season so long as food
is not scarce. Breeding occurs throughout the year, with the most young being
born in the dry season. Young are reared in a sheltered scrape in the forest
floor, obscured by dense vegetation; male takes no part in rearing young. Female
is sexually mature at one year, gestation is around 40 days. Usually 4 young
are born; these can occasionally be two sets of twins. Young reach full size
within 6 months. Lifespan is up to 13 years due to defensive spines, only relatively
persistent or smart predators, such as raptorial birds and predatory marsupials,
may succeed in preying upon it.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Rapa
Nui rat (Geomurinus brachyurus)
Order: Rodents (Rodentia)
Family: Mice and rats (Muridae)
Habitat: Easter Island.
While humans purposefully introduced some species in new habitats, others were
inadvertently introduced. The rats (Rattus) followed the humans to several places,
benefiting from their presence. When they were extinct, some of these rodents
were able to survive and continue their evolution. In Easter Island, the populations
of rodents that survived evolved into an endemic species, the Rapa Nui rat.
The Rapa Nui rat has 13 cm of body length, with a short tail of 5 cm. Their
physical appearance reminds that of a Cricetidae hamster, with them having a
fossorial lifestyle. They have strong forelimbs that are used to excavate the
ground to make tunnels to be used as their dens. It has reddish-brown fur with
a white underside and three black stripes on the back.
This species feeds mostly on fruits, roots and seeds. Insects and eggs are eaten
very occasionally. They spend the day on their dens, only leaving to forage
at night.
The Rapa Nui rats are very territorial and intolerant of each other, with attacks
against each other being commonplace. Exceptions do occur, usually when a female
and male meet when the female is in heat, but even so, the female may attack
the male after mating. Even siblings, once mature, may attack one another. Same-sex
groups of siblings can stay with each other until they are about eight weeks
old, at which point they will become territorial and fight with one another,
sometimes to the death. Infanticide is not uncommon among females, usually due
to stress from the environment. They also eat their dead young.
These rodents mark their burrows with secretions from special scent glands on
their hips. The males in particular lick their bodies near the glands, creating
damp spots on the fur, then drag their sides along objects to mark their territory.
Females also use bodily secretions and feces. They can have two to four litters
per year. The Rapa Nui rat has a 15 days long gestation, giving birth to a litter
of 1 to 12 that wean after three weeks of age.
It communicates by squeaking or with ultrasound. The females are sexually mature
with eight weeks, while the males only become mature with six months. Their
lifespan is 3 years, but many are killed by predators, such as the owl-harrier.
This mammal species was discovered by João Vitor Coutinho, Brazil.
False
Hamster (Cricetimimus novazealandiae)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: Open country, scrub and light forest, vegetated mountainsides, North
and South Islands of New Zealand.
The house mouse (Mus musculus) was a ubiquitous pest of mankind in the Holocene,
and it made its way to wherever man came to settle. Plagues of mice would often
occur on grain farms, and infestations in human dwellings were common.
In the Neocene, apart from rather typical kinds of mouse, New Zealand plays
host to various Murids with a somewhat more specialized lifestyle, like the
False Hamster. This animal is far more common on the cooler, drier South Island,
where open country is more prevalent, North Island populations are smaller in
size and more sparsely distributed. Adults reach 15 centimetres long and weigh
up to 50 grams, having only short, stubby tails, physical shape is rounder and
more robust than its ancestor, with large cheek pouches for storing food. Coloration
of fur is a dark reddish brown with a yellowish belly, face is marked with a
whitish “mask” , feet, nose and naked tail are pinkish. Its diet consists of
various grains and seeds as well as the stems of grasses, and insects occasionally.
These animals use their cheek pouches to store large amounts of food underground.
Individuals are solitary and indeed anti-social outside the mating season, and
live in small, deep set burrows, in which they sleep, store food, and raise
their young. This rodent is most active in the twilight and early evening. Males
and females make glandular secretions in order to mark and maintain territorial
borders, and a latrine is left near the burrow for this purpose also. Call is
a rather typical squeak, a high chirp, hissing when alarmed or fighting.
Sexual maturity is reached at 8 weeks, and females come into oestrus multiple
times per month, gestation is 25 days. Litters consist of up to 10 babies, the
mother will nurse them for a short period of time, and if she feels threatened,
may kill and eat her pups. Lifespan is up to 3 years.
This species of mammals was discovered by Timothy Donald Morris, Adelaide, Australia.
Causeway
Mouse (Commutomys semistriatus)
Order: Rodents (Rodentia)
Family: Mice and Rats (Muridae)
Habitat: tropical savannahs and semideserts of Central and southern Africa.

Picture by Wovoka

Animals commonly evolve habits and specializations in order
to avoid predation, rodents seem to have excelled at this, from kinds
with spines to poisonous forms, to
ones that run quickly. One such example of predatory avoidance in habit is the
Causeway Mouse.
This rodent is generally unspectacular in appearance, small, pale brown with
indistinct dark longitudinal stripes, in shape generally very much like a typical
mouse, apart from legs and feet which are longer and more slender. Head and
body reach up to 10 centimeters long, with the tail reaching a further 10 to
12 centimeters, weight is up to 30 grams. Descended from Striped Grass Mouse
(Lemniscomys striatus), it has passed to a diet of insects, fallen berries,
and the softer parts of herbaceous plants.
The habits of this animal are extraordinary. It clears paths in the grass which
it runs along to access areas where it usually feeds, this is not so unusual,
some small mammals of human era did this. But what is unusual is that it protects
itself by enclosing these pathways in a dense latticework of grass stems which
act to form tunnels out of these paths, from this habit it gains the name “Causeway
Mouse”. When these tunnels are disturbed or damaged, the mouse proceeds to mend
them, and they are being maintained constantly, with new ones being made when
food sources fail. These tunnels serve to conceal it from nearby predators,
a nest which is a globular bundle of woven grass stems, sits at the centre of
its territory, most often sheltered beneath a bush or in lee of a rock or fallen
tree.
It is diurnal and solitary, being most active in the morning. Breeding occurs
year round, a male will venture out of his usual causeways in search of a female’s
nest, with which he may visit to breed multiple times a year. Oestrus cycle
of the female is about six days, and oestrus lasts a day and a half, gestation
is twenty days. Young are born blind and hairless, maturing and weaning at 30
days. Lifespan is up to 2 years. Predators include small cats and various birds
of prey, some kinds of predator may be smart enough to make a hole in the tunnel,
and wait for this mouse to pass, in order to catch it.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Hawaiian
mole mouse (Xenospalax subterraneus)
Order: Rodents (Rodentia)
Family: Pacific mole mice (Xenospalacidae)
Habitat: Hawaii (Big Island), ground in valleys and woods.
Before human occurrence at Hawaii mammals were absent at islands and only insects
and their larvae could occupy an ecological niche of underground digging animals.
But historical colonization epoch the fauna of islands had replenished with
various animals, alien to indigenious fauna on fault of people. At Hawaii various
species of ground mammal, which began to develop ecological niches inaccessible
to birds, had appeared. Moles and other digging animals were not introduced
to Hawaii, but their place uniquitous rodents had occupied: they are descendants
of mice avoiding a competition to descendants of two species of rats, also introduced
to islands by people.
The ground in woods of Hawaiian Islands is penetrated with set of underground
burrows in which their builders - completely blind rat-sized animals – run.
By body shape they are similar to mole rats (Spalax) living in Eurasia, but
they are descendants of house mouse. Therefore they are named Hawaiian mole
mice. They differ from true mole rats in features of behavior: if true mole
rats frequently left on the ground surface for feeding, their Hawaiian “doubler”
does it extremely seldom, eating only roots and tubers of plants.
Hawaiian mole mice are similar to continental species by features of anatomy
developed at both groups independently. At them there are the same cylindrical
body, short paws and large head. Main digging adaptations of Hawaiian mole mouse
are its head and strong incisors. Skull of this animal is wide and short, and
incisors of top jaw are very big. Lips of animal densely close mouth from the
ground by special outgrowths, not impeding incisors. Occipital muscles are very
strong, for their attachment on back edge of skull of the animal the special
crest stretches. Muscles lowering head are also strongly advanced. During the
ground digging this small mammal lifts head making effort and ramming a vault
of formed hole, and then lowers it, ploughing by incisors deep furrow in the
ground before itself. Animal rakes away the loosened ground by forepaws under
stomach and pushes it out from hole, or simply stamps by paws.
This animal is completely blind, and its eyes are reduced even in greater degree,
than at moles – eyeballs are abscent and the optic nerve had reduced. The top
of head and shoulders of Hawaiian mole mouse are covered with thick cornificate
skin – animal forms a vault of hole by them. Corneous armour on head closes
the place where ancestors of this animal had eyes, and also covers nose from
above. It is additionally strengthened with bones of skull of animal grown together
in continuous osseous vault.
Because paws do not accept direct participation in digging, they are rather
short. Animal only rakes away the friable ground back by them. Tail of Hawaiian
mole mouse is also very short and hairless. Animal is covered with velvety fur
of black color. On muzzle of Hawaiian mole mice long whiskers, with which help
animals are guided in space of underground courses, grow.
Hawaiian mole mice build extended systems of burrows in wood, and arrange the
special drainage tunnels through which the water leaking to burrows because
of daily rains is removed from them. As animals are vegetarians, the competition
between them is not as sharp, as, for example, at moles. Hawaiian mole mice
eat roots and tubers of plants, and even parts of such species which are not
eaten by other animals.
Hawaiian mole mice breed practically the year round. For one year the female
can draw up to five hatches numbering 6 – 8 cubs. They grow quickly, and at
monthly age already begin independent life. At three-monthly age they already
can give birth to posterity. But, despite of such high rate of breeding, the
significant amount of young growth perishes: when the colony appears overpopulated,
the part of its inhabitants (mainly young animals) feels stress. It occurs because
of too big frequency of meetings between animals, and less strong animals aspire
to leave earlier safe holes. At this time animals frequently settle not in systems
of holes, but on the ground surface. It happens that such animals movable by
aspiration to avoid of stress appear on ground surface in day time, run in wood,
not trying to hide, and at times even try to force rivers. The significant part
of these of “victims of stress” perishes, but some ones succeed to leave far
enough and to found a new settlement.
Under the ground one of main dangers – the descendant of intruder like them,
the large digging blind snake
catches Hawaiian mole mice. And on the ground surface local
predatory birds, and even descendants of rats catch them.
At the next islands of the Hawaiian archipelago there are close species of these
rodents:
Mole
mouse of Lanai Island (Xenospalax lanaiensis) is the small species
(equal to large mouse in size), distinguished by spotty colouring: on light
grey background there are dark spots. Separate individuals of this species are
almost completely black. This species has similar biology, but spends more time
on ground surface. At night it leaves to feed among bushes.
Mole
mouse of Maui Island, or half-naked mole mouse (Xenospalax
mauiensis) differs from other species in strange appearance: the fur
of yellowish-grey color had remained at this species only on the bottom part
of body and on shoulders. Head, back and sides of this rodent are hairless,
covered with rough grayish skin. This rodent lives in warm rainforests, and
is able to swim well. The young growth can even settle during floodings.
Giant
mole mouse (Xenospalax giganteus) lives at small Kahulawi Island near
southwest coast of Maui Island. It is the largest species of genus: the length
of body reaches 25 cm. It differs in greater, than at other species, propensity
to leave on ground surface. Its wool is colored rusty-brown color, and corneous
“helmet” on head is rather small.
Cataphractherium (“armored beast”) (Cataphractherium protectus)
Order: Rodents (Rodentia)
Family: Porcupines (Hystricidae)
Habitat: forests of Zinj Land, the broken sites of forest stand with plentiful
grassy vegetation.
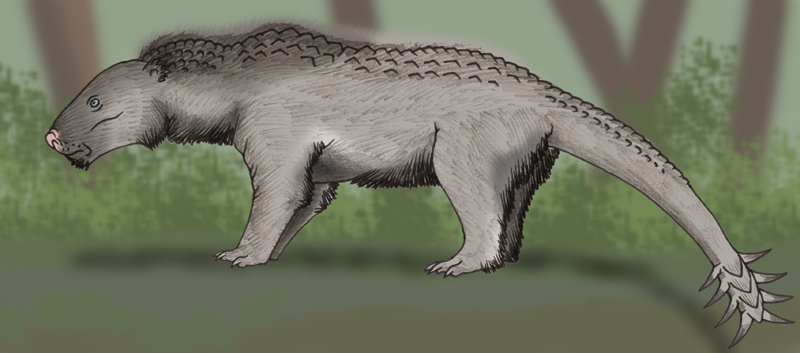
Picture by Tim Morris
In Neocene rodents had an opportunity to pass to large dimensional
class. Earlier this process proceeded on the Earth in limited scale – really
large species
appeared in Neogene of South America, and in other continents this group
was represented by rather small animals. Extinction of large species of animals
in Holocene has enabled rodents to occupy new ecological niches and to become
larger species. The increase of size took place independently in different
groups of rodents. Huge mighty
grasscuttes, kangoohoppers and giant
kangaroo jerboas live in Old World of Neocene epoch. And at the East
African subcontinent, in Zinj Land, the unusual representative of Old World
porcupines, cataphractherium
lives. It is a large porcupine of massive constitution – weight of adult
animal reaches 200 kg, body length is about 150 cm not including tail. This
animal
has convergent similarity to large ground porcupines of
North America and stegoechidna from
Meganesia – it moves on four legs, sometimes rears on hind legs and actively
uses forepaws for food getting. This is a plantigrade animal with large head
and long tail.
The anatomy of skin of cataphractherium is unusual: it is an animal chained
in bone-corneous “armour” similarly to extinct ground sloths of late Cenozoic
(hence the name: “cataphractus” means “armored” in Latin), but having means
of active defense. Needles, characteristic for true porcupines, have lost
function of thrust weapon at cataphractherium. On back and sides of this
animal needles
are truncated and became very thick. They have lost mobility and have turned
to corneous plates of wide pyramidal shape with the dulled apex. On neck
they are joined to cross lines, and on trunk form something like “cobblestone
road”
with chaotic arrangement of plates. Inside these plates located along a backbone
the centres of ossification are formed, and at old animals the corneous tissue
of plates is gradually replaced by firm bone. These plates are mostly immersed
in thickness of skin and only tops jut outside. They are hidden with rich
wool of ash-grey color, darker on head and shoulders.
Cataphractherium is a sluggish herbivorous animal to which it is vital to
be able to render worthy repulse to predators. Tail of this animal has turned
to truly dangerous weapon. It is strong and muscled, because at food getting
heavy cataphractherium supports on it. The spikes, inherited from ancestral
porcupines, had changed and had turned to strong corneoud “daggers” on the
tail of this animal. Terminal vertebrae of tail are fused together, having
formed a kind of “mace”, as at prehistoric ankylosaurs. Lateral shoots of
“mace”
vertebrae are enlarged, and “mace” has flattened shape and rounded outlines.
The bottom surface of “mace” is covered with small prickles, which rest against
the ground when cataphractherium reaches to branches of low trees, having
reared on hind legs. “Dagger” spikes grow on edges of this “mace”, and on
its top
part there are some corneous plates. Defensive tactics of cataphractherium
is very simple – it meets a predator, standing on four paws and swinging
tail. If the predator attacks, animal makes impacts to its legs and stomach
by tail.
Easily turning on hind legs, the animal constantly turns sideways to predator
and keeps up its movements.
Beyond the danger this animal behaves very peacefully. Males may display
aggression to each other only at the presence of the female ready to pairing.
Thus they
try to frighten the contender, striking impacts by tail on grass and thin-stalked
bushes and making a lot of noise. If the contender does not recede, males
combat, having risen on hind legs and “boxing” by forepaws. As cataphractheriums
live
in tropical climate, the courtship season is not expressed precisely, though
the majority of females is ready to pairing in rainseason.
Cataphractheriums are peaceful foliage browsing herbivores. They eat greenery
of undersized trees, bushes and large grassy plants. Powerful incisors allow
them to bite off young branches of bushes and fibrous leafstalks of grassy
plants. With the help of long claws on forepaws cataphractheriums dig the
ground in searches of tubers and rhizomes of plants. It is a solitary species
of animals:
only the female and its posterity keep together.
The female gives rise to two cubs once a year. The young growth is not protected
by plates – they start to grow only at the age of two months. Cubs have soft
wool and spotty colouring – on dark background there are randomly scattered
black spots. As corneous plates grow, spots disappear, being kept for longer
time on shoulders and head. Young animals quickly learn to eat food of adult
animals, but are fed with milk up to five-monthly age, and further require
only protection and the help in getting of vegetative forage. Usually at
this time female is already coupled again, and the young animal leaves mother
shortly
before birth the next cub at her. The young animal reaches the size of adult
individual at the age of three years and becomes sexually mature the same time.
This species of mammals was discovered by Simon, the forum member.
Otter
dormouse (Glirolutra hamiota)
Order: Rodents (Rodentia)
Family: Dormice (Gliridae)
Habitat: southeast of Europe, valleys of rivers running into Fourseas.

Picture by Alexander Smyslov
The last congelation at the boundary of Holocene and Neocene
in had radically changed the geography of Eurasia. The glacier had moved huge
masses of the ground as if huge “bulldozer” and has changed the direction of
current of European rivers. Only the lower reaches of Dnepr, Don and Volga rivers
had remained approximately in the same places, as in human epoch. But their
upper courses are already absolutely different, rather than in human epoch.
Number of inhabitants of European fresh waters had been reduced in great degree,
and many fish-eating animals had died out. In particular, European otters were
among them. Earlier their number was undermined by people, and it was an additional
factor caused their extinction.
After glacier thawing in early Neocene, when fresh waters of Europe had returned
to the territory occupied earlier and had formed new river channels. In rivers
freshwater fishes and crustaceans had settled again, but otters did not hunt
them any more – they had died out irrevocably. However, such favourable ecological
niche has not remained empty - rodents had undertaken a role of fish-eating
river predator. It is remarkable, that in South America about the same situation
had taken place in parallel and independently, but the descendant of completely
another animal became aquatic predator there.
And in Europe the role of Neocene “otter” is played by the descendant of fat
dormouse (Glis glis), by otter dormouse. It is rather large rodent: it is approximately
equal to marten in size. Top side of body of this animal is gray-brown; throat,
breast and stomach are milky-white. Fur is very dense and rich; animal spends
a lot of time looking after it and greases it regularly with fatty secretions
of special glands. Head of otter dormouse is pointed, ears are small, and muzzle
is lengthened. On muzzle there are very much advanced rigid and long vibrissae,
helping to be guided even in muddy water.
The basic body of underwater movement represents hind legs with long toes between
which membranes are advanced. In addition this rodent swims with the help of
wide and flattened tail similar a little to beaver’s tail. Forepaws are shorter,
rather than hind ones, and at movement they are pressed against chest. Fingers
lack of membranes, but the claws are advanced, and finger pads are very sensitive.
The basic food of this rodent includes fish catching by otter dormouse in thickness
of water. Occasionally it eats large crustaceans – crayfishes and large scuds.
This rodent kills small prey (small crustaceans, tadpoles, fish fry and insects)
by bite and hides in cheek pouches. Having emerged on surface of water, otter
dormouse eats the saved up prey. Otter dormouse eats large fish on land, holding
food in forepaws.
Otter dormice live in clans, forming small settlements on riverbanks. Such colony
totals up to ten adult animals, and also some individuals of young growth of
the present year. “Lair” is arranged in holes in riverbanks. Holes are located
no farther than one meter from each other; the single rodent and growing up
posterity till some time lives in each one. Occasionally among otter dormice
the convinced singles appear. They are less conservative in a choice of refuge
and sometimes settle even in hollows of trees or in nests of large birds, arranging
“ground floor” under nest (due to tenacious claws this rodent is able to climb
up trees). But life alone for this rodent is rather compelled circumstances,
than a rule. But the clan of these animals represents simply some individuals
living nearby from each other and not connected with relationships in the majority.
At them the hierarchy constructed by a principle of strict matriarchy is established:
females are larger than males and settle in the center of colony.
During the hunting each rodent operates separately and does not miss an opportunity
to steal prey at weaker individual. Therefore the young growth, leaving mother,
stays in colony rather seldom, especially if the place is not too rich in food.
The only thing uniting these rodents is the danger. Even large wide-headed catfishes
are afraid to attack group of these rodents, preying only young or single animals.
Otter dormice have voice and can utter some different signals based on whistle.
Loud whistle is, perhaps, a sound most important for them – it’s a signal of
threat from air: birds of prey are usual enemies of these rodents. Rolling chirring
is a signal of underwater alarm. At occurrence of a stranger from other clan,
or a single individual, old residents of colony meet it with sibiliant whistle,
accompanying it with threating poses. If the shortage of food takes place, otter
dormice may be aggressive to strangers.
Despite of descending from dormice, these rodents keep activity in winter. They
punch thin ice on river surface with claws or gnaw it through. But usually in
habitats of these rodents river does not become covered by ice – otter dormice
do not settle far to the north.
Two times per one year the female gives rise up to 5 – 6 cubs. They come into
the world blind, but covered with wool. The first litter is born when winter
colds have not passed yet, and the second litter appears in the middle of summer.
Shortly before it female banishes the previous posterity. At one-year-old age
young animals begin able to bring posterity.
This species of mammals was discovered by Simon, the forum member.
Mountain
dormouse, chinchilla-dormouse (Raptoglis chinchilloides)
Order: Rodents (Rodentia)
Family: Dormice (Gliridae)
Habitat: mountains of Caucasian peninsula, Middle East.

Picture by Alexander Smyslov
In early Neocene rodents got an opportunity to prove once
again their evolutionary success. They evolved quickly, occupying all habitats
suitable for them, and already in early Neocene their specific variety was comparable
to the level of Pliocene and an early Pleistocene, before the epoch of anthropogenous
pressure. In Neocene rodents are widespread everywhere on Earth, except for
Antarctica – from equatorial forests to cold northern meadows and tops of mountains.
In Eurasia in Neocene there were some species of dormice family which use food
of animal origin much more often than their ancestors. These are the
sable dormouse (Raptoglis zibellinus) from Three-River-Land steppes and
the otter-dormouse (Glirolutra hamiota)
from rivers of eastern part of Europe. One more species closely related to them
lives to the south from Fourseas, in mountains of Caucasian peninsula and mountains
of the Middle East – it is the mountain dormouse called also the chinchilla-dormouse
for its thick warm fur. This representative of northern fauna prospers in the
south exactly thanks to dwelling in cold highland climate.
The mountain dormouse is the large rodent reaching weight of about 3 kilograms
and the size of a small cat. Due to tail covered with long wool the animal seems
even larger. The head of this rodent is rounded, has shortened muzzle and small
ears covered with wool from outside. Mountain dormouse has large eyes and good
sight.
This species mastered successfully life in highlands due to thick fur which
helps to keep heat even in hard frost or at the great elevations. Summer fur
of this animal is reddish-brown, velvety and rather short. There always is a
white spot on breast and throat, head top is darker. Winter fur differs in greater
wool length and thickness, and also in lighter coloring.

Picture by Alexander Smyslov
Constitution of this animal is solid, and paws are short (hinder
legs are rather longer than forelegs). Fingers and toes are armed with pointed
claws due to which mountain dormouse is capable to climb on almost steep mountainsides
and among a bush. This animal can make leaps up to 2 – 3 meters long.
Mountain dormouses are almost exceptionally zoophagous, adding to their diet
no more than 10% of vegetable food, though in case of starvation they can temporarily
turn to completely vegetative food. These rodents often eat carrion, searching
for it by smell. Additionally mountain dormouses readily gnaw even bones. For
them the habit to search and to pull down bones to “bonemill”, a special place
hidden from predators, where these animals gnaw their finds, is characteristic.
They almost completely assimilate collagen from the eaten bones, and the remains
are insignificant. Usual prey of mountain dormice includes small rodents, birds
and their eggs, and reptiles and insects in lowlands.
This species of rodents lives singly and rarely in couples. The female is more
aggressive than male. The shelter of this rodent represents the deep hole made
between stones in the way hardening the digging out of the hole by predators.
Like other representatives of family, mountain dormice fall the long hibernation
which can last up to half a year with short breaks. Animals grow fat intensively
to the winter, gaining the weight of about 4,5 kg. Fur becomes longer and denser.
During the winter the animal wakes up for several times to leave the dung, which
is very condensed and dry at this time.
The posterity is born at the end of spring when there is a lot of food. The
litter numbers up to 5 cubs which grow quickly and at the age of about two weeks
already start tasting meat. Sometimes at the end of summer animals can have
the second litter in which only sometimes there are more than two cubs. Life
expectancy doesn’t exceed 5 years.
Translated by Alexander Smyslov
Viverrine
Dormouse (Viverroglis maculatus)
Order: Rodents (Rodentia)
Family: Dormice (Gliridae)
Habitat: Southern Europe, areas bordering Mediterranean hypergaline swamp; Middle-east,
forests, woods and scrub.
Specialized forms of rodent carnivorous in diet can be found in the Neocene,
among them the Predatory Dormice (Raptoglis). Relatives of these, descended
from a common ancestor, are less specialized in diet and just as adaptable,
Viverrine Dormouse. Living in forested areas of southern Europe and the Middle-east,
this rodent somewhat resembles the Sable Dormouse (Raptoglis
zibellinus), but is a bit more elongate in shape, legs are more bent
and claws larger. Animal is an analog of the genets and civets of Holocene,
feeding not only on small vertebrates, but insects, fruit, fungi and large seeds.
It is an opportunist, eating whatever is most available; in part of range bordering
the Mediterranean swamp it may feed profusely on wild olives.
Viverrine Dormouse is a fair sized animal, reaching a weight of 4 kilograms,
animal seems longer due to low build and long torso and tail. Tail is long and
used for balance, legs are short and bent, claws on all four feet are large
and curved. This species climbs trees well and spends a good deal of time there.
This animal is very agile and can leap between trees, or make pouncing jumps
onto prey. The face is pointed and the base of the skull is long, nose is bare
but the face has long whiskers, feet are bare. Ears are relatively large and
bare on the inner surface.
Its fur is fairly short (except on the belly), giving it a sleek appearance
like a cat, colouring is light yellow brown with numerous dark brown spots and
cross stripes, tail is banded with a white tip.
Generally solitary, they are not averse to their own kind and may form couples.
Unlike its relatives, it nests in trees, preferring a large tree hole or broad
fork in the crown of the tree.
As opposed to its relatives, this animal does not hibernate but it may become
less active during the cold of winter.
Breeding occurs in late winter and early spring. Up to 5 young are born and
they develop very quickly, eating solid food within 3 weeks. Sometimes pairs
make a successful second litter in the early summer. Lifespan is up to 5 years,
predators and competitors include carnivores like cats and true civets, as well
as large birds of prey.
This mammal species was discovered by Timothy Donald Morris, Adelaide, Australia.
Giant
kangaroo jerboa (Macrodipodes wallabiiformis)
Order: Rodents (Rodentia)
Family: Jerboas (Dipodidae)
Habitat: steppes of South-Eastern Europe, along the Fourseas coast to Middle
Asia.

Picture by Alexey Tatarinov
Steppe ecosystems of Eurasia before the beginning of the vigorous
human activity had not conceded to savannas of Africa in biological efficiency.
Various hoofed mammals – aurochses, wild horses and antelopes – had been dominant
species of steppe herbivores. People ploughed steppes, transforming these areas
into primitive agrocenoses, and exterminated steppe animals. The result of human
activity was a degrading of steppe ecosystems by the end of human era. In Neocene
the population of newly appearing ecosystems had been formed, actually, from
zero. In the Euroasian steppes of temperate climatic zone representatives of
new groups of herbivorous mammals dominate – porcippulas,
cursorial boar relatives, gracile harelopes, and
some other forms of herbivores. Among new steppe herbivores of Neocene large
representatives of rodents took the important place. In Neocene rodents represent
one of the most successful orders of mammals.
Jerboas were the characteristic group of rodents of the Old World lived in open
spaces. In ice age at the boundary of Holocene and Neocene, in conditions of
desertification and deforestation, some representatives of jerboa family had
got a wide distribution and had considerably increased in size. This adaptation,
favourable in conditions of cold climate, has allowed one species of these rodents
to occupy an ecological niche of the analogue of small antelopes in steppes
of Eurasia. This animal is giant kangaroo jerboa, a gregarious middle-sized
mammal.
This rodent has kept features of anatomy, characteristic for family representatives,
adjusted for the giant size: the animal weighs up to 15 kg. In behaviour and
a habit of movement giant kangaroo jerboa is similar to small kangaroos like
wallaby. The body length of this animal reaches 60 cm, and tail is about one
meter long. Long strong tail at this rodent serves for support when animal eats,
bringing food to the mouth by forepaws. Giant kangaroo jerboa moves almost exclusively
on rear legs, accelerating momentum up to 70 kms per hour In case of danger
it makes jumps up to 6 meters long. At jumps tail of animal serves as the balance
weight. On the tip of tail the switch of white hair is advanced.
Ears of giant kangaroo jerboa are rather short and rounded. This animal does
not dig holes, and does not run into hibernation in winter – these changes of
behaviour are consequences of increase of body size. In winter this animal keeps
activity, and it does not need overcooling.
Summer wool of giant kangaroo jerboa has sand yellow color with small dark spots
merging to short cross strokes on back. In winter the wool of animal turns lighter
in color and becomes thicker. At this species of rodents wool is weakly attached
to skin. If the predator will seize this animal, wool easily drops out and allows
the animal to escape from its teeth. Moreover in winter “dress” at giant kangaroo
jerboa “ski” grow on feet – it is a border of rigid wool. This adaptation helps
an animal to jump easily on snow, not falling in.
In summer giant kangaroo jerboa is a solitary animal. Each individual has territory,
and its borders are marked by urine and secretions of smell glands. The centre
of territory is a hole or any other ready shelter. Giant kangaroo jerboa is
not able to dig a hole itself, therefore it uses ready shelter – usually holes
dug out by goliath mole rat, huge local rodent.
Usually the territory with a hole is occupied by female. Males are not so choosy
in the choice of shelter: rich bushes as shelter are frequently enough for them.
This species of rodents has rather long pregnancy time, and the female brings
only one litter in one year, in which there are about 5 cubs. Newborn cubs are
helpless and female hides them in hole, closing an entrance with a bunch of
prickly grass or bush pulled out from the ground. Cubs are born in the beginning
of summer, and within the first month of life they do not leave shelter. The
female watches the vicinities of shelter, and if necessary it is ready to distract
a predator in order to withdraw it far away from shelter. Cubs behave very cautiously,
even having learned to move. While the female is near to posterity, cubs can
leave a hole and play, but at a first sign of danger they hide in. The posterity
leaves a hole for ever at the age of about one and a half months. At this time
colouring of cubs is cross-striped, that helps them to mask in grass better.
It is the only protection of young animal. Usually young animals wait for returning
of the female, having hidden in grass. In case of need cubs can escape from
the enemy, quickly jumping and sharply changing direction of movement. When
danger passes, they come back to a former place, waiting for the female returning.
At the same time young animals begin to eat plants. At the end of summer the
litter is fed in territory of the female, gaining weight. Young animals become
independent to an autumn, having reached the weight of 8 – 9 kg. This is a very
important time in their life: the young animal, not having accumulated a sufficient
stock of hypodermic fat, easily becomes a victim of frost. In the first winter
about a half of the young growth born in summer perishes because of various
reasons. But the next year in autumn females become capable to pairing, and
at the age of two years, after the second wintering in their life, they bring
posterity the first time.
In winter, when it is more difficult to find food and danger of predator attacks
is more probable, giant kangaroo jerboas gather to herds numbering over fifty
animals, and wander on extensive territory. In spring, when snow thaws, the
common territory of herd is broken into individual sites, and animals become
solitary ones again.
Bush-climbing jerboa (Galagops scandens)
Order: Rodents (Rodentia)
Family: Jerboas (Dipodidae)
Habitat: Central Asia, bush thickets.
At the boundary of Holocene and Neocene ecosystems of the Earth had passed
through a short-term stage of impoverishment of specific variety. As a result
of human activity and climatic changes the majority of large species of vertebrates
had died out. However, this process was accompanied by outburst of number and
growth of specific variety of small mammals, mainly of rodents. In course of
evolution these animals adapted to inhabiting in newly formed ecosystems. One
example of this process is a small species of rodents from Central Asia, bush-climbing
jerboa. Approximately 10 million years after end of Holocene congelation, when
climate became humid, the area of steppes and semideserts of Central Asia was
reduced, and they had partly given up the place to hardy bushy vegetation.
The part of animal species lived in these places had died out, but some steppe
species managed to adapt to inhabiting in places of new type of vegetation.
Rodents have successfully mastered these habitats, having formed a significant
amount of new species. Bush-climbing jerboa is one of them; being a descendant
of certain low-specialized species of this family, it has developed a new way
of movement – climbing and jumps among branches.
This small-sized rodent is a little similar to bushbaby prosimian in shape
and a manner of movement. Bush-climbing jerboa reaches length of 25 cm; tail
is up to 50 – 55 cm. Its rear legs are lengthened and have narrow long foot
and short mobile toes. Forepaws of animal are also prehensile. Bush-climbing
jerboa jumps in branches on rear legs, using tail as a balance weight, but
it also can move in “galloping” movements, seizing branch alternately by front
and rear paws. It is capable to climb down along tree trunk turning a tail
downwards, but simply jumps off right on the ground from undersized trees and
bushes (up to 3 meters high). This animal prefers to live in top part of tree
crones where there is an opportunity to make long jumps. At the contact with
branch toes of animal automatically grasp it.
Ears of this rodent are short, rounded and wide. They do not prevent to move
on branches, but help to catch environmental sounds and serve as the good device
of heat radiating. In hot weather blood vessels extend, and animal moistens
external surface of auricles with urine at which evaporation there is better
heat radiation.
Wool of bush-climbing jerboa has yellowish-brown colouring with several longitudinal
strips of brown color; stomach is white. On tip of tail there is a switch of
black color with white top. With its help animal gives signals to relatives
and distracts attention of predators.
This is an omnivore eating seeds and buds of plants, and also small insects.
Having an opportunity, bush-climbing jerboa can ravage bird nests. This rodent
frequently lives near bushgophers, repairs their abandoned shelters and occupies
them. In winter this rodent runs into hibernation. It spends winter in reliable
shelter – usually in deep holes of other species of rodents. Frequently some
animals gather and run into hibernation in common hole. Animals close the entrance
to this hole with plug of dry grass. Feeling rigorous winter, animals may make
some grass plugs.
Within spring and summer female has time to give birth to two litters of three
– five cubs. To give birth to posterity the female uses abandoned bird nest
in which it simply drags more stalks and leaves. Cubs are born naked and blind,
but develop quickly. At the age of two weeks they already study to climb on
branches.
In warmer areas of Eurasia bush-climbing jerboa is substituted by close species:
grass-hopping jerboa (Galagops minusculus). This is dwarf similarity of bush-climbing
jerboa: the adult grass-hopping jerboa is mouse-sized animal. It lives in thickets
of high grasses, eats grains of various degrees of ripeness. In spring this
little mammal eats buds and flowers of plants, insects and their larvae. It
inhabits steppes of Three-Rivers-Land, and is spreaded to the west along northern
coast of Fourseas. Grass-hopping jerboa frequently occupies a part of hole
of digging rodents, peacefully getting on with hole owners.
Giant
wood porcupine (Aepythizon megatherioides)
Order: Rodents (Rodentia)
Family: Wood porcupines (Erethizontidae)
Habitat: woods at plains and mountains of northwest of Northern America (to the
west up to southern areas of Beringia).

Initial picture by Amplion, colorization by Lambert
Initial picture by Amplion |
After extinction of plenty of hoofed mammals species at the
border of Holocene and Neocene other herbivore mammals had occupied become empty
ecological niches having caused fantastical species. There were rodents among
them, huge by variety order including in Holocene basically small and absolutely
tiny species. Certainly in various epochs of Cenozoic rodents more than once
“tried” to come to middle- and large-sized class, and sometimes it happened
most advantageously: the rhinoceros-sized rodent lived in South America and
Pleistocene North-American species of beavers reached the size of bear had appeared
among them. In Holocene largest representative of this group were beaver and
capybara lived in rivers. But in Neocene having occupied various ecological
niches rodents have fairly increased their size. In South America among rodents
forms similar to antelopes and pigs, and also giants similar to ground sloths
of Pliocene and Pleistocene have appeared. At the territory of Mexican plateau
the bear-sized porcupine has
lodged in Neocene. But in wood zone of Northern America this porcupine has even
larger relative.
Giant wood porcupine is very large representative of Neocenic rodents (it weights
up to 1 ton; growth of animal standing on hinder legs is up to 3 meters, general
length is over 5 meters including tail). Back legs of this animal are longer
than front ones; due to it animal can stand on two legs eating plants. Feet
of hinder legs are covered with thick cornificate skin therefore animal equally
easily can walk both on stones heated up by sun and on winter ice. It is a herbivorous
animal; its food includes leaves and branches of various deciduous trees. In
winter giant wood porcupine can eat even poorly nutritious branches of coniferous
trees and roots digging out from ground. On forepaws of porcupine there are
curve claws with which help animal gets food to itself and effectively protects
from enemies. When the animal moves on four legs, it leans against lateral surface
of fingers, claws thus are directed by tops to each other.
This species of animals lives in temperate zone frequently rising highly in
mountains. Therefore in winter it should endure rather strong frosts. Fur helps
it to do it becoming to winter thicker and denser. Colouring of fur is changeable,
as well as at its desert neighbour: there are variants of colouring from light
brown up to grey and straw-coloured. In fur the main protective weapon of the
huge wood porcupine is hidden – very thick (up to 2 cm at the basis) needles
up to 40 - 60 cm long. They are especially numerous on neck (from above and
from sides) and shoulders and then grow by strip along back (up to the basis
of tail). Thus, all places which predator prefers to attack are reliably protected.
Needles are mobile; animal can lift and lower them depending on a situation.
Sometimes (especially if the predator is weak or inexperienced) giant porcupine
itself can attack it, striking impacts by needles growing on shoulders. Besides
it can strongly bite by huge incisors. During fight the porcupine roars for
frightening. But usually adult animals seldomly show their abilities: while
they are strong and healthy they practically have no enemies. And cubs of huge
porcupines can become easy catch of predators departing far from mother.
At the female in pack there are 1 – 2 cubs. The new litter appears once a year,
but young growth long time stays with the female: last year's cubs continue
to feed with mother after birth of new ones, abandoning her up to the time of
sexual maturity (at the third year of life). Cubs are born without needles being
overgrown by them at the first month of life. But the newborn wood porcupine
is covered with wool, can see and already in half an hour after birth studies
to walk. The wool of cubs of the first year of life is dark but after the first
winter, during spring moult, colouring is shown which the animal will have all
next life.
Sexual maturity comes at the third year of life. At this time young animals
are already independent, and take part in courtship games which take place at
the end of summer. On borders of individual possessions battles of males for
the right of pairing with females happen. But these tournaments are not as substantial
as at the desert bear porcupine: usually two males battle for one or two females.
But fights happen fierce seldomly: more often all events may end by might demonstration.
Contenders peel bark on tree trunks, pull out bunches of grass from the ground,
urinate trees plentifully and snort loudly. Females observe of the duel from
the side stimulating contenders by characteristic rumbling and spraying strongly
smelling urine.
Pairing proceeds not for long and animals miss soon. But during carings the
excited males can take cubs of any female for contenders and attack them. Frequently
grown up young growth survived in winter and has got stronger, perishes from
attacks of males in courtship season. But porcupine has successfully survived
in youth can live about 60 years.
Striped
tree porcupine (Bradycoendu lanceolatus)
Order: Rodents (Rodentia)
Family: Wood porcupines (Erethizontidae)
Habitat: woodlands of South America.
At the border of Holocene and Neocene, when the most part of rainforest of South
America disappeared as a result of global cooling and climate aridization, the
most part of tropical rainforest animals died out or had to change the way of
life to survive. Among them there were various New World porcupines from South
America, quite unusual rodents, which felt safe from the numerous enemies due
to their quills.
Among mammals, rodents express a very wide adaptability, and have adapted to
live everywhere, from the mountain slopes to fresh water. Some of their Neocene
descendants even turned to marine animals,
but the evolution of South American porcupines did not reach such extremes.
Most part of them (for example, the sidespiny
porcupine) still represents tree-dewelling animals, but there are exceptions
in any rule, and the striped tree porcupine is one of these animal exceptions.
Striped tree porcupine is a relative of sidespiny porcupine and is a relatively
large rodent, reaching almost 1.5 m in length and weighing up to 15 kg. It is
covered with brown fur, from which very brightly colored quills protrude; the
contrast combination of colour of quills and fur creates a striped zebra-like
pattern, which makes this porcupine less noticeable among vegetation. The quills
of this animal are longer than those of its Holocene predecessors. This species
has short strong tail that serves as a support at tree-climbing and ground digging.
Unlike its relatives, striped tree porcupine is rather ponderous and spends
only part of its life in the trees. It often comes down on the ground, searching
there for some edible plants, bathes in dust and escapes from enemies (mostly
birds of prey). This lifestyle has developed in connection with the fact that
this rodent is one of the most southerly species of its family, and lives not
in rainforest, where the climate is humid year-round, and the land is often
waterlogged, but much farther south, where the climate is drier, with pronounced
seasonality, and drought may take place in summer. As a result, the ancestors
of this animal had to develop a new way of life.
The food of this animal includes various fruits and lush greens of ground-dwelling
plants. In dry season striped tree porcupine eats roots, digging them out from
the ground with its powerful claws. It supplements this diet with cockroaches
and grubs, founding them in the ground and among the fallen leaves.
In autumn the mating season of this species begins and females call to the males
loudly; this call sounds like cat’s meowing. If several males gather around
a single female, they will begin to push each other away from the female, until
the most powerful and enduring male remains. From that point on, the male porcupine
continually keeps near the female. The male mates with the female until the
latter is ready to accept the male’s courtship, and it also drives away other
males from the female. While the female is pregnant, male stays close to her
– this species forms a family to rear the posterity. 1 – 2 well-developed cubs
with opened eyes are born at the female three months later. The cubs are cared
for by both parents, and they even carry them (sometimes in turns) on their
backs. Unlike the side-spined porcupine, this species’ quills grow both on sides
and on the middle part of the back. Only the hind part of the back and the base
of the tail do not have any quills. The cubs sit on this part of the body of
adults, and the adult animal covers them up with its tail, curving it upwards.
There are long and moveable quills around this part of the porcupine’s body.
In case of danger they lock over the cubs to protect them.
The young of this species grow rapidly, and the next year they become completely
independent. They become capable of reproduction at the age of two years.
The maximum life span of striped tree porcupine is up to 20 years, but the most
part of animals survives only up to 10-12 years.
This species of mammals is discovered by Bhut, the forum
member.
Translated by Bhut.
Andean
social cuy (Sociocavia andina)
Order: Rodents (Rodentia)
Family: Cavy (Caviidae)
Habitat: South America, Andean Plateau.
In face of the anthropogenic onslaught, some resilient species were capable
of surviving. In South America, the guinea pigs (genus Cavia) were one of these
survivors. In the Neocene one of their direct descendants is the genus Sociocavia,
the social cuy. The members of this genus are highly social animals that form
large colonies with burrows, which can be used by other animals. One of the
representatives of this genus is the Andean social cuy, a species endemic to
the Andean Plateau.
The Andean social cuy have 42 cm of length, with males being slightly heavier
than the females. They are generally tan in color, with lighter-colored bellies.
However, there could be color variation in their pelt, such as dark fur on their
back in black and brown tones. Their forelimbs are strong, with them using it
to dig complex tunnels. The tail is very short.
This mammal is a herbivore and feeds on grasses and other herbs. It's diurnal,
mainly emerging in the early morning to forage and again in the evening.
During the winter months, these rodents don't hibernate. They continue to leave
the burrow to forage, but will enter a state of torpor at night to conserve
energy. Torpor is categorized by a drop in metabolism, heart rate and respiration
similar to hibernation, but is involuntary and shorter in duration. On average,
an Andean social cuy will lose twenty percent of its body weight during the
fall and winter seasons when they go through bouts of torpor. As winter progresses,
the amount of time spent in torpor increases.
The Andean social cuy is a highly social rodent. They form large colonies with
several individuals. Colony size may range from five to thousands of individuals,
and may be subdivided into two or more subgroups, based on topographic features.
These groups may contain 15–26 family groups. There may also be subgroups, called
“wards”, which are separated by a physical barrier. Family groups exist within
these wards. Most social cuy family groups are made up of one adult breeding
male, two to three adult females and one to two male offspring and one to two
female offspring. Females remain in their natal groups for life and are thus
the source of stability in the groups. Males leave their natal groups when they
mature to find another family group to defend and breed in. Breeding takes place
at the beginning of austral spring. The gestation period is about 62 days and
females can have five litters in a year. The number of young born averages two
and ranges from one to five. The minimum age at which the young adults can breed
is thirty days. They have a lifespan of 7 years.
This mammal species was discovered by João Vitor Coutinho, Brazil.
Koriko
pexerumen (Koriko insularis)
Order: Rodents (Rodentia)
Family: Cavy (Caviidae)
Habitat: Archipelago of Fernando de Noronha, forests and mountain slopes.
During the Holocene, humans had caused several impacts on the environment. Among
their actions was the introduction of exotic species and the places that most
suffered with it were the islands. With the disappearance of the human species,
these animals remained and some evolved in new species during the Neocene. One
of these cases was the koriko pexerumen, the descendant of the population of
rock cavy (Kerodon rupestris) that was introduced in the main island of Archipelago
of Fernando de Noronha, close to the coast of Eastern South America. Koriko
pexerumen is the name in the Xukuru language of Pernambuco for the rock cavy.
The koriko pexerumen is endemic to the largest island of the Archipelago of
Fernando de Noronha, inhabiting its forests and rocky areas.
The body length of the koriko pexerumen is 95 cm, with a shoulder height of
30 cm. The fur is auburn and pale grey at the underbelly. Its legs are longer
than that of their ancestors, giving them the appearance of a small and primitive
ungulate. However, it has no hooves, but instead several small nails that remind
hooflets. Their feet are adapted to allow them to move through rocky surfaces.
The ears are short and the tail is nonexistent. They developed strong incisors,
which is used to break hard fruits and shells.
These animals eat fallen fruit, leaves and roots. When feeding, they will stay
near each other as a protection from predators. Sometimes they can eat eggs
and even shellfish on the seashore.
These animals are diurnal and polygamous, with a male and two females mating
and staying together for the rest of their lives. They form groups composed
by the male, his females and their offspring. The female gives birth to a litter
of 2 to 4 young after a gestation of 4 months. There is no defined mating season,
with births occurring many times during the year. The birth usually occurs hidden
in bushes and dense vegetation. The young are born well developed and are capable
to follow the parents shortly after the birth. The old offspring will help the
parents to take care of the young siblings. Both females will help each other
to take care of the offspring. They are weaned with 16 weeks and reach sexual
maturity with 2 years. Within 4 years, koriko pexerumen leaves the family to
form their own. The lifespan of this species is of 19 years.
This mammal species was discovered by João Vitor Coutinho, Brazil.
Rafter
coypu (Paracoypu totorae-lacustris)
Order: Rodents (Rodentia)
Family: Coypus (Myocastoridae)
Habitat: Totora Lake, Andes.
Along Pacific coast of South America the extended mountain circuit – Andes –
stretches. Conditions of life in this area sharply contrast with an environment
of rainforests of Amazonia or savannas of Gran Chaco. The high mountain world
is an empire of cold, sharply expressed daily and seasonal change of temperatures
and superfluous sunlight. In ice age the high-mountainous climate was dry and
very cold. In Neocene the climate began more humid; the increase of amount of
rains had caused occurrence of large lake at Altiplano plateau. In human epoch
at this plateau there were some lakes among which Titicaca lake was the most
known one. Gradually all lakes of Altiplano had merged together, and at the
plateau the huge Totora Lake appeared.
Coypu reached significant evolutionary success in South America. Among descendants
of this animal there is a unique group of mammals – algocetids, huge aquatic
animals. It is the most deviated and specialized branch of rodents making the
separate order. Other descendants of coypus are less specialized, but it does
not prevent them to prosper in nearwater biotops. In South America they are
analogues of beaver and muskrat, and settle down to the cold south of continent.
One descendants of coypu has developed high mountains, and its population lives
at Totora Lake. It is rafter coypu, animal leading unusual way of life and differing
in advanced building behaviour.
Rafter coypu is large species of aquatic rodents keeping anatomic features of
the ancestor. The velvety fur with small amount of guard hair serves to this
species as fine thermoisolation in very cold water of the lake. In fur colouring
significant polymorphism is observed – among animals there are individuals from
grey and straw-coloured up to brown and black ones. Rafter coypu carefully looks
after fur, and combs it by claws for a long time. Forepaws at this animal have
mobile fingers and are adapted to manipulation with small objects.
Ears of this mammal are short, and eyes are small. This animal receives a significant
part of information about the world around with the help of smell sense. On
a muzzle of rafter coypu numerous whiskers grow, helping to orientate in darkness.
Main moving organ of this animal at the swimming is its tail. It is wide, rounded
and looks like cut off at the back edge. Tail is covered with hairless tight
skin and contains the advanced fat layer; therefore warm does not leave an organism
of animal through naked skin of tail. In water rafter coypu swims with the help
of tail, having pressed paws to body and making wavy bends by backbone. During
feeding at shoaliness animal swims slowly, with the help of webby hind legs.
Rafter coypu is able to dive to the depth up to 10 meters and stays under water
till about 5 minutes. But it prefers to use this skill only in case of extreme
danger, because it feeds in coastal shallow zone.
This species is strictly herbivorous animal, eating rhizomes of totora reed
(in language of Indians of Andes it is the name of the reed known in human epoch,
hence the name of lake) and tubers of water plants. Eating rafter coypu simultaneously
receives building material for its shelters. This animal digs out sprouts of
reed with the help of strong incisors and claws of forepaws. At the dug out
plants rafter coypu bites off and eats rhizome and the bottom part of stalks,
and pulls a bunch of poorly nutritious stalks to original shelter in which it
spends night.
These animals build rafts and huts on them of reed. To construct a raft, rafter
coypu bites off stalks and branches of coastal plants, drags them to common
heap floating on surface of water, and constantly renews the construction. As
a rule, some animals begin simultaneously construction of raft. They constantly
drag the cut off rods and stalks of reed on rafts, and trample them down, moving
on rafts. On such raft the hut is placed, in which the pair of adult animals
and their posterity lives. Hut has the hemispherical shape, reaches one meter
height and has thick friable walls. The entrance opens sideways; if the raft
is great enough, near to an entrance animals specially make an outlet to the
water and clear it regularly, dragging off from it the rests of plants.
Small rafts, constructed by separate family groups, may gather to form big floating
islands and give a haven for other creatures. When some rafts are linked together,
their inhabitants do not try to uncouple them – rafter coypus are sociable enough
and between them there are no serious conflicts. They simply throw more reed
stalks on edges of linked rafts and form unite large raft. Running on it, animals
trample down its surface, and the rests of their food serve as a building material.
Such rafts may exist for many years, constantly being renewed. The part of reed
rhizomes not eaten by rafter coypus sprouts in thickness of rotten stalks, and
additionally strengthens the construction. Sprouts of reed make the way through
surface of raft, and serve as good top dressing for these animals. In the middle
of raft animals eat sprouts completely, and on edges the bitten stalks of totora
reed plentifully branch, forming green fringe.
The pair of adult animals rises within summer one litter – up to five cubs.
The young growth of these animals is born with open eyes and covered with rich
wool. The female cares of posterity within two months, and at this time male
is engaged in repair and construction of raft alone. Young animals leave dwelling
already at week age, and monthly cubs swim perfectly. They live in common with
parents within the first winter. At this time young females reach sexual maturity,
and males become large enough (they mature later). Females leave parental raft
and settle on rafts of other males, forming pairs with them. Young males are
banished by their own father, and they start to build their own rafts or occupy
and renew empty huts.
The idea about existence of this species was proposed by Momus, the forum member.
Chilean algocetus (Algocetus chiliensis)
Order: Algocetiforms, “algae whales” (Algocetiformes)
Family: Algocetids (Algocetidae)
Habitat: Pacific coast of South America.
Human activity and climatic changes at the boundary of Holocene
and Neocene had caused fast disappearance of plenty of species of sea animals.
Sirens had become extinct irrevocably in that epoch, and cetaceans and pinnipeds
are present as rare relic forms in Neocene. But after mankind disappearance
and stabilization of climatic conditions life began to restore the lost positions
and to form new ecosystems again. Seas of northern hemisphere in Neocene are
occupied by birds – gannetwhales
and flightless loons. These
animals dominate in Atlantic and Arctic oceans. In Pacific Ocean large flightless
hatchetbills (a kind of auks) replace them, and at the Asian coast algal geese
and penguin duck also live. But in cold and temperate waters of southern hemisphere
birds are presented only by flightless penguigulls
of several species. These birds eat animals, and mammals came to the place of
herbivorous marine animals.
Sea mammals of southern hemisphere descend from South America. They are descendants
of coypu (Myocastor coypus) widely settled in a moderate and subtropical zone
of continent. Coypu had successfully passed the period of anthropogenous pressure,
and even had widely settled due to human activity – it was domesticated.
Sea mammals of Neocene are herbivores. In early Neocene when sirens – completely
aquatic herbivorous mammal – had disappeared, evolution of coypu had gone especially
roughly. Till some millions years descendants of coypu had passed approximately
the same way, than cetaceans in early Eocene epoch did – from small semiaquatic
creatures up to completely aquatic large mammals lost connection with land.
Descendants of coypu form the separate order of mammals – Algocetiformes, “algae
whales”. In their anatomy features of rodents and features of obligatory aquatic
mammals (whales and sirens) developed convergently are combined. Algocetids
are large animals; smallest of them reach the length of 3 meters, and at coast
of Antarctica and at the south of South America the giant about 7 meters long,
weighting over 6 tons, lives. They resemble sirens externally a little; it is
the result of similar habit of life.
The muzzle of algocetes differs in advanced, mobile and partly doubled upper
lip on which bottom side lines of corneous combs grow. With the help of such
lip the animal easily scrapes off from stones seaweed which it eats. The bottom
jaw differs in massiveness, and incisors in it are very large and wide. Such
incisors help to tear off from stones not only seaweed, but also sedentary animals
– mollusks and barnacles.
Nostrils of algocetids are shifted to the top part of head – such structure
permits the animal to breathe, having put out from water only small part of
head. Large individuals emerge for some minutes, and then can hold the breath
till half an hour. Cubs emerge to breathe much more often.
Though algocetids are completely marine animals, not capable to leave on land,
they swim not so well. These animals prefer to move in shallow water similarly
to Steller’s sea cow – making steps on bottom by well advanced mobile forward
extremities. On them two large claws (on II and III fingers), assisting to cling
to bottom, are present. IV and V fingers are reduced, lack of mobility, and
only support a plane of flipper. The thick layer of fat serves not only thermoisolation,
but also a fine float, reducing loading to extremities of animal. Rear legs
of algocetids had reduced completely, and on the tip of tail the wide rounded
fin is developed. Algocetes are sluggish swimmers, and during storm they prefer
to hide in rich thickets of seaweed which soften force of waves.
Eyes at algocetes are small, but sight is rather quite good. Animals prefer
to live in places with transparent water, supporting visual contact to each
other. They also can produce sounds with the help of air bladders in nasal ducts.
External auditory meatus at algocetids is not closed, but an eardrum is thick.
Bones of internal ear are isolated from skull by special fatty tissue, like
at cetaceans. The upper lip is covered with thick whiskers (hair thickness is
up to 3 mm), which help to search for food in muddy water.
Algocetes keep in families including a breeding pair or the male and two females,
and their posterity of last two – three generations. At the largest species
living near Falkland Islands and in Antarctic Region, herds of several tens
of individuals including some breeding groups are formed. Pregnancy lasts about
16 months at large species and about 11 months at smaller ones. As a rule, the
female at all algocetid species gives rise to two well advanced cubs. Only at
the largest species there is only one cub. Cubs are born covered with thin fur
which drops out till first days of life. At the delivery the female is surrounded
by neighbours push the cub out to the surface of water. When the cub starts
to move, they leave it, and only its own mother cares of it. But if necessary,
females of algocetuses protect young growth against large predatory fishes together.
Nipples of the female are located on sides of body, approximately at the level
of pelvic girdle hided in thickness of muscles. To make more convenient to cub
to be fed, the female lies down on the side, and the cub sucks milk, having
put nostrils out to the surface of water, and breathing. Cubs suckle milk about
half-year, but already from bi-monthly age start to eat food of adult animals.
In Neocenic fauna algocetids are presented by one family and four species.
Chilean algocetus lives at Pacific coast of South America, and the separate
population with doubtful specific status inhabits waters of Galapagos Islands.
The length of this species reaches 4 – 4,5 meters. This species lives in coastal
zone, preferring areas of coast protected from storm waves by barrier of small
islands. Its distribution is closely connected to cold current flowing from
coast of Antarctica. These animals eat brown and green macroalgae. Due to their
fast growth animals lead almost settled way of life, moving within the borders
of the territory to some kilometers. This species willingly swims to lower reaches
of short rapid rivers flowing down from Andes to Pacific Ocean. In such way
these animals escape of ectoparasites perishing in fresh water.

Picture by Alexander Smyslov
Patagonian coypocetus (Coypocetus patagonicus) inhabits rivers of Atlantic coast of South America. It is the smallest species of algocetes – the length of adult individuals hardly exceeds 3 meters. It constantly lives in fresh water and eats aquatic flowering plants. At this species there are rather large claws – with their help the animal digs out rhizomes of water plants and protects itself from enemies. Its forepaws are mobile; with their help animal can bring food to mouth. Digging out silt and eating water plants, these animals interfere with bogging of the rivers. To wintering these animals migrate from upper streams and move down to the mouth of the river, forming there big congestions destroying water vegetation along many kilometers of riverbanks. At the lack of forage they may swim for the short time to coastal sea waters.

Picture by Alexander Smyslov
Closely related to the previous species coastal coypocetus (Coypocetus
littoralis) has wide area: it lives along the Atlantic coast
of South America. This animal does not meet in fresh water at all. Its
length
is about 4 meters. Coastal
coypocetus is rather heat-loving, and does not settle far to the south.
But it also does not love very warm water; therefore the northern border
of its
area does not reach mouths of Amazon and Hyppolite rivers.
All species of algocetuses described above live in shallow waters and
do not swim far from coast. Therefore Falkland paralgocetus (Paralgocetus
falklandicus),
the largest species of algocetes, looks the exception among them. It
is the true giant among algocetes, reaching the length of 7 meters. It
lives
at
the coast of Falkland Islands and rather frequently leaves in open ocean.
This
animal frequently feeds in congestions of floating brown seaweed in southern
part of Atlantic. It differs from other species of algocetes in very
big fat incrustation expanding on forward part of head between nostrils
and
upper lip.
This pillow is used as ram during intraspecific duels. At this species
there are small dulled claws on forward fins, and sometimes the claw
on II finger
is completely reduced. A tail fin of Falkland paralgocetus is triangular-shaped,
but it is not such wide, as at whales.
Due to the size and thick layer of fat (up to 15 cm) it is the most cold-resistant
species of algocetids, reached the certain success in evolution. Rather
recently (according the geological measures) the small population of
this species
migrated to coast of Antarctica, and later descendants of first settlers
settled generally
coasts of continent. The Antarctic representatives of this species became
more omnivorous, and at polar night marine invertebrates frequently make
up their
food.
Caribbean algocetus (Xenalgocetus diodon)
Order: Algocetuses (Algocetiformes)
Family: Algocetuses (Algocetidae)
Habitat: Caribbean Sea.

Picture by Carlos Pizcueta (Electreel)
Herbivorous sea mammals in human epoch were presented by
sirenian order – not numerous group of sluggish aquatic mammals. These animals
bred slowly and differed
in poor ingenuity. It made them very vulnerable for hunting, and their number
began to reduce even in early historical time. Despite of strict protection,
residual populations of these animals had not gone through human epoch. In
Neocene various groups of vertebrates competed for a role of sea herbivores.
In Tanganyika passage algal
turtles (Thalassotestudo algophaga) live, but they
did not manage to become dominant herbivores of seas – their distribution
is limited only to warm waters of passage. Descendants of South American
rodent
coypu (Myocastor coypus) have achieved the much greater success. At coast
of South America the representatives of new group of mammals descended from
them
very fast to evolutionary measures; these ones are algocetuses, herbivorous
animals permanently living in sea. The majority of algocetuses does not cross
equators and is settled only in coastal waters of South America and Antarctica.
Only one species managed to step to the north, to Caribbean Sea. Thickets
of sea spinach (Thalassolatucca crystallophylla),
floating aquatic plant, are the basic source of food for Caribbean algocetus,
heat-loving species of herbivorous
water mammals.
Caribbean algocetus is a second-largest species in order after Falkland paralgocetus.
The body length of adult individual reaches 5 – 6 meters. Layer of blubber
at this species is thin – it is a consequence of life in tropics. However
the blubber layer is necessary for it serving as an adaptation facilitating
swimming.
Caribbean algocetus spends a significant part of life at the surface of water.
At this species the extra adaptations were developed, allowing the improving
of heat radiation: skin is covered with the numerous cross wrinkles increasing
its surface. Tail fin is wide with rounded flukes – it is not only swimming
organ, but also the adaptation for heat emitting. Caribbean algocetus swims
slowly and dives only to depth of about 20 – 30 meters only in case of extreme
danger, for example, in storm. At its way of life speed and ability to dive
deeply become almost excessive, because its forage is always on surface of
water.
Rear limbs of this animal had disappeared completely –only rudiments of sacrum
and hip remained from them. But front flippers of this species are very mobile
– they are rather long and can bring food to mouth. Front limbs keep mobility
in elbows and even prehensile function is kept partly at them. The second
and third fingers of forward limb are covered with the common skin cover,
are separated
from the fourth and fifth fingers by small interval and can move independently
of them. Due to this feature the limb capable to grasp food is formed. On
second and third fingers large claws are advanced.
Caribbean algocetus eats mostly sea spinach – floating species of aquatic
plants living on surface of Caribbean Sea. This plant lives in stable conditions
and
always gives plentiful growth, therefore groups of these animals are always
provided with habitual forage. Caribbean algocetus, however, does not leave
far in sea – it is possible to find only separate males at great distance
from the coast. The basic population prefers to be fed in thickets of sea
spinach
above depths up to 100 meters. This animal may be seen frequently in shallow
waters; it swims there for thickets of sea spinach carried by wind, and also
for aquatic plants growing on shoaliness. In silent weather this animal occasionally
comes to edges of mangrove forests and in high inflow browses leaves of mangrove
trees. Feeding on branches of mangrove trees, Caribbean algocetus swims in
shallow water, lies sideways and drags branches of mangrove trees to mouth
by forelimb. In outflow this animal tries to swim to deeper places. Occasionally
the careless animal can get in a trap among mangrove trees: when passages
become too shallow, animal stays on shoaliness and may die from heatstroke
or choke
under weight of its own body.
Soft vegetative food had resulted in significant changes of dental system
of this species. Molars are lack completely, and they are replaced with corneous
plates with ribbed chewing surface. Incisors in top jaw had turned to tusks
similar to tusks of walrus or dugong, but shorter, rather than their ones.
They are used in tournament fights of males. Female tusks are smaller. In
lower
jaw incisors are reduced completely.
Once in two years female gives rise to one large cub. Once in ten – fifteen
cases of birth twins may be born. The one and a half meters long cub quickly
grows, eating fat milk. Till two months of life it doubles its initial weight
and gradually passes to feeding on plants though it is fed with milk almost
up to one-year-old age. At four-year-old age the young animal is ready for
breeding.
Caribbean algocetus lives in herds under the leadership of large female.
Usually such herd totals up to 20 – 30 adult females and their posterity
of the first
years of life. Young males stay in herd up to their sexual maturity. Adult
males form large barchelor herds which break up in breeding season. The female
ready to pairing emits with urine the substance which taste helps males to
find her out. In courtship season male behaves aggressively relatively to
contenders. Battling for the female, males snap each other’s flippers and
tail flukes.
Battling with the help of tusks, they strike each other lateral impacts by
head. Tusk points are used only for protection against predatory fishes.
Life expectancy of Caribbean algocetus reaches 35 – 40 years. Sharks are
main enemies of these animals, but these mammals are more often suffer from
the
parasitic invertebrates boring into skin. Animals escape from them, coming
into fresh water where parasites perish. With the same purpose during the
inflow algocetuses scratch against roots and trunks of mangrove trees.