

Bestiarium of
the future - Reptiles and Amphibians
|
 |
| In this section descriptions of various animals and plants, which could live on the Earth in Neocene epoch. The section will be supplemented as new ideas about possible ways of evolution of life will appear. If readers will not find here any species placed here earlier, it means that it is a reason to search for a new chapter in English version of "The Neocene Project". |  |
|
Amphibians and Reptiles |
Reptiles
|
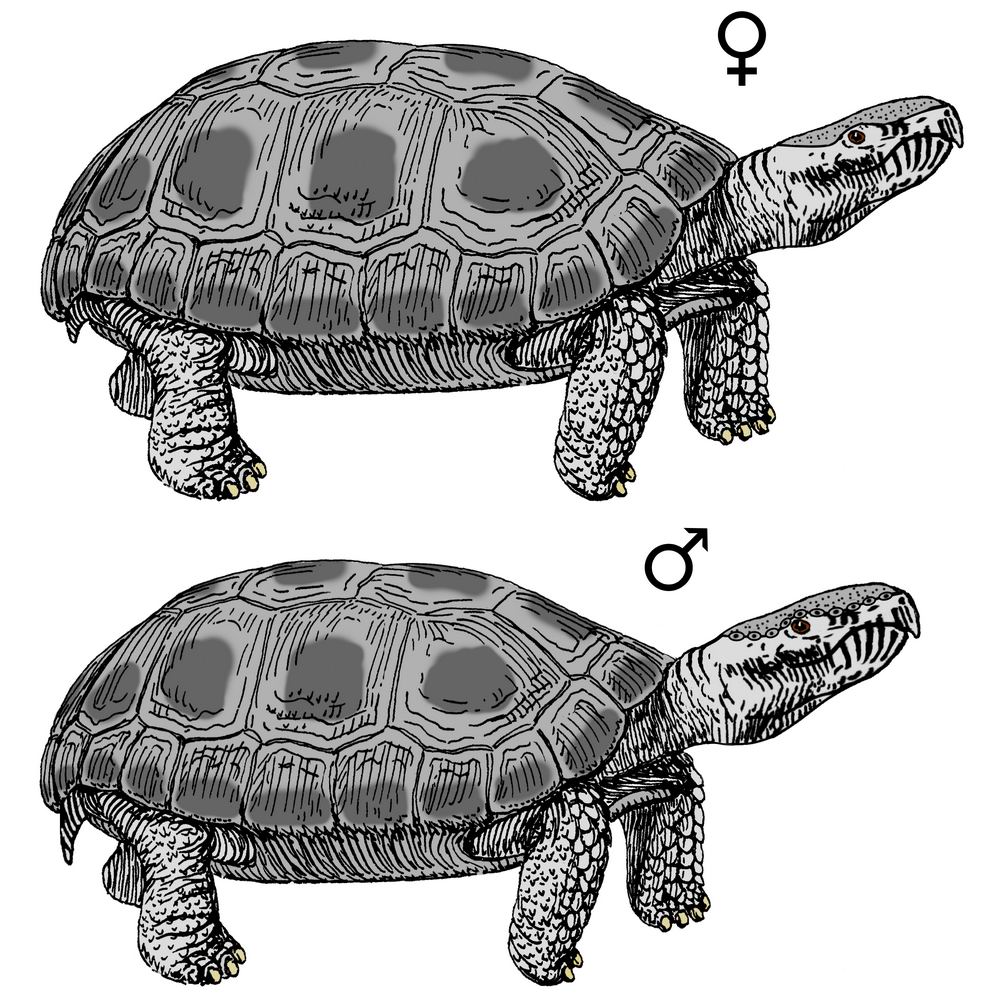
Long-bearded
dracochelys (Dracochelys palustris)
Order: Turtles (Testudines)
Family: Chelydridae (Chelydridae)
Habitat: freshwater reservoirs of Central America.
Despite the fact that the end of the Holocene and the beginning of the Neocene
were marked by significant changes in the biosphere and were accompanied by
mass extinction, turtles – at that time, perhaps, the most ancient reptiles
of the Earth – survived and subsequently evolved successfully, occupying ecological
niches in newly emerging natural communities. Even among this group of reptiles,
the Chelydridae family was of a quite ancient origin, but its representatives
successfully exist in the Neocene. The rivers and lakes of North America are
populated by a giant trapperturtle,
and to the south of it, at the territory of Central America, in the tropics
south of the Mexican deserts and highlands, its smaller relative, long-bearded
dracochelys, lives.
This species is a medium-sized turtle compared to its ancestor of human era:
males grow up to 1 m in length, including a long tail; females are up to about
70 cm long. This reptile has a well-developed carapace with three rows of conical
scutes with sharp edges, which gives the long-bearded dracochelys protection
from predators – large mammals and birds, as well as from other freshwater turtles.
The carapace itself is initially of a dark shade, from dark green to almost
black, but in adults it is usually completely overgrown with filamentous and
other algae – like most snapping turtles, long-bearded dracochelys is non-mobile
and spends most of its life buried in soft bottom mud, hiding its much lighter
belly (the color of the abdominal plates in this species ranges from sandy to
gray). The plastron is cross-shaped, leaving the bases of all limbs open. The
hidden reptile lies with only its neck and head sticking forward.
The nostrils and eyes are shifted to the upper part of the head. Thus, long-bearded
dracochelys can breathe without betraying its presence to predators: it only
needs to stick out its nostrils above the surface of the water. In addition,
in this way it lures prey. The North American snapping turtles had worm-like
“baits” on their tongues, thanks to which small fish swam into the mouths of
these reptiles themselves; instead of it, long-bearded dracochelys has a “beard”
on its neck – numerous thin skin outgrowths that attract small underwater creatures,
which this turtle species feeds on. In part, these outgrowths function as a
skin respiration organ, allowing the reptile to stay underwater for a longer
time – they are covered with a thin epithelium penetrated with a network of
blood vessels. Long-bearded dracochelys is an omnivorous reptile compared to
the trapperturtle: it eats plants (in small amounts), aquatic invertebrates,
fish, amphibians, reptiles (including smaller turtles of its own specied), catches
chicks of aquatic birds and does not disdain carrion.
Long-bearded dracochelys is active all year round; like other tropical species,
this turtle does not have a pronounced mating season. Like all turtles, long-bearded
dracochelys does not form pairs, but this species is not very territorial, and
individuals of approximately the same size and age are tolerant of each other.
Fertilized females of this species leave the water and look for a place with
loose soil to lay their eggs – there are up to 15-20 eggs in clutches. This
is where the parental care of the long-bearded dracochelys females ends, and
they never return to their eggs again; the juveniles hatch after about 2 months
and immediately search for a suitable place to live – as a rule, it is a freshwater
swamp, preferably densely overgrown with aquatic vegetation, with a soft mud
or muddy bottom. They grow slowly; sexual maturity comes at the age of 18-20
years. Having reached adult size, these reptiles have actually no enemies.
The lifespan of long-bearded dracochelys is over a hundred years, sometimes
up to 130 years.
This reptile species was discovered by Bhut, the forum member.
Ink
turtle (Sepiochelys atramentophora)
Order: Turtles (Testudines)
Family: Sliders and Terrapins (Emydidae)
Habitat: rivers and lakes of Central Africa.

Picture by Alexander Smyslov
Conservative and not too numerous group of turtles had suffered the big damage
as a result of human activity. Their largest species dwelt in islands had
died out the first. After them some endemic continental species which habitats
were
destroyed by people had died out. The most mass species of sea and freshwater
turtles had suffered because of overhunt and unlimited egg gathering. In
human epoch areas of the majority of turtles had strongly reduced. Approach
of the
ice age became one more impact to the variety of this order and besides appreciably
reduced the areas suitable for life of these reptiles.
In Neocene the warm climate had permitted to realize chance of a survival
to not numerous survived representatives of turtles. In various places of
Earth
there were species adapted to
extreme conditions of inhabiting, or on the contrary, prospering and
dominant in favorable conditions. In some habitats turtles even have turned
to main predators of the
ecosystem. But the majority of turtles remained rather small animals, especially
at the continents.
Presence of predators in the ecosystem stimulates the development of various
protective adaptations. In rivers of Central Africa the place of crocodiles
extinct till Holocene is occupied by the huge representative of Squamata
order – huge crocodile monitor lizard.
This reptile hunts with the help of sight and sense of smell, and force of
its jaws permits this reptile to eat any animal
from ones living in rivers. One of turtles living in the neighbourhood of
this monster had developed the way to avoid the danger coming from the giant.
It is the aquatic turtle of rather usual appearance – it has flat carapace
of rounded outlines, webby paws and large head on mobile and strong neck.
Usually it slowly swims near the bottom, searching for forage: freshwater
crabs, snails
and carrion. When the crocodile monitor lizard or predatory fish starts to
chase it, this turtle tries to rescue by flight. But, if the distance between
the turtle and its persecutor became critical, the reptile exposes “the second
line of defense”: it emits a cloud of black liquid from cloaca and receives
some seconds to hide. Hence the name of this reptile – it is ink turtle.
Ink turtle belongs to rather large species: carapace length of adult individual
is up to 60 cm at weight over 30 kg. The carapace of ink turtle is colored
olive with small black speckles. At old individuals it becomes covered by
algal layer, and even by aquatic moss. It serves as additional masking of
the reptile.
Skin on legs, neck and head is brownish with set of light spots. On head
of animal spots merge to longitudinal strips, and the throat looks much more
lighter,
rather than skin. Paws of the reptile have palamas between toes – ink turtle
swims very well, accelerating momentum under water up to 30 kms per hour.
On forepaws of males increased claws grow: they are very long (up to 5 cm),
strong
and sharp. With their help males fight in courtship season. Claws also help
the male to be kept on the carapace of the female till the pairing.
The main weapon of ink turtle is the liquid of black color emitting out in
case of danger. In walls of cloacal bladders at this species numerous ink
glands accumulating pastelike black dye had appeared. When the turtle is
in danger,
it squeezes out their secret to cloacal bladders where it mixs up with their
contents and is emitting in water as black liquid with unpleasant smell.
Ink of this turtle is a little bit similar by action to cephalopods ink:
it has
poorly narcotic property, and may paralyse olfactory nerves of predator.
It has an action at reptiles in some minutes, but sense of smell of fish
may be
paralysed till half an hour and more. Even the most young ink turtles having
length of carapace about 5 centimeters are able to emit ink.
Breeding at ink turtles lasts the year round. Courtship displays of these
reptiles are rather primitive: males try to banish each other from the female.
The strongest
male, having got rid from contenders, starts to swim near the female, trying
to bite slightly its paw or neck. When the female shows the submission, stopping
attempts to departure, male couples to it and right after this act abandons
the female.
Eggs form in organism of the female till 25 – 30 days. For egg laying the
reptile leaves in wood, digs out the small hole in ground and lays simultaneously
up
to 8 – 11 eggs. Having dug the nest, it is not interested in destiny of posterity
any more. The incubating lasts about 40 days. For one year the female can
make up to three clutches.
Ephemeral
Slider (Ephemeremys dormus)
Order: Turtles (Tedustines)
Family: Sliders and Terrapins (Emydidae)
Habitat: Yearly drying lakes and ponds found in the deserts of south-western
Meganesia.
Aestivation is a technique used by many animals to survive times of drought
and heat. Amphibians of various sorts do this, but reptiles are better adapted
to deal with drying conditions. The domestic Red Eared Slider (Trachemys) was
introduced by man to Australia, and has developed into many freshwater forms
in the Neocene of Meganesia, sometimes displacing the smaller kinds of native
side-necked turtle. One kind is specially adapted to aestivate during periods
of drought, Ephemeral Slider.
This species is a relatively small turtle, with a carapace length of no more
than 35 centimetres. Shell is broad and slightly tapered, relatively flat on
top with an up-turned front margin, and plastron is extensive. Shell is coloured
buff brown with radial bands of dull yellow on each scute, skin of this turtle
is pale brown spotted with grey, to avoid overheating. The head generally resembles
its ancestor, but the forefeet bear large robust claws for digging aestivation
burrows. Tail of the male is thicker and longer, and females are slightly larger
than males. It is able to both swim strongly and clamber about overland for
some distance.
This turtle feeds mainly on animal matter, crustaceans, insects, fish and carrion,
but will consume algae and detritus. They will sometimes bask during the day
to regulate their temperature but generally have no need to given how exposed
their habitat is. Breeding occurs sporadically due to rain cycles, and on a
bad year may not happen at all, at the start of the rainy months, courting begins
in earnest with males pursuing females, pawing at them and eventually mounting
them. The female spends a considerable time with her eggs developing inside
her body, and will try her best to regulate her body temperature. During the
height of the rainy months, 3-5 large eggs are laid in mound of earth, heated
by the sun. Babies hatch well developed and are fully able to aestivate during
the following dry season. An internal portion of yolk sac remains after hatching
and may sustain the babies during aestivation. Smaller less vigorous young generally
do not survive the aestivation period. Unlike many other kinds of freshwater
turtle, skin can completely dry out without the turtle becoming ill or dehydrated,
and they are adapted to retain water in preparation for aestivation. They are
most active during the day, and will submerge themselves during the cold of
the night.
Each year when the ephemeral ponds and lakes dry up, these turtles seek a place
to aestivate until the rains return. They seek out an area above the old waterline,
where the soil is damp, but not wet, so as not to obstruct breathing with mud.
The turtle then digs a vertical tunnel some 50 to 70 centimetres deep and makes
a small chamber which it them backfills over itself. During the preceding weeks,
the turtle passes less waste water and builds up retention of body fluids that
will sustain it during aestivation. When aestivating they enter a state of dormancy
where bodily functions and metabolism are severely reduced, and are only stimulated
into activity again when the soil becomes waterlogged after rain. Rainy period
generally happens late in the year and only lasts a few months. If an individual
is uncovered at the wrong time, perhaps by a predator, it goes into shock and
dies. Predators include raptorial birds, large lizards and predatory marsupials.
Lifespan is up to 30 years, though generally shorter.
This species of reptiles was discovered by Timothy Donald Morris, Adelaida, Australia.
Algal
turtle (Thalassotestudo algophaga)
Order: Turtles (Testudines)
Family: Tortoises (Testudinidae)
Habitat: Tanganyika passage, coastal thickets of seaweed. Young growth lives
in estuaries of the rivers.

Picture by Tim Morris, the improvement by Alexander Smyslov
Picture by Alexey Tatarinov - the initial image |
Before man began attack to nature, at sea meadows overgrown
with seaweed and sea grasses, large herbivorous water mammals – sirens (Sirenia)
– grazed. Unlimited hunting has resulted to that dugong and manatees became
very rare, and Steller’s sea cow dwelt in Pacific Ocean had been exterminated
at all. Ecological crisis of the end of Holocene had resulted in extinction
of the rests of sea herbivorous animals. When balance in ecosystems of Earth
was restored, seaweed and sea flowering plants began to occupy much more extensive
territories, than in Holocene – they have taken place of productive communities
earlier belonging to coral reeves. In early Neocene at algal “pastures” only
numerous shoals of omnivorous fishes fed. But later new eaters, reptiles, huge
algal turtles, had added to them.
They are not descendants of sea turtles, existed millions years and suffered
from the human in Holocene, but completely independent group of reptiles. They
had descended from one of species of herbivorous tortoises, and the combination
of unpretentiousness and diet has permitted them to gain sea meadows. Certainly,
many species of water turtles had gone through ecological crisis, but they are
predators and can not pass to vegetarian diet completely.
Algal turtle is rather large species of reptiles: the length of its carapace
reaches 1 meter. As at all turtles living in water, the carapace of algal turtle
has the streamline oval form. Head at this species is very big, with strong
cutting jaws. In case of need this turtle can defend itself from any predator
by bites. Head does not retract under carapace, therefore additional protection
was developed on it: it is covered with special convex osseous scute. The top
part of carapace at algal turtle is colored light brown, but this colouring
can be invisible because of layer of the special microscopic algae settling
on carapace. When turtle creeps out on coast, algae cells form mucous membranes
protecting from drying in the sun, and due to what survive. Therefore carapaces
of turtles, as a rule, have more or less appreciable greenish shade. The plastron
of turtle is colored yellowish-white. Skin on neck and basis of legs is grey-green,
on head and legs gray-brown. Males are larger than females, their plastron in
bend, as at all turtles; around of eye of the adult male the ring of large thick
scales of yellow color develops – it is a distinctive attribute of the sexual
matured male. Besides at males on forepaws the thick claws grow, necessary for
female keeping at pairing.
Column-like legs of tortoises have turned at algal turtle to flat oar-like flippers.
This reptile perfectly swims and can dive for a long time, feeding with underwater
vegetation. But on land flipper-like paws are almost useless: the turtle moves
on sand hardly, dragging body like a seal. However it does not create problems
for turtle: this species of reptiles inhabits eastern coast of Tanganyika passage
belonging to Eastern - African subcontinent (Zinj Land) where large predators
are not found. And against small and medium-sized predators both in water and
on land the algal turtle is reliably protected with carapace.
The most important danger, with which the descendant of ground animals faces
passing to life in sea, is the danger of dehydration: concentration of salts
in sea water is more, than in blood of animals. The organism of ground turtles
is adapted to economical expenditure of internal stocks of water that had permitted
to ancestors of algal turtle to occupy sea. For more effective life in sea water
at turtle in eye-sockets large salt glands were developed. When turtle gets
out on land, it can seem, that it “weeps”. Actually it is the work of salt gland,
removing surplus of salts from blood. The brine flows down on each side of heads
of the turtle having a rest on coast. From time to time the reptile rubs head
against forepaws, cleaning off crust of salt.
On the coast turtles are basking after nourishing dinner, having stretched flippers
in sides. The gathering of turtles from afar is a little similar to rookery
of seals, but almost full silence here reigns: turtles do not utter loud sounds.
Knock of carapaces when any turtle pushes another one, crawling on sand, or
hissing when two turtle males sort out their relationships in breeding season,
is only heard sometimes. Turtles live in gathering in common and at the same
time separately: each of them perceives neighbours only as an annoying obstacle
at the way to meal or the sand heated up by sun. They are united only with presence
of meal – thickets of sea grasses and seaweed waving in shallow waters.
In water awkwardness of turtles vanishes: alternately moving by paws, they dive
to the bottom and bite sprouts of sea grasses. Each turtle is constantly accompanied
with school of small fishes: so it is much safer to them, besides turtle muddles
sand, facilitating search of tiny invertebrates. Some small fishes scrap seaweed
overgrown back of the reptile, others
tear rags of skin exfoliating at shed. The turtle has nobody to be afraid of:
monstrous sharks, capable to bite through half-and-half its carapace are not
found here. Only before storm turtles creep out to the coast and even go deep
into coastal wood: waves easily can carry away any algal turtle to the sea or
(that it is even worse) simply break against rock.
At young algal turtles salt gland is not advanced yet. However, it is not so
vital for them: in youth they do not live in sea water. Algal turtle, as against
sea turtles of Holocene, does not bury eggs at the sea coast. Fertilized females
leave coast, swim up rivers deep into woods of Zinj Land. Turtles from islands
near continental coast should cross a passage for this purpose. In forest turtles
crawl out searching for soft ground, suitable for the nest arranging.
Having found the likely place, turtle digs out by hinder legs small hole where
lays about 30 rounded white eggs. After that it fills nest up with wood dust,
some times crawls across it, and then comes back to the river. Weakened after
egg laying turtles are floating in current, having a rest. At this time they
eat of nothing.
If the laying will not be dug out by inhabitants of wood, after 3 - 4 months
young turtles will hatch out. They at once search for water, with the help of
sense of smell precisely determining concentration of water vapor in air. Having
found the river, young turtles live in it the first year of life, gradually
passing down from wood streams to the basic channel and further to river mouth.
Later, when salt glands will develop, young turtles will leave in the sea. Sexual
maturity at them will come at the age of 10 years, and common life expectancy
can reach 150 years and even more.
Spike-headed
tortoise (Cephalotestudo strobilocervix)
Order: Turtles (Testudines)
Family: Tortoises (Testudinidae)
Habitat: Galapagos Islands, bush and forest.
In human epoch Galapagos Islands were rather well-known due to huge tortoises.
Sluggish reptiles had become original “visiting card” of islands, and even
other name of Galapagos Islands is “Tortoise Islands”. The destiny of these
reptiles in historical epoch had balanced at the edge of nonexistence: in the
beginning because of extermination of tortoises by people, and then because
of their competition with introduced mammals. But tortoises managed to survive
successfully due to the world surrounded them. Galapagos Islands differ in
driest climate though are located in the ocean. At these islands there are
no rivers, and there are no constant water sources. In such conditions in combination
with hot climate few mammal could exist normally. Therefore an ice age when
the amount of deposits had even more reduced, introduced animals had started
to die out, and tortoises, on the contrary, began to master rather actively
becoming empty territories from which they were superseded earlier by introduced
and feral mammals.
Spike-headed tortoise is one of few descendants of representatives of native
fauna of islands. This is a large creature which has kept characteristic shape
of the tortoise: the carapace length reaches two meters at one meter height
and width about one and half meters. Outlines of carapace are rounded, above
the neck the edge of carapace is bent upwards, forming characteristic “collar”.
Carapace colouring is light grey with darker middle of scutes. Legs of this
tortoise are similar to elephant’s ones; they have large hoof-like claws and
thick corneous scutes. Weight of adult reptile reaches 500 kg and more; males
are approximately 20 % heavier than females.
This tortoise eats leaves and branches, and can reach them due to a long neck.
It is able to eat plants up to height of one and half meters above the ground.
But the neck creates a vital issue for this tortoise: it can not retract under
the carapace completely, and predators appeared at islands together with people,
could easily attack these reptiles. Therefore till the process of evolution
at tortoises the interesting protective adaptation giving to them fantastical
appearance was developed. At young tortoises on back edge of head and on neck
some ring lines of corneous spikes protecting head from bite of predator are
located. At adult tortoises bases of thorns gradually expand in continuous
rings of corneous scutes, and sharp tips of spikes gradually erase. But the
adult tortoise is able to protect itself actively: it bites, and its jaws are
very strong: they easily bite off pieces of lignificated trunks of local huge
cactuses. Young tortoises, protecting themselves from attack, brandish a head
(neck at this species is very flexible), trying to strike a blow to predator.
Spike-headed tortoises avoid open places, but in woods and bushes they thread
out the true circuit of wide tracks. These creatures are not inclined to communication
with congeners: they meet together only at rich in forage sites, but even in
this case each tortoise keeps separately, not paying attention to congeners.
These reptiles eat various vegetation including plants inedible for other herbivores:
they willingly eat leaves and branches of poisonous plants of spurges family,
and also prickly sprouts of cactuses making a basis of flora of Galapagos Islands
in Neocene.
The courtship season at these reptiles lasts practically the year round: presence
of the female ready to pairing stimulates activity of males. Courtship ritual
at these tortoises does not differ in special complexity: males push each other
away from the chosen female, hiss and strike each other by heads, trying to
hit in unprotected place between neck and forward legs. The male had won in
duel shows itself to the female: he walks around of her on extended legs, from
time to time striking on edge of her carapace. The female at this time retracts
head and legs under carapace, showing submission to him.
Till the year female lays eggs up to three times. In clutch it may be about
20 eggs a little bit smaller than goose egg by size. Female lays them in secluded
place in bush, and does not come back any more to the nest. Incubating lasts
about two months. Young tortoises at once leave nest and hide from enemies
in thickets of cactuses. They grow slowly, and reach the maturity only at 30-th
year of life at carapace length of about one meter. Up to this time many of
them perish from enemies – rodents and birds. But all the same the population
of tortoises of this species at Galapagos Isles is numerous. Life expectancy
at this species is extraordinary long; it also may make over 200 years.
Collarbill
tortoise (Ramphotestudo dorsognatha)
Order: Turtles (Testudines)
Family: Tortoises (Testudinidae)
Habitat: tropical forests of Zinj Land.

Picture by Mex, colorization by Biolog
Initial picture by Mex |
Turtles are rather conservative group of reptiles: having evolved
at Earth in Triassic period, they actually did not change the anatomy features,
differing mainly by the habit of life. Protective tactics of these animals remained
practically identical from the moment of occurrence of these reptiles – at any
age they entirely relied on passive protection with the help of carapace.
Passive protection is not the best one – not receiving due repulse, predator
finds an opportunity to force a reptile by ruse to jut out from under the carapace,
and to kill it. Therefore various species of turtles used active defense for
protection: some of them became fast swimmers, and others began to bite. But
the most original way of active defense was developed by tortoises living in
tropical forests of Zinj Land. On the top part of the carapace above a head
at them the narrow strong wedge-shaped outgrowth of edge scutes, sticking up
forward, is developed. This outgrowth resembles a horn of the rhinoceros beetle
or a beak of some bird species. For this feature this tortoise is named the
collarbill tortoise.
Collarbill tortoise is the medium-sized reptile: length of carapace of adult
individual is about 30 cm, but usually it is much smaller. Its shape is typical
for turtles, but there are some essential differences. The head of this reptile
is very big and consequently it can not be retracted under the carapace completely.
But this feature does not prevent, and on the contrary, helps this tortoise
to defend. On the head of this tortoise the thick corneous scute with several
jags sticking upwards above nostrils and eyes was developed. Head and the outgrowth
at forward edge of carapace form the kind of “pincers”, like at males of Hercules
beetle (Dynastes): having pressed head to this outgrowth, the tortoise “bites”
predator, clamping its paw or tail, as if any alive trap. Strong cervical muscles
attaching to the top edge of carapace help this tortoise to compress “jaws”.
Force of the “bite” is so great, that even the adult tortoise can hang on aggressor
and do it till some minutes.
Despite of such protection, this tortoise nevertheless prefers to hide from
predators. It lives in underbrush, and leaves to feed in hottest time of day
when the activity mammal is reduced. This reptile eats mainly plants and mushrooms,
and especially willingly regales itself with fruit dropped from trees. Moreover
the collarbill tortoise eats eggs of birds nesting on the ground, and snails.
The carapace of the collarbill tortoise is colored contrastly: it is light yellow
with dark spots in the centre of each scute. Young tortoises have no such protective
adaptation; they are colored almost black color; also they are timid and hide
from enemies in bushes. “Beak” starts to grow at them at the approach of sexual
maturity – it happens approximately to the tenth year of life. In same time
on edges of scutes light corneous cover appears, and the tortoise becomes spotty.
The basic destination of this “beak” is to be the weapon in tournament fights
of males. In courtship season they become even more aggressive, than usually
are: reptiles creep in forest, trying to “bite” by outgrowths any moving object
suitable in the size. It happens they hook by head and crush lizards and smaller
tortoises of other species, and even try to attack birds. Having met each other,
males try to hook by head and to overturn the contender. Thus they puff throat
and hiss – it is a unique sound which they can utter. Having met the female,
male begins displays: it creeps around the female, pushing her by “beak”. During
the display male taps with a head on “beak” at the carapace from below. The
female has lesser “beak”, and she does not render resistance to the displaying
male.
Pairing occurs quickly, and male abandons the female. From six to seven weeks
after pairing female lays 6 – 8 eggs in simple nest as a small hole, dug out
by hinder legs in wood ground. Having laid eggs, she crawls across the nest
some times, and then masks it additionally. For this purpose the tortoise digs
out some small shallow holes (“false nests”) near the true nest, fills them
up with the ground, simulating the dug nest, and does not care of clutch any
more. The incubating lasts about three months; young tortoises at once leave
nest and hide in bushes.
At the western coast of Zinj Land the close species lives: it is “fanged”
tortoise (Ramphotestudo odontophora). At this tortoise above the head
deflexed edges of scutes stick up, forming something like toothed jaw. Its head
is wide and angular, and above eyes strong sharp spikes serving for “biting”
grow. This tortoise also differs in increased aggression.
Mole
tortoise (Subterraneotestudo talpina)
Order: Turtles (Testudines)
Family: Tortoises (Testudinidae)
Habitat: islands (“oases”) of Mediterranean swamps.

Picture by Alexander Smyslov
Live creatures select various strategies of survival at change
of environment conditions. They can move in searches of suitable to life conditions
or to change habit of life, adapting to new environment. Such choice had been
chosen by Mediterranean tortoise (Testudo graeca) when conditions of life in
the Mediterranean had started to change. The sea without the connection with
ocean because of formation of the Gibraltar isthmus gradually had dried up,
having transformed district favorable for life earlier to rigorous desert with
extreme conditions of environment. Rather favorable conditions had kept only
at tops of mountains sticking up among salt desert - at former islands of the
Mediterranean. Tortoises dwelt there, had been compelled to adapt to such conditions.
And the part of population of Mediterranean tortoise had acted easy and more
artfully: they simply began to avoid influence of extreme factors, having disappeared
under the ground and having turned to the special kind of digging tortoises.
It is too difficult to believe that this descendant of Mediterranean tortoise
is the close relative of huge Antheos
tortoise playing the same role at these small islands, that large ground
herbivorous mammal at continents. The mole tortoise almost constantly lives
under the ground, occasionally creeping out on a surface to get warm in beams
of the morning and evening sun. In the afternoon this reptile digs holes and
disappears in them from the sun drying up heat.
Armour of mole tortoise is extended and rather high; its length is no more than
30 cm. Scutes of carapace are smooth: it reduces resistance at movement in holes.
Main digging instruments of this creature is head with strong beak, and thick
sharp claws on forepaws. Eyes of this turtle are very small, and sight sense
is bad. During digging the ground the reptile blinks thick eyelids at all. The
firmest and dry ground easily yields to pressure of this creature: by beak the
tortoise loosens ground, and wide forepaws rakes it back. The back edge of carapace
is movably jointed with other carapace, and can fall down with the help of contraction
of special muscles. This adaptation, on the one hand, protects tortoise from
attacks behind, and on the other hand, helps to rake away and push the dug out
ground from hole. Hinder legs of mole tortoise are armed with strong claws and
become reliable support during ground digging.
This reptile eats mainly at the surface of the ground, leaving holes at night.
It eats greenery of treelike
salttrees and other plants meeting in “oases” of the Mediterranean, adding
to leaves ripe fruits and small invertebrates. At night these tortoises also
can ravage ground nests of birds and eat carrion. In the afternoon this reptile
does not go hungry too: it undermines roots of plants and eats them. Also it
eats tubers and bulbs of grassy plants.
During the rigorous Mediterranean spring when rains from Atlantic and Northern
Sea overcome mountain ridges, at mole tortoises the courtship season begins.
At night, when fog falls on the ground, these tortoises leave holes. Between
males there are duels accompanying with knock of carapaces and hissing of excited
“grooms”. The male tries to impress the female, accompanying the courtship with
impacts of claws of forward legs on her carapace. Pairing occurs at night on
the ground surface. The second wave of breeding takes place in an autumn but
then pairing occurs not so roughly. After pairing the female lays in deep hole
eggs in amount no more than 5 – 6 ones. For egg laying the female chooses a
hole in bottom layers of ground, digs out a small hole in its bottom, lays eggs
and buries them. After that female crawls out and does not care any more of
posterity. Such process repeats twice per year. At this tortoise the interesting
feature had developed: oosperms can develop with high degree of probability
further, and from such kind of eggs the normal posterity hatches. The sex of
posterity is determined by an ambient temperature, therefore parthenogenetic
populations at this tortoise do not appear. But in different “oases” there are
the subspecies distinguished in size and coloring of carapace.
The incubating of eggs lasts about two months. Young tortoises are completely
independent. In winter when the temperature is cooler, the posterity of autumn
laying can remain in nest till the next spring. Young animals live in tunnels
dug by adult tortoises, and eat the same food, as adult individuals. They become
sexual mature at ten years' age and can live over hundred years.
Cutting-beaked
tortoise (Agrionemys teratognathus)
Order: Tortoises (Testudines)
Family: Tortoises (Testudinidae)
Habitat: semidesert in Persian Ridge region.
Picture by Alexey Tatarinov
Human era had caused great damage to the representatives of
turtle group – the largest representatives of these reptiles died out or became
a rarity in the historical epoch. But in fact the negative influence began much
earlier: in Pliocene and Pleistocene the diversity of large tortoises decreased
at the species level at the background of development and proliferation of ground-dwelling
hominids: first australopithecines, and then humans.
At the end of the historic epoch tortoises in the wild had been present exclusively
as small and medium-sized forms, and the larger species remained in a small
number almost exclusively in captivity and in conditions of strict protection.
After the end of human epoch and the ice age the tortoises started the return
to their lost positions - among them many large species appeared, and they evolved
independently in various places of the Earth.
In the semi-deserts appeared in Persian Ridge area the large species of tortoises
appeared – it is a descendant of Horsfield’s tortoise (Agrionemys horsfieldii).
Its peculiar feature is very large head, which does not fit under carapace and
has its own protective corneous shield. A significant part of the head is made
of huge jaws with sharp edges, hence the name of this tortoise.
This is a fairly large species of terrestrial reptiles: the length of an adult
one’s carapace reaches 70-80 cm and it weighs of up to 60-70 kg. Females are
larger than males, but males have larger head, and the schield on it is covered
with lots of bumps on edges and is used as a weapon.
Cutting-beaked tortoises have a certain food specialization, allowing them to
avoid competition to closely related species and to use a very accessible source
of food. These reptiles feed on large hard grasses and on branches of shrubs.
Due to the low sensitivity to alkaloids they easily eat large bulbs of stony
onion (Petroallium petrops). The opened jaws of cutting-beaked tortoise
really look like garden shears – the cutting edge of lower jaw is curved up
and it looks heavier than the upper one. Edges of horny beak are self-sharpening
and do not get blunt when the tortoise eats hard food. Sometimes these reptiles
eat food of animal origin: small vertebrates and carrion. To replenish the calcium
stock in its body cutting-beaked tortoise gnaws dry bones. If there is water
nearby, these tortoises drink it willingly and often.
To protect themselves from overheating these reptiles have very light colour
of shells: scutes are grey with darker centre. The reptile’s dry and wrinkled
skin is greyish-white. Besides these tortoises live in places where the ground
vegetation – tall herbaceous plants and shrubs – is present.
The life of these reptiles is exposed to the seasonal rhythm. These animals
spend the winter hibernating in burrows they dig with paws and beak. In early
spring these tortoises emerge from wintering shelters and in 10-12 days they
are already ready to breeding. During the mating time males are very active
– they quickly pursue each other and bite the legs and edges of the carapace.
At the high number of tortoises on their “display grounds” the continuous sounds
of striking shells (the males encourage females to copulation with their shell
strikes) and the hoarse rumbling of males are heard. In May, at the beginning
of the dry season, the females lay 2-5 eggs 5 cm long in underground nest, making
three nests in a short time. In October the tortoise eggs hatch and the hatchlings
stay under ground for wintering, leaving for the surface only at the next year
spring. Although they grow up a bit during the winter due to the remnants of
egg yolk, their shells are still soft and therefore they are very vulnerable.
At the age of 25 years young reptiles reach the average adult size and maturity.
Their life expectancy is great: some individuals live up to 125 years.
The idea of the existence of this species of reptiles is
proposed by Nick, the forum member.
Translated by Bhut
Ugunqu
(Ugunqu kubantwana)
Order: Turtles (Testudines)
Family: Softshell turtles (Trionychidae)
Habitat: freshwater reservoirs of tropical Africa, the Saharan Nile and rivers
south of the Sahara.
Turtles are among the oldest reptiles of the Earth that survived to the age
of man – they appeared at the very beginning of the Triassic, but their separation
occured already in the late Paleozoic. They distinguish significantly from the
rest of the reptile orders anatomically and behaviorally. Endurance and the
ability to survive adverse conditions helped them survive the end of the Holocene
– the era of anthropogenic pressure, as well as the beginning of the Neocene
with its climatic cataclysms. In the time of Neocene warming, a spate of active
speciation began; they gave rise to many new species and occupied new ecological
niches. The large African crocoturtle
is one of the turtles of the new Neocene formation, and a related ugunqu species
lives next to it.
Ugunqu is a fairly large turtle; adults grow up to 4 m in length. Although it
is related to the crocoturtle, this reptile is not so predatory, and eats a
significant amount of plant food – as a rule, underwater and near-water plants.
Ugunqu has a shorter neck than a crocoturtle, and in general its proportions
are more typical for turtles: ugunqu prefers not to swim, but to walk on the
bottom of reservoirs. Like a crocoturtle, it lacks corneous shell cover, but
a fatty hump appeared on its back under the skin, due to which ugunqu survives
during the drought (this is especially important in the north of its range),
when it has nothing to eat. The skin is gray with a greenish tinge, often overgrown
with microalgae, folded, mobile and stretchable on the neck and legs, thick
and dense on the shell. The reptile has powerful legs and claws, and it easily
digs holes at the beginning of a drought, and waits out an unfavorable period
in them.
Ugunqu eats much less amount of animal food than a crocturtle, and it usually
represents not live animals, but carrion. Ugunqu has very powerful jaws, and
it is able to break and swallow any solid food – hard plants, bones and cartilage
of a dead mammal or bird. It is hunted by various predators, but mainly small
young turtles fall their prey: if ugunqu grows to at least two meters long,
it is no longer afraid of anyone, and can even drive the predator away from
its legitimate prey, using its size and weight for this. It often feeds on the
trophies of the crocoturtle, sometimes joining the predator’s feast and not
waiting for it to get enough and leave the prey.
The mating season at ugunqu begins at the beginning of the rainseason: males
of this species push each other with their bodies, trying to overturn or push
each other away from the chosen area on the river bank. Here the victory does
not always depend on the size – sometimes a smaller male, having taken a comfortable
position, can defeat a larger one, and sometimes the female, while two males
are establishing their hierarchy, leaves their territories and appears in the
possession of a third male, but the conflicting rivals do not even pay attention
to it in the rage of battle. Like all other turtles, ugunqus have no families
and no more or less permanent pairs form: females lay their clutches in the
ground near rivers and lakes where they live, and no longer take care of them.
There are about 200 medium-sized rounded eggs in the clutch. The clutch is not
guarded, and a significant number of eggs die from predators. Usually, after
one and a half to two months, the juvenile turtles hatch from the eggs and begin
an independent life, like adults. They stay in shallow areas of the riverbed,
where they can feel like in safety from an accidental attack of an adult one.
Sexual maturity comes at the age of about 20 years, with a body length of 1.75-2
meters. If ugunqu does not die in the first years of life, then it will be able
to live 200-300 years, or even more.
This reptile species was discovered by Bhut, the forum member.
Honu-i’no
(Honuino rapax)
Order: Turtles (Testudines)
Family: Softshell turtles (Trionychidae)
Habitat: Hawaiian Archipelago, Hawaii island and neighboring islands, rivers
on the lowland part of the islands.
The human epoch is characterized by the enormous scale of settling of residents
of various floral and faunal realms to territories previously not peculiar to
them, where they could not get naturally. Not all species settled by humans
have become extinct in new habitats – many of them have settled down in their
new homeland, and in due course of evolution have given rise to new forms. Honu-i’no
is a large carnivorous descendant of the Chinese softshell turtle (Pelodiscus
sinensis), once introduced by people to Hawaii (hence the name meaning “evil
turtle” in Hawaiian). It became very large and occupied a special ecological
niche, avoiding competition with its neighbors, which remained small, although
descended from the same ancestor. This reptile has got a convergent resemblance
to the crocoturtle – another
giant softshell turtle inhabiting North Africa, because it occupies a similar
ecological niche of the top predator of freshwater reservoirs.
The length of the shell of honu-i’no reaches 2 m, and the neck and head make
additional 1.5 meters to it. The body is wide and oval in shape. The front legs
are quite long and mobile, with a large first claw on hand. The hind legs are
shorter, but also well developed, with webbing between the toes. The neck is
long and thick enough; the head is large, the snout is shortened compared to
other softshell turtles, but the jaws are wide and strong. The back and almost
the entire body are greenish or gray in color, but the belly is yellow with
large black spots.
This unusual reptile lives in small Hawaiian rivers flowing in the undergrowth
in mountain valleys on relatively flat terrain. It usually stays in shallow
water, basking in the sun or waiting for prey. The main prey of adults includes
mammals and large birds (the attacking honu-i’no is able to drag even an adult
kuahana under water), but a significant part of
the diet consists of aquatic animals – fish, smaller turtles, including juvenile
bat turtles, and waterfowl. Cannibalism
is typical of this species. To hunt large ground-dwelling prey, the turtle buries
itself in river debris at the bottom near the river bank and waits for the prey
to be nearby. When it happens, honu-i’no stretches its neck, grabs the prey
with a death grip and drags it under the water, where holds it until it chokes,
and then eats it, tearing off pieces of meat with its jaws. The remains of prey
can be eaten by shorebirds.
This turtle species has soft skin, so it is affected by various skin parasites,
from which birds and small fish relieve honu-i’no. To clean the skin, honu-i’no
lies down in shallow water, sticking its head out of the water, and stays motionless.
The adult turtle has no other enemies, but the juveniles can be threatened by
fishes, small turtles of other species and adult relatives, as well as some
local lizards, some birds and mammals.
Due to the ability to resist salt oceanic water for a short time, honu-i’nos
can settle across the archipelago. There is always a rare but regular exchange
of individuals between populations of different islands.
There is no clearly defined breeding season for this species. The female lays
eggs on the shore, buries them in the ground and stays near the nest for about
two months – a parental care unusual for turtles. At this time, she occasionally
leaves and goes to feed. When the juveniles begin to hatch, the turtle helps
them out by digging up the ground layer above the nest with its head and front
paws. Crawling through the thickets near the nest, it scares away small animals
that can damage the offspring. When the young turtles crawl into the water,
the female leaves the offspring. Little honu-i’nos live in quiet forest rivers
and at first feed on small fish, frogs and invertebrates (aquatic insects and
their larvae, crustaceans, snails), gradually switching to adult food with age.
Sexual maturity occurs at the age of about 20 years. Life expectancy is very
long – up to 150 years and more.
This reptile species was discovered by Mamont, the forum member.
Hawaiian
spotty softshell turtle (Pelodiscus hawaiiensis)
Order: Turtles (Testudines)
Family: Softshell turtles (Trionychidae)
Habitat: Hawaiian archipelago, Hawaii island, forest rivers.
In the human era, Chinese softshell turtle (Pelodiscus sinensis) was introduced
to the Hawaii. It managed to survive there in the era of the global ecological
crisis, actively evolved, and its descendants occupied several different ecological
niches in the Hawaiian archipelago of the Neocene epoch. In addition to the
giant honu-i’no, it gave rise to a number of
smaller species that do not differ in size from the ancestor. One of these species
is the Hawaiian spotty softshell turtle.
It is a small turtle whose shell length does not exceed 20 cm. The flat soft
shell is of oval shape. The legs are quite short, but wide, with long digits
and large claws, adapted for swimming and digging. The tail is quite long, protruding
from under the shell edge. The neck is long and thick enough, the muzzle is
pointed, with a long movable nose. From above, the shell is willow green with
numerous small black spots; the upper part of the head, paws and the tail are
of the same color. The plastron is yellow with several pairs of large black
spots. The underside of the legs is light yellow and lacks spots. Sometimes
there are individuals of dark brown, almost black color. In such dark individuals,
the black spots on the underside of the body merge with each other, and only
the edges of the plastron have a dark yellow border.
Spotty softshell turtle lives in quiet forest rivers, slowly flowing and littered
with tree trunks. This turtle is not territorial, sometimes several reptiles
of this species can even bask in a large group on a fallen tree trunk. But most
often the Hawaiian spotty softshell turtle lurks on the river bottom (usually
near the shore) and waits for the approach of possible prey. Attacking it, the
reptile thrusts its head forward on a long neck and catches its prey with a
death grip. The diet of this species is diverse – crustaceans, insects, numerous
fish and other small vertebrates. The shells of arthropods are crushed by the
jaws of a turtle, terrestrial vertebrates are usually drowned and held under
water. These turtles masterfully deal with large sawleg
grasshoppers – it
grabs the insect by the abdomen and drags it into the water, where it suffocates,
and then tears it apart, leaving only its legs and wings. The enemies of these
turtles are honu-i’nos, as well as some birds and
mammals.
This species has no specific breeding season. The female lays eggs near the
shore, buries them in the ground and crawls away. Fertility is up to 10 round
eggs with a leathery shell. After 4 months, small turtles hatch, quickly crawl
into the water and for the first time feed on crustaceans, aquatic insects and
small fish. At this time, they have a lot of enemies, and less than a quarter
of the total number of the offspring survive until the end of the first year
of life.
Sexual maturity comes at the age of 5 years, life expectancy is over 50 years.
This reptile species was discovered by Mamont, the forum member.
Mossy
langawarin (Langawarin orectoloboides)
Order: Turtles (Testudines)
Family: Snake-necked turtles (Chelidae)
Habitat: Meganesia, freshwater bodies of tropical and subtropical zones.
The merging of Australia and New Guinea into a single continent Meganesia, as
well as the shift of the continent to the north favored the evolution of freshwater
fauna. Among the inhabitants of rivers, lakes and swamps of the continent, snake-necked
turtles are very characteristic, making a worthy competition for the descendants
of the red-eared slider introduced here in the human era. In due course of evolution,
they have developed a variety of strategies to avoid competition and occupy
various ecological niches in accessible habitats. One of the most peculiar strategies
was developed by a turtle from shallow reservoirs with a slow current – mossy
langawarin (“langawarin” is the name of the snake-necked turtle among the aborigines
of northern Australia). This representative of snake-necked turtles has leathery
fringed outgrowths on the edges of the body and neck, which gives it an excellent
disguise and helps to get food.
Mossy langawarin is a large turtle: the length of the shell is about 50 cm;
the length of the neck is up to 70 cm. The reptile has a short flattened head,
a movable neck, a rounded shell and a very short thick tail. Along the edges
of the neck and limbs of this reptile, brush-like skin outgrowths develop, which
simultaneously serve as additional organs of gas exchange. The surface of these
outgrowths is covered with numerous villi, increasing the total area of gas
exchange. Capillaries penetrate the villi, supplying the reptile with additional
oxygen from the water; due to it, the turtle can hold its breath for 2-3 hours.
There are four rows of outgrowths on the neck – large brush-like ones on the
edges of the neck, and two parallel rows of small tufts on the upper surface
of the neck. Each limb is bordered by one row of fan-shaped skin outgrowths.
The reptile’s head is flat; small brown eyes are turned up. The mouth can open
wide; the edges of the jaws are cutting.
The color of the upper side of the body is faint – brown with greenish spots.
The outgrowths are colored dark green. This color, together with a layer of
algae film on the shell, provides langawarin with an excellent disguise. The
lower part of the body is grayish-white, but at rest it is completely invisible.
This reptile species is a benthic ambush predator living in shallow waters at
a depth of no more than one meter. Langawarin catches its prey by thrusting
its head forward and sucking prey into its jaws with a strong current of water
rushing into the animal’s throat. This species feeds on fish, crayfish and frogs.
If possible, langawarin can grab the chicks of waterfowl from the water surface.
Like all turtles, this species does not care about offspring and does not form
permanent pairs. The female is larger than the male, which, in turn, has more
developed claws on its front paws. The primitive mating ritual is limited to
the male’s attempts to keep on the shell of a resisting female. The nest is
arranged on the shore; a clutch numbers up to 15 eggs. After egg laying is completed,
the female masks the nest with plant debris. Incubation lasts about 120 days.
Egg laying is repeated 2 times a year.
Young animals reach sexual maturity at the age of 8 years. Life expectancy is
60 years or more.
Giant
snake-necked turtle (Chelodina giganthea)
Order: Turtles (Testudines)
Family: Snake-necked turtles (Chelidae)
Habitat: freshwater reservoirs of northern and eastern Meganesia.

Picture by Alexey Tatarinov
Due to the arid climate, the reservoirs of Australia were extremely
rare, and their wildlife was less diverse than in other tropical areas. The
situation is changing dramatically in the Neocene – due to the shift of the
continent to the equator, the total amount of precipitation increases, and two
large lakes of marine
origin form in the north. Against the background of the ongoing changes, the
biodiversity of reservoirs is also increasing, new ecological niches are emerging.
One of them was occupied by a giant snake-necked turtle. Unlike its African
counterpart, it is not a top predator and preys mainly on small fish.
The giant snake-necked turtle is one of the largest turtles in Meganesia and
is second in size only to Brontochelys. Despite some changes in lifestyle, the
giant turtle is similar to other members of its genus. The carapace of an adult
reptile can be up to 1.5 meters long, and combined with a long neck, the total
length of this turtle reaches 2.5 meters. The carapace is smooth, narrowed in
front, of dark brown color. The plastron and the lower part of the turtle’s
body are pale yellow or white. The scales can be of various colors – from dark
purple and brown to light gray. The tail is so short that it is almost invisible
under the rear edge of the carapace. A long and very mobile neck smoothly turns
into a small head, at the tip of which the nostrils and eyes are located. The
paws have well-developed webbing and claws.
The giant snake-necked turtle lives in stagnant and slow-flowing freshwater
reservoirs, mainly on shallows or near the shore. It chooses places no more
than one meter deep, so that it can raise its head at any moment and take air
into its lungs. The turtle leads a sedentary lifestyle and almost does not move,
being in constant waiting for a prey. This species differs significantly from
its relatives in hunting tactics – instead of actively searching for prey, it
lays motionless with its head hidden and waits for careless fish. When the prey
is close enough, the turtle abruptly darts its head and snaps it. The reptile’s
diet also includes large invertebrates, waterfowl, frogs and other animals of
a suitable size. In turn, large crocodiles pose a danger to an adult turtle,
and young individuals die in large numbers from large fish and birds.
The mating season begins in mid-spring; at this time, males become active and
move in search of a mate. The mating ritual is very simple and includes “neck
wrestling”, when the male, clinging to the female’s carapace, pulls her head
down by pressing his neck on her neck. After mating, the animals leave each
other. Pregnancy lasts about two months, after which the females go ashore.
For laying eggs, they choose warm and not flooded places near the water. There
can be from 15 to 30 eggs in a clutch, but most of them are eaten by predators,
since the nests are not guarded. Depending on the external temperatures, the
incubation period lasts from 120 to 180 days.
Giant snake-necked turtles live up to 90 years, become sexually mature at the
age of 9 years.
This reptile species was discovered by Gleb_97, the forum member.
Wollunqua
(Chelodina wollunqua)
Order: Turtles (Testudines)
Family: Snake-necked turtles (Chelidae)
Habitat: northern Meganesia, including Arafura and Carpentaria lakes.
Snake-necked turtles, like other members of the order, suffered in human epoch
from habitat destruction and excessive hunting. The second impact to them was
the aridization of the climate of Australia during the ice age. However, some
of them survived to more favorable times, giving rise to a number of new species.
One of them is wollunqua, a descendant of the New Guinea snake-necked turtle
(Chelodina novaeguineae).
This species is a relatively large reptile: the length of the carapace is up
to 70 cm, the neck and head are about 60 cm long. The body shape of this species
is typical for this group of reptiles: an oval carapace of a streamlined shape,
powerful paws with long claws, a short tail and a neck that folds sideways under
the carapace. The reptile’s head resembles a cartoon boa constrictor’s head
with nostrils shifted to the tip of the muzzle and equipped with leathery valves.
The body color is not bright – from yellow-olive to brown with various transitional
variants, depending on the population.
This species is a predator: the food includes mainly fish and amphibians, less
often reptiles and arthropods that have fallen into the water. During the hunt,
this turtle lies on the bottom, hiding its body in the silt and among underwater
plants. Any fish or amphibian of suitable size that carelessly swims up to the
ambush site is captured and eaten. When the reptile does not hunt, most often
only the head and neck are visible from the side, resembling a snake sticking
out of the thickets (hence the name: wollunqua – the snake-like rain spirit
of the Australian aborigines). The wollunqua spends the night in a hole dug
on the shore, the exit of which is under water. In this reptile, the mucous
membrane of the mouth and pharynx forms numerous villi that promote absorbing
oxygen directly from the water. Due to this feature, the turtle can stay under
water for up to 3 hours.
The mating season in the southern part of the range begins about 4 months before
the rainy season, in the northern part of the range the seasonality in reproduction
is not pronounced. During courtship, the male and female wrap their necks around
each other, touching their sides at the same time, and the male clings to the
front edge of the female’s carapace with his claws. If the female does not accept
the courtship of the male, she breaks out and swims away. Eggs are laid by females
in the same place where they hatched – in a heated place near a non-drying reservoir.
The same nesting sites have been used for many generations, and a lot of eggshells
and bones of dead juveniles accumulate in the ground in such places. The wollunqua
eggs are oval, 39-42 mm long and 29-30 mm wide; there are up to 30 eggs in a
clutch. After 100-110 days, shortly before the rains begin, young turtles hatch.
In the northern part of the range, young turtles of various ages are found in
the reservoirs. In the first year of life, during the settling, about 60% of
the hatched wollunquas die. They reach puberty at the age of 9 years with a
carapace length of 50 cm . Life expectancy can reach 120 years.
This reptile species was discovered by Nick, the forum member.
Elasmosaurine
hydromedusa (Herpetomedusa elasmosauroides)
Order: Turtles (Testudines)
Family: Snake-necked turtles (Chelidae)
Habitat: rivers of tropical zone of South America.

Picture by Carlos Pizcueta (Electreel)
Sometimes successful types of anatomy met in any group of animals,
can be successfully shown in another one, though in another form. This situation
had taken place at Neocenic water turtles of South America: among them one absolutely
unusually looking creature had appeared more similar to plesiosaurs for a long
time missed from face of Earth. Turtles are rather conservative by their anatomy
group of reptiles, but features of their structure have permitted developing
of such original design.
The inhabitant of great rivers of South America, the elasmosaurine hydromedusa,
is externally very similar to any dwarfish plesiosaurus: the neck length one
and half time surpasses length of carapace. It precisely repeats principle of
constitution of this sea reptile: long flexible neck permits to seize dexterously
the most agile catch under water, and the massive body serves as reliable point
of support, allowing making throw. In cervical of this turtle the number of
vertebrae is increased (up to 30 vertebrae), and joints between them are improved;
due to it neck has got significant flexibility and mobility. But such structure
has the return side: head of this turtle can not retract under carapace. But
this reptile lives far from coast, in thickness of water, and large predators
of South-American rivers, able to harm it, prefer coastal habitats.
Elasmosaurine hydromedusa is adapted to pelagic habit of life: it is tireless
swimmer seldom getting out on riverbank. Extremities of this turtle are oar-like,
wide and long, and lengthened fingers form similarity of flippers. At males
on forepaw two long thick claws are advanced, and on hips of hinder legs there
are long corneous heels. The reptile moves basically due to simultaneous flaps
of forepaws, hinder legs during swimming serve as rudders. To improve this animal
covered with carapace, keeping in water easily and do not sink, under carapace
thick layer of fat serving as float is accumulated. Additional buoyancy to turtle
is given with the porous bones impregnated with fat, and voluminous lungs. Horn
plates of carapace at this turtle in youth are as thick and strong, as at other
turtles of comparable size, but at the adult reptile they are appreciably thinner,
than at other turtles. Length of carapace is about 1.7 m, weight of adult animal
is up to 250 kgs. Tail at this turtle is short, serving basically for storage
of fat stock.
Scutes of carapace of the elasmosaurine hydromedusa are smooth and skintight
to each other. On medial scutes of carapace at turtles there are small vertical
keels, at males a little more sticking up, than at females.
The top part of carapace at the elasmosaurine hydromedusa is colored light brown
with darker spots in the centre of each scute. Abdominal shell (plastron) is
white with contrast black spots. Neck and extremities at the adult turtle are
colored gray-blue with dim white longitudinal strips, at young individuals –
dark-brown with grey-green strips. At males an iris of the eye is red, at females
it is pale yellow.
Head of the elasmosaurine hydromedusa is wide with strong jaws. Males are more
“large-headed”, than females. Edges of jaws are sharp, cutting, and tips of
jaws are bent like beak. This turtle eats fishes and other water animals. Young
turtles of this species search for water insects, river shrimps and crabs. They
live in coastal zone (it is the reason of their darker colouring in comparison
with adult reptiles) and do not compete to larger representatives of the species.
Growing up, they start to hunt fish in thickness of water and to develop deep-water
sites of the river. Adult turtles live far from coast and eat mainly fishes.
They also can catch water birds (seizing them from below from under water) and
eat corpses of sunken animals. The elasmosaurine hydromedusa almost does not
creep to land, only sometimes it has a rest, having clung by paw to trees floating
in river or fallen in water.
These turtles are solitary animals meeting together only in pairing season.
Because in river it is difficult to mark any conditional borders between sites,
turtles simply try to avoid company of each other. Sometimes some turtles surround
one large shoal of fishes, but during hunting they operate not together, differing,
for example, from dolphins or pelicans.
Hunting turtle tries to catch up planned fish and to push it to water surface.
It makes sharp lunges by head, biting fish. Sharp jaws of turtle can literally
shear across smaller catch, and to larger animal it puts deep wounds and catches
prey weakened from loss of blood. If the preyed fish is too great, turtle simply
bites off from it pieces and swallows them. Usually it eats catch while it keeps
on water, and does not dive for sunken one. The full turtle has a rest and is
basked at the sun, floating on surface of water and occasionally flapping flippers
to keep on current.
The pairing season is time when turtles pay attention to the neighbours though
any time. Elasmosaurine hydromedusas breed the most part of year, making a break
only in dry season. In northern part of area seasonal prevalence in breeding
of this turtle is not present. The female ready to pairing emits in water musk
liquid, involving males by smelling trace. Sometimes the chain of several males
follows such female showing the keenest interest to it. At turtles constant
pairs do not form, and female basically is indifferent, what male will be father
of its youngs. Males moving by breeding instinct, push away each other from
the female, and try to hook by claws for edge of its carapace.
Males never leave water, except for cases when the river or lake where they
live, become shallow and dries up. And females annually should make hard way
to land to lay eggs. They hardly move overland, dragging massive body forward
by flippers, and pushing by back ones. On land the elasmosaurine hydromedusa
is very sluggish and clumsy.
However, the female occupied with the nest making, is not defenceless, similarly
to sea turtles of Holocene epoch: powerful jaws and long neck allow it to keep
nest ravaging lovers at respectful distance. If any predator especially desiring
to eggs (for example, the otterodent,
large local river rodent) will approach too close, turtle warns of the intention
to protect, opening wide mouth and loudly hissing. If threats of the reptile
are not taken seriously, it can seize by sharp attack the uninvited visitor
and then it is possible to get rid of its stranglehold, perhaps, only having
torn off turtle’s head or having left in its jaws a fair piece of own skin.
And due to flexibility of the neck turtle can keep all-round defense, simultaneously
digging nest for the posterity.
In one clutch it can be up to 20 - 25 eggs. Their incubating lasts more than
three months, in colder southern areas delaying up to five months. Young turtles
with length of carapace about 5 cm live in shallow wood streams, eating small
fishes and shrimps. Dark colouring helps them to mask at the bottom among fallen
leaves. For improvement of masking at young turtles on sides of neck and legs
skinny scalloped outgrowths develop. Young turtles catch prey by sharp throw
of head, not leaving shelter.
At growing up reptiles behavior and shape change: colouring brightens, and young
turtle starts to catch fishes, chasing them in thickness of water more often,
and, at last, at the age of six years completely turns to pelagic predator.
The ten years' turtle becomes able to breed, and the age limit of this species
can pass for 130-150 years.
Robust
chelonicerops (Chelonicerops robustus)
Order: Turtles (Testudines)
Family: Podocnemididae (Podocnemididae)
Habitat: Atlantic Ocean, New Tortuga island.
Islands formed before the human epoch and preserved in the Neocene display the
effects of human influence on their nature – the flora and fauna of such islands
is often represented almost exclusively by descendants of alien species. At
the same time, the nature of the younger islands bears all the features characteristic
of the island fauna and flora. Among these islands there is New
Tortuga, an island in the tropical Atlantic. Most of the terrestrial
inhabitants of the island are represented by descendants of South American podocnemidid
turtles. They are very diverse – several herbivorous forms live here, and even
tree-climbing and predatory species. Perhaps only
the lack of competition with other vertebrates allowed most of them to arise.
Among the turtles of the island even a horned species – robust chelonicerops
– exists.
The body length of this reptile is up to 1.1 m, weight is up to 150 kg. This
turtle has a thick saddle-shaped carapace with an extension over its head looking
like a kind of a “collar”. The legs and the carapace are protected by large
shields forming numerous blunt spikes. A conical horn 15 cm long grows on the
head, pointing upwards, and there are several smaller outgrowths behind the
eyes. The reptile’s head is large, equipped with strong cutting jaws.
Chelonicerops feeds on plant food – branches of shrubs and low trees, large
grasses. It is a solitary animal, leading a diurnal lifestyle. The only thing
the adult chelonicerops fears is cheloniraptor,
a local predator. Despite the hard shields, the predatory turtle is able to
injure this one. However, the chelonicerops is able to defend itself – it launches
sharp blows with its horned head and spiked shell, keeping the predator at a
distance. Therefore, unlike gravichelonias, in
this species young individuals and eggs die more often. When an adult reptile
outgrows a predator in size, it steeply increases the chances of winning a fight
against it.
The mating ritual of this species is primitive: males fight for a female, hitting
each other with their heads and clinging with their horns. The reproduction
rate is very slow – the female makes only two clutches of twenty eggs the size
of a chicken one per year. Incubation lasts up to three months. Young turtles
reach sexual maturity at the age of 17-19 years; a significant part of the offspring
die from predation by cheloniraptor and birds. The life expectancy of these
reptiles reaches 170-200 years.
This reptile species was discovered by Nick, the forum member.
Atlantic
bat turtle (Pteromedusa vespertilionina)
Order: Tortoises (Testudinata)
Family: False scuteless turtles (Neoathecae)
Habitat: tropical latitudes of Atlantic Ocean, Caribbean Sea, Pacific coast
of Central America.

Picture by Alexander Smyslov
In Neocene reptiles have made one more resolute attempt to
win a sea inhabitancy. Warming of climate in Neocene epoch, and also the mass
extinction of cetaceans, pinnipeds and many other sea inhabitants had permitted
them to develop pelagic habit of life. In tropical latitudes of Pacific Ocean
the huge sharkodile,
one of last species of crocodiles reigns. Except for it, various turtles live
in sea. They are not descendants of sea turtles of Holocene epoch. Some species
of sea turtles of Neocene descend even from tortoises. The algal
turtle from Tanganyika Passage separating Africa and Zinj Land is those,
for example. But this reptile does not leave far from the coast. But other turtles
have developed true pelagic habit of life. They meet at open ocean far from
coast, and need the ground only for egg laying.
Water literally boils under impacts of mighty flippers, when one of such turtles
emerges to the ocean surface to make some breaths. It represents an impressive
show: the body is up to five – six meters in length at width over two meters.
And the span of huge forward flippers even exceeds the general length of the
reptile. They are very similar to wings of huge bat, hence the name of the turtle
– the bat turtle. Some loud breaths follow, and the giant plunges in water again.
Having emerged to the surface and having recovered breath properly, this turtle
may not emerge till two hours. It even sleeps hanging in thickness of water,
and only slightly moving by flippers.
The body of the bat turtle keeps the flattened streamline form characteristic
for turtles, but has no main feature of turtles – firm armour. Instead of corneous
scutes it is covered with thick rough skin, as at the leatherback turtle (Dermochelys)
extinct to Neocene. But bat turtles are not lineal descendants of these animals
– they descend from one species of freshwater turtles of South America. The
heavy armour is not necessary for this reptile - it prevents to swim, and speed
protects it from enemies, but not only it. The principal organ of movement of
this turtle is the pair of long forward extremities similar to wings. Flapping
them up and down, the turtle can swim accelerating for a long time the speed
more than 30 kms per hour, and at the short distance it even may accelerate
up to 60 kms per hour. Shoulder and forearm at this reptile are rather short,
but hand and fingers are very long: they amount more than half of length of
forward extremity. Bones of fingers are wide and flattened, connected by elastic
skin membrane, and the short thumb is not connected by membrane and has some
mobility. With its help the male keeps the female at pairing, and the female
clings to ground creeping out to the land for egg laying. The back edge of forward
extremity is joined to side of turtle with the help of rag of mobile and extensible
skin. This feature makes the bat turtle very similar to gigantic bat swimming
under water. Back extremities of the reptile are modified to short and wide
flippers serving for turns and maintenance of balance. Their length hardly probable
makes third of length of forward flippers.
The body of this reptile is colored softly dark grey color with small amount
of dim dark spots on back. The bottom part of body at these turtles is light
grey. Such colouring helps the animal to mask in thickness of water. But masking
and speed not always can help to escape from predators, especially to young
turtles. Therefore this species has one more weapon very effective against underwater
predators, from large squids to the sharkodile. Already at early age at young
turtles in cloacal bladders glands secreting very poisonous paralytic substance
develop. It represents transparent yellowish liquid; escaping from predator,
the turtle emits it out in water. When the predator swims through the cloud
of such substance, receptors in its mouth and olfactory bodies appear paralyzed
till some hours. Besides these turtles are able to protect themselves actively,
biting the enemy by strong jaws. The adult turtle of this species having reached
the maximal size does not have enemies.
At bat turtle there is wide flattened head with short, narrow and high beak.
They eat various food which may be found in sea. Atlantic bat turtle differs
in bias to vegetarianism. It often meets by big groups at algal fields near
New Azora Island, and near thickets of sea salad
in Caribbean Sea. Young turtles of this species keep near to coast and eat macroalgae
growing at the bottom, and adult individuals pass to pelagic life. Only females
once a year come back to land to lay eggs.
Pairing at these turtles takes place at open ocean. It is preceded by primitive
courtship displays. At this time males chase the female and push each other
away from her by body. During courtship displays the skin on head of the male
becomes bright: along the side of head through an eye black strip stretches,
and the throat turns bright blue color. The iris of the eye at the male is white,
and it is clearly distinguished on the background of head. The winner male tries
to keep the female, seizing her by beak for edge of the carapace. When he succeeds
to do it, he creeps on the female and couples with it. Pairing lasts till some
hours.
After the pairing in organism of the female eggs ripen within approximately
5 months. They are too small for the multiton giant, but all the same they have
the impressive size: length is up to 20 cm at the width about 10 cm. The clutch
of this reptile may number up to 100 – 120 eggs. All turtles of this species
breed in the same place – in South America, at the land area between Amazon
and Hyppolite rivers. They swim up-stream in these rivers to tens kilometers
far from the coast, and lay eggs at sandy riverbanks. The nest of this turtle
is a hole about one and half meters deep. The female digs eggs in sand and leaves
them to the mercy of fate.
If 3 months of incubating pass successfully, young little turtle with the length
of carapace about 15 cm hatches from egg. The hatching of young turtles passes
almost simultaneously, but each young turtle should independently dig the way
through the thickness of sand for itself. Such work lasts up to three or four
hours. Usually it starts in the afternoon when the sun warms up sand well, and
is finished up to the night. Young turtles appear in water simultaneously, form
schools and swim downstream to the sea. The mass downstream migration of young
bat turtles takes place at night within approximately one week, but separate
singles swim downstream till about next two weeks. The considerable part of
them becomes prey of fishes and other inhabitants of the river whereas schools
of young turtles suffer less damage from predators.
Young bat turtles grow rather quickly: till the first years of life they increase
2 – 3 cm in length; farther up to 60-years age they grow up to 5 cm per one
year. At this time they reach maturity, but further their growth is slowed down.
But they reach the really the monstrous size due to the large life expectancy:
the bat turtle may live over 250 years.
Other species of this group live in oceans:
Pacific
bat turtle (Pteromedusa velonectes) differs from the Atlantic congener
in smaller size: the length of the adult reptile exceeds 5 meters seldom, and
usually exceeds 4 meters only a little. At this reptile colouring of body is
gray-blue without dark spots. Sometimes the reptile gets an appreciable greenish
shade because of symbiotic green algae settling on its skin. Forward flippers
of this turtle are narrower, and back ones are more lengthened, rather than
at the Atlantic species. This turtle swims faster, than the Atlantic species.
It also differs in greater aggression, and frequently attacks the enemy: some
sharkodiles have deep scars on a body, and sometimes the tip of the flipper
at them appears bitten off – these are traces of counterattacks of turtles of
this species. Pacific bat turtle eats mainly pelagic invertebrates – jellyfishes
and gardenersalpas. It breeds
at Hawaii, swims upstream in rivers and lays eggs in soft forest ground.

Picture by Alexander Smyslov
Dwarf bat turtle (Micropteromedusa australis) lives in the Southern hemisphere. This species, on contrary to the relatives, do not keep away from coast. It lives along the coast of Meganesia and New Zealand, and the separate population inhabits Eyre Gulf, not having contact to ocean populations of this species at all. The length of body of dwarf bat turtle does not exceed two meters. It also differs in more advanced claws on forward flippers and massive head, and its beak is not cutting, but crushing. This reptile is zoophagous – it eats molluscs, crabs and dead fishes. Populations of open ocean breed at small islands near the coast of New Zealand; for this purpose individuals from the coast of Meganesia gather to schools and cross Tasman Sea. The population from Eyre Gulf breeds at northern coast of Tasmania. For the breeding females of this population had to cross sandy shallows in the mouth of gulf till the inflow.
The idea about the existence of this group of animals was stated by Tim Morris, Adelaide, Australia.
Leaf-looking
gecko (Phyllogekko platysomus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckoes (Gekkonidae)
Habitat: woods of Equatorial Africa.
If large animals can expect to own force and size in case of danger, tiny creatures
had to resort more often to tricks to not get to somebody for dinner. One of
prevalent dodges in animal kingdom is masking. Managing to pretend to what
you are not (and mainly - does not causing appetite in predator), it is possible
to receive quite good chance to survive. Various insects frequently act so,
simulating branches, knots, leaves of various degrees of freshness. In Neocene
epoch in tropical woods of Africa another kind of imitator had appeared: it
is small forest lizard, the gecko simulating tree leaf.
Leaf-looking gecko lives on trees, inhabiting branches in forest canopy at
height of several tens meters above the ground. It is rather large for the
gecko though its body length including tail does not exceed 20 cm, and tail
accounts about one third of general length. The body of this lizard is strongly
flattened in dorsoventral direction, simulating leaves of tree. On edges of
body and tail the wide skinny border grows, forming scalloped outgrowths: it
helps animal to look rather convincingly, simulating insect-eating leaves.
Besides outgrowths on edges of body help to reduce up to a minimum treacherous
shadow can come to naught masking of the reptile. Head of leaf-looking gecko
is flat, eyes are strongly spread and look in sides. Above eyes triangular
skinny outgrowths stick up in sides, masking them at sight from above.
The body shape is harmoniously supplemented with colouring. The body of leaf-looking
gecko is colored from above bright green. Along back thin light strip passes
simulating middle nerve of leaf, and on each side there are short cross strips.
At edges of lateral outgrowths of body there are some brown irregular-shaped
spots: it is the imitation of damaged leaves. Even eyes of this lizard are
perfectly masked: the iris of the eye is colored green, and vertical pupil
may be voluntary narrowed up to the thinnest crack, imperceptible at passing
to the hidden lizard. When this gecko sits on branch or on leaf, having turned
up paws under body, it cannot be founded while it’ll move.
But the belly of leaf-looking gecko is colored deliberately contrast: at males
it is black with red spots and dubs, and at females it is one-color black one.
Such distinction helps to individuals of different sexes to distinguish each
other.
To live on the tree, it is not enough to have only cryptic colouring. Paws
of many geckoes, even largest ones, are well advanced and also are very tenacious
due to set of microscopic fibers taking place on their bottom side. The leaf-looking
gecko is not exception here: its wide flat fingers are adapted to keeping even
on smooth surface of leaves.
Main food of this species of geckoes is insects. Large cockroaches and small
termites – all of them are devoured with identical pleasure. The leaf-looking
gecko supplements insect diet with small snails and ripe fruits with soft pulp,
even little bit begun to rot.
By their nature leaf-looking geckoes are solitary animals. They occupy the
certain territory somewhere in crone of tree, and zealously preserve it against
neighbours. Each lizard declares its rights to territory by the loud cries
similar simultaneously to bark of tiny doggy and voice of the newborn crocodile.
If any gecko will come to the territory occupied by another one, owner of territory
at first tries to banish it, having started to shout more loudly and more often.
If the newcomer does not go away, the owner leaves shelter and creeps towards
to the unbidden visitor. Both opponents start to show themselves, having risen
on legs and from time to time sharply nodding heads. But such demonstration
does not proceed long: the newcomer either leaves in good time or fight begins.
Both lizards bite each other, drag each other by legs or lateral outgrowths
of body. Usually in this case both opponents get numerous scratches from sharp
teeth of the neighbour.
Only time when such selfish creatures willingly suffer presence of strangers
(but not everyone!) at their territory is breeding time. In courtship season
males of leaf-looking gecko show bright belly, involving females. For this
purpose they hang on branch headfirst, gripping it only by back paws. Thus
they utter the special cry similar to chirp of bird. The female has entered
to territory of the male, should show two signals: belonging to fair sex and
humility. Coming nearer to the male, she turns a head by bottom jaw to his
side. It allows to see color of throat of the female, having no bright spots,
and simultaneously tames aggression of the male as very weak spot is shown
to him.
Pairing lasts some minutes, and then female leaves territory of the male and
twice zealously starts to protect her own one: in her organism ripening of
eggs begins. About three weeks after pairing the female lays in tree-trunk
hollow or behind peeled off tree bark two or three eggs. For one year the female
can make up to three - five such clutches.
First minutes shell of freshly layed egg is soft and sticky, but in air it
quickly hardens, and egg appears firmly pasted to substratum. About two months
later tiny (about 4 cm long) copy of the parent hatches from it. Young leaf-looking
geckoes have smooth outgrowths on sides of body and uniform green colouring
without brown spots. At their growing up jags at edges of body appear, and
in colouring in the beginning the pattern of leaf nerves and then dark stains
simulating decay will be shown. At the age of 18 - 20 months young lizards
can breed.
The leaf-looking gecko keeps mainly on branches of trees. And the closely related
species, lichen
gecko (Ph. lichenodermatus), lives on wood trunks and large branches.
It is similar to leaf-looking gecko, differing from it in larger size (up to
30 cm) and some features of appearance. On surface of its body numerous warts
and knobs imitating lichens on tree bark are located. Colouring of this species
is well matched with habitat: it is the combination of grey and brown colors
with greenish spots. Skinny edging of body at it is smoother.
The lichen gecko, just as leaf-looking one, eats insects have sat on bark.
Generally the diet of lichen gecko includes butterflies and capricorn beetles;
it catches them creeping closely and sharply rushing at prey. As many butterflies,
spending daytime on tree bark, have cryptic colouring, this gecko finds them
with the help of very keen sense of smell.
Flower
gecko (Anthogekko scandens)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family Geckoes (Gekkonidae)
Habitat: Northern Australia, rainforest canopy.

Picture by Alexander Smyslov
Lizards pass to vegetative food very seldom. The majority
of their species eats insects or small vertebrates. One of reasons of this
phenomenon is considered
to be easier digestion of food of animal origin in comparison with vegetative
food. But in tropics where climate is constantly warm, there is a significant
amount of herbivorous reptiles. During evolution vegetarian species appeared
in various families. And even among especially carnivorous lizards there
are the species eating sweet and high-calorific fruits of plants. In Neocenic
woods
of Northern Australia one species of geckoes have completely changed its
habits, having turned from predator to vegetarian. But the food which it uses
differs
in refinement: it is flower nectar.
The Australian flower gecko spends all life in forest canopy at dizzy height.
Here, in empire of wood crones and epiphytic plants it finds enough flowers
rich in nectar.
This lizard grows only to 15 cm length, and about half of it thin tail accounts.
Flower gecko is dexterous acrobat: its legs are rather long, and on fingers
characteristic for these lizards combs, permitting clambering on even smooth
surface of leaves, are advanced. This gecko moves slowly, but in case of
danger it can make very fast throws and even jumps up to meter long.
The skin of this lizard is colored green with bright red legs and spot on
nape; stomach and throat are white. Large eyes have straw-colored iris with
narrow
vertical pupil.
The head of flower gecko is narrow, and tongue can extend rather far. Practically
constantly these geckoes walk over the territory, searching for flowers of
plants. Because stocks of this food are rather small, lizard constantly inspects
territory, zealously preserving it from competitors. Each lizard declares
rights to territory, uttering loud sharp sounds similar to peal of hammer
against
anvil.
On the tip of tongue of flower gecko fibers forming hairy brush, convenient
for nectar gathering, grow. In addition to nectar plenty of pollen gets in
stomach of gecko. Pollen of many species of plants is not digested, but pollen
of some plants serves as additional food source for lizard. Also flower gecko
willingly regales itself with fruits begun to rot.
The breeding period of this species stretches practically for the all year:
at any time in wood there are young lizards of this species. The female lays
only 2 - 3 eggs, but egg layings can repeat up to four times once a year.
For breeding female chooses deep tree-trunk hollows or cracks under peeled
out
bark where eggs are inaccessible to casual predators. In breeding season
males willingly admit females to their territories, but after pairing banish
them.
The young growth hatches from eggs after 10 - 11 weeks. Young geckoes differ
from adults in brighter colouring: at them head, paws and tail are red, and
only on back there are some green spots. They spend the most part of time
on flowers of trees and epiphytes, disappearing at approaching of adult neighbours.
At their growing up more and more green spots merging in a continuous background
appear on their bodies.
Mouse-eating
gecko (Macrogekko myophagus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family Geckoes (Gekkonidae)
Place of an inhabiting: temperate forests of New Zealand, wood litter.
Picture by Carlos Pizcueta

Picture by Alexander Smyslov
In human epoch fauna of New Zealand had undergone radical
changes: in this “lost world” inhabitants unknown earlier, mammals, had appeared.
Among them
there were both herbivores and predatory species. Newcomers had started to
destroy local animals, and to the end of human era only separate species
had remained from endemic fauna.
But influence of alien species to fauna of New Zealand was not only negative.
Some species of local animals had developed new kinds of food being absent
at islands before human colonization. Murine rodents and even small predators
belonging to weasel family became the basic prey of one surprising creature.
In deciduous woods of New Zealand ground is covered with thick layer of fallen
foliage. In it numerous insects crawl, and small rodents hunt them. They
obviously feel like in safety, but this impression is deceptive Attentive
eye of predator
hidden literally right at mammal’s very nose observes of carefree rodent.
One careless movement – and rodent is seized. For a share of second leaves
laying
before it turn to head and paws of large-headed lizard. Reptile catches small
mammals by wide mouth and compresses by jaws. Quiet crunch is heard, and
little body of small mammal lifelessly hangs, and the hopeless peep breaks.
Lizard
slightly inclines head sideways, catches prey more conveniently, and starts
to swallow it, inclining head alternately to both sides. Eventually only
tail, sticking up from mouth of lizard, remains from unlucky rodent.
When the job is done, lizard creeps out of shelter: the successful hunter
has received dinner which will be sufficient to it to nearest few days. Now
main
task is to find good shelter where nobody will disturb this lizard.
The large lizard is the mouse-eating gecko - one of representatives of endemic
New Zealand fauna, the descendant of native species of these islands. When
small predatory mammal had appeared at islands due to human will, the majority
of New Zealand reptiles had been exterminated by them. But some gecko lizards
had survived, and one of their species has turned to predator.
It is a reptile up to 70 cm long; this one is true giant among geckoes, usually
do not exceeding 20 cm in length. It is remarkable, that before human colonization
of these islands in New Zealand very large species of geckoes was found.
At the mouse-eating gecko there are large head with strong jaws, rather short
paws and short tail.
This lizard hides in leaf litter, trapping small mammal, birds and reptiles,
and catching them by fast throw. This species avoids competition with predatory
mammals due to habit of life: it is active in the afternoon while predatory
animals go to hunting at night. For improvement of masking the skin of mouse-eating
gecko is covered with lines of warty outgrowths, and flat pointed outgrowths
stick up above eyes. The skin is colored brown with yellow “nerves” and dark
spots. Even eyes of reptile are masked: the iris of the eye is colored beige
with twisting vertical brown strips. The pupil of reptile looks like vertical
crack. Having slightly dug in rotten leaves, lizard becomes imperceptible
for possible prey.
At wood geckoes tail serves as the balance weight during tree-climbing. The
mouse-eating gecko spends most part of time at the ground, therefore the
tail at it had became much shorter, than at wood relatives. In tail stock
of fat
is accumulating permitting to wait cool winter days. Though in habitats of
mouse-eating gecko snow never falls, in winter it becomes appreciably colder
and appetite of reptile reduces. Mouse-eating gecko spends winter in hibernation,
hiding in deep tree-trunk hollow or in crack between stones. Lizard keeps
some activity even in the most cool winter time, slowly creeping in the refuge.
At the end of winter when sun heats up stones and trunks of trees, the lizard
creeps out to get warm not for a long time.
When short cool winter comes to an end, mouse-eating geckoes leave shelters
and start to feed again. During first weeks after hibernation they look poorly:
tail is very thin, and skin on body here and there is wrinkled. But gradually
they are eaten off: at first they catch insects, and then small animals.
Eggs of birds are special delicacy, and geckoes willingly climb for them
on bushes
and trees.
When lizards restore forces after wintering, tournament fights of males begin.
The male at this species is larger and more large-headed than the female.
In courtship season its eyes change color: the iris of the eye becomes bright
red. This color is supplemented with light pink mucous membrane of mouth.
Gecko males share forest to set of sites. The owner of site creeps on tree
trunk or stone, and declares rights to territory by cries. The voice of the
mouse-eating gecko resembles yelping of small dog. Having noticed the female,
the male starts to cry even more often and louder.
In clutch there are 2 - 3 eggs by size like chicken ones, with dense skinny
shell. Shell of freshly layed egg is sticky. The female lays them usually
in tree-trunk hollow or in crack between large stones. Here, in conditions
of
more stable microclimate, the incubating passes more successfully. The female
protects place of clutch, and drives neighbours off from it, puffing throat
and hissing.
The incubating lasts about two months. When young lizards hatch from eggs,
female leaves post and any time continues usual life. For a season she can
make only two layings, but survival rate of posterity due to protection of
a laying is rather high.
Young mouse-eating geckoes eat insects. Later they pass to feed by rodents,
and in the beginning ravage their nests and eat newborn cubs. At the age
of three years they become able to breed.
Butterfly
gecko (Papiliostellio quasivolans)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckoes (Gekkonidae)
Habitat: Indonesia (Jakarta Coast), crones of trees of tropical rainforest.

Picture by Lambert
Sites of land open to ocean winds near equator are a place
where a plenty of pre-glacial forms of life had kept. In Neocene some of them
have died out, not having sustained a competition to newcomers from more cool
areas, but as a whole variety and originality of life in these places remained
up to the mark. Reptiles are presented especially richly in equatorial area.
In Neocene they have started to occupy new ecological niches, turning to surprising
creatures.
One of such Neocenic reptile species had evolved on the long site of land earlier
representing the Big Sunda isles which in Neocene had merged to common belt
of land - Jakarta Coast. In conditions of winds constantly blowing from ocean
this creature had adapted to gliding flight. It is the small lizard – gecko
about 20 cm long including tail. It would look quite usual lizard, marking among
many neighbours only with bright red eyes. But at once feature for which this
lizard is named "butterfly gecko" is evident: on each side of the
body scales of this lizard are extended and have turned in feather-like outgrowths
with fibrous back edges. Such feature already had appeared during evolution
process at the Triassic reptile Longisquama dwelt in the Central Asia. But at
it such scales had grew along back in two parallel lines. But if about their
applicability at the fossil reptile long disputes had taken place their role
at the butterfly gecko does not cause doubts: with their help it can make long
gliding jumps from tree to tree. The length of "feathers" reaches
10 cm, they are slightly bent back. Especially long "feathers" grow
on sides at once near forepaws. To hips they gradually become little, on the
tail basement they are very small, but on the tip of tail they form the small
blade, suitable for jump driving. Small muscles are connected to the basis of
every "feather". Therefore the butterfly gecko can lift and lower
them and also change their inclination during a jump. All fingers of the butterfly
gecko are wide, tenacious, with hooked claws.
The top side of body of the butterfly gecko is painted not strikingly: it is
green with brown spots and grey points. By color and the texture the skin on
back of the lizard successfully imitates bark and lichen. But in breeding season
the reptile changes: from below "feathers" are colored much more brightly,
red-colored with white tips, and they are actively used in courtship games.
The male shows them to the female, having lifted upwards and having opened,
as if the peacock tail. Thus it marks time on stretched legs, trying to keep
to the female by side. The throat of the male gets at this moment white color
with black longitudinal strip.
This lizard is a predator eating insects and snails, cracking their shells.
It can catch flying insects like flies and butterflies.
This species breeds, laying eggs. In clutch there are only 2 rather large eggs
hiding by female in tree-trunk hollow. For one year egg laying can repeat up
to 10 times. Zygotes are kept in female organism for a long time and quickly
turn to eggs, therefore after one pairing the female can lay fertilized eggs
5 – 6 times.
The incubation lasts about 2 months. Just hatched geckoes are about 5 cm long.
They have no “feathers”, but their growth begins almost at once, and at the
age of 2 month reptile already completely becomes “fully fledged” and can make
long jumps. At the age of half year gecko becomes completely mature, and its
life expectancy in wildlife is about 5-6 years.
Midge
gecko (Muscigekko muscicarpus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckos (Gekkonidae)
Habitat: equatorial regions of Africa, forests.

Picture by Pavel Volkov
In the Neocene, the warm climate favours the life of reptiles,
and their diversity is comparable to what was observed in the pre-human era.
The areas of the greatest diversity of reptiles are the evergreen forests of
the equatorial regions of the planet. In order to avoid interspecific competition,
numerous species of reptiles become highly specialized in due course of evolution,
differing from neighboring species by food specialization or the choice of specific
habitats. An example of such specialization is demonstrated by the midge gecko
– one of the smallest reptile species on the Earth in the Neocene epoch. The
body length of an adult of this species is about 25 mm along with the tail.
This lizard species is remarkable in characteristic proportions: it has a large
bug-eyed head, a short wide trunk, well-developed legs and a shortened tail
– its length is less than a third of the total length of the lizard. The coloration
of this gecko is very dull: brown with dark spots along the midline of the back.
The throat and belly are white; the eyes are also circled around by narrow white
rings. The throat of males has an inflating air sac of red color, which is hidden
in a longitudinal skin fold on the throat at rest. When a lizard claims its
place of residence, it inflates its throat like a bubble, and then its coloration
becomes noticeable. At the same time, the lizard emits a pulse of high-frequency
sound. In the female, the throat swells weaker and the color of the gular sac
is white.
This species is a symbiote of large tortoises. Midge geckos live in pairs under
the carapace of the tortoise and hide there from the enemy at the first sign
of danger. Settling down, the midge gecko moves from one reptile to another
during their joint rest and mating. This reptile is a predator and feeds on
insects and ticks that get on the reptile’s body. Due to the activity of these
reptiles, the probability of infection of the host tortoise with infectious
diseases and parasites transmitted by blood-sucking arthropods is noticeably
reduced.
Despite living in a tropical climate, the midget gecko is viviparous: 3-4 times
a year, the female gives birth to 1-2 large well-developed juveniles almost
half the length of the mother. Young geckos have a monotonous brown color. They
settle by crawling over the bodies of other tortoises during their encounters
in the forest. They reach sexual maturity at the age of 4-5 months; life expectancy
is about 5 years.
Brush-tongued
gecko (Penicillingua ceriphaga)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckos (Gekkonidae)
Habitat: Jakarta Coast, tropical rainforests.
The restoration of tropical forests in the Neocene contributed to active speciation
among their inhabitants who survived after the human epoch. The restoration
of highly productive natural communities led to the appearance of species displaying
a strict food specialization as a way of avoiding competition. The productivity
of the community allows coexisting of species with extremely bizarre specializations;
one example of this is the brush-tongued gecko from the canopy of the tropical
rainforests of the Jakarta Coast.
This reptile species is very small – the length of an adult does not exceed
8 centimeters along with the tail. The length of the tail is about a third of
the total length of the reptile; it has a relatively large head with large eyes.
Legs are well developed, thin, and five-toed, with grasping fingers and toes.
The outline of the body of the brush-tongued gecko is quite unusual. The head
and trunk are strongly flattened, expanded and equipped with scalloped skin
outgrowths along the edges. The tail, on the contrary, lacks such outgrowths.
The coloration of the animal is relatively bright, with a characteristic color
pattern. The upper side of the body is bright green with a longitudinal stripe
of yellow from the tip of the snout to the tail. The tail is also bright yellow.
On the edges of the body there is a yellowish border with separate brown specks.
On the shoulders and back of the head, the yellow zone expands and is complemented
by large brown spots along the edge. Thus, the shape and color of the animal
accurately imitate a slightly insect-eaten plant leaf. The underside of the
body is yellowish-white. Large eyes are also camouflaged: the iris is yellow
with thin horizontal strokes of green. The pupil is vertical and silt-shaped.
This lizard keeps among the foliage at the ends of branches and among growing
shoots, where its prey is most often found. This reptile species is insectivorous,
and the basis of its diet consists of sucking sedentary and slow-moving insects
– coccids, aphids and scale insects. Due to the symbiotic bacteria living in
the intestines, this gecko is able to digest and assimilate wax, from which
these insects make protective structures.
Living in tropical conditions, this reptile species breeds all year round. The
mating ritual of this gecko is relatively simple: males chase and bite each
other, driving each other away from the female. The male quickly copulates with
the female and expels her from his territory. The fertility of this species
is low: the female lays only 2 eggs every two weeks; one mating is enough for
2-3 complete clutches. During the year, the female lays about 20 eggs, which
she hides in shady wet shelters: in the dust at the bottom of a tree-trunk hollow,
under the tree bark or in thickets of epiphytes. Incubation lasts about 5 weeks.
Young animals feed on small invertebrates with soft body coverings – slugs,
aphids and springtails. Juvenile has a narrow body and only the head is expanded
in the back. With age, skin outgrowths appear on the body and tail, providing
camouflage. Sexual maturity comes at the age of 6-7 months. Life expectancy
is no more than 6 years.
Salt
gecko (Salinogekko setipes)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckos (Gekkonidae)
Habitat: Mediterranean lowland, breeding in oases.
The closure of the Strait of Gibraltar led to the rapid drying up of the Mediterranean
Sea and the transformation of large areas between Africa and Europe into almost
uninhabitable salt-covered deserts. The harsh climate and lack of water forced
the few inhabitants of these places to gather in “oases” – on the highlands
that were once islands of the Mediterranean. Here, water condenses from the
air in small amounts, supporting the life of the inhabitants of these “shelters”.
Drought-resistant reptiles thrive here, one of which is the salt gecko, a descendant
of the small Kotschy’s gecko (Mediodactylus kotschyi), one of whose subspecies
has adapted to life among salt.
It is a rather large representative of the gecko family: large individuals reach
a length of 15-20 cm, which allows them to store large internal water stocks.
The physique is robust, the tail is short and thick – it serves as a place of
fat storing. Large legs are armed with claws. The skin on the throat, belly
and paws is light yellow; the eyes are quite large and have a marble-white iris
and narrow vertical pupils. The nostrils are equipped with corneous caps with
keratinous villi along the edge, protecting against inhaling salty dust.
The salt gecko is one of the best specialists in camouflage in the Neocene:
the skin on its head, back and sides is covered with large “faceted” scales
and the texture is similar to salt crystals. The salt gecko has unusual physiological
features: its kidneys are adapted so that it can sometimes drink salt water,
but it rarely does this, receiving most of the moisture from its food. The tip
of its hunting tongue is covered with keratinized tissue with notches. On the
underside of the paws there are many corneous ridges keeping the body above
a layer of sun-heated salt.
This lizard lives among the salt desert, withstanding considerable heat in the
absence of moisture. At night, the reptile buries itself in salt, which preserves
the warmth of the sun, during the day it is awake – mainly in the morning and
evening, waiting the hottest daytime hours in shelters. In addition, the salt
gecko tries to live near reservoirs, where the amount of prey is greater. The
usual diet consists of a variety of insects adapted to life among salt, such
as beetles and ants, which it catches by picking up with the tip of its tongue.
The keratinized tissue on the tongue allows it to keep prey, salt drops away
between the bristles, and so the lizard avoids swallowing it unnesessary. Sometimes
salt geckos visit reservoirs. There is much more food there, and it is much
more diverse. From time to time, these lizards enter shallow waters, where they
catch larvae of saltland bloodfly,
marsh brine shrimps,
young larvae of water turtlebeetles
and hawk robberfly. Lizards
avoid going deep into the water. During the breeding seasons of crested
flamingos, this gecko can eat their eggs or dead chicks. If it is noticed
by adult birds, the gecko quickly runs away and hides among the stones or buries
itself in salt. Also these lizards sometimes eat the eggs of large Antheos
tortoises.
Despite its apparent clumsiness, the salt gecko is a good jumper; it is able
to hunt large flying insects by jumping into the air. These geckos often climb
on the carapaces of Antheos tortoises to catch blood-sucking insects attracted
by large reptiles. Finding the corpse of an animal larger than itself, the salt
gecko can tear off pieces of meat, even dried up, from it. Sometimes it also
eats the fruits of the berry
salttree, spreading its seeds. The enemies of this species are salt
hawks, as well as migrating birds of prey on the passage across the Mediterranean
hollow.
The breeding season is in winter, when temperatures are not so extreme. During
primitive courtship games, males chase and bite each other, trying to break
off pieces of camouflage scales. The winner mates with the female and immediately
leaves her. Despite the high temperatures in the habitats of these reptiles,
this species is viviparous – dry air and salt create unfavorable conditions
for egg incubation. After the only fertilization, the female can give offspring
up to 4 times in a row – there are “spare” egg cells in the oviducts, which
are dormant. Pregnancy lasts up to 3 months, and the youngs are born in the
spring, at a time favorable for life. Fertility is very low: 1-2 large juveniles
(up to 10 cm long) are born at once. Delivery usually takes place in “oases”,
and the female actively feeds after it. Young geckos live in oases until they
reach a length of about 15 cm. At this time, they form large “crystalline” scales
on their backs. Before that, they look like ordinary geckos and hunt insects,
spiders, other arthropods and snails in the undergrowth of oases.
Sexual maturity comes at the age of 4-5 years; life expectancy is up to 20 years.
This reptile species was discovered by Mamont, the forum member.
Whistling
jumping gecko (Saltarogekko sibilator)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckos (Gekkonidae)
Habitat: Africa, from savannas to tropical forests, bodies of large herbivores.
In the process of ecosystem restoration in the early Neocene, new forms of coexistence
between the descendants of the surviving species were established. In the process
of evolution, they acquired bizarre specializations that allow them to survive
in specific conditions. One example of such adaptations is showed by the whistling
jumping gecko, a descendant of one small gecko species that inhabited Africa
in the human era.
Whistling jumping gecko is a very small reptile about 13 cm long, of which 6
cm falls on the body. Its physique is unusual: the hind legs are very long and
muscular, adapted to jumping, the front ones are not very large, but have tenacious
fingers. The head is quite broad, with an elongated muzzle, the neck is short,
the tongue is wide and mobile. The body color is brown with thin longitudinal
stripes, allowing the reptile to disguise itself among the fur of animals; the
lower part of the head and belly are white. A resonator sac is developed on
the throat.
The whole life of these reptiles represents a wandering on large
animals living in the African savannas. However, sometimes whistling jumping
geckos stay on “clients” for a long time. They are able to move on the ground
and tree branches by running and jumping, and when they meet a large enough
animal, they jump on it and stay on it to feed. Once on the “set table”, the
reptile begins running around the animal and catches parasites such as lice
and ticks, which are constantly found on mammals. The ability to jump allows
this gecko catching flies and other flying insects hovering around the giants.
In addition, it is not difficult for the lizard to pull out the gadfly larvae
embedded in the skin, strongly squeezing the fistula with its jaws: the bite
is very strong. If the host animal runs away from enemies, the whistling jumping
gecko clings to the wool or feathers to avoid falling. Living on a large animal
allows the reptile to avoid many enemies, but these geckos fall prey of birds
searching for parasites on large animals, and on the ground small predatory
reptiles, birds and mammals are dangerous for this lizard.
Each lizard guards its food-bearing animal, preventing competitors from entering
it. If the lizard sees a competitor, it stands on its hind legs and whistles,
inflating the resonator bag. Only a female, ready to reproduce, is able to tolerate
the presence of a male for a while, but after mating, she drives him away.
This species does not have a pronounced breeding season. If a male and a female
are nearby, then they mate. The rate of reproduction is quite slow: the female
gives birth to 2 juveniles every 3 months. One mating with a male is enough
for normal reproduction for two years. Cases of parthenogenesis are not uncommon,
with all offspring consisting exclusively of females.
The juveniles stay on the host animal for a short time. They quickly leave it
and take cover on the ground. At the beginning of life, whistling jumping geckos
feed on insects and small spiders and sometimes catch termites in termite mounds,
and later switch to adult food and begin to travel in animal fur.
Sexual maturity comes at the age of 2 years; life expectancy is up to 15 years.
This reptile species was discovered by Mamont, the forum member.
Greater
Fruit-Gecko (Durophelsuma gigas)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Geckoes (Gekkonidae)
Habitat: Madagascar, canopies and understory of rainforests and other woods
containing fruiting trees.
Charismatic reptile found in Holocene Madagascar other than various chameleons
was the Giant Day Gecko (Phelsuma grandis), diurnal gecko of fairly large size
which fed on insects and nectar and enjoyed licking the pulp of fruit. From
this form has evolved fruit-eating geckoes in the Neocene, including giant forms
equal in size to the largest extinct forms of day gecko made extinct by man
in the 1800s.
Greater Fruit-Gecko is the largest of these kinds of gecko, reaching 40 cm long
and being powerfully built. Form of body and limbs generally resembles its ancestor,
but is more robust, limbs are slightly longer and more muscular, feet bear gripping
pads typical of most geckos. Differs from ancestor is the form of the head,
jaws are adapted to bite into and husk large fruit, being thicker and more rounded,
teeth are spade shaped and weakly serrated. Tail is fairly short and thick,
serves as a store of fat. Coloration is much less bright than its ancestor,
dark brownish green with pale yellow stripes and spots along sides, belly is
whitish yellow. This lizard is diurnal and adapted to distinguish colours of
ripe fruit, hence eyes are large and have a round pupil.
This lizard spends almost all of its time in the trees, climbing on branches
or sitting against boughs or trunk in order to conceal itself of bask in the
sun, occasionally it may descend to the ground to eat fallen fruit. Animal swallows
small fruits or berries whole, passing seeds in dung, but is also able to husk
and bite pieces off larger fruits. When confronted with a predator, it may flee
but will usually scream loudly and gape its mouth in a threat display.
Breeding occurs in the dry season, males will make loud barking chirp in order
to attract the female. If more than one male pursues a female, a fight will
break out in which males lock jaws and attempt to exhaust one another.
Female lays a clutch of up to 4 large eggs, which are initially very sticky,
fastened to a secluded shady spot on a tree branch or trunk, sometimes in a
tree hole or the underside of a bough, the female will leave the eggs to hatch
on their own. The incubation lasts up to two month. Female may make two layings
within the breeding season. Baby geckoes are lighter in colour and feed on insects
and nectar, sexual maturity is reached at three years.
This reptile species was discovered by Timothy Donald Morris, Adelaide, Australia.
Nocturnal
thief gecko (Aeluroscalabotes latronox)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Eyelid geckos (Eublepharidae)
Habitat: primary and secondary rainforests of Southeast Asia.
In the Holocene, the genus Aeluroscalabotes, represented by the species A. felinus,
existed in the Eublepharidae family. It was a mobile animal led a nocturnal
lifestyle, which managed to survive extinction in some shelters – pockets of
forest stand in remote mountainous areas. It became the progenitor of a new
species of the Neocene epoch – the nocturnal thief gecko.
This animal is slightly larger than its predecessor, being 18-20 cm long. This
species is a slender gecko with a triangular, flattened head, elongated muzzle
and large eyes. The animal’s legs are relatively short and muscular, with long
thick claws and widely spaced fingers/toes. The tail is slightly shorter than
half the body length; it is thick and strong, capable of spiraling – when this
gecko climbs vines, the tail acts like a “fifth paw”, allowing the animal attaching
itself more firmly to the support. Two small corneous tubercles grow between
the eyes – these are greatly enlarged scales. The body is of brown color: the
underside is light, turning creamy on the belly, and the back is very dark,
with grayish and white spots, making the reptile hardly noticeable against the
background of the tree bark. The tail is decorated with white spots, and males
have in addition a white stripe on its sides. In case of danger, the reptile
can easily drops its tail, which then grows back, but loses its former flexibility
and has a darker color. The irises of the eyes are dark, the pupils are vertical
and slit-shaped, and the bumps on the head are black.
The disturbed gecko first tries to freeze, relying on its cryptic coloration.
If this trick fails, the lizard gets a typical threatening pose, strongly arching
its back, and exposes a brightly colored tail. In extreme cases, the reptile
opens its mouth wide to display a dark blue tongue tip.
This animal is nocturnal, getting out of its shelter at dusk. During the day,
it hides among the roots of epiphytic plants and lianas, in hollows – much higher
than its ancestor, that preferred to live closer to the forest floor. Thus,
its shelter is located 5-6 meters above the ground and inaccessible to terrestrial
predators. Arboreal predators from the forest canopy also rarely attack this
species. The nocturnal thief gecko feeds on eggs, nestlings and young mammals,
ravaging the nests of small animals. The young lizards of this species feed
on insects.
The nocturnal thief gecko is a very moisture-loving species that actively consumes
lots of water, so it prefers to arrange its shelters near thickets of epiphytes,
in which foliage water accumulates, or on trees growing along the banks of forest
rivers.
Pregnancy lasts 50-65 days, depending on the temperature. Mating has two waves
of intensity: from October to December and from March to April. The female lays
2-3 eggs, which she buries in rotting plants. Incubation lasts about 2 months.
Life expectancy reaches 10 years, and these reptiles become sexually mature
in a year and a half.
This reptile species was discovered by Ilia, the forum member.
Ridge-backed
Marine Iguana (Marephilosaura epicristata)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Iguanas (Iguanidae)
Habitat: Galapagos Islands, coastal areas and near shore waters of all islands.
The presence of humans on the Galapagos Islands during the Holocene disturbed
and endangered the unique reptiles living there. Giant Tortoises became very
few in number, land iguanas became extinct, and Marine Iguana (Amblyrhynchus
cristatus) became rarer, and was forced to breed in more remote places. However
it survived to leave descendants in the Neocene.
Ridge-backed Marine Iguana inhabits the coasts and coastal waters of most of
the Galapagos Islands, but in order to breed it migrates to remote islands free
of pigs and other nest-wreckers. It reaches about 1.8 meters long but usually
less, of which more than half is tail, colour is similar to that of its ancestor,
perhaps a little darker, dark bluish black with light blue and orange highlights.
Legs are strongly built and feet bear large claws, with these it grips tenaciously
to rocks underwater in order to feed. On the crown of the head corneous scales
of conical shape grow, culminating in a cluster of larger “horns” which are
used in courtship displays and fighting. Appearance differs from ancestor in
one important way, tail is deep and flattened and this continues into a moderately
sized “sail” or ridge along the back, which has a peak on it, similarly to the
Philippine Water Dragon (Hydrosaurus). This feature is made even more prominent
by the presence of a crest of pointed scales, which is taller in males. As with
its ancestor, excess salt is secreted into the nose and expelled with a snort.
These lizards congregate at the coasts in large groups that may number hundreds,
feeding upon seaweed and filamentous algae that grow in rocky coastal waters.
Individuals dive under the water and cling to submerged rocks, scraping growth
with their teeth, this forms all of its diet. When they become too chilled by
the water, they emerge to bask and sun themselves.
Animals are diurnal, they become sluggish and sleep by night.
Breeding season occurs in the dry season, males and females venture inland in
order to mate, and for egg laying, females swim to the most remote islets devoid
of nest-ruining predators. Males compete to impress females, bobbing heads,
pushing each other and jousting with facial horns, as well as biting and wrestling.
Dominant males may proclaim their domain by marking their territory with a waxy
pheromone secretion from special glands. After mating, female produces eggs
after 50 days, and it is in this context that females have horns too, they fight
and compete over nesting grounds in a similar way to the fighting of the males.
Courtship involves the dominant male chasing, nipping and pawing at the female,
copulation may last several minutes, and males do not stay with the female after
mating. Eggs are typically about 4 centimeters long, young are independent from
birth, but differ from adults in smoother skin and darker colour. Incubation
lasts up to 110 days, up to 15 eggs are laid; sexual maturity is reached in
about 2 years but the first clutch may not be laid until a couple of years later.
Lifespan of this animal may be as long as 20 years but usually much shorter.
Predators include birds of prey and the Galapagos
Nano-Otorongo, wild pigs such as the Cerdito-peluda
may dig up and consume their eggs, their young are preyed upon by many kinds
of other reptile and birds.
This reptile species is discovered by Timothy Donald Morris, Adelaide, Australia
Duckweed
basilisk (Nanobasiliscus lemnodromus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Casquehead lizards (Corytophanidae)
Habitat: South America, slowly flowing rivers, swamps and lakes.
Picture by Alexey Tatarinov
At the boundary of Holocene and Neocene the area of tropical
woods had been reduced everywhere at Earth. Only in Indonesia, Equatorial Africa
and South America small sites of rainforests had kept. Only few inhabitants
of this type of ecosystems had managed to survive up to Neocene. Some inhabitants
of rainforest had died out because of chasing from the part of people; others
had not managed to keep viable populations at isolated sites of solid earlier
area. But the small part of inhabitants of tropical wood had kept up to Neocene
and evolved actively. Among them there were mainly medium-sized animals not
demanding a plenty of food and able to endure short-term deterioration of inhabiting
conditions. Reptiles having such abilities, had received in Neocene the right
of the further evolution. Among them fantastical and original lifeforms have
appeared.
One of surprising reptiles lives in rainforest reservoirs. It is tiny lizard
with brightly yellow corneous crest on head. It can be seen more often on leaves
of water lilies where this reptile is heated, having stretched paws in sides.
Length of this creature is about 20 cm including long tail making more of half
of the general length.
While the lizard is quiet, it differs from other neighbours with nothing. Only
its rear paws are approximately twice larger than front ones, and toe tips are
edged with thin bristle-like outgrowths of scales. When above surface of leaves
the fly flies by, the lizard reacts instantly: it jumps up, and rushes for the
fly. When the insect turns off to water, the reptile does not lag behind: slaping
by paws on surface of water, tiny lizard jumps, snaps fly right in air, and
runs back to leaves right on water surface. Lizards of basilisk genus (Basiliscus)
dwelt in historical time in South America had this skill. This reptile has inherited
and has developed this unique skill, having specialized to life on water surface.
It weighs so little, that can easily run on thickets of duckweed (Lemna) spread
on surface of water. Hence the name of this lizard – the duckweed basilisk.
To be simply very small – it is obviously insufficiently to have ability to
run on water surface. In body of duckweed basilisk also qualitative changes
have taken place: its bones are strongly pneumatized, air bags penetrate them,
as at birds. The constitution of this reptile is very fragile – at it there
are thin paws and long thin tail. The adult reptile weighs no more than 20 grammes.
The skin of the lizard is colored bright green which shade can vary depending
on condition and mood of the lizard. Stomach of the reptile is white. The skinny
crest on head of the lizard is colored lemon yellow shade. It is an attribute
using which lizards distinguish congeners.
Duckweed basilisk can not only run but also swim, though it makes it reluctantly.
In water the lizard easily can fall prey to various predators – fishes and turtles.
Besides duckweed basilisk is too tiny to swim quickly enough and to escape from
underwater predators. And one more danger is that this tiny lizard can easily
cool down in water and becomes inactive. Therefore duckweed basilisk escapes
in water only when there is no another way to hide from predator (for example
when the reptile is attacked from air by bird of prey or bat). The main way
of rescue from enemies at this reptile is fast run.
Duckweed basilisk eats almost exclusively insects living on surface of water.
It eats water scaters and catches larvae of mosquitoes in leaf axils of floating
plants. Occasionally duckweed basilisk eats small snails (more often it is made
by females shortly before to egg laying), fry and tadpoles living at the surface
of water. In dexterous jumps duckweed basilisk catches damselflies and the flies
hovering above water.
Duckweed basilisk is a tiny creature which is quickly heated up and quickly
loses heat. At night when it becomes colder, reptile gets into axils of leaves
and falls into catalepsy. At night the duckweed basilisk is very sluggish and
easily can fall prey to various predators, therefore it tries to hide more reliably.
In the morning lizard gets out on leaves of plants and is heated in first beams
of sun.
It is unprofitable to be too heavy to animal having such habit of life, therefore
at females of duckweed basilisk one ovary is reduced, and eggs develop and are
laid by two – three ones. The courtship season at duckweed basilisk lasts a
year round. The male of this species is smaller than female, and it also differs
with more advanced red coloured gular pouch. It arranges displays for females,
climbing on stalks and peduncles of aquatic plants sticking up above the surface
of reservoir. Involving females, male stirrs head up, puffing ular pouch. When
two males meet, they begin bloodless and transient tournament. Males show gular
pouches to each other, are shaken on peduncles, and in near fight try to push
each other out in water. At this moment they become striped.
The female ready to pairing shows the submission to the male, having light green
colouring. After pairing it undertakes risky travel on coast, and lays eggs
among moss thickets on tree trunks at height up to five meters. Cycles of breeding
repeat each two weeks.
Young reptiles hatch approximately after three weeks. Young lizards go down
from tree trunks and search for reservoirs where they will spend, probably,
all life. They easily settle on floating plants.
Life expectancy of duckweed basilisk makes no more than 4 – 5 years. But the
female starts to lay eggs at the age of five months.
The idea of existing of this animal was stated by Rafael Silva do Nascimento, Brazil.
Cockatrix
(Cocatrix cocatrix)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Casquehead lizards (Corytophanidae)
Habitat: plain areas of South America.
The end of the Holocene and the beginning of the Neocene were the time of major
changes in the Earth’s biosphere. Human activity caused a drastic decrease of
biological diversity, and the ice age changed the boundaries of natural zones.
At this time, South America once again separated from the North America, and
became an island continent again. Its flora and fauna began the development
in isolation again, acquiring their unique features.
The brown basilisk (Basiliscus vittatus) was similar in many ways to its more
famous counterpart, the common basilisk, and differed from it mostly in color.
In the Neocene, the descendants of these two related species began differing
sharply from each other in their ways of life and acquired various adaptations
for survival. The descendants of the common basilisk remained residents of the
Central American rainforests, while the descendant of the brown basilisk managed
to master life in the almost treeless open spaces of South America – it clearly
took this evolutionary step during the ice age, when the area of tropical forests
reduced to a minimum. Outwardly, it still resembles other members of its family,
but its proportions have changed somewhat: the hind legs have become more powerful
and longer than the front ones, the tail has become longer and turned to a good
counterweight for a proportionally larger head, which seems even larger due
to the leathery crest growing on it – hence the name, in honor of the rooster-like
monster of ancient and medieval bestiaries. It is a fairly large lizard – males
can be almost one and a half meters long, females are slightly smaller and more
massive. Unlike its mythological prototype, the cockatrix is quite harmless
and omnivorous: it eats insects, smaller lizards and other small vertebrates,
as well as various parts of plants, especially flowers, fruits and seeds. Quite
often, this reptile follows groups of hunting
herons at a respectful distance in order to pick up the remains of their
prey or catch small animals startled by these birds. This lizard is a very good
runner, and a healthy individual of this species can usually run away and hide
from an enemy (usually from a large snake, a predatory mammal or a bird), but
the imperfection of physiology does not allow the lizard to run for a long time.
The large mass no longer allows the animal to run on the surface of the water,
but these lizards can swim well and cross narrow rivers. The cockatrix, unlike
other basilisks, does not climb trees – the physique and especially its hind
legs no longer allow it.
The mating season of the cockatrix takes place during the drought period. At
this time, males get a bright coloration: the crest acquires an orange color
with a black upper edge, the upper part of the head also turns orange, and the
cheeks, nape and neck become dark brown. A pattern of wide vertical brown stripes
appears on the body. The coloration of the females becomes marble-speckled with
a pale yellow head. Males gather in groups among the grass and attract females
by quickly nodding their heads. A mandatory component of the mating ritual is
a group run of males following the female. After mating, the female lays an
average of 10-12 eggs, buries them in the ground or in fallen leaves, and does
not guard the clutch. In the summer, juveniles hatch from eggs. They differ
from adults in their slender build, yellowish coloration with longitudinal dark
stripes and narrow heads. They live in the grass, climb its stems and hunt insects.
In case of danger, they freeze in an upright position among the stems, clutching
them with their paws. Freezing in place in an upright position near objects
remains a protective reaction even in adults.
Cockatrix can live up to 20 years, but most of these reptiles die much earlier.
This reptile species was discovered by Bhut, the forum member.
Gliding
anolis (Pteroanolis volans)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Anoles (Polychrotidae)
Habitat: south and southeast of North America.

Picture by Lambert
Sometimes evolution makes experiments, which may not come
to mind. It had produced not one, but three groups of actively flying vertebrate
animals – pterosaurs, birds and bats – and a number of species, which can passively
glide down, and even snakes are among them. In Neocene this situation remained
unchanged. Active flight is an exceptional property of bats and birds only,
and numerous species from other groups of vertebrates in different places of
the Earth glide from tree to another. Gliding anole from south and southeastern
part of North America is one of them.
Compared to its ancestor, a certain kind of the anole lizard (genus Anolis)
of Holocene epoch, this lizard has barely changed in size: its length it up
to 16 centimeters, except for tail. This reptile is not green, but reddish-brown
in color, because it leads mostly crepuscular lifestyle, spending the days in
shelter on tree trunk, in tree hollow, or among dense branches, and at night
and dusk it awakens and begins hunting for various insects and other invertebrates.
Gliding anole has rather large eyes and it sees pretty well at dusk, when it
is searching in the trees for food and females and makes gliding jumps. Like
in the case of unrelated flying dragon (Draco volans) of Holocene epoch, its
“wings” are located on sides; they represent flaps of skin of yellowish-white
color stretched on framework made of gristle rods: during the day, if a gliding
anole is startled by a predator, it opens these skin flaps harshly. The sudden
appearance of bright colors may startle the predator and give the anole few
extra moments to run or glide away. White spots on wings are also used by these
anoles to signal to each other in twilight.
In late winter or early spring the gliding anoles’ breeding season begins. Males
begin calling the females by uttering cries sounding like squealing and barking
of tiny dog. When females fly closer, males court them, inflating their throat
sacs and making courtship dance. The throat sac of the male reddens up in breeding
season, and the reptile displays itself by flashing the gliding membrane and
bending its head on the back; at this moment throat sac is clearly visible on
their background and is seen by a female staying nearby.
In late spring the females bury their clutches numbering from 1-2 to 10 eggs
in leaf litter or into any other substrate, and do not care for them anymore.
A month and a half later the young lizards hatch from the eggs. They do not
different in colour from adults, but their “wings” have the same coloration
as the body. The sexual maturity comes at the age of one year.
The maximum life span of gliding anole is 18-20 years, but the most part of
these reptiles dies much earlier.
This species of reptiles is discovered by Bhut, the forum
member.
Translated by Bhut.
Snail-eating
anole, mokoli’i (Paranolis mokolii)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Anoles (Polychrotidae)
Habitat: Hawaii, tropical forests.

Picture by Pavel Volkov
Human activity has left an indelible mark on the biogeography
of the Neocene: there are great numbers of biological species whose ancestors
originate from other biogeographic regions and could not settle in their habitats
naturally. The tropical forests of the Hawaiian Islands harbor the mokoli’i
lizard (the name means “lizard” or “dragon” in Hawaiian), or snail-eating anole,
which is a descendant of the North American green anole (Anolis carolinensis),
introduced to the archipelago by people in the historical era. In the process
of evolution, it specialized in feeding on snails, differing in this feature
from related species, which are mainly herbivorous.
This species is a relatively large reptile of a lizard-like appearance, with
well-developed limbs. The body length of the snail-eating anole is about 30
cm; the tail is of the same length. Legs are strong, widely spaced, adapted
for walking, running and climbing trees. The head is large, with powerful jaws
and large eyes adapted for vision in the semi-darkness of the undergrowth. A
distinctive feature of this species is its teeth appearance – they are massive,
wide and blunted, of crushing type. In the depth of the mouth, several cusps
develop on each tooth.
The body color is usually green, less often brown, with numerous transitional
shades and patterns – there is a significant number of populations that differ
in background coloring. The belly is always grayish-white, but in dark-colored
individuals it is gray. There is a red gular sac designed for courtship display
and recognition of relatives. The eyes are light yellow with a round pupil.
Mokoli’i lives mainly in the undergrowth; each individual occupies a certain
territory, marking it with odorous feces smeared against the tree roots. This
is due to a narrow food specialization: the reptile constantly inspects its
territory in search of various snails. Having found a snail, the reptile snaps
it with its mouth and squeezes its jaws hard. When the crunch of the destroyed
shell is heard, the anole takes the soft body of the mollusk from the shell
fragments and eats it, and spits out the inedible fragments of the shell. It
is able to eat almost all kinds of snails in Hawaii, even a burning
snail, which has almost no other enemies – the lizard is insensitive
to its venom. Sometimes the snail-eating anole can climb trees, where it also
searches for gastropods. Having found a large Hawaiian
bird-eating snail at a bird’s nest, the lizard defeats it and eats it, additionally
having a snack of bird eggs. In swampy areas of the forest, mokoli’i is able
to hunt aquatic snails in shallow water. In addition to snails, mokoli’i also
eats insects, land-dwelling crustaceans, bird eggs and small mammals, and very
rarely feeds on carrion. The activity is diurnal; the lizard prefers to hunt
after the rain, when snails crawl out of their shelters in large numbers. Among
the enemies of this species, there are mammals, birds of prey and larger reptiles.
The snail-eating anole runs away from them, and if it doesn’t help, it bites
very hard. It can also climb trees in case of danger.
There is no determined breeding season. If the male finds a female, they mate,
and the female lays eggs under the roots of the tree, buries the clutch, and
no longer cares about the offspring. The clutch numbers 9-12 eggs. 5-6 weeks
later, the juveniles hatch. At first, they feed on small insects and snails,
gradually beginning to consume larger food with age.
Sexual maturity comes in the third year of life; life expectancy is up to 15
years.
This reptile species was discovered by Mamont, the forum member.
Salt-loving
skinkfish (Ichthyoscincus halophilus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Skinks (Scincidae)
Habitat: Mediterranean salt swamps.

Picture by Alexander Smyslov

Picture by Saurrian
When in early Neocene Mediterranean sea has completely dried up, rich fauna
of fishes and other water animals inhabited it earlier had left to non-existence.
After changes of climate water by means of plentiful rains gradually began
to come back to extensive salt desert of Mediterranean, having formed extensive
shallow reservoirs and bogs. But in these places no one fish may be found:
the salinity of water is too great for them, and Mediterranean is not connected
to any sea of World ocean. Nevertheless, at the bottom of Mediterranean reservoirs
live creatures teem: larvae of different species of flies, and also small crustaceans,
steady against the high contents of salts in water. But if the local birds,
flightless crested
flamingoes eat crustaceans,
larvae of flies living at the bottom of reservoirs, feel like in relative safety
from them. And nevertheless they have enemy.
In greenish because of seaweed and muddy water long white creatures slide,
wriggling. They are similar to fishes, but they are not fishes: from time to
time they emerge to surface of water, putting out muzzle tip, and breathe with
characteristic puffing. But they also are not amphibians: any newts or salamanders
will not endure so salt water. When one such creature creeps out on stone,
it becomes evident, that it is the reptile: its body is covered with dense
granular scales. It is the lizard – the skinkfish.
Desert inhabitants, skinks have developed adaptations for survival in conditions
of shortage of water. They prosper in most droughty places of planet. And in
bogs of Mediterranean these features were useful to them, having permitted
to master new habitat, atypical for lizards – supersalt reservoirs.
The length of body of skinkfish is about 60 cm; from which tail amounts about
2/3 (males are longer, than females, but their body is more graceful). Body
of animal is cylindrical; tail is flattened from sides and edged from above
and from below by wide skinny plica, permitting to swim. Head of the reptile
is pointed in front, covered with thick horn scales. Eyes of skinkfish are
protected by eyelids grown together – at this reptile it is unblinking “snake”
sight. Nostrils of skinkfish are shifted upwards and protected by skinny valves
which are closed when lizard dives. Paws of reptile are similar to mole’s ones
– they are short and thick, with long claws. Hinder legs are little bit longer
than forepaws. Because the reptile spends significant part of life in water,
belts of extremities are poorly joint to backbone that gives the skinkfish
some advantages.
The body of reptile is covered with dense corneous cover shedding similarly
to snake skin – it exfoliates from edges of mouth, and gets off by entire piece.
During shedding skinkfish rubs body against stones and twists to rings, rubbing
different parts of body against each other. Colouring of skin of skinkfish
is grayish-white without spots and strips.
In physiology of this reptile there is one feature directly connected to habitat:
very large kidneys stretched practically along all body from shoulders up to
pelvis, partly coming even in tail due to elastic connection of pelvis and
backbone. These kidneys effectively eliminate from blood salt, plentifully
treating to animal with food.
Skinkfish feeds with few species of insects had mastered life in Mediterranean
swamps: larvae of flies and water
beetles. Fodder areas of skinkfishes stretch in shallow waters, where under
the bright sunlight microscopic algae expand on stones plentifully. The basic
catch of skinkfish, larvae of blood-sucking
flies, scraping this seaweed by mouths, keeps there. Usually skinkfish
cautiously creeps to catch, drifting in thickness of water. It is not afraid,
that it will be noticed – larvae of flies are completely blind. But they sensitively
react to slightest movements of water; therefore any careless movement of lizard
will frighten them off. Having swum up closely enough, skinkfish makes sharp
movement sideways, creating whirlpool. Current of water tears larvae off from
stone, and lizard picks them up by mouth. But even if larvae have felt approaching
of the enemy and have hided between pebbles at the bottom, skinkfish will not
stay hungry: it simply will dig out bottom by powerful sharp-clawed forepaws
and tip of muzzle, and will seize larvae.
When the lizard seizes larvae, it faces with main difficulty for all creatures
living in this place: in its organism salt can get. And here the interesting
mechanism of protection was developed at the reptile. In soft palate skinkfish
has plica tightly closing gullet. It interferes to casual swallowing of salt
water. In mouth of lizard there is submaxillar throat pouch for catch, therefore
skinkfish may not emerge to surface too frequently - having filled this bag
with catch up to the full, lizard creeps out on stones and outside of water
eats caught larvae. But under water lizard all the same should open mouth and
to prevent getting of salt in blood through mucous membranes of mouth, they
are protected from brine by slime rich in protein with addition of fats making
this slime water-repellent.
From time to time skinkfishes creep out to coast to have a rest and swallow
caught larvae. Usually each lizard has stone chosen for this purpose, but outside
of water they quietly tolerate company of each other, and at times at one convenient
stone up to ten lizards gather. They do not try to take away each other’s catch,
because, swallowing caught larvae, lizard may not open mouth at all. Only having
crept out on land, it accurately contracts throat, squeezing out water from
mouth. Sitting on stone, skinkfish from time to time raises head, squeezes
out from throat pouch by effort of its ring muscles caught larvae one by one,
and swallows them.
On land skinkfish moves hardly: it pushes body by movements of tail, because
hinder legs of reptile are rather weak, and pelvis is attached to backbone
by flexible and extensible ligaments. Such feature of anatomy permits to skinkfish,
however, to avoid some difficulties which wait for reptiles in this salty world.
Skinkfishes do not lay eggs – they are live-bearing reptiles. Pairing of skinkfishes
takes place on land: it excludes salt getting in organism of female through
oviducts. Before pairing males arrange courtship demonstrations: they lift
heads upwards, inflate throat pouches and shake them. Thus skin on throat of
the lizard becomes bright red because of extended blood vessels. When the female
comes nearer to chosen male, showing by lowered head the submission, he twists
around of her body, and the fast pairing occurs.
Pregnancy of the female lasts about three months, and then she gives rise to
unique young on land. Young skinkfish is practically a copy of the adult reptile;
its length makes approximately half of body length of mother. To bear and to
give rise easily to such large posterity extensible connection of pelvis and
backbone helps to the female. Birth of youngs occurs 2 times per one year:
during pairing there is a fertilization of several ovocytes from which only
one develops, and other thus are “inhibited”. After birth of first young the
second ovocyte starts to develop.
After birth of second cub other zygotes do not collapse: they survive through
the most adverse season, but their destiny is not determined yet. If the female
will not receive visual signals from other males, at “spare” fertilized ovocytes
there is a chance to develop to high-grade youngs. In such way the population
can restore, basically, from few fertilized females. Presence of other males
causes changes of hormonal level of the female, and “spare” zygotes collapse.
But at the same time new ovocytes ripen, ready to fertilization.
Tatzelwurm,
giant horned skink (Scincodraco tatzelwurm)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Skinks (Scincidae)
Habitat: Southern Europe – the southern slopes of the Alps, the northern extremity
of the Mediterranean Basin to Asia Minor.

Picture by Alexander Smyslov
At the end of the human epoch, the skink family lost part
of the species, especially endemic and local ones, but the species that survived
the “anthropogenic crisis” later gave rise to a new variety of skinks. Even
specialized forms have appeared among them: one
of their species lives in a salt-covered Mediterranean basin. To the north
of this survival champion a giant representative of the family – tatzelwurm
– lives.
This species is a descendant of one species of Chalcides genus. The warm climate
allowed it becoming much larger than its ancestors: the length of this reptile
is 80-110 cm, of which a blunted tail makes about a third. The color of the
skin is not bright: brown spots on a gray background, smaller on the head and
tail, larger on the trunk, stretched across the trunk. The throat, belly and
underside of the tail are dirty white. Unlike the ancestors covered with smooth
scales, tatzelwurm’s skin is largely ossified, with several longitudinal rows
of osteoderms stretching from the neck to the middle of the tail. Osteoderms
prominently protrude above it, forming solid bumps, and the two largest bumps
protrude on the head, resembling small “horns”. The eyes are small, yellow in
color.
Tatzelwurm is a bad runner – its legs are somewhat reduced: they are relatively
short and have only 3 fingers on them. Therefore, escaping from large predators
from among mammals, it curls up in a ring, hiding its unprotected stomach and
exposing a strong skin shell. If the predator continues to attack, tatzelwurm
fights it off, making sharp movements with its whole body, hissing, hitting
its head and biting. After biting the aggressor, the reptile clenches its jaws
on its body with a death grip and hangs, dragging after the aggressor when it
starts to run.
This reptile has two periods of relative rest. By winter, the tatzelwurm eats
off, storing fat in its tail, and lies dormant in a deep hole for about two
months. On the hottest summer days, tatzelwurm also runs aestivation for about
2-3 weeks. Individuals from the Balkans, as a rule, do without aestivation.
Tatzelwurm is a zoophagous creature: it feeds on large arthropods, terrestrial
mollusks and small vertebrates. Occasionally, this lizard can steal a harelope
cub or eat the afterbirth of a large mammal. Cannibalism is occasionally possible
in these reptiles, but usually the juveniles stay away from adults and try to
hide in narrow burrows and crevices between stones.
Breeding takes place in March-April. The male finds the female by scent, and
then simply holds her until the end of mating, wrapping his body around her
and squeezing her across the trunk with short hind legs. When two males meet
near one female, a fight ensues between them: they butt and hiss, opening their
mouths wide until one of them retreats. 3 weeks after mating, the female lays
5-7 large eggs. Two months later, juveniles 15-20 cm long hatch. They have relatively
large legs, and they look more like lizards, whereas an adult reptile looks
more like a snake, since the legs are less noticeable when moving. Young individuals
initially feed on invertebrates, and they switch to larger animal prey from
about the age of three. At the age of 5, tatzelwurm reaches sexual maturity,
and life expectancy is often about 40-45 years.
This reptile species was discovered by Nick, the forum member.
Hubal
(Tiligugu hubal)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Skinks (Scincidae)
Habitat: grasslands of Arabia.
Chalcids, like other skinks, lost part of their species in the human era due
to the ploughing and loss of habitats, and were unable to fully recover after
that. By the Neocene, chalcids represent a fairly conservative group of reptiles,
prone to limb reduction. Few ones go against the conservatism of this group
of lizards. These ones are a large tatzelwurm
(Scincodraco tatzelwurm) from the Southern European maquis, and hubal from
Arabia, the descendant of the ocellated skink (Chalcides ocellatus).
Hubal is a relatively large species among skinks in general: the length of its
robust cylindrical body is up to 65-70 cm, of which about a third is a thickened
blunted tail. The limbs are three-toed, but quite developed and actively involved
in movement. The coloration is dim – a combination of gray, brown and black
colors forming a marble pattern with a darker back. The ventral side is lighter;
a pair of longitudinal stripes stretches along the sides – from the shoulders
to the tip of the tail. A crescent-shaped spot on the head is always present
(hence the name: Hubal is the moon god of the ancient Arabs), facing backwards:
in light individuals it is dark, in darker ones it is light; also hard bumps
from enlarged scales develop in this place.
Hubal is a twilight animal, hiding in the sand or in thickets of plants during
the day and at midnight, appearing on the ground surface at dawn and sunset.
In the evenings, the reptile warms up and catches insects on large stones heated
by the sun during the day. On moonlit nights, the lizard hunts while there is
a moon in the sky. It is an exclusively zoophagous reptile, feeding on any small
prey – arthropods, mollusks and terrestrial vertebrates. Several individuals
gather near carrion and the remains of prey of predators. When they meet, these
lizards establish a hierarchy, rattling with modified scales on the sides of
their tails (a similar device existed in the common wonder gecko of the human
era) and nod their heads, displaying a sickle-shaped spot. The main importance
is its size: the brighter and larger it is, the healthier the individual is
– it allows them avoiding a fight. Usually animals confine themselves to displaying,
but individuals approximately equal in strength can butt, resting on each other’s
corneous tubercles. When attacked by medium-sized predators, hubal strikes them
with its head and bites, often clinging to the aggressor’s body with a death
grip and hanging on to it. The reptile tries to escape from larger enemies,
hiding in the grass or in burrows.
Reproduction takes place in the spring. Ready to reproduce, the male chases
the female, searching her by smell. Catching up with her, he rattles with his
tail, nods his head, puffs out his throat and displays his size and force. Accepting
his courtship, the female presses herself to the ground and moves her tail from
side to side, producing a faint rattle. It is an oviparous reptile species,
with up to 15 large eggs in a clutch; incubation lasts about 2 months. After
laying eggs in a hole in the ground, the female buries the clutch and leaves
it. Usually the juveniles hatch from 8-12 eggs, the rest die for various reasons.
Also, in the first months of life, up to half of the hatched juveniles die,
including due to cannibalism of larger relatives. The growth rate of hubal is
slow: sexual maturity comes only at the age of 4 years. Life expectancy is up
to 30 years.
This reptile species was discovered by Nick, the forum member.
Wedgehead
(Herpethotalpa caeca)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Skinks (Scincidae)
Habitat: Meganesia, tropical forests.

Picture by Fanboyphilosopher
Tropical forest is a community of plants growing on sand and
clay in literal sense. The soil layer in tropical forest is much thinner, rather
than in deciduous forests of temperate latitudes, therefore roots of tropical
trees penetrate into it not so deep. The underground fauna of tropical forests
is also poorer, than it is possible to expect for so productive community. Nevertheless,
various insects and their larvae, snails, spiders and worms are found in wood
litter of tropical rainforest. These animals represent a source of food for
larger underground inhabitants, and also for not numerous ground animals digging
wood litter in searches of food. One species of reptiles, which almost never
comes in sight, lives in wood litter of tropical rainforests of Meganesia. It
is wedgehead, large underground skink – the total length of adult reptile of
this species reaches 40 cm.
Appearance of wedgehead demonstrates its fitness to totally underground way
of life. The body of this animal has wedge-like shape ideally suitable for digging
in thickness of friable wood ground. Wedgehead does not build constant tunnels
like moles and other digging mammals do. It simply moves in thickness of the
ground near to the surface, and the tunnel falls off behind it. On the ground
surface the trace of creeping wedgehead is visible as a faltering not deep flute
on surface of ground. The behaviour of this reptile is very primitive – wedgeheads
are solitary animals and behave aggressively to their congeners, and courtship
before pairing has very formal character.
Forepaws of this reptile are reduced. On body surface only two strong short
claws from each side of body have remained from them, and in thickness of muscles
reduced in great degree limb bones lay. Hinder legs are advanced much better.
They are short, but well appreciable outside, with mobile joints, short feet
and strong claws.
The armored head of reptile is using for ground drilling. It has wedge-like
outlines; the lower jaw is shifted a little back and above it the strong snout
overhangs. Wedgehead pushes by hind legs, making its way, and moves ground apart
by head. Instead of scales on head of animal the continuous corneous cover constantly
renewing on forward part of head is developed. Eyes of animal lack of eyelids
and are slightly reduced. They are covered with continuous transparent corneous
pellicle. During the moult this cover is remewing in common with other parts
of skin, as if at snakes. Sight sense shows attributes of degeneration: reptile
is short-sighted and also does not discern colors. Colouring of reptile is dim
enough – it is lead-coloured with darker cross strips, which number and width
varies in great degree at different individuals. There are animals, which back
and head are covered with 20 – 25 thin and clearly distinct strips, and there
are animals on which back no more than ten pale strips are stretched.
Tail of wedgehead is short and almost immoveable. It serves as a place for stock
of fat and as a support at ground drilling. Its muscles are very strong, and
on the bottom surface large plates are advanced; their back edge is extended
like strong spike. Supporting by such tail against the ground, reptile moves
forward.
On the ground surface wedgehead feels like very uncomfortably and tries to be
dug as soon as possible. It moves with the help of pushes of hind legs and tail,
trying to find an eminence in order to drill in it by head and to be dug in
the ground. In case of predator attack this reptile prefers active defense:
wedgehead dug out from the ground coils by sharp movements, striking the enemy
impacts by body, and also bites it. Having seized in body of the enemy, reptile
hangs with death grip and continues making sharp movements, shaking and sticking
teeth deeper in wounds.
Wedgehead eats any ground animals it can overcome – worms, spiders, grubs, fly
larvae, small snakes and lizards. Because of secretive way of life it becomes
anybody’s prey very seldom.
It is a viviparous reptile – once a year female gives birth up to three large
youngs, capable to dig tunnels and to lead independent life at once. At the
third year of life they begin able to breed. Many young reptiles perish because
of cannibalism of adult individuals. Life expectancy of wedgehead reaches 20
years and more.
Spiny
skink (Tiliquosoma spinifera)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Skinks (Scincidae)
Habitat: Meganesia, savannas and shrub thickets.
After the extinction of placental predators in Neocene Australia, marsupials
quickly developed the ecological niches returned to them. Native herbivores
had a harder time – not all placental herbivores “left the scene” after the
human extinction. However, as in the rest of the world, with the warming of
the climate in Meganesia, a number of niches were occupied by reptiles that
increased in size. Here, among the herbivores, a descendant of the common blue-tongued
skink (Tiliqua scincoides) appeared. Many related species became extinct or
gave carnivorous descendants, but this ancestral species diverged into several
herbivorous and omnivorous species. The spiny skink is one of them: it is a
giant descendant of the common blue-tongued skink, one of two skinks exceeding
in length the modern species of the family.
The body length of this species is 150 cm along with the tail; the weight of
an adult is about 45 kg. The animal has a robust build, a wide trunk, a massive
head and a long movable tail. The main color of the skin is yellowish-brown,
the belly is lighter. Unlike most relatives that have not lost their legs, this
species has strong and straightened limbs, because they have to endure a significant
load – the weight of the reptile’s body.
The increase in body size in this species is associated with the peculiarities
of the diet. The spiny skink inhabits savannas and shrubby thickets, feeding
on vegetative food – rough grasses, roots, and branches of shrubs with leaves.
The animal digs out the roots with lateral movements of the head. Osteoderms
on the lower jaw are large and convex – they help to dig the ground. Swallowed
gastrolith stones help to digest food. The increase in body size is associated
with the need to have a very long intestine, which allows it to effectively
digest rough low-nutritious food.
The animal digs deep burrows or expands existing abandoned burrows of mammals.
In the hot midday hours, the reptile hides in the bushes or in the shade of
trees.
Since the legs of the spiny skink are quite short and it cannot run away from
predators, it has developed a good passive defense. It is made of bony spikes
that have arisen on the basis of osteoderms; their length is 12-15 cm. They
are located on the animal’s body in several longitudinal rows. The middle row
stretches to the base of the tail. The skink can erect and lower them. The reptile’s
tail is covered with rings of convex osteoderms. When attacked by small predators,
the skink goes into a counterattack, lashing its tail, hissing and turning sharply.
Such a blow of the tail leaves a scar on the predator’s body. When attacked
by larger predators like the marsupial
panther or the gladiator eagle,
the spiny skink clings to the ground, protecting its belly, and not every predator
manages to turn it over. At the attempt of doing it, the reptile fights back
with blows of the tail and erects all the spikes on the body.
This species is viviparous. Mating takes place in September-November. This is
the only time of the year when this animal is interested in something other
than food and predators. The rate of reproduction of this species is relatively
slow: the female brings offspring once every 2 years. Males find a female ready
to reproduce by scent and mate with her. If several males meet near her, a fight
begins: the opponents hiss and strike each other with their tails. The female
sometimes mates with the third male while the first two males fight, forgetting
about her. After 3 months, the female gives birth to 12-15 well-developed large
juveniles up to 20 cm long. Newborn ones lack thorns, but they appear already
in the first 2 months of their life. At the age of 5, young reptiles reach adult
size, and at 6 years old they are ready to mate. The spiny skink lives for 55-60
years.
This reptile species was discovered by Nick, the forum member.
Meganesian
green raptor (Prasinohaema hexadactyla)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Skinks (Scincidae)
Habitat: northern part of Meganesia, mountain rainforests.
Picture by Lambert
This reptile is a representative of the green-blooded skinks
genus and is the largest species of this genus. The length of an adult reptile
is about 1 meter. This species has an appearance typical for skinks: a muzzle
with a pointed nose, a weakly pronounced neck narrowing, and an elongated flexible
body with a long tail. The limbs are short and strong, well adapted for climbing
rocks and trees: the thumb on the front and hind paws is opposed to other digits,
strengthening the grip. In addition, there is a strip of “sticky” scales on
the tips and edges of the digits, equipped with a specialized fringe, like geckos
(this similarity is convergent). There are 6 toes on the hind legs – an additional
toe is short, equipped with a powerful claw, directed outward and provides a
firm grip of branches when acting together with the thumb of the foot.
Like its ancestors existed in the human epoch, this species accumulates the
green pigment biliverdin in its saliva (and to a lesser extent in the blood).
The saliva of this reptile has a thick consistency and resembles a green paste;
the pigment is filtered out of the blood and accumulates in saliva. Its concentration
is so high that the reptile’s saliva is poisonous to small animals. Also, several
mildly toxic poisons of a protein nature accumulate in saliva. The green raptor
has an element of warning coloration: “spectacles” of white color around the
eyes, clearly visible against the background of skin coloration. The eggs that
the female lays in burrows at the roots of trees or in spaces between stones
are greenish-yellow due to biliverdin, but are not poisonous. There are up to
5 large eggs in a clutch; incubation lasts about 2 months. After laying eggs,
the female guards the burrow, but not the clutch itself. Outside of the breeding
season, each reptile alternately uses several shelters located on the inhabited
territory. The care of the offspring shown by the female is interesting: she
feeds the hatched cubs on eggs (both fertilized and unfertilized). During the
feeding of the offspring, the female periodically lays eggs in a thinned eggshell
or simply in a filmy envelope, which is easy to break through with the weak
jaws of the juvenile. The remains of eggs attract small insects, which are also
eaten by young individuals. The Meganesian green raptor lives in monogamous
family groups (a couple and offspring up to about one month of age), less often
in polyandrous groups, including 2-3 males. Sometimes a group of males “shares”
two females living next to each other.
Sexual dimorphism is clearly expressed in this species. The female is larger
and more robust than the male, but the male is characterized by pronounced aggressiveness.
The coloration of the male and female also differs. The male has a bright green
color with transverse brown strokes on the back and at the base of the tail.
The female color is brown with black transverse strokes on the back and tail.
The mucous membrane of the mouth in animals of both sexes is blue-green. Defending
itself, the animal opens its mouth wide and displays its coloring by sticking
out its tongue. When attacked by a predator, the lizard bites it, clings to
its body and secretes abundantly the saliva, which gets into the wounds.
The Meganesian green raptor is zoophagous. This reptile eats bird eggs and nestlings,
as well as skinks of other species, young monitor lizards and snakes. Young
individuals feed on insects.
Sexual maturity in this species occurs at the age of 3 years, and the life expectancy
is 25 years or more.
This reptile species was discovered by Dmitry Korshin, the forum member.
Darter
chameleon (Belonoglossaurus aviphagus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Chameleons (Chameleontidae)
Habitat: rainforests at the east of Madagascar island.
Representatives of chameleon family live mainly in Africa though in human epoch
their separate representatives lived in Asia, and even in Southern Europe. But
these lizards have reached the remarkable variety at Madagascar, in conditions
of island isolation. In ice age, when the areas of tropical rainforests had
been reduced, the variety of chameleons had decreased, and many of their species
had died out. In Neocene the remained species of chameleons had started to evolve
actively, and among them the species occupying new ecological niches had appeared.
Among chameleons small and medium-sized animals prevail. And at Madagascar in
human epoch the tiniest of all chameleons, representatives of genus Brookesia,
lived. The size of the largest chameleon of human epoch did not exceed 60 cm.
But, if it’d meet the largest chameleon of Neocene epoch, it should have the
most pitiable consequences for it. The biggest chameleon of Neocene, darter
chameleon, reaches the length of about one meter though tail makes half of it.
And the menu of this reptile includes not insects, but small vertebrates: birds,
mammals, tree frogs and lizards.
It is difficult for such giant to remain unnoticed on tree branch, but darter
chameleon has inherited from the ancestors the skill of camouflage and has developed
it up to refined perfection. Head and back of this lizard are covered with small
knobs and outgrowths imitating a bark of tree. On head of adult reptile the
corneous outgrowths imitating knots of tree stick: one is sticking out forward
on the nose, and two ones directed upwards and in sides are located above eyes.
At different individuals they have a little bit different shape, and even the
different size. Masking is supplemented with scalloped outgrowths on legs, hiding
in part a shadow cast by the hidden lizard.
Color of skin of this chameleon is changeable, and easily varies from light
grey up to green or dark brown with very diverse patterns. In rest darter chameleon
has a pattern of two colors: the top of body and head have soft green color,
and the bottom part of sides, tail, stomach and paws are grey. “Horns” on head
of lizard are also grey. Border between grey and green colors is sharp, but
rough, with strong individual deviations. On the general background of the body
small white and yellowish spots are scattered, imitating spots of lichens on
bark.
Like the overwhelming majority of chameleons, this species lives in crones of
trees, despite of its size. Paws and tail of darter chameleon are prehensile,
fingers and toes on paws are opposed as two groups: two fingers (toes) against
three ones, as at all lizards of this family. Usually this reptile arranges
ambush on large branches, having nestled against the bark of tree, or having
hidden among epiphytic plants. Darter chameleon leads diurnal way of life; at
night it hides in hollows or among rich branches.
The main hunting weapon of all chameleons is their tongue capable to extend
far forward. But at small species on tip of tongue the sucker is developed,
with which help it is possible to keep small creature only. Darter chameleon
catches more powerful prey and strikes it with its tongue. Tongue of this reptile
may extend forward to half-meter distance. But it is only a half of affair to
get prey by tongue. The main thing is to keep it, and tongue of darter chameleon
is perfectly adapted for this purpose. The tip of its tongue has turned to spear
instead of sucker: on it the corneous spike is developed; it kills prey or does
not give it an opportunity to escape. If prey is only wounded and got away,
but has not flied out and has not fallen down, chameleon may pursue it, finishing
with some more “shots” of tongue. Inside the tongue the longitudinal beam of
elastic fibres stretches, giving to it durability and serving for retraction
of tongue with prey pinned on it.
Like many species of predators, this animal is territorial: each reptile actively
protects the fodder site from competitors. Darter chameleon tries to frighten
off contenders and small predators (for example, mammals), inflating air bags
inside the body and rocking on all four paws. At these moments its skin gets
crimson colouring with white spots on sides. The lizard accompanies threating
display with loud hissing, blowing air out between densely compressed lips.
But this threatening is mostly illusory: animal never uses the tongue for protection
against large contender, being afraid to injure it. It only can bite the opponent
by strong jaws. And the bite of darter chameleon is very strong.
In courtship season males turn especially intolerant to each other: they actively
drive away each other from the female and combat, “butting” by horns. If fight
becomes especially persistent, males can bite each other. The winner male starts
to walk around the female, rocking on straightened legs. Its body gets crimson
color with white cross strips and spots on head. As a sign of submission the
female turns pale and nestles against the branch.
Darter chameleon is an egg-laying species. Female goes down on the ground only
to lay eggs. In clutch of this species there are up to 10 eggs like small chicken
eggs in size, covered with soft leathery shell. Embryos start development in
female’s organism, and to the moment of egg laying they are approximately half
developed. Sometimes female leaves eggs in deep hollow of tree trunk, digging
them in dust. “Ripening” of eggs lasts three weeks more.
Young chameleons at hatching are up to 15 cm lond (half of their length thin
tenacious tail makes). They eat insects, gradually including inth their diet
larger animals – young frogs, nestlings of small birds and cubs of forest rodents.
Young lizards reach the typical size of adult individual at the age of 6 – 7
years. But they reach sexual maturity earlier – females bring posterity for
the first time at the age of 4 years, and continue to grow after becoming sexually
mature.
Three-lobed
petalobrookesia (Petalobrookesia trilobata)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Chameleons (Chameleonidae)
Habitat: tropical forests of eastern Madagascar, forest canopy.
Human economic activity has caused great damage to the tropical forests of Madagascar:
their area has been drastically reduced, and many species of animals and plants
have become extinct. In the Neocene, the restoration of tropical forests was
accompanied by the appearance of new species occupying peculiar ecological niches
in the renewed world. One of these species is a dwarf species of chameleons,
three-lobed petalobrookesia, an inconspicuous inhabitant of the canopy of the
tropical forests of Madagascar.
This species is a representative of a peculiar genus of dwarf chameleons, which
differs from its relatives in food preferences: petalobrookesias lick the flowers
of epiphytic plants and get the insects from them, and pollen and nectar are
used as an extra nutrition. Three-lobed petalobrookesia is a reptile about 6
cm long, with a large head and a short tail. The head and trunk are deep, the
limbs are thin. This dwarf reptile seems somewhat larger because of the unusual
body shape: along the spine there are three very long leathery blades – on the
head, trunk and tail. Their height is about 3 cm; their upper parts are expanded
compared to the base.
The unusual shape of the body is combined with the characteristic coloring.
The body of three-lobed petalobrookesia is not brightly colored: it is dark
green with transverse light green veins. The coloring of the body is complemented
by the bumpy texture of the skin. Contrasting with the dull coloration of the
body, the skin lobes on the head, back and tail are very brightly colored: they
are dark red with a yellow upper edge and individual yellow specks on the surface.
Body coloration may vary within certain limits. The body can change its color
from light green to brown, and the blades from brownish-yellow to reddish-brown.
In this way, this chameleon camouflages itself on the inflorescences of orchids
and other epiphytic plants from predators and possible prey. In case of danger,
the animal curls into a ring, mimicking a bud opening. The enemies of this species
are ants, large wasps, large predatory beetles, less often birds and small snakes.
Usually the reptile crawls slowly among the flowers, from time to time thrusting
its tongue into the flower’s fauces and licking small insects that get inside
it. Occasionally, the animal crawls over to other thickets of epiphytes. During
the migration, its color becomes as dull and inconspicuous as possible.
Sexual dimorphism is very weakly expressed: the male is about 1/5 shorter than
the female and slimmer than her. The male finds the female by smell; the mating
ritual is reduced exclusively to mating itself. There is no seasonality in reproduction,
only at the edges of the range when the dry season comes, the reptile suspends
egg laying. The rate of reproduction is relatively slow: the female lays one
egg every three weeks. In the absence of copulation, the female lays parthenogenetic
eggs, which develop normally. Incubation lasts about 4 weeks. A young individual
with a length of only about 11 mm has only weakly pronounced ridges at the place
of future outgrowths. The skin color is dark brown, changing to green with age.
Outgrowths begin to appear at the age of 2 months, with a length of about 4
cm. Their color is initially light green, by the onset of sexual maturity (at
the age of 7 months) it changes to a typical one for the species. Life expectancy
rarely exceeds 3 years.
In the tropical forests of Madagascar, there are similar species of Petalobrookesia
genus:
Spure-bearing
petalobrookesia (Petalobrookesia plectrifera) is an arboreal reptile
up to 65-70 mm long. It differs from the previous species in the shape of outgrowths:
the outgrowth on the tail is narrow and thin, resembles a spur of flower at
some plants. This outgrowth is colored dimmer than the “petals”: it usually
has a greenish-white color. The “petals” are yellow with a greenish base and
streaks mimicking veins; the reptile’s body is green with brownish specks on
the back.
Coral
petalobrookesia (Petalobrookesia corallina) lives on tree trunks near
“epiphyte gardens” and mimics the fruit bodies of fungi in its color and body
shape. The body length is about 75 mm. The body color is dark; the outgrowths
are orange with white tips; each is divided into several lobes. Sometimes there
are short parallel outgrowths on the body, repeating the color of the main ones.
The rate of reproduction is similar to the previous species, but at a favorable
time it can lay two eggs at once.
Prickly
agama (Echinagama spinosa)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family Agamas (Agamidae)
Habitat: steppes to the west from Fourseas.
Agamas are one of the most successful families of lizards. In Old World their
representatives during Cenozoic superseded representatives of other groups,
adapting to various conditions of inhabiting: from deserts up to tropical woods.
High abilities to adaptation have permitted these reptiles to survive successfully
during an ice age.
From eastern coast of Fourseas (Garagumy beach) up to bush wastelands of Mongolia
steppes, overgrown by grasses, were stretched. Here there are not many rivers,
and fauna is presented by mainly drought-resistant species. The interesting
species of agamas – prickly, or hedgehog, agama – lives here.
When this lizard is quiet, it almost by nothing differs from other representatives
of family. It is the lizard of approximately half meter long including tail
and large head. On body and lateral faces of tail scales are lengthened, having
turned to spikes. Scales on shoulders are especially long – up to 3 cm. By
shape scales resemble three-edged blade: they are flattened, and on the external
side longitudinal edge passes. These scales are colored dark-brown, being especially
visible on background of grayish-yellow skin. They grow by longitudinal lines,
forming on the top side of body of animal regular pattern. On stomach spikes
are much less, and skin is colored lighter, being of almost white color. At
the male throat is colored bright red and spike scales are white. At the female
color of throat does not differ from color of stomach. The bright throat pouch
is using by male in courtship demonstrations.
Prickly agama like its neighbours is afraid of birds and large mammals, and
hides from them in shelter – cracks in stones and holes of rodents. But it
is not afraid at all of snakes being enemies of many lizards of such size,
because it is able to protect from them originally.
Having seen snake, lizard takes menacing pose: it rises on straightened legs
as the toad, and puffs. It has some air bladders connected with lungs, and
special valves allow to force in them air at an exhalation. As a result the
reptile quickly inflates, and its visible size is increased more than twice.
Covering its body lengthened scales stick out, turning to the terrible weapon.
Puffed up, lizard keeps at predator’s movement. If the distance too decreases,
it swings by tail in sides. From birds and mammals prickly agama uses one more
way of protection: being strongly puffed up, it slightly “blows off”, and thus
utters loud whistle through densely compressed jaws. If enemy does not recede,
lizard beats it by prickly tail, or makes lunge, striking it by spikes of sides
and shoulders.
Courtship games of prickly agama are similar by something to displaying at
man-o-war birds from far Holocene. Males get on separately sticking up branches,
and puff throat pouch, becoming appreciable from afar. Each male protects the
territory from contenders.
During rain season (spring) the female makes 2 layings: in first one there
are up to 10 - 12 eggs, in second one (2 – 3 weeks later) - no more than 10
eggs. The posterity hatches in second half of summer. They spend winter in
holes of rodents, fencing off from restless neighbours by several ground “fuses”.
The young reptile reaches maturity at the age of two years.
Leaf-headed agama (Compressisaura angustissima)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: humid subtropical woods at southern and western coasts of Fourseas.
During an ice age the rests of heat-loving European flora and fauna appeared
“compressed” at the narrow strip between cold glacial tundras stretching from
the north, and dry salt deserts at the place of Mediterranean Sea dried up
when strait of Gibraltar had closed for ever at movement of Africa to the north.
There were only few “oases” where the rests of Holocene flora and fauna dragged
out a miserable existence among new species appeared in glacial epoch.
In early Neocene the flora and fauna of Europe had gone through the rebirth:
when glaciers had thawn and at Earth extensive shallow seas had appeared again,
the climate became more humid and steady. The huge Fourseas lake isolated from
the ocean, formed in early Neocene, softens the heated breath of Mediterranean
hollow and supplies with moisture the mysterious world of subtropics of South-East
of Europe. This world is isolated from tropics of Asia and Africa by mountains
and salt desert, therefore it was occupied mainly by descendants of local fauna
and flora. Representatives of heat-loving southern fauna are not very numerous
in woods of Balkans and Caucasus: there are only separated species of small
vertebrate animals, insects and few plants had reached here over mountains.
In warm and damp woods at the southern and western coast of Fourseas reptiles
are numerous. One of their representatives is the leaf-headed agama, the small
tree-climbing lizard living on tree trunks.
Lizards represent easy catch for various predators. The majority of their species
has no poison and is too small to protect itself actively. Therefore till the
evolution process lizards either studied quickly to run, or developed various
ways of masking for protection against predators. The leaf-headed agama shows
one of such kinds of protection.
Body of this reptile is strongly compressed from sides and high: it hardly
exceeds 2 cm in width at length of the reptile about half meter. The height
of body of this lizard amounts about 10 cm; on its back the skinny crest with
slightly cut up edge grows, on head there is also thin jagged comb. When the
lizard freezes sitting vertically on bark of tree, it is similar to the exfoliating
piece of bark. One more advantage of this skinny crest is an opportunity to
be warmed faster: in the morning lizard creeps out on tree trunk and turns
sideways under beams of rising sun.
The fantastical body shape is supplemented with cryptic colouring which varies
over a wide range: from green with narrow white strips simulating leaf nerves
up to light grey or brown with longitudinal lines imitating color of tree bark.
At this reptile even eyes are disguised – the iris of the eye is colored yellow
with tiny brown speckles. Such colouring is successfully combined with any
variant of skin colouring.
Paws of the leaf-headed agama are thin and have tenacious sharp-clawed toes.
To keep on tree trunk stronger, at back edge of forearms and on the internal
side of shins spike-like scales develop. When the lizard sits on tree bark
upwards a head, they are stuck in bark and the reptile keeps on trunk without
the special efforts. The tail is long (it accounts more than half of general
length of animal) and thin; its tip is extended. It serves as the balance weight
when the leaf-headed agama jumps from branch to another. Also the attacked
lizard can whip small enemies by tail. The basis of tail has some small skinny
outgrowths continuing a crest on back at the top side.
The leaf-headed agama is the diurnal reptile. Usually it sits on tree trunk
as precisely as possible imitating color of its bark. This lizard is a predator
and eats insects and spiders, but can attack small wood frogs and ravage bird
nests. Usually it expects while suitable catch will come nearer to it itself,
catches it by short throw, quickly swallows and freezes again not to be noticed
by larger predators. It spends night in shelters – cracks of tree trunk and
in narrow cracks under scaled out bark. The hidden lizard supports by paws
against bark and tree trunk, and tenaciously keeps by claws. Only few nocturnal
predators are able to pull out the leaf-headed agama hidden this way.
Features of constitution impose restrictions to some vital processes of this
lizard. In thin body of the female many eggs can not form at once, therefore
the female of this species lays eggs in small portions - 2 – 3 ones at once.
The egg of leaf-headed agama is very much extended and covered with limy shell.
Till first minutes after laying the shell is soft and sticky, then on air it
hardens quickly. This feature permits the reptile to lay eggs in well-protected
shelters – on tree trunk under bark where this animal usually hides at night.
The pasted egg keeps strongly on tree trunk and is inaccessible so to the majority
of predators.
In connection with such breeding feature the courtship season at leaf-headed
agamas is strongly dragged out: it takes place since late spring up to middle
of an autumn. Courtship display of the male happens rough: having felt a smell
of the female ready to pairing, it begans the display – the male rises on extended
legs, puffs an extensible throat pouch of brightly-yellow color with black
spots, frequently “nods” a head and expects for the appearing of the female.
If the female is not shown for too long time, male starts to follow its tracks.
When the male sees the female, his demonstrations become even more active:
he starts to utter abrupt whistling sounds, blowing air from throat pouch through
slightly opened lips. When near one female two males appear, between them fight
is possible; in this situation contenders bite each other, beat by tails and
try to push each other away from the female. But usually males avoid fight
and simply try to make impression of the bellicosity against each other. They
puff throat pouches, whistle and whip tails, trying to strike the contender.
The male had losed fight in any case has an opportunity to breed: till the
peak of breeding season in forest females ready to pairing meet frequently.
Readiness of the female for pairing happens about each 10 days, right after
the next laying. For one season the female may lay more than 30 eggs: it is
even more, rather than it happens at other lizards living in the neighbourhood.
The incubating of eggs lasts about 2 months, but last autumn layings develop
much longer: their development is delayed for all winter. From such eggs young
animals hatch approximately in middle of spring. Adult reptiles hibernate in
deep tree-trunk hollows. Their aggression to each other is reduced in an autumn,
and they gather for wintering by groups of some tens of animals. In winter
animals are inactive, but approximately at the end of March when it becomes
relatively warm, they one by one leave the common shelter and occupy territory
suitable to life.
Rainbow agama (Variolepis iris)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamids (Agamidae)
Habitat: thickets of large-leaved plants on riverbanks, flooding areas of forest
in continental part of Southeast Asia.
Restoration of tropical forest areas in Southeast Asia became the stimulus
for intensive speciation among small species of forest reptiles. Snakes and
lizards of various sizes and colours are typical inhabitants of Neocene forests.
Some species of these animals have freakish shape and bright colouring. These
creatures are ideally “dissolving” in their native inhabitancy, despite of
brightness of colouring, and represent its organic continuation. Tree-living
rainbow agama, the descendant of common butterfly lizard (Leiolepis belliana)
of Holocene epoch, is those one.
It is a typical representative of family – medium-sized reptile: total body
length including thin tail is about 50 cm, weight is about 300 g. This reptile
swarms up trees very good, using strong hooked claws, and is the often visitor
of tree crones. Its head is rather deep and rather narrow, with the dulled
conic muzzle. Colouring has determined a name of this lizard: on its body and
tail white and pink spots of various sizes and shape are scattered on emerald-green
background. This colouring helps rainbow agama to mask among inflorescences
of tropical trees: the lizard simply climbs on inflorescence and freezes among
opened flowers. Trees of tropical forest as a whole blossom the year round,
but the flowering period of of each separately revealing species of trees may
be short. Therefore as far as trees are deflowering reptiles of this species
are compelled to move to the next trees. Their flowers frequently differ in
colouring, but the rainbow agama has ability to change color of skin. In rate
and accuracy of these metamorphosises it considerably concedes to such masters
of camouflage, as chameleons and cephalopods. Reptile needs about two hours
to change the color of background and spots. The basic colouring can vary from
bright green up to very dark, almost black. And pink spots if necessary can
turn to crimson on any background. Also the size of spots varies a little.
The camouflage of reptile is supplemented by crest stretching along the back.
It is made of irregular-shaped skin blades which improve imitation of plant
and “break off” a contour of animal. The largest blades, up to 3 cm long, grow
from nape up to shoulders of reptile. On tail skin blades get the shape of
tiny denticles. They are a little bit darker than other parts of body – it
is an adaptation for the best warming. In early morning rainbow agama turns
body perpendicularly to sun rays and the crest is quickly warmed.
Rainbow agama has the expressed sexual dimorphism: male is larger than female,
though weighs less than it, and has higher crest.
This reptile prefers for life a site including several trees of different species
bound by lianas, usually not very far from water. Escaping from the enemy,
rainbow agama can simply jump in water, having drawn in paws and having closed
eyes. Swinging by tail in air during the falling from tree, it has time to
turn headfirst, and enters into water almost without splash. Under water reptile
turns paws in sides and slowly emerges, having holded breath.
Rainbow agama is omnivorous one. It eats fruits, flowers and large insects.
It is most active in the morning and at the sunset, avoiding afternoon heat.
Sometimes after rain it keeps activity almost till all daytime. In hot weather
rainbow agama becomes languid. It prefers to hide in shadow of inflorescences
of trees where it is more difficult for finding out to any casual predator,
and eats vegetative food – flowers. It hunts for insects in the evening more
often.
The breeding season begins at the end of spring and proceeds till about three
months. At this time female succeeds to do up to two clutches. In courtship
season lizards become much more aggressive to each other. Males actively struggle
for females, displaying to each other might and physical shape. They hiss,
widely opening mouths, whip tail and actively restrict the contender from branch
or tree trunk, pushing it by side and delivering impacts of tail. If there
are two opponents equal in force, they can establish relationships with the
help of bites. During the courtship combat and courting for the female male
of rainbow agama gets especially bright and saturated colors. Standing in front
of the female, male actively “nods” the head slightly turned aside in order
to show its crest.
Soon after pairing female lays 5 – 7 eggs in dust at the bottom of deep hollow.
In 3 weeks young lizards hatch. They look absolutely not like adult individuals:
they do not have crest, and colouring represents irregular cross strokes of
grey color on green background. They become sexually mature at the age of 9
months, but get completely adult colouring approximately to the age of one
and a half years. Life expectancy in nature usually is about 8 – 9 years, but
separate individuals live up to 11 years.
The idea about existence of this species of reptiles was proposed by Nick, the forum member.
Patch-skinned
agama (Allopogona hemialata)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: deserts of southeastern Meganesia.
In the Neocene, agamids represent one of the most prosperous lizard families.
They are found everywhere in the Old World and Meganesia, showing adaptability
to life in a variety of habitats, from tropical rainforests to deserts. In the
desert areas of Meganesia one of the unusual representatives of agamas lives
– the patch-skinned agama, a descendant of the central bearded dragon (Pogona
vitticeps) of the human era. It is a relatively large reptile, reaching about
50 cm in length; half of this length is the tail, in which fat reserves store.
This reptile has a short, robust body and a large head. The patch-skinned agama
has a blunted muzzle and strong jaws – it is a predator. It differs from other
species by the unusual structure of the skin: from the sides of the body to
the wrists of the front paws, a membrane stretches on a cartilaginous “frame”,
making the lizard look a little like a flying squirrel (hence the name). This
membrane is an adaptation for thermoregulation: in the morning, the patch-skinned
agama crawls out onto a hill open to the rays of the sun, stretches its hind
legs and tail, and stands on this “tripod”, spreading its front paws to the
sides and stretching its “wings”. To improve the heating, it changes the color
of the membrane to black. In the heat, the lizard, on the contrary, folds its
“wings” and changes the color of the membrane to white. The membrane tissues
are abundantly penetrated with blood vessels, which contribute to better heat
exchange; a line of large pointed scales stretches along the edge of the membrane.
The color of the whole body of the patch-skinned agama is camouflaging sandy
yellow with an indistinct pattern of darker transverse stripes. The tip of the
tail is black; the lower part of the body is yellowish-white.
Camouflage coloring helps the animal to escape from birds. The patch-skinned
agama escapes from small mammals and large lizards by inflicting bites. The
mucous membrane of the mouth is colored bright red, and the tongue is white.
Defending itself, the reptile opens its mouth wide and hisses, warning about
the ability to defend itself. When attacked by a large terrestrial predator,
this reptile flees. It runs mainly on very muscular hind legs, which are free
of webbing. In this feature, it is similar to the frilled lizard extinct to
the Neocene. The patch-skinned agama digs burrows for protection from predators
and daytime heat; they are shallow and are located under bushes of desert plants.
Unlike many other lizards, this species lives in small harem groups consisting
of a large male, several adult females and young of the current year. Eggs are
laid in a specially dug nest hole, usually under boulders that flatten daily
temperature fluctuations. There are usually up to 20 eggs in a clutch. This
species is characterized by caring for offspring: the female guards the clutch
until the hatchlings appear, attacking enemies and inflicting strong bites.
As a rule, adult reptiles do not hunt their own young living in the vicinity
of the burrow. However, some “vagrant” individuals are cannibals. Intraspecific
communication in the group is carried out by means of sound signals: whistling,
hissing and rustling of tail scales. During the mating season, tournaments for
the female take place among males, but they proceed in a very peculiar way:
reptiles rear on their hind legs, spreading their forelegs to the sides and
stretching their membranes, and begin pushing each other with their breasts,
trying to knock down the opponent. These duels are strictly ritualized, since
a lizard with a torn membrane can die from blood loss. The winner chases the
opponent from his territory and bites his tail. Seeking the attention of the
female, the male walks around her on outstretched legs and inflates his throat,
which gets a bluish color at this time. In dominant males, the throat is dark
blue.
Depending on its age, patch-skinned agama hunts insects or other lizards, using
its superiority in speed, and also feeds on berries. It hunts lizards in the
morning, when they are still sluggish. Often, this lizard stands in ambush on
its hind legs, holding onto a branch or a grass stalk with its claws in order
to get off the ground faster and rush to run, having detected prey or danger.
In addition, this reptile likes to lure large spiders with the movements of
its contrast-colored and rustling tip of the tail.
Sexual maturity comes at the age of two years; life expectancy is about 20 years.
This reptile species was discovered by Dmitry Korshin, the forum member.
Orchidonia
(Orchidonia stumpkei)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamids (Agamidae)
Habitat: tropical forests of India, Indochina.

Picture by fanboyphilosopher
Tropical areas represent a habitat of numerous reptiles. Here
these creatures, not capable to support a high body temperature independently,
find optimum conditions for life. And the riches of fauna and flora permit reptiles
to evolve roughly, developing very freakish strategies of survival. Tropical
forest canopy is a residence of various small reptiles like lizards and snakes.
The strange species of tree-climbing lizards belonging to diverse agamid family
lives in forests of Southern and Southeast Asia. It is an orchidonia; this reptile
developed an unusual way of hunting for insects. It is the lizard of gracile
constitution, up to 25 – 30 cm long; about half of total lenght a long tail
makes. Paws of this reptile are well advanced, long and thin, with tenacious
fingers and toes armed with claws. The head of orchidonia moves on mobile neck
and can turn to the angle of 180 degrees. Body is covered with smooth scales,
and on tip of tail and on internal surface of paws numerous corneous bristles
grow. They permit this reptile to be fixed firmly on smooth stalks of epiphytic
plants among which reptile waits for prey.
The most remarkable feature in anatomy of this lizard is the presence of several
leathery outgrowths around of head. On sides of head two symmetric outgrowths
with rounded edges grow, on top of head there is one unpaired outgrowth with
the pointed top, and on the lower jaw three medium-sized lanceolate outgrowths
are developed. All of them stretch on thin elastic gristle cores which mobile
bases are supplied with muscles. Due to it lizard can open all outgrowths at
its own choosing, imitating the flower of orchid plant. Masking is supplemented
with rounded cross outgrowth on nose.
But masking of reptile would be incomplete without exact imitation of color.
Orchidonia has surpassed even chameleon in this skill. Color of its head varies
from greyish-white up to crimson red, orange and citreous. This reptile is able
to create patterns of spots and strips on head outgrowths, imitating colouring
of petals of orchid, among which flowers it is hidden at present. This time
its body gets green color. In case of danger orchidonia is hidden among roots
of epiphytic plants or on bark. At this moment its body gets yellowish-brown
color with speckles and strokes, imitating tree bark, and outgrowths lay along
the body and turn rusty-brown with dark border, looking like withered leaf.
Skills of colouring change are connected to the diet of orchidonia, which includes
various insects. Usually reptile traps them, having hidden in inflorescence
of orchid. Thus tail of orchidonia is twisted around of flower-bearing stem
and helps the reptile to keep on plant. The smell of flowers masks a certain
imperfection of its masking, and reptile catches insects which may even settle
right on its head in searches of food.
Sexual dimorphism at orchidonia is expressed only in body shape of male – it
is a little bit larger, rather than female, but has more graceful constitution.
The courtship season at this species lasts the year round, but males become
especially active during the monsoonal rains. They combat for the female, showing
each other stretched “petals” plicas. Thus they hiss and make attacks against
each other, trying to bite “petal”. The torn places, however, regenerate quickly.
During the courtship combat pattern on male’s “petals” is especially bright
and contrast. Usually it represents spots and dark border on light background.
The female lays up to five small eggs in shelter under bark of tree or among
dust in tree-trunk hollow. The egg laying repeats 3 – 4 times per one year.
The incubation lasts up to about six weeks; eggs do not endure drying, because
they absorb a significant part of water from environment through soft shell.
Young lizards have no outgrowths on head and eat insects, finding them under
bark and among moss thickets. At the age of 2 – 3 months at them “petals” begin
to grow, and they pass to hunting on flowers of plants. One-year-old reptiles
are already capable to breed.
Life expectancy makes no more than 6 years.
Stick
agama (Bacillisaura tenuissima)
Order: Squamates (Squamata), Suborder: Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitats: savannas of Northern Africa (the plain to the north from Saharan Nile),
high herbage.

Picture by Alexander Smyslov
Family Agamidae was one of the most progressive families of
lizards during Holocene epoch. They successfully developed both forests and
savannas, and also desert areas. The latter quality allowed these reptiles to
endure successfully ice age when the considerable areas covered earlier with
forests turned into deserts. During the period when the extreme climate of glacial
epoch was replaced by mild and favorable climate of Neocene, agamids evolved
actively and formed the new species adapted to dreat diversity of habitats.
In savannas of North Africa stretched at the place of Sahara, among thickets
of high grasses, one of unusual species of this family lives – fragile and graceful
stick agama. It is a small species of lizards – the length of its body including
tail reaches only 20 cm. This agama has very thin body and rather long, pointed
head with prominent eyes. Paws are rather long, prehensile and adapted for climbing.
Tail is thin; it makes about a half of general length of this reptile.
Coloring of body of stick agama is cryptic: it represents thin brown longitudinal
strips on yellowish background of body, like a color of dry grass. One dark
strip stretches across an eye, masking it. During rain season, when the fresh
grass grows, color of background changes from yellow to green, and this lizard
keeps possibility to mask. Pupil of the eye is slit-like and horizontal (thus
at the perched lizard the crack of eye pupil is placed vertically, improving
the circular view). Eye iris is brown.
In habit of life stick agama resembles in some details the corresponding insect.
It lives on plants in the same way, in everything hoping for the protective
coloring and shape. This lizard keeps on stalks of graminoids. In rest it perches
along the stalk and keeps the body in vertical position. In such position lizard
can easily climb to the next stalks, and even makes jumps sideways, pushing
at the same moment by front and hind leg of the same side of body. In case of
danger it tries to mask, hides under leaves or nestles against stalks and stays
motionless until the last minute. At the extreme necessity the reptile seeks
safety in flight, but at the first possibility tries to hide again.
Stick agama eats only insects, catching them with a short rush when they creep
up rather close. This lizard drinks, licking the dew accumulating on stalks
of grasses in the morning. It can not drink for a long time, receiving enough
amount of moisture from food. Being a predator, it protects a fodder site from
relatives, pushing them by side out from territory borders. Males conflict only
with males, and females with females. As a whole the territories of individuals
of opposite genders can overlap completely.
Courtship season at stick agamas stretches to almost all year round, interrupting
only before a drought which happens on northern border of their area. Males
notify neighbors that the territory is occupied, uttering the characteristic
sounds similar to crash of broken twig. When the female comes closer to the
male, it determines her readiness for pairing by a smell. If the female shows
submission (it takes up the position on stalk lower than male), the male starts
courtship ritual. It opens throat bag on which bright red middle fold bcomes
visible, and starts to shake the head up-down. He climbs down to the female
and keeps side by side with her. After fast pairing animals leave each other.
In clutch there are only 2-3 rather large eggs of very elongated shape, but
the egg laying can repeat every month almost all the year round, interrupting
only for the period of severe drought. The female lays eggs in the middle of
large bush of graminoids, in depth where they will be protected from casual
damage. The incubation lasts till about a month. Young lizards don’t differ
from adults in habit of life, only keeping on plants near bushes where it is
easier to take cover from hot sun and it is possible to find small insects.
At one-year-old age they become adults.
The cannibalism is not characteristic for these lizards.
Translated by Alexander Smyslov.
Knave-tail
lizard (Fraudatia candicauda)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: the outskirts of the Mediterranean lowland, Asia Minor, the Middle
East to the Persian Ridge.
The evolution of some groups of living organisms can take the most unexpected
courses, depending on which feature will ensure reproductive success in a particular
habitat. A striking example of the “whims” of evolution was the appearance of
an unusual lizard – a knave-tail lizard – in arid areas of eastern Eurasia.
By itself, the knave-tail lizard is a rather nondescript insectivorous reptile
that successfully disguises itself among the surrounding landscape, but if necessary,
it can lift its tail over its head with a sharp movement and, thanks to some
simple adaptations, turn into one of the most noticeable creatures in its habitat.
The total length of the reptile is about 30 cm, of which about half is on the
tail. The head and body of the knave-tail lizard have a rather ordinary appearance:
a wide head with a pointed snout, small eyes, a wide trunk and strong legs with
tenacious claws that allow climbing on rocks and plants. The upper part of the
body is covered with small scales; there are small shields on the head. The
color of the body is sandy yellow; a darker stripe stretches along the spine.
The throat and belly are yellowish-white.
The tail of this reptile contrasts in structure with the body and head and is
an original example of mimicry. Along the edges of the tail large flaps of keratinized
skin grow, capable of both pressing against the body and rising up almost perpendicular
to the spine due to the action of small muscles and blood vessels that can fill
with blood a lot. The inner surface of these flaps is brightly colored, and
along their posterior edge long hair-like outgrowths (modified scales) grow,
that exaggerate the imaginary size of the animal during its display. The color
of the colored areas varies from pale blue to orange and scarlet, and is fixed
genetically. At the tip of the tail, under one of the pairs of these outgrowths,
blood vessels and connective tissue form bubbles that can quickly fill with
blood. The surface of these bubbles is covered with smooth elastic skin with
an ocular pattern. When swelling, these bubbles very accurately mimic the eyes,
and the smooth underside of the skin flaps and “eyelashes” further exaggerate
their size. Such a display, complemented by a loud hissing, allows the knave-tail
lizard scaring away many predators. In addition, the knave-tail lizard uses
its tail during the mating season – thus, natural selection favors the owners
of the most striking and visually complex tails. When the displaying is not
needed, the skin flaps lie on the tail like a roof, hiding bright spots, and
the lizard differs from other species only by a slightly more extended tail
and “hairs” sticking out along the edge of the tail, which are noticeable only
when the reptile is not moving.
Mating takes place immediately after emerging from the short hibernation, and
in the south of the range – after spring rains. During the mating season, males
arrange tournaments, displaying each other tails with raised and spaced flaps
of skin. Standing side by side to each other, males begin to raise and lower
rhythmically these flaps, becoming either very noticeable or almost invisible.
When things get heated, males begin to walk on outstretched paws, raising their
tails up and displaying their false “eyes”. In some males, under the influence
of hormones, another pair of bubbles is formed on the tail behind the false
“eyes”, also nimicking the eyes.
Knave-tail lizards are oviparous reptiles. In midsummer, the female lays up
to ten eggs, burying them in moist soil, usually at the roots of large plants.
In the laid eggs, the embryo is already quite well developed, and further incubation
takes about a month. In the first year of life, young lizards have tails of
a typical appearance, with only small serrations along the edge. In the third
year of life, with the coming of sexual maturity, the tail acquires an appearance
typical for this species. Life expectancy is about 15 years.
This reptile species was discovered by Mex, the forum member.
Umbrella-headed
agama (Membranobarba dracocephala)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: steppe and semi-desert regions of Central Asia.
The agamid family is one of the most widespread groups of lizards in the Old
World. During the human epoch, some species of this group suffered from human
activity and became extinct; another part of the species disappeared due to
changes in the biosphere at the turn of the Holocene and Neocene. However, the
remaining species were quite diverse, so they did not give up their positions
in the Neocene. In due course of evolution, various species of these reptiles
developed peculiar adaptations for survival, often giving their owners a bizarre
appearance. The umbrella-headed agama is one of the peculiar species of the
Asian herpetofauna of the Neocene epoch.
This reptile species is a lizard about half a meter long, of which a thick powerful
tail covered with large scales takes about a third. This species has a large
flattened head on a movable neck, a flexible trunk and well-developed legs.
The lizard is able to run quickly on erect legs, as well as climb bushes and
low trees and dig holes. The background color of the body is sandy-brown, and
the belly is yellowish-white.
A characteristic feature of this species is the bizarre decoration on the head:
red skin folds grow along the edges of the mouth and lower jaw in adults, covered
with small corneous spikes along the edge and bordered by a lighter stripe with
a large number of black spots. While the lizard is at rest, these skin formations
are folded and pressed against the neck and shoulders, and their cryptically
colored upper edge covers the bright markings, so the catchy coloring is not
visible. If necessary, the reptile stretches them, accompanying the displaying
of bright coloring with a loud hiss. In males, these skin folds are especially
well developed. During the fights, the males open their “collars” and display
them to the opponents. In the same way, individuals of this species react to
the attack of predators. In such situation, the threat is reinforced by strong
bites. Umbrella-headed agama escapes from large predators by running away.
In the spring, during the breeding season, the umbrella-headed agamas arrange
ritual duels, standing on their hind legs and uttering a loud hiss. At such
moments, males stand in front of each other on their hind legs, keeping their
balance with the help of the tail and opening the skin collar, which takes bright
and contrasting color in the mating season. If the opponents are not inferior
to each other, they begin to attack each other, while maintaining an upright
posture. While courting the female, the male also displays her opened collar,
holding the body vertically, and trembles slightly at the same time.
This species is territorial; disputes over territory are also accompanied by
a display of skin collars.
The umbrella-headed agama feeds mainly on insects and small vertebrates – lizards
of other species, rodents, bird eggs and nestlings. Up to a third of the diet
consists of plant food – flowers and fruits of shrubs and herbs. This species
often ravages rodent burrows.
Living in a seasonal climate, the umbrella-headed agama falls into a hibernation.
Before wintering, the lizard stores a large amount of fat in its tail, which
makes it look like a club in autumn. This reptile overwinters in burrows, which
it digs out on its own. The depth of wintering burrows reaches 2 meters or more.
This species often arranges collective wintering shelters – reptiles of various
ages gather in the same wintering hole. Young individuals usually overwinter
in the burrows of adults, and reptiles reached maturity dig their own burrows
or chambers in a common burrow.
The umbrella-headed agama is an oviparous species; the female lays eggs 2 times
a year: up to 6-10 eggs in spring, and additionally 2-4 eggs in mid-summer.
In some isolated populations from mountainous areas, there is a transition to
ovovivipary – they reproduce only once a year, and 3-6 eggs develop in the female’s
body almost until the offspring are ready to hatch. From such eggs, the juveniles
hatch 2-3 days after they are laid.
Sexual maturity comes at the 4th year of life; life expectancy reaches 25 years.
The idea of the existence of this reptile species was proposed by DarkSp17, the forum member.
Shy
toadhead agama (Phrynocephalus crypticus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: steppes of Central Asia, east of the Fourseas.
Not all toadhead agamas survived the human era and the ice age. Irrigated agriculture,
habitat destruction and climatic fluctuations have destroyed the most vulnerable
local species. However, the surviving representatives of the group had gone
through a new flourishing during the ice age at the turn of the Holocene and
Neocene, when the deserts of the Old World expanded significantly. Later, when
the climate became more humid and the area of the steppes expanded, the toadhead
agamas experienced a crisis period. However, in the Neocene they are still a
fairly successful group of lizards of the arid regions of the Earth. And while
the umbrella-headed agama (Membranobarba
dracocephala) is a rather unusual representative of their group, the shy
toadhead agama is a more typical species, one of the common species of the Asian
steppes. It is a descendant of the sunwatcher toadhead agama (Phrynocephalus
helioscopus) of the human epoch.
It is a relatively small reptile: the total body length including the tail is
up to 17.5 cm; at the same time, the tail itself is 40% of the length of the
animal. The general coloration of the skin varies from yellowish-gray to grayish-brown;
the shade may change slightly in a couple of days when moving to a new area.
Darker spots are scattered on this background. The underside of the body is
pale yellow or white. The shy toadhead lizard is a very cautious animal; in
case of danger, it stays motionless until the very last. Coupled with the coloration,
this behavior pattern often hides it from predators and potential prey – arthropods
and small snails, although the lizard does not disdain the predators’ leftovers.
The shy toadhead lizard often freezes on a stone, a grass stalk or a shrub branch,
somewhat resembling an African “grass fish” skink
having a similar lifestyle. At night, the shy toadhead lizard hides in rodent
burrows, or in its own burrows, that it easily digs out with its claws. There
are always several emergency shelters on the territory of each individual. Especially
deep, up to half a meter, is a wintering hole dug under a stone or among bushes.
Perhaps this is the only animal that a farmer
hamster can tolerate on its territory: in the summer and autumn period,
a shy toadhead lizard eats insects that harm wheat.
However, the reptile still winters in its own burrow, because this rodent can
easily eat a lizard that has settled down for the winter in its burrows.
The breeding season begins in late April – early May. At this time, shy toadhead
lizard males get bright spots on the head and sides and begin to behave less
covertly. They climb up on the branches of shrubs and large stones, arranging
courtship displays. Males raise their tails and swing them, attracting females,
and arrange battles with each other, showing brightly colored skin folds at
the corners of the mouth. Females can make 2 clutches per season (from May to
July), numbering usually 2 eggs per clutch. About two months after laying, young
lizards hatch. Sexual maturity comes at the age of one year; life expectancy
is up to 12 years.
This reptile species was discovered by Nick, the forum member.
Emela-ntouka
(Emela ntouka)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: Sub-Saharan Africa, tropical forests, shrubs.
The Neocene is an era when old ecosystems and food chains collapsed, and new
ones took their place; at this time, the descendants of the surviving species
of the human epoch often mastered a different ecological niche. The extinction
of mammals of all sizes in Africa allowed some reptiles to become larger than
their ancestors were, and warming allowed them to displace mammals in the small-
and medium-sized class. Most of these new species are predators: snakes, monitor
lizards and their descendants – water monitors. However, among the large reptiles
herbivorous species exist also; one of the largest among them is the African
emela-ntouka, one of the agamid lizards.
Emela-ntouka grows up to 2 meters in length and its physique resembles American
chuckwallas, although they belong to the iguanid family and the similarity between
them is exceptionally convergent. Like the chuckwallas, emela-ntouka eats various
plants – herbs, flowers, leaves and fruits of shrubs: everything it can reach
from the ground. Unlike smaller reptiles, it is unable to climb trees as well,
as American iguanas – its physique is too robust for it. However, it can stand
up on its hind legs, like monitor lizards, and eat vegetation located above
its head, pulling branches to itself with its front paws. During feeding, the
reptile relies on the tail; its length is about 45% of the total length of the
animal. Defending itself from the enemy, the reptile strikes its legs with the
tail.
The head on a short neck is massive, with a blunted snout and strong jaws. The
teeth are leaf-shaped, of cutting type – the reptile chaffs food with them,
but does not chew it. Emela-ntouka can inflict strong bites on the enemy: teeth
leave deep cut wounds on the enemy’s body.
One of the characteristic features of the animal’s appearance is a single conical
horn growing on its nose; hence the name – it is the name of an African cryptid
with a long horn on its head. The horn grows on the bridge of the nose, its
base stretches from the nostrils to the eye sockets. Emela-ntouka is not so
much protected by it from predators, as establishes intraspecific relations:
males fence with them for females, avoiding using their jaws. The emela-ntouka
females also have horns, but they are shorter. While moving in the undergrowth,
the reptile pushes the vegetation aside with its horn. The reptile can run fast
for short distances and swims well, so it often escapes from enemies in the
water.
The emela-ntouka is colored not brightly – usually it is of various shades of
gray, but there are also darker, almost black individuals: it is not a separate
subspecies, but just a color morph. The belly and throat are slightly lighter.
The eyes are small, the iris is yellow; the pupil is round.
Emela-ntouka does not demonstrate a particularly complex behavior; it is a species
tolerant of its relatives, and quite often several individuals feed nearby to
each other. However, it is not a herd, which is characteristic for more highly
organized vertebrates: there are no social ties and hierarchy in such group.
In case of danger, each lizard defends itself from a predator on its own. Only
a rich source of food makes these reptiles stick together.
There is no expressed breeding period at this species, and the emela-ntouka
females also do not look at their eggs: they bury them in the ground in a fairly
dry place and leave forever. There are up to 20-25 medium-sized eggs in the
clutch. Juveniles hatch after a few months; they are usually 10-15 cm long.
They lack horns on their noses; this feature appears only some time later, when
they reach maturity. The main part of a diet of young animals is small invertebrates;
they also search for and eat fallen tree fruits. They are able to eat the fruits
of plants of Solanaceae family, which are poisonous to warm-blooded vertebrates.
The enemies of these lizards are birds of prey, snakes and large lizards, especially
monitor lizards. Large individuals can be attacked by predatory mammals. If
emela-ntouka is not eaten being a juvenile, then it can live for several decades.
This reptile species was discovered by Bhut, the forum member.
Mortal
agamoraptor (Agamoraptor atrox)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: oases of the Mediterranean lowland.
Representatives of the numerous agamid family in the Neocene show a considerable
diversity and adaptability: some of their species are found even in the Mediterranean
lowland, the place characterized by extreme natural conditions. They have developed
these habitats due to the peculiarities of their physiology: reptiles are able
to live in a regime of strict economy of water, which is obtained mainly from
food.
One of these agama species has become partially bipedal – it is a mortal agamoraptor,
an inhabitant of the mountain oases of the Mediterranean lowland. Walking on
two legs helps it to reduce contact with the hot surface of stones and salt
in the midday sun. The body length of this reptile is about half a meter; half
of the total length is the balancer tail. The hind legs are twice as long as
the front ones, with a narrow elongated foot, used for running; the front paws
are tenacious, of the anatomy typical for lizards. Agamoraptor climbs rocks,
runs and jumps well. The head is triangular in shape, with blunted muzzle and
folds of skin on the sides, opening in case of danger. The eyes are large and
surrounded by “eyelashes” – thin scales that diffuse bright sunlight. The color
is of light shades and mottled: light brown with black and dark brown speckles.
Agamoraptors live in oases, both in the forest and on rocks. They usually stay
in areas with a mixed landscape, where areas of vegetation and rocks alternate.
Activity is all day and night round, but in extreme heat reptiles hide in shelters
(in burrows or in the forest floor). At night, agamoraptors can settle, running
from one oasis to another. If the day finds this lizard on its way across the
salt plain, it looks for temporary shelters and keeps in the shadow of blocks
of salt, continuing its journey after sunset.
The basis of the lizard’s diet consists of small animals: insects (agamoraptor
licks off the smallest of them with its tongue), spiders, other arthropods,
snails, small lizards, juvenile snakes. Agamoraptors often ravage the nests
of local birds and eat eggs and chicks. If a lizard finds carrion, it can stay
near it for a long time, eating dried meat and insect larvae. In fact, the mortal
agamoraptor is one of the top predators of the oases, which in its adult condition
should only be afraid of snakes and birds of prey. Adopting a threatening pose,
the reptile opens the folds of skin on the sides of the head, trying to scare
off the predator. If the threat does not help, the lizard runs away. Out of
danger, agamoraptors move on 4 legs, like all common lizards. Chasing its prey,
the agamorpator is able to run on its hind legs for several minutes.
Courtship games begin in the second half of autumn and include male-to-female
races, fights between rivals and demonstrations of straightened folds of skin
on the head. At this time, in males, they turn bright red with a black spot
in the middle. After mating, the male leaves the female, no permanent pair is
formed.
This species is ovoviviparous; fertility is up to 3-4 large well-developed juveniles.
The offspring develops in the mother’s body the whole winter and the beginning
of spring. Delivery takes place in a shelter and proceeds very quickly. Having
freed from the egg shells, the juveniles run away. The female hides in a shelter
for some time, her feeding behavior is suppressed; after recovering from delivery,
the female can easily eat her own offspring. The diet of small agamoraptors
consists of insects and lizards, including the juveniles of the salt
gecko.
Sexual maturity comes at the age of 4 years; life expectancy is up to 20 years.
This reptile species was discovered by Mamont, the forum member.
Bonnacon
lizard (Bonnaconosaurus foetidus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Agamas (Agamidae)
Habitat: forests and forest-steppes of southern Europe, between the Alps and
the Mediterranean basin.
Agamids represent an extraordinarily diverse and successful group of Old World
lizards. Like other groups of reptiles, they have suffered significant damage
from human activity, and a number of their species have become extinct – mainly
local forms and inhabitants of tropical forests. In the Neocene, the descendants
of the surviving species gave rise to a great number of new forms, which acquired
incredibly diverse defense mechanisms and strategies in the process of evolution.
Grass snakes of the Holocene epoch were able to scare off predators by producing
a protective substance with an unpleasant odor from the cloacal glands. In the
process of evolution, a species with a similar protective mechanism appeared
among the agamas.
According to legends, the bonnacon is a mythical bull-like creature that bombarded
its enemies with fiery excrement. Like its mythical prototype, the bonnacon
lizard raises its tail when threatened, and shoots a stream of pungent, fetid
liquid out of the cloaca. The liquid is very sticky and difficult to clean out
of the wool, keeping an unpleasant smell for a long time. The disturbed reptile
can make up to three “shots”, after which it must restore the supply of these
secretions within a day.
The lizard itself is very small, reaching only 15 cm in length; tail makes slightly
less than half of its total length. The animal has a massive round head on a
short neck, strong and relatively short legs, and a wide tail. The body has
an inconspicuous gray coloration with small black and brown specks from above;
the underside of the body and the area under the tail are creamy-white.
This reptile species is a peaceful, timid creature. Bonnakon lizard feeds on
insects and small vertebrates – rodent cubs and chicks of birds nesting on the
ground and in bushes.
The breeding season falls in early spring, while the spring vegetation supports
the existence of fodder insects. Males acquire a courtship dress – the head
turns white, a black “mask” appears around the eyes and on the muzzle. At the
end of spring, the female lays 4-6 eggs, burying the clutch in a warm and moist
place – between the roots or in the forest litter. The juveniles hatch in the
middle of summer. They hide in shady shelters, escaping from the hot sun, and
are most active in the morning and evening. In the morning, they willingly lick
the dew that appears on the stones, and in the evenings they hunt insects.
Sexual maturity comes at the age of 2 years. Life expectancy is 13-15 years.
This reptile species was discovered by Ilia, the forum member.
Pangolin
lizard (Neocordylus armatura)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Girdle-tailed lizards (Cordylidae)
Habitat: Southern Africa, savannas of various types.
Human epoch is characterized by change of borders of natural zones. At this
time there was a reduction of areas occupied with forests and grasslands, but
areas of deserts extended. It took place because of irrational economic activity
of people. In historical epoch the significant territories occupied earlier
with productive natural communities, had turned to deserts and became unsuitable
for human life and for agriculture. But in these places rather large number
of animal species was kept. The greatest distribution of deserts was typical
for Africa to the north of equator, and for Near East. In South Africa extensive
territories also were occupied with deserts – Namib, Kalahari and Carroo. In
these deserts rich fauna of reptiles evolved and was kept. One of Neocene species
of South-African fauna is pangolin lizard.
Pangolin lizard is a small reptile of strong constitution, with very powerful
and clawed forepaws. It is the descendant of Holocene girdle-tailed lizards
(Cordylus) characteristic for South Africa.
This species has a little changed in comparison with the ancestor: the length
of adult individual seldom exceeds 20 cm. This lizard has rounded head covered
from above with strong corneous plate, flattened body covered with cross lines
of large pointed scales (hence the name), and thick armored tail covered with
scale rows overlapping each other, and similar to pine cone. This tail serves
for reserving of fat stock, and in courtship season is used for displaying to
females – they more willingly are coupled with successful and full males which
tail is full of fat. Protecting itself from small predators, lizard beats them
by tail, as if by buldgeon.
Living in small groups – 1 – 2 males (one of which is obvious subdominant) and
5 – 6 females – these yellowish-brown reptiles something resemble miniature
anteater monitors from deserts of Meganesia. Peaks of activity of pangoline
lizard fall to morning and first half of day when their forage – various small
and medium-sized invertebrates – has not come to the senses yet after night
catalepsy, and is easy for catching. Lizards are heated in beams of rising sun
on stones or walls of termitaries, and then start search of food. Forage of
these reptiles includes termites, ants and other insects and invertebrates.
Due to strong jaws these lizards easily chew strong armours of beetles.
By way of life pangoline lizards divide in two groups – “tramps” and “homebodies”.
“Homebodies” are lucky beggars managed to settle near large ant hill, or, even
better, near termitary which is exposed periodically to their attacks: due to
thick skin pangolin lizard can rather painlessly suffer relatively large number
of stings of ants, termites, and others small, but poisonous insects. It breaks
a surface of termitary by forepaws, and simply licks off running out insects.
“Tramps”, on the contrary, have no constant residence. Almost all of them are
young males did not manage to secure a constant colony of fodder insects. Therefore
all the time they are occupied with searches of their own colony, and are compelled
to spend much more time for search of daily portion of food – any insects or
arachnids, which they can eat. This is rather dangerous occupation, because,
despite of hardness of skin and camouflage colouring, pangolin lizards are regularly
eaten by various animals, from larger lizards, and up to large spiders and scorpions,
attacking young reptiles. Besides they constantly should search for shelter
to wait in shadow hotter second half of day, and to spend the night in safety.
At the middle of dry season pangolin lizards have a new care: the courtship
season begins. Homebody males get on any eminences – for example, on tops of
the “own” termitaries, and start to hiss loudly, puffing and showing bright
throat having violet or yellow spots. Basically, this representation is intended
not for females, but for tramp males of this species, which would like to expel
the owner from its house after long fight. During the courtship tournament two
males push each other by breast, creeping on top of termitary, ant hill, stone
or other eminence. Two or three fights usually are enough to reveal the weakest
one, which leaves a place of struggle, conceding to the winner territory and
females, which are immediately fertilized.
This species of lizards is ovoviviparous. At the end of summer, in the beginning
of rainseason, females give rise to 2 – 3 live youngs. Among females of pangolin
lizards there are no “tramps”. If any female does not live in constant colony,
it may be only very young female had left to search for new place for residence.
Adult females also can be “tramps”, but only when the colony of insects in which
they ate, was exhausted and lost, that happens, but too seldom. On the contrary,
almost all males leave a place of their birth to the beginning of the next dry
season, and do not come back to it even to the beginning of courtship season,
preferring to win a place for life somewhere at the distance. In such way pangolin
lizards succeed to avoid inbreeding.
Birth of young pangolin lizards is dated to the beginning of rainseason, because
at this time at termites, ants and other social insects of South Africa courtship
flights begin. In same time other insects, grasshoppers and locusts, burst.
At this time in nature there is a greatest variety of food for young pangolin
lizards, opportunities for growth of their colony are greatest, and aggression
of adult individuals is reduced. But further, somewhere in second half of rainseason,
all young males and some females leave a place of the birth, being banished
by old male and sometimes by other females.
This species of reptiles was discovered by Bhut, the forum member.
Legless
chamaesaura (Chamaesaura serpentina)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Girdle-tailed lizards (Cordylidae)
Habitat: arid savannas of South Africa.
Chamaesauras, inhabitants of southern and eastern Africa, suffered in the human
era during the plowing of savannas and other economic activities, but in small
numbers survived at the border of the Holocene and the Neocene. Their adaptability
to life in arid areas helped them survive during the ice age, when the area
of forests decreased. However, even after the end of the ice age, they have
changed very little, remaining a kind of Holocene relic. The legless chamaesaura
is a reptile that demonstrates the strongest changes in the appearance of this
group of animals. It is a descendant of the Cape snake lizard (Chamaesaura anguina)
of the Holocene epoch.
The body length of this reptile is 1.5 meters, of which more than half is a
brittle tail. Ossified rings of scales grow on the tail, forming a protective
device: if the reptile is hiding among the stones, they make it difficult to
take the animal from its shelter. The coloration of this animal is nondescript,
gray-brown; the belly is lighter. The serpentine body is slightly compressed
from the sides. The legs, reduced already in the ancestral form, disappeared
almost completely – from the front legs only rudiments of the pectoral girdle
under the skin remained, and from the rear ones there are two tubercles on the
sides of the cloaca orifice. The legless chamaesaura feeds on large invertebrates,
sometimes hunts small vertebrates, and spends the night in burrows or crevices
between stones. The favorite habitat of this species is thickets of bushes or
tall grass, where it can easily hide from enemies.
The courtship season in this species falls at the end of the dry season. Males
chase females, guided by the scent left by an adult female. When two males meet,
a power struggle begins: both rivals push with their sides and heads, trying
to push each other away from the female. The winner male chases the female and
mates with her in the shelter, fixing himself with the help of his tail and
holding her with the curves of his body. The legless chamaesaura is an ovoviviparous
species. 1.5 months after mating, the female gives birth to 8-12 thin worm-like
juveniles, depending on the length of the female. The juveniles become sexually
mature at the age of 2 years. Life expectancy is up to 30 years.
This reptile species was discovered by Nick, the forum member.
Lizardboa
(Sauroboa trachycephalus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Lateral fold lizards (Anguidae)
Habitat: humid subtropical forests at the coast of Fourseas – from the Balkan
up to Caucasian Peninsula.

Picture by Alexey Tatarinov
Ice age at the boundary of Holocene and Neocene had become
serious test for European herpetofauna. Glaciers held down by continuous armour
the most part of Europe had forced heat-loving reptiles to recede to the south,
or had simply exterminated their populations. Therefore in early Neocene the
herpetofauna of Europe had began to form practically from zero. Some widely
settled European species had survived at the south of former area, and had returned
to habitats of their forefathers in new quality. Among such species there was
one representative of blindworm family, the glass-snake (Ophisaurus apodus).
This species of snake-like lizards had successfully survived in agrarian landscapes
of human epoch, and its descendants had taken a dominant position in herpetofauna
of European subtropical forests.
The descendant of this large lizard had turned to true giant over 3 meters long.
By its powerful constitution this reptile is similar to small boas and hence
is named lizardboa. As against to true snakes which are strict predators, lizardboa
is omnivorous one. This reptile feeds on various large insects and small vertebrate
animals, and also willingly eats soft fruits fallen from trees. In appearance
of lizardboa there is one more feature distinguishing it from snakes - at this
lizard there are mobile eyelids, and it can blink. Also at lizardboa the cervical
narrowing very characteristic for snakes is poorly expressed.
The skin of this reptile is covered with small scales forming a continuous cover.
Colouring of lizardboa helps to mask in underbrush, waiting for prey approaching.
The basic background of colouring is yellowish brown. On it a spots of brown
color are scattered making three faltering longitudinal lines – along back and
on each side. On tail spots merge to continuous strips with rough edges. The
head of reptile is covered with set of small irregular-shaped spots.
Lizardboa leads passive enough habit of life. In day time it hides in wood litter
or under bushes, waiting for approaching of small animals suitable for eating.
Attacking them, this reptile strikes a blow by head which is covered with strong
corneous armour; impact stuns prey, or at once kills it. Protecting against
large predators, this lizard applies similar tactics: it rises on tail like
cobra, loudly hisses, and if the enemy does not recede, strikes sharp and strong
impacts by head.
At the end of the summer and in autumn lizardboa is actively fattened, gathering
the stock of fat necessary for successful wintering. Per bumper-crop years the
diet of this reptile may more, than half consist of berries and fruits. Before
wintering reptile does not eat, exempting intestines from the rests of food.
Few large reptiles expand far to the north. Only the huge lizard gila-dragon
(Helodermopsis monstrosus) lives in North America almost so far at the north.
In conditions of seasonal climate reptiles face to the necessity to go through
winter period adverse for life, running into more or less long hibernation.
Shortly before an autumn cold snap lizardboas begin to search shelters for wintering.
Lizardboa winters in burrows which digs itself, or occupies ready burrows of
various mammals. Usually few animals can resist to this reptile if it has chosen
their burrow for wintering; therefore lizardboa usually does not have problems
with search of winter habitation. In areas where there are karstic caves with
narrow entrances, these reptiles gather for wintering by tens. At this time
lizardboas are inactive and rather tolerant to relatives.
In spring, when stabile warming comes, lizardboas leave shelter in which they
had spent cold season, and start to feed actively. At this time they behave
extremely aggressively, and the adult reptile can even eat young growth of own
species. It is connected to vital necessity – after wintering the exhausted
reptiles as soon as possible should restore the physical shape and start breeding.
The courtship season at these reptiles begins approximately one month after
the end of hibernation. Males colour very brightly: their colouring becomes
very contrast. In courtship dress male is almost black-and-white with slight
yellowish shade on white areas of body. Males become very active. They creep
in forest, expelling contenders from the territory, and mark borders with odorous
liquid. If two males about identical in force meet, they arrange noisy combat,
coiling bodies and biting each other. Having met the female, male tries to press
her body to the ground and to turn on back in order to couple with her. If the
female is not ready to breeding, she tries to turn to dense ball preventing
male to unwrap her body. If the female is ready to pairing, the pair of reptiles
coils and coupled during several hours. At this time male is very aggressive,
and attacks even animals much larger than its own size.
Lizardboa belongs to egg-laying reptiles. During two months after pairing in
organism of the female up to ten large eggs develop. During the pregnancy the
female becomes cautious and keeps mainly at warm forest clearings where sometimes
some individuals of this species gather at once. Such behaviour is connected
with the fact that eggs of lizardboa pass the first stage of incubation in organism
of the female. Then the female lays them, digging in forest litter. The further
care of posterity is not characteristic for this species. The incubation proceeds
till about one month, and young individuals about 30 cm hatch. They differ from
adults in cross-striped colouring. The diet of young ones consists of insects
and berries.
Lizardboa becomes completely developed at the age of 4 years, at length up to
170 cm. Life expectancy of this reptile makes more than 60 years.
Scaly
bird-snatcher (Paraophisaurus aviphagus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Lateral fold lizards (Anguidae)
Habitat: southeastern North America.
During the existence of humankind, many large animal species were subjected
to significant anthropogenic pressure. Among reptiles, crocodiles, sea turtles
and large tortoises, as well as large lizards and snakes have suffered from
human activity. The latter, however, suffered less than crocodiles, but despite
this, many large snakes failed to withstand the anthropogenic impact like direct
hunting and habitat destruction, and became extinct. However, at the same time,
there were much more species of small reptiles in the world – tortoises and
freshwater turtles, snakes and lizards. Among lizards, there were many legless
forms from several families, and lateral fold lizards in particular. Being lizards
by their nature, these reptiles lost their limbs and began to move like snakes.
They quite succeeded in the struggle for existence – in the subtropical forests
of the Balkans, a large omnivorous lizardboa
(Sauroboa trachycephalus) appeared, and on the other side of the Atlantic
Ocean, in equally humid forests of North America, its distant relative lives
– scaly bird-snatcher, descending from a certain American species of the genus
Ophisaurus.
It is a fairly large reptile, vaguely similar to a snake. Individuals can grow
up to 2 meters long, but usually scaly bird-snatchers are no longer than 120-150
cm. The body of this reptile is covered with a continuous layer of thick, tightly
fitting scales with a characteristic longitudinal fold of stretchable skin along
each side. The main background color is greenish-yellow, sometimes with irregular
spots of a lighter shade. This coloring allows the reptile hiding among the
vegetation. The young of the scaly bird-snatcher is darker, with a predominance
of gray-green shade in color. Coloring helps the scaly bird-snatcher not only
to avoid enemies, but also to lie in wait for its prey: unlike the lizardboa,
the scaly bird-snatcher is an exclusive predator. In terms of lifestyle, the
scaly bird-snatcher also differs from its European counterpart – this reptile
leads an arboreal lifestyle in adulthood and hunts birds, ambushing them or
catching them in nests, at an altitude of more than 10 meters above the ground.
To improve the grip on the support, on the lower part of the animal’s body the
scales end with sharp edges facing backwards. The main prey of this reptile
is birds of various sizes. Usually this reptile preys on small birds, but sometimes
a pigeon-sized species can fall its prey. In addition to birds, the scaly bird-snatcher
attacks arboreal rodents and bats. The front teeth of this reptile are pointed;
palatine teeth, well developed in the front of the mouth, also help to hold
its prey.
The mating games of these reptiles begin with the rainy season. At this time,
the males begin to behave aggressively – they chase each other and try to expel
the opponent from the inhabited territory, inflicting bites on each other’s
tail and sides. A characteristic element of mating tournaments is a head fight,
when opponents “box”, striking each other hard blows with hard snouts. During
the breeding season, males darken, and the throat and front of the belly turn
white in them. Females are more phlegmatic and tolerant of their own kind. A
female ready for copulation attracts males with a specific smell, and mates
with the winner of the mating tournament. In summer – from June to August –
scaly bird-snatcher females descend from trees and lay eggs – from 16 to 34
small elongated eggs in one clutch – in heaps of dead foliage or among dead
wood. After two months, young animals hatch. Like other lizards, they immediately
lead a completely independent life. Within an hour after hatching, they move
away in search of food – mainly other reptiles, small amphibians and invertebrates.
However, they grow quickly and by the next spring, they pass to an arboreal
lifestyle and begin to hunt almost exclusively for birds.
The arboreal lifestyle of this lizard is caused by the abundance of terrestrial
and semi-aquatic snakes in its habitats. Many snakes are able to kill and eat
even an adult scaly bird-snatcher. At the same time, various small birds forming
the basis of the diet of this species also live in trees, away from land enemies.
In the conditions of the tropics and subtropics, these reptiles gain an advantage
in the struggle for existence over predatory mammals, whose homoiothermy does
not give a tangible gain in the struggle for existence in a hot climate. The
scaly bird-snatcher is able not only to lie in wait for birds on branches, but
also to climb into hollows for nestlings and eggs.
A young animal reaches sexual maturity in the second year of life. The maximum
life span of scaly bird-snatcher is 15-18 years.
This species of reptiles was discovered by Bhut, the forum member.
Monai
(Notovaranus monai)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Teiids (Teiidae)
Habitat: Pampas of South America.
As a rule, in the human era, large reptiles suffered more from human activity
than smaller ones. There were only some exceptions, and one of them was the
large South American common tegu (Tupinambis teguixin). It was human activity
that saved this species: lizards were easily tamed and often became pets. This
circumstance, as well as its wide distribution, helped the populations of this
species to survive both in the jungle and in open spaces. In the middle of the
Neocene, the tegus became more diverse and colonized a variety of habitats,
giving rise to a number of new species. Monai is one of them, inhabiting the
Pampas with separate shrub thickets (the name comes from the name of the deity
of open spaces in Guarani mythology).
This species is the South American counterpart of the Old World monitor lizards
in size and lifestyle. The body length of an adult is about 2.5 meters, of which
40% falls on the tail. The weight of the animal reaches 55 kg. The skin is covered
with smooth scales; the background color of the body is gray-green, but it can
change: when moving to the thicket of bushes, darker stripes may appear on the
body in a couple of days. The best ability to change color is demonstrated by
young individuals; old reptiles lose this ability. The hind legs of this reptile
are longer and stronger than the front ones: sometimes the monai pursues prey,
running a short distance on its hind legs, but prefers to ambush and catch small
animals with a short rush from its shelter.
The teeth of this lizard are cutting and blade-like; during the bite, they cause
deep incised wounds. There are lots of bacteria on the reptile’s teeth, so after
an unsuccessful monai’s attack, animals often get sick and may die.
Monai feeds on small animals, occasionally daring attacking old and weak deermaras.
However, much more often it eats their cubs, rodents, birds (including young
hunting herons), amphibians
and reptiles. This species is undemanding in terms of food and does not disdain
carrion and the remains of prey from other predators. If necessary, the reptile
can drive away small predators like canines or small cats from their prey.
The monai is a territorial animal; each individual marks its territory with
scratches on high places – on cactus stems and tree trunks. Rivals are violently
driven to the borders of the territory; occasionally fights for territory happen
between these reptiles. In old monai individuals, fingers may be bitten off
– this is a result of intraspecific contractions. Competitors of this species
are small relic representatives of crocodile order – land
caimans (Ziphocaiman terrestris). In places where both of these reptile
species occur, monai prefers drier places on watersheds, since it is less demanding
on the availability of drinking water.
Mating takes place during the dry season. After mating, the female searches
for a place to lay eggs. Usually it is a well-warmed place, most often where
the female hatched. Good areas for egg laying are used for decades by many generations
of these reptiles in succession. The clutch numbers from 4 to 12 eggs; the female
buries them in the ground and no longer cares about the offspring. Young monais
hatch in 150-160 days. They differ from adults in a brighter coloration and
the ability to change it within certain limits. At the age of 8 years, having
reached a length of about 1.8 m, they become sexually mature. Life expectancy
is up to 70 years.
The llanos of the northern part of the continent are inhabited by a larger (up
to 3 meters long) relative of this species – azure-headed
monai (Notovaranus azuriceps). This species hunts not only small prey,
but also medium-sized herbivores. It is distinguished by a more massive physique
and is unable to rear on its hind legs. The scales on its head have a lapis
lazuli color.
This reptile species was discovered by Nick, the forum member.
Eonesian
eonesaura (Eonesaura eonensiensis)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Teiids (Teiidae)
Habitat: Eonesia Islands (Burotu Archipelago).
Among the wingless vertebrate colonizers of various islands, lizards and snakes
are the most successful ones; they are able to reach places that are inaccessible
to mammals, except bats. This was the case before the Holocene, in the human
epoch and after – in the Neocene. Lizards and snakes of small and medium size
spread almost everywhere and appeared even in places where it seemed impossible
to live without wings.
Burotu Archipelago is one such place. It is just a chain of 15 small and medium-sized
islands. 4 of the largest of them are covered with forests, the rest ones –
with thickets of shrubs with a clearly marked coastal beach zone. Such an ecosystem
allows the most difficult compatible groups of birds to get along – ground pigeons
and parrots (plus flying songbirds) with sea birds (whose flight abilities have
been preserved) – and also with lizards.
It is difficult to say when the ancestor of the eonesaura got to Burotu from
South America, and how it did it – most likely, the ancestors of these reptiles
were brought there by a storm on a snag or a “raft” of plants. Once in a new
place, this lizard began to flourish, and its descendant reached very large
size – up to 80 cm in length. In addition, at the beginning of their evolution,
when parthenogenesis was not yet the only reproduction option, and representatives
of the ancestral species settled on various islands, they formed several subspecies
that differ from each other in size and shape of the head.
The main subspecies, the common eonesaura (Eonesaura eonensiensis vulgaris),
has a striated green-black color. The stripes on the sides are clearly defined,
but on the back they become paler and blur on the green background; on the tail
the stripes are much clearer. The tail is long, but proportionally shorter than
that of small teiids from the mainland. The legs are relatively short, but strong
and well developed, with tenacious digits and claws. This lizard is quite capable
of climbing trees for the sake of chicks, social insects or fruits; this species
is omnivorous. Its teeth are small, but numerous and pointed; the jaws are well
developed. The skull is noticeably thickened from above, and the snout is blunted.
There are two longitudinal ridges on the skull, which makes the head appear
tetrahedral. The eyes of this Eonesian theiid are small, in addition, covered
from above and slightly in front with peculiar bone visors – the protrusions
of the skull that protect them from damage. This reptile does not see well right
in front of its head and is guided largely by the sense of smell, hearing and
touch – it feels well the vibrations from the movement of small animals on the
ground or wood. Since this lizard reproduces parthenogenetically, it is quite
peaceful and relatively slow. The common eonesaura lives on large islands covered
with forest, and prefers to live under the forest canopy and in forest clearings.
Its natural enemies are predatory birds,
and the juveniles are attacked by large sawleg
grasshoppers. When attacked by an enemy, this lizard prefers to defend itself
actively: it clings to the enemy with a bulldog attitude, hits it with its tail
and tears with its claws. This method is quite effective, and most predators
attack only young lizards or feed on eggs of this species. However, cases of
cannibalism are quite frequent.
Living in a mostly tropical climate, eonesaura does not have a clearly defined
breeding season. In addition, it reproduces parthenogenetically, and it means
that the population has no males fighting for the right to mate with a female.
There are 16-17 eggs in the clutch, which the female buries to a shallow depth
and covers with rotten leaves. These lizards don’t care about offspring.
Other subspecies inhabit small islands and differ greatly from the nominate
form, mainly in the size and shape of the skull. The subspecies horned
eonesaura Eonesaura eonensiensis moloch differs from the nominate form
in much smaller sizes – no more than 30 cm in length. In addition, the crest
above the eyes is set along the edge with corneous spines that greatly expand
it, especially at the back of the head. This feature makes it difficult for
predators to swallow this lizard. The reptile’s skin color is also green, but
with a yellowish tinge and with gray vertical strokes on the sides. In addition,
representatives of this subspecies can be of different sizes. Although this
subspecies also reproduces parthenogenetically, females engage in pseudocopulation,
and court each other before laying eggs. Therefore, in the habitats of this
species, both larger and aggressive individuals, and smaller ones may be found.
Each lizard lays eggs separately; clutches number fewer eggs (up to 10 on average),
and this subspecies is found closer to the coastal strip than the previous one.
Nevertheless, the horned eonesaura also climbs well, but rather on bushes than
on trees, and leads a similar lifestyle.
The lesser eonesaura Eonesaura eonensiensis minor subspecies
is the smallest of all the subspecies of this species: the length is no more
than 20 cm in adult lizards. Unlike its relatives, this subspecies occurs mainly
near water, swims and dives well; it inhabits large humid islands of the archipelago,
where there are small reservoirs. This reptile is found among coastal shrubs,
tends to stay on the ground and avoids dry places. It feeds mainly on invertebrates
– insects and crabs; small fish and carrion are also present in its diet; plant
food is almost absent in its diet, unlike other subspecies. The clutches are
small (usually half a dozen eggs), and the young hatch quickly, within two weeks;
but the female bears them in oviducts for a long time, much longer than other
subspecies. There are no spikes or other ornaments on its head, pseudocopulation
is also not observed.
This reptile species was discovered by Bhut, the forum member.
Tessellated
eonesaura (Eonesaura psittacophila)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Teiids (Teiidae)
Habitat: Eonesia, Burotu Islands.
In the Neocene, the islands formed after the end of the human epoch had a typical
island fauna, in which there were no consequences of human activity. Small reptiles
that can withstand occasional sea voyages and easily get accustomed in new habitats
represent one of the components of this fauna. A significant number of lizard
species inhabit the islands of the Pacific Ocean, including the Eonesia islands.
The most remote of the new archipelagos is Burotu, where only a few species
of teiids live – the result of adaptive radiation after only one successful
settling. Their ability to reproduce parthenogenetically helped them to develop
new habitats.
Avoiding competition, some species of these lizards have developed unusual ways
of life and even entered into alliances with neighbors on the islands. A small
lizard, tessellated eonesaura, living at the Burotu Islands, became a symbiont
of the infantopsitta parrot,
a large local flightless bird. It constantly stays in the burrows of this bird
and next to them, and outside of bird’s nesting it lives on the body of this
parrot and helps it to get rid of parasites.
The length of an adult of this species is up to 10 cm. The physique of the tessellated
eonezaura is gracile; the lizard looks like a salamander with a slender cylindrical
body, small legs and a very long tail. This species has a conical head and small
golden-yellow eyes with vertical pupils. The background color of the skin of
this reptile is black, large white spots are located in pairs on its sides.
On the tail, the spots merge and turn into transverse bands. The surface of
the scales is smooth; due to this feature, the reptile can easily climb in the
plumage of its host, searching for parasites. The digits are expanded at the
tips and have transverse ridges developed from the edges of the scales and hooked
claws.
The symbiotic relationship of the lizard and the host bird is not obligate:
if necessary, the tessellated eonesaura is able to live independently, inhabiting
tree trunks and thickets of grasses. The reptile keeps on the lower part of
tree trunks and hunts small insects, but at the first opportunity it moves to
the bird’s body or into its burrow.
The symbiosis of these reptiles with birds makes it difficult to meet mates,
therefore, in this species, the main method of reproduction is parthenogenesis;
the female lays 1-2 eggs once a month and buries them in the forest litter.
Incubation lasts up to 5 weeks; young individuals differ from adults by the
brown background color of the body and randomly scattered white spots. Sexual
maturity comes at the age of 2 years; by this time the pattern on the body becomes
orderly, and the coloration changes to adult black and white. Life expectancy
reaches 15 years.
The idea of the existence of this reptile species was proposed by Bhut, the forum member.
Aepylacerta
(Aepylacerta viridis)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: True Lizards (Lacertidae)
Habitat: New Azora, rainforest.
True lizards of the human era are small, often dimly colored creatures, among
which there are almost no species remarkable in bizarre appearance or unusual
behavior. In the Neocene, the situation on the continents has hardly changed.
However, on some islands, lacertids, in the absence of competitors, managed
to evolve into unusual new species. One of these islands is New
Azora in the Atlantic.
All the lizards of the New Azora belong to the true lizard family and descend
from a single North African or Southern European species that got to the island
by sea – obviously on a vegetation “raft”. There were no competitors – other
reptile species – at the island, and this circumstance, along with a favorable
climate, allowed the newcomer to give rise to a whole range of species that
greatly differ in food specialization and size. The successful colonization
was clearly a one-time event. Aepylacerta is the largest descendant of this
ancestral species; its maximum length reaches 1.4 m with a weight of up to 4
kg. This size is reached by the oldest females; aepylacerta’s growth is lifelong.
The appearance of this reptile is typical for true lizard family, but it has
some differences: for example, its muzzle is shortened. The appearance of the
aepylacerta reflects its arboreal lifestyle: hooked claws are developed on strong
legs, and a long prehensile tail, which makes up more than a third of the total
length of the animal, is used as an additional limb. These lizards are characterized
by sexual dimorphism: the male is smaller and slimmer than the female, and is
of brighter coloration: the main body color is bright green, on the sides there
is a row of five ring-shaped spots of black color. The bottom of the head and
throat are bright blue, the top of the head is pinkish-gray, and there are a
number of blurred black transverse stripes on the tail. The male becomes the
brightest after exuviation, before the beginning of the breeding season. The
female’s head is colored grayish-green; it lacks dark areas on the body and
tail. The color intensity of these lizards is variable; it strongly depends
on the color of the surrounding vegetation and the physical and emotional state
of the animal.
Aepylacerta is an arboreal reptile; it spends most of its life on the branches
of trees. However, these lizards walk well on the ground and can even swim a
little. The diet of the aepylacerta, quite unusual for a reptile, consists of
70 percent vegetative food: fruits, leaves and young shoots. A long intestine
with a rich microflora helps to digest this hard-to-digest food; there are even
bacteria capable of splitting the wax covering the fruits of trees of the laurel
family. Interestingly, intestinal symbionts are transmitted to the juveniles
“by inheritance”: near the clutch, the female leaves her feces, eaten then by
the hatched juveniles in the first day of their life, acquiring useful microflora
in this way. However, this reptile needs animal protein, so in addition to vegetation,
it eats large insects, mollusks, young small vertebrates, and sometimes ravages
bird nests. Aepylacertas get water from food and absorb the mist with their
skin, but sometimes they lick off the dew accumulating on the leaves. In the
search for food, these reptiles are helped by well-developed color vision and
a keen sense of smell.
Aepylacertas are rather phlegmatic animals that often lie motionless on branches,
and bask in the sun for a long time in the morning (this reptile leads a daytime
lifestyle). Only during the mating season, which takes place in the spring,
they become more active. At this time, males become intolerant of violators
of their territory, show aggression to each other and fight, and try to attract
females by displaying the bright coloring of the head and body sides. Approximately
50 days after mating, the female lays 8-12 eggs in the rotten foliage at the
foot of the tree, simultaneously emptying her intestines. This is where her
concern for offspring ends. The hatched juveniles, having got out of the ground,
scatter through the forest and climb trees. They differ from adults in size
and have a monotonous bright green coloration. They will become sexually mature
only two years after hatching, and they can live up to 20-25 years, although
the average life span does not exceed 10 years.
Young aepylacertas have many enemies, but their number decreases with age: the
oldest individuals should be afraid only of the largest predators of the New
Azora – martillas, for example.
Despite the fact that this reptile can fight back against the enemy by biting
and hitting it with its tail, aepylacertas usually rely on camouflage and try
to freeze or stay in areas of the tree inaccessible to the predator when a predator
appears.
This reptile species was discovered by Simon, the forum member.
One-colour
viviparous lizard (Zootoca palearctica)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: True Lizards (Lacertidae)
Habitat: Northern and Central Europe, forests.
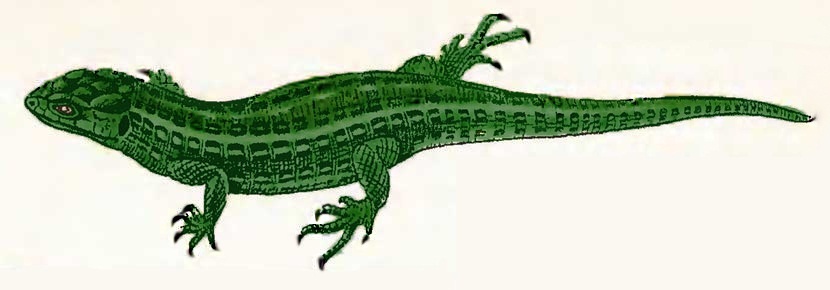
Picture by Lambert

Picture by Alexander Smyslov
In the Holocene epoch, the viviparous lizard (Zootoca vivipara)
was the northernmost of all lizards in the world. One of its descendants, the
taiga lizard, has firmly established itself in
this niche and has developed several additional adaptations that allow it existing
in the seasonal climate of Siberia, which is not too favorable for ectothermic
animals. Moreover, its close relative, the one-color viviparous lizard, replaces
it in Northern and Central Europe.
Unlike its taiga relative, this species does not enter into symbiosis with ants,
preferring to dig its own burrows – relatively shallow, but in initially protected
places: under a deadfall, under a layer of leaf litter, sometimes between the
roots of trees. The burrow is shallow and is used for a short time – representatives
of this species are not territorial and can both dig a new burrow and use an
empty someone’s else one. Sometimes they use ready-made shelters of natural
origin.
The one-color viviparous lizard reaches 15-20 centimeters in length; its tail
is shorter than that of the Siberian species, and by autumn it becomes much
thicker due to fat stock. Unlike the taiga lizard, it is usually monotonous
green in color, and other color morphs are almost absent in this species; like
the taiga lizard, males also have a small skin ridge on the back.
This lizard comes out of hibernation earlier than representatives of a related
species from Siberia, and goes to winter later. By the end of April, females
of this species begin leaving odorous marks that indicate their readiness to
mate. Males follow them, guided mainly by the sense of smell, and if several
males meet near one female, a fight ensues between them, sometimes quite ferocious,
until a winner is revealed.
In the first half to the middle of summer, a female of one-color lizard gives
birth to 2-4 live juveniles, which differ from their parents only in size. They
are completely independent and immediately start their own life.
The main enemies of this lizard are small predatory mammals and birds, less
often snakes. Like their eastern relatives, these lizards sometimes die from
the winter cold, but it happens less often – they live in the zone of a milder
winter climate. This species feeds on various invertebrates: insects and spiders,
less often snails. Sometimes cases of cannibalism happen, but they are rare.
Lizards tend to hide in the forest floor or run away from enemies, but, not
having such an opportunity, they actively defend themselves and bite, injecting
weakly poisonous saliva with a strong irritating effect into the wound.
The total lifespan of a one-color viviparous lizard is 10-12 years.
This reptile species was discovered by Bhut, the forum member.
Feyi
(Zootoca feyi)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: True Lizards (Lacertidae)
Habitat: steppes of Asia.
During the Holocene epoch, the viviparous lizard was one of the most common
lizards of Eurasia. In the Neocene, its descendants form several different species
– it managed to populate the mountain
range of the Alps and was even to colonize
the volcanic Loki island in the Atlantic; one of its descendants is found
even in the Siberian taiga (taiga lizard), and
another species, feyi, is found further south, in the steppes of Mongolia and
neighboring territories (the species is named after the monster from Chinese
mythology).
Feyi is a small lizard: about 10 cm long with the tail is the same length. It
has a strong physique and relatively short strong legs equipped with claws:
this lizard is a good digger and equally well runs on the ground and digs tunnels
under the roots of plants. At the tip of the muzzle, the horn shields are thickened.
The reptile has a gray-brown color with a pattern of thin transverse ripple
on the back and upper side of the tail. On the sides there are two brighter
longitudinal stripes of green color, starting on the cheeks and ending on the
proximal third of the tail. The male is usually longer and slimmer than the
female.
Feyi is usually active at night, when the ground still retains the warmth of
the sun. The lizard runs on the ground surface and hunts mainly various invertebrates;
its favorite food includes orthopters – crickets, locusts and others. With the
help of strong legs, it easily digs their egg pods out of the ground. Sometimes
it eats small vertebrates – usually chicks of the ground-nesting birds or baby
rodents.
The enemies of this species are local snakes and owls, less often carnivores
and large lizards. When caught, this lizard resists, wriggles, hisses loudly
and tries to bite its enemy; sometimes it manages to escape by slipping out
of the enemy’s grasp thanks to the smooth body coverings.
In spring, the breeding season begins at feyis: the females leave odorous marks,
the males search for them by smell and mate with females. The courtship ritual
is absent, and feyi female can mate with several males per night. This species
does not form stable breeding pairs; if several males meet near the female,
they can fight with each other, including without interrupting mating. A few
weeks later, the feyi female give birth to several – from 2 to 4 – juveniles.
They look like a smaller copy of adult lizard, and immediately begin an independent
life. The survival rate of the offspring is good. Sexual maturity comes at the
2nd year of life.
Life expectancy is up to 15 years.
This reptile species was discovered by Bhut, the forum member.
Mountain
alpinosaurus (Alpinosaurus montanophilus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: True Lizards (Lacertidae)
Habitat: Alpine valleys in the southern part of the Alps.
The human epoch is characterized by the mass extinction of local endemic forms
due to the direct extermination and destruction of their habitats. Species with
a large populations and a wide range survived the human era relatively successfully
and in the Neocene began to evolve rapidly, filling empty ecological niches.
In the Neocene, various local lizard species appeared in the Alps mountain range,
which inhabit only some valleys of the mountain range. The mountain alpinosaurus
is one of the many descendants of the viviparous lizard (Zootoca vivipara) that
appeared in the warm valleys of the Alps, a representative of a wide complex
of related species that differ in their appearance, behavior and physiology.
The maximum length of this lizard is about half a meter, of which the tail is
half the length. In its habitats, it is one of the largest reptiles along with
the tatzelwurm skink. This species
has rather long legs and a large head; in proportions alpinosaurus looks like
a sand lizard. The coloration is variable in wide limits: alpinosaurs from different
populations may have different background colors – brown, gray, green and brown-green.
A common element of coloring is a pattern of black spots along the back and
on the sides. In addition, there are often other variants of the spotted pattern:
small spots all over the body, large strokes merging into transverse rings on
the legs. The belly is light and of one color, usually white or yellowish. The
eyes with an orange iris contrast with the camouflage coloration; in males they
are usually brighter.
Alpinosaurus lives in the warm valleys of the southern Alps. It prefers open
spaces where there are lots of rocks and grass. The lizard is active during
the day, likes to bask on the rocks, but is very careful and hides between the
rocks in case of danger. The diet is very diverse: insects, spiders, scorpions,
snails, amphibians, smaller reptiles, bird eggs and chicks, small insectivores
and rodents, as well as carrion. The lizard licks small prey from substratum,
grabs larger prey with its jaws, and tears off pieces from carrion. Sometimes
the reptile looks for prey on the banks of rapid mountain rivers, where it looks
for fish, crustaceans, tadpoles, as well as fish and frog eggs. The lizard does
not like cold water and looks for such kinds of food only near the shore.
The enemies of this species are small carnivorous mammals and birds of prey,
but the mountain alpinosaurus bites painfully, and this is one of its main defensive
techniques. Also, the enemies of its juveniles are snakes. The animal winters
in burrows under rocks or in forests growing in mountain valleys.
This species has a pronounced seasonality in reproduction. The breeding season
is in April; the male searches for the female by scent, chases her and mates
quickly. Each male guards his territory; during the breeding season, the color
of his eyes becomes brighter and serves as a signal for rivals. When males meet,
a fight ensues between them, which often ends with fractures of digits and torn
flaps of skin.
The female bears eggs for several weeks and gives birth to 5-6 cubs up to four
centimeters long, ready for independent life. The basis of their diet consists
of insects and snails, with age the diet expands. They also have rapidly growing
jaws, and juvenile individuals seem to have a larger head than adults.
Sexual maturity comes at the age of 3 years; life expectancy is up to 20 years.
This reptile species was discovered by Mamont, the forum member.
Fafnirella
(Fafnirella nigra)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: True Lizards (Lacertidae)
Habitat: Atlantic Ocean, Loki Island.
The viviparous lizard (Zootoca vivipara) contemporary to people is one of the
most cold-resistant reptiles. Some parts of its range reached the Arctic Ocean.
It is not surprising that significant resistance to cold, as well as a wide
geographical distribution and high number of the population allowed the viviparous
lizard surviving during the ice age that marked the end of the Holocene. In
the Neocene, when the climate became much warmer, the species descending from
it repopulated northern Europe and Asia. And one of the distant descendants,
which settled the Loki island rose from the bottom of the Atlantic Ocean after
the end of the human era, became one of the largest representatives of the family.
This animal is called fafnirella – in honor of the character of Scandinavian
myths, the dragon Fafnir, who guarded untold treasures. The ancestors of this
species came to Loki island by sea from Europe. Fafnirella is not directly related
to the lizards of New Azora and is the
result of another attempt of colonization of the islands of the Atlantic by
the lizards of Eurasia.
This is a fairly large lizard – the body length of adults is 55-70 cm with a
weight of up to one kilogram. The male is slimmer and slightly smaller than
the female. The base of the reptile’s tail is thickened – fat for the winter
is stored in it. From the point of view of proportions, these reptiles look
more robust than the ancestral species – they have a shorter tail, thick paws
and a large head. Fafnirellas are colored shining black – this color allows
them absorbing solar energy with maximum efficiency. It is curious that such
a color was often found in viviparous lizards of the human era. Only on the
belly there are dark brown spots of irregular shape, and in males the throat
is colored white.
These lizards are terrestrial animals. They are active during the day, and spend
the night in burrows. These reptiles treat their relatives quite aggressively,
although they are not strictly territorial animals.
Fafnirells belong to the zoophagous reptiles. Most often, their menu includes
large invertebrates – insects, spiders and terrestrial mollusks. They also often
eat carrion, ravage the nests of land birds and eat their eggs and chicks. Fafnirellas
are too slow to hunt adult birds, but the most experienced individuals can get
birds by hiding and using the effect of surprise. These reptiles often look
for food on the seashore: they gather animals cast ashore by the surf, catch
crabs and cause damage to seashore bird rookeries. The prey items of adult fafnirellas
are often young individuals of their own species. Vegetative food is almost
not used by these reptiles: only sometimes they eat juicy fruits and berries.
The breeding cycle of fafnirellas begins in mid-spring. At this time, they get
out of the shelters where they spent the winter, and gather in large numbers
in places well warmed by the sun, as well as at hot springs where underground
volcanic heat comes out. At this time, males arrange peculiar tournaments: hiss
menacingly, inflating their white throats, bite and fight, standing on their
hind legs and clasping the opponent with their forelegs. The winners of the
tournaments get the right to mate with a female. About a month and a half after
mating, the female begins laying eggs – the fafnirella lost the ovovivipary
characteristic of the ancestral species. The reason for this is that these lizards
lay eggs (usually 10-12) in the soil warmed by volcanic heat – in such natural
incubators, the offspring develop faster than in the female’s body. Skirmishes
often occur between fafnirella females in places of mass egg laying. The best
places for laying eggs are occupied by the strongest females, and not everyone
has enough space. Outsider females have to lay eggs in ordinary sand on the
seashore. Their offspring are born later, and they have less chance of survival
– young lizards are often attacked by seabirds. In volcanic sand, egg incubation
lasts a little bit longer than a month. The newly hatched juveniles have a chocolate-brown
color. During the first months of life, they feed on small insects and live
on trees and shrubs. Only in the second year of life they descend to the ground
(adult fafnirellas lead an exclusively terrestrial lifestyle). The main enemies
of the juveniles are adults not missing the opportunity to eat one of them.
Since October, the fafnirellas reduce their activity. They become sluggish,
non-aggressive to each other and gather in places, where the soil is warmed
by volcanic heat. Here they dig holes where they spend the winter. Despite the
fact that the winters at Loki island are mild, fafnirellas are very inactive
at this time of year, although they do not hibernate. Every few days, they come
out of their burrows to search for food, but do not go far. The fat stored in
the tail helps them to survive the cold season: they accumulated it by actively
eating, starting from the middle of summer.
Given the huge role that endogenous heat plays in fafnirellas’ life, we can
say that it is the main factor that allows such a large reptile to exist in
the cool climate of Loki island. Another factor is the lack of competition from
mammals.
The main enemies of fafnirellas are birds of prey. These lizards actively defend
themselves from them by biting and using their tail as a whip. If the forces
are unequal, the reptile flees and hides in a hole. Only young individuals are
capable to autotomy (tail dropping). Adults with a regenerated tail usually
enjoy less success during the mating season.
The life span of this reptile can reach 26-28 years. Such longevity, usually
uncharacteristic for lizards, is achieved due to periods of slowed down activity.
This reptile species was discovered by Simon, the forum member.
Tree-climbing
goanna (Varanus arboreus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: Meganesia, tropical rainforests in the north of the mainland, tree
crowns.
Goannas – Australian representatives of the monitor lizard family – represent
one of the most successful experiments of evolution for durability. One representative
of their kind, the huge Megalania lizard, was one of the top predators of Australia
of the Pliocene epoch, before the arrival of humans and placental predators
on the island continent. Now both humans and megalanias have long disappeared
from the face of the Earth, and a new large representative of the goannas –
the terrible dracovaranus – terrifies the animals
of the Neocene Meganesia. Nevertheless, dracovaranus, like Megalania before
it, is rather an exception to the rule in the Cenozoic, when not reptiles or
dinosaurs, but birds and mammals are at the top of the food chains. Other representatives
of the monitor lizard family confirm this.
The tree-climbing goanna is a reptile about one meter long, weighing up to 12
kg. It is covered with green scales with black stripes, slightly resembling
South American iguanas of the human era in its coloration; mature males have
a white or yellow gular sac, which they inflate during the mating season, at
the end of the Meganesian summer. Unlike dracovaranus, tree-climbing goannas
are not the top predators of their ecosystem, the rainforest in the north of
Meganesia; large warm-blooded creatures like predatory
descendants of cassowary and larger species of marsupial
civets do not miss the opportunity to attack even such a large lizard,
especially early in the morning or at night, when large reptiles are less active.
The juveniles of tree-climbing goanna, inferior to the adult representatives
of this species in size, are vulnerable even to small owls. Their only defense
is the speed of movement and the ability to hide in the narrowest shelters.
In addition, young individuals have another protective device. In the process
of evolution, the tree-climbing goanna has developed subcutaneous glands that
open with pores on the sides of the body and secrete a special slippery greasing
on their scales. This feature makes young goannas, firstly, very slippery, and
secondly, unpleasant to the taste for most predators. Nevertheless, cannibalism
is common in these reptiles, and these secretions do not protect much from an
adult cannibal attack. Young individuals are colored somewhat differently than
adults – they have more black than green in their coloration, and they can hide
among epiphytes, remaining motionless and merging with the shadows of their
leaves on the tree bark.
In adult arboreal goannas (especially in males, which sometimes reach more than
a meter in length) this greasing is secreted in much smaller quantities, and
they defend themselves (and attack) by inflicting bites and painful tail blows
on enemies. Like some Holocene monitor lizards, the mouth of tree-climbing goanna
is a home to bacteria that make the bite of this lizard poisonous and dangerous
to health. Secretions have another function – they prevent the development of
bacteria. Therefore, having received a wound from the teeth of a predator or
a relative, the reptile secretes intensely this greasing and applies it with
its paw to the wounded areas of its skin.
Tree-climbing goannas feed mainly on small and medium-sized animals – arboreal
marsupials, small birds, sometimes their own relatives and other reptiles; smaller
and younger individuals also eat invertebrates – snails and insects. Larger
animals, being less agile on trees (goannas climb using their claws, not “stickers”
like geckos), sometimes descend to the ground and hunt there; these lizards
do not disdain carrion, which they search for with the help of a keen sense
of smell.
At the beginning or middle of the Meganesian autumn, tree-climbing goanna female
lays a clutch of 10 to 15 eggs in tree hollows and covers them with the foliage
of the same trees. To do this, the reptile gets out on thin branches, and, at
the risk of falling off, tears off the leaves. After taking some leaves in her
mouth, the female returns to the nest and puts them there.
After the clutch is laid and covered from prying eyes with foliage and green
twigs, the female leaves it and does not take care of it anymore. At least half
of the young goannas simply do not live to puberty, despite their protective
adaptations. Young goannas, however, are tolerant of each other, and juveniles
from the same brood stay together for quite a long time, and it compensates
somewhat for mortality at an early age. Adult tree-climbing goannas are also
quite tolerant of each other due to the high availability of prey in the crowns
of the rainforest. If during the mating season two or four males have to face
each other near one female, they establish hierarchy without a fight, displaying
each other the size of their gular sacs: the male with the largest one wins.
Tree-climbing goannas live up to 80-90 years, but usually less, because they
are hunted by a large number of various predators.
This reptile species was discovered by Bhut, the forum member.
Hook-snouted
goanna (Varanus protorhynchus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: Meganesia, tropical rainforests at the north of the continent.
At the genetic level the closest relatives of tree-climbing goannas are hook-snouted
goannas living at the same area, but having completely different ecology. These
ones are larger, one-coloured dark green (up to black) reptiles (old males sometimes
reach more than three meters in length) living not on trees, but in thickets
along valleys of wide streams and forest rivers where in forest canopy gleams
appear and the plentiful underbrush develops on the ground. Their males do not
have advanced gular sac, but along their backbone darker crest of corneous plaques
stretches.
Though hook-snouted goannas live near water, they do not behave like their African
relatives resembling crocodiles in their way of life. On the contrary, hook-snouted
goannas prefer to watch for the prey at the riverbanks and seize it on land,
or snatch out from water, standing on land. At these lizards the semblance of
collective behaviour is advanced: large male gathers a harem of 5 – 6 females,
and reptiles are capable to attack large animal in common. Hunting and eating
prey together, these lizards can defend the prey even at predatory cassowaries,
large predators of Meganesian jungle.
These lizards are named because of the shape of snout: their maxillae are deflexed
and their shape resembles fish-hook a little. In common with powerful jaw muscles
and sharp teeth it permits hook-snouted goannas to peel meat from the prey completely,
up to bones, or to break and to eat these bones in common with meat. As a rule,
these reptiles eat the parts of prey body containing small bones, right with
bones – hands, feet, tails and a part of thoracic cage with ribs. If prey has
rather small head (kangaroo, for example), due to mobile jaws and flexible ligaments
between them lizard can swallow also a head of prey together with skin, wool
and teeth. Reptile will digest also such dinner successfully, and not digested
teeth and wool of prey will leave with dung.
As well as at their tree-climbing relatives, the courtship period at hook-snouted
goannas begins at the end of Meganesian summer. But it takes place roughly and
aggressively, being accompanied by roar of dominant males warning thus possible
opponents and notify unfamiliar and lonely females on the force and readiness
for breeding. Sometimes between males combats take place; at these moments males
rear on hind legs and try to tumble down each other on back and to press against
the ground. Defeated male hastily leaves territory of the winner; it doesn’t
matter, whether it belonged to him earlier or not. The winner expels losed male,
pursuing him and biting its sides or tail.
Though females live together, each of them digs clutch in the ground separately,
carefully hiding the nest from congeners; in clutch of this species there are
10 – 12 eggs. Like tree-climbing goannas, they do not care of the clutch and
also leave the posterity to the mercy of fate. The majority of more-less serious
predators prefers to leave the territory of hook-snouted goannas – these lizards
are dangerous opponents. And this circumstance saves many young individuals.
But the advantage of it is reduced to zero by active cannibalism of adult individuals.
Young individuals have not won yet the harem or have not adjoined to already
existing one are especially dangerous to young ones. And juveniles fall prey
of adult reptiles.
As well as at tree-climbing goanna, life expectancy of hook-snouted goanna may
reach 90 years. If the reptile of this species had not died up to achievement
of one-and-a-half-meter length, it has all chances to reach age limit.
This species of reptiles was discovered by Bhut, the forum member.
Ant-monitor
(Myrmivaranus flagellicaudis)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: savannas of Meganesia.
Picture by Timothy Donald Morris
Initial sketch by Arseny Zolotnikov |
Long and thin tongue of reptiles of order Squamata, is constantly put outside
to gather information about the environment and to carry it to Jacobson’s organ
at the palate. It is frequently able to penetrate into various cracks and apertures.
Therefore would be quite possible to it to get in the future any more function,
which was earlier not met at present group of animals.
In human epoch monitor lizards (Varanidae) were considered as the most progressive
family of lizards. One of variety centers of this family is Australia. In Neocene
one of survived representatives of monitor lizards family from savannas and
semi-deserts of this continent, using an abundance of various species of social
insects, had passed from predating habit of life to feeding by this specific
catch. Before this species Australian echidna (Tachyglossus) and less specialized
banded anteater, or numbat (Myrmecobius) had such diet. This reptile became,
thus, their ecological analogue, taken place of these animals in savannas of
Neocene Meganesia.
Reaching more than 2 meters in length, the Meganesian ant-monitor
is rather sluggish and slow, and rather peaceful in comparison with its relatives.
It
has powerful enough claws, both on front and on hind legs, with which breaks
the ground and nests of colonial insects. Long and thin tongue, doubled at
the end and covered with sticky saliva, allows to catch insects and to draw
them in mouth. Saliva also involves by taste and smell some ants, which get
on tongue when lizard pushes it in their nest. Teeth of this reptile are considerably
reduced; they are small and sharp, located only in forward part of jaws.
Thick scaly skin protects body from stings which are put to monitor lizard
by insects, furiously protecting ravaging nests. Head of this monitor lizard
is covered from above with almost continuous corneous armour. On back of ant-monitor
there are sticking up enlarged scutes also used as passive protection. They
form cross belts on all back of the reptile, representing reliable protection
against medium-sized predators.
In case of danger the ant-monitor protects itself by impacts of tail, on which
end some rows of pointed scales were developed, and also by big powerful claws,
making aggressive lunges aside the enemy.
Colouring of body is soft: from yellowish-grey to dark brown tones (there are
individual deviations in color), lighter yellowish strip through lateral part
of neck and shoulders serves for recognition of neighbours. For strengthening
of contrast with colouring of body it is shaded from above and from below with
darker strips. Color of this strip directly depends on physical condition of
animal. At meeting two reptiles of this species turn heads aside, showing this
strip and estimating thus physical opportunities of each other.
The basic food of ant-monitor is social insects. This monitor lizard feeds
with them as in nests (destroying them with strong claws), as on their tracks
or in thickets. Perhaps, only soldier ants devouring all alive on their way,
represent the certain danger for it, but their more peaceful neighbours frequently
become its food. In searches of insects ant-monitor is able to rise of hinder
legs, and base by forepaws against trees trunks, surveying bushes and branches
in searches of insect nests. But because of massive armour this reptile does
not differ in abilities of tree-climber. Also this reptile can feed on ground,
digging out underground nests of insects, or eating underground larvae of beetles
and cicadas.
Ant-monitors are single animals, but in areas of dense settlements of social
insects they keep by groups of 3 – 5 individuals. Usually these reptiles constantly
wander in savanna, not being late for a long time in any place.
Males at this species are larger than females. Ant-monitor lays eggs (up to
20 ones) to unusual incubator: into termitarium. After all clutch is laid,
female leaves it, and termites repair the damaged wall of nest. Eggs appear
in very favorable conditions, and besides, they get a smell of termitarium.
Therefore practically nothing threatens to clutch and young ant-monitors, and
the first time they grow in environment of own food. The adult female, contrary
to habits of majority of lizards, does not leave nest at all. It marks termitarium
becoming an incubator for its clutch, and also remarkable objects in its vicinities
with musk liquid from glands opening on hips, and keeps beside. It will drive
off adult neighbours from nest during all term of incubating, but approximately
when the young growth hatches from eggs, it leaves a post. The first ant-monitor,
destroying termitarium in searches of food, involuntarily releases young growth
from imprisonment.
Young ant-monitors have brighter and contrast colouring, than adults. Primary
color of their body is yellow, on it brown cross strips pass. Stomach is white;
along neck and on shoulder brown strip passes. Later colouring changes: strips
on body disappear, and strip at shoulder becomes brighter: on it light stains
appear, merging at the adult reptile to continuous strip.
Young ant-monitors are still too weak to break open nests of termites independently.
Therefore they keep near to adult reptiles, and feed after them at cracked
termitariums. They will reach the size of adult animal approximately to 6 -
to 7 years, though they’ll become able to breed already at 3-rd - 4-th year
of life.
This reptile species was discovered by Arseny Zolotnikov
Pouncing
monitor lizard (Varanus dendrobius)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: Meganesia, tropical rainforests.
The warm climate of the Neocene contributed to the evolution and spread of reptiles.
Mass extinction and the ice age reduced the number and diversity of these animals
in the early Neocene, but subsequent warming gave impetus to a new wave of their
evolution.
In the Holocene, monitor lizards represented a characteristic group of reptiles
of Meganesia. In prehistoric times, the largest monitor lizard was found on
the territory of Australia, and in the human era, the smallest species of this
group of lizards lived there. In the Neocene, the monitor lizards of Meganesia
became as diverse as in the pre-human era again. The north of the continent
is covered with dense tropical forests, and among them, there are species living
on different levels of the forest.
In the undergrowth of the tropical rainforests of Meganesia, a peculiar species
of Neocene monitor lizard lives – a pouncing monitor lizard. It is a large lizard
of light build, reaching two meters in length. It has strong legs; the hind
legs are about twice as long as the front ones. Characteristic features of this
reptile also are hooked claws and long tail.
The head of the pouncing monitor lizard is elongate, flat from above, bearing
two flat leathery outgrowths above large eyes. Pointed teeth with longitudinal
grooves along the posterior edge grow in the reptile’s mouth. The reptile can
open its mouth very wide, and its neck is very mobile and flexible.
The upper side of the body of the pouncing monitor lizard has camouflage coloration
– brown with sparsely scattered gray and green spots. The belly and throat of
the reptile, on the contrary, are clearly visible, white with a yellowish tinge.
This color is associated with the peculiarities of the behavior of this monitor
lizard: this predator hides on tree trunks in an upright position, tail down,
holding on to the bark of the tree with all paws. Thanks to the coloring, it
is so invisible that the prey can approach it almost at point-blank range. When
the chosen prey is close enough, the monitor lizard abruptly pushes off from
the tree trunk with its hind legs. In a jump, it turns around like a cat, and
falls on the prey already with its paws down. The pouncing monitor lizard attacks
large ground-dwelling birds and mammals ranging in size from a cat to a piglet.
The strength of the monitor lizard would not be enough to deal with large prey,
biting and torturing it, as other monitor lizards do. The reptile only grabs
the prey with its paws, holds it for a few seconds and inflicts a strong bite,
after which it lets prey go. The prey can escape, but now the monitor lizard
begins to follow it relentlessly. It moves at a walk, guided by the smell of
prey, which it remembered during the attack. No matter how long the pursuit
lasts, the result will always be the same.
In the teeth of a pouncing monitor lizard bacteria live, that produce a strong
poison. The reptile has no special glands or other organs in which bacteria
could settle, so they simply inhabit the cavity of its mouth. The most comfortable
habitat for bacteria is the grooves in the teeth of this monitor lizard. After
the bite, the bacteria enter the wound and begin breeding and secreting poison
actively. Prey infected with them quickly dies from poisoning. Even if it managed
to escape far away, the predator will definitely find it by smell. If the prey
is weakened, but still retains strength and escapes from pursuit, the pouncing
monitor lizard catches it up with running and inflicts several more bites, from
which it bleeds and dies.
Since the pouncing monitor lizard lives in a tropical climate, seasonality in
reproduction is not expressed, and individuals ready for reproduction are found
at any time of the year. Mating displaying at this species include standing
and walking on its hind legs – this way the male shows the female a light belly,
clearly visible even in the semi-darkness of the undergrowth. If two males meet
near one female, a fight begins between them. Since an accidental bite can be
life-threatening, males fight by striking each other with their tails. An important
element of courtship in this species is “vertical racing”, when the female climbs
onto the trunk of a tree, and the male chases her. The finale of this ritual
is mating, which takes place on a branch high above the ground.
The female lays 5-8 eggs in the soil under the roots of a large tree or in an
empty burrow and no longer cares about the offspring. After 7 to 8 weeks, young
monitor lizards hatch from the eggs. They are very different from adults in
color – emerald green with a white stripe along the back and along the upper
part of the tail. The belly, chest and throat of young individuals are bright
yellow. Young pouncing monitor lizards live in trees, thereby avoiding attacks
from adults. Due to their small size, they deftly run and jump on branches,
hunting insects and frogs. They reach sexual maturity at the age of 4 years.
The coloration begins changing to adult type already in the second year of life:
dark spots appear on the sides, which gradually increase and merge with each
other, replacing the juvenile coloration. The three-year-old reptile moves to
the lower part of the tree trunk and begins to lead a lifestyle characteristic
of adults, attacking only smaller animals.
Zahhak
(Varanus zahhak)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: Sunda Land, Kalimantan, Jakarta Coast and Southeast Asia.
By the beginning of the Neocene, monitor lizards had lost a number of rare and
local species; the number and range of other species also decreased in great
degree. However, after the disappearance of humankind, the remaining species
easily restored their former numbers and range, and even occupied a previously
unusual ecological niche of an aquatic
predator, when the number and distribution of crocodiles drastically decreased
due to human activity. Among the land-dwelling monitor lizards, both excellent
runners of light build and carnivorous giants
appeared. Among the latter, there is zahhak, the large Asian monitor lizard
(its name is the name of the Iranian dragon).
The zahhak is a descendant of the large Bengal monitor (Varanus bengalensis)
and is widespread over a large area of Southeast Asia. On the islands, there
are small local forms about 3 meters long (Sunda Land), and on the mainland
there are individuals up to 5 meters long. The appearance of this reptile is
typical for monitor lizards: large head with a wide mouth and sharp teeth, mobile
neck and long strong tail. The skin color is greenish-brown, usually with small
yellowish spots and specks that form transverse stripes on the body and the
base of the tail. A low skin ridge stretches along the back and tail.
The reptile’s teeth are laterally compressed, blade-like and serrated along
the edge. Zahhak feeds on a wide variety of animals – from large arthropods
and mollusks to grown-up juveniles of large mammals and does not disdain carrion.
Up to 3-4 adult lizards can gather around a large carcass; they behave aggressively
towards each other and quickly establish a hierarchy and feeding order. Usually,
smaller individuals stay away from large adult reptiles and approach the carcass
only after they have eaten: the zahhak can eat its own relatives. Thanks to
the notches on the teeth of the zahhak, pieces of meat keep, where putrefactive
bacteria develop – because of this, even large mammals, such as kong
and ganda, get sick for a long time after its bite.
These reptiles hunt relatively large prey from ambush, sometimes knocking the
prey down with blows of a powerful tail, and often breaking its legs this way.
Usually the reptile tries to bite through the tendons on the prey’s legs to
immobilize it.
Adult zahhaks have no enemies – they are at the top of the food pyramid. However,
young reptiles are eaten by carnivorous mammals, varaha
and kong. An adult lizard fights off large animals by lashing its tail and biting.
Thanks to the reptilian metabolism, the zahhak eats relatively rarely – if it
eats large invertebrates every other day, then a small vertebrate the size of
a chicken is enough for a week. It eats large prey only once every 2-3 months,
while stuffing its stomach to the full.
The breeding season falls at the beginning of the dry season. By this time,
the male’s crest turns red, and the female, on the contrary, has a lighter coloration.
The male chases the female, guided by the scent left by her. Preparing for mating,
the male displays himself to the female: he straightens his legs, nods and shakes
his skin ridge. When two males meet near one female, they start a fight: rising
on their hind legs, they embrace and try to knock down each other. After mating,
the female lays 5-8 eggs in the soil under the roots of a large tree, in a pile
of leaves or just in an empty hole, buries them and no longer cares about the
offspring. Incubation lasts about two months: the offspring hatch shortly before
the rain season. At this time, the juveniles get enough food – insects and small
rodents. Sexual maturity comes at the age of 4 years, when approximately 70%
of the length of an adult animal is reached. At the age of 6-7 years, having
reached the size typical for this species, young males take part in fights for
females as equals. With smaller body sizes, they had been easily driven away
by larger individuals, who could even eat a young rival.
Life expectancy reaches 60 years.
This reptile species was discovered by Nick, the forum member.
Emerald
goanna (Varanus smaragdinus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: Meganesia, tropical rainforests in the north of the continent.
Monitor lizards as a family have always been characterized by a variety of predatory
and insectivorous species. Herbivorous forms have appeared among them only a
few times in the entire history of their existence. In the Holocene, these were
large monitor lizards living in the Philippines, while the second species is
the Neocene emerald goanna. It originated from the tree-dwelling monitor lizards
of the subgenus Euprepiosaurus from New Guinea, which also sometimes diversify
their diet with leaves and fruits. This goanna is an analogue of the green iguana
in Meganesia, it is the only Neocene arboreal phytophagous monitor lizard.
The emerald goanna reaches large size. It grows up to 1.5 meters, 60% of which
falls on the tail, used as a fifth limb. An adult individual weighs up to 14
kg. The skin has emerald green color, effectively camouflaging the reptile among
the foliage. The emerald goanna is diurnal and leads an arboreal lifestyle,
almost without descending to the ground. Very often, the reptile goes basking,
which takes place on the upper branches illuminated by sunlight. These monitor
lizards are territorial: on one tree there can be only one male and a harem
of 2-4 females, as well as their young, to which adults are quite tolerant.
Emerald monitor lizards live high in the crowns of gallery forests, feed on
fruit pulp, plant leaves and flowers. Of the adaptations that allow these goannas
to eat vegetation, a smaller stomach and a more developed colon, as well as
the presence of a caecum may be noted. This species lacks a pathogenic microflora
in the mouth, since a favorable environment for bacteria is created when monitor
lizards feed on carrion, which is not included in the diet of the emerald goanna.
In case of danger, this reptile rushes down like an iguana – into the water
or into dense foliage; if it is impossible to hide, the monitor lizard can switch
to active defense, lashing the enemy with its tail and inflating the throat
sac to appear larger. For an adult, the greatest danger is posed by large birds
and some predatory marsupials. Young monitor lizards that have not reached a
meter in length most often fall prey of tree-climbing
and pouncing goannas, and tree snakes,
although they are under the care of their parents for some time.
The breeding season begins in early autumn, later than at other goannas. The
male mates with the females living next to him in the same tree. If another
male enters his domain, then a fight takes place between them, in which they
stand on their hind legs and try to overturn each other.
Pregnancy lasts 6 weeks, after which the female descends to the ground and lays
5 eggs in the roots of plants or in a hole dug by her. After 6 months of incubation,
the juveniles hatch, which return to the tree to their parents, where they live
until the sexual maturity comes. Grown-up individuals are banished to other
trees.
The life expectancy of this reptile is 15-20 years. Sexual maturity comes in
the first year of life.
This reptile species was discovered by Gleb_97, the forum member.
Lilliputian
monitor lizard (Varanus pumilio)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: Meganesia, savannas and woodlands.
The warm and humid climate of the Meganesian continent is ideal for many reptiles.
In the Neocene, one of the dominant groups here is monitor lizard family, which
reached the greatest diversity at this time. The adaptive abilities of these
lizards are potent; they easily occupy a variety of niches. The place of the
land mollusk-eating animal remains not empty: here, the descendant of the small
insectivorous monitor lizards of Australia, the lilliputian monitor lizard,
has now settled. In the human era, skinks occupied a similar niche, but monitor
lizards turned out to be competitive enough for such forms to appear among them.
The lilliputian monitor lizard fully corresponds to its name: it belongs to
the number of the smallest monitor lizards of the Neocene. Some individuals
reach 40 cm in length, and the size of the majority is no more than 30 cm. This
species has an elongated body, a relatively short thick tail and small short
legs with pointed claws. The lilliputian monitor lizard has a brick-red color
with black speckles, which makes this monitor lizard poorly visible against
the red sand.
This species is diurnal, most active in the evening hours and cloudy days. It
spends the night in someone else’s burrows, termite mounds, under pieces of
bark or stones. Early in the morning, when the air is still cool, it is often
possible to observe several basking monitor lizards located on the tops of shrubs,
small trees or stones. They feed mainly on snails and less often on insects,
which are actively searched for in moist places – in foliage, in tree roots,
under the bark. Lilliputian monitor lizards, in turn, often fall prey to snakes,
small marsupials, birds and other monitor lizards. In case of danger, this lizard
tries to climb a tree or shrub, hide in a shelter.
The breeding season is not pronounced; females lay eggs at any time of the year.
Pregnancy lasts up to 5 weeks, the female lays from 3 to 10 eggs in an abandoned
termite mound. They are incubated in it for 3 months, after which small monitor
lizards up to 9 centimeters long hatch.
The life expectancy of lilliputian monitor lizard is about 8 years. Newly hatched
ones become sexually mature 10 months after birth.
This reptile species was discovered by Gleb_97, the forum member.
Humbaba
(Varanus humbaba)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: southern and eastern margins of the Mediterranean lowland to the Persian
Ridge, Arabia, northeast to the southern shore of the Fourseas.
Neocene monitor lizards are one of the successful groups of Neocene reptiles.
At the end of the Holocene and during the ice age, some of them died out; the
reason for this was most often – directly or indirectly – human activity. Nevertheless,
in the early Neocene, they not only restored species diversity, but also formed
a new family of aquatic monitor lizards – large semi-aquatic reptiles. Other
species, inhabitants of open spaces, have retained a more familiar way of life.
One of these species is the humbaba, a descendant of the desert monitor (Varanus
griseus).
The length of the humbaba is 2.5 meters, and the weight is about 20 kg. Animals
from the outskirts of the Mediterranean lowland are smaller and slimmer, growing
up to 1.7 m with a weight of 10 kg. The coloration of adult animals is dark
brown, usually with small yellowish spots and specks on the upper side of the
body, the belly and the inner side of the legs are pinkish-gray. The reptile
has a heavy body, powerful limbs and a large short head with small horns behind
the eyes. Blade-like teeth with finely serrated edges grow in the jaws; they
are adapted for cutting meat and cartilage. The animal’s mouth is inhabited
by bacteria, which cause blood poisoning and release toxins (hence the name:
Humbaba is the monster of Sumerian myths).
Humbaba feeds mainly on warm-blooded animals of medium size – from rat-sized
rodents to a harelope. Occasionally this reptile catches small reptiles and
amphibians or ravages bird nests. Humbaba attacks prey from ambush, because
its physiology does not allow for a long active pursuit. The humbaba grabs the
small animal with its teeth and swallows it alive, inflicting wounds on it with
its teeth. The larger prey is first knocked down by a blow of the tail, or the
predator bites prey’s soft side and shakes its head, causing deep wounds with
its teeth. Sometimes an animal that has escaped from the reptile’s jaws dies
from shock and blood loss, or from rapidly progressing sepsis. Therefore, after
such attacks, the reptile follows the escaped prey for some time, waiting for
its inevitable death. This species does not disdain carrion, eating even mummified
corpses along with bones and larvae of necrophagous insects.
Humbaba has nly few natural enemies due to its large size. Young animals most
often fall prey of birds and lizards, including larger relatives. Occasionally,
in lean years, large cats and canids can attack humbaba, but at the same time
they risk getting blood poisoning after an accidental reptile bite. Sometimes
one-toed ostriches also trample
and peck them during migrations.
Mating takes place during the dry season. Male grows a skin fold on the throat,
which it displays to female by walking around it on straightened legs and hissing
loudly. If several males gather near the female, the rivals hiss, whip their
tails and strike each other with horned heads. Teeth are not used during intraspecific
conflicts. The winner presses the defeated one to the ground and holds its neck
for a while, lying on the former rival by stomach or pressing him with the paw.
During the mating, the male twitches his head and rubs his lower jaw against
the female’s neck. After mating, the female buries 15-20 eggs in well-warmed
places. After 3 months, young individuals hatch. They are careful, avoid open
spaces and try to hide in the bushes so as not to get caught by adult animals.
Sexual maturity comes at the age of 4 years with a total body length of about
100 cm, in the Mediterranean subspecies – with a length of 85 cm. Life expectancy
is up to 45 years.
This reptile species was discovered by Nick, the forum member.
River
constrictor (Varanguis truncatus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Monitor lizards (Varanidae)
Habitat: East and Southeast Asia, tropical rivers and lakes.
The disappearance of limbs is quite common phenomenon among the lizards. It
is observed in different families in parallel on different continents and at
different times, and is usually associated with the change of habitats to aquatic
or underground ones. Perhaps the ancestors of the first snakes were lizards
like the skinks of the Holocene epoch passed to an underground lifestyle and
eventually completely lost their legs back in the time of the dinosaurs. The
new way of life helped ancient snakes survive the mass extinction at the end
of the Mesozoic, which exterminated the dinosaurs.
Since then, lizards have repeated this transition quite regularly – for example,
slowworms and skinks of the Holocene epoch, or their Neocene descendants. The
river constrictor is another such attempt made by reptiles of another family.
The river constrictor is a descendant of the Asian water monitor (Varanus salvator).
The Asian water monitor itself was a very large lizard, reaching an average
of 2 m in length with a weight of about 20 kg, but its snake-like descendant
is much larger. The river constrictor reaches the length of about 5 meters with
a weight of up to 100 kg. Externally, the river constrictor resembles large
snakes – it is a heavy reptile with almost completely reduced limbs: from each
of them only one claw remains on the outside, hardly distinguishable among the
scales. The tail of this reptile is about a third of the total length; the head
is narrow and equipped with long jaws. In the upper part of the body, there
is a skin ridge stretching from the middle of the back to the tip of the tail,
expanding on the tail into a kind of trapezoidal vertical caudal fin. This reptile
is not a relative of the water monitors
(Aquavaranidae family), and the similarity in lifestyle and anatomy features
was acquired by them independently from each other.
The color of the river constrictor is not bright, but masking. The skin is olive-brown,
covered with blurry vertical stripes of black and dirty yellow. Frozen among
the underwater vegetation, the reptile is very similar to the tree trunk. The
similarity is complemented by filamentous algae, which often settle on the skin
of the animal, especially on the head and back. During molting, the reptile
dumps the old skin, shedding it in large chunks, and loses its algal camouflage.
However, after a few weeks, the algae thickets are restored again. In young
reptiles, the stripes are clearer than in mature ones, and the color is generally
brighter and more contrasting. The eyes are large, well developed, with a bright
yellow iris and movable eyelids. This species has a forked tongue, sensitive
to substances dissolved in water, and it allows the animal searching for prey
even in the dark or in muddy water. The river constrictor is able to hold its
breath for an hour.
The river constrictor differs from snakes in its mouth structure: its jaws are
more similar to the jaws of other monitor lizards and lack the elastic ligaments
characteristic of snakes. The mouth is full of cutting teeth with a jagged edge,
which cause deep lacerations to the prey. The reptile hunts large land-dwelling
mammals and birds, lying in wait for them at the watering hole and dragging
them into the water, as well as waterfowl and large fish. The river constrictor
can hold prey by wrapping its body around it. The rings of the river constrictor’s
body squeeze the prey’s chest, and at this time the predator bites the prey,
inflicting deadly wounds. Since the river constrictor’s mouth stretches worse
than that of true snakes, it prefers to dismember prey in the process of eating,
holding it with body rings and tearing off small pieces. It swallows small prey
wholly, sometimes even alive; sometimes the teeth break off and remain in the
body of the prey, but new teeth grow quickly enough.
The river constrictor dwells exclusively in reservoirs and moves with difficulty
on the ground. Usually it is forced to move along the ground after floods, finding
itself in too small reservoirs, and also if it needs to overcome a low waterfall.
Occasionally, the reptile can move to the sea and swim across the sea straits,
but it rarely does so. Usually, the reason for the sea voyage of a river constrictor
is a flood or a high population density. This species has settled on coastal
islands, forming populations communicating with the mainland one.
As with many tropical inhabitants, there is no pronounced breeding period in
the river constrictor, and females can be ready to copulation at any time of
year. Often several males meet around one female, and then they actively fight
with each other, wrapping each other in rings and trying to drown the opponent
in the water. Mating is also similar to a struggle – sexual dimorphism in the
river constrictor is poorly expressed, and males almost do not differ from females
externally. The male holds the female by wrapping his tail around her, and his
weight prevents her from swimming, keeping her in shallow water for several
hours.
Pregnancy lasts 5-7 months, and the female gives birth to several live and rather
large juveniles – this is an adaptation caused by the passing to an aquatic
lifestyle. Unlike crocodiles, she does not care about offspring and can eat
her own posterity if they stay too long next to her. The enemies of young individuals
are birds of prey, catfishes and large snakeheads. The offspring becomes sexually
mature at the age of about 10 years, and the average life expectancy of a snake
is 40-60 years.
This reptile species was discovered by Bhut, the forum member.
Crocodile
monitor lizard (Pseudosuchus varanoides)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: rivers of Southern and Eastern Africa, south of Zinj Land.

Picture by Sauron from FurNation
After the ice age number of crocodiles, last representatives
of prospering in far past Archosauria subclass, had sharply reduced and they
have died
out practically at all area. But in warm and damp climate of Neocene when
many
finny rivers and lakes have appeared, the place of large fish-eating predators
did not remain empty: it was occupied with representatives of large monitor
lizards. Once in Cretaceous period monitor lizards have already achieved
short-term success, having evolved to predatory mososaurs – true horror
of seas. In Neocene
the opportunity to make the second attempt has appeared at them, and they
again have taken advantage of it, becoming ancestors of separate family
of water
monitors, including predators similar to crocodiles of last geological epoch.
They are settled in rainy tropic areas of Old World: in Africa, at East-African
subcontinent, south of Arabia, Hindustan, in South-Eastern Asia and Indonesia.
In Nile which in Neocene had changed the channel and flows across former
Sahara desert as in prehistoric times, representatives of water monitors
are not found:
there they are replaced by other predatory river reptile, huge
crocoturtle (Archotrionyx vorax), the representative of soft-shelled
turtles.
At the coast of Tanganyika passage separating the East-African subcontinent
from Africa, the crocodile monitor lizard, one of widespread species of this
family, lives. It is the huge water reptile (its length is up to 5 - 6 meters),
externally similar to crocodile with longer neck and without characteristic
skin osseous plates. The skull of crocodile monitor lizard is more than half
meter long. It is rather wide and flat. Eyes of reptile are shifted in top
part of head and stick up above it a little. Haw has a role of “underwater
glasses”, permitting monitor lizard to see normally under water. Nostrils
of crocodile monitor lizard open at the end of muzzle that allows the reptile
to breathe, only minimally displaying itself for catch. Edges of nostrils
can
close automatically at diving. Under water this monitor lizard finds possible
catch with the help of sight and strongly advanced chemical feeling: the
reptile from time to time analyzes water by tongue like snake.
Paws of crocodile monitor lizard are well advanced, animal easily moves on
the ground. On toes claws, with which help monitor lizard tears apart large
catch, are advanced. Palamas between toes of reptile are not present. The
main swimming organ at the crocodile monitor lizard is long tail (its length
is
about half of general length of reptile). On tail the skinny edge is advanced,
stretching up to middle of back. The skin of this monitor lizard is colored
greenish-grey with pattern of longitudinal lines of darkly-green spots. Stomach,
chest and throat are covered with soft yellowish-white skin.
Crocodile monitor lizard is active predator and scavenger. Its basic forage
includes fishes, but the reptile equally willingly eats ground animals, for
any reasons got in water, and during river floods searches corpses of sunk
animals. Teeth of this reptile, as against to crocodile’s ones, resemble
more teeth of shark: they are leaf-shaped with sharp saw-toothed edges. Attacking
large animal, reptile puts to it deep cut wounds by teeth.
These reptiles are rather tolerant to each other, and frequently it is possible
to see, as they gather on riverbank by small even-aged groups. The most part
of day adult crocodile monitor lizards bask at the sun. If they become hot,
huge lizards slip in water, wet skin and creep out to riverbank again, or
hide in shadow of trees, or wallow in dirt. But such picture can be observed
only
when reptiles are full. Hungry crocodile monitor lizard is dangerous creature.
It arranges ambush on shallow, near to favorite watering places of ground
animals. Usually this reptile lays at the bottom, occasionally putting out
from water
tip of muzzle to breathe. Through river bottom monitor lizard feels steps
of animals going to watering place. When the catch is located, monitor lizard
cautiously creeps up in bottom as close as it is possible to animals drinking
water, and makes sharp throw, trying to seize the chosen prey to the muzzle.
If the attack is successful, reptile jerkily fells catch and drags it in
water
to drown. Thus it strongly grips it by jaws, not giving to prey any chance
to escape.
Young crocodile monitor lizards eat at first insects, and then pass to more
essential catch: frogs, small fishes, birds and animals had fallen in water.
Crocodile monitor lizard eats exterminated catch alone when it is not so
large. But if carcass is too large, it should hasten: the successful hunter
is immediately
surrounded by hungry neighbours, and everyone strives to bite off from another's
catch richer piece. Sometimes near catch serious fight springs up. But teeth
are not used, in other case after each such conflict the population promptly
would decrease. Monitor lizards fight, pushing away opponents by all body,
striking impacts by head and scratching by claws. Teeth are used only for
demonstration of force: driving away neighbours from lawful catch, crocodile
monitor lizard
widely opens mouth, showing lines of sharp teeth, but does not try to bite
the contender.
Being adapted to life in water, the crocodile monitor lizard confidently
feels on land. It easily walks, and can even run to small distances. Sometimes
it
hunts on land, hiding near track of large animals. Here excellent masking
colouring helps reptile. It attacks nothing suspecting prey when it walks
beside. Sharply
having turned around, monitor lizard strikes blow of tail on legs of prey,
and then, having turned to it by head and rushes on animal trying to rise,
seizing its neck or thorax. Even if the prey will escape, it is practically
doomed: teeth of crocodile monitor lizard put to prey such wounds, that it
quickly perishes from loss of blood. And the reptile easily finds wounded
prey by blood trace. As against water turtles, the crocodile monitor lizard
easily
can swallow catch outside of water, and teeth with which it is possible to
bite off suitable piece easily, favorably distinguish it from crocodiles.
Crocodile is compelled to snap for its any sticking outside part and to turned
around
axis to dismember large catch. And such trick succeeds only in water.
The crocodile monitor lizard is very enduring, and it can even stay short
time in sea water. Sometimes on “rafts” of floating trees these reptiles
can be
carried away far to the ocean, receiving an opportunity of settle at islands.
They had crossed this way Tanganyika passage and have occupied the East-African
subcontinent, and also had settled in Southern Arabia. At the East-African
subcontinent (Zinj Land) this species had formed the separate dwarf
form (Pseudosuchus varanoides carcinivorus) up
to 3 meters long. The dwarf crocodile monitor lizard eats mainly fishes and
freshwater crabs.
The breeding season at these reptiles stretches almost for all year, except
for dry season at edges of natural habitat. Usually the male (on the average
it is about meter longer than the female) begins caring: it chases female
ready to pairing, seizing by paws for the basis of her tail (usually males
of lizards
keep females by teeth, but teeth of crocodile monitor lizard can put painful
wounds). If the female is not ready to accept caring of the male, it can
shake him from herself, having swept by the ground or having swam through
driftwood.
If some males try to care to one female, there can be severe fights between
them: contenders push each other away from the female, strike impacts by
head and scratch by claws. If the competition becomes especially sharp, teeth
are
used and then fight may terminate in heavy traumas. In colonies of crocodile
monitor lizards it is quite often possible to see animals with bitten off
toes, injured skin and knocked out eyes, crippled during courtship tournaments.
Caring for the female, male pays to her signs of attention: it rubs against
her head and throat. On riverbank male lays near to female, driving away
from her probable competitors and when the female leaves in water, follows
her.
Pairing of reptiles takes place in water and repeats some times. After pairing
male some time stays near to the female.
Pregnancy at crocodile monitor lizard lasts about three weeks. When the female
feels, that there comes time to lay eggs, she leaves riverbank and searches
for secluded place in forest. In moisturized and friable wood ground she
digs out by hinder leg hole about half meter depth, and lays in it about
two tens
eggs by size like goose ones. When all eggs are laid, female digs nest in
and by bottom jaw rakes up atop leaves and branches of it. The care of posterity
at this species of reptiles comes to an end at this moment, and female leaves
nest.
The incubating lasts about two months. The length of just hatched young retile
is only about 25 cm. By colouring it differs from adults: its skin is emerald-green
with faltering cross strips of black color. Fin plica on tail is well advanced,
and the young growth is able to swim and dive since the first day of life.
But the first day of life is the hardest for the future river tyrant.
Young monitor lizards should fulfill very hard task: to dig independently
the exit to the surface, and then to find way to water. It succeeds not to
all
ones: if the place for nest is chosen unsuccessfully, the overflow after
rain river may simply inundate nest. A part of clutches may be founded and
dug out
by predatory animals, they destroy part of young monitors. In the river both
predatory fishes and adult neighbours hunt young crocodile monitor lizards.
Therefore the just hatched young monitor lizards searches shelter from cannibal
neighbours in thickets of marsh plants where adult crocodile monitor lizard
is not able to creep through. In total only posterity from one third of laid
eggs survive up to monthly age. But at two-year-old age they reach one-and-a-half-meter
length, and many of former enemies easily can turn to catch. And the eight-year
reptile being about four meters long has practically no enemies. At the same
age crocodile monitor lizards can breed. Life expectancy of this reptile
can reach 70 years.
Montane
water monitor (Pseudosuchus nhang)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: mountains of East Africa (Zinj Land).
Monitor lizards represent one of the most successful lizards on Earth; even
in the Mesozoic, prehistoric seas were plowed by mosasaurs – giant sea lizards,
relatives of monitor lizards of the Cenozoic. The monitor lizards themselves
never adapted to aquatic lifestyle deeply, until their descendants appeared
in the early Neocene, belonging to the water monitor family, the inhabitants
of reservoirs in Africa and Asia. Usually representatives of this group are
large lizards resembling crocodiles and mosasaurs. In the mountains of East
Africa microcontinent (Zinj Land) one of the smaller species lives – the montane
water monitor. In appearance, it is a scaled copy of a larger
mainland species from Central Africa – it is a relic of the early evolution
of this group of reptiles, preserved in isolation on the East African microcontinent.
The montane water monitor grows no more than 150 cm in length, of which a little
more than half falls on the tail. At the montane water monitor it is long, strong
and compressed from the sides – it is the main propulsion when swimming, while
the legs are pressed to the sides and are used only when turning and when moving
on land. The legs are short, but strong and well-developed, and the montane
water monitor walks equally good on the ground and on the river bottom, clinging
to the stones with its claws. The coloration is not green, as in most representatives
of this genus, but more motley – ochre-brown with a black irregular mesh pattern,
which makes the montane water monitor less noticeable both under water in mountain
reservoirs and on their rocky shores. This species has few enemies in the mountains
of Zinj Land, and the behavior of this lizard is relatively primitive.
There are no deep rivers in the mountains, and the montane water monitor avoids
rivers with particularly fast currents, preferring shallower reservoirs, not
deep rivers and lakes where the water warms up better. In cold water, the reptile
quickly loses mobility and must bask on the shore for a long time before diving
again. Being a predator, this species is territorial and aggressive towards
its relatives, especially outside the breeding season. The food of this species
includes fish, frogs and crustaceans. It also eats rodents and waterfowl chicks.
In mountain reservoirs, this species is the apex predator, and it has very few
enemies. Occasionally it falls an accidental prey of large snakes, birds of
prey and carnivorous mammals. Sometimes montane water monitors die in local
disasters like rockfalls and earthquakes. If the territory appears to be unsuitable
as a place for life, the reptile can undertake long journeys overland in search
of new habitats.
The courtship behavior involves males fighting for a female and the chase of
a female by a male on land and in water. There are 15-20 eggs in the clutch
of the montane water monitor, which the female lays for one to two weeks in
a small nest arranged in the ground. The male does not take part in the parental
care, and the female guards only the territory where the nest is located. After
45-50 days, young reptiles hatch and immediately begin an independent life,
trying to get away from the mother’s possessions – she can easily eat them.
During the search for territory and in the first years of life, most young individuals
die. If the montane water monitor does not die in its youth and grows to at
least 60 cm, it has every opportunity to live for several decades. However,
only a few individuals survive to old age.
This reptile species was discovered by Bhut, the forum member.
Gavialodon
(Gavialodon angustirostris)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: Congo Basin.
The water monitor family in the fauna of the Neocene is an analogue of crocodiles
of the human epoch. Having suffered from human activity in the historical epoch,
crocodiles are represented only by few species in the Neocene. And water monitors
have become the dominant predators in fresh waters, having occupied the top
of the food pyramid in ecosystems. Their competitors in Africa are large
carnivorous turtles, and the water
monitor species from the rivers of West Africa have developed the certain
features of specialization, avoiding competition with turtles. One of the specialized
species of water monitors is gavialodon, a relative of the crocodile monitor
lizard that inhabits the Congo River and its tributaries. It is a specialized
fish-eating reptile, although sometimes it can catch small reptiles and amphibians
(including tadpoles).
The body length of an adult gavialodon reaches 4.5 meters; the male is almost
one meter longer than the female, but the female has a more massive build. Externally,
this species resembles a crocodile monitor lizard, but is of darker color and
has a long rostrum: the length of the jaws is three times their width at the
base. The teeth are numerous, thin and needle-shaped. The basic body color is
dark green with lighter narrow transverse stripes on the back. The belly is
yellowish-white.
Gavialodon is deeply adapted to an aquatic lifestyle and spends a lot of time
in the water. Its legs are somewhat reduced; they retain five digits and a small
membrane between them, but are no longer able to lift the body off the ground
and hold it in this position for a long time. In the water, the gavialodon is
quite agile, dives well and can hold its breath for half an hour or more. The
tail of the animal is bordered by a large skin plica that stretches from the
loin almost to the cloaca. While swimming, the reptile presses its limbs to
its sides and bends its whole body. Only in shallow water and among plant thickets
the animal moves with the help of limbs. This predator rarely gets out on land
outside of the breeding season, preferring to bask in the shallows and climbing
half-sunken fallen trees.
The teeth allow the gavialodon hunting small fish effectively. Usually during
the hunt, the reptile lies in ambush at the river bottom. When a shoal of fish
swims by, the gavialodon opens its mouth slightly and makes a sharp lateral
movement with its head. Fishes impale themselves upon its teeth or get injured
and sink to the bottom, where they are gathered by a predator. In addition,
gavialodon eats frogs and grabs waterfowl and their chicks from under the water.
In addition to small vertebrates, it catches arthropods got in the water and
does not disdain rotten carrion.
The breeding season of this species stretches for the whole year. Males display
their claims to the territory by swimming near the water surface and slapping
their tails loudly. The male chases a female ready to reproduce and holds her
in the water, displaying his strength to her. The female ready for mating shows
submission by stopping and sinking to the bottom under the pressure of the male’s
body. If she is not ready to reproduce or does not like the male, she resists
actively, pressing the male to the bottom and pounding him with her tail. During
intraspecific conflicts, jaws are not used because of the fragility of bones
and teeth. The male has a harem of several females and protects it from other
males, pushing them away with his body and tail blows. 20 days after mating,
the female lays up to a dozen eggs. To do it, she gets ashore at night, digs
a nest 3-5 m from the water with one of her hind legs and covers the eggs with
sand and leaves. The clutch is not guarded; during the year, the female can
make up to three clutches, after which the break of up to six months follows,
when the female’s body recovers to prepare to a new breeding cycle. After 8
weeks, young gavialodons hatch from the eggs. They get out of the nest and immediately
crawl into the water. At first, young animals hunt insects and shrimps, and
later switch to adult food. Avoiding encounters with large predators, they migrate
to shallow rivers and descend into large rivers as they mature.
The gavialodon lives up to 70 years and reaches sexual maturity at a length
of 3 m, at the age of about 10 years.
This reptile species was discovered by Nick, the forum member.
Ganges
aquavaranus (Aquavaranus gangeticus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: South Asia, rivers of Hindustan.
The warm climate of the Neocene and the extinction of many groups of mammals
contributed to the emergence of greater diversity in the Neocene reptiles. Thus,
due to constant pressure from humans and the environmental consequences of their
activities, almost all aquatic forms of mammals disappear. However, their niche
has not remained empty: now rivers and lakes are occupied by representatives
of the water monitor family. One of them, the Ganges aquavaranus, settled in
the large river systems of South Asia – in the rivers of the Hindustan peninsula,
where the majestic Indus and Ganges flowed in the human era.
This monitor lizard has almost completely lost contact with land – only females
return there to lay eggs. It has a snake-like body shape, reduced webbed limbs
and a long tail that it uses as a rudder. In the water, the aquavaranus is quite
agile: it moves like a snake, wriggling its whole body. Despite the great similarity
of aquavaranus with mosasaurs, the former did not adapt so well to life in the
water. In particular, the water monitor family had not developed live-bearing
yet, which their prehistoric predecessors managed to do.
Record specimens of males reach a length of 6 meters; females are much smaller,
growing up to 4 meters. This monitor is colored dirty grayish-brown and greenish.
This species has relatively small eyes; the tip of the monitor’s muzzle is equipped
with numerous effectors, making it very sensitive. This feature indicates that
the Ganges aquavaranus lives in muddy water and rarely uses vision, using mainly
the sense of smell. The basis of the diet of this predator is fish and shellfish.
In addition, the water monitor often feeds on waterfowl, animals crossing the
river and carrion. Adults, whose length has exceeded two meters, have enemies
only in the face of their larger relatives. Each individual occupies its own
territory, beyond which it almost never goes. These water monitors are very
fiercely guarding their sites. If someone violates the border, a fierce fight
takes place between the reptiles, during which many fighters remain with ripped
or torn off tails and fins, or with broken jaws. This very often affects females,
which, due to their smaller size, cannot give a decent rebuff to large mature
males.
The courtship season falls in early spring; at this time the males leave their
sites and begin the search of females. For this short period, they are tolerant
of females, but relate with even greater malice towards other males. Having
found the female, the male behaves extremely aggressively towards competitors.
The largest and strongest of them drives the other males away from the territory
where mating takes place.
Pregnancy lasts from two to four months. The female spends more time basking
in the shallows and near the surface of the water, and feeds more often than
the male. At the very beginning of summer, many females come ashore, where eggs
are laid. On land, these animals are clumsy and helpless, but because of their
large size, no one dares to attack these monitors. In addition, reptiles defend
themselves from enemies with bites and tail blows.
The nest is a simple hole in the sand, which the female digs out with the movements
of her hind limbs, and buries with lateral movements of the body. After laying
eggs, the females return to the water. The number of eggs can vary from 8 to
25. The female does not guard the clutch, so more than half of the eggs are
eaten by small predators. Hatching of young animals occurs after 4 months; juveniles
independently overcome a long way from the place of hatching to the water.
The life span of the Ganges aquavaranus can reach 60 years; they become capable
of reproduction five years after hatching.
This reptile species was discovered by Gleb_97, the forum member.
Mangrove
aquavaranus (Aquavaranus mangrophylus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: South Asia, mangrove forests, estuaries; coasts of the Bay of Bengal
and the South China Sea
Holocene reptiles were represented mainly by land animals, since many niches
in the water were already occupied by mammals. Despite this, many reptiles,
including lepidosaurs, have much more advantages over their warm-blooded competitors.
In the Mesozoic, it was they, not dinosaurs, who conquered the sea and reigned
there for more than 180 million years. In the Neocene, they again attempted
to conquer aquatic habitats, giving rise to water monitor family, but so far
these reptiles are unable to completely break off contact with land – they are
still returning to the shore to lay eggs.
The sizes of different subspecies of mangrove aquavaranus vary greatly: the
nominate subspecies reaches a length of 3 meters, while the coastal subspecies
grows up to 7 meters. The mangrove aquavaranus has a body even closer in shape
to the snake than its relatives. It helps this one to move better among the
thick roots and trunks of mangrove trees, as well as penetrate into the cracks
where fish can hide. This monitor lizard is colored yellowish and greenish tones,
the belly and large spots on the tail are white. Unlike the Ganges
aquavaranus, this species lives in salty and brackish water. It gets rid
of excess salts due to well-developed salt glands. The reptile is not picky
in food; all animals suitable in size fall on the menu – most often these are
fish, shellfish and crustaceans abundant in its habitats. The water monitor
cannot always keep up with the fish, so most often it hides at the bottom or
among the roots, from where it makes a rush at the right moment. In an ambush
under water, it can stay up to half an hour, having previously taken air into
its lungs. The mangrove aquavaranus is not as aggressive as its freshwater relative
is, but skirmishes often occur between individuals of this species due to the
protection of the territory or females. The consequences of fights are not very
serious – most often, the weaker individual just leaves.
The breeding season begins in March or April. Males quickly find females by
scent. As a rule, having found her, they meet several more competitors. The
strongest male drives away the smallest individuals by his appearance alone;
a fight occurs with the largest ones who do not want to give way without a fight.
The winning individual has the right to mate with a female.
Pregnancy lasts more than three months; in early summer, the female lays up
to 18 eggs on the shore above the highest tide level. The embryos in them are
already well formed: they were actively developing even when the eggs were in
the oviducts. A month passes before hatching, after that many newly hatched
monitor lizards move towards the water. Along the way, many of them will be
eaten by birds, another part will die already at sea. Only a few pairs of juveniles
from the brood will eventually reach maturity.
These reptiles live from 40 to 65 years; they begin to reproduce five years
after birth.
The species has two well-distinguished subspecies:
The common mangrove aquavaranus (A. m. mangrophylus) lives
in brackish reservoirs of South Asia and prefers mangrove forests and estuaries.
Sometimes this subspecies enters rivers upstream, including the rivers of Hindustan,
where it can meet with a larger Ganges aquavaranus. In Southeast Asia, it is
replaced by another species – the pike monitor
lizard. It grows up to 3 meters.
The coastal aquavaranus (A. m. litus) is a much larger subspecies
with a wide range. It lives in the Bay of Bengal and the South China Sea, along
the coasts of India, the Moluccas Peninsula, the islands of Kalimantan, Sumatra,
Sri Lanka. This form lives only in salt water, never entering rivers, and reaches
a length of 7 meters.
This reptile species was discovered by Gleb_97, the forum member.
Mollusk-eating
aquavaranus (Aquavaranus malacophagus)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: freshwater reservoirs of South and Southeast Asia
Monitors represent a very successful and diverse group of reptiles. Generalists
among them are medium-sized lizards that feed on small vertebrates and insects.
Atypical representatives include several Neocene aquatic forms that evolved
as a result of the decline of aquatic mammals and crocodiles at the boundary
of the Holocene and Neocene. One of the most bizarre species is the mollusk-eating
aquavaranus, specialized in feeding on shellfish. Like many other Neocene animals,
the mollusk-eating aquavaranus is not unique in its kind: it just repeated the
history of its relatives. One of the most famous malacophage varanoids of the
past is the Globidens mosasaurus, which lived at the end of the Mesozoic. Due
to the same lifestyle, they are very similar in appearance – both are characterized
by wide blunted crushing teeth and a large head with very powerful jaw muscles.
Some monitor lizards of the human epoch had similar adaptations – the African
species Varanus exanthematicus and several related species.
The mollusk-eating aquavaranus is not the largest representative of the family:
separate individuals reach a length of 3.5 meters. It has stockier proportions,
a powerful tail and a large head. In the jaws of this monitor lizard, there
are rounded teeth that allow it crushing the hard integuments of invertebrates.
It is covered with fairly strong scales, which serves as a passive defense against
predators. If necessary, this monitor lizard is capable of inflicting strong
bites – its jaw muscles are very powerful. The mollusk-eating aquavaranus has
a coloration of soft gray and dark green tones – spots forming an irregular
cross-striped pattern are scattered on the back on a greenish background. It
is very slow, because it does not need to chase prey. This species prefers to
stay away from the shore or at the bottom to avoid encountering other Aquavaranus
species. It searches for prey with the help of a long forked tongue and a Jacobson’s
organ, and is able to hold its breath for 30 minutes or more. The diet of this
species includes mainly benthic animals with good protection like shells – crustaceans,
bivalves and gastropods. Sometimes it eats dead fish, small turtles and carrion.
Mollusk-eating aquavaranus is peaceful towards its relatives; fights between
males happen very rarely and end without serious consequences. The breeding
season lasts the whole spring.
Up to two months pass from mating to egg laying. In the middle or late summer,
many pregnant females can be observed coming ashore to lay eggs. At one time,
the female lays up to 30 eggs, more than half of which are eaten by small predators
later. Juveniles begin hatching after 2-3 months. They are well protected by
armor from small predators. For the first few weeks, young aquavaranuses stay
close to the shore, but later they need to leave coastal waters, since they
can easily fall prey of large Ganges aquavaranuses.
The advanced age of these aquavarans comes to 40 years, but some individuals
live up to 50 years. They become sexually mature at the age of four.
This reptile species was discovered by Gleb_97, the forum member.
Pike
monitor lizard (Varanesox velonatans)
Order: Squamates (Squamata), suborder Lizards (Lacertilia)
Family: Water monitors (Aquavaranidae)
Habitat: islands of Indonesia, South-Eastern Asia.

Picture by Roman Val
Initial picture by Arseny Zolotnikov |
Change of climatic
conditions and natural accidents accompanying them were reflected at the
reproduction of plankton by the most pernicious way. Biological
efficiency of seas had sharply decreased, that had caused extinction of most
part of coastal fish-eating organisms. But ecosystems of rivers not so strongly
depend on sea plankton, and only separate species of animals had suffered
here. General efficiency of rivers had practically not decreased.
It had permitted to some species of terrestrial animals to make attempt to
pass to aquatic habit of life. In Indonesia conditions had especially favored
to it. At the ice age the ocean level was lowered, and islands had merged
to some common areas of land that had allowed to ground animal to settle
widely.
Among successful migrants there were various reptiles, including monitor
lizards. When glaciers have thawed, the common land appeared divided to set
of islands.
Due to plentiful rains at islands the set of rivers had appeared. One of
optimum ways of adaptation to this change of inhabitancy was the passing
to life in
water.
Coast estuaries are rich in life – here larvae of fishes growing up in brackish
water and small shrimps meet, amphibians and insects live in freshwater sites
of flowing rivers, and also there are different species of waterfowl. Medium-sized
lizards of monitor lizards family had started development of this promising
inhabitancy, having evolved to essentially new form – pike monitor lizard.
This almost completely aquatic lizard most advantageously lives in shallow
warm gulfs. Its close relative is African crocodile
monitor lizard (Pseudosuchus varanoides). It differs in larger sizes
being less adapted to aquatic habit of life.
The pike monitor lizard, on the contrary, spends significant part of time
in water. It has streamline body shape, but has kept many features of common
terrestrial
reptiles. Main movement organ at this lizard is the tail; it is short and
compressed from sides with oblong fin at the end. Extremities are rather
small, flatten
like crocodile paws, having claws at fingers and toes. Hinder legs are larger
than front ones. At the land pike monitor lizard moves clumsily and reluctantly;
it does not creep far from reservoir.
Pike monitor lizard reaches one and half meters long, but more often it is
smaller. The skin is covered with smooth, small, densely fitted scales colored
dark green with cryptic vertical stripes of grayish-yellow color.
Eyes and nostrils are shifted to the top part of muzzle that permits to look
over vicinities, only slightly having put out muzzle from water. Head of
the reptile is rather large: approximately the fifth part of the general
length
of animal. Neck is almost not expressed.
This reptile swims by means of tail and paws. Certainly, in quickness this
monitor lizard concedes to fishes, however it successfully hunts them from
an ambush. Usually the reptile hides among water plants, or driftwood. Thoroughly
having recovered breath and having inhaled air in lungs, it can stop breathe
during approximately quarter of hour. Thus its blood circulation is switched
in such manner that blood supplies first of all brain and heart. Muscles
exist due to internal stocks of chemically fixed oxygen.
Less often this monitor lizard attacks amphibians and nestlings of waterfowl,
and also eats carrion.
It swallows small catch entirely, taking it to swallow from head. This reptile
can bring large fishes or dead animal which cannot be swallowed at once,
for convenience to shallow water, and then tears it apart by teeth and claws
of
forepaws.
In the morning, or after meal, pike monitor lizard quite often creeps out
of water to get warm on driftwood or to sandy spit in river channel, keeping
up
for environmental conditions. On land this reptile behaves very cautiously.
Any sharp sound or suspicious movement it regards as danger, rushing in water
and hide deeply in thickets of underwater plants. Being caught, this monitor
lizard is able to defend itself actively, biting by mouth full of rather
large needle-like sharp teeth.
Courtship season at this species takes place rather roughly, that is especially
remarkable for such cautious animal. Pairing is preceded with tournaments
in the water, accompanying with jumps and fountains of splashes. At this
time
males chase and bite each other by mouth, trying slightly subsurface the
contender, showing the superiority. Loosed one stops resistance, having pressed
paws to
sides, and when the competitor male lets it off, immediately retires.
Appreciating the origin, for egg laying this reptile comes out to land. Pike
monitor lizard is egg-laying reptile. In clutch it happens up to ten eggs
by size like small chicken ones. Females of this species dig eggs by back
paws
in coastal ground above line of inflow. For breeding these monitor lizards
gather at small islands inaccessible to the majority of ground predators.
Nevertheless, the specific habit of life has already an effect for features
of breeding of these reptiles. Term of incubating of laid eggs at pike monitor
lizard is less, than at overland reptiles of the similar size. It is connected
with the feature of its development: development of embryo begins still long
before egg laying, in organism of mother. Pregnant females become inactive,
and spend more time in well warmed up gulfs. At this time they eat much less,
and frequently meet in groups up to ten reptiles and more.
Young pike monitor lizards, distinguishing from adults by brighter spotty
colouring, live in thickets of plants in shallow water, catching insects,
larvae of fishes
and small frogs. They keep up-stream to avoid the possible attack of adult
reptiles of this species. Right after hatching the young growth aspires to
get in rivers, instinctively moving against current. The greatest mortality
among young animals is observed at this particular time: fishes and adult
pike monitor lizards attack young ones. Due to smaller weight young pike
monitor
lizards move overland, passing through watersheds and settling in other rivers
easily. Adult reptiles are usually adhered to places of inhabiting though
among them there are some “tramps” overcoming even rather narrow sea passages
between
islands.
It is possible, that in more long-term future descendants of this monitor
lizard may at all lose connection with land and pass to life in high sea,
taken place
of dolphins and large predatory fishes in tropical seas of Earth.
This species was discovered by Arseny Zolotnikov
Underground
crown-tailed snake (Coronocauda caeca)
Order: Squamates (Squamata), suborder Snakes (Srepentes)
Family: Blind snakes (Typhlopidae)
Habitat: Hawaiian Islands, rainforests.
Hawaiian Islands are the land of volcanic origin separated from continents by
extensive water spaces. By virtue of reliable isolation on islands some groups
of animals typical for continents were abscent. So, at these islands there were
no ground tetrapods (except for birds): Pacific Ocean appeared practically insuperable
to them. But till the human colonisation of islands the situation had sharply
changed: similarly to many other islands, Hawaii became a shelter for set of
animals from various continents imported intentionally or casually. Their presence
had completely changed a direction of evolution of local flora and fauna, and
many species had completely disappeared. But nevertheless as a result in Neocene
at Hawaii the balanced but absolutely another ecosystem had generated, rather
than it could develop by natural way.
In the ground of Hawaiian rainforests the set of burrows forming extended circuits
is dug. They were dug out by local digging rodents, Hawaiian false mole rats,
similar to large-headed moles. In tunnels they are protected from ground predators,
but they never disappear from other creature also leading underground habit
of life. Pinkish body of this creature almost silently slides in tunnels of
rodents, and sharp sense of smell prompts this predator where it may search
for holes in which young rodents hide.
Tireless hunter for rodents, living at Hawaii, belongs to the primitivest snakes
on Earth – blind snakes. But by size this creature surpasses any of its continental
relatives – its length reaches one meter at thickness about 6 cm. Flattened
head of this snake is covered with thick corneous armour, and tail, on the contrary,
is obtuse. The tip of tail is topped with flat corneous “incrustation” with
small denticles at the edges, directed sideways and back. This formation looks
similarly to crown, and for that snake is named crown-tailed snake.
Present species of reptiles is the descendant of bramin’s blind snake (Typhlops
braminus) or “pottery snake” – small blind snake about 10 – 12 cm long, casually
introduced to Hawaii with indoor plants. At the islands this small creature
had received a fine food source as insects, including introduced ones. Its descendant
has reached length of one meter – millions years of evolution practically out
of competition had an effect. Large descendants of bramin’s blind snake passed
to feeding with small vertebrates including their own congeners. Gradually they
began to include in diet rodents digging in ground – descendants of mice introduced
to Hawaii. Underground crown-tailed snake became the largest digging creature
at islands.
Sight had not any significant role in life of ancestors of this creature, therefore
eyes of crown-tailed snake had completely disappeared, and even their seen traces
did not remain. Head of this reptile is covered with integral corneous shell,
becoming the fine instrument hole burrowing in wood ground. In behavior of blind
snake living under ground, colouring of body also does not matter, therefore
covers of body of this reptile became semi-translucent, pinkish-yellow. Skin
is covered with thin scales forming integral smooth cover. On the tip of tail
of snake the corneous scute with denticles sticking back on edges, similar to
crown, grows. This formation was developed as a support at movement under ground
– snake supporst by denticles against walls of hole. Usually crown-tailed snake
lives in holes of rodents, but easily burrows own holes in friable wood ground.
On ground surface this snake becomes absolutely helpless: crown-tailed snake
tries to find a point of support for tail to push and to burrow in the ground
again. Its muscles are very strong; therefore snake literally “dives” into the
ground and quickly digs in.
Crown-tailed snake has kept predating habits characteristic for all snakes.
It eats small rodents, Hawaiian false mole rats, chasing them in their own holes.
Keen chemical feeling helps crown-tailed snake to search for rodents by smell.
Teeth of this snake are sharp and recurved. Having grasped the catch, snake
keeps it in teeth while catch bleeds to death, and then swallows it. It equally
easily swallows catch both from head, and from tail.
Similarly to the majority of snakes the crown-tailed snake is oviparous species.
Once per half-year female lays up to five lengthened eggs covered with thin
pergamen-like shell. Males of this species may be met rather seldom. They are
longer than females, but differ in narrower head. Male finds female ready to
pairing by the special smell. For pairing snakes bury deeply in ground, and
arrange under tree roots the special hole ending by spherical chamber. In this
chamber male and female twist as if a knot, and spend some hours in such position
while pairing lasts. But as a rule, these snakes have parthenogenetic breeding
without participation of males. Stimulating the increase of hormonal level,
females simulate pairing, gathering by some individuals in the certain similarity
of “marriage holes”. Such behaviour promotes development of ovocites, and approximately
after two weeks females lay eggs. Female hides eggs in hole, digging it and
stamping ground by pushes of head.
Approximately in ten weeks from eggs young snakes hatch. They at once leave
parental holes and dig own burrows: adult snakes frequently eat their own posterity.
Young snakes become able to breeding at the age of about one year. First clutches
are parthenogenetic; completely advanced individuals pair with males and lay
normal eggs.
Nyoka
(Nyoka subterranea)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Boas (Boidae)
Habitat: savannas of North Africa, dung heaps of large mammals – ndipinotheres.
Such an interesting group of reptiles as snakes has not lost its diversity in
the Neocene. Among these legless reptiles there are also many unusual forms
– with a relatively small anatomical diversity, they often demonstrate an unusual
way of life. One of the unusual representatives of snakes is nyoka (the name
means “snake” in Swahili) – a descendant of the javelin sand boa (Eryx jaculus),
which lives in the savannas of North Africa near the dung heaps of large mammals,
and feeds mainly on insects.
The body length of the nyoka is about 1 meter, sometimes a little more, with
a thickness of up to 5-6 cm. This species has a well-developed musculature and
a smooth scaly coat. The length of the tail is up to 10 cm, the head is about
the same length, with a narrow hard nose. The tip of the tail is blunt and equipped
with large scales, serving as a support when moving. The eyes are quite large,
black in color. The tongue is long; the jaws are very strong and have large
teeth slightly curved back. An interesting feature of this reptile is the large
movable scales on the back of the head, capable of rising when the reptile inflates
its throat. In males they are bright red, in females they are red-and-black;
also in males they are larger in size.
Nyoka arranges a dwelling next to a dung heap regularly visited by large mammals.
Thickets of bushes serve as a dwelling, but more often it lives in a pile of
stones with narrow cracks between them, or in a hole dug in the ground. Nyoka
often occupies the burrows of rodents, with which it coexists relatively peacefully:
they are not a part of the reptile’s diet. Nyoka feeds at any time of the day
and night, although it is most active during the daytime. The snake crawls on
a manure pile of ndipinotherium,
or bores manure, laying temporary tunnels in it. The basis of the diet consists
of invertebrates living in these dung heaps: termites,
cockroaches, ants, larvae of dung beetles and dipterans, spiders and others.
To catch diverse prey, the animal uses various hunting techniques. Termites,
cockroaches, ants and spiders with relatively thin integuments are grabbed by
the jaws of nyoka. Strong jaws easily crush the chitinous armor of such arthropods;
the snake regurgitates the undigested armors in the form of tightly compressed
balls, which are often found near the manure piles of ndipinotherium. To catch
the horsefly
larva, the snake crawls in the thickness of manure, sticking to the tunnels
it has laid. It swallows soft-bodied larvae whole, having previously killed
them with several bites. The reptile does the same with the larvae of large
dung beetles. It does not attack adult beetles – largely, it is facilitated
by the presence of solid horn-like outgrowths in beetles.
Sometimes nyoka is faced with the fact that the upper layer of the manure has
dried and hardened. In this case, it can catch arthropods creeping around the
dung heap.
The enemies of this species are birds of prey and predatory mammals. The snake
either crawls away from them, or ruffles its scales and hisses, intimidating
the predator. When a herd of herbivores comes to the manure pile, the legless
predator prefers to crawl away and hides in tunnels.
Males are less attached to the dung heaps of ndipinotherium than females. They
prefer to live in rodent burrow systems, eating insects trapped in them, and
at night they crawl across the savannah in search of females. Mating lasts about
an hour and takes place mainly in a burrow or in a manure pile. After mating,
the male crawls away, and the female continues her habitual life. This species
is viviparous, 5 months after mating, the female gives birth to 10-20 juveniles,
which quickly crawl away, and for the first time they do not live in dung heaps,
but in the grass.
Life expectancy is up to 25 years.
This reptile species was discovered by Mamont, the forum member.
Forest
worm-tailed python (Vermicaudophis venator)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Pythons (Pythonidae)
Habitat: Equatorial Africa, tropical forests, wood litter.

Picture by Fanboyphilosopher
In Neocene tropical rainforests had restored in areas occupied
earlier. But they differ from forests known to people. The part of species of
plants and animals had died out irrevocably, and the part of new forest species
represents descendants of rather recent immigrants succeeded to settle in newly
forming ecosystems.
Tropical rainforest represents a fine habitat for reptiles. From forest canopy
up to soil layer under tree roots – it is possible to find reptiles in any level
of forest. The part of them is herbivores, a significant amount of small species
represents insectivores, and large species are predators and hunt vertebrate
animals. Among predatory reptiles snakes dominate, and pythons belong to the
largest snakes of Tropical Africa.
Many specialized species of these snakes live in tropical forests. Some of them
live in crones of trees, others lodge near water. And in wood litter representatives
of original genus of worm-tailed pythons live; these ones are hunters for ground
animals from chicken up to dog in size. The most widespread species of this
genus is forest worm-tailed python, a reptile about 4 meters long.
Worm-tailed pythons differ from other snakes in several very characteristic
attributes of anatomy. Body of these snakes is wide and slightly flattened,
as at adders of genus Bitis known in human epoch. Neck is feebly marked, and
head is flattened. Eyes and nostrils are shifted to the top part of head. It
is connected to inactive way of life of these snakes – worm-tailed pythons spend
a significant part of time having dug in wood litter. The body of snake has
camouflage color with the soft pattern, helping to hide from prey. At forest
worm-tailed python the pattern on back represents yellowish-brown irregular-shaped
spots on dark brown background. These spots are located on body in pairs and
have slightly oblong form and wavy outlines. In the middle of each spot there
is a slanting stroke of dark brown color, and edges of these spots are shaded
with chocolate-brown strokes. When snake lays in wood litter, having slightly
dug in it and having bent its body, this pattern on its back ideally merges
with environmental background. On head of reptile there is a leaf-shaped pattern
of yellowish-brown color. Stomach of worm-tailed python is light yellow or white.
Compared to body tail of these reptiles is sharply narrowed and thin. Its tip
is long, whip-like and very mobile. Its colouring sharply differs from colouring
of other parts of body – it is light pink with thin cross lines. Tail has such
colouring at last 20 – 30 cm of its length. This is an important hunting adaptation
of the snake.
Forest worm-tailed python is dug in wood litter by lateral movements of body
and freezes immoveably, having bent body so, that tail is near to head. This
snake can keep immovability during many hours, waiting for approaching of prey.
At occurrence of ground birds or mammals of suitable size hidden snake starts
to move with a tip of tail, imitating movement of worms and other small animals
in wood litter. Usually large herbivores or carnivores do not pay attention
to this bait, but birds and insectivorous animals or rodents frequently fall
prey of reptile. Worm-tailed python kills the prey by bites or strangles it,
having compressed in rings of body.
At these snakes the sexual dimorphism is clearly expressed: at male tail is
longer, rather than at female and also male is smaller – his general length
is 20 % less, than at female.
Worm-tailed pythons breed at any time of year. Male finds the female by smell
and is coupled with her, twisting her with body rings. Pairing may last till
about some days. At this time snakes are aggressive and bite the animal disturbed
them, but the food behaviour at reptiles is suppressed this time. Pregnancy
of the female lasts till about three months. It lays up to two tens of small
eggs and protects them, having twisted around of clutch. Additionally female
warms clutch with movements of body. The incubation lasts till about one month:
at the moment of egg laying embryos in them are already advanced enough. Young
snakes eat small rodents and insects. They live in bushes and in tree crones,
not competing to adult individuals.
In Equatorial Africa Some species of worm-tailed pythons live:
Simple-tailed
python (Vermicaudophis simplicicaudis) is the most primitive species
of the genus. On its tail there are no areas differing in colouring from other
parts of body. This reptile up to 2 meters long lives in tree crones, but often
moves down on the ground. The body has cylindrical form typical for the majority
of snakes, and also is rather thin. This species allures prey simply with movements
of tail. Colouring of simple-tailed python is grey with a pattern of faltering
black strokes stretched along the back and sides; on sides strokes merge to
lines of dark spots. Small birds, frogs and lizards become prey of this species
more often. The female arranges nest in tree hollow.
Lizard-tailed
python (Vermicaudophis saurocaudis) specializes on feeding on ground
mammals. Length of this reptile is up to four meters. Its colouring represents
a line of large grey spots along the back on reddish-brown background. Tail
of this reptile very precisely imitulates head and body of lizard. It is bright
green, and its tip is expanded, forming leaf-like structure. On lateral faces
of bait tail there is a pair of oculate spots of yellow color with black stroke
in the centre: they imitate eyes of reptile. Various omnivorous mammals and
birds become prey of this species.
Aquatic
worm-tailed python (Vermicaudophis congensis) differs from other species
of genus in a way of life. It is semi-aquatic reptile about 2 meters long which
lives in boggy areas and in the shallow rivers of Congo basin, and eats fishes
and frogs. Like other representatives of genus, this python allures prey by
tail. The body of this species has bluish-grey color with two parallel chains
of large black strokes along the back. The tip of tail is covered with large
scales of black color which form cross lines, imitating the segmented body of
crustacean. This python arranges ambush in thickets of aquatic plants, holding
head near surface of water; the tail of reptile lays on the bottom, among the
vegetative parts. For breeding female leaves water and siuts nest in reeds,
frequently in nests of water birds. The incubation of eggs passes mainly in
body of the female and it protects clutch no more than two weeks. Young individuals
at once start to live and hunt in water. The tip of tail at them is bright red:
it is an imitation of benthic larvae of some mosquitoes. As snake grows, the
tail changes color to yellowish, and further darkens, turnting color, characteristic
for adult individuals.
Oar-tailed
python (Alopicaudophis percussicaudis)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Pythons (Pythonidae)
Habitat: Africa, the Congo basin, the rivers of West Africa, the lower reaches
of the Saharan Nile.
In the human era, the species diversity of large reptiles decreased due to habitat
destruction, hunting and a reduction of the populations of prey. In the Neocene
epoch, after the retreat of glaciers, warming and restoration of the area of
suitable habitats, a gradual increase in the species diversity of reptiles began,
and large forms began to evolve among them again. It was also a time of developing
new life strategies and ways of hunting. Among African pythons, a genus of worm-tailed
pythons (Vermicaudophis), using their tails as bait for prey, appeared.
A related genus of pythons uses the tail as the main hunting tool – these are
oar-tailed pythons.
The oar-tailed python is a robust semi-aquatic snake inhabiting large bodies
of water with a slow current in the tropics of Africa. The body length is no
more than 4 meters, and up to a third of the body falls on the tail; the male
is about 20% shorter than the female. This snake has a very characteristic appearance:
the body is massive, and the tail is flattened from the sides – like at thresher
shark (Alopias) of the human era. The back half of the tail is bordered by a
skin plica. When looking at the body of a snake, it is possible to determine
clearly, where the body ends and the narrower tail begins. The way this reptile
hunts is similar to the one used by the thresher shark: with a blow of its tail,
this snake stuns or drowns the prey, and then takes it freely with its mouth
and eats it. The food of the oar-tailed python includes waterfowl, small mammals
swimming across reservoirs and medium-sized fish. For hunting, the snake hides
in thickets of water plants, usually under the leaves of water lilies, and one
ring of its body is wrapped around a support – a snag or a bunch of stems of
a plant sticking out of the water. In this position, the snake can stay for
many hours, occasionally carefully exposing its head to breathe. In addition,
the heavy body of the reptile serves as an additional support for the tail at
the moment of striking. Due to the fin plica on the tail, the reptile can swim
well.
The coloring of this python is very simple: brown upper body and head, grayish
stripe along the sides and grayish-white belly. A pattern of a double zigzag
row of ring-shaped spots with a lighter middle is noticeable on the upper part
of the body. The eyes are large, the reptile’s vision is very good – this species
of snake searches for prey mainly with the help of vision.
In the tropical climate, the breeding season is not pronounced. Ready to breed,
males slap loudly on the water with their tails, allowing the female assessing
their strength by the waves formed when the tail hits the water. Males also
feel the force of the opponents’ tails hitting the water, and decide whether
they should enter into a duel for the female, or retire. If there are males
of equal strength nearby, they arrange a duel, flapping their tails on the water
alternately. While courting the female, the male twists himself around her body,
holds her with anal spurs and the tip of his tail “shivers”, sticking out of
the water. Sometimes a female joins him, sticking her tail out of the water.
Pregnancy lasts up to 2 months; there are up to 20 eggs in a clutch. The female
lays eggs in the forest litter, buries them with lateral movements of the body
and leaves the clutch without caring about the offspring. Young snakes about
40 cm long live in shallow backwaters of rivers. Their coloration is grayish-green
with a light belly and a noticeable pattern of black ring-shaped spots. They
hunt by hiding at the surface of the water among floating plants and stun the
chicks of waterfowl and rodents caught in the water with blows of their tails.
As they grow, the background color becomes brown. At the age of 5-6 years, young
reptiles become sexually mature and their growth slows down.
The idea of the existence of this species was proposed by Simon, the forum member.
Atargatis
(Atargatis platicauda)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Aquatic natricids (Aquanatricidae)
Habitat: southern part of Fourseas.

Picture by FanboyPhilosopher
Fourseas, huge brackish lake in South-Eastern Europe, had
appeared in Neocene literally from non-existence. When climate began more humid,
water had filled hollows at the place of Black, Azov, Caspian and Aral seas,
having formed common midland reservoir. Sea animals could not occupy it: Mediterranean
Sea nearest to Fourseas had dried up, and because of increase of amount of rains
in its hollow the set of bitterly-salt swamps had appeared. Certainly, in such
conditions any sea animal could not survive physically. Therefore Fourseas was
occupied with rests of former fauna of Caspian and Black seas – fishes and invertebrates
had survived in salt lakes which existence was supported during ice age by rivers
of Eastern Europe.
The ichthyofauna of Fourseas is made basically by gobies and some other species
of fishes. But except for them in huge reservoir the set of various species
of vertebrates lives. In the sea-like lake there are even water snakes: it is
atargatis named after ancient Phoenician sea goddess.
By appearance atargatis resembles sea snakes dwelt in tropical seas in Holocene.
But those sea snakes lived on Earth in human epoch were poisonous representatives
of order belonging to separate family of sea snakes (Hydrophiidae), and atargatis
is the specialized descendant of colubrid snakes, having been classified, of
course, in independent family because of big differences in anatomy and habit
of life.
Atargatis is rather large snake: the length of its body is up to 3 meters; the
basic swimming organ, tail flatted from sides, accounts about half from them.
Up to tail body is cylindrical, slightly flattened at the stomach. The skin
is covered with homogeneous small scales, adjoining “end-to-end” and forming
smooth surface. Scale has “marble” grey pattern of irregular-shaped spots on
blue background, perfectly masking reptile in thickness of water. One detail
spoils this masking a little: on tail of snake there is oculate red spot with
white border – one from each side. This element of colouring plays double role:
during courtship season male (which is are longer and more harmonious than female
is) shows these spots to the female, and in case of danger spots, simulating
eyes, distract attention of predators from the true head.
Head of atargatis is flattened; at sight from above forward part of head is
rounded. In mouth of snake there is plenty of recurved teeth. This snake eats
fishes, killing them biting through head by teeth (poisonous glands at atargatis
are not present). The basic catch is fishes up to 30 cm long, usually small
and medium-sized gobies with cylindrical body. Also this snake willingly eats
crayfishes and large scuds. Due to rather thin forward part of body atargatis
can catch fishes and crayfishes even in holes. Usually this snake slowly swims
above thickets of water plants, from time to time putting out tongue. Due to
chemoreceptors atargatis catches chemical substances indicating presence of
fishes or crustaceans. Having caught presence of possible catch by smell, snake
starts to try water by tongue, shaking head in sides more often: it like scans
water, catching direction to source of smell. When the snake “detects” catch,
it bends tail S-shapely and makes throw by forward part of body, overtaking
prey. The lateral surface of tail serves to snake as support at throws to catch.
Atargatis kills prey not by poison, but by bite of long teeth. Snake tries to
seize fish by head and at once to bite through its skull, and seizes crayfishes
by armour from above (to avoid their pincers) and some time “chews” by jaws
to shatter its shell. Scuds having weak legs do not cause the big harm to snake:
at atargatis there is armored head. Snake swallows catch entirely: fishes are
swallowing from head (to avoid wounds by spikes), crayfishes and scuds, on the
contrary, from the part of abdomen.
Due to slow metabolism atargatis can hold breath at the whole hour, and even
prefers to sleep under water, having hided in thickets of plants in shallow
water.
This species of snakes had completely severed life on land, having reconstructed
breeding process. Even some species of sea snakes of Holocene epoch were compelled
to creep out on land to lay eggs. Atargatis had escaped of this difficulty:
it is ovoviviparous species.
The female ready to pairing emits in water odorous substance from cloacae glands.
Having felt it, males start to search and chase the female. During courtship
ritual male twists around of female, bites it by edge of mouth to the basis
of tail, pushes by head to surface of water. If near female at once some males
gather, they start to struggle with each other, twisting by bodies and trying
as it is possible to squeeze contender stronger. Only trembling of tips of their
tails indicates the huge effort to which there is a struggle. Usually weaker
contender, surrendering, starts to pull head in sides, trying to free. In this
case hard embraces are immediately unclenched, defeated male swims away, and
the winner starts to care at the female.
After long pregnancy (about half-year) the female gives rise to 2 – 3 rather
large youngs (up to 60 cm long). At the birthing moment each young reptile is
covered with thin membrane, and it immediately breaks it off by several sharp
movements. Having released from egg membrane, young snake emerges to surface
of water and makes the first breath. From the moment of birth it is completely
independent. First weeks youngs eat small fishes and large plankton crustaceans.
Sexual maturity at young snakes comes at the age of three years, and life expectancy
can reach 30 – 40 years and more.
Blind
cavesnake (Cavernerpeton caecus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: caves in mountains of Central Africa.

Picture by Alexander Smyslov
Usually in caves rather small animals live: insects, spiders
and other invertebrates. More progressive animals, as a rule, do not belong
to number of permanent inhabitants of caves. Only in cave reservoirs there are
fishes and amphibians. But in Neocene mountains of Central Africa became the
native place to one of most unusual cave animals.
Food resources in caves of equatorial area differ in abundance in comparison
with those in temperate latitudes. The abundance of sunlight in equatorial latitudes
favors to growth of mosses and algae in caves even at significant distance from
an entrance. And such animals as bats and birds, settling in caves, become a
constant and reliable source of organic substances. Therefore tropical caves
have more various and numerous population.
In caves of mountains of Central Africa there is the original ecosystem including
different insects: beetles, cockroaches and larvae of different butterflies.
Some of them eat dung of bats, others gnaw seeds and sprouts of plants which
have got in cave in stomachs of bats. There are scavengers eating dead small
mammals, or tiny predators.
But all of them do not occupy top of feeding pyramid of caves: other predator
lives here. Between stones the long body of this rather large creature (its
length is about 1 meter) slides. It is covered with smooth shining scale, and
moves practically silently.
When the animal creeps out from stones to open place, it can be seen better.
It is a snake of ivory color with pale pattern like chain of yellowish spots
on back, with white stomach. The snake lifts head and then one feature of its
appearance at once is evident: it is blind. Eyes of this snake are latent under
skin, and scutes, closing eyes, differ from others practically with nothing.
The blind cavesnake – it is the name of this reptile.
The cavesnake never leaves underground world. Here it finds everything necessary
for life. The climate in cave is constant, here dangers of overheat or overcooling
are absent. Due to narrow body this snake easily squeezes even in “bottle-neck”,
therefore it in due course had widely settled in caves of Central Africa.
Sight at snakes has already undergone a partial degeneration when they passed
to partly underground habit of life during evolution. At the blind cavesnake
this process has gone even further, and there are only eye rudiments under skin
at this species. But instead of sight at this snake other feelings, senses of
smell and touch, are developed, permitting to receive the adequate information
about the world around.
Nostrils of blind cavesnake are well advanced and very large.
Because blind cavesnake is a predator, it lives only in caves where there are
colonies of bats. Cavesnake eats basically insects inhabiting such caves, but
also willingly eats fallen down cubs and weakened adult individuals of bats.
As against to many snakes, blind cavesnake eats carrion as willingly, as fresh
catch: food resources of caves are rather poor, and it is more favorable to
use them maximally full. Teeth of this reptile are numerous, but short, thin
and recurved.
Snakes are known for the ability to feel vibrations of ground, determining in
such way approaching of large animals or possible catch. Blind cavesnake has
unusually keenly advanced ability to feel vibration of objects. Due to it this
reptile it can feel the cockroach or cricket running on stone. During hunting
the snake constantly puts bottom jaw to stones or ground, determining presence
of moving catch. Snake makes solving throw completely blindly, having found
catch by speed and direction of movement. After throw the reptile promptly rolls
up to spiral, trying to “overflow” by loops of body environmental space, and
then compresses to dense knot. The sense of this tactics is, that even if the
insect at last moment will change movement direction, one of rings of snake
will necessarily cover and will press it down.
Cavesnake can not digest chitin of insects, therefore it from time to time spits
out the rigid armors of prey pressed to dense lump.
Despite of relative food abundance, each separate cave can support only limited
population of predators which stand at the top of feeding pyramid. But here
there is other problem: the population of snakes containing too little number
of breeding females, easily can degenerate. Therefore blind cavesnake during
process of evolution has got one more property: it became the hermaphrodite
from diecious animal. Sexual glands at this species had changed: one of them
became masculine, and another feminine one. Snake ready to pairing emits small
amount of odorous liquid, leaving appreciably smelling trace. By the same traces
left by other snakes, it finds partners for pairing.
During pairing two snakes mutually fertilize each other. Pregnancy lasts about
4 months. Blind cavesnake concerns to ovoviviparous species: each snake once
a year lays 5 - 7 large eggs covered with thin filmy shell, in which there is
already completely advanced posterity. Young snakes about 20 cm long hatch from
eggs practically at once. At them there is more contrast colouring, and eyes
appear under skin like dark spots. They can distinguish light and darkness,
and vaguely distinguish contours of subjects. After the first shed they start
to eat small insects, obviously preferring larvae of beetles and butterflies
with soft covers.
Young snakes can leave cave, and at night crawl out in searches of new places
of inhabiting. Thus they expand an area of species, and interfere with inbreeding
which leads to degeneration. Traveling snakes spend hot time of day in holes
of rodents or under stones. These snakes are guided, analyzing streams of air,
and finding wind carrying specific smells of caves. They grow slowly: sexual
maturity comes only at 6 - 7-years age at length of body up to 70 cm. But duration
of their life frequently exceeds 30 years.
Chameleon snake (Mutabilophis mutabilis)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: Japan Islands, humid tropical forests of south-east of islands, forest
canopy.
Snakes have very original evolutional history. In the beginning of formation
of this group of reptiles its early representatives passed to partly underground
habit of life; therefore their eyes had started to reduce partly. Further the
direction of evolution had changed: they had become active predators at the
ground surface again, and even had developed the world of forest crones. But
eyes earlier started to reduce had not restored to the full. At snakes cones
in eye retina are absent, and though some snakes have sharp sight, all their
species are not able to distinguish colors. Because of it snakes had lost many
features of behavior connected to the color signal system usual for lizards.
And one more feature of lizards had remained inaccessible to snakes of Holocene
epoch – they had not mastered the skill of color change for masking in environmental
district. At lizards of various species such ability was developed up to refinement
– in human epoch the word “chameleon” in some languages became the nominal
one, emphasizing variability of subject of conversation.
The world varies unpredictably, and the direction of evolution of any group
of live creatures can vary sharply. In this case attributes which gave advantage
earlier, may turn to serious difficulty. But occasionally evolution finds original
decisions for the similar problems formed at ancestors of evolving group in
connection with their further evolutionary history. In Neocene at Earth one
species of snakes able to change colouring partly – the chameleon snake – had
appeared. This reptile lives in Japan Islands, preferring warm rainforests.
Japanese chameleon snake has kept habitual shape of snakes. It is rather massive
reptile about 2 meters long with flattened body. It spends practically all
life in tree crones where it eats birds, wood mammals, amphibians and even
large insects. The chameleon snake prefers to swarm up thick branches and trees
trunks, and is also sluggish reptile.
At this reptile there is large head with expressed cervical interception. Chameleon
snake is not poisonous, but its teeth are numerous, sharp and slightly bent.
Slow moving does not prevent this snake to hunt successfully because it is
compensated by the most remarkable feature of chameleon snake – the ability
of color change. The mechanism of this phenomenon permits to bypass such lack,
as absence of color sight. Abdominal scutes of this snake are connected to
numerous receptors, and allow the reptile to define features of surface on
which it creeps. According to these sensations color of skin of the snake varies.
Sight of all snakes is bad, therefore chameleon snake is not able to estimate
precisely color of objects among which it moves. The mechanism of color change
at chameleon snake is very approximate; it is based at the estimation of contrast
of environmental district and character of tactile sensations. At chameleon
snake there are few variants of colouring, connected with certain tactile sensations,
and this reptile can change color only in these rigidly determined limits.
On rough bark snake gets brownish color with longitudinal wavy strips. Among
smooth leaves the chameleon snake becomes spotty – on green background black
spots appear. At night colouring of the reptile darkens, and at the bright
sun it turns lighter.
Scales on the top side of body of this reptile are small and transparent. On
skin chromatophores are placed, due to which contraction and expansion this
reptile can change color. The stomach of this snake does not change color –
it is white; on throat and neck there are black spots.
The chameleon snake lives in wood crones, only occasionally creeping down to
the ground. Usually snakes living on trees are thin and mobile. But this species
is massive reptile not able to chase catch for a long time. This snake can
make only short throws for catch, therefore it prefers to expect while catch
will approach to it itself closely. The chameleon snake suits ambush on branches,
using its ability to change color. Usually it nestles against the bark or hides
among moss and foliage. The reptile kills catch with the help of teeth, not
strangling it like boas or other snakes.
In courtship season males actively search for females, defining their presence
by smell. The male ready to pairing changes color: it becomes black with white
stomach and separate white spots on body. The male shows to the female the
throat, lifting head upwards at the right angle to body. If two males meet
near the female, they try to push each other away from it, striking the contender
impacts by lateral face of head. Fights of males in courtship season resemble
the competition of heavyweight wrestlers – it is more force struggle, rather
than effective demonstrations. Sometimes one of males may simply “bind” the
contender by rings of body to the branch and show to the female readiness for
pairing, physically keeping the contender. Recognizing defeat in courtship
duel, the contender strongly turns pale, becoming almost completely white.
Then won male lets the contender off.
The female turns pale only a little, admitting the male to pairing. Pairing
occurs in tree-trunk hollow and may last some hours in succession.
The chameleon snake belongs to oviparous reptiles. The female is compelled
to creep down to the ground to lay eggs. It cautiously creeps down the trunk,
clinging by scutes tor roughnesses of bark. On the ground the female finds
a secluded place (frequently right among roots of tree on which it lives),
and lays to simple small hole in the ground some tens of eggs. Having dug eggs,
snake comes back to the tree crone and does not care any more of posterity.
The incubating of eggs lasts about 55 – 60 days. Length of just hatched chameleon
snake is about 20 cm. At young snakes ability to change color is less advanced.
They are green with brown irregular-shaped dabs and live in bush and among
young trees. Later, having had reached the length about half meter, young chameleon
snake gets ability to change color and moves to the tree crone.
Hopping
tree snake (Trachygasterophis saltatus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: Japan, rainforests.
Picture by Fanboyphilosopher
Japan islands are the volcanic ridge in the place where the
bottom of Pacific Ocean “dives” under the continental plate. Pacific Ocean renders
enormous influence to the climate of this region. In Neocene, when the climate
of islands became warmer and more humid, southern islands of archipelago had
become covered by rich rainforest. This forest is rich in various lifeforms,
among which extermely specialized ones frequently live.
The canopy of Japan rainforests is strongly close and bound by lianas: it is
an adaptation for survival in conditions of often hurricanes blowing from Pacific
Ocean. It represents very favorable inhabitancy for species of tree-climbing
animals. Some of them never go down to the ground.
Among inhabitants of forest canopy there is a lot of climbing mammal and reptiles.
And one of the most interesting snakes of the world lives in rainforests of
Japan – it is the hopping tree snake. It is the reptile up to three meters long
with very thin body and long tenacious tail (its length amounts more than half
of general length of the reptile).
The hopping tree snake has cryptic colouring: top of its body is green, bottom
is brown, and lines of short slanting strokes of black color pass on sides.
This reptile eats mainly small birds, showing the big dexterity in hunting.
It seizes them hurriedly, at throw clinging for branch only by tail. In some
cases it rushes on birds, completely coming off from branches and catching prey
in flight. Having caught prey in air, the reptile falls back in branches, and
manages to seize branch by tail in falling. In dexterity of movement in branches
it does not concede to monkeys.
Eyes of this snake are rather large and also shifted to the forward part of
head. They provide practically three-dimensional sight that is very important
at an estimation of distance to catch or next branch. In retina there is lack
of cones (it is usual attribute of all snakes), but by density of rods the retina
of this snake is comparable to retina of nocturnal mammals. Pupil is chinky,
longitudinal, with small expansions on edges. It results to that in eye of hopping
tree snake two areas of the best perception are present: one of them is formed
by first line of pupil and helps to consider catch and to estimate the distance,
and the second one, perceiving images transmitted from back edge of pupil, provides
the snake good lateral sight. Having slightly turned head, snake can look objects
behind its head.
The tail of hopping tree snake is very strong and tenacious, it is easily bent
in any side, and vertebrae can even turn a little about the axis. On the bottom
side of tail corneous scutes have small spike-like outgrowths on edges assisting
to fix in throw. Scutes on the bottom side of tail also can rise with the help
of special muscles, improving durability of branch capture.
To lead such way of life, it is not enough only to have tenacious tail. Wood
mammal and lizards, jumping from branch to branch, amortize inevitable pushes
by paws, and their internals do not feel impacts. At the hopping tree snake
paws are not present, and it must accept impact against the branch by all body.
To not injure internals this reptile had got additional protection. Abdominal
scutes at this species of snakes are firm and elastic; at impact against the
branch they spring, protecting internals of snake from traumas. The snake weighs
a little; therefore corneous scutes protect its interiors well.
Ribs of hopping tree snake also had changed a little: they cover internals from
sides, and their tips are connected by extensible sheaf, also amortizing at
impact. Such additional protection has imposed the certain restrictions to the
diet of hopping tree snake. Usually snakes eat rather large catch which can
be larger than diameter of their body. When the usual snake swallows catch,
its internals and muscles stretch strongly. The hopping tree snake does not
presume to itself such diet because of ligaments between ribs and strong abdominal
scutes. It eats only small birds, and even attacks large insects: it eats butterflies
and big caterpillars. But the thin body and ability to jumps allow this reptile
to eat nestlings of small birds nesting in narrow tree-trunk hollows or attaching
nests to tips of thin twigs. Food specialization of this reptile is rather strict;
therefore hopping tree snakes are territorial very much. Each reptile marks
borders of hunting site by secretions of glands located near cloaca, and does
not tolerate strangers at its territory.
hopping tree snake is very much specialized to tree climbing. It spends all
life in forest canopy, never creeping down to the ground. This reptile prefers
to live in old woods with rich canopy interwined with lianas. This specialization
considerably limits opportunities of its settling; therefore the distribution
of this species is limited only to sites of rainforest at the south-east of
Japan Islands.
Pairing of these reptiles occurs unusually: male and female weave together,
hanging on tails under tree branch. Frequently pairing takes place at height
up to 30 – 40 meters on branch shaken in wind, but it does not frighten reptiles:
in such place they are perfectly protected from the majority of predators. Only
predatory birds can attack them at this moment. They are main enemies of hopping
tree snake.
The female in which ovaries eggs are forming, becomes less mobile and more cautious.
It prefers to hunt on branches and does not make risky jumps for catch. Fertility
of hopping tree snake is small, and does not exceed 5 lengthened eggs in one
clutch, but the egg laying may repeat each month. This reptile lays eggs in
tree-trunk hollows, burying them in dust at the bottom of hollow. Frequently
the female makes some clutches in same tree-trunk hollow, and at the bottom
of such hollow there are simultaneously egg shells, eggs at various stages of
incubating, and young snakes.
Young snakes hatch after 6 – 7 weeks of egg laying. Young snakes are colored
green with white stomach. They become sexual matured at three-year age.
Lancing snake (Hastatoserpenta sylvatica)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: Eastern Madagascar, a rain tropical wood.
Tropical regions of Earth differ in surprising variety of reptiles, among which
there are numerous snakes. Till the ice age at the boundary of Holocene and
Neocene many species of these reptiles had died out, as the area of rainforests
had strongly reduced. But in Neocene changes of climate had resulted in expansion
of the area of tropical forests, and as the consequence to the increasing of
number and variety of snakes. Some of them had got new features of behavior
which had not been present earlier.
In rainforests of Eastern Madagascar there is one interesting species of forest
snakes. It is usual medium-sized reptile – its length is no more one and half
meters, and its body is thin. This snake has brownish-grey color, masking it
on the background of wood bark. Its body is covered with irregular mesh pattern
imitating wood bark, and on head from above there is a yellowish-white spot.
This reptile lives in tropical woods of Madagascar, equally well feeling like
on trees and in wood litter.
This snake is really surprising. The descendant of Madagascar leaf-nosed snake
(Langaha madagascariensis), the lancing snake differs in interesting way of
food getting. The matter is that the nose of this snake, both at males and
at females, terminates by long narrowed edge. This outgrowth is approximately
equal the other part of head of the snake; it is very durable, and also is
sharp: it is formed by lengthened and partly grown together bones of skull
covered with dense ridge scales. Hence this snake has received the name “lancing
snake”. This reptile differs in inactive way of life, and usually expects catch,
having hidden on branch. Thus colouring of its body merges with texture of
bark, and head precisely simulates the broken bough with light wood. As soon
as it notices any potential prey (large wood frog, bird or rodent), it makes
flash-like strike by head, piercing prey by nasal outgrowth, as if by dagger.
Having struck prey, snake at once pulls out the killing instrument from the
wound. “Dagger-like” impact lasts only a split second. If the prey was not
killed at once and had managed to escape, it dies later from loss of blood
and damage of internals. The snake simply finds it by smell and swallows entirely,
as well as it is characteristic for all snakes. It may happen that catch will
remain pined on snake’s nose, especially when it is small creature. In this
case lancing snake removes prey from nose, having griped it in rings of body.
One more interesting feature of the lancing snake is the structure of its tongue:
on its tip there is the fleshy pillow, serving to clear eyes and head of blood.
At the attack of large animal lancing snake can use rostrum as means of protection:
it puts painful wounds to nose and eyes of aggressor. The similar way of protection
is developed at snake of other species living in savannas of North Africa –
egg drillsnake (Ovisugerpeton ovisugus). But it strikes impacts to the enemy
not by rostrum, but by strong teeth sticking forward.
It is the oviparous snake breeding at any time of year. Eggs in amount of 10
– 15 ones are layed in ground, and the female does not care of clutch protection.
Strong rostrum helps to young snakes to punch skinny egg shell, but it is too
short to be used as the hunting weapon. Small lancing snakes first time catch
insects and tiny vertebrates, biting them by teeth. But at them the spike develops
rather quickly, helping to hunt the same way, as it is done by the adult reptiles.
The closely related species of snakes lives in rivers of Eastern Madagascar:
the river
lancing snake, or the harpoon snake (Potamohasta ichthyophaga).
It is similar to the wood relative, but differs a little in colouring and the
body shape. Top of body at this snake is silver-grey and stomach is milky-white.
Body of this reptile in back part is rather wide and massive, and head and
forward third of body are thin. It eats fish (preferring small fishes with
long and not deep body) and water frogs, killing catch the same way as its
forest “cousine”. Thus the wide body serves as a support for snake at the rush.
Harpoon snake often hides among driftwood and water plants, and even is
dug by tail in river bottom. Being externally similar to sea snakes of Holocene,
this reptile is not as specialized, as they were: it can creep on land, easily
crosses marshy sites of wood, and besides lays eggs in ground.
These species of reptiles are discovered by Simon, the forum member.
Dwarf vinegar snake (Acetophis foetidissimus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: Japan Islands, humid forests at the southeast of archipelago.
Snakes are youngest groups of reptiles existing in a Neocene. Having evolved
in cretaceous period, they had kept rather monotonous and recognizeable shape,
and differ only by some features of anatomy. In human epoch basically large
or local species of snakes had died out – mainly because of destruction of
habitats, and partly because of hunting. But smaller snakes frequently adapted
to life in the neighbourhood with people and found rich prey as rodents and
small birds, and in tropics more lizards and large insects besides of them.
One small insectivorous snake of Neocene lives in tropical forests at the southeast
of Japan Islands. It is dwarf vinegar snake, the only representative of genus
endemic for Japan Islands, harmless reptile which spends the most part of
time, creeping in forest litter and searching for prey. It feeds exclusively
on insects
(preferring soft-bodied cockroaches and grubs) and slugs. Body length of dwarf
vinegar snake is about 40 cm. It has rounded head, cylindrical body and very
short tail.
Appearance of this snake is very bright and well-remembered. It imitates very
precisely tiger millipede – a kind of poisonous millipeds living in the same
place. Colouring of body of this snake is orange with black cross strips. The
tip of muzzle of snake is black (it is an imitation of head of millipede),
and eye is masked by black vertical strip. Edges of belly plates are white
with black border – they imitate legs of millipede. Usually dwarf vinegar snake
tries to hide in depth of leaf litter in order to avoid meeting with possible
predators. The disturbed snake imitates a way of protection of milliped – it
rolls in spiral and hides head among coils of body. Also it imitates smell
of protecting milliped, emitting from cloaca an odorous liquid having vinegar
smell and unpleasant bitter taste. But, as against to tiger millipede, this
species is absolutely poisonless. The only active protection of which this
snake is capable is a bite of pointed teeth. It may be enough to support warning
colouring which it has.
Due to warm climate these snakes are active the year round. Dwarf vinegar snake
searches for food from midday till evening, and in the rest of the time it
prefers to stay in shelter, having turned to spiral like a protective pose
of milliped.
Seasonal prevalence in breeding at this species is not expressed. Male differs
from female in a little bit longer tail and thinner body (it weighs less, rather
than female at equal body length). Male searches for the female ready to pairing
with the help of smell sence. Pairing takes place at night in shelter, where
the female is usually hidden.
Dwarf vinegar snake is ovoviviparous and differs in very low fertility – in
ovoduct of the female no more than two eggs in thin leathery pellicle are incubating
simultaneously. Their development lasts till about 4 months. The female bears
posterity two times within one year. Young snakes leave egg covers right at
birth. The length of young snake makes almost half of length of the parent.
Young snakes of this species frequently search for carrion or dung of large
mammals, and eat larvae of flies developing in such substratum. They reach
length of adult individuals in three years, and one more year later become
able to breeding.
Skunk
snake (Foetinatrix jaculator)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrid snakes (Colubridae)
Habitat: Europe, subtropical areas at the coast of Fourseas – the Balkan, Caucasus,
Crimea.
Grass snake (Natrix natrix) in human epoch was one of the most widespread species
of snakes of Eurasia. This reptile successfully lived in human-changed landscapes
and by the end of human era had the great number and wide area – a pledge to
successful survival and evolution. Descendants ужа mastered new habitats and
developed various tactics of a survival. One of grass-snake descendants, living
in subtropics of Europe, has the original adaptation, permitting it to protect
itself against the enemies. This reptile is named skunk snake, and its name
indicates the protective tactics of this species.
Skunk snake is ordinary enough species of reptiles – it is a snake up to 150
cm long, conducting ground way of life. It lives in forests and bushes and eats
small vertebrate animals – frogs and rodents. But, being disturbed, it uses
spectacularly looking demonstration before to use the protective weapon. The
bottom part of body of skunk snakes from cloacal area up to tip of tail has
very appreciable colouring – it is cross-striped black-and-white one. Protecting
itself, snake rears back part of the body and directs cloaca to the enemy, simultaneously
turning tail the bottom part aside the enemy. This is a kind of warning: tail,
black-and-white from lower side, is clearly visible even to a predator lack
of color sight. Cloaca of skunk snake forms a large sac in which unpleasantly
smelling oily liquid is gathered. This sac has ring muscles, and at their contraction
it splashes out a jet of liquid to the enemy. Liquid has very proof smell which
is felt within several days.
The top side of body of skunk snake is colored ochre-red with dark chain-like
pattern along the back; right behind eyes there is citreous cross band.
In areas of seasonal climate skunk snake runs into the hibernation lasting up
to about two months. In southern part of area this species keeps activity the
year round.
Pairing begins in spring, right after the ending of hibernation. In southern
areas pairing is dated for the beginning of spring rains. In courtship season
males behave aggressively relatively to each other; they pursue and bite competitors.
Pursuing the female, male creeps side by side to her, cautiously bites her body
and forces to stop, twisting around of her body. Female ready to pairing lets
a small amount of odorous secret, and males find herby smell.
After pairing female lays up to 20 lengthened eggs in abandoned hole of any
rodent, fills up an entrance with ground and does not care of posterity any
more. The incubation lasts till about 3 months. Young snakes hatch in the middle
of summer. They eat frogs and keep near water, finding there food and protection.
In case of danger young skunk snakes can dive and hide at the bottom among silt
and driftwood. They produce very small amount of odorous secret and are not
able to splash it to the enemy. In colouring of young snakes yellow cross stripe
behind eyes is absent – it appears after the second year of life. Young skunk
snake grows intensively during first four years of life, and for the third year
can take part in breeding. Life expectancy of this reptile reaches 50 years.
Reed
snake (Natrix scirpophilus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: Europe, old overgrown reservoirs, wetlands – northeast to Wenedian
Lake, south to the Pyrenees, west to the Atlantic coast.
Snakes adapt well to the aquatic environment. One of the most successful species
of the human epoch was the dice snake (Natrix tesselata), which left many descendants
in the Neocene. One of them is the reed snake, an aquatic reptile from southern
and western Europe.
This species is a small snake; its body length is about 40 cm. The body is long
and thin, like at other colubrid snakes. There is a kind of “fin” on the back,
which is a skin plica. The tail has a different kind of specialization: it is
flexible and very tenacious, equipped with scales protruding from the sides.
Despite the predominantly aquatic lifestyle, this snake can crawl out onto land.
The body color is green, the back is of different shades in different individuals;
there are gray or black spots arranged in chains on the back and sides; the
belly is white. The eyes are orange, with a round pupil.
Reed snakes inhabit large bodies of water with a large amount of aquatic vegetation,
preferring oxbows and lakes. The reptile usually hides among coastal and aquatic
vegetation – reeds, cattails, etc. Also, sometimes reed snake crawls on stones,
leaves of water lilies, trees floating in the pond. For hunting, it swims into
the thickets of plants and attaches by its tail to the stem, like a seahorse.
When a prey of a suitable size swims nearby, the snake makes a rush in its direction
and catches it with its jaws, and then swallows it. The food specialization
of this species is wide: crustaceans, insects and their larvae, fish, frogs,
newts, juvenile turtles fall prey to reptiles. For the reed snake itself, birds
(for example, gulls and herons), large predatory fish and turtles, and less
often terrestrial predatory mammals are dangerous. To protect against them,
the reed snake either leaves the water, climbing on the stems of plants, or
dives and hides in the mud, holding its breath for half an hour or more. In
autumn, snakes from northern populations crawl ashore, where they dig holes
in the ground or occupy ready-made shelters, and hibernate there. Reptiles from
the southern part of the range overwinter in the water.
In the spring, after mating, the female crawls away from the water, to a guaranteed
dry place, and arranges a nest hole there with a wide nest chamber at the end,
where she lays about a dozen eggs, surrounds them with body rings, like a female
python or cobra, and protects from enemies. After 8 weeks of incubation, the
juveniles hatch from the eggs. They immediately crawl away to the nearest pond,
and after them the adult one leaves the nest. For the first time of their life,
young individuals lead a secretive lifestyle, hiding in a layer of plant debris
at the bottom and feeding on crustaceans, insects and tadpoles. With age, they
change their diet and the way they hunt.
Sexual maturity comes at the age of 3 years; life expectancy is up to 30 years.
This reptile species was discovered by Mamont, the forum member.
Asian
egg snake (Niduophis oviphagus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: East Asia, Japan Archipelago; forests and shrubs.
Snakes form a group of exclusively zoophagous reptiles: there are no omnivorous
or herbivorous species among them. The food of snakes is made mainly of terrestrial
vertebrates ranging in size from small to medium and large. Less often snakes
eat fish and invertebrates. But some snakes demonstrate a kind of specialization
to feeding on eggs. It is a seasonal food, available only part of the year,
but snakes use it successfully due to their reptilian metabolism and the ability
to starve for a long time. In Africa of the human epoch, there was a genus of
egg-eating snakes Dasypeltis, and in South Asia – the Indian egg-eating snake
Elachistodon. In the Neocene on the territory of Africa, a genus of drill
snakes (Ovisugerpeton) appeared, forming a small separate family and
specialized in eating eggs of large bird species. In Asia, another type of snake,
the Asian egg snake, has become a specialized consumer of bird eggs.
This reptile species is a descendant of the Schrenck’s rat snake (Elaphe schrenki),
which showed a tendency to feed on bird eggs even in the human era. Specialization
in feeding on eggs has caused a bunch of anatomical changes in this species.
It also has a “can opener” for cracking eggshells, which is formed of elongated
processes of vertebrae facing the pharynx. This adaptation developed in this
one independently of the “true” egg-eating snakes, thus presenting an example
of parallelism in evolution. When swallowing an egg, the reptile strongly squeezes
it with the circular muscles of the pharynx and the processes of the vertebrae
punch a hole in the shell. By contracting the muscles, the reptile squeezes
the contents of the egg into the esophagus and spits out the crumpled shell.
This reptile species climbs trees very well when its physique allows it. The
Asian egg runner reaches a meter and a half in length; the body is colored brown
with a pattern of several parallel intermittent stripes stretching along the
back and sides. The background color of the sides has a yellowish hue, and the
belly is creamy-white. The head of this snake is narrow and pointed.
The Asian egg snake lives in the tree crowns and feeds exclusively on the eggs
of small birds. This reptile lives in conditions where the seasonal climate
makes the usual food available for only a few months of the year. This circumstance
imposes certain restrictions on the life cycle of the reptile. The Asian egg
snake is able to go without food for a long time and to fatten very quickly
in the presence of available food. The snake feeds especially intensively in
spring and early summer, accumulating a significant amount of fat under the
skin and in the liver. The back of the body becomes thicker and heavier than
the front one, and the snake has difficulty climbing trees. The second peak
of feeding activity takes place in the middle of summer, when small birds make
a second clutch. At this time, mainly small individuals fatten; large reptiles
are limited to the spring “feeding fenzy”. At this time, these snakes treat
each other very aggressively, protecting their feeding territories and driving
out competitors from their possessions. Fights, accompanied by bites and a forceful
“struggle” of twisted bodies, often take place between adults
Sexual dimorphism in the Asian egg snake is poorly expressed; the male has a
longer tail and a lighter build than the female. The breeding season of these
snakes takes place in the middle of summer, when they are well-fed and intraspecific
aggressiveness is reduced. This snake lays eggs in the hollows of trees. Eggs
are not numerous (2-4 eggs are laid at the same time) and have a characteristic
elongated shape. Laying is repeated up to three times per season. Young snakes
develop during the summer and the first half of autumn; in the northern areas
of the range, where the seasonality of the climate is pronounced, the juveniles
overwinter in eggs and hatch in the spring. After the first molt, young snakes
begin to feed on the eggs of small birds. Due to their light build, they are
able to climb even reed stalks and often feed in wetlands, where adult snakes
of this species are not found. In the southern parts of the range, where the
seasonality of bird nesting is not pronounced, breeding and hatching of young
animals occur throughout the year.
Sexual maturity comes in the third year of life. Life expectancy is more than
20 years.
Bird-loving
Tree Snake (Macroboiga tigrina)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrid snakes (Colubridae)
Habitat: Rainforests of northern Meganesia, swamp forests and mangrove forests
bordering Carpentary and Arafura sea lakes.
Australian rainforests are known for various snakes, among them the Brown Tree
Snake or Cat Eyed Snake (Boiga). This species was little threat to humans, and
has left a more fearsome descendant in the Neocene, Bird-loving Tree Snake.
Up to 3.1 meters in length this snake looks a good deal like its ancestor, but
is somewhat bigger and thicker, however body is thin, long, sinuous and prehensile,
snake slithers through the treetops quite fast, can also climb rocky outcrops.
Coloration varies from light reddish brown to an olive green with a pale yellow
belly, it invariably has dark brown banding. Eyes are large and protuberant,
with a pupil resembling a cat. Head is broader and more triangular than its
ancestor, and this is connected to its diet, this snake feeds on vertebrate
prey including frogs, lizards, rodents, bats and birds up to the size of a thrush.
Snake is rear fanged, and must bite hard and firmly onto prey to administer
venom. The head and throat are also especially adapted to steal eggs from the
nests of birds, and this snake will engulf a single egg by expanding its jaws,
muscular pressure exerted on keratinous spikes at the back of the throat pierce
the egg and drains its contents, the shell is spat out.
This snake is strictly nocturnal and will hunt birds as they sleep or frighten
brooding birds away to feed on the eggs. When alarmed by a predator, it can
jump a fair distance in order to catch itself on branches lower down. Reproductive
age is reached at about 3 years old, male finds the female by smell, and breeding
takes place in summer. A clutch of up to 12 eggs is laid in a tree hollow, dead
log or a cave, and females may lay 2 clutches in a year. The eggs are not cared
for, and hatch 90 days later. Lifespan is 10 to 15 years; predators include
other snakes, large lizards, raptorial birds, and predatory marsupials.
This species of reptiles was discovered by Timothy Donald Morris, Adelaida, Australia.
Giant
regina, goliath regina (Regina goliath)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: rivers of southern North America.
The Neocene was a period of significant changes in the fauna and flora of the
Earth. During the human epoch and the ice age, many large groups of animals
(up to the order level) died out, many ecological niches became empty, and new
species occupied them. The giant regina, or goliath regina, the aquatic snake
of the rivers of North America, is one of the new species of the Neocene epoch.
It is a descendant of the queen snake (Regina septemvittata), a representative
of a fairly common and numerous group of American snakes, but it differs markedly
from its ancestors. The maximum length of some individuals can reach 150 cm,
which is more than twice the length of the ancestor of this species. Goliath
regina is covered with unicoloured brownish-gray scales, since this reptile
spends most of the time at the bottom of the river, waiting for its main prey
– river fish, less often amphibians and other reptiles. Its nostrils are located
almost at the tip of the muzzle, very much shifted upwards and equipped with
skin valves; the eyes are also located in the upper part of the head, but they
are relatively small. Vision does not play a decisive role in the life of these
snakes; the main sense organ of this reptile is the tongue with which it catches
smells and tastes in its environment.
Although goliath regina is an aquatic reptile, it is a relatively poor swimmer:
it prefers attacks from an ambush. Usually it hides in underwater vegetation
or buries itself in bottom silt, holding its breath for almost an hour. It is
not poisonous, but it has powerful jaws and sharp teeth bent back, so the prey
has little chance to escape: after the reptile’s teeth have sunk into its body,
it wraps its prey with body rings and immobilizes it, gradually swallowing it
underwater.
Goliath regina is viviparous, and in the warm season, females of this species
give birth to an average of 4-5 juveniles, which are already ready to live on
their own. At the northern part of the range, courtship games begin at the end
of summer, while the water is still warm, and the females winter already pregnant,
giving birth to offspring in the spring, after sufficient warming of the water.
In the southern part of the range, breeding takes place all year round.
At first, young snakes stay in shallow water overgrown with plants, away from
their adult relatives and predatory fish, their main enemies, and eat water
insects and their larvae, as well as small fish. Over time, they migrate to
deeper waters. Sexual maturity comes at the age of 3 years. The average life
expectancy of goliath regina is about 18 years.
This reptile species was discovered by Bhut, the forum member.
Bøygen
(Regina boygen)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: rivers of North America.
The Neocene was marked by large-scale biosphere rearrangements – nature was
recovering after a period of anthropogenic pressure that reduced biological
diversity, and the ice age that changed the boundaries of climatic zones and
natural communities. During the restoration of biological diversity, new ecological
niches emerged, and they were occupied by the descendants of the species that
survived in the human era. The rivers of the southern part of North America
are inhabited by descendants of the queen snake (Regina septemvittata), a reptile
quite common in the Holocene. One of them is goliath
regina, but a related species lives side by side with it – bøygen
(named after the monster from the “Peer Gynt” play).
The bøygen grows up to 120 cm in length, has a monotonous gray color
without a pattern, and is able not only to ambush at the bottom in search of
prey, but also to swim in the water column, chasing prey. Like all reptiles,
it breathes atmospheric air; its nostrils are shifted from the tip of the muzzle
higher and closer to the eyes, as was the case with the phytosaurs of the Triassic,
so the reptile can both see above the water and breathe while slightly sticking
the top of its head out of the water. In addition, the oral mucosa forms numerous
papillae and this species can absorb oxygen through the oral mucosa directly
from water, which its ancestor was unable to do. Due to this feature, boygen
prefers oxygen-rich water and does not live in well-warmed and swampy reservoirs
with muddy water.
This snake is able to wrap rings around and drown its prey, but its body is
more compressed from the sides, especially in the rear half of the body and
on the tail. This snake comes to land even less often than its relatives, and
hunts mainly underwater animals – fish, crustaceans and amphibians. Sometimes
the bøygen attacks the waterfowl chicks, but it happens quite rarely;
despite its size, it is a cautious reptile. Its graceful and fragile physique
does not allow it hunting large prey.
Bøygen is an ovoviviparous snake, and in the warm season, females of
this species give birth to an average of 6-8 juveniles about a quarter of the
length of the mother. Young snakes are able to swim right after birth; they
immediately leave their mother and hide in the thickets in shallow water. They
have a slightly different color than adult snakes – longitudinally striped pattern
with a greenish tinge, which gives them additional camouflage among underwater
plants. As they grow older, these stripes fade, young snakes get adult coloring
and leave the underwater thickets to lead an active lifestyle. Young snakes
feed on tadpoles and aquatic insects, and hunt fish fry. Their survival rate
is relatively high: thanks to their camouflage abilities, they fall only accidental
prey for nearwater birds, predatory fish and turtles.
The average life expectancy of boygen is about 14-16 years.
This reptile species was discovered by Bhut, the forum member.
Tlamatini,
or anteater-snake (Thamnophis tlamatini)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Colubrids (Colubridae)
Habitat: seasonally humid forests and tropical rainforests of Central America.
Among the numerous group of garter snakes of North America, a number of species
and subspecies became extinct during the human epoch and during the climatic
changes at the border of the Holocene and Neocene. This snake is a descendant
of the Western ribbon snake (Thamnophis proximus), a snake species widespread
in the human era from the southern United States to Costa Rica. For many peoples,
the snake is a symbol of wisdom, so this snake was named tlamatini, which means
“thinker, sage, philosopher, scientist” in Nahuatl.
Tlamatini is a miniature representative of its group: the total length of the
snake ranges from 40 to 50 cm. The body is slender, with keeled scales; the
head is narrow and has small eyes shifted forward. The reptile’s vision is partially
binocular. The main background color of the body is olive, yellowish on the
belly. Compared to the top of the head, the lip shields are of much lighter
coloration. There are an orange stripe stretching along the middle of the back,
and a cream stripe on each side.
This species loves wooded wetland terrain, inhabiting lake shores and rainforests.
Activity is mainly diurnal. This species feeds on frogs, tadpoles, fish, small
arthropods, worms, snails and slugs. The main criterion for the choice of food
objects is their small size.
These snakes can often be found in considerable numbers (up to 100 snakes at
a time) in the vicinity of megacolonies of local pochteca
ants, which this snake likes to feed on. In the strategy of ant hunting,
it is similar to the Australian moloch lizard. While getting its food, tlamatini
arranges an ambush near the ant path leading from the bushes with coccids
to the colony. With a movement of its head, it grabs ergate ants with abdomens
filled with honey-fall of coccids, as well as forager ants carrying their prey.
The reptile ignores ants carrying droppings for Calathea
fertilizing, distinguishing them by an unpleasant smell. After catching its
prey, the snake turns its head to the side in order to prevent the alarm in
other ants due to the smell of formic acid from the crushed insect. In one day,
tlamatini can eat up to several thousand ants.
Breeding season takes place in spring. Males compete for a female, pushing each
other away from a ready-to-mate female. Occasionally there are skirmishes between
them, accompanied by a power struggle. Mating lasts for several hours. It is
a viviparous reptile species; the female gives birth to up to 27 juveniles.
Life expectancy is up to 20 years.
The idea of the evolution of this species was expressed
by Mamont, the forum member.
The description was composed by Wovoka, the forum member.
Green
cobra (Naja crudus)
Order: Squamates (Squamata), Suborder Snakes (Serpentes)
Family: Elapids (Elapidae)
Habitat: underbrush of jungles of Southeastern Asia and Hindustan.

Picture by Alexey Tatarinov
Unlike some other snakes, cobras and other elaphid snakes kept
a significant variety of species after the epoch of anthropogenic pressure of
Holocene and the climatic cataclysms of the beginning of Neocene. The restoration
of forest localities in Neocene promoted the evolution of forest-dwelling forms
of this group. Green cobra is a descendant of one of Asian cobras of Holocene,
one of new species of venomous snakes evolved only in Neocene.
This is relatively small reptile that reaches 1.5 m length on the average. Its
body is thin and flattened, but since this snake does not climb on trees, hunts
medium-sized prey, kills it by venom and leads solitary and hidden way of life,
strongly developed musculature is not required for these snakes.
Green cobra is covered in sclaes of dark green color with brighter colored belly;
sometimes individuals with brighter pigmentation appear. This coloration contrasts
wildly with the head of ochre-red and the figure on hood of the black and white
“glasses”, which makes this snake noticeable during the daytime.
Like other elapid snakes, green cobra hunts at night, and during daytime it
sleeps in fallen foliage, in burrow of other animals or in hollows of fallen
trees. If other animals disturb it or attempt to attack, this snake hisses first
– after raising its head and showing-off its hood – and then bites. Venom of
green cobra is neurotoxic, but also has cytotoxic side-effects. Small rodents
and birds die from its bite after few minutes, but larger animals can survive.
The prey of this species includes small mammals and birds, more rarely reptiles
and frogs, which have a considerably greater resistance to green cobra’s venom.
Sometimes green cobra eats larvae of large beetles and other soft-bodied insects.
Green cobra does not have clearly determined mating season, and at any time
of the year (but usually during the rain season) it is possible to meet females
of this species that guard their clutches buried in fallen foliage. The clutch
of green cobra usually numbers 10 to 15 eggs; the hatchlings’ appearance resembles
that of the adult snakes in the miniature: the length of newly-hatched snakes
is approximately 20 cm.
The greater number of young cobras perish before they will reach at least half-meter
in length. The first food of young snakes is insects and small frogs.
The average life expectancy of green cobra is about 40 years.
This species of reptiles is discovered by Bhut, the forum
member.
Translated by Bhut.
Markupo
(Naja markupo)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Elapids (Elapidae)
Habitat: Southeast Asia, Jakarta Coast, islands of Indonesia; tropical forests.
Unlike some other snakes, cobras and related species in the Neocene are represented
in fairly large species diversity, although they still remain limited in their
range by Old World (Africa and Asia). Their general appearance is also easily
recognizable, with rare exceptions.
The green cobra of Neocene Asia and Hindustan
is one of the most common species of Asian cobras. Its close relative, markupo,
lives next to it.
Markupo reaches a length of 2-2.5 m. Its appearance is typical for elapid snakes:
slender body, small head without a cervical waist, wide-opening mouth and fixed
poisonous teeth. In elapids, unlike crotalids and vipers, venomous teeth are
not very large. Their venom is also different – in elapids, it is neurotoxic
and causes paralysis of the nervous system. The venom of markupo is strong enough
to kill not only small animals (especially rodents) and birds, which this snake
species feeds on. The poison is also dangerous for larger jungle inhabitants
if they try to eat markupo or attack this reptile (hence the name: in the Philippines,
markupo is a very poisonous snake from the folk tales). Despite the venom of
this snake, some jungle inhabitants – for example, the predatory pig varaha
and some other species attack it and eat it without much fear.
Markupo has a green coloration, but this snake has a number of black and white
rings on its tail. In a hidden snake, the body is poorly visible against the
background of vegetation, so predators usually attack a more noticeable tail,
which allows markupo turning to the enemy for the bite. Like all cobras, it
has a hood, which is monotonously green from the back, without “spectacles”
or other markings. However, on the lower side it has contrasting coloration:
dark spots on a lighter background. It is an aposematic coloration that markupo
displays to scare off the enemy if it has not yet managed to attack the snake.
This snake species lives in coastal areas of tropical forests near rivers and
lakes, where dense undergrowth develops and thickets of large herbaceous plants
grow. The snake swims willingly and searches for prey in the water, often attacks
broods of waterfowl.
Like many tropical species, markupo does not have an expressed courtship season,
but usually clutches are done during the rain season. They are buried in fallen
leaves, less often in loose ground. In the clutch, there are usually 7-10 eggs
covered with light shells. From them juveniles hatch, which are outwardly similar
to their parents, but are no longer than 20-25 cm. They are poisonous already
when they hatch, but they have many enemies, and many of them die in the first
weeks of their life. Adult markupos do not care about offspring. At the age
of 5, young snakes take part in reproduction.
The average life expectancy of markupo is 50-60 years.
This reptile species was discovered by Bhut, the forum member.
King
Tiger Snake/ False King Cobra (Neoechis gigantea)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Elapids (Elapidae)
Habitat: Found throughout much of tropical and subtropical Meganesia, prefers
well vegetated areas, absent from deserts.
Australia was characterized in the Holocene as the home of various exceedingly
venomous Elapid snakes, Tiger Snakes, Brown Snakes, Taipans etc. These forms
were devastating predators of warm-blooded animals such as birds and mammals,
though their fangs were quite small compared to those of a cobra or viper.
This trend has continued in the Neocene, even in spite of human persecution.
Various kinds of Elapid predominate throughout the continent, with the largest
and most fierce being the King Tiger Snake, a descendant of Notechis, the tiger
snake. This snake is both long and massive, reaching a length of 4.5 meters,
its head is very broad and bears larger fangs proportionally than its ancestor.
Ecologically it is more general than its ancestors, feeding partly on small
and medium sized warm blooded animals but also a great deal on other snakes,
and lizards such as monitors. In this way it is an analogue of the Holocene
King Cobra. The animal is banded with chocolate brown and olive green, with
a creamy yellow underbelly.
Temperament of this animal is generally aggressive, when threatened it will
rear up and show a flattened neck “hood” much like a true cobra, and even as
its ancestor did. An opponent may be enough of a threat to pursue and bite in
defensive action. Vocalisations range from a soft hiss to a loud churring hiss
or a high pitched whistle. Predators are relatively few, but may include large
land birds or raptorial flying birds. This snake prefers vegetated areas and
will stay relatively close to water sources, it is absent from deserts, it is
also a good swimmer. It is active mostly in the mid morning and afternoon, after
being warmed by the sun.
Mating occurs opportunistically in summer, and females give birth to up to 50
live young which have potent venom from birth. Young have a distinctive black
chevron on the head that serves as a warning to potential predators.
This species of reptiles was discovered by Timothy Donald Morris, Adelaida, Australia.
Illuyiankas
(Illuiyankas phoeniceana)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: Near East, from eastern edge of Mediterranean hollow up to Persian
Ridge.
After closing of strait of Gibraltar and drying of Mediterranean Sea the climate
of Near East began considerably more rigorous, rather than in human epoch. Mountains
do not pass clouds from Fourseas to these places, and the extensive territory
to the east from the Mediterranean hollow represents dry desert. It is the world
where mammals are not too diverse, but birds and reptiles prosper. Among the
last ones there are huge forms.
One of the largest species of poisonous snakes of the world lives at Near East.
It is illuyiankas – huge snake, the descendant of Field’s horned viper (Pseudocerastes
fieldi) lived in these places in Holocene. The shape of illuyiankas causes memoirs
on ancient myths told in early historical epoch and subsequently forgotten and
missed for ever in common with mankind. By its size illuyiankas resembles boa
most of all: thick body of reptile reaches the length of 8 meters. Colouring
of top part of body is light grey with indistinct marble pattern on sides; stomach
is white. Along the backbone of reptile white or light yellow ledges are located,
forming something like dragon’s serrated crest on back. Flattened head is divided
from body by obviously expressed neck narrowing. At this reptile species sexual
dimorphism is expressed: on back part of male’s head a line of lengthened scales
resembling short mane is developed. Besides male is longer and more aggressive,
rather than female, and is colored a little bit brighter. Female is approximately
one meter shorter, rather than male, but is heavier and stronger.
Illuyiankas is the top predator in desert ecosystems. Diet of this reptile includes
harelopes, rodents and small carnivores – any animals weighting up to 20 kg.
Thus illuyiankas can steal up to prey absolutely imperceptibly, despite of the
size. In spring this reptile is active in day time, and in summer leaves to
hunt at night. In hot weather this reptile prefers to hide in shadow of stones
and crevices in rocks. At this time it is slow active.
Attacking prey, illuyiankas seizes it by teeth and simultaneously tries to turn
the caught animal in rings of body or to press it against the ground by its
own weight. During the bite illuyiankas “chews” prey, making short pulsing movements
by jaws. Thus poison from glands is squeezed out into the body of prey. Poison
of illuyiankas influences blood of prey, starting the mechanism of blood coagulation.
Within several minutes in blood system of animal bitten by illuyiankas blood
clots, closing the vital blood vessels are formed, and prey perishes.
Short winter serves as stimulus for the subsequent courtship behaviour. In winter
illuyiankas is hidden in natural caves and wide holes of mammals. Young individuals
dig their own holes. The courtship season begins at the end of winter when the
temperature of air becomes high enough to keep the activity of reptile. Males
gather on well warmed up stones and arrange courtship displays - having risen
forward part of body, they shake heads. That one of them which lifts head higher
than others and keeps in such position for longer time is winner. Force struggle
in such tournaments is reduced to pushing the contender by head and forward
part of body forcing it to fall on the ground. The winner receives an opportunity
to pair with the female. Pairing lasts till some hours and at this time male
twists around the body of the female.
This species of reptiles is ovoviviparous. After pairing within two months eggs
develop in the organism of female which lays in spring from 8 up to 40 large
eggs in leathery shell depending on her size and general condition. The laying
takes place in shelter – more often in cave near to the entrance where stones
well get warm in the sun. In four weeks young snakes 50 – 60 cm long leave eggs.
They grow rather quickly, reaching the length of 5 m at three-year age. From
that moment they reach sexual maturity.
Illuyiankas is remarkable in its considerable longevity: the maximal life expectancy
is about 100 years.
The idea about existence of this species of reptiles was proposed by Nick, the forum member.
Tauridian
adder (Vipera taurica)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: Fourseas, Crimea island.
Crimea turned into an isolated island at the beginning of the Neocene, when
the basin of the Fourseas was filled with water. Up to this point, during the
ice age at the boundary of the Holocene and Neocene, the Crimea was only a hill
among a dry plain with separate patches of salt marshes. Representatives of
the terrestrial fauna easily settled the Crimea, and after the formation of
the Fourseas began to evolve independently of the mainland populations.
One of the reptiles of the Crimea island is the Tauridian viper, a descendant
of the meadow viper (Vipera ursinii) and endemic to the island. During the ice
age, the meadow viper appeared to be one of the few species capable of migrating
from the Crimea to the mainland not only along the Perekop Isthmus, but also
through the salty bottom of the dried-up Black Sea. On the peninsula, only small
and graceful individuals remained, being less dependent on food. In the conditions
of island isolation, the remaining snakes increased in size, but remained relatively
slender. Over time, a separate species, clearly different from relatives from
the mainland, has formed from this population.
The length of this snake is 2.3-2.5 meters; females are more robust than males,
but inferior to them in length. The head of this species retains an appearance
typical for vipers: it is wide and has a pronounced cervical waist. However,
in its physique, Tauridian viper resembles colubrids – the reptile is quite
slender, with smooth scales and long tail. The background color is dark brown
to black. Against this background, there are not numerous white or yellow narrow
transverse stripes and half-rings covering part of the back and side. There
are no differences in coloration between males and females. Melanistic individuals
often appear.
This reptile feeds on rodents, small birds and eggs. Occasionally this snake
can swallow a cub of great Crimean hare, but it
is a rare piece of luck. They usually hunt only newborn cubs of this species,
which they can swallow, so such prey is available to them for only some weeks
a year. Tauridian viper kills its prey with poison, and additionally fixes large
rodents with body rings.
Like other vipers, this reptile is an ovoviviparous species. Mating takes place
in the second half of April-May. Males actively search for females by scent
and chase them, pressing them to the ground with body rings. If several males
meet near one female, they arrange ritualized duels. During the duel, the opponents
make a characteristic “stand” with the front part of the body, trying to rise
above the opponent. When an opponent falls, the winning male tries to pin his
opponent to the ground with his body and hold him for several minutes. Three
months after mating, the female gives birth to 5-7 juveniles up to 20-25 cm
long. Sexual maturity in the Tauridian viper comes quite late: at 4-5 years
old with a length of 175-180 cm. Life expectancy is 20-25 years.
This reptile species was discovered by Nick, the forum member.
Siberian
viper (Vipera boreasiatica)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: taiga zone of Eurasia.
Siberian taiga zone was always an inconvenient place for life for cold-blooded
animals, amphibians and reptiles, but, nevertheless, some of their representatives
gradually settled farther to the north. Among the lizards the viviparous lizard
(Zootoca vivipara) had been one of such species, and also common European adder
(Vipera berus) among the snakes.
Millions of years passed since the time of Holocene. In general, the world climate
had time to turn somewhat warm, which allowed the taiga zone to shift to higher
latitudes. Forest-dwelling animals settled following forests, and lizards and
snakes among them.
In appearance Siberian viper does not seem any special species: this is rather
large snake, about one meter long, of massive constitution, covered with thick,
one-coloured dark grey or even black scales. In the autumn, having accumulated
the stock of hypodermic fat, this snake also grows fat and wide, and its body
becomes flatter a little bit.
Head of Siberian viper is both wide and flat, and it is clearly separated from
the body with the neck narrowing. Its eyes are also large, sight is sharp by
standards of the snakes; therefore it is hard for a predator to sneak up upon
this viper. Nevertheless, a main sense in this snake is the sense of smell;
furthermore, it can feel ground tremors in case of any large animal approaching.
The food of Siberian viper includes various small mammals and birds, terrestrial
or arboreal, and it attacks them from ambush (heaps of fallen conifer needles
and branches, or grass thickets) and makes venomous bite. Long teeth and powerful
venom kill bird or bat in a few minutes, and the smaller animals – for example,
taiga lizards or young snakes of the same species, which have a low sensitivity
to the venom of this snake – are swallowed alive, as a whole.
The viper itself must fear of owls and other predatory birds that attack this
snake from the top, falling from the sky. Predatory animals, even berls
and Siberian sabertooths,
prefer to avoid encounters with this snake: the venom of Siberian viper is not
fatal for animals of their size, but it causes rather long and unhealthy weakness
in the body, which easily overgrows into disease often capable of killing even
berl, especially when animal is young.
Besides the predatory birds and the intraspecific cannibalism, the main enemy
of Siberian viper is the Siberian frost. Like taiga lizard, this viper resists
it with the aid of thermo-insulating scales, layer of hypodermic fat and group
wintering. Sometimes, however, these snakes spend winter alone as well. Nevertheless,
in contrast to the lizards, vipers do not have a partially symbiotic connection
with local shaaz ants, but vipers winter in any convenient place, including
heaps of conifer needle on ground surface; however, larger size than lizards’
and the corresponding fat stock for wintering give this reptile a greater advantage
in resistance to winter cold, which compensates the wintering on ground surface,
where it is colder than under ground.
As in case of the taiga lizard, the breeding period at Siberian viper begins
in spring, when these snakes creep out from their burrows and shed their skin.
At this time females make odorous markers along their paths, and the smell attracts
males. Mating resembles a fight: two snakes wind themselves like a ball and
hiss in a drawling manner.
At the end of spring and in the beginning of summer the female viper gives birth
to several living youngs (in case of large females their number is up to 20)
less than half meter long. They differ from their parents only by size and begin
independent life immediately.
The lifespan of Siberian viper is about 60-80 years, maximum of up to 100 years.
This species of reptiles is discovered by Bhut, the forum
member.
Translated by Bhut.
Karelic,
or northern adder (Vipera karelica)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: taiga and krummholz of Northern Europe, east to the Urals.
Not all vipers managed to survive the age of man. Extermination of snakes, venom
gathering and habitat destruction have dealt a blow to a number of representatives
of the genus, causing their extinction or significant reduction in numbers and
range. Nevertheless, the common European viper survived. It appeared to be numerous
and widespread enough to survive not only the era of anthropogenic pressure,
but also the ice age. Moreover, with the stabilization of natural conditions,
this ancestor gave rise to a number of new species. One of them is the Karelic,
or northern adder.
This species is one of the northernmost snake species in Eurasia. Body length
is up to 85-90 cm; weight is about 180 g. The appearance of the Karelic addder
is quite recognizable: it is a brown or black snake with gray spots with a cervical
waist characteristic for the family. The belly is lighter, of bluish-gray color.
The reptile feeds on rodents, shrews, small weasels and ground-dwelling birds.
Snake venom is quite strong: a cat-sized animal dies from a bite, a harelope-sized
mammal gets sick for a long time, and larger animals have a painful tumor at
the bite site for several days, and blood appears in their urine (the venom
has a hemolytic effect).
The period of activity of this species is very short. Karelic adders leave to
the hibernation already at the end of September – October, having stopped eating
two weeks before that moment. Up to several dozen snakes gather in wintering
shelters. Hibernation usually takes place in a deep rodent burrow, or in natural
shelters between rocks. The coming out from hibernation is timed to the period
of steady warming: in late April – early May.
The rate of reproduction of this species is relatively slow: it is so due to
the high energy costs of raising offspring. Each female is capable of reproduction
once every 3 years, restoring the physical condition of the body during breaks.
Mating takes place 2 weeks after coming out from hibernation. Males find females
ready to reproduce by scent; if several males meet near one female, they fight,
intertwining with each other. Sometimes it happens that at this time the female
manages to mate with another male and just crawls away. After mating, the rapid
development of embryos begins in the female’s body, which consumes a significant
amount of the resources of mother’s body. The pregnant female becomes cautious,
basks in the sun for a long time and almost does not eat. After 2 months, she
gives birth to 5-6 small (12-15 cm long) well-developed juveniles ready for
independent life. During the delivery, it often wraps around a stone, root,
or the bottom of a tree. Young snakes stay for some days near their birthplace
until they shed for the first time. After that, they begin feeding, stocking
a reserve of fat for wintering. They reach sexual maturity at a length of 55
cm, by about 5 years. Life expectancy is up to 25 years, although 2/3 of females
do not survive the third birth due to profound physiological changes associated
with pregnancy. The enemies of this species are small predatory animals: owls,
hedgehogs, weasels and canids. Young animals often die during wintering.
This reptile species was discovered by Nick, the forum member.
Zoureg
(Vipera zureg)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: deserts of Central Asia.
The boundary of the Holocene and Neocene was a time of global changes in the
biosphere, when the species that survived in the human era were subjected to
a kind of test for survival and evolutionary flexibility. Viperids of the Holocene
epoch did not become extinct at this time, but their range and diversity in
Eurasia decreased. Africa has remained the center of a significant species diversity
of viperids, and Eurasia is inhabited by relatively unnumerous species, one
of which is the zoureg.
Zoureg is a somewhat unusual snake: it is a kind of ecological analogue of the
desert sand boa (Eryx miliaris) of the Holocene, adjusted for size and presence
of venom. It does not exceed 30-40 cm in length, and its body is adapted for
life in the thickness of free-flowing sand of Asian deserts. It is teretial;
the head is covered with thick shield-like scales (partly resembles the heads
of amphisbaenians) and has very small eyes. The cervical waist characteristic
of vipers is actually not expressed. Like the amphisbaenians, zoureg almost
does not come to the surface of the sand and has a sandy color lacking any patterns;
there are also lighter and darker color morphs. It lies just below the sand
surface and waits for prey – any small animals that this reptile is able to
swallow. Unlike crotalid snakes, zoureg cannot catch the heat of other animals,
but it has a keen sense of smell, and it is also able to sense the vibrations
of the medium surrounding it, giving out the movement on the surface of the
sand. Unlike crotalid snakes, it does not specialize in catching small mammals.
Zoureg has a disproportionately powerful poison that kills small animals very
quickly, and larger desert dwellers usually get sick from a bite for a very
long time. But more often zoureg avoids encounters with larger animals and hides
from them – feeling the shaking of the substrate, it “swims” very quickly in
the thickness of the sand and is able to crawl away from a dangerous place in
a few minutes. If a predator digs up sand, the snake can dig itself to the depth
of up to half a meter, almost completely holding its breath.
Zoureg’s main enemies are local monitor lizards, which are resistant to its
strong poison and are able to dig it out from under the sand and eat it, and
other snakes that can detect and attack it before it feels them. Cannibalism
is also characteristic of this species.
The breeding season of the zoureg begins in the spring: males of this species
search for females by smell, and after catching them up, they press females
by their body rings to the ground, because they are on average longer and slimmer
than females. This species is polyandrous: one female can mate with several
males; some weeks later she gives birth to one to four juveniles up to 15-16
cm long, but already indistinguishable externally from adults. If several juveniles
develop in the female’s body, it is likely that they may descend from different
males – this feature allows maintaining genetic diversity in the population.
They become sexually mature when they grow up to 25 (females) or 30 (males)
centimeters in length.
The life span of zoureg can reach 15 years, but many of these reptiles die earlier,
especially at an early age.
This reptile species was discovered by Bhut, the forum member.
Cyprus
kontonoura (Macrovipera cypriotica)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: Mediterranean hollow, Cyprus mountain oasis.
The drying up of the Mediterranean Sea has made the climate of North Africa
and southern Europe extremely hard – arid and hot. Moisture-loving animals left
this area, but the region of the Mediterranean hollow has become a place where
reptiles thrive, being adapted to the economical consumption of moisture. Mountain
“oases” instead of the former islands of the Mediterranean are the birthplace
of endemic species that developed in isolation. One inhabitant of this region
is Cyprus kontonoura, which is a descendant of the Levant viper (Macrovipera
lebetina), that has adapted to a new habitat.
This snake differs little from its ancestor – it has changed little in appearance,
its body is robust and quite thick, but it has become smaller (length up to
150 cm) due to more severe living conditions. The head is broad, with a blunted
rostrum and pronounced cervical waist. The coloration of the body has noticeably
changed: there are wavy transverse stripes on the back and dark brown spots
on the sides; the background color is sandy yellow. The eyes are quite large;
the night vision is good.
Like most reptiles, the Cyprus kontonoura is active around the clock, holed
up in a shelter only during the hottest daytime hours. In winter, it remains
active and hunts mainly during the day. This snake inhabits all areas of the
Cyprus mountain oasis, from the rocky foot to the forested top, and hunts both
in the forest and in places where trees do not form continuous thickets. In
extreme heat, the reptile crawls into the shelters and sometimes eats insects
hiding in burrows from the heat.
With age, the diet of this snake changes greatly. Young individuals feed mainly
on invertebrates (insects, spiders, scorpions, woodlice, snails and millipedes),
adolescent individuals hunt lizards and drying toads (Xerobufo
xeroderma), adults eat large lizards and birds. The Cyprus kontonoura
occupies one of the tops of the food pyramid. If a young snake can still fall
prey of predators (other reptiles or birds), then an adult snake has no enemies,
and it has every chance to live a long life while continuing to grow. A hunting
snake buries itself in leaf litter or lies in the shade of a shrub, which does
not allow possible prey recognizing the outlines of its body. When the prey
gets close enough, the snake attacks and puts it a venomous bite. The venom
has a strong hemolytic effect.
The courtship season comes at the end of winter and is accompanied by duels
of males who make “stands” and try to knock each other down by pushing with
their bodies. The fertility of this species is relatively low: after mating,
the female lays only 5-6 eggs in the forest litter, buries them with body bends
and crawls away. After 5-6 weeks, the juveniles hatch, dark brown in color without
a pattern. The initial diet consists of insects and other invertebrates. At
the age of about 2 months, young snakes begin to settle in new habitats and
switch to feeding on small reptiles.
Young snakes begin breeding at the age of 4 years; life expectancy is up to
27-30 years.
This reptile species was discovered by Mamont, the forum member.
Daeva,
spiny snake (Echinovipera daeva)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: South and Central Asia from the Persian Ridge to the Hindu Kush and
from the ocean to the Fourseas, in the mountains up to a height of 550 m.
The viperids, despite the human relation to them and the periodic extermination,
appeared a fairly common group of snakes of the Neocene. Despite the extinction
of some species during the era of anthropogenic pressure and the subsequent
ice age, they fully restored species diversity and range after the retreat of
glaciers. Numerous new species have appeared among the vipers, including very
large forms. One of them is daeva, an inhabitant of the arid regions of Asia,
a descendant of Levant viper (Macrovipera lebetina).
This reptile species is very large not only among vipers, but also among snakes
in general: the length of an adult snake is 3.5-4.5 meters; weight is up to
33 kg. The body color is dark, most often brown with a pattern of black cross
stripes on the back, although there are unicoloured gray and black individuals.
The head and back are overgrown with spines – these are modified strongly elongated
scales, slightly protruding and having pointed tips. When a snake lies in ambush
among rocks, stripes and spikes mask it. The reptile inhabits arid rocky areas
with sparse thickets of shrubs.
Daeva feeds on rodents, including quite large ones, as well as birds and large
reptiles, occasionally catches cubs of stoneloppers
and small predators. The venom of the spiny snake is very strong, quickly killing
even reptiles, and an adult stonelopper dies in convulsions 2 minutes after
the bite (hence the name: daeva is the evil spirit in Persian mythology).
The mating season lasts from April to early June. The male finds the female
with the help of the pheromones that she secretes during the mating period.
When approaching, the male tries to touch the female’s head with his head. If
the female is favorable to him, then the snakes begin a mating display, during
which their bodies intertwine. When the males encounter, both begin to reach
up, trying to raise their heads above the opponent’s head and knock him down.
Occasionally power struggle between them takes place, during which they hook
the enemy with their spikes.
Like its ancestor, daeva is an oviparous reptile. Around the middle of summer,
the female lays 10-20 eggs in a shallow hole and buries them with lateral body
movements, raking a small mound above them. Then she wraps around the clutch
and waits for the juveniles to hatch, guarding the clutch and driving away large
animals that happen to be nearby. She eats almost nothing, allowing herself
attacking only small animals that happen to be nearby. The incubation period
lasts 76-80 days. Young snakes hatch from eggs with the help of an egg tooth,
with which they break through the shell. After leaving the nest, they molt on
the 2nd-3rd day of life and begin hunting. Daeva reaches sexual maturity at
the age of 4 years at the length of 1.8 meters. Life expectancy is very long
– over 50 years.
This reptile species was discovered by Nick, the forum member.
Bird-eating
adder (Paratheris ornithophagus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: savannas of South Africa, areas of woody vegetation in river valleys.
Climate changes at the boundary of the Holocene and Neocene led to a reduction
in forest areas and to the spread of biotopes where herbaceous vegetation dominates.
Further climate changes in the early Neocene promoted the recovering of forest
vegetation and stimulated the evolution of species associated with this type
of habitat.
One species of tree-dwelling snakes in South Africa is a large bird-eating adder,
a descendant of a certain species of Holocene bush vipers (Atheris). It is a
large brown-green snake that lives in the tree crowns and bush thickets. The
length of an adult snake of this species is from 2 to 3 m, the male is larger
than the female. These reptiles are characterized by a triangular-shaped head
with a pronounced cervical waist, large eyes, tenacious prehensile tail and
elongated scales on the sides of the head forming two crests behind the eyes.
The build is very slender and gracile; the tail is about a half the total length
of the reptile.
The body color is brown with numerous grayish spots on the sides; the belly
is gray. On the upper part of the head there is a large diamond-shaped spot
with ragged edges and a black border, simulating a tree leaf, and along the
back a chain of black diamond-shaped spots with a green spot in the middle stretches.
In populations living in more humid forests, the scales on the back lean on
each other greatly and have a pronounced keel.
This species feeds mainly on birds ranging in size from sparrow to pigeon, and
less often catches arboreal rodents. The bird-eating adder hunts from ambush
and lies in wait for prey, curled up in a thicket of branches. It easily catches
birds perching on branches and can even seize a flying bird, being fixed on
a branch with its tail. The bite of this reptile is venomous and kills prey
within seconds. The snake swallows its prey, hanging upside down from a branch.
Despite the presence of venom, when an enemy (large bird, monkey or a large
animal moving through the bush) appears, this reptile most often freezes among
the branches, hoping for its camouflage color. At such moments, the snake’s
body is partially suspended and curved, simulating nearby branches. Such trick
is usually enough to avoid danger. The bird-eating adder bites only when it
is impossible to avoid a meeting; large animals get sick from its bite for a
long time, and smaller animals can die.
The breeding season of the bird-eating adder falls at the end of the rainseason.
The male finds the female by smell; if several males meet near one female, they
arrange duels for the right to mate, twisting on the branches in a spiral. A
tangle of struggling snakes often rolls down to the ground and the struggle
continues below for a while. The bird-eating adder is viviparous, like its ancestors.
The female gives birth to 3-4 juveniles 30 cm long. After the second molt, they
begin to feed on insects, nestlings of small birds and rodent cubs. Sexual maturity
comes at the age of 5 years, and adult reptiles live up to 55 years.
A related species, the golden-eared
adder (Paratheris chrysotus), lives in the sparse woodlands and dry
forests in the south of Zinj Land. It is characterized by smaller size (length
no more than 170 cm) and a larger head. In this reptile, elongated scales on
the head form two pronounced “crests” of lemon-yellow color, sticking out sideways
and backward behind the eyes. This species clearly prefers to hunt arboreal
rodents and bats, which make up about 2/3 of its diet, and has mainly nocturnal
activity. The color of this snake is gray with longitudinal strokes of brown
color, simulating the pattern of tree bark.
This reptile species was discovered by Nick, the forum member.
Incanyamba
(Hydrocausus incanyamba)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Vipers (Viperidae)
Habitat: rivers of South Africa, north to the Tanganyika Strait.
When the ice age ended and the climate became more humid, some species of land-dwelling
snakes from various groups passed independently from each other to an aquatic
lifestyle. In the non-numerous rivers of South Africa, night adders (Causus)
appeared one of these groups. In the Neocene, one of their descendants turned
into the incanyamba – a large freshwater reptile. The name of this species was
given after a large water snake from Zulu myths. Another aquatic viper species
lives in the Saharan Nile – it is the river adder (Potamobitis
planiceps), descended from the Gaboon viper (Bitis).
Incanyamba is a fairly large reptile: the body length is about 3-5 m, females
are larger than males. It leads an almost completely aquatic lifestyle and rarely
gets out on land. However, this species has not lost the ability of moving on
land, and during a drought, a snake can easily crawl from one reservoir to another.
In its appearance, inkanyamba resembles a cross between a viper and a python:
it has a viper’s flat head of a characteristic appearance, but the cervical
waist is unclear; the body is slender and resembles more the python’s body.
The coloration is usually brown with a longitudinal “marble” pattern: incanyamba
often hides at the bottom among the leaves, or buries itself in the silt near
the shore. The belly is usually white or yellowish. Unlike its ancestor, the
specialized herpetophage, this snake feeds on chicks of waterfowl and small
mammals that have come to drink. Occasionally, the cubs of medium-sized mammals
and small birds that have fallen into the water fall its prey. This snake catches
up with the prey in a rush; having sunk its teeth into the prey, it fixes the
attacked animal with two or three turns of its body and does not free it until
death from venom occurs.
In drought, these snakes hide in reservoirs that remain in the deepest places
of the riverbed. If the river dries up completely, the reptiles leave the riverbed
and look for large burrows or natural spaces between the stones, where they
wait out the drought in a condition resembling aestivation. Animals prefer to
go out on land in the early evening or in the morning, avoiding the heat of
the day and the cold of the night.
At the beginning of the rainseason, this snake begins courtship games. The reptiles
return to the river and their mating games take place in well-warmed shallow
waters. Males seek out a female by the secretions emitted from her cloaca. During
the courtship ritual, the male wraps around the female, bites her side slightly
with the edge of his mouth and pushes her into shallow water. If several males
appear near the female at once, they begin to fight with each other, twisting
their bodies and trying to squeeze the opponent as much as possible. Sometimes
a tangle of struggling snakes even turns out to be on the shore, where they
roll around not loosening their embraces until the strongest of the males remains.
The weaker opponent usually shakes his whole body (the release signal) and the
winner releases him. However, during the mating duels, it happens that the females
are fertilized by less aggressive males who do not take part in grueling duels.
The female lays 20 eggs in the sand or a pile of plant debris on the coast.
After 4 months, young snakes hatch. At 3 years of age, with a length of about
2/3 of the average size of an adult, the incanyamba reaches sexual maturity.
This species lives about 20-25 years.
This reptile species was discovered by Nick, the forum member.
Swamp
rattler (Agkistrotalus giganteus)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Crotalid snakes (Crotalidae)
Habitat: North America, marshlands of warm-temperate climate.
During the time of human existence many large species of animals had undergone
to persecution for various reasons. Among them there were also large reptiles:
crocodiles, turtles and snakes. In human epoch the area of crocodiles had been
reduced in great degree because of extermination and destruction of habitats
of these animals. The ice age at the boundary of Holocene and Neocene also had
caused extinction of residual populations of these animals. As a result only
few species of crocodiles and caymans had remained from four families of crocodiles,
existed in the beginning of human era.
With the disappearance of the basic predators of fresh waters various species
of other groups began making attempts to take a place of the top predators of
ecosystems. Monitor lizards have succeeded in Old World in it, and even the
separate family of water monitors (Aquavaranidae) had evolved. In North America
monitor lizards were absent, and the place of the top predator of freshwater
ecosystems the snake had occupied here. It is swamp rattler – the descendant
of cottonmouth (Agkistrodon piscivorus), semi-aquatic crotalid snake.
In comparison with the ancestor swamp rattler is very large reptile: old individuals
may reach 2,5 meters in length (the sexual maturity comes at one and a half
meter length), but in the rest the similarity caused by relationship is quite
appreciable: it is a snake of dark colouring with characteristic white mucous
membrane of mouth. Besides the size, this reptile has two more characteristic
attributes.
Swamp rattler has slightly lengthened muzzle, and larger head, rather than snakes
of comparable size. Accordingly, at this reptile large pointed teeth were developed.
Jaw bones, as well as at all snakes, are connected by extensible ligaments that
permits this reptile to open mouth widely. Such features of skull allow swamp
rattler to catch and to eat even rather large prey, for example, small heron-sized
waterfowl. Body of this snake is large and thick, therefore snake is very heavy
for its length: the adult two-meter individual weighs about 20 kgs.
Though swamp rattler is only distant relative of rattlesnakes of Pleistocene
and Holocene, parallel evolution had made its own rattle which makes quite heard
sound which can serve as the warning for large animals casually appeared near
it, though it is small in comparison with typical rattlesnakes, both from human
epoch, and from Neocene. If the animal does not go away, snake can warn it otherwise:
having bent forward part of body vertically, reptile opens mouth and displays
white mucous membrane. Thus maxillary bones move in sides, increasing the seen
size of mouth. If the animal does not leave or continues an attack, the snake
counterattacks and delivers bite by poisonous teeth.
This snake prefers to settle in densely overgrown reservoirs with slow current,
sites of reed and areas of open water.
Swamp rattler eats basically warm-blooded animals: mammals and birds living
near water or in water. Young snakes eat murine rodents and chicks of waterfowl.
Large adult snakes easily swallow swan-sized birds, and even herons. Snake swallows
such long-legged and long-billed birds, snapping by shoulders and the basis
of neck. At swallowing the neck of prey is bent back; beak and legs of bird
stick out from its mouth for any time and at swallowing they will not wound
a wall of digestive path.
As well as other crotalid snakes, swamp rattler gives rise to live youngs. In
typical case one female brings about 8 – 10 young snakes up to 50 cm long. They
grow quickly and reach sexual maturity already at the age of 3 – 5 years. In
southern parts of area growth of this snake is faster. General life expectancy
of swamp rattler may be 80 years and more. Active growth proceeds during the
first third of life; further the snake grows much slower.
In the north of an area activity of this snake has a seasonal rhythm. In winter
when the temperature of water falls down to +10°C, these snakes practically
do not eat and spend the most part of time on land near shelters – trunks of
trees with rotten out core or near holes. If there is a short-term cold snap
and frosts, the reptile does not leave shelter and runs into not deep catalepsy.
In the south of area activity of this snake is kept the year round.
This species of snakes was discovered by Bhut, the forum member.
Velachief
(Aquabothrops velachief)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Crotalid snakes (Crotalidae)
Habitat: Central America, freshwater reservoirs.
During the existence of humankind, large reptile species had a hard time, especially
crocodiles and turtles. But while turtles partially returned to their former
prosperity after the human disappearance and the climate stabilization, even
forming their own ecosystem at the New Tortuga
island, then crocodiles and their relatives had almost disappeared: only some
rather specialized
species of this group persist on the
Earth. Their place in the ecosystem was taken by a variety of animals – large
fish and caudate
amphibians, freshwater turtles,
as well as various lizards. Snakes
also passed to an aquatic lifestyle: in North America, there is a swamp
rattler, a descendant of the Holocene swamp moccasin, and in Central America,
there is another aquatic crotalid snake – velachief.
Velachief is a medium-sized reptile, about one and a half meters long, sometimes
longer. This species has a long, slender, laterally compressed body, especially
in the tail area. On land, this snake feels insecure and does not crawl far
away from the water. At the same time, its head looks quite large because of
the well-developed venom glands. If this reptile is attacked by a predator (mammal
or bird, large predatory fish in the water), the velachief hisses threateningly
and opens its mouth wide, displaying the mucous membrane of the mouth – it is
red, surrounded by white lip shields in this species. Velachief is a descendant
of American lanceheads of the Holocene epoch.
Velachief has a variegated coloration – on a dark brown, even black background,
lighter, brown and reddish-brown irregular transverse stripes stretch. The pattern
on the skin makes this snake inconspicuous, especially in water and under water,
where it often waits for prey, slightly buried in the bottom silt and the remains
of plants. Like at other aquatic reptiles, the velachief’s eyes and nostrils
are set high on the head, so the reptile can only stick out a small part of
its head to breathe and look around. The thermolocation pits characteristic
of its family have been preserved, but their temperature sensitivity is weaker
than that of other crotalid snakes: most of the prey of this species are various
fish, amphibians and large freshwater crustaceans; warm-blooded birds and animals
also fall its prey, but less often. However, the thin skin in these places is
very sensitive to the waves spreading in the water, allowing it determining
the presence of animals in the neighborhood. Larger animals sometimes attack
the velachief, but it is mainly saved by camouflage coloring and the ability
to hold its breath for a long time, as well as a secretive lifestyle. Velachief
avoids large bodies of water, preferring shallow lakes and swamps overgrown
with amphibious plants.
Velachif is a viviparous species that has no seasonality in reproduction. Females
usually give birth to 3 to 5 juveniles 15-18 cm long. They resemble adult snakes
in everything and are independent from the birth, feeding on small crayfish,
aquatic insects and fish. Intraspecific cannibalism is very rare in this species.
Sexual maturity comes in 2-3 years. The average life expectancy is up to 30-40
years, but some individuals live much longer.
This reptile species was discovered by Bhut, the forum member.
Leaf-headed
snake (Foliocephalophis imitator)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Crotalid snakes (Crotalidae)
Habitat: Florida, tropical forests.
Climatic changes in the Neocene caused the appearance of extensive rainforests
in the southern part of North America. Their area stretches as a strip along
the coast of the Gulf of Mexico, and the largest arrays are located in Florida,
and at Cuba and Great Antigua islands. The development of complex multilayered
forests contributed to the evolution of local animals and the emergence of a
diverse forest fauna. Among the characteristic representatives of the local
fauna, there are reptiles that thrive in a tropical climate.
The leaf-headed snake, a small tree-climbing reptile well adapted to an arboreal
lifestyle, lives in the upper level of the tropical forests of Florida. The
appearance of this species is very distinctive and recognizable. The body of
the leaf-headed snake is thin and gracile, up to 150 cm long. The snake mimics
the vine stalk very convincingly by the coloring of its scales: it is green
with transverse yellowish bands imitating the nodes of the stem. On the green
background there are numerous brown and yellowish longitudinal strokes, making
the reptile’s body looking even more like a vine. The leaf–headed snake often
hangs among the vines, hooked by its tail, and spends a long time in immobility,
waiting for the appearing of prey – lizard, frog, small bird or large insect.
Sometimes small arboreal rodents or bats, which it catches during daytime rest,
fall its prey.
The distinctive feature of this species is the shape of its head – it is wide
and leaf-shaped, with triangular horns above the eyes, directed to the sides.
The color of the upper side of the head is bright green; an intermittent brown
border, the pattern of spots of which is individual, stretches along the head
edges. The underside of the head is white. The tip of the muzzle is slightly
bent up.
There are two venom-injecting fangs in the mouth. The venom of this reptile
is very strong: a sparrow-sized bird dies from it in less than a minute. On
the underside of the head, slightly before the eyes, there are sensitive pits
of thermolocators that help to detect warm-blooded prey at dusk or early in
the morning.
The leaf-headed snake inhabits the upper level of the forest, preferring parts
of the forest with an abundance of lianas. This reptile arranges an ambush,
disguising itself among the stems and branches; at this time, most part of the
body is held in the air, and the tail and the back of the trunk wraps around
the support.
Sexual dimorphism is almost not pronounced in these snakes: the male differs
from the female only by a longer tail. During the courtship, he wraps around
the female’s body and sometimes even hampers her by tying her with his own body
to the vines. Copulation takes place in “suspended” condition and lasts about
an hour.
This snake species lays eggs. The fertility of the female is 3-5 eggs, which
are most often laid in a tree-trunk hollow, among rotting wood. Incubation lasts
up to 15 weeks. Young leaf-headed snakes differ from adults in the absence of
outgrowths above the eyes and a blunted tip of the muzzle. As they grow up,
they change their body color to green with transverse rings, and then they grow
characteristic outgrowths on their heads. By the time of sexual maturity (in
the third year of life), the appearance of the leaf-headed snake becomes characteristic
for adults of this species. The lifespan of these snakes reaches 25 years.
Tzaotu
(Gloydius tzaotu)
Order: Squamates (Squamata), suborder Snakes (Serpentes)
Family: Crotalid snakes (Crotalidae)
Habitat: steppes of Central Asia – from the southern border of forests to the
marshy plains of East Asia and from the Fourseas to the Pacific Ocean.
In the historical era, the range of Asian ground pit vipers decreased due to
the plowing of the steppes, and during the ice age, some species and populations
disappeared. However, this group escaped from extinction due to specialization
in feeding on small animals – this food resource was available even at the peak
of the global ecological crisis. In the Neocene, their diversity has increased
compared to the human era.
Tzaotu is a descendant of the Siberian pitviper (Gloydius halys) common in the
human epoch. It is a large species: the length of the male is up to 2.25 m,
the females are 35 cm shorter (hence the name: Tzaotu is one of the Chinese
dragons). The general background color is yellowish-grayish-cream, sometimes
olive-brown or gray-brown (the color combination varies in different populations).
On the body there is a pattern of a chain of large dark spots on a lighter background,
small dark specks are scattered on the sides. At this snake species, the whitish
edges of some scales create a thin light mesh pattern on the dark background
of spots on the back. Sexual dimorphism is expressed in this species: in males,
the belly is pearl-white, in females it is yellow.
Tzaotu feeds on rodents, small terrestrial birds and reptiles, clearly preferring
mammals. Sometimes an adult snake attacks larger animals: it can swallow a heterolopa
or a zibetonyx cub, or a dwarf
harelope. However, it is a rare piece of luck: a snake does not often venture
attacking such prey for fear of injury. However, the large prey is enough for
reptile for a month: the snake prefers to spend this time in shelters. Tzaotu’s
bite is very painful; the venom has a hemolytic effect. An adult heterolopa
dies from it in 30 minutes, larger animals will be sick for a long time, but
they are likely to survive. In spring and autumn, this snake is active during
the day, and in summer it turns to a crepuscular lifestyle, spending the hottest
daytime hours in the shelter. The coming out from wintering takes place from
the beginning of March to the end of April depending on the geographical latitude.
Snakes rest most often in burrows and under rocks; they also winter there, simply
deepening the summer burrow. Comfortable shelters are expanded by movements
of the head and body; the reptile throws the ground out of the shelter with
the bends of its body. Sometimes the snake occupies the burrow of a farmer
hamster, having swallowed its owner before that.
The mating season begins about 2 weeks after the ending of the wintering and
lasts for a month. Tzaotu mating is characterized by the so-called mating dance,
when the male and female wrap each other with the back halves of the body, lifting
their heads and the front parts of the body up. In this position, they can crawl,
rubbing against each other at the same time. Competing males, on the contrary,
try to bend the opponent’s head to the ground and press it down with their weight.
At the moment of mating, the male holds the female’s neck with his jaws. After
mating, the male stays close to the female for a while, driving rivals away,
but then leaves her.
Tzaotu is a viviparous reptile species. In August, the female brings 3 to 5
juveniles with a body length of 20-22 cm. They feed on insects and baby rodents.
Sexual maturity comes at the body length of 1.2-1.4 meters, at the age of about
3 years. Life expectancy is up to 50 years. Tzaotu’s natural enemies are deinapers
and birds of prey, juveniles can be eaten by zibetonyx, cats and lizards.
A related species, the swamp
tzaotu (Gloydius paludidraco), lives in the wetlands of East Asia.
It is smaller – only 1.6-1.9 m long, brown in color with a blurred spotted pattern
and light yellow eyes. This species is more moisture-loving; it prefers swampy
forests and shrubs, and is a good swimmer. This species feeds on small mammals,
chicks of wading birds and amphibians, hunting only during the day. Otherwise,
the biology is similar to the steppe species.
This reptile species was discovered by Nick, the forum member.
Armored
crocodile (Neomekosuchus loricatus)
Order: Crocodiles (Crocodylia)
Family: Crocodiles (Crocodylidae)
Habitat: Meganesia, wooded tropical swamps.
Crocodiles of the Neocene epoch represent a group on its decline, superseded
by a variety of animals: monitor lizards,
sharks and even turtles. Due
to human activity, most of the crocodiles died out, and the rest adapted to
the new conditions. Some of them became marine
inhabitants, while others, on the contrary, returned
to the land. One of the land-dwelling species is the armored crocodile,
a descendant of the Johnstone’s crocodile.
There are many examples of terrestrial crocodiles, but they all appeared on
various continents only in the absence of advanced placental mammalian predators.
Australia, therefore, was the center of the evolution of these strange animals,
because until the human era there were almost no placentals here. Then, after
numerous epizootics of distemper and rabies, placental predators died out, and
now the niche of a large terrestrial predator has become vacant again. However,
monitor lizards, being true land-dwelling animals, are much better suited for
this role, and crocodiles still remain tied to their niche of a semi-aquatic
predator. Semi-terrestrial crocodiles, which have never become the rightful
owners of the savannahs and forests of Meganesia, appear in the flooded tropical
forests of the north of the continent. Their representative is the armored crocodile.
This reptile is quite modest in size, 1-1.5 meters long, weighting up to 30-35
kg. Like the dwarf caimans from the
New World, this species is characterized by a high-set head and long limbs.
The position of the limbs has also changed: now they are directly under the
body, just like in mammals. Because of their small size, armored crocodiles
have become more vulnerable to predators, so their osteoderms are even better
developed and form a strong shell on the back and tail. Behind the eyes, there
are large bony protrusions resembling horns.
The reptile’s muzzle is narrow and relatively short. It has large brown-red
eyes indicating a nocturnal lifestyle. The black color masks the animal at dusk;
the motionless crocodile is almost indistinguishable from a stone. In the dark,
they come out of shelters and reservoirs and start searching for all kinds of
food: large insects, snails, crabs, fish and small vertebrates. Young crocodiles
like to ravage bird nests arranged low from the ground. This is their peculiarity:
unlike adults, they often spend a long time in the trees, basking in the sun.
Very often small crocodiles lay on top of each other, periodically pushing a
neighbor off a branch. When the sun rises, they return to reservoirs or hide
in holes dug near the shore or under a snag. Due to the well-developed shell
of osteoderms, they rarely fall prey of terrestrial predators, but some large
birds with powerful beaks manage sometimes to kill an adult. In case of danger,
armored crocodiles quickly rush into the water, or defend themselves by trying
to bite.
Armored crocodiles are not territorial, but each one has its own burrow in which
the reptile spends a lot of time. Sexual maturity comes at the age of 5. The
mating season begins in the middle of summer and is accompanied by a ringing
squeak uttered by numerous males. Every year at this time, crocodiles meet their
permanent mate living in the neighboring territory (this species is monogamous).
After mating, another month passes, and the female lays eggs in her burrow.
Depending on the temperature, incubation lasts from 60 to 90 days. For the first
few months, the juveniles will live in the vicinity of the nest under the care
of their mother, but then they will have to move away.
The life expectancy of the armored crocodile is 30-50 years.
This reptile species was discovered by Gleb_97, the forum member.
Meganesian
crab-eating crocodile (Brevicroco destructor)
Order: Crocodiles (Crocodylia)
Family: Crocodiles (Crocodylidae)
Habitat: northern part of Meganesia, Indonesia, the nearest islands in the tropical
Pacific Ocean.
In the Neocene, crocodiles largely lost their positions of the top predators
of freshwater ecosystems. It happened mainly due to human activity, which undermined
the number of most species of these reptiles. In the tropics of the Old World,
aquatic monitor lizards, and in the
New World – fish and
turtles have become ecological
analogues of crocodiles. Meganesia was one of the few places where crocodiles
retained a sufficiently high number and diversity to struggle for existence
in new conditions. In addition, specialization has become one of the ways of
survival.
In the coastal waters and on the reefs of the northern coast of Meganesia, the
Meganesian crab-eating crocodile lives – a saltwater crocodile with a short
wide muzzle, very powerful jaws and crushing back teeth. This reptile species
feeds on prey with hard body coverings: crabs and shellfish,
as well as juvenile sea turtles (it
is too slow for fishing). It is a 3-meter stocky crocodile with very powerful
webbed hind legs and a thick dorsal shell composed of osteoderms. To protect
against bites, the osteoderms on the neck have a conical shape. The tail of
this reptile is deep and compressed from the sides. Usually, a crab-eating crocodile
swims with the help of its hind legs, but in case of danger, it uses its tail
and for a short time develops a sufficiently high speed to escape from an attack.
The reptile’s back is colored very light green with a pattern of short transverse
strokes of a darker shade. The belly is milky white with a bluish tinge.
This species does not form large congestions like freshwater croc species, and
exhibits pronounced territorial behavior. It lives and feeds in monogamous pairs
– this way it is easier to notice the danger in the sea. The pair persists for
many years in a row. During the rain season, the female lays about 8-10 very
large eggs in the nest, which she arranges above the level of the highest tide,
and guards the nest in common with the male. Incubation lasts about 2 months,
and all this time the female feeds only on random prey and leaves the nest only
briefly. When the offspring hatch, the female digs out the nest and carries
the juveniles to shallow water where sharks and sharkodiles
do not appear. To raise offspring, reptiles use a small shallow bay, which entrance
is protected from the surf by reefs. The female often carries the juveniles
on her back and feeds them: after catching a crab, she leaves its cracked legs
and claws to the juveniles. The male hunts nearby at this time and protects
the approaches to the “nursery” from possible danger. Sexual maturity comes
at the age of 7-9 years.
Adult reptiles take care of the posterity for about a year, but later adult
reptiles do not drive them away from their territory, since adult and young
crab-eating crocodiles hunt different invertebrates. The young ones are more
mobile than adults and eat shrimp and small bottom fish; the grown up juveniles
are expelled as potential food competitors. If the usual food is not enough,
the crab-eating crocodile hunts large prey – young saw-nosed
crocodiles and even sharkodiles. They also use the tactics of a “live trap”,
but they do it in fresh water, hunting mammals and birds at a watering hole.
However, such hunting is usually less successful due to its too light coloration,
and brings a result only in thickets of coastal plants.
This species has large air sacs that provide an additional supply of oxygen
and buoyancy, and can easily settle to the islands separated by tens of kilometers
of sea, and dive to a depth of up to 100 m, where false
rudistes mollusks still grow, which it feeds on.
This reptile species was discovered by Dmitry Korshin, the forum member.
Land
Caiman (Ziphocaiman terrestris)
Order: Crocodiles (Crocodilia)
Family: Alligators + Caimans (Alligatoridae)
Habitat: Tropical plains of central South America.
Picture by Tim Morris
Large, dangerous crocodiles suffered during the Holocene, persecuted and
hunted by man. Few larger Crocodylians survived into the Neocene. Smaller
types fared
better, the African Dwarf Crocodile and Johnston's Freshwater crocodile prospered
because of their less intimidating nature. In South America, smaller Caimans
of the genus Paleosuchus evolved into new forms.
As the South American continent moved southwards, parts of caiman habitat
became dryer and more open. Paleosuchus already had some predisposition towards
land,
excavating burrows, travelling over land on occasion, and being able to lift
its head and neck above its body, unlike its cousins.
Neosuchus became more terrestrial in the Neocene, becoming the Land Caiman
(Ziphocaiman terrestris). This caiman shows clear adaptations to a terrestrial
lifestyle. Its snout is deeper and less flat, slightly resembling the face
of a dog or theropod dinosaur, its head is held high. Its legs are longer
and more muscular, and hold the animal in a more erect gait; its feet are
more
compact and less splayed, ending in conical claws for traction. It is able
to move quickly by a gallop and fast walk. The middle rows of dorsal armor
are very prominent and well developed, attaching very tightly to the back
muscles. Its tail is less paddle-shaped, and more rounded in cross-section,
like that
of a large lizard, and it is held higher. Its teeth are serrated and curved
like a theropod dinosaur, being Ziphodont.
The animal is colored a light caramel brown with darker banding, along the
side the banding becomes mottled spots, and the belly is a beige-cream color.
Its osteoderms lack the serrated crest seen in its ancestors.
The Land Caiman measures 3-3.5 meters long and weighs up to 80 kilograms.
It preys predominately on small to medium sized animals such as large rodents,
ground birds, lizards and snakes, Deermara and young of the Giant Paca are
taken. It is like all crocodiles, an opportunist, scavenging and plundering
eggs and nestlings. Each Land Caiman excavates at least one burrow, which
is
several meters long, it is most active in the afternoon, morning, and twilight,
sheltering during the middle of the day.
The mating season for land caimans is during the end of spring and the beginning
of summer. Usually they are aggressive at close quarters, being able to avoid
each other actively by sight on the plains. In the breeding season, males
will choose a patch of ground fairly close to their burrow, and will call
in the
afternoon with loud bellows. The male will mate with any females that arrive,
and will repel any interloping males with biting and shoving. The female
will bury the 15-20 eggs in a nest of rotting vegetation, which she usually
places
inside her burrow, blocking access to it by sitting at the entrance, actively
defending against intruders. The young will remain with her mother around
her burrow for up to a fortnight, after which they disperse into nearby undergrowth.
Lifespan of the Land Caiman can be from 60-70 years, older males usually
accumulate scars and scrapes across their armor.
In the Subtropical Pampas to the south occurs a smaller relative, the Grass
Caiman (Ziphocaiman varanoides). This Caiman reaches between 1.5
and 2 meters and weighs up to 35 kilograms, and is lighter in coloration
than its relative.
It feeds mostly on small animals, particularly invertebrates, lizards, young
birds and small rodents. The mating season is in late summer, and the female
lays up to 20 eggs. Lifespan of this animal is between 40-55 years.
This reptile species is discovered by Timothy Donald Morris, Adelaide, Australia.
Amphibians
|
Cave
wormsalamander (Typhlodraco branchiatus)
Order: Tailed amphibians (Caudata)
Family: Salamanders (Salamandridae)
Habitat: caves of Balkan and Near East.

Picture by Alexander Smyslov
Caves represent the specific habitat. The cave environment
substantially differs from surface of the ground. Here there is no light; therefore
biological
efficiency
of caves is very low: plants practically do not grow here. Only bacteria
can provide food needs of inhabitants of caves in any degree. The majority
of food
chains of cave ecosystems stretches to the ground surface. Bats, birds and
insects deliver to caves organic substances from world around which animals,
never seeing sunlight, eat. Also organic substances can get in caves at river
floods.
But caves differ in stability of conditions of inhabiting: here daily and
seasonal fluctuations of temperatures are practically not felt, and humidity
of air
is constant and close to 100 %. Not all live creatures can adapt for life
in conditions of eternal darkness, but nevertheless caves are plentifully
inhabited.
Amphibians are largest inhabitants of these places in Europe.
In caves frequently there are reservoirs settled by various live creatures.
Usually there are worms, small crustaceans and fishes in them. Their food
includes rests of plants and the dead animals delivered by water. But above
them the
predator reigns, crowning feeding pyramid of caves.
In rivers flowing from Balkan caves, it is possible to find unusual animals.
They swim like small fishes, but their body is covered with naked skin, and
on each side of heads three pairs of soft feather-like pinkish gills stick
up. But at them even a rest of legs is not present, and eyes are closed by
transparent thin skin. Bodies of these creatures are delicate, and through
skin on the belly interiors appear through. They obviously are larvae of
any amphibians.
The river has taken out young growth to the surface, but their parents have
remained in gloomy world of cave. Here, in outer darkness, they search for
livelihood - other inhabitants of cave rivers. Naturally, these creatures
are larger than larvae meeting at the surface. Their bodies are pale, and
eyes
completely are absent. Only two stains indicate their past site on head.
Parents are as poorly similar to any adult amphibian, as their posterity.
The inhabitant of caves, the cave wormsalamander is neotenic larva of one
of salamander species dwelt once at the ground surface. Escaping from climate
cooling and drying in ice age, ancestors of these animals have receded in
damp
caves. Life in gloom has left traces at shape of these animals, having transformed
them to underdeveloped, but nevertheless adult larvae.
The cave wormsalamander is legless worm-like animal about 40 cm long. About
40 % of length of animal tail accounts. At this amphibian there is delicate
translucent skin of rosy-white color through which interiors appear through.
But at animals living in caves where the sunlight gets, pigmentation of skin
develops: small white spots on brown background. This animal spends all life
in water, therefore at it there is mainly branchial breath with the help
of three pairs of external feather-like gills. Because of them this animal
resembles
by something fantastic dragons (the name of animal means “blind dragon”),
invented by people which had missed long time ago. But by properties it is
rather far
from frightening prototypes.
At the wormsalamander also underdeveloped lungs have remained, and the animal
from time to time emerges to surface of water, swallowing a portion of air
with silent smacking sound. Air in lungs serves this amphibian as certain
analogue of swimming bladder, helping to remain swimming.
In darkness of cave sight is not necessary, therefore the wormsalamander
is completely blind. But instead of sight at it other sensor organs are advanced.
At the wormsalamander the lateral line, detecting vibrations of water, proceeding
from swimming or creeping live creatures, is well advanced. Also it has bodies
of electric feeling allowing more precisely to determine presence of live
creatures.
The tail is bordered from above and from below by fin plica.
The male is larger than the female; at him tail is longer, making up to half
of general length of animal. Ritual of courtship is reduced to some necessary
actions: male finds female by smell, catches her up and snaps by tail fin.
Twisting around of body of female, male keeps her, and begins courtships.
He swims around the female, rubbing by throat against her body, and from
time
to time pushing her by head. When courtship ritual reaches apogee, male emits
spermatophores on stones.
Wormsalamander is a live-bearing amphibian; in oviducts of female within
3 months after fertilization only two larvae about 10 cm long develop. They
are
a little more advanced, than adult amphibian of this species: at them eyes
covered with thin translucent skin are more advanced.
Larvae can distinguish light and darkness, and vaguely distinguish contours
of subjects. They have very important role in life cycle of this species,
settling from one cave to another through reservoirs at the ground surface.
Usually
it happens during rain season when underground rivers overflow with water
and leave on surface.
Similarly to all representatives of amphibians, the cave wormsalamander is
a predator. It eats any live creatures which can find in caves: fry, small
crustaceans, and sometimes even its own posterity. Metabolism at this animal
is very slow, and after plentiful meal the wormsalamander may desist from
eat about one month.
Midwife salamander (Batrachodraco parentalis)
Order: Tailed amphibians (Caudata)
Family: Salamanders (Salamandridae)
Habitat: North America, the mixed forests of the Atlantic coast.
In Neocene variety of anuran amphibians almost does not differ from their variety
in human epoch. Anurans are the largest and the most successful group of the
classus. As against them, caudate amphibians look rather conservative group.
They are still characteristic only for northern hemisphere, and penetrate far
to the south only along mountain ridges. The majority of them has not developed
any unusual adaptations, helping to survive in non-typical inhabitancy or to
care of posterity. But among them the species had evolved, which had developed
unusual for caudate amphibians way of care of posterity.
Usually caudate amphibians leave the fertilized eggs to the mercy of fate.
Also among them there are the species taking places at various stages of transition
to vivipary. Some species protect eggs, staying near to clutch during the incubation.
But in Neocene at the territory of North America the separate species of salamanders
has appeared, which displays the wonderful for caudate amphibians form of care
of posterity – the parental individual bears eggs on itself. By analogy to
midwife toad of human epoch this species is named as midwife salamander.
As against its European “double”, at American midwife salamander only the female
cares of posterity. She has characteristic shape which is connected to features
of care of posterity. This species has clearly expressed sexual dimorphism.
The female of midwife salamander is an amphibian about 30 cm long, of rather
heavy constitution, with strong paws and rather short tail. Its background
color is dim reddish-brown color with small white spots on head and paws. On
hands and feet spots become larger and merge to uniform color field. In this
feature it differs from other species of salamanders only a little, but in
its shape there is a remarkable feature – a line of skin outgrowths forming
some kind of crest stretches from neck up to middle of tail. Because of it
the animal resembles tiny dragon a little. It has wide shovel-like head and
small eyes on its edges. Nose and edges of upper jaw of this species are covered
with cornificated callouses, permitting to dig out wood litter in searches
of beetles, their larvae and worms which this animal eats. Also this salamander
willingly eats snails and slugs. The female of midwife salamander lives on
the ground in thickets of plants, where air is constantly humid.
Male of this species is the creature of significantly smaller size and lighter
constitution. The maximal length of male does not exceed 20 cm. It has greyish-brown
colouring of body with short vertical strokes. On its back the crest also stretches,
but it differs in great degree from crest of the female: it lasts from shoulders
up to the basis of tail and has not numerous, but rather large blades. Blades
of crest have dark brown colouring with orange-yellow edges. Their colouring
very precisely imitates tinder funguses of some species. Male is able to climb
on trees and frequently lives among thickets of moss at height up to 2 meters
above the ground. In case of danger it simply turns body across tree trunk
and hangs on bark. Blades on its back imitate fungi, and it succeeds to avoid
dangers. It eats smaller insects which can be found among moss – cockroaches,
earwigs and larvae of small beetles.
The reasons of occurrence of crest become especially obvious in courtship season
– in spring. When days become warmer, males actively start to search for females.
They turn brighter – edges of skin blades get bright red colouring (and the
border becomes much wider), and on sides white spots appear, “flowing down”
to the stomach. Courting the female, male creeps in front of her, displaying
to her the crest and from time to time tiny shuddering by the whole body. If
the female is ready to breeding, she admits male closer to herself, and soon
he simply creeps on her back. The female creeps away th the thickets, and male
finds on her side suitable place where leaves spermatophore on skin of the
female. After that he quickly leaves her body. The female takes spermatophore
by paw and places it to the cloaca. In ovoducts of the female the internal
fertilization takes place.
In one week after fertilization female starts to lay eggs. At this time there
are appreciable changes at its crest – the skin swells and becomes softer.
The female rises on paws, holding one of hinder legs directly under cloaca.
When egg leaves a cloaca, it at once drops in toes of animal, and female puts
it to lateral face of one of blades of crest. The environment of egg is pasted
to skin of the female, and the amphibian pastes the next egg to crest. Pasting
eggs to forward and back parts of crest, the female is bent as a ring. Now
within one month it will have to bear eggs on its own body.
Eggs are not simply pasted to skin – otherwise they will turn away, when the
female gets into close shelter. Eggs are partly immersed in the top layer of
skin as it takes place at frogs of genus Pipa. Eggs contain a plenty of nutrients,
and the embryo can continue development successfully without additional nutrition
from the part of parent organism. Slime of female forms a protective cover
on surface of eggs, protecting them from drying. Blood vessels penetrate into
skin of crest of the female and closely adjoin to egg environments. But blood
of the female supplies posterity only with additional oxygen and water, but
not with nutrients. Therefore in any case embryos develop only up to the stage
of aquatic larva, because in egg there will be no nutrients for full metamorphosis
of embryos. When they develop sufficiently, their secretions to the blood of
the female stimulate her to go down in water and to let larvae out. Shortly
before hatching yolk sac completely resolves and tiny thin-bodied larva is
turned in ball inside the egg. When the female is in water, the top layer of
her skin swells, and the protective cover of stiffened slime is dissolved.
Feeling it, larvae leave egg environments and hide at the bottom among plants
and silt. They feed on tiny worms and larvae of insects and breathe with the
help of plumose external gills. The body of the larva of first year of life
is completely transparent; it helps it to be hidden from enemies – at danger
the larva simply rushes to the bottom and is dug in silt. At the second year
of life at them paws grow and gills resolve completely. At the age of four
years these amphibians are capable to breed.
Zme’eved, the forum member, has helped to specify features of biology of this species.
Red
unnewt (Contraphthalmus rufus)
Order: Tailed amphibians (Caudata)
Family: Salamanders (Salamandridae)
Habitat: deciduous, mixed and coniferous forests of eastern North America.

Picture by Alexander Smyslov
Amphibians are not only the oldest quadrupedal vertebrates,
but also the most “conservative” ones: unlike reptiles, birds and mammals, they
remained connected with water at least in the early stages of life, and some
species for their entire lives.
However, in Neocene, this situation has changed. Back in the Holocene, some
species of American newts and salamanders, in particular, the eastern newt (Notophthalmus
viridiscens), had a special juvenile form adapted to life on land. Adult newts
of this species lived mainly in water. At the turn of the Holocene and Neocene,
several species descended from the eastern newt; one of them is almost an analogue
of the ancestral species, the second one is a completely neotenic species that
lives only in water, and the third one almost does not need water for its life
at all.
The red unnewt resembles outwardly the juvenile terrestrial form of the ancestral
species –it has actually lost the adult aquatic stage, having passed to life
on land. It is a fairly large amphibian, reaching up to 20 cm in length along
with the tail. Its skin is red or red-brown, with darker spots of irregular
shape on the sides. Unlike other amphibians, the skin of the red unnewt is poorly
permeable to water, and it is one of the reasons that it is able to live on
land all the time, unlike its ancestor and other amphibians. The second reason
is hidden in the peculiarities of its metamorphosis.
In amphibians, the larval stage lives in water: their offspring usually have
gills and initially lack the limbs. The situation is different for the red unnewt:
its legs are well-developed already at birth, but the gills are small, and disappear
within a maximum period of a month. The adult land-dwelling form does not differ
much in appearance from a lizard – except the absence of scales, claws and an
ear openings.
The red unnewt spends most of its life on the ground and under ground: its limbs
are well developed, strong and equipped with special horny calluses that make
them good tools for ground digging. As a rule, this amphibian spends its daylight
hours in its own shelter, and at night wanders through the forest and catches
its prey – mainly invertebrates, as well as small vertebrates. This animal approaches
the water only during the breeding season.
The red unnewt is a viviparous species. Its breeding season takes place in the
first half of spring, and is not especially graceful. The male’s courtship for
the female is not much different from the beating that males of this species
give each other when establish a relationship of dominance. The male lays the
spermatophore on the broad leaves of plants or on smooth stones, and literally
forces the female to take it with her cloaca. At the end of summer, female of
this species lays one or two well-developed larvae in shallow water bodies,
and they turn very soon into a land-dwelling morph. The larva has four legs,
short brush-like outer gills and greenish coloration with black speckles on
the back and sides. In autumn, they leave the reservoirs and pass to terrestrial
lifestyle.
The red unnewt has few enemies – mostly various birds, including owls, as well
as omnivorous terrestrial mammals. This amphibian species is somewhat poisonous
and has an unpleasant taste. Its poison is not fatal, but has a strong irritating
effect on the mucous membranes. In addition, the red unnewt has a rather powerful
bite, so most small predators prefer avoiding the contact with this amphibian.
The red unnewt hibernates in burrows in the north of its range, often in groups,
which is an exception to the rules for this solitary and somewhat aggressive
amphibian, and in the south, where winters are not very cold or snowy, it sometimes
does not hibernate at all, remaining active all year round.
Red unnewt can live for several decades.
This amphibian species was discovered by Bhut, the forum member.
Amaru
(Herpetotriturus amaru)
Order: Tailed amphibians (Caudata)
Family: Salamanders (Salamandridae)
Habitat: southeastern North America, stagnant freshwater reservoirs – lakes
and marshes.
Like many other animal groups, amphibians have suffered from habitat destruction
caused by humans. Some of their species became extinct due to hunting for food
or captivity, and also became victims of diseases imported from other continents.
The ice age of the beginning of the Neocene caused the extinction of a number
of local and especially thermophilic forms, and also led to the disappearance
of residual populations of a number of species. However, in the early Neocene,
the few surviving forms gave a wide range of descendants differing in ecology
and habitat requirements. One mainstream direction of amphibian evolution was
neoteny, and new neotenic and legless forms also evolved in nature, among which
there is amaru, the large descendant of the eastern newt (Notophthalmus viridiscens)
– the common species of the human epoch.
This amphibian species has a significant number of juvenile traits that persist
until the end of life. The body length of an adult is up to 80-90 cm, of which
the laterally compressed tail bordered by a fin plica makes about a half. This
animal has an elongated head with well-developed eyes and a wide-opening mouth.
The skin color is greenish-brown, lighter on the belly. The limbs are very short
and have three digits; the animal rarely goes out on land and moves mainly by
crawling, slightly clinging with its limbs. Under water, the front legs help
to push prey into the mouth, and the hind legs are used in the breeding ritual.
Amaru lives mainly in water and retains external gills, although it has also
poorly developed lungs; the animal is able to breathe through the skin. If the
pond where the amphibian lives turns shallow or filled with rotting plants,
amaru emerges for air every 7 minutes, inhaling it with a characteristic squeaking.
In clean water, the animal pops up once every 25 minutes. When the reservoir
dries up, the animal can crawl to the next one by land, guided by the smell
of water. This species is exclusively zoophagous: the amaru feeds on arthropods
– both aquatic and fallen into the water, mollusks, fish fry, adult small fish
and tadpoles. It can also eat its own young, froglets and fish and amphibian
eggs.
Breeding takes place from December to April. At this time, the fin plica expands
in males and a bright orange color appears on the throat and chest, and the
head becomes almost black. Males chase females ready to spawning, being guided
on their scent. Having found the female, the male wraps his body around her,
holding her by the hind legs, and the animals bring their cloacae closer. At
this point, the spermatophore is transferred. After mating, the male holds the
female for some more hours (usually all night until morning) and releases her
when she stops emitting the smell of readiness for reproduction. In February-June,
the female lays 120-130 large eggs in secluded moist places, wrapping them with
her body. Crayfish burrows or old burrows of aquatic mammals in their upper
part, located above the water level, are often used as places for egg laying.
At this time, the female rarely leaves the eggs, does not eat and loses a lot
of her weight. The development of this species is slow: incubation lasts 3 months,
after which the female leaves the juveniles. At hatching, larvae have a length
of about 6 cm and well-developed forelegs. In the early stages of development,
they feed on daphnia and mosquito larvae, gradually switching to larger prey.
Sexual maturity in amaru comes at the age of 4 years with a length of 65 cm.
Life expectancy is up to 28-30 years.
This species of amphibians was discovered by Nick, the forum member.
Monsternewt
(Siluroherpeton cryophilus)
Order: Tailed amphibians (Caudata)
Family: Swampers (Pseudoandriidae)
Habitat: Northern Europe up to Ural, Scandinavia.

Picture by Alexander Smyslov
After human extinction till millions years the Earth had gradually
cleared of various kinds of pollution left by this species. Polluting and poisonous
substances had been gradually decomposed as a result of activity of bacteria
and other organisms. Chemical elements making them had included natural geochemical
cycles, and in Neocene epoch the planet had returned to protogenic cleanliness
in ecosystems.
In human epoch the species sensitive to pollution of environment died out or
receded from places where people conducted economic activities. Among them there
were especially many species of fishes and amphibious. Human extinction had
permitted to evolve to new species of these animals, exacting to cleanliness
of inhabitancy.
The territory of Europe had strongly suffered from human activity, and a number
of species of amphibians lived there became rarity. The glaciers came from the
north, had almost completely destroyed marks of presence of people in this part
of the world. When people had died out, amphibians had returned to themselves
primordial habitats and evolved to strange and unusual species.
In head waters of rivers of Europe running into Arctic Ocean at the north, and
into Fourseas at the south, the largest European species of amphibians, the
monsternewt, is found. It is the close relative of Siberian gluttonous
swamper (Pseudoandrias silurops) which is the largest species of amphibians
in Neocene world.
Monsternewt considerably concedes in size and weight to glutton swamper, but
is also a large amphibian. Body length of this amphibian is about one meter
including tail. At monsternewt there are large head, wide body, deep tail with
fin plica stretched up to middle of back, and strong paws without claws. This
amphibian has soft dark brown skin with small darker spots, lighter bottom side
of body. On stomach skin forms the set of longitudinal plicas taking part in
skin breath. Also at this animal branchiate apertures are advanced and there
are small external gills.
Monsternewt is inactive ambush predator and is not able to chase prey at long
distance. This amphibian waits for prey, having hidden among water plants, or
having slightly dug in silt. It has flat head with wide mouth and strong jaws.
Teeth of this animal are tiny; they serve only for keeping of rather small prey
– frogs, crayfishes, fish, mammals and small aquatic birds. Eyes at this amphibian
are extremely small, located on edges of head near corners of mouth. Bodies
of lateral line play the vital role in life of monsternewt – they form a dense
system of sensitive channels on head of animal, and stretch as double line along
each side. With their help animal can define the location of prey even in complete
darkness. These animals well feel like in karstic caves.
Monsternewt lives in cold water containing a plenty of oxygen and because of
it is intolerant to overheat of water. The greatest number of these amphibians
lives in rivers and lakes at the north of Eurasia. The period of maximal activity
of monsternewt falls to the time of high water. At this time amphibians settle
to new habitats, especially if favorable habitats are overpopulated. In high
water young individuals frequently meet in forest, and appear in traps – in
small isolated ponds, whence they can not get out. This amphibian can not move
overland – on air wet skin of animal dries up quickly, and monsternewt chokes.
In summer monsternewt is inactive and hides in deep cold whirlpools where underwater
springs gush out. In winter this amphibian creeps on the bottom under ice and
hunts on fishes wintering in underwater holes. Animals from cold river heads
and karstic reservoirs keep activity even in the middle of summer. In winter
at complete freezing of shallow-water reservoir monsternewt digs in silt and
falls into catalepsy. When ice thaws, it continues habitual life.
In early spring monsternewts spawn. Spawning begins when rivers free from ice
and the time of high water begins. At the male during the spawning time colouring
becomes much brighter. It has bright orange-red color of stomach. Courtship
dress of the male also includes high skin crest on back and rounded “horns”
above eyes. During the spawning males “butt” for females, pushing away each
other by heads. Also with the help of “horns” male pushes female to spawning
place – to thickets of underwater vegetation. Clutch of monsternewt is rather
large – it numbers over 10 thousand eggs strongly swelling in water and covering
with the common slime cover. The incubation lasts till about one month. From
eggs long-bodied legless larvae with well advanced plumose gills hatch. They
lead predatory habit of life, and for them cannibalism is very characteristic.
As against to adult monsternewts, larvae well enough exist at higher temperature.
Till the summer they manage to grow up to 10 cm, and legs develop at them. Since
the second year of life young animals gradually start to avoid the high temperature,
and pass to autumnal, winter and spring activity.
The sexual maturity at monsternewt comes at fifth year of life. Life expectancy
of this amphibian is not less than hundred years.
Ahtosuchus
(Ahtosuchus rex)
Order: Tailed amphibians (Caudata)
Family: Swampers (Pseudoandriidae)
Habitat: cold freshwater reservoirs of Northern Europe.
In the Neocene epoch, the characteristic group of tailed amphibians of Eurasia
are the swampers – large descendants of newts inhabiting fresh water bodies
of the cold regions of the continent. The rivers of the Kola Peninsula are inhabited
by ahtosuchus, a very large representative of this family. The maximum size
of this amphibian is up to 2.5 m, but only some individuals in the population
reach this size. Usually animals of this species are slightly smaller. Outwardly,
it looks like a giant newt: an elongated slender body, a rounded, slightly elongated
head tapering to the muzzle, large eyes, a fin plica at the tip of the tail
(flattened from the sides) and rather long and functional limbs on which it
can crawl on land. The body color is brown, suitable for camouflage at the bottom
of the reservoir. The belly is colored lighter and brighter – numerous red spots
on a yellowish-white background. The lower jaw and throat are yellow. The outer
gills are small and feather-like; there are 3 pairs of them.
Ahtosuchus usually swims slowly in the slow-flowing rivers of Northern Europe
(the species is named after Ahto, the Finnish sea god), preferably deeper and
with a large layer of plant rests on the bottom. This species is sympathetic
to the monsternewt (Siluroherpeton cryophilus)
and differs from it in lifestyle features. It is most active in the summer,
when the monsternewt turns numble, and feeds on larger prey. This species is
more thermophilic, and shows the peak activity in summer, when the water in
rivers and lakes warms up. At this time, ahtosuchus can be seen lying in shallow
water, on the large stone or log jutting out from the water, or on land at the
water’s edge. The disturbed animal quickly “falls” into the water, pushing its
tail off the ground.
This species is exclusively zoophagous and can hunt in various ways. The first
way is to swim slowly over the bottom and literally “suck” the benthic prey
like small fish, insect larvae, crustaceans, snails and frogs. This is how young
individuals usually hunt. The second way is to hide in the plant debris at the
bottom, slightly burrowing itself into the bottom debris, open the mouth and
wait until a careless fish or frog swims closer, and then catch it. An adult
animal is a well-swimming and strong predator capable of chasing pelagic fish.
Also, it is not difficult for it to grab birds floating on the water, pick up
the corpses of small land animals or eat the bodies of large ones decomposing
in the water, tearing off pieces of meat. In addition, this amphibian is also
a real nightmare for land animals coming to drink. It eats not only rodents
and other small mammals, but also harelope
cubs. Ahtosuchus seizes the prey by the throat or head, and, writhing with its
whole body like a snake, drags it to the depths. At a depth, ahtosuchus simply
waits until the prey suffocates, and then swallows it whole.
Fish, other amphibians, birds and mammals pose a danger to the juveniles, and
the adult ahtosuchus has almost no enemies. The monsternewt can catch the young
of this amphibian, but ahtosuchus also poses a danger to its juveniles, hunting
in places where young individuals spend the summer. This amphibian winters at
the bottom of the river, burrowing into the silt, and very often lies in the
pools. The favorite wintering places of this species are located in pits under
waterfalls.
In the spring, the awakened amphibians begin courtship displays: males show
to each other bright body parts, bump with their sides and bite each other.
According to the results of the tournaments, the female chooses the best partner,
that delivers her his spermatophore and stays next to her, waiting for the egg
laying, and after laying eggs leaves them in the care of the male. Ahtosuchus
male guards about a hundred eggs of 5 cm in diameter (after laying, the eggs
swell considerably) for about 2.5 weeks. An interesting feature is that there
is a smaller amount of eggs in the clutch compared to its Holocene ancestor,
but the male takes an active part in caring for the offspring and eats any animal
that can harm the offspring. After the 7 cm long larvae hatch, the male continues
to look after them as carefully as possible for about two more weeks. Despite
this, a part of the offspring anyway falls prey of small predators. At the beginning
their of life, the achtosuchus larvae feed on small worms, insects, snails and
crustaceans, switching later to feeding on fish fry, tadpoles, small frogs.
A significant part of the offspring dies in the first two years of life.
Individual development is very slow. Sexual maturity comes only at 8-10 years
of age. Ahtosuchus has a significant longevity: it can live up to 200 years.
This species of amphibians was discovered by Mamont, the forum member.
Catfish-faced
mole salamander (Silurotriton rubroviridis)
Order: Tailed amphibians (Caudata)
Family: Mole salamanders (Ambystomatidae)
Habitat: rivers and lakes of the temperate zone of North America.
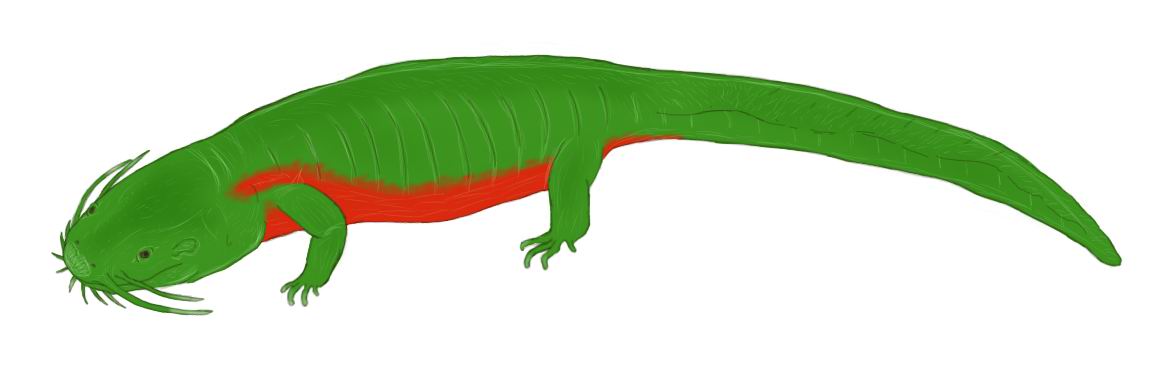
Picture by FanboyPhilosopher
In the human era, amphibians suffered significantly as a result
of habitat destruction, environmental pollution and diseases, the pathogens
of which were spread through human activity. The early Neocene was a time of
active speciation of surviving forms that occupied ecological niches emptied
at the end of the human epoch. Some of the new forms survived due to developing
of new strategies for the struggle for existence.
In North America, some of the numerous tailed amphibians began using venom not
only as a defensive, but also as an offensive weapon. One of their typical representatives
is catfish-faced mole salamander, widespread in the east of the continent.
It is a fairly large poisonous ambystoma: the body length is up to 45 cm, along
with a tail that is about half the length of an adult. Its overall appearance
is typical of mole salamanders: it is an animal similar to a robust lizard with
short strong limbs. The animal’s head is flattened, of rounded outlines. Small
eyes are located on the edges of the head; the animal’s vision is poor. The
quality of eyesight is largely compensated by a well-developed chemical sense.
On the upper jaw of this amphibian, several pairs of elongated wattles grow,
similar to the wattles of catfish and being about a half the animal’s head length,
with fleshy bases. The surface of these wattles is covered with numerous sensitive
cells responsible for tactile sensations and chemical sense. There are also
several short wattles (up to 4 pairs) on the lower jaw. At the end of the muzzle,
there is a corneous growth – an adaptation for digging the ground.
The body of the catfish-faced mole salamander is colored green with a red stripe
along the body in the area of the shoulder blades and with a red belly. In case
of danger, the animal bends with its belly up and curls like a ring, displaying
the bright coloration of its belly. In addition, such a pose makes it difficult
for snakes to attack this amphibian. The bright coloration is aposematic; the
animal is armed with two types of venom glands. The glands on the skin synthesize
bufotenin, which is abundantly released in case of danger; in the mouth there
are modified salivary glands located on the upper jaw, which synthesize a neuroparalytic
venom of protein nature for hunting fish. The teeth of this amphibian are very
powerful; the venom is released during the bite. Biting its prey, the animal
“chews” it with its jaws for some time, facilitating the penetration of venom
into the wound. A fish of the size equal with this amphibian dies in 5-10 minutes,
and catfish-faced mole salamander easily finds it by smell.
The animal spends a lot of time in the water, although it has no gills in the
adult stage (only the spiracle hole does not close). Catfish-faced mole salamander
can settle overland and, if necessary, crosses the distances of up to several
kilometers to a habitable reservoir. When this amphibian is on land for a long
time, the wattles shrink significantly and look like rounded growths, and the
skin is covered with a layer of viscous slime that helps to preserve water in
the body.
This species is a predator and feeds mainly on fish and amphibians. Less often,
catfish-faced mole salamander eats crayfish, preferring small ones that are
still soft after molting. It also eats larvae of aquatic insects.
Reproduction in this species takes place in water, as in most tailed amphibians.
Spawning takes place twice a year – in spring and autumn. Usually the autumn
clutch is smaller than the spring one. By the time of mating games, a deep skin
flap grows on the male’s back, which turns completely red, and numerous black
spots appear on his body. The female is paler, fuller and larger than the male.
It spawns a small amount of large, up to 1 cm in diameter, eggs in thickets
of small-leaved aquatic plants. The male protects the spawned eggs, and later
the larvae until the age of one month. At this time, he attacks even animals
larger than his size and bites them.
Hibernation in this species takes place on land, in a hole dug by the amphibian
by its own. In the south of its range, the catfish-faced mole salamander waits
through the dry summer in its burrow. This amphibian grows quite slowly: sexual
maturity comes at the age of 3 years, and active growth continues until the
age of 10, after which it slows down significantly. Life expectancy can reach
40 years.
This amphibian species was discovered by Dmitry Korshin, the forum member.
Pouched
mole salamander (Fissurotriton paterfamilias)
Order: Tailed amphibians (Caudata)
Family: Mole salamanders (Ambystomatidae)
Habitat: northern part of North America, swampy coniferous forests.
North America of the human epoch was remarkable by a great species diversity
of tailed amphibians. Human activity led to a decrease in their species diversity,
but later, as suitable habitats had restored, this damage was compensated by
their rapid speciation. Among the Neocene tailed amphibians, species have emerged
that use new strategies for the struggle for existence – in particular, new
forms of caring for offspring.
Tailed amphibians are more conservative in terms of ways of parental care compared
to tailless amphibians. But in the Neocene, a species appeared among them, partly
copying the way of parental care characteristic of marsupial frogs (genus Gastrotheca).
This animal is a pouched mole salamander, a short-tailed and stocky salamander
living in the reservoirs of taiga zone of North America. This animal reaches
a length of about 30 cm along with the tail. It leads a semi-terrestrial lifestyle
and often crawls from one reservoir to another. The pouched mole salamander
has strong limbs that allow the animal digging the layer of leaf litter in search
of shelter and food. The head of this amphibian is broad and short, with strong
jaws and areas of keratinized skin on the front edge of the jaws.
The skin coloration of the pouched mole salamander is bright and contrasting.
The limbs, sides and base of the tail are colored brown with black speckles,
and the head, back and upper side of the tail are very bright orange, with a
reddish tinge in some individuals. In males, two pronounced skin folds stretch
parallel to the spine. When attacked by a predator, this amphibian rises on
straightened limbs and begins making sharp movements with its tail and head
from side to side. It is a warning display: the pouched mole salamander is armed
with skin glands that are located on the shoulders and back of the head and
spray poisonous secretions containing bufotenin over a short distance. In addition,
in the mouth of this animal there are venomous teeth in the upper jaw, equipped
with grooves, along which during the bite the venom of a protein nature flows
down.
Like all other amphibians, pouched mole salamander is a predator. It hunts tadpoles,
frogs and fry of various fish, and on land, it eats insects and small amphibians.
The tongue is short, so the animal grabs the prey with its mouth and delivers
a venomous bite.
This species lives in a seasonal climate with a cold winter, so the pouched
mole salamander hibernates in self-dug burrows or in rodent burrows. After coming
out from hibernation, the animals migrate to reservoirs, where reproduction
takes place then. Males get bright coloration: brown color is replaced by orange,
and black spots increase in size. Females, on the contrary, turn very dark,
and the orange area forms a snake-like pattern. In the south of the range, breeding
begins around the middle of spring, and in the north in early summer, when the
water warms up well.
During the courtship ritual, the male lays the spermatophore on the plants,
and the female takes it with the edges of the cloaca; so the internal fertilization
takes place. The male holds the female with his limbs, waiting for the eggs
to develop. At this time, the skin ridges on his back swell and form two longitudinal
skin folds stretching from the shoulders to the base of the tail. The cavity
formed by them is lined with spongy skin, into which blood vessels penetrate.
The female lays up to 100-120 large eggs in a prepared brooding pouch, and then
the male bears eggs on his own back for several weeks. Blood vessels supply
developing embryos with nutrition and oxygen. Due to it, the male’s pouch is
left by already well-developed larvae having buds of the forelimbs. Bearing
eggs, the male can transfer them from one reservoir to another. When the offspring
leaves the pouch, the male cleans with his paws and eats the detached epithelium,
and the size of the folds decreases back. At this time, he “shepherds” the larvae
for several days, protecting them from fish and its own relatives.
In the northern part of the range, larvae develop for two years, in the south
metamorphosis occurs in mid-autumn and young animals run to hibernation. Life
expectancy reaches 20 years.
This species of amphibians was discovered by Dmitry Korshin, the forum member.
Burrowing
mole salamander (Ambystoma sauris)
Order: Tailed amphibians (Caudata)
Family: Mole salamanders (Ambystomatidae)
Habitat: deciduous, mixed and coniferous forests of eastern and northeastern
North America.
Mole salamanders form one of the most widespread kinds of tailed amphibians
in North America, and have a typical set of features of salamanders: these are
lizard-like creatures with slimy smooth and sometimes spotty skin. These amphibians
can often be poisonous, but not deadly.
In the Neocene, mole salamanders survived and remained very diverse. They have
achieved distinct species diversity in western North America, where a stable
humid climate contributes to their prosperity. However, there are also interesting
species in the east of the continent. One their species is the burrowing mole
salamander. A descendant of the Holocene yellow-spotted salamander (Ambystoma
maculatum), it partly resembles a larger representative of the ancestral species
– the average length of this amphibian reaches 20-25 cm. However, it has no
spots – almost the entire body is dark brown, almost black, and only on the
shoulders and neck there is a bright yellow spot, as if separating the head
from the rest of the body. The physique is more robust compared to the ancestor;
the cervical waist is almost not pronounced, the head is flattened, and eyes
are small.
The burrowing mole salamander has well-developed, short and muscular limbs,
since this amphibian spends a considerable part of its time under ground, where
it digs tunnels to find its prey – invertebrates, mainly insects and worms –
and also to escape from the sun. It is a good digger in its size class, and
lives in places where competing burrowing mammals (moles, rodents, etc.) are
not usually found – in moist, sometimes even swampy soil along the shores of
various reservoirs and in humid forests. Each individual marks the tunnels with
slime with its own smell, and does not tolerate the presence of relatives there.
Sometimes adult animals attack young animals that happen to be in their burrows.
Adults of this species breathe with their lungs and skin, so they regularly
come to the surface to breathe air and clean their skin – they have long enough
and well-developed legs to do it. At this time, the animal is most often attacked
by predators – mainly shorebirds and snakes. However, it is not often hunted
specifically: ambistoma digger is venomous, although not fatal, but its slime
has an irritating effect, and its taste is still unpleasant. Eggs and larvae
lack such protection and are readily devoured by various predators. This species
also suffers from various parasites.
The burrowing mole salamander spawns its eggs into the water in the spring:
it has relatively small clutches, numbering up to 70-200 or more eggs, and they
float across the shallow water like sticky tapes. Males actively fight for females,
trying to overcome each other in the fight, and sometimes bite off the opponent’s
leg or part of the tail. The lost body parts regenerate soon, but a male crippled
in a fight, as a rule, drops out of the battles for the female. The male actively
swims around the female, pushing her and grabbing her with his limbs, and forces
her to take with her cloaca the sprematophore left on plants. Fertilization
is internal. Incubation of eggs lasts up to 3-4 weeks. The larvae of this species
resemble the larvae of other amphibians, have external gills and can only live
in water. However, by autumn they already complete metamorphosis and leave the
water for life on land, where they immediately begin to build tunnels, escaping
from predators and dry air on the ground surface. Sexual maturity comes at the
age of 2 years; the average life expectancy of the burrowing mole salamander
is up to 20 years.
This amphibian species was discovered by Bhut, the forum member.
Eel-like
salamander (Neoplethodon anguiliformis)
Order: Tailed amphibians (Caudata)
Family: Lungless salamanders (Plethodontidae)
Habitat: shallow waters of Mishe-Nama Lake, other large bodies of water.
From the point of view of classification, amphibians, including tailed ones
– salamanders and newts – belong to quadrupedal vertebrates (tetrapods). Nevertheless,
a fairly large number of amphibians (not related to tailless ones) have not
four legs, but only two, or, like caecilians, lack them completely. This was
also the case at the very beginning of amphibian evolution, in the Carboniferous
period, when legless amphibians existed, although they are not close relatives
of modern amphibians. In the Neocene, amphibians also lost their legs in due
course of evolution, and some species of such appearance live in North America,
where the diversity of tailed amphibians is especially great.
Lungless salamanders were one of the most successful families of tailed amphibians,
especially in North America, and in the Neocene they managed to occupy new ecological
niches and give rise to new species, sometimes quite bizarre in appearance.
A number of species of this family live in the cool water of rivers and lakes
in the north of the continent, and one of them is the eel-like salamander.
It is an amphibian with a thin body, up to 20-25 cm long, of reddish-brown color
with a lighter belly, covered with darker specks and spots. The shape of its
body resembles it of an an eel, because its paws are reduced completely and
even in adulthood there is no trace of them left. In addition, the eel-like
salamander lacks gills – they are replaced by numerous skin folds, especially
on the back and belly. They are abundantly penetrated with blood vessels and
provide the animal’s body with oxygen exclusively due to skin respiration. The
body of the eel-like salamander is also somewhat compressed from the sides,
and it gives its body a leaf-like appearance. It helps this animal to hide in
the thickets of underwater plants, where it usually stays, avoiding its main
enemies – predatory fish and large tailed amphibians. This amphibian keeps almost
exclusively in the water, staying in coastal thickets during breeding.
The eel-like salamander is also a predator; it feeds on various aquatic invertebrates
– insects, crustaceans, snails, etc. It has well-developed bulging eyes, but
it also uses sense of smell and touch, especially in search of prey and females.
The breeding season of the eel-like salamander is at the end of September and
October: before wintering, males search by smell for females ready to mate,
fertilize them and no longer care about their offspring: this salamander species
is an exclusively solitary animal. The only exception is wintering taking place
in non-freezing springs and at the river bottom.
In April, after the end of wintering, females of this species lay about 20 large
eggs in secluded places among coastal vegetation – they are large, opaque and
cream-colored. The female protects the clutch from predators and infection by
wrapping her body around it and secreting bactericidal slime.
This species does not care about hatched offspring; young salamanders are independent
from the moment of hatching and look like a reduced copy of their parent. Having
hatched out of the eggs, they immediately leave their native places and look
for a place for their own housing. Sexual maturity comes at 2 years; the average
lifespan of the eel-like salamander is 11-16 years.
This amphibian species was discovered by Bhut, the forum member.
Fanged
salamander (Neoplethodon odontata)
Order: Tailed amphibians (Caudata)
Family: Lungless salamanders (Plethodontidae)
Habitat: shallow waters of Mishe-Nama Lake, other large freshwater bodies.
Tailed amphibians are among the conservative inhabitants of the Earth, including
in terms of realized adaptation opportunities. Unlike tailless amphibians –
frogs, toads and others – which occupied a wide variety of habitats, from the
desert to the rainforest, numerous tailed amphibians
remained inhabitants of the water – usually slow-flowing and even stagnant.
Some of them managed to develop life on land to some extent, while others returned
to the habitat of their ancestors and, due to the neoteny phenomenon, turned
to completely underwater inhabitants. The fanged salamander is one of such animals.
Despite the passing to the aquatic lifestyle, it looks quite unusual – it lacks
external gills; instead, the fanged salamander developed numerous skin folds
on the body (mainly on the back, boy and tail sides), which is also characteristic
of some tailless amphibians of the Holocene and Neocene. It is an amphibian
of small size – 10-15 cm in length, but due to its skin texture it looks very
exotic. The coloration of this species is not bright – dark gray with a greenish
or brownish tinge; when the animal freezes at the bottom, it is very difficult
to notice it.
A special feature of the fanged salamander is a pair of “fangs”: the bony processes
of the skull protruding at the edges of the upper jaw, which are used to break
the shells of its prey – gastropods, less often bivalves. This amphibian feeds
almost exclusively on them, and almost does not eat other prey. Because of this
feeding specialization, the fanged salamander has become a very slow animal:
being an exclusively aquatic form, it actually does not swim, but only walks
on the bottom. It has well-developed limbs, and the fanged salamander is able
to dig the ground well: it is able not only to dig its prey out of the sandy
or muddy bottom, but also to dig itself in case of an attack by any predator
– fish, diving bird or the predatory amphibian like the lionhead
waterdog.
The breeding season of the fanged salamander falls on September and October.
At this time, the females emit a smell that attracts males – the fanged salamander
has a poor vision, but a keen sense of smell and touch, and also feels subtly
the movements of the water around itself due to the numerous sensitive cells
in the skin. Males find females by scent and fertilize them via depositing of
spermatophore directly into the female’s cloaca. The development of eggs continues
until spring, and in early April, having fattened up after wintering, the female
spawns a bunch of large eggs (usually from 10 to 15 in number), connected by
slimy threads in a row. After 3 weeks, the larvae hatch from them. During incubation,
the female protects them from predators and diseases by wiping them with her
slime. Young animals look like a miniature copy of their parents, and immediately
pass to an independent life, hunting small crustaceans like Daphnia. They become
sexually mature at the age of one year, and at the same time switch completely
to a diet characteristic of this species.
Mishe-Nama Lake is a body of water that never freezes completely, and significant
areas of the bottom at depth remain free of ice. Therefore, the fanged salamander
never go into a true hibernation and is active all year round, looking for its
prey – snails and other mollusks. Her average life expectancy is 11 years or
more.
This amphibian species was discovered by Bhut, the forum member.
Lionhead
waterdog (Necturus leonatus)
Order: Tailed amphibians (Caudata)
Family: Mudpuppies (Proteidae)
Habitat: North America, Mishe-Nama Lake.
Neocene is a time of great changes in the history of the Earth. Many features
of the landscape of various continents have changed significantly, and these
changes are clearly visible on the territory of North America.
The natural zones of North America are torn apart by a vast expanse of prairies
that lie in the center of the continent and turn into deserts in the south.
To the west and east of them there are wetter lands overgrown with forests and
inhabited by species that often had a common ancestor that existed on Earth
in the human era.
The northeast of North America is distinguished from other regions of the continent
by a unique natural complex – Mishe-Nama Lake, which has its own unique ecosystem,
including numerous endemic animals. It is a home to species of clearly marine
origin: local gannetwhales
and cartilaginous fishes – giant stingrays. However,
side by side with them in the lake true freshwater animals live, which got into
Mishe-Nama Lake from the south, populating it as the glacier retreated and the
upper water layers desalinated. One of these animals is the lionhead waterdog.
The lionhead waterdog is a descendant of the common mudpuppy (Necturus maculosus).
It is one of the largest amphibians in North America, reaching an average of
50-60 cm in length, which makes it a Neocene analogue of the Holocene era hellbender.
Unlike them, the lionhead waterdog is quite an active creature. It has a strong,
laterally compressed tail, which is its main propeller in the water column,
and well-developed (though mainly in adults) limbs used by this salamander to
move through shallow water and along the bottom. The lionhead waterdog got its
name because of the well-developed external gills, vaguely resembling a lion’s
mane. This amphibian is an exclusively aquatic animal and usually avoids both
shallow and deep waters. It usually stays at a depth of 2 to 20 meters.
Unlike some other Neocene amphibians, the lionhead waterdog has not lost pigmentation:
on the contrary, its skin is dark- or olive-brown with darker spots. This species
is able to change the color of the skin partially, making it lighter or darker
depending on the transparency of the surrounding water. Sometimes there are
albinos among them, but they usually die at the larval stage. Direct sunlight
frights these animals.
The lionhead waterdog breathes primarily through its gills: they are well developed
even in adult amphibians and allow them absorbing oxygen from the water. But
lionhead waterdogs also absorb a certain amount of the oxygen through the skin.
If the weather is hot and the content of oxygen in the water decreases, they
are able to swim up to the surface and breathe through their lungs, which they
still have. But their lungs mainly represent an analogue of a swimming bladder,
helping the animal to keep in the water column.
Lionhead waterdogs are voracious predators, and feed on any small animals that
they are able to prey – small fish, invertebrates, other amphibians, etc. They
do not disdain carrion, as well as the remains of the prey of other, larger
predators like gannetwhales and other birds. They are hunted by gannetwhales
and large fish, as well as aquatic
mammals, but the lionhead waterdog is a rather cautious animal. Its eyesight
is not very good, but the lateral line is well developed, which helps it to
feel the water movements, and there is also a keen sense of smell: at the slightest
sign of danger, the lionhead waterdog swims to the depths, hides under driftwood
or buries itself in silt.
The breeding period of the lionhead waterdog takes place in the autumn, before
wintering. Like most tailed amphibians, these animals are not remarkable in
complexity of behavior and are solitary ones, but due to the relative abundance
of food and space for life in Mishe-Nama Lake, lionhead waterdogs treat each
other neutrally and often live side-by-side, although they hunt in different
places and can compete for shelters. It happens, that the same male mates from
year to year with the same female, but this is not a permanent breeding pair
like in more developed vertebrates, and if one of the amphibians dies, the second
one easily forms a pair with another individual of the appropriate sex.
Lionhead waterdogs lay eggs in the first half of spring. Their eggs are quite
large compared to ancestral species (up to 30 mm in diameter), and their number
is usually small: 5-6 ones only. Females guard their offspring: they lay eggs
in shallow burrows at the lake bottom and protect them from enemies, and create
the constant water flow around the eggs. Incubating the eggs, the female creates
a current of water with her tail and turns eggs with her hind legs. All this
time she does not eat anything, but because of the slow metabolism, it does
not harm her body. Large larvae hatch by summer and quickly leave the burrow,
since a hungry female can easily attack her own offspring. Larvae mature slowly,
reaching sexual maturity by the fourth or fifth year of life.
The average life expectancy of a lionhead waterdog is several decades.
This amphibian species was discovered by Bhut, the forum member.
Poisonous acrobatrachus (Acrobatrachus toxicus)
Order: Tailless amphibians (Anura)
Family: Fire-bellied toads (Bombinatoridae)
Habitat: Europe, deciduous forests and bushes of various types.

Picture by Alexander Smyslov
Warming of the climate in Neocene has caused change of vegetation
of the Earth and redistribution of landscapes. At this time the territory of
Europe had
considerably extended due to deviation of North Sea – instead of it the extensive
territory covered with wetlands of various degree of salinity had appeared.
The cold-loving vegetation is kept only in mountains, and in plains deciduous
forests dominate. Warming has caused increase of variety of amphibians and
reptiles in European forests. Evolving actively, they had developed new tactics
and strategies of protection against enemies. New species of these animals
in the majority weren’t descendants of heat-loving southern forms – the mountain
ridge of the Alpes has risen across the way of their moving as an insuperable
barrier. Therefore the majority of these animals represents the descendants
of local forms managed to go through ice age.
In deciduous forests of Europe, to the north of the Alpes, in river valleys
one representative of new amphibians of Europe lives; it is a tiny frog covered
with spots of dark green and orange-red color. The length of its body does
not exceed 3 – 4 cm. Appearance of this animal is characteristic for frogs,
but forepaws are strong, and rear legs are rather short, with tenacious toes.
This frog lives on trunks and branches of plants. Being disturbed, it seizes
bark or branch by forepaws and is overturned upside down, displaying bright
colouring of belly where red color prevails, and green is present as separate
spots. This species is acrobatrachus, or the acrobat frog. Tiny frogs of
this species are very poisonous, and their skin is supplied with a whole
set of
glands secreting poisonous slime. It is the descendant of fire-bellied toads
(Bombina), at which bright colouring of belly was kept and along with enchancement
of poisonous properties was distributed to the whole body. If the predator
continues an attack, acrobatrachus inflates throat bag and utters grumbling
sounds, warning about its inedibility.
Acrobatrachuses secret very strong poison, and one eaten frog is enough for
bear-sized animal to feel the symptoms of poisoning. The smaller predators,
having tried such frog carelessly, do not perish, but feel stomach ache within
several days and can not hunt. Therefore acrobatrachuses have no need for
fast movement. Usually these frogs creep slowly on branches and leaves of
bushes
and undersized trees of underbrush, hunting insects and spiders. These frogs
have short muzzle and mobile prominent eyes, helping to estimate distance
up to the object or the next branch. The swimming membrane on hind legs is
reduced
and does not reach the middle of toes. Paws of these frogs are mobile and
strong; fingers automatically clench, having touched a substratum, and frog
can easily
hang on one paw. Tips of fingers and toes are covered from below with corneous
callouses that allows an animal to be fixed reliably on branches and tree
bark. Due to mobile limbs frog easily makes a daily toilet. In the morning
it emits
from glands a small amount of poisonous secret and smears it on body by paws.
Only one paw takes part in it, but frog greases skin by hinder legs as dexterously,
as by front ones. As a result the poisonous secret is distributed on the
whole body as an even layer.
Acrobatrachuses lead the solitary way of life. Each individual protects the
fodder site from contenders, uttering from time to time the squeaky sounds
similar to bark of tiny dog. If two frogs of this species meet each other,
between them the duel takes place, during which they inflate bodies and try
to push the contender away by strongly inflated throat. If the contender
does not recede, frogs struggle with each other, pushing each other by heads.
In
courtship season males bite each other, but these bites seldom result in
serious injures. Usually fight between males begins at the presence of the
female ready
to breeding. The winner male creeps on the female and she carries him on
her body to spawning place. At this time male drives contenders away from
the female,
uttering abrupt barking sounds and inflating throat.
Breeding of acrobatrachuses proceeds in the way traditional for frogs: adult
individuals gather in stagnant reservoirs suitable to spawning and almost
synchronously spawn a plenty of eggs. It is the most vulnerable stage of
life cycle of frogs:
eggs are not poisonous, and the plenty of them is devoured by fishes. Young
tadpoles are also edible, but they start to accumulate poison gradually.
Tadpoles just hatched from eggs are translucent with yellowish shade. They
hide in thickets
of plants, attaching to leaves with the help of sucker-like mouth. As they
grow, their colouring turns spotty. The tadpoles accumulated enough of poisonous
substances in skin cease to hide and in numerous schools feed on algal films
on shoaliness. They are afraid only of insects – water bugs and grubs of
water beetles simply pierce their skin and inject poison, dissolving their
tissues.
As tadpoles grow, in their colouring red and black spots appear, composing
at young frog the characteristic pattern.
Poisonous acrobatrachus is the most usual species of the genus in Europe.
It is settled across the whole southern part of Europe up to the Alpes, avoiding
only dry deforested areas. This frog is especially numerous in areas of deciduous
and river forests. Close species of these amphibians live in other places
of
Europe:

Picture by Alexander Smyslov
Orange acrobatrachus (Acrobatrachus citrinus) lives at the Pyrenean Ridge, isolated from an area of other species. It is the large species reaching the length of 6 cm. The basic colouring of body is orange and varies from dark yellow up to orange-red. On the top part of body there is a pattern of large black irregular-shaped spots. Protecting itself from the enemy, frog is not only overturned upside down, but also changes colouring: black color fades, and background turns more sated. This frog eats large ground insects and worms, and keeps mainly in underbrush among grassy vegetation.
Picture by Alexander Smyslov
Dwarf
acrobatrachus (Acrobatrachus pusillus) lives in southeast of Europe,
inhabiting subtropical forests of the Balkan. It keeps in valleys where formed
the numerous local forms connected to each other by transitive variants of colouring.
The length of this frog does not exceed 3 cm. In typical case colouring represents
a twisting pattern of spots of black and citreous colors. Among local forms
there are some variants of colouring:
1) Almost entirely black with separate yellow spots of rounded shape.
2) Longitudinal-striped with a little bit irregular pattern, usually on sides.
3) Black with cross-striped paws and yellow irregular-shaped spots.
4) Yellow with black irregular-shaped spots of various sizes.
This species of amphibians eats mainly tiny flying insects and lives in tree
crones. More often dwarf acrobatrachuses occupy hollow trees where rain water
accumulates. In these places separate individuals even breed.
Saharan
namomubatrachus, or Protopterus frog (Namomubatrachus protopteroides)
Order: Tailless amphibians (Anura)
Family: Spadefoot toads (Pelobatidae)
Habitat: Sahara, vicinities of temporary reservoirs.
Amphibians are associated with water for at least a small part of their life
cycle: most species absolutely need water during the breeding season. Therefore,
most of their species live near reservoirs, and competition between species
is very great. Some species have evolved along the way of developing of arid
habitats, but they are still associated with water during the breeding season.
In the process of evolution, some amphibian species have adapted to reproduction
in temporary reservoirs, and it imposes the significant restrictions on the
duration of their larval stage. Nevertheless, one of the species in the process
of evolution has adapted to circumvent this restriction in a very peculiar way.
The Saharan namomubatrachus lives in North Africa, inhabiting the outskirts
of the Saharan Nile basin. There are very few frog species found here, and this
species does not experience serious competition from them. Namomubatrachus tadpoles
develop in temporary reservoirs, feeding at first on algal films and silt. They
grow quickly and soon switch to a more nutritious diet – the tadpoles of other
frogs. Feeding on meat allows the namomubatrachus tadpoles growing rapidly.
By the time the puddle dries, the tadpoles of namomubatrachus resemble catfish
about 20 cm long. They have a strongly elongated body; about 2/3 of the length
is the tail, bordered by a leathery fin plica. The body is cylindrical, with
a flat head, a wide mouth with corneous teeth and small eyes. One flexible conical
wattle grows at each corner of the mouth. The color of the tadpole is brown
or ochre-red with a pinkish belly and a marble pattern on the back. This tadpole
makes the most of the opportunities provided by the reservoir until it dries
up. When liquid smelly mud remains in a puddle instead of water, this tadpole
has not yet passed to metamorphosis. Instead, it begins to dig out the bottom
of the puddle, and the rest of the water accumulates in the resulting hole.
The tadpole gradually deepens the bottom of the hole and turns it into a vertical
shaft. At the bottom of this shaft, it curls up into a ball and secretes mucus,
which glues the surrounding dirt into a protective cocoon, like the Protopterus
lungfish acts. Hence the name of the species: “namomu” is the name of the Protopterus
in Sudan. In the dry season, this tadpole lies in its burrow, curled up like
a ball.
When the new rainy season begins, the tadpole frees itself from the cocoon and
gets to the surface. At this time, competing species of frogs are still spawning,
and the namomubatrachus tadpole simply feeds on their eggs, gaining weight quickly.
Its length reaches 25-30 cm, but the weight increases many times; after that,
the time of metamorphosis comes – the tail resolves quickly, limbs develop,
and the body shape becomes characteristic of an adult. Next year, young individuals
will be able to reproduce.
Adult namomubatrachus is an insect-eating frog up to 15 cm long, with a pointed
head and powerful front limbs. The outer edge of the hind legs is covered with
corneous calluses – it is a tool for ground digging. The eyes are small, with
a slit-like vertical pupil characteristic of representatives of the family.
The body color is clay-red or brown with dark spots on the back; the belly is
yellowish-gray. The female is much larger than the male, which behaves more
aggressively during the mating season. At this time, the male almost constantly
keeps on the back of the female. A couple of adults occupies a vast territory,
on which there are several reservoirs. In each of them, the couple spawns a
small portion of eggs – in order to avoid competition between their own tadpoles.
This species has a clearly expressed territorial behavior – the breeding pair
actively drives away rivals with the help of sound signals, and sometimes attacks
them. In each puddle on the territory of a pair of adults, one or more tadpoles
grow up, depending on the size of the reservoir.
In a drought, an adult burrows into the ground, often occupying rodent burrows.
The frog is fenced off from its neighbors by a loose plug made of the ground.
Life expectancy is up to 14-15 years.
Mephistopheles’
toad (Bufo mephisto)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: Philippines, riverbanks and thickets of large herbaceous plants.
In the human epoch a significant number of amphibian species became extinct
due to habitat destruction and pollution. But some species due to human activities
have greatly expanded their range, which gave them an advantage at the end of
human era. Such species include the cane toad (Bufo marinus), which from the
tropics of the New World settled around the world and has successfully got acclimatized
in places where it never live previously. The efforts of people to exterminate
this amphibian mostly proved unsuccessful. At the end of the global ecological
crisis in various parts of the world numerous descendants of this amphibian
evolved. At Philippine Islands the Mephistopheles’ toad is found – a large toxic
species of amphibians.
The body length of this animal is more than 30 cm and its weight is up to 2.5-3
kg (female is larger than the male). This is a massive short-legged animal:
Mephistopheles’ toad cannot jump, but quickly walks “in a trot” and even runs
“at a gallop” – it is the only thing that remains of its ability to jump. The
appearance and color of this species are very recognizable. On the head of Mephistopheles’
toads hornlike thickenings over the eyes grow, which determined the name of
the animal. They have larger size at males, and in the mating season they also
become bright orange, being noticeable to potential rivals. The main body color
is brown with bright red spots, the largest of which is located on the lower
back and sides, like a saddle. At different subspecies coloring and pattern
vary. The subspecies B. m. diabolicus from the north of the
Philippine archipelago differs in a black background color with a lot of small
white spots and white fingertips. At the southern subspecies, B. m.
cernunos “horns” above the eyes are doubled, and the skin is brown
with small red dots. At the subspecies B. m. tuberculata from
the sea coast of the southern Philippine archipelago the growths over the eyes
are blunted, and there are many outgrowths on the skin of the parotid glands.
The locally restricted subspecies B. m. satanas inhabits a
small island on the southern coast. It has a very large size (weight up to 4
kg) and is a major predator of the island. It is characterized by brown color
with red spots scattered mainly on the sides, and by very large triangular growths
on the eyes. All subspecies of Mephistopheles’ toads have a light belly, the
color of which varies from white to lemon yellow.
These toads are predators that feed on small vertebrates. The jaws of the animal
are shortened and strong and the snout is blunt. Mephistopheles’ toad ambush
hunts small rodents and reptiles, and may eat a chick or a large insect. Near
rivers, these toads attack ground-dwelling crabs and crush their shells with
their jaws. The subspecies B. m. satanas is able to eat pigeon-sized
birds and replaces absent small predators on its native island. It is possible
that competing species of predatory mammals were ousted by these amphibians
and died out. The main hunting method of these sluggish animals is an ambush.
Mephistopheles’ toad usually hides in the forest floor, often near water. When
masking itself, it is buried in the ground about halfway and threw on itself
leaves and pieces of bark. This toad hunts most actively is in the afternoon,
when the heat of the day subsides, but it is not very cool yet. Some individuals
stay active at dusk and in the early hours of the night, when small mammals
leave their shelters.
At this species there are not natural enemies among large vertebrates, as Mephistopheles’
toad is poisonous, like its ancestor. The main part of the poison is contained
in large parotid glands, but the poison is also produced by numerous skin glands.
A captured toad strongly swells, utters loud gurgling sounds and “sweats”, releasing
the abundant poison from skin glands. In addition, it can bite the enemy with
strong jaws. This amphibian’s poison is its primary means of protection. Only
eggs are vulnerable to predators (some of which even eat the tadpoles of this
species), and the poison begins to produce at the tadpole an hour after hatching.
For a while the tadpoles completing metamorphosis, as well as young toads, just
having left the water, lack the poison – it takes place due to the peculiarities
of the physiological processes accompanying the restructuring of the animal’s
organism. Some days after the completion of metamorphosis the producing of the
poison in quantities sufficient for protection starts again.
The breeding season of this species stretches over six months during the rainy
season. At this time males occupy shallow water and compete in the volume of
their voice. The courtship song of Mephistopheles’ toads is like the trill of
birds, but continuous, monotonous and unvaried. Fertility is up to 50-80 thousand
eggs, incubation lasts 3-4 days. Tadpoles feed on microscopic algae, developing
for about 10 weeks. Maturity of this species occurs at half a year of age, and
the lifespan is 18-20 years.
The idea of the existence of this species of amphibians is
proposed by Nick, the forum member.
Translated by FanboyPhilosopher.
Rospo,
or Sicilian digging toad (Bufo fossoris)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: light forests and thickets of shrubs in the mountain “oases” of the
Mediterranean basin at the place of the Sicily island.
The drift of Africa towards Eurasia resulted in the closure of the Strait of
Gibraltar and the catastrophically rapid drying up of the Mediterranean Sea.
The change in the natural environment led to the disappearance of a significant
part of the moisture-loving fauna, which was forced to retreat to the mountains
– to the north, to the mountain range of the Alps, and to the former islands
of the Mediterranean, towering like mountains among the salty desert the sea
bottom turned into.
Rospo is a descendant of the European toad (Bufo bufo) living in Sicily, one
of the few amphibian species of this region. The name means “toad” in Italian.
The total length reaches 15 cm, weight is up to 500 g; sexual dimorphism is
expressed – females are larger than males. The body of representatives of this
species is wide and squat, with shortened limbs; the head is large, with a wide
mouth and relatively small eyes. On the back and sides, there are longitudinal
folds of skin of various lengths. The skin is dry and bumpy, permeated with
a network of thin capillaries. The fore and hind limbs are very muscular, there
are horny calluses on the outer edge of the hand and foot, designed for digging
the soil. The swimming membranes between the digits are almost absent.
The eyes are orange, and the pupils are horizontal. The color of the upper side
of the body varies depending on sex and age, as well as the time of year. It
may be not only gray, but also olive, dark brown, terracotta, sandy, sometimes
with dark spots or a light stripe along the backbone – the coloration features
are characteristic of local subpopulations. The belly, underside of the head
and limbs are yellowish, often with dark spots on the throat.
Rospo inhabits places protected from the daytime sun: sparse woodlands (mainly
thickets of spiral-leaved oak), as well as thickets
of shrubs (mainly thickets of berry
salttree) along the shores of salty reservoirs. This species is resistant
to salt water and can get rid of it: salt is excreted from the body by spraying
brine through the nostrils. At night and in the morning, the toad absorbs dew
through the skin, replenishing water stock in its body. This species leads a
terrestrial lifestyle, burrows into the ground during the day, and hunts small
snakes, lizards, shrews and invertebrates at night. Rospo catches the prey with
a sticky tongue and holds it with its front limbs.
In summer, this species runs into aestivation. Before doing it, toads stock
fat, which prevents them from dying from exhaustion during aestivation. Fat
also serves as a source of metabolic water. For aestivation, the animal buries
itself in the ground to a depth of 1 meter, often using rodent burrows.
Rospo is quite aggressive and can utter sounds similar to the bull roar. To
protect itself against enemies, the animal uses a protective masking color and
toxic secretions of glands located behind the eyes. The glands secrete a thick
sticky poison when a predator tries to swallow the toad. Being attacked by a
not very large animal, the digging toad rises on its paws and takes on a threatening
appearance, swelling up and making jerky sounds. Having seen a larger animal,
the toad freezes and can dig slightly into the ground. If this does not help,
it flees, jumping like a frog, although in a calm state it moves at a walk.
Its enemies are snakes and birds of prey (salt
hawks and maned crows);
shrews and lizards are dangerous for young animals.
Reproduction is very irregular. The breeding season begins during the spring
rains, which can fall only once in some years. When temporary reservoirs appear,
in adult animals, the rapid development of germ cells begins, and males begin
to sing their courtship songs intensively. When forming a pair, the male firmly
grabs the female with his front paws and holds her, scaring off competitors
at the same time. The female lays up to 400 eggs 2 mm in diameter. Male and
female immediately take their parts of the clutch in the mouth, where incubation
takes place. While there is an opportunity, the animals remain in the water,
simultaneously filling their bladders with it. Tadpoles appear after 2 days,
eat the remains of the egg shells and unfertilized eggs, and then feed on the
secretions of the epithelium of the oral cavity of the parents. With a lack
of food, the cannibalism is common among them. Development lasts 2-3 weeks,
no more than 20-30 individuals survive to it at each parent. In case of danger,
an adult individual can swallow the offspring, but in a calm environment regurgitates
the young back immediately: the adult toad does not feed all the time of bearing
offspring. After the end of metamorphosis, young individuals leave the parent’s
mouth. Sexual matyrity comes at the age of 4-5 years.
This species is characterized by very slow growth and significant longevity
– animals often reach 50 years of age.
This amphibian species was discovered by Wovoka, the forum member.
Louhi’s
toad (Bufo louhi)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: Fennoscandia and eastern Europe, forests of various types.
Neocene toads are no less diverse than Holocene ones. After the era of human
domination and the ice age, the species diversity of toads had greatly decreased;
local endemics and tropical forms have suffered in greatest degree. Nevertheless,
quite a large number of species survived, and the family persisted. After the
disappearance of humankind and the stabilization of natural conditions, toads
resettled across the former range and began active speciation, occupying ecological
niches of extinct species. Louhi’s toad is one of the common toads of northeastern
Europe, a descendant of the European toad (Bufo bufo).
It is a small amphibian species – the body length of an adult is up to 15 cm
(males are smaller than females). The body is broad, with a large head and relatively
short legs. The skin color is gray with a brownish tinge; there are several
dark spots near the eyes and on the muzzle, which give the impression of an
expression of disgust. The belly is grayish-white and lacks any spots. The skin
looks wrinkled because of the folds and bumps of the skin glands (hence the
name in honor of Louhi, an old witch from the Kalevala, the mistress of Pohjola).
Louhi’s toad feeds on land invertebrates – mollusks, insects and their larvae,
woodlice and worms. After hunting, it stays for a long time in wet shelters.
In the short northern summer, it actively eats away, stocking fat for hibernation:
in well-fed toads, folds and wrinkles are less noticeable. This species is remarkable
for its resistance to low temperatures: animals begin to appear on the ground
surface already during the snow thaw and often wander through the snow in search
of invertebrates. Wintering usually takes place in the burrows of small mammals,
or under rocks in a self-dug shelter. However, this toad regularly winters at
the bottom of reservoirs, breathing through its skin. When attacked by medium-sized
predators, the Louhi’s toad rises on outstretched legs and inflates the body.
Like its ancestor, it escapes by jumping only in case of extreme necessity.
Usually, this animal moves along the ground at a walk.
The breeding season begins very early – just a few days after the end of hibernation,
in March-April. Louhi’s toads reproduce all their lives in the same reservoirs
– most often in the places where they hatched. For reproduction, animals migrate
several kilometers through the forest. In the reservoir, each male has his own
territory, which he protects from other applicants. Within a week after the
males share the pond, they call the females with their “bre-ke-keks” calls.
Females prefer to mate with the most vociferous males, although others sometimes
manage to “intercept” them on their way to the most attractive partners, and
then two or three males can fertilize the clutch of one female simultaneously.
The female lays 1600 eggs; the clutch looking like slimy cords is wound on aquatic
plants, etc. Soon after spawning, adult toads leave the pond and do not protect
the clutch. A month later, tadpoles hatch from the survived eggs, which spend
from a year (in the south of the area) to 3 years (in Scandinavia and in the
Khibiny) in the water. The Louhi’s toad reaches sexual maturity 2 years after
metamorphosis; life expectancy in adulthood is up to 22 years.
This species of amphibians was discovered by Nick, the forum member.
Hammon’s
toad (Bufo baalhammonis)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: savannas of North Africa.
During the global ecological crisis of the late Holocene – early Neocene, warm-blooded
predators experienced a crisis, and their ecological niches were partially occupied
by “generalists” from other taxa. The significant depletion of the species composition
of ecosystems during the mass extinction gave a ghostly opportunity for a breakthrough
even to representatives of non-numerous taxa that were on the sidelines in ecosystems.
Amphibians managed to take advantage of this opportunity: one of the successful
species was the European green toad (Bufo viridis). Although its range has decreased
in Eurasia due to the ice age, the African part of the population has not been
affected. Moreover, the descendants of this species managed to grow up and slightly
displace rodent-eaters from among the reptiles. One of them is Hammon’s toad,
named after the supreme Carthaginian deity – the god of the sun, weather and
fertility.
The body length of this amphibian is up to 20-25 cm; females are larger than
males. The skin is gray-green in color; several slightly keratinized prickles
grow on the head, resembling a crown. Because of the drying up of the Mediterranean
Sea, the climate of North Africa has become more arid, so Hammon’s toads avoid
showing up on the ground surface during the day and lead a twilight lifestyle,
getting out of hiding late in the evening and early in the morning. Low-growing
desert shrubs, rocks or someone’s burrows can serve as shelters for them. The
Hammon’s toad is interested in reservoirs only during the courtship season,
although during the dry season it regularly approaches the shore and lies down
on wet areas or buries itself in mud, replenishing moisture losses by the body
through the skin. This species is a predator; it feeds on large arthropods,
mollusks and small vertebrates – mainly rodents, but does not disdain reptiles
and other amphibians. Sometimes it eats small individuals of its own species.
The adult Hammon’s toad has few enemies – it is poisonous, and only monitor
lizards eat it without harm to themselves.
The breeding season begins in spring, in the first half of the rainy season.
If the rains do not fall, or their amount is insufficient, toads may not breed
this season. The hammon’s toad males gather in the shallow waters of any more
or less large reservoirs and call the females with a rumble that can be heard
for several kilometers. The female chooses the largest and loudest male. When
the choice is made, the male embraces the mate, who swims to the depth. There
she spawns a clutch of 550-600 eggs enclosed in slimy ribbons that swell strongly
in water. After 10 days, tadpoles hatch from eggs. They grow fast, feeding mainly
on algae. Metamorphosis takes place 1.5-2 months after hatching. Soon after
metamorphosis young toads stay near permanent reservoirs – they are not as resistant
to moisture loss as adults. As the animals grow and mature, they settle in drier
areas. Sexual maturity comes at 2 years; life expectancy is up to 30 years.
This species of amphibians was discovered by Nick, the forum member.
Giant
Chirping Toad (Terrabufo gigantea)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: Meganesia, any vegetated area with sufficient seasonal water.
The invasion of Australia by man let to many dire consequences for both australian
flora and aboriginal people. Many introduced animals decimated the indigenous
fauna, including perhaps the most ravenous of all, the Cane Toad (Bufo marinus).
The Chirping giant toad is a descendant of this animal, and reaches considerable
size, even greater than its ancestor. Snout to vent length is 35 centimeters,
legs are shortened and heavily ossified, hopping is slow and ponderous, usually
the animal moves by clambering. Shoulder glands posess toxin which is exuded
when the animal is harassed by a predator. The mouth is enlarged (moreso than
its ancestor), and the animal posesses large bony “fangs” with which it kills
and manipulates food. Males posess thickened skulls used in courtship conflicts,
there are bony ridges above the eyes. The dorsal surface of the animal is fortified
by large corneous warts which meet at the edges, acting as rudimentary armor.
The belly of this toad is relatively soft, and predators are sometimes able
to flip the animal to rip out its belly. Color of upper side of the body is
olive brown, speckled with dark brown, and creamy yellow on the underside.
Depending on the temperature and humidity, it may be active during the day or
night. These toads are able to dig shallow burrows and scrapes from which they
ambush their prey, such as mice, rats, lizards, small birds, frogs, and large
insects. During favorably cool, damp, or shady hours they will wander in search
of food, which they will hold down and kill with their jaws, and swallow whole.
Smaller prey is taken and swallowed alive.
Males compete aggressively for females, using their thickened skulls to flip
eachother onto their backs. Females are held and spawned with in shallow water
with the help of the male’s front limbs. Breeding season is all year, where-ever
sufficient water for spawning exists. Tadpoles mature quickly, and commonly
fall prey to insects, fishes and snakes which are resistant to their toxin.
Lifespan up to 20 years.
Related, Lake
Chirping Toad (Terrabufo lacustris), slightly smaller, coloring is
dull grey with black spots. This toad spends most of its time in the marsh and
swamp-forest on the southern fringes of Carpentaria and Arafura lakes, and is
able to spawn in brackish water, poisonous tadpoles are commonly prey to specialised
fish and snakes.
Forest Chirping
Toad (Terrabufo media) found in forests of Northern Meganesia. Medium-sized
form posessing bright warning colors advertising high toxicity, bright orange
with black speckles on the dorsal side, and patterned white and yellow on the
belly.
These species of amphibians were discovered by Timothy Donald Morris, Adelaide, Australia.
Egg-bearing
sac toad (Saccobufo ovifera)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: Southern Europe (south of the Alps mountain range), dry laurel and
oak forests.
In the Neocene, the territory of the southern part of Europe became an extremely
unfavorable place for amphibians to live. The drying up of the Mediterranean
Sea has turned vast areas into deserts, depriving amphibians of a vital resource
– water necessary to support the life of adults and for the development of offspring.
The most part of the species diversity of these animals is located in the basins
of rivers flowing down from the Alps, but some species, avoiding fierce competition,
began to develop drier habitats. The main direction of the evolution of these
animals was to weaken the connection with water and reduce the time of the vulnerable
aquatic stage of development. Toads have been particularly successful in developing
dry areas; among them, there are even species that can
lose a lot of moisture without risking their lives. In the dry areas
of the Mediterranean hollow, several species of the sac toad genus (Saccobufo)
live, demonstrating various degrees of attachment to the aquatic environment
during the rearing of offspring.
The egg-bearing sac toad is the least specialized species of the sac toad genus.
In this species, sexual dimorphism is clearly expressed in size: the body length
of the male is about 5 cm, and the female is up to 10 cm. These animals have
an appearance typical for representatives of the family: almost dry warty skin
and short limbs, designed more for walking than jumping. However, the pads of
the paws of all species of the genus are covered with thick keratinized skin,
which allows them walking on sun-heated stones or dry clay. The head is large,
with relatively small eyes. The coloration of the egg-bearing sac toad is gray-green
with brighter sides; the belly is grayish-white. The iris of the eye is red
with a brownish tinge.
Like other toad species, this species is a solitary insect-eating creature.
During the day, it hides in shelters – usually in burrows that it digs by itself,
or occupies and expands abandoned ones. At night, the egg-bearing sac toad goes
hunting and searches for breeding partners. In the winter drought, this species
becomes numb in the shelter. Males of this species search for females in the
fall and “ride” them for a while without letting go. Both animals aestivate
in the same shelter.
A characteristic feature of the egg-bearing sac toad is the cloacal sac stretching
along the entire belly part of the body. In the dry season, this sac is filled
with liquid, which is used as an additional water reserve. In the spring, when
the breeding partners become more active, even before the rains begin, the courtship
games take place. The mating songs of this species have actually disappeared:
the male only “formally” utters short squeaky trills while in the same burrow
with the female. The male fertilizes the female’s eggs by injecting seminal
fluid into her cloaca and then leaves the female. After that, the female lays
up to 200 eggs, which immediately enter the cloacal sac and begin to develop
almost to the stage of the tadpole’s hatching. Within a few days after spawning
eggs, the female carries them with her (hence the name) and searches for a suitable
reservoir for the development of offspring – usually at this time the rains
already begin. When the female spawns the eggs into the water, tadpoles hatch
after about an hour; in the first days of life, they devour the eggs of other
amphibians and are even able to gnaw the soaked corpses of animals that died
from drought. Later they begin to feed on planktonic crustaceans and algae.
If the reservoir begins to dry up, they turn to predation and cannibalism, and
then the last surviving individuals get an opportunity to complete metamorphosis.
Normally, the development of tadpoles lasts about two weeks.
Young individuals of the egg-bearing sac toad take part in reproduction in the
third year of life. Life expectancy can reach 17-20 years.
False-viviparous
sac toad (Saccobufo pseudovivipara)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: Southern Europe, semi-desert areas, the vicinity of drying reservoirs.
Various species of the sac toad genus (Saccobufo) show various degrees of adaptation
to life in arid conditions. Some species of this genus only keep developing
eggs in the body for a short time, throwing it off relatively quickly, and thus
are more similar to ancestral forms that spawned eggs in water. In false-viviparous
sac toad, the care of offspring is more complicated. This species reproduces
before the rainy season and in a similar way bears its eggs in the cloacal sac,
where they develop for some time, and thereby buys time for the development
of offspring during the rainy season. The fertility of the false-viviparous
sac toad is relatively small: only 40-50 eggs. Nevertheless, this is compensated
by the fact that the eggs are very large and receive part of the necessary moisture
from the parent’s body through the epithelium lining the cloacal sac. In this
species, the male not only fertilizes the eggs, but also releases part of the
stored liquid into the cloacal sac of the female, making a much greater contribution
to the care of the offspring than in the egg-bearing
sac toad (Saccobufo ovifera). The female stays in hiding for some time,
and the male protects her from possible competitors, not letting her go even
on the ground surface until she releases her offspring into the pond. At this
time, the male, keeping on the female’s back, utters loud warning sounds, forcing
relatives to keep their distance. The female releases from her cloacal sac already
formed tadpoles, which immediately begin to hunt small animals, including tadpoles
of other species, and only from 3-4 days of independent life begin to scrape
algae from the bottom of temporary reservoir. Their development takes no more
than 2 weeks.
The appearance of adults of this species is typical for toads: they have short
hind legs, a robust physique and relatively dry warty skin. A strong skull helps
to dig the ground during the making of a shelter, and on the outer side of the
forearm and hand, individuals of both sexes have a row of corneous tubercles
that help to dig even dense soil. The eyes are small, located on the edges of
the head and are able to sink completely into the sockets during the ground
digging. The body color of this species is reddish-brown with small specks on
the back and crown; the belly is grayish-white. The adult female of this species
reaches a length of 14-16 cm; the male is up to 12 cm long.
This species reaches sexual maturity at the age of 4 years, and life expectancy
is up to 25 years.
False-pregnant
sac toad (Saccobufo pseudogravida)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: “oasis” in the Mediterranean hollow in the place of the island of Crete.
The Mediterranean hollow is one of the harshest places on the Neocene Earth.
It is a vast land area between North Africa and Southern Europe, covered with
saline lands. In some places, there are vast hypergaline lakes, and the former
Mediterranean islands have turned into dry mountains towering over the salt-covered
plain. On their tops, peculiar “oases” are formed, where small representatives
of fauna live, being able to survive with a minimum amount of water or without
it at all. Such an area looks completely unsuitable for amphibians; however,
they are found even in such places.
In the oak-laurel forests of the “oasis” left over from the island of Crete,
one representative of amphibians of the Mediterranean hollow, the false-pregnant
sac toad, lives. This toad species reaches 7-9 cm in length and is remarkable
in its powerful physique, short strong paws and relatively dry skin. Sexual
dimorphism is not expressed in this species. The body color of this amphibian
is modest – grayish-brown with darker specks on the back and upper part of the
head. There is a lesser number of specks on the sides, and the belly is yellowish-white.
This species has large corneous growths on the fingers and the outer edge of
the front paws, which help this amphibian to dig the ground.
The most vulnerable stage of the life cycle of amphibian is the tadpole – the
aquatic larval stage. Living in a limited area actually lacking even temporary
reservoirs, the false-pregnant sac toad has developed a special method of reproduction
for which a reservoir is not required. The false-pregnant sac toad spends a
significant part of its life underground, awake or in a condition of aestivation.
Usually males and females of this species run into aestivation together so that
in the spring they do not waste time searching for a mate. In spring, the male
and female mate in the underground chamber: they backwards turn to each other
and connect with cloacae for a long time. Fertilization takes place in the female’s
body. The fecundity of this species is very low: no more than 10 large eggs;
it pays off with a high survival rate of offspring.
This species has the most specialized form of parental care among all sac toads:
an adult individual bears eggs in a “false uterus”, which role the sac-like
outgrowth of the cloaca plays, being located under the belly skin. Fertilized
eggs are shared between a male and a female, equally involved in the rearing
of offspring. The eggs attach to the wall of the cloacal sac, which in this
place abundantly sprouts with villi penetrated with blood vessels. Through the
blood, the parent body supplies with oxygen and feeds the tadpole, which eventually
turns out to be attached by its belly to the wall of the cloacal bladder of
the parent. Its tail is initially reduced, but its limbs develop very soon.
During their “pregnancy”, adult toads of this species continue teir usual life.
After 2 months of “pregnancy”, young individuals leave the cloacal sac of the
parent and begin leading an independent life.
Sexual maturity in young individuals comes at 4 years of life, and the life
expectancy is about 30 years.
Dwarf
sac toad (Saccobufo minima)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: small “oases” in the Mediterranean hollow.
The vast hollow that remains on the site of the Mediterranean Sea is one of
the most unfavorable places for life on the Neocene Earth. Here, there are occasional
rains coming from the Atlantic, and rivers flow from the peaks of the Alps,
which sometimes reach the vicinity of the Mediterranean hollow and support the
existence of small salt lakes. Otherwise, only the most resilient species survive
in these places: the salt left after the drying of the Mediterranean Sea makes
life in these places extremely difficult. Nevertheless, there are hypergaline
marshes and peculiar dry “oases” that were islands in the human era. Drought-resistant
plants grow in these “oases”, supporting the life of rare animals.
Invertebrates, reptiles and birds form the basis of the fauna of the “oases”,
but other groups of animals are also represented there. In particular, representatives
of amphibians adapted to life in extremely arid conditions – sac toads – live
in small “oases” of the Mediterranean hollow. Some of their species managed
to get rid of the need to develop in water, having developed a semblance of
a true vivipary: a large cloacal sac turned from a temporary storage for eggs
and moisture reserves into an incubator for offspring, and after spawning eggs,
parental individuals equally share the worries associated with offspring bearing.
In conditions of limited habitats in small “oases” of the Mediterranean hollow,
a special amphibian species, the dwarf sac toad, survives successfully. The
body length of an adult of this species is about 25 mm. It is distinguished
by a robust physique and good ground-digging abilities, digging deep burrows
for her life. Part of the burrows lies under small stones that serve as natural
condensers of moisture brought by the wind. On the outer edge of the front limbs
of this species are large corneous growths that help loosen and shovel the ground.
The skull is strong, and its anterior edge is thickened; the eye sockets are
shifted to the sides of the head. The body coloration is brown with yellowish
speckles on the sides. Sexual dimorphism in this species is not expressed, and
even the border between the sexes has been erased – it is also a consequence
of the low number and sparsity of populations. Nevertheless, this species is
found in many “oases” of the Mediterranean hollow, and the secret of success
in survival is associated with this feature.
In the dwarf sac toad, the sex is not genetically fixed and can change depending
on life circumstances during several months, by the next breeding season. Initially,
each individual develops according to the female type and is able to lay parthenogenetic
eggs, which normally develop in its cloacal sac. The fertility of this species
is very low: each individual gives birth to only 2-3 well-developed juveniles.
However, due to parthenogenesis, the slow reproduction rate is compensated by
the potential ability of all born individuals to give offspring. When a population
reaches a certain density, some of the females begin to turn into males – it
restrains the growth of the population and creates conditions for the growth
of genetic diversity simultaneously. In fact, one individual can give rise to
a functional population. This amphibian species has also another feature: small
juveniles search for sleeping birds at night and hide on their body among the
plumage. Doing it, they get a possibility of transferring by the bird to a new
“oasis”, where it can establish a new population or join an existing one. Probably,
this species has spread widely in the oases of the Mediterranean hollow mainly
with the help of birds.
This amphibian species feeds exclusively on insects and is nocturnal.
Sexual maturity in the dwarf sac toad comes at the 2nd year of life; the life
expectancy is up to 15 years.
Mossy
corpse-mimic toad (Cadaverobufo heli)
Order: Tailless amphibians (Anura)
Family: Toads (Bufonidae)
Habitat: Philippine Archipelago, lowland rainforests.
One of the important ways of survival in nature was mimicry and disguise. In
the process of evolution, a kind of competition takes place between predator
and prey – both try to hide, to be unrecognized, to look not who they really
are. These tendencies were traced in the coloring of the skins of zebras and
tigers, and in the imitation of harmless insects by bright-colored well-armed
wasps, and in the bizarre appearance of stick insects and mantises, and in many
other cases. In the Neocene, this tactic remained relevant and even rose to
a new level.
In the humid depths of the Philippine forests, near the corpses of animals,
strange toads that hunt corpse-eating insects can often be seen. Their back
and paws are covered with green growths, perfectly mimicking moss, due to which
the amphibian looks like a green moss hummock. These growths contain glands
that produce poisonous secretions, which cover these growths in drops, like
dew. Such an unusual disguise protects amphibians from predators, which also
do not miss the opportunity to search for food near carrion. This toad is a
mossy corpse-mimic toad, or a corpse toad. The name Hel, which became a specific
epithet of the animal, belonged to the Old Norse goddess – the mistress of the
kingdom of the dead, a half of whose body had a color of raw meat. The peculiarities
of the biology of these amphibians fully justify this choice: these toads can
be found very often near the corpses of animals that they find by smell, and
the lower part of the body of this toad has meat-red coloration with whitish
spots of irregular shape. The animal’s body is short, squat and widened, with
strong short limbs. There are swimming membranes on the hind limbs between the
toes, but they are not present on the front ones. During the breeding season,
sexually mature males develop nuptial pads on the first two fingers of their
front limbs, which help the male keep on the female during spawning. Sexual
dimorphism is clearly expressed: the length of an adult female reaches 10 centimeters,
males are half as small.
In addition to catching prey near large corpses, this amphibian has two more
hunting tactics. One of them is the following: having found the corpse of some
small rodent, the toad drags it to a secluded place, where it also ambushes
necrophagous insects without fear of predators.
The second hunting tactic is more curious. When there are no suitable corpses
for hunting for a long time, several toads come together. One or two of them
turn upside down with their bellies imitating the color of decomposing meat,
and begin to emit a noticeable cadaverous smell, mimicking a decomposing corpse
and attracting insects that are caught by their fellows lurking nearby. The
bait toad can also catch several insects that happen to be near its head. Due
to this hunting tactic, the amphibian got the name “corpse-mimic”. In case of
danger, the toad shrinks strongly, dragging its limbs under the body, and emits
poisonous secretions with its whole body, accompanying it with a strong cadaverous
smell.
The amphibian’s activity is predominantly diurnal and twilight. It prefers dense
humid forests and the banks of rivers and streams, and moves on the ground in
short jumps. At low temperature (in the mountains) these amphibians become sluggish
and become numb. Usually in the dry season, toads hide under boulders along
rivers, under a layer of fallen leaves, in abandoned burrows of other animals,
under dense grass sod and in other shelters where moist air is preserved. During
the molting period, the corpse-mimic toad climbs into a secluded place, and
inflates its body until the layer of old skin bursts on the back, and then,
shifting the skin to the mouth, eats it. During the mating season, when the
water temperature reaches 25°C, males occupy territories along the banks of
forest streams and begin uttering mating calls – a loud whistle in one tonality.
The corpse-mimic toad reproduces in water with a slow current, preferring reservoirs
with clear water. At night, the males move into the shallow water and begin
calling females. When the female appears, the male climbs on her back and fertilizes
the eggs she spawns. A female may carry a male for several days to a week before
spawning eggs. One female can be fertilized by several males. Each adult female
lays 2,000 to 5,000 eggs in one season. Eggs are spawned in long transparent
jelly-like threads among aquatic vegetation. After the egg bunches are fixed,
the parents cease to show any care for the offspring. The female can spawn not
once, but twice a year, depending on the habitat – repeated laying is more often
done by individuals from wetter areas. The reproduction of the corpse-mimic
toad is associated with the beginning of the rainseason, when temporary reservoirs
form in plenties.
Tadpoles develop for about 4 weeks; after metamorphosis, young animals hide
among the forest floor and catch mainly flies, luring them by smell. Sexual
maturity comes at the 2nd year of life; life expectancy is up to 25-28 years.
This species of amphibians was discovered by Morgot, the forum member.
“Termite
queen” (Batrachoregina hygrophila)
Order: Tailless amphibians (Anura)
Family: Narrow-mouthed toads (Microhylidae)
Habitat: South Africa, dry savanna.

Picture by Alexander Smyslov
Amphibians are the primal vertebrate land inhabitants.
They had not completely left the water environment – water is vital for them
in spawning season,
and the majority of amphibians prefers to live in wet places. But despite
of such
vital requirements these animals had managed to adapt even to inhabiting
in arid areas. Though Neocene differs from Holocene in more humid and warm
climate,
deserts and savannas cover a significant part of planet. One of such places
is South Africa. Sometimes it is too hard to find water here, and termites,
which are very moisture-loving insects, are compelled to dig deep vertical
shafts, reaching up to aquiferous stratum. And various animals searching
for moisture and safety willingly use their constructions. It is difficult
to not
cause aggression of furious termites from soldier caste, but nevertheless
in buildings of termites the set of various animals lives. Basically there
are
insects among them, and occasionally millipeds and spiders are. But one of
large “guests” of termites is the strange big-bellied frog about 8 centimeters
long. It behaves rather confidently and feels like in the full safety, surrounded
by insects equipped with protective weapon. In dry season this frog meets
only in constructions of termites. For these features it has received the
name “termite
queen”.
The secret of loyal relation of termites to their “queen” is contained in
chemical substances secreting with slime of “termite queen”. Skin secretion
of this
frog reduces aggression of termites. These insects perceive this frog almost
like their relative: they lick secretions of skin of “termite queen”, and
do not attack her, even when the frog eats several termites one by one.
“Termite queen” has short and thick, almost spherical body with rather short
paws. This frog is not able to jump, and moves on the ground, walking like
a toad. Skin of “termite queen” is smooth with strongly advanced skin glands
producing secretions attractive to termites. Its colouring is dusky – grayish-brown
with small dark spots at the top part of body; stomach is white. Head of
this frog is short and wide; eyes are small, protected from stings of termites
by
transparent eyelid. “Termite queen” has bad developed sight sense – it spends
the most part of life in complete darkness of galleries of termitary, and
appears at the ground surface only at night during the rain season. But this
animal
orientates very well in termitary due to sense of smell and advanced touch
sense.
This frog is adapted to digging habit of life. On its paws large corneous
callouses develop on external edge of foot and forearm. It digs holes in
the basis of
termitary and frequently uses ready galleries of termites for movement. The
similar way of life is led by desert mole toad (Cryptophrys
cornicephalus),
living in Meganesia; it is an example of the convergence, because the species
from Meganesia belongs to another family.
“Termite queen” eats termites and other insects living in termitaries. It
obviously prefers large insects – crickets and cockroaches, and also adult
termites.
But in each termitary only one or two adult frogs of this species live (and
also some small not sexually mature individuals may exist near them), therefore
damage put by such “queen” to the colony of termites is small. Even more
so, it eats various symbiotes and parasites of colony of termites more often,
than
termites.
This frog spawns during short rain season. When it feels that the ground
becomes wetter, it leaves termitary using galleries, and digs a tunnel to
the ground
surface. Having reached a surface, frog gets out of hole not at once – it
makes it only when night comes. At this time tens of “termite queens” hasten
to temporary
reservoirs to spawn. Males get out on surface earlier, than females. They
share coastal sites of reservoir to set of territories, and involve females
with
the help of call similar to long descending whistle. In day time males prefer
to dig in ground on coast of reservoir, or hide in thickets of marsh plants.
Females creep to the surface later. Within one night pairs form, frogs spawn
and immediately leave a reservoir, coming back to the same places where they
lived before. “Termite queen” is never mistaken, returning after spawning
only to the same termitary. At the second night only separate females come
to reservoirs,
and third spawning night, as a rule, does not come.
In clutch of these frogs it is totaled up to 300 eggs stuck to one mucous
ball. Tadpoles of these frogs in the beginning eat algae, and from 9 - 10-day’s
age
become predators and cannibals. Up to the metamorphosis (at the age of 45
– 50 days) no more than 10% of the general number of tadpoles survives. Young
frogs dig in ground and search for galleries of termites along which they
reach
dwellings of these insects. The part of frogs perishes from stings of termites
– not all species of these insects accept such “visitors” peacefully. But
the frog managed to lodge at termites of suitable species can live about
ten years.
Motley
midgefrog (Miniranula multicolor)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: Western Siberia, swamps and marshy forests.

Picture by Alexander Smyslov
Significant territories of Eurasia differ in rigorous continental
climate, and this circumstance does not favour to settling of amphibians and
reptiles there. Only few species of these animals had adapted to seasonal climate.
Those species which could make it had developed various adaptations, permitting
to resist to the most terrible enemy – to winter cold.
When swamps and lakes of Western Siberia release from ice cover, it is possible
to see various aquatic animals in pieces of ice floating on surface of reservoirs.
Some of them had got in ice casually and lost, and others normally endure such
test and after will thaw, continue the habitual life. In ice captivity there
are larvae of various insects, small crustaceans and their eggs, and also small
long-bodied creatures similar to thin transparent fishes. They are tadpoles
of local midgefrogs – animals prospering in Siberian climate. They are alive,
though are frozen, and simply wait while warmth of sun will release them. But
in ice there are only separate tadpoles had got there casually. The much greater
number of tadpoles is hidden in layer of silt at the bottom of reservoir. When
ice starts thawing, their multithousand shoals leave the refuges and gather
at warmed up shoalinesses. Jaws of midgefrog tadpoles are scraping – with their
help they gather algal scurf from the surface of plants and eat tiny sedentary
animals. One of the main enemies of midgefrog tadpoles is giant
bladderwort, carnivorous aquatic plant.
One of the most terrible inhabitants of Siberia are not predators, but numerous
blood-sucking insects. Their larvae successfully survive in winter and from
the beginning of summer there is their mass metamorphosis to adult mosquitoes
and midges. These insects do not give rest to mammals and birds – they attack
by whole flights during all night and the most part of day. But just at the
moment of their metamorphosis tadpoles also turn to frogs, and adult individuals
of midgefrogs appear provided with food of suitable size.
Adult midgefrogs are very tiny creatures. The length of normally advanced individual
at all species of this genus does not exceed 20 – 25 millimeters (male is smaller,
than female). They are reliably protected from predators by strong poison which
contains in their slime. During the evolution at midgefrogs bright warning colouring
had developed. Many species of this genus of frogs distinguishing in ecology
and well distinct on features of colouring live in Siberia. All species of these
amphibians are able to climb dexterously on branches of bushes and leaves of
marsh grasses with the help of tenacious fingers and toes. At front paws the
palm forms a kind of sucker.
The adult midgefrog grows not for long, but very actively. Its length after
metamorphosis does not exceed 14 mm, but right till one month it reaches the
size characteristic for adult individual. In the same time it becomes sexually
mature and starts spawning. So high growth rate is explaining by one feature
of its physiology: is an annual frog. Closer to an autumn adult midgefrogs quickly
grow old. Besides when small two-winged insects vanish, these frogs lose an
only source of forage and are quickly exhausted.
Motley midgefrog lives in wet places – under leaves of perennial grassy plants.
This species is widely settled in swamps of Siberia and eats mosquitoes. This
tiny frog has spotty colouring – in it orange and brownish-green colors, located
by chaotic spots are combined. The size and amount of spots may strongly vary
at different individuals.
Courtship games of motley midgefrog begin in the middle of summer. Males call
females, perching on branches of bushes and high stalks of grasses. Voice of
this frog is shrill squeak. Short sound signals follow one by one in some seconds
during 2 – 3 minutes in succession, if the male is not disturbed by anybody.
At the meeting of two males sound signals follow one by one, almost not interrupting.
Eventually, weaker male stops “duel” by the first and leaves out.
Female in common with male moves to water and spawns near the coast up to hundred
very small eggs. In them also there is a poisonous substance, and it is enough
quantity of it to protect eggs from encroachments of small aquatic predators.
But tadpoles of this species are lack of such protection, and can hope only
for their own speed of movement and reaction. They gather to big shoals and
keep in well warmed up places of reservoir.
Close species of midgefrogs live in Siberia:

Picture by Alexander Smyslov
Little Red Riding Hood midgefrog (Miniranula crystallodermata) is one of tiniest species of the genus: the length of adult female is about 19 mm, male – up to 14 mm. At this frog skin is poor pigmented and so thin, that through it interiors are visible. Only on head of this frog there is a big red spot, hence it has received its nickname. It lives in shady places and eats exclusively midges and fungus gnats. Adult frogs frequently can be found on old mushrooms bored by insects. Voice of this species is lingering silent trill.

Picture by Alexander Smyslov
Leopard midgefrog (Miniranula leopardina) is one of most colourfully looking midgefrogs. Its length is about 25 mm. Background color of skin is ochre-yellow, on it small black an irregular-shaped spots are scattered. Limbs of this frog are rather short, and it seems more robust, than its relatives. It swarms up plants good, and meets on stalks and leaves at height of more than one meter above the ground. This frog can eat not only mosquitoes, but also larger insects – small moths.

Picture by Alexander Smyslov
Lemon midgefrog (Miniranula citrina) is unusually looking species of frogs. Skin of this frog is monotonous canary-yellow, and on throat there is bright yellow area. Eyes are rather large: this species leads nocturnal habit of life, and hides in foliage and holes of small animals in day time. At night these frogs become more active, jump on stalks of grasses and bush leaves. Voice of lemon midgefrog is abrupt “yelping” sounds.

Picture by Alexander Smyslov
Moonlight midgefrog (Miniranula selena) differs from other species by “varnish” blue colouring. Like previous species it is a nocturnal animal and has large eyes. It is tiny species of midgefrogs reaching the length of 18 – 19 mm. It lives mainly in thickets of marsh plants at riverbanks and lake coasts. The voice of this frog resembles cricket chirring.
Ice
frog (Cryorana glaciophila)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: bogs of Western Siberia.

Picture by Shervin
In Neocene the climate of the Earth became warmer, but in
northern hemisphere still there is a huge “refrigerator” – Eurasia, an extensive
land area of the continental climate. Fourseas influences only the European
part of continent, and beyond Ural the extensive landmass far from warm seas
is stretched. Therefore for Eurasia the seasonal climate with the great annual
amplitude of temperatures is still characteristic – from almost tropical heat
in summer up to rigorous frosts in winter. In such conditions at the most part
of continent only few species of amphibians and reptiles can exist – the winter
cold is limiting factor of their settling.
In Neocene rivers of Western Siberia overflow, forming an extensive circiut
of lakes and bogs. Here amphibians live, which have developed the various adaptations,
permitting to survive in rigorous winter. The largest one among them, the gluttonous
swamper, huge caudate amphibian, spends the winter, having dug deeply
in silt. Near to it numerous species of amphibians live, looking less grotesquely.
Dwarf midgefrogs do not winter at all
in adult condition: they perish after the first night frosts, having had time
to breed the posterity which winters under ice. But some amphibians are capable
to endure rigorous Siberian winter many times during their life and had developed
a way permitting to avoid harmful influence of cold. One of Neocene species
of frogs is capable to survive, simply freezing in ice. It is an ice frog –
one of characteristic inhabitants of bogs of Western Siberia.
Appearance of ice frog is quite ordinary for tailless amphibians. It is a frog
about 10 cm long of dark green, almost black color, with white stomach and small
yellow eyes. Hind legs at this species are long; between toes there is a wide
elastic membrane; body and head are wide and flattened.
Ice frog almost constantly lives in water, and appears on land only then when
the reservoir in which it lives dries up. Hind legs serve it almost only for
swimming, and on land ice frog walks like a toad, supporting on foot and raising
upwards toes with membrane in order to protect it against injuring. Only in
case of danger it escapes from enemy, making jumps up to one meter long. But
ice frog is too heavy to move this way to long distances. In not drying up reservoirs
ice frog lives almost the whole its life. Young individuals of this species,
recently gone through metamorphosises, leave water for a short time while their
excretory system of “adult” type is forming.
Ice frog is a predator eating various prey. It eats larvae of insects, small
crustaceans, fish fry and even tadpoles of its own species.
These frogs begin active life when ice floes still float on the surface of reservoirs.
Ice frogs emerge on the surface of water, putting their backs to rays of spring
sun, and quickly get warm due to dark colouring. When ice thaws completely and
rivers overflow banks, at this species courtship games begin. Самцы involve
females with loud rolling croaking, inflating throat. At this time they are
brightly colored – the male’s back becomes emerald-green with bluish shade,
and on forepaws dark “gloves” appear. Females (they are larger, rather than
males), on the contrary, darken, and male formed a pair with the female, is
clearly visible on the background of her body. These frogs spawn a plenty of
eggs – up to 1000 ones from one female. Eggs quickly swell in water, but their
development proceeds about 20 days because of low temperature of water. Tadpoles
of this species are tiny and translucent till first days of life. Their food
at first includes the tiniest fresh-water plankton, after which they pass to
vegetarian diet. By the end of summer tadpoles reach the length of 25 – 30 mm
and turn to young frogs. Up to the first frosts they live on land in thickets
of marsh plants. When cold season begins, they move to water and do not leave
it without emergency.
In winter ice frogs remain in reservoir even if it freezes through to the bottom.
These amphibians are not afraid of frost as they are able to resist to it. In
cold water in blood of frogs the glycerin accumulates in a plenty, interfering
growth of ice crystals. When the reservoir freezes, and fishes are hidden in
deep whirlpools, ice frogs keep activity though stop feeding. They float in
thickness of water and “hand” on the bottom surface of ice. The body of frog
becomes covered with a layer of viscous slime which interferes skin freezing.
When the frog appears “trapped” by ice crystals, it turns into ball inside slime
cocoon, having pressed paws densely to the body. At this time it closes eyes
and eyeballs move down into the head, not going beyond the surface. The slime
capsule interferes freezing of frog’s skin, but is freely pervious to cold.
However, due to natural “antifreeze” frog does not freeze through. Ice continues
grow thicker and ice frogs appear enclosed in it entirely. In such condition
they spend winter while ray of spring sun will melt ice. At this moment dark
colouring of frog helps it to get warm faster and simultaneously causes ice
around of its body to thaw.
Due to winter “preservation” this frog grows old very slowly and has significant
life expectancy – up to 40 – 45 years.
Talvisammakko,
Finnish ice frog (Cryorana fennica)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: swamps and lakes of Europe in the zone of temperate forests, taiga
and forest tundra.
Despite the warmer climate compared to the Holocene, the far north of Eurasia
in the Neocene epoch is still a very harsh, cold area where survival is not
easy for all living organisms. Rivers flowing into the Arctic Ocean and long-gone
glaciers of the Holocene-Neocene glaciation era have formed numerous wetlands
and lakes where amphibians thrive. One of the amphibians characteristic of this
frosty region is the talvisammakko, or Finnish ice frog.
Talvisammakko belongs to the same genus as the eastern
ice frog (Cryorana glaciophila), which lives in the wetlands of Western
Siberia. In contrast, the Finnish ice frog lives in Europe, from Scandinavia
to the Urals – both species originate from a common ancestor. It also is able
to freeze into ice without harm to health (hence the name: from the Finnish
words talvi – winter and sammakko – frog).
Externally, talvisammakko differs little from its Asian relative: it is also
a relatively large (about 8-10 cm long) frog with a wide body, having a dark,
rich green color (slightly lighter than that of the Siberian species) with small,
darker oblong spots on the back and sides; the belly is yellowish-white, without
spots. There are well-developed swimming membranes between the toes of the hind
paws. Females are larger than males.
Talvisammakko is an almost completely aquatic amphibian, which almost never
leaves the reservoir in which it lives, like the clawed frogs of the human era.
It is connected with the land even less than its Siberian relative. Populations
from mountain lakes with steep rocky shores may not come to land at all for
dozens of generations.
The Finnish ice frog feeds on aquatic invertebrates, fish fry, tadpoles and
other small aquatic animals.
The breeding season of these amphibians begins immediately after the ice melts.
It even happens that males begin mating calls while sitting on pieces of melting
ice. Courtship games are characteristic for true frogs – males attract females
by uttering a dull, low croak: “guaaaa-guaaaa-guaaaa” and inflating the resonator
bladder. They also change color, becoming dark blue with black “gloves” on the
front limbs. Females, on the contrary, become maroon-brown with a pure white
belly during the breeding season.
The number of eggs in one female reaches 1200. Their development before hatching
offspring takes 16-18 days; in cold years and in the north of the area, it is
delayed up to 1 month. Parents do not guard the eggs. Tadpoles, like those of
the Siberian species, are small and translucent for the first few days, then
turn brownish-gray. They also feed first on small plankton, then on microscopic
vegetation and sedentary animals. By the end of summer, having reached a length
of 2-2.5 centimeters, tadpoles undergo metamorphosis and turn to young frogs,
which during the first year of life are able to leave the water for a long time,
and even winter in the forest litter, but after the first wintering they switch
to permanent habitation in the water. Growth is very slow; sexual maturity comes
at the 5th year of life.
The wintering of talvisammakko is identical to the wintering of its eastern
relative. This amphibian lives up to 35-45 years; occasionally there are centenarians
who have crossed the half-century mark.
This amphibian species was discovered by a Dohlosavr, the forum member.
Baikal lungless frog (Apulmorana baikalensis)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: Baikal lake, depths up to 100 meters.

Picture by Alexander Smyslov
Baikal lake is original in many respects. This lake is one
of the most ancient on the Earth, and simultaneously the deepest one, over
one kilometer deep.
The lake is located in rift zone, and its coasts gradually move apart. It
explains the significant term of existence of lake. The fauna of Baikal is
very original,
including the set of endemic species, and at the top of food pyramid there
is one of few relic species of cetaceans – Baikal hardbeaked
dolphin (Delphinapterops odontorostrum).
Among small species of the vertebrates inhabiting the lake, the frog about
10 cm long is one of the most original inhabitants. It differs externally
only a little from the majority of typical frogs, except for the wrinkled
skin freely
fitting a body, and hair-like outgrowths appearing on body in courtship season.
Such attributes are present also at other species of frogs, but the Baikal
species differs from them, living in depth, constantly keeping in benthonic
layer of water and having lungs reduced in great degree and lost respiratory
function. It is Baikal lungless frog. Its ancestor is any kinds of numerous
genus Rana, to which this species is very close. Most likely, it descends
from the representative of group of pond frogs, which are more connected
to water,
rather than brown frogs.
Baikal lungless frog has typically frog-looking appearance, not showing any
neotenical features connected to aquatic way of life. It is externally similar
a little to African clawed frog (platanna, genus Xenopus), has small eyes
directed upwards and flattened body. Back of this amphibian is glaucous,
and belly is
silvery. Due to camouflage colouring it easily hides from predators at the
bottom, among Baikal sponges and bottom sediments. As it lives in a zone
where the sunlight is strongly weakened by thickness of water, sight does
not play
in its life such great role, as at ground species. Eyes of this frog are
poorly advanced and are directed upwards and in sides; it sees almost nothing
right
in front of itself. But at this species the lateral line, characteristic
for tadpole stage, is kept during the whole life. Baikal lungless frog creeps
at
the bottom, occasionally rising above the bottom to some meters. In thickness
of water it swims with the help of movements of hind legs, like other frogs.
This frog has strictly skin breathing. The Baikal water is cold and rich
in oxygen, therefore it completely provides needs of animal for oxygen. Lungs
of this species are reduced and filled with fat, but on stomach skin is wrinkled
for increase of surface. In courtship season when frogs become more active
and require more oxygen, at their back and rear legs the fringe of hair-like
skin outgrowths appears.
These frogs live at the depths up to 100 m, eat invertebrates and fish fry,
searching for them with the help of sense of smell and bodies of lateral
line. This frog is capable to swallow fish of size almost equal to itself.
For this
species cannibalism is frequent – adult individuals eat tadpoles and small
individuals recently had gone through metamorphosises. Frogs become prey
of fishes, and large scuds can eat wintering individuals.
Baikal lungless frog winters in thickness of bottom sediments or in narrow
cracks of rocks where the predatory fish can not penetrate. Frequently tens
individuals of approximately equal size gather in convenient places for wintering.
Wintering frogs can move down deeper – up to 150 and even up to 200 meter
depth.
Breeding of Baikal lungless frogs begins in spring when the surface of lake
turns free of ice. At this time the contents of oxygen in water is increased
and it gets warm a little. Males search for females with the help of sense
of smell. Having found out the female ready to spawning, male seizes her
with forepaws and swims with her, driving others males away from her. These
amphibians
spawn eggs in colony of Baikal sponges where it is better aerated due to
the currents of water created by sponges. The incubation lasts from two up
to four
weeks, depending on temperature of water. Young tadpoles also live among
sponges where they eat tiny invertebrates and silt. Growing older, they leave
bottom
and form schools far from the coast. Tadpoles up to 6 – 8 cm long including
tail live closer to surface, rather than adult frogs. They eat fish fry and
small species of pelagic scuds. Their metamorphosis takes place at the second
year of life, and even at the third year in cold years. Because Baikal lungless
frog lives in cold water, it grows slowly, and its life expectancy reaches
50 years.
This species of amphibians was discovered by Anton, the forum member.
Jelly-making
frog (Mucobatrachus glutinosus)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: The Far East, East Asia, rivers and lakes.
In the process of evolution, animals are constantly perfecting weapons of attack
and defense. Sometimes the same type of weapon appears independently in representatives
of completely different groups of animals. For example, in the course of evolution,
saber-like canines appeared independently in different families of mammals and
mammal-like reptiles. Similarly, representatives of primitive jawless hagfishes,
known back in the human era, as well as tailless amphibians of the Neocene epoch,
acquired similar weapons independently. This weapon is a slime that can swell
strongly in the water, preventing the predator from catching the animal.
The jelly-making frog is a predominantly aquatic frog species inhabiting freshwater
reservoirs of East Asia in temperate and subtropical latitudes, which uses slime
weapons. In its appearance, this frog differs little from the species of the
Rana genus known in the human era: it has only a larger head and strong hind
legs with elongated toes connected by an extensive swimming membrane. The skin
of the jelly-making frog is smooth, green in color with brown spots along the
back and numerous small brown specks on the limbs from the elbow and knee to
the digits. The belly is light yellow or white. The male differs from the female
in smaller sizes (up to 10 cm long, female – up to 12 cm) and a more saturated
belly color.
Courtship games in this amphibian species begin in spring, when the water level
in rivers and lakes rises. Males get a bright courtship dress: the belly becomes
lemon yellow, and the back is emerald green with black spots. Males utter mating
calls very similar to the croaking of frogs of Rana genus in the human era.
Having found the female, the male hugs her with his front limbs and swims with
her for a while. The spawning begins at dawn. Clutches of this species represent
long cords of eggs packed in a dense slimy mass and containing up to 50 thousand
eggs. Parents don’t care about the clutch. Incubation lasts about 10 days. Tadpoles
of this species keep in shallow water among plants in large shoals.
This frog species has numerous mucus glands located in the skin. If an adult
frog is captured by a heron or another bird, the amphibian’s skin becomes covered
with a very slippery slime, and the frog slips out of the predator’s beak and
falls into the water. In the water column, a predator like a catfish or a snakehead
catching jelly-making frog faces another result of the action of this slime:
it swells strongly in the water, and clots of slime clog the gills, preventing
the fish from breathing. The slime secreted by this amphibian contains a small
percentage of proteins that can swell strongly in water, turning it into a viscous
slimy mass. The slime secreted by tadpoles has exactly the same properties:
having felt the “smell of fear” (substances from the damaged skin of a relative)
in the water, the tadpoles simultaneously emit slime, turning significant areas
of water into a viscous mass resembling jelly. This method of self-defense may
well stop and kill a predatory insect, blocking the access to the surface of
the water for breathing for it. In addition, small fish, getting into such a
mass, just suffocates. The tadpoles themselves quickly “dump” the slimy mass
from their bodies and escape outside the protective slime barrier.
Metamorphosis ends completely at the age of 3 months, but the frogs almost never
leave the water. Only when the native reservoir shallows, they come to land
and search for a new place to live. Sexual maturity in young animals comes at
the age of 2 years, and life expectancy can be up to 15 years.
Island
lithobates (Lithobates minor)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: freshwater reservoirs of the island of Great Antigua.

Picture by Pavel Volkov
Neocene is a time when new species and families of animals,
fungi and plants came on the scene and conquered the planet, but representatives
of the nature of the Earth who survived in the historical era also did not always
give up their positions. They continued to evolve, and even colonized new ecosystems.
In the Holocene epoch, the American bullfrog (Lithobates catesbeianus) was one
of the most common amphibian species in North America. Large size, voracity,
and relative undemanding to the habitat allowed this amphibian to spread throughout
the eastern half of the United States and penetrate far to the west. Human activity
and immunity to chytridiomycosis, which killed many tailless amphibians, benefited
the bullfrog and allowed it to expand its range and get rid of competitors.
The Holocene-Neocene glaciation forced the part of the bullfrog population to
settle to the south, including to the Caribbean islands, where they settled
via the land sections of the Antilles land bridge formed at that time. When
the ice age ended, part of the bullfrog population remained in the south, cut
off from North America by saltwater straits insurmountable for amphibians. In
the conditions of island isolation, the bullfrog persisted and evolved into
new species, including the island lithobates.
The island lithobates is a relatively small ground-dwelling amphibian, not exceeding
9 cm in length. It resembles its ancestor in body shape, but its back and the
outer side of the legs are darker, almost black, the head is yellow, and the
belly and inner side of the legs are brown in females and reddish in males.
This coloration makes the island lithobates a more noticeable species, but since
it lives on islands where predatory mammals and birds are less numerous than
on the mainland, and its main predators, aquatic snakes and turtles, do not
have color vision, this mutation was able to fix in the new species without
harm. This frog feeds mainly on invertebrates, less often on small vertebrates,
which it can overcome by attacking from an ambush. Cannibalism is common for
this species, and a significant part of the young die in the jaws of their adult
relatives.
The preferred habitats of this species are humid evergreen forests and thickets
of large moisture-loving grasses.
The island lithobates has no pronounced breeding season, but prefers to spawn
after heavy rains. Then the males of this species gather in large groups near
freshwater reservoirs – ponds, small lakes and rivers – and begin to croak loudly,
attracting females from afar. This species is even more louder-voiced than its
ancestor was, and the voices of males can be heard from afar. The females come
to the place, begin spawning eggs, and the males rush to the female in a group
and fight right on her back for the right to fertilize her eggs. As a rule,
the strongest male wins, but it happens that the females die in all this turmoil.
The eggs of one female may be often fertilized by several males at once.
The eggs of this species are pelagic, floating freely in the water column in
well-warmed shallow waters, and after some days (on average after a week), tadpoles
hatch out of them, similar to the tadpoles of the ancestral species, except
for a smaller size. Like many tropical amphibians, the larvae of the island
lithobates develop quite quickly, and within one and a half to two months, they
are ready for metamorphosis. Sexual maturity comes only after a year. The maximum
life expectancy of the island lithobates is 8-10 years.
This amphibian species was discovered by Bhut, the forum member.
Yalubba
(Lithobates yalubba)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitats: reservoirs in the subtropical forests of North America in the southeastern
United States.

Picture by Wovoka
In the animal world, the phenomenon of neoteny is widespread:
the larval stage gets the ability to reproduce without starting metamorphosis.
Among vertebrates in the Holocene, this phenomenon is demonstrated by some tailed
amphibians: mole salamanders (Ambystoma), olm (Proteus) and sirens (Siren).
In the human era, such a phenomenon was not known among tailless amphibians.
However, by the Neocene in various parts of the world, the tailless amphibians
evolved, that developed a similar feature independently of each other: branchial
tadpole in South America, trapmouth
frog (partially) in Africa, and in New Zealand a whole new family arose,
New Zealand false
salamanders.
The same process had taken place in North America with the descendant of the
American bullfrog (Lithobates catesbeianus). Yalubba is a descendant of the
American bullfrog that has completely lost the ability to metamorphose, i.e.
to transformation to the adult stage (it lacks even limbs), and has passed to
reproduction at the larval stage. The name “yalubba” means “tadpole” in the
language of the Choctaw Indians. It reaches a maximum size of 20 cm and lives
in various reservoirs in the subtropical forests of the southeastern edge of
North America – east to the swamps of Florida, to the northeast of the Mexican
Plateau. The settling of this animal across small ponds and lakes takes place
due to shore birds and waterfowl, on whose legs the eggs of a tadpole frog can
be carried. Yalubba has an olive-brown color with small yellow (sometimes black)
spots and a white or yellowish belly. Individual coloring may partly vary depending
on the prevailing color of the bottom of the reservoir.
The amphibian’s mouth is quite wide, capable of stretching greatly and swallowing
large prey. Yalubba is a predator that feeds on fry, tadpoles, small frogs,
small crustaceans and insect larvae. Young individuals feed on algae films.
The breeding season begins in spring and lasts until the end of summer; the
peak in reproduction falls at the beginning of summer. In the north of the range,
there is usually only one spawning, in the south, two spawnings per season are
common. The animal leaves the clutch among the aquatic vegetation. During the
breeding season, males occupy areas of thickets of underwater plants at a depth
of up to 1 meter and protect their own water area from rivals. Skirmishes occur
at the boundaries of the individual territories: males push each other with
their heads, sometimes striking the opponent hard. At this time, the color of
the male’s body becomes more contrasting and a jagged dark border appears on
the fin plica. The width of the border is determined by the content of sex hormones,
and females prefer males with a wider border. Males guard only the territory
of the nest, but do not take care of the offspring.
The male fertilizes all the eggs that the females spawn on his site. Each female
spawns up to 1000 eggs packed in long slime cords, which are easily torn. At
the first stage of embryonic development, eggs are resistant to drying – at
this time they are easiest to transfer through the air on the legs of water
birds. Translucent larvae hatch after 15 days. If they escape the predators,
they reach sexual maturity at the age of 3 years. The life span of this species
can reach up to 16 years.
This amphibian species was discovered by Wovoka, the forum member.
Halonlabi
(Halonlabi caecus)
Order: Tailless amphibians (Anura)
Family: True frogs (Ranidae)
Habitat: North America, karst cave systems in the Appalachians.
In the Neocene, in the Appalachian Mountains, there are systems of ancient karst
caves stretching underground for tens of kilometers. This is an extreme place
to live, but some caves turn to a home for the communities of very peculiar
animals specialized to such a habitat. Organic substances get into these caves
due to colonies of bats, and in some cases, they are brought by rivers from
the ground surface. During the day, when bats rest in caves, hanging upside
down, they fall prey of peculiar local predators, which include the blind Appalachian
halonlabi cave frogs. These absolutely transparent frogs are descendants of
the American bullfrog (Lithobates catesbianus), which passed to the cave lifestyle;
the name of this frog means “bullfrog” in the language of the Choctaw Indians.
After millions of years of living in dark caves, they have completely lost their
sight, but have got an excellent sense of smell and hearing, as well as spatial
memory. Their main diet is bats, which they catch, moving freely along the cave
roof due to powerful suction cups on their limbs. Each individual remembers
well the “map” of its possessions and convenient routes to bat colonies. Animals
have translucent skin, through which the insides, bones and muscles are visible.
Although these frogs have completely lost their sight, they still have eyes,
although they perform an unusual function. The retina of the eyes has long degraded
and does not perceive light, the vitreous body has become dense, and the eyes
themselves are covered with skin from the outside and protrude above the surface
of the head like two tubercles. They are needed exclusively for pushing prey
into the esophagus.
In small caves, where the number of bats is lesser, halonlabis have to be content
with cave mosquitoes and other insects, catching them with the help of hearing
and smell. They feed rarely, but to the full: in one hunt, the halonlabi is
able to eat up to 5-7 individuals of small bat species. Once full, the frog
may not eat for up to 3 months, digesting its prey and storing nutrients in
the body. When the frog stores enough resources, the instinct of reproduction
activates. Well-fed males and females descend to the cave reservoirs and begin
to call for more and more frogs. In this species, females are also able to utter
a semblance of a courtship song, but they do it only when they do not hear the
voices of males. Males utter loud humming sounds, amplified by the cave echo.
Mating songs serve as a powerful incentive to gather adults for reproduction.
After some time, dozens of halonlabis ready to spawn gather from all over the
cave, and a rough skrimmage begins: copulation of one and all. The eggs of one
female (up to 5000 eggs) spawned in water can be fertilized by several males
at once. In huge systems of karst caves, where tens of thousands of bats belonging
to several different species live, it repeats every week for a whole year due
to the even climate in the caves almost all year round. In smaller caves, where
the number of fodder bats is low, or they fly away for the winter, frogs are
forced to be more content with insects, and such celebrations of life occur
much less often.
After tadpoles appear from eggs in cave reservoirs, they begin to live their
own independent life. However, there is very little food for them, so they are
almost omnivorous, and their development is greatly delayed. Their main food
is bat droppings, adult halonlabi eggs, smaller tadpoles, as well as larvae
of cave mosquitoes. They also feed on scarce groundwater plankton, consisting
mainly of small crustaceans. The favorite dish of tadpoles is the bat corpse:
when it gets into the water, a shoal of tadpoles rushes to it, and in less than
half an hour only the skeleton remains from it. They also eat the corpses of
adult halonlabis that could not withstand the stress of the breeding season.
Under favorable conditions, the development of the tadpole in halonlabi lasts
up to 3-6 months. In small caves, where the food is scarce, development can
delay up to 1 year. Halonlabis become sexually mature at the age of 8-9 years
(six months earlier in females). In small caves, the growth of animals slows
down greatly, and sexual maturity comes at 12-14 years. If the halonlabi grows
to the stage of an adult frog (only some tadpoles from the brood manage to do
it), it will be able to live up to 60-70 years. Significant longevity of this
species is associated with stable living conditions in caves and a slow metabolism.
Halonlabi is a very polymorphic species: despite their similarity, over millions
of years of living in different caves, different populations of this frog have
accumulated differences from each other, and some of them have lost the ability
to interbreed with each other. Halonlabis living in karst cave systems, where
there is a plenty of fodder bats, grow very large (larger than their bullfrog
ancestors) – up to 30 cm long, and are able to gain weight up to 700 grams.
However, body of such frog is more muscular compared to its bullfrog ancestor,
because it needs to perform constantly acrobatic tricks when crawling on the
walls and roof of caves. The frogs that live in smaller caves, where the bat
population is little, are much smaller: the smallest halonlabis grow up to 15
cm in length and weigh about 100 grams. For such small individuals, one bat
is enough to eat to the full. Populations from different caves exchange genes
due to the ability of tadpoles to go without food for a long time and settle
via underground rivers.
This amphibian species was discovered by Wovoka, the forum member.
Crab-eating
quasireptile (Quasireptilia cancrivora)
Order: Tailless amphibians (Anura)
Family: Forked-tongue frogs (Dicroglossidae)
Habitat: mangrove forests and brackish swamps of South and Southeast Asia from
India to the Philippines and from southern China to southern Indonesia.
Several species of fishes appeared in the Neocene, which independently developed
separate features of anatomy and lifestyle of amphibians – for example, the
Antarctic amphibious flounder and froggy
mudskipper. However, by the Neocene, another interesting evolutionary
transformation happened: a kind of quasi-reptile descended from the amphibian
– it is a descendant of the crab-eating frog (Fejervarya cancrivora), which
was the only amphibian of the human era capable of staying more or less a long
time in salty sea water without harm to it.
The crab-eating quasireptile has grown larger than its ancestor: the length
of adult male reaches about 10 cm, and adult female is about 12 cm long; however,
the largest quasireptile specimens can reach a length of 20 cm. It retains a
typical frog appearance, but its skin has become significantly coarsened, which
is why it actually lost skin respiration, having passed to pulmonary respiration
completely. This feature gave the frog the opportunity to reduce significantly
the intake of salt into the body and stay in salty seawater much longer. The
kidneys play a major role in the excretion of salt from the organism. In addition,
a significant amount of urea accumulates in the blood and musculature of this
animal. In addition, there is a protein in the animal’s musculature that resembles
cetacean myoglobine in function – it allows this animal accumulating a significant
amount of bonded oxygen, compensating for the relatively imperfect lungs of
the animal. After searching for prey, the crab-eating quasireptile stays motionless
on mangrove trees for a long time, restoring the reserves of bonded oxygen before
hunting.
The coloration of this frog is of relatively light shade: the back is gray with
some black specks and smears on the limbs, imitating perfectly the texture of
mangrove tree bark, and the belly is light green with large white spots of irregular
shape, making it difficult to recognize the outlines of the animal when viewed
from below. The animal has front legs with prehensile fingers and corneous claws
that allow it to capture soft-bodied prey.
Due to the physiological transformation of the organism, this frog managed to
expand its feeding territories significantly: it was able to feed not only in
mangroves, but also to swim some distance into the sea, having developed the
hunting for small fish, shrimp, crabs and even young octopuses in coastal waters.
The way of parental care has undergone significant changes in this species.
To prevent the offspring from dying in salt water, the frog bears not numerous
eggs, and then tadpoles, in a special brooding pouch on its back (like the marsupial
tree frog of Gastrotheca genus). This is an exclusively female’s occupation.
In the process of reproduction, the male fertilizes yolk-rich eggs laid by the
female on above-water objects (usually on the branches of a mangrove tree),
and then assists the female in placing them into the brooding pouch. In this
species, the larval stage passes inside the egg shell, and the pouch is left
by already formed froglets, up to 7 ones in one brood. The gestation of eggs
and posterity lasts about 5 weeks. Sexual maturity comes at the age of 14-19
months, and life expectancy reaches 20 years.
This amphibian species was discovered by Wovoka, the forum member.
Deafening
frog (Decibellator abalienarus)
Order: Tailless amphibians (Anura)
Family: African reed frogs (Hyperoliidae)
Habitat: forests of Equatorial Africa, forest canopy.
Tree-climbing frogs of the Hyperoliidae family, which are peculiar analogues
of the tree frogs of Eurasia and North America, as well as South American leptodactylid
frogs and poison dart frogs, are very characteristic inhabitants of the tropical
forests of Africa. Hyperoliids are characterized by small size and bright coloration,
often with contrasting spots. Representatives of this family have survived in
the era of climate change, although a significant number of their species have
died out – mainly inhabitants of tropical forests and local species. In the
Neocene epoch, the diversity of hyperoliids was restored.
One species of Neocene hyperoliids is a small tree frog, named as deafening
frog for its loud voice. This animal has a characteristic “frog-like” appearance
with a large head and a blunted muzzle. The body length of the deafening frog
is about 5 cm. It has thin limbs with prehensile digits and relatively large
eyes. The coloration of the deafening frog is soft – green with brown spots
on the sides and transverse stripes on the limbs. When the frog squats with
its legs pursed, the spots on the limbs and sides merge with each other, making
it difficult to perceive the outlines of the animal. The belly of animals of
both sexes is milky-white or yellowish. The eyes have a golden iris and a pupil
looking like a horizontal slit. At night, the pupil opens and has a diamond
shape. The male differs from the female in a more slender body and a bright
red throat, which color is especially noticeable when the frog croaks. The female
has a white throat.
Deafening frog is characterized by a very loud voice. The calls of males, resembling
the dog barking, represent one of the distinctive features of the rainforest.
The male calls more actively in the evening, but is also active in the morning,
although he calls less actively.
Deafening frog feeds on small insects living in the tree crowns.
This species is characterized by an unusual parental care. The deafening frog
does not descend to the ground for reproduction, using the water sources available
in the trees. This species lays eggs in the water-filled cup-shaped fruit bodies
of a fungus called a “frog’s cup”
and formed a symbiotic relationship with this fungus. Tadpoles develop in these
improvised mini-reservoirs protected by the male. Usually the male keeps on
the outer side of the mushroom near the edge, head up. He drives away from the
chosen mushroom any competitors, who can eat a clutch or tadpoles while occupying
the settled mushroom. Defending the offspring from larger animals (birds, small
mammals), the male utters a loud, hard-to-bear sound. If necessary, he bites
and behaves aggressively even towards animals such as monkeys.
In the clutch of this species there are up to 15-20 eggs, which the female lays
in the water accumulating in the fruit body of the fungus. Spawning repeats
on average every month. For spawning, the female chooses only young fruit bodies,
determining their age by the quality of water in them. Tadpoles hatch 24 hours
after fertilization of eggs. They feed on the secretions of the fruit body of
the fungus and have a mouth of scraping type. During the feeding, they swallow
the spores of the “frog’s cup”, and the fungus infects them for a while. Tadpoles
undergo metamorphosis at the age of 2 weeks and leave the fruit body, which
collapses soon. The male remains near the fruit body, because in good conditions
the fungus soon forms a new one, where a new portion of eggs can be spawned.
For about two weeks after metamorphosis, young frogs cope with the fungus, but
during this time they manage to spread it along the branches with their droppings.
At the age of 6-7 months, they become sexually mature. The life expectancy of
this species does not exceed 5 years.
Orange
frog (Mantella madagascarensis)
Order: Tailless amphibians (Anura)
Family: Malagasy poison frogs (Mantellidae)
Habitat: tropical forests of Madagascar.
In nature, similarity does not always mean kinship; among its repertoire there
is the phenomenon of convergent evolution, when unrelated or only distantly
related families, genera and species develop superficial similarities, sometimes
very large. So, in the American tropics there are poison dart frogs (Dendrobatidae),
while in Madagascar there are Malagasy poison frogs, very similar to poison
dart frogs outwardly, but much more closely related to the Asian families of
tailless amphibians. In the human era, this endemic family demonstrated many
examples of covergence with representatives of other frog families of the rest
of the world. Their diversity fell at the turn of the Holocene and Neocene,
but recovered after the stabilization of natural conditions. Among the Neocene
representatives of this family, the orange frog is notable.
Like other mantellids, it is a fairly small amphibian, no more than 2-3 cm long.
It has a solid, reddish-orange color (hence the name). Such a bright, warning
color deters most predators, since this frog is poisonous, but some local species
of solitary wasps have developed a certain resistance to its venom and have
become parasites of this species. The orange frog feeds on various small invertebrates,
and its tadpoles are vegetarians. This frog species is a good tree-climber and
is found at an altitude of up to 20 meters above the ground; these amphibians
almost never descend to the ground.
The mating season of the orange frog takes place during the rainseason. At that
time, the males search for suitable reservoirs – as a rule, these are local
species of epiphytic plants growing on trees and accumulating rainwater in their
rosettes (usually ferns or epiphytic ladies’-slipper orchids) – and then they
resound their possessions with loud calls, attracting females and driving away
rival males. If the latter are not afraid and challenge the owner of the territory,
then a struggle begins, during which both frogs try to knock each other down
and throw the rival off the branch. The loser leaves the battlefield, and the
winner gets the plant. The females, who arrived at the call, examine the plant,
and if they like it, they stay with the male. These amphibians are monogamous
and live in peculiar families, where there is a division of responsibilities
for raising offspring: females leave fertilized eggs, from which tadpoles hatch,
in the water and then lay there unfertilized ones, which these tadpoles feed
on. At the same time, males drive outsiders and potential enemies from the territory.
In addition to unfertilized eggs, tadpoles eat microscopic algae and a film
of unicellular animals. After some weeks, the tadpoles turn into froglets, leave
their parents, and the families break up. Nevertheless, these frogs are more
stay-at-home than wanderers, and do not go far from habitable plants at all,
so adults of this species very often reunite old pairs than start new ones.
During the year they have 2-3 breeding cycles. Sexual maturity comes at the
age of 8-9 months.
The lifespan of an orange frog is less than 10 years, usually up to 5 years.
This amphibian species was discovered by Bhut, the forum member.
Erlik’s
frog (Erlikobatrachus sibiricus)
Order: Tailless amphibians (Anura)
Family: Afroasian tree frogs (Rhacophoridae)
Habitat: Siberia, taiga lakes, rivers and swamps.
During the Holocene epoch, Afroasian tree frogs were one of the most successful
amphibian families. They thrived mainly in South Asia, from rainforests to mountains.
However, people have caused severe damage to these frogs – the destruction of
habitats and pollution of reservoirs have destroyed many local species. The
pied warty frog (Theloderma asperum) was among the few surviving species. These
small frogs adapted successfully to the moderately warm and even cold mountain
climate, and during the restoration of the fauna, their descendants began to
spread to the plains, including north of the Himalayas. The ice age was not
a serious problem for them; on the contrary, it helped them to “cast away” the
competition, because the populations of thermophilic species declined in places,
and the descendants of Theloderma quickly occupied their niches. And it was
during the glaciation era that one of the most interesting species of plain-dwelling
thelodermas appeared on the northern tip of the range of the family – the Erlik’s
frog.
Erlik’s frog is a small (5-7.5 cm) tailless amphibian, similar in anatomy to
its ancestor. It lacks suckers on its digits – unlike other thelodermas, Erlik’s
frog does not climb trees, and lives exclusively on the ground and among stones.
The skin of this frog, like that of the ancestor, is bumpy and not smooth, providing
it an excellent camouflage. The Erlik’s frog mimics simply the forest litter,
and in addition this frog partially changes its coloration depending on which
substrate it squats on. However, in this species, the camouflage is not limited
to coloration change – the Erlik’s frog has much more interesting “decorations”.
Both sexes have skin horns (hence the name in honor of Erlik – the death deity
from Altai mythology); male’s ones are much longer than female’s. These horns,
like the outgrowths on the back, are formed of the skin. They grow right above
the eyes and have a flattened shape with a pointed tip (like at Asian horned
frogs). Their color is usually grass green in spring, rich green in summer and
dull rusty in autumn. The horns help frogs hide their body shape and mimic the
fallen leaves.
The behavior of the Erlik’s frog is unusual. It is a nocturnal animal, so it
is inactive during the day: usually it spends the daytime huddled in moss or
fallen leaves, and in the evening it goes hunting. It feeds on insects and other
invertebrates, which it catches on land and in water – this amphibian is also
a good swimmer. On rainy days, it prefers to stay on land, and on dry days,
even during the daytime, it sometimes keeps in some shelter in a pond – more
often among the stems of marsh plants in the coastal zone. In case of danger,
it clings to the ground and changes its coloration, hiding from the threat.
If the predator found it, this frog can quickly jump away from it or escape
in the water.
This species overwinters in natural caves in the ground, as well as in rodent
burrows, burrowing to a depth of 2 meters. Up to several dozen frogs can winter
in one shelter. Shelters are used from year to year, and frogs remember their
location well. Young frogs find the shelters of adults by smell, and join them
for wintering.
The breeding season at Erlik’s frogs begins in mid-May. At this time, they gather
in large reservoirs, where males attract females by croaking, sounding like
“rrrrrrrrrrkikiki”, changing their coloration to bright orange with red spots.
The female spawns about 20 eggs at a time on the stems of marsh plants low above
the water, and the male fertilizes them. The cluster of eggs is covered with
slime capsule; the tadpoles hatch after 7-10 days, fall into the water and form
a school, hiding from enemies among plants. The spawning repeats 3-4 times within
a month after the first clutch. Tadpoles are omnivorous, eating various foods
– from algae to fish corpses. Metamorphosis begins after about 30 days. The
froglets reach sexual maturity at the age of 2 years. Life expectancy is up
to 12 years.
This amphibian species was discovered by Dohlosavr, the forum member.
Moss-mimic
frog (Cryptorana bryomima)
Order: Tailless amphibians (Anura)
Family: Afroasian tree frogs (Rhacophoridae)
Habitat: mountains in South Asia, karst caves, bamboo forests.
During the Holocene epoch, the rhacophorid genus Theloderma experienced a crisis
due to habitat reduction. Many local species became extinct, species from tropical
forests disappeared also, and among the few survivors there was pied warty frog
(Theloderma asperum), which survived the climatic cooling. In the tropics of
Asia, its descendants live – an extensive genus of small hiding frogs (Cryptorana).
Like its ancestors, the moss-mimic frog is an inhabitant of mountains and caves,
where it settles in areas overgrown with shade-requiring vegetation. Some individuals,
due to their small size, settle even in water-filled stalks of bamboo and other
large graminoids. Like the ancestral species, the moss-mimic frog escapes from
predators by skillful mimicking of the moss inedible for them. An adult of this
species is about 2.5-3 centimeters long, has an elegant build, a narrow body,
long hind limbs and greenish-brown skin with a bluish tinge on the belly. On
the head, there are large orange eyes. On its back, the skin is rough and has
small outgrowths resembling the tops of moss shoots. Blood vessels enter these
outgrowths, and due to it the animal satisfies a significant part of the oxygen
needs via skin respiration. The eyelids are also covered with bumps and growths
along the edges, resembling eyelashes, and in this way the animal masks brightly
colored eyes.
This species is terrestrial and lives on rocks overgrown with moss, escaping
from enemies due to an excellent imitation of the substrate surface. The moss-mimic
frog feeds on small insects like midges, also eats spiders, snails and slugs.
It breeds once a year, spawning eggs in water-filled karst caves. To spawn eggs,
these frogs gather in large groups and migrate to reservoirs. The male’s mating
call sounds similar to “pirrrr-pic-pic-pic-pic-pic”. The female spawns 100-120
eggs; the parents do not guard the eggs. Tadpoles hatch 2 weeks later and feed
on microorganisms (such as infusoria) and mold fungi growing on organic debris
got into the caves. Development is slow; metamorphosis takes place 2 months
after hatching. Winters are cold in the habitats of this species, so frogs winter
in caves – usually in the same ones where bats hibernate. The development of
this species is slow; frogs become sexually mature in 4-5 years. The life span
reaches 25 years.
Related species live in the mountains of South Asia:
Leaf-mimic
frog (Cryptorana foliomima) is also a mountain dweller, but it lives
lower down the slopes, in forests. It has outgrowths on the back and above the
eyes that make the animal’s back look like a leaf. The color changes throughout
the year – in spring it is brown with spots, in summer it is green with yellowish
spots on the edges, in autumn it turns reddish and gradually turns brown by
winter. The length of an adult is 4-5 cm. This species spawns eggs in shallow
reservoirs, and winters in the same places. Outside of the breeding season,
the animal lives in trees and feeds on insects. The male’s call sounds like
“peeee-rr-rr-rr-peeee-peeee”.
Stone-like
frog (Cryptorana lithops) lives in mountainous areas side-by-side with
moss-mimic one, but spends less time out of the water and lives on the rocky
banks of mountain streams. It has a wider, rounded body 2-3 cm long, gray coloration
and unsmooth skin, resembling outwardly a small stone. Usually it stays in shallow
water or at the edge of the water, pursing its limbs and imitating a stone.
This species feeds on insects. Breeding and wintering occur in streams. Tadpoles
stay on the underside of the rocks, where the current is weaker, and feed on
sessile invertebrates. Wintering takes place in the non-freezing part of the
streambed. The mating song of the male sounds like “peeee-peeee-peeee-prr”.
This amphibian species was discovered by Allosaurus12, the forum member.
Bush-bearer
frog (Ctenodorsa santhamnos)
Order: Tailless amphibians (Anura)
Family: Afroasian tree frogs (Rhacophoridae)
Habitat: Southeast Asia, Jakarta Coast, tropical forests.
One of the evolutionary trends in the Afroasian tree frog family of the Neocene
epoch was aimed at camouflage improving by developing various skin outgrowths
on the body surface, designed to give the animal a resemblance to objects inedible
for predators. Some members of the family have skin outgrowths on the body that
enhance their camouflage. A similar adaptation developed independently in giant
mossy tree frog (Bryodorsa titan) from East Asia, unrelated to rhacophorids,
but these formations are less specialized. In some rhacophorids of the Neocene
epoch, the development of these formations reached an extreme degree.
One of the most bizarre amphibians of the Neocene is the bush-bearer frog from
the tropical rainforests. Skin on its back and limbs is covered with numerous
long dense outgrowths forming a number of ridges, which make it very difficult
to notice the animal itself, and even more so to identify it as a frog. In the
bush-bearer frog, these structures perform several important functions at once.
Firstly, they significantly increase the area of the animal’s body surface,
allowing it to receive maximum oxygen from the humid tropical air. Secondly,
they provide an excellent disguise – the brown color of the body and the green
ridges very much resemble a small plant or moss thickets, and also distort the
silhouette of the creature beyond recognition. Thirdly, in this way this amphibian
protects itself from enemies, since damage to the outgrowths is not fatal for
it – due to the muscle fibers inside the outgrowths, they contract when damaged,
preventing blood loss. In addition, with the help of outgrowths, this frog regulates
the temperature of its body very effectively, exposing them to feeble wind or
sunlight, and holding a layer of air around the trunk at other times. However,
they greatly interfere with movement under water, because, even when pressed
against the body, they noticeably disrupt the flowability. For this reason,
the bush-bearer frog leads a terrestrial lifestyle, and hunts from ambush –
it waits until the prey comes within jumping distance, sharply squeezes the
crests and jumps, catching the prey with its short front paws and jaws. Having
a length of 10-15 centimeters (the female is larger), it is dangerous only for
large insects and small invertebrates. During the breeding season, some of the
bush-bearer frog’s outgrowths get a bright red or orange color, without breaking
the camouflage as a whole, but becoming quite noticeable for mates.
Large eyes have a yellow iris and a silt-like horizontal pupil. There are bright
red-orange spots on the sides, hidden at the squatting frog with its hind legs
pressed to the body. The limbs are elongated and have prehensile digits equipped
with suction cups. The hind legs are much longer than the front ones. Animals
are good at climbing branches and tree bark. Due to suckers, they even climb
trees with smooth bark.
The breeding season in tropical rainforests is not pronounced, and these frogs
make two full-scale clutches per year. In monsoon forests, it begins with the
rainy season, and there is only one spawning per year. The male in the courtship
dress calls the female, uttering mating songs that sound like a long-repeated
rhythmic ticking. This species has developed parental care: the female bears
its eggs on the back. Fertility is up to 60-100 eggs. The male fertilizes eggs
spawned on a branch or among moss, and smears a lump of eggs enclosed in the
slimy mass on the female’s back. The eggs fall between the skin outgrowths,
stick to them and incubate on the mother’s back for 9-12 days. Hatched tadpoles
eat the shells of eggs and attach with their mouths to the female’s skin. Feeling
the movements of the tadpoles on her skin, the female descends to the ground
and moves into the pond, where the tadpoles leave her back. Metamorphosis begins
at the age of 3 months, and at the age of 5 months, the growth of camouflage
outgrowths begins in a young frog. Sexual maturity comes at the age of 16-20
months, life expectancy is up to 15 years.
This species of amphibians was discovered by Mex, the forum member.
Sticky
ditch frog (Glutinodorsum ruderophorus)
Order: Tailless amphibians (Anura)
Family: Southern frogs (Leptodactylidae)
Habitat: South America, Chaco, grasslands and shrubs.
Frogs of the leptodactylid family are widespread in South America and occupy
a variety of ecological niches, replacing true frogs on this continent. In the
process of evolution, some species have developed peculiar adaptations for survival.
Being non-poisonous, these amphibians escape from predators with the help of
a kind of camouflage: representatives of this genus carry debris that they gather
in their habitats. In all representatives of this genus, the sides and the back
of the body are surrounded by a characteristic skin fold that greatly expands
the area of the back and hides the front and partly hind limbs of the squatting
animal with its edges. The upper side of the body has very sensitive skin and
its surface has a lot of glands; highly developed skin glands secrete a sticky
and very viscous slime that hardens in the air into a rubbery mass. To this
slime, the frog glues fallen leaves, sand, bird feathers and pieces of tree
bark, providing camouflage for the animal. The animal’s head is greatly expanded,
and the edge of the skin fold bordering the trunk covers the eyeball from above.
In the bushes and among the thickets of grass near reservoirs, one of the usual
species of the genus lives – the sticky ditch frog. The body length of this
species is up to 12 cm; the female is larger than the male. The background color
of the upper side of the body is brown, and beige-yellow below the skin fold;
the belly is yellowish-white with a darker marble pattern. The male’s eyes have
a peculiar bright green color, and in females they are light yellow. This species
camouflages itself by sticking leaves and stems of graminoids and sedges, as
well as fallen leaves of trees, on its back. The hind legs of this species are
characterized by mobility, with their help the amphibian places debris on its
back for camouflage purposes. Usually, the frog crawls under the object of its
interest, secretes some slime and presses the object against it, holding it
for several minutes. Slime is secreted in places contacting with the object:
the stimulus for such reaction is skin irritation. In case of danger, the frog
clings to the ground and closes its eyes. With a direct threat to life, the
frog can escape by making long jumps. The camouflage needs constant renewal:
during heavy rain or in water, the slime liquefies and the debris peels off.
Also, the frog has to re-camouflage itself after molting.
In the breeding season, coinciding with the rainseason, males occupy the shores
of reservoirs and utter plangent rumbling trills, attracting females. Usually
their competition comes down to attempts to drown out the voice of an opponent
or push an approaching female away from his territory. The peak activity of
males takes place at night, but some individuals still vocalize after sunrise.
This species has up to 200-300 large eggs in the clutch; adults spawn eggs in
water and do not take care of them anymore. Incubation lasts about a week. Tadpoles
feed on algae films; their development lasts about 50 days. Young frogs have
a noticeable skin fold on the sides of the body by the time they come to land.
Sexual maturity comes the next year; life expectancy is about 10-13 years.
Sand-carrying
ditch frog (Glutinodorsum crispus) lives in temporary reservoirs in
the river valleys of the subtropical part of South America. It is one of the
most drought-resistant species of the genus, able to withstand the dry season
for 3-4 months (at this time it switches to a nocturnal lifestyle). The upper
surface of its body is colored brown, the fringing skin fold is wavy and scalloped;
the sides and underside of the body are grayish-yellow, the belly is lighter.
The iris of the eye is golden brown in animals of both sexes. This species camouflages
itself by bestrewing sand and lumps of ground over itself. Reproduction takes
place in seasonal reservoirs.
Several species of ditch frogs live in forests. These are the smallest and most
moisture-loving species of the genus:
Leaf-sticker
(Glutinodorsum foliiphorus) lives in the forest floor of mountain rainforests,
avoiding places flooded during river overflows. The body length of an adult
is about 8 cm; the male is smaller than the female, but has a wider back. The
main body color is green, but the edges of the body and limbs are colored brown
with separate rusty spots. This species glues the fallen leaves of trees on
itself, clearly preferring large wide leaves that cover the entire body of the
animal. This species has a remarkable parental care: the female “smears” the
mass of fertilized eggs on the back of the male, who covers it with a tree leaf,
masking and protecting it from drying out. The male carries eggs on land; in
dry weather, he stays close to water sources and dabbles the clutch with water,
scooping it with one of his hind legs and pouring it into the gap between the
back and the leaf glued to it. Incubation lasts up to 10 days and hatched tadpoles
stay on the male’s back for about a day. At this time, feeling the movements
of the tadpoles on his back, the male returns to the water and drops the tadpoles
into the water with one of his hind legs. This is where his parental care ends.
Moss-sticking
ditch frog (Glutinodorsum inconspicuus) inhabits the canopy of a tropical
forest and prefers to stay on the branches of large trees overgrown with epiphytic
plants. This species is up to 10 cm long and has a slimmer emerald-green body
with a white belly and golden eyes. The lateral fold of the skin is divided
into a series of scalloped outgrowths, which surface is fitted with numerous
glue-producing glands. It is not fastidious in choosing a camouflage and willingly
glues its back with both flaccid leaves and butterfly wings or flowers. Long-toed
paws with a highly reduced swimming membrane help this frog to climb quickly
and easily reach the back to stick the new found debris on it. The male bears
a clutch of eggs on his back, but after hatching the tadpoles, he is forced
to descend from the tree, since the tadpoles continue their development in forest
swamps and slow-flowing rivers. After metamorphosis, young frogs leave the water
and climb trees.

Picture by FanboyPhilosopher
Orchid-carrier, or Sticky gardener frog (Glutinodorsum orchidifer) is the smallest species of the genus: the body length of an adult does not exceed 5 cm. This species lives in the rainforests of northern South America and inhabits the forest canopy. It differs from other species of the genus in its clearly expressed adaptation to the arboreal lifestyle: it has relatively long limbs with tenacious digits. The swimming membrane on the hind legs is highly reduced and reaches only to the middle of the toes. The body color is bright green with slight variations from dark green to yellowish-green; the belly is white. This species is remarkable in its “refined” preferences in the choice of material for camouflage: it gathers fallen bright flowers and petals. Often the frog tears off and places a fresh orchid flower on its back and thus lures the insects it feeds on. It happens that this species ambushes among the inflorescences of epiphytic plants. The male is remarkable in parental care: he carries fertilized eggs on his back and leaves one or two eggs in the axils of bromeliad leaves or in hollows filled with rainwater. Tadpoles feed on small invertebrates.
Panama
poison frog (Boreodendrobates panamicus)
Order: Tailless amphibians (Anura)
Family: Poison dart frogs (Dendrobatidae)
Habitat: seasonally humid forests and rainforests of southern North America,
Central America.

Picture by Wovoka
Like other amphibians of the tropics, poison dart frogs suffered
greatly in the Holocene. Deforestation, pollution of reservoirs and epizootics
of chytridiomycosis have exterminated numerous species of this family. However,
populations with immunity to this fungus appeared among several species, and
it allowed them survival in the pockets of tropical forests. Among the surviving
species there was the green-and-black poison dart frog (Dendrobates auratus),
whose range remained mostly in North America after the formation of the Panama
Strait. After the restoration of natural conditions, its descendants settled
far to the north, and are found throughout North America. One of them is the
Panama poison frog, a small amphibian: the females grow up to 2.8 cm long, males
– up to 1.9 cm.
Like the poison dart frog species of the human era, the Panama one is very poisonous
– even such a large animal as yaraha will die from a small dose of its poison.
It shows a clearly visible bright aposematic coloration. The usual coloring
pattern includes black body with irregular-shaped spots and stripes (each individual
has a unique pattern), changing its color from purple on the head to turquoise
on the back, and yellow-orange “half-toes” on black paws, usually completely
covering only the digits and a part of the hand or foot. Other color morphs
exist also, that have different colors or patterns. Its physique is the same
as that of all other poison dart frogs, features of the anatomy like suckers
on the digits also remained the same.
Like the ancestral species, the Panama poison frog feeds on ticks, ants and
other small invertebrates. It lives mostly in trees, but can descend to the
ground. Activity is mostly twilight. Interestingly, these frogs usually are
not drawn towards swamps and other bodies of water, but in seasonally humid
forests during the drought season, they move closer to the water. These frogs
often live near thickets of ground bromeliads.
In tropical rainforests, these frogs breed all year round, and in seasonally
humid ones only during the monsoon season. Males occupy a certain territory,
choose a place for spawning, drive other males away from it and vocalize, calling
for females. The mating song of the male is a quiet “kikiki-kikiki” with interruptions.
After the appearing of the female, the breeding pair goes to the place of spawning
– usually to the area of wet moss thickets. There, the female lays about 10
eggs, hatching after 15 days. The male guards the eggs before hatching and carries
the tadpoles to the nearest bromeliads or shallow reservoirs. After that, he
stays with them and guards them until they undergo metamorphosis and leave the
pond. It usually happens after 2 months. If the reservoir dries up, the male
can move his offspring to a safe place where there is water.
At the age of 6-9 months, young poison dart frogs are able to breed, and they
live about 6-7 years.
This amphibian species was discovered by a Dohlozavr, the forum member.
Aklaipo
(Herbobates aklaipo)
Order: Tailless amphibians (Anura)
Family: Poison dart frogs (Dendrobatidae)
Habitat: savannas and woodlands on the coast of the Gulf of Mexico.
In the Holocene, most of the poison dart frogs were inhabitants of tropical
forests, and many of their species died out along with the forests cut down.
The restoration of the natural environment in the early Neocene became an incentive
for the active adaptive radiation of the descendants of the surviving species,
and in the Neocene, the descendants of poison dart frogs made an attempt to
occupy a new ecological niche – dry forests and woodlands. Aklaipo is one of
their species. Its ancestor was the green-and-black poison dart frog (Dendrobates
auratus), which lived in Central America in the Holocene and survived thanks
to several populations resistant to chytridiomycosis. In the Neocene, its descendants
settled to the north, having developed the subtropics of North America. In due
course of this settlement, some species adapted to survive in savannas and prairies.
Aklaipo is a typical representative of them (“aklaipo” is the word “frog” in
the language of the Comecrudo Indians). This frog is very large for poison dart
frogs: males grow up to 6.2 cm long, and females are up to 7 cm long. The anatomy
of this species has changed somewhat – the animal has acquired a more robust
physique and is better adapted to jumping. The suckers on the digits decreased,
and the fingers became tenacious, with small corneous pads on their underside.
Aklaipo is poisonous and brightly colored. Most of the color morphs of this
species have a body of a rich green background color with purple-blue spots
on the back and upper side of the thighs, but there are other varieties of coloration,
including large purple spots on the back. Like the poison arrow frogs of the
human era, this frog species does not synthesize its own poison. To obtain poison,
aklaipo eats ants, ticks and other poisonous arthropods.
Usually, aklaipos live along the shores of reservoirs, spending a hot day near
the water, and at night they hunt and often go far from the reservoir. These
frogs are able to climb shrubs and stems of large grasses. Due to changes in
habitat conditions, the breeding mode of this species has also changed. The
males simply gather in the water and call the females with a mating song that
sounds like “kirr-kek-kek”. Each male guards his part of the coast from competitors.
Males often dig up the soft shore, making individual pits connected with the
reservoir. The female spawns about 50 eggs at the male’s territory, and the
male protects them, having pasted to his own stomach. At this time, he also
does not leave the water and constantly drags eggs on his body. After 10 days,
tadpoles hatch, and the father also guards them until they begin to swim on
their own. Metamorphosis begins after 2 months; sexual maturity comes at the
age of 2 years, and aklaipos live up to 15 years.
There are related species:
Uruna (Herbobates
uruna) is a species related to aklaipo, that lives on the watersheds
of rivers in the lower reaches of the Mississippi basin (“uruna” is the name
of the frog in the Tunica Indian language). It lives in reed thickets among
shrubs and has a slightly smaller size than the aklaipo: the length of the female
is up to 6 cm, the male is up to 4.8 cm. The color is very distinctive – an
orange background with 3 blue longitudinal stripes on the back and the spots
of the same color on the limbs, sometimes merging into transverse bands. The
male also bears eggs on his stomach, but does not guard the offspring.
Tlalok
(Herbobates tlalok) is the smallest (females up to 5.8 cm long, males
up to 4.5 cm) species from the group of savanna poison dart frogs living in
the northern part of the Mexican savannas, sometimes entering maquis in western
North America (Tlalok is the Aztec god of rain). The background color is reddish-brown
with bright purple “neon” intermittent stripes on the back, sides and limbs.
It can go a little longer without water than other species of the genus – water
is needed only for raising offspring. The slime is viscous and has a high protein
content; it protects this frog against water loss. The animal gets moisture
from insects and licks dew in the morning. The breeding season begins during
the spring rains; at this time, loud whistling voices of males are heard at
night.
This amphibian species was discovered by a Dohlozavr, the forum member.
Saputka,
or Floridan poison dart frog (Lignobates saputka)
Order: Tailless amphibians (Anura)
Family: Poison dart frogs (Dendrobatidae)
Habitat: subtropical forests of North America on the coast of the Gulf of Mexico
and the Atlantic Ocean (from east Texas to Florida and north to North Carolina).
Neocene is a time of significant redistribution of the areas of various groups
of living organisms. It was partly the result of human activity, which moved
various species to unusual habitats, and partly the result of changes in nature
after the human extinction. An example of the latter scenario was the existence
of poison dart frogs in North America. The ancestor of the North American poison
dart frog clade was the grren-and-black poison dart frog (Dendrobates auratus),
which lived in Central America in the Holocene and survived due to several populations
resistant to chytridiomycosis. The settlement of the descendants of this species
began from the south to the north. At first, Panama poison frog appeared, whose
descendants gradually settled to the north, having mastered the humid savannas
on the coast of the Gulf of Mexico first, where aklaipo
and related species appeared, and later made their way to the subtropical forests
in the southern extremity of North America, where the Floridan poison dart frog,
or saputka lives (“saputka” – frog in the language of the Seminole Indians).
It is a small frog: females grof up to 3 cm long, males are up to 2 cm long.
Saputka lives mainly in trees, but can also descend to the ground in shrubs,
where a humid microclimate persists. The animal retains suckers on its digits,
allowing it climbing smooth leaves and stems. The activity of this species is
mostly crepuscular. Like other species of poison dart frogs, the Floridan species
is very poisonous. The poison is not produced by the frog itself, but accumulates
from food. To get poison, it eats ants, ticks and other poisonous arthropods.
The poisonous properties of the slime are reinforced by a bright aposematic
coloration. This species has a purple back, and the head, limbs and belly are
lurid pink.
The incentive for reproduction is an increase in the water level after rains,
when the forest is flooded. During the mating season, frogs croak loudly on
branches hanging over the water, and eggs are spawned in shallow water near
the shore.
At the age of 1 year, tree climbers are able to breed, and they live for about
10 years.
The main enemy of this species is anenui,
or cannibal tree frog, which eats poison dart frogs for their poison, which
is completely harmless to it.
This amphibian species was discovered by Wovoka, the forum member.
Shukatti
(Lignobates shukatti)
Order: Tailless amphibians (Anura)
Family: Poison dart frogs (Dendrobatidae)
Habitat: subtropical forests of North America along the coast of the Gulf of
Mexico and the Atlantic Ocean (from east Texas to Florida).

Picture by Wovoka
The green-and-black poison dart frog (Dendrobates auratus),
which inhabited the Central America during the human era, became the ancestor
of a whole clade of North American poison dart frog species adapted to new habitats.
The diversity of habitats has led to the divergence of various species of North
American poison dart frogs, differing from each other in size and coloration
features, but their common feature is the presence of poisonous slime protecting
from enemies. One of the most toxic species of the group is shukatti, a widespread
species from southern North America.
Despite its considerable range, this animal is difficult to find, because shukatti
is one of the smallest species of frogs of the Neocene epoch: females are up
to 1.5 cm long, males grow up to 1 cm. In the language of the Choctaw Indians
(lived once in the states of Mississippi, Alabama and Louisiana), the word “shukatti”
means “little frog”. The animal is remarkable in its gracile physique and elongated
limbs. The body coloration is acid green with bright red spots of various shapes
and sizes on the back. In different populations, the size and number of spots
can vary from some small spots on the back to an almost completely red back
from the eyes to the hips. The belly is bright green in color; there are black
“gaiters” and bright red “gloves” and “socks” on the legs. Sometimes the red
color remains only at the tips of the digits, and occasionally it may disappear
completely.
Due to the small body size, frogs of this species are at risk of dehydration,
so they live mainly in river valleys, in humid forests with abundant undergrowth
of large grasses. The favorite shelter of this frog species is the leaf axils
of large herbaceous plants, where a small amount of water accumulates after
rains. These frogs often spend the day in the leaf axils, sticking out only
their front legs and head, and leave this shelter at night. In cloudy weather,
they are active during the day.
Despite the metabolic rate characteristic of amphibians, these animals must
feed almost continuously – fasting for several hours leads to exhaustion. Shukatti
feeds on very small insects with soft covers – physopods, aphids and dipterans.
This frog also eats small slugs.
Reproduction of this species does not depend on a specific season, and is limited
only by the presence of reservoirs suitable for growing offspring – with a slow
current and thickets of small-leaved marsh plants. Males attract females with
calls resembling the clock ticking. The female spawns no more than 10 eggs at
once, repeating spawning up to 5 times a year. The male does not protect the
clutch. Tadpoles are thin-bodied; they hide in thickets and feed on sessile
protists and rotifers. By the time of metamorphosis, the length of the tadpole
is almost twice the length of an adult due to the long thin tail. After metamorphosis,
the length of a young animal is about 2/3 of the length of an adult. At the
age of 3 months, the young animal becomes sexually mature. The maximum life
expectancy is up to 2-3 years.
The main enemy of this species is anenui,
or cannibal tree frog, which eats poison dart frogs for their poison, which
is completely harmless to this one.
This amphibian species was discovered by Wovoka, the forum member.
Deadly
wareko (Wareko mortiferus)
Order: Tailless amphibians (Anura)
Family: Poison dart frogs (Dendrobatidae)
Habitat: Amazon rainforest.
In the Holocene, the most poisonous animals on Earth were poison dart frogs
of the Phyllobates genus, which contained huge concentrations of batrachotoxin
in the body. However, the poison did not save the poison dart frogs from the
damage caused to nature by humans. As a result of the destruction of habitats
and the spread of diseases, only 2 of the 5 species of the genus known in the
historical era survived. In the Neocene, the descendants of these surviving
species still exist, but they are no longer the most venomous poison dart frogs
– they were overtaken by another species. The deadly wareko is the most venomous
animal of the Neocene, at least among amphibians. The name “wareko” means “poison
dart frog” in the language of the South American Caribs tribe.
It is a descendant of one species of Ranitomeya frogs of the Holocene, which
survived in small populations having immunity to chytridiomycosis. Like its
ancestors, wareko is a small frog: females reach a length of 3 centimeters,
and males – 2.4 centimeters. The body proportions also remained similar ti the
ancestor. It is a brightly colored frog: the body is black and orange with a
gradient (the head and eyes are black), the limbs are blue with black spots.
All poison dart frogs are poisonous, but wareko is particularly toxic. It contains
3.5 times more batrachotoxin than in proportionate related species. It gets
this substance from ants, beetles and other poisonous arthropods. Perhaps it
was the appearance of a parasitic fungus that became a factor determined the
direction of the evolution of the wareko’s ancestors towards increased toxicity:
in this way, their slime got increased antifungal properties. This species is
active at dusk and lives mainly on large old trees, rarely descending to the
ground. This species prefers areas of old undisturbed forest with extensive
thickets of epiphytes on the tree branches.
Reproduction in wareko is similar to the reproduction of other poison dart frogs
– the male occupies a certain territory in the thickets of epiphytes, chooses
a place for spawning eggs and calls the female. Mating song sounds like very
quiet monotonous “kri-kri-kri”. After the eggs (11-15 eggs in one clutch) are
spawned, the male protects them for 15 days until the offspring hatch, and then
moves the tadpoles to the bromeliad pools, where he also guards them until the
end of metamorphosis. The froglets leave the water at 2 months, become sexually
mature at 6-7 months, and live up to 8 years.
This amphibian species was discovered by a Dohlozavr, the forum member.
Mantid-mimic
frog (Mimohyla mantidomima)
Order: Tailless amphibians (Anura)
Family: Tree Frogs (Hylidae)
Habitat: swamp regions of China.
Reservoirs in tropics (however, in any latitudes) are the true battlefield.
Predators from infusorians up to insects, fishes and amphibians are plentifully
represented here. Even plants in reservoirs may be predators often. Therefore
ones unable to be a predator, or unable to resist stronger predator, develop
fantastical and refined strategies of survival. Some animals hope for passive
protection, growing armours and needles interfering attack of predator. Others
prefer to seek safety in flight or to hide, and third ones hope for strong poison.
But the most skillful tactics is to show a predator, that things seen by it
are actually not what it expects. Imitators are among various animals and in
the various habitats. But the indispensable condition of their occurrence is
a presence in the same habitat of the creature which is able to protect itself
and warns predators about it. The interesting insect lives in bogs of China
– it is aquatic soothsayer.
This creature is successfully protected from water bugs and predatory medium-sized
fishes. Noticing a predator, aquatic soothsayer demonstrates strong trapping
legs, showing readiness to self-defence. Having received a serious chipped wound,
the predator recedes. At such protected creature completely unexpected simulator
had appeared – it is one local tree frog named mantid-mimic frog. Due to dexterous
imitation of aquatic soothsayer this amphibian can feel like protected from
smaller water predators. It is very important for the frog, because its length
is only about 7 – 8 cm.
The frog imitates not whole soothsayer – it too differs from this insect in
body shape. But to frighten underwater predator it is enough to imitate the
forward part of body of aquatic soothsayer shipped in water. At mantid-mimic
frog there is thin body simulating a prothorax of insect. It is colored yellowish
with thin faltering longitudinal strips. Hips and shins expanded as blades simulate
the terrible weapon of insect – the forward pair of legs armed with spikes.
Like trapping legs of soothsayer they are colored green from above, and grey
from below. The basis of hips has two light spots (it is the imitation of insect
eyes), and on back there is the turned triangle darker rather than other colouring.
It takes off the head of insect. On expanded lobe-like hips and shins along
the edge there is a pattern like slanting short strokes imitating thorns on
trapping legs of soothsayer.
Forward paws of mantid-mimic frog are very thin and have tenacious fingers.
When the amphibian floats at the surface of water, having grasped any floating
plant, and its bottom part freely hangs down in water, the complete illusion
of aquatic soothsayer trapping catch is created. The small fishes having the
experience of this insect attack, try to keep aside the floating frog. To simulate
the aquatic soothsayer even more precisely, mantid-mimic frog moves back paws,
imitating movements of insect clearing. Toes with swimming membranes at this
moment are compressed and do not break accuracy of imitation.
The mantid-mimic frog leaves water seldom, and prefers to live on the cover
of floating plants. It eats small insects hovering above floating plants in
searches of flowers, and catches them by long tongue.
This amphibian breeds similarly to the majority of frog species. The mantid-mimic
frog does not have complex forms of building behaviour, as at some other species
of amphibians. Pair of animals spawns eggs (up to 500 grains of roe in one laying)
in thickets of cane or other coastal vegetation: here eggs are better protected
from casual predators. Approximately after 5 – 8 days from eggs tadpoles hatch.
They are a little bit similar to parents: their body is also narrow and slender.
Tadpoles of mantid-mimic frog live among coastal vegetation, and eat smallest
animals, scraping them from stalks of plants. They sit among stalks vertically,
and do not like to swim to long distances. At danger tadpoles prefer to hide
among plants, and freeze, nestling against stalks. For the best masking their
colouring represents longitudinal dark green lines on brown background.
At the second month of life the length of tadpole surpasses the length of adult
frog, and to the beginning of metamorphosis the tadpole of mantid-mimic frog
is almost twice longer, rather than adult individual. About two thirds of its
length muscled tail amounts. Till the metamorphosis tail is resorbing and simultaneously
the body of tadpole gets the shape characteristic for adult amphibian of this
species. On hinder legs characteristic lobe-like outgrowths appear, and head
becomes wide and flat. Passed through metamorphosis young mantid-mimic frog
is only a quarter shorter than adult individual. This is the original adaptation
to imitation of aquatic soothsayer - the amphibian “slips” through the age when
soothsayers are too small, and there is no sense to imitate them.
Life expectancy of mantid-mimic frog is about four years.
Butterfly tree frog (Papiliohyla membranata)
Order: Tailless amphibians (Anura)
Family: Tree frogs (Hylidae)
Habitat: rainforests of Southeast Asia, trees.
Tropical rainforest is very favorable place for life of various species of
amphibians. Tailless amphibians – various species of frogs and toads – are
the most typical representatives of this class in tropics. They differ from
each other in size and diet, and also lead different ways of life, inhabiting
tropical forest from wood litter up to crones of emergent trees. Some of them
have camouflage colouring, and predator can find them out only after long searches.
Other species, on the contrary, compete to tropical flowers and birds in brightness
of colors. One representative of tree frog family, butterfly tree frog, belongs
to the number of such species.
It is small and gracile climbing frog – the length of its body does not exceed
20 mm. It is able to make long gliding jumps in forest canopy. Due to light
weight it effectively uses weak air streams for jumps. Butterfly tree frog
is not unique in this skill: it shares habitats with glidefrog, the amphibian
having very much advanced membrane between limbs. But this species differs
from the glidefrog in features of anatomy: butterfly tree frog makes gliding
jumps with the help of very advanced membranes between the lengthened fingers
and toes of all paws. In this feature it is convergently similar to flying
frogs of genus Rhacophorus belonging to family Rhacophoridae. Fingers and toes
at this frog are of almost identical length.
The basic coloration of the top side of body of this tree frog is cryptic –
brownish background with small black specks composing faltering longitudinal
lines. But, displaying the colouring to relatives, this frog is changing. Elastic
membranes between fingers and toes are brightly colored – they are citreous
with red border at the external edge and small black specks on the whole surface.
Stomach of amphibian is white. Females differ from males in dimmer color of
membranes.
Displaying colouring of membranes, frog jumps from trees, straightening like
fans fingers and toes of all extremities. Due to small size the butterfly tree
frog can easily overcome distance of about ten meters in one jump. In flight
pattern of paws is clearly visible; at this moment frog is similar a little
to the butterfly – hence its name. One more way of displaying of bright spots
is used at direct meeting of competing individuals: declaring the rights to
the territory, the frog keeps on tree bark, having opened widely fingers and
having straightened membranes between them.
Displaying of colouring of membranes between fingers and toes is one element
of courtship behaviour. Having met each other, males by turns lift one of forepaws,
showing each other colouring of membranes. Size and brightness of colouring
of gliding membranes are determining factors at the establishment of domination
relations.
Butterfly tree frog eats tiny insects and spiders.
This amphibian breeds, spawning large eggs in hollows of trees filled with
rain water. In clutch it may be up to 30 eggs of reddish color, rich in carotene.
Male protects nest against competitors, uttering the sounds similar to mouse
squeak. In first days of life tadpoles scrape slightly rotten wood, digesting
bacteria and fungi. At the age of 2 weeks they become predators and hunt mosquito
larvae. If it is not enough food for them, they turn to cannibals. At monthly
age they undergo metamorphosis and turn to little frogs about 8 – 9 mm long.
They differ from adult individuals in poorly advanced membranes between fingers
and toes and are not able to make gliding jumps.
Giant
mossy tree frog (Bryodorsa titan)
Order: Tailless amphibians (Anura)
Family: Tree frogs (Hylidae)
Habitat: China, swamp forests.
Increase of climate humidity in Neocene has resulted in occurrence of marshlands
at the territory of East Asia. Mountains serve as natural condensers of moisture
– they interfere to penetration of rain clouds into central areas of Asia, supplying
numerous rivers flowing down from their slopes to the east, aside Pacific Ocean.
Swamp forests growing in basins of these rivers serve as a habitat of numerous
moistureloving animals. Amphibians living in these forests are especially various.
Among them there is especially remarkable very large tree frog developing the
most interesting way of care of posterity – giant mossy tree frog.
This species is huge (about 25 cm long) frog of heavy constitution with strong
paws and very tenacious fingers and toes. It skilfully swarms up trees and lives
in swamp forests from the top part of tree trunks up to top of crone. This frog
goes down to the ground extremely seldom, only in case of need. However, when
the river burst the banks floods forests, giant mossy tree frog willingly goes
down in water and swims rather long distances from one tree to another. Thus
to have additional buoyancy it is simply inflated.
This amphibian is remarkable in heavy constitution and is not able to swarm
up thin branches. Giant mossy tree frog keeps mainly on trunks and thick branches
of trees, preferring thickets of epiphytes and thick stalks of lianas where
it is easier to cling by fingers. Appearance of this frog is very characteristic:
back is wide and flat, head is dulled with rather small eyes, and hind legs
are short. Coloration of this amphibian is cryptic – brown with green irregular-shaped
spots, shaded with strokes of black color on sides. The sexual dimorphism at
this species is expressed in size first of all. Male is larger than female in
size though it is approximately quarter lighter, rather than female. It is connected
with special role of male of this species in breeding process. At males of giant
mossy tree frog on back numerous fibres and outgrowths develop, which make back
of this animal similar to thickets of moss or lichen. Undoubtedly, these outgrowths
serve as additional bodies for air breath, but their main function is clearly
appreciable during the spawning. In courtship season these outgrowths swell
and extend a little. Males involving the female with calls gather somewhere
in crone of tree. The female comes guided by male voices and begins to estimate
development of these outgrowths on their backs, touching by throat the back
of calling male. When the female stops, having put head on male’s back, it is
a signal for him. He stops calling and gets on back of the female which carries
him away for egg spawning.
During the spawning male still stays on female’s back, but keeps one of hind
legs opposite to her cloaca. When the female spawns a portion of eggs, male
picks them up and distributes on the back, pasting eggs to fibres and outgrowths.
Almost at once he produces a portion of sperm liquid and greases eggs with it.
After that the cycle repeats and as a result eggs are placed as the even layer
on male’s back. In clutch it is totaled up to 200 eggs. The rate of fertilization
of eggs is almost 100%. When the female finishes egg spawning, male leaves her
and entirely devotes itself to care of posterity. He regularly moistens eggs
with water, not allowing them to dry up. For this purpose male sits under drops
of rain and crawls through thickets of epiphytic plants. If such humidifying
is not enough, he puts on eggs by paws thin layer of slime which is slowing
down the process of drying.
Male carries eggs on back within one week, but his care of posterity does not
terminate after it. Usually the greatest death rate of posterity is observed
at the moment of passing to independent life. At these amphibians such moment
is delayed for some time. Male feeds tadpoles with skin secretions which are
produced between fibres. Tadpoles have scraping mouthes and lengthened fish-like
body. Yolk sac quickly resolves at them and they pass to feeding on skin secretions
of male. They remain on his back up to developing of rear legs, growing up to
20 – 25 mm. When the period of bringing up of posterity comes to an end at the
male, he goes down on the ground and hastens to any reservoir. Having got into
water, male cautiously removes tadpoles from back by hind leg.
Tadpoles of giant mossy tree frog grow quickly, eating small sedentary invertebrates.
They are colored grayish-green with bright yellow belly and keep near to bottom.
At danger they are quickly dug in silt. At length of about 10 cm (the tail makes
a little more than half of total length) tadpoles undergo metamorphosis and
turn to young frogs. At this time young individuals suffer from cannibalism
in greatest degree – adult tree frogs of this species frequently eat them alongside
with frogs of other species. Young frogs eat tiny insects, but, as they grow,
their diet changes – they begin to hunt large spiders and cockroaches. Adult
amphibians hunt small vertebrates and frequently ravage bird nests.
Anenui,
or Cannibal tree frog (Anenuihyla batrachophaga)
Order: Tailless amphibians (Anura)
Family: Tree frogs (Hylidae)
Habitat: subtropical forests of North America along the coast of the Gulf of
Mexico and the Atlantic Ocean (from east Texas to Florida and north to North
Carolina).
A descendant of the barking treefrog (Hyla gratiosa), that lived in the subtropical
forests of North America during the human era. In the historical era, barking
treefrogs, like many other amphibians of North and South America, were severely
affected by the fungus Batrachochytrium dendrobatidis, but part of the population
managed to develop immunity to the fungus, and by the Neocene tree frogs managed
to restore their population and even form several new species. One of these
species is anenui (the name means “frog” in the language of the Atakapa Indians.
Before the arrival of the white colonizers, the Atakapa Indians practiced cannibalism).
Anenui is a specialized predator and cannibal – it eats almost exclusively other
tailless amphibians, including its own relatives. Anenui can even eat toxic
poison dart frogs, whose venom is completely harmless for it, and due to the
venom of poison dart frogs, it also becomes poisonous. Because of it, many predators
try to avoid it, although its venom does not stop some snake species at all,
and they hunt it. Anenui can also eat various invertebrates (worms, slugs, millipedes,
arachnids, insects and woodlice), small reptiles, rodents and birds (especially
their nestlings). Anenui tadpoles are also predators: they hunt other tadpoles,
fish fry and insect larvae. Tadpoles can be eaten not only by relatives, but
also by predatory fish.
Anenui is much larger than its ancestor, and reaches a length of up to 15 cm.
Its head is also larger than that of its ancestor, with strong jaws. Body is
compact; back is slightly arched. Males have a throat resonator sac. The frog
climbs trees and large herbaceous plants well. For fixing on smooth leaves,
its fingers and toes are equipped with round suction cups. The coloration is
quite bright: the back is yellowish-green with many dark green and bright red
spots. The belly is bright yellow. In the mating season, the resonator of the
calling male is clearly visible against the background of vegetation due to
its bright yellow color.
This tree frog spends a significant part of its life on trees or shrubs; it
is also found in grassy vegetation along the riverbanks and shores of lakes,
swamps and just puddles. At the sight of snakes, it is able to burrow into the
mud, but caught on the trees, it escapes by making long jumps. Activity is mainly
nocturnal and crepuscular, during the day the frog rests in a shelter among
the leaves.
During the breeding season, the male calling the female utters loud sounds resembling
dog barking, like its ancestor. This species is polygamous, and the male can
fertilize the eggs of up to a dozen females in some days. At the same time,
he has to exercise a certain caution and dexterity, because after spawning,
the female can easily snack on the male. Breeding takes place from March to
August in reservoirs. The female spawns up to 5000 eggs in the water, during
the breeding season the spawning can repeat twice. Tadpoles hatch after 10-12
days. Depending on the temperature, their development takes from 40 to 160 days;
in the north of the range in the cold winter, tadpoles can dig into the silt
and complete metamorphosis in the spring. They reach a length of up to 9 cm
along with the tail. Females become sexually mature at 3 years old, males at
4 years old. This species can live up to 15 years, but most anenui tree frogs
die earlier, mainly due to predation by relatives or snakes.
This amphibian species was discovered by Wovoka, the forum member.
Carpentary
Saline Frog (Halorana aquatica)
Order: Tailless Amphibians (Anura)
Family: Australian Tree-frogs (Pelodryadidae)
Habitat: Vegetated margins and swamps of Carpentary lake.
Frogs are notable for being sensitive to chemical imbalances due to their porous
skin, through which they breathe and exchange moisture. But in some cases species
emerge that are able to tolerate certain imbalances, as a specialist found nowhere
else. Lakes of high acidity levels in the Holocene once hosted frogs which were
adapted to this change, and various frogs and toads can tolerate higher salinity
levels.
And this we see again in the Neocene the Carpentary Saline Frog. In many ways
it is a typical aquatic frog, it greatly resembles a smaller version of its
ancestor, the Bell Frog (Litoria inermis), reaching a body length of up to 7
centimetres. The body is a dirty olive green with dark brown stripes, the belly
is greyish white. This frog is a strong swimmer and spends most of its time
in or near water, though it is competent on land. This species is most active
in the mornings and afternoons, and calls during twilight.
The brackish water of the Carpentary Lake is not equable to most kinds of frog,
and so the Saline Frog thrives here in great numbers. It can even be found within
the swamps that border the lake, which have a higher level of salinity than
the lake itself. As well as this form, some descendants of the Cane Toad are
able to breed in these waters. Carpentary Salt Frogs breed mainly during the
wet season, from November until April, when rains may freshen the water, but
tadpoles and eggs are able to even tolerate the higher brackishness of the deeper
layers of the lake. Males court the female, attracting her with a deep “Wonk-wonk”
growling croak, calling occurs more frequently during rain. Amplexus for mating
is achieved in water, and it may last as long as 10 hours. Egg laying and fertilisation
take about 6 minutes, and as many as 5000 eggs may be laid. Eggs will develop
and hatch relatively quickly, in as little as a day. Tadpoles are about half
a centimetre long at birth and grow exponentially from that time, soon reaching
a centimetre or two within a month. Tadpoles are brown with a typical scraping
mouth, but lighten as they near adulthood. It may take as long as 4.5 months
for the tadpole to lose its tail and become a froglett, which are usually about
3 centimeters long. Both tadpoles and adults may engage in cannibalism. Lifespan
of this frog may reach 10 years, they are commonly preyed upon by predatory
fish and water birds as well as snakes.
This amphibian species was discovered by Timothy Donald Morris, Adelaida, Australia.
New
Zealand two-wattled frog (Xenolitoria barbata)
Order: Tailless amphibians (Anura)
Family: Australian Tree-frogs (Pelodryadidae)
Habitat: New Zealand, swamps and lakes on the plains, in the foothills and mountain
valleys.
In the human era, the green tree frog (Litoria caerulea) was introduced to the
territory of New Zealand. This event led to significant changes in the course
of amphibian evolution in the archipelago. Native amphibian species were driven
out by the Litoria descendants, and in the Neocene, the descendants of this
tree frog demonstrated the phenomenon of adaptive radiation, having occupied
most of the habitats available to amphibians. One branch of the descendants
of this species has lost the adult stage and is represented by a variety
of neotenic forms,
representing the counterparts of the tailed amphibians of the northern hemisphere.
Another branch of these amphibians retained the adult stage and the familiar
appearance of tailless amphibians. One of the representatives of this group
is the New Zealand two-wattled frog.
This amphibian species is a peculiar analogue of toads: it is a ground-dwelling
frog, very poisonous due to the ability to secrete bufotenin. The body length
of an adult is up to 40 cm; the female is more robust, the male is slimmer and
smaller. The body coloration is brown with blurred marble pattern on the back.
The eyes are large and well camouflaged: the iris is brown with thin vertical
strokes imitating the pattern of the tree bark. There are large warty growths
on the eyelids. The belly is white with pale blue spots. This species moves
well on land, which allows it to settle effectively; the limbs of the animals
are shortened, in its body proportions this species resembles a toad. It is
unable to climb trees, and the suckers on its digits represent just rounded
skin outgrowths. The head of this frog is remarkable: it is equipped with two
small wattles (like in catfish, but shorter), which grow in the middle of the
lower jaw and have some mobility. These are the tactile organs and organs of
chemical sense. Instead of atrophied teeth, the keratinized lips are covered
with numerous sharp horny denticles – it is a larval feature that has been preserved
and further developed in an adult animal due to the peculiarities of the diet.
Keratinized lips also help the animal to dig the mud in search of crustaceans
and insect larvae, but these items make up a significant part of the diet only
in young animals. Adult amphibians of this species are ambush predators.
This species lives in marshes, less often in tiny forest reservoirs. The New
Zealand two-wattled frog feeds on small terrestrial vertebrates, fish and swamp
species of false salamanders, which are not poisonous.
The mating season begins in spring. The incentive for reproduction is an increase
in precipitation and a change in the chemical composition of water. Males move
into reservoirs, change their coloration to a lighter one and wait for the appearance
of females, starting mating songs. Their vocalization resembles the squeaky
yapping of a small dog, repeated in series several dozen times. Each male occupies
a fairly large territory from which competitors are expelled. In the presence
of a female, the male behaves aggressively towards other animals, displaying
his physical condition to her.
There are up to 300 large eggs in the clutch. This species is characterized
by parental care: it is necessary because tadpoles do not secrete bufotenin.
The female occupies the territory previously chosen by the male and grows her
offspring there. She guards the brood, feeds the tadpoles with unfertilized
eggs, and when the reservoir dries up, she moves the tadpoles in her gular sac
to another reservoir. The male can also take part in the transportation of tadpoles.
Tadpoles of this species grow to large sizes and begin metamorphosis at a length
of about 10 cm. This species is polygamous: the male guards his territory and
does not allow other males entering it (as a rule, the territory represents
a small lake). At the same time, there may be several females with their tadpole
broods at different stages of development on the territory of one male.
This amphibian species was discovered by Dmitry Korshin, the forum member.
Tramper
False Toad (Bufolitoria terrestris)
Order: Frogs and Toads (Anura)
Family: Australian Tree Frogs (Pelodryadidae)
Habitat: Vegetated areas of North and South Island, New Zealand, including swamps,
scrub-land, forest, mountain forest etc.
Until the arrival of Europeans, the amphibians of New Zealand were distinctive
and primitive (family Leiopelmatidae). Europeans introduced Australian tree
frogs (genus Litoria), which quickly reproduced and proliferated. Neocene amphibia
of New Zealand consists entirely of descendants of this introduced species,
and have radiated into unique forms, including ones that retain juvenile features
into adulthood, and have an unusual appearance (Virilogyrinidae).
Among the more typical frogs of Aotearoa, some have adapted into partly terrestrial
species which are common in vegetated areas. The Tramper False Toad is so-called
because it is adept at moving overland, and usually does so by walking or short
hops. Outwardly it resembles a mid-sized toad or a large warty frog, hind-legs
are shorter than its ancestors, but both fore and hind legs are strong, and
posture is upright. As it is technically a frog, it lacks the large poison glands
on the shoulder that typify a true toad. Body is covered in coarse small bumps,
as well as glands which secrete a bitter-tasting mucus that serves to defend
it from predators. Head is large, with big protruding eyes, mouth is large and
broad. Total body-length may be up to 20 centimetres not including outstretched
hind legs. Coloration is an all-over dark brown, spotted with light grey and
black, stomach is pale white with distinct yellow spots.
As with true toads, it is a roving predator that spends all of its time on the
ground, it may actively search for prey, or sit in wait for prey to pass by.
Its diet includes various insects and larvae, including non-poisonous beetles,
roaches, earwigs, maggots, caterpillars, crickets etc. The frog pounces upon
prey with a short lunge and stuffs it into the mouth using the forelegs. When
confronted with a predator, it hunkers close to the ground and tucks its legs
underneath it, it may let out a loud squealing call in order to startle the
predator. Skin is coated in a very bitter-tasting mucous that often deters predators
from swallowing it. This amphibian is active by day and sometimes also during
dusk or dawn.
Breeding occurs in the wettest part of the year, between June and late September.
Males will chose water-sources such as creeks, ditches and large puddles, as
potential spawning sites. They sit in a slightly elevated position close to
the water, and call loudly in order to attract a female, the male's call is
a loud high-pitched “breddik,-breddik-breddik”. Once a female submits to be
mounted, amplexus takes place in the water, and up to 1500 eggs are laid, which
immediately stick to each-other in masses, and sink. Development of the tadpoles
after hatching takes about 6 weeks to reach full metamorphosis. Miniature young
frogs emerge on to land and seek damp patches of vegetation in which to hunt
small insects, and hide from predators. Lifespan is up to 20 years, predators
of adults can include birds, mustelids and snakes,
tadpoles are consumed by fish, predatory
water insects, and other amphibians.
This amphibian species was discovered by Timothy Morris, Adelaide, Australia.
New
Zealand cave frog (Virilohyla spealea)
Order: Tailless amphibians (Anura)
Family: Australian Tree-frogs (Pelodryadidae)
Habitat: New Zealand, caves of Northern Island.
In human epoch New Zealand had the most ancient flora and fauna of great level
of endemism: at birds and reptiles the endemism reached an order level. It is
connected to long-term isolation of islands – presumably, from the end of Mesozoic.
But in human epoch isolation of island flora and fauna had been terminated.
People had introduced to islands great number of species completely alien for
islands – mainly ground mammals of various orders, and also fishes, birds and
plants. Among island immigrants large Australian tree frogs of genus Litoria
appeared. These intruders had got accustomed well at the islands and had given
rise to various species of local tree frogs. Moreover, during the evolution
of larval stage of tree frogs in water bodies of New Zealand the original endemic
group of “tailed amphibians”, New Zealand false
salamanders, had appeared. These amphibians proved to be very successful
group, and had occupied habitats, characteristic for tailed amphibians in Northern
hemisphere. Besides at the islands the species of amphibians is kept, throwing
light on the origin of this endemic group.
Mountain areas of New Zealand are rich in caves. Cracks in the ground and narrow
clefts in mountains lead to extensive underground cavities, the part of which
is water-filled. The underground rivers, flowing from glaciers, may run into
small underground lakes and penetrate to ground surface as springs. These reservoirs
are populated with various creatures – insects, crustaceans and even representatives
of false salamanders. And some creatures are only temporary visitors of
caves.
In spring a plenty of small tree frogs gathers to cave entrances. These ones
are creatures only about 5 centimeters long with characteristic appearance of
tree frogs – large eyes, flattened head and sucking disks on fingers and toes.
Colouring of these tree frogs is bright green with dim brownish strip from nape
up to the back end of body. Atop of this strip the narrow precisely outlined
stripe of white color lasts. Such colouring helps these creatures to hide from
predators among foliage. This masking is broken only with large yellow eyes
with brownish nerves and narrow vertical pupils. However, having closed eyes,
this tree frog completely merges with surrounding foliage. It is New Zealand
cave frog which frequently keeps near to caves and in damp gorges overgrown
with tropical vegetation.
All these frogs are females. They lead way of life typical for these amphibians,
swarm up trees and large-leaved bushes, eat insects and other invertebrates.
In spring when rains become more plentiful and streams in mountains overflow
banks and turn to rough rivers, at these amphibians courtship season begins.
Females of New Zealand cave frog migrate to places where they had hatched from
eggs a long time ago – to underground rivers flowing in caves. Bellies of these
frogs are overfilled with eggs, which even are visible through thin belly skin
of these animals.
In darkness of caves shrill voices of males do not meet these females. Having
found water, females enter cold stream and wait while males will find them.
As against females, males of New Zealand cave frog never leave underground rivers.
They are not able to leave water at the will at all. These creatures differ
from females even in appearance: male of this species is about 8 cm long and
looks like very big tadpole with the advanced back legs. The muscled tail with
well advanced fin makes about half of its total length. Only the male’s head
partly takes the shape characteristic for adult frog, but eyes remain small
and underdeveloped. In sides of male’s mouth short triangular wattles with wide
bases supplied with sensitive cells are advanced. Due to well advanced chemical
feeling male easily finds the female ready to lay eggs in darkness of cave.
In caves where the sunlight doesn’t penetrate, males have pinkish-white depigmentated
skin, and in reservoirs where the sunlight gets from surface, their colouring
becomes grey with brown speckles on sides. In such case eyes also develop at
them.
Such sexual division has the great biological sense. Females introduce into
the poor ecosystem of cave a significant amount of organic substance – eggs
and their own bodies (not all of them succeed to get out and to return to the
forest), and males do not vaste resources for migrations and metamorphosis.
Living in cold water, they require a small amount of food.
Having met the female, male sticks to her back by mouth and clasps her by hind
legs. The pair of animals swims in such position near the bottom, only from
time to time emerging to the surface for air. Male, despite of its larval shape,
has lung breath though it receives a significant part of necessary oxygen with
the help of gills.
Animals lay eggs in natural deepenings at the bottom of reservoir, or dig a
special hole for it. The size of clutch makes no more than one hundred of eggs,
but usually totals no more than 75 ones. Having spawned eggs, female leaves
the reservoir, and does not care of posterity any more. Male stays near clutch.
Due to advanced lateral line he distinguishes approach of predators and rises
in defence of clutch. It strikes to the attacker impacts by head and tries to
push it aside from eggs by side and impacts of tail. Till the time of egg incubation
(it makes about 2 months) male eats of nothing. When tiny long-bodied tadpoles
hatch from eggs, male eats the rests of egg shells and abandons the rests of
clutch. There are numerous animals dangerous to eggs: crustaceans, worms and
even the hungry congeners – males not busy with care of posterity and tadpoles
of advanced ages. Cannibalism in conditions of scarcity of resources represents
the usual phenomenon for this species.
Tadpoles and tadpole-looking males of New Zealand cave frpg are omnivores. They
willingly eat food of animal origin, scrape from stones layer of bacteria and
protozoans, and also eat the vegetative parts getting into the reservoir. Large
tadpoles and males also hunt small crustaceans.
This species is very close to primitive false salamanders keeping a significant
number of tadpole features in adult condition. Obviously, this frog is a relict
of early stage of evolution of New Zealand false salamanders.
The idea about existence of this species was proposed by Simon, the forum member.
Armored
pouched frog (Barogastrotheca armata)
Order: Tailless amphibians (Anura)
Family: Egg-brooding frogs (Hemiphractidae)
Habitat: swampy tropical forests of South America.
Neocene is a time when new species and families of various animals, fungi and
plants came on the scene and conquered the planet, but at the same time there
were a significant number of forms that survived in the human era in the living
world of the planet. The descendants of these “old residents” of the Earth also
did not always give up their positions, and even developed new ecosystems, giving
rise to new and unusual species. One of these species is the armored pouched
frog, an inhabitant of tropical areas of South America.
The armored pouched frog is an amphibian of very large size compared to its
relatives, about 16 cm long (hence the prefix baro- (“heavy”) to the genus name),
and colored various shades of green and brown. Having increased in size, it
almost lost the ability to climb trees, and moved into the undergrowth of the
tropical forest. This amphibian spends most of its life buried in the wood litter,
where it waits for smaller vertebrates or invertebrates to appear next to it,
and devours them. This frog happens to fall prey of various mammals and birds,
from which it escapes mainly due to camouflage coloring. Another notable feature
of this species is a significant number of subcutaneous ossifications (hence
the species epithet) – they cover mainly its head, but are also arranged in
rows along the back. These ossifications protect it from large predatory frogs,
and from fish under water.
The armored pouched frog does not have an expressed breeding season – like most
tropical species, this amphibian is ready to breed at any time of the year.
Therefore, the mating songs of males of this species – a shrill, non-melodic
croaking – are heard at any time of the year. The peculiarity of this species
is that it does not need water for reproduction, and its clutch is very small:
instead of hundreds and thousands of eggs, like in other tailless amphibians,
there are only two or three large eggs rich in yolk, which the female carries
on her body after the male fertilizes the eggs. The eggs are placed into the
skin “pouch” on her back – it is a specialized organ that protects first the
eggs, and then the tadpoles. Tadpoles of this species attach to the epithelium
of the walls of the brooding pouch and live there for several weeks, receiving
nutrition from the mother’s blood, until they turn to froglets and leave their
mother. The length of a young frog at birth is about 4 centimeters. After bearing
offspring, the female eats off for a while, sheds, and when some days or weeks
pass, she is ready to start a new breeding cycle.
Armored pouched frog can live about 11-12 years, but usually its lifespan is
shorter. It reproduces much slower than most other tailless amphibians, but
thanks to parental care, infant mortality in this species is much lower, so
this species maintains a stable population and is not rare.
This amphibian species was discovered by Bhut, the forum member.
Branchial
tadpole (Branchiosuga nosferatu)
Order: Tailless amphibians (Anura)
Family: Neotenic frogs (Pseudocaudidae)
Habitat: tropical ponds and rivers of South America.
Picture by Alexey Tatarinov
Colorization by Carlos Pizcueta
In fauna the phenomenon of neoteny is very much distributed:
larval stage of individual development gets ability to breed, not starting
to metamorphosis.
Among vertebrate animals such phenomenon is shown by some tailed amphibians
like mole salamanders (Ambystoma). Larvae of these amphibians are known as
“axolotl”. Some other tailed amphibians, for example, Proteus and Siren,
represent, in the main, becoming adult larva which had lost the adult stage.
In human
epoch among tailless amphibians such phenomenon was not known.
But in Neocene one of numerous species of South-American frogs had made that
earlier was possible only to its tailed relatives: it had lost adult “frog”
condition under rather unexpected circumstances. Tadpoles of this species
able to normal breeding, live in rather unusual place: at gills of huge fishes boltergillers
(Potamocetus balaenognathus), eating river plankton, and occasionally
on gills of other big fishes. They are species of amphibians had mastered
parasitic
habit of life - being mature they eat exclusively blood of fishes.
These creatures completely passed to breeding at larval stage and had lost
an adult stage of the frog. The parasitic way of life influenced upon anatomy
of this animal. Body of branchial tadpole is flat and oblong; mouth has transformed
to sucker. In mouth there are two corneous plates, with which tadpole bites
through epithelium of gills of the host fish. The body of animal is about
5 - 6 cm long; it is covered with thin translucent skin of grayish color
through
which interiors and blood vessels appear through. Because the tadpole spends
all adult life on fish, it does not have necessity to swim to anywhere. Therefore
its tail is very short, accounting about quarter of general length of animal.
In spite of the fact that this creature is only the adult tadpole, at it
hinder legs serving for keeping on gills of fish develop. At these paws only
two toes
develop, forming some kind of “pincers” clasping branchial arch of fish.
Sense organs at branchial tadpoles are advanced very poorly: eyes are appreciably
reduced, and there were only few tens of sensitive cells of former lateral
line. Organs of smell sense are well advanced only at young individuals,
but not at adult males.
At these tadpoles males are smaller than females, and are more active than
them: male can move in branchial cavity of fish, finding for females ready
to breeding. The female is larger; her body is convex, and oral sucker is
stronger. Once per some days pair makes a plenty of small eggs (up to 100
ones that is
a big number for small animals). Eggs develop in thickness of water: they
swell in water, and after two - three days from them very small larvae hatch.
The early-aged tadpole of this species lives in plankton and eats it, filtering
through gills. It is tiny and practically completely transparent. Later,
having reached length about 2 cm (tail accounts more than half of length),
the tadpole
settles on gills of the host fish. It is easy for making: the fish, properly,
finds it itself together with other plankton. First time the young branchial
tadpole does not harm the host fish: it only scrapes plankton accumulated
on its gills. Adult animal passes to parasitism – it starts to bite through
gill
epithelium and to suck blood.
To lower an intraspecific competition in the limited vital space, at these
parasitic creatures the original mechanism of regulation of population amount
had developed. Adult individuals eat not only blood of fish. They also may
be cannibals, killing significant part of young tadpoles settling on gills
of fish. Thus the overpopulation of the host animal by parasites is prevented.
Young tadpoles have real chance of survival only if at the infected fish
there is a small population of adult tadpoles.
Toby
frog (Vesicorana toxidermata)
Order: Tailless amphibians (Anura)
Family Spherical frogs (Sphaeroranidae)
Habitat: the wood rivers of South America.

Picture by Alexander Smyslov
After ice age, marked with itself the border of Holocene and Neocene, climate
became more humid, and woods had occupied again extensive territories in
equatorial zone of Earth. In territory of South America the impassable
selva again
was spread. Current of rivers of Amazon region have a little changed after
droughts of glacial epoch, but in Neocene the basin of Amazon and Hyppolite
flowing in parallel to it has occupied approximately the same area, as an
Amazon region in Holocene epoch.
The damp, periodically flooded rainforest, or igapo, is a fine place for
life of various frogs and toads. Here it is possible to find various shelters
and
reservoirs for development of tadpoles, therefore the frog population of
this place is especially numerous and various. At night woods of Amazon region
are
resounded by voices of various frogs. Among them the voice of one of local
frogs, sharp abrupt whistle, is remarkable. The owner of so remarkable voice
is frog about 10 cm long keeping among wide-leafed marsh grasses. By body
shape it differs from the majority of frogs unless by larger and wide head,
and small
eyes are shifted to sides a little. It is colored rather brightly: back is
black with small red spots on shoulders, hips and waist; belly has red spots
on white background. This amphibian does not hide in presence of large animals,
and it looks, they diligently avoid meetings with it. The name of this species
is toby frog.
Two features unite this frog with its “double” among fishes. First, the skin
of this frog contains strong poison. And second, the toby frog has developed
an interesting way of the warning about its virulence. Skin on its belly
is very elastic. In case of danger this frog quickly inflates as a sphere,
swallowing
air or water. Thus it rises on paws so, that bright colouring of stomach
becomes especially distinct. Similarly the toby fish (Tetraodon) turns to
sphere, being
disturbed. At puffed frog on skin drops of poison involuntarily appear squeezed
from skin glands on sides. Poison at it has unpleasant sharp smell, warning
possible aggressors about danger.
However, in nature absolute protection does not happen. Some birds have adapted
to eat toby frogs, avoiding their poisonous protection. They kill and tear
apart frog by claws, and then simply peck meat, not touching poisonous skin.
Here toby frog loses to fish “double” in armament: at fish there are even
poisonous meat and interiors.
When threat of life will pass, frog simply weakens stomach sphincter and
by contraction of abdominal muscles lets air or water outside through a mouth.
This species has staked on its own virulence, and during all life protect
itself by chemical weapon – eggs and tadpoles of toby frog are also poisonous.
Eggs of this frog are colored red because of abundance of carotene – they
develop in reservoirs with slow current, poor in oxygen, and carotene has
in addition
property to fix oxygen. At the same time red colouring warns water animals
that eggs are poisonous. In clutch at this species there may be up to 200
grains of roe. Parents simply leave eggs to the mercy of fate: the survival
rate of
posterity at this species is rather great even without additional care. Tadpoles
of the toby frog have bright colouring too: they are red with black speckles
on sides. They have feather-like external gills necessary for life in water
poor in oxygen. Besides at tadpoles lungs develop early. They eat filamentous
algae and keep in shallow waters, where they have no competitors. These tadpoles
are not afraid of coming nearer animals – they, as well as adult frogs, are
poisonous. The stage of tadpole lasts about 2 months. Having reached length
about 4 cm, the tadpole turns to young frog and goes to land. At the age
of 7 – 8 months young frog can take part in spawning.
Stream suckermouth (Ancistrogyrinus lampetrifrons)
Order: Tailless amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae)
Habitat: mountain rivers of New Zealand.

Picture by Alexander Smyslov
In historical epoch the fauna of New Zealand had suffered the large damage
from the human activity connected to “enrichment” of it by various species
of animals, unusual for natural ecosystems of islands. Among invaders there
were various species of invertebrates and vertebrates, and also plants. In
woods of islands various mammals, and in rivers fishes from various continents
had appeared. There were large Australian tree frogs of genus Litoria among
animals introduced to New Zealand.
Climatic changes at the boundary of Holocene and Neocene had determined the
unusual direction of evolution of these creatures. At the cooling of climate
these amphibians suffered the tightening of metamorphosis. This phenomenon
had been well-known at newts of various species and other tailed amphibians
dwelt in Northern hemisphere. In New Zealand some populations of descendants
of introduced tree frogs had gradually turned to sexual matured tadpoles
and had lost the ability to turn to adult frogs. Actually they had became
tailed
tailless amphibians, once again emphasizing the reserve of names given by
people.
In mountain areas of New Zealand, in clean quickly flowing rivers there are
many various species of such “overgrown tadpoles” occupying various ecological
niches in reservoirs. One of usual species is the stream suckermouth, an
amphibian about 40 cm long. Its body is lengthened; strong muscled tail makes
approximately
2/3 of general length.
Head of this creature is poorly separated from the body; it also is wide
and flat. Small eyes are placed at edges of head and are directed forward
and sideways.
Scraping mouth looking like sucker is placed at the bottom part of head.
The specific name of the animal means “having the head of lamprey” because
of the
similarity in structure of mouth, and the mouth shape also had determined
its everyday name. On top and bottom jaws of this amphibian strips of corneous
denticles, bordering mouth in front and behind, are advanced. With their
help
stream suckermouth scrapes larvae of midges and other sedentary and inactive
invertebrates from stones. In connection with such way of feeding branchial
apertures are turned upwards. Lungs at this species are rather small – this
animal receives oxygen using branchial and skin respiration.
Paws of stream suckermouth are short; hinder legs are only a little bit longer
than front ones. The animal can rake by paws stones in searches of forage,
and at danger it can quickly dig in ground. When stream suckermouth swims,
paws are densely pressed to sides, and the tail is the main organ of movement.
Usual colouring of this animal is soft: the top part of body and sides is
grey with small black spots on back and head. On sides there is less amount
of spots,
and the stomach is absolutely lack of them. The bottom half of body at females
and males out of spawning season is colored similarly – it is milky-white.
In spawning season stream suckermouthes change: the throat and chest of males
turn orange-red, head blackens, and on sides black faltering vertical strips
appear. Besides on tip of tail of males in spawning season the long string
grows (similar to “ornaments” of some armored catfishes of family Loricariidae
of Holocene epoch), and the general length of the male at this time may exceed
half-meter. Upon termination of spawning season this string is resorbed.
The spawning season begins in spring (in Southern hemisphere it is the end
of August – first half of September), when mountain snows thaw, and also
rivers become deeper. Males occupy individual sites and protect them from
congeners.
At borders of sites there are skirmishes at which males show to each other
bright stomach and try to drive the contender off by impacts of head to sides.
When the female swims to the site of male, it begins courtship rituals and
tries to keep the female, sticking by mouth to her side and keeping by paws
for stones and driftwood. The center of the site of the male is the flat
stone which surface is carefully cleared of algae and sedentary animals.
The inhabiting in quickly flowing streams causes the occurrence of the special
reproductive behaviour at these amphibians. The male does not produce liquid
sperm, and lays the spermatophore on the cleared stone. The female swims
above the stone and takes it by mobile edges of cloaca. Thus, at stream suckermouthes
the internal fertilisation takes place. It raises the probability of fertilisation
– in quickly flowing water the liquid sperm may be simply washed off by current.
The female lays on the bottom side of stones a small amount of large eggs
(about 60 – 70 ones about 1 cm in diameter) with sticky environment. Approximately
in one month the tadpoles distinguished from adult animals by absence of
legs
and dark colouring hatch from them. They reach sexual maturity at the age
of 2 years. Life expectancy of this species may be about 15 – 18 years.
The idea about the existence of this group of animals was proposed by Timothy Morris, Adelaide, Australia.
Weed-eating
Suckermouth (Herbogyrinus loricariifrons)
Order: Tailless amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae)
Habitat: Lowland rivers, lakes and marshes of North and South Islands of New
Zealand, areas of aquatic plant growth.
Adaptive radiation occurring in the Neocene has given rise to numerous new groups
of live organisms; in New Zealand a completely new kind of amphibian has arisen
from introduced tree frogs; the false salamanders, which take on many forms.
In weedy lowland fresh waters of New Zealand occurs a relative on the Stream
Suckermouth (Ancistrogyrinus lampetrifrons), the Weed-eating Suckermouth.
This amphibian is distinguished by feeding entirely on the growth of underwater
plants, thus making it the only adult amphibian existing to be an herbivore.
This form is more robust than its mountain relative, having a shorter tail that
is deeper and slightly triangular in shape, body is short and deep, neck is
absent. Adult reaches a length of about 28 centimetres. Hind limbs are larger
than forelimbs and hind feet have extensive webbing extending to the knee. Head
is broad and flat, bearing a mouth which is thick-lipped, moustached and shaped
like a sucker, recalling a South American armored catfish (Loricariidae). Oral
sucker bears rows of large, coarse-ridged horny teeth, which allow this animal
to consume plant material by scraping it. Diet consists of films of algae, thick
growths of filamentous algae, water weeds, and submerged vegetation that are
not too coarse or woody. Body coloration is a greenish brown, with a cream coloured
underbelly; upper side and tail are lined with dark spots of various sizes.
Colouring of the tail becomes more contrasting in the male during breeding season.
This creature is diurnal, and not socially active outside of the breeding season,
but they may form large aggregations in waterways that are thickly choked with
water plants, as would any tadpole.
Spawning season runs from August to early November, in the spring. Males establish
small territories which they defend against other males. Males compete by fluttering
their tail and attempting to bite one another. When a male makes an overture
to the female, he swims back and forth, fluttering his tail vigorously. Male
attaches himself to the female by mouth and leads her to site intended for egg
laying. Unlike its relative, spawning occurs in the usual way, amplexus is somewhat
different, with male retaining the female by gripping gently with its mouth.
Up to 100 eggs are laid in a string that is woven around underwater plants.
Young tadpoles without legs hatch from the eggs in about 12 days, sexual maturity
is reached at 2 years, and life expectancy is up to 17 years.
This species of amphibians was discovered by Timothy Morris, Adelaide, Australia.
Batrachogymnotus
(Batrachogymnotus rivularis)
Order: Tailless amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae)
Habitat: New Zealand (Northern Island), the flatland parts of rivers overgrown
with aquatic plants.
Picture by Carlos Pizcueta
Having evolved in conditions of island isolation and at almost
full absence of competitors, New Zealand false salamanders became the original
analogues
of tailed amphibians of Northern hemisphere. Among them species of various
sizes appeared, similar to tailed amphibians in various degrees. The most
progressive species had almost completely lost the similarity to tadpole, and
the most
primitive species, which inhabit the rivers of islands, keep completely larval
shape. To such species batrachogymnotus living in subtropical climate of
Northern Island belongs; it is a large species of New Zealand false salamanders.
It
never creeps to land and constantly lives in water among thickets of underwater
plants.
This creature is a long-bodied species of eel-like shape, reaching the length
of 80 – 90 cm, and sometimes up to 100 cm; males are larger than females,
but are more graceful than fems are. Body and head are short, and tail makes
more
than 3/4 of general length of animal. It is muscled and rather deep, bordered
with leathery fin plica. On the top part of body and tail fin is reduced
in great degree and represents only low skin elevation. But the bottom part
of
tail fin is wide; the fin is stretched along the belly side of body up to
cloacal aperture. Batrachogymnotus swims with the help of wavy movements
of the bottom
part of tail fin. It this feature it is very similar to South American gymnotid
fishes and demonstrates, thus, a vivid example of convergence. Such manner
of swimming is connected to features of habitat of animal: batrachogymnotus
lives in dense thickets of plants where it is difficult to turn around. Like
gymnotid fishes it can easily swim back to front, hiding among plants from
predators or competitors.
Limbs of batrachogymnotus are rather short, forward paws are much shorter
than rear ones. At 30 – 40% of representatives of population forward limbs
may not
develop at all. Hind legs lack membranes between toes and can be used at
movement among plants. Hip joint in them is very mobile, and paw can be turned
forward,
grasping stalks and leaves of plants by toes.
Head of this amphibian is flat, with well advanced eyes located on its edges.
Mouth of animal is wide, capable to be stretched vastly and swallow large
prey. In corners of mouth long sensitive wattles grow, supplied with chemoreceptors.
Colouring easily permits batrachogymnotus to hide among plants and stones.
The basic background of colouring is yellowish-brown, on it there are are
randomly scattered spots of dark brown and black color, forming a marble
pattern. Stomach
is almost white with separate black specks.
Batrachogymnotus is an active ambush predator. It feeds on fishes and crustaceans,
eats frogs and other small vertebrates, and also aquatic larvae of insects.
At swallowing of large prey stomach and belly wall of animal can stretch
greatly; animal easily swallows fishes in length up to third of own length
of the hunter.
For the significant part of time batrachogymnotuses hide in thickets of water
plants and in driftwood. These animals behave aggressively to each other,
and fight is fastened between them at their meeting. Usually animals mark
borders
of the territory, rubbing body against underwater objects.
Spawning at this amphibian takes place in spring. When it is raining in mountains,
the rivers overflow banks and water is considerably enriched in oxygen. It
is a stimulus for the beginning of courtship behaviour at these animals.
Males turn brighter – their throat reddens, and on sides distinct dark vertical
strips
appear. Male’s skin turns lighter, and the back becomes almost white. The
female gets black colouring with white stomach. Male courts the female, swimming
around
her and displaying striped colouring of sides. He “leads” the female to rich
thickets of plants or to driftwood and trees fallen in water. Here both parents
clear a site of bottom of dust with the help of lips, and then female spawns
eggs directly on the ground. Among small-leaved plants adult individuals
arrange a kind of nest, whirling among stalks and trampling them down. The
female spawns
large eggs – up to 100 – 150 pea-sized grains of roe. Male fertilizes clutch
and at once banishes the female. He protects eggs, pasting them to his own
stomach and fin. First minutes after spawning eggs gets glutinosity and male
at once lay on them. Eggs stick to his skin and does not come unstuck even
at sharp movements. Protecting eggs, male becomes very cautious; he hides
in shelters and eats almost of nothing, except for small larvae casually
coming
almost right up to him. For batrachogymnotus the care of posterity of first
weeks after hatching is very characteristic. Male carefully protects school
of tadpoles, and they keep near to the parent. Tadpoles in first of all remember
his smell, but later begin to react to his appearance and movements. Male
drives predators away from them and digs bottom in searches of small invertebrates.
The young ones leave male at length of 2 – 3 cm. Back legs start to grow
at
them at the age of about half-year, at length about 15 cm. At the age of
2 – 2.5 years young individuals become capable to breeding. Life expectancy
of
batrachogymnotus may reach 20 years.
The idea about existence of this group of animals was proposed bt Timothy Morris, Adelaide, Australia.
New Zealand cave neosalamander (Cavernogyrinus caecus)
Order: Tailless amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae)
Habitat: underground reservoirs of New Zealand.

Picture by Alexander Smyslov
In Neocene at New Zealand the separate group of amphibians, New Zealand false
salamanders, prospers. They are descendants of Australian tree frogs of
genus Litoria introduced to islands, though all representatives of this
group resemble
externally any caudate amphibians of North Hemisphere. They had appeared
as the result of climatic changes at the boundary of Holocene and Neocene.
The
cold climate had caused the delaying of metamorphosis and occurrence of
group of neotenic amphibians keeping in adult stage the set of larval features.
In early Neocene their active adaptive radiation began, and as the result
of it,
at the islands many species of these animals had appeared. They are adapted
to various conditions of life, and one species, New Zealand cave neosalamander,
had settled underground reservoirs.
Cave reservoirs represent the habitat distinguished by poverty of food
resources. Therefore the majority of cave inhabitants is small creatures.
Besides the
basic sources of food for cave inhabitants are other animals, lower fungi
and bacteria, and also carrion. It considerably reduces opportunities of
settling
of caves by live organisms.
The predator having low rate of metabolism is the creature prospering in
such ecosystems. New Zealand cave neosalamander belongs just to such species.
This
is small animal about 20 cm long, with delicate translucent skin of “ivory”
yellowish-white color. At this amphibian there is thin long body; tail
makes about half of its general length. It is bordered with wide fin plica
stretched
on back almost up to the level of forepaws. Head at this species of amphibians
is flat, rounded, with wide mouth. This amphibian breathes mainly by gills
– lungs are very poorly developed.
Paws of New Zealand cave neosalamander are short and have three small toes.
This amphibian gets out on land seldom, preferring to swim in cold water
of underground rivers rich in oxygen. Eyes of this amphibian are completely
reduced
– on head of the animal only two dark spots instead of eyes are appreciable.
At New Zealand neosalamander even rudiments of optic nerve did not remain,
but there is a curious sense of “skin sight” – the animal reacts to light
falling on skin and tries to hide from it in dark places. Such phenomenon
was known
at caudate amphibians of Holocene epoch. Two short conic wattles in corners
of mouth help this blind animal in search of small invertebrates dug in
ground. Along each side of New Zealand cave neosalamander the line of lateral
line
cells passes. Very sensitive organs of lateral line help the animal to
define presence of moving objects in absolute darkness. Also for this amphibian
keen sense of smell is characteristic. Nostrils are surrounded with two
leaf-looking
outgrowths which permit to define the direction to source of a smell.
Due to so advanced sense organs New Zealand cave neosalamander occupies
high place in food pyramid of cave reservoirs. It eats crustaceans living
nearby,
and gathers insects fallen in water. The consuming of insects – plentiful
and nutritious food – permits this species to reach rather large number:
in large
cave lakes populations total some thousand individuals of these amphibians.
When in underground waters the dead animal from ground surface gets, New
Zealand cave neosalamander can eat carrion, finding it by smell and tearing
off small
pieces of food by mouth.
Seasonal prevalence in spawning at this amphibian is not present. Females
are ready to breeding depending on physical condition up to 2 – 3 times
once a
year. The female ready to spawning emits the odorous substance strongly
involving males. Near one female up to ten males of various ages and sizes
may gather.
Gradually stronger males drive off weaker ones, and soon only one male
starts to escort the female. Occasionally the applicant male keeps nearby
to them,
trying to wedge in courtship display.
These amphibians breed, spawning up to 250 small eggs in cracks between
stones. During the egg spawning male is twisted around of female’s body
and kept
on her, holding her paw. Pair may keep “embraces” till some minutes, and
then
simultaneous spawning of sexual products in water follows.
Eggs develop for rather long time – about three weeks. For this time the
part of them may be lost from predators, but eggs fallen in remote places,
develop
successfully. Small larvae of New Zealand cave neosalamander feed with
bacterial film and fungi, scraping the food from stones. They keep solitarily
and grow
slowly: legs develop at them at the age of about eight months. Larvae are
very cautious: their main enemies are crustaceans and adult congeners.
Young New
Zealand cave neosalamanders reach the sexual maturity only to 3-years age,
but can easily live up to 30 – 35 years.
The idea about the existence of this group of animals is stated by Tim Morris, Adelaide, Australia.
Swamp terragyrinus (Terragyrinus paludiphilus)
Order: Tailless amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae)
Habitat: New Zealand, cold Alpine swamps and marshy meadows.

Picture by Alexander Smyslov
Among Neocene inhabitants of New Zealand the family of “adult tadpoles”,
the separate group of amphibians had lost the adult stage has the special
rank.
Various species of these amphibians had occupied on islands the ecological
niches similar to those at tailed amphibians of northern hemisphere. One
species of these amphibians is swamp terragyrinus. The majority of its
relatives remained
aquatic animals, but this species spends on land much more time, than they
do. It is the inhabitant of swamps of Alpine zone overgrown with plants.
Swamp terragyrinus is very similar to large salamander. This is long-bodied
animal at which the tail makes about 2/3 of general length. The length of
this amphibian is about 20 cm.
At terragyrinus paws are well advanced, and this amphibious can freely move
overland. It lives in damp places - in swamps and lakes near to edge of mountain
glaciers where water and air are very cold. This amphibian lives among grass,
and due to thin flexible body freely creeps among stalks, escaping from enemies.
Head at terragyrinus is narrow and has pointed muzzle. Branchial apertures
are small; for protection of gills from drying they are closed by special
valves of skin. Eyes at this amphibian are small.
Body of animal has grey color with dark “marble” pattern; stomach is white.
In courtship season at males throat gets bright red color. Skin has very
important role in gas exchange – terragyrinus has small sac-like lungs which
can hardly
provide even a half of requirement of animal in oxygen. When this amphibian
dives, instead of lungs gills start to work. But the skin also provides animal
with oxygen. Terragyrinus does not endure rise of temperature of water, because
thus the contents of oxygen in it falls. In hottest days of summer these
animals leave land and hide in cold bottom layers of water of mountain lakes.
But terragyrinus
keeps activity even at minus temperatures (up to - 2 °С).
Terragyrinuses eat insects and small snails crushing their shells with the
help of special corneous plates on jaws.
The courtship season at these amphibians begins in spring when the rough
thawing of snow and ice takes place in mountains. After short court ritual
the female
lays on coastal vegetation large slimy mucous clod of eggs (about 500 ones
in clutch), covered with the common environment. All this time the male drives
competitors away from the female. It behaves very aggressively, and attacks
even fishes. When eggs swell, male penetrates into the clutch and ejaculates
sperm liquid. After fertilization of eggs both parents leave clutch.
The incubation lasts till about three weeks. Young ones are similar to tadpoles
of frogs and differ in translucent covers of body. Larvae of terragyrinus
live among vegetation and eat microscopic algae and microscopic animals.
At the
age of two years they reach the size of adult animal and begin able to breed.
Terragyrinuses have the slow metabolism and grow slowly. But they differ
in significant longevity – on the average, life expectancy of these animals
makes
55 – 58 years.
The idea about existence of this group of animals is proposed by Tim Morris, Adelaide, Australia.
Frogwurm
(Anuroserpens aotearoae)
Order: Tail-less Amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae), Subfamily: Legless
False-Salamanders (Anuroserpentinae)
Habitat: North Island and northern part of South Island, New Zealand, forested
and marshy areas with damp ground and abundant leaf-litter.
The evolution false-salamanders (Virilogyrinidae) from introduced tree-frogs
(Litoria) in New Zealand, proved to be an abundant radiation of new forms, some
of which differed greatly fro the ancestral species. The most modified of this
group are Anuroserpentinae, legless false-salamanders, elongate forms, in which
the frog-bodyform is modified and almost completely lost.
Typical form is the Frogwurm, a burrowing legless ampibian which is similar
in niche to Caecilians. Though it appears shaped like a worm or snake, only
the first third of its length actually consists of the body, reduced number
of vertebrae in ancestor caused this condition. The nine body vertebrae are
extended and more flexible than at its ancestor, hipbones are rodlike and elongate,
torso and hip region is well-muscled and tubular. Remaining length of body consists
of serpentine tail, smooth, cylindrical and muscular without a fin. Total length
including tail can be up to 37 centimetres, body minus tail is usually no longer
than 16 centimetres. Animal is a subterranean burrower in loose damp soil and
leaf-litter; it moves by writhing its muscular tail, wriggling its body and
pushing forward with its spade-shaped head. Head is cylindrical and flat, resembling
a narrow spade, eyes are small and weak, set forward. Teeth are mid-sized and
close-set and conical, small sensitive wattles adorn the upper lip. Prey consists
of worms, larvae and other soil-dwelling insects located by smell, touch and
vibration. Colour of body is bluish grey with a pale yellowish underside.
Animal is mostly solitary, it is most active during the night and early morning,
when the air is cooler. Breeding occurs during the wettest part of the year,
between May and September, female invites the attention of males my exuding
a trail of mucus secretions that have a distinct odour. More than one male may
court a female at once, and they will coil around the female, attempting to
mate. Fertilisation is internal, inseminated female gestates eggs for as long
as 9 months, before giving birth to up to 7 large, well developed young. For
as long as 2 months, the mother will remain with the young in the nest, she
produces loose growths of greasy shed skin which the babies feed on. After this,
the young become independent and the female leaves the nesting chamber, resuming
normal activities. Lifespan is up to 15 years, predators include carnivorous
birds, hedgehogs, mustelids
and occasionally large lizards.
This amphibian species was discovered by Timothy Morris, Adelaide, Australia.
Frog-siren
(Anurosiren natans)
Order: Tail-less Amphibians (Anura)
Family: New Zealand false salamanders (Virilogyrinidae), Subfamily: Legless
False-Salamanders (Anuroserpentinae)
Habitat: North and South Islands of New Zealand, marshes, ponds, slow-flowing
weed-choked rivers and streams, shallow lakes.
The false-salamanders of Aotearoa are an extremely diverse group, all descended
from tree-frogs introduced during the age of man. The most-modified are the
Anuroserpentinae subfamily, which converge on Caecelians and Sirens.
One notable form is the Frog-siren, an aquatic amphibian which is elongate and
legless, almost resembling an eel or siren. Both body and tail are somewhat
cylindrical, partly laterally flattened, tail consists of 2 thirds of the entire
length, and has ridge-like fin running its length above and below. Full length
including tail is up to 55 centimetres, of which only a third consists the tubular,
legless body. Head is cylindrical and slightly pointed, jaws are moderately
powerful, teeth are close-packed and pointed. Eyes are of moderate size, set
forward on the head, eyesight is fair, sensitive to movement. Body coloration
is a muddy greenish-brown, underside is pale yellowish cream colour. Animal
typically inhabits marshes, ponds, slow-flowing weed-choked rivers and shallow
lakes. Animal usually moves by swimming in an eel-like manner, sinuous lateral
undulations, it is able to crawl overland in the case that it needs to find
a better habitat. Prey consists of small fish, frogs, aquatic arthropods, frog-spawn,
cannibalism on smaller individuals occurs.
They are usually solitary, but will tolerate each-other during overcrowding
of shallow waterways. This species is mostly diurnal, more active in the morning
and afternoon.
Breeding occurs during periods of higher rainfall, May to September. Females
advertise receptiveness by exuding mucous secretions that have a particular
chemical odour. Multiple males may attempt to court a female at once, wrapping
around her in an attempt to inseminate. Insemination is internal, males have
a penis-like organ, female is pregnant with eggs for 2 or 3 months, then laying
a sticky tangle of up to 10 large gelatinous eggs in a secluded patch of aquatic
vegetation. Female broods eggs until they hatch, which may take as long as 2
months. Young are exact miniatures of the adults, fully independent, and begin
feeding immediately. Lifespan may be up to 10 years, predators include water-birds,
large fish, other kinds of amphibian,
and aquatic snakes.
This amphibian species was discovered by Timothy Morris, Adelaide, Australia.