

Bestiarium of
the future - Invertebrates
|
 |
| In this section descriptions of various animals and plants, which could live on the Earth in Neocene epoch. The section will be supplemented as new ideas about possible ways of evolution of life will appear. If readers will not find here any species placed here earlier, it means that it is a reason to search for a new chapter in English version of "The Neocene Project". |  |
|
Invertebrates |
Tunicates
|
“Amphitrite’s
beads” (Doliophora amphithritae)
Order: Doliolids (Doliolida)
Family: Colonial doliolids (Doliophoridae)
Habitat: Indian Ocean and Tanganyika Passage, top layers of water.
Salps, pyrosomas and doliolids are close relatives of vertebrate animals. But
it is difficult to believe in it to unsophisticated observer. At the first sight
at these creatures it can seem, that they are nearest relatives of jellyfishes:
they are fragile and transparent too. But features of embryonic development
evidently show that they are representatives of chordates (Chordata).
The body of these animals has turned to true pomp which tirelessly pumps over
water through filtering device of huge gullet. Due to it animals can move in
thickness of water, similarly to rockets. Some representatives of invertebrate
chordates form colonies numbering from two zooids up to several thousand ones
at the bottom of ocean or in thickness of water.
“Amphitrite’s beads” is original colonial species of doliolids order, usually
including species, existing as singles in adult condition. For usually single
animal transformation into the whole colony is very hard process. But invertebrate
chordates have one special feature, permitting to carry out such transition
easily. In life cycle of these creatures there is the alternation of generations:
sexual, breeding by egg laying, and sexless one, breeding by gemmation. It is
enough for the individuals developing from gemmas “to be late” in this condition
longer and also to become adults quicker and the colony is ready in general.
So, obviously, it also had taken place at ancestors of doliolid “Amphitrite’
beads”.
This colonial animal has not lost features of ancestral appearance: it is also
ephemeral, watery and transparent. The colony of this species consists of several
types of zooids (individuals being as though “bricks” of colony) having had
partly lost independence. In the top part of colony six large mover individuals,
gathered in one “block” settle down. This is the posterity of initial zooid
developed from eggs. “Founder” is at top of all colony.
When the colony was young, mover zooids had well advanced filtering device and
could support their own existing. But in due course the colony expands, and
the speciality to move colony is “laid on shoulders” of these individuals. In
due course at mover zooids filtering device is reduced, but powerful ring muscles,
strengthening jet push, develop. They strongly increase in size in process of
colony development. Besides on external (concerning to other zooids) surface
of each mover zooid the special keel develops, permitting to keep direction
of movement.
Long double chain of eating zooids numbering up to fifty individuals branches
off from the bottom mover zooid (bottom zooid does not have keel, and runner
with zooids of other type branches off from its back side inverted downwards).
Due to it all colony is similar to hanging down string of beads. Eating zooids
have well advanced filtering device: their duty includes supplying by food of
all colony. By common channels of blood system all individuals in colony are
supplied with nutrients acquired by these zooids.
For breeding at the basis of eating zooids from special gemmas strongly reduced
fertile individuals grow. At them the digestive system is reduced, filtering
device is underdeveloped, and they completely depend on nutrients delivered
by other members of colony. But at them ovaries, in which some large eggs ripen,
are advanced.
Inside eggs larvae develop, right in eggs turning to single sexless individuals.
These future founders of colony hatch and also live solitary life for some time.
Then on the bottom surface of single individual some gemmas appear, from which
additional mover zooids grow, and single individual turns to basis of the future
colony. Such “underdeveloped” colonies can be mistaken for separate species
of live creatures, but it is only special stage of development. On the bottom
side of body of the bottom mover zooid special outgrowth starts to develop,
and in body a plenty of gemmas, migrating to this outgrowth, is formed. They
settle down on it in two longitudinal lines and turn to eating individuals.
And later to this outgrowth new generation of gemmas migrates, turning to breeding
fertile individuals.
In rest the colony of “Amphitrite’s beads” dangles in water almost vertically:
mover zooids by rhythmical contractions of ring muscles slowly move colony in
water, and thick runner on which other members of colony sit, hangs downwards.
In case of need other members of colony may be included in movement. “Amphitrite’s
beads” is inhabitant of top layer of water: colony keeps in layer of water,
rich in phytoplankton. But, having felt beginning of storm, fragile colony swims
down to depth up to 50 meters. Storm and sea inhabitants frequently tear this
fragile colony to pieces, but it does not harm to “Amphitrite’s beads”: colony
easily regenerates. Usually the colony is broken off in area of runner. Then
on that part where there are mover zooids, simply the formation of new gemmas
begins, and the colony is restored till the initial size. On torn off runner
eating zooids sitting closely to place of break, gradually turn to mover zooids.
Such recycled colony differs from initial only that on its top part there is
no initial zooid from which the development of colony had began. And function
of formation of gemmas for growing colony at once two bottom zooids have turned
to movers incur.
In Tanganyika passage where strong gales are rather rare, intact colonies of
these creatures frequently may be met, but in Indian Ocean approximately two
thirds of colonies have signs of damages.
This species of doliolids has ability to light: at all members of colony, except
for fertile zooids, organs of luminescence develop. These are small sacs, located
near ring muscles, and containing inside luminous bacteria. The colony of “Amphitrite’s
beads” can shine greenish and bright blue light.
Whale
salpa (Titanosalpa crystallocetus)
Order: Salps (Salpida)
Family: Giant salps (Titanosalpidae)
Habitat: cold subpolar waters of Southern hemisphere, circumantarctic species.
Primitive chordates represent the original transitive group between invertebrates
and vertebrates. There is an opinion, that vertebrate animals had descended
as the result of loss of adult stage by larva of lower chordates (this phenomenon
is named pedomorphosis). Adult individuals of lower chordates may appreciably
differ from larvae of the same species, and during the life cycle two completely
unlike against each other generations may be replaced.
Lower chordates of Tunicata subtype are very similar to jellyfishes and their
relatives – at them there is the same transparent body, which significant part
is made of water. But this similarity is only external – tunicates essentially
differ from jellyfishes by features of structure and development.
Pelagic tunicates are active filtrating organisms passing through the body
hundreds of litres of water per day. During the extinction of cetaceans and
large sea fishes, in crisis epoch at the boundary of Holocene and Neocene,
these invertebrates had got the advantage in impoverished ecosystems of open
ocean. Primarily among them many large species of filtrating organisms evolved.
But the majority of them had become extinct later, when in seas true pelagic
fishes had appeared again. But nevertheless some species of giant tunicates
had survived in subpolar areas, rich in plankton.
Whale salpa is one of such huge species of solitary tunicates, a relict of
early Neocene. It represents an animal up to 4 meters long at deep of body
about 2 meters and width a little bit more one meter. By its appearance whale
salpa resembles more the case of ancient sailing ship without masts: its sides
are slightly compressed, bottom part of animal is extended, and on it gristle-like
“keel”, helping to keep stability, is developed. The consistence of body of
whale salpa is rather fragile, and covers are translucent; therefore animal
seems made of glass. Ring muscles constantly and rhythmically contracting,
passing water through the body of animal, are visible through covers of body.
Such large animal could not survive, not having even any similarity of skeleton.
At whale salpa the half rings of dense tissue, covering body from sides, develop.
From above they are strapped with easily extensible ligament, and the bottom
ends of these formations merge with keel.
Due to rather fragile body consistence for this animal ability to feel approach
of storm is vital. Long before approach of bad weather whale salpa submerses
to depth and motionlessly hangs in thickness of water. This animal is the sluggish
swimmer, and its covers are frequently injured by various animals. But whale
salpa, similarly to the majority of relatives, can easily repair injured by
storm or predators parts of a body. The surface of tunica of this species is
overgrown with small symbiotic polyps. They slow down movement of whale salpa
in water, and partly compete to it for forage, overtaking a part of plankton,
but there is a considerable benefit to whale salpa from such union: polyps
protect animal from enemies with the help of burning tentacles. Some pelagic
fishes and invertebrates lay eggs on covers of body of animal, because of what
old individuals of whale salpa turn to true swimming “kindergarten”. In oral
siphon of whale salpa small crustaceans – symbiotic scuds – frequently lodge;
in cloacal siphon parasitic isopods frequently live.
As at all tunicates, at whale salpa there is an alternation of generations
in life cycle. Single individuals of whale salpa represent sexless generation
hatching from eggs. These giants reach the maximal size approximately for two
years, and after the attainment of full physiological maturity make the colonial
generation sharply distinguishing from single individuals externally. On the
bottom side of body of whale salpa, on front edge of keel the runner develops
– the outgrowth on which the colonial generation of this species is formed.
On runner some whorls of gemmas are formed, from which young individuals of
sexual generation develop. The young growth is formed and matures in “clusters”
of 2 – 3 pulled together whorls of 6 – 8 individuals in every one. Runner reaches
the length about one meter. When young individuals on its tip reach the size
of plums, runner is broken off right above them, and young ones swim out from
adult individual. Within several months they reach the length about 20 cm (the
length of colony of such individuals may be up to half meter), and become able
to breeding. The colony of sexual generation of whale salps makes up to 20
large eggs per one week. In the common cavity of colony eggs develop, and already
generated young individuals up to 15 cm long, the single sexless generation,
leave the colony. They grow to giants which have determined the name of this
species of animals.
Whale salps reach the utmost size in cold, rich in plankton waters of Subantarctic
region. Sometimes currents flowing to the north along the coast of Antarctica
and South America, carry away individuals of this species in tropical latitudes.
In warm water this animal does not breed and lives not for a long time. In
tropical latitudes whale salpa quickly grows old, but even faster it becomes
prey of heat-loving pelagic predators. More often these salps are eaten by
huge bat turtles.
Echinoderms
|
Armour-bearing
starfish (Ruderasterias litophoros)
Order: Forcipulatida (Forcipulatida)
Family: Asteriidae (Asteriidae)
Habitat: Europe, a littoral zone of northern seas.
In Neocene the inclination of continental plate of Eurasia has changed: glaciers
haв stopped to press it with the whole weight, and northern edge of continent
had risen a little. It has had an effect on geography of continent: the rivers
of Asia from Arctic Ocean basin spill widely now and flow much slower, and
in Europe Northern Sea had receded, having exempted extensive land areas. The
northwest extremity of Europe forms the coastal line cut up capriciously with
sea gulfs. Along the coast there are numerous low islets and shallows, overflowing
by sea, and also the numerous marshes overgrown with grasses steady to sea
water. In this world the creatures capable to survive both in water, and on
land prosper.
Among littoral inhabitants there are various echinoderms. Being sensitive to
change of water salinity, they avoid river mouths and keep in pools of sea
water at the coast. Here they are vunerable for birds searching for food at
the shoaliness. Therefore in due course of evolution they had developed various
protective adaptations promoting their survival.
At the coast of Europe, in area of temperate climate, the armour-bearing starfish
lives; this one developed interesting strategy of masking. In Holocene epoch
such feature was more characteristic for crabs, but in Neocene this echinoderm
has partly repeated it. Common starfish (Asterias rubens), widely settled in
human epoch in Northern Atlantic, was an ancestor of this species.
Armour-bearing starfish is a radially symmetric animal about 1 foot in diameter.
The body of this animal has small central disk and mobile flexible arms. Colouring
of top (aboral) side of body is spotty – on bright yellow background large
red or brown spots are scattered. The bottom (oral) side of body is white.
The structure of ambulacral tube feet of this echinoderm is unusual. Along
each arm on the oral side of body two lines of tube feet of usual structure
stretch; they serve for movement. And on the top side of body in intervals
between arms and along the edges of arms the large mobile tube feet, capable
to grasp and to keep small objects are developed. With the help of these tube
feet the armour-bearing starfish keeps on itself the various dust picked up
on sea-floor. It gives obvious preference to flat firm objects like bivalve
shells and large flat pebbles. Animal gathers these objects, creeping on bottom.
Having found suitable object, starfish feels it with grasping tube feet. If
it may be lifted, animal sticks to it and tightens it on itself. In total this
animal can keep on itself up to twenty pebbles and shells.
Objects kept by armour-bearing starfish on its body help it to mask and complicate
an attack of predators as the attacked starfish only keeps the seized objects
stronger. In addition to it this species lives at the areas with strong ebb-tide
current, therefore stones help armour-bearing starfish to stay in its place.
This species eats molluscs and other invertebrates, not differing in this respect
from its ancestor.
This species breeds with the help of the planktonic larva, as well as all related
species. Fertility reaches 1 million tiny eggs. The larva lives in plankton
for about three months, and then turns to bottom-dwelling stage. Young armour-bearing
starfishes keep in quiet sites of littoral zone and mask, keeping on their
bodies parts of seaweed and other light weighting objects. In process of resettlement
to the sites with stronger current they choose heavier objects for masking.
At the sites with weaker current smaller species lives – shield-bearing
starfish (Ruderasterias scutifer). This animal does not exceed 15 cm in diameter. It
has similar anatomy of the body, but aboral side of body has yellowish-white
color. This animal carries on its body large flat cockleshell of bivalve, under
which it hides from enemies. Being attacked by predator, shield-bearing starfish
keeps shell strongly and sticks to substratum, not giving an opportunity to
hook the shell from the edge. The same way it spends outflow, having densely
covered by shell and having hidden under it the body from wind and the sun.
This species lives at the the top border of littoral zone and frequently stays
outside of water.
Life expectancy of both species does not exceed 5 – 6 years.
Surprising
planktostella (Planktostella inexpectata)
Order: Larvasterida (Larvasterida)
Family: Plankton starfishes (Planktostellidae)
Habitat: temperate latitudes of Southern hemisphere.
“Planktonic accident” at the boundary of Holocene and Neocene had caused significant
updating in structure of World ocean zooplankton. In Neocene among planktonic
organisms there are representatives of various groups of animals, including
such ones, which had not existed in human epoch.
The echinoderms, developing with larval stage, spent a short part of life cycle
in plankton. Their larvae – doliolaria, ophiopluteus, echinopluteus, bipinnaria
etc. – lived for some time in plankton, but then had a deep metamorphosis and
turned to benthic adult stage, looking completely unlike the larva. But in
Neocene one group of starfishes of order Asterida began to spend in plankton
considerably longer time. It was connected to presence of free ecological niches
in planktonic community. The planktonic larva, brachiolaria, had actually turned
at them to independent stage of development like asexual generation of tunicates.
This way the separate larvasterid order had appeared among starfishes. As against
microscopic brachiolariae of ancestral forms, the larva of larvasterids is
long-living (its term of life makes more than 2 years) and reaches great length
to measures of larvae: up to 3 – 5 cm. But simultaneously it keeps a typical
anatomy of larvae, eats tiny planktonic organisms and swims actively with the
help of numerous bristles. As against brachiolariae of typical starfishes,
it produces star-shaped adult stage repeatedly – under covers of brachiolaria
a row of germs (imaginal disks) is formed, giving rise to new starfishes.
The adult form of starfishes of this group serves for breeding. It leads planktonic
way of life and eats small animals. In connection to change of habit of life
the anatomy of planktonic starfishes had changed in great degree in comparison
with their ancestors. The body of these animals is flattened in oral-aboral
direction. Long ambulacral tube feet border flattened arms. They serve for
soaring in water, and also grasp small planktonic animals transporting then
into the mouth of animal.
In internal anatomy of the animal also great changes had taken place. The part
of ambulacral systems of planktostellids is separated and turned to gas floater
bladders, arranged in double line along each arm of starfish. Gas glands fill
them with nitrogen and the carbonic dioxyde taken from sea water.
The armour is reduced in connection with planktonic way of life; only unary
line of sharp plates along each arm remains from it. The body of animal keeps
an elastic consistence, but is semi-transparent. Planktostellids are poisonous.
As additional means of protection at them autotomy and ability to regeneration
are developed. The seized starfish easily throws away one of arms which wriggling
distracts attention of predator. The lost arm easily grows back, and on the
torn off arm due to available nutrients missing ones grow, and it turns to
complete organism. The central disk at these stars is very small, and arms
in typical case are expanded in middle part and resemble lily petals in shape.
Surprising planktostella is large starfish reaching almost one meter in diameter.
It swims, making synchronous waves by all arms (normally there are 5 arms at
this species, but there are separate four- and six-armed individuals). Despite
of the size, it is the species leading the least predating habit of life. At
feeding it turns stomach out in water and soaks up organic chemical substances
dissolved of water with its surface, and also catches the smallest plankton
which is pasted to the surface of the turned out stomach. Thus the star keeps
immovability and drifts passively in current.
The stomach of surprising planktostella forms a set of pockets of 1-st, 2-nd
and 3-rd orders, which, being turned out, turn to root-like outgrowths. Due
to it the summary surface of stomach is increased. Bowels at this species,
on the contrary, are very short.
Breeding of all planktostellids has no expressed seasonal rhythm. These animals
are hermaphrodites, and two genetically different individuals mutually fertilize
each other. If two individuals being clones of initial one meet each other
casually, fertilization does not occur – this species is self-sterile. When
eggs develop, the starfish spawns them in water, and from them larvae hatch,
which grow till two months and further pass to asexual breeding.
Waters of World ocean are also inhabited by other species of planktostellids:
Jellyfish-eating planktostella (Planktostella medusiphaga) lives in subtropical
waters of Southern hemisphere. Diameter of this animal is about 40 cm; usually
it has 6 – 7 arms. This species eats jellyfishes and other soft-bodied pelagic
animals (including even small cephalopods) with the help of the stomach turned
inside out. The stomach of this species forms a number of suckers which keep
prey. Usually this star drifts in thickness of water, having turned out its
stomach opened as an umbrella with suckers on its internal side. When prey
casually touches it, the star seizes the prey by sucker of stomach and envelops
it with the whole stomach, starting the digestion. If prey shows resistance,
the starfish puts all arms together, keeping the caught animal between them.
The starfish gradually involves prey in mouth in due course of digestion.
“Sea sundew” (Planktostella droseroforma) is a small species about 10 cm in
diameter with five-armed body. It is the species actively hunting planktonic
crustaceans, worms and molluscs with the help of ambulacral tube feet. Arms
of this species have the ribbon-like shape, dulled on the tip. They are bordered
with numerous ambulacral tube feet capable to extend to the distance up to
2 cm. The tips of tube feet are supplied with the chemoreceptors catching presence
of crustaceans. Having felt prey, the tube foot extends in its side and sticks
to the armour of crustacean. The mechanical irritation serves as stimulus for
activity of the near feet, and they gradually transport prey to the mouth of
animal. This species inhabits temperate and cold waters of Southern hemisphere
and is circumantarctic.
Insects
|
Giant
Cave Springtail (Cavernella spp.)
Order: Springtails (Entomobryomorpha)
Family: Isotomidae (Isotomidae) Subfamily: Giant Cave Springtails (Cavernellinae)
Habitat: New Zealand, North Island, walls and floors of the depths of certain
caves. Numerous similar species within this genus inhabit different cave systems.

Picture by Biolog
Caves provide areas of isolation that allow speciation and
vicariance of animals much like islands or other natural barriers. In New Zealand
we see similarly, caves host specialized forms of invertebrate living in the
depths, such as the Giant Cave Springtail.
This insect is relatively very large for a springtail, up to a centimetre long.
It is more sedentary than its ancestor, flatter and heavier in shape, and its
ability to utilize its tail “spring” is less in agility. Cuticle of exoskeleton
is relatively thin, body, legs are relatively longer than its ancestor, tail
“spring” is shorter. Insect is pale opaque white in color, as with many troglobiota.
Antennae are relatively long and finger-shaped to aid in tactile sensing of
surroundings, springtail feeds mostly on fungal and algal films that form near
sources of water, nematodes and other microfauna as well as rotting organic
matter and carrion.
This species reproduces by producing small spermatophore droplets which females
seek out and fertilize. Eggs are laid in secluded crevices and are vulnerable
to desiccation. A female may lay as many as 350 eggs per year, year round. Babies
are tiny replicas of their parents, apart from being much smaller, and almost
completely transparent, maturity is reached in about 4 months. Springtails are
predated upon by cave-dwelling mites, beetles and harvestman. This genus is
restricted to certain cave systems in the North Island, with different species
restricted to individual caves, but being nearly identical outwardly.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Stinky
cave cockroach (Cavernoblattula foetida)
Order: Cockroaches (Blattodea)
Family: Cockroaches (Blattidae)
Habitat: caves of the Balkan and the Alpes.

Picture by Biolog
Cockroaches existed always and everywhere, up to times of
dinosaurs – having appeared on the Earth in Carboniferous, they strongly kept
their place in ecosystems of all subsequent geological epoch. They always rustled
under legs of larger creatures, eating various organic dust and dross. Before
human epoch cockroaches prospered in areas of warm climate, and only few small
species had penetrated far to the north. Expansion of communication and trade
since Middle Ages has allowed to cockroaches of some species to expand considerably
the area and to settle almost on global scale.
Despite of it, synanthropic cockroaches had not become rather hardy insects,
and at the extinction of people as a biological species cockroaches had receded
from temperate warm and cold areas too. Nevertheless, Holocene epoch and human
domination had not passed without results for cockroaches: they managed to stay
at the south of Europe, having evolved to some specialized species. In caves
of the Balkan formed after receding of glaciers, some underground species had
appeared, one of which is stinky cave cockroach. Its ancestor was, obviously,
oriental cockroach (Blatta orientalis) – the species widely settled in human
epoch.
Stinky cave cockroach is rather sizable insect: sometimes there are individuals
of this species up to 6 centimeters long. It has flattened oval body of brown,
black, or grey colouring, covered with oily liquid, plentifully secreting by
special gland. Regularly secreting a small amount of liquid, this cockroach
pounds it on the top part of body by back pair of legs. Oily liquid is its most
important protection, especially if the cockroach can not be escaped from the
enemy by another way. This liquid smells very unpleasantly and has bad taste;
the scared insect secrets it in plenty. Therefore the majority of rather large
predators living in caves prefers not to eat cockroaches. And from small enemies
the disturbed cockroaches usually seek safety in flight – they run quickly and
dexterously swarm up walls of cave. Stinky cave cockroach does not have wings.
Sight sense at adult insects is very bad, but young cockroaches have well advanced
large eyes, which are reduced with each subsequent moult. But instead of them
long antennas develop, which exceed body length at adult insect.
This is unpretentious insect developed a special way of surviving in winter
colds – when winter comes, cockroaches are dug in layer of bird and bat guano,
and run into anabiosis till spring. It has allowed them to settle in systems
of caves far to the north – to the area of seasonal climate. In caves of the
Balkan located in area of rather warm climate, these insects keep activity the
year round.
Cave cockroaches can not dispense for a long time without water – it is used
for secretion of liquid covering their body. Therefore they should run out from
cave at night to drink from pools, if the rain took place recently, or, on the
contrary, to go down to the depth of cave, if there is an underground reservoir
there.
Stinky cave cockroaches are not fussy – they are ready to eat any organic material,
including each other. Usually they eat dung of bats, small worms eating dung,
sprouts of seeds belched by birds nesting in cave, and dead animals of any species.
Some times per one year the female lays up to 20 eggs made in special capsule,
an ootheca, and carries them on its abdomen till about two weeks. Then it leaves
an ootheca in secluded place where young cockroaches leave it one week later.
In northern parts of area this cockroach makes no more than two clutches per
one year. Development from the bursting from egg up to adult individual lasts
about three months. The majority of cave cockroaches perishes in youth, when
they yet have not reached the size of adults, and their chitinous covers is
much softer. They produce less amount of odorous liquid, and rely more in their
self-protection to speed of legs; the main enemies of cockroaches are other
cave insects, like beetles
and soothsayers.
Young cockroaches settle from caves, creeping out to the forest. But they frequently
use “services” of birds and bats living in their native cave. Clinging to their
body, young stinky cockroaches have an opportunity to appear in other cave.
Sometimes they appear in forest, and can live any time like usual cockroaches
living there. But they search by smell for entrances in caves and cracks, and
occupy them.
This species of insects was discovered by Bhut, the forum member.
Scatophagous
Cave-roach (Scatoblatta chiropterophilus)
Order: Cockroaches (Blattodea)
Family: Cockroaches (Blattidae)
Habitat: New Zealand, caves containing bat colonies, entrances of caves.

Picture by Pavel Volkov
The various verminous insects brought to New Zealand by humans
had resulted in many new species, some having a specialized lifestyle. Descendants
of the American cockroach (Periplaneta americana) came to inhabit many parts
of New Zealand, including caves.
Scatophagous Cave Roach is one such example, a flightless roach feeding almost
entirely on bat dung. It has changed in appearance from its ancestor, body is
broader and legs have robust spiny shape to aid digging in piles of guano. This
roach is flightless as an adult, but retains short wings. Like its ancestor
it is relatively large, reaching up to 5 centimetres long and 3 centimetres
across. They form large congregations under the sites of bat colonies, piles
of dung may appear to move with the motion of digging and feeding roaches. Carapace
and legs are a uniform dark brown, slightly darker above.
Eyesight of the adults is somewhat weak; eyes are smaller in adults than young,
which can see well. Insects are active most times of day, becoming more agitated
by day when bats occupy the cave and actively produce dung. It gains most of
its water from its food, but it may suck liquid from dead bats and occasionally
scavenge small portions of carrion.
Females lay a large ootheca of up to 30 eggs, which they carry with them for
a week and a half, young develop quickly, and females may make up to 3 clutches
per year. Eggs take about a week to hatch, and young reach full size after two
months. Both young and adults are commonly preyed upon by predatory beetles,
earwigs and crickets. Lifespan may be up to 30 weeks.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Elephant
Cockroach (Gigantopanesthia hoplitochelys)
Order: Cockroaches (Blattodea)
Family: Blaberids (Blaberidae)
Habitat: Tropical rainforests of northern Meganesia, avoids swampy coasts of
Carpentary and Arafura lakes.

Picture by Biolog
Among native insects of Australia of Holocene epoch, apart
from more charismatic beetles and butterflies, giant litter dwelling roaches
are notable. One large species, the Queensland Giant Cockroach (Macropanesthia
rhinoceros) was common in subtropical forests and even able to be kept as pets
by humans. From this species, the Elephant Cockroach descends, which inhabits
the leaf litter of northern tropical rainforests in Meganesia.
Even larger than its ancestor, this roach reaches 12 centimetres in length and
45 grams in weight. It is a thick-shelled, wingless cockroach which burrows
in the leaf litter and soil, consuming fallen and rotting leaves. Unlike its
ancestor, which fed mostly on dry eucalypt leaves, Elephant Cockroach is unpretentious
in feeding and will process many different kinds of rainforest leaves.
The cuticle of this roach is even thicker and more shell-like than its ancestor,
hence the specific name “hoplitochelys”. Margins of its carapace flare out from
the body, and when discovered by a predator, it will attempt to bury itself
so deep that it cannot be extracted. The pronotum is shovel shaped and aids
greatly in digging. Its legs are thick but short, covered in large spines which
aid it in digging as well as defence. This roach is usually a dark black colour,
but females may be chocolate brown instead. Immature stage nymphs emerge pale
but as soon as their shells harden they become a dark brown.
These insects are important components of the ecosystem, as they constantly
aid in decomposing fallen leaves. They may dig as deep as 1.2 meters in the
soil, forming deep burrows which are their permanent home, where they store
and consume leaf litter which they have gathered. The burrow is lined with dried
plant matter and is used to compost the leaves which it eats. The composting
chamber is large, up to a meter wide, large amounts of leaves are composted
and consumed there. Entrance to the burrow forms a dish-like semi-circle but
is often obscured by plant litter, and the shaft of the burrow may stretch up
to a meter.
Some social behaviour is seen, as nymphs remain with their mother for some months.
Males are solitary and often fight when they meet, disputing over feeding territory.
Fighting consists of pushing, barging, and hissing in an attempt to flip the
opponent over. These roaches are active mostly at night, searching for dead
leaves which they drag back to their burrows. Breeding also occurs at night;
males groom and nudge the female in an attempt to court her. Females may mate
once or multiple times, and are ovoviparous, eggs are incubated internally and
reared in a maternal burrow away from the male. The mother cares for the nymphs
until they mature, which can take up to 6 months. Brood size is up to thirty
nymphs which are produced in spring after an incubation of about 3 months. Life
cycle consists of shedding their skin about 14 times. Lifespan is up to 10 years.
When threatened, this roach can emit a loud hiss to startle a predator. Species
which prey on this animal may include large lizards, omnivorous marsupials and
occasionally birds, but the roach is generally bad tasting and not preferred
food.

Picture by Biolog
A close relative is the Southern Digging Cockroach (Gigantopanesthia brunnus), which occurs in the subtropical woods of Southeastern Australia, and actually more closely resembles the ancestral type, as it mostly feeds by processing dead eucalyptus leaves. It is about 7 centimeters in length and about 27 grams in weight. Physical differences from the nominal species are mainly size and colour – brown, but the carapace is also more rugose.
This species of invertebrates was discovered by Timothy Donald Morris, Adelaida, Australia.
Modest
dung termite (Apteroregina modesta)
Order: Termites (Isoptera)
Family: Termites (Termitidae)
Habitat: savannas of Northern Africa.

Picture by Ilia
In Neocene extensive areas of Northern Africa, Western Europe
and the former Mediterranean region represent savanna which provides with food
numerous herbivorous mammals. Among them the significant place is occupied by
descendants of hyraxes – flathorns
and ndipinotheres.
They had inherited from their ancestors a behavioural habit to leave manure
in the same place every time. In due course in “lavatories” of these animals
high heaps of manure are formed, and around of them the special ecosystem of
set of species of invertebrates and mushrooms utilizing manure of these animals
is formed. Among the dung-eating insects termites occupy the important place;
colonies of these insects usually settle down under manure heaps, and thickness
of manure is penetrated with their tunnels.
One species of termites making these ecosystems is modest dung termite, very
small species of termites. The length of adult ergate (working individual) of
this species is only about 4 mm, and adult “queen” does not exceed 3 – 4 cm
in length. The foundator male is also rather small – about 5 mm long only. Only
“soldier” individuals reach the length of 6 – 8 mm including large head and
massive mandibles. This species of insects differs from others in short term
of life of colony – no more than three years, but usually about two years only.
It is connected to rather small term of existence of manure heaps in which they
live.
Covers of these termites are very gentle and translucent, colored creamy white.
Head capsule of black color and wide brownish mandibles represent the firmest
parts of body of these insects. Modest dung termite lives only in thickness
of manure heaps and under ground, in conditions 100% humidity of air. This species
of insects settles in heaps of flathorn manure.
Symbiosis of this species of termites with large dung beetle, “termite
bus” beetle, is remarkable. Grubs of this dung beetle of various ages constantly
live in colony of termites. Grubs are suppliers of building material – their
dung at drying gives durability to walls of tunnels and galleries which termites
make. Even after complete decomposition of manure heap the rests of constructions
of this termite are kept as tubes with porous walls, sticking out of the ground.
Termites also eat dung of the grub, taking from it vitamins necessary for them,
which are made by bacteria living in intestines of the beetle.
Modest dung termite lacks clearly expressed period of mass flight of winged
foundator individuals. But every time in colony both winged males, and the females
ready to fertilization are present. Males of modest dung termite regularly leave
termitary in small groups and search for females with the help of smell sense
like males of termites of other species. The female, foundress of new colony,
at modest dung termite lacks of wings: it saves the resources necessary for
the foundation of new colony. Settling of females happens in unusual way. The
female, constantly surrounded with some ergates, waits while “termite bus” beetle,
having finished metamorphosis, leaves a cocoon where the transformation took
place. When the beetle starts to dig a tunnel to the surface, the female of
termite with several ergates creeps in its prothorax where there is a deepening
protected by beetle’s horn from the front side. During the beetle’s flight the
female of termite emits pheromone, involving males. Male fertilizes her right
on back of the beetle and at the same time ergates from termitary of the female
break off his wings. The beetle carries a colony to new place – to fresh heaps
of manure of flathorn. When it is dug in manure, termites on its back appear
in safety and start to build a nest at once.
Sailing
damselfly (Antarctovelum navigator)
Order: Dragonflies (Odonata)
Family: Damselflies (Calopterygidae)
Habitat: freshwater reservoirs of Antarctica.

Picture by Biolog
In Neocene the territory of Antarctica started gradually free
of ice cover. In polar areas glacier still exists, but edges of continent already
represent tundra with rare bush islets. Plants and animals of Antarctica represent
a mix of descendants of sea species and the species had got to the continent
by air. Among insects of Antarctica dragonflies are very typical. Due to great
ability to flight they could cross ocean and settle in this rigorous world where
they had few competitors, but a great number of prey like other species of insects.
Some dragonflies of Antarctica in larval stage occupy an ecological niche of
small fresh-water fishes and form an original group of fish dragonflies in damselfly
family. Imago of these insects does not differ in way of life from typical dragonflies,
except for one species which had lost ability to flight.
During the Antarctic summer on surface of water of Antarctic lakes the genuine
flotillas of original creatures appear – these ones are rather large damselflies
drifting on surface of water, having lifted wings upwards, as if sails. This
insect is sailing damselfly, the species of dragonfly order, not capable to
fly. This species has rather large size – body length of adult individual is
about 15 – 18 cm. The large size of insect is an exaggeration in many respects:
the abdomen of this dragonfly is very thin, and thorax seems large because of
two floater air bags developed on each side of the body. The adult individual
of this species leads rather passive way of life – it is a filtrator of tiny
planktonic animals. Using wind power and operating position of the wings as
if sails, it floats passively on the surface of water. As the requirement for
oxygen at this one is low, it does not choke, and tracheal system quite successfully
supplies this insect with oxygen. Under water this insect has similarity of
sea anchor made of rather long legs covered with spikes and long bristles. They
stabilize the position of insect in water and do not give it to be overturned.
At strong wind sailing damselfly simply folds wings along the abdomen. Wings
of this insect resemble a little a yacht sail in their shape – they are strongly
expanded in the bottom part and smoothly narrowed to the tip. Some strong nerves
stretch from front edge of wing to back one, providing its elasticity and resistance
to wind.
For adult dragonflies gnawing oral parts with well advanced mandibles are typical.
Sailing damselfly in adult condition eats tiny animals, and the typical oral
parts of dragonflies are not suitable for it. During the evolutional development
of this species of insects the partial neoteny has appeared, which had made
accessible such way of feeding: adult insect keeps the larval oral parts with
the advanced lower lip – so called mask which can fold on the bottom side of
body. The terminal part of lip is expanded and covered with rich hairs on edge,
and on its edges palps covered with bristles on the internal side are developed.
Movements of this lip gather zooplankton in thickness of water and bring it
directly to mouth. Thus palps are widely opened, increasing a trapping surface
of mask. And if the larva of mosquito or other small animal is near, the lip
of dragonfly works as a hand: tentacles instantly snap, as if a trap, and keep
prey.
Filtrator at this species is only the adult individual. The large larva is an
active predator, keeps in thickness of water and in thickets of underwater plants,
and catches larvae of others fish dragonflies. Sailing damselfly lives the most
part of life as the larva; development takes about two years, and in cold areas
of Antarctica it lasts for three years. Only at the end of life for some summer
months it turns to imago and is coupled. Imagoes live not for long and perish
already in second half of summer when the amount of plankton in water is reduced.
Sailing damselfly lives in reservoirs of Antarctica far from the sea, where
sea birds fly seldom, and frequently drifts far from the coast, therefore it
is almost not threatened with coastal birds. And dragonflies of other species
simply do not hunt such large prey.
This species of dragonflies has great problem of settling. Adult individuals
are not able neither to fly nor to creep overland, and only the larva is able
to active moving. Therefore sailing damselflies live only in large reservoirs,
connected by rivers or flooded in the spring during the snow thawing period.
In the closed isolated reservoirs this species does not live, or forms temporary
small populations.
Imago of different genders differ from each other in colouring. Female imagoes
have yellow cross strips on dark abdomen, and their body has the velvety black
color, helping them to warm up in sun light. Their wings are completely transparent.
Males have long and mobile abdomen of blue color with metal shine, and their
wings are black in the basis.
Competing for the female, males swim to each other and catch the contender by
mask for legs or for the basis of abdomen. Courting the female, male keeps her
legs with the mask and is coupled with her, having bent abdomen above the head
like a scorpion. The clutch of several hundreds small eggs is laid by the female
in of a plants floating on waters surface.
This species of insects was discovered by Arthropod, the forum member.
Butterfly
dragonfly (Papilianax multicolor)
Order: Dragonflies (Odonata)
Family: Butterfly dragonflies (Papiliopterygidae)
Habitat: Antarctica, fresh water.

Picture by Fanboyphilosopher
Till millions years Antarctica was the continent absolutely
lack of terrestrial lifeforms. Last ice age at the boundary of Holocene and
Neocene had completely destroyed all forms of terrestrial life at this continent.
When climate started to change and warm currents from the north had warmed coasts
of Antarctica, the surface of continent had exempted from ice cover. The areas
exempted from ice gradually began to be settled. But isolation of continent
had resulted to the fact that only swimming and flying animals, and plants which
seeds could be moved by wind, could settle there. Free ecological niches were
distributed among these animals by the most unexpected image.
Fresh waters of Antarctica belong to dragonflies. These insects gave surprising
variety of species – from small up to very large ones. Some dragonflies of Antarctica
surpass in size the largest dragonflies of Neocene tropics of the Earth. And
in Neocene among the Antarctic dragonflies one completely unusual species not
having of analogues anywhere in the world, Antarctic butterfly dragonfly, had
evolved.
This species of dragonflies differs from the relatives living in the neighbourhood
in many respects. The butterfly dragonfly belongs to the number of damselflies
and differs in flitting flight and very thin prolonged body. It is the largest
species of damselflies of Neocene – the length of its body from head up to the
tip of abdomen amounts 15 cm at wingspan up to 25 cm. But the size is not the
most remarkable feature of this insect. When butterfly dragonfly flies above
the swamp, it is very difficult for not noticing. This insect has motley colouring
of wings; they have soft blue color with black mesh pattern in the basis, and
on tips of wings there are oculate spots with iridescent play of colours from
lilac up to pink. The flying butterfly dragonfly sparkles in light of poor Antarctic
sun like a brilliant. The body of this dragonfly has strict black color with
golden cross stripes on each segment of abdomen. Such bright colouring appreciable
from apart permits individuals of this species to distinguish each other. These
dragonflies gather for pairing in flights numbering some tens individuals. Adult
dragonflies should hasten to find each other for pairing – at this species of
insects imago does not feed and lives only due to stocks of nutrients had been
accumulated by larva. The maximal life expectancy of adult dragonfly is no longer,
than one week.
Time of the adult life given out to dragonfly passes very roughly. Males actively
compete to each other for the right to couple to the female, and push each other
aside from it, arranging air maneuvers and actively displaying itself to the
female. As against to the male, female saves vital resources - at the moment
of male rivalry it perches on stalk of grass and waits for the ending of their
duel. When the winner is defined, he clings to the body of female and insects
make courtship flight together. Having fertilized the female, male protects
her for some time, not admitting to her other males. Next day after fertilization,
when in organism of the female eggs start development, male lets her off and
perishes soon.
For egg laying the female of butterfly dragonfly chooses coastal plants with
thick stalks rich in air-containing tissue. It immerses an abdomen in water
and makes vertical cuts on integumentary tissues of stalk by sharp ovipositor.
In each cut female places one egg, and can lay about 400 eggs in total. The
laying of eggs proceeds a very long time – about some hours. This process exhausts
the female so much, that it perishes soon after laying of last eggs, and its
body becomes prey of water predators. Frequently the female perishes from exhaustion,
having not laid last portion of eggs.
The larva of butterfly dragonfly, as opposed to peaceful adult individual, is
gluttonous and ruthless predator. It lives in thickness of water, is a good
swimmer and has wide flattened body supplied by rounded outgrowths on edges
of each segment. Branchial outgrowths on back end of abdomen are wide and have
leaf-shaped form. When larva moves in water with the help of vertical waves
of body, they work as a tail fin. If it is necessary to swim slower, the larva
of butterfly dragonfly uses walking legs covered from two sides by bristles
and acting like oars.
The larva of butterfly dragonfly eats various small animals. The young larva
eats larvae of two-winged flies, scraping them by its lower lip from substratum.
The larva of advanced age reached the length of 18 cm at width of body about
2 cm eats larvae of other species of dragonflies and attacks larger prey: freshwater
shrimps and small fishes. Frequently larvae of butterfly dragonflies attack
the adult females of their own species laying eggs, or gather their dead bodies
from surface of water. The larva of this insect develops rather slowly, and
spends under water five years before it will be ready to metamorphosis and will
turn to sparkling fragile insect.
Aquatic
soothsayer (Dimorphomantis semiaquaticus)
Order: Soothsayers (Mantodea)
Family: Soothsayers (Mantidae)
Habitat: China, cane thickets at riverbanks.

Picture by Amplion, colorization by Tony Johnes
 From
the moment of the origin at the Earth insects became one of prospering groups
of live creatures. Features of respiratory and blood system do not permit them
to grow up large size (to become larger, rather than crabs, for example), but
among tiny inhabitants of Earth insects are in the lead unconditionally. They
easily adapt to changes of environment and among them new species evolve very
quickly. At rather small morphological reorganization insects can develop new
habitats easily. One of such species had appeared in marshy plains of China.
From
the moment of the origin at the Earth insects became one of prospering groups
of live creatures. Features of respiratory and blood system do not permit them
to grow up large size (to become larger, rather than crabs, for example), but
among tiny inhabitants of Earth insects are in the lead unconditionally. They
easily adapt to changes of environment and among them new species evolve very
quickly. At rather small morphological reorganization insects can develop new
habitats easily. One of such species had appeared in marshy plains of China.
In Neocene the climate began more humid, and the plain part of China represents
a circuit of rivers, lakes and swamps constantly changing the outlines. Winds
bring rains from Pacific Ocean, and in plains flooding often happen. And after
flooding the part of water inhabitants may be trapped in shallow ponds drying
up quickly. In such conditions inhabitants of water environment develop adaptations
for life on land, and ground inhabitants study to develop water.
As a rule, soothsayers, insects having very ancient origin, were inhabitants
of woods and dry habitats. But in swamps of China one species of these insects
till the evolution process had got ability to live in water, at least, half.
Among water insects water bugs and beetles are very characteristic. These creatures
differ in predating behavior and eat any animals they can catch. For prey catching
modified front legs working as claws serve to bugs. These insects spend life
in thickness of water, regularly emerging to the surface for the portion of
air. In comparison with them aquatic soothsayer looks “amateur”, but its habit
of existence has the certain grace.
Aquatic soothsayer is completely not able to swim. Nevertheless this insect
is not less successful hunter, rather than armed and quickly swimming water
bugs. Aquatic soothsayer applies traditional tactics of the ambuscader for hunting.
This insect simply sits among water plants upwards an abdomen, having immersed
the forward part of body in water. Thus respiratory apertures of insect remain
above water, and soothsayer does not risk sinking.
Because aquatic soothsayer actually lives simultaneously in two environments,
in air and in water, it seems made of two halves of completely different insects.
This feature has determined the generic name of this insect – Dimorphomantis,
“the soothsayer of two shapes”. The length of this creature exceeds 15 cm: it
is rather large species of soothsayers though its larger congeners live in tropical
rainforests.
Forward part of body of the aquatic soothsayer is colored yellowish, very much
lengthened and thin. It makes approximately two thirds of general length of
insect. Forward pair of legs of aquatic soothsayer is well advanced and very
strong. Segments of forward legs are expanded and also resemble a pair of leaves
growing on thin stem. From outside they are colored bright green, and their
internal surface is grayish-white. Head of this insect is flat and triangular;
spherical eyes are very big. It is very mobile, and eyes provide the full circular
field of view. Aquatic soothsayer catches prey using sight and chemical feeling.
Usually it perches motionlessly at the surface of water, waiting while catch
of suitable size will swim up to it closely enough. This soothsayer catches
fry of fishes and various water insects. Sharp thorns on internal surface of
legs permit it to kill even dangerous insect like water bug quickly.
Abdomen of aquatic soothsayer, on the contrary, is wide and flat. It imitates
leaves of marsh plant sticking up from water rather good. Wings folded on back
and having pattern of light nerves on green background strengthen the accuracy
of this imitation. Walking legs at this insect have usual for soothsayer structure,
but are only more tenacious. Aquatic soothsayer frequently masks among water
plants, having perched on the bottom side of leaf. So it becomes invisible to
predators from air, and protective colouring masks insect from sights of underwater
predators. Wings of aquatic soothsayer are very short, and this insect can not
fly. But even if wings would be advanced enough, the structure of body would
not permit this insect to fly up. Therefore wings of aquatic soothsayer had
lost initial function of flight, and serve for masking. At males second pair
of wings has bright strips and spots – bases of wings have small brown spots
on yellow background, edge of wing is bright yellow. Opening wings and flapping
them, male shows to the female readiness for pairing. Also the male of aquatic
soothsayer is “musical”: calling the female, it rubs jags on forward pair of
legs against each other. The sound is well audible in water, and the ear at
soothsayers is located between forward legs. Therefore female reached the maturity
and ready to breeding hears courtship calls of the male well. It answers them
with short clicks even more stimulating “musical” abilities of the male.
Similarly to all soothsayers, aquatic soothsayer breeds once in life. During
the pairing female devours the male. The stock of sperm received by it till
the pairing is sufficient to make some clutches. Female lays among reed few
ootecas (clutches packed into common dense environment), and then quickly perishes
of the exhaustion.
Approximately after 8 – 10 days from eggs larvae very similar to adult insects,
but wingless, burst. Young growth of aquatic soothsayer is also connected to
water in life, but it occurs differently, rather than at adult individuals.
Young aquatic soothsayers are very tiny; therefore it is difficult for them
to break through the surface tension film. But it not the obstacle for them:
larvae of aquatic soothsayer settle on surface film of water and live in places
protected of wind. Aquatic soothsayers settled in other reservoirs mainly at
the larval stage, often migrating on floating plants. Adult insects lead settled
life.
Larvae have hairs on tips of walking legs with which help they keep on surface
tension film. Hairs are covered with wax-like substance that makes them hydrophobous.
This substance is emitted from glands on the tip of abdomen, and insect not
occupied with hunting serially greases legs with it. Thill first days of life
young aquatic soothsayers catch springtails (Collembola) living on water surface,
then start to gather small insects fallen in water. Water bugs become catch
of larvae of advanced age. Gradually growing up larvae start to hunt fish fry
hiding among water plants, applying the same mode, as adult insects.
At later stages of development (after the penultimate moult) young insects start
exclusively underwater hunt. At this time wax gland starts to produce much larger
amount of wax, and insect starts to grease with it an abdomen. Such feature
of behavior helps to immerse in water deeper in searches of catch, and rescues
when the insect casually falls in water – in this case aquatic soothsayer has
some minutes to get out of water.
The aquatic soothsayer is successfully protected from predatory fishes, striking
impacts by spiny forward legs. In ponds of China it has the dexterous imitator
– one local frog species.
Super-longest
archobaculum (Archobaculum prelongissimus)
Order: Walking sticks (Phasmoptera)
Family: Baculids (Baculidae)
Habitat: rainforests of Meganesia, a forest canopy.

Picture by Pavel Volkov
In the world of insects, it is hard to be a giant. Insects
have certain features of physiology which impose essential restrictions to opportunities
of size increasing. Those ones are tracheal respiring and loss of ability to
transfer oxygen by blood (hemolymph). Therefore even largest species of insects,
living in atmosphere with low contents of oxygen, can not become giants. Epoch
of Carboniferous with the high contents of oxygen in air and absence of competition
to flying vertebrates gone irrevocably. Therefore in Neocene the largest species
of insects all the same have small weight and represent the unique phenomenon
among small relatives. Walking stick species super-longest archobaculum, living
in tropical forests of Meganesia, evidently shows the restrictions connected
to body weight. This is one of the largest insects of Neocene: the length of
its body reaches 40 centimeters. But thus its thickness does not exceed 15 mm.
This giant has lost ability to fly, and its wings are strongly decreased in
size; at the female of this insect wings are completely reduced.
Archobaculum is, in fact, the huge variant of small walking sticks. Its body
has cylindrical form, and head is short and almost spherical, with short antennas.
Legs of this insect are very long and thin – they are only a little shorter
than the body. The grayish-colored body of archobaculum with knobs and dark
spots imitates tree bark texture. On head and thoracal segments knobs are numerous,
and covers of abdomen are smoother. Only on terminal segment of abdomen there
are some small outgrowths helping to male and female to fix stronger during
the copulation.
At archobaculum sexual dimorphism is well expressed. Males of this insect are
smaller and more movable, rather than females – their maximal length is no more
30 centimeters. Male also differs from the female in presence of the small outgrowths,
imitating parts of leaf almost completely eaten by caterpillars, on shins of
forward pair of legs. These sites of body are dark green with bright green longitudinal
line on shin imitating middle nerve of leaf. This adaptation serves for courtship
combat – males push each other away from the female by front pair of legs. Demonstrating
itself to the female, male displays to it courtship ritual: it spreads legs
of front pair and opens bright reduced wings of back pair – they are yellow
with black border and several spots at the root. These wings can not lift this
insect in air, and are used only for courtship display. In rest they are covered,
as if by covers, by narrow front wings.
This species is almost defenseless to predators. A plenty of young ones perishes
from small insectivorous birds, larger individuals are eaten by reptiles and
mammals. The basic protection of super-longest archobaculum is the protective
similarity to branch of tree. Having felt danger, insect clings to branch and
extends forward front pair of legs pressed together. Thus the walking stick
seems the continuation of branch. The disturbed insect emits unpleasant smell,
but can not give even any repulse to predator at all.
Males of super-longest archobaculum meet less often, than females, but such
disproportion does not interfere with breeding of insects. At absence of males
females simply lay parthenogenetic eggs from which normal viable young ones
hatch – but only females do. On some islands this species of insects is presented
only by females.
The success of this species in settling is caused by two important features
of these insects. First, super-longest archobaculum is herbivorous species consuming
wide spectrum of vegetative food. At other species of walking sticks the number
of fodder plants species may be strongly limited, and it interferes with their
settling.
The second component of success of this insect is the feature of eggs structure.
The majority of walking sticks lays eggs with dense dry shell, which fall on
the ground and can rest during very long time. The female of super-longest archobaculum
lays eggs on the bottom side of leaves with the help of mobile tip of abdomen.
The egg of this insect is placed on long threadlike stem and has shell sticky
because of slime not drying up for a long time. It endures gusts, but is easily
pasted to feathers of birds and wool of mammals. Vertebrate animals have considerably
larger abilities to settling, than these insects, therefore due to their “services”
super-longest archobaculum is very widely settled in rainforests of Meganesia.
Besides if the egg will fall on the ground from forest canopy, it will be very
difficult for larva bursting from it to rise back to forest canopy to find fodder
plants. Eggs at the female of super-longest archobaculum are produced, as if
on the conveyor. It can lay no more than 15 – 20 eggs per day, but keeps such
daily fertility during the whole adult life (after last moult).
Larvae of super-longest archobaculum differ from adult individuals only in smaller
size and smoother body. Outgrowths and knobs characteristic for adult insect
appear at them only after penultimate moult. Development of the larva proceeds
till about 15 months, and life expectancy of adult insect seldom exceeds half-year.
Saw-legged
dragon grasshopper (Serratipodus dracocephalus)
Order: Orthopters (Orthoptera)
Family: “Awful grasshoppers” (Horrodecticidae)
Habitat: woods at the islands of Pacific Ocean.

Picture by Amplion, colorization by Biolog
Initial picture by Amplion |
Islands are fairly named as “hearth of evolution”. Here there
is a fastest rate of new species formation, and here life gets the most fantastical
forms.
As a rule, at the islands never been a part of continent, the fauna consists
of species able to flight, or lost this ability already after island settling.
At islands there are no representatives of many groups of animals and plants,
characteristic for continents. And at islands there are many free ecological
niches which sometimes are occupied with species completely unexpected in this
role. At islands of Pacific Ocean the place of small predators was occupied
with the huge representative of insects - the distant relative of common grasshoppers,
the saw-legged dragon grasshopper.
On contrary to popular belief, the significant part of grasshoppers is predators
(and herbivorous locusts belong to other family). The saw-legged dragon grasshopper
is the huge representative of grasshoppers: the length of its body reaches 20
cm, and because of long antennae and wings he seems even larger. Among the impoverished
fauna of islands it occupies an ecological niche of small predators, attacking
large insects and small vertebrates - birds and rodents. This species also eats
carrion: after storm or during outflow saw-legged dragon grasshoppers wander
along ocean coast, eating dead sea animals or gathering inhabitants of the ocean
has got in pools at the coast.
Adaptation to predating is visible in appearance of insect: forward legs of
saw-legged grasshopper are very long (they are equal to back legs by length),
on their internal side there are sharp thorns: this is the adaptation for catch
seizing. Powerful mandibles of insect easily crush both armour of beetle, and
bones of small vertebrates. Frequently saw-legged dragon grasshopper swarms
up trees searching for bird nests: it equally willingly eats eggs and nestlings
of small species of birds.
Back legs of this insect as at the majority of representatives of order, are
rather long, but because of large weight this insect is not able to hop. But
this huge grasshopper swarms up trees well: on its paws tenacious claws, permitting
to heavy insect to cling to slightest jogs of bark, are advanced.
On large head of saw-legged dragon grasshopper there is a plenty of knobs and
outgrowths especially advanced at males. They give to this insects fantastical
appearance, and serve for tournament fights in breeding season. antennae of
saw-legged dragon grasshoppers are very long too: they are equal to body by
length. On thorax at males two bent horns are advanced sticking up upwards and
in sides.
Colouring of this insect quietly sitting on tree seems modest: body is gray-brown
with small black speckles. The brightest part of body of saw-legged dragon grasshopper,
the back pair of wings, at this moment is latent. But during courtship duels
or at caring for the female males open them, showing bright pattern: black “grid”
of nerves on yellow background; near the basement of wing there is big red spot.
Top wings of insect are reddish-brown with grey cross strips, on edge nerves
the sound device is advanced: a line of tiny denticles. During chirring the
insect rubs one wing against another, uttering sounds heard at the distance
of one hundred meters.
Despite of size, this insect is able to fly, and even can make distant flights
between islands, especially at good fair wind. Such way saw-legged dragon grasshoppers
had settled practically at all in slightest degree suitable for life islands
in tropical zone of Pacific Ocean. The unique limiting factor for them is the
presence of fresh water. But at small islands these insects find enough moisture
in puddles accumulating after rain in bases of palm leaf petioles.
These large insects can breed all the year round. The female differs from the
male with fuller abdomen, long saber-like ovipositor and pale colouring of wings.
Males find females ready to breeding by smell. During acquaintance male creeps
up to the female from the side of head, widely opening and planting in sides
bright wings. Thus it moves by antennae, touching by them to antennae of the
female. When the female reciprocates, male closes wings and starts to chirr
loudly, rubbing one wing against another. Thus it declares the rights on the
female. If near one female some males gather, they begin original “tournament”
in which the strongest one is displaying. Males up to the last moment avoid
to enter direct collision which can be finished by death of one of them, and
superiority is defined by loudness of chirring and brightness of coloring of
wings. Males start to chirr, to short time interrupting warbles with demonstration
of unwrapped wings. Weaker males at once leave a place of competitions, and
gradually near the female only one or two contenders stay. If it is impossible
to define the strongest one in the peace way, and not any male wish to concede,
between them fight is possible. Strong forward legs are in use, with which contenders
grip each other by bases of antennae and try to push aside from the female.
Fight is accompanied with menacing chirring and demonstration of opened wings.
Pairing lasts not for long, then the male loses interest to the female and crawls
out. With the help of long ovipositor female places portion of two - three tens
large eggs in friable, well warmed up ground, and does not care any more about
them. For all life she can make up to ten clutches.
All orthopters are insects with incomplete metamorphosis: from eggs larvae similar
to an adult insect excluse. Young saw-legged dragon grasshoppers about 5 millimeters
long eat different delicate insects - plant lice and tiny caterpillars. At half-year
age they grow up to the size of adult insect, and become able to breeding. At
the lack of forage they will turn to “scanty” ones with thin long body and wide
wings. Such individuals fly perfectly, and easily move from island to another
one searching of favorable conditions for life.
Hawaiian
red-masked sawleg (Horrodecticus larva-rubra)
Order: Orthopters (Orthoptera)
Family: Awful grasshoppers (Horrodecticidae)
Habitat: Hawaii Archipelago, rainforest canopy.

Picture by Fanboyphilosopher
In human epoch in New Zealand huge endemic orthopters lived
– slow-moving herbivorous wetas (family Deinacridae). These creatures were typical
inhabitants of the ecosystem lack of terrestrial predators of small and medium
size, and appeared low resistant to competitors in a human epoch when in New
Zealand due to human activity terrestrial mammals have appeared. In Neocene
epoch wetas had died out at the large islands, and only separate relic species
had been kept at the separate small islets where rats and predators of mustelid
family had not got.
In Neocene at the tropical islands of Pacific Ocean evolution of insects had
created new group of orthopters – sawlegs, large predating insects. They descend
from grasshoppers, and, possible, Indonesia was the centre of their origin.
Because sawleg ancestors evolved surrounded by possible predators and competitors,
they had not lost ability to self-defense. At predator’s attack sawlegs protect
themselves actively by bites, seize an aggressor with strong forward legs armed
with sharp spikes. Besides as against wetas, sawlegs are good flyers, and one
of species, saw-legged dragon grasshopper
(Serratipodus dracocephalus), had settled to many islands of Pacific Ocean.
Hawaiian Islands became one of the centres of specific variety of sawlegs, where
some species of these predatory insects live. One of typical Hawaiian species
is Hawaiian red-masked sawleg.
It is rather large species of orthopters kept ability to fly. It had easily
settled to all islands of archipelago and lives in crones of trees. Body length
of this sawleg is about 18 centimeters. Body and front pair of wings have camouflage
colouring – brown with dark faltering lines imitating the facture of wood bark,
and separate irregular-shaped light spots. In different populations these spots
may differ in color from light grey up to yellow and greenish. The second pair
of wings is colored very brightly – on transparent background bright blue spots
with iridescent shine and black border are scattered. Head of this insect has
no outgrowths and lumps, except for low cross crest between the bases of antennae;
it has shining black color, but front part of head and forehead are bright red.
It is warning colouring: this insect is well protected from small predators.
Hawaiian red-masked sawleg has very large mandibles with jagged internal edges.
The external edge of mandibles is colored white, contrasting with colouring
of head.
The enlarged front pair of legs is a general feature of sawleg family. At this
species legs of forward pair differ in expanded segments and are similar a little
to legs of soothsayers. The external surface of legs has camouflage color, and
internal side is snow-white and smooth. Threatening an aggressor or the congener,
males of sawleg spread in sides front pair of legs, displaying well appreciable
colouring of their internal surface. The internal edges of segments adjoining
with each other, have saw-like lines of sharp prickles, helping to keep prey.
This species is a predator; it hunts large insects, snails, cubs of rodents
and nestlings of small birds nesting on trees. Due to strong mandibles this
insect easily cracks shells of ground snails and bones of small vertebrates.
Hawaiian red-masked sawleg is well protected from attack of predators. In case
of danger it emits acrid liquid from abdomen – it is stored in special outgrowth
of hindgut. At threat this insect also displays to aggressor red “mask” and
widely opens mandibles. Displaying the threatening pose, sawleg rises on legs
and rocks, seizing for branch. If the enemy does not recede, insect opens back
wings, showing spots, and raises an abdomen. After this pose “shot” of jet of
caustic liquid into the aggressor follows.
The female of red-masked sawleg is heavy-built and moves slowly, but the male
is good flyer. Males of Hawaiian red-masked sawleg frequently fly from island
to the next one, carrying their genes into new populations, therefore local
forms of these insects have a set of transitive variants and are indistinctly
separated from each other.
The stridulation organ at these insects is located at the edges of front pair
of wings leaning against each other. Courting the female, male sings special
“love song”, producing short series of melodious chirring. At the different
species living in common, sounding of songs strongly differs.
If near the same female two males meet, they arrange the duel accompanying with
loud lingering chirring. As these insects live in the area of tropical climate,
seasonal prevalence in breeding is not expressed. The female lays up to 200
large eggs in hole, making it in wood ground with the help of ovipositor. Egg
laying repeats every five – six days during eight months (the deadline of imago
life is those). Young insects develop within almost two years, and during almost
whole life they keep camouflage colouring. They lead terrestrial way of life
and can jump – it helps them to escape from ground crabs.
At Hawaii close species of sawlegs live:
Hawaiian
mimic sawleg (Horrodecticus trixter) evolved side-by-side with Hawaiian
red-masked sawleg and shows an example of Mullerian mimicry. It also has red
“mask” and is able to protect itself against enemies. It can not splash out
acrid liquid, and only belches from mouth unpleasantly smelling sticky substance.
But this insect aggressively defends itself from the enemy and puts to it painful
wounds by thorns of front legs.
Reef
sawleg (Horrodecticus thalassophilus) lives in mangrove thickets and
has intensive green color with brownish nerves on wings. Forward part of body
of this insect is yellowish. The length of body of this insect reaches 18 cm.
It has rather thin and long front legs; that’s why the insect seems larger.
During the outflow reef sawleg hunts small sea animals in littoral pools. This
insect immerses forward part of body in water and catches prey by front legs.
This insect usually eats young fishes, small crabs and molluscs. If there are
no mangrove thickets at coast, this insect lives in forests and hunts at the
reef during the outflow.
Flat-legged sawleg
(Platydecticus platypodus) is a terrestrial species
of sawlegs. It has camouflage colouring, and only internal surfaces of front
legs have white colouring. Its body is covered with tiny tubercles, and this
insect is successfully masking in fallen foliage. This sawleg has muscled front
legs. It eats carrion and and hunts ground crabs. Frequently it digs out holes
of crabs and pulls out from them the moulted animals still having soft carapace.
New
Zealand Green Sawleg (Notohorrodecticus rex)
Order: Orthopters (Orthoptera)
Family: “Awful grasshoppers” (Horrodecticidae)
Habitat: Woods and forests, dense scrub, North and South Islands of New Zealand.
The introduction of rodents and other mammals to New Zealand proved doom for
the native Orthoptera, the Wetas (Anostostomatidae). These insects were generally
slow, ponderous and lacking in aggression.
Orthoptera living in other countries were both able to be large, and aggressive
enough in defence of themselves to exist alongside mammalian predators, even
during the Holocene. And thus, the predatory saw-leg grasshoppers evolved in
the Neocene from such forms. Some of these inhabit New Zealand, and have come
to form nostalgia of the long lost Wetas.
Most prominent form is the New Zealand Green Sawleg, which reaches up to 15
centimetres long, because of wings and antenna this length seems longer. Preferring
areas of dense cover, it is chequered and striped with light green, dark green,
and yellow, as with some kinds of predatory Holocene katydid. As with all sawlegs,
it is a predator, consuming insects, small rodents, bird eggs and nestlings,
lizards and frogs, as well as occasionally snails.
The front legs are large and powerful, armed with trenchant spines for catching
prey. This grasshopper has large hooked mandibles which can easily kill and
butcher large insects and small vertebrates.
Back legs are long and spiky but because of size, this species does not hop,
it may kick fiercely with its legs to repel predators. Living in forests and
woods, it is an exceptional climber, but may also forage on the ground.
Unlike some of its relatives, the
head is un-ornamented. Wings have bright orange and black eyespots which they
display pre-emptively if they are aroused by a predator or intruder. As with
all sawlegs, this insect stridulates by rubbing its wings together to make a
loud chirring call which is quite deep. This insect is able to fly fairly well
but usually only does so in order to find new territory or flee.
Breeding occurs from September to March, when conditions are warm. The female
is plumper in the abdomen and has a large ovipositor. Males locate females by
smell, and will display to them by opening their wings and chirring loudly.
If more than one male approaches a female, they will call and display loudly
to each other in a group, and if forced to fight will lock mandibles and attempt
to throw each other off their perch. Once the female shows willingness, the
male will call loudly in an overture to the brief coupling. The female lays
with her ovipositor two or three masses of about 30 eggs each which she places
in the ground at the base of a bush or tree, females may reproduce up to 10
times in their life. Young wingless nymphs about 4 millimetres long, hatch from
the eggs, these babies resemble their parents in miniature, but are spotted
with bright yellow, and feed on small insects. Adult size is achieved at about
6 months.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Insular
Boar Weta (Suinacris insularis)
Order: Crickets and Grasshoppers (Orthoptera)
Family: Wetas (Anostostomatidae)
Habitat: Small series of remote islets to the north-east of mainland New Zealand,
scrub and sparse woodland.
Introduced species of mammal had a severe effect on many kinds of native animal
in New Zealand during the Holocene. Along with the extinction of native birds
and reptiles, large native insects, Wetas, suffered badly too.
In the Neocene, descendants of Wetas (Anostostomatidae) only remain on off shore
islets far enough away from the mainland to not have been colonized by invasive
mammals. Some of these small islands are relatively newly formed, such as those
to the north-east of North Island. Here, we do not see giant weta of past human
age, but descendants of more common Tree Weta (Hemideina thoracica), which had
become vagrants after washing out to sea on rafts of vegetation.
The Insular Boar Weta is actually a complex of similar sub-species spread across
this group of small islands. It is quite large for an insect, reaching about
7 centimeters in length, excluding the legs. Males have a much larger head proportionately,
bearing greatly enlarged mandibles which form prominent protruding “tusks”,
used in mating contests. Cuticle covering is heavy and rough, hindmost legs
are large, powerful and spiny, it is able to kick forward defensively if attacked
by a bird or lizard. Colour is an overall light brown, with dark markings on
the abdomen, and a darker head and jaws. Diet consists of the leaves, buds and
stems of trees and shrubs, as well as some invertebrates. This insect is most
active at night. Boar wetas are able to stridulate with their legs to call,
and during the mating season, males can drum their abdomen against the ground
percussively to attract females.
They will nest in tree hollows or abandoned holes dug by birds, in the breeding
season, the male will keep a harem of up to 8 females, which he defends fiercely.
Males compete over harems by locking mandibles and attempting to flip each other
over, but females may secretly mate with multiple interloping males. Females
later disperse and lay their eggs in soft soil. The eggs hatch in the spring,
and 8 instars are required before reaching adulthood. Lifespan of this insect
is 1 and a half years.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Hunchbacked
Cave Weta (Ankylodrosoptera nanus)
Order: Orthopters (Orthoptera)
Family: Camelback Crickets (Rhaphidophoridae)
Habitat: New Zealand, the mouths and upper reaches of caves.
The native Orthoptera of New Zealand suffered heavily from the introduction
of mammalian predators, mainly rodents. The large Wetas went extinct entirely,
and the Cave Wetas were obliged to decrease in size and become more alert and
furtive. Example of these is the Hunchbacked Cave Weta.
This insect generally resembles its ancestor, it is a wingless, cricket-like
animal with a globular body, and long spidery legs, antennae are very long,
insect seems larger because of long extremities. Body of this insect is only
about 2 centimetres long, but span of legs may reach as much as 12 centimetres,
antennae are even longer. As with many species in its family, its body is rounded
and the arch of the back forms a hump-like shape (relatives of cave weta were
called “Camel Crickets” for this reason). Body colour is a pale brown, the posterior
part of each segment is darker, and legs are an overall dark colour. This animal
only inhabits the upper reaches of caves, and regularly ventures into the open
at night, hence sight is still well developed and eyes are large. Individuals
navigate the darkness of the caves with ease, and may climb some distance on
cave walls; they also sometimes forage in or near shallow pools of water. These
insects are generally scavengers, eating dead animal and plant matter, as well
as soft plants and fungi. Individuals remain on the walls and floor of the cave
by day, emerging at night to forage and feed; they tend to prefer damp conditions.
Breeding season is in winter when it is moist, they form large aggregations
at the entrances of caves, forming pairs which mate multiple times during the
course of the day. Females venture away from the congregations to lay their
eggs by depositing them in soft clay or soil close to the entrance of the cave.
Eggs take about 3 months to hatch, lifespan can be as much as two years. Nymph
stage lasts about 10 months, females do not die after egg-laying but are usually
not lucky enough to lay twice. These insects are most abundant in late autumn
and early winter, when it is damp and relatively cool.
This species of invertebrates was discovered by Timothy Donald Morris, Adelaide, Australia.
Scorpio
earwig (Scorpiforficula cervicera)
Order: Dermapters (Dermaptera)
Family: Earwigs (Forficulidae)
Habitat: Balkan, southern and western coast of Fourseas.

Picture by Alexey Tatarinov
Forests of temperate latitudes differ from tropical rainforests
in presence of thick layer of soil. In tropical forest soil layer is very thin,
and under it there is a layer of infertile mineral substratum. In forests of
temperate latitudes and subtropics ground layer is considerably thicker. Ground
of these forests is rich in various live beings which process dying off organic
substance to the condition easier absorbing by plants.
Among ground animals of temperate latitudes earwigs (order Dermaptera), the
insects changed externally a little from Paleozoic era, are characteristic.
Separate species of these insects had successfully survived till the ice age
at the boundary of Holocene and Neocene, and had given rise to several interesting
species of Neocene epoch. In subtropical forests of Balkan and coastal areas
of Fourseas there is one very large species of this group, the scorpio earwig.
The body length of scorpio earwig reaches 5 – 6 cm; it is the largest representative
of order in Neocene. This is heavy-built wingless insect with strong long legs
which is able to run quickly – escaping from the enemy, scorpio earwig accelerates
momentum up to 20 cm per one second.
Scorpio earwig looks very impressively – its covers have black color with faint
metal shine. This insect is not able to fly, and its wings had undergone the
reduction – the back pair of wings had vanished completely, and two small outgrowths
on thorax remain from forward wings. Cercuses at this insect are large, reddish-brown,
differing in shape at insects of different genders. At males they have the additional
outgrowth directed inside – hence the specific epithet meaning “deer-horned”.
At females cercuses are wider and lack an additional outgrowth. The difference
in cercuses shape is connected to their different role in life of insects of
different genders. Males use cercuses for courtship combat, and female hatches
posterity on them.
In human epoch at earwigs various forms of care of posterity were recorded.
Scorpio earwig is not exception here. It differs in strongly advanced parental
instinct and original care of posterity. The laying of this species totals about
fifty large eggs. It is covered with the common foamy environment which dries
up and forms a semblance of porous cocoon. Female constantly carries clutch
in cercuses, having bent them above head (at this moment it is similar to scorpion,
hence the name).
Common earwig (Forficula auricularia) which, most likely, was an ancestor of
this species, perished right after bursting of young ones and the posterity
ate remains of their own mother. The female of scorpio earwig does not perish
after the bursting of posterity, because before it had led almost habitual way
of life. It continues to carry the hatched young growth on back, and additionally
feeds young ones with belched semidigested food. For this reason the survival
rate of posterity of early age at this species is very high. During the feeding
of young growth female raises head upwards, and belches a part of food. Young
insects (similar externally to adult individual, but translucent) creep right
on its head and eat belched food.
Scorpio earwig is a predator. Its prey includes slugs and sot-bodied insects
(caterpillars and grubs). Also this insect eats carrion.
Rabbit
earwig (Graciliforficula cuninculiphila)
Order: Dermapters (Dermaptera)
Family: Earwigs (Forficulidae)
Habitat: New Zealand, colonies of castle rabbit,
caves.

Picture by Alexey Tatarinov
Species of animals, widely settled at the Earth due to human
activity, occupied ecological niches in new ecosystems. Frequently it proceeded
to the detriment of local flora and fauna, but occasionally such process passed
not so sharply. After extinction of mankind species of animals and plants introduced
to habitats unusual for them began to evolve and to survive in equal conditions
with native species. Species got advantage because of ability to live in anthropogenous
landscape died out, as a rule. Other species adapted to life in natural environment
and became a part of new ecosystems.
In New Zealand common earwig (Forficula auricularia) was one of numerous introduced
species. Its descendants had adapted to life in forests of islands and differ
from the ancestor a little, but one species had chosen absolutely special inhabitancy.
For fauna of Neocene mammals of New Zealand castle rabbit is characteristic.
It is a social species of herbivorous mammals building strong and long-term
constructions in which all colony lives. Many species of invertebrates being
symbiotes, commensals and parasites of rabbits live in system of tunnels and
chambers of these animals. The descendant of common earwig living in nest litter
is completely blind rabbit earwig.
This insect is perfectly adapted to life in warm and humid holes of rabbit,
but hardly can survive at the ground surface. Its covers are thin and soft,
as if at termite, colored yellowish-white, even translucent on the bottom side
of abdomen. The length of insect does not exceed 25 millimeters. A constitution
of this earwig is very fragile and delicate – it has long flexible body with
short legs, and thin cercuses bent on tips. Wings at rabbit earwig are reduced
– it never leaves rabbit holes and spends the whole life in litter of their
nests.
Head and cercuses are the firmest parts of body of this insect. Despite of delicate
constitution, rabbit earwig is an active predator. Its prey includes various
insects living temporarily or constantly in dwellings of rabbits. More often
flea larvae living in litter of nest fall prey of this insect, but occasionally
rabbit earwig eats larvae of crickets and cockroaches.
For breeding this insect chooses burrows not visited by rabbits for a long time.
In friable walls of holes it digs a short tunnel ending by nest chamber. In
chamber the female of this earwig lays up to 50 eggs and protects them right
to bursting of young ones, having turned around of them. Frequently females
of rabbit earwig form small colonies, arranging holes near each other. Larvae
and adult individuals at overpopulation of colony move in new habitats. For
this purpose they use “services” of castle rabbits – insects simply creep in
their wool.
This species of insects can live not only in colonies of castle rabbits. Rabbit
earwig can form small populations in caves populated by bats. In caves they
hunt cockroaches and larvae of flies settling in dung of bats.
Spiderweb
scorpion fly (Xenopanorpa arachnophila)
Order: Scorpion flies (Mecoptera)
Family: Scorpion flies (Panorpidae)
Habitat: warm-moderate areas of Europe.
In Holocene epoch scorpion flies (Panorpa) were among the most widespread insects
in temperate latitudes of Eurasia. These insects, eating dead small animals,
had one interesting feature: having got in spider web, scorpion fly easily
escapes from it, washing sticky strings from legs by saliva. In Neocene among
scorpion flies one species appeared, at which this feature has received unexpected
development – it is spiderweb scorpion fly. This species of insects lives in
places where it simply dangerous for life to appear for other insects: near
spider webs of various species of spiders, where always there is an opportunity
to find the insects got in trap placed by spider. Actually, this scorpion fly
plunders spiders, sucking out the caught insects. Thus, it displays the phenomenon
of cleptoparasitism among arthropods.
Spiderweb scorpion fly has very gracile constitution – it has long body with
mobile abdomen, which in rest is inflexed by the way characteristic for these
insects. Thin long legs help spiderweb scorpion fly to move quickly. It is
able to fly good, but during the feeding on a web moves basically run. Only
at approach of spider and occurrence of real danger to life this insect flies
up. Body of spiderweb scorpion fly is light grey, head is black, and the tip
of abdomen is reddish-brown with yellowish border. Males have this border a
little bit wider, than females. Also самец has spender and more graceful addition.
Length of body of this insect is about 2 cm.
Head of this insect is extended to long proboscis slightly bent from top to
bottom. Due to mobile joint of head with thorax this insect can turn head on
a corner up to 180 degrees.
On paws of spiderweb scorpion fly glands are advanced, secreting rich gel-like
substance. Spiderweb scorpion fly additionally greases paws with saliva, which
provides even more free movement on spiderweb. Due to such adaptation the insect
does not stick to web and can move freely on it in searches of prey.
Strong narrow wings allow spiderweb scorpion fly to fly quickly though this
insect basically runs.
Spiderweb scorpion flies keep near web of large species of spiders. In rest
the insect keeps near edge of spiderweb, having put tips of antennae to web
strings. When in web the insect is caught, scorpion fly feels its movements.
If the spider moves to prey faster, scorpion fly waits, while it will braid
prey by web and will inject digestive enzymes in it. After that the insect
cautiously creeps to the prey prepared by spider and sucks it out with the
help of a proboscis.
Pairing at spiderweb scorpion flies takes place outside of spiderweb, but near
to habitat of the female. Males are less adhered to their inhabitancy, and
frequently feed on web near which the female lives. Within one summer the female
lays up to 200 eggs in small portions.
The larva of spiderweb scorpion fly lives in wood litter. It is similar to
caterpillars of butterflies, but differs from them in high “crest” of gland-bearing
hairs. Bulb-like tips of hairs emit unpleasantly smelling liquid with which
help the larva protects itself against ants. The same liquid serves as original
“glue”: with its help the larva fixes on the body pieces of rotten leaves and
bark, providing itself additional masking. It eats small digging invertebrates.
Development of the larva proceeds within about one year; it winters, having
dug in wood litter.
Vinegar
ground beetle (Deinocarabus acetiferus)
Order: Beetles (Coleoptera)
Family: Ground beetles (Carabidae)
Habitat: Southwest Europe, North Africa, Atlas Mountains.
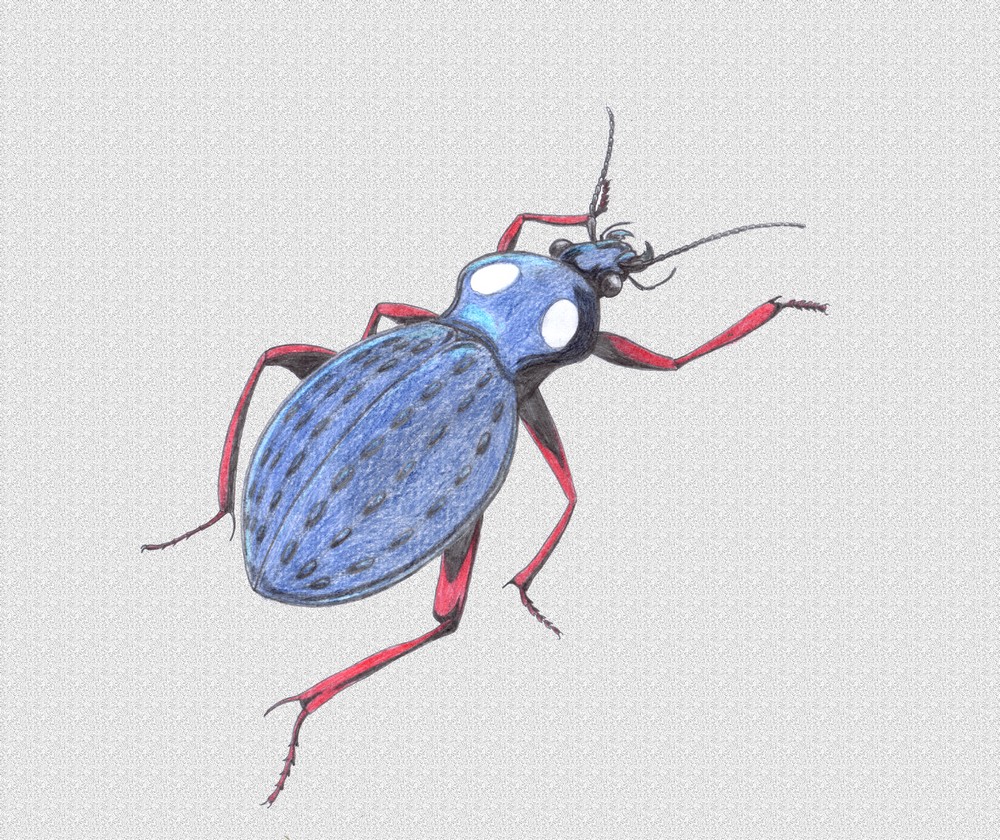
Picture by Amplion
Insects are one of the most successful groups of terrestrial
inhabitants. Features of anatomy of respiratory system do not permit them to
turn to large animals, but in small-sized class they are out of competition
among other groups of terrestrial animals by variety and abilities to adaptation.
Huge number of populations, wide spectrum of variability and fast alternation
of generations successfully allow them to adapt to various changes of environment.
Among insects there are species having interesting adaptations for survival.
In Neocene strait of Gibraltar was closed for ever because of movement of Africa
to the north, and Mediterranean Sea had turned to circuit of salt lakes and
swamps with few islets of land. The western Europe and North Africa in Neocene
became unite land where rather dry climate dominates. On grasslands overgrown
with dry light forest, there is penetrating heat in summer, and the majority
of animals hide in shadow places, or at all pass to underground way of life.
However heat is not dangerous to insects, and they become even more active when
sun is almost in zenith.
Large predatory beetle, the vinegar ground beetle lives in grasslands of Western
Europe and North Africa. The length of this insect reaches 7 – 8 cm: it is the
largest representative of ground beetles living beyond the area of tropical
forests. Vinegar ground beetle is not able to fly (it is the common feature
of all ground beetles), but runs very quickly. Elytrums form strong armour on
its body, protecting insect from birds. However, the main protective adaptation
of this beetle is not armour. Colouring of insect is dark blue with metal shine.
Elytrums are covered with set of knobs forming longitudal lines. Prothorax of
vinegar ground beetle is smooth, with white oculate spots. Long legs of insect
strongly contrast with body colouring – they are bright red, covered with shining
shell. Such colouring is warning – vinegar ground beetle easily defends from
small predators belong to vertebrates.
Sight of vinegar ground beetle is good; eyes are large and spherical. Strong
mandibles of the beetle with sharp cutting edges can bite through even skin
of vertebrate animals, therefore vinegar ground beetle has very few enemies
among animals of size comparable to it. But its main protective adaptation is
the chemical weapon, able to stop an attack even of large predator. Ground beetles
of Holocene epoch differed in ability to emit caustic chemical substances with
unpleasant smell. Hit of liquid of large species on skin could cause painful
chemical burns in aggressor. Vinegar ground beetle has even more succeeded in
chemical war leading: it has transformed the poison inherited from ancestors
to offensive weapon. This insect can be dangerous to rodents and other inhabitants
of holes – the beetle emits poisonous easily evaporating substance having suffocating
action. This way vinegar ground beetle quickly kills small animals in closed
space of holes, where the ventilation is insufficient. Especially frequently
cubs of rodents and others burrowing animals become prey of vinegar ground beetle.
Secretions of the beetle strongly smell as vinegar – hence its name.
On the ground surface vinegar ground beetle also attacks small vertebrates –
frogs and small lizards. As the chemical weapon on ground surface influences
less effectively, vinegar ground beetle chases them. Also this beetle eats large
insects and spiders. Having killed prey large in comparison with itself, beetle
stays near to it while it starts to spoil. Thus the insect furiously protects
catch from congeners. If near dead animal two beetles of this species meet,
between them severe fight is fastened.
Holes of mammals are not only hunting territory, but also an incubator for eggs
of vinegar ground beetle. This insect lays eggs one by one and leaves them slightly
dug in top layer of ground inside the tunnels dug by rodents. Fertility of the
female makes up to 300 eggs for 6 – 7 months (imago of this beetle lives so
much time). In conditions of constant humidity and good aeration eggs of vinegar
ground beetle incubating successfully, and after 9 – 12 days from them small
worm-like larvae burst. They quickly dig in ground, and till first some weeks
of life eat ground invertebrates. Development of larvae of vinegar ground beetle
is delayed for two years. The larva of advanced age shortly before metamorphosis
is frightening creature in shining armour of black color, up to 15 cm long,
with advanced legs. It lives in friable ground where digs a system of its own
tunnels and hunts digging insects and their larvae. In droughty districts larvae
of vinegar ground beetles meet mainly under stones, where ground is rather damp,
or in deep layers of ground, in holes of mammals. Larvae of vinegar ground beetle
are creatures on which well-being the set of animals in extensive territory
in Western Europe and North Africa depends. These creatures eat clutches of
locusts dug in the ground, and thus adjust number of these gluttonous orthopters.
Deceiver-beetle
(Deceptoroptera aromatica)
Order: Beetles (Coleoptera)
Family: Ground Beetles (Carabidae)
Habitat: Forested areas of New Zealand, nests of Neovespula wasps.

Picture by Biolog
The colonies of Neovespula
wasps have invited many parasites and commensals, one such creature is the Deceiver
Beetle. Beetles are notorious for being highly adaptable to new niches, and
so we see here.
This beetle reaches about 1.7 centimeters long, and feeds by predating on individual
wasps within the nest. Its appearance partly mimics its host, elytra are striped
in black and yellow, but otherwise it resembles the genus Carabus quite closely,
apart from a squarer head. It also produces a pheromone scent that mimics that
of a young wasp. This is connected to its method of parasitism; it feeds to
the wasps a sweet-smelling narcotic substance from its mouth. The substance
causes the wasps to become slavish protectors, guarding this beetle as if it
were their offspring. Only leaving the nest in order to find a mate, it is mostly
nocturnal, and solitary outside of the breeding season.
Breeding occurs in late winter and early spring, from August to October, this
is the only time adult beetles leave the nest. Males will seek the females by
scent, and actively compete for her attention. The female may mate with multiple
males during one breeding season, up to 30 eggs are laid in a burrow. The female
will attend to the eggs until they hatch, larvae are independent and feed by
predating on smaller insects. After pupation, both males and females instinctually
seek out Neovespula wasp nests in which to spend the rest of their lives. Lifespan
is up to three years.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Pale
Diving Beetle (Baptoptera cavernicola)
Order: Beetles (Coleoptera)
Family: Predatory Diving Beetles (Dytiscidae)
Habitat: Cave waterways and some surface waters downstream. North and South
Islands of New Zealand.
Cave faunas are dependant on the inhabitants of the surface, from which endemic
specialised species arise after venturing there. And so it was that the typical
Cosmopolitan Diving Beetle (Rhantus suturalis) gave rise a form living within
cave waterways in New Zealand.
This beetle is outwardly a little similar to its ancestor, a streamlined aquatic
beetle up to 1.3 centimetres long. It differs in that the antennae and forelegs
are longer and coated in long bristles, prey is usually located by touch and
olfaction. Eyes are reduced in the size and number of lenses, but eyesight is
still retained. Individuals may live in surface waters, a small distance downstream
from caves. Body coloration is a pale yellowish brown, extremities and edges
of exoskeleton are slightly darker. Swimming legs are large and covered with
backward pointing bristles, apart from bristly forelegs and antennae the front
of the animal is streamlined and rounded, a bullet shape. Larvae are slightly
longer than the adult, and are voracious predators in their own right. Head
of larvae are large and flat, bearing curved pointed mandibles which seize prey,
as with the adult the front legs are covered in long sensory hairs. Body is
elongate and tapered, coloured similarly to the adult, a breathing siphon protrudes
from the back end. Adults trap air bubbles beneath their wing cases to breathe,
larvae must protrude a snorkel-like siphon above the surface to breathe.
This species hunts by swimming and diving about in the water column, in search
of smaller aquatic invertebrates, usually little crustaceans, larvae and even
smaller individuals of its own species. Olfaction and tactile senses are used
to locate prey, beetle is sensitive to scents in the water and the movement
of water around it. Prey is seized by the curved claws on the forelegs, and
passed to the mouth-parts.
Breeding occurs between October and late January, males locate receptive females
by scent, fertilisation is internal. Up to 30 eggs are deposited adhering to
stones or hard surfaces underwater, larvae hatch within 4 days. Larval stage
involves three instars, and towards the end of the final stage, it looks for
exposed mud away from the water in which to pupate. A burrow is dug in the mud
where the larva pupates, and emerges after two weeks. Usually one generation
is produced per year. Predators include other aquatic insects such as fishfly
larvae, and even their own predatory larvae, cave dwelling frogs,
tadpoles and false-salamanders
commonly prey on this insect too. Lifespan may be up to 7 years.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Narrow-sided
fur beetle (Pellibius compressipleurus)
Order: Beetles (Coleoptera)
Family: Leather-winged beetles (Cantaridae)
Habitat: South America, moderate and cool areas, commensal at large animals.
Large species of animals are an extensive habitat for various parasites. On
each species the unique community of parasitic animal’s species (parasitocenosis)
is formed – some of their species are strict special for the present species
of large animals. Abundance of parasites involves various predators. Some of
them only incidentally meet on large parasitocenosis host animals, and others
spend on them at least all life. Usually these predators are insects though
among parasite hunters there are even bats
living on body of obda – the huge
herbivorous animal of Eurasia. On groundsloth
rodents living in Neocene epoch in Patagonia, the separate species of tiny
marsupial mammals lives. But the original beetle – narrow-sided fur beetle
– shares with this mammal places of inhabiting. For quite explained reasons
the doctor caenolestes does not settle in feathering of large birds, but this
beetle can live even there though it obviously prefers for life mammal wool.
Narrow-sided fur beetle had not changed predating habits of representatives
of its family, but it hunts specific catch: mites, trichodecteses and other
parasites plentifully inhabiting wool of large mammals. Strong chewing jaws
permit this beetle easily crack even firm shells of mites.
The beetle is perfectly adapted to movement among hair – as at the flea, its
body is strongly compressed at sides, short and deep. Elytrums of the beetle
have the specific shape: they do not cover body from above, but cover it from
sides, adjoining along the bottom side of abdomen. Narrow-sided fur beetle has
not lost ability to fly. Moreover, this ability is vital for it – having exterminated
parasites on any animal, this beetle simply flies to another one. In flight
wide elytrums are stretched sideways, creating some elevating force similarly
to plane wings.
This species of beetles ise poisonous, as well as many representatives of its
family – in body of this insect the small amount of cantarid poison is accumulated.
Colouring of elytrums of the beetle is bright and warning: it is orange with
faltering longitudinal black strips. Usually birds or mammals do not search
this beetle specially, but can injure it, cleaning themselves. In case of danger
(for example when the animal is scratched or cleans wool by claws) the beetle
simply draws legs in and slips out from wool or feathers of the host on the
ground, and then flies up and returns to the body of the host.
At the fur beetle there are short tenacious legs, and this insect can quickly
run among wool or feathers of the host animal.
Eyes at this beetle are well advanced, because it leads an active habit of life
and can fly. With the help of sharp sense of smell the beetle easily finds parasites
sitting on large animal among wool. antennae of this beetle are feather-like
(it is an attribute of well advanced sense of smell) and short. When the beetle
creeps on body of the host animal, they are wrapped and hided under head.
This beetle lives in conditions of seasonal climate. It spends winter in shelters
on the ground, but in northern (warmer) parts of the area it occasionally spends
winter on animals.
Fertility of narrow-sided fur beetle is not so big: the female lays for all
life no more than thousand eggs in small fine portions. It pastes eggs one by
one on covers of the host animal. The larva bursts in one week. It develops
right on the body of host animal within approximately two months (larvae bursted
later, spend winter on the body of host animal). Larva is active, worm-looking,
with well advanced legs. Covers of its body are rather soft, but head capsule
is firm, and mandibles are well advanced. It also is a predator and attacks
parasitic and blood-sucking insects. While the larva is small, it feeds with
mortifying particles of host animal skin. Larva pupates in ground. Duration
of metamorphosis is about two weeks.
Wyrm
Beetle (Vermiaoptera vorax)
Order: Beetles (Coleoptera)
Family: Rove beetles (Staphylinidae)
Habitat: New Zealand, both islands, caves inhabited by colonies of bats.

Picture by Biolog
The existence of large colonies of roosting bats in caves
gives rise to a particular set of conditions. Air is warmer, more humid, and
piles of dung accumulate on the cave floor. This allows for a small ecosystem
to exist around this resource, and one insect that has specialized to this is
the Wyrm Beetle.
This beetle is a kind of large rove beetle, inhabiting the cave floors of bat
colonies, feeding on the other kinds of insects attracted to feed on the dung,
as well as on baby bats which have fallen down onto the floor of the cave. Various
insects are taken, including carrion beetles, cockroaches, earwigs and small
cave wetas.
It is unusual for its family in being a good deal larger, up to 10cm long, and
by its original shape. Body and abdomen of Wyrm Beetle is elongated and slender,
resembling a worm, this allows it to burrow and wriggle amongst the masses of
guano in search of prey. Legs are short but relatively powerful, wings are absent
(it is flightless), and head forms an oblong rectangle. Mandibles are very large,
curved and sharp, with this it kills and butchers its prey. Antennae are long,
with lobe shaped segments, face is hairy and sense of smell and touch is very
keen, eyes are extremely reduced, insect almost blind. Colour of body is a light
brown, turning to dark brown and black on the head and mandibles, where chitinous
covering is thicker, legs are slightly darker than body. This insect is generally
more active when the bats are roosting, but may be active any time of day.
The female lays up to 200 eggs with an incubation of about 2 or 3 days. Female
will lay eggs 8 at a time, in a secluded crevice close to the cave wall. Larvae
are tiny when they hatch, and may sometimes cling to the feathers of birds which
visit the cave, and be spread to other locations, in this way it is distributed
throughout most of New Zealand’s caves systems. As with their parents, larvae
are predators of insects, initially feeding on tiny ones, larvae become larger
and pupate after 3 moults. Individual Wyrm Beetle may live as long as 200 days,
predators include centipedes, birds and lizards which venture into the cave.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Menthol
leaf beetle (Menthodoromela chemica)
Order: Beetles (Coleoptera)
Family: Leaf beetles (Chrysomelidae)
Habitat: Southern and South-Eastern Asia, thickets of mint tree.

Picture by Biolog
At each species of plants numerous species of animals feed.
Sometimes the plant protects itself from numerous eaters, accumulating in leaves
and stalks various chemical substances frightening off the majority of herbivores.
But evolution develops the absolute weapon seldom: for some animals chemical
protection of plants is not a problem, and due to the adaptation produced by
plant, they practically lose competitors.
Among shades of greenery of tropical rainforests silvery “caps” of crones of
mint tree (Menthoxylon leucophyllum)
are remarkable. This plant accumulates in foliage plenty of menthol giving to
greenery of plant characteristic smack. But not all insects bypass its foliage.
Many leaves of this tree are perforated by holes, and their edges are gnawed.
Originators of damages openly feed on leaves of tree. They are big-bellied beetles
of emerald-green color about 1 cm long. At them there are thin short antennae
and tenacious legs. They lazily creep on branch, or motionlessly sit on leaves,
gnawing their edges. Near to adult insects larvae feed. They gnaw soft pulp
of leaves, leaving from them only net of nerves. It is menthol leaf beetle -
one of few insects eating leaves of mint tree.
By the habit of life these beetles differ only a little from other leaf beetles.
But in connection with specific diet at this insect the special protective adaptation
has appeared. For protection against enemies many insects accumulate various
liquids having unpleasant taste. Menthol leaf beetle is not exception here.
But it has ability to accumulate in special glands menthol from plant, and adds
it in pungent liquid secreting for protection. The principle of action of the
chemical weapon of this beetle is, that menthol irritates flavoring receptors
of predator and by that strengthens action of poison.
Menthol leaf beetles lay eggs on bottom side of leaves where they will not dry
up right in the sun. Larvae in first days of life keep on the bottom side of
leaves, then creep to top one. They are light green, but through translucent
cuticle interior and intestines, filled with chewed greenery are visible. Development
of larvae proceeds about one month. For one year the menthol leaf beetle can
produce up to 4 generations.
Hairy
Forest Longicorn (Prionoplus neohuhu)
Order: Beetles (Coleoptera)
Family: Longhorns (Cerambycidae)
Habitat: Conifer forests in both the North and South Islands of New Zealand,
including montane forests.
An Entomological icon of New Zealand during the age of man was the Huhu Beetle
(Prionoplus reticularis), a fairly large longhorn beetle that was ubiquitous
wherever there were forests or trees. Being notable to the Maori, the grubs
were fatty and good to eat, and it was an unpretentious animal which only needed
rotting wood in order to grow. It was also notable for doing significant damage
to commercial timbers and houses as a pest.
One of its descendants in the Neocene is the Hairy Forest Longicorn, an even
larger beetle than its ancestor, measuring up to 7.2 centimetres as an adult.
Elytra are dark brown, almost black, and un-patterned, though bearing longitudinal
ridges, the thorax, head and abdomen are chocolate-coloured and covered in a
dense coat of long bristly hair. Antennae almost surpass the length of the animal
(those of the males are longer and more sensitive), mandibles are large and
sharp and the legs bear rows of defensive spines. Adults do not feed, and the
entire life of the grub consists of eating in order to pupate and eventually
breed. The larvae are large and pale, reaching up to 9 centimetres long, feeding
exclusively on conifer wood, and contributing to the bio-degrading of the forest
floor.
Adult males rove about in search o f females, and tussle over the right to breed
by biting with their mandibles and grappling fiercely. Egg-laying may occur
at any time of year so long as it is not too dry, and each female lays as many
as 70 eggs (sometimes as few as 40) in crevices in or under bark. Each egg is
3 millimetres long and the larva emerges by biting the casing open. The larval
stage lasts up to three years, and the grubs may grow to be quite large and
fat. The mature grub makes a special pupal chamber inside a space within the
wood lined with faeces, pupal phase last 25 days. The adult stage lives for
only 2 weeks in which they endeavour to mate and lay eggs, both sexes can fly
powerfully in short bursts. Both adults and grubs have many predators including
insectivorous mammals such as hedgehogs and rodents, as well as foraging birds,
particularly parrots that actively extract them from the wood, reptiles such
as lizards also feed on the adults.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Shepherd beetle (Dolichocurculio pastor)
Order: Beetles (Coleoptera)
Family: True weevils (Curculionidae)
Habitat: Mediterranean deserts, plants of leafless
paunchstem invaded by water-bearing
plant louse.

Picture by Amplion, colorization by Biolog
Initial image by Amplion |
Symbiosis is very favourable life strategy: mutually helping
to each other, representatives of different species get ability to survive in
extreme conditions and to form productive communities. Examples of this benefit
are represented, accordingly, by lichens and coral polyps. Besides them, in
nature there is a set of other species of live beings forming symbiotic connections
and receiving success in survival due to it.
In desert at the edge of ultrahaline Mediterranean swamps sources of food are
poor, but some species find an opportunity for specialization and exist due
to it successfully even in extreme conditions. Among plants of this area there
are many succulents reliably protecting stocks of water from herbivores with
the help of poisons. One of these plants is leafless paunchstem, a succulent
umbellate plant with thick leafless stalks. This plant is a habitat of various
species of invertebrates. One of inhabitants of this plant is a large species
of weevil beetles about 3 cm long, named shepherd beetle. This insect has ash-grey
color with black legs and bottom part of body.
Adult insect of this species is omnivore. Shepherd beetle feeds on nectar of
paunchstem, which is plentifully secreting at the inflorescences, but receives
additional protein food, eating various small insects flying to inflorescences
of paunchstem. The plant does not receive from its presence of any benefit,
but there is one species of insects, for which the union with shepherd beetle
has a vital importance.
In desert water is a biggest value for animals. Sap of paunchstem contains a
plenty of alkaloids and is not suitable for drinking almost for all species
of animals, except for small species of sucking insects, the water-bearing plant
louse. In colonies of this plant louse shepherd beetle has found a plentiful
source of drinking water. Each individual of plant louse from time to time secrets
a drop of almost clean water not containing poisons and suitable for drinking.
With the help of head extended as a proboscis this beetle easily gathers water
in colony of plant lice, and due to it survives in hot desert.
The head of shepherd beetle is extended to very long proboscis exceeding in
length thorax and abdomen in total. The proboscis is arc-like bent from top
to bottom, the mouth and short antennae are located on its tip. On the top part
of proboscis two or three pairs of acute spikes develop, sticking upwards and
in sides. Neck of this insect is very mobile. Due to its movements head may
be turned in under body and be curved forward by sharp movement – this way beetle
strikes a blow by proboscis, armed with spikes. By such impacts of proboscis
shepherd beetle drives away from plant lice of large insects which can injure
them – predatory beetles, solitary wasps and grasshoppers. However, at food
shortage it also can eat a small amount of water-bearing plant lice from preserving
colony. But this casual damage is not comparable with the benefit, brought by
this beetle to colonies of water-bearing lice, preserving them from enemies.
Legs of this beetle are very long – they are longer than a body of insect. Usually
shepherd beetle cautiously walks in colony of plant lice, gathering liquid secreted
by them. By short antennae and tentacles of lower lip this beetle tickles plant
lice, and licks off their secretions.
It is able to fly, but makes it reluctantly and flies slowly because of massive
head. Shepherd beetle flies at night and in the morning, while birds have not
woken up. Being attacked by bird it folds wings or turns in legs, and falls
on the ground, and then is hidden in cracks of ground under the plant.
When paunchstem plants begin blossoming, beetles move to their inflorescences
and eat nectar. Inflorescences of paunchstem, reaching one meter in diameter,
serve as a place of meeting of beetles of opposite sexes, and also as arena
for determine of relations between contender males.
Fertilized female of shepherd beetle searches for egg laying for absolutely
other species of plants – roots of paunchstem contain not less alkaloids, rather
than its overground part, and larvae of this species do not eat them. Due to
keen sense of smell the female of shepherd beetle searches for tubers of other
deserted plants, located deeply in ground. It is dug in the ground above the
tuber, and at the ground surface only a part of proboscis is visible. Having
extended an abdomen, the beetle female lays eggs in ground as deep, as possible,
and as close, as possible to the surface of tuber. The larva bursts from eggs
and at once searches by smell for a tuber of fodder plant. It gnaws through
the tuber and develops in its pulp during dry season. The larva keeps activity
in winter – it is necessary to have time to pass the metamorphosis. The next
year in spring it leaves tuber and pupates in the top layer of ground near to
sprouts of fodder plant. Within one month it turns to adult beetle and flies
out to search for fodder plant. Adult shepherd beetles perish at the end of
an autumn.
Kaikai
Harakeke (Pseudanagotus novis)
Order: Beetles (Coleoptera)
Family: Weevils (Curculionidae)
Habitat: Vegetated habitats on the North Island and the northern part of the
South Island, restricted to places where Flax grows.
The wildlife emblems of New Zealand suffered heavily from mankind's occupation,
many kinds such as Kiwi and Tuatara became completely extinct, leaving no descendants.
Botanical emblems of this country fared much better, including the Kauri Tree,
and in this case, the Flax plant (Phormium). Previously, during the Holocene,
a flightless species of Weevil (genus Anagotus) was specialised to feed exclusively
on this plant, but due to its vulnerable nature, it became extinct from predation
by introduced rats.
During human occupation a local un-specialised kind of boring-weevil (genus
Rhynchodes) was common and thrived all over the country. In the late Holocene
it developed into a form that preferred the fibrous leaves of the native flax.
The result is the Kaikai Harakeke, name derives from the Maori words for “Flax
Eater”.
The adult of this beetle contrasts to its ancestor somewhat larger size, commonly
reaching up to 3.5 centimetres long. Its exoskeleton is mostly devoid of hairs
on its upper side, and is covered in thick longitudinal ridges, spotted with
light grey on a dark brown background. Underside bears fair-coloured, scale-like
hairs. Its snout is shorter and thicker than that of its ancestor, being adapted
for chewing the fibrous leaf-edges of the flax plant, just as with the extinct
Anagotus weevils. Legs are splayed and relatively long for a weevil, bearing
large claws at their tips, this beetle is an adept but slow climber. By necessity,
it retains the ability to fly, and when disturbed it will launch itself and
flee.
Feeding habits somewhat resemble its archetype of the Holocene, it chews the
edges of the large flax leaves into a distinct crescent shaped feeding trace,
but will also bore holes into the base of the leaf. This species is most active
at night, hiding beneath the leaves of the flax, or inside the base of the plant,
by day. Differences between the sexes are limited to the placement of the antenna,
farther back along the rostrum in females, closer to the front in males.
Egg-laying occurs between December and March, the female bores a hole at the
base of the flax plant and lays up to 60 eggs over about a week. After about
8 days, the larvae hatch and feed by boring into the bases of the flax leaves.
After two weeks, they begin to pupate, the pupa stage lasts a further 2 weeks.
A single female may lay as many as 2 clutches in one season, by the time the
following summer arrives, the newly emerged adults are ready to breed.
Predators include various insect-feeding birds, lizards, rodents, and hedgehogs,
as well as larger predatory insects, lifespan of a single beetle may be as much
as 2 years.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Mould-loving
Cave-lady (Spelaeococcinella mucorophila)
Order: Beetles (Coleoptera)
Family: Ladybirds (Coccinellidae)
Habitat: New Zealand, guano-coated floors of caves inhabited by roosting bats.
Large congregations of animals that are more-or-less permanent, often eventually
result in communities of organisms that have evolved to exploit them. And so
it usually is for caves in which large numbers of bats roost, they attract roving
predators, insect decomposers that feed on their dung and corpses, and other
insects that feed on the decomposers. The bat colonies of New Zealand have large
piles of guano coating the cave floor, on which grows Velvet Guano Mould and
other kinds of decomposing fungus, these are fed upon by a specialised kind
of beetle, the Mould-loving Cave-lady.
This small beetle is in fact a kind of ladybird, descended from the fungus-eating
Illeis galbula, which was accidentally introduced into the North Island in the
age of man. Adults are up to 5 millimetres in length, body and legs are coloured
a pale brownish white, darker towards the tips of the legs. The thorax and elytra
are very pale, greyish white, with 3 patchy strips of slightly darker grey running
across it, this is a remnant of the contrasting colours of its ancestor. The
eyes are smaller than those of its ancestor, and have a smaller number of compound
lenses, sight is poor but still able to discern light and movement. Larvae are
usually only 8 millimetres long, pale white with darker grey spots running along
their sides.
They feed mostly on Velvet
Guano Mould and are attracted to patchy growths of it by the smell of its
spores. Many individual beetles may congregate to feed on a patch of mould,
remaining there until their stomachs are full, and sometimes even longer. Other
than this, it is usually a very active beetle, and not only crawls about but
frequently flies in search of fresh mould. Larva are also fairly active and
will commonly stand on end with their back end in the air, when not moving about.
When threatened by a predator, adults are able to secrete a bad-tasting substance
in defence.
They do not need to migrate to other areas in cold weather, as the warm cave
environment is usually stable. Reproduction can occur as many as 3 times a year,
and is generally dependant on the abundance of food, and humidity levels. Both
males and females produce pheromones when they are ready to breed, by which
they locate each-other, a copulating pair may stay joined for up to 2 hours.
Females are able to store sperm for a couple of months before laying eggs, if
they need to, eggs are laid near abundant sources of food such as a growth of
mould. The eggs are laid in clusters of up to 15, and 50-70 may be laid in one
breeding cycle. A single female may lay up to 1000 eggs within her lifetime.
Larva grow for about a month before pupating, pupa stage lasts up to 14 days,
lifespan may be as long as 1.5 years. Predators include various hunting kinds
of cave insect such as predatory beetles, centipedes and Cave
Wetas.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
“Termite
bus” beetle (Termitophorus caementiferus)
Order: Coleopters, or Beetles (Coleoptera)
Family: Dung beetles (Geotrupidae)
Habitat: savannas of Northern Africa.
In Neocene African savannas are populated with large animals like in human epoch.
But in the majority these ones are the representatives of other systematic groups,
rather than in human epoch. Elephants, rhinos and giraffes had been replaced
by other animals – the descendants
of hyraxes, cursorial lagomorphs
and giant ostriches. And
their manure represents the source of food for numerous invertebrates, mainly
for insects. Constantly renewed heaps of manure, left in savanna by large animals,
represent the nutrient medium where the special kind of biocenosis of various
invertebrates and fungi develops.
The important role in decomposition of the manure gathering at the pastures
is played by dung beetles. Among the set of species of these insects there is
one species surviving due to symbiotic relations with tiny-sized
species of manure-eating termites. With the greatest probability it may
be met in termitaries, and because of special role of this beetle in settling
of termites it is named “termite bus” beetle. This rather large insect begins
the life cycle as the thick worm-like grub carefully raising by termites in
their dwelling in thickness of manure heap. It feeds on manure, and at early
age while it is rather small, termites feed it as if their own congeners. Large
larvae can feed independently. As they can destroy galleries of termites, eating
manure, termites “move out” such larvae to the edge of termitary. In larval
stage of development “termite bus” beetle is the supplier of building material
for termites: its dung hardens in air as firm paste, and termites form of it
vaults and galleries of their dwelling. Hence the specific epithet: “caementiferus”
means “cement-bearing”. At the end of larval stage of life cycle this insect
makes of dung pulp strong cocoon in which termites can not disturb it in time
of metamorphosis. In addition the larva serves as the supplier of some vitamins
and other chemical compounds: the microflora in its intestines differs from
microflora of termites, and bacteria produce biologically active substances
which are partly used by the larva, and partly by the termites. This is one
more advantage receiving by termites from the larva of “termite bus” beetle.
The adult beetle of this species looks very impressively: this is an insect
of about 7 centimeters in length, of black color with metal green shine on elythra
and bluish shine on legs. Elythra of this insect have the coarse-grained facture.
Pronotum of this beetle has the special form at individuals of both genders:
on back part of pronotum there is the deep hole covered at the front edge with
original flat “screen” made of three fused horns. The internal part of hole
is covered with rigid bristles.
The flight of beetles coincides to a greater or lesser extent with swarming
of termites. During the courtship flight of termites, taking place at night,
short-winged and almost flightless young “queen” leaves termitary in common
with the part of “citizens”. The beetle leaves termitary, carrying such “passengers”
on itself. During the flight of the beetle termite “queen” emits pheromones
involving males. The adult beetle searches for heaps of manure suitable to settling,
and frequently flies to manure heap already carrying the couple of termites
ready to breeding. When the beetle is dug in manure, “screen” on its pronotum
protects soft-bodied termites from damages. In heap of manure the female of
“termite bus” beetle lays eggs. Larvae of this beetle can develop without the
help of termites. When the colony of termites becomes stronger, ergates start
to care of beetle larvae. Frequently females of this beetle lay eggs in available
colony of termites, partly destroying it. But termites quickly restore damages,
and eggs of the beetle appear in good conditions for incubation.
Besides termites, this bug carries on the body spores of mushroom Copragaricus
growing on manure of large herbivorous mammals.
This species of insects was discovered by Mutant, the forum member.
Tri-horned
camel-loving Dung Beetle (Camelophaga tricornis)
Order: Beetles (Coleoptera)
Family: Dung beetles (Geotrupidae)
Habitat: Savannah and wooded grassland of central Meganesia.
Foreign dung beetles were introduced to Australia in order to make the farming
of bovid livestock sustainable, and to suppress the propagation of dung flies
that spread disease. Native dung beetles already existed but could not process
profuse cow and sheep dung in the same way. In the Neocene, many species descended
from the introduced kinds exist to feed on the dung of ungulates, which are
also descended from species brought by man.
Camel-loving dung beetles are the most common kind on the bushy savannahs of
central Meganesia, and thrive feeding on the dung left by various
kinds of camel.
This species is small and rounded, 3 centimetres long, fully matching the typical
scarab shape. 2 short horns and one long horn are present on the pronotum of
the male. The carapace is black with a blue shiny iridescence, and the digging
pair of legs is large. Wings are whitish and only visible in flight. Mouthparts
are adapted to eating and shovelling dung.
This species reproduces in spring and summer; the female attracts the male with
pheromones. As is typical for scarabs, the male and female dig a circular hole
to bury the dung in and lay the eggs inside. Dung is taken into the burrow as
a ball which is rolled in the typical way. The female lays the eggs in the burrow
soon after, and the young soon hatch and start feeding on the dung. Grubs go
through 3 skin changes until they reach the non-eating pupa stage, conditions
and availability of food can determine the size of males. The larvae can survive
unfavourable conditions such as drought during a bad year. After pupation the
newly emerged beetles dig to the surface in order to find dung of their own.
Sexual maturity is reached at 2 weeks.
These insects serve not only to process animal waste and fertilize the soil,
but to aerate the soil in order to promote plant growth. They are ubiquitous
on the bushy savannah and are a constant presence around dung. 5 similar species
exist within this genus, and differ by number and size of horns, colour, and
the time of year in which they are active, so as to avoid competition. Lifespan
is up to 3 years, and both adults and grubs are preyed upon by frogs, toads,
lizards, birds and small marsupials.
This species of invertebrates was discovered by Timothy Donald Morris, Adelaida, Australia.
Four-striped
honey bee (Apis tetrafasciata)
Order: Hymenopters (Hymenoptera)
Family: True bees (Apidae)
Habitat: tropical forests of South America.

Picture by Biolog

Picture by Pavel Volkov
In Holocene, people produced the Africanized bees by the cross-breeding
between the Western honey bee (Apis mellifera) and African form of the same
species. The Africanized honey bee was first introduced to South America in
order to increase honey production, but swarms escaped quarantine and the species
has spread throughout South America and North America. In Neocene Africanized
bees give rise to the subgenus Striatoapis – striped honey bees. The type species
of this subgenus is the four-striped honey bee, an insect that lives in tropical
areas of South America.
This species has black coloration with characteristic pattern of four bright
reddish-orange cross-stripes on the abdomen. Not measuring more than 30 mm and
with tongues about 7 mm long, these bees frequently form swarms and belong to
the most common pollinators of tropical trees. Their nests are made in hollowed
tree, but this insect also makes them in the ground. Four-striped honey bee
is very defensive of their hive, forming swarms to attack predators.
Like their ancestors, four-striped honey bee is eusocial insect forming colonies
with a single fertile female (or “queen”), many normally non-reproductive females
or “workers”, and seasonally a small proportion of fertile males or “drones”.
Some colonies can house tens of thousands of bees. Colony activities are organized
by complex communication between individuals, through both pheromones and the
dance language.
Their life cycle is nearly the same as their ancestors. Eggs are laid singly
in a cell in a wax honeycomb produced and shaped by the worker bees. Using her
spermatheca, the queen can choose to fertilize the egg she is laying, usually
depending on into which cell she is laying. Drones develop from non-fertilized
eggs and are haploid, while females (queens and worker bees) develop from fertilized
eggs and are diploid. Larvae are initially fed on royal jelly produced by worker
bees, switching later to honey and pollen. The exception is a larva fed solely
on royal jelly, which will develop into a queen bee. The larva undergoes several
moultings before making a cocoon within the cell, and pupating.
Young worker bees, sometimes called “nurse bees”, clean the hive and feed the
larvae. When their royal jelly-producing glands begin to atrophy, they begin
building comb cells. They progress to other within-colony tasks as they become
older, such as receiving nectar and pollen from foragers, and guarding the hive.
Later still, a worker takes its first orientation flights and finally leaves
the hive and typically spends the rest of its life as a forager. Worker bees
cooperate to find food and use a pattern of “dancing” to communicate information
regarding food resources with each other.
Virgin queens go on mating flights away from their home colony to a drone congregation
area, and mate with multiple drones before returning. The drones die in the
act of mating. Queens of four-striped honey bee do not mate with drones from
their home colony.
Colonies are established not by solitary queens, as in most bees, but by groups
known as “swarms”, which consist of a mated queen and a large contingent of
worker bees. This group moves to a nest site which was scouted by worker bees
beforehand and whose location is communicated with a special type of dance.
Once the swarm arrives, they immediately construct a new wax comb and begin
to raise new worker brood.
The subgenus Striatoapis includes some other species; the coloration pattern
of all of them is uniform and represents completely black background with bright
reddish-orange stripes. The number of stripes varies between the species. The
species are following:
Three-striped
honey bee (Apis trifasciata) living in southern South America is well
adapted to cold winter; they stop flights when the temperature drops and begin
crowding into the central area of the hive to form a “winter cluster”. The worker
bees huddle around the queen bee at the center of the cluster, shivering to
keep the temperature in the center between 27 °C at the beginning of winter
(during the broodless period) and 34 °C once the queen resumes laying eggs.
During the winter they consume their stored honey to produce body heat. Large
colony can store up to 5-6 kg of honey and protects it ferociously against rodents
and small carnivores. The worker bees rotate through the cluster from the outside
to the inside so that no one bee gets too cold. They measure 20 mm, with tongues
of 5 mm long. Their name comes from the three reddish-orange stripes on their
abdomens.
Two-striped
honey bee (Apis difasciata) inhabits elevated areas of the Andes; this
species has the same strategy for survive in the winter that the three-striped
honey bee. Its name comes from the two reddish-orange stripes on its abdomen.
These bees measure 25 mm, with tongues of 6 mm, and are “hairy” compared to
other species. Two-striped honey bees construct their hives in the ground and
between large stones.

Picture by Pavel Volkov
Antiguan banded honey bee (Apis monofasciata) lives in forest of Great Antigua island, being isolated from related continental forms. This bee is 18 mm long, and its tongue is 9 mm long – it is an adaptation for pollinating of tropical plants having tube-like flowers. This species has only one red stripe across its abdomen. Antiguan honey bee can made hives both in trees and ground.
This species was discovered by João Vitor Coutinho, Brazil
Great
Lonesome Humble Bee (Notoneobombus rex)
Order: Bees, Wasps, Ants (Hymenoptera)
Family: Bees (Apidae)
Habitat: Sub-tropical lowland forest and woods of North Island, New Zealand.

Picture by Biolog
Invasive insects are a feature of human occupation in the
Holocene, any place colonized by man brings introduced forms as well as stow-aways.
Bumblebees (genus Bombus) found their way to New Zealand in the age of man,
and have left descendants in the Neocene. Most of these resemble their ancestor,
with some exceptions, such as the Great Lonesome Humble Bee.
This bee has become a solitary nester, and does not form the small family colonies
that its ancestors did. It inhabits the sub-tropical forests of the North Island,
feeding on nectar, pollen, rotting fruit and freely flowing tree resin. It is
very large for a bee, length is about 4 centimeters, and wingspan is 6.5 centimeters,
body is very fat and covered in black hairs, wings are iridescent. Great Lonesome
Humble Bees are active by day, and produce a very distinct loud buzz in flight.
Unlike true solitary bees it does sting, and this is quite painful, a necessary
defense in a forest full of predatory birds. Flight is powerful but slow, and
they will also crawl or climb to reach food.
Breeding and nesting occur from May until August, mating occurs once a year.
The female digs a vertical, cylindrical burrow in firm soil, in which it will
deposit a gallery of multiple egg chambers, several are laid over a matter of
months. Their development is synchronized so that they all emerge at once and
do not compete. The female guards the nest fiercely; stinging and killing predatory
insects that venture too near. Young bees can fly at about 3 days after emerging
from the pupa. Males fertilize females during the courtship flight and die soon
after that. Females make nests, rear the posterity, take care of it during the
wintering, then reach senescence and die in early spring.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Manduri
(Manduri infestantibus)
Order: Hymenopters (Hymenoptera)
Family: Bees (Apidae)
Habitat: tropical and subtropical areas of South America.

Picture by Pavel Volkov
In the South America of the Neocene, not only the Africanized
bees survived. Some of the members of the Genus Melipona were capable of
surviving the pressure that it suffered during the Holocene. These stingless
bee survived competition and destruction of their habitats by becoming aggressive
and developing large heads with large mandibles capable of cutting wood. The
Manduri is one of the species of this genus. This word was used as a regional
name in Brazil for their ancestors. This insect inhabits the tropical and subtropical
areas of South America.
They have dark black, rounded bodies with slightly curved antennae and translucent
wings. Size is from 29 to 30 mm, and they are more heavily build than the Apis
bee. This bee can be identified by the large bright yellow head and black mandibles.
Their mandibles are large and well developed, giving them a wasp-like appearance.
The abdomens of the queens swell with ovarian development, making older queens
larger than workers which is typical of most social bees. Queens vary slightly
in their coloring, having brown eyes and brown hair compared to the black eyes
and hair of worker bees. Workers are smaller than the queen. Workers have black
eyes and black hair on their thorax and abdomen. Older workers will go out foraging
while younger workers, 12 to 21 days old, will construct and provision cells
in the comb.
Like their ancestors, they are highly eusocial bees that are characterized by
having perennial colonies that are typically headed by a single-mated queen.
The average number of adult workers and queens within a colony is 300–400. There
is rare cases of temporal polygyny within a colonies having more than one egg-laying
queen.
New colonies are established in a slow process, when the number of worker bees
exceed 500 or 600 individuals in the parent colony. Then, a number of worker
bees start to build a new nest by using their large mandibles to carve a tree
cavity well suited for this purpose, and store honey and pollen in there. When
the new nest is ready, a "princess bee" (mated gyne) join the workers,
and if accepted it starts laying eggs and becomes the new queen. After a while
the abdomen of the new queen expands to 3 or more times the initial size (a
phenomenon called physogastrism) and it becomes incapable of flying, never leaving
the nest again.
Eggs are laid singly in a cell in a wax honeycomb, produced and shaped by the
worker bees. Using her spermatheca, the queen can choose to fertilize the egg
she is laying, usually depending on into which cell she is laying. Drones develop
from unfertilised eggs and are haploid, while females (queens and worker bees)
develop from fertilised eggs and are diploid. Larvae are initially fed with
royal jelly produced by worker bees, later switching to honey and pollen. The
exception is a larva fed solely on royal jelly, which will develop into a queen
bee. The larva undergoes several moultings before spinning a cocoon within the
cell, and pupating.
Young worker bees, sometimes called "nurse bees", clean the hive and
feed the larvae. When their royal jelly-producing glands begin to atrophy, they
begin building comb cells. They progress to other within-colony tasks as they
become older, such as receiving nectar and pollen from foragers, and guarding
the hive. Later still, a worker takes her first orientation flights and finally
leaves the hive and typically spends the remainder of her life as a forager.
Worker bees cooperate to find food and use a pattern of "dancing” to communicate
information regarding resources with each other.
While they feed on nectar of different flowers, the manduri and honey bee (subgenus
Striatoapis) compete for hollowed trees to make their hives. The first will
carve their own nests that can be usurped by the second. Such conflicts can
result in fights between swarms of workers of both species. The honey bees will
try to sting their rivals, while the manduri will use their mandibles to cut
off their heads. The species emerging victorious in such disputes can depend
of several factors, such as the number of bees involved.
This insect species was discovered by João Vitor Coutinho, Brazil.
Bird-eating
wasp (Ornithosphex ornithophagus)
Order: Hymenoptera (Hymenoptera)
Family: Digger wasps (Sphecidae)
Habitat: Great Antigua, tropical forests.

Picture by Fanboyphilosopher
During evolution solitary wasps had turned to extremely specialized
predators with complex behaviour. At these insects female specially prepares
for the posterity insects or other invertebrates of certain species – it paralyzes
them by sting, pulls to specially prepared nest and lays egg on the body of
prey. The larva of wasp devours the paralyzed prey alive, protected by nest.
In Neocene separate species of parasitic Hymenoptera had made original “breakthrough”
in development of parasitism. They passed to parasitizing on vertebrate animals.
In Southeast Asia doctor
wasp (Hygeiosphex antibioticus) lays eggs in hypoderm of small mammals,
and its larva develops in body of animal, releasing it from other parasites.
The species imitating it, the special kind of ichneumons, killer
wasp (Pseudohygeia placebo), also attacks mammals. Its larva even breeds
parthenogenetically in host organism. At Great Antigua Island some kinds of
wasps parasitizing on vertebrates live. Frog-devouring
wasp (Batrachovespa batrachovora) infects tadpoles of local frogs, and develops
for a long time in body of animal, leaving it when host animal has undergone
metamorphosis and has settled on land. And one more species of wasps from Great
Antigua had passed to parasitism on birds independently of these species, and
makes it by very complex way.
Antiguan bird-eating wasp is rather large insect. Length of its body is about
35 mm. It differs in fragile and graceful constitution: it has a long thin body,
and short drop-like abdomen is located on long pedicle. The female of bird-eating
wasp is covered with velvety grey hairs, giving to its body silky shine. The
tip of abdomen at this insect is black and hairless, and has bright metal shine.
Male differs from female in smaller size and more expressed silvery shine.
This wasp lays eggs in incubating eggs of medium-sized birds – pigeons, small
parrots, passerines and other forest birds. Wasp searches a clutch with
the help of sense of smell, and easily penetrates even into the spherical nests
or nests made in hollows. The ovipositor at this species of wasps is very strong;
it easily pierces egg-shell near the air chamber. During the egg laying special
glands produce substance, sticking on the spot an aperture from prick of sting
and thus the development of bird's egg is not broken. In egg of bird tiny larva
finds a developing bird embryo, creeps into its digestive path and sticks to
wall of intestines. Modified legs turned into suckers with small claws on edges
and semicircular sucker on the back end of body work as organs an attachment
at this one. The larva of bird-eating wasp feeds on blood of developing embryo.
Usually from the eggs, infected by this wasp, weaker nestling having more chances
to die hatches. If it happens, larva turns to active scavenger: in body of dead
nestling wasp larva starts to eat meat, growing up to size necessary for further
development much faster. If the nestling survives, the wasp larva continues
eating of its blood and grows slower. In this case development of larva lasts
till about six weeks. To the moment of preparation to metamorphosis it reaches
length of about 10 cm. The body of larva at this stage is very long and flat,
therefore bird in which body it parasitizes, continues rather normal existence.
The larva ready to metamorphosis leaves body of host bird over intestines, and
pupates in ground, in litter of nest of host bird, in forest litter or in moss
thickets.
Mole
wasp (Talposphex terrestris)
Order: Hymenoptera (Hymenoptera)
Family: Burrowing fossores (Terrasphecidae)
Habitat: Great Antigua, ground in rainforests.

Picture by Amplion
During evolution the species can get advantage in struggle
for existence, having changed habit of life. As the stimulus to it the free
ecological niche, or
the big competitiveness of the beginner in comparison with species, that
occupied this habitat earlier, can serve. At the island Great Antigua which
separates
Atlantic Ocean and Caribbean Sea, one of species of solitary wasps has gone
such way of evolution. From the mobile flyer this insect has partly turned
to digging creature which does not see sunlight. The new species of insects,
the mole wasp, differs from neighbours by original habit of life: males look
as usual wasps, hide in various shelters in daytime, and fly at night; females
are not able to fly, burrow in ground and creep out to the surface only before
pairing.
Mole wasp is a small insect. The shape of male and female sharply differs
in connection with difference in way of life.
Length of body of the female is up to 3 cm. Body is lengthened, flexible,
covered with strong chitinous armour. Wide shovel-like head is the main instrument
of ground digging. Eyes of the female are small, hided in small holes on
edges
of head and protected by “eyebrows” composed by bristles. The upper lip is
well advanced, serving for ground digging. Wings of the female have lost
function of flight – they are dense and skinny, covering body of insect similarly
to
elytrums of beetle. On edges of wings small hooks grow, due to which wings
are densely linked with each other. All legs of the female are rather short,
with wide paws. The forward pair of legs is supplied with small thorns on
edge – the wasp loosens ground by them, burrowing and repairing holes.
Male strongly differs from the female: it is a long-legged wasp about the
same size, but of more graceful constitution with short rounded abdomen sitting
on long stem. At it there is mobile head with sensitive feather-like antennae
and big eyes. At the male there is very keen sense of smell – its success
in
breeding depends on how soon it will find out the female and will couple
with her. The body of the male is colored black with violet metal shine.
Wings of
male are narrow and translucent, with black basic nerves.
Male and female eat different kinds of food: male sucks nectar of colors
and wood sap flowing down from damaged bark, and female is a predator. Her
catch
is small digging insects and their larvae, and also small soil worms.
This species of wasps is parasite of invertebrates. Usually single wasps
lay eggs on caterpillars of butterflies and other insects living openly,
and also
on spiders. Frequently they paralyze prey by poison, and larva simply eats
alive “canned food”, reserved in nest. Mole wasp avoids competition to them
due to that its larvae develop in completely other species of invertebrates.
This species of wasps parasitizes on earthworms and other digging invertebrates
– larvae of cicadas and beetles, root scale insects and other sedentary insects.
For pairing at night female creeps out on ground surface and hides under
leaves. She involves males with smell which is emitting out from the special
gland.
Having felt it, male starts to run on leaves, as if a police dog, trying
to find its source. Having found out the female, he rakes leaves above her,
quickly
fertilizes her and departs for searches of new females.
Fertilized female digs in the ground. One pairing is enough for her to lay
normal eggs all next life. The female of mole wasp moves under ground in
its own tunnels, but more often it uses burrows dug by its potential prey
– worms
and other insects.
For egg laying the female searches by smell holes have been dug by suitable
species of animals. This species has some ecological races preferring for
development different species of host animal: some races prefer worms, others
like larvae
of beetles, third race is not so legible in choice of host insect. The form
parasitizing on cicadas is especially rare: it is connected with the circumstance
that at some cicadas there is very long term of development (much longer,
than at the wasp), and their larvae meet not so frequently as at other species.
Having found prey, the female wasp chases it and gets to it’s back. Supporting
against ceiling of hole, it presses prey to the ground and deprives with
an opportunity to move, then wasp puts to it sting, laying egg in body of
chosen
animal. Larvae of insects and worms on which this species parasitizes, in
some cases many times over surpass the adult mole wasp in weight. In some
hours
from eggs thin translucent larva bursts immediately starting to eat from
within their alive cradle. Sooner or later it puts to the host animal damages
incompatible
to life. Worms on which the wasp parasitizes, are in best condition, rather
than insects - they even can tear itself half-and-half and to be released
in such a way from the parasite, and regenerate after that. After destruction
of the host animal larva very quickly eats everything, that it is possible
to have eaten, and pupates under protection of remains of the had eaten animal.
For all adult life (approximately 2 months) the female can lay up to 20 -
30
eggs.
If the larva turns to male, difficult travel upward to ground surface is
necessary to it. Many males can not make it and perish. But it is quite enough
the number
of survived males to fertilize females. Female right after emerging appears
in native habitat. It eats various soil animals for some days, accumulating
in body the fat necessary for formation of eggs. Only after that at hot tropical
night it digs burrow to the ground surface, and waits for the male.
Snail
diving wasp (Aquavespilla helicivora)
Order: Hymenoptera (Hymenoptera)
Family: Diving wasps (Aquavespidae)
Habitat: rivers and lakes of South America, islands of Caribbean region, Florida.
South America of Neocene epoch, as well as in human epoch, has an extensive
circuit of rivers and lakes. The huge river of the past, Amazon, had divided
to two rivers flowing almost in parallel each other, but their basins nevertheless
remain biggest ones in the world. For inhabitants of these places it appears
very favorable to adapt to life in water: here it is possible to find a lot
of food, and the competition is sometimes lower, than on land or in wood crones.
Therefore in rivers of Amazon region it is possible to meet representatives
of various groups of animals, at times of rather unexpected ones.
In shallow, but rather wide forest river, which banks are overgrown with set
of plants, it is possible to see small insects sitting on leaves above water.
Their bodies are colored grey; therefore such insect is clearly visible at
the background of green and reddish leaves. Being frightened off, insect immediately
jumps off in water, turning as if in quicksilver ball, and quickly swims between
plants and driftwood. In some minutes it emerges to the surface of water, clings
to petiole of leaves, and stops for some time. Having convinced, that danger
has passed, this insect creeps on leaves and then it can be distinguished in
full details.
It is obviously not the beetle: it does not have characteristic smooth elytrums.
Narrow “waist” indicates that it is not a fly. This insect is snail diving wasp,
a solitary wasp, the representative of Hymenoptera order. In New World, these
insects have adapted to aquatic life, having formed the separate family which
has reached certain success.
Length of body of this insect is about 2 cm. The body has features of adaptation
to swimming: it is short and rounded. Abdominal stem at this species is rather
short and thick. Colouring of body is metallic – grey; wings are transparent.
Legs of snail diving wasp also are adapted to aquatic way of life. Two forward
pairs of legs have kept the universality, but the hind pair became larger;
shin and last segments on it are flat and wide, forming oar-like structure.
The main adaptation to aquatic way of life is at the surface of body of this
insect: the wasp is covered with water-repellent hairs, and glands on abdomen
secret paste-like wax substance. Under water this wasp is similar to water
spider (Argyroneta): due to hairs and greasing its body is surrounded with
layer of air; that’s why the body of insect seems silvery, as if quicksilver.
Only rowing hinder pair of legs juts out from air bubble.
The unusual habit of life of the diving wasp is connected with the special circumstance:
this species of hymenopters parasitizes on numerous snails inhabiting rivers
and bogs of tropical area of New World. Wasp searches under water snails by
smell, feeling by antennae trace of slime leaving by mollusk at movement. Wasp
finds snail and cautiously swims up to it. Having felt approaching of insect,
snail hides in shell, but wasp is able to wait. It cautiously clings to shell
of snail, and stands motionless. When the snail, deceived by imaginary calmness,
creeps out from shell, wasp pricks it by sting, laying egg in creeping sole
of mollusk. In some days from it small larva bursts. It parasitizes in creeping
sole of snail, gradually eating away its muscles. Also the larva eats part of
liver and sexual glands of mollusk. It eats gradually, and mollusk has time
to restore partly injured organs. Except for direct physical harm larva influences
life of host snail with the help of biologically active substances emitting
with it. Had reached the certain age, the larva with the help of secreted hormones
is forcing snail to leave water. Actually it is the death sentence to snail.
When the mollusk creeps out to damp ground of riverbank, it still can live any
time. But the larva of the wasp will not permit it to make: it starts to eat
actively, and for any some hours eats everything, leaving from a snail only
empty shell. During this time larva grows up larger then adult wasp. Having
to had eaten snail, wasp larva blocks shell fauces with mix of dung and secretions
of special rectal glands, forming some kind of partition. Under its protection
larva pupates in empty shell. About one week after the partition bursts, and
from behind it the young wasp creeps out. Having dried, the insect spreads wings,
and flies for searches of the male. Young females emit odorous substances by
which they are found by males. Fertilized females fly to bogs and begin life
of the swimming hunter, searching for snails. For the short life the female
wasp may lay up to 20 eggs.
The genus of diving wasps includes some species; each of them parasitizes on
certain species of mollusks:
Tiny
diving wasp (Aquavespilla minimissima) is tiny wasp about
4 mm long at all. It differs in coal-black colouring with metal
shine; by proportions it is similar to winged ant. This species
parasitizes on small orb snails (Planorbidae). Because of very
small size is not able to swim, creeping under water. To remain
under water longer, the insect has developed an interesting way
to update a stock of air: wasp gnaws by mandibles leaves of aquatic
plant, and simply rises above bladders of gas rich the oxygen formed
at photosynthesis, emitting from plant. Wasp shakes surplus of
gas from body by legs. Due to such adaptation the insect can remain
under water all the day.
Lung
diving wasp (Aq. pulmovora) is the internal parasite of
the snails, breathing by lung. It meets in vicinities of savanna
rivers and others partly or completely drying up reservoirs. It
is small (length of a body about 6 mm), metallic green wasp with
reduced wings. It swims and dives very well, similarly to the whirligig
beetle (Gyrinus). It parasitizes in large snails, settling in their
lung cavities. Wasp lays only one egg in wall of respiratory tube
of snail. Further in development the embryo divides to some parts,
and as a result from one eggs it appears at once about ten larvae
(similar feature was known at some representatives of ichneumons
(Ichneumonidae), close relatives of wasps). They settle in walls
of lung cavities, rich in blood vessels, and eat blood of snail.
In dry season when reservoirs dry up, and snail digs in silt, larvae
of the wasp start to develop roughly. Soon they devour the struck
snail, and wait a dry season under protection of its shell. In
the beginning of rain season they abandon shell and depart to searches
of new prey.
Frog-devouring
wasp (Batrachovespa batrachovora)
Order: Hymenoptera (Hymenoptera)
Family: Diving wasps (Aquavespidae)
Habitat: rivers and lakes of South America and the south of Northern America.
In family of water wasps not only the species infecting small animals had appeared.
In shallow rivers and ponds usual in selva of South America and the south of
Northern America, the set of frog species lives. And one species of water wasps
has adapted to parasitize in them.
Frog-devouring wasp is appreciably larger and more massive than its relatives
parasitizing in snails. Its body reaches length 2.5 - 3 cm (male is smaller
than female). This wasp differs in very thin body, its abdomen sits on long
stem. The body is colored black with red cross band on stem and spot on thorax
between wings. Males of this species have more red color in coloration, than
females, sometimes all abdomen of the male is colored red.
Back legs at males are usual, and at females they are modified to swimming ones.
Shins at them are wide and flat, and at external edge of shin there is a line
of hairs increasing rowing surface. The frog-devouring wasp also has glands
emitting water-repellent greasing. As against snail wasps, it does not grease
all body, limiting only with abdomen (where at insects respiratory apertures
open) and wings. In case of need, for example, rescuing from fish, this wasp
can, having gathered speed under water, jump out in air, and at once depart.
This species differs in some features of life cycle connected with parasitizing
in vertebrate animals.
For breeding female ready to lay eggs, dives and searches for flight of tadpoles.
It correctly defines by smell among tadpoles of some species of frogs representatives
of the necessary species. The wasp chooses tadpoles of largest species of frogs
and toads. Usually wasp creeps to flight of tadpoles on floating plants, having
turned over upwards by abdomen. Having tracked down prey of the necessary species,
wasp does not wait, while it will come closely to it. Insect puts all forces
to one successful throw. In case of successful throw the wasp grasps the tadpole
in two forward pairs of legs, and lays egg in its body. Then it lets it off,
and slowly emerges to the surface of water.
The tadpole in which wasp had injected egg, practically does not differ in rate
of growth from neighbours. The matter is that the larva of wasp develops very
long and slowly, therefore its presence does not lie down excessive loading
to the organism of amphibian. Moving in blood vessels, the larva of the wasp
is fixed in bladder of the host animal. The tadpole continues the life, and
in necessary time turns to frog. All this time in its bladder larva of the wasp
sits, gradually growing up. At last, when the frog becomes adult (at one-year-old
age, and even later), larva finishes the development, and begins metamorphosis.
The young wasp abandons body of the frog through cloaca. Because traumas, put
to the frog at this moment, are minimal, the frog survives in most cases. And
wasps flying above pond or river bank are not so terrible to it.
The adult wasp lives not for long time, as against its larva: only 2-3 months.
But for this time it has time to lay in tadpoles about 250-300 eggs: 2-3 eggs
per day.
Civil
Wasp Cuckoo (Neostilbum cryptica)
Order: Wasps (Hymenoptera)
Family: Cuckoo Wasps (Chrysididae)
Habitat: Forested areas of New Zealand, nests of Neovespula wasps.

Picture by Biolog
Within the colonies of wasps that inhabit New Zealand forests,
various parasitic forms exist which plague them to some extent, one example
is the New Zealand Wasp-Cuckoo.
This wasp is rather typical of the group in shape and size, with a body about
1.3 centimetres long, but it differs in colour, being a less prominent, dull
brown with no metallic sheen. This coloration is linked to its habits; it forms
a parasite of the colonies of Neovespula
wasps, so it must remain inconspicuous to its host. Body surface is grainy.
This wasp is diurnal and always solitary, feeding on nectar from various flowers
that bloom in the forest during the hotter months. To reproduce, they invade
the Neovespula wasp colonies, where they lay eggs individually in nest combs,
next to the growing larva, Wasp-cuckoo may lay multiple batches of eggs. When
the cuckoo larva hatches, it will consume the already developing larva, and
afterward be fed and protected by the Neovespula colony. This wasp has developed
a pheromone that mimics the scent of the Neovespula wasps, in order to not be
disturbed. Lifespan is about 1 year.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Civil
Wasp (Neovespula communis)
Order: Hymenopterans (Hymenoptera)
Family: True Wasps (Vespidae)
Habitat: Forested areas of New Zealand, nesting in large trees.
Some species of insect have been introduced widely across the globe, either
by accident or intent, and one of these was the German Wasp (Vespula germanica),
which made its way to many parts of the world, including New Zealand. Here it
has produced an unusual specialized form, the communal species Civil and Martial
Wasps.
These are a pair of specialized species which live communally in vast colonies
up to a million strong. Their nests are large enough to take up most of the
crown of a tree, so heavy that trees sometimes break under their weight. Nest
is built from a mixture of plant fibres and clay, reaching a hard consistency
unlike the paper nests of its ancestor.
Civil wasps are of a similar shape and size to their ancestor, about 1.3cm long,
queens may reach up to 5 centimeters long when mature. They have the same warning
colours, bands of yellow and black, as their ancestor. Fully grown queens become
larger and distended, with a bloated abdomen and no wings, having lost her wings
after her mating flight. Only the younger females retain wings and functioning
legs, until they potentially become queens. Workers have relatively large mouthparts
to chew material to build the nest, and long antenna, which they use these to
stimulate symbiotic plant louse species into producing honeydew.
This species of wasp performs various different duties to do with the communal
life of the nest. It produces and applies construction material to build the
nest, it gathers honeydew from symbiotic plant
louse as sustenance for the nest, and it cares for and cleans the larvae
and pupae, also eradicating brood parasites.
The workers will regurgitate the liquid contents of their stomach in order to
feed the queen and developing larvae. This species of wasp is less inclined
to sting than the martial wasp. Civil wasps do not feed themselves, but are
fed by the Martial Wasp, which regurgitate food for them. This species generally
outnumbers the Martial Wasp in the nest, and queens are more fertile.
The breeding behavior of this wasp is not typical of its ancestor, nests are
almost permanent as opposed to seasonal, and there is more than one queen. Individual
queens succeed each other so that the colony will last many years. Nests reach
up to a ton in weight and may eventually cause the tree to break. Queens mate
with multiple males and breed all year round, though highest fecundity occurs
in summer and spring. In areas of warmer climate, nests may be more successful
and have higher numbers of queens. All species within the colony breed communally,
and the young are attended to by the workers, which regurgitate food for them
that they in turn were brought by the Martial Wasp. The colony is entirely diurnal,
becoming inactive at night. Civil Wasps live longer than their ancestors, as
their nest is permanent; queens may live for up to 3 years.
Martial Wasp
(Neovespula atrox) is a closely related species of wasp, which performs
the more vigorous and violent tasks related to the communal nest. It hunts for
slow moving insects and caterpillars, as well as gathering oily seeds and sweet
berries; these are chewed up and digested, to be regurgitated as a liquid to
feed to the Civil Wasp workers, and its own larvae. They also defend the nest
from predators and intruders, bearing a long sting with potent venom, when one
individual stings an intruder, it releases a pheromone that signals others in
a frenzy to gather and sting together. The Martial Wasp workers are larger than
those of the Civil Wasp, up to 3.5 centimeters long, and bear a bolder warning
color, bright orange and black bands. In order to perform their duty, they are
equipped with large, sharp mouthparts for killing, and barbed grasping forelegs
for manipulating and carrying food and prey, body is slightly more narrow than
Civil Wasp (though heavier and longer), with a constricted waist. Queens of
this species are smaller and less distended than those of the Civil Wasp, slightly
fewer individuals of this species are produced to serve the nest, overall. Lifespan
and development duration of this species is almost identical to that of the
Civil Wasp, queens and young are reared communally and not segregated.
This species of invertebrates was described by Timothy Morris, Adelaide, Australia, based on ideas of Anton, the forum member.
Сontravirgo ichneumon (Micrevirator contravirginus)
Order: Hymenoptera (Hymenoptera)
Family: Ichneumons (Ichneumonidae)
Habitat: Siberia. The area coincides with the area of contravirgo moths.
In human epoch ichneumons were one of prospering groups of insects. They had
safely gone through epoch of global ecological crisis – among them only some
endemic and highly specialized species had died out. But as a whole in Neocene
the specific variety of ichneumons is comparable with the situation in human
epoch.
Intensive speciation of host insects (in reality each species of insects is
the host of any ichneumon) had caused the occurrence of parasites at which
life cycle was coordinated with host development. Some kinds of moths of bears
family, contravirgo and antivirgo, live in Siberia. These insects are remarkable
by the feature of breeding; at them individuals of one gender reach sexual
maturity being in larval stage. Poison protects them against large predators
from among vertebrates, but it does not save them from parasitic insects. One
species of ichneumons living in Eurasia, parasitizes on caterpillars of moths
of Contravirgo genus, which turn to males.
Contravirgo ichneumon is tiny insect with thin body and long legs. The length
of adult female does not exceed 1 cm, but its ovipositor is three times longer
than the body of insect. Male is considerably smaller, than female is – its
length is about 5 mm. Covers of this species of insects are colored black with
metal shine. By its shape contravirgo ichneumon differs only a few from ichneumons
of other genera, including ones known in human epoch.
The female of contravirgo ichneumon searches for caterpillars of contravirgo
moth by smell. It attacks caterpillar promptly and is not late near it for
a long time. The female makes fast sting by ovipositor to last segments of
caterpillar, almost not causing anxiety to it. Due to long ovipositor ichneumon
female avoids a prick of poisonous hairs of contravirgo caterpillar. It lays
tiny egg to friable connecting tissue of body of caterpillar. Some hours later
from egg the microscopic underdeveloped larva hatches and starts to eat caterpillar
gradually. At early stages of development it almost does not harm to the host,
and the infected caterpillar practically does not differ from healthy individuals
by rate of growth and appearance. However hormonal signals of maturing caterpillar
change the direction of development of parasite larva. At last stages of metamorphosis
of contravirgo caterpillar the development of ichneumon larva becomes more
active, and it starts to grow roughly. The growing larva of parasite moves
into testicles of contravirgo male. After that event instead of sperm liquid
in male organism the larva of ichneumon matures, eating growing tissue of testicles
of caterpillar (hence the scientific name meaning “small castrator”).
The larva of contravirgo ichneumon starts in turn to influence to the development
of host caterpillar. Secretions of ichneumon larva block the development of
smell glands at the male caterpillar of contravirgo at late stages of development.
Also colouring of infected contravirgo male is dimmer, than at healthy males.
As a result it less visible not only for contravirgo females, but also for
insectivorous birds. Obeying an instinct, contravirgo male creeps on leaves
and starts to wait for females. But it does not emit smell involving females,
and they are not interested in infected male. Such male exists for a long time
“in waiting mode”, imperceptible neither for females, nor for predators. In
such way the larva of contravirgo ichneumon prepares to itself an opportunity
of successful end of development. In some days a body of contravirgo male caterpillar
gradually mummyficates alive and it falls on the ground. The significant amount
of mummified bodies of such males is hidden among forest litter, and they are
not found by insectivorous animals. Inside a mummy of contravirgo male the
larva of ichneumon successfully passes metamorphosis. In spring the contravirgo
ichneumon leaves the shelter. Pairing at this species takes place on flowers,
and adult insects eat mainly nectar, licking it off from opened flowers. Approximately
in one week after fertilization in organism of the female eggs start to develop,
and it searches for young contravirgo caterpillars suitable for infection.
Cunning spider miner (Arachnomina arguta)
Order: Hymenoptera (Hymenoptera)
Family: Spider miners (Arachnominidae)
Habitat: temperate and subtropical climatic zones of Europe, Atlas Mountains;
deciduous forests and bush.
In Neocene among hymenopters various species, which larvae parasitize in other
animals, had appeared. Some species of ichneumons and solitary wasps had adapted
to parasitizing in vertebrates: mammals, birds and amphibians. Others had remained
“faithful” to former food source – invertebrates. The special family of Neocene
hymenopters specialized in parasitism exclusively at spiders. For features
of individual development these insects are called “spider miners”. These insects
are the specialized branch of ichneumons.
These insects are extremely tiny – the length of largest tropical species hardly
exceeds 5 millimeters, but usually they are twice lesser. Spider miners are
similar externally to small fruit flies, but their thick abdomen is placed
on clearly visible pedicle as at wasps and is very mobile. The ovipositor of
female differs in durability and can pierce covers of body of various insects.
On narrow head of spider miners there are big eyes consisting of set of facets,
and short plumose antennae. Jaws of spider miners are gnawing, and the lower
lip is mobile and covered by hairs on the tip. Wings of these insects are short,
rounded, covered by hairs on edge.
Males of spider miners are smaller, than females. Adult insects of this group
are long-term and eat flower nectar. They are fed on liquid food with the help
of lower lip, dipping its tip into liquid. In spring spider miners gather on
trees and lick the tree sap flowing down from cracks in a bark.
Ichneumons lay eggs directly in animals become food of their larvae. At spider
miners this process proceeds in two stages. First the fertilized females of
spider miners search for web of spider of suitable species, and constantly
“are on duty” near it. As patiently, as spiders, they wait, while prey will
get in web. When any insect appears in web, the female of spider miner rushes
to it, trying to get to it before spider, lays some eggs to its body and flies
at once. The spider seizes prey, bites it and injects enzymes diluting contents
of exoskeleton of prey. Digestive enzyme dissolves tissues and organs of prey,
but has no influence on eggs of spider miners. And at the second stage of infection
the spider soaks up in common with dissolved tissues of prey eggs of spider
miners. They are tiny, but covered with dense environment. Enzymes of spider
make an environment of eggs more permeable and stimulate development of the
larva. The larva of spider miner is very small. It penetrates into the wall
of intestines of spider and moves to its body cavity. Here larva eats tissues
of spider and develops. At this time it additionally pedogenetically breeds.
During the term of development one larva gives rise to several ones – in body
of one large spider it can develop in total up to ten larvae.
The significant part of term of development larva harms to spider a little,
and it continues normal life. At them differentiation of organs and tissues
proceeds, but they grow very little – they increased in length approximately
three times in comparison with the initial size. Shortly before the ending
of development larvae start to influence stronger to the life of spider in
which they parasitize – it is a part of preparation to their metamorphosis.
With the help of chemical substances they change behaviour of spider and force
it to bury itself alive in literal sense. Under influence of substances secreting
by larvae of spider miner the spider stops usual way of life. It leaves web,
finds shelter in forest and makes in it the similarity of cocoon for itself.
Soon it perishes here, because larvae begin to grow and eat its tissues. In
its mummificated body larvae finish development. They quickly devour soft tissues
of prey, grow intensively and soon undergo metamorphosis inside the exoskeleton
of spider.
At spider miners fertilized females winter. At species living in the north
of area of family, pupae winter right in shelter which is made by spider before
its death.
Cunning spider miner belongs to widespread European species and parasitizes
in the majority of species of European spiders except for wolf spiders (Lycosidae)
and other species not making web for prey catching. Females of this species
differ in grey colouring of abdomen; head and thorax are black.
The idea about existence of this group of animals is proposed by Nem, forum member.
Revolutionary ant (Formica comandante)
Order: Hymenoptera (Hymenoptera)
Family: Ants (Formicidae)
Habitat: Central and East Europe, deciduous and coniferous forests.
Between ants of different species, as a rule, there are antagonistic relations.
In rare cases ants of various species can enter relations of symbiosis, for
example, recruit ant (Recrutoformica
latrunculus). In human epoch so-called
slave-making ants were rather usual: these species had no their own caste of
workers, and for updating the number of working individuals in ant hill they
simply grasped pupae of other species of ants, from which new working individuals
appeared.
Such tactics brings to slave-making ants the great benefit, therefore species
leading such way of life are kept in Neocene. But constant “military actions”
between different species of ants resulted in formation of new defensive tactics
during the evolution process. One of species of ants, which ancestors had been
exposed to attacks of aggressive neighbours, had developed most interesting
tactics of protection, because of which it is named revolutionary ant.
Appearance of revolutionary ant is quite usual for these insects. The size
of adult fertile female reaches 14 mm; working individuals are much smaller
– up to 9 mm. This species has one unusual caste of large workers which are
only a little bit smaller, than “queen” – about 12 mm long.
These insects are colored reddish-brown color with white “mask” of short hairs
on forward part of head.
Colonies of revolutionary ant lead life standard for ants of Formica genus.
They build ant hills about 30 cm high around of mouldering stubs, and penetrate
stub through with their tunnels, transforming it to well fortified “palace”.
Under roots of stub there is a large chamber where some fertile foundress females
live. Working individuals drag various small insects to ant hill, clearing
a site of forest from pests.
In posterity of revolutionary ant it is possible to see clear difference of
larvae and pupae into castes by size. Working individuals protect the posterity
from predators, but are not always able to resist to impact of slave-making
ants, which steal many pupae of this species for updating the number of “slaves”
in their own ant hills. In this situation that property, for which revolutionary
ant has received the loud name, is expressing.
Slave-making ants steal pupae of this species of ants, and pupae, from which
large working ants develop, frequently get to their ant hills. But it becomes
the original “biological bomb” for a colony of slave-making ants. Large working
individuals of revolutionary ant have a special feature: they are females turning
to parthenogenetic “queens”. Slave-making ants do not perceive these individuals
as threat, because they have a smell peculiar to all members of colony. Being
in safety, parthenogenetic “queens” of revolutionary ant at good feeding (if
the colony with captured ants prospers) start to lay eggs actively; working
ants care of them as of eggs of slave-making species. In this case number of
individuals of revolutionary ant in mixed colony gradually grows and soon exceeds
number of the former oppressor. Actually, revolutionary ant simply biologically
supersedes the slave-making ant from its own ant hill, and gradually forms
a normal colony of this species. In it winged females, which normally couple
with males of this species, develop and settle.
Hawaiian
shell ant (Cochleoformica ostracophila)
Order: Hymenopters (Hymenoptera)
Family: Ants (Formicidae)
Habitat: Hawaii, rainforests.

Picture by Biolog
During the human colonization of Hawaiian Islands intentionally
or casually the set of species of animals had been imported there. When large
species of vertebrates (pigs, deer, domestic cattle, mongooses) had been introduced
purposefully, small ones, especially insects, mostly got to islands casually,
as “unbidden visitors”: in the ground with exotic plants, or with any cargoes.
So the set of species of insects had settled at Hawaii and among them more than
40 species of ants has appeared there. Being mainly natives of tropical countries,
not all these species had gone through the ice age, but survived ones had received
a prize – an opportunity of settling of islands. At these islands evolution
had caused set of unique species of ants distinguishing by interesting features
of habit of life.
Among tens of ant species inhabiting Hawaii, shell ants belong to smallest ones.
The length of working ant at this species is about 5 millimeters, and only queen
reaches length about one centimeter. These ants are colored rather dim: at them
there is brown body with metal shine and only at working individuals (“combining”
this occupation with the speciality of “soldiers”) head is colored bright orange
with black mandibles. This colouring is warning: sting of this species of ants
is poisonous, despite of their tiny size. The sting of ten adult ants of this
species may kill pigeon-sized birds, and the solitary ant of this species can
kill the beetle or the soldier ant of larger species.
Poisoness of this species of ants is the defensive weapon, rather than hunting
adaptation. Shell ants eat small insects, gathering them on the ground, and
seldom keep in big groups, gathering food. Such food predilections are partly
connected to habit of life of this species: their dwellings are arranged so,
that large catch simply does not place in them.
This ant is connected in life with some local birds: most frequently shell ant
arranges nests under “dining rooms” of Hawaiian hookbill – the local bird. It
lodges in shells of the snails had eaten by bird, therefore many features of
its way of life are dictated by opportunities of its dwelling. The colony of
shell ants occupies some empty shells of snails. One of these shells, the largest
one, is occupied with the queen-foundress. Queen chooses this shell once per
life, during the breeding flight, and never leaves it more. Queen sits in the
most distant curl of shell, surrounded with set of workers which carry off the
eggs laid by it to “kindergartens” located in next shell, and bring to it food
– fresh insects. Workers preserve their queen, clear her body, remove dust,
and every time are ready rush to its protection if somebody disturbs “imperial”
shell. The majority of workers lives in the next bowls, in the same place there
are warehouses of food.
About two times per one year at shell ants breeding flight takes place: one
month prior to this event female starts to lay large eggs, and workers, obeying
her chemical signals, rear from them the generation of future “kings” and “queens”
– fertile males and females. For this purpose they clear one of best shells
in vicinities of colony, and care of larvae very attentively. Larvae, from which
fertile individuals of new generation will develop, receive the increased feeding.
If they hatch before time of the common fly of young insects from the next colonies,
they stay in shelter shell, and working ants continue to care of them. But usually
different colonies synchronize breeding fly, and young insects, founders of
new colonies, direct to breeding flight simultaneously.
After pairing which occurs in air, before the fertilized female there is a difficult
task: it must find empty shells of snails which collect under “dining rooms”
of hookbills. Sometimes the instinct “misfires”: the insect finds and occupies
a single shell of snail had perished from any casual reasons. Then growth of
colony slows down: it is limited only to this shell, and the place for expansion
of ant settlement is absent. Such colonies quickly become victims of ants of
other species, or perish from casual reasons.
The fertilized female gets into depth of shell and there lays the first portion
of eggs – about ten ones largest of ever laid by it. From them larvae hatch,
and the female any time feeds them. They receive partly cut down diet though
turn to normal working individuals: development follows due to stocks of nutrients
in egg. After they develop, queen female leaves “household chores”, and starts
to lay eggs only.
The colony exists until the queen-foundress is alive – till three years.
Toka
Ahi Tahu (Antipodopyra invicta)
Order: Wasps, bees, ants (Hymenoptera)
Family: Ants (Formicidae)
Habitat: Many different vegetated habitats across the North island and the northern
part of South Island of New Zealand. Prefers forests and disturbed areas, absent
from swampy areas and cold mountainous areas.
The necessity of trade and commerce between islands and the mainland commonly
introduced accidental stow-aways, during the Holocene occupation by man. Plants
and produce exported to New Zealand would sometimes contains insects, worms
or spiders that went on to become invasive pests. One of these was the notorious
Fire Ant (Solenopsis invicta), which quickly came to dominate and out-compete
various forms of native ant.
The most common descendant of these Fire Ants is the Toka Ahi Tahu (meaning
literally “burning ant” in Maori). Their appearance is very similar to their
ancestor, but in general they attain slightly larger sizes on average. Worker
ants are polymorphic, with minor ones measuring about 3 millimetres, and major
ones up to 7 millimetres. The smaller workers do not forage, and instead help
with brood-tending duties. Major workers mature later and are able to lift larger
loads than the minor ones, their head are larger and wider, they also serve
as soldiers. A queen may be up to 2.9 centimetres long, and has a much larger,
darker coloured abdomen. Coloration of workers is generally a reddish brown,
the head being darker, and having a yellowish reflective sheen. Males are about
1.5 centimetres long and coloured much darker than the other castes, almost
black.
The nest contains a central mound above ground, that may be up to 60 centimetres
tall and 50 centimetres across, smaller mounds are located within a radius of
about 30 feet. Tunnels penetrate from the centre outward and down, linking the
mounds to the surface, and the tunnels are linked to subterranean chambers.
Parts protruding above the ground are oriented so that they only bear the full
heat of the sun in early morning and just before sunset. Tunnels and chambers
penetrate a maximum of 1 meter below ground. These ants exhibit hygienic behaviour
where refuse, dead ants and individuals infected by fungus are removed and deposited
away from the nest. If the immediate area floods, the colony will form a dense
mass or “raft” with workers on the outside and the queen in the centre, this
mass will float until the waters subside. Outermost tunnels of the colony lead
out into the main areas where workers forage, and they communicate in order
to cooperate in foraging, using pheromones. Members of one colony identify each
other by a common odour that is specific to that colony. Each worker is capable
of biting, and administering a painful sting to intruders or would-be predators.
Groups of ants may swarm over an intruder, biting and stinging, stings are a
very painful burning sensation.
These ants forage for dead insects and carrion, seeds, and sweet liquid substances
like honey, nectar, or the honeydew produced by certain insects. Food is shared
within the colony, and individual workers can regurgitate liquid food in order
to share with each other. They also stockpile specific dry food such as pieces
of dead insects, and seeds, in order to provide food in the future. Workers
tending the brood will regurgitate food to the mouths of the larvae, and each
larva must be feed very frequently.
The nuptial flight begins between late spring and early summer (November to
early January), the males emerge in the late afternoon, and the virgin queens
emerge a little while after. Males fly higher than the virgin queens, and these
queens will usually have flown almost 2 kilometres by the time they land after
mating. Mating occurs on the wing, and the males find their mates via pheremone
signals, a colony may be founded by one fertilized queen, or by up to 3. The
queen will have laid about 15 eggs within 24 hours of mating, within a week
after, she will have laid up to 140 eggs. Larvae hatch after about one week,
and go through 4 instars, within 7 to 10 days they pupate, pupa stage lasts
up to 16 days. Once the first generation of workers mature, they begin constructing
the nest, within 6 months, the colony will number several thousand. Within 2
years, the colony contains over 25 thousand, and doubles within the next year.
A mature colony can number as many as 300,000, a mature queen will lay as many
as 1500 eggs per day, which all hatch into workers. Each worker may live as
long as 100 days, a queen may live up to 7 years. Predators include many other
kinds of carnivorous insect (especially wasps) as well as insectivorous
birds and small mammals.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Recruit
ant (Recrutoformica latrunculus)
Order: Hymenopters (Hymenoptera)
Family: Ants (Formicidae)
Habitat: moderate and subtropical areas of Europe – from Atlantic coast up
to Three-Rivers-Land steppes; woods and bushes.
Ants are one of most typical groups of forest insects. Due to their activity
some plants settle, and number of pest insects restrains in reasonable limits.
Among ants there are no solitary species, though colonies of some species of
Holocene epoch could include some tens individuals. In colonies of ants more
or less expressed division to castes exists. Relations of ants of different
species show the variety though more often they may be reduced to interspecific
antagonism. Among ants of Holocene epoch there were species having “slaves”
were known: they took larvae of other species of ants from which then individuals
carrying out functions of workers in colonies of the aggressor ant grew. In
Neocene one species of ants had developed this form of relations with other
species in other direction – it had turned to the original “mercenary” living
in colonies of other ants. This feature has determined its name: the recruit
ant.
This insect has only two castes: fertile queens living in colonies of other
species of ants, and furious “soldiers”, which replace own “soldiers” of host
species and protect its colony on equal terming of high-grade inhabitants.
The shape of representatives of these castes considerably differs.
During the top expression of breeding function the queen resembles the fertile
female of termites. At this time it reaches the length of 35 – 40 millimeters,
from which only about 10 mm falls to head and thorax, and strongly swollen
abdomen makes other lenght. The adult female of recruit ant is completely helpless
and can not move independently. It lives in most protected bottom part of ant
hill, in special chamber. It is fed and cleaned by working individuals of host
species, and all cares of larvae of recruit ant are completely their duty.
Such way of life transforms the female to true “factory” of egg producing.
In summer when all ants are most active, the recruit ant queen lays one egg
each minute. Working individuals of host species immediately carry eggs away
and look after them the same way as after eggs of their own species.
The most part of summer from eggs larvae hatch, turning to individuals of other
caste – furious “soldiers”. “Soldiers” of recruit ant are absolutely not similar
to queen. These ants are easy for detecting in colonies of other species: recruit
ants differ in black colouring with metal shine, and white mandibles. The length
of these insects reaches 20 – 25 millimeters: these ones belong to the number
of largest ants of Eurasia. They concede in size only to some tropical species.
But this feature also imposes the certain restriction to opportunities of existence
of recruit ant. It can live only in colonies of ants of size comparable to
them. Smaller species of ants will have not enough food resources to support
giant “soldiers”, and also large “soldiers” simply can not move normally in
too narrow passages. Recruit ant can settle in colonies of various species
of ants, including ones having their own “soldiers”. The colony protected by
recruit ants develops more successfully, rather than other colonies of the
same species of host ants. Sting of “soldier” of recruit ant is very painful:
in addition the insect injects in wound the poisonous liquid containing ant
acid. Efficiency of protection of nest by recruit ants is very great: mouse-sized
mammal perishes from ten stings of such ants. And joint attack of recruit ant
“soldiers” can turn into flight even large mammal.
In second half of summer queen of recruit ant lays eggs from which winged individuals
taking part in breeding develop. These insects are similar to winged ants of
other species, but appreciable metal shine of body covers, and also larger
mandibles distinguish them. They fly off from populated ant hills of various
species of ants almost simultaneously, pair quickly, and males perish soon
after that. Females of recruit ant must carry out the important mission just
after pairing: to find the colony of other species not populated with congeners,
and to settle in it. The female of recruit ant produces special pheromone which
reduces aggression of ants of potential host species. Having found a colony,
the female of recruit ant defines by smell, whether individuals of its species
are present in colony. If the smell of congeners is weak, or it is not present
absolutely, the female enters into ant hill, surrounding itself by pheromone
smell. It searches for the chamber suitable by size, and settles in it. First
time young female of recruit ant is very cautious: it does not lay eggs, and
emits pheromones actively. Gradually it starts to smell similarly to environmental
ants, and moves in ant hill galleries more freely. When ants of host species
“recognize” the female of recruit ant and start to feed it under its requirement,
it settles in chosen chamber and starts to produce eggs. After several weeks
of intensive feeding its abdomen hypertrophies, and female becomes similar
to “queen” of termites.
If the smell of recruit ants is present in ant hill chosen by the female, but
is rather weak (that means in colony there is a little number of ants of this
species), the female also settles in such ant hill. The large colony of any
host ants may feed up to five recruit ant females, using their “soldiers”.
Life expectancy of the female of this species is about three years.
The idea about existence of this species of insects was proposed by Momus, the forum member.
Lantern ant, twilight lanternomyrma (Lanternomyrma
crepusculina)
Order: Hymenoptera (Hymenoptera)
Family: Ants (Formicidae)
Habitat: rainforests of Meganesia; the symbiote of lamplighter
bird.
In tropical forest live beings receive the set of opportunities for evolution
– abundance of various food sources and habitats. But the inhabiting in such
ecosystem has an underside – strict competition. As a result various species
of live organisms during the evolution develop the oddest and keen life strategies
permitting to use effectively an opportunity given by nature and to avoid
unnecessary competition. One of effective forms of survival is symbiosis.
One of mysterious and secretive inhabitants of rainforests of North Meganesia
is lamplighter bird, the nocturnal species of bowerbirds. This bird builds
for courtship displays simple constructions – heaps of pieces of rotten wood.
But on them special pileate fungi
of Mycolanterna genus grow; these ones
have ability to glow. In their illusive light male of this bird arranges
courtship
displays.
Male of lamplighter bird constantly renews the construction, dragging on
it new pieces of rotten wood, which it gathers in forest. And such activity
of
bird is very favorable for constant settler of these constructions – small
ant named twilight lanternomyrma, or lantern ant.
Twilight lanternomyrma is a species of ants a little distinguishing externally
from the majority of these insects. It is an ant of red-brown colouring with
rather large head and mandibles extended forward and forming similarity of
tweezers. Working individuals of twilight lanternomyrma are no more than
4 millimeters long, and the adult “queen” female grows up to 8 – 9 mm. In
colony
of twilight lanternomyrma there are some breeding “queens”.
Life of colony begins from the important event – fertilized winged female
finds a heap of pieces of rotten wood suitable to life which the male of
lamplighter
bird started to gather. If such opportunity is not present, the female can
lodge in already existing, but weak colony which had already rendered habitable
construction of lamplighter bird. First days the female leads a secretive
way of life, but gradually gets smell characteristic for colony, and working
individuals
accept her.
The colony of twilight lanternomyrma leads nocturnal habit of life. Ants
of this species turn more active in twilight, and hide in nest soon after
dawn.
For night these insects have time to gather enough forage for feeding of
colony. They should not care of repairing and expansion of nest - all work
on delivery
of “building material” is incurred by lamplighter bird, certainly, not understanding
its role in life of this ant.
Nevertheless lamplighter bird receives the certain benefit from presence
of such symbiote. When parasitic insects start to annoy bird male vastly,
it simply
digs a hole on the construction, and lays in it. Ants creep into bird’s plumage
and carefully clear it of parasites. Besides stings of ants cause in bird
the behavioural reaction similar to euphoria. And ants receive food – parasitic
insects and mites.
Male of lamplighter bird would not achieve only by its activity even a half
of success in cultivation of glowing mushrooms without these ants. The second
species of live organisms with which these insects had entered symbiosis
is the glowing mushroom settling on heaps of lamplighter bird. Working individuals
of lanternomyrma create conditions for prosperity of these mushrooms – they
destroy other species of fungi settling on constructions of lamplighter bird.
The saliva of ants contains the substances overwhelming growth of some species
of fungi, particularly mold ones. Besides working individuals of lanternomyrma
creep on fruit bodies of mushroom and exterminate insects which damage them.
Certainly, all this activity is selfish – ants eat a part of mycelium. The
mycelium of glowing mushroom forms original galls in places of stings of
these
ants, and ants from time to time gather a rich “harvest” which serves as
addition to usual food consisting of insects and other invertebrates.
Flight of lanternomyrma also takes place in twilight. Males of this species
differ in good sense of smell – they can feel the presence of the female
for some tens meters, that is too good result for tiny insects. Pairing occurs
in air, and then the female at once flies to search the suitable place for
future colony. Female is guided by sense of smell in choice of settling place
– it searches for mycelium of glowing mushrooms by its smell. Frequently
females
settle simply on rotten tree trunks decomposing by mushrooms. But such colonies
live not for long and degrade after 1 – 2 years.
Gall
ant (Nanocamponotus symbioticus)
Order: Hymenopters (Hymenoptera)
Family: Ants (Formicidae)
Habitat: forests at the southern slopes of the Alpes, Balkan and Apennines;
symbiosis with gall-forming insects.
In human epoch ants almost have not suffered damage, except for species lived
in tropical forests undergone to large-scale cutting down. After human disappearance
evolution of ants had proceeded almost in former rate. One of directions of
evolution of ants is the symbiosis with various species of plants. In this case
plants form special cavities in roots and stalks, which are inhabited by ants
saving the plant from herbivorous insects. But at the south of Europe, in dry
mountain forests of the Mediterranean area, evolution of ants had gone in opposite
direction: these insects have entered symbiosis with the parasitic insect which
due to fodder plant supplies them with shelter and food in exchange for protection.
The ant chosen such way of evolution is the descendant of carpenter ants (genus
Camponotus). These ants made their dwellings in old dry stubs of trees, but
symbiote insect has offered them available dwelling in alive tree, and the ant
has turned to its ally. This ant inhabits the galls formed on branches of trees
populated by larvae of laurel and oak
gallfly (Oncofaber myrmecophila), and hence the ant is named gall ant.
Colonies of gall ant are rather small – they total some hundreds of adult insects.
It is a tiny species of ants: length of adult ergate is no more than 3 mm, and
flying “queen” females may grow up to 5 mm. Colouring of gall ant is black with
white tips of mandibles and longitudinal stroke of brown color on thoracal segments.
The ancestral species of ants had original caste of large-headed “gatekeepers”
which closed entrances to the nest by massive heads. At gall ant they are absent,
but there is a plenty of the soldier ants. Ergates at this species are not specialized
– gall ant lives in ready cavities, and its building activity is reduced to
minimum. Ergates only expand passageways between cavities inside galls or gnaw
new holes. Also they gather food – small soft-bodied insects. This kind of food
is rare in dry forests of Southern Europe, and here gall ants almost don’t have
competitors.
Activity of larvae of laurel and oak gallfly forms complex galls with set of
cavities, representing almost ready shelters for ants of this species. Moreover
gallfly supplies ants with a significant amount of food. Secretions of larvae
change biochemical processes in plant tissues, and due to it internal walls
of galls supply ants with formations rich in proteins and starch – it is so-called
“ant bread”.
Ergates of ants look after the larvae living in friable parenchyma on internal
walls of galls. They remove dung of larvae and cautiously lick them, clearing
from pollution. If larvae are placed too densely, ants move a part of larvae
to new places. In the rest the biology of gall ant differs only a little from
biology of other species of ants. In the colony of this species there are some
females, capable to lay eggs. They are descendants of the first foundator female,
and they are impregnated by small wingless males hatched from her eggs. This
inbreeding provides genetic uniformity of colony that facilitates the recognizing
of “своих” and “strangers” by ants on smell. Colonies of this species exist
for rather short time – no longer than three years in succession. It is connected
to features of breeding of gallflies – they live in cavity of the gall for approximately
the same time, and then pass metamorphosis or perish by virtue of natural ageing.
It is inevitable payment for advantages of symbiosis, but for this time ants
have time to found some more colonies on tree, simply having dragged there the
fertilized females of gallfly. During the existence of colony of ants inside
it some generations of females and males of gallflies pass metamorphosis, and
ant ergates care of them.
Prospering due to symbiosis with gallflies, ants incur care on their settling
– females of laurel and oak gallflies had lost ability to fly. Flight at gall
ant begins in spring, when snows in the Alpes thaw, rivers become deep for a
short time and trees grow intensively. Young “queen” makes courtship flight,
having taken in mandibles the flightless female of gallfly, and one or two more
individuals can cling to its legs. During the courtship flight winged males
by smell define, whether the female has such “wedding gift”, necessary for the
foundation of the colony. If the female does not carry any gallfly, male simply
does not pay to attention her, despite of own smell of the female. In some cases
females from weak and growing old colonies where shortage of gallfly females
for courtship flight is felt, can be engaged in “robbery”, attacking on the
females carrying two and more gallflies. In this case they have a real opportunity
to be fertilized and to found a new colony. But such cases are rare – the female
from a viable colony usually is stronger and easily seeks safety in flight from
attacking individual from weaker colony.
The critical moment in life of new colony is its foundation. After courtship
flight the fertilized ant female chooses a branch on which the new colony will
be founded. She cautiously places on branch the brought gallfly females and
leaves them in order to give them time to lay eggs. After that the gallfly female
dies, and the ant female carefully protects the branch, where gallfly had placed
eggs, from other insects, including from its own congeners. At this time it
lives in casual shelter and is very vulnerable for insectivorous animals. When
the first gall is formed, ant female gnaws in it the entrance and creeps into
the internal cavity. There it lays the first eggs and feeds on “ant bread”.
The first small ergates start to care of her and expand the shelter, finding
new cavities and connecting them with convenient tunnels. Also they supply a
colony with food including small insects.
Honey-bearing
Plant-louse (Vespocacopsylla meliproductor)
Order: True bugs (Hemiptera)
Family: Jumping plant lice (Psyllidae)
Habitat: forested areas of New Zealand.
Various invasive species came to New Zealand during colonial times, both plant
and animal. The apple tree, so useful to man as a food crop, was dispersed widely
across the globe, and it has left various descendants, though these produce
smaller, less heavy fruit than their ancestor.
One inhabitant of these trees is the Honey Bearing Plant Louse, a descendant
of the genus Cacopsylla. This louse has become larger and more sedentary than
its ancestor, reaching about 1 centimeter long. It feeds entirely on the sap
of the native apple trees, and generally lives in large congregations that are
not inclined to move around unless disturbed. When retreating from a predator,
they are able to both hop and fly, but are less inclined to do so than their
ancestor. This species is also a partial symbiote of the Neovespula genus of
wasps, it produces honeydew specifically when stimulated by the Civil
wasp, but also produces honeydew as a by-product of its feeding, which may
also be gathered by the wasp. Outwardly, apart from its larger size, it generally
resembles its ancestor, being a small, vaguely cicada-shaped insect, though
in this case the louse is bright green instead of brown.
Eggs are laid on the surface of leaves, and galls are formed around them, in
which the young develop, this generally does not harm the tree, as they do not
lay eggs in large enough numbers to cause the leaves to fall off. Egg laying
begins during the spring when new leaf buds are forming, and may take several
weeks. Larvae are flat and oval shaped, wingless.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Water-bearing
plant louse (Toxiaphis aquifera)
Order: Homopters (Homoptera)
Family: Plant lice (Aphidiidae)
Habitat: deserts of Mediterranean Lowland, plants of leafless
paunchstem.

Picture by Biolog
Plant lice are tiny and delicate insects living in huge amount
on whole Earth. As against the majority of insects, the body of plant lice is
covered with very thin cuticle. It makes plant lice very vulnerable for any
predators who will want to attack them. But protection of these insects is not
in the reliable reservation at all. Fast rate of development and the highest
fertility save them. Plant lice enter symbiosis with other animals, for example,
with ants, and use their protection in exchange for drops of sugary secretions.
Ability to increase number quickly, ease of settling and high adaptive ability
easily had permitted plant lice to go through changes in biosphere at the boundary
of Holocene and Neocene. Their specific variety had not changed almost, and
in Neocene increased even more. Among plant lice the specialized species distinguished
by interesting adaptations to inhabitancy had appeared.
It is possible to meet plant lice everywhere. Even at the border of severe saline
desert of former Mediterranean these insects find house and food. The most important
condition for life of plant lice is the presence of fodder plant. In places,
where small streams flow in spring, thick not branchy trunks of leafless paunchstem
plant stick up from ground. This unpretentious plant is a food source for one
Neocene species of plant lice. Insects form white, as if powdered colonies on
tops of trunks of this plant where covers of stem still thin, and sap flows
more actively.
On surface of colony of plant lice drops of their secretions gleam. It is not
dense sugary syrup, but something more valuable in desert – almost clean water.
For this feature plant louse living on paunchstem is named water-bearing plant
louse.
This insect is the stenophagous species feeding strictly on paunchstem plants.
Paunchstem is poisonous, but this plant louse during the evolution had adapted
to neutralize its chemical protection. It even defends from enemies, accumulating
poisonous substances from plant sap in body. Except for predatory insects, water-bearing
plant louse has other enemy – the drying up sun. To escape from it, the insect
produces wax, protecting itself from drying. Colouring of body of water-bearing
plant louse is light grey, and wax gives to its body whiteness. It does not
form continuous cover, and lays on body of plant louse by friable flakes, creating
isolation from overheat.
Similarly to all species of plant lice, water-bearing plant louse is inactive
insect. It almost constantly stays immovable, having pierced by proboscis covers
of plant. From paunchstem sap the insect receives all substances necessary for
life. Secretory glands are well advanced; their tips stick up from wax coating
as two tubules. Plant lice known in human epoch entered symbiosis with ants.
They “bribed” ants, secreting sugary liquid at stimulation. Water-bearing plant
louse lives in desert, and secrets substance more valuable to inhabitants of
desert – clean water. Ants of various species live in desert, and some of them
simply can eat these plant lice. But this plant louse has entered relations
of symbiosis with other insect. Plant lice are protected by shepherd
beetle of true weevils family. It preserves plant lice, attacking other
insects appearing in colony of plant lice. The beetle receives appreciable benefit
from mutual relations with these plant lice: it drinks water secreted by them.
The paunchstem plant contains a plenty of water, but not many animals can use
it, because the plant is poisonous. Plant lice serve as original natural filter,
supplying the beetle with water.
For plant lice the phenomenon of alternation of generations is typical. Depending
on conditions these insects form various intraspecific forms – winged and wingless,
parthenogenetic and breeding with fertilization. In hot summer water-bearing
plant louse forms only parthenogenetic generations. They settle on surface of
chosen plant of paunchstem and actively breed. In autumn, when weather becomes
cooler, and at night fogs form, in colonies of water-bearing plant lice winged
males and large mobile females of sexual generation appear. Males find and fertilize
such females, and they lay hibernating eggs covered with dense shell. Using
cracks in the ground such female penetrates under ground. It creeps on the surface
of rhizome and lays eggs on buds, from which in spring new stalks will grow.
After the laying of all eggs the female perishes. In winter eggs of water-bearing
plant louse are in inactive condition. In spring the stalk of plant starts to
grow and takes eggs of plant louse out on ground surface. From them larvae,
which settle on tips of sprouts, burst. In process of stalk growth larvae creep
higher. The part of larvae turns to generation of small winged settling females.
They fly out from the plant where they were born, and search for new fodder
plants. The majority of them perishes: winds carry them away in salt desert
of former Mediterranean, or in Alps. But those few individuals succeeded to
find fodder plant start to give rise to larvae from which the colony develops
soon. For one summer season the water-bearing plant louse gives more than twenty
generations, and fertility of one female can reach fifty larvae. They quickly
grow, and in one week can rise posterity.
Sea wax scale (Thalassorthezia cereifera)
Order: Homopters (Homoptera)
Family: Scales (Ortheziidae)
Habitat: Atlantic coast of Europe; the parasite of leafless
sea asters.
Various insects of homopters order – scales, plant lice and bark lice – are
among the most specialized parasites of plants. The parasitic habit of life
has resulted in simplification of way of life at simultaneous complication
of breeding ways. Some insects of this group had lost mobility at adult stage
of development, and live, having constantly stuck to fodder plant. Frequently
the parasite is highly specialized species, and evolution of host plant results
in occurrence of surprising adaptations in its parasite.
In salt marshes of littoral zone of North Europe leafless sea asters grow.
These ones are flowering plants enduring regular flooding by tidal waves. This
species of plants is a fodder plant for one species of parasitic insects which
had also adapted to existence in tidal zone. It is the sea wax scale living
on succulent stalks of leafless sea aster. Similarly to all relatives, this
is inactive insect almost constantly sitting on plant and sucking its sap with
the help of long proboscis.
Males and females of sea wax scale strongly differ by appearance. The female
of this species is wingless one about 1 centimeter long. At it very short legs
are kept. Because this insect leads motionless way of life, legs do not serve
for walking any more. Two forward pairs of legs are reduced and weak. The back
pair of legs is longer, than the others; they are mobile, with wide paws. These
legs serve for the vital process: using them insect smears over the body wax
secreted by special glands. Around of body of the female the scute of wax serving
for protection against sea water develops. This scute protects not only the
female, but also its posterity, forming brooding chamber in back part of body
of the female. For fertilization of the female the male creeps up under her
scute.
The male of sea wax scale is tiny – about 2 – 3 mm long. At him wings are advanced,
and it flies from one plant to another in searches of females. For the short
life males eat of nothing. They have two overall purposes in life – to fertilize
female and to settle posterity. However, females easily breed without fertilization.
Fertilizing the female, the male involuntarily takes away on itself tiny larvae
from her brooding chamber, and carries them in vicinities. Larvae also easily
settle with the help of wind and sea water. They weight so little, that do
not break a film of surface tension, and are carrying by currents to long distances
along the coast. But they survive only in places, where the fodder plant grows.
This insect receives feed from unique source – from sap of leafless sea asters.
The sea wax scale in adult condition constantly sucks sap of plant with the
help of very long proboscis three times exceeding the length of body of insect.
As sap is not balanced ideally for feeding of insects, the sea wax scale is
compelled to remove the substances received in superfluous amount from the
organism. Relatives of this insect, plant lice, secret surplus of sugars entering
to the organism, with the help of special glands. Sea wax scale emits with
their help not only sugar, but also surplus of salt: in sap of fodder plant
there is much more salt, rather than in sap of ground plants. Secretory pores
on the top side of sacciform body of the female look like two tubules. Through
them sweetish brine from time to time is secreting. Salt dries up atop of wax
coating and falls off from wax cover of insect as translucent scales. Sometimes
butterflies fly to the colony of these scales and suck liquid secreted by them.
Salt dissolved of sea water is a main problem for animals, compelled to develop
life in sea. Sea wax scale solves it very simply: insect does not suppose contact
of water with its own body. This insect produces plentiful wax coating, and
the top part of its body is covered with hairs. Due to such adaptations the
insect can endure flooding – around of its body the thin film of air protecting
the organism of insect from influence of salt and serving for breath is kept.
But air surrounding the body of insect, would not suffice for stay under water
during the inflow. However this insect has practically unlimited source of
oxygen for breath, and sea wax scale is able to use it. Stigmas on abdomen
of insect are shifted downwards and pulled together. They are protected by
special kind of scales, which compose the peaked tube. Due to this feature
the insect does not choke during inflow: the scale simply sticks tube through
thin skin of stalks of plant and respires the gases formed in parenchyma of
plant. The gas mix inside the plant is rich in oxygen formed at the photosynthesis,
and thus, the plant serves to the insect not only as “dining room”, but also
as “aqualung”.
This species can maintain stay in sea water and settle on floating branches
of leafless sea aster. Therefore sea wax scale is settled almost in all places
where the leafless sea aster grows at the Atlantic coast of Old World. This
insect does not live only at New Azora and in Iceland – these islands are too
far from the coast of Europe and Africa.
Great mantis bug (Rhinocoris gigas)
Order: Bugs (Hemiptera)
Cемейство: Assassin bugs (Reduviidae)
Habitat: temperate areas of Eurasia, from the Central Europe up to Eastern
Siberia.

Picture by Amplion, colorization by Biolog
Initial picture by Amplion |
Bugs represent one of the largest orders of insects alongside
with coleopters, lepidopters, hymenopters and dipters. Various species of bugs
live in all natural zones, down to the water surface of the tropical seas. Usually
they reach very large size in tropics and subtropics, but in Neocene separate
very large representatives of this group had evolved in the areas of Eurasia,
where seasonal climate is typical. Some of them are descendants of tropical
forms, and others descend from local species. One species of large bugs of Eurasia
is great mantis bug, the descendant of certain European assassin bugs of genus
Rhinocoris. This insect lives among grassy and bushy vegetation in deciduous
and mixed forests, especially where the forest alternates with sites of bushes
and open area, and also at the riverbanks. This species differs from other assassin
bugs in more extended and narrowed body shape. The size is a reason to regard
this bug as one of the largest species of insects of temperate latitudes: adult
males reach length 4.5 – 5 cm, and females are 5 – 6 cm long. Colouring of adult
individuals is changeable depending on inhabitancy: from ochreous up to dark
brown, but more often it is light brown or yellowish with darker spots. Larvae
of this species are darker, rather than adult individuals. Shine of chitinous
covers, characteristic for Rhinocoris genus, is absent at great mantis bug.
Tergites of abdomen have strips which may be seen on original “boards” - the
edges of abdomen rising a little above the back.
This species of bugs is carnivorous and in absolutely not fussy in food choice.
It eats almost everything that may be caught and kept. Due to the size it can
attack spiders and large grasshoppers. This species has convergent similarity
to mantids in some features of anatomy. The forward pair of legs has original
outgrowths on internal surface – spikes for prey keeping. Simple eyes at this
species are not present, but complex eyes are large and are combined from multiply
facets that provide sharp sight to this insect. Short antennae are 4-segmented,
as well as at all bugs of the present genus, and proboscis has three segments.
The sexual dimorphism is clearly expressed – the female has larger sizes and
underdeveloped wings. At this species only males can fly. Great mantis bugs
can utter sounds with the help of friction of proboscis against the oblong groove
supplied with cross thickenings and located between coxae of front legs (it
is the distinctive feature of all assassin bugs).
Great mantis bug starts food search in the evening, and hunting proceeds during
the whole night. In daytime this insect tries to find shelter or simply stays
motionless among vegetation, trying not to give out its own presence. Pairing
takes place from June to July. The female relates to males tolerantly: despite
of similarity to the mantis, cannibalism on the part of the female is not present.
The female hides clutch among plants. The clutch has no protective adaptations,
and the number of eggs in clutch reaches up to 60 ones on the average, but may
vary from 40 up to 80 ones. Within one summer the egg laying repeats up to 3
– 4 times. From eggs mobile larvae hatch. They differ from adult individuals
only in size and underdeveloped wings. They also are predators, and plant lice
usually are their first prey. Great mantis bug hibernates at the late larval
stage, and at this time does not form congestions.
Among invertebrates these bugs have rather little number of natural enemies.
They are more vulnerable at young age and then they are attacked by other predatory
insects and spiders. Clutches are frequently attacking by ichneumons. At any
age birds and small mammals eat these bugs. Protecting itself from large enemies,
this bug can put painful wounds by proboscis – its “sting” in morbidity is comparable
with hornet’s sting.
This species of insects was discovered by Amplion, the forum member.
Running
skimmer (Dolichogerris cursor)
Order: Bugs (Hemiptera)
Family: Water striders (Gerridae)
Habitat: Antarctica, stony areas with sparse grass near glacier edges.
Adaptive radiation of terrestrial representatives of water striders led to the
emergence of various kinds of predatory bugs inhabiting a variety of habitats
and using diverse tactics in search of prey.
On the surface of rocks warmed up by the summer sun one of the representatives
of terrestrial water striders hunts – the running skimmer. This species is a
small insect – the body length of an adult is about 12 mm. Its body is elongated,
cylindrical, with a spherical head and big bulging eyes. Its front pair of grasping
legs at rest is folded under its body. The second and third pairs of legs are
of equal length and are developed in equal degree. They are only slightly longer
than the body. All individuals have wings.
The main body color is gray or brownish, like the color of rocks prevailing
in a given area. The upper side of the body is red, but it is noticeable only
during flight or the threatening displaying of the insect.
Waiting for prey, this bug lies on the surface of a stone, having sprawled to
the sides its middle pair of legs. At the same time the rear pair is bent under
the body and is prepared to pounce. Sensitive hairs on the legs allow the insect
to feel the slightest tremor on the surface of the stone produced by insects
running on it. When suitable prey is close, bug catches up with it, moving by
a “gallop” and keeping its front pair of legs ready. Having overtaken its prey,
the predator inflicts a poisonous stab by its proboscis and drains it.
When attacked by a bird the insect opens its wings and shows its raised up red-colored
abdomen. A running skimmer has a durable proboscis capable of piercing the skin
of birds, and strong poison, causing a short-lived, but very painful sensation.
Within a year, this species provides only one generation. Nymphs of an early
age hibernate and begin feeding intensively after the snow melts. Due to their
constitution, they are able to withstand even freezing in ice and continue normal
development after several years of captivity in the ice.
Translated by FanboyPhilosopher.
Humped
nurse stinkbug (Pentatonutrix gibbosus)
Order: Bugs (Hemiptera)
Family: Stinkbugs (Pentatomidae)
Habitat: tropical rainforests of South America.

Picture by Amplion, colorization by Biolog
Initial picture by Amplion |
In Neocene epoch the great number of insects species lives
in equatorial areas of the Earth. During the struggle for existence they developed
the various strategies, allowing to avoid the competition: new kinds of forage,
new ways of protection, new ways of care of posterity. Among South American
bug species had appeared, which have got an original way of care of posterity;
these ones are nurse stinkbugs.
One of typical species in this genus is humped nurse stinkbug. It is a large
bug: the adult individual reaches length from 4 cm (males) up to 5 (females)
centimeters. Its body shape is characteristic for typical stinkbug: body of
insect is wide and flat, without lateral outgrowths. The shield differs only;
its structure is a characteristic attribute of genus – it is large, covers the
most part of upper side of body and has the lid shape (it is convex). Usually
it adjoins to the body densely enough, but in some cases it is capable to rise
above the back – this is promoted also by bends of body.
Colouring of humped nurse stinkbug is bright, but the pattern on the body is
rather simple: on each segment of abdomen there are black and light (from bright
red up to beige-yellow) strips. Shield is colored light with two black spots;
sometimes these spots reach the edge of shield. Prothorax has more or less T-shaped
light spot on black general background. On head (also black) there may be an
M-shaped spot. Hemelythra are black with a line of light spots along the edge.
The present species has sexual dimorphism: female is larger due to more lengthened,
pointed back end of body; its hemelythra are short and wings are reduced.
This bug lives in tropical rainforests on large trees. It consumes vegetative
food, similarly to other bugs, piercing covers of plants by proboscis. Due to
smell glands, producing unpleasantly smelling secret, bug is rather inedible.
It feeds in daytime, and at night hides in secluded places, but sometimes it
stays in feeding place, simply staying motionless and relying on warning colouring
and smell.
During the individual development this bug undergoes four moults, reaching a
stage of sexually mature imago at the 30-th – 35-th day of life. Males capable
to fly are engaged in search of the partner. When male finds the female, the
short phase of recognition follows, and then the pairing takes place, continuing
from several hours up to the whole day After pairing the female turns back to
male and begins laying eggs on its back, right under the risen shield. The egg
laying lasts for some hours; all this time the female keeps male’s back pair
of legs. Letting him off, the female lets know, that the laying is completed
and male is free.
 Picture by Amplion
Picture by Amplion
Eggs are very small; their amount may reach 90 ones. Their development is
finished for 20-th day. Being under shield, eggs are protected not only from
excessive
drying and egg-eating predators, but also from egg parasites from which other
bugs suffer in great degree. Moreover, male actively moves all the time while
it carries clutch on itself – it allows females hatching from them to settle
faster. However, it can not fly at this time – eggs are pasted not only to
the internal side of shield, but also to wings.
At the third day after hatching, right after the first moult, young bugs
leave their daddy and move to “adult life”. Having removed from under shield
the
rests of egg shells, male can go to search for the new female. At successful
coincidence of circumstances the adult bug can live till 2 years, couple
and bear posterity up to 4 – 5 times during its life. The clearly expressed
dependence
of breeding to any season at these bugs it is not observed.
The basic enemies of these bugs are the spiders, separate species of birds,
and also the certain number of large beetles. At the moment of laying bug
eggs are vulnerable for tiny ichneumons.
This species of insects was discovered by Amplion, the forum member.
Puena
Ana, Cave Fishfly (Spelaeochauliodes rex)
Order: Dobsonflies (Megaloptera)
Family: Dobsonflies (Corydalidae)
Habitat: Ephemeral insect with long life cycle, larvae live within cave waterways.
Mature adults are short lived. North and South Islands of New Zealand.
Freshwater environments usually play host to a miniature zoo of aquatic invertebrates,
often including predators pugnacious and fierce for their size. In the subterranean
cave rivers of New Zealand, one of the top aquatic predators is the Puene Ana
or Cave Fishfly.
This aquatic insect has a long life-cycle, distinguished by a larval stage of
some years, and a brief ephemeral adult stage. The larva is somewhat maggot-shaped,
up to 3 centimetres long. Abdominal segments are soft and bear tentacle-like
gills, which aid in swimming and give the insect a centipede-like appearance.
Only the head and thorax bear a hard cuticle, and 3 pairs of grasping legs,
mandibles are large and pointed for catching prey. The head bears clusters of
long filaments, by which it senses the movement of prey, eyesight is very weak
and eyes are small. Rest of the body is covered in sparse long filaments that
alert it to movement in the water. Coloration is pale, soft abdomen is whitish
and opaque, head and thorax are pale yellowish brown, darker at the extremities.
The adult is a flying insect with protruding eyes and long antennae. It has
2 pairs of large wings, wingspan is up to 4 centimetres, body length up to 2.5
centimetres. Colour of the adult is dark brown spotted with grey, wings are
clear.
Larva lives for up to 7 years in cave waterways, hunting smaller aquatic invertebrates
such as small crustaceans, snails
and other submerged insects, it will commonly consume insects from outside that
stray into cave systems and fall into the water. Prey is located with tactile
senses provided by clusters of sensory whiskers, large pointed mandibles seize
and impale the prey. Usually it will creep along the stream-bed using its forelegs,
but is also able to swim by undulating and thrashing its abdomen from side to
side. Adults live usually no longer than 10 days, larval stage is the only one
that feeds.
After up to 6 years, during the time between July and November, larva enters
the prepupal stage which lasts up to 10 weeks, it crawls around near the margins
of cave rivers. After this it enters the pupa stage (lasting about 20 days,
occurring between October and February) where it comes to resemble an adult
that lacks wings, it crawls about on rock faces and exposed mud on the banks
of cave waterways. It darkens in colour and moults into the winged adult, which
flies around inside the caves in search of a mate, this stage only lasts less
than 10 days. Pregnant females lay masses of eggs on the rocky margins of cave
rivers and streams, total number of eggs may be as many as 400. Predators include
cave dwelling amphibians and
other members of their own species.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Antiguan
giant hawk moth (Toxicomanduca longissimoproboscis)
Order: Butterflies (Lepidoptera)
Family: Hawk moths (Sphingidae)
Habitat: Great Antigua, tropical forest areas.
Butterflies and moths represent the group of insects almost exclusively adapted
to feeding on nectar. Only their primitive species, tiny moths, have kept mandibles
and are capable to eat pollen. The majority of species of butterflies and moths
eats exclusively and only liquid food with the help of long proboscis. Certainly,
among them there are the species eating tree sap, liquid fraction of dung of
large animals, fermenting rotten fruits, blood of live animals and products
of carcass decomposition. But the majority of species of butterflies and moth
all the same meets on flowers. Coevolution of flowers and lepidopters is directed
to the maximal conformity of both species to each other’s requirements and features
of structure at simultaneous disposal of any alien species not bringing advantage
to the plant and being competitors of its pollinator.
One of tops of this coevolution is represented by the union of plant and insect,
appeared in Neocene at the Great Antigua island. At the broken areas of tropical
forest where there are “windows” in solid forest canopy and the sunlight reaches
the ground, pipe-flowered
tobacco tree grows; it is a large species of Solanaceae family with treelike
stalk. Its tubular flowers reaching half-meter length, hide deeply the nectar
from the pollinators not adapted to its extraction. And only one species of
moths has ability free to feed on nectar of this plant – it is Antiguan giant
hawk moth.
This insect is one of the largest species of lepidopters in Neocene. Wingspan
reaches 20 cm at the female and 15 cm at male. In outlines of the body this
moth is similar to the majority of representatives of the family – its wings
are rather narrow and long, providing excellent flight abilities; body is streamlined.
Antiguan giant hawk moth is the excellent flyer. In flight this moth is easy
for confusing with any small bird. It is capable to hover above the flower and
to suck nectar in flight, as all hawk moths do. Colouring of front wings is
cryptic, green with brown spots forming a pattern similar to spots of moss and
lichens on tree bark. Back wings are purple-red with dark blue oculate spots.
In middle of these spots there are sites of sunlight reflecting scales. During
rest the moth keeps on bark of tree, having combined front wings atop of back
ones, and this pattern is not visible. At danger moth displays back wings, turning
front ones in sides. Thus it slightly rises on legs and vibrates by back wings.
The sunlight reflects from oculate spots and frightens the enemy, creating the
illusion of eyes of moving large animal.
This moth lives in the top part of tree crones and after the metamorphosis it
does not fly down to the ground.
The most remarkable feature of Antiguan great hawk moth is its half-meter long
proboscis – the longest one in nature. Even at larger butterflies and moths
it does not reach such length. This species is adapted exclusively to feeding
in flowers of pipe-flowered tobacco tree. Also this moth is the only pollinator
of this plant. During the flight the proboscis is turned in hard spiral and
is located between legs of insect.
Fertility of this species up to 1000 eggs. It can seem rather low for such large
insect, but at Antiguan great hawk moth there are no food competitors, because
caterpillars of this species also feed on plants of pipe-flowered tobacco tree.
The female lays eggs in flight, simply dumping them on leaves of fodder plant.
Due to sticky shell eggs at once paste to leaves and in some days from them
caterpillars burst. Shortly before pupating the caterpillar reaches length up
to 15 cm. It has bright green colouring with longitudinal black strips. Right
behind the head of caterpillar on thoracal segments there is a pair of black
oculate spots with red edge. On the back end of body there is long (up to 2
cm) orange tail thorn. At danger caterpillar takes pose of sphynx characteristic
for these insects. Very young caterpillars have monotonous green colouring.
Age change of colouring is connected to features of protection strategy of caterpillars.
As they grow, caterpillars accumulate in their bodies alkaloids of tobacco,
and to the final stage they become impregnable for predators. Young caterpillars
are much more edible, and green colouring helps them to hide from the enemy.
Union with the poisonous plant gives this insect one more benefit: it has only
few ichneumon parasites – not all species of these insects can sustain that
amount of alkaloids which is accumulated in tissues of caterpillar.
Having reached the age limit, caterpillars pupate in ground directly under fodder
plant. Pupae and imago of this species also are poisonous.
Any adaptation has its own price. Antiguan giant hawk moth in its life completely
depends on fodder plant, and the reduction of population of pipe-flowered tobacco
tree can cause fast extinction of the moth, which, in turn, will result in complete
disappearance of the plant. However, the existence of both these species so
closely interconnected to each other, indicates that environment of their habitat
differs in certain stability.
This species of insects was discovered by Anton, the forum member.
Long-nosed
hawk moth (Nasulosphinx longiproboscis)
Order: Butterflies (Lepidoptera)
Family: Hawk moths (Sphingidae)
Habitat: subtropic areas of Europe, deciduous forests.
The role of insects in life of plants is difficult for overestimating. Various
species of insects eat plants (less often it happens on the contrary) and are
pollinators and seed carriers. Frequently coevolution of plant and insect species
results in occurrence of freakish shapes and strict specialization at both sides
of this union. One example of it is a tropical plant pipe-flowered
tobacco tree (Nicotiana longissimoflora), pollinated only by one
species of moth. In Europe relations of plant and insect do not reach such
extreme degree of specialization, but among them also there are examples of
close interrelation.
On roots of trees of humid subtropical forests of the south of Europe the parasitic
plant alien rootflower (Rhizoflosculus
xenos) lives. Its stalk and leaves had turned to a circuit of tissue cords
stretching under bark of the host tree, causing formation of outgrowths on tree
trunk. From under bark flower buds sprout, which form large labiate flowers
of lengthened shape, which represent the feeding place of one species of European
insects.
Flowers of parasitic plants are pollinated by the large moth found in underbrush
of the European subtropical forests. It is long-nosed hawk moth, an insect about
5 cm long, about 8 – 9 cm in wingspan. The majority of hawk moths flies in day
time, and some of them are nocturnal. Long-nosed hawk moth belongs to diurnal
species, but its habitat is the gloomy humid underbrush. This insect never rises
even up to the lowest large branches, and spends night on bark of trees.
Long-nosed hawk moth is excellent flyer. Its body has the streamline shape,
and wings are narrow and peaked. Front wings have camouflage colouring – dark
grey with faltering longitudinal strips and strokes of black color. In rest
the moth keeps on tree bark a head upwards, having folded wings like a roof.
Their pattern thus merges with background colouring of bark. Back pair of wings,
on the contrary, is very appreciable – they are white with black spot in the
basis and cyaneous shining border. The body of insect is covered with downiness
of grey color, and legs are black. The disturbed moth emits sharply smelling
substance – in such way it frightens off predatory insects and mammals.
In connection with strict food specialization at this species the long proboscis
is developed: it is up to 15 cm long. In rest the proboscis is curtailed to
hard spiral. Long-nosed hawk moth is feeding, hanging above the flower in flight.
Strict specialization of adult insect is compensated by omnivorousity of its
larva. The caterpillar of this insect reaching length of 7 cm and lives in tree
crones. It is fed on many species of deciduous trees, eating off completely
foliage on separate branches. It obviously prefers soft foliage of maples and
horse-chestnut, but avoids foliage of laurels and ivy. The appearance of the
caterpillar is typical for hawk moths: in back part of the body there is a peaked
horn, and forward part of body grows massive. The caterpillar is colored bright
green with several slanting longitudinal strokes. Being scared, it freezes,
imitating the tortuous leaf. The bottom part of body is bright red. The disturbed
caterpillar bends head and thorax back, displaying to the aggressor red colored
areas. It is capable to emit the acrid substances causing in predator plentiful
secretion of foamy saliva.
Caterpillar winters in cracks of tree bark and in hollows, in layer of dust.
It feeds on young spring leaflets, reaches the condition optimal for metamorphosis,
and pupates at the end of spring or in the beginning of summer. In the beginning
of summer, to the moment of flowering of fodder plant, metamorphosis is over
and adult moths appear.
Conifer
silk moth (Neobombyx abietes)
Order: Butterflies (Lepidoptera)
Family: Bombycids (Bombycidae)
Habitat: coniferous forests of Eurasia.
In typical cases lepidopterans and coniferous forests are two mutually excluding
things: no matter how voracious caterpillars are, but it is easier for them
to eat the leaves of fowering plants than less nutritious and hard needles of
conifers.
This rule had been broken in Neocene: the relative warming of global climate
had created new specialized species, conifer silk moth.
This species of lepidopterans has retained all of the features of this family.
Caterpillars hatch from eggs within a week or so and immediately begin eating.
Unlike its ancestor, they are able to eat conifer needles instead of leaves,
and even transform some substances from needles to their own biological weapons:
the body of grown up caterpillar, pupa and imago has such a high concentration
of substances derived from pine needles that they can cause poisoning (though
not lethal for large vertebrates) even to such giants as shurga
and Siberian obda. And small insectivorous
animals like predatory arthropods and taiga lizards can even die from food poisoning.
Besides the inedible taste caterpillars also accumulate in their bodies substances
that initially help the conifer needles not to freeze in winter. These substances
help caterpillars and pupae to survive in Siberian winter, but the cold weather
kills about as much or even more silkworms than the number of them perishing
in stomachs of insectivorous animals.
Imago emerges from pupal cover in late spring; it can fly, unlike some species
of these insects living farther at south. Moreover, conifer silk moth has very
strong wings for 5 cm long insect – they help the insects to find a partner
and to mate it at night. The next morning, or in rare cases during the day after,
males die and females die around the time of sunset – imagoes do not eat anything,
for there it is nothing to eat for them in boreal forest: there are no flowers,
nor any other food source.
Despite their enemies and the autumn-winter frosts, the population of conifer
silk moth resembles the population of lemmings or white hares of Holocene: once
in 12-15 years the population peak of these insects takes place and extensive
territories of taiga are deforested by their caterpillars, and it is followed
by the population crash of this species. The role of predators in this case
is done by predatory insects and spiders. The total life span of this moth is
one year.
This species of insects is discovered by Bhut, the forum
member.
Translated by Bhut.
Gold-winged
metal swallowtail (Metallopapilio chrysopterus)
Order: Butterflies (Lepidoptera)
Family: Swallowtails (Papilionidae)
Habitat: rainforest canopy of South-Eastern Asia.

Picture by Biolog
Butterflies of swallowtails family were among the most remarkable
insects of Holocene epoch. Large size and bright contrast colouring draw attention
to these insects when they promptly fly by above flowers somewhere at the meadow
of temperate zone of Earth or above trees of tropical rainforest. After ecological
accident and mass extinction carried away to non-existence tropical woods of
Holocene epoch with all inhabitants, some species of swallowtails managed to
survive. When the climate became warmer and soft, they had occupied again formed
habitats, having given rise to set of effectively looking species. Among descendants
of common swallowtails (Papilio) butterflies of separate genus of metal swallowtails
(Metallopapilio), distinguished with the special structure of wing scales are
especially remarkable.
At metal swallowtails light-reflecting properties of wing scales have amplified;
that’s why representatives of this genus flash in various metal shades: from
cold silvery steel up to gold and green and reddish metal of amazing beauty.
Metal shine is combined at these butterflies with velvety matte blackness at
edges of wings. At all species at back wings there are characteristic “tails”.
Shine of wings permits insects to find out representatives of species at large
distance.
The body of metal swallowtails is usually covered with thick short hairs of
grey and black color. At some species at the middle line of body there passes
white or yellow strip. Metal swallowtails differ in large size of body: the
length of body of the butterfly is about 5 cm, and wingspan at some species
exceeds 15 see.
These swallowtails have very long proboscises - up to 10 cm being unwrapped.
It is the specialization to feeding at trees with long tubular flowers. Adult
insects practically never leave forest canopy.
Caterpillars of these swallowtails feed on trees of other species, rather than
adult butterflies. Especially frequently it is possible to meet caterpillars
on plants which leaves are rich in essential oils. Caterpillars of metal swallowtails
are large (length is up to 10 cm), thick, and they have bright colouring. Usually
in their coloring green color is combined with red and white spots and strips.
Behind the head of caterpillar there is a repugnatorial gland, which in case
of danger turns out as bubble and emits unpleasant smell.
Gold metal swallowtail is one of the most impressive species of genus: its wingspan
reaches 18 cm. Wings of this insect are covered with brilliant scales, playing
in all shades of gold. On edge of wings there passes narrow black border, corners
of forward wings are black too. At every back wing there are two “tails” with
the expanded end and rounded tip. The border on back wings is wider, than on
front ones.
Caterpillars of this species are about 12 cm long; they are almost entirely
black with narrow longitudinal green strips. They feed on trees of family of
labiates (Lamiaceae).
Other species of this genus live in woods of South-Eastern Asia:
Mourning
metal swallowtail (M. hypochondricus) lives at former Sumatra Island
(in Neocene it is a part of Jakarta land). Its wingspan is up to 10 cm. Wings
are almost entirely black, with several lines of silvery scales along main ribs
and large site of silvery color near a root of forward wing. At every back wing
there is one pointed “tail”.
Copper
swallowtail (M. chalcopterus) inhabits islands of Indonesia near Australian-New
Guinean continent. It is very large species: wingspan is up to 20 cm. Wings
are colored copper-red with rust-colored shade; only corners of forward wings
and narrow border at back wings are black. At back wing there is one big “tail”
with rounded tip.
Steel
swallowtail (M. ferrum) lives at the south of islands of Indonesia
and at the north of Australian-New Guinean continent. Wingspan is about 10 cm.
The basic colouring is grey with bluish shade; black border on back wings forms
characteristic indented pattern, on front wings there is only narrow black strip.
On back wing there are three very thin “tails” with pulled together bases.
Urushi
swallowtail (Necropapilio urushiphilus)
Order: Butterflies (Lepidoptera)
Family: Swallowtails (Papilionidae)
Habitat: Japan Islands, humid mountain woods.

Picture by Biolog
Between plants and animals continuous “race of arms” always
proceeds. Plants put forward acute spikes against four-footed herbivores, and
edible parts are penetrated with rigid fibres. Protecting from eating, plants
developed various poisons, pitch and lacteal sap. But the answer to these adaptations
of plants is the next coil of evolution of herbivores. Uniquitous insects have
especially succeeded in counteraction to plants: any kind of plants protected
from the majority of herbivores, appears a forage for any species of insects.
As the consequence of such evolutionary transformations, these insects are very
highly specialized species. It can result adversely in perspectives of their
further evolution, but while nothing threatens to species of fodder plants,
they prosper and appear out of competition.
In warm and humid mountain woods of Japan Islands the plant had reached the
great successes in chemical war against herbivores prospers. All its parts are
literally impregnated with poison (hence its name: “death
tree”), and in air the bitterish smell warning possible lovers of greenery
about danger is felt. Around of this tree only few insects hover, and on the
ground under roots of this tree dead and dying butterflies lay – they all belong
to the same species. Their number is too large to explain their presence in
such place as simple accident.
Actually large bright butterflies, whose life comes to an end at roots of “death
tree”, appeared here quite naturally – they had finished their life cycle. Besides,
it is quite possible, that they began life on the plant of this species. And
this is one of few insects successfully coexisting the “death tree”. The butterfly
is named urushi swallowtail (“urushi” is the Japanese name of poison dogwood
(Rhus vernix), the ancestor of “death tree”, and urushiol is one of poisons
of “death tree”).
The urushi swallowtail is rather large species of swallowtails: the wingspan
at this species reaches 15 cm. Colouring of wings is bright and contrast – orange
background with red edges of wings and black nerves especially well expressed
at the basis of wings. On tips of back wings there are some “tails” among which
the middle one is largest. The basis of this “tail” has round black spot with
white “eye”. Male of urushi swallowtail is colored contraster, rather than female
– it has more black color in coloring, and at separate ones even narrow black
border on wings appears. Body of this butterfly is black with red longitudinal
strips on sides. The abdomen of female is greater, rather than at male.
Caterpillars of urushi swallowtail feed on leaves of “death tree”: usually damages
on leaves of this tree are left by them. Gluttonous caterpillars of advanced
ages leave from leaves of “death tree”only thick middle nerve.
The caterpillar of this butterfly is very big – right before the metamorphosis
it reaches the length 12 – 15 cm. Colouring of caterpillar is black with big
red spots on each segment. On prothorax there are two white oculate spots which
help the caterpillar to imitate a head of small snake or lizard. For additional
protection at them smell gland of two outgrowths is advanced – at danger it
turns outside and emits unpleasant smell. Tissues of this gland are colored
orange and contrastly allocate on the background of caterpillar’s back.
Such colouring of adult butterfly and its larva has the large biological sense
– it is warning one. In fat tissue of insect a lot of poison is accumulated:
the caterpillar receives it from leaves of “death tree”. Even after the metamorphosis
poison remains in body of imago in amount enough to make the butterfly completely
inedible. Development of caterpillar of urushi swallowtail lasts approximately
5 weeks, and for one year at this species up to three generations may develop.
Last generation of insects hibernates in pupal stage. The pupa of urushi swallowtail
also has warning colouring: it is cross-striped red-and-black.
The adult butterfly eats nectar of flowers of various plants, avoiding, however,
flowers of “death tree”. Poisonous substances in its body are accumulated in
fat organ and are reliably isolated from organs and tissues. Its immunity to
poison of plants in which its caterpillar is fed, is much below, rather than
at caterpillar. Adult urushi swallowtails often feed at flowers of Urushiphla
orchid parasitizing on roots of sumach and “death tree”.
Females ready to pairing gather in well lighted places among tree crones. They
fly in sunlight in dense swarms well seen from apart. Females emit pheromones
involving males from the long distance.
Female of this butterfly lays eggs once a life. It finds a fodder tree and sits
on foliage, choosing young sprouts. At this time the substances evaporating
from leaves of plant start to influence an insect. Development of eggs in body
of the female demands a lot of energy, and exhausts an organism of insect. Thus
ability to resist to poisonous substances of “death tree” is reduced, and the
insect appears poisoned.
Female leaves eggs by some ones on leaves of tree. Common fertility of this
species reaches half a thousand eggs and more, therefore laying of all eggs
takes a lot of time. Up to the finish of egg laying the female appears fatally
poisoned with evaporations of plant and perishes. Dead or poisoned insect falls
on the ground under tree. At mass flight of urushi swallowtails the ground under
trees appears interspersed with dead butterflies. Males overlive females not
for a long time – after pairing they stop eating and perish in some days.
Altjira
(Aepypapilio altjira)
Order: Butterflies (Lepidoptera)
Family: Swallowtails (Papilionidae)
Habitat: Meganesia, rainforests.

Picture by FanboyPhilosopher
In human epoch New Guinea was known as a motherland of the
largest butterfly of the Earth – Queen Alexandra’s birdwing (Ornithoptera alexandrae)
from swallowtails family (Papilionidae). This species alongside with a plenty
of bright tropical insects had become extinct in epoch of global ecological
crisis which had come at the end of human epoch. In Neocene tropical rainforests
had covered equatorial areas of the Earth again. And there new species of insects
had evolved, many of which do not concede in beauty and size to the most known
species of human epoch. One of such species – is swallowtail butterfly altjira
from rainforests of the north of Meganesia. The adult butterfly of this species
has wingspan over 30 centimeters, and caterpillars reach the length of about
20 centimeters. Such insects had evolved due to stable conditions and an abundance
of food in places of their inhabiting. The name “altjira” descends from mythology
of one tribe of Australian aborigenes – it is the name of the mythical lord
of sky.
Caterpillars of altjira feed on leaves of forest species of leguminous plants.
They are rather easy for seeing on the background of vegetation: the caterpillar
has black colouring due to which it is quickly warmed up after cold night and
has higher speed of metabolism. On the second thoracal segment caterpillar has
a pair of large oculate spots of orange color with short vertical stroke in
the middle. These spots imitate eyes of small reptiles. The first thoracal segment
is orange-red, and head is black, as the rest of body. The disturbed caterpillar
draws head in thorax, and thus the forward part of its body becomes similar
to head with opened mouth. In such way it frightens off birds. Also it has one
more way of defense: on thoracal segments odorous glands of bright yellow colouring
stuck out, emitting disgusting smell. With the help of such smell caterpillar
frightens off animals searching for food with the help of sense of smell – reptiles
and mammals. If both ways do not work, the caterpillar shoots at the enemy by
partly digested food, adding to it a secretion of modified salivary glands,
stiffening on air in gum-like mass.
Development of the caterpillar, having so numerous protective adaptations, lasts
about 3 months. The adult butterfly of this species looks very impressive –
long peaked front wings give to it the remote similarity to swallows. Forward
nerves on wings are thicken and very strong. Back wings are rounded, and at
their external and back edges some tails grow. Back pair of tails is very long
– at males they reach length up to 15 cm and extend on tips. Adults males of
altjira are colored blue with green metal shine and black front edge of the
wing. In the basis of back wing they have some small black spots. Females of
altjira are green with slight metal shine. On their front wings the thin cross
strokes are scattered, forming faltering mesh pattern, varying at different
individuals. Tails on back wings at representatives of both sexes are colored
black. At males on larger tails of back pair along the middle line the narrow
silvery strip stretches. The bottom side of wings at butterflies of both sexes
is colored similarly – it has yellowish-brown tone, imitating colouring of dried
leaf. In rest these butterflies hang on bottom side of branches and among foliage,
hiding in such way from predators. Body at butterflies of both sexes is black
with greyish-white longitudinal strip stretching from head up to tip of abdomen.
Altjira flies quickly, and in flight tips of wings clap against each other from
above and from below of the body of butterfly with rather loud sound. In courtship
season male, displaying itself to the female, hangs before her in air, loudly
flapping wings. After courtship season tips of wings at males look shabby, and
their colouring fades up to semi-transparency.
Altjira feeds on flowers of various trees and lianas, not giving preference
to any separate species. Due to a long proboscis (up to 12 cm long) this species
can feed even on flowers inaccessible to metal
swallowtails – distant relatives of this species. The maximal life expectancy
of imago makes about 3 – 4 weeks, but usually it is less. For the life the female
has time to make up to five clutches of 300 – 400 eggs in each one. The greatest
death rate at caterpillars falls to the first weeks of life, while at them all
means of protection are not advanced to the full.
Aquatic swallowtail (Exopapilio aquaticus)
Order: Butterflies (Lepidoptera)
Family: Swallowtails (Papilionidae)
Habitat: Sunda Land, freshwater reservoirs.
Neocene is a time of new and unusual animals. After human disappearance the
nature has restored the lost specific variety of ecosystems, and the set of
new species had occupied the newly formed habitats. Representatives of some
groups of live organisms made more or less successful attempts to develop new
inhabitancies where their ancestors did not live. An example of such success
is an aquatic swallowtail, the butterfly from Southeast Asia, the descendant
of emerald swallowtail (Papilio palinurus) of Holocene, a beautiful large insect
having 10 cm wingspan. But anthropogenous pressure (collecting of insects and
destruction of their habitats) had reduced the number of that species and had
resulted in size decreasing of these insects, and then after the ending of
ice age epoch the climate of Southeast Asia started to change. Following the
climate plants, and after them animals dependent on them, including various
butterflies, and among butterflies – the survived species of swallowtails changed
also. As a result of influence of these circumstances to butterfly evolution
in Neocene world absolutely unusual descendants of old genera had appeared.
The aquatic swallowtail is a butterfly occupied completely new to swallowtails
ecological niche due to the small size and ecological flexibility of ancestral
species: freshwater reservoirs of Sunda Land. This island was formed at merge
of several islands of Indonesia due to the influence of volcanic activity and
raising of lithosperic plate at th movement of Meganesia to the north. The
passing of this swallowtail to aquatic way of life had partly happened because
Sunda Land is the area of numerous reservoirs, medium-sized and small – from
streams and rivers up to ponds and lakes. They receive water from daily rains
– it is a consequence of hot and humid equatorial climate. The abundance of
fresh water and presence of various isolated biotops promotes the development
of various freshwater lifeforms, particulary invertebrates, fishes and amphibians.
The aquatic swallowtail is simultaneously both one of the strangest animals
of fresh water, and one of the most usual inhabitants of this island. Its ancestors
passed to semi-aquatic way of life in order to take advantage of new fodder
resource representing plentifully growing marsh and aquatic plants. These plants
give plentiful food to various aquatic and semi-aquatic animal, including caterpillars
of aquatic swallowtail. Its ancestor, an emerald swallowtail, had no bias to
life near reservoirs, preferring tree crones more, but in human epoch the area
of tropical forests in Asia had been reduced, that had placed this swallowtail
literally to the edge of extinction. The swallowtail had adapted to new conditions
similarly to some other butterflies and moths (for example, Saturnia and bagworm
moths): adult individuals stopped feeding, and their role in life cycle of
the butterfly is reduced only to breeding. To any time this strategy had helped
these swallowtails to survive, when they existed mainly as gluttonous caterpillars,
nonpoisonous, but protected by unpleasant smell. But the situation continued
changing. While swallowtails adapted to life on not numerous fodder trees of
habitual species, these plants became rare and smaller, conceding places to
new plant species. The swallowtail had avoided the complete extinction, having
changed the diet qualitatively. Its descendants developed new species of fodder
plants, and some populations had moved to the banks of tropical reservoirs
where they began feeding first on amphibious, and then on semi-aquatic plants.
Caterpillars of new species of swallowtails adapted for temporary existence
in aquatic environment, having developed primitive system of breath both in
air, and under water – their body haв become covered by hydrophobous hair which
kept an air cover around the body of caterpillar, allowing it to breathe. This
adaptation was effective in lower degree, especially before the next moult
and pupating, but it was the beginning of the origin of one of the most unusual
species of lepidopters.
As well as all tropical species, aquatic swallowtails lay eggs all the year
round – they do not have mass simultaneous metamorphosis and imago hatching.
As they spend the life under water not completely, adult females paste eggs
on leaves of plants growing above water. Hatching caterpillars at once fall
in water where they grow, eating various plants. They eat both upwater shoots
of plants and soft underwater leaves. Duckweed frequently becomes the first
food for them. Caterpillars of aquatic swallowtail have ordinary-looking greenish-brown
color, masking them from predators. Their skins have the numerous plicas working
similarly to tracheal gills of underwater larvae of insects of other groups;
caterpillars receive through them oxygen directly from water. Though they have
not so pleasant taste, because of large number of this species many caterpillars
become food for the various aquatic animals, especially for amphibians and
fishes. At growing up caterpillars (life cycle of this species lasts no more
than half-year), skin plicas smooth gradually, and absorption of oxygen from
water through covers is worsened. Larger larvae of aquatic swallowtail move
at first to shoaliness and then creep out from water, living on floating leaves
of marsh plants. They pass metamorphosis on land; caterpillars creep on trees
and spend in pupa stage for 2 – 3 weeks. From pupae small butterflies (wingspan
does not exceed 3 cm) hatch; in their adult life they resemble May flies –
ancient winged insects which also continue the existence in Neocene. Back wings
of this swallowtail keep tails characteristic for the family. The butterfly
is colored white color with black nerves of front wings and wide black border
along the edge of back wings. In the basis of every tail there is a large oculate
spot – red one with white stroke in middle. When the butterfly lands, having
risen wings, they imitate its head, driving the attention of predator from
the true head of the insect. However, it is more the atavistic feature which
is not bearing real advantage for survival of the species. Life at the imago
stage at this species is very short: adult insects mature in some hours after
burst from pupal skin. They die during 1 – 2 day, but before the death males
have time to fertilize females, and females paste eggs on leaves of floating
plants, and so the life cycle of this species repeats again.
The idea about existence of this species of insects was proposed
by Simon, the forum member.
This species of insects was discovrered by Bhut, the forum member.
Ribby
flower pierid (Petalopterina flosculimima)
Order: Butterflies (Lepidoptera)
Family: Pierids (Pieridae)
Habitat: dry woods and semi-deserts of Southwest Asia.
In wild each species of plants coexists with various species of animals, directly
or indirectly dependent on it. It happens, that any species of insects, for
example, lives on one plant, feeds on next one, and its larvae eat greenery
of third species. It happens on the contrary, when two species, plant and animal,
adapt only to each other, and evolve together.
In semi-deserts surrounding the dried up Mediterranean Sea, there are groups
of thick-trunk low true cabbage
trees (Brassixylon crassus) belonging to crucifers family (Brassicaceae).
For some animals these plants are unique source of moisture and food, and sometimes
the unique house.
In spring and in the beginning of summer when clouds from Indian ocean and
Tanganyika passage reach this droughty district and humidify ground, the true
cabbage tree begins to blossom: its crone is crowned by set of large inflorescences
with bright pink flowers. They will bloom not for long, and wind breaks them
from tree and carries away. It looks, however, that not all petals agree to
leave a tree so easily: some of them rather confidently fly against a wind,
and sit back on branches. At the nearest acquaintance it becomes completely
evident, that they are not flowers, and not plants at all, but animals – butterflies,
skillfully imitating by shape and colouring of body flower of cabbage tree.
This species of butterflies is named ribby flower pierid, and belongs to family
of pierids among which there are species, rather not indifferent to plants
of crucifers family. In human epoch some species of these insects were considered
as plant pests, but the nature had put the situation to rights when people
have disappeared and huge plantations, where one or two species of plants were
cultivated have become a thing of the past follow them.
Ribby flower pierid is rather large butterfly: the length of its body is up
to 5 cm, and wingspan reaches 8 – 9 cm. Rounded wings of this insect are colored
pinkish with dark red nerves. The body and antennae of the butterfly are yellowish,
covered with downiness of short hairs. In case of danger this butterfly imitates
flower of the true cabbage tree, on which it lives: it plants wings forward
and back as a cross, slightly raises them (it is the imitation of petals),
lifts up and bends on back the abdomen, lifting head with antennae (imitating
stamens and pestle). Its similarity to flowers is so great, that even in the
beginning of dry season when the tree has already faded, birds do not peck
butterflies sitting among drying up rests of inflorescences of cabbage tree.
The caterpillar eats leaves of the same tree, and for masking is colored green.
Usually these caterpillars sit on edges of leaves, eating soft tissues. After
them only “skeletons” of thickest ribs remains from leaves. Disturbed caterpillars
bend the forward end of body as letter S and belch swallowed and semi-digested
greenery, from which the disgusting smell emits. It is their unique protection,
and the part of them perishes in beaks of birds and teeth of lizards. But fertility
of butterflies of this species giving up to two generations during short rain
season, permits to compensate these losses. Butterflies of the first generation
are “early”: they are small, grow and perish quickly. From their eggs caterpillars
excluse, which gather weight during first half of droughty season, eating withering
leaves of tree. They pupate in cracks of ground and among vegetative dust.
The second generation of butterflies survives dry season at the stage of pupa,
and emerges after first rains of the next year.
“Winged
guard” (Pugnatonymphalis lord-protector)
Order: Butterflies (Lepidoptera)
Family: Nymphalids (Nymphalidae)
Habitat: Far East, subtropical and warm-temperate areas, forests.

Picture by Lambert
Forests are rich in various species of butterflies. Nature
gives them a generous choice of forages at any stage of development – from caterpillar
to imago. In tropical forests extremely specialized species of butterflies frequently
meet, connected in life cycle to unique species of fodder plants. And in temperate
and cold areas of the Earth butterflies may be very unpretentious in choice
of food. Besides of nectar, diet of adult butterflies includes other kinds of
food: tree sap (even fermenting), liquid of decomposed bodies of animals, dung
of large animals. However, some butterflies not feed in adult condition at all.
Evolution does not stay, and frequently live beings can develop odd and unexpected,
but effective ways of survival. At the Far East one species of butterflies had
completely stopped to feed on flowers. This small (wingspan is about 6 cm) bright
butterfly meets on large grassy plants where colonies of plant lice have settled.
This butterfly constantly keeps near the colony of plant lice and drives the
congeners away from it. For this feature it is named “winged guard”.
The sugary liquid producing by plant lice is needed for “winged guard”. It quite
replaces nectar to the butterfly, and “winged guard” only occasionally may be
seen on flowers. Most likely, these ones are “tramps” at which the fodder colony
of plant lice was lost. Within several days such butterflies search for new
colony and continue to lead habitual way of life.
Similarly to ants, “winged guard” stimulates a plant louse to secret sweet liquid.
The butterfly sits in colony of plant lice and cautiously tickles insects by
short antennae. Replying to stimulation plant lice secret sweetish liquid which
“winged guard” licks off by short mobile proboscis.
Such food source is not as accessible, as flower nectar, therefore “winged guard”
is very territorial. Butterflies of this species leave on plants the odorous
marks warning congeners that the territory is occupied. Colouring of top side
of wings is very bright and clearly visible from apart: they are yellow with
black border on edge of wing. Black color of border is shaded with blood-red
strip. On bottom surface of front wings of “winged guard” has bright labels
– on grey background big there are white spots with grayish scales scattered
on them. Scales reflect ultra-violet part of solar spectrum, making the butterfly
especially visible for congeners. The back pair of wings has brownish bottom
side and brown external edge. When the butterfly hides from predators, it closes
bottom side of forward pair of wings by back ones, and light shine does not
give out its presence. The butterfly is very cautious: having noticed a flying
bird it closes wings, covers shining spots and simply falls in grass.
If the congener interferes to the fodder territory of “winged guard”, the owner
of territory protects colony of plant lice from strangers. Driving competitors
away, the butterfly arranges bloodless, but active “duels” in air, pushing congeners
aside from the chosen colony of plant lice. If the contender flies at the edge
of territory, the lawful owner of colony of plant lice flies up and makes some
circles along borders of territory. From time to time butterfly stops in air
for some seconds, trembling wings and showing ultra-violet “flashes” on inside
of wings. Other contenders of “winged guard” are ants. This butterfly can make
of nothing against them, and is content with feeding on plant lice colonies
located at the distance from ants colonies. If such colonies are not present,
butterfly makes a strike on colonies of plant lice occupied by ants, and feeds
while ants had not gathered for protection of plant lice.
At “winged guard” male has dimmer colouring, than female – on top side of its
wings there is no red strip, black strip is narrower, and background color of
wing is paler. It has no constant fodder colony of plant lice and leads “vagrant”
life, searching for females. Pairing at these butterflies lasts more than one
hour. Thus the female sits vertically on plant stalk, and male hangs headfirst,
having closed wings and having drawn legs in. Such position does not cause inconvenience
in him: pairing male simultaneously “plunders” fodder colony of the female,
actively licking off secretion of plant lice. It is its basic source of food.
After pairing the female leaves for a short time the territory to lay eggs.
Caterpillars of “winged guard” feed on leaves of hazel grove and birch, therefore
fertilized female flies to the forest for searches of fodder tree. It makes
it in evening, in order to prevent congeners to occupy its territory. After
egg laying the female flies back, being guided by smell of its own labels. For
the life the female has time to make 3 – 4 layings numbering 200 – 300 eggs
in each one. Caterpillars have not striking green colouring with brown longitudinal
strip on the top part of body. They keep on the bottom side of leaves and skeletonize
them.
For summer this species of butterflies gives two generations. Pupae of the second
generation hibernate. In spring their metamorphosis is finished, and they turn
to butterflies lead a way of life typical for this species.
Sky
pilgrim (Caelestoviator transtetramarinus)
Order: Butterflies (Lepidoptera)
Family: Nymphalids (Nymphalidae)
Habitat: Europe, the Caucasian peninsula.

Picture by Lambert
Events of rather recent past had changed the face of southeast
part of Europe. In ice age Black, Azov, Caspian and Aral have dried up, but
after glacier thawing all of them became parts of the unite reservoir – the
Fourseas. While hollows of the seas were empty, above them numerous routes of
the birds of passage travelling between the north of Eurasia, Asian Near East
and Africa ran. Formation of Fourseas had taken place rather quickly in geological
sense, and a part of birds, keeping adherence to old routes, had been compelled
to cross Fourseas from the north to the south. In addition to birds other flying
creatures, butterflies, use the same route. Some species of migrating butterflies
belonging to different families live in Neocene Eurasia. One of their species
is sky pilgrim making annually mass migrations between places of wintering and
breeding.
Sky pilgrim is the medium-sized butterfly. Wingspan of the female does not exceed
6 – 7 cm, male is smaller a little bit. Wings of this species are strong, dense
and lengthened. When butterfly flaps them, tips of wings touch each other above
and under the body of insect. The clap of wings of the separate butterfly is
almost not audible, but the flock of butterflies, numbering millions of individuals,
makes in flight the noise similar to noise of pouring rain.
Wings of this butterfly are colored brightly enough. The basic background of
colouring is ochre red; edges are white with separate strokes of black color.
It makes a border of wings similar to miniver lining of royal cloak. Edges of
wings are angular and have small dredgings between nerves. Front edge of wing
is smooth and very dense. Back wings are rounded and have narrower white border.
At males wings have more expressed red shade, and white border almost lacks
black strokes. The bottom side of wings at insects of both genders has citreous
color with grey irregular-shaped spots. It is warning colouring; butterflies
are poisonous, because at the caterpillar stage they synthesize a plenty of
cantharidin.
Sky pilgrim winters in forests of Caucasian peninsula. Similarly to monarch
butterflies from America it gathers in unnumerable congestions on branches of
conifers and other evergreen trees. This butterfly spends winter in slow active
condition, making short flights only in the warmest days. Birds do not eat these
butterflies because of acrid taste, and wintering passes successfully. Pairing
at this species occurs in wintering places. When weather becomes warm enough,
insects restore forces, eating nectar of the first spring flowers. Males ready
to pairing begin courtship flight. They gather above the forest in great flocks
similar to clubs of smoke from apart. At once some males start following the
female moving near such flock, but always only one of them becomes a winner.
Pairing occurs on tree branches right under male flock. In these cases some
tens pairs of copulating butterflies may be found on branches simultaneously.
Pairing lasts till some hours. After it male strongly sticks the genital ducts
of the female with quickly stiffening substance. The “chastity belt” falls off
only some days later, when the female loses ability to fertilization. After
this action male lives about two hours. It does not fly anywhere, and only creeps
on branches. After the period of pairing under trees little heaps of male bodies
remain, and wind drives them as if fallen leaves. At females formation of eggs
has a pause which is connected to necessity of migration.
The impregnated females should do long and hard way across Fourseas and Three-Rivers-Land
steppe. After pairing, having released from the males, females begin prepare
to travel. They gather to great flocks above forests of the Caucasian peninsula
and gradually move to coastal areas. Here flock of butterflies, numbering hundred
thousand individuals, are fed within several days. Having accumulated a small
stock of fat, insects begin their travel. They cross Fourseas in the bottleneck,
but all the same non-stop flight above the sea within many hours strongly tires
them. At this time the subjects floating on sea surface become much desired
find for them. Trunks of trees, reed “rafts”, and even the carcasses of terrestrial
animals carried by the rivers, appear covered with a carpet of insects having
a rest. The part of butterflies inevitably appears in water and perishes, but
their death only raises chances of other individuals to leave posterity: On
dead or dying butterflies floating on surface of water frequently their congeners
land to have a rest.
Having overcome Fourseas, flocks of sky pilgrims fly by above steppe areas,
stopping for night in dense congestions on branches of separate trees or simply
at tops of heights. Breeding area of sky piligrim is a zone of mixed and small-leaved
forests of Europe. Here huge flights scatter in searches of suitable fodder
plants. Females quickly restore the physical form, and in their organism eggs
start to ripen again. This butterfly lays eggs on leaves of trees in middle
part of crone. Caterpillars have clearly expressed warning colouring – they
are bright orange with three longitudinal lines of black points along back and
sides. Head of caterpillar has white color. The disturbed caterpillar lets out
portions of strongly smelling dung, but all the same the part of caterpillars
perishes, especially at early stages of development – ants, carnivorous beetles
and also small birds occasionally eating caterpillars of this species hunt for
them in order to get rid of internal parasites. Ichneumons attack larger caterpillars.
Caterpillars of sky pilgrim grow quickly and pupate at the end of summer After
the first frosts, when warm weather returns, from them butterflies burst. Flocks
of butterflies of this species, migrating to the south, reach the even greater
number – they include males also. The beginning of migration is quick: in warm
autumn day of butterflies gather in flocks and in the evening simultaneously
fly to the south. The small concentration of the odorous substances secreted
by representatives of their species is necessary for launching of migration
instinct at this one. Flockr of flying out butterflies keep close to the ground
and in wood zone prefer moving along river valleys where the vegetation is lower.
In steppe zone butterflies unite large flocks and cross Fourseas in common.
At the coast of Caucasian peninsula they restore forces, feeding on nectar of
last flowers, juice of fruit beginning to rot and wood sap. When cold snap approaches,
they gather on trees and hibernate.
Summer
contravirgo (Contravirgo aestiva)
Order: Butterflies (Lepidoptera)
Family: Bears (Arctiidae)
Habitat: Siberia, areas overgrown with grass vegetation and bushes.

Top: summer contravirgo - female, larvae, larva-like
male
Bottom: hibernating contravirgo
Picture by Fanboyphilosopher
In Neocene Eurasia has remained huge landmass where the severe
continental climate dominates. Therefore in central part of this continent the
period favorable for active life of invertebrates is rather short. Evolution
of insects in such conditions frequently appears directed to development of
adaptations permitting as soon, as possible, to develop and to use resources
of the environment more economically.
In this sense among Neocene insects of Siberia moths from Arctiidae family forming
the separate genus Contravirgo are interesting. At these insects development
was sped up so, that they partly even have got ability to breed, remaining in
larval stage. The most common species of this genus is summer contravirgo. The
moth of this species flies in middle and in second half of summer.
For contravirgo the expressed sexual dimorphism is characteristic. The female
of summer contravirgo develops in usual order, turning from caterpillar to winged
imago up to 4 cm in wingspan. The female of this species of insects is poisonous
– it synthesizes poisonous substances, being a caterpillar, and keeps them in
the body after metamorphosis. Colouring of wings of summer contravirgo is warning:
front wings are white with clear black nerves. The bases of front wings and
thorax of moth are covered with rich black hairs. Back wings of summer contravirgo
are bright red with mesh pattern of shining dark blue nerves. Usually adult
insect is rather inactive: in day time the moth hides in grass and sits on bottom
side of leaves, having put wings V-shaped. The disturbed moth stretches front
wings and shows bright coloring of back wings. But in the evening adult females
of summer contravirgo fly, feed and search for males.
There is one more feature of shape of contravirgo – at females of these moths
feather-like antennae, fluffy because of additional hairs increasing sensitive
surface of antennae, are strongly advanced. Usually males of moths differ in
advanced antennae and sharp sense of smell, but at contravirgo the shape of
male is completely other, rather than at usual moths.
The male of moths of genus Contravirgo is actually reduced up to larva able
to pairing. Usually the survival rate of males at larva stage is higher, than
at females – they are smaller and keep secretively, therefore at larval stage
the number of males is more, than females. The male caterpillar of contravirgo
has dim grey colouring with greenish strip along the back and black strokes
on sides. It grows slower, than females, and undergoes less number of moults.
After last moult at it sexual glands roughly develop, and the body strongly
changes externally: the male caterpillar becomes bright orange, starts to creep
out on top side of leaves of fodder plant and emits the special smell attractive
to females. At this stage sexual glands filled with sperm liquid occupy about
the half of body volume of the male caterpillar. At it tenacious walking legs
similar to legs of adult butterfly of other species develop, but abdominal false
legs, characteristic for caterpillars, do not disappear. The head of such male
also gets separate features of shape of adult insect – eyes become larger, and
pectoral segments from above four small shoots appear – these ones are reduced
wings. Mouth parts of the male at this stage degrade, and from them only nonfunctional
rudiments remain. The oral aperture completely grows out. In such condition
the unique applicability of the male is to fertilize the female.
The female flies involved by smell of the male, sits near to it and turns to
pose of pairing, having slightly stretched wings and having extended an abdomen
aside the male. The male creeps on the female, and goes with her to courtship
flight, having clung to her abdomen from below. The male keeps on body of female
by walking legs, enters in sexual ducts of the female all seed liquid which
was formed at him, and quickly perishes after pairing. Frequently it simply
falls down from the flying female on the ground.
Spermatozoids keep viability in sexual ducts of the female for a long time.
As against the male, after metamorphosis she can eat and lives for a long time
– about two weeks. For the stayed life she makes some clutches numbering 200
- 300 eggs each after the single pairing. Also for this time she can fly tens
kilometers, settling to new places of inhabiting. The female finds suitable
fodder plants, lays eggs and perishes usually after the first light frost.
Caterpillars of summer contravirgo are unpretentious regarding a feeding: they
eat leaves of various evergreen bushes wintering under snow. This species has
some externally indiscernible ecological races, feeding on different plants.
Eggs of contravirgo hibernate in dormant condition, and start to develop after
snow thawing. Caterpillars burst from eggs in early spring, and at once start
to eat. They are very hardy: during light frosts they simply remain on foliage
of fodder plant, easily tolerating freezing. After night light frost caterpillars
easily thaw and continue feeding. Being frozen and motionless, they could become
easy catch of various insectivorous animals, but they are protected with poisonous
bristles richly covering the body. At first stages of development caterpillars
of summer contravirgo of both sexes are similar in colouring to the male caterpillar
of advanced age.
Female caterpillars of advanced age have typically warning colouring – they
have black color with cross white strips. Besides the female caterpillar is
richly covered with hairs. These hairs easily break off and stuck in skin of
aggressor, causing irritation. Also they easily get in respiratory ways and
lungs, causing strong burning. Colouring of caterpillar is precisely remembered
by insectivorous birds or small mammals, and they leave alone dangerous larvae.
But in places of inhabiting of summer contravirgo there are some species of
birds able to eat caterpillars protected in similar way. Also ichneumons are
natural enemies of these moths.
In Siberia there is the close species of moths: hibernating
contravirgo (Contravirgo hibernata). This insect eats narrower set
of forages, than summer contravirgo. Fodder plants for hibernating contravirgo
are poisonous species of buttercups family. Therefore hibernating contravirgo
more often lives in damper district where there fodder plants necessary to it
grow usually. This species differs from summer contravirgo in larger size –
the caterpillar of this moth feeds for longer time and pupates later. Female
caterpillars at this species are much larger than males, grow faster and moult
more often. They pupate an early autumn, for some days before males. But the
basic difference from summer contravirgo is that the male in condition of caterpillar
is more active. It finds by smell and fertilizes females being at the pupa stage.
Males of these moths also do not turn to imago, but live longer, than at summer
contravirgo, and perish after fertilization of several females. The pupa of
this species is protected by poison which is received from fodder plants and
is accumulated in hemolymph. The female pupa hibernates and in spring the moth
undergone the metamorphosis, already lays fertile eggs on suitable plants. The
female of hibernating contravirgo has wingspan of about 6 – 7 cm. It also is
colored very brightly – front wings have black color with dark blue shade and
slight metal shine. On back wings of the female dark red irregular-shaped spots
are scattered on white background. The female of hibernating contravirgo does
not search for males by smell; therefore its antennae are rather short and only
a little hairy. The imago of this species eats nectar of spring flowers and
lives for rather long time – up to three – five weeks. The female lays eggs
in several portions of 400 – 500 ones within two weeks, and then perishes.
Two-shaped antivirgo (Antivirgo dimorpha)
Order: Butterflies (Lepidoptera)
Family: Bears (Arctiidae)
Habitat: Siberia, areas of landscape overgrown with grassy vegetation and bushes.
Some moths of bears family living in Neocene at the territory of Siberia had
developed expressed sexual dimorphism as a result of adaptation to rigorous
continental climate. At moths of genus contravirgo
(Contravirgo) males in fact
remain in condition of caterpillar and do not turn to normal imago stage. Very
sharp sexual dimorphism also had developed at moths of other close genus –
antivirgo. At this genus the female matures, remaining a caterpillar, and small
winged male does not differ by features of development from moths of other
species. It finds sexually mature caterpillar females and fertilizes them.
During the life male succeeds to fertilize several females. At non-fertilized
caterpillar females eggs also develop. During the first division chromosomes
in such cells do not separate and new cellular wall is not formed. From such
eggs females hatch exclusively. From eggs of fertilized female both females
and males excluse. Besides in ovoduct of growing caterpillar female eggs continue
to form some time after fertilization. Last portions of eggs in clutch of fertilized
female are unfertilized, and from them small haploid males develop.
Fertilized caterpillar normally undergoes metamorphosis and turns to imago.
Imago of antivirgo serves only for settling of species – it finds trees suitable
for development of caterpillars, lays eggs and perishes. Imago of antivirgo
of both genders does not feed, oral parts of these butterflies are underdeveloped.
Eggs of antivirgo winter, and in spring when growth of plants begins, from
them caterpillars burst.
Caterpillars and moths of genus antivirgo are protected from predators by poison.
7 – 8 cm long caterpillars of this species are covered in rich black hairs.
On black background the white longitudinal strip on back side of body is clearly
visible. It is warning colouring supported with effective way of protection.
Hairs of antivirgo caterpillars are poisonous and easily break off at the basis.
Sticking to mucous membranes they cause strong irritation and hypostasis. Caterpillars
of antivirgo produce poison by themselves. They feed on nonpoisonous plants
of various species – dogrose, hazel grove and large grassy plants. Male caterpillars
grow slower, rather than female ones.
Imagoes of this genus also are poisonous. Male and female of two-shaped antivirgo
sharply differ externally (hence the specific name of this moth). Male represents
small insect (wingspan is no more than 3 cm) of red-brown color with black
tip of abdomen and white spot on thorax. Wing nerves are a little darker, than
the basic background of wing colouring. The female is much larger, than male
– its wingspan reaches 7 cm. Front wings of female have cross-striped black-and-white
pattern; strips are irregular-shaped. Back wings are colored bright blue with
metal shine, contrasting with strict colouring of forward wings. The body of
female is covered with rich velvety hairs of cherry-red color. The disturbed
female opens front wings and displays shining of back wings. It supports the
warning of inedibility, emitting sharp bitter smell in air.
Antivirgo males fly in August, and imago females appear at the end of August
and in beginning of September. They fly till only some days, but appear simultaneously
in plenty. Females of antivirgo are most active within midday hours when their
body is well warmed by sun. Flight of antivirgo females marks the end of Siberian
summer.
River silkworm (Potamobombyx socialis)
Order: Caddis flies (Trichoptera)
Family: Neotenical caddis flies (Pedopsychidae)
Habitat: Eurasia; rivers, streams and lakes of Siberia.
Representatives of caddis flies order were very characteristic inhabitants
of freshwater reservoirs in human epoch. But drainage and pollution of reservoirs
had caused the great damage to their populations, and many species became very
rare and had disappeared at the significant part of the area. Few populations
of caddis flies had been kept only in small rivers, but it was enough for rather
quick returning of these insects to the lost positions after human extinction.
Among the caddis flies from cold reservoirs of Holarctic the special family
evolved, which representatives developed rare for insects ability to breed
at larval stage. One species of this family is the river silkworm living in
reservoirs of Siberia.
Similarly to all caddis flies, this species spends a significant part of life
at the stage of larva. The size of larvae is about 8 – 12 mm. Their body is
long and thin; legs are short (about 0.8 – 1 mm long), but keep all segments
and are very tenacious. Tracheal gills are present at these larvae, but they
are very small and breath is almost exclusively through skin.
In summer, when water plants grow actively, this species forms colonies on
floating leaves of water lilies. River silkworm had lost the ability of decoration
of shelters with various small subjects, and instead of it makes complex constructions
of silk on the bottom side of leaf. Each leaf (or some leaves floating near
each other) is occupied with the numerous colony of larvae numbering some hundreds
of individuals. Each individual in colony has its individual habitation looking
like silk tube hanging downwards. On bottom surface of leaf these tubes are
connected at the basis with a circuit of randomly directed tunnels in which
walls casually getting dust is intertwined. In common silk tunnels these caddis
fly larvae move, bending the body like inchworm caterpillars; the middle pair
of their legs at this time is folded and nestled against lateral parts of thoracal
segments in order to not prevent movement. In its own tubes it simply creeps,
pulling the body by two pairs of walking legs (1-st and 3-rd ones), having
folded and pressed to sides legs of the second pair, and pushing by cerci.
They make “steps” due to 2 or 3 pairs of prolegs located on first abdominal
segments.
The larva spends a significant part of time in its own shelter, and only at
its destruction by predators abandons it, escaping in the common tunnels of
colony. The larva is a filtering organism; it hangs on the bottom surface of
leaf, keeping by cercae on internal surface of shelter and keeping slime-made
trap in middle pair of legs. It almost constantly makes wavy movements by the
whole body, simultaneously filtering water and promoting skin breath (approximately
as many modern sea worms act). Silk glands of the larva produce slime of special
sort which inflates in water and turns to long sticky strings. Middle pair
of legs of the larva is lengthened: they reach about half of length of the
larva body. When it is put out from shelter, these legs extend downwards and
in sides. The larva preliminary smears them with such slime, and tips of these
legs turn to mucous trap for small prey. The larva makes raking movements by
middle pair of legs, and from time to time brings them to the mouth to lick
off the gathered plankton. If larger prey is caught, the larva pulls it from
slime by forward pair of legs. If near the colony there is a clutch of eggs
(usually snail clutch), the larva of river silkworm eats these eggs, covering
them with silk web in order to protect it from food competitors.
In case of danger the larva is hidden in silk tube, closing the entrance by
large flat head. If danger is great (for example, fish or water beetle tries
to break off silk constructions), the larva abandons the tube and escapes in
the common tunnels of colony. If the colony is attacked only by one large predator,
larvae are easily escaping, simply creeping in tunnels to another edge of colony.
In the colony of river silkworm the complex combination of various ways of
breeding takes place. It is a dioecious species, and in some cases for breeding
fertilization is necessary. Among representatives of the family this species
is primitive enough as it keeps an adult flying stage. The river silkworm breeds
during the whole larval life (one larva lives till about 6 – 7 months). Terms
of existence of different generations are imposed against each other, especially
in cold rivers and lakes where development of larvae is delayed. The generation,
at which second half of life falls to spring and the beginning of summer, undergoes
metamorphosis and turns to winged imago stage.
This species of insects does not have strict alternation of generations, and
development of one or another generation is controlled by pheromons. Larvae
have two pheromones, A and B. A pheromone is produced by growing up settling
individuals and “prohibits” to other larvae to develop to settlers, and B pheromone
is produced by filtrating ones and in higher concentration (for example, at
the overpopulation of colony) stimulates larvae to develop to settlers. Development
of settlers is possible at high concentration of B pheromone and low concentration
of A pheromone. Usually settlers develop from first half of summer when colonies
of filtering larvae become numerous enough. At high temperature of water settlers
are capable to turn to imago. Growing up settlers can not turn to filterers
because of absence of slime-producing glands. They feed, scraing bacterial
cover inside the tunnels of colony, and also eat secretions of anal glands
of filtering individuals.
The way of life of larvae of river silkworm depends on their age. Larvae of
younger age (up to 2-nd moult), not able to make slime traps yet, are feed
on edible secretions of anal glands of larvae of more advanced age, and then
turn to filterinf organisms. If they should grow outside of a colony, they
survive successfully and feed on algae and the sedentary rotifers attached
to plants, but at “independent” life their death rate is much higher. Having
found a colony, such larvae build silk tubes and lay in them up to 20 – 30
parthenogenetic eggs from which the second generation bursts. They feed up
these larvae with their secretions, but these new larvae quickly enough pass
to feeding by filtering water. The following generation of larvae is settled
on the whole surface of leaf, making narrow temporary tunnels. Further larvae
expand and strengthen them, and at the edges of tunnels there are tubes of
new filtering individuals. From time to time filtering larvae lay eggs with
haploid set of chromosomes, from which males hatch. Male larvae of filtering
generation grow slowly and quickly perish, and under the influence of B pheromone
develop normally, feeding on secretions of filtering viduals and develop to
normal settler larvae.
In an autumn, from the end of September up to middle of October, but before
the temperature of water will lower below +5 … +7 degrees Celsius, larvae of
settling generations get the tight cuticle during the next moult and leave
a colony. Settler individuals of this generation do not turn to imago, but
keep activity even in cold water that gives them advantage – fishes pursue
them less actively. Settler larvae have stronger walking legs, and back pair
of legs becomes swimming organs – on them the fringe of rigid bristles develops.
They are inclined to settling with the help of phoresia: these ones frequently
cling to floating vegetative dust, feathers and legs of waterfowl and wool
of aquatic mammals. Shortly before approach of frosts they creep out on land,
creep away to thickets of coastal plants and run into anabiosis in wood litter.
These larvae easily endure freezing. In spring they copulate, females creep
to the nearest reservoirs and lay eggs gathered to large clutch on the bottom
side of leaves of water plants, and then perish. When leaves of water lilies
die off, larvae eat a significant part of silk of their constructions (it is
literally almost pure protein), and the colony moves to the bottom where spends
winter in thickness of friable ground. The next spring survived individuals
found new colonies.
In summer representatives of filtering generation are settled also – some kind
of “budding” of the colony takes place. They leave their shelters and simply
creep to the next leaves.
This species of insects was discovered by Mutant, the forum member.
Crab
botfly (Carcinotaba vorax)
Order: Two-winged flies (Diptera)
Family: Horseflies (Tabanidae)
Habitat: South-Eastern Asia, Jakarta Coast, Sunda Land and nearby islands.

Picture by Biolog
Among insects of two-winged flies order parasitism is widely
spread. Some of these insects suck blood (mosquitoes, midges, some flies), others
parasitize in body of large vertebrates (larvae of botflies). In tropics of
Asia one of species of horseflies has adapted to parasitizing on ground crustaceans.
Adult flies of this species suck blood of crabs and ground hermit crabs. Larvae
are predators as at the majority of horseflies. But their catch is not soil
animals, but eggs and young growth of the same crabs.
Externally crab botfly only a little differs from other flies. But at this insect
the sexual dimorphism is advanced. The male of this species eats of nothing,
and lives not for long time. It is colored white with the silvery shine reflecting
ultra-violet rays, and its eyes are emerald-green. It “displays” on the lighted
place on tree trunk, uttering characteristic buzzing and involving females by
specific smell. The female is colored grey with black longitudinal strip; its
eyes are brown. Right after hatching from pupa females search for “displaying”
males, pair with them and depart away. At females organs of smell sense are
very much advanced: it is necessary for search of the specific host animal on
which this species parasitizes. Right after pairing the female quenches blood,
and in its organism… not eggs, but at once alive larvae start to develop. Having
got to the host animal, they at once start to grow and develop.
For cultivation of posterity the adult crab botfly finds the female of crab
had recently laid eggs. Fly cautiously flies up behind, and sits on carapace
of crab. Having waited some minutes while the animal will calm down, the insect
strongly extends an abdomen, and postpones in cavity between cephalothorax and
abdomen of crab the live larva covered only with thin film. If the crab is large
(this insect frequently parasitizes on huge “mangrove
robber” crab (Archocancer mangrophilus), some larvae can get at once to
it. The larva of crab botfly at once starts to eat eggs of crab, and frequently
destroys them almost completely. If it does not suffice eggs for feeding, larva
can bite through covers of crab and lick off flowing out blood. In case of shortage
of forage one larva can to have eaten other ones if they appear together on
one crab. Having fattened up to the necessary size, the larva abandons a body
of crab, and falls in wood litter. Having buried in the ground, it pupates,
and in one week turns to adult insect.
This species was discovered by Arseny Zolotnikov.
Infectiofly
(Dolichomusca infectans)
Order: Two-winged flies (Diptera)
Family: Robber flies (Asilidae)
Habitat: tropics and humid subtropic areas of Old World – Asia and the southeast
of Europe.

Picture by Biolog
In Neocene on the Earth many species of large insects had
evolved. They are usual mainly in warm climatic zone and belong to various systematic
groups. Each species of such insects indicates by its existence the stability
of habitat, because the very existence of such species is on the verge of physiological
opportunities of insect organism in whole.
In tropical areas of Old World where rainforests expand and a plenty of species
of vertebrates lives, many kinds of carrion-eating insects live. Among them
the large fly having body shape similar to dragonfly, 8 – 10 cm long, is remarkable
in its appearance. It is one representative of predatory robber flies adapted
in due course of evolution to feeding on carrion. But this fly had developed
the special mechanism which simultaneously allows it to feed, avoiding a competition,
and also protects it from enemies – it had entered symbiosis with various species
of bacteria among which there are the varieties pathogenic even for large vertebrates.
Hence it is named infectiofly.
This fly has bright colouring – it is cross-striped like wasp: on dark blue
background with expressed metal shine wide cross strips of yellow color stretch.
An abdomen of this fly is rather narrow and mobile, and the thorax is very expanded
– inside it there are strong flying muscles, which permit this insect to fly
at speed making inferior to speed of birds and dragonflies only a few. Eyes
of infectiofly are large and have numerous facets, providing almost complete
circular field of view. But the main sense for this insect is sense of smell.
Infectiofly catches in air even the slightest smells indicating the death and
the beginning of corpse decomposition, and due to strong wings it appears on
the corpse among the first scavengers.
The mouth parts of this insect represent pointed stabbing proboscis which in
rest is placed along the body between the bases of legs. This proboscis is rather
long (about one third of general body length of this insect) and strong – it
can pierce even thick skin of corpse of large herbivore. Infectiofly has an
original analogue of a craw – the pocket in gullet where symbiotic pathogenic
bacteria, helpers and defenders of this insect, breed. They easily endure the
influence of digestive enzymes of insect.
This fly transfers bacteria, pathogenic to vertebrates, therefore birds and
insectivorous mammals seldom hunt it. For the majority of arthropods this fly
is too large to hunt it – only large spiders and soothsayers can catch and eat
these insects without harm for themselves. Infectiofly eats mainly carrion though
it can diversify the diet with other arthropods. This fly is feeding, injecting
digestive juice in thickness of decomposed carrion, and sucking out the made
“broth” with the help of proboscis.
The care of posterity observed at this insect is original. Larvae of infectiofly
develop in carrion. Also this insect is live-bearing, and in ovoducts of the
female always there are some larvae ready to be placed to suitable substratum.
The insect precisely enough estimates the size of dead animal, and leaves in
it exactly as much larvae, as much can successfully live in it. Before the leaving
of larvae the female pierces carrion by proboscis some times. It injects in
these places the small amount of digestive juice and lets out from “craw” the
certain amount of bacteria developing in it. Then female sticks firm ovipositor
into the carrion infected with bacteria and places the larva in thickness of
meat. In large carrion this fly lays some tens larvae, in small (mouse-sized)
only one or two larvae may be placed. Vertebrates do not disturb them, because
this carrion is infected and inedible for them. The enzymes produced by symbiotic
bacteria slow down process of rotting, and the larva has time to eat food before
it will decompose. Also symbiotic bacteria interfere with an eating of the carcass
by other invertebrates. Activity of bacteria transforms meat of the carcass
to the food easily digesting by infectiofly larvae. Having developed completely,
larvae pupate and after the metamorphosis turn to adult flies. In adult condition
infectioflies copulate and search for carrion. Male differs from the female
in larger eyes and thinner abdomen. Pairing occurs in air; males actively search
for females and arrange combat in air forf them, driving contenders away from
the female. After pairing the female can lay up to 100 larvae.
The idea about existence of this species of insects was proposed by Arthropod, the forum member.
Sambio
(Sambio mortalis)
Order: Two-winged flies (Diptera)
Family: Robber flies (Asilidae)
Habitat: Tropical Africa, equatorial areas of Zinj Land.
Picture by Biolog |
In human epoch tsetse fly (Glossina palpalis), a carrier of
some fatal diseases, had been one of the most terrible insects of Africa. After
human epoch it difficultly enough to trace the history of this species and its
descendants. Probably, it became rare in epoch of anthropogenous pressure when
number of large species of mammals had been strongly reduced. Probably, it had
died out during the ice age, which had followed the extinction of mankind. It
is possible also, that any of its descendants passed to feeding on blood of
small vertebrates. However, in Neocene, when herds of large animals roam on
the Earth again, another fly, also inspiring fear to animals, had replaced tsetse
fly. This species is the deadly sambio fly, the representative of robber flies
family. Neocene robber flies had evolved very large infectiofly
(Dolichomusca infectans), widely settled in tropics of Old World and feeding
on carrion. But in due course of evolution of this species at the open spaces
of Africa the lateral line of carnivorous flies branched off; these ones get
their food in different way. They do not search for already dead animals, and
kill live ones by sting. One species of these flies is sambio fly living in
tropical rainforests of Africa and Zinj Land.
Small warm-blooded animals – rodents, bats, insectivores – fall prey of sambio.
Mouth parts of sambio represent long and pointed proboscis, which length makes
about half of total body length of insect. Having tracked down prey by smell,
sambio female pursues it, keeping aloof. Having chosen a right moment, sambio
rushes to its prey and puts fatal sting to it. At the moment of sting the saliva
mixed with contents of “craw”, in which symbiotic bacteria live, is injected
to the body of prey. Actually, sambio female poisons the prey (hence the name:
“sambio” is a poisonous substance in language of kisi tribe). After that fly
continues pursuing of the bitten animal, distinguishing it with the help of
smell. The bitten animal is doomed – during one or two hours it perishes from
the poisoning. The corpse of animal teeming with pathogenic bacteria becomes
food for larvae: sambio female lays larvae in tissues of still warm corpse.
Shape of sambio is considerably less impressing, rather than shape of infectiofly.
Length of sambio female is about 30 mm. Its body has ordinary-looking grayish-black
color with dim cross rings on abdominal segments. Wings of sambio female are
transparent, and only at the basis are dark, as if smoked. Eyes of this insect
are dark green. An abdomen of the female is lengthened; its tip is very mobile
and supplied with strong ovipositor. After fertilization sambio female turns
to the conveyor on larvae producing – within one day in its organism 2 – 3 larvae
ready to independent life develop.
Sambio male differs from the female by the shape in great degree. This insect
is up to 25 mm long, with very thin abdomen. Colouring of male body is dark
blue with sparkling metal shine – some hairs on its body are turned to light-reflecting
scales. Wings of male have dark violet colouring, and eyes are emerald-green.
Male actively searches for females with the help of sense of smell.
At insects of both genders thorax is strongly inflated and expanded. The digestive
path in this place forms the shoot directed upwards – the “craw”, in which symbiotic
bacteria develop. The role of these bacteria in life of sambio is very great
– they take part in prey killing and prepare it for feeding of larvae. Vertebrates
do not eat the dead body of the animal infected with bacteria – it gets a special
smell. Tissues of the corpse are decomposed much slower, and sambio larvae eat
them up to the moment of metamorphosis. They grow very quickly, and are ready
to metamorphosis within one week.
Sambio attacks exclusively mammals, because symbiotic bacteria develop successfully
only in their bodies. The birds bitten by sambio survive due to higher body
temperature at which development of bacteria is suppressed. And reptiles do
not fall ill because of significant distinction from mammals in biochemistry
– bacteria do not develop in their organisms and quickly perish. In forests
of Africa some related species of these predatory flies attacking other animals
are present:
Tormentor
sambio (Sambio cruciator) is the large species of flies parasitizing
in large tetrapods. The length of adult individual of this species reaches 40
– 45 mm. Female and male differ in colouring in small degree - they both are
dark grey with separate silvery hairs on body. Male differs from the female
in narrower body and larger eyes. The female lays larvae under skin of large
mammals and birds, having preliminary put the sting by proboscis. In place of
sting the center of an inflammation is formed, causing strong pain to the host
animal. Action of bacteria causes inflow of blood to the bitten place, and the
larva eats blood and friable connecting tissue. This species inhabits Africa
at the border of savanna and forests.
Snake-eating
sambio (Sambio ophiophagus) inhabits tropical rainforests in Equatorial
Africa. This species specializes in feeding on reptiles. It attacks usually
small snakes up to half meter long, and alongside with predatory birds and mammals
exterminates a significant part of young ones of these reptiles. Also snake-eating
sambio attacks medium-sized lizards. At this species female is colored goldenyellow
with dim shine; wings are transparent, and eyes are dark red. Male is silver-grey
with ruby-red eyes; the bases of wings are dark grey.
Bird-eating
sambio (Sambio ornitivorus) inhabits rainforest canopy in Equatorial
Africa and Zinj Land. This species attacks exclusively birds. Its symbiotic
bacteria are capable to maintain high body temperature of attacked animal and
differ in high virulence – the pigeon-sized bird, bitten by this fly, perishes
less, than in one hour. This species of flies has the smallest size among representatives
of the genus: the female is 14 – 15 mm long, and the male’s size is only 12
mm. Colouring of these flies is imperceptible – black with dim shine. The female
behaves extremely cautiously, and more often waits for birds, having hidden
on branches and from the bottom side of leaves. Sometimes this fly attacks the
nestlings having not leaved the nest, and is capable to kill the entire hatch
for one visiting of the nest.
Bird
leechfly (Bdellomusca ornitina)
Order: Two-winged flies (Diptera)
Family: Leechflies (Bdellomuscidae)
Habitat: temperate, subtropical and tropical areas of Old World: Eurasia (except
for Siberia and Beringia), Africa, Zinj Land.
Picture by Biolog |
Two-winged flies represent successful group of insects in
evolutional aspect. They are cosmopolites and occupy great amount of various
ecological niches. Some species
had developed even hyperhaline reservoirs of Mediterranean hollow, and others
became predators in this
inhospitable area. But nevertheless the majority of two-winged flies lives in
less extreme conditions.
Among two-winged flies there are many parasites of other animals, including
vertebrates. These insects parasitize at various stages of development: some
ones at larval stage and others in adult condition. In Neocene in Old World
representatives of new family of two-winged flies, leechflies, had widely settled.
These insects are adapted to parasitism on vertebrate animals of various species.
Some species of these flies are highly specialized parasites of separate species
of vertebrates, and others use wide set of species for feeding. Leechflies bear
live larvae which destiny at different species develops differently. Some species
leave them directly on skin of the host (usually it is characteristic for highly
specialized species); at such species fertility is small, but larvae are larger,
and also survival rate of posterity is higher. Other species leave larvae in
places of most probable occurrence of possible hosts, and do not care of posterity
any more. At such species larvae are small, and their survival rate is lower,
because they not always succeed to find suitable host in time. High fertility
of such species (up to 200 – 300 larvae from one female) compensates death rate
of posterity. Right before metamorphosis the length of leechfly larva 2 – 2,5
times exceeds length of adult individual. Imagoes at all species of leechflies
eat other food, rather than larvae: rotten organic material, dung of animals,
nectar. Depending on breeding way the shape of these insects differs. At leechflies
leaving larvae directly on body of host, the sexual dimorphism is sharply expressed.
Males of such species have bright and beautiful colouring, and females have
ordinary-looking grey or black colouring, and differ from other species of flies
only a little. The species which simply leave larvae in suitable places, have
bright colouring with metal shine at both genders. The way of breeding is precisely
connected to the size of adult individual: more prolific species are larger,
than specialized ones. On tips of wings of leechflies there are dark spots;
at males they are larger, than at females. Females of leechflies also have the
feature connected to gender: on their back pair of legs special attaching hooks
and suckers are advanced, permitting to keep on the body of the host.
Larvae of leechflies differ from larvae of other species of flies in few features
of anatomy. They have worm-like segmented legless body and lack the expressed
head capsule. But, as against larvae of other flies, larvae of leechflies have
a special sucker with hooks on forward end of body. Having searched the suitable
host, larva of leechfly sticks to its body and drinks its blood. The larva sticks
to skin where it is thin or where there are wounds. It keeps on skin of host
with the help of attaching hooks. At some species there are additional hooks
on body segments. Larvae of flies lack mandibles with which help it is possible
to gnaw through or to pierce skin of host. Therefore they solve a problem of
feeding rather simply: larvae produce active digestive enzymes which dissolve
the top layer of skin of host animal and stick to the wound. Due to action of
digestive enzymes of leechflies larvae wounds made by these larvae do not get
inflammation and do not fester, and after striking off of the parasite heal
rather quickly. The saliva of leechfly larva contains the substance interfering
blood fibrillation and rendering local anesthetizing effect.
Larval stage is limiting part in settling of these insects. The adult insect
is well protected from drying by chitinous armour, but larva has thin covers
and rather quickly loses moisture in dry air. Therefore leechflies meet mainly
in areas, where air is damp – in forests and near reservoirs.
Bird leechfly parasitizes on various birds living in forest canopy, and is the
typical representative of family. Its length does not exceed 5 – 6 millimeters.
The female of this species has grey colouring with darker thorax, and reddish-brown
eyes. Male strongly differs from the female in appearance – it is larger and
has golden-yellow colouring with metal shine. This insect meets in forests of
Southern and Southeast Europe. It attacks small birds, mainly passerines, but
occasionally meets on birds of other taxonomic groups.
Males of this species are easily appreciable: during the most part of summer
they arrange courtship flights. Males display themselves to females, flying
in forest canopy. Involving the female, male can stop for a long time in air
in ray of sunlight appear through forest canopy. If the competitor appears near,
male rushes to it and drives it away. If the female moves nearer to male, it
arranges chase for it, from time to time stopping in air and uttering special
hum of higher tone. Pairing occurs in air and lasts for some seconds.
After pairing in ovoduct of female larva starts to develop. It eats secretions
of walls of ovoduct, and quickly reaches the weight of approximately one third
of weight of adult female. When the larva is ready to independent life, the
female searches for host bird of suitable size. Usually the female of leechfly
leaves larvae on skin of birds in the evening or in the morning when bird is
less active. The insect creeps under plumage of bird, and tries to find by smell
the injured site of skin of bird. When suitable place is found, the leechfly
female gives rise to the larva which at once starts to feed. Newborn larva attaches
to skin of bird by viscous secretions of special glands on the bottom side of
body and starts to suck blood.
Development of larva lasts about five weeks. Thus, till the summer bird leechfly
can give three, and in warm years even four (at the south of area up to five)
generations. At leechflies pupae winter.
The stuck larva eats almost continuously. Nevertheless, it adequately reacts
to world around. When bird cleans feathers or searches for parasites, larva
unfastens from wound and starts to creep between feathers quickly, escaping
from beak of bird. When conditions become quieter, it finds by smell the place
of feeding, and continues blood sucking. When development of larva comes to
end, it leaves the host and pupates in moss growing on branches. Larvae of other
species of leechflies pupate in wood dust, forest litter, or in soft ground.
This genus includes many species differing by habit of life:
Hollow
leechfly (Bdellomusca alveophila) lives in forests of Europe from Atlantic
up to Three-Rivers-Land steppes and parasitizes on wide spectrum of species
of animals – its larvae feed on bats, small carnivores, various birds. This
species is easily distinctive from other leechflies by reddish-brown colouring
with faint shine, and by bright orange eyes at insects of both genders. Hollow
leechfly belongs to largest species of genus – the length of adult insect reaches
10 – 12 mm. Fertility of this species makes up to 30 – 40 small larvae for one
laying. The female leaves them not on skin of the host animal, but in hollows
of trees where the probability to meet suitable animals for feeding is more.
Larvae not constantly live on animal, but only temporarily stick to it for feeding.
They prefer to attack bats at which wing membranes are plentifully penetrated
by blood vessels. Adult individuals eat fermenting tree sap and fruits of plants
beginning to rot: yeast are the main component of food of imago. For one year
this leechfly gives only two generations.
Burrow
leechfly (Bdellomusca subterranea) lives in Three-Rivers-Land steppes,
at Balkan, at the edges of Mediterranean swamps and in savannas at the north
of Africa. It parasitizes strictly on burrowing animals – digging mammals and
their neighbours (usually on birds). Very seldom larvae of this species may
be met on lizards and toads. Adult insect is about 8 mm long and has grey color
with metal shine. On each segment of body larva has a fringe of short strong
bristles permitting to keep more durable on body of host animal.
Corroding
leechfly (Bdellomusca dermatolysa) is the largest species of the genus
living in Southern Europe and Southern Asia (to the south from Himalayas mountain
circuit). Length of adult female of this insect is about 15 mm; male is about
10 mm long. Male has contrast colouring: body has copper shade, reddish metal
shine and green eyes. Female is yellowish-grey with black longitudinal strip
on abdomen. This species of flies parasitizes on large mammals. The larva is
well adapted to reach blood vessels through thick skin of host animal. It injures
skin producing active digestive enzymes. Usually the forward part of body of
larva is immersed in skin of mammal. The disturbed larva quickly gets out from
the wound and tries to hide in wool of host animal. The female gives rise to
large larvae (about 30% of weight of the female) with periodicity in 2 – 3 days,
sitting on skin of large mammals. Adult individuals feed on dung of mammals.
Predatory
leechfly (Bdellomusca rapax) from North Africa and Western Europe represents
the exception among representatives of genus. Its larva is not an ectoparasite,
but an active predator. It feeds on large mammals and birds. The adult individual
of this species feeds on flowers; its proboscis is long enough, and helps to
reach nectar in deep flowers. As against the majority of leechflies which females
have dim colouring, at this species the female is colored silver-grey with green
eyes. Male, on the contrary, is coal-black with metal shine. It also has green
translucent eyes. The animals, instinctively feeling danger of blood-sucking
insects, are not afraid of this leechfly. It freely creeps on the body of large
animals, and leaves on their skin large larvae. Young larvae of predatory leechfly
exhaust parasitic insects and ticks (especially sated of blood), and later start
to swallow them entirely. Larva pupates in ground.
Representatives of close genus of aquatic leechflies (Aquabdellomusca) are even
more adhered to humid habitats. They parasitize on various amphibians and other
water vertebrates. Their larva has the special adaptations, permitting to receive
oxygen from water - on its body segments long elastic outgrowths penetrated
with tracheal tubules develop. When larva is in water, its stigmas close, and
outgrowths extended and promote gas exchange - oxygen from water diffuses through
thin covers directly in tracheal system. Besides at larvae of aquatic leechflies
additional attaching hooks on two back segments of body develop – it is the
adaptation which permits the larva to keep on the body of host at its movement
in denser environment.
Duck
leechfly (Aquabdellomusca anatina) lives on bottom part of body of
waterfowl – ducks and grebes. It has very wide area – from Fourseas and rivers
of Southern and Southeast Europe up to Equatorial Africa and South Asia. The
settling of this species is obviously connected to migratory ways of European
birds. The length of adult individual of duck leechfly does not exceed 6 mm.
The female has dark grey color with white points on thorax and the first segments
of abdomen. Male is bright green with metal shine. Eyes at individuals of both
genders are orange-red.
The imago of this species feeds on rotten parts of water plants, and frequently
meets at the coast of reservoirs in innumerable flights. Adult males and females
form the common swarm, and feed in common. After pairing when in ovoduct of
the female some small larvae start the development, it searches for the host
bird suitable for the posterity. The female cautiously sits on plumage of bird,
and waits, while the bird will calm down and the will cease to pay attention
to it. Then the female creeps in plumage of bird, and quickly bears several
larvae on its skin. Larvae creep, find blood vessels stretching close to skin
surface, and stick to it. When the bird dives, in its plumage the layer of air
which permits to larvae to breathe is kept. At larvae of this species adaptations
to inhabiting in aquatic environment are expressed in lesser degree.
Frog
leechfly (Aquabdellomusca ranophila) parasitizes on frogs of various
species. It reaches length of 6 – 7 mm and lives in tropical and subtropical
areas of Old World. Females of this insect have expressed camouflage colouring
varying at different populations from sand grey up to brown and black. Eyes
at females have about the same color, as a body. Males of this leechfly sparkle
golden-yellow colouring with greenish metal shine and emerald-green eyes. They
keep near swamps and marshy areas of river channel, gathering to dense swarm.
When the female flies nearer to the male congestion, they rush to chase her,
and only after several minutes of chaotic flight one male is coupled to the
female.
The female shows very big care, searching for the host animal for larvae. Having
found a frog, the female of frog leechfly flies behind where frog does not see
it, and cautiously creeps nearer to the frog. Some last steps it moves an abdomen
forward, having extended it. The female of leechfly slightly touches skin of
frog, throwing out the larva on frog skin by contraction of muscles of abdomen.
The larva at once is attached to the body of frog, and only after that starts
to search on its body for a place suitable for feeding. The body of larva is
translucent, and is poorly appreciable on the background of motley skin of frog.
Turtle
leechfly (Aquabdellomusca testudophaga) is the largest species of genus.
Its length is about 15 mm. It inhabits areas where turtles live – tropics of
Old World from Equatorial Africa up to Japan Islands. This insect has copper
red colouring, and male differs from female only in yellow color of eyes (at
females they are brown) and presence of black spots on wings. This insect leaves
larvae on bodies of various turtles - from huge African crocoturtle
up to small turtles of Southeast Asia. The female bears larvae on back part
of body of turtles, near the basis of tail and under carapace. Larvae are covered
with dense cuticle, and tracheal gills at them are advanced only on last segments
of body. The adult insect feeds on decomposed corpses of various animals.
Flat-bodied
leechfly (Aquabdellomusca platysoma) is named because of body shape
of the larva, which is very wide, and resembles flat cake in rounded outlines.
On edges of larva body the additional suckers develop from plicas of integumentary
tissues, permitting it to keep on smooth skin of host animals. This species
lives in Southeast Asia and parasitizes on body of amphibians living in quickly
current mountain rivers. More often this leechfly attacks flat
toads. The adult fly keeps in thickets of wide-leaved grassy plants,
on the bottom side of leaves, waiting while in thickets amphibian of needed
species appears. Attack of the female of flat leechfly is very fast – during
a split of second it “shoots” to flat toad the larva, and simultaneously throws
out a drop of sticky secretions which help the larva to attach to the body of
host animal. Flat leechfly is rather large insect: the length of adult individual
makes about 15 mm. It has massive constitution (the specific name is determined
exclusively by the body shape of larva) and heavy slow flight. Therefore adult
flat leechflies prefer to lead a secretive way of life. Bodies of imagoes of
both genders are ruby-red with metal shine, and eyes are bordered by thin silvery
strips. The same strip stretches along the abdomen. At males of this species
wings are almost entirely black; only back edge of wing is transparent. Imagoes
of this species are active in twilight and eat pulp of rotting fruits.
Fourseas
leechfly (Aquabdellomusca tetramarina) is found in thickets of reed
and giant cat's tail at the coast of Fourseas, and is especially numerous at
the southern coast and at Caucasian Peninsula. This species is a polyphage;
larva feeds on various birds and frogs. By habit of life the larva of this species
considerably differs from other species. At the majority of species of leechflies
the female by the behaviour provides contact of larva and host animal. At Fourseas
leechfly larva leads mobile way of life and searches for the host animal itself.
It waits for approach of suitable host, having hidden in leaf axils of marsh
plants like ground leeches of tropical forest. Larvae of Fourseas leechfly are
rather large. During the life the female can give birth up to 50 such larvae.
It leaves them in leaf axils of huge cat's tail where the small amount of water
gathers.
Adult Fourseas leechfly feeds on corpses of various animals. It is rather easy
to define because of bronze colouring of body with reddish metal shine, and
of golden-yellow eyes with tiny black points. Male and female at this species
are similar to each other, and differ only in colouring wings – at male they
are darker.
The idea about existence of present group of insects is proposed by Nem, the forum member.
Wasp
Louse (Vespobraula parasitica)
Order: Two-winged flies (Diptera)
Family: Bee Lice (Braulidae)
Habitat: New Zealand, colonies of Neovespula wasps.
The Neovespula wasp colonies have invited
a whole host of parasites and commensals, not the least, the Wasp Louse, a relative
of Bee Lice (Braula). Their ancestors were brought along with honeybees during
colonisation, but this kind was able to become a parasite of wasp colonies.
Individuals may be found congregating on apple trees, where they may be brought
back to the nest. Individuals are tiny, about 1.5mm long, their bodies are round
and truncated, wingless, with relatively long limbs which allow them to clamber
over their hosts. Adults feed by roaming around the mouths of the wasps, feeding
on their mouth secretions. They prefer the mouths of queens, as well as workers
of Civil wasp species, and they will rush to cluster around the mouth. Large
infestations of these on any one individual may cause irritation. Wasp lice
breed year round, and their abundance is linked to the health of the colony,
and an overly large infestation may negatively affect the fecundity and health
of the wasps. Eggs are laid in large casings close to the combs of developing
wasp larvae, and the newly hatched wasp lice larvae will feed directly from
the mouths of wasp larvae and their attendants, sometimes negatively affecting
the health of the developing wasp.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Golden
dorsobia (Dorsobia chrysaea)
Order: Two-winged flies (Diptera)
Family: Flesh flies (Calliphoridae)
Habitat: Eastern Asia, swamp forests of China.

Picture by Biolog
Insects had firmly kept place in low-size class of inhabitants
of the Earth; they had easily gone through epoch of anthropogenous crisis. The
most vulnerable and highly specialized species had died out only, but as a whole
a specific variety of insects had decreased very unsignificantly. In early Neocene,
when ecosystems began to restore again, insects had quickly occupied new habitats.
The new species, adapted in the best way to their inhabitancy, had evolved;
in stable conditions a plenty of highly specialized forms had appeared. Occurrence
of new species of vertebrates had caused a new round of evolution of parasitic
insects. The new species of them had appeared, using the most unusual sources
of food.
In swamp forests of China the large species of amphibians had evolved – giant
mossy tree frog (Bryodorsa titan). In due course of evolution this amphibian
had developed the original form of care of posterity – male of this species
bears clutch of eggs on back, among numerous skin outgrowths. When from eggs
tadpoles hatch, it brings them up with nutritious skin secretions and carries
on itself, while they will reach the needed size.
Male can protect the posterity from drying, famine and medium-sized predators
like water beetles and small fishes. But the posterity of this amphibian can
not feel like in full safety.
Male of giant mossy tree frog carrying its clutch may become an object of steadfast
attention of females of one species of insects. Small (4-5 mm long) fly of golden-yellow
color with brown eyes and spherical abdomen persistently follows the amphibian
male surpassing it in thousand times in weight. Having chosen a right moment,
the fly lands on its back among eggs and begins laying eggs one by one in various
places of clutch. In some hours from eggs worm-like larvae burst. Their first
food is slimy secretions on skin of this tree frog, that provide connection
of eggs and surface of male’s skin. While larvae are small, they do not harm
to eggs, because they feed as commensals. Obviously, it was a first step in
the adaptation to parasitic way of life. When tadpoles hatch, male’s skin begins
producing of more nutritious slime containing proteins and a small amount of
carbohydrates. Eating such secretions, larvae begin to grow quickly and in two
days reach the length of 6 – 8 mm. After the next moult they start to attack
tadpoles and to eat them. The enzymes produced by larvae of golden dorsobia
corrode skin of tadpoles and cause occurrence of wounds in them. Larvae of the
fly, feeling a smell of blood, creep to wounded tadpole and eat it. The dead
tadpole falls off from the back of the parent soon, and fly larvae attack the
next one. If larvae got to the body of the dead tadpole, they can finish their
development, feeding on its meat. If the dead tadpole had fallen in water, the
larvae kept on it perish. During the time of clutch bearing by the male of giant
mossy tree frog some individuals of this fly can lay eggs on it. Larvae do not
attack against each other, and on the infected tree frog male it is possible
to find the larvae being at completely different stages of development. As a
result the activity of larvae of golden dorsobia may destroy up to the half
of litter of the amphibian. Having finished their development, larvae leave
the host and pupate in layer of soft vegetative dust and in cracks of tree bark.
Males of golden dorsobia differ from females in narrower body and large eyes
adjoining each other at the most part of head. Adult insects eat tree sap and
flower nectar. After pairing the female can make up to three clutches.
Laurel
and oak gallfly (Oncofaber myrmecophilus)
Order: Two-winged flies (Diptera)
Family: Gallflies (Cecidomyidae)
Habitat: dry forests in mountain areas of the Mediterranean area.

Picture by Biolog
Gallflies represent the separate family of plant-parasitizing
Diptera. In human epoch their species had been listed as agriculture pests,
and many species of them simply lived near to people, feeding on decorative
plants and in park zone of cities. Therefore the human epoch had passed absolutely
imperceptibly for many species of them, and the specific variety of this group
was kept in general. Restoration of the large forests broken by people and the
climatic changes, connected to movement of continents, had caused the redistribution
of borders of natural zones in Neocene, and it had resulted in changes of fauna
of these plant pest insects.
After the drying of Mediterranean Sea the climate of Southern Europe became
very dry and hot. In the Mediterranean hollow the remarkable communities of
plants steady to salinization of soil and the lack of water had formed. And
in mountains, where the conditions are more humid, forests of drought-resistant
trees are formed, mainly of oaks and laurel trees of various species. On these
plants the tiny insect from the number of gallflies parasitizes – laurel and
oak gallfly. This species had developed the special form of relations with small
species of gall ants living on
trees. The coevolution has resulted in changes of shape and life cycle of these
insects.
Larvae of this species, blind worm-like creatures about 3 mm long, settle on
young shoots of trees. Their secretions cause ugly growth of branch with formation
of large galls with complex internal structure. The gall represents a chain
of 4 – 8 apple-sized growths, covered with tiny leaves randomly scattered on
their surface keeping the typical form of leaves of the host plant. Inside each
gall there is true circuit of cavities of different forms and sizes connected
to each other by apertures. Larvae live in external layer of galls, under its
strong environment. Influence of chemical substances, secreted by larvae, forces
the plant to produce rich in starch and proteins parenchymatose cells on walls
of some chambers of galls. Congestions of such tissue form some kind of “ant
bread”, which is used for feeding of gall ants. Activity of larvae of laurel
and oak gallfly provides both firm dwelling and feed for ants. From their side
ants protect galls and their inhabitants from predators and parasites.
The galls, formed by this insect, exist for some years. For the second year
of existence of the gall on its surface some shoots sprout. Ant ergates infect
them with larvae, and the gall expands further. Larvae of gallfly breed under
their protection. Even in human epoch the phenomenon of pedogenesis was known
for them, at which the larva produces new generation of larvae without fertilization,
not passing to adult condition. These new larvae develop in the body of parental
individual, break it off and eat it; further they continue to lead the same
life, as their mother did. Ants carefully watch for them in order to prevent
the overpopulation among larvae. If necessary they transfer a part of young
larvae to new branches where new galls begin forming soon.
Adult males of this species are very similar to other gallflies. These ones
are tiny winged insects similar to mosquitoes of about 1 millimeter long, of
citreous color with shining black head. Antennae of males are long; their segments
are thickened, and tips of antennae have glands. They produce the odorous substance
calming ants greatly. Due to such “passing” males penetrate freely in the galls
inhabited with ants in searches of females, even if they carry on their bodies
a smell of another colony of ants. After the metamorphosis males leave the colony,
and ants do not prevent them in it.
Females passed the metamorphosis strictly differ from males. They are larger
– up to 3 millimeters long, with thick, almost spherical abdomen. Their antennae
are short, but wide, and also are supplied with glands, producing the odorous
substance forcing ants to behave to them gently and carefully. Females lack
of wings, but have normally advanced chitinous cover. Ants care of them: transfer
females with hardened covers to special chambers where they can feed on “ant
bread”, and where males flied from other colonies can easily find them. As required
ergates drag off the fertilized gallfly females to other branches, where females
lay eggs. The part of females lays eggs on the same branch where they were born,
on shoots atop of old galls, “attaching” the new gall in younger part of the
shoot. After laying of eggs females die.
During the flight of ants the settling of fertilized females takes place – ant
females carry away them to courtship flight. One ant female may carry up to
three fertilized gallfly females, which keep for its legs. The fertilized ant
female finds the tree of suitable species and puts on it the gallfly female.
The female lays eggs, and in one day larvae burst. They start to change the
shoot of plant with their secretions, forming the gall for new colony of ants.
False-flowermaker (Nanofloricultor pseudanthoformis)
Order: Two-winged flies (Diptera)
Family: Gallflies (Cecidomyidae)
Habitat: tropical forests of Southeast Asia.
Among insects in due course of evolution the special ecological group of gall-producing
insects was developed. Species of the insects belonging to different orders
in due course of evolution had developed skill of changing of character of
growth of plants with the help of secretions, causing at them formation of
galls of various appearance and size. Frequently galls represent excrescences,
but sometimes they may look completely unusually – like leaves and flowers,
but not of such appearance that are formed in typical case on parent plant.
One of tops in art of gall-producing belongs to false-flowermaker living in
Southeast Asia. This insect lives in plants of Bignoniaceae family. Usually
fodder plants of this insect represent lianas forming the inflorescences consisting
of large tubular flowers of bright colouring, pollinated by birds and large
butterflies. But under the influence of secretions of parasitic insect the
character of growth of the plant changes. On young shoot the small swelling
is formed, in which the larva – worm-like creature up to 6 – 8 mm long – originally
lives. This larva never turns to adult individual – its life task includes
breeding only. Its secretions result in gradual formation of spherical thickening
on stalk of the plant. It is covered with thin elastic bark, under which some
buds are forming. One of these buds sprouts and turns to similarity of the
reduced flower – narrow green tube on which internal surface tubercles grow,
appropriating approximately to stamens of a normal flower. Occasionally a small
amount of sterile pollen appears on them. Edges of this flower are surrounded
with fleshy blades of brown color – in cells chlorophyll and anthocyan are
generated simultaneously. In depth of this “flower” a certain similarity of
a nectary develops – the gland producing smell involving males of false-flowermaker.
This adaptation develops for attraction of the male which penetrates through
this flower inside the gall and fertilizes the female larva. Male of this species
represents a tiny winged insect of lacquer-black color with white-and-yellow
head. Its total length is less than 2 millimeters. Having coupled with the
female larva it perishes and its body stays in gall cavity. Frequently the
larva devours its body.
After fertilization the female larva gradually changes and turns similar to
any nematode worm: its body grows fat strongly because of forming eggs which
number may be up to 200 – 300 ones. The complete portion of eggs ripens simultaneously;
and the female larva distributes them on internal surface of its dwelling.
Having laid the last egg, it perishes. As it stops the producing of the substances
stimulating the formation of “flower”, it dies off and the entrance to the
gall cavity is closed with a fuse of friable parenchymatose tissue. Larvae
of the second generation have other destiny: they turn to imagoes, to normal
winged males and wingless immovable females capable to independent movement
at short distance. Females of false-flowermaker have the inflated abdomen and
short legs, and also firm ovipositor. They are capable to creep on branches
in searches of suitable place for egg laying.
In due course of growth of larvae of new generation the buds generated by activity
of their mother gradually germinate. Buds develop to hypertrophied similarity
of flowerbuds. When larvae turn to females and males, these “flowers” start
blossoming. In outward appearance they resemble normal flowers of the fodder
plant, but surpass them in size. At first from these “flowers” males fly out,
searching for new females to fertilize them. Then females creep out from the
gall. Their abilities to independent moving are very limited; therefore they
apply another way of settling. Females stay in depth of “flowers”, waiting
for animals which will undertake function of pollinators. In the tube of such
“flower” gall nectaries develop, and bright colouring of perianth involves
insects and small nectarivorous birds. In searches of nectar beaks of birds
and proboscises of butterflies penetrate deeply into the refuge of females
which cling to them. Because of their tiny size birds and insects do not notice
such “passengers” and carry females to new plants. Females lay under thin skin
of plant shoots parthenogenetic eggs, from which the larvae burst, penetrate
into the tissues of the plant and form galls.
Greenish
Glow-Worm (Arachnocampa semiviridis)
Order: Flies (Diptera)
Family: Fungus Gnats (Keroplatidae)
Habitat: New Zealand, the depths of caves, cavities under ledges and riverbanks
in humid forests.
A rather famous insect inhabitant of New Zealand was the Waitomo Glow-worm (Arachnocampa
luminosa), famously creating great underground light shows with their luminescence,
which were popular with tourists. This species has descended into the Neocene
without mishap, resulting in the Greenish Glow-Worm.
This gnat generally greatly resembles its ancestor, being a mid-sized kind of
fly with long, spidery legs and an overall light brown colour. Larva is a worm-like
grub that measures up to 4 centimeters long, but is usually obscured by the
nest which it makes it of strands of silk and mucus.
Larva hunts by suspending up to 30 long silk threads from the ceiling of the
cave, which are dotted with drops of extremely sticky mucus, serving to trap
flying insects that have ventured into the cave. Victims are enticed towards
this trap by the bioluminescence produced by this worm, a bright, greenish-blue
light which glows very intensely, produced by an organ at its rear end. Large
groups of these insects in a dark cave resemble a starlit sky at night. When
an insect becomes caught in a strand, the worm pulls in the strand and consumes
the prey.
Adults, pupa and larvae all emit light in some form, and it is by this light
that the male gnat finds the female in order to mate while they are still outside
during the night. In the spring, when gravid, the females fly into the caves
and deposit about 100 eggs directly onto the wall of the cave, which take 20
days to hatch. Upon hatching, larvae are only half a centimetre long and immediately
glow. They begin searching for a good place on the ceiling in order to produce
their nest and begin hunting, feeding larval stage lasts up to one year. After
shedding its skin five times it will spin a silken pupa within its nest; pupal
phase lasts about two weeks. Adults are relatively poor fliers, and are ephemeral,
living for only 70 to 90 hours, in which they must find each other and mate,
adults do not eat. Predators include cave dwelling harvestman, beetles, and
parasitic fungi.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Southern
Spotted Mosquito (Antipoaedes pardus)
Order: Flies and Mosquitoes (Diptera)
Family: Mosquitos (Culicidae)
Habitat: Ubiquitous across warm habitats in Meganesia and the Northern part
of New Zealand, various south-Pacific islands, requires standing water to breed.
In the Neocene, as it was in the age of man, various parasitic sanguiphages
actively seek to feed on the blood of warm blooded animals. In the south-Pacific,
including Meganesia and New Zealand, the most common mosquito is the Southern
Spotted Mosquito, which is descended from the species Aedes notoscriptus.
This species is functionally very similar to its ancestor, a mid-sized mosquito
whose head and body reach lengths of about 7 millimetres. It is shaped very
similarly to its ancestor, though the white spots that adorn its blackish-colored
body smaller and more distinct. Its long feathery legs are banded near the joints
with white, also.
Both males and females shelter in shady areas of cover during the day, usually
among the branches of trees and shrubs. Females actively search for blood, and
bite their hosts during the early evening, and early morning, crepuscular behavior,
males feed on the nectar of different flowers. The most common victims of the
blood-sucking females are mammals and birds, both small and large. Females may
bite and consume blood by day in shady or dark areas like caves and forest glades,
caves that contain large amounts of roosting bats usually attract many of these
insects to suck the blood of both adults and their young.
Eggs are laid in puddles or ponds of still water which are adjacent to dense
vegetation, a female may lay up to 100 eggs at a time. Breeding is more frequent
and more abundant in the warmer months, from October to March. Eggs hatch a
few days after being laid in water, young are aquatic, and will pupate within
as little as 5 days. Eggs can survive drying out, in the event that their nesting
site evaporates, they can remain this way for up to 8 months. Females are ready
to breed about a day after reaching adulthood, a single female can produce up
to 100 broods of eggs in her lifetime, lifespan is generally less than 30 days.
Predators include various birds, predatory flying insects and many kinds of
night-flying small bat, larvae may be consumed by aquatic insects and their
young, as well as small fish and tadpoles.
This insect is able to carry and transmit various dangerous diseases and internal
parasites, and may spread them by biting multiple individuals.
This insect species was discovered by Timothy Donald Morris, Adelaide, Australia.
Millipedes
|
Tear
gas millipede (Lacrimacausa irritans)
Order: Snake millipedes (Juliformia)
Family: Snake millipedes (Julidae)
Habitat: Great Antigua, underbrush of rainforest.

Picture by Biolog
Millipedes are among the most ancient terrestrial animals
of the Earth. Having evolved in Devonian, they had safely gone through the
time of human domination, and in Neocene had kept their positions in ecosystems.
The main protective weapon of millipedes includes various chemical substances.
Some of them have strong poison and inject it in wounds during the sting.
Others, escaping from the enemy, secret odorous substances with addition of
hydrocianic acid vapor. Third ones leave a trace of the caustic substances
irritating skin.
In Neocene one of millipedes living at the island Great Antigua, had developed
even more effective way of protection against enemies. This species named
tear gas millipede lives in underbrush of rainforest. When this millipede
feels danger, it emits a mix of easily evaporating substances which influences
as tear gas (its Latin name literally means “the reason of crying”) and strongly
irritates mucous membranes of predator.
Presence of such way of protection is very useful for tear gas millipede –
it does not have other ways of protection against enemies. By the nature it
is peaceful sluggish animal. The length of tear gas millipede reaches 20 cm,
and thickness of cylindrical body is up to 15 mm. In its body it is totaled
60 segments. This species lives on the ground, and only if necessary can dig
in rotten foliage. Occasionally the tear gas millipede gets on trees by thick
stalks of lianas.
Tear gas millipede is herbivorous one; its diet mostly consists of fallen
leaves and mushrooms. However, tear gas millipede eats snails and carrion
equally willingly. This animal belongs to large group of snake millipedes,
and has practically all their typical features. Though on each body segment
except for first three ones at tear gas millipede there are two pairs legs
(it is the common feature of snake millipedes), it can not escape from the
enemy by flight – legs are too short. But it is no need in speed because the
chemical protection of this millipede is very reliable. It is supported with
bright orange colouring of body, and across the body on each segment the strip
of silvery color passes, that makes tear gas millipede more beautiful and
appreciable from apart. On back end of its body one more protective adaptation
is located: two terminal segments of body are colored black with two white
points on sides. They form “false head”, which distracts attention of predator
from the true head of tear gas millipede.
This species of millipedes breeds the year round. The female lays eggs by
small portions of 20 – 30 ones. For each laying it builds a special nest.
For this purpose the female digs small jug-like hole by the back end of body.
It loosens the ground by legs and densely stamps walls of nest. Thus special
secretions from glands on one of segments cement the ground and disinfect
an internal part of nest.
From eggs larvae with small amount of legs hatch. First weeks of life they
eat only mycelium of mushrooms, later start to eat foliage processed by fungi,
and only after reaching of about one third of length of adult individual begin
to eat just fallen leaves of trees, rich in cellulose.
Tiger
millipede (Bradyjulius rubro-nigrum)
Order: Snake millipedes (Juliformia)
Family: Snake millipedes (Julidae)
Habitat: humid forests of Japan Islands.

Picture by Biolog
Initial picture by Tony Johnes |
Warming in Neocene had caused expansion of area of tropical
climate at the Earth. The abundance of food and rather equal temperature conditions
favoured to occurrence of large species with long cycle of development among
Neocene invertebrates. Warming had caused the development of humid tropical
forests at the southeast of Japan Islands, at mountain slopes, open to winds
from Pacific Ocean. As in any tropical forest, in wood litter of humid forests
of Japan Islands the set of invertebrates lives – beetles, cockroaches and
large snails. Also the largest ground invertebrate at the islands, large millipede
named tiger millipede, lives here. This is an animal with cylindrical body
up to 20 cm long and about 3 cm thick. In body of this millipede it is about
100 segments.
Despite of large size, this animal does not represent danger to other inhabitants
of underbrush. It is a harmless species of ground invertebrates, the exclusive
vegetarian eating mushrooms and fallen foliage. The body of tiger millipede
is covered with strong armour. Wide and flat head helps it to dig in ground.
Such feature helps this millipede to dig out underground fruit bodies of truffle
mushrooms, which tiger millipede can sense through the 10 – 15-centimetric
layer of ground. Eating mushrooms, this millipede carries their spores. Also
tiger millipede can eat tubers and bulbs of various grassy plants. But all
the same in its diet fallen leaves of various plants prevail. These millipeds
have an important role in processes of decomposition of organic substances
in forest.
This millipede has short antennae, developing under the bottom part of head
during ground digging, and very small eyes. Eyesight at this species is very
weak: tiger millipede confidently distinguishes only light and darkness, and
can distinguish movement of large objects around of it. However, it is not
required to it of sharp sight – food is always present near it and does not
disappear anywhere. And tiger millipede is reliably protected from predators,
though is not able to bite at all. Weak mandibles are a common feature of
snake millipedes, and it is not an exception.
This large millipede is difficult for not noticing in forest: it has very
bright colouring seen from apart. Tiger millipede has red body with cross
black strips – one stripe on each segment. It is warning colouring, and any
predator, had not stopped the attack in time, in the full can test all abilities
of protection of this millipede.
Under attack tiger millipede instinctively turns body off as a spiral, having
hidden head in coils of body. It already should become the warning for predator,
because after it this animal will involve the second line of defense. Tiger
millipede is poisonous, and, being disturbed, lets out easily evaporating
liquid with the unpleasant smell, containing a plenty of formaldehyde and
organic acids. Having got on skin of aggressor, this liquid causes strong
inflammation, which lasts till some days, but seldom comes to an end in fatal
outcome. Usually only frogs and toads die from the poison of tiger millipede,
because their naked skin is very sensitive to its secretions. Due to the effective
chemical weapon of this millipede, there are some imitators at tiger millipede
– one small species of snakes
and large ground flatworm. As they
also are protected, though weaker, than this one, these three species form
a “ring”, and represent a good example of Mullerian mimicry.
This species of millipedes arranges spectacular courtship rituals. For breeding
a plenty of these millipedes gathers. The congestion may occupy up to hundred
square meters, and on each square meter it may be up to two hundreds of animals.
All of them emit a specific sharp smell which is felt from the large distance.
In such congestions individuals of opposite genders find each other and are
coupled within several hours at night. It is interesting, that smell emitting
by these millipedes involves some local birds very much. Breeding congestions
of this millipede are frequently visited by local bird forest
ronin (Roninornis mortifer). These birds usually aggressively relating
to congeners do not pay attention to each other, sitting among creeping millipedes.
They fluff feathers and try to come nearer to the greater congestion of tiger
millipedes. Obviously, birds in such way get rid of parasites living in their
plumage.
The female lays eggs, having dug by back part of body in friable ground near
roots of tree. In clutch of this species there is up to 50 large eggs, and
such clutches may repeat each 2 weeks. The young animal reaches the size of
adult individual only at the second year of life.
Arachnids
|
South
American aquatic scorpion (Potamoscorpio aquaticus)
Order: Scorpions (Scorpionida)
Family: Botriurids (Bothriuridae)
Habitat: freshwater reservoirs in tropical part of South America.
Scorpions belong to oldest representatives of arthropods and arachnids in
particular; they have appeared at the world scene of life in Palaeozoic era
(Silurian and Devonian epochs). The first representatives of scorpions were
underwater animals, but later they settled to the land and have mastered the
new way of life so successfully that they have outlived their underwater relatives
by more than 300 million years.
In Neocene several kinds of scorpions returned back to aquatic lifestyle,
taking over the ecological niches vacated after the climatic disasters and
anthropogenic impact. South American river scorpion belongs to largest species
of secondary aquatic scorpions and of arthropods in general; it reaches a
length of 27-30 cm from tips of its massive claws to the sting at the tip
of its abdomen. Being covered with thick dark armor of olive color, it’s too
heavy to swim, so this creature just creeps on river bottom in search of various
invertebrates, carrion and wounded fish or amphibians; its kills prey with
its venomous sting.
Being an underwater inhabitant, aquatic scorpion still depends on atmosphere.
It breathes not with gills already lost by its Paleozoic ancestors, but with
lungs, which had increased relatively to the size and weight of its and allow
it breathing under water and on land, when the scorpion crawls from one reservoir
to another during the dry season in search of shelter and prey. Perhaps, the
adaptation to aquatic lifestyle was a response of terrestrial creature to
the periodic flooding of the forests in tropics of South America.
To breathe South American river scorpion crawls to the surface of water on
stalks of aquatic plants. If necessary, it can swim a short distance, moving
its limbs intensively: this scorpion emerges for breath from the depth of
1 meter and more and clings to aquatic plants to get to the surface. The lower
part of abdomen, where the respiratory holes open, is covered with dense water-resistant
hair and under water the air layer covers it, giving the silvery sheen to
animal’s underside.
The main enemies of aquatic scorpion are some large spiders, scorpions and
army ants able to overpower it, as well as various microbes and fungi that
destroy its organism from inside. Sometimes these scorpions are attacked by
large fishes, and during the moving on land it is vulnerable to crabs and
shorebirds.
With the onset of rain season aquatic scorpions turn paler in color and much
more visible to each other; at this time females and males of this species
search for each other to couple. They mate during the bizarre mating dance,
like many other representatives of this arachnid subclass, but the dance and
the transferring of spermatophore occur under water. After mating male and
female remain together for some time, hiding in burrows excavated near each
other in coastal mud. This species is ovoviviparious; female protects the
posterity carrying youngs on its back. During this time the female stays on
dry land and eats almost of nothing. Young scorpions leave their mother soon
and prefer to stay alone, hiding among floating plants or in the coastal thickets.
During this time cannibalism is common among them: the juveniles often fall
victims of the adult representatives of this species. They become fully mature
at the age of about two years.
The average life expectancy of South American aquatic scorpion is quite big:
up to 17-20 years.
This species of arachnids is discovered by Bhut, the forum
member.
Translated by Bhut.
Four-legged
net spider (Tetrapodarachna tetramana)
Order: Spiders (Araneae)
Family: Orb-web spiders (Araneidae)
Habitat: rain tropical forests of South-Eastern Asia, rainforest canopy.

Picture by FanboyPhilosopher
During the evolutional history spiders had developed various
ways of prey trapping: from simple chasing up to web traps reaching sometimes
a high degree of refinement in design. The majority of spiders trap catch,
having stretched web constructions among branches. But some spiders hunt by
special way, combining features of hunting spiders and spider web builders.
The Neocene inhabitant of woods of South-Eastern Asia, the representative
of orb-web spiders, four-legged net spider is those.
It is a large spider: the length of body is about 6 – 7 cm from which big
rounded abdomen accounts about 60 per cent. The cephalothorax is wide, in
its forward part there is an eminence on which eyes are located: two large
shining eyes look forward, and six ones provide the circular field of view.
The cephalothorax is covered with rich hairs of black color. Abdomen is cross-striped,
“wasp-colored”: on black background there are yellow cross strips. Legs are
light green with longitudinal black strips on top side.
Woods of South-Eastern Asia differ in changeable weather. Here is rainy every
day, and rain is accompanied with strong winds from ocean. Therefore four-legged
net spider does not arrange huge nets: they may be frequently torn by wind.
This representative of arachnids has no constant shelter: it hunts, wandering
over branches and lianas of forest canopy in searches of catch, similarly
to representatives of hunting spiders of family Lycosidae. Each shelter happens
occupied in succession of some days, and then spider leaves it. But this species
of spiders also is not stopped weaving web, though it makes it extremely originally.
Legs of four-legged net spider are of different length: first two pairs are
very long, approximately twice exceeding length of cephalothorax and abdomen
taken together. Two back pairs of legs are much shorter, only one and half
times longer than the body of spider. Such structure is connected to features
of web weaving. For this purpose the spider rises on two forward pairs of
legs, and begins making of web on tips of two back pairs of legs. In the beginning
it connects claws of back pairs of legs by basis strings, then stretches between
them radial basic strings, and braids them by sticky circular string. The
web of characteristic circular form, by length and width approximately equal
to body of spider turns out as a result. When it is ready, spider gradually
takes its corners by long legs of forward two pairs, changing them one by
one. At the end of this operation it stands on two back pairs of legs. When
the web appears stretched on long forward legs, it is ready for hunting.
The hunting spider moves on two pairs legs, making short dashes and jumps.
The web is always ready to prey catching: the spider holds it above eyes,
having stretched asides. Having noticed suitable catch (any small animal from
cockroach up to tiny lizard), the spider drives it by jumps, or simply moves
down from above on web string, and covers catch by web.
The female of this spider looks and behaves so. The male of four-legged net
spider is much smaller than the female similarly to males of the majority
of spiders. This creature is more similar to large ant - at it there is narrow
cephalothorax colored black. Such coloring combined with the body shape gives
him quite good chances of escape from insectivorous birds. For improvement
of masking it holds up a forward pair of legs put up before head, imitating
ant head.
In courtship season the male should be especially cautious: if it will approach
to the female too close without the appropriate ritual, she simply will “swaddle”
him by web, and will have eaten. Therefore ritual of acquaintance at this
spider begins from very cautious acquaintance of the future parental pair.
The male defines shelter of the female by smell, cautiously sneaks to it,
and leaves odorous labels. Just in case he keeps near suitable shelters, ready
to hide at the first threat from the side of female. But gradually the female
gets used to his presence, and once the male begins courtship dance near her
dwelling. During this ritual the male imitates the actions made at prey capture.
Only he holds simply plant leaf or the bird feather in two forward pairs of
legs instead of web. The male “covers” by this object an imagined prey, but
does not finish action, being limited only by “prey capture”. If the female
does not show aggression, the male “presents” her with this object, and while
the female holds it in cheliceras, quickly couples with her. After that the
male occasionally succeeds to escape, but more often the female eats him.
The female of net spider lays up to 200 eggs simultaneously. She keeps them
in web cocoon, carefully clearing it from dust and preserving from damages.
Approximately in ten days the posterity hatches from eggs. Young spiders first
days “ripe” in cocoon then leave it and get over on back of the female. Having
got stronger, they start to lead independent life. First time young spiders
hunt plant lice and small termites, and then start to attack larger and better
protected insects: ants and beetles.
Mangrove
fishing spider (Arachnopiscatorius mangrophilus)
Order: Spiders (Araneae)
Family: Orb-web spiders (Araneidae)
Habitat: Meganesia, mangrove thickets of Eyre Gulf.
Among spiders there are only few species leading completely aquatic habit of
life – only separate species do it. For comparison, among insects representatives
of several orders had developed aquatic inhabitancy, and they spend in water
whole life, or any stage of development (usually larval one). However, the significant
amount of species of spiders leads near-water habit of life, and even has skills
of catching of water animals. One of such spiders lives in thickets of marsh
plants of Eyre Gulf in Meganesia.
Eyre Gulf represents shallow-water reservoir with variable salinity of water
in which action of ocean inflow is felt. Two times per day water of gulf directs
to the ocean over sandy shallows in mouth, and two times in day it is replaced
with tidal wave from ocean. As a result the water level in gulf strongly fluctuates.
Rivers drift in some places of gulf a circuit of small sandy shallows and islets
separating from gulf extensive sites of quieter water. At these shallows original
“mangrove forests” made mainly of Pandanus trees grow. Among sites of such forest
various animals find shelter and food – birds, insects, mollusks and other animals.
One inhabitant of Pandanus thickets in Eyre Gulf is a large spider of reddish-brown
color with white spots on abdomen. The length of its massive abdomen reaches
4 cm, and leg span exceeds 15 cm. It is mangrove fishing spider which has adapted
to inhabiting in changeable world of mangrove forest.
Mangrove fishing spider is very poisonous animal – its sting may kill pig-sized
animal in some minutes. Signaling about presence of such strong weapon, mangrove
fishing spider has developed very bright and appreciable colouring. All legs
of this spider have cross-striped colouring of wide brown and snow-white strips.
On reddish-brown abdomen of spider white strokes form a rough border, and along
the middle line of abdomen the strip of white round spots is stretched. The cephalothorax
of this spider colored white is covered with short velvety hair; pedipalps and
killing cheliceras have black colouring. The body of spider is covered with water-repellent
hair, which keep air layer on abdomen and permit the spider to breathe any time,
having plunged in water. Legs of forward pair are very long – their length is
up to 10 cm.
Eyes at this spider are large and shine green light in darkness.
Mangrove fishing spider lives in thickets of Pandanus and keeps on border of
usual level of inflow. This spider is too heavy to be able to swim. Nevertheless,
its food is made of various water animals. This spider builds webs, having the
characteristic circular form, among trunks and roots of Pandanus and other plants.
It is engaged in construction and repair of web during the outflow time. The
web of mangrove fishing spider is strong and elastic enough to sustain rather
strong tidal wave. All way of hunting of this spider is designed for rough waves
of inflow and outflow. At this time in web prey gets – fish fry and small crustaceans,
crabs and shrimps. At hunting for such prey having slow metabolism it is very
important to kill it fast; the strong poison of spider is designed for it.
Web is plaiting so that spider has convenient place above it. If there is no
firm support, spider makes for itself small web of dry strings, forming similarity
of a hammock. During inflow and outflow mangrove fishing spider keeps front legs
on web, thus its body is outside of water. Having felt presence of prey, it slips
in water (approximately up to half of abdomen length) and seizes prey by front
pair of legs. It accurately breaks off strings of web keeping prey, and kills
it by sting. During the hunting spider keeps by one of back legs for strong “safety
cable” made of web.
Male at this species is lighter than female in some tens times, but it is equally
poisonous. The most part of time it leads the same way of life as the female,
but hunts smaller prey. In courtship season male finds the female and starts
cautious courting ritual. When female catches prey, male cautiously comes nearer
to web from the side and cautiously starts to pull web in special rhythm, warning
the female about its presence. When the female replies the male with a special
signal, he cautiously comes nearer to her, keeping in sight and observing a distance.
At the slightest sign of aggression from the part of the female male hides and
begins courting all over again.
Pairing at these spiders occurs quickly, but it is preceded with special preparation
– male at first ties legs of the female together by web. After pairing he has
some minutes to leave a web of female and to survive. But approximately in one
case from four it has not time to make it, and becomes a prey of the female.
The female lays about 200 eggs in large cocoon of strong web which she always
carries in back legs. For young spiders she makes a special “children’s” web
above the maximal inflow level, and feeds posterity, bringing prey to “children’s”
web. Young spiders quickly leave parental nest and settle, flying on web thread.
Many of them perish in waves of Eyre Gulf and become prey of fishes. But the
part of young spiders survives and reaches places, suitable for life. The first
months of life they hunt small invertebrates with the help of simple web traps,
and only having reached the size of approximately one third of the length of
adult individual, start to make underwater webs.
Tiny republican spider (Microsociarachne numerosus)
Order: Spiders (Araneae)
Family: Orb-web spiders (Araneidae)
Habitat: North America, areas of warm-temperate climate, deciduous forests.
Usually spiders are solitary hunters, and even relations of male and female
may be very strained. Occasionally male succeeds to tie the female by web
thread and to stay alive, but more often the female catches and eats him
after pairing. Among spiders social species represent rather exception, than
a rule. In Neocene among spiders there are some social species, which had
evolved independently from each other in various areas of the Earth, mainly
in tropics.
One of the most northern species of the spiders leading social way of life
is tiny republican spider inhabiting forests of North America. Length of
the female of this species is up to 9 mm including legs, and male is about
6 mm long. Colonies total up to 100000 individuals.
The appearance of tiny republican spider is typical for orb-web spiders.
It has no outgrowths, characteristic for some tropical spiders, on the body,
and body also has no rich hair cover. On the body and walking legs of this
spider only short tactile bristles are present. This spider has dim cryptic
coloration – grey with brown “marble” pattern. Males differ from females
not only in size, but also in more gracile constitution.
Presence of spiders of this species is easy for finding out: the tree chosen
by a colony of tiny republican spider may be entirely surrounded by spider
web. Trapping webs of this spider are circular, characteristic for orb-web
spiders. The diameter of a separate web may reach one meter. On each of such
webs some adult females of this species wait for prey. Webs are connected
with each other by separate alarm strings; therefore spiders from any web
receive the information about what is happening in the next web.
On one branch the center of colony is located – the common nest tailored
by web strings from live green leaves. These spiders are not specialized
to any certain kind of prey, and sometimes catch huge insects comparatively
to their own size, obviously preferring dipters and lepidopters. Large prey
is killed by stings of some females at once.
In colony of tiny republican spider females work mainly – their duties include
prey killing, and they also lay eggs from which the new generation of spiders
hatches. When the colony is normally reproduced, there is constantly a set
of “superfluous” females, which should not reach sexual maturity, because
their posterity may undermine a forage reserve of the colony. In order to
make it at such females last moult is delayed: pheromones secreting by mature
females have an influence on them. Outside of a colony “superfluous” females
can normally develop further, and their fertility does not differ from fertility
of usual females of the colony.
Males at tiny republican spider also live in a colony. Among them there are
the “homebodies” making a part of functions of colony members, and there
are also settling males, which leave a colony after last moult. Male do not
differ in any remarkable “labour activity”; they basically perform work on
patroling and local repair of the damaged web. It takes place at night when
danger of attack of birds and solitary wasps is less. Participation of males
in hunting is minimal: they do not kill the caught insects, but having found
large prey, they signal about it to females with the help of fluctuations
of web threads. Males also actively catch prey on periphery of the web where
it is more difficult to females to be kept.
Males in the majority also are exposed to pheromonal “taming”: they do not
have last moult. This phenomenon arises at simultaneous influence on the
male of pheromones of other mature males and the fertilized females. And
the individuals which succeed to moult (it takes place also at the high concentration
in atmosphere of the nest of pheromones of not impregnated females) remain
in native colony. If the competition between them is great, they become settlers.
“Tamed” males are defenders of colony from small enemies. They show hyperactivity
in struggle against ichneumons and the solitary spider-eating wasps. “Superfluous”
females struggle with larger enemies – at them poisonous glands are better
advanced.
In the nest there are the females bearing cocoons, and also young spiders.
They are looked after by the small “tamed” females. The tiny republican spider
has exchange of the food, similar the one characteristic for social insects.
Hunting individuals living on web are suppliers of food. Like all spiders,
tiny republican spiders inject in prey digestive enzymes which transform
prey’s tissues into “broth” which is sucked out by females then. Having filled
stomachs with food liquid, they move to nest. Here hunting females belch
a part of food for the females protecting clutch, and for males looking after
them and struggling with parasitic insects. Having received a portion of
the liquid forage, males share it with youngest spiders. Male going to feed
posterity shakes a web in a special rhythm, involving young spiders. When
they gather on his body, male hangs down on web and belches a drop of nutritious
liquid, keeping it in cheliceras and pedipalps, and young spiders simply
lick off this liquid.
Colonies of tiny republican spider multiply through “division” or “budding”:
the large colony is gradually divided to two or three independent ones, or
from it the small colony is separated, in which all types of males and females
are present. The settled not impregnated females followed by males are frequently
observed. Frequently males from other colonies meet such females. Moving
of young ones on spiderweb parachutes at this species is not present.
Colonies of tiny republican spider may be met on branches during the most
part of year when there is foliage on trees. These spiders winter in multilayered
web nest arranged in the abandoned hole of small mammals. For protection
against cold spiders make multilayered web “fuse”, which provides heat insulation.
From outside “fuse” is encrusted with lumps of the ground and the vegetative
rubbish. In spring the colony leaves winter shelter and builds the new nest
surrounded with trapping spider webs.
This species of spiders was discovered by Mutant, the forum member.
Bird-catching
spider (Avireticulum mortalis)
Order: Spiders (Araneae)
Family: Orb-web spiders (Araneidae)
Habitat: Central America, tropical forests.
In human epoch at studying of features of the spiders leading social way of
life, it was established, that adult spiders in small colonies tend to being
larger, than adult spiders in larger colonies. Among the Neocene spiders,
forming colonies, this dependence also is kept. One species of social spiders
of Neocene, tiny republican spider
(Microsociarachne numerosus) from North America, displays this phenomenon.
It is the small spider forming large colonies, numbering up to hundred thousand
individuals. To the south of it, in tropical areas of Central America, its
larger relative lives, displaying the opposite tendency – the bird-catching
spider.
The colony of the bird-catching spider totals no more than 20 adult females
– it is very little number in comparison with colonies of other spiders leading
a similar way of life. But each female in colony has length of body about
6 cm, and legspan reaches 20 cm. Female has lengthened cylindrical abdomen
and wide cephalothorax. The body of the female is colored black with white
spotty pattern on the bottom side of abdomen. Legs of the female are long
and thin, black with thin cross strips of white color in the basis. On upper
side of cephalothorax there is a white figure as a white irregular-shaped
spot. Cheliceras and pedipalps of the female are covered by large red spots.
It is a warning colouring – if necessary, the female of bird-catching spider
can attack an animal surpassing it in size manyfold.
In colony of bird-catching spider there are males also. They are very tiny
– body length of the adult male is about 10 mm. As against females males have
grayish-green camouflage colouring, and only pedipalps have white colouring.
Bird-catching spiders are active predators. The bird spiders (family Aviculariidae),
known in human epoch, were not the specialized bird hunters, despite of their
name received speculatively. As against them, bird-catching spider is true,
literal “bird-eater” in tropical forest canopy. Birds like small pigeon in
size sometimes fall prey of these spiders though usually they catch smaller
birds. Also these spiders eat small mammals, frogs, lizards and young snakes.
The sting of bird-catching spider is very poisonous – one sting is enough
to kill the hen-sized bird within one minute.
This spider builds the big web trap consisting of several circular spider
webs of regular shape made of thick sticky web threads. Spider webs are located
in different planes and cover a site of large tree branch. From them the alarm
strings stretch to the shelter where adult individuals hide. The shelter represents
a ball made of leaves stuck together by spider web threads. The compound plumose
leaf is especially well suitable for the arrangement of the shelter. Spiders
bend it and attach parts of leaf to each other. Gradually the whole leaf turns
to ball with narrow entrance chink, in which spiders hide during the rain
or at night.
Males of bird-catching spider are small and vulnerable, but it does not prevent
them to have strong poison, and to defend themselves successfully from small
predators. Males almost never play a role of hunters – all need for food is
provided by females, and males can catch only casual small prey. But males
clear bodies of females of parasites. Moreover females leave egg cocoons in
the common nest where males care of them. Males clear surface of cocoons of
dust and protect posterity. They kill and eat various parasitic flies and
ichneumons which constantly penetrate into the nest in order to lay eggs in
spider cocoons. Males also look after posterity, feeding up young spiders
with belched food. In order to use prey effectively at this spider feature
of behaviour, characteristic for termites and other social insects was developed
– an exchange of food. Gorged on female under the male’s request (a special
sort of touch to her cheliceras) belches liquid semidigested food which is
distributed by males among posterity of the colony.
Nurse
spider (Curarachne nutrix)
Order: Spiders (Araneae)
Family: Weaver Spider (Araneidae)
Habitat: tropical forests of the Far East (Asia Mainland and nearby islands).
Among the insects, there are many social species, and some taxa are represented
exclusively by social species. In contrast to insects, spiders relatively
uncommonly form colonies. But when an independent transition to a social way
of life occurs among spiders, a considerable variety of colony establishments
is found. Social spiders appeared during the Neocene era on different continents,
and populated predominantly the tropical belt.
In the tropical forests of the Far East one of the original social spiders,
nurse spider, is found. It is a representative of araneidae species, and it
forms a colony, numbering up to 500 individuals, most of whom are young and
small males. This species has pronounced sexual dimorphism. The female nurse
spider reaches length of 15 mm (leg span about 30 mm); it has a big round
abdomen. The coloration of an adult female is dark brown, with white leg tips
and pedipalps. A male’s length is approximately 5 mm (10 mm leg span); it
has a very catchy coloration: the upper part of the abdomen is bright red
with some white spots along the midline; the rest of the body is white, with
translucent legs. These males have very strong venom and in case of danger
they emerge on the web and even sit astride the females, defending them from
possible enemies. The bite of several males is enough to kill a small bird.
Young individuals remain greenish-white in colour for a long time.
The web of this species is a large structure, with a diameter of up to 3 meters.
Usually it is stretched among the branches and consists of several rather
regular circular nets from which individual solid web strands stretch to the
neighbouring branches. Insects, caught in the nets attract the attention of
young and sexually mature females, who attack it, bite it and partially suck
it out. Then the old females crawl onto the web, receiving a much larger proportion
of the nutrients from prey. Males and juveniles are also present on the net,
but keep out of the way, in shelters. Hunting females rarely gorge themselves
– usually they leave the prey, as it barely begins to feel the effect of the
digestive enzymes. The stated females creep into shelters and use the movements
of the legs on the net to send a special “feeding invitation” signal, in response
to which the juveniles and males climb on its body. The female belches out
a nutritious liquid, and young spiders and males feed on it. Also, the old
females distribute nutrients among young females, which are busy in caring
for the young.
In this species, despite the sharply pronounced sexual dimorphism, the female
is loyal to the male and does not eat it after mating. Sometimes during the
mating, the female is fed with “broth” burped out by a “nanny”, and it reduces
its aggressiveness. One clutch contains up to 200 small hatchlings, many of
which survive to independence. At the age of about 6 months they become sexually
mature. Life expectancy is about 2-3 years.
Translated by Bhut.
Amazon spider (Latrodectus bellicosus)
Order: Spiders (Araneae)
Family: Cobweb spiders (Theridiidae)
Habitat: subtropical and warm-temperate areas of Europe, Near East and Central
Asia; woody areas, bushes.
Among Neocene spiders some species leading social way of life had evolved.
Representatives of different groups of spiders passed to formation of colonies
independently from each other. Relations between individuals in colonies
of different species have different nature – from simple aspiration to live
near to relatives up to complex regulation of development of colony members
depending on condition of the whole colony.
In tropics and subtropics at the east of Europe, in Levant and Middle Asia
one species of spiders forming colonies lives – it is small, but aggressive
and very poisonous amazon spider. Length of body of this animal is about
15 mm; its close relative is Mediterranean black widow spider (Latrodectus
tredecimguttatus), known in human epoch.
Amazon spider is similar in shape to its ancestor, but differs from it in
colouring – its general colouring of body is black, and on abdomen there
is only one big red irregular-shaped spot surrounded with randomly scattered
white points. It is typical warning colouring – the spider is poisonous and
aggressive, and each individual can repulse a predator attacking the colony.
Male of this species is a small creature of grey color of about 5 mm long.
Colonies of amazon spider reach the different size depending on efficiency
of habitats. In droughty areas colonies of this species do not exceed 1000
individuals, and in subtropical forests the colonies numbering 10000 individuals
are not rare. Amazon spider leads strictly nocturnal way of life. At night
these spiders show hyperactivity and catch any prey of suitable size in radius
20 – 100 meters from the nest. Colonies of this species do not stay for a
long time in the chosen place: when amazon spiders exterminate all suitable
prey, they simply move to new area. The web at such colonies represents the
separate irregular-shaped pieces made among vegetation or between stones.
Stationary colonies in places, rich in food, have the whole system of web
traps among which there is carefully made nest in which all members of a
colony spend the daytime.
The sociality at amazon spider is advanced in lesser degree, rather than
at other social species of spiders. In particular, this spider hadn’t develop
the exchange of food between members of the colony and between adult individuals
and young growth. In habitats poor in food the colony is more similar to
a simple congestion of individuals. Spiders spend the daytime together in
the shelter plaited of web threads, encrusted with sand grains and vegetative
dust. At the exhaustion of food resources spiders easily abandon such shelter
and move to a new habitat in common.
In long-term colonies strongly advanced influence of pheromones to development
of individuals is expressed in full degree. In densely populated colony restriction
of birth rate is shown: superfluous (“not wanted”) females moult and reach
the typical size, but their ovaries remain undeveloped. Actually, in the
colony from 300 to 1000 breeding females exist, depending on the size of
a colony. Though in large colonies the quantitative gain is more, than in
smaller ones, it is counterbalanced with outflow of settling individuals.
Males also are exposed to chemical “taming”. For them a plenty of pheromones
of mature males and smell of egg cocoons are the factors of “taming” (pheromones
of fertilized females have lesser effect). Pheromones of not impregnated
females render on them the opposite effect – faster growth and maturing of
males. Braking of development of ovaries at superfluous females also takes
place under action of “egg pheromone” which is emitted by the females had
generated egg cocoons. Influence of smell of egg cocoons on females is much
stronger, rather than on males. Males have higher losses at moving and in
the colony of any type their number is less, than number of females. Any
individual can turn free of influence of chemical substances, leaving a colony.
Usually males act so, but the not impregnated females sometimes also leave
a colony.
Large colonies of amazon spider can separate freely to some independent colonies,
or from them settling groups are separated – small groups of individuals
of both genders, settling together to new habitats. The fertilized females
also sometimes move to the periphery of a colony, promoting its “budding”.
Amazon spider gives 1 – 2 generations per one year depending on temperature.
In areas of seasonal climate with approach of winter the colony moves to
the burrow of a rodent or to other shelter, covering its walls by silk.
This species of spiders was discovered by Mutant, the forum member.
Moehau
Spider (Macrodelena moehau)
Order: Spiders (Araneae)
Family: Huntsman Spiders (Sparassidae)
Habitat: North Island of New Zealand, forest, woods, bushland, dense forest.

Picture by Pavel Volkov
With any human occupation during the Holocene, invertebrates
of many kinds may find their way to live there as invasive pests, as stowaways
on boats or shipments of goods such as fruit or wood. And so it was that the
Flat Huntsman (Delena cancerides) that came to settle in the North Island
of New Zealand, with some infamy.
Among its descendants is the Moehau Spider, an analog of large tropical spiders
such as those found in Australia, named after hairy ogre-monster of Maori
mythology. Being larger than its ancestor, its body is overall bigger and
fatter, reaching 50 millimetres in body length, with a leg span of up to 20
centimetres Constitution is overall heavier and a little slower than its ancestor,
females are larger than males, and both sexes are quite hairy. Coloration
is dark brown on the cephalothorax and legs, with the ends of the legs being
darker, abdomen is covered in light grey hairs with a star-shaped dark pattern,
pedipalps, fangs and face are darker again, almost black.
These spiders hunt their food rather than spinning webs, catching catch mid
sized and large insects and other spiders, as well as small vertebrates such
as lizards, frogs, baby rodents and nestling birds. It moves slowly and tentatively
generally but strikes prey quickly, and can retreat quickly when revealed
to a predator. Generally shy, they are reluctant to bite unless seized by
a predator. When not active they usually hide under loose bark, fallen logs
or rocks.
Breeding occurs in the summer, from late November to mid February. The male
makes overtures to the female by circling her and caressing her abdomen, as
well as drumming his pedipalps against a flat surface to make a percussive
sound. After fertilisation, the female produces a large oval egg sac made
out of silk, in which are laid up to 200 large eggs. She then attaches it
to a hidden surface under a rock or tree bark, and guards it fiercely; incubation
varies by the conditions of climate that year from 3 to 4 weeks. When the
babies are ready to hatch, the mother moistens and tears open the sac to release
her young. Young may remain with their mother for a few weeks, until their
third moult generally. Lifespan of this spider is 2 to 3 years.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Heraldic
spider, shield-spider (Heraldarachne scutiferus)
Order: Spiders (Araneae)
Family: Wolf spiders (Lycosidae)
Habitat: Southern and South-Eastern Europe, Balkans, coast of Fourseas, Caucasus;
humid subtropical woods.
In Neocene when the significant warming of climate had taken place, borders
of natural zones had changed: the border of humid deciduous woods had moved
far to poles. It appeared especially appreciably in areas close to ocean and
seas while the central areas of Eurasia had remained a zone of sharply expressed
continental climate. In Europe the climate became warmer and humid due to
influence of Fourseas – huge brackish lake. It promoted the occurrence of
the zone of humid evergreen subtropical forests along western and southern
coasts of Fourseas. The fauna of these places is almost as rich, as in subtropics
of Holocene epoch. But it is formed mainly by descendants of European species
– from the south these woods are separated from Africa and South Asia by mountains,
and primal tropical groups of animals and plants are submitted in them by
separate species.
In warm tropics and subtropics arthropods are very diverse and often reach
large size. Among them as in the world of vertebrates, there are predators
and prey. Spiders are one of main predators among arthropods. One large species
of spiders, obviously the descendant of European wolf spiders (Lycosa), lives
in forests of Balkans. The length of its body is about 8 cm, and legspan may
reach about 12 cm. This creature lives on trunks and branches of trees, and
leads the vagrant habit of life, spending night in casual shelters. Its appearance
differs from habitual shape of spider: this is flat creature with tenacious
legs and very originally-shaped abdomen. The abdomen of this spider is expanded,
its top part is extended forward and hangs like cap peak above the cephalothorax.
The first line of abdominal shield is expanded and is bend a little. The abdomen
of animal at sight from above resembles the Medieval French knight’s shield,
and its coloring strengthens this similarity. For it the spider is named shield-spider,
or heraldic spider. At quietly creeping spider the forward part of cephalothorax
with eyes juts out from under “cap peak”, and animal has good circular field
of view. In case of danger animal completely hides under abdominal shield,
moving it on body from above. Thus due to cryptic colouring and shape spider
becomes indistinguishable from any outgrowth on tree bark. In case of need
heraldic spider can defend from enemies by stings. Its main enemies are birds
searching these spiders with the help of sight and simply pecking them having
pressed by paw against bark.
This spider has kept features characteristic for wolf spiders: it also does
not build a spiderweb, and catches prey, suddenly snatching on it by jump.
Its catch often appear moths spending day on tree trunk. Occasionally heraldic
spider catches even young frogs and lizards. Cheliceres of this animal are
long (up to half of cephalothorax length), their sting is poisonous and dangerous
to pigeon-sized birds. Heraldic spider has fine sight: it has two large forward
eyes giving the stereoimage of high quality. Four lateral eyes are small,
and the back pair of eyes is shifted upwards, enabling an animal to see everything
happening behind of it.
Legs and bottom part of body of heraldic spider are covered with hairs, and
top part of shieldis hairless with wrinkled surface – it imitates bark of
tree well. Colouring of shield of heraldic spider is cryptic: on brown background
there is a pattern of grey and yellowish spots and points, forming figures
approximally similar to heraldic figures. Under the armour this spider is
colored much brighter, rather than outside. The bottom surface of shield at
the male is covered with red hair reflecting light and giving to inner side
of shield silky shine. Bases of walking legs of the male are coal-black, and
legs are cross-striped black-and-white. At the female the inner side of shield
is colored red-brown color, and bases of striped legs are covered with hair
of bright blue color.
The male of heraldic spider is approximately 20% lighter, rather than the
female. Such small difference in weight permits it to avoid lethal outcome
after pairing which is very characteristic for many spiders. The male ready
to pairing actively searches for females, running on tree trunk. It tries
to keep in places where through forest canopy sun beams penetrate – in bright
light it looks very effectively. Having met the female, the male begins courtship
demonstration: having risen on three pairs of legs, it turns towards to the
female bright inner side of shield so, that sun light reflects from it. When
the female ready to pairing freezes (it is a part of ritual too), male gets
to it on shield, and densely pastes the female by web against bark. Certainly,
it is not enough to of it to keep the female stopped reliably, but the male
may win some seconds to escape from the female after pairing.
The female differs from the male not only in size. At it there is the special
structure of abdomen: it has asymmetric from of the bottom side forming original
“pocket” (usually from the right side). This adaptation is necessary for carrying
of cocoon, which this spider hides under the shield.
Heraldic spiders breed the most part of year, except for the coldest winter
months when their activity is reduced and they spend the most part of time
in shelters. For one year the female bears 3 – 4 cocoons numbering 200 – 250
eggs. Last clutch is usually smaller, rather than others, but eggs in it are
larger, and young spiders have chance to go through winter better. Eggs develop
within 15 days; young spiders sit in cocoon till three days, and then get
over to the bottom side of the shield of the female. Having cast the coat,
they abandon the female for ever and lead independent life. Young heraldic
spiders reach sexual maturity at the age of one year. Life expectancy of heraldic
spiders is rather great and can make up to 9 – 10 years.
All arthropods for growth are compelled to pass through hard process of molt.
Usually at molting spider the top part of cephalothorax armour is separated,
and it gets out through this aperture. At heraldic spider this process is
complicated with very large abdomen of specific shape. Therefore it molts
differently a little: at molting spider the seam separating the top side of
armour from other body bursts. When the top part on abdomen exfoliates, animal
gets out through it, as through the hatch, back to front.
Closely related species of spiders live in subtropics of Europe:
Bullhorn
spider (Heraldarachne taurocornis) lives in woods of Caucasus Peninsula.
It is smaller species, rather than heraldic spider – it is 6 cm long at all.
This species lives on bottom parts of tree trunks in moss cover. The shield
outgrowth on its abdomen forms two “horns” sticking forward. The top part
of body has masking color: it is black with bright green irregular-shaped
spots and thick green hairs on edge. Bottom side of body at this spider is
bright orange; legs at males are silver-gray, at females black. In fight for
territory males “butt” each other by horn-shaped outgrowths. In the heat of
battle spider can fling the contender away or dump it from trunk down by movement
of “horns”.
Leaf-bellied
spider (Heraldarachne foliomimus) lives in forests at the western
coast of Fourseas. It reaches the size of 7 – 8 cm. This species is colored
dimmer, rather than its relatives: from the bottom side of shield there are
no bright hairs, legs are colored grey with dim white cross strips, and only
on forward pair of legs at the male there are reddish-brown “brushes”. But
this spider has excellent masking: the abdomen of this species is flat and
wide, having on edges some jags with tips extended and slightly bent upwards.
Colouring of abdomen precisely imitates dry leaf: the basic background is
yellowish-brown with darker spots. Light strokes simulate leaf nerves and
form irregular mesh-like pattern. This spider eats insects and can kill spiders
of smaller species.
Eared
spider (Heraldarachne auriculatus) inhabits Balkan forests. It is
the largest species in genus: its length exceeds 10 cm at legspan more than
15 cm. It differs from other heraldic spiders by the shape of shield formed
by abdomen overgrown forward: on it forward and upwards two large flat lateral
outgrowths stick. Eared spider lives in forest canopy and does not go down
on trunks. It is very poisonous, and bright warning colouring is developed
at it: shield is bright yellow with black warts on the surface; outgrowths
are black with blue oculate spots in the center. Legs of animal are black
with red tips. The sting of this spider may kill an animal weighting up to
50 kg. Eared spider eats small vertebrates – frogs, lizards and nestlings
of small birds. It also eats large insects – cockroaches and crickets.
Stick
spider (Arachnobaculum inconspicuus)
Order: Spiders (Araneae)
Family: Wolf spiders (Lycosidae)
Habitat: tropical forests of Southeast Asia, forest canopy.
In due course of evolution there is a constant competition between predator
and prey in which new means of survival undergo rigid selection. Speed, force
and poison are the results of such struggle. But it would be incorrect to
reduce competition between predator and prey only to rough power struggle.
The mimicry and procryptic colouring are also results of struggle for existence;
thus not the strongest fighter survives, but the skilfulest imitator. And
both predators and their prey equally succeed in such competitions. Among
invertebrates walking sticks and orthopters among herbivores and mantids among
predators are skilful imitators. Among spiders there is also a lot of imitators
– some of them imitate inanimate objects, and others mimic well protected
animal. In Neocene some spiders in order to masking developed the body shape,
more characteristic for insects of phasmopter order (so called “walking sticks”).
One of such species, living in Southeast Asia, is named stick spider because
of it.
This species has very characteristic body shape. It has squeezed, thin and
mobile abdomen. Because of it the length of its body reaches 90 – 100 mm though
the width of abdomen does not exceed 8 mm. Cephalothorax is also long, and
distance between pairs of legs is about 5 mm. Legs of stick spider are also
very long – up to 15 centimeters. This spider differs in fragility of constitution,
and is not able to run quickly. It is capable of fast movements only at the
moment of prey seizing, and moves in branches simply with fast steps. In case
of danger this spider simply jumps in air and stretches legs in gliding. Due
to small weight and long legs of spider the jump is very slow, and spider
has time to hook for a branch or liana stalk.
The body of stick spider is covered with rich short hair. Colouring of body
is camouflaging – greyish-brown with short cross strokes of black color. When
the spider freezes among foliage and branches, it cannot be made out. Sometimes
insects land directly on its body, not having noticed a spider. Feeling danger,
stick spider can run into similarity of catalepsy – it freezes, having extended
forward two front pairs of legs, and strongly seizes a branch by back pairs
of legs. It keeps immovability even when the predator casually touches its
body. But at the first opportunity of escaping by flight the spider jumps
off from branch.
Eyes of this spider are large. Their location on cephalothorax is very unusual.
All eyes (two large and six smaller ones) are taken out upwards and forward
on “tower” – a special outgrowth of cephalothorax which hangs above cheliceras
and pedipalps. Due to unusual arrangement of eyes this spider has very wide
field of view. Large eyes are directed forward and give the three-dimensional
image of an area directly ahead of spider, a line of four tiny eyes is arranged
under them, and two medium-sized eyes located above the pair of large eyes,
are directed upwards.
Male of stick spider is only a little smaller, rather than the female. In
colouring both genders do not differ from each other. Courtship ritual at
these spiders is accompanied by special courtship dance – male walks around
the female, highly lifting legs and having rised upwards a front pair of legs.
Male holds in cheliceras a symbolical gift – a piece of bark or dry leaf slightly
wound with web. Usually after pairing male succeeds to escape from the female,
but approximately in the tenth part of cases female manages to catch male
and to eat him. Some males can make up to four pairings during their life.
The female carries cocoon under the abdomen, having pasted it to spinnerets
(it is a typical feature of wolf spiders). In order to hide the white colouring
of the cocoon from predators female pastes on its surface small parts of vegetative
dust – fluffy cops of seeds, pieces of bark and rotten leaves. When the posterity
hatches, the female bears young spiders on the body till some days. The grown
up and got stronger youing spiders leave their mother and start to lead independent
life. Their first prey includes plant lice or other tiny insects with soft
covers more often. After several moults young spiders pass to life among air
roots of plants where they hunt larvae of crickets and cockroaches. They reach
sexual maturity at the age of 13 – 14 months.
Zakolo
spider (Zakolo saurophagus)
Order: Spiders (Araneae)
Family: Jumping spiders (Salticidae)
Habitat: tropical forests of Africa.

Male

Female
Pictures by Pavel Volkov
In the Neocene epoch the jumping spider family experienced
an explosion of species. Among its representatives relatively large species
appeared, which could partially replace wolf and tarantula spiders in tropical
habitats, but have not parted with these spiders’ characteristics of speed
and vagrant lifestyles.
In the undergrowth of African tropical forests, and on the roots of large,
mossy trees, one of the largest representatives of this family lives: the
zakolo spider. This is a rather large spider with a body length of 7-8.5 cm,
and a leg length of about 5 cm. The physique of the zakolo spider is quite
massive: the cephalothorax is expanded and has a raised front, and the abdomen
is relatively small (shorter than the cephalothorax), flattened, and with
a rounded shape when viewed from above. The body of the female of this spider
is brown with some light spots at the front. Two big eyes look forward, providing
great stereoscopic vision. They shine brightly and “glow” in the dark due
to a layer of light-reflecting pigments. Two more pairs of small eyes are
on the side of the cephalothorax, directed to the sides, as well as a pair
of eyes looking back. The front pair of eyes is bordered with a white stripe
that gives them the impression of eyes of an animal of much larger size. The
pedipalps and the thickened bases of the chelicerae are hairless and have
a shiny surface, and are colored a bright red color. Claws at the tips of
the chelicerae are white, curved, and about 15 mm long.
The male zakolo spider differs from the female in smaller size (a length of
about 3-4 cm, usually less), a gracile physique, and a different color. He
has brown upper parts of the cephalothorax and abdomen, but the sides are
gray. On the tips of the front pair of legs he has noticeable white cross
stripes edged with red hairs. During his “dance” in front of a female he holds
his front legs lifted upwards and outwards, showing these signals to the female.
His chelicerae and pedipalps are also bright red and lack hairs.
The zakolo spider attacks small vertebrates, or rarely large arthropods. Most
often, it attacks small lizards; its bite kills prey within 10 seconds. In
larger vertebrates the zakolo’s poison causes painful swelling. The spider
prefers to hunt by jumping on prey from above: from tree roots or the branches
of young, low trees. This species is diurnally active; at nights it builds
a temporary shelter, weaving together several large dried leaves with some
threads.
The breeding season for this species is not emphasized, but in areas with
seasonally dry climates most juvenile are common in the wet season. The female
lays about 30 relatively large eggs and wears them with her in a cocoon of
cobwebs. The male often flees after mating, but sometimes it becomes a victim
of the female. Spiderlings appear within 10 days and are immediately cared
for by the female. For the first few days they sit on their mother’s back
and eat part of her prey by sucking up the victim’s tissue dissolved by the
female’s poison. Gradually the spiderlings leave their mother and live independently.
The last spiderlings leave their mother after their first molt. The first
3 molts occur every month, the following 4 are once a month and a half, and
the eighth molt is two months after the previous one. After that, the growth
of the spider stops and all the body’s resources are spent on reproduction.
The female has time to bear up to three broods.
The idea about the existence of this species was proposed
by Nick, the forum member.
Translated by FanboyPhilosopher
Clusterweb
Communal Spider (Congregaranea lineata)
Order: Spiders (Araneae)
Family: Hackled Orb-Spiders (Uloboridae)
Habitat: Forested areas of the North Island of New Zealand.
Islands in particular are vulnerable to invasive species, both those intentionally
brought by man, and accidental stowaways that become established. Some of
these species have left descendants in the Neocene.
So it was that the Little Humped Spider (Philoponella congregabilis) was accidentally
introduced into Christchurch, New Zealand. Finding little competition, it
spread across the warmer parts of the South Island, and via rafting invaded
the North Island too. It has descendants on both islands, but the most notable
is the Clusterweb Communal Spider.
As with its ancestor, it is a small orb-weaving spider, females measure up
to 7 millimetres long, males reach 5 millimetres long. It still bears the
distinctive humped abdomen that gave its ancestor the name, but coloration
is a lighter more reddish shade of brown. A black longitudinal line runs along
the abdomen and cephalothorax, legs are greyish yellow with fine dark bands.
Legs are of moderate length, this spider mainly climbs around the confines
of its web, and those of its neighbours. Pedipalps are large, especially in
males, but fangs are greatly reduced, this species is unable to bite. It kills
insect prey by wrapping it so tightly in webbing that it is crushed.
As with its ancestor, this spider is social, living in communal colonies that
consist of many overlapping webs, maintained by individual spiders. Usually
a colony is made in the branches of a large tree, individual webs are bowl-shaped
and open slanting downward. Each web is a dense tangle of messy fibres, and
may border another web on one or both sides, or be some distance from its
neighbour, linked by pathways of webbing. One web can be as much as 25 centimetres
across, and the entire colony can consist of up to 600 individual webs, spread
over as much as 17 square meters. The spiders will react to intruders or kleptoparasites
as a group, driving them away, and spiders in adjacent webs may cooperate
in catching and consuming prey. Each web is maintained by its owner, but the
webbing that connects them is maintained cooperatively.
Prey usually consist of flying insects, flies, smaller bees, wasps, midges,
mosquitoes, and moths. The spider will quickly attack trapped prey, sometimes
helped by other spiders from nearby webs, immediately it is wrapped in silk
over and over, crushing it to death. After this, the spider spits digestive
juices onto it, and allows it to degrade into a slurry, which it then consumes.
Prey is sometimes shared with others from neighbouring webs. Breeding occurs
in the drier months of the year, between April and June, females within the
colony seek an unrelated male with which to mate. The female produces an egg
sac which is about 10 millimetres long and oval shaped, containing up to 30
large sticky eggs. Mating and breeding occurs within most fertile members
of the colony at roughly the same time, and egg sacs are deposited in a communal
nest, usually in the upper part of the colony. Here, each female deposits
its egg sac, suspended by webs connected to the nest, and to neighbouring
egg sacs. The females guard and brood the eggs in a group, and are brought
captured and wrapped prey by the males. Mothers all feed regurgitated liquid
food to the spiderlings as they grow. Spiderlings are independent and in early
adulthood after about 3 months, many of the mature young will stay and join
the colony, but usually about 30% of them will leave to establish new colonies.
When a colony becomes too large or unruly, it may split into two, or send
part of its number to set up a new colony.
These spiders are commonly preyed upon by mantids and predatory wasps, as
well as birds and small arboreal lizards, each spider may live as long as
2.5 years.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Giant
Fourseas soliphuga (Galeodes tetramarina)
Order: Soliphuges (Solifugae)
Family: Galeodids (Galeodidae)
Habitat: Fourseas area, zone of coastal thickets; forested areas at the west
and south-west of range.

Picture by Biolog
At the boundary of Holocene and Neocene the areas of inland
seas in Eurasia have undergone drastic changes. During the ice age the seas
dried up, replaced by deserts, and after the melting of glaciers the seas
returned to form a large brackish body of water – Fourseas. In a corresponding
manor the composition of the fauna has changed. Some species from arid zones
have adapted to living in the new environment, and among them there is a giant
Fourseas soliphuga, the descendant of the Central Asian solifuge (Galeodes
caspius). Its ancestors adapted to life on the steppes, learning to occupy
holes of other animals, which was important in the dry climate of the glacial
epoch.
This is a very large arachnid species – the length of the body without limbs
reaches 10 cm. The cephalothorax is gray, the abdomen is black with a longitudinal
red stripe. Limbs are gray, with darker tips. The entire body is covered with
dense hairs; on limbs the hairs are sparse and longer, performing tactile
functions.
A distinctive feature of the giant Fourseas soliphuga is very large chelicerae,
vaguely resembling the claws of some crabs. This species feeds on large arthropods
(including its own kind), and occasionally on small vertebrates (mice, shrews,
young lizards and snakes). The chelicerae are able to break beetle armor or
cut the body of small lizard or snake to bones. In case of danger the animal
raises on the two rear pairs of legs and widely spreads apart its front legs.
At the same time the chelicerae are opened, and claws are straightened out.
This species can utter chirping, rubbing its chelicerae against one another.
This species has a nocturnal way of life, hiding during the day in the burrows
of small animals or in other shelters, which are re-built to suit its needs.
This animal also hibernates in burrows, demonstrating a strong attachment
to the chosen habitats. At the east and north-east of its range the animal
digs special wintering holes to the depth of a meter, beginning under a rock
and directed vertically.
The giant Fourseas soliphuga lives in a seasonal climate, and therefore its
reproduction has a clearly pronounced seasonal nature. This species breeds
in August (in the western and south-western parts of its range earlier), with
mating behavior resembling that of scorpions – for some time male and female
crawl, having linked chelicerae and pedipalps. The female lays up to 30 eggs
under rocks in a special chamber whose walls are strenghtened with stones.
Young solifuges hatch after hibernation in the spring, shortly after the melting
of snow in the northern part of the area, and in the south – about the middle
of March.
This species has a large life expectancy – about 10 years, and starts breeding
from the second year of life.
This species of spider was discovered by Nick, the forum member.
Translated by FanboyPhilosopher.
Great-clawed
Longlegs (Onychopillion spp.)
Order: Harvestman (Opiliones)
Family: Neopilionidae (Neopilionidae)
Habitat: New Zealand, North Island, various cave systems, depths of caves.
Numerous similar species within this genus inhabit different cave systems.
Arachnida have commonly given rise to various specialized forms, spiders,
harvestmen, mites and scorpions have all given rise to forms inhabiting specific
habitats, including caves. And thus, native forest harvestmen (Pantopsalis
listeri) of New Zealand have given rise to troglobiotic forms, Great Clawed
Longlegs.
General shape is typical for a harvestman, globular body attached to very
long thin legs, but it differs by having enlarged, long-armed chelicerae that
are bent at the elbows and possessing trenchant, barbed claws. Chelicera and
claws bear long sensory hairs, with which it is able to sense prey and obstacles
when hunting. Ancestor of this species only in males had similar claws, absent
in females, but in this form both sexes have them, as they are used for predation.
This feature is possible due to the changes of the gene expression pattern
resulted in possibility of the occurrence of hermaphroditism. Such a trick
allows these arachnids breeding in small populations Body is about a centimetre
long, leg span reaches as much as 13 centimetres, grasping chelicerae are
about a third shorter than the legs. Coloration is a pale cream-white colour,
becoming yellowish at extremities where chitin cuticle is thicker. This species
is one of the apex predators of its ecosystem, being able to catch and consume
various other kinds of troglobiotic arthropods. Water bodies contain some
predators which may eat it, amphibians and fish. When it senses nearby prey,
it rapidly lashes out, striking with its elongated chelicerae, claws of the
chelicerae crush and impale the prey immediately.
It mates year round, with the male seeking out the female by chemical scent,
sometimes presenting a nuptial gift of food to the female. Male may pursue
the female for some time. If more than one male pursues a female, confrontation
will involve waving and locking of chelicerae in an attempt to overpower one
another. Mating is via penetrative copulation. Females deposit eggs using
an ovipositor, up to 20 eggs are laid in a large cluster in a crevice or depression.
The male remains at the nest after egg-laying, and guards the eggs from predators
and nest raiding cannibals, after hatching there is no other care for offspring.
Hatchlings are tiny and nearly transparent; they consume the tiny young of
other cave dwelling invertebrates, as well as chewing biofilms of fungus and
bacteria. Life expectancy is up to 2 and half years.
This arachnid species was discovered by Timothy Donald Morris, Adelaide, Australia.
Acatu
feather mite (Acatoubius plumophilus)
Order: Mesostigmata (Mesostigmata)
Family: Syringophilids (Syringophilidae)
Habitat: South America, giant carnivorous acatu
bird.

Picture by Biolog
In nature any animal, large or small, represents true “zoo”:
in its organism the plenty of various parasites settles. They are protozoans,
flatworms and nematode worms, various arthropods. Among bird parasites mites
are rather usual. Various species of these arachnids live on feathering of
bird, in pipes and in skin. Some species only temporarily live on bird and
hide in litter of its nest. Other species of mites, on the contrary, live
on the body of bird constantly or the most part of life, excepting the short
periods of settling. Various species of parasites are strictly connected to
certain parts of body of the host animal.
When the giant bird acatu opens wings and wants to fly up, it becomes especially
appreciable, that in one wing some minor primary feathers are deformed and
short. It almost does not prevent this bird to fly, but reduces its maneuverability
at prey chasing. Such development of feathers is the result of activity of
one acatu parasite, the acatu feather mite.
This tiny arthropod lives in shafts of primary and tail feathers of bird.
The adult female of this species represents the translucent worm-like four-legged
creature about 3 cm long at the thickness of body about 2 mm. It lives in
the basis of feather shaft and does not leave it up to the end of life. When
the feather falls out, the female living in it perishes, but more often it
perishes before this moment when the basis of feather placed in skin of bird,
stops the development.
The male of this species sharply differs from the female: it has typical shape
of free-living mite. It is thin-bodied eight-legged creature about 2 mm long.
In essence, the male differs only a little from larvae of last age. Larvae
and males live on skin of acatu and eat exfoliating epidermis, not harming
a bird.
The male fertilizes the female out of feather before her last molt. After
fertilization in behaviour and anatomy of the female striking changes take
place: she starts to search feather papillae from which largest feathers in
acatu feathering – primary feathers in wings and quill feathers in tail develop.
The female gnaws in the tip of developing feather an aperture and creeps inside
of it. Having found the shelter, she at once molts and turns to inactive creature.
The female loses two back pairs of legs, and its abdomen stays covered with
soft elastic cuticle. Only in forward part of her cephalothorax rather firm
chitinous cover remains. On it there are some elastic bristles which allow
the mite to fix inside of feather shaft.
Feather shaft is filled with friable keratinous fibers, and the female partly
eats them away from within. She gradually reaches a basement of feather. To
this moment it is not formed yet, therefore the female has access to blood
vessels surrounding the feather bursa. She pierces an environment of developing
feather and starts to suck blood. At this moment at her in an abdomen the
development of eggs fertilized before begins. For short life the female of
acatu feather mite can form and incubate in the body over 50 thousand eggs.
They ripen in small portions in abdomen of the female, and gradually, as on
the conveyor, move along the oviduct. From them right in oviduct hexapod nymphs
excluse. After birth they live some time inside the feather shaft, eating
dung of the female and the rests of keratinous fibers. The female of acatu
feather mite prepares for them a kind of “baby food”, only partly digesting
the exhausted blood of bird. Nymphs eat liquid dung of the female which actually
consists of semidigested blood of acatu and is rather nutritious. Eating such
food young acatu feather mites pass some molts inside the feather. Nymphs
of advanced age scrape walls of feather from within, making it fragile. Because
of their activity the feather breaks or broken off, when the bird cleans feathers,
and mite nymphs leave it. They begin life as commensals, crawling on the body
of acatu. Nymphs of acatu feather mite settle during the contact of adult
birds, or bird and nestling. But they have one more way of moving which they
frequently use. Nymphs find larvae of Goliath
feather louse (Megalaembothrion goliath) – large parasitic insect, and
cling to them. The nymph can not eat very long time, staying in inactive condition
on the body of Goliath feather louse larva. They expect, while the Goliath
feather louse larva will cling itself to any flying insect and will get on
body of new host bird. The part of nymphs, however, stays on body of bird
and also turns to adult mites.
Crustaceans
|
Meganesian
Tadpole Shrimp (Triops aridophilus)
Order: Tadpole Shrimp (Notostraca)
Family: Tadpole Shrimp (Triopsidae)
Habitat: Seasonal drying-up lakes of the desert areas of south-western Meganesia.

Picture by Biolog
Extreme, ephemeral environments can sometimes provide refuges
for species which have a long evolutionary history, such as the Tadpole Shrimp
(Triops), which was commonly found in temporary reservoirs in Australia.
In the temporary drying lakes of the western Meganesian deserts, a descendant
of Triops lives. Generally it greatly resembles its ancestor in habits, size
and shape. Individuals reach up to 7 centimetres long, and the fore body is
dominated by a large carapace that covers the thorax and head, with 2 large
compound eyes and a central eye-spot, the segmented abdomen and uropods trail
behind. Arrangement of legs and appendages is generally primitive for a crustacean,
with paired appendages at the front (thoracopods), and clusters of up to six
appendages per segment further back, the rear of the abdomen forms a “tail”
which bears no appendages. The very first appendages are raptorial and used
in feeding, with branched swimming and breathing appendages behind those.
Food is stirred up using the branching appendages of the thorax, and larger
food particles are caught and torn up at the front. Colour of the animal is
a spotted olive green, and the carapace is opaque brownish green. These shrimp
feed on edible particles and carrion that are suspended in the water and sediment,
including dead animals, vegetative detritus, and they will also practise cannibalism.
Unlike its ancestor, a slight spiny serration is seen on the outer edge of
the carapace, and the midline bears small tubercles.
When the water level is relatively high, late in the year, they begin to prepare
to breed, sexual activity is rare and asexual reproduction predominates (parthenogenesis).
The eleventh pair of appendages is developed in the female into egg carriers,
where the eggs are stored as they develop, to be released in batches, a female
may release as many as 500 eggs in one day. Eggs have a hard shell and can
dry out completely, remaining dormant until they are wetted again. Dormancy
can be sustained for up to 20 years. As soon as the reservoirs fill with water,
the larvae hatch and develop within a day through 5 instar phases, eventually
looking like miniature adults. Lifespan and life cycle coincide with the yearly
rain cycle, unless a drought prolongs the dry period, in which case eggs stay
dormant until the next rain. These shrimp are eaten by fish, other crustaceans,
frogs and birds.
This species of invertebrates was discovered by Timothy Donald Morris, Adelaida, Australia.
Ghostly
Water-flea (Phantodaphnia tenebra)
Order: Water Fleas (Anomopoda)
Family: Daphniidae
Habitat: Slow-flowing parts of underground cave waterways, some areas downstream,
New Zealand.
Any freshwater habitat will contain very small, planktonic organisms that
are suspended in the water, which usually consists of various tiny crustaceans.
And so it is within the caves of New Zealand, subterranean waterways that
are deep and slow-flowing host swarms of the Ghostly Water-flea, descended
from the common Daphnia water-flea.
This crustacean is usually no larger than 2 millimetres long, its body is
encased in a carapace that has 2 sides, with a cleft in the lower-middle from
which the fluttering hairy legs protrude slightly. The second pair of antenna
are large compared to the body, covered in long hairs, and are the main means
of propulsion. Head segment is fused to the body, and the eye is absent, carapace,
antenna and legs are completely transparent, visible internal organs are opaque
and pale. Post-abdomen protrudes from the lower part of the shell, and bears
a series of curved spikes, this allows it to cling to surfaces when not swimming.
Diet consists of microscopic algae, protists and bacteria which are filtered
from the water. The legs beat constantly, which creates a current pushing
water and food particles into the digestive system, the second pair of legs
catch edible matter and pass it to the mouth.
Females are capable of parthenogenesis, and produce eggs in this manner for
most of their life cycle, one brood is produced at each moult. Each brood
at this stage contains up to 80 eggs that hatch within a day. Towards the
end of the life-cycle they switch to sexual reproduction, stressed clonal
females are able to become male in overcrowded places. After mating, a brood
of hard-shelled eggs are cast off during the next moult, and these can withstand
drying out or extreme cold. Lifespan is generally between 5 and 8 months,
predators include various small aquatic invertebrates and even those that
filter feed.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Voodoo
sacculina (Voodoocancer zombiefabricatus)
Order: Cirripedians (Cirripedia), suborder Root-headed crustaceans (Rhizocephala)
Family: Sacculinas (Sacculinidae)
Habitat: tropical areas of Old World and Meganesia: from Zinj Land and Tanganyika
passage at the west through Hindustan, Southeast Asia and Indonesia up to
northern coast of Meganesia.
Root-headed crustaceans represent one of the tops of evolution of parasitic
crustaceans. The body of the parasite almost completely loses the features
typical not only for crustaceans, but also for arthropods in general. The
whole body of sacculina represents root-like outgrowths completely penetrating
the body of the host – usually decapode. Only the sac-like body of the parasite
in which genitals of animal are placed is visible from outside.
Sacculina parasitizes in host crustacean, not destroying its organs. The animal
inhabited by it keeps normal viability and adequately reacts to occurrence
of predators, escaping itself and its parasite in common. But sacculinas affect
its maturing, causing in various cases parasitic castration or regeneration
to the individual of opposite gender. Usually sacculina is a hermaphrodite,
but any number of species of close genera has dwarf males. In this respect
the species evolved in Neocene and named voodoo sacculina strongly differs
from them. At this species both genders are approximately equally advanced
– males are only a little smaller, rather than females, and do not cause deep
physiological changes in host’s organism during their growth and development.
Voodoo sacculina does not differ externally from other related species. Its
body represents mainly root-like bands penetrating the body of the crustacean
infected with it up to tips of antennae and legs. Under the abdomen between
segments of infected animal sac-like outgrowth of yellowish color, about 20
mm in diameter, covered with dense chitinized cuticle without traces of segmentation
is stuck out. It is easy to distinguish males and females of this species
by the appearance of this sac. Females have only two apertures of oviducts
on it. Males have a pair of special closed sacs; in each of them there is
a genital looking like thin tubule. During the pairing the sac is opened and
it stucks out, searching for oviduct aperture of the female.
Sexual breeding with fertilization has the great advantage to its owners.
At the pairing of two genetically different individuals in their posterity
the recombination of their features takes place and it promotes evolution
of the species as a whole and complicates the infection with viruses. But
for the parasite its complete dependence on its host represents a great difficulty.
Voodoo sacculina has one way to overcome this obstacle. Penetrating the host’s
body, this animal changes the shape of the host with its secretions: in comparison
with the not infected representatives of the host species individuals carrying
voodoo sacculina have more advanced antennae covered with lengthened bristles
with a plenty of sensor cells. This adaptation aggravates sense of smell of
the infected animal in great degree, making it more cautious, that at the
same time saves the life of the parasite. But the basic function of this adaptation
is absolutely different. Having reached the maturity, voodoo sacculina begins
to influence the nerve system of the host animal, changing its behaviour.
In breeding season the parasite forces the host to search with the help of
smell sense not for the partner of its own species, but for the crustacean
of any kind also infected by individual of opposite gender of voodoo sacculina.
Hence the trivial name of voodoo sacculina – the parasite completely subordinates
the behaviour of the host crustacean. As a result of it at the sea bottom
the strange scenes resembling courtship games take place, performed by crustaceans
not only of different species and genera, but also of different families (voodoo
sacculina is not specialized in choice of the host). Sometimes it happens,
that male of voodoo sacculina infects any shrimp, and it, being more mobile,
searches for carriers of sacculina females including ones belonging to species
eating shrimps in a typical case. But in this case smell of the approaching
male forces the female of voodoo sacculina to interrupt the aggressive behaviour
of the host in order to pairing. During the pairing the carrier of the female
extends an abdomen, making accessible an outgrowth in which genitals of the
parasite female are located. Thus male individuals of host species display
the sexual behaviour of the female, allowing carriers of voodoo sacculina
males to approach the parasite to sufficient distance to make fertilization
at parasites. Spermatozoa of the male may keep viability in ovaries of the
female till about half-year, but the female is regularly coupled with new
самцами and their spermatozoa as more mobile and viable fertilize her ovocytes
in the primary order. If the pairing had not taken place for a long time,
the last group of impregnated ovocytes divide repeatedly, and from one initial
ovocyte some hundreds eggs may be generated.
The further development of voodoo sacculina differs in nothing from life cycle
of other species of this group. Fertility of the female makes up to 10 thousand
tiny eggs per one week. Planktonic nauplius turns to cypris and is attached
to covers of the host. After that it moults once again and penetrates into
the body of the host as an unstructured lump of cells of which the whole body
of animal develops inside its host. Life expectancy of voodoo sacculina male
is short and does not exceed two years. The female of this species lives till
five years.
The idea about existence of this species of crustaceans was proposed by Momus, the forum member.
Fish sacculina (Piscisacculina quasisymbiotica)
Order: Root-headed crustaceans (Rhizocephala)
Family: Fish sacculinas (Piscisacculinidae)
Habitat: northern part of Pacific Ocean, seas of Southern hemisphere.
In seas of Holocene one surprising crustacean was known – sacculina, the
endoparasite of crabs, developing in the infected crab as long roots in all
its body, exhausting nutrients. In Neocene this kind of parasitism had promoted
further away, practically up to a level of symbiosis. The crustacean similar
to crab sacculina infects fishes of certain species leading mainly schooling
habit of life. These fishes do not make long migrations, remaining within
the limits of zone of inhabiting of the population, near to coast of continent,
and eat seaweed, invertebrates, ground and silt inhabitants.
The microscopic larva of crustacean – the nauplius – finds young fish, is
swallowed by it with water and settles on gills. Having attached to epithelium,
it has metamorphosis. As a result of it the crustacean larva sprouts inside
of host animal body as long tissue outgrowths, and the rests of the nauplial
cover on the surface of fish gills disappear after the molt. Gradually “sprouts”
pass through all its body and organs, absorbing nutrients of the host organism.
The fish does not feel the special discomfort; the parasite has adapted to
not affect for the time being the vital centers, and also develops the connections
overwhelming unpleasant sensations. Taking root in gonads, the invader dilutes
sexual products of fish at early stages of development, and develops its
own ovo- or spermatogonies, depending on its sex, occupying all volume of
former gonads. Ripening of sexual products of crustacean coincides to terms
of egg ripening of the not invaded individuals of fishes of this species.
In required term the fish comes to spawning with other congeners. But it
can not spawn eggs any more, and generates in its gonads eggs or sperm cells
of the parasite. And soon naupliuses of the parasite appear, swimming to
searching of new hosts.
The parasite creates alternative communication system in the body of fish,
providing transport of substances, oxygen, and participating in their metabolisme.
By developed substances it interferes with development of congeners again
getting on this fish, and they perish still larvae at attempt of “sprouting”
into the fish. Except for it, thr parasite creates a chemical barrier to
all this school of fishes for other larvae, frightening them off and overwhelming
their development, and it is as stronger, as more is the rate of infected
fishes in school. If in fish school there is more than the one fifth of infected
fishes, defeats new practically do not occur and fishes receive some kind
of “immunity” from new attacks - the parasite has “an interest” to keep a
significant part of fishes healthy to permit them to breed.
Through skin of fish long hair-like outgrowths of the parasite sprout. They
are supplied with sensitive receptors reacting to the slightest signs of
food keener, than fish’s own ones, and fish through system of the mutual
biochemical communications moves there, and it is followed also by other
fishes of school. Except for it, the poisonous secret of the parasite emitting
through the same outgrowths, makes fish almost up to the end of life uneatable.
The predators had tried such fish few times, at all try to avoid catching
of fishes of this species. The infected fishes live till 5-6 years.
Only at the end of host’s life the parasite takes root into brain of fish,
and its coordination of movements is broken. The struck fish swims badly,
and larger predator usually swallows such individuals. But it will not be
infected, because its biochemistry is not the same, as at host fishes to
which the physiology of parasite is very well adapted. In digestive path
of predatory fish after digestion of catch beforehand firm formations of
the parasite similar to cysts are formed. They will be kept, and the fish
will distribute them wandering in open spaces of ocean in wider areas, than
migratory routes of the separate population of the host fish. After removing
of cysts from the organism with excrement from them plankton larvae of parasitic
crustacean will burst.
This species is discovered by Arseny Zolotnikov.
Surveyor
scud (Geocaprella geometra)
Order: Scuds, or Side-swimmers (Amphipoda)
Family: Skeleton shrimps (Caprellidae)
Habitat: shores of northern part of Pacific ocean.
Active processes of orogenesis in northern part of Pacific Ocean have resulted
to radical changes of the coastal line of Asia and Northern America in comparison
with situation known in Holocene. A plenty of new islands has appeared, and
it has permitted to some sea inhabitants to master land.
In ebb-tide strip of north - Pacific islands life has to found a convenient
place for itself. True “forests” of brown and green seaweed develop here, and
in them tens species of invertebrates crawl. In these places one equally well
feeling like both in water, and on land, survives successfully.
Among crabs scurrying among rotting thalli of seaweed at the coast, it is possible
to find strange creatures of unusual appearance: they are long-bodied, also
creep “by steps” similarly to caterpillar of geometer moth. But antennae and
well advanced legs indicate, that they are not caterpillars. The body of these
creatures is covered with firm armour and colored red - brown with dark irregular-shaped
spots. These animal are lineal descendants of crustaceans skeleton shrimps
(Caprella). For characteristic manner of movement this creature is named surveyor
scud. They are rather large representatives of order: body length of surveyor
scud is up to 10 cm.
The body of surveyor scud is covered with numerous knobs and outgrowths, and
on antennae and legs rounded scalloped outgrowths stick up. It helps this crustacean
to mask among seaweed from enemies and possible catch. During the inflow the
surveyor scud stays in thickets of brown seaweed, waiting for small animals:
fry, other crustaceans and snails. Having clung by back legs to seaweed, the
surveyor scud expects, while the prey will come nearer too close to be seized.
The caught prey is kept by forward pair of legs on which tips there are small
spikes. During walking the surveyor scud folds these legs under thorax.
Being the inhabitant of an intertidal zone, the surveyor scud is able to live
not only in sea, but also on land. In outflow tens of these crustaceans creep
in intertidal zone, eating animals cast ashore. But at this time they are
endangered: many sea birds fly above intertidal zone, and even ground predators
at times search for catch at the coast. Therefore, having remained at the
coast, surveyor scuds keep near to natural shelters - usually near heaps of
seaweed. At first signs of danger surveyor scud rushes to the nearest shelter.
Due to colouring and body shape it easily masks. When the hidden crustacean
keeps in seaweed, it may be easily mistaken for a part of plant. Like crabs
and fishes surveyor scuds rescue from small predators by jumps: having turned
up the back end of body under itself and sharply having straightened, the
scud jumps at the distance up to meter. Being overtaken at the flat area,
it can escape, very quickly having buried in sand. As a last resort seized
surveyor scud easily rejects limbs, which completely restore after two molts.
The female of this species carries eggs and posterity on itself. At this species
there is direct development, and from large eggs posterity similar to adult
scuds bursts. The young growth keeps on body of the female and eats the rests
of her catch. Before the first molt the posterity abandons mother and further
lives independently.
Plankton living stick (Thalassophasma tenuissima)
Order: Scuds, or Side-swimmers (Amphipoda)
Family: Skeleton shrimps (Caprellidae)
Habitat: Pacific Ocean, top water layers.
After extinction of significant part of plankton organisms the set of ecological
niches of open ocean appeared empty. It stimulated the evolution of survived
ocean inhabitants and had permitted to some inhabitants of sea shallow waters
to make attempt to develop habitats of open ocean. At the initial stage of
open ocean settling in plankton there had been a plenty of descendants of
shallow water forms, but gradually their amount had decreased, and few lines
of new immigrants to plankton had survived only. Even descendants of one
rather specialized group of scuds – skeleton shrimps (Caprellidae) were among
them. These animals had developed pelagic habit of life rather originally:
they had left shallow water adapting to live on huge brown or green algae
floating in top layers of water. And only therefrom skeleton shrimps passed
to inhabiting in thickness of water, becoming true plankton animals. One
species of these creatures differs in very large size and characteristic
appearance. It is named plankton living stick.
This crustacean looks the real giant among scuds: it is the animal up to
20 cm long. But the large size does not mean that it is very strong and dangerous
creature. Plankton living stick quite justifies its name – it is the fragile
and graceful creature adapted to plankton life. The body of this animal is
very thin, and four forward pectoral segments are lengthened very much –
they amount approximately 80% of the general length of animal. Four forward
pairs of legs are also very long. They are covered with hairs facilitating
soaring in water. The forward pair of legs is little bit shorter, rather
than the others – it is used for prey grabbing. On tips of forward pair of
legs small pincer-like outgrowths develop, helping to keep catch.
Segments of back part of thorax appreciably differ from front ones – they
are flat; two pairs of short and strong swimming legs with dense bristles
on edge grow on them. In case of need the plankton living stick is able to
swim – for this purpose it folds long forward legs back, and rows by swimming
legs. But this animal is not so good swimmer, also is is not able to swim
for a long time. The longest moving it makes is the vertical migration. Having
a presentiment of storm, this fragile crustacean falls to the depth of about
20 meters.
The pleon of plankton living stick is reduced (this condition had been present
already at skeleton shrimps, ancestors of this animal) and is extended to
needle-shaped outgrowth.
Usually plankton living stick motionlessly soars in thickness of water, having
stretched pectoral legs in sides. Occasionally one or two swimming legs come
in movement, slowly turning animal to the necessary direction. This sluggish
creature eats small plankton animals, catching them one by one. At this animal
there are maxillipeds of cutting type – plankton living stick bites off pieces
of catch.
Fertility at plankton living stick there is rather low for plankton animals
– the female lays at all only some hundreds eggs and bears them on segments
of back part of thorax (at females these segments are wider, rather than
at males) within approximately three weeks. From eggs young animals similar
to adults hatch. They sit on body of the female about one week and catch
smallest plankton animals floating near it, then abandon the female and live
independently. The young growth is able to swim faster, rather than adult
individuals – at just passed to independent life plankton living sticks body
is shorter and swimming legs are rather long. Also at them the forward pair
of pectoral legs is longer than the others. With each molt proportions of
young animal advance to proportions of adult individual.
Mantis
scud (Deinogammarus rapax)
Order: Scuds, or Side-swimmers (Amphipoda)
Family: Scuds, or Side-swimmers (Gammaridae)
Habitat: Fourseas, coastal thickets of plants. This crustacean can rise against
current of rivers and form freshwater populations.
The history of Fourseas had begun from zero, or from the size close to it. When
four internal seas at the south of Europe had dried up, all sea fauna appeared
simply destroyed. And after rigorous ice age when Fourseas had formed, this
reservoir became populated with numerous species of live creatures. They were
not descendants of representatives of the extinct fauna, but animals, whose
ancestors lived in rivers and lakes.
In geological sense Fourseas had formed very quickly: for some hundreds thousand
years thawed snow of glaciers of Northern hemisphere have filled in hollows
of dried up seas, and rains have lifted sea level even more. Tectonic processes
have closed Bosporus passage, and Fourseas actually appeared a little bit above
ocean level.
Processes of evolution of live creatures in new reservoir have gone especially
roughly. The set of free ecological niches has permitted to descendants of few
founder species to avoid the competition. Among crustaceans which have occupied
Fourseas, there were scuds. These crustaceans had survived in glacial lakes
with cold and clean water. When glaciers had started to thaw, filling in hollows
of dried up seas, scuds first of all began to develop the new world, forming
new species better adapted to inhabitancy which Fourseas could give them. They
became the most mass group of invertebrates in reservoir. Among them both fine
plankton species, and various ground inhabitants have appeared. And one of species,
the mantis scud, became the true predator.
The length of this crustacean is about 10 cm: it is one of largest species of
scuds of Fourseas. But it seems even larger because of very long antennae, surpassing
in length its body. Except for them, on head of the crustacean there are very
well advanced eyes consisting of set of very small facets. Due to it mantis
scud can distinguish tiny details of environmental district. The body of this
crustacean is lengthened and flexible, numbering set of segments.
This animal is active ambuscader eating water insects (larvae of dragonflies,
water beetles, etc.), leeches and small vertebrates (fishes, tadpoles, frogs).
Also this species willingly eats carrion. In this connection at it the adaptation
for keeping of catch and meat tearing are developed: the forward pair of legs
has increased, segments on edge are supplied with spikes and jags. In rest these
trap legs are put together, but when the animal attacks, they quickly open and
grasp catch. This crustacean can swim quickly: three back pairs of legs are
feather-like, covered with long elastic hairs. Back pairs of legs also serve
for attachment to substratum: on their tips sharp bent claws are advanced.
Colouring is cryptic: the top side of body is olive-brown, and bottom is reddish.
It permits the crustacean to mask in thickets of pondweed, waiting for catch.
Usually this crustacean hides in plants, and expects for suitable catch. But
the hungry crustacean may chase catch, swimming to small distance from shelter.
These crustaceans, similarly to many single predators, are strictly territorial:
invisible borders share thickets of underwater grasses to set of sites, and
each of which is vigilantly protected. If the neighbour has crossed border and
does not hurry up to leave another's possession, the lawful owner warns it.
For this purpose on trap legs of the mantis scud the special knobs, to which
on the next segment small holes are opposed, are advanced. Bending the leg,
and pushing knob in hole, crustacean utters sharp clicking sound. The click
distinguished by loudness and sounding, is an alarm signal. When above places
of inhabiting of mantis scuds the large fish swims, its movement is accompanied
by alarm signals of these animals. If the neighbor or predatory fish attacks
the mantis scud, the animal is able to protect itself with the help of spiny
trap legs. Protection is accompanied by the special gnashing sound which will
be remembered by any predator, having received deep painful wounds from the
mantis scud.
The pledge of success of survival consists not only in skill of protecting against
the enemy, but also in care of posterity. This species differs in child-loving:
the female drags eggs and larvae on the body. From large eggs (up to 100 ones
in clutch) hot helpless larvae, but tiny advanced youngs – small copies of parents,
hatch. Since the first minutes they can move independently, but do not leave
mother before hardening of carapace after the first shed. They eat the rests
of mother’s catch, and successfully grow up till the size guaranteeing their
survival. Young mantis scuds are good swimmers, and can attack animals of size
equal to them.
Variable
Troglodyte Land-Scud (Cavernalitrus variablis)
Order: Scuds (Amphipoda)
Family: Land Shrimps/Shore Hoppers (Talitridae)
Habitat: Cave systems throughout New Zealand, species is variable, surface-dwelling
variant exists similar to ancestor.
Crustaceans have become masters at colonizing land, but have almost never fully
divorced themselves from water. Prime example is the Land Shrimp, a kind of
terrestrial scud which required damp vegetative conditions to survive away from
water. These successful crustaceans prospered wherever they were found, and
even became stowaways in garden plants and colonized Europe as an invasive species.
One local descendant of these is the Variable Troglodyte Land-Scud, an amphibious
crustacean about a centimetre and a half long. It is descended from a species
introduced from Australia (Arcitalitrus sylvaticus), the endemic species having
become much rarer. Two main forms of this species exist, cave-dwelling strain
and surface dwelling strain. Surface-dwelling strain differs by darker coloration
and slightly better eyesight. Shape of this crustacean resembles its ancestor,
hunched, abbreviate shrimp shape with numerous segments, and short abdomen tucked
under the body, antennae are prominent. Rear segments bear pleopods, front 2
segments bear legs jaws (gnathopods), there are 4 pairs of walking legs, posterior
pair of legs is robust and used to jump or swim away from danger. Cave dwelling
strain is pale, with opaque, almost translucent coverings, insides are faintly
visible. Eyes are present, but in cave-dwelling strain are generally weak from
disuse.
Surface-dwelling individuals are only active at night, being only present in
areas with enough moisture in the vegetation, such as wet forests or moss. Cave
dwelling form is active all day, dwelling mostly in the deeper parts of caves,
but is also present in moister upper reaches of caves, it is absent from caves
containing bat colonies. This crustacean feeds on biofilms of algae and fungus,
carrion, rotting matter and occasionally slow-moving invertebrates caught underwater.
Being amphibious, it is able to both crawl and swim; rear pair of legs allows
this crustacean to either jump quickly or swim backstroke in order to move quickly.
Cave-dwelling individuals breed year round, surface ones breed only when they
sense conditions are moist enough. Fertilization is internal; the female lays
her eggs within a fluid-filled brood sack which she carries on her underbelly.
Young are released upon hatching, and resemble miniatures of their parents,
going through a series of moults until reaching adult size. Lifespan is short,
only about 2 years.
This crustacean species was discovered by Timothy Donald Morris, Adelaide, Australia.
Sea bird louse (Thalassophthirus aviphilus)
Order: Isopods (Isopoda)
Family: Idoteids (Idoteidae)
Habitat: Northern hemisphere, Atlantic and Arctic oceans. The parasite of large
sea birds.
Extinction of cetaceans and others sea mammal in epoch of ecological crisis
had carried away to non-existence the fauna of their parasites – worms, crustaceans,
and other species of invertebrates. In Neocene the place of large sea vertebrates
in Northern hemisphere is occupied by large flightless birds. Various species
of parasitic animals had settled these birds evolved in appropriate way. Some
of them descend from own parasites of birds, and others had settled newly evolving
species of huge birds from zero.
One of new settlers of huge sea birds is the parasitic species of isopod crustaceans,
externally similar to louse - because of it this species is named sea bird
louse. It settles on skin of birds under feathers and eats blood. Length of
body of this crustacean is about 7 – 11 mm. Obviously, it is the descendant
of one of species of sea crustaceans of genus Idotea – usual inhabitants of
interdial zone in Holocene epoch.
The body of sea bird louse is flattened, covered from above and from sides
with firm shell. On the bottom side of body chitinous cover is thin and folded.
Plicas increase the area of body surface, and help in respiring. And when this
animal sucks a plenty of blood, plicas are smoothed, and the body becomes slightly
bulging. Mouth feet of this crustacean are modified to thin long proboscis.
At sea bird louse five pairs of walking legs are advanced. The forward pair
of legs is similar to hunting legs of soothsayer – on their internal side there
are pointed thin denticles. These limbs serve for attachment to the host body
– sea bird louse clasps by them feather shaft of host bird, or clings the stretched
legs to its skin, sticking denticles in epidermis of the bird. Next three pairs
of walking legs are short and hook-like. On penultimate segment of walking legs
there is a sharp spike, and last segment is hooked. These two segments form
strong claw, therefore sea bird louse is not washed away from host bird by casual
wave, and even bird carefully cleaning feathering not always can deliver from
these parasites. Last pair of walking legs has changed to similarity of flippers
– they are lack of spikes, but last segment of these legs is very wide and is
bordered by rigid hairs. With the help of such legs sea bird louse swims well,
and in rest these legs cover from below very short abdomen on which gills are
located, and protect them from drying.
Though sea bird louse is able to swim, it can stay on land for a long time
and even rather quickly creeps on the ground. It settles on head of gannetwhales and
other sea flightless birds – plesioloons and laughterloons. Occasionally
this species settles on skin of other birds, but, as a rule, it lives on them
not for long: small birds clean off these crustaceans from themselves or peck
them off one from another. Sea bird louse settles at direct contact of birds,
or swims from one bird to another when birds hunt in sea. This crustacean makes
risky travels on the ground in cool foggy night when the risk to dry or to
be eaten is less.
Sea bird louse breeds, laying many small eggs – up to 2 – 3 thousands in one
clutch. When the host bird stays on land, female protects eggs from drying,
covering them by swimming legs. Term of incubating of eggs is short – about
3 – 4 days at all. When the host bird dives, tiny free-living pelagic larvae
hatch from eggs, quickly leaving egg shells. After that female within day cleans
off rests of clutch from the body, and becomes ready to breeding again. For
one year one female can make over thirty clutches.
Mouth parts at sea bird louse at any stage of development represent the proboscis.
Larvae of early age live among seaweed and eat small soft-bodied animals, exhausting
them. They are similar to adult individuals, but at them there are transparent
body and only two pairs of walking legs. Later they start to attack large invertebrates.
Gradually larvae settle closer to surface of water. Larvae of advanced age,
which has reached the size of about third of length of adult individual, pass
to amphibiotic habit of life: they creep out on surface of algae carpet, waiting
for occurrence of the host bird. Having got on the body of bird, crustaceans
pass to feeding on blood, and start to grow quickly, turning to adult individual.
Shogashi (Mesidoteopsis shogashi)
Order: Isopods (Isopoda)
Family: Idoteids (Idoteidae)
Habitat: North America, Mishe-Nama lake.
At the northeast of North America the wide Mishe-Nama lake is stretched. In
human epoch at this place Hudson bay was, but in ice age at the boundary of
Holocene and Neocene it was repeatedly covered with glacier shield and all
live creatures there died out or receded to cold freshwater reservoirs formed
along the edge of glacier. Waves of glaciation one after another gradually
deposited ground in mouth of the gulf, making poorer its connection to the
sea. At the end of glacial epoch Hudson bay appeared completely separated from
Atlantic and turned to Mishe-Nama lake.
Among inhabitants of this lake there are some kinds of animals of sea origin.
Among them there were isopod crustaceans of genus Mesidotea. Their descendant
is shogashi – completely fresh-water species of crustaceans.
Shogashi is rather large species: the length of its body reaches 15 cm. Body
of animal is flattened, with four pairs of strong walking legs. The abdomen
of animal is short and ends with shovel-like telson dulled at top. Body colouring
of shogashi is brown with lighter pattern, but in many respects it depends
on color of the ground. Shogashi is capable to change colouring according to
color of the ground in places of its inhabiting. In shallow sandy areas of
river mouths formed of deposits of light sand there are yellowish-white individuals,
and completely black individuals live in deep whirlpools. Nevertheless, each
animal is able to change shell color within several days, according the colors
of environment. And after the moult color varies much faster – while armour
is hardening.
Sight sense plays rather small role in life of shogashi. Eyes of animal are
small and consist of several hundreds facets. Sight allows shogashi to distinguish
objects at the distance of up to half meter far around of it. Shogashi receives
the information about far objects due to sense of smell. Antennae of animal
are short and mobile, and have thick bases. They are supplied with the set
of chemoreceptors, allowing keen distinguishing of smells.
Shogashi is flesh-eating crustacean. It is mainly a scavenger and small animal
hunter. Its three forward pairs of thoracal legs are prehensile – this feature
is inherited from ancestors. However, the first pair is increased and stronger,
and the similarity of subchelae develops on it, like at mantis shrimps. Subchelae
of shogashi are supplied with cutting edge and several pressing tubercles on
opposite surfaces of segments. With its help shogashi crushes shells of molluscs
and crustaceans which it succeeds to catch, and even cuts bones of prey like
small fishes and frogs. Usually shogashi eats snails, catches small crayfishes
and eats carrion, but it also can catch quicker prey from an ambush. Hunting
fishes and frogs, shogashi hides among ground dust and keeps immovability while
the careless animal comes too close. Shogashi frequently settles in water near
nesting areas of fish-eating birds and takes prey drop by them.
This crustacean lives at the shoalinesses and hides among plants. In case of
danger shogashi can quickly dig in the ground, using its telson like a shovel.
This species is also capable to protect itself actively – if the aggressor
is not too large, or there is no another way to escape, shogashi seizes the
enemy in forward pair of thoracal legs.
This crustacean has direct type of development, and the young ones completely
similar to adult individuals leave the female. Shogashi female raises young
ones in pair brooding pouches located on abdomen. The brooding pouch contains
up to 50 eggs from which transparent young crustaceans hatch. The courtship
season at shogashi begins at the end of winter while water is still cold –
it is a heritage of life of its ancestors in cold water. In summer, when water
gets much warmer, shogashi moves to the depths of lake.
The young individual moults 8 times before it will reach the size characteristic
for adult individual. Life expectancy is about 8 – 9 years.
Tridacna louse (Malacophthirus parasiticus)
Order: Isopods (Isopoda)
Family: Aegids (Aegidae)
Habitat: reeves of tropical area of Indian Ocean, shells of giant bivalves.
At the reeves of Neocene corals have the subordinated position. The majority
of species of madreporarian corals had died out in human epoch as a result
of pollution of oceans and destructions of habitats of these animals. In early
Neocene corals with firm calcareous skeleton had been substituted as reef-builders
with the set of representatives of other groups of animals. At the reeves of
Indian Ocean very large bivalve “blossoming”
tridacna (Obliquitridacna florida) lives; it had turned to almost autotrophic species due to symbiotic algae and
cyanobacteria which inhabit outgrowths of the mantle, giving to them bluish-green
color.
In shells of these huge molluscs various animals settle: crabs, shrimps and
even small fishes. Some of them are harmless to their host, others are parasites
and harm a huge mollusc, and the third ones help it to clean parasites. One
of shell inhabitants is a small species of isopods living on the surface of
edge outgrowths of the mantle containing symbiotic unicellular organisms. Because
of similarity in habit of life with the appropriate parasitic insects it is
named tridacna louse.
It is an active and mobile species of parasitic crustaceans feeding in an adult
condition exclusively on the “blossoming tridacna”. Its body had not undergone
significant changes which are usually characteristic for parasitic forms, including
isopods. Eyes of this animal are large, with a plenty of facets, providing
good sight. Tridacna louse reacts to appearing of fishes and other large animals,
and quickly hides in shell of the host. The body of tridacna louse is wide
and flat, allowing squeezinge inside of the shell of host mollusc through a
crack between valves. This crustacean is able to run quickly on the surface
of mantle of the mollusc; it has seven pairs of well advanced walking legs.
Pleopods of this species are transformed into organs of attachment – they are
mobile and lengthened, and on their edges small hooks grow. Tridacna louse
can be hooked with their help to outgrowths of the body of host mollusc, as
if by nipper. Body cover of this crustacean is greenish, providing the masking
on the background of the color of surface of mollusc body.
Oral parts of tridacna louse are transformed into the proboscis made of two
halves which press against each other during the feeding. This species sucks
colonies of symbiotic algae from tissues of the host mollusc. From them tridacna
louse receives not only nutrients. From blue-green algae this crustacean takes
poison which is concentrated in its body, making it absolutely inedible for
fishes.
Adult tridacna louse can spend the whole its life on the same mollusc, therefore
the probability of meeting of the partner of opposite gender could be much
less, than at free-living forms. But tridacna louse is a hermaphrodite, and
two any individuals of this species mutually fertilize each other at the meeting.
The sperm liquid is kept for a long time in special pocket of oviduct, and
serves for fertilization of several sets of eggs. Right before pairing these
crustaceans clean from each other’s pockets the sperm left by the previous
partner. If other individual is not met for too long time, at this species
parthenogenesis is possible. Tridacna louse bears eggs (their amount is about
several hundreds ones) in a pair of brooding pouches on the abdomen. The egg
laying can repeat up to ten times a year.
The larva of this species is free-living; at this stage tridacna louse has
an only opportunity to settle to new habitats and to find a shell of the host
mollusc. As against adult individuals, it is very mobile and at the certain
stage of development is able to swim – the back pair of its walking legs is
transformed to swimming legs because pleopods are modified to organs of attachment
even at early stages of development. Larvae of tridacna louse keep for some
time on the abdomen and back pair of walking legs of the female, but they leave
mother rather quickly – right after the first moult. In the first weeks of
life swimming legs are not advanced, and larvae of this species live on the
surface of reef. The mouth parts of larva at this time are typical for isopods,
and larvae eat algae growing on the surface of reef. In addition to algae they
eat small sedentary animals – hydroid polyps and bryozoans. At this stage of
life larvae are prey for numerous predators, and at early age the majority
of larvae of this species perishes. After several moults at them swimming legs
develop, and larvae of tridacna louse start active settling. When the large
fish swims near, larvae quickly swim to it and cling to its body. But larvae
do not parasitize on fish, and use it only as means of transportation. Being
attached to the body of fish, the larva catches smells of inhabitants of reef.
Having felt a smell of the probable host, it is unhooked from a fish, moves
down to the bottom and begins search of the mollusc. Having got on outgrowths
of mantle of “blossoming tridacna”, the larva undergoes metamorphosis during
the next moult; its swimming legs turn to walking ones, and mandibles form
a proboscis.
River driftwood borer (Fluvilimnoria xyloterebra)
Order: Isopods (Isopoda)
Family: Limnoriidae (Limnoriidae)
Habitat: rivers of Iceland, Northern and Northwest Europe.
The rivers of all continents, except for Antarctica, take out in ocean daily
hundreds trunks of various species of trees, and also numerous nuts and other
fruits in a ligneous environment. In addition at the coasts of the tropical
seas mangrove trees grow. The combination of lignificate vegetative material
and sea water had caused a special ecological niche for sea animals settling
in wood and drilling it. In sea water numerous species of “ship worms” live
– these ones are bivalve molluscs drilling wood by rudimentary shell. In addition
to them, isopod Limnoria was engaged in drilling of wood in human epoch. In
Iceland where the flora and fauna had been actually generated from zero after
the ice age, an ecological niche of aquatic driftwood-drilling animal had been
empty for some time. On land capricorn beetles settled to this island from
Europe play this part, and in fresh water their “colleague” had soon evoloved
– river driftwood borer, a separate species of crustaceans descended from one
species of genus Limnoria, the sea inhabitant.
River driftwood borer is rather large isopod species; length of adult individual
is about 20 mm. Body of this animal is flattened and extended in great degree;
because of it this one it can be misinterpreted as any variety of millipeds
if it should live on land. River driftwood borer has wide flat head with strong
mandibles and very short antennae. It drills tunnels in wood mainly with the
help of maxillipeds. Legs are short and strong; with their help this crustacean
easily moves in the tunnels forward and back. In connection with this ability
the last pair of walking legs of river driftwood borer is lengthened like antennae.
During the reverse movement of animal these legs feel the way. Eyes of this
crustacean are very small, presented only by several tens facets from each
side of head. The body of animal is covered with pale grey chitinous cover
and is transparent: tubular heart appears through the armour.
This species of crustaceans eats bacteria and fungi developing in rotten wood.
Mandibles of animal allow scraping a surface of wood, and the intestines form
the similarity of spiral valve characteristic for sharks. This adaptation extends
a way of food in intestines and enables to take from it all edible content
in fullest degree. The crustacean throws undigested sawdust out in water, cautiously
putting out the back end of body from an entrance of its tunnel. From time
to time river driftwood borer makes “cleaning” in the tunnels, pushing out
from them the sawdust gathered at drilling, by the back end of body.
River driftwood borer is a dioecious animal. The female of this species is
larger a little, rather than male (usually about 5 mm longer) and it has wider
back end of the body. It bears about 30 – 40 eggs in brooding pouches at the
basis of back pair of walking legs. Development of this species is direct,
and larvae are rather large – their length reaches 2 – 3 millimeters. They
spend first two weeks of life on the body of mother, and then settle in its
tunnels. Passing to independent life, young animals leave tunnels and creep
on river bed, searching for the driftwood. At this stage larvae settle very
actively. They differ from adult individuals not only in size, but also in
shorter body and longer legs. Larvae can endure sea water during long time,
and frequently appear at the ocean with floating pieces of wood. Cold coastal
current Antigulf Stream carries trunks with larvae along the Atlantic coast
of Europe where pieces of wood are cast ashore by storm. In river mouths larvae
feel the fall of salinity of water and begin to move against current. River
driftwood borers can eventually rise along river channels up to river heads,
and move from one river basin to another through bogs.
“Sea
broomer” (Pelagoniscus chaetopus)
Order: Isopods (Isopoda)
Family: Sea centipedes (Thalassomillipedidae)
Habitat: Sea of Okhotsk, floating macroalgae.
Thickets of floating algae at th surface of sea of Okhotsk serve as a perfect
habitat for various slowly moving, creeping and even sedentary animals though
they are located at the great distance from the coast. During millions years
in thickets of brown algae the original inhabitants evolved, which do not live
at the seacoasts. In addition the life far from the coast above the great depths
promoted development of swimming ability at benthic animals. Some species are
able only of short rushes in thickness of water, and others are able to swim
as much, as necessary, only for a short time sitting on algae for rest. Among
the animals developed a new way of movement there were representatives of isopod
order. One species of swimming isopods is a long-bodied crustacean named “sea
broomer”, the representative of separate family of sea centipedes, which representatives
resemble their terrestrial namesakes in lengthened body and numerous pairs of
legs.
When “sea broomer” swims in thickness of water, it is easy to think it as any
shrimp or small fish. It has thin flexible body about 4 cm long, including the
increased number of thoracal segments (15) bearing short legs. During the swimming
they are pressed against the body and do not brake movements of animal. Head
of “sea broomer” is flattened and wide. The principal organ of swimming at this
one includes two forward pairs of walking legs. They strongly differ in structure
from other legs: their surface is covered with elastic bristles forming a kind
of small brushes on top and bottom sides of leg. These brushes stretch in the
same plane, giving the feather shape to two pairs of legs of this crustacean.
On maxillipeds of this crustacean there are also bristle brushes. This is an
adaptation for feeding: during the swimming of crustacean legs “sweep” plankton
which is gathered on brushes. From time to time one or another pair of legs
stands motionless, and at this moment maxillipeds rake up got stuck planktonic
organisms and transfer them into the mouth. “Sea broomer” swims with the help
of the same movements, but at this time maxillipeds are pressed against the
head from below and do not prevent movement. If it is necessary to swim faster,
“sea broomer” helps itself with antennae, also are covered with bristles.
The back pair of limbs (uropods) does not take part in movement. These legs
are lengthened; their size reaches half of body length of animal. They are forked
and also are covered with bristles. These limbs serve for balance keeping in
water. When “sea broomer” stops, it stretches uropods in sides and opens their
branches. In such position they work as some kind of “parachute”, which is slowing
down the immersing. If the crustacean gets in rich enough congestion of plankton,
it drifts in such position, from time to time catching a portion of plankton
in forward pairs of legs. During the swimming branches of uropods are densely
pressed to each other, and their resistance to movement is minimal.
“Sea broomer” spends a significant part of life in thickness of water. The body
of this crustacean is transparent, with yellowish shade and small black spots
on the top part of body. When “sea broomer” rests on algae, it is almost imperceptible.
This animal is able to creep on algae, but uses them only as temporary shelter.
This species is dioecious; male is a little bit smaller, than female, and also
differs in tapered back end of body. After fertilization the female lays a portion
of 40 – 50 rather large eggs; the egg laying repeats up to seven times a year
with a break in winter months. Young animals spend first some days of life on
walking legs of the female and on the bottom side of her body. They leave mother
after the first moult and live in algae thickets for some time. At the length
of about 1 cm at them swimming legs develop, and they begin able to eat plankton.
At the age of 6 months young “sea broomers” become sexually mature. Life expectancy
of this species does not exceed 4 years.
Garbaging
Woodlouse (Hirsutopus scatophaga)
Order: Isopods (Isopoda)
Family: Woodlice (Porcellionidae)
Habitat: Forested areas of New Zealand, colonies of wasps.
The nests of Neovespula wasps not only
bear parasites, but some commensals and symbiotes. One such creature is the
Garbaging Woodlouse, descended from the introduced Porcellio woodlouse, brought
to New Zealand during English colonization.
These woodlice look somewhat different than their ancestor, bearing a coat of
hairs on their underside to help retain moisture. The rest of the body resembles
a typical woodlouse (Porcellio), though slightly bigger and fatter, reaching
3 centimeters long and having a brown colour, occasionally reddish individuals
are born. This species exists as a symbiote and commensal of the Neovespula
colony, by feeding on wasp droppings and food scraps, individuals are universally
tolerated by the wasps. Behavior involves seeking out the most humid parts of
the nest when not feeding, individuals may cluster together in crevices in the
nest, to conserve moisture when not seeking out food.
Females carry up to 80 fertilized eggs in a brood sack held under the abdomen
for about 40 days. Babies mature in about 2 months, and lifespan is about 3
years. Predators within the colony occasionally include kleptoparasitic beetles
such as the Deceiver Beetle.
This species of invertebrates was discovered by Timothy Morris, Adelaide, Australia.
Aotearoan
Cave-loving Woodlouse (Spelaeopoda spp.)
Order: Isopods (Isopoda)
Family: Porcellionidae (Porcellionidae)
Habitat: New Zealand, North Island, deep parts of various cave systems. Numerous
similar species within this genus inhabit different cave systems.

Picture by Biolog
Introduced species are often generalists, and it is in this
way that they come to dominate in new lands, but even these may pass into specialized
forms during the course of evolution. So we see with Porcellio woodlice, which
have come to colonize cave systems and become true troglobiota.
Aotearoan Cave-loving Woodlouse measures about 2 centimetres long, in shape
it mostly resembles its ancestor, apart from smoother, thinner-shelled carapace.
Coloration is a yellowish white, legs and underside are paler. This crustacean
feeds mostly on biofilms of bacteria, algae and fungi that grow in damp stone
surfaces in the depths of caves; they also feed on any dead organic matter such
as carrion. Individuals are very tolerant of each other and may cluster together
to conserve moisture when the cave air becomes dry. Legs of this animal are
tenacious and it is able to climb cave walls fairly well.
Breeding occurs year round. Males find females by following their scent trails.
The female will lay up to 50 eggs and carry them in a sac that she carries on
her underbelly; they hatch within about 40 days. Sexual maturity and full size
is reached at about 3 months, lifespan is up to 4 years. Predators include large
mites, spiders, and harvestmen, individuals that fall into bodies of water are
consumed by fish and amphibians.
This crustacean species was discovered by Timothy Donald Morris, Adelaide, Australia.
“Phantomic
murderer” (Vitromantella microhorrida)
Order: Stomatopods, or Mantis shrimps (Stomatopoda)
Family: Pedomantids (Pedomantidae)
Habitat: Indian Ocean, tropical and subtropical latitudes.
Pedomorphosis is the special way of evolutional transformations when the adult
stage is gradually lost and replaced by previous larval one. This phenomenon
is closely connected with pedogenesis – the aquiring by larvae of ability to
breed not turning to adult stage.
In the epoch of global ecological crisis there was an extinction of plankton
organisms and inhabitants of coral reeves. Descendants of a few survived genera
(mainly inhabitants of seas of moderate climate zone) occupied the exempted
habitats in next epoch of climate stabilization. Among them branches which representatives
developed a plankton habit of life had been separated. Such process occured
simultaneously in several groups of sea crustaceans, therefore in Neocene plankton
there is a plenty of species of “adult larvae” of various crustaceans belonging
to different groups. Among decapods the family of infantoportunid crabs (Infantoportunidae)
had appeared, and mantis shrimps had “answered” this step of crabs with occurrence
of plankton species of pedomantids.
It was easier to stomatopod crustaceans to pass to such way of life, because
all their species in development have plankton larva. The result of such evolutionary
step became the occurrence of stomatopod species which had received the name
“phantomic murderer”. This animal is a plankton predator about 4 – 5 cm long.
At it there is translucent body, poorly distinct in water – it is a common attribute
of the majority of plankton organisms. In anatomy of “phantomic murderers” many
features characteristic for a larva of benthic mantis shrimps are kept.
The main hunting weapon of the “phantomic murderer” is the pair of catching
legs, characteristic for representatives of the order. They are big relatively
to the body size – the span of completely opened and extended legs exceeds the
length of animal almost twice. Catching legs of this crustacean are very thin;
their last segment has a small hook on the tip. Thorns with which help the animal
keeps and kills catch, are thin and sharp, similar to bristles. Food of “phantomic
murderers” includes various small plankton animals with soft and thin body covers,
and it is not necessary of the big efforts for their capture.
This predator has very good sight. Eyes of “phantomic murderer” are large, spherical
and shining; they are located on thin stems – it improves the field of view
and simultaneously facilitates soaring in thickness of water. Facets are small;
therefore the animal sees even tiny plankton inhabitants very well.
On the head of the “phantomic murderer” there is long thorn (its length is a
half of thorax lenght), directed forward. On it from both sides hairs grow,
allowing soaring in thickness of water without efforts.
Abdomen of this crustacean is very thin and rather short: its length makes about
a half of general length of the crustacean (at benthic mantis shrimps abdomen
is much larger and much wider, rather than thorax).
Not hunting animal slowly floats in thickness of water, flapping by wide fanlike
“oars” (propodits of 4-th pair of mouth feet). They are long (their length makes
about the one fifth of general length of animal), covered with hairs on edges.
With their help the crustacean turns in water, being aimed on prey. Sometimes
“phantomic murderer” passively soars in thickness of water, having stretched
propodits in sides. If necessary to swim faster (for example, attacking the
prey), the crustacean can increase speed, moving by strong abdominal legs. Abdominal
legs are covered with hairs on edges, which increase a rowing surface. On the
tip of abdomen there is a wide telson plate. Last pair of abdominal legs is
also wide and flattened. Joining the telson they form a fin having a role of
rudder of depth and turn.
This crustacean is an active predator, hunting small plankton animals – worms,
crustaceans, fish fry, larvae and young cephalopods. For “phantomic murderers”
the cannibalism is very characteristic – about a quarter of young animals of
this species become the prey of their congeners. The crustacean catches prey
by sharp throw (accelerating momentum about half meter per one second), and
seizes it by trap legs. Per one day the “phantomic murderer” eats the amount
of food approximately as much, as it weighs itself. But actually it kills much
more animals, because it eats only the richest with nutrients parts of preyed
ones body and leaves the rests.
This crustacean is the solitary animal showing interest to congeners only during
hunting and in breeding season. Pairing at the north of an area (in tropical
latitudes) occurs at any time year; at the south it takes place in summer.
Walking legs at different genders of “phantomic murderers” are advanced unequally:
it is connected to their different roles in breeding. At the female all three
pairs of walking legs are advanced though they do not participate in walking:
the crustacean lives far from coast above the big depths. The female bears eggs
(up to thousand fine eggs in one clutch) on walking legs covered with hairs.
At the male the last pair of walking legs turned into short strong hooked appendages
is advanced only: they serve for keeping of the female at pairing. First pairs
of walking legs are reduced up to thin and weak hooks.
Breeding and development of this crustacean is connected to lunar cycles. The
peak of sexual activity falls to a full moon and some days after it. The incubating
lasts about one week, and from eggs plankton larvae hatch, at once leaving the
female. The larva moults about 10 times before it will turn to adult crustacean.
The cycle of development of “phantomic murderer” lasts about 2 months, and its
life expectancy usually does not exceed half-year.
Cricket crayfish (Symbiosquilla
innoxia)
Order: Stomatopods, or Mantis shrimps (Stomatopoda)
Family: Lysiosquillids (Lysiosquillidae)
Habitat: mollusk-spongial reeves of Indian and Pacific oceans, shells of
huge bivalves.
Plancton communities of ocean form an enormous biomass. And the greatest
variety of species is concentrated at the reeves forming in shallow waters.
Reeves
of Neocene are constructed not by corals, but by other live organisms: practically
all reef-building corals had died out in epoch of ecological crisis at the
boundary of Holocene and Neocene. The reef of Neocene is formed mainly by
slowly growing and living a long time massive bivalves and sponges. But it
is as rich
in life, as coral reeves of Holocene were. Various predators, often highly
specialized are richly presented here: it indicates a variety of food resources.
At reeves of Neocene mantis shrimps of various species live. In epoch of
ecological crisis a lot of species of these crustaceans had died out: all
of them pass
a part of life cycle in plankton as a floating larva.
In majority mantis shrimps are solitary predators and active cannibals. But
among them there is a small amount of species living in breeding pairs in
common shelter. Obviously, from any of similar species one kind of mantis
shrimp living
on reeves had evolved – the cricket crayfish. It is a tiny species of mantis
shrimps – the length of adult crustacean is about 3 – 5 cm (the male of this
species is appreciably larger, rather than the female). Such creature would
have a lot of enemies on a reef. But it had adapted to inhabiting in perfectly
protected place - in huge shells of goblet-like
false rudistes, which tower
above the body of reef, as if fortifications. Pair of adult animals lives
in each such shell, but occasionally in very large shell of old mollusk one
male
and two or three females settles.
Life in safety and twilight of shell of huge mollusk has resulted at the
appearance of this crustacean. The majority of mantis shrimps living in shallow
waters
is colored very brightly or has the cryptic colouring which harmonizes with
color of environmental district. And the cricket crayfish resembles any cave
animals by the shape: at this shell inhabitant the body is depigmentated,
translucent and brownish. Eyes of this crustacean are very small, and sight
sense is much
weaker, rather than at freely living mantis shrimps. Long antennae help the
animal to orientate itself in the shell.
The cricket crayfish is, perhaps, the most peaceful species among stomatopods.
Inside shells where it lives there is no catch, characteristic for the diet
of its freely living congeners. Its trapping limbs are modified to scrapers
with which help these crustaceans gather settling suspension from gills of mollusk.
It is the basic food of cricket crayfish. On trapping legs of this animal many
tiny denticles, not adapted to pierce and kill catch, grow. Presence of such
small symbionts renders the big benefit to mollusk: these crustaceans clear
its body of parasites, eat worms and smaller parasitic crustaceans which injure
gills of goblet-like false rudistes.
The population of shell protects the habitat from neighbours. Except for
trapping legs, in set of natural weapon of mantis shrimps there are “clubs”
on pectoral
legs with which help typical stomatopods break shells of mollusks. This crustacean
uses them in intraspecific conflicts. Cricket crayfishes living in one shell,
learn each other by smell. The number of animals living in same shell is
determined by frequency of their meetings with each other. At too often meetings
crustaceans
turn more aggressive, and start to attack against each other. Thus the weakest
animal abandons shell, or may be killed. “Indigenes” drive off from the habitat
of unfamiliar neighbours, trying to lodge in shell, and can attack larvae
of their species, getting to the shell in current of water. Avoiding an opportunity
to enter fight, these mantis shrimps show in the beginning their presence
to
unfamiliar congeners. They utter warning sounds, passing by mouth feet along
denticles of trapping legs. Such sound is similar to chirring of some orthopters
– this feature combined with long antennae and secretivity has determined
the name of the animal. Protecting the shell, these crustaceans creep out
to the
edge of shell of mollusk in which they live, keep in shadow of the closing
fold and chirr continuously. If the neighbour does not leave the shell, fight
which can be finished by fatal outcome is possible. Protecting the shell,
the cricket crayfish can leave it for a short time. It swims with the help
of abdominal
legs fast and manoeuvrably. Such small creature can put strong and even fatal
impact to small animals by “clubs” on armour or head of the opponent. It
lasts a split of second, and stuns or kills the opponent. Attacking predatory
fishes,
the animal quickly comes up from the shell, “clicks” in nose or eye of fish,
and immediately hides back.
Under the protection of shell the cricket crayfish breeds the year round.
In shell only one male can live. He fertilizes all females living in its
shell,
and even visits for this in next shells. The female has wider abdomen, than
the male, and bears some hundreds of small eggs for one clutch. The incubating
lasts about one week; at this time the female is very cautious, and does
not take place in habitat protection, because it swims much slower. The egg
laying
repeats each two weeks.
Transparent plankton larvae hatch from eggs. The larva spends the time up
to the first moult on abdomen of the female. Then the female creeps out on
edge
of shell and throws larvae off in water, intensively moving by abdominal
legs. So the plankton stage of development of young crustaceans of this species
begins.
It is reduced in comparison with time of development of freely living congeners.
The larva spends some days in plankton above the reef; at this time it is
settled and with the help of sea current may overcome the big distances between
islands.
Due to this ability the cricket crayfish is widely settled in tropical area
of Pacific Ocean and has penetrated to the eastern part of Indian Ocean.
The animal occupies pallial cavity of huge mollusk, getting in it with a
current
of water while the mollusk opens shell and lets out lobes of pallium with
symbiotic algae. If it manages to occupy the shell, after 2 moults it will
reach the
half of length of adult crustacean and will completely lose larval features.
At the age of 4 months the young animal is able to breed.
Life expectancy of cricket crayfish is insignificant and exceeds two years
seldom.
Umbrella
shrimp (Umbellocaris furtivus)
Order: Decapods (Decapoda)
Family: Palaemonids (Palaemonidae)
Habitat: Pacific Ocean, top water layers.

Picture by Fanboyphilosopher
Plankton is the special community of live organisms. Here
there are species of predators and scavengers, parasites and symbionts. The
competition in plankton is very strong, and predators of different trophic levels
are diverse. Therefore various live organisms enter the unions with each other
to resist to predators in common. Often one well armed species becomes the involuntary
defender to tens species of smaller animals. Such unions are not always safe.
Jellyfishes are one of the most widespread animals in plankton. They are a food
for many larger predators, but nevertheless they are not too defenceless. Stinging
cells of jellyfishes save these animals of the majority of predators, and only
few specialized species can unpunishedly eat jellyfishes or parasitize
on them.
Animals looking for protection at jellyfishes must be cautious – the casual
touch of tentacle may transform them to easy dinner of this animal. But there
are live creatures venturing to live in dangerous affinity from poisonous tentacles
of these coelenterates.
Under the bell of jellyfishes it is possible to find often the small shrimp
keeping by thin hooked pincers for edge of the jellyfish bell. For this feature
it received the name of the umbrella shrimp: it looks like hidden from the sun
under the umbrella of jellyfish. This is small crustacean: length of its body
is about 5 cm. Body of umbrella shrimp is translucent and has bluish shade.
Therefore it is absolutely imperceptible in water. Only sometimes, when the
jellyfish emerges to water surface, the sun shine in round eyes indicates the
presence of this crustacean.
Pleon of this animal is very short and has advanced swimming legs. Walking legs
are not adapted to movement on firm surface. They are covered from edges with
rigid hairs and help the shrimp to swim. But nevertheless this crustacean spends
the most part of life having clung to the bell of jellyfish. At the umbellate
shrimp two pairs of pincers twice exceeding length of body are well advanced.
Tips of these pincers are very thin and long. The forward pair of pincers is
used more often to keep for edge of the jellyfish bell. In case of necessity
to hide from the predator, umbrella shrimp bends joints of these pincers, and
by one somersault hides under umbrella of the jellyfish. By second pair of pincers
of the same length the umbrella shrimp “handles” tentacles of the jellyfish
on which it lives. It takes off from tentacles and eats small animals had got
to the jellyfish. Hence the specific epithet of this shrimp meaning “thievish”.
Such habit of life is possible for it because this shrimp is tolerant to poison
of jellyfishes.
Umbrella shrimp spends all life on jellyfishes. If the host jellyfish perishes,
shrimp can move to other jellyfish. It swims very reluctantly and mostly soars
in water, having stretched antennae in sides. When the jellyfish approaches
to it, shrimp seizes it to the edge of bell. First time umbrella shrimp gets
accustomed to poison of new host jellyfish and develops immunity. It cautiously
touches tentacles of the jellyfish, receiving easy stings. At this time the
immune system of shrimp accumulates antibodies to poison of jellyfish. Having
got accustomed to new host jellyfish, shrimp starts to live habitual life, taking
away a part of catch right from tentacles of jellyfish.
Umbrella shrimps are strongly expressed egoists. Each individual protects the
jellyfish from contenders, getting rid them from bell and pinching by pincers.
If the opponent is stronger, the crustacean seizes edge of jellyfish bell by
all pincers waiting while the contender will leave out. Sometimes after such
passive defense the part of legs and antennae of shrimp appears bitten off,
but they quickly grow after the molt.
But for the sake of breeding umbrella shrimps are compelled to overcome individualism
and to search each other in the ocean. The female stays the homebody, but the
male should undertake risky travel to thickness of water in searches of the
female. It folds long pincers under the body and swims using walking legs. Male
searches for female by smell and leaves the jellyfish only when the female ready
to pairing is felt absolutely beside. At the male there are long antennae at
the basis with numerous hairs. They are rich in chemoreceptors; therefore male
precisely finds the necessary female at the distance of several meters in congestion
of jellyfishes. Having found the female, male as soon as possible must couple
with it. But for this purpose it should convince the female of peace intentions.
At meeting with the female it is very cautious: when the meeting of the male
and the female takes place, the male cautiously touches the female by tip of
antenna. It strokes the cephalothorax of the female in the area of eyes and
maxillipeds, accurately approaching nearer to the female. Courtship ritual lasts
till some minutes. Having convinced that the male does not express aggression,
the female supposes it to itself. Pairing proceeds fast: male clings to female,
leaves on its pleon two large spermatophores and departs. More often it can
not return back to the “own” jellyfish, and after each pairing is compelled
to search new one. But during one “wedding travel” male has time to fertilize
several females.
In some hours after pairing the female lays large eggs and bears them on abdominal
legs. At the umbellate shrimp there is direct development: tiny copies of parents
burst from eggs. This species has a little posterity in comparison with other
plankton animals, but young shrimps are very much advanced. They perch on abdominal
legs of the female till some days, then abandon the mother and begin independent
life. Since the first days of life young shrimps start to cooperate with jellyfishes.
First they live on bell of jellyfishes and clear them of parasites – infusorians
and microscopic worms – for any time. Then young umbrella shrimp move on edge
of its bell and start to eat its catch.
This shrimp grows very quickly – at three-monthly age the young shrimp reaches
the size of adult one and starts to breed. Every five – six weeks the female
makes a new clutch.
Dragonfly prawn (Libellulocaris prolongus)
Order: Decapods (Decapoda)
Family: Penaeids (Penaeidae)
Habitat: Pacific Ocean, top water layers.
Waters of open ocean represent very specific habitat. Here it is almost impossible
to find a point of support, and the majority of plankton inhabitants is adapted
to soaring or swimming in water thickness. Some sea inhabitants spend in water
thickness only a part of life cycle, and others constantly live in this world.
All of them form the special community named plankton. All plankton creatures
have one common property – they are not able to resist to current, and constantly
wander in ocean.
In plankton constant struggle for existence, not less rigid, rather than in
the world of big creatures, takes place. Peaceful vegetarians eat plants –
microscopic algae growing in top layers of water. In turn, they serve as food
to various predators. Their activity is so great, that plankton inhabitants
practically do not have chance to die a natural death. Some predators are passive,
and others chase the planned prey actively. One original prawn had received
the name dragonfly prawn for features of appearance and habits concerns to
number of active predators of plankton.
The length of this crustacean reaches approximately 10 – 13 cm. Body of dragonfly
prawn is long, thin and transparent. Through covers of its body it is visible,
as heart contracts, and in intestines the digested food moves. This creature
becomes well appreciable when it appears in day time in the top layers of water.
Then its huge spherical eyes on short stems brightly shine. Eyes of dragonfly
prawn are rather large in comparison with the size of its body, and also number
hundreds thousand tiny facets. Due to it dragonfly prawn easily distinguishes
even the slightest movement in thickness of water. By casual light shine on
body covers it finds plankton inhabitants which often are almost completely
transparent. Organs of sense of smell are located on long antennae. When this
prawn chases prey, antennae are pressed to the body.
Land dragonflies chase catch using opportunities of wings. Dragonfly prawn,
certainly, has no wings, but it does not prevent it to chase catch. This crustacean
swims with the help of strong abdominal legs. In connection with such way of
movement the body of dragonfly prawn had partly changed: the cephalothorax
of this crustacean became very short (its length is about one fifth of the
general body length), but the pleon is strongly extended. At dragonfly prawn
segmentation had changed - the number of pleon segments reaches one and half
tens. The fin formed by back pair of abdominal legs serves as rudder for sharp
turns – catch of dragonfly prawn often makes sharp jumps aside, forcing predator
to stop chasing.
Usually small plankton crustaceans and larvae of fishes become food of this
prawn, but occasionally this prawn can catch fry, large crustaceans or floating
mollusks. Walking legs of dragonfly prawn are one more feature giving to this
crustacean similarity to winged prototype from among insects. At pelagic habit
of life ability to walk appeared useless (land dragonflies can not walk too),
but legs had got other, much more useful function. On internal surface of all
five pairs of walking legs at dragonfly prawn lines of long sharp spikes are
developed. Put together, legs represent similarity of fishing-basket. For capture
of small animals dragonfly prawn simply rushes through their congestion and
catches them by some ones at once, not undertaking special efforts. Usual dragonflies
catch their prey, midges and mosquitoes, the same way. Larger animals also
can not avoid deadly “embraces” of this prawn – it grasps large animal and
strongly compresses it by legs, piercing through by spikes.
When prey is caught, maxillipeds are used. Bristles on their bottom side gather
tiny plankton animals to one clod which is swallowing by animal. The prawn
dismembers large catch with the help of cutting edges of maxillipeds. For one
day the prawn can eat up to ten rather large fish fry or smaller animals in
amount approximately equal to body weight of this animal.
The dragonfly prawn is a predator and active cannibal; therefore it does not
form congestions in plankton. Only in breeding time the male searches for the
female not having the purpose of attack. The male of this species differs from
the female in larger back pair of walking legs with which help it keeps the
female till the pairing.
The female ready to pairing emits the special substance involving males. By
odorous trace they find it – often near one female some competing males gather.
One of males (as a rule, the largest one) drives off others and pairs to the
female, and then swims away. The fertilized female stops the odorous substance
emitting, and becomes dangerous to own congeners again.
Tiniest eggs (up to 20 thousand grains of roe) are bearing on abdominal legs
because of what the female loses speed of movement and lives half-starving
for some time. Through 3 – 5 days from eggs translucent plankton larvae with
long back outgrowth facilitating soaring in thickness of water burst. They
are predators, and immediately start to eat unicellular organisms. At one month
age the larva finally turns to tiny copy of adult animal. But to this time
up to 90 % of larvas perishes of various predators including their own congeners.
Sexual maturity comes to them at the age of 7 months. After that the adult
female regularly starts to lay eggs with an interval equal to lunar month.
The maximal life duration of dragonfly prawn does not exceed one and half years.
Tentacle prawn (Furocaris toxiresista)
Order: Decapods (Decapoda)
Family: Penaeids (Penaeidae)
Habitat: warm and temperate areas of Indo-Pacific.
In Neocene plankton fauna had undergone some changes in comparison with Holocene.
The reason of it had been the mass extinction connected to global ecological
changes. In early Neocene when conditions of life had stabilized, planktonic
habit of life had been developed by plenty of new groups of animals. They had
been mainly various descendants of coastal and shallow-water forms. Shrimps
are one of typical inhabitants of plankton in epoch Neocene. Among them there
is a set of various species – from peaceful filtrating organisms up to predators.
Some species of planktonic shrimps had entered symbiotic relations with other
animals living in plankton. Generally speaking, forms of relations of shrimps
and their neighbours change from symbiosis up to predating with various variants.
One of such variants is shown by tentacle prawn living in waters of Southern
hemisphere.
It is rather large carnivorous prawn about 15 centimeters long. It keeps near
to congestions of jellyfishes, and even constantly lives in tentacles of some
species of these coelenterates. Body of this crustacean is lengthened; cephalothorax
is rather short. Tentacle prawn is a little similar to another pelagic crustacean,
its distant relative, dragonfly prawn. Antennae of tentacle prawn are very
long – they exceed body length of this crustacean. Walking legs of this prawn
are covered with small bristles and adapted to drifting in water – tentacle
prawn frequently floats, keeping for a tentacle of jellyfish by one claw, and
having freely stretched legs in thickness of water. Also with the help of such
legs shrimp easily keeps on tentacles of jellyfish on which it lives. At the
male the back pair of walking legs is larger, than others: these legs serve
for keeping on the female during pairing.
In spite of the fact that this prawn is rather large, it is difficult for seeing
in water. Armour of animal is almost transparent and has slight bluish shade.
Through covers of this prawn it is perfectly visible, as its heart pulsates
and food moves in intestines. A parameter of refraction at tissues of tentacle
prawn is almost same as at sea water, therefore small grey spots on pleon segments
and shining dark eyes are only things visible at this crustacean in water at
dim illumination.
The food of this prawn consists mainly of prey captured by jellyfish – the
main sense of its relations with jellyfishes consists in it. Tentacle prawn
constantly swims near jellyfish, inspecting its tentacles. This occupation,
however, is unsafe – tentacles of jellyfishes are poisonous. But tentacle prawn
has strong immunity to poison of jellyfishes. This immunity is acquired, and
is developed during some time. At first tentacle prawn cautiously moves nearer
to jellyfish, touching up to its tentacle by antennae. Usually at this moment
it receives weak burn of stinging cells. After attack of jellyfish the prawn
does not swim out, but stays near this animal, and even sits on its dome from
above, outside of zone of reach of jellyfish tentacles. Within approximately
one day prawn actively develops antibodies and gets immunity to poison of present
species of jellyfishes, if it had not it earlier. After that it actively creeps
in tentacles of jellyfish in searches of its prey. Immunity is kept to whole
further life, therefore the “skilled” prawn had replaced host jellyfishes of
several species becomes a universal predator. Sometimes this prawn can harm
to jellyfish: it simply bites off a tentacle in common with prey, and eats
it entirely. Poison of jellyfish accumulates in liver of prawn, and additionally
makes it inedible.
Tentacle prawn is a solitary animal. It concerns to congeners neutrally, but
at the case of lack of food it can drive them away from jellyfishes on which
it is fed by impacts of rostrum.
This prawn is the descendant of pelagic animals, and has kept a way of breeding
characteristic for them. After short courtship ritual and pairing female lays
set of tiny eggs – up to several thousand ones. 3 – 5 days later from them
tiny planktonic larvae hatch; they pass some stages of metamorphosis before
turning to tiny similarity of adult individual. Tentacle prawn breeds monthly
during half-year, and then in coldest months of year the break follows.
Young tentacle prawns lead a pelagic way of life. They gather to schools up
to several hundreds individuals, and avoid meeting with jellyfishes. At young
age their immune system is not ready to development of antibodies against poison
of these animals, and they frequently become prey of jellyfishes. Only at length
of about 5 cm young prawns become advanced physiologically enough to pass to
inhabiting on jellyfishes without danger to their own life.
Feather-legged shrimp (Baculophthalmocaris plumopes)
Order: Decapods (Decapoda)
Family: Penaeids (Penaeidae)
Habitat: northern part of Pacific Ocean (temperate and polar latitudes), top
layers of water.
In Neocene among oceanic plankton the essential place was occupied by decapod
crustaceans. In Holocene many species of these animals spent in plankton only
a part of life cycle. But in Neocene among them the true planktonic forms competing
to other groups of crustaceans had appeared. One of such species is tiny feather-legged
shrimp. This is pelagic crustacean adapted to inhabiting in water thickness.
It has translucent body, therefore it is difficult to notice feather-legged
shrimp in water. The body length of this crustacean does not exceed 3 – 4 cm.
This crustacean passively drifts in water thickness, having stretched in sides
long walking legs. The length of legs is almost equal to body length of this
crustacean. Legs from both sides are densely “feathered” with hairs – this
adaptation facilitates drifting in water thickness. Claws of feather-legged
shrimp are long: they twice exceed body length. In rest they are usually folded
along the body on its bottom side, and do not prevent movement of shrimp.
The pleon of feather-legged shrimp is short; it makes less than half of general
length of animal (not including antennae and legs). It is wide, and pleopods
are covered with hairs on edges and are adapted to drifting in water. When
feather-legged shrimp drifts in water thickness, pleopods are spread wide in
sides, increasing the common area of body.
The structure of eyes of this crustacean is remarkable. Eyes are rather big
– they are cherry stone-sized. They are located on long mobile stalks which
can move almost in any side. Eyes of spherical form give this shrimp the full
circular field of view. They are combined by set of tiny facets providing sharp
sight. Stalks of eyes also serve as the additional organs facilitating drifting
in water: the similar attribute was present at fry of deep-water fishes Idiacanthus
known to people. The animal can change position of body in water thickness,
moving by long eye stalks.
Despite of passive way of life, feather-legged shrimp is a predator. It catches
small invertebrates, seizing them by mobile claws and moving to mouth. Occasionally
it can make short throws to prey or to escape from a predator, rowing by pleon.
This species of shrimps is dioecious. Male is smaller, than female, and also
weighs almost twice less. It is able to swim actively, rowing by walking legs.
Pairing occurs quickly and is not anticipated by any ritual. In some days after
pairing the female spawns a lot of tiny eggs (up to 3 – 5 thousand ones), and
bears it on pleopods within two weeks. From egg pelagic larva (zoea) hatches,
which at once leaves the female. Within one year the female can make up to
5 – 6 clutches.
Development of larva takes about three months. Till this time it undergoes
up to 8 – 9 moults and gradually turns to tiny similarity of adult individual.
To the end of metamorphosis its length reaches about 20 mm. Two months later
it becomes sexually mature.
Ice shrimp (Cryopenaeus crystallinus)
Order: Decapods (Decapoda)
Family: Penaeids (Penaeidae)
Habitat: Antarctica, shallow rivers.
After thawing of glacier covered Antarctica during late Tertiary and Quaternary
periods, at this continent marshes, rivers and lakes had formed. The freshwater
fauna of Antarctica formed from zero – its former representatives were destroyed
by congelation. Settlers of fresh waters get to the continent in two ways –
by air (insects) and from the sea (crustaceans and fishes). In reservoirs of
Antarctica invertebrates take more significant place, rather than at other
continents – it is connected to small number of local fishes. Among local invertebrates
the species having various adaptations had appeared, permitting to survive
in extreme conditions which had developed at this continent.
In southern seas shrimps of penaeids family (Penaeidae) are usual. Usually
their representatives spend in fresh water only a part of life, and for breeding
necessarily migrate to the sea. Among freshwater animals of Antarctica these
shrimps are very usual. One of their species is ice shrimp.
Body length of this crustacean is up to 10 cm; male is smaller, than female.
The body of ice shrimp is almost completely transparent: through thin covers
internals, heart pulsation and movement of food in intestines are visible.
On armour small grey points are visible only. Such colouring provides good
masking. Only the bases of legs, mandibles and maxillipeds, and also eyes of
this shrimp are opaque. Long serrated rostrum, and also large reddish eyes
consisting of numerous facets are characteristic features of shape of the ice
shrimp. This shrimp is able to swim well with the help of pleopods. The forward
pair of walking legs is not transformed into claws, but they are thicker and
stronger, than the others. On bottom side of these legs sharp prickles, with
which help shrimp keeps food, grow.
Ice shrimp is a predator. It eats larvae of insects – dragonflies and two-winged
flies. This shrimp hunts from an ambush. Usually it hangs upside down on plants
floating on surface of water, or on stalks of water plants, and waits for occurrence
of prey. It catches small animals by throw; having seized prey, shrimp returns
to stalk and eats it, keeping by forward pair of walking legs. Having eaten
prey, ice shrimp simply lets off the rests, and they are carried away by current.
This species of crustaceans has adapted to survival in shallow reservoirs of
Antarctica completely freezing through in winter. Ice shrimp survives in winter,
having dug among rests of vegetation at the bottom. While the environment is
liquid, shrimp keeps any activity and can even slowly move in thickness of
silt impregnated with water, clinging by strong forward pair of walking legs.
When the reservoir freezes through to the bottom, this crustacean falls into
anabiosis and permits body liquids to freeze. In tissues of shrimp the substances
constraining formation of large ice crystals accumulate, and ice does not destroy
cells. In such condition ice shrimp can endure frosts up to - 40°С.
In spring, when reservoirs get warm enough, ice shrimps release from winter
captivity. They start to fatten actively, searching for larvae of insects fallen
into catalepsy, and sometimes eat their own congeners. Approximately one month
after clearing of reservoirs of Antarctica from ice, having restored forces,
these shrimps start to prepare for breeding. Males compete for females, arranging
fierce, but not fatal combats with the help of forward pair of legs. They grasp
by forward pair of legs leg or antenna of the opponent, and try to turn it
and to keep in such position. But usually the amount of males in population
is less, than females – they grow slower and more often become prey of other
water animals.
Ice shrimp breeds in sea water. Fertilized females migrate downstream, gathering
to numerous schools, numbering tens thousand individuals. At this time in ovaries
of females eggs develop; they which will be laid only in water of ocean salinity,
far from coast. There are many eggs in clutch – up to 50000 ones; but they
are very small. After young ones hatch, the female dumps the rests of clutch
from pleopods and moves back to the river. Females come back upstream along
the riverbank, where current is slower, and at this time do not gather in schools.
Using well advanced sense of smell, each individual finds the river in which
it lived before breeding. The egg laying repeats 2 – 3 times per polar summer,
but the mass breeding migration takes place in late spring. Males of this species
never leave fresh water.
Development of ice shrimp includes planktonic larval stage. After the metamorphosis
(at the age of 3 months) young shrimps enter rivers and move upstream. The
first year of life young ones stay in lower reaches and central channels of
rivers not freezing completely. Strong enough and mature individuals rise upstream
farther and settle in lakes and streams. One-year-old shrimps can breed; life
expectancy seldom exceeds three years.
Badger prawn (Pedocrangon minimeles)
Order: Decapods (Decapoda)
Family: Crangonid prawns (Crangonidae)
Habitat: rivers of Pacific coast of Asia - from Big Kurils and Far East coast
of Asia up to the south of Japan.
In Holocene prawns of crangonid family had started to develop freshwater habitats;
separate species of these crustaceans passed the bigger or smaller part of
life cycle in fresh water. Among Neocene crangonids species completely passed
to freshwater habit of life had evolved, and they had settled rivers and lakes
far from seas. Transformation of life cycle of these prawns was the result
of this act – they are lack numerous larval stages and the development had
to greater or lesser extent come nearer to ametabolous development. In Asian
rivers of Pacific Ocean basin the genus of crangonid prawns had evolved, which
had a special life cycle during which the individual changes gender depending
on circumstances. It is a genus of prawns of massive constitution, which representatives
have massive short claws adapted to digging. In dense ground composing riverbanks
they dig burrows in which hide during the day time.
Dark colouring and longitudinal white strip along the middle line of cephalothorax,
and also skill of burrow digging had denominated this species as badger prawn.
At this species of crustaceans the sexual dimorphism is sharply expressed.
The female of badger prawn is massive – up to 20 cm long, with short wide claws
and strong legs. It swims not so good, but walks on the bottom much better.
The armour of this crustacean is sculptured – on cephalothorax and pleon segments
of adult prawns quaint relief of knobs and grooves is expressed. On body numerous
tiny hooks and bristles are located. It is necessary for masking – badger prawn
fastens on itself parts of plants. Antennae at the female of this species are
short.
Male sharply differs from female. It is very small – not larger than 2 cm.
It is connected to features of life cycle of badger prawn. The gender at these
crustaceans is not fixed genetically, and connected exclusively to age of animal.
Badger prawn has ametabolous development: from eggs tiny similarities of adult
individual hatch. They keep on pleopods of the female within several days.
After the first molt young individuals ready to independent life leave female’s
pleon and the first year lead independent life. The next summer they reach
sexual maturity and every one of them turns to male. Males find mature females,
and sit to bottom side of their pleon. Walking legs of males have special attaching
claws. In breeding season (in the beginning of spring) male fertilizes the
female but does not leave her after that. He lives on her body as a commensal,
eats prey of female and clears her body of parasites. When the female bears
posterity, male cares of eggs and young prawns, sitting on bottom side of body
of the female. He clears eggs of dust, moves off dead or non-fertilized eggs
and protects just hatched posterity from small predators. When posterity leaves
the female, male also leaves off. Its male life is finished in this time, and
within one year after some molts its transformation into the female takes place.
After that transformation up to the end of life this individual will be the
female, and male of new generation will look after eggs, sitting on its pleon.
At the second year of life females are able to breeding. Active growth proceeds
at them up to the seventh year of life, and general life expectancy makes till
20 years.
Badger prawn digs holes, using claws and massive rostrum. Edges of claws of
badger prawn are covered with denticles, with which help animal loosens clay
ground and even “saws” roots of plants. The hole of this crustacean is made
at the depth up to one meter. The length of hole may exceed two meters. Summer
holes of this species are horizontal; individuals from southern sites of an
area dig similar holes. In areas of seasonal climate and in cold winter badger
prawn makes deep hibernating holes directed downwards vertically to the depth
up to 160 cm.
The idea about existence of present species of crustaceans is proposed by Nem, the forum member.
Antarctic
swamp false jabby (Antarctocancer palustris)
Order: Decapods (Decapoda)
Family: Antarctic false crayfishes (Pseudocancridae)
Habitat: meadows and marshy areas in Antarctica.
Antarctica of Neocene epoch is true “lost world”. It remained isolated from
other continents for a long time, and the whole terrestrial fauna of this continent
had been exterminated by glaciation held down Antarctica to millions of years.
As a result after climate warming in Neocene this continent had been settled
by terrestrial animals almost from zero.
Absence of ground mammals in New Zealand (before human colonization of islands)
had resulted in occurrence of giant flightless orthopters of Deinacridae family,
which became ecological analogues of rodents. In Antarctica the similar situation
had repeated. But insects have the limited opportunities for increase of size
because of features of anatomy of respiratory system. Therefore in Antarctica
they had lost to representatives of decapod crustaceans in struggle for existence.
In Antarctica there are no river crayfishes, fauna of sea crabs is very poor,
and ground crabs live far overseas, and their larvae can not get to this isolated
continent remarkable by cold climate. But in coastal waters of Antarctica there
are many kinds of shrimps. Some shrimps had developed life in fresh water, and
one genealogical line of these crustaceans had given rise to unique terrestrial
forms. Antarctic swamp false jabby is the largest terrestrial arthropod of Antarctica.
It is the true terrestrial species of crustaceans, the close relative of prawns
of crangonid family (Crangonidae). It is very large species not only to measures
of prawns – the length of this crustacean reaches 20 cm.
Appearance of Antarctic swamp false jabby is freakish and even grotesque. It
has rough granular armour and sharp denticles on carapace, growing in three
longitudinal lines. Pleon of this crustacean is short and wide; on each segment
a cross line of large knobs grows. Edges of carapace at Antarctic swamp false
jabby are convex, and on each side of the body extensive cavities are formed.
In them there are plicas of thin epithelium penetrated with blood vessels and
substituting lungs. The similar adaptation was present at terrestrial coconut
crab (Birgus latro) of human epoch. Due to such adaptation Antarctic swamp false
jabby well feels like on land many hours in succession. Nevertheless, this crustacean
is compelled to fill up stocks of water regularly. For this purpose Antarctic
swamp false jabby keeps near to marshes and various temporary reservoirs where
dives for wetting of respiratory cavity.
From four pairs of walking legs first pair is advanced rather poorly. These
legs are thin and mobile; with their help animal gathers food. The following
three pairs of walking legs are rather large and strong. They easily maintain
weight of body of Antarctic swamp false jabby on land. With their help animal
can even climb on bushes in searches of food – bird eggs and insects. Powerful
claws are inherited by this crustacean from ancestors – prawns of crangonid
family. Dactyl (“thumb”) is very short, and the propodus (outgrowth of penultimate
segment), forming the basic part of claw, is wide and shovel-like, with serrated
internal edge and knobby surface. With the help of claws Antarctic swamp false
jabby digs holes in which rests and molts.
Male differs from female in smaller size, narrow body and larger claws. At females
bottom side of claws is grey with white edge, and at males it is bright pink.
Besides female is colored darker, than male.
This crustacean has short antennae – their length does not exceed length of
carapace. Basal segments of antennae are thick and covered with tiny thorns.
In courtship season males of Antarctic swamp false jabby arrange combat for
females, pushing each other away with the massive antennae bases. Eyes of this
crustacean are large and located on short and thick eyestalks. The set of facets
provides good sight, but animal is short-sighted and in search of food uses
sense of smell first of all.
Antarctic swamp false jabby is a predator and scavenger. It eats terrestrial
invertebrates – insects and their larvae, and also its own smaller relatives.
It willingly eats nestlings of local flightless mousebirds
(Musornis spp.), pulling them out from holes. In autumn and in spring, when
in Antarctica day and night alternate, at night Antarctic swamp false jabbies
may creep out to the coast of ocean in searches of sea animals cast ashore.
This crustacean has almost interrupted the connection with aquatic environment,
and does not depend on sea water in breeding season at all. After pairing the
female lays large eggs (up to 400 ones about 6 mm in diameter) staying on land.
It bears clutch on pleon up to the hatching of young ones. Only just before
the ending of incubation female searches for fresh-water reservoir. At this
time it behaves very aggressively to any congener, attacking on it and expelling
from the reservoir occupied by her. Young jabbies hatch in water, and female
with young ones lives in shallow water till some time. After 5 – 6 days young
jabbies leave the female, and she comes back to land. To the end of summer young
Antarctic swamp false jabbies reach the length of 3 – 4 cm and leave a reservoir
in searches of place for wintering.
Features of Antarctic climate had forced this crustacean to develop special
forms of behaviour permitting to survive. Antarctic swamp false jabby winters
in deep hole (its depth is up to 3 meters) dug almost vertically. In hole there
are some horizontal ells closed by friable ground fuses – it provides thermo-insulating
and the minimal air change. Along the hole, depending on weather in forthcoming
winter, this crustacean arranges up to five fuses. Young individuals winter
in casual shelters, and frequently penetrate into holes of adult individuals.
In spring nothing threatens to them: while weather is cool, at adult individuals
appetite is reduced, and they do not pay attention to young growth, leaving
a hole. But in summer, when there is a polar day and the sun shines all day
and night, in meadows of Antarctica it is warm weather, and adult false jabbies
can eat young ones. Also young jabbies frequently become prey of terrestrial
birds and bird-catching sundew (Droserophyllum ornitivorum)
– large carnivorous plant.
Young animals reach sexual maturity at the age of 3 years at length of a body
about 15 cm. Life expectancy of this crustacean seldom exceeds 15 years.
Kōeke
Ana (Speleatya pellucida)
Order: Decapods (Decapoda)
Family: Atyid shrimp (Atyidae)
Habitat: Cave waterways within glacially made cave systems, some areas downstream.
North and South Island of New Zealand.
Freshwater crustaceans of many sorts are notable for being hardy and ubiquitous.
The common shrimp Paratya curvirostris of New Zealand managed to survive the
onslaught of predation by invasive fishes, by its wide distribution and unpretentious
diet. Descendants in the Neocene incude cave-dwelling specialists like the Kōeke
Ana (meaning “cave shrimp” in Maori).
This small shrimp is somewhat like its ancestor in appearace, apart from being
smaller (usually no longer than 3 centimetres) and having completely transprent
body tissues and exoskeleton. Another difference is that eyes are very reduced,
eyestalks are very sort and only bear small clusters of lenses that merely allow
to distinguish light from dark. The first pair of feeding legs is modified into
a hairy brush-shape at the end, this is connected to its diet. It gathers organic
detritus and algal films with these specialized forelegs, and passes this food
to the mouth, it will also eat very small invertebrates, and eachothers young.
Able to hover in the water by undulating its pleopods, if alarmed or chased
by a predator it can flex its bod rapidly to dart away.
Breeding occurs when water levels are high, usually between May and October.
Females carry broods of up to 40 eggs on their abdominal legs and ventilate
them by leg movements. Larval stages pass in eggs, and miniature semblance of
adult hatches. Juveniles often wash downstream, sometimes out into surface waters.
Young adults migrate back up into the deeper reaches of cave waterways, where
they mature and breed. Lifespan is up to 2 years, predators include cave-dwelling
amphibians and aquatic insects.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Driftwood-boring
shrimp (Xylocaris terebratus)
Order: Decapods (Decapoda)
Family: Long-armed shrimps (Macrobrachiidae)
Habitat: woods of Amazon region, shallow rivers.
Tropical rainforest and river are practically indissoluble combination. Many
forest inhabitants are connected in their life with rivers, and the wood roofs
and foods some river inhabitants. In Neocene the set of interrelations between
inhabitants of two these worlds has replenished with one more example.
Rainforests of an Amazon region from time to time are overflowing by water.
Trees have not sustained flooding, fall and lay across rivers. In their roots
and branches fishes and other inhabitants of the river find shelter, and some
fishes even rather willingly gnaw mossy moldering wood. In Neocene one more
animal has joined to set of dead wood destructors.
The tree has fallen in water about two months back, at first sight looks almost
intact. Rotten pieces of bark here and there have fallen off, and dead leaves
are carried away by river. But at close look it becomes visible, that tree trunk
obviously has traces of someone’s work: in some places wood is pierced by apertures
in which small creatures sit. Being scared, they disappear in burrows, but being
calmed, they dare to creep out. And then it becomes evident, that they are crustaceans:
at them there are long antennae and eyes sit on stems.
It is the special kind of shrimps from tropical rivers of America – the driftwood-boring
shrimp. It serves here as the original analogue of bivalve mollusk “ship worm”
(Teredo): this crustacean drills holes in wood, destroying trees have got in
water. The driftwood-boring shrimp lives in minks by whom itself does(makes)
in wood. Sometimes this shrimp lodges even in roots of trees, for any reasons
flooded with the river and remaining in water the year round. These species
prefer a society to itself similar, therefore settle usually the whole colonies
numbering up to hundred of individuals.
Length of the driftwood-boring shrimp is about 5 cm. Body of animal at the back
edge is translucent, armour is yellow-colored with black longitudinal strip.
Pincers are white color, very short and strong, with jags along the edge. They
have turned to powerful drilling apparatus with which help this shrimp destroys
wood of the majority of tropical trees.
Eyes are large; they are placed on long stems. When shrimp sits in shelter,
from hole only eyes and antennae stick up. Occasionally the crustacean leaves
holes, but does not leave far from them. At danger this animal quickly swims
and hides into the nearest hole.
The colony occupies tree and pierces it with holes where crustaceans live and
breed, while the tree will decay finally. Holes are deep, pulling mainly in
top layer of wood. They join, forming complex system in process of colony increasing.
The driftwood-boring shrimp almost does not eat wood: it eats, scratching out
from walls of holes fungoid film and colonies of bacteria, and also eats small
animals swimming away in holes in searches of shelters. Naturally, as an addition
to food it swallows some wood dust, but it is not digested. Due to strong pincers
the crustacean easily splits shells of snails.
Development is direct, from eggs tiny shrimps similar to adult individual hatch.
In clutch it is about 30 – 50 rather large eggs. Young shrimps live in holes
of adults, but in process of growth they are gradually superseded to edge of
colony, where they start to drill their own holes.
Needle
shrimp (Acuticaris incola)
Order: Decapods (Decapoda)
Family: Tubocarids (Tubocaridae)
Habitat: reeves of Indian and Pacific oceans, colonies of tube worms.
After extinction of reef-building corals, a role of main architects of ocean
shallow waters had been undertaken by various animals, first
of all by mollusks.
But the considerable part of reeves is constructed by sedentary annelid worms both
with red algae sedimenting limestone. Settlements of worms form on sea bottom
true “towns” of numerous directed upwards straight or bent tubes. Usually from
tubes tentacles of these animals stick up, but if any large animal appears
nearby, worms immediately hide in tubes. But they will not manage to disappear
from some animals of reef thus even because that ones share “apartments”, their
own tubes. One of inhabitants of worm tubes is thin transparent shrimp with
extended legs and pincers. When it is shown at few seconds from tube of one
worm to get into the next tube, it is practically not visible in sea water
- only two black beads of eyes give out presence of this crustacean.
The needle shrimp almost constantly sits in tubes of worms. Certainly, these
animals are not always glad to meet visitors: the tube is designed only for
one worm. But they suffer presence of this shrimp not vainly: this crustacean
plays a role of cleaner, cleaning from tube every possible dust, and also gathering
from bodies of worms every possible parasites which this sedentary animal is
not able to shake out from itself.
The thin and flexible shrimp scurries in dwelling of worm, touching by antennae
to its body. By its physical condition shrimp estimates environment and chooses
the moment to get over to the tube of the next worm. If worms have quietly
opened their tentacles, shrimp can travel in colony under their covering. But
if worms are disturbed and start to fold tentacles, shrimp immediately dives
into the nearest tube. Its body is flexible and worm-like: armour covering
cephalothorax is rather short, and shrimp resembles more any primitive crustaceans.
The body of this shrimp is strongly extended (about 5 cm long at thickness
only of 3 - 4 mm), and legs are very thin. Pincers account the body length;
they are thin and sensitive: with their help shrimp gathers dust and parasites
from cautious “host”. On the forward end of a body of needle shrimp the strong
spike grows – it is unique weapon of this fragile creature. When too curious
fish shows interest to tube where this shrimp is hidden, it risks getting sensitive
sting of such spike.
Swimming abdominal legs of needle shrimp are hairless, and serve only to bear
eggs - this shrimp is not able to swim, and can only creep clumsily in a bottom
outside of worm tubes.
Because needle shrimp is the homebody, at it a breeding problem could arise:
eggs should be fertilized to make the posterity genetically various. And at
the same time populations of worms in which tubes these shrimps live, are far
from each other, and frequently are too small to settle in them many individuals
of needle shrimps. But this crustacean has to found a simple way out: the needle
shrimp is hermaphrodite, also in case of need it can be simply self-fertilized.
But it is the extreme way: more often in colony of worms there are even two
shrimps mutually fertilizing each other.
In clutch of needle shrimp it is usually up to 300 very small eggs. From them
smallest plankton larvae decorated with long needle-shaped outgrowths on sides
and back hatch. Their development passes at the reef, usually at deepest sites
of it. This shrimp breeds when for some weeks quiet weather without storm is
established. At larvae there are two main problems in life: to survive in plankton
where everybody can to have eaten them and find a colony of worms. After the
next molt larva loses “ornaments” on body and gets rather recognizable shape
of adult shrimp, only without needle on head. It finds tentacles of worm and
settles in its tube. It is a crucial stage in life of young shrimp: if it will
not to find a colony of worms, it’ll appear completely defenceless against
numerous reef predators. If the young shrimp is lucky, it starts to grow and
at the age of about 10 weeks already becomes adult. To this moment at it the
nasal needle grows.
The first contact of young shrimp and worms never passes smoothly. Worms, one
might, “mistrustfully” concern to occurrence of shrimp, and perceive it hostilely:
hide, close tube by operculum, or jet out to the shrimp the poisonous substance
at all. But persistence and care of shrimp make the business: it starts to
clear accurately the future hosts of parasites and dust, and eventually between
it and worms communication is established. Gradually the shrimp starts to get
into tube constructed by the worm, and soon grows roots in it and takes over
“trusteeship” some part of colony of sedentary worms.
Spike shrimp (Paracuticaris echinophila)
Order: Decapods (Decapoda)
Family: Tubocarids (Tubocaridae)
Habitat: reeves of Pacific Ocean, symbiote of sea urchins.
Reeves are favorable place for life, and are comparable to tropical forest
on specific variety. In Neocene the main reef-builders, madreporarian corals,
had become extinct, and the body of reef is formed by other invertebrates:
mollusks and worms. Between inhabitants of reef there is strict competition
inducing strict specialization. And the abundance of predators forces to develop
more and more effective ways of protection. Some inhabitants of reeves use
reliable means tested during millions years of evolution. Sea urchins of various
colors are armed with spikes and firm armours. It makes them impregnable for
the majority of reef inhabitants. Protection of sea urchins serves not only
to them – among spikes of these animals tiny graceful shrimps hide. Pointed
spikes with poisonous slime in grooves serve as an absolute obstacle for predators
eating crustaceans. The specific pointed body shape had determined the name
of this crustacean – spike shrimp.
This tiny (about 5 cm long) crustacean occupies the ecological niche of fishes
– shrimpfishes (Aeoliscus strigatus) and urchin clingfishes (Diademichthys
lineatus) used such shelter in human epoch. Spike shrimp constantly keeps between
spikes of sea urchins – here it finds simultaneously food and shelter. This
crustacean gathers parasites and dust from body of echinoderm, and receives
safe dwelling in return for it.
Body of spike shrimp is long and narrow. Cephalothorax is short; pleon is cylindrical
with wide fin formed by telson and back pair of pleopods. On head of this crustacean
there is pointed straight outgrowth directed forward and little bit similar
to spikes of sea urchin. If necessary, spike shrimp is able to change color
of body cover, adapting to shade of spikes of sea urchin on which it lives.
This crustacean sits among spikes directing head outside. Shrimp can move among
spikes of sea urchin in any direction, and at danger hides deeper into spikes,
closer to body of host animal.
With the help of thin claws shrimp gathers from spikes of sea urchin larvae
of sedentary animals settling on them, and kills parasites attached to it.
Thus, relations of sea urchin and spike shrimp represent mutually advantageous
symbiosis.
On each sea urchin some such shrimps live, forming breeding group. The gender
at these shrimps is not genetically determined, and depends on age of animal
and its position in hierarchy. Usually the colony is presented by females among
which one male lives – it is the largest and strongest shrimp. If for any reasons
male leaves colony or perishes, strongest female takes its place in hierarchy.
Shrimps growing alone initially develop as males.
Each female regularly lays eggs – up to 100 eggs once in four weeks (the laying
is dated for full moon), and bears them within two weeks. At this species there
is development without larval stage, and from eggs tiny similarities of adult
individuals leave. They at once leave parental colony and search new host for
themselves. Young shrimps can live on sea urchins with rather short spikes,
and larger individuals whenever possible move to long-spiked species. Spike
shrimp becomes adult at the age of about 7 months. Life expectancy of these
animals seldom exceeds two years.
Hive
snapper shrimp (Alpheus alvearosus)
Order: Decapods (Decapoda)
Family: Snapper shrimps (Alpheidae)
Habitat: Pacific Ocean, reeves in tropical zone.

Queen female
Picture by Toron, colorization by Biolog
One way of survival in conditions of competition is the social habit of life. Insects (and among them termites and hymenopters did it independently from each other) and to a lesser degree spiders (in Neocene some new species leading social way of life had evolved) selected such way of evolution on land. For social species of arthropods formation of castes is typical; relations between them are regulating with the help of secretion of physiologically active substances having influence on development of colony members. The similar system of relations between members of the colony had formed independently of them at some species of crustaceans living at the reeves of Pacific Ocean. Here hive snapper shrimp lives; it is the social species of snapping shrimp living in reef-building sponges growing in shallow parts of tropical seas. It is the remote descendant of colonial snapping shrimps of Holocene epoch.

Castes of the hive snapper shrimp:
Top left - guardian male;
Top right - male;
Bottom - juvenile specimen.
Picture by Toron, colorization by Biolog
Colonies of this species reach the number up to 5000 individuals.
In colony there is rather stable ratio of individuals of different genders and
ages between females, males not sexually mature young ones – approximately 1:5:20.
There are appreciable morphological differences between individuals of different
genders and ages. As against free-living relatives, this species is omnivorous,
but has a bias to vegetarianism.
The colony occupies cavities inside the same large sponge of a special kind.
There is only one fertile female in colony, not leaving a body of sponge and
devoted only to reproduction. It is a “queen” 10 – 12 cm long. It has rather
small “working” claw, and it can not utter ultrasonic clicks characteristic
for snapper shrimps. Colouring of its shell is pale, yellowish-grey. Its eyes
are rather small, and sight is bad. The “queen” female is adapted for bearing
a plenty of posterity – its pleon is expanded, and pleopods are covered with
bristles. Fertilization is performed by stranger males from other colonies,
which live freely and move between different colonies. The number of eggs in
one clutch reaches 150-300 ones. Development of this species is direct, without
larval stages. In addition to metabolic products the “queen” female secrets
pheromones determining the further development of young ones.
At low number of the colony and small inhabited cavity inside the host sponge
pheromones of queen females cause sterilization of growing up females. Sterile
females sharply enough differ in morphology from fertile ones. Length of sterile
hive female is 6 – 7 cm. It has strongly expanded pleon with large pleopods,
but the female of this caste is not able to swim, bending the pleon. Such females
constantly live inside the sponge body and perform a set of functions on maintenance
of life of the colony. They provide an additional current of water through the
body of the host sponge, protect entrances to the colony cavities, and also
are engaged in driving and clearing of tunnels in the body of the host sponge.
Right cheliped (claw) at them is strongly increased, and its terminal segments
have disc-like shape. Such claw is supplied with additional denses on edges
and serves for entrance closing at protection of the colony. Function of ultrasound
generation at such females is weakened. Colouring of body of these females is
pale, except for the right claw, which has dark brown colouring.
 |
 |
| Initial images by Toron |
|
Colony’s own males reach the length of about 5-6 cm. Their
sexual function is also oppressed with action of pheromones of queen females.
Males perform the functions of active protection of colony and food gathering.
They constantly leave cavities of the host sponge and creep in vicinities of
the colony. Colouring of males appreciably differs from colouring of females
– they are pinkish with the reddish-brown marble pattern well combined to colouring
of surface of reef. They continuously check and protect the algae gardens, which
grow in littoral pools at the the internal side of reeves. The food competitors
encroached on vegetation are ruthlessly destroyed, or frightened off with the
help of ultrasonic impacts. Killed thus small animals (fishes, shrimps, molluscs)
are used as a food of the colony. Males have good sight; their eyes larger,
rather than at females, and are supplied with a great number of facets. Protecting
of a site of vegetation they operate in groups of 10-20 individuals, supporting
tactile contact with each other with the help of antennae. Protective reaction
of own colony’s male is powerful stimulus for joint attack – having heard click
of the relative, male take a threatening pose and start clicking. If in their
field of view any moving object appears, intensity of clicks amplifies and they
turn aside the infringer of territory.
The sexually immature young ones after first two moults are included in active
life of the colony. The young ones of both genders differ from adult individuals
both in morphology, and in ecology. Besides the feeding on vegetative and animal
food like adult individuals, young individuals of hive snapper shrimp have an
ability of water filtering. They frequently stay on the surface of sponge near
the entrances, filtering water, but at first signs of danger they hide in depth
of sponge. In colony young individuals perform functions of cleaners and tunnellers.
In addition to clearing of tunnels in the body of the host sponge of dust, they
also are engaged in grooming of adult individuals of the colony, especial of
females. Due to thin claws they can clear of parasites branchial cavity of adult
individuals. Some young individuals always keep close to “queen female”.
When colony grows up to limiting number, when it occupies large enough space
inside the sponge, the amount of pheromones emitting by queen femal falls below
critical concentration, and it causes exceeding by the part of sexually mature
males of the bounds of the colony and activization of their sexual function.
The present process is accompanied by change of colouring and behaviour of males
– they turn much darker, undergo an additional moult and become larger. Sexually
active males begin to react keenly to smells of other colonies, preferring maximal
genetically remote ones. They abandon the native colony and wander on surface
of reeves, hiding from enemies in casual shelters. Having reached an unrelated
colony, such male emits the substances calming guardian females. Due to it male
easily penetrates into the colony and copulates with foreign queen female. Such
males do not live in a colony constantly, and the majority of them perishes
during the migrations between colonies.
Lack of pheromones also causes changes in growth of sexually immature females.
Their specialization does not take place and at the edges of the colony they
freely reach sexual maturity and then abandon parental colony. Young queen females
search for free host sponge; then the copulation with migrating stranger males
takes place.
This species of crustaceans was discovered by Toron, the forum member.
Gill
crayfish (Cryptocambarus branchiophilus)
Order: Decapods (Decapoda)
Family: American crayfishes (Cambaridae)
Habitat: the south of North America, an area coincides with the area of manatee
fish.
Every species of live organisms is an inhabitancy for numerous parasites and
symbiotes. Some of them cause essential harm to their host, others use it only
as a shelter, and presence of the third ones brings mutual benefit to both species.
Frequently the species belonging to systematic groups very far from each other
may be interconnected.
In southern areas of North America, in the wide rivers with well warmed up water
the large manatee fish reaching the length of 4 meters lives. This peace herbivorous
creature gives a shelter to very original representative of decapods.
In almost every case the couple of very small gill crayfishes lives in branchial
cavity of manatee fishes. Possible, the development of such way of life by these
ones is rather recent evolutional step, because in their anatomy features of
crayfishes living in the same reservoirs, but leading a way of life traditional
for them, are quite recognizeable.
Appearance of gill crayfish is traditional for decapods. This is a creature
with four pairs of walking legs and well advanced thin claws. Last pair of walking
legs of gill crayfish is increased a lot, and on its internal side numerous
thorns and bristles develop. These legs serve for the attachment to substratum
– at the way of life which this crayfish leads always there is a probability
to be washed away by current of water through gills to an environment where
this animal is doomed for immediate death. The body of gill crayfish is transparent,
covered with thin armour. The strongest parts of body are claws and back pair
of legs. Last pair of pleopods forming a fin, is reduced in great degree and
is transformed to attaching hooks with which help animal clasps gill arch of
fish. Eyes of gill crayfish are considerably reduced. They are placed on short
stalks and are almost motionless – the crayfish can only retract them under
carapace a little. Antennae, on the contrary, are very well advanced – their
length exceeds total length of body of this animal.
The couple of adult animals of this species, large female and smaller male,
always lives in branchial cavity of fish. The gender at gill crayfish has no
genetical fixation: the first individual settled on fish turns to female, and
the second one, having found her and having attached to her, turns to male.
Sexually mature male constantly keeps on the female with the help of back pair
of walking legs. He fertilizes eggs and looks at young crayfishes until they
leave mother. Usually male keeps on the belly side of body of the female, but
he is mobile and can creep on all parts of her body. The length of the adult
female reaches 30 – 35 mm; male is not longer than 20 mm.
Gill crayfish eats tiny invertebrates casually swallowed by fish with its food.
It renders the host fish of cleaning service, gathering from its gills parasitic
crustaceans and worms which constantly settle on fish.
Gill crayfishes breed the year round, giving for one year up to eight litters
numbering 100 – 150 young crayfishes in each one. This species has direct development
– from eggs tiny animals able to creep and to swim hatch. They are not too similar
to adult individuals: they have normally advanced back pair of legs and rather
large eyes. Young animals stay on pleopods of the female till about one week,
and male cares of them. Passing to independent life, they leave gills of manatee
fish with current of water and settle in plant thickets waiting for approaching
of new host fish. If they will stay on gills of fish, on which their parents
live, they, most likely, will be eaten by their own parents, which perceive
them as food.
The young crayfish can lead free life for a long time. As against common crayfishes,
it keeps mainly in the top circle of reservoirs – on stalks and leaves of floating
and long-stem plants. When the manatee fish comes nearer, young crayfish clings
to its skin and then creeps on gills. Here it undergoes metamorphosis, getting
after several moults prominent features of adult representatives of this species.
Kōura
Mārō (Hoploastacus australis)
Order: Decapods (Decapoda)
Family: Southern Crayfish (Parastacidae)
Habitat: cold, well-oxygenated rivers in the South Island of New Zealand, and
the southern part of the North Island.
Since the later part of the Tertiary, New Zealand has showed a tendency to cool
climates, which was greatly catalysed by its isolation, and its vicinity to
the circumpolar current. Even in the Neocene as it begins to travel north, much
of it is temperate and cool. Cold temperatures in aquatic habitats often promote
a degree of gigantism in the invertebrates that live there, ones closer to the
poles will be larger than their tropical counterparts. Such a situation was
seen with the Tasmanian Giant Crayfish (Astacopsis gouldi) which inhabited southernmost
Australia during the Holocene.
In the Neocene, a similar circumstance has played out in the cold rivers of
the southern part of New Zealand. The Kōura (genus Paranephrops), native freshwater
crayfish, has developed into a large, well armoured and pugnacious crustacean
found only in sheltered, cold, well-oxygenated rivers. This is the Kōura Mārō,
Maori for “Armored Crayfish”.
Development within the depredations of New Zealand's Neocene has shaped this
crustacean into a formidable creature. Size approaches that seen in marine “spiny
lobsters” found in neighbouring oceans, males may reach as much as 50 centimetres
and females 45 centimetres, not counting the fore-claws or antenna, weight may
be as much as 6 kilograms. Exoskeleton is heavy and covered in ridges and large
spines, coloration is most often a blackish brown with the underparts reddish
brown, but bluish or reddish variants may occasionally be seen. Fore-claws are
large and bear distinct “teeth” on the cutting surface of the claw, males have
larger claws than females, used in territorial combat.
The diet of this crustacean is mostly scavenging, it will consume dead wood,
carrion, dead-leaves, water-plants and occasionally aquatic invertebrates such
as worms and snails. Most active at night, they are more common along riverbeds
that are deep, with a lot of snags, submerged wood and deep depressions. Annually
they are the most active in the warmer months of spring and summer, and in the
depths of winter may restrict themselves to a smaller territory. Males are territorial
and maintain a harem of several females. Adults may travel overland in order
to establish new territories, but invariably do so in the wetter months, and
at night.
Maturation is not quite as slow as some of its Holocene relatives, females reach
sexual maturity at 10 years or so, with males reaching maturity after about
8. During the spawning season, antagonism between males is heightened, and they
will fight over territory and the right to breed. Females usually only spawn
once per year, after their summer moult in the height of autumn (between March
and May). Number of eggs may vary between 300 and 1500 based on the size of
the female, and the gestating eggs are carried through the winter, hatching
after about 6 months. The hatchlings are about half a centimetre long and cling
to their mother's tail for another 2 and a half months until they can feed on
their own.
An adult crayfish has relatively few predators, but the young may fall prey
to predatory fish, water-birds, parrots and aquatic mammals such as the Waitoreke
and Unotter. Lifespan may
be as long as 60 years.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Hawaiian boring crab (Terebrocancer geograpsus)
Order: Decapods (Decapoda)
Family: Grapsid crabs (Grapsidae)
Habitat: Hawaii, ground in tropical woods.
Islands never having connections with other land differ in big originality
of animal and plant population. The fauna of such places is impoverished by
typical continental species (especially by animals unable to fly or not suffering
salt water), but is frequently replenished due to sea species passing to overland
life. One of ways to leave the sea is to become digging creature and gradually
to pass to existing in damp atmosphere of underground burrows.
Rather small competition of soil animals at Hawaiian islands had permitted
to one of sea crabs widely settled in Pacific Ocean to pass to this inhabitancy.
This way the boring crab, the next overland descendant of sea inhabitants,
had appeared at Hawaii. It is small species of digging crustaceans – width
of its carapace is about 15 cm, and whole crab is up to 20 cm width. It could
be rather significant size for crabs of habitual constitution, but not for
this species. Hawaiian boring crab combines usual for crabs habit of movement
sideways with hole digging, therefore proportions of its body had strongly
changed. At such significant width of the body the height and length of this
crab do not exceed 5 – 6 cm: this animal is as if “stretched” in sides.
Pincers of this crab have unequal sizes (this feature is characteristic for
crabs in general), and fulfil different functions: one pincer serves as the
drill, the second is a shovel. Usually crab bores ground with the help of right
pincer which strongly differs from left one externally. On the right pincer
the penultimate segment is very big with the expanded gear edges and numerous
knobs on the external surface. “Thumb” (last segment) on this pincer is small,
but strong: with its help this crustacean easily cuts roots of plants, preventing
to dig a hole. Left pincers is lesser than right one, expanded, with smooth
edges. It rakes away ground dug by crab. Also with its help crab brings food
to mouth. If the crab had injured boring pincer, or it had been bitten off
by any predator, it has reorganization of sense organs and anatomy: the remained
intact pincer for some molts turns to boring one, and the crab turns to “lefthander”
from “right-handed one”. The restored right pincer becomes “shovel”. However,
about 10 % of population of boring crabs are hereditary “lefthanders”. The
walking legs of this crustacean located from the same side as “boring” pincer,
also fulfil the function of shovels, raking away the loosened ground back,
under “shovel” pincer. For this purpose the fringe of rigid bristles on their
edge also serves. When near of “shovel” pincer the heap of ground gathers,
crab walks to other side, raking away friable ground on the ground surface
or to burrows of other animals which contact with its holes, partly shifting
the work to them.
Sight does not play the significant role in life of Hawaiian boring crab, and
its eyes are appreciably reduced: they are very small, located on short thick
stems and at ground digging are hided in deepenings of the front edge of the
carapace. From outside they are protected from the ground by rich “eyebrows”
of bristles.
Boring crabs live in holes at the depth about half meter, at night rising to
the ground surface. They eat various invertebrates, but can eat and fruits
beginning to rot: obviously, a strong smell of fruits involves crabs. Occasionally
at night they get out on the surface of the ground and catch small ground animals.
If the dead animal (for example, the carcass of Hawaiian
forest goose – the
large local bird) lays on the ground, crabs can sap under ground the carcass
and eat it from below, and also gather larvae of flies and beetles eating carrion
and dug in the ground for pupating.
Being the ground inhabitant, this crab has not lost the connection with water.
It had left the sea, but goes to fresh water for breeding. Male differs from
female in narrow wedge-shaped pleon and in larger size. It couples with female
in specially dug wide hole, and does not accept any more participation in care
of posterity. But the female, ready to lay eggs, settles closer to water: it
digs in riverbank a hole which is filled with water filtered through ground.
In the formed reservoir female bears eggs within approximately two weeks. In
clutch it may be about 200 large eggs. When the time to hatch larvae appears,
female abandons the shelter. At late night it gets out of hole and hastens
to the reservoir where the simultaneous hatching of young growth occurs (fresh
and rich in oxygen water serves as stimulus of this process). If not all posterity
had hatched within night, female throws off the rests of eggs from the abdomen,
and always comes back in a hole to the morning.
The posterity of this species, looking like tiny transparent crabs, lives in
water some weeks similarly to other species of crabs. Having reached the diameter
about 3 cm, they leave water and pass to life in wood litter. Having got stronger
enough, they start to bore holes in ground. Half-year aged crabs having width
of carapace about 6 cm are able to breed, and they reach the size typical for
this species at 3-rd – 5-th year of life.
Azorean
nocturnal crab, “night robber” (Azograpsus fur-nocturnus)
Order: Decapods (Decapoda)
Family: Grapsid crabs (Grapsidae)
Habitat: New Azora island, forests.

Picture by Pavel Volkov
Terrestrial island ecosystems differ from continental ones
in one feature – recent natives of sea play rather large role in them, and many
groups of terrestrial animals, characteristic for continents, can be absent
completely. At New Azora island, which is located in tropical latitudes of Atlantic
Ocean, in an ecosystem there is a plenty of descendants of mammals, terrestrial
continental animals. This is a consequence of human activity – people had introduced
some species of terrestrial mammals to Azorean Islands. But in ecosystem had
formed at the island after extinction of mankind, there are descendants of sea
animals – various crabs of separate genus Azograpsus, or Azorean crab (Azograpsus).
Various species of these crabs occupy ecological niches of scavengers and small
predators. Among these animals the largest species is “night robber”, or Azorean
nocturnal crab – scavenger and active predator representing danger to rat-sized
and smaller animals.
“Night robber” is large crustacean – the width of its carapace reaches 12 –
15 centimeters, but because of legs and large claws it seems even larger. Its
armour is flattened from above and has rounded outlines. Claws are rather large,
have short fingers and serrated cutting edges. With the help of such claws crab
easily breaks snails shells and crushes bones of small vertebrates.
Colouring of armour of crab is black with bluish shade; bottom part of body
is grey. Cutting edges of claws are brightly visible on background of body –
they are snow-white. On claws there are some more white spots.
Azorean nocturnal crab lives in underbrush and can climb on trees, especially
at young age. Small crabs of this species spend much more time on trees, rather
than adults. Due to this feature of behaviour the probability of survival of
young ones of this species raises – this way young crabs avoid meeting with
adult individuals which frequently are cannibals. Adult Azorean nocturnal crabs
lead nocturnal habit of life and hide in holes in day time. They dig hole by
themselves, but can occupy and re-build ready hole of any small animal (frequently
the lawful inhabitant of hole is simply eaten thus). At night this crab moves
to searches of food. It easily copes with small rodents, eats eggs and nestlings
of birds nesting on the ground, and easily finds carrion and rests of prey of
local predators with the help of sharp sense of smell. Azorean nocturnal crabs
living near the ocean coast can hunt crabs of other species.
All crabs of genus Azograpsus had breaked off the dependence on the water environment
during the breeding to greater or lesser extent. Only some species of these
animals make migrations to ocean, and the majority of species is content with
temporary reservoirs for posterity hatching. At all species of these crustaceans
in clutch there is only a few sizable eggs from which well advanced young crabs,
quickly passing to independent life, hatch. Females of Azorean nocturnal crab
dig holes at the coast of reservoirs, and on bottom of such holes from ground
a lot of water filters. In formed puddle female spends all day and some part
of night, and only in darkest night hours it hunts. Last days of incubation
the female does not leave shelter at all.
In clutch of this species of crustaceans up to 70 – 80 rather large eggs is
totaled. Young crabs are translucent, colored light grey. The female does not
pay attention to the posterity, and does not feed young growth specially. First
some days of life young crabs hide in hole of the female and eat tiny ground
animals. After the first molt they leave parental hole and begin independent
life. At the age of four years young crab reaches the size, characteristic for
adult individuals, and its growth slows down. The sexual maturity at these crabs
comes at the age of about one and a half years.
At New Azora some other species of crabs of this genus live:
Azorean
signaler crab (Azograpsus signalis) differs in very bright colouring.
It is medium-sized (carapace width is about 7 – 9 cm) diurnal species of crabs.
It has good sight, and large spherical eyes are located on long mobile eyestalks.
The body of this crab has dark red colouring, and claws are colored scarlet
color with white spots. Claws of this crab are very strong and serve for digging
of larvae of insects from ground. Besides with the help of such claws crab protects
itself, and bright colouring serves to defending animal for warning signals
injection. The disturbed crab rises claws and starts to click by them. On finger
of claw it has small knob which enters into hole on claw. When crab closes claw,
loud click is heard. The similar mechanism was developed at this species independently
from snapping shrimps of family Alpheidae known in human epoch.
This species of crabs eats small invertebrates and oily fruits of palm trees
and trees of laurel family. Females bear eggs on abdomen during almost all term
of incubation. When time of posterity hatching comes, females gather in littoral
marshes and drop young crabs hatching from eggs in water. Fertility of this
species makes up to 200 eggs in one clutch.
Arboreal
Azorean crab (Azograpsus arboreus) is small species of the genus. The
width of carapace of adult animal does not exceed 4 – 5 cm. It differs in dim
greenish colouring of body with dark brown legs and claws. The bottom surface
of claws is colored snow-white – crabs of this species inject signals to each
other, raising claws and showing their bottom side. This species constantly
lives in crones of trees and is able to swarm up branches in searches of fruits
and small tree invertebrates. Females of this species bear clutch, gathering
in tree hollows where rain water accumulates. Clutch of this species totals
no more 50 large eggs from which young crabs hatch. Adult animals do not attack
young growth, and the survival rate of posterity at this species is rather great.
Young crabs right after hatching from eggs start to lead independent life. They
creep away from hollow where they were born, and hide in moss or in other places
where the moisture is kept. This species of crabs does not go down to the ground
during whole life cycle.
Lemon,
or frugivorous Azorean crab (Azograpsus citrinus) lives in
tropical forest litter. This medium-sized (carapace width is about 8 cm) crustacean
differs in very bright colouring – lemon-yellow with black points on tips of
claws; the bottom part of body is white. Frugivorous Azorean crab eats mainly
vegetative food; the most part of its diet is made of fruits of plants of laurel
and heathers families characteristic for forests of New Azora island. Occasionally
it eats seeds of coniferous trees.
In day time crabs of this species dig in wood litter or hide in shelters of
various kinds, and in the evening goes to search of fallen fruits. In cloudy
and damp weather they are active in day time. This species of crabs makes mass
migrations during breeding season, and females with clutch spend some weeks
in shallow waters of island while incubation of eggs proceeds.
Tiniest
Azorean crab (Azograpsus minutissimus) is the most numerous crab of
the island. This tiny (carapace width is no larger than 4 cm) species of crustaceans
is the important part in ecosystem of island, because the basis of its food
is made of rotten leaves of trees and grassy plants. Such accessible forage
allows these crabs to reach great number – on the average, at each 5 square
meters of territory of New Azora forests one crab of this species lives. Colouring
of tiniest Azorean crab is dark, but is remarkable by its refinement: body is
reddish-brown, legs and claws are black, and cutting edges of claws are snow-white.
Due to activity of these crabs fallen leaves quickly turn to humus, from which
plant easily take mineral and organic substances. These crabs dig in wood ground
deep (up to 60 – 70 cm) holes in which spend the day. The female incubates eggs
on land, and right before hatching of posterity searches for shallow reservoirs.
Young crabs spend some days in fresh water, and then creep out on land.
Aotearoan
Forest Crab (Novaeterracancer furtivus)
Order: Decapods (Decapoda)
Family: Land Crabs (Gecarcinidae)
Habitat: Warm tropical forests in the northern-most parts of the North Island
of New Zealand, forest floor.
The islands of the south Pacific usually play host to land crabs of different
species (family Gecarcinidae). After the extinction of man as one of their main
predators, New Zealand was fit for these forms to eventually colonise the northern-most
tropical forests there.
The Aotearoan Forest Crab is a common sight in the humid tropical forests that
grow in the most Northern regions of the North Island. These crabs are of moderate
size, carapace measures up to 12 centimetres across, they greatly resemble other
land-crab species, claws are relatively large, and males reach a larger size
than females. Their legs are fairly long and strong with pointed trenchant tips,
crab will stand upright and can crawl quickly over snags and rocks, also climbing
into the lower branches of trees and bushes. Coloration is usually a orange-brown
colour, with brighter orange on the carapace, extremities of limbs and claws
are darker, occasionally bright red or purple colour strains are found.
Breathing is done with internal gills, and these animals endeavour to stay in
damp areas to conserve moisture. Individuals will usually dig burrows in the
soil in which to shelter, usually each crab resides for a long time in one hole.
Crab is diurnal, becoming slow-moving and inactive by night. During the drier
part of the year, they may cover the entrance to their burrow and hide until
the ground becomes wet again. These crabs are opportunistic scavengers, feeding
on fallen fruit, dead leaves, seeds, animal dung, carrion, and smaller slow-moving
animals such as snails or worms, the eggs of ground nesting birds are consumed
too.
Between late September and Mid December, these crabs will migrate in large swarms
in order to find estuaries and coastal areas where they breed and deposit their
eggs. Males will excavate burrows in mud or sand, which they defend from intruders,
mating is done in the burrows. After this the male leaves and female lays and
broods her eggs, incubation lasts up to 2 weeks. After incubation, the female
releases the eggs into the water, where the eggs are stimulated to immediately
hatch and drift out to sea. The babies take about 4 weeks to mature into juvenile
crabs that resemble miniatures of their parents, after which they come ashore
and migrate inland, usually hiding in damp vegetation to conserve moisture.
Moulting in adults occurs once per year, lifespan may be up to 11 years, predators
of adults include hawks, parrots, mustelids,
canids and large
hedgehogs, the young may be consumed in the water by many kinds of fish,
and upon reaching land are commonly eaten by lizards,
frogs and large insects. Some kinds of swarming ant
will overwhelm and kill even adult crabs, and consume their meat.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
"Mangrove
robber" (Archocancer mangrophilus)
Order: Decapods (Decapoda)
Family: Mangrove crabs (Archocancridae)
Habitat: mangrove thickets of South-Eastern Asia – Jakarta Coast, Sunda Land
and near islands, fresh and brackish water.

Picture by Alexey Tatarinov
Mangrove swamps are very changeable world. Twice per day
inflow and outflow replace each other here, and the salinity of water strongly
changes – from
almost fresh up to almost oceanic. Trees form continuous thickets, alternating
with sites of viscous oozy ground. Everywhere from silt respiratory roots
of trees stick up, forming almost impassable “fences”. Here it is difficult
to
survive to both oceanic and freshwater inhabitants. Neither aquatic animal,
nor the ground one can not feel like as at home in this place. But animals
had been adapted to this changeable world can feel like true kings.
Largest of local inhabitants has all properties permitting it to survive
here. It has tenacious legs, and it can swarm up roots of trees. Stepping
through
branches and air roots, it actively searches for food. But if it will fall
down in water, it simply will continue to move in a bottom. It easily tolerates
change of salinity of water, and its food is made almost by everything, that
it is possible to find in swamp. It surveys a surface of mangrove swamps
walking on long legs as if on stilts. Sharp twigs do not harm to it – its
body is covered
with shell.
This creature looking monstrously from the point of human view is one of
species of crabs. In Neocene when number and variety of many of dominating
before groups
of animals has decreased, some of “supernumeraries” had an opportunity to
evolve actively. And from one crab of Cardisoma genus, able to live both
in fresh,
and in sea water, the huge crab “mangrove robber”, the largest arthropod
able to live on land, had evolved. To tell the truth, it spends on land not
all
life – for breeding and gill humidifying these huge crabs are compelled to
come back to the water, but not for a long time.
The width of the armour decorated from above with “hornets” and lumps, reaches
half meter, and leg-span exceeds one and half meter. Carapace has the “angular”
shape; in front it is almost straight, hind edge is rounded. “Mangrove robber”
lives mostly in air environment therefore its branchial cavity functions
as lung. However sometimes it comes into water to humidify gills. During
the rain
this huge crustacean willingly puts body to floods of the water flowing down
from leaves.
Colouring of this crustacean is rather bright. The animal is perfectly armed;
therefore it is favorable to it to warn the predator about consequences of
attack by the colouring. The trunk of crab is colored from above yellowish
- brown color with small dark speckles, legs outside are colored the same
color. But an internal surface of legs and the bottom side of flat body is
ivory-colored.
Pincers of crab are wide and flat; at males they are larger than at females.
Surface of pincers is smooth and shining. They are colored outside bright
red with white strips along cutting edge on “thumb”. The bottom side of pincers
is light pink. In courtship season males show to females bottom side of pincers
to lower their aggression.
Eyes of crab are increased and placed on mobile stems: it permits an animal
to have the full circular field of view. Small facets provide rather sharp
sight, and the crab can easily define a possible predator. Though in mangrove
swamps almost nobody is danger to it, in forest predatory mammals or birds
can easily trap “mangrove robber”. They attack young growth especially frequently.
But the adult crab in intervals between molts is practically impregnable,
and it will reflect any attack by strong pincers.
“Mangrove robber” is omnivorous animal. It willingly eats any food of animal
origin: it hunts mollusks creeping in oozy shallow water, gathers fishes,
shrimps, worms and snails staying in pools during outflow, catching them
by big long
pincers having on tips sharp jags and hooks. It does not squeamish also to
eat carrion: after storm such crabs frequently leave on a shore, searching
for dead animals have been cast ashore. In forest “mangrove robbers” appear
for the sake of fruits – they eat sweet and strongly smelling fruits of tropical
trees with great pleasure, being one of main distributors of seeds on tropical
islands.
This is solitary animal not having certain territory. “Mangrove robbers”
meet at the distance no more than one kilometer from the nearest sea coast,
but
along the rivers they come deeper into land areas: up to 3 – 4 kilometers.
They frequently lodge on coast of brackish lakes where sea water filters
from sand. Usually the crab uses as shelter natural niches, but it also can
dig
own holes or occupy and expand abandoned holes of rodents.
For breeding females of this crab right after pairing migrate to the sea:
eggs of these animals develop only in salt water (salinity must be not below
the
half of oceanic one). At “mangrove robber” the development is direct: from
eggs tiny crab babies hatch. First days of their life the female carries
them under an abdomen. Gradually the young growth molts, and gets over on
back of
female. At this time female shows surprising for large crustaceans care for
posterity: during meal the young growth can drag slices of parental meal,
and the female patiently waits, while the posterity will be sated. At this
time
it is ready to attack any animal of size comparable to it, if it will consider
its presence dangerous to young growth. Crabs of this species breed the year-round,
giving 2 – 3 hatches of 150 – 200 young crabs.
Though “mangrove robber” has no large enemies, its life from the very beginning
is threatened by various tiny creatures. During the bearing of posterity
crabs are threatened with flies of the special kind – crab
botflies (Carcinotaba vorax). They actually are not true botflies, and
belong to… horseflies. Adult flies of this species suck blood of crab, and
larvae develop among eggs, destroying
them almost entirely. The small agile coastal bird resembling nuthatch, the crab-cleaning rattlebird
(Carcinornis crepitaculum), having the tenacious paws,
permitting to perch on carapace of crab, saves “mangrove robber” from crab
botflies. It feeds with these flies, trapping them, perching right on body
of crab. At an opportunity it also clears crab of other parasites – small
ground leeches and ticks. But sometimes these birds turn from friends to
enemies:
they equally willingly eat young crabs.
This species was discovered by Arseny Zolotnikov.
Grooming
crab (Hygeioportunus accuratus)
Order: Decapods (Decapoda)
Family: Swimming crabs (Portunidae)
Habitat: reeves of Pacific Ocean, tropical and subtropical regions.

Picture by Fanboyphilosopher
Coral reeves of Holocene epoch had represented the community
of live organisms, not conceding in variability of species to tropical rainforests.
In conditions of food abundance inhabitants of coral reeves frequently showed
very strict specialization in feeding and habit of life. But this circumstance
had made them dependent on existence of ecosystem as a whole. Therefore, when
at the boundary of Holocene and Neocene reef-building corals had died out, the
most part of rich reef fauna had disappeared after them.
In Neocene instead of former reeves new ones have appeared. Not corals, but
other animals – sponges, huge bivalves
and other sedentary animals form them. At these reeves like at Holocene ones,
various sea animals search for shelter and food. In struggle for existence they
also show miracles of specialization. Among inhabitants of Neocene reeves crabs
of various families are characteristic – these animals are mainly predators
and scavengers of reef. They often
develop very effective and deadly ways of food getting. But one of crabs had
evolved to other way of specialization – it had staked not on bloodthirstiness,
but on cooperation. And, of course, it is very successful from the point of
view of survival.
Around of one huge shell growing at some distance from body of reef the school
of fishes swims. They stay near the shell for rather long time, though obviously
they are not going to be fed. Fishes only replace each other above the shell,
turning in round dance. The reason of interest of fish school to this shell
is a small motley crab creeping on skin of one fish. This creature is not afraid
that fishes will eat it: crab slowly moves on fish scales, searching for parasites
attached to it. Having finished to clear the fish, crab leaves it and quickly
moves to other fish.
This animal is grooming crab, the descendant of one species of predatory swimming
crabs. It differs a little in anatomy from congeners leading predating way of
life in other parts of ocean. At grooming crab there is the same flattened back
pair of walking legs with which help it swims quickly. On the bottom side of
these legs small holes are advanced – they are suckers helping the grooming
crab to attach to smooth sides of fishes. This crab is rather small – the carapace
width of the adult female reaches 6 – 7 cm; males are a little bit smaller,
but have larger pincers.
The main hunting weapon of swimming crabs is pincers with which help these predators
kill fishes and other prey animals. At the grooming crab pincers had turned
to thin tweezers with which help animal gathers parasites from skin and gills
of fishes, and also “cuts out” sites of dying off tissues from wounds and grazes.
Sharp sight helps the grooming crab to search even for small parasites: his
eyes are spherical and placed on short mobile stems. They are supplied with
set of facets providing recognition of small details.
For “advertising” of rendering services contrast colouring serves to grooming
crab. Body of this crab is coffee-black, but the middle part of carapace is
snow-white with small round black spots. Pincers of animal are also white with
black tips and separate black spots. Swimming legs are red on tips: when crab
swims near the shelter, their movement involves fishes wishing to be cleared
of parasites.
Obviously, first time only young growth of any reef species of swimming crabs
rendered cleaning service to fishes, attacking parasites of large fishes, as
if the prey. They were too small to be prey of large fishes, but near to them
the constant protection had been provided. This vital strategy appeared favourable:
adult grooming crab, actually, eats food of animal origin, gathering it from
bodies of fishes, being well protected – its behaviour combined with bright
colouring became a signal of lowering of aggression for fishes.
Each grooming crab has the certain territory on reef zealously protecting from
congeners. As a rule, the best possession is one having any object seen from
apart – group of sponges, stone or mollusk shell. But near to them the shelter
is necessary located, in which crab hides in case of danger.
The crab constantly lives at its own territory. Only male can leave the possession
in searches of females ready to pairing. But in this case there is a danger
that it may find at the territory any stranger lodged there during its absence.
Usually such ones are “tramps” – small crabs not having constant territory and
eating casual food. Such animals are weaker in comparison with “thrifty” crabs
– they strongly concede to them in growth. To banish such animals is easy for
the male of grooming crab. Females of this species are larger and more aggressive,
rather than males, therefore their territory becoomes free only owing to death
of the animal.
The female lays some thousand eggs from which in three weeks tiny young growth
bursts. Young animals spend about two weeks in plankton above reeves, and then
fall on bottom and pass to habit of life characteristic of adult individuals.
Young crabs are transparent with several black spots on sides. They clean of
parasites shrimps and small fishes. Approximately at one-year-old age young
crab is able to breed.
On reeves of Pacific Ocean grooming crab has imitator from among fishes – the
impostor boxfish. This fish
imitates colouring of crab, but does not render cleaner services to fishes.
But being attacked this boxfish emits a jet of poisonous liquid. Such way of
protection benefits not only to it, but also to grooming crab which is also
not attacking by predatory fishes.
Crystal swimming crab (Crystallonectes crystallinus)
Order: Decapods (Decapoda)
Family: Swimming crabs (Portunidae)
Habitat: Indian and Pacific oceans, thickness of water.
In early Neocene many ecological niches of plankton organisms appeared empty
after mass extinction of plankton caused by natural accidents at the boundary
of Holocene and Neocene. But the biosphere as a whole has one remarkable property:
ecosystems develop to the way of maximal use of resources given by environment.
Therefore right after extinction of plankton organisms of Holocene epoch the
thickness of water began to be settled again.
Among new representatives of plankton crustaceans are especially characteristic.
Except for myzids, scuds and shrimps, even crabs had appeared in plankton.
Representatives of swimming crabs family were one of first invaders of thickness
of water. In Neocene they had generated even the separate family which has
kept neotenic features. But in parallel to them in the ocean there are also
descendants of the basic branch – true swimming crabs of specialized species.
Benthic swimming crabs were predators, and they still live on reeves. And pelagic
swimming crabs became predators of plankton.
The crystal swimming crab is one of predatory pelagic crabs. By features of
anatomy this animal is very close to representatives of family Infantoportunidae.
It is the descendant of crabs actually became transitive forms between families
Portunidae and Infantoportunidae. Independently of true Infantoportunidae it
had got similar adaptations to life in plankton.
Body of this crustacean is transparent, flattened, about 4 cm long and about
5 cm width. For simplification of soaring in water two long spikes on each
side of body serve to crystal swimming crab. They are slightly bent back and
covered from back edge with the bristles increasing a surface of shell.
Walking legs of first three pairs are short and thin, reduced up to small hooks
keeping segmentation. They can not serve for walking any more, and animal uses
them only for catch keeping. At males legs of third pair are larger, rather
than at females. They are a little similar to trapping legs of soothsayer,
and on their internal side small jags are advanced. Such legs of the male help
it to keep the female at pairing. In rest they are fold at the bottom side
of crab body and pressed to pleon to not prevent to swimming. The male differs
from the female a little: it is smaller, but has larger pincers. For pairing
the male tries to swim up to the female behind and to seize it by third pair
of walking legs. It avoids so an attack from the part of female – at this species
the cannibalism is usual.
Back pair of legs serves for active swimming as at all swimming crabs. They
are flattened, and the terminal segment is expanded and supplemented with folding
hairs along the edge. When crab pushes by legs, they unbend, strengthening
the effectiveness of the rowing.
At crystal swimming crab there are flat and wide pincers. When it is swimming,
pincers serve as rudders of depth and turn, permitting to change fast the direction
of movement. Hunting small animals, this crab is able to make sharp rushes
and turns on the spot. Also it is very important for predator to have well
advanced sense organs. This crab distinguishes prey and danger with the help
of sight. At it there are large shining eyes numbering numerous facets. Due
to such feature of eyes crystal swimming crab is able to distinguish slightest
movements around of itself. Short antennae of this crustacean are feather-like
– they help to catch a smell of prey.
Prey of the crystal swimming crab includes others plankton animal – worms,
jellyfishes and fish fry. Usually this crustacean passively soars in thickness
of water, having stretched pincers and legs in sides. Having noticed catch,
animal instantly turns to the condition of “alertness”: it folds pincers in
front of itself and presses walking legs to pleon. When catch is too small,
crab simply waits while it will approach nearer to it. But it can chase large
catch at the distance of several meters, that it very long for so small creature.
Fertility of plankton animals usually reaches astronomical scopes. But survival
rate of their posterity is parts of one per cent. At crystal swimming crab
it is, on the contrary, rather small: no more than 200 – 300 eggs. But it is
compensated by care of posterity. Youngs of this species are tiny copies of
parents about 3 mm long. The posterity of this crab keeps for a long time on
the carapace of the female – till one week. For this time young crabs have
time to pass through two molts. Swimming legs of young crystal swimming crabs
are modified to suckers and serve for the attachment to the female. Their pincers
also are not similar to pincers of adult crab: they are thin and serve for
grab of food particles from water. The larvae of this species feed on rests
of female’s catch. The female bearing posterity eats catch not the same mode,
as the male or the female without posterity does it: it does not tear off pieces,
but chews the caught prey and does not swallow it. Particles of food get in
water, and the posterity catches them. Till the bearing of young growth the
female eats a little and prefers to swim slowly to keep offsprings on its back.
After the second molt swimming legs of young crabs of this species get the
shape characteristic for them, and they abandon mother ready to independent
life. At the age of four months they reach the size of adult crab and start
to breed. Life expectancy of crystal swimming crab does not exceed two years.
Bloodsucker crab (Infantoportunus sanguisugus)
Order: Decapods (Decapoda)
Family: Infantoportunids (Infantoportunidae)
Habitat: Indian and Pacific oceans, from moderate up to tropical latitudes
of both hemispheres, the parasite of pelagic animals.
In the process of evolution it happens, that the new group of live organisms
occurs from representatives of one of already existing groups by the evolution
of larval stage. The larva as if “replaces” an adult stage, getting ability
to breeding. This process occured independently at different live organisms
not related to each other. In Neocene by such way the separate family of crabs
– Infantoportunidae family – evolved from swimming crabs (Portunidae). Larval
stage began prevailing at them, and even adult crabs keep some larval features.
The most appreciable attribute of these animals is flattened body with transparent
shell through which internal organs are visible.
Swimming crabs during the history were active predators. Small fishes and various
invertebrates were their catch. Specialization to predating at infantoportunids
was developed even deeper – some of their species passed to parasitic habit
of life at all. Jaws of these crustaceans had changed: they became narrow and
long. During the feeding of the animal they form a kind of pointed proboscis
which length at some species can exceed width of carapace of the animal. Predatory
infantoportunids catch small animals by pincers and suck them out.
The bloodsucker crab is the least specialized to pelagic way of life species
of family. It lives in sea shallow waters and sucks blood of various fishes.
This is rather small translucent crustacean with the advanced swimming legs
and disc-shaped body. The own colouring of crab is greenish, dark liver and
intestines with food appear through the shell. Width of carapace of the bloodsucker
crab is about 6 – 7 cm. Walking legs at this species are very small and weak,
turned to similarity of hooks. Pincers of this crab are also thin and lengthened.
Swimming legs (last pair of walking legs) are very well advanced – with their
help the bloodsucker crab is able to swim quickly, chasing fishes.
At the bloodsucker crab sense organs are well advanced. At it there are large
eyes and good sight, and also there is the keen sense of smell by which it
is guided in search of animals on which it eats. This crustacean eats exclusively
blood of fishes. Having noticed the school of fishes, hungry bloodsucker crab
hides, and attacks when they appear near to it. This animal is able to swim
quickly, sharply changing the direction of movement; therefore not any fish
can catch it. But the crab easily reaches the chosen fish, especially when
on its body there are small wounds – the smell of blood strongly attracts this
animal.
Bloodsucker crab attaches to wounded areas of fish body, and when the fish
is not wounded, it prefers to perch behind a head, near gills. Having chosen
by smell the suitable place, crab finds blood vessel and sticks proboscis in
skin of fish. When the animal is hungry, it is almost not visible on skin of
fish because of translucent body. But when the crab starts to be sated, the
exhausted blood of fish appears visible through its intestines. Due to elasticity
of shell the crab can suck blood in quantity up to 50 % of its body weight.
The full animal unhooks from fish, and tries to hide itself at sea-bottom.
Hiding from enemies, crab can be dug in sand, raking it on itself by back legs.
Bloodsucker crabs breed the year round. The male at this species is much smaller
than female, and also leads more active habit of life. Females of bloodsucker
crabs live at the certain territory and come back to it after the attack on
fish. Males have no constant territory: they wander in shallow waters, searching
for females ready to fertilisation.
Fertility of the female of bloodsucker crab is about 500 large eggs. Egg-laying
repeats each 2 months. Eggs are left with floating pelagic larvae which will
spend in plankton over three months. In plankton they are active predators
also attacking mainly young growth of fishes. The young bloodsucker crab settles
on bottom, being about 2 cm long, and gradually passes to parasitic way of
life.
The species close to the bloodsucker crab, jellyfish pilferer (Infantoportunus
medusivorus), has lodged in open ocean. It is small species of crustaceans
- width of its shell is usually no more than 4 cm, and the length is even less.
Walking legs at this animal are reduced up to thin hooks, and on the bottom
side of swimming legs suckers develop. This crustacean lives and eats on jellyfishes.
The crab sits on the cupola of the jellyfish from above, avoiding touches of
poisonous tentacles. On one jellyfish one female of this species lives always,
and all noted characteristics of species concern to it. Males of jellyfish
pilferer sharply differ from females: they are animals not exceeding 1 cm length.
The male actually represents the larva with developed sexual glands. Covers
of body at it are transparent, and the body is short and also very much extended
in width. It lives on the female in her branchial cavity, and eats blood of
female, piercing its covers. Actually, the male parasitizes on the female.
Its contribution to breeding is limited to that it regularly lays spermatophores
to sexual apertures of female. In the rest time male is completely motionless.
His walking legs are reduced, only former swimming legs (back pair of walking
legs) modified in suckers are advanced. In connection with parasitic habit
of life the sight of jellyfish pilferer is very weak – in each eye there is
at all about one hundred facettes. But at it the sense of smell permitting
this crab to search the host animal is perfectly advanced.
At jellyfish pilferer there is the sharp proboscis reaching 10 cm length. It
is almost constantly dipped in body of jellyfish on which this animal lives.
The proboscis of jellyfish pilferer pierces covers of the jellyfish and reaches
its digestive cavity. The crustacean eats only animals semidigested by jellyfish.
The female of jellyfish pilferer lays hundreds of eggs per one week. From them
small pelagic larvae having no sex hatch. First days of life they are predators,
and attack pelagic crustaceans. Further larvae search for young jellyfishes
and settle on their bodies. On the jellyfish larva transforms to young crab,
and the pointed proboscis develops at it. Such larvae turn to females. Having
settled on the female, larva turns to the male.
If the jellyfish perishes, the female of jellyfish pilferer moves to other
jellyfishes – it is able to swim and can spend any time in thickness of water,
expecting for new host jellyfish.
Gill-sucking
crab (Branchioportunus parasiticus)
Order: Decapods (Decapoda)
Family: Infantoportunids (Infantoportunidae)
Habitat: Pacific Ocean, from equator up to Subantarctic.
In Neocene crabs of swimming crabs family (Portunidae) distinguished by active
predating way of life had made the attempt to develop the pelagic habit of life
among many groups of coastal sea animals. They had successfully realized the
opportunity received during the global changes in biosphere and had formed the
separate family of crabs Infantoportunidae distinguished by preservation of
some juvenile features in adult condition. Representatives of Infantoportunidae
are adapted to inhabiting in thickness of water where their whole life cycle
passes.
Representatives of new family had actively started to evolve, developing various
ways of food getting. They became numerous predators of plankton, and some species
passed to parasitic habit of life. One
species of infantoportunids is feeding, exhausting contents of gastral cavity
of jellyfishes. And its relative, the gill-sucking crab, had passed to parasitism
at vertebrate animals. It lives on gills of sailerfish
– very large pelagic fish of southern part of Pacific Ocean.
Similarly to the majority of specialized parasitic species of animals this crab
had undergone the deep degeneration in comparison with ancestors. The adult
individual of the branchial crab female is about 2 cm long, and the male is
even smaller. Body of this animal it very thin and translucent; its bottom side
is flat and top is slightly convex. Walking legs at the female of branchial
crab are strongly reduced and modified to hooks with which the animal keeps
for branchial arch of fish. For additional fixing suckers serve to branchial
crab: they are modified terminal blades of back legs used for swimming earlier.
The adult female of branchial crab almost constantly keeps motionlessly on gills
of fish, and is unhooked from it only for the period of molt. Sometimes the
animal can slowly creep from one site of gills to another, moving suckers alternately.
Males of this species are much more active, rather than females – they are able
to creep on branchial arches of fish, fertilizing females. At them walking legs
are more advanced. Among males living on gills of the same fish, battles may
happen, resulting in death of one of them – strongest male kills all weaker
ones and privately-owned fertilizes females living on gills. Sometimes in population
there are hermaphrodites able to fertilize each other (self-fertilisation does
not take place).
Mouth organs of branchial crab are modified to pointed proboscis with which
help animal is feeding. In adult condition branchial crab is able to eat only
liquid food – blood of fishes.
In connection with almost motionless habit of life the adult crab has degeneration
of all sense organs. Only the chemical feeling and touch appear more or less
advanced at adult individual. Till the process of growth at branchial crab eye
reduce.
The female of branchial crab lays weekly about some hundreds eggs. After 2 –
3 days from them tiny plankton larvae burst leading the predating habit of life.
They attack mainly spineless invertebrates. After 5 – 6 molts (at the age of
approximately 2 months) young animal should get on host fish, otherwise death
threatens to it. The young branchial crab does not search for the host fish:
it finds the crab itself. The animal gets in mouth of fish during its feeding:
when sailerfish is swimming in congestion of plankton with widely opened mouth,
young crabs sediment on its gills and attach to them. First time they keep features
of diet of young animal – eat plankton sedimenting on gills of sailerfish. But
after each new molt the developing branchial crab loses more and more attributes
of freely living animal – at it legs shorten, suckers develop and eyes reduce.
When pincers of animal reduce so that crab could not gather plankton animals
sedimented on gills of fish, it passes to feeding exclusively on blood of fish.
Velvet worms |
Velvet
poison-bearer (Venenophorus holosericum)
Order: Euonychophora (Euonychophora)
Family: Diverse-legged peripatopsids (Heteroperipatopsidae)
Habitat: north of Meganesia, rainforests.

Picture by FanboyPhilosopher
In the age of people there lived in New Guinea the Onychophora
family of peripatopsids, which was characterized by reproduction by egg-laying.
Their Neocenic descendants form a seperate family of diverse-legged peripatopsids,
which is characterized by uneven development and the specialization of glue
glands. One of the two glue glands (the left) has become a poisonous gland emitting
a powerful nerve poison of viscous consistency, while the right one remains
a “glue gun” as at ancestral forms. The walking legs of diverse-legged peripatopsids
have also undergone some changes, differing from the legs of ancestral forms
that retain approximately the same structure along the entire length of the
body. The two pairs of front legs of these animals were drawn together; they
are elongate and grasping. In a calm state, they convolve into a spiral and
are not involved in movement. The front part of the body when moving is slightly
raised above the substrate.
These strange creatures move in two ways: by contractions of their dermomuscular
tube (for hunting grubs in the soil) or by folding up like a leech or inchworm,
when hunting for termites on the bark of trees. Following the hunting legs are
two pairs of legs which have become very powerful and tenacious, their tips
developing into corneous claws. These legs form the front “attachment disk.”
Several pairs of legs, following the front walking ones, are reduced, and next
to them lung sacs appear. The last 4 pairs of legs had also undergone specialization:
the last pair of legs became a cutting scoop, the penultimate were lengthened
and turned into a kind of antennae, and the remaining two pairs of legs formed
the rear attachment disk and gained strong claws.
The body length of the velvet poison-bearer reaches 10 cm. This animal looks
like the caterpillar of some butterflies, but differs from them in its manner
of movement. The underside of the animal is creamy white, and the top is red
with a violet hue. The animal leads a nocturnal life and is only found in the
day time in wet weather, after a rain.
In case of danger the velvet poison-bearer lifts up the front part of its body,
spreading its trapping limbs and moving them in the air. In this case, the white
body is visible in the low light of night. If this warning does not force the
enemy to retreat, the animal splashes onto it a jet of poison, determining the
direction of the “shot” by smell and movement in the air. This poison is very
dangerous for animals with thin skin: predatory snails, amphibians, and small
reptiles. Prolonged exposure to the poison can kill terrestrial arthropods (ingested
into the body through the trachea or cuticles in the joints of the legs). For
larger reptiles with thick scales, birds, or mammals the poison is dangerous
if it gets into the eyes, nostrils, or mouth.
In the forests of northern Meganesia a close species lives: the drill
poison-bearer (Venenophorus militaris), which behaves differently:
it is a social species, forming clusters of a few dozen individuals. The body
length of this species is much less: no more than 4 cm. Its body is colored
a pale pearlescent pink. Drill poison-bearers creep in “caravans”, as some caterpillars
of butterflies, keeping track of a creeping individual in front. Termites represent
their main prey. These velvet worms look for the termite trails in the undergrowth,
and their perianal scrapers break through the roof of the gallery and they begin
to hunt for termite workers. If they are attacked by soldier termites, the animals
glue their legs with sprays of glue and eat the immobilized insects. Also, these
animals are not afraid of ants and often feed near their trails. The drill poison-bearer’s
sticky liquid is used for primitive forms of construction: they often make a
shelter, sticking to the ground the leaf of a plant, or rolling it in half with
the underside on the inside. This shelter always provides the optimum microclimate.
This species of invertebrate was discovered by Korshin D.E.,
a member of the forum.
Translated by FanboyPhilosopher
Mollusks
|
Shore
melania (Paludimelanoides subterrestris)
Order: Sorbeoconcha(Sorbeoconcha)
Family: Melanias (Melaniidae)
Habitat: rainforests and swamps of Zinj Land.
Earthworms are fairly named as “guts of the ground”: their role in soil
formation processes is really irreplaceable. But they live not everywhere:
worms avoid damp marsh soils. And in such biotopes of the Eastern - African
subcontinent they are replaced by other digging animal: shore melania, the
representative of gastropods. This species have evolved from one of species
of melania snails (Melanoides), widely settled in tropical fresh and brackish
waters. These snails spend a lot of time digging in ground, therefore they
needed only few changes to pass to life in damp marsh ground.
Shore melania is digging snail about 4 cm long. At it there is strong conic
shell – the main ground digging instrument. Coils of shell are covered with
cross lines of limy knobs, that’s why shell is similar to drill (and it
works approximately like it). Shell color is grey with cross brown strips.
Knobs on shell are quickly erased; therefore they frequently look as white
dulled hillocks. The creeping sole of snail can extend and compress, extend
forward and contract sharply - this organ helps snail to bore ground. In
friable ground snail digs burrows, pushing aside and ramming ground by shell.
And in more dense ground it works differently: snail pushes creeping sole
far forward between clods of grounds, and then sharply contracts it, tightening
shell forward and simultaneously moving apart the ground. For protection
against enemies the thick corneous operculum closing fauces serves to snail.
The head of shore melania is extended to long proboscis assisting to snail
to get food from narrow cracks in ground. Shore melania eats parts of plants
begun to rot, but sometimes it also eats soft tips of live roots. Shore
melania plays the important role in biocenoses of swamps and flooded rainforests:
boring damp marsh and wood ground, it improves conditions for growth of
plant roots.
At night snail creeps out on surface of ground in searches of rests of plants,
hiding in holes in day time. As well as its ancestors, shore melania is
live-bearing animal. This snail is hermaphrodite, therefore two any individuals
may mutually fertilize each other. Young snails develop in pallium cavity
of parental individual within one week. Completely generated little snail
about 2 - 3 millimeters long is born under ground and at once lives independently.
At it there is rather strong shell, but nevertheless the young growth frequently
becomes prey of soil mites and small beetles.
Glassy
Cave Snail (Vitriconcha sp.)
Order: Sea and River Snails (Littorinimorpha)
Family: Small Operculate River Snails (Tateidae)
Habitat: Glacier-fed cave-waterways and adjacent areas downstream, North
and South Island of New Zealand.
Cave ecosystems often produce unique species of animal, descended from more
typical ancestors found in surrounding countryside. So it was that the New
Zealand Mud Snail (Potamopyrgus antipodarum) gave rise to the genus Vitriconcha,
which are common grazers within the cave waterways of New Zealand.
A small aquatic snail, it possesses an operculum and an elongate, coiled
shell with between 5 and 8 whorls, shell is transparent and glassy reaching
a length of 9 millimetres maximum. Its body is pale, transparent and slightly
opaque, its gut can be seen from the outside. One pair of sensory tentacles
protrude from the head, the mouth extends into a short proboscis, eye-spots
are absent and this snail does not react to light. Diet consists of algal
and microbial films that it scrapes from submerged surfaces, it sometimes
may venture a small distance out of water to feed on mildew that covers
damp surfaces. This snail is sometimes found living outside of caves, in
waterways a small distance downstream from cave rivers. It is able to survive
a fair range of salinities, and pools containing concentrations of dissolved
mineral are tolerated by it easily.
The snails breed at any time of year and are able to reproduce sexually
or by parthenogenesis, breeding occurs once or twice within a year. The
production of parthenogenic young is usually catalysed by stress or low
population numbers within a given area. Distinct sexes exist, fertile males,
fertile females, and clonal parthenogenic females. A female will invite
the attention of males by secreting a strong chemical scent in her slime
trail, males seek to inseminate the female, and the female is mounted by
their apertures fitting together. Mothers will seek a sheltered underwater
crevice to brood a clutch of eggs which may produce as many as 30 live young,
a single female may produce as many as 100 young within a year. Females
reach breeding size at about 4 millimetres, about 6 to 7 months of age.
Lifespan may be up to 1 and a half years, predators include crustaceans,
tadpoles and aquatic insects.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Poisonous
orb snail (Paraplanorbis virosus)
Order: Basommatophora
Family: Orb snails (Planorbidae)
Habitat: fresh waters of Europe.

Picture by Pavel Volkov
Pulmonate molluscs are widespread in fresh waters of the world. The epoch
of anthropogenic pressure passed completely unnoticeably for many kinds
of these animals – the ability to live in polluted waters made it possible
for many species to keep a sufficient number of populations and vast area.
After the end of the human era in several tens of thousands of years all
pollutants became completely decomposed or they got to be buried under the
deposits. The freshwater bodies became clean and the snails continued their
normal existence. The majority of species of aquatic snails developed no
new features in comparison with their ancestors: their habitat and their
way of life proved to be rather conservative, and the stabilizing selection
kept the aquatic snails as completely recognizable ones even the millions
years after the human disappearance. But nevertheless, among them species
having no analogs in human epoch evolved.
In the fresh waters of Europe rather large species of orb snails lives.
The diameter of its flat-spiral shell reaches 35 – 40 mm. The coloration
of this snail is quite distinct against the background of the surrounding
aquatic plants and is very garish – broad black bands stretch on light yellow
background along the turns of shell. The body of mollusc is black. An animal
that will decide to attack this snail can suffer seriously. This species
is completely justified in being named a poisonous orb snail – a strong
poison is accumulated in the body of this mollusc, and its coloration serves
as warning for the predators.
The poisonous orb snail independently produces only a part of its poison.
This mollusc obtains a large part of its poisonous substances from its food:
the poisonous orb snail feeds on the poisonous water plants of Alismataceae
family, and also on the poisonous marshy plants of the Apiaceae family.
The alkaloids from the plants are accumulated in a special gland, which
opens into the mantle cavity of mollusc. Having sensed an approach of carnivorous
worms (leeches, planarians) or insect larvae, poisonous orb snail emits
into the water a portion of its accumulated poison and then hides into the
shell, tightly adhering to the substratum. Spreading through water, the
poison frightens away the snail’s enemies. This snail is also poisonous
for the fishes – a part of the alkaloids is accumulated in the musculature,
giving this snail a bitter taste.
Like the majority of the related species, poisonous orb snail is a hermaphrodite.
This species is not capable of the self-fertilization, and for successful
reproduction a mating of two individuals is necessary. In the rare cases,
in the rarefied populations of this species that are located at the edges
of species area, separate individuals develop the ability of parthenogenesis:
their unfertilized eggs double their number of chromosomes to the diploid
while still in the oviduct of their parent, and further on, when laid, they
begin division and develop into normal embryos. This snail glues flat slimy
batches of eggs to plant leaves, making up to twenty batches of eggs during
the season. The young snails have dim coloured shells: they do not have
a poison in their bodies, and they are vulnerable to predators. The young
snails accumulate a quantity of poison sufficient for the self-defence later
on, and a large number of the young snails perishes from the aquatic predators.
The sexual maturity of the young snails begins at the age of two months,
and the lifespan reaches three to four years.
Translated by Bhut, the forum member.
Reed-eating
pond snail (Galba calamivora)
Order: Basommatophora (Basommatophora)
Family: Pond snails (Lymnaeidae)
Habitat: Eurasia from the Three Rivers Land through Siberia to the rivers of
the Far East
The Neocene is the time of appearance in nature of new, and often unusual species.
But, alongside them, many species of the living organisms exist, that are almost
unremarkable. However, this circumstance does not understate the role they
play in the ecosystems of Neocene epoch.
The high stems of reeds, cane and macereed form extensive thickets in the shoals
of rivers and lakes of Eurasia. Here the large animals feed, which have the
adaptations that make it possible to digest this rough and fibrous food. But
alongside them the whole world of small creatures dwells, and they are also
adapted to feed on such sort of plants. The insects gnaw throug the soft core
of stems or drill through the rhizomes of the plants, slowing their growth.
And on the edge of thickets the stems of these plants lean down to the water,
chewed at their very bases. These are the traces of feeding of another species
of herbivorous animal – the reed-eating pond snail, a representative of the
pulmonate molluscs.
This aquatic snail is one of the basic species in the freshwater reservoirs.
The appearance of reed-eating pond snail has changed rather little in comparison
with its ancestors of the human epoch. This is a descendant of the pond snail
Galba truncatula, a common species of Eurasia. Because of their unpretentiousity
pond snails did not suffer from human activity. They successfully inhabited
polluted reservoirs, gaining an advantage over the more sensitive species.
The reed-eating pond snail is well adapted to feed on the tough, fibrous stems
of aquatic plants. It feeds exclusively on the stems of the reeds and other
coastal plants, without competing with the other gastropods, which prefer algal
cover or the greenery of softer plants.
The length of the shell of this species is up to 40 mm. The shell of this species
is short-ened and slightly flattened, with blunted top; in its shape it is
a transitional type to the planispiral shell like shells of orb snails, but
currently with a definite top. In a transverse shear it appears to be rather
oval in shape, excluding the increased last turn, where the lung of the mollusc
is located. The surface of shell has pattern of brown spots against a yellowish
background, and the soft body of the mollusc is yellowish-grey. On the head
of this pond snail there is a pair of blunted conical tentacles with two small
eyes at their base.
The reed-eating pond snail feeds constantly, interrupting only to creep to
the surface from under the water to breath air. The air amount in its lung
in enough to stay under water for approximately 45 minutes. This mollusc can
digest the toughest parts of the reed stems – its digestive path produces enzyme,
which helps to decompose the cellulose that contained in it. The guarantee
of success of this mollusc is its strong radula (“tongue”), at the tip of which
large corneous teeth grow, helping it to scrape the rigid plant fibres. Each
motion of radula cuts off a layer of plant tissue in common with the fibres.
This species feeds under water, gnawing the stems at their bases. Among the
thickets such stem is simply immersed in the water due to its own gravity,
keeping its vertical position, and the snail continues to eat it up from below,
until only the top remains. At the edge of thickets such stems frequently fall
into the water and are carried away by the flow.
This species is hermaphroditic like its ancestor. The reed-eating pond snail
lays eggs from May to the middle of September. The break between the egg laying
is about 3 – 4 days. The snail lays 10 to 15 eggs at the same time, enclosed
in slime capsule. Small snails hatch after a week. They rapidly grow and reach
sexual maturity at the age of 1.5 months. The reed-eating pond snails live
for about 2 years.
This species of molluscs is discovered by Nick, the forum
member.
Translated by Bhut, the forum member.
Hawaiian
bird-eating snail (Horrohelix ovisuga)
Order: Stylommatophors (Stylommatophora)
Family: Spiraxids (Spiraxidae)
Habitat: Hawaiian islands, rainforests of hillsides and valleys.

Picture by Alexander Smyslov
In human epoch at Hawaiian Islands the set of ground snails had lived. They
formed tens of various species distinguished by shell coloring. But their prosperity
had ended when people had introduced from Africa the huge snail achatina (Achatina
fulica). This mollusk multiplied, and became the agriculture pest. And as biological
measures of struggle from Florida predatory snail Euglandina rosea (Rosy Wolfsnail)
had been introduced. In common with collectors shells it had exterminated a
plenty of endemic local species of snails, but not introduced achatina. Due
to continental “training” and rather soft climate of Hawaii till the epoch
of biosphere crisis it had successfully gone through climatic cataclysms of
the boundary of Holocene and Neocene. The Hawaiian bird-eating snail is its
lineal descendant.
It is rather large mollusk – shell length is about 15 cm, weight of adult snail
is approximately 400 grams. Shell of animal is egg-shaped with the blunt tip
and wide fauces; last curl of it is the biggest. The shell is colored very
floridly and brightly: red - brown with pattern of black strips of different
width stretched along curls.
Body of bird-eating snail is long; its forward part is flexible and head is
rather small. Covers of bird-eating snail have mesh pattern: dark red gauze
on pinkish background. On head there are two pairs of feelers; on long feelers
(“hornet”) eyes are placed, and another pair grows near the mouth.
This mollusk lives in forests and is rather mobile: the snail easily gets on
trees and creeps on branches in searches of food.
The Hawaiian bird-eating snail has completely kept, and even has strengthened
the injurious bents inherent in its ancestor. It eats various ground mollusks,
and attacks small vertebrate animals: it sucks bird eggs and even eats nestlings.
Sight sense of this mollusk is bad; therefore it is not able to track down
catch, as a cat. But sharp sense of smell helps this animal to find nest with
eggs or nestlings. Having found out a clutch of any bird, this snail bites
through eggshell by radula, and licks contents of egg. Then it bites edges
of the bore, expanding it, and thrusts head inside of shell. It takes about
twenty minutes to eat one egg by size like pigeon’s one at this snail. Feeding
by eggs is especially important during growth of mollusk: this way the snail
fills up stocks of calcium necessary for shell forming.
Bird-eating snails are hermaphrodites, and also breed the year round. Before
egg laying the animal feels need for protein food, and much more willingly
eats various invertebrates and nestlings of small birds. The saliva of this
snail has paralyzing properties, therefore the animal bitten by it any more
cannot escape by flight.
When snail has received enough protein food, in its organism development of
eggs begins. In oviduct the cocoon containing about hundred of rather large
eggs is formed. The snail hides it in wood ground between roots of trees. For
one year it can make up to ten such clutches. In two weeks from cocoon young
snails hatch. They eat small insects, and at the age of about one year their
shell reaches size about 10 cm. Further growth of snail is slowed down, but
it starts to breed.
Despite of successful existence in ecosystem of Hawaiian Islands, this mollusk
has many enemies. Ground crabs easily break open with pincers shells of these
snails, and some birds have adapted to eat various ground snails including
this species.
Ant
snail (Myrmiachatina melanoleuca)
Order: Stylommatophors (Stylommatophora)
Family: Achatinids (Achatinidae)
Habitat: tropical woods of Hawaiian islands, ant hills.

Picture by Pavel Volkov
Social insects are representatives of two absolutely not related
orders: hymenopters (ants) and termites. Both those insects differ in various
degrees of development of building art, but always live in colonies with complex
system of hierarchy and castes. Any aggressor solved to attack settlements of
these insects, takes the risk to receive serious repulse from insects of soldier
caste, and can perish easily.
But despite of real danger which is represented by these insects for other live
creatures solved to penetrate into their dwelling, constructions of both groups
of insects became a habitat for set of species of other animals - from smallest
insects up to lizards. To escape in society of these well armed insects, other
invertebrates develop smart methods of interaction with owners of dwelling.
Some of them are very cautious, and whenever possible try to slip away from
owners of dwelling, others camouflage to look like lawful owners of dwelling
by smell and appearance. But the most widespread way of interaction is to become
“friends” of armed insects and to provide to itself safety by that at life side
by side with them. The simplest way of this interaction is to produce substances
which ants or termites like. In nests of these insects it is possible to find,
for example, beetles and strange wingless flies secreting substances liking
to ants. For the sake of sweet secretions ants carefully preserve, and even
“breed” plant lice, protecting them and sometimes constructing shelters above
them. For the same purpose ants take care to caterpillars of hairstreak butterflies
(Lycaenidae family) which inconsiderately devour their own posterity. In historical
epoch ants represented an alien component of fauna on Hawaiian islands, but
to Neocene descendants of first introduced species became a part of the ecosystem
formed on islands, and some other animals began to “search for friendship” at
these well protected insects. Among them there was even one species of local
ground snails had lodged near to ant hills.
Hawaiian ant snail is rather small species of gastropods: diameter of its shell
does not exceed 5 cm. Certainly, it looks large in comparison with the majority
of snails known to humans, but much larger giant African snail (Achatina fulica)
introduced to Hawaii had been its ancestor - the shell length of this giant
reached 15 cm. For millions years of natural selection smaller individuals of
ancestral forms of this snail survived, and gradually it had turned to the dwarf.
At ant snail there is not bright, but contrast black-and-white shell colouring
forming striped pattern - at various individuals the ratio of black and white
colors varies, and striped pattern is individual. The body of snail is covered
with an epithelium of yellowish color.
The ant snail uses protection of ants because it secrets slime which involves
ants with taste and smell. Special glands specially intended for this purpose
are advanced on edges of mollusk’s creeping sole and marked by black points.
Insects thirsting for secretions, gather around the snail, and tickle it by
antennae. Replying to their action, the ant snail emits a portion of slime,
continuing feeding. These mollusks keep near settlements of some local species
of ants, expecting upon their “protection”.
This snail uses not only protection of ants, but also feed near to ant hills.
It eats not only plants, but also insects brought by ants in colony. It eats
even dead ants from colony which protects it.
The only time when it must leave a colony is the time of breeding. These snails
are hermaphrodites, and two any individuals can mutually fertilize each other.
When in organism of snail eggs ripen, it finds a secluded place away from tracks
of ants, and hides in wood litter about hundred of small transparent eggs. In
2 weeks from them the posterity hatches.
Young ant snails have no glands developing a liquid which involves ants. Therefore
they keep in thickets of grasses and on the bottom side of leaves. Later, when
shell diameter of snail reaches about 15 mm, these glands develop, and snails
find a colony of ants. It happens that some ants search for these snails, and
specially drag them to their dwelling.
Green
Leafy Slug (Phytolimax mimeticus)
Order: Air-breathing Snails (Stylommatophora)
Family: Leaf-veined Slugs (Athoracophoridae)
Habitat: Dense Tropical forest in the north of the North Island, New Zealand.
The invertebrates of New Zealand in the Holocene was typified by a high degree
of endemism and striking adaptations. One example were the leaf-veined slugs,
unusual flat slugs with somewhat degenerate anatomy (mantle is only small, and
face has only one pair of tentacles), but highly unusual outward appearance.
Unlike many other endemic forms here, some of these managed to survive and leave
descendants in the Neocene.
This slug is usually flat in shape and tapered, up to 12 centimetres long, coloured
bright leafy green with darker green speckles and lines. Body is covered with
unusual ridges that resemble the veins of a leaf. It has a thin, plate-like
internal shell, face has only a single pair of sensory tentacles. Body may become
thicker and shorter when moving or when disturbed. Usually this species appears
almost flat, on the surface of forest branches and leaves, mimicking the appearance
of a leaf completely, edges of its body can be wrinkled in order to resemble
an immature-or-imperfect leaf. This differs from its ancestors, which would
imitate fallen leaves instead of freshly-growing-ones. Like its ancestors, it
feeds by scraping off algae and mold that accumulate on the surface of forest
leaves and branches. This species, like all in its family are usually only active
at night, during the day they tend to cluster together in shady spots or on
the undersides of branches.
Individuals are hermaphrodites and usually mate by casually meeting upon the
surface of a leaf, and mutually inserting their genitals in order to exchange
sperm. A cluster of un-shelled eggs are fertilised and laid in a moist spot
under tree bark. Lifespan may be up to 4 years, and predators most often include
carnivorous snails or predatory insects, occasionally birds.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Hissing
slug (Virolimax stridor)
Order: Stylommatophors (Stylommatophora)
Family: Slugs (Limacidae)
Habitat: humid forests of temperate and subtropical zones of Pacific
coast of North America.

Picture by Alexander Smyslov
At the narrow strip between Rocky Mountains and Pacific coast of North America
in Neocene there were conditions favorable for growth of deciduous forests of
special type, so-called “moss forests”. In such forests there are favorable
conditions for growth of moistureloving plants and life of moistureloving animals
– amphibians and terrestrial mollusks. In warm Neocene climate some invertebrates
evolved to original and remarkable species. Large and brightly colored species
of slugs named hissing slug is very characteristic for moss forests.
It is herbivorous species of shell-less gastropods. The length of its spindle-shaped
body reaches 10 cm. Such large invertebrates frequently appear very vulnerable
for various predators – birds, reptiles and amphibians. But hissing slug protects
against them with the help of strong poison which is accumulating in slime covering
its body. At this mollusk bright warning colouring was developed – it is black
with longitudinal rusty-red strip on each side of the body. Disturbed hissing
slug additionally displays its self-protecting with the help of some features
of behaviour. At danger it starts to swallow air and inflates body. At this
moment bright colouring of its covers becomes especially appreciable - at puffed
up slug the forward part of the body becomes similar to small bubble. On body
covers at this moment large drops of poisonous slime are secreted. Puffed slug
moves head, displaying warning colouring to aggressor. When the enemy recedes,
mollusk starts to blow off, letting air out from the mouth. Thus it hisses and
even whistles loudly enough. Such sounds in addition frighten off from it small
predators from among vertebrates.
Hissing slug is peaceful vegetarian; this one eats moss and mushrooms. And it
eats poisonous mushrooms without harm for itself, and uses their poison for
self-defense – poisons of mushrooms (muscarine and even more poisonous amanitine)
secret plentifully in slime of disturbed mollusk. This slug keeps mainly on
the ground, among windfallen trees and large grassy plants.
Hissing slug is hermaphrodite (it is typical of the present group of mollusks).
Sexual partners find each other by smell. Courtship ritual at this mollusk resembles
a little ritual competition of snakes – two mollusks raise forward part of body
and inflate it. They push each other by inflated bodies while one of partners
will start to blow off, displaying its defeat. After that winner slug couples
to it and creeps away on searches of new partner. But in case of loss in competition
it also may be fertilized by other slug.
After fertilization slug lays transparent eggs similar to fish eggs in moss.
In each clutch it is totaled up to 50 – 100 eggs which incubation lasts about
ten days. Young slugs have no warning colouring and emit very small amount of
poison. They lead secretive way of life and hide in moss. As they grow, they
pass to feeding on mycelium of poisonous species of mushrooms and gradually
start to eat their fruit bodies. Till the process of poison accumulation colouring
of growing mollusks becomes brighter and they start to lead undisguised life.
Enemies of hissing slug are grubs of lighting bugs and other predatory insects
eating young ones. Grubs of lighting bugs of advanced age can attack adult slugs.
During sudden attack slug has not time to produce enough of poisonous substances,
and insect can quietly eat it, having torn open body covers of mollusk.
Goblet-like
false rudistes (Rudistoconcha ostreoides)
Order: Rudistokonchs (Rudistoconchomorpha)
Family: False rudists (Pseudorudistidae)
Habitat: reeves of tropical and subtropical zones of Pacific Ocean.
After extinction of reef-building corals their place in ecosystem of ocean
was occupied by other animals. In tropical, and even in moderate waters of
Neocene seas and oceans reeves, rich in life, have appeared again. But they
are formed not by coelenterates. In tropical seas separate relic species of
soft corals were kept, but at reeves of Neocene they play a supporting role.
Perhaps, most important builders of Neocene reeves are bivalve mollusks. Having
entered symbiotic relations with algae representatives, they have turned to
amazing sedentary animals: their role at reeves of Neocene is comparable only
to role of corals in earlier geological epoch.
Reeves of a Neocene are made by these mollusks, covered by goblet-like shell.
In the top part such shell is closed by flat operculum. On surface of shell
there are traces of annual increase as relief ring-formed belts are visible.
According them, highest, meter tall shells reach, probably, age of centenary.
But more often such big and old shells are almost completely overgrown by various
sea inhabitants - sponges, worms and limy algae. Only uppermost, recently grown
edge of shell is still rather clean. Its natural color is black, but numerous
algae and sedentary animals will quickly hide it.
When the mollusk is disturbed with nothing, the operculum is slightly opened,
and from under it two tubes of siphons and the whole fan of flat pinkish tentacles
jut out. They spread under sun beams, as if leaves of plant. In crack between
operculum and shell light pink body of animal is appreciable. From time to
time, when small animals touch tentacles, they contract for shares of second,
and then spread again. But when larger animal shows interest to them, all tentacles
are instantly involved, and operculum densely slams.
This builder of reeves is goblet-like false rudistes, very big bivalve mollusk
with shell valves of unequal size. In Mesozoic era rudists, large sedentary
mollusks lived in sea, having had similar habit of life. But false rudistes
is not their lineal descendant: it is the close relative of oysters (Ostrea),
sedentary bivalve mollusks of Cenozoic era.
False rudists form very original reeves. Mollusks of various age form either
“walls”, or dense congestions, in which animals are distributed at regular
intervals in the big area. Mollusks frequently form dense joints on shells
of older individuals.
In connection with motionless habit of life shell, and after it the body of
false rudistes have strongly changed, having lost symmetry. The left valve
of this mollusk adheres to substratum when soaring larva settles on bottom
and finds favorable place for life. This valve is strongly curved - in it all
body of mollusk settles down. It grows up along all edge, gradually extending
and increasing in height. Right valve became flat – it serves as an operculum.
The ligament between valves is reduced, due to what shell forms the characteristic
goblet-like structure. When the larva of mollusk settles on inclined surface,
shell gradually bends and directs up – at adult mollusk it grows strictly vertically.
The body of false rudistes is approximately in the top third of shell. Gills
of animal from the right side of body (inverted upwards) are reduced, but gills
from the left side have grown up, forming the accomplished filtration system
occupying a significant part of pallial cavity. The filtering system of this
mollusk works not only due to ciliar epithelium, but also is supplied with
valves actively pumping over water through the left gill. These valves are
formed by the edge of pallium turned to tube. When the mollusk is not disturbed,
this tube juts out from shell and makes slow pulsing movements.
To be only filtrating organism is not so favorable, especially in congestions
formed by false rudists. But mollusks have developed more progressive way of
feeding due to which during evolution process they have occupied prevailing
position on reef. At these animals at the edge of pallium there is set of tentacles
(on right (top) valve they are short, on left (bottom) one much longer). In
tentacles there is a main ally of these mollusks in struggle for existence
– symbiotic red algae. Because of them feelers of false rudists have pinkish
color. Red algae favorably differ from green and brown ones in their ability
to lead photosynthesis at the greater depth because they are content basically
with beams of blue and green part of spectrum, penetrating in water especially
deeply. Therefore false rudists easily grow at the depth up to 15 meters, and
separate individuals survive even at depth of 25 meters. Algae get into the
body of young mollusk at the stage of spores (probably, some larvae may grasp
them, soaring in plankton).
False rudists are hermaphrodites. In time of maximum high inflow they simultaneously
spawn in water clouds of tiny eggs (up to 10 – 13 million ones from one adult
mollusk) and sperm liquid. Larvae spend first 10 days of life in plankton,
then settle to the surface of firm objects and turn to young mollusks. At the
first year of life up to 60 % of all larvae perish, and to the end 5 - 6-th
year of life only separate young mollusks from the generation remain. To this
moment they reach height 4 - 5 cm at diameter up to 10 cm. Further at them
active growth in height begins, and the 10-years old mollusk may reach height
up to 20 cm at same diameter.
Longevity of these mollusks is too sizeable: the majority of adult mollusks
easily live up to 100 years, and in lagoons of some islands there are even
170-years old “patriarchs”.
Limestone drill (Petroteredopsis calcareoterebra)
Order: Venerids (Venerida)
Family: Pholadids (Pholadidae)
Habitat: reeves of Indian and Pacific Ocean.
Reeves of Neocene epoch differ from Holocene ones. The basic reef-builders
of Holocene reeves were corals. But at the boundary of Holocene and Neocene
number of these coelenterates had appreciably decreased, and many species of
corals had died out. In Neocene the role of reef-building organisms was partly
undertaken with mollusks, worms and sponges. They form prolong solids of limestone,
as against porous and lacy structures of Holocene coral reef. Such feature
of Neocene reef favours to evolution of drilling forms of animals. On reeves
even sedentary octopuses drilling holes in reef body had evolved. Activity
of rock-boring organisms raises productivity of reef: many species of animals
settle in their holes.
Bivalve mollusks inclined to formation of various sedentary and inactive lifeforms
had succeeded in drilling of reef limestone. One of their species, living on
reef, is named limestone drill. It forms numerous congestions at the reef,
and area of limestone, drilled by these mollusks, turns similar to bee honeycombs.
This large bivalve mollusk has convergent similarity with so-called shipworms,
mollusks of close family Teredinidae drilled burrows in wood.
The shell of limestone drill is reduced up to two strong ridge limy plates
on the forward end of body. It does not serve any more to this animal as protection,
and is intended for limestone drilling. The body of this mollusk more than
one meter long at thickness about 2 cm is covered with thin delicate skin of
pinkish-white color. The pallium of limestone drill has grown together almost
at all extent to original “case”, and opens only on the back end of worm-like
body of mollusk. Right behind rudimentary shell on pallium there are some ring
lines of corneous knobs, with which help mollusk moves in tunnel back and ahead.
Pallium is “threaded” with longitudinal and ring muscular fiber, and also has
mobility.
Limestone drill bores tunnels, using shell. Mollusk supports by corneous outgrowths
against walls of hole, and starts to rotate shell from side to side. At the
forward end of its body there are glands secreting weak solution of hydrochloric
and acetic acids. Limestone is the carbonate easily dissolving even by weak
acids. At influence of mollusk secretions it turns to products soluble in water,
therefore limestone drill does not have problems with removal of dust. Secreting
some surplus of acids, mollusk expands tunnel, dissolving a part of limestone
on its walls. From time to time mollusk sharply contracts body some times,
throwing out from tunnel water with products of reaction and small insoluble
dust. The tunnel of limestone drill has the special form: in the top third
(bored by mollusk at young age) it has S-shaped bent, forming ell protecting
this animal against fishes with tweezers-like jaws, and against crustaceans.
Living in tunnels, this mollusk has kept a way of feeding characteristic for
the relatives. Limestone drill feeds, continuously passing water through gills.
Gills located at the forward end of body had practically lost respiration function.
They are covered with set of corneous bristles and represent the effective
filtration device. Mollusk gathers from them edible particles by mobile tentacle
– the modified rudiment of foot. It passes through itself water at speed up
to five liters at one hour. Water sucking proceeds through siphon formed by
pallium plica, and water is thrown out by sharp contraction of ring muscles
of pallium.
To compensate the inconveniences caused by transformation of gills into filtrating
device, this mollusk has false pallial “gills” representing several outgrowths
similar to tentacles growing on the internal side of pallium near to edge on
the back end of its body. In rest the mollusk juts out back end of body from
tunnel, and stretches pallial “gills” like a flower. As mollusks of this species
settle in colonies, they form a certain similarity of colony of hydroids or
original underwater “flower bed”. Limestone drill is very sensitive to movements
of water – on pallial “gills” the set of cells feeling change of pressure,
touch, and even change of light exposure is located. At the slightest attribute
of alarm mollusk retracts pallial “gills” inside of pallium and sharply contracts
body, supporting by corneous knobs against walls of hole.
Pallial “gills” also have one more function – they are covered with very thin
epithelium, through which this mollusk can acquire the organic substances dissolved
of water. It is the additional feeding source of limestone drill.
The form of hole, in which it is difficult to push straight jaws and claws
deep, protects this mollusk from large and nonflexible predators. But it does
not help to protect against flexible small predators – annelids. Against them
limestone drill uses the chemical weapon – it emits in water strongly smelling
liquid which frightens worms off. The same means helps limestone drill to compete
to sedentary worms: having settled near to them, mollusk oppresses worms with
its secretions. At the same time larvae of limestone drill willingly settle
near to adult individuals, forming colonies.
Limestone drill is hermaphrodite. At this sedentary mollusk there is external
fertilization – breeding individuals, settling beside, synchronize the development
with the help of hormones secreting in water, and simultaneously throw out
in water eggs and sperm liquid. At these mollusks self-sterility had developed,
therefore the risk of self-fertilization and the subsequent degeneration does
not exist. Each individual lays up to 30 thousand tiny eggs at once; breeding
at limestone drill repeats each lunar month.
The larva of this mollusk spends in reef plankton about one week, and then
settles down at the reef and turns to young sedentary mollusk. Having attached
to surface of reef, it starts to produce an acid and to bore a vertical tunnel.
Within one year the young mollusk grows up to 20 cm, and larger specimens are
10 years old ones.
“Palm worm” (Foliosiphon palmatus)
Order: Venerids (Venerida)
Family: Pholadids (Pholadidae)
Habitat: reeves of Indian and Pacific Ocean.
At the reeves of oceans tropical zone in Neocene epoch drilling bivalve mollusks
live. They play an important role in life of reef: boring massive of reef
limestone by firm shells, these animals promote settling of thickness of
reef and increasing
of biomass of reef inhabitants. In burrows made by them crustaceans, worms
and fishes settle. Drilling mollusks also have various vital strategies.
Limestone drill (Petroteredopsis
calcareoterebra) belongs to widely spread
species of drilling bivalves. Its long worm-like body is hided in tunnel
bored in thickness of reef, and only the back end of body is seen from outside
–
a tube of pallium with pallial “gills” jut out from it. This evolutionary
idea has received original continuation. At reeves of tropical Indo-Pacific
some
species of leaf-siphon mollusks (genus Pholiosiphon) related to limestone
drill live; they are adapted to different way of feeding.
Pallial “gills” of limestone drill have a permeable epithelium, through which
this mollusk absorbs the organic substances contained in water. Leaf-siphon
mollusks also absorb organic and besides of it also mineral substances through
epithelium of pallial “gills”. But these substances intend not to mollusk,
but to numerous “lodgers” in its tissues – to unicellular algae. In coexistence
with them there is the secret of well-being of this group of mollusks. Mollusk
receives oxygen and feeding from microalgae, giving them carbonic gas, mineral
substances and protection against enemies.
The typical representative of leaf-siphon mollusks is “palm worm”. Its name
indicates two features of shape of this animal.
The body of “palm worm” is lengthened and flexible, covered by tube-like
pallium. This species is very large representative of bivalve mollusks: its
length reaches
200 cm at thickness of about 4 cm. This mollusk drills burrows in thickness
of limestone, using the rudimentary bi-valved shell similar to shell of limestone
drill. Its tunnels differ in spiral form – only young mollusk bores vertical
shaft, and further it is bent, making the animal inaccessible to many predators.
The second feature of this mollusk distinguishing it from limestone drill
is the shape of its pallial “gills”. They are very big (up to 15 cm long
being
straightened) and have a plumose structure. Mollusk has 4 – 6 pallial “gills”
and consequently the back end of its body jut out from hole resembles small
palm tree, and the colony of these mollusks is similar to palm grove. In
tissues of pallial “gills” there is a plenty of symbiotic green microalgae,
which give
to them green colouring. In case of danger mollusk quickly retracts pallial
“gills” inside of pallium, and hides in hole. Each leaf of pallial “gills”
has a longitudinal muscular fiber contracting in case of danger and shorting
“gill”.
On the forward end of body of “palm worm” there are some ring lines of peaked
knobs, with which animal supports against walls of hole. Shell of “palm worm”
is reduced, thick and ridge. It is transformed into drilling tool like at
limestone drill. This mollusk produces smaller amount of acid at tunnel drilling,
using
mainly the movements of shell.
On reeves one more species of leaf-siphon mollusks, “sea rose” (Foliosiphon
rosa-marina), lives. Its pallial “gills” are wider and have rounded
shape with longitudinal plicas. Because of it the back part of body of “sea
rose”
looks
like a flower – it has determined the name of this species. This species
has much smaller size, than “palm worm”: body of “sea rose” is about 60 cm
long
at thickness of no more than 2 cm.
Both species of these mollusks settle at well lighted shallow sites of reeves.
But “sea rose” lives at sites which may dry at outflow, and “palm worm” prefers
deeper places. During outflow “sea rose” is hidden in hole and sticks an
entrance by mucous fuse.
The breeding biology at these species is similar to those at limestone drill.
Perseus's
octopus (Perseoctopus medusophorus)
Order: Octopuses (Octopoda)
Family: True octopuses (Octopodidae)
Habitat: Pacific Ocean, top water layers.

Picture by Timothy Morris, colorization by Biolog
Initial picture by Timothy Morris |
The open ocean is very specific environment. Top layers of
water are inhabited by huge amount of plankton organisms soaring in thickness
of water and are constantly carrying by current being not able to resist to
it. Here constantly severe struggle for existence proceeds, in which various
live organisms use the most refined ways of survival. After mass extinction
of plankton organisms at the boundary of Holocene and Neocene the new plankton
community was generated by descendants of coastal and ground animals survived
in ecosystems poorly affected by extinction. Among new plankton organisms there
are various worms, crustaceans and swimming gastropods. Characteristic representatives
of Neocene plankton are cephalopods of various species. Descendants of coastal
species had passed to existence in thickness of water even in Holocene, and
mass extinction of plankton animals had increased their chances of success.
Very favourable form of mutual relation in various communities is the union
of two species of organisms – mutualism (mutually advantageous), commensalism
(with unilateral benefit) or parasitism (causing any damage to one participant
of the community). One species of cephalopods had solved set of problems having
concluded the favourable union with jellyfishes – one of the most dangerous
plankton inhabitants. This transparent creature about 10 cm long with thin one
meter long tentacles lives in congestions of jellyfishes and is named Perseus's
octopus to commemorate the mythical hero being victorious over monstrous gorgone
Medusa.
The Perseus's octopus is a pelagic cephalopod. It spends all life in thickness
of water, and is not connected to the oceanic bottom in any period of life.
This animal almost nothing differs externally from other octopuses. The body
of Perseus’s octopus is transparent like bodies of the majority of cephalopods
living in plankton. When the octopus is quiet, only its big eyes partly disguised
by brilliant film on external surface of eyeball are visible only. On skin of
the octopus on closer examination a plenty of tiny black points is visible:
it is special iridocyte cells containing light-reflecting substances. Opening
them, animal becomes shining: it is a protective reaction (alternation of shine
and transparency distracts predator, preventing it to concentrate at the octopus
searching). The disturbed octopus represents very interesting show – it opens
iridocytes till one second, brightly flashing in sunlight, and then instantly
retracts them, becoming translucent and poorly appreciable among jellyfishes
swimming around.
The Perseus's octopus protects itself and obtains food, constantly holding in
tentacles two jellyfishes. Besides it has immunity to poison of jellyfishes
and other coelenterates, therefore it can freely swim among jellyfishes, impudently
robbing their tentacles. Besides slime of Perseus’s octopus has inhibiting properties
– it interrupts the reaction of stinging cells of jellyfish tentacles. At Perseus’s
octopus immunity to poison of jellyfish of certain species using at present
is developed. But being compelled to coexist the jellyfish of other species,
the mollusk receives from it slight stings first time. But it just is necessary,
because at this moment the mollusk develops the immunity and accumulates the
inhibitor to new “pet”. Tentacles of Perseus’s octopus have some sensitive suckers
in the middle part which are not damaging delicate body of the jellyfish kept
by the octopus. If the octopus is attacked by predatory fish, it tries to stick
to it into the head tentacles of jellyfish kept by it. It happens, that mollusk
even kills an attacking animal this way. In this case it uses an opportunity
and simply eats it, and jellyfishes get shreds of meat floating in water.
The male of Perseus’s octopus is smaller by size, rather than the female. It
prefers to court to it “keeping the distance”, being afraid of tentacles of
jellyfishes kept by it. The male arranges “light show” for the female, during
which on its tentacles and body shining waves glance. For this purpose animal
emerges holding jellyfishes to the surface of water where it is shined better
with sunlight. If the female reacts positively to courts of the male, one of
his tentacles, filled with sperm liquid (it is called “hectocotylus”) is torn
off and searches the entrance to pallial cavity of the female itself. Swimming
hectocotylus continues courtship “show”: on it iridocytes open by waves, that’s
why it seems flickering. After the courtship ritual male can grow the lost feeler,
and during life it repeats courtship ritual up to five times with different
females.
The female of Perseus’s octopus lays some hundreds of small eggs. She bears
them on turned inside abdominal pair of tentacles within three months. Till
this time the female eats of nothing. It sits on the bell of large jellyfish
from above, having clasped it by all free tentacles, and having hidden the clutch
under the body. If there is lack of suitable jellyfish, the female of Perseus’s
octopus covers eggs with the top pair of tentacles and holds two smaller jellyfishes
by lateral tentacles, choosing for self-defense the most poisonous species.
From eggs young octopuses hatch; they are similar to adult individual, but have
short tentacles and are lack of iridocytes. They begin the life, searching for
jellyfishes of less poisonous species and gradually developing immunity. Their
immune system is rather weak; therefore a sting of strongly poisonous jellyfish
may kill them. But till the process of growth young animals get immunity and
develop life even on the most poisonous jellyfishes of the ocean.
Similarly to all octopuses, Perseus's octopus grows quickly, and reaches the
limiting size at the second year of life.
Balloon
octopus (Sphaeroctopus inflatus)
Order: Octopuses (Octopoda)
Family: True octopuses (Octopodidae)
Habitat: reeves of Caribbean Sea.

Picture by Timothy Morris, colorization by Alexander Smyslov
Initial image by Timothy Morris |
The rich fauna of shallow waters of Caribbean Sea had strongly
suffered during the global ecological crisis at the boundary of Holocene and
Neocene. Reef-building corals had especially big damage – they had practically
completely become extinct. The plenty of species of sea animals connected in
daily life with corals had died out in common with them. Subsequently in early
Neocene on sea shallows new ecosystems had started to develop – heat and abundance
of sunlight favored to their prosperity. These communities are as rich in life,
as coral reeves of Holocene were; here also there is strict competition and
there is a set of predators of various trophic levels.
In similar conditions at various animals various ways of protection develop.
But also it often happens in nature, that the same “technical decision” is embodied
in different groups of live creatures. The most popular tactics of protection
is to exaggerate the own size. Various plicas of skin and outgrowths unwrapped
aside the contender, and also limbs stretched in sides are different ways of
the decision of such problem. One more popular way to exaggerate is to be inflated.
Such tactics is used by different vertebrates: amphibians, reptiles and fishes.
In Neocene among such deceivers the octopus had appeared.
The balloon octopus living on Caribbean reeves is small species of cephalopods.
Length of its body is about 10 cm, and tentacles reach the length of half meter.
This species is colored rather brightly: the basic background of skin is light
yellow; on it brick-red spots with silvery border are scattered. Behind eyes
there are two larger spots. The annoyed octopus can turn almost black for a
split of second. But colouring is not main feature of this mollusk.
When the predator comes nearer, balloon octopus does not hide. It is able to
repulse predators: in the mouth of this mollusk there are poisonous glands secreting
deadly poison. One bite of balloon octopus can kill fish larger than one meter
long for some seconds. But poison is not fighting, but the hunting weapon, therefore
octopus tries to avoid near fight with predator, and uses poison only in case
of extreme danger, when there is no another way to escape. Warning the enemy
about the protection, octopus puffs body, exaggerating the own size, and colors
skin brightly, becoming appreciable from apart. Being disturbed, it emits in
water poisonous ink which paralyze olfactory nerves of fish attacking it for
some time. After such protection of octopus fish remembers its colouring, and
does not attack these mollusks any more.
It is simple to the octopus to use such way of demonstration – its anatomy favors
to ability to change the body shape any way. Octopuses do not have internal
skeleton; and soft tissues are easily extendable. Pallial cavity of balloon
octopus has strong ring muscle on the edge, “tightening” it when the animal
is inflated; and edges of pallium are covered with spongy tissue skintight to
body. Swimming funnel can work as the delivery pompe – it pumps up water in
pallial cavity by pulsing movements. The disturbed animal quickly gathers water
in pallial cavity, and its visible size increases some times: the body is inflated
up to half meter diameter sphere. Colouring of animal fades, and on skin behind
eyes the special pattern appears: two big black oculate spots, frightening off
predator. Inflated animal keeps tentacles pressed to each other like the “beak”
of huge bird's head.
Such demonstration very effectively affects to medium-sized predators: fishes
are frightened of suddenly appeared in front of them “live head”, and swim out.
But balloon octopus can stay not for long in such position: when pallial cavity
is closed, water in it is not freshened, and animal simply begins to choke.
mollusk compensates this circumstance partly by skin respiring, but if the demonstration
is delayed, octopus prefers to seek safety in flight: it sharply “exhales” water
from pallial cavity, and uses the ensued strong jet push to hide in the nearest
shelter. At this moment it sharply changes well appreciable colouring to pale
grey one and as if “is dissolved” on the background of water. It also gives
to it precious seconds to escape from predator.
Similarly to all cephalopods, balloon octopus is a predator. It eats small fishes
and crustaceans, getting them from cracks and holes by long tentacles, or catching
by throws from ambush.
Balloon octopus breeds once a life – it is the characteristic feature of octopuses.
Pairing of these mollusks is preceded with original courtship displays. During
courtship ritual the male is inflated and shows to the female various spotty
patterns on the body, simultaneously touching her by tips of tentacles. If near
the female two males meet, their courtship becomes much more active and more
colorful. They are inflated, and, holding by tips of tentacles for reef, try
to push away each other from the female by body. Competing males try to inflate
as much, as possible, and to keep in such condition longer, than the contender.
The male “blew off” before the competitor, loses. It leaves the place of courtship
display, having replaced bright courtship dress to cryptic coloration. And the
winner starts to make court. It cautiously moves nearer to the female, and enters
in her pallial cavity the tentacle carrying spermatophors. At the culmination
moment muscles in the basis of tentacle tear from sharp contraction, and it
stays in pallial cavity of the female. If the female was already courted by
other male, new partner tries to pull out the tentacle left by the previous
male. After pairing the lost feeler quickly grows. The male is able to repeat
courtship ritual two – three times per life, and then perishes.
When the female starts to lay eggs, tentacle of the male in her pallial cavity
bursts from contractions of the special muscles, and eggs are fertilized. The
female lays up to 200 thousands of small eggs. She places them in shelter –
deep crack or large bivalve shell. The incubating lasts about two months. From
eggs small larvae differing from adult mollusk by short tentacles and pointed
translucent body hatch. After their hatching the organism of the female quickly
degrades, and she dies.
Young animals grow very quickly, and already at one-year-old age reach the size
of adult mollusk. Males become able to breeding at the age of 16 – 18 months,
and females up to 20 months.
“Crystal
demon” (Ophthalmoctopus crystallinus)
Order: Octopuses (Octopoda)
Family: True octopuses (Octopodidae)
Habitat: Sea of Okhotsk, the upper water layers to the depths of 200 – 300 meters.

Picture by Timothy Morris, colorization by Biolog
Initial image by Timothy Morris |
In Neocene the Sea of Okhotsk became a semi-enclosed body of
water. The volcanic activity in the region of the “Pacific Ocean’s ring of fire”
resulted in transformation of Kurile Islands into Big Kuriles – a chain of large
islands, separated only by narrow straits, through which the Sea of Okhotsk
is connected to Pacific Ocean. On the surface of the Sea of Okhotsk a unique
biocenosis was formed by various brown algae, and it resembles the natural community
of the Sargasso Sea from the human epoch. The rafts of interwoven algae represent
the living place for many small animals – fishes, crustaceans, molluscs and
worms. The abundance of small animals attracts predators to the floating algae.
Some of them climb deeply into the depths of the rafts; others hunt on the edges
of algae congestions, and the third ones catch animals, which accidentally appeared
in the water far away from their shelters.
Among the predators of algal fields of Sea of Okhotsk there are many species
of octopi. One of their species passed to untypical way of life – it barely
descends to the substratum, and prefers to keep in thickness of water at the
edges of the algal congestions. Its transparent body and carnivorous way of
life has determined its name – the “crystal demon”. This animal is rather common
in the algal fields, although it is difficult to be observed.
The “crystal demon” is swimming octopus species. This is a definite descendant
of pelagic species settled in the fodder-rich environment – in the outskirts
of rafts of brown algae.
The body of this mollusc is about 10 cm long, flat and wide, resembling tree
leaf in shape. Almost all of the internal organs of the animal are semi-transparent,
except for its eyes, cartilaginous “skull” and ink bag. However, these body
parts of mollusc are disguised by the thin layer of tissues that contain guanine
crystals. This gives them a silvery shine and makes it possible to reflect the
surrounding objects, making animal almost indistinguishable in water. The mantle
adheres to the abdominal side of body along the central line, separating the
mantle cavity into two “pockets” along the sides of body. Strong circular muscles
are developed in their walls. The funnel is reduced and serves only to pull
water into the “pockets”. “Crystal demon” swims due to powerful contractions
of circular muscles of the “pockets” of mantle cavity. Lack of the internal
skeleton gives a problem to soft-bodied animals: their bodies take their initial
form only due to the elasticity of the tissues. At the “crystal demon”, “the
pockets” of mantle cavity are supported in their straightened state by the incomplete
rings of cartilage-like tissue that developed in their walls. This makes it
possible to fill them rapidly with water before making the reactive push.
The body of “crystal demon” is semi-transparent and barely noticeable in sea
water. The eyes of the animal are large, luminous and very mobile: they can
be turned sideways, upwards and downwards. The head of “crystal demon” is thickened,
and the eyes are located at its edges. The tentacles of this mollusc are about
15 cm long, but if necessary they can stretch out to double that, when the mollusc
attacks its prey that is hidden among the algae.
The “crystal demon” possesses a surprising method to lure prey. This mollusc
can imitate a school of small swimming animals: it demonstrates “running” spots
of small size – dark or silvery in color – against transparent background. Some
pigment cells in the external tissues of the mollusc accumulate guanine, and
it helps animal to imitate silvery fishes. Showing on its skin a simulacrum
of school of small animals, this octopus can easily swim closer to an unsuspecting
shrimp, which observes its “show”, as if being hypnotized. In the same way this
mollusc catches small fish as well.
This species of cephalopods is monocyclic: all of the adults die after breeding.
The male of “crystal demon” is considerably smaller, rather than the female.
It fertilizes the female with the aid of the special tentacle – the hectocotyle.
The female bears the clutch of several tens of thousands transparent eggs on
its tentacles for three months. At this time it leaves the algal fields and
drifts in the water depth, where there is smaller amount of predators. During
the time of clutch bearing the body of the female is completely transparent,
and predator can discover it only by accident. The lifespan of this octopus
reaches five years.
Translated by Bhut, the forum member.
Monkey
octopus (Platytentaculum acrobaticus)
Order: Octopuses (Octopoda)
Family: True octopuses (Octopodidae)
Habitat: Sea of Okhotsk, fields of floating algae.

Picture by Timothy Morris, colorization by Alexander Smyslov
Initial image by Timothy Morris |
Among the predators of the algal fields of the Sea of Okhotsk,
cephalopods occupy an important place. These inhabitants of the sea bottom had
found far from the coasts an environment completely suitable for life and representing
interwoven brown algae, where it is easy to find shelter from the enemies and
suitable prey like numerous small animals settling among the algae. The only
limitation for cephalopods at the settling at the algal congestions is connected
to the body size: the coastal giants cannot live among the algae. However the
small octopi, populating the algal fields of Sea of Okhotsk, are very diverse.
The absence of hard skeleton makes possible for them to squeeze into the narrowest
slots, and in the thick interlacements of algae they feel themselves like a
duck to water, creeping among the algae with the aid of their tentacles.
One of the small octopi populating the algal fields of Sea of Okhotsk is the
monkey octopus, a mollusc having body length of about 6 cm. The body of monkey
octopus is flattened into an oblate shape. The tentacles are about 20 cm long,
wide and flattened, ribbon-like with rounded tips. Their suckers are arranged
in two rows. At this octopus the suckers have significant mobility and they
can make “steps” like movements of the caterpillars. With the aid of the suckers’
movements monkey octopus can slowly creep up to its prey, without giving itself
away by superfluous motions. When the monkey octopus does not hunt, it moves
among the algae, grasping them by its tentacles as monkey does with its paws.
The adult mollusc of this specie has lost the ability to swim – being out of
the algae thickets monkey octopus feels itself very insecure. Having found itself
in open water, this mollusc attempts to keep itself afloat, waving its tentacles.
Simultaneously it stretches out its tentacles in the direction of the nearest
strands of algae, attempting to find support. The funnel of this mollusc is
underdeveloped, and the mantle cavity is considerably smaller than of the octopi
leading a mobile way of life and able to swim.
Skin respiration plays an essential role in the respiratory metabolism of monkey
octopus. At the upper side and along the sides body of this mollusc is covered
with the leaf-like outgrowths, which simultaneously perform the function of
“additional gills” and provide camouflage. When resting, the octopus stretches
them out, increasing the surface of its skin, but in case of danger they contract
strongly and take the shape of small tubercles on the mollusc’s skin.
The body of monkey octopus is coloured in brown with greenish specks and orange
spots above the eyes and along the sides of body. The lower side of the tentacles
is white. Depending on its emotional and physical condition, this mollusc can
change colour from white to dark red, orange, green or brown with various switches
between aforementioned colors and their combinations. The eyes of this mollusc
are large and mobile, can rise above the body. Above each eye there are several
branching outgrowths of skin that disguise the eye. The sight of monkey octopus
has an important special feature – it notes the slightest motion in the motionless
environment. It helps the octopus to find prey among the algae, where visibility
is very poor. Also the monkey octopus has very keen sense of smell. The bite
of monkey octopus is poisonous for the crustaceans and fishes – the mollusc
feeds on them predominantly.
This mollusc breeds once a life. The male avoids direct encounters with the
female – at this species cannibalism is very common, and the male (it is smaller,
than female) has a chance to fall prey of the female. Having found a female,
the male approaches it from a safe distance and with the sharp reduction of
muscles breaks off one of its tentacles – the hectocotylus, filled with sperm.
This tentacle finds the female by itself via smell and creeps into her mantle
cavity, where the fertilization of eggs takes place. After the detaching of
hectocotylus male remains alive. It grows new hectocotylus and can fertilize
the next female. At this time the male does not eat, and perishes after the
loss of second hectocotylus.
The clutch of this species amounts 40 – 50 thousand of small eggs; female bears
them on the front pair of tentacles. When bearing the clutch, the female builds
for itself a kind of a nest, bending the strands of algae to speric construction,
in middle of which it hides, keeping the “walls” of its shelter by tentacles.
The incubation lasts for three months, and all this time the female eats of
nothing. After the hatching of larvae it dies soon.
The young monkey octopi can swim and settle to great distances before they grow
up and pass to settled way of life. The sexual maturity of this species begins
at the third year of life.
Translated by Bhut, the forum member.
Sun-shaped
octopus (Soleoctopus platyssimus)
Order: Octopuses (Octopoda)
Family: True octopuses (Octopodidae)
Habitat: tropical waters of Central America – Caribbean Sea, Pacific and Atlantic
coasts of North and South America.
The “plankton catastrophe” on the boundary of the Holocene and Neocene had a
severe impact to marine ecosystems. In the era of restoration of biological
diversity squids made a number of successful attempts to master pelagic habitats,
and their relatives, octopuses and cuttlefish, have continued to evolve in the
coastal waters, and among them a number of bizarre forms appeared. One species
of such mollusks is the sun-shaped octopus from tropical waters of the New World.
This species is a flat crawling octopus dwelling in marine shallows. Its tentacles
are enclosed in a thick elastic membrane almost to their tips and have limited
mobility. The tips of the tentacles, however, retain sufficient mobility, and
with their help the mollusk can crawl along the seabed. Also well-developed
suckers participate in crawling; they are located in double rows along each
tentacle. The body of this mollusk is strongly flattened; its contours are outlined
with the semicircular fold of its mantle, which extends some distance behind
its eyes. The eyes are movable, bulging, with a horizontally slit pupil; the
iris is golden brown.
Being adapted to a creeping lifestyle, the sun-shaped octopus has practically
lost the ability of reactive movement. Only occasionally a scared octopus may
emit a stream of water, rising from the seabed. It is able to swim in strokes
using tentacles, resembling at this point a tailless ray or a large flatworm.
The coloring of the sun-shaped octopus is very changeable: from almost white
to green and cherry red. The animal has good vision, allowing it to simulate
easily the color of the surrounding area. At rest, this species has a brown
color with rows of white strokes, radiating from the body on the upper side
of the tentacles. When agitated the coloring becomes sharper, and in the webbing
between tentacles additional spots appear, merging with the main pattern.
The sun-shaped octopus, despite leading a fairly sedentary lifestyle, differs
by aggressive behavior. Inverted or washed out from the sea bottom by a jet
of water, the octopus tries to cling to its enemy and to bite it. Its poisonous
bite is excellent protection: a shark with a slow metabolism feels weak for
several days, and warm-blooded animals can die from the poison in a few minutes.
The venom of this mollusk, however, is a hunting weapon: the animal eats benthic
invertebrates with slow metabolism. The crushing jaws of the octopus can cause
strong bites and are able to stretch from its mouth on movable ligaments when
the octopus examines the reef in search of burrowing animals – crustaceans,
worms and mollusks. This species eats starfishes as well, including poisonous
species.
As at all octopi, ink at the sun-shaped octopus is poisonous. But in the method
of its application, this species differs from its relatives: releasing ink into
the water, it does not leave its cloud, and hides, clinging to the bottom, its
eyes closed and mantle cavity locked tightly. At the same time, on the body
of octopus a mucus layer is released that protects it from its own poison. If
necessary, sun-shaped octopus can make up to three “shots” of ink.
The passive way of life at this species is combined with a high capacity for
regeneration: the animal can easily grow back all its tentacles, if lost them
simultaneously. If this octopus damages its eye, it is easily restored within
a few weeks if even a small part of it remains.
This species grows rapidly and reaches sexual maturity at the age of two years.
The male is smaller than the female, and differs from her by elongated tentacles.
In the process of “dating” the male gently draws its tentacles and strokes the
female’s tentacle. Displaying itself, it swims circle-wise above the female,
and then sits down on top of her body and puts a spermatophore into her mantle
cavity.
The female lays up to 1000 small eggs and harbors them on the underside of the
body, having pasted them between the suction cups. Usually she selects as a
shelter a narrow crack in the reef and ventilates it with her undulating movements
of body and tentacles. Incubation lasts about a month. Juveniles immediately
leave their mother who does not eat after posterity hatching and dies within
a month.
Translated by FanboyPhilosopher.
Hermit
octopus (Diogenopus vermisomus)
Order: Octopuses (Octopodida)
Family: Porter octopuses (Phoroctopidae)
Habitat: mollusk-spongial reeves of Pacific Ocean.

Picture by Timothy Morris
Initial image by Timothy Morris |
Octopuses are cephalopods completely lost their shell: even
a rudiment of this structure is not remained at them. Therefore they can not
rely upon passive protection as it was made by their relatives – ammonites and
nautiloids. Feature of behavior of octopuses is their disposition to hide into
different kinds of shelters. And absence of firm parts of body gives them big
advantages in this sense: octopus can hide in very narrow shelter, where predator
having a firm skeleton, will not squeeze through. But shelter is not always
“near at hand”: the mollusk should leave it to hunt. In Neocene some octopuses
have solved this problem by very simple way: they simply began carry shelter
on themselves. The most simple and accessible among small and enough strong
shelters on Neocene reeves are mollusk shells. During evolution process the
special family of octopuses, which have mastered using these shells for daily
life similarly to hermit crabs, has appeared.
The characteristic representative of this family is the hermit octopus. It is
medium-sized mollusk: its tentacles are up to half meter long (measurements
of the female are described here, male is much smaller). At usual octopuses
leading free life there is short and thick body, but at this mollusk body is
long and flexible. Hermit octopus practically constantly drags on itself found
mollusk shells in which it hides in case of danger, therefore such body shape
permits it to use volume of portable shelter most rationally. In connection
with such unusual habit of life its tentacles have a little changed: from eight
tentacles two ones (last pair) became short, flat and wide, and their bases
have moved back and upwards. These tentacles are intended for keeping of shell
on the body of mollusk, therefore getting it out from the shelter is rather
uneasy: for keeping at smooth internal surface of shell on these tentacles suckers
are well advanced. Other tentacles have a structure typical for octopuses, and
are used the same way as usually. The hermit octopus has kept ability to change
colouring of body, but its body nevertheless makes it less successfully, than
head and tentacles. In rest colouring of tentacles is grey with pinkish and
white “marble” pattern of cross strips and spots, but it also can change from
white up to rubiginous and black color. Eyes are large, greenish with white
rings around, exaggerating visually their size.
At all species of hermit octopuses both males and females search and carry shells.
This kind of octopuses is named after the philosopher of human epoch Diogenes,
lived, as the legend says, in barrel. Such name rather precisely indicates features
of behavior of this octopus: characteristic for these cephalopods aspiration
to occupy various shelter has resulted to that hermit octopuses have originally
returned to feature which their far ancestors have refused millions years ago.
However, as against fossil cephalopods, carrying shell till all their life,
the hermit octopus can leave it at the will not for a long time. It makes it
very seldom, and only when there is no other way to reach any significant purpose.
However it does not leave from the house far: by one tentacle the hermit octopus
constantly keeps for shell, being ready at any moment to involve itself in shelter
by sharp contraction of this tentacle.
Because of heavy shell and thin long body the hermit octopus has lost the ability
to swim. Usually it creeps on the bottom, extending forward the forward pair
of tentacles and tighten on them. It does not have need to swim quickly: its
food in case of danger does not aspire to rescue by flight. Similarly to all
cephalopod, this octopus is a predator. It eats crustaceans and fishes dug at
danger in sand, or hiding in holes. Usually such octopuses one by one wander
on reef, tracing for smells dissolving in water. Having felt smell of possible
catch, octopus slowly moves to its source. It does not try to disappear: it
has no need to creep as closer, as it is possible, and then to rush and to chase
prey; it hunts by other way.
Having noticed a predator, any fishes and shrimps swim out, and any ones hide
themselves in sand. This mollusk also hunts for them. It immerses tentacles
in sand, and sensitive chemoreceptors on their surface permit this mollusk to
define, where there is a catch. Though the mollusk does not see prey, it chases
it in thickness of sand by tentacle by smell. If the catch dug in sand will
try to slip away, electroreceptors catching activity of nervous system of hidden
catch include to hunting.
Strong suckers at tips of tentacles help to keep seized catch. When the chased
animal has got, octopus jerky contracts tentacle and pulls catch out from sand.
Even if at this moment it will escape, it will not be possible to rescue to
it: octopus covers a place of hunting by shell in which it is sitting, and catch
slipped away all the same appears in trap.
Octopus kills caught prey by sting which is very poisonous and instantly kills
even animals with slow metabolism. With the help of stings octopus protects
itself from enemies – predatory fishes and other species of octopuses. For protection
another traditional for cephalopod kind of weapon, poisonous ink, is also used.
But it is the extreme case: ink is poisonous for the octopus, and it can not
leave quickly the poisoned zone. Therefore it splashes ink through swimming
funnel extending like tube, and at once presses shell to bottom. If the bottom
is sandy, octopus even “anchors”, deeply immersing free tentacles in sand and
being dug in it by edges of shell.
For breeding male finds female by smell emitting by it. Having found shell of
the female, male cautiously gets on it, and waits, while the female will find
out his presence herself. If he will be careless, the female may easily have
eaten him. When the male will be convinced, that the female knows about his
presence, he cautiously starts closer acquaintance: he warns her about his intentions
by easy touches to tentacles of the female. The female unready to pairing simply
banishes the unlucky “groom” by impact of tentacle. If the female is ready to
pairing, she lets tentacles out and touches by them the male, answering him.
An accustoming of animals to each other proceeds so. For pairing the male pushes
tentacle filled with sperm liquid (it is larger than others) in pallial cavity
of the female, and then by sharp contraction of muscles in the basis of this
tentacle tears it off. His mission in breeding is executed, and the male leaves
out. The torn off tentacle will regenerate, and he one – two times again can
repeat courtship. His tentacle some time lives in pallial cavity of the female.
When she starts to lay eggs, this tentacle bursts and fertilizes them.
The female of this species carries eggs pasted in vault of shell like grapes.
As all octopuses, during protection of posterity she becomes inactive and eats
of nothing. Development of eggs lasts about one month, and then from them tiny
translucent youngs hatch. First some days of life they spend in shell near to
mother. They hang on wall of shell almost motionlessly. At this time at them
large yolk sac resolves, and body gradually starts to get colouring. After the
young growth abandons shell, female lives not for long: about one week. In her
organism irreversible physiological changes take place, and she perishes.
Leaving from parental shell, young hermit octopuses first two months live usual
life, characteristic for the majority of octopuses. Later their body starts
to extend, and they look for a shell to themselves.
Hermit octopuses are short-lived: duration of their life does not exceed three
years.
Close species:
Flat-bodied
hermit octopus (Diogenopus platysomus). It is small species: the length
of tentacles does not exceed 10 cm. At this mollusk body is strongly flattened:
it lives in shells of mollusks with narrow chink-like fauces. All body of flat-bodied
hermit octopus is translucent; covers have pattern coloration of grey spots,
and eyes are silvery. This species eats mainly small crustaceans and sedentary
worms. Due to strong beak-looking jaws it can break open sideways limy tubes
of sedentary worms and shells of small snails.
Garbaging
octopus (D. ostracophylus) is the most low-specialized species of genus:
the mollusk uses valves of shells of bivalve mollusks as shelters. In case of
danger this octopus presses shelter to firm substratum and sticks by all tentacles;
therefore it avoids sites with soft sandy bottom and keeps basically on rocky
and calcareous sites of reeves. This species differs in small sizes (length
of tentacles is about 8 cm) and short body. Covers are colored rather brightly:
they are yellow-colored with small black spots, and the body is colored as brightly,
as tentacles are. It eats different small animals, and also willingly eats rests
of catch of large predators.
Polyp
octopus (Polypoctopus sessilis)
Order: Octopuses (Octopodida)
Family Sedentary octopuses (Sessiloctopidae)
Habitat: coastal waters of Indian and Pacific oceans, oyster reeves.
When coral reeves have disappeared, new productive natural community, reeves
constructed by bivalve mollusks, basically by stone oysters and various species
of tridacnas, has appeared to replace them. Entering commonwealth with algae
they have constructed an ecosystem populated almost as is rich and various,
as coral reeves. Cephalopods in any epoch were characteristic inhabitants of
such coastal communities. Mastering different ecological niches, they sometimes
have fantastical and unexpected shape.
In Neocene among cephalopods living at algal-mollusk reeves completely unexpected
creatures have appeared: octopuses replaced active life of free hunters to
life of passive homebodies. And they became so adhered to the house, that seldom
leave it at own will, spending at times the most part of life sitting at same
place.
The polyp octopus, one of such “homebodies”, is medium-sized species of octopuses
with sharp sexual dimorphism. The female is about 10 cm long, with very long
tentacles which can extend up to 1 meter length. She constantly sits in once
chosen shelter: empty shell or crack in rocks. In connection with such life
she had strong changes in appearance. The body of such female grows, filling
by itself the chosen shelter and accepting at times the most fantastical and
wrong form. Sense organs also have undergone changes: sight at the female of
polyp octopus is very weak, eyes are substantially reduced. But instead of
sight at her the sharp chemical feeling sense was developed, permitting to
receive the exact information about world around.
The body of female of polyp octopus is colored light pink, skin on it is translucent,
and interiors of animal appear through it. Tentacles are colored brown with
greenish and white irregular-shaped spots. However, their colouring can easily
vary.
On the back side of body skin sucker, assisting to keep in shelter, develops.
Similarly to all representatives of cephalopod classus, the polyp octopus is
a predator. The female of this species arranges ambushes, having stretched
on surface of reef tentacles with well advanced suckers. Having felt presence
of catch, mollusk by sharp movement of tentacles seizes it and draws to mouth.
For catch killing the polyp octopus uses poisonous sting. Jaws can be pushed
forward, putting sting to the seized animal. The basic catch of females of
polyp octopus is small and medium-sized fish.
The male of this species is radically differing from the female by appearance
and habit of life. It is very small (length is about 6 - 7 cm with tentacles),
leads active and mobile life. On his body there are muscled fins with which
help the mollusk can swim very quickly. At him eyes and organs of chemical
feeling are well advanced: all sense of his life is finding of females for
the sake of breeding. The skin of the male is colored red - brown, but it can
change shade from grayish-white up to spotty and black. After approach of sexual
maturity (at the age of half-year) the male actively searches and fertilizes
females. But the male lives not for long time: about nine or ten months.
For fertilization of the female the male falls on bottom outside of her tentacles
reach zone. One of tentacles of the male tears off the body by the sharp muscular
contraction, and independently creeps to the female. To come nearer to her
for fulfillment of courtship ritual is unsafe for the tiny male: she easily
can take him for catch and to have eaten. But independently creeping tentacle
does not cause in her reaction of attack, and easily reaches oviducts of the
female, attaching there to wall and at the moment of egg laying emitting liquid
sperm. At the male new tentacle grows soon, and he searches for the next female
which may be fertilized. Sometimes in oviduct of one female some tentacles
of different males can wait for “hour of triumph”.
The female pastes clutch of large translucent eggs (about 200 ones) looking
like cluster of grapes in shelter, and then diligently protects the future
posterity within one month, eating of nothing. After larvae hatching (actively
swimming transparent larvae are externally similar to the male) she lives not
for long and soon perishes. Life expectancy at females of this species does
not exceed two years.
The young growth first two months leads active life; later females look for
themselves any shelter and turn to inactive sedentary creatures. Males continue
swimming and hunting, but their growth is sharply slowed down, and they remain
dwarfs to all stayed life.
Worm-like
octopus (Vermoctopus serpuloides)
Order: Octopuses (Octopodida)
Family: Sedentary octopuses (Sessiloctopidae)
Habitat: coastal waters of Pacific Ocean, spongial and mollusk reeves.

Picture by Alexey Tatarinov
This species belong besides to family, as previous one, but
its specialization to inactive habit of life has gone any other way: this mollusk
arranges holes
in calcareous basis of reef. As the adaptation to such way of life the large
gland producing acid secret was developed at the back end of body. Because
limestone is easily dissolved even by the weakest acids, its drilling does
not cause any difficulties.
At this species the sexual dimorphism is less expressed, than at previous
one: male is only a little smaller, than female. Body of mollusks of both
sexes
is worm-like: its length at the female is about 30 cm (male is about 20 cm
long) at thickness of body only 3 - 4 cm. Tentacles are strongly extendible
(in quiet condition their length is about 15 cm, extended ones can reach
length of 60 - 70 cm), suckers are only on their tips. Skin of worm-like
octopuses
is grey with small black speckles on tentacles. On the back end of body,
near the acid gland, there is a large sucker assisting to animal to keep
in hole.
Eyes of these mollusks are rather well advanced: animals can see colors and
small moving objects, but they are too near-sighted: at the distance of two
meters the large fish turns for their sight to blurred shadow. But well advanced
chemical feeling permits them to perceive presence of the enemy and to hide
in hole before it will get in their field of vision.
For simplification of breath the respiratory siphon of these mollusks became
long and extensible. Its end is covered with set of receptors detecting movement
of water and presence of chemical substances in it. Even when whole mollusk
sits in hole, the siphon serves to it as though as “window in the world”,
and permits to keep under the control conditions around of hole.
The worm-like octopus lives, as the sedentary tube worm (Serpula): it juts
tentacles out from hole and waits, while any small animal will come nearer
to the distance from which it may be seized easily. In case of danger this
mollusk instantly disappears in hole and closes an entrance by “fuse” (stone
or massive shell) which it constantly holds in one of tentacles. If the enemy
appears excessively curious or persevering, the mollusk can frighten him
off by jet of ink containing unpleasantly smelling and poisonous substances.
But
in this case the octopus should hold its breath itself for some minutes,
expecting while poisonous liquid will be carried by current aside.
As against to majority of octopuses, worm-like octopus is not gloomy single.
The male lives in hole of the female, having drilled for itself separate
lateral burrow. Both animals in common gather food and share it with each
other.
Ritual of acquaintance of male and female proceeds very interestingly: the
young male, still living independently, finds a hole, bored by female, by
smell. He cautiously comes nearer to her hole, ready to slip away any minute
at first
sign of displeasure from the female. Gradually the female gets used to smell
of the extraneous octopus, and the second stage of acquaintance begins. The
male settles near to the female in temporary shelter. He starts feeding up
the female, throwing to her hole killed fishes and shrimps. Gradually he
passes to the third stage of acquaintance, to tactile contact. He any more
not at
once releases catch intended to the female, but any time keeps it. At this
time female can feel his body by tentacles, and chemoreceptors located on
them supplement with the new information an image of the male applying for
pair
forming. If all stages of acquaintance are passed successfully, the male
may fearlessly get into her hole and start to bore his own “apartments”.
The female
helps him, by strong “exhalations” of water throwing out from hole “building
dust” – sand and products of limestone dissolution.
However, sometimes mollusks of this species use temporary shelters: they
dig holes in sand, strengthening their walls by slime. Not sexual matured
males
and very young females were not found suitable place for arranging of constant
hole make it.
The generated pair of mollusks hunts more successfully, than the single.
Such mollusks quickly grow and expand hole, dissolving by secret of acid
gland layers
of limestone on walls. And once the female starts to build the big chamber
in lateral wall of hole. It means, that the male had fertilized her, and
she had stand on the way of materinity. The nest chamber is bell-shaped one.
On
vault of the chamber female lays a cluster of small eggs (about 500 ones),
and starts to protect them both with the male. The male keeps closer to entrance
of hole. Under action of the substances secreted by the female, at him large
poisonous glands start to develop, and he turns to fatally dangerous creature.
After one month from eggs translucent larvae hatch. They swim out from native
hole, and lead secretive life in crevices of a reef the first month of life.
Later females start to bore holes fit to live in, using natural cavities
as a basis of construction. Males live at the sandy bottom of shallow waters,
but later they move to holes of females.
As at all cephalopods, this animal breeds once a life. Its life expectancy
does not exceed 1 year, and after larvae hatching the parental pair perishes.
Vineglass
cuttlerfish (Utriculosepia pustulosa)
Order: Cuttlerfishes (Sepiida)
Family: Cuttlerfishes (Sepiidae)
Habitat: Pacific Ocean, top layers of water.

Picture by Timothy Morris
In human epoch one of tendencies in evolution of cephalopods
was the pelagization – the transition to life in thickness of water. Such way
of development was selected mainly by squids, though among other groups of cephalopods
also pelagic species evolved. People know species of pelagic octopuses and cuttlerfishes.
In Neocene mass extinction of plankton animals stimulated the new wave of settling
of open ocean by cephalopods. Among them many species at which the complete
life cycle passes far from coast had appeared. Some of them became strong and
muscled necton animals, fast swimmers overcoming longest distances. Others had
turned to fragile plankton organisms passively soaring in thickness of water
and freely transferring by currents. Life in plankton has made some cephalopods
fantastical creatures with strange, but effective strategies of life. The part
of plankton cephalopods has almost lost the art of swimming. These creatures
soar in water, moving only when someone will disturb them, or when they are
threatened. The rest of time they lead passive, perhaps, even “vegetative” life.
And sometimes in anatomy of animals practically direct parallels with features
of structure of some plants are shown.
Among the plankton organisms inhabiting waters of southern part of Pacific Ocean
there is one species of pelagic cuttlerfishes. This is small creature (length
of its body is only 12 – 15 cm) with big eyes and almost completely transparent.
Covers of body of this mollusk have bluish color; therefore it seems that the
cuttlerfish is made of blue glass. In thickness of water this mollusk is completely
invisible, and its presence is given out only by big eyes, shining when the
ray of sunlight from the surface of water gets in them.
Many similar mollusks live in thickness of water, and simply the pelagic cuttlerfish
of one more species may get lost among their variety. But it differs in very
specific receptions of hunting which do not meet at other cephalopods. At this
species of cuttlerfishes there are very long tentacles (extending to one meter
and even more), on which strongly changed suckers are located. Each sucker represents
formation like the transparent bladder placed on thin stem, or simply small
vineglass. For this feature the mollusk has received the name vineglass cuttlerfish.
Edges of suckers are strongly narrowed and supplied with the valve, which is
closed by tiny cover. The corneous hook on sucker is reduced and represents
thin sensitive bristle. When any small creature (fry or crustacean) touches
it, bladder opens and engulfs prey. The similar adaptation was developed on
leaves of floating plant bladderwort (Utricularia) – this feature represents
a surprising example of convergence of animal and plant.
Such adaptation was developed for feeding on the most accessible plankton catch
– small crustaceans. Vineglass cuttlerfish simply passively soars in thickness
of water, having stretched tentacles. Its fins are reduced up to two small “patches”.
With their help mollusk supports the necessary position in water, but can not
swim quickly. Soaring in thickness of water, cuttlerfish waits, when plankton
crustaceans will touch its tentacles. Having filled suckers with caught crustaceans,
cuttlerfish stretches feelers near the mouth one by one, squeezing out prey
by contractions of bladder right in mouth. Dwarf males of this species differ
from females not only externally, but also by preferred diet: they eat small
shrimps.
For self-defense the vineglass cuttlerfish uses two tentacles with pointed corneous
hooks - the disturbed animal extends them aside the aggressor. Thus the body
and trapping tentacles blacken. After attack the animal sharply turns transparent
and contracts tentacles. It can swim, pushing water from pallial cavity. If
the enemy is small and represents the same fragile plankton creature, vineglass
cuttlerfish can attack it, but against the large and well-protected animals
it is defenceless. In case of attack of fishes it tries to leave from chasing,
using the ability to autotomy: with the help of the special muscle one or two
tentacles tore off from its body and start to contract and to wriggle actively,
distracting attention of predator from the mollusk.
Vineglass cuttlerfish lives within about two years. It breeds once a life, bearing
the clutch of several hundreds of small eggs on tentacles during three – four
months. This time the female eats of nothing and leads the extremely passive
way of life – it moves only when it is necessary for protection of posterity.
At this mollusk the male is very small – its length does not exceed 5 cm. At
it there are reduced tentacles, except for one, about 10 cm long. With the help
of this tentacle the male fertilizes females. In fact the male is the larva
finished growth early and having reached sexual maturity. It lives less than
half-year, but for this time it can fertilize several females. During the courtship
ritual the male blackens, showing itself to the female. It accurately “perches”
on the body of the female from above, trying to not touch her tentacles – the
female can easily catch and eat the own groom. If the female is ready to fertilization,
male enters spermatophors in her pallial cavity with the help of long tentacle,
and then leaves the female.
Siphonophore
squid (Natarospectrum mollissimus)
Order: Squids (Theutida)
Family: Onychotheutids, or Clawed squids (Onychotheutidae)
Habitat – deep-water zone of Pacific Ocean.

Picture by Timothy Morris, colorization by Biolog
Initial picture by |
From the moment of their occurrence on the Earth cephalopods
were mainly active swimmers and strong contenders of pelagic fishes. But some
species of these mollusks in different geological eras passed from life in top
layers of water to life in oceanic depths, and some ones even on oceanic bottom.
Here, in conditions of low temperatures, darkness and enormous pressure, they
varied up to unrecognizability, getting the most fantastical shapes. In depths
at lack of catch it is energetically unprofitable to be active swimmer, but
the passive predator, patiently expecting suitable prey achieves much more success
in survival. Therefore squids lodged in abyss, have almost lost ability to swim.
Siphonophore squid is one of such deep-water pelagic squids. It spends all life
in thickness of water at the depth from one and half up to three kilometers.
This mollusk passively floats in water, having stretched tentacles in sides.
To facilitate floating this squid in water, its large liver with stock of easy
fat serves as a float. Body of this mollusk is very soft, jelly-like and translucent.
Siphonophore squid is a creature, not much more dense, than the jellyfish.
Eyes of this creature are large, telescopic, sit on flexible “stems” and can
turn in various sides.
Principal organs of movement at squids are muscled fins and the jet device of
swimming funnel and pallial cavity. Siphonophore squid had lost ability to jet
swimming because its muscles are strongly reduced. Fins at this species are
short and wide, a little bit similar to fly wings by shape. Squid usually passively
soars in thickness of water and swims only when it is disturbed. In this case
it flaps by fins, as by wings. However it can not swim for a long time, and
quickly becomes exhausted.
For all cephalopods presence of different amount of tentacles is typical: it
is a former forward part of creeping sole (the back part had turned to swimming
funnel). Tentacles of the siphonophore squid are long, and also have very characteristic
shape for what the animal has received the name. They very much resemble a colony
of any coelenterates. Tentacle bases have relatively advanced muscles, and due
to this tentacles can move. But in the significant part of muscles they are
reduced, and cannot be bent any more. Distal part of tentacles is thin; suckers
are partly reduced and strongly modified. The part of suckers has turned to
glue glands, secreting on the surface of tentacles sticky secret. Here and there
among them there are luminous glands - the modified large suckers looking like
bulbs. They are almost closed, forming bladder in which secretions of the special
gland containing luciferin and luciferase enzyme mix up. When the squid simultaneously
emits two reagents in cavity of bladder, it brightly flashes for any time.
Tentacles are the main hunting adaptation of this squid. Being a passive animal,
this squid has not ceased to be a predator. Simply prey itself comes to a predator
now. Lighting squid involves to itself small fishes and invertebrates. These
creatures are simply pasted to feelers, and while they have failed to free,
the squid grips and eats them.
Except for usual tentacles, for squids two trapping tentacles are characteristic.
At this species they are very well advanced, and muscles easily permit them
to bend in any direction. Trapping tentacles are thin, easily extensible, and
on their ends suckers and horn hooks are advanced. Having felt that to tentacles
small catch is pasted, the siphonophore squid lets out one trapping tentacle
(usually it keeps them involved), and seizes prey before it will free.
Siphonophore squid is rather large creature: though length of its body is only
about 25 – 30 cm, tentacles of this cephalopod are very long – up to 2 meters.
The male is much smaller than the female – its length is about one and half
meters including tentacles. At it organs of smell sense are strongly advanced,
and it swims much more actively than the female, searching for her by odorous
trace.
Similarly to all squids, this species breeds only once a life. The female bears
clutch of 400 – 500 large eggs about half-year, then young squids remain on
the body of female till one month. They feed with prey caught by parental tentacles
(after their hatching the female does not eat any more, and her body degrades).
The posterity cautiously creeps on parent tentacles, eating the catch pasted
to them. They do not paste to tentacles of the female because covers of body
at them secret the substances interfering action of glue of this species. Usually
young squids abandon the female when it perishes and also its tentacles cease
to emit sticky secret. They become able to breed at the age of approximately
five years.
Unequal-handed
angler squid (Lophiotheutis anisomanus)
Order: Squids (Theutida)
Family: Angler squids (Lophiotheutidae)
Habitat: Atlantic Ocean, depth from 200 up to 2000 meters.

Picture by Timothy Morris, colorization by Biolog
Initial picture by Timothy Morris |
“Planktonic accident” at the border of Holocene and Neocene
rendered enormous influence to fauna of deepwater organisms. Inhabitants of
depths, where the sunlight does not reach, entirely depend on efficiency of
top layers of water. Therefore in epoch of mass extinction of pelagic animals
deepwater fauna had suffered serious damage.
In Neocene depths of oceans are populated as richly, as in Holocene, but their
inhabitants belong to descendants of shallow-water and coastal forms. In depths,
as well as at the surface of water, fishes and cephalopods compete to each other.
Among deepwater cephalopods the group of passive predators evolved – these ones
are angler squids, or unequal-handed squids. They live in all oceans and populate
the thickness of water. Some species keep near to continental slope and at underwater
mountains. These ones are passive and slowly moving forms with rounded fins
and short body. They swim slowly, and mostly passively hang in thickness of
water, having stretched tentacles, which serve only for supporting of body in
water thickness.
Unequal-handed squid is the typical representative of this group of animals.
It is widely settled in Atlantic from temperate latitudes of both hemispheres
up to equator. Length of body of this mollusc is about 20 cm. It has short sac-like
body with rounded fins. In rest the squid supports the necessary position in
thickness of water, slightly waving by fins. Maintenance of buoyancy is promoted
also by fatty liver shifted to the forward part of its body.
Tentacles of unequal-handed squid are wide and flat. They had lost almost all
suckers (only a few pairs of suckers directly near the mouth are kept), and
had turned to organs promoting soaring in water thickness. The length of tentacles
does not exceed length of body of squid. In rest this squid soars in thickness
of water, having turned tentacles upwards and having stretched them in sides,
as petals of flower.
Angler squids are predators; they catch prey with the help of the trapping hands
modified in special way. Trapping hands of these molluscs have different length
and different purposes. Right hand is very long and extensible – usually it
may be extended to the length of up to three body lengths of the animal, but
at some species it extends even more. Along this trapping hand a bunch of muscular
fibres stretches, due to which this hand may be contracted very quickly. The
tip of right hand frequently may be translucent and leaf-shaped. On it organs
of luminescence are located; their size and amount vary at various species.
These are the modified suckers becoming a refuge of colonies of luminous bacteria.
The left hand of angler squids, on the contrary, is short and muscled, with
well advanced suckers and hooks. This animal allures prey by right hand, imitating
movements of small animals, and seizes and drags prey to the mouth by left hand.
In salivary glands of these molluscs the strong poison, killing fishes and other
animals with slow metabolism for some seconds, is producing.
At unequal-handed angler squid right trapping hand is extended to the length
of about one meter. On its tip some bubbles (modified suckers), containing colony
of luminous bacteria, are advanced. They are located in one line and resemble
a colony of doliolids or siphonophoras, when they are visible in darkness of
depths as a chain of fires. The hunting squid weakens muscles, extending bait
hand, and the luminous bait does not give out its presence. Some muscles force
bait to coil, strengthening similarity to colony of small live organisms. Bait
hand imitates movement of small animal so skilfully, that it seems, as if other
live being is adhered to squid – the difference between inactive animal and
mobile bait is expressed so. When the prey involved with bait swims up closer,
unequal-handed squid gradually contracts muscles of tentacle, forcing prey to
come nearer to trapping hand.
Trapping hand of this squid is really similar to trap. It is short, but its
tip is wide and is divided into two lobes with elastic cartilaginous “skeleton”.
Cartilages are connected by adductor muscles, which “slam” trapping hand, when
prey touches it. The inner surface of trapping hand is supplied with plenty
of suckers, and on its edge sharp corneous hooks grow.
At angler fishes of several families males attached to the female to all further
life and led parasitic way of life, but at other species males were simply tinier,
rather than females. At angler squids males always lead an active way of life
and are smaller, than females. They keep an anatomy and symmetry of body typical
for squids, and trapping hands at them are not modified. Males of angler squids
swim quickly and actively search for females ready to fertilization. Sexually
mature male can fertilize several females before it will die from old age.
Ritual of “acquaintance” at these squids passes in some stages. Coming nearer
to the female, male keeps in the distance from its tentacles – the female can
seize him and eat. In order to allow the female to identify him, male emits
in water the special chemical substance, signalling to the female about his
presence. After that, being careful, male begins direct “acquaintance”. He gradually
comes nearer to the female, keeping directed to her by tentacles and slowly
waving by fins. Such pose enables him to feel movements of the female with the
help of receptor cells on tentacles, and to escape from the female with the
help of one sharp jet pulse in case of danger. If the female does not show aggression
and is ready to pairing, male goes to closer contact. He cautiously creeps on
head to the female and starts to touch her body. Having found out an entrance
to pallial cavity, he enters in it spermatophore of lengthened form in dense
cover, and abandons the female.
After fertilization the female actively eats till some time, and in her body
the clutch ripens – some hundreds thousands tiny eggs. In same time her internal
organs gradually start to degrade – inevitable process of ageing begins. The
female lays eggs covered by common slimy jelly-like mass, which strongly swells
in water. She keeps clutch by tentacles and trapping hand. In same time the
luminous bait on her hand degrades. But the part of luminous bacteria has time
to penetrate into clutch, and infects developing embryoes; therefore young angler
squids are already able to luminescence. After young growth hatching the female
perishes.
The complete cycle of development of this squid takes about three years, from
which last eight months of life fall to incubation of clutch.
Dwarf
unequal-handed squid (Anisomanella subtilis)
Order: Squids (Theutida)
Family: Angler squids (Lophiotheutidae)
Habitat: Atlantic, depth from 200 up to 2000 meters.

Picture by Timothy Morris, colorization by Biolog
Initial picture by Timothy Morris |
Representatives of angler squid family appeared rather successful
inhabitants of depths. After the type of the passive predator squid alluring
prey with the help of tentacle had formed, this group of molluscs had evolved
a plenty of species adapted to various prey. The majority of these squids hunts
fishes, but some species prefer small prey like shrimps. Dwarf unequal-handed
squid, the smallest species of angler squids, belongs to the number of these
species.
Dwarf unequal-handed squid is very small squid. The body length of adult individual
of this species does not exceed 5 cm. This squid is not adapted to swimming
and has the inflated body with small head. Fins are very wide; with their help
animal can move to small distances and supports position of body convenient
for hunting. Short flat tentacles with rudimentary suckers and advanced horn
hooks in rest are stretched in sides. They form the floating “anchor”, assisting
to stop at prey catching.
This tiny mollusc has very long bait hand – it can be extended to the distance
up to half meter. The bait on the tip of this hand is lengthened and consists
of the line of closely put luminous organs. It precisely imitates pelagic luminous
worms which deep-water shrimps, the basic food of this squid, eat. Besides them,
dwarf unequal-handed squid eats worms also involved with light of bait. The
modified suckers turned to organs of luminescence, are placed on each side of
the bait on long stalks, imitating articulated body of worm with outgrowths
on each side of segments. On tip of bait two corneous hooks with jagged internal
edge are advanced. They assist to drag prey closer to trapping hand. The animal
grapples the deceived shrimps by hooks, and sharply contracts bait hand. It
simultaneously tightens prey to itself, and moves nearer to it. Squid sticks
to armour of shrimp by trapping hand, not giving it an opportunity to escape,
and kills, putting to the crustacean a poisonous bite.
After fertilization by dwarf male the female lays eggs on internal surface of
trapping hand and keeps them to the development of young ones till about 3 months.
At this time it eats of nothing and is quickly exhausted; its body deeply degenerates.
The significant part of muscles resolves, volume of liver decreases, and the
digestive path disappears almost completely: only a tissue cord without lumen
remains from it only. The female perishes soon after hatching of young ones
from eggs. The complete life cycle of this species lasts about 16 months.
Toady
squid (Bufotheutis dioneomanus)
Order: Squids (Theutida)
Family: Angler squids (Lophiotheutidae)
Habitat: Pacific Ocean, depth of 500 – 3000 meters.

Picture by Timothy Morris, colorization by Alexander Smyslov
Initial image by Timothy Morris |
The family of lophiotheutid squids (“angler squids”) is widespread
in the deep water area of the oceans of the world. These passive cephalopods
are the ecological analogs of deepwater anglerfishes of Holocene, and in due
course of evolution they got a distant similarity to these fish in their appearance.
They almost lost all capability for the rapid reactive swimming and became passively
drifting planktonic animals, capable of only swimming slowly using the strokes
of their wide triangular fins.
Toady squid is widespread practically everywhere in Pacific Ocean, living in
deep water layers far from the coasts and underwater mountains. The body length
of this mollusc is approximately 20 cm not including tentacles. The passive
way of life of toady squid had led to the striking changes in appearance of
this mollusc in comparison with its rapidly swimming ancestors from upper layers
of water. This species has short wide body, covered in flabby skin of pale gray
color, which can both extend and get rumpled. The tentacles of this animal are
short and wide, with well developed suckers and large corneous hooks at the
tips. Their function is keeping of the grasped prey. Near the mouth suckers
are reduced, but the corneous hooks are very large and are wide.
This mollusc has good sight. It has large mobile eyes located at the edges of
its flattened head. This squid easily notices the lights of deepwater animals
in gloomy depths.
The trapping tentacle of toady squid is short and wide. Its trapping part can
fold along like the leaf of Venus flytrap plant (Dionea muscipula). Sharp corneous
hooks grow along the edges of this tentacle. Thin and flexible baiting tentacle
can stretch out to the distance of one-and-a-half meters. When squid hunts,
this tentacle “lures” prey from a distance with the help of the bait and grasps
it with the “trap” of the trapping tentacle. The strong squeeze of trapping
tentacle and corneous hooks stab the prey and kill it.
The beak of this squid is wide, and mouth can open very wide as well. The poisonous
glands are not developed, because squid kills its prey with corneous hooks.
This species is adapted to hunting large prey, whose mass can exceed its own
one three times. Large animals are caught by the trapping tentacle right behind
the head. When the prey is killed, squid grasps its head by tentacles and begins
“stretching” onto its prey with the help of tentacle movements. Moving the tentacles,
the predator crawls along the prey body from head to tail, keeping with the
help of the hooks. Its mouth extends strongly, and the squid gradually swallows
its prey, forcing it into a ring-like shape in its stomach. The lack of firm
solid skeleton greatly helps this mollusc in carrying-out this specific method
of feeding. After filling its stomach, the animal takes almost globular form
and floats in the water depth, slightly moving its fins to keep stability. The
prey is digested almost completely.
This species of squid bears eggs on its own body. After the mating the male
stays alive – its energy is enough to fertilize several females. The female
lays several hundred thousands of very small eggs, encased in jelly-like shell
looking like wide ribbon. It winds the clutch around the baiting tentacle and
guards it during several months, driving off enemies with the help of trapping
tentacle. The degeneration of the tissues of its body occurs at this time –
the fat becomes completely used-up, the reduction of intestine and digestive
glands takes place. Toward the end of the incubation the female becomes slow-moving
and sluggish, but it reacts to the approaching of predators and makes threatening
movements with its trapping tentacles. After the hatching of young squids it
dies.
Young animals easily come to the surface because of drops of fat in their liver.
They feed in upper layers of water on plankton crustaceans and fish fry. As
they grow, the young toady squids migrate into the depths. The development of
this species occurs very rapidly: at the age of 4 months, the young animal already
has a body shape typical for the adult individuals, and it has half of the body
length of adult mollusc. During their first year they reach the typical size
of the adult individuals, and at the second year of life they become sexually
mature.
Translated by Bhut, the forum member.
Worms
|
Spat
leech (Serpentobdella jaculata)
Order: Proboscisless leeches (Arhynchobdellida)
Family: Jaw leeches (Gnathobdellidae)
Habitat: tropical forests of Southern Asia.
The damp climate of Neocene epoch substantially favors to development of most
various groups of moisture-loving animals. In tropical rainforests where each
day it is rainy and it is always possible to find pools of fresh water, there
is especially favorable inhabitancy for various invertebrates: mollusks and
worms. But if snails are sluggish vegetarians, worms have turned to original
predators of forest canopy. Certainly, not all worms have achieved success
in development of wood habitats, and only their one group – leeches had become
such group. Ground leeches were known in Holocene, but they were basically
medium-sized animals. In Neocene they have grown to monsters of frightening
size, becoming among appreciable predators in wood ecosystems. Huge slobber
leech Titanobdella glutinosa places sticky traps on branches, from which
even the cat-sized animal may not escape. Having excellent masking and prompt
reaction adderleech Viperobdella
arborea catches small vertebrate animals, killing them by poisonous sting.
One more relative of these deadly creatures is the spat leech, developing an
original way of hunting.
Spat leech is large predatory worm living only on trees. Life of spat leech
is closely connected to water: its covers are soft, mucous, and very quickly
lose water. But this worm regularly humidifies skin, using water gathering
in crowns of epiphytic plants. The body length of this worm is up to 50 cm
at width about 8 - 9 cm, but it is rather changeable size: the leech can contract,
compressing almost to sphere or to stretch up more than twice. Colouring of
the top side of its body is cryptic: grey with longitudinal brown strips and
black strokes. The bottom side at this leech is colored brighter: it is yellow
with round black spots.
Spat leech eats insects and small vertebrates – frogs, lizards, and even tiny
birds. It is sluggish creature, but catches these mobile animals, spitting
out in them jet of saliva which in air becomes sticky and viscous (by the same
way Peripatus similar to hybrid of centipede and caterpillar, the representative
of ancient by origin phylum Onychophora, catches forage to itself). In salivary
glands of this leech two components are formed, each of which separately is
a liquid. But it splashes them simultaneously, and under action of oxygen of
air there is a chemical reaction between them.
Saliva splashing occurs due to strong ring muscles surrounding every gland.
Nerves approach to them from one pair of nerve ganglions, and under influence
of one impulse substances from two pairs of glands splash synchronously. The
distance of this “shot” may account about 1 meter. In searches of catch this
worm is guided first of all by sight: the spat leech has six well advanced
eyes. Sight at this worm is rather keen: this leech notices small beetle at
the distance of two meters, and especially sharply reacts to moving objects.
This worm spends all life on trees. More often leech passively waits, while
possible catch will come nearer to critical distance itself. After that well-aimed
“spittle” follows, and saliva of leech, having stiffened to rubber-like viscous
substance, pastes bird or insect to leaves. If the bird brought down by the
leech will fall on branches outside of reach of the leech, it has chance to
escape: the saliva of the leech thickens very quickly, and in some minutes
loses the property of sticking. Then it is easy to clean it off from feathers.
But more often at the pasted prey anywhere it is not time to leave – leech
snatches it, killing by sting of three cutting jaws, and then swallows prey
entirely.
This leech, similarly to all neighbours, is hermaphrodite. Adult animal leaves
mucous cocoons with eggs in tree-trunk hollows filled with water, or in crowns
of epiphytic plants and in leaf axils filled with water. In each cocoon it
is totaled up to 200 - 300 small eggs stuck together by slime in sticky paste.
In 2 months of incubating from eggs thin translucent young leeches hatch. They
crawl out on tree, search for damp places and more often settle in moss. Here
they are not threatened with danger to dry, and it is possible to find a plenty
of prey – small snails and insects with delicate covers. They grow and mature
slowly: only at the fifth year of life at length about 30 cm the leech becomes
adult. But these worms differ in longevity enviable for their size: the age
limit at spat leeches can reach 50 years.
Hanging
wood leech (Dendrobdella pendula)
Order: Proboscisless leeches (Arhynchobdellida)
Family: Jaw leeches (Gnathobdellidae)
Habitat: rainforests of South-Eastern Asia, forest canopy.

Picture by Biolog
In Neocene epoch when the ancient tropical fauna had strongly
suffered as a result of mass extinction, in new rainforests representatives
of various groups of animals, occupied before this time the subordinated position,
had appeared. In conditions of hot and damp climate in woods of South-Eastern
Asia ground leeches had reached the big prosperity. Some especially large representatives
of this group can successfully compete even to snakes, hunting small animals,
birds and insects. They have developed various tactics of hunting that has permitted
them to avoid a competition with each other – some of them put sticky substance
on branch, others catch small animals, masking in the environment. Due to different
tactics of food getting these worms do not compete with each other.
Next large land leech lives in woods of South-Eastern Asia is the hanging wood
leech. It is rather sizable worm though some giants of rainforest from among
its distant relatives surpass this species in size much more. The length of
body of hanging wood leech in rest reaches 15 – 17 cm at width about 4 cm, but
it can stretch almost up to 30 cm, moving or attacking catch. Similarly to the
majority of leeches, this is rather passive predatory animal. Justifying the
name, hanging wood leech waits for catch, hanging for a long time on bottom
side of leaves and branches headfirst, keeping by strong back sucker and having
compressed. It arranges an ambush above the branch or leaves where small animals
appear oftenly. Sometimes it hides above flowers the where probability to meet
potential catch is higher. To be less visible for catch, and also to not fall
prey to large predators of different kind, this leech till the process of evolution
has got excellent masking. By colouring and shape of body it simulates plant
leaves: on the top part of body on green background light longitudinal strip
crossed from sides by short strokes stretches, and on edge light points bordering
a body like faltering strip are placed. The bottom side of body is grayish-white
– it simulates the bottom side of leaf.
Having hung under branch, this worm waits for catch. Quickly stretching, this
leech seizes by three sharp jaws small animal (usually insects, less often small
vertebrates – frogs, lizards, mammals) moving on branch in the bottom. Smell
organs and eyes are well advanced at this species – the animal determines the
presence of catch with the help of sight and sense of smell. Eyes of this leech
are rather large, located on edge of forward part of head. The animal badly
distinguishes colors, but determines contours of objects well and reacts to
movements. Especial interest at this leech is caused with subjects which movements
are well visible on general background. In connection with presence of such
predator some insects are adapted to mask from the hanging wood leech, rocking
in a step to movements of leaves.
Having caught prey, leech kills it by bite and swallows entirely. Having swallowed
catch, predatory worm becomes sluggish. The full leech hides on bottom side
of branch, having attached to it simultaneously by mouth and back sucker (thus
its body is bent U-shapedly), and digests food in relative rest.
Similarly to all leeches, this worm is the hermaphrodite. For breeding the leech
does not leave crones of trees: it lays slimy cocoons containing about two hundred
large eggs in top part of trunk hollow of large tree in which rain water gathers.
Frequently for this purpose some leeches use the same tree-trunk hollow, and
in one tree-trunk hollow it may be totally up to fifty cocoons. The incubating
of eggs lasts about two months. Young leech gets out of cocoon and falls in
pool of water accumulated in tree-trunk hollow. First some days of life young
leeches swim in this water, eating insects fallen in water, and even their own
smaller congeners. Later they abandon the hollow, and lead terrestrial life
in foliage of forest canopy.
Egg leech (Oobdella ovivora)
Order: Proboscisless leeches (Arhynchobdellida)
Family: Jaw leeches (Gnathobdellidae)
Habitat: tropics, subtropics and temperate areas of Old World, coasts of reservoirs.
In epoch of biological crisis at the boundary of Holocene and Neocene the variety
of leeches remained almost the same, as in human epoch. Destruction of tropical
forests and global congelation had resulted in extinction of some tropical
species of leeches, but stabilization of natural conditions in early Neocene
had resulted in a new wave of speciation of these worms. Among them the species
using new food sources not developed by these worms earlier had evolved.
In significant territory of Eurasia and in the north of Zinj Land near reservoirs
one species of leeches lives, mastered an ecological niche new to these worms.
It is an amphibious species of leeches which eats contents of the bird’s eggs,
biting small apertures in egg shell with powerful jaws. It is the egg leech,
a worm up to 7 – 9 cm long. Body of this worm is narrow, almost cylindrical;
head end is expanded and rounded in outlines. On it six small eyes are located.
The oral sucker is small in size, but jaws of egg leech are extremely powerful,
with slightly jagged edges. The back sucker is well advanced – with its help
worm is kept on the substratum during the feeding. The body of egg leech is
colored green with several longitudinal lines of brown spots. On edges body
is bordered by a line of tiny red dots.
This species of leeches is stenophagous – it feeds strictly on contents of
bird eggs. Salivary glands of egg leech produce substances interfering protein
denaturation, and worm exhausts freely the contents of the whole medium-sized
bird egg. In connection to such specific diet this worm is starving during
the most part of year. Egg leech actively feeds till only few weeks a year
when nesting of waterfowl begins. At this time it actively grows and gathers
full intestines of contents of bird eggs. For feeding this leech creeps out
on land and searches for hatching bird. Having found a clutch suitable for
attack, leech crawls into the nest from below, bites through egg-shell and
drinks its contents. If the egg is large, leech can creep inside of the shell
and live there for several days, continuously feeding. When in egg the embryo
develops, leech can not eat such egg any more; therefore the most part of attacks
is made in the beginning of nesting of birds. The egg leech defines a stage
of development of bird eggs by smell. In tropics, where seasonal prevalence
in bird nesting is not expressed, this leech attacks on bird clutches up to
five times a year.
When the leech exhausts egg white, it actively excretes the water received
with food.
After feeding in organism of the leech eggs develop. Fertility of this worm
species makes up to 150 eggs for one clutch covered with a cocoon with dense
environment. The amount of clutches within one year is in direct ratio to amount
of attacks on bird nests. Cocoons are rich in nutrients therefore young ones
does not need for food for a long time. The first food of young leeches is
the contents of a cocoon – nutritious slime.
Young leeches eat eggs of small songbirds nesting in reeds. In searches of
such nest the young egg leech can rise on stalks of marsh plants to the height
of more than one meter from the ground. In search of clutches it is helped
very much by very advanced chemical feeling: the leech defines the presence
of birds by the smell of their dung falling in water. This smell is so attractive
to egg leeches that they gather under a nest of bird in numerous groups and
almost simultaneously start to search for a nest. As a rule, one or several
leeches succeed to find a nest, and the clutch may be destroyed within several
hours. If it is not enough bird eggs, leeches can search for eggs of reptiles
– snakes and turtles. Sometimes young egg leeches attack eggs of frogs or fishes.
Within two years the egg leech grows up to the size characteristic for adult
individual.
This species of worms was discovered by Nem, the forum member.
Dwarf
bird leech (Ornithoclepsis dimorpha)
Order: Proboscis leeches (Rhynchobdellida)
Family: Snail leeches (Glossiphoniidae)
Habitat: warm-temperate areas of Europe, Northern Africa, Near East.

Picture by Biolog
Land species of leeches are especially characteristic for
tropical rainforests, where they often reach very
large size. Independently of the tropical species leading the origin from
ground leeches of Holocene epoch, some species of aquatic leeches of Eurasia
passed to terrestrial way of life. One of such species is dwarf bird leech,
the tiny species of leeches feeding on blood of various waterfowl and ground
birds. Body of this worm is up to 10 – 20 mm long, flattened in dorsoventral
direction. The body shape is rather unusual for leeches: the body consists of
two halves sharply differing in shape. Forward part of the body is very lengthened
and thin, separated by expressed constriction from the back part. Back part
is expanded and has rounded outlines – in it large lateral outgrowths of intestines
are located, in which the exhausted blood is kept. Because of this distinction
of body parts there is a specific epithet “dimorpha”, meaning “two-shaped”.
The top side of body has grey color with black cross strokes; the bottom side
is white.
This species of leeches spends a lot of time on land; sight has great importance
in its life. Large eyes are located on the head end of the body; their number
can change from 4 up to 8 ones. Moreover, some small secondary eyes develop
on the back end of the body in parallel to the edge of sucker. Dwarf bird leech
can not distinguish colors, but distinguishes keenly contours of subjects at
the distance up to 20 – 30 cm from itself. Also at this species sense of smell
is well advanced; with its help this leech can find the birds sleeping on the
ground.
This worm eats blood of birds, sticking to various parts of their bodies. Waiting
for prey, leech hides among grass stalks or near stones. When the bird appears
beside, leech quickly creeps up to it and creeps in plumage along its leg. On
the body of bird leech is fixed on skin by back sucker and extends forward part
of body in searches of area of skin under which the blood vessel lays. In addition
to blood this species eats small invertebrate parasites of bird – feather lice,
chewing lice and larvae of tineids moths. Sometimes leech bites through a body
of mites full of sucked out blood, and drinks contents of their intestines,
already begun to be digested.
During the contact of the host bird to its congeners this leech can creep on
them.
Dwarf bird leech spends the most part of life on land, appearing in water only
during the flooding or if the host bird decides to bathe. Therefore the problem
of water preservation is especially important for it. This species can produce
the slime stiffening as lumps inside which it waits through the droughty periods
and frosts. In northern parts of area where snow can fall, dwarf bird leech
winters under the layer of wood litter, sticking for itself a cocoon of slime
and small vegetative rests.
This species lays the cocoons containing up to 40 eggs in wet ground and moss,
at the coast of shallow reservoirs. The wall of cocoon is denser, than at other
species of family, but through it a part of water necessary for development
of eggs is penetrating. The cocoon is protected by the adult leech, the young
ones live for some time on its body (like at related species of a human epoch).
During first two months of life young leeches do not leave water and eat blood
of tadpoles and frogs.
The idea about existence of this species of worms was proposed by Nem, the forum member.
Horse-headed
Fishing-Leech (Hippoobdella gigas)
Order: Proboscis leeches (Rhynchobdellida)
Family: Snail leeches (Glossiphoniidae)
Habitat: Permanent rivers and lakes of Equatorial Africa.
Leeches of various sorts had become more specialized during the onset of the
Neocene, due to stresses put on species competing with them, and opportunities
to diversify. In the rivers of Africa, aquatic leeches had passed to becoming
larger, and predating on fish, one such form is the Horse-headed Fishing Leech.
It is rather large for a leech or non-digging worm, reaching a length of 20cm,
width of body in the middle is 4.5cm Body is of an original shape, fore and
mid part of the body are muscular, belt-shaped and flattened, resembling some
of its ancestors. Front of the animal forms a constricted “neck” able to coil
in an s-shape, and oral sucker is large and flares out at the sides, these features
allow the leech to quickly seize and fasten onto its prey, small fish, tadpoles
and frogs, when attacking prey, animal can extend itself up to 30cm long. Coloration
is a plain greenish brown, lighter on underside, lacking the spots or stripes
possessed by other kinds of water leech. Both vision and sense of smell are
very keen in this animal, compared to other kinds of worm, the three pairs of
eyes are large and located toward the front of the head, being most sensitive
to movement.
Hunting is achieved passively from ambush; animal coils itself around submerged
vegetation, fastening with rear sucker, forming its head and forward end into
an s shape, which allow it to strike effectively. It is from this pose that
it gains its name “horse-headed”. After prey is secured with its large oral
sucker, it begins to feed by extensive its long, powerful proboscis in order
to drill into the prey, extracting its juices and flesh. If disturbed by a predator
or interloper, it will produce a bitter mucus from its skin which is distasteful
to predators.
Breeding takes place year round but is most frequent in the wet season. This
species lays a few cocoons containing up to 80 eggs, cocoons are sticky and
are attached to aquatic plants as they are laid. Female will guard the eggs,
and young spend first part of their life attached to the underside of their
mother, eventually dispersing and feeding on small crustaceans, insects, and
fish fry. Individuals of this species may breed up to four times in their life
but usually less, lifespan is up to 8 years.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia
North
Island Giant Earthworm (Titanolumbricus sylvanus)
Order: Earthworms (Opisthopora)
Family: Garden Earthworms (Lumbricidae)
Habitat: Layers of soil within the tropical and subtropical zones of the North
Island of New Zealand, especially heavily vegetated areas.
Mankind introduced many common garden invertebrates to New Zealand inadvertently
or during the processes of horticulture, among them were garden earthworms (Lumbricidae).
The descendants of these largely out-competed many native kinds of New Zealand
earthworm, especially those specialised for certain habitats.
As a result, the native kinds of giant earthworm, some reaching as much as 1.4
meters, became extinct, and were replaced by a functional analogue, the North
Island Giant Earthworm.
This worm is large and relatively thick, reaching 1.6 meters long and as much
as 1.1 centimetres thick. Its body is an overall pinkish-brown in colour, annulations
of the segments are darker than the rest of the body. It is found mostly in
damp soils in vegetated areas, tunnels may reach a depth of 3.5 meters. This
species feeds by consuming already decaying plant matter and enriching the soil,
as such it is greatly important in forested ecosystems. Sensory organs on the
front end are sensitive to the chemicals in decaying plant matter and leaves.
It prefers warm temperatures that are humid, moist and not too dry, to develop
optimally. As with many earthworms it only emerges above ground at night (if
at all) and is fairly photophobic.
This worm is hermaphroditic, and usually breed when the weather is wet and warm,
breeding may occur frequently when the conditions are right. Individuals slide
close to each-other and produce a lot of mucus, after which they exchange sperm.
Eggs are encased in a mucous cocoon which is placed in a chamber underground,
each cocoon may be as long as 4 centimetres. The larger an individual is, the
more eggs it will lay at a time, with a maximum of about 4, but as many as 50
cocoons may be laid in one season. Worm-lings are sexually mature at about 2.5
years old. Predators include carnivorous birds, hedgehogs,
lizards and badger-like
mustelids, lifespan may be up to 5 years.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Stomach
lightworm (Gasteronereis luciferus)
Order: Phyllodocids (Phyllodocida)
Family: Nereids (Nereididae)
Habitat: Pacific Ocean, depths from 500 up to 1000 meters, symbiote of bathycrystallichthys
fish.
At the process of evolution the strategy directed to formation of symbiotic
connections between species, frequently led both species to success in struggle
for existence. Algae and fungi form lichens – unpretentious and hardy organisms.
Corals and other coelenterates, and also bivalve mollusks tridacna and goblet-like
false rudistes had formed strong symbiosis with unicellular microorganisms.
Glowing bacteria and fishes (or crustaceans) are the successful combination
for development of gloomy depths of ocean. There are many examples of success
of symbiotic species in struggle for existence.
In Neocene in depths of Pacific Ocean completely improbable symbiosis has appeared.
It is formed by two species, which at other trend of developments could play
roles of prey and predator. One species of bristle-bearing worms had exchanged
the ability to glow brightly to safety and full life, becoming a symbiote of
predatory deep-water fish. The strange habit of life of this worm has caused
its name – stomach lightworm.
This worm balances on thin side between symbiosis and parasitism. Its way of
life is rather interesting to that the animal lives simultaneously in two environments
considerably differing by conditions of existence. The back part of body of
stomach lightworm is attached in branchiate cavity, and front one – in stomach
of fish non-glowing bathycrystallichthys
(Bathycrystallichthys unlucens). Length of this worm is about 20 cm, and
thickness of body is only about 5 mm.
In connection with unusual way life forward and back halves of worm body strongly
differ in shape. The forward half of worm constantly located in stomach of fish
is protected from action of digestive juice by dense covers. On segments of
forward half there are no gills, and parapodiums are shifted to the bottom side
of body and modified to suckers permitting to attach strongly to stomach wall
of fish. The head of stomach lightworm has no eyes and tentacles, but is supplied
with strong jaws by which worm bites off food swallowed by fish. Strong smooth
rings of armour protect body of worm against traumas when fish swallows large
prey. The forward half of worm can move inside fish stomach.
On forward segments there are also large organs of luminescence. They are located
on lateral parts of segments in pairs and emit bright blue light which penetrates
through walls of stomach of fish and is seen from outside.
The back part of body of this worm strongly differs from front one. Distinctions
begin from segments which are at the level of entrance to gullet of fish. On
such segments there are gills – on first segments they are small, and closer
to the end of body turn larger, similar to bunches of thin red strings (blood
of stomach lightworm contains hemoglobin). Therefore worm does not choke, existing
more, than half inside the body of fish. On two back pairs of segments there
are corneous hooks serving for attachment. With their help stomach lightworm
is attached to branchial arch of fish. Also on back segments of worm paired
genital organs grow.
Stomach lightworm is a dioecious species. Males and females at this species
sharply differ in appearance; the present description concerns to the female.
Dwarf male no more 10 mm long adheres to back part of female body near one of
sexual apertures and feeds on her body as a parasite. It is actually necessary
for her only for egg fertilization. The gender at these worms is not determined
genetically and depends on destiny of the larva. If the larva succeeded to attach
to gills of fish, it develops as a female. When young worm starts to develop
in organism of bathycrystallichthys by “female” variant, young bathycrystallichthys
still swims in top layers of water, where the meeting with other larva is probable.
In this case it can attach to back part of body of young worm female and turn
to male. If the meeting of female and male had not taken place, the female of
stomach lightworm can breed parthenogenetically. Male has very simplified anatomy;
its body keeps only reasts of segmentation and is supplied with several suckers
on the forward end. After attachment to the female sense organs reduce at him.
The gullet of larva developing to male pierces covers of the female and is turned
out inside her body, supplying male with food.
Stomach lightworm breeds, regularly laying some hundreds tiny eggs made in slime
cocoon. The back part of male body keeps mobility, and it fertilizes the female,
entering sperm liquid in her ovoducts. Ovisacs are thrown out from branchial
cavity of fish by current of water, and emerge on ocean surface. At this time
from eggs planktonic trochophore larvae hatch. They gradually develop to pelagic
worm-like individuals which settle on gills of young bathycrystallichthyses.
The idea about existence of this species is proposed by Simon, the forum member.
Sprouting
tube worm (Phytoserpulopsis proliferus)
Order: Sabellids (Sabellida)
Family: Symbiotic tube worms (Symbioserpulidae)
Habitat: reeves of Indian and Pacific oceans, well illuminated areas of sea
bottom.
Corals took the separate place in ecosystems of Earth due to the symbiosis
with algae: they were original “autotrophic animals”, received a part of necessary
for growth organic substances as result of processes of photosynthesis. After
their extinction a plenty of various live organisms began “sharing” of their
original ecological niche – some species form body of reef from their firm
armors and shells, others catch tiny plankton, and third
ones increased organic substance due to energy of sunlight. So at Earth
of Neocene epoch the new generation of reef builders has appeared.
When water filtering and increasing of firm shell is not a problem for animals,
the function of photosynthesis at the reef was undertaken basically by green
and red limy algae forming at the bottom and underwater objects multi-colored
limy crusts and outgrowths. However among animals species, challenging at plants
their role at the reef due to symbiosis with microscopic algae, also have appeared.
Basically they are various species of mollusks, which had lodged algae in edges
of the pallium. But there are also other organisms which have offered cooperation
to algae.
On the firm bottom of reef somewhere at the depth about two meters it is possible
to see true “thickets” of limy tubes about 30 – 40 cm height, sticking up vertically.
Keeping immobility, it is possible to wait, while from tubes their builders
and inhabitants will appear. Really, some minutes after above separate tubes
clouds of dregs appear, and then tips feather-like tentacles of their inhabitants
are put outside. Gradually, having convinced of safety of world around, tube
inhabitants put out from tubes rich plumes of tentacles, unwrapped, as if exotic
flowers. Tips of tentacles are white, but their other part is colored emerald-green.
Gradually it appears more and more of them, while the congestion of tubes turns
to similarity of flower bed. However, it is enough to too curious fish or the
shrimp to swim up, and magnificent plumes disappear in a flash, having involved
in tubes.
Secretive inhabitants of limy tubes are sprouting tube worms, the special species
of sedentary worms. Their very delicate segmented bodies are constantly latent
in limy tubes. Only the head surrounded approximately with ten pinnate feather-like
tentacles up to 10 cm long, with which help the worm eats, is put outside.
The tube built by the worm, consists of layers of rather soft organic substance
inlaid with sand particles. But on it limy algae settle, giving to tubes durability,
sedimenting on them from outside thin layer of limestone.
On the caudal end of body of the worm some segments have formed sucker on which
edges there are bristles transformed to tenacious hooks permitting to be kept
strongly in tube.
At this species of worms in connection with sedentary way of life eyes have
completely disappeared. But absence of sight is repeatedly compensated by sharply
advanced organs of touch and chemical feeling. The cautious worm is difficult
for deceiving: it is enough too sharp or strong movement of water, and worms
instantly disappear in tubes.
Except for eyes, sprouting tube worm does not have also another, much more
important for life system of organs – digestive tract. The mouth at these worms
is completely disappeared, and only tissue band has remained from intestines.
But worms at all do not suffer difficulties with feeding because for some millions
years before Neocene their ancestors have entered symbiosis with photosynthesizing
protozoans. Due to this adaptation worms became rather independent organisms
living due to the symbionts.
In tentacles of worms, in friable connective tissue, colonies of photosynthesizing
protozoans, giving to tentacles of worm bright green colouring, live and breed.
Tentacles are repeatedly ramified, and this feature increases surface of their
contact with water. They are covered with permeable epithelium permitting to
symbionts of these worms to receive from water all mineral and organic substances
necessary for life. Symbiotic unicellular organisms penetrate into feelers
of each separate individual of the worm from outside. The immune system of
sprouting tube worm is arranged to allow the “own” species of protozoans to
penetrate into feelers and to occupy them, and against “strangers” the immune
system of animal works, tearing away them.
Just under wreath of tentacles some segments, producing building material of
tube, are placed. They increase tube by layers which are clearly visible on
its surface.
Body segments of the worm differ a little in structure. So, segments from middle
of an animal on which hooks are advanced, in themselves are thick and rather
not elastic. In them fat is gathering, serving as a stock of nutrients if the
forward end of worm will be nevertheless bitten off by fish or crab. And segments
from forward part of animal can stretch strongly and contract sharply: they
are responsible for impellent reactions of animal.
Most back segments of this species have completely surprising ability for animals:
they can form germs sprouting in new worms. Branches of all systems of organs
of the worm come into each such germ, and it starts to sprout through a wall
of tube. At first at the basis of tube the aperture appears, through which
it is appreciable as additional “head” moves and expands hole. Gradually this
“head” starts building of short horizontal tunnel sharply bent upwards. When
it reaches about two thirds of height of initial tube, at an affiliated individual
tentacles start to grow, and later they’ll be occupied by symbiotic protozoans
from an environment. In one colony it can be up to four generations of worms
connected together by channels of blood system. Growth of these “additional”
individuals occurs especially actively, when individuals of previous generations
are injured. Then, if damages are too strong, the founder individual may be
simply resolved by formed affiliated individuals, and the central tube becomes
empty.
At the time of highest inflow colonies of sprouting tube worms (and also of
other related species) as if blow up by eggs and sperm liquid. Eggs are carrying
away by waves to open ocean. In some hours from them larvae covered with ciliums
hatch. They will spend about one month in plankton, and then few larvae have
survived for this time will come to reef with next high inflow. They settle
on bottom and give rise to new colonies. Young worms have no symbionts, but
settle on sunny sites of reef. They still have intestines, and they differ
by nothing from other sedentary organisms. But gradually symbionts of the necessary
species find their tentacles and breed in them. Then intestines gradually degenerate,
and worm starts to live exclusively due to “lodgers”, providing them house
and safety.
Reeves of Indian and Pacific oceans are inhabited also by other species of
tube worms entered symbiosis with photosynthesizing organisms:
Poisonous
tube worm (Phytoserpulopsis toxicus).
This species, perhaps, is protected
from enemies better than others:
it enters symbiotic relations with
poisonous protozoan Gymnodinium.
Some species of these protozoans
cause deadly for many other animals
“red inflow”, but worms easily coexist
with such dangerous creatures due
to especial enzyme, neutralizing
poisonous substances getting in blood.
But fish or shrimp, having tried
reddish-orange tentacles of this
worm, will not want to make it second
time any more. As if understanding
its own safety, this worm is not
so cautious, as other species: it
hides only at rather strong movement
of water or direct touch to tentacles.
Despite of fine protection, this
worm is rather rare: the symbiotic
protozoan species occupying its tentacles,
meets too seldom, and most part of
larvae of the worm perishes, not
having met such symbiont. Tubes of
this species are narrow, but grow
in dense groups and reach about half
meter height.
Grey
tube worm (Phytoserpulopsis glaucus) has
given a shelter to cyanobacteriae,
and it also has made it inedible
for many fishes. Tentacles of this
species have bluish-green colouring,
and their edges form the expanded
blades which the worm keeps perpendicularly
to sun beams. For recognition of
sunlight in tissues of tentacles
special cells reacting to light were
developed. Due to wide prevalence
of symbiotic algae this species may
occupy extensive areas of reeves.
It differs in rather small size:
height of tubes is only 10 – 13 cm.
Golden
tube worm (Phytoserpulopsis chrysus) lives
in symbiosis with yellow-green algae
giving yellowish shade to translucent
feelers. It is the smallest species
of genus – length of body of the
adult worm is about 5 cm. Its tubes
grow basically in horizontal direction,
only their tips rise upwards. At
times limy algae completely cover
them, but it does good to worms:
they live in most restless part of
reef – on the top of valves of shells
of bivalve mollusks, on stones near
to a surface of water. Here the probability
to be eaten by fishes is minimal
for this worm.
Awful teratonema (Teratonema horribilis)
Order: Spirurida (Spirurida)
Family: Filariidae (Filariidae)
Habitat: Meganesia, vicinities of Carpentaria Lake and the bogs surrounding
it.
Mass extinction of species at the end of human epoch rendered the essential
influence to the species connected to them at various stages of life cycle.
First of all it had affected on parasitic species of invertebrates, and helminths
in particular. But restoration of biological variety in early Neocene had resulted
in rough evolution of parasites, including various species of pathogenic organisms.
Parasites of new species of live organisms evolved, arranging their life cycle
to features of connections between the species forming ecosystems in which
the host species lives. One of new species of parasitic worms lives at the
territory of tropical forests of Meganesia. Some kinds of animals connected
between each other by life cycle of this species of worms live in rainforests
of this region.
The representative of nemathod worms, awful teratonema (literally: “the monstrous
string”) lives in blood vessels of large land vertebrates inhabiting areas
near lakes in northern part of Meganesia – brackish Carpentaria Lake and more
salt Arafura Lake. Its final hosts are various species of mammals – mainly
marsupials and artiodactyls (descendants of species introduced by people).
Awful teratonema has pronounced sexual dimorphism. The female of this species
is about 120 mm long at body thickness of less than 1 mm, and male is approximately
twice shorter. Worms of this species have smooth cylindrical body with thin
covers. Adult worms parasitize in blood vessels of mammals, preferring places
with thinner skin. At ungulates they are located in vessels in the field of
neck, near eyes and on breast. Marsupials with thinner skin are infected in
the field of front limbs, on breast and at the lower jaw, and also in a groin
and in the field of brooding pouch. Worms acquire nutrients, absorbing them
by the whole surface of the body, and also exhaust erythrocytes. In thin blood
vessels with slow current of blood the worm is fixed, simply bending inside
the vessel and resting by sides against its walls.
The worm female gives rise to thousands of microscopic larvae daily till the
whole adult life; they are constantly present in blood of the infected animal.
Mosquitoes and other blood-sucking dipters suck blood of the infected animal.
In their organism larvae pass the further development. They migrate from digestive
path of insect to its mouth, and turn to inactive larva of the second generation
in their salivary glands. The blood-sucking insect transfers larvae of the
parasite to organism of the second intermediate host. In life cycle a role
of the second intermediate host is performed by very original inhabitant of
Meganesia – small mammal marsupial
bloodsucker.
Getting in blood of marsupial bloodsucker at the sting of insect, the larvae
of the second generation of awful teratonema live not for long. They do not
harm this intermediate host, and during the development in its organism make
a lot of larvae of the third generation. These larvae settle in cells of mucous
membrane of mouth of the animal. The larva lives in depth of hypoderm, but
infects cells of epithelium with larvae of the fourth generation which stay
in inactive condition inside epithelial cells. The parasite is located under
the tongue and on the internal side of lips of marsupial bloodsucker, forming
traces as small aqueous vesicles, containing larvae of inactive stage. At the
bite the appearing of blood of prey in mouth of this mammal serves as an original
chemical signal for the further development of larvae. They leave mouth epithelium
of marsupial bloodsucker and get in blood of the final host, infecting it.
In blood of large mammals awful teratonema develops without problems. Occasionally
the marsupial bloodsucker attacks on yagil – large species of flightless bird
of Meganesia. In blood of infected yagil at the influence of higher body temperature
of bird worms develop more intensively and grow larger. They stop up a gap
of blood vessels that has rather serious consequences for the infected bird:
occurrence of tumours or tissue necrosis.
Giant
snail planaria (Titanoplana helicivora)
Order: Tricladids (Tricladida)
Family: Toothed planarians (Odontoplanariidae)
Habitat: Central and Eastern Europe, rivers and lakes.

Picture by Biolog
At the boundary of Holocene and Neocene the Earth had gone
through severe congelation, even more severe, than all ones had taken place
before it. This event rendered colossal influence to the nature of the Earth.
The climate became drier and colder, and the amount of rains was strongly reduced.
The congelation rendered serious influence on freshwater reservoirs of Eurasia
and North America. Glaciers had changed character of river current, and because
of drier climate in ice age the part of reservoirs had simply vanished.
Freshwater flora and fauna had suffered in lesser degree, rather than sea ones.
The majority of kinds of invertebrates managed to keep the species variability.
The broken off areas and difference in conditions of inhabiting promoted the
evolution and active speciation. In places where favorable conditions and abundance
of food resources had kept, specialized species evolved. One of the main centers
of speciation in Europe is the Balkan region. In ice age this area appeared
in conditions of favorable enough temperature mode and moderate humidifying.
Mountain areas served as stores of moisture, and from glaciers rivers constantly
flew, filling reservoirs in valleys. A plenty of isolated habitats and remoteness
from the glacier promoted preservation of variability of species of freshwater
fauna.
In Neocene the climate of Balkan became damp due to influence of Fourseas stretched
in northeast. Forests favor to preservation of reservoirs, and surprising species
of live creatures prosper in them. One of new Neocene species is large freshwater
planaria up to 3 – 4 cm long. It lives in coastal thickets of water plants and
shows itself seldom.
By appearance this worm is the typical representative of its kind. The flat
body of giant snail planaria is covered with caustic poisonous slime. Its color
is grey with light cross strips forming “marble” pattern. On front edge of the
body there are two short blunt tentacles, sticking in sides, and small eyes
– 8 ones smaller, than poppy seed by size. Planaria sees badly and vaguely distinguishes
only contours of objects.
This worm is an active predator. Giant snail planaria eats snails, eating them
away from shells. For feeding on such specific protected prey at it the hunting
adaptation had developed. The characteristic feature of planarias is the gullet
supplied with muscles, able to extend and even to live independent life. At
giant snail planaria the gullet is considerably improved in comparison with
its tiny relatives. Due to ring muscular fibers it is able to extend strongly
(up to 10 cm long). On the tip of gullet some pointed corneous denticles grow.
This worm hunts snails very effectively. Having found out suitable snail, the
worm simply pushes the gullet in fauces of its shell and eats snail away alive
entirely – down to the last coil of shell. Certainly, mollusks with operculum
on the shell are more protected from this worm, but at young mollusks of such
species giant snail planaria gnaws through the operculum and also eats them
away.
Such large worm is well protected from attacks of fishes and water beetles due
to the cover of poisonous slime. But nevertheless these worms meet in European
reservoirs not so often – they also have enemies. Larvae of water beetles and
water bugs can attack this planaria. They do not bite it, inevitably receiving
a portion of slime, but simply exhaust the body of worm by proboscis or through
mandibles, not touching its slime. Sometimes water bugs attack on planaria devouring
snail and exhaust semidigested food right from its intestinal cavity, having
pierced the wall of body by proboscis.
Giant snail planaria has interesting feature of sexless breeding: having had
eaten large enough catch, the worm is able to divide spontaneously. The body
of planaria divides across with the help of contractions of longitudinal muscles.
After that each half completes missing organs till some days, using nutrients
of semidigested prey which had preserved in intestines. Thus gullet of planaria
also is separated and lives by its own life – it can creep, contracting muscles,
and reacts to approach of catch. Gullet of planaria attacks small snails and
other invertebrates, obviously preferring soft-bodied ones. It eats them, and
using received nutrients completes on the back end temporarily absent body.
If hunting was unsuccessful, gullet all the same restores the body, using the
nutrients which are accumulated in its tissues. Thus it “dries out” and strongly
decreases in size, but survives and even lives much longer the normal worm of
this species.
Giant snail planaria lives in conditions of seasonal climate. In autumn, when
water becomes colder, at this worm sexual breeding begins. Similarly to all
planarias, this species is hermaphrodite, and two any individuals can fertilize
each other. Eggs of this species develop in slimy cocoons which the animal pastes
to roots of trees or driftwood. Cocoons hibernate, and in spring young individuals
left them. In autumn young worms lay the first clutch, but do not reach the
maximal size. At the second year of life they reach the sizes of adult individual,
and start breeding by division. Life expectancy at giant snail planaria may
reach 5 years.
Burning
flatworm (Nipponoplana tigrina)
Order: Tricladids (Tricladida)
Family: Bipaliids (Bipaliidae)
Habitat: Japan Islands, humid forests of southeast.

Picture by Biolog
Tropical rainforests form the community supporting its own
existence. Moss, wood litter and roots of trees keep water as if a sponge, and
the leaves falling from trees, at once are decomposed and go to circulation
of substances again. The rich forest canopy supports a specific microclimate
– equal and moist. In such conditions life is especially rich, and some water
inhabitants willingly occupy land. Various species of large
ground leeches
settle in forests of South-East Asia, and one species of fishes lives in South
America in wood litter – it is almost blind worm-like
catfish. One more species from the number of recent immigrants from water
lives in tropical forests of Japan – it is a large ground planaria named burning
flatworm because of bright colouring and burning taste of the slime covering
its body.
Burning flatworm is the true giant among planarias – it reaches the length of
30 – 35 cm at width of about 2 cm. It is smaller, than at record by size ground
planarias of Holocene epoch, but all the same it surpasses the length of the
majority of species of ground planarias. Colouring of body at this worm is very
much remembered: it is cross-striped, on red background with orange shade on
edges and in forward part of body the set of cross strips of black color passes.
This large worm imitates the appearance of local tiger
millipede (Bradyjulius rubro-nigrum). Appearance of burning flatworm warns
that the animal is very well protected from an attack. Any animal which will
snap this worm, will feel the burning taste of its slime. Slime will irritate
mucous membranes of predator within several hours, and the memoirs on unpleasant
meeting will be remembered to the aggressor for a long time. Burning flatworm
“maintains the reputation”, having the security common for the species included
to the “ring” with such type of colouring. Besides this worm, tiger millipede
is imitated by one local snake.
But burning flatworm has one more unique way of protection which any of species
from the “ring” does not own. If the body of animal is injured, or any predator
had seized it by paw, burning flatworm can break off its body by voluntary retraction
of muscles to some parts, slipping out from predator’s grip. It does not bring
any serious harm to it – each piece easily regenerates, restoring the lost organs,
and turns to small normal worm.
This planaria lives in leaf litter and hunts earthworms and insects. Gullet
located in the middle of the body, as if lives by independent life, searching
for food irrespectively of direction of movement of the worm. It can extend
to any side to the distance about equal to the body length of a worm. At edges
of the gullet sensitive cells and chemoreceptors assisting in search of prey
are located. Gullet can even dig in friable ground in searches of worms, catching
and swallowing them right in their own hole.
Head of this worm is flat, crescent and two-lobed. Very tiny eyes (this worm
has 10 eyes) are located in line on its front edge. They can distinguish light
from darkness and distinguish contours of large objects – it is quite enough
of it for worm, which gullet searches for prey by smell itself.
Slimy and thin covers of burning flatworm easily lose moisture, therefore burning
flatworm lives only in moist places. This planaria though is ground worm, can
behave as an amphibian: it can dive into cool water of forest streams, rich
in oxygen, and receive oxygen from water through external covers till any time.
It avoids pools with warmed up water, but can catch water animals, using mobile
gullet, which stretches under water. The favourite forage of this worm includes
aquatic snails. At the drought burning flatworm is dug in the ground and contracts
almost to ball, forming around of itself a slimy cover. Sand particles stick
to slime, and, drying up, the cocoon becomes covered with dense crust. After
rains it becomes soggy, worm absorbs moisture from ground, restoring its amount
in organism, and continues to lead usual life.
This animal is a hermaphrodite. After pairing the worm lays cocoons containing
up to 20 eggs in moist ground. After 3 – 4 weeks very small young worms having
as burning slime, as adult ones, leave them.
Enemies of this worm are predatory bugs, which can pierce by proboscis covers
of its body, passing a layer of slime, inject their poisonous saliva rendering
digestive action, and exhaust the dissolved tissues of worm. The adult worm
can escape from a bug, having divided to pieces, only one of which will be exhausted
by the insect, but young worms frequently have no time to make it and perish
after a sting of bug.
Pale
Tyrant Flatworm (Platytyrannus pallidus)
Order: Tricladids (Tricladida)
Family: Land Planarians (Geoplanidae)
Habitat: Damp cave floors and cave waterways within the depths of glacially
produced cave systems, North and South Islands of New Zealand.
Every ecosystem consists of an abundance of prey animals, preyed upon by various
predators, the miniature menagerie living within cave systems is no different.
One of the dominant inveterate predators in the cave systems of New Zealand
is the Pale Tyrant Flatworm, descended from the common New Zealand Flatworm
(Arthurdendyus triangulatus).
This terrestrial flatworm is elongate and flattened in shape, the head comes
to a point, which feels about in order to find its way, and locate prey. Coloration
is pale and opaque, whitish, edges of the body that are thinner are slightly
translucent. Body reaches 15 centimetres long at full extension, when resting,
it will wedge itself under a stone or within a crevice, and wind itself into
a spiral ball.
This worm is able to hunt both on land and underwater, feeding on small crustaceans,
insects, and their eggs, dead animal matter is also eaten. They can remain submerged
for up to half an hour, but must return to land after feeding, breathing is
by diffusion through the body wall. When prey is located, the worm wraps around
it, secreting mucous which contains strong digestive juices, liquefied food
and small pieces are sucked through the mouth into the pharynx to the gut. It
moves around by crawling using muscular contractions, the head moves about in
order for the animal to find its way, prey is located through chemo-reception
and touch. During times of starvation, it is able to digest its own body tissues
for food.
Breeding occurs year round, sexual reproduction occurs after one worm locates
the other by smell, all individuals are hermaphrodites. A single large oblong
egg capsule is laid in a crevice or under a stone. The outside hardens, and
after a few weeks the young emerge as fully formed miniatures of the adults.
Individual worms are also able to reproduce by fission, splitting into two parts
that both grow into separate individuals. Lifespan can be up to 3 years, apart
from those individuals that reproduce by splitting.
This invertebrate species was discovered by Timothy Donald Morris, Adelaide, Australia.
Pelagic
multi-eyed worm (Crystallodiscus polyoculatus)
Order: Polycladids (Polycladida)
Family: Planocerids (Planoceridae)
Habitat: temperate and cold waters of Southern hemisphere.
In plankton there are various species of animals which spend in this community
as a part of life (larval stage), and all life entirely. All planktonic inhabitants
are well adapted to soaring in thickness of water, developing freakish outgrowths,
spikes and flat parts of body which help to keep in thickness of water. Food
predilections of plankton inhabitants are diverse, but the most part of them
are predators of various trophic levels.
Among the planktonic animals living in productive areas of oceans of Southern
hemisphere, there is a pelagic flat worm with wide body of rounded outlines
– pelagic multi-eyed worm. It is rather large species of the group: the length
(and diameter) of the body reaches approximately 10 cm. Along the front line
of the body many tiny eyes (up to 10 – 16 ones) are located, which distinguish
light from darkness and can distinguish contours of large objects. From both
edges a line of eyes is bordered by short triangular tentacles. Body of pelagic
multi-eyed worm is translucent, through it very much ramified intestines penetrating
the whole body are seen. This worm has long extensible gullet looking like proboscis
which grows right in the middle of the belly side of body and is supplied with
a plenty of muscular fibres allowing it to extend, bend and retract.
Being a planktonic animal, this worm is capable to swim actively with the help
of wavy movements of lateral parts of the body combined to bends along an axis
of the body. Speed of such movement can make up to 3 meters per minute. Pelagic
multi-eyed worm is a predator eating various small planktonic animals: larvae
of fishes and cephalopods, worms and crustaceans. Usually this worm passively
hovers in thickness of water, keeping an immovability of the body and resembling
at this time any kind of jellyfish. Its gullet is extended waiting for approach
of suitable prey and makes slow movements in sides. Its edges are supplied with
chemoreceptors, and gullet can search for prey independently of the body of
worm. When the small animal swims up close enough to the tip of gullet, the
worm extends it in its side, trying to seize prey. If the attempt is successful,
pelagic multi-eyed worm pulls prey to itself and swallows it entirely.
In parenchyma between branches of intestines the fat inclusions improving buoyancy
develop. They are visible in the body of worm as numerous white semitransparent
lumps. At well fatten worm they form the pattern repeating the contours of branches
of intestines. The worm starved for a long time spends stocks of fat entirely
and becomes completely transparent.
As at all free-living flat worms, at pelagic multi-eyed worms ability to regeneration
is advanced in great degree. The seized worm spontaneously breaks up to some
pieces. From each piece of body, irrespective of its size, the whole animal
is soon restored. Regeneration proceeds especially quickly at the presence of
stocks of fat in parenchyma.
Pelagic multi-eyed worm is the hermaphrodite; a lot of pairs of masculine and
feminine sexual glands are located at the edge of body. Masculine and feminine
sexual products develop in the body of animal not simultaneously, that excludes
self-fertilization. At this species development is direct – from eggs tiny copies
of adult individual hatch. They at once start to hunt pelagic nanoplankton.
At the age of 4 – 5 months the young ones reach the size of adult individual.
Coelenterates
|
Anchor jellyfish (Anchorotentacula sessilis)
Order: Trachylids (Trachylida)
Family: Anchor jellyfishes (Anchorotentaculidae)
Habitat: Atlantic Ocean, algal fields near New Azora.
Relief of bottom of Atlantic Ocean in combination to currents has reasoned
the occurrence of extensive fields of floating seaweed near the New Azora Island.
Fields of seaweed, quickly expanding and equally quickly dying off, form the
favorable conditions for life of various sea inhabitants. Here they find not
only shelter and food, but also safe place for life: fields of seaweed soften
force of storm.
The majority of inhabitants of algal fields are represented by descendants
of coastal animals. Their ancestors settled at the stage of plankton pelagic
larva, and some larvae had to found the convenient place among floating seaweed,
having given rise to the population of algal fields of New Azora. Some inhabitants
of water thickness also had adapted to life in seaweed. One of these creatures
is the small jellyfish attaching to seaweed. In Holocene epoch jellyfish Lucernaria
led the attached way of life was known. It attached to substratum by dome,
upwards the mouth, and kept radial symmetry. But the Neocene jellyfish attaching
to seaweed differs from that species. At it the radial symmetry characteristic
for coelenterates is lost in some degree. From four tentacles of this jellyfish
one has the function of attaching – with its help the animal holds thalli of
seaweed. For this feature this coelenterate is named anchor jellyfish. Other
three tentacles have kept trapping ability. They are thin, ramified, and also
are able to extend strongly. On their tips urticators (lasso cells) are advanced.
Usually anchor jellyfish freely soars in water, having fixed by attaching tentacle
and having stretched in sides trapping tentacles. This is almost transparent
creature, practically invisible in water. Its body has spherical form with
thick mesogloea through which ramified gastral cavity is visible. The size
of adult anchor jellyfish may reach 10 cm, and the “anchor” tentacle is extended
to one and half meter. The anchor jellyfish belongs to number of most poisonous
inhabitants of ocean. Its food includes pelagic worms, crustaceans and small
fishes.
This creature is almost not able to swim. If the anchor jellyfish casually
tears off from substratum, it motionlessly soars in thickness of water, having
stretched tentacles in sides. When one of them touches seaweed or other substratum,
it is fixed on it and the jellyfish is dragging to substratum.
The attaching tentacle is thicker and longer, rather than trapping ones. On
it muscular fibrillas develop, able to contract in wide degree. When storm
comes, or beside large animal swims, muscular fibrillas contract, and the jellyfish
hides in seaweed thickets, having shrinked in mucous ball. Lasso cells of attaching
tentacle have changed: they had lost lasso and poison, and the sensitive hair
had turned to strong cornificate prickle. The attaching tentacle is ramified
on the end to some branches. The animal twists them around seaweed thallus
and keeps on the spot so. Sometimes casual movement of large animal or storm
wave breaks off attaching tentacle and tears off the jellyfish from substratum.
In this case the animal attaches to substratum by any of kept tentacles, and
it gradually turns to attaching one. And regenerating attaching tentacle becomes
trapping one.
Breeding of jellyfishes usually includes alternation of generations: polypoid
and medusoid stages. The anchor jellyfish belongs to species at which the stage
of polyp is lost a long time ago. Jellyfishes of this species lay eggs, from
which pelagic planula larva hatches. Growing up, it at first leads mobile life
under the carpet of seaweed, where there is a lot of food and few plankton
predatory animals. Having reached the diameter about 3 cm, young jellyfish
passes to sedentary habit of life. Life expectancy of this species does not
exceed three months.